CN107286146B - 4-aminopyrimidine derivatives as adenosine A2A receptor antagonists and uses thereof - Google Patents
4-aminopyrimidine derivatives as adenosine A2A receptor antagonists and uses thereof Download PDFInfo
- Publication number
- CN107286146B CN107286146B CN201710542349.1A CN201710542349A CN107286146B CN 107286146 B CN107286146 B CN 107286146B CN 201710542349 A CN201710542349 A CN 201710542349A CN 107286146 B CN107286146 B CN 107286146B
- Authority
- CN
- China
- Prior art keywords
- adenosine
- receptor
- compound
- cancer
- aminopyrimidine
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 150000005007 4-aminopyrimidines Chemical class 0.000 title claims abstract description 16
- 229940123702 Adenosine A2a receptor antagonist Drugs 0.000 title 1
- 239000002467 adenosine A2a receptor antagonist Substances 0.000 title 1
- OIRDTQYFTABQOQ-KQYNXXCUSA-N adenosine Chemical compound C1=NC=2C(N)=NC=NC=2N1[C@@H]1O[C@H](CO)[C@@H](O)[C@H]1O OIRDTQYFTABQOQ-KQYNXXCUSA-N 0.000 claims abstract description 126
- 150000001875 compounds Chemical class 0.000 claims abstract description 104
- 239000002126 C01EB10 - Adenosine Substances 0.000 claims abstract description 63
- 229960005305 adenosine Drugs 0.000 claims abstract description 63
- 239000002464 receptor antagonist Substances 0.000 claims abstract description 35
- 206010028980 Neoplasm Diseases 0.000 claims abstract description 25
- 229940044551 receptor antagonist Drugs 0.000 claims abstract description 22
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 claims abstract description 15
- 201000010099 disease Diseases 0.000 claims abstract description 14
- 230000004770 neurodegeneration Effects 0.000 claims abstract description 14
- OYRRZWATULMEPF-UHFFFAOYSA-N pyrimidin-4-amine Chemical class NC1=CC=NC=N1 OYRRZWATULMEPF-UHFFFAOYSA-N 0.000 claims abstract description 14
- 208000015122 neurodegenerative disease Diseases 0.000 claims abstract description 13
- 208000011580 syndromic disease Diseases 0.000 claims abstract description 13
- 208000027776 Extrapyramidal disease Diseases 0.000 claims abstract description 12
- 125000002971 oxazolyl group Chemical group 0.000 claims abstract description 12
- 208000019901 Anxiety disease Diseases 0.000 claims abstract description 11
- 206010012601 diabetes mellitus Diseases 0.000 claims abstract description 10
- 229910052736 halogen Inorganic materials 0.000 claims abstract description 10
- 208000013403 hyperactivity Diseases 0.000 claims abstract description 10
- 125000003226 pyrazolyl group Chemical group 0.000 claims abstract description 10
- 208000019116 sleep disease Diseases 0.000 claims abstract description 10
- 150000002367 halogens Chemical class 0.000 claims abstract description 9
- 125000000719 pyrrolidinyl group Chemical group 0.000 claims abstract description 9
- 230000036506 anxiety Effects 0.000 claims abstract description 7
- 125000004093 cyano group Chemical group *C#N 0.000 claims abstract description 6
- 238000002360 preparation method Methods 0.000 claims description 32
- 238000011282 treatment Methods 0.000 claims description 16
- 239000003814 drug Substances 0.000 claims description 12
- 206010058467 Lung neoplasm malignant Diseases 0.000 claims description 7
- 201000005202 lung cancer Diseases 0.000 claims description 7
- 208000020816 lung neoplasm Diseases 0.000 claims description 7
- 239000008194 pharmaceutical composition Substances 0.000 claims description 5
- 229940079593 drug Drugs 0.000 claims description 4
- 201000001441 melanoma Diseases 0.000 claims description 4
- 208000002154 non-small cell lung carcinoma Diseases 0.000 claims description 4
- 208000029729 tumor suppressor gene on chromosome 11 Diseases 0.000 claims description 4
- 206010005003 Bladder cancer Diseases 0.000 claims description 3
- 206010006187 Breast cancer Diseases 0.000 claims description 3
- 208000026310 Breast neoplasm Diseases 0.000 claims description 3
- 206010009944 Colon cancer Diseases 0.000 claims description 3
- 208000001333 Colorectal Neoplasms Diseases 0.000 claims description 3
- 206010033128 Ovarian cancer Diseases 0.000 claims description 3
- 206010061535 Ovarian neoplasm Diseases 0.000 claims description 3
- 206010061902 Pancreatic neoplasm Diseases 0.000 claims description 3
- 206010060862 Prostate cancer Diseases 0.000 claims description 3
- 208000000236 Prostatic Neoplasms Diseases 0.000 claims description 3
- 208000006265 Renal cell carcinoma Diseases 0.000 claims description 3
- 208000005718 Stomach Neoplasms Diseases 0.000 claims description 3
- 208000007097 Urinary Bladder Neoplasms Diseases 0.000 claims description 3
- 208000006990 cholangiocarcinoma Diseases 0.000 claims description 3
- 206010017758 gastric cancer Diseases 0.000 claims description 3
- 208000005017 glioblastoma Diseases 0.000 claims description 3
- 201000010536 head and neck cancer Diseases 0.000 claims description 3
- 208000014829 head and neck neoplasm Diseases 0.000 claims description 3
- 206010073071 hepatocellular carcinoma Diseases 0.000 claims description 3
- 231100000844 hepatocellular carcinoma Toxicity 0.000 claims description 3
- 208000015486 malignant pancreatic neoplasm Diseases 0.000 claims description 3
- 201000002528 pancreatic cancer Diseases 0.000 claims description 3
- 208000008443 pancreatic carcinoma Diseases 0.000 claims description 3
- 239000000546 pharmaceutical excipient Substances 0.000 claims description 3
- 201000011549 stomach cancer Diseases 0.000 claims description 3
- 201000005112 urinary bladder cancer Diseases 0.000 claims description 3
- 229940000425 combination drug Drugs 0.000 claims 1
- 230000003042 antagnostic effect Effects 0.000 abstract description 7
- 125000005843 halogen group Chemical group 0.000 abstract description 5
- 125000001715 oxadiazolyl group Chemical group 0.000 abstract description 5
- 125000001425 triazolyl group Chemical group 0.000 abstract description 5
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 abstract description 5
- 108020003175 receptors Proteins 0.000 description 50
- 102000005962 receptors Human genes 0.000 description 50
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 45
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 36
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 36
- IAZDPXIOMUYVGZ-WFGJKAKNSA-N Dimethyl sulfoxide Chemical compound [2H]C([2H])([2H])S(=O)C([2H])([2H])[2H] IAZDPXIOMUYVGZ-WFGJKAKNSA-N 0.000 description 30
- PCLIMKBDDGJMGD-UHFFFAOYSA-N N-bromosuccinimide Chemical compound BrN1C(=O)CCC1=O PCLIMKBDDGJMGD-UHFFFAOYSA-N 0.000 description 24
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 21
- 238000006243 chemical reaction Methods 0.000 description 20
- 238000005160 1H NMR spectroscopy Methods 0.000 description 15
- RWRDLPDLKQPQOW-UHFFFAOYSA-N Pyrrolidine Chemical compound C1CCNC1 RWRDLPDLKQPQOW-UHFFFAOYSA-N 0.000 description 15
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 14
- 210000004027 cell Anatomy 0.000 description 13
- 239000000243 solution Substances 0.000 description 13
- WFDIJRYMOXRFFG-UHFFFAOYSA-N Acetic anhydride Chemical compound CC(=O)OC(C)=O WFDIJRYMOXRFFG-UHFFFAOYSA-N 0.000 description 12
- 230000000694 effects Effects 0.000 description 12
- -1 pyrazole compound Chemical class 0.000 description 12
- 230000027455 binding Effects 0.000 description 11
- 230000002829 reductive effect Effects 0.000 description 11
- WTKZEGDFNFYCGP-UHFFFAOYSA-N Pyrazole Chemical compound C=1C=NNC=1 WTKZEGDFNFYCGP-UHFFFAOYSA-N 0.000 description 10
- VYFYYTLLBUKUHU-UHFFFAOYSA-N dopamine Chemical compound NCCC1=CC=C(O)C(O)=C1 VYFYYTLLBUKUHU-UHFFFAOYSA-N 0.000 description 10
- 239000002904 solvent Substances 0.000 description 10
- 241000700159 Rattus Species 0.000 description 9
- 238000000034 method Methods 0.000 description 9
- 239000002287 radioligand Substances 0.000 description 9
- JRNVZBWKYDBUCA-UHFFFAOYSA-N N-chlorosuccinimide Chemical group ClN1C(=O)CCC1=O JRNVZBWKYDBUCA-UHFFFAOYSA-N 0.000 description 8
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 8
- FJDQFPXHSGXQBY-UHFFFAOYSA-L caesium carbonate Chemical compound [Cs+].[Cs+].[O-]C([O-])=O FJDQFPXHSGXQBY-UHFFFAOYSA-L 0.000 description 8
- 229910000024 caesium carbonate Inorganic materials 0.000 description 8
- 238000012360 testing method Methods 0.000 description 8
- 230000005764 inhibitory process Effects 0.000 description 7
- 239000000203 mixture Substances 0.000 description 7
- 239000012044 organic layer Substances 0.000 description 7
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 6
- PAOANWZGLPPROA-RQXXJAGISA-N CGS-21680 Chemical compound O[C@@H]1[C@H](O)[C@@H](C(=O)NCC)O[C@H]1N1C2=NC(NCCC=3C=CC(CCC(O)=O)=CC=3)=NC(N)=C2N=C1 PAOANWZGLPPROA-RQXXJAGISA-N 0.000 description 6
- 239000005557 antagonist Substances 0.000 description 6
- 230000001413 cellular effect Effects 0.000 description 6
- KDLHZDBZIXYQEI-UHFFFAOYSA-N palladium Substances [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 6
- 239000000047 product Substances 0.000 description 6
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical class O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 6
- 101150049660 DRD2 gene Proteins 0.000 description 5
- ZCQWOFVYLHDMMC-UHFFFAOYSA-N Oxazole Chemical compound C1=COC=N1 ZCQWOFVYLHDMMC-UHFFFAOYSA-N 0.000 description 5
- 208000018737 Parkinson disease Diseases 0.000 description 5
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 5
- 230000008485 antagonism Effects 0.000 description 5
- 229960003638 dopamine Drugs 0.000 description 5
- 239000012528 membrane Substances 0.000 description 5
- 210000002381 plasma Anatomy 0.000 description 5
- 239000000018 receptor agonist Substances 0.000 description 5
- 238000010898 silica gel chromatography Methods 0.000 description 5
- 208000010110 spontaneous platelet aggregation Diseases 0.000 description 5
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 4
- 102000009346 Adenosine receptors Human genes 0.000 description 4
- 108050000203 Adenosine receptors Proteins 0.000 description 4
- 241000699670 Mus sp. Species 0.000 description 4
- MZRVEZGGRBJDDB-UHFFFAOYSA-N N-Butyllithium Chemical compound [Li]CCCC MZRVEZGGRBJDDB-UHFFFAOYSA-N 0.000 description 4
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 4
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 4
- 230000004913 activation Effects 0.000 description 4
- 238000003556 assay Methods 0.000 description 4
- 230000006399 behavior Effects 0.000 description 4
- ZPUCINDJVBIVPJ-LJISPDSOSA-N cocaine Chemical compound O([C@H]1C[C@@H]2CC[C@@H](N2C)[C@H]1C(=O)OC)C(=O)C1=CC=CC=C1 ZPUCINDJVBIVPJ-LJISPDSOSA-N 0.000 description 4
- 230000033001 locomotion Effects 0.000 description 4
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 4
- 230000004044 response Effects 0.000 description 4
- 238000010561 standard procedure Methods 0.000 description 4
- 238000003756 stirring Methods 0.000 description 4
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 4
- 210000004881 tumor cell Anatomy 0.000 description 4
- KURQKNMKCGYWRJ-HNNXBMFYSA-N 7-(5-methylfuran-2-yl)-3-[[6-[[(3s)-oxolan-3-yl]oxymethyl]pyridin-2-yl]methyl]triazolo[4,5-d]pyrimidin-5-amine Chemical compound O1C(C)=CC=C1C1=NC(N)=NC2=C1N=NN2CC1=CC=CC(CO[C@@H]2COCC2)=N1 KURQKNMKCGYWRJ-HNNXBMFYSA-N 0.000 description 3
- 208000012661 Dyskinesia Diseases 0.000 description 3
- 208000023105 Huntington disease Diseases 0.000 description 3
- WTDRDQBEARUVNC-LURJTMIESA-N L-DOPA Chemical compound OC(=O)[C@@H](N)CC1=CC=C(O)C(O)=C1 WTDRDQBEARUVNC-LURJTMIESA-N 0.000 description 3
- WTDRDQBEARUVNC-UHFFFAOYSA-N L-Dopa Natural products OC(=O)C(N)CC1=CC=C(O)C(O)=C1 WTDRDQBEARUVNC-UHFFFAOYSA-N 0.000 description 3
- UTLPKQYUXOEJIL-UHFFFAOYSA-N LSM-3822 Chemical compound N1=CC=2C3=NC(C=4OC=CC=4)=NN3C(N)=NC=2N1CCC1=CC=CC=C1 UTLPKQYUXOEJIL-UHFFFAOYSA-N 0.000 description 3
- 206010034010 Parkinsonism Diseases 0.000 description 3
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 3
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 3
- 230000009471 action Effects 0.000 description 3
- 239000000164 antipsychotic agent Substances 0.000 description 3
- 210000004227 basal ganglia Anatomy 0.000 description 3
- 201000011510 cancer Diseases 0.000 description 3
- 210000003169 central nervous system Anatomy 0.000 description 3
- 239000003153 chemical reaction reagent Substances 0.000 description 3
- 230000006735 deficit Effects 0.000 description 3
- 239000003136 dopamine receptor stimulating agent Substances 0.000 description 3
- 230000003291 dopaminomimetic effect Effects 0.000 description 3
- 125000000623 heterocyclic group Chemical group 0.000 description 3
- 230000002519 immonomodulatory effect Effects 0.000 description 3
- 229960004502 levodopa Drugs 0.000 description 3
- 210000001577 neostriatum Anatomy 0.000 description 3
- 229910052757 nitrogen Inorganic materials 0.000 description 3
- 230000001575 pathological effect Effects 0.000 description 3
- 230000037361 pathway Effects 0.000 description 3
- 230000035790 physiological processes and functions Effects 0.000 description 3
- NROKBHXJSPEDAR-UHFFFAOYSA-M potassium fluoride Chemical compound [F-].[K+] NROKBHXJSPEDAR-UHFFFAOYSA-M 0.000 description 3
- 239000002244 precipitate Substances 0.000 description 3
- 230000002035 prolonged effect Effects 0.000 description 3
- 229940044601 receptor agonist Drugs 0.000 description 3
- 230000009467 reduction Effects 0.000 description 3
- 230000007958 sleep Effects 0.000 description 3
- 238000005406 washing Methods 0.000 description 3
- PLRACCBDVIHHLZ-UHFFFAOYSA-N 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine Chemical compound C1N(C)CCC(C=2C=CC=CC=2)=C1 PLRACCBDVIHHLZ-UHFFFAOYSA-N 0.000 description 2
- MEKOFIRRDATTAG-UHFFFAOYSA-N 2,2,5,8-tetramethyl-3,4-dihydrochromen-6-ol Chemical compound C1CC(C)(C)OC2=C1C(C)=C(O)C=C2C MEKOFIRRDATTAG-UHFFFAOYSA-N 0.000 description 2
- NHQDETIJWKXCTC-UHFFFAOYSA-N 3-chloroperbenzoic acid Chemical compound OOC(=O)C1=CC=CC(Cl)=C1 NHQDETIJWKXCTC-UHFFFAOYSA-N 0.000 description 2
- ATTAJQLEJJIUAA-UHFFFAOYSA-N 6-chloro-2-methylsulfinylpyrimidin-4-amine Chemical compound CS(=O)C1=NC(N)=CC(Cl)=N1 ATTAJQLEJJIUAA-UHFFFAOYSA-N 0.000 description 2
- 206010001540 Akathisia Diseases 0.000 description 2
- 208000024827 Alzheimer disease Diseases 0.000 description 2
- FFBDFADSZUINTG-UHFFFAOYSA-N DPCPX Chemical compound N1C=2C(=O)N(CCC)C(=O)N(CCC)C=2N=C1C1CCCC1 FFBDFADSZUINTG-UHFFFAOYSA-N 0.000 description 2
- LCGLNKUTAGEVQW-UHFFFAOYSA-N Dimethyl ether Chemical compound COC LCGLNKUTAGEVQW-UHFFFAOYSA-N 0.000 description 2
- 208000014094 Dystonic disease Diseases 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 2
- 108010052285 Membrane Proteins Proteins 0.000 description 2
- 102000018697 Membrane Proteins Human genes 0.000 description 2
- 241001465754 Metazoa Species 0.000 description 2
- 208000016285 Movement disease Diseases 0.000 description 2
- CBENFWSGALASAD-UHFFFAOYSA-N Ozone Chemical compound [O-][O+]=O CBENFWSGALASAD-UHFFFAOYSA-N 0.000 description 2
- 208000001431 Psychomotor Agitation Diseases 0.000 description 2
- CZPWVGJYEJSRLH-UHFFFAOYSA-N Pyrimidine Chemical compound C1=CN=CN=C1 CZPWVGJYEJSRLH-UHFFFAOYSA-N 0.000 description 2
- WQDUMFSSJAZKTM-UHFFFAOYSA-N Sodium methoxide Chemical compound [Na+].[O-]C WQDUMFSSJAZKTM-UHFFFAOYSA-N 0.000 description 2
- 210000001744 T-lymphocyte Anatomy 0.000 description 2
- 208000003721 Triple Negative Breast Neoplasms Diseases 0.000 description 2
- 238000010521 absorption reaction Methods 0.000 description 2
- 102000030621 adenylate cyclase Human genes 0.000 description 2
- 108060000200 adenylate cyclase Proteins 0.000 description 2
- 230000002411 adverse Effects 0.000 description 2
- 238000004220 aggregation Methods 0.000 description 2
- 238000004458 analytical method Methods 0.000 description 2
- 230000001430 anti-depressive effect Effects 0.000 description 2
- 230000000702 anti-platelet effect Effects 0.000 description 2
- 239000003146 anticoagulant agent Substances 0.000 description 2
- 229940125681 anticonvulsant agent Drugs 0.000 description 2
- 239000001961 anticonvulsive agent Substances 0.000 description 2
- 239000000935 antidepressant agent Substances 0.000 description 2
- 229940005513 antidepressants Drugs 0.000 description 2
- 229940005529 antipsychotics Drugs 0.000 description 2
- 230000004071 biological effect Effects 0.000 description 2
- 230000015572 biosynthetic process Effects 0.000 description 2
- 210000004369 blood Anatomy 0.000 description 2
- 239000008280 blood Substances 0.000 description 2
- 210000001772 blood platelet Anatomy 0.000 description 2
- 230000031709 bromination Effects 0.000 description 2
- 238000005893 bromination reaction Methods 0.000 description 2
- 238000005660 chlorination reaction Methods 0.000 description 2
- 229910052801 chlorine Inorganic materials 0.000 description 2
- 239000000460 chlorine Substances 0.000 description 2
- 229960003920 cocaine Drugs 0.000 description 2
- 208000029742 colonic neoplasm Diseases 0.000 description 2
- DOBRDRYODQBAMW-UHFFFAOYSA-N copper(i) cyanide Chemical compound [Cu+].N#[C-] DOBRDRYODQBAMW-UHFFFAOYSA-N 0.000 description 2
- 230000008878 coupling Effects 0.000 description 2
- 238000010168 coupling process Methods 0.000 description 2
- 238000005859 coupling reaction Methods 0.000 description 2
- 239000012043 crude product Substances 0.000 description 2
- 230000001419 dependent effect Effects 0.000 description 2
- 208000010118 dystonia Diseases 0.000 description 2
- 238000002474 experimental method Methods 0.000 description 2
- 230000002349 favourable effect Effects 0.000 description 2
- 229910052731 fluorine Inorganic materials 0.000 description 2
- 230000006870 function Effects 0.000 description 2
- BTCSSZJGUNDROE-UHFFFAOYSA-N gamma-aminobutyric acid Chemical compound NCCCC(O)=O BTCSSZJGUNDROE-UHFFFAOYSA-N 0.000 description 2
- 210000001905 globus pallidus Anatomy 0.000 description 2
- 238000010438 heat treatment Methods 0.000 description 2
- 210000002865 immune cell Anatomy 0.000 description 2
- 238000000338 in vitro Methods 0.000 description 2
- 230000002401 inhibitory effect Effects 0.000 description 2
- 208000014674 injury Diseases 0.000 description 2
- 230000003993 interaction Effects 0.000 description 2
- 230000002147 killing effect Effects 0.000 description 2
- 238000011813 knockout mouse model Methods 0.000 description 2
- 239000003446 ligand Substances 0.000 description 2
- 210000002540 macrophage Anatomy 0.000 description 2
- 230000002503 metabolic effect Effects 0.000 description 2
- 239000011259 mixed solution Substances 0.000 description 2
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 2
- 210000000822 natural killer cell Anatomy 0.000 description 2
- 230000000926 neurological effect Effects 0.000 description 2
- 230000009871 nonspecific binding Effects 0.000 description 2
- DIVDFFZHCJEHGG-UHFFFAOYSA-N oxidopamine Chemical compound NCCC1=CC(O)=C(O)C=C1O DIVDFFZHCJEHGG-UHFFFAOYSA-N 0.000 description 2
- NFHFRUOZVGFOOS-UHFFFAOYSA-N palladium;triphenylphosphane Chemical compound [Pd].C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 NFHFRUOZVGFOOS-UHFFFAOYSA-N 0.000 description 2
- 230000000737 periodic effect Effects 0.000 description 2
- 229910052698 phosphorus Inorganic materials 0.000 description 2
- 239000013612 plasmid Substances 0.000 description 2
- 229910000027 potassium carbonate Inorganic materials 0.000 description 2
- 230000003389 potentiating effect Effects 0.000 description 2
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 2
- 238000010992 reflux Methods 0.000 description 2
- UIIMBOGNXHQVGW-UHFFFAOYSA-M sodium bicarbonate Substances [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 2
- 229910000029 sodium carbonate Inorganic materials 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- 238000006467 substitution reaction Methods 0.000 description 2
- 239000000725 suspension Substances 0.000 description 2
- 208000024891 symptom Diseases 0.000 description 2
- 150000003852 triazoles Chemical group 0.000 description 2
- MWKJTNBSKNUMFN-UHFFFAOYSA-N trifluoromethyltrimethylsilane Chemical compound C[Si](C)(C)C(F)(F)F MWKJTNBSKNUMFN-UHFFFAOYSA-N 0.000 description 2
- 208000022679 triple-negative breast carcinoma Diseases 0.000 description 2
- OGNSCSPNOLGXSM-UHFFFAOYSA-N (+/-)-DABA Natural products NCCC(N)C(O)=O OGNSCSPNOLGXSM-UHFFFAOYSA-N 0.000 description 1
- RIRGCFBBHQEQQH-SSFGXONLSA-N (-)-n6-(2-phenylisopropyl)adenosine Chemical compound C([C@@H](C)NC=1C=2N=CN(C=2N=CN=1)[C@H]1[C@@H]([C@H](O)[C@@H](CO)O1)O)C1=CC=CC=C1 RIRGCFBBHQEQQH-SSFGXONLSA-N 0.000 description 1
- UKSZBOKPHAQOMP-SVLSSHOZSA-N (1e,4e)-1,5-diphenylpenta-1,4-dien-3-one;palladium Chemical compound [Pd].C=1C=CC=CC=1\C=C\C(=O)\C=C\C1=CC=CC=C1.C=1C=CC=CC=1\C=C\C(=O)\C=C\C1=CC=CC=C1 UKSZBOKPHAQOMP-SVLSSHOZSA-N 0.000 description 1
- IVTMXOXVAHXCHI-YXLMWLKOSA-N (2s)-2-amino-3-(3,4-dihydroxyphenyl)propanoic acid;(2s)-3-(3,4-dihydroxyphenyl)-2-hydrazinyl-2-methylpropanoic acid Chemical compound OC(=O)[C@@H](N)CC1=CC=C(O)C(O)=C1.NN[C@@](C(O)=O)(C)CC1=CC=C(O)C(O)=C1 IVTMXOXVAHXCHI-YXLMWLKOSA-N 0.000 description 1
- KWTSXDURSIMDCE-QMMMGPOBSA-N (S)-amphetamine Chemical compound C[C@H](N)CC1=CC=CC=C1 KWTSXDURSIMDCE-QMMMGPOBSA-N 0.000 description 1
- QWENRTYMTSOGBR-UHFFFAOYSA-N 1H-1,2,3-Triazole Chemical compound C=1C=NNN=1 QWENRTYMTSOGBR-UHFFFAOYSA-N 0.000 description 1
- SVUOLADPCWQTTE-UHFFFAOYSA-N 1h-1,2-benzodiazepine Chemical class N1N=CC=CC2=CC=CC=C12 SVUOLADPCWQTTE-UHFFFAOYSA-N 0.000 description 1
- UPVBKNZVOJNQKE-UHFFFAOYSA-N 2,6-dichloropyrimidin-4-amine Chemical compound NC1=CC(Cl)=NC(Cl)=N1 UPVBKNZVOJNQKE-UHFFFAOYSA-N 0.000 description 1
- JKMHFZQWWAIEOD-UHFFFAOYSA-N 2-[4-(2-hydroxyethyl)piperazin-1-yl]ethanesulfonic acid Chemical class OCC[NH+]1CCN(CCS([O-])(=O)=O)CC1 JKMHFZQWWAIEOD-UHFFFAOYSA-N 0.000 description 1
- GETQKLUOZMYHGE-UHFFFAOYSA-N 2-[[3-(3,6-dichlorocarbazol-9-yl)-2-hydroxypropyl]amino]-2-(hydroxymethyl)propane-1,3-diol Chemical compound ClC1=CC=C2N(CC(O)CNC(CO)(CO)CO)C3=CC=C(Cl)C=C3C2=C1 GETQKLUOZMYHGE-UHFFFAOYSA-N 0.000 description 1
- LWXNQQIVUUSCSL-UHFFFAOYSA-N 2-amino-2-(hydroxymethyl)propane-1,3-diol trihydrochloride Chemical compound Cl.Cl.Cl.OCC(N)(CO)CO LWXNQQIVUUSCSL-UHFFFAOYSA-N 0.000 description 1
- JHFIINJNJBFZRV-UHFFFAOYSA-N 5-bromo-6-chloro-2-(1,3-oxazol-2-yl)pyrimidin-4-amine Chemical compound BrC=1C(=NC(=NC=1Cl)C=1OC=CN=1)N JHFIINJNJBFZRV-UHFFFAOYSA-N 0.000 description 1
- OKCBVYPRVWJFRT-UHFFFAOYSA-N 5-bromo-6-chloro-2-methylsulfinylpyrimidin-4-amine Chemical compound CS(=O)C1=NC(N)=C(Br)C(Cl)=N1 OKCBVYPRVWJFRT-UHFFFAOYSA-N 0.000 description 1
- ISUXMAHVLFRZQU-UHFFFAOYSA-N 6-chloro-2-methylsulfanylpyrimidin-4-amine Chemical compound CSC1=NC(N)=CC(Cl)=N1 ISUXMAHVLFRZQU-UHFFFAOYSA-N 0.000 description 1
- 206010001541 Akinesia Diseases 0.000 description 1
- 201000001320 Atherosclerosis Diseases 0.000 description 1
- 206010003658 Atrial Fibrillation Diseases 0.000 description 1
- 208000006096 Attention Deficit Disorder with Hyperactivity Diseases 0.000 description 1
- 208000036864 Attention deficit/hyperactivity disease Diseases 0.000 description 1
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 1
- 208000009132 Catalepsy Diseases 0.000 description 1
- 241001456553 Chanodichthys dabryi Species 0.000 description 1
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 1
- 206010008748 Chorea Diseases 0.000 description 1
- 241001415939 Corvus Species 0.000 description 1
- 206010061818 Disease progression Diseases 0.000 description 1
- 208000007590 Disorders of Excessive Somnolence Diseases 0.000 description 1
- 102000004980 Dopamine D2 Receptors Human genes 0.000 description 1
- 108090001111 Dopamine D2 Receptors Proteins 0.000 description 1
- 229940098778 Dopamine receptor agonist Drugs 0.000 description 1
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 1
- 108091006027 G proteins Proteins 0.000 description 1
- 108090000045 G-Protein-Coupled Receptors Proteins 0.000 description 1
- 102000003688 G-Protein-Coupled Receptors Human genes 0.000 description 1
- 102000030782 GTP binding Human genes 0.000 description 1
- 108091000058 GTP-Binding Proteins 0.000 description 1
- 108091006065 Gs proteins Proteins 0.000 description 1
- 241000282412 Homo Species 0.000 description 1
- 208000000269 Hyperkinesis Diseases 0.000 description 1
- 206010021143 Hypoxia Diseases 0.000 description 1
- 206010061218 Inflammation Diseases 0.000 description 1
- 241000581650 Ivesia Species 0.000 description 1
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 description 1
- 206010025323 Lymphomas Diseases 0.000 description 1
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 1
- 241000124008 Mammalia Species 0.000 description 1
- 208000037273 Pathologic Processes Diseases 0.000 description 1
- CXOFVDLJLONNDW-UHFFFAOYSA-N Phenytoin Chemical compound N1C(=O)NC(=O)C1(C=1C=CC=CC=1)C1=CC=CC=C1 CXOFVDLJLONNDW-UHFFFAOYSA-N 0.000 description 1
- 208000005793 Restless legs syndrome Diseases 0.000 description 1
- 241000283984 Rodentia Species 0.000 description 1
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical class [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 1
- 206010041349 Somnolence Diseases 0.000 description 1
- 208000005392 Spasm Diseases 0.000 description 1
- FZWLAAWBMGSTSO-UHFFFAOYSA-N Thiazole Chemical compound C1=CSC=N1 FZWLAAWBMGSTSO-UHFFFAOYSA-N 0.000 description 1
- 102000002938 Thrombospondin Human genes 0.000 description 1
- 108060008245 Thrombospondin Proteins 0.000 description 1
- 206010044565 Tremor Diseases 0.000 description 1
- 229940123445 Tricyclic antidepressant Drugs 0.000 description 1
- 206010047853 Waxy flexibility Diseases 0.000 description 1
- 208000027418 Wounds and injury Diseases 0.000 description 1
- VKIJXFIYBAYHOE-VOTSOKGWSA-N [(e)-2-phenylethenyl]boronic acid Chemical compound OB(O)\C=C\C1=CC=CC=C1 VKIJXFIYBAYHOE-VOTSOKGWSA-N 0.000 description 1
- OIPILFWXSMYKGL-UHFFFAOYSA-N acetylcholine Chemical compound CC(=O)OCC[N+](C)(C)C OIPILFWXSMYKGL-UHFFFAOYSA-N 0.000 description 1
- 229960004373 acetylcholine Drugs 0.000 description 1
- 230000003213 activating effect Effects 0.000 description 1
- 239000002598 adenosine A1 receptor antagonist Substances 0.000 description 1
- 230000004520 agglutination Effects 0.000 description 1
- 230000002776 aggregation Effects 0.000 description 1
- 238000013019 agitation Methods 0.000 description 1
- 239000000556 agonist Substances 0.000 description 1
- 230000001270 agonistic effect Effects 0.000 description 1
- 230000004075 alteration Effects 0.000 description 1
- 229940025084 amphetamine Drugs 0.000 description 1
- 230000033115 angiogenesis Effects 0.000 description 1
- 230000000561 anti-psychotic effect Effects 0.000 description 1
- 208000006673 asthma Diseases 0.000 description 1
- 208000015802 attention deficit-hyperactivity disease Diseases 0.000 description 1
- 210000003719 b-lymphocyte Anatomy 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 229960000911 benserazide Drugs 0.000 description 1
- BNQDCRGUHNALGH-UHFFFAOYSA-N benserazide Chemical compound OCC(N)C(=O)NNCC1=CC=C(O)C(O)=C1O BNQDCRGUHNALGH-UHFFFAOYSA-N 0.000 description 1
- 230000002146 bilateral effect Effects 0.000 description 1
- 210000004204 blood vessel Anatomy 0.000 description 1
- 230000009045 body homeostasis Effects 0.000 description 1
- KGBXLFKZBHKPEV-UHFFFAOYSA-N boric acid Chemical compound OB(O)O KGBXLFKZBHKPEV-UHFFFAOYSA-N 0.000 description 1
- 239000004327 boric acid Substances 0.000 description 1
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 1
- 229910052794 bromium Inorganic materials 0.000 description 1
- 239000000872 buffer Substances 0.000 description 1
- FFGPTBGBLSHEPO-UHFFFAOYSA-N carbamazepine Chemical compound C1=CC2=CC=CC=C2N(C(=O)N)C2=CC=CC=C21 FFGPTBGBLSHEPO-UHFFFAOYSA-N 0.000 description 1
- 229960000623 carbamazepine Drugs 0.000 description 1
- 210000000748 cardiovascular system Anatomy 0.000 description 1
- 230000005779 cell damage Effects 0.000 description 1
- 208000037887 cell injury Diseases 0.000 description 1
- 230000007969 cellular immunity Effects 0.000 description 1
- 210000004978 chinese hamster ovary cell Anatomy 0.000 description 1
- 125000001309 chloro group Chemical group Cl* 0.000 description 1
- 208000012601 choreatic disease Diseases 0.000 description 1
- 238000004587 chromatography analysis Methods 0.000 description 1
- 230000001684 chronic effect Effects 0.000 description 1
- 208000037976 chronic inflammation Diseases 0.000 description 1
- 208000037893 chronic inflammatory disorder Diseases 0.000 description 1
- 230000001149 cognitive effect Effects 0.000 description 1
- 230000003920 cognitive function Effects 0.000 description 1
- 230000001276 controlling effect Effects 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- 231100000433 cytotoxic Toxicity 0.000 description 1
- 230000001472 cytotoxic effect Effects 0.000 description 1
- 230000006378 damage Effects 0.000 description 1
- 230000003111 delayed effect Effects 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 210000002249 digestive system Anatomy 0.000 description 1
- 230000005750 disease progression Effects 0.000 description 1
- 208000037765 diseases and disorders Diseases 0.000 description 1
- MWEQTWJABOLLOS-UHFFFAOYSA-L disodium;[[[5-(6-aminopurin-9-yl)-3,4-dihydroxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-oxidophosphoryl] hydrogen phosphate;trihydrate Chemical compound O.O.O.[Na+].[Na+].C1=NC=2C(N)=NC=NC=2N1C1OC(COP(O)(=O)OP([O-])(=O)OP(O)([O-])=O)C(O)C1O MWEQTWJABOLLOS-UHFFFAOYSA-L 0.000 description 1
- 208000035475 disorder Diseases 0.000 description 1
- 238000010494 dissociation reaction Methods 0.000 description 1
- 230000005593 dissociations Effects 0.000 description 1
- 230000009429 distress Effects 0.000 description 1
- 229940052760 dopamine agonists Drugs 0.000 description 1
- 231100000673 dose–response relationship Toxicity 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 230000000142 dyskinetic effect Effects 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 230000017188 evasion or tolerance of host immune response Effects 0.000 description 1
- 210000000744 eyelid Anatomy 0.000 description 1
- 239000011790 ferrous sulphate Substances 0.000 description 1
- 235000003891 ferrous sulphate Nutrition 0.000 description 1
- 210000002950 fibroblast Anatomy 0.000 description 1
- 239000000706 filtrate Substances 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- 239000011737 fluorine Substances 0.000 description 1
- 125000001153 fluoro group Chemical group F* 0.000 description 1
- 230000005714 functional activity Effects 0.000 description 1
- 210000001222 gaba-ergic neuron Anatomy 0.000 description 1
- 229960003692 gamma aminobutyric acid Drugs 0.000 description 1
- 229930195712 glutamate Natural products 0.000 description 1
- 229940049906 glutamate Drugs 0.000 description 1
- 230000012010 growth Effects 0.000 description 1
- 230000007954 hypoxia Effects 0.000 description 1
- 230000008076 immune mechanism Effects 0.000 description 1
- 230000003832 immune regulation Effects 0.000 description 1
- 230000008629 immune suppression Effects 0.000 description 1
- 210000000987 immune system Anatomy 0.000 description 1
- 230000006058 immune tolerance Effects 0.000 description 1
- 230000007365 immunoregulation Effects 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 238000010874 in vitro model Methods 0.000 description 1
- 238000001727 in vivo Methods 0.000 description 1
- 230000004054 inflammatory process Effects 0.000 description 1
- 239000003112 inhibitor Substances 0.000 description 1
- 230000010354 integration Effects 0.000 description 1
- 230000037041 intracellular level Effects 0.000 description 1
- 238000001990 intravenous administration Methods 0.000 description 1
- 238000010253 intravenous injection Methods 0.000 description 1
- 229910052742 iron Inorganic materials 0.000 description 1
- BAUYGSIQEAFULO-UHFFFAOYSA-L iron(2+) sulfate (anhydrous) Chemical compound [Fe+2].[O-]S([O-])(=O)=O BAUYGSIQEAFULO-UHFFFAOYSA-L 0.000 description 1
- 229910000359 iron(II) sulfate Inorganic materials 0.000 description 1
- 230000000302 ischemic effect Effects 0.000 description 1
- 210000000867 larynx Anatomy 0.000 description 1
- 239000010410 layer Substances 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 238000012417 linear regression Methods 0.000 description 1
- 229910003002 lithium salt Inorganic materials 0.000 description 1
- 159000000002 lithium salts Chemical class 0.000 description 1
- 238000011866 long-term treatment Methods 0.000 description 1
- 230000007774 longterm Effects 0.000 description 1
- 210000001165 lymph node Anatomy 0.000 description 1
- 230000003211 malignant effect Effects 0.000 description 1
- 230000001404 mediated effect Effects 0.000 description 1
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004899 motility Effects 0.000 description 1
- 238000010172 mouse model Methods 0.000 description 1
- 210000003205 muscle Anatomy 0.000 description 1
- 210000000653 nervous system Anatomy 0.000 description 1
- 239000003176 neuroleptic agent Substances 0.000 description 1
- 210000002569 neuron Anatomy 0.000 description 1
- 230000004112 neuroprotection Effects 0.000 description 1
- 230000000324 neuroprotective effect Effects 0.000 description 1
- 210000000440 neutrophil Anatomy 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 210000000056 organ Anatomy 0.000 description 1
- WCPAKWJPBJAGKN-UHFFFAOYSA-N oxadiazole Chemical compound C1=CON=N1 WCPAKWJPBJAGKN-UHFFFAOYSA-N 0.000 description 1
- 230000036961 partial effect Effects 0.000 description 1
- 230000009054 pathological process Effects 0.000 description 1
- 230000002093 peripheral effect Effects 0.000 description 1
- 210000001428 peripheral nervous system Anatomy 0.000 description 1
- 230000000144 pharmacologic effect Effects 0.000 description 1
- 210000003800 pharynx Anatomy 0.000 description 1
- 229960002036 phenytoin Drugs 0.000 description 1
- 208000028591 pheochromocytoma Diseases 0.000 description 1
- 239000013641 positive control Substances 0.000 description 1
- 239000011698 potassium fluoride Substances 0.000 description 1
- 235000003270 potassium fluoride Nutrition 0.000 description 1
- 229960003089 pramipexole Drugs 0.000 description 1
- FASDKYOPVNHBLU-ZETCQYMHSA-N pramipexole Chemical compound C1[C@@H](NCCC)CCC2=C1SC(N)=N2 FASDKYOPVNHBLU-ZETCQYMHSA-N 0.000 description 1
- 239000002243 precursor Substances 0.000 description 1
- 230000035755 proliferation Effects 0.000 description 1
- 239000003368 psychostimulant agent Substances 0.000 description 1
- 150000003222 pyridines Chemical class 0.000 description 1
- 150000003230 pyrimidines Chemical class 0.000 description 1
- 125000000714 pyrimidinyl group Chemical group 0.000 description 1
- 239000011541 reaction mixture Substances 0.000 description 1
- 230000001373 regressive effect Effects 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 230000003252 repetitive effect Effects 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 210000002345 respiratory system Anatomy 0.000 description 1
- 238000007363 ring formation reaction Methods 0.000 description 1
- 229960001879 ropinirole Drugs 0.000 description 1
- UHSKFQJFRQCDBE-UHFFFAOYSA-N ropinirole Chemical compound CCCN(CCC)CCC1=CC=CC2=C1CC(=O)N2 UHSKFQJFRQCDBE-UHFFFAOYSA-N 0.000 description 1
- 230000000276 sedentary effect Effects 0.000 description 1
- 230000001953 sensory effect Effects 0.000 description 1
- 239000000741 silica gel Substances 0.000 description 1
- 229910002027 silica gel Inorganic materials 0.000 description 1
- 150000003384 small molecules Chemical class 0.000 description 1
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 1
- 239000007858 starting material Substances 0.000 description 1
- 125000001424 substituent group Chemical group 0.000 description 1
- 150000003462 sulfoxides Chemical class 0.000 description 1
- 230000005062 synaptic transmission Effects 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- 238000004885 tandem mass spectrometry Methods 0.000 description 1
- 238000010998 test method Methods 0.000 description 1
- 210000001103 thalamus Anatomy 0.000 description 1
- 231100001274 therapeutic index Toxicity 0.000 description 1
- 238000002560 therapeutic procedure Methods 0.000 description 1
- XSKZXGDFSCCXQX-UHFFFAOYSA-N thiencarbazone-methyl Chemical compound COC(=O)C1=CSC(C)=C1S(=O)(=O)NC(=O)N1C(=O)N(C)C(OC)=N1 XSKZXGDFSCCXQX-UHFFFAOYSA-N 0.000 description 1
- 238000004809 thin layer chromatography Methods 0.000 description 1
- RZWIIPASKMUIAC-VQTJNVASSA-N thromboxane Chemical compound CCCCCCCC[C@H]1OCCC[C@@H]1CCCCCCC RZWIIPASKMUIAC-VQTJNVASSA-N 0.000 description 1
- 210000001519 tissue Anatomy 0.000 description 1
- CFOAUYCPAUGDFF-UHFFFAOYSA-N tosmic Chemical compound CC1=CC=C(S(=O)(=O)C[N+]#[C-])C=C1 CFOAUYCPAUGDFF-UHFFFAOYSA-N 0.000 description 1
- 238000002054 transplantation Methods 0.000 description 1
- 230000008733 trauma Effects 0.000 description 1
- 239000003029 tricyclic antidepressant agent Substances 0.000 description 1
- 238000001291 vacuum drying Methods 0.000 description 1
- 230000002792 vascular Effects 0.000 description 1
- 230000021542 voluntary musculoskeletal movement Effects 0.000 description 1
- 230000029663 wound healing Effects 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/14—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings
- C07D413/04—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing three or more hetero rings
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
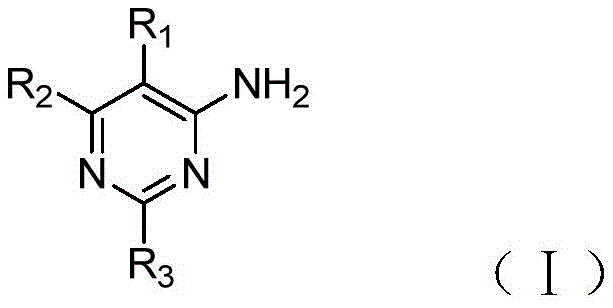
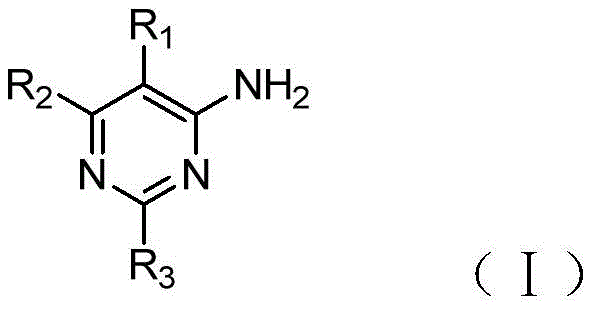
The invention discloses a compound serving as adenosine A2A4-aminopyrimidine derivatives of receptor antagonists having the general structural formula (i):wherein R is1Selected from halogen, cyano or trifluoromethyl; r2Selected from pyrazolyl, pyrrolidinyl, or substituted by one or more halogens or C1‑3Alkyl-substituted pyrazolyl or pyrrolidinyl; r3Selected from oxazolyl, oxadiazolyl, triazolyl, or substituted with one or more halogens or C1‑3An alkyl-substituted oxazolyl group. The 4-aminopyrimidine derivative provided by the invention is human adenosine A2AThe receptor has obvious antagonistic effect, and can be used for treating A2AAntagonism-responsive diseases or conditions, particularly neurodegenerative diseases, extra-pyramidal syndrome, depression, hyperactivity syndrome, sleep disorders, anxiety, diabetes or tumours.
Description
Technical Field
The invention relates to the field of medicine and organic chemistry, in particular to adenosine A2A4-aminopyrimidine derivatives of receptor antagonists and their use.
Background
As endogenous regulators of many physiological functions in the Central (CNS) and peripheral nervous systems, adenosine is widely distributed in multiple tissues and organs such as the nervous system, cardiovascular system, digestive system, respiratory system, etc., and plays a role in regulating various important physiological processes. It exerts its biological effects through a class of membrane-specific receptors belonging to the superfamily of G protein-coupled receptors, for which 4 subtypes of adenosine receptors have been identified, which are: a. the1、A2A、A2BAnd A3. Wherein A is1And A2AIs a highly expressed receptor which acts at physiologically low adenosine concentrations, and A2BAnd A3The expression level of (A) is low, and the adenosine can be activated only when the adenosine is greatly increased under pathological conditions, so that a series of pathological effects are generated. A. the1And A3Receptors down-regulate cellular levels by coupling them to G proteins that inhibit adenylate cyclase; in contrast, A2AAnd A2BThe receptor is coupled to Gs proteins that activate adenylate cyclase and increase intracellular levels. Through the action of these receptors, adenosine is capable of achieving a wide range of modulation of physiological functions.
A2AReceptors are mostly found in dopamine rich areas such as basal ganglia elements, striatum and globus pallidus in various mammals including humans. The basal ganglia and striatum asThe central element, which is involved in the integration of information in the cortex, thalamus and peripheral lobes to produce motor behavior. A has been found in striatum2AThe receptor and dopamine D2 receptor are tightly co-localized to striatal globus pallidus gabaergic neurons, forming a so-called indirect export pathway from the striatum that is associated with inhibition of motility. A. the2AThe receptor regulates the neurotransmission of gamma-aminobutyric acid, dopamine, acetylcholine and glutamate through a variety of pathways facilitating control of motor behaviour. In general, A2ABy interaction with the D2 receptor, especially A2AThe effect as an antagonist is of great benefit for the treatment of parkinson's disease, which can lead to a reduction in dopamine levels. A. the2AThe receptor interacts tightly and antagonistically with the D2 receptor, causing a decrease in the affinity of the D2 receptor for dopamine when stimulated. Thus, A2AReceptor antagonists can potentiate the effects of endogenous dopamine as well as clinically used dopamine receptor agonists and increase the drug response time limit of dopaminergic activity.
Selectivity A2AReceptor agonists and antagonists have been extensively described in experiments of pharmacology, behavior, and neuroprotection in rodents and non-human primates. At D2 receptor antagonist and A2AD2 and A can be clearly exemplified in the model of receptor agonist-induced catalepsy2AThe close interaction of the receptor in catalepsy models, which is mediated by A2AReceptor antagonists and D2 receptor agonists. At present, a number of researchers have reported that2APotential of receptor antagonists against defibrillation. For example, adenosine A2AThe receptor antagonists SCH58261 and KW-6002, both enhanced bilateral rotation induced by sub-domain doses of levodopa in unilateral 6-hydroxydopamine (6-OHDA) injured mice and rats. In addition, adenosine A has been generally reported2AThe receptor antagonist KW-6002 significantly ameliorates non-dyskinetic motor impairment induced by 1-methyl-4-phenyl-1, 2,3, 6-tetrahydropyridine (MPTP) in non-human primates by long-term treatment with the dopamine receptor agonist levodopa. Thus, since it not only reverses motor impairment but also can slow or stop disease progression by extending cell life, a2AReceptor antagonists as parpFuture drugs for long-term use by patients with parkinson's disease show great potential.
Several preclinical studies have shown adenosine A2AReceptor antagonists are useful for the treatment of neurodegenerative diseases such as parkinson's disease, huntington's disease or alzheimer's disease. And has been reported to be A2AReceptor antagonists have neuroprotective effects in vivo and in vitro models of different neurodegenerative diseases. In summary, A2AReceptor antagonists are effective in protecting different neurons from various forms of injury-induced neurodegeneration.
Has the research finding A2AReceptor knockout mice are less sensitive to "inhibitor" challenge than their wild-type counterparts. Consistent with this study, A was tested in the mouse tail suspension experiment2AReceptor antagonists SCH58261 and KW6002 reduced total immobility time. It was also found that the antagonists SCH8261 and ZM241385 reduced immobility when administered to previously screened mice for high immobility time, while SCH58261 reduced immobility in mice selectively raised for their "helpless" in this model. With A2AKnockout mouse studies have shown that these animals are sluggish to psychostimulants such as amphetamine and cocaine. Thus, there is evidence to suggest that adenosine A2AReceptor antagonists may have antidepressant and/or antipsychotic properties via a dopaminergic pathway that modulates the mesostriatum or the mesocorticoid. A. the2AReceptor activation can contribute to the amelioration of a range of neuropsychiatric diseases and disorders, such as depression, excessive daytime sleepiness, restless leg syndrome (R L S), attention deficit hyperactivity disorder and cognitive fatigue.
Extrapyramidal syndrome (EPS) is a generic term for a series of adverse neurological reactions associated with the use of antipsychotics. There are 6 different classes of EPS-associated neurological syndromes, of which 4, dystonia, akathisia, pseudoparkinsonism (Parkinson's syndrome) and delayed dyskinesia, are particularly prevalent in patients treated with antipsychotic drugs. Dystonia is a painful spasm of the muscle groups, particularly the neck, chin, back, pharynx and larynx. It is most commonly used in young men treated with antipsychotics, but may also be associated with the use of cocaine, tricyclic antidepressants, lithium salt anticonvulsants (e.g., phenytoin and carbamazepine). Pseudoparkinsonism manifests itself as akinesia (rigidity, stiffness and slow voluntary movement, humpback, trails) and tremor, and these symptoms occur weeks or months after the start of therapy. Sedentary sitting fails to manifest itself as hyperkinetic movement, subjective internal sensory distress or discomfort, often misinterpreted as agitation or anxiety, a common syndrome that is often undiagnosed and minimally responsive to treatment. Prolonged dyskinesia is a later-occurring syndrome associated with chronic use of neuroleptic drugs. It occurs more often in elderly patients and is characterized by stereotypic, repetitive, involuntary, rapid chorea-like movements of the face, eyelids, mouth, tongue, limbs and body.
Akathisia is also characteristic of R L S and P L MS (periodic limb movements during sleep) and P L MD (periodic leg (or limb) movement disorders) R L S is a general disorder that causes patients to have an intolerant and unpleasant desire to move their legs, often times develops at rest and/or at night, and may disturb sleep patients who do not have the typical R L S symptoms, but who exhibit intermittent leg movements that adversely affect sleep are diagnosed as P L ms.r L S and P L MS treatments have included levodopa/carbidopa, levodopa/benserazide, dopamine agonists (such as pramipexole and ropinirole), benzodiazepinesOpioids, anticonvulsants and iron (ferrous sulfate).
In the CNS, data show A2AReceptors are present in high density in the basal ganglia, which are important in controlling good movement. Furthermore, A2ASelective antagonists of the receptor are pharmacologically important because they exhibit efficacy in reducing motor impairment, thereby improving function in neurodegenerative diseases, such as parkinson's disease and related movement disorders (e.g., huntington's chorea). Compared to current dopaminergic treatments that bring about an increase in the therapeutic index, A2AAntagonists appear to exhibit a decrease inLow propensity for side effects (e.g., no dyskinesia). A. the2AAntagonists also have antidepressant properties and stimulate cognitive function.
Thus, it is desirable to find novel, highly effective and selective adenosine A2AReceptor antagonists are of increasing interest. Some effective adenosine A discovered by pharmaceutical companies2AAntagonists have entered clinical trials and show positive results, indicating a2AReceptor antagonists are expected to be useful not only in the treatment of neurodegenerative diseases such as parkinson's disease, huntington's disease or alzheimer's disease, but also in the treatment of other CNS related diseases such as depression, hyperactivity syndrome, sleep disorders and anxiety disorders.
In addition, adenosine A2AReceptors are also closely related to immunomodulation. Immunoregulation is an important means for keeping the body homeostasis and resisting external harmful stimuli. Adenosine, an important transmitter and modulator of the body, can be greatly increased in metabolic disturbance and cell injury, activates adenosine receptors to play a biological effect, and participates in the immune regulation of the body. Recent studies have shown that adenosine A is involved in many pathological processes such as ischemic hypoxia, inflammation, trauma, transplantation, etc2AActivation of the receptor may play an important immunomodulatory role, possibly in conjunction with A2AThe receptor is related to high expression level on various immune cells such as T cells, B cells, mononuclear macrophages, neutrophils and the like.
A2AThe receptor is closely related to the tumor. Normally, the body can rely on an intact immune mechanism to effectively monitor and reject cancerous cells, such as: in the aspect of cellular immunity, T lymphocytes, antibody-dependent cytotoxic cells (K cells), NK cells and macrophages all have killing effects on tumor cells. However, if the function of the cancer cells themselves or the above immune cells is changed, the cancer cells may escape the immune system and may be malignant to form tumor. Studies have shown that A2AThe activation of the receptor can promote the organism to generate immune tolerance, and is closely involved in the formation of 'immune escape' or 'immune suppression' of tumor cells, thereby creating favorable conditions for the occurrence and development of tumors. A. the2AReceptor activation can be achieved by inhibiting vascular endotheliumThe expression of the cellular thrombospondin promotes angiogenesis in a dose-dependent manner, so that a favorable environment is created for the growth of the blood vessel-dependent tumor; a. the2AThe activation of the receptor can also inhibit the killing of natural killer cells to tumor cells by raising cAMP and activating PKA; can promote proliferation of tumor cells such as melanoma A375 cells, fibroblast NIH3T3 cells and pheochromocytoma PC12 cells. It has been shown that A2AThe receptor antagonist can be used for treating various tumors, such as lung cancer, especially non-small cell lung cancer.
Is suitable for A2ACandidate products for receptor antagonists requiring potent co-A2AReceptor binding but not strong binding to other adenosine receptors, i.e. higher A is required2AReceptor subtype selectivity, which helps to reduce potential side effects. A number of small molecules of adenosine A are now available2AReceptor antagonists are entering clinical phase I studies for the treatment of tumors. Such as CPI-444 from Corvus corporation, showing binding A2AThe affinity (Ki) of the receptor was 3.5nm, A1The receptor affinity (Ki) was 192nm for A1Based on these results, researchers believe that CPI-444 has sufficient safety and potential efficacy at the currently expected dose levels over a reasonable dose range, CPI-444 has been tested in 3 mouse models of different growing tumors, namely the E L-4 lymphoma model, the MC38 colon tumor model and the CT26 colon tumor model, and has shown a significant reduction in the number of lymph nodes at the cancer cells, or a significant reduction in tumor volume at the primary site, or a steady or regressive tumor volume, or even some mice have shown complete cure2AThe receptor antagonist can be used for treating tumor.
Furthermore, A2AThe immunomodulatory effects of receptors in chronic inflammatory diseases such as asthma and atherosclerosis are also increasingly recognized and appreciated, a2AReceptors are also closely related to wound healing or atrial fibrillation. A number of studies have shown that A2AReceptor antagonists may also be useful in the treatment of diabetes。
Chinese invention patent (CN102892761) provides a new adenosine A2A4-aminopyrimidine derivatives of receptor antagonists:
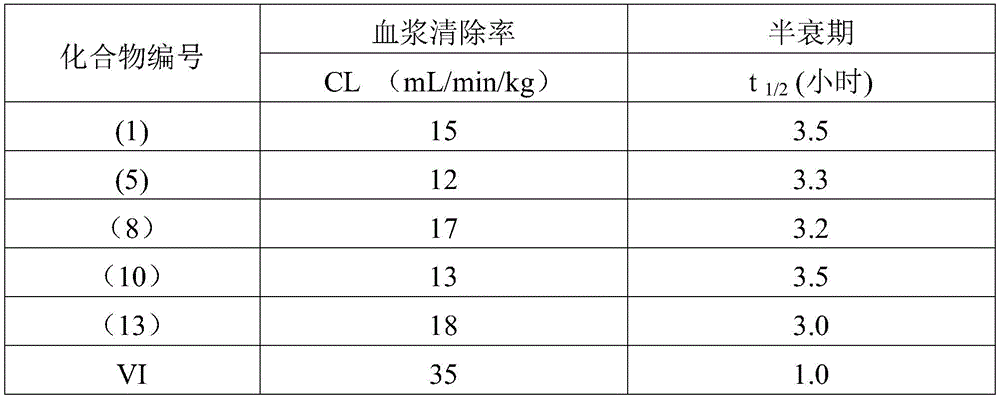
wherein R is1Represents a pyrazole, thiazole or triazole ring optionally substituted by one or two halogen atoms or by one or two methyl or trifluoromethyl groups. The compound is useful for adenosine A2AThe receptor has better antagonism and is shown to be A2AIs stronger, but it is for A1Also, the selectivity of (A) is low, resulting in1In the presence of an acceptor, which is to A2AThe selectivity of the receptor is low. Meanwhile, the pyrazole compound has poor pharmacokinetic characteristics, high plasma clearance rate and short half-life period in a rat body.
Disclosure of Invention
It is an object of the present invention to provide adenosine A as2A4-aminopyrimidine derivatives of receptor antagonists and their use in the treatment of A2AAntagonizing the responsive disease or condition.
In order to achieve the above object, the present invention provides a compound which is adenosine A2A4-aminopyrimidine derivatives of receptor antagonists having the general structural formula (i):
wherein:
R1selected from halogen, cyano or trifluoromethyl;
R2selected from pyrazolyl, pyrrolidinyl, or substituted by one or more halogens or C1-3Alkyl-substituted pyrazolyl or pyrrolidinyl;
R3selected from oxazolyl, oxadiazolyl, triazolyl, or substituted with one or more halogens or C1-3An alkyl-substituted oxazolyl group.
Preferably, the aboveAs adenosine A2A4-aminopyrimidine derivatives of receptor antagonists, wherein R2Selected from pyrazolyl or pyrrolidinyl; r3Selected from oxazolyl, oxadiazolyl, triazolyl, or via a C1-3An alkyl-substituted oxazolyl group.
Preferably, the above is adenosine A2AA 4-aminopyrimidine derivative of a receptor antagonist, wherein the 4-aminopyrimidine derivative is selected from the following compounds:
preferably, the 4-aminopyrimidine derivative is selected from the following compounds: (1) (5), (8), (10), (13), (14), (17) or (18).
The invention also provides the application of the 4-aminopyrimidine derivative in preparing the medicine for treating A2AAntagonizing a disease or condition responsive thereto.
Further, the disease or condition comprises any one or more of a neurodegenerative disease, extra pyramidal syndrome, depression, hyperactivity syndrome, sleep disorders, anxiety, diabetes, or a tumor.
Further, the tumor is lung cancer, ovarian cancer, pancreatic cancer, gastric cancer, breast cancer, glioblastoma, melanoma, renal cell carcinoma, triple negative breast cancer, colorectal cancer, head and neck cancer, bladder cancer, prostate cancer, hepatocellular carcinoma, or cholangiocarcinoma.
Further, the lung cancer is non-small cell lung cancer.
The invention also provides a pharmaceutical composition, wherein the pharmaceutical composition contains the adenosine A serving as the adenosine A2A4-aminopyrimidine derivatives of receptor antagonists, and pharmaceutically acceptable excipients therefor.
The invention also provides a compound medicine, which comprises the 4-aminopyrimidine derivative and a medicine capable of being combined with the derivative.
Further, the combination is a compound for the treatment of the following diseases or conditions: neurodegenerative diseases, extrapyramidal syndrome, depression, hyperactivity syndrome, sleep disorders, anxiety disorders, diabetes or tumors.
The invention has the following beneficial effects: the 4-aminopyrimidine derivative provided by the invention is adenosine A2AThe receptor has obvious selective antagonism and better pharmacokinetic characteristics, and can be applied to the treatment of A2AAntagonism-responsive diseases or conditions, particularly neurodegenerative diseases, extra-pyramidal syndrome, depression, hyperactivity syndrome, sleep disorders, anxiety, diabetes or tumours.
Detailed Description
The present invention is further described by the following specific examples, which are only for illustrating the present invention and are not intended to limit the scope of the present invention.
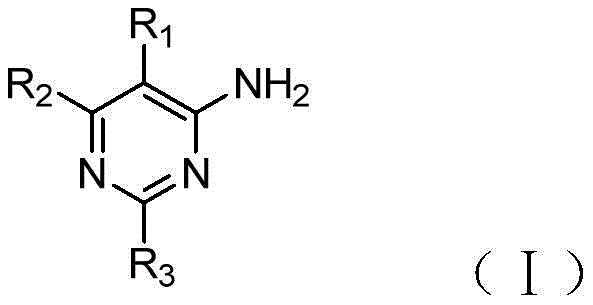
The invention provides a compound serving as adenosine A2A4-aminopyrimidine derivatives of receptor antagonists having the general structural formula (i):
wherein:
R1selected from halogen, cyano or trifluoromethyl;
R2selected from pyrazolyl, pyrrolidinyl, or substituted by one or more halogens or C1-3Alkyl-substituted pyrazolyl or pyrrolidinyl;
R3selected from oxazolyl, oxadiazolyl, triazolyl, or substituted with one or more halogens or C1-3An alkyl-substituted oxazolyl group.
Preferably, the above is adenosine A2A4-aminopyrimidine derivatives of receptor antagonists, wherein R2Selected from pyrazolyl or pyrrolidinyl; r3Selected from oxazolyl, oxadiazolyl, triazolyl, or via a C1-3An alkyl-substituted oxazolyl group.
Preferably, the above is adenosine A2A4-aminopyrimidines as receptor antagonistsA pyridine derivative, wherein the 4-aminopyrimidine derivative is selected from the following compounds:
preferably, the 4-aminopyrimidine derivative is selected from the following compounds: (1) (5), (8), (10), (13), (14), (17) or (18).
The invention also provides the application of the 4-aminopyrimidine derivative in preparing the medicine for treating A2AAntagonizing a disease or condition responsive thereto.
Further, the disease or condition comprises any one or more of a neurodegenerative disease, extra pyramidal syndrome, depression, hyperactivity syndrome, sleep disorders, anxiety, diabetes, or a tumor.
Further, the tumor is lung cancer, ovarian cancer, pancreatic cancer, gastric cancer, breast cancer, glioblastoma, melanoma, renal cell carcinoma, triple negative breast cancer, colorectal cancer, head and neck cancer, bladder cancer, prostate cancer, hepatocellular carcinoma, or cholangiocarcinoma.
Further, the lung cancer is non-small cell lung cancer.
The invention also provides a pharmaceutical composition, wherein the pharmaceutical composition contains the adenosine A serving as the adenosine A2A4-aminopyrimidine derivatives of receptor antagonists, and pharmaceutically acceptable excipients therefor.
The invention also provides a compound medicine, which comprises the 4-aminopyrimidine derivative and a medicine capable of being combined with the derivative.
Further, the combination is a compound for the treatment of the following diseases or conditions: neurodegenerative diseases, extrapyramidal syndrome, depression, hyperactivity syndrome, sleep disorders, anxiety disorders, diabetes or tumors.
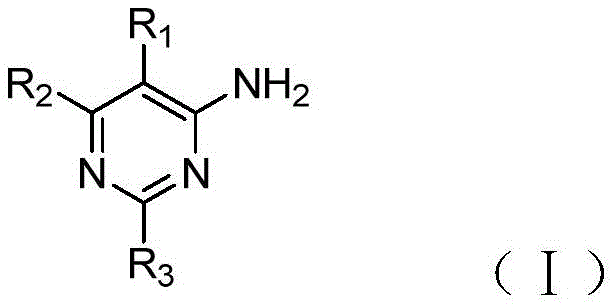
Provided by the invention as adenosine A2A4-aminopyrimidine derivatives of receptor antagonists can be synthesized by one of the following synthetic routes:
scheme 1:
the reagent and the condition are (a) acetic anhydride and reflux, (b) pyrazole, cesium carbonate, Dimethylformamide (DMF) at the temperature of 80 ℃, (c) trans- β -styrene boric acid, sodium carbonate, dioxane and room temperature, and tetrakis (triphenylphosphine) palladium (Pd (PPh)3)4) At 90 ℃; (d) ozone, methanol (MeOH)/dichloromethane (CH)2Cl2) -78 deg.C; (e) p-toluenesulfonylmethyl isonitrile (TOSMIC), potassium carbonate (K)2CO3) MeOH, 80 ℃; (f), N-bromosuccinimide and DMF.
The preparation of the compounds (1) - (3), (6) - (8), (11) - (13), (15) - (17) is carried out according to scheme 1. See example 1 for a specific method of implementation. Compounds in which the 5-position fluorine of the pyrimidine is substituted may also be prepared starting from fluorine atom-containing starting materials.
Scheme 2:
the reagent and the conditions are (a), m-chloroperbenzoic acid, dichloromethane, room temperature, (b), N-bromosuccinimide (NBS), DMF, room temperature, (c), 1, oxazole, butyl lithium (N-Bu L i), Tetrahydrofuran (THF), 78-20 ℃,2, Pd (PPh)3)4At the temperature of 80 ℃; (d) 1H-pyrazole, cesium carbonate, DMF, 90 ℃; (e) pyrrolidine, cesium carbonate, DMF, 90 ℃.
The sulfoxide of formula (G) is reacted with various commercially available five-membered heterocyclic (e.g., pyrazole, oxazole, oxadiazole or triazole) derivatives in the presence of a base (e.g., cesium carbonate or butyllithium) using DMF as a solvent at room temperature or low temperature to produce a derivative substituted at the 2-position with a five-membered heterocyclic ring. For example, reaction of an intermediate of formula (G) with oxazole under these conditions affords a derivative of formula (H).
The 6-position chlorine atom of the pyrimidine derivative is further replaced by a five-membered heterocyclic derivative (e.g., pyrazole or pyrrolidine) using DMF as a solvent in the presence of a base (e.g., cesium carbonate or sodium methoxide). For example, reaction of derivative (H) with pyrazole or pyrrolidine under these conditions can produce compounds (19) and (23).
The preparation of the compounds (19), (20), (23-25) is carried out according to scheme 2. See example 15 for a specific method of implementation.
Scheme 3:
reagents and conditions: (a) cuprous cyanide, pyridine, Microwave (MW), reaction at 250 ℃ for 20 minutes. (b) Potassium fluoride (KF), bis (dibenzylideneacetone) palladium (Pd (dba)2)2- (dicyclohexylphosphonium) -3, 6-dimethoxy-2 ',4',6 '-triisopropyl-1, 1' -biphenyl, trimethyl (trifluoromethyl) silane, dioxane, 20h, 140 ℃.
For the synthesis of the substituent R as defined above in the 5-position of the pyrimidine1Pyrimidine derivatives, which are cyano or trifluoromethyl, can be prepared using analogous precursor compounds as shown for compound (1) using the methods described in scheme 3, respectively.
The preparation of the compounds (4), (5), (9), (10), (14), (18), (21), (22) is carried out according to scheme 3. See examples 4 and 5 for specific methods of implementation.
Example 1:
preparation of Compound (1), the structural formula of which is as follows:
preparation of compound (1) is carried out according to scheme 1 above:
first, preparation of intermediate (A)
Suspending 4-amino-2, 6-dichloropyrimidine (DCAP, 4g, 24.4mmol) in acetic anhydride (80m L, 860mmol), heating under reflux for 4 hours under stirring, cooling the reaction solution, concentrating under vacuum, adding toluene to the residual acetic anhydride, distilling off, dissolving the residue in ethyl acetate and water, adding 10% NaHCO3Solution to pH of solution7. Washing the organic layer with saturated brine, recovering solvent, dissolving the residue in acetic anhydride (40M L), stirring at 0-5 deg.C for 2 hr, filtering, collecting precipitate, and vacuum drying at 40 deg.C to obtain intermediate (A), MS M/z (ESI) 206.0[ M +1 ]]+。
Second step, preparation of intermediate (B)
Dissolving intermediate (A) (1g, 5mmol) in anhydrous DMF (15M L), adding pyrazole (340mg, 5mmol) and cesium carbonate (1.6g, 5mmol), stirring at 80 deg.C for 2 hr, pouring into water, extracting with ethyl acetate, washing the organic layer with water and saturated brine, drying over anhydrous sodium sulfate, removing solvent under reduced pressure, separating and purifying the residue by silica gel column chromatography (3% methanol: dichloromethane) to obtain product (B), MS M/z (ESI) 238.0[ M +1 ]: 238.0]+。
Third step, preparation of intermediate (C)
Intermediate (B) (0.4g, 1.68mmol), trans- β -styreneboronic acid (0.5g, 3.36mmol) and sodium carbonate (1.08g, 10.1mmol) were added to a dioxane/water solution, nitrogen was purged for about 30min, and Pd (PPh) was added3)4(0.2g, 0.16mmol), the mixture was heated to 90 ℃ and stirred for 20 hours, then poured into water and extracted with ethyl acetate. The organic layer was washed with water and saturated brine in this order, dried over anhydrous sodium sulfate, and the solvent was removed under reduced pressure. The residue was subjected to silica gel column chromatography (3% methanol: dichloromethane) to obtain the product (C). MS M/z (ESI) 306.1[ M +1 ]]+。
The fourth step, preparation of intermediate (D)
Intermediate (C) (0.4g, 1.6mmol) was dissolved in a mixed solution of methanol/dichloromethane (4/1, 20ml), cooled to-78 deg.C, and ozone was introduced for 10 minutes. And after the reaction is finished, introducing nitrogen for 20 minutes, adding dimethyl ether, heating the reaction solution to room temperature, and introducing nitrogen to completely volatilize the solvent to obtain a crude product (D). MS M/z (ESI) 232.1[ M +1 ]]+。
Fifth step, preparation of intermediate (E)
Intermediate (D) (460mg, 2mmol), TOSMIC (p-toluenesulfonylmethylisocyan, 50mg, 4mmol), potassium carbonate (860mg, 6mmol) were mixed with methanol, heated to 80 ℃ and reacted for 16 hours, after which methanol was recovered and the residue was poured into water, ethylAnd (5) extracting with ethyl acetate. The organic layer was washed with water, saturated brine, dried over anhydrous sodium sulfate, and the solvent was removed under reduced pressure. Chromatography of the residue on a silica gel column (5% methanol: dichloromethane) gave the product (E). MS M/z (ESI) 229.1[ M +1 ]]+。
Fifth step, Compound (1) is prepared
0.2g (1.25mmol) NBS (N-bromosuccinimide) was slowly added to a cold DMF solution in which 0.2g of intermediate (E) was suspended. After stirring at room temperature for 1 hour, the solvent was removed under reduced pressure. The residue was poured into water and extracted with ethyl acetate. The organic layer was washed with water, saturated brine, dried over anhydrous sodium sulfate, and the solvent was removed under reduced pressure. The residue was purified by silica gel column chromatography (5% methanol: dichloromethane) to obtain compound (1). MS M/z (ESI) 307.1[ M +1 ]]+,1HNMR(400MHz,DMSO-d6) 8.55(d,1H),8.32(s,1H),7.84(d,1H),7.53(s,1H),7.58(s,2H),6.58(dd, 1H)。
Example 2:
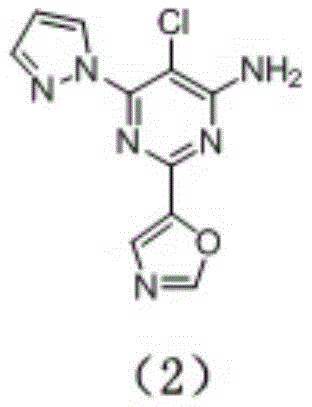
preparation of Compound (2), having the formula:
preparation of compound (2) is carried out according to scheme 1, with specific methods as referenced in example 1. Substituting NCS (N-chlorosuccinimide) for NBS (N-bromosuccinimide) in the reaction of the step 6 to perform chlorination to obtain a compound (2). MS M/z (ESI) 263.0[ M +1 ]]+,1HNMR(400MHz,DMSO-d6) 8.53(d,1H),8.26(s,1H),7.82(d,1H),7.49(s,1H),7.54(s,2H),6.55(dd, 1H)。
Example 3
Preparation of Compound (3), the structural formula of which is as follows:
compound (3) is prepared according to scheme 1. MS M/z (ESI) 247.0[ M +1 ]]+,1HNMR (400MHz,DMSO-d6)8.51(d,1H),8.27(s,1H),7.83(d,1H),7.48(s,1H), 7.55(s,2H),6.54(dd,1H)。
Example 4:
preparation of Compound (4), having the formula:
a mixture of 11.3mg (20. mu. mol) Pd (dba)2And 15.8mg (29.4. mu. mol) of 2- (dicyclohexylphosphorus) -3, 6-dimethoxy-2 ',4',6 '-triisopropyl-1, 1' -biphenyl were added to 3ml of dioxane, and then the mixed solution was added to a mixture containing 0.1g (0.33mmol) of the compound (1) (example 1), 0.04g (0.65mmol) of potassium fluoride, 0.093g (0.65mmol) of trimethyl (trifluoromethyl) silane was added, the reaction solution was stirred at 140 ℃ for 20 hours and then filtered through celite, and the filtrate was concentrated in vacuo. The residue was purified by silica gel column chromatography (dichloromethane: methanol) to give compound (4). MS M/z (ESI) 297.1[ M +1 ]]+,1HNMR(400MHz,DMSO-d6)8.51(d,1H),8.31 (s,1H),7.85(d,1H),7.52(s,1H),7.55(s,2H),6.58(dd,1H)。
Example 5:
preparation of Compound (5), having the formula:
the preparation method is as shown in synthetic route 3, the compound (1) and 0.06g (0.72mmol) cuprous cyanide are added into pyridine, the mixture is placed in a microwave reactor for reaction at 250 ℃ for 20 minutes, after the reaction is monitored by thin layer chromatography (T L C), ethyl acetate is added, then the mixture is filtered by using kieselguhr, saturated sodium bicarbonate solution and saturated common salt water are used for washing in sequence, an organic layer is dried by anhydrous magnesium sulfate, and the product (5) is obtained by concentration, wherein the mass ratio of MS M/z (ESI) is 254.1[ M +1 ]]+,1HNMR(400MHz,DMSO-d6)8.52(d,1H),8.29(s,1H),7.83 (d,1H),7.51(s,1H),7.57(s,2H),6.59(dd,1H)。
Example 6:
preparation of Compound (6), having the formula:
compound (6) was prepared according to scheme 1, wherein the reaction of step 5 replaced p-toluenesulfonylmethyl isocyan (TOSMIC) with methyl-p-toluenesulfonylmethyl isocyan (Me-TOSMIC) to prepare an analog of compound (E). MS M/z (ESI) 261.1[ M +1 ]]+,1HNMR(400MHz, DMSO-d6)8.55(d,1H),8.36(s,1H),7.84(d,1H),7.57(s,2H),6.61(dd,1H), 2.16(s,3H)。
Example 7:
preparation of Compound (7), having the formula:
prepared according to synthetic route 1. Substitution of TOSMIC with Me-TOSMIC at the reaction of step 5 an analog of Compound (E) was prepared, followed by chlorination with NCS (N-chlorosuccinimide) in place of NBS (N-bromosuccinimide) to give Compound (7). MS M/z (ESI) 277.1[ M +1 ]]+,1HNMR (400MHz,DMSO-d6)8.52(d,1H),8.33(s,1H),7.82(d,1H),7.55(s,2H), 6.60(dd,1H),2.14(s,3H)。
Example 8:
preparation of Compound (8), having the formula:
prepared according to synthetic route 1. Substitution of TOSMIC with Me-TOSMIC at the reaction of step 5 an analog of compound (E) was prepared and then brominated with NBS (N-bromosuccinimide) to give compound (8). MS M/z (ESI) 321.0[ M +1 ]]+,1HNMR(400MHz,DMSO-d6)8.53(d,1H),8.34 (s,1H),7.82(d,1H),7.56(s,2H),6.59(dd,1H),2.12(s,3H)。
Example 9:
preparation of Compound (9), having the formula:
replacing compound (1) with compound (8)Compound (9) was prepared by the method of example 4. MS M/z (ESI) 311.1[ M +1 ]]+,1HNMR(400MHz,DMSO-d6)8.50(d,1H),8.31 (s,1H),7.79(d,1H),7.55(s,2H),6.57(dd,1H),2.13(s,3H)。
Example 10:
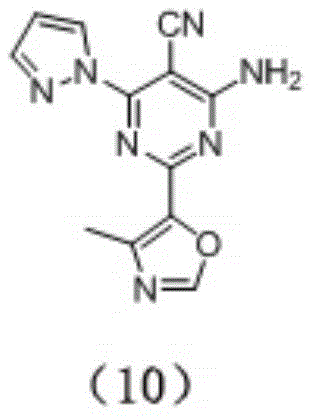
preparation of Compound (10), having the following structural formula:
compound (10) was prepared by the method of example 5, substituting compound (8) for compound (1). MS M/z (ESI) 268.1[ M +1 ]]+,1HNMR(400MHz,DMSO-d6)8.51(d,1H),8.30 (s,1H),7.81(d,1H),7.53(s,2H),6.58(dd,1H),2.16(s,3H)。
Example 11:
preparation of Compound (13), having the following structural formula:
prepared according to synthetic route 1. In the second step, pyrrolidine is used to replace pyrazole to obtain an analogue of the intermediate (B), and the compound (13) is prepared by coupling, cyclization, bromination and other reactions. MS M/z (ESI) 310.0 [ M +1 ]]+,1HNMR(400MHz,DMSO-d6)8.51(d,1H),7.82(d,1H),6.90(s, 2H),3.51-3.74(m,4H),1.82-1.85(m,4H)。
Example 12:
preparation of Compound (14), having the following structural formula:
compound (14) was obtained in the same manner as in example 5 except that Compound (1) was replaced with Compound (13). MS M/z (ESI) 257.1[ M +1 ]]+,1HNMR(400MHz,DMSO-d6)8.50(d,1H),7.81 (d,1H),6.94(s,2H),3.53-3.75(m,4H),1.83-1.87(m,4H)。
Example 13:
preparation of Compound (17), having the formula:
prepared according to synthetic route 1. The step 2 reaction replaces pyrazole with pyrrolidine to prepare an analog of intermediate (B), the step 5 reaction replaces TOSMIC with Me-TOSMIC to prepare an analog of intermediate (E), and then bromination with NBS (N-bromosuccinimide) affords compound (17). MS M/z (ESI) 324.0[ M +1 ]]+,1HNMR(400MHz,DMSO-d6)8.50(s,1H),7.70(s,2H),3.52- 3.76(m,4H),2.16(s,3H),1.82-1.86(m,4H)。
Example 14:
preparation of Compound (18), having the structural formula:
compound (17) in place of compound (1), compound (18) was obtained in the same manner as in example 5. MS M/z (ESI) 271.1[ M +1 ]]+,1HNMR(400MHz,DMSO-d6)8.52(d,1H),7.69 (s,2H),3.53-3.77(m,4H),1.82-1.86(m,4H)。
Example 15:
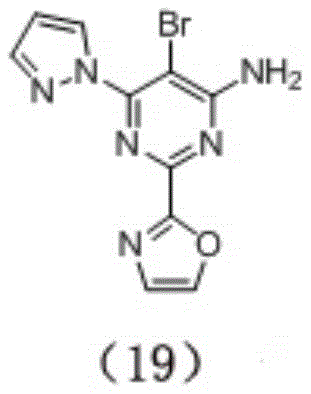
preparation of Compound (19), having the formula:
prepared according to synthetic route 2.
The first step is as follows: preparation of intermediate 6-chloro-2- (methylsulfinyl) pyrimidin-4-amine (F)
To a solution of 1.0g (5.7mmol) of 6-chloro-2- (methylthio) pyrimidin-4-amine in 50ml of dichloromethane was added a solution of 1.5g (6.9mmol) of m-chloroperbenzoic acid (77%) in 30ml of dichloromethane over 30 minutes. The reaction mixture was stirred at room temperature for 4 hours. The resulting white precipitate was filtered, washed several times with dichloromethane and dried to obtain 1.0g of intermediate (F). MS M/z (ESI) 192.0 [ M +1 ]]+。
The second step is that: preparation of intermediate 5-bromo-6-chloro-2-(methylsulfinyl) pyrimidin-4-amine (G): 1.12G (6.3 mmol) of N-bromosuccinimide (NBS) was slowly added to a cooled suspension of 1.0G (5.3mmol) of 6-chloro-2- (methylsulfinyl) pyrimidin-4-amine in 30ml of DMF, stirred at room temperature for 50 minutes, the precipitate was filtered, washed with cold DMF, washed several times with cold water, and dried under vacuum to give (G), MS M/z (ESI):271.9[ M +1 ]]+。
The third step: preparation of intermediate 5-bromo-6-chloro-2- (oxazol-2-yl) pyrimidin-4-amine (H):
oxazole (260mg, 3.7mmol) was dissolved in anhydrous THF at-78 deg.C, nBu L i (1.6M in n-hexane) was added and stirred for 15 minutes, the temperature was naturally raised to-20 deg.C, 1G (3.7mmol) of intermediate (G) and Pd (PPh)3)4Added to anhydrous THF. And then added to the above oxazole solution. After the mixture was reacted at 80 ℃ for 2 hours, the solution was poured into 1N hydrochloric acid, extracted with ethyl acetate, the organic layer was washed with water, washed with saturated brine, dried over anhydrous sodium sulfate, and subjected to silica gel column chromatography (5% methanol in dichloromethane) to obtain the desired product. MS M/z (ESI) 276.9[ M +1 ]]+。
The fourth step: preparation of Compound (19)
To a solution of 0.15g of intermediate (H) in 3ml of DMF were added 0.11g (1.64mmol) of 1H-pyrazole and 0.18g (0.55mmol) of cesium carbonate. The mixture was stirred at 90 ℃ for 24 hours. The solvent DMF was concentrated under reduced pressure. The crude product was washed with water and dried to give compound (19). MS M/z (ESI) 307.1[ M +1 ]]+。
Example 16:
preparation of Compound (25), the structural formula of which is as follows:
prepared according to scheme 2 above, in a manner similar to that described in example 15. In the third step of reaction, the intermediate (G) and the 1H-triazole are stirred in a DMF solution for reaction for several hours at room temperature under the action of cesium carbonate to prepare the corresponding intermediate (H) analogue. The 6-chloro position of pyrimidine was replaced with pyrazole in a similar manner to that in example 15 to give compound (25). MS M/z (ESI) 307.1[ M +1 ]]+,1HNMR(400MHz, DMSO-d6)9.14(s,1H),8.60(s,1H),8.45(d,1H),7.64(d,1H),6.55(dd,1H)。
Pharmacological activity:
(1) para adenosine A2AReceptor binding affinity assay:
by determination of human adenosine A using standard techniques2AReceptor selective radioligands
[3H]Conversion of CGS-21680 to determine in vitro binding of the Compounds of the invention to human adenosine A2AThe binding affinities of the receptors and the results are summarized in table 1.
By encoding human adenosine A2APlasmids of the receptor were stably transfected on HEK-293 cells, which were used to prepare membranes using standard techniques in Tris (hydroxymethyl) aminomethane (Tris-HCl) buffer (pH 7.4). About 15. mu.g of a membrane protein and 50nM of a radioligand3H]CGS-21680, 10. mu.M test compound were mixed and incubated for 90 min at 25 ℃. Nonspecific binding was determined by the addition of 50 μ M NECCA (adenosine-5' -N-ethylcarboxyamide). The membrane was filtered and washed 3 times to remove unbound radioligand. The filter measures bound ligand with a scintillation counter. The concentration-response binding competition curve was determined by analyzing a plurality of different concentrations. Calculating IC using a non-linear fitting program50The value is obtained. The inhibition constant (Ki) of the compounds was calculated by Cheng-Prusoff equation (II).
Ki=IC50/(1+[L]/KD) (Ⅱ)
Wherein the IC50Is the concentration of compound that converts 50% of radioligand binding, [ L]Is the free concentration of the radioligand and KD is the dissociation constant of the radioligand. IC (integrated circuit)50Values were obtained by Prism software fitting the data with non-linear regression. The smaller the Ki value indicates that the compound is at human adenosine A2AThe more pronounced the antagonistic effect of the receptor.
(2) Para adenosine A1Receptor binding affinity assay:
the compound DPCPX (1, 3-dipropyl-cyclopentaxanthine) is known as highly active adenosine A1Receptor antagonists (reported in the literature as Ki 0.45 nM). By determination of human adenosine A using standard techniques1Receptor-selective radioligand [ alpha ]3H]Determination of binding of Compounds of the invention to human adenosine A in vitro1Binding affinity of receptor to determine the Compound of the present invention to A2AThe selective strength of the receptor. The results are summarized in table 1.
By encoding human adenosine A1The plasmid of the receptor was stably transfected on CHO cells, and the cells were used to prepare membranes using standard techniques in modified HEPES buffer (pH 7.4). About 10. mu.g of a membrane protein and 1nM of a radioligand3H]DPCPX, 10. mu.M test compound were mixed and incubated for 90 minutes at 25 ℃. Nonspecific binding was determined by adding 100. mu. M R (-) -PIA (R-phenylisopropyladenosine). The membrane was filtered and washed 3 times to remove unbound radioligand. The filter measures bound ligand with a scintillation counter. The concentration-response binding competition curve was determined by analyzing a plurality of different concentrations. The inhibition constants (Ki) of the compounds were calculated as described previously.
(3) Human adenosine A2AAnalysis of anti-platelet aggregation activity of receptor:
CGS-21680 is adenosine A2AHigh activity agonists of the receptor acting on adenosine A2AThe receptor promotes platelet aggregation. Adenosine A at the cellular level was obtained by testing compounds to inhibit CGS-21680-induced platelet aggregation2AAntagonistic activity of the receptor. The results are summarized in table 1.
At 37 deg.C, plasma rich in human blood platelets is in thromboxane A2Platelet-rich plasma (6 × 10) in the upper layer under the action of receptor agonist U-46619(10 μ M)8Platelets/ml) to produce aggregation, detected by optical agglutination. Using 1 μ M CGS-21680 as a control, it was shown that the test substance may have adenosine A if platelet aggregation reached 50% or more (. gtoreq.50%) in the test compound (30 μ M) system within 5 minutes2AReceptor agonist activity.
When no significant agonistic activity was observed at a certain test substance concentration, it reduced CGS-21680 (1. mu.M) induced inhibitory response by 50% or more (. gtoreq.50%), indicating that the test compound has adenosine A2AReceptor antagonistic activity. According to the above formulaThe inhibition constant (Ki) of the compound is calculated by the method, and the smaller the Ki value is, the lower the inhibition constant (Ki) of the compound is, the human adenosine A of the compound is shown2AThe more pronounced the antagonistic effect of the receptor.
TABLE 1 human adenosine A obtained in affinity assay and platelet aggregation assay for Compounds (1) - (25)2AAnd A1Inhibition constant of receptor ("/" indicates not tested)
As can be seen from Table 1, the compounds prepared according to the present invention were found to be able to express human adenosine A at nM concentration level2AThe receptor has obvious antagonism. And when R is1The group has enhanced charge absorption, such as when the halogen atom is changed from bromine to chlorine or when the halogen atom is changed from halogen atom to cyano, the compound of the invention is used for preparing the human adenosine A2AThe antagonistic activity of the receptor is obviously improved.
Furthermore, the compound VI is the compound of example 1 in CN102892761, which is a pyrazole compound having the following structural formula:
it is now used as a positive control in the present invention. In patent CN102892761, the compound VI is reported to be of human origin A2AThe receptor has an affinity Ki of 12nM at CHO-A2AKi values for cAMP on cells were 25 nM. The present invention adopts the above test method, and the antagonistic activity of the tested compound VI is poorer than that reported in the patent CN102892761 (see Table 1).
Among the 4 receptor subtypes of adenosine, due to A1And A2AIs a highly expressed receptor which acts at physiologically low adenosine concentrations, and A2BAnd A3Is low, and can be activated to cause diseases only when adenosine is greatly increased under pathological conditionsPhysical effect. The oxazole compound of the invention is relative to A1Receptor, pair A2AThe receptor has obvious high selectivity (the selectivity coefficient is that the compound in the table 1 is in the human adenosine A)1Inhibition constant of receptor affinity and inhibition of human adenosine A2AThe ratio of the inhibition constants of the receptor affinities), and the selectivity coefficient of the partial compound is 3-4 times that of compound VI reported in CN102892761, which is considered to be a significant advance in the art.
Human adenosine A by the compounds of the invention2AAnalysis of the anti-platelet aggregation activity of the receptors proves that the receptors have antagonism A on the cellular level2AFunctional role of the receptor. Most of the tested compounds of the present invention showed stronger cellular activity than the pyrazole compound VI described in CN102892761, e.g. compound (5) showed about 4.5 times stronger cellular activity than VI.
Therefore, compared with the pyrazole compound VI described in CN102892761, the oxazole compound provided by the invention is used for human adenosine A2AThe antagonistic activity and the functional activity of the receptor are equal to or better than those of the receptor, and the receptor is human adenosine A1The affinity of the receptor is obviously reduced and shows that the receptor has the affinity for A2AHigh selectivity of the receptor.
Evaluation of pharmacokinetics:
the compounds (1), (5), (8), (10), (13) of the present invention were tested for pharmacokinetics.
According to the conventional method, SD rats are used as test animals, the L C/MS/MS method is used for measuring the drug concentration in the blood plasma of the rats at different moments after the rats are administrated with the compounds (1), (5), (8), (10) and (13) by intravenous injection, the pharmacokinetic behavior of the compounds in the rats is researched, and the pharmacokinetic characteristics of the compounds are evaluated.
TABLE 2 pharmacokinetic parameters of the Compounds of the invention (1), (5), (8), (10), (13) (SD rat intravenous 1.0mg/kg)
The results in table 2 show that the oxazole compound of the present invention has better pharmacokinetic characteristics, and has a significantly lower plasma clearance than the pyrazole compound VI in CN102892761, such that a higher blood concentration and a longer effective time can be maintained; the half-life period of the oxazole compound in a rat body is more than 3 times of that of a compound VI, the half-life period is prolonged by 20-40% in the technical field to be considered as better, and the half-life period is prolonged by more than 1 time to be considered as obvious progress; therefore, compared with the pyrazole compound VI described in CN102892761, the compound of the invention has remarkable improvement in maintaining higher blood concentration and longer effective treatment time.
In conclusion, the 4-aminopyrimidine derivative provided by the invention is human adenosine A2AThe receptor has obvious antagonism and shows high A2AReceptor selectivity and good metabolic absorption in rat body, and can be used for treating A2AAntagonism-responsive diseases or conditions, particularly neurodegenerative diseases, extra-pyramidal syndrome, depression, hyperactivity syndrome, sleep disorders, anxiety, diabetes or tumours.
While the present invention has been described in detail with reference to the preferred embodiments, it should be understood that the above description should not be taken as limiting the invention. Various modifications and alterations to this invention will become apparent to those skilled in the art upon reading the foregoing description. Accordingly, the scope of the invention should be determined from the following claims.
Claims (8)
1. A compound used as adenosine A2A4-aminopyrimidine derivatives of receptor antagonists, characterized by the general structural formula (i) below:
wherein:
R1selected from halogen, cyano;
R2selected from pyrazolyl, pyrrolidinyl;
R3selected from oxazolyl or C1-3An alkyl-substituted oxazolyl group;
the 4-aminopyrimidine derivative is selected from the following compounds:
2. use of a 4-aminopyrimidine derivative according to claim 1 for the preparation of a medicament for the treatment of para-a2AAntagonizing a disease or condition responsive thereto.
3. The use of claim 2, wherein the disease or condition comprises any one or more of a neurodegenerative disease, extra-pyramidal syndrome, depression, hyperactivity syndrome, sleep disorders, anxiety, diabetes or tumors.
4. The use of claim 3, wherein the tumor is lung cancer, ovarian cancer, pancreatic cancer, gastric cancer, breast cancer, glioblastoma, melanoma, renal cell carcinoma, colorectal cancer, head and neck cancer, bladder cancer, prostate cancer, hepatocellular carcinoma, or cholangiocarcinoma.
5. The use of claim 4, wherein the lung cancer is non-small cell lung cancer.
6. A pharmaceutical composition comprising as adenosine a according to claim 12A4-aminopyrimidine derivatives of receptor antagonists, and pharmaceutically acceptable excipients therefor.
7. A combination drug comprising the 4-aminopyrimidine derivative according to claim 1 in combination with a drug.
8. The combination according to claim 7, wherein the combination is a compound for the treatment of: neurodegenerative diseases, extrapyramidal syndrome, depression, hyperactivity syndrome, sleep disorders, anxiety disorders, diabetes or tumors.
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201710542349.1A CN107286146B (en) | 2017-07-05 | 2017-07-05 | 4-aminopyrimidine derivatives as adenosine A2A receptor antagonists and uses thereof |
| PCT/CN2018/085021 WO2019007140A1 (en) | 2017-07-05 | 2018-04-28 | 4-aminopyrimidine derivative used as adenosine a2a receptor antagonist and application thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201710542349.1A CN107286146B (en) | 2017-07-05 | 2017-07-05 | 4-aminopyrimidine derivatives as adenosine A2A receptor antagonists and uses thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN107286146A CN107286146A (en) | 2017-10-24 |
| CN107286146B true CN107286146B (en) | 2020-07-31 |
Family
ID=60100843
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201710542349.1A Active CN107286146B (en) | 2017-07-05 | 2017-07-05 | 4-aminopyrimidine derivatives as adenosine A2A receptor antagonists and uses thereof |
Country Status (2)
| Country | Link |
|---|---|
| CN (1) | CN107286146B (en) |
| WO (1) | WO2019007140A1 (en) |
Families Citing this family (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN107286146B (en) * | 2017-07-05 | 2020-07-31 | 上海肇钰医药科技有限公司 | 4-aminopyrimidine derivatives as adenosine A2A receptor antagonists and uses thereof |
| CN109662966A (en) * | 2017-10-16 | 2019-04-23 | 北京莱科金基因科技有限责任公司 | Purposes of the istradefylline in the drug that preparation is used for oncotherapy |
| JOP20210164A1 (en) | 2019-01-11 | 2023-01-30 | Omeros Corp | Methods and combinations of cancer treatment |
| US12453738B2 (en) | 2019-03-12 | 2025-10-28 | Arcus Biosciences, Inc. | Treatment of oncogene-driven cancers |
| KR20210144821A (en) * | 2019-03-29 | 2021-11-30 | 아르커스 바이오사이언시즈 인코포레이티드 | Cancer treatment method using the identified adenosine fingerprint |
| CN111943938A (en) * | 2019-05-17 | 2020-11-17 | 上海再极医药科技有限公司 | Synthetic method of A2A adenosine receptor antagonist |
| JP7109684B2 (en) | 2019-11-01 | 2022-07-29 | ユニマテック株式会社 | Fluorine-containing pyrimidine compound and method for producing the same |
| US12428400B2 (en) | 2020-06-01 | 2025-09-30 | Unimatec Co., Ltd. | Fluorine-containing fused ring pyrimidine compound and method for producing same |
| EP4317154A4 (en) * | 2021-03-22 | 2025-03-12 | Unimatec Co., Ltd. | FLUORINATED PYRIMIDINE COMPOUND AND METHOD FOR THE PREPARATION THEREOF |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101679371A (en) * | 2006-12-04 | 2010-03-24 | 艾美罗股份公司 | Substituted pyrimidines as adenosine receptor antagonists |
| CN101687850A (en) * | 2007-03-21 | 2010-03-31 | 艾美罗股份公司 | Substituted pyrimidines as adenosine receptor antagonists |
| CN102892761A (en) * | 2010-03-31 | 2013-01-23 | 帕罗生物制药有限公司 | 4-aminopyrimidine derivatives and their use as adenosine A2A receptor antagonists |
| CN103626741A (en) * | 2013-11-26 | 2014-03-12 | 苏州大学 | Heterocyclic aminopyrimidine compound with adenosine receptor antagonist activity |
| CN103664908A (en) * | 2013-12-10 | 2014-03-26 | 苏州大学 | Aminopyrimidine heterocyclic compound having adenosine receptor antagonizing activity |
| CN105008346A (en) * | 2013-01-24 | 2015-10-28 | 帕罗生物制药有限公司 | Novel pyrimidine derivatives as phosphodiesterase 10 (PDE-10) inhibitors |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN107286146B (en) * | 2017-07-05 | 2020-07-31 | 上海肇钰医药科技有限公司 | 4-aminopyrimidine derivatives as adenosine A2A receptor antagonists and uses thereof |
-
2017
- 2017-07-05 CN CN201710542349.1A patent/CN107286146B/en active Active
-
2018
- 2018-04-28 WO PCT/CN2018/085021 patent/WO2019007140A1/en not_active Ceased
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101679371A (en) * | 2006-12-04 | 2010-03-24 | 艾美罗股份公司 | Substituted pyrimidines as adenosine receptor antagonists |
| CN101687850A (en) * | 2007-03-21 | 2010-03-31 | 艾美罗股份公司 | Substituted pyrimidines as adenosine receptor antagonists |
| CN102892761A (en) * | 2010-03-31 | 2013-01-23 | 帕罗生物制药有限公司 | 4-aminopyrimidine derivatives and their use as adenosine A2A receptor antagonists |
| CN105008346A (en) * | 2013-01-24 | 2015-10-28 | 帕罗生物制药有限公司 | Novel pyrimidine derivatives as phosphodiesterase 10 (PDE-10) inhibitors |
| CN103626741A (en) * | 2013-11-26 | 2014-03-12 | 苏州大学 | Heterocyclic aminopyrimidine compound with adenosine receptor antagonist activity |
| CN103664908A (en) * | 2013-12-10 | 2014-03-26 | 苏州大学 | Aminopyrimidine heterocyclic compound having adenosine receptor antagonizing activity |
| CN104447708A (en) * | 2013-12-10 | 2015-03-25 | 苏州大学 | Aminopyrimidine heterocyclic compound having adenosine receptor antagonistic activity |
Non-Patent Citations (3)
| Title |
|---|
| 2-Amino-N-pyrimidin-4-ylacetamides as A2A Receptor Antagonists: 1. Structure-Activity Relationships and Optimization of Heterocyclic Substituents;Deborah H. Slee等;《J. Med. Chem.》;20080229;第51卷;第1719-1729页 * |
| 2-Amino-N-pyrimidin-4-ylacetamides as A2A Receptor Antagonists: 2. Reduction of hERG Activity, Observed Species Selectivity, and Structure-Activity Relationships;Deborah H. Slee等;《J. Med. Chem.》;20080229;第51卷;第1730-1739页 * |
| N-[6-Amino-2-(heteroaryl)pyrimidin-4-yl]acetamides as A2A Receptor Antagonists with Improved Drug Like Properties and in Vivo Efficacy;Marion C. Lanier等;《J. Med. Chem.》;20090113;第52卷;第709-717页 * |
Also Published As
| Publication number | Publication date |
|---|---|
| CN107286146A (en) | 2017-10-24 |
| WO2019007140A1 (en) | 2019-01-10 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN107286146B (en) | 4-aminopyrimidine derivatives as adenosine A2A receptor antagonists and uses thereof | |
| CN113135896A (en) | Methylpyrazole derivatives as RET inhibitors | |
| JP5551066B2 (en) | New compounds | |
| EP1849465A1 (en) | Agent for controlling function of gpr34 receptor | |
| WO2018022761A1 (en) | Substituted cyclopentane-amides for treating disorders related to ret | |
| TW201116532A (en) | Metabotropic glutamate receptor modulators | |
| JP2004533447A (en) | Substituted pyrazinones, pyridines and pyrimidines as corticotropin releasing factor ligands | |
| TW201533044A (en) | Substituted xanthines and methods of use thereof | |
| TW201522347A (en) | Heterocyclic derivatives | |
| MXPA06004361A (en) | NOVEL TETRAYDROSPIRO{PIPERIDINE-2,7aCO -PYRROLO[3,2-b]PYRIDINE DERIVATIVES AND NOVEL INDOLE DERIVATIVES USEFUL IN THE TREATMENT OF 5-HT6 RECEPTOR -RELATED DISORDERS. | |
| WO2011076744A1 (en) | Disubstituted heteroaryl-fused pyridines | |
| EP4079734A1 (en) | Triazolopyridazine derivative, preparation method therefor, pharmaceutical composition thereof, and use thereof | |
| CN104093712A (en) | 1H-Indazole-3-carboxamide compounds as inhibitors of glycogen synthase kinase 3β | |
| CN104955826A (en) | Spiro-quinazolinone derivatives useful for the treatment of neurological diseases and conditions | |
| WO2012020725A1 (en) | Heterocyclic derivative having npy y5 receptor antagonism | |
| CN106687464B (en) | Macrocyclic RIP2 kinase inhibitors | |
| JP6370294B2 (en) | Cyclic aminomethylpyrimidine derivatives | |
| CA3214756A1 (en) | Isoindolinone compounds and imaging agents for imaging huntingtin protein | |
| CA2982232C (en) | Indole derivatives | |
| JP2006525309A (en) | Pyrrolo (1,2-b) pyridazine compounds and their use as CRF-1 receptor antagonists | |
| JP2025531050A (en) | Free base crystalline form of polycyclic compound containing nitrogen heterocycle and method for producing same | |
| WO1996024594A1 (en) | Benzoxazepine derivatives, salts thereof, and drugs containing the same | |
| WO2023160614A1 (en) | Compound as fak inhibitor and use thereof | |
| JP2006519251A (en) | Novel aminopyridine derivatives as mGluR5 antagonists | |
| KR101082227B1 (en) | Methanesulfonic Acid Salts of Pyrazolopyrimidine Compounds, Their Crystals and Methods for Manufacturing the Same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |