CN107174586B - Pharmaceutical composition and application thereof using luteolin derivatives as active ingredients - Google Patents
Pharmaceutical composition and application thereof using luteolin derivatives as active ingredients Download PDFInfo
- Publication number
- CN107174586B CN107174586B CN201710092931.2A CN201710092931A CN107174586B CN 107174586 B CN107174586 B CN 107174586B CN 201710092931 A CN201710092931 A CN 201710092931A CN 107174586 B CN107174586 B CN 107174586B
- Authority
- CN
- China
- Prior art keywords
- derivatives
- melatonin
- pharmaceutical composition
- application
- preparation
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 239000008194 pharmaceutical composition Substances 0.000 title claims abstract description 16
- 239000004480 active ingredient Substances 0.000 title abstract description 6
- 150000002660 luteolin derivatives Chemical class 0.000 title 1
- DRLFMBDRBRZALE-UHFFFAOYSA-N melatonin Chemical compound COC1=CC=C2NC=C(CCNC(C)=O)C2=C1 DRLFMBDRBRZALE-UHFFFAOYSA-N 0.000 claims abstract description 24
- YJPIGAIKUZMOQA-UHFFFAOYSA-N Melatonin Natural products COC1=CC=C2N(C(C)=O)C=C(CCN)C2=C1 YJPIGAIKUZMOQA-UHFFFAOYSA-N 0.000 claims abstract description 23
- 229960003987 melatonin Drugs 0.000 claims abstract description 22
- 102000001419 Melatonin receptor Human genes 0.000 claims abstract description 21
- 108050009605 Melatonin receptor Proteins 0.000 claims abstract description 21
- 239000003814 drug Substances 0.000 claims abstract description 14
- 239000000952 serotonin receptor agonist Substances 0.000 claims abstract description 9
- 208000015114 central nervous system disease Diseases 0.000 claims abstract description 5
- 238000002360 preparation method Methods 0.000 claims description 25
- 102000040125 5-hydroxytryptamine receptor family Human genes 0.000 claims description 8
- 108091032151 5-hydroxytryptamine receptor family Proteins 0.000 claims description 8
- 102000005962 receptors Human genes 0.000 abstract description 13
- 108020003175 receptors Proteins 0.000 abstract description 13
- 229940079593 drug Drugs 0.000 abstract description 7
- 229940076279 serotonin Drugs 0.000 abstract description 3
- MRNPHCMRIQYRFU-KXUMSINMSA-N Arundoin Chemical class C([C@]1(C)[C@@H](C(C)C)CC[C@H]1[C@]1(C)CC=C23)C[C@]1(C)[C@H]3CC[C@@H]1[C@]2(C)CC[C@H](OC)C1(C)C MRNPHCMRIQYRFU-KXUMSINMSA-N 0.000 abstract 2
- QZAYGJVTTNCVMB-UHFFFAOYSA-N serotonin Chemical compound C1=C(O)C=C2C(CCN)=CNC2=C1 QZAYGJVTTNCVMB-UHFFFAOYSA-N 0.000 description 29
- 239000000546 pharmaceutical excipient Substances 0.000 description 13
- 230000001270 agonistic effect Effects 0.000 description 11
- 238000000034 method Methods 0.000 description 11
- 239000000203 mixture Substances 0.000 description 10
- 238000005481 NMR spectroscopy Methods 0.000 description 9
- 150000001875 compounds Chemical class 0.000 description 9
- 238000009472 formulation Methods 0.000 description 8
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 6
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 6
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 6
- 239000000843 powder Substances 0.000 description 6
- YJYPHIXNFHFHND-UHFFFAOYSA-N agomelatine Chemical compound C1=CC=C(CCNC(C)=O)C2=CC(OC)=CC=C21 YJYPHIXNFHFHND-UHFFFAOYSA-N 0.000 description 5
- 210000004027 cell Anatomy 0.000 description 5
- 230000000144 pharmacologic effect Effects 0.000 description 5
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 4
- QUSNBJAOOMFDIB-UHFFFAOYSA-N Ethylamine Chemical compound CCN QUSNBJAOOMFDIB-UHFFFAOYSA-N 0.000 description 4
- NQRYJNQNLNOLGT-UHFFFAOYSA-N Piperidine Chemical compound C1CCNCC1 NQRYJNQNLNOLGT-UHFFFAOYSA-N 0.000 description 4
- KAESVJOAVNADME-UHFFFAOYSA-N Pyrrole Chemical compound C=1C=CNC=1 KAESVJOAVNADME-UHFFFAOYSA-N 0.000 description 4
- 229960002629 agomelatine Drugs 0.000 description 4
- 239000006144 Dulbecco’s modified Eagle's medium Substances 0.000 description 3
- YLXDSYKOBKBWJQ-LBPRGKRZSA-N N-[2-[(8S)-2,6,7,8-tetrahydro-1H-cyclopenta[e]benzofuran-8-yl]ethyl]propanamide Chemical compound C1=C2OCCC2=C2[C@H](CCNC(=O)CC)CCC2=C1 YLXDSYKOBKBWJQ-LBPRGKRZSA-N 0.000 description 3
- 239000003937 drug carrier Substances 0.000 description 3
- 230000000694 effects Effects 0.000 description 3
- 210000002919 epithelial cell Anatomy 0.000 description 3
- 238000001914 filtration Methods 0.000 description 3
- 238000002347 injection Methods 0.000 description 3
- 239000007924 injection Substances 0.000 description 3
- 239000007788 liquid Substances 0.000 description 3
- 239000013641 positive control Substances 0.000 description 3
- 229960001150 ramelteon Drugs 0.000 description 3
- 239000000243 solution Substances 0.000 description 3
- 229960000660 tasimelteon Drugs 0.000 description 3
- PTOIAAWZLUQTIO-GXFFZTMASA-N tasimelteon Chemical compound CCC(=O)NC[C@@H]1C[C@H]1C1=CC=CC2=C1CCO2 PTOIAAWZLUQTIO-GXFFZTMASA-N 0.000 description 3
- FQUYSHZXSKYCSY-UHFFFAOYSA-N 1,4-diazepane Chemical compound C1CNCCNC1 FQUYSHZXSKYCSY-UHFFFAOYSA-N 0.000 description 2
- WHKWMTXTYKVFLK-UHFFFAOYSA-N 1-propan-2-ylpiperazine Chemical compound CC(C)N1CCNCC1 WHKWMTXTYKVFLK-UHFFFAOYSA-N 0.000 description 2
- 241000709675 Coxsackievirus B3 Species 0.000 description 2
- RCYLUNPFECYGDW-UHFFFAOYSA-N N-(4-phenyl-1,2,3,4-tetrahydronaphthalen-2-yl)propanamide Chemical compound C12=CC=CC=C2CC(NC(=O)CC)CC1C1=CC=CC=C1 RCYLUNPFECYGDW-UHFFFAOYSA-N 0.000 description 2
- WVVXBPKOIZGVNS-UHFFFAOYSA-N N-[2-[2-(phenylmethyl)-1H-indol-3-yl]ethyl]acetamide Chemical compound N1C2=CC=CC=C2C(CCNC(=O)C)=C1CC1=CC=CC=C1 WVVXBPKOIZGVNS-UHFFFAOYSA-N 0.000 description 2
- GLUUGHFHXGJENI-UHFFFAOYSA-N Piperazine Chemical compound C1CNCCN1 GLUUGHFHXGJENI-UHFFFAOYSA-N 0.000 description 2
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 2
- REAYFGLASQTHKB-UHFFFAOYSA-N [2-[3-(1H-pyrazol-4-yl)phenoxy]-6-(trifluoromethyl)pyridin-4-yl]methanamine Chemical compound N1N=CC(=C1)C=1C=C(OC2=NC(=CC(=C2)CN)C(F)(F)F)C=CC=1 REAYFGLASQTHKB-UHFFFAOYSA-N 0.000 description 2
- 150000001413 amino acids Chemical group 0.000 description 2
- 230000000561 anti-psychotic effect Effects 0.000 description 2
- 239000000164 antipsychotic agent Substances 0.000 description 2
- 239000002775 capsule Substances 0.000 description 2
- 210000003169 central nervous system Anatomy 0.000 description 2
- 239000012043 crude product Substances 0.000 description 2
- 230000000120 cytopathologic effect Effects 0.000 description 2
- HPNMFZURTQLUMO-UHFFFAOYSA-N diethylamine Chemical compound CCNCC HPNMFZURTQLUMO-UHFFFAOYSA-N 0.000 description 2
- 201000010099 disease Diseases 0.000 description 2
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 2
- 238000002330 electrospray ionisation mass spectrometry Methods 0.000 description 2
- 239000012091 fetal bovine serum Substances 0.000 description 2
- 239000008187 granular material Substances 0.000 description 2
- MWDZOUNAPSSOEL-UHFFFAOYSA-N kaempferol Natural products OC1=C(C(=O)c2cc(O)cc(O)c2O1)c3ccc(O)cc3 MWDZOUNAPSSOEL-UHFFFAOYSA-N 0.000 description 2
- IQPNAANSBPBGFQ-UHFFFAOYSA-N luteolin Chemical compound C=1C(O)=CC(O)=C(C(C=2)=O)C=1OC=2C1=CC=C(O)C(O)=C1 IQPNAANSBPBGFQ-UHFFFAOYSA-N 0.000 description 2
- LRDGATPGVJTWLJ-UHFFFAOYSA-N luteolin Natural products OC1=CC(O)=CC(C=2OC3=CC(O)=CC(O)=C3C(=O)C=2)=C1 LRDGATPGVJTWLJ-UHFFFAOYSA-N 0.000 description 2
- 235000009498 luteolin Nutrition 0.000 description 2
- RKHCTAKUYDTFHE-QMMMGPOBSA-N ly-156,735 Chemical compound C1=C(Cl)C(OC)=CC2=C1NC=C2[C@@H](C)CNC(C)=O RKHCTAKUYDTFHE-QMMMGPOBSA-N 0.000 description 2
- RCGABEQHDDFFRR-UHFFFAOYSA-N n-ethyl-n-(1h-indol-3-ylmethyl)ethanamine Chemical compound C1=CC=C2C(CN(CC)CC)=CNC2=C1 RCGABEQHDDFFRR-UHFFFAOYSA-N 0.000 description 2
- 210000004560 pineal gland Anatomy 0.000 description 2
- 239000000741 silica gel Substances 0.000 description 2
- 229910002027 silica gel Inorganic materials 0.000 description 2
- 238000010898 silica gel chromatography Methods 0.000 description 2
- 150000003384 small molecules Chemical class 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- 239000002904 solvent Substances 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- CZDYPVPMEAXLPK-UHFFFAOYSA-N tetramethylsilane Chemical compound C[Si](C)(C)C CZDYPVPMEAXLPK-UHFFFAOYSA-N 0.000 description 2
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 2
- YIMPWIJIKJBRDM-UHFFFAOYSA-N 1-indol-1-yl-n,n-dimethylmethanamine Chemical compound C1=CC=C2N(CN(C)C)C=CC2=C1 YIMPWIJIKJBRDM-UHFFFAOYSA-N 0.000 description 1
- FXHRAKUEZPSMLJ-UHFFFAOYSA-N 1-methyl-1,4-diazepane Chemical compound CN1CCCNCC1 FXHRAKUEZPSMLJ-UHFFFAOYSA-N 0.000 description 1
- XQJMXPAEFMWDOZ-UHFFFAOYSA-N 3exo-benzoyloxy-tropane Natural products CN1C(C2)CCC1CC2OC(=O)C1=CC=CC=C1 XQJMXPAEFMWDOZ-UHFFFAOYSA-N 0.000 description 1
- 241001494508 Arundo donax Species 0.000 description 1
- 241000283690 Bos taurus Species 0.000 description 1
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- 241000699800 Cricetinae Species 0.000 description 1
- 239000012981 Hank's balanced salt solution Substances 0.000 description 1
- 241000282412 Homo Species 0.000 description 1
- 241000124008 Mammalia Species 0.000 description 1
- 229940123784 Melatonin receptor antagonist Drugs 0.000 description 1
- 241001465754 Metazoa Species 0.000 description 1
- YBKLLOANIOYZAN-UHFFFAOYSA-N N-(1H-indol-3-ylmethyl)piperazin-1-amine Chemical compound N1(CCNCC1)NCC1=CNC2=CC=CC=C12 YBKLLOANIOYZAN-UHFFFAOYSA-N 0.000 description 1
- QQXLDOJGLXJCSE-UHFFFAOYSA-N N-methylnortropinone Natural products C1C(=O)CC2CCC1N2C QQXLDOJGLXJCSE-UHFFFAOYSA-N 0.000 description 1
- VFSMDMJLLJMJQD-UHFFFAOYSA-N N1C(=CC=C1)NCC1=CNC2=CC=CC=C12 Chemical compound N1C(=CC=C1)NCC1=CNC2=CC=CC=C12 VFSMDMJLLJMJQD-UHFFFAOYSA-N 0.000 description 1
- 102000004960 NAD(P)H dehydrogenase (quinone) Human genes 0.000 description 1
- 108020000284 NAD(P)H dehydrogenase (quinone) Proteins 0.000 description 1
- 241000209504 Poaceae Species 0.000 description 1
- QIZDQFOVGFDBKW-DHBOJHSNSA-N Pseudotropine Natural products OC1C[C@@H]2[N+](C)[C@H](C1)CC2 QIZDQFOVGFDBKW-DHBOJHSNSA-N 0.000 description 1
- 241000270942 Rana pipiens Species 0.000 description 1
- 235000001484 Trigonella foenum graecum Nutrition 0.000 description 1
- 244000250129 Trigonella foenum graecum Species 0.000 description 1
- 241000700605 Viruses Species 0.000 description 1
- 230000004913 activation Effects 0.000 description 1
- 239000002671 adjuvant Substances 0.000 description 1
- 230000008484 agonism Effects 0.000 description 1
- 239000005557 antagonist Substances 0.000 description 1
- 230000001153 anti-wrinkle effect Effects 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 230000003078 antioxidant effect Effects 0.000 description 1
- 235000006708 antioxidants Nutrition 0.000 description 1
- 238000003556 assay Methods 0.000 description 1
- 230000004071 biological effect Effects 0.000 description 1
- 230000037396 body weight Effects 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 239000006143 cell culture medium Substances 0.000 description 1
- PBAYDYUZOSNJGU-UHFFFAOYSA-N chelidonic acid Natural products OC(=O)C1=CC(=O)C=C(C(O)=O)O1 PBAYDYUZOSNJGU-UHFFFAOYSA-N 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 230000027288 circadian rhythm Effects 0.000 description 1
- 238000004440 column chromatography Methods 0.000 description 1
- 238000001784 detoxification Methods 0.000 description 1
- 238000010586 diagram Methods 0.000 description 1
- 239000003085 diluting agent Substances 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 239000000945 filler Substances 0.000 description 1
- 238000004108 freeze drying Methods 0.000 description 1
- 229940088597 hormone Drugs 0.000 description 1
- 239000005556 hormone Substances 0.000 description 1
- 229940053999 hypnotics and sedatives melatonin receptor agonists Drugs 0.000 description 1
- 210000003016 hypothalamus Anatomy 0.000 description 1
- 230000002401 inhibitory effect Effects 0.000 description 1
- 238000010255 intramuscular injection Methods 0.000 description 1
- 239000007927 intramuscular injection Substances 0.000 description 1
- 238000010253 intravenous injection Methods 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 108010082117 matrigel Proteins 0.000 description 1
- 239000002609 medium Substances 0.000 description 1
- 238000002844 melting Methods 0.000 description 1
- 230000008018 melting Effects 0.000 description 1
- 230000003020 moisturizing effect Effects 0.000 description 1
- 229930014626 natural product Natural products 0.000 description 1
- 230000000955 neuroendocrine Effects 0.000 description 1
- 239000002858 neurotransmitter agent Substances 0.000 description 1
- 231100000252 nontoxic Toxicity 0.000 description 1
- 230000003000 nontoxic effect Effects 0.000 description 1
- 238000000655 nuclear magnetic resonance spectrum Methods 0.000 description 1
- -1 organic acid tropine ester Chemical class 0.000 description 1
- 238000004806 packaging method and process Methods 0.000 description 1
- 239000000825 pharmaceutical preparation Substances 0.000 description 1
- 238000004382 potting Methods 0.000 description 1
- VLEUZFDZJKSGMX-ONEGZZNKSA-N pterostilbene Chemical class COC1=CC(OC)=CC(\C=C\C=2C=CC(O)=CC=2)=C1 VLEUZFDZJKSGMX-ONEGZZNKSA-N 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 238000012216 screening Methods 0.000 description 1
- 238000007789 sealing Methods 0.000 description 1
- 239000008227 sterile water for injection Substances 0.000 description 1
- 230000001954 sterilising effect Effects 0.000 description 1
- 238000004659 sterilization and disinfection Methods 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 239000006228 supernatant Substances 0.000 description 1
- 210000000221 suprachiasmatic nucleus Anatomy 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 230000001225 therapeutic effect Effects 0.000 description 1
- 238000004809 thin layer chromatography Methods 0.000 description 1
- 235000001019 trigonella foenum-graecum Nutrition 0.000 description 1
- 239000005526 vasoconstrictor agent Substances 0.000 description 1
- 230000029812 viral genome replication Effects 0.000 description 1
- 239000008215 water for injection Substances 0.000 description 1
- 230000002087 whitening effect Effects 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/55—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having seven-membered rings, e.g. azelastine, pentylenetetrazole
- A61K31/551—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having seven-membered rings, e.g. azelastine, pentylenetetrazole having two nitrogen atoms, e.g. dilazep
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/40—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil
- A61K31/403—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil condensed with carbocyclic rings, e.g. carbazole
- A61K31/404—Indoles, e.g. pindolol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/40—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil
- A61K31/403—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil condensed with carbocyclic rings, e.g. carbazole
- A61K31/404—Indoles, e.g. pindolol
- A61K31/4045—Indole-alkylamines; Amides thereof, e.g. serotonin, melatonin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
- A61K31/445—Non condensed piperidines, e.g. piperocaine
- A61K31/4523—Non condensed piperidines, e.g. piperocaine containing further heterocyclic ring systems
- A61K31/454—Non condensed piperidines, e.g. piperocaine containing further heterocyclic ring systems containing a five-membered ring with nitrogen as a ring hetero atom, e.g. pimozide, domperidone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/496—Non-condensed piperazines containing further heterocyclic rings, e.g. rifampin, thiothixene or sparfloxacin
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Indole Compounds (AREA)
Abstract
Description
技术领域technical field
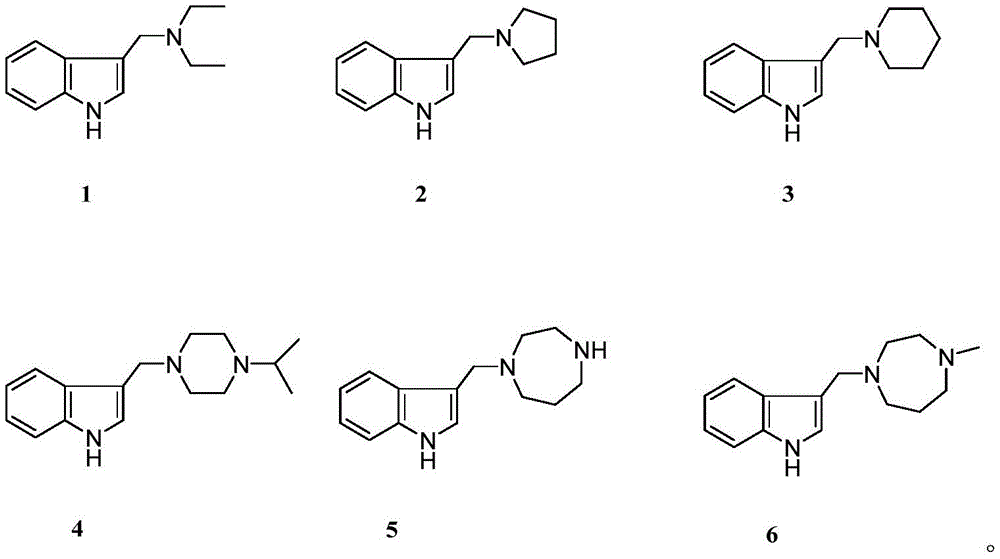
本发明属于药物技术领域,具体地说,涉及以结构式1-6所示芦竹碱衍生物为活性成分的药物组合物,它们的制备方法,以及该类衍生物及其药物组合物用作为褪黑素和5-羟色胺受体激动剂,它们在制备治疗或改善与褪黑素和五羟色胺受体相关的中枢神经系统疾病的药物中的应用。The invention belongs to the technical field of medicine, and in particular, relates to a pharmaceutical composition using the rhodophylline derivatives shown in structural formulas 1-6 as active ingredients, their preparation method, and the use of such derivatives and their pharmaceutical compositions as Melatonin and serotonin receptor agonists, and their use in preparing medicines for treating or improving central nervous system diseases related to melatonin and serotonin receptors.
背景技术Background technique
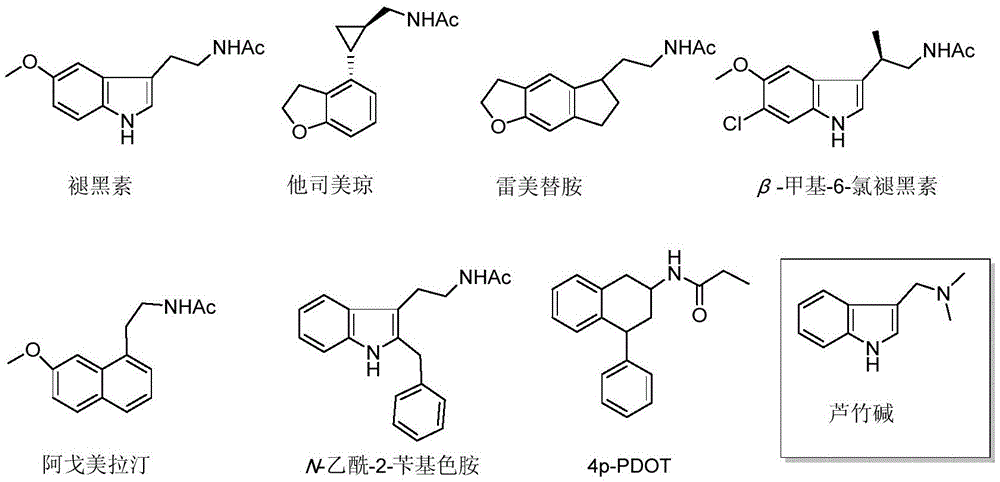
褪黑素(Melatonin)受体是松果体分泌的一种神经内分泌激素,由下丘脑交叉上核控制,可以调节昼夜节律,这种节律性波动表现为夜间达到高峰而白昼降至谷值。McCord和Allan于1917年发现牛松果体可以调节环境中Rana pipien蝌蚪皮肤的变化,并确定了褪黑素的结构为N-乙酰基-5-甲氧基色胺。按照褪黑激素功能和药理特性,褪黑素分为MT1,MT2和MT3三种亚型,这些受体被视为潜在的药物活性靶标。其中MT3受体亲和位点主要位于具有排毒属性的、人类同源染色体仓鼠的醌还原酶(QR2) 上。哺乳动物中存在的褪黑素受体亚型MT1和MT2已经被克隆,两个受体亚型同属于 G-偶联蛋白受体家族,分享一些特定的氨基酸序列,在氨基酸水平显示60%的同源性和独特的药理活性。目前,常见的褪黑素受体激动剂:他司美琼(Tasimelteon, VEC-162)、雷美替胺(Ramelteon,TAK-375)、β-甲基-6-氯褪黑素(LY-156735, TIK-301),阿戈美拉汀(Agomelatine)[11]等;常见的褪黑素受体拮抗剂:N-乙酰-2-苄基色胺(Luzindole),4-Phenyl-2-propionamidotetralin(4-P-PDOT)等。Melatonin receptor is a neuroendocrine hormone secreted by the pineal gland, which is controlled by the suprachiasmatic nucleus of the hypothalamus and can regulate the circadian rhythm, which is characterized by peaks at night and troughs during the day. McCord and Allan in 1917 discovered that the bovine pineal gland can regulate changes in the skin of the Rana pipien tadpole in the environment, and determined the structure of melatonin as N-acetyl-5-methoxytryptamine. Melatonin is divided into three subtypes, MT 1 , MT 2 and MT 3 , according to its functional and pharmacological properties, and these receptors are regarded as potential drug-active targets. Among them, the MT 3 receptor affinity site is mainly located on the quinone reductase (QR 2 ) of the human homologous hamster with detoxification properties. The melatonin receptor subtypes MT 1 and MT 2 present in mammals have been cloned. The two receptor subtypes belong to the G-coupled protein receptor family and share some specific amino acid sequences, showing 60 at the amino acid level. % homology and unique pharmacological activity. At present, common melatonin receptor agonists: tasimelteon (Tasimelteon, VEC-162), ramelteon (Ramelteon, TAK-375), β-methyl-6-chloromelatonin (LY- 156735, TIK-301), Agomelatine [11] , etc.; common melatonin receptor antagonists: N-acetyl-2-benzyltryptamine (Luzindole), 4-Phenyl-2-propionamidotetralin (4-P-PDOT) etc.
随着人们对天然产物研究的深入,越来越多的天然小分子引起药物化学家的兴趣。天然小分子芦竹碱(N,N-二甲基胺甲基吲哚)最初由Orekhov和Norkina从亚洲禾本科(Gramineae)芦竹属植物芦竹(Arundo Donax L.)中分离得到,具有与褪黑素受体相似的化学结构和广泛的生物活性:包括收缩血管,抗氧化,神经递质5-HT2A受体的拮抗剂等作用。芦竹碱可以抑制CVB3宿主细胞病变效应 (CPE),通过抑制早期CVB3病毒在宿主细胞的复制,作为一种抗 CVB3病毒药物[CN 105748474]。芦竹碱或者医药上可接受的盐作为活性成分对人体具有很好的皮肤美白,弹性,抗起皱或皮肤保湿效果 [KR 2016020240]。With the in-depth study of natural products, more and more natural small molecules have attracted the interest of medicinal chemists. The natural small molecule rhodophylline (N,N-dimethylaminomethyl indole) was originally isolated from the Asian Gramineae plant Arundo Donax L. by Orekhov and Norkina. Similar chemical structure and broad biological activity of melatonin receptor: including vasoconstrictor, antioxidant, antagonist of neurotransmitter 5-HT 2A receptor, etc. Lubaline can inhibit the CVB3 host cytopathic effect (CPE), by inhibiting the early CVB3 virus replication in host cells, as an anti-CVB3 virus drug [CN 105748474]. Lubaline or pharmaceutically acceptable salt as an active ingredient has good skin whitening, elasticity, anti-wrinkle or skin moisturizing effect on human body [KR 2016020240].
目前,本发明提供的芦竹碱衍生物(1-6)作为褪黑素和5-羟色胺受体激动剂并用于抗精神疾病有效成分的药物未见报道,其衍生物或其药物组合物作为褪黑素和5-羟色胺受体激动剂在制备或治疗抗精神疾病药物中未见报道。At present, there is no report on the rutabine derivatives (1-6) provided by the present invention as melatonin and serotonin receptor agonists and as medicines for antipsychotic active ingredients. Melatonin and serotonin receptor agonists have not been reported in the preparation or treatment of antipsychotic drugs.
发明内容SUMMARY OF THE INVENTION
本发明旨在提供一种新的具有药用价值的芦竹碱衍生物(1‐6)为活性成分的药物组合物,其制备方法,以及该类衍生物及其药物组合物作为褪黑素和5‐羟色胺受体激动剂,以及其在制备治疗或改善与褪黑素和5‐羟色胺受体相关的中枢神经系统疾病的药物中的应用。The present invention aims to provide a new pharmaceutical composition with medicinal value rudoline derivatives (1-6) as active ingredients, a preparation method thereof, and the derivatives and pharmaceutical compositions thereof as melatonin and serotonin receptor agonists, and their use in the manufacture of a medicament for the treatment or amelioration of central nervous system diseases associated with melatonin and serotonin receptors.
为了实现本发明的上述目的,本发明提供了如下的技术方案:In order to achieve the above-mentioned purpose of the present invention, the present invention provides the following technical solutions:
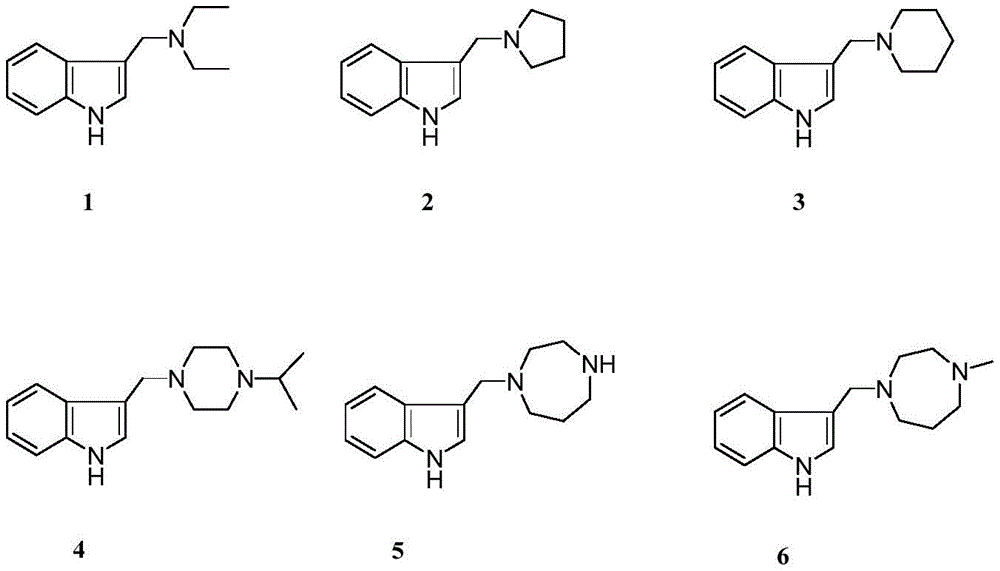
药物组合物,含有如下结构式所示的的芦竹碱衍生物1‐6,以及可药用载体或赋形剂。A pharmaceutical composition, comprising the rutabine derivatives 1-6 represented by the following structural formulas, and a pharmaceutically acceptable carrier or excipient.
本发明同时提供了制备所述的药物组合物中芦竹碱衍生物1‐6的方法,芦竹碱分别与乙胺、吡咯、哌啶、N‐异丙基哌嗪、N‐甲基高哌嗪、高哌嗪,于无水甲苯中反应,110℃回流至反应结束,减压回收溶剂,制备得到粗产品,硅胶柱层析纯化(二乙胺/甲醇/氯仿,v/v/v,2/4/94~3/5/92)制备得到目标化合物1‐6。At the same time, the present invention provides a method for preparing the
本发明还提供了所述的药物组合物在制备褪黑素和5‐羟色胺受体激动剂中的应用,以及所述的药物组合物在制备治疗或改善与褪黑素和5‐羟色胺受体相关的中枢神经系统疾病的药物中的应用。The present invention also provides the application of the pharmaceutical composition in the preparation of melatonin and serotonin receptor agonists, and the preparation of the pharmaceutical composition in the preparation of treatment or improvement with melatonin and serotonin receptors The use of medicines related to diseases of the central nervous system.
本发明另外还提供了如下结构式所示的的芦竹碱衍生物1‐6在制备治疗或改善与褪黑素和5‐羟色胺受体相关的中枢神经系统疾病的药物中的应用,以及芦竹碱衍生物1‐6在制备抗精神疾病药物中的应用,以及芦竹碱衍生物1‐6在制备褪黑素和5‐羟色胺受体激动剂中的应用。The present invention also provides the use of the rutabine derivatives 1-6 represented by the following structural formulas in the preparation of medicaments for treating or improving the diseases of the central nervous system related to melatonin and serotonin receptors, as well as the application of lucidum The application of base derivatives 1-6 in the preparation of antipsychotic drugs, and the application of rutabine derivatives 1-6 in the preparation of melatonin and serotonin receptor agonists.
以褪黑素和5‐羟色胺受体激动活性为指导发现芦竹碱衍生物1‐6具有抗精神疾病作用。本发明通过对芦竹碱的结构修饰得到6个衍生物具有较好激动活性,其中化合物2和6的活性较好,对褪黑素受体MT1的EC50值分别为0.51和0.50mΜ;对5‐HT1A的EC50值分别为0.28和0.23 mΜ。Guided by the agonistic activities of melatonin and serotonin receptors, rubarine derivatives 1-6 were found to have antipsychotic effects. The present invention obtains 6 derivatives by modifying the structure of luteolin and has better agonistic activity, wherein
本发明化合物用作药物时,可以直接使用,或者以药物组合物的形式使用。该药物组合物含有 0.1–99%,优选为0.5–90%的本发明化合物,其余为药物学上可接受的,对人和动物无毒和惰性的可药用载体或赋形剂。When the compound of the present invention is used as a medicine, it can be used directly or in the form of a pharmaceutical composition. The pharmaceutical composition contains 0.1-99%, preferably 0.5-90% of the compound of the present invention, and the rest are pharmaceutically acceptable, non-toxic and inert pharmaceutically acceptable carriers or excipients for humans and animals.
所述的药用载体或赋形剂是一种或多种固体、半固体和液体稀释剂、填料以及药物制品辅剂。将本发明的药物组合物以单位体重服用量的形式使用。本发明的药物可经注射(静注、肌注)和口服两种形式给药。The pharmaceutically acceptable carrier or excipient is one or more solid, semi-solid and liquid diluents, fillers and pharmaceutical preparation adjuvants. The pharmaceutical composition of the present invention is used in a dosage per body weight. The medicament of the present invention can be administered by injection (intravenous injection, intramuscular injection) and oral administration.
附图说明:Description of drawings:
图1为本发明芦竹碱衍生物(1-6)结构示意图。Fig. 1 is the structural schematic diagram of rhodophylline derivatives (1-6) of the present invention.
具体实施方式Detailed ways
为了更好地理解本发明的实质性内容,用本发明的实施例来说明本发明芦竹碱衍生物(1‐6)的制备方法和药理作用结果,但不以此来限定本发明。In order to better understand the essential content of the present invention, the examples of the present invention will be used to illustrate the preparation method and pharmacological effect results of the fenugreek derivatives (1-6) of the present invention, but the present invention is not limited by this.
下面实施例中,高分辨电喷雾电离质谱(HRESIMS)在LCMS‐IT‐TOF质谱仪(Shimadzu,Kyoto,Japan) 上测定,核磁共振谱(1H和13C NMR)由Bruker AM 400(1H/13C,400MHz/100MHz)核磁共振仪(Bruker, Bremerhaven,Germany)测定,以TMS(四甲基硅烷)为内标。柱色谱硅胶(200~300目)和薄层色谱硅胶GF254均为青岛美高集团有限公司生产。反应试剂购自Alfa Aesar、百灵威和Acros公司。In the following examples, high-resolution electrospray ionization mass spectrometry (HRESIMS) was measured on a LCMS-IT-TOF mass spectrometer (Shimadzu, Kyoto, Japan), and nuclear magnetic resonance spectra ( 1 H and 13 C NMR) were obtained by Bruker AM 400 ( 1 H / 13 C, 400MHz/100MHz) nuclear magnetic resonance apparatus (Bruker, Bremerhaven, Germany), with TMS (tetramethylsilane) as the internal standard. Column chromatography silica gel (200-300 mesh) and thin layer chromatography silica gel GF254 are produced by Qingdao Meigao Group Co., Ltd. Reagents were purchased from Alfa Aesar, Bailingwei and Acros.
化合物制备实施例1:Compound Preparation Example 1:
芦竹碱(2mmol)分别与乙胺、吡咯、哌啶、N‐异丙基哌嗪、N‐甲基高哌嗪及高哌嗪(2~10mmol) 溶于10mL无水甲苯,110℃回流至反应结束,减压回收溶剂,制备得到粗产品,硅胶柱层析(二乙胺/甲醇/氯仿,v/v/v,2/4/94~3/5/92)纯化得到目标化合物(1‐6):Lubaline (2mmol) and ethylamine, pyrrole, piperidine, N-isopropylpiperazine, N-methylhomopiperazine and homopiperazine (2-10mmol) were dissolved in 10mL of anhydrous toluene, refluxed at 110℃ To the end of the reaction, the solvent was recovered under reduced pressure to prepare a crude product, which was purified by silica gel column chromatography (diethylamine/methanol/chloroform, v/v/v, 2/4/94~3/5/92) to obtain the target compound ( 1‐6):
化合物1‐6结构确定数据:Structure determination data for compounds 1-6:
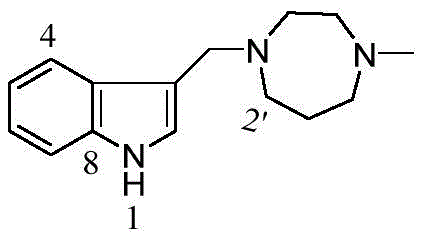
N,N‐二乙基‐3‐吲哚甲胺(N,N‐Diethyl‐3‐indolylmethylamine,1):白色粉末,收率97%.1H NMR (400MHz,CDCl3)δH:8.93(s,1H,NH),7.78‐7.00(m,5H,H‐2,4,5,6,7),3.87(s,2H,‐CH2N),2.69‐2.65(m,4H, N,N-Diethyl-3-indolylmethylamine (N,N-Diethyl-3-indolylmethylamine, 1): white powder, yield 97%. 1 H NMR (400 MHz, CDCl 3 ) δ H : 8.93 ( s,1H,NH),7.78-7.00(m,5H,H-2,4,5,6,7),3.87(s,2H,-CH 2 N),2.69-2.65(m,4H,
N‐吡咯基吲哚甲胺(N‐Pyrrolyl‐3‐indolylmethylamine,2):白色粉末,收率96%.1H NMR(400MHz, CDCl3)δH:9.27(s,1H,NH),7.75‐7.00(m,5H,H‐2,4,5,6,7),3.91(s,2H,CH2N),2.68(m,4H,H‐2',5'),Piperazinyl‐3‐indolylmethylamine,4):白色粉末,收率93%.1H NMR(400MHz,CDCl3)δH:9.57(s,1H,NH), 7.73‐6.94(m,5H,H‐2,4,5,6,7),3.78(s,2H,CH2N),2.69‐2.60(m,9H,H‐1”,2',3',5',6'),1.05(d,6H,Me). 13C NMR(100MHz,CDCl3)δ:136.2(s,C‐8),128.3(s,C‐9),124.4(d,C‐2),121.7(d,C‐6),119.3(d,C‐5),119.3 (d,C‐4),111.3(s,C‐3),111.2(d,C‐7),54.5(d,C‐1”),53.1(t,CH2N),53.1(t,C‐3',5'),48.7(t,C‐2',6'),18.7(q,7'),2.17(s,1H,NH),1.82‐1.76(m,2H,H‐3'),.13C NMR(100MHz,CDCl3)δ:136.2 (s,C‐8),128.1(s,C‐9),123.8(d,C‐2),121.7(d,C‐6),119.3(d,C‐5),119.2(d,‐3‐indolylmethylamine,6):黄色无定型粉末,收率80%.1H NMR(400MHz,CDCl3)δH:10.05(s,1H,NH),7.80‐7.02(m,5H,H‐2,4,5,6,7),3.89(s,2H,CH2N),2.89‐2.85(m,4H,H‐2',4'), 2.75‐2.69(m,4H,H‐6',7'),2.40(s,3H,NMe),1.89‐1.85(m,2H,H‐3').13C NMR(100MHz,CDCl3)δ:136.4(s, C‐8),128.1(s,C‐9),124.3(d,C‐2),121.6(d,C‐6),119.4(d,C‐5),119.1(d,C‐4),112.6(s,C‐3),111.3(d,C‐7),57.5(t,C‐6'),56.8(t,C‐7'),54.3(t,C‐4'),54.0(t,C‐2'),53.6(t,CH2N),46.9(q,NMe),27.0(t,C‐3').ESIMS: m/z 244[M+H]+,HRESIMS:C15H21N3[M+H]+测定值244.1742,计算值为244.1808.N-Pyrrolyl-3-indolylmethylamine, 2: white powder, yield 96%. 1 H NMR (400 MHz, CDCl 3 ) δ H : 9.27(s, 1H, NH), 7.75 ‐7.00(m,5H,H‐2,4,5,6,7),3.91(s,2H,CH 2 N),2.68(m,4H,H‐2',5'), Piperazinyl‐3‐indolylmethylamine, 4): white powder, yield 93%. 1 H NMR (400MHz, CDCl 3 ) δ H : 9.57(s, 1H, NH), 7.73‐6.94(m, 5H, H‐2, 4,5,6,7),3.78(s,2H,CH 2 N),2.69‐2.60(m,9H,H‐1",2',3',5',6'),1.05(d, 6H,Me). 13 C NMR (100MHz, CDCl 3 )δ: 136.2(s, C-8), 128.3(s, C-9), 124.4(d, C-2), 121.7(d, C-6 ), 119.3 (d, C-5), 119.3 (d, C-4), 111.3 (s, C-3), 111.2 (d, C-7), 54.5 (d, C-1”), 53.1 ( t, CH 2 N), 53.1(t, C-3', 5'), 48.7(t, C-2', 6'), 18.7(q, 7'), 2.17 (s, 1H, NH), 1.82-1.76 (m, 2H, H-3'), . 13 C NMR (100MHz, CDCl 3 )δ: 136.2 (s, C-8), 128.1 ( s,C‐9),123.8(d,C‐2),121.7(d,C‐6),119.3(d,C‐5),119.2(d, ‐3‐indolylmethylamine, 6): yellow amorphous powder, yield 80%. 1 H NMR (400MHz, CDCl 3 )δ H : 10.05(s,1H,NH),7.80-7.02(m,5H,H-2,4,5,6,7),3.89(s,2H,CH 2 N),2.89-2.85 (m,4H,H‐2',4'), 2.75‐2.69(m,4H,H‐6',7'),2.40(s,3H,NMe),1.89‐1.85(m,2H,H‐ 3'). 13 C NMR (100MHz, CDCl 3 )δ: 136.4(s, C-8), 128.1(s, C-9), 124.3(d, C-2), 121.6(d, C-6) ,119.4(d,C-5),119.1(d,C-4),112.6(s,C-3),111.3(d,C-7),57.5(t,C-6'),56.8(t ,C‐7'),54.3(t,C‐4'),54.0(t,C‐2'),53.6(t,CH 2 N),46.9(q,NMe),27.0(t,C‐3 ').ESIMS: m/z 244[M+H] + , HRESIMS: C 15 H 21 N 3 [M+H] + found 244.1742, calculated 244.1808.
下面以褪黑素和5‐羟色胺受体激动活性药理作用试验说明本发明的芦竹碱衍生物的药理作用结果:The pharmacological action results of the rubaline derivatives of the present invention are described below by the pharmacological action test of melatonin and serotonin receptor agonistic activity:
试验例1:Test Example 1:
本发明上述的化合物制备实施例制备得到的芦竹碱衍生物(1‐6)在人体肾上皮细胞MT1‐HEK293 和5‐HT1A‐HEK293细胞模型上,测定其对受体的激动活性。The rhodophylline derivatives (1-6) prepared in the above-mentioned compound preparation examples of the present invention were tested for their agonistic activity on receptors on human renal epithelial cell MT 1 -HEK293 and 5-HT 1A -HEK293 cell models.
1激动活性实验部分1 Agonistic activity experimental part
1.1材料和仪器1.1 Materials and Instruments
活性筛选使用的MT1和5‐HT1A细胞株分别对应人体肾上皮细胞MT1‐HEK293和 5‐HT1A‐HEK293,含有10%胎牛血清的细胞培养基(Dulbecco's Modified Eagle Medium,DMEM),10% FBS,HBSS购自GIBCO;褪黑素(CAS:73‐31‐4)购自Damas‐beta公司(Basel,瑞士);阳性对照药物阿戈美拉汀(CAS:138112‐76‐2)和5‐羟色胺(CAS:50‐67‐9)分别购自于萨恩化学技术(上海)有限公司和云南泽浩商贸有限公司;免洗钙8剂盒(Wash Free Fluo‐8Calcium Asaay Kit,HD03‐0010,HDB Biosciences Co.Ltd,上海,中国)。CO2恒温培养箱Thermo Forma 3310(美国);倒置生物显微镜XD‐101 型(南京);Flexstation 3台式多功能酶标仪(Molecular Devices,加利福尼亚,美国)。The MT 1 and 5-HT 1A cell lines used in the activity screening correspond to human renal epithelial cells MT 1 -HEK293 and 5-HT 1A -HEK293, respectively, in cell culture medium (Dulbecco's Modified Eagle Medium, DMEM) containing 10% fetal bovine serum, 10% FBS, HBSS were purchased from GIBCO; Melatonin (CAS: 73-31-4) was purchased from Damas-beta (Basel, Switzerland); positive control drug agomelatine (CAS: 138112-76-2) and 5-hydroxytryptamine (CAS: 50-67-9) were purchased from San Chemical Technology (Shanghai) Co., Ltd. and Yunnan Zehao Trading Co., Ltd. respectively; Wash Free Fluo-8 Calcium Asaay Kit, HD03 ‐0010, HDB Biosciences Co. Ltd, Shanghai, China). CO2 constant temperature incubator Thermo Forma 3310 (USA); Inverted biological microscope XD-101 (Nanjing); Flexstation 3 desktop multi-plate reader (Molecular Devices, California, USA).
1.2实验过程1.2 Experimental process
制备得到不同骨架有机酸托品酯,在人体肾上皮细胞MT1‐HEK293和5‐HT1A‐HEK293细胞模型上,以阿戈美拉汀和5‐羟色胺为阳性对照,对所合成衍生物进行MT1和5‐HT1A受体激动活性测定。 MT1和5‐HT1A受体激动活性用化合物对MT1和5‐HT1A受体激动率和半数有效浓度表达(EC50), EC50-药物对相应症状产生50%最大效应时的浓度。利用GraphPadSoftware公司开发的基础生物统计及绘图综合软件GraphPad Prism 5.0进行EC50计算,具体试验方法参考文献[17‐19]。采用 Dulbecco's改良的Eagle培养基,将细胞以4×104/孔的密度,在Matrigel包被的96孔黑壁透底板中,于CO2浓度为5%的37℃恒温培养箱中培养24h。吸取上清液然后,弃去原培养基,加入新鲜配置的染液100μl/孔,37℃避光60min。准备待测样品:配制不同浓度的待测样品,测试样品置入另一个透底板中。两块板同时放入FlexStation 3台式多功能酶标仪中。实验数据由Flex Station 3台式多功能酶标仪读取,EC50值通过Graph Pad Prism 5软件计算得到;激动率=Δδa/Δδ c×100%(a:测试样品;c:阳性对照)。The organic acid tropine ester with different backbones was prepared, and the synthesized derivatives were tested on human renal epithelial cell MT 1 -HEK293 and 5-HT 1A -HEK293 cell models, with agomelatine and serotonin as positive controls. MT 1 and 5-HT 1A receptor agonistic activity assay. MT 1 and 5-HT 1A receptor agonistic activity is expressed by compound on MT 1 and 5-HT 1A receptor agonism rate and half effective concentration (EC 50 ), EC 50 - the concentration at which the drug produces 50% of the maximal effect on the corresponding symptom . EC 50 was calculated by using GraphPad Prism 5.0, the basic biostatistics and drawing comprehensive software developed by GraphPad Software. Using Dulbecco's modified Eagle's medium, cells were cultured at a density of 4 × 104/well in Matrigel-coated 96-well black-walled permeable bottom plates in a 37°C incubator with a CO concentration of 5% for 24h. Aspirate the supernatant, then discard the original medium, add 100 μl/well of freshly prepared dye solution, and protect from light at 37°C for 60 min. Prepare the sample to be tested: prepare the sample to be tested with different concentrations, and place the test sample into another transparent bottom plate. Both plates were placed in the
2.结果:浓度为1.00mM,芦竹碱及其衍生物(1‐6)的激动活性如表(1)所示:2. Results: At a concentration of 1.00 mM, the agonistic activities of luteolin and its derivatives (1-6) are shown in Table (1):
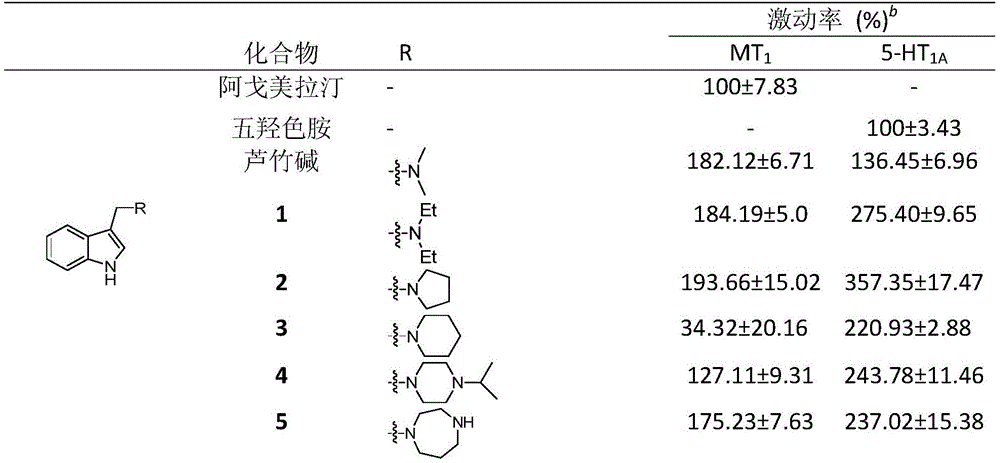
表1 芦竹碱衍生物对MT1和5‐HT1A受体的激动活性a Table 1 Agonistic activities of rubarine derivatives on MT 1 and 5-HT 1A receptors a
注:a所有测试化合物浓度约为1Mm,阿戈美拉汀测试浓度1.11μM,5‐羟色胺测试浓度6.67μM. 为三次测定的平均值,激动活性表达为(n=3).Notes: a All tested compounds were at a concentration of approximately 1 Mm, agomelatine was tested at a concentration of 1.11 μM, and serotonin was tested at a concentration of 6.67 μM. is the average of three determinations, and the agonistic activity is expressed as (n=3).
表2 衍生物2和6半数有效浓度(EC50,mM)Table 2 The half effective concentration of
注:化合物1‐6测试浓度范围为0.02~1.52μM;为三次测定的平均值,EC50表达为(n=3)。Note: The tested concentration range of compound 1-6 is 0.02-1.52 μM; is the mean of three determinations, and the EC50 is expressed as (n=3).
3.结论:实验结果显示,已测定的芦竹碱衍生物对MT1和5‐HT1A受体具有较强的激动活性,具有与褪黑素和受体相关的潜在的调节或治疗作用。3. Conclusion: The experimental results show that the determined rutabine derivatives have strong agonistic activity on MT 1 and 5-HT 1A receptors, and have potential regulatory or therapeutic effects related to melatonin and receptors.
制剂实施例1:Formulation Example 1:
按制备实施例1的方法制备得到的得芦竹碱衍生物1‐6,分别或混合用少量的DMSO溶解后,按常规加注射用水,精滤,灌封灭菌制成注射液。The rutabine derivatives 1-6 prepared according to the method of Preparation Example 1, respectively or mixed with a small amount of DMSO to dissolve, conventionally add water for injection, fine filtration, potting and sterilization to prepare an injection solution.
制剂实施例2:Formulation Example 2:
按制备实施例1的方法先制备得到芦竹碱衍生物1‐6,分别或混合,用少量的DMSO溶解后,将其溶于无菌注射用水中,搅拌使溶解,用无菌抽滤漏斗过滤,再无菌精滤,分装于安瓿中,低温冷冻干燥后无菌熔封得粉针剂。According to the method of Preparation Example 1, first prepare the derivatives 1-6 of rutabine, separately or mix them, dissolve them with a small amount of DMSO, dissolve them in sterile water for injection, stir to dissolve, and use a sterile suction filter funnel to dissolve them. Filtration, then sterile fine filtration, sub-packaging in ampoules, freeze-drying at low temperature, aseptic melting and sealing to obtain powder injection.
制剂实施例3:Formulation Example 3:
按制备实施例1的方法先制备得到芦竹碱衍生物1‐6,分别或混合,按其与赋形剂重量比为9:1 的比例加入赋形剂,制成粉剂。According to the method of Preparation Example 1, first prepare the rudbine derivatives 1-6, separate or mix, add excipients in a ratio of 9:1 by weight to excipients, and prepare powders.
制剂实施例4:Formulation Example 4:
按制备实施例1的方法先制备得到得到芦竹碱衍生物1‐6,分别或混合,按其与赋形剂重量比为 5:1的比例加入赋形剂,制粒压片。According to the method of Preparation Example 1, firstly prepare and obtain Pterostilbene derivatives 1-6, separately or mixed, add excipients in a ratio of 5:1 by weight to excipients, and granulate and press into tablets.
制剂实施例5:Formulation Example 5:
按制备实施例1的方法先制备得到的芦竹碱衍生物1‐6,分别或混合,按常规口服液制法制成口服液。According to the method of Preparation Example 1, the rudbine derivatives 1-6 obtained first were prepared separately or mixed, and made into an oral liquid according to the conventional oral liquid preparation method.
制剂实施例6:Formulation Example 6:
按制备实施例1的方法先制备得到的芦竹碱衍生物1‐6,分别或混合,按其与赋形剂重量比为5:1 的比例加入赋形剂,制成胶囊。According to the method of Preparation Example 1, the rudbine derivatives 1-6 were prepared separately or mixed, and the excipients were added in a ratio of 5:1 by weight to the excipients to prepare capsules.
制剂实施例7:Formulation Example 7:
按制备实施例1的方法先制得芦竹碱衍生物1‐6,分别或混合,按其与赋形剂重量比为3:1的比例加入赋形剂,制成胶囊。According to the method of Preparation Example 1, the rudbine derivatives 1-6 were prepared separately or mixed, and the excipients were added in a ratio of 3:1 by weight to the excipients to prepare capsules.
制剂实施例8:Formulation Example 8:
按制备实施例1的方法先制备得到的芦竹碱衍生物1‐6,分别或混合,按其与赋形剂重量比为5:1 的比例加入赋形剂,制成颗粒剂。According to the method of Preparation Example 1, the rudbine derivatives 1-6 obtained first were prepared separately or mixed, and the excipients were added in a ratio of 5:1 by weight to the excipients to prepare granules.
Claims (4)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201710092931.2A CN107174586B (en) | 2017-02-21 | 2017-02-21 | Pharmaceutical composition and application thereof using luteolin derivatives as active ingredients |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201710092931.2A CN107174586B (en) | 2017-02-21 | 2017-02-21 | Pharmaceutical composition and application thereof using luteolin derivatives as active ingredients |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN107174586A CN107174586A (en) | 2017-09-19 |
| CN107174586B true CN107174586B (en) | 2020-08-11 |
Family
ID=59830435
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201710092931.2A Active CN107174586B (en) | 2017-02-21 | 2017-02-21 | Pharmaceutical composition and application thereof using luteolin derivatives as active ingredients |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN107174586B (en) |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1720225A (en) * | 2002-11-28 | 2006-01-11 | 苏文生命科学有限公司 | N-arylsulfonyl-3-substituted indole with 5-HT receptor affinity, its preparation method and pharmaceutical composition containing it |
-
2017
- 2017-02-21 CN CN201710092931.2A patent/CN107174586B/en active Active
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1720225A (en) * | 2002-11-28 | 2006-01-11 | 苏文生命科学有限公司 | N-arylsulfonyl-3-substituted indole with 5-HT receptor affinity, its preparation method and pharmaceutical composition containing it |
Non-Patent Citations (3)
| Title |
|---|
| Indole;美国化学会;《Registry》;20090914;1-3 * |
| Oxytocic activity of basic (aminomethyl) derivatives of phenols and related compounds;COHEN A.等;《Brit.J.Pharmacol.》;19571231;第12卷;194-208 * |
| Structure-Activity Relationships at 5-HT1A Receptors: Binding Profiles and Intrinsic Activity;DAVID L. NELSON等;《Pharmacology Biochemistry & Behavio》;19911231;第40卷;1041-1051 * |
Also Published As
| Publication number | Publication date |
|---|---|
| CN107174586A (en) | 2017-09-19 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN109232439A (en) | For treating the alkyl derivative of virus infection and other disease | |
| KR101406727B1 (en) | Agomelatine hydrochloride hydrate and preparation thereof | |
| WO2018219281A1 (en) | 4-pyrimidinediamine small molecular organic compounds and derivatives thereof and use thereof | |
| CN107305202A (en) | The HPLC methods and impurity that analysis methanesulfonic acid pleasure is cut down for Buddhist nun and its preparation impurity make the purposes of reference standard | |
| CN110088092B (en) | Compounds, processes for their preparation, pharmaceutical compositions, uses of compounds and methods for treating psychiatric and/or sleep disorders | |
| WO2022129256A1 (en) | Selective hdac6 inhibitor | |
| WO2024193678A1 (en) | Crystal form of compound used as kinase inhibitor, preparation method therefor and use thereof | |
| CN107573348B (en) | Crystal form B of baricitinib trifluoroacetate and preparation method thereof | |
| CN108623601A (en) | The eutectic and preparation method thereof of Temozolomide and baicalein | |
| CN107174586B (en) | Pharmaceutical composition and application thereof using luteolin derivatives as active ingredients | |
| CN109809971B (en) | Poly-benzyl derivative, pharmaceutical composition thereof, preparation method and application thereof | |
| KR102738924B1 (en) | A novel compound for treating cancer in which kras g12d mutation exists and composition for preventing or treating cancer using the same | |
| CN108143741B (en) | Application of magnolol glucoside in preparation of medicine for treating central nervous system diseases | |
| CN108997121A (en) | Application of Magnolol Derivatives in the Preparation of Medicines for Treating Central Nervous System Diseases | |
| CN118176202B (en) | A crystal form of a cyano-substituted polypeptide compound and a preparation method thereof | |
| JP2014527524A (en) | 5,6,7,8-Tetrahydro-6- [N, N-bis [(2-thienyl) ethyl]] amino-1-naphthol, process for its preparation and use thereof | |
| CN114262305A (en) | Alkynyl-substituted thiazolinone-based-N-aryl benzamide compound and preparation method and application thereof | |
| CN106905300A (en) | Giantreed alkali derivant and its pharmaceutical composition and its application in pharmacy | |
| JP2025528495A (en) | TRKA(G667C) AND FLT3 TARGETED INHIBITORS AND COMPOSITIONS THEREOF WITH TUSIDINOSTAT | |
| CN105693819A (en) | Tetrahydroindole-4ketone tripeptide compound, preparation method and application of compound to anti-tumor drugs | |
| CN116253743B (en) | (+) -Paeovaitol derivative, pharmaceutical composition thereof, preparation method and application thereof | |
| CN116751186B (en) | Preparation and use of an estrogen receptor modulator | |
| CN116253704A (en) | Paeovaitol D derivative, pharmaceutical composition thereof, preparation method and application thereof | |
| CN112912373B (en) | Angiogenesis inhibitor, preparation method and application thereof | |
| CN111217777A (en) | A new preparation process of high-purity escitalopram pamoate |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |