CN106635974B - Separation and culture method of human umbilical cord mesenchymal stem cells - Google Patents
Separation and culture method of human umbilical cord mesenchymal stem cells Download PDFInfo
- Publication number
- CN106635974B CN106635974B CN201610910131.2A CN201610910131A CN106635974B CN 106635974 B CN106635974 B CN 106635974B CN 201610910131 A CN201610910131 A CN 201610910131A CN 106635974 B CN106635974 B CN 106635974B
- Authority
- CN
- China
- Prior art keywords
- umbilical cord
- pbs
- culture
- culture bottle
- temperature
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 210000003954 umbilical cord Anatomy 0.000 title claims abstract description 101
- 210000002901 mesenchymal stem cell Anatomy 0.000 title claims abstract description 29
- 238000000926 separation method Methods 0.000 title description 4
- 238000012136 culture method Methods 0.000 title description 3
- 108091003079 Bovine Serum Albumin Proteins 0.000 claims abstract description 34
- 102000004190 Enzymes Human genes 0.000 claims abstract description 34
- 108090000790 Enzymes Proteins 0.000 claims abstract description 34
- 239000001963 growth medium Substances 0.000 claims abstract description 27
- 239000007788 liquid Substances 0.000 claims abstract description 25
- 238000000034 method Methods 0.000 claims abstract description 23
- 238000012258 culturing Methods 0.000 claims abstract description 15
- 238000002791 soaking Methods 0.000 claims abstract description 14
- 239000006228 supernatant Substances 0.000 claims abstract description 13
- 239000012091 fetal bovine serum Substances 0.000 claims abstract description 11
- 238000005406 washing Methods 0.000 claims abstract description 10
- 239000006144 Dulbecco’s modified Eagle's medium Substances 0.000 claims abstract description 9
- 238000002156 mixing Methods 0.000 claims abstract description 8
- 238000010008 shearing Methods 0.000 claims abstract description 8
- 238000000338 in vitro Methods 0.000 claims abstract description 5
- UCSJYZPVAKXKNQ-HZYVHMACSA-N streptomycin Chemical compound CN[C@H]1[C@H](O)[C@@H](O)[C@H](CO)O[C@H]1O[C@@H]1[C@](C=O)(O)[C@H](C)O[C@H]1O[C@@H]1[C@@H](NC(N)=N)[C@H](O)[C@@H](NC(N)=N)[C@H](O)[C@H]1O UCSJYZPVAKXKNQ-HZYVHMACSA-N 0.000 claims description 98
- 239000008055 phosphate buffer solution Substances 0.000 claims description 80
- 239000000243 solution Substances 0.000 claims description 67
- 229960005322 streptomycin Drugs 0.000 claims description 49
- 229930182555 Penicillin Natural products 0.000 claims description 47
- JGSARLDLIJGVTE-MBNYWOFBSA-N Penicillin G Chemical compound N([C@H]1[C@H]2SC([C@@H](N2C1=O)C(O)=O)(C)C)C(=O)CC1=CC=CC=C1 JGSARLDLIJGVTE-MBNYWOFBSA-N 0.000 claims description 47
- 229940049954 penicillin Drugs 0.000 claims description 47
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims description 42
- 210000004027 cell Anatomy 0.000 claims description 33
- 229940088598 enzyme Drugs 0.000 claims description 32
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 32
- 239000002609 medium Substances 0.000 claims description 24
- 239000012894 fetal calf serum Substances 0.000 claims description 23
- 102000029816 Collagenase Human genes 0.000 claims description 16
- 108060005980 Collagenase Proteins 0.000 claims description 16
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 claims description 16
- 229960002424 collagenase Drugs 0.000 claims description 16
- 239000000203 mixture Substances 0.000 claims description 14
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 claims description 12
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 claims description 12
- 230000004927 fusion Effects 0.000 claims description 12
- 239000008103 glucose Substances 0.000 claims description 12
- 230000001079 digestive effect Effects 0.000 claims description 11
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 claims description 9
- 108010003272 Hyaluronate lyase Proteins 0.000 claims description 8
- 102000009066 Hyaluronoglucosaminidase Human genes 0.000 claims description 8
- 102000004157 Hydrolases Human genes 0.000 claims description 8
- 108090000604 Hydrolases Proteins 0.000 claims description 8
- 230000029087 digestion Effects 0.000 claims description 8
- 229960002989 glutamic acid Drugs 0.000 claims description 8
- 229960002773 hyaluronidase Drugs 0.000 claims description 8
- DAEPDZWVDSPTHF-UHFFFAOYSA-M sodium pyruvate Chemical compound [Na+].CC(=O)C([O-])=O DAEPDZWVDSPTHF-UHFFFAOYSA-M 0.000 claims description 8
- JKMHFZQWWAIEOD-UHFFFAOYSA-N 2-[4-(2-hydroxyethyl)piperazin-1-yl]ethanesulfonic acid Chemical compound OCC[NH+]1CCN(CCS([O-])(=O)=O)CC1 JKMHFZQWWAIEOD-UHFFFAOYSA-N 0.000 claims description 7
- 239000007995 HEPES buffer Substances 0.000 claims description 7
- 238000001816 cooling Methods 0.000 claims description 6
- 235000011389 fruit/vegetable juice Nutrition 0.000 claims description 6
- 230000010355 oscillation Effects 0.000 claims description 6
- 239000011780 sodium chloride Substances 0.000 claims description 6
- 210000000130 stem cell Anatomy 0.000 claims description 6
- 230000001464 adherent effect Effects 0.000 claims description 4
- 238000010009 beating Methods 0.000 claims description 4
- 238000007664 blowing Methods 0.000 claims description 4
- 229940054269 sodium pyruvate Drugs 0.000 claims description 4
- 230000003068 static effect Effects 0.000 claims description 4
- 210000004369 blood Anatomy 0.000 claims description 3
- 239000008280 blood Substances 0.000 claims description 3
- 125000000291 glutamic acid group Chemical group N[C@@H](CCC(O)=O)C(=O)* 0.000 claims description 3
- 238000007789 sealing Methods 0.000 claims description 3
- 239000002904 solvent Substances 0.000 claims description 3
- 230000003321 amplification Effects 0.000 abstract description 8
- 238000003199 nucleic acid amplification method Methods 0.000 abstract description 8
- 230000000694 effects Effects 0.000 abstract description 2
- 238000004321 preservation Methods 0.000 abstract 1
- 238000010257 thawing Methods 0.000 abstract 1
- 238000009489 vacuum treatment Methods 0.000 abstract 1
- 238000002360 preparation method Methods 0.000 description 17
- 239000008367 deionised water Substances 0.000 description 8
- 229910021641 deionized water Inorganic materials 0.000 description 8
- 102100022464 5'-nucleotidase Human genes 0.000 description 7
- 101000678236 Homo sapiens 5'-nucleotidase Proteins 0.000 description 7
- 239000000523 sample Substances 0.000 description 7
- 238000005303 weighing Methods 0.000 description 5
- 239000000047 product Substances 0.000 description 4
- 238000003756 stirring Methods 0.000 description 4
- 239000000427 antigen Substances 0.000 description 3
- 108091007433 antigens Proteins 0.000 description 3
- 102000036639 antigens Human genes 0.000 description 3
- 238000012512 characterization method Methods 0.000 description 3
- 239000003550 marker Substances 0.000 description 3
- 239000013074 reference sample Substances 0.000 description 3
- 239000000725 suspension Substances 0.000 description 3
- ZDXPYRJPNDTMRX-VKHMYHEASA-N L-glutamine Chemical compound OC(=O)[C@@H](N)CCC(N)=O ZDXPYRJPNDTMRX-VKHMYHEASA-N 0.000 description 2
- 229930182816 L-glutamine Natural products 0.000 description 2
- FCPVYOBCFFNJFS-LQDWTQKMSA-M benzylpenicillin sodium Chemical compound [Na+].N([C@H]1[C@H]2SC([C@@H](N2C1=O)C([O-])=O)(C)C)C(=O)CC1=CC=CC=C1 FCPVYOBCFFNJFS-LQDWTQKMSA-M 0.000 description 2
- 238000000684 flow cytometry Methods 0.000 description 2
- 239000002994 raw material Substances 0.000 description 2
- 239000011734 sodium Substances 0.000 description 2
- 102000008186 Collagen Human genes 0.000 description 1
- 108010035532 Collagen Proteins 0.000 description 1
- 239000007836 KH2PO4 Substances 0.000 description 1
- 229930040373 Paraformaldehyde Natural products 0.000 description 1
- 230000003698 anagen phase Effects 0.000 description 1
- 239000007640 basal medium Substances 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 230000007910 cell fusion Effects 0.000 description 1
- 230000010261 cell growth Effects 0.000 description 1
- 238000002659 cell therapy Methods 0.000 description 1
- 238000005119 centrifugation Methods 0.000 description 1
- 229920001436 collagen Polymers 0.000 description 1
- 230000004069 differentiation Effects 0.000 description 1
- 210000003981 ectoderm Anatomy 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 210000003754 fetus Anatomy 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 238000001415 gene therapy Methods 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 210000003716 mesoderm Anatomy 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 229910000402 monopotassium phosphate Inorganic materials 0.000 description 1
- 229920002866 paraformaldehyde Polymers 0.000 description 1
- 239000002504 physiological saline solution Substances 0.000 description 1
- 210000001778 pluripotent stem cell Anatomy 0.000 description 1
- 230000001954 sterilising effect Effects 0.000 description 1
- 239000002699 waste material Substances 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0652—Cells of skeletal and connective tissues; Mesenchyme
- C12N5/0662—Stem cells
- C12N5/0668—Mesenchymal stem cells from other natural sources
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2500/00—Specific components of cell culture medium
- C12N2500/30—Organic components
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2500/00—Specific components of cell culture medium
- C12N2500/30—Organic components
- C12N2500/32—Amino acids
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2500/00—Specific components of cell culture medium
- C12N2500/30—Organic components
- C12N2500/34—Sugars
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2500/00—Specific components of cell culture medium
- C12N2500/60—Buffer, e.g. pH regulation, osmotic pressure
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2509/00—Methods for the dissociation of cells, e.g. specific use of enzymes
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Biomedical Technology (AREA)
- Zoology (AREA)
- Developmental Biology & Embryology (AREA)
- Organic Chemistry (AREA)
- Biotechnology (AREA)
- Chemical & Material Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Genetics & Genomics (AREA)
- Wood Science & Technology (AREA)
- Microbiology (AREA)
- Cell Biology (AREA)
- Biochemistry (AREA)
- General Engineering & Computer Science (AREA)
- General Health & Medical Sciences (AREA)
- Rheumatology (AREA)
- Medicines Containing Material From Animals Or Micro-Organisms (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
Abstract
The invention discloses a method for separating and culturing human umbilical cord mesenchymal stem cells, which comprises the following steps: fresh in vitro umbilical cord of a newborn → low-temperature PBS liquid preservation, washing → shearing in low-temperature soaking liquid → low-temperature PBS liquid washing, low-temperature vacuum treatment → thawing → PBS liquid soaking → first mixed enzyme solution adding, vibration enzymolysis treatment → second mixed enzyme solution adding, vibration enzymolysis treatment → fetal bovine serum, PBS liquid and LG-DMEM culture medium adding, standing and then centrifuging, removing supernatant → adding PBS liquid, standing and then centrifuging, removing supernatant → adding fetal bovine serum, PBS liquid and LG-DMEM culture medium, mixing and → amplification culture; has the effect of improving the purity of the umbilical cord mesenchymal stem cells.
Description
Technical Field
The invention relates to a preparation technology of mesenchymal stem cells, in particular to a separation and culture method of human umbilical cord mesenchymal stem cells.
Background
Mesenchymal Stem Cells (MSCs) are a class of pluripotent stem cells with high self-renewal capacity and multipotent differentiation potential derived from early-developing mesoderm and ectoderm, which can be continuously subcultured and cryopreserved under suitable conditions in vitro. Since MSCs can differentiate into different cells under specific conditions, they are becoming a source of cells of great practical value in the fields of cell therapy and gene therapy. The umbilical cord belongs to extra-embryonic tissues, contains a large number of stem cells differentiated in multiple directions, has high cell growth and amplification speed, becomes waste after the fetus is delivered, is convenient to obtain materials, has rich sources, has no ethical problems, and can be used as an excellent source of mesenchymal stem cells.
Chinese patent publication No. CN101643719A, published as 2010, 2/10/discloses a simplified method for isolated culture of umbilical cord mesenchymal stem cells, which comprises the following steps: umbilical cord removal, physiological saline washing → shearing → centrifugation and supernatant removal → tissue mass culture with 10% α MEM, fluid exchange every 3 days → passage until about 80% of cells fuse about 10 days → passage every 3-5 days thereafter.
The research on the phenotype identification of the cells shows that the purity of the umbilical cord mesenchymal stem cells obtained by the separation and culture of the method still needs to be improved.
Disclosure of Invention
The invention aims to provide a method for separating and culturing human umbilical cord mesenchymal stem cells, which has the effect of improving the purity of the umbilical cord mesenchymal stem cells.
The technical purpose of the invention is realized by the following technical scheme:
a method for separating and culturing human umbilical cord mesenchymal stem cells comprises the following steps:
i. taking a fresh in-vitro umbilical cord of a newborn, placing the umbilical cord in PBS (phosphate buffer solution) which is controlled at the temperature of 2-5 ℃ and contains 100U/ml penicillin and 100U/ml streptomycin, and preserving for later use;
ii, taking out the umbilical cord treated in the step i, and washing the umbilical cord for multiple times by using PBS (phosphate buffer solution) with the temperature controlled at 2-5 ℃ and containing 100U/ml penicillin and 100U/ml streptomycin until no blood stain remains;
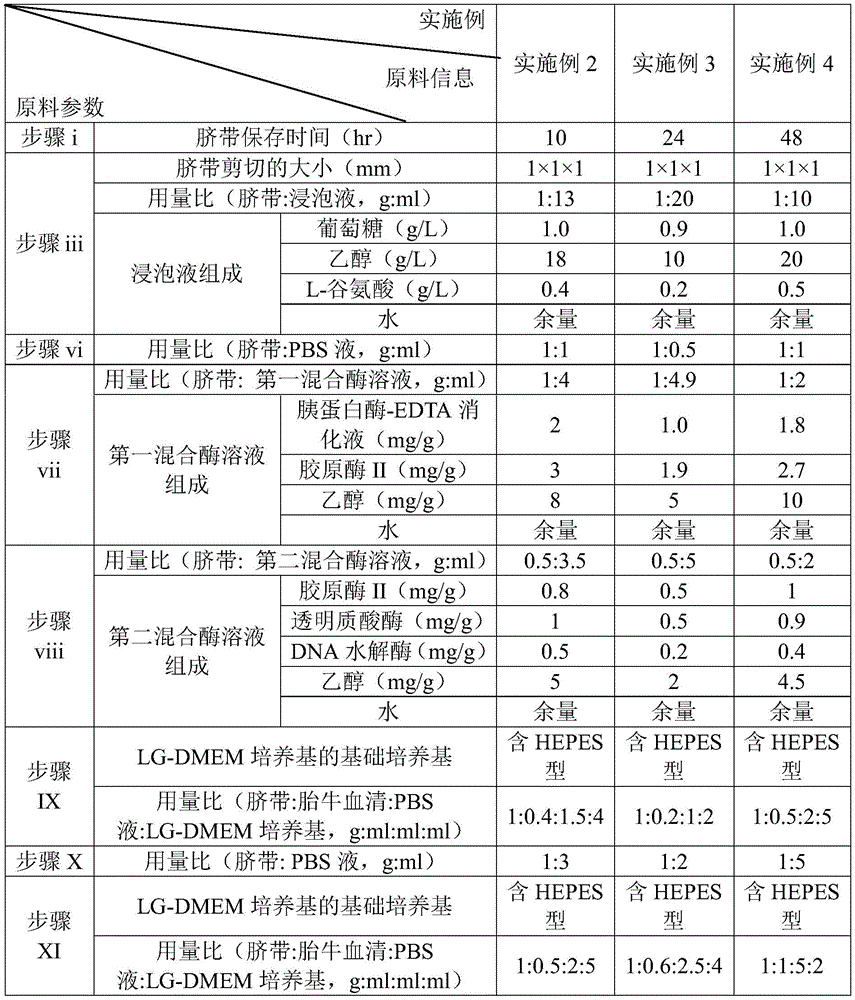
iii, shearing the washed umbilical cord in a soaking solution environment with the temperature controlled at 2-5 ℃, and removing arteriovenous in the shearing process; the using amount ratio of the umbilical cord to the soaking solution is 1g: 10-20 ml, the soaking solution comprises 0.9-1.0 g/L of glucose, 10-20 g/L of ethanol and the balance of water;
iv, taking the cut umbilical cord, washing the umbilical cord for 2-3 times by using PBS (phosphate buffer solution) which is controlled at the temperature of 2-5 ℃ and contains 100U/ml penicillin and 100U/ml streptomycin, putting the umbilical cord into a culture bottle, placing the culture bottle in an environment with the temperature controlled at-80-70 ℃ and the vacuum degree controlled at 10-30 Pa for 5-20 min, and removing the solvent on the umbilical cord;
v, taking the culture bottle for sealing treatment, placing the culture bottle in an environment with the temperature controlled between-40 ℃ and-35 ℃, transferring the culture bottle to the environment with the temperature controlled between-5 ℃ and 0 ℃ when the temperature of the culture bottle is increased to between-40 ℃ and-35 ℃, and transferring the culture bottle to the environment with the temperature controlled between 37 ℃ when the temperature of the culture bottle is increased to between-5 ℃ and 0 ℃ until the umbilical cord in the culture bottle is completely thawed;
taking the culture bottle treated in the step v, and adding PBS (phosphate buffer solution) containing 100U/ml penicillin and 100U/ml streptomycin into the culture bottle; wherein the dosage ratio of the umbilical cord to the PBS is 1g: 0.5-1 ml, and the umbilical cord and the PBS are fully contacted by oscillating for 1-5 min;
and vii, taking the culture bottle treated by the step vi, adding a first mixed enzyme solution into the culture bottle, and oscillating the culture bottle, wherein the temperature control curve of a water bath for oscillation is as follows: 0-5min, constant temperature of 15 ℃ → 5.01-20min, uniform temperature rise to 25 → 20.01-30min, constant temperature of 25 → 30.01-40min, constant temperature rise to 37 → 40.01-75min, constant temperature of 37 ℃ → 75.01-90min, natural cooling to 25 ℃; the dosage ratio of the umbilical cord to the first mixed enzyme solution is 1g: 2-5 ml, the first mixed enzyme solution comprises 1-2 mg/g of trypsin-EDTA digestive juice, 2-3 mg/g of collagenase II, 5-10 mg/g of ethanol, and the balance of water;
and viii, taking the culture bottle treated by the step vii, adding a second mixed enzyme solution into the culture bottle, and oscillating the culture bottle, wherein the temperature control curve of a water bath for oscillation is as follows: 0-2min, constant temperature of 25 → 2.01-10min, uniform temperature rise to 30 ℃ → 10.01-15min, constant temperature of 30 ℃ → 15.01-20min, constant temperature rise to 37 ℃ → 20.01-60min, constant temperature of 37 ℃ → 60.01-75min, natural cooling to 25 ℃; the dosage ratio of the umbilical cord to the second mixed enzyme solution is 0.5g: 2-5 ml, the second mixed enzyme solution comprises 0.5-1 mg/g of collagenase II, 0.5-1 mg/g of hyaluronidase, 0.2-0.5 mg/g of DNA hydrolase, 2-5 mg/g of ethanol, and the balance of water;
IX. taking the culture bottle treated by the step viii, adding fetal calf serum, PBS containing 100U/ml penicillin and 100U/ml streptomycin, LG-DMEM culture medium containing 100U/ml penicillin and 0.1g/L streptomycin, standing for 2-5 min at 20-25 ℃, and centrifuging for 5-8 min at the temperature; wherein the dosage ratio of the umbilical cord, the fetal calf serum, the PBS liquid and the LG-DMEM culture medium is 1g: 0.2-0.5 ml: 1-2 ml: 2-5 ml;
x, adding the culture bottle treated in the step IX into PBS containing 100U/ml penicillin and 100U/ml streptomycin, standing for 2-5 min at 20-25 ℃, centrifuging for 5-8 min at the temperature, and removing the supernatant; wherein the dosage ratio of the umbilical cord to the PBS is 1g: 2-5 ml;
XI, adding fetal calf serum, PBS (phosphate buffer solution) containing 100U/ml penicillin and 100U/ml streptomycin and LG-DMEM (glycerol-free medium) containing 100U/ml penicillin and 100U/ml streptomycin into the culture bottle treated in the step X, standing for 2-5 min at 20-25 ℃, centrifuging for 5-8 min at the temperature, removing the supernatant, and repeating for 2-3 times; wherein the dosage ratio of the umbilical cord, the fetal calf serum, the PBS liquid and the LG-DMEM culture medium is 1g: 0.5-1 ml: 2-5 ml;
XII, taking the culture bottle treated in the step XI, adding fetal calf serum, PBS (phosphate buffer solution) containing 100U/ml penicillin and 100U/ml streptomycin and LG-DMEM (glycerol modified eagle medium) containing 100U/ml penicillin and 100U/ml streptomycin, and uniformly mixing; wherein the dosage ratio of the umbilical cord, the fetal calf serum, the PBS liquid and the LG-DMEM culture medium is 1g:0.5ml:1ml:1 ml;
XIII, 100. mu.L of the sample treated in step XII was sampled and 0.5ml of fetal bovine serum, 12ml of DMEM/F12 medium and 1mg of Na were added to itCl, in 5% by volume CO at 37 ℃2Standing and culturing in an environment with the humidity of 95% RH; changing the solution after 4 days, removing the non-adherent cells, and changing every 2 days; after the cells in the culture dish reach 80% fusion, removing the culture solution, rinsing with PBS for 1 time, adding trypsin-EDTA digestive liquid at 37 ℃ for digestion for 2min, slightly beating the bottle wall to promote the cells to be separated from the bottle bottom, then adding PBS containing 100U/ml penicillin and 100U/ml streptomycin, LG-DMEM culture medium containing 100U/ml penicillin and 100U/ml streptomycin to stop digestion, collecting the cells obtained after blowing, centrifuging, discarding the supernatant, mixing the mixture again with 0.5ml fetal calf serum, 12ml DMEM/F12 culture medium and 1mg NaCl, subculturing at the ratio of 1:2, placing the mixture at 37 ℃ and 5% of CO by volume fraction2And carrying out static culture in an environment with the humidity of 95% RH, replacing the culture solution for 1 time every 2 days, and digesting and passaging by the same method when the primary stem cells in the culture dish reach 80% fusion.
The cell phenotype identification research shows that the fusion time of 80% of the cells can be shortened to be within 5 days during the amplification culture in the technical scheme, so that the culture speed is increased; and the positive rate of mouse anti-human PE labeled CD73 (as a marker antigen of mesenchymal stem cells) is improved to over 91 percent, the purity of the mesenchymal stem cells is improved, and the product quality is improved.
More preferably: treating the umbilical cord treated in step i with step ii within 48 hr.
More preferably: in step iii, the cord is cut to a size of 1mm × 1mm × 1 mm.
More preferably: in the step iii, the soaking solution further comprises 0.2-0.5 g/L of L-glutamic acid.
More preferably: the soak solution comprises 1.0g/L glucose, 0.4 g/L-glutamic acid, 18g/L ethanol and the balance of water.
More preferably: in step vi, the dosage ratio of the umbilical cord to the PBS is 1g:1 ml.
More preferably: in step vii, the dosage ratio of the umbilical cord and the first mixed enzyme solution is 1g:4ml, and the first mixed enzyme solution comprises 2mg/g trypsin-EDTA digestive juice, 3mg/g collagenase II, 8mg/g ethanol, and the balance of water;
in step viii, the dosage ratio of the umbilical cord and the second mixed enzyme solution is 0.5g:3.5ml, and the second mixed enzyme solution comprises 0.8mg/g collagenase II, 1mg/g hyaluronidase, 0.5mg/g DNA hydrolase, 5mg/g ethanol, and the balance of water.
More preferably: in the step IX, the dosage ratio of the umbilical cord, the fetal calf serum, the PBS and the LG-DMEM medium is 1g:0.4ml:1.5ml:4 ml;
in the step X, the dosage ratio of the umbilical cord to the PBS is 1g to 3 ml;
in the step XI, the dosage ratio of the umbilical cord, the fetal calf serum, the PBS liquid and the LG-DMEM medium is 1g:0.5ml:2ml:5 ml.
More preferably: the basic culture medium of the LG-DMEM culture medium comprises 1000mg/L glucose, 4.0mM L-glutamic acid and 110mg/L sodium pyruvate.
More preferably: the basic medium of the LG-DMEM medium further comprises 25mM HEPES.
The cell phenotype identification research shows that the fusion time of the cells reaching 80 percent can be further shortened to be within 3 days when the cells are subjected to amplification culture by the technical scheme, so that the culture speed is further increased; and the positive rate of mouse anti-human PE labeled CD73 (as a marker antigen of mesenchymal stem cells) is further improved to more than 98%, the purity of the mesenchymal stem cells is further improved, and the product quality is improved.
In conclusion, the invention has the following beneficial effects:
the cell phenotype identification research shows that when the cell is subjected to amplification culture, the amplification culture speed is high, the purity of the obtained mesenchymal stem cells is high, and the product quality is improved to a certain extent, wherein the fusion time of the cells reaches 80% within 5 days, the positive rate of the mouse anti-human PE labeled CD73 is higher than 91%, the fusion time of the best sample reaches 80% within 3 days, and the positive rate of the mouse anti-human PE labeled CD73 is higher than 98%.
Detailed Description
The present embodiment is only for explaining the present invention, and it is not limited to the present invention, and those skilled in the art can make modifications of the present embodiment without inventive contribution as needed after reading the present specification, but all of them are protected by patent law within the scope of the present invention.
Example 1: preparation of various raw materials
(1) Umbilical cord: collected from obstetrical and gynecological delivery rooms in hospitals.
(2) PBS containing 100U/ml penicillin and 100U/ml streptomycin:
weighing 1.42g Na2HPO4、0.24g KH2PO4Adding 8g of NaCl and 0.2g of KCl into 800ml of deionized water, stirring and dissolving, adding 0.06g of penicillin sodium and 0.1g of streptomycin, adjusting the pH value of the solution to 7.4 by using HCl, adding deionized water to a constant volume of 1L, sterilizing at high temperature and high pressure, and storing at room temperature; the converted effective price of penicillin and streptomycin in the solution is 100U/ml and 100U/ml respectively;
of these, penicillin sodium and streptomycin were purchased from Gibco.
(3) Soaking liquid:
weighing a certain amount of glucose, (part of the glucose is added with L-glutamic acid) and ethanol as required, adding part of deionized water, stirring and dissolving, and finally fixing the volume with the deionized water to obtain the glucose-glutamic acid-glutamic; see the specific examples for specific concentrations of each component.
(4) First mixed enzyme solution
Weighing a certain amount of trypsin-EDTA digestive juice, collagenase II and ethanol according to the requirement, adding part of deionized water, stirring and dissolving, and finally fixing the volume by using the deionized water to obtain the composition; see the specific examples for specific concentrations of each component, wherein trypsin-EDTA digest and collagenase II were both purchased from Gibco.
(5) Second mixed enzyme solution
Weighing a certain amount of collagenase II, hyaluronidase, DNA hydrolase and ethanol, adding a part of deionized water, stirring and dissolving, and finally fixing the volume with the deionized water to obtain the collagen; see the specific examples for specific concentrations of each component, wherein collagenase II is available from Gibco, and hyaluronidase and DNA hydrolase are both available from Amresco, usa.
(6) Basic culture medium of LG-DMEM (Ligno-modified eagle-medium) culture medium
Purchased from Gibco, and of two types, respectively HEPES-containing and HEPES-free,
wherein, the glucose in the HEPES type is 1000mg/L, the L-glutamine is 4.0mM, the sodium pyruvate is 110mg/L, and the HEPES is 25 mM;
the HEPES-free type had glucose of 1000mg/L, L-glutamine of 4.0mM, sodium pyruvate of 110mg/L and it contained no HEPES.
(7) Fetal bovine serum: purchased from Hyclone.
(8) LG-DMEM medium containing 100U/ml penicillin and 0.1g/L streptomycin, or LG-DMEM medium containing 100U/ml penicillin and 100U/ml streptomycin
Weighing a certain amount of penicillin and streptomycin with the same source as the penicillin and streptomycin in the step (2) according to the requirement, adding the basic culture medium of the LG-DMEM culture medium in the step (6), uniformly dispersing and fixing the volume.
Examples 2 to 4: a method for separating and culturing human umbilical cord mesenchymal stem cells comprises the following steps:
i. taking a fresh in-vitro umbilical cord of a newborn, placing the umbilical cord in PBS (phosphate buffer solution) which is controlled at the temperature of 2-5 ℃ and contains 100U/ml penicillin and 100U/ml streptomycin, and preserving for later use;
ii, taking out the umbilical cord treated in the step i, and washing the umbilical cord for multiple times by using PBS (phosphate buffer solution) with the temperature controlled at 2-5 ℃ and containing 100U/ml penicillin and 100U/ml streptomycin until no blood stain remains;
iii, shearing the washed umbilical cord in a soaking solution environment with the temperature controlled at 2-5 ℃, and removing arteriovenous in the shearing process; the using amount ratio of the umbilical cord to the soaking solution is 1g: 10-20 ml, and the soaking solution comprises 0.9-1.0 g/L glucose, 0.2-0.5 g/L-glutamic acid, 10-20 g/L ethanol and the balance of water;
iv, taking the cut umbilical cord, washing the umbilical cord for 2-3 times by using PBS (phosphate buffer solution) which is controlled at the temperature of 2-5 ℃ and contains 100U/ml penicillin and 100U/ml streptomycin, putting the umbilical cord into a culture bottle, placing the culture bottle in an environment with the temperature controlled at-80-70 ℃ and the vacuum degree controlled at 10-30 Pa for 5-20 min, and removing the solvent on the umbilical cord;
v, taking the culture bottle for sealing treatment, placing the culture bottle in an environment with the temperature controlled between-40 ℃ and-35 ℃, transferring the culture bottle to the environment with the temperature controlled between-5 ℃ and 0 ℃ when the temperature of the culture bottle is increased to between-40 ℃ and-35 ℃, and transferring the culture bottle to the environment with the temperature controlled between 37 ℃ when the temperature of the culture bottle is increased to between-5 ℃ and 0 ℃ until the umbilical cord in the culture bottle is completely thawed;
taking the culture bottle treated in the step v, and adding PBS (phosphate buffer solution) containing 100U/ml penicillin and 100U/ml streptomycin into the culture bottle; wherein the dosage ratio of the umbilical cord to the PBS is 1g: 0.5-1 ml, and the umbilical cord and the PBS are fully contacted by oscillating for 1-5 min;
and vii, taking the culture bottle treated by the step vi, adding a first mixed enzyme solution into the culture bottle, and oscillating the culture bottle, wherein the temperature control curve of a water bath for oscillation is as follows: 0-5min, constant temperature of 15 ℃ → 5.01-20min, uniform temperature rise to 25 → 20.01-30min, constant temperature of 25 → 30.01-40min, constant temperature rise to 37 → 40.01-75min, constant temperature of 37 ℃ → 75.01-90min, natural cooling to 25 ℃; the dosage ratio of the umbilical cord to the first mixed enzyme solution is 1g: 2-5 ml, the first mixed enzyme solution comprises 1-2 mg/g of trypsin-EDTA digestive juice, 2-3 mg/g of collagenase II, 5-10 mg/g of ethanol, and the balance of water;
and viii, taking the culture bottle treated by the step vii, adding a second mixed enzyme solution into the culture bottle, and oscillating the culture bottle, wherein the temperature control curve of a water bath for oscillation is as follows: 0-2min, constant temperature of 25 → 2.01-10min, uniform temperature rise to 30 ℃ → 10.01-15min, constant temperature of 30 ℃ → 15.01-20min, constant temperature rise to 37 ℃ → 20.01-60min, constant temperature of 37 ℃ → 60.01-75min, natural cooling to 25 ℃; the dosage ratio of the umbilical cord to the second mixed enzyme solution is 0.5g: 2-5 ml, the second mixed enzyme solution comprises 0.5-1 mg/g of collagenase II, 0.5-1 mg/g of hyaluronidase, 0.2-0.5 mg/g of DNA hydrolase, 2-5 mg/g of ethanol, and the balance of water;
IX. taking the culture bottle treated by the step viii, adding fetal calf serum, PBS containing 100U/ml penicillin and 100U/ml streptomycin, LG-DMEM culture medium containing 100U/ml penicillin and 0.1g/L streptomycin, standing for 2-5 min at 20-25 ℃, and centrifuging for 5-8 min at the temperature; wherein the dosage ratio of the umbilical cord, the fetal calf serum, the PBS liquid and the LG-DMEM culture medium is 1g: 0.2-0.5 ml: 1-2 ml: 2-5 ml;
x, adding the culture bottle treated in the step IX into PBS containing 100U/ml penicillin and 100U/ml streptomycin, standing for 2-5 min at 20-25 ℃, centrifuging for 5-8 min at the temperature, and removing the supernatant; wherein the dosage ratio of the umbilical cord to the PBS is 1g: 2-5 ml;
XI, adding fetal calf serum, PBS (phosphate buffer solution) containing 100U/ml penicillin and 100U/ml streptomycin and LG-DMEM (glycerol-free medium) containing 100U/ml penicillin and 100U/ml streptomycin into the culture bottle treated in the step X, standing for 2-5 min at 20-25 ℃, centrifuging for 5-8 min at the temperature, removing the supernatant, and repeating for 2-3 times; wherein the dosage ratio of the umbilical cord, the fetal calf serum, the PBS liquid and the LG-DMEM culture medium is 1g: 0.5-1 ml: 2-5 ml;
XII, taking the culture bottle treated in the step XI, adding fetal calf serum, PBS (phosphate buffer solution) containing 100U/ml penicillin and 100U/ml streptomycin and LG-DMEM (glycerol modified eagle medium) containing 100U/ml penicillin and 100U/ml streptomycin, and uniformly mixing; wherein the dosage ratio of the umbilical cord, the fetal calf serum, the PBS liquid and the LG-DMEM culture medium is 1g:0.5ml:1ml:1 ml;
XIII, 100. mu.L of the sample treated in step XII was sampled, 0.5ml of fetal bovine serum, 12ml of DMEM/F12 medium and 1mg of NaCl were added thereto, and the mixture was placed at 37 ℃ in 5% by volume CO2Standing and culturing in an environment with the humidity of 95% RH; changing the solution after 4 days, removing the non-adherent cells, and changing every 2 days; after the cells in the culture dish reach 80% fusion, removing the culture solution, rinsing with PBS for 1 time, adding trypsin-EDTA digestive liquid at 37 ℃ for digestion for 2min, slightly beating the bottle wall to promote the cells to be separated from the bottle bottom, then adding PBS containing 100U/ml penicillin and 100U/ml streptomycin, LG-DMEM culture medium containing 100U/ml penicillin and 100U/ml streptomycin to stop digestion, collecting the cells obtained after blowing, centrifuging, discarding the supernatant, mixing the mixture again with 0.5ml fetal calf serum, 12ml DMEM/F12 culture medium and 1mg NaCl, subculturing at the ratio of 1:2, placing the mixture at 37 ℃ and 5% of CO by volume fraction2And carrying out static culture in an environment with the humidity of 95% RH, replacing the culture solution for 1 time every 2 days, and digesting and passaging by the same method when the primary stem cells in the culture dish reach 80% fusion.
Specific parameters of examples 2 to 4 are shown in Table 1.
TABLE 1 raw material information for examples 2-4
Example 5: a method for separating and culturing human umbilical cord mesenchymal stem cells, which is different from the method in example 1 in that in the step IX and the step XI, the basal medium of LG-DMEM medium does not contain HEPES type.
Example 6: a method for isolating and culturing human umbilical cord mesenchymal stem cells, which is different from the method in example 1 in that the composition of the soaking solution in the step iii does not contain L-glutamic acid.
Example 7: a method for isolating and culturing human umbilical cord mesenchymal stem cells, which is different from example 1 in that the umbilical cord treated in step i is treated in step ii after being preserved for 100 hr.
Example 8: a method for isolating and culturing human umbilical cord mesenchymal stem cells, which is different from example 1 in that, in step iii, the umbilical cord is cut into a size of 3mm × 3mm × 3 mm.
Performance characterization
(1) Preparation of reference sample
Reference 1, the preparation method of which differs from example 1 in that in step iii the soaking solution consists of: 0.45g/L of glucose, 5g/L of ethanol and the balance of water; the dosage ratio of the umbilical cord to the soak solution is 1g to 5 ml.
Reference 2, the preparation method differs from example 1 in that in step iii, the soaking solution consists of: 2g/L of glucose, 40g/L of ethanol and the balance of water; the dosage ratio of the umbilical cord to the soak solution is 1g to 40 ml.
Reference 3, the preparation method thereof differs from example 1 in that, in step vii, the composition of the first mixed enzyme solution is: 0.5mg/g of trypsin-EDTA digestive solution, 1mg/g of collagenase II, 2.5mg/g of ethanol and the balance of water; and the dosage ratio of the umbilical cord to the first mixed enzyme solution is 1g:1 ml.
Reference 4, the preparation method thereof differs from example 1 in that, in step vii, the composition of the first mixed enzyme solution is: 4mg/g of trypsin-EDTA digestive juice, 6mg/g of collagenase II, 20mg/g of ethanol and the balance of water; and the dosage ratio of the umbilical cord to the first mixed enzyme solution is 1g to 10 ml.
Reference 5, the preparation method thereof differs from example 1 in that, in step viii, the composition of the second mixed enzyme solution is: 0.25mg/g of collagenase II, 0.25mg/g of hyaluronidase, 0.1mg/g of DNA hydrolase, 1mg/g of ethanol and the balance of water; and the dosage ratio of the umbilical cord and the second mixed enzyme solution is 0.5g to 1 ml.
Reference 6, the preparation method thereof differs from example 1 in that, in step viii, the composition of the second mixed enzyme solution is: 2mg/g of collagenase II, 2mg/g of hyaluronidase, 1mg/g of DNA hydrolase, 10mg/g of ethanol and the balance of water; and the dosage ratio of the umbilical cord to the second mixed enzyme solution is 0.5g to 10 ml.
Reference 7, the preparation method thereof differs from example 1 in that in step IX, umbilical cord, fetal bovine serum, PBS solution, LG-DMEM medium are used in a ratio of 1:0.1:0.25: 1.
Reference 8, the preparation method thereof differs from example 1 in that in step IX, umbilical cord, fetal bovine serum, PBS, LG-DMEM medium are used in a ratio of 1:1:4: 10.
Reference 9, the preparation method thereof differs from example 1 in that in step X, the ratio of the umbilical cord to the PBS liquid is 1: 1.
Reference 10, the preparation method differs from example 1 in that in step X, the ratio of the umbilical cord to the PBS liquid is 1: 10.
Reference 11, the preparation method thereof differs from example 1 in that in step XI, umbilical cord, fetal bovine serum, PBS solution, LG-DMEM medium are used in a ratio of 1:0.25:1: 1.
Reference 12, the preparation method thereof differs from example 1 in that in step XI, umbilical cord, fetal bovine serum, PBS solution, LG-DMEM medium are used in a ratio of 1:2:10: 10.
Reference 13, the method of preparation differs from example 1 in that steps v and vi are not employed.
Reference 14, the preparation method differs from example 1 in that step XIII is changed to: a100. mu.L aliquot of the homogenized sample (as primary stem cells) was taken and placed in sterile culture flasks, one for each1ml of fetal bovine serum (from Hyclone) and 10ml of DMEM/F12 medium (1:1, from Hyclone) were added to each flask and placed at 37 ℃ in a 5% volume CO2Standing and culturing in an environment with the humidity of 95% RH; changing the solution after 4 days, removing the non-adherent cells, and changing every 2 days; after the cells in the culture dish reach 80% fusion, removing the culture solution, rinsing with PBS for 1 time, adding trypsin-EDTA digestive liquid at 37 ℃ for digestion for 2min, slightly beating the bottle wall to promote the cells to be separated from the bottle bottom, then adding PBS containing 100U/ml penicillin and 100U/ml streptomycin and LG-DMEM culture medium containing 100U/ml penicillin and 100U/ml streptomycin to terminate digestion, collecting the cells obtained after blowing, centrifuging, discarding the supernatant, mixing with the culture solution again, carrying out subculture at the ratio of 1:2, placing at 37 ℃, and carrying out CO with the volume fraction of 5 percent2And carrying out static culture in an environment with the humidity of 95% RH, replacing the culture solution for 1 time every 2 days, and digesting and passaging by the same method when the primary stem cells in the culture dish reach 80% fusion.
Reference 15, prepared according to CN101643719A example 1.
(2) Phenotypic characterization of cells
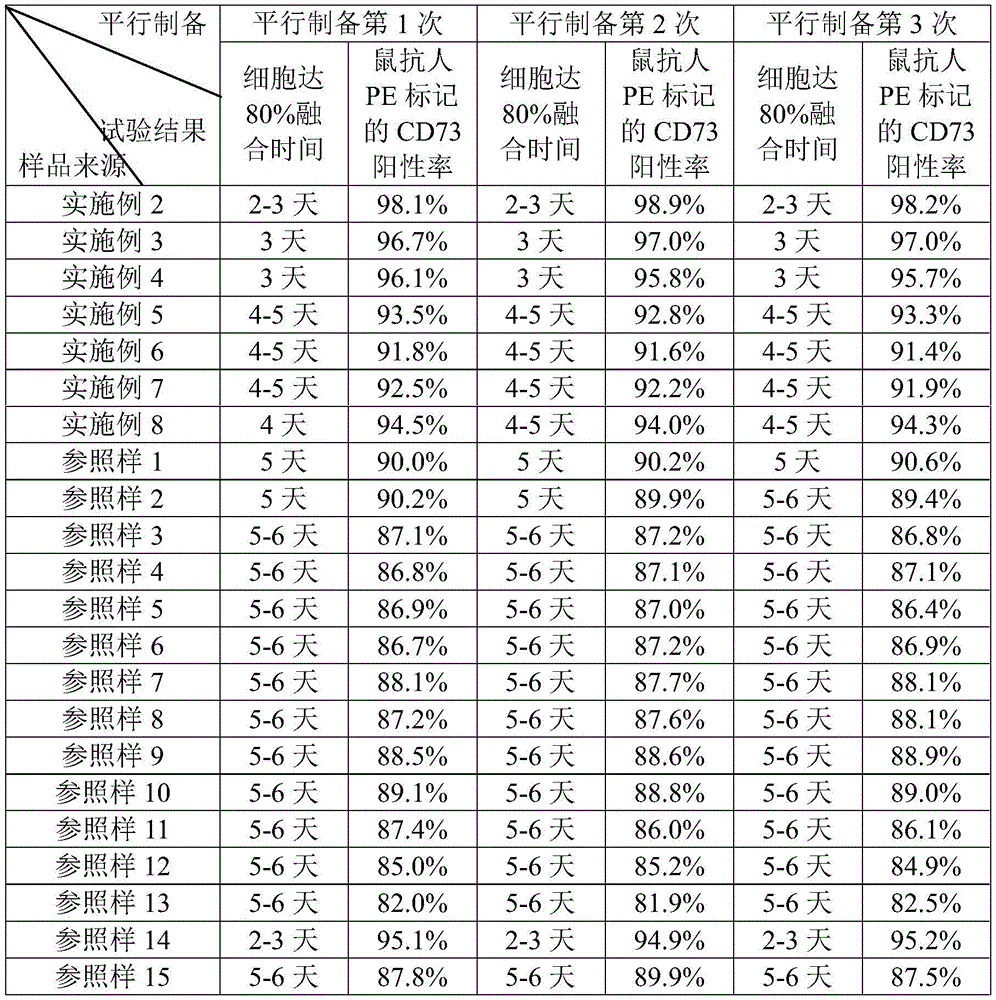
Test subjects: the phenotypic characterization of cells was performed using examples 2-8 as test samples and reference samples 1-12 as controls.
The test contents are as follows: after culturing for passage 3, the 4 th generation cell in logarithmic growth phase is taken to prepare 1X 109L-1Suspending liquid, incubating with mouse anti-human PE labeled CD73 and isotype control 10 μ L at 4 deg.C in dark for 30min, washing with PBS for 3 times, fixing with 10g/L paraformaldehyde, and analyzing by flow cytometry. Each sample was prepared 3 times in parallel, the phenotype of the prepared sample cells was identified 3 times each time and the positive rate of mouse anti-human PE-labeled CD73 obtained by flow cytometry analysis was averaged.
And (3) test results: as shown in table 2. Table 2 shows that the suspensions prepared in examples 2-8 have a short cell fusion time of 80% and high positive rate of mouse anti-human PE labeled CD73 (as a marker antigen of mesenchymal stem cells) and the results of parallel tests are close to each other when they are subjected to amplification culture, compared to the reference sample, which indicates that the suspensions prepared in examples 2-8 have a high amplification culture speed, high purity of the obtained mesenchymal stem cells, and higher product quality than the reference sample, and the suspension prepared in example 2 is the best sample.
TABLE 2 results of phenotypic identification of cells
Claims (3)
1. A method for separating and culturing human umbilical cord mesenchymal stem cells is characterized by comprising the following steps:
i. taking a fresh in-vitro umbilical cord of a newborn, placing the umbilical cord in PBS (phosphate buffer solution) which is controlled at the temperature of 2-5 ℃ and contains 100U/ml penicillin and 100U/ml streptomycin, and preserving for later use;
ii, taking out the umbilical cord treated in the step i, and washing the umbilical cord for multiple times by using PBS (phosphate buffer solution) with the temperature controlled at 2-5 ℃ and containing 100U/ml penicillin and 100U/ml streptomycin until no blood stain remains;
iii, shearing the washed umbilical cord in a soaking solution environment with the temperature controlled at 2-5 ℃, and removing arteriovenous in the shearing process; the dosage ratio of the umbilical cord to the soak solution is 1g to 13ml, and the soak solution comprises 1.0g/L glucose, 18g/L ethanol, 0.4 g/L-glutamic acid and the balance of water;
iv, taking the cut umbilical cord, washing the cut umbilical cord for 2-3 times by using PBS (phosphate buffer solution) containing 100U/ml penicillin and 100U/ml streptomycin at the temperature of 2-5 ℃, putting the umbilical cord into a culture bottle, placing the culture bottle in an environment with the temperature controlled at-80 to-70 ℃ and the vacuum degree controlled at 10-30 Pa for 5-20 min, and removing the solvent on the umbilical cord;
v, sealing the culture bottle, placing the culture bottle in an environment with the temperature controlled within-40 to-35 ℃, transferring the culture bottle to the environment with the temperature controlled within-5 to 0 ℃ when the temperature of the culture bottle is increased to-40 to-35 ℃, and transferring the culture bottle to the environment with the temperature controlled within 37 ℃ when the temperature of the culture bottle is increased to-5 to 0 ℃ until the umbilical cord in the culture bottle is completely thawed;
taking the culture bottle treated in the step v, and adding PBS (phosphate buffer solution) containing 100U/ml penicillin and 100U/ml streptomycin into the culture bottle; wherein the dosage ratio of the umbilical cord to the PBS is 1g to 1ml, and the umbilical cord and the PBS are fully contacted by oscillating for 1-5 min;
and vii, taking the culture bottle treated by the step vi, adding a first mixed enzyme solution into the culture bottle, and oscillating the culture bottle, wherein the temperature control curve of a water bath for oscillation is as follows: 0-5min, constant temperature of 15 ℃ → 5.01-20min, uniform temperature rise to 25 → 20.01-30min, constant temperature of 25 → 30.01-40min, constant temperature rise to 37 → 40.01-75min, constant temperature of 37 ℃ → 75.01-90min, natural cooling to 25 ℃; the dosage ratio of the umbilical cord to the first mixed enzyme solution is 1g:4ml, the first mixed enzyme solution comprises 2mg/g of trypsin-EDTA digestive juice, 3mg/g of collagenase II, 8mg/g of ethanol and the balance of water;
and viii, taking the culture bottle treated by the step vii, adding a second mixed enzyme solution into the culture bottle, and oscillating the culture bottle, wherein the temperature control curve of a water bath for oscillation is as follows: 0-2min, constant temperature of 25 → 2.01-10min, uniform temperature rise to 30 ℃ → 10.01-15min, constant temperature of 30 ℃ → 15.01-20min, constant temperature rise to 37 ℃ → 20.01-60min, constant temperature of 37 ℃ → 60.01-75min, natural cooling to 25 ℃; wherein the dosage ratio of the umbilical cord to the second mixed enzyme solution is 0.5g:3.5ml, the second mixed enzyme solution comprises 0.8mg/g collagenase II, 1mg/g hyaluronidase, 0.5mg/g DNA hydrolase, 5mg/g ethanol and the balance of water;
IX. taking the culture bottle treated by the step viii, adding fetal calf serum, PBS containing 100U/ml penicillin and 100U/ml streptomycin, LG-DMEM culture medium containing 100U/ml penicillin and 0.1g/L streptomycin, standing for 2-5 min at 20-25 ℃, and centrifuging for 5-8 min at the temperature; wherein the dosage ratio of the umbilical cord, the fetal calf serum, the PBS liquid and the LG-DMEM culture medium is 1g:0.4ml:1.5ml:4 ml;
x, adding the culture bottle treated in the step IX into PBS containing 100U/ml penicillin and 100U/ml streptomycin, standing for 2-5 min at 20-25 ℃, centrifuging for 5-8 min at the temperature, and removing the supernatant; wherein the dosage ratio of the umbilical cord to the PBS is 1g to 3 ml;
XI, adding fetal calf serum, PBS (phosphate buffer solution) containing 100U/ml penicillin and 100U/ml streptomycin and LG-DMEM (glycerol-free medium) containing 100U/ml penicillin and 100U/ml streptomycin into the culture bottle treated in the step X, standing for 2-5 min at 20-25 ℃, centrifuging for 5-8 min at the temperature, removing the supernatant, and repeating for 2-3 times; wherein the dosage ratio of the umbilical cord, the fetal calf serum, the PBS liquid and the LG-DMEM culture medium is 1g:0.5ml:2ml:5 ml;
XII, taking the culture bottle treated in the step XI, adding fetal calf serum, PBS (phosphate buffer solution) containing 100U/ml penicillin and 100U/ml streptomycin and LG-DMEM (glycerol modified eagle medium) containing 100U/ml penicillin and 100U/ml streptomycin, and uniformly mixing; wherein the dosage ratio of the umbilical cord, the fetal calf serum, the PBS liquid and the LG-DMEM culture medium is 1g:0.5ml:1ml:1 ml;
XIII, 100. mu.L of the sample treated in step XII was sampled, 0.5ml of fetal bovine serum, 12ml of DMEM/F12 medium and 1mg of NaCl were added thereto, and the mixture was placed at 37 ℃ in 5% by volume CO2Standing and culturing in an environment with the humidity of 95% RH; changing the solution after 4 days, removing the non-adherent cells, and changing every 2 days; after the cells in the culture dish reach 80% fusion, removing the culture solution, rinsing with PBS for 1 time, adding trypsin-EDTA digestive liquid at 37 ℃ for digestion for 2min, slightly beating the bottle wall to promote the cells to be separated from the bottle bottom, then adding PBS containing 100U/ml penicillin and 100U/ml streptomycin, LG-DMEM culture medium containing 100U/ml penicillin and 100U/ml streptomycin to stop digestion, collecting the cells obtained after blowing, centrifuging, discarding the supernatant, mixing the mixture again with 0.5ml fetal calf serum, 12ml DMEM/F12 culture medium and 1mg NaCl, subculturing at the ratio of 1:2, placing the mixture at 37 ℃ and 5% of CO by volume fraction2Performing static culture in an environment with the humidity of 95% RH, replacing the culture solution for 1 time every 2 days, and digesting and passaging by the same method when the primary stem cells in the culture dish reach 80% fusion;
in the step IX, the step XI, the step XII and the step XIII, the basic medium of LG-DMEM medium comprises 1000mg/L glucose, 4.0mM L-glutamic acid, 110mg/L sodium pyruvate and 25mM HEPES.
2. The method of claim 1, wherein the umbilical cord treated in step i is treated in step ii within 48 hr.
3. The method for isolating and culturing human umbilical cord mesenchymal stem cells according to claim 1, wherein in step iii, the umbilical cord is cut to a size of 1mm x 1 mm.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201610910131.2A CN106635974B (en) | 2016-10-19 | 2016-10-19 | Separation and culture method of human umbilical cord mesenchymal stem cells |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201610910131.2A CN106635974B (en) | 2016-10-19 | 2016-10-19 | Separation and culture method of human umbilical cord mesenchymal stem cells |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN106635974A CN106635974A (en) | 2017-05-10 |
| CN106635974B true CN106635974B (en) | 2020-10-27 |
Family
ID=58856599
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201610910131.2A Active CN106635974B (en) | 2016-10-19 | 2016-10-19 | Separation and culture method of human umbilical cord mesenchymal stem cells |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN106635974B (en) |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN107513518A (en) * | 2017-10-10 | 2017-12-26 | 重庆金时代生物技术有限公司 | A kind of umbilical cord mesenchymal stem cells extracorporeal culturing method |
| CN108251358B (en) * | 2017-12-15 | 2021-10-26 | 广东药科大学 | Multi-batch primary separation method of human mesenchymal stem cells from same donor source |
| CN110684726A (en) * | 2018-07-06 | 2020-01-14 | 上海中溢精准医疗科技有限公司 | Method for separating and culturing umbilical cord mesenchymal stem cells |
| CN112011505B (en) * | 2020-09-09 | 2021-07-30 | 陕西中港万海生命科学研究院有限公司 | Umbilical cord mesenchymal stem cell separation method |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| TWI548747B (en) * | 2006-09-13 | 2016-09-11 | 艾伯維有限公司 | Cell culture improvement |
| WO2011101834A1 (en) * | 2010-02-22 | 2011-08-25 | Advanced Neuro-Science Allies Private Limited | A method for obtaining mesenchymal stem cells, media, methods and composition thereof |
| CN101974484B (en) * | 2010-11-03 | 2013-01-30 | 江苏省北科生物科技有限公司 | Method for preparing human umbilical cord mesenchymal stem cells |
| CN102127522B (en) * | 2010-12-27 | 2012-11-21 | 协和干细胞基因工程有限公司 | Human umbilical mesenchymal stem cell and preparation method thereof |
| CN102660497B (en) * | 2012-05-21 | 2013-06-12 | 博雅干细胞科技有限公司 | Method for freezing and reviving umbilical cord tissues and for separating and increasing stem cells |
| JP2014172899A (en) * | 2013-03-13 | 2014-09-22 | Nobuhiko Miwa | Bio activation agent and composition thereof, and method of manufacturing it |
| CN104988117B (en) * | 2015-07-03 | 2016-06-15 | 深圳中基恒润投资有限公司 | Separate and cultivate mescenchymal stem cell from umbilical cord and to the method for chondrogenic differentiation |
-
2016
- 2016-10-19 CN CN201610910131.2A patent/CN106635974B/en active Active
Also Published As
| Publication number | Publication date |
|---|---|
| CN106635974A (en) | 2017-05-10 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR100908481B1 (en) | Mesenchymal stem cell culture medium and culture method of mesenchymal stem cells using the same | |
| EP4663241A2 (en) | Enhanced msc preparations | |
| KR20120008223A (en) | A medium composition for culturing amnion derived mesenchymal stem cells and a method of culturing amnion derived mesenchymal stem cells using the same | |
| CN106635974B (en) | Separation and culture method of human umbilical cord mesenchymal stem cells | |
| CN105713871A (en) | Human chorion mesenchymal stem cell isolated culture method | |
| CN108184818B (en) | A kind of human placental mesenchymal stem cell suspension protective agent | |
| CN105238751A (en) | Umbilical cord tissue mesenchymal stem cell isolated culture method | |
| CN105670986A (en) | Culture medium for inducing human umbilical cord mesenchymal stem cells to differentiate into islet-like cells and induction method therefor | |
| CN105420179A (en) | Method for simultaneously extracting epithelial cells and mesenchymal stem cells from umbilical cord and placenta amnion tissues | |
| WO2017094879A1 (en) | Method for producing mesenchymal stem cells | |
| CN113287603A (en) | Biological sample preservation solution and preparation method and application thereof | |
| CN115851587A (en) | Optimized culture medium, kit and culture method of human placenta-derived mesenchymal stem cells | |
| CN108504628A (en) | The cultural method of human umbilical cord mesenchymal stem cells | |
| CN106399235A (en) | Method for isolating human umbilical cord mesenchymal stem cells | |
| CN118064359A (en) | Preparation method of human umbilical cord mesenchymal stem cell bank with high immunoregulation capacity | |
| CN110747162B (en) | Application of small molecular compound 4-aminobiphenyl in promoting stem cell proliferation and chondrogenic differentiation | |
| WO2012070001A1 (en) | An explant culture technique for isolation of mesenchymal stem cells from adipose tissue | |
| CN108496957A (en) | The deepfreeze store method of umbilical cord mesenchymal stem cells culture supernatant | |
| CN106434546B (en) | Method for preparing mesenchymal stem cells by fully utilizing umbilical cord resources | |
| CN105624115B (en) | A culture medium for inducing human umbilical cord mesenchymal stem cells to differentiate into neural-like cells and its induction method | |
| CN116240167A (en) | A preparation method for extracting dental pulp stem cells | |
| CN110484491B (en) | Method for obtaining amniotic membrane and amniotic fluid derived endothelial progenitor cells and purification culture method thereof | |
| CN109666627B (en) | Method for inducing umbilical cord mesenchymal stem cells to differentiate into Leydig cells with small molecules | |
| CN114276980A (en) | A method for culturing pancreatic islet cells suitable for clinical application | |
| CN118546875A (en) | A method for extracting and characterizing mesenchymal stem cells derived from human fallopian tube |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |