CN105315319B - Hepatitis C virus inhibitor and its application - Google Patents
Hepatitis C virus inhibitor and its application Download PDFInfo
- Publication number
- CN105315319B CN105315319B CN201510453910.XA CN201510453910A CN105315319B CN 105315319 B CN105315319 B CN 105315319B CN 201510453910 A CN201510453910 A CN 201510453910A CN 105315319 B CN105315319 B CN 105315319B
- Authority
- CN
- China
- Prior art keywords
- ring
- fluoro
- alkyl
- amino
- methyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 241000711549 Hepacivirus C Species 0.000 title abstract description 43
- 239000003112 inhibitor Substances 0.000 title description 6
- 150000001875 compounds Chemical class 0.000 claims abstract description 241
- 239000003814 drug Substances 0.000 claims abstract description 12
- 125000000217 alkyl group Chemical group 0.000 claims description 143
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 72
- 229910052736 halogen Inorganic materials 0.000 claims description 70
- 150000002367 halogens Chemical class 0.000 claims description 70
- 150000003839 salts Chemical class 0.000 claims description 46
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 30
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 25
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical group C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 claims description 18
- 229910052799 carbon Inorganic materials 0.000 claims description 16
- 238000011282 treatment Methods 0.000 claims description 15
- 239000008194 pharmaceutical composition Substances 0.000 claims description 14
- YTPLMLYBLZKORZ-UHFFFAOYSA-N Thiophene Chemical group C=1C=CSC=1 YTPLMLYBLZKORZ-UHFFFAOYSA-N 0.000 claims description 13
- 125000002883 imidazolyl group Chemical group 0.000 claims description 13
- 125000000842 isoxazolyl group Chemical group 0.000 claims description 13
- 125000002971 oxazolyl group Chemical group 0.000 claims description 13
- 125000003226 pyrazolyl group Chemical group 0.000 claims description 13
- FZWLAAWBMGSTSO-UHFFFAOYSA-N Thiazole Chemical group C1=CSC=N1 FZWLAAWBMGSTSO-UHFFFAOYSA-N 0.000 claims description 12
- 125000000168 pyrrolyl group Chemical group 0.000 claims description 12
- 230000009385 viral infection Effects 0.000 claims description 11
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 claims description 8
- 239000011737 fluorine Substances 0.000 claims description 8
- 229910052731 fluorine Inorganic materials 0.000 claims description 8
- SIKJAQJRHWYJAI-UHFFFAOYSA-N Indole Chemical compound C1=CC=C2NC=CC2=C1 SIKJAQJRHWYJAI-UHFFFAOYSA-N 0.000 claims description 6
- KTZQTRPPVKQPFO-UHFFFAOYSA-N 1,2-benzoxazole Chemical group C1=CC=C2C=NOC2=C1 KTZQTRPPVKQPFO-UHFFFAOYSA-N 0.000 claims description 5
- 150000001721 carbon Chemical group 0.000 claims description 5
- 125000003453 indazolyl group Chemical group N1N=C(C2=C1C=CC=C2)* 0.000 claims description 5
- 239000003937 drug carrier Substances 0.000 claims description 3
- 238000004519 manufacturing process Methods 0.000 claims description 2
- 238000011321 prophylaxis Methods 0.000 claims description 2
- 125000001153 fluoro group Chemical group F* 0.000 claims 2
- 125000001164 benzothiazolyl group Chemical group S1C(=NC2=C1C=CC=C2)* 0.000 claims 1
- 208000006454 hepatitis Diseases 0.000 claims 1
- 231100000283 hepatitis Toxicity 0.000 claims 1
- 238000002360 preparation method Methods 0.000 abstract description 89
- 239000000203 mixture Substances 0.000 abstract description 14
- 208000015181 infectious disease Diseases 0.000 abstract description 13
- 230000000840 anti-viral effect Effects 0.000 abstract description 6
- 231100000135 cytotoxicity Toxicity 0.000 abstract description 5
- 230000003013 cytotoxicity Effects 0.000 abstract description 5
- 208000035473 Communicable disease Diseases 0.000 abstract description 4
- 230000002401 inhibitory effect Effects 0.000 abstract description 4
- 108010017384 Blood Proteins Proteins 0.000 abstract description 2
- 102000004506 Blood Proteins Human genes 0.000 abstract description 2
- 229940126585 therapeutic drug Drugs 0.000 abstract description 2
- -1 nucleoside phosphate compounds Chemical class 0.000 description 180
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 166
- XHXFXVLFKHQFAL-UHFFFAOYSA-N phosphoryl trichloride Chemical compound ClP(Cl)(Cl)=O XHXFXVLFKHQFAL-UHFFFAOYSA-N 0.000 description 118
- 125000003545 alkoxy group Chemical group 0.000 description 111
- 238000004895 liquid chromatography mass spectrometry Methods 0.000 description 89
- 125000001072 heteroaryl group Chemical group 0.000 description 84
- 238000000034 method Methods 0.000 description 80
- 125000000592 heterocycloalkyl group Chemical group 0.000 description 70
- 125000000266 alpha-aminoacyl group Chemical group 0.000 description 66
- 125000005257 alkyl acyl group Chemical group 0.000 description 65
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 description 65
- 125000004366 heterocycloalkenyl group Chemical group 0.000 description 61
- 125000004890 (C1-C6) alkylamino group Chemical group 0.000 description 60
- 239000002994 raw material Substances 0.000 description 59
- XBNGYFFABRKICK-UHFFFAOYSA-N 2,3,4,5,6-pentafluorophenol Chemical compound OC1=C(F)C(F)=C(F)C(F)=C1F XBNGYFFABRKICK-UHFFFAOYSA-N 0.000 description 58
- IZUPBVBPLAPZRR-UHFFFAOYSA-N pentachloro-phenol Natural products OC1=C(Cl)C(Cl)=C(Cl)C(Cl)=C1Cl IZUPBVBPLAPZRR-UHFFFAOYSA-N 0.000 description 58
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 55
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 51
- 238000006243 chemical reaction Methods 0.000 description 50
- 238000005481 NMR spectroscopy Methods 0.000 description 48
- XBDQKXXYIPTUBI-UHFFFAOYSA-M Propionate Chemical compound CCC([O-])=O XBDQKXXYIPTUBI-UHFFFAOYSA-M 0.000 description 47
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 42
- 239000012453 solvate Substances 0.000 description 42
- 125000005913 (C3-C6) cycloalkyl group Chemical group 0.000 description 41
- YAQKNCSWDMGPOY-JEDNCBNOSA-N propan-2-yl (2s)-2-aminopropanoate;hydrochloride Chemical compound Cl.CC(C)OC(=O)[C@H](C)N YAQKNCSWDMGPOY-JEDNCBNOSA-N 0.000 description 39
- 229910052739 hydrogen Inorganic materials 0.000 description 38
- IJMWOMHMDSDKGK-UHFFFAOYSA-N Isopropyl propionate Chemical compound CCC(=O)OC(C)C IJMWOMHMDSDKGK-UHFFFAOYSA-N 0.000 description 36
- 239000013078 crystal Substances 0.000 description 36
- 125000000475 sulfinyl group Chemical group [*:2]S([*:1])=O 0.000 description 36
- 125000000472 sulfonyl group Chemical group *S(*)(=O)=O 0.000 description 35
- PXQLVRUNWNTZOS-UHFFFAOYSA-N sulfanyl Chemical group [SH] PXQLVRUNWNTZOS-UHFFFAOYSA-N 0.000 description 34
- 238000004440 column chromatography Methods 0.000 description 32
- 125000003118 aryl group Chemical group 0.000 description 31
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 31
- ARKKGZQTGXJVKW-VPCXQMTMSA-N 1-[(2r,3r,4r,5r)-3-fluoro-4-hydroxy-5-(hydroxymethyl)-3-methyloxolan-2-yl]pyrimidine-2,4-dione Chemical compound C[C@@]1(F)[C@H](O)[C@@H](CO)O[C@H]1N1C(=O)NC(=O)C=C1 ARKKGZQTGXJVKW-VPCXQMTMSA-N 0.000 description 30
- LFGREXWGYUGZLY-UHFFFAOYSA-N phosphoryl Chemical group [P]=O LFGREXWGYUGZLY-UHFFFAOYSA-N 0.000 description 30
- 239000007858 starting material Substances 0.000 description 30
- 239000001257 hydrogen Substances 0.000 description 28
- 125000003342 alkenyl group Chemical group 0.000 description 27
- IAZDPXIOMUYVGZ-WFGJKAKNSA-N Dimethyl sulfoxide Chemical compound [2H]C([2H])([2H])S(=O)C([2H])([2H])[2H] IAZDPXIOMUYVGZ-WFGJKAKNSA-N 0.000 description 26
- 125000005256 alkoxyacyl group Chemical group 0.000 description 26
- 125000000304 alkynyl group Chemical group 0.000 description 26
- 125000004433 nitrogen atom Chemical group N* 0.000 description 26
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 24
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 24
- COIQUVGFTILYGA-UHFFFAOYSA-N (4-hydroxyphenyl)boronic acid Chemical compound OB(O)C1=CC=C(O)C=C1 COIQUVGFTILYGA-UHFFFAOYSA-N 0.000 description 23
- 210000004027 cell Anatomy 0.000 description 23
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 23
- 239000000243 solution Substances 0.000 description 22
- 150000002431 hydrogen Chemical class 0.000 description 21
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 20
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 20
- YLQBMQCUIZJEEH-UHFFFAOYSA-N Furan Chemical group C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 19
- 239000002253 acid Substances 0.000 description 17
- 229940049706 benzodiazepine Drugs 0.000 description 16
- ATUOYWHBWRKTHZ-UHFFFAOYSA-N dimethylmethane Natural products CCC ATUOYWHBWRKTHZ-UHFFFAOYSA-N 0.000 description 15
- FHGWEHGZBUBQKL-UHFFFAOYSA-N 1,2-benzothiazepine Chemical group S1N=CC=CC2=CC=CC=C12 FHGWEHGZBUBQKL-UHFFFAOYSA-N 0.000 description 14
- SVUOLADPCWQTTE-UHFFFAOYSA-N 1h-1,2-benzodiazepine Chemical compound N1N=CC=CC2=CC=CC=C12 SVUOLADPCWQTTE-UHFFFAOYSA-N 0.000 description 14
- 241000700605 Viruses Species 0.000 description 14
- 125000004432 carbon atom Chemical group C* 0.000 description 14
- ZCXLTWVZYXBHJS-UHFFFAOYSA-N 1,2-benzoxazepine Chemical group O1N=CC=CC2=CC=CC=C12 ZCXLTWVZYXBHJS-UHFFFAOYSA-N 0.000 description 13
- 125000000753 cycloalkyl group Chemical group 0.000 description 13
- 230000000694 effects Effects 0.000 description 13
- 125000004434 sulfur atom Chemical group 0.000 description 13
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 12
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 12
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 12
- 125000004169 (C1-C6) alkyl group Chemical group 0.000 description 11
- 125000003282 alkyl amino group Chemical group 0.000 description 11
- 150000004677 hydrates Chemical class 0.000 description 11
- 239000012074 organic phase Substances 0.000 description 11
- 239000001294 propane Substances 0.000 description 11
- 125000003373 pyrazinyl group Chemical group 0.000 description 11
- 125000000714 pyrimidinyl group Chemical group 0.000 description 11
- 229920006395 saturated elastomer Polymers 0.000 description 11
- 125000006552 (C3-C8) cycloalkyl group Chemical group 0.000 description 10
- UGUHFDPGDQDVGX-UHFFFAOYSA-N 1,2,3-thiadiazole Chemical group C1=CSN=N1 UGUHFDPGDQDVGX-UHFFFAOYSA-N 0.000 description 10
- PCLIMKBDDGJMGD-UHFFFAOYSA-N N-bromosuccinimide Chemical compound BrN1C(=O)CCC1=O PCLIMKBDDGJMGD-UHFFFAOYSA-N 0.000 description 10
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 10
- 239000012141 concentrate Substances 0.000 description 10
- 235000008504 concentrate Nutrition 0.000 description 10
- ZLTPDFXIESTBQG-UHFFFAOYSA-N isothiazole Chemical group C=1C=NSC=1 ZLTPDFXIESTBQG-UHFFFAOYSA-N 0.000 description 10
- WCPAKWJPBJAGKN-UHFFFAOYSA-N oxadiazole Chemical group C1=CON=N1 WCPAKWJPBJAGKN-UHFFFAOYSA-N 0.000 description 10
- PBMFSQRYOILNGV-UHFFFAOYSA-N pyridazine Chemical group C1=CC=NN=C1 PBMFSQRYOILNGV-UHFFFAOYSA-N 0.000 description 10
- 239000011780 sodium chloride Substances 0.000 description 10
- 150000003852 triazoles Chemical group 0.000 description 10
- 241000710781 Flaviviridae Species 0.000 description 9
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 9
- 239000003153 chemical reaction reagent Substances 0.000 description 9
- 238000000605 extraction Methods 0.000 description 9
- 108010050904 Interferons Proteins 0.000 description 8
- 102000014150 Interferons Human genes 0.000 description 8
- 125000001183 hydrocarbyl group Chemical group 0.000 description 8
- 229940079322 interferon Drugs 0.000 description 8
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 8
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 7
- 208000036142 Viral infection Diseases 0.000 description 7
- 238000001514 detection method Methods 0.000 description 7
- 229940079593 drug Drugs 0.000 description 7
- 125000001188 haloalkyl group Chemical group 0.000 description 7
- 239000002609 medium Substances 0.000 description 7
- IYUKFAFDFHZKPI-DFWYDOINSA-N methyl (2s)-2-aminopropanoate;hydrochloride Chemical compound Cl.COC(=O)[C@H](C)N IYUKFAFDFHZKPI-DFWYDOINSA-N 0.000 description 7
- 125000001624 naphthyl group Chemical group 0.000 description 7
- 229910052757 nitrogen Inorganic materials 0.000 description 7
- 125000006559 (C1-C3) alkylamino group Chemical group 0.000 description 6
- 125000006656 (C2-C4) alkenyl group Chemical group 0.000 description 6
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 6
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 6
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 6
- ZHNUHDYFZUAESO-UHFFFAOYSA-N Formamide Chemical compound NC=O ZHNUHDYFZUAESO-UHFFFAOYSA-N 0.000 description 6
- 108060001084 Luciferase Proteins 0.000 description 6
- 239000005089 Luciferase Substances 0.000 description 6
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 6
- 238000004364 calculation method Methods 0.000 description 6
- 239000000460 chlorine Substances 0.000 description 6
- 125000005842 heteroatom Chemical group 0.000 description 6
- 230000005764 inhibitory process Effects 0.000 description 6
- 239000002777 nucleoside Substances 0.000 description 6
- 229930195734 saturated hydrocarbon Natural products 0.000 description 6
- 229910052717 sulfur Inorganic materials 0.000 description 6
- OYIYNIONWDBJIF-UHFFFAOYSA-N (4-hydroxy-2-methylphenyl)boronic acid Chemical compound CC1=CC(O)=CC=C1B(O)O OYIYNIONWDBJIF-UHFFFAOYSA-N 0.000 description 5
- 125000006650 (C2-C4) alkynyl group Chemical group 0.000 description 5
- DQFQCHIDRBIESA-UHFFFAOYSA-N 1-benzazepine Chemical group N1C=CC=CC2=CC=CC=C12 DQFQCHIDRBIESA-UHFFFAOYSA-N 0.000 description 5
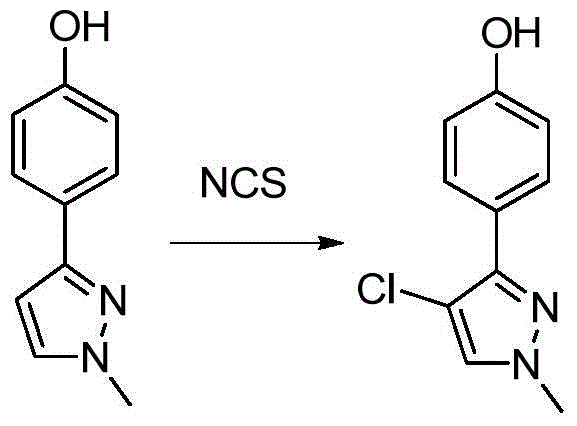
- WINBZWXSAVBKHU-UHFFFAOYSA-N 4-(1-methylpyrazol-3-yl)phenol Chemical compound CN1N=C(C=C1)C1=CC=C(C=C1)O WINBZWXSAVBKHU-UHFFFAOYSA-N 0.000 description 5
- RUZWJMSQCFYNAM-UHFFFAOYSA-N 4-thiophen-2-ylphenol Chemical compound C1=CC(O)=CC=C1C1=CC=CS1 RUZWJMSQCFYNAM-UHFFFAOYSA-N 0.000 description 5
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 5
- 239000006144 Dulbecco’s modified Eagle's medium Substances 0.000 description 5
- 125000003310 benzodiazepinyl group Chemical group N1N=C(C=CC2=C1C=CC=C2)* 0.000 description 5
- 229910052801 chlorine Inorganic materials 0.000 description 5
- VZWXIQHBIQLMPN-UHFFFAOYSA-N chromane Chemical group C1=CC=C2CCCOC2=C1 VZWXIQHBIQLMPN-UHFFFAOYSA-N 0.000 description 5
- 125000004438 haloalkoxy group Chemical group 0.000 description 5
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 5
- 125000001972 isopentyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 5
- 125000000740 n-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 5
- 125000004430 oxygen atom Chemical group O* 0.000 description 5
- 230000002265 prevention Effects 0.000 description 5
- 238000010992 reflux Methods 0.000 description 5
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 5
- VTSRCHXIIMBAKT-WNQIDUERSA-N (2s)-2-aminopropanoic acid;2-chloropropane Chemical compound CC(C)Cl.C[C@H](N)C(O)=O VTSRCHXIIMBAKT-WNQIDUERSA-N 0.000 description 4
- SLLFVLKNXABYGI-UHFFFAOYSA-N 1,2,3-benzoxadiazole Chemical group C1=CC=C2ON=NC2=C1 SLLFVLKNXABYGI-UHFFFAOYSA-N 0.000 description 4
- CSNIZNHTOVFARY-UHFFFAOYSA-N 1,2-benzothiazole Chemical group C1=CC=C2C=NSC2=C1 CSNIZNHTOVFARY-UHFFFAOYSA-N 0.000 description 4
- 125000000355 1,3-benzoxazolyl group Chemical group O1C(=NC2=C1C=CC=C2)* 0.000 description 4
- IANQTJSKSUMEQM-UHFFFAOYSA-N 1-benzofuran Chemical group C1=CC=C2OC=CC2=C1 IANQTJSKSUMEQM-UHFFFAOYSA-N 0.000 description 4
- HZNVUJQVZSTENZ-UHFFFAOYSA-N 2,3-dichloro-5,6-dicyano-1,4-benzoquinone Chemical compound ClC1=C(Cl)C(=O)C(C#N)=C(C#N)C1=O HZNVUJQVZSTENZ-UHFFFAOYSA-N 0.000 description 4
- XQJAHBHCLXUGEP-UHFFFAOYSA-N 2-bromo-1-(4-methoxyphenyl)ethanone Chemical compound COC1=CC=C(C(=O)CBr)C=C1 XQJAHBHCLXUGEP-UHFFFAOYSA-N 0.000 description 4
- ZDXNEAWEPCZBQO-UHFFFAOYSA-N 4-(1,3-thiazol-4-yl)phenol Chemical compound C1=CC(O)=CC=C1C1=CSC=N1 ZDXNEAWEPCZBQO-UHFFFAOYSA-N 0.000 description 4
- VWIPREOWTHPLGZ-UHFFFAOYSA-N 4-(3-fluoropyridin-2-yl)phenol Chemical compound C1=CC(=C(N=C1)C2=CC=C(C=C2)O)F VWIPREOWTHPLGZ-UHFFFAOYSA-N 0.000 description 4
- DQICBPNUMVEVIA-UHFFFAOYSA-N 4-(3-methylthiophen-2-yl)phenol Chemical compound C1=CSC(C=2C=CC(O)=CC=2)=C1C DQICBPNUMVEVIA-UHFFFAOYSA-N 0.000 description 4
- RDTKGSYKMUXXSY-UHFFFAOYSA-N 4-(4-methoxyphenyl)-1,3-oxazole Chemical compound C1=CC(OC)=CC=C1C1=COC=N1 RDTKGSYKMUXXSY-UHFFFAOYSA-N 0.000 description 4
- OQLREJFMSQEHQR-UHFFFAOYSA-N 4-(5-chloro-1,3-thiazol-4-yl)phenol Chemical compound ClC1=C(N=CS1)C1=CC=C(C=C1)O OQLREJFMSQEHQR-UHFFFAOYSA-N 0.000 description 4
- LCBPXERZXJUXQS-UHFFFAOYSA-N 4-(5-methylthiophen-2-yl)phenol Chemical compound S1C(C)=CC=C1C1=CC=C(O)C=C1 LCBPXERZXJUXQS-UHFFFAOYSA-N 0.000 description 4
- MGYPKUHNAMYABF-UHFFFAOYSA-N 4-bromo-5-methyl-1,3-thiazole Chemical compound CC=1SC=NC=1Br MGYPKUHNAMYABF-UHFFFAOYSA-N 0.000 description 4
- NTPLXRHDUXRPNE-UHFFFAOYSA-N 4-methoxyacetophenone Chemical compound COC1=CC=C(C(C)=O)C=C1 NTPLXRHDUXRPNE-UHFFFAOYSA-N 0.000 description 4
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 description 4
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 4
- WTDHULULXKLSOZ-UHFFFAOYSA-N Hydroxylamine hydrochloride Chemical compound Cl.ON WTDHULULXKLSOZ-UHFFFAOYSA-N 0.000 description 4
- 108010047761 Interferon-alpha Proteins 0.000 description 4
- 102000006992 Interferon-alpha Human genes 0.000 description 4
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 4
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Phenol Chemical compound OC1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 4
- SMWDFEZZVXVKRB-UHFFFAOYSA-N Quinoline Chemical group N1=CC=CC2=CC=CC=C21 SMWDFEZZVXVKRB-UHFFFAOYSA-N 0.000 description 4
- IWUCXVSUMQZMFG-AFCXAGJDSA-N Ribavirin Chemical compound N1=C(C(=O)N)N=CN1[C@H]1[C@H](O)[C@H](O)[C@@H](CO)O1 IWUCXVSUMQZMFG-AFCXAGJDSA-N 0.000 description 4
- DTQVDTLACAAQTR-UHFFFAOYSA-N Trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 4
- 125000002252 acyl group Chemical group 0.000 description 4
- 125000003785 benzimidazolyl group Chemical group N1=C(NC2=C1C=CC=C2)* 0.000 description 4
- 125000005605 benzo group Chemical group 0.000 description 4
- BNBQRQQYDMDJAH-UHFFFAOYSA-N benzodioxan Chemical group C1=CC=C2OCCOC2=C1 BNBQRQQYDMDJAH-UHFFFAOYSA-N 0.000 description 4
- IOJUPLGTWVMSFF-UHFFFAOYSA-N benzothiazole Chemical group C1=CC=C2SC=NC2=C1 IOJUPLGTWVMSFF-UHFFFAOYSA-N 0.000 description 4
- 125000003354 benzotriazolyl group Chemical group N1N=NC2=C1C=CC=C2* 0.000 description 4
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 4
- ILAHWRKJUDSMFH-UHFFFAOYSA-N boron tribromide Chemical compound BrB(Br)Br ILAHWRKJUDSMFH-UHFFFAOYSA-N 0.000 description 4
- 230000003833 cell viability Effects 0.000 description 4
- WCZVZNOTHYJIEI-UHFFFAOYSA-N cinnoline Chemical group N1=NC=CC2=CC=CC=C21 WCZVZNOTHYJIEI-UHFFFAOYSA-N 0.000 description 4
- 125000004122 cyclic group Chemical group 0.000 description 4
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 4
- JCXLZWMDXJFOOI-WCCKRBBISA-N ethyl (2s)-2-aminopropanoate;hydrochloride Chemical compound Cl.CCOC(=O)[C@H](C)N JCXLZWMDXJFOOI-WCCKRBBISA-N 0.000 description 4
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 4
- KJIFKLIQANRMOU-UHFFFAOYSA-N oxidanium;4-methylbenzenesulfonate Chemical compound O.CC1=CC=C(S(O)(=O)=O)C=C1 KJIFKLIQANRMOU-UHFFFAOYSA-N 0.000 description 4
- 229910052760 oxygen Inorganic materials 0.000 description 4
- 229910000027 potassium carbonate Inorganic materials 0.000 description 4
- 238000012545 processing Methods 0.000 description 4
- 238000010791 quenching Methods 0.000 description 4
- JWVCLYRUEFBMGU-UHFFFAOYSA-N quinazoline Chemical group N1=CN=CC2=CC=CC=C21 JWVCLYRUEFBMGU-UHFFFAOYSA-N 0.000 description 4
- 125000001567 quinoxalinyl group Chemical group N1=C(C=NC2=CC=CC=C12)* 0.000 description 4
- HZCAHMRRMINHDJ-DBRKOABJSA-N ribavirin Natural products O[C@@H]1[C@H](O)[C@@H](CO)O[C@H]1N1N=CN=C1 HZCAHMRRMINHDJ-DBRKOABJSA-N 0.000 description 4
- 229960000329 ribavirin Drugs 0.000 description 4
- 125000004191 (C1-C6) alkoxy group Chemical group 0.000 description 3
- FNQJDLTXOVEEFB-UHFFFAOYSA-N 1,2,3-benzothiadiazole Chemical group C1=CC=C2SN=NC2=C1 FNQJDLTXOVEEFB-UHFFFAOYSA-N 0.000 description 3
- FCEHBMOGCRZNNI-UHFFFAOYSA-N 1-benzothiophene Chemical group C1=CC=C2SC=CC2=C1 FCEHBMOGCRZNNI-UHFFFAOYSA-N 0.000 description 3
- PRIGFEJKMMRJSF-UHFFFAOYSA-M 1-fluoro-2,4,6-trimethylpyridin-1-ium;trifluoromethanesulfonate Chemical compound [O-]S(=O)(=O)C(F)(F)F.CC1=CC(C)=[N+](F)C(C)=C1 PRIGFEJKMMRJSF-UHFFFAOYSA-M 0.000 description 3
- FSUYMKXZLQOFQY-UHFFFAOYSA-N 3,4-dihydro-1,2-benzodithiine Chemical group C1=CC=C2SSCCC2=C1 FSUYMKXZLQOFQY-UHFFFAOYSA-N 0.000 description 3
- WPWNEKFMGCWNPR-UHFFFAOYSA-N 3,4-dihydro-2h-thiochromene Chemical group C1=CC=C2CCCSC2=C1 WPWNEKFMGCWNPR-UHFFFAOYSA-N 0.000 description 3
- NBUTXWIFTKWSMK-UHFFFAOYSA-N 4-(1-methylimidazol-4-yl)phenol Chemical compound CN1C=NC(C=2C=CC(O)=CC=2)=C1 NBUTXWIFTKWSMK-UHFFFAOYSA-N 0.000 description 3
- HOAYTTLLVORNLA-UHFFFAOYSA-N 4-(2-methyl-1,3-thiazol-4-yl)phenol Chemical compound S1C(C)=NC(C=2C=CC(O)=CC=2)=C1 HOAYTTLLVORNLA-UHFFFAOYSA-N 0.000 description 3
- DTQHSTVFRWIPNR-UHFFFAOYSA-N 4-(5-chloro-2-methyl-1,3-thiazol-4-yl)phenol Chemical compound ClC1=C(N=C(S1)C)C1=CC=C(C=C1)O DTQHSTVFRWIPNR-UHFFFAOYSA-N 0.000 description 3
- VDTIGYKLTROQAH-UHFFFAOYSA-N 4-bromo-1,3-thiazole Chemical compound BrC1=CSC=N1 VDTIGYKLTROQAH-UHFFFAOYSA-N 0.000 description 3
- IOTSLMMLLXTNNH-UHFFFAOYSA-N 4-bromo-1-methylimidazole Chemical compound CN1C=NC(Br)=C1 IOTSLMMLLXTNNH-UHFFFAOYSA-N 0.000 description 3
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 3
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 3
- NLXLAEXVIDQMFP-UHFFFAOYSA-N Ammonium chloride Substances [NH4+].[Cl-] NLXLAEXVIDQMFP-UHFFFAOYSA-N 0.000 description 3
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 3
- 208000005176 Hepatitis C Diseases 0.000 description 3
- OFOBLEOULBTSOW-UHFFFAOYSA-N Propanedioic acid Natural products OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 3
- QOSMNYMQXIVWKY-UHFFFAOYSA-N Propyl levulinate Chemical compound CCCOC(=O)CCC(C)=O QOSMNYMQXIVWKY-UHFFFAOYSA-N 0.000 description 3
- 235000002597 Solanum melongena Nutrition 0.000 description 3
- 150000001335 aliphatic alkanes Chemical class 0.000 description 3
- 125000004429 atom Chemical group 0.000 description 3
- OKTCJUNXOQMLRW-UHFFFAOYSA-N benzene 1H-1,2-benzodiazepine Chemical compound C1=CC=CC=C1.N1N=CC=CC2=C1C=CC=C2 OKTCJUNXOQMLRW-UHFFFAOYSA-N 0.000 description 3
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 3
- 229910052794 bromium Inorganic materials 0.000 description 3
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 3
- 201000010099 disease Diseases 0.000 description 3
- 108010048367 enhanced green fluorescent protein Proteins 0.000 description 3
- 150000002148 esters Chemical class 0.000 description 3
- 238000009472 formulation Methods 0.000 description 3
- 239000001963 growth medium Substances 0.000 description 3
- 125000005843 halogen group Chemical group 0.000 description 3
- 208000010710 hepatitis C virus infection Diseases 0.000 description 3
- 125000000623 heterocyclic group Chemical group 0.000 description 3
- 239000005457 ice water Substances 0.000 description 3
- 239000002547 new drug Substances 0.000 description 3
- 150000003833 nucleoside derivatives Chemical class 0.000 description 3
- 108090000623 proteins and genes Proteins 0.000 description 3
- 238000000746 purification Methods 0.000 description 3
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 3
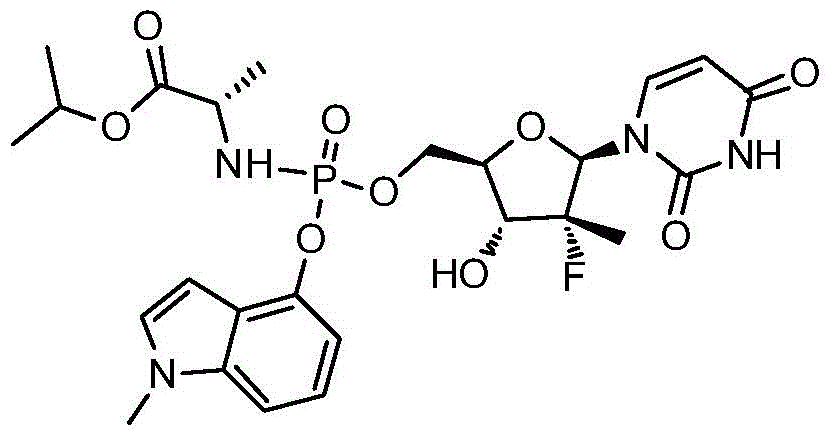
- TTZHDVOVKQGIBA-IQWMDFIBSA-N sofosbuvir Chemical compound N1([C@@H]2O[C@@H]([C@H]([C@]2(F)C)O)CO[P@@](=O)(N[C@@H](C)C(=O)OC(C)C)OC=2C=CC=CC=2)C=CC(=O)NC1=O TTZHDVOVKQGIBA-IQWMDFIBSA-N 0.000 description 3
- 238000006467 substitution reaction Methods 0.000 description 3
- 239000000758 substrate Substances 0.000 description 3
- 230000003612 virological effect Effects 0.000 description 3
- RARYKHVUFJIGKT-UHFFFAOYSA-N (2-fluoro-4-hydroxyphenyl)boronic acid Chemical compound OB(O)C1=CC=C(O)C=C1F RARYKHVUFJIGKT-UHFFFAOYSA-N 0.000 description 2
- RDCLVZNNBVGGFW-UHFFFAOYSA-N (4-bromo-1,3-thiazol-5-yl)methanol Chemical compound OCC=1SC=NC=1Br RDCLVZNNBVGGFW-UHFFFAOYSA-N 0.000 description 2
- 108091032973 (ribonucleotides)n+m Proteins 0.000 description 2
- BMRZGYNNZTVECK-UHFFFAOYSA-N 1-benzothiophen-4-ol Chemical compound OC1=CC=CC2=C1C=CS2 BMRZGYNNZTVECK-UHFFFAOYSA-N 0.000 description 2
- FNNWTXMUBMHNOD-UHFFFAOYSA-N 1-methylindol-5-ol Chemical compound OC1=CC=C2N(C)C=CC2=C1 FNNWTXMUBMHNOD-UHFFFAOYSA-N 0.000 description 2
- SULYEHHGGXARJS-UHFFFAOYSA-N 2',4'-dihydroxyacetophenone Chemical compound CC(=O)C1=CC=C(O)C=C1O SULYEHHGGXARJS-UHFFFAOYSA-N 0.000 description 2
- SSJFBYYLOSHJER-RGMNGODLSA-N 2,2-dimethylpropyl (2s)-2-aminopropanoate;hydrochloride Chemical compound Cl.C[C@H](N)C(=O)OCC(C)(C)C SSJFBYYLOSHJER-RGMNGODLSA-N 0.000 description 2
- QNCDPGOJVGDTAN-UHFFFAOYSA-N 2-bromo-1-(4-methoxyphenyl)propan-1-one Chemical compound COC1=CC=C(C(=O)C(C)Br)C=C1 QNCDPGOJVGDTAN-UHFFFAOYSA-N 0.000 description 2
- GOHBBINNYAWQGO-UHFFFAOYSA-N 2-bromo-3-chloropyridine Chemical compound ClC1=CC=CN=C1Br GOHBBINNYAWQGO-UHFFFAOYSA-N 0.000 description 2
- IFGLECYAEGYLSJ-UHFFFAOYSA-N 2-bromo-3-fluoropyridine Chemical compound FC1=CC=CN=C1Br IFGLECYAEGYLSJ-UHFFFAOYSA-N 0.000 description 2
- BZUUVQCSPHPUQA-UHFFFAOYSA-N 2-bromo-5-chloropyridine Chemical compound ClC1=CC=C(Br)N=C1 BZUUVQCSPHPUQA-UHFFFAOYSA-N 0.000 description 2
- YVCOYZFLAVPLLH-UHFFFAOYSA-N 2-fluoro-4-(1,3-thiazol-4-yl)phenol Chemical compound C1=C(F)C(O)=CC=C1C1=CSC=N1 YVCOYZFLAVPLLH-UHFFFAOYSA-N 0.000 description 2
- IOKCWQIRAQHNPH-UHFFFAOYSA-N 2-fluoro-4-(1-methylpyrazol-3-yl)phenol Chemical compound FC1=C(C=CC(=C1)C1=NN(C=C1)C)O IOKCWQIRAQHNPH-UHFFFAOYSA-N 0.000 description 2
- ZGEVJEQMVRIEPX-UHFFFAOYSA-N 3-bromo-1-methylpyrazole Chemical compound CN1C=CC(Br)=N1 ZGEVJEQMVRIEPX-UHFFFAOYSA-N 0.000 description 2
- XJXJAKANYJZWKA-UHFFFAOYSA-N 3-fluoro-4-(1,3-thiazol-4-yl)phenol Chemical compound FC1=CC(O)=CC=C1C1=CSC=N1 XJXJAKANYJZWKA-UHFFFAOYSA-N 0.000 description 2
- DQZKZJXIFWECIZ-UHFFFAOYSA-N 3-fluoro-4-(1-methylimidazol-4-yl)phenol Chemical compound FC=1C=C(C=CC=1C=1N=CN(C=1)C)O DQZKZJXIFWECIZ-UHFFFAOYSA-N 0.000 description 2
- IZKYNZFXZSIBLH-UHFFFAOYSA-N 3-methyl-1,2-benzoxazol-6-ol Chemical compound OC1=CC=C2C(C)=NOC2=C1 IZKYNZFXZSIBLH-UHFFFAOYSA-N 0.000 description 2
- QWZZKVQXNPQSQT-UHFFFAOYSA-N 3-methyl-4-(1,3-thiazol-4-yl)phenol Chemical compound CC1=CC(O)=CC=C1C1=CSC=N1 QWZZKVQXNPQSQT-UHFFFAOYSA-N 0.000 description 2
- PRPXETNOKTXAMQ-UHFFFAOYSA-N 3-methyl-4-(1-methylimidazol-4-yl)phenol Chemical compound CC=1C=C(C=CC=1C=1N=CN(C=1)C)O PRPXETNOKTXAMQ-UHFFFAOYSA-N 0.000 description 2
- QMIPCYMONNSFTD-UHFFFAOYSA-N 4-(1,3-oxazol-4-yl)phenol Chemical compound C1=CC(O)=CC=C1C1=COC=N1 QMIPCYMONNSFTD-UHFFFAOYSA-N 0.000 description 2
- RRBHMAJENUAYKG-UHFFFAOYSA-N 4-(1-methylpyrazol-4-yl)phenol Chemical compound C1=NN(C)C=C1C1=CC=C(O)C=C1 RRBHMAJENUAYKG-UHFFFAOYSA-N 0.000 description 2
- KYMRRZDVCYCEKW-UHFFFAOYSA-N 4-(2-chloropyridin-4-yl)phenol Chemical compound Oc1ccc(cc1)-c1ccnc(Cl)c1 KYMRRZDVCYCEKW-UHFFFAOYSA-N 0.000 description 2
- GOUDLCDKAKGSBL-UHFFFAOYSA-N 4-(2-fluoropyridin-4-yl)phenol Chemical compound C1=CC(=CC=C1C2=CC(=NC=C2)F)O GOUDLCDKAKGSBL-UHFFFAOYSA-N 0.000 description 2
- KETDAHHUUHZPRG-UHFFFAOYSA-N 4-(3,5-dimethyl-1,2-oxazol-4-yl)phenol Chemical compound CC1=NOC(C)=C1C1=CC=C(O)C=C1 KETDAHHUUHZPRG-UHFFFAOYSA-N 0.000 description 2
- WUTLAZSDFCABRR-UHFFFAOYSA-N 4-(3-chloropyridin-2-yl)-3-methylphenol Chemical compound ClC=1C(=NC=CC=1)C1=C(C=C(C=C1)O)C WUTLAZSDFCABRR-UHFFFAOYSA-N 0.000 description 2
- XXFBPIBVOIZSGC-UHFFFAOYSA-N 4-(3-fluoropyridin-2-yl)-3-methylphenol Chemical compound FC=1C(=NC=CC=1)C1=C(C=C(C=C1)O)C XXFBPIBVOIZSGC-UHFFFAOYSA-N 0.000 description 2
- NDYHGKODLQFNIS-UHFFFAOYSA-N 4-(4-chloro-1-methylpyrazol-3-yl)phenol Chemical compound ClC=1C(=NN(C=1)C)C1=CC=C(C=C1)O NDYHGKODLQFNIS-UHFFFAOYSA-N 0.000 description 2
- LKTNDBBNDGVMOX-UHFFFAOYSA-N 4-(4-hydroxyphenyl)-1,3-thiazole-5-carbaldehyde Chemical compound C(=O)C1=C(N=CS1)C1=CC=C(C=C1)O LKTNDBBNDGVMOX-UHFFFAOYSA-N 0.000 description 2
- ZXTBHLCOMGMOIP-UHFFFAOYSA-N 4-(4-hydroxyphenyl)-1,3-thiazole-5-carbonitrile Chemical compound C1=CC(=CC=C1C2=C(SC=N2)C#N)O ZXTBHLCOMGMOIP-UHFFFAOYSA-N 0.000 description 2
- JNCUHNIZNRLHDV-UHFFFAOYSA-N 4-(4-hydroxyphenyl)-1-methylpyrazole-3-carbonitrile Chemical compound N#CC1=NN(C)C=C1C1=CC=C(O)C=C1 JNCUHNIZNRLHDV-UHFFFAOYSA-N 0.000 description 2
- WUHYWCIHDXJYQR-UHFFFAOYSA-N 4-(4-hydroxyphenyl)-N,1-dimethylpyrazole-3-carboxamide Chemical compound CN1N=C(C(=C1)C1=CC=C(C=C1)O)C(=O)NC WUHYWCIHDXJYQR-UHFFFAOYSA-N 0.000 description 2
- DTXLNIJOPMFKHR-UHFFFAOYSA-N 4-(5-chloro-1,3-oxazol-4-yl)phenol Chemical compound C1=CC(=CC=C1C2=C(OC=N2)Cl)O DTXLNIJOPMFKHR-UHFFFAOYSA-N 0.000 description 2
- WXXCYPVPGONROC-UHFFFAOYSA-N 4-(5-chloro-1-methylimidazol-4-yl)phenol Chemical compound ClC1=C(N=CN1C)C1=CC=C(C=C1)O WXXCYPVPGONROC-UHFFFAOYSA-N 0.000 description 2
- WVBOOOSPTYEDLI-UHFFFAOYSA-N 4-(5-chloropyridin-2-yl)-3-methylphenol Chemical compound ClC=1C=CC(=NC=1)C1=C(C=C(C=C1)O)C WVBOOOSPTYEDLI-UHFFFAOYSA-N 0.000 description 2
- BFKBMBGHLWQRLI-UHFFFAOYSA-N 4-(5-chloropyridin-2-yl)phenol Chemical compound C1=CC(O)=CC=C1C1=CC=C(Cl)C=N1 BFKBMBGHLWQRLI-UHFFFAOYSA-N 0.000 description 2
- YRKVYQSAXKWMOX-UHFFFAOYSA-N 4-(5-fluoro-1,3-thiazol-4-yl)phenol Chemical compound FC1=C(N=CS1)C1=CC=C(C=C1)O YRKVYQSAXKWMOX-UHFFFAOYSA-N 0.000 description 2
- BEOXIFBRANCPFD-UHFFFAOYSA-N 4-(5-fluoropyridin-2-yl)phenol Chemical compound C1=CC(O)=CC=C1C1=CC=C(F)C=N1 BEOXIFBRANCPFD-UHFFFAOYSA-N 0.000 description 2
- ZRWIYIAZSGOCHK-UHFFFAOYSA-N 4-(5-methyl-1,3-oxazol-4-yl)phenol Chemical compound CC1=C(N=CO1)C1=CC=C(C=C1)O ZRWIYIAZSGOCHK-UHFFFAOYSA-N 0.000 description 2
- IRPJVORBRFNQAG-UHFFFAOYSA-N 4-(5-methyl-1,3-thiazol-4-yl)phenol Chemical compound S1C=NC(C=2C=CC(O)=CC=2)=C1C IRPJVORBRFNQAG-UHFFFAOYSA-N 0.000 description 2
- FLPSVRVLOVLWCQ-UHFFFAOYSA-N 4-bromo-1,3-thiazole-5-carbaldehyde Chemical compound BrC=1N=CSC=1C=O FLPSVRVLOVLWCQ-UHFFFAOYSA-N 0.000 description 2
- NTTLCOOFUGHMJS-UHFFFAOYSA-N 4-bromo-1-methylpyrazole-3-carbonitrile Chemical compound CN1C=C(Br)C(C#N)=N1 NTTLCOOFUGHMJS-UHFFFAOYSA-N 0.000 description 2
- LEEPGDCCHVRYHK-UHFFFAOYSA-N 4-bromo-1-methylpyrazole-3-carboxylic acid Chemical compound CN1C=C(Br)C(C(O)=O)=N1 LEEPGDCCHVRYHK-UHFFFAOYSA-N 0.000 description 2
- ZGENVPIDMFBEBJ-UHFFFAOYSA-N 4-bromo-5-(bromomethyl)-1,3-thiazole Chemical compound BrCc1scnc1Br ZGENVPIDMFBEBJ-UHFFFAOYSA-N 0.000 description 2
- VQHMPVXKDCHHSR-UHFFFAOYSA-N 4-pyridin-2-ylphenol Chemical compound C1=CC(O)=CC=C1C1=CC=CC=N1 VQHMPVXKDCHHSR-UHFFFAOYSA-N 0.000 description 2
- WQTVIWDQIXIEOD-UHFFFAOYSA-N 4-pyridin-3-ylphenol Chemical compound C1=CC(O)=CC=C1C1=CC=CN=C1 WQTVIWDQIXIEOD-UHFFFAOYSA-N 0.000 description 2
- DANMQSWQAGVFKV-UHFFFAOYSA-N 4-pyridin-4-ylphenol Chemical compound C1=CC(O)=CC=C1C1=CC=NC=C1 DANMQSWQAGVFKV-UHFFFAOYSA-N 0.000 description 2
- HNWBVGFOXICLIL-UHFFFAOYSA-N 5-chloro-4-(4-methoxyphenyl)-1,3-oxazole Chemical compound ClC1=C(N=CO1)C1=CC=C(C=C1)OC HNWBVGFOXICLIL-UHFFFAOYSA-N 0.000 description 2
- HQNPKVBTBJUMTR-UHFFFAOYSA-N 5-methoxy-1-methylindole Chemical compound COC1=CC=C2N(C)C=CC2=C1 HQNPKVBTBJUMTR-UHFFFAOYSA-N 0.000 description 2
- RLYUNPNLXMSXAX-UHFFFAOYSA-N 5-methylthiazole Chemical compound CC1=CN=CS1 RLYUNPNLXMSXAX-UHFFFAOYSA-N 0.000 description 2
- 125000004070 6 membered heterocyclic group Chemical group 0.000 description 2
- SIQFFBSANOANLX-UHFFFAOYSA-N COC1=CC=C(C=C1)C=1N=COC=1C Chemical compound COC1=CC=C(C=C1)C=1N=COC=1C SIQFFBSANOANLX-UHFFFAOYSA-N 0.000 description 2
- WGAQMMYWJSYOJO-UHFFFAOYSA-N ClC=1C(=NC=CC=1)C1=CC=C(C=C1)O Chemical compound ClC=1C(=NC=CC=1)C1=CC=C(C=C1)O WGAQMMYWJSYOJO-UHFFFAOYSA-N 0.000 description 2
- 241000710831 Flavivirus Species 0.000 description 2
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 description 2
- 229940126656 GS-4224 Drugs 0.000 description 2
- 241000124008 Mammalia Species 0.000 description 2
- 108060004795 Methyltransferase Proteins 0.000 description 2
- JGFZNNIVVJXRND-UHFFFAOYSA-N N,N-Diisopropylethylamine (DIPEA) Chemical compound CCN(C(C)C)C(C)C JGFZNNIVVJXRND-UHFFFAOYSA-N 0.000 description 2
- ZKNARUVAUOSLGV-UHFFFAOYSA-N N1N=CC=CC=C1.C1=CC=CC=C1 Chemical group N1N=CC=CC=C1.C1=CC=CC=C1 ZKNARUVAUOSLGV-UHFFFAOYSA-N 0.000 description 2
- 101800001014 Non-structural protein 5A Proteins 0.000 description 2
- 101150003085 Pdcl gene Proteins 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 2
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 2
- VMHLLURERBWHNL-UHFFFAOYSA-M Sodium acetate Chemical compound [Na+].CC([O-])=O VMHLLURERBWHNL-UHFFFAOYSA-M 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 2
- DRTQHJPVMGBUCF-XVFCMESISA-N Uridine Chemical compound O[C@@H]1[C@H](O)[C@@H](CO)O[C@H]1N1C(=O)NC(=O)C=C1 DRTQHJPVMGBUCF-XVFCMESISA-N 0.000 description 2
- 229910052786 argon Inorganic materials 0.000 description 2
- 150000007657 benzothiazepines Chemical class 0.000 description 2
- FJDQFPXHSGXQBY-UHFFFAOYSA-L caesium carbonate Chemical compound [Cs+].[Cs+].[O-]C([O-])=O FJDQFPXHSGXQBY-UHFFFAOYSA-L 0.000 description 2
- 229910000024 caesium carbonate Inorganic materials 0.000 description 2
- 238000002038 chemiluminescence detection Methods 0.000 description 2
- 208000019425 cirrhosis of liver Diseases 0.000 description 2
- 238000012258 culturing Methods 0.000 description 2
- 150000001924 cycloalkanes Chemical class 0.000 description 2
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 2
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 2
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 description 2
- 239000003085 diluting agent Substances 0.000 description 2
- 238000010790 dilution Methods 0.000 description 2
- 239000012895 dilution Substances 0.000 description 2
- XBDQKXXYIPTUBI-UHFFFAOYSA-N dimethylselenoniopropionate Natural products CCC(O)=O XBDQKXXYIPTUBI-UHFFFAOYSA-N 0.000 description 2
- 241001493065 dsRNA viruses Species 0.000 description 2
- 238000002474 experimental method Methods 0.000 description 2
- KTWOOEGAPBSYNW-UHFFFAOYSA-N ferrocene Chemical compound [Fe+2].C=1C=C[CH-]C=1.C=1C=C[CH-]C=1 KTWOOEGAPBSYNW-UHFFFAOYSA-N 0.000 description 2
- 108700008776 hepatitis C virus NS-5 Proteins 0.000 description 2
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N lactic acid Chemical compound CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- 208000019423 liver disease Diseases 0.000 description 2
- 239000000463 material Substances 0.000 description 2
- 229940098779 methanesulfonic acid Drugs 0.000 description 2
- YVLMYTLBNBNEPH-UHFFFAOYSA-N methyl 4-(4-hydroxyphenyl)-1-methylpyrazole-3-carboxylate Chemical compound CN1N=C(C(=C1)C1=CC=C(C=C1)O)C(=O)OC YVLMYTLBNBNEPH-UHFFFAOYSA-N 0.000 description 2
- PFKHXPUGZRGBPX-UHFFFAOYSA-N methyl 4-bromo-1-methylpyrazole-3-carboxylate Chemical compound COC(=O)C1=NN(C)C=C1Br PFKHXPUGZRGBPX-UHFFFAOYSA-N 0.000 description 2
- PIBWKRNGBLPSSY-UHFFFAOYSA-L palladium(II) chloride Chemical compound Cl[Pd]Cl PIBWKRNGBLPSSY-UHFFFAOYSA-L 0.000 description 2
- 239000000546 pharmaceutical excipient Substances 0.000 description 2
- 239000012071 phase Substances 0.000 description 2
- 229910052698 phosphorus Inorganic materials 0.000 description 2
- 239000011574 phosphorus Substances 0.000 description 2
- LYKMMUBOEFYJQG-UHFFFAOYSA-N piperoxan Chemical compound C1OC2=CC=CC=C2OC1CN1CCCCC1 LYKMMUBOEFYJQG-UHFFFAOYSA-N 0.000 description 2
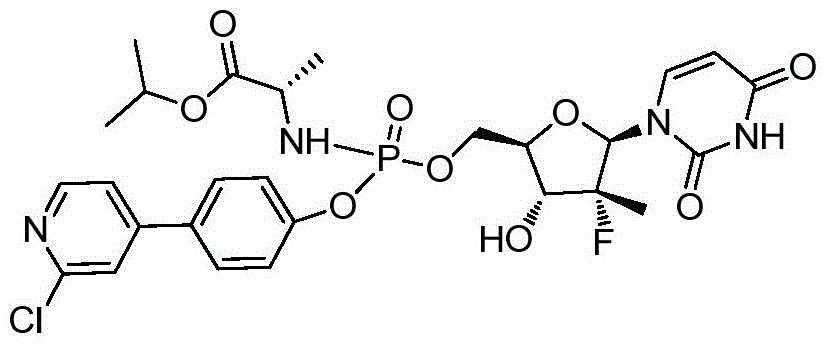
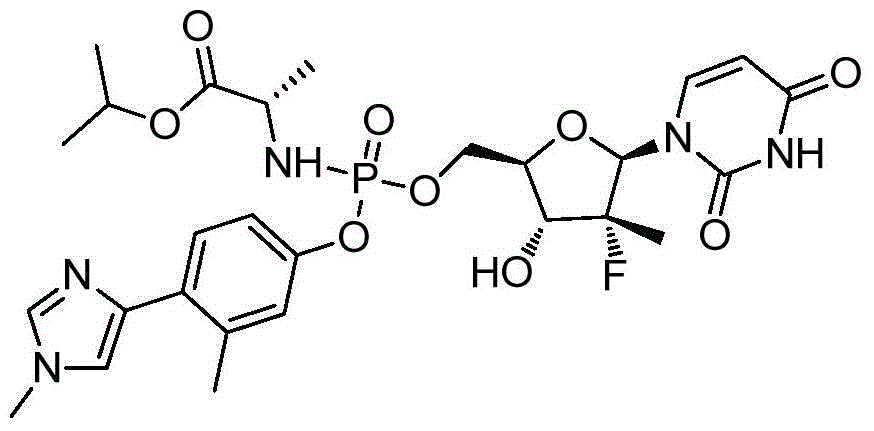
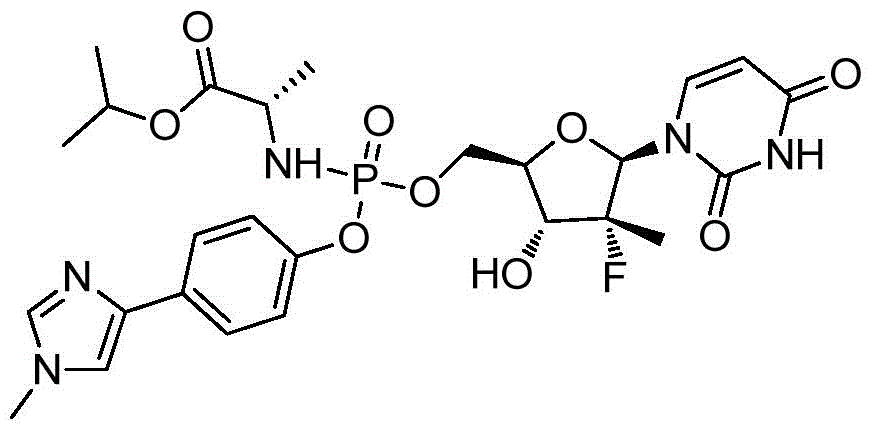
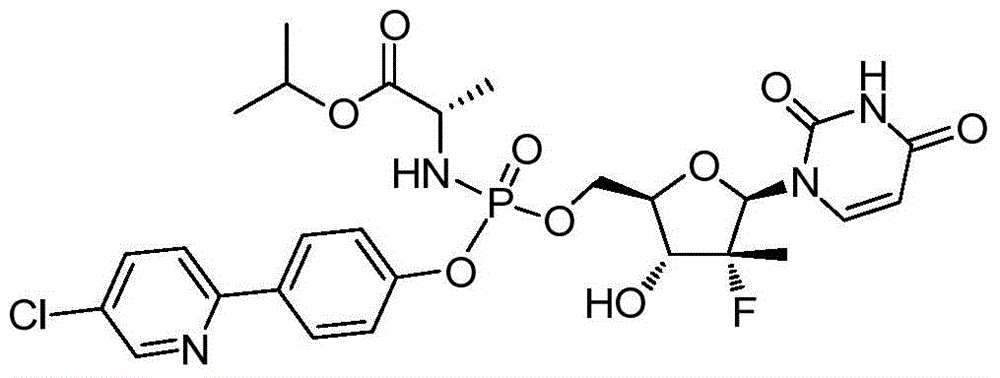
- NASPUKHMRFFREY-WQZFGADUSA-N propan-2-yl (2s)-2-[[[(2r,3r,4r,5r)-5-(2,4-dioxopyrimidin-1-yl)-4-fluoro-3-hydroxy-4-methyloxolan-2-yl]methoxy-[4-(1,3-thiazol-4-yl)phenoxy]phosphoryl]amino]propanoate Chemical compound N1([C@@H]2O[C@@H]([C@H]([C@]2(F)C)O)COP(=O)(N[C@@H](C)C(=O)OC(C)C)OC=2C=CC(=CC=2)C=2N=CSC=2)C=CC(=O)NC1=O NASPUKHMRFFREY-WQZFGADUSA-N 0.000 description 2
- 235000018102 proteins Nutrition 0.000 description 2
- 102000004169 proteins and genes Human genes 0.000 description 2
- 230000010076 replication Effects 0.000 description 2
- 238000012216 screening Methods 0.000 description 2
- 239000012363 selectfluor Substances 0.000 description 2
- 239000001632 sodium acetate Substances 0.000 description 2
- 235000017281 sodium acetate Nutrition 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- 238000003756 stirring Methods 0.000 description 2
- 239000011550 stock solution Substances 0.000 description 2
- VZGDMQKNWNREIO-UHFFFAOYSA-N tetrachloromethane Chemical compound ClC(Cl)(Cl)Cl VZGDMQKNWNREIO-UHFFFAOYSA-N 0.000 description 2
- FYSNRJHAOHDILO-UHFFFAOYSA-N thionyl chloride Chemical compound ClS(Cl)=O FYSNRJHAOHDILO-UHFFFAOYSA-N 0.000 description 2
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 2
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 2
- QAEDZJGFFMLHHQ-UHFFFAOYSA-N trifluoroacetic anhydride Chemical compound FC(F)(F)C(=O)OC(=O)C(F)(F)F QAEDZJGFFMLHHQ-UHFFFAOYSA-N 0.000 description 2
- 230000035899 viability Effects 0.000 description 2
- QBYIENPQHBMVBV-HFEGYEGKSA-N (2R)-2-hydroxy-2-phenylacetic acid Chemical compound O[C@@H](C(O)=O)c1ccccc1.O[C@@H](C(O)=O)c1ccccc1 QBYIENPQHBMVBV-HFEGYEGKSA-N 0.000 description 1
- HBZBAMXERPYTFS-SECBINFHSA-N (4S)-2-(6,7-dihydro-5H-pyrrolo[3,2-f][1,3]benzothiazol-2-yl)-4,5-dihydro-1,3-thiazole-4-carboxylic acid Chemical compound OC(=O)[C@H]1CSC(=N1)c1nc2cc3CCNc3cc2s1 HBZBAMXERPYTFS-SECBINFHSA-N 0.000 description 1
- BJEPYKJPYRNKOW-REOHCLBHSA-N (S)-malic acid Chemical compound OC(=O)[C@@H](O)CC(O)=O BJEPYKJPYRNKOW-REOHCLBHSA-N 0.000 description 1
- JPJGNZQDELRZGE-UHFFFAOYSA-N (phenyl-$l^{2}-phosphanyl)benzene Chemical compound C=1C=CC=CC=1[P]C1=CC=CC=C1 JPJGNZQDELRZGE-UHFFFAOYSA-N 0.000 description 1
- 102000040650 (ribonucleotides)n+m Human genes 0.000 description 1
- KZPYGQFFRCFCPP-UHFFFAOYSA-N 1,1'-bis(diphenylphosphino)ferrocene Chemical compound [Fe+2].C1=CC=C[C-]1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=C[C-]1P(C=1C=CC=CC=1)C1=CC=CC=C1 KZPYGQFFRCFCPP-UHFFFAOYSA-N 0.000 description 1
- OGYGFUAIIOPWQD-UHFFFAOYSA-N 1,3-thiazolidine Chemical compound C1CSCN1 OGYGFUAIIOPWQD-UHFFFAOYSA-N 0.000 description 1
- AZQWKYJCGOJGHM-UHFFFAOYSA-N 1,4-benzoquinone Chemical compound O=C1C=CC(=O)C=C1 AZQWKYJCGOJGHM-UHFFFAOYSA-N 0.000 description 1
- UBCHPRBFMUDMNC-UHFFFAOYSA-N 1-(1-adamantyl)ethanamine Chemical compound C1C(C2)CC3CC2CC1(C(N)C)C3 UBCHPRBFMUDMNC-UHFFFAOYSA-N 0.000 description 1
- NNIUCKWMURTFEZ-UHFFFAOYSA-N 1-methylindazol-5-ol Chemical compound OC1=CC=C2N(C)N=CC2=C1 NNIUCKWMURTFEZ-UHFFFAOYSA-N 0.000 description 1
- WSMJTXVQAZYLAV-UHFFFAOYSA-N 1-methylindol-4-ol Chemical compound C1=CC=C2N(C)C=CC2=C1O WSMJTXVQAZYLAV-UHFFFAOYSA-N 0.000 description 1
- 238000005160 1H NMR spectroscopy Methods 0.000 description 1
- YYJBWYBULYUKMR-UHFFFAOYSA-N 2-bromo-3-methylthiophene Chemical compound CC=1C=CSC=1Br YYJBWYBULYUKMR-UHFFFAOYSA-N 0.000 description 1
- UODINHBLNPPDPD-UHFFFAOYSA-N 2-bromo-5-fluoropyridine Chemical compound FC1=CC=C(Br)N=C1 UODINHBLNPPDPD-UHFFFAOYSA-N 0.000 description 1
- ACDLOOGOFKSUPO-UHFFFAOYSA-N 2-bromo-5-methylthiophene Chemical compound CC1=CC=C(Br)S1 ACDLOOGOFKSUPO-UHFFFAOYSA-N 0.000 description 1
- IMRWILPUOVGIMU-UHFFFAOYSA-N 2-bromopyridine Chemical compound BrC1=CC=CC=N1 IMRWILPUOVGIMU-UHFFFAOYSA-N 0.000 description 1
- TUCRZHGAIRVWTI-UHFFFAOYSA-N 2-bromothiophene Chemical compound BrC1=CC=CS1 TUCRZHGAIRVWTI-UHFFFAOYSA-N 0.000 description 1
- 125000004493 2-methylbut-1-yl group Chemical group CC(C*)CC 0.000 description 1
- VQGHOUODWALEFC-UHFFFAOYSA-N 2-phenylpyridine Chemical compound C1=CC=CC=C1C1=CC=CC=N1 VQGHOUODWALEFC-UHFFFAOYSA-N 0.000 description 1
- BMYNFMYTOJXKLE-UHFFFAOYSA-N 3-azaniumyl-2-hydroxypropanoate Chemical compound NCC(O)C(O)=O BMYNFMYTOJXKLE-UHFFFAOYSA-N 0.000 description 1
- NYPYPOZNGOXYSU-UHFFFAOYSA-N 3-bromopyridine Chemical compound BrC1=CC=CN=C1 NYPYPOZNGOXYSU-UHFFFAOYSA-N 0.000 description 1
- JBGDUBFDRQXGGK-UHFFFAOYSA-N 3-methyl-1-benzothiophen-4-ol Chemical compound C1=CC(O)=C2C(C)=CSC2=C1 JBGDUBFDRQXGGK-UHFFFAOYSA-N 0.000 description 1
- FQPRUMXSHZSJGM-WEVVVXLNSA-N 4-[(E)-N-hydroxy-C-methylcarbonimidoyl]benzene-1,3-diol Chemical compound C\C(=N/O)C1=C(O)C=C(O)C=C1 FQPRUMXSHZSJGM-WEVVVXLNSA-N 0.000 description 1
- IXJSDKIJPVSPKF-UHFFFAOYSA-N 4-bromo-1-methylpyrazole Chemical compound CN1C=C(Br)C=N1 IXJSDKIJPVSPKF-UHFFFAOYSA-N 0.000 description 1
- ONHMWUXYIFULDO-UHFFFAOYSA-N 4-bromo-2-chloropyridine Chemical compound ClC1=CC(Br)=CC=N1 ONHMWUXYIFULDO-UHFFFAOYSA-N 0.000 description 1
- PTPTZLXZHPPVKG-UHFFFAOYSA-N 4-bromo-2-fluoropyridine Chemical compound FC1=CC(Br)=CC=N1 PTPTZLXZHPPVKG-UHFFFAOYSA-N 0.000 description 1
- LYRXILTUZBBMNS-UHFFFAOYSA-N 4-bromo-2-methyl-1,3-thiazole Chemical compound CC1=NC(Br)=CS1 LYRXILTUZBBMNS-UHFFFAOYSA-N 0.000 description 1
- GYHZPSUAMYIFQD-UHFFFAOYSA-N 4-bromo-3,5-dimethyl-1,2-oxazole Chemical compound CC1=NOC(C)=C1Br GYHZPSUAMYIFQD-UHFFFAOYSA-N 0.000 description 1
- BSDGZUDFPKIYQG-UHFFFAOYSA-N 4-bromopyridine Chemical compound BrC1=CC=NC=C1 BSDGZUDFPKIYQG-UHFFFAOYSA-N 0.000 description 1
- ATEWBFOJLQMYEA-UHFFFAOYSA-N 4-methoxy-1-methylindole Chemical compound COC1=CC=CC2=C1C=CN2C ATEWBFOJLQMYEA-UHFFFAOYSA-N 0.000 description 1
- LUNOXNMCFPFPMO-UHFFFAOYSA-N 4-methoxy-1h-indole Chemical compound COC1=CC=CC2=C1C=CN2 LUNOXNMCFPFPMO-UHFFFAOYSA-N 0.000 description 1
- DWAQDRSOVMLGRQ-UHFFFAOYSA-N 5-methoxyindole Chemical compound COC1=CC=C2NC=CC2=C1 DWAQDRSOVMLGRQ-UHFFFAOYSA-N 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- HBAQYPYDRFILMT-UHFFFAOYSA-N 8-[3-(1-cyclopropylpyrazol-4-yl)-1H-pyrazolo[4,3-d]pyrimidin-5-yl]-3-methyl-3,8-diazabicyclo[3.2.1]octan-2-one Chemical class C1(CC1)N1N=CC(=C1)C1=NNC2=C1N=C(N=C2)N1C2C(N(CC1CC2)C)=O HBAQYPYDRFILMT-UHFFFAOYSA-N 0.000 description 1
- 206010056519 Abdominal infection Diseases 0.000 description 1
- 206010065051 Acute hepatitis C Diseases 0.000 description 1
- 229940077274 Alpha glucosidase inhibitor Drugs 0.000 description 1
- 108020005544 Antisense RNA Proteins 0.000 description 1
- 208000003950 B-cell lymphoma Diseases 0.000 description 1
- LSNNMFCWUKXFEE-UHFFFAOYSA-M Bisulfite Chemical compound OS([O-])=O LSNNMFCWUKXFEE-UHFFFAOYSA-M 0.000 description 1
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical group [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- 108090000994 Catalytic RNA Proteins 0.000 description 1
- 102000053642 Catalytic RNA Human genes 0.000 description 1
- 208000006154 Chronic hepatitis C Diseases 0.000 description 1
- 108091026890 Coding region Proteins 0.000 description 1
- 208000003322 Coinfection Diseases 0.000 description 1
- 206010010741 Conjunctivitis Diseases 0.000 description 1
- 208000019707 Cryoglobulinemic vasculitis Diseases 0.000 description 1
- 206010016654 Fibrosis Diseases 0.000 description 1
- 206010018364 Glomerulonephritis Diseases 0.000 description 1
- 239000007821 HATU Substances 0.000 description 1
- 229940124771 HCV-NS3 protease inhibitor Drugs 0.000 description 1
- 206010018910 Haemolysis Diseases 0.000 description 1
- 206010019663 Hepatic failure Diseases 0.000 description 1
- 208000005331 Hepatitis D Diseases 0.000 description 1
- 206010019799 Hepatitis viral Diseases 0.000 description 1
- 241000709715 Hepatovirus Species 0.000 description 1
- 101000600434 Homo sapiens Putative uncharacterized protein encoded by MIR7-3HG Proteins 0.000 description 1
- 206010061218 Inflammation Diseases 0.000 description 1
- 206010022004 Influenza like illness Diseases 0.000 description 1
- 101710200424 Inosine-5'-monophosphate dehydrogenase Proteins 0.000 description 1
- 102100040018 Interferon alpha-2 Human genes 0.000 description 1
- 108010079944 Interferon-alpha2b Proteins 0.000 description 1
- 102000013462 Interleukin-12 Human genes 0.000 description 1
- 108010065805 Interleukin-12 Proteins 0.000 description 1
- 102000000588 Interleukin-2 Human genes 0.000 description 1
- 108010002350 Interleukin-2 Proteins 0.000 description 1
- 102000004889 Interleukin-6 Human genes 0.000 description 1
- 108090001005 Interleukin-6 Proteins 0.000 description 1
- 102000005741 Metalloproteases Human genes 0.000 description 1
- 108010006035 Metalloproteases Proteins 0.000 description 1
- RJUFJBKOKNCXHH-UHFFFAOYSA-N Methyl propionate Chemical compound CCC(=O)OC RJUFJBKOKNCXHH-UHFFFAOYSA-N 0.000 description 1
- BAVYZALUXZFZLV-UHFFFAOYSA-N Methylamine Chemical compound NC BAVYZALUXZFZLV-UHFFFAOYSA-N 0.000 description 1
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 1
- 101800001019 Non-structural protein 4B Proteins 0.000 description 1
- JWIPGAFCGUDKEY-UHFFFAOYSA-L O[Cr](Cl)(=O)=O.C1=CC=NC=C1 Chemical compound O[Cr](Cl)(=O)=O.C1=CC=NC=C1 JWIPGAFCGUDKEY-UHFFFAOYSA-L 0.000 description 1
- 108091034117 Oligonucleotide Proteins 0.000 description 1
- 108010019160 Pancreatin Proteins 0.000 description 1
- 241000710778 Pestivirus Species 0.000 description 1
- 206010035148 Plague Diseases 0.000 description 1
- 241000097929 Porphyria Species 0.000 description 1
- 208000010642 Porphyrias Diseases 0.000 description 1
- 102100037401 Putative uncharacterized protein encoded by MIR7-3HG Human genes 0.000 description 1
- IWYDHOAUDWTVEP-UHFFFAOYSA-N R-2-phenyl-2-hydroxyacetic acid Natural products OC(=O)C(O)C1=CC=CC=C1 IWYDHOAUDWTVEP-UHFFFAOYSA-N 0.000 description 1
- 108010052090 Renilla Luciferases Proteins 0.000 description 1
- 108700008625 Reporter Genes Proteins 0.000 description 1
- PLXBWHJQWKZRKG-UHFFFAOYSA-N Resazurin Chemical compound C1=CC(=O)C=C2OC3=CC(O)=CC=C3[N+]([O-])=C21 PLXBWHJQWKZRKG-UHFFFAOYSA-N 0.000 description 1
- JAXLHKGQYFBYIG-UHFFFAOYSA-N S1N=NC=C1.C1=CC=CC=C1 Chemical group S1N=NC=C1.C1=CC=CC=C1 JAXLHKGQYFBYIG-UHFFFAOYSA-N 0.000 description 1
- 102000012479 Serine Proteases Human genes 0.000 description 1
- 108010022999 Serine Proteases Proteins 0.000 description 1
- 101800001838 Serine protease/helicase NS3 Proteins 0.000 description 1
- KEAYESYHFKHZAL-UHFFFAOYSA-N Sodium Chemical compound [Na] KEAYESYHFKHZAL-UHFFFAOYSA-N 0.000 description 1
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical class [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 1
- 244000061458 Solanum melongena Species 0.000 description 1
- 101710172711 Structural protein Proteins 0.000 description 1
- 108091027544 Subgenomic mRNA Proteins 0.000 description 1
- KDYFGRWQOYBRFD-UHFFFAOYSA-N Succinic acid Natural products OC(=O)CCC(O)=O KDYFGRWQOYBRFD-UHFFFAOYSA-N 0.000 description 1
- 206010046274 Upper gastrointestinal haemorrhage Diseases 0.000 description 1
- 241000607479 Yersinia pestis Species 0.000 description 1
- JLCPHMBAVCMARE-UHFFFAOYSA-N [3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-[[3-[[3-[[3-[[3-[[3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-hydroxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methyl [5-(6-aminopurin-9-yl)-2-(hydroxymethyl)oxolan-3-yl] hydrogen phosphate Polymers Cc1cn(C2CC(OP(O)(=O)OCC3OC(CC3OP(O)(=O)OCC3OC(CC3O)n3cnc4c3nc(N)[nH]c4=O)n3cnc4c3nc(N)[nH]c4=O)C(COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3CO)n3cnc4c(N)ncnc34)n3ccc(N)nc3=O)n3cnc4c(N)ncnc34)n3ccc(N)nc3=O)n3ccc(N)nc3=O)n3ccc(N)nc3=O)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cc(C)c(=O)[nH]c3=O)n3cc(C)c(=O)[nH]c3=O)n3ccc(N)nc3=O)n3cc(C)c(=O)[nH]c3=O)n3cnc4c3nc(N)[nH]c4=O)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)O2)c(=O)[nH]c1=O JLCPHMBAVCMARE-UHFFFAOYSA-N 0.000 description 1
- 230000002159 abnormal effect Effects 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- GPWHDDKQSYOYBF-UHFFFAOYSA-N ac1l2u0q Chemical compound Br[Br-]Br GPWHDDKQSYOYBF-UHFFFAOYSA-N 0.000 description 1
- 208000037621 acute hepatitis C virus infection Diseases 0.000 description 1
- 125000004442 acylamino group Chemical group 0.000 description 1
- 150000001345 alkine derivatives Chemical class 0.000 description 1
- 239000003888 alpha glucosidase inhibitor Substances 0.000 description 1
- BJEPYKJPYRNKOW-UHFFFAOYSA-N alpha-hydroxysuccinic acid Natural products OC(=O)C(O)CC(O)=O BJEPYKJPYRNKOW-UHFFFAOYSA-N 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- DKNWSYNQZKUICI-UHFFFAOYSA-N amantadine Chemical compound C1C(C2)CC3CC2CC1(N)C3 DKNWSYNQZKUICI-UHFFFAOYSA-N 0.000 description 1
- 229960003805 amantadine Drugs 0.000 description 1
- 235000019270 ammonium chloride Nutrition 0.000 description 1
- 235000011114 ammonium hydroxide Nutrition 0.000 description 1
- 208000007502 anemia Diseases 0.000 description 1
- 230000000692 anti-sense effect Effects 0.000 description 1
- 239000003443 antiviral agent Substances 0.000 description 1
- 238000013459 approach Methods 0.000 description 1
- 125000002029 aromatic hydrocarbon group Chemical group 0.000 description 1
- 238000003556 assay Methods 0.000 description 1
- ZVSKZLHKADLHSD-UHFFFAOYSA-N benzanilide Chemical class C=1C=CC=CC=1C(=O)NC1=CC=CC=C1 ZVSKZLHKADLHSD-UHFFFAOYSA-N 0.000 description 1
- LZLOFGMGFADIKQ-UHFFFAOYSA-N benzene;1,4-dioxane Chemical compound C1COCCO1.C1=CC=CC=C1 LZLOFGMGFADIKQ-UHFFFAOYSA-N 0.000 description 1
- DRTQHJPVMGBUCF-PSQAKQOGSA-N beta-L-uridine Natural products O[C@H]1[C@@H](O)[C@H](CO)O[C@@H]1N1C(=O)NC(=O)C=C1 DRTQHJPVMGBUCF-PSQAKQOGSA-N 0.000 description 1
- KDYFGRWQOYBRFD-NUQCWPJISA-N butanedioic acid Chemical compound O[14C](=O)CC[14C](O)=O KDYFGRWQOYBRFD-NUQCWPJISA-N 0.000 description 1
- 239000002775 capsule Substances 0.000 description 1
- 239000000969 carrier Substances 0.000 description 1
- 238000004113 cell culture Methods 0.000 description 1
- 239000006143 cell culture medium Substances 0.000 description 1
- 230000010261 cell growth Effects 0.000 description 1
- 238000003570 cell viability assay Methods 0.000 description 1
- 208000020403 chronic hepatitis C virus infection Diseases 0.000 description 1
- 230000007882 cirrhosis Effects 0.000 description 1
- 238000010367 cloning Methods 0.000 description 1
- 238000002648 combination therapy Methods 0.000 description 1
- 239000003184 complementary RNA Substances 0.000 description 1
- 238000003271 compound fluorescence assay Methods 0.000 description 1
- 201000003278 cryoglobulinemia Diseases 0.000 description 1
- 239000000134 cyclophilin inhibitor Substances 0.000 description 1
- 238000002784 cytotoxicity assay Methods 0.000 description 1
- 231100000263 cytotoxicity test Toxicity 0.000 description 1
- 230000001419 dependent effect Effects 0.000 description 1
- OKVJWADVFPXWQD-UHFFFAOYSA-N difluoroborinic acid Chemical compound OB(F)F OKVJWADVFPXWQD-UHFFFAOYSA-N 0.000 description 1
- VAYGXNSJCAHWJZ-UHFFFAOYSA-N dimethyl sulfate Chemical compound COS(=O)(=O)OC VAYGXNSJCAHWJZ-UHFFFAOYSA-N 0.000 description 1
- GPAYUJZHTULNBE-UHFFFAOYSA-O diphenylphosphanium Chemical compound C=1C=CC=CC=1[PH2+]C1=CC=CC=C1 GPAYUJZHTULNBE-UHFFFAOYSA-O 0.000 description 1
- 229940042399 direct acting antivirals protease inhibitors Drugs 0.000 description 1
- 239000000839 emulsion Substances 0.000 description 1
- 210000000981 epithelium Anatomy 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 206010016256 fatigue Diseases 0.000 description 1
- 239000012091 fetal bovine serum Substances 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- 239000001530 fumaric acid Substances 0.000 description 1
- 125000002541 furyl group Chemical group 0.000 description 1
- 108020001507 fusion proteins Proteins 0.000 description 1
- 102000037865 fusion proteins Human genes 0.000 description 1
- 230000002068 genetic effect Effects 0.000 description 1
- 208000005017 glioblastoma Diseases 0.000 description 1
- 229930182470 glycoside Natural products 0.000 description 1
- 150000002338 glycosides Chemical class 0.000 description 1
- 239000008187 granular material Substances 0.000 description 1
- 230000008588 hemolysis Effects 0.000 description 1
- 208000007386 hepatic encephalopathy Diseases 0.000 description 1
- 208000002672 hepatitis B Diseases 0.000 description 1
- 206010073071 hepatocellular carcinoma Diseases 0.000 description 1
- 201000011200 hepatorenal syndrome Diseases 0.000 description 1
- 230000000887 hydrating effect Effects 0.000 description 1
- 229960002751 imiquimod Drugs 0.000 description 1
- DOUYETYNHWVLEO-UHFFFAOYSA-N imiquimod Chemical compound C1=CC=CC2=C3N(CC(C)C)C=NC3=C(N)N=C21 DOUYETYNHWVLEO-UHFFFAOYSA-N 0.000 description 1
- 230000028993 immune response Effects 0.000 description 1
- 238000000338 in vitro Methods 0.000 description 1
- 230000002458 infectious effect Effects 0.000 description 1
- 230000004054 inflammatory process Effects 0.000 description 1
- 238000001802 infusion Methods 0.000 description 1
- 239000007924 injection Substances 0.000 description 1
- 238000002347 injection Methods 0.000 description 1
- 239000002348 inosinate dehydrogenase inhibitor Substances 0.000 description 1
- 230000002452 interceptive effect Effects 0.000 description 1
- 229940117681 interleukin-12 Drugs 0.000 description 1
- 229940100601 interleukin-6 Drugs 0.000 description 1
- 238000007918 intramuscular administration Methods 0.000 description 1
- 238000007912 intraperitoneal administration Methods 0.000 description 1
- 238000001990 intravenous administration Methods 0.000 description 1
- 239000011630 iodine Substances 0.000 description 1
- 229910052740 iodine Inorganic materials 0.000 description 1
- INQOMBQAUSQDDS-UHFFFAOYSA-N iodomethane Chemical compound IC INQOMBQAUSQDDS-UHFFFAOYSA-N 0.000 description 1
- 125000004491 isohexyl group Chemical group C(CCC(C)C)* 0.000 description 1
- 125000001786 isothiazolyl group Chemical group 0.000 description 1
- 239000004310 lactic acid Substances 0.000 description 1
- 235000014655 lactic acid Nutrition 0.000 description 1
- 201000011486 lichen planus Diseases 0.000 description 1
- 239000008297 liquid dosage form Substances 0.000 description 1
- 201000007270 liver cancer Diseases 0.000 description 1
- 208000007903 liver failure Diseases 0.000 description 1
- 231100000835 liver failure Toxicity 0.000 description 1
- 208000014018 liver neoplasm Diseases 0.000 description 1
- 238000004020 luminiscence type Methods 0.000 description 1
- CQRPUKWAZPZXTO-UHFFFAOYSA-M magnesium;2-methylpropane;chloride Chemical compound [Mg+2].[Cl-].C[C-](C)C CQRPUKWAZPZXTO-UHFFFAOYSA-M 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- 239000011976 maleic acid Substances 0.000 description 1
- 239000001630 malic acid Substances 0.000 description 1
- 235000011090 malic acid Nutrition 0.000 description 1
- 229960002510 mandelic acid Drugs 0.000 description 1
- FFEARJCKVFRZRR-UHFFFAOYSA-N methionine Chemical compound CSCCC(N)C(O)=O FFEARJCKVFRZRR-UHFFFAOYSA-N 0.000 description 1
- 229940017219 methyl propionate Drugs 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 210000002200 mouth mucosa Anatomy 0.000 description 1
- 125000001280 n-hexyl group Chemical group C(CCCCC)* 0.000 description 1
- 125000001971 neopentyl group Chemical group [H]C([*])([H])C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 229910017604 nitric acid Inorganic materials 0.000 description 1
- TVMXDCGIABBOFY-UHFFFAOYSA-N octane Chemical compound CCCCCCCC TVMXDCGIABBOFY-UHFFFAOYSA-N 0.000 description 1
- 125000001715 oxadiazolyl group Chemical group 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- ZJVAWPKTWVFKHG-UHFFFAOYSA-N p-Methoxypropiophenone Chemical compound CCC(=O)C1=CC=C(OC)C=C1 ZJVAWPKTWVFKHG-UHFFFAOYSA-N 0.000 description 1
- KDLHZDBZIXYQEI-UHFFFAOYSA-N palladium Substances [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 1
- 229940055695 pancreatin Drugs 0.000 description 1
- 238000007911 parenteral administration Methods 0.000 description 1
- 239000000137 peptide hydrolase inhibitor Substances 0.000 description 1
- 206010034674 peritonitis Diseases 0.000 description 1
- 230000000144 pharmacologic effect Effects 0.000 description 1
- SCOAVUHOIJMIBW-UHFFFAOYSA-N phenanthrene-1,2-dione Chemical class C1=CC=C2C(C=CC(C3=O)=O)=C3C=CC2=C1 SCOAVUHOIJMIBW-UHFFFAOYSA-N 0.000 description 1
- 239000008363 phosphate buffer Substances 0.000 description 1
- 239000006187 pill Substances 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 239000000047 product Substances 0.000 description 1
- QDQVXVRZVCTVHE-YFKPBYRVSA-N propan-2-yl (2s)-2-aminopropanoate Chemical compound CC(C)OC(=O)[C@H](C)N QDQVXVRZVCTVHE-YFKPBYRVSA-N 0.000 description 1
- 235000019260 propionic acid Nutrition 0.000 description 1
- 125000002098 pyridazinyl group Chemical group 0.000 description 1
- 125000004076 pyridyl group Chemical group 0.000 description 1
- 239000002718 pyrimidine nucleoside Substances 0.000 description 1
- IUVKMZGDUIUOCP-BTNSXGMBSA-N quinbolone Chemical compound O([C@H]1CC[C@H]2[C@H]3[C@@H]([C@]4(C=CC(=O)C=C4CC3)C)CC[C@@]21C)C1=CCCC1 IUVKMZGDUIUOCP-BTNSXGMBSA-N 0.000 description 1
- 238000005215 recombination Methods 0.000 description 1
- 210000000664 rectum Anatomy 0.000 description 1
- 238000012827 research and development Methods 0.000 description 1
- 206010039073 rheumatoid arthritis Diseases 0.000 description 1
- 108091092562 ribozyme Proteins 0.000 description 1
- 229960000888 rimantadine Drugs 0.000 description 1
- 239000003730 rna directed rna polymerase inhibitor Substances 0.000 description 1
- 229910000029 sodium carbonate Inorganic materials 0.000 description 1
- 239000012312 sodium hydride Substances 0.000 description 1
- 229910000104 sodium hydride Inorganic materials 0.000 description 1
- SNOOUWRIMMFWNE-UHFFFAOYSA-M sodium;6-[(3,4,5-trimethoxybenzoyl)amino]hexanoate Chemical compound [Na+].COC1=CC(C(=O)NCCCCCC([O-])=O)=CC(OC)=C1OC SNOOUWRIMMFWNE-UHFFFAOYSA-M 0.000 description 1
- 229960002063 sofosbuvir Drugs 0.000 description 1
- 239000007909 solid dosage form Substances 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 229940076563 sovaldi Drugs 0.000 description 1
- 238000011272 standard treatment Methods 0.000 description 1
- 229960005322 streptomycin Drugs 0.000 description 1
- 238000007920 subcutaneous administration Methods 0.000 description 1
- 239000006228 supernatant Substances 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 239000006188 syrup Substances 0.000 description 1
- 235000020357 syrup Nutrition 0.000 description 1
- 230000009885 systemic effect Effects 0.000 description 1
- 239000003826 tablet Substances 0.000 description 1
- NHKZSTHOYNWEEZ-AFCXAGJDSA-N taribavirin Chemical compound N1=C(C(=N)N)N=CN1[C@H]1[C@H](O)[C@H](O)[C@@H](CO)O1 NHKZSTHOYNWEEZ-AFCXAGJDSA-N 0.000 description 1
- 229950006081 taribavirin Drugs 0.000 description 1
- 125000003831 tetrazolyl group Chemical group 0.000 description 1
- 230000001225 therapeutic effect Effects 0.000 description 1
- 125000001113 thiadiazolyl group Chemical group 0.000 description 1
- 150000003548 thiazolidines Chemical class 0.000 description 1
- 125000000335 thiazolyl group Chemical group 0.000 description 1
- 125000001544 thienyl group Chemical group 0.000 description 1
- RYYWUUFWQRZTIU-UHFFFAOYSA-K thiophosphate Chemical compound [O-]P([O-])([O-])=S RYYWUUFWQRZTIU-UHFFFAOYSA-K 0.000 description 1
- 238000004448 titration Methods 0.000 description 1
- 229940044616 toll-like receptor 7 agonist Drugs 0.000 description 1
- 229940044655 toll-like receptor 9 agonist Drugs 0.000 description 1
- 239000003053 toxin Substances 0.000 description 1
- 231100000765 toxin Toxicity 0.000 description 1
- 108700012359 toxins Proteins 0.000 description 1
- 238000013519 translation Methods 0.000 description 1
- 125000004306 triazinyl group Chemical group 0.000 description 1
- 125000001425 triazolyl group Chemical group 0.000 description 1
- DRTQHJPVMGBUCF-UHFFFAOYSA-N uracil arabinoside Natural products OC1C(O)C(CO)OC1N1C(=O)NC(=O)C=C1 DRTQHJPVMGBUCF-UHFFFAOYSA-N 0.000 description 1
- 229940045145 uridine Drugs 0.000 description 1
- 239000003981 vehicle Substances 0.000 description 1
- 201000001862 viral hepatitis Diseases 0.000 description 1
- 238000005303 weighing Methods 0.000 description 1
- 230000004580 weight loss Effects 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7042—Compounds having saccharide radicals and heterocyclic rings
- A61K31/7052—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides
- A61K31/706—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides containing six-membered rings with nitrogen as a ring hetero atom
- A61K31/7064—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides containing six-membered rings with nitrogen as a ring hetero atom containing condensed or non-condensed pyrimidines
- A61K31/7068—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides containing six-membered rings with nitrogen as a ring hetero atom containing condensed or non-condensed pyrimidines having oxo groups directly attached to the pyrimidine ring, e.g. cytidine, cytidylic acid
- A61K31/7072—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides containing six-membered rings with nitrogen as a ring hetero atom containing condensed or non-condensed pyrimidines having oxo groups directly attached to the pyrimidine ring, e.g. cytidine, cytidylic acid having two oxo groups directly attached to the pyrimidine ring, e.g. uridine, uridylic acid, thymidine, zidovudine
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic Table
- C07F9/02—Phosphorus compounds
- C07F9/06—Phosphorus compounds without P—C bonds
- C07F9/22—Amides of acids of phosphorus
- C07F9/24—Esteramides
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Organic Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Molecular Biology (AREA)
- Biochemistry (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
本发明属于医药化学领域,具体涉及一类具有良好丙型肝炎病毒抑制效果的化合物,其制备方法,含有该化合物的组合物,以及所述化合物或组合物作为丙型肝炎病毒感染性疾病治疗药物的用途。本发明的化合物表现出优异的抗病毒活性,同时具有小的细胞毒性,安全性好,且血浆蛋白结合率良好,适于成药,具有非常好的临床应用前景。The invention belongs to the field of medicinal chemistry, and in particular relates to a class of compounds with good hepatitis C virus inhibitory effect, a preparation method thereof, a composition containing the compound, and the compound or composition as a therapeutic drug for hepatitis C virus infectious disease the use of. The compound of the present invention exhibits excellent antiviral activity, has small cytotoxicity, good safety, and good plasma protein binding rate, is suitable for medicine, and has a very good clinical application prospect.
Description
技术领域technical field
本发明属于医药化学领域,具体涉及一类具有良好丙型肝炎病毒抑制效果的化合物,其制备方法,含有该化合物的组合物,以及所述化合物或组合物作为丙型肝炎病毒感染性疾病治疗药物的用途。The invention belongs to the field of medicinal chemistry, and in particular relates to a class of compounds with good hepatitis C virus inhibitory effect, a preparation method thereof, a composition containing the compound, and the compound or composition as a therapeutic drug for hepatitis C virus infectious disease the use of.
背景技术Background technique
丙型肝炎病毒(HCV)感染是世界范围内流行的疾病,全球慢性感染者已超过2亿,中国感染率为3.2%,位列世界前三。丙型肝炎病毒感染的临床表现多样,轻至炎症,重至肝硬化、肝癌。慢性丙型肝炎还可以并发某些肝外表现,包括类风湿性关节炎、干燥性结膜角膜炎、扁平苔藓、肾小球肾炎、混合型冷球蛋白血症、B细胞淋巴瘤和迟发性皮肤卟啉症等,可能是机体异常免疫反应所致。丙肝肝硬化失代偿期时,可以出现各种并发症,如腹水腹腔感染,上消化道出血,肝性脑病,肝肾综合征,肝衰竭等表现。Hepatitis C virus (HCV) infection is a prevalent disease worldwide, with more than 200 million chronically infected people worldwide. The infection rate in China is 3.2%, ranking among the top three in the world. The clinical manifestations of HCV infection are diverse, ranging from mild inflammation to severe liver cirrhosis and liver cancer. Chronic hepatitis C can also be complicated by certain extrahepatic manifestations, including rheumatoid arthritis, conjunctivitis sicca, lichen planus, glomerulonephritis, mixed cryoglobulinemia, B-cell lymphoma, and late-onset Skin porphyria, etc., may be caused by abnormal immune response of the body. In the decompensated stage of hepatitis C cirrhosis, various complications can occur, such as ascites and abdominal infection, upper gastrointestinal bleeding, hepatic encephalopathy, hepatorenal syndrome, and liver failure.
HCV属于黄病毒科肝病毒属病毒,其与黄病毒科中的另外两个属即瘟病毒属和黄病毒属的基因结构相似。目前,治疗HCV感染的标准方法有干扰素以及干扰素和利巴韦林联合疗法。不过,仅50%的治疗者对该方法有反应,且干扰素具有明显的副作用,例如流行性感冒样症状、体重减低以及疲乏无力,而干扰素和利巴韦林联合疗法则产生相当大的副作用,包括溶血、贫血症和疲乏等。HCV belongs to the Flaviviridae family of viruses of the genus Hepatovirus, which is similar in genetic structure to two other genera in the Flaviviridae family, Pestivirus and Flavivirus. Currently, the standard treatments for HCV infection are interferon and combination therapy with interferon and ribavirin. However, only 50% of those treated responded to the approach, and interferon had significant side effects such as influenza-like symptoms, weight loss, and fatigue, while the combination of interferon and ribavirin produced considerable Side effects, including hemolysis, anemia, and fatigue.
另外,已开发的用于治疗HCV感染的药物包括蛋白酶抑制剂、噻唑烷衍生物、噻唑烷和N-苯甲酰苯胺、菲醌、解旋酶抑制剂、核苷聚合酶抑制剂和胶霉毒素、反义硫代磷酸酯寡核苷酸、依赖于IRES的翻译的抑制剂、核酶以及核苷类似物等。In addition, drugs that have been developed for the treatment of HCV infection include protease inhibitors, thiazolidine derivatives, thiazolidine and benzoic anilides, phenanthrenequinones, helicase inhibitors, nucleoside polymerase inhibitors and glioblastoma Toxins, antisense phosphorothioate oligonucleotides, inhibitors of IRES-dependent translation, ribozymes, and nucleoside analogs, etc.
目前,核苷磷酸酯类化合物用于治疗黄病毒科病毒尤其是HCV感染是本领域的重要研发方向。WO 2006/065335公开了一种抑制HCV病毒的氟化吡咯并[2,3,d]嘧啶核苷化合物。US 2006/0241064公开了用于治疗黄病毒科家族病毒如HCV引起的病毒感染的核苷化合物。WO 2008/121634公开了用于治疗哺乳动物病毒性感染的核苷氨基磷酸酯化合物。At present, the use of nucleoside phosphate compounds for the treatment of Flaviviridae viruses, especially HCV infection, is an important research and development direction in this field. WO 2006/065335 discloses a fluorinated pyrrolo[2,3,d]pyrimidine nucleoside compound that inhibits HCV virus. US 2006/0241064 discloses nucleoside compounds for the treatment of viral infections caused by viruses of the Flaviviridae family, such as HCV. WO 2008/121634 discloses nucleoside phosphoramidate compounds for use in the treatment of viral infections in mammals.
尽管存在上述公开,目前仍然非常需要用于治疗和/或预防HCV感染的有效化合物。Despite the above disclosures, there is still a great need for effective compounds for the treatment and/or prevention of HCV infection.
发明内容SUMMARY OF THE INVENTION
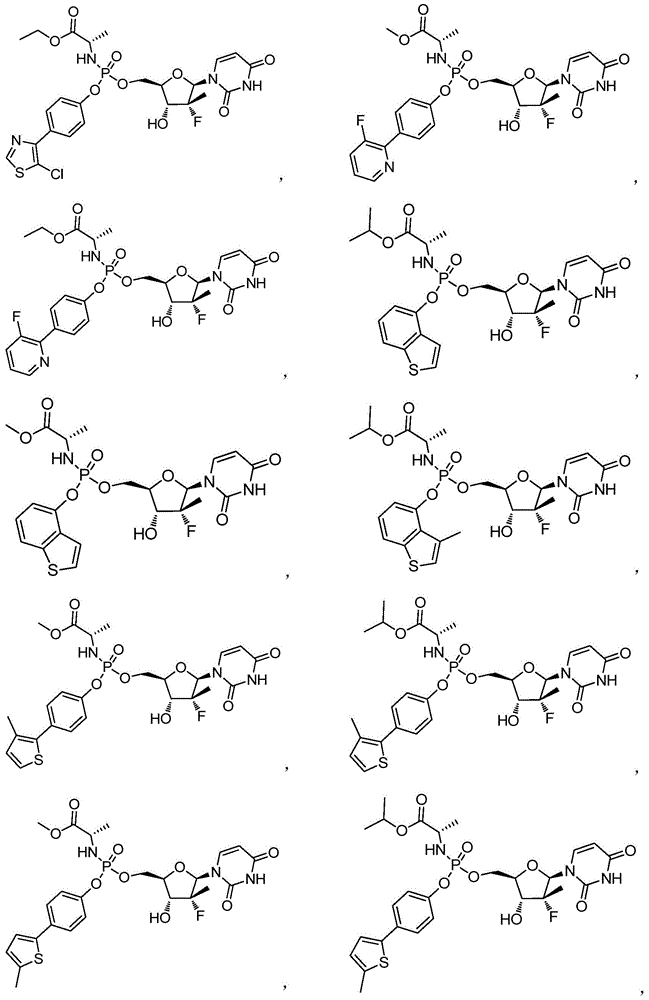
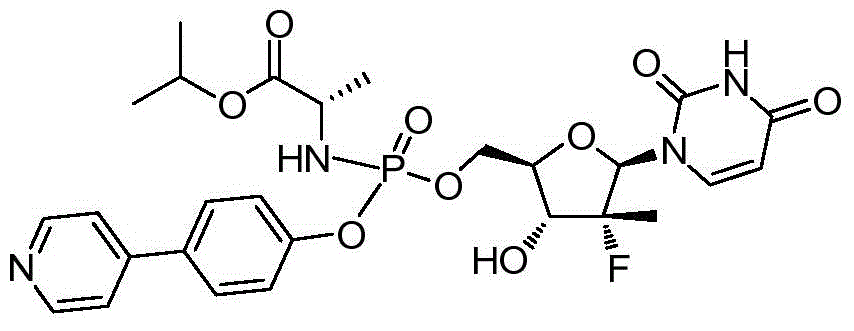
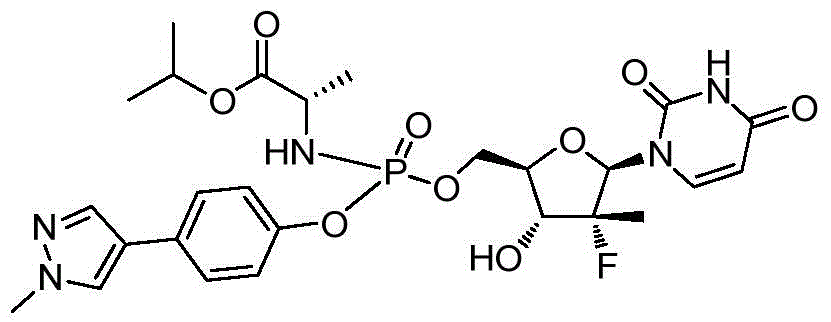
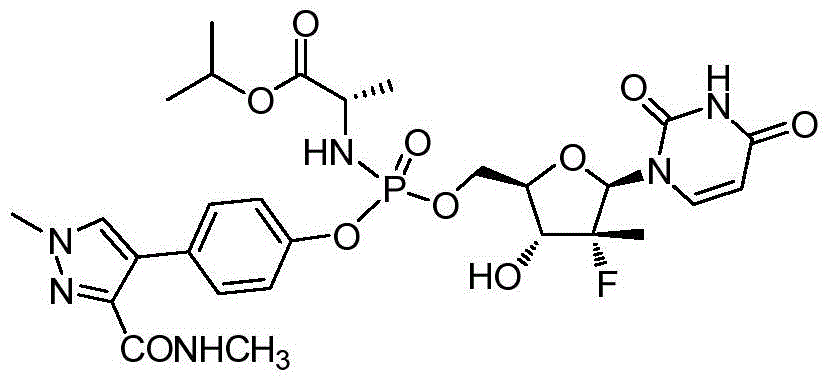
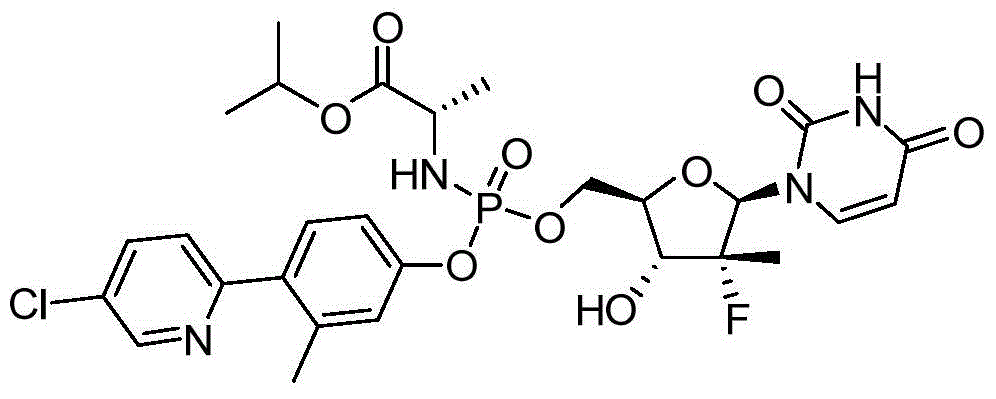
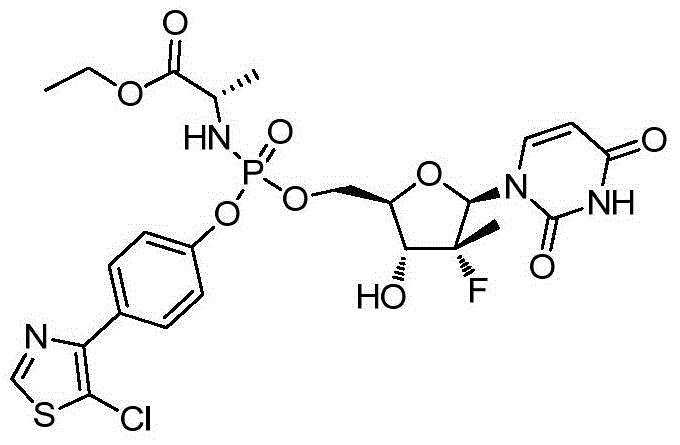
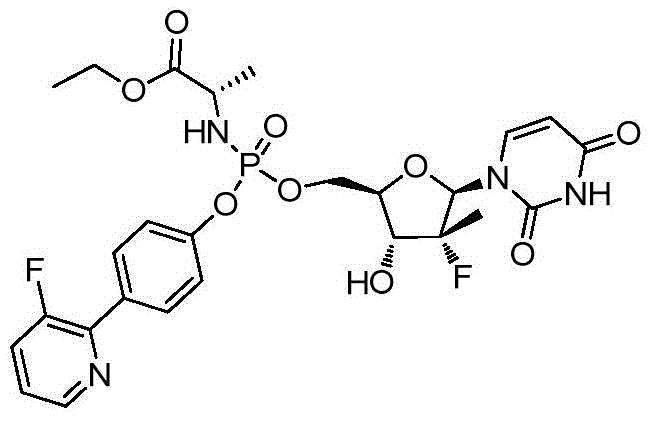
本发明的一个目的是提供用于治疗和/或预防丙型肝炎病毒感染的通式I所示的化合物或其立体异构体、药学可接受的盐、水合物、溶剂合物或结晶:One object of the present invention is to provide a compound of general formula I or its stereoisomer, pharmaceutically acceptable salt, hydrate, solvate or crystal for treating and/or preventing hepatitis C virus infection:
本发明的另一个目的是提供制备本发明的通式I的化合物或其立体异构体、药学可接受的盐、水合物、溶剂合物或结晶的方法。Another object of the present invention is to provide a process for preparing the compounds of general formula I of the present invention or stereoisomers, pharmaceutically acceptable salts, hydrates, solvates or crystals thereof.
本发明的再一个目的是提供包含本发明的通式I的化合物或其立体异构体、药学可接受的盐、水合物、溶剂合物或结晶和药效可接受的载体的组合物以及包含本发明的通式I的化合物或其立体异构体、药学可接受的盐、水合物、溶剂合物或结晶和另一种抗病毒药的组合物。A further object of the present invention is to provide a composition comprising a compound of general formula I or a stereoisomer, a pharmaceutically acceptable salt, hydrate, solvate or crystalline and a pharmaceutically acceptable carrier of the present invention and a composition comprising The composition of the compound of general formula I of the present invention or its stereoisomer, pharmaceutically acceptable salt, hydrate, solvate or crystal and another antiviral drug.
本发明的还一个目的是提供本发明的通式I的化合物或其立体异构体、药学可接受的盐、水合物、溶剂合物或结晶治疗和/或预防丙型肝炎病毒感染的方法以及本发明的通式I的化合物或其立体异构体、药学可接受的盐、水合物、溶剂合物或结晶在制备用于治疗和/或预防病毒感染的药物中的应用。Still another object of the present invention is to provide a compound of general formula I or a stereoisomer, a pharmaceutically acceptable salt, hydrate, solvate or crystal of the present invention for the treatment and/or prevention of hepatitis C virus infection and Use of the compound of general formula I or its stereoisomer, pharmaceutically acceptable salt, hydrate, solvate or crystal of the present invention in the preparation of a medicament for the treatment and/or prevention of viral infection.
针对上述发明目的,本发明提供以下技术方案:For the above-mentioned purpose of the invention, the present invention provides the following technical solutions:
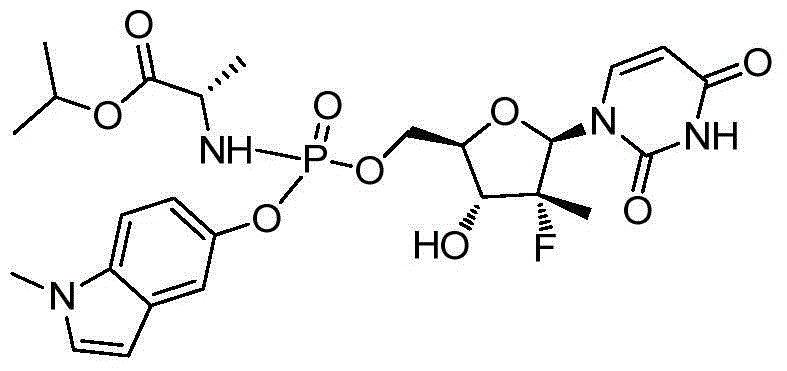
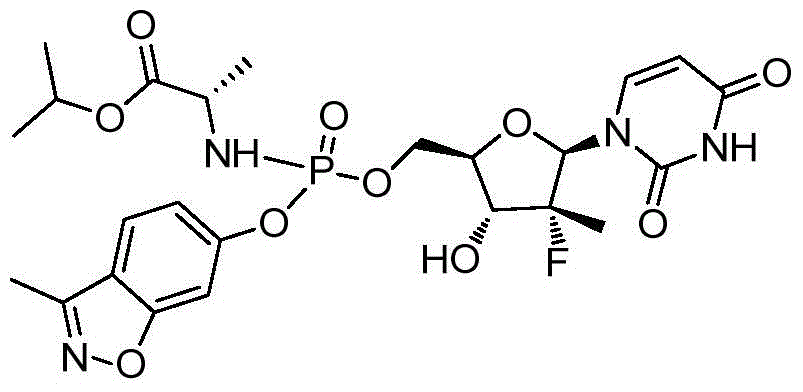
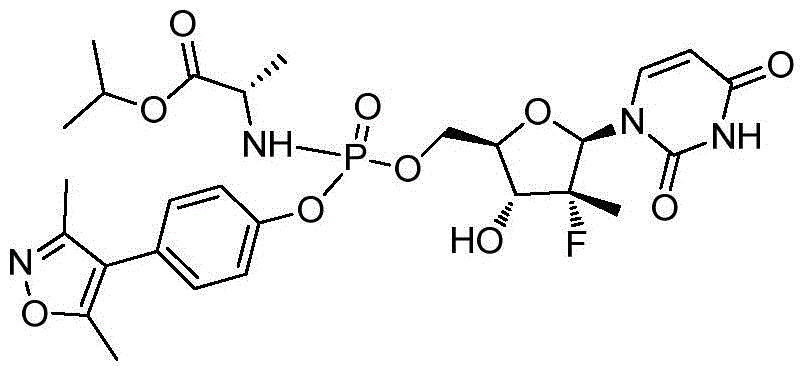
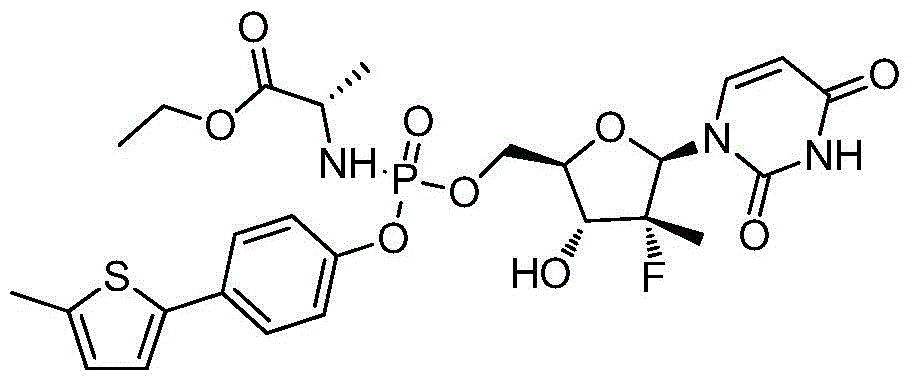
第一方面,本发明提供通式I所示的化合物或其立体异构体、药学可接受的盐、水合物、溶剂合物或结晶:In the first aspect, the present invention provides the compound represented by the general formula I or its stereoisomer, pharmaceutically acceptable salt, hydrate, solvate or crystal:
其中:in:
R1选自H、烷基和卤代烷基;R 1 is selected from H, alkyl and haloalkyl;
R2选自H和卤素;R is selected from H and halogen;
R3选自H、OH和烷氧基; R is selected from H, OH and alkoxy;
R4选自H和烷基,其中所述烷基任选被选自烷基、环烷基、杂环烷基、烷氧基、烷基氨基、卤素、羟基、氨基、硝基、氰基、烷基酰基、氨基酰基、烷氨基酰基、磺酰基、亚磺酰基、巯基、芳基和杂芳基中的一个或多个基团取代; R4 is selected from H and alkyl, wherein said alkyl is optionally selected from alkyl, cycloalkyl, heterocycloalkyl, alkoxy, alkylamino, halo, hydroxy, amino, nitro, cyano , alkyl acyl, amino acyl, alkylamino acyl, sulfonyl, sulfinyl, mercapto, aryl and heteroaryl group substituted with one or more groups;
R5选自H、烷基、环烷基、芳基和杂芳基,其中所述烷基、环烷基、芳基和杂芳基任选被选自烷基、环烷基、杂环烷基、烷氧基、烷基氨基、卤素、羟基、氨基、硝基、氰基、烷基酰基、氨基酰基、烷氨基酰基、磺酰基、亚磺酰基、巯基、芳基和杂芳基中的一个或多个基团取代;R is selected from H, alkyl, cycloalkyl, aryl and heteroaryl, wherein said alkyl, cycloalkyl, aryl and heteroaryl are optionally selected from alkyl, cycloalkyl, heterocycle Among alkyl, alkoxy, alkylamino, halogen, hydroxyl, amino, nitro, cyano, alkylacyl, aminoacyl, alkylaminoacyl, sulfonyl, sulfinyl, mercapto, aryl and heteroaryl substituted with one or more groups;
Cy1选自芳基和杂芳基,其中所述的芳基和杂芳基任选被选自卤素、羟基、烷基、环烷基、卤代烷基、烷氧基、卤代烷氧基、氨基、烷基氨基、烷基酰基氨基、烷基酰基、氨基酰基、烷基氨基酰基、硝基、氰基中的一个或多个基团取代;Cy 1 is selected from aryl and heteroaryl, wherein said aryl and heteroaryl are optionally selected from halogen, hydroxyl, alkyl, cycloalkyl, haloalkyl, alkoxy, haloalkoxy, amino, Substitution of one or more groups of alkylamino, alkylacylamino, alkylacyl, aminoacyl, alkylaminoacyl, nitro, and cyano;
Cy2选自氢、芳基、杂芳基、杂环烷基和杂环烯基,其中所述的芳基、杂芳基、杂环烷基和杂环烯基任选被选自卤素、羟基、烷基、烯基、炔基、环烷基、卤代烷基、烷氧基、卤代烷氧基、氨基、烷基氨基、烷基酰基氨基、烷基酰基、氨基酰基、烷基氨基酰基、硝基、氰基中的一个或多个基团取代;Cy 2 is selected from hydrogen, aryl, heteroaryl, heterocycloalkyl and heterocycloalkenyl, wherein said aryl, heteroaryl, heterocycloalkyl and heterocycloalkenyl are optionally selected from halogen, Hydroxy, alkyl, alkenyl, alkynyl, cycloalkyl, haloalkyl, alkoxy, haloalkoxy, amino, alkylamino, alkylacylamino, alkylacyl, aminoacyl, alkylaminoacyl, nitro One or more groups in cyano group and cyano group are substituted;
条件是Cy1为芳基时,Cy2不为氢或芳基,且本发明通式I的化合物不是以下化合物:Provided that Cy 1 is an aryl group, Cy 2 is not hydrogen or aryl, and the compound of general formula I of the present invention is not the following compounds:
(2S)-2-(((4-(1,2,3-噻二唑-4-基)苯-1-基)氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯;(2S)-2-(((4-(1,2,3-thiadiazol-4-yl)phenyl-1-yl)oxy)(((2R,3R,4R,5R)-5-( 2,4-Dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino ) isopropyl propionate;
(2S)-2-(((2-(喹喔啉-5-基)苯-1-基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯;(2S)-2-(((2-(quinoxalin-5-yl)phenyl-1-yloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo -3,4-Dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate isopropyl;
(2S)-2-(((4-(噻唑-2-基)苯-1-基)氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯;(2S)-2-(((4-(thiazol-2-yl)phenyl-1-yl)oxy)(((2R,3R,4R,5R)-5-(2,4-dioxo- Isopropyl 3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate;
(2S)-2-(((4-(噻唑-5-基)苯-1-基)氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯;(2S)-2-(((4-(thiazol-5-yl)phenyl-1-yl)oxy)(((2R,3R,4R,5R)-5-(2,4-dioxo- Isopropyl 3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate;
(2S)-2-(((1H-吲哚-5-基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯;(2S)-2-(((1H-Indol-5-yloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo-3,4-dihydropyrimidine Isopropyl 1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate;
(2S)-2-(((4-氟-2-甲基-1H-吲哚-5-基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯;(2S)-2-(((4-Fluoro-2-methyl-1H-indol-5-yloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo Isopropyl substituted-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate ;
(2S)-2-(((3-甲氧羰基甲基-苯并呋喃-7-基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯;(2S)-2-(((3-methoxycarbonylmethyl-benzofuran-7-yloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo- Isopropyl 3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate;
(2S)-2-(((4-氧代-2-苯基-4H-苯并吡喃-6-基)氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯;(2S)-2-(((4-oxo-2-phenyl-4H-benzopyran-6-yl)oxy)(((2R,3R,4R,5R)-5-(2, 4-Dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propane isopropyl acid;
(2S)-2-(((喹喔啉-5-基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯;(2S)-2-(((quinoxalin-5-yloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo-3,4-dihydropyrimidine- Isopropyl 1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate;
(2S)-2-(((4-氧代-2-苯基苯并二氢吡喃-7-基)氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯;(2S)-2-(((4-oxo-2-phenylchroman-7-yl)oxy)(((2R,3R,4R,5R)-5-(2,4 -Dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionic acid isopropyl ester;
(2S)-2-((((4-(1H-1,2,4-三唑-1-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯;(2S)-2-((((4-(1H-1,2,4-triazol-1-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2, 4-Dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propane isopropyl acid;
(2S)-2-(((4-(嘧啶-2-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯;(2S)-2-(((4-(pyrimidin-2-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo-3,4- Dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate isopropyl;
(2S)-2-(((S)-((4-氟-1,2-二甲基-1H-吲哚-5-基)氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯;(2S)-2-(((S)-((4-Fluoro-1,2-dimethyl-1H-indol-5-yl)oxy)(((2R,3R,4R,5R)- 5-(2,4-Dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphorus Acyl)amino)isopropyl propionate;
(2S)-2-((((6-氟-3-甲基苯并[d]异噁唑-5-基)氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯;(2S)-2-((((6-Fluoro-3-methylbenzo[d]isoxazol-5-yl)oxy)(((2R,3R,4R,5R)-5-(2 ,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino) isopropyl propionate;
(2S)-2-((((6-氟苯并[c][1,2,5]噻二唑-5-基)氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯;和(2S)-2-((((6-Fluorobenzo[c][1,2,5]thiadiazol-5-yl)oxy)(((2R,3R,4R,5R)-5- (2,4-Dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl) amino) isopropyl propionate; and
(2S)-2-(((4-(噻唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯。(2S)-2-(((4-(thiazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo-3,4- Dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionic acid isopropyl ester.
在一些优选的实施方案中,R1选自H、C1-6烷基、卤代C1-6烷基,R2选自H、氟、氯、溴,R3选自H、OH。In some preferred embodiments, R 1 is selected from H, C 1-6 alkyl, haloC 1-6 alkyl, R 2 is selected from H, fluorine, chlorine, bromine, and R 3 is selected from H, OH.
在另一些优选的实施方案中,R1选自H、C1-3烷基,R2选自氟、氯,R3选自H、OH。In other preferred embodiments, R 1 is selected from H, C 1-3 alkyl, R 2 is selected from fluorine, chlorine, and R 3 is selected from H, OH.
在一些优选的实施方案中,R4选自H、C1-6烷基,R5选自C1-6烷基和C3-8环烷基,其中所述的烷基和环烷基任选被选自烷基、环烷基、杂环烷基、烷氧基、烷基氨基、卤素、羟基、氨基、硝基、氰基、烷基酰基、氨基酰基、烷氨基酰基、磺酰基、亚磺酰基、巯基、芳基和杂芳基中的一个或多个基团取代。In some preferred embodiments, R 4 is selected from H, C 1-6 alkyl, and R 5 is selected from C 1-6 alkyl and C 3-8 cycloalkyl, wherein said alkyl and cycloalkyl optionally selected from alkyl, cycloalkyl, heterocycloalkyl, alkoxy, alkylamino, halo, hydroxy, amino, nitro, cyano, alkylacyl, aminoacyl, alkylaminoacyl, sulfonyl , sulfinyl, mercapto, aryl and heteroaryl are substituted with one or more groups.
在另一些优选的实施方案中,R4选自H、C1-3烷基,R5选自C1-6烷基和C3-6环烷基,其中所述的烷基和环烷基可以被选自C1-6烷基、C3-8环烷基、C3-8杂环烷基、C1-6烷氧基、C1-6烷基氨基、卤素、羟基、氨基、硝基、氰基、C1-6烷基酰基、氨基酰基、C1-6烷氨基酰基、磺酰基、亚磺酰基、巯基、芳基和杂芳基中的一个或多个基团取代。In other preferred embodiments, R 4 is selected from H, C 1-3 alkyl, R 5 is selected from C 1-6 alkyl and C 3-6 cycloalkyl, wherein said alkyl and cycloalkane The group can be selected from C 1-6 alkyl, C 3-8 cycloalkyl, C 3-8 heterocycloalkyl, C 1-6 alkoxy, C 1-6 alkylamino, halogen, hydroxyl, amino , nitro, cyano, C 1-6 alkyl acyl, aminoacyl, C 1-6 alkylamino acyl, sulfonyl, sulfinyl, mercapto, aryl and heteroaryl substituted with one or more groups .
在另一些优选的实施方案中,R4选自H、C1-3烷基,R5选自C1-3烷基和C3-6环烷基,其中所述的烷基和环烷基可以被选自C1-3烷基、C3-6环烷基、C3-6杂环烷基、C1-3烷氧基、C1-3烷基氨基、卤素、羟基、氨基、硝基、氰基、C1-3烷基酰基、氨基酰基、C1-3烷氨基酰基、磺酰基、亚磺酰基、巯基、苯基和杂芳基中的一个或多个基团取代。In other preferred embodiments, R 4 is selected from H, C 1-3 alkyl, R 5 is selected from C 1-3 alkyl and C 3-6 cycloalkyl, wherein said alkyl and cycloalkane The group can be selected from C 1-3 alkyl, C 3-6 cycloalkyl, C 3-6 heterocycloalkyl, C 1-3 alkoxy, C 1-3 alkylamino, halogen, hydroxyl, amino , nitro, cyano, C 1-3 alkyl acyl, aminoacyl, C 1-3 alkylamino acyl, sulfonyl, sulfinyl, mercapto, phenyl and heteroaryl substituted with one or more groups .
在一些优选的实施方案中,Cy1选自苯环,Cy2选自杂芳基、杂环烷基和杂环烯基,其中所述的苯环、杂芳基、杂环烷基和杂环烯基任选被选自卤素、羟基、C1-6烷基、C2-4烯基、C2-4炔基、C3-6环烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、氨基、C1-6烷基氨基、C1-6烷基酰基氨基、C1-6烷基酰基、氨基酰基、C1-6烷基氨基酰基、硝基、氰基中的一个或多个基团取代。In some preferred embodiments, Cy 1 is selected from benzene ring, Cy 2 is selected from heteroaryl, heterocycloalkyl and heterocycloalkenyl, wherein said benzene ring, heteroaryl, heterocycloalkyl and heterocycle Cycloalkenyl is optionally selected from halogen, hydroxy, C 1-6 alkyl, C 2-4 alkenyl, C 2-4 alkynyl, C 3-6 cycloalkyl, halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 alkoxy, amino, C 1-6 alkylamino, C 1-6 alkyl acylamino, C 1-6 alkyl acyl, aminoacyl, C 1 -6 Substituted with one or more groups of alkylaminoacyl, nitro, and cyano.
在另一些优选的实施方案中,Cy1选自苯环,Cy2选自杂芳基、C3-8杂环烷基和C3-8杂环烯基,其中所述的苯环、杂芳基、杂环烷基和杂环烯基任选被选自卤素、羟基、C1-3烷基、C2-4烯基、C2-4炔基、C3-6环烷基、卤代C1-3烷基、C1-3烷氧基、卤代C1-3烷氧基、氨基、C1-3烷基氨基、C1-3烷基酰基氨基、C1-3烷基酰基、氨基酰基、C1-3烷基氨基酰基、硝基、氰基中的一个或多个基团取代。In other preferred embodiments, Cy 1 is selected from benzene ring, Cy 2 is selected from heteroaryl, C 3-8 heterocycloalkyl and C 3-8 heterocycloalkenyl, wherein said benzene ring, heterocyclo Aryl, heterocycloalkyl and heterocycloalkenyl are optionally selected from halogen, hydroxy, C 1-3 alkyl, C 2-4 alkenyl, C 2-4 alkynyl, C 3-6 cycloalkyl, Halogenated C 1-3 alkyl, C 1-3 alkoxy, halogenated C 1-3 alkoxy, amino, C 1-3 alkylamino, C 1-3 alkylacylamino, C 1-3 Substituted with one or more groups of alkylacyl, aminoacyl, C1-3 alkylaminoacyl, nitro, and cyano.
在另一些优选的实施方案中,Cy1选自苯环,Cy2选自杂芳基、C3-6杂环烷基和C3-6杂环烯基,其中所述的苯环、杂芳基、杂环烷基和杂环烯基任选被选自卤素、羟基、C1-3烷基、C2-4烯基、C2-4炔基、C3-6环烷基、卤代C1-3烷基、C1-3烷氧基、卤代C1-3烷氧基、氨基、C1-3烷基氨基、C1-3烷基酰基氨基、C1-3烷基酰基、氨基酰基、C1-3烷基氨基酰基、硝基、氰基中的一个或多个基团取代。In other preferred embodiments, Cy 1 is selected from benzene ring, Cy 2 is selected from heteroaryl, C 3-6 heterocycloalkyl and C 3-6 heterocycloalkenyl, wherein said benzene ring, heterocyclo Aryl, heterocycloalkyl and heterocycloalkenyl are optionally selected from halogen, hydroxy, C 1-3 alkyl, C 2-4 alkenyl, C 2-4 alkynyl, C 3-6 cycloalkyl, Halogenated C 1-3 alkyl, C 1-3 alkoxy, halogenated C 1-3 alkoxy, amino, C 1-3 alkylamino, C 1-3 alkylacylamino, C 1-3 Substituted with one or more groups of alkylacyl, aminoacyl, C1-3 alkylaminoacyl, nitro, and cyano.
在另一些优选的实施方案中,Cy1选自苯环,Cy2选自至少含有一个N原子的五元或六元杂芳基、杂环烷基和杂环烯基,其中所述的苯环、五元或六元杂芳基、杂环烷基或杂环烯基任选被选自卤素、羟基、C1-6烷基、C2-4烯基、C2-4炔基、C3-6环烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、氨基、C1-6烷基氨基、C1-6烷基酰基氨基、C1-6烷基酰基、氨基酰基、C1-6烷基氨基酰基、硝基、氰基中的一个或多个基团取代。In other preferred embodiments, Cy 1 is selected from benzene ring, Cy 2 is selected from five- or six-membered heteroaryl, heterocycloalkyl and heterocycloalkenyl containing at least one N atom, wherein the benzene ring, five- or six-membered heteroaryl, heterocycloalkyl or heterocycloalkenyl optionally selected from halogen, hydroxy, C1-6 alkyl, C2-4 alkenyl, C2-4 alkynyl, C 3-6 cycloalkyl, halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 alkoxy, amino, C 1-6 alkylamino, C 1-6 alkane One or more groups of acylamino, C 1-6 alkyl acyl, aminoacyl, C 1-6 alkyl aminoacyl, nitro, and cyano are substituted.
在另一些优选的实施方案中,Cy1选自苯环,Cy2选自含有1-3个N原子的五元或六元杂芳基、杂环烷基和杂环烯基,其中所述的苯环、五元或六元杂芳基、杂环烷基或杂环烯基任选被选自卤素、羟基、C1-6烷基、C2-4烯基、C2-4炔基、C3-6环烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、氨基、C1-6烷基氨基、C1-6烷基酰基氨基、C1-6烷基酰基、氨基酰基、C1-6烷基氨基酰基、硝基、氰基中的一个或多个基团取代。In other preferred embodiments, Cy 1 is selected from benzene ring, Cy 2 is selected from five- or six-membered heteroaryl, heterocycloalkyl and heterocycloalkenyl containing 1-3 N atoms, wherein the The benzene ring, five- or six-membered heteroaryl, heterocycloalkyl or heterocycloalkenyl are optionally selected from halogen, hydroxyl, C1-6 alkyl, C2-4 alkenyl, C2-4 alkyne base, C 3-6 cycloalkyl, halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 alkoxy, amino, C 1-6 alkylamino, C 1- Substituted with one or more groups of 6 alkylacylamino, C1-6 alkylacyl, aminoacyl, C1-6 alkylaminoacyl, nitro, and cyano.
在另一些优选的实施方案中,Cy1选自苯环,Cy2选自仅含有1个S原子的五元或六元杂芳基、杂环烷基和杂环烯基,其中所述的苯环、五元或六元杂芳基、杂环烷基或杂环烯基任选被选自卤素、羟基、C1-6烷基、C2-4烯基、C2-4炔基、C3-6环烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、氨基、C1-6烷基氨基、C1-6烷基酰基氨基、C1-6烷基酰基、氨基酰基、C1-6烷基氨基酰基、硝基、氰基中的一个或多个基团取代。In other preferred embodiments, Cy 1 is selected from benzene ring, Cy 2 is selected from five- or six-membered heteroaryl, heterocycloalkyl and heterocycloalkenyl containing only 1 S atom, wherein said Benzene ring, five- or six-membered heteroaryl, heterocycloalkyl or heterocycloalkenyl optionally selected from halogen, hydroxy, C1-6alkyl , C2-4alkenyl , C2-4alkynyl , C 3-6 cycloalkyl, halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 alkoxy, amino, C 1-6 alkylamino, C 1-6 One or more groups of alkylacylamino, C1-6 alkylacyl, aminoacyl, C1-6 alkylaminoacyl, nitro, and cyano are substituted.
在一些具体的实施方案中,Cy1选自苯环,Cy2选自含有1个N原子的六元杂芳基、杂环烷基和杂环烯基,其中所述的苯环、杂芳基、杂环烷基或杂环烯基任选被选自卤素、羟基、C1-6烷基、C2-4烯基、C2-4炔基、C3-6环烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、氨基、C1-6烷基氨基、C1-6烷基酰基氨基、C1-6烷基酰基、氨基酰基、C1-6烷基氨基酰基、硝基、氰基中的一个或多个基团取代。In some specific embodiments, Cy 1 is selected from benzene ring, Cy 2 is selected from six-membered heteroaryl, heterocycloalkyl and heterocycloalkenyl containing 1 N atom, wherein said benzene ring, heteroaryl group, heterocycloalkyl or heterocycloalkenyl optionally selected from halogen, hydroxy, C 1-6 alkyl, C 2-4 alkenyl, C 2-4 alkynyl, C 3-6 cycloalkyl, halogen Substituted C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 alkoxy, amino, C 1-6 alkylamino, C 1-6 alkyl acylamino, C 1-6 alkane One or more groups of acyl, aminoacyl, C 1-6 alkylaminoacyl, nitro, and cyano are substituted.
在另一些具体的实施方案中,Cy1选自苯环,Cy2选自仅含有1个S原子的五元杂芳基、杂环烷基和杂环烯基,其中所述的苯环、杂芳基、杂环烷基或杂环烯基任选被选自卤素、羟基、C1-6烷基、C2-4烯基、C2-4炔基、C3-6环烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、氨基、C1-6烷基氨基、C1-6烷基酰基氨基、C1-6烷基酰基、氨基酰基、C1-6烷基氨基酰基、硝基、氰基中的一个或多个基团取代。In other specific embodiments, Cy 1 is selected from benzene ring, Cy 2 is selected from five-membered heteroaryl, heterocycloalkyl and heterocycloalkenyl containing only 1 S atom, wherein the benzene ring, Heteroaryl, heterocycloalkyl or heterocycloalkenyl is optionally selected from halogen, hydroxy, C1-6 alkyl, C2-4 alkenyl, C2-4 alkynyl, C3-6 cycloalkyl , halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 alkoxy, amino, C 1-6 alkylamino, C 1-6 alkyl acylamino, C 1- Substituted with one or more groups of 6 alkyl acyl, amino acyl, C 1-6 alkyl amino acyl, nitro, and cyano.
在另一些具体的实施方案中,Cy1选自苯环,Cy2选自含有2个N原子的五元杂芳基、杂环烷基和杂环烯基,其中所述的苯环、杂芳基、杂环烷基或杂环烯基任选被选自卤素、羟基、C1-6烷基、C2-4烯基、C2-4炔基、C3-6环烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、氨基、C1-6烷基氨基、C1-6烷基酰基氨基、C1-6烷基酰基、氨基酰基、C1-6烷基氨基酰基、硝基、氰基中的一个或多个基团取代。In some other specific embodiments, Cy 1 is selected from benzene ring, Cy 2 is selected from five-membered heteroaryl, heterocycloalkyl and heterocycloalkenyl containing 2 N atoms, wherein said benzene ring, heterocyclic Aryl, heterocycloalkyl or heterocycloalkenyl are optionally selected from halogen, hydroxy, C1-6 alkyl, C2-4 alkenyl, C2-4 alkynyl, C3-6 cycloalkyl, Halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 alkoxy, amino, C 1-6 alkylamino, C 1-6 alkylacylamino, C 1-6 Substituted with one or more groups of alkylacyl, aminoacyl, C 1-6 alkylaminoacyl, nitro, and cyano.
在另一些具体的实施方案中,Cy1选自苯环,Cy2选自含有1个N原子和1个硫原子的五元杂芳基、杂环烷基和杂环烯基,其中所述的苯环、杂芳基、杂环烷基或杂环烯基任选被选自卤素、羟基、C1-6烷基、C2-4烯基、C2-4炔基、C3-6环烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、氨基、C1-6烷基氨基、C1-6烷基酰基氨基、C1-6烷基酰基、氨基酰基、C1-6烷基氨基酰基、硝基、氰基中的一个或多个基团取代。In other specific embodiments, Cy 1 is selected from benzene ring, Cy 2 is selected from five-membered heteroaryl, heterocycloalkyl and heterocycloalkenyl containing 1 N atom and 1 sulfur atom, wherein the The benzene ring, heteroaryl, heterocycloalkyl or heterocycloalkenyl is optionally selected from halogen, hydroxyl, C 1-6 alkyl, C 2-4 alkenyl, C 2-4 alkynyl, C 3- 6 cycloalkyl, halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 alkoxy, amino, C 1-6 alkylamino, C 1-6 alkylacylamino , C 1-6 alkyl acyl group, amino acyl group, C 1-6 alkyl amino acyl group, nitro group, cyano group is substituted with one or more groups.
在另一些具体的实施方案中,Cy1选自苯环,Cy2选自含有1个N原子和1个氧原子的五元杂芳基、杂环烷基和杂环烯基,其中所述的苯环、杂芳基、杂环烷基或杂环烯基任选被选自卤素、羟基、C1-6烷基、C2-4烯基、C2-4炔基、C3-6环烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、氨基、C1-6烷基氨基、C1-6烷基酰基氨基、C1-6烷基酰基、氨基酰基、C1-6烷基氨基酰基、硝基、氰基中的一个或多个基团取代。In other specific embodiments, Cy 1 is selected from benzene ring, Cy 2 is selected from five-membered heteroaryl, heterocycloalkyl and heterocycloalkenyl containing 1 N atom and 1 oxygen atom, wherein the The benzene ring, heteroaryl, heterocycloalkyl or heterocycloalkenyl is optionally selected from halogen, hydroxyl, C 1-6 alkyl, C 2-4 alkenyl, C 2-4 alkynyl, C 3- 6 cycloalkyl, halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 alkoxy, amino, C 1-6 alkylamino, C 1-6 alkylacylamino , C 1-6 alkyl acyl group, amino acyl group, C 1-6 alkyl amino acyl group, nitro group, cyano group is substituted with one or more groups.
在另一些优选的实施方案中,Cy1选自苯环,Cy2选自呋喃环、吡咯环、噻吩环、咪唑环、吡唑环、噻唑环、异噻唑环、噁唑环、异噁唑环、三氮唑环、噻二唑环、噁二唑环、吡啶环、嘧啶环、吡嗪环、哒嗪环、苯并氮杂环、苯并氧杂环、苯并硫杂环、苯并二氮杂环、苯并二氧杂环、苯并二硫杂环、苯并氧氮杂环、苯并硫氮杂环,其中所述的呋喃环、吡咯环、噻吩环、咪唑环、吡唑环、噻唑环、异噻唑环、噁唑环、异噁唑环、三氮唑环、噻二唑环、噁二唑环、吡啶环、嘧啶环、吡嗪环、哒嗪环、苯并氮杂环、苯并氧杂环、苯并硫杂环、苯并二氮杂环、苯并二氧杂环、苯并二硫杂环、苯并氧氮杂环、苯并硫氮杂环任选被选自卤素、羟基、C1-6烷基、C2-4烯基、C2-4炔基、C3-6环烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、氨基、C1-6烷基氨基、C1-6烷基酰基氨基、C1-6烷基酰基、氨基酰基、C1-6烷基氨基酰基、硝基、氰基中的一个或多个基团取代。In other preferred embodiments, Cy 1 is selected from benzene ring, Cy 2 is selected from furan ring, pyrrole ring, thiophene ring, imidazole ring, pyrazole ring, thiazole ring, isothiazole ring, oxazole ring, isoxazole Ring, triazole ring, thiadiazole ring, oxadiazole ring, pyridine ring, pyrimidine ring, pyrazine ring, pyridazine ring, benzoazepine ring, benzoxane ring, benzothiane ring, benzene Diazepine ring, benzodioxane ring, benzodithiane ring, benzoxazepine ring, benzothiazepine ring, wherein said furan ring, pyrrole ring, thiophene ring, imidazole ring, Pyrazole ring, thiazole ring, isothiazole ring, oxazole ring, isoxazole ring, triazole ring, thiadiazole ring, oxadiazole ring, pyridine ring, pyrimidine ring, pyrazine ring, pyridazine ring, benzene Benzodiazepine, benzoxazepine, benzothiazepine, benzodiazepine, benzodiazepine, benzodiazepine, benzoxazepine, benzothiazepine Ring is optionally selected from halogen, hydroxy, C1-6 alkyl, C2-4 alkenyl, C2-4 alkynyl, C3-6 cycloalkyl, halogenated C1-6 alkyl, C1 -6 alkoxy, halogenated C 1-6 alkoxy, amino, C 1-6 alkylamino, C 1-6 alkyl acylamino, C 1-6 alkyl acyl, aminoacyl, C 1-6 One or more groups of alkylaminoacyl, nitro, and cyano are substituted.
在另一些优选的实施方案中,Cy1选自苯环,Cy2选自呋喃环、吡咯环、噻吩环、咪唑环、吡唑环、噻唑环、异噻唑环、噁唑环、异噁唑环、三氮唑环、噻二唑环、噁二唑环、吡啶环、嘧啶环、吡嗪环、哒嗪环、苯并氮杂环、苯并氧杂环、苯并硫杂环、苯并二氮杂环、苯并二氧杂环、苯并二硫杂环、苯并氧氮杂环、苯并硫氮杂环,其中所述的呋喃环、吡咯环、噻吩环、咪唑环、吡唑环、噻唑环、异噻唑环、噁唑环、异噁唑环、三氮唑环、噻二唑环、噁二唑环、吡啶环、嘧啶环、吡嗪环、哒嗪环、苯并氮杂环、苯并氧杂环、苯并硫杂环、苯并二氮杂环、苯并二氧杂环、苯并二硫杂环、苯并氧氮杂环、苯并硫氮杂环任选被选自卤素、羟基、C1-6烷基、C2-4烯基、C2-4炔基、C3-6环烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、氨基、C1-6烷基氨基、C1-6烷基酰基氨基、C1-6烷基酰基、氨基酰基、C1-6烷基氨基酰基、硝基、氰基中的一个或多个基团取代,且Cy2在Cy1的对位或间位。In other preferred embodiments, Cy 1 is selected from benzene ring, Cy 2 is selected from furan ring, pyrrole ring, thiophene ring, imidazole ring, pyrazole ring, thiazole ring, isothiazole ring, oxazole ring, isoxazole Ring, triazole ring, thiadiazole ring, oxadiazole ring, pyridine ring, pyrimidine ring, pyrazine ring, pyridazine ring, benzoazepine ring, benzoxane ring, benzothiane ring, benzene Diazepine ring, benzodioxane ring, benzodithiane ring, benzoxazepine ring, benzothiazepine ring, wherein said furan ring, pyrrole ring, thiophene ring, imidazole ring, Pyrazole ring, thiazole ring, isothiazole ring, oxazole ring, isoxazole ring, triazole ring, thiadiazole ring, oxadiazole ring, pyridine ring, pyrimidine ring, pyrazine ring, pyridazine ring, benzene Benzodiazepine, benzoxazepine, benzothiazepine, benzodiazepine, benzodiazepine, benzodiazepine, benzoxazepine, benzothiazepine Ring is optionally selected from halogen, hydroxy, C1-6 alkyl, C2-4 alkenyl, C2-4 alkynyl, C3-6 cycloalkyl, halogenated C1-6 alkyl, C1 -6 alkoxy, halogenated C 1-6 alkoxy, amino, C 1-6 alkylamino, C 1-6 alkyl acylamino, C 1-6 alkyl acyl, aminoacyl, C 1-6 One or more groups of alkylaminoacyl, nitro, and cyano are substituted, and Cy 2 is at the para or meta position of Cy 1 .
在一些优选的实施方案中,根据本发明的通式I的化合物或其立体异构体、药学可接受的盐、水合物、溶剂合物或结晶,其中Cy2在Cy1的对位或间位。本发明的发明人意料不到地发现,当通式I中的Cy2在Cy1的对位或间位,本发明的化合物具有非常优异的抗病毒活性。In some preferred embodiments, the compound of general formula I according to the present invention, or a stereoisomer, pharmaceutically acceptable salt, hydrate, solvate or crystal thereof, wherein Cy 2 is in the para or meta position of Cy 1 bit. The inventors of the present invention unexpectedly found that when Cy 2 in the general formula I is in the para or meta position of Cy 1 , the compounds of the present invention have very excellent antiviral activity.
在另一些优选的实施方案中,Cy1选自萘环、杂芳基,Cy2选自氢、芳基、杂芳基、杂环烷基和杂环烯基,其中所述的萘环、芳基、杂芳基、杂环烷基和杂环烯基任选被选自卤素、羟基、C1-6烷基、C2-4烯基、C2-4炔基、C3-6环烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、氨基、C1-6烷基氨基、C1-6烷基酰基氨基、C1-6烷基酰基、氨基酰基、C1-6烷基氨基酰基、硝基、氰基中的一个或多个基团取代。In other preferred embodiments, Cy 1 is selected from naphthalene ring, heteroaryl, Cy 2 is selected from hydrogen, aryl, heteroaryl, heterocycloalkyl and heterocycloalkenyl, wherein said naphthalene ring, Aryl, heteroaryl, heterocycloalkyl and heterocycloalkenyl are optionally selected from halogen, hydroxy, C 1-6 alkyl, C 2-4 alkenyl, C 2-4 alkynyl, C 3-6 Cycloalkyl, halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 alkoxy, amino, C 1-6 alkylamino, C 1-6 alkylacylamino, One or more groups of C 1-6 alkyl acyl, aminoacyl, C 1-6 alkyl aminoacyl, nitro, and cyano are substituted.
在另一些优选的实施方案中,Cy1选自萘环、杂芳基,Cy2选自氢、芳基、杂芳基、C3-8杂环烷基和C3-8杂环烯基,其中所述的萘环、芳基、杂芳基、杂环烷基和杂环烯基任选被选自卤素、羟基、C1-6烷基、C2-4烯基、C2-4炔基、C3-6环烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、氨基、C1-6烷基氨基、C1-6烷基酰基氨基、C1-6烷基酰基、氨基酰基、C1-6烷基氨基酰基、硝基、氰基中的一个或多个基团取代。在另一些优选的实施方案中,Cy1选自萘环、杂芳基,Cy2选自氢、芳基、杂芳基、C3-6杂环烷基和C3-6杂环烯基,其中所述的萘环、芳基、杂芳基、杂环烷基和杂环烯基任选被选自卤素、羟基、C1-3烷基、C2-4烯基、C2-4炔基、C3-6环烷基、卤代C1-3烷基、C1-3烷氧基、卤代C1-3烷氧基、氨基、C1-3烷基氨基、C1-3烷基酰基氨基、C1-3烷基酰基、氨基酰基、C1-3烷基氨基酰基、硝基、氰基中的一个或多个基团取代。In other preferred embodiments, Cy 1 is selected from naphthalene ring, heteroaryl, Cy 2 is selected from hydrogen, aryl, heteroaryl, C 3-8 heterocycloalkyl and C 3-8 heterocycloalkenyl , wherein the naphthalene ring, aryl, heteroaryl, heterocycloalkyl and heterocycloalkenyl are optionally selected from halogen, hydroxyl, C 1-6 alkyl, C 2-4 alkenyl, C 2- 4alkynyl , C 3-6 cycloalkyl, halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 alkoxy, amino, C 1-6 alkylamino, C Substituted with one or more groups of 1-6 alkylacylamino, C1-6 alkylacyl, aminoacyl, C1-6 alkylaminoacyl, nitro, and cyano. In other preferred embodiments, Cy 1 is selected from naphthalene ring, heteroaryl, Cy 2 is selected from hydrogen, aryl, heteroaryl, C 3-6 heterocycloalkyl and C 3-6 heterocycloalkenyl , wherein the naphthalene ring, aryl, heteroaryl, heterocycloalkyl and heterocycloalkenyl are optionally selected from halogen, hydroxyl, C 1-3 alkyl, C 2-4 alkenyl, C 2- 4alkynyl , C 3-6 cycloalkyl, halogenated C 1-3 alkyl, C 1-3 alkoxy, halogenated C 1-3 alkoxy, amino, C 1-3 alkylamino, C Substituted with one or more groups of 1-3 alkylacylamino, C1-3 alkylacyl, aminoacyl, C1-3 alkylaminoacyl, nitro, and cyano.
在另一些优选的实施方案中,Cy1选自苯并至少含有一个N原子的五元或六元杂环,其中所述苯并至少含有一个N原子的五元或六元杂环任选被选自卤素、羟基、C1-6烷基、C2-4烯基、C2-4炔基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、氨基、C1-6烷基氨基、C1-6烷基酰基氨基、C1-6烷基酰基、氨基酰基、C1-6烷基氨基酰基、硝基、氰基中的一个或多个基团取代,且Cy2为氢。在另一些优选的实施方案中,Cy1选自苯并仅含有一个S原子的五元或六元杂环,其中所述苯并仅含有一个S原子的五元或六元杂环任选被选自卤素、羟基、C1-6烷基、C2-4烯基、C2-4炔基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、氨基、C1-6烷基氨基、C1-6烷基酰基氨基、C1-6烷基酰基、氨基酰基、C1-6烷基氨基酰基、硝基、氰基中的一个或多个基团取代,且Cy2为氢。In other preferred embodiments, Cy 1 is selected from a five- or six-membered heterocyclic ring containing at least one N atom of benzo, wherein the five- or six-membered heterocyclic ring containing at least one N atom of benzo is optionally Selected from halogen, hydroxyl, C 1-6 alkyl, C 2-4 alkenyl, C 2-4 alkynyl, halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 Of alkoxy, amino, C 1-6 alkylamino, C 1-6 alkyl acylamino, C 1-6 alkyl acyl, amino acyl, C 1-6 alkyl amino acyl, nitro, cyano One or more groups are substituted, and Cy 2 is hydrogen. In other preferred embodiments, Cy 1 is selected from a five- or six-membered heterocycle containing only one S atom, wherein the five- or six-membered heterocycle containing only one S atom is optionally Selected from halogen, hydroxyl, C 1-6 alkyl, C 2-4 alkenyl, C 2-4 alkynyl, halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 Of alkoxy, amino, C 1-6 alkylamino, C 1-6 alkyl acylamino, C 1-6 alkyl acyl, amino acyl, C 1-6 alkyl amino acyl, nitro, cyano One or more groups are substituted, and Cy 2 is hydrogen.
在一些具体的实施方案中,Cy1选自苯并五元或六元含1、2或3个氮原子的杂芳环,其中所述苯并五元或六元含1、2或3个氮原子的杂芳环任选被选自卤素、羟基、C1-6烷基、C2-4烯基、C2-4炔基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、氨基、C1-6烷基氨基、C1-6烷基酰基氨基、C1-6烷基酰基、氨基酰基、C1-6烷基氨基酰基、硝基、氰基中的一个或多个基团取代,且Cy2为氢。In some specific embodiments, Cy 1 is selected from a benzo five- or six-membered heteroaromatic ring containing 1, 2, or 3 nitrogen atoms, wherein the benzo five- or six-membered contains 1, 2, or 3 The heteroaromatic ring of the nitrogen atom is optionally selected from halogen, hydroxy, C 1-6 alkyl, C 2-4 alkenyl, C 2-4 alkynyl, halogenated C 1-6 alkyl, C 1-6 alkane Oxy, halogenated C 1-6 alkoxy, amino, C 1-6 alkylamino, C 1-6 alkyl acylamino, C 1-6 alkyl acyl, aminoacyl, C 1-6 alkylamino One or more groups of acyl, nitro, and cyano are substituted, and Cy 2 is hydrogen.
在另一些具体的实施方案中,Cy1选自苯并五元含1或2个氮原子的杂芳环,其中所述苯并五元含1或2个氮原子的杂芳环任选被选自卤素、羟基、C1-6烷基、C2-4烯基、C2-4炔基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、氨基、C1-6烷基氨基、C1-6烷基酰基氨基、C1-6烷基酰基、氨基酰基、C1-6烷基氨基酰基、硝基、氰基中的一个或多个基团取代,且Cy2为氢。In other specific embodiments, Cy 1 is selected from a benzoquinta-membered heteroaromatic ring containing 1 or 2 nitrogen atoms, wherein the benzoquinta-membered heteroaromatic ring containing 1 or 2 nitrogen atoms is optionally Selected from halogen, hydroxyl, C 1-6 alkyl, C 2-4 alkenyl, C 2-4 alkynyl, halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 Of alkoxy, amino, C 1-6 alkylamino, C 1-6 alkyl acylamino, C 1-6 alkyl acyl, amino acyl, C 1-6 alkyl amino acyl, nitro, cyano One or more groups are substituted, and Cy 2 is hydrogen.
在另一些具体的实施方案中,Cy1选自苯并五元含1个氮原子和1个氧原子的杂芳环,其中所述苯并五元含1个氮原子和1个氧原子的杂芳环任选被选自卤素、羟基、C1-6烷基、C2-4烯基、C2-4炔基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、氨基、C1-6烷基氨基、C1-6烷基酰基氨基、C1-6烷基酰基、氨基酰基、C1-6烷基氨基酰基、硝基、氰基中的一个或多个基团取代,且Cy2为氢。In other specific embodiments, Cy 1 is selected from a benzoquinone-membered heteroaromatic ring containing 1 nitrogen atom and 1 oxygen atom, wherein the benzoquinone contains 1 nitrogen atom and 1 oxygen atom The heteroaromatic ring is optionally selected from halogen, hydroxy, C 1-6 alkyl, C 2-4 alkenyl, C 2-4 alkynyl, halogenated C 1-6 alkyl, C 1-6 alkoxy, Halogenated C 1-6 alkoxy, amino, C 1-6 alkylamino, C 1-6 alkyl acylamino, C 1-6 alkyl acyl, aminoacyl, C 1-6 alkyl aminoacyl, nitro One or more groups of cyano group and cyano group are substituted, and Cy 2 is hydrogen.
在另一些具体的实施方案中,Cy1选自苯并仅含1个S原子的五元杂芳环,其中所述苯并仅含1个S原子的五元杂芳环任选被选自卤素、羟基、C1-6烷基、C2-4烯基、C2-4炔基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、氨基、C1-6烷基氨基、C1-6烷基酰基氨基、C1-6烷基酰基、氨基酰基、C1-6烷基氨基酰基、硝基、氰基中的一个或多个基团取代,且Cy2为氢。In other specific embodiments, Cy 1 is selected from a five-membered heteroaromatic ring containing only one S atom, wherein the five-membered heteroaromatic ring containing only one S atom is optionally selected from Halogen, hydroxyl, C 1-6 alkyl, C 2-4 alkenyl, C 2-4 alkynyl, halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 alkoxy one of C1-6 alkylamino, C1-6 alkylamino, C1-6 alkylacylamino, C1-6 alkylacyl, aminoacyl, C1-6 alkylaminoacyl, nitro, cyano or Multiple groups are substituted, and Cy 2 is hydrogen.
在另一些优选的实施方案中,Cy1选自呋喃环、吡咯环、噻吩环、咪唑环、吡唑环、噻唑环、异噻唑环、噁唑环、异噁唑环、三氮唑环、噻二唑环、噁二唑环、吡啶环、嘧啶环、吡嗪环、哒嗪环、苯并氮杂环、苯并氧杂环、苯并硫杂环、苯并二氮杂环、苯并二氧杂环、苯并二硫杂环、苯并氧氮杂环、苯并硫氮杂环,Cy2选自氢、苯环、呋喃环、吡咯环、噻吩环、咪唑环、吡唑环、噻唑环、异噻唑环、噁唑环、异噁唑环、三氮唑环、噻二唑环、噁二唑环、吡啶环、嘧啶环、吡嗪环、哒嗪环、苯并氮杂环、苯并氧杂环、苯并硫杂环、苯并二氮杂环、苯并二氧杂环、苯并二硫杂环、苯并氧氮杂环、苯并硫氮杂环,其中所述的苯环、噻吩环、咪唑环、吡唑环、噻唑环、异噻唑环、噁唑环、异噁唑环、三氮唑环、噻二唑环、噁二唑环、吡啶环、嘧啶环、吡嗪环、哒嗪环、苯并氮杂环、苯并氧杂环、苯并硫杂环、苯并二氮杂环、苯并二氧杂环、苯并二硫杂环、苯并氧氮杂环、苯并硫氮杂环任选被选自卤素、羟基、C1-6烷基、C2-4烯基、C2-4炔基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、氨基、C1-6烷基氨基、C1-6烷基酰基氨基、C1-6烷基酰基、氨基酰基、C1-6烷基氨基酰基、硝基、氰基中的一个或多个基团取代。In other preferred embodiments, Cy 1 is selected from furan ring, pyrrole ring, thiophene ring, imidazole ring, pyrazole ring, thiazole ring, isothiazole ring, oxazole ring, isoxazole ring, triazole ring, Thiadiazole ring, oxadiazole ring, pyridine ring, pyrimidine ring, pyrazine ring, pyridazine ring, benzazepine ring, benzoxane ring, benzothiane ring, benzodiazepine ring, benzene Dioxane, benzodithia, benzoxazepine, benzothiazepine, Cy 2 is selected from hydrogen, benzene ring, furan ring, pyrrole ring, thiophene ring, imidazole ring, pyrazole Ring, thiazole ring, isothiazole ring, oxazole ring, isoxazole ring, triazole ring, thiadiazole ring, oxadiazole ring, pyridine ring, pyrimidine ring, pyrazine ring, pyridazine ring, benzazepine Heterocycles, benzoxazepines, benzothiazepines, benzodiazepines, benzodioxanes, benzodiazepines, benzoxazepines, benzothiazepines, Wherein the benzene ring, thiophene ring, imidazole ring, pyrazole ring, thiazole ring, isothiazole ring, oxazole ring, isoxazole ring, triazole ring, thiadiazole ring, oxadiazole ring, pyridine ring , pyrimidine ring, pyrazine ring, pyridazine ring, benzazepine ring, benzoxane ring, benzodiazepine ring, benzodiazepine ring, benzodioxane ring, benzodithiane ring , benzoxazepine, benzothiazepine optionally selected from halogen, hydroxy, C 1-6 alkyl, C 2-4 alkenyl, C 2-4 alkynyl, halogenated C 1-6 Alkyl, C 1-6 alkoxy, halogenated C 1-6 alkoxy, amino, C 1-6 alkylamino, C 1-6 alkyl acylamino, C 1-6 alkyl acyl, aminoacyl , C 1-6 alkylaminoacyl, nitro, cyano group is substituted with one or more groups.
在另一些优选的实施方案中,Cy1选自呋喃环、吡咯环、噻吩环、咪唑环、吡唑环、噻唑环、异噻唑环、噁唑环、异噁唑环、三氮唑环、噻二唑环、噁二唑环、吡啶环、嘧啶环、吡嗪环、哒嗪环、苯并呋喃环、苯并吡咯环、苯并噻吩环、苯并咪唑环、苯并吡唑环、苯并噻唑环、苯并异噻唑环、苯并噁唑环、苯并异噁唑环、苯并三氮唑环、苯并噻二唑环、苯并噁二唑环、苯并吡啶环、苯并嘧啶环、苯并吡嗪环、苯并哒嗪环,Cy2为氢,其中所述的Cy1任选被选自卤素、羟基、C1-6烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、氨基、C1-6烷基氨基、C1-6烷基酰基氨基、C1-6烷基酰基、氨基酰基、C1-6烷基氨基酰基、硝基、氰基中的一个或多个基团取代。In other preferred embodiments, Cy 1 is selected from furan ring, pyrrole ring, thiophene ring, imidazole ring, pyrazole ring, thiazole ring, isothiazole ring, oxazole ring, isoxazole ring, triazole ring, Thiadiazole ring, oxadiazole ring, pyridine ring, pyrimidine ring, pyrazine ring, pyridazine ring, benzofuran ring, benzopyrrole ring, benzothiophene ring, benzimidazole ring, benzopyrazole ring, benzothiazole ring, benzisothiazole ring, benzoxazole ring, benzisoxazole ring, benzotriazole ring, benzothiadiazole ring, benzoxadiazole ring, benzopyridine ring, Benzopyrimidine ring, benzopyrazine ring, benzopyridazine ring, Cy 2 is hydrogen, wherein said Cy 1 is optionally selected from halogen, hydroxyl, C 1-6 alkyl, halogenated C 1-6 Alkyl, C 1-6 alkoxy, halogenated C 1-6 alkoxy, amino, C 1-6 alkylamino, C 1-6 alkyl acylamino, C 1-6 alkyl acyl, aminoacyl , C 1-6 alkylaminoacyl, nitro, cyano group is substituted with one or more groups.
在一些优选的实施方案中,本发明提供通式Ia所示的化合物或其立体异构体、药学可接受的盐、水合物、溶剂合物或结晶:In some preferred embodiments, the present invention provides a compound of formula Ia or a stereoisomer, pharmaceutically acceptable salt, hydrate, solvate or crystal thereof:
其中:in:
Ra1选自杂芳基、杂环烷基和杂环烯基,其中所述的杂芳基、杂环烷基和杂环烯基任选被选自卤素、羟基、氨基、烷基、烯基、炔基、环烷基、卤代烷基、烷氧基、卤代烷氧基、烷氨基、烷基酰基、烷氧基酰基、氨基酰基、烷氨基酰基、磺酰基、亚磺酰基、巯基、硝基、氰基中的一个或多个基团取代;R a1 is selected from heteroaryl, heterocycloalkyl and heterocycloalkenyl, wherein said heteroaryl, heterocycloalkyl and heterocycloalkenyl are optionally selected from halogen, hydroxy, amino, alkyl, alkene alkynyl, alkynyl, cycloalkyl, haloalkyl, alkoxy, haloalkoxy, alkylamino, alkylacyl, alkoxyacyl, aminoacyl, alkylaminoacyl, sulfonyl, sulfinyl, mercapto, nitro , one or more groups in the cyano group are substituted;
Ra2选自氢、卤素、羟基、氨基、烷基、环烷基、卤代烷基、烷氧基、卤代烷氧基、烷氨基、烷基酰基、烷氧基酰基、氨基酰基、烷氨基酰基、磺酰基、亚磺酰基、巯基、硝基和氰基;或者R a2 is selected from hydrogen, halogen, hydroxyl, amino, alkyl, cycloalkyl, haloalkyl, alkoxy, haloalkoxy, alkylamino, alkylacyl, alkoxyacyl, aminoacyl, alkylaminoacyl, sulfonic acid Acyl, sulfinyl, mercapto, nitro and cyano groups; or
Ra1、Ra2与它们连接的碳原子一起构成杂芳基、杂环烷基或杂环烯基;R a1 , R a2 together with the carbon atoms to which they are attached form a heteroaryl, heterocycloalkyl or heterocycloalkenyl;
Ra3、Ra4、Ra5独立地选自氢、卤素、羟基、氨基、烷基、环烷基、卤代烷基、烷氧基、卤代烷氧基、烷氨基、烷基酰基、烷氧基酰基、氨基酰基、烷氨基酰基、磺酰基、亚磺酰基、巯基、硝基、氰基;R a3 , R a4 , R a5 are independently selected from hydrogen, halogen, hydroxy, amino, alkyl, cycloalkyl, haloalkyl, alkoxy, haloalkoxy, alkylamino, alkylacyl, alkoxyacyl, Aminoacyl, alkylaminoacyl, sulfonyl, sulfinyl, mercapto, nitro, cyano;
R1、R2、R3、R4、R5具有通式I中的定义。R 1 , R 2 , R 3 , R 4 , R 5 have the definitions in formula I.
在一些实施方案中,根据本发明的通式Ia的化合物或其立体异构体、药学可接受的盐、水合物、溶剂合物或结晶,其中R1选自H、C1-6烷基和卤代C1-6烷基,优选选自H和C1-3烷基;R2选自H、氟、氯和溴,优选选自氟和氯;R3选自H和OH;R4选自H和C1-6烷基,其中所述烷基任选被选自C1-6烷基、C3-8环烷基、C3-8杂环烷基、C1-6烷氧基、C1-6烷基氨基、卤素、羟基、氨基、硝基、氰基、C1-6烷基酰基、氨基酰基、C1-6烷氨基酰基、磺酰基、亚磺酰基、巯基、芳基和杂芳基中的一个或多个基团取代,优选地,R4选自H和C1-3烷基,其中所述烷基任选被选自C1-3烷基、C3-6环烷基、C3-6杂环烷基、C1-3烷氧基、C1-3烷基氨基、卤素、羟基、氨基、硝基、氰基、C1-3烷基酰基、氨基酰基、C1-3烷氨基酰基、磺酰基、亚磺酰基、巯基、芳基和杂芳基中的一个或多个基团取代;和R5选自H、C1-6烷基、C3-8环烷基和芳基,其中所述的烷基、环烷基和芳基任选被选自C1-6烷基、C3-8环烷基、C3-8杂环烷基、C1-6烷氧基、C1-6烷基氨基、卤素、羟基、氨基、硝基、氰基、C1-6烷基酰基、氨基酰基、C1-6烷氨基酰基、磺酰基、亚磺酰基、巯基、芳基和杂芳基中的一个或多个基团取代,优选地,R5选自H、C1-3烷基、C3-6环烷基和芳基,其中所述的烷基任选被选自C1-3烷基、C3-6环烷基、C3-6杂环烷基、C1-3烷氧基、C1-3烷基氨基、卤素、羟基、氨基、硝基、氰基、C1-3烷基酰基、氨基酰基、C1-3烷氨基酰基、磺酰基、亚磺酰基、巯基、芳基和杂芳基中的一个或多个基团取代。In some embodiments, a compound of general formula Ia or a stereoisomer, pharmaceutically acceptable salt, hydrate, solvate or crystal thereof according to the present invention, wherein R 1 is selected from H, C 1-6 alkyl and halogenated C 1-6 alkyl, preferably selected from H and C 1-3 alkyl; R 2 is selected from H, fluorine, chlorine and bromine, preferably selected from fluorine and chlorine; R 3 is selected from H and OH; R 4 is selected from H and C 1-6 alkyl, wherein said alkyl is optionally selected from C 1-6 alkyl, C 3-8 cycloalkyl, C 3-8 heterocycloalkyl, C 1-6 Alkoxy, C 1-6 alkylamino, halogen, hydroxyl, amino, nitro, cyano, C 1-6 alkyl acyl, aminoacyl, C 1-6 alkylaminoacyl, sulfonyl, sulfinyl, One or more groups in mercapto, aryl and heteroaryl are substituted, preferably, R 4 is selected from H and C 1-3 alkyl, wherein said alkyl is optionally selected from C 1-3 alkyl , C 3-6 cycloalkyl, C 3-6 heterocycloalkyl, C 1-3 alkoxy, C 1-3 alkylamino, halogen, hydroxyl, amino, nitro, cyano, C 1-3 One or more group substitutions of alkylacyl, aminoacyl, C1-3 alkylaminoacyl, sulfonyl, sulfinyl, mercapto, aryl and heteroaryl ; and R5 is selected from H, C1- 6 alkyl, C 3-8 cycloalkyl and aryl, wherein said alkyl, cycloalkyl and aryl are optionally selected from C 1-6 alkyl, C 3-8 cycloalkyl, C 3 -8 Heterocycloalkyl, C 1-6 alkoxy, C 1-6 alkylamino, halogen, hydroxyl, amino, nitro, cyano, C 1-6 alkyl acyl, aminoacyl, C 1-6 One or more groups in alkylaminoacyl, sulfonyl, sulfinyl, mercapto, aryl and heteroaryl are substituted, preferably, R 5 is selected from H, C 1-3 alkyl, C 3-6 ring Alkyl and aryl, wherein said alkyl is optionally selected from C 1-3 alkyl, C 3-6 cycloalkyl, C 3-6 heterocycloalkyl, C 1-3 alkoxy, C 1-3 alkylamino, halogen, hydroxyl, amino, nitro, cyano, C1-3 alkylacyl, aminoacyl, C1-3 alkylaminoacyl, sulfonyl, sulfinyl, mercapto, aryl and One or more groups in the heteroaryl group are substituted.
在一些优选的实施方案中,本发明提供通式Ia的化合物或其立体异构体、药学可接受的盐、水合物、溶剂合物或结晶,其中Ra1选自C4-8杂芳基、C4-8杂环烷基和C4-8杂环烯基,其中所述的杂芳基、杂环烷基和杂环烯基任选被选自卤素、羟基、氨基、C1-6烷基、C2-4烯基、C2-4炔基、C3-6环烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、C1-6烷氨基、C1-6烷基酰基、C1-6烷氧基酰基、氨基酰基、C1-6烷氨基酰基、磺酰基、亚磺酰基、巯基、硝基、氰基中的一个或多个基团取代;In some preferred embodiments, the present invention provides a compound of general formula Ia, or a stereoisomer, pharmaceutically acceptable salt, hydrate, solvate or crystal thereof, wherein R a1 is selected from C 4-8 heteroaryl , C 4-8 heterocycloalkyl and C 4-8 heterocycloalkenyl, wherein said heteroaryl, heterocycloalkyl and heterocycloalkenyl are optionally selected from halogen, hydroxy, amino, C 1- 6 alkyl, C 2-4 alkenyl, C 2-4 alkynyl, C 3-6 cycloalkyl, halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 alkane oxy, C 1-6 alkylamino, C 1-6 alkyl acyl, C 1-6 alkoxy acyl, aminoacyl, C 1-6 alkylamino acyl, sulfonyl, sulfinyl, mercapto, nitro, One or more groups in the cyano group are substituted;
Ra2选自氢、卤素、羟基、氨基、C1-6烷基、C3-6环烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、C1-6烷氨基、C1-6烷基酰基、C1-6烷氧基酰基、氨基酰基、C1-6烷氨基酰基、磺酰基、亚磺酰基、巯基、硝基和氰基;或者R a2 is selected from hydrogen, halogen, hydroxyl, amino, C 1-6 alkyl, C 3-6 cycloalkyl, halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 Alkoxy, C 1-6 alkylamino, C 1-6 alkyl acyl, C 1-6 alkoxy acyl, aminoacyl, C 1-6 alkylamino acyl, sulfonyl, sulfinyl, mercapto, nitro and cyano; or
Ra1、Ra2与它们连接的碳原子一起构成C4-8杂芳基、C4-8杂环烷基或C4-8杂环烯基。R a1 , R a2 together with the carbon atom to which they are attached constitute a C 4-8 heteroaryl, C 4-8 heterocycloalkyl or C 4-8 heterocycloalkenyl.
在另一些优选的实施方案中,本发明提供通式Ia的化合物或其立体异构体、药学可接受的盐、水合物、溶剂合物或结晶,其中Ra1选自至少含有一个N原子的五元或六元杂芳基、杂环烷基和杂环烯基,其中所述的杂芳基、杂环烷基和杂环烯基任选被选自卤素、羟基、氨基、C1-6烷基、C2-4烯基、C2-4炔基、C3-6环烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、C1-6烷氨基、C1-6烷基酰基、C1-6烷氧基酰基、氨基酰基、C1-6烷氨基酰基、磺酰基、亚磺酰基、巯基、硝基、氰基中的一个或多个基团取代;In other preferred embodiments, the present invention provides a compound of general formula Ia, or a stereoisomer, pharmaceutically acceptable salt, hydrate, solvate or crystal thereof, wherein R a1 is selected from those containing at least one N atom Five- or six-membered heteroaryl, heterocycloalkyl and heterocycloalkenyl, wherein said heteroaryl, heterocycloalkyl and heterocycloalkenyl are optionally selected from halogen, hydroxy, amino, C 1- 6 alkyl, C 2-4 alkenyl, C 2-4 alkynyl, C 3-6 cycloalkyl, halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 alkane oxy, C 1-6 alkylamino, C 1-6 alkyl acyl, C 1-6 alkoxy acyl, aminoacyl, C 1-6 alkylamino acyl, sulfonyl, sulfinyl, mercapto, nitro, One or more groups in the cyano group are substituted;
Ra2选自氢、卤素、羟基、氨基、C1-6烷基、C3-6环烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、C1-6烷氨基、C1-6烷基酰基、C1-6烷氧基酰基、氨基酰基、C1-6烷氨基酰基、磺酰基、亚磺酰基、巯基、硝基和氰基;或者R a2 is selected from hydrogen, halogen, hydroxyl, amino, C 1-6 alkyl, C 3-6 cycloalkyl, halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 Alkoxy, C 1-6 alkylamino, C 1-6 alkyl acyl, C 1-6 alkoxy acyl, aminoacyl, C 1-6 alkylamino acyl, sulfonyl, sulfinyl, mercapto, nitro and cyano; or
Ra1、Ra2与它们连接的碳原子一起构成至少含有一个N原子的五元或六元杂芳基、杂环烷基或杂环烯基。在另一些优选的实施方案中,本发明提供通式Ia的化合物或其立体异构体、药学可接受的盐、水合物、溶剂合物或结晶,其中Ra1选自含有1-3个N原子的五元或六元杂芳基、杂环烷基和杂环烯基,其中所述的杂芳基、杂环烷基和杂环烯基任选被选自卤素、羟基、氨基、C1-6烷基、C2-4烯基、C2-4炔基、C3-6环烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、C1-6烷氨基、C1-6烷基酰基、C1-6烷氧基酰基、氨基酰基、C1-6烷氨基酰基、磺酰基、亚磺酰基、巯基、硝基、氰基中的一个或多个基团取代;R a1 , R a2 together with the carbon atom to which they are attached constitute a five- or six-membered heteroaryl, heterocycloalkyl or heterocycloalkenyl group containing at least one N atom. In other preferred embodiments, the present invention provides a compound of general formula Ia, or a stereoisomer, pharmaceutically acceptable salt, hydrate, solvate or crystal thereof, wherein R a1 is selected from the group consisting of 1-3 N Atoms of five- or six-membered heteroaryl, heterocycloalkyl and heterocycloalkenyl, wherein said heteroaryl, heterocycloalkyl and heterocycloalkenyl are optionally selected from halogen, hydroxy, amino, C 1-6 alkyl, C 2-4 alkenyl, C 2-4 alkynyl, C 3-6 cycloalkyl, halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1- 6 alkoxy, C 1-6 alkylamino, C 1-6 alkyl acyl, C 1-6 alkoxy acyl, aminoacyl, C 1-6 alkylamino acyl, sulfonyl, sulfinyl, mercapto, nitro One or more groups in cyano group and cyano group are substituted;
Ra2选自氢、卤素、羟基、氨基、C1-6烷基、C3-6环烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、C1-6烷氨基、C1-6烷基酰基、C1-6烷氧基酰基、氨基酰基、C1-6烷氨基酰基、磺酰基、亚磺酰基、巯基、硝基和氰基;或者R a2 is selected from hydrogen, halogen, hydroxyl, amino, C 1-6 alkyl, C 3-6 cycloalkyl, halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 Alkoxy, C 1-6 alkylamino, C 1-6 alkyl acyl, C 1-6 alkoxy acyl, aminoacyl, C 1-6 alkylamino acyl, sulfonyl, sulfinyl, mercapto, nitro and cyano; or
Ra1、Ra2与它们连接的碳原子一起构成含有1-3个N原子的五元或六元杂芳基、杂环烷基或杂环烯基。R a1 , R a2 together with the carbon atoms to which they are attached form a five- or six-membered heteroaryl, heterocycloalkyl or heterocycloalkenyl group containing 1-3 N atoms.
在另一些优选的实施方案中,本发明提供通式Ia的化合物或其立体异构体、药学可接受的盐、水合物、溶剂合物或结晶,其中Ra1选自仅含有一个S原子的五元或六元杂芳基、杂环烷基和杂环烯基,其中所述的杂芳基、杂环烷基和杂环烯基任选被选自卤素、羟基、氨基、C1-6烷基、C2-4烯基、C2-4炔基、C3-6环烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、C1-6烷氨基、C1-6烷基酰基、C1-6烷氧基酰基、氨基酰基、C1-6烷氨基酰基、磺酰基、亚磺酰基、巯基、硝基、氰基中的一个或多个基团取代;In other preferred embodiments, the present invention provides a compound of general formula Ia, or a stereoisomer, pharmaceutically acceptable salt, hydrate, solvate or crystal thereof, wherein R a1 is selected from those containing only one S atom Five- or six-membered heteroaryl, heterocycloalkyl and heterocycloalkenyl, wherein said heteroaryl, heterocycloalkyl and heterocycloalkenyl are optionally selected from halogen, hydroxy, amino, C 1- 6 alkyl, C 2-4 alkenyl, C 2-4 alkynyl, C 3-6 cycloalkyl, halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 alkane oxy, C 1-6 alkylamino, C 1-6 alkyl acyl, C 1-6 alkoxy acyl, aminoacyl, C 1-6 alkylamino acyl, sulfonyl, sulfinyl, mercapto, nitro, One or more groups in the cyano group are substituted;
Ra2选自氢、卤素、羟基、氨基、C1-6烷基、C3-6环烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、C1-6烷氨基、C1-6烷基酰基、C1-6烷氧基酰基、氨基酰基、C1-6烷氨基酰基、磺酰基、亚磺酰基、巯基、硝基和氰基;或者R a2 is selected from hydrogen, halogen, hydroxyl, amino, C 1-6 alkyl, C 3-6 cycloalkyl, halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 Alkoxy, C 1-6 alkylamino, C 1-6 alkyl acyl, C 1-6 alkoxy acyl, aminoacyl, C 1-6 alkylamino acyl, sulfonyl, sulfinyl, mercapto, nitro and cyano; or
Ra1、Ra2与它们连接的碳原子一起构成仅含有一个S原子的五元或六元杂芳基、杂环烷基或杂环烯基。R a1 , R a2 together with the carbon atom to which they are attached form a five- or six-membered heteroaryl, heterocycloalkyl or heterocycloalkenyl group containing only one S atom.
在一些具体的实施方案中,本发明提供通式Ia的化合物或其立体异构体、药学可接受的盐、水合物、溶剂合物或结晶,其中Ra1选自含有1个N原子的六元杂芳基、杂环烷基和杂环烯基,其中所述的杂芳基、杂环烷基和杂环烯基任选被选自卤素、羟基、氨基、C1-6烷基、C2-4烯基、C2-4炔基、C3-6环烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、C1-6烷氨基、C1-6烷基酰基、C1-6烷氧基酰基、氨基酰基、C1-6烷氨基酰基、磺酰基、亚磺酰基、巯基、硝基、氰基中的一个或多个基团取代;和In some specific embodiments, the present invention provides a compound of general formula Ia, or a stereoisomer, pharmaceutically acceptable salt, hydrate, solvate or crystal thereof, wherein R a1 is selected from the group consisting of six atoms containing 1 N atom. membered heteroaryl, heterocycloalkyl and heterocycloalkenyl, wherein said heteroaryl, heterocycloalkyl and heterocycloalkenyl are optionally selected from halogen, hydroxy, amino, C 1-6 alkyl, C 2-4 alkenyl, C 2-4 alkynyl, C 3-6 cycloalkyl, halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 alkoxy, C Of 1-6 alkylamino, C 1-6 alkyl acyl, C 1-6 alkoxy acyl, aminoacyl, C 1-6 alkylamino acyl, sulfonyl, sulfinyl, mercapto, nitro, cyano One or more groups are substituted; and
Ra2选自氢、卤素、羟基、氨基、C1-6烷基、C3-6环烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、C1-6烷氨基、C1-6烷基酰基、C1-6烷氧基酰基、氨基酰基、C1-6烷氨基酰基、磺酰基、亚磺酰基、巯基、硝基和氰基。R a2 is selected from hydrogen, halogen, hydroxyl, amino, C 1-6 alkyl, C 3-6 cycloalkyl, halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 Alkoxy, C 1-6 alkylamino, C 1-6 alkyl acyl, C 1-6 alkoxy acyl, aminoacyl, C 1-6 alkylamino acyl, sulfonyl, sulfinyl, mercapto, nitro and cyano.
在另一些具体的实施方案中,本发明提供通式Ia的化合物或其立体异构体、药学可接受的盐、水合物、溶剂合物或结晶,其中Ra1、Ra2与它们连接的碳原子一起构成含有1个N原子的五元或六元杂芳基、杂环烷基或杂环烯基,其中所述的杂芳基、杂环烷基和杂环烯基任选被选自卤素、羟基、氨基、C1-6烷基、C2-4烯基、C2-4炔基、C3-6环烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、C1-6烷氨基、C1-6烷基酰基、C1-6烷氧基酰基、氨基酰基、C1-6烷氨基酰基、磺酰基、亚磺酰基、巯基、硝基、氰基中的一个或多个基团取代。In other specific embodiments, the present invention provides compounds of general formula Ia, or stereoisomers, pharmaceutically acceptable salts, hydrates, solvates or crystals thereof, wherein R a1 , R a2 and the carbon to which they are attached atoms together to form a five- or six-membered heteroaryl, heterocycloalkyl or heterocycloalkenyl containing 1 N atom, wherein said heteroaryl, heterocycloalkyl and heterocycloalkenyl are optionally selected from Halogen, hydroxyl, amino, C 1-6 alkyl, C 2-4 alkenyl, C 2-4 alkynyl, C 3-6 cycloalkyl, halogenated C 1-6 alkyl, C 1-6 alkoxy group, halogenated C 1-6 alkoxy, C 1-6 alkylamino, C 1-6 alkyl acyl, C 1-6 alkoxy acyl, aminoacyl, C 1-6 alkylamino acyl, sulfonyl, One or more groups of sulfinyl, mercapto, nitro, and cyano are substituted.
在另一些具体的实施方案中,本发明提供通式Ia的化合物或其立体异构体、药学可接受的盐、水合物、溶剂合物或结晶,其中Ra1选自含有2个N原子的五元杂芳基、杂环烷基和杂环烯基,其中所述的杂芳基、杂环烷基和杂环烯基任选被选自卤素、羟基、氨基、C1-6烷基、C2-4烯基、C2-4炔基、C3-6环烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、C1-6烷氨基、C1-6烷基酰基、C1-6烷氧基酰基、氨基酰基、C1-6烷氨基酰基、磺酰基、亚磺酰基、巯基、硝基、氰基中的一个或多个基团取代;In other specific embodiments, the present invention provides a compound of general formula Ia, or a stereoisomer, pharmaceutically acceptable salt, hydrate, solvate or crystal thereof, wherein R a1 is selected from the group consisting of 2 N atoms Five-membered heteroaryl, heterocycloalkyl and heterocycloalkenyl, wherein said heteroaryl, heterocycloalkyl and heterocycloalkenyl are optionally selected from halogen, hydroxy, amino, C 1-6 alkyl , C 2-4 alkenyl, C 2-4 alkynyl, C 3-6 cycloalkyl, halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 alkoxy, C 1-6 alkylamino, C 1-6 alkyl acyl, C 1-6 alkoxy acyl, aminoacyl, C 1-6 alkylamino acyl, sulfonyl, sulfinyl, mercapto, nitro, cyano substituted with one or more groups;
Ra2选自氢、卤素、羟基、氨基、C1-6烷基、C3-6环烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、C1-6烷氨基、C1-6烷基酰基、C1-6烷氧基酰基、氨基酰基、C1-6烷氨基酰基、磺酰基、亚磺酰基、巯基、硝基和氰基;或者R a2 is selected from hydrogen, halogen, hydroxyl, amino, C 1-6 alkyl, C 3-6 cycloalkyl, halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 Alkoxy, C 1-6 alkylamino, C 1-6 alkyl acyl, C 1-6 alkoxy acyl, aminoacyl, C 1-6 alkylamino acyl, sulfonyl, sulfinyl, mercapto, nitro and cyano; or
Ra1、Ra2与它们连接的碳原子一起构成含有2个N原子的五元杂芳基、杂环烷基或杂环烯基。R a1 , R a2 together with the carbon atoms to which they are attached form a five-membered heteroaryl, heterocycloalkyl or heterocycloalkenyl group containing 2 N atoms.
在另一些具体的实施方案中,本发明提供通式Ia的化合物或其立体异构体、药学可接受的盐、水合物、溶剂合物或结晶,其中Ra1选自含有1个N原子和1个硫原子的五元杂芳基、杂环烷基和杂环烯基,其中所述的杂芳基、杂环烷基和杂环烯基任选被选自卤素、羟基、氨基、C1-6烷基、C2-4烯基、C2-4炔基、C3-6环烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、C1-6烷氨基、C1-6烷基酰基、C1-6烷氧基酰基、氨基酰基、C1-6烷氨基酰基、磺酰基、亚磺酰基、巯基、硝基、氰基中的一个或多个基团取代;In other specific embodiments, the present invention provides a compound of general formula Ia, or a stereoisomer, pharmaceutically acceptable salt, hydrate, solvate or crystal thereof, wherein R a1 is selected from containing 1 N atom and Five-membered heteroaryl, heterocycloalkyl and heterocycloalkenyl with 1 sulfur atom, wherein said heteroaryl, heterocycloalkyl and heterocycloalkenyl are optionally selected from halogen, hydroxyl, amino, C 1-6 alkyl, C 2-4 alkenyl, C 2-4 alkynyl, C 3-6 cycloalkyl, halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1- 6 alkoxy, C 1-6 alkylamino, C 1-6 alkyl acyl, C 1-6 alkoxy acyl, aminoacyl, C 1-6 alkylamino acyl, sulfonyl, sulfinyl, mercapto, nitro One or more groups in cyano group and cyano group are substituted;
Ra2选自氢、卤素、羟基、氨基、C1-6烷基、C3-6环烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、C1-6烷氨基、C1-6烷基酰基、C1-6烷氧基酰基、氨基酰基、C1-6烷氨基酰基、磺酰基、亚磺酰基、巯基、硝基和氰基;或者R a2 is selected from hydrogen, halogen, hydroxyl, amino, C 1-6 alkyl, C 3-6 cycloalkyl, halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 Alkoxy, C 1-6 alkylamino, C 1-6 alkyl acyl, C 1-6 alkoxy acyl, aminoacyl, C 1-6 alkylamino acyl, sulfonyl, sulfinyl, mercapto, nitro and cyano; or
Ra1、Ra2与它们连接的碳原子一起构成含有1个N原子和1个硫原子的五元杂芳基、杂环烷基或杂环烯基。R a1 , R a2 together with the carbon atoms to which they are attached form a five-membered heteroaryl, heterocycloalkyl or heterocycloalkenyl group containing 1 N atom and 1 sulfur atom.
在另一些具体的实施方案中,本发明提供通式Ia的化合物或其立体异构体、药学可接受的盐、水合物、溶剂合物或结晶,其中Ra1选自含有1个N原子和1个氧原子的五元杂芳基、杂环烷基和杂环烯基,其中所述的杂芳基、杂环烷基和杂环烯基任选被选自卤素、羟基、氨基、C1-6烷基、C2-4烯基、C2-4炔基、C3-6环烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、C1-6烷氨基、C1-6烷基酰基、C1-6烷氧基酰基、氨基酰基、C1-6烷氨基酰基、磺酰基、亚磺酰基、巯基、硝基、氰基中的一个或多个基团取代;In other specific embodiments, the present invention provides a compound of general formula Ia, or a stereoisomer, pharmaceutically acceptable salt, hydrate, solvate or crystal thereof, wherein R a1 is selected from containing 1 N atom and Five-membered heteroaryl, heterocycloalkyl and heterocycloalkenyl with 1 oxygen atom, wherein said heteroaryl, heterocycloalkyl and heterocycloalkenyl are optionally selected from halogen, hydroxyl, amino, C 1-6 alkyl, C 2-4 alkenyl, C 2-4 alkynyl, C 3-6 cycloalkyl, halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1- 6 alkoxy, C 1-6 alkylamino, C 1-6 alkyl acyl, C 1-6 alkoxy acyl, aminoacyl, C 1-6 alkylamino acyl, sulfonyl, sulfinyl, mercapto, nitro One or more groups in cyano group and cyano group are substituted;
Ra2选自氢、卤素、羟基、氨基、C1-6烷基、C3-6环烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、C1-6烷氨基、C1-6烷基酰基、C1-6烷氧基酰基、氨基酰基、C1-6烷氨基酰基、磺酰基、亚磺酰基、巯基、硝基和氰基;或者R a2 is selected from hydrogen, halogen, hydroxyl, amino, C 1-6 alkyl, C 3-6 cycloalkyl, halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 Alkoxy, C 1-6 alkylamino, C 1-6 alkyl acyl, C 1-6 alkoxy acyl, aminoacyl, C 1-6 alkylamino acyl, sulfonyl, sulfinyl, mercapto, nitro and cyano; or
Ra1、Ra2与它们连接的碳原子一起构成含有1个N原子和1个氧原子的五元杂芳基、杂环烷基或杂环烯基。R a1 , R a2 together with the carbon atoms to which they are attached constitute a five-membered heteroaryl, heterocycloalkyl or heterocycloalkenyl group containing 1 N atom and 1 oxygen atom.
在另一些具体的实施方案中,本发明提供通式Ia的化合物或其立体异构体、药学可接受的盐、水合物、溶剂合物或结晶,其中Ra1选自仅含有1个S原子的五元杂芳基、杂环烷基和杂环烯基,其中所述的杂芳基、杂环烷基和杂环烯基任选被选自卤素、羟基、氨基、C1-6烷基、C2-4烯基、C2-4炔基、C3-6环烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、C1-6烷氨基、C1-6烷基酰基、C1-6烷氧基酰基、氨基酰基、C1-6烷氨基酰基、磺酰基、亚磺酰基、巯基、硝基、氰基中的一个或多个基团取代;In other specific embodiments, the present invention provides a compound of general formula Ia, or a stereoisomer, pharmaceutically acceptable salt, hydrate, solvate or crystal thereof, wherein R a1 is selected from containing only 1 S atom The five-membered heteroaryl, heterocycloalkyl and heterocycloalkenyl, wherein the heteroaryl, heterocycloalkyl and heterocycloalkenyl are optionally selected from halogen, hydroxyl, amino, C 1-6 alkane base, C 2-4 alkenyl, C 2-4 alkynyl, C 3-6 cycloalkyl, halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 alkoxy , C 1-6 alkylamino, C 1-6 alkyl acyl, C 1-6 alkoxy acyl, aminoacyl, C 1-6 alkylamino acyl, sulfonyl, sulfinyl, mercapto, nitro, cyano One or more groups in the substituted;
Ra2选自氢、卤素、羟基、氨基、C1-6烷基、C3-6环烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、C1-6烷氨基、C1-6烷基酰基、C1-6烷氧基酰基、氨基酰基、C1-6烷氨基酰基、磺酰基、亚磺酰基、巯基、硝基和氰基;或者R a2 is selected from hydrogen, halogen, hydroxyl, amino, C 1-6 alkyl, C 3-6 cycloalkyl, halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 Alkoxy, C 1-6 alkylamino, C 1-6 alkyl acyl, C 1-6 alkoxy acyl, aminoacyl, C 1-6 alkylamino acyl, sulfonyl, sulfinyl, mercapto, nitro and cyano; or
Ra1、Ra2与它们连接的碳原子一起构成仅含有1个S原子的五元杂芳基、杂环烷基或杂环烯基。R a1 , R a2 together with the carbon atoms to which they are attached form a five-membered heteroaryl, heterocycloalkyl or heterocycloalkenyl group containing only 1 S atom.
在一些优选的实施方案中,本发明提供通式Ia的化合物或其立体异构体、药学可接受的盐、水合物、溶剂合物或结晶,其中R1为H、甲基、乙基、正丙基或异丙基,R2为F,R3为OH,R4选自H、甲基、乙基、正丙基或异丙基,R5选自甲基、乙基、正丙基、异丙基、环丙基、正丁基、异丁基、叔丁基、环丁基、正戊基、异戊基、环戊基或苄基,Ra1选自呋喃环、吡咯环、噻吩环、咪唑环、吡唑环、噻唑环、异噻唑环、噁唑环、异噁唑环、三氮唑环、噻二唑环、噁二唑环、吡啶环、嘧啶环、吡嗪环、哒嗪环、苯并氮杂环、苯并氧杂环、苯并硫杂环、苯并二氮杂环、苯并二氧杂环、苯并二硫杂环、苯并氧氮杂环、苯并硫氮杂环,其中所述的Ra1任选被选自卤素、羟基、C1-6烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、氨基、C1-6烷基氨基、C1-6烷基酰基氨基、C1-6烷基酰基、氨基酰基、C1-6烷基氨基酰基、硝基、氰基中的一个或多个基团取代;Ra2、Ra3、Ra4、Ra5独立地选自氢、卤素、羟基、氨基、烷基、C3-8环烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、C1-6烷氨基、C1-6烷基酰基、C1-6烷氧基酰基、氨基酰基、C1-6烷基氨基酰基、磺酰基、亚磺酰基、巯基、硝基、氰基。In some preferred embodiments, the present invention provides a compound of general formula Ia, or a stereoisomer, pharmaceutically acceptable salt, hydrate, solvate or crystal thereof, wherein R 1 is H, methyl, ethyl, n-propyl or isopropyl, R 2 is F, R 3 is OH, R 4 is selected from H, methyl, ethyl, n-propyl or isopropyl, R 5 is selected from methyl, ethyl, n-propyl base, isopropyl, cyclopropyl, n-butyl, isobutyl, tert-butyl, cyclobutyl, n-pentyl, isopentyl, cyclopentyl or benzyl, R a1 is selected from furan ring, pyrrole ring , thiophene ring, imidazole ring, pyrazole ring, thiazole ring, isothiazole ring, oxazole ring, isoxazole ring, triazole ring, thiadiazole ring, oxadiazole ring, pyridine ring, pyrimidine ring, pyrazine ring, pyridazine ring, benzodiazepine, benzoxane, benzothiazepine, benzodiazepine, benzodioxane, benzodiazepine, benzoxazepine Ring, benzothiazepine heterocycle, wherein said R a1 is optionally selected from halogen, hydroxyl, C 1-6 alkyl, halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 alkoxy, amino, C 1-6 alkylamino, C 1-6 alkyl acylamino, C 1-6 alkyl acyl, amino acyl, C 1-6 alkyl amino acyl, nitro, One or more groups in the cyano group are substituted; R a2 , R a3 , R a4 , R a5 are independently selected from hydrogen, halogen, hydroxyl, amino, alkyl, C 3-8 cycloalkyl, halogenated C 1 -6 alkyl, C 1-6 alkoxy, halogenated C 1-6 alkoxy, C 1-6 alkylamino, C 1-6 alkyl acyl, C 1-6 alkoxy acyl, aminoacyl, C 1-6 alkylaminoacyl, sulfonyl, sulfinyl, mercapto, nitro, cyano.
在一些优选的实施方案中,本发明提供通式Ia的化合物或其立体异构体、药学可接受的盐、水合物、溶剂合物或结晶,其中R1为H或甲基,R2为F,R3为OH,R4选自H、甲基、乙基、正丙基或异丙基,R5选自甲基、乙基、正丙基、异丙基、环丙基、正丁基、异丁基、叔丁基、环丁基、正戊基、异戊基、环戊基或苄基,Ra1选自呋喃环、吡咯环、噻吩环、咪唑环、吡唑环、噻唑环、异噻唑环、噁唑环、异噁唑环、三氮唑环、噻二唑环、噁二唑环、吡啶环、嘧啶环、吡嗪环、哒嗪环、苯并呋喃环、苯并吡咯环、苯并噻吩环、苯并咪唑环、苯并吡唑环、苯并噻唑环、苯并异噻唑环、苯并噁唑环、苯并异噁唑环、苯并三氮唑环、苯并噻二唑环、苯并噁二唑环、苯并吡啶环、苯并嘧啶环、苯并吡嗪环、苯并哒嗪环,其中所述的Ra1任选被选自卤素、羟基、C1-6烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、氨基、C1-6烷基氨基、C1-6烷基酰基氨基、C1-6烷基酰基、氨基酰基、C1-6烷基氨基酰基、硝基、氰基中的一个或多个基团取代;Ra2、Ra3、Ra4、Ra5独立地选自氢、卤素、羟基、氨基、烷基、C3-8环烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、C1-6烷氨基、C1-6烷基酰基、C1-6烷氧基酰基、氨基酰基、C1-6烷基氨基酰基、磺酰基、亚磺酰基、巯基、硝基、氰基。In some preferred embodiments, the present invention provides a compound of general formula Ia, or a stereoisomer, pharmaceutically acceptable salt, hydrate, solvate or crystal thereof, wherein R1 is H or methyl and R2 is F, R 3 is OH, R 4 is selected from H, methyl, ethyl, n-propyl or isopropyl, R 5 is selected from methyl, ethyl, n-propyl, isopropyl, cyclopropyl, n-propyl Butyl, isobutyl, tert-butyl, cyclobutyl, n-pentyl, isopentyl, cyclopentyl or benzyl, R a1 is selected from furan ring, pyrrole ring, thiophene ring, imidazole ring, pyrazole ring, Thiazole ring, isothiazole ring, oxazole ring, isoxazole ring, triazole ring, thiadiazole ring, oxadiazole ring, pyridine ring, pyrimidine ring, pyrazine ring, pyridazine ring, benzofuran ring, benzopyrrole ring, benzothiophene ring, benzimidazole ring, benzopyrazole ring, benzothiazole ring, benzisothiazole ring, benzoxazole ring, benzisoxazole ring, benzotriazole ring, benzothiadiazole ring, benzoxadiazole ring, benzopyridine ring, benzopyrimidine ring, benzopyrazine ring, benzopyridazine ring, wherein said R a1 is optionally selected from halogen , hydroxy, C 1-6 alkyl, halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 alkoxy, amino, C 1-6 alkylamino, C 1- Substituted by one or more groups in 6 alkyl acylamino, C 1-6 alkyl acyl, aminoacyl, C 1-6 alkyl amino acyl, nitro, cyano; R a2 , R a3 , R a4 , R a5 is independently selected from hydrogen, halogen, hydroxyl, amino, alkyl, C 3-8 cycloalkyl, halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 alkoxy base, C 1-6 alkylamino, C 1-6 alkyl acyl, C 1-6 alkoxy acyl, aminoacyl, C 1-6 alkylaminoacyl, sulfonyl, sulfinyl, mercapto, nitro, cyano group.
在一些优选的实施方案中,本发明提供通式Ia的化合物或其立体异构体、药学可接受的盐、水合物、溶剂合物或结晶,其中R1为H、甲基、乙基、正丙基或异丙基,R2为F,R3为OH,R4选自H、甲基、乙基、正丙基或异丙基,R5选自甲基、乙基、正丙基、异丙基、正丁基、异丁基、叔丁基、正戊基、异戊基或苄基,Ra1、Ra2与它们连接的碳原子一起构成苯并氮杂环、苯并氧杂环、苯并硫杂环、苯并二氮杂环、苯并二氧杂环、苯并二硫杂环、苯并氧氮杂环、苯并硫氮杂环,其中所述的苯并氮杂环、苯并氧杂环、苯并硫杂环、苯并二氮杂环、苯并二氧杂环、苯并二硫杂环、苯并氧氮杂环、苯并硫氮杂环任选被选自卤素、羟基、C1-6烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、氨基、C1-6烷基氨基、C1-6烷基酰基氨基、C1-6烷基酰基、氨基酰基、C1-6烷基氨基酰基、硝基、氰基中的一个或多个基团取代;Ra3、Ra4、Ra5独立地选自氢、卤素、羟基、氨基、烷基、C3-8环烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、C1-6烷氨基、C1-6烷基酰基、C1-6烷氧基酰基、氨基酰基、C1-6烷基氨基酰基、磺酰基、亚磺酰基、巯基、硝基、氰基。In some preferred embodiments, the present invention provides a compound of general formula Ia, or a stereoisomer, pharmaceutically acceptable salt, hydrate, solvate or crystal thereof, wherein R 1 is H, methyl, ethyl, n-propyl or isopropyl, R 2 is F, R 3 is OH, R 4 is selected from H, methyl, ethyl, n-propyl or isopropyl, R 5 is selected from methyl, ethyl, n-propyl base, isopropyl, n-butyl, isobutyl, tert-butyl, n-pentyl, isopentyl or benzyl, R a1 , R a2 together with the carbon atoms to which they are attached form benzazepine, benzo Oxygen heterocycle, benzothiazepine, benzodiazepine, benzodioxane, benzodiazepine, benzothiazepine, benzothiazepine, wherein the benzene Benzodiazepine, benzoxazepine, benzothiazepine, benzodiazepine, benzodiazepine, benzodiazepine, benzoxazepine, benzothiazepine Ring is optionally selected from halogen, hydroxy, C1-6 alkyl, halogenated C1-6 alkyl, C1-6 alkoxy, halogenated C1-6 alkoxy, amino, C1-6 Substitution of one or more groups in alkylamino, C 1-6 alkyl acylamino, C 1-6 alkyl acyl, aminoacyl, C 1-6 alkyl amino acyl, nitro, and cyano; R a3 , R a4 , R a5 are independently selected from hydrogen, halogen, hydroxyl, amino, alkyl, C 3-8 cycloalkyl, halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1 -6 alkoxy, C 1-6 alkylamino, C 1-6 alkyl acyl, C 1-6 alkoxy acyl, aminoacyl, C 1-6 alkylaminoacyl, sulfonyl, sulfinyl, mercapto , nitro, cyano.
在一些优选的实施方案中,本发明提供通式Ia的化合物或其立体异构体、药学可接受的盐、水合物、溶剂合物或结晶,其中R1为H、甲基、乙基、正丙基或异丙基,R2为F,R3为OH,R4选自H、甲基、乙基、正丙基或异丙基,R5选自甲基、乙基、正丙基、异丙基、正丁基、异丁基、叔丁基、正戊基、异戊基或苄基,Ra1、Ra2与它们连接的碳原子一起构成苯并呋喃环、苯并吡咯环、苯并噻吩环、苯并咪唑环、苯并吡唑环、苯并噻唑环、苯并异噻唑环、苯并噁唑环、苯并异噁唑环、苯并三氮唑环、苯并噻二唑环、苯并噁二唑环、苯并吡啶环、苯并嘧啶环、苯并吡嗪环、苯并哒嗪环,其中所述的苯并呋喃环、苯并吡咯环、苯并噻吩环、苯并咪唑环、苯并吡唑环、苯并噻唑环、苯并异噻唑环、苯并噁唑环、苯并异噁唑环、苯并三氮唑环、苯并噻二唑环、苯并噁二唑环、苯并吡啶环、苯并嘧啶环、苯并吡嗪环、苯并哒嗪环任选被选自卤素、羟基、C1-6烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、氨基、C1-6烷基氨基、C1-6烷基酰基氨基、C1-6烷基酰基、氨基酰基、C1-6烷基氨基酰基、硝基、氰基中的一个或多个基团取代;Ra3、Ra4、Ra5独立地选自氢、卤素、羟基、氨基、烷基、C3-8环烷基、卤代C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、C1-6烷氨基、C1-6烷基酰基、C1-6烷氧基酰基、氨基酰基、C1-6烷基氨基酰基、磺酰基、亚磺酰基、巯基、硝基、氰基。In some preferred embodiments, the present invention provides a compound of general formula Ia, or a stereoisomer, pharmaceutically acceptable salt, hydrate, solvate or crystal thereof, wherein R 1 is H, methyl, ethyl, n-propyl or isopropyl, R 2 is F, R 3 is OH, R 4 is selected from H, methyl, ethyl, n-propyl or isopropyl, R 5 is selected from methyl, ethyl, n-propyl base, isopropyl, n-butyl, isobutyl, tert-butyl, n-pentyl, isoamyl or benzyl, R a1 , R a2 together with the carbon atoms to which they are attached form a benzofuran ring, benzopyrrole Ring, benzothiophene ring, benzimidazole ring, benzopyrazole ring, benzothiazole ring, benzisothiazole ring, benzoxazole ring, benzisoxazole ring, benzotriazole ring, benzene Thiadiazole ring, benzoxadiazole ring, benzopyridine ring, benzopyrimidine ring, benzopyrazine ring, benzopyridazine ring, wherein said benzofuran ring, benzopyrrole ring, benzene ring thiophene ring, benzimidazole ring, benzopyrazole ring, benzothiazole ring, benzisothiazole ring, benzoxazole ring, benzisoxazole ring, benzotriazole ring, benzothiadi azole ring, benzoxadiazole ring, benzopyridine ring, benzopyrimidine ring, benzopyrazine ring, benzopyridazine ring is optionally selected from halogen, hydroxyl, C 1-6 alkyl, halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 alkoxy, amino, C 1-6 alkylamino, C 1-6 alkyl acylamino, C 1-6 alkyl acyl , aminoacyl, C 1-6 alkylaminoacyl, nitro, cyano group is substituted with one or more groups; R a3 , R a4 , R a5 are independently selected from hydrogen, halogen, hydroxyl, amino, alkyl , C 3-8 cycloalkyl, halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 alkoxy, C 1-6 alkylamino, C 1-6 alkyl acyl , C 1-6 alkoxyacyl, aminoacyl, C 1-6 alkylaminoacyl, sulfonyl, sulfinyl, mercapto, nitro, cyano.
在上述优选的实施方案中,本发明通式Ia的化合物,不是以下化合物:In the above-mentioned preferred embodiments, the compounds of general formula Ia of the present invention are not the following compounds:
(2S)-2-(((4-(1,2,3-噻二唑-4-基)苯-1-基)氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯;(2S)-2-(((4-(1,2,3-thiadiazol-4-yl)phenyl-1-yl)oxy)(((2R,3R,4R,5R)-5-( 2,4-Dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino ) isopropyl propionate;
(2S)-2-(((2-(喹喔啉-5-基)苯-1-基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯;(2S)-2-(((2-(quinoxalin-5-yl)phenyl-1-yloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo -3,4-Dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate isopropyl;
(2S)-2-(((4-(噻唑-2-基)苯-1-基)氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯;(2S)-2-(((4-(thiazol-2-yl)phenyl-1-yl)oxy)(((2R,3R,4R,5R)-5-(2,4-dioxo- Isopropyl 3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate;
(2S)-2-(((4-(噻唑-5-基)苯-1-基)氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯;(2S)-2-(((4-(thiazol-5-yl)phenyl-1-yl)oxy)(((2R,3R,4R,5R)-5-(2,4-dioxo- Isopropyl 3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate;
(2S)-2-(((1H-吲哚-5-基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯;(2S)-2-(((1H-Indol-5-yloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo-3,4-dihydropyrimidine Isopropyl 1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate;
(2S)-2-(((4-氟-2-甲基-1H-吲哚-5-基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯;(2S)-2-(((4-Fluoro-2-methyl-1H-indol-5-yloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo Isopropyl substituted-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate ;
(2S)-2-(((3-甲氧羰基甲基-苯并呋喃-7-基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯;(2S)-2-(((3-methoxycarbonylmethyl-benzofuran-7-yloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo- Isopropyl 3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate;
(2S)-2-(((4-氧代-2-苯基-4H-苯并吡喃-6-基)氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯;(2S)-2-(((4-oxo-2-phenyl-4H-benzopyran-6-yl)oxy)(((2R,3R,4R,5R)-5-(2, 4-Dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propane isopropyl acid;
(2S)-2-(((喹喔啉-5-基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯;(2S)-2-(((quinoxalin-5-yloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo-3,4-dihydropyrimidine- Isopropyl 1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate;
(2S)-2-(((4-氧代-2-苯基苯并二氢吡喃-7-基)氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯;(2S)-2-(((4-oxo-2-phenylchroman-7-yl)oxy)(((2R,3R,4R,5R)-5-(2,4 -Dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionic acid isopropyl ester;
(2S)-2-((((4-(1H-1,2,4-三唑-1-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯;(2S)-2-((((4-(1H-1,2,4-triazol-1-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2, 4-Dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propane isopropyl acid;
(2S)-2-(((4-(嘧啶-2-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯;(2S)-2-(((4-(pyrimidin-2-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo-3,4- Dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate isopropyl;
(2S)-2-(((S)-((4-氟-1,2-二甲基-1H-吲哚-5-基)氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯;(2S)-2-(((S)-((4-Fluoro-1,2-dimethyl-1H-indol-5-yl)oxy)(((2R,3R,4R,5R)- 5-(2,4-Dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphorus Acyl)amino)isopropyl propionate;
(2S)-2-((((6-氟-3-甲基苯并[d]异噁唑-5-基)氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯;(2S)-2-((((6-Fluoro-3-methylbenzo[d]isoxazol-5-yl)oxy)(((2R,3R,4R,5R)-5-(2 ,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino) isopropyl propionate;
(2S)-2-((((6-氟苯并[c][1,2,5]噻二唑-5-基)氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯;和(2S)-2-((((6-Fluorobenzo[c][1,2,5]thiadiazol-5-yl)oxy)(((2R,3R,4R,5R)-5- (2,4-Dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl) amino) isopropyl propionate; and
(2S)-2-(((4-(噻唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯。(2S)-2-(((4-(thiazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo-3,4- Dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionic acid isopropyl ester.
本发明提供了以下具体化合物:The present invention provides the following specific compounds:
另一方面,本发明提供本发明的通式I化合物的制备方法,该方法包括以下步骤:On the other hand, the present invention provides the preparation method of the compound of general formula I of the present invention, the method comprises the following steps:
a)式(1)的化合物在碱性条件下与三氯氧磷反应,制得式(2)的化合物;a) The compound of formula (1) is reacted with phosphorus oxychloride under basic conditions to prepare the compound of formula (2);
b)式(2)的化合物与式(3)的化合物反应制得式(4)的化合物;b) the compound of formula (2) is reacted with the compound of formula (3) to obtain the compound of formula (4);
c)式(4)的化合物与五氟苯酚反应制得式(5)的化合物;c) The compound of formula (4) is reacted with pentafluorophenol to obtain the compound of formula (5);
d)式(5)的化合物与式(6)的化合物反应制得通式(I)的化合物。d) Compounds of formula (5) are reacted with compounds of formula (6) to produce compounds of general formula (I).
其中,R1、R2、R3、R4、R5、Cy1、Cy2如以上通式I中所定义。wherein, R 1 , R 2 , R 3 , R 4 , R 5 , Cy 1 , Cy 2 are as defined in general formula I above.
第三方面,本发明提供药物组合物,其包含本发明的通式I或Ia所示化合物、立体异构体、药学可接受的盐、水合物、溶剂合物或结晶。In a third aspect, the present invention provides a pharmaceutical composition comprising the compound represented by the general formula I or Ia, a stereoisomer, a pharmaceutically acceptable salt, a hydrate, a solvate or a crystal of the present invention.
在一些实施方案中,本发明提供本发明的化合物、立体异构体、药学可接受的盐、水合物、溶剂合物或结晶及包含本发明的化合物、立体异构体、药学可接受的盐、水合物、溶剂合物或结晶的药物组合物,所述化合物或药物组合物用于治疗和/或预防由丙型肝炎病毒引起的肝脏疾病。In some embodiments, the present invention provides compounds, stereoisomers, pharmaceutically acceptable salts, hydrates, solvates or crystals of the present invention and compounds, stereoisomers, pharmaceutically acceptable salts comprising the present invention , hydrate, solvate or crystalline pharmaceutical composition for the treatment and/or prevention of liver disease caused by hepatitis C virus.
在一些实施方案中,本发明提供药物组合物,其包含通式I或Ia所示化合物、立体异构体、药学可接受的盐、水合物、溶剂合物或结晶,还包含选自下列组成的一种或多种另外的抗HCV治疗剂:HCV NS3蛋白酶抑制剂、HCV NS5B RNA依赖性RNA聚合酶抑制剂、核苷类似物、干扰素α、聚乙二醇化的干扰素、利巴韦林、左旋韦林、韦拉米啶、TLR7激动剂、TLR9激动剂、亲环蛋白抑制剂、α葡萄糖苷酶抑制剂、NS5A抑制剂和NS3解螺旋酶抑制剂。In some embodiments, the present invention provides a pharmaceutical composition comprising a compound, stereoisomer, pharmaceutically acceptable salt, hydrate, solvate or crystal of Formula I or Ia, and further comprising a composition selected from the group consisting of: One or more additional anti-HCV therapeutics of: HCV NS3 protease inhibitor, HCV NS5B RNA-dependent RNA polymerase inhibitor, nucleoside analog, interferon alpha, pegylated interferon, ribavir Lin, levovirine, viramidine, TLR7 agonists, TLR9 agonists, cyclophilin inhibitors, alpha glucosidase inhibitors, NS5A inhibitors, and NS3 helicase inhibitors.
可以将本发明的通式I和Ia所示化合物、立体异构体、药学可接受的盐、水合物、溶剂合物或结晶与药学上可接受的载体、稀释剂或赋形剂混合制备成药物制剂,以适合于经口或胃肠外给药。给药方法包括,但不限于皮内、肌内、腹膜内、静脉内、皮下、鼻内和经口途径。所述制剂可以通过任何途径施用,例如通过输注或推注,通过经上皮或皮肤粘膜(例如口腔粘膜或直肠等)吸收的途径施用。给药可以是全身的或局部的。经口施用制剂的实例包括固体或液体剂型,具体而言,包括片剂、丸剂、粒剂、粉剂、胶囊剂、糖浆、乳剂、混悬剂等。所述制剂可通过本领域已知的方法制备,且包含药物制剂领域常规使用的载体、稀释剂或赋形剂。Compounds, stereoisomers, pharmaceutically acceptable salts, hydrates, solvates or crystals of the present invention shown in general formula I and Ia can be mixed with pharmaceutically acceptable carriers, diluents or excipients to prepare. Pharmaceutical formulations suitable for oral or parenteral administration. Methods of administration include, but are not limited to, intradermal, intramuscular, intraperitoneal, intravenous, subcutaneous, intranasal, and oral routes. The formulations may be administered by any route, such as by infusion or bolus injection, by route of absorption through the epithelium or mucocutaneous (eg, oral mucosa or rectum, etc.). Administration can be systemic or local. Examples of formulations for oral administration include solid or liquid dosage forms, specifically, tablets, pills, granules, powders, capsules, syrups, emulsions, suspensions and the like. The formulations can be prepared by methods known in the art and include carriers, diluents or excipients conventionally used in the art of pharmaceutical formulations.
第四方面,本发明提供本发明的通式I和Ia所示化合物、立体异构体、药学可接受的盐、水合物、溶剂合物或结晶或本发明的药物组合物治疗黄病毒科病毒感染的受治疗者的方法,包括向所述受治疗者施用通式I和Ia的化合物、立体异构体、药学可接受的盐、水合物、溶剂合物或结晶或者包含通式I和Ia的化合物、立体异构体、药学可接受的盐、水合物、溶剂合物或结晶的药物组合物,以有效减少所述受治疗者中所述病毒的病毒载量的量施用。在一个实施方案中,本发明提供用于治疗和/或预防RNA病毒例如黄病毒科病毒感染的方法,包括向需要此治疗的个体给予本发明的化合物、其立体异构体、药学可接受的盐、水合物、溶剂合物或结晶或其药物组合物。在另一个实施方案中,本发明提供抑制RNA病毒例如黄病毒科病毒感染的方法,包括使所述病毒与治疗有效量的本发明的化合物、其立体异构体、药学可接受的盐、水合物、溶剂合物或结晶或其药物组合物接触。In the fourth aspect, the present invention provides the compounds, stereoisomers, pharmaceutically acceptable salts, hydrates, solvates or crystals of the present invention shown in general formulas I and Ia or the pharmaceutical compositions of the present invention to treat Flaviviridae viruses A method for an infected subject comprising administering to said subject a compound, stereoisomer, pharmaceutically acceptable salt, hydrate, solvate or crystal of or comprising formula I and Ia The compound, stereoisomer, pharmaceutically acceptable salt, hydrate, solvate or crystalline pharmaceutical composition of the compound is administered in an amount effective to reduce the viral load of the virus in the subject. In one embodiment, the present invention provides a method for the treatment and/or prevention of infection by an RNA virus, such as a Flaviviridae virus, comprising administering to an individual in need of such treatment a compound of the present invention, a stereoisomer thereof, a pharmaceutically acceptable Salts, hydrates, solvates or crystals or pharmaceutical compositions thereof. In another embodiment, the present invention provides a method of inhibiting infection by an RNA virus, such as a Flaviviridae virus, comprising hydrating the virus with a therapeutically effective amount of a compound of the present invention, a stereoisomer, a pharmaceutically acceptable salt, compound, solvate or crystal or a pharmaceutical composition thereof.
“黄病毒科病毒”是指黄病毒科的任何病毒,包括感染人和非人动物的那些病毒,例如黄病毒、鼠疫病毒和丙型肝炎病毒。本发明的化合物和组合物可特别用于治疗和/或预防性治疗HCV。"Flaviviridae" refers to any virus of the Flaviviridae family, including those viruses that infect humans and non-human animals, such as flaviviruses, plague virus, and hepatitis C virus. The compounds and compositions of the present invention are particularly useful in the therapeutic and/or prophylactic treatment of HCV.
另一方面,本发明提供本发明的通式I和Ia所示化合物、立体异构体、药学可接受的盐、水合物、溶剂合物或结晶用于预防或治疗病毒性感染,尤其是黄病毒感染疾病的应用,以及在制备预防和/或治疗病毒性感染疾病的药物中的应用,特别是在制备预防和/或治疗HCV病毒感染,如HCV病毒性肝炎疾病的药物中的应用。该类疾病的实例有急性丙型肝炎、慢性丙型肝炎以及丙型肝炎和乙型肝炎或丁型肝炎的混合感染。On the other hand, the present invention provides compounds, stereoisomers, pharmaceutically acceptable salts, hydrates, solvates or crystals of the present invention shown in general formula I and Ia for preventing or treating viral infections, especially yellow The application of viral infectious diseases, and the application in the preparation of medicines for preventing and/or treating viral infectious diseases, especially the application in preparing medicines for preventing and/or treating HCV viral infections, such as HCV viral hepatitis diseases. Examples of such diseases are acute hepatitis C, chronic hepatitis C and co-infection of hepatitis C and hepatitis B or D.
在一些实施方案中,本发明提供用于治疗和/或预防病毒性感染的方法,所述方法包括向有此需要的个体给予治疗和/或预防有效量的本发明的化合物或其立体异构体、药学可接受的盐、水合物、溶剂合物或结晶或本发明的药物组合物。可以向有需要的哺乳动物给予本发明的化合物或其立体异构体、药学可接受的盐、水合物、溶剂合物或结晶或本发明的药物组合物以抑制病毒,阻止病程进展。In some embodiments, the present invention provides methods for treating and/or preventing viral infections, the methods comprising administering to an individual in need thereof a therapeutically and/or prophylactically effective amount of a compound of the present invention or a stereoisomer thereof body, pharmaceutically acceptable salt, hydrate, solvate or crystal or the pharmaceutical composition of the present invention. The compounds of the present invention or their stereoisomers, pharmaceutically acceptable salts, hydrates, solvates or crystals or the pharmaceutical compositions of the present invention can be administered to mammals in need thereof to inhibit the virus and prevent the progression of the disease course.
在另一些实施方案中,所述治疗和/或预防病毒性感染的方法或用途还包括向所述个体给予本发明的式I的化合物或其立体异构体、药学可接受的盐、水合物、溶剂合物或结晶或含有它们的药物组合物,且在给予本发明的式I的化合物或其立体异构体、药学可接受的盐、水合物、溶剂合物或结晶或含有它们的药物组合物之前、之后或同时给予至少一种具有抗HCV活性的其它化合物。In other embodiments, the method or use of treating and/or preventing a viral infection further comprises administering to the individual a compound of formula I of the present invention or a stereoisomer, pharmaceutically acceptable salt, hydrate thereof , solvates or crystals or pharmaceutical compositions containing them, and in the administration of the compounds of formula I of the present invention or their stereoisomers, pharmaceutically acceptable salts, hydrates, solvates or crystals or drugs containing them At least one other compound having anti-HCV activity is administered before, after or concurrently with the composition.
在一些实施方案中,所述其它化合物中的至少一种为干扰素或利巴韦林。在一些具体的实施方案中,所述干扰素选自干扰素α2B、PEG化的干扰素α、同感干扰素、干扰素α2A和成淋巴细胞样干扰素τ。在另一些实施方案中,所述其它化合物中的至少一种选自白细胞介素2、白细胞介素6、白细胞介素12、干扰RNA、反义RNA、咪喹莫特、利巴韦林、5’-单磷酸肌苷脱氢酶抑制剂、金刚烷胺和金刚乙胺。在另一些实施方案中,所述其它化合物中的至少一种可有效抑制靶标的功能以治疗HCV感染,所述靶标选自HCV金属蛋白酶、HCV丝氨酸蛋白酶、HCV聚合酶、HCV解旋酶、HCV NS4B蛋白、HCV NS5B蛋白、HCV进入、HCV组装、HCV释出、HCVNS3/4A蛋白和IMPDH。In some embodiments, at least one of the other compounds is interferon or ribavirin. In some specific embodiments, the interferon is selected from the group consisting of interferon alpha 2B, PEGylated interferon alpha, syngeneic interferon, interferon alpha 2A, and lymphoblastoid interferon tau. In other embodiments, at least one of the other compounds is selected from the group consisting of interleukin 2, interleukin 6, interleukin 12, interfering RNA, antisense RNA, imiquimod, ribavirin, Inosine 5'-monophosphate dehydrogenase inhibitors, amantadine and rimantadine. In other embodiments, at least one of the other compounds is effective to inhibit the function of a target selected from the group consisting of HCV metalloprotease, HCV serine protease, HCV polymerase, HCV helicase, HCV NS4B protein, HCV NS5B protein, HCV entry, HCV assembly, HCV release, HCV NS3/4A protein and IMPDH.
术语定义Definition of Terms
除非另外定义,本文使用的所有技术和科学术语具有与本发明所属领域的普通技术人员通常理解的相同的含义。Unless otherwise defined, all technical and scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art to which this invention belongs.
术语“立体异构体”是指由分子中原子在空间上排列方式不同所产生的异构体。包括顺反异构体、对映异构体和构象异构体。所有立体异构体均属于本发明的范围。本发明的化合物的单独立体异构体可基本上不含其它异构体或可以混合成例如外消旋体,或者与所有其它的立体异构体混合。The term "stereoisomer" refers to isomers that result from different arrangements of atoms in a molecule in space. Including cis-trans isomers, enantiomers and conformational isomers. All stereoisomers are within the scope of the present invention. Individual stereoisomers of the compounds of the present invention may be substantially free of other isomers or may be mixed, for example, as racemates, or with all other stereoisomers.
术语“药学可接受的盐”是指本发明所述的化合物与酸形成的药学上可接受的盐,所述的酸例如可选自,但不限于:磷酸、硫酸、盐酸、氢溴酸、柠檬酸、马来酸、丙二酸、扁桃酸、琥珀酸、富马酸、醋酸、乳酸、硝酸、磺酸、对甲苯磺酸、苹果酸、甲烷磺酸或其类似物。The term "pharmaceutically acceptable salt" refers to a pharmaceutically acceptable salt formed by the compound of the present invention and an acid, for example, the acid can be selected from, but not limited to: phosphoric acid, sulfuric acid, hydrochloric acid, hydrobromic acid, Citric acid, maleic acid, malonic acid, mandelic acid, succinic acid, fumaric acid, acetic acid, lactic acid, nitric acid, sulfonic acid, p-toluenesulfonic acid, malic acid, methanesulfonic acid, or the like.
术语“溶剂合物”是指通过与溶剂分子配位形成固态或液态的配合物的本发明化合物的形式。水合物是溶剂合物的特殊形式,其中与水发生配位。在本发明范围内,溶剂合物优选是水合物。The term "solvate" refers to a form of a compound of the present invention that forms a complex in a solid or liquid state by coordination with solvent molecules. Hydrates are a special form of solvates in which coordination occurs with water. In the context of the present invention, solvates are preferably hydrates.
术语“结晶”是指本发明所述的化合物形成的各种固体形态,包括晶型、无定形。The term "crystalline" refers to various solid forms formed by the compounds of the present invention, including crystalline forms and amorphous forms.
术语“烷基”是指直链或支链的饱和烃基,优选6个碳原子以下的烃基。烷基的实施例包括甲基、乙基、正丙基、异丙基、正丁基、异丁基、叔丁基、正戊基、异戊基、新戊基、正己基、异己基、2,2-甲基丁基和2,3-二甲基丁基。术语“C1-6烷基”是指含有1-6个碳原子的直链或支链的饱和烃基。术语“C1-4烷基”是指含有1-4个碳原子的直链或支链的饱和烃基。The term "alkyl" refers to a straight or branched chain saturated hydrocarbon group, preferably a hydrocarbon group having less than 6 carbon atoms. Examples of alkyl groups include methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, tert-butyl, n-pentyl, isopentyl, neopentyl, n-hexyl, isohexyl, 2,2-methylbutyl and 2,3-dimethylbutyl. The term "C 1-6 alkyl" refers to a straight or branched chain saturated hydrocarbon group containing 1 to 6 carbon atoms. The term "C 1-4 alkyl" refers to a straight or branched chain saturated hydrocarbon group containing 1 to 4 carbon atoms.
术语“卤代烷基”是指至少被一个卤素原子取代的烷基。The term "haloalkyl" refers to an alkyl group substituted with at least one halogen atom.
术语“环烷基”是指环状的饱和烃基,优选8个碳原子以下的烃基。术语“C3-8环烷基”是指含有3-8个碳原子的环状饱和烃基。术语“C3-6环烷基”是指含有3-6个碳原子的直链或支链的饱和烃基。The term "cycloalkyl" refers to a cyclic saturated hydrocarbon group, preferably a hydrocarbon group of 8 or less carbon atoms. The term "C 3-8 cycloalkyl" refers to a cyclic saturated hydrocarbon group containing 3-8 carbon atoms. The term "C 3-6 cycloalkyl" refers to a straight or branched chain saturated hydrocarbon group containing 3 to 6 carbon atoms.
术语“烷氧基”是指-O-烷基。The term "alkoxy" refers to -O-alkyl.
术语“卤素”是指氟、氯、溴、碘。The term "halogen" refers to fluorine, chlorine, bromine, iodine.
术语“烷基氨基”是指-NH-烷基或-N-(烷基)(烷基)。The term "alkylamino" refers to -NH-alkyl or -N-(alkyl)(alkyl).
术语“烷基酰基”是指-C(O)-烷基。The term "alkylacyl" refers to -C(O)-alkyl.
术语“氨基酰基”是指-C(O)-NH2,术语“烷氨基酰基”是指-C(O)-NH-烷基或-C(O)-N-(烷基)(烷基)。The term "aminoacyl" refers to -C(O) -NH2 and the term "alkylaminoacyl" refers to -C(O)-NH-alkyl or -C(O)-N-(alkyl)(alkyl ).
术语“磺酰基”是指-S(O)2-烷基,术语“亚磺酰基”是指-S(O)-烷基。The term "sulfonyl" refers to -S(O) 2 -alkyl and the term "sulfinyl" refers to -S(O)-alkyl.
术语“芳基”是指含有一个或多个苯环的芳烃基团。合适的芳基包括苯基、萘基。The term "aryl" refers to an aromatic hydrocarbon group containing one or more benzene rings. Suitable aryl groups include phenyl, naphthyl.
术语“杂芳基”是指芳基中至少有一个碳原子被杂原子替代的芳香性基团。所述的杂原子为O、S、N。例如,本文所述杂芳基包括但不限于吡啶基、吡唑基、噁唑基、噻唑基、噻吩基、呋喃基、吡咯基、异噁唑基、异噻唑基、咪唑基、三唑基、四唑基、噁二唑基、噻二唑基、嘧啶基、哒嗪基、吡嗪基和三嗪基。The term "heteroaryl" refers to an aromatic group in which at least one carbon atom in the aryl group is replaced by a heteroatom. Said heteroatoms are O, S, N. For example, heteroaryl groups described herein include, but are not limited to, pyridyl, pyrazolyl, oxazolyl, thiazolyl, thienyl, furyl, pyrrolyl, isoxazolyl, isothiazolyl, imidazolyl, triazolyl , tetrazolyl, oxadiazolyl, thiadiazolyl, pyrimidinyl, pyridazinyl, pyrazinyl and triazinyl.
术语“杂环烷基”是指至少含有一个杂原子的饱和环状基团,其中杂原子为N、O或S。The term "heterocycloalkyl" refers to a saturated cyclic group containing at least one heteroatom, wherein the heteroatom is N, O or S.
术语“杂环烯基”是指至少含有一个杂原子的不饱和环状基团,其中杂原子为N、O或S。The term "heterocycloalkenyl" refers to an unsaturated cyclic group containing at least one heteroatom, wherein the heteroatom is N, O or S.
具体实施方式Detailed ways
下面结合实施例对本发明作进一步详细阐述,但本发明不限于这些实施例。本发明使用的试剂和原料均为商购所得。The present invention will be described in further detail below in conjunction with the examples, but the present invention is not limited to these examples. The reagents and raw materials used in the present invention are all commercially available.
实施例1(2S)-2-(((4-(吡啶-2-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 1(2S)-2-(((4-(pyridin-2-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo-3 ,4-Dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate isopropyl ester
步骤1 4-(吡啶-2-基)苯酚的制备Step 1 Preparation of 4-(pyridin-2-yl)phenol
称取1.38g 4-羟基苯硼酸(10mmol),1.58g 2-溴吡啶(10mmol),0.4g[1,1'-双(二苯基磷)二茂铁]二氯化钯(Pd(dppf)Cl2,0.5mmol)和9g碳酸铯(30mmol)于250mL的茄形瓶中,加入50mL 1,4-二氧六环和5mL水,氩气保护下90℃反应1.5h,反应结束后,加入100mL乙酸乙酯和50mL饱和氯化钠水溶液萃取,收集有机相,用饱和氯化钠水溶液洗涤(3×50mL),无水硫酸钠干燥,过滤,减压浓缩,柱层析纯化得标题化合物。Weigh 1.38g of 4-hydroxyphenylboronic acid (10mmol), 1.58g of 2-bromopyridine (10mmol), 0.4g of [1,1'-bis(diphenylphosphorus)ferrocene]palladium dichloride (Pd(dppf) )Cl 2 , 0.5mmol) and 9g cesium carbonate (30mmol) in a 250mL eggplant-shaped flask, add 50mL 1,4-dioxane and 5mL water, and react at 90°C for 1.5h under argon protection. After the reaction, Add 100 mL of ethyl acetate and 50 mL of saturated aqueous sodium chloride solution for extraction, collect the organic phase, wash with saturated aqueous sodium chloride solution (3×50 mL), dry over anhydrous sodium sulfate, filter, concentrate under reduced pressure, and purify by column chromatography to obtain the title compound .
LC-MS m/z:[M+H]+=172。LC-MS m/z: [M+H] + =172.
步骤2 2-(((五氟苯氧基)(4-(吡啶-2-基)苯氧基)磷酰基)氨基)丙酸异丙酯的制备Step 2 Preparation of isopropyl 2-(((pentafluorophenoxy)(4-(pyridin-2-yl)phenoxy)phosphoryl)amino)propionate
量取0.32mL三氯氧磷于反应瓶中,加入5mL无水四氢呋喃,-60℃下滴加10mL溶解有342mg步骤1所得物4-(吡啶-2-基)苯酚和0.42mL三乙胺的无水四氢呋喃溶液,滴毕,升温到25℃反应2h,然后于-60℃下加入334mg丙氨酸异丙酯盐酸盐,搅拌10min后滴加5mL溶解有1.05mL三乙胺的无水四氢呋喃,滴加完毕,缓慢升温到-5℃,加入5mL溶解有0.385g五氟苯酚和0.42mL三乙胺的无水四氢呋喃液体,滴加完毕,室温反应30分钟,反应结束后,柱层析纯化得到标题化合物。Measure 0.32 mL of phosphorus oxychloride into a reaction flask, add 5 mL of anhydrous tetrahydrofuran, and dropwise add 10 mL of a solution containing 342 mg of 4-(pyridin-2-yl)phenol obtained in step 1 and 0.42 mL of triethylamine at -60°C. Anhydrous tetrahydrofuran solution, after dripping, warmed up to 25°C for reaction for 2h, then added 334mg of alanine isopropyl ester hydrochloride at -60°C, stirred for 10min, and added dropwise 5mL of anhydrous tetrahydrofuran dissolved with 1.05mL of triethylamine , the dropwise addition was completed, the temperature was slowly raised to -5°C, 5 mL of anhydrous tetrahydrofuran liquid in which 0.385 g of pentafluorophenol and 0.42 mL of triethylamine were dissolved was added, the dropwise addition was completed, and the reaction was carried out at room temperature for 30 minutes. The title compound was obtained.
步骤3(2S)-2-(((4-(吡啶-2-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯的制备Step 3(2S)-2-(((4-(pyridin-2-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo-3, Preparation of isopropyl 4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate
称取260mg(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷(1mmol)于反应瓶中,密封,氩气保护下注射10mL无水四氢呋喃(THF),0℃注射10mLt-BuMgCl溶液(1M,于THF中),室温反应3h后,注射10mL溶有600mg步骤2所得物2-(((五氟苯氧基)(4-(吡啶-2-基)苯氧基)磷酰基)氨基)丙酸异丙酯的四氢呋喃溶液,25℃反应16h,反应完毕后,加入15mL 2N HCl溶液淬灭反应,乙酸乙酯萃取,有机相用饱和碳酸氢钠溶液洗涤,干燥,减压浓缩,柱层析纯化,得标题化合物。Weigh 260mg (2'R)-2'-deoxy-2'-fluoro-2'-methyluridine (1mmol) in a reaction bottle, seal it, inject 10mL anhydrous tetrahydrofuran (THF) under argon protection, 0 ℃, inject 10 mL of t-BuMgCl solution (1M in THF), and after 3 hours of reaction at room temperature, inject 10 mL of 2-(((pentafluorophenoxy)(4-(pyridin-2-yl)benzene dissolved in 600 mg of step 2) The tetrahydrofuran solution of isopropyl oxy)phosphoryl)amino)propionate was reacted at 25°C for 16h. After the reaction was completed, 15mL of 2N HCl solution was added to quench the reaction, extracted with ethyl acetate, and the organic phase was washed with saturated sodium bicarbonate solution, Dry, concentrate under reduced pressure, and purify by column chromatography to obtain the title compound.
1H NMR(300MHz,DMSO-d6)δppm:11.51(s,1H),8.64-8.65(m,1H),8.08-8.11(m,2H),7.93-7.95(m,2H),7.56-7.59(m,1H),7.32-7.35(m,3H),6.12-6.16(m,2H),5.84-5.87(m,1H),5.58(m,1H),4.87-4.89(m,1H),4.36-4.38(m,1H),4.26-4.28(m,1H),4.01(m,1H),3.82-3.84(m,2H),1.26-1.29(m,6H),1.13-1.16(m,6H)。 1 H NMR (300MHz, DMSO-d 6 ) δppm: 11.51 (s, 1H), 8.64-8.65 (m, 1H), 8.08-8.11 (m, 2H), 7.93-7.95 (m, 2H), 7.56-7.59 (m,1H),7.32-7.35(m,3H),6.12-6.16(m,2H),5.84-5.87(m,1H),5.58(m,1H),4.87-4.89(m,1H),4.36 -4.38(m,1H),4.26-4.28(m,1H),4.01(m,1H),3.82-3.84(m,2H),1.26-1.29(m,6H),1.13-1.16(m,6H) .
LC-MS m/z:[M+H]+=607。LC-MS m/z: [M+H] + =607.
实施例2(2S)-2-(((4-(3-氟吡啶-2-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 2(2S)-2-(((4-(3-fluoropyridin-2-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo Isopropyl substituted-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate
步骤1 4-(3-氟吡啶-2-基)苯酚的制备Step 1 Preparation of 4-(3-fluoropyridin-2-yl)phenol
以4-羟基苯硼酸和2-溴-3-氟吡啶为原料,同实施例1步骤1的方法得到标题化合物。Using 4-hydroxyphenylboronic acid and 2-bromo-3-fluoropyridine as starting materials, the title compound was obtained by the same method as in Example 1, step 1.
LC-MS m/z:[M+H]+=190。LC-MS m/z: [M+H] + =190.
步骤2(2S)-2-(((4-(3-氟吡啶-2-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯的制备Step 2(2S)-2-(((4-(3-fluoropyridin-2-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo -3,4-Dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)isopropyl propionate preparation
以步骤1所得物4-(3-氟吡啶-2-基)苯酚、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。4-(3-Fluoropyridin-2-yl)phenol, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol and (2'R)-2'-deoxygenate obtained in step 1 -2'-Fluoro-2'-methyluridine was used as the raw material, and the target compound was obtained by the same method as steps 2 and 3 of Example 1.
1H NMR(300MHz,DMSO-d6)δppm:11.51(s,1H),8.53-8.54(m,1H),7.92-7.95(m,2H),7.83-7.86(m,1H),7.54-7.57(m,1H),7.45-7.47(m,1H),7.33-7.35(m,2H),6.19-6.23(m,1H),6.07-6.15(m,1H),5.91-5.93(m,1H),5.57-5.60(m,1H),4.87-4.90(m,1H),4.41-4.46(m,1H),4.30-4.30(m,1H),4.05(m,1H),3.79-3.82(m,2H),1.20-1.27(m,6H),1.13-1.17(m,6H)。 1 H NMR (300 MHz, DMSO-d 6 ) δppm: 11.51 (s, 1H), 8.53-8.54 (m, 1H), 7.92-7.95 (m, 2H), 7.83-7.86 (m, 1H), 7.54-7.57 (m,1H),7.45-7.47(m,1H),7.33-7.35(m,2H),6.19-6.23(m,1H),6.07-6.15(m,1H),5.91-5.93(m,1H) ,5.57-5.60(m,1H),4.87-4.90(m,1H),4.41-4.46(m,1H),4.30-4.30(m,1H),4.05(m,1H),3.79-3.82(m, 2H), 1.20-1.27 (m, 6H), 1.13-1.17 (m, 6H).
LC-MS m/z:[M+H]+=625。LC-MS m/z: [M+H] + =625.
实施例3(2S)-2-(((4-(2-氟吡啶-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 3(2S)-2-(((4-(2-fluoropyridin-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo Isopropyl substituted-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate
步骤1 4-(2-氟吡啶-4-基)苯酚的制备Step 1 Preparation of 4-(2-fluoropyridin-4-yl)phenol
以4-羟基苯硼酸和2-氟-4-溴吡啶为原料,同实施例1步骤1的方法制得标题化合物。Using 4-hydroxyphenylboronic acid and 2-fluoro-4-bromopyridine as starting materials, the title compound was obtained by the same method as Example 1, step 1.
LC-MS m/z:[M+H]+=190。LC-MS m/z: [M+H] + =190.
步骤2(2S)-2-(((4-(2-氟吡啶-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯的制备Step 2(2S)-2-(((4-(2-fluoropyridin-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo -3,4-Dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)isopropyl propionate preparation
以步骤1所得物4-(2-氟吡啶-4-基)苯酚、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。4-(2-Fluoropyridin-4-yl)phenol, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol and (2'R)-2'-deoxygenate obtained in step 1 -2'-Fluoro-2'-methyluridine was used as the raw material, and the target compound was obtained by the same method as steps 2 and 3 of Example 1.
1H NMR(300MHz,DMSO-d6)δppm:11.51(s,1H),8.28-8.30(m,1H),7.89-7.92(m,2H),7.68-7.70(m,1H),7.55-7.58(m,1H),7.52(m,1H),7.36-7.39(m,2H),6.11-6.19(m,1H),5.98-6.05(m,1H),5.84-5.86(m,1H),5.54-5.56(m,1H),4.81-4.90(m,1H),4.37-4.39(m,1H),4.26-4.28(m,1H),3.89-4.01(m,1H),3.83-3.85(m,2H),1.22-1.29(m,6H),1.14-1.16(m,6H)。 1 H NMR (300MHz, DMSO-d 6 ) δppm: 11.51 (s, 1H), 8.28-8.30 (m, 1H), 7.89-7.92 (m, 2H), 7.68-7.70 (m, 1H), 7.55-7.58 (m,1H),7.52(m,1H),7.36-7.39(m,2H),6.11-6.19(m,1H),5.98-6.05(m,1H),5.84-5.86(m,1H),5.54 -5.56(m, 1H), 4.81-4.90(m, 1H), 4.37-4.39(m, 1H), 4.26-4.28(m, 1H), 3.89-4.01(m, 1H), 3.83-3.85(m, 2H), 1.22-1.29 (m, 6H), 1.14-1.16 (m, 6H).
LC-MS m/z:[M+H]+=625。LC-MS m/z: [M+H] + =625.
实施例4(2S)-2-(((4-(吡啶-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 4(2S)-2-(((4-(pyridin-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo-3 ,4-Dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate isopropyl ester
步骤1 4-(吡啶-4-基)苯酚的制备Step 1 Preparation of 4-(pyridin-4-yl)phenol
以4-羟基苯硼酸和4-溴吡啶为原料,同实施例1步骤1的方法制得标题化合物。LC-MS m/z:[M+H]+=172。Using 4-hydroxyphenylboronic acid and 4-bromopyridine as starting materials, the title compound was prepared in the same manner as in Example 1, step 1. LC-MS m/z: [M+H] + =172.
步骤2(2S)-2-(((4-(吡啶-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯的制备Step 2(2S)-2-(((4-(pyridin-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo-3, Preparation of isopropyl 4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate
以步骤1所得物4-(吡啶-4-基)苯酚、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。4-(Pyridin-4-yl)phenol, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol and (2'R)-2'-deoxy-2' obtained in step 1 -Fluoro-2'-methyluridine was used as the raw material, and the target compound was obtained by the same method as steps 2 and 3 of Example 1.
1H NMR(500MHz,DMSO-d6)δppm:11.52(s,1H),8.61-8.62(m,2H),7.82-7.84(m,2H),7.67-7.68(m,2H),7.36-7.37(m,1H),7.32-7.34(m,2H),6.19-6.21(m,1H),6.13-6.16(m,1H),5.90-5.91(m,1H),5.56-5.58(m,1H),4.83-4.86(m,1H),4.30-4.31(m,1H),4.26-4.29(m,1H),4.01-4.05(m,1H),3.82-3.84(m,2H),1.26-1.28(m,6H),1.13-1.15(m,6H)。 1 H NMR (500MHz, DMSO-d 6 ) δppm: 11.52 (s, 1H), 8.61-8.62 (m, 2H), 7.82-7.84 (m, 2H), 7.67-7.68 (m, 2H), 7.36-7.37 (m,1H),7.32-7.34(m,2H),6.19-6.21(m,1H),6.13-6.16(m,1H),5.90-5.91(m,1H),5.56-5.58(m,1H) ,4.83-4.86(m,1H),4.30-4.31(m,1H),4.26-4.29(m,1H),4.01-4.05(m,1H),3.82-3.84(m,2H),1.26-1.28( m, 6H), 1.13-1.15 (m, 6H).
LC-MS m/z:[M+H]+=607。LC-MS m/z: [M+H] + =607.
实施例5(2S)-2-(((4-(1-甲基-1H-吡唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 5(2S)-2-(((4-(1-methyl-1H-pyrazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2 ,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino) isopropyl propionate
步骤1 4-(1-甲基-1H-吡唑-4-基)苯酚Step 1 4-(1-Methyl-1H-pyrazol-4-yl)phenol
以4-羟基苯硼酸和1-甲基-4-溴-1H-吡唑为原料,同实施例1步骤1的方法制得标题化合物。Using 4-hydroxyphenylboronic acid and 1-methyl-4-bromo-1H-pyrazole as starting materials, the title compound was obtained by the same method as Example 1, step 1.
LC-MS m/z:[M+H]+=175。LC-MS m/z: [M+H] + =175.
步骤2(2S)-2-(((4-(1-甲基-1H-吡唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯的制备Step 2(2S)-2-(((4-(1-methyl-1H-pyrazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2, 4-Dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propane Preparation of isopropyl acid
以步骤1所得物4-(1-甲基-1H-吡唑-4-基)苯酚、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。4-(1-Methyl-1H-pyrazol-4-yl)phenol, phosphorus oxychloride, L-alanine isopropyl hydrochloride, pentafluorophenol and (2'R) obtained in step 1 -2'-Deoxy-2'-fluoro-2'-methyluridine was used as the raw material, and the target compound was obtained by the same method as steps 2 and 3 of Example 1.
1H NMR(300MHz,DMSO-d6)δppm:11.49(s,1H),8.06(s,1H),7.79(m,1H),7.52-7.58(m,3H),7.15-7.17(m,2H),6.00-6.06(m,2H),5.86-5.88(m,1H),5.55-5.60(m,1H),4.83-4.90(m,1H),4.38-4.42(m,1H),4.27-4.29(m,1H),4.03-4.06(m,1H),3.81-3.83(m,3H),3.78-3.81(m,2H),1.20-1.28(m,6H),1.14-1.17(m,6H)。 1 H NMR (300MHz, DMSO-d 6 ) δppm: 11.49(s,1H), 8.06(s,1H), 7.79(m,1H), 7.52-7.58(m,3H), 7.15-7.17(m,2H) ),6.00-6.06(m,2H),5.86-5.88(m,1H),5.55-5.60(m,1H),4.83-4.90(m,1H),4.38-4.42(m,1H),4.27-4.29 (m,1H),4.03-4.06(m,1H),3.81-3.83(m,3H),3.78-3.81(m,2H),1.20-1.28(m,6H),1.14-1.17(m,6H) .
LC-MS m/z:[M+H]+=610。LC-MS m/z: [M+H] + =610.
实施例6(2S)-2-(((4-(1-甲基-1H-吡唑-3-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 6(2S)-2-(((4-(1-methyl-1H-pyrazol-3-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2 ,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino) isopropyl propionate
步骤1 4-(1-甲基-1H-吡唑-3-基)苯酚Step 1 4-(1-Methyl-1H-pyrazol-3-yl)phenol
以4-羟基苯硼酸和3-溴-1-甲基-1H-吡唑为原料,同实施例1步骤1的方法制得标题化合物。Using 4-hydroxyphenylboronic acid and 3-bromo-1-methyl-1H-pyrazole as starting materials, the title compound was obtained by the same method as Example 1, step 1.
LC-MS m/z:[M+H]+=175。LC-MS m/z: [M+H] + =175.
步骤2(2S)-2-(((4-(1-甲基-1H-吡唑-3-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯的制备Step 2(2S)-2-(((4-(1-Methyl-1H-pyrazol-3-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2, 4-Dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propane Preparation of isopropyl acid
以步骤1所得物4-(1-甲基-1H-吡唑-3-基)苯酚、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。4-(1-Methyl-1H-pyrazol-3-yl)phenol, phosphorus oxychloride, L-alanine isopropyl hydrochloride, pentafluorophenol and (2'R) obtained in step 1 -2'-Deoxy-2'-fluoro-2'-methyluridine was used as the raw material, and the target compound was obtained by the same method as steps 2 and 3 of Example 1.
1H NMR(300MHz,DMSO-d6)δppm:11.49(s,1H),8.06(s,1H),7.79(m,1H),7.52-7.58(m,3H),7.15-7.17(m,2H),6.00-6.06(m,2H),5.86-5.88(m,1H),5.55-5.60(m,1H),4.83-4.90(m,1H),4.38-4.42(m,1H),4.27-4.29(m,1H),4.03-4.06(m,1H),3.81-3.83(m,3H),3.78-3.81(m,2H),1.20-1.28(m,6H),1.14-1.17(m,6H)。 1 H NMR (300MHz, DMSO-d 6 ) δppm: 11.49(s,1H), 8.06(s,1H), 7.79(m,1H), 7.52-7.58(m,3H), 7.15-7.17(m,2H) ),6.00-6.06(m,2H),5.86-5.88(m,1H),5.55-5.60(m,1H),4.83-4.90(m,1H),4.38-4.42(m,1H),4.27-4.29 (m,1H),4.03-4.06(m,1H),3.81-3.83(m,3H),3.78-3.81(m,2H),1.20-1.28(m,6H),1.14-1.17(m,6H) .
LC-MS m/z:[M+H]+=610。LC-MS m/z: [M+H] + =610.
实施例7(2S)-2-(((4-(1-甲基-3-甲氨基羰基-1H-吡唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 7(2S)-2-(((4-(1-methyl-3-methylaminocarbonyl-1H-pyrazol-4-yl)phenyloxy)(((2R,3R,4R,5R )-5-(2,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy )phosphoryl)amino)isopropyl propionate
步骤1 4-溴-1-甲基-1H-吡唑-3-甲酸甲酯Step 1 Methyl 4-bromo-1-methyl-1H-pyrazole-3-carboxylate
称取4.10g 4-溴-1-甲基-1H-吡唑-3-羧酸(20mmol)于250mL的茄形瓶中,加入100ml甲醇,0-4℃下,缓慢加入5ml二氯亚砜,常温下反应结束后,加入500mL乙酸乙酯和250mL饱和氯化钠水溶液萃取,收集有机相,水洗3遍,无水硫酸钠来干燥,过滤,浓缩,柱层析纯化得到标题化合物。Weigh 4.10g of 4-bromo-1-methyl-1H-pyrazole-3-carboxylic acid (20mmol) into a 250mL eggplant flask, add 100ml methanol, and slowly add 5ml thionyl chloride at 0-4°C , after the reaction at room temperature, 500 mL of ethyl acetate and 250 mL of saturated aqueous sodium chloride were added for extraction, the organic phase was collected, washed with water 3 times, dried over anhydrous sodium sulfate, filtered, concentrated, and purified by column chromatography to obtain the title compound.
LC-MS m/z:[M+H]+=219。LC-MS m/z: [M+H] + =219.
步骤2 4-(1-甲基-3-甲氧基羰基-1H-吡唑-4-基)苯酚Step 2 4-(1-Methyl-3-methoxycarbonyl-1H-pyrazol-4-yl)phenol
以步骤1所得物4-溴-1-甲基-1H-吡唑-3-甲酸甲酯和4-羟基苯硼酸为原料,同实施例1步骤1的方法制得标题化合物。Using methyl 4-bromo-1-methyl-1H-pyrazole-3-carboxylate obtained in step 1 and 4-hydroxybenzeneboronic acid as starting materials, the title compound was prepared in the same manner as in step 1 of Example 1.
LC-MS m/z:[M+H]+=233。LC-MS m/z: [M+H] + =233.
步骤3 4-(1-甲基-3-甲氨基羰基-1H-吡唑-4-基)苯酚Step 3 4-(1-Methyl-3-methylaminocarbonyl-1H-pyrazol-4-yl)phenol
称取2.33g步骤2所得物4-(1-甲基-3-甲氧基羰基-1H-吡唑-4-基)苯酚(10mmol)于250mL的茄形瓶中,加入100ml甲氨水溶液和10ml甲醇,常温下反应,反应结束后,加入500mL乙酸乙酯,250mL饱和氯化钠水溶液萃取,收集有机相,水洗3遍,无水硫酸钠来干燥,过滤,浓缩,柱层析纯化得标题化合物。Weigh 2.33 g of 4-(1-methyl-3-methoxycarbonyl-1H-pyrazol-4-yl)phenol (10 mmol) obtained in step 2 into a 250 mL eggplant-shaped flask, add 100 mL of methyl ammonia solution and 10ml of methanol was reacted at room temperature. After the reaction was completed, 500ml of ethyl acetate was added, and 250ml of saturated aqueous sodium chloride solution was added for extraction. The organic phase was collected, washed three times with water, dried over anhydrous sodium sulfate, filtered, concentrated, and purified by column chromatography to obtain the title. compound.
LC-MS m/z:[M+H]+=232。LC-MS m/z: [M+H] + =232.
步骤4(2S)-2-(((4-(1-甲基-3-甲氨基羰基-1H-吡唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Step 4(2S)-2-(((4-(1-Methyl-3-methylaminocarbonyl-1H-pyrazol-4-yl)phenyloxy)(((2R,3R,4R,5R) -5-(2,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy) Phosphoryl)amino)isopropyl propionate
以步骤3所得物4-(1-甲基-3-甲氨基羰基-1H-吡唑-4-基)苯酚、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。Using the product obtained in step 3, 4-(1-methyl-3-methylaminocarbonyl-1H-pyrazol-4-yl)phenol, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol Using (2'R)-2'-deoxy-2'-fluoro-2'-methyluridine as a raw material, the target compound was prepared in the same manner as in steps 2 and 3 of Example 1.
1H NMR(300MHz,DMSO-d6)δppm:11.45(s,1H),8.03-8.04(m,1H),7.95(m,1H),7.53-7.56(m,3H),7.11-7.14(m,2H),6.06-6.10(m,2H),5.88-5.98(m,1H),5.56-5.59(m,1H),4.85-4.87(m,1H),4.38-4.39(m,1H),4.25-4.29(m,1H),4.02-4.04(m,1H),3.81-3.87(m,3H),3.72-3.78(m,2H),2.86-2.70(m,3H),1.21-1.26(m,6H),1.13-1.16(m,6H)。 1 H NMR (300MHz, DMSO-d 6 ) δppm: 11.45(s, 1H), 8.03-8.04(m, 1H), 7.95(m, 1H), 7.53-7.56(m, 3H), 7.11-7.14(m ,2H),6.06-6.10(m,2H),5.88-5.98(m,1H),5.56-5.59(m,1H),4.85-4.87(m,1H),4.38-4.39(m,1H),4.25 -4.29(m, 1H), 4.02-4.04(m, 1H), 3.81-3.87(m, 3H), 3.72-3.78(m, 2H), 2.86-2.70(m, 3H), 1.21-1.26(m, 6H), 1.13-1.16 (m, 6H).
LC-MS m/z:[M+H]+=667。LC-MS m/z: [M+H] + =667.
实施例8(2S)-2-(((4-(1-甲基-3-氰基-1H-吡唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 8(2S)-2-(((4-(1-methyl-3-cyano-1H-pyrazol-4-yl)phenyloxy)(((2R,3R,4R,5R) -5-(2,4-Dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy) Phosphoryl)amino)isopropyl propionate
步骤1 1-甲基-3-氨基酰基-4-溴-1H-吡唑的制备Step 1 Preparation of 1-methyl-3-aminoacyl-4-bromo-1H-pyrazole
取4.10g 4-溴-1-甲基-1H-吡唑-3-羧酸(20mmol)、2.14g氯化铵(40mmol)、15.2gHATU(40mmol)和6.98ml DIPEA(40mmol)于250mL的茄形瓶中,加入100mlDMF,常温下反应12h,反应结束后,加入500mL乙酸乙酯和250mL饱和氯化钠水溶液萃取,收集有机相,水洗3遍,无水硫酸钠干燥,过滤,浓缩,柱层析纯化得标题化合物。Take 4.10g 4-bromo-1-methyl-1H-pyrazole-3-carboxylic acid (20mmol), 2.14g ammonium chloride (40mmol), 15.2g HATU (40mmol) and 6.98ml DIPEA (40mmol) in 250mL of eggplant 100ml of DMF was added to the flask, and the reaction was carried out at room temperature for 12h. After the reaction was completed, 500ml of ethyl acetate and 250ml of saturated aqueous sodium chloride were added for extraction. The organic phase was collected, washed three times with water, dried over anhydrous sodium sulfate, filtered, concentrated, and the column layer was The title compound was obtained by analytical purification.
LC-MS m/z:[M+H]+=204。LC-MS m/z: [M+H] + =204.
步骤2 1-甲基-3-氰基-4-溴-1H-吡唑的制备Step 2 Preparation of 1-methyl-3-cyano-4-bromo-1H-pyrazole
称取2.03g步骤1所得物1-甲基-3-氨基酰基-4-溴-1H-吡唑(10mmol)于250mL的茄形瓶中,加入100ml吡啶,0-4℃下,缓慢加入30ml三氯氧磷,常温下反应12h,反应结束后,加入饱和碳酸钠水溶液中和,再加入500mL乙酸乙酯和250mL饱和氯化钠水溶液萃取,收集有机相,水洗3遍,无水硫酸钠干燥,过滤,浓缩,柱层析纯化,得标题化合物。Weigh 2.03 g of 1-methyl-3-aminoacyl-4-bromo-1H-pyrazole (10 mmol) obtained in step 1 into a 250 mL eggplant flask, add 100 mL of pyridine, and slowly add 30 mL of pyridine at 0-4°C Phosphorus oxychloride was reacted at room temperature for 12 hours. After the reaction, saturated aqueous sodium carbonate solution was added to neutralize, and then 500 mL of ethyl acetate and 250 mL of saturated aqueous sodium chloride solution were added for extraction. The organic phase was collected, washed with water 3 times, and dried over anhydrous sodium sulfate. , filtered, concentrated, and purified by column chromatography to obtain the title compound.
LC-MS m/z:[M+H]+=186。LC-MS m/z: [M+H] + =186.
步骤3 4-(1-甲基-3-氰基-1H-吡唑-4-基)苯酚Step 3 4-(1-Methyl-3-cyano-1H-pyrazol-4-yl)phenol
以4-羟基苯硼酸和步骤2所得物1-甲基-3-氰基-4-溴-1H-吡唑为原料,同实施例1步骤1的方法制得标题化合物。Using 4-hydroxyphenylboronic acid and 1-methyl-3-cyano-4-bromo-1H-pyrazole obtained in step 2 as starting materials, the title compound was prepared in the same manner as in step 1 of Example 1.
LC-MS m/z:[M+H]+=200。LC-MS m/z: [M+H] + =200.
步骤4(2S)-2-(((4-(1-甲基-3-氰基-1H-吡唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Step 4(2S)-2-(((4-(1-methyl-3-cyano-1H-pyrazol-4-yl)phenyloxy)(((2R,3R,4R,5R)- 5-(2,4-Dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphorus Acyl)amino)isopropyl propionate
以步骤3所得物4-(1-甲基-3-氰基-1H-吡唑-4-基)苯酚、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。Using the resultant of step 3, 4-(1-methyl-3-cyano-1H-pyrazol-4-yl)phenol, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol and (2'R)-2'-deoxy-2'-fluoro-2'-methyluridine was used as the raw material, and the target compound was obtained by the same method as steps 2 and 3 of Example 1.
1H NMR(300MHz,DMSO-d6)δppm:11.49(s,1H),8.03-8.09(m,1H),7.61-7.62(m,2H),7.55-7.57(m,1H),7.33-7.34(m,2H),6.09-6.11(m,1H),6.04-6.07(m,1H),5.98-6.00(m,1H),5.54-5.56(m,1H),4.83-4.88(m,1H),4.38-4.40(m,1H),4.24-4.27(m,1H),4.01-4.03(m,1H),3.82-3.84(m,3H),3.81(m,2H),1.23-1.27(m,6H),1.15-1.15(m,6H)。 1 H NMR (300 MHz, DMSO-d 6 ) δppm: 11.49 (s, 1H), 8.03-8.09 (m, 1H), 7.61-7.62 (m, 2H), 7.55-7.57 (m, 1H), 7.33-7.34 (m,2H),6.09-6.11(m,1H),6.04-6.07(m,1H),5.98-6.00(m,1H),5.54-5.56(m,1H),4.83-4.88(m,1H) ,4.38-4.40(m,1H),4.24-4.27(m,1H),4.01-4.03(m,1H),3.82-3.84(m,3H),3.81(m,2H),1.23-1.27(m, 6H), 1.15-1.15 (m, 6H).
LC-MS m/z:[M+H]+=635。LC-MS m/z: [M+H] + =635.
实施例9(2S)-2-(((4-(吡啶-3-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 9(2S)-2-(((4-(pyridin-3-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo-3 ,4-Dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate isopropyl ester
步骤1 4-(吡啶-3-基)苯酚的制备Step 1 Preparation of 4-(pyridin-3-yl)phenol
在100mL单口瓶中加入1g 3-溴吡啶(6.4mmol)、0.96g对羟基苯硼酸(7.0mmol)、2.6g碳酸钾(21mmol)、0.45g PdCl2(PPh3)2(0.65mmol)、15mL水和30mL1,4-二氧六环,氮气保护下,80℃反应2h,反应结束后,冷却至室温,过滤,浓缩,加乙酸乙酯和水萃取,收集有机相,无水硫酸钠干燥,过滤,浓缩,柱层析纯化得标题化合物。In a 100mL single-neck flask, add 1g 3-bromopyridine (6.4mmol), 0.96g p-hydroxyphenylboronic acid (7.0mmol), 2.6g potassium carbonate (21mmol), 0.45g PdCl 2 (PPh 3 ) 2 (0.65mmol), 15mL Water and 30 mL of 1,4-dioxane were reacted at 80°C for 2 h under nitrogen protection. After the reaction was completed, cooled to room temperature, filtered, concentrated, extracted with ethyl acetate and water, the organic phase was collected, dried over anhydrous sodium sulfate, Filtration, concentration, and purification by column chromatography gave the title compound.
1HNMR((300MHz,DMSO-d6)δppm:9.67(s,1H),8.82(s,1H),8.49(d,1H),7.99(d,1H),7.65-7.54(m,2H),7.46-7.42(m,1H),6.88(d,2H)。 1 HNMR ((300MHz, DMSO-d6) δppm: 9.67(s, 1H), 8.82(s, 1H), 8.49(d, 1H), 7.99(d, 1H), 7.65-7.54(m, 2H), 7.46 -7.42(m, 1H), 6.88(d, 2H).
LC-MS m/z:[M+H]+=172。LC-MS m/z: [M+H] + =172.
步骤2(2S)-2-(((4-(吡啶-3-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Step 2(2S)-2-(((4-(pyridin-3-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo-3, Isopropyl 4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate
以步骤1所得物4-(吡啶-3-基)苯酚、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。4-(Pyridin-3-yl)phenol, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol and (2'R)-2'-deoxy-2' obtained in step 1 -Fluoro-2'-methyluridine was used as the raw material, and the target compound was obtained by the same method as steps 2 and 3 of Example 1.
1H NMR(300MHz,DMSO-d6)δppm:11.50(s,1H),8.87(s,1H),8.56(d,1H),8.05(d,1H),7.74(d,2H),7.59-7.46(m,2H),7.35(d,2H),6.20-6.00(m,2H),5.90(d,1H),5.58(d,1H),4.90-4.82(m,1H),4.43(m,1H),4.31(m,1H),4.05(m,1H),3.84-3.78(m,2H),1.28-1.20(m,6H),1.17-1.14(m,6H)。 1 H NMR (300MHz, DMSO-d 6 ) δppm: 11.50(s, 1H), 8.87(s, 1H), 8.56(d, 1H), 8.05(d, 1H), 7.74(d, 2H), 7.59- 7.46(m, 2H), 7.35(d, 2H), 6.20-6.00(m, 2H), 5.90(d, 1H), 5.58(d, 1H), 4.90-4.82(m, 1H), 4.43(m, 1H), 4.31 (m, 1H), 4.05 (m, 1H), 3.84-3.78 (m, 2H), 1.28-1.20 (m, 6H), 1.17-1.14 (m, 6H).
LC-MS m/z:[M+H]+=607。LC-MS m/z: [M+H] + =607.
实施例10(2S)-2-(((4-(5-氟吡啶-2-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 10(2S)-2-(((4-(5-fluoropyridin-2-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo Isopropyl substituted-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate
步骤1 4-(5-氟吡啶-2-基)苯酚的制备Step 1 Preparation of 4-(5-fluoropyridin-2-yl)phenol
以2-溴-5-氟吡啶和4-羟基苯硼酸为原料,同实施例9步骤1的方法制得标题化合物。Using 2-bromo-5-fluoropyridine and 4-hydroxybenzeneboronic acid as starting materials, the title compound was prepared in the same manner as in Example 9, step 1.
1HNMR((300MHz,CDCl3-d6)δppm:8.50(s,1H),7.82(d,2H),7.64(m,1H),7.46(m,1H),6.90(d,2H),7.53(s,1H)。 1 HNMR ((300MHz, CDCl3-d 6 ) δppm: 8.50(s, 1H), 7.82(d, 2H), 7.64(m, 1H), 7.46(m, 1H), 6.90(d, 2H), 7.53( s, 1H).
步骤2(2S)-2-(((4-(5-氟吡啶-2-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Step 2(2S)-2-(((4-(5-fluoropyridin-2-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo -3,4-Dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate isopropyl ester
以步骤1所得物4-(5-氟吡啶-2-基)苯酚、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。4-(5-Fluoropyridin-2-yl)phenol, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol and (2'R)-2'-deoxygenate obtained in step 1 -2'-Fluoro-2'-methyluridine was used as the raw material, and the target compound was obtained by the same method as steps 2 and 3 of Example 1.
1H NMR(300MHz,DMSO-d6)δppm:11.50(s,1H),8.64(d,1H),8.04(d,3H),7.80(m,1H),7.56(d,1H),7.33(d,2H),6.15-6.07(m,2H),5.84(d,1H),5.56(d,1H),4.90-4.83(m,1H),4.43(m,1H),4.27(m,1H),4.01(m,1H),3.88-3.78(m,2H),1.29-1.22(m,6H),1.16-1.14(m,6H)。 1 H NMR (300MHz, DMSO-d 6 ) δppm: 11.50(s, 1H), 8.64(d, 1H), 8.04(d, 3H), 7.80(m, 1H), 7.56(d, 1H), 7.33( d,2H),6.15-6.07(m,2H),5.84(d,1H),5.56(d,1H),4.90-4.83(m,1H),4.43(m,1H),4.27(m,1H) , 4.01 (m, 1H), 3.88-3.78 (m, 2H), 1.29-1.22 (m, 6H), 1.16-1.14 (m, 6H).
LC-MS m/z:[M+H]+=625。LC-MS m/z: [M+H] + =625.
实施例11(2S)-2-(((4-(2-氯吡啶-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 11(2S)-2-(((4-(2-chloropyridin-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo Isopropyl substituted-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate
步骤1 4–(2-氯吡啶-4-基)苯酚的制备Step 1 Preparation of 4-(2-chloropyridin-4-yl)phenol
以2-氯-4-溴吡啶和对羟基苯硼酸为原料,同实施例9步骤1的方法制得标题化合物。Using 2-chloro-4-bromopyridine and p-hydroxybenzeneboronic acid as starting materials, the title compound was prepared by the same method as in Example 9, step 1.
1HNMR((300MHz,CDCl3-d6)δppm:8.50(s,1H),7.82(d,2H),7.64(m,1H),7.46(m,1H),6.90(d,2H),7.53(s,1H)。 1 HNMR ((300MHz, CDCl3-d 6 ) δppm: 8.50(s, 1H), 7.82(d, 2H), 7.64(m, 1H), 7.46(m, 1H), 6.90(d, 2H), 7.53( s, 1H).
LC-MS m/z:[M+H]+=206。LC-MS m/z: [M+H] + =206.
步骤2(2S)-2-(((4-(2-氯吡啶-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Step 2(2S)-2-(((4-(2-chloropyridin-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo -3,4-Dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate isopropyl ester
以步骤1所得物4-(2-氯吡啶-4-基)苯酚、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。4-(2-Chloropyridin-4-yl)phenol, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol and (2'R)-2'-deoxygenate obtained in step 1 -2'-Fluoro-2'-methyluridine was used as the raw material, and the target compound was obtained by the same method as steps 2 and 3 of Example 1.
1H NMR(300MHz,DMSO-d6)δppm:11.51(s,1H),8.46(d,1H),7.92(m,3H),7.42(d,1H),7.62-7.56(m,1H),7.37(d,2H),6.20-5.99(m,2H),5.86(d,1H),5.54(d,1H),4.88-4.81(m,1H),4.39(m,1H),4.27(m,1H),4.04(m,1H),3.89-3.81(m,2H),1.29-1.22(m,6H),1.16-1.14(m,6H)。 1 H NMR (300MHz, DMSO-d 6 ) δppm: 11.51(s, 1H), 8.46(d, 1H), 7.92(m, 3H), 7.42(d, 1H), 7.62-7.56(m, 1H), 7.37(d, 2H), 6.20-5.99(m, 2H), 5.86(d, 1H), 5.54(d, 1H), 4.88-4.81(m, 1H), 4.39(m, 1H), 4.27(m, 1H), 4.04 (m, 1H), 3.89-3.81 (m, 2H), 1.29-1.22 (m, 6H), 1.16-1.14 (m, 6H).
LC-MS m/z:[M+H]+=641。LC-MS m/z: [M+H] + =641.
实施例12(2S)-2-(((4-(5-甲基噻唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 12(2S)-2-(((4-(5-methylthiazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-di Isopropyl oxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propanoate ester
步骤1 4-溴-5甲基噻唑的制备Step 1 Preparation of 4-bromo-5-methylthiazole
在100mL单口瓶中加入1g 5-甲基噻唑(10mmol),1.8g N-溴代丁二酰亚胺(10mmol)和30mL二氯甲烷,室温反应12h,反应结束后,浓缩,柱层析纯化得标题化合物。1g 5-methylthiazole (10mmol), 1.8g N-bromosuccinimide (10mmol) and 30mL dichloromethane were added in a 100mL single-neck flask, and the reaction was carried out at room temperature for 12h. After the reaction was completed, it was concentrated and purified by column chromatography. The title compound was obtained.
步骤2 4-(5-甲基噻唑-4-基)苯酚的制备Step 2 Preparation of 4-(5-methylthiazol-4-yl)phenol
以步骤1所得物4-溴-5-甲基噻唑和对羟基苯硼酸为原料,同实施例9步骤1的方法制得标题化合物。Using 4-bromo-5-methylthiazole obtained in step 1 and p-hydroxyphenylboronic acid as starting materials, the title compound was prepared in the same manner as in step 1 of Example 9.
1HNMR((300MHz,CDCl3-d6)δppm:8.65(s,1H),7.52(d,2H),6.88(d,2H),2.57(s,3H)。 1 H NMR ((300 MHz, CDCl 3 -d6) δ ppm: 8.65 (s, 1H), 7.52 (d, 2H), 6.88 (d, 2H), 2.57 (s, 3H).
步骤3(2S)-2-(((4-(5-甲基噻唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Step 3(2S)-2-(((4-(5-Methylthiazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo Isopropyl substituted-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate
以步骤2所得物4-(5-甲基噻唑-4-基)苯酚、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。4-(5-methylthiazol-4-yl)phenol, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol and (2'R)-2'- Deoxy-2'-fluoro-2'-methyluridine was used as a raw material, and the target compound was obtained by the same method as steps 2 and 3 of Example 1.
1H NMR(300MHz,DMSO-d6)δppm:11.49(s,1H),7.89(d,2H),7.58(m,2H),7.33(d,2H),6.18-5.98(m,2H),5.86(d,1H),5.57(d,1H),4.88-4.81(m,1H),4.39(m,1H),4.27(m,1H),4.04(m,1H),3.89-3.81(m,2H),2.50(s,3H),1.29-1.22(m,6H),1.16-1.14(m,6H)。 1 H NMR (300MHz, DMSO-d 6 ) δppm: 11.49(s, 1H), 7.89(d, 2H), 7.58(m, 2H), 7.33(d, 2H), 6.18-5.98(m, 2H), 5.86(d,1H),5.57(d,1H),4.88-4.81(m,1H),4.39(m,1H),4.27(m,1H),4.04(m,1H),3.89-3.81(m, 2H), 2.50 (s, 3H), 1.29-1.22 (m, 6H), 1.16-1.14 (m, 6H).
LC-MS m/z:[M+H]+=627。LC-MS m/z: [M+H] + =627.
实施例13(2S)-2-(((1-甲基-1H-吲唑-5-基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 13(2S)-2-(((1-methyl-1H-indazol-5-yloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo -3,4-Dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate isopropyl ester
以5-羟基-1-甲基-1H-吲唑、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。With 5-hydroxy-1-methyl-1H-indazole, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol and (2'R)-2'-deoxy-2'- Fluoro-2'-methyluridine was used as the raw material, and the target compound was prepared in the same manner as in steps 2 and 3 of Example 1.
1H NMR(300MHz,DMSO-d6)δppm:11.49(s,1H),7.99(s,1H),7.63(d,1H),7.54(d,2H),7.26(d,1H),6.18-5.98(m,2H),5.86(d,1H),5.57(d,1H),4.88-4.81(m,1H),4.39(m,1H),4.27(m,1H),4.04(m,1H),3.99(s,3H),3.89-3.81(m,2H),1.29-1.22(m,6H),1.16-1.14(m,6H)。1H NMR (300MHz, DMSO-d 6 ) δppm: 11.49(s, 1H), 7.99(s, 1H), 7.63(d, 1H), 7.54(d, 2H), 7.26(d, 1H), 6.18-5.98 (m, 2H), 5.86(d, 1H), 5.57(d, 1H), 4.88-4.81(m, 1H), 4.39(m, 1H), 4.27(m, 1H), 4.04(m, 1H), 3.99(s, 3H), 3.89-3.81(m, 2H), 1.29-1.22(m, 6H), 1.16-1.14(m, 6H).
LC-MS m/z:[M+H]+=584。LC-MS m/z: [M+H] + =584.
实施例14(2S)-2-(((1-甲基-1H-吲哚-5-基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 14(2S)-2-(((1-methyl-1H-indol-5-yloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo -3,4-Dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate isopropyl ester
步骤1 5-甲氧基-1-甲基-1H-吲哚的制备Step 1 Preparation of 5-methoxy-1-methyl-1H-indole
称取5g 5-甲氧基-1H-吲哚(0.034mol)于500mL的单口瓶中,加入200mL DMF溶解,0-4℃下加入2.7g氢化钠(0.068mol),加毕搅拌0.5h后,滴加4.4g硫酸二甲酯(0.035mol),加毕,室温反应0.5h,反应结束后,冰浴下加水淬灭,过滤,干燥,得到标题化合物。Weigh 5g of 5-methoxy-1H-indole (0.034mol) into a 500mL single-neck flask, add 200mL of DMF to dissolve, add 2.7g of sodium hydride (0.068mol) at 0-4°C, and stir for 0.5h after the addition is complete. , 4.4g of dimethyl sulfate (0.035mol) was added dropwise, the addition was completed, and the reaction was carried out at room temperature for 0.5h. After the reaction was completed, water was added under an ice bath to quench, filtered, and dried to obtain the title compound.
步骤2 5-羟基-1-甲基-1H-吲哚的制备Step 2 Preparation of 5-hydroxy-1-methyl-1H-indole
称取0.7g步骤1所得物5-甲氧基-1-甲基-1H-吲哚(4.34mmol)于100mL单口瓶中,加入20mL二氯甲烷溶解,-30℃下滴加0.52g三溴化硼(21mmol),加毕,室温搅拌12h,反应结束后,冰浴下加二氯甲烷稀释,加水淬灭,萃取,干燥,浓缩,柱层析纯化得标题化合物。Weigh 0.7 g of 5-methoxy-1-methyl-1H-indole (4.34 mmol) obtained in step 1 into a 100 mL single-neck flask, add 20 mL of dichloromethane to dissolve, and dropwise add 0.52 g of tribromide at -30°C Boronide (21 mmol) was added, and stirred at room temperature for 12 h. After the reaction was completed, dichloromethane was added to dilute in an ice bath, quenched by adding water, extracted, dried, concentrated, and purified by column chromatography to obtain the title compound.
步骤3(2S)-2-(((1-甲基-1H-吲哚-5-基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Step 3(2S)-2-(((1-Methyl-1H-indol-5-yloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo- Isopropyl 3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate
以步骤2所得物5-羟基-1-甲基-1H-吲哚、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。5-Hydroxy-1-methyl-1H-indole, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol and (2'R)-2'-deoxygenated from step 2 -2'-Fluoro-2'-methyluridine was used as the raw material, and the target compound was obtained by the same method as steps 2 and 3 of Example 1.
1H NMR(300MHz,DMSO-d6)δppm:11.49(s,1H),8.37(d,1H),7.55-7.34(m,3H),7.02(d,1H),6.36(d,1H),6.04-5.85(m,3H),5.48(d,1H),4.88-4.81(m,1H),4.35(m,1H),4.22(m,1H),4.043.78(m,1H),3.89-3.31(m,5H),1.29-1.22(m,6H),1.16-1.14(m,6H)。 1 H NMR (300MHz, DMSO-d 6 ) δppm: 11.49(s, 1H), 8.37(d, 1H), 7.55-7.34(m, 3H), 7.02(d, 1H), 6.36(d, 1H), 6.04-5.85(m, 3H), 5.48(d, 1H), 4.88-4.81(m, 1H), 4.35(m, 1H), 4.22(m, 1H), 4.043.78(m, 1H), 3.89- 3.31 (m, 5H), 1.29-1.22 (m, 6H), 1.16-1.14 (m, 6H).
LC-MS m/z:[M+H]+=583。LC-MS m/z: [M+H] + =583.
实施例15(2S)-2-(((2-氟-4-(1-甲基-1H-吡唑-3-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 15(2S)-2-(((2-Fluoro-4-(1-methyl-1H-pyrazol-3-yl)phenyloxy)(((2R,3R,4R,5R)- 5-(2,4-Dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphorus Acyl)amino)isopropyl propionate
步骤1 4-(1-甲基-1H-吡唑-3-基)苯酚的制备Step 1 Preparation of 4-(1-methyl-1H-pyrazol-3-yl)phenol
以1-甲基-3-溴-1H-吡唑和4-羟基苯硼酸为原料,同实施例1步骤1的方法制得标题化合物。Using 1-methyl-3-bromo-1H-pyrazole and 4-hydroxybenzeneboronic acid as starting materials, the title compound was obtained by the same method as in Example 1, step 1.
LC-MS m/z:[M+H]+=175。LC-MS m/z: [M+H] + =175.
步骤2 2-氟-4-(1-甲基-1H-吡唑-3-基)苯酚的制备Step 2 Preparation of 2-fluoro-4-(1-methyl-1H-pyrazol-3-yl)phenol
称取1.71g步骤1所得物4-(1-甲基-1H-吡唑-3-基)苯酚、3.54g 1-氯甲基-4-氟-1,4-二氮双环[2.2.2.]辛烷双氟硼酸盐(selectfluor)于100mL的茄形瓶中,加入50mL三氟乙酸,60℃反应12h,反应结束后,调节反应液pH值至7左右,加入100mL乙酸乙酯和50mL饱和氯化钠水溶液萃取,水洗3遍,收集有机相,无水硫酸钠来干燥,过滤,浓缩,柱层析纯化得标题化合物。Weigh 1.71g of the obtained 4-(1-methyl-1H-pyrazol-3-yl)phenol in step 1, 3.54g of 1-chloromethyl-4-fluoro-1,4-diazabicyclo[2.2.2 .]Octane difluoroborate (selectfluor) was placed in a 100 mL eggplant-shaped flask, 50 mL of trifluoroacetic acid was added, and the reaction was carried out at 60°C for 12 h. After the reaction, the pH value of the reaction solution was adjusted to about 7, and 100 mL of ethyl acetate and ethyl acetate were added. Extracted with 50 mL of saturated aqueous sodium chloride solution, washed three times with water, collected the organic phase, dried over anhydrous sodium sulfate, filtered, concentrated, and purified by column chromatography to obtain the title compound.
LC-MS m/z:[M+H]+=193。LC-MS m/z: [M+H] + =193.
步骤3(2S)-2-(((2-氟-4-(1-甲基-1H-吡唑-3-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯的制备Step 3(2S)-2-(((2-Fluoro-4-(1-methyl-1H-pyrazol-3-yl)phenyloxy)(((2R,3R,4R,5R)-5 -(2,4-Dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl ) preparation of amino) isopropyl propionate
以步骤2所得物2-氟-4-(1-甲基-1H-吡唑-3-基)苯酚、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。Using the resultant of step 2, 2-fluoro-4-(1-methyl-1H-pyrazol-3-yl)phenol, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol and ( 2'R)-2'-deoxy-2'-fluoro-2'-methyluridine was used as a raw material, and the target compound was obtained by the same method as steps 2 and 3 of Example 1.
1H NMR(300MHz,DMSO-d6)δppm:11.50(s,1H),7.70-7.73(m,2H),7.60-7.66(m,2H),7.44-7.47(m,1H),6.67(s,1H),6.21-6.24(m,1H),6.19-6.20(m,1H),5.86-5.89(m,1H),5.56-5.59(m,1H),4.81-4.90(m,1H),4.38-4.42(m,1H),4.27-4.28(m,1H),4.01-4.06(m,1H),3.81-3.84(m,3H),3.78-3.80(m,2H),1.27-1.29(m,6H),1.14-1.16(m,6H)。 1 H NMR (300MHz, DMSO-d 6 ) δppm: 11.50 (s, 1H), 7.70-7.73 (m, 2H), 7.60-7.66 (m, 2H), 7.44-7.47 (m, 1H), 6.67 (s) ,1H),6.21-6.24(m,1H),6.19-6.20(m,1H),5.86-5.89(m,1H),5.56-5.59(m,1H),4.81-4.90(m,1H),4.38 -4.42(m, 1H), 4.27-4.28(m, 1H), 4.01-4.06(m, 1H), 3.81-3.84(m, 3H), 3.78-3.80(m, 2H), 1.27-1.29(m, 6H), 1.14-1.16 (m, 6H).
LC-MS m/z:[M+H]+=628。LC-MS m/z: [M+H] + =628.
实施例16(2S)-2-(((4-(5-氰基噻唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 16(2S)-2-(((4-(5-cyanothiazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-di Isopropyl oxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propanoate ester
步骤1 4-溴-5-甲基噻唑的制备Step 1 Preparation of 4-bromo-5-methylthiazole
称取15g 5-甲基噻唑于500mL的单口瓶中,加入250mL二氯甲烷溶解,分批加入27gNBS,加毕,室温反应12h,反应结束后,浓缩,柱层析纯化得标题化合物。Weigh 15g of 5-methylthiazole into a 500mL single-necked bottle, add 250mL of dichloromethane to dissolve, add 27g of NBS in batches, complete the addition, and react at room temperature for 12h. After the reaction is completed, concentrate and purify by column chromatography to obtain the title compound.
步骤2 4-溴-5-溴甲基噻唑的制备Step 2 Preparation of 4-bromo-5-bromomethylthiazole
称取5g步骤1所得物4-溴-5-甲基噻唑和5g NBS于250mL的单口瓶中,加入80mL四氯化碳溶解,回流反应2h,反应结束后,冷却至室温,浓缩,柱层析纯化得标题化合物。Weigh 5g of the obtained 4-bromo-5-methylthiazole in step 1 and 5g of NBS into a 250mL single-necked bottle, add 80mL of carbon tetrachloride to dissolve, reflux for 2h, after the reaction is completed, cool to room temperature, concentrate, column layer The title compound was obtained by analytical purification.
步骤3 4-溴-5-羟甲基噻唑的制备Step 3 Preparation of 4-bromo-5-hydroxymethylthiazole
称取2g步骤2所得物4-溴-5-溴甲基噻唑,3.2g碳酸钾于100ml的单口瓶中,加入50mL 1,4-二氧六环和20mL水中溶解,90℃反应1h,反应结束后,浓缩,柱层析纯化得标题化合物。Weigh 2g of the obtained 4-bromo-5-bromomethylthiazole in step 2, 3.2g of potassium carbonate in a 100ml single-neck bottle, add 50mL of 1,4-dioxane and 20mL of water to dissolve, react at 90°C for 1h, the reaction After completion, concentration and column chromatography were performed to obtain the title compound.
步骤4 4-溴-5-甲酰基噻唑的制备Step 4 Preparation of 4-bromo-5-formylthiazole
称取1g步骤3所得物4-溴-5-羟甲基噻唑于100mL的单口瓶中,加入30mL二氯甲烷溶解,分批加入1.5g氯铬酸吡啶(PCC),加毕,室温反应3h,反应结束后,浓缩,柱层析纯化得到标题化合物。Weigh 1 g of 4-bromo-5-hydroxymethylthiazole obtained in step 3 into a 100 mL single-neck flask, add 30 mL of dichloromethane to dissolve, add 1.5 g of pyridine chlorochromate (PCC) in batches, complete the addition, and react at room temperature for 3 h , after the reaction was completed, concentrated and purified by column chromatography to obtain the title compound.
步骤5 4-(5-甲酰基噻唑-4-基)苯酚的制备Step 5 Preparation of 4-(5-formylthiazol-4-yl)phenol
以步骤4所得物4-溴-5-甲酰基噻唑和4-羟基苯硼酸为原料,同实施例9步骤1的方法得到标题化合物。Using 4-bromo-5-formylthiazole obtained in step 4 and 4-hydroxyphenylboronic acid as raw materials, the title compound was obtained by the same method as step 1 in Example 9.
步骤6 4-(噻唑-5-甲醛肟-4-基)苯酚Step 6 4-(thiazol-5-carbaldehyde oxime-4-yl)phenol
称取1g步骤5所得物4-(5-甲酰基噻唑-4-基)苯酚、0.51g盐酸羟胺和0.6g乙酸钠于100mL的单口瓶中,加入30mL乙醇和5mL水溶解,回流反应1h,反应结束后,冷却至室温,浓缩,柱层析纯化得标题化合物。Weigh 1 g of 4-(5-formylthiazol-4-yl)phenol obtained in step 5, 0.51 g of hydroxylamine hydrochloride and 0.6 g of sodium acetate into a 100-mL single-neck flask, add 30 mL of ethanol and 5 mL of water to dissolve, and reflux for 1 h. After the reaction was completed, it was cooled to room temperature, concentrated, and purified by column chromatography to obtain the title compound.
步骤7 4-(5-氰基噻唑-4-基)苯酚的制备Step 7 Preparation of 4-(5-cyanothiazol-4-yl)phenol
称取1g步骤6所得物4-(噻唑-5-甲醛肟-4-基)苯酚于100ml的单口瓶中,加入10mLTHF溶解后,加入8mL吡啶和8mL三氟乙酸酐,室温反应12h,反应结束后,冷却至室温,浓缩,柱层析纯化得标题化合物。Weigh 1g of 4-(thiazole-5-carbaldehyde oxime-4-yl)phenol obtained in step 6 into a 100ml single-necked flask, add 10mL of THF to dissolve, add 8mL of pyridine and 8mL of trifluoroacetic anhydride, and react at room temperature for 12h, the reaction ends After that, it was cooled to room temperature, concentrated, and purified by column chromatography to obtain the title compound.
步骤8(2S)-2-(((4-(5-氰基噻唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯的制备Step 8(2S)-2-(((4-(5-Cyanothiazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo Isopropyl substituted-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate preparation
以步骤7所得物4-(5-氰基噻唑-4-基)苯酚、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。4-(5-Cyanothiazol-4-yl)phenol obtained in step 7, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol and (2'R)-2'- Deoxy-2'-fluoro-2'-methyluridine was used as a raw material, and the target compound was obtained by the same method as steps 2 and 3 of Example 1.
1H NMR(300MHz,DMSO-d6)δppm:11.49(s,1H),8.74(d,1H),8.05(d,2H),7.02(d,1H),7.54(d,1H),7.41(d,2H),6.22(m,1H),6.17(m,1H),5.85(m,1H),4.88-4.81(m,1H),4.35(m,1H),4.22(m,1H),4.043.78(m,1H),3.89-3.31(m,2H),1.29-1.22(m,6H),1.16-1.14(m,6H)。 1 H NMR (300MHz, DMSO-d 6 ) δppm: 11.49(s, 1H), 8.74(d, 1H), 8.05(d, 2H), 7.02(d, 1H), 7.54(d, 1H), 7.41( d,2H),6.22(m,1H),6.17(m,1H),5.85(m,1H),4.88-4.81(m,1H),4.35(m,1H),4.22(m,1H),4.043 .78(m,1H), 3.89-3.31(m,2H), 1.29-1.22(m,6H), 1.16-1.14(m,6H).
LC-MS m/z:[M+H]+=638。LC-MS m/z: [M+H] + =638.
实施例17(2S)-2-(((3-甲基苯并[d]异噁唑-6-基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 17(2S)-2-(((3-methylbenzo[d]isoxazol-6-yloxy)(((2R,3R,4R,5R)-5-(2,4- Dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)isopropionic acid Propyl ester
步骤1(Z)-1-(2,4-二羟基苯基)乙酮肟的制备Step 1 (Z)-Preparation of 1-(2,4-dihydroxyphenyl)ethanone oxime
称取5g 1-(2,4-二羟基苯基)乙酮于100mL的单口瓶中,加入50mL乙醇溶解后,加入2.3g盐酸羟胺,2.7g乙酸钠和5mL水,回流反应2h,反应结束后,浓缩,柱层析纯化得标题化合物。Weigh 5g of 1-(2,4-dihydroxyphenyl)ethanone into a 100mL single-necked bottle, add 50mL of ethanol to dissolve, add 2.3g of hydroxylamine hydrochloride, 2.7g of sodium acetate and 5mL of water, reflux for 2h, and the reaction ends After that, it was concentrated and purified by column chromatography to obtain the title compound.
步骤2 6-羟基-3-甲基苯并[d]异噁唑的制备Step 2 Preparation of 6-hydroxy-3-methylbenzo[d]isoxazole
称取1g二氯二氰基苯醌(DDQ)、1.2g PPh3于100mL的单口瓶中,加入30mL二氯甲烷搅拌溶解后,滴加30mL溶解有0.5g步骤1所得物(Z)-1-(2,4-二羟基苯基)乙酮肟的二氯甲烷溶液,室温反应0.5h,反应结束后,浓缩,柱层析纯化得标题化合物。Weigh 1 g of dichlorodicyanobenzoquinone (DDQ) and 1.2 g of PPh 3 in a 100 mL single-necked bottle, add 30 mL of dichloromethane, stir to dissolve, and add dropwise 30 mL to dissolve 0.5 g of the result of step 1 (Z)-1 The solution of -(2,4-dihydroxyphenyl)ethanone oxime in dichloromethane was reacted at room temperature for 0.5 h, after the reaction was completed, concentrated and purified by column chromatography to obtain the title compound.
步骤3(2S)-2-(((3-甲基苯并[d]异噁唑-6-基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯的制备Step 3(2S)-2-(((3-methylbenzo[d]isoxazol-6-yloxy)(((2R,3R,4R,5R)-5-(2,4-di Isopropyl oxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propanoate Preparation of esters
以步骤2所得物6-羟基-3-甲基苯并[d]异噁唑、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。6-Hydroxy-3-methylbenzo[d]isoxazole, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol and (2'R)-2 obtained in step 2 '-Deoxy-2'-fluoro-2'-methyluridine was used as the raw material, and the target compound was obtained by the same method as steps 2 and 3 of Example 1.
1H NMR(500MHz,DMSO-d6)δppm:11.49(s,1H),7.63(d,1H),7.53(d,2H),7.19(d,1H),6.10-6.07(m,2H),5.84(m,1H),5.54(m,1H),4.88-4.81(m,1H),4.35(m,1H),4.22(m,1H),4.043.78(m,1H),3.89-3.31(m,2H),2.56(s,3H),1.29-1.22(m,6H),1.16-1.14(m,6H)。 1 H NMR (500MHz, DMSO-d 6 ) δppm: 11.49(s, 1H), 7.63(d, 1H), 7.53(d, 2H), 7.19(d, 1H), 6.10-6.07(m, 2H), 5.84(m,1H),5.54(m,1H),4.88-4.81(m,1H),4.35(m,1H),4.22(m,1H),4.043.78(m,1H),3.89-3.31( m, 2H), 2.56 (s, 3H), 1.29-1.22 (m, 6H), 1.16-1.14 (m, 6H).
LC-MS m/z:[M+H]+=585。LC-MS m/z: [M+H] + =585.
实施例18(2S)-2-(((4-(5-氯噻唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 18(2S)-2-(((4-(5-chlorothiazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo Isopropyl substituted-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate
步骤1 4-(噻唑-4-基)苯酚的制备Step 1 Preparation of 4-(thiazol-4-yl)phenol
以4-溴噻唑和4-羟基苯硼酸为原料,同实施例1步骤1的方法制得标题化合物。Using 4-bromothiazole and 4-hydroxybenzeneboronic acid as starting materials, the title compound was obtained by the same method as in Example 1, step 1.
LC-MS m/z:[M+H]+=178。LC-MS m/z: [M+H] + =178.
步骤2 4-(5-氯噻唑-4-基)苯酚的制备Step 2 Preparation of 4-(5-chlorothiazol-4-yl)phenol
称取1g步骤1所得物4-(噻唑-4-基)苯酚于100mL的单口瓶中,加入50mL二氯甲烷溶解后,加入0.75g NCS,加毕,室温反应12h,反应结束后,浓缩,柱层析纯化得标题化合物。Weigh 1 g of 4-(thiazol-4-yl)phenol obtained in step 1 into a 100 mL single-necked flask, add 50 mL of dichloromethane to dissolve, add 0.75 g of NCS, complete the addition, and react at room temperature for 12 h. After the reaction is completed, concentrate, Column chromatography gave the title compound.
步骤3(2S)-2-(((4-(5-氯噻唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯的制备Step 3(2S)-2-(((4-(5-chlorothiazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo -3,4-Dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)isopropyl propionate preparation
以步骤2所得物4-(5-氯噻唑-4-基)苯酚、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。4-(5-Chlorothiazol-4-yl)phenol, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol and (2'R)-2'-deoxygenate obtained in step 2 -2'-Fluoro-2'-methyluridine was used as the raw material, and the target compound was obtained by the same method as steps 2 and 3 of Example 1.
1H NMR(300MHz,DMSO-d6)δppm:11.51(s,1H),9.13(s,1H),7.92(d,2H),7.57(d,1H),7.35(d,2H),6.17-6.07(m,2H),5.85(m,1H),5.56(m,1H)4.88-4.81(m,1H),4.35(m,1H),4.22(m,1H),4.043.78(m,1H),3.89-3.31(m,2H),1.29-1.22(m,6H),1.16-1.14(m,6H)。 1 H NMR (300MHz, DMSO-d 6 ) δppm: 11.51(s, 1H), 9.13(s, 1H), 7.92(d, 2H), 7.57(d, 1H), 7.35(d, 2H), 6.17- 6.07(m, 2H), 5.85(m, 1H), 5.56(m, 1H) 4.88-4.81(m, 1H), 4.35(m, 1H), 4.22(m, 1H), 4.043.78(m, 1H) ), 3.89-3.31 (m, 2H), 1.29-1.22 (m, 6H), 1.16-1.14 (m, 6H).
LC-MS m/z:[M+H]+=647。LC-MS m/z: [M+H] + =647.
实施例19(2S)-2-(((4-(3,5-二甲基异噁唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 19(2S)-2-(((4-(3,5-dimethylisoxazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-( 2,4-Dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino ) isopropyl propionate
步骤1 4-(3,5-二甲基异噁唑-4-基)苯酚的制备Step 1 Preparation of 4-(3,5-dimethylisoxazol-4-yl)phenol
以4-溴-3,5-二甲基异噁唑和对羟基苯硼酸为原料,同实施例1步骤1的方法制得标题化合物。Using 4-bromo-3,5-dimethylisoxazole and p-hydroxyphenylboronic acid as starting materials, the title compound was obtained by the same method as Example 1, step 1.
步骤2(2S)-2-(((4-(3,5-二甲基异噁唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Step 2(2S)-2-(((4-(3,5-Dimethylisoxazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2 ,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino) isopropyl propionate
以步骤1所得物4-(3,5-二甲基异噁唑-4-基)苯酚、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。4-(3,5-Dimethylisoxazol-4-yl)phenol, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol and (2'R) obtained in step 1 )-2'-deoxy-2'-fluoro-2'-methyluridine was used as the raw material, and the target compound was obtained by the same methods as steps 2 and 3 of Example 1.
1H NMR(500MHz,DMSO-d6)δppm:11.49(s,1H),7.57(s,1H),7.39(d,2H),7.31(d,2H),6.00-6.11(m,2H),5.85(s,1H),5.51(m,1H),4.83-4.89(m,1H),4.39(m,1H),4.27(m,1H),4.02(m,1H),3.83-3.87(m,2H),2.38(s,3H),2.21(s,3H),1.23-1.28(m,6H),1.14-1.16(m,6H)。 1 H NMR (500MHz, DMSO-d 6 ) δppm: 11.49(s, 1H), 7.57(s, 1H), 7.39(d, 2H), 7.31(d, 2H), 6.00-6.11(m, 2H), 5.85(s, 1H), 5.51(m, 1H), 4.83-4.89(m, 1H), 4.39(m, 1H), 4.27(m, 1H), 4.02(m, 1H), 3.83-3.87(m, 2H), 2.38(s, 3H), 2.21(s, 3H), 1.23-1.28(m, 6H), 1.14-1.16(m, 6H).
LC-MS m/z:[M+H]+=625。LC-MS m/z: [M+H] + =625.
实施例20(2S)-2-(((4-(4-氯-1-甲基-1H-吡唑-3-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 20(2S)-2-(((4-(4-Chloro-1-methyl-1H-pyrazol-3-yl)phenyloxy)(((2R,3R,4R,5R)- 5-(2,4-Dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphorus Acyl)amino)isopropyl propionate
步骤1 4-(4-氯-1-甲基-1H-吡唑-3-基)苯酚Step 1 4-(4-Chloro-1-methyl-1H-pyrazol-3-yl)phenol
称取0.5g实施例15步骤1制备的4-(1-甲基-1H-吡唑-3-基)苯酚(2.9mmol)、0.385gNCS(2.9mmol)于50mL的茄形瓶中,加入5mL THF和5mL CH3CN,50℃反应3h,反应结束后,加入100mL乙酸乙酯和50mL饱和氯化钠水溶液萃取,水洗3遍,然后收集有机相,无水硫酸钠来干燥,过滤,浓缩,柱层析纯化得标题化合物。Weigh 0.5 g of 4-(1-methyl-1H-pyrazol-3-yl)phenol (2.9 mmol) and 0.385 g of NCS (2.9 mmol) prepared in step 1 of Example 15 into a 50 mL eggplant-shaped flask, add 5 mL THF and 5 mL of CH 3 CN were reacted at 50°C for 3 h. After the reaction was completed, 100 mL of ethyl acetate and 50 mL of saturated aqueous sodium chloride were added for extraction, washed three times with water, and then the organic phase was collected, dried over anhydrous sodium sulfate, filtered, and concentrated. Column chromatography gave the title compound.
LC-MS m/z:[M+H]+=209。LC-MS m/z: [M+H] + =209.
步骤2(2S)-2-(((4-(4-氯-1-甲基-1H-吡唑-3-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯的制备Step 2(2S)-2-(((4-(4-Chloro-1-methyl-1H-pyrazol-3-yl)phenyloxy)(((2R,3R,4R,5R)-5 -(2,4-Dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl ) preparation of amino) isopropyl propionate
以步骤1所得物4-(4-氯-1-甲基-1H-吡唑-3-基)苯酚、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。Using the resultant of step 1, 4-(4-chloro-1-methyl-1H-pyrazol-3-yl)phenol, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol and ( 2'R)-2'-deoxy-2'-fluoro-2'-methyluridine was used as a raw material, and the target compound was obtained by the same method as steps 2 and 3 of Example 1.
1H NMR(300MHz,DMSO-d6)δppm:11.49(s,1H),8.02(s,1H),7.78-7.80(m,2H),7.56-7.57(m,1H),7.29-7.31(m,2H),6.00-6.09(m,2H),5.82-5.84(m,1H),5.57-5.58(m,1H),4.83-4.88(m,1H),4.37-4.40(m,1H),4.24-4.26(m,1H),4.00-4.03(m,1H),3.80-3.86(m,5H),1.23-1.29(m,6H),1.14-1.15(m,6H)。 1 H NMR (300MHz, DMSO-d 6 ) δppm: 11.49(s, 1H), 8.02(s, 1H), 7.78-7.80(m, 2H), 7.56-7.57(m, 1H), 7.29-7.31(m ,2H),6.00-6.09(m,2H),5.82-5.84(m,1H),5.57-5.58(m,1H),4.83-4.88(m,1H),4.37-4.40(m,1H),4.24 -4.26(m, 1H), 4.00-4.03(m, 1H), 3.80-3.86(m, 5H), 1.23-1.29(m, 6H), 1.14-1.15(m, 6H).
LC-MS m/z:[M+H]+=644。LC-MS m/z: [M+H] + =644.
实施例21(2S)-2-(((4-(5-甲基噁唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 21(2S)-2-(((4-(5-methyloxazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4- Dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)isopropionic acid Propyl ester
步骤1 2-溴-1-(4-甲氧基苯基)-1-丙酮的制备Step 1 Preparation of 2-bromo-1-(4-methoxyphenyl)-1-propanone
称取1g 1-(4-甲氧基苯基)-1-丙酮(6.1mmol)、1.2g对甲苯磺酸一水合物(6.1mmol)和1.1g NBS(6.1mmol)于100mL的单口瓶中,加入30mL乙腈溶解,回流反应4h,反应结束后,浓缩,柱层析纯化得标题化合物。Weigh 1g 1-(4-methoxyphenyl)-1-propanone (6.1mmol), 1.2g p-toluenesulfonic acid monohydrate (6.1mmol) and 1.1g NBS (6.1mmol) in a 100mL single-neck flask , 30 mL of acetonitrile was added to dissolve, and the reaction was refluxed for 4 h. After the reaction was completed, the mixture was concentrated and purified by column chromatography to obtain the title compound.
步骤2 4-(4-甲氧基苯基)-5-甲基噁唑的制备Step 2 Preparation of 4-(4-methoxyphenyl)-5-methyloxazole
称取1g步骤1所得物2-溴-1-(4-甲氧基苯基)-1-丙酮(4.4mmol)于100mL的单口瓶中,加入2mL甲酰胺和20mL DMF,回流反应2h。反应结束后,浓缩,将浓缩液倒入冰水中,乙酸乙酯萃取,干燥,过滤,浓缩,柱层析纯化得标题化合物。Weigh 1 g of 2-bromo-1-(4-methoxyphenyl)-1-propanone (4.4 mmol) obtained in step 1 into a 100 mL single-neck flask, add 2 mL of formamide and 20 mL of DMF, and react under reflux for 2 h. After the reaction, concentrated, poured the concentrated solution into ice water, extracted with ethyl acetate, dried, filtered, concentrated, and purified by column chromatography to obtain the title compound.
步骤3 4-(5-甲基噁唑-4-基)苯酚的制备Step 3 Preparation of 4-(5-methyloxazol-4-yl)phenol
称取0.4g步骤2所得物4-(4-甲氧基苯基)-5-甲基噁唑(1.7mmol)于100mL的单口瓶中,加入20mL二氯甲烷溶解,冰浴下滴加8mL溶解有8mmol三溴化硼的二氯甲烷溶液,加毕,室温反应12h,原料结束后,冰浴下加水淬灭,二氯甲烷萃取,干燥,过滤,浓缩,柱层析纯化得标题化合物。Weigh 0.4 g of the obtained 4-(4-methoxyphenyl)-5-methyloxazole (1.7 mmol) in step 2 into a 100 mL single-neck flask, add 20 mL of dichloromethane to dissolve, and add 8 mL dropwise under an ice bath Dissolve 8 mmol of boron tribromide in dichloromethane solution, complete the addition, and react at room temperature for 12 h. After the completion of the raw materials, add water under ice bath to quench, extract with dichloromethane, dry, filter, concentrate, and purify by column chromatography to obtain the title compound.
步骤4(2S)-2-(((4-(5-甲基噁唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯的制备Step 4(2S)-2-(((4-(5-methyloxazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-di Isopropyl oxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propanoate Preparation of esters
以步骤3所得物4-(5-甲基噁唑-4-基)苯酚、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。4-(5-Methyloxazol-4-yl)phenol, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol and (2'R)-2' obtained in step 3 -Deoxy-2'-fluoro-2'-methyluridine is used as the raw material, and the target compound is obtained by the same method as steps 2 and 3 of Example 1.
1H NMR(300MHz,DMSO-d6)δppm:11.51(s,1H),8.28(s,1H),7.68(d,2H),7.56(d,1H),7.31(d,2H),6.13-6.05(m,2H),5.87(m,1H),5.56(m,1H),4.88-4.81(m,1H),4.35(m,1H),4.22(m,1H),4.04-3.78(m,1H),3.89-3.31(m,2H),2.50(s,3H),1.29-1.22(m,6H),1.16-1.14(m,6H)。 1 H NMR (300MHz, DMSO-d 6 ) δppm: 11.51(s, 1H), 8.28(s, 1H), 7.68(d, 2H), 7.56(d, 1H), 7.31(d, 2H), 6.13- 6.05(m, 2H), 5.87(m, 1H), 5.56(m, 1H), 4.88-4.81(m, 1H), 4.35(m, 1H), 4.22(m, 1H), 4.04-3.78(m, 1H), 3.89-3.31 (m, 2H), 2.50 (s, 3H), 1.29-1.22 (m, 6H), 1.16-1.14 (m, 6H).
LC-MS m/z:[M+H]+=611。LC-MS m/z: [M+H] + =611.
实施例22(2S)-2-(((1-甲基-1H-吲哚-4-基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 22 (2S)-2-(((1-methyl-1H-indol-4-yloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo -3,4-Dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate isopropyl ester
步骤1N-甲基-4-甲氧基吲哚Step 1N-Methyl-4-methoxyindole
称取1.47g 4-甲氧基吲哚(10mmol),1.38g碳酸钾(10mmol)于250mL的茄形瓶中,加入100mL丙酮溶解后,缓慢加入2.84g碘甲烷(20mmol),室温反应,反应结束后,向反应液中加入100mL乙酸乙酯和50mL饱和氯化钠水溶液萃取,水洗3遍,然后收集有机相,用无水硫酸钠来干燥,过滤,浓缩,柱层析纯化得标题化合物。Weigh 1.47g of 4-methoxyindole (10mmol), 1.38g of potassium carbonate (10mmol) in a 250mL eggplant-shaped flask, add 100mL of acetone to dissolve, slowly add 2.84g of methyl iodide (20mmol), and react at room temperature. After the completion, 100 mL of ethyl acetate and 50 mL of saturated aqueous sodium chloride solution were added to the reaction solution for extraction, washed three times with water, and then the organic phase was collected, dried over anhydrous sodium sulfate, filtered, concentrated, and purified by column chromatography to obtain the title compound.
LC-MS m/z:[M+H]+=162。LC-MS m/z: [M+H] + =162.
步骤2N-甲基-4-羟基吲哚Step 2N-Methyl-4-hydroxyindole
以步骤1所得物N-甲基-4-甲氧基吲哚为原料,同实施例14步骤2的方法制得标题化合物。Using N-methyl-4-methoxyindole obtained in step 1 as a starting material, the title compound was prepared in the same manner as in step 2 of Example 14.
LC-MS m/z:[M+H]+=148。LC-MS m/z: [M+H] + =148.
步骤3(2S)-2-(((1-甲基-1H-吲哚-4-基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯的制备Step 3(2S)-2-(((1-Methyl-1H-indol-4-yloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo- Preparation of isopropyl 3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate
以步骤2所得物N-甲基-4-羟基吲哚、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。N-methyl-4-hydroxyindole obtained in step 2, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol and (2'R)-2'-deoxy-2' -Fluoro-2'-methyluridine was used as the raw material, and the target compound was obtained by the same method as steps 2 and 3 of Example 1.
1H NMR(300MHz,DMSO-d6)δppm:11.47(s,1H),7.46-7.48(m,1H),7.31-7.32(m,1H),7.25-7.27(m,1H),7.05-7.11(m,1H),6.95-6.97(m,1H),6.49-6.50(m,1H),5.96-6.04(m,2H),5.83-5.85(m,1H),5.33-5.35(m,1H),4.82-4.90(m,1H),4.35-4.41(m,1H),4.20-4.28(m,1H),3.88-4.00(m,1H),3.84-3.87(m,2H),3.78-3.82(m,3H),1.20-1.27(m,6H),1.14-1.16(m,6H)。 1 H NMR (300MHz, DMSO-d 6 ) δppm: 11.47(s,1H), 7.46-7.48(m,1H), 7.31-7.32(m,1H), 7.25-7.27(m,1H), 7.05-7.11 (m,1H),6.95-6.97(m,1H),6.49-6.50(m,1H),5.96-6.04(m,2H),5.83-5.85(m,1H),5.33-5.35(m,1H) ,4.82-4.90(m,1H),4.35-4.41(m,1H),4.20-4.28(m,1H),3.88-4.00(m,1H),3.84-3.87(m,2H),3.78-3.82( m, 3H), 1.20-1.27 (m, 6H), 1.14-1.16 (m, 6H).
LC-MS m/z:[M+H]+=583。LC-MS m/z: [M+H] + =583.
实施例23(2S)-2-(((4-(噁唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 23 (2S)-2-(((4-(oxazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo- Isopropyl 3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate
步骤1 2-溴-1-(4-甲氧基苯基)乙酮的制备Step 1 Preparation of 2-bromo-1-(4-methoxyphenyl)ethanone
称取1g 1-(4-甲氧基苯基)乙酮(6.7mmol),1.5g对甲苯磺酸一水合物(6.7mmol)和1.2g NBS(6.7mmol)于100mL的单口瓶中,加入30mL乙腈溶解,回流反应4小时,反应结束后,浓缩,柱层析纯化得标题化合物。Weigh 1g 1-(4-methoxyphenyl)ethanone (6.7mmol), 1.5g p-toluenesulfonic acid monohydrate (6.7mmol) and 1.2g NBS (6.7mmol) in a 100mL single-neck flask, add 30 mL of acetonitrile was dissolved, and the reaction was refluxed for 4 hours. After the reaction was completed, it was concentrated and purified by column chromatography to obtain the title compound.
步骤2 4-(4-甲氧基苯基)噁唑的制备Step 2 Preparation of 4-(4-methoxyphenyl)oxazole
称取1g步骤1所得物2-溴-1-(4-甲氧基苯基)乙酮(4.4mmol)于100mL的单口瓶中,加入2mL甲酰胺和20mL DMF,回流反应2h,原料结束后,浓缩,将反应液倒入冰水中,乙酸乙酯萃取,干燥,过滤,浓缩,柱层析纯化得标题化合物。Weigh 1g of step 1 gained 2-bromo-1-(4-methoxyphenyl)ethanone (4.4mmol) in a 100mL single-necked flask, add 2mL formamide and 20mL DMF, reflux for 2h, after the raw material finishes , concentrated, the reaction solution was poured into ice water, extracted with ethyl acetate, dried, filtered, concentrated, and purified by column chromatography to obtain the title compound.
步骤3 4-(噁唑-4-基)苯酚的制备Step 3 Preparation of 4-(oxazol-4-yl)phenol
称取0.4g步骤2所得物4-(4-甲氧基苯基)噁唑(2.3mmol)于100mL的单口瓶中,加入20mL二氯甲烷溶解后,冰浴下加入8mL三溴化硼的二氯甲烷溶液(1.0mol/L,8mL),加毕,室温反应24h,反应结束后,冰浴下加水淬灭,二氯甲烷萃取,干燥,过滤,浓缩,柱层析纯化得标题化合物。Weigh 0.4 g of step 2 obtained 4-(4-methoxyphenyl) oxazole (2.3 mmol) in a 100 mL single-necked flask, add 20 mL of dichloromethane to dissolve, and add 8 mL of boron tribromide under an ice bath. Dichloromethane solution (1.0 mol/L, 8 mL) was added and reacted at room temperature for 24 h. After the reaction, water was added under ice bath to quench, extracted with dichloromethane, dried, filtered, concentrated, and purified by column chromatography to obtain the title compound.
步骤4(2S)-2-(((4-(噁唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯的制备Step 4(2S)-2-(((4-(oxazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo-3 Preparation of isopropyl ,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate
以步骤3所得物4-(噁唑-4-基)苯酚、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。4-(oxazol-4-yl)phenol, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol and (2'R)-2'-deoxy-2 obtained in step 3 '-Fluoro-2'-methyluridine was used as the raw material, and the target compound was obtained by the same methods as steps 2 and 3 of Example 1.
1H NMR(500MHz,DMSO-d6)δppm:11.51(s,1H),8.59(s,1H),8.45(s,1H),7.78(d,2H),7.56(d,1H),7.31(d,2H),6.13-6.05(m,2H),5.87(m,1H),5.56(m,1H),4.88-4.81(m,1H),4.35(m,1H),4.22(m,1H),4.04-3.78(m,1H),3.89-3.31(m,2H),1.29-1.22(m,6H),1.16-1.14(m,6H)。 1 H NMR (500MHz, DMSO-d 6 ) δppm: 11.51(s, 1H), 8.59(s, 1H), 8.45(s, 1H), 7.78(d, 2H), 7.56(d, 1H), 7.31( d,2H),6.13-6.05(m,2H),5.87(m,1H),5.56(m,1H),4.88-4.81(m,1H),4.35(m,1H),4.22(m,1H) , 4.04-3.78 (m, 1H), 3.89-3.31 (m, 2H), 1.29-1.22 (m, 6H), 1.16-1.14 (m, 6H).
LC-MS m/z:[M+H]+=597。LC-MS m/z: [M+H] + =597.
实施例24(2S)-2-(((4-(5-氟噻唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 24 (2S)-2-(((4-(5-fluorothiazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo Isopropyl substituted-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate
步骤1 4-(5-氟-噻唑-4-基)苯酚的制备Step 1 Preparation of 4-(5-Fluoro-thiazol-4-yl)phenol
称取3g实施例18步骤1所得物4-(噻唑-4-基)苯酚(16mmol),6.0g 1-氯甲基-4-氟-1,4-二氮杂双环[2.2.2]辛烷二(四氟硼酸)盐(16mmol)于100mL的单口瓶中,加入50mL1,4-二氧六环溶解,回流反应12h,反应结束后,浓缩,柱层析纯化得标题化合物。Weigh 3 g of 4-(thiazol-4-yl)phenol (16 mmol) obtained in step 1 of Example 18, 6.0 g of 1-chloromethyl-4-fluoro-1,4-diazabicyclo[2.2.2]octane Alkane bis(tetrafluoroborate) salt (16 mmol) was placed in a 100 mL single-necked flask, 50 mL of 1,4-dioxane was added to dissolve, and the reaction was refluxed for 12 h. After the reaction was completed, concentrated and purified by column chromatography to obtain the title compound.
步骤2(2S)-2-(((4-(5-氟噻唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯的制备Step 2(2S)-2-(((4-(5-fluorothiazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo -3,4-Dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)isopropyl propionate preparation
以步骤1所得物4-(5-氟-噻唑-4-基)苯酚、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。4-(5-Fluoro-thiazol-4-yl)phenol, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol and (2'R)-2'- Deoxy-2'-fluoro-2'-methyluridine was used as a raw material, and the target compound was obtained by the same method as steps 2 and 3 of Example 1.
1H NMR(500MHz,DMSO-d6)δppm:11.51(s,1H),8.75(s,1H),7.84(d,2H),7.56(d,1H),7.35(d,2H),6.17-6.05(m,2H),5.86(m,1H),5.56(m,1H),4.88-4.81(m,1H),4.35(m,1H),4.22(m,1H),4.04-3.78(m,1H),3.89-3.31(m,2H),1.29-1.22(m,6H),1.16-1.14(m,6H)。 1 H NMR (500MHz, DMSO-d 6 ) δppm: 11.51(s, 1H), 8.75(s, 1H), 7.84(d, 2H), 7.56(d, 1H), 7.35(d, 2H), 6.17- 6.05(m, 2H), 5.86(m, 1H), 5.56(m, 1H), 4.88-4.81(m, 1H), 4.35(m, 1H), 4.22(m, 1H), 4.04-3.78(m, 1H), 3.89-3.31 (m, 2H), 1.29-1.22 (m, 6H), 1.16-1.14 (m, 6H).
LC-MS m/z:[M+H]+=631。LC-MS m/z: [M+H] + =631.
实施例25(2S)-2-(((2-氟-4-(噻唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 25(2S)-2-(((2-Fluoro-4-(thiazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-di Isopropyl oxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propanoate ester
步骤1 2-氟-4-(噻唑-4-基)苯酚的制备Step 1 Preparation of 2-fluoro-4-(thiazol-4-yl)phenol
称取3g 4-(噻唑-4-基)苯酚(0.017mol),6.0g 1-氯甲基-4-氟-1,4-二氮杂双环[2.2.2]辛烷二(四氟硼酸)盐(Selectfluor,0.017mol)于250mL的单口瓶中,加入30mL三氟乙酸,95℃反应3h,反应完全后,浓缩,加水和乙酸乙酯萃取,干燥,过滤,浓缩,柱层析纯化得标题化合物。Weigh 3g of 4-(thiazol-4-yl)phenol (0.017mol), 6.0g of 1-chloromethyl-4-fluoro-1,4-diazabicyclo[2.2.2]octanebis(tetrafluoroboric acid) ) salt (Selectfluor, 0.017mol) in a 250mL single-neck flask, add 30mL trifluoroacetic acid, react at 95°C for 3h, after the reaction is complete, concentrate, add water and ethyl acetate for extraction, dry, filter, concentrate, and purify by column chromatography to obtain title compound.
步骤2(2S)-2-(((2-氟-4-(噻唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯的制备Step 2(2S)-2-(((2-Fluoro-4-(thiazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo Isopropyl substituted-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate preparation
以步骤1所得物2-氟-4-(噻唑-4-基)苯酚、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。2-Fluoro-4-(thiazol-4-yl)phenol, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol and (2'R)-2'- Deoxy-2'-fluoro-2'-methyluridine was used as a raw material, and the target compound was obtained by the same method as steps 2 and 3 of Example 1.
1H NMR(500MHz,DMSO-d6)δppm:11.50(s,1H),9.21(s,1H),8.25(s,1H),7.96-7.81(m,2H),7.66-7.56(m,2H),6.13-6.05(m,2H),5.87(m,1H),5.56(m,1H),4.88-4.81(m,1H),4.35(m,1H),4.22(m,1H),4.04-3.78(m,1H),3.89-3.75(m,2H),1.29-1.22(m,6H),1.16-1.14(m,6H)。 1 H NMR (500MHz, DMSO-d 6 ) δppm: 11.50(s, 1H), 9.21(s, 1H), 8.25(s, 1H), 7.96-7.81(m, 2H), 7.66-7.56(m, 2H) ),6.13-6.05(m,2H),5.87(m,1H),5.56(m,1H),4.88-4.81(m,1H),4.35(m,1H),4.22(m,1H),4.04- 3.78 (m, 1H), 3.89-3.75 (m, 2H), 1.29-1.22 (m, 6H), 1.16-1.14 (m, 6H).
LC-MS m/z:[M+H]+=631。LC-MS m/z: [M+H] + =631.
实施例26(2S)-2-(((4-(噻吩-2-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 26(2S)-2-(((4-(thiophen-2-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo-3 ,4-Dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate isopropyl ester
步骤1 4-(噻吩-2-基)苯酚的制备Step 1 Preparation of 4-(thiophen-2-yl)phenol
称取3g 4-羟基苯硼酸(22mmol),3g 2-溴-噻吩(18mmol),7.5g碳酸钾(55mmol),1.2g PdCl2(PPh3)2(1.7mmol)于250mL的单口瓶中,加入50mL 1,4-二氧六环和10mL水,氮气保护下95℃反应3h,反应结束后,浓缩,加水和乙酸乙酯萃取,干燥,过滤,浓缩,柱层析纯化得标题化合物。Take by weighing 3g 4-hydroxyphenylboronic acid (22mmol), 3g 2-bromo-thiophene (18mmol), 7.5g potassium carbonate (55mmol), 1.2g PdCl 2 (PPh ) 2 ( 1.7mmol) in the single-necked flask of 250mL, Add 50 mL of 1,4-dioxane and 10 mL of water, react at 95°C for 3 h under nitrogen protection, after the reaction, concentrate, add water and ethyl acetate for extraction, dry, filter, concentrate, and purify by column chromatography to obtain the title compound.
步骤2(2S)-2-(((4-(噻吩-2-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯的制备Step 2(2S)-2-(((4-(thiophen-2-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo-3, Preparation of isopropyl 4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate
以步骤1所得物4-(噻吩-2-基)苯酚、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。4-(thiophen-2-yl)phenol, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol and (2'R)-2'-deoxy-2' obtained in step 1 -Fluoro-2'-methyluridine was used as the raw material, and the target compound was obtained by the same method as steps 2 and 3 of Example 1.
1H NMR(500MHz,DMSO-d6)δppm:11.50(s,1H),7.66-7.56(m,3H),7.42-7.22(m,2H),7.26(d,2H),7.12(m,1H),6.13-6.05(m,2H),5.87(m,1H),5.56(m,1H),4.88-4.81(m,1H),4.35(m,1H),4.22(m,1H),4.04-3.78(m,1H),3.89-3.75(m,2H),,1.29-1.22(m,6H),1.16-1.14(m,6H)。 1 H NMR (500MHz, DMSO-d 6 ) δppm: 11.50 (s, 1H), 7.66-7.56 (m, 3H), 7.42-7.22 (m, 2H), 7.26 (d, 2H), 7.12 (m, 1H) ),6.13-6.05(m,2H),5.87(m,1H),5.56(m,1H),4.88-4.81(m,1H),4.35(m,1H),4.22(m,1H),4.04- 3.78 (m, 1H), 3.89-3.75 (m, 2H), 1.29-1.22 (m, 6H), 1.16-1.14 (m, 6H).
LC-MS m/z:[M+H]+=612。LC-MS m/z: [M+H] + =612.
实施例27(2S)-2-(((3-氟-4-(噻唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 27 (2S)-2-(((3-fluoro-4-(thiazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-di Isopropyl oxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propanoate ester
步骤1 3-氟-4-(噻唑-4-基)苯酚的制备Step 1 Preparation of 3-fluoro-4-(thiazol-4-yl)phenol
以4-羟基-2-氟苯硼酸、4-溴-噻唑为原料,同实施例26步骤1的方法制得标题化合物。Using 4-hydroxy-2-fluorophenylboronic acid and 4-bromo-thiazole as starting materials, the title compound was obtained by the same method as step 1 of Example 26.
步骤2(2S)-2-(((3-氟-4-(噻唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯的制备Step 2(2S)-2-(((3-Fluoro-4-(thiazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo Isopropyl substituted-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate preparation
以步骤1所得物3-氟-4-(噻唑-4-基)苯酚、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。3-Fluoro-4-(thiazol-4-yl)phenol, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol and (2'R)-2'- Deoxy-2'-fluoro-2'-methyluridine was used as a raw material, and the target compound was obtained by the same method as steps 2 and 3 of Example 1.
1H NMR(300MHz,DMSO-d6)δppm:11.49(s,1H),9.23(s,1H),8.17(m,1H),8.13(s,1H),7.55(d,1H),7.30-7.19(m,2H),6.13-6.05(m,2H),5.87(m,1H),5.56(m,1H),4.88-4.81(m,1H),4.35(m,1H),4.22(m,1H),4.04-3.78(m,1H),3.89-3.75(m,2H),1.29-1.22(m,6H),1.16-1.14(m,6H)。 1 H NMR (300MHz, DMSO-d 6 ) δppm: 11.49(s, 1H), 9.23(s, 1H), 8.17(m, 1H), 8.13(s, 1H), 7.55(d, 1H), 7.30- 7.19(m, 2H), 6.13-6.05(m, 2H), 5.87(m, 1H), 5.56(m, 1H), 4.88-4.81(m, 1H), 4.35(m, 1H), 4.22(m, 1H), 4.04-3.78 (m, 1H), 3.89-3.75 (m, 2H), 1.29-1.22 (m, 6H), 1.16-1.14 (m, 6H).
LC-MS m/z:[M+H]+=631。LC-MS m/z: [M+H] + =631.
实施例28(2S)-2-(((3-甲基-4-(噻唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 28(2S)-2-(((3-methyl-4-(thiazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4- Dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)isopropionic acid Propyl ester
步骤1 3-甲基-4-(噻唑-4-基)苯酚的制备Step 1 Preparation of 3-methyl-4-(thiazol-4-yl)phenol
以4-羟基-2-甲基苯硼酸和4-溴噻唑为原料,同实施例26步骤1的方法制得标题化合物。Using 4-hydroxy-2-methylphenylboronic acid and 4-bromothiazole as starting materials, the title compound was prepared in the same manner as in Example 26, step 1.
步骤2(2S)-2-(((3-甲基-4-(噻唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Step 2(2S)-2-(((3-methyl-4-(thiazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-di Isopropyl oxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propanoate ester
以步骤1所得物3-甲基-4-(噻唑-4-基)苯酚、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。3-methyl-4-(thiazol-4-yl)phenol, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol and (2'R)-2' obtained in step 1 -Deoxy-2'-fluoro-2'-methyluridine is used as the raw material, and the target compound is obtained by the same method as steps 2 and 3 of Example 1.
1H NMR(500MHz,DMSO-d6)δppm:11.51(s,1H),9.19(s,1H),7.80(s,1H),7.60(d,2H),7.17(m,2H),6.13-6.05(m,2H),5.87(m,1H),5.56(m,1H),4.88-4.81(m,1H),4.35(m,1H),4.22(m,1H),4.04-3.78(m,1H),3.89-3.75(m,2H),2.39(s,3H),1.29-1.22(m,6H),1.16-1.14(m,6H)。 1 H NMR (500MHz, DMSO-d 6 ) δppm: 11.51(s, 1H), 9.19(s, 1H), 7.80(s, 1H), 7.60(d, 2H), 7.17(m, 2H), 6.13- 6.05(m, 2H), 5.87(m, 1H), 5.56(m, 1H), 4.88-4.81(m, 1H), 4.35(m, 1H), 4.22(m, 1H), 4.04-3.78(m, 1H), 3.89-3.75 (m, 2H), 2.39 (s, 3H), 1.29-1.22 (m, 6H), 1.16-1.14 (m, 6H).
LC-MS m/z:[M+H]+=627。LC-MS m/z: [M+H] + =627.
实施例29(2S)-2-(((3-甲基-4-(1-甲基-1H-咪唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 29(2S)-2-(((3-methyl-4-(1-methyl-1H-imidazol-4-yl)phenyloxy)(((2R,3R,4R,5R)- 5-(2,4-Dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphorus Acyl)amino)isopropyl propionate
步骤1 3-甲基-4-(1-甲基-1H-咪唑-4-基)苯酚的制备Step 1 Preparation of 3-methyl-4-(1-methyl-1H-imidazol-4-yl)phenol
以4-羟基-2-甲基苯硼酸和4-溴-N-甲基咪唑为原料,同实施例26步骤1的方法制得标题化合物。Using 4-hydroxy-2-methylbenzeneboronic acid and 4-bromo-N-methylimidazole as starting materials, the title compound was obtained by the same method as in Example 26, step 1.
步骤2(2S)-2-(((3-甲基-4-(1-甲基-1H-咪唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Step 2(2S)-2-(((3-Methyl-4-(1-methyl-1H-imidazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5 -(2,4-Dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl ) amino) isopropyl propionate
以步骤1所得物3-甲基-4-(1-甲基-1H-咪唑-4-基)苯酚、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。Using the resultant of step 1, 3-methyl-4-(1-methyl-1H-imidazol-4-yl)phenol, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol and ( 2'R)-2'-deoxy-2'-fluoro-2'-methyluridine was used as a raw material, and the target compound was obtained by the same method as steps 2 and 3 of Example 1.
1H NMR(300MHz,DMSO-d6)δppm:11.51(s,1H),7.80-7.31(m,4H),7.07(m,2H),6.13-6.05(m,2H),5.87(m,1H),5.56(m,1H),4.88-4.81(m,1H),4.35(m,1H),4.22(m,1H),4.04-3.78(m,1H),3.89-3.75(m,2H),3.71(s.3H),3.54(s,3H),1.29-1.22(m,6H),1.16-1.14(m,6H)。 1 H NMR (300MHz, DMSO-d 6 ) δppm: 11.51 (s, 1H), 7.80-7.31 (m, 4H), 7.07 (m, 2H), 6.13-6.05 (m, 2H), 5.87 (m, 1H) ), 5.56(m, 1H), 4.88-4.81(m, 1H), 4.35(m, 1H), 4.22(m, 1H), 4.04-3.78(m, 1H), 3.89-3.75(m, 2H), 3.71(s.3H), 3.54(s,3H), 1.29-1.22(m,6H), 1.16-1.14(m,6H).
LC-MS m/z:[M+H]+=624。LC-MS m/z: [M+H] + =624.
实施例30(2S)-2-(((4-(1-甲基-1H-咪唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 30(2S)-2-(((4-(1-methyl-1H-imidazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2, 4-Dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propane isopropyl acid
步骤1 4-(1-甲基-1H-咪唑-4-基)苯酚的制备Step 1 Preparation of 4-(1-methyl-1H-imidazol-4-yl)phenol
以4-羟基苯硼酸和4-溴-N-甲基咪唑为原料,同实施例26步骤1的方法制得标题化合物。Using 4-hydroxyphenylboronic acid and 4-bromo-N-methylimidazole as starting materials, the title compound was prepared in the same manner as in Example 26, step 1.
步骤2(2S)-2-(((4-(1-甲基-1H-咪唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯的制备Step 2(2S)-2-(((4-(1-methyl-1H-imidazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4 -Dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionic acid Preparation of isopropyl ester
以步骤1所得物4-(1-甲基-1H-咪唑-4-基)苯酚、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。4-(1-Methyl-1H-imidazol-4-yl)phenol, phosphorus oxychloride, L-alanine isopropyl hydrochloride, pentafluorophenol and (2'R)- 2'-Deoxy-2'-fluoro-2'-methyluridine was used as the raw material, and the target compound was obtained by the same method as steps 2 and 3 of Example 1.
1H NMR(500MHz,DMSO-d6)δppm:11.510(s,1H),7.72-7.58(m,5H),7.20(m,2H),6.04-6.00(m,2H),5.89(m,1H),5.58(m,1H),4.88-4.81(m,1H),4.35(m,1H),4.22(m,1H),4.043.78(m,1H),3.89-3.31(m,2H),3.68(s,3H),1.28-1.22(m,6H),1.16-1.14(m,6H)。 1 H NMR (500MHz, DMSO-d 6 ) δppm: 11.510 (s, 1H), 7.72-7.58 (m, 5H), 7.20 (m, 2H), 6.04-6.00 (m, 2H), 5.89 (m, 1H) ), 5.58(m, 1H), 4.88-4.81(m, 1H), 4.35(m, 1H), 4.22(m, 1H), 4.043.78(m, 1H), 3.89-3.31(m, 2H), 3.68(s, 3H), 1.28-1.22(m, 6H), 1.16-1.14(m, 6H).
LC-MS m/z:[M+H]+=610。LC-MS m/z: [M+H] + =610.
实施例31(2S)-2-(((4-(3-氯吡啶-2-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 31 (2S)-2-(((4-(3-chloropyridin-2-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo Isopropyl substituted-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate
步骤1 3-氯-2-(4-羟基苯基)吡啶的制备Step 1 Preparation of 3-chloro-2-(4-hydroxyphenyl)pyridine
称取1.38g 4-羟基苯硼酸(10mmol),1.91g 2-溴-3-氯吡啶(10mmol),0.4g[1,1'-双(二苯基磷)二茂铁]二氯化钯(0.5mmol),9g碳酸铯(30mmol)于250mL的茄形瓶中,加入50mL 1,4-二氧六环和5mL水,氮气保护下90℃反应1.5h,反应结束后,加入100mL乙酸乙酯和50mL饱和氯化钠水溶液萃取,水洗3遍,然后收集有机相,用无水硫酸钠来干燥,过滤,浓缩,柱层析纯化得标题化合物。Weigh 1.38g of 4-hydroxyphenylboronic acid (10mmol), 1.91g of 2-bromo-3-chloropyridine (10mmol), 0.4g of [1,1'-bis(diphenylphosphonium)ferrocene]palladium dichloride (0.5mmol), 9g of cesium carbonate (30mmol) was placed in a 250mL eggplant-shaped flask, 50mL of 1,4-dioxane and 5mL of water were added, and the reaction was carried out at 90°C for 1.5h under nitrogen protection. After the reaction, 100mL of ethyl acetate was added. The ester was extracted with 50 mL of saturated aqueous sodium chloride solution, washed three times with water, and then the organic phase was collected, dried over anhydrous sodium sulfate, filtered, concentrated, and purified by column chromatography to obtain the title compound.
LC-MS m/z:[M+H]+=206。LC-MS m/z: [M+H] + =206.
步骤2(2S)-2-(((4-(3-氯吡啶-2-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯的制备Step 2(2S)-2-(((4-(3-chloropyridin-2-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo -3,4-Dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)isopropyl propionate preparation
以步骤1所得物3-氯-2-(4-羟基苯基)吡啶、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。3-Chloro-2-(4-hydroxyphenyl)pyridine, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol and (2'R)-2'- Deoxy-2'-fluoro-2'-methyluridine was used as a raw material, and the target compound was obtained by the same method as steps 2 and 3 of Example 1.
1H NMR(300MHz,DMSO-d6)δppm:11.47(s,1H),8.61-8.62(m,1H),8.02-8.05(m,1H),7.69-7.72(m,2H),7.56-7.59(m,1H),7.41-7.45(m,1H),7.32-7.35(m,2H),6.14-6.17(m,1H),6.05-6.09(m,1H),5.84-5.98(m,1H),5.57-5.60(m,1H),4.83-4.91(m,1H),4.38-4.43(m,1H),4.25-4.29(m,1H),4.01-4.05(m,1H),3.80-3.89(m,2H),1.22-1.29(m,6H),1.15-1.17(m,6H)。 1 H NMR (300MHz, DMSO-d 6 ) δppm: 11.47 (s, 1H), 8.61-8.62 (m, 1H), 8.02-8.05 (m, 1H), 7.69-7.72 (m, 2H), 7.56-7.59 (m,1H),7.41-7.45(m,1H),7.32-7.35(m,2H),6.14-6.17(m,1H),6.05-6.09(m,1H),5.84-5.98(m,1H) ,5.57-5.60(m,1H),4.83-4.91(m,1H),4.38-4.43(m,1H),4.25-4.29(m,1H),4.01-4.05(m,1H),3.80-3.89( m, 2H), 1.22-1.29 (m, 6H), 1.15-1.17 (m, 6H).
LC-MS m/z:[M+H]+=641。LC-MS m/z: [M+H] + =641.
实施例32(2S)-2-(((4-(5-氯吡啶-2-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 32(2S)-2-(((4-(5-chloropyridin-2-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo Isopropyl substituted-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate
步骤1 4-(5-氯吡啶-2-基)苯酚的制备Step 1 Preparation of 4-(5-chloropyridin-2-yl)phenol
以4-羟基苯硼酸和2-溴-5-氯吡啶为原料,同实施例31步骤1的方法制得标题化合物。Using 4-hydroxyphenylboronic acid and 2-bromo-5-chloropyridine as starting materials, the title compound was prepared by the same method as in Example 31, step 1.
LC-MS m/z:[M+H]+=206。LC-MS m/z: [M+H] + =206.
步骤2(2S)-2-(((4-(5-氯吡啶-2-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯的制备Step 2(2S)-2-(((4-(5-chloropyridin-2-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo -3,4-Dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)isopropyl propionate preparation
以步骤1所得物4-(5-氯吡啶-2-基)苯酚、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。4-(5-Chloropyridin-2-yl)phenol, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol and (2'R)-2'-deoxygenate obtained in step 1 -2'-Fluoro-2'-methyluridine was used as the raw material, and the target compound was obtained by the same method as steps 2 and 3 of Example 1.
1H NMR(300MHz,DMSO-d6)δppm:11.47(s,1H),8.68(m,1H),8.08-8.11(m,1H),7.82-7.85(m,1H),7.55-7.59(m,1H),7.48-7.53(m,1H),7.31-7.35(m,2H),7.15-7.20(m,1H),6.07-6.14(m,2H),5.99(m,1H),5.57-5.60(m,1H),4.85-4.89(m,1H),4.37-4.41(m,1H),4.26-4.28(m,1H),4.01(m,1H),3.80-3.87(m,2H),1.22-1.29(m,6H),1.13-1.16(m,6H)。 1 H NMR (300MHz, DMSO-d 6 ) δppm: 11.47(s, 1H), 8.68(m, 1H), 8.08-8.11(m, 1H), 7.82-7.85(m, 1H), 7.55-7.59(m ,1H),7.48-7.53(m,1H),7.31-7.35(m,2H),7.15-7.20(m,1H),6.07-6.14(m,2H),5.99(m,1H),5.57-5.60 (m,1H),4.85-4.89(m,1H),4.37-4.41(m,1H),4.26-4.28(m,1H),4.01(m,1H),3.80-3.87(m,2H),1.22 -1.29(m, 6H), 1.13-1.16(m, 6H).
LC-MS m/z:[M+H]+=641。LC-MS m/z: [M+H] + =641.
实施例33(2S)-2-(((4-(5-氯吡啶-2-基)-3-甲基苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 33(2S)-2-(((4-(5-chloropyridin-2-yl)-3-methylphenyloxy)(((2R,3R,4R,5R)-5-(2 ,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino) isopropyl propionate
步骤1 4-(5-氯吡啶-2-基)-3-甲基苯酚的制备Step 1 Preparation of 4-(5-chloropyridin-2-yl)-3-methylphenol
以2-甲基-4-羟基苯硼酸和2-溴-5-氯吡啶为原料,同实施例31步骤1的方法制得标题化合物。Using 2-methyl-4-hydroxybenzeneboronic acid and 2-bromo-5-chloropyridine as starting materials, the title compound was obtained by the same method as Example 31, step 1.
LC-MS m/z:[M+H]+=220。LC-MS m/z: [M+H] + =220.
步骤2(2S)-2-(((4-(5-氯吡啶-2-基)-3-甲基苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Step 2(2S)-2-(((4-(5-chloropyridin-2-yl)-3-methylphenyloxy)(((2R,3R,4R,5R)-5-(2, 4-Dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propane isopropyl acid
以步骤1所得物4-(5-氯吡啶-2-基)-3-甲基苯酚、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。4-(5-Chloropyridin-2-yl)-3-methylphenol obtained in step 1, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol and (2'R) -2'-Deoxy-2'-fluoro-2'-methyluridine was used as the raw material, and the target compound was obtained by the same method as steps 2 and 3 of Example 1.
1H NMR(300MHz,DMSO-d6)δppm:11.51(s,1H),8.69(m,1H),7.54-7.57(m,2H),7.39-7.42(m,1H),7.14-7.17(m,2H),6.05-6.12(m,2H),5.85(m,1H),5.54-5.57(m,1H),4.82-4.91(m,1H),4.36-4.42(m,1H),4.21-4.27(m,2H),3.90-4.01(m,1H),3.82-3.88(m,2H),2.30(m,3H),1.21-1.29(m,6H),1.14-1.16(m,6H)。 1 H NMR (300MHz, DMSO-d 6 ) δppm: 11.51 (s, 1H), 8.69 (m, 1H), 7.54-7.57 (m, 2H), 7.39-7.42 (m, 1H), 7.14-7.17 (m ,2H),6.05-6.12(m,2H),5.85(m,1H),5.54-5.57(m,1H),4.82-4.91(m,1H),4.36-4.42(m,1H),4.21-4.27 (m, 2H), 3.90-4.01 (m, 1H), 3.82-3.88 (m, 2H), 2.30 (m, 3H), 1.21-1.29 (m, 6H), 1.14-1.16 (m, 6H).
LC-MS m/z:[M+H]+=655。LC-MS m/z: [M+H] + =655.
实施例34(2S)-2-(((4-(3-氟吡啶-2-基)-3-甲基苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 34 (2S)-2-(((4-(3-fluoropyridin-2-yl)-3-methylphenyloxy)(((2R,3R,4R,5R)-5-(2 ,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino) isopropyl propionate
步骤1 4-(3-氟吡啶-2-基)-3-甲基苯酚的制备Step 1 Preparation of 4-(3-fluoropyridin-2-yl)-3-methylphenol
以2-甲基-4-羟基苯硼酸和2-溴-3-氟吡啶为原料,同实施例31步骤1的方法制得标题化合物。Using 2-methyl-4-hydroxybenzeneboronic acid and 2-bromo-3-fluoropyridine as starting materials, the title compound was obtained by the same method as Example 31, step 1.
LC-MS m/z:[M+H]+=204。LC-MS m/z: [M+H] + =204.
步骤2(2S)-2-(((4-(3-氟吡啶-2-基)-3-甲基苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Step 2(2S)-2-(((4-(3-Fluoropyridin-2-yl)-3-methylphenyloxy)(((2R,3R,4R,5R)-5-(2, 4-Dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propane isopropyl acid
以步骤1所得物4-(3-氟吡啶-2-基)-3-甲基苯酚、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。4-(3-Fluoropyridin-2-yl)-3-methylphenol obtained in step 1, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol and (2'R) -2'-Deoxy-2'-fluoro-2'-methyluridine was used as the raw material, and the target compound was obtained by the same method as steps 2 and 3 of Example 1.
1H NMR(300MHz,DMSO-d6)δppm:11.51(s,1H),8.53(m,1H),8.13(m,1H),7.79-7.86(m,1H),7.50-7.57(m,1H),7.31-7.34(m,1H),7.15-7.20(m,2H),6.06-6.13(m,2H),5.84(m,1H),5.58-5.60(m,1H),4.85-4.89(m,1H),4.39(m,1H),4.28(m,1H),4.03(m,1H),3.83-3.85(m,2H),2.15(m,3H),1.22-1.29(m,6H),1.15-1.17(m,6H)。 1 H NMR (300MHz, DMSO-d 6 ) δppm: 11.51 (s, 1H), 8.53 (m, 1H), 8.13 (m, 1H), 7.79-7.86 (m, 1H), 7.50-7.57 (m, 1H) ),7.31-7.34(m,1H),7.15-7.20(m,2H),6.06-6.13(m,2H),5.84(m,1H),5.58-5.60(m,1H),4.85-4.89(m ,1H),4.39(m,1H),4.28(m,1H),4.03(m,1H),3.83-3.85(m,2H),2.15(m,3H),1.22-1.29(m,6H), 1.15-1.17 (m, 6H).
LC-MS m/z:[M+H]+=639。LC-MS m/z: [M+H] + =639.
实施例35(2S)-2-(((4-(5-氯噁唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 35 (2S)-2-(((4-(5-chlorooxazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-di Isopropyl oxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propanoate ester
步骤1 2-溴-1-(4-甲氧基苯基)乙酮的制备Step 1 Preparation of 2-bromo-1-(4-methoxyphenyl)ethanone
以1-(4-甲氧苯基)乙酮为原料,同实施例21步骤1的方法制得标题化合物。Using 1-(4-methoxyphenyl)ethanone as starting material, the title compound was prepared in the same manner as in Example 21, step 1.
步骤2 4-(4-甲氧基苯基)噁唑的制备Step 2 Preparation of 4-(4-methoxyphenyl)oxazole
以步骤1所得物2-溴-1-(4-甲氧基苯基)乙酮和甲酰胺为原料,同实施例21步骤2的方法制得标题化合物。Using 2-bromo-1-(4-methoxyphenyl)ethanone obtained in step 1 and formamide as starting materials, the title compound was prepared in the same manner as in step 2 of Example 21.
LC-MS m/z:[M+H]+=176。LC-MS m/z: [M+H] + =176.
步骤3 5-氯-4-(4-甲氧基苯基)噁唑的制备Step 3 Preparation of 5-chloro-4-(4-methoxyphenyl)oxazole
称取3.0g步骤2所得物4-(4-甲氧基苯基)噁唑(0.0171mol)于100ml的单口瓶中,加入30mL乙腈溶解后,加入2.29g NCS(0.0171mol),室温反应2h,反应结束后,浓缩,柱层析纯化得到标题化合物。Weigh 3.0 g of 4-(4-methoxyphenyl) oxazole (0.0171 mol) obtained in step 2 into a 100 ml single-neck flask, add 30 mL of acetonitrile to dissolve, add 2.29 g of NCS (0.0171 mol), and react at room temperature for 2 h , after the reaction was completed, concentrated and purified by column chromatography to obtain the title compound.
LC-MS m/z:[M+H]+=210。LC-MS m/z: [M+H] + =210.
步骤4 4-(5-氯噁唑-4-基)苯酚的制备Step 4 Preparation of 4-(5-chlorooxazol-4-yl)phenol
称取1.3g步骤3所得物5-氯-4-(4-甲氧基苯基)噁唑(6.22mmol)于50mL的单口瓶中,加入15mL甲磺酸溶解后,加入3.71g DL-Met(24.8mmol),加毕,60℃反应24h,反应结束后,冷却,倒入冰水,乙酸乙酯萃取(50mL x 6),干燥,过滤,浓缩,柱层析纯化得标题化合物。Weigh 1.3g of step 3 obtained 5-chloro-4-(4-methoxyphenyl)oxazole (6.22mmol) in a 50mL single-neck flask, add 15mL of methanesulfonic acid to dissolve, add 3.71g of DL-Met (24.8 mmol), the addition was completed, and the reaction was carried out at 60 °C for 24 h. After the reaction, cooled, poured into ice water, extracted with ethyl acetate (50 mL x 6), dried, filtered, concentrated, and purified by column chromatography to obtain the title compound.
1H NMR(300MHz,DMSO-d6)δppm:9.76(s,1H),8.48(s,1H),7.69(d,2H),6.88(d,2H)。 1 H NMR (300 MHz, DMSO-d 6 ) δ ppm: 9.76 (s, 1H), 8.48 (s, 1H), 7.69 (d, 2H), 6.88 (d, 2H).
LC-MS m/z:[M+H]+=196。LC-MS m/z: [M+H] + =196.
步骤5(2S)-2-(((4-(5-氯噁唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯的制备Step 5(2S)-2-(((4-(5-chlorooxazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo Isopropyl substituted-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate preparation
以步骤4所得物4-(5-氯噁唑-4-基)苯酚、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。4-(5-Chloroxazol-4-yl)phenol, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol and (2'R)-2'- Deoxy-2'-fluoro-2'-methyluridine was used as a raw material, and the target compound was obtained by the same method as steps 2 and 3 of Example 1.
1H NMR(300MHz,DMSO-d6)δppm:11.50(s,1H),8.55(s,1H),7.87(d,2H),7.56(d,1H),7.37(d,2H),5.90-6.20(m,2H),5.85(d,1H),5.57(d,1H),4.77-4.92(m,1H),4.20-4.45(m,2H),3.95-4.10(m,1H),3.70-3.95(m,2H),1.18-1.40(m,6H),1.15(d,6H)。 1 H NMR (300MHz, DMSO-d 6 ) δppm: 11.50(s, 1H), 8.55(s, 1H), 7.87(d, 2H), 7.56(d, 1H), 7.37(d, 2H), 5.90- 6.20(m, 2H), 5.85(d, 1H), 5.57(d, 1H), 4.77-4.92(m, 1H), 4.20-4.45(m, 2H), 3.95-4.10(m, 1H), 3.70- 3.95(m, 2H), 1.18-1.40(m, 6H), 1.15(d, 6H).
LC-MS m/z:[M+H]+=631。LC-MS m/z: [M+H] + =631.
实施例36(2S)-2-(((4-(3-氯吡啶-2-基)-3-甲基苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 36 (2S)-2-(((4-(3-chloropyridin-2-yl)-3-methylphenyloxy)(((2R,3R,4R,5R)-5-(2 ,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino) isopropyl propionate
步骤1 4-(3-氯吡啶-2-基)-3-甲基苯酚的制备Step 1 Preparation of 4-(3-chloropyridin-2-yl)-3-methylphenol
以2-溴-3-氯吡啶和4-羟基-2-甲基苯硼酸为原料,同实施例1步骤1的方法制得标题化合物。Using 2-bromo-3-chloropyridine and 4-hydroxy-2-methylbenzeneboronic acid as starting materials, the title compound was obtained by the same method as Example 1, step 1.
LC-MS m/z:[M+H]+=220。LC-MS m/z: [M+H] + =220.
步骤2(2S)-2-(((4-(3-氯吡啶-2-基)-3-甲基苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯的制备Step 2(2S)-2-(((4-(3-chloropyridin-2-yl)-3-methylphenyloxy)(((2R,3R,4R,5R)-5-(2, 4-Dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propane Preparation of isopropyl acid
以步骤1所得物4-(3-氯吡啶-2-基)-3-甲基苯酚、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得标题化合物。4-(3-Chloropyridin-2-yl)-3-methylphenol obtained in step 1, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol and (2'R) -2'-Deoxy-2'-fluoro-2'-methyluridine was used as the starting material, and the title compound was obtained by the same method as steps 2 and 3 of Example 1.
1HNMR(300MHz,DMSO-d6)δppm:11.48(s,1H),8.61(d,1H),8.01(d,1H),7.58(d,1H),7.46(d,1H),7.23-7.02(m,3H),6.20-5.90(m,3H),5.58(m,1H),4.88-4.83(m,1H),4.39-4.26(m,2H),4.01-3.98(m,1H),3.90-3.78(m,2H),2.04(s,3H),1.29-1.22(m,6H),1.16-1.14(m,6H)。 1 HNMR (300MHz, DMSO-d6) δppm: 11.48(s, 1H), 8.61(d, 1H), 8.01(d, 1H), 7.58(d, 1H), 7.46(d, 1H), 7.23-7.02( m,3H),6.20-5.90(m,3H),5.58(m,1H),4.88-4.83(m,1H),4.39-4.26(m,2H),4.01-3.98(m,1H),3.90- 3.78(m, 2H), 2.04(s, 3H), 1.29-1.22(m, 6H), 1.16-1.14(m, 6H).
LC-MS m/z:[M+H]+=655。.LC-MS m/z: [M+H] + =655. .
实施例37(2S)-2-(((4-(5-氯-1-甲基-1H-咪唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 37(2S)-2-(((4-(5-Chloro-1-methyl-1H-imidazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5 -(2,4-Dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl ) amino) isopropyl propionate
步骤1 4-(5-氯-1-甲基-1H-咪唑-4-基)苯酚的制备Step 1 Preparation of 4-(5-chloro-1-methyl-1H-imidazol-4-yl)phenol
称取0.81g实施例30步骤1所得物4-(1-甲基-1H-咪唑-4-基)苯酚(5.14mmol),0.62g NCS(5.14mmol)于100mL的单口瓶中,加入20mL乙腈溶解,回流反应1h,反应结束后,浓缩,柱层析纯化得标题化合物。Weigh 0.81g of Example 30 step 1 obtained 4-(1-methyl-1H-imidazol-4-yl)phenol (5.14mmol), 0.62g NCS (5.14mmol) in a 100mL single-neck flask, add 20mL of acetonitrile Dissolved, refluxed for 1 h, after the reaction, concentrated and purified by column chromatography to obtain the title compound.
步骤2(2S)-2-(((4-(5-氯-1-甲基-1H-咪唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯的制备Step 2(2S)-2-(((4-(5-Chloro-1-methyl-1H-imidazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5- (2,4-Dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl) Preparation of isopropyl amino)propionate
以步骤1所得物4-(5-氯-1-甲基-1H-咪唑-4-基)苯酚、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。4-(5-Chloro-1-methyl-1H-imidazol-4-yl)phenol obtained in step 1, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol and (2 'R)-2'-deoxy-2'-fluoro-2'-methyluridine was used as a raw material, and the target compound was obtained by the same method as in steps 2 and 3 of Example 1.
1H NMR(500MHz,DMSO-d6)δppm:11.51(s,1H),8.40(s,1H),7.84(d,2H),7.56(d,1H),7.35(d,2H),6.17-6.05(m,2H),5.86(m,1H),5.56(m,1H),4.88-4.81(m,1H),4.35(m,1H),4.22(m,1H),4.043.78(m,1H),3.89-3.31(m,2H),3.62(s,3H),1.29-1.22(m,6H),1.16-1.14(m,6H)。 1 H NMR (500MHz, DMSO-d 6 ) δppm: 11.51(s, 1H), 8.40(s, 1H), 7.84(d, 2H), 7.56(d, 1H), 7.35(d, 2H), 6.17- 6.05(m, 2H), 5.86(m, 1H), 5.56(m, 1H), 4.88-4.81(m, 1H), 4.35(m, 1H), 4.22(m, 1H), 4.043.78(m, 1H), 3.89-3.31 (m, 2H), 3.62 (s, 3H), 1.29-1.22 (m, 6H), 1.16-1.14 (m, 6H).
LC-MS m/z:[M+H]+=644。LC-MS m/z: [M+H] + =644.
实施例38(2S)-2-(((3-氟-4-(1-甲基-1H-咪唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 38(2S)-2-(((3-Fluoro-4-(1-methyl-1H-imidazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5 -(2,4-Dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl ) amino) isopropyl propionate
步骤1 3-氟-4-(1-甲基-1H-咪唑-4-基)苯酚Step 1 3-Fluoro-4-(1-methyl-1H-imidazol-4-yl)phenol
以4-溴-1-甲基-1H-咪唑和2-氟-4-羟基苯硼酸为原料,同实施例26步骤1的方法制得标题化合物。Using 4-bromo-1-methyl-1H-imidazole and 2-fluoro-4-hydroxybenzeneboronic acid as starting materials, the title compound was obtained by the same method as step 1 of Example 26.
步骤2(2S)-2-(((3-氟-4-(1-甲基-1H-咪唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯的制备Step 2(2S)-2-(((3-Fluoro-4-(1-methyl-1H-imidazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5- (2,4-Dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl) Preparation of isopropyl amino)propionate
以步骤1所得物3-氟-4-(1-甲基-1H-咪唑-4-基)苯酚、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。3-fluoro-4-(1-methyl-1H-imidazol-4-yl)phenol obtained in step 1, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol and (2 'R)-2'-deoxy-2'-fluoro-2'-methyluridine was used as a raw material, and the target compound was obtained by the same method as in steps 2 and 3 of Example 1.
LC-MS m/z:[M+H]+=628。LC-MS m/z: [M+H] + =628.
实施例39(2S)-2-(((4-(2-甲基噻唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 39 (2S)-2-(((4-(2-methylthiazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-di Isopropyl oxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propanoate ester
步骤1 4-(2-甲基噻唑-4-基)苯酚的制备Step 1 Preparation of 4-(2-methylthiazol-4-yl)phenol
以2-甲基-4-溴噻唑和4-羟基苯硼酸为原料,同实施例1步骤1的方法制得标题化合物。Using 2-methyl-4-bromothiazole and 4-hydroxybenzeneboronic acid as starting materials, the title compound was obtained by the same method as in Example 1, step 1.
LC-MS m/z:[M+H]+=192。LC-MS m/z: [M+H] + =192.
步骤2(2S)-2-(((4-(2-甲基噻唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯的制备Step 2(2S)-2-(((4-(2-Methylthiazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo Isopropyl substituted-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate preparation
以步骤1所得物4-(2-甲基噻唑-4-基)苯酚、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。4-(2-Methylthiazol-4-yl)phenol, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol and (2'R)-2'- Deoxy-2'-fluoro-2'-methyluridine was used as a raw material, and the target compound was obtained by the same method as steps 2 and 3 of Example 1.
LC-MS m/z:[M+H]+=627。LC-MS m/z: [M+H] + =627.
实施例40(2S)-2-(((4-(5-氯-2-甲基噻唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 40(2S)-2-(((4-(5-Chloro-2-methylthiazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2 ,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino) isopropyl propionate
步骤1 4-(5-氯-2-甲基噻唑-4-基)苯酚的制备Step 1 Preparation of 4-(5-chloro-2-methylthiazol-4-yl)phenol
以实施例39步骤1所得物4-(2-甲基噻唑-4-基)苯酚为原料,同实施例37步骤1的方法制得标题化合物。Using 4-(2-methylthiazol-4-yl)phenol obtained in step 1 of Example 39 as a starting material, the title compound was prepared in the same manner as in step 1 of Example 37.
LC-MS m/z:[M+H]+=226。LC-MS m/z: [M+H] + =226.
步骤2(2S)-2-(((4-(5-氯-2-甲基噻唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯的制备Step 2(2S)-2-(((4-(5-Chloro-2-methylthiazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2, 4-Dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propane Preparation of isopropyl acid
以步骤1所得物4-(5-氯-2-甲基噻唑-4-基)苯酚、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。4-(5-Chloro-2-methylthiazol-4-yl)phenol, phosphorus oxychloride, L-alanine isopropyl hydrochloride, pentafluorophenol and (2'R) obtained in step 1 -2'-Deoxy-2'-fluoro-2'-methyluridine was used as the raw material, and the target compound was obtained by the same method as steps 2 and 3 of Example 1.
LC-MS m/z:[M+H]+=661。LC-MS m/z: [M+H] + =661.
实施例41(2S)-2-(((4-(5-氯-2-甲基噻唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸甲酯Example 41 (2S)-2-(((4-(5-Chloro-2-methylthiazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2 ,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino) methyl propionate
以实施例40步骤1所得物4-(5-氯-2-甲基噻唑-4-基)苯酚、三氯氧磷、L-丙氨酸甲酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。4-(5-Chloro-2-methylthiazol-4-yl)phenol, phosphorus oxychloride, L-alanine methyl ester hydrochloride, pentafluorophenol and (2' R)-2'-deoxy-2'-fluoro-2'-methyluridine was used as the raw material, and the target compound was obtained by the same method as steps 2 and 3 of Example 1.
LC-MS m/z:[M+H]+=633。LC-MS m/z: [M+H] + =633.
实施例42(2S)-2-(((4-(5-氯-噻唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸甲酯Example 42 (2S)-2-(((4-(5-Chloro-thiazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-di Methyl oxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate
以实施例18步骤2所得物4-(5-氯噻唑-4-基)苯酚、三氯氧磷、L-丙氨酸甲酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。4-(5-Chlorothiazol-4-yl)phenol, phosphorus oxychloride, L-alanine methyl ester hydrochloride, pentafluorophenol and (2'R)-2' obtained in step 2 of Example 18 -Deoxy-2'-fluoro-2'-methyluridine is used as the raw material, and the target compound is obtained by the same method as steps 2 and 3 of Example 1.
1H NMR(300MHz,DMSO-d6)δppm:11.50(s,1H),9.13(s,1H),7.92(d,2H),7.56(d,1H),7.35(d,2H),6.20-6.02(m,2H),5.87(m,1H),5.58(m,1H),4.39-4.03(m,2H),4.01-3.98(m,1H),3.93-3.85(m,2H),3.58(s,3H),1.28-1.22(m,6H)。 1 H NMR (300MHz, DMSO-d6) δppm: 11.50(s, 1H), 9.13(s, 1H), 7.92(d, 2H), 7.56(d, 1H), 7.35(d, 2H), 6.20-6.02 (m,2H),5.87(m,1H),5.58(m,1H),4.39-4.03(m,2H),4.01-3.98(m,1H),3.93-3.85(m,2H),3.58(s , 3H), 1.28-1.22 (m, 6H).
LC-MS m/z:[M+H]+=619。LC-MS m/z: [M+H] + =619.
实施例43(2S)-2-(((4-(5-氯噻唑-4-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸乙酯Example 43 (2S)-2-(((4-(5-chlorothiazol-4-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo Ethyl substituted-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate
以实施例18步骤2所得物4-(5-氯噻唑-4-基)苯酚、三氯氧磷、L-丙氨酸乙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。4-(5-Chlorothiazol-4-yl)phenol, phosphorus oxychloride, L-alanine ethyl ester hydrochloride, pentafluorophenol and (2'R)-2' obtained in step 2 of Example 18 -Deoxy-2'-fluoro-2'-methyluridine is used as the raw material, and the target compound is obtained by the same method as steps 2 and 3 of Example 1.
LC-MS m/z:[M+H]+=633。LC-MS m/z: [M+H] + =633.
实施例44(2S)-2-(((4-(3-氟吡啶-2-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸甲酯Example 44 (2S)-2-(((4-(3-fluoropyridin-2-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo Methyl substituted-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propanoate
以实施例2步骤1所得物4-(3-氟吡啶-2-基)苯酚、三氯氧磷、L-丙氨酸甲酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。4-(3-fluoropyridin-2-yl)phenol, phosphorus oxychloride, L-alanine methyl ester hydrochloride, pentafluorophenol and (2'R)-2' obtained in Example 2, step 1 -Deoxy-2'-fluoro-2'-methyluridine is used as the raw material, and the target compound is obtained by the same method as steps 2 and 3 of Example 1.
LC-MS m/z:[M+H]+=597。LC-MS m/z: [M+H] + =597.
实施例45(2S)-2-(((4-(3-氟吡啶-2-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸乙酯Example 45 (2S)-2-(((4-(3-fluoropyridin-2-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo Ethyl substituted-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate
以实施例2步骤1所得物4-(3-氟吡啶-2-基)苯酚、三氯氧磷、L-丙氨酸乙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。4-(3-fluoropyridin-2-yl)phenol, phosphorus oxychloride, L-alanine ethyl ester hydrochloride, pentafluorophenol and (2'R)-2' obtained in Example 2, step 1 -Deoxy-2'-fluoro-2'-methyluridine is used as the raw material, and the target compound is obtained by the same method as steps 2 and 3 of Example 1.
LC-MS m/z:[M+H]+=611。LC-MS m/z: [M+H] + =611.
实施例46(2S)-2-(((苯并[b]噻吩-4-基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 46(2S)-2-(((Benzo[b]thiophen-4-yloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo-3, Isopropyl 4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate
以4-羟基苯并噻吩、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。With 4-hydroxybenzothiophene, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol and (2'R)-2'-deoxy-2'-fluoro-2'-methyl Using uridine as a raw material, the target compound was prepared by the same methods as steps 2 and 3 in Example 1.
1H NMR(500MHz,DMSO-d6)δppm:11.49(s,1H),7.84-7.78(m,2H),7.51(d,2H),7.37-7.30(m,2H),6.20-6.02(m,2H),5.88(m,1H),5.43(m,1H),4.86(m,1H),4.43-4.03(m,2H),4.01-3.98(m,1H),3.93-3.85(m,2H),1.28-1.22(m,6H),1.14-1.12(m,6H)。 1 H NMR (500MHz, DMSO-d6) δppm: 11.49 (s, 1H), 7.84-7.78 (m, 2H), 7.51 (d, 2H), 7.37-7.30 (m, 2H), 6.20-6.02 (m, 2H), 5.88(m, 1H), 5.43(m, 1H), 4.86(m, 1H), 4.43-4.03(m, 2H), 4.01-3.98(m, 1H), 3.93-3.85(m, 2H) , 1.28-1.22 (m, 6H), 1.14-1.12 (m, 6H).
LC-MS m/z:[M+H]+=586。LC-MS m/z: [M+H] + =586.
实施例47(2S)-2-(((苯并[b]噻吩-4-基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸甲酯Example 47(2S)-2-(((Benzo[b]thiophen-4-yloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo-3, Methyl 4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propanoate
以4-羟基苯并噻吩、三氯氧磷、L-丙氨酸甲酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。With 4-hydroxybenzothiophene, phosphorus oxychloride, L-alanine methyl ester hydrochloride, pentafluorophenol and (2'R)-2'-deoxy-2'-fluoro-2'-methylurine Using glycosides as raw materials, the target compound was obtained by the same methods as steps 2 and 3 in Example 1.
LC-MS m/z:[M+H]+=558。LC-MS m/z: [M+H] + =558.
实施例48(2S)-2-(((3-甲基苯并[b]噻吩-4-基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 48(2S)-2-(((3-methylbenzo[b]thiophen-4-yloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo Isopropyl substituted-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate
以3-甲基-4-羟基苯并噻吩、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。With 3-methyl-4-hydroxybenzothiophene, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol and (2'R)-2'-deoxy-2'-fluoro- 2'-methyluridine was used as the raw material, and the target compound was obtained by the same method as steps 2 and 3 in Example 1.
LC-MS m/z:[M+H]+=600。LC-MS m/z: [M+H] + =600.
实施例49(2S)-2-(((4-(3-甲基噻吩-2-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸甲酯Example 49 (2S)-2-(((4-(3-methylthiophen-2-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-di Methyl oxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate
步骤1 4-(3-甲基噻吩-2-基)苯酚的制备Step 1 Preparation of 4-(3-methylthiophen-2-yl)phenol
以2-溴-3-甲基噻吩和4-羟基苯硼酸为原料,同实施例1步骤1的方法制得标题化合物。Using 2-bromo-3-methylthiophene and 4-hydroxybenzeneboronic acid as starting materials, the title compound was prepared in the same manner as in Example 1, step 1.
LC-MS m/z:[M+H]+=191。LC-MS m/z: [M+H] + =191.
步骤2(2S)-2-(((4-(3-甲基噻吩-2-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸甲酯Step 2(2S)-2-(((4-(3-Methylthiophen-2-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo Methyl substituted-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate
以步骤1所得物4-(3-甲基噻吩-2-基)苯酚、三氯氧磷、L-丙氨酸甲酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。4-(3-Methylthiophen-2-yl)phenol, phosphorus oxychloride, L-alanine methyl ester hydrochloride, pentafluorophenol and (2'R)-2'-deoxygenated from step 1 -2'-Fluoro-2'-methyluridine was used as the raw material, and the target compound was obtained by the same method as steps 2 and 3 of Example 1.
1H NMR(500MHz,DMSO-d6)δppm:11.50(s,1H),7.57(s,1H),7.46-7.43(m,3H),7.30(d,2H),7.00(d,1H),6.20-6.02(m,2H),5.87(m,1H),5.58(m,1H),4.39-4.03(m,2H),4.01-3.98(m,1H),3.93-3.85(m,2H),3.60(s,3H),2.24(s,3H),1.28-1.22(m,6H)。LC-MSm/z:[M+H]+=598。 1 H NMR (500MHz, DMSO-d6) δppm: 11.50(s, 1H), 7.57(s, 1H), 7.46-7.43(m, 3H), 7.30(d, 2H), 7.00(d, 1H), 6.20 -6.02(m, 2H), 5.87(m, 1H), 5.58(m, 1H), 4.39-4.03(m, 2H), 4.01-3.98(m, 1H), 3.93-3.85(m, 2H), 3.60 (s, 3H), 2.24 (s, 3H), 1.28-1.22 (m, 6H). LC-MS m/z: [M+H] + =598.
实施例50(2S)-2-(((4-(3-甲基噻吩-2-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯Example 50(2S)-2-(((4-(3-methylthiophen-2-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-di Isopropyl oxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propanoate ester
以实施例49步骤1所得物4-(3-甲基噻吩-2-基)苯酚、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。4-(3-Methylthiophen-2-yl)phenol, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol and (2'R)- 2'-Deoxy-2'-fluoro-2'-methyluridine was used as the raw material, and the target compound was obtained by the same method as steps 2 and 3 of Example 1.
1H NMR(500MHz,DMSO-d6)δppm:11.50(s,1H),7.57(s,1H),7.46-7.43(m,3H),7.30(d,2H),7.00(d,1H),6.20-6.02(m,2H),5.87(m,1H),5.58(m,1H),4.88-4.83(m,1H),4.39-4.03(m,2H),4.01-3.98(m,1H),3.93-3.85(m,2H),2.26(s,3H),1.28-1.22(m,6H),1.17-1.14(m,6H)。 1 H NMR (500MHz, DMSO-d6) δppm: 11.50(s, 1H), 7.57(s, 1H), 7.46-7.43(m, 3H), 7.30(d, 2H), 7.00(d, 1H), 6.20 -6.02(m, 2H), 5.87(m, 1H), 5.58(m, 1H), 4.88-4.83(m, 1H), 4.39-4.03(m, 2H), 4.01-3.98(m, 1H), 3.93 -3.85(m, 2H), 2.26(s, 3H), 1.28-1.22(m, 6H), 1.17-1.14(m, 6H).
LC-MS m/z:[M+H]+=626。LC-MS m/z: [M+H] + =626.
实施例51(2S)-2-(((4-(5-甲基噻吩-2-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸甲酯Example 51 (2S)-2-(((4-(5-methylthiophen-2-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-di Methyl oxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propanoate
步骤1 4-(5-甲基噻吩-2-基)苯酚的制备Step 1 Preparation of 4-(5-methylthiophen-2-yl)phenol
以2-溴-5-甲基噻吩和4-羟基苯硼酸为原料,同实施例1步骤1的方法制得标题化合物。Using 2-bromo-5-methylthiophene and 4-hydroxybenzeneboronic acid as starting materials, the title compound was prepared in the same manner as in Example 1, step 1.
LC-MS m/z:[M+H]+=191。LC-MS m/z: [M+H] + =191.
步骤2(2S)-2-(((4-(5-甲基噻吩-2-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸甲酯Step 2(2S)-2-(((4-(5-Methylthiophen-2-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo Methyl substituted-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propanoate
以步骤1所得物4-(5-甲基噻吩-2-基)苯酚、三氯氧磷、L-丙氨酸甲酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1的方法,制得目标化合物。4-(5-Methylthiophen-2-yl)phenol, phosphorus oxychloride, L-alanine methyl ester hydrochloride, pentafluorophenol and (2'R)-2'-deoxygenated from step 1 -2'-Fluoro-2'-methyluridine was used as the raw material, and the target compound was obtained by the method of Example 1.
1H NMR(300MHz,DMSO-d6)δppm:11.51(s,1H),7.59-7.55(m,3H),7.25-7.21(m,3H),6.81(d,1H),6.20-6.02(m,2H),5.87(m,1H),5.58(m,1H),4.39-4.03(m,2H),4.01-3.98(m,1H),3.93-3.85(m,2H),3.30(s,3H),2.46(s,3H),1.29-1.22(m,6H)。 1 H NMR (300MHz, DMSO-d6) δppm: 11.51(s, 1H), 7.59-7.55(m, 3H), 7.25-7.21(m, 3H), 6.81(d, 1H), 6.20-6.02(m, 2H), 5.87(m, 1H), 5.58(m, 1H), 4.39-4.03(m, 2H), 4.01-3.98(m, 1H), 3.93-3.85(m, 2H), 3.30(s, 3H) , 2.46(s, 3H), 1.29-1.22(m, 6H).
LC-MS m/z:[M+H]+=598。LC-MS m/z: [M+H] + =598.
实施例52(2S)-2-(((4-(5-甲基噻吩-2-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸异丙酯酯Example 52 (2S)-2-(((4-(5-methylthiophen-2-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-di Isopropyl oxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propanoate Ester
以实施例51步骤1所得物4-(5-甲基噻吩-2-基)苯酚、三氯氧磷、L-丙氨酸异丙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。4-(5-methylthiophen-2-yl)phenol, phosphorus oxychloride, L-alanine isopropyl ester hydrochloride, pentafluorophenol and (2'R)- 2'-Deoxy-2'-fluoro-2'-methyluridine was used as the raw material, and the target compound was obtained by the same method as steps 2 and 3 of Example 1.
1H NMR(300MHz,DMSO-d6)δppm:11.50(s,1H),7.59-7.55(m,3H),7.25-7.22(m,3H),6.80(d,1H),6.20-6.02(m,2H),5.87(m,1H),5.58(m,1H),4.88-4.83(m,1H),4.39-4.03(m,2H),4.01-3.98(m,1H),3.93-3.85(m,2H),2.46(s,3H),1.28-1.22(m,6H),1.16-1.14(m,6H)。 1 H NMR (300MHz, DMSO-d6) δppm: 11.50(s, 1H), 7.59-7.55(m, 3H), 7.25-7.22(m, 3H), 6.80(d, 1H), 6.20-6.02(m, 2H), 5.87(m, 1H), 5.58(m, 1H), 4.88-4.83(m, 1H), 4.39-4.03(m, 2H), 4.01-3.98(m, 1H), 3.93-3.85(m, 2H), 2.46 (s, 3H), 1.28-1.22 (m, 6H), 1.16-1.14 (m, 6H).
LC-MS m/z:[M+H]+=626。LC-MS m/z: [M+H] + =626.
实施例53(2S)-2-(((4-(5-甲基噻吩-2-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸乙酯Example 53 (2S)-2-(((4-(5-methylthiophen-2-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-di Ethyl oxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate
以实施例51步骤1所得物4-(5-甲基噻吩-2-基)苯酚、三氯氧磷、L-丙氨酸乙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。4-(5-methylthiophen-2-yl)phenol, phosphorus oxychloride, L-alanine ethyl ester hydrochloride, pentafluorophenol and (2'R)-2 obtained in Example 51, step 1 '-Deoxy-2'-fluoro-2'-methyluridine was used as the raw material, and the target compound was obtained by the same method as steps 2 and 3 of Example 1.
1H NMR(300MHz,DMSO-d6)δppm:11.50(s,1H),7.55-7.57(m,3H),7.21-7.25(m,3H),6.80-6.81(m,1H),6.60-6.61(m,1H),5.98-6.04(m,1H),5.83-5.86(m,1H),5.55-5.58(m,1H),4.34-4.40(m,1H),4.20-4.26(m,1H),3.90-3.93(m,1H),3.85-3.88(m,2H),3.58(m,2H),2.45-2.49(m,3H),1.21-1.29(m,9H)。 1 H NMR (300MHz, DMSO-d6) δppm: 11.50(s, 1H), 7.55-7.57(m, 3H), 7.21-7.25(m, 3H), 6.80-6.81(m, 1H), 6.60-6.61( m,1H),5.98-6.04(m,1H),5.83-5.86(m,1H),5.55-5.58(m,1H),4.34-4.40(m,1H),4.20-4.26(m,1H), 3.90-3.93 (m, 1H), 3.85-3.88 (m, 2H), 3.58 (m, 2H), 2.45-2.49 (m, 3H), 1.21-1.29 (m, 9H).
LC-MS m/z:[M+H]+=612。LC-MS m/z: [M+H] + =612.
实施例54(2S)-2-(((4-(噻吩-2-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸乙酯Example 54 (2S)-2-(((4-(thiophen-2-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo-3 ,4-Dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate ethyl ester
以实施例26步骤1所得物4-(噻吩-2-基)苯酚、三氯氧磷、L-丙氨酸乙酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。4-(thiophen-2-yl)phenol, phosphorus oxychloride, L-alanine ethyl ester hydrochloride, pentafluorophenol and (2'R)-2'-deoxy- 2'-Fluoro-2'-methyluridine was used as the raw material, and the target compound was prepared in the same manner as in steps 2 and 3 of Example 1.
1H NMR(300MHz,DMSO-d6)δppm:11.50(s,1H),7.65(d,2H),7.58-7.52(m,2H),7.45(d,1H),7.25(d,2H),7.13-7.10(m,1H),6.14-6.06(m,2H),5.85-5.82(m,1H),5.58-5.55(m,1H),4.40-4.36(m,1H),4.26-4.22(m,1H),4.07-4.00(m,3H),3.89-3.81(m,2H),1.29-1.23(m,6H),1.16-1.11(t,3H)。 1 H NMR (300MHz, DMSO-d6) δppm: 11.50(s, 1H), 7.65(d, 2H), 7.58-7.52(m, 2H), 7.45(d, 1H), 7.25(d, 2H), 7.13 -7.10(m,1H),6.14-6.06(m,2H),5.85-5.82(m,1H),5.58-5.55(m,1H),4.40-4.36(m,1H),4.26-4.22(m, 1H), 4.07-4.00 (m, 3H), 3.89-3.81 (m, 2H), 1.29-1.23 (m, 6H), 1.16-1.11 (t, 3H).
LC-MS m/z:[M+H]+=598。LC-MS m/z: [M+H] + =598.
实施例55(2S)-2-(((4-(噻吩-2-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸甲酯Example 55(2S)-2-(((4-(thiophen-2-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo-3 Methyl ,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate
以实施例26步骤1所得物4-(噻吩-2-基)苯酚、三氯氧磷、L-丙氨酸甲酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。4-(thiophen-2-yl)phenol, phosphorus oxychloride, L-alanine methyl ester hydrochloride, pentafluorophenol and (2'R)-2'-deoxy- 2'-Fluoro-2'-methyluridine was used as the raw material, and the target compound was prepared in the same manner as in steps 2 and 3 of Example 1.
1H NMR(500MHz,DMSO-d6)δppm:11.48(s,1H),7.66(d,2H),7.58-7.51(m,2H),7.46(d,1H),7.26(d,2H),7.13(m,1H),6.15-6.00(m,2H),5.87(m,1H),5.56(m,1H),4.41-4.37(m,1H),4.28-4.21(m,1H),4.05(m,1H),4.01-3.80(m,2H),3.60(s,3H),1.29-1.17(m,6H)。 1 H NMR (500MHz, DMSO-d6) δppm: 11.48(s, 1H), 7.66(d, 2H), 7.58-7.51(m, 2H), 7.46(d, 1H), 7.26(d, 2H), 7.13 (m,1H),6.15-6.00(m,2H),5.87(m,1H),5.56(m,1H),4.41-4.37(m,1H),4.28-4.21(m,1H),4.05(m , 1H), 4.01-3.80 (m, 2H), 3.60 (s, 3H), 1.29-1.17 (m, 6H).
LC-MS m/z:[M+H]+=584。LC-MS m/z: [M+H] + =584.
实施例56(2S)-2-(((4-(噻吩-2-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸新戊酯Example 56 (2S)-2-(((4-(thiophen-2-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-dioxo-3 ,4-Dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionate neopentyl
以实施例26步骤1所得物4-(噻吩-2-基)苯酚、三氯氧磷、L-丙氨酸新戊酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。4-(thiophen-2-yl)phenol, phosphorus oxychloride, L-alanine neopentyl ester hydrochloride, pentafluorophenol and (2'R)-2'-deoxygenate obtained in Example 26, step 1 -2'-Fluoro-2'-methyluridine was used as the raw material, and the target compound was obtained by the same method as steps 2 and 3 of Example 1.
1H NMR(500MHz,DMSO-d6)δppm:11.51(s,1H),7.65(d,2H),7.58-7.51(m,2H),7.45(d,1H),7.26(d,2H),7.14-7.11(m,1H),6.17-6.00(m,2H),5.86(d,1H),5.58(d,1H),4.40-4.21(m,2H),4.05-3.98(m,1H),3.93-3.85(m,3H),3.64(m,1H),1.31-1.21(m,6H),0.86(s,9H)。 1 H NMR (500MHz, DMSO-d6) δppm: 11.51(s, 1H), 7.65(d, 2H), 7.58-7.51(m, 2H), 7.45(d, 1H), 7.26(d, 2H), 7.14 -7.11(m,1H),6.17-6.00(m,2H),5.86(d,1H),5.58(d,1H),4.40-4.21(m,2H),4.05-3.98(m,1H),3.93 -3.85(m, 3H), 3.64(m, 1H), 1.31-1.21(m, 6H), 0.86(s, 9H).
LC-MS m/z:[M+H]+=640。LC-MS m/z: [M+H] + =640.
实施例57(2S)-2-(((4-(3-甲基噻吩-2-基)苯基氧基)(((2R,3R,4R,5R)-5-(2,4-二氧代-3,4-二氢嘧啶-1(2H)-基)-4-氟-3-羟基-4-甲基四氢呋喃-2-基)甲氧基)磷酰基)氨基)丙酸新戊酯Example 57 (2S)-2-(((4-(3-methylthiophen-2-yl)phenyloxy)(((2R,3R,4R,5R)-5-(2,4-di Oxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)phosphoryl)amino)propionic acid neopentyl ester
以实施例49步骤1所得物4-(3-甲基噻吩-2-基)苯酚、三氯氧磷、L-丙氨酸新戊酯盐酸盐、五氟苯酚和(2'R)-2'-脱氧-2'-氟-2'-甲基尿苷为原料,同实施例1步骤2和3的方法,制得目标化合物。4-(3-Methylthiophen-2-yl)phenol, phosphorus oxychloride, L-alanine neopentyl ester hydrochloride, pentafluorophenol and (2'R)- 2'-Deoxy-2'-fluoro-2'-methyluridine was used as the raw material, and the target compound was obtained by the same method as steps 2 and 3 of Example 1.
1H NMR(500MHz,DMSO-d6)δppm:11.50(s,1H),7.58(d,1H),7.45(m,3H),7.30(d,2H),7.00(d,1H),6.17-6.02(m,2H),5.87(m,1H),5.56(m,1H),4.40-4.21(m,2H),4.05-3.98(m,1H),3.93-3.85(m,3H),3.64(m,1H),2.26(s,3H),1.31-1.23(m,6H),0.87(s,9H)。 1 H NMR (500MHz, DMSO-d6) δppm: 11.50(s, 1H), 7.58(d, 1H), 7.45(m, 3H), 7.30(d, 2H), 7.00(d, 1H), 6.17-6.02 (m,2H),5.87(m,1H),5.56(m,1H),4.40-4.21(m,2H),4.05-3.98(m,1H),3.93-3.85(m,3H),3.64(m , 1H), 2.26 (s, 3H), 1.31-1.23 (m, 6H), 0.87 (s, 9H).
LC-MS m/z:[M+H]+=654。LC-MS m/z: [M+H] + =654.
药理活性评价Pharmacological activity evaluation
实验例1本发明的化合物在HCV复制子系统中的抗病毒活性检测Experimental Example 1 Detection of antiviral activity of the compounds of the present invention in the HCV replication system
1.实验材料1. Experimental materials
1.1试剂:1.1 Reagents:
表1试剂列表Table 1 List of reagents
1.2Huh71b细胞系:1.2Huh71b cell line:
Huh71b细胞系由上海药明康德新药开发有限公司提供,为包含带有稳定的荧光素酶(Luc)报告子的HCV 1b复制子的Huh7细胞系。其通过基因重组技术将HCV非结构蛋白基因、neo(G418抗性)及荧光素酶报告基因克隆入pBR载体构建。然后将携带有HCV复制子的载体转染入huh7细胞,通过G418抗性筛选,HCV复制子可稳定复制且相关蛋白和荧光素酶在huh7细胞内稳定表达。该细胞模型用于抗HCV化合物体外筛选。通过检查荧光素酶发光底物的化学发光水平而测定化合物的抗HCV的活性。参见Lohmann V,et al.1999.Replicationof subgenomic hepatitis C virus RNAs in a hepatoma cell line.Science.285(5424):110-113.The Huh71b cell line, provided by Shanghai WuXi AppTec New Drug Development Co., Ltd., is a Huh7 cell line containing the HCV 1b replicon with a stable luciferase (Luc) reporter. It is constructed by cloning HCV non-structural protein gene, neo (G418 resistance) and luciferase reporter gene into pBR vector through gene recombination technology. Then, the vector carrying the HCV replicon was transfected into huh7 cells, and through G418 resistance screening, the HCV replicon could be stably replicated and related proteins and luciferase were stably expressed in huh7 cells. This cell model is used for in vitro screening of anti-HCV compounds. The anti-HCV activity of the compounds was determined by examining the level of chemiluminescence of the luciferase luminescent substrate. See Lohmann V, et al. 1999. Replication of subgenomic hepatitis C virus RNAs in a hepatoma cell line. Science. 285(5424): 110-113.
2.实验方法:2. Experimental method:
1)化合物准备:使用本发明以上实施例制备的化合物,每个化合物用DMSO配制成母液,然后用含0.5%DMSO的DMEM完全培养液稀释至10μM作为化合物起始终浓度,3倍稀释,共10个浓度,用POD 810全自动微孔板预处理系统(LabCyte公司,美国)将化合物分别加入到96孔板中;同时设立无效作用对照组(含0.5%DMSO的完全培养液代替化合物)和100%有效作用对照组(只加入含0.5%DMSO的完全培养液);1) Compound preparation: using the compounds prepared in the above examples of the present invention, each compound was prepared into a stock solution with DMSO, and then diluted to 10 μM with DMEM complete culture medium containing 0.5% DMSO as the compound concentration, 3-fold dilution, a total of 10 Compounds were added to 96-well plates with POD 810 automatic microplate pretreatment system (LabCyte, USA) at different concentrations; at the same time, an ineffective control group (complete medium containing 0.5% DMSO instead of compounds) and 100 % effective control group (only complete culture medium containing 0.5% DMSO was added);
2.1细胞准备:收集对数期的Huh71b细胞,用DMEM完全培养液重悬后,接种到96孔板中,每孔125μl,8×103个细胞/孔,置于37℃、5%CO2培养箱培养细胞72小时;2.1 Cell preparation: Collect log-phase Huh71b cells, resuspend them in DMEM complete culture medium, and inoculate into 96-well plates, 125 μl per well, 8×10 3 cells/well, placed at 37°C, 5% CO 2 Cells were cultured in an incubator for 72 hours;
2.2细胞活力检测:每孔加30μl细胞生长荧光滴定检测试剂,37℃、5%CO2培养箱培养细胞1小时,分光光度仪上检测荧光信号值,所得数据用于化合物细胞毒性计算;2.2 Cell viability detection: add 30 μl of cell growth fluorescence titration detection reagent to each well, incubate the cells for 1 hour at 37°C, 5% CO 2 incubator, and detect the fluorescence signal value on a spectrophotometer, and the obtained data is used for compound cytotoxicity calculation;
2.3Bright-Glo检测:每孔加100μl荧光素酶发光底物Bright-Glo,5分钟内用化学发光检测系统EnVision(PerkinElmer公司,美国)检测荧光信号值,所得数据用于化合物活力计算。2.3 Bright-Glo detection: add 100 μl of luciferase luminescent substrate Bright-Glo to each well, and use chemiluminescence detection system EnVision (PerkinElmer, USA) to detect the fluorescence signal value within 5 minutes, and the obtained data is used for compound activity calculation.
2.4数据处理:使用如下公式将所得数据转换为细胞活力百分比(Viability%):2.4 Data processing: Convert the obtained data to percent cell viability (Viability%) using the following formula:
CPD:化合物孔的荧光信号值CPD: Fluorescence signal value of compound wells
HPE(Hundred percent effect):100%有效作用对照荧光信号值HPE (Hundred percent effect): 100% effective control fluorescence signal value
ZPE(Zero percent effect):无效作用对照荧光信号值ZPE (Zero percent effect): no effect control fluorescence signal value
使用如下公式将原始数据处理为抑制百分数(Inhibition%):The raw data was processed as a percent inhibition (Inhibition %) using the following formula:
CPD:化合物孔的化学发光信号值CPD: Chemiluminescence signal value of compound wells
HPE(Hundred percent effect):100%有效作用化学发光信号值HPE (Hundred percent effect): 100% effective chemiluminescence signal value
ZPE(Zero percent effect):无效作用化学发光信号值ZPE (Zero percent effect): Invalid effect chemiluminescence signal value
将抑制百分数导入GraphPad Prism进一步处理得出对应曲线和EC50值。The percent inhibition was imported into GraphPad Prism for further processing to generate corresponding curves and EC50 values.
实验结果表明,本发明化合物的EC50在约0.05μM-1μM之间,具有非常好的抑制HCV病毒的能力。部分数据见表2。The experimental results show that the EC 50 of the compound of the present invention is between about 0.05 μM-1 μM, and has a very good ability to inhibit HCV virus. Some data are shown in Table 2.
表2Table 2
如Gilead公司的官方网站上所公布的,目前最好的抗HCV药物sofosbuvir(GS7977)在HCV复制子测定中对基因1b型的EC50值为0.102μM,具体参见http://www.gilead.com/~/media/Files/pdfs/medicines/liver-disease/sovaldi/sovaldi_pi.pdf。本发明的化合物具有与sofosbuvir相当或更优的抗HCV病毒活性。As announced on Gilead's official website, the current best anti-HCV drug, sofosbuvir (GS7977), has an EC 50 value of 0.102 μM for genotype 1b in the HCV replicon assay, see http://www.gilead for details. com/~/media/Files/pdfs/medicines/liver-disease/sovaldi/sovaldi_pi.pdf. The compounds of the present invention have anti-HCV virus activity equivalent or superior to sofosbuvir.
实验例2本发明的化合物对HCV细胞感染模型(Cell-culture-derivedinfectious HCV,HCVcc)的抗病毒活性检测Experimental Example 2 Detection of antiviral activity of compounds of the present invention on HCV cell infection model (Cell-culture-derived infectious HCV, HCVcc)
1实验材料1 Experimental material
1.1化合物1.1 Compounds
本实验例使用以上实施例制备的本发明的化合物,用DMSO配制成10mM母液后,用含0.5%DMSO的DMEM完全培养液稀释至1000nM,然后依次3倍稀释,共8个浓度。In this experimental example, the compounds of the present invention prepared in the above examples were used to prepare a 10 mM stock solution with DMSO, and then diluted to 1000 nM with DMEM complete medium containing 0.5% DMSO, and then diluted 3 times successively, with a total of 8 concentrations.
1.2细胞1.2 Cells
Huh 7.5.1细胞,由上海药明康德新药开发有限公司提供。Huh 7.5.1 cells were provided by Shanghai WuXi AppTec New Drug Development Co., Ltd.
1.3病毒1.3 Viruses
J399EM(HCV genotype 2a)病毒,即转染了EGFP(增强型绿色荧光蛋白)的HCV全长突变株,与JFH-1野生型具有相同的感染能力,同时通过在NS5A区域插入EGFP编码序列,可以直接在感染细胞内观察NS5A-EGFP融合蛋白荧光,由上海药明康德新药开发有限公司提供。J399EM (HCV genotype 2a) virus, that is, a full-length HCV mutant strain transfected with EGFP (enhanced green fluorescent protein), has the same infectivity as the JFH-1 wild type. At the same time, by inserting the EGFP coding sequence in the NS5A region, it can Direct observation of NS5A-EGFP fusion protein fluorescence in infected cells, provided by Shanghai WuXi AppTec New Drug Development Co., Ltd.
1.4试剂1.4 Reagents
2实验方法2 Experimental methods
2)Huh 7.5.1细胞准备:收集对数期的Huh 7.5.1细胞,用DMEM完全培养液重悬后,接种于96孔板中(7×103个细胞/孔),置于37℃、5%CO2培养箱中培养过夜;2) Preparation of Huh 7.5.1 cells: collect Huh 7.5.1 cells in log phase, resuspend them in DMEM complete medium, and inoculate them in a 96-well plate (7×10 3 cells/well) at 37°C , 5% CO 2 incubator overnight;
3)药物处理:将本发明化合物分别加入到96孔板中,化合物起始终浓度均为1000nM,每个化合物做双复孔,3倍稀释,共8个浓度,DMSO终浓度0.5%;同时设立无效作用对照组(含0.5%DMSO的完全培养液代替化合物)和100%有效作用对照组(加入无病毒感染的细胞);3) Drug treatment: The compounds of the present invention were added to the 96-well plate respectively, the compound concentration was 1000nM from beginning to end, and each compound was made into double wells, 3-fold dilution, a total of 8 concentrations, the final concentration of DMSO was 0.5%; Ineffective control group (complete medium containing 0.5% DMSO instead of compound) and 100% effective control group (adding cells without virus infection);
4)病毒感染:将J399EM病毒上清以每孔MOI 0.2的浓度加入96孔板中。随后置于37℃、5%CO2培养箱中培养3天。4) Virus infection: J399EM virus supernatant was added to a 96-well plate at a concentration of MOI 0.2 per well. It was then placed in a 37°C, 5% CO2 incubator for 3 days.
5)抗病毒活性测定:培养结束后,每孔加入荧光素酶发光底物,用化学发光检测系统Envision检测Luminescence信号值,所得数据用于化合物抑制HCV活性计算,计算公式为:5) Determination of antiviral activity: after culturing, luciferase luminescent substrate was added to each well, and the chemiluminescence detection system Envision was used to detect the Luminescence signal value, and the obtained data was used for the calculation of compound inhibition of HCV activity, and the calculation formula was:
Inhibition%=(RLUs-RLUc)/(RLUV-RLUc)×100Inhibition%=(RLUs- RLUc )/( RLUv - RLUc ) ×100
其中,RLUV表示病毒对照组(含0.5%DMSO的完全培养液代替化合物)的化学发光强度,RLUS表示对应化合物处理组的化学发光强度,RLUC表示细胞对照组(无病毒感染的细胞)的化学发光强度;Among them, RLU V represents the chemiluminescence intensity of the virus control group (complete medium containing 0.5% DMSO instead of the compound), RLU S represents the chemiluminescence intensity of the corresponding compound treatment group, and RLU C represents the cell control group (cells without virus infection) chemiluminescence intensity;
6)细胞毒性测定:细胞和化合物处理方法同以上步骤,但以培养基替代病毒加入实验板中。同样条件37℃,5%CO2培养箱中培养3天后,加入细胞活力检测试剂AlamarBlue,用分光光度仪检测Fluorescence信号值。所得数据用于化合物细胞毒性计算,计算公式为:6) Cytotoxicity assay: The cell and compound treatment methods were the same as the above steps, but the medium was added to the experimental plate instead of the virus. After culturing in a 5% CO 2 incubator for 3 days under the same conditions, AlamarBlue, a cell viability detection reagent, was added, and the Fluorescence signal value was detected by a spectrophotometer. The obtained data is used for compound cytotoxicity calculation, and the calculation formula is:
Viability%=RFUS/RFUM×100Viability%=RFU S /RFU M ×100
其中,RFUM表示溶媒对照组(含0.5%DMSO的完全培养液代替化合物)的荧光强度,RFUS表示对应化合物处理组的荧光强度;Wherein, RFU M represents the fluorescence intensity of the vehicle control group (complete medium containing 0.5% DMSO instead of compound), and RFU S represents the fluorescence intensity of the corresponding compound treatment group;
7)数据处理:将抑制百分数、细胞活力百分数分别导入GraphPad Prism软件进行数据处理,得出化合物对应的曲线及其对HCV的抑制活性(EC50)和细胞毒性(CC50)数值。7) Data processing: The inhibition percentage and cell viability percentage were respectively imported into GraphPad Prism software for data processing, and the corresponding curve of the compound and its HCV inhibitory activity (EC 50 ) and cytotoxicity (CC 50 ) values were obtained.
实验结果显示,本发明的化合物的EC50值基本上在40nM-150nM之间,CC50均大于1000nM,例如实施例20的化合物的EC50为53.4nM,CC50>1000nM。部分本发明的化合物的EC50值更小,例如实施例26的EC50为9.3nM,CC50>1000nM。由此可见,本发明的化合物具有优异的抗病毒活性,同时具有小的细胞毒性,安全性好。The experimental results show that the EC 50 values of the compounds of the present invention are basically between 40 nM and 150 nM, and the CC 50 values are all greater than 1000 nM. Some of the compounds of the invention have smaller EC50 values, eg Example 26 has an EC50 of 9.3 nM and a CC50 >1000 nM. It can be seen that the compound of the present invention has excellent antiviral activity, small cytotoxicity and good safety.
另外,本发明的发明人发现,本发明的化合物具有良好的血浆蛋白结合率,适于成药,具有非常好的临床应用前景。尽管以上已经对本发明作了详细描述,但是本领域技术人员理解,在不偏离本发明的精神和范围的前提下可以对本发明进行各种修改和改变。本发明的权利范围并不限于上文所作的详细描述,而应归属于权利要求书。In addition, the inventors of the present invention found that the compound of the present invention has a good plasma protein binding rate, is suitable for medicine, and has a very good clinical application prospect. Although the present invention has been described in detail above, it will be understood by those skilled in the art that various modifications and variations can be made in the present invention without departing from the spirit and scope of the invention. The scope of the right of the present invention is not limited to the detailed description above, but belongs to the claims.
Claims (5)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201510453910.XA CN105315319B (en) | 2014-07-30 | 2015-07-29 | Hepatitis C virus inhibitor and its application |
Applications Claiming Priority (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2014103709236 | 2014-07-30 | ||
| CN201410370923 | 2014-07-30 | ||
| CN2014104443916 | 2014-09-02 | ||
| CN201410444391 | 2014-09-02 | ||
| CN201510453910.XA CN105315319B (en) | 2014-07-30 | 2015-07-29 | Hepatitis C virus inhibitor and its application |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN105315319A CN105315319A (en) | 2016-02-10 |
| CN105315319B true CN105315319B (en) | 2020-11-20 |
Family
ID=55216774
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201510453910.XA Active CN105315319B (en) | 2014-07-30 | 2015-07-29 | Hepatitis C virus inhibitor and its application |
Country Status (2)
| Country | Link |
|---|---|
| CN (1) | CN105315319B (en) |
| WO (1) | WO2016015638A1 (en) |
Families Citing this family (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN106554382B (en) * | 2015-09-29 | 2019-12-03 | 四川科伦博泰生物医药股份有限公司 | Application in nucleoside phosphoramidite class compound and preparation method thereof and drug |
| CN108137630A (en) * | 2016-09-23 | 2018-06-08 | 鲁汶天主教大学 | It is fluorinated the prodrug of acyclic nucleoside phosphate ester |
| WO2018082503A1 (en) * | 2016-11-02 | 2018-05-11 | 四川科伦博泰生物医药股份有限公司 | Heterocyclic compound and preparation method and application thereof |
| WO2019080724A1 (en) * | 2017-10-23 | 2019-05-02 | 四川科伦博泰生物医药股份有限公司 | Nucleoside phosphate compound and preparation method therefor and use thereof |
| JP7161716B2 (en) * | 2018-04-13 | 2022-10-27 | 国立大学法人 熊本大学 | INCUBATOR DEVICE, CELL CULTURE ENVIRONMENT CONTROL SYSTEM AND CELL CULTURE ENVIRONMENT CONTROL METHOD |
| CN113861223B (en) * | 2021-09-07 | 2022-09-16 | 暨南大学 | A kind of synthetic method and application of thiazolo[3,2-a]benzimidazole compounds |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN104031104A (en) * | 2013-03-08 | 2014-09-10 | 南京圣和药业有限公司 | New Nucleoside Phosphoramidate Compounds and Their Applications |
Family Cites Families (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| AR056327A1 (en) * | 2005-04-25 | 2007-10-03 | Genelabs Tech Inc | NUCLEOSID COMPOUNDS FOR THE TREATMENT OF VIRAL INFECTIONS |
| US7964580B2 (en) * | 2007-03-30 | 2011-06-21 | Pharmasset, Inc. | Nucleoside phosphoramidate prodrugs |
| EA019341B1 (en) * | 2008-12-23 | 2014-02-28 | Джилид Фармассет, Ллс. | Nucleoside phosphoramidates |
| CN104231023B (en) * | 2013-06-06 | 2019-02-05 | 南京汇诚制药有限公司 | Tricyclic fused heterocycle nucleoside phosphoramidate compound, preparation method and application |
| UA117375C2 (en) * | 2013-09-04 | 2018-07-25 | Медівір Аб | Hcv polymerase inhibitors |
| CN104761604A (en) * | 2014-01-02 | 2015-07-08 | 江苏豪森药业股份有限公司 | Uridine monophosphate analogue, and preparation method and applications thereof |
-
2015
- 2015-07-29 CN CN201510453910.XA patent/CN105315319B/en active Active
- 2015-07-29 WO PCT/CN2015/085380 patent/WO2016015638A1/en not_active Ceased
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN104031104A (en) * | 2013-03-08 | 2014-09-10 | 南京圣和药业有限公司 | New Nucleoside Phosphoramidate Compounds and Their Applications |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2016015638A1 (en) | 2016-02-04 |
| CN105315319A (en) | 2016-02-10 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN104031104B (en) | New Nucleoside Phosphoramidate Compounds and Their Applications | |
| CN105315319B (en) | Hepatitis C virus inhibitor and its application | |
| EP3423451B1 (en) | Inhibitors of wdr5 protein-protein binding | |
| JP5735482B2 (en) | Fused hepatitis C inhibitor | |
| TWI486348B (en) | Inhibitors of hepatis c virus | |
| CN102119152B (en) | Organic compounds | |
| EP1849465A1 (en) | Agent for controlling function of gpr34 receptor | |
| TW200906826A (en) | Anti-viral inhibitors and methods of use | |
| CA2593450A1 (en) | Indole derivatives for treating viral infections | |
| CN104231023B (en) | Tricyclic fused heterocycle nucleoside phosphoramidate compound, preparation method and application | |
| TWI846853B (en) | Pyrrole compounds | |
| CN113423712A (en) | Bicyclic sulfonamides | |
| JP2022542390A (en) | Dihydropyrimidine derivatives and their use in the treatment of HBV infection or HBV-induced disease | |
| EP3962915B1 (en) | Selective ligands of human constitutive androstane receptor | |
| HK1210619B (en) | Nucleoside phosphoramidate compounds for use in the treatment of hcv |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant | ||
| TR01 | Transfer of patent right |
Effective date of registration: 20250109 Address after: No. 68 Zhaoqiao Henan Road, Changlu Street, Jiangbei New District, Nanjing City, Jiangsu Province, 210000 Patentee after: Nanjing Huicheng Pharmaceutical Co.,Ltd. Country or region after: China Address before: 210038 No. 9 Zhong Hui Road, Nanjing economic and Technological Development Zone, Jiangsu Patentee before: NANJING SANHOME PHARMACEUTICAL Co.,Ltd. Country or region before: China |
|
| TR01 | Transfer of patent right |