CN103638987A - Hydrogenation catalyst carrier for coal liquefaction oil, catalyst as well as preparation method and application of hydrogenation catalyst carrier - Google Patents
Hydrogenation catalyst carrier for coal liquefaction oil, catalyst as well as preparation method and application of hydrogenation catalyst carrier Download PDFInfo
- Publication number
- CN103638987A CN103638987A CN201310638434.XA CN201310638434A CN103638987A CN 103638987 A CN103638987 A CN 103638987A CN 201310638434 A CN201310638434 A CN 201310638434A CN 103638987 A CN103638987 A CN 103638987A
- Authority
- CN
- China
- Prior art keywords
- carrier
- catalyst
- catalyst carrier
- hydrogenation catalyst
- cnt
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 239000003054 catalyst Substances 0.000 title claims abstract description 130
- 238000005984 hydrogenation reaction Methods 0.000 title claims abstract description 54
- 239000003245 coal Substances 0.000 title claims abstract description 43
- 238000002360 preparation method Methods 0.000 title claims abstract description 28
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 claims abstract description 30
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 claims abstract description 26
- 239000000377 silicon dioxide Substances 0.000 claims abstract description 13
- 239000004408 titanium dioxide Substances 0.000 claims abstract description 10
- PNEYBMLMFCGWSK-UHFFFAOYSA-N Alumina Chemical compound [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 claims description 43
- 239000000843 powder Substances 0.000 claims description 43
- 239000002184 metal Substances 0.000 claims description 30
- 229910052751 metal Inorganic materials 0.000 claims description 29
- 239000011148 porous material Substances 0.000 claims description 18
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 18
- 238000000034 method Methods 0.000 claims description 11
- 239000002245 particle Substances 0.000 claims description 11
- 102000040350 B family Human genes 0.000 claims description 10
- 108091072128 B family Proteins 0.000 claims description 10
- 239000012752 auxiliary agent Substances 0.000 claims description 10
- 238000004898 kneading Methods 0.000 claims description 10
- PXHVJJICTQNCMI-UHFFFAOYSA-N nickel Substances [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 claims description 9
- 150000003839 salts Chemical class 0.000 claims description 8
- 150000001868 cobalt Chemical class 0.000 claims description 7
- 238000005516 engineering process Methods 0.000 claims description 7
- 150000002815 nickel Chemical class 0.000 claims description 7
- 238000001125 extrusion Methods 0.000 claims description 6
- 239000011261 inert gas Substances 0.000 claims description 6
- 239000000463 material Substances 0.000 claims description 6
- 238000000465 moulding Methods 0.000 claims description 6
- 239000002048 multi walled nanotube Substances 0.000 claims description 6
- 239000011230 binding agent Substances 0.000 claims description 5
- 239000003795 chemical substances by application Substances 0.000 claims description 5
- 239000000203 mixture Substances 0.000 claims description 3
- 238000002156 mixing Methods 0.000 claims description 2
- OGIDPMRJRNCKJF-UHFFFAOYSA-N titanium oxide Inorganic materials [Ti]=O OGIDPMRJRNCKJF-UHFFFAOYSA-N 0.000 claims description 2
- 239000003921 oil Substances 0.000 abstract description 12
- 238000006243 chemical reaction Methods 0.000 abstract description 10
- 230000003197 catalytic effect Effects 0.000 abstract description 5
- 238000006477 desulfuration reaction Methods 0.000 abstract description 5
- 230000023556 desulfurization Effects 0.000 abstract description 5
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 abstract description 4
- 238000006392 deoxygenation reaction Methods 0.000 abstract 2
- 239000002041 carbon nanotube Substances 0.000 abstract 1
- 229910021393 carbon nanotube Inorganic materials 0.000 abstract 1
- 239000008161 low-grade oil Substances 0.000 abstract 1
- TWNQGVIAIRXVLR-UHFFFAOYSA-N oxo(oxoalumanyloxy)alumane Chemical compound O=[Al]O[Al]=O TWNQGVIAIRXVLR-UHFFFAOYSA-N 0.000 abstract 1
- 235000012239 silicon dioxide Nutrition 0.000 abstract 1
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 36
- 239000000243 solution Substances 0.000 description 19
- 238000007598 dipping method Methods 0.000 description 17
- 238000001291 vacuum drying Methods 0.000 description 15
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 12
- 230000000694 effects Effects 0.000 description 12
- NLYAJNPCOHFWQQ-UHFFFAOYSA-N kaolin Chemical compound O.O.O=[Al]O[Si](=O)O[Si](=O)O[Al]=O NLYAJNPCOHFWQQ-UHFFFAOYSA-N 0.000 description 11
- 229910052622 kaolinite Inorganic materials 0.000 description 11
- QGZKDVFQNNGYKY-UHFFFAOYSA-O Ammonium Chemical compound [NH4+] QGZKDVFQNNGYKY-UHFFFAOYSA-O 0.000 description 10
- 241000219782 Sesbania Species 0.000 description 10
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 9
- 241000219793 Trifolium Species 0.000 description 9
- 229910017604 nitric acid Inorganic materials 0.000 description 9
- 238000003756 stirring Methods 0.000 description 8
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 7
- 229910000480 nickel oxide Inorganic materials 0.000 description 7
- 229910018104 Ni-P Inorganic materials 0.000 description 6
- 229910018536 Ni—P Inorganic materials 0.000 description 6
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 6
- 239000008367 deionised water Substances 0.000 description 6
- 229910021641 deionized water Inorganic materials 0.000 description 6
- 229910052731 fluorine Inorganic materials 0.000 description 6
- GNRSAWUEBMWBQH-UHFFFAOYSA-N oxonickel Chemical compound [Ni]=O GNRSAWUEBMWBQH-UHFFFAOYSA-N 0.000 description 6
- APUPEJJSWDHEBO-UHFFFAOYSA-P ammonium molybdate Chemical compound [NH4+].[NH4+].[O-][Mo]([O-])(=O)=O APUPEJJSWDHEBO-UHFFFAOYSA-P 0.000 description 5
- 229940010552 ammonium molybdate Drugs 0.000 description 5
- 235000018660 ammonium molybdate Nutrition 0.000 description 5
- 239000011609 ammonium molybdate Substances 0.000 description 5
- 229910052799 carbon Inorganic materials 0.000 description 5
- 239000011737 fluorine Substances 0.000 description 5
- 229910052739 hydrogen Inorganic materials 0.000 description 5
- 239000001257 hydrogen Substances 0.000 description 5
- KBJMLQFLOWQJNF-UHFFFAOYSA-N nickel(ii) nitrate Chemical compound [Ni+2].[O-][N+]([O-])=O.[O-][N+]([O-])=O KBJMLQFLOWQJNF-UHFFFAOYSA-N 0.000 description 5
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 4
- YCKRFDGAMUMZLT-UHFFFAOYSA-N Fluorine atom Chemical compound [F] YCKRFDGAMUMZLT-UHFFFAOYSA-N 0.000 description 4
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 4
- 239000000654 additive Substances 0.000 description 4
- 230000000996 additive effect Effects 0.000 description 4
- 235000011114 ammonium hydroxide Nutrition 0.000 description 4
- 229910052796 boron Inorganic materials 0.000 description 4
- 230000000052 comparative effect Effects 0.000 description 4
- WQOXQRCZOLPYPM-UHFFFAOYSA-N dimethyl disulfide Chemical compound CSSC WQOXQRCZOLPYPM-UHFFFAOYSA-N 0.000 description 4
- 238000012360 testing method Methods 0.000 description 4
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 3
- ZOXJGFHDIHLPTG-UHFFFAOYSA-N Boron Chemical compound [B] ZOXJGFHDIHLPTG-UHFFFAOYSA-N 0.000 description 3
- 229910019142 PO4 Inorganic materials 0.000 description 3
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 3
- 229910000428 cobalt oxide Inorganic materials 0.000 description 3
- IVMYJDGYRUAWML-UHFFFAOYSA-N cobalt(ii) oxide Chemical compound [Co]=O IVMYJDGYRUAWML-UHFFFAOYSA-N 0.000 description 3
- 239000007788 liquid Substances 0.000 description 3
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 3
- 239000010452 phosphate Substances 0.000 description 3
- 239000002994 raw material Substances 0.000 description 3
- 102000004160 Phosphoric Monoester Hydrolases Human genes 0.000 description 2
- 108090000608 Phosphoric Monoester Hydrolases Proteins 0.000 description 2
- 229910004298 SiO 2 Inorganic materials 0.000 description 2
- 229910000831 Steel Inorganic materials 0.000 description 2
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 2
- YTPLMLYBLZKORZ-UHFFFAOYSA-N Thiophene Chemical compound C=1C=CSC=1 YTPLMLYBLZKORZ-UHFFFAOYSA-N 0.000 description 2
- MCMNRKCIXSYSNV-UHFFFAOYSA-N Zirconium dioxide Chemical compound O=[Zr]=O MCMNRKCIXSYSNV-UHFFFAOYSA-N 0.000 description 2
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 2
- 239000012876 carrier material Substances 0.000 description 2
- 239000010941 cobalt Substances 0.000 description 2
- 229910017052 cobalt Inorganic materials 0.000 description 2
- GUTLYIVDDKVIGB-UHFFFAOYSA-N cobalt atom Chemical compound [Co] GUTLYIVDDKVIGB-UHFFFAOYSA-N 0.000 description 2
- UFMZWBIQTDUYBN-UHFFFAOYSA-N cobalt dinitrate Chemical compound [Co+2].[O-][N+]([O-])=O.[O-][N+]([O-])=O UFMZWBIQTDUYBN-UHFFFAOYSA-N 0.000 description 2
- 229910001981 cobalt nitrate Inorganic materials 0.000 description 2
- 239000000446 fuel Substances 0.000 description 2
- 229910052759 nickel Inorganic materials 0.000 description 2
- 229910052757 nitrogen Inorganic materials 0.000 description 2
- 235000006408 oxalic acid Nutrition 0.000 description 2
- JMANVNJQNLATNU-UHFFFAOYSA-N oxalonitrile Chemical compound N#CC#N JMANVNJQNLATNU-UHFFFAOYSA-N 0.000 description 2
- 229910052760 oxygen Inorganic materials 0.000 description 2
- 239000001301 oxygen Substances 0.000 description 2
- 239000000047 product Substances 0.000 description 2
- 238000005507 spraying Methods 0.000 description 2
- 239000010959 steel Substances 0.000 description 2
- 229910052717 sulfur Inorganic materials 0.000 description 2
- 239000011593 sulfur Substances 0.000 description 2
- LWIHDJKSTIGBAC-UHFFFAOYSA-K tripotassium phosphate Chemical compound [K+].[K+].[K+].[O-]P([O-])([O-])=O LWIHDJKSTIGBAC-UHFFFAOYSA-K 0.000 description 2
- MPPQGYCZBNURDG-UHFFFAOYSA-N 2-propionyl-6-dimethylaminonaphthalene Chemical compound C1=C(N(C)C)C=CC2=CC(C(=O)CC)=CC=C21 MPPQGYCZBNURDG-UHFFFAOYSA-N 0.000 description 1
- 239000004254 Ammonium phosphate Substances 0.000 description 1
- 241000196324 Embryophyta Species 0.000 description 1
- 240000005702 Galium aparine Species 0.000 description 1
- 235000014820 Galium aparine Nutrition 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- 239000002841 Lewis acid Substances 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- 241000612118 Samolus valerandi Species 0.000 description 1
- 229920002472 Starch Polymers 0.000 description 1
- 239000005864 Sulphur Substances 0.000 description 1
- FZQSLXQPHPOTHG-UHFFFAOYSA-N [K+].[K+].O1B([O-])OB2OB([O-])OB1O2 Chemical compound [K+].[K+].O1B([O-])OB2OB([O-])OB1O2 FZQSLXQPHPOTHG-UHFFFAOYSA-N 0.000 description 1
- 230000002159 abnormal effect Effects 0.000 description 1
- 238000009825 accumulation Methods 0.000 description 1
- 238000005054 agglomeration Methods 0.000 description 1
- 230000002776 aggregation Effects 0.000 description 1
- 229910000148 ammonium phosphate Inorganic materials 0.000 description 1
- 235000019289 ammonium phosphates Nutrition 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 150000004945 aromatic hydrocarbons Chemical class 0.000 description 1
- 125000003118 aryl group Chemical group 0.000 description 1
- 229910001593 boehmite Inorganic materials 0.000 description 1
- 238000009835 boiling Methods 0.000 description 1
- KGBXLFKZBHKPEV-UHFFFAOYSA-N boric acid Chemical compound OB(O)O KGBXLFKZBHKPEV-UHFFFAOYSA-N 0.000 description 1
- 239000004327 boric acid Substances 0.000 description 1
- 238000001354 calcination Methods 0.000 description 1
- 239000001913 cellulose Substances 0.000 description 1
- 229920002678 cellulose Polymers 0.000 description 1
- 235000010980 cellulose Nutrition 0.000 description 1
- 235000015165 citric acid Nutrition 0.000 description 1
- 239000004927 clay Substances 0.000 description 1
- 229910052570 clay Inorganic materials 0.000 description 1
- 239000013065 commercial product Substances 0.000 description 1
- 150000001875 compounds Chemical class 0.000 description 1
- 238000009833 condensation Methods 0.000 description 1
- 230000005494 condensation Effects 0.000 description 1
- 238000005336 cracking Methods 0.000 description 1
- 230000007423 decrease Effects 0.000 description 1
- 230000008021 deposition Effects 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- MNNHAPBLZZVQHP-UHFFFAOYSA-N diammonium hydrogen phosphate Chemical compound [NH4+].[NH4+].OP([O-])([O-])=O MNNHAPBLZZVQHP-UHFFFAOYSA-N 0.000 description 1
- 239000002283 diesel fuel Substances 0.000 description 1
- 238000009826 distribution Methods 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 125000001153 fluoro group Chemical group F* 0.000 description 1
- 239000000295 fuel oil Substances 0.000 description 1
- 239000003502 gasoline Substances 0.000 description 1
- 239000010439 graphite Substances 0.000 description 1
- 229910002804 graphite Inorganic materials 0.000 description 1
- 239000002010 green coke Substances 0.000 description 1
- 125000005842 heteroatom Chemical group 0.000 description 1
- 150000002431 hydrogen Chemical class 0.000 description 1
- FAHBNUUHRFUEAI-UHFFFAOYSA-M hydroxidooxidoaluminium Chemical compound O[Al]=O FAHBNUUHRFUEAI-UHFFFAOYSA-M 0.000 description 1
- 239000003350 kerosene Substances 0.000 description 1
- 238000003475 lamination Methods 0.000 description 1
- 150000007517 lewis acids Chemical class 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 230000001404 mediated effect Effects 0.000 description 1
- 229910044991 metal oxide Inorganic materials 0.000 description 1
- 150000004706 metal oxides Chemical class 0.000 description 1
- -1 nickel aluminate Chemical class 0.000 description 1
- 239000005416 organic matter Substances 0.000 description 1
- 238000011056 performance test Methods 0.000 description 1
- 235000011007 phosphoric acid Nutrition 0.000 description 1
- 229910052698 phosphorus Inorganic materials 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 229910000160 potassium phosphate Inorganic materials 0.000 description 1
- 235000011009 potassium phosphates Nutrition 0.000 description 1
- 230000002441 reversible effect Effects 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 238000001179 sorption measurement Methods 0.000 description 1
- 239000008107 starch Substances 0.000 description 1
- 235000019698 starch Nutrition 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 238000010998 test method Methods 0.000 description 1
- 229930192474 thiophene Natural products 0.000 description 1
- 238000012546 transfer Methods 0.000 description 1
- 238000004073 vulcanization Methods 0.000 description 1
Landscapes
- Catalysts (AREA)
Abstract
The invention relates to a hydrogenation catalyst carrier for coal liquefaction oil, a catalyst as well as a preparation method and application of the hydrogenation catalyst carrier and belongs to the field of hydrogenation catalysts. The carrier comprises the following components (counted by 100% of total weight of the carrier): 5-60wt% of carbon nanotube, 32-90wt% of aluminum oxide, 0-8wt% of silicon dioxide and 0-8wt% of titanium dioxide. When the hydrogenation catalyst carrier for the coal liquefaction oil is used for carrying out a hydrorefining reaction for directly liquefying low-grade oil by coal, after the reaction is continuously carried out for 48 hours, the desulfurization rate can reach 99.7%, the denitrification rate can reach 95.1%, and the deoxygenation rate can reach 99.5%; after the reaction is continuously carried out for 168 hours, the desulfurization rate can reach 98.9%, the denitrification rate can reach 94.6%, and the deoxygenation rate can reach 98.6%; the hydrogenation catalyst carrier has high catalytic activity and stability.
Description
Technical field
The present invention relates to a kind of liquefied coal coil catalyst carrier for hydrgenating, Catalysts and its preparation method and application, belong to catalyst field, particularly relate to a kind of hydrogenation catalyst for liquefied coal coil hydrofining technology, and prepare this catalyst carrier, with and preparation method separately.
Background technology
Along with the accelerated development of process of industrialization, worldwide oil shortage cannot be avoided.Coal resources in China is the most sufficient in three macrofossil energy resources, makes full use of the coal resources of China's abundant, and Devoting Major Efforts To Developing coal for replacing oil, coal liquefaction technology, be to reduce the effective way that oil lacks risk.At present, liquefied coal coil technology is to realize the main method of coal for replacing oil, coal liquefaction.Liquefied coal coil technology is under HTHP, and hydrogenation makes the organic matter in coal be converted into liquid fuel, and this liquid fuel is liquefied coal coil.Liquefied coal coil, by further hydrofinishing, can form the fuel oil such as gasoline, diesel oil.
Patent CN101607204A discloses a kind of take hydrodesulfurization that CNT is carrier, hydrodenitrogenation catalyst and preparation method thereof.Catalyst is comprised of Mo/Co and CNT, and each component is: Mo:6.44-8.04%, Co:1.32-1.75%; CNT is multi-walled carbon nano-tubes, specific area 110-156m
2/ g.The slaine of metering metal component is made to the aqueous solution, and then step load is on CNT, and drying, obtains catalyst after calcining.This catalyst activity is high, and hydrogenation of thiophene desulfurization, second cyanogen hydrodenitrogeneration conversion ratio are high.
Above-mentioned patent be take CNT as carrier Kaolinite Preparation of Catalyst, because the active force between carrier and active metal is low, and in catalytic action process, the easy agglomeration of active component, reduce in activated centre gradually, and catalyst activity reduces; And the carrier using CNT as hydrogenation catalyst, cost is relatively high, is difficult to heavy industrialization.
Summary of the invention
On the one hand, the invention provides a kind of liquefied coal coil catalyst carrier for hydrgenating, for liquefied coal coil, the described carrier gross weight of take is 100%, described carrier comprises the CNT of 5-60wt%, the silica of the aluminium oxide of 32-90wt%, 0-8wt% and the titanium dioxide of 0-8wt%, and other composition can be same or similar with general hydrogenation catalyst.
CNT has the features such as mechanical strength is large, own wt is light, its intensity is steel 100 times, weight only has 1/6 of steel; CNT has the structure of hollow, and the passage while can be used as reaction demonstrates the excellent locational choice in reaction; Because CNT has the structural pipe wall of modifiable surface, class graphite, nano level tube chamber, good electronics transmits performance, heat transfer property and abnormal mechanics and chemical property, its reversible adsorption ability machine to hydrogen is strong, and may produce hydrogen overflow effect; Therefore, CNT can reduce the Lewis acid site on carrier, be conducive to regulate and take the character of its catalyst surface that is carrier, can significantly improve mechanical strength and the carbon accumulation resisting ability of catalyst, thus stability and the service life of improving hydrogenation catalyst.
Wherein, the preferred multi-walled carbon nano-tubes of described CNT, its specific area is 180-350m
2/ g, pore volume is 0.4-1.5cm
3/ g; Selection has the alumina powder on the large surface of macropore, and preferably pore volume is 0.45-1.20cm
3/ g, the alumina powder that average pore size is 8-17nm; Preferably, the average diameter of described silica and described titanium dioxide is 30-70nm.Owing to having more condensation aromatic ring structure in liquefied coal coil composition, its average molecular structure is larger, and easily green coke, therefore selects the carrier material of macropore bigger serface to be conducive to for hydrogenation reaction provides better reaction channel, makes the catalyst of making have certain anti-carbon performance.
Preferably, the content of described multi-walled carbon nano-tubes in described carrier is 5-45wt%, and the content of described silica is 2-8wt%, and the content of described titanium dioxide is 2-8wt%, more preferably the content of described silica is 2-6wt%, and the content of described titanium dioxide is 2-6wt%.Because CNT cost is higher, when guaranteeing catalytic activity, should at utmost reduce catalyst cost, when the content of above-mentioned carrier material is this scope, can guarantee that catalyst has the production cost of the reduction catalyst that good catalytic activity again can maximum possible.
Preferably, the described carrier gross weight of take is 100%, also comprises NiO and/or the CoO of 2.8-5.6wt% in described carrier.By the appropriate NiO of load on carrier and/or CoO, can prepare Al
2o
3the active phase catalyst of II type, Al
2o
3during the active phase full cure of II type, be and pile up lamination MoS
2structure, is the active phase of Co-Mo-S of high-sulfur coordination, and it is conventionally piled up and be laminated on together by larger sheet, is not connected with carrier, and active each activated centre mutually of II type is dived active high, and hydrodesulfurization and hydrodenitrogenationactivity activity are higher than I type.
Preferably, the described carrier gross weight of take is 100%, also comprises the auxiliary agent of 0-2.6wt%, the peptizing agent of the binding agent of 1-4wt%, 0-2wt% and the extrusion aid of 0-2.5wt% in described carrier.By adding above additive, be conducive to carrier moulding, and can regulate the acidity of aluminium oxide, make it have suitable Acidity.
Preferably, described auxiliary agent is one or more in phosphate builder, fluorine auxiliary agent, boron additive, described phosphate builder, fluorine auxiliary agent, boron additive are the solable matter containing P, F, B, described phosphate builder is selected from one or more in phosphoric acid, ammonium phosphate, potassium phosphate etc., described fluorine auxiliary agent is selected from one or more in ammonium fluosilicate, prodan, potassium fluosilicate etc., and described boron additive is selected from one or more in boric acid, sodium fluoborate, dipotassium tetraborate etc.; Described binding agent is one or more in clay, silica, aluminium oxide, boehmite (SB powder), zirconia and titanium oxide; Described peptizing agent is one or more in citric acid, nitric acid, acetic acid, oxalic acid etc.; Described extrusion aid is sesbania powder, one or more in citric acid, oxalic acid, cellulose, starch etc.More preferably, described auxiliary agent is fluorine auxiliary agent; Described binding agent is high sticky SB powder; Described peptizing agent is citric acid; Described extrusion aid is sesbania powder, the compound extrusion aid that citric acid, oxalic acid form.
Preferably, the specific area of described liquefied coal coil catalyst carrier for hydrgenating is 250-400m
2/ g, pore volume is 0.45-0.75ml/g, and average pore size is 8-12nm, and diameter is the 75%-85% in Zhan Zong hole, 6-12nm hole; The particle diameter of described catalyst carrier for hydrgenating is 0.8-1.5mm.
On the other hand, the invention provides a kind of preparation method of liquefied coal coil catalyst carrier for hydrgenating, described method comprises:
By CNT, aluminium oxide joins in certain water gaging, and selectively add silica, titanium dioxide, auxiliary agent, binding agent, peptizing agent or extrusion aid, in the catalyst carrier that makes to make, contain the CNT with the total restatement 5-60wt% of carrier, the aluminium oxide of 32-90wt%, the titanium dioxide of 0-8wt%, the silica of 0-8wt%, the consumption of described water is enough to make the above-mentioned material that adds moistening, so that above-mentioned raw materials mixes, above-mentioned raw materials is mixed, after kneading 0.5-1.5h, extruded moulding, support shapes after moulding can be sheet, spherical, cylindric or different in nature bar, if cross section is the rectangular of clover or bunge bedstraw herb type, preferred different in nature bar, the particle diameter of carrier is controlled at 0.8-1.5mm,
Under the product of described extruded moulding is air-dry, 80-120 ℃ vacuum, roasting 3-9h under dry 1-6h, 350-650 ℃ inert gas atmosphere, obtains described catalyst carrier.
Preferably, described CNT also comprises before mixing with other support material:
Nickel salts and/or Cobalt salts are loaded on CNT, roasting 2-5h under dry 1-5h, 300-500 ℃ inert gas atmosphere under air-dry, 70-120 ℃ vacuum, obtain the CNT of carried metal nickel salt and/or Cobalt salts, wherein nickel salts and/or Cobalt salts can be configured to solution, by equi-volume impregnating, load on CNT, the catalyst carrier gross weight making of take is 100%, and it is 2.8-5.6wt% that the nickel salts of load and/or Cobalt salts be take the content of oxide in described carrier.By part active metal is directly loaded on CNT, can avoid this part active metal and aluminium oxide to form unreducible material, be conducive to improve the hydrogenation reaction activity of catalyst.
Another aspect, the invention provides a kind of liquefied coal coil hydrogenation catalyst, and for liquefied coal coil, it comprises:
Above-mentioned carrier;
Active component: VI B family active metal and/or VIII family active metal, described VI B family active metal is W and/or Mo, VIII family active metal is Co and/or Ni;
Wherein: the described hydrogenation catalyst gross weight of take is 100%, to take the content of oxide in described hydrogenation catalyst be 20-55wt% to active metal.
Preferably, the pore volume of described catalyst is 0.35-0.75cm
3/ g, specific area is 220-390m
2/ g.
Again on the one hand, the invention provides a kind of preparation method of liquefied coal coil hydrogenation catalyst, described method comprises:
Prepare as stated above catalyst carrier for hydrgenating;
The salt of JiangⅥ B family active metal and/or the salt of VIII family active metal load on described carrier, air-dry, and under 100-120 ℃ of vacuum, roasting 2-5h under dry 1-4h, 400-650 ℃ inert gas atmosphere, obtains described catalyst;
Wherein: the salt of the salt of VI B family active metal and/or VIII family active metal can be dissolved in the deionized water of certain pH value, make dipping solution, with this dipping solution, according to equi-volume impregnating, flood described carrier, thereby the salt of ShiⅥ B family active metal and/or the salt of VIII family active metal load on described carrier; Described VI B family active metal is W and/or Mo, and VIII family active metal is Co and/or Ni; The described hydrogenation catalyst gross weight of take is 100%, and it is 20-55wt% that active metal be take the content of oxide in described hydrogenation catalyst.
On the one hand, the invention provides the application of above-mentioned liquefied coal coil hydrogenation catalyst in liquefied coal coil hydrofining technology again.
Hydrogenation catalyst provided by the invention at least has following effect:
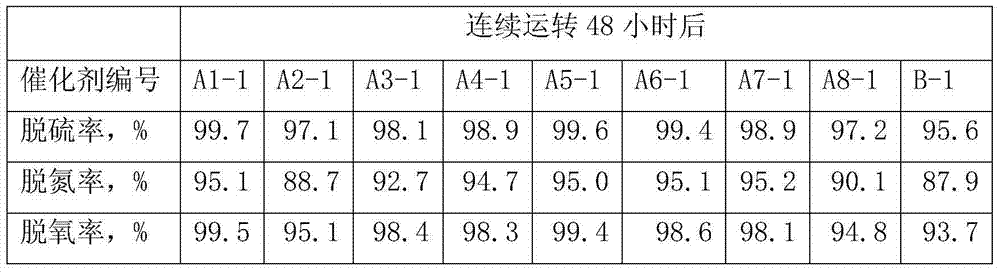
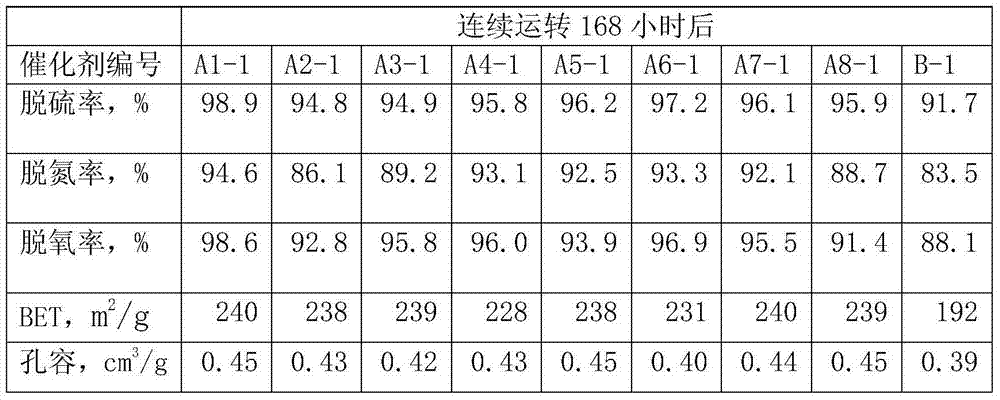
Hydrogenation catalyst provided by the invention is when the low minute oily hydrofining reaction of DCL/Direct coal liquefaction, and after 48 hours, desulfurization degree can reach 99.7%, denitrification percent can reach 95.1%, deoxidation rate can reach 99.5% in continuous operation; After continuous operation 168 hours, desulfurization degree can reach 98.9%, denitrification percent can reach 94.6%, deoxidation rate can reach 98.6%, has higher catalytic activity and stability.
Above-mentioned explanation is only the general introduction of technical solution of the present invention, in order to better understand technological means of the present invention, and can be implemented according to the content of description, below with preferred embodiment of the present invention, is described in detail as follows.
The specific embodiment
Below in conjunction with specific embodiment, the invention will be further described, but not as a limitation of the invention.
In the embodiment of the present invention, all raw materials are commercial product, and wherein CNT is multi-walled carbon nano-tubes, and its specific area is 180-350m
2/ g, pore volume is 0.4-1.5cm
3/ g; Alumina powder pore volume is 0.45-1.20cm
3/ g, average pore size is 8-17nm; The average diameter of silica and titania powder is 30-70nm; High sticky SB powder specific area is 240-350m
2/ g, pore volume is 0.42-0.62ml/g, colloidal sol index is greater than>=and 95%, common its specific area of SB powder 260-420m
2/ g, pore volume is 0.5-0.9ml/g, colloidal sol index>=80%.
Embodiment 1
Kaolinite Preparation of Catalyst carrier:
Nickel nitrate is dissolved in ethanol, is configured in nickel oxide, the solution of 15g NiO/100ml ethanol, the solution spraying preparing is impregnated in 80g CNT, under room temperature, place air-dry, under 80 ℃ of temperature conditions of vacuum drying oven dry 4 hours, N in Muffle furnace
2under the lower 350 ℃ of temperature conditions of atmosphere, roasting is 4 hours, obtains the CNT of load nickel oxide, the nickeliferous CNT of mentioning in following step for this load the CNT of nickel oxide;
The nitric acid that is 66% by 200g alumina powder, the nickeliferous high sticky SB powder of CNT, 16g titania powder, 50g, 6g sesbania powder, 4g citric acid, 7.6g ammonium fluosilicate, 15g carbonic hydroammonium, the 9ml concentration of 52g, adding water 210ml stirs, putting into kneading machine mediates 45 minutes, on banded extruder, be extruded into little of the clover of particle diameter 1.2mm, air-dry, under 110 ℃ of temperature conditions of vacuum drying oven, be dried 4 hours, N in Muffle furnace
2under the lower 550 ℃ of temperature conditions of atmosphere, roasting is 4 hours, makes catalyst carrier A1.
Prepare hydrogenation catalyst:
Get respectively ammonium metatungstate 145g, ammonium molybdate 102g, nickel nitrate 69g, phosphoric acid 32ml and deionized water, by ammoniacal liquor, allocate pH value and be mixed with the limpid dipping solution S1 of 500mlW-Mo-Ni-P.
The limpid dipping solution S1 of the above-mentioned W-Mo-Ni-P of carrier A 1 use is pressed to equi-volume impregnating dipping 4 hours, air-dry, under 110 ℃ of temperature conditions of vacuum drying oven, be dried 4 hours, then N in Muffle furnace
2under the lower 550 ℃ of temperature conditions of atmosphere, roasting is 4 hours, makes hydrogenation catalyst A1-1.
Embodiment 2
Kaolinite Preparation of Catalyst carrier:
With the method identical with embodiment 1 CNT of nickel oxide of having prepared load;
The nitric acid that is 66% by 200g alumina powder, the nickeliferous high sticky SB powder of CNT, 16g titania powder, 50g, 6g sesbania powder, 4g citric acid, 15g carbonic hydroammonium, the 9ml concentration of 52g, adding water 210ml stirs, putting into kneading machine mediates 45 minutes, on banded extruder, be extruded into little of the clover of particle diameter 1.2mm, air-dry, under 110 ℃ of temperature conditions of vacuum drying oven, be dried 4 hours, N in Muffle furnace
2under the lower 550 ℃ of temperature conditions of atmosphere, roasting is 4 hours, makes catalyst carrier A2.
Prepare hydrogenation catalyst:
Preparation method is identical with embodiment 1, obtains hydrogenation catalyst A2-1.
Embodiment 3
Kaolinite Preparation of Catalyst carrier:
Identical with embodiment 1, unique different be to have substituted high sticky SB powder with common SB powder, obtain catalyst carrier A3.
Prepare hydrogenation catalyst:
Preparation method is identical with embodiment 1, obtains hydrogenation catalyst A3-1.
Embodiment 4
Kaolinite Preparation of Catalyst carrier:
The nitric acid that is 66% by 200g alumina powder, 45g CNT, 16g titania powder, the high sticky SB powder of 50g, 6g sesbania powder, 4g citric acid, 7.6g ammonium fluosilicate, 15g carbonic hydroammonium, 9ml concentration, adding water 210ml stirs, putting into kneading machine mediated after 45 minutes, on banded extruder, be extruded into little of the clover of particle diameter 1.2mm, air-dry, under 110 ℃ of temperature conditions of vacuum drying oven, be dried 4 hours, N in Muffle furnace
2under the lower 550 ℃ of temperature conditions of atmosphere, roasting is 4 hours, makes catalyst carrier A4.
Kaolinite Preparation of Catalyst:
Get respectively ammonium metatungstate 73g, ammonium molybdate 51g, nickel nitrate 70g, phosphatase 11 5ml and deionized water, by ammoniacal liquor, allocate pH value and be mixed with the limpid dipping solution S2 of 250mlW-Mo-Ni-P.
The limpid dipping solution S2 of the above-mentioned W-Mo-Ni-P of carrier A 4 use is pressed to equi-volume impregnating dipping 4 hours, air-dry, under 110 ℃ of temperature conditions of vacuum drying oven, be dried 4 hours, then N in Muffle furnace
2under the lower 550 ℃ of temperature conditions of atmosphere, roasting is 4 hours, makes hydrogenation catalyst A4-1.
Embodiment 5
Kaolinite Preparation of Catalyst carrier:
Cobalt nitrate is dissolved in ethanol, is configured in cobalt oxide, the solution of 15g CoO/100ml ethanol, the solution spraying preparing is impregnated in 80g CNT, under room temperature, place air-dry, under 80 ℃ of temperature conditions of vacuum drying oven dry 4 hours, N in Muffle furnace
2under the lower 350 ℃ of temperature conditions of atmosphere, roasting is 4 hours, obtains the CNT of load cobalt oxide, in following step, mention containing the CNT of the cobalt CNT of cobalt oxide that has been this load;
The nitric acid that is 66% containing the high sticky SB powder of CNT, 17g SiO 2 powder, 50g, 6g sesbania powder, 4g citric acid, 7.6g ammonium fluosilicate, 15g carbonic hydroammonium, the 9ml concentration of cobalt by 200g alumina powder, 52g, adding water 210ml stirs, putting into kneading machine mediates 45 minutes, on banded extruder, be extruded into little of the clover of particle diameter 1.2mm, air-dry, under 110 ℃ of temperature conditions of vacuum drying oven, be dried 4 hours, N in Muffle furnace
2under the lower 550 ℃ of temperature conditions of atmosphere, roasting is 4 hours, makes catalyst carrier A5.
Prepare hydrogenation catalyst:
Get respectively ammonium metatungstate 145g, ammonium molybdate 102g, cobalt nitrate 69g, phosphoric acid 32ml and deionized water, by ammoniacal liquor, allocate pH value and be mixed with the limpid dipping solution S3 of 500mlW-Mo-Co-P.
The limpid dipping solution S3 of the above-mentioned W-Mo-Co-P of carrier A 5 use is pressed to equi-volume impregnating dipping 4 hours, air-dry, under 110 ℃ of temperature conditions of vacuum drying oven, be dried 4 hours, then N in Muffle furnace
2under the lower 550 ℃ of temperature conditions of atmosphere, roasting is 4 hours, makes hydrogenation catalyst A5-1.
Embodiment 6
Kaolinite Preparation of Catalyst carrier:
Nickel nitrate is dissolved in ethanol, is configured in nickel oxide, the solution of 30g NiO/100ml ethanol, uses the method identical with embodiment 1 CNT of nickel oxide of having prepared load;
The nitric acid that is 66% by 250g alumina powder, the nickeliferous high sticky SB powder of CNT, 36g, 6g sesbania powder, 4g citric acid, 7.6g ammonium fluosilicate, the 9ml concentration of 21g, adding water 230ml stirs, putting into kneading machine mediates 45 minutes, on banded extruder, be extruded into little of the clover of particle diameter 1.2mm, air-dry, under 110 ℃ of temperature conditions of vacuum drying oven, be dried 4 hours, N in Muffle furnace
2under the lower 550 ℃ of temperature conditions of atmosphere, roasting is 4 hours, makes catalyst carrier A6.
Prepare hydrogenation catalyst:
Get respectively ammonium metatungstate 210g, ammonium molybdate 174g and deionized water, be mixed with the limpid dipping solution S4 of 250mlW-Mo.
The limpid dipping solution S4 of the above-mentioned W-Mo of carrier A 6 use is pressed to equi-volume impregnating dipping 4 hours, air-dry, under 110 ℃ of temperature conditions of vacuum drying oven, be dried 4 hours, then N in Muffle furnace
2under the lower 550 ℃ of temperature conditions of atmosphere, roasting is 4 hours, makes hydrogenation catalyst A6-1.
Embodiment 7
Kaolinite Preparation of Catalyst carrier:
The nitric acid that is 66% by 160g alumina powder, 135g CNT, the high sticky SB powder of 35g, 22g SiO 2 powder, 4g sesbania powder, 4g citric acid, 9ml concentration, adding water 220ml stirs, putting into kneading machine mediates 45 minutes, on banded extruder, be extruded into little of the clover of particle diameter 1.2mm, air-dry, under 110 ℃ of temperature conditions of vacuum drying oven, be dried 4 hours, N in Muffle furnace
2under the lower 550 ℃ of temperature conditions of atmosphere, roasting is 4 hours, makes catalyst carrier A7.
Prepare hydrogenation catalyst:
Preparation method is identical with embodiment 1, obtains hydrogenation catalyst A7-1.
Embodiment 8
Kaolinite Preparation of Catalyst carrier:
The nitric acid that is 66% by 90g alumina powder, 180g CNT, the high sticky SB powder of 35g, 4g sesbania powder, 4g citric acid, 9ml concentration, adding water 210ml stirs, putting into kneading machine mediates 45 minutes, on banded extruder, be extruded into little of the clover of particle diameter 1.2mm, air-dry, under 110 ℃ of temperature conditions of vacuum drying oven, be dried 4 hours, N in Muffle furnace
2under the lower 550 ℃ of temperature conditions of atmosphere, roasting is 4 hours, makes catalyst carrier A8.
Prepare hydrogenation catalyst:
Get respectively ammonium metatungstate 45g, ammonium molybdate 42g, nickel nitrate 32g, phosphatase 11 4ml and deionized water, by ammoniacal liquor, allocate pH value and be mixed with the limpid dipping solution S5 of 250mlW-Mo-Ni-P.
The limpid dipping solution S5 of the above-mentioned W-Mo-Ni-P of carrier A 8 use is pressed to equi-volume impregnating dipping 4 hours, air-dry, under 110 ℃ of temperature conditions of vacuum drying oven, be dried 4 hours, then N in Muffle furnace
2under the lower 550 ℃ of temperature conditions of atmosphere, roasting is 4 hours, makes hydrogenation catalyst A8-1.
Comparative example
This comparative example is the conventional preparation method of hydrogenation catalyst, and concrete preparation process is as follows:
Kaolinite Preparation of Catalyst carrier:
The nitric acid that is 66% by 200g alumina powder, the common SB powder of 50g, 6g sesbania powder, 4g citric acid, 9ml concentration, adding water 210ml stirs, putting into kneading machine mediates 45 minutes, on banded extruder, be extruded into little of the clover of particle diameter 1.2mm, air-dry, under 110 ℃ of temperature conditions of vacuum drying oven, be dried 4 hours, N in Muffle furnace
2under the lower 550 ℃ of temperature conditions of atmosphere, roasting is 4 hours, makes catalyst carrier B.
Prepare hydrogenation catalyst:
Preparation method is identical with embodiment 1, obtains hydrogenation catalyst B-1.
The catalyst carrier that embodiment 1-8 and comparative example provide and catalyst property are referring to table 1 and table 2:
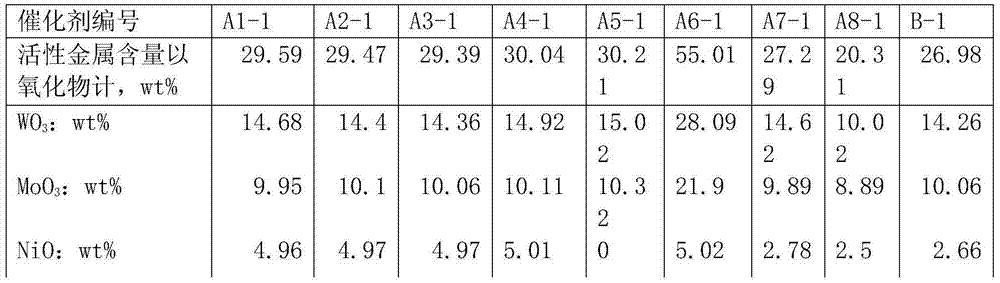
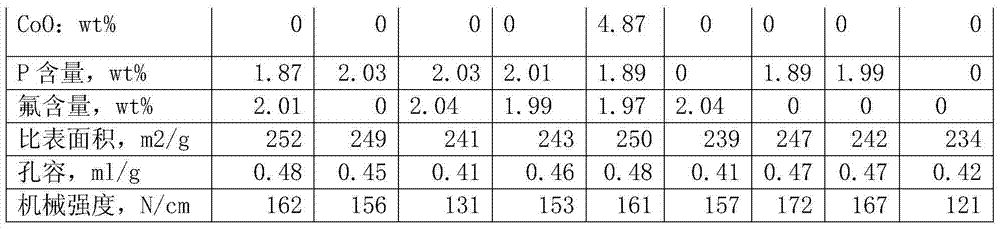
Each catalyst carrier data parameters of table 1
Each catalyst data parameter of table 2
The catalyst that embodiment 1-8 and comparative example are provided carries out performance test, and method of testing is as follows:
Feedstock oil: low minute oil of DCL/Direct coal liquefaction, its character is referring to table 3;
Test condition: on 100ml hydrogenation plant, feedstock oil is carried out to hydrofinishing, after catalyst fills by device designing requirement, first adopt wet method vulcanization process under hydrogen effect, adopt containing the aviation kerosine of 2wt% dimethyl disulfide (DMDS) catalyst is processed, make catalyst be converted into sulphided state, then carry out hydrogenation reaction.Reaction condition is: reaction temperature is 360 ℃, hydrogen dividing potential drop 10MPa, liquid hourly space velocity (LHSV) 1.5h
-1, hydrogen to oil volume ratio 800:1.The test result of device steady and continuous running after 48 hours, 168 hours is referring to table 4 and table 5.
Table 3 feedstock oil nature parameters table
| Project | Feedstock oil |
| Density, kg/m 3 | 948.3 |
| Boiling range | ℃ |
| IBP/10% | 63/177 |
| 50%/90% | 276/355 |
| 95%/EP | -/385 |
| Sulfur content, ug/g | 490 |
| Nitrogen content, ug/g | 4603 |
| Oxygen content, ug/g | 12000 |
The test result of table 4 device steady and continuous running after 48 hours
The test result of table 5 device steady and continuous running after 168 hours
Liquefied coal coil arene content is high, the decline of catalyst activity is mainly because catalyst surface carbon distribution causes, by table 2 and table 4, can be found out, it is all less that the catalyst that the embodiment of the present invention provides moves is continuously active after a period of time, specific area declines, and the anti-carbon performance of catalyst is good.The catalyst carrier that the embodiment of the present invention 1 provides is owing to having added multi-walled carbon nano-tubes, used high sticky SB powder, can make the hole wall of catalyst thin and closely knit, improve under the prerequisite of catalyst pore volume, specific area, improve by a relatively large margin mechanical strength and the anti-carbon deposition ability of catalyst carrier.By part active component nickel is loaded on CNT, avoided forming between nickel and aluminium oxide the nickel aluminate material of difficult reduction, make hydrogenation activity higher, hydrogenation activity is more stable, and in catalyst, introduces fluorine auxiliary agent in the present invention, modulation the acidity of catalyst, make it have certain cracking open loop effect, the hydrodenitrogeneration effect of catalyst is increased, can remove to greatest extent the hetero atoms such as sulphur in liquefied coal coil, nitrogen, oxygen, improve the stability of product.
Claims (10)
1. a liquefied coal coil catalyst carrier for hydrgenating, is characterized in that, the described carrier gross weight of take is 100%, and described carrier comprises the CNT of 5-60wt%, the silica of the aluminium oxide of 32-90wt%, 0-8wt% and the titanium dioxide of 0-8wt%.
2. liquefied coal coil catalyst carrier for hydrgenating according to claim 1, is characterized in that:
Described CNT is multi-walled carbon nano-tubes, and its specific area is 180-350m
2/ g, pore volume is 0.4-1.5cm
3/ g; Described aluminium oxide is activated alumina, and its specific area is 195-410m
2/ g, pore volume is 0.45-1.20cm
3/ g, average pore size are 8-17nm; The average diameter of described silica and described titanium dioxide is 30-70nm.
3. liquefied coal coil catalyst carrier for hydrgenating according to claim 1, is characterized in that, the described carrier gross weight of take is 100%, also comprises NiO and/or the CoO of 2.8-5.6wt% in described carrier.
4. according to the liquefied coal coil catalyst carrier for hydrgenating described in claim 1-3 any one, it is characterized in that:
The specific area of described catalyst carrier for hydrgenating is 250-400m
2/ g, pore volume is 0.45-0.75ml/g, and average pore size is 8-12nm, and diameter is the 75%-85% in Zhan Zong hole, 6-12nm hole; The particle diameter of described catalyst carrier for hydrgenating is 0.8-1.5mm.
5. a preparation method for liquefied coal coil catalyst carrier for hydrgenating, is characterized in that, described method comprises:
CNT, alumina powder are added to the water, and selectively add silica, titanium oxide, auxiliary agent, binding agent, peptizing agent or extrusion aid, in the catalyst carrier that makes to make, contain with the CNT of the total restatement 5-60wt% of carrier, the titanium dioxide of the aluminium oxide of 32-90wt%, 0-8wt%, the silica of 0-8wt%, the consumption of described water is enough to make the material that adds moistening, mix, after kneading, extruded moulding;
Under the product of described extruded moulding is air-dry, 80-120 ℃ vacuum, roasting 3-9h under dry 1-6h, 350-650 ℃ inert gas atmosphere, obtains described catalyst carrier.
6. the preparation method of liquefied coal coil catalyst carrier for hydrgenating according to claim 5, is characterized in that, described CNT also comprises before mixing with other support material:
Nickel salts and/or Cobalt salts are loaded on CNT, and roasting 2-5h under dry 1-5h, 300-500 ℃ inert gas atmosphere under air-dry, 70-120 ℃ vacuum, obtains the CNT of carried metal nickel salt and/or Cobalt salts;
Wherein: the catalyst carrier gross weight making of take is 100%, to take the content of oxide in described carrier be 2.8-5.6wt% for the nickel salts of load and/or Cobalt salts.
7. a liquefied coal coil hydrogenation catalyst, is characterized in that, it comprises:
The carrier that claim 1-4 any one provides;
Active component: VI B family active metal and/or VIII family active metal, described VI B family active metal is W and/or Mo, VIII family active metal is Co and/or Ni;
Wherein: the described hydrogenation catalyst gross weight of take is 100%, to take the content of oxide in described hydrogenation catalyst be 20-55wt% to active metal.
8. liquefied coal coil hydrogenation catalyst according to claim 7, is characterized in that:
The pore volume of described catalyst is 0.35-0.75cm
3/ g, specific area is 220-390m
2/ g.
9. a preparation method for liquefied coal coil hydrogenation catalyst, is characterized in that, described method comprises:
The method providing according to claim 5 or 6 is prepared catalyst carrier for hydrgenating;
The salt of JiangⅥ B family active metal and/or the salt of VIII family active metal load on described carrier, air-dry, and under 100-120 ℃ of vacuum, roasting 2-5h under dry 1-4h, 400-650 ℃ inert gas atmosphere, obtains described catalyst;
Wherein: described VI B family active metal is W and/or Mo, and VIII family active metal is Co and/or Ni; The described hydrogenation catalyst gross weight of take is 100%, and it is 20-55wt% that active metal be take the content of oxide in described hydrogenation catalyst.
10. the application of the liquefied coal coil hydrogenation catalyst described in claim 7 or 8 in liquefied coal coil hydrofining technology.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201310638434.XA CN103638987B (en) | 2013-12-02 | 2013-12-02 | Liquefied coal coil catalyst carrier for hydrgenating, catalyst and preparation method and application |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201310638434.XA CN103638987B (en) | 2013-12-02 | 2013-12-02 | Liquefied coal coil catalyst carrier for hydrgenating, catalyst and preparation method and application |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN103638987A true CN103638987A (en) | 2014-03-19 |
| CN103638987B CN103638987B (en) | 2016-08-17 |
Family
ID=50244353
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201310638434.XA Active CN103638987B (en) | 2013-12-02 | 2013-12-02 | Liquefied coal coil catalyst carrier for hydrgenating, catalyst and preparation method and application |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN103638987B (en) |
Cited By (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN104084227A (en) * | 2014-06-26 | 2014-10-08 | 上海第二工业大学 | Direct coal liquefaction catalytic additive, preparation method and application of direct coal liquefaction catalytic additive |
| CN108329943A (en) * | 2018-03-12 | 2018-07-27 | 江苏华达化工集团有限公司 | A kind of processing method of heavy benzol naphtalene oil |
| CN108409522A (en) * | 2018-03-12 | 2018-08-17 | 江苏华达化工集团有限公司 | A kind of heavy benzol naphthalene production refined naphthalene method |
| CN108579778A (en) * | 2018-03-19 | 2018-09-28 | 煤炭科学技术研究院有限公司 | A kind of carbon nanotube is the hydrogenation catalyst and the preparation method and application thereof of carrier |
| CN108927143A (en) * | 2018-09-06 | 2018-12-04 | 王淑英 | A kind of preparation method for treating open-angle glaucoma pharmaceutical intermediate |
| CN109364919A (en) * | 2018-09-28 | 2019-02-22 | 广东国能中林实业有限公司 | A kind of hydrogenation catalyst based on CNT-alumina/silica composite carrier and its preparation method and application |
| CN110256191A (en) * | 2018-03-12 | 2019-09-20 | 中国科学院宁波材料技术与工程研究所 | A refined naphthalene method for industrial naphthalene production |
| CN110256187A (en) * | 2018-03-12 | 2019-09-20 | 中国科学院宁波材料技术与工程研究所 | A kind of production method of refined naphthalene |
| CN110252273A (en) * | 2018-03-12 | 2019-09-20 | 中国科学院宁波材料技术与工程研究所 | Catalyst carrier for selective hydrogenation of industrial naphthalene and preparation method thereof |
| CN110252325A (en) * | 2018-03-12 | 2019-09-20 | 中国科学院宁波材料技术与工程研究所 | Industrial naphthalene selective hydrogenation catalyst and preparation method thereof |
| CN116532111A (en) * | 2023-04-27 | 2023-08-04 | 清华大学 | Conductive and semi-hydrophobic composite carrier catalyst, preparation method and application thereof |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101085936A (en) * | 2006-06-07 | 2007-12-12 | 神华煤制油研究中心有限公司 | Method for preparing thermal conductive oil by using coal liquefied oil |
| CN101507929A (en) * | 2009-03-20 | 2009-08-19 | 抚顺新瑞催化剂有限公司 | Catalyst for coal tar hydrogenation modification and its preparation method and use |
| CN101513612A (en) * | 2009-03-23 | 2009-08-26 | 浙江大学 | Catalyst for removing nitrogen oxide and organic pollutant discharged in burning and method for preparing same |
| CN102247838A (en) * | 2010-05-21 | 2011-11-23 | 中国石油化工股份有限公司 | Supported palladium catalyst with characteristic of specific infrared absorption |
| CN102284300A (en) * | 2010-06-21 | 2011-12-21 | 中国石油天然气股份有限公司 | A kind of inferior diesel oil hydrogenation treatment catalyst and preparation method |
| CN102701897A (en) * | 2012-05-23 | 2012-10-03 | 河南宝舜化工科技有限公司 | Method for preparing cyclic hydrocarbon compounds by hydrogenation of wash oil fraction |
| CN103182310A (en) * | 2011-12-28 | 2013-07-03 | 中国石油天然气股份有限公司 | A kind of distillate oil hydrogenation treatment catalyst preparation method |
-
2013
- 2013-12-02 CN CN201310638434.XA patent/CN103638987B/en active Active
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101085936A (en) * | 2006-06-07 | 2007-12-12 | 神华煤制油研究中心有限公司 | Method for preparing thermal conductive oil by using coal liquefied oil |
| CN101507929A (en) * | 2009-03-20 | 2009-08-19 | 抚顺新瑞催化剂有限公司 | Catalyst for coal tar hydrogenation modification and its preparation method and use |
| CN101513612A (en) * | 2009-03-23 | 2009-08-26 | 浙江大学 | Catalyst for removing nitrogen oxide and organic pollutant discharged in burning and method for preparing same |
| CN102247838A (en) * | 2010-05-21 | 2011-11-23 | 中国石油化工股份有限公司 | Supported palladium catalyst with characteristic of specific infrared absorption |
| CN102284300A (en) * | 2010-06-21 | 2011-12-21 | 中国石油天然气股份有限公司 | A kind of inferior diesel oil hydrogenation treatment catalyst and preparation method |
| CN103182310A (en) * | 2011-12-28 | 2013-07-03 | 中国石油天然气股份有限公司 | A kind of distillate oil hydrogenation treatment catalyst preparation method |
| CN102701897A (en) * | 2012-05-23 | 2012-10-03 | 河南宝舜化工科技有限公司 | Method for preparing cyclic hydrocarbon compounds by hydrogenation of wash oil fraction |
Non-Patent Citations (1)
| Title |
|---|
| 林磊 等: "用作催化剂载体的碳纳米管-氧化铝复合材料的制备", 《福州大学学报(自然科学版)》, vol. 37, no. 2, 30 April 2009 (2009-04-30), pages 277 - 278 * |
Cited By (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN104084227A (en) * | 2014-06-26 | 2014-10-08 | 上海第二工业大学 | Direct coal liquefaction catalytic additive, preparation method and application of direct coal liquefaction catalytic additive |
| CN110256187B (en) * | 2018-03-12 | 2021-11-02 | 中国科学院宁波材料技术与工程研究所 | A kind of production method of refined naphthalene |
| CN108329943A (en) * | 2018-03-12 | 2018-07-27 | 江苏华达化工集团有限公司 | A kind of processing method of heavy benzol naphtalene oil |
| CN108409522A (en) * | 2018-03-12 | 2018-08-17 | 江苏华达化工集团有限公司 | A kind of heavy benzol naphthalene production refined naphthalene method |
| CN110256191B (en) * | 2018-03-12 | 2021-11-02 | 中国科学院宁波材料技术与工程研究所 | A kind of refined naphthalene method for industrial naphthalene production |
| CN110256191A (en) * | 2018-03-12 | 2019-09-20 | 中国科学院宁波材料技术与工程研究所 | A refined naphthalene method for industrial naphthalene production |
| CN110256187A (en) * | 2018-03-12 | 2019-09-20 | 中国科学院宁波材料技术与工程研究所 | A kind of production method of refined naphthalene |
| CN110252273A (en) * | 2018-03-12 | 2019-09-20 | 中国科学院宁波材料技术与工程研究所 | Catalyst carrier for selective hydrogenation of industrial naphthalene and preparation method thereof |
| CN110252325A (en) * | 2018-03-12 | 2019-09-20 | 中国科学院宁波材料技术与工程研究所 | Industrial naphthalene selective hydrogenation catalyst and preparation method thereof |
| CN108329943B (en) * | 2018-03-12 | 2019-11-05 | 江苏华达化工集团有限公司 | A kind of processing method of heavy benzol naphtalene oil |
| CN108579778A (en) * | 2018-03-19 | 2018-09-28 | 煤炭科学技术研究院有限公司 | A kind of carbon nanotube is the hydrogenation catalyst and the preparation method and application thereof of carrier |
| CN108927143A (en) * | 2018-09-06 | 2018-12-04 | 王淑英 | A kind of preparation method for treating open-angle glaucoma pharmaceutical intermediate |
| CN108927143B (en) * | 2018-09-06 | 2020-01-03 | 北京宝诺康医药科技有限公司 | Preparation method of pharmaceutical intermediate for treating open-angle glaucoma |
| CN109364919B (en) * | 2018-09-28 | 2021-09-07 | 广东国能中林实业有限公司 | A kind of hydrogenation catalyst based on CNT-alumina/silica composite carrier and its preparation method and application |
| CN109364919A (en) * | 2018-09-28 | 2019-02-22 | 广东国能中林实业有限公司 | A kind of hydrogenation catalyst based on CNT-alumina/silica composite carrier and its preparation method and application |
| CN116532111A (en) * | 2023-04-27 | 2023-08-04 | 清华大学 | Conductive and semi-hydrophobic composite carrier catalyst, preparation method and application thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| CN103638987B (en) | 2016-08-17 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN103638987A (en) | Hydrogenation catalyst carrier for coal liquefaction oil, catalyst as well as preparation method and application of hydrogenation catalyst carrier | |
| CN102166521B (en) | Preparation method of hydrofining catalyst | |
| CN102166520B (en) | Hydrofining Catalyst | |
| CN101927197B (en) | Hydrogenation catalyst with gradient decreasing distribution of active metal and acidic additive concentration and preparation method thereof | |
| JP2007512132A5 (en) | ||
| CN101376110B (en) | Preparation of hydrogenation catalyst | |
| CN102794181A (en) | Hydrodeoxygenation catalyst for Fischer Tropsch synthesis oil and preparation method and application of hydrodeoxygenation catalyst | |
| CN101590417A (en) | Hydrofining catalyst and preparation method thereof | |
| CN101590424A (en) | A kind of distillate oil hydrogenation catalyst and preparation method thereof | |
| CN109722280A (en) | A kind of carrier for hydrotreating, catalyst and preparation method and application thereof | |
| CN102847541A (en) | Coal tar hydrodemetalization catalyst and preparation method thereof | |
| CN106914251A (en) | Residual oil hydrodesulfurization catalyst and preparation method thereof | |
| CN102039148B (en) | A kind of preparation method of petroleum wax hydrorefining catalyst | |
| CN109718753A (en) | For the carrier of hydrotreating, catalyst and its preparation method and application | |
| JP4689198B2 (en) | Hydrocarbon hydrotreating catalyst, process for producing the same, and hydrotreating process for hydrocarbon oil | |
| CN105233848A (en) | Ni, W and P loaded hydrofining catalyst with three-peak type pore size distribution for anthracene oil, and preparation method and application | |
| CN104001539B (en) | A kind of coal tar hydrocracking pretreatment catalyst and preparation method thereof | |
| CN106622314B (en) | High-activity hydrofining catalyst and preparation method thereof | |
| CN102019201A (en) | A catalyst for heavy distillate oil hydrotreating containing molecular sieves | |
| CN102451748B (en) | A kind of Hydrodewaxing catalyst and preparation method thereof | |
| CN100486699C (en) | Catalyst used for producing refined benzene by hydrogenation, desulfurization and denitrification of crude benzene, and preparation method thereof | |
| CN106669796A (en) | Preparation method of hydro-upgrading catalyst | |
| CN109772400B (en) | Hydrotreating catalyst and preparation method and application thereof | |
| CN110090654A (en) | Hydrotreating catalyst and its preparation method and application | |
| JP2011245418A (en) | Catalyst for hydrotreating heavy oil and method for producing the catalyst |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C41 | Transfer of patent application or patent right or utility model | ||
| TA01 | Transfer of patent application right |
Effective date of registration: 20160321 Address after: 100013 Chaoyang District, Hepingli Youth Ditch Road, No. 5, No. Applicant after: Co., Ltd of coal science and technology academy Address before: 100013 Chaoyang District, Hepingli Youth Ditch Road, No. 5, No. Applicant before: General Research Institute of Coal Science |
|
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant |