CN103242152A - Method for synthesizing compound 3-(benzyloxy)-1-cyclobutanone - Google Patents
Method for synthesizing compound 3-(benzyloxy)-1-cyclobutanone Download PDFInfo
- Publication number
- CN103242152A CN103242152A CN2013101639200A CN201310163920A CN103242152A CN 103242152 A CN103242152 A CN 103242152A CN 2013101639200 A CN2013101639200 A CN 2013101639200A CN 201310163920 A CN201310163920 A CN 201310163920A CN 103242152 A CN103242152 A CN 103242152A
- Authority
- CN
- China
- Prior art keywords
- benzyloxy
- cyclobutanone
- ether
- phenmethyl
- compound
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- GPPSQLLIFNWNSB-UHFFFAOYSA-N 3-phenylmethoxycyclobutan-1-one Chemical compound C1C(=O)CC1OCC1=CC=CC=C1 GPPSQLLIFNWNSB-UHFFFAOYSA-N 0.000 title claims abstract description 33
- 150000001875 compounds Chemical class 0.000 title claims abstract description 15
- 238000000034 method Methods 0.000 title abstract description 8
- 230000002194 synthesizing effect Effects 0.000 title abstract 3
- 238000006243 chemical reaction Methods 0.000 claims abstract description 31
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 claims description 50
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 claims description 27
- QYKIQEUNHZKYBP-UHFFFAOYSA-N Vinyl ether Chemical compound C=COC=C QYKIQEUNHZKYBP-UHFFFAOYSA-N 0.000 claims description 18
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 claims description 15
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 claims description 14
- 238000010189 synthetic method Methods 0.000 claims description 13
- FAGXDAYDIVWFAE-UHFFFAOYSA-N 2,2-dichloro-3-phenylmethoxycyclobutan-1-one Chemical compound C1C(=O)C(Cl)(Cl)C1OCC1=CC=CC=C1 FAGXDAYDIVWFAE-UHFFFAOYSA-N 0.000 claims description 12
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 claims description 12
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 claims description 12
- 238000002360 preparation method Methods 0.000 claims description 12
- KCXMKQUNVWSEMD-UHFFFAOYSA-N benzyl chloride Chemical compound ClCC1=CC=CC=C1 KCXMKQUNVWSEMD-UHFFFAOYSA-N 0.000 claims description 10
- 239000003960 organic solvent Substances 0.000 claims description 10
- RIOQSEWOXXDEQQ-UHFFFAOYSA-N triphenylphosphine Chemical compound C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 RIOQSEWOXXDEQQ-UHFFFAOYSA-N 0.000 claims description 10
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 claims description 9
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 claims description 9
- TVZPLCNGKSPOJA-UHFFFAOYSA-N copper zinc Chemical compound [Cu].[Zn] TVZPLCNGKSPOJA-UHFFFAOYSA-N 0.000 claims description 9
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 claims description 8
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 claims description 7
- 229910052757 nitrogen Inorganic materials 0.000 claims description 7
- 239000003513 alkali Substances 0.000 claims description 6
- 125000004969 haloethyl group Chemical group 0.000 claims description 6
- 229910052500 inorganic mineral Inorganic materials 0.000 claims description 6
- 239000011707 mineral Substances 0.000 claims description 6
- XHXFXVLFKHQFAL-UHFFFAOYSA-N phosphoryl trichloride Chemical compound ClP(Cl)(Cl)=O XHXFXVLFKHQFAL-UHFFFAOYSA-N 0.000 claims description 6
- -1 BOE Substances 0.000 claims description 5
- DKGAVHZHDRPRBM-UHFFFAOYSA-N Tert-Butanol Chemical compound CC(C)(C)O DKGAVHZHDRPRBM-UHFFFAOYSA-N 0.000 claims description 5
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 claims description 5
- 229960001701 chloroform Drugs 0.000 claims description 5
- LPNYRYFBWFDTMA-UHFFFAOYSA-N potassium tert-butoxide Chemical compound [K+].CC(C)(C)[O-] LPNYRYFBWFDTMA-UHFFFAOYSA-N 0.000 claims description 5
- 239000007795 chemical reaction product Substances 0.000 claims description 4
- 229910052736 halogen Inorganic materials 0.000 claims description 4
- 150000002367 halogens Chemical class 0.000 claims description 4
- 239000002904 solvent Substances 0.000 claims description 4
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 claims description 3
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 claims description 3
- 229910052794 bromium Inorganic materials 0.000 claims description 3
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 claims description 2
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 claims description 2
- ZAFNJMIOTHYJRJ-UHFFFAOYSA-N Diisopropyl ether Chemical compound CC(C)OC(C)C ZAFNJMIOTHYJRJ-UHFFFAOYSA-N 0.000 claims description 2
- BZLVMXJERCGZMT-UHFFFAOYSA-N Methyl tert-butyl ether Chemical compound COC(C)(C)C BZLVMXJERCGZMT-UHFFFAOYSA-N 0.000 claims description 2
- 239000003054 catalyst Substances 0.000 claims description 2
- 239000000460 chlorine Substances 0.000 claims description 2
- 229910052801 chlorine Inorganic materials 0.000 claims description 2
- 239000011630 iodine Substances 0.000 claims description 2
- 229910052740 iodine Inorganic materials 0.000 claims description 2
- 239000011701 zinc Substances 0.000 claims description 2
- 229910052725 zinc Inorganic materials 0.000 claims description 2
- 239000002994 raw material Substances 0.000 abstract description 6
- 238000003786 synthesis reaction Methods 0.000 abstract description 5
- 230000015572 biosynthetic process Effects 0.000 abstract description 2
- 230000008030 elimination Effects 0.000 abstract description 2
- 238000003379 elimination reaction Methods 0.000 abstract description 2
- 238000004519 manufacturing process Methods 0.000 abstract description 2
- 238000007363 ring formation reaction Methods 0.000 abstract description 2
- 238000006266 etherification reaction Methods 0.000 abstract 1
- 230000026030 halogenation Effects 0.000 abstract 1
- 238000005658 halogenation reaction Methods 0.000 abstract 1
- 150000005172 methylbenzenes Chemical class 0.000 abstract 1
- 238000006798 ring closing metathesis reaction Methods 0.000 abstract 1
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 15
- 238000001035 drying Methods 0.000 description 15
- 238000001556 precipitation Methods 0.000 description 12
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 12
- LCGLNKUTAGEVQW-UHFFFAOYSA-N Dimethyl ether Chemical compound COC LCGLNKUTAGEVQW-UHFFFAOYSA-N 0.000 description 9
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 9
- 125000005998 bromoethyl group Chemical group 0.000 description 9
- 238000005406 washing Methods 0.000 description 9
- 239000000047 product Substances 0.000 description 8
- 238000003756 stirring Methods 0.000 description 7
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 6
- 238000004821 distillation Methods 0.000 description 6
- 239000000243 solution Substances 0.000 description 6
- MZRVEZGGRBJDDB-UHFFFAOYSA-N N-Butyllithium Chemical compound [Li]CCCC MZRVEZGGRBJDDB-UHFFFAOYSA-N 0.000 description 4
- VLTRZXGMWDSKGL-UHFFFAOYSA-N perchloric acid Chemical compound OCl(=O)(=O)=O VLTRZXGMWDSKGL-UHFFFAOYSA-N 0.000 description 4
- 238000011084 recovery Methods 0.000 description 4
- 238000005303 weighing Methods 0.000 description 4
- 239000007864 aqueous solution Substances 0.000 description 3
- 230000006837 decompression Effects 0.000 description 3
- 230000000694 effects Effects 0.000 description 3
- 238000000605 extraction Methods 0.000 description 3
- 239000000706 filtrate Substances 0.000 description 3
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 3
- 235000010755 mineral Nutrition 0.000 description 3
- 239000011259 mixed solution Substances 0.000 description 3
- 239000003208 petroleum Substances 0.000 description 3
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 3
- 235000017557 sodium bicarbonate Nutrition 0.000 description 3
- 150000003457 sulfones Chemical class 0.000 description 3
- HRPGUVFWKDCSSX-UHFFFAOYSA-N 2-phenylmethoxycyclobutan-1-one Chemical compound O=C1CCC1OCC1=CC=CC=C1 HRPGUVFWKDCSSX-UHFFFAOYSA-N 0.000 description 2
- 239000003814 drug Substances 0.000 description 2
- 230000003647 oxidation Effects 0.000 description 2
- 238000007254 oxidation reaction Methods 0.000 description 2
- HVELAOQZAFXLRD-UHFFFAOYSA-N 1,3-dibromopropan-2-yloxymethylbenzene Chemical compound BrCC(CBr)OCC1=CC=CC=C1 HVELAOQZAFXLRD-UHFFFAOYSA-N 0.000 description 1
- 101900297506 Human immunodeficiency virus type 1 group M subtype B Reverse transcriptase/ribonuclease H Proteins 0.000 description 1
- GOOHAUXETOMSMM-UHFFFAOYSA-N Propylene oxide Chemical compound CC1CO1 GOOHAUXETOMSMM-UHFFFAOYSA-N 0.000 description 1
- RCTYPNKXASFOBE-UHFFFAOYSA-M chloromercury Chemical compound [Hg]Cl RCTYPNKXASFOBE-UHFFFAOYSA-M 0.000 description 1
- KCXMKQUNVWSEMD-PTQBSOBMSA-N chloromethylbenzene Chemical group Cl[13CH2]C1=CC=CC=C1 KCXMKQUNVWSEMD-PTQBSOBMSA-N 0.000 description 1
- 238000004440 column chromatography Methods 0.000 description 1
- 238000006298 dechlorination reaction Methods 0.000 description 1
- 229940079593 drug Drugs 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 239000012467 final product Substances 0.000 description 1
- 238000004817 gas chromatography Methods 0.000 description 1
- 125000005843 halogen group Chemical group 0.000 description 1
- 238000003987 high-resolution gas chromatography Methods 0.000 description 1
- 238000009776 industrial production Methods 0.000 description 1
- 239000003112 inhibitor Substances 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 238000000425 proton nuclear magnetic resonance spectrum Methods 0.000 description 1
- 239000003419 rna directed dna polymerase inhibitor Substances 0.000 description 1
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 1
- 229920002554 vinyl polymer Polymers 0.000 description 1
Images
Landscapes
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
Abstract
The invention provides a new method for synthesizing compound 3-(benzyloxy)-1-cyclobutanone, belonging to the technical field of organic synthesis. The method comprises the steps of performing five steps of reaction, namely etherification, halogenation, elimination, ring closure and dechloridation by taking halogenated methyl benzene as an initial raw material, and synthesizing to obtain the target product 3-(benzyloxy)-1-cyclobutanone with high yield and high purity. According to the method, the raw materials are low-cost and easily available, the reaction conditions are mild (especially the ring closure reaction is carried out at room temperature), the synthesis cost can be effectively lowered, the requirement on equipment can be reduced, and the method has good industrialization production prospects.
Description
Technical field
The invention belongs to the organic chemical synthesis field, relate to the synthetic method of a kind of compound 3-(benzyloxy)-1-cyclobutanone.
Background technology
Compound 3-(benzyloxy)-1-cyclobutanone is important organic synthesis intermediate, is the synthesis material of a lot of medicines and auxiliary, for example preparation of HIV-1 reverse transcriptase inhibitors, PLK inhibitor etc.; Also can be used for the preparation of medicine and medical developer.Its structural formula is:
Synthetic for 3-(benzyloxy)-1-cyclobutanone, domestic do not have a bibliographical information, abroad more existing documents or patent report,
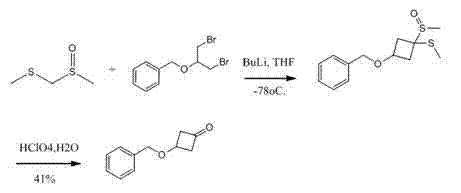
Bioorganic ﹠amp; Biological and the pharmaceutical chemistry wall bulletin of Medicinal Chemistry Letters(), 22 (12), 4064-4067; 2012 have reported the synthetic method of 3-(benzyloxy)-1-cyclobutanone; be with semi-synthetic intermediate 2-benzyloxy-1; 3-dibromopropane and methyl sulfo-first sulfone are raw material; under the butyllithium effect; subzero 78 ℃ of degree reactions; make 1-methylsulfinyl-1-methylthio group-3-benzyloxy tetramethylene, obtained 3-(benzyloxy)-1-cyclobutanone by the perchloric acid oxidation then.Because semi-synthetic intermediate 2-benzyloxy-1,3-dibromopropane and methyl sulfo-first sulfone are difficult in market buy, cost is higher, and the condition harshness, is not easy to amplify produce; Total recovery is not high yet.Its circuit is as follows:
WO2009114512 provides the method for a kind of synthetic 3-(benzyloxy)-1-cyclobutanone; be raw material with brooethyl propylene oxide and cylite; reaction makes 2-benzyloxy-1 under 160 ℃ under the effect of mercury chloride; the 3-dibromopropane; again under the butyllithium effect; 2-benzyloxy-1; 3-dibromopropane and methyl sulfo-first sulfone are in-78 ℃ of reactions down; make 1-methylsulfinyl-1-methylthio group-3-benzyloxy tetramethylene, obtained 3-(benzyloxy)-1-cyclobutanone by the perchloric acid oxidation then, this method has been finished reaction by three steps; purifying products uses column chromatography; the cost height, yield is low, is unfavorable for suitability for industrialized production.Its synthetic line is as follows:
Summary of the invention
The objective of the invention is the problem at the prior art existence, the novel method of synthetic compound 3-(benzyloxy)-1-cyclobutanone that a kind of cost is low, yield is high is provided.
The synthetic method of The compounds of this invention 3-(benzyloxy)-1-cyclobutanone comprises following processing step:
(1) preparation of BOE
Under the mineral alkali condition, monochloromethyl benzene and ethylene glycol are with the mol ratio of 1:2 ~ 1:10, and ℃ reaction 1 ~ 6h in room temperature ~ 70 obtains BOE---intermediate 2;
Described monochloromethyl benzene is chloromethylbenzene or brooethyl benzene;
Described mineral alkali is sodium hydroxide or potassium hydroxide; The molar weight of mineral alkali is 1 ~ 1.5 times of monochloromethyl benzene.
(2) preparation of haloethyl phenmethyl ether
In organic solvent, BOE, triphenylphosphine, halogen in room temperature reaction 1 ~ 5h, obtain haloethyl phenmethyl ether---intermediate 3 with the mol ratio of 1:1:0.5 ~ 1:1.5:1.5;
Described halogen is chlorine, bromine or iodine;
Described organic solvent is methylene dichloride, trichloromethane or tetracol phenixin.
(3) preparation of phenmethyl vinyl ether
Be solvent with the trimethyl carbinol, haloethyl phenmethyl ether and potassium tert.-butoxide are with the mol ratio of 1:1.0 ~ 1:1.5, and ℃ reaction 2 ~ 8h in room temperature ~ 70 obtains the phenmethyl vinyl ether---intermediate 4.
(4) benzyloxy-2, the preparation of 2-dichloro cyclobutanone
Under the nitrogen protection, in organic solvent, be catalyzer with the zinc-copper, phenmethyl vinyl ether and trichoroacetic chloride are with the mol ratio of 1:1 ~ 1:3, and room temperature reaction 1 ~ 3h gets benzyloxy-2,2-dichloro cyclobutanone---intermediate 5;
Or under the nitrogen protection, in organic solvent, be catalyzer with the zinc-copper, make phenmethyl vinyl ether, trichoroacetic chloride, phosphorus oxychloride with the mol ratio of 1:1:0.2 ~ 1:3:0.3, in room temperature reaction 0.5 ~ 3.0h, get benzyloxy-2,2-dichloro cyclobutanone---intermediate 5;
Described zinc-copper catalyst consumption is 0.4 ~ 1 times of phenmethyl vinyl ether molar weight;
Described organic solvent is methyl tertiary butyl ether, ether or isopropyl ether.
(5) preparation of 3-(benzyloxy)-1-cyclobutanone
Be solvent with the aqueous acetic acid, benzyloxy-2,2-dichloro cyclobutanone and metallic zinc are with the mol ratio of 1:2 ~ 1:3.5, and room temperature reaction 1 ~ 3h gets final product 6---3-(benzyloxy)-1-cyclobutanone;
In the described aqueous acetic acid, the percent by volume of acetic acid is 40 ~ 60%.
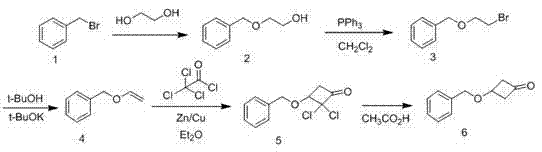
The synthetic route of 3-of the present invention (benzyloxy)-1-cyclobutanone is as follows:
Synthetic product of the present invention is through proton nmr spectra, and high resolution gas chromatography is measured, and shows that the finished product that the present invention synthesizes are 3-(benzyloxy)-1-cyclobutanone, and purity is 95 ~ 98%, and yield is more than 50%.
The present invention is starting raw material with monochloromethyl benzene, pass through into ether, halo, elimination, pass ring, dechlorination five steps reaction, high yield, high purity have been synthesized target product 3-(benzyloxy)-1-cyclobutanone, its raw material is cheap and easy to get, reaction conditions gentleness (particularly ring closure reaction is at room temperature to carry out), effectively reduce synthetic cost, reduced the requirement to equipment simultaneously, have good industrial production foreground.
Description of drawings
Fig. 1 is the gas-chromatography of synthetic product of the present invention;
The nuclear magnetic spectrogram that Fig. 2 synthesizes for the present invention.
Embodiment
Be further described below by the synthetic method of specific embodiment to 3-of the present invention (benzyloxy)-1-cyclobutanone.
(1) BOE is synthetic
In the reaction flask that thermometer, prolong are housed, add 800mL(15.74mol) ethylene glycol, 285.6g(7.14mol) sodium hydroxide, be stirred to dissolving in 40 ℃, slowly add brooethyl benzene 684g(4mol), stirred 1 hour, add 2000mL water, use extracted with diethyl ether, anhydrous sodium sulfate drying, precipitation, get BOE 556g, yield 91%;
(2) the bromo ethyl phenenyl methyl ether is synthetic
Take by weighing triphenylphosphine 832g(3.58mol) be dissolved among the chloroform 2000mL, drip bromine 290g(1.79mol) to the solution becomes yellowly, drip 400g(2.62mol under the room temperature) BOE, 400mL triethylamine and 600mL chloroform mixed solution, dripped off in 1 hour, reacted 0.5 hour, the decompression precipitation, residuum adds chloroform 1000mL, wash anhydrous sodium sulfate drying, distillation with water, get product bromo ethyl phenenyl methyl ether 544g, yield 97%;
(3) the phenmethyl vinyl ether is synthetic
Take by weighing bromo ethyl phenenyl methyl ether 450g(2.1mol) be dissolved in the 1000mL trimethyl carbinol, add potassium tert.-butoxide 330g(2.9mol), 55 ℃ of stirring reactions 2 hours, reaction solution is poured in the 3000mL water, uses petroleum ether extraction, washing, anhydrous sodium sulfate drying, precipitation gets phenmethyl vinyl ether 247g, yield 88%;
(4) benzyloxy-2,2-dichloro cyclobutanone synthetic
Under nitrogen protection, with 160g(1.2mol) the phenmethyl vinyl ether is dissolved in the 3000mL ether, adds zinc-copper catalyzer 50g(0.77mol), drip 300mL(2.7mol under the room temperature) trichoroacetic chloride and 30mL(0.33mol) phosphorus oxychloride, stirred 1 hour, and added sodium bicarbonate aqueous solution 1000mL, get with sherwood oil, washing, anhydrous sodium sulfate drying, precipitation gets benzyloxy-2,2-dichloro cyclobutanone 250g, yield 85%;
(5) 3-(benzyloxy)-1-cyclobutanone is synthetic
With 250g(1mol) benzyloxy-2,2-dichloro cyclobutanone is dissolved in 2000mL water and the 2000mL acetic acid, slowly add zinc powder 200g(3.07mol), stirring at room 1 hour is filtered, filtrate is used extracted with diethyl ether, washing, anhydrous sodium sulfate drying, distillation, get 3-(benzyloxy)-1-cyclobutanone 150g, yield 84%.The end product total recovery is 55.46%.
(1) BOE is synthetic
Thermometer is being housed, add 800mL(15.74mol in the reaction flask of prolong) ethylene glycol, 400g(7.14mol) potassium hydroxide, 70 ℃ are stirred to dissolving, slowly add 522.0g(4.1mol) chloromethylbenzene, stirred 1 hour, and added the water of 2000mL, use extracted with diethyl ether, anhydrous sodium sulfate drying, precipitation gets BOE 586g, yield 96%.
(2) the bromo ethyl phenenyl methyl ether is synthetic
Triphenylphosphine 832g(3.58mol) be dissolved among the methylene dichloride 3000mL Dropwise 5 72g(3.5mol) to the solution becomes yellowly, drip 400g(2.62mol under the room temperature) BOE, 400mL triethylamine and 600mL methylene dichloride mixed solution, dripped off in 1 hour, and dripped off reaction 0.5 hour, the decompression precipitation, residuum adds ether 1000mL, wash anhydrous sodium sulfate drying, distillation with water, get product bromo ethyl phenenyl methyl ether 521g, yield 93%.
(3) the phenmethyl vinyl ether is synthetic
Take by weighing bromo ethyl phenenyl methyl ether 450g(2.1mol) be dissolved in the 1000mL trimethyl carbinol, add potassium tert.-butoxide 330g(2.9mol), 60 ℃ of stirring reactions 2 hours, reaction solution is poured in the 3000mL water, uses petroleum ether extraction, washing, anhydrous sodium sulfate drying, precipitation gets phenmethyl vinyl ether 252g, yield 90%.
(4) 3-(benzyloxy)-1-cyclobutanone is synthetic
Under nitrogen protection, with 160g(1.2mol) the phenmethyl vinyl ether is dissolved in the 3000mL ether, adds zinc-copper catalyzer 50g(0.77mol), drip 300mL(2.7mol under the room temperature) trichoroacetic chloride, stirred 1 hour, and added sodium bicarbonate aqueous solution 1000mL, get with sherwood oil, washing, anhydrous sodium sulfate drying, precipitation gets benzyloxy-2,2-dichloro cyclobutanone 232g, yield 79%;
(5) 3-(benzyloxy)-1-cyclobutanone is synthetic
With 125g(0.5mol) benzyloxy-2,2-dichloro cyclobutanone is dissolved in 1000mL water and the 1000mL acetic acid, slowly add zinc powder 100g(1.5mol), stirring at room 1 hour is filtered, filtrate is used extracted with diethyl ether, washing, anhydrous sodium sulfate drying, distillation, get 3-(benzyloxy)-1-cyclobutanone 77.6g, yield 87%.The end product total recovery is 55.22%.
(1) BOE is synthetic
Thermometer is being housed, add 800mL(15.74mol in the reaction flask of prolong) ethylene glycol, 400g(7.14mol) potassium hydroxide, stirring at room to the dissolving, slowly add 522.0g(4.1mol) chloromethylbenzene, stirred 6 hours, and added the water of 2000mL, use extracted with diethyl ether, anhydrous sodium sulfate drying, precipitation gets BOE 525g, yield 86%.
(2) the bromo ethyl phenenyl methyl ether is synthetic
Triphenylphosphine 832g(3.58mol) be dissolved among the methylene dichloride 3000mL Dropwise 5 72g(3.5mol) to the solution becomes yellowly, drip 400g(2.62mol under the room temperature) BOE, 400mL triethylamine and 600mL methylene dichloride mixed solution, dripped off in 1 hour, and dripped off reaction 0.5 hour, the decompression precipitation, residuum adds ether 1000mL, wash anhydrous sodium sulfate drying, distillation with water, get product bromo ethyl phenenyl methyl ether 521g, yield 93%.
(3) the phenmethyl vinyl ether is synthetic
Take by weighing bromo ethyl phenenyl methyl ether 450g(2.1mol) be dissolved in the 1000mL trimethyl carbinol, add potassium tert.-butoxide 330g(2.9mol), stirring at room reaction 8 hours, reaction solution is poured in the 3000mL water, uses petroleum ether extraction, washing, anhydrous sodium sulfate drying, precipitation gets phenmethyl vinyl ether 246g, yield 88%.
(4) benzyloxy-2,2-dichloro cyclobutanone synthetic
Under nitrogen protection, with 160g(1.2mol) the phenmethyl vinyl ether is dissolved in the 3000mL ether, adds zinc-copper catalyzer 78g(1.2mol), drip 300mL(2.7mol under the room temperature) trichoroacetic chloride, stirred 1 hour, and added sodium bicarbonate aqueous solution 1000mL, get with sherwood oil, washing, anhydrous sodium sulfate drying, precipitation gets benzyloxy-2,2-dichloro cyclobutanone 246g, yield 84%;
(5) 3-(benzyloxy)-1-cyclobutanone is synthetic
With 125g(0.5mol) benzyloxy-2,2-dichloro cyclobutanone is dissolved in 1000mL water and the 1000mL acetic acid, slowly add zinc powder 112g(1.75mol), stirring at room 1.5 hours is filtered, filtrate is used extracted with diethyl ether, washing, anhydrous sodium sulfate drying, distillation, get 3-(benzyloxy)-1-cyclobutanone 78.5g, yield 88%.The end product total recovery is 52%.
Claims (8)
1. the synthetic method of compound 3-(benzyloxy)-1-cyclobutanone comprises following processing step:
(1) preparation of BOE: under the mineral alkali condition, monochloromethyl benzene and ethylene glycol are with the mol ratio of 1:2~1:10, and ℃ reaction 1~6h obtains BOE in room temperature~70;
(2) preparation of haloethyl phenmethyl ether: in organic solvent, BOE, triphenylphosphine, halogen in room temperature reaction 1~5h, obtain haloethyl phenmethyl ether with the mol ratio of 1:1:0.5~1:1.5:1.5;
(3) preparation of phenmethyl vinyl ether: be solvent with the trimethyl carbinol, haloethyl phenmethyl ether and potassium tert.-butoxide are with the mol ratio of 1:1.0~1:1.5, and ℃ reaction 2~8h obtains the phenmethyl vinyl ether in room temperature~70;
(4) benzyloxy-2, the preparation of 2-dichloro cyclobutanone: under the nitrogen protection, in organic solvent, be catalyzer with the zinc-copper, phenmethyl vinyl ether and trichoroacetic chloride are with the mol ratio of 1:1~1:3, and room temperature reaction 1~3h gets benzyloxy-2,2-dichloro cyclobutanone; Or under the nitrogen protection, in organic solvent, be catalyzer with the zinc-copper, make phenmethyl vinyl ether, trichoroacetic chloride, phosphorus oxychloride with the mol ratio of 1:1:0.2~1:3:0.3, in room temperature reaction 0.5~3.0h, get benzyloxy-2,2-dichloro cyclobutanone;
(5) preparation of 3-(benzyloxy)-1-cyclobutanone: be solvent with the aqueous acetic acid, benzyloxy-2,2-dichloro cyclobutanone and metallic zinc are with the mol ratio of 1:2~1:3.5, and room temperature reaction 1~3h gets end product 3-(benzyloxy)-1-cyclobutanone.
2. the synthetic method of compound 3-(benzyloxy)-1-cyclobutanone according to claim 1, it is characterized in that: the described monochloromethyl benzene of step (1) is chloromethylbenzene or brooethyl benzene.
3. the synthetic method of compound 3-(benzyloxy)-1-cyclobutanone according to claim 1, it is characterized in that: the described mineral alkali of step (1) is sodium hydroxide or potassium hydroxide; The molar weight of mineral alkali is 1~1.5 times of monochloromethyl benzene.
4. the synthetic method of compound 3-(benzyloxy)-1-cyclobutanone according to claim 1, it is characterized in that: the described halogen of step (2) is chlorine, bromine or iodine.
5. the synthetic method of compound 3-(benzyloxy)-1-cyclobutanone according to claim 1, it is characterized in that: the described organic solvent of step (2) is methylene dichloride, trichloromethane or tetracol phenixin.
6. the synthetic method of compound 3-(benzyloxy)-1-cyclobutanone according to claim 1, it is characterized in that: the described zinc-copper catalyst consumption of step (4) is 0.4~1 times of phenmethyl vinyl ether molar weight.
7. the synthetic method of compound 3-(benzyloxy)-1-cyclobutanone according to claim 1, it is characterized in that: the described organic solvent of step (4) is methyl tertiary butyl ether, ether or isopropyl ether.
8. the synthetic method of compound 3-(benzyloxy)-1-cyclobutanone according to claim 1, it is characterized in that: in the described aqueous acetic acid of step (5), the percent by volume of acetic acid is 40~60%.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201310163920.0A CN103242152B (en) | 2013-05-07 | 2013-05-07 | The synthetic method of compound 3-(benzyloxy)-1-cyclobutanone |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201310163920.0A CN103242152B (en) | 2013-05-07 | 2013-05-07 | The synthetic method of compound 3-(benzyloxy)-1-cyclobutanone |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN103242152A true CN103242152A (en) | 2013-08-14 |
| CN103242152B CN103242152B (en) | 2015-12-02 |
Family
ID=48922061
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201310163920.0A Expired - Fee Related CN103242152B (en) | 2013-05-07 | 2013-05-07 | The synthetic method of compound 3-(benzyloxy)-1-cyclobutanone |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN103242152B (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN118496070A (en) * | 2024-04-12 | 2024-08-16 | 武汉软件工程职业学院(武汉开放大学) | Synthesis method of 3- (benzyloxy) -1-cyclobutanone |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2002060362A (en) * | 2000-08-17 | 2002-02-26 | Nippon Synthetic Chem Ind Co Ltd:The | Method for producing ethylene glycol monobenzyl ether |
| WO2004056725A1 (en) * | 2002-12-20 | 2004-07-08 | Ge Healthcare Limited | Solid-phase preparation of 18f-labelled amino acids |
| WO2011006000A1 (en) * | 2009-07-08 | 2011-01-13 | Haiyan Liu | Berberine derivatives useful for modulating lipid levels and their methods of synthesis |
-
2013
- 2013-05-07 CN CN201310163920.0A patent/CN103242152B/en not_active Expired - Fee Related
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2002060362A (en) * | 2000-08-17 | 2002-02-26 | Nippon Synthetic Chem Ind Co Ltd:The | Method for producing ethylene glycol monobenzyl ether |
| WO2004056725A1 (en) * | 2002-12-20 | 2004-07-08 | Ge Healthcare Limited | Solid-phase preparation of 18f-labelled amino acids |
| WO2011006000A1 (en) * | 2009-07-08 | 2011-01-13 | Haiyan Liu | Berberine derivatives useful for modulating lipid levels and their methods of synthesis |
Non-Patent Citations (2)
| Title |
|---|
| LAURENT MARTARELLO等: "Synthesis of syn- and anti-1-Amino-3-[18F]fluoromethyl-cyclobutane-1-carboxylic Acid (FMACBC), Potential PET Ligands for Tumor Detection", 《J. MED. CHEM.》, vol. 45, no. 11, 26 April 2002 (2002-04-26), pages 2250 - 2259, XP 002276531, DOI: doi:10.1021/jm010242p * |
| VIJAY KAIWAR等: "Synthesis of 9- [ cis-3-(hydroxymethyl)cyclobutyl] -adenine and-guanine", 《J . CHEM. SOC. PERKIN TRANS.》, vol. 1, 31 December 1995 (1995-12-31), pages 2281 - 2287, XP 000926767, DOI: doi:10.1039/p19950002281 * |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN118496070A (en) * | 2024-04-12 | 2024-08-16 | 武汉软件工程职业学院(武汉开放大学) | Synthesis method of 3- (benzyloxy) -1-cyclobutanone |
Also Published As
| Publication number | Publication date |
|---|---|
| CN103242152B (en) | 2015-12-02 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN107629032A (en) | A kind of preparation method of cyclic sulfates | |
| CN107337596B (en) | Preparation method of 4-methoxy-1, 3-phthalic acid | |
| CN105367526A (en) | Preparation method of high-purity n-butylphthalide | |
| CN103242152B (en) | The synthetic method of compound 3-(benzyloxy)-1-cyclobutanone | |
| CN103012275A (en) | Method for producing high-purity N-alkyl imidazole | |
| CN107629004A (en) | 13The pyraoxystrobin and its synthetic method of C flag | |
| CN109956871A (en) | A kind of preparation method of 3,4,5-trifluoro-2'-nitrobiphenyl | |
| CN104262450A (en) | Method for preparing and refining eplerenone | |
| CN102701964A (en) | Method for synthesizing 4, 4' -biphenyldicarboxylic acid | |
| CN104326936B (en) | The synthetic method of trans N-ethyl-N-(2 '-alkyl phenyl)-2-butylene acid amides | |
| CN110452198B (en) | Preparation method of feloxicib | |
| CN103951538A (en) | Method for synthesizing diarylketone under catalysis of nickel at normal pressure | |
| CN109627228A (en) | A kind of synthetic method of 3- oxetanone | |
| CN109422709A (en) | A kind of synthetic method of oxetanone | |
| CN101659612B (en) | Selective esterification method | |
| CN105566201A (en) | Green synthesis method of carbazole oxime ester photoinitiator | |
| CN105949217A (en) | Preparation method of milbemycin oxime intermediate | |
| CN104447262A (en) | Method for synthesizing 1-chloro-1-chloroacetyl-cyclopropane by adopting one-pot reaction | |
| CN103224485B (en) | Method for preparing 3-thionphene ethyl formate compound | |
| CN104961629A (en) | Synthetic method of pentafluorophenol | |
| CN103539666B (en) | Preparation method of 2-methyl-3-butenoic acid ester | |
| CN103613479B (en) | A kind of preparation method of aromatic carboxylic acid compound | |
| JP6235932B2 (en) | Method for producing 2-cyanophenylboronic acid derivative | |
| CN102040650B (en) | Method for constructing sterides-6beta, 7beta-methylene structure by cascade reaction | |
| CN108276254A (en) | A method of the synthesis chloro- 4- trifloro methyl phenols of 2- |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C41 | Transfer of patent application or patent right or utility model | ||
| TA01 | Transfer of patent application right |
Effective date of registration: 20150915 Address after: 730010 Gansu Province Chengguan District of Lanzhou City Yantan Power Plant Road Building Room 2404 Applicant after: LANZHOU AGLI BIOCHEMICAL TECHNOLOGY Co.,Ltd. Address before: 730020, Gansu Province, Chengguan District, Lanzhou Province, 177 section 7, 3 floor, 301 Jia Lu Road House Applicant before: LANZHOU SHUYA BIOCHEMICAL TECHNOLOGY Co.,Ltd. |
|
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| CF01 | Termination of patent right due to non-payment of annual fee | ||
| CF01 | Termination of patent right due to non-payment of annual fee |
Granted publication date: 20151202 |