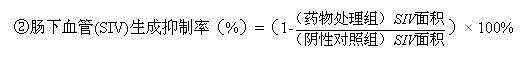CN103142601A - Application of PCI (Percutaneous Coronary Intervention)-32765 for preparing anti-angiogenic medicines - Google Patents
Application of PCI (Percutaneous Coronary Intervention)-32765 for preparing anti-angiogenic medicines Download PDFInfo
- Publication number
- CN103142601A CN103142601A CN2013100792612A CN201310079261A CN103142601A CN 103142601 A CN103142601 A CN 103142601A CN 2013100792612 A CN2013100792612 A CN 2013100792612A CN 201310079261 A CN201310079261 A CN 201310079261A CN 103142601 A CN103142601 A CN 103142601A
- Authority
- CN
- China
- Prior art keywords
- pci
- blood vessel
- angiogenic
- application
- medicine
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 239000003814 drug Substances 0.000 title claims abstract description 57
- 229940079593 drug Drugs 0.000 title abstract description 35
- 230000001772 anti-angiogenic effect Effects 0.000 title abstract description 8
- 238000013146 percutaneous coronary intervention Methods 0.000 title abstract 4
- XYFPWWZEPKGCCK-GOSISDBHSA-N ibrutinib Chemical compound C1=2C(N)=NC=NC=2N([C@H]2CN(CCC2)C(=O)C=C)N=C1C(C=C1)=CC=C1OC1=CC=CC=C1 XYFPWWZEPKGCCK-GOSISDBHSA-N 0.000 claims abstract description 83
- 239000002177 L01XE27 - Ibrutinib Substances 0.000 claims abstract description 73
- 229960001507 ibrutinib Drugs 0.000 claims abstract description 73
- 206010028980 Neoplasm Diseases 0.000 claims abstract description 13
- 230000007850 degeneration Effects 0.000 claims description 21
- 206010025421 Macule Diseases 0.000 claims description 19
- VEEGZPWAAPPXRB-BJMVGYQFSA-N (3e)-3-(1h-imidazol-5-ylmethylidene)-1h-indol-2-one Chemical class O=C1NC2=CC=CC=C2\C1=C/C1=CN=CN1 VEEGZPWAAPPXRB-BJMVGYQFSA-N 0.000 claims description 10
- 239000007924 injection Substances 0.000 claims description 9
- 238000002347 injection Methods 0.000 claims description 9
- 239000007788 liquid Substances 0.000 claims description 9
- 210000001367 artery Anatomy 0.000 claims description 8
- 239000002552 dosage form Substances 0.000 claims description 6
- 239000002246 antineoplastic agent Substances 0.000 claims description 4
- 239000002775 capsule Substances 0.000 claims description 4
- 239000008187 granular material Substances 0.000 claims description 4
- 239000003826 tablet Substances 0.000 claims description 4
- 229940041181 antineoplastic drug Drugs 0.000 claims description 3
- 210000004877 mucosa Anatomy 0.000 claims description 2
- 241000252212 Danio rerio Species 0.000 abstract description 68
- 238000002474 experimental method Methods 0.000 abstract description 21
- 206010064930 age-related macular degeneration Diseases 0.000 abstract description 12
- 230000033115 angiogenesis Effects 0.000 abstract description 10
- 230000012010 growth Effects 0.000 abstract description 8
- 208000002780 macular degeneration Diseases 0.000 abstract description 6
- 201000011510 cancer Diseases 0.000 abstract description 5
- 230000000259 anti-tumor effect Effects 0.000 abstract description 4
- 230000000694 effects Effects 0.000 abstract description 4
- 230000003285 pharmacodynamic effect Effects 0.000 abstract description 3
- 238000001727 in vivo Methods 0.000 abstract description 2
- 230000002491 angiogenic effect Effects 0.000 abstract 1
- 210000004204 blood vessel Anatomy 0.000 description 63
- 210000001161 mammalian embryo Anatomy 0.000 description 42
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 29
- 230000000968 intestinal effect Effects 0.000 description 22
- 230000001629 suppression Effects 0.000 description 19
- 210000002257 embryonic structure Anatomy 0.000 description 13
- 230000004720 fertilization Effects 0.000 description 13
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 13
- 238000000034 method Methods 0.000 description 11
- 230000005764 inhibitory process Effects 0.000 description 9
- 230000008175 fetal development Effects 0.000 description 8
- 230000000630 rising effect Effects 0.000 description 8
- 230000002792 vascular Effects 0.000 description 8
- 206010033128 Ovarian cancer Diseases 0.000 description 7
- 230000008569 process Effects 0.000 description 7
- 239000004037 angiogenesis inhibitor Substances 0.000 description 6
- 229940121369 angiogenesis inhibitor Drugs 0.000 description 6
- 230000002401 inhibitory effect Effects 0.000 description 6
- 239000013642 negative control Substances 0.000 description 6
- 238000009827 uniform distribution Methods 0.000 description 6
- 206010020880 Hypertrophy Diseases 0.000 description 5
- 230000005856 abnormality Effects 0.000 description 5
- 201000010099 disease Diseases 0.000 description 5
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 5
- 238000002360 preparation method Methods 0.000 description 5
- 230000009261 transgenic effect Effects 0.000 description 5
- 238000002054 transplantation Methods 0.000 description 5
- 238000011282 treatment Methods 0.000 description 5
- 230000015572 biosynthetic process Effects 0.000 description 4
- 230000017531 blood circulation Effects 0.000 description 4
- 210000004027 cell Anatomy 0.000 description 4
- GVPFVAHMJGGAJG-UHFFFAOYSA-L cobalt dichloride Chemical compound [Cl-].[Cl-].[Co+2] GVPFVAHMJGGAJG-UHFFFAOYSA-L 0.000 description 4
- 229940097267 cobaltous chloride Drugs 0.000 description 4
- 238000011156 evaluation Methods 0.000 description 4
- 238000013507 mapping Methods 0.000 description 4
- 239000013641 positive control Substances 0.000 description 4
- 239000000243 solution Substances 0.000 description 4
- 230000001225 therapeutic effect Effects 0.000 description 4
- WLCZTRVUXYALDD-IBGZPJMESA-N 7-[[(2s)-2,6-bis(2-methoxyethoxycarbonylamino)hexanoyl]amino]heptoxy-methylphosphinic acid Chemical compound COCCOC(=O)NCCCC[C@H](NC(=O)OCCOC)C(=O)NCCCCCCCOP(C)(O)=O WLCZTRVUXYALDD-IBGZPJMESA-N 0.000 description 3
- 201000009030 Carcinoma Diseases 0.000 description 3
- 206010027476 Metastases Diseases 0.000 description 3
- PCZOHLXUXFIOCF-UHFFFAOYSA-N Monacolin X Natural products C12C(OC(=O)C(C)CC)CC(C)C=C2C=CC(C)C1CCC1CC(O)CC(=O)O1 PCZOHLXUXFIOCF-UHFFFAOYSA-N 0.000 description 3
- 150000001875 compounds Chemical class 0.000 description 3
- 230000006378 damage Effects 0.000 description 3
- 238000011161 development Methods 0.000 description 3
- 230000018109 developmental process Effects 0.000 description 3
- 239000005090 green fluorescent protein Substances 0.000 description 3
- 229960004844 lovastatin Drugs 0.000 description 3
- PCZOHLXUXFIOCF-BXMDZJJMSA-N lovastatin Chemical compound C([C@H]1[C@@H](C)C=CC2=C[C@H](C)C[C@@H]([C@H]12)OC(=O)[C@@H](C)CC)C[C@@H]1C[C@@H](O)CC(=O)O1 PCZOHLXUXFIOCF-BXMDZJJMSA-N 0.000 description 3
- QLJODMDSTUBWDW-UHFFFAOYSA-N lovastatin hydroxy acid Natural products C1=CC(C)C(CCC(O)CC(O)CC(O)=O)C2C(OC(=O)C(C)CC)CC(C)C=C21 QLJODMDSTUBWDW-UHFFFAOYSA-N 0.000 description 3
- 238000011017 operating method Methods 0.000 description 3
- 238000002428 photodynamic therapy Methods 0.000 description 3
- 108090000623 proteins and genes Proteins 0.000 description 3
- 238000011160 research Methods 0.000 description 3
- 210000003462 vein Anatomy 0.000 description 3
- 241000251468 Actinopterygii Species 0.000 description 2
- 201000001320 Atherosclerosis Diseases 0.000 description 2
- MLDQJTXFUGDVEO-UHFFFAOYSA-N BAY-43-9006 Chemical compound C1=NC(C(=O)NC)=CC(OC=2C=CC(NC(=O)NC=3C=C(C(Cl)=CC=3)C(F)(F)F)=CC=2)=C1 MLDQJTXFUGDVEO-UHFFFAOYSA-N 0.000 description 2
- 206010012689 Diabetic retinopathy Diseases 0.000 description 2
- 108010043121 Green Fluorescent Proteins Proteins 0.000 description 2
- 102000004144 Green Fluorescent Proteins Human genes 0.000 description 2
- 239000002147 L01XE04 - Sunitinib Substances 0.000 description 2
- 239000005511 L01XE05 - Sorafenib Substances 0.000 description 2
- 241000283984 Rodentia Species 0.000 description 2
- 108010081667 aflibercept Proteins 0.000 description 2
- 210000001643 allantois Anatomy 0.000 description 2
- 210000004369 blood Anatomy 0.000 description 2
- 239000008280 blood Substances 0.000 description 2
- 238000009395 breeding Methods 0.000 description 2
- 230000001488 breeding effect Effects 0.000 description 2
- 230000010261 cell growth Effects 0.000 description 2
- 210000003837 chick embryo Anatomy 0.000 description 2
- 230000000875 corresponding effect Effects 0.000 description 2
- 238000009792 diffusion process Methods 0.000 description 2
- 230000000857 drug effect Effects 0.000 description 2
- 239000011888 foil Substances 0.000 description 2
- 238000002372 labelling Methods 0.000 description 2
- 229940092110 macugen Drugs 0.000 description 2
- 210000002189 macula lutea Anatomy 0.000 description 2
- 230000000873 masking effect Effects 0.000 description 2
- 235000015097 nutrients Nutrition 0.000 description 2
- 102000004169 proteins and genes Human genes 0.000 description 2
- 229960003876 ranibizumab Drugs 0.000 description 2
- 238000012827 research and development Methods 0.000 description 2
- 238000001223 reverse osmosis Methods 0.000 description 2
- 235000002639 sodium chloride Nutrition 0.000 description 2
- 238000012109 statistical procedure Methods 0.000 description 2
- 238000002560 therapeutic procedure Methods 0.000 description 2
- 238000012546 transfer Methods 0.000 description 2
- 210000004881 tumor cell Anatomy 0.000 description 2
- 230000004614 tumor growth Effects 0.000 description 2
- 210000003556 vascular endothelial cell Anatomy 0.000 description 2
- AYJGABFBAYKWDX-IAGOWNOFSA-N (-)-Solenopsin A Chemical compound CCCCCCCCCCC[C@@H]1CCC[C@@H](C)N1 AYJGABFBAYKWDX-IAGOWNOFSA-N 0.000 description 1
- UEJJHQNACJXSKW-UHFFFAOYSA-N 2-(2,6-dioxopiperidin-3-yl)-1H-isoindole-1,3(2H)-dione Chemical compound O=C1C2=CC=CC=C2C(=O)N1C1CCC(=O)NC1=O UEJJHQNACJXSKW-UHFFFAOYSA-N 0.000 description 1
- 208000023275 Autoimmune disease Diseases 0.000 description 1
- 208000010839 B-cell chronic lymphocytic leukemia Diseases 0.000 description 1
- 201000004085 CLL/SLL Diseases 0.000 description 1
- 208000005590 Choroidal Neovascularization Diseases 0.000 description 1
- 206010060823 Choroidal neovascularisation Diseases 0.000 description 1
- 208000017604 Hodgkin disease Diseases 0.000 description 1
- 208000031422 Lymphocytic Chronic B-Cell Leukemia Diseases 0.000 description 1
- 206010025323 Lymphomas Diseases 0.000 description 1
- 241001465754 Metazoa Species 0.000 description 1
- JMPFSEBWVLAJKM-UHFFFAOYSA-N N-{5-chloro-4-[(4-chlorophenyl)(cyano)methyl]-2-methylphenyl}-2-hydroxy-3,5-diiodobenzamide Chemical compound ClC=1C=C(NC(=O)C=2C(=C(I)C=C(I)C=2)O)C(C)=CC=1C(C#N)C1=CC=C(Cl)C=C1 JMPFSEBWVLAJKM-UHFFFAOYSA-N 0.000 description 1
- 206010061535 Ovarian neoplasm Diseases 0.000 description 1
- 206010034972 Photosensitivity reaction Diseases 0.000 description 1
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 1
- 239000002671 adjuvant Substances 0.000 description 1
- 230000032683 aging Effects 0.000 description 1
- 238000010171 animal model Methods 0.000 description 1
- 230000003527 anti-angiogenesis Effects 0.000 description 1
- 230000000118 anti-neoplastic effect Effects 0.000 description 1
- 210000000709 aorta Anatomy 0.000 description 1
- 229940120638 avastin Drugs 0.000 description 1
- 210000003050 axon Anatomy 0.000 description 1
- 210000003719 b-lymphocyte Anatomy 0.000 description 1
- 229960000397 bevacizumab Drugs 0.000 description 1
- 229960000074 biopharmaceutical Drugs 0.000 description 1
- 210000001185 bone marrow Anatomy 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 210000002987 choroid plexus Anatomy 0.000 description 1
- 208000032852 chronic lymphocytic leukemia Diseases 0.000 description 1
- 208000023738 chronic lymphocytic leukemia/small lymphocytic lymphoma Diseases 0.000 description 1
- 229950004178 closantel Drugs 0.000 description 1
- 230000002596 correlated effect Effects 0.000 description 1
- 230000008034 disappearance Effects 0.000 description 1
- 239000013583 drug formulation Substances 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 108700008165 endostar Proteins 0.000 description 1
- 210000002889 endothelial cell Anatomy 0.000 description 1
- 230000003511 endothelial effect Effects 0.000 description 1
- 238000013210 evaluation model Methods 0.000 description 1
- 239000003889 eye drop Substances 0.000 description 1
- 229940051306 eylea Drugs 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 230000009036 growth inhibition Effects 0.000 description 1
- 230000012447 hatching Effects 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 206010020718 hyperplasia Diseases 0.000 description 1
- 239000000893 inhibin Substances 0.000 description 1
- 239000003112 inhibitor Substances 0.000 description 1
- ZPNFWUPYTFPOJU-LPYSRVMUSA-N iniprol Chemical compound C([C@H]1C(=O)NCC(=O)NCC(=O)N[C@H]2CSSC[C@H]3C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@H](C(N[C@H](C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC=4C=CC(O)=CC=4)C(=O)N[C@@H](CC=4C=CC=CC=4)C(=O)N[C@@H](CC=4C=CC(O)=CC=4)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)NCC(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CSSC[C@H](NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CC=4C=CC=CC=4)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CCCCN)NC(=O)[C@H](C)NC(=O)[C@H](CCCNC(N)=N)NC2=O)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CSSC[C@H](NC(=O)[C@H](CC=2C=CC=CC=2)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H]2N(CCC2)C(=O)[C@@H](N)CCCNC(N)=N)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(O)=O)C(=O)N2[C@@H](CCC2)C(=O)N2[C@@H](CCC2)C(=O)N[C@@H](CC=2C=CC(O)=CC=2)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(=O)N2[C@@H](CCC2)C(=O)N3)C(=O)NCC(=O)NCC(=O)N[C@@H](C)C(O)=O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@H](C(=O)N[C@@H](CC=2C=CC=CC=2)C(=O)N[C@H](C(=O)N1)C(C)C)[C@@H](C)O)[C@@H](C)CC)=O)[C@@H](C)CC)C1=CC=C(O)C=C1 ZPNFWUPYTFPOJU-LPYSRVMUSA-N 0.000 description 1
- 229940076783 lucentis Drugs 0.000 description 1
- 230000001926 lymphatic effect Effects 0.000 description 1
- 210000004698 lymphocyte Anatomy 0.000 description 1
- 238000007726 management method Methods 0.000 description 1
- 230000005906 menstruation Effects 0.000 description 1
- 230000009401 metastasis Effects 0.000 description 1
- 238000000520 microinjection Methods 0.000 description 1
- 238000013508 migration Methods 0.000 description 1
- 230000005012 migration Effects 0.000 description 1
- 239000000203 mixture Substances 0.000 description 1
- LBWFXVZLPYTWQI-IPOVEDGCSA-N n-[2-(diethylamino)ethyl]-5-[(z)-(5-fluoro-2-oxo-1h-indol-3-ylidene)methyl]-2,4-dimethyl-1h-pyrrole-3-carboxamide;(2s)-2-hydroxybutanedioic acid Chemical compound OC(=O)[C@@H](O)CC(O)=O.CCN(CC)CCNC(=O)C1=C(C)NC(\C=C/2C3=CC(F)=CC=C3NC\2=O)=C1C LBWFXVZLPYTWQI-IPOVEDGCSA-N 0.000 description 1
- 239000002547 new drug Substances 0.000 description 1
- 229940080607 nexavar Drugs 0.000 description 1
- 238000011275 oncology therapy Methods 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 229960003407 pegaptanib Drugs 0.000 description 1
- 238000001050 pharmacotherapy Methods 0.000 description 1
- 208000007578 phototoxic dermatitis Diseases 0.000 description 1
- 230000000270 postfertilization Effects 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 210000001525 retina Anatomy 0.000 description 1
- 239000013037 reversible inhibitor Substances 0.000 description 1
- 201000003068 rheumatic fever Diseases 0.000 description 1
- 206010039073 rheumatoid arthritis Diseases 0.000 description 1
- 229960000672 rosuvastatin Drugs 0.000 description 1
- BPRHUIZQVSMCRT-VEUZHWNKSA-N rosuvastatin Chemical compound CC(C)C1=NC(N(C)S(C)(=O)=O)=NC(C=2C=CC(F)=CC=2)=C1\C=C\[C@@H](O)C[C@@H](O)CC(O)=O BPRHUIZQVSMCRT-VEUZHWNKSA-N 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 231100000075 skin burn Toxicity 0.000 description 1
- 229940126586 small molecule drug Drugs 0.000 description 1
- 150000003384 small molecules Chemical class 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 229960003787 sorafenib Drugs 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- WINHZLLDWRZWRT-ATVHPVEESA-N sunitinib Chemical compound CCN(CC)CCNC(=O)C1=C(C)NC(\C=C/2C3=CC(F)=CC=C3NC\2=O)=C1C WINHZLLDWRZWRT-ATVHPVEESA-N 0.000 description 1
- 229960001796 sunitinib Drugs 0.000 description 1
- 229940034785 sutent Drugs 0.000 description 1
- 230000008685 targeting Effects 0.000 description 1
- 229960003433 thalidomide Drugs 0.000 description 1
- 238000010200 validation analysis Methods 0.000 description 1
- 229950000578 vatalanib Drugs 0.000 description 1
- YCOYDOIWSSHVCK-UHFFFAOYSA-N vatalanib Chemical compound C1=CC(Cl)=CC=C1NC(C1=CC=CC=C11)=NN=C1CC1=CC=NC=C1 YCOYDOIWSSHVCK-UHFFFAOYSA-N 0.000 description 1
- 230000009978 visual deterioration Effects 0.000 description 1
- 230000000007 visual effect Effects 0.000 description 1
- 239000012224 working solution Substances 0.000 description 1
- 210000001325 yolk sac Anatomy 0.000 description 1
Images
Landscapes
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
The invention relates to application of PCI (Percutaneous Coronary Intervention)-32765 for preparing anti-angiogenic medicines. Related reports on PCI-32765 anti-angiogenic activity have not been available so far. The invention provides application of PCI-32765 in preparing anti-angiogenic medicines, antitumor medicines and anti-moisture age-related macular degeneration medicines. In-vivo pharmacodynamic experiment by a zebra fish angiogenic model verifies that PCI-32765 can remarkably inhibit the zebra fish angiogenesis and growth of human cancer cells transplanted and has a treating effect to moisture aged-related macular degeneration. Therefore, PCI-32765 can be used for preparing anti-angiogenic, anti-tumor or anti-moisture age-related macular degeneration medicines.
Description
Technical field
The present invention relates to medical technical field, the specifically application of PCI-32765 in preparing angiogenesis inhibitor class medicine.
Background technology
Angiogenesis (angiogenesis) and the multiple major disease height correlation of the mankind, as malignant tumor, look degeneration of macula (Age-related macular degeneration old age, AMD), atherosclerosis (Atherosclerosis), rheumatic arthritis (Rheumatoid arthritis), diabetic retinopathy (Diabetic retinopathy) and neoplasm metastasis (Tumor metastasis) etc.
[1].Along with the Chinese population aging aggravates gradually, these major diseases life and health of serious harm China people at present.
1971, Judah professor Folkman of Harvard University proposed the antineoplastic vascular therapy first, and he thinks that the growth of entity tumor and diffusion depend on the formation of neovascularity in tumor, and obtains nutrient by new vessels; The formation of neovascularity and growth, promoted the transfer of tumor cell
[2 – 4].Through a large amount of fundamental research in 40 years, at present based on this therapy, the listing of existing a plurality of heavy pound patent new drug, Bevacizumab (trade name Avastin as Roche, within 2010, the global marketing volume is 6,700,000,000 dollars), the Sorafenib of Bayer (trade name Nexavar, the global marketing volume was 9.94 hundred million dollars in 2010), the Sunitinib of Pfizer (trade name Sutent, the global marketing volume was 10.7 hundred million dollars in 2010)
[5 – 6] .but these drug prices are very expensive, be the medicine giant of foreign enterprise monopolization.Domestic only have the anti-tumor angiogenesis drug of 1 real meaning to ratify listing (rhEndostatin, first sign Pharmaceutical, listing in 2005) by SFDA at present
[7], but annual sales amount seldom (2010 annual sales amounts are 2.5 hundred million RMB only
), the method that there is no participates in global competition.Secondly, due to rhEndostatin (recombinant human vascular endothelial inhibin injection, Recombinant Human Endostatin Injection) be macro-molecular protein class medicine, produce the safe preparation of this class high-purity and need very high technical threshold, and the medicine of this type half-life in vivo is shorter, thereby limited the clinical practice of these activated proteins
[7].
Looking degeneration of macula (Age-related macular degeneration is called for short AMD) old age is a kind of degeneration ophthalmic of involving macula retinae district, optical fundus.It produces the macular area degeneration because of age growth, can cause that central vision sharply descends.According to statistics, this disease is suffered from over 3,000 ten thousand people in the whole world.Degeneration of macula is divided into dryness and moist two kinds.Look degeneration of macula moist old age main because choroidal artery generates extremely, seepage appears in newborn invalid blood capillary, and the liquid of vascular leakage and then destruction macula lutea, cause central vision significantly to descend, and affects quality of life, even cecutiency.Look degeneration of macula (AMD) moist old age and become the blind arch-criminal of over-65s old people
[8-9].
The method for the treatment of moist AMD mainly contains photodynamic therapy and anti-angiogenic pharmacotherapy
[8-9].Photodynamic therapy is mainly injected photosensitive drug by vein, then adopts the non-thermal energy laser irradiation choroidal neovascularization focus of specific wavelength, and photosensitive drug is activated.Treating moist AMD by photodynamic therapy, can only stablize or reduce the risk of moist AMD visual deterioration, is not etiological treatment, can not stop the possibility of recurrence.Generally need to repeatedly treat.And want lucifuge 48 hours after treatment, and to avoid photosensitivity reaction, cause skin burn, therefore, to the patient, bring a lot of miseries.Treat at present moist AMD, the medicine gone on the market mainly contains: the Macugen (Pegaptanib of Pfizer, trade name Macugen), the ranibizumab of Novartis (Ranibizumab, trade name Lucentis), the Ai Liya (VEGF-Trap-eye of Bayer, trade name Eylea), the price of these medicines is very expensive, generally needs the administration of every menstruation intravitreal injection, and this tediously long administration process is difficult to be accepted by the patient.Therefore, to treat age-related macular degeneration be following development trend to the eye drop of development of new cheapness.
In sum, seek new angiogenesis inhibitor and effectively treat above-mentioned disease and become the research and development focus in micromolecular compound, it is extremely urgent that exploitation has the patent targeting angiogenesis inhibitor small-molecule drug of independent intellectual property right.
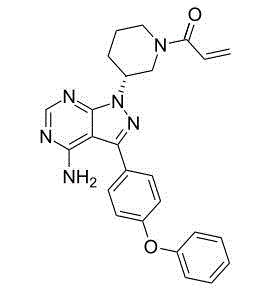
PCI-32765, English common name: PCI-32765 (Ibrutinib); English name: 1-[(3R)-3-[4-Amino-3-(4-phenoxyphenyl)-1H-pyrazolo[3,4-d] pyrimidin-1-yl]-1-piperidinyl]-2-propen-1-one; Chinese: 1-[(3R)-3-[4-amino-3-(4-Phenoxyphenyl)-1H-pyrazolo [3,4-d] pyrimidine-1-yl]-piperidino]-2-propylene-1-ketone; Molecular weight: 440.5; No. Cas: 936563-96-1.PCI-32765 is the non-reversible inhibitor of new small molecule Btk of the Pharmacyclics of U.S. biopharmaceutical company research and development.Chronic lymphocytic leukemia/small lymphocyte lymphoma (CLL/SLL) curative effect is obvious and toleration is good in the PCI-32765 treatment; In addition, PCI-32765 also likely comprises that to autoimmune disease and bone marrow derived cell (B-cell) are relevant the disease such as non-Hodgkin′s lymphatic cancer has certain curative effect
[10-17].The chemical structural formula of PCI-32765 is as follows:
There is not yet the active relevant report of relevant PCI-32765 angiogenesis inhibitor (anti-angiogenesis).
Summary of the invention
The present inventor is unexpected to be found, PCI-32765 has the effect of angiogenesis inhibitor.
Therefore, the invention provides the application of PCI-32765 in preparing angiogenesis inhibitor class medicine.
The present invention also provides the application of PCI-32765 in preparing antitumor drug, and described PCI-32765 prevents or treat tumor by the generation that suppresses neovascularity in tumor.
The present invention also provides PCI-32765 preparing the application of moisture resistance in looking the degeneration of macula medicine old age, and described PCI-32765 prevents or treats and look degeneration of macula moist old age by suppressing the choroidal artery paraplasm.
Said medicine is oral administered dosage form, injecting medicine-feeding form, mucosa delivery dosage form or percutaneous dosing dosage form, more particularly, and as tablet, capsule, granule, oral liquid, injection, patch or gel form.Can pass through the methods known in the art useful in preparing drug formulations.
The present invention utilizes the Brachydanio rerio angiogenesis model to carry out the anti-angiogenic pharmacodynamic experiment of PCI-32765.With traditional blood vessel study model (mouse of Rodents and chick embryo allantois mould), compare, a large amount of studies confirm that at present, Brachydanio rerio is optimal Vascular Biology and anti-angiogenic medicaments evaluation model
[18-27].There are shortcoming separately in the mouse of Rodents and chick embryo allantois mould
[21 – 22].By utilizing the Brachydanio rerio angiogenesis model to carry out pharmacodynamic evaluation and the checking of medicine novel targets, existing branched cancer therapy drug enters clinical front experiment (Pre-clinical Trial) or clinical trial (Clinical Trial) stage (comprising the medicine that obtains FDA approval listing), as Vatalanib (Novartis)
[23], Compound 6 (TargeGen)
[24], Rosuvastatin
[25], Solenopsin (Eli Lilly)
[26]deng; A kind of old medicine for the treatment of malignant tumor of mainly finding based on Brachydanio rerio angiogenesis inhibitor model is newly used medicine---and reaction stops (Thalidomide) and has obtained FDA approval listing
[27].
In the Brachydanio rerio body, vascular pattern confirms, PCI-32765 is to (the intersegm-ental vessel of blood vessel between the Brachydanio rerio body segment, ISV) blood vessel (Subintestinal vessel and under intestinal, SIV) function of remarkable inhibition is arranged, and present certain dosage according to patience, therefore, can be used for preparing the angiogenesis inhibitor inhibitor.
As cancer transplantation model and moist looking aspect the degeneration of macula model, large quantity research is also arranged the research Brachydanio rerio both at home and abroad
[28-33].Through Brachydanio rerio human ovarian carcinoma (A2780) transplantation model, confirm, PCI-32765 can significantly suppress the growth of ovarian cancer (A2780); Look the degeneration of macula model validation through Brachydanio rerio, PCI-32765 has significant therapeutic effect to looking degeneration of macula moist old age.Therefore, PCI-32765 can be used for antitumor and treats and look degeneration of macula moist old age.
PCI-32765 of the present invention is safe, raw material sources are extensive, is aided with pharmaceutically acceptable adjuvant, adopts the conventional formulation technology to can be made into various oral, injections, external preparation, has good DEVELOPMENT PROSPECT.
The accompanying drawing explanation
fig. 1for blood vessel (ISV) model between after fertilization 48h of the present invention (48hpf) blood vessel transgenic fluorescence Brachydanio rerio body segment.Confine the position of zone for the local amplifying observation of Brachydanio rerio body segment blood vessel network.Arrow indication body intersegmental blood vessel (ISV, intersegmental vessel).
fig. 2for the inhibition of qualitative observation PCI-32765 of the present invention to angiogenesis (angiogenesis) between the Brachydanio rerio body segment.Figure a-c, the Brachydanio rerio of after fertilization 23hpf, through drug treating 24h, is 48hpf during observation mutually.The figure negative contrast of a (0.1%DMSO), figure b is the PCI-32765 processed group, the figure positive contrast of c (10 μ M lovastatin).With negative control, compare, 10 μ M PCI-32765 can suppress the generation of blood vessel between the Brachydanio rerio body segment (ISV) fully.
fig. 3for the suppression ratio of PCI-32765 of the present invention to blood vessel (ISV) generation between the Brachydanio rerio body segment.The suppression ratio that PCI-32765 generates blood vessel (ISV) between the Brachydanio rerio body segment presents step increase along with the rising of concentration, the suppression ratio that each concentration PCI-32765 group generates blood vessel (ISV) between the Brachydanio rerio body segment is respectively: 1 μ M(0.1%), 2.5 μ M(19%), 5 μ M(43%), 10 μ M(67%), 25 μ M(100%), IC
50be 5.91 μ M.
fig. 4for blood vessel (SIV) model under 72 hours (72hpf) blood vessel transgenic fluorescence Brachydanio rerio intestinal of after fertilization of the present invention.Confine the position of zone for the local amplifying observation of blood vessel (SIV) network under intestinal.Blood vessel (SIV, subintestinal vessel) under arrow indication intestinal.
fig. 5for the inhibition of quantitative observation PCI-32765 of the present invention to blood vessel (SIV) under the Brachydanio rerio intestinal.Figure a-c, the Brachydanio rerio of after fertilization 48hpf, through drug treating 24h, is 72hpf during observation mutually.The figure negative contrast of a (0.1%DMSO), figure b is the PCI-32765 processed group, the figure positive contrast of c (10 μ M lovastatin).The position that shown in dotted line, zone is blood vessel under intestinal (SIV) area amplifying observation.With negative control, compare, 10 μ M PCI-32765 can significantly suppress the generation of blood vessel under the Brachydanio rerio intestinal (SIV), show as blood vessel area under intestinal and reduce.
fig. 6for the suppression ratio of PCI-32765 of the present invention to blood vessel (SIV) generation under the Brachydanio rerio intestinal.The suppression ratio that PCI-32765 generates blood vessel (SIV) under the Brachydanio rerio intestinal presents step increase along with the rising of concentration, the suppression ratio that each concentration PCI-32765 group generates blood vessel (SIV) under the Brachydanio rerio intestinal is respectively: 1 μ M(11%), 2.5 μ M(25%), 5 μ M(43%), 10 μ M(62%), 25 μ M(98%), IC
50be 7.64 μ M.
fig. 7antitumor drug effect for Brachydanio rerio human ovarian carcinoma of the present invention (A2780) transplantation model evaluation PCI-32765.Figure a-f, transplant the Brachydanio rerio of the rear 2dpf of human ovarian carcinoma (A2780) through drug treating 4d, during observation, is 6dpf mutually.Figure a is blank, the figure negative contrast of b (0.1%DMSO), the PCI-32765 processed group that figure d-f is variable concentrations, the figure positive contrast of c (1000 μ M5-FU).
fig. 8for the growth inhibition ratio of PCI-32765 of the present invention to the carcinoma transplanted cell.PCI-32765 presents step increase to the suppression ratio of mankind's carcinoma transplanted Growth of Cells along with the rising of concentration, three concentration PCI-32765 group suppression ratio are respectively: 1 μ M(7.9%), 2.5 μ M(26.6%), 10 μ M(34.9%).
fig. 9for quantitative assessment PCI-32765 of the present invention to looking the therapeutical effect of degeneration of macula.Figure a-e, the Brachydanio rerio of after fertilization 1dpf, through drug treating 4d, is 5dpf during observation mutually.In border circular areas shown in dotted line, it is choroidal artery.The figure negative contrast of a (0.1%DMSO), figure b is model group (1mg/ml cobaltous chloride), the PCI-32765 processed group that figure c-e is various dose.
figure 10for PCI-32765 of the present invention to the paraplasm suppression ratio of choroidal artery.PCI-32765 presents step increase to the paraplasm suppression ratio of choroidal artery along with the rising of injected dose, three PCI-32765 dosage group suppression ratio are respectively: 0.44 μ g(8.3%), 1.47 μ g(23.8%), 4.41 μ g(36.4%).
The specific embodiment
Below in conjunction with Figure of description and embodiment, the present invention is further elaborated, but protection scope of the present invention is not limited to this.
The Brachydanio rerio initialism of being correlated with
After fertilization hourage: hpf-hours postfertilization
Back of the body major axis blood vessel: DLAV-dorsal longitudinal anastomotic vessel
Blood vessel between body segment: ISV-intersegmental vessel
Dorsal aorta: DA-dorsal aorta
Posterior cardinal vein: PCV-posterior cardinal vessel
Subintestinal vein blood vessel: SIV-subintestinal vessel
Green fluorescent protein: GFP-green fluorescent protein
embodiment 1the inhibition of qualitative observation PCI-32765 to blood vessel (ISV) generation model between the Brachydanio rerio body segment
Brachydanio rerio:
The Brachydanio rerio that the present embodiment is used is blood vessel transgenic green fluorescence Brachydanio rerio (a kind of gene of being expressed by the Brachydanio rerio endothelial-cell specific drives green fluorescent protein at Brachydanio rerio vascular endothelial cell specifically expressing as driven element)
(Fig. 1), raising and Application standard strictly carry out with reference to the requirement of U.S.'s management of laboratory animal and use committee (IACUC).
The water (Fish water) of breeding fish:
Compound method: 1L reverse osmosis water (reverse osmosis (RO) water) adds 0.3g sea salt (Instant Ocean salts).
Dimethyl sulfoxide (DMSO, analytical pure):
Buy in Aladdin.0.1% DMSO solution (negative control) preparation: during use, with the water of breeding fish, be mixed with the working solution that concentration is 0.1%, now with the current.
Lovastatin (positive control):
Buy the U.S. logical sequence in Dalian, purity is greater than 98%.During use, with 0.1% DMSO solution preparation, become the required concentration of experiment, in this experiment, the working concentration of positive control drug is 10 μ M.
PCI-32765(Closantel):
(No. Cas: 936563-96-1), become the PCI-32765 solution of variable concentrations during use with 0.1% DMSO solution preparation, working concentration is 10 μ M to buy in Shanghai vast fragrant bio tech ltd.
experimental technique:
(1) experiment grouping and embryo process: get 45 well-developed zebrafish embryos, during fetal development, be after fertilization 23hpf (hour-postfertilization mutually, hpf), be divided at random 3 groups of (negative control group, the drug treating group, positive controls), every group of embryo's quantity is 15.During operation by embryo's uniform distribution to 48 porocyte culture plate (Greiner, Germany), 15, every hole embryo, every hole embryo raises water 1ml.
(2) drug treating: the medicinal liquid that will prepare in advance rapidly with pipettor (range 100~1000 μ l, Eppendorf) adds Zhong,Mei hole, the hole 1ml that 48 porocyte culture plates are corresponding.Before adding medicinal liquid, with pipettor (range 10~1000 μ l, Eppendorf), the raising water of hatching the embryo in 48 orifice plates is shifted out as possible, this operation needs to complete in advance at short notice, to prevent embryo's drying.The experimental situation temperature is controlled at 28.5 ℃ of left and right, relative humidity 40~70%.Then with masking foil, 48 orifice plates are wrapped, carry out the experiment labelling, be positioned over rapidly in the Brachydanio rerio incubator and continue to cultivate 24h (the incubator temperature is controlled at 28.5 ± 0.5 ℃).
(3) Phenotypic Observation and statistics: observe each hole embryo's phenotype under Stereo microscope, observation index: observe medicine to fetal development, blood circulation, the impact of the aspects such as heartbeat.Then, the affected embryo of blood circulation being placed under body formula fluorescence microscope (Nikon AZ100 body formula fluorescence microscope) and further observing and take pictures, is 48hpf mutually while taking pictures, and to confirm angiogenesis, suppresses phenotype.
Experimental result is shown in
fig. 2: 10 μ M PCI-32765 significantly suppress the generation of Brachydanio rerio intersegmental blood vessel (ISV), show as the intersegmental blood vessel disappearance.
embodiment 2the inhibition of quantitative assessment PCI-32765 to blood vessel (ISV) generation model between the Brachydanio rerio body segment
The Brachydanio rerio vascular endothelial cell sprouts from after fertilization 20hpf, and the 30-31hpf left and right forms blood vessel network between main body segment, and as blood vessel (ISV) between back of the body major axis blood vessel (DLAV) and body segment, 48hpf forms complete axon blood vessel network basically
[34], blood vessel (ISV) between high-visible complete body segment now.Between complete body segment, blood vessel mainly refers to connect that section blood vessel between (DLAV) between dorsal aorta (DA) and back of the body major axis blood vessel, sees
fig. 1(vascular pattern between 48hpf blood vessel transgenic fluorescence Brachydanio rerio body segment).The Brachydanio rerio one of 48hpf has blood vessel (ISVs) between 28 pairs of complete body segments.Experimental technique is as follows:
(1) experiment grouping and embryo process: getting 210 well-developed zebrafish embryos, is after fertilization 23hpf (hour-postfertilization, hpf) mutually during fetal development, is divided at random 7 groups, sees the following form:
30 of every group of zebrafish embryo quantity.During operation by embryo's uniform distribution to 48 porocyte culture plate (Greiner, Germany), 15, every hole embryo, each drug level is processed 30 embryos, every hole embryo raises water 1ml.
(2) drug treating: see the experimental technique operating procedure (2) in embodiment 1.
(3) Phenotypic Observation and quantitative statistics: the embryo after each drug level is processed observes and takes pictures under body formula fluorescence microscope (Nikon AZ100 body formula fluorescence microscope), while taking pictures, be 48hpf mutually, impact blood vessel (ISV) between the Brachydanio rerio body segment generated to analyze each drug level.Get at random 10 embryos from each experimental group and carry out quantitative statistics, statistical indicator is as follows:
1. blood vessel (ISVs) quantity between complete body segment: connect between dorsal aorta (DA) and back of the body major axis blood vessel (DLAV)
Between blood vessel
Utilize GraphPad Prism software to add up mapping, and calculate PCI-32765 and suppress the IC that between the Brachydanio rerio body segment, blood vessel (ISV) generates
50.Experimental result is shown in
fig. 3: the suppression ratio that PCI-32765 generates blood vessel (ISV) between the Brachydanio rerio body segment presents step increase along with the rising of concentration, the suppression ratio that each concentration PCI-32765 group generates blood vessel (ISV) between the Brachydanio rerio body segment is respectively: 1 μ M(0.1%), 2.5 μ M(19%), 5 μ M(43%), 10 μ M(67%), 25 μ M(100%), IC
50be 5.91 μ M.
embodiment 3the inhibition of qualitative observation PCI-32765 to blood vessel (SIV) generation model under the Brachydanio rerio intestinal
Blood vessel under the Brachydanio rerio intestinal (SIV, subintestinal vessel) is grown in the yolk sac both sides, and its shape is like one basket, and blood vessel under intestinal (SIV) is about 50~100 μ m by the body segment veutro to the length of downward-extension
[22-23].See
fig. 4(vascular pattern under 72hpf blood vessel transgenic fluorescence Brachydanio rerio intestinal).Experimental technique is as follows:
(1) experiment grouping and embryo process: get 45 well-developed zebrafish embryos, during fetal development, be after fertilization 48hpf (hour-postfertilization mutually, hpf), be divided at random 3 groups of (negative control group, the medicine group, positive controls), every group of embryo's quantity is 15.During operation by embryo's uniform distribution to 48 porocyte culture plate (Greiner, Germany), 15, every hole embryo, every hole embryo raises water 1ml.
(2) drug treating: see the experimental technique operating procedure (2) in embodiment 1.
(3) Phenotypic Observation and statistics: observe each hole embryo's phenotype under Stereo microscope, observation index: observe medicine to fetal development, blood circulation, the impact of the aspects such as heartbeat.Then, the affected embryo of blood circulation being placed under body formula fluorescence microscope (Nikon AZ100 body formula fluorescence microscope) and further observing and take pictures, is 72hpf mutually while taking pictures, and to confirm angiogenesis, suppresses phenotype.
Experimental result is shown in
fig. 5: 10 μ M PCI-32765 significantly suppress the generation of blood vessel under the Brachydanio rerio intestinal (SIV), show as blood vessel area under intestinal and reduce.
embodiment 4the inhibition of quantitative assessment PCI-32765 to blood vessel (SIV) generation model under the Brachydanio rerio intestinal
(1) experiment grouping and embryo process: getting 210 well-developed zebrafish embryos, is after fertilization 48hpf (hour-postfertilization, hpf) mutually during fetal development, is divided at random 7 groups, sees the following form:
30 of every group of zebrafish embryo quantity.During operation by embryo's uniform distribution to 48 porocyte culture plate (Greiner, Germany), 15, every hole embryo, each drug level is processed 30 embryos, every hole embryo raises water 1ml.
(2) drug treating: see the experimental technique operating procedure (2) in embodiment 1.
(3) Phenotypic Observation and quantitative statistics: the embryo after each drug level is processed observes and takes pictures under body formula fluorescence microscope (Nikon AZ100 body formula fluorescence microscope), while taking pictures, be 72hpf mutually, impact blood vessel (SIV) under the Brachydanio rerio intestinal generated to analyze each drug level.Get at random 10 embryos from each experimental group and carry out quantitative statistics, statistical indicator is as follows:
1. blood vessel area (SIV area) under intestinal: utilize Nikon AZ100 body formula fluorescence microscope configuration
NIS-Elements 3.1 softwares are calculated
Utilize GraphPad Prism software to add up mapping, experimental result is shown in
fig. 6: the suppression ratio that PCI-32765 generates blood vessel (SIV) under the Brachydanio rerio intestinal presents step increase along with the rising of concentration, the suppression ratio that each concentration PCI-32765 group generates blood vessel (SIV) under the Brachydanio rerio intestinal is respectively: 1 μ M(11%), 2.5 μ M(25%), 5 μ M(43%), 10 μ M(62%), 25 μ M(98%), IC
50be 7.64 μ M.
embodiment 5brachydanio rerio human ovarian carcinoma (A2780) transplantation model is estimated the antitumor drug effect of PCI-32765
The growth of entity tumor and diffusion depend on the formation of neovascularity in tumor, and obtain nutrient by new vessels; The formation of neovascularity and growth, promoted the transfer of tumor cell.The present embodiment is for illustrating that PCI-32765 can suppress growth and the migration of tumor.Experimental technique is as follows:
(1) experiment grouping and embryo process: getting the zebrafish embryo that 150 transplanting have human ovarian carcinoma (A2780) cell, is after fertilization 2dpf (day-postfertilization, dpf) mutually during fetal development, is divided at random 5 groups, sees the following form:
30 of every group of zebrafish embryo quantity.During operation by embryo's uniform distribution to 6 porocyte culture plate (Greiner, Germany), 30, every hole embryo, each drug level is processed 30 embryos, every hole embryo raises water 3ml.
(2) drug treating: the medicinal liquid that will prepare in advance rapidly with pipettor adds Zhong,Mei hole, the hole 3ml that 6 porocyte culture plates are corresponding.Then with masking foil, 6 orifice plates are wrapped, carry out the experiment labelling, be positioned in the Brachydanio rerio incubator and continue to cultivate 4d (the incubator temperature is controlled at 35.5 ± 0.5 ℃).
(3) Phenotypic Observation and quantitative statistics: the embryo after each concentration drug treating is observed and takes pictures under body formula fluorescence microscope (Nikon AZ100 body formula fluorescence microscope), while taking pictures, be 6dpf mutually, to analyze the inhibitory action of each drug level to Brachydanio rerio human ovarian carcinoma (A2780) transplantation model.Get at random 10 embryos from each experimental group and carry out quantitative statistics, statistical indicator is as follows:
1. the inhibitory action of qualitative evaluation PCI-32765 to neoplasm metastasis;
2. the inhibitory action of quantitative assessment PCI-32765 to tumor growth: utilize Nikon NIS-Elements 3.1 computed in software tumor cell fluorescence intensities (S), the statistical procedures result means with mean ± SE; PCI-32765 is as follows to the inhibition computing formula of tumor growth:
Utilize GraphPad Prism software to add up mapping, experimental result is shown in
fig. 7~Fig. 8: PCI-32765 presents step increase to the suppression ratio of mankind's carcinoma transplanted Growth of Cells along with the rising of concentration, three concentration PCI-32765 group suppression ratio are respectively: 1 μ M(7.9%), 2.5 μ M(26.6%), 10 μ M(34.9%).
embodiment 6quantitative assessment PCI-32765 looks the therapeutical effect of degeneration of macula to moist old age
Look degeneration of macula moist old age main because choroidal artery generates extremely, seepage appears in newborn invalid blood capillary, the liquid of vascular leakage and then destruction macula lutea.Cobaltous chloride can be induced Brachydanio rerio retina choroid plexus blood vessel hyperplasia, visual cell degeneration, is similar to the change that the mankind look degeneration of macula moist old age
[35-37].The present embodiment is for illustrating that PCI-32765 has therapeutic effect to looking degeneration of macula moist old age.Experimental technique is as follows:
(1) experiment grouping and embryo process: getting 150 well-developed zebrafish embryos, is after fertilization 1dpf (day-postfertilization, dpf) mutually during fetal development, is divided at random 5 groups, sees the following form:
30 of every group of zebrafish embryo quantity.During operation by embryo's uniform distribution to 6 porocyte culture plate (Greiner, Germany), 30, every hole embryo, every hole embryo raises water 3ml.
(2) drug treating: add DMSO in negative control group, making its final concentration is 0.1%; Add cobaltous chloride in model group, making its final concentration is 1 mg/ml; PCI-32765, by the administration of microinjection mode, all injects 30 embryos for every group, after injection, the embryo is put into respectively to the raising water that 3ml contains 1 mg/ml cobaltous chloride by group.
(3) Phenotypic Observation and quantitative statistics: the embryo after each dose drug is processed observes and takes pictures under body formula fluorescence microscope (Nikon AZ100 body formula fluorescence microscope), while taking pictures, be 5dpf mutually, to analyze the inhibitory action of each drug dose to Brachydanio rerio eye choroidal abnormalities hypertrophy blood vessel.Get at random 10 embryos from each experimental group and carry out quantitative statistics, statistical indicator is as follows:
1. the inhibitory action of qualitative evaluation PCI-32765 to eye choroidal abnormalities hypertrophy blood vessel;
2. the inhibitory action of quantitative assessment PCI-32765 to choroidal abnormalities hypertrophy blood vessel: utilize NIS-Elements 3.1 computed in software choroidal abnormalities hypertrophy blood vessel fluorescence intensities (S), the statistical procedures result means with mean ± SE; PCI-32765 is as follows to the inhibition computing formula of choroidal abnormalities hypertrophy blood vessel:
Utilize GraphPad Prism software to add up mapping, experimental result is shown in
fig. 9~Figure 10: PCI-32765 presents step increase to the paraplasm suppression ratio of choroidal artery along with the rising of dosage, three PCI-32765 dosage group suppression ratio are respectively: 0.44 μ g(8.3%), 1.47 μ g(23.8%), 4.41 μ g(36.4%).
list of references
[1] Folkman J. Angiogenesis: an organizing principle for drug discovery
Nat Rev Drug Discov.2007 Apr;6(4):273-86.
[2] Hanahan D, Folkman J. Patterns and emerging mechanisms of the angiogenic switch during tumorigenesis.
Cell.1996 Aug 9;86(3):353-64.
[3] Li CY, Shan S, Cao Y, et al. Role of incipient angiogenesis in cancer metastasis.
Cancer Metastasis Rev. 2000;19(1-2):7-11.
[4] Carmeliet P, Jain RK. Angiogenesis in cancer and other diseases.
Nature.2000
Sep 14;407(6801):249-57.
[5] Ma WW, Adjei AA. Novel agents on the horizon for cancer therapy.
CA
Cancer J Clin.
2009 Mar-Apr;59(2):111-37.
[6] Cook KM, Figg WD. Angiogenesis inhibitors: current strategies and future prospects.
CA Cancer J Clin. 2010 Jul-Aug;60(4):222-43.
[7] Guan great Gang. Research of Recombinant Human Endostatin combined chemotherapy Clinical advances [J]. modern tumor medical science, 2011,19(2): 400-403.
[8] Fang Yuxin, often quiet. the anti-vascular endothelial growth factor of newly wishing of patients with age-related macular degeneration is treated [J]. Chinese medical information Leader, 2007,22(24): 16-17.
[9] Li Hairong, Li Jianhua .Anti-VEGF treats clinical assessment and the application of moist age-related macular degeneration. the Chinese Medicine guide, and 2010,8(25): 32-35.
[10] Lee A. Honigberg etc. " PNAS " 107 volume in 2010 is 13075 pages.
[11] Dasmahapatra G, Patel H, Dent P, et al. The Bruton tyrosine kinase (BTK) inhibitor PCI-32765 synergistically increases proteasome inhibitor activity in diffuse large-B cell lymphoma (DLBCL) and mantle cell lymphoma (MCL) cells sensitive or resistant to bortezomib.
Br J Haematol. 2013 Jan 30.
[12] Brown JR. Ibrutinib (PCI-32765), the First BTK (Bruton's Tyrosine Kinase) Inhibitor in Clinical Trials.
Curr Hematol Malig Rep. 2013 Jan 8.
[13] Advani RH, Buggy JJ, Sharman JP, et al. Bruton tyrosine kinase inhibitor ibrutinib (PCI-32765) has significant activity in patients with relapsed/refractory B-cell malignancies.
J Clin Oncol. 2013 Jan 1;31(1):88-94.
[14] Winer ES, Ingham RR, Castillo JJ. PCI-32765: a novel Bruton's tyrosine kinase inhibitor for the treatment of lymphoid malignancies.
Expert Opin Investig Drugs.2012 Mar;21(3):355-61.
[15] Ponader S, Chen SS, Buggy JJ, et al. The Bruton tyrosine kinase inhibitor PCI-32765 thwarts chronic lymphocytic leukemia cell survival and tissue homing in vitro and in vivo.
Blood. 2012 Feb 2;119(5):1182-9.
[16] de Rooij MF, Kuil A, Geest CR,et al. The clinically active BTK inhibitor PCI-32765 targets B-cell receptor- and chemokine-controlled adhesion and migration in chronic lymphocytic leukemia.
Blood.2012 Mar 15;119(11):2590-4.
[17] Chang BY, Huang MM, Francesco M, et al. The Bruton tyrosine kinase inhibitor PCI-32765 ameliorates autoimmune arthritis by inhibition of multiple effector cells.
Arthritis Res Ther.2011 Jul 13;13(4):R115.
[18] Adeghate E, Adem A, Hasan MY, et al. Medicinal Chemistry and Actions of Dual and Pan PPAR Modulators.
Open Med Chem J. 2011;5(Suppl 2):93-8.
[19]De Filippis B, Giancristofaro A, Ammazzalorso A, et al. Discovery of gemfibrozil analogues that activate PPARα and enhance the expression of gene CPT1A involved in fatty acids catabolism.
Eur J Med Chem. 2011 Oct;46(10): 5218-24.
[20] Ogata M, Tsujita M, Hossain MA, et al. On the mechanism for PPAR agonists to enhance ABCA1 gene expression.
Atherosclerosis. 2009 Aug;205(2):413-9.
[21] Cheng J, Gu YJ, Wang Y, et al. Nanotherapeutics in angiogenesis: synthesis and in vivo assessment of drug efficacy and biocompatibility in zebrafish embryos.
Int J Nanomedicine. 2011;6:2007-21.
[22] Serbedzija GN, Flynn E, Willett CE. Zebrafish angiogenesis: a new model for drug screening.
Angiogenesis. 1999;3(4):353-9.
[23] Hasan J, Shnyder SD, Bibby M, et al. Quantitative angiogenesis assays in vivo--a review.
Angiogenesis.2004; 7: 1-16.
[24] Nicoli S, Presta M. The zebrafish/tumor xenograft angiogenesis assay.
Nat Protoc. 2007;2(11):2918-23.
[25] Chan J, Bayliss PE, Wood JM,et al. Dissection of angiogenic signaling in zebrafish using a chemical genetic approach.
Cancer Cell.2002 Apr;1(3): 257-67.
[26] Murphy EA, Shields DJ, Stoletov K, et al. Disruption of angiogenesis and tumor growth with an orally active drug that stabilizes the inactive state of PDGFRbeta/B-RAF.
Proc Natl Acad Sci U S A. 2010 Mar 2;107(9):4299-304.
[27] Wang C, Tao W, Wang Y, et al. Rosuvastatin, identified from a zebrafish chemical genetic screen for antiangiogenic compounds, suppresses the growth of prostate cancer.
Eur Urol. 2010 Sep;58(3):418-26.
[28] Arbiser JL, Kau T, Konar M, et al. Solenopsin, the alkaloidal component of the fire ant (Solenopsis invicta), is a naturally occurring inhibitor of phosphatidylinositol-3-kinase signaling and angiogenesis.
Blood.2007 Jan 15;109(2):560-5.
[29] Yabu T, Tomimoto H, Taguchi Y, et al. Thalidomide-induced antiangiogenic action is mediated by ceramide through depletion of VEGF receptors, and is antagonized by sphingosine-1-phosphate.
Blood. 2005 Jul 1;106(1):125-34.
[30] Herpers R, van de Kamp E, Duckers HJ, et al. Redundant Roles for Sox7 and Sox18 in Arteriovenous Specification in Zebrafish.
Circ Res. 2008 Jan 4;102(1):12-5.
[31] Marques IJ, Weiss FU, Vlecken DH, et al. Metastatic behaviour of primary human tumours in a zebrafish xenotransplantation model.
BMC Cancer.2009
Apr 28;9(1):128.
[32] Konantz M, Balci TB, Hartwig UF, et al. Zebrafish xenografts as a tool for in vivo studies on human cancer[J].
Ann. N.Y. Acad. Sci. 2012, (1266): 124-137.
[33] Chu DLH, Li VWT, Yu RMK. Leptin: Clue to poor appetite in oxygen-starved fish[J].
Molecular and Cellular Endocrinology, 2010, (319): 143-146.
[34] Rooijen E, Voest EE, Logister I, et al. Von Hippel-Lindau tumor suppressor mutants faithfully model pathological hypoxia-driven angiogenesis and vascular retinopathied in zebrafish[J].
Disease Models & Mechanisms, 2010, (3): 343-353.
[35] Bibliowicz J, Tittle RK, Gross JM. Towards a better understanding of human eye disease: insights from the zebrafish, Danio rerio[J].
Prog Mol Biol Transl Sci, 2011, (100): 287-330.
[36] Siekmann AF, Lawson ND. Notch signalling limits angiogenic cell behaviour in developing zebrafish arteries[J].
Nature.2007 Feb 15;445(7129):781-4.
[37] Yu RM, Chu DL, Tan TF, et al. Leptin-mediated modulation of steroidogenic gene expression in hypoxic zebrafish embryos: implications for the disruption of sex steroids[J].
Environ Sci Technol, 2012, 46(16): 9112-9119.
Claims (9)
1.PCI-32765 the application in preparing angiogenesis inhibitor class medicine.
2. application according to claim 1, is characterized in that, described medicine is oral administered dosage form, injecting medicine-feeding form, mucosa delivery dosage form or percutaneous dosing dosage form.
3. application according to claim 1, is characterized in that, described medicine is tablet, capsule, granule, oral liquid, injection, patch or gel form.
4.PCI-32765 the application in preparing antitumor drug.
5. application according to claim 4, is characterized in that, described PCI-32765 prevents or treat tumor by the generation that suppresses neovascularity in tumor.
6. application according to claim 4, is characterized in that, described medicine is tablet, capsule, granule, oral liquid, injection, patch or gel form.
7.PCI-32765 preparing the application of moisture resistance in looking the degeneration of macula medicine old age.
8. application according to claim 7, is characterized in that, described PCI-32765 prevents or treats and look degeneration of macula moist old age by suppressing the choroidal artery paraplasm.
9. application according to claim 7, is characterized in that, described medicine is tablet, capsule, granule, oral liquid, injection, patch or gel form.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2013100792612A CN103142601A (en) | 2013-03-13 | 2013-03-13 | Application of PCI (Percutaneous Coronary Intervention)-32765 for preparing anti-angiogenic medicines |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2013100792612A CN103142601A (en) | 2013-03-13 | 2013-03-13 | Application of PCI (Percutaneous Coronary Intervention)-32765 for preparing anti-angiogenic medicines |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN103142601A true CN103142601A (en) | 2013-06-12 |
Family
ID=48541111
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN2013100792612A Pending CN103142601A (en) | 2013-03-13 | 2013-03-13 | Application of PCI (Percutaneous Coronary Intervention)-32765 for preparing anti-angiogenic medicines |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN103142601A (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN104327085A (en) * | 2013-11-27 | 2015-02-04 | 苏州晶云药物科技有限公司 | PCI-32765 crystal form A and preparation method thereof |
| CN106866684A (en) * | 2015-12-10 | 2017-06-20 | 杭州雷索药业有限公司 | Macrocyclic derivatives for treating tumour |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101610676A (en) * | 2006-09-22 | 2009-12-23 | 药品循环公司 | Inhibitors of Bruton's tyrosine kinase |
| US20120071497A1 (en) * | 2010-06-03 | 2012-03-22 | Pharmacyclics, Inc. | Methods of treating abc-dlbcl using inhibitors of bruton's tyrosine kinase |
-
2013
- 2013-03-13 CN CN2013100792612A patent/CN103142601A/en active Pending
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101610676A (en) * | 2006-09-22 | 2009-12-23 | 药品循环公司 | Inhibitors of Bruton's tyrosine kinase |
| US20120071497A1 (en) * | 2010-06-03 | 2012-03-22 | Pharmacyclics, Inc. | Methods of treating abc-dlbcl using inhibitors of bruton's tyrosine kinase |
Non-Patent Citations (2)
| Title |
|---|
| SAMIR PAREKH,ET AL.: "New molecular targets in mantle cell lymphoma", 《SEMINARS IN CANCER BIOLOGY》 * |
| 樊代明: "《肿瘤研究前沿(第10卷)》", 31 December 2010 * |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN104327085A (en) * | 2013-11-27 | 2015-02-04 | 苏州晶云药物科技有限公司 | PCI-32765 crystal form A and preparation method thereof |
| CN104327085B (en) * | 2013-11-27 | 2016-08-24 | 苏州晶云药物科技有限公司 | Crystal formation A of PCI-32765 and preparation method thereof |
| CN106866684A (en) * | 2015-12-10 | 2017-06-20 | 杭州雷索药业有限公司 | Macrocyclic derivatives for treating tumour |
| CN106866684B (en) * | 2015-12-10 | 2020-06-09 | 吴耀东 | Macrocyclic derivatives for the treatment of tumors |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Lis et al. | Circadian timing in cancer treatment: the biological foundation for an integrative approach | |
| MXPA03010528A (en) | Methods for inhibiting angiogenesis. | |
| Kaczmarek et al. | Intravitreal HDAC inhibitor belinostat effectively eradicates vitreous seeds without retinal toxicity in vivo in a rabbit retinoblastoma model | |
| CN101647801A (en) | Application of tetracyclic triterpenoids compound in preparing anti-angiogenic drugs | |
| US10709757B2 (en) | Pharmaceutical composition for anti-angiogenesis containing cyclic pentadepsipeptide as an effective ingredient | |
| Yan et al. | Effects of vestibular damage on the sleep and expression level of orexin in the hypothalamus of rats and its correlation with autophagy and Akt tumor signal pathway | |
| CN103479662A (en) | Application of auranofin in preparing anti-angiogenesis medicines | |
| CN103142601A (en) | Application of PCI (Percutaneous Coronary Intervention)-32765 for preparing anti-angiogenic medicines | |
| CN102217980A (en) | Preparation method of rhesus monkey choroidal angiogenesis model | |
| CN105418769B (en) | Fusion protein with functions of resisting tumor and inflammation and treating ophthalmic diseases and preparation method and application thereof | |
| CN111562394A (en) | Application of heat shock factor 2 binding protein in liver ischemia-reperfusion injury and drug-induced liver injury | |
| CN103054866A (en) | Application of chloroxine in preparing anti-angiogenesis medicine | |
| CN103040823A (en) | Application of vinpocetine in preparation of anti-angiogenic medicament | |
| CN103054858A (en) | Application of oxibendazole in preparing anti-angiogenesis medicine | |
| CN103251588A (en) | Application of phenytoin sodium in preparing anti-angiogenesis medicine | |
| CN103054846B (en) | Anti-angiogenic compound and usage thereof | |
| CN103040800A (en) | Application of gemfibrozil in preparation of anti-angiogenic medicaments | |
| CN1837169A (en) | Compound capable of inhibiting zinc ion metalloproteinases | |
| CN103271897A (en) | Applications of mesna in preparation of anti-angiogenesis drugs | |
| CN103251633A (en) | Application of phenazopyridine in preparing anti-angiogenesis medicine | |
| CN116751816A (en) | A plasmid composition, construction method and application of rat hydrodynamic liver cancer model | |
| CN103110614B (en) | Application of suloctidil to prepare anti-angiogenesis medicine | |
| CN104622874B (en) | Application of the CCR4 antagonists in cancer growth and transfer is suppressed | |
| CN113577066B (en) | Use of arylguanidine compound or pharmaceutically acceptable salt thereof | |
| CN103054840B (en) | Application of levalbuterol to preparation of anti-angiogenesis drugs |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| RJ01 | Rejection of invention patent application after publication | ||
| RJ01 | Rejection of invention patent application after publication |
Application publication date: 20130612 |