CN103130663A - Cinnamylate ester compound and application thereof - Google Patents
Cinnamylate ester compound and application thereof Download PDFInfo
- Publication number
- CN103130663A CN103130663A CN2013100437540A CN201310043754A CN103130663A CN 103130663 A CN103130663 A CN 103130663A CN 2013100437540 A CN2013100437540 A CN 2013100437540A CN 201310043754 A CN201310043754 A CN 201310043754A CN 103130663 A CN103130663 A CN 103130663A
- Authority
- CN
- China
- Prior art keywords
- compound
- cinnamylate
- ester compound
- formula
- plant growth
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Classifications
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02A—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE
- Y02A40/00—Adaptation technologies in agriculture, forestry, livestock or agroalimentary production
- Y02A40/10—Adaptation technologies in agriculture, forestry, livestock or agroalimentary production in agriculture
Landscapes
- Agricultural Chemicals And Associated Chemicals (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
Abstract
Description
技术领域 本发明属于农用植物生长调节剂领域,具体涉及一种肉桂酸酯类化合物及其应用。Technical field The invention belongs to the field of agricultural plant growth regulators, in particular to a cinnamate compound and its application.
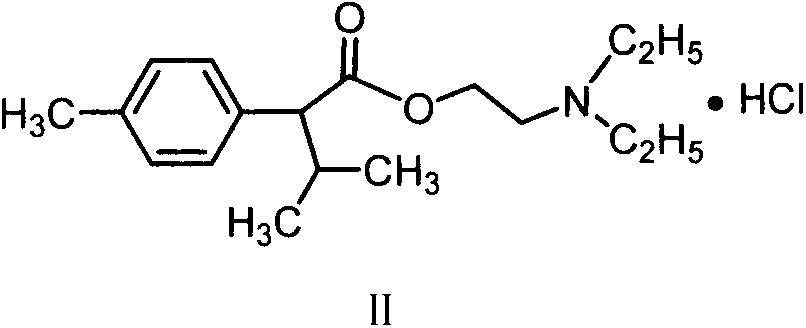
背景技术 随着人口增长及可耕地面积的减少,在有限的土地上生产更多的粮食是解决粮食安全问题的重要手段。植物生长调节剂作为植物化控手段在农作物增产、早熟、改善品质及抗逆等方面取得了广泛应用。目前国内普遍供用的植物生长剂品种主要有复硝酚钠、胺鲜脂、萘乙酸钠、乙烯利等,存在着功能单一、使用安全性及药效不够好等欠缺,因此开发综合性能好、使用安全的绿色植物生长调节剂品种是我国农业发展之必需。CN1091123报道了下列化合物(式II)的制备及植物生长调节活性,用于小麦和油菜等作物增产17%以上。Background Art With the increase of population and the reduction of arable land, producing more food on limited land is an important means to solve the problem of food security. Plant growth regulators have been widely used as plant chemical control methods in crop yield increase, early maturity, quality improvement and stress resistance. At present, the types of plant growth agents commonly used in China mainly include sodium nitrophenolate, amine fresh fat, sodium naphthalene acetate, and ethephon, etc., which have shortcomings such as single function, use safety, and insufficient efficacy. Therefore, the development of comprehensive performance is good, The use of safe green plant growth regulator varieties is necessary for the development of agriculture in our country. CN1091123 reports the preparation of the following compound (formula II) and its plant growth regulating activity, which is used to increase the yield of crops such as wheat and rapeseed by more than 17%.
在现有技术中,如本发明所述的肉桂酸酯类化合物及其植物生长调节活性未见公开In the prior art, there is no disclosure of cinnamate compounds and their plant growth regulating activity as described in the present invention
发明内容 本发明的目的是提供一种促生根、增产作用好、成本低及使用安全的植物生长调节剂,具体地说提供一种肉桂酸酯类化合物,它可用于农业、林业的大幅度增产或成活率提高。SUMMARY OF THE INVENTION The object of the present invention is to provide a plant growth regulator that promotes rooting, has a good yield-increasing effect, is low in cost, and is safe to use, and specifically provides a cinnamic acid ester compound, which can be used to greatly increase yield in agriculture and forestry or increase the survival rate.
本发明的技术方案如下:Technical scheme of the present invention is as follows:
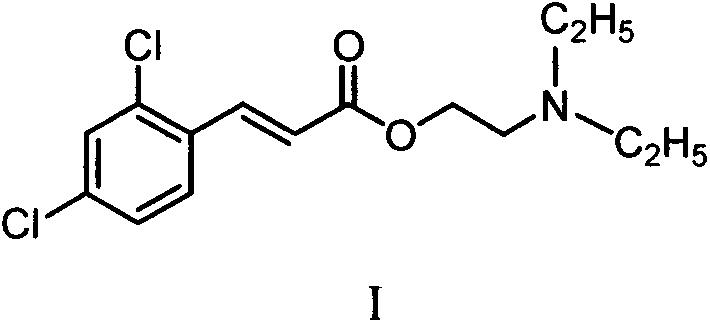
一种肉桂酸酯类化合物,结构如式I所示:A cinnamate compound, the structure is as shown in formula I:
式I化合物及其盐的制备反应式如下:The preparation reaction formula of formula I compound and salt thereof is as follows:
其中RCOOH:柠檬酸,草酸,富马酸,琥珀酸二氯肉桂酸在适宜的溶剂中与氯化亚砜在10℃至回流温度下反应2-4h,减压蒸出溶剂和过量的氯化亚砜,制得2,4-二氯肉桂酰氯;将制得的酰氯加适当的溶剂配成溶液,控温-10℃-10℃搅拌下滴加到由二乙氨基乙醇与适当的溶剂配成的溶液中,制得式I化合物的盐酸盐;将该化合物盐酸盐溶于水中,用饱和碳酸钠溶液中和,制得式I化合物;式I化合物与有机酸按等摩尔反应制得式I化合物的有机酸盐。反应过程适宜的溶剂可选自二氯甲烷、苯、甲苯、乙酸乙酯。二乙氨基乙醇既是反应原料又是缚酸剂,故式I化合物盐酸盐的生成无需另加缚酸剂。制备其有机酸盐过程中,有机酸选自柠檬酸、草酸、富马酸、琥珀酸,优选柠檬酸制备式I化合物的柠檬酸盐。由于式I化合物为水不溶性流动液体,接触空气易氧化变色,故作为植物生长调节剂使用时通常以无机盐或有机酸盐的形式出现。同市面上已知的同类化合物相比,本发明的肉桂酸酯类化合物具有更高的植物生长调节活性,其促生根性能和增产作用不仅显著优于同类产品DA-6(胺鲜脂),也显著优于广泛应用的“复硝酚钠”、“萘乙酸钠”等植物生长调节剂。本发明的肉桂酸酯类化合物及其盐具有合成方法简便、环保、合成成本低、有效用量少等优点。因此可以实现以较低的使用成本和突出的植物生长调节活性达到农作物大幅度增产或林业提高成活率的目的。Among them, RCOOH: citric acid, oxalic acid, fumaric acid, succinic acid dichlorocinnamic acid react with thionyl chloride in a suitable solvent at 10°C to reflux temperature for 2-4h, and distill off the solvent and excess chlorinated sulfoxide to obtain 2,4-dichlorocinnamoyl chloride; add the obtained acid chloride to a suitable solvent to form a solution, and add it dropwise to the solution prepared by diethylaminoethanol and a suitable solvent under stirring at -10°C-10°C In the resulting solution, the hydrochloride of the compound of formula I is obtained; the compound hydrochloride is dissolved in water, and neutralized with a saturated sodium carbonate solution to obtain the compound of formula I; the compound of formula I is reacted with an organic acid in equimolar The organic acid salt of the compound of formula I is obtained. Suitable solvents for the reaction process can be selected from dichloromethane, benzene, toluene, ethyl acetate. Diethylaminoethanol is both a reaction raw material and an acid-binding agent, so the generation of the compound hydrochloride of formula I does not need additional acid-binding agent. In the process of preparing its organic acid salt, the organic acid is selected from citric acid, oxalic acid, fumaric acid, succinic acid, preferably citric acid to prepare the citrate of the compound of formula I. Since the compound of formula I is a water-insoluble flowing liquid, it is easy to oxidize and change color when exposed to air, so it usually appears in the form of inorganic salt or organic acid salt when used as a plant growth regulator. Compared with similar compounds known on the market, the cinnamic acid ester compound of the present invention has higher plant growth regulating activity, and its rooting-promoting performance and production-increasing effect are not only significantly better than similar products DA-6 (amine fresh fat), It is also significantly better than widely used plant growth regulators such as "sodium nitrophenolate" and "sodium naphthalene acetate". The cinnamate ester compound and the salt thereof of the present invention have the advantages of simple synthesis method, environmental protection, low synthesis cost, less effective dosage and the like. Therefore, the purpose of greatly increasing the yield of crops or increasing the survival rate of forestry can be achieved with lower use cost and outstanding plant growth regulating activity.
本发明还包括式I化合物或其盐作为活性组分的植物生长调节剂组合物,该组合物中活性组分的重量百分含量在0.5~95%之间,该植物生长调节剂组合物中还包括农业或林业上可接受的载体。The present invention also includes the compound of formula I or its salt as the plant growth regulator composition of the active component, and the weight percentage of the active component in the composition is between 0.5% and 95%, and in the plant growth regulator composition Also included are agriculturally or forestry acceptable carriers.
本发明的组合物可以制剂的形式施用,式I化合物或其盐作为活性组分溶解或分散于载体中配制成水剂、微乳剂、水乳剂、乳油或可溶性粉剂,为了提高有效组分利用率,在配制制剂时通常加入适量的表面活性剂。The composition of the present invention can be administered in the form of preparations, and the compound of formula I or its salt is dissolved or dispersed in the carrier as an active component to be formulated into an aqueous agent, a microemulsion, an emulsion in water, an emulsifiable concentrate or a soluble powder, in order to improve the utilization rate of the effective component , usually add an appropriate amount of surfactant when preparing the preparation.
本发明的技术方案还包括调节植物生长的方法:将本发明组合物按活性组分0.1~500ppm浓度施于需要调节生长的介质上,优选的使用浓度为20~200ppm。The technical scheme of the present invention also includes a method for regulating plant growth: applying the composition of the present invention to the medium requiring growth regulation at a concentration of 0.1-500 ppm of the active component, and preferably using a concentration of 20-200 ppm.
对于某些应用,可根据需要在本发明的植物生长调节剂组合物中加一种或几种具他的杀菌剂、杀虫剂、植物生长调节剂或肥料等,由此可产生附加的优点和效果。For some applications, one or more other fungicides, insecticides, plant growth regulators or fertilizers, etc. can be added to the plant growth regulator composition of the present invention as required, which can produce additional advantages and effects.
应明确的是,在本发明的权力要求所限定的范围内,可进行各种交换和变动。It should be understood that various exchanges and changes can be made within the scope defined by the claims of the present invention.
具体实施方式Detailed ways
下列合成实例,制剂实施例及生测实验结果可用来进一步说明本发明,但不意味着限制本发明。The following synthesis examples, preparation examples and bioassay results can be used to further illustrate the present invention, but are not meant to limit the present invention.
合成实例synthetic example
实例1、式I化合物的制备Example 1, the preparation of formula I compound
(1)2,4-二氯肉桂酰氯的合成(1) Synthesis of 2,4-dichlorocinnamoyl chloride
向500mL反应瓶中依次加入43.2g(0.2mol)2,4-二氯肉桂酸、200mL甲苯和35.4g氯化亚砜(0.3mol),搅拌下缓慢升温(放出HCl和SO2气体,使用20%NaOH溶液吸收),分别在40~60℃、60~80℃、80~100℃反应1h,然后回流1h;降至室温,旋蒸出甲苯和过量氯化亚砜得浅黄色油状液体45.0g,收率95.5%。Add 43.2g (0.2mol) 2,4-dichlorocinnamic acid, 200mL toluene and 35.4g thionyl chloride (0.3mol) successively in the 500mL reaction flask, slowly heat up under stirring (release HCl and SO 2 gas, use 20 % NaOH solution absorption), react at 40-60°C, 60-80°C, and 80-100°C for 1 hour, then reflux for 1 hour; cool down to room temperature, rotary distill off toluene and excess thionyl chloride to obtain 45.0 g of light yellow oily liquid , yield 95.5%.
(2)2,4-二氯肉桂酸二乙氨基乙醇酯盐酸盐的合成(2) Synthesis of 2,4-dichlorocinnamic acid diethylaminoethanol ester hydrochloride
向500mL反应瓶中依次加入23.4g(0.2mol)二乙氨基乙醇,150mL甲苯,冰盐浴降温至-5~0℃,搅拌下滴加上述制得的47.1g(0.2mol)2,4-二氯肉桂酰氯与50mL甲苯组成的溶液,控温0~5℃,滴加完毕,室温搅拌反应2h;抽滤,滤饼用适量甲苯洗涤,滤液重蒸后套用,干燥得白色固体67.0g,收率96.0%。熔点207-212℃。Add 23.4g (0.2mol) diethylaminoethanol and 150mL toluene to a 500mL reaction flask in sequence, cool down to -5-0°C in an ice-salt bath, and add dropwise 47.1g (0.2mol) of 2,4- A solution composed of dichlorocinnamoyl chloride and 50mL toluene was controlled at a temperature of 0-5°C. After the dropwise addition was completed, the reaction was stirred at room temperature for 2 hours; filtered with suction, the filter cake was washed with an appropriate amount of toluene, the filtrate was re-distilled and used mechanically, and dried to obtain 67.0 g of a white solid. Yield 96.0%. The melting point is 207-212°C.
(3)2,4-二氯肉桂酸二乙氨基乙醇酯(式I化合物)的制备(3) 2, the preparation of 4-dichlorocinnamic acid diethylaminoethanol ester (formula I compound)
向500mL反应瓶中加入上述制得的式I化合物盐酸盐53.0g(0.15mol)、150mL水,搅拌溶解.加入100mL二氯甲烷,室温搅拌下滴加由16g(0.15mol)碳酸钠和80mL水配成的溶液,滴毕,继续搅拌0.5h,静置分层,分出下层有机相,用水洗涤两次(30mL×2),有机相用无水硫酸钠干燥,旋蒸出溶剂二氯甲烷,得到浅黄色油状液43.5g,收率92%,式I化合物的1H NMR(500MHz,DMSO-d6)δ(ppm):0.978~1.007(t,-CH3,6H),2.528~2.571(q,-CH2,4H),2.708~2.732(t,-CH2,2H),4.210~4.234(t,-CH2,2H),6.339~6.371(d,J=16,-CH,1H),7.163~7.473(m,Ar-H,3H),7.908~7.940(d,J=16,-CH,1H)。Add 53.0g (0.15mol) of the compound hydrochloride of formula I prepared above, 150mL water in the 500mL reaction flask, stir and dissolve. Add 100mL dichloromethane, add dropwise under stirring at room temperature 16g (0.15mol) sodium carbonate and 80mL The solution made of water, after dripping, continued to stir for 0.5h, allowed to stand for stratification, separated the lower organic phase, washed twice with water (30mL×2), dried the organic phase with anhydrous sodium sulfate, and rotary evaporated the solvent dichloromethane Methane, to obtain 43.5g of light yellow oily liquid, yield 92%, 1 H NMR (500MHz, DMSO-d6) δ (ppm) of the compound of formula I: 0.978~1.007 (t, -CH 3 , 6H), 2.528~2.571 (q, -CH 2 , 4H), 2.708~2.732(t, -CH 2 , 2H), 4.210~4.234(t, -CH 2 , 2H), 6.339~6.371(d, J=16, -CH, 1H ), 7.163~7.473 (m, Ar-H, 3H), 7.908~7.940 (d, J=16, -CH, 1H).
实例2、2,4-二氯肉桂酸二乙氨基乙醇酯有机酸盐的制备Example 2, 2, the preparation of 4-dichlorocinnamic acid diethylaminoethanol ester organic acid salt
(1)2,4-二氯肉桂酸二乙氨基乙醇酯柠檬酸盐的制备(1) Preparation of 2,4-dichlorocinnamic acid diethylaminoethanol citrate
向250mL反应瓶中依次加入0.05mol柠檬酸和80mL乙醇,搅拌溶解,搅拌下缓慢滴加0.05mol2,4-二氯肉桂酸二乙氨基乙醇酯(式I化合物)与20mL乙醇的混合液,滴毕,继续搅拌0.5h,旋蒸出溶剂,干燥得白色固体25.4g,收率约100%。Add 0.05mol citric acid and 80mL ethanol successively to the 250mL reaction flask, stir to dissolve, slowly add dropwise the mixed solution of 0.05mol2,4-dichlorocinnamic acid diethylaminoethanol ester (formula I compound) and 20mL ethanol under stirring, drop After completion, the stirring was continued for 0.5 h, the solvent was evaporated by rotary evaporation, and 25.4 g of white solid was obtained by drying, with a yield of about 100%.
(2)2,4-二氯肉桂酸二乙氨基乙醇酯草酸盐的制备(2) Preparation of 2,4-dichlorocinnamic acid diethylaminoethanol oxalate
向250mL反应瓶中依次加入0.05mol的草酸、100mL乙酸乙酯和0.05mol的2,4-二氯肉桂酸二乙氨基乙醇酯(式I化合物),升温回流1h,降温,抽滤,干燥得白色固体19.9,收率98%。Add 0.05 mol of oxalic acid, 100 mL of ethyl acetate and 0.05 mol of 2,4-dichlorocinnamic acid diethylaminoethanol (compound of formula I) to a 250 mL reaction flask successively, heat up and reflux for 1 h, cool down, suction filter, and dry to obtain White solid 19.9, yield 98%.
按照方法(1)或(2)可以分别制得式I化合物的琥珀酸盐、富马酸盐。According to method (1) or (2), the succinate and fumarate of the compound of formula I can be prepared respectively.
组合物制剂实施例Composition Formulation Example
实例3、(1)5%2,4-二氯肉桂酸二乙氨基乙醇酯盐酸盐水剂Example 3, (1) 5% 2,4-dichlorocinnamic acid diethylaminoethanol ester hydrochloride aqueous solution
向250mL反应瓶中分别加入5g式I化合物盐酸盐和90g水,搅拌溶解,加入5g吐温-80,搅拌0.5h.得5%2,4-二氯肉桂酸二乙氨基乙醇酯盐酸盐水剂。Add 5g of formula I compound hydrochloride and 90g of water to a 250mL reaction flask, stir to dissolve, add 5g of Tween-80, and stir for 0.5h to obtain 5% diethylaminoethanol 2,4-dichlorocinnamate hydrochloride agent.
(2)50%2,4-二氯肉桂酸二乙氨基乙醇酯柠檬酸盐可湿性粉剂(2) 50% 2,4-dichlorocinnamic diethylaminoethanol citrate wettable powder
称取50g2,4-二氯肉桂酸二乙氨基乙醇酯柠檬酸盐,45g氯化钾和5g茶皂素,混合均匀后粉碎至80~100目,得50%2,4-二氯肉桂酸二乙氨基乙醇酯柠檬酸盐可湿性粉剂。Weigh 50g of 2,4-dichlorocinnamic acid diethylaminoethanol citrate, 45g of potassium chloride and 5g of tea saponin, mix them evenly and grind them to 80-100 meshes to obtain 50% 2,4-dichlorocinnamic acid Diethylaminoethanol Citrate Wettable Powder.
生物活性测试Biological activity test
实例4、黄瓜子叶生根测定Example 4, measurement of cucumber cotyledon rooting
选用黄瓜种子津研4号,浸种2h后播于0.7%的琼脂上,25℃暗室培养72h,摘取子叶备用,取10片子叶放在直径6cm的培养皿中,内有一张滤纸片和30mL待测试液,25℃暗室培养5天,测叶柄茎部生根,第6天测根长和根鲜重。Select cucumber seeds Jinyan No. 4, sow the seeds on 0.7% agar after soaking for 2 hours, culture in a dark room at 25°C for 72 hours, remove cotyledons for later use, take 10 cotyledons and put them in a 6cm-diameter petri dish, which contains a piece of filter paper and 30mL The solution to be tested was cultured in a dark room at 25°C for 5 days, and the rooting of petioles and stems was measured, and the root length and root fresh weight were measured on the 6th day.
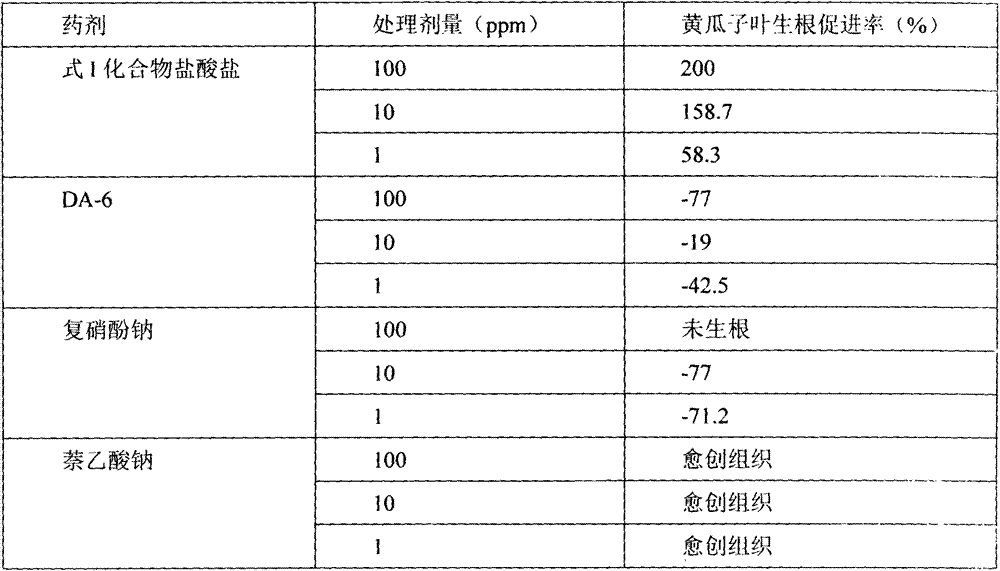
对本发明合成的化合物(式I)盐酸盐进行测试,以市面购得的复硝酚钠、DA-6、萘乙酸钠为对比药剂,实验结果见表1The compound (formula I) hydrochloride synthesized by the present invention is tested, and commercially available sodium binitrophenolate, DA-6, and sodium naphthalene acetate are used as contrast agents, and the experimental results are shown in Table 1
表1不同植物生长调节剂生物活性测定结果Table 1 The results of bioactivity assays of different plant growth regulators
从表1测定结果看出,本发明化合物(式I)对黄瓜子叶生根具有极显著的促进作用,并随浓度的升高促进作用增强。说明本发明化合物不仅促生根作用强,且使用安全,而对比生长调节剂则出现抑制作用或药害。As can be seen from the measurement results in Table 1, the compound of the present invention (formula I) has a very significant promoting effect on the rooting of cucumber cotyledons, and the promoting effect is enhanced with the increase of the concentration. It shows that the compound of the present invention not only has a strong rooting-promoting effect, but also is safe to use, while the inhibitory effect or phytotoxicity occurs in the comparison growth regulator.
将组合物制剂实施例3中(1)制备的5%2,4-二氯肉桂酸二乙氨基乙醇酯盐酸盐水剂稀释500倍(100ppm)于作物生长期喷洒2次,两次喷洒间隔期15天,对花生、生姜和玉米增产率分别达21%、16.7%和22.4%,显著优于同浓度下DA-611.5%、8.3%和14.0%的增产效率。Dilute 500 times (100ppm) of 5% 2,4-dichlorocinnamic acid diethylaminoethanol ester hydrochloride aqueous solution prepared in composition preparation embodiment 3 (1) and spray 2 times in the crop growth period, and the interval between two sprays After 15 days, the yield-increasing rates of peanut, ginger and corn reached 21%, 16.7% and 22.4% respectively, significantly better than the 11.5%, 8.3% and 14.0% yield-increasing efficiencies of DA-6 at the same concentration.
Claims (3)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201310043754.0A CN103130663B (en) | 2013-01-25 | 2013-01-25 | Cinnamylate ester compound and application thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201310043754.0A CN103130663B (en) | 2013-01-25 | 2013-01-25 | Cinnamylate ester compound and application thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN103130663A true CN103130663A (en) | 2013-06-05 |
| CN103130663B CN103130663B (en) | 2014-06-18 |
Family
ID=48491169
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201310043754.0A Expired - Fee Related CN103130663B (en) | 2013-01-25 | 2013-01-25 | Cinnamylate ester compound and application thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN103130663B (en) |
Cited By (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN103641781A (en) * | 2013-12-26 | 2014-03-19 | 青岛科技大学 | Application of naphthalimide type compounds and salts thereof as plant growth regulators |
| CN103694129A (en) * | 2013-12-26 | 2014-04-02 | 青岛科技大学 | Beet alkaline plant growth regulator containing cinnamamide groups |
| CN103719082A (en) * | 2014-01-22 | 2014-04-16 | 杭州宇龙化工有限公司 | Application of taking cinnamamides compound as plant growth regulator |
| CN104956862A (en) * | 2015-05-29 | 2015-10-07 | 蚌埠市乔峰农业蔬菜专业合作社 | Anti-disease broccoli cultivation method |
| CN105481762A (en) * | 2016-01-26 | 2016-04-13 | 青岛科技大学 | Cinnamic acid pyridine ethyl ester compound and application |
| CN106614765A (en) * | 2016-12-20 | 2017-05-10 | 韦界飞 | Agricultural insecticide |
| CN108348449A (en) * | 2016-09-13 | 2018-07-31 | 株式会社爱茉莉太平洋 | Dermatologic preparation composition is used containing germination the anti-aging of G. gracilis extract as active ingredient |
| CN108558683A (en) * | 2018-05-09 | 2018-09-21 | 青岛科技大学 | 2- (2- diethylamino ethoxies) -5- nitrophenols and its preparation method and application |
| CN111087305A (en) * | 2019-12-13 | 2020-05-01 | 南京工业大学 | β -fluoroalkyl cinnamate compound and preparation method thereof |
| CN116675604A (en) * | 2023-04-26 | 2023-09-01 | 镇江广为生物科技有限公司 | Biological preparation process and application of high-activity cinnamyl ester compound |
| CN118005519A (en) * | 2024-01-02 | 2024-05-10 | 四川施成收生态科技有限公司 | Compound containing oxalate structure, preparation method and application |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4808208A (en) * | 1986-08-04 | 1989-02-28 | Canadian Patents & Development Ltd. | Phenolic safeners for glyphosate herbicides |
| CN1091123A (en) * | 1993-02-18 | 1994-08-24 | 武汉大学 | Plant-growth regulator N, N dimethylamine base ethyl-4-chloro-α-isopropyl benzyl carboxylate hydrochloride synthetic |
| WO1997035471A2 (en) * | 1996-03-25 | 1997-10-02 | Proguard, Inc. | Use of aromatic aldehydes as pesticides |
| JP2011046631A (en) * | 2009-08-26 | 2011-03-10 | National Institute For Agro-Environmental Science | Cis-cinnamic acid derivative compound and plant growth regulator |
-
2013
- 2013-01-25 CN CN201310043754.0A patent/CN103130663B/en not_active Expired - Fee Related
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4808208A (en) * | 1986-08-04 | 1989-02-28 | Canadian Patents & Development Ltd. | Phenolic safeners for glyphosate herbicides |
| CN1091123A (en) * | 1993-02-18 | 1994-08-24 | 武汉大学 | Plant-growth regulator N, N dimethylamine base ethyl-4-chloro-α-isopropyl benzyl carboxylate hydrochloride synthetic |
| WO1997035471A2 (en) * | 1996-03-25 | 1997-10-02 | Proguard, Inc. | Use of aromatic aldehydes as pesticides |
| JP2011046631A (en) * | 2009-08-26 | 2011-03-10 | National Institute For Agro-Environmental Science | Cis-cinnamic acid derivative compound and plant growth regulator |
Non-Patent Citations (4)
| Title |
|---|
| 《Journal of the Pennsylvania Academy of Science》 20011231 Beres,James A 等 Inhibition of cytochrome oxidase by beta-dimethylaminoethyl cinnamates 第43-47页 1 第74卷, 第2&3期 * |
| BERES,JAMES A 等: "Effects of structural modification of N,N-dimethylaminoethyl cinnamates on inhibition of acetylcholinesterase", 《JOURNAL OF THE PENNSYLVANIA ACADEMY OF SCIENCE》, vol. 69, no. 3, 31 December 1995 (1995-12-31), pages 115 - 120 * |
| BERES,JAMES A 等: "Inhibition of cytochrome oxidase by β-dimethylaminoethyl cinnamates", 《JOURNAL OF THE PENNSYLVANIA ACADEMY OF SCIENCE》, vol. 74, no. 23, 31 December 2001 (2001-12-31), pages 43 - 47 * |
| 安悦 等: "七种芥子碱类似物的合成及其对家兔离体肠平滑肌张力的影响", 《药学学报》, vol. 46, no. 12, 12 December 2011 (2011-12-12), pages 1467 - 1 * |
Cited By (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN103694129A (en) * | 2013-12-26 | 2014-04-02 | 青岛科技大学 | Beet alkaline plant growth regulator containing cinnamamide groups |
| CN103694129B (en) * | 2013-12-26 | 2015-03-25 | 青岛科技大学 | Beet alkaline plant growth regulator containing cinnamamide groups |
| CN103641781A (en) * | 2013-12-26 | 2014-03-19 | 青岛科技大学 | Application of naphthalimide type compounds and salts thereof as plant growth regulators |
| CN103719082A (en) * | 2014-01-22 | 2014-04-16 | 杭州宇龙化工有限公司 | Application of taking cinnamamides compound as plant growth regulator |
| CN104956862A (en) * | 2015-05-29 | 2015-10-07 | 蚌埠市乔峰农业蔬菜专业合作社 | Anti-disease broccoli cultivation method |
| CN105481762A (en) * | 2016-01-26 | 2016-04-13 | 青岛科技大学 | Cinnamic acid pyridine ethyl ester compound and application |
| CN105481762B (en) * | 2016-01-26 | 2018-04-20 | 青岛科技大学 | A kind of cinnamic acid pyridine ethyl ester type compound and purposes |
| CN108348449B (en) * | 2016-09-13 | 2021-09-10 | 株式会社爱茉莉太平洋 | Anti-aging skin external composition containing extract of germinated semi-wild soybean as effective component |
| CN108348449A (en) * | 2016-09-13 | 2018-07-31 | 株式会社爱茉莉太平洋 | Dermatologic preparation composition is used containing germination the anti-aging of G. gracilis extract as active ingredient |
| CN106614765A (en) * | 2016-12-20 | 2017-05-10 | 韦界飞 | Agricultural insecticide |
| CN108558683A (en) * | 2018-05-09 | 2018-09-21 | 青岛科技大学 | 2- (2- diethylamino ethoxies) -5- nitrophenols and its preparation method and application |
| CN111087305A (en) * | 2019-12-13 | 2020-05-01 | 南京工业大学 | β -fluoroalkyl cinnamate compound and preparation method thereof |
| CN111087305B (en) * | 2019-12-13 | 2021-08-27 | 南京工业大学 | Beta-fluoroalkyl cinnamate compound and preparation method thereof |
| CN116675604A (en) * | 2023-04-26 | 2023-09-01 | 镇江广为生物科技有限公司 | Biological preparation process and application of high-activity cinnamyl ester compound |
| CN118005519A (en) * | 2024-01-02 | 2024-05-10 | 四川施成收生态科技有限公司 | Compound containing oxalate structure, preparation method and application |
Also Published As
| Publication number | Publication date |
|---|---|
| CN103130663B (en) | 2014-06-18 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN103130663A (en) | Cinnamylate ester compound and application thereof | |
| CN101941872B (en) | Pesticidal foliage fertilizer mixed with plant growth factors and preparation method thereof | |
| CN102762530B (en) | Substituted diphenylamine compounds, preparation method and use thereof | |
| JP2012528803A (en) | E-phenyl acrylate compounds containing substituted anilinopyrimidine groups and their use | |
| CN104926724B (en) | Naphthalene-dicarboxamide-contained bisquaternary ammonium salt type plant growth regulator | |
| CN102249996A (en) | 2-(quinoline-8-yl)carbonyl-cyclohexane-1,3-dione compounds | |
| JPH0578522B2 (en) | ||
| CN103641781A (en) | Application of naphthalimide type compounds and salts thereof as plant growth regulators | |
| CN103130769B (en) | 3-difluoro ethoxy-pyrazole amides compounds and application thereof | |
| CN107980784B (en) | Application of 5-aminopyrazole compounds in regulating plant growth | |
| CN105669549B (en) | A kind of naphthalene dicarboxamido ethyl naphthalene acetate compound and its application | |
| CN103709158B (en) | Pyrazolopyrimidine ureas compound and application thereof | |
| CN103539694B (en) | A kind of polysubstituted chrysanthemum anilide derivative and application thereof | |
| CN103636602B (en) | Application of naphthaleneacetamide type compounds and salts thereof as plant growth regulators | |
| CN103724265A (en) | Naphthalenedicarboxamide-containing compound and preparation method thereof as well as plant growth regulator composition comprising same | |
| CN105481762B (en) | A kind of cinnamic acid pyridine ethyl ester type compound and purposes | |
| CN105777618B (en) | A kind of methyl α-naphthyl acetate pyridine ethyl ester type compound and purposes | |
| CN103694128B (en) | A kind of Beet alkaline plant growth regulator containing sorb amide group | |
| CN106866505A (en) | Acetylsalicylic acid esters plant growth regulator | |
| CN105669550B (en) | A kind of aphthalimide base thylhexoic acid esters compound and its application | |
| CN106748979A (en) | A kind of 2,4,6 trichlorophenyl pyridine ethyl ether plant growth regulator | |
| CN107216291A (en) | Fragrant phenoxy group propionic acid compounds containing bishydrazide and preparation method and application | |
| CN103694129B (en) | Beet alkaline plant growth regulator containing cinnamamide groups | |
| CN103641732B (en) | Glycine betaine type plant growth regulator containing naphthalene acetamide groups | |
| CN108822082B (en) | A kind of fluorine-containing pyridine piperazine amide compound and its application |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| ASS | Succession or assignment of patent right |
Owner name: QINGDAO LVWANDA AGRICULTURAL CHEMICALS CO., LTD. Free format text: FORMER OWNER: QINGDAO UNIVERSITY OF SCIENCE + TECHNOLOGY Effective date: 20141010 |
|
| C41 | Transfer of patent application or patent right or utility model | ||
| COR | Change of bibliographic data |
Free format text: CORRECT: ADDRESS; FROM: 266061 QINGDAO, SHANDONG PROVINCE TO: 266000 QINGDAO, SHANDONG PROVINCE |
|
| TR01 | Transfer of patent right |
Effective date of registration: 20141010 Address after: 266000 Shandong province Qingdao City, Wuyang Road No. 7 Patentee after: Qingdao Wanda Green Agricultural Chemicals Co. Ltd. Address before: 266061 Shandong Province, Qingdao city Laoshan District Songling Road No. 99 Patentee before: Qingdao University of Science & Technology |
|
| CF01 | Termination of patent right due to non-payment of annual fee |
Granted publication date: 20140618 Termination date: 20190125 |
|
| CF01 | Termination of patent right due to non-payment of annual fee |