CN103084161A - Ce-Zr-Al-based composite oxide rare earth oxygen-storage material and preparation method thereof - Google Patents
Ce-Zr-Al-based composite oxide rare earth oxygen-storage material and preparation method thereof Download PDFInfo
- Publication number
- CN103084161A CN103084161A CN2011103464930A CN201110346493A CN103084161A CN 103084161 A CN103084161 A CN 103084161A CN 2011103464930 A CN2011103464930 A CN 2011103464930A CN 201110346493 A CN201110346493 A CN 201110346493A CN 103084161 A CN103084161 A CN 103084161A
- Authority
- CN
- China
- Prior art keywords
- cerium
- oxide
- zirconium
- aluminum
- rare earth
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Landscapes
- Catalysts (AREA)
- Inorganic Compounds Of Heavy Metals (AREA)
Abstract
一种铈锆铝基复合氧化物稀土储氧材料及其制备方法,所述铈锆铝基复合氧化物稀土储氧材料是由氧化铈、氧化锆、氧化铝和除铈以外的其他稀土氧化物组成的复合氧化物,其重量百分比如下:氧化铈:15-65%,氧化锆:12-55%,氧化铝:5-50%,除铈以外其他稀土氧化物:4-10%;与现有的产品相比,通过对氧化铝进行表面处理,加强氧化铝与铈锆基复合氧化物之间的相互作用,并充分地保证氧化铝与铈锆基复合氧化物之间的均一性;通过对沉淀剂选择及复配,控制沉淀终点的,确保了产品的收率;通过加入表面处理剂,保证了沉淀物前驱体在焙烧后的孔径、孔容较大,提高了材料的高温抗老化能力。
A cerium-zirconium-aluminum-based composite oxide rare-earth oxygen storage material and a preparation method thereof, the cerium-zirconium-aluminum-based composite oxide rare-earth oxygen storage material is composed of cerium oxide, zirconia, aluminum oxide and other rare earth oxides except cerium The composition of the composite oxide, its weight percentage is as follows: cerium oxide: 15-65%, zirconia: 12-55%, aluminum oxide: 5-50%, other rare earth oxides except cerium: 4-10%; Compared with some products, the interaction between alumina and cerium-zirconium-based composite oxides is strengthened by surface treatment of alumina, and the uniformity between alumina and cerium-zirconium-based composite oxides is fully guaranteed; through The selection and compounding of the precipitating agent and the control of the precipitation end point ensure the yield of the product; by adding the surface treatment agent, the pore diameter and pore volume of the precipitate precursor after roasting are ensured, which improves the high temperature anti-aging of the material ability.
Description
技术领域 technical field
本发明涉及一种稀土储氧材料及其制备方法,特别涉及一种用于汽车尾气净化、有机废气消除和天然气催化燃烧等废气净化或催化燃烧的铈锆铝基复合氧化物稀土储氧材料及其制备方法。The invention relates to a rare earth oxygen storage material and a preparation method thereof, in particular to a cerium-zirconium-aluminum-based composite oxide rare earth oxygen storage material used for exhaust gas purification or catalytic combustion such as automobile exhaust purification, organic waste gas elimination, and natural gas catalytic combustion, and its preparation method.
背景技术 Background technique
减少汽车尾气污染、保护大气环境质量,实现生态环境和社会的可持续发展,是目前及未来相当长时间内我国环境保护的重要课题之Reducing vehicle exhaust pollution, protecting the quality of the atmospheric environment, and realizing the sustainable development of the ecological environment and society are one of the important issues of my country's environmental protection at present and for a long time to come.
为减少污染,必须从提高汽油品质、改善发动机燃烧状态和增加尾气净化催化效率三方面进行突破。到目前为止,使用汽车尾气净化催化剂是最有效途径就是通过三元催化装置来降低尾气中HC、CO和NOx三种有毒气体的含量。In order to reduce pollution, breakthroughs must be made in three aspects: improving the quality of gasoline, improving the combustion state of the engine, and increasing the catalytic efficiency of exhaust gas purification. So far, the most effective way to use automobile exhaust purification catalysts is to reduce the content of three toxic gases, HC, CO and NOx, in the exhaust gas through a three-way catalytic device.
三效催化剂主要是由起催化作用的贵金属活性组分、含CeO2催化助剂(储氧材料)和用于支撑活性组分的载体组成。而合适的汽车尾气净化储氧涂层材料对贵金属的分散和稳定起到关键作用,同时储氧涂层材料可以提高高温条件下催化剂的储氧能力,扩大其空燃比操作窗口,使催化剂在十分苛刻的操作条件下活性有明显改善。The three-way catalyst is mainly composed of a noble metal active component that plays a catalytic role, a catalytic promoter (oxygen storage material) containing CeO 2 and a carrier for supporting the active component. A suitable oxygen storage coating material for automobile exhaust purification plays a key role in the dispersion and stability of precious metals. At the same time, the oxygen storage coating material can improve the oxygen storage capacity of the catalyst under high temperature conditions, expand its air-fuel ratio operating window, and make the catalyst operate at a very high temperature. The activity is significantly improved under harsh operating conditions.
目前商业上用的含铈的稀土储氧材料和具有高比表面积和较强抗热老化能力的γ-Al2O3是分别制备的。The currently commercially used cerium-containing rare earth oxygen storage materials and γ-Al 2 O 3 with high specific surface area and strong thermal aging resistance were prepared separately.
由于CeO2的热稳定性较差,限制了它的使用。因此,在高温下使用时必须抑制CeO2的烧结。当将CeO2中的铈离子用其它阳离子部分取代时,可产生结构缺陷,明显提高其高温热稳定性。Due to the poor thermal stability of CeO2 , its use is limited. Therefore, the sintering of CeO2 must be suppressed when used at high temperature. When the cerium ions in CeO2 are partially replaced with other cations, structural defects can be generated, which significantly improves its high-temperature thermal stability.
ZrO2添加到CeO2中,可形成CeO2-ZrO2固溶体而改善CeO2的体相特性,利于体相氧原子的迁移和扩散,使体相反应过程变得活泼,能够提高CeO2的储氧能力和高温稳定性。同时,Pr、Nd、Y、Nb、La和Ba等稀土及碱土元素的加入可进一步增强储氧材料的抗高温老化性,提高其储氧量。储氧材料可有效稳定活性组分的分散,因此具有大的比表面积、较大的孔体积及合适的孔径分布,并具有良好的抗高温老化性能、优异的低温催化性能的铈锆基储氧材料成为新三效催化剂的关键材料。Adding ZrO 2 to CeO 2 can form a CeO 2 -ZrO 2 solid solution to improve the bulk phase properties of CeO 2 , facilitate the migration and diffusion of bulk oxygen atoms, make the bulk phase reaction process more active, and improve the storage capacity of CeO 2 Oxygen capacity and high temperature stability. At the same time, the addition of rare earth and alkaline earth elements such as Pr, Nd, Y, Nb, La, and Ba can further enhance the high temperature aging resistance of the oxygen storage material and increase its oxygen storage capacity. The oxygen storage material can effectively stabilize the dispersion of the active components, so it has a large specific surface area, a large pore volume and a suitable pore size distribution, and has good high-temperature aging resistance and excellent low-temperature catalytic performance. Cerium-zirconium-based oxygen storage Material becomes the key material of the new three-way catalyst.
γ-Al2O3的热稳定性、比表面积、孔容、孔径分布、表面酸性以及NO吸附能力在很大程度上影响催化剂的催化性能。提高γ-Al2O3的高温热稳定性和表面性能对提高催化剂的活性和使用寿命具有重要意义。研究表明。通过添加稀土、碱土元素等可以提高氧化铝的高温热稳定性,抑制氧化铝由γ相向α相的转变。工业上目前广泛使用的是通过稀土元素或过渡金属元素对拟薄水铝石进行表面改性后经焙烧制备具有较强抗热老化能力的氧化铝。The thermal stability, specific surface area, pore volume, pore size distribution, surface acidity and NO adsorption capacity of γ-Al 2 O 3 largely affect the catalytic performance of the catalyst. Improving the high-temperature thermal stability and surface properties of γ-Al 2 O 3 is of great significance for improving the activity and service life of the catalyst. research shows. The high-temperature thermal stability of alumina can be improved by adding rare earth elements, alkaline earth elements, etc., and the transformation of alumina from γ phase to α phase can be inhibited. At present, it is widely used in the industry to modify the surface of pseudo-boehmite with rare earth elements or transition metal elements and then roast it to prepare alumina with strong thermal aging resistance.
用铈锆基固溶体等稀土材料与氧化铝相互改性,制备铈锆铝基复合材料,使其兼具储氧材料和氧化铝的共同优点,提高储氧材料及氧化铝的高温抗老化能力,是新一代催化涂层的关键技术。Use rare earth materials such as cerium-zirconium-based solid solution and alumina to modify each other to prepare cerium-zirconium-aluminum matrix composite materials, so that it has the common advantages of oxygen storage materials and alumina, and improves the high-temperature anti-aging ability of oxygen storage materials and alumina. It is the key technology of the new generation of catalytic coating.
专利号为200510020615.1的发明专利公开了一种铈锆铝基储氧材料及其制备方法,其基本组成是由氧化铈、氧化锆、氧化铝和稳定剂,稳定剂选自氧化镧、氧化钇及碱土金属氧化物的至少一种;其制备工艺是(1)按给定的材料组成组分制备硝酸盐混合溶液及碱性沉淀剂;(2)将制备好的两种溶液并流导入反应容器中进行沉淀,沉淀结束后在90-100℃陈化不小于2h;(3)陈化后的反应料液进行固液分离,分离出的固相沉淀物进行洗涤、然后与表面活性剂和水配成浆液;(4)浆液通过蒸发干燥、然后焙烧得铈锆铝基储氧材料。The invention patent with the patent number of 200510020615.1 discloses a cerium-zirconium-aluminum-based oxygen storage material and its preparation method. Its basic composition is cerium oxide, zirconia, aluminum oxide and a stabilizer. At least one of alkaline earth metal oxides; the preparation process is (1) preparing a mixed solution of nitrate and an alkaline precipitant according to a given material composition; (2) introducing the prepared two solutions into a reaction vessel in parallel Precipitate at 90-100°C for no less than 2 hours after the precipitation; (3) The solid-liquid separation of the aged reaction feed liquid is carried out, and the separated solid-phase precipitate is washed and then mixed with surfactant and water Prepare a slurry; (4) The slurry is dried by evaporation and then fired to obtain a cerium-zirconium-aluminum-based oxygen storage material.
用上述方法制备的储氧材料的比表面剂在1000℃焙烧5h后最高可达110m2/g以上,至少可达到38m2/g以上;但是,该方法以硝酸铝为原料,成本较高,同时制备出的储氧材料孔径较小、储氧材料的高温抗老化性能较差。The specific surface agent of the oxygen storage material prepared by the above method can reach a maximum of 110m 2 /g or more than 38m 2 /g after calcination at 1000°C for 5 hours; however, this method uses aluminum nitrate as a raw material, and the cost is relatively high. At the same time, the pore size of the prepared oxygen storage material is small, and the high temperature anti-aging performance of the oxygen storage material is poor.
专利公开号为CN101940921A的发明专利公开了一种双层结构储氧材料及其制备方法,由铈锆固溶体与大比表面γ-Al2O3复合而成,其制备方法是:(1)取硝酸镧溶液、硝酸铈溶液、硝酸钇及硝酸锆溶液,置入容器内加入氨水进行沉淀得到沉淀物;(2)在步骤(1)的沉淀物中加入氧化铈质量的工业双氧水,得到铈锆前驱化合物;(3)将前驱体与γ-Al2O3质量比1∶1混合、搅拌均匀得到混合物;(4)将步骤(3)中的混合物经洗涤、过滤、加入表面活性剂后,再经烘干、焙烧、气流粉碎,得双层结构的粉体储氧材料。The invention patent with the patent publication number CN101940921A discloses a double-layer structure oxygen storage material and its preparation method, which is composed of a cerium-zirconium solid solution and a large specific surface γ-Al 2 O 3 . Lanthanum nitrate solution, cerium nitrate solution, yttrium nitrate and zirconium nitrate solution are placed in a container and ammonia water is added to precipitate to obtain a precipitate; (2) industrial hydrogen peroxide of cerium oxide quality is added to the precipitate in step (1) to obtain cerium-zirconium Precursor compound; (3) Mixing the precursor and γ-Al 2 O 3 in a mass ratio of 1:1 and stirring to obtain a mixture; (4) Washing, filtering, and adding a surfactant to the mixture in step (3), After drying, roasting, and jet crushing, a powder oxygen storage material with a double-layer structure is obtained.
上述方法以铈锆前驱体与γ-Al2O3机械混合,这就导致了氧化铝与铈锆复合氧化物的混合均匀性及氧化铝与铈锆复合氧化物的粘结性较差,影响储氧材料的催化性能及高温热稳定剂。The above method uses cerium-zirconium precursors and γ-Al 2 O 3 to mechanically mix, which leads to poor mixing uniformity of alumina and cerium-zirconium composite oxides and poor adhesion between alumina and cerium-zirconium composite oxides, which affects Catalytic properties of oxygen storage materials and high temperature thermal stabilizers.
专利号为98108256.4的发明专利公开了一种具有高热稳定性的储氧材料及其制备该材料的方法,把氧化铈和至少一种选自氧化镨、氧化镧、氧化钇及氧化钕的稳定剂高度分散在氧化铝、氧化锆、氧化钛、二氧化硅或其他氧化物等大表面积载体的比表面上,然后再经焙烧而得;其制备方法是把溶解的所述的稳定剂的前体物和氧化铈加到一种事先准备好的、不断搅拌着的所选载体物质的水分散物中,然后通过加碱,慢慢提高分散物的pH值至8-10,从而使稳定剂和氧化铈沉淀在载体物质上。Patent No. 98108256.4 discloses an oxygen storage material with high thermal stability and a method for preparing the material. Cerium oxide and at least one stabilizer selected from praseodymium oxide, lanthanum oxide, yttrium oxide, and neodymium oxide It is highly dispersed on the specific surface of large surface area carriers such as alumina, zirconia, titania, silica or other oxides, and then roasted; its preparation method is to dissolve the precursor of the stabilizer and cerium oxide are added to a previously prepared, constantly stirring aqueous dispersion of the selected carrier material, and then slowly increase the pH of the dispersion to 8-10 by adding alkali, so that the stabilizer and Cerium oxide is precipitated on the support material.
上述方法没有对载体进行表面处理,而载体的颗粒大小、形貌等均影响着包覆的均匀性及涂覆层与载体的粘结力,影响储氧材料的催化活性及高温热稳定性。The above method does not carry out surface treatment on the carrier, and the particle size and shape of the carrier affect the uniformity of the coating and the adhesion between the coating layer and the carrier, and affect the catalytic activity and high temperature thermal stability of the oxygen storage material.
综上,现有对于铈锆铝基稀土储氧材料的研究较少,且铈锆与氧化铝之间的结合力及高温抗老化性存在着较大的矛盾,材料的高温抗老化性较差。In summary, there are few studies on cerium-zirconium-aluminum-based rare earth oxygen storage materials, and there is a big contradiction between the binding force between cerium-zirconium and alumina and the high-temperature aging resistance, and the high-temperature aging resistance of the material is poor. .
因此,特别需要一种铈锆铝基复合氧化物稀土储氧材料及其制备方法,已解决上述现有存在的问题。Therefore, there is a special need for a cerium-zirconium-aluminum-based composite oxide rare earth oxygen storage material and a preparation method thereof, which have solved the above-mentioned existing problems.
发明内容 Contents of the invention
本发明的目的在于提供一种铈锆铝基复合氧化物稀土储氧材料及其制备方法,具有比表面积大、高温抗老化能力好、储放氧能力高等特点,适用于汽车尾气净化、有机废气消除和天然气催化燃烧等废气净化或催化燃烧。。The purpose of the present invention is to provide a cerium-zirconium-aluminum-based composite oxide rare earth oxygen storage material and its preparation method, which has the characteristics of large specific surface area, good high temperature anti-aging ability, high oxygen storage and release ability, etc., and is suitable for automobile exhaust gas purification, organic waste gas Exhaust gas purification or catalytic combustion such as elimination and catalytic combustion of natural gas. .
为了实现上述目的,本发明的技术方案如下:In order to achieve the above object, the technical scheme of the present invention is as follows:
一方面,本发明提供一种铈锆铝基复合氧化物稀土储氧材料,其特征在于,所述铈锆铝基复合氧化物稀土储氧材料是由氧化铈、氧化锆、氧化铝和除铈以外的其他稀土氧化物组成的复合氧化物,其重量百分比如下:On the one hand, the present invention provides a cerium-zirconium-aluminum-based composite oxide rare-earth oxygen storage material, characterized in that the cerium-zirconium-aluminum-based composite oxide rare-earth oxygen storage material is made of cerium oxide, zirconium oxide, aluminum oxide and decerized The composite oxide composed of other rare earth oxides, the weight percentage is as follows:
氧化铈:15-65%Cerium oxide: 15-65%
氧化锆:12-55%Zirconia: 12-55%
氧化铝:5-50%Alumina: 5-50%
除铈以外其他稀土氧化物:4-10%。Rare earth oxides other than cerium: 4-10%.
在本发明的一个实施例中,所述除铈以外的其他稀土氧化物选自氧化镧、氧化钇、氧化镨和氧化钕中的一种或几种。In one embodiment of the present invention, the rare earth oxides other than cerium are selected from one or more of lanthanum oxide, yttrium oxide, praseodymium oxide and neodymium oxide.
另一方面,本发明提供一种铈锆铝基复合氧化物稀土储氧材料的制备方法,其特征在于,它包括如下步骤:In another aspect, the present invention provides a method for preparing a cerium-zirconium-aluminum-based composite oxide rare earth oxygen storage material, which is characterized in that it includes the following steps:
(1)铝溶胶的制备,将氧化铝原料与水混合,加入硝酸调节至pH为2-3,剪切分散,高能球磨,形成透明的铝溶胶A;(1) Preparation of aluminum sol, mixing alumina raw material with water, adding nitric acid to adjust the pH to 2-3, shearing dispersion, high-energy ball milling, to form transparent aluminum sol A;
(2)混合金属盐溶液的制备,根据材料组分构成,确定氧化铈、氧化锆及除铈以外的其他稀土氧化物的含量,换算成相应的硝酸盐或碳酸盐的重量含量,然后将硝酸铈、除铈以外的其他稀土可溶性硝酸盐和硝酸锆溶于水,得混合金属硝酸盐溶液B;(2) the preparation of mixed metal salt solution, according to material composition, determine the content of cerium oxide, zirconium oxide and other rare earth oxides except cerium, convert into the weight content of corresponding nitrate or carbonate, then Cerium nitrate, soluble nitrates of rare earths other than cerium and zirconium nitrate are dissolved in water to obtain mixed metal nitrate solution B;
(3)活性组分前驱体的制备,根据材料的组成,将铝溶胶A和混合金属硝酸盐溶液B混合,得活性组分前驱体C;(3) Preparation of the active component precursor, according to the composition of the material, the aluminum sol A and the mixed metal nitrate solution B are mixed to obtain the active component precursor C;
(4)沉淀,在一定温度下,将碱性沉淀剂加入上述活性组分前驱体C中,直到沉淀终点,加入化学计量的双氧水,保温一定时间,得沉淀物D;(4) Precipitation, at a certain temperature, add an alkaline precipitating agent to the above-mentioned active component precursor C until the end of the precipitation, add stoichiometric hydrogen peroxide, keep warm for a certain period of time, and obtain a precipitate D;
(5)沉淀物的表面处理,将沉淀物D进行脱水、洗涤;将脱水后的沉淀物D与水以质量比2∶1重新打浆,加入一定量的表面处理剂,剪切分散;(5) Surface treatment of the precipitate, dehydrating and washing the precipitate D; repulping the dehydrated precipitate D and water at a mass ratio of 2:1, adding a certain amount of surface treatment agent, and shearing and dispersing;
(6)焙烧,将经过表面处理的沉淀物D在450-800℃下焙烧4-8h,得所述铈锆铝基固溶体稀土储氧材料。(6) Roasting, calcining the surface-treated precipitate D at 450-800° C. for 4-8 hours to obtain the cerium-zirconium-aluminum-based solid solution rare earth oxygen storage material.
在本发明的一个实施例中,步骤(1)中,所述氧化铝原料选自大孔γ-Al2O3或孔容≥0.9cm3/g的大孔拟薄水铝石。In one embodiment of the present invention, in step (1), the alumina raw material is selected from macroporous γ-Al 2 O 3 or macroporous pseudo-boehmite with a pore volume ≥ 0.9 cm 3 /g.
在本发明的一个实施例中,所述氧化铝与水的混合浊液的固含量为5-10%。In one embodiment of the present invention, the solid content of the mixed turbid liquid of alumina and water is 5-10%.
在本发明的一个实施例中,步骤(2)中,所述氧化铈来源选自硝酸铈或碳酸铈;所述氧化锆来源选自碳酸氧锆;所述除铈以外的其他稀土可溶性硝酸盐选自硝酸镧、硝酸钇、硝酸镨和硝酸钕中的一种或几种或硝酸与氧化镧、氧化钇、氧化镨和氧化钕反应生成相应的硝酸盐溶液。In one embodiment of the present invention, in step (2), the cerium oxide source is selected from cerium nitrate or cerium carbonate; the zirconia source is selected from zirconium oxycarbonate; the other rare earth soluble nitrates except cerium One or more selected from lanthanum nitrate, yttrium nitrate, praseodymium nitrate and neodymium nitrate or nitric acid reacts with lanthanum oxide, yttrium oxide, praseodymium oxide and neodymium oxide to form a corresponding nitrate solution.
在本发明的一个实施例中,步骤(4)中,所述碱性沉淀剂选自氨水和碳酸氢铵中的一种或几种;所述沉淀的温度为60-90℃;所述沉淀终点为:当选用氨水为沉淀剂时,沉淀的终点pH为9,当沉淀剂含有碳酸氢铵时,沉淀的终点为6~6.5;所述保温时间为1-3h。In one embodiment of the present invention, in step (4), the alkaline precipitation agent is selected from one or more of ammonia water and ammonium bicarbonate; the temperature of the precipitation is 60-90°C; the precipitation The end point is: when ammonia water is selected as the precipitant, the pH of the end point of the precipitation is 9, and when the precipitant contains ammonium bicarbonate, the end point of the precipitation is 6-6.5; the holding time is 1-3h.
在本发明的一个实施例中,步骤(5)中,所述脱水为板框压滤或离心脱水的常规脱水方式;所述表面处理剂选自月桂酸、硬脂酸和烯基琥珀酸酐中的一种或几种,所述表面处理剂的加入量为重量百分比30-50%。In one embodiment of the present invention, in step (5), the dehydration is a conventional dehydration method of plate and frame filter press or centrifugal dehydration; the surface treatment agent is selected from lauric acid, stearic acid and alkenyl succinic anhydride One or more of them, the added amount of the surface treatment agent is 30-50% by weight.
本发明的铈锆铝基复合氧化物稀土储氧材料及其制备方法,与现有的产品相比,通过对氧化铝进行表面处理,加强氧化铝与铈锆基复合氧化物之间的相互作用,并充分地保证氧化铝与铈锆基复合氧化物之间的均一性;通过对沉淀剂选择及复配,控制沉淀终点的,确保了产品的收率;通过加入表面处理剂,保证了沉淀物前驱体在焙烧后的孔径、孔容较大,提高了材料的高温抗老化能力,使制备得到的铈锆铝基复合氧化物储氧材料,具有储氧能力高、储放氧速度快,高温抗老化能力强等优点,实现本发明的目的。Compared with the existing products, the cerium-zirconium-aluminum-based composite oxide rare earth oxygen storage material and the preparation method thereof of the present invention strengthen the interaction between the alumina and the cerium-zirconium-based composite oxide by surface-treating the alumina , and fully ensure the uniformity between alumina and cerium-zirconium-based composite oxides; through the selection and compounding of precipitating agents, the precipitation endpoint is controlled to ensure the product yield; by adding surface treatment agents, the precipitation is guaranteed The pore diameter and pore volume of the precursor after calcination are larger, which improves the high-temperature anti-aging ability of the material, so that the prepared cerium-zirconium-aluminum-based composite oxide oxygen storage material has high oxygen storage capacity and fast oxygen storage and release speed. The invention has the advantages of strong anti-aging ability under high temperature and the like, and realizes the object of the present invention.
本发明的特点可参阅本案图式及以下较好实施方式的详细说明而获得清楚地了解。The features of the present invention can be clearly understood by referring to the drawings of the present invention and the detailed description of the following preferred embodiments.
附图说明 Description of drawings
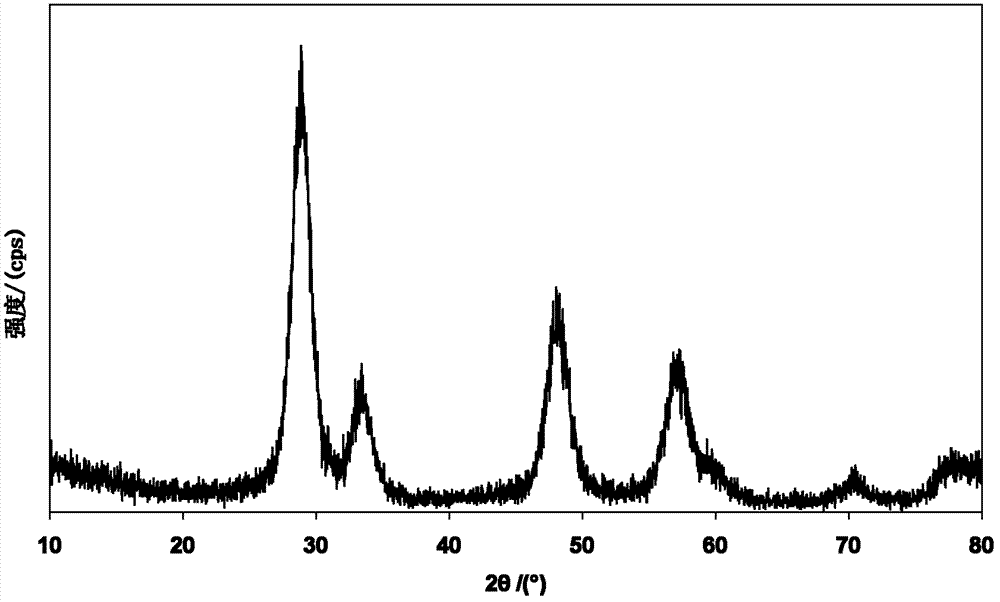
图1为本发明的实施例1制备纳米铈锆基固溶体稀土储氧材料的XRD示意图。Fig. 1 is an XRD schematic diagram of the nano-cerium-zirconium-based solid solution rare earth oxygen storage material prepared in Example 1 of the present invention.
具体实施方式 Detailed ways
为了使本发明实现的技术手段、创作特征、达成目的与功效易于明白了解,下面结合具体实施例进一步阐述本发明。In order to make the technical means, creative features, objectives and effects achieved by the present invention easy to understand, the present invention will be further described below in conjunction with specific embodiments.
本发明的铈锆铝基复合氧化物稀土储氧材料,所述铈锆铝基复合氧化物稀土储氧材料是由氧化铈、氧化锆、氧化铝和除铈以外的其他稀土氧化物组成的复合氧化物,其重量百分比如下:The cerium-zirconium-aluminum-based composite oxide rare earth oxygen storage material of the present invention, the cerium-zirconium-aluminum-based composite oxide rare earth oxygen storage material is a composite composed of cerium oxide, zirconium oxide, aluminum oxide and other rare earth oxides except cerium Oxide, its weight percent is as follows:
氧化铈:15-65%Cerium oxide: 15-65%
氧化锆:12-55%Zirconia: 12-55%
氧化铝:5-50%Alumina: 5-50%
除铈以外其他稀土氧化物:4-10%;本发明的铈锆铝基复合氧化物稀土储氧材料的XRD示意图如图1所示。Rare earth oxides other than cerium: 4-10%; the XRD schematic diagram of the cerium-zirconium-aluminum-based composite oxide rare earth oxygen storage material of the present invention is shown in FIG. 1 .
在本发明中,所述除铈以外的其他稀土氧化物选自氧化镧、氧化钇、氧化镨和氧化钕中的一种或几种。In the present invention, the rare earth oxides other than cerium are selected from one or more of lanthanum oxide, yttrium oxide, praseodymium oxide and neodymium oxide.
本发明的铈锆铝基复合氧化物稀土储氧材料的制备方法,它包括如下步骤:The preparation method of the cerium-zirconium-aluminum-based composite oxide rare earth oxygen storage material of the present invention comprises the following steps:
(1)铝溶胶的制备,将氧化铝原料与水混合,加入硝酸调节至pH为2-3,剪切分散,高能球磨,形成透明的铝溶胶A;(1) Preparation of aluminum sol, mixing alumina raw material with water, adding nitric acid to adjust the pH to 2-3, shearing dispersion, high-energy ball milling, to form transparent aluminum sol A;
(2)混合金属盐溶液的制备,根据材料组分构成,确定氧化铈、氧化锆及除铈以外的其他稀土氧化物的含量,换算成相应的硝酸盐或碳酸盐的重量含量,然后将硝酸铈、除铈以外的其他稀土可溶性硝酸盐和硝酸锆溶于水,得混合金属硝酸盐溶液B;(2) the preparation of mixed metal salt solution, according to material composition, determine the content of cerium oxide, zirconium oxide and other rare earth oxides except cerium, convert into the weight content of corresponding nitrate or carbonate, then Cerium nitrate, soluble nitrates of rare earths other than cerium and zirconium nitrate are dissolved in water to obtain mixed metal nitrate solution B;
(3)活性组分前驱体的制备,根据材料的组成,将铝溶胶A和混合金属硝酸盐溶液B混合,得活性组分前驱体C;(3) Preparation of the active component precursor, according to the composition of the material, the aluminum sol A and the mixed metal nitrate solution B are mixed to obtain the active component precursor C;
(4)沉淀,在一定温度下,将碱性沉淀剂加入上述活性组分前驱体C中,直到沉淀终点,加入化学计量的双氧水,保温一定时间,得沉淀物D;(4) Precipitation, at a certain temperature, add an alkaline precipitating agent to the above-mentioned active component precursor C until the end of the precipitation, add stoichiometric hydrogen peroxide, keep warm for a certain period of time, and obtain a precipitate D;
(5)沉淀物的表面处理,将沉淀物D进行脱水、洗涤;将脱水后的沉淀物D与水以质量比2∶1重新打浆,加入一定量的表面处理剂,剪切分散;(5) Surface treatment of the precipitate, dehydrating and washing the precipitate D; repulping the dehydrated precipitate D and water at a mass ratio of 2:1, adding a certain amount of surface treatment agent, and shearing and dispersing;
(6)焙烧,将经过表面处理的沉淀物D在450-800℃下焙烧4-8h,得所述铈锆铝基固溶体稀土储氧材料。(6) Roasting, calcining the surface-treated precipitate D at 450-800° C. for 4-8 hours to obtain the cerium-zirconium-aluminum-based solid solution rare earth oxygen storage material.
在本发明中,步骤(1)中,所述氧化铝原料选自大孔γ-Al2O3或孔容≥0.9cm3/g的大孔拟薄水铝石。In the present invention, in step (1), the alumina raw material is selected from macroporous γ-Al 2 O 3 or macroporous pseudoboehmite with a pore volume ≥ 0.9 cm 3 /g.
在本发明中,所述氧化铝与水的混合浊液的固含量为5-10%。In the present invention, the solid content of the mixed turbid liquid of alumina and water is 5-10%.
在本发明中,步骤(2)中,所述氧化铈来源选自硝酸铈或碳酸铈;所述氧化锆来源选自碳酸氧锆;所述除铈以外的其他稀土可溶性硝酸盐选自硝酸镧、硝酸钇、硝酸镨和硝酸钕中的一种或几种或硝酸与氧化镧、氧化钇、氧化镨和氧化钕反应生成相应的硝酸盐溶液。In the present invention, in step (2), the source of cerium oxide is selected from cerium nitrate or cerium carbonate; the source of zirconia is selected from zirconyl carbonate; the other rare earth soluble nitrates other than cerium are selected from lanthanum nitrate One or more of yttrium nitrate, praseodymium nitrate and neodymium nitrate or nitric acid reacts with lanthanum oxide, yttrium oxide, praseodymium oxide and neodymium oxide to form the corresponding nitrate solution.
在本发明中,步骤(4)中,所述碱性沉淀剂选自氨水和碳酸氢铵中的一种或几种;所述沉淀的温度为60-90℃;所述沉淀终点为:当选用氨水为沉淀剂时,沉淀的终点pH为9,当沉淀剂含有碳酸氢铵时,沉淀的终点为6~6.5;所述保温时间为1-3h。In the present invention, in step (4), the alkaline precipitating agent is selected from one or more of ammonia water and ammonium bicarbonate; the temperature of the precipitation is 60-90°C; the end point of the precipitation is: When ammonia water is used as the precipitant, the pH of the end point of the precipitation is 9, and when the precipitant contains ammonium bicarbonate, the pH of the end point of the precipitation is 6-6.5; the holding time is 1-3h.
在本发明中,步骤(5)中,所述脱水为板框压滤或离心脱水的常规脱水方式;所述表面处理剂选自月桂酸、硬脂酸和烯基琥珀酸酐中的一种或几种,所述表面处理剂的加入量为重量百分比30-50%。In the present invention, in step (5), the dehydration is a conventional dehydration method of plate and frame filter press or centrifugal dehydration; the surface treatment agent is selected from one of lauric acid, stearic acid and alkenyl succinic anhydride or Several kinds, the adding amount of the surface treatment agent is 30-50% by weight.
实施例1Example 1
将5kg的γ-Al2O3加入140kg水中,用硝酸调节至pH为2,经剪切分散、高能球磨得透明溶胶A;将57kg碳酸氧锆、3.5kg的氧化钇用硝酸溶解,加入14.25kg硝酸镧、172kg硝酸铈,2000kg水,得澄清透明溶液B。将溶液B和A混合,加热至60℃后,缓慢加入氨水,控制终点的pH为9,加入双氧水24kg,保温3h。经沉淀物离心脱水后与水按照质量比2∶1进行混合,加入月桂酸30kg,剪切分散。将分散后的浊液经450℃焙烧8h,得所述铈锆铝基稀土储氧材料。Add 5kg of γ-Al 2 O 3 into 140kg of water, adjust the pH to 2 with nitric acid, and obtain transparent sol A through shear dispersion and high-energy ball milling; dissolve 57kg of zirconyl carbonate and 3.5kg of yttrium oxide with nitric acid, and add kg of lanthanum nitrate, 172 kg of cerium nitrate, and 2000 kg of water to obtain a clear and transparent solution B. Mix solutions B and A, heat to 60°C, slowly add ammonia water, control the pH at the end point to 9, add 24 kg of hydrogen peroxide, and keep warm for 3 hours. After the sediment was centrifuged and dehydrated, it was mixed with water at a mass ratio of 2:1, and 30 kg of lauric acid was added for shear dispersion. The dispersed turbid liquid was calcined at 450° C. for 8 hours to obtain the cerium-zirconium-aluminum-based rare earth oxygen storage material.
其中,氧化铈63.7%,氧化锆22.8%,氧化铝5%,氧化钇3.5%,氧化镧5%。Among them, 63.7% of cerium oxide, 22.8% of zirconia, 5% of aluminum oxide, 3.5% of yttrium oxide, and 5% of lanthanum oxide.
实施例2Example 2
将70kg大孔拟薄水铝石将入到700kg水中,用硝酸调节至pH为3,经剪切分散、高能球磨得透明溶胶A;将7kg氧化镨、7kg氧化钕、140kg碳酸氧锆用硝酸溶解,加入81kg硝酸铈,2000kg水,得澄清透明溶液B;将溶液B和A混合,加热至90℃后,缓慢加入用氨水与碳酸氢铵饱和溶液按照质量比1∶1混合的沉淀剂,控制终点的pH为6.5,加入双氧水11kg,保温1h。经沉淀物离心脱水后与水按照质量比2∶1进行混合,加入硬脂酸酸50kg,剪切分散。将分散后的浊液经800℃焙烧4h,得所述铈锆铝基稀土储氧材料。Put 70kg of macroporous pseudo-boehmite into 700kg of water, adjust the pH to 3 with nitric acid, and obtain transparent sol A through shear dispersion and high-energy ball milling; mix 7kg of praseodymium oxide, 7kg of neodymium oxide, and 140kg of zirconium oxycarbonate with nitric acid Dissolve, add 81kg of cerium nitrate and 2000kg of water to obtain a clear and transparent solution B; mix solution B and A, heat to 90°C, slowly add a precipitant mixed with ammonia water and saturated ammonium bicarbonate solution in a mass ratio of 1:1, Control the pH at the end point to 6.5, add 11 kg of hydrogen peroxide, and keep warm for 1 hour. After the precipitate is centrifuged and dehydrated, it is mixed with water at a mass ratio of 2:1, and 50 kg of stearic acid is added for shear dispersion. The dispersed turbid liquid was calcined at 800° C. for 4 hours to obtain the cerium-zirconium-aluminum-based rare earth oxygen storage material.
其中,氧化铈15%,氧化锆28%,氧化铝50%,氧化镨3.5%,氧化镧3.5%。Among them, 15% of cerium oxide, 28% of zirconia, 50% of aluminum oxide, 3.5% of praseodymium oxide, and 3.5% of lanthanum oxide.
实施例3Example 3
将5kg大孔拟薄水铝石加入140kg水中,用硝酸调节至pH为2,经剪切分散、高能球磨得透明溶胶A;将2kg氧化镨、2kg氧化镧、125kg碳酸氧锆用硝酸溶解,加入110kg硝酸铈,2000kg水,得澄清透明溶液B;将溶液B和A混合,加热至80℃后,缓慢加入氨水,控制终点的pH为9,加入双氧水16kg,保温2h。经沉淀物离心脱水后与水按照质量比2∶1进行混合,加入月桂酸20kg,烯基琥珀酸酐30kg剪切分散。将分散后的浊液经600℃焙烧6h,得所述铈锆铝基稀土储氧材料。Add 5 kg of macroporous pseudo-boehmite into 140 kg of water, adjust the pH to 2 with nitric acid, and obtain transparent sol A through shear dispersion and high-energy ball milling; dissolve 2 kg of praseodymium oxide, 2 kg of lanthanum oxide, and 125 kg of zirconium oxycarbonate with nitric acid, Add 110kg of cerium nitrate and 2000kg of water to obtain clear and transparent solution B; mix solution B and A, heat to 80°C, slowly add ammonia water, control the pH at the end point to 9, add 16kg of hydrogen peroxide, and keep warm for 2 hours. After the sediment was centrifuged and dehydrated, it was mixed with water at a mass ratio of 2:1, and 20 kg of lauric acid and 30 kg of alkenyl succinic anhydride were added for shear dispersion. The dispersed turbid liquid was calcined at 600° C. for 6 hours to obtain the cerium-zirconium-aluminum-based rare earth oxygen storage material.
其中,氧化铈41%,氧化锆50%,氧化铝5%,氧化镨2%,氧化镧2%。Among them, 41% of cerium oxide, 50% of zirconia, 5% of aluminum oxide, 2% of praseodymium oxide, and 2% of lanthanum oxide.
实施例4Example 4
将26kg大孔拟薄水铝石将入到500kg水中,用硝酸调节至pH为2,经剪切分散、高能球磨得透明溶胶A;将5kg氧化镨、5kg氧化钕、30kg碳酸氧锆、133kg碳酸铈用硝酸溶解,加入2000kg水,得澄清透明溶液B;将溶液B和A混合,加热至80℃后,缓慢加入碳酸氢铵饱和溶液,控制终点的pH为6,加入双氧水22.3kg,保温2h。经沉淀物离心脱水后与水按照质量比2∶1进行混合,加入烯基琥珀酸酐35kg,剪切分散。将分散后的浊液经600℃焙烧5h,得所述铈锆铝基稀土储氧材料。Put 26kg of large-pore pseudo-boehmite into 500kg of water, adjust the pH to 2 with nitric acid, and obtain transparent sol A through shear dispersion and high-energy ball milling; 5kg of praseodymium oxide, 5kg of neodymium oxide, 30kg of zirconium Dissolve cerium carbonate with nitric acid, add 2000kg of water to obtain a clear and transparent solution B; mix solution B and A, heat to 80°C, slowly add saturated ammonium bicarbonate solution, control the pH of the end point to 6, add 22.3kg of hydrogen peroxide, and keep warm 2h. After the precipitate was centrifuged and dehydrated, it was mixed with water at a mass ratio of 2:1, and 35 kg of alkenyl succinic anhydride was added for shear dispersion. The dispersed turbid liquid was calcined at 600° C. for 5 hours to obtain the cerium-zirconium-aluminum-based rare earth oxygen storage material.
其中,氧化铈60%,氧化锆12%,氧化铝18%,氧化镨5%,氧化镧5%。Among them, 60% of cerium oxide, 12% of zirconia, 18% of aluminum oxide, 5% of praseodymium oxide, and 5% of lanthanum oxide.
以上显示和描述了本发明的基本原理、主要特征和本发明的优点。本行业的技术人员应该了解,本发明不受上述实施例的限制,上述实施例和说明书中描述的只是本发明的原理,在不脱离本发明精神和范围的前提下本发明还会有各种变化和改进,这些变化和改进都落入要求保护的本发明的范围内。本发明要求的保护范围由所附的权利要求书及其等同物界定。The basic principles, main features and advantages of the present invention have been shown and described above. Those skilled in the art should understand that the present invention is not limited by the above-mentioned embodiments. What are described in the above-mentioned embodiments and the description are only the principles of the present invention. Variations and improvements, which fall within the scope of the claimed invention. The scope of protection required by the present invention is defined by the appended claims and their equivalents.
Claims (8)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2011103464930A CN103084161A (en) | 2011-11-04 | 2011-11-04 | Ce-Zr-Al-based composite oxide rare earth oxygen-storage material and preparation method thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2011103464930A CN103084161A (en) | 2011-11-04 | 2011-11-04 | Ce-Zr-Al-based composite oxide rare earth oxygen-storage material and preparation method thereof |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN103084161A true CN103084161A (en) | 2013-05-08 |
Family
ID=48197590
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN2011103464930A Pending CN103084161A (en) | 2011-11-04 | 2011-11-04 | Ce-Zr-Al-based composite oxide rare earth oxygen-storage material and preparation method thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN103084161A (en) |
Cited By (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102824904A (en) * | 2012-09-20 | 2012-12-19 | 上海华明高纳稀土新材料有限公司 | Composite aluminium, cerium and zirconium oxide catalytic material and method for producing same |
| CN102962047A (en) * | 2012-11-12 | 2013-03-13 | 上海华明高纳稀土新材料有限公司 | Aluminum-cerium-zirconium based composite oxide catalytic material and preparation method thereof |
| CN103816886A (en) * | 2014-01-27 | 2014-05-28 | 江苏振邦尾气净化有限公司 | Method for preparing rare earth oxygen-storage material with large specific surface |
| CN103908959A (en) * | 2014-03-27 | 2014-07-09 | 台州欧信环保净化器有限公司 | Ce-Zr composite alumina oxide material and preparation method thereof |
| CN104437458A (en) * | 2014-11-12 | 2015-03-25 | 诺轩化学科技(上海)有限公司 | Cerium-zirconium-based composite oxide catalytic material and preparation method thereof |
| CN107107036A (en) * | 2014-11-06 | 2017-08-29 | 巴斯夫欧洲公司 | Mixed-metal oxides compound for storing up oxygen |
| CN109453823A (en) * | 2018-11-15 | 2019-03-12 | 新地能源工程技术有限公司 | A kind of cerium zirconium aluminum composite metal oxide and using it as the nickel-base catalyst of carrier |
| CN109529802A (en) * | 2018-11-12 | 2019-03-29 | 山东国瓷功能材料股份有限公司 | A kind of cerium zirconium compound oxide and its preparation method and application |
| CN109999804A (en) * | 2019-05-06 | 2019-07-12 | 昆明理工大学 | A kind of CO reduction SO2With catalyst and the preparation method and application thereof |
| CN110366445A (en) * | 2016-12-23 | 2019-10-22 | 罗地亚经营管理公司 | Age-resistant mixed oxides made of cerium, zirconium, aluminium and lanthanum for catalytic converters of motor vehicles |
| CN110721683A (en) * | 2019-10-29 | 2020-01-24 | 昆明理工大学 | Catalyst for liquid nitrogen tail gas washing oxidation under oxygen critical condition and preparation method and application thereof |
| CN110724460A (en) * | 2019-11-13 | 2020-01-24 | 刘通 | Preparation method of cerium-aluminum composite oxide polishing powder |
| CN113797915A (en) * | 2021-10-24 | 2021-12-17 | 合肥神舟催化净化器股份有限公司 | Diesel oxidation catalyst based on metal oxide nanoparticles, and preparation method and application thereof |
| CN114551910A (en) * | 2022-02-25 | 2022-05-27 | 内蒙古科技大学 | A kind of composite rare earth oxide and its preparation method and application |
| CN117899848A (en) * | 2023-12-28 | 2024-04-19 | 江门市科恒实业股份有限公司 | Oxygen storage material and preparation method thereof |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0706980A1 (en) * | 1994-10-05 | 1996-04-17 | Santoku Metal Industry Co., Ltd. | Composite oxide having oxygen absorbing and desorbing capability and method for preparing same |
| CN1695798A (en) * | 2005-03-30 | 2005-11-16 | 四川大学 | Cerium-zirconium-aluminum-based oxygen storage material and preparation method thereof |
| CN101745375A (en) * | 2008-12-15 | 2010-06-23 | 比亚迪股份有限公司 | Cerium zirconium aluminum based multiple oxide material and preparing method thereof |
| CN102000558A (en) * | 2010-10-27 | 2011-04-06 | 重庆海特汽车排气系统有限公司 | Composite oxide for automobile exhaust catalytic purification and preparation method thereof |
-
2011
- 2011-11-04 CN CN2011103464930A patent/CN103084161A/en active Pending
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0706980A1 (en) * | 1994-10-05 | 1996-04-17 | Santoku Metal Industry Co., Ltd. | Composite oxide having oxygen absorbing and desorbing capability and method for preparing same |
| CN1695798A (en) * | 2005-03-30 | 2005-11-16 | 四川大学 | Cerium-zirconium-aluminum-based oxygen storage material and preparation method thereof |
| CN101745375A (en) * | 2008-12-15 | 2010-06-23 | 比亚迪股份有限公司 | Cerium zirconium aluminum based multiple oxide material and preparing method thereof |
| CN102000558A (en) * | 2010-10-27 | 2011-04-06 | 重庆海特汽车排气系统有限公司 | Composite oxide for automobile exhaust catalytic purification and preparation method thereof |
Non-Patent Citations (1)
| Title |
|---|
| 焦毅,等: ""不同制备方法对CeO2-ZrO2-Al2O3材料性能的影响"", 《无机材料学报》, vol. 26, no. 8, 31 August 2011 (2011-08-31), pages 813 - 814 * |
Cited By (22)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102824904A (en) * | 2012-09-20 | 2012-12-19 | 上海华明高纳稀土新材料有限公司 | Composite aluminium, cerium and zirconium oxide catalytic material and method for producing same |
| CN102824904B (en) * | 2012-09-20 | 2015-09-02 | 上海华明高纳稀土新材料有限公司 | Aluminium cerium zirconium compound oxide catalysis material and preparation method thereof |
| CN102962047A (en) * | 2012-11-12 | 2013-03-13 | 上海华明高纳稀土新材料有限公司 | Aluminum-cerium-zirconium based composite oxide catalytic material and preparation method thereof |
| CN102962047B (en) * | 2012-11-12 | 2014-10-08 | 上海华明高纳稀土新材料有限公司 | Aluminum-cerium-zirconium based composite oxide catalytic material and preparation method thereof |
| CN103816886A (en) * | 2014-01-27 | 2014-05-28 | 江苏振邦尾气净化有限公司 | Method for preparing rare earth oxygen-storage material with large specific surface |
| CN103908959A (en) * | 2014-03-27 | 2014-07-09 | 台州欧信环保净化器有限公司 | Ce-Zr composite alumina oxide material and preparation method thereof |
| CN107107036A (en) * | 2014-11-06 | 2017-08-29 | 巴斯夫欧洲公司 | Mixed-metal oxides compound for storing up oxygen |
| CN104437458A (en) * | 2014-11-12 | 2015-03-25 | 诺轩化学科技(上海)有限公司 | Cerium-zirconium-based composite oxide catalytic material and preparation method thereof |
| CN104437458B (en) * | 2014-11-12 | 2017-05-17 | 诺轩化学科技(上海)有限公司 | Cerium-zirconium-based composite oxide catalytic material and preparation method thereof |
| CN110366445A (en) * | 2016-12-23 | 2019-10-22 | 罗地亚经营管理公司 | Age-resistant mixed oxides made of cerium, zirconium, aluminium and lanthanum for catalytic converters of motor vehicles |
| CN109529802A (en) * | 2018-11-12 | 2019-03-29 | 山东国瓷功能材料股份有限公司 | A kind of cerium zirconium compound oxide and its preparation method and application |
| CN109529802B (en) * | 2018-11-12 | 2021-12-28 | 山东国瓷功能材料股份有限公司 | Cerium-zirconium composite oxide and preparation method and application thereof |
| CN109453823A (en) * | 2018-11-15 | 2019-03-12 | 新地能源工程技术有限公司 | A kind of cerium zirconium aluminum composite metal oxide and using it as the nickel-base catalyst of carrier |
| CN109999804A (en) * | 2019-05-06 | 2019-07-12 | 昆明理工大学 | A kind of CO reduction SO2With catalyst and the preparation method and application thereof |
| CN109999804B (en) * | 2019-05-06 | 2020-07-17 | 昆明理工大学 | A kind of catalyst for CO reduction SO2 and its preparation method and application |
| CN110721683A (en) * | 2019-10-29 | 2020-01-24 | 昆明理工大学 | Catalyst for liquid nitrogen tail gas washing oxidation under oxygen critical condition and preparation method and application thereof |
| CN110724460A (en) * | 2019-11-13 | 2020-01-24 | 刘通 | Preparation method of cerium-aluminum composite oxide polishing powder |
| CN113797915A (en) * | 2021-10-24 | 2021-12-17 | 合肥神舟催化净化器股份有限公司 | Diesel oxidation catalyst based on metal oxide nanoparticles, and preparation method and application thereof |
| CN113797915B (en) * | 2021-10-24 | 2023-11-21 | 合肥神舟催化净化器股份有限公司 | Diesel oxidation catalyst based on metal oxide nano particles, preparation method and application |
| CN114551910A (en) * | 2022-02-25 | 2022-05-27 | 内蒙古科技大学 | A kind of composite rare earth oxide and its preparation method and application |
| CN114551910B (en) * | 2022-02-25 | 2023-09-22 | 内蒙古科技大学 | Composite rare earth oxide and preparation method and application thereof |
| CN117899848A (en) * | 2023-12-28 | 2024-04-19 | 江门市科恒实业股份有限公司 | Oxygen storage material and preparation method thereof |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN103084161A (en) | Ce-Zr-Al-based composite oxide rare earth oxygen-storage material and preparation method thereof | |
| CN102824904B (en) | Aluminium cerium zirconium compound oxide catalysis material and preparation method thereof | |
| CN102580719B (en) | A kind of nano cerium zirconium solid solution and preparation method thereof | |
| CN103127925B (en) | Cerium-zirconium-based solid solution rare earth oxygen-storage material and preparation method thereof | |
| CN101773830B (en) | High-temperature stability catalyst used for three-effect catalysis | |
| CN104492415B (en) | Preparation method of cerium-zirconium based solid solution catalytic material | |
| CN100360222C (en) | Cerium-zirconium-aluminum-based oxygen storage material and preparation method thereof | |
| CN101185887A (en) | Catalyst for wet oxidation process and preparation method thereof | |
| CN101249459A (en) | High-performance nanocomposite catalytic coating material for automobile exhaust purifier and preparation method thereof | |
| CN102989442A (en) | Heat-resistant modified alumina and preparation method thereof | |
| CN103908959A (en) | Ce-Zr composite alumina oxide material and preparation method thereof | |
| CN100563821C (en) | Low cerium type oxygen storage material and preparation method thereof | |
| CN102824918B (en) | Graphene oxide rare-earth composite methane catalyst | |
| CN103349978A (en) | Cerium-zirconium-aluminium-based composite oxide catalytic material and preparation method thereof | |
| CN102962047B (en) | Aluminum-cerium-zirconium based composite oxide catalytic material and preparation method thereof | |
| CN101940921A (en) | Bilayer structured oxygen storage material and preparation method thereof | |
| CN102847559A (en) | Cerium/aluminum-based composite oxide with high specific surface area and its preparation method | |
| US8357626B2 (en) | Oxygen storage/release material and exhaust gas purifying catalyst comprising the same | |
| CN104492414B (en) | Preparation method of cerium-zirconium based solid solution | |
| CN103406116B (en) | High heat resistance modified aluminium oxide and preparation method thereof | |
| CN105251485A (en) | Motor vehicle exhaust purification catalyst and production process thereof | |
| CN102962104A (en) | Composite carrier of exhaust gas cleaning catalyst and preparation method of carrier | |
| CN100493697C (en) | A spherical cerium-zirconium-based composite oxide and its preparation method | |
| EP2155365B1 (en) | Oxygen storage/release material and exhaust gas purifying catalyst comprising the same | |
| CN107349923B (en) | Mixed component material and preparation method thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C02 | Deemed withdrawal of patent application after publication (patent law 2001) | ||
| WD01 | Invention patent application deemed withdrawn after publication |
Application publication date: 20130508 |