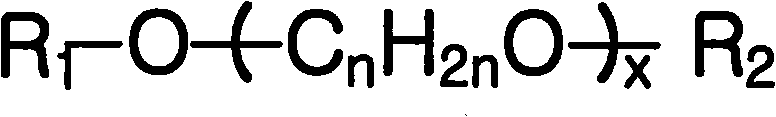CN102965214A - Cleaning composition - Google Patents
Cleaning composition Download PDFInfo
- Publication number
- CN102965214A CN102965214A CN201110270579XA CN201110270579A CN102965214A CN 102965214 A CN102965214 A CN 102965214A CN 201110270579X A CN201110270579X A CN 201110270579XA CN 201110270579 A CN201110270579 A CN 201110270579A CN 102965214 A CN102965214 A CN 102965214A
- Authority
- CN
- China
- Prior art keywords
- cleaning combination
- substrate
- cleaning
- ether
- ethylene glycol
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 238000004140 cleaning Methods 0.000 title claims abstract description 65
- 239000000203 mixture Substances 0.000 title claims abstract description 13
- 239000000758 substrate Substances 0.000 claims abstract description 56
- -1 polyol compound Chemical class 0.000 claims abstract description 19
- 150000001875 compounds Chemical class 0.000 claims abstract description 14
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims abstract description 14
- 239000000908 ammonium hydroxide Substances 0.000 claims abstract description 11
- 229920005862 polyol Polymers 0.000 claims abstract description 11
- 125000001453 quaternary ammonium group Chemical group 0.000 claims abstract description 11
- 229920000036 polyvinylpyrrolidone Polymers 0.000 claims abstract description 7
- 239000001267 polyvinylpyrrolidone Substances 0.000 claims abstract description 7
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 claims abstract description 7
- 239000004372 Polyvinyl alcohol Substances 0.000 claims abstract description 6
- 229920002451 polyvinyl alcohol Polymers 0.000 claims abstract description 6
- 229920002125 Sokalan® Polymers 0.000 claims abstract description 5
- 239000001913 cellulose Substances 0.000 claims abstract description 5
- 229920002678 cellulose Polymers 0.000 claims abstract description 5
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 claims description 47
- 238000000034 method Methods 0.000 claims description 16
- 239000003960 organic solvent Substances 0.000 claims description 14
- WGTYBPLFGIVFAS-UHFFFAOYSA-M tetramethylammonium hydroxide Chemical compound [OH-].C[N+](C)(C)C WGTYBPLFGIVFAS-UHFFFAOYSA-M 0.000 claims description 12
- RTZKZFJDLAIYFH-UHFFFAOYSA-N ether Substances CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 claims description 10
- 230000014509 gene expression Effects 0.000 claims description 7
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 claims description 6
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 claims description 6
- ZNQVEEAIQZEUHB-UHFFFAOYSA-N 2-ethoxyethanol Chemical compound CCOCCO ZNQVEEAIQZEUHB-UHFFFAOYSA-N 0.000 claims description 5
- PUPZLCDOIYMWBV-UHFFFAOYSA-N (+/-)-1,3-Butanediol Chemical compound CC(O)CCO PUPZLCDOIYMWBV-UHFFFAOYSA-N 0.000 claims description 4
- UXFQFBNBSPQBJW-UHFFFAOYSA-N 2-amino-2-methylpropane-1,3-diol Chemical compound OCC(N)(C)CO UXFQFBNBSPQBJW-UHFFFAOYSA-N 0.000 claims description 4
- UNXHWFMMPAWVPI-UHFFFAOYSA-N Erythritol Natural products OCC(O)C(O)CO UNXHWFMMPAWVPI-UHFFFAOYSA-N 0.000 claims description 4
- 229920003171 Poly (ethylene oxide) Polymers 0.000 claims description 4
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 claims description 4
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 claims description 4
- 239000004973 liquid crystal related substance Substances 0.000 claims description 4
- HEBKCHPVOIAQTA-UHFFFAOYSA-N meso ribitol Natural products OCC(O)C(O)C(O)CO HEBKCHPVOIAQTA-UHFFFAOYSA-N 0.000 claims description 4
- VDZOOKBUILJEDG-UHFFFAOYSA-M tetrabutylammonium hydroxide Chemical compound [OH-].CCCC[N+](CCCC)(CCCC)CCCC VDZOOKBUILJEDG-UHFFFAOYSA-M 0.000 claims description 4
- ARXJGSRGQADJSQ-UHFFFAOYSA-N 1-methoxypropan-2-ol Chemical group COCC(C)O ARXJGSRGQADJSQ-UHFFFAOYSA-N 0.000 claims description 3
- SBASXUCJHJRPEV-UHFFFAOYSA-N 2-(2-methoxyethoxy)ethanol Chemical compound COCCOCCO SBASXUCJHJRPEV-UHFFFAOYSA-N 0.000 claims description 3
- XNWFRZJHXBZDAG-UHFFFAOYSA-N 2-METHOXYETHANOL Chemical compound COCCO XNWFRZJHXBZDAG-UHFFFAOYSA-N 0.000 claims description 3
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 claims description 3
- 229930195725 Mannitol Natural products 0.000 claims description 3
- XXJWXESWEXIICW-UHFFFAOYSA-N diethylene glycol monoethyl ether Chemical compound CCOCCOCCO XXJWXESWEXIICW-UHFFFAOYSA-N 0.000 claims description 3
- 229940075557 diethylene glycol monoethyl ether Drugs 0.000 claims description 3
- 235000011187 glycerol Nutrition 0.000 claims description 3
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 3
- 239000000594 mannitol Substances 0.000 claims description 3
- 235000010355 mannitol Nutrition 0.000 claims description 3
- 229940073455 tetraethylammonium hydroxide Drugs 0.000 claims description 3
- LRGJRHZIDJQFCL-UHFFFAOYSA-M tetraethylazanium;hydroxide Chemical compound [OH-].CC[N+](CC)(CC)CC LRGJRHZIDJQFCL-UHFFFAOYSA-M 0.000 claims description 3
- JLGLQAWTXXGVEM-UHFFFAOYSA-N triethylene glycol monomethyl ether Chemical compound COCCOCCOCCO JLGLQAWTXXGVEM-UHFFFAOYSA-N 0.000 claims description 3
- MEIRRNXMZYDVDW-MQQKCMAXSA-N (2E,4E)-2,4-hexadien-1-ol Chemical compound C\C=C\C=C\CO MEIRRNXMZYDVDW-MQQKCMAXSA-N 0.000 claims description 2
- 229940058015 1,3-butylene glycol Drugs 0.000 claims description 2
- WFSMVVDJSNMRAR-UHFFFAOYSA-N 2-[2-(2-ethoxyethoxy)ethoxy]ethanol Chemical compound CCOCCOCCOCCO WFSMVVDJSNMRAR-UHFFFAOYSA-N 0.000 claims description 2
- AIDLAEPHWROGFI-UHFFFAOYSA-N 2-methylbenzene-1,3-dicarboxylic acid Chemical compound CC1=C(C(O)=O)C=CC=C1C(O)=O AIDLAEPHWROGFI-UHFFFAOYSA-N 0.000 claims description 2
- FBPFZTCFMRRESA-KAZBKCHUSA-N D-altritol Chemical compound OC[C@@H](O)[C@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KAZBKCHUSA-N 0.000 claims description 2
- UNXHWFMMPAWVPI-QWWZWVQMSA-N D-threitol Chemical compound OC[C@@H](O)[C@H](O)CO UNXHWFMMPAWVPI-QWWZWVQMSA-N 0.000 claims description 2
- 239000004386 Erythritol Substances 0.000 claims description 2
- ZJCCRDAZUWHFQH-UHFFFAOYSA-N Trimethylolpropane Chemical compound CCC(CO)(CO)CO ZJCCRDAZUWHFQH-UHFFFAOYSA-N 0.000 claims description 2
- TVXBFESIOXBWNM-UHFFFAOYSA-N Xylitol Natural products OCCC(O)C(O)C(O)CCO TVXBFESIOXBWNM-UHFFFAOYSA-N 0.000 claims description 2
- SRBFZHDQGSBBOR-QMKXCQHVSA-N alpha-L-arabinopyranose Chemical group O[C@H]1CO[C@@H](O)[C@H](O)[C@H]1O SRBFZHDQGSBBOR-QMKXCQHVSA-N 0.000 claims description 2
- BMRWNKZVCUKKSR-UHFFFAOYSA-N butane-1,2-diol Chemical compound CCC(O)CO BMRWNKZVCUKKSR-UHFFFAOYSA-N 0.000 claims description 2
- 235000019437 butane-1,3-diol Nutrition 0.000 claims description 2
- WERYXYBDKMZEQL-UHFFFAOYSA-N butane-1,4-diol Chemical compound OCCCCO WERYXYBDKMZEQL-UHFFFAOYSA-N 0.000 claims description 2
- OWBTYPJTUOEWEK-UHFFFAOYSA-N butane-2,3-diol Chemical compound CC(O)C(C)O OWBTYPJTUOEWEK-UHFFFAOYSA-N 0.000 claims description 2
- HEBKCHPVOIAQTA-NGQZWQHPSA-N d-xylitol Chemical compound OC[C@H](O)C(O)[C@H](O)CO HEBKCHPVOIAQTA-NGQZWQHPSA-N 0.000 claims description 2
- UNXHWFMMPAWVPI-ZXZARUISSA-N erythritol Chemical compound OC[C@H](O)[C@H](O)CO UNXHWFMMPAWVPI-ZXZARUISSA-N 0.000 claims description 2
- 235000019414 erythritol Nutrition 0.000 claims description 2
- 229940009714 erythritol Drugs 0.000 claims description 2
- 238000004519 manufacturing process Methods 0.000 claims description 2
- 229940059574 pentaerithrityl Drugs 0.000 claims description 2
- WXZMFSXDPGVJKK-UHFFFAOYSA-N pentaerythritol Chemical compound OCC(CO)(CO)CO WXZMFSXDPGVJKK-UHFFFAOYSA-N 0.000 claims description 2
- LPSKDVINWQNWFE-UHFFFAOYSA-M tetrapropylazanium;hydroxide Chemical compound [OH-].CCC[N+](CCC)(CCC)CCC LPSKDVINWQNWFE-UHFFFAOYSA-M 0.000 claims description 2
- QXJQHYBHAIHNGG-UHFFFAOYSA-N trimethylolethane Chemical compound OCC(C)(CO)CO QXJQHYBHAIHNGG-UHFFFAOYSA-N 0.000 claims description 2
- 239000000811 xylitol Substances 0.000 claims description 2
- HEBKCHPVOIAQTA-SCDXWVJYSA-N xylitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)CO HEBKCHPVOIAQTA-SCDXWVJYSA-N 0.000 claims description 2
- 235000010447 xylitol Nutrition 0.000 claims description 2
- 229960002675 xylitol Drugs 0.000 claims description 2
- 239000004584 polyacrylic acid Substances 0.000 claims 1
- MTHSVFCYNBDYFN-UHFFFAOYSA-N diethylene glycol Chemical compound OCCOCCO MTHSVFCYNBDYFN-UHFFFAOYSA-N 0.000 abstract description 3
- 239000002202 Polyethylene glycol Substances 0.000 abstract 2
- 229920001223 polyethylene glycol Polymers 0.000 abstract 2
- 229920006187 aquazol Polymers 0.000 abstract 1
- 239000004210 ether based solvent Substances 0.000 abstract 1
- 239000011521 glass Substances 0.000 description 13
- 239000011248 coating agent Substances 0.000 description 12
- 238000000576 coating method Methods 0.000 description 12
- 239000003921 oil Substances 0.000 description 10
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 9
- 229910052802 copper Inorganic materials 0.000 description 9
- 239000010949 copper Substances 0.000 description 9
- 239000002957 persistent organic pollutant Substances 0.000 description 9
- 229920001721 polyimide Polymers 0.000 description 8
- 239000000976 ink Substances 0.000 description 7
- 238000009736 wetting Methods 0.000 description 7
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 6
- 239000004642 Polyimide Substances 0.000 description 6
- 239000012528 membrane Substances 0.000 description 6
- 239000002245 particle Substances 0.000 description 6
- 238000005406 washing Methods 0.000 description 6
- 239000000809 air pollutant Substances 0.000 description 5
- 231100001243 air pollutant Toxicity 0.000 description 5
- 230000000694 effects Effects 0.000 description 5
- 229910052751 metal Inorganic materials 0.000 description 5
- 239000002184 metal Substances 0.000 description 5
- 229910000881 Cu alloy Inorganic materials 0.000 description 4
- 238000002438 flame photometric detection Methods 0.000 description 4
- 239000007921 spray Substances 0.000 description 4
- 229910000838 Al alloy Inorganic materials 0.000 description 3
- 229910052581 Si3N4 Inorganic materials 0.000 description 3
- 239000004411 aluminium Substances 0.000 description 3
- 229910052782 aluminium Inorganic materials 0.000 description 3
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 3
- 238000001035 drying Methods 0.000 description 3
- 229910052757 nitrogen Inorganic materials 0.000 description 3
- 230000008569 process Effects 0.000 description 3
- HQVNEWCFYHHQES-UHFFFAOYSA-N silicon nitride Chemical compound N12[Si]34N5[Si]62N3[Si]51N64 HQVNEWCFYHHQES-UHFFFAOYSA-N 0.000 description 3
- 239000000243 solution Substances 0.000 description 3
- 229910021642 ultra pure water Inorganic materials 0.000 description 3
- 239000012498 ultrapure water Substances 0.000 description 3
- 229910004205 SiNX Inorganic materials 0.000 description 2
- 239000008367 deionised water Substances 0.000 description 2
- 229910021641 deionized water Inorganic materials 0.000 description 2
- 238000010586 diagram Methods 0.000 description 2
- 239000007888 film coating Substances 0.000 description 2
- 238000009501 film coating Methods 0.000 description 2
- 230000005660 hydrophilic surface Effects 0.000 description 2
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 2
- 238000002156 mixing Methods 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 238000002360 preparation method Methods 0.000 description 2
- KRHYYFGTRYWZRS-UHFFFAOYSA-M Fluoride anion Chemical compound [F-] KRHYYFGTRYWZRS-UHFFFAOYSA-M 0.000 description 1
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 1
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 1
- 125000000217 alkyl group Chemical group 0.000 description 1
- 150000001412 amines Chemical group 0.000 description 1
- 150000003868 ammonium compounds Chemical class 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 229910052799 carbon Inorganic materials 0.000 description 1
- 125000004432 carbon atom Chemical group C* 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 229940125904 compound 1 Drugs 0.000 description 1
- 229940126214 compound 3 Drugs 0.000 description 1
- 238000005260 corrosion Methods 0.000 description 1
- 230000007797 corrosion Effects 0.000 description 1
- 230000008021 deposition Effects 0.000 description 1
- 238000004090 dissolution Methods 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 230000007613 environmental effect Effects 0.000 description 1
- 238000005530 etching Methods 0.000 description 1
- 230000002349 favourable effect Effects 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 150000002500 ions Chemical class 0.000 description 1
- 229910021645 metal ion Inorganic materials 0.000 description 1
- 150000002894 organic compounds Chemical class 0.000 description 1
- 239000011146 organic particle Substances 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 230000002940 repellent Effects 0.000 description 1
- 239000005871 repellent Substances 0.000 description 1
- 239000004065 semiconductor Substances 0.000 description 1
- 229910052710 silicon Inorganic materials 0.000 description 1
- 239000010703 silicon Substances 0.000 description 1
- 238000001179 sorption measurement Methods 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 229910000597 tin-copper alloy Inorganic materials 0.000 description 1
- ZMANZCXQSJIPKH-UHFFFAOYSA-O triethylammonium ion Chemical compound CC[NH+](CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-O 0.000 description 1
Images
Landscapes
- Detergent Compositions (AREA)
Abstract
The invention discloses a cleaning composition which can endow the surface of a substrate with hydrophilicity. The composition includes: 0.0001-5wt% of at least one water-soluble high molecular compound selected from the group composed of polyvinyl alcohol, polyethylene glycol (PEG), cellulose, polyacrylic acid (PAA), polyethyloxazoline and polyvinylpyrrolidone; 0.01-5wt% of a polyol compound; 0.05-10wt% of quaternary ammonium hydroxide; 0.05-40wt% of a glycol ether-based solvent expressed by the following molecular formula; and the balance water.
Description
Technical field
The present invention relates to a kind of cleaning combination, more specifically, relate to a kind of for such as liquid-crystal display, plasma display, the cleaning combination of the flat-panel monitor of flexible display and similar indicating meter (being referred to as hereinafter, " FPDs ").
Background technology
FPDs, its representative instance are liquid-crystal display (LCDs), be to form by film, and exposure, the same technique of etching and the like and semiconductor device is made.But in these techniques, the various organic or inorganic particulate with 1 μ m or following size is attached to the surface of substrate, thereby causes the pollution of substrate.When adhering to particle on it and carry out subsequently technique, form pin hole or hole at film, and rupture of line or bridge joint, thereby reduce product yield.Therefore, in order to remove these particles, implement cleaning, thereby develop various clean-out systems.
The ability of removing the organic or inorganic particulate of these clean-out systems must be excellent, and they must not corrode substrate or made by aluminium, aluminium alloy, copper, copper alloy or analogue and be formed at metal wire on the substrate.In addition, in order easily to implement technique subsequently, need these clean-out systems to improve the coating performance that cleans metacoxa.
Korean patent registration No. 10-0305314 discloses and has a kind ofly comprised the alkaline aqueous solution of metal ion not and have 2 to 10 hydroxyl (clean-out systems of poly-hydroxy group OH).But, because this clean-out system does not comprise the organic solvent such as glycol ether, so the problem that exists is it does not have enough abilities of removing such as the organic pollutant of oil and analogue thereof, and it can not fully demonstrate and gives substrate surface hydrophilic effect after cleaning.
In addition, Korean patent registration No. 10-073052 discloses a kind of glycol ether that comprises, the clean-out system of water-miscible organic solvent and ammonium compound.Yet, this clean-out system can be favourable by effectively using removing the photo-resist process aspect at it, but do not give aspect the substrate surface wetting ability and a large amount of organic solvent residuals is problematic aspect on substrate at it, thereby increased the surface contact angle of substrate.
In addition, Korean patent registration No. 10-0522845 discloses a kind of clean-out system, comprising: at least a dissolution accelerator that is selected from amine and fluorochemical; With water-soluble high-molecular compound such as polyvinylpyrrolidone.But this clean-out system also has problem, the fluoride corrosion that silicon-based glass substrate or silicon nitride film are added, and metal such as copper also are corroded.
Summary of the invention
Therefore, found out the present invention to address the above problem, the purpose of this invention is to provide a kind of clean-out system, it has removes pollution glass substrate or the organic pollutant of metallic membrane or the excellent ability of particle in making FPD substrate process process, it has and prevents the excellent ability that is corroded by the metal wire that aluminium, aluminium alloy, copper, copper alloy or analogue are made, and it has and gives the substrate surface wetting ability to improve the excellent ability of the coating performance that cleans metacoxa.
In order to finish above purpose, one aspect of the present invention provides a kind of cleaning combination that can give the surface hydrophilicity of substrate, comprise: based on total composition, 0.0001~5wt% is selected from least a water-soluble high-molecular compound in the group that is comprised of polyvinyl alcohol, polyoxyethylene glycol (PEG), Mierocrystalline cellulose, polyacrylic acid (PAA), PEOz and polyvinylpyrrolidone; 0.01 the polyol compound of~5wt%; 0.05 the quaternary ammonium hydroxide of~10wt%; 0.05 the ethylene glycol ether organic solvent by following molecular formula 1 expression of~40wt%; With remaining water.
Molecular formula 1
R wherein
1Methyl or ethyl, R
2Be hydrogen atom, n is 2 to 5 integer, and x is 1 to 5 integer.
Another aspect of the present invention provides a kind of manufacturing to be used for the method for the array substrate of liquid-crystal display, comprises the step of using described cleaning combination cleaning base plate.
Description of drawings
To more clearly understand above and other purpose of the present invention by following detailed description by reference to the accompanying drawings, feature and advantage, wherein:
Fig. 1 has shown to use the cleaning combination of embodiment according to the present invention 16 to remove the result's of oil pen's pollutent photo from substrate;
Fig. 2 has shown to be used for assessing organic membrane to the ink-jet printer synoptic diagram of the coating ability of the substrate that cleaned by cleaning combination of the present invention;
Fig. 3 has shown that the assessment organic membrane is to the synoptic diagram of the method for the coating ability of the substrate that cleaned by cleaning combination of the present invention.
Embodiment
Hereinafter, explain with reference to the accompanying drawings preferred embodiment of the present invention.
The invention provides a kind of cleaning combination that can give the surface hydrophilicity of substrate, comprise: based on total composition, (a) 0.0001~5wt%'s is selected from least a water-soluble high-molecular compound in the group that is comprised of polyvinyl alcohol, polyoxyethylene glycol (PEG), Mierocrystalline cellulose, polyacrylic acid (PAA), PEOz and polyvinylpyrrolidone; (b) polyol compound of 0.01~5wt%; (c) quaternary ammonium hydroxide of 0.05~10wt%; (d) the ethylene glycol ether organic solvent by following molecular formula 1 expression of 0.05~40wt%; (e) remaining water.
Molecular formula 1
R wherein
1Methyl or ethyl, R
2Being that hydrogen is former gives, and n is 2 to 5 integer, and x is 1 to 5 integer.
Cleaning combination of the present invention is characterised in that it has the excellent ability of cleaning base plate, with since it comprise an amount of above water-soluble high-molecular compound, so it has the substrate surface of giving wetting ability, thus the excellent ability of the coating performance of the substrate after raising is cleaned.
Hereinafter, describe the composition of cleaning combination of the present invention in detail.
(a) water-soluble high-molecular compound
Cleaning combination of the present invention comprises at least a water-soluble high-molecular compound that is selected from the group that is comprised of polyvinyl alcohol, polyoxyethylene glycol (PEG), Mierocrystalline cellulose, polyacrylic acid (PAA), PEOz and polyvinylpyrrolidone.
Water-soluble high-molecular compound has high water-soluble, with insulating film, copper film, the water repellent surface improvement of tin-copper alloy film WU or similar film is hydrophilic surface, in technique subsequently, advantageously to utilize its hydrophilic surface, such as the photo-resist coating, use organic coating of polyimide and analogue thereof, metal film deposition, and similar technique.
Based on total composition, the amount of the water-soluble high-molecular compound that comprises is preferred, 0.0001~5wt%, more preferably 0.001~2wt%.When the amount of water-soluble high-molecular compound was less than 0.0001wt%, the problem of existence was that the hydrophilic ability of substrate surface of giving of cleaning combination significantly reduces.And, when its amount is higher than 5wt%, the problem that exists is that the viscosity of cleaning combination increases, therefore produce too much bubble, workability is worsened, with the surface adsorption increase of cleaning combination, so cleaning combination remains in the surface of substrate, thereby causes that water-soluble high-molecular compound remains in the substrate in cleaning.
(b) polyol compound
In cleaning combination of the present invention, polyol compound plays by increasing the wetting capacity of substrate surface, improve cleaning combination and remove the effect of the ability (cleansing power) of oiliness composition from substrate surface, and play deliquescent effect in the raising cleaning combination water.
Based on the total amount of composition, the amount of the polyol compound that comprises is 0.01~5wt%, preferred 0.1~3wt%.When the amount of polyol compound was less than 0.01wt%, it is not enough that the moistening ability of glass baseplate surface of giving of cleaning combination becomes.When its amount surpassed 5wt%, the effect of giving wetting capacity no longer increased, and it is inefficient.
The example of polyol compound can comprise ethylene glycol, propylene glycol, 1, ammediol, 1,2-butyleneglycol, 1,3-butyleneglycol, 2,3-butyleneglycol, 1,4-butyleneglycol, 2-methyl isophthalic acid, ammediol, glycerine, trimethylolethane, TriMethylolPropane(TMP), tetramethylolmethane, Xylitol, N.F,USP MANNITOL, sorbyl alcohol, erythritol, Pentitol, threitol, the pure and mild talitol of pectinose.These examples can use separately or two kinds or above being combined with.
(c) quaternary ammonium hydroxide
Cleaning combination of the present invention comprises the quaternary ammonium hydroxide by following molecular formula 2 expressions:
Molecular formula 2
[N-(R
3)
4]
+·OH
-
R wherein
3That carbon atom is 1 to 5 alkyl.
Because quaternary ammonium hydroxide has high alkalescence, it is converted into low molecular compound and increases separability by the organic compound with polymer organic pollutant or oil-containing, plays a part to improve cleansing power.
Based on the total amount of composition, the amount of the quaternary ammonium hydroxide that comprises is 0.05~10wt%, preferred 0.1~5wt%.When the amount of quaternary ammonium hydroxide is less than 0.05wt%, be difficult for removing the particle or the organic pollutant that are attached on the substrate.When its amount surpassed 10wt%, cleaning performance no longer increased, and was uneconomic therefore, and caused environmental problem.
The example of quaternary ammonium hydroxide can comprise tetramethylammonium hydroxide, tetraethylammonium hydroxide, hydroxide tetrapropylammonium, and tetrabutylammonium hydroxide.Wherein, preferably use tetramethylammonium hydroxide (being referred to as hereinafter, " TMAH ").These examples can be separately or two kinds or above being combined with.
(d) ethylene glycol ether organic solvent
Cleaning combination of the present invention comprises the ethylene glycol ether organic solvent by following molecular formula 1 expression.
Molecular formula 1
R wherein
1Methyl or ethyl, R
2Be hydrogen atom, n is 2 to 5 integer, and x is 1 to 5 integer.
Example by the ethylene glycol ether organic solvent of above molecular formula 1 expression can comprise ethylene glycol monomethyl ether (MG), diethylene glycol monomethyl ether (MDG), diethylene glycol monoethyl ether (Trivalin SF), ethylene glycol monoethyl ether (EG), Triethylene glycol ethyl ether (ETG), triethylene glycol monomethyl ether (MTG), DPE (MFDG), propylene glycol monomethyl ether (MFG).These examples can be separately or two kinds or above being combined with.
Based on the total amount of composition, the amount of the ethylene glycol ether organic solvent that comprises is 0.05~40wt%, preferred 0.5~20wt%.When the amount of ethylene glycol ether organic solvent is less than 0.05wt%, can not expect to increase the solvability of organic pollutant in organic solvent.When its amount surpassed 40wt%, the effect that increases owing to wetting capacity no longer increased.
(e) water
The water that uses among the present invention can be deionized water, but is not particularly limited.More preferably, the deionized water with 18M Ω/cm or higher ratio resistance can be used as water and uses, and the ratio resistance of water represents the degree that ion has been removed from water.
Use the technique of cleaning combination cleaning base plate of the present invention to implement by usually known method.Usually the example of known method can comprise spray, and rotation is soaked, and ultrasonic wave is soaked and similar approach.Cleaning combination of the present invention is at 20~80 ℃, and preferred 20~50 ℃ temperature has shown the most excellent cleaning performance, uses cleaning combination of the present invention, and implementing cleaning base plate technique was preferred in 30 seconds~10 minutes.
Hereinafter, illustrate in greater detail the present invention according to following example.But following example just is used for explaining of the present invention, and scope of the present invention is not limited.Following example can be within the scope of the invention by those skilled in the art's change and modification.
Example 1 to 16 and comparison example 1 to 5: the preparation of cleaning combination
Cleaning combination is by mixing the component that provides in the following table 1 with corresponding ratio of components in being equipped with the mixing section of agitator, and then at room temperature stir the mixture with the speed of rotation of 500rpm prepared in 1 hour.
Table 1
Explain
1): polyvinyl alcohol
2): polyvinylpyrrolidone
3): PEOz
A): ethylene glycol
B): glycerine
C): N.F,USP MANNITOL
TMAH: tetramethylammonium hydroxide
TEAH: tetraethylammonium hydroxide
EG: ethylene glycol monoethyl ether
MG: ethylene glycol monomethyl ether
EDG: diethylene glycol monoethyl ether
MDG: diethylene glycol monomethyl ether
MTG: triethylene glycol monomethyl ether
Test examples: the assessment of removing particle and organic pollutant ability
For the ability of removing particle and organic pollutant of the cleaning combination of assessing preparation, carried out following test.
1)
Remove the assessment of air pollutant ability
Example 1 to 16 and comparison example 1 to 5 each prepared cleaning combination 100ml are put into the beaker of a 250ml.In order to assess the ability of removing air pollutant, glass substrate with each 5cmx5cm size, insulating film (SiNx: silicon nitride) substrate and copper base, in air, placed 3 days, so that it is contaminated, at room temperature contaminated substrate was washed 1 minute with cleaning combination respectively with Spray-cleaning Machine.Afterwards, further with ultrapure water washing washed substrate 30 seconds originally, then use nitrogen drying.
The ability of removing air pollutant of each cleaning combination be by the washing before and after contact angle reduce assess, its result provides in following table 2.
2) remove the assessment of fingerprint pollution thing ability
In order to assess the ability of removing the fingerprint pollution thing, pollute with the glass substrate of fingerprint with the 2cmx5cm size, at room temperature contaminated glass substrate was washed 1 minute with cleaning combination with Spray-cleaning Machine.Afterwards, further with ultrapure water washing washed glass substrate 30 seconds originally, then use nitrogen drying.
The situation that the fingerprint pollution thing is removed represents that with zero the situation that the fingerprint pollution thing is not removed represents with X.Its result provides in following table 2.
Table 2
Explain)
Remove the assessment of air pollutant ability from glass substrate
◎: excellent (reducing 30 ° or larger)
Zero: good (reduce 20 or larger~be less than 30 °)
△: not enough (reduce 5 or larger~be less than 20 °)
X: poor (minimizing is less than 5 °)
Clump insulating film substrate and copper base are removed the assessment of air pollutant ability
◎: excellent (reducing 15 ° or larger)
Zero: good (reduce 10 or larger~be less than 15 °)
△: not enough (reduce 0 or larger~be less than 10 °)
X: poor (minimizing is less than 0 °)
From the test-results that upper table 2 provides, can determine that all cleaning combinations of example 1 to 16 have shown the excellent ability of removing pollutent.Yet can determine that in the cleaning combination of comparison example 1 to 5 each does not comprise water-soluble high-molecular compound, polyol compound, any in basic cpd and the ethylene glycol ether organic solvent shown low-down ability of removing pollutent.
3) remove the assessment of oil pen's pollutent ability
For assess remove organic pollutant ability, the glass substrate of a 2cmx5cm size is polluted with the oil pen, with Spray-cleaning Machine at room temperature with the glass substrate that pollutes with example 3,4,9,12,14 and 16, and each cleaning combination of comparison example 1 and 3 washed 1 minute.Afterwards, then the glass substrate that further originally is washed with ultrapure water washing 30 seconds uses nitrogen drying.
Situation about being removed when oil pen's pollutent represents that with zero situation about not being removed when oil pen's pollutent represents with X.Its result provides in following table 3.And the result of the ability of removing oil pen's pollutent of the cleaning combination of example 16 is illustrated among Fig. 1.
4) assessment of polyimide solution coating performance
For organic membrane after being evaluated at the washing substrate to the coating performance of substrate surface, glass substrate with each 5cmx5cm size, (SiNx: silicon nitride) substrate and copper base are at room temperature used respectively example 3 to insulating film substrate, 4,9,12 and 14 and the cleaning combination of comparison example 1 and 3 use the Spray-cleaning Machine washing.
As shown in Figure 2, use the coating performance of spray ink Printing machine and polyimide China ink assessment organic membrane.Namely use the spray ink Printing machine with the polyimide ink droplet to substrate 10, this spray ink Printing machine is equipped with the stamp pad 11 of having placed substrate 10 on it, comprise most nozzles 13 and be used for ink-jet printer 14 to the ink supply parts 15 of ink-jet printer 14 ink supply, form thus drop 12.The ability of utilizing ink-jet printer 14 usefulness polyimide solutions to be applied to substrate is to drip to the diameter 16 (referring to Fig. 3) that 10 skins on the substrate 10 rise drop with regular interval and assess by measuring.In the assessment of polyimide solution to the coating performance of substrate 10, when surpassing the situation of 100 μ m, the diameter of the drop on dripping to substrate 10 represents (well) with zero, when its diameter is the situation of 100~80 μ m, represent (generally) with △, and represent (poor) with X during less than the situation of 80 μ m when its diameter.
Table 3
From the test-results that upper table 3 provides, can determine example 3,4, all cleaning combinations of 9,12 and 14 have shown excellent ability and the excellent polyimide film coating performance of removing oil pen's pollutent.Yet, can determine the cleaning combination of each comparison example that does not comprise water-soluble high-molecular compound 1 and 3, shown and removed the poor ability of oil pen's pollutent and poor polyimide film coating performance.
As mentioned above, cleaning combination of the present invention has from being used for the glass substrate of FPDs, and insulating film or by copper is removed the excellent ability of organic pollutant or particle on the metallic membrane that copper alloy or analogue are made.And this cleaning combination has and prevents by aluminium, aluminium alloy, and copper, that copper alloy or analogue are made and be formed at the excellent ability that the metal wire on the substrate is corroded.And this cleaning combination has the substrate surface of giving wetting ability, thereby improves the excellent ability of the coating performance of the substrate after cleaning.In addition, this cleaning combination is owing to comprise a large amount of water, so can easily be processed and environmentally friendly.
Although disclose for illustrative purposes preferred embodiment of the present invention, it will be understood by those skilled in the art that not breaking away from such as appended claim under the disclosed scope and spirit of the present invention, carry out various modifications, add, and replacement is possible.
Claims (5)
1. the cleaning combination that can give the surface hydrophilicity of substrate, comprise, total amount based on described composition: 0.0001~5wt% is selected from by polyvinyl alcohol, polyoxyethylene glycol, Mierocrystalline cellulose, polyacrylic acid, at least a water-soluble high-molecular compound in the group that PEOz and polyvinylpyrrolidone form;
0.01 the polyol compound of~5wt%;
0.05 the quaternary ammonium hydroxide of~10wt%;
0.05 the ethylene glycol ether organic solvent by following molecular formula 1 expression of~40wt%; With
Remaining water:
[molecular formula 1]
R wherein
1Methyl or ethyl, R
2Be hydrogen atom, n is 2 to 5 integer, and x is 1 to 5 integer.
2. cleaning combination according to claim 1, wherein said polyol compound comprises and being selected from by ethylene glycol, propylene glycol, 1, ammediol, 1,2-butyleneglycol, 1,3-butyleneglycol, 2,3-butyleneglycol, BDO, 2-methyl isophthalic acid, at least a in the group that the pure and mild talitol of ammediol, glycerine, trimethylolethane, TriMethylolPropane(TMP), tetramethylolmethane, Xylitol, N.F,USP MANNITOL, sorbyl alcohol, erythritol, Pentitol, threitol, pectinose forms.
3. cleaning combination according to claim 1, wherein said quaternary ammonium hydroxide comprise and being selected from by tetramethylammonium hydroxide, tetraethylammonium hydroxide, hydroxide tetrapropylammonium, and at least a in the group that forms of tetrabutylammonium hydroxide.
4. cleaning combination according to claim 1, wherein said ethylene glycol ether organic solvent by 1 expression of above molecular formula comprises and being selected from by ethylene glycol monomethyl ether, diethylene glycol monomethyl ether, diethylene glycol monoethyl ether, ethylene glycol monoethyl ether, Triethylene glycol ethyl ether, triethylene glycol monomethyl ether, at least a in the group that DPE, propylene glycol monomethyl ether form.
5. a manufacturing is used for the method for the array substrate of liquid-crystal display, comprises the step that right to use requires any one the described cleaning combination cleaning base plate in 1 to 4.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201110270579XA CN102965214A (en) | 2011-08-31 | 2011-08-31 | Cleaning composition |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201110270579XA CN102965214A (en) | 2011-08-31 | 2011-08-31 | Cleaning composition |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN102965214A true CN102965214A (en) | 2013-03-13 |
Family
ID=47795650
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201110270579XA Pending CN102965214A (en) | 2011-08-31 | 2011-08-31 | Cleaning composition |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN102965214A (en) |
Cited By (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN103160385A (en) * | 2013-04-03 | 2013-06-19 | 济南瑞东实业有限公司 | Full cleaning agent for oil refining device and preparation method of full cleaning agent |
| WO2016086012A1 (en) | 2014-11-26 | 2016-06-02 | Microban Products Company | Surface disinfectant with residual biocidal property |
| CN106701352A (en) * | 2016-12-02 | 2017-05-24 | 安徽长庚光学科技有限公司 | Environment-friendly type efficient lens cleanser and preparation method thereof |
| CN108008862A (en) * | 2017-12-19 | 2018-05-08 | 上海天马微电子有限公司 | Touch film layer, touch panel and touch display device thereof |
| CN108102807A (en) * | 2017-12-21 | 2018-06-01 | 广东德新科技孵化器有限公司 | A kind of wood furniture surface cleaning renovation agent and preparation method thereof |
| CN108653098A (en) * | 2018-07-11 | 2018-10-16 | 中山市智联企业孵化器发展有限公司 | A kind of plant body wash and preparation method thereof |
| CN108697078A (en) * | 2015-11-23 | 2018-10-23 | 美可帮产品公司 | Surface disinfection agent with remaining biocidal properties |
| CN108697082A (en) * | 2015-11-23 | 2018-10-23 | 美可帮产品公司 | Surface disinfection agent with remaining biocidal properties |
| CN109310696A (en) * | 2016-05-23 | 2019-02-05 | 美可帮产品公司 | Touchscreen cleaning and protection compositions |
| CN110868858A (en) * | 2017-06-16 | 2020-03-06 | W·M·巴尔公司 | Surface disinfectants with residual biocidal properties |
| US10842147B2 (en) * | 2014-11-26 | 2020-11-24 | Microban Products Company | Surface disinfectant with residual biocidal property |
| US10925281B2 (en) | 2014-11-26 | 2021-02-23 | Microban Products Company | Surface disinfectant with residual biocidal property |
| US11033023B2 (en) | 2014-11-26 | 2021-06-15 | Microban Products Company | Surface disinfectant with residual biocidal property |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1683487A (en) * | 2004-01-23 | 2005-10-19 | 东京応化工业株式会社 | Stripping cleaning solution, semiconductor substrate cleaning method, and metal wiring forming method |
| CN1849386A (en) * | 2003-06-18 | 2006-10-18 | 东京応化工业株式会社 | Cleaning composition, method of cleaning semiconductor substrate, and method of forming wiring on semiconductor substrate |
-
2011
- 2011-08-31 CN CN201110270579XA patent/CN102965214A/en active Pending
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1849386A (en) * | 2003-06-18 | 2006-10-18 | 东京応化工业株式会社 | Cleaning composition, method of cleaning semiconductor substrate, and method of forming wiring on semiconductor substrate |
| CN1683487A (en) * | 2004-01-23 | 2005-10-19 | 东京応化工业株式会社 | Stripping cleaning solution, semiconductor substrate cleaning method, and metal wiring forming method |
Cited By (28)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN103160385A (en) * | 2013-04-03 | 2013-06-19 | 济南瑞东实业有限公司 | Full cleaning agent for oil refining device and preparation method of full cleaning agent |
| CN103160385B (en) * | 2013-04-03 | 2014-10-08 | 济南瑞东实业有限公司 | Full cleaning agent for oil refining device and preparation method of full cleaning agent |
| EP3223612A4 (en) * | 2014-11-26 | 2018-09-26 | Microban Products Company | Surface disinfectant with residual biocidal property |
| US10834922B2 (en) | 2014-11-26 | 2020-11-17 | Microban Products Company | Surface disinfectant with residual biocidal property |
| US11026418B2 (en) | 2014-11-26 | 2021-06-08 | Microban Products Company | Surface disinfectant with residual biocidal property |
| CN107205383A (en) * | 2014-11-26 | 2017-09-26 | 美可帮产品公司 | Surface disinfectants with residual biocidal properties |
| CN107231806A (en) * | 2014-11-26 | 2017-10-03 | 美可帮产品公司 | Surface disinfectant with remaining biocidal properties |
| WO2016086012A1 (en) | 2014-11-26 | 2016-06-02 | Microban Products Company | Surface disinfectant with residual biocidal property |
| EP3885394A3 (en) * | 2014-11-26 | 2021-12-08 | Microban Products Company | Surface disinfectant with residual biocidal property |
| EP3224300A4 (en) * | 2014-11-26 | 2018-08-29 | Microban Products Company | Surface disinfectant with residual biocidal property |
| US11033023B2 (en) | 2014-11-26 | 2021-06-15 | Microban Products Company | Surface disinfectant with residual biocidal property |
| US11134678B2 (en) | 2014-11-26 | 2021-10-05 | Microban Products Company | Surface disinfectant with residual biocidal property |
| US10925281B2 (en) | 2014-11-26 | 2021-02-23 | Microban Products Company | Surface disinfectant with residual biocidal property |
| US10842147B2 (en) * | 2014-11-26 | 2020-11-24 | Microban Products Company | Surface disinfectant with residual biocidal property |
| WO2016086014A1 (en) | 2014-11-26 | 2016-06-02 | Microban Products Company | Surface disinfectant with residual biocidal property |
| US11134674B2 (en) | 2014-11-26 | 2021-10-05 | Microban Products Company | Surface disinfectant with residual biocidal property |
| EP3379931A4 (en) * | 2015-11-23 | 2019-06-05 | Microban Products Company | SURFACE DISINFECTANT HAVING RESIDUAL BIOCIDAL PROPERTY |
| EP3379932A4 (en) * | 2015-11-23 | 2019-05-01 | Microban Products Company | SURFACE DISINFECTANT HAVING RESIDUAL BIOCIDAL PROPERTY |
| CN108697082A (en) * | 2015-11-23 | 2018-10-23 | 美可帮产品公司 | Surface disinfection agent with remaining biocidal properties |
| CN108697078A (en) * | 2015-11-23 | 2018-10-23 | 美可帮产品公司 | Surface disinfection agent with remaining biocidal properties |
| CN109310696A (en) * | 2016-05-23 | 2019-02-05 | 美可帮产品公司 | Touchscreen cleaning and protection compositions |
| US11503824B2 (en) | 2016-05-23 | 2022-11-22 | Microban Products Company | Touch screen cleaning and protectant composition |
| CN106701352A (en) * | 2016-12-02 | 2017-05-24 | 安徽长庚光学科技有限公司 | Environment-friendly type efficient lens cleanser and preparation method thereof |
| EP3638027A4 (en) * | 2017-06-16 | 2021-03-17 | W.M. Barr & Company Inc. | Surface disinfectant with residual biocidal property |
| CN110868858A (en) * | 2017-06-16 | 2020-03-06 | W·M·巴尔公司 | Surface disinfectants with residual biocidal properties |
| CN108008862A (en) * | 2017-12-19 | 2018-05-08 | 上海天马微电子有限公司 | Touch film layer, touch panel and touch display device thereof |
| CN108102807A (en) * | 2017-12-21 | 2018-06-01 | 广东德新科技孵化器有限公司 | A kind of wood furniture surface cleaning renovation agent and preparation method thereof |
| CN108653098A (en) * | 2018-07-11 | 2018-10-16 | 中山市智联企业孵化器发展有限公司 | A kind of plant body wash and preparation method thereof |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN102965214A (en) | Cleaning composition | |
| US6191086B1 (en) | Cleaning composition and method for removing residues | |
| JP3441715B2 (en) | Aqueous rinse composition and method using the same | |
| JP5801594B2 (en) | Cleaning composition, cleaning method using the same, and semiconductor device manufacturing method | |
| JP2007510173A (en) | Process of using bischoline and trischoline in cleaning quartz coated polysilicon and other materials | |
| JP6488507B2 (en) | Stripper composition for removing photoresist and photoresist stripping method using the same | |
| CN1724626A (en) | Composition and use thereof for removing photoresist and/or etch residues from substrates | |
| JP2009231354A (en) | Cleaning liquid for semiconductor device and cleaning method | |
| JP2005043873A (en) | Photoresist stripping liquid composition and method for stripping photoresist by using the same | |
| CN103725455B (en) | Cleaning composition and cleaning method for semiconductor manufacturing process | |
| JP5824249B2 (en) | Cleaning liquid composition | |
| JP4902898B2 (en) | Stabilized non-aqueous cleaning composition for microelectronic substrates | |
| KR101847208B1 (en) | Detergent composition for flat panel display device | |
| KR101459725B1 (en) | Stripper composition for removing post-etch residues and photoresist etch polymer | |
| KR20090045859A (en) | Release agent for polyimide | |
| JP2005060660A (en) | Cleaning solution for semiconductor substrates | |
| KR20120007339A (en) | Detergent composition | |
| JP4485786B2 (en) | Cleaning solution for semiconductor substrates | |
| JP5603948B2 (en) | Cleaning composition for glass substrate of flat panel display device | |
| EP1883863B1 (en) | Compositions for the removal of post-etch and ashed photoresist residues and bulk photoresist | |
| KR101799592B1 (en) | A Detergent Composition For A Substrate Of Flat Panel Display Device And Cleaning Method Using The Same | |
| TWI491729B (en) | Cleaning composition | |
| JP4215537B2 (en) | Resist stripper | |
| JP2011028146A (en) | Resist removing agent | |
| JP5903228B2 (en) | Cleaning composition and method for producing array substrate for liquid crystal display device using the same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C53 | Correction of patent of invention or patent application | ||
| CB02 | Change of applicant information |
Address after: Jeonbuk, South Korea Applicant after: Tongwoo Fine Chemicals Co., Ltd. Address before: South Korea Jeonbuk Iksan city and new hole 740-30 Applicant before: Dongwoo Fine Chem Co Ltd |
|
| COR | Change of bibliographic data |
Free format text: CORRECT: APPLICANT; FROM: DONGWOO FINE-CHEM CO., LTD. TO: TONGWOO FINE CHEMICALS CO., LTD. |
|
| RJ01 | Rejection of invention patent application after publication | ||
| RJ01 | Rejection of invention patent application after publication |
Application publication date: 20130313 |