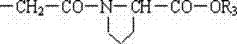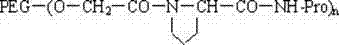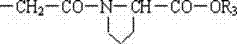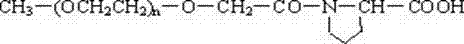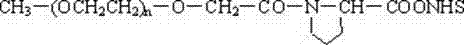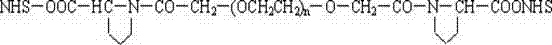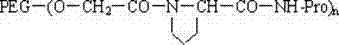CN102964587A - Preparation method and application of acid or active ester of polyethylene glycol with tail end connected with proline - Google Patents
Preparation method and application of acid or active ester of polyethylene glycol with tail end connected with proline Download PDFInfo
- Publication number
- CN102964587A CN102964587A CN2012104460857A CN201210446085A CN102964587A CN 102964587 A CN102964587 A CN 102964587A CN 2012104460857 A CN2012104460857 A CN 2012104460857A CN 201210446085 A CN201210446085 A CN 201210446085A CN 102964587 A CN102964587 A CN 102964587A
- Authority
- CN
- China
- Prior art keywords
- proline
- pro
- ester
- peg
- acid
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 229920001223 polyethylene glycol Polymers 0.000 title claims abstract description 54
- 239000002202 Polyethylene glycol Substances 0.000 title claims abstract description 46
- 150000002148 esters Chemical class 0.000 title claims abstract description 32
- ONIBWKKTOPOVIA-UHFFFAOYSA-N Proline Natural products OC(=O)C1CCCN1 ONIBWKKTOPOVIA-UHFFFAOYSA-N 0.000 title claims abstract description 27
- 238000002360 preparation method Methods 0.000 title claims abstract description 17
- 239000002253 acid Substances 0.000 title claims abstract description 14
- 230000007062 hydrolysis Effects 0.000 claims abstract description 8
- 238000006460 hydrolysis reaction Methods 0.000 claims abstract description 8
- 239000003814 drug Substances 0.000 claims abstract description 7
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 claims description 28
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 claims description 25
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 claims description 20
- 239000000047 product Substances 0.000 claims description 19
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 claims description 18
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 claims description 18
- 102000004169 proteins and genes Human genes 0.000 claims description 17
- 108090000623 proteins and genes Proteins 0.000 claims description 17
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 claims description 12
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 claims description 12
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 claims description 12
- -1 polyoxyethylene Polymers 0.000 claims description 12
- QOSSAOTZNIDXMA-UHFFFAOYSA-N Dicylcohexylcarbodiimide Chemical compound C1CCCCC1N=C=NC1CCCCC1 QOSSAOTZNIDXMA-UHFFFAOYSA-N 0.000 claims description 11
- 238000006243 chemical reaction Methods 0.000 claims description 10
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 claims description 10
- 102000004190 Enzymes Human genes 0.000 claims description 9
- 108090000790 Enzymes Proteins 0.000 claims description 9
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims description 8
- IXCSERBJSXMMFS-UHFFFAOYSA-N hydrogen chloride Substances Cl.Cl IXCSERBJSXMMFS-UHFFFAOYSA-N 0.000 claims description 6
- 229910000041 hydrogen chloride Inorganic materials 0.000 claims description 6
- 238000000034 method Methods 0.000 claims description 6
- 229940088623 biologically active substance Drugs 0.000 claims description 5
- 230000004048 modification Effects 0.000 claims description 5
- 238000012986 modification Methods 0.000 claims description 5
- 238000003756 stirring Methods 0.000 claims description 5
- PQJJJMRNHATNKG-UHFFFAOYSA-N ethyl bromoacetate Chemical compound CCOC(=O)CBr PQJJJMRNHATNKG-UHFFFAOYSA-N 0.000 claims description 4
- 239000007789 gas Substances 0.000 claims description 4
- 239000003607 modifier Substances 0.000 claims description 4
- 235000006408 oxalic acid Nutrition 0.000 claims description 4
- LPNYRYFBWFDTMA-UHFFFAOYSA-N potassium tert-butoxide Chemical compound [K+].CC(C)(C)[O-] LPNYRYFBWFDTMA-UHFFFAOYSA-N 0.000 claims description 4
- 229920006395 saturated elastomer Polymers 0.000 claims description 4
- 239000002502 liposome Substances 0.000 claims description 3
- 239000000975 dye Substances 0.000 claims description 2
- 239000000706 filtrate Substances 0.000 claims description 2
- 239000002777 nucleoside Substances 0.000 claims description 2
- 125000003835 nucleoside group Chemical group 0.000 claims description 2
- 229920001184 polypeptide Polymers 0.000 claims description 2
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 claims description 2
- 102000004196 processed proteins & peptides Human genes 0.000 claims description 2
- 108090000765 processed proteins & peptides Proteins 0.000 claims description 2
- 229920003171 Poly (ethylene oxide) Polymers 0.000 claims 7
- 239000004698 Polyethylene Substances 0.000 claims 2
- QPNJHVDIRZNKOX-LURJTMIESA-N ethyl (2s)-pyrrolidine-2-carboxylate Chemical compound CCOC(=O)[C@@H]1CCCN1 QPNJHVDIRZNKOX-LURJTMIESA-N 0.000 claims 2
- 229920000573 polyethylene Polymers 0.000 claims 2
- 238000001035 drying Methods 0.000 claims 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims 1
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 claims 1
- 238000001556 precipitation Methods 0.000 claims 1
- 238000004062 sedimentation Methods 0.000 claims 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 abstract description 12
- 239000013543 active substance Substances 0.000 abstract description 5
- 229960002317 succinimide Drugs 0.000 abstract description 4
- 125000003277 amino group Chemical group 0.000 abstract description 3
- 229920000642 polymer Polymers 0.000 abstract description 2
- ONIBWKKTOPOVIA-BYPYZUCNSA-N L-Proline Chemical compound OC(=O)[C@@H]1CCCN1 ONIBWKKTOPOVIA-BYPYZUCNSA-N 0.000 description 16
- 239000002244 precipitate Substances 0.000 description 9
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 8
- DUJGQVVONTYHLT-RGMNGODLSA-N ethyl (2s)-pyrrolidine-2-carboxylate;hydrochloride Chemical compound Cl.CCOC(=O)[C@@H]1CCCN1 DUJGQVVONTYHLT-RGMNGODLSA-N 0.000 description 8
- 230000009257 reactivity Effects 0.000 description 8
- 238000010992 reflux Methods 0.000 description 6
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 5
- 239000000284 extract Substances 0.000 description 5
- ADFXKUOMJKEIND-UHFFFAOYSA-N 1,3-dicyclohexylurea Chemical compound C1CCCCC1NC(=O)NC1CCCCC1 ADFXKUOMJKEIND-UHFFFAOYSA-N 0.000 description 4
- NQTADLQHYWFPDB-UHFFFAOYSA-N N-Hydroxysuccinimide Chemical compound ON1C(=O)CCC1=O NQTADLQHYWFPDB-UHFFFAOYSA-N 0.000 description 4
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 4
- 230000015572 biosynthetic process Effects 0.000 description 4
- 239000012141 concentrate Substances 0.000 description 4
- 239000008367 deionised water Substances 0.000 description 4
- 229910021641 deionized water Inorganic materials 0.000 description 4
- 238000003786 synthesis reaction Methods 0.000 description 4
- 229940079593 drug Drugs 0.000 description 3
- 229920001427 mPEG Polymers 0.000 description 3
- FALRKNHUBBKYCC-UHFFFAOYSA-N 2-(chloromethyl)pyridine-3-carbonitrile Chemical compound ClCC1=NC=CC=C1C#N FALRKNHUBBKYCC-UHFFFAOYSA-N 0.000 description 2
- 230000003213 activating effect Effects 0.000 description 2
- 230000004913 activation Effects 0.000 description 2
- 150000001408 amides Chemical class 0.000 description 2
- KZNICNPSHKQLFF-UHFFFAOYSA-N dihydromaleimide Natural products O=C1CCC(=O)N1 KZNICNPSHKQLFF-UHFFFAOYSA-N 0.000 description 2
- 238000004090 dissolution Methods 0.000 description 2
- 230000005847 immunogenicity Effects 0.000 description 2
- 239000003960 organic solvent Substances 0.000 description 2
- 239000002904 solvent Substances 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- KDYFGRWQOYBRFD-UHFFFAOYSA-N succinic acid Chemical compound OC(=O)CCC(O)=O KDYFGRWQOYBRFD-UHFFFAOYSA-N 0.000 description 2
- 229940014800 succinic anhydride Drugs 0.000 description 2
- 238000013268 sustained release Methods 0.000 description 2
- 239000012730 sustained-release form Substances 0.000 description 2
- 238000005292 vacuum distillation Methods 0.000 description 2
- 229920003169 water-soluble polymer Polymers 0.000 description 2
- UBUGOZFEGIKNLN-RGMNGODLSA-N (2s)-1-ethylpyrrolidine-2-carboxylic acid;hydrochloride Chemical compound Cl.CCN1CCC[C@H]1C(O)=O UBUGOZFEGIKNLN-RGMNGODLSA-N 0.000 description 1
- ODIGIKRIUKFKHP-UHFFFAOYSA-N (n-propan-2-yloxycarbonylanilino) acetate Chemical compound CC(C)OC(=O)N(OC(C)=O)C1=CC=CC=C1 ODIGIKRIUKFKHP-UHFFFAOYSA-N 0.000 description 1
- QXZGLTYKKZKGLN-UHFFFAOYSA-N 4-(2,5-dioxopyrrolidin-1-yl)oxy-4-oxobutanoic acid Chemical compound OC(=O)CCC(=O)ON1C(=O)CCC1=O QXZGLTYKKZKGLN-UHFFFAOYSA-N 0.000 description 1
- XBDQKXXYIPTUBI-UHFFFAOYSA-M Propionate Chemical class CCC([O-])=O XBDQKXXYIPTUBI-UHFFFAOYSA-M 0.000 description 1
- XBDQKXXYIPTUBI-UHFFFAOYSA-N Propionic acid Chemical group CCC(O)=O XBDQKXXYIPTUBI-UHFFFAOYSA-N 0.000 description 1
- 238000005904 alkaline hydrolysis reaction Methods 0.000 description 1
- 230000017531 blood circulation Effects 0.000 description 1
- 150000001732 carboxylic acid derivatives Chemical class 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 125000004093 cyano group Chemical group *C#N 0.000 description 1
- 230000007547 defect Effects 0.000 description 1
- 230000008030 elimination Effects 0.000 description 1
- 238000003379 elimination reaction Methods 0.000 description 1
- 238000005886 esterification reaction Methods 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- 239000012074 organic phase Substances 0.000 description 1
- 230000020477 pH reduction Effects 0.000 description 1
- 239000012071 phase Substances 0.000 description 1
- UQDJGEHQDNVPGU-UHFFFAOYSA-N serine phosphoethanolamine Chemical compound [NH3+]CCOP([O-])(=O)OCC([NH3+])C([O-])=O UQDJGEHQDNVPGU-UHFFFAOYSA-N 0.000 description 1
- 238000001179 sorption measurement Methods 0.000 description 1
- 239000001384 succinic acid Substances 0.000 description 1
Landscapes
- Medicinal Preparation (AREA)
- Peptides Or Proteins (AREA)
Abstract
本发明公开了一种末端连接脯氨酸的聚乙二醇的酸或活性酯的制法和应用,该发明应用于医药领域。此聚合物活性酯在水中水解半衰期为10.9分钟,具有理想的与生物活性物质的氨基反应的活性。其结构式如下:R1-(OCH2CH2)n-O-R2其中n=43~680;R1可以是CH3-,也可以是R2R2的结构如下:R3可以是H-,也可以是-N-丁二酰亚胺基。The invention discloses a preparation method and application of polyethylene glycol acid or active ester whose end is connected with proline, and the invention is applied in the field of medicine. The polymer active ester has a hydrolysis half-life of 10.9 minutes in water, and has an ideal activity of reacting with amino groups of biologically active substances. Its structural formula is as follows: R 1 -(OCH 2 CH 2 ) n -OR 2 where n=43~680; R 1 can be CH 3 - or R 2 The structure of R 2 is as follows: R 3 may be H- or -N-succinimide.
Description
技术领域 technical field
本发明涉及聚乙二醇水溶性聚合物及其制备方法和应用,尤其涉及聚乙二醇活性酯及其制备方法和应用。 The invention relates to a polyethylene glycol water-soluble polymer and a preparation method and application thereof, in particular to a polyethylene glycol active ester and a preparation method and application thereof.
背景技术 Background technique
使用化学方法将水溶性聚合物聚乙二醇(Polyethylene Glycol,PEG)结合到分子和表面在生物技术中具有重大的应用意义。PEG在生物技术领域有着广泛的应用:一些PEG的活性衍生物结合到蛋白质、酶的分子上,即对酶进行修饰,由于PEG可以溶于有机溶剂,PEG结合到酶分子可以使PEG与酶的结合物溶于有机溶剂;与未修饰的蛋白质相比,将PEG连接到蛋白质上可减小蛋白的免疫原性、降低肾脏排除速率,并可从中促使结合物在血液循环中的半衰期的增加;PEG结合到表面可减小蛋白质和细胞的吸附并改变表面的带电性质,PEG结合到脂质体上可以使其半衰期有极大增加,并能够增加其作为药物缓释的介质的能力。 The use of chemical methods to bind the water-soluble polymer polyethylene glycol (PEG) to molecules and surfaces has significant applications in biotechnology. PEG has a wide range of applications in the field of biotechnology: some active derivatives of PEG are bound to proteins and enzyme molecules, that is, enzymes are modified. Since PEG can be dissolved in organic solvents, PEG combined with enzyme molecules can make PEG and enzyme molecules The conjugate is soluble in organic solvents; compared with unmodified protein, linking PEG to the protein can reduce the immunogenicity of the protein, reduce the rate of renal elimination, and thereby increase the half-life of the conjugate in blood circulation; The binding of PEG to the surface can reduce the adsorption of proteins and cells and change the charged properties of the surface. The binding of PEG to liposomes can greatly increase its half-life and increase its ability as a medium for drug sustained release.
PEG只有经过活化才能够结合到分子或表面上。因此,一系列的活化衍生物被制备出来。目前应用较广的修饰剂活化类型为羧酸的活化酯类。该类修饰剂是通过活化酯对蛋白的自由氨基进行酰化,从而将PEG链结合到蛋白质分子上实现修饰的。 PEG can only bind to molecules or surfaces after activation. Therefore, a series of activated derivatives were prepared. At present, the widely used modifier activation type is the activated ester of carboxylic acid. This type of modifier is modified by activating the ester to acylate the free amino group of the protein, thereby binding the PEG chain to the protein molecule.
目前,许多用于修饰的活化的羧基PEG具有较差的反应活性。例如,羧甲基化PEG(CM-PEG)的NHS酯的反应活性太强,以至溶解在水中立刻水解(在pH8、25°C下的水解半衰期为0.75 min),这种高反应活性是羧甲基化PEG活性酯的严重缺陷。而第一代修饰剂中活化的SC-PEG虽然有较好的反应活性(在pH8、25°C下的半衰期为20.4 min),但由于在分子内含有不稳定的酯键,现已较少使用。又如丁二酸酐丁二酰亚胺酯活化的PEG(succinimidyl succinate,SS-PEG)。SS-PEG的制备是通过mPEG与丁二酸酐发生酯化反应,使丁二酸酐开环,从而在PEG的链端造成一个羧基,然后又将羧基活化成丁二酰亚胺酯。该试剂在与蛋白质结合后还存在一个酯键,而酯键由于其固有的不稳定性在PEG高分子结合到蛋白质上时非常容易被水解。这种水解不仅会使PEG链从蛋白上脱落,丧失PEG修饰带来的益处,而且留在蛋白质上的丁二酸尾部还可作为半抗原导致蛋白质的免疫原性。Harris等人在mPEG的末端引入丙酸单位,使得这种丙酸PEG活性酯的水解半衰期增加到16.5分钟,但该方法涉及了在PEG一端引入氰基,需要再进行强酸和强碱的水解才能得到产物,极有可能破坏PEG主链。 Currently, many activated carboxy-PEGs used for modification have poor reactivity. For example, the NHS ester of carboxymethylated PEG (CM-PEG) is so reactive that it hydrolyzes immediately upon dissolution in water (hydrolysis half-life of 0.75 min at pH 8, 25°C), and this high reactivity is A serious drawback of methylated PEG active esters. Although the activated SC-PEG in the first generation of modifiers has better reactivity (half-life of 20.4 min at pH 8, 25°C), due to the unstable ester bond in the molecule, it is now less use. Another example is PEG (succinimidyl succinate, SS-PEG) activated by succinic anhydride succinimide ester. The preparation of SS-PEG is through the esterification reaction between mPEG and succinic anhydride to open the ring of succinic anhydride, thereby forming a carboxyl group at the chain end of PEG, and then activating the carboxyl group into succinimide ester. The reagent still has an ester bond after it is bound to the protein, and the ester bond is very easily hydrolyzed when the PEG polymer is bound to the protein due to its inherent instability. This hydrolysis will not only detach the PEG chain from the protein and lose the benefits of PEG modification, but also the succinic acid tail left on the protein can act as a hapten to cause the immunogenicity of the protein. Harris et al. introduced propionate units at the end of mPEG, which increased the hydrolysis half-life of this propionate PEG active ester to 16.5 minutes, but this method involved the introduction of a cyano group at one end of PEG, which required further hydrolysis with strong acid and strong base. Obtained products are very likely to destroy the PEG main chain.
目前,许多用于修饰的活化的羧基PEG具有很差的反应活性:不是反应太快就是反应太慢。例如,羧甲基化PEG(CM-PEG)的NHS酯的反应活性太强,以至溶解在水中立刻水解,这种高反应活性是羧甲基化PEG活性酯的严重缺陷。 Currently, many activated carboxyl PEGs used for modification have poor reactivity: they either react too quickly or react too slowly. For example, the NHS ester of carboxymethylated PEG (CM-PEG) is so reactive that it hydrolyzes immediately upon dissolution in water, and this high reactivity is a serious drawback of the active ester of carboxymethylated PEG.
发明内容 Contents of the invention
本发明需要解决的技术问题是公开一种末端连接脯氨酸的聚乙二醇的酸或活性酯的制法和应用,以克服现有技术存在的缺陷,满足医药领域和生物工程领域发展的需要。 The technical problem to be solved in the present invention is to disclose a preparation method and application of a polyethylene glycol acid or active ester connected to proline at the end, so as to overcome the defects in the prior art and meet the development needs of the fields of medicine and bioengineering need.
本发明的末端连接脯氨酸的聚乙二醇的活性酸或酯的结构式如下: The structural formula of the active acid or ester of polyethylene glycol whose end is connected to proline of the present invention is as follows:
R1-(OCH2CH2)n-O-R2 R 1 -(OCH 2 CH 2 ) n -O-R 2
其中n=43~680; where n=43~680;
R1可以是CH3-,也可以是R2 R 1 can be CH 3 - or R 2
R2的结构如下: The structure of R2 is as follows:
R3可以是H-,也可以是-N-丁二酰亚胺基。 R 3 may be H- or -N-succinimide.
本发明提供的连接脯氨酸的聚乙二醇的活性酯具有合适的反应活性,活性酯在水中的水解半衰期为10.9分钟,且不含有其它的酯键连接。 The active ester of polyethylene glycol linked to proline provided by the invention has suitable reactivity, the hydrolysis half-life of the active ester in water is 10.9 minutes, and does not contain other ester linkages.
本发明的连接脯氨酸的聚乙二醇的活性酯的制备方法包括如下步骤: The preparation method of the active ester of polyethylene glycol linked to proline of the present invention comprises the steps:
(1)CM-PEG(羧甲基化聚乙二醇)的制备: (1) Preparation of CM-PEG (carboxymethylated polyethylene glycol):
将聚乙二醇溶于甲苯中,加入叔丁醇钾回流反应1~2小时,加入溴乙酸乙酯回流反应2~4小时,然后室温反应18~24小时,浓缩后用乙醚沉淀,沉淀物碱水解然后酸化,采用常规的方法收集产物; Dissolve polyethylene glycol in toluene, add potassium tert-butoxide to reflux for 1-2 hours, add ethyl bromoacetate to reflux for 2-4 hours, then react at room temperature for 18-24 hours, concentrate and precipitate with ether, the precipitate Alkaline hydrolysis and then acidification, using conventional methods to collect the product;
(2)脯氨酸乙酯盐酸盐的制备: (2) Preparation of proline ethyl ester hydrochloride:
将氯化氢气体通入含有脯氨酸的乙醇直到饱和,然后收集脯氨酸乙酯盐酸盐; Pass hydrogen chloride gas into ethanol containing proline until saturated, then collect proline ethyl ester hydrochloride;
(3)末端连接脯氨酸的聚乙二醇酸衍生物的制备: (3) Preparation of polyethylene glycol acid derivatives with proline connected at the end:
取步骤(1)中制备的CM-PEG溶于二氯甲烷,然后依次加入二环己基碳二亚胺、脯氨酸乙酯盐酸盐,然后用三乙胺将溶液的pH值调节到8~9,反应8~12小时,过滤,滤液加入乙醚,收集沉淀产物,将产物干燥后溶于水,加入氢氧化钠溶液调节pH至10~12,反应1~3小时,然后用草酸将溶液pH调节到2~4,收集产物,获得末端连接脯氨酸的聚乙二醇酸衍生物。 Take the CM-PEG prepared in step (1) and dissolve it in dichloromethane, then add dicyclohexylcarbodiimide and proline ethyl ester hydrochloride in sequence, and then adjust the pH value of the solution to 8 with triethylamine ~9, react for 8~12 hours, filter, add diethyl ether to the filtrate, collect the precipitated product, dry the product and dissolve it in water, add sodium hydroxide solution to adjust the pH to 10~12, react for 1~3 hours, and then dilute the solution with oxalic acid The pH was adjusted to 2-4, and the product was collected to obtain a polyethylene glycol acid derivative whose end was linked to proline.
(4)取步骤(3)的产物溶于二氯甲烷,加入1.5~3倍摩尔量的二环己基碳二亚胺和1.5~3倍摩尔量的N-羟基丁二酰亚胺,10~30℃搅拌反应20~28小时,收集产物,获得末端连接脯氨酸的聚乙二醇活性酯。 (4) Dissolve the product of step (3) in dichloromethane, add 1.5 to 3 times the molar amount of dicyclohexylcarbodiimide and 1.5 to 3 times the molar amount of N-hydroxysuccinimide, 10 to 3 times the molar amount The reaction was stirred at 30°C for 20-28 hours, and the product was collected to obtain a polyethylene glycol active ester whose end was linked to proline.
所说的脯氨酸为D型、L型或外消旋体; Said proline is D-form, L-form or racemate;
聚乙二醇优选的分子量为2000~30000 Da。 The preferred molecular weight of polyethylene glycol is 2000~30000 Da.
本发明的末端连接脯氨酸的聚乙二醇的活性酯,在水中拥有10.9分钟的水解半衰期,具有理想的与生物活性物质氨基的反应活性,可用于与生物活性物质相连接,制备PEG修饰的生物活性物质,用于制备缓释药物的介质等。 The active ester of polyethylene glycol whose end is connected to proline has a hydrolysis half-life of 10.9 minutes in water, has ideal reactivity with the amino group of biologically active substances, and can be used to link with biologically active substances to prepare PEG modification Biologically active substances, media for the preparation of sustained-release drugs, etc.
本发明还涉及一种所说的末端连接脯氨酸的聚乙二醇活性酸或酯与生物活性物质的结合物,结构通式如下: The present invention also relates to a combination of said polyethylene glycol active acid or ester connected to proline at the end and a biologically active substance, the general structural formula of which is as follows:
其中:n为1或2,NH-Pro部分代表生物活性物质的氨基位点。 Wherein: n is 1 or 2, and the NH-Pro part represents the amino site of the biologically active substance.
生物活性物质选自蛋白质、酶、多肽、药物、染料、核苷、酯类或脂质体。 Biologically active substances are selected from proteins, enzymes, polypeptides, drugs, dyes, nucleosides, esters or liposomes.
采用上述方法制备的末端连接脯氨酸的聚乙二醇活性酯,具有良好的反应活性,可用于与蛋白质或酶的分子相连接,以及用于制备缓释药物的介质。 The proline-linked polyethylene glycol active ester prepared by the above method has good reactivity and can be used for linking with protein or enzyme molecules and for preparing a medium for slow-release medicine.
具体实施方式 Detailed ways
实施例1 Example 1
与分子量为5000 Da的单甲氧基PEG以酰胺键连接的脯氨酸活性酯的合成 Synthesis of proline active ester linked with monomethoxy PEG with a molecular weight of 5000 Da via amide bond
步骤1:CM-PEG(羧甲基化聚乙二醇)的制备: Step 1: Preparation of CM-PEG (Carboxymethylated Polyethylene Glycol):
将分子量为5000 Da的单甲氧基聚乙二醇15 g(3 mmol)溶于甲苯中,蒸馏出一部分甲苯以共沸除水,然后加入4 mmol的叔丁醇钾回流反应1.5小时,而后缓慢加入4 mmol的溴乙酸乙酯回流反应3小时,然后室温反应21小时,过滤除去沉淀后减压蒸去溶剂,残留物加少量二氯甲烷溶解,用干燥乙醚沉淀出产物,将该产物溶于去离子水中,加入0.1 mol/L的氢氧化钠溶液直到溶液的pH值稳定在pH10,然后用0.1 mol/L的盐酸调节溶液的pH到3,用等量的氯仿萃取溶液三次,合并有机相并用无水硫酸钠干燥,浓缩后用乙醚沉淀干燥得到分子量为5000 Da的CM-PEG,分子式如下:CH3O-(CH2CH2O)n-CH2COOH。 Dissolve 15 g (3 mmol) of monomethoxypolyethylene glycol with a molecular weight of 5000 Da in toluene, distill off part of the toluene to remove water azeotropically, then add 4 mmol of potassium tert-butoxide to reflux for 1.5 hours, and then Slowly add 4 mmol of ethyl bromoacetate to reflux reaction for 3 hours, then react at room temperature for 21 hours, remove the precipitate by filtration, evaporate the solvent under reduced pressure, dissolve the residue with a small amount of dichloromethane, precipitate the product with dry ether, dissolve the product In deionized water, add 0.1 mol/L sodium hydroxide solution until the pH value of the solution is stable at pH 10, then adjust the pH of the solution to 3 with 0.1 mol/L hydrochloric acid, extract the solution three times with an equal amount of chloroform, and combine the organic The phases were combined and dried with anhydrous sodium sulfate, concentrated and then precipitated and dried with ether to obtain CM-PEG with a molecular weight of 5000 Da, the molecular formula is as follows: CH 3 O-(CH 2 CH 2 O) n -CH 2 COOH.
步骤2:脯氨酸乙酯盐酸盐的制备: Step 2: Preparation of proline ethyl ester hydrochloride:
将20 g 脯氨酸加入到300 mL无水乙醇中,在搅拌下通入干燥的氯化氢气体直到饱和,然后加入无水乙醇进行减压蒸馏,将多余的氯化氢除去,接着将溶液浓缩至合适的体积,用干燥无水的乙醚沉淀得到脯氨酸乙酯盐酸盐。 Add 20 g of proline to 300 mL of absolute ethanol, pass in dry hydrogen chloride gas under stirring until saturated, then add absolute ethanol for vacuum distillation to remove excess hydrogen chloride, and then concentrate the solution to a suitable volume, and precipitated with dry anhydrous ether to obtain proline ethyl ester hydrochloride.
步骤3:合成以酰胺键连接的mPEG-脯氨酸缀合物: Step 3: Synthesis of mPEG-proline conjugate linked by amide linkage:
取7.5 g(1.5 mmol)步骤(1)中制备的CM-PEG溶于40 mL干燥的二氯甲烷中,然后依次加入2 mmol的二环己基碳二亚胺、2 mmol的脯氨酸乙酯盐酸盐,然后用三乙胺将溶液的pH值调节到8,搅拌反应12小时,过滤除去沉淀后,加入无水乙醚沉淀产物,将产物干燥后溶于去离子水,加入0.1 mol/L氢氧化钠溶液调节pH至11,反应2小时,然后用草酸将溶液pH调节到3,用氯仿萃取溶液三次,合并萃取液用无水硫酸钠干燥,溶液浓缩后再用无水乙醚沉淀得到分子量为5000 Da的以酰胺键连接的mPEG-脯氨酸缀合物。 Take 7.5 g (1.5 mmol) of CM-PEG prepared in step (1) and dissolve in 40 mL of dry dichloromethane, then add 2 mmol of dicyclohexylcarbodiimide and 2 mmol of ethyl proline Hydrochloride, then adjust the pH value of the solution to 8 with triethylamine, stir for 12 hours, filter to remove the precipitate, add anhydrous ether to precipitate the product, dry the product and dissolve it in deionized water, add 0.1 mol/L Sodium hydroxide solution adjusted the pH to 11, reacted for 2 hours, then adjusted the pH of the solution to 3 with oxalic acid, extracted the solution three times with chloroform, combined the extracts and dried them with anhydrous sodium sulfate, concentrated the solution and then precipitated with anhydrous ether to obtain the molecular weight 5000 Da amide-linked mPEG-proline conjugate.
步骤4: Step 4:
取步骤3制备的分子量为5000 Da的mPEG-脯氨酸,溶于干燥精制的无水二氯甲烷中,加入1.2倍摩尔量的二环己基碳二亚胺(DCC)和1.2倍摩尔量的N-羟基丁二酰亚胺,室温搅拌反应24小时,停止反应滤除沉淀二环己基脲(DCU),溶液用无水乙醚沉淀得到产物分子量为5000 Da的mPEG-脯氨酸的N-羟基丁二酰亚胺酯。 Take the mPEG-proline with a molecular weight of 5000 Da prepared in step 3, dissolve it in dry and refined anhydrous dichloromethane, add 1.2 times the molar amount of dicyclohexylcarbodiimide (DCC) and 1.2 times the molar amount of N-hydroxysuccinimide, stirred at room temperature for 24 hours, stopped the reaction and filtered off the precipitated dicyclohexyl urea (DCU), and the solution was precipitated with anhydrous ether to obtain the N-hydroxyl group of mPEG-proline with a molecular weight of 5000 Da Succinimide ester.
实施例2 Example 2
与分子量为20000 Da的PEG二酸以酰胺键连接的脯氨酸活性酯的合成 Synthesis of proline active ester linked with PEG diacid with molecular weight of 20000 Da by amide bond
步骤1:CM-PEG二酸的制备: Step 1: Preparation of CM-PEG diacid:
将分子量为20000 Da的单甲氧基聚乙二醇20 g(1 mmol)溶于适量的甲苯中,蒸馏出一部分甲苯以共沸除水,然后加入3 mmol的叔丁醇钾回流反应2小时,而后缓慢加入3 mmol的溴乙酸乙酯回流反应4小时,然后室温反应18小时,过滤除去沉淀后减压蒸去溶剂,残留物加入二氯甲烷溶解,用干燥乙醚沉淀出产物,将该产物溶于去离子水中,逐渐加入0.1 mol/L的氢氧化钠溶液直到溶液的pH值稳定在pH10,然后用0.1 mol/L的盐酸调节溶液的pH到3,用等量的氯仿萃取溶液三次,合并有机相并用无水硫酸钠干燥,浓缩后用乙醚沉淀干燥得到分子量为20000 Da的CM-PEG二酸,化学式如下:HOOCCH2O-(CH2CH2O)n-CH2COOH。 Dissolve 20 g (1 mmol) of monomethoxypolyethylene glycol with a molecular weight of 20,000 Da in an appropriate amount of toluene, distill a part of the toluene to remove water azeotropically, and then add 3 mmol of potassium tert-butoxide to reflux for 2 hours , and then slowly add 3 mmol of ethyl bromoacetate to reflux reaction for 4 hours, then react at room temperature for 18 hours, filter and remove the precipitate, evaporate the solvent under reduced pressure, add dichloromethane to the residue to dissolve, and precipitate the product with dry ether. Dissolve in deionized water, gradually add 0.1 mol/L sodium hydroxide solution until the pH value of the solution is stable at pH 10, then adjust the pH of the solution to 3 with 0.1 mol/L hydrochloric acid, extract the solution three times with an equal amount of chloroform, The organic phases were combined and dried with anhydrous sodium sulfate, concentrated and then precipitated and dried with ether to obtain a CM-PEG diacid with a molecular weight of 20000 Da. The chemical formula is as follows: HOOCCH 2 O-(CH 2 CH 2 O) n -CH 2 COOH.
步骤2:脯氨酸乙酯盐酸盐的制备: Step 2: Preparation of proline ethyl ester hydrochloride:
将20 g 脯氨酸加入到300 mL无水乙醇中,在搅拌下通入干燥的氯化氢气体直到饱和,然后加入无水乙醇进行减压蒸馏,将多余的氯化氢除去,接着将溶液浓缩至合适的体积,用干燥无水的乙醚沉淀得到脯氨酸乙酯盐酸盐。 Add 20 g of proline to 300 mL of absolute ethanol, pass in dry hydrogen chloride gas under stirring until saturated, then add absolute ethanol for vacuum distillation to remove excess hydrogen chloride, and then concentrate the solution to a suitable volume, and precipitated with dry anhydrous ether to obtain proline ethyl ester hydrochloride.
步骤3:合成以酰胺键连接的PEG-二脯氨酸缀合物: Step 3: Synthesis of PEG-diproline conjugates linked by amide bonds:
取10 g(0.5 mmol)步骤(1)中制备的CM-PEG二酸溶于40 mL干燥的二氯甲烷中,然后依次加入2 mmol的二环己基碳二亚胺(DCC)、2 mmol的脯氨酸乙酯盐酸盐,然后用三乙胺将溶液的pH值调节到8,搅拌反应过夜,过滤除去沉淀后,加入无水乙醚沉淀产物,将产物干燥后溶于去离子水,加入0.1 mol/L氢氧化钠溶液调节pH至11,然后用草酸将溶液pH调节到3,用氯仿萃取溶液三次,合并萃取液用无水硫酸钠干燥,溶液浓缩后再用无水乙醚沉淀得到分子量为20000 Da的PEG-二脯氨酸。 Take 10 g (0.5 mmol) of the CM-PEG diacid prepared in step (1) and dissolve it in 40 mL of dry dichloromethane, then add 2 mmol of dicyclohexylcarbodiimide (DCC), 2 mmol of Proline ethyl ester hydrochloride, then adjust the pH value of the solution to 8 with triethylamine, stir the reaction overnight, filter to remove the precipitate, add anhydrous ether to precipitate the product, dry the product and dissolve it in deionized water, add Adjust the pH to 11 with 0.1 mol/L sodium hydroxide solution, then adjust the pH of the solution to 3 with oxalic acid, extract the solution three times with chloroform, dry the combined extracts with anhydrous sodium sulfate, concentrate the solution and then precipitate with anhydrous ether to obtain the molecular weight PEG-diproline of 20000 Da.
步骤4: Step 4:
取步骤(3)制备的分子量为20000 Da的PEG-二脯氨酸,溶于干燥精制的无水二氯甲烷中,加入3倍摩尔量的二环己基碳二亚胺(DCC)和3倍摩尔量的N-羟基丁二酰亚胺,室温搅拌反应24小时,停止反应滤除沉淀二环己基脲(DCU),溶液用无水乙醚沉淀得到产物分子量为20000 Da的PEG-二脯氨酸的N-羟基丁二酰亚胺酯。 Take the PEG-diproline with a molecular weight of 20000 Da prepared in step (3), dissolve it in dry and refined anhydrous dichloromethane, add 3 times the molar amount of dicyclohexylcarbodiimide (DCC) and 3 times Molar amount of N-hydroxysuccinimide, stirred at room temperature for 24 hours, stopped the reaction and filtered off the precipitated dicyclohexyl urea (DCU), and the solution was precipitated with anhydrous ether to obtain PEG-diproline with a molecular weight of 20000 Da of N-hydroxysuccinimide esters.
Claims (8)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2012104460857A CN102964587A (en) | 2012-11-09 | 2012-11-09 | Preparation method and application of acid or active ester of polyethylene glycol with tail end connected with proline |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2012104460857A CN102964587A (en) | 2012-11-09 | 2012-11-09 | Preparation method and application of acid or active ester of polyethylene glycol with tail end connected with proline |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN102964587A true CN102964587A (en) | 2013-03-13 |
Family
ID=47795040
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN2012104460857A Pending CN102964587A (en) | 2012-11-09 | 2012-11-09 | Preparation method and application of acid or active ester of polyethylene glycol with tail end connected with proline |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN102964587A (en) |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5965566A (en) * | 1993-10-20 | 1999-10-12 | Enzon, Inc. | High molecular weight polymer-based prodrugs |
| CN1660920A (en) * | 2005-02-01 | 2005-08-31 | 华东理工大学 | Polyglycolic acid or active ester with ω-amino acid connected at the end, its preparation method and application |
| CN101870769A (en) * | 2010-06-13 | 2010-10-27 | 河北科技大学 | PEG, mPEG chemical modifier and method for preparing water-soluble resveratrol prodrug |
-
2012
- 2012-11-09 CN CN2012104460857A patent/CN102964587A/en active Pending
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5965566A (en) * | 1993-10-20 | 1999-10-12 | Enzon, Inc. | High molecular weight polymer-based prodrugs |
| CN1660920A (en) * | 2005-02-01 | 2005-08-31 | 华东理工大学 | Polyglycolic acid or active ester with ω-amino acid connected at the end, its preparation method and application |
| CN101870769A (en) * | 2010-06-13 | 2010-10-27 | 河北科技大学 | PEG, mPEG chemical modifier and method for preparing water-soluble resveratrol prodrug |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JPH08507549A (en) | Water-soluble non-immunogenic polyamide crosslinker | |
| CN1232567C (en) | End-amino water-soluble ultrabranching polyamide and preparation process thereof | |
| JPS61243026A (en) | Method for manufacturing polymerizable drugs | |
| WO2006088248A1 (en) | Polyoxyalkylene derivative | |
| CN111298132A (en) | A kind of dendrimer gemcitabine self-assembled nano-prodrug and its preparation method and application | |
| CN104926920A (en) | Protein polypeptide drug N-terminal fixed-point polyethylene glycol modification method | |
| JPH04501121A (en) | New biologically active drugs/polymer derivatives and their production methods | |
| CN102321239B (en) | Preparation method of water-soluble toluylene compound prodrugs | |
| WO2003059987A1 (en) | Multidrop tree branching functional polyethylene glycol, the preparation method and the use of it | |
| CN102643420B (en) | Poly alkyl ether compound with strange end group and double functional groups and application thereof | |
| CN109908084B (en) | A kind of platinum-crosslinked camptothecin prodrug micelle nanomedicine and its preparation method and application | |
| CN106188222A (en) | A kind of double dipeptide structure supermolecular gel factor and preparation method thereof | |
| CN110669810B (en) | A method for enzymatically preparing lysine oligopeptide and its monomethoxypolyethylene glycol modification | |
| CN102964588A (en) | Preparation method and application of acid or active ester of polyethylene glycol with tail end connected with aminophenyl propionic acid | |
| CN100546656C (en) | A kidney-targeted drug carrier and its prodrug, preparation method and application | |
| CN102964587A (en) | Preparation method and application of acid or active ester of polyethylene glycol with tail end connected with proline | |
| CN1210310C (en) | N-lactose acyl chitosan | |
| CN111454457B (en) | A kind of chiral peptide antibacterial polymer with dendrimer as side chain and preparation method thereof | |
| CN105017522A (en) | Polyethylene glycol-modified unnatural amino acid preparation method | |
| CN101307018A (en) | Diethylenetriaminepentaacetic acid carboxyl activated ester or analogue and its synthesis method and application | |
| JP2008308690A (en) | POLY (ETHYLENE GLYCOL) FUNCTIONAL DERIVATIVE AND METHOD FOR PRODUCING THE SAME | |
| CN116178733B (en) | Branched monodisperse PEG derivative based on trifunctional amino acid, preparation method and application | |
| Huang et al. | Synthesis of an ortho-phthalaldehyde-functionalized copolymer for rapid, chemoselective and efficient conjugation with native proteins under physiological conditions | |
| US5124437A (en) | Galactosamine substitute of poly-ω-substituted-L-glutamic acid (or aspartic acid) | |
| CN117126397A (en) | Synthesis method and application of protein-binding type precise polyethylene glycol |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| C02 | Deemed withdrawal of patent application after publication (patent law 2001) | ||
| WD01 | Invention patent application deemed withdrawn after publication |
Application publication date: 20130313 |