CN102885796A - Lacosamide tablet for treating epilepsy and preparation method for lacosamide tablet - Google Patents
Lacosamide tablet for treating epilepsy and preparation method for lacosamide tablet Download PDFInfo
- Publication number
- CN102885796A CN102885796A CN201210266439XA CN201210266439A CN102885796A CN 102885796 A CN102885796 A CN 102885796A CN 201210266439X A CN201210266439X A CN 201210266439XA CN 201210266439 A CN201210266439 A CN 201210266439A CN 102885796 A CN102885796 A CN 102885796A
- Authority
- CN
- China
- Prior art keywords
- lacosamide
- weight portion
- scheme
- tablet
- enclose
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- VPPJLAIAVCUEMN-GFCCVEGCSA-N lacosamide Chemical compound COC[C@@H](NC(C)=O)C(=O)NCC1=CC=CC=C1 VPPJLAIAVCUEMN-GFCCVEGCSA-N 0.000 title claims abstract description 70
- 229960002623 lacosamide Drugs 0.000 title claims abstract description 68
- 206010015037 epilepsy Diseases 0.000 title claims abstract description 14
- 238000002360 preparation method Methods 0.000 title claims abstract description 14
- 239000000463 material Substances 0.000 claims abstract description 38
- 239000011248 coating agent Substances 0.000 claims abstract description 25
- 238000000576 coating method Methods 0.000 claims abstract description 25
- 239000000945 filler Substances 0.000 claims abstract description 17
- 239000000314 lubricant Substances 0.000 claims abstract description 17
- 239000000080 wetting agent Substances 0.000 claims abstract description 11
- 238000004090 dissolution Methods 0.000 claims abstract description 8
- 239000003814 drug Substances 0.000 claims abstract description 7
- 239000000203 mixture Substances 0.000 claims description 24
- 238000000034 method Methods 0.000 claims description 21
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 19
- 239000003795 chemical substances by application Substances 0.000 claims description 16
- 239000008187 granular material Substances 0.000 claims description 16
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 claims description 16
- 229920002472 Starch Polymers 0.000 claims description 14
- 239000001253 polyvinylpolypyrrolidone Substances 0.000 claims description 14
- 235000013809 polyvinylpolypyrrolidone Nutrition 0.000 claims description 14
- 229920000523 polyvinylpolypyrrolidone Polymers 0.000 claims description 14
- 239000008107 starch Substances 0.000 claims description 14
- 235000019698 starch Nutrition 0.000 claims description 14
- 229940031703 low substituted hydroxypropyl cellulose Drugs 0.000 claims description 12
- 229920000858 Cyclodextrin Polymers 0.000 claims description 11
- HFHDHCJBZVLPGP-UHFFFAOYSA-N schardinger α-dextrin Chemical compound O1C(C(C2O)O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC(C(O)C2O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC2C(O)C(O)C1OC2CO HFHDHCJBZVLPGP-UHFFFAOYSA-N 0.000 claims description 11
- 239000011230 binding agent Substances 0.000 claims description 10
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims description 8
- 229920000168 Microcrystalline cellulose Polymers 0.000 claims description 8
- 238000005516 engineering process Methods 0.000 claims description 8
- 235000019359 magnesium stearate Nutrition 0.000 claims description 8
- 235000019813 microcrystalline cellulose Nutrition 0.000 claims description 8
- 239000008108 microcrystalline cellulose Substances 0.000 claims description 8
- 229940016286 microcrystalline cellulose Drugs 0.000 claims description 8
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 claims description 7
- 239000000741 silica gel Substances 0.000 claims description 7
- 229910002027 silica gel Inorganic materials 0.000 claims description 7
- DPXJVFZANSGRMM-UHFFFAOYSA-N acetic acid;2,3,4,5,6-pentahydroxyhexanal;sodium Chemical compound [Na].CC(O)=O.OCC(O)C(O)C(O)C(O)C=O DPXJVFZANSGRMM-UHFFFAOYSA-N 0.000 claims description 5
- 239000001768 carboxy methyl cellulose Substances 0.000 claims description 5
- FPAFDBFIGPHWGO-UHFFFAOYSA-N dioxosilane;oxomagnesium;hydrate Chemical group O.[Mg]=O.[Mg]=O.[Mg]=O.O=[Si]=O.O=[Si]=O.O=[Si]=O.O=[Si]=O FPAFDBFIGPHWGO-UHFFFAOYSA-N 0.000 claims description 5
- 235000019812 sodium carboxymethyl cellulose Nutrition 0.000 claims description 5
- 229920001027 sodium carboxymethylcellulose Polymers 0.000 claims description 5
- JLPULHDHAOZNQI-ZTIMHPMXSA-N 1-hexadecanoyl-2-(9Z,12Z-octadecadienoyl)-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCCCCCC\C=C/C\C=C/CCCCC JLPULHDHAOZNQI-ZTIMHPMXSA-N 0.000 claims description 4
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 claims description 4
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 claims description 4
- 229920000881 Modified starch Polymers 0.000 claims description 4
- 125000002057 carboxymethyl group Chemical group [H]OC(=O)C([H])([H])[*] 0.000 claims description 4
- 235000019441 ethanol Nutrition 0.000 claims description 4
- 239000008101 lactose Substances 0.000 claims description 4
- 238000005498 polishing Methods 0.000 claims description 4
- 238000009702 powder compression Methods 0.000 claims description 4
- 239000011734 sodium Substances 0.000 claims description 4
- 229910052708 sodium Inorganic materials 0.000 claims description 4
- 239000008347 soybean phospholipid Substances 0.000 claims description 4
- 210000002784 stomach Anatomy 0.000 claims description 4
- 238000004132 cross linking Methods 0.000 claims description 3
- IIZPXYDJLKNOIY-JXPKJXOSSA-N 1-palmitoyl-2-arachidonoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCC\C=C/C\C=C/C\C=C/C\C=C/CCCCC IIZPXYDJLKNOIY-JXPKJXOSSA-N 0.000 claims description 2
- 235000021355 Stearic acid Nutrition 0.000 claims description 2
- 235000010489 acacia gum Nutrition 0.000 claims description 2
- 239000001785 acacia senegal l. willd gum Substances 0.000 claims description 2
- 238000007906 compression Methods 0.000 claims description 2
- 230000006835 compression Effects 0.000 claims description 2
- 238000001035 drying Methods 0.000 claims description 2
- 235000010445 lecithin Nutrition 0.000 claims description 2
- 239000000787 lecithin Substances 0.000 claims description 2
- 229940067606 lecithin Drugs 0.000 claims description 2
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 claims description 2
- OQCDKBAXFALNLD-UHFFFAOYSA-N octadecanoic acid Natural products CCCCCCCC(C)CCCCCCCCC(O)=O OQCDKBAXFALNLD-UHFFFAOYSA-N 0.000 claims description 2
- 239000000843 powder Substances 0.000 claims description 2
- 229920006395 saturated elastomer Polymers 0.000 claims description 2
- 238000001694 spray drying Methods 0.000 claims description 2
- 239000008117 stearic acid Substances 0.000 claims description 2
- 238000005550 wet granulation Methods 0.000 claims description 2
- 239000000853 adhesive Substances 0.000 abstract 1
- 230000001070 adhesive effect Effects 0.000 abstract 1
- 239000007884 disintegrant Substances 0.000 abstract 1
- 239000000523 sample Substances 0.000 description 15
- 238000012360 testing method Methods 0.000 description 14
- 239000008213 purified water Substances 0.000 description 13
- 238000000227 grinding Methods 0.000 description 10
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 6
- 239000000047 product Substances 0.000 description 6
- 238000007605 air drying Methods 0.000 description 5
- 239000012467 final product Substances 0.000 description 5
- 238000007689 inspection Methods 0.000 description 5
- -1 oven dry Substances 0.000 description 5
- 238000012856 packing Methods 0.000 description 5
- 235000011837 pasties Nutrition 0.000 description 5
- 239000007779 soft material Substances 0.000 description 5
- 239000013068 control sample Substances 0.000 description 4
- 229940068196 placebo Drugs 0.000 description 4
- 239000000902 placebo Substances 0.000 description 4
- 230000000694 effects Effects 0.000 description 3
- 230000007774 longterm Effects 0.000 description 3
- 239000013558 reference substance Substances 0.000 description 3
- 206010010904 Convulsion Diseases 0.000 description 2
- DHMQDGOQFOQNFH-UHFFFAOYSA-N Glycine Chemical compound NCC(O)=O DHMQDGOQFOQNFH-UHFFFAOYSA-N 0.000 description 2
- HOKKHZGPKSLGJE-GSVOUGTGSA-N N-Methyl-D-aspartic acid Chemical compound CN[C@@H](C(O)=O)CC(O)=O HOKKHZGPKSLGJE-GSVOUGTGSA-N 0.000 description 2
- 230000036461 convulsion Effects 0.000 description 2
- 239000012738 dissolution medium Substances 0.000 description 2
- 238000005286 illumination Methods 0.000 description 2
- 238000000746 purification Methods 0.000 description 2
- 238000013112 stability test Methods 0.000 description 2
- 229940089285 vimpat Drugs 0.000 description 2
- 239000004471 Glycine Substances 0.000 description 1
- 108010052164 Sodium Channels Proteins 0.000 description 1
- 102000018674 Sodium Channels Human genes 0.000 description 1
- 238000002835 absorbance Methods 0.000 description 1
- 239000002671 adjuvant Substances 0.000 description 1
- 239000005557 antagonist Substances 0.000 description 1
- 239000001961 anticonvulsive agent Substances 0.000 description 1
- 230000007423 decrease Effects 0.000 description 1
- 206010012601 diabetes mellitus Diseases 0.000 description 1
- 238000011978 dissolution method Methods 0.000 description 1
- 238000005265 energy consumption Methods 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 230000002779 inactivation Effects 0.000 description 1
- 229940089422 lacosamide 200 mg Drugs 0.000 description 1
- 239000002609 medium Substances 0.000 description 1
- 208000004296 neuralgia Diseases 0.000 description 1
- 208000021722 neuropathic pain Diseases 0.000 description 1
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 238000002560 therapeutic procedure Methods 0.000 description 1
- 238000000870 ultraviolet spectroscopy Methods 0.000 description 1
Images
Landscapes
- Medicinal Preparation (AREA)
Abstract
The invention relates to a tablet for treating epilepsy and a preparation method for the tablet, in particular to a lacosamide tablet for treating epilepsy and a preparation method for the lacosamide tablet. The lacosamide tablet comprises lacosamide, an inclusion material, filler, disintegrant, lubricant and/or wetting agent, adhesive and a coating material. The main medicine lacosamide is coated by the inclusion material, and other auxiliary materials are added into the lacosamide, so that a tablet core is obtained, and a finished product is obtained after the tablet core is coated by the coating material. The obtained lacosamide tablet is relatively high in dissolution rate, medicine stability is greatly improved, and medicine-using risks are reduced.
Description
Technical field
The present invention relates to a kind of tablet for the treatment of epilepsy and preparation method thereof, be specifically related to a kind of scheme for lacosamide tablet for the treatment of epilepsy and preparation method thereof.
Background technology
Scheme for lacosamide (lacosamide), chemistry (R)-2-acetamido by name-N-benzyl-3-methoxy propionamide, that a kind of novel NMDA (NMDA) receptor glycine site is in conjunction with antagonist, belonging to the new class functional amino, is the anticonvulsant drug of the brand-new double mechanism of tool.It optionally promotes the slow inactivation in sodium channel and regulates the reaction mediating proteins-2 (CRMP-2) that subsides, and CRMP-2 may be slowly even the neuropathic pain that blocks epilepsy and alleviate diabetes.
Schwarz Fa Mu drugmaker of subsidiary (Schwarz Pharma) than company (UCB Pharma) when scheme for lacosamide is excellent by Belgium develops, commodity are called Vimpat by European Union Germany and UK and USA FDA approval listing respectively in August, 2008, September And October.The positive data of safety and curative effect shows in the III clinical trial phase: oral scheme for lacosamide 200mg/d and two groups of 400mg/d show than placebo group statistically significant and clinical relevant improvement.Used in the past 3 kinds of different antuepileptics still to add simultaneously with the generally fine tolerance of energy behind the scheme for lacosamide among out of contior patient.In the test of multicenter, double blinding, placebo, to compare with placebo, the convulsions rate of 200mg/d group reduces by 14.4% (P=0.02), and the convulsions rate of 400mg/d group reduces by 15.0% (P=0.03).The 400mg/d group obtains 50% remission rate person 40.5%, and placebo group is 25.8% (P=0.006).This medicine generally can fine tolerance.
But because scheme for lacosamide is slightly water-soluble and character is unstable, although at present commercially available scheme for lacosamide tablet dissolution is higher, stability is not good enough.
Therefore, be badly in need of clinically a kind of scheme for lacosamide tablet that when improving dissolution, greatly improves medicine stability now.
Summary of the invention
Primary and foremost purpose of the present invention provides a kind of scheme for lacosamide tablet for the treatment of epilepsy.
Another object of the present invention provides the purposes of scheme for lacosamide tablet in treatment epilepsy.
Another purpose of the present invention provides a kind of method for preparing the scheme for lacosamide tablet.
The scheme for lacosamide tablet for the treatment of epilepsy provided by the invention comprises the each component of following portions by weight proportioning:
(a) sheet core segment:
Scheme for lacosamide 30-200 weight portion;
Enclose material 10-100 weight portion;
Filler 80-250 weight portion;
Disintegrating agent 5-70 weight portion; With
Lubricant 0.5-4 weight portion;
Perhaps, sheet core segment:
Scheme for lacosamide 30-200 weight portion;
Enclose material 10-100 weight portion;
Filler 80-250 weight portion;
Disintegrating agent 5-70 weight portion;
Wetting agent or binding agent 40-180 weight portion; With
Lubricant 0.5-4 weight portion; And
(b) clothing layer segment:
Coating material 6-30 weight portion.
In the embodiment of first aspect, described tablet can comprise the each component of following portions by weight proportioning:
(a) sheet core segment:
Scheme for lacosamide 70-125 weight portion;
Enclose material 40-80 weight portion;
Filler 100-180 weight portion;
Disintegrating agent 8-60 weight portion; With
Lubricant 1-4 weight portion;
Perhaps, sheet core segment:
Scheme for lacosamide 70-125 weight portion;
Enclose material 40-80 weight portion;
Filler 100-180 weight portion;
Disintegrating agent 8-60 weight portion;
Wetting agent or binding agent 50-170 weight portion; With
Lubricant 1-4 weight portion; And
(b) clothing layer segment:
Coating material 10-25 weight portion.
In the embodiment of first aspect, described enclose material can be selected from cyclodextrin, lecithin and soybean phospholipid; Described filler can be selected from one or more in starch, lactose, microcrystalline Cellulose, micropowder silica gel and the pregelatinized Starch; Described disintegrating agent can be selected from one or more in polyvinylpolypyrrolidone, cross-linking sodium carboxymethyl cellulose, carboxymethyl starch sodium and the low-substituted hydroxypropyl cellulose; Described wetting agent or binding agent can be selected from water, ethanol, polyvinylpolypyrrolidone, arabic gum, starch and sodium carboxymethyl cellulose; Described lubricant can be selected from Pulvis Talci, magnesium stearate and stearic acid; And described coating material can be selected from the stomach dissolution type coating powder.In one embodiment, described enclose material can be cyclodextrin or soybean phospholipid, preferred cyclodextrin; Described filler can be in starch, microcrystalline Cellulose and the pregelatinized Starch one or more, preferred starch or microcrystalline Cellulose; Described disintegrating agent can be in polyvinylpolypyrrolidone, carboxymethyl starch sodium and the low-substituted hydroxypropyl cellulose one or more, the mixture of preferred polyvinylpolypyrrolidone and low-substituted hydroxypropyl cellulose, and described wetting agent or binding agent can be ethanol; Described lubricant can be magnesium stearate or Pulvis Talci; And described coating material can be the stomach dissolution type Opadry.In one embodiment, the mass ratio of polyvinylpolypyrrolidone and low-substituted hydroxypropyl cellulose can be 1: 2.
In the embodiment of first aspect, described label can be standby by direct powder compression or wet granule compression tablet legal system.
The method for preparing the scheme for lacosamide tablet provided by the invention can may further comprise the steps:
(a) using the enclose material that scheme for lacosamide is carried out enclose processes;
(b) enclose of gained is processed product and the abundant mix homogeneously of filler, disintegrating agent and lubricant;
(c) mixture that step (b) is obtained carries out direct powder compression, obtains label; With
(d) use coating material that the label that step (c) obtains is carried out coating, obtain described scheme for lacosamide tablet;
Perhaps, said method comprising the steps of:
(I) using the enclose material that scheme for lacosamide is carried out enclose processes;
(II) enclose of gained is processed product and filler and the abundant mix homogeneously of disintegrating agent;
(III) add wetting agent or binding agent and carry out wet granulation, drying;
(IV) the dried granule that step (III) is obtained carries out granulate, adds lubricant, product in the middle of obtaining;
(V) content of scheme for lacosamide in the middle product that obtain of detecting step (IV) is determined the weight of label according to content, tabletting obtains label; With
(VI) use coating material that the label that step (V) obtains is carried out coating, obtain described scheme for lacosamide tablet.
In the embodiment of the third aspect, the use enclose material in step (a) and the step (I) carries out enclose to scheme for lacosamide to be processed and can be undertaken by being selected from following mode: saturated water solution method, polishing, supercritical ultrasonics technology and spray drying method.In a preferred embodiment, the use enclose material in step (a) and the step (I) carries out enclose to scheme for lacosamide and processes and can be undertaken by polishing.
The scheme for lacosamide tablet for the treatment of epilepsy provided by the invention is compared with existing scheme for lacosamide tablet, has following advantage:
(1) scheme for lacosamide tablet of the present invention has obtained beyond thought effect aspect the medicine stability improving, and stability is better, has reduced the clinical application risk;
(2) safe and effective, the steady sources of the related adjuvant of scheme for lacosamide tablet of the present invention, the common and easy operating of related process equipment, energy consumption is lower, pollution-free in the preparation process;
(3) at present state's internal therapy epilepsy medicine is less and do not have the listing of scheme for lacosamide tablet, and scheme for lacosamide tablet provided by the present invention will enrich domestic clinical application.
The accompanying drawing summary
Fig. 1 is scheme for lacosamide tablet and the stripping curve of reference substance in the hydrochloric acid solution of pH 1.0 of expression the present invention preparation.
The specific embodiment
Below each embodiment further explain to of the present invention, as to further restriction of the present invention.The material that adopts among the embodiment is commercially available if no special instructions.
Embodiment 1:
Prescription: (specification 30mg, 1000)
Preparation technology:
1. the 60g cyclodextrin is dissolved in the 180ml purified water, the 30g scheme for lacosamide is dissolved in a small amount of 65-80 ℃ purified water, place grinding machine for grinding to become pasty state two kinds of solution, 50~60 ℃ of forced air dryings obtain samples, with this sample and 80g microcrystalline Cellulose, 6g low-substituted hydroxypropyl cellulose, 1.2g micropowder silica gel mix homogeneously;
2. the mixture that obtains in 1 is added an amount of purified water and prepare soft material, cross 20 mesh sieves and granulate, oven dry, granulate;
3. add 6g polyvinylpolypyrrolidone, 2g magnesium stearate mix homogeneously, tabletting;
4. coating, packing, full inspection and get final product.
Embodiment 2:
Prescription: (specification 50mg, 1000)
Preparation technology:
1. the 100g cyclodextrin is dissolved in the 300ml purified water, the 50g scheme for lacosamide is dissolved in a small amount of 65-80 ℃ purified water, place grinding machine for grinding to become pasty state two kinds of solution, 50~60 ℃ of forced air dryings obtain samples, with this sample and 50g lactose, 50g starch, 5g micropowder silica gel mix homogeneously;
2. the mixture that obtains in 1 is added that water prepares soft material in the purification, cross 20 mesh sieves and granulate, oven dry, granulate;
3. add 10g cross-linking sodium carboxymethyl cellulose, 3.0g Pulvis Talci mix homogeneously, tabletting;
4. coating, packing, full inspection and get final product.
Embodiment 3:
Prescription: (specification 70mg, 1000)
Preparation technology:
1. the 140g cyclodextrin is dissolved in the 420ml purified water, the 70g scheme for lacosamide is dissolved in a small amount of 65-80 ℃ purified water, place grinding machine for grinding to become pasty state two kinds of solution, 50~60 ℃ of forced air dryings obtain samples, with this sample and 140g microcrystalline Cellulose, 20g low-substituted hydroxypropyl cellulose, 10g micropowder silica gel mix homogeneously;
2. the mixture that obtains in 1 is added that water prepares soft material in the purification, cross 20 mesh sieves and granulate, oven dry, granulate;
3. add 8g polyvinylpolypyrrolidone, 2g magnesium stearate mix homogeneously, tabletting;
4. coating, packing, full inspection and get final product.
Embodiment 4:
Prescription: (specification 100mg, 1000)
Preparation technology:
1. the 100g cyclodextrin is dissolved in the 600ml purified water, the 100g scheme for lacosamide is dissolved in a small amount of 65-80 ℃ purified water, place grinding machine for grinding to become pasty state two kinds of solution, 50~60 ℃ of forced air dryings obtain samples, with this sample and 100g starch, 25g low-substituted hydroxypropyl cellulose, 15g micropowder silica gel mix homogeneously;
2. the mixture that obtains in 1 is added in the purified water and prepare soft material, cross 20 mesh sieves and granulate, oven dry, granulate;
3. add 25g polyvinylpolypyrrolidone, 5g magnesium stearate mix homogeneously, tabletting
4. coating, packing, full inspection and get final product.
Embodiment 5:
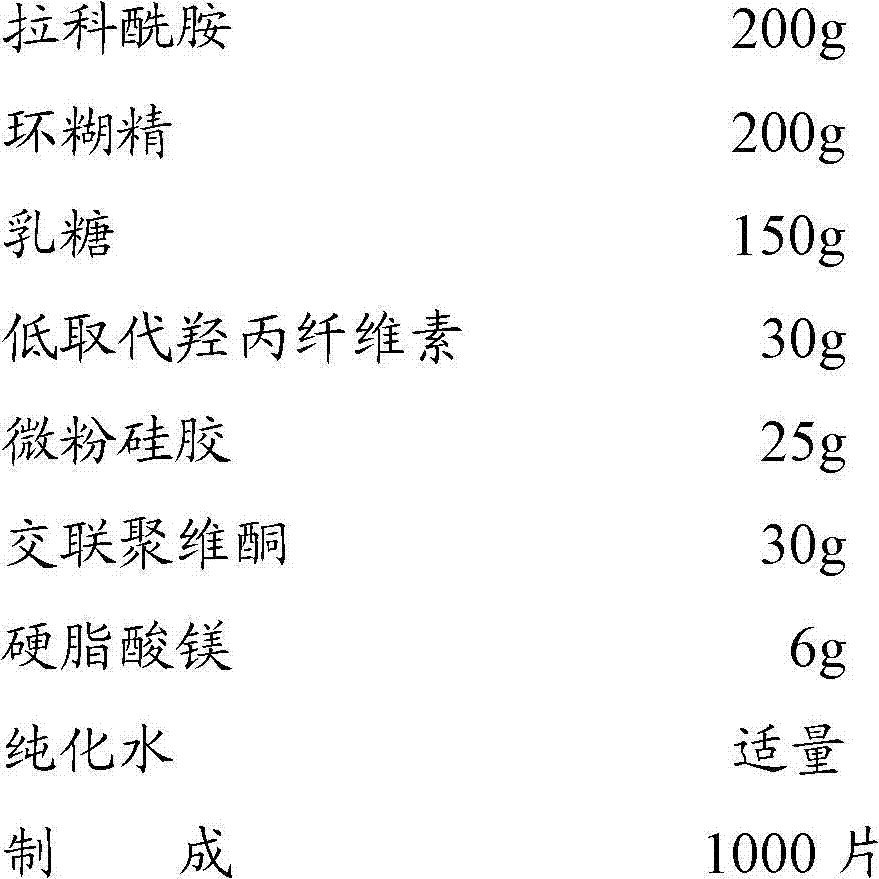
Prescription: (specification 200mg, 1000)
Preparation technology:
1. the 200g cyclodextrin is dissolved in the 600ml purified water, the 200g scheme for lacosamide is dissolved in a small amount of 65-80 ℃ purified water, place grinding machine for grinding to become pasty state two kinds of solution, 50~60 ℃ of forced air dryings obtain samples, with this sample and 150g lactose, 30g low-substituted hydroxypropyl cellulose, 25g micropowder silica gel mix homogeneously;
2. the mixture that obtains in 1 is added in the purified water and prepare soft material, cross 20 mesh sieves and granulate, oven dry, granulate;
3. add 30g polyvinylpolypyrrolidone, 6g magnesium stearate mix homogeneously, tabletting;
4. coating, packing, full inspection and get final product.
Estimate:
Test one:
The scheme for lacosamide tablet that embodiment 1-5 is made, according to dissolution method (two appendix XC the second methods of Chinese Pharmacopoeia version in 2010), 0.1mol/L hydrochloric acid solution 900ml is dissolution medium, rotating speed is that per minute 50 turns, in accordance with the law operation in the time of 10,15,20,30,45 minutes, is got solution 10ml (replenishing with the volume dissolution medium simultaneously), filter, as need testing solution.Other gets scheme for lacosamide reference substance [self-control tablet, lot number: LX110610-D], adds the solution that the stripping medium becomes 55.6 μ g/ml, and product solution adopts ultraviolet visible spectrophotometry to detect in contrast.By the stripping quantity of every of the ratio calculation of both absorbances, calculate the accumulative total stripping quantity.The results are shown in Table 1 and Fig. 1.
The sample that table 1 various embodiments of the present invention make and the reference substance stripping quantity in the hydrochloric acid solution of pH 1.0
| Time (min) | 10 | 15 | 20 | 30 | 45 |
| |
92.5 | 99.7 | 100.4 | 100.4 | 100.2 |
| |
89.8 | 100.1 | 100.1 | 100.2 | 100.2 |
| |
93.2 | 99.6 | 101.7 | 101.5 | 101.2 |
| |
90.7 | 100.3 | 100.5 | 100.4 | 100.5 |
| Embodiment 5 | 91.6 | 101.4 | 102.1 | 102.0 | 102.0 |
| Comparison film (100mg) | 90.7 | 100.5 | 100.5 | 100.4 | 100.4 |
Can find out by table 1 and Fig. 1, the prepared sample of the present invention is compared with control sample, and the stripping situation is all fine.
Test two
This test is carried out with reference to Chinese Pharmacopoeia (two appendix XIXC of version in 2010) stability test guideline, the sample that respectively embodiment 4 is made and control sample [scheme for lacosamide sheet (VIMPAT), Lot No.56090, UCB.lnc.] be placed under the environment of 60 ℃ of high temperature, illumination, high humidity 92.5%, detected respectively contrast at 0 day, the 5th day, the 10th day.The result sees Table respectively 2 and table 3:
Table 2 embodiment 4 gained sample effects factorial experiments results
Table 3 control sample influence factor result of the test
Can obviously find out by the above results, the prepared sample of the present invention is under influence factor's experimental condition, and each test item obviously is better than existing control formulation, under high humidity environment, control sample swells, and dissolution obviously descends, content decreases, and related substance increases.Under other conditions, all reflect the advantage of the prepared sample of the present invention such as illumination, high temperature.
Test three
This test is carried out with reference to Chinese Pharmacopoeia (two appendix XIXC of version in 2010) stability test guideline.In accelerated test, the sample that makes among the embodiment 4 is placed in the environment of 40 ℃ ± 2 ℃ of temperature, relative humidity 75% ± 5%, when 1 month, 2 months and 3 months, detect respectively; In long term test, the sample that makes among the embodiment 4 is placed in the environment of 25 ℃ ± 2 ℃ of temperature, relative humidity 60% ± 10%, in the time of 3 months, detect.The results are shown in Table 4:
The sample of table 4 embodiment 4 accelerates, long-term test results
Can find out by the above results, the prepared sample of the present invention accelerate, under the long term test condition, each test item is substantially unchanged, product stability is fine, can satisfy the requirement of clinical application.
Claims (10)
1. scheme for lacosamide tablet for the treatment of epilepsy, described tablet comprises the each component of following portions by weight proportioning:
(a) sheet core segment:
Scheme for lacosamide 30-200 weight portion;
Enclose material 10-100 weight portion;
Filler 80-250 weight portion;
Disintegrating agent 5-70 weight portion; With
Lubricant 0.5-4 weight portion;
Perhaps, sheet core segment:
Scheme for lacosamide 30-200 weight portion;
Enclose material 10-100 weight portion;
Filler 80-250 weight portion;
Disintegrating agent 5-70 weight portion;
Wetting agent or binding agent 40-180 weight portion; With
Lubricant 0.5-4 weight portion; And
(b) clothing layer segment:
Coating material 6-30 weight portion.
2. scheme for lacosamide tablet as claimed in claim 1, described tablet comprises the each component of following portions by weight proportioning:
(a) sheet core segment:
Scheme for lacosamide 70-125 weight portion;
Enclose material 40-80 weight portion;
Filler 100-180 weight portion;
Disintegrating agent 8-60 weight portion; With
Lubricant 1-4 weight portion;
Perhaps, sheet core segment:
Scheme for lacosamide 70-125 weight portion;
Enclose material 40-80 weight portion;
Filler 100-180 weight portion;
Disintegrating agent 8-60 weight portion;
Wetting agent or binding agent 50-170 weight portion; With
Lubricant 1-4 weight portion; And
(b) clothing layer segment:
Coating material 10-25 weight portion.
3. scheme for lacosamide tablet as claimed in claim 1 or 2, wherein said enclose material is selected from cyclodextrin, lecithin and soybean phospholipid; Described filler is selected from one or more in starch, lactose, microcrystalline Cellulose, micropowder silica gel and the pregelatinized Starch; Described disintegrating agent is selected from one or more in polyvinylpolypyrrolidone, cross-linking sodium carboxymethyl cellulose, carboxymethyl starch sodium and the low-substituted hydroxypropyl cellulose; Described wetting agent or binding agent are selected from water, ethanol, polyvinylpolypyrrolidone, arabic gum, starch and sodium carboxymethyl cellulose; Described lubricant is selected from Pulvis Talci, magnesium stearate and stearic acid; And described coating material is selected from the stomach dissolution type coating powder.
4. scheme for lacosamide tablet as claimed in claim 3, wherein said enclose material is cyclodextrin or soybean phospholipid, preferred cyclodextrin; Described filler is one or more in starch, microcrystalline Cellulose and the pregelatinized Starch, preferred starch or microcrystalline Cellulose; Described disintegrating agent is one or more in polyvinylpolypyrrolidone, carboxymethyl starch sodium and the low-substituted hydroxypropyl cellulose, the mixture of preferred polyvinylpolypyrrolidone and low-substituted hydroxypropyl cellulose, and described wetting agent or binding agent are ethanol; Described lubricant is magnesium stearate or Pulvis Talci; And described coating material is the stomach dissolution type Opadry.
5. scheme for lacosamide tablet as claimed in claim 4, wherein the mass ratio of polyvinylpolypyrrolidone and low-substituted hydroxypropyl cellulose is 1: 2.
6. scheme for lacosamide tablet as claimed in claim 1 or 2, wherein said label is standby by direct powder compression or wet granule compression tablet legal system.
In the claim 1 to 6 each described scheme for lacosamide tablet for the preparation of the treatment epilepsy medicine in purposes.
8. method for preparing such as each described scheme for lacosamide tablet in the claim 1 to 6 said method comprising the steps of:
(a) using the enclose material that scheme for lacosamide is carried out enclose processes;
(b) enclose of gained is processed product and the abundant mix homogeneously of filler, disintegrating agent and lubricant;
(c) mixture that step (b) is obtained carries out direct powder compression, obtains label; With
(d) use coating material that the label that step (c) obtains is carried out coating, obtain described scheme for lacosamide tablet;
Perhaps, said method comprising the steps of:
(I) using the enclose material that scheme for lacosamide is carried out enclose processes;
(II) enclose of gained is processed product and filler and the abundant mix homogeneously of disintegrating agent;
(III) add wetting agent or binding agent and carry out wet granulation, drying;
(IV) the dried granule that step (III) is obtained carries out granulate, adds lubricant, product in the middle of obtaining;
(V) content of scheme for lacosamide in the middle product that obtain of detecting step (IV) is determined the weight of label according to content, tabletting obtains label; With
(VI) use coating material that the label that step (V) obtains is carried out coating, obtain described scheme for lacosamide tablet.
9. method as claimed in claim 8, wherein the use enclose material in step (a) and the step (I) carries out enclose to scheme for lacosamide and processes and undertaken by being selected from following mode: saturated water solution method, polishing, supercritical ultrasonics technology and spray drying method.
10. method as claimed in claim 9, wherein the use enclose material in step (a) and the step (I) carries out enclose to scheme for lacosamide and processes and undertaken by polishing.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201210266439XA CN102885796A (en) | 2012-07-30 | 2012-07-30 | Lacosamide tablet for treating epilepsy and preparation method for lacosamide tablet |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201210266439XA CN102885796A (en) | 2012-07-30 | 2012-07-30 | Lacosamide tablet for treating epilepsy and preparation method for lacosamide tablet |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN102885796A true CN102885796A (en) | 2013-01-23 |
Family
ID=47529617
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201210266439XA Pending CN102885796A (en) | 2012-07-30 | 2012-07-30 | Lacosamide tablet for treating epilepsy and preparation method for lacosamide tablet |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN102885796A (en) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN104784134A (en) * | 2015-04-12 | 2015-07-22 | 石家庄四药有限公司 | Lacosamidesolid preparation and preparation method thereof |
| CN109010301A (en) * | 2018-09-05 | 2018-12-18 | 上海上药第生化药业有限公司 | A kind of lacosamide crystal form II tablet and its preparation method and application |
| CN112043681A (en) * | 2019-06-06 | 2020-12-08 | 上海奥科达生物医药科技有限公司 | Lacosamide pharmaceutical composition and pharmaceutical preparation thereof |
| US12478587B1 (en) | 2025-02-06 | 2025-11-25 | Shanghai Aucta Pharmaceuticals Co., Ltd. | Formulation for lacosamide |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2010083843A1 (en) * | 2009-01-26 | 2010-07-29 | Egalet A/S | Controlled release formulations with continuous efficacy |
| WO2010088911A1 (en) * | 2009-02-06 | 2010-08-12 | Egalet A/S | Pharmaceutical compositions resistant to abuse |
| WO2011008298A2 (en) * | 2009-07-16 | 2011-01-20 | Nectid, Inc. | Novel axomadol dosage forms |
| CN102020585A (en) * | 2009-09-15 | 2011-04-20 | 成都普康生物科技有限公司 | New preparation method of (R)-2-acetamido-N-benzyl-3-methoxy propionamide (lacosamide) |
| WO2011055385A1 (en) * | 2009-11-03 | 2011-05-12 | Lupin Limited | Modified release formulation of lacosamide |
| WO2011101863A2 (en) * | 2010-02-19 | 2011-08-25 | Cadila Healthcare Limited | Extended release pharmaceutical compositions of lacosamide |
| EP2468261A1 (en) * | 2010-12-02 | 2012-06-27 | UCB Pharma GmbH | Formulation of lacosamide |
-
2012
- 2012-07-30 CN CN201210266439XA patent/CN102885796A/en active Pending
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2010083843A1 (en) * | 2009-01-26 | 2010-07-29 | Egalet A/S | Controlled release formulations with continuous efficacy |
| WO2010088911A1 (en) * | 2009-02-06 | 2010-08-12 | Egalet A/S | Pharmaceutical compositions resistant to abuse |
| WO2011008298A2 (en) * | 2009-07-16 | 2011-01-20 | Nectid, Inc. | Novel axomadol dosage forms |
| CN102020585A (en) * | 2009-09-15 | 2011-04-20 | 成都普康生物科技有限公司 | New preparation method of (R)-2-acetamido-N-benzyl-3-methoxy propionamide (lacosamide) |
| WO2011055385A1 (en) * | 2009-11-03 | 2011-05-12 | Lupin Limited | Modified release formulation of lacosamide |
| WO2011101863A2 (en) * | 2010-02-19 | 2011-08-25 | Cadila Healthcare Limited | Extended release pharmaceutical compositions of lacosamide |
| EP2468261A1 (en) * | 2010-12-02 | 2012-06-27 | UCB Pharma GmbH | Formulation of lacosamide |
Cited By (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN104784134A (en) * | 2015-04-12 | 2015-07-22 | 石家庄四药有限公司 | Lacosamidesolid preparation and preparation method thereof |
| CN104784134B (en) * | 2015-04-12 | 2018-05-29 | 石家庄四药有限公司 | A kind of scheme for lacosamide solid pharmaceutical preparation and preparation method thereof |
| CN109010301A (en) * | 2018-09-05 | 2018-12-18 | 上海上药第生化药业有限公司 | A kind of lacosamide crystal form II tablet and its preparation method and application |
| CN109010301B (en) * | 2018-09-05 | 2021-01-26 | 上海上药第一生化药业有限公司 | Lacosamide crystal form II tablet and preparation method and application thereof |
| CN112043681A (en) * | 2019-06-06 | 2020-12-08 | 上海奥科达生物医药科技有限公司 | Lacosamide pharmaceutical composition and pharmaceutical preparation thereof |
| CN114404393A (en) * | 2019-06-06 | 2022-04-29 | 上海奥科达生物医药科技有限公司 | Lacosamide pharmaceutical composition and pharmaceutical preparation thereof |
| US11337943B2 (en) | 2019-06-06 | 2022-05-24 | Shanghai Aucta Pharmaceuticals Co., Ltd. | Lacosamide pharmaceutical composition and dosage form thereof |
| CN114404393B (en) * | 2019-06-06 | 2023-02-24 | 上海奥科达生物医药科技有限公司 | Lacosamide pharmaceutical composition and pharmaceutical preparation thereof |
| US11883374B2 (en) | 2019-06-06 | 2024-01-30 | Shanghai Aucta Pharmaceuticals Co., Ltd. | Lacosamide pharmaceutical composition and dosage form thereof |
| US12478587B1 (en) | 2025-02-06 | 2025-11-25 | Shanghai Aucta Pharmaceuticals Co., Ltd. | Formulation for lacosamide |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CA2805974C (en) | Use of binders for manufacturing storage stable formulations | |
| JP5636364B2 (en) | Preparation of sustained release skeletal muscle relaxant drug form | |
| CN102885796A (en) | Lacosamide tablet for treating epilepsy and preparation method for lacosamide tablet | |
| RS56570B1 (en) | Compositions and methods for overcoming resistance to tramadol | |
| JP6243351B2 (en) | Fixed dose drug combination therapy for Parkinson's disease | |
| CN104146978B (en) | A kind of disulfiram enteric coated tablet and preparation method thereof | |
| EP2361616A1 (en) | Extended release compositions for high solubility, high permeability active pharmaceutical ingredients | |
| CN105228608A (en) | Pharmaceutical composition capable of easily adjusting the dissolution profile of lacosamide or a pharmaceutically acceptable salt thereof | |
| CN101991559B (en) | Stable agomelatine capsule pharmaceutical composition | |
| Chattaraj et al. | Effect of formulation variables on dissolution profile of diclofenac sodium from ethyl-and hydroxypropylmethyl cellulose tablets | |
| CN104546781A (en) | Sevelamer carbonate tablet and preparation method thereof | |
| CN103405395A (en) | Sodium picosulfate enteric-coated tablet and preparation method thereof | |
| US11793852B2 (en) | Application of Longhu Rendan in preparing medicament for preventing and/or treating liver fibrosis | |
| WO2013158638A1 (en) | Stable dosage forms of skeletal muscle relaxants with extended release coating | |
| CN104666263B (en) | A kind of tablet containing Levetiracetam and preparation method thereof | |
| CN101574341B (en) | Oral solid medicine composition containing ropinirole | |
| CN102871981B (en) | Tablet medicine compound containing levetiracetam | |
| CN104382873A (en) | Mycophenolate mofetil dispersible tablet | |
| Dineshmohan et al. | Effect of HPMC and ethyl cellulose polymeric granules and its combinations in press coated tablets of lornoxicam: fabrication and in vitro characterization | |
| KR20140146095A (en) | Method of treating acne | |
| CN106692094A (en) | Rolapitant medicine oral preparation and preparation method thereof | |
| CN104337783B (en) | A kind of capecitabine tablet and preparation method thereof | |
| CN101112363A (en) | An in vivo cross-linked chip for colon-targeted drug delivery | |
| CN103006618A (en) | Anti-depression slow-release capsule | |
| Liang et al. | Preparation of colon-specific and synchronous release pellet containing total alkaloids of Sophora alopecuroides |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C12 | Rejection of a patent application after its publication | ||
| RJ01 | Rejection of invention patent application after publication |
Application publication date: 20130123 |