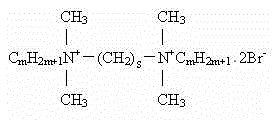CN102746396A - Cm-s-Cm Surfactant Reverse Micellar Extraction and Transfer Method of Bovine Serum Albumin - Google Patents
Cm-s-Cm Surfactant Reverse Micellar Extraction and Transfer Method of Bovine Serum Albumin Download PDFInfo
- Publication number
- CN102746396A CN102746396A CN2011104027300A CN201110402730A CN102746396A CN 102746396 A CN102746396 A CN 102746396A CN 2011104027300 A CN2011104027300 A CN 2011104027300A CN 201110402730 A CN201110402730 A CN 201110402730A CN 102746396 A CN102746396 A CN 102746396A
- Authority
- CN
- China
- Prior art keywords
- serum albumin
- bovine serum
- extraction
- reverse micelles
- type surfactant
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 108091003079 Bovine Serum Albumin Proteins 0.000 title claims abstract description 70
- 229940098773 bovine serum albumin Drugs 0.000 title claims abstract description 70
- 238000000605 extraction Methods 0.000 title claims abstract description 51
- 239000004094 surface-active agent Substances 0.000 title claims abstract description 46
- 238000000034 method Methods 0.000 title claims abstract description 27
- 239000000693 micelle Substances 0.000 claims abstract description 49
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims abstract description 20
- 238000009777 vacuum freeze-drying Methods 0.000 claims abstract description 3
- 239000000243 solution Substances 0.000 claims description 19
- 150000001335 aliphatic alkanes Chemical class 0.000 claims description 6
- 239000007853 buffer solution Substances 0.000 claims description 6
- 150000001298 alcohols Chemical class 0.000 claims description 5
- TVMXDCGIABBOFY-UHFFFAOYSA-N octane Chemical compound CCCCCCCC TVMXDCGIABBOFY-UHFFFAOYSA-N 0.000 claims description 5
- AMQJEAYHLZJPGS-UHFFFAOYSA-N N-Pentanol Chemical compound CCCCCO AMQJEAYHLZJPGS-UHFFFAOYSA-N 0.000 claims description 4
- 239000000872 buffer Substances 0.000 claims description 4
- KBPLFHHGFOOTCA-UHFFFAOYSA-N caprylic alcohol Natural products CCCCCCCCO KBPLFHHGFOOTCA-UHFFFAOYSA-N 0.000 claims description 4
- ZSIAUFGUXNUGDI-UHFFFAOYSA-N hexan-1-ol Chemical compound CCCCCCO ZSIAUFGUXNUGDI-UHFFFAOYSA-N 0.000 claims description 4
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims description 3
- OFBQJSOFQDEBGM-UHFFFAOYSA-N Pentane Chemical compound CCCCC OFBQJSOFQDEBGM-UHFFFAOYSA-N 0.000 claims description 3
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 claims description 3
- 239000012074 organic phase Substances 0.000 claims description 3
- 239000000843 powder Substances 0.000 claims description 3
- 239000007864 aqueous solution Substances 0.000 claims description 2
- 238000005119 centrifugation Methods 0.000 claims 1
- 150000003839 salts Chemical class 0.000 abstract description 12
- 230000000694 effects Effects 0.000 abstract description 5
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 6
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 6
- LZZYPRNAOMGNLH-UHFFFAOYSA-M Cetrimonium bromide Chemical compound [Br-].CCCCCCCCCCCCCCCC[N+](C)(C)C LZZYPRNAOMGNLH-UHFFFAOYSA-M 0.000 description 5
- 239000000284 extract Substances 0.000 description 5
- 239000008346 aqueous phase Substances 0.000 description 4
- 239000012454 non-polar solvent Substances 0.000 description 4
- 239000012071 phase Substances 0.000 description 4
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- 229910017053 inorganic salt Inorganic materials 0.000 description 3
- 238000002360 preparation method Methods 0.000 description 3
- 102000004169 proteins and genes Human genes 0.000 description 3
- 108090000623 proteins and genes Proteins 0.000 description 3
- DHMQDGOQFOQNFH-UHFFFAOYSA-N Glycine Chemical compound NCC(O)=O DHMQDGOQFOQNFH-UHFFFAOYSA-N 0.000 description 2
- VMHLLURERBWHNL-UHFFFAOYSA-M Sodium acetate Chemical compound [Na+].CC([O-])=O VMHLLURERBWHNL-UHFFFAOYSA-M 0.000 description 2
- 230000015572 biosynthetic process Effects 0.000 description 2
- 239000006184 cosolvent Substances 0.000 description 2
- BNIILDVGGAEEIG-UHFFFAOYSA-L disodium hydrogen phosphate Chemical compound [Na+].[Na+].OP([O-])([O-])=O BNIILDVGGAEEIG-UHFFFAOYSA-L 0.000 description 2
- 238000000746 purification Methods 0.000 description 2
- 239000001632 sodium acetate Substances 0.000 description 2
- 235000017281 sodium acetate Nutrition 0.000 description 2
- JHJLBTNAGRQEKS-UHFFFAOYSA-M sodium bromide Chemical compound [Na+].[Br-] JHJLBTNAGRQEKS-UHFFFAOYSA-M 0.000 description 2
- -1 2-ethylhexyl Chemical group 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- 239000004471 Glycine Substances 0.000 description 1
- WCUXLLCKKVVCTQ-UHFFFAOYSA-M Potassium chloride Chemical compound [Cl-].[K+] WCUXLLCKKVVCTQ-UHFFFAOYSA-M 0.000 description 1
- SWLVFNYSXGMGBS-UHFFFAOYSA-N ammonium bromide Chemical compound [NH4+].[Br-] SWLVFNYSXGMGBS-UHFFFAOYSA-N 0.000 description 1
- 230000000712 assembly Effects 0.000 description 1
- 238000000429 assembly Methods 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 238000011095 buffer preparation Methods 0.000 description 1
- 238000009792 diffusion process Methods 0.000 description 1
- CBMPTFJVXNIWHP-UHFFFAOYSA-L disodium;hydrogen phosphate;2-hydroxypropane-1,2,3-tricarboxylic acid Chemical compound [Na+].[Na+].OP([O-])([O-])=O.OC(=O)CC(O)(C(O)=O)CC(O)=O CBMPTFJVXNIWHP-UHFFFAOYSA-L 0.000 description 1
- 230000009881 electrostatic interaction Effects 0.000 description 1
- 238000004945 emulsification Methods 0.000 description 1
- 230000002708 enhancing effect Effects 0.000 description 1
- 238000004108 freeze drying Methods 0.000 description 1
- 238000000622 liquid--liquid extraction Methods 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 239000007974 sodium acetate buffer Substances 0.000 description 1
- BHZOKUMUHVTPBX-UHFFFAOYSA-M sodium acetic acid acetate Chemical compound [Na+].CC(O)=O.CC([O-])=O BHZOKUMUHVTPBX-UHFFFAOYSA-M 0.000 description 1
- 229940074404 sodium succinate Drugs 0.000 description 1
- ZDQYSKICYIVCPN-UHFFFAOYSA-L sodium succinate (anhydrous) Chemical compound [Na+].[Na+].[O-]C(=O)CCC([O-])=O ZDQYSKICYIVCPN-UHFFFAOYSA-L 0.000 description 1
- CIJQGPVMMRXSQW-UHFFFAOYSA-M sodium;2-aminoacetic acid;hydroxide Chemical compound O.[Na+].NCC([O-])=O CIJQGPVMMRXSQW-UHFFFAOYSA-M 0.000 description 1
- 238000000638 solvent extraction Methods 0.000 description 1
- BDHFUVZGWQCTTF-UHFFFAOYSA-M sulfonate Chemical compound [O-]S(=O)=O BDHFUVZGWQCTTF-UHFFFAOYSA-M 0.000 description 1
- 239000006228 supernatant Substances 0.000 description 1
Landscapes
- Peptides Or Proteins (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
Abstract
Cm-s-Cm型表面活性剂反胶束萃取转移牛血清蛋白的方法,属于生物工程中反胶束萃取技术领域。包括以下步骤:用Cm-s-Cm型表面活性剂配制反胶束;配制牛血清蛋白溶液;前萃取;后萃取;真空冷冻干燥得到牛血清蛋白产品。本发明用Cm-s-Cm型表面活性剂形成的反胶束对牛血清蛋白进行萃取(即前萃取),无需加入无机盐即可使水相的牛血清蛋白完全转移至反胶束;在后萃取阶段,即将牛血清蛋白从反胶束再次转移至水相的过程中,仅需调节水的pH值,无需加入无机盐,即可使BSA高效地富集在水相中。本方法操作简单,无需加入无机盐,表面活性剂用量少,牛血清蛋白的天然活性不受影响。The invention discloses a method for extracting and transferring bovine serum albumin by reverse micelles of Cm - sCm type surfactant, which belongs to the technical field of reverse micelles extraction in bioengineering. The method comprises the following steps: preparing reverse micelles with C m -sC m type surfactants; preparing bovine serum albumin solution; pre-extracting; post-extracting; vacuum freeze-drying to obtain bovine serum albumin products. The present invention uses reverse micelles formed by Cm - sCm type surfactants to extract bovine serum albumin (i.e. pre-extraction), and the bovine serum albumin in the water phase can be completely transferred to the reverse micelles without adding inorganic salts; In the post-extraction stage, that is, in the process of transferring bovine serum albumin from reverse micelles to the water phase again, it is only necessary to adjust the pH value of the water without adding inorganic salts, so that BSA can be efficiently enriched in the water phase. The method is simple to operate, does not need to add inorganic salts, uses less surfactant, and does not affect the natural activity of the bovine serum albumin.
Description
技术领域 technical field
本发明属于生物工程中反胶束萃取技术领域,特别涉及一种新型表面活性剂形成的反胶束萃取转移牛血清蛋白的方法。 The invention belongs to the technical field of reverse micelles extraction in bioengineering, in particular to a method for extracting and transferring bovine serum albumin by reverse micelles formed by a novel surfactant.
背景技术 Background technique
反胶束是表面活性剂在非极性溶剂中形成的一种分子有序组合体。它具有特殊的纳米空间——“水池”,该“水池”能将酶和蛋白质等增溶于其中,并为它们提供一个特殊的微环境,使其周围均为水分子,从而可以很好的保持其天然活性。 Reverse micelles are molecularly ordered assemblies of surfactants in non-polar solvents. It has a special nano-space - "water pool", which can solubilize enzymes and proteins in it, and provide them with a special microenvironment, so that they are surrounded by water molecules, so that they can be well maintain its natural activity.
反胶束对牛血清蛋白的液-液萃取是蛋白质分离、转移、富集和纯化的一种方法,包括前萃取(即将牛血清蛋白萃取至反胶束中)和后萃取(即将牛血清蛋白从反胶束再次转移至水相)两步。一般在反胶束萃取牛血清蛋白技术中用到的表面活性剂为1,4-二(2-乙基己基)丁二酸酯磺酸钠 (AOT) 和十六烷基三甲基溴化铵 (CTAB)。虽然AOT极易形成反胶束,但是它对牛血清蛋白进行萃取时,不仅用量大(0.16 M,即70 g/L),而且需要2 M无机盐(KCl)存在才能保证萃取效率。CTAB用于牛血清蛋白萃取时,虽然用量远远低于AOT(CTAB用量不能低于0.04 M,即14.5 g/L),但是仍然要求至少1.7 M的无机盐(NaBr)存在才能保证萃取效率。在无盐存在的情况下,这两种表面活性剂对牛血清蛋白的萃取效率接近于0。高浓度盐的存在不仅对牛血清蛋白的后续纯化带来不便,同时对工艺设备的要求也比较高,以防止盐对有关工艺设备造成腐蚀。此外,牛血清蛋白萃取过程中尚存在以下无法避免的不利因素:(1)AOT浓度高时易于使水相乳化,(2)CTAB在萃取过程中容易与牛血清蛋白形成白色不溶的复合物,(3)待萃取的牛血清蛋白含量不得高于6 g/L,否则萃取率会急剧降低。本发明将一种新型表面活性剂用于牛血清蛋白的萃取转移,不仅表面活性剂用量少(大于3 g/L即可),无需加入无机盐,且待萃取牛血清蛋白的含量可高至28 g/L。此外,本发明所用的表面活性剂在浓度达到90 g/L的情况下也不会导致水相乳化,亦不会与牛血清蛋白形成不溶复合物。 The liquid-liquid extraction of bovine serum albumin by reverse micelles is a method of protein separation, transfer, enrichment and purification, including pre-extraction (that is, bovine serum albumin is extracted into reverse micelles) and post-extraction (that is, bovine serum albumin Transfer from reverse micelles to aqueous phase again) in two steps. The surfactants generally used in reverse micellar extraction of bovine serum albumin are 1,4-di(2-ethylhexyl) sodium succinate sulfonate (AOT) and hexadecyltrimethyl bromide ammonium (CTAB). Although AOT is very easy to form reverse micelles, when it extracts bovine serum albumin, it not only needs a large amount (0.16 M, or 70 g/L), but also requires the presence of 2 M inorganic salt (KCl) to ensure the extraction efficiency. When CTAB is used for bovine serum albumin extraction, although the amount used is much lower than that of AOT (the amount of CTAB should not be lower than 0.04 M, that is, 14.5 g/L), it still requires at least 1.7 M of inorganic salt (NaBr) to ensure the extraction efficiency. In the absence of salt, the extraction efficiencies of these two surfactants for bovine serum albumin were close to zero. The presence of high-concentration salt not only brings inconvenience to the subsequent purification of bovine serum albumin, but also requires relatively high requirements on process equipment to prevent salt from corroding related process equipment. In addition, there are still the following unavoidable unfavorable factors in the extraction process of bovine serum albumin: (1) when the concentration of AOT is high, it is easy to emulsify the water phase, (2) CTAB is easy to form white insoluble complexes with bovine serum albumin during the extraction process, (3) The content of bovine serum albumin to be extracted shall not be higher than 6 g/L, otherwise the extraction rate will drop sharply. The present invention uses a new type of surfactant for the extraction and transfer of bovine serum albumin, not only the amount of surfactant is less (just greater than 3 g/L), no need to add inorganic salts, and the content of bovine serum albumin to be extracted can be high to 28 g/L. In addition, the surfactant used in the present invention will not cause emulsification of the water phase when the concentration reaches 90 g/L, nor will it form an insoluble complex with bovine serum albumin.
发明内容 Contents of the invention
本发明的目的是提供一种操作简单、表面活性剂用量少、无需无机盐即可萃取转移牛血清蛋白,并使牛血清蛋白保持天然活性的Cm-s-Cm型表面活性剂反胶萃取转移牛血清蛋白的方法。 The object of the present invention is to provide a Cm - sCm type surfactant reverse gel extraction with simple operation, less surfactant consumption, extraction and transfer of bovine serum albumin without inorganic salts, and keeping the natural activity of bovine serum albumin Method for transferring bovine serum albumin.
本发明方法是:将牛血清蛋白配制成待萃取的牛血清蛋白溶液后以反胶束进行前萃取,取得有机相后再用缓冲液进行后萃取,离心取得牛血清蛋白水溶液,再通过真空冷冻干燥得到牛血清蛋白干粉;本发明的特点是:采用Cm-s-Cm型表面活性剂配制反胶束。 The method of the invention is as follows: pre-extract the bovine serum albumin into the bovine serum albumin solution to be extracted with reverse micelles, obtain the organic phase and then use the buffer solution to perform post-extraction, centrifuge to obtain the bovine serum albumin aqueous solution, and then vacuum freeze drying to obtain bovine serum albumin dry powder; the feature of the invention is: adopting Cm - sCm type surfactant to prepare reverse micelles.
本发明用Cm-s-Cm型表面活性剂形成的反胶束对牛血清蛋白进行萃取(即前萃取),在萃取过程中仅需少量的表面活性剂,且在无盐的条件下就能使水相的牛血清蛋白完全转移至反胶束,此过程操作简单,萃取过程中,牛血清蛋白被反胶束内的水环境包围,能使BSA保持天然活性。在后萃取阶段,即将BSA从反胶束再次转移至水相的过程中,仅需调节溶液的pH值就能使牛血清蛋白转移并富集在水相中。本发明方法操作简单,无需无机盐,在表面活性剂用量极少的情况下即可高效地萃取转移牛血清蛋白,并能保持牛血清蛋白的天然活性。 The present invention uses reverse micelles formed by C m -sC m type surfactants to extract bovine serum albumin (i.e. pre-extraction), only a small amount of surfactant is needed in the extraction process, and it can be extracted under the condition of no salt The bovine serum albumin in the water phase is completely transferred to the reverse micelles. This process is simple to operate. During the extraction process, the bovine serum albumin is surrounded by the water environment in the reverse micelles, which can keep the natural activity of BSA. In the post-extraction stage, that is, in the process of transferring BSA from the reverse micelles to the aqueous phase again, the bovine serum albumin can be transferred and enriched in the aqueous phase only by adjusting the pH value of the solution. The method of the invention is simple to operate, does not need inorganic salts, can efficiently extract and transfer the bovine serum albumin under the condition that the amount of surfactant is very small, and can maintain the natural activity of the bovine serum albumin.
另,本发明所述反胶束由Cm-s-Cm型表面活性剂、正构烷烃、短链醇和水组成。 In addition, the reverse micelles of the present invention are composed of C m -sC m type surfactants, normal alkanes, short chain alcohols and water.
本发明所述Cm-s-Cm型表面活性剂结构式为: The structural formula of the Cm - sCm type surfactant of the present invention is:
其中,8≤ m≤ 16,s=8。 Among them, 8≤ m≤ 16, s=8.
适当增大表面活性剂浓度有利于提高前萃取效率,但是浓度太大可能会阻碍蛋白质的扩散,使前萃取效率反而降低。故所述反胶束中Cm-s-Cm型表面活性剂的浓度为0.003 g/ml ~ 0.09 g/ml。 Appropriately increasing the concentration of surfactant is beneficial to improve the efficiency of pre-extraction, but too high a concentration may hinder the diffusion of protein and reduce the efficiency of pre-extraction instead. Therefore, the concentration of the C m -sC m type surfactant in the reverse micelles is 0.003 g/ml-0.09 g/ml.
少量的助溶剂(即短链醇)有助于反胶束的形成,但助溶剂过多会影响溶液的极性以及反胶束的大小,导致前萃取效率降低。故本发明所述反胶束中正构烷烃和短链醇的体积比为12 ~ 5 : 1。 A small amount of co-solvent (that is, short-chain alcohol) is helpful for the formation of reverse micelles, but too much co-solvent will affect the polarity of the solution and the size of reverse micelles, resulting in a decrease in the efficiency of pre-extraction. Therefore, the volume ratio of normal alkanes and short-chain alcohols in the reverse micelles of the present invention is 12 to 5: 1.
水含量直接影响反胶束的大小,且水含量过小或过大都不利于反胶束的形成。故本发明所述反胶束中水与Cm-s-Cm型表面活性剂的摩尔比为5 ~ 20 : 1。 Water content directly affects the size of reverse micelles, and too small or too large water content is not conducive to the formation of reverse micelles. Therefore, the molar ratio of water to Cm - sCm type surfactant in the reverse micelles of the present invention is 5-20:1.
本发明所述正构烷烃为正戊烷、正己烷或正辛烷中的任意一种。 The n-alkane in the present invention is any one of n-pentane, n-hexane or n-octane.
本发明所述短链醇为正戊醇、正己醇或正辛醇中的任意一种。 The short-chain alcohol described in the present invention is any one of n-pentanol, n-hexanol or n-octanol.
由于牛血清蛋白的等电点为4.7,控制pH值大于其等电点,牛血清蛋白带负电,有利于增强它与本发明所述的表面活性剂之间的静电作用,提高前萃取效率。故本发明以pH为5 ~ 10的缓冲液溶解牛血清蛋白制成待萃取的牛血清蛋白溶液。 Because the isoelectric point of the bovine serum albumin is 4.7, the pH value is controlled to be greater than the isoelectric point, and the bovine serum albumin is negatively charged, which is conducive to enhancing the electrostatic interaction between it and the surfactant of the present invention and improving the pre-extraction efficiency. Therefore, the present invention dissolves bovine serum albumin in a buffer solution with a pH of 5 to 10 to make the bovine serum albumin solution to be extracted.
本发明后萃取所用缓冲液的pH为3.0 ~ 4.5。本发明将缓冲液的pH控制在小于BSA的等电点范围内,BSA带正电,从而导致它与反胶束内核中的表面活性剂头基之间发生静电排斥而被排出反胶束水核,有利于后萃取过程。 The pH of the buffer used in the post-extraction of the present invention is 3.0 ~ 4.5. In the present invention, the pH of the buffer is controlled within the range of the isoelectric point of less than BSA, and BSA is positively charged, thereby causing electrostatic repulsion between it and the surfactant head group in the reverse micelle core and being discharged from the reverse micelle water nuclei, which facilitates the post-extraction process.
具体实施方式 Detailed ways
1、配制反胶束: 1. Preparation of reverse micelles:
先将正构烷烃和短链醇以12 ~ 5 : 1的体积比混合,形成非极性溶剂。 Firstly, normal alkanes and short-chain alcohols are mixed at a volume ratio of 12 to 5:1 to form a non-polar solvent.
再将3 ~ 90 g Cm-s-Cm型表面活性剂溶于1 L 非极性溶剂中,再加入1 ~ 35 mL水,充分振荡至溶液澄清透明,即为反胶束。 Dissolve 3 ~ 90 g of C m -sC m type surfactant in 1 L of non-polar solvent, then add 1 ~ 35 mL of water, shake fully until the solution is clear and transparent, that is, reverse micelles.
表1 反胶束的配制 Table 1 Preparation of reverse micelles
2、配制待萃取的牛血清蛋白溶液: 2. Prepare the bovine serum albumin solution to be extracted:
缓冲液的配制: Buffer preparation:
用磷酸氢二钠和柠檬酸或醋酸和醋酸钠,配pH范围为4.7~8的磷酸氢二钠-柠檬酸缓冲液(0.01 M)。 Use disodium hydrogen phosphate and citric acid or acetic acid and sodium acetate to prepare disodium hydrogen phosphate-citric acid buffer solution (0.01 M) with a pH range of 4.7~8.
用甘氨酸和氢氧化钠,配pH范围为9~10的甘氨酸-氢氧化钠缓冲液(0.01 M)。 Glycine and sodium hydroxide are used to prepare glycine-sodium hydroxide buffer (0.01 M) with a pH range of 9-10.
取以上两种缓冲液分别溶解牛血清蛋白,形成待萃取的牛血清蛋白溶液。 Take the above two buffer solutions to dissolve the bovine serum albumin respectively to form the bovine serum albumin solution to be extracted.
3、前萃取 3. Pre-extraction
将以上配制的反胶束与待萃取的牛血清蛋白溶液经涡旋震荡10 min以及14000 rpm下离心15 min后,取上层反胶束清液。 The above-prepared reverse micelles and the bovine serum albumin solution to be extracted were vortexed for 10 min and centrifuged at 14000 rpm for 15 min, and then the supernatant of reverse micelles was taken.
通过以下公式分别计算前萃取的效率: The efficiency of the pre-extraction was calculated respectively by the following formula:
前萃取情况表 Pre-extraction table
本方法无需在体系中额外加入无机盐即可使牛血清蛋白完全萃取至有机相,这是其它表面活性剂无法做到的。 The method can completely extract the bovine serum albumin into the organic phase without adding additional inorganic salts to the system, which cannot be achieved by other surfactants.
(1)如用其它表面活性剂如CTAB或AOT,若使前萃取效率达到100 %,待萃取牛血清蛋白溶液中牛血清蛋白的浓度不得高于6 mg/ml;(2)本发明中,当待萃取牛血清蛋白溶液中牛血清蛋白浓度高于表格中所给数值后,本方法所用的反胶束不能保证牛血清蛋白100 %的从水相萃取到反胶束 (1) If other surfactants such as CTAB or AOT are used, if the pre-extraction efficiency reaches 100%, the concentration of bovine serum albumin in the bovine serum albumin solution to be extracted shall not be higher than 6 mg/ml; (2) in the present invention, When the bovine serum albumin concentration in the bovine serum albumin solution to be extracted is higher than the value given in the table, the reverse micelles used in this method cannot guarantee that the bovine serum albumin is 100% extracted from the aqueous phase to the reverse micelles
4、后萃取 4. Post-extraction
后萃液的配制:用磷酸氢二钠和柠檬酸或醋酸和醋酸钠,配pH范围为3 ~ 4.5的醋酸-醋酸钠缓冲液(0.01 M),该溶液即为后萃液。 Preparation of post-extraction: Use disodium hydrogen phosphate and citric acid or acetic acid and sodium acetate to prepare acetic acid-sodium acetate buffer (0.01 M) with a pH range of 3 to 4.5, and this solution is the post-extraction.
按体积比为1:1,将后萃液与步骤2所得的反胶束溶液相混合,涡旋震荡10 min并于14000 rpm下离心15 min后,取下层牛血清蛋白溶液。 According to the volume ratio of 1:1, the post-extraction was mixed with the reverse micellar solution obtained in step 2, vortexed for 10 min and centrifuged at 14000 rpm for 15 min, and then the lower bovine serum albumin solution was removed.
通过以下公式计算后萃取的效率: The efficiency of post-extraction was calculated by the following formula:
后萃取情况表 Post-extraction table
本方法无需在体系中额外加入无机盐即可使牛血清蛋白高效的从反胶束中萃取到水相。如不加入无机盐,其它表面活性剂对BSA的后萃取率为0 。 The method can efficiently extract the bovine serum albumin from the reverse micelles to the water phase without adding additional inorganic salts to the system. If no inorganic salt is added, the post-extraction rate of BSA by other surfactants is 0.
5、冷冻干燥 5. Freeze drying
将步骤4所得的牛血清蛋白溶液,在压强为50 Pa、温度为-60℃下冷冻干燥36 h,得到纯牛血清蛋白干粉。 The bovine serum albumin solution obtained in step 4 was freeze-dried at a pressure of 50 Pa and a temperature of -60°C for 36 h to obtain a dry powder of pure bovine serum albumin.
Claims (9)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2011104027300A CN102746396A (en) | 2011-12-07 | 2011-12-07 | Cm-s-Cm Surfactant Reverse Micellar Extraction and Transfer Method of Bovine Serum Albumin |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2011104027300A CN102746396A (en) | 2011-12-07 | 2011-12-07 | Cm-s-Cm Surfactant Reverse Micellar Extraction and Transfer Method of Bovine Serum Albumin |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN102746396A true CN102746396A (en) | 2012-10-24 |
Family
ID=47026931
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN2011104027300A Pending CN102746396A (en) | 2011-12-07 | 2011-12-07 | Cm-s-Cm Surfactant Reverse Micellar Extraction and Transfer Method of Bovine Serum Albumin |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN102746396A (en) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN105399650A (en) * | 2015-12-16 | 2016-03-16 | 齐鲁工业大学 | Method for extracting alliin through reverse micelles system |
| US11604026B2 (en) | 2019-03-14 | 2023-03-14 | Terumo Bct Biotechnologies, Llc | Lyophilization loading tray assembly and system |
| US11634257B2 (en) | 2017-10-09 | 2023-04-25 | Terumo Bct Biotechnologies, Llc | Lyophilization container and method of using same |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1966137A (en) * | 2005-11-22 | 2007-05-23 | 华东理工大学 | Gemini surface active agent |
| CN101348779A (en) * | 2008-09-12 | 2009-01-21 | 南昌市浩然生物医药有限公司 | Method for extracting purified elastase from pig pancreas |
| CN101554569A (en) * | 2009-04-21 | 2009-10-14 | 西安楚龙达化工有限公司 | Anionic type Gemini surface active agent and synthesizing method |
-
2011
- 2011-12-07 CN CN2011104027300A patent/CN102746396A/en active Pending
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1966137A (en) * | 2005-11-22 | 2007-05-23 | 华东理工大学 | Gemini surface active agent |
| CN101348779A (en) * | 2008-09-12 | 2009-01-21 | 南昌市浩然生物医药有限公司 | Method for extracting purified elastase from pig pancreas |
| CN101554569A (en) * | 2009-04-21 | 2009-10-14 | 西安楚龙达化工有限公司 | Anionic type Gemini surface active agent and synthesizing method |
Non-Patent Citations (3)
| Title |
|---|
| 19941231: "反胶束液-液萃取牛血清白蛋白的动力学研究 I.萃取过程", 《华东理工大学学报》, vol. 20, no. 6, 31 December 1994 (1994-12-31), pages 723 - 729 * |
| 肖静等: "Gemini表面活性剂/正己烷/正己醇/水反胶束体系萃取牛血清蛋白的研究", 《中国化学会第十三届胶体与界面化学会议论文摘要集》, 20 July 2011 (2011-07-20) * |
| 陆强等: "从反胶束溶液中反萃牛血清白蛋白的研究", 《生物工程学报》, vol. 12, no. 2, 23 April 1996 (1996-04-23) * |
Cited By (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN105399650A (en) * | 2015-12-16 | 2016-03-16 | 齐鲁工业大学 | Method for extracting alliin through reverse micelles system |
| US11634257B2 (en) | 2017-10-09 | 2023-04-25 | Terumo Bct Biotechnologies, Llc | Lyophilization container and method of using same |
| US11604026B2 (en) | 2019-03-14 | 2023-03-14 | Terumo Bct Biotechnologies, Llc | Lyophilization loading tray assembly and system |
| US11609043B2 (en) | 2019-03-14 | 2023-03-21 | Terumo Bct Biotechnologies, Llc | Lyophilization container fill fixture, system and method of use |
| US11609042B2 (en) | 2019-03-14 | 2023-03-21 | Terumo Bct Biotechnologies, Llc | Multi-part lyophilization container and method of use |
| US11740019B2 (en) | 2019-03-14 | 2023-08-29 | Terumo Bct Biotechnologies, Llc | Lyophilization loading tray assembly and system |
| US11747082B2 (en) | 2019-03-14 | 2023-09-05 | Terumo Bct Biotechnologies, Llc | Multi-part lyophilization container and method of use |
| US11815311B2 (en) | 2019-03-14 | 2023-11-14 | Terumo Bct Biotechnologies, Llc | Lyophilization container fill fixture, system and method of use |
| US11994343B2 (en) | 2019-03-14 | 2024-05-28 | Terumo Bct Biotechnologies, Llc | Multi-part lyophilization container and method of use |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| RU2009117170A (en) | DRIED RESTORED VESICULES FOR PHARMACEUTICAL USE | |
| CN103735532A (en) | Lutein ester microcapsule and preparation method thereof | |
| BR112017000805B1 (en) | METHOD FOR PREPARING A PROTEIN ISOLATE FROM MICROALGAE BIOMASS OF THE GENUS CHLORELLA | |
| CN113698506B (en) | Temperature response type eutectic solvent and method for extracting lycium barbarum polysaccharide | |
| CN102746396A (en) | Cm-s-Cm Surfactant Reverse Micellar Extraction and Transfer Method of Bovine Serum Albumin | |
| CN109956856B (en) | Method for extracting coenzyme Q10 and phospholipid from coenzyme Q10 fermentation strain powder | |
| CN103951739B (en) | A kind of method of composite reverse micelle system purifying cottonseed protein | |
| CN103988975A (en) | Method for extracting protein from soybean meal by adopting ultrasonic-assisted reverse micelle extraction process | |
| CN107412546A (en) | A kind of method of Lilium lancifo1ium Thunb extraction saponin(e | |
| Hemavathi et al. | Reverse micellar extraction of bromelain from Ananas comosus L. Merryl | |
| FR2495939A1 (en) | PROCESS FOR PURIFYING POLYOSIDES OF STREPTOCOCCUS PNEUMONIAE AND VACCINE BASED ON POLYOSIDES SO PURIFIED | |
| US2488565A (en) | Preparation of hyaluronidase | |
| CN103343114B (en) | A kind of method preparing high vigor pancreatin | |
| US2488564A (en) | Preparation of hyaluronidase | |
| CN114052253A (en) | Method for preparing lycium barbarum zeaxanthin microcapsules by lycium barbarum polysaccharide composite wall material | |
| CN105294822A (en) | Process for extracting rapeseed protein | |
| CN117756959B (en) | A method for preparing streptococcus pneumoniae capsular polysaccharide | |
| CN102718854A (en) | Method for extracting and transferring haemocyanin under neutral and alkaline conditions | |
| CN101619099B (en) | Method for extracting rapeseed protein from reverse micelles | |
| CN104531643A (en) | Method for separating and purifying of bromelain from pineapple peels | |
| CN101360762A (en) | Water-soluble polysaccharides derived from rice bran, process for producing the same, and emulsifier using the same | |
| CN106317234A (en) | Nostoc commune polysaccharide extraction method | |
| CN104338117A (en) | Preparation method of bivalirudin for injection | |
| CN105920591B (en) | A kind of preparation method of bright mung bean superoxide dismutase liposome | |
| CN121182736A (en) | Extraction method and application of exosomes |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C02 | Deemed withdrawal of patent application after publication (patent law 2001) | ||
| WD01 | Invention patent application deemed withdrawn after publication |
Application publication date: 20121024 |