CN102727468B - Dry powder inhaler of interferon alpha - Google Patents
Dry powder inhaler of interferon alpha Download PDFInfo
- Publication number
- CN102727468B CN102727468B CN201210235912.8A CN201210235912A CN102727468B CN 102727468 B CN102727468 B CN 102727468B CN 201210235912 A CN201210235912 A CN 201210235912A CN 102727468 B CN102727468 B CN 102727468B
- Authority
- CN
- China
- Prior art keywords
- interferon
- formoterol fumarate
- alpha
- foradil aerolizer
- aerolizer formoterol
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 108010047761 Interferon-alpha Proteins 0.000 title claims abstract description 85
- 102000006992 Interferon-alpha Human genes 0.000 title claims abstract description 85
- 229940112141 dry powder inhaler Drugs 0.000 title abstract 8
- 239000003223 protective agent Substances 0.000 claims abstract description 10
- 239000003085 diluting agent Substances 0.000 claims abstract description 9
- 239000002245 particle Substances 0.000 claims abstract description 6
- OBRNDARFFFHCGE-QDSVTUBZSA-N arformoterol fumarate Chemical compound OC(=O)\C=C\C(O)=O.C1=CC(OC)=CC=C1C[C@@H](C)NC[C@H](O)C1=CC=C(O)C(NC=O)=C1.C1=CC(OC)=CC=C1C[C@@H](C)NC[C@H](O)C1=CC=C(O)C(NC=O)=C1 OBRNDARFFFHCGE-QDSVTUBZSA-N 0.000 claims description 160
- 229960000193 formoterol fumarate Drugs 0.000 claims description 81
- 230000000694 effects Effects 0.000 claims description 30
- 239000000243 solution Substances 0.000 claims description 15
- 230000033228 biological regulation Effects 0.000 claims description 13
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 claims description 11
- 239000008101 lactose Substances 0.000 claims description 11
- 239000003795 chemical substances by application Substances 0.000 claims description 10
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 claims description 9
- 229930195725 Mannitol Natural products 0.000 claims description 9
- 108010010648 interferon alfacon-1 Proteins 0.000 claims description 9
- 229960003358 interferon alfacon-1 Drugs 0.000 claims description 9
- 239000000594 mannitol Substances 0.000 claims description 9
- 235000010355 mannitol Nutrition 0.000 claims description 9
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 claims description 8
- DHMQDGOQFOQNFH-UHFFFAOYSA-N Glycine Chemical compound NCC(O)=O DHMQDGOQFOQNFH-UHFFFAOYSA-N 0.000 claims description 8
- ROHFNLRQFUQHCH-YFKPBYRVSA-N L-leucine Chemical compound CC(C)C[C@H](N)C(O)=O ROHFNLRQFUQHCH-YFKPBYRVSA-N 0.000 claims description 8
- ROHFNLRQFUQHCH-UHFFFAOYSA-N Leucine Natural products CC(C)CC(N)C(O)=O ROHFNLRQFUQHCH-UHFFFAOYSA-N 0.000 claims description 8
- 239000012752 auxiliary agent Substances 0.000 claims description 8
- 102000009027 Albumins Human genes 0.000 claims description 4
- 108010088751 Albumins Proteins 0.000 claims description 4
- 239000004471 Glycine Substances 0.000 claims description 4
- COLNVLDHVKWLRT-QMMMGPOBSA-N L-phenylalanine Chemical compound OC(=O)[C@@H](N)CC1=CC=CC=C1 COLNVLDHVKWLRT-QMMMGPOBSA-N 0.000 claims description 4
- AYFVYJQAPQTCCC-GBXIJSLDSA-N L-threonine Chemical compound C[C@@H](O)[C@H](N)C(O)=O AYFVYJQAPQTCCC-GBXIJSLDSA-N 0.000 claims description 4
- AYFVYJQAPQTCCC-UHFFFAOYSA-N Threonine Natural products CC(O)C(N)C(O)=O AYFVYJQAPQTCCC-UHFFFAOYSA-N 0.000 claims description 4
- 239000004473 Threonine Substances 0.000 claims description 4
- 235000001014 amino acid Nutrition 0.000 claims description 4
- 150000001413 amino acids Chemical class 0.000 claims description 4
- COLNVLDHVKWLRT-UHFFFAOYSA-N phenylalanine Natural products OC(=O)C(N)CC1=CC=CC=C1 COLNVLDHVKWLRT-UHFFFAOYSA-N 0.000 claims description 4
- JLPULHDHAOZNQI-ZTIMHPMXSA-N 1-hexadecanoyl-2-(9Z,12Z-octadecadienoyl)-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCCCCCC\C=C/C\C=C/CCCCC JLPULHDHAOZNQI-ZTIMHPMXSA-N 0.000 claims description 3
- OUYCCCASQSFEME-QMMMGPOBSA-N L-tyrosine Chemical compound OC(=O)[C@@H](N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-QMMMGPOBSA-N 0.000 claims description 3
- 239000004472 Lysine Substances 0.000 claims description 3
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Natural products NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 claims description 3
- 229910019142 PO4 Inorganic materials 0.000 claims description 3
- 239000007853 buffer solution Substances 0.000 claims description 3
- 239000008366 buffered solution Substances 0.000 claims description 3
- 239000007979 citrate buffer Substances 0.000 claims description 3
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 claims description 3
- 239000010452 phosphate Substances 0.000 claims description 3
- 229940083466 soybean lecithin Drugs 0.000 claims description 3
- OUYCCCASQSFEME-UHFFFAOYSA-N tyrosine Natural products OC(=O)C(N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-UHFFFAOYSA-N 0.000 claims description 3
- 150000003904 phospholipids Chemical class 0.000 claims description 2
- 125000003289 ascorbyl group Chemical class [H]O[C@@]([H])(C([H])([H])O*)[C@@]1([H])OC(=O)C(O*)=C1O* 0.000 claims 1
- 102000014150 Interferons Human genes 0.000 abstract description 16
- 108010050904 Interferons Proteins 0.000 abstract description 16
- 229940079322 interferon Drugs 0.000 abstract description 16
- 239000002131 composite material Substances 0.000 abstract description 3
- 102000004169 proteins and genes Human genes 0.000 abstract description 3
- 108090000623 proteins and genes Proteins 0.000 abstract description 3
- 239000000546 pharmaceutical excipient Substances 0.000 abstract 2
- 239000000654 additive Substances 0.000 abstract 1
- 230000000996 additive effect Effects 0.000 abstract 1
- 239000000825 pharmaceutical preparation Substances 0.000 abstract 1
- 230000000087 stabilizing effect Effects 0.000 abstract 1
- 239000008187 granular material Substances 0.000 description 30
- 238000000034 method Methods 0.000 description 22
- 238000005507 spraying Methods 0.000 description 22
- 238000001035 drying Methods 0.000 description 11
- 239000007789 gas Substances 0.000 description 11
- 239000007921 spray Substances 0.000 description 11
- 201000010099 disease Diseases 0.000 description 9
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 9
- 239000003814 drug Substances 0.000 description 9
- 238000002360 preparation method Methods 0.000 description 8
- 238000001694 spray drying Methods 0.000 description 8
- 238000006243 chemical reaction Methods 0.000 description 6
- 239000000463 material Substances 0.000 description 6
- 239000000203 mixture Substances 0.000 description 6
- 208000009421 viral pneumonia Diseases 0.000 description 6
- 206010035737 Pneumonia viral Diseases 0.000 description 5
- 238000000889 atomisation Methods 0.000 description 5
- 239000002775 capsule Substances 0.000 description 5
- 238000002347 injection Methods 0.000 description 5
- 239000007924 injection Substances 0.000 description 5
- 210000002345 respiratory system Anatomy 0.000 description 5
- 239000002552 dosage form Substances 0.000 description 4
- 229940098458 powder spray Drugs 0.000 description 4
- 230000002685 pulmonary effect Effects 0.000 description 4
- 238000004458 analytical method Methods 0.000 description 3
- 230000004071 biological effect Effects 0.000 description 3
- 238000002144 chemical decomposition reaction Methods 0.000 description 3
- 239000000470 constituent Substances 0.000 description 3
- 238000011049 filling Methods 0.000 description 3
- 238000005457 optimization Methods 0.000 description 3
- 238000011160 research Methods 0.000 description 3
- 238000013112 stability test Methods 0.000 description 3
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 2
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 2
- 101000959820 Homo sapiens Interferon alpha-1/13 Proteins 0.000 description 2
- 102000008100 Human Serum Albumin Human genes 0.000 description 2
- 108091006905 Human Serum Albumin Proteins 0.000 description 2
- 102100040019 Interferon alpha-1/13 Human genes 0.000 description 2
- 241001465754 Metazoa Species 0.000 description 2
- 241000475481 Nebula Species 0.000 description 2
- 241000700605 Viruses Species 0.000 description 2
- 239000002671 adjuvant Substances 0.000 description 2
- 239000006185 dispersion Substances 0.000 description 2
- 238000004090 dissolution Methods 0.000 description 2
- 238000005516 engineering process Methods 0.000 description 2
- 238000009472 formulation Methods 0.000 description 2
- 108010002492 human interferon alfa-1b Proteins 0.000 description 2
- 102000000707 human interferon alfa-1b Human genes 0.000 description 2
- 230000002401 inhibitory effect Effects 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- 238000012423 maintenance Methods 0.000 description 2
- 238000012545 processing Methods 0.000 description 2
- 235000018102 proteins Nutrition 0.000 description 2
- 238000013441 quality evaluation Methods 0.000 description 2
- 238000005070 sampling Methods 0.000 description 2
- 150000005846 sugar alcohols Polymers 0.000 description 2
- 231100000419 toxicity Toxicity 0.000 description 2
- 230000001988 toxicity Effects 0.000 description 2
- HDTRYLNUVZCQOY-UHFFFAOYSA-N α-D-glucopyranosyl-α-D-glucopyranoside Natural products OC1C(O)C(O)C(CO)OC1OC1C(O)C(O)C(O)C(CO)O1 HDTRYLNUVZCQOY-UHFFFAOYSA-N 0.000 description 1
- IIZPXYDJLKNOIY-JXPKJXOSSA-N 1-palmitoyl-2-arachidonoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCC\C=C/C\C=C/C\C=C/C\C=C/CCCCC IIZPXYDJLKNOIY-JXPKJXOSSA-N 0.000 description 1
- 229920000858 Cyclodextrin Polymers 0.000 description 1
- 102000004127 Cytokines Human genes 0.000 description 1
- 108090000695 Cytokines Proteins 0.000 description 1
- 208000035240 Disease Resistance Diseases 0.000 description 1
- -1 Foradil Aerolizer formoterol fumarate Chemical class 0.000 description 1
- 102100040018 Interferon alpha-2 Human genes 0.000 description 1
- 108010079944 Interferon-alpha2b Proteins 0.000 description 1
- 150000000996 L-ascorbic acids Chemical class 0.000 description 1
- KDXKERNSBIXSRK-YFKPBYRVSA-N L-lysine Chemical compound NCCCC[C@H](N)C(O)=O KDXKERNSBIXSRK-YFKPBYRVSA-N 0.000 description 1
- HDTRYLNUVZCQOY-WSWWMNSNSA-N Trehalose Natural products O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@H](O)[C@@H](O)[C@@H](O)[C@@H](CO)O1 HDTRYLNUVZCQOY-WSWWMNSNSA-N 0.000 description 1
- TVXBFESIOXBWNM-UHFFFAOYSA-N Xylitol Natural products OCCC(O)C(O)C(O)CCO TVXBFESIOXBWNM-UHFFFAOYSA-N 0.000 description 1
- 229940124532 absorption promoter Drugs 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- 239000003570 air Substances 0.000 description 1
- HDTRYLNUVZCQOY-LIZSDCNHSA-N alpha,alpha-trehalose Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 HDTRYLNUVZCQOY-LIZSDCNHSA-N 0.000 description 1
- 239000012080 ambient air Substances 0.000 description 1
- 230000000844 anti-bacterial effect Effects 0.000 description 1
- 230000002155 anti-virotic effect Effects 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 229940072107 ascorbate Drugs 0.000 description 1
- 235000010323 ascorbic acid Nutrition 0.000 description 1
- 239000011668 ascorbic acid Substances 0.000 description 1
- 230000037396 body weight Effects 0.000 description 1
- 239000000337 buffer salt Substances 0.000 description 1
- 238000011443 conventional therapy Methods 0.000 description 1
- 235000012343 cottonseed oil Nutrition 0.000 description 1
- 238000002425 crystallisation Methods 0.000 description 1
- 230000008025 crystallization Effects 0.000 description 1
- 239000002274 desiccant Substances 0.000 description 1
- 229910001873 dinitrogen Inorganic materials 0.000 description 1
- 238000009826 distribution Methods 0.000 description 1
- 229940079593 drug Drugs 0.000 description 1
- 238000001647 drug administration Methods 0.000 description 1
- 230000000857 drug effect Effects 0.000 description 1
- 239000003623 enhancer Substances 0.000 description 1
- 230000008014 freezing Effects 0.000 description 1
- 238000007710 freezing Methods 0.000 description 1
- 239000000787 lecithin Substances 0.000 description 1
- 229940067606 lecithin Drugs 0.000 description 1
- 235000010445 lecithin Nutrition 0.000 description 1
- 238000011068 loading method Methods 0.000 description 1
- 230000014759 maintenance of location Effects 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 230000010534 mechanism of action Effects 0.000 description 1
- HEBKCHPVOIAQTA-UHFFFAOYSA-N meso ribitol Natural products OCC(O)C(O)C(O)CO HEBKCHPVOIAQTA-UHFFFAOYSA-N 0.000 description 1
- 230000004060 metabolic process Effects 0.000 description 1
- 210000004877 mucosa Anatomy 0.000 description 1
- 229910052756 noble gas Inorganic materials 0.000 description 1
- 239000003961 penetration enhancing agent Substances 0.000 description 1
- 230000035479 physiological effects, processes and functions Effects 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 238000003825 pressing Methods 0.000 description 1
- 230000002265 prevention Effects 0.000 description 1
- 230000029058 respiratory gaseous exchange Effects 0.000 description 1
- HFHDHCJBZVLPGP-UHFFFAOYSA-N schardinger α-dextrin Chemical compound O1C(C(C2O)O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC(C(O)C2O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC2C(O)C(O)C1OC2CO HFHDHCJBZVLPGP-UHFFFAOYSA-N 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 239000011550 stock solution Substances 0.000 description 1
- 238000003860 storage Methods 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 238000004808 supercritical fluid chromatography Methods 0.000 description 1
- 238000012360 testing method Methods 0.000 description 1
- 230000001225 therapeutic effect Effects 0.000 description 1
- 238000002560 therapeutic procedure Methods 0.000 description 1
- 231100000331 toxic Toxicity 0.000 description 1
- 230000002588 toxic effect Effects 0.000 description 1
- ODLHGICHYURWBS-LKONHMLTSA-N trappsol cyclo Chemical compound CC(O)COC[C@H]([C@H]([C@@H]([C@H]1O)O)O[C@H]2O[C@@H]([C@@H](O[C@H]3O[C@H](COCC(C)O)[C@H]([C@@H]([C@H]3O)O)O[C@H]3O[C@H](COCC(C)O)[C@H]([C@@H]([C@H]3O)O)O[C@H]3O[C@H](COCC(C)O)[C@H]([C@@H]([C@H]3O)O)O[C@H]3O[C@H](COCC(C)O)[C@H]([C@@H]([C@H]3O)O)O3)[C@H](O)[C@H]2O)COCC(O)C)O[C@@H]1O[C@H]1[C@H](O)[C@@H](O)[C@@H]3O[C@@H]1COCC(C)O ODLHGICHYURWBS-LKONHMLTSA-N 0.000 description 1
- 230000003612 virological effect Effects 0.000 description 1
- 239000000811 xylitol Substances 0.000 description 1
- 235000010447 xylitol Nutrition 0.000 description 1
- HEBKCHPVOIAQTA-SCDXWVJYSA-N xylitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)CO HEBKCHPVOIAQTA-SCDXWVJYSA-N 0.000 description 1
- 229960002675 xylitol Drugs 0.000 description 1
Landscapes
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
Abstract
The invention belongs to the field of protein pharmaceutical preparations, and relates to a dry powder inhaler of interferon alpha. The dry powder inhaler contains curative effective dosage of composite interferon and an appropriate dose of dry powder inhaler pharmaceutical adjuvant, wherein the dry powder inhaler pharmaceutical adjuvant comprises one or a plurality of active protective agent, disperse additive, pH stabilizing regulator, diluent and/or carrier with large particle size according to the function. In the optimized embodiment, the single administration dose of the dry powder inhaler of interferon alpha contains 1-200 mu grams of composite interferon. Compared with the dry powder inhaler of interferon alpha in the prior art, the stability of the dry powder inhaler of interferon alpha is obviously improved.
Description
Technical field
The inhalation dosage form that relates to interferon-ALPHA that the present invention is general, relates to the Foradil Aerolizer formoterol fumarate of interferon-ALPHA especially.
Background technology
Interferon (interferon, IFN) be a kind of cytokine class medicine with broad-spectrum disease resistance toxic action being produced by animal body at first, according to it, produce that position is different from the mechanism of action can be divided into the type that α, β, γ, λ etc. are large, and every kind of large type can be divided into some little hypotypes, in same large type, between different hypotype, on primary structure, difference is very little, very approaching on the above higher structure of secondary.In several large types, α type is most widely used a kind of, and this kind of interferon of clinical practice at present mainly comprises interferon-ALPHA 2a, interferon alpha 2 b, Interferon α1 b, Interferon Alfacon-1 etc.
In prior art, there are much reports for respiration system virus disease therapeutic about interferon-ALPHA.In the treatment of diseases of respiratory system toxicity disease, the mode administration that interferon-ALPHA generally sucks or sprays with injection, atomization, is prepared into respectively injection, atomized inhalation and spray.The curative effect of injecting recombinant human interferon alpha 1 b treatment infantile viral pneumonia reported in the article " interferon therapy viral pneumonia observation of curative effect " being for example published on the < < modern internal medicine > > magazine of China the 3rd volume the 6th phase 645-647 page in 2006, and Epidemiological Analysis shows to add with comparing and have significant difference in curative effect between the treatment group of recombinant human interferon alpha 1 b treatment and the matched group of employing conventional therapy by statistics.The article " interferon atomization inspiration treatment viral pneumonia efficacy analysis " being and for example published on the practical medical > > of < < China the 4th volume the 9th phase 151-152 page in 2009 has reported that atomization sucks the curative effect of recombinant human interferon-alpha treatment infantile viral pneumonia, and Epidemiological Analysis shows that the treatment group adding with recombinant human interferon-alpha atomization inspiration treatment compares and in curative effect, have significant difference with the matched group of employing general treatment by statistics.For another example Chinese patent CN 03147580.9 discloses a kind of recombinant human interferon alpha nebula for the treatment of viral pneumonia; it is equipped with suitable protective agent, mucosa absorption promoter, antibacterial etc. by recombinant human interferon-alpha stock solution and makes; pH value is at 5.0-8.0, and the recombinant human interferon alpha nebula that extracorporeal antivirus effect test shows above-mentioned formula is to respiratory tract or the susceptible HSV of pulmonary
1and HSV
2virus all has stronger inhibitory action, but to HSV
2the inhibitory action of virus is more responsive.
Although interferon-ALPHA is prepared into injection, atomized inhalation or spray, for the treatment of diseases of respiratory system toxicity disease, have clear and definite curative effect, the feature of general metabolism is had a greatly reduced quality the dosage of the actual performance of injection dosage form drug effect; Atomized inhalation and spray dosage form exist effective inhalation dose low, the shortcoming that dosage cannot accurately be controlled.
Interferon-ALPHA is prepared into Foradil Aerolizer formoterol fumarate, is also that powder spray can overcome through Pulmonary inhalation these shortcomings that above injection, atomized inhalation and spray dosage form exist completely from formulation characteristic.For example Chinese patent 95193669.7 discloses a kind of Foradil Aerolizer formoterol fumarate of interferon, it is prepared and is contained the interferon of the treatment effective dose of being combined with the acceptable adjuvant of medicine by interferon aqueous solution, this Foradil Aerolizer formoterol fumarate does not preferably contain penetration enhancer but preferably contains adjuvant human serum albumin.And for example Chinese patent application 200410018796.X discloses a kind of interferon powder spray; its material by following percentage by weight forms: interferon 0.0002%-0.8%; diluent 70%-97.9%; activity protecting agent 0.01%-5%; the buffer salt system of dispersibility auxiliary agent 0%-25% and maintenance pH 4-9 scopes, this powder spray does not preferably contain human albumin and absorption enhancer.
The Foradil Aerolizer formoterol fumarate of the interferon-ALPHA of above prior art report is mainly from the kind of Foradil Aerolizer formoterol fumarate pharmaceutically acceptable auxiliaries and the preparation of content research optimization of C/C composites composition, but substantially do not relate to the optimization of principal agent interferon-ALPHA hypotype and content, therefore likely by the optimization of principal agent interferon-ALPHA hypotype and content, obtain the Foradil Aerolizer formoterol fumarate of the more excellent interferon-ALPHA of performance, with the better treatment for viral diseases such as viral pneumonias.
Summary of the invention
The Foradil Aerolizer formoterol fumarate that the object of this invention is to provide a kind of interferon-ALPHA, the Foradil Aerolizer formoterol fumarate stability of its more existing interferon-ALPHA obviously improves.
For realizing this object; in basic embodiment; the invention provides a kind of Foradil Aerolizer formoterol fumarate of interferon-ALPHA; it contains treats the Interferon Alfacon-1 of effective dose and the Foradil Aerolizer formoterol fumarate pharmaceutically acceptable auxiliaries of Sq, and described Foradil Aerolizer formoterol fumarate pharmaceutically acceptable auxiliaries is divided and comprised one or more in activity protecting agent, dispersibility auxiliary agent, the agent of pH stable regulation, diluent and/or coarsegrain carrier by function.
Activity protecting agent play protection interferon-ALPHA Foradil Aerolizer formoterol fumarate preparation, store with transportation in activity do not lose or the effect of less forfeiture; it can be one or more the combination in albumin, aminoacid, polyhydric alcohol, cyclodextrin, lecithin matter; the preferably combination of one or more in albumin, lysine, 2-hydroxy propyl-Beta-cyclodextrin, soybean lecithin, and more preferably do not comprise one or more the combination in albuminous preferred substance.
Dispersibility auxiliary agent plays the granule of the Foradil Aerolizer formoterol fumarate that disperses the interferon-ALPHA prepare to suitable dispersion, prevent between granule reuniting each other and the effect of adhesion, can be one or more the combination in tyrosine, leucine, phenylalanine, glycine, preferably leucine, phenylalanine.
The effect that the pH after Foradil Aerolizer formoterol fumarate dissolving is stabilized in to physiology appropriate pH is played in the agent of pH stable regulation, including, but not limited to phosphate buffered solution system, citrate buffer solution system, Ascorbate buffer solution system, require physiological safety and can and be stabilized between 5.0-8.0 pH regulator.
Diluent plays to fill on the basis of drug content and other pharmaceutically acceptable auxiliaries content increases Foradil Aerolizer formoterol fumarate particle weight to the effect of required loadings or suction volume, it can be one or more the combination in polyhydric alcohol, amino acids material, the preferably combination of one or more in mannitol, xylitol, lactose, trehalose, cottonseed sugar, leucine, threonine, glycine, and the more preferably combination of one or more in mannitol, lactose, leucine, threonine.
Coarsegrain carrier plays and helps Foradil Aerolizer formoterol fumarate granule to be transported to from medicine-feeding part the effect of throat.When the Foradil Aerolizer formoterol fumarate grain graininess of the interferon-ALPHA preparing is less and dispersion is higher, it is suitable for by respiratory tract, being transported to pulmonary from throat, but be not suitable for being transported to throat from medicine-feeding part, now need in the Foradil Aerolizer formoterol fumarate granule preparing, add coarsegrain carrier; Thereby although and when the Foradil Aerolizer formoterol fumarate granule of the interferon-ALPHA preparing single less but have to each other looser adhesion can form when granularity reaches the loose aggregates of 50-100 μ m that (a little applies external force, as increase ambient air stream, the Foradil Aerolizer formoterol fumarate granule of adhesion will be dispersed into each other independently Foradil Aerolizer formoterol fumarate granule), in the granule without the Foradil Aerolizer formoterol fumarate of the interferon-ALPHA preparing, add coarsegrain carrier.Such coarsegrain carrier can be selected one or more the combination in the materials such as lactose, mannitol, phospholipid, aminoacid, the preferably combination of one or both in lactose and mannitol, and more preferably lactose.The particle mean size of coarsegrain carrier should be between 25-300 μ m, preferably between 50-200 μ m, and more preferably between 50-100 μ m.The ratio that Foradil Aerolizer formoterol fumarate granule mixes with coarsegrain carrier by weight should be between 100:1-1:100, preferably between 50:1-1:50, and more preferably between 20:1-1:20.
It should be pointed out that the situation of selecting an a kind of or class material simultaneously to play the function of above-mentioned multiple Foradil Aerolizer formoterol fumarate pharmaceutically acceptable auxiliaries of not getting rid of.
The Foradil Aerolizer formoterol fumarate granule of above-mentioned interferon-ALPHA can adopt the preparation such as the method that well known to a person skilled in the art lyophilizing-mechanical crushing method, spray drying method, supercritical fluid method, crystallization process, preferably adopts spray drying method preparation.When preparing the Foradil Aerolizer formoterol fumarate granule of interferon-ALPHA, employing spray drying method should first prepare the desiccant solution of spraying, also the various compositions that soon form Foradil Aerolizer formoterol fumarate except coarsegrain carrier are dissolved into solution state, then that this solution is dry for follow-up spraying.Spraying is dry can affect with the solid content in solution the performance that spray-dired efficiency and spraying are dried the Foradil Aerolizer formoterol fumarate granule obtaining, and it generally should be at 0.5-10%(m/v) between, preferably at 1-5%(m/v) between, and more preferably at 1-3%(m/v) between.
In spraying is dry, the main preparation technology parameter of the performance of the Foradil Aerolizer formoterol fumarate granule of the interferon-ALPHA that impact prepares comprises spraying dry gas inlet temperature, spray gas flow, hydrojet flow etc.
Spraying dry gas inlet temperature is higher, rate of drying is faster, material disposal ability is larger, but more easily there is bioinactivation and/or chemical degradation/reaction in interferon-ALPHA and Foradil Aerolizer formoterol fumarate pharmaceutically acceptable auxiliaries in spray-drying process, therefore suitable inlet temperature is 80-140 ℃, preferred 90-130 ℃, and more preferably 100-120 ℃.
Spray gas flow is higher, rate of drying is faster, material disposal ability is larger, but power consumption is simultaneously larger, in spray-drying process, more easily there is bioinactivation and/or chemical degradation/reaction in interferon-ALPHA and Foradil Aerolizer formoterol fumarate pharmaceutically acceptable auxiliaries, therefore for the small spraying drying instrument as Buchi B-290, suitable gas flow is 200-1000L/h, preferred 300-800 L/h, and more preferably 500-700 L/h, the spray gas flow of the spraying drying instrument of other types can utilize the General Principle that well known to a person skilled in the art operating parameter conversion when scaling-up dwindles to convert.
Hydrojet flow is lower, gas-liquid flow-rate ratio is larger, rate of drying is faster, the granularity of the Foradil Aerolizer formoterol fumarate granule obtaining is less, but treating capacity can obviously reduce simultaneously, processing time can obviously extend, and the processing time longly more easily causes that bioinactivation and/or chemical degradation/reaction occur in spray-drying process for interferon-ALPHA and pharmaceutically acceptable auxiliaries, therefore for the small spraying drying instrument as Buchi B-290, suitable hydrojet flow is 1-10ml/min, preferred 2-8ml/min, and more preferably 3-5ml/min, the gas flow of the spraying drying instrument of other types can utilize the General Principle that well known to a person skilled in the art operating parameter conversion when scaling-up dwindles to convert.
Other drying condition aspects of spraying, kind to spraying dry gas does not generally have specific (special) requirements, with air, but in order to prevent spray dry gas and interferon-ALPHA and Foradil Aerolizer formoterol fumarate pharmaceutically acceptable auxiliaries generation chemical reaction, also can select nitrogen or noble gas as spraying dry gas.The temperature of hydrojet generally selects freezing point temperature (0 ℃) to the temperature between room temperature (25 ℃).
By the control to operating parameter in above-mentioned spray-drying process, should make between the granularity 0.1-10 μ m of 50% above granule in the Foradil Aerolizer formoterol fumarate granule of the interferon-ALPHA for preparing, preferably the granularity of 70% above granule is between 1-5 μ m, and more preferably the granularity of 80% above granule between 2-4 μ m.
Because causing the Foradil Aerolizer formoterol fumarate mobility of the interferon-ALPHA for preparing, the too high meeting of moisture reduces, in storage process, grain graininess increases, principal agent biologic activity reduces, therefore by the control of operating parameter in above-mentioned spray-drying process, should make moisture in the Foradil Aerolizer formoterol fumarate granule of the interferon-ALPHA for preparing be controlled at below 5%, preferably below 3%, and most preferably below 2.5%.
The Foradil Aerolizer formoterol fumarate granule of the interferon-ALPHA preparing by said method should optionally determine whether mix with the carrier of coarsegrain.
In a preferred embodiment, the invention provides a kind of Foradil Aerolizer formoterol fumarate of interferon-ALPHA, the Interferon Alfacon-1 that contains 1-200 μ g in single-dose dosage.
In a preferred embodiment, the invention provides a kind of Foradil Aerolizer formoterol fumarate of interferon-ALPHA, the Interferon Alfacon-1 that contains 50-200 μ g in single-dose dosage.
In a preferred embodiment, the invention provides a kind of Foradil Aerolizer formoterol fumarate of interferon-ALPHA, in every 20mg, contain the activity protecting agent of 0.3-1mg; the dispersibility auxiliary agent of 0.6-5mg; the pH stable regulation agent of 1-4.8mg, the diluent of 3-9mg, the coarsegrain carrier of 0-15mg.
In a preferred embodiment, the invention provides a kind of Foradil Aerolizer formoterol fumarate of interferon-ALPHA, wherein said activity protecting agent is selected from one or more the combination in albumin, lysine, 2-HP-BETA-CD, soybean lecithin.
In a preferred embodiment, the invention provides a kind of Foradil Aerolizer formoterol fumarate of interferon-ALPHA, wherein said dispersibility auxiliary agent is selected from one or more the combination in tyrosine, leucine, phenylalanine, glycine.
In a preferred embodiment, the invention provides a kind of Foradil Aerolizer formoterol fumarate of interferon-ALPHA, the agent of wherein said pH stable regulation is selected from one or more the combination in phosphate buffered solution, citrate buffer solution, ascorbic acid salt buffer solution.
In a preferred embodiment, the invention provides a kind of Foradil Aerolizer formoterol fumarate of interferon-ALPHA, wherein said diluent is selected from one or more the combination in mannitol, lactose, leucine, threonine.
In a preferred embodiment, the invention provides a kind of Foradil Aerolizer formoterol fumarate of interferon-ALPHA, wherein said coarsegrain carrier is selected from one or both the combination in lactose and mannitol.
The term using in the present invention " Foradil Aerolizer formoterol fumarate " (Dry Power Inhalation, DPI), also referred to as " powder spray ", refer to micronized medicine and/or carrier with single dose or multiple dose depot forms, adopt special powder inhaler, by patient initiatively or passive suction atomization medicine to the preparation of respiratory tract and even pulmonary.
The term using in the present invention " treatment effective dose " represents to be used for the treatment of or during prevent disease, the amount of active constituents of medicine is enough to realization to the treatment of disease or prevention when drug administration active component.Treatment effective dose is by the age according to active constituents of medicine, disease and its seriousness and the patient that treats, body weight etc. and different.
The term using in the present invention " Foradil Aerolizer formoterol fumarate pharmaceutically acceptable auxiliaries " refers to when Foradil Aerolizer formoterol fumarate Formulation, for solving the problems such as mouldability, effectiveness, stability, safety of Foradil Aerolizer formoterol fumarate, join self other auxiliary element to human body or animal body safety except active constituents of medicine in Foradil Aerolizer formoterol fumarate prescription.
The term using in the present invention " Interferon Alfacon-1 " (consensus interferon or integrated interferon) refer in Chinese patent application CN02159950.5 in the open sequence of sequence table 3 and in description embodiment the Interferon Alfacon-1 of open preparation method.
The specific embodiment
By following embodiment, enforcement of the present invention is described further, but embodiments of the present invention are not limited to following embodiment.
Embodiment 1: the preparation of the Foradil Aerolizer formoterol fumarate granule of interferon-ALPHA
By as dry with solution in each spraying of the formulated in following table 1, and by dry this solution of Buchi B-290 spraying drying instrument spraying for the spraying drying condition of table 2 correspondence (whole liquid sprays complete after continuation maintenance spray dry gas inlet temperature and flow 15 minutes) thus prepare the Foradil Aerolizer formoterol fumarate granule of interferon-ALPHA.
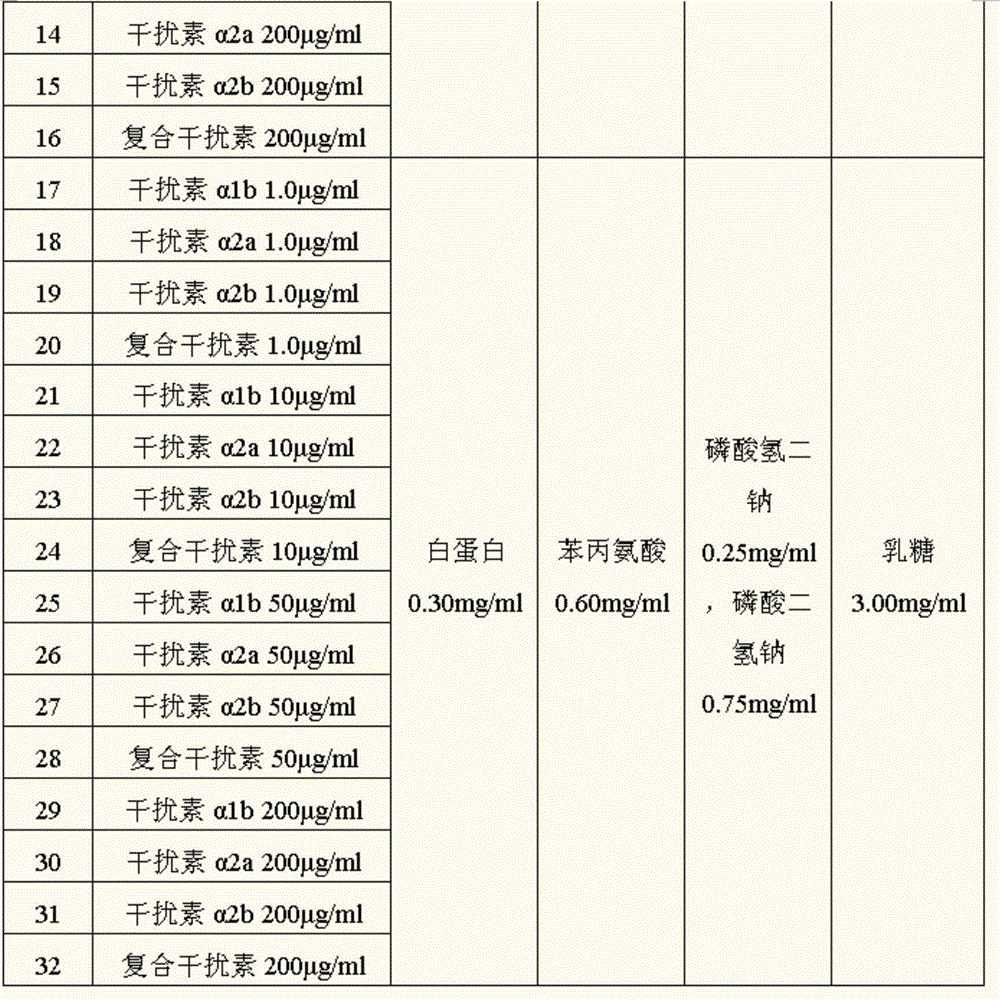
Table 1 is prepared the dry solution formula of using of spraying of the Foradil Aerolizer formoterol fumarate granule of interferon-ALPHA
The spraying drying condition of the Foradil Aerolizer formoterol fumarate of table 2 interferon-ALPHA
Embodiment 2: the quality evaluation of the Foradil Aerolizer formoterol fumarate granule of interferon-ALPHA
The activity (IU/ml) of interferon-ALPHA in solution after the Foradil Aerolizer formoterol fumarate grain dissolution of the interferon-ALPHA of each formula preparing by the regulation mensuration of < < Chinese Pharmacopoeia 2010 editions (three) > > appendix " interferon biological activity algoscopy "; By the regulation of < < Chinese Pharmacopoeia 2010 editions (three) > > appendix " protein determination " the second method Lowry method, measure after the Foradil Aerolizer formoterol fumarate grain dissolution of interferon-ALPHA of each formula preparing the concentration (mg/ml) of interferon-ALPHA in solution, both the be divided by specific activities (IU/mg) of interferon-ALPHA in the Foradil Aerolizer formoterol fumarate granule of interferon-ALPHA of each formula of obtaining preparing.The dry specific activity with interferon-ALPHA in solution of each spraying of blank determination, and the reservation of calculating thus spray-dried rear interferon-ALPHA specific activity.
Moisture in the Foradil Aerolizer formoterol fumarate granule of the interferon-ALPHA of each formula preparing by the regulation mensuration of < < Chinese Pharmacopoeia 2010 editions (three) > > appendix " aquametry " first method.
By the regulation of < < Chinese Pharmacopoeia 2010 editions (two) > > appendix " granularity and particle size distribution method " first method, measure the particle mean size of the Foradil Aerolizer formoterol fumarate granule of each interferon-ALPHA of filling a prescription preparing.
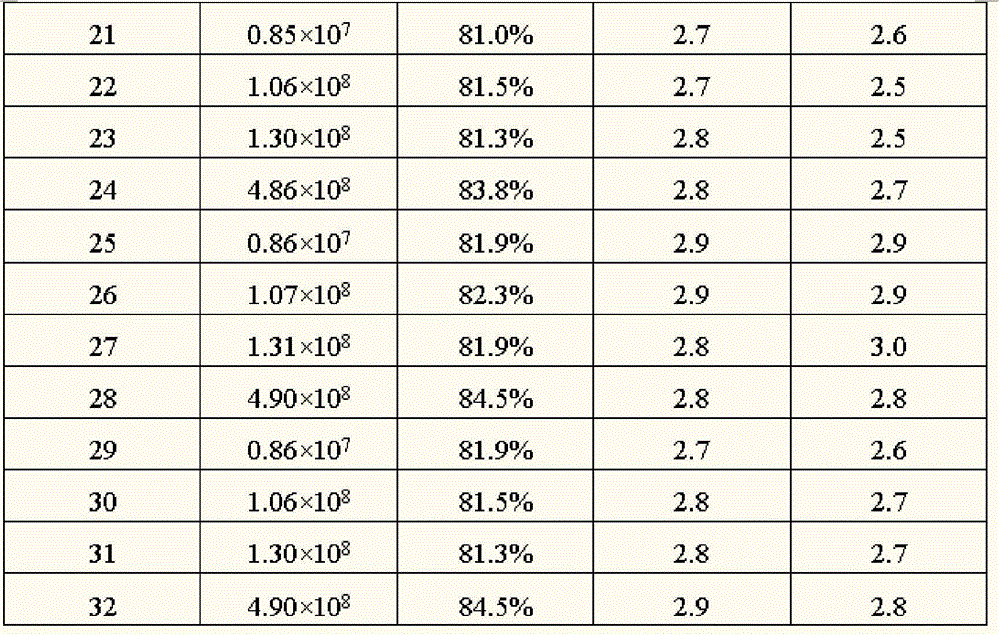
All measurement results are as shown in table 3 below.
The quality evaluation of the Foradil Aerolizer formoterol fumarate granule of the dry interferon-ALPHA preparing of table 3 spraying
Embodiment 3: the Foradil Aerolizer formoterol fumarate of being prepared interferon-ALPHA by the Foradil Aerolizer formoterol fumarate granule of interferon-ALPHA
The Foradil Aerolizer formoterol fumarate particulate samples of the interferon-ALPHA of the formula 1-16 in each formula that the method for pressing embodiment 1 is obtained does not mix with coarsegrain carrier, and direct amount of usining every capsule 20mg is carried out after subpackage the research for embodiment 4 as the Foradil Aerolizer formoterol fumarate of interferon-ALPHA; The Foradil Aerolizer formoterol fumarate particulate samples of interferon-ALPHA of formula 17-32 is mixed homogeneously with commercially available coarsegrain lactose carrier granule with the mass ratio of 1:3, and the amount of every capsule 20mg of usining is carried out after subpackage the research for embodiment 4 as the Foradil Aerolizer formoterol fumarate of interferon-ALPHA.
Embodiment 4: the accelerated stability test of the Foradil Aerolizer formoterol fumarate of interferon-ALPHA
Each many of the capsules of each formula are placed in to 40 ℃, the environment of 75%RH is placed March, the activity (IU/ml) of interferon-ALPHA in solution after the Foradil Aerolizer formoterol fumarate of the interferon-ALPHA of each formula that monthly sampling once prepares by the regulation mensuration of < < Chinese Pharmacopoeia 2010 editions (three) > > appendix " interferon biological activity algoscopy " takes out and dissolves in capsule; The concentration (mg/ml) of interferon-ALPHA in solution after the Foradil Aerolizer formoterol fumarate of measuring the interferon-ALPHA of each formula preparing by the regulation of < < Chinese Pharmacopoeia 2010 editions (three) > > appendix " protein determination " the second method Lowry method takes out and dissolves in capsule, both the be divided by specific activities (IU/mg) of interferon-ALPHA in the Foradil Aerolizer formoterol fumarate of each interferon-ALPHA of filling a prescription of obtaining preparing.Each sample of filling a prescription sampled divided by corresponding 0 month the specific activity recording with the specific activity that January, February, March, sampling recorded respectively, obtained the specific activity relative retention value of interferon-ALPHA in accelerated stability test process, and result is as shown in table 4 below.
The accelerated stability test result of the Foradil Aerolizer formoterol fumarate of table 4 interferon-ALPHA
Claims (5)
1. the Foradil Aerolizer formoterol fumarate of interferon-ALPHA; it is characterized in that the Interferon Alfacon-1 that contains 50-200 μ g in single-dose dosage; the activity protecting agent that contains 0.3-1mg in the Foradil Aerolizer formoterol fumarate of every 20mg; the dispersibility auxiliary agent of 0.6-5mg; the pH stable regulation agent of 1-4.8mg; the diluent of 3-9mg, the coarsegrain carrier of 0-15mg
Described Interferon Alfacon-1 is the Interferon Alfacon-1 of SEQ ID No.3 in CN02159950.5,
Described Foradil Aerolizer formoterol fumarate pharmaceutically acceptable auxiliaries is divided and is comprised one or more in activity protecting agent, dispersibility auxiliary agent, the agent of pH stable regulation, diluent and/or coarsegrain carrier by function,
Described dispersibility auxiliary agent is selected from one or more the combination in tyrosine, leucine, phenylalanine, glycine,
Described coarsegrain carrier is selected from one or more the combination in the lactose of particle mean size between 25-300 μ m, mannitol, phospholipid, aminoacid.
2. the Foradil Aerolizer formoterol fumarate of interferon-ALPHA according to claim 1, is characterized in that described activity protecting agent is selected from one or more the combination in albumin, lysine, 2-HP-BETA-CD, soybean lecithin.
3. the Foradil Aerolizer formoterol fumarate of interferon-ALPHA according to claim 1, is characterized in that described pH stable regulation agent is selected from one or more the combination in phosphate buffered solution, citrate buffer solution, ascorbic acid salt buffer solution.
4. the Foradil Aerolizer formoterol fumarate of interferon-ALPHA according to claim 1, is characterized in that described diluent is selected from one or more the combination in mannitol, lactose, leucine, threonine.
5. the Foradil Aerolizer formoterol fumarate of interferon-ALPHA according to claim 1, is characterized in that described coarsegrain carrier is selected from one or both the combination in lactose and mannitol.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201210235912.8A CN102727468B (en) | 2012-07-07 | 2012-07-07 | Dry powder inhaler of interferon alpha |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201210235912.8A CN102727468B (en) | 2012-07-07 | 2012-07-07 | Dry powder inhaler of interferon alpha |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN102727468A CN102727468A (en) | 2012-10-17 |
| CN102727468B true CN102727468B (en) | 2014-03-12 |
Family
ID=46984239
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201210235912.8A Active CN102727468B (en) | 2012-07-07 | 2012-07-07 | Dry powder inhaler of interferon alpha |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN102727468B (en) |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN105770862B (en) * | 2016-04-22 | 2022-01-11 | 北京三元基因药业股份有限公司 | Dry powder inhalant of interferon |
| CN105770863A (en) * | 2016-04-22 | 2016-07-20 | 北京三元基因药业股份有限公司 | Dry powder inhaler of interferon Lambda |
| CN105920586A (en) * | 2016-04-22 | 2016-09-07 | 北京三元基因药业股份有限公司 | Interferon aerosol propellant |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1151168A (en) * | 1994-05-18 | 1997-06-04 | 吸入治疗系统公司 | Method and compositions for the dry powder formulation of interferons |
| CN1478545A (en) * | 2003-07-18 | 2004-03-03 | 中国科学院微生物研究所 | Application of compound interferon in treating SARS disease |
| CN1511849A (en) * | 2002-12-30 | 2004-07-14 | 北京三元基因工程有限公司 | Novel alpha interferon mutant and its preparing process |
| CN1672731A (en) * | 2004-03-26 | 2005-09-28 | 天津药物研究院 | Interferon spray for lung administration and its prepn process |
-
2012
- 2012-07-07 CN CN201210235912.8A patent/CN102727468B/en active Active
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1151168A (en) * | 1994-05-18 | 1997-06-04 | 吸入治疗系统公司 | Method and compositions for the dry powder formulation of interferons |
| CN1511849A (en) * | 2002-12-30 | 2004-07-14 | 北京三元基因工程有限公司 | Novel alpha interferon mutant and its preparing process |
| CN1478545A (en) * | 2003-07-18 | 2004-03-03 | 中国科学院微生物研究所 | Application of compound interferon in treating SARS disease |
| CN1672731A (en) * | 2004-03-26 | 2005-09-28 | 天津药物研究院 | Interferon spray for lung administration and its prepn process |
Non-Patent Citations (2)
| Title |
|---|
| L-亮氨酸对干扰素粉雾剂性能的影响;江荣高等;《中国医药工业杂志》;20051231;第36卷(第12期);第747-750页 * |
| 江荣高等.L-亮氨酸对干扰素粉雾剂性能的影响.《中国医药工业杂志》.2005,第36卷(第12期), |
Also Published As
| Publication number | Publication date |
|---|---|
| CN102727468A (en) | 2012-10-17 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| FI116195B (en) | Method and compositions for delivering insulin via lungs | |
| FI111909B (en) | Process for the preparation of inhalation compositions | |
| KR100391873B1 (en) | Powder Formulations Containing Melezitose as a Diluent | |
| KR100384353B1 (en) | Methods and compositions for preparing dry powder formulations of interferon | |
| US20230113539A1 (en) | Traditional Chinese medicine for dispersing lung qi and detoxicating | |
| JPH05508616A (en) | therapeutic aerosol | |
| CN102716105B (en) | Dry powder inhalant of interferon Alpha | |
| CN111202722A (en) | Lopinavir inhalation dry powder pharmaceutical composition and preparation method thereof | |
| KR20220045214A (en) | Pharmaceutical composition comprising ensifentrine | |
| US20030148925A1 (en) | Insulin formulation for inhalation | |
| CN114949183A (en) | Tinepotide powder aerosol and preparation method thereof | |
| CN102727468B (en) | Dry powder inhaler of interferon alpha | |
| US10596144B2 (en) | Treating or preventing nephrogenic diabetes insipidus | |
| JP2009513682A (en) | Stabilized formulation containing γ- and α-interferon at a rate that exhibits a synergistic effect | |
| Saha et al. | Inhalable dry powder containing remdesivir and disulfiram: Preparation and in vitro characterization | |
| CN105770862B (en) | Dry powder inhalant of interferon | |
| CN116327743A (en) | Application of baicalein inhalation preparation in preparation of acute lung injury treatment drugs | |
| CN102716469B (en) | Dry powder inhalant of interferon alpha | |
| CN105920586A (en) | Interferon aerosol propellant | |
| CN102727469B (en) | Dry powder inhalant of interferon alpha | |
| CN102727467B (en) | Dry powder inhalant of interferon alpha | |
| CN1289143C (en) | Method for reducing fasting blood-glucose of diabetic and weight | |
| RU2175556C2 (en) | Methods and compositions for light delivery of insulin | |
| US20160296574A1 (en) | Ephedra alata extracts and methods of use thereof | |
| US20230248722A1 (en) | Clofazimine composition and method for the treatment or prophylaxis of viral infections |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant | ||
| C56 | Change in the name or address of the patentee | ||
| CP01 | Change in the name or title of a patent holder |
Address after: 102600 Beijing City, Daxing District Daxing Industrial Development Zone Jinyuan Road No. 1 Patentee after: BEIJING TRI-PRIME GENE PHARMACEUTICAL CO., LTD. Address before: 102600 Beijing City, Daxing District Daxing Industrial Development Zone Jinyuan Road No. 1 Patentee before: Beijing Sanyuan Gene Engineering Co., Ltd. |