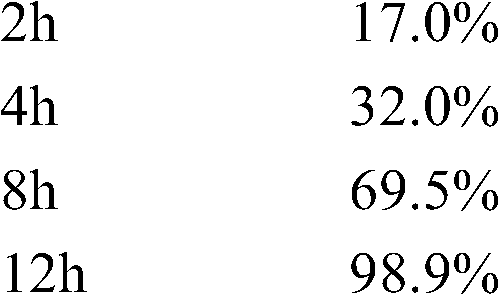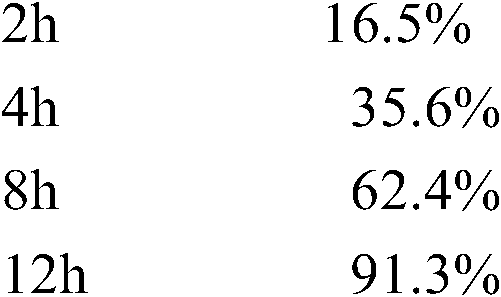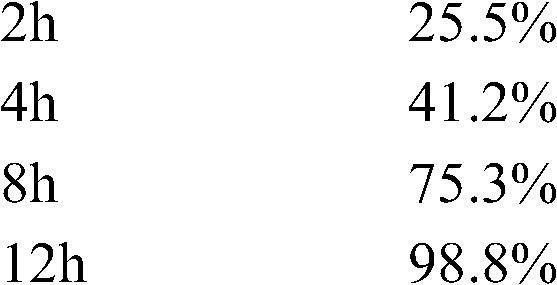CN102727459A - A formulation having first and second active agents - Google Patents
A formulation having first and second active agents Download PDFInfo
- Publication number
- CN102727459A CN102727459A CN2012101072455A CN201210107245A CN102727459A CN 102727459 A CN102727459 A CN 102727459A CN 2012101072455 A CN2012101072455 A CN 2012101072455A CN 201210107245 A CN201210107245 A CN 201210107245A CN 102727459 A CN102727459 A CN 102727459A
- Authority
- CN
- China
- Prior art keywords
- pharmaceutical preparation
- pioglitazone
- preparation
- release
- active
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
- A61K31/4427—Non condensed pyridines; Hydrogenated derivatives thereof containing further heterocyclic ring systems
- A61K31/4439—Non condensed pyridines; Hydrogenated derivatives thereof containing further heterocyclic ring systems containing a five-membered ring with nitrogen as a ring hetero atom, e.g. omeprazole
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/13—Amines
- A61K31/155—Amidines (), e.g. guanidine (H2N—C(=NH)—NH2), isourea (N=C(OH)—NH2), isothiourea (—N=C(SH)—NH2)
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0002—Galenical forms characterised by the drug release technique; Application systems commanded by energy
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/28—Dragees; Coated pills or tablets, e.g. with film or compression coating
- A61K9/2806—Coating materials
- A61K9/2833—Organic macromolecular compounds
- A61K9/284—Organic macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyvinyl pyrrolidone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/28—Dragees; Coated pills or tablets, e.g. with film or compression coating
- A61K9/2806—Coating materials
- A61K9/2833—Organic macromolecular compounds
- A61K9/286—Polysaccharides, e.g. gums; Cyclodextrin
- A61K9/2866—Cellulose; Cellulose derivatives, e.g. hydroxypropyl methylcellulose
Landscapes
- Health & Medical Sciences (AREA)
- Epidemiology (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Medicinal Preparation (AREA)
Abstract
Description
技术领域 technical field
本发明涉及一种具有第一和第二活性药物的制剂及其在制备II型糖尿病的药物中的用途。 The present invention relates to a preparation with a first and a second active drug and its use in the preparation of a medicament for type II diabetes. the
背景技术 Background technique
目前人们已经使用许多技术来提供控制释放的和缓释的药物剂型以维持药物治疗血清浓度和减少由于患者缺乏顺从性所引起的药物剂量不足带来的影响。例如,现有技术已经公开了缓释片剂,其具有被半透膜包裹的渗透活性的药物芯。这些片剂的功能是通过使体液中的水溶性成分例如胃肠液渗透过包衣膜并溶解活性成分,从而所产生的药物溶液可通过包衣膜中的通道释放出来。或者,如果活性成分在渗透液中不溶解,它可以通过膨胀剂如水凝胶通过通道。在美国专利Nos.3,845,770;3,916,899;4,034,758;4,077,407和4,783,337中公开了这些渗透性片剂系统的某些代表性的实例。美国专利No.3,952,741公开了一种渗透性装置,其中只有在膜内已经具有足够的压力而能在膜的薄弱部分上使膜裂解或破裂后,活性剂才从被半透膜包裹的芯释放。 A number of techniques have been used to provide controlled-release and sustained-release drug dosage forms to maintain therapeutic serum levels of the drug and to reduce the effects of drug underdosing due to lack of patient compliance. For example, the prior art has disclosed sustained release tablets having an osmotically active drug core surrounded by a semipermeable membrane. These tablets function by allowing water-soluble components in body fluids such as gastrointestinal fluids to permeate through the coating and dissolve the active ingredient, so that the resulting drug solution can be released through channels in the coating. Alternatively, if the active ingredient is insoluble in the permeate, it can be passed through the channels by a swelling agent such as a hydrogel. Some representative examples of these osmotic tablet systems are disclosed in US Patent Nos. 3,845,770; 3,916,899; 4,034,758; 4,077,407 and 4,783,337. U.S. Patent No. 3,952,741 discloses an osmotic device in which the active agent is released from a core surrounded by a semi-permeable membrane only after there has been sufficient pressure within the membrane to crack or rupture the membrane over a weakened portion of the membrane . the
目前,人们已经努力对在上述引用的专利中所描述的基本渗透装置进行了改进以提供对活性成分更好的控制释放。例如,美国专利Nos.4,777,049和4,851,229描述了包括包裹芯的半透膜的渗透剂型。该芯包含一种活性成分和一种调节剂,其中所述调节剂使活性成分通过半透膜上的通道以脉冲方式释放。更进一步的改进包括改进包裹活性芯的半透膜,例如美国专利Nos.5,178,867,4,587,117和4,522,625中所述那样改变成膜成分的比例,或者例如美国专利Nos.5,650,170和4,892,739中所述那样增加包裹活性芯的包衣的数目。 Currently, efforts have been made to modify the basic osmotic devices described in the above cited patents to provide better controlled release of the active ingredient. For example, US Patent Nos. 4,777,049 and 4,851,229 describe osmotic dosage forms comprising a semipermeable membrane surrounding a core. The core contains an active ingredient and a modulator, wherein the modulator causes the active ingredient to be released in a pulsatile manner through channels in the semipermeable membrane. Further improvements include modifying the semipermeable membrane surrounding the active core, such as changing the ratio of membrane-forming components as described in U.S. Patent Nos. 5,178,867, 4,587,117 and 4,522,625, or increasing the wrapping The number of coatings on the active core. the
某些使用抗高血糖药物例如盐酸二甲双胍的控制释放或缓释制剂已被限制于使用膨胀或胶凝剂来控制药物从剂型中释放。该受限制的研究由W096/08243公开的内容和购自Bristol-Myers Squibb Co.的盐酸 二甲双胍控制释放产品GLUCOPHAGETM XR所例证。 Certain controlled release or sustained release formulations using antihyperglycemic drugs such as metformin hydrochloride have been limited to the use of bulking or gelling agents to control the release of the drug from the dosage form. This limited study is exemplified by the publication of WO96/08243 and GLUCOPHAGE™ XR, a controlled release product of metformin hydrochloride available from Bristol-Myers Squibb Co. the
在美国专利No.4,687,777中已描述了噻唑烷二酮衍生物。这些化合物在联合治疗中的治疗作用已被进一步地描述于在美国专利Nos.5,859,037;5,952,356;5,965,584;6,150,384和6,172,090中。 Thiazolidinedione derivatives have been described in US Patent No. 4,687,777. The therapeutic effect of these compounds in combination therapy has been further described in US Patent Nos. 5,859,037; 5,952,356; 5,965,584; 6,150,384 and 6,172,090. the
在本领域中已经公开使用包含抗高血压药物和噻唑烷二酮衍生物的组合的药物剂型。例如,EPOO 749751(将其引入本文作为参考)公开了包含胰岛素增敏剂(其可以为噻唑烷二酮化合物)的药物组合物,并包含其它抗糖尿病药。更特别地,EPOO 749751公开了优选的胰岛素增敏剂是吡格列酮,其可以与其它抗糖尿病药例如二甲双胍、苯乙双胍或丁基双胍组合,并且进一步地这些药物可以与常规赋形剂组合(混合和/或包衣)来提供掩盖气味或缓释。抗高血糖药物和噻唑烷二酮衍生物组合的另一个实例是美国专利No.6,011,049(将其引入本文作为参考)。该专利公开了以缓释形式比如渗透泵或皮肤贴片包含吡格列酮或曲格列酮和二甲双胍的单一药物组合物。其它将抗高血糖药物和噻唑烷二酮衍生物的组合可发现于美国专利Nos.6,524,621;6,475,521;6,451,342和6,153,632和PCT专利申请WO01/3594和W001/3594中。 The use of pharmaceutical dosage forms comprising combinations of antihypertensive drugs and thiazolidinedione derivatives has been disclosed in the art. For example, EPOO 749751 (which is incorporated herein by reference) discloses pharmaceutical compositions comprising an insulin sensitizer, which may be a thiazolidinedione compound, and comprising other antidiabetic agents. More particularly, EPOO 749751 discloses that a preferred insulin sensitizer is pioglitazone, which can be combined with other antidiabetic drugs such as metformin, phenformin or butylbiguanide, and further these drugs can be combined with conventional excipients (mixing and/or coating) to provide odor masking or sustained release. Another example of a combination of an antihyperglycemic drug and a thiazolidinedione derivative is US Patent No. 6,011,049 (herein incorporated by reference). This patent discloses a single pharmaceutical composition comprising pioglitazone or troglitazone and metformin in a sustained release form such as an osmotic pump or a dermal patch. Other combinations of antihyperglycemic agents and thiazolidinedione derivatives can be found in US Patent Nos. 6,524,621; 6,475,521; 6,451,342 and 6,153,632 and PCT Patent Applications WO01/3594 and WO01/3594. the
在本领域中也已知W099/47125和美国专利No.6,099,862中公开了用立即释放包衣来包衣的二甲双胍渗透片剂,所述包衣包含抗高血糖或降糖药物。 Also known in the art are WO 99/47125 and US Patent No. 6,099,862 which disclose metformin osmotic tablets coated with an immediate release coating comprising an antihyperglycemic or hypoglycemic drug. the
CN100544717C公开了一种具有第一和第二活性药物的药物剂型,其中采用的是低粘性水溶性粘合剂这一常规技术,并未创造性地涉及高粘性或水不溶性粘合剂。 CN100544717C discloses a pharmaceutical dosage form with first and second active drugs, which adopts the conventional technology of low-viscosity water-soluble adhesives, and does not creatively involve high-viscosity or water-insoluble adhesives. the
发明内容 Contents of the invention
本发明的内容在于提供一种具有第一和第二活性药物的制剂,所述制剂包括: The content of the present invention is to provide a kind of preparation with first and second active drug, described preparation comprises:
a)控制释放片芯,其含有抗高血糖药物和至少一种药学上可接受的赋形剂;和 a) a controlled release tablet core containing an antihyperglycemic drug and at least one pharmaceutically acceptable excipient; and
b)速释包衣层,该包衣层包括立即释放的第二活性药物和高粘性水溶性粘合剂或水不溶性粘合剂。 b) An immediate release coating comprising an immediate release second active drug and a highly viscous water soluble or water insoluble binder. the
优选地,第一活性药物为二甲双胍及其药学上可接受的盐,或者 第二活性药物为吡格列酮及其药学上可接受的盐。 Preferably, the first active drug is metformin and a pharmaceutically acceptable salt thereof, or the second active drug is pioglitazone and a pharmaceutically acceptable salt thereof. the
进一步优选地,所述制剂包括: Further preferably, the preparation includes:
a)控制释放片芯,其中包含约60%~90%的第一活性药物、5%~40%的粘合剂、0~20%的吸收促进剂及0~3%的润滑剂; a) Controlled-release tablet core, which contains about 60%-90% of the first active drug, 5%-40% of binder, 0-20% of absorption enhancer and 0-3% of lubricant;
b)半透膜; b) semi-permeable membrane;
c)速释包衣层,该包衣层含有:第二活性药物、高粘性水溶性粘合剂或水不溶性粘合剂及吸收促进剂或表面活性剂。 c) an immediate-release coating layer, which contains: the second active drug, a high-viscosity water-soluble binder or a water-insoluble binder, and an absorption promoter or a surfactant. the
进一步优选地,在20℃作为2%的水溶液测定时,所述高粘性水溶性粘合剂具有大于6mPa.s的粘度。 Further preferably, the highly viscous water-soluble adhesive has a viscosity greater than 6 mPa.s when measured as a 2% aqueous solution at 20°C. the
进一步优选地,立即释放层的粘合剂为羟丙甲基纤维素或者丙烯酸树脂。 Further preferably, the binder of the immediate release layer is hydroxypropylmethylcellulose or acrylic resin. the
本发明所提供的药物制剂克服了现有技术中的技术偏见,大胆采用了非常规的粘合剂,取得了较现有技术相当甚至更好的成药效果及药效表现。 The pharmaceutical preparation provided by the invention overcomes the technical prejudice in the prior art, boldly adopts unconventional adhesives, and achieves comparable or even better drug-making effect and drug performance than the prior art. the
具体实施方式 Detailed ways
以下提供的例子仅用于说明而非表示限制。 The examples provided below are for illustration only and not meant to be limiting. the
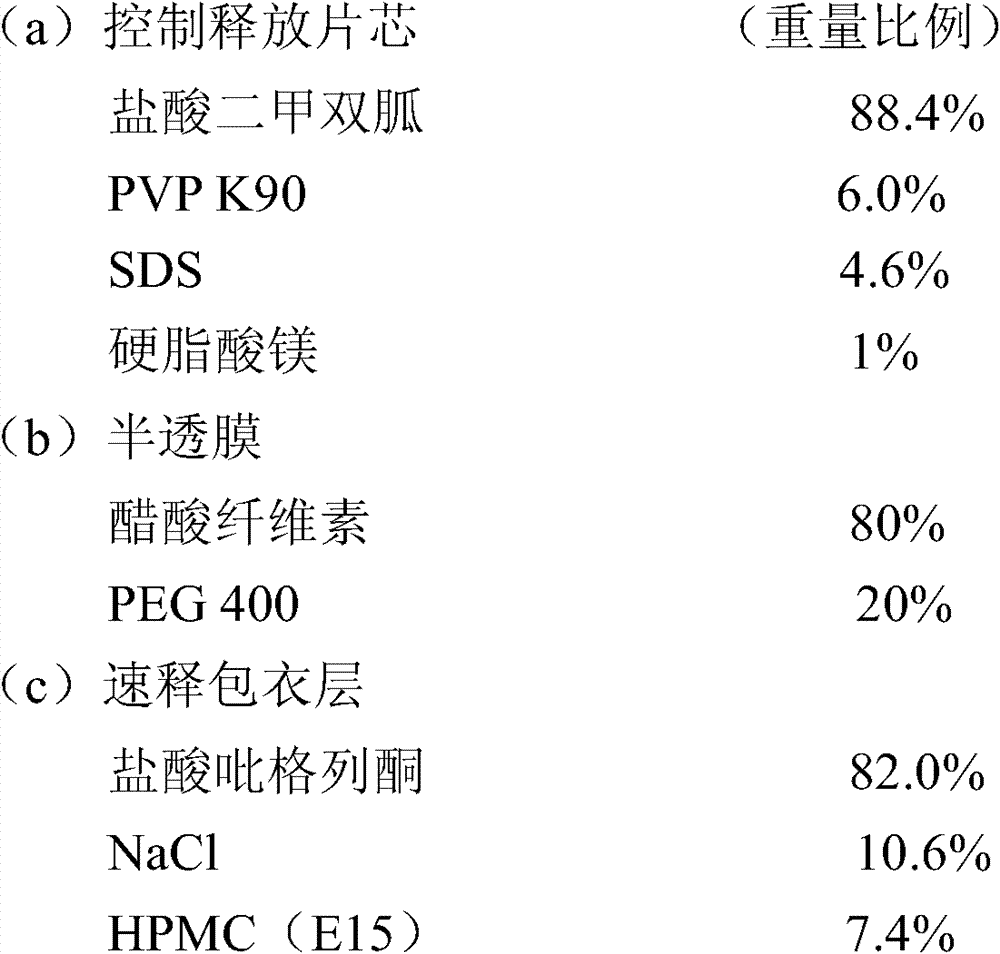
实施例1包含33.07mg盐酸吡格列酮(以吡格列酮计30mg)和1000mg的盐酸二甲双胍控制释放片剂的制备 Embodiment 1 comprises the preparation of 33.07mg pioglitazone hydrochloride (30mg in terms of pioglitazone) and 1000mg metformin hydrochloride controlled release tablet
制备方法: Preparation:
1.将1~5%的PVP K90配制成15%的水溶液,剩余的PVP与盐酸二甲双胍混合,以湿法制粒工艺将上述混合物制备成颗粒并压制成片芯; 1. Prepare 1-5% PVP K90 into a 15% aqueous solution, mix the remaining PVP with metformin hydrochloride, prepare the above mixture into granules by wet granulation process and press into tablet cores;
2.半透膜包衣液的配制:先将醋酸纤维素配制成澄清的丙酮溶液,聚乙二醇400配制成澄清的水溶液,将两种溶液混合,搅拌至澄清溶液,备用; 2. Preparation of semi-permeable membrane coating solution: first prepare cellulose acetate into a clear acetone solution, polyethylene glycol 400 into a clear aqueous solution, mix the two solutions, stir until a clear solution, and set aside;
3.速释层包衣液的配制:将NaCl溶于少量的水后加入95%的乙醇中,再分别加入HPMC和盐酸吡格列酮,分散后制得包衣液; 3. Preparation of immediate-release layer coating solution: Dissolve NaCl in a small amount of water and add 95% ethanol, then add HPMC and pioglitazone hydrochloride respectively, and disperse to prepare the coating solution;
4.用激光打孔机在片芯上下两面各打一个孔; 4. Use a laser drilling machine to punch a hole on the upper and lower sides of the core;
5.包衣结束后,于40℃条件下放置24小时,取出,即可。 5. After coating, place it at 40°C for 24 hours, then take it out. the
盐酸二甲双胍释放度的测定: Determination of Metformin Hydrochloride Release Rate:
取样品,照释放度的测定法,采用中国药典2005年版二部附录XD第一法,溶出度测定法第一法的装置,以0.1N的盐酸溶液900mL为释放介质,转速为100rpm,依法操作,在第2、4、8、12小时取点,采用高效液相色谱法进行释放度测定,药物的释放数据如下: Get the sample, according to the assay method of release rate, adopt the first method of appendix XD of Chinese Pharmacopoeia 2005 edition two, the device of dissolution assay method first method, take 0.1N hydrochloric acid solution 900mL as release medium, rotating speed is 100rpm, operate according to law , take points at 2, 4, 8, and 12 hours, and use high-performance liquid chromatography to measure the release rate. The release data of the drug is as follows:
盐酸吡格列酮溶出度的测定: Determination of the dissolution rate of pioglitazone hydrochloride:
取样品,照释放度的测定法,采用中国药典2005年版二部附录XD第一法,溶出度测定法第一法的装置,以0.1N的盐酸溶液900mL为释放介质,转速为50rpm,依法操作,在第15、30、45分钟取点,采用高效液相色谱法进行释放度测定,药物的溶出结果如下: Get the sample, according to the assay method of release rate, adopt the first method of appendix XD of Chinese Pharmacopoeia 2005 edition two, the device of dissolution assay method first method, take 0.1N hydrochloric acid solution 900mL as release medium, rotating speed is 50rpm, operate according to law , at the 15th, 30th, and 45th minute points were taken, and the release rate was measured by high performance liquid chromatography. The dissolution results of the drug were as follows:
15min 94.8% 15min 94.8%
30min 98.6% 30min 98.6%
45min 98.9% 45min 98.9%
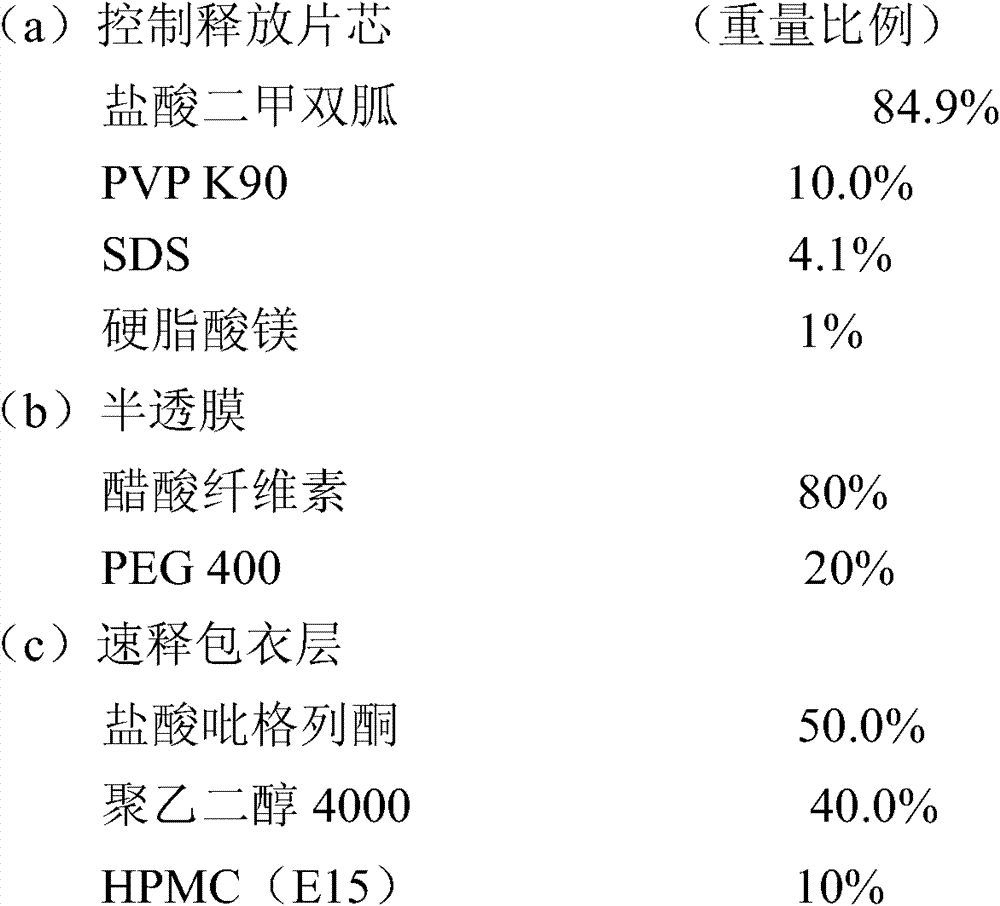
实施例2包含33.07mg盐酸吡格列酮(以吡格列酮计30mg)和 1000mg的盐酸二甲双胍控制释放片剂的制备 Embodiment 2 comprises the preparation of 33.07mg pioglitazone hydrochloride (30mg as pioglitazone) and 1000mg metformin hydrochloride controlled release tablet
制备方法: Preparation:
1.片芯、半透膜制备同实施例1。 1. The preparation of tablet core and semipermeable membrane is the same as in Example 1. the
2.速释层包衣液的配制:将聚乙二醇4000溶于少量的水后加入95%的乙醇中,再分别加入HPMC和盐酸吡格列酮,分散后制得包衣液; 2. Preparation of immediate-release layer coating solution: Dissolve polyethylene glycol 4000 in a small amount of water and add 95% ethanol, then add HPMC and pioglitazone hydrochloride respectively, and disperse to prepare the coating solution;
3.用激光打孔机在片芯上下两面各打一个孔; 3. Use a laser drilling machine to punch a hole on the upper and lower sides of the core;
4.包衣结束后,于40℃条件下放置24小时,取出,即可。 4. After coating, place it at 40°C for 24 hours, then take it out. the
盐酸二甲双胍释放度的测定: Determination of Metformin Hydrochloride Release Rate:
测定法同实施例1,其释放数据如下: Assay method is the same as embodiment 1, and its release data are as follows:
盐酸吡格列酮溶出度的测定: Determination of the dissolution rate of pioglitazone hydrochloride:
测定法同实施例1,其溶出结果如下: Assay method is with embodiment 1, and its stripping result is as follows:
15min 85.2% 15min 85.2%
30min 99.5% 30min 99.5%
45min 98.7% 45min 98.7%
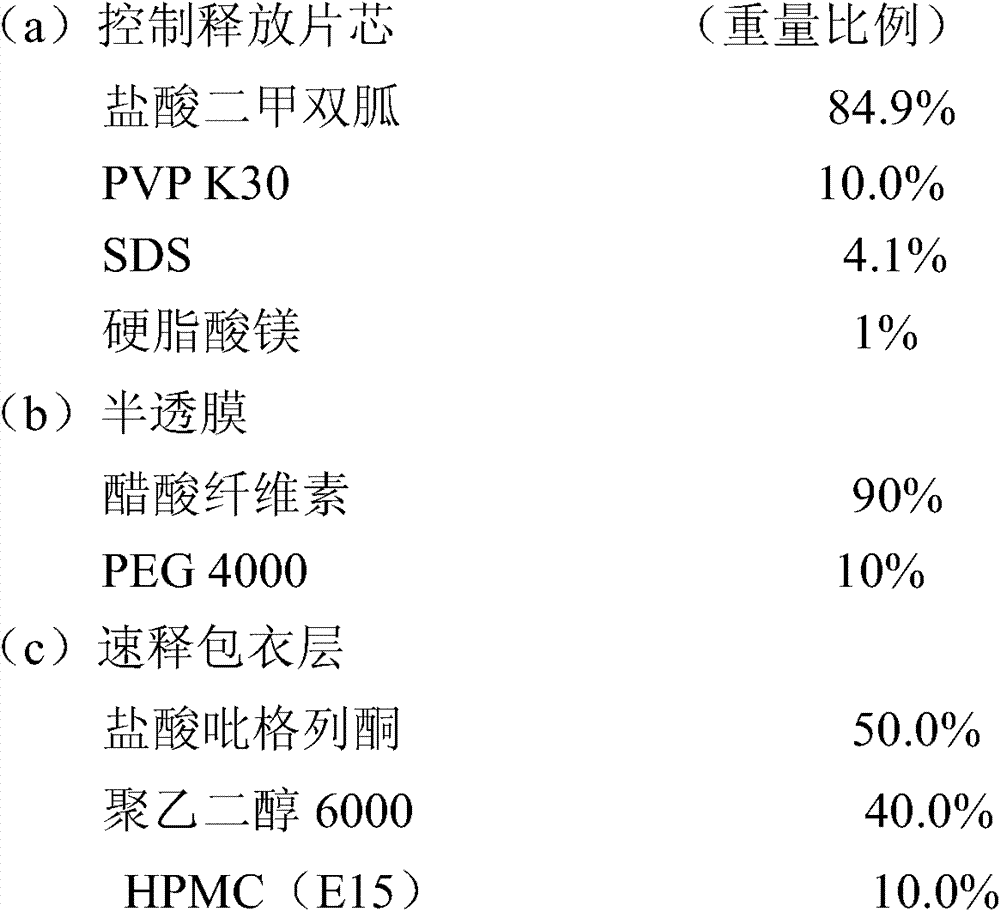
实施例3包含33.07mg盐酸吡格列酮(以吡格列酮计30mg)和1000mg的盐酸二甲双胍控制释放片剂的制备 Embodiment 3 comprises the preparation of 33.07mg pioglitazone hydrochloride (30mg in terms of pioglitazone) and 1000mg metformin hydrochloride controlled release tablet
制备方法: Preparation:
1.将1~5%的PVP K30配制成30%的水溶液,剩余的PVP与盐酸二甲双胍混合,以湿法制粒工艺将上述混合物制备成颗粒并压制成片芯; 1. Prepare 1-5% of PVP K30 into a 30% aqueous solution, mix the remaining PVP with metformin hydrochloride, prepare the above mixture into granules by wet granulation process and press into tablet cores;
2.半透膜包衣液的配制:先将醋酸纤维素配制成澄清的丙酮溶液,聚乙二醇4000配制成澄清的水溶液,将两种溶液混合,搅拌至澄清溶液,备用; 2. Preparation of semi-permeable membrane coating solution: first prepare cellulose acetate into a clear acetone solution, polyethylene glycol 4000 into a clear aqueous solution, mix the two solutions, stir until a clear solution, and set aside;
3.速释层包衣液的配制:将聚乙二醇6000溶于少量的水后加入95%的乙醇中,再分别加入HPMC和盐酸吡格列酮,分散后制得包衣液; 3. Preparation of immediate-release layer coating solution: Dissolve polyethylene glycol 6000 in a small amount of water and add 95% ethanol, then add HPMC and pioglitazone hydrochloride respectively, and disperse to prepare the coating solution;
4.用激光打孔机在片芯上下两面各打一个孔; 4. Use a laser drilling machine to punch a hole on the upper and lower sides of the core;
5.包衣结束后,于40℃条件下放置24小时,取出,即可。 5. After coating, place it at 40°C for 24 hours, then take it out. the
盐酸二甲双胍释放度的测定: Determination of Metformin Hydrochloride Release Rate:
测定法同实施例1,其的释放数据如下: Assay method is the same as embodiment 1, and its release data are as follows:
盐酸吡格列酮溶出度的测定: Determination of the dissolution rate of pioglitazone hydrochloride:
测定法同实施例1,其溶出结果如下: Assay method is with embodiment 1, and its stripping result is as follows:
15min 55.2% 15min 55.2%
30min 85.7% 30min 85.7%
45min 97.6% 45min 97.6%
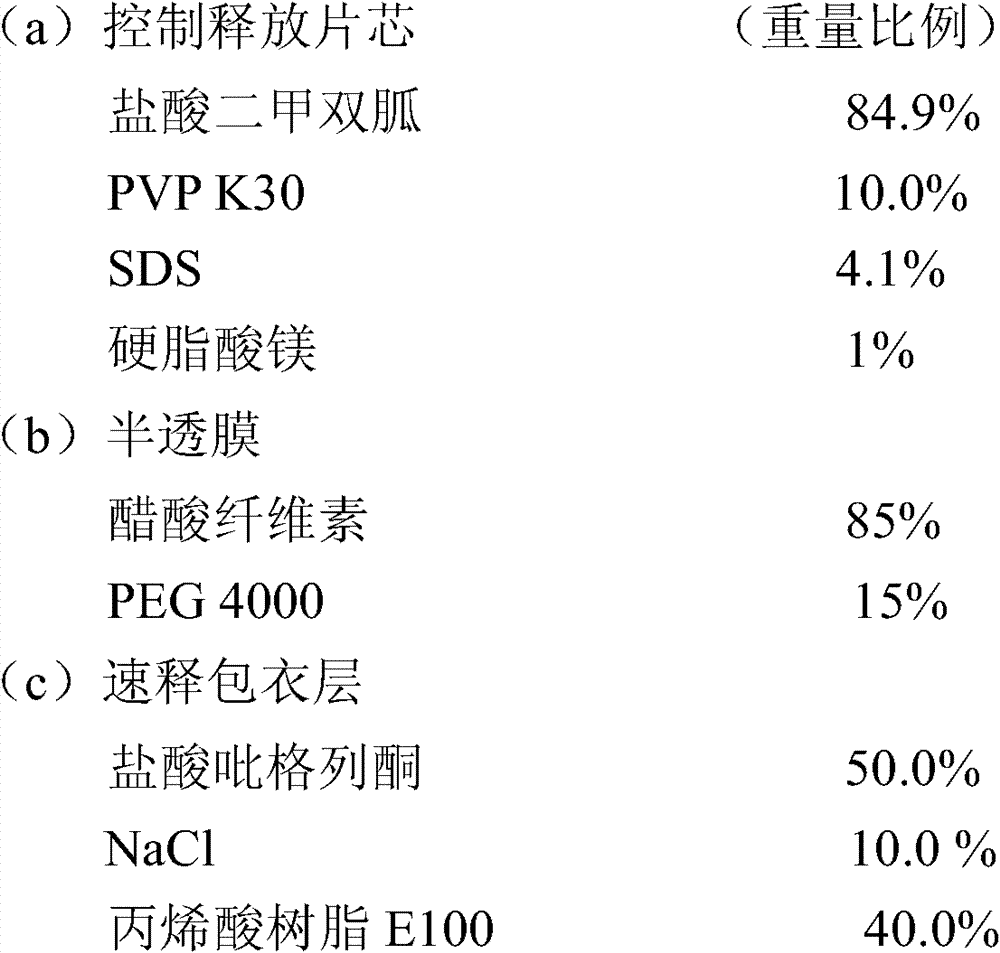
实施例4包含33.07mg盐酸吡格列酮(以吡格列酮计30mg)和1000mg的盐酸二甲双胍控制释放片剂的制备 Embodiment 4 comprises the preparation of 33.07mg pioglitazone hydrochloride (30mg in terms of pioglitazone) and 1000mg metformin hydrochloride controlled release tablet
制备方法: Preparation:
1.片芯、半透膜制备同实施例3。 1. The preparation of tablet core and semipermeable membrane is the same as in Example 3. the
2.速释层包衣液的配制:将NaCl溶于适量水中,丙烯酸树脂E100溶入95%的乙醇中,二者混合后再将盐酸吡格列酮分散其中后制得包衣液; 2. Preparation of immediate-release layer coating solution: dissolve NaCl in an appropriate amount of water, dissolve acrylic resin E100 in 95% ethanol, mix the two, and then disperse pioglitazone hydrochloride in it to prepare the coating solution;
3.用激光打孔机在片芯上下两面各打一个孔; 3. Use a laser drilling machine to punch a hole on the upper and lower sides of the core;
4.包衣结束后,于40℃条件下放置24小时,取出,即可。 4. After coating, place it at 40°C for 24 hours, then take it out. the
盐酸二甲双胍释放度的测定: Determination of Metformin Hydrochloride Release Rate:
检测方法同实施例1,其释放数据如下: Detection method is the same as embodiment 1, and its release data is as follows:
盐酸吡格列酮溶出度的测定: Determination of the dissolution rate of pioglitazone hydrochloride:
测定法同实施例1,其溶出结果如下: Assay method is with embodiment 1, and its stripping result is as follows:
15min 95.4% 15min 95.4%
30min 98.6% 30min 98.6%
45min 99.4% 45min 99.4%
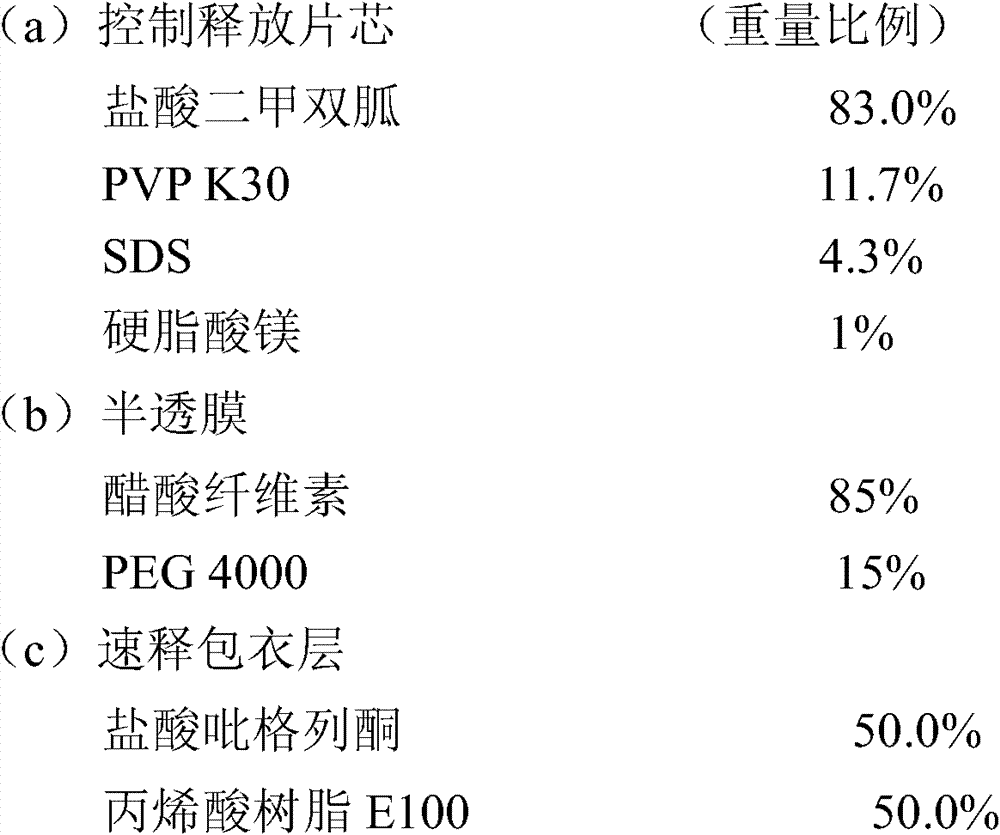
实施例5包含33.07mg盐酸吡格列酮(以吡格列酮计30mg)和1000mg的盐酸二甲双胍控制释放片剂的制备 Embodiment 5 comprises the preparation of 33.07mg pioglitazone hydrochloride (30mg in terms of pioglitazone) and 1000mg metformin hydrochloride controlled release tablet
制备方法: Preparation:
1.片芯、半透膜制备同实施例3。 1. The preparation of tablet core and semipermeable membrane is the same as in Example 3. the
2.速释层包衣液的配制:将乳糖溶于适量水中,丙烯酸树脂E100溶入95%的乙醇中,二者混合后再将盐酸吡格列酮分散其中后制得包衣液; 2. Preparation of immediate-release layer coating solution: dissolve lactose in an appropriate amount of water, dissolve acrylic resin E100 in 95% ethanol, mix the two, and then disperse pioglitazone hydrochloride in it to prepare the coating solution;
3.用激光打孔机在片芯上下两面各打一个孔; 3. Use a laser drilling machine to punch a hole on the upper and lower sides of the core;
4.包衣结束后,于40℃条件下放置24小时,取出,即可。 4. After coating, place it at 40°C for 24 hours, then take it out. the
盐酸二甲双胍释放度的测定: Determination of Metformin Hydrochloride Release Rate:
检测方法同实施例1,其释放数据如下: Detection method is the same as embodiment 1, and its release data is as follows:
盐酸吡格列酮溶出度的测定: Determination of the dissolution rate of pioglitazone hydrochloride:
测定法同实施例1,其溶出结果如下: Assay method is with embodiment 1, and its stripping result is as follows:
15min 89.5% 15min 89.5%
30min 95.7% 30min 95.7%
45min 96.8% 45min 96.8%
实施例6包含33.07mg盐酸吡格列酮(以吡格列酮计30mg)和1000mg的盐酸二甲双胍控制释放片剂的制备 Embodiment 6 comprises the preparation of 33.07mg pioglitazone hydrochloride (30mg in terms of pioglitazone) and 1000mg metformin hydrochloride controlled release tablet
(a)控制释放片芯同实施例5 (a) controlled release tablet core is the same as embodiment 5
(b)半透膜同实施例5 (b) semipermeable membrane is the same as embodiment 5
(c)速释包衣层 (c) Immediate release coating layer
盐酸吡格列酮 30.0% Pioglitazone Hydrochloride 30.0%
丙烯酸树脂E100 70.0% Acrylic resin E100 70.0%
制备方法:同实施例5 Preparation method: with embodiment 5
盐酸吡格列酮溶出度的测定: Determination of the dissolution rate of pioglitazone hydrochloride:
测定法同实施例1,其溶出结果如下: Assay method is with embodiment 1, and its stripping result is as follows:
15min 56.5% 15min 56.5%
30min 89.8% 30min 89.8%
45min 99.6% 45min 99.6%
实施例7包含16.54mg盐酸吡格列酮(以吡格列酮计15mg)和1000mg的盐酸二甲双胍控制释放片剂的制备 Embodiment 7 comprises the preparation of 16.54mg pioglitazone hydrochloride (15mg in terms of pioglitazone) and 1000mg metformin hydrochloride controlled release tablet
(a)控制释放片芯同实施例5 (a) controlled release tablet core is the same as embodiment 5
(b)半透膜同实施例5 (b) semipermeable membrane is the same as embodiment 5
(c)速释包衣层 (c) Immediate release coating layer
盐酸吡格列酮 70.0% Pioglitazone Hydrochloride 70.0%
丙烯酸树脂E100 30.0% Acrylic resin E100 30.0%
制备方法:同实施例5。 Preparation method: with embodiment 5. the
盐酸吡格列酮溶出度的测定: Determination of the dissolution rate of pioglitazone hydrochloride:
测定法同实施例1,其溶出结果如下: Assay method is with embodiment 1, and its stripping result is as follows:
15min 95.5% 15min 95.5%
30min 96.7% 30min 96.7%
45min 96.8% 45min 96.8%
实施例8包含16.54mg盐酸吡格列酮(以吡格列酮计15mg)和500mg的盐酸二甲双胍控制释放片剂的制备 Embodiment 8 comprises the preparation of 16.54mg pioglitazone hydrochloride (15mg in terms of pioglitazone) and 500mg metformin hydrochloride controlled release tablet
制备方法:同实施例5. Preparation method: with embodiment 5.
盐酸二甲双胍释放度的测定: Determination of Metformin Hydrochloride Release Rate:
检测方法同实施例1,其释放数据如下: Detection method is the same as embodiment 1, and its release data is as follows:
盐酸吡格列酮溶出度的测定: Determination of the dissolution rate of pioglitazone hydrochloride:
测定法同实施例1,其溶出结果如下: Assay method is with embodiment 1, and its stripping result is as follows:
15min 92.3% 15min 92.3%
30min 96.7% 30min 96.7%
45min 98.8% 。 98.8% in 45 minutes.
Claims (10)
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201610012615.5A CN105663130B (en) | 2011-04-15 | 2012-04-12 | A formulation having first and second active agents |
| CN2012101072455A CN102727459A (en) | 2011-04-15 | 2012-04-12 | A formulation having first and second active agents |
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201110098544 | 2011-04-15 | ||
| CN201110098544.2 | 2011-04-15 | ||
| CN2012101072455A CN102727459A (en) | 2011-04-15 | 2012-04-12 | A formulation having first and second active agents |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201610012615.5A Division CN105663130B (en) | 2011-04-15 | 2012-04-12 | A formulation having first and second active agents |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN102727459A true CN102727459A (en) | 2012-10-17 |
Family
ID=46984232
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN2012101072455A Pending CN102727459A (en) | 2011-04-15 | 2012-04-12 | A formulation having first and second active agents |
| CN201610012615.5A Expired - Fee Related CN105663130B (en) | 2011-04-15 | 2012-04-12 | A formulation having first and second active agents |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201610012615.5A Expired - Fee Related CN105663130B (en) | 2011-04-15 | 2012-04-12 | A formulation having first and second active agents |
Country Status (1)
| Country | Link |
|---|---|
| CN (2) | CN102727459A (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN114929210A (en) * | 2020-09-22 | 2022-08-19 | 赛乐医药科技有限公司 | Antidiabetic pharmaceutical composition and preparation method thereof |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1681496A (en) * | 2002-09-20 | 2005-10-12 | 安壮奇实验室公司 | New formulations of drugs containing biguanides and thiazolidinedione derivatives |
| CN101222912A (en) * | 2005-03-30 | 2008-07-16 | 华生制药公司 | New dosage forms of drugs comprising biguanides and thiazolidinedione derivatives |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20040081697A1 (en) * | 1998-11-12 | 2004-04-29 | Smithkline Beecham P.L.C. | Pharmaceutical composition for modified release of an insulin sensitiser and another antidiabetic agent |
| GB0318824D0 (en) * | 2003-08-11 | 2003-09-10 | Glaxo Group Ltd | Novel composition |
-
2012
- 2012-04-12 CN CN2012101072455A patent/CN102727459A/en active Pending
- 2012-04-12 CN CN201610012615.5A patent/CN105663130B/en not_active Expired - Fee Related
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1681496A (en) * | 2002-09-20 | 2005-10-12 | 安壮奇实验室公司 | New formulations of drugs containing biguanides and thiazolidinedione derivatives |
| CN101222912A (en) * | 2005-03-30 | 2008-07-16 | 华生制药公司 | New dosage forms of drugs comprising biguanides and thiazolidinedione derivatives |
Non-Patent Citations (2)
| Title |
|---|
| 张兆旺等: "《中药药剂学》", 31 January 2002, 中国中医药出版社 * |
| 彼得厄特: "《尤特奇(聚甲基丙烯酸酯)应用技术指南》", 31 August 2009, 化学工业出版社 * |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN114929210A (en) * | 2020-09-22 | 2022-08-19 | 赛乐医药科技有限公司 | Antidiabetic pharmaceutical composition and preparation method thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| CN105663130B (en) | 2019-08-09 |
| CN105663130A (en) | 2016-06-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR100762847B1 (en) | Multiple unit type sustained release oral preparation and method for preparing same | |
| AU2005320362B2 (en) | Controlled release complex formulation for oral administration of medicine for diabetes and method for the preparation thereof | |
| Kumar et al. | Oral extended release drug delivery system: A promising approach | |
| JP5775464B2 (en) | Delayed release oral dosage composition containing amorphous CDDO-ME | |
| US8246986B2 (en) | Drug coating providing high drug loading | |
| AU737121B2 (en) | Tramadol multiple unit formulations | |
| EP1515701B1 (en) | Process for the manufacture of multilayer tablet compositions comprising thiazolidinedione and biguanide | |
| JP4848558B2 (en) | Rapid-release tablets containing metformin hydrochloride | |
| JP2011148819A (en) | Novel pharmaceutical formulation containing biguanide and thiazolidinedione derivative | |
| CN109689036A (en) | Pulsed drug delivery system for the treatment of morning akinesia | |
| RU2007115544A (en) | NEW DRUGS FOR PROTON PUMP INHIBITORS IN THE FORM OF TABLETS WITH MODIFIED RELEASE | |
| MXPA04005667A (en) | Extended release pharmaceutical tablet of metformin. | |
| TWI573587B (en) | Composite formulation for oral administration comprising metformin and rosuvastatin | |
| MX2008002795A (en) | Extended release pharmaceutical composition of metformin and a process for producing it. | |
| WO2007114376A1 (en) | Solid pharmaceutical preparation | |
| RU2007115537A (en) | NEW DRUGS FOR PROTON PUMP INHIBITORS IN THE FORM OF PELLET WITH MODIFIED RELEASE | |
| CN105663130B (en) | A formulation having first and second active agents | |
| CN115607544A (en) | Sustained release composition of sacubitril and valsartan sodium, its preparation method and application | |
| CN105878256A (en) | Controlled-release preparation containing metformin hydrochloride and glimepiride and preparation method of controlled-release preparation | |
| AU2010328353B2 (en) | Partial dip coating of dosage forms for modified release | |
| HK1214136A1 (en) | Extended release compositions of an aminoalkyl nitrate | |
| RU2008150430A (en) | PHARMACEUTICAL COMPOSITIONS CONTAINING A COMBINATION OF PIPERIDINOALKANOL AND ANTI-FLOW AGENT | |
| CN102525991A (en) | Compound preparation containing pioglitazone hydrochloride and metformin hydrochloride and method for preparing compound preparation containing pioglitazone hydrochloride and metformin hydrochloride | |
| AU2004258949B2 (en) | Antibiotic product, use and formulation thereof | |
| JP2006528190A (en) | Antibiotic preparations, their use and preparation |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| ASS | Succession or assignment of patent right |
Free format text: FORMER OWNER: JIANGSU HANSOH MEDICAL RESEARCH INSTITUTE CO., LTD. Effective date: 20140618 |
|
| C41 | Transfer of patent application or patent right or utility model | ||
| TA01 | Transfer of patent application right |
Effective date of registration: 20140618 Address after: 222047 Lianyungang Development Zone, Jiangsu Applicant after: Jiangsu Hansoh Medical Group Co.,Ltd. Address before: Tenth Industrial Zone, Lianyungang, Jiangsu, China, 222047 Applicant before: Jiangsu Hansoh Medical Group Co.,Ltd. Applicant before: Jiangsu Hansoh Medical Research Institute Co., Ltd. |
|
| C12 | Rejection of a patent application after its publication | ||
| RJ01 | Rejection of invention patent application after publication |
Application publication date: 20121017 |