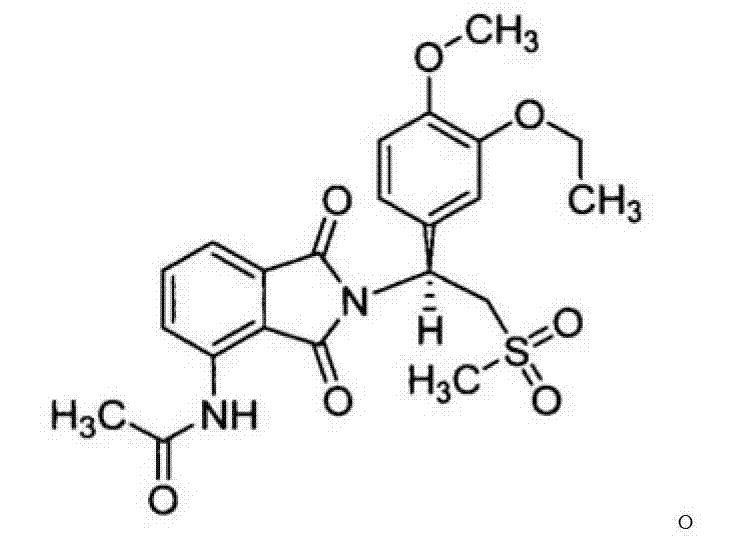
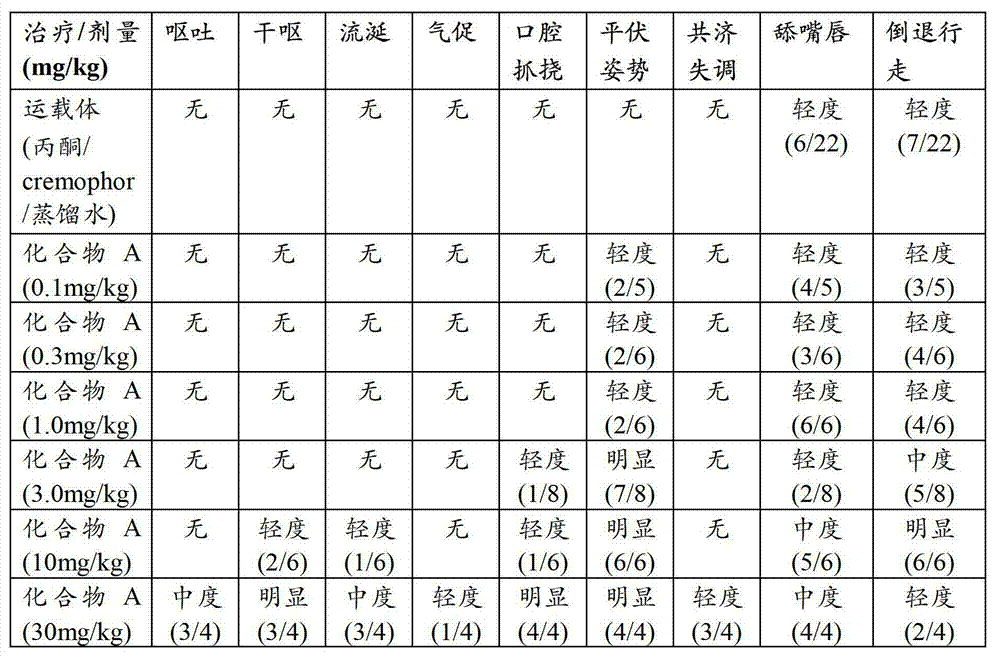
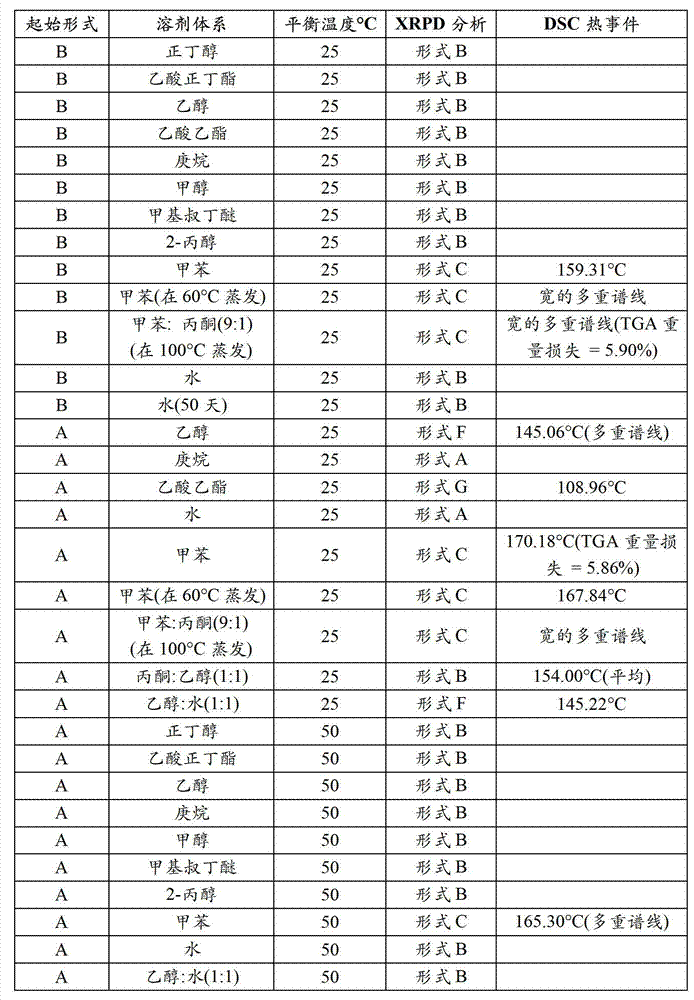
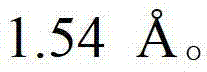CN102702070A - Solid form containing (+)-2-[1-(3-oxethyl-4-methoxyphenyl)-2-methylsulfonylethyl]-4-acetylaminoisoindoline-1,3-dione, composition and application thereof - Google Patents
Solid form containing (+)-2-[1-(3-oxethyl-4-methoxyphenyl)-2-methylsulfonylethyl]-4-acetylaminoisoindoline-1,3-dione, composition and application thereof Download PDFInfo
- Publication number
- CN102702070A CN102702070A CN2012101389499A CN201210138949A CN102702070A CN 102702070 A CN102702070 A CN 102702070A CN 2012101389499 A CN2012101389499 A CN 2012101389499A CN 201210138949 A CN201210138949 A CN 201210138949A CN 102702070 A CN102702070 A CN 102702070A
- Authority
- CN
- China
- Prior art keywords
- compd
- certain embodiments
- disease
- solid form
- present
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
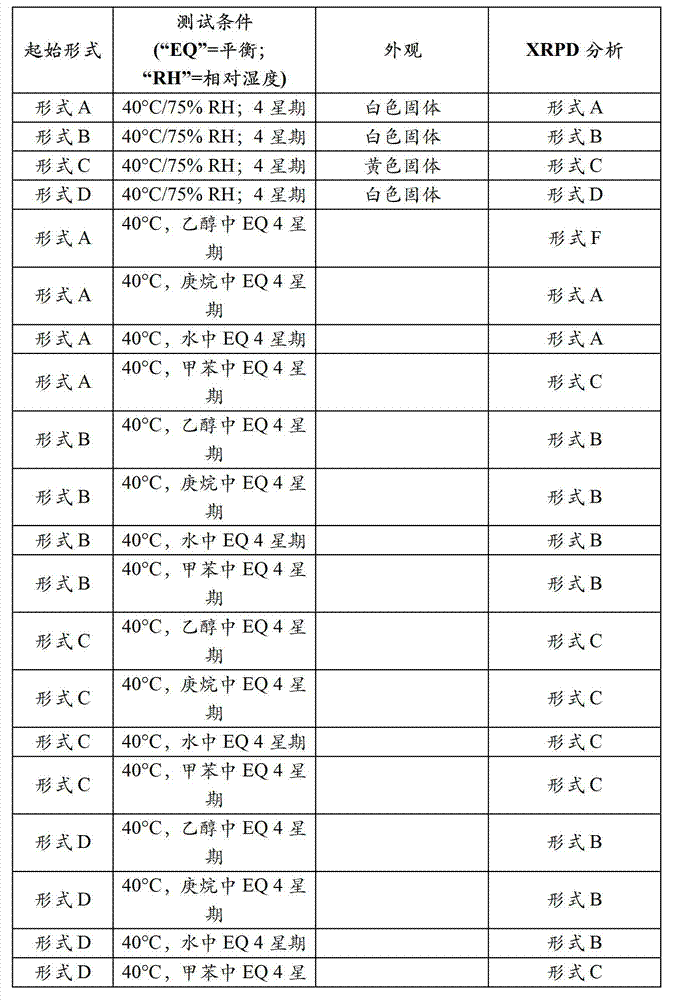
- 239000007787 solid Substances 0.000 title claims abstract description 207
- 239000000203 mixture Substances 0.000 title abstract description 105
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 claims abstract description 71
- 201000010099 disease Diseases 0.000 claims abstract description 61
- 108060008682 Tumor Necrosis Factor Proteins 0.000 claims abstract description 45
- 102000000852 Tumor Necrosis Factor-alpha Human genes 0.000 claims abstract description 45
- MZOFCQQQCNRIBI-VMXHOPILSA-N (3s)-4-[[(2s)-1-[[(2s)-1-[[(1s)-1-carboxy-2-hydroxyethyl]amino]-4-methyl-1-oxopentan-2-yl]amino]-5-(diaminomethylideneamino)-1-oxopentan-2-yl]amino]-3-[[2-[[(2s)-2,6-diaminohexanoyl]amino]acetyl]amino]-4-oxobutanoic acid Chemical compound OC[C@@H](C(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](CC(O)=O)NC(=O)CNC(=O)[C@@H](N)CCCCN MZOFCQQQCNRIBI-VMXHOPILSA-N 0.000 claims abstract description 44
- 101100296720 Dictyostelium discoideum Pde4 gene Proteins 0.000 claims abstract description 27
- 101100082610 Plasmodium falciparum (isolate 3D7) PDEdelta gene Proteins 0.000 claims abstract description 27
- 150000001875 compounds Chemical class 0.000 claims description 108
- 238000000634 powder X-ray diffraction Methods 0.000 claims description 82
- 238000004458 analytical method Methods 0.000 claims description 50
- 239000008194 pharmaceutical composition Substances 0.000 claims description 46
- 238000002360 preparation method Methods 0.000 claims description 40
- 239000003814 drug Substances 0.000 claims description 37
- 206010028980 Neoplasm Diseases 0.000 claims description 30
- 238000010521 absorption reaction Methods 0.000 claims description 20
- 230000001185 psoriatic effect Effects 0.000 claims description 19
- 230000001684 chronic effect Effects 0.000 claims description 18
- 201000011510 cancer Diseases 0.000 claims description 17
- 206010040070 Septic Shock Diseases 0.000 claims description 16
- 230000002757 inflammatory effect Effects 0.000 claims description 14
- 206010040047 Sepsis Diseases 0.000 claims description 13
- 208000006673 asthma Diseases 0.000 claims description 13
- 208000013223 septicemia Diseases 0.000 claims description 13
- 208000004232 Enteritis Diseases 0.000 claims description 11
- 206010039073 rheumatoid arthritis Diseases 0.000 claims description 11
- 208000023275 Autoimmune disease Diseases 0.000 claims description 10
- 201000004624 Dermatitis Diseases 0.000 claims description 10
- 206010035664 Pneumonia Diseases 0.000 claims description 10
- 208000006454 hepatitis Diseases 0.000 claims description 10
- 231100000283 hepatitis Toxicity 0.000 claims description 10
- 201000008482 osteoarthritis Diseases 0.000 claims description 10
- 230000036303 septic shock Effects 0.000 claims description 9
- 208000011580 syndromic disease Diseases 0.000 claims description 9
- 208000006545 Chronic Obstructive Pulmonary Disease Diseases 0.000 claims description 8
- 206010009900 Colitis ulcerative Diseases 0.000 claims description 8
- 201000006704 Ulcerative Colitis Diseases 0.000 claims description 8
- 206010001052 Acute respiratory distress syndrome Diseases 0.000 claims description 7
- 208000019838 Blood disease Diseases 0.000 claims description 7
- 206010006895 Cachexia Diseases 0.000 claims description 7
- 208000013616 Respiratory Distress Syndrome Diseases 0.000 claims description 7
- 208000011341 adult acute respiratory distress syndrome Diseases 0.000 claims description 7
- 201000000028 adult respiratory distress syndrome Diseases 0.000 claims description 7
- 208000019664 bone resorption disease Diseases 0.000 claims description 7
- 208000026500 emaciation Diseases 0.000 claims description 7
- 208000014951 hematologic disease Diseases 0.000 claims description 7
- 208000018706 hematopoietic system disease Diseases 0.000 claims description 7
- 230000035939 shock Effects 0.000 claims description 7
- 206010002556 Ankylosing Spondylitis Diseases 0.000 claims description 6
- 201000003883 Cystic fibrosis Diseases 0.000 claims description 6
- 206010024229 Leprosy Diseases 0.000 claims description 6
- 201000009906 Meningitis Diseases 0.000 claims description 6
- 208000034578 Multiple myelomas Diseases 0.000 claims description 6
- 208000001132 Osteoporosis Diseases 0.000 claims description 6
- 206010035226 Plasma cell myeloma Diseases 0.000 claims description 6
- 208000019155 Radiation injury Diseases 0.000 claims description 6
- 206010063837 Reperfusion injury Diseases 0.000 claims description 6
- 206010052779 Transplant rejections Diseases 0.000 claims description 6
- 208000027418 Wounds and injury Diseases 0.000 claims description 6
- 230000006378 damage Effects 0.000 claims description 6
- 230000003176 fibrotic effect Effects 0.000 claims description 6
- 230000001435 haemodynamic effect Effects 0.000 claims description 6
- 230000000222 hyperoxic effect Effects 0.000 claims description 6
- 208000014674 injury Diseases 0.000 claims description 6
- 201000006417 multiple sclerosis Diseases 0.000 claims description 6
- 201000000596 systemic lupus erythematosus Diseases 0.000 claims description 6
- 208000009137 Behcet syndrome Diseases 0.000 claims description 5
- 206010012438 Dermatitis atopic Diseases 0.000 claims description 5
- 201000008937 atopic dermatitis Diseases 0.000 claims description 5
- 230000001976 improved effect Effects 0.000 claims description 5
- 201000001441 melanoma Diseases 0.000 claims description 5
- 208000005615 Interstitial Cystitis Diseases 0.000 claims description 4
- 201000001263 Psoriatic Arthritis Diseases 0.000 claims description 4
- 208000036824 Psoriatic arthropathy Diseases 0.000 claims description 4
- 208000035250 cutaneous malignant susceptibility to 1 melanoma Diseases 0.000 claims description 4
- 208000005017 glioblastoma Diseases 0.000 claims description 4
- 206010025135 lupus erythematosus Diseases 0.000 claims description 4
- 208000009329 Graft vs Host Disease Diseases 0.000 claims description 3
- 208000024908 graft versus host disease Diseases 0.000 claims description 3
- 201000009273 Endometriosis Diseases 0.000 claims description 2
- 208000007465 Giant cell arteritis Diseases 0.000 claims description 2
- 208000031671 Large B-Cell Diffuse Lymphoma Diseases 0.000 claims description 2
- 201000009053 Neurodermatitis Diseases 0.000 claims description 2
- 206010037779 Radiculopathy Diseases 0.000 claims description 2
- 208000021386 Sjogren Syndrome Diseases 0.000 claims description 2
- 206010046851 Uveitis Diseases 0.000 claims description 2
- 208000003728 Vulvodynia Diseases 0.000 claims description 2
- 206010069055 Vulvovaginal pain Diseases 0.000 claims description 2
- 201000001981 dermatomyositis Diseases 0.000 claims description 2
- 206010012818 diffuse large B-cell lymphoma Diseases 0.000 claims description 2
- 201000008319 inclusion body myositis Diseases 0.000 claims description 2
- 201000011486 lichen planus Diseases 0.000 claims description 2
- 208000005987 polymyositis Diseases 0.000 claims description 2
- 201000007094 prostatitis Diseases 0.000 claims description 2
- 208000017940 prurigo nodularis Diseases 0.000 claims description 2
- 208000009954 pyoderma gangrenosum Diseases 0.000 claims description 2
- 201000000306 sarcoidosis Diseases 0.000 claims description 2
- 206010043207 temporal arteritis Diseases 0.000 claims description 2
- 208000018737 Parkinson disease Diseases 0.000 claims 1
- 238000011282 treatment Methods 0.000 abstract description 86
- 238000000034 method Methods 0.000 abstract description 70
- 230000002265 prevention Effects 0.000 abstract description 44
- 208000024891 symptom Diseases 0.000 abstract description 11
- 230000002401 inhibitory effect Effects 0.000 abstract description 2
- 229940126062 Compound A Drugs 0.000 description 91
- NLDMNSXOCDLTTB-UHFFFAOYSA-N Heterophylliin A Natural products O1C2COC(=O)C3=CC(O)=C(O)C(O)=C3C3=C(O)C(O)=C(O)C=C3C(=O)OC2C(OC(=O)C=2C=C(O)C(O)=C(O)C=2)C(O)C1OC(=O)C1=CC(O)=C(O)C(O)=C1 NLDMNSXOCDLTTB-UHFFFAOYSA-N 0.000 description 91
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 83
- 239000002904 solvent Substances 0.000 description 81
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 70
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 66
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 64
- 210000004027 cell Anatomy 0.000 description 52
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 49
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 49
- 238000009472 formulation Methods 0.000 description 46
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 45
- ZWEHNKRNPOVVGH-UHFFFAOYSA-N 2-Butanone Chemical compound CCC(C)=O ZWEHNKRNPOVVGH-UHFFFAOYSA-N 0.000 description 40
- 230000008859 change Effects 0.000 description 38
- 239000013078 crystal Substances 0.000 description 38
- 239000003795 chemical substances by application Substances 0.000 description 37
- IMNFDUFMRHMDMM-UHFFFAOYSA-N N-Heptane Chemical compound CCCCCCC IMNFDUFMRHMDMM-UHFFFAOYSA-N 0.000 description 36
- 230000015572 biosynthetic process Effects 0.000 description 35
- 238000005755 formation reaction Methods 0.000 description 35
- 239000000463 material Substances 0.000 description 35
- 239000000243 solution Substances 0.000 description 35
- 238000011160 research Methods 0.000 description 31
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical group CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 30
- 230000001939 inductive effect Effects 0.000 description 30
- 238000012360 testing method Methods 0.000 description 30
- 235000019441 ethanol Nutrition 0.000 description 29
- -1 alkyl group acid amides Chemical class 0.000 description 28
- 238000002425 crystallisation Methods 0.000 description 26
- 239000002158 endotoxin Substances 0.000 description 26
- BZLVMXJERCGZMT-UHFFFAOYSA-N Methyl tert-butyl ether Chemical compound COC(C)(C)C BZLVMXJERCGZMT-UHFFFAOYSA-N 0.000 description 24
- 230000003203 everyday effect Effects 0.000 description 24
- 239000002512 suppressor factor Substances 0.000 description 24
- 239000003981 vehicle Substances 0.000 description 21
- 239000000651 prodrug Substances 0.000 description 20
- 229940002612 prodrug Drugs 0.000 description 20
- 230000008025 crystallization Effects 0.000 description 19
- 239000002245 particle Substances 0.000 description 19
- 239000012071 phase Substances 0.000 description 19
- 239000000843 powder Substances 0.000 description 19
- 230000001225 therapeutic effect Effects 0.000 description 19
- 230000000694 effects Effects 0.000 description 18
- 239000000047 product Substances 0.000 description 18
- 230000001154 acute effect Effects 0.000 description 17
- 230000008021 deposition Effects 0.000 description 17
- 230000007062 hydrolysis Effects 0.000 description 17
- 238000006460 hydrolysis reaction Methods 0.000 description 17
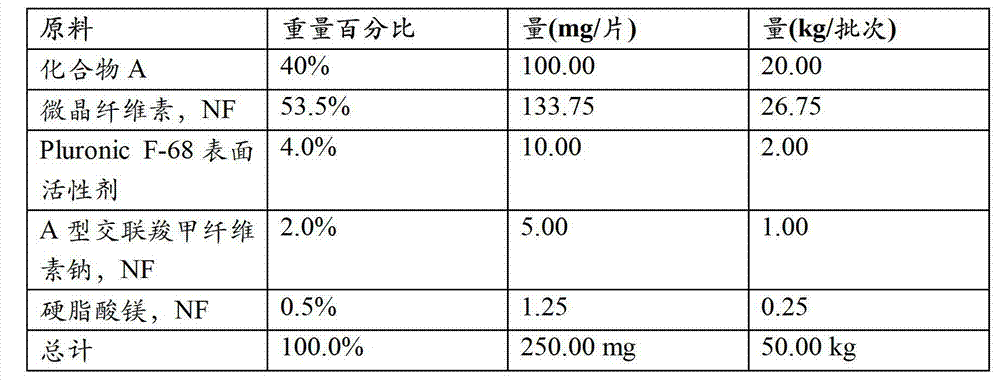
- 239000003826 tablet Substances 0.000 description 17
- 210000003714 granulocyte Anatomy 0.000 description 16
- 230000003448 neutrophilic effect Effects 0.000 description 16
- 238000001179 sorption measurement Methods 0.000 description 16
- 239000000126 substance Substances 0.000 description 16
- 230000006399 behavior Effects 0.000 description 15
- 239000002775 capsule Substances 0.000 description 15
- 230000000295 complement effect Effects 0.000 description 14
- 241001465754 Metazoa Species 0.000 description 13
- DKPFZGUDAPQIHT-UHFFFAOYSA-N butyl acetate Chemical compound CCCCOC(C)=O DKPFZGUDAPQIHT-UHFFFAOYSA-N 0.000 description 13
- 238000005516 engineering process Methods 0.000 description 13
- 239000007788 liquid Substances 0.000 description 13
- 230000008569 process Effects 0.000 description 13
- 231100001274 therapeutic index Toxicity 0.000 description 13
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 12
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 12
- 210000001744 T-lymphocyte Anatomy 0.000 description 12
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 12
- 230000009471 action Effects 0.000 description 12
- 230000005764 inhibitory process Effects 0.000 description 12
- 102000004127 Cytokines Human genes 0.000 description 11
- 108090000695 Cytokines Proteins 0.000 description 11
- 206010061218 Inflammation Diseases 0.000 description 11
- 210000004369 blood Anatomy 0.000 description 11
- 239000008280 blood Substances 0.000 description 11
- 230000007423 decrease Effects 0.000 description 11
- 239000002895 emetic Substances 0.000 description 11
- 238000001704 evaporation Methods 0.000 description 11
- 230000004054 inflammatory process Effects 0.000 description 11
- 210000004072 lung Anatomy 0.000 description 11
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 10
- 241000282341 Mustela putorius furo Species 0.000 description 10
- 206010047700 Vomiting Diseases 0.000 description 10
- 238000001574 biopsy Methods 0.000 description 10
- 239000002178 crystalline material Substances 0.000 description 10
- IJGRMHOSHXDMSA-UHFFFAOYSA-N nitrogen Substances N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 10
- 239000003921 oil Substances 0.000 description 10
- 238000002336 sorption--desorption measurement Methods 0.000 description 10
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 9
- 229920000168 Microcrystalline cellulose Polymers 0.000 description 9
- 206010029379 Neutrophilia Diseases 0.000 description 9
- 201000004681 Psoriasis Diseases 0.000 description 9
- CKMXBZGNNVIXHC-UHFFFAOYSA-L ammonium magnesium phosphate hexahydrate Chemical compound [NH4+].O.O.O.O.O.O.[Mg+2].[O-]P([O-])([O-])=O CKMXBZGNNVIXHC-UHFFFAOYSA-L 0.000 description 9
- 239000012296 anti-solvent Substances 0.000 description 9
- 238000006243 chemical reaction Methods 0.000 description 9
- 229940079593 drug Drugs 0.000 description 9
- 230000008020 evaporation Effects 0.000 description 9
- 210000002216 heart Anatomy 0.000 description 9
- 230000006872 improvement Effects 0.000 description 9
- 239000003112 inhibitor Substances 0.000 description 9
- 239000002207 metabolite Substances 0.000 description 9
- 235000019813 microcrystalline cellulose Nutrition 0.000 description 9
- 239000008108 microcrystalline cellulose Substances 0.000 description 9
- 229940016286 microcrystalline cellulose Drugs 0.000 description 9
- 235000019198 oils Nutrition 0.000 description 9
- 238000007614 solvation Methods 0.000 description 9
- 229910052567 struvite Inorganic materials 0.000 description 9
- 210000001519 tissue Anatomy 0.000 description 9
- 230000008673 vomiting Effects 0.000 description 9
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 8
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 8
- 108010002350 Interleukin-2 Proteins 0.000 description 8
- 102000000588 Interleukin-2 Human genes 0.000 description 8
- 235000013339 cereals Nutrition 0.000 description 8
- 238000001816 cooling Methods 0.000 description 8
- 230000008034 disappearance Effects 0.000 description 8
- 150000002148 esters Chemical class 0.000 description 8
- 230000005496 eutectics Effects 0.000 description 8
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 8
- 239000006186 oral dosage form Substances 0.000 description 8
- 238000012856 packing Methods 0.000 description 8
- 239000006201 parenteral dosage form Substances 0.000 description 8
- 150000003839 salts Chemical class 0.000 description 8
- 210000003491 skin Anatomy 0.000 description 8
- 229940124597 therapeutic agent Drugs 0.000 description 8
- 102100035793 CD83 antigen Human genes 0.000 description 7
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 7
- 101000946856 Homo sapiens CD83 antigen Proteins 0.000 description 7
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 7
- 241000124008 Mammalia Species 0.000 description 7
- 108090001050 Phosphoric Diester Hydrolases Proteins 0.000 description 7
- 102000004861 Phosphoric Diester Hydrolases Human genes 0.000 description 7
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 7
- 230000002411 adverse Effects 0.000 description 7
- 239000002585 base Substances 0.000 description 7
- 229910002091 carbon monoxide Inorganic materials 0.000 description 7
- PRQROPMIIGLWRP-BZSNNMDCSA-N chemotactic peptide Chemical compound CSCC[C@H](NC=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@H](C(O)=O)CC1=CC=CC=C1 PRQROPMIIGLWRP-BZSNNMDCSA-N 0.000 description 7
- 238000009792 diffusion process Methods 0.000 description 7
- 210000004400 mucous membrane Anatomy 0.000 description 7
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Substances [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 7
- 238000001953 recrystallisation Methods 0.000 description 7
- 238000003860 storage Methods 0.000 description 7
- 241000894006 Bacteria Species 0.000 description 6
- IAZDPXIOMUYVGZ-WFGJKAKNSA-N Dimethyl sulfoxide Chemical compound [2H]C([2H])([2H])S(=O)C([2H])([2H])[2H] IAZDPXIOMUYVGZ-WFGJKAKNSA-N 0.000 description 6
- 102000004190 Enzymes Human genes 0.000 description 6
- 108090000790 Enzymes Proteins 0.000 description 6
- 102100037850 Interferon gamma Human genes 0.000 description 6
- 108010074328 Interferon-gamma Proteins 0.000 description 6
- 102100029438 Nitric oxide synthase, inducible Human genes 0.000 description 6
- 101710089543 Nitric oxide synthase, inducible Proteins 0.000 description 6
- OIRDTQYFTABQOQ-KQYNXXCUSA-N adenosine Chemical compound C1=NC=2C(N)=NC=NC=2N1[C@@H]1O[C@H](CO)[C@@H](O)[C@H]1O OIRDTQYFTABQOQ-KQYNXXCUSA-N 0.000 description 6
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 6
- 239000005557 antagonist Substances 0.000 description 6
- 230000008901 benefit Effects 0.000 description 6
- 239000003153 chemical reaction reagent Substances 0.000 description 6
- 239000002552 dosage form Substances 0.000 description 6
- 239000000839 emulsion Substances 0.000 description 6
- 229940088598 enzyme Drugs 0.000 description 6
- 210000002615 epidermis Anatomy 0.000 description 6
- 238000002474 experimental method Methods 0.000 description 6
- 238000004128 high performance liquid chromatography Methods 0.000 description 6
- 210000003734 kidney Anatomy 0.000 description 6
- 239000000314 lubricant Substances 0.000 description 6
- 238000005259 measurement Methods 0.000 description 6
- 210000000214 mouth Anatomy 0.000 description 6
- 210000000582 semen Anatomy 0.000 description 6
- RNLYVRKMBPOHNI-UNDPIUDWSA-N (2s)-5-(diaminomethylideneamino)-n-[11-[4-[4-[4-[11-[[2-[4-[(2r)-2-hydroxypropyl]triazol-1-yl]acetyl]amino]undecanoyl]piperazin-1-yl]-6-[2-[2-(2-prop-2-ynoxyethoxy)ethoxy]ethylamino]-1,3,5-triazin-2-yl]piperazin-1-yl]-11-oxoundecyl]-2-[4-[(2s)-2-methylbut Chemical compound N1=NC(C[C@@H](C)CC)=CN1[C@@H](CCCN=C(N)N)C(=O)NCCCCCCCCCCC(=O)N1CCN(C=2N=C(N=C(NCCOCCOCCOCC#C)N=2)N2CCN(CC2)C(=O)CCCCCCCCCCNC(=O)CN2N=NC(C[C@@H](C)O)=C2)CC1 RNLYVRKMBPOHNI-UNDPIUDWSA-N 0.000 description 5
- 101100407335 Dictyostelium discoideum pde7 gene Proteins 0.000 description 5
- 101001046686 Homo sapiens Integrin alpha-M Proteins 0.000 description 5
- 101000935040 Homo sapiens Integrin beta-2 Proteins 0.000 description 5
- 102100022338 Integrin alpha-M Human genes 0.000 description 5
- 102100022297 Integrin alpha-X Human genes 0.000 description 5
- 102100025390 Integrin beta-2 Human genes 0.000 description 5
- 108090001007 Interleukin-8 Proteins 0.000 description 5
- 102000004890 Interleukin-8 Human genes 0.000 description 5
- 208000014767 Myeloproliferative disease Diseases 0.000 description 5
- 229920002472 Starch Polymers 0.000 description 5
- 238000002441 X-ray diffraction Methods 0.000 description 5
- 229920000392 Zymosan Polymers 0.000 description 5
- 239000002253 acid Substances 0.000 description 5
- 239000000443 aerosol Substances 0.000 description 5
- 238000007605 air drying Methods 0.000 description 5
- 150000001412 amines Chemical class 0.000 description 5
- 230000004071 biological effect Effects 0.000 description 5
- 238000013270 controlled release Methods 0.000 description 5
- 238000012258 culturing Methods 0.000 description 5
- 239000003405 delayed action preparation Substances 0.000 description 5
- 210000004907 gland Anatomy 0.000 description 5
- 208000027866 inflammatory disease Diseases 0.000 description 5
- 239000007924 injection Substances 0.000 description 5
- 238000002347 injection Methods 0.000 description 5
- 229910052757 nitrogen Inorganic materials 0.000 description 5
- 239000002953 phosphate buffered saline Substances 0.000 description 5
- 230000000704 physical effect Effects 0.000 description 5
- 229940068196 placebo Drugs 0.000 description 5
- 239000000902 placebo Substances 0.000 description 5
- 108090000765 processed proteins & peptides Proteins 0.000 description 5
- 210000000664 rectum Anatomy 0.000 description 5
- 210000002966 serum Anatomy 0.000 description 5
- 239000002002 slurry Substances 0.000 description 5
- 229940032147 starch Drugs 0.000 description 5
- 239000008107 starch Substances 0.000 description 5
- 235000019698 starch Nutrition 0.000 description 5
- 238000005406 washing Methods 0.000 description 5
- KMOUUZVZFBCRAM-OLQVQODUSA-N (3as,7ar)-3a,4,7,7a-tetrahydro-2-benzofuran-1,3-dione Chemical compound C1C=CC[C@@H]2C(=O)OC(=O)[C@@H]21 KMOUUZVZFBCRAM-OLQVQODUSA-N 0.000 description 4
- 238000005160 1H NMR spectroscopy Methods 0.000 description 4
- KXDHJXZQYSOELW-UHFFFAOYSA-M Carbamate Chemical compound NC([O-])=O KXDHJXZQYSOELW-UHFFFAOYSA-M 0.000 description 4
- 206010007559 Cardiac failure congestive Diseases 0.000 description 4
- 229920002785 Croscarmellose sodium Polymers 0.000 description 4
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 4
- 206010019280 Heart failures Diseases 0.000 description 4
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 4
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 4
- 229920000881 Modified starch Polymers 0.000 description 4
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 4
- 239000008156 Ringer's lactate solution Substances 0.000 description 4
- DPXJVFZANSGRMM-UHFFFAOYSA-N acetic acid;2,3,4,5,6-pentahydroxyhexanal;sodium Chemical compound [Na].CC(O)=O.OCC(O)C(O)C(O)C(O)C=O DPXJVFZANSGRMM-UHFFFAOYSA-N 0.000 description 4
- 239000003005 anticarcinogenic agent Substances 0.000 description 4
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 4
- 210000004556 brain Anatomy 0.000 description 4
- BMLSTPRTEKLIPM-UHFFFAOYSA-I calcium;potassium;disodium;hydrogen carbonate;dichloride;dihydroxide;hydrate Chemical compound O.[OH-].[OH-].[Na+].[Na+].[Cl-].[Cl-].[K+].[Ca+2].OC([O-])=O BMLSTPRTEKLIPM-UHFFFAOYSA-I 0.000 description 4
- 229940063834 carboxymethylcellulose sodium Drugs 0.000 description 4
- 210000000038 chest Anatomy 0.000 description 4
- 238000005336 cracking Methods 0.000 description 4
- 238000000354 decomposition reaction Methods 0.000 description 4
- 210000003238 esophagus Anatomy 0.000 description 4
- 210000001508 eye Anatomy 0.000 description 4
- 239000012530 fluid Substances 0.000 description 4
- 239000000499 gel Substances 0.000 description 4
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 description 4
- 210000000936 intestine Anatomy 0.000 description 4
- 210000000867 larynx Anatomy 0.000 description 4
- VNYSSYRCGWBHLG-AMOLWHMGSA-N leukotriene B4 Chemical compound CCCCC\C=C/C[C@@H](O)\C=C\C=C\C=C/[C@@H](O)CCCC(O)=O VNYSSYRCGWBHLG-AMOLWHMGSA-N 0.000 description 4
- 210000004185 liver Anatomy 0.000 description 4
- 235000019359 magnesium stearate Nutrition 0.000 description 4
- 239000002609 medium Substances 0.000 description 4
- 108020004999 messenger RNA Proteins 0.000 description 4
- 230000004048 modification Effects 0.000 description 4
- 238000012986 modification Methods 0.000 description 4
- 238000007911 parenteral administration Methods 0.000 description 4
- 229910052697 platinum Inorganic materials 0.000 description 4
- 238000002203 pretreatment Methods 0.000 description 4
- 238000010992 reflux Methods 0.000 description 4
- 239000012266 salt solution Substances 0.000 description 4
- 238000012216 screening Methods 0.000 description 4
- 238000007390 skin biopsy Methods 0.000 description 4
- 239000011734 sodium Substances 0.000 description 4
- 229910052708 sodium Inorganic materials 0.000 description 4
- 239000008354 sodium chloride injection Substances 0.000 description 4
- PFNFFQXMRSDOHW-UHFFFAOYSA-N spermine Chemical compound NCCCNCCCCNCCCN PFNFFQXMRSDOHW-UHFFFAOYSA-N 0.000 description 4
- 210000002784 stomach Anatomy 0.000 description 4
- 201000008827 tuberculosis Diseases 0.000 description 4
- 230000003612 virological effect Effects 0.000 description 4
- 238000001644 13C nuclear magnetic resonance spectroscopy Methods 0.000 description 3
- QNKJFXARIMSDBR-UHFFFAOYSA-N 3-[2-[bis(2-chloroethyl)amino]ethyl]-1,3-diazaspiro[4.5]decane-2,4-dione Chemical compound O=C1N(CCN(CCCl)CCCl)C(=O)NC11CCCCC1 QNKJFXARIMSDBR-UHFFFAOYSA-N 0.000 description 3
- WGLQHUKCXBXUDV-UHFFFAOYSA-N 3-aminophthalic acid Chemical compound NC1=CC=CC(C(O)=O)=C1C(O)=O WGLQHUKCXBXUDV-UHFFFAOYSA-N 0.000 description 3
- HIQIXEFWDLTDED-UHFFFAOYSA-N 4-hydroxy-1-piperidin-4-ylpyrrolidin-2-one Chemical compound O=C1CC(O)CN1C1CCNCC1 HIQIXEFWDLTDED-UHFFFAOYSA-N 0.000 description 3
- 239000002126 C01EB10 - Adenosine Substances 0.000 description 3
- 208000031229 Cardiomyopathies Diseases 0.000 description 3
- OIFBSDVPJOWBCH-UHFFFAOYSA-N Diethyl carbonate Chemical compound CCOC(=O)OCC OIFBSDVPJOWBCH-UHFFFAOYSA-N 0.000 description 3
- ZQZFYGIXNQKOAV-OCEACIFDSA-N Droloxifene Chemical compound C=1C=CC=CC=1C(/CC)=C(C=1C=C(O)C=CC=1)\C1=CC=C(OCCN(C)C)C=C1 ZQZFYGIXNQKOAV-OCEACIFDSA-N 0.000 description 3
- 238000002965 ELISA Methods 0.000 description 3
- 102000006354 HLA-DR Antigens Human genes 0.000 description 3
- 108010058597 HLA-DR Antigens Proteins 0.000 description 3
- 102000015271 Intercellular Adhesion Molecule-1 Human genes 0.000 description 3
- 108010064593 Intercellular Adhesion Molecule-1 Proteins 0.000 description 3
- 108010050904 Interferons Proteins 0.000 description 3
- 102000014150 Interferons Human genes 0.000 description 3
- 102000003814 Interleukin-10 Human genes 0.000 description 3
- 108090000174 Interleukin-10 Proteins 0.000 description 3
- 108010000817 Leuprolide Proteins 0.000 description 3
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 3
- LRHPLDYGYMQRHN-UHFFFAOYSA-N N-Butanol Chemical compound CCCCO LRHPLDYGYMQRHN-UHFFFAOYSA-N 0.000 description 3
- WHNWPMSKXPGLAX-UHFFFAOYSA-N N-Vinyl-2-pyrrolidone Chemical compound C=CN1CCCC1=O WHNWPMSKXPGLAX-UHFFFAOYSA-N 0.000 description 3
- 208000001388 Opportunistic Infections Diseases 0.000 description 3
- 241000283973 Oryctolagus cuniculus Species 0.000 description 3
- 208000002193 Pain Diseases 0.000 description 3
- 235000019483 Peanut oil Nutrition 0.000 description 3
- 239000012980 RPMI-1640 medium Substances 0.000 description 3
- 206010038776 Retching Diseases 0.000 description 3
- OTKJDMGTUTTYMP-ROUUACIJSA-N Safingol ( L-threo-sphinganine) Chemical compound CCCCCCCCCCCCCCC[C@H](O)[C@@H](N)CO OTKJDMGTUTTYMP-ROUUACIJSA-N 0.000 description 3
- BZHJMEDXRYGGRV-UHFFFAOYSA-N Vinyl chloride Chemical compound ClC=C BZHJMEDXRYGGRV-UHFFFAOYSA-N 0.000 description 3
- 230000002159 abnormal effect Effects 0.000 description 3
- 229960000583 acetic acid Drugs 0.000 description 3
- 239000013543 active substance Substances 0.000 description 3
- 229960005305 adenosine Drugs 0.000 description 3
- 210000005091 airway smooth muscle Anatomy 0.000 description 3
- 235000010443 alginic acid Nutrition 0.000 description 3
- 229920000615 alginic acid Polymers 0.000 description 3
- 150000001408 amides Chemical class 0.000 description 3
- 230000003110 anti-inflammatory effect Effects 0.000 description 3
- 229940125715 antihistaminic agent Drugs 0.000 description 3
- 239000000739 antihistaminic agent Substances 0.000 description 3
- 210000000988 bone and bone Anatomy 0.000 description 3
- 210000000481 breast Anatomy 0.000 description 3
- 239000000872 buffer Substances 0.000 description 3
- 206010009887 colitis Diseases 0.000 description 3
- 210000001072 colon Anatomy 0.000 description 3
- 238000013016 damping Methods 0.000 description 3
- 238000004807 desolvation Methods 0.000 description 3
- 238000009826 distribution Methods 0.000 description 3
- 238000001035 drying Methods 0.000 description 3
- 210000003725 endotheliocyte Anatomy 0.000 description 3
- 239000003623 enhancer Substances 0.000 description 3
- 238000001914 filtration Methods 0.000 description 3
- OVBPIULPVIDEAO-LBPRGKRZSA-N folic acid Chemical compound C=1N=C2NC(N)=NC(=O)C2=NC=1CNC1=CC=C(C(=O)N[C@@H](CCC(O)=O)C(O)=O)C=C1 OVBPIULPVIDEAO-LBPRGKRZSA-N 0.000 description 3
- 235000013305 food Nutrition 0.000 description 3
- 238000007710 freezing Methods 0.000 description 3
- 230000008014 freezing Effects 0.000 description 3
- SDUQYLNIPVEERB-QPPQHZFASA-N gemcitabine Chemical compound O=C1N=C(N)C=CN1[C@H]1C(F)(F)[C@H](O)[C@@H](CO)O1 SDUQYLNIPVEERB-QPPQHZFASA-N 0.000 description 3
- 239000011521 glass Substances 0.000 description 3
- 238000000227 grinding Methods 0.000 description 3
- 208000014829 head and neck neoplasm Diseases 0.000 description 3
- 230000036541 health Effects 0.000 description 3
- 208000019622 heart disease Diseases 0.000 description 3
- 238000010438 heat treatment Methods 0.000 description 3
- 239000001257 hydrogen Substances 0.000 description 3
- 229910052739 hydrogen Inorganic materials 0.000 description 3
- 238000001727 in vivo Methods 0.000 description 3
- 208000015181 infectious disease Diseases 0.000 description 3
- 229940079322 interferon Drugs 0.000 description 3
- 238000001361 intraarterial administration Methods 0.000 description 3
- 230000003834 intracellular effect Effects 0.000 description 3
- 238000007918 intramuscular administration Methods 0.000 description 3
- 238000011835 investigation Methods 0.000 description 3
- 150000002500 ions Chemical class 0.000 description 3
- RGLRXNKKBLIBQS-XNHQSDQCSA-N leuprolide acetate Chemical compound CC(O)=O.CCNC(=O)[C@@H]1CCCN1C(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](CC(C)C)NC(=O)[C@@H](NC(=O)[C@H](CO)NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@H](CC=1N=CNC=1)NC(=O)[C@H]1NC(=O)CC1)CC1=CC=C(O)C=C1 RGLRXNKKBLIBQS-XNHQSDQCSA-N 0.000 description 3
- YDTFRJLNMPSCFM-YDALLXLXSA-M levothyroxine sodium anhydrous Chemical compound [Na+].IC1=CC(C[C@H](N)C([O-])=O)=CC(I)=C1OC1=CC(I)=C(O)C(I)=C1 YDTFRJLNMPSCFM-YDALLXLXSA-M 0.000 description 3
- 239000008297 liquid dosage form Substances 0.000 description 3
- 210000004962 mammalian cell Anatomy 0.000 description 3
- VNWKTOKETHGBQD-UHFFFAOYSA-N methane Natural products C VNWKTOKETHGBQD-UHFFFAOYSA-N 0.000 description 3
- 208000010125 myocardial infarction Diseases 0.000 description 3
- 210000003739 neck Anatomy 0.000 description 3
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 3
- 239000002674 ointment Substances 0.000 description 3
- 210000001672 ovary Anatomy 0.000 description 3
- 230000003647 oxidation Effects 0.000 description 3
- 238000007254 oxidation reaction Methods 0.000 description 3
- 210000000496 pancreas Anatomy 0.000 description 3
- 206010033675 panniculitis Diseases 0.000 description 3
- 239000000312 peanut oil Substances 0.000 description 3
- 230000035515 penetration Effects 0.000 description 3
- 150000003016 phosphoric acids Chemical class 0.000 description 3
- 229920000642 polymer Polymers 0.000 description 3
- 229920001184 polypeptide Polymers 0.000 description 3
- 230000003449 preventive effect Effects 0.000 description 3
- 102000004196 processed proteins & peptides Human genes 0.000 description 3
- 238000012545 processing Methods 0.000 description 3
- 238000011321 prophylaxis Methods 0.000 description 3
- 210000002307 prostate Anatomy 0.000 description 3
- 208000005333 pulmonary edema Diseases 0.000 description 3
- BBNQQADTFFCFGB-UHFFFAOYSA-N purpurin Chemical compound C1=CC=C2C(=O)C3=C(O)C(O)=CC(O)=C3C(=O)C2=C1 BBNQQADTFFCFGB-UHFFFAOYSA-N 0.000 description 3
- 230000009467 reduction Effects 0.000 description 3
- 230000002829 reductive effect Effects 0.000 description 3
- 238000003757 reverse transcription PCR Methods 0.000 description 3
- 229920006395 saturated elastomer Polymers 0.000 description 3
- 210000001599 sigmoid colon Anatomy 0.000 description 3
- 238000001228 spectrum Methods 0.000 description 3
- 229950006050 spiromustine Drugs 0.000 description 3
- 230000000638 stimulation Effects 0.000 description 3
- 238000007920 subcutaneous administration Methods 0.000 description 3
- 239000006228 supernatant Substances 0.000 description 3
- RCINICONZNJXQF-MZXODVADSA-N taxol Chemical compound O([C@@H]1[C@@]2(C[C@@H](C(C)=C(C2(C)C)[C@H](C([C@]2(C)[C@@H](O)C[C@H]3OC[C@]3([C@H]21)OC(C)=O)=O)OC(=O)C)OC(=O)[C@H](O)[C@@H](NC(=O)C=1C=CC=CC=1)C=1C=CC=CC=1)O)C(=O)C1=CC=CC=C1 RCINICONZNJXQF-MZXODVADSA-N 0.000 description 3
- 238000002054 transplantation Methods 0.000 description 3
- 229940121358 tyrosine kinase inhibitor Drugs 0.000 description 3
- 238000003828 vacuum filtration Methods 0.000 description 3
- 206010047470 viral myocarditis Diseases 0.000 description 3
- HZSBSRAVNBUZRA-RQDPQJJXSA-J (1r,2r)-cyclohexane-1,2-diamine;tetrachloroplatinum(2+) Chemical compound Cl[Pt+2](Cl)(Cl)Cl.N[C@@H]1CCCC[C@H]1N HZSBSRAVNBUZRA-RQDPQJJXSA-J 0.000 description 2
- FELGMEQIXOGIFQ-CYBMUJFWSA-N (3r)-9-methyl-3-[(2-methylimidazol-1-yl)methyl]-2,3-dihydro-1h-carbazol-4-one Chemical compound CC1=NC=CN1C[C@@H]1C(=O)C(C=2C(=CC=CC=2)N2C)=C2CC1 FELGMEQIXOGIFQ-CYBMUJFWSA-N 0.000 description 2
- SWXOGPJRIDTIRL-DOUNNPEJSA-N (4r,7s,10s,13r,16s,19r)-10-(4-aminobutyl)-n-[(2s)-1-amino-3-(1h-indol-3-yl)-1-oxopropan-2-yl]-19-[[(2r)-2-amino-3-phenylpropanoyl]amino]-16-[(4-hydroxyphenyl)methyl]-13-(1h-indol-3-ylmethyl)-6,9,12,15,18-pentaoxo-7-propan-2-yl-1,2-dithia-5,8,11,14,17-pent Chemical compound C([C@H]1C(=O)N[C@H](CC=2C3=CC=CC=C3NC=2)C(=O)N[C@@H](CCCCN)C(=O)N[C@H](C(N[C@@H](CSSC[C@@H](C(=O)N1)NC(=O)[C@H](N)CC=1C=CC=CC=1)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(N)=O)=O)C(C)C)C1=CC=C(O)C=C1 SWXOGPJRIDTIRL-DOUNNPEJSA-N 0.000 description 2
- LKJPYSCBVHEWIU-KRWDZBQOSA-N (R)-bicalutamide Chemical compound C([C@@](O)(C)C(=O)NC=1C=C(C(C#N)=CC=1)C(F)(F)F)S(=O)(=O)C1=CC=C(F)C=C1 LKJPYSCBVHEWIU-KRWDZBQOSA-N 0.000 description 2
- OJRZEKJECRTBPJ-NGAMADIESA-N (z,5s)-5-acetamido-1-diazonio-6-hydroxy-6-oxohex-1-en-2-olate Chemical compound CC(=O)N[C@H](C(O)=O)CC\C([O-])=C\[N+]#N OJRZEKJECRTBPJ-NGAMADIESA-N 0.000 description 2
- OOMDVERDMZLRFX-UHFFFAOYSA-N 2,2-bis(aminomethyl)propane-1,3-diol;cyclobutane-1,1-dicarboxylic acid;platinum Chemical compound [Pt].NCC(CN)(CO)CO.OC(=O)C1(C(O)=O)CCC1 OOMDVERDMZLRFX-UHFFFAOYSA-N 0.000 description 2
- QXLQZLBNPTZMRK-UHFFFAOYSA-N 2-[(dimethylamino)methyl]-1-(2,4-dimethylphenyl)prop-2-en-1-one Chemical compound CN(C)CC(=C)C(=O)C1=CC=C(C)C=C1C QXLQZLBNPTZMRK-UHFFFAOYSA-N 0.000 description 2
- UZFPOOOQHWICKY-UHFFFAOYSA-N 3-[13-[1-[1-[8,12-bis(2-carboxyethyl)-17-(1-hydroxyethyl)-3,7,13,18-tetramethyl-21,24-dihydroporphyrin-2-yl]ethoxy]ethyl]-18-(2-carboxyethyl)-8-(1-hydroxyethyl)-3,7,12,17-tetramethyl-22,23-dihydroporphyrin-2-yl]propanoic acid Chemical compound N1C(C=C2C(=C(CCC(O)=O)C(C=C3C(=C(C)C(C=C4N5)=N3)CCC(O)=O)=N2)C)=C(C)C(C(C)O)=C1C=C5C(C)=C4C(C)OC(C)C1=C(N2)C=C(N3)C(C)=C(C(O)C)C3=CC(C(C)=C3CCC(O)=O)=NC3=CC(C(CCC(O)=O)=C3C)=NC3=CC2=C1C UZFPOOOQHWICKY-UHFFFAOYSA-N 0.000 description 2
- CLPFFLWZZBQMAO-UHFFFAOYSA-N 4-(5,6,7,8-tetrahydroimidazo[1,5-a]pyridin-5-yl)benzonitrile Chemical compound C1=CC(C#N)=CC=C1C1N2C=NC=C2CCC1 CLPFFLWZZBQMAO-UHFFFAOYSA-N 0.000 description 2
- XAUDJQYHKZQPEU-KVQBGUIXSA-N 5-aza-2'-deoxycytidine Chemical compound O=C1N=C(N)N=CN1[C@@H]1O[C@H](CO)[C@@H](O)C1 XAUDJQYHKZQPEU-KVQBGUIXSA-N 0.000 description 2
- KXBCLNRMQPRVTP-UHFFFAOYSA-N 6-amino-1,5-dihydroimidazo[4,5-c]pyridin-4-one Chemical compound O=C1NC(N)=CC2=C1N=CN2 KXBCLNRMQPRVTP-UHFFFAOYSA-N 0.000 description 2
- FHVDTGUDJYJELY-UHFFFAOYSA-N 6-{[2-carboxy-4,5-dihydroxy-6-(phosphanyloxy)oxan-3-yl]oxy}-4,5-dihydroxy-3-phosphanyloxane-2-carboxylic acid Chemical compound O1C(C(O)=O)C(P)C(O)C(O)C1OC1C(C(O)=O)OC(OP)C(O)C1O FHVDTGUDJYJELY-UHFFFAOYSA-N 0.000 description 2
- RTHKPHCVZVYDFN-UHFFFAOYSA-N 9-amino-5-(2-aminopyrimidin-4-yl)pyrido[3',2':4,5]pyrrolo[1,2-c]pyrimidin-4-ol Chemical compound NC1=NC=CC(C=2C3=C(O)C=CN=C3N3C(N)=NC=CC3=2)=N1 RTHKPHCVZVYDFN-UHFFFAOYSA-N 0.000 description 2
- 206010000830 Acute leukaemia Diseases 0.000 description 2
- 208000031261 Acute myeloid leukaemia Diseases 0.000 description 2
- 208000036762 Acute promyelocytic leukaemia Diseases 0.000 description 2
- 229920001817 Agar Polymers 0.000 description 2
- 208000032791 BCR-ABL1 positive chronic myelogenous leukemia Diseases 0.000 description 2
- FVLVBPDQNARYJU-XAHDHGMMSA-N C[C@H]1CCC(CC1)NC(=O)N(CCCl)N=O Chemical compound C[C@H]1CCC(CC1)NC(=O)N(CCCl)N=O FVLVBPDQNARYJU-XAHDHGMMSA-N 0.000 description 2
- 101100289995 Caenorhabditis elegans mac-1 gene Proteins 0.000 description 2
- 208000010833 Chronic myeloid leukaemia Diseases 0.000 description 2
- PTOAARAWEBMLNO-KVQBGUIXSA-N Cladribine Chemical compound C1=NC=2C(N)=NC(Cl)=NC=2N1[C@H]1C[C@H](O)[C@@H](CO)O1 PTOAARAWEBMLNO-KVQBGUIXSA-N 0.000 description 2
- 206010009944 Colon cancer Diseases 0.000 description 2
- UHDGCWIWMRVCDJ-CCXZUQQUSA-N Cytarabine Chemical compound O=C1N=C(N)C=CN1[C@H]1[C@@H](O)[C@H](O)[C@@H](CO)O1 UHDGCWIWMRVCDJ-CCXZUQQUSA-N 0.000 description 2
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 2
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 2
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 2
- 108010002156 Depsipeptides Proteins 0.000 description 2
- 206010012442 Dermatitis contact Diseases 0.000 description 2
- 241000233866 Fungi Species 0.000 description 2
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 2
- 102100039619 Granulocyte colony-stimulating factor Human genes 0.000 description 2
- 208000032843 Hemorrhage Diseases 0.000 description 2
- XDXDZDZNSLXDNA-TZNDIEGXSA-N Idarubicin Chemical compound C1[C@H](N)[C@H](O)[C@H](C)O[C@H]1O[C@@H]1C2=C(O)C(C(=O)C3=CC=CC=C3C3=O)=C3C(O)=C2C[C@@](O)(C(C)=O)C1 XDXDZDZNSLXDNA-TZNDIEGXSA-N 0.000 description 2
- 206010061217 Infestation Diseases 0.000 description 2
- 102000000589 Interleukin-1 Human genes 0.000 description 2
- 108010002352 Interleukin-1 Proteins 0.000 description 2
- 102000003777 Interleukin-1 beta Human genes 0.000 description 2
- 108090000193 Interleukin-1 beta Proteins 0.000 description 2
- 108010065637 Interleukin-23 Proteins 0.000 description 2
- 102000013264 Interleukin-23 Human genes 0.000 description 2
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 2
- ZDXPYRJPNDTMRX-VKHMYHEASA-N L-glutamine Chemical compound OC(=O)[C@@H](N)CCC(N)=O ZDXPYRJPNDTMRX-VKHMYHEASA-N 0.000 description 2
- 208000028018 Lymphocytic leukaemia Diseases 0.000 description 2
- 229930195725 Mannitol Natural products 0.000 description 2
- HRHKSTOGXBBQCB-UHFFFAOYSA-N Mitomycin E Natural products O=C1C(N)=C(C)C(=O)C2=C1C(COC(N)=O)C1(OC)C3N(C)C3CN12 HRHKSTOGXBBQCB-UHFFFAOYSA-N 0.000 description 2
- 206010062207 Mycobacterial infection Diseases 0.000 description 2
- 241000186359 Mycobacterium Species 0.000 description 2
- QJMCKEPOKRERLN-UHFFFAOYSA-N N-3,4-tridhydroxybenzamide Chemical compound ONC(=O)C1=CC=C(O)C(O)=C1 QJMCKEPOKRERLN-UHFFFAOYSA-N 0.000 description 2
- ZDZOTLJHXYCWBA-VCVYQWHSSA-N N-debenzoyl-N-(tert-butoxycarbonyl)-10-deacetyltaxol Chemical compound O([C@H]1[C@H]2[C@@](C([C@H](O)C3=C(C)[C@@H](OC(=O)[C@H](O)[C@@H](NC(=O)OC(C)(C)C)C=4C=CC=CC=4)C[C@]1(O)C3(C)C)=O)(C)[C@@H](O)C[C@H]1OC[C@]12OC(=O)C)C(=O)C1=CC=CC=C1 ZDZOTLJHXYCWBA-VCVYQWHSSA-N 0.000 description 2
- LYPFDBRUNKHDGX-SOGSVHMOSA-N N1C2=CC=C1\C(=C1\C=CC(=N1)\C(=C1\C=C/C(/N1)=C(/C1=N/C(/CC1)=C2/C1=CC(O)=CC=C1)C1=CC(O)=CC=C1)\C1=CC(O)=CC=C1)C1=CC(O)=CC=C1 Chemical compound N1C2=CC=C1\C(=C1\C=CC(=N1)\C(=C1\C=C/C(/N1)=C(/C1=N/C(/CC1)=C2/C1=CC(O)=CC=C1)C1=CC(O)=CC=C1)\C1=CC(O)=CC=C1)C1=CC(O)=CC=C1 LYPFDBRUNKHDGX-SOGSVHMOSA-N 0.000 description 2
- UFWIBTONFRDIAS-UHFFFAOYSA-N Naphthalene Chemical compound C1=CC=CC2=CC=CC=C21 UFWIBTONFRDIAS-UHFFFAOYSA-N 0.000 description 2
- 108091034117 Oligonucleotide Proteins 0.000 description 2
- 240000007594 Oryza sativa Species 0.000 description 2
- 235000007164 Oryza sativa Nutrition 0.000 description 2
- 229930012538 Paclitaxel Natural products 0.000 description 2
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 2
- 241000282320 Panthera leo Species 0.000 description 2
- 241000228143 Penicillium Species 0.000 description 2
- 108091005804 Peptidases Proteins 0.000 description 2
- GLUUGHFHXGJENI-UHFFFAOYSA-N Piperazine Chemical compound C1CNCCN1 GLUUGHFHXGJENI-UHFFFAOYSA-N 0.000 description 2
- RJKFOVLPORLFTN-LEKSSAKUSA-N Progesterone Chemical compound C1CC2=CC(=O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H](C(=O)C)[C@@]1(C)CC2 RJKFOVLPORLFTN-LEKSSAKUSA-N 0.000 description 2
- 206010060862 Prostate cancer Diseases 0.000 description 2
- 208000000236 Prostatic Neoplasms Diseases 0.000 description 2
- 102000003923 Protein Kinase C Human genes 0.000 description 2
- 108090000315 Protein Kinase C Proteins 0.000 description 2
- 238000001237 Raman spectrum Methods 0.000 description 2
- 240000004808 Saccharomyces cerevisiae Species 0.000 description 2
- 206010039361 Sacroiliitis Diseases 0.000 description 2
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 2
- DBMJMQXJHONAFJ-UHFFFAOYSA-M Sodium laurylsulphate Chemical compound [Na+].CCCCCCCCCCCCOS([O-])(=O)=O DBMJMQXJHONAFJ-UHFFFAOYSA-M 0.000 description 2
- HVUMOYIDDBPOLL-XWVZOOPGSA-N Sorbitan monostearate Chemical compound CCCCCCCCCCCCCCCCCC(=O)OC[C@@H](O)[C@H]1OC[C@H](O)[C@H]1O HVUMOYIDDBPOLL-XWVZOOPGSA-N 0.000 description 2
- 241000191967 Staphylococcus aureus Species 0.000 description 2
- NSOXQYCFHDMMGV-UHFFFAOYSA-N Tetrakis(2-hydroxypropyl)ethylenediamine Chemical class CC(O)CN(CC(C)O)CCN(CC(C)O)CC(C)O NSOXQYCFHDMMGV-UHFFFAOYSA-N 0.000 description 2
- 108010050144 Triptorelin Pamoate Proteins 0.000 description 2
- 102100040247 Tumor necrosis factor Human genes 0.000 description 2
- XSQUKJJJFZCRTK-UHFFFAOYSA-N Urea Chemical compound NC(N)=O XSQUKJJJFZCRTK-UHFFFAOYSA-N 0.000 description 2
- ZMQRJWIYMXZORG-GZIFKOAOSA-N [(1e,3r,4r,6r,7z,9z,11e)-3,6,13-trihydroxy-3-methyl-1-[(2s)-6-oxo-2,3-dihydropyran-2-yl]trideca-1,7,9,11-tetraen-4-yl] dihydrogen phosphate Chemical compound OC/C=C/C=C\C=C/[C@H](O)C[C@@H](OP(O)(O)=O)[C@@](O)(C)\C=C\[C@@H]1CC=CC(=O)O1 ZMQRJWIYMXZORG-GZIFKOAOSA-N 0.000 description 2
- USZYSDMBJDPRIF-SVEJIMAYSA-N aclacinomycin A Chemical compound O([C@H]1[C@@H](O)C[C@@H](O[C@H]1C)O[C@H]1[C@H](C[C@@H](O[C@H]1C)O[C@H]1C[C@]([C@@H](C2=CC=3C(=O)C4=CC=CC(O)=C4C(=O)C=3C(O)=C21)C(=O)OC)(O)CC)N(C)C)[C@H]1CCC(=O)[C@H](C)O1 USZYSDMBJDPRIF-SVEJIMAYSA-N 0.000 description 2
- 229960004176 aclarubicin Drugs 0.000 description 2
- SMPZPKRDRQOOHT-UHFFFAOYSA-N acronycine Chemical compound CN1C2=CC=CC=C2C(=O)C2=C1C(C=CC(C)(C)O1)=C1C=C2OC SMPZPKRDRQOOHT-UHFFFAOYSA-N 0.000 description 2
- RJURFGZVJUQBHK-UHFFFAOYSA-N actinomycin D Natural products CC1OC(=O)C(C(C)C)N(C)C(=O)CN(C)C(=O)C2CCCN2C(=O)C(C(C)C)NC(=O)C1NC(=O)C1=C(N)C(=O)C(C)=C2OC(C(C)=CC=C3C(=O)NC4C(=O)NC(C(N5CCCC5C(=O)N(C)CC(=O)N(C)C(C(C)C)C(=O)OC4C)=O)C(C)C)=C3N=C21 RJURFGZVJUQBHK-UHFFFAOYSA-N 0.000 description 2
- 239000000654 additive Substances 0.000 description 2
- 230000000996 additive effect Effects 0.000 description 2
- 230000001464 adherent effect Effects 0.000 description 2
- 239000008272 agar Substances 0.000 description 2
- 239000000556 agonist Substances 0.000 description 2
- 229940072056 alginate Drugs 0.000 description 2
- 229960000473 altretamine Drugs 0.000 description 2
- 229940024606 amino acid Drugs 0.000 description 2
- 235000001014 amino acid Nutrition 0.000 description 2
- 150000001413 amino acids Chemical class 0.000 description 2
- 229960001220 amsacrine Drugs 0.000 description 2
- XCPGHVQEEXUHNC-UHFFFAOYSA-N amsacrine Chemical compound COC1=CC(NS(C)(=O)=O)=CC=C1NC1=C(C=CC=C2)C2=NC2=CC=CC=C12 XCPGHVQEEXUHNC-UHFFFAOYSA-N 0.000 description 2
- 229960002932 anastrozole Drugs 0.000 description 2
- YBBLVLTVTVSKRW-UHFFFAOYSA-N anastrozole Chemical compound N#CC(C)(C)C1=CC(C(C)(C#N)C)=CC(CN2N=CN=C2)=C1 YBBLVLTVTVSKRW-UHFFFAOYSA-N 0.000 description 2
- 238000010171 animal model Methods 0.000 description 2
- 238000013459 approach Methods 0.000 description 2
- 150000003851 azoles Chemical class 0.000 description 2
- 230000001580 bacterial effect Effects 0.000 description 2
- 239000011324 bead Substances 0.000 description 2
- 229960000997 bicalutamide Drugs 0.000 description 2
- 230000000975 bioactive effect Effects 0.000 description 2
- 229960000074 biopharmaceutical Drugs 0.000 description 2
- 229950008548 bisantrene Drugs 0.000 description 2
- 210000003995 blood forming stem cell Anatomy 0.000 description 2
- 150000007516 brønsted-lowry acids Chemical class 0.000 description 2
- 150000007528 brønsted-lowry bases Chemical class 0.000 description 2
- 230000015556 catabolic process Effects 0.000 description 2
- 210000002421 cell wall Anatomy 0.000 description 2
- 235000010980 cellulose Nutrition 0.000 description 2
- 229920002678 cellulose Polymers 0.000 description 2
- 208000037976 chronic inflammation Diseases 0.000 description 2
- 238000000576 coating method Methods 0.000 description 2
- 208000010247 contact dermatitis Diseases 0.000 description 2
- 230000002596 correlated effect Effects 0.000 description 2
- SBRXTSOCZITGQG-UHFFFAOYSA-N crisnatol Chemical compound C1=CC=C2C(CNC(CO)(CO)C)=CC3=C(C=CC=C4)C4=CC=C3C2=C1 SBRXTSOCZITGQG-UHFFFAOYSA-N 0.000 description 2
- 229950007258 crisnatol Drugs 0.000 description 2
- 238000002447 crystallographic data Methods 0.000 description 2
- 239000000850 decongestant Substances 0.000 description 2
- 238000006731 degradation reaction Methods 0.000 description 2
- 230000018044 dehydration Effects 0.000 description 2
- 238000006297 dehydration reaction Methods 0.000 description 2
- 230000006866 deterioration Effects 0.000 description 2
- 229950010621 dezaguanine Drugs 0.000 description 2
- WVYXNIXAMZOZFK-UHFFFAOYSA-N diaziquone Chemical compound O=C1C(NC(=O)OCC)=C(N2CC2)C(=O)C(NC(=O)OCC)=C1N1CC1 WVYXNIXAMZOZFK-UHFFFAOYSA-N 0.000 description 2
- 235000005911 diet Nutrition 0.000 description 2
- 230000037213 diet Effects 0.000 description 2
- 238000002050 diffraction method Methods 0.000 description 2
- 239000003085 diluting agent Substances 0.000 description 2
- 238000010790 dilution Methods 0.000 description 2
- 239000012895 dilution Substances 0.000 description 2
- LOKCTEFSRHRXRJ-UHFFFAOYSA-I dipotassium trisodium dihydrogen phosphate hydrogen phosphate dichloride Chemical compound P(=O)(O)(O)[O-].[K+].P(=O)(O)([O-])[O-].[Na+].[Na+].[Cl-].[K+].[Cl-].[Na+] LOKCTEFSRHRXRJ-UHFFFAOYSA-I 0.000 description 2
- 238000007599 discharging Methods 0.000 description 2
- 238000004090 dissolution Methods 0.000 description 2
- 229960003668 docetaxel Drugs 0.000 description 2
- 229950004203 droloxifene Drugs 0.000 description 2
- 229950005133 duazomycin Drugs 0.000 description 2
- 229930192837 duazomycin Natural products 0.000 description 2
- 238000004043 dyeing Methods 0.000 description 2
- 230000002526 effect on cardiovascular system Effects 0.000 description 2
- VLCYCQAOQCDTCN-UHFFFAOYSA-N eflornithine Chemical compound NCCCC(N)(C(F)F)C(O)=O VLCYCQAOQCDTCN-UHFFFAOYSA-N 0.000 description 2
- 238000002565 electrocardiography Methods 0.000 description 2
- 210000003038 endothelium Anatomy 0.000 description 2
- 206010015037 epilepsy Diseases 0.000 description 2
- HCZKYJDFEPMADG-UHFFFAOYSA-N erythro-nordihydroguaiaretic acid Natural products C=1C=C(O)C(O)=CC=1CC(C)C(C)CC1=CC=C(O)C(O)=C1 HCZKYJDFEPMADG-UHFFFAOYSA-N 0.000 description 2
- FRPJXPJMRWBBIH-RBRWEJTLSA-N estramustine Chemical compound ClCCN(CCCl)C(=O)OC1=CC=C2[C@H]3CC[C@](C)([C@H](CC4)O)[C@@H]4[C@@H]3CCC2=C1 FRPJXPJMRWBBIH-RBRWEJTLSA-N 0.000 description 2
- 239000000328 estrogen antagonist Substances 0.000 description 2
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 2
- 239000000284 extract Substances 0.000 description 2
- 229950011548 fadrozole Drugs 0.000 description 2
- 230000002349 favourable effect Effects 0.000 description 2
- NMUSYJAQQFHJEW-ARQDHWQXSA-N fazarabine Chemical compound O=C1N=C(N)N=CN1[C@H]1[C@@H](O)[C@H](O)[C@@H](CO)O1 NMUSYJAQQFHJEW-ARQDHWQXSA-N 0.000 description 2
- 238000011832 ferret model Methods 0.000 description 2
- 239000012997 ficoll-paque Substances 0.000 description 2
- 238000000684 flow cytometry Methods 0.000 description 2
- GIUYCYHIANZCFB-FJFJXFQQSA-N fludarabine phosphate Chemical compound C1=NC=2C(N)=NC(F)=NC=2N1[C@@H]1O[C@H](COP(O)(O)=O)[C@@H](O)[C@@H]1O GIUYCYHIANZCFB-FJFJXFQQSA-N 0.000 description 2
- GVEPBJHOBDJJJI-UHFFFAOYSA-N fluoranthene Chemical compound C1=CC(C2=CC=CC=C22)=C3C2=CC=CC3=C1 GVEPBJHOBDJJJI-UHFFFAOYSA-N 0.000 description 2
- 235000011194 food seasoning agent Nutrition 0.000 description 2
- 238000004108 freeze drying Methods 0.000 description 2
- CHPZKNULDCNCBW-UHFFFAOYSA-N gallium nitrate Chemical compound [Ga+3].[O-][N+]([O-])=O.[O-][N+]([O-])=O.[O-][N+]([O-])=O CHPZKNULDCNCBW-UHFFFAOYSA-N 0.000 description 2
- 239000007789 gas Substances 0.000 description 2
- 229960005277 gemcitabine Drugs 0.000 description 2
- 239000003292 glue Substances 0.000 description 2
- UUVWYPNAQBNQJQ-UHFFFAOYSA-N hexamethylmelamine Chemical compound CN(C)C1=NC(N(C)C)=NC(N(C)C)=N1 UUVWYPNAQBNQJQ-UHFFFAOYSA-N 0.000 description 2
- 229960001101 ifosfamide Drugs 0.000 description 2
- HOMGKSMUEGBAAB-UHFFFAOYSA-N ifosfamide Chemical compound ClCCNP1(=O)OCCCN1CCCl HOMGKSMUEGBAAB-UHFFFAOYSA-N 0.000 description 2
- 230000003308 immunostimulating effect Effects 0.000 description 2
- 239000012535 impurity Substances 0.000 description 2
- CGIGDMFJXJATDK-UHFFFAOYSA-N indomethacin Chemical compound CC1=C(CC(O)=O)C2=CC(OC)=CC=C2N1C(=O)C1=CC=C(Cl)C=C1 CGIGDMFJXJATDK-UHFFFAOYSA-N 0.000 description 2
- 230000028709 inflammatory response Effects 0.000 description 2
- 238000002329 infrared spectrum Methods 0.000 description 2
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N lactic acid Chemical class CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 description 2
- 108010021336 lanreotide Proteins 0.000 description 2
- 235000012204 lemonade/lime carbonate Nutrition 0.000 description 2
- 229960003881 letrozole Drugs 0.000 description 2
- HPJKCIUCZWXJDR-UHFFFAOYSA-N letrozole Chemical compound C1=CC(C#N)=CC=C1C(N1N=CN=C1)C1=CC=C(C#N)C=C1 HPJKCIUCZWXJDR-UHFFFAOYSA-N 0.000 description 2
- 229960004338 leuprorelin Drugs 0.000 description 2
- 239000006210 lotion Substances 0.000 description 2
- 239000007937 lozenge Substances 0.000 description 2
- 208000012804 lymphangiosarcoma Diseases 0.000 description 2
- 201000004792 malaria Diseases 0.000 description 2
- 239000000594 mannitol Substances 0.000 description 2
- 235000010355 mannitol Nutrition 0.000 description 2
- 238000004519 manufacturing process Methods 0.000 description 2
- 229960003951 masoprocol Drugs 0.000 description 2
- HCZKYJDFEPMADG-TXEJJXNPSA-N masoprocol Chemical compound C([C@H](C)[C@H](C)CC=1C=C(O)C(O)=CC=1)C1=CC=C(O)C(O)=C1 HCZKYJDFEPMADG-TXEJJXNPSA-N 0.000 description 2
- HAWPXGHAZFHHAD-UHFFFAOYSA-N mechlorethamine Chemical compound ClCCN(C)CCCl HAWPXGHAZFHHAD-UHFFFAOYSA-N 0.000 description 2
- 208000023356 medullary thyroid gland carcinoma Diseases 0.000 description 2
- 238000002844 melting Methods 0.000 description 2
- 230000008018 melting Effects 0.000 description 2
- LWYJUZBXGAFFLP-OCNCTQISSA-N menogaril Chemical compound O1[C@@]2(C)[C@H](O)[C@@H](N(C)C)[C@H](O)[C@@H]1OC1=C3C(=O)C(C=C4C[C@@](C)(O)C[C@H](C4=C4O)OC)=C4C(=O)C3=C(O)C=C12 LWYJUZBXGAFFLP-OCNCTQISSA-N 0.000 description 2
- 229950002676 menogaril Drugs 0.000 description 2
- ODDHBYXHXZCAGQ-UHFFFAOYSA-N methyl 2-anilinobenzoate Chemical class COC(=O)C1=CC=CC=C1NC1=CC=CC=C1 ODDHBYXHXZCAGQ-UHFFFAOYSA-N 0.000 description 2
- HRHKSTOGXBBQCB-VFWICMBZSA-N methylmitomycin Chemical compound O=C1C(N)=C(C)C(=O)C2=C1[C@@H](COC(N)=O)[C@@]1(OC)[C@H]3N(C)[C@H]3CN12 HRHKSTOGXBBQCB-VFWICMBZSA-N 0.000 description 2
- 230000000813 microbial effect Effects 0.000 description 2
- 238000002156 mixing Methods 0.000 description 2
- 239000007932 molded tablet Substances 0.000 description 2
- 210000001616 monocyte Anatomy 0.000 description 2
- 230000035772 mutation Effects 0.000 description 2
- 208000027531 mycobacterial infectious disease Diseases 0.000 description 2
- 208000025113 myeloid leukemia Diseases 0.000 description 2
- NJSMWLQOCQIOPE-OCHFTUDZSA-N n-[(e)-[10-[(e)-(4,5-dihydro-1h-imidazol-2-ylhydrazinylidene)methyl]anthracen-9-yl]methylideneamino]-4,5-dihydro-1h-imidazol-2-amine Chemical compound N1CCN=C1N\N=C\C(C1=CC=CC=C11)=C(C=CC=C2)C2=C1\C=N\NC1=NCCN1 NJSMWLQOCQIOPE-OCHFTUDZSA-N 0.000 description 2
- 229940021182 non-steroidal anti-inflammatory drug Drugs 0.000 description 2
- 231100000252 nontoxic Toxicity 0.000 description 2
- 230000003000 nontoxic effect Effects 0.000 description 2
- 210000001331 nose Anatomy 0.000 description 2
- 229960005343 ondansetron Drugs 0.000 description 2
- 229950008017 ormaplatin Drugs 0.000 description 2
- 229960001592 paclitaxel Drugs 0.000 description 2
- 201000010198 papillary carcinoma Diseases 0.000 description 2
- 208000035824 paresthesia Diseases 0.000 description 2
- 229960001744 pegaspargase Drugs 0.000 description 2
- 108010001564 pegaspargase Proteins 0.000 description 2
- VPAWVRUHMJVRHU-VGDKGRGNSA-N perfosfamide Chemical compound OO[C@@H]1CCO[P@@](=O)(N(CCCl)CCCl)N1 VPAWVRUHMJVRHU-VGDKGRGNSA-N 0.000 description 2
- 229950009351 perfosfamide Drugs 0.000 description 2
- 230000010412 perfusion Effects 0.000 description 2
- 229920001983 poloxamer Polymers 0.000 description 2
- 235000010482 polyoxyethylene sorbitan monooleate Nutrition 0.000 description 2
- 229920000053 polysorbate 80 Polymers 0.000 description 2
- 229960004293 porfimer sodium Drugs 0.000 description 2
- 229950004406 porfiromycin Drugs 0.000 description 2
- 239000002243 precursor Substances 0.000 description 2
- 230000035935 pregnancy Effects 0.000 description 2
- 230000000770 proinflammatory effect Effects 0.000 description 2
- 201000003651 pulmonary sarcoidosis Diseases 0.000 description 2
- RXWNCPJZOCPEPQ-NVWDDTSBSA-N puromycin Chemical compound C1=CC(OC)=CC=C1C[C@H](N)C(=O)N[C@H]1[C@@H](O)[C@H](N2C3=NC=NC(=C3N=C2)N(C)C)O[C@@H]1CO RXWNCPJZOCPEPQ-NVWDDTSBSA-N 0.000 description 2
- XSCHRSMBECNVNS-UHFFFAOYSA-N quinoxaline Chemical compound N1=CC=NC2=CC=CC=C21 XSCHRSMBECNVNS-UHFFFAOYSA-N 0.000 description 2
- 230000002285 radioactive effect Effects 0.000 description 2
- 230000008521 reorganization Effects 0.000 description 2
- 230000004044 response Effects 0.000 description 2
- 235000009566 rice Nutrition 0.000 description 2
- QXKJWHWUDVQATH-UHFFFAOYSA-N rogletimide Chemical compound C=1C=NC=CC=1C1(CC)CCC(=O)NC1=O QXKJWHWUDVQATH-UHFFFAOYSA-N 0.000 description 2
- 229950005230 rogletimide Drugs 0.000 description 2
- MOCVYVBNJQIVOV-TVQRCGJNSA-N rohitukine Chemical compound O[C@@H]1CN(C)CC[C@@H]1C1=C(O)C=C(O)C2=C1OC(C)=CC2=O MOCVYVBNJQIVOV-TVQRCGJNSA-N 0.000 description 2
- CGFVUVWMYIHGHS-UHFFFAOYSA-N saintopin Chemical compound C1=C(O)C=C2C=C(C(=O)C=3C(=C(O)C=C(C=3)O)C3=O)C3=C(O)C2=C1O CGFVUVWMYIHGHS-UHFFFAOYSA-N 0.000 description 2
- 239000012047 saturated solution Substances 0.000 description 2
- 229960003440 semustine Drugs 0.000 description 2
- 238000000926 separation method Methods 0.000 description 2
- 230000019491 signal transduction Effects 0.000 description 2
- 239000000377 silicon dioxide Substances 0.000 description 2
- 235000019333 sodium laurylsulphate Nutrition 0.000 description 2
- 229910052938 sodium sulfate Inorganic materials 0.000 description 2
- 235000011152 sodium sulphate Nutrition 0.000 description 2
- 238000000935 solvent evaporation Methods 0.000 description 2
- 239000000600 sorbitol Substances 0.000 description 2
- 235000010356 sorbitol Nutrition 0.000 description 2
- 229940063675 spermine Drugs 0.000 description 2
- 238000003756 stirring Methods 0.000 description 2
- PVYJZLYGTZKPJE-UHFFFAOYSA-N streptonigrin Chemical compound C=1C=C2C(=O)C(OC)=C(N)C(=O)C2=NC=1C(C=1N)=NC(C(O)=O)=C(C)C=1C1=CC=C(OC)C(OC)=C1O PVYJZLYGTZKPJE-UHFFFAOYSA-N 0.000 description 2
- 239000000758 substrate Substances 0.000 description 2
- 235000000346 sugar Nutrition 0.000 description 2
- 235000012222 talc Nutrition 0.000 description 2
- 239000000454 talc Substances 0.000 description 2
- 229910052623 talc Inorganic materials 0.000 description 2
- URLYINUFLXOMHP-HTVVRFAVSA-N tcn-p Chemical compound C=12C3=NC=NC=1N(C)N=C(N)C2=CN3[C@@H]1O[C@H](COP(O)(O)=O)[C@@H](O)[C@H]1O URLYINUFLXOMHP-HTVVRFAVSA-N 0.000 description 2
- 229960001674 tegafur Drugs 0.000 description 2
- WFWLQNSHRPWKFK-ZCFIWIBFSA-N tegafur Chemical compound O=C1NC(=O)C(F)=CN1[C@@H]1OCCC1 WFWLQNSHRPWKFK-ZCFIWIBFSA-N 0.000 description 2
- 229960002197 temoporfin Drugs 0.000 description 2
- NRUKOCRGYNPUPR-QBPJDGROSA-N teniposide Chemical compound COC1=C(O)C(OC)=CC([C@@H]2C3=CC=4OCOC=4C=C3[C@@H](O[C@H]3[C@@H]([C@@H](O)[C@@H]4O[C@@H](OC[C@H]4O3)C=3SC=CC=3)O)[C@@H]3[C@@H]2C(OC3)=O)=C1 NRUKOCRGYNPUPR-QBPJDGROSA-N 0.000 description 2
- 229960001278 teniposide Drugs 0.000 description 2
- 238000011287 therapeutic dose Methods 0.000 description 2
- WYWHKKSPHMUBEB-UHFFFAOYSA-N tioguanine Chemical compound N1C(N)=NC(=S)C2=C1N=CN2 WYWHKKSPHMUBEB-UHFFFAOYSA-N 0.000 description 2
- ORYDPOVDJJZGHQ-UHFFFAOYSA-N tirapazamine Chemical compound C1=CC=CC2=[N+]([O-])C(N)=N[N+]([O-])=C21 ORYDPOVDJJZGHQ-UHFFFAOYSA-N 0.000 description 2
- TVPNFKRGOFJQOO-UHFFFAOYSA-N topsentin b1 Chemical compound C1=CC=C2C(C3=CN=C(N3)C(=O)C=3C4=CC=C(C=C4NC=3)O)=CNC2=C1 TVPNFKRGOFJQOO-UHFFFAOYSA-N 0.000 description 2
- XFCLJVABOIYOMF-QPLCGJKRSA-N toremifene Chemical compound C1=CC(OCCN(C)C)=CC=C1C(\C=1C=CC=CC=1)=C(\CCCl)C1=CC=CC=C1 XFCLJVABOIYOMF-QPLCGJKRSA-N 0.000 description 2
- 229960005026 toremifene Drugs 0.000 description 2
- 231100000419 toxicity Toxicity 0.000 description 2
- 230000001988 toxicity Effects 0.000 description 2
- LZFOPEXOUVTGJS-ONEGZZNKSA-N trans-sinapyl alcohol Chemical compound COC1=CC(\C=C\CO)=CC(OC)=C1O LZFOPEXOUVTGJS-ONEGZZNKSA-N 0.000 description 2
- 229950003873 triciribine Drugs 0.000 description 2
- NOYPYLRCIDNJJB-UHFFFAOYSA-N trimetrexate Chemical compound COC1=C(OC)C(OC)=CC(NCC=2C(=C3C(N)=NC(N)=NC3=CC=2)C)=C1 NOYPYLRCIDNJJB-UHFFFAOYSA-N 0.000 description 2
- 229960001099 trimetrexate Drugs 0.000 description 2
- VXKHXGOKWPXYNA-PGBVPBMZSA-N triptorelin Chemical compound C([C@@H](C(=O)N[C@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N1[C@@H](CCC1)C(=O)NCC(N)=O)NC(=O)[C@H](CO)NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@H](CC=1N=CNC=1)NC(=O)[C@H]1NC(=O)CC1)C1=CC=C(O)C=C1 VXKHXGOKWPXYNA-PGBVPBMZSA-N 0.000 description 2
- 229960004824 triptorelin Drugs 0.000 description 2
- 210000001215 vagina Anatomy 0.000 description 2
- 229960002730 vapreotide Drugs 0.000 description 2
- 108700029852 vapreotide Proteins 0.000 description 2
- YTZALCGQUPRCGW-ZSFNYQMMSA-N verteporfin Chemical compound N1C(C=C2C(=C(CCC(O)=O)C(C=C3C(CCC(=O)OC)=C(C)C(N3)=C3)=N2)C)=C(C=C)C(C)=C1C=C1C2=CC=C(C(=O)OC)[C@@H](C(=O)OC)[C@@]2(C)C3=N1 YTZALCGQUPRCGW-ZSFNYQMMSA-N 0.000 description 2
- 238000001845 vibrational spectrum Methods 0.000 description 2
- 229940061392 visudyne Drugs 0.000 description 2
- 229960001771 vorozole Drugs 0.000 description 2
- XLMPPFTZALNBFS-INIZCTEOSA-N vorozole Chemical compound C1([C@@H](C2=CC=C3N=NN(C3=C2)C)N2N=CN=C2)=CC=C(Cl)C=C1 XLMPPFTZALNBFS-INIZCTEOSA-N 0.000 description 2
- 239000008215 water for injection Substances 0.000 description 2
- 238000009736 wetting Methods 0.000 description 2
- 239000000080 wetting agent Substances 0.000 description 2
- 229950003017 zeniplatin Drugs 0.000 description 2
- QCHFTSOMWOSFHM-WPRPVWTQSA-N (+)-Pilocarpine Chemical compound C1OC(=O)[C@@H](CC)[C@H]1CC1=CN=CN1C QCHFTSOMWOSFHM-WPRPVWTQSA-N 0.000 description 1
- BMKDZUISNHGIBY-ZETCQYMHSA-N (+)-dexrazoxane Chemical compound C([C@H](C)N1CC(=O)NC(=O)C1)N1CC(=O)NC(=O)C1 BMKDZUISNHGIBY-ZETCQYMHSA-N 0.000 description 1
- GCPUVEMWOWMALU-HZMBPMFUSA-N (1s,3s)-1-hydroxy-8-methoxy-3-methyl-1,2,3,4-tetrahydrobenzo[a]anthracene-7,12-dione Chemical compound C1[C@H](C)C[C@H](O)C2=C1C=CC1=C2C(=O)C(C=CC=C2OC)=C2C1=O GCPUVEMWOWMALU-HZMBPMFUSA-N 0.000 description 1
- MNHVIVWFCMBFCV-AVGNSLFASA-N (2S)-2-[[(2S)-2-[[(4S)-4-amino-4-carboxybutanoyl]amino]-6-diazo-5-oxohexanoyl]amino]-6-diazo-5-oxohexanoic acid Chemical compound OC(=O)[C@@H](N)CCC(=O)N[C@@H](CCC(=O)C=[N+]=[N-])C(=O)N[C@@H](CCC(=O)C=[N+]=[N-])C(O)=O MNHVIVWFCMBFCV-AVGNSLFASA-N 0.000 description 1
- MXABZXILAJGOTL-AUYMZICSSA-N (2S)-N-[(2S)-1-[(2S)-1-[(2S,3S)-1-[(2S)-1-[2-[(2S)-1,3-dihydroxy-1-[(E)-1-hydroxy-1-[(2S,3S)-1-hydroxy-3-methyl-1-[[(2Z,6S,9S,12R)-5,8,11-trihydroxy-9-(2-methylpropyl)-6-propan-2-yl-1-thia-4,7,10-triazacyclotrideca-2,4,7,10-tetraen-12-yl]imino]pentan-2-yl]iminobut-2-en-2-yl]iminopropan-2-yl]imino-2-hydroxyethyl]imino-1,5-dihydroxy-5-iminopentan-2-yl]imino-1-hydroxy-3-methylpentan-2-yl]imino-1-hydroxy-3-methylbutan-2-yl]imino-1-hydroxy-3-phenylpropan-2-yl]-2-[[(2S)-2-[[(2S)-2-[[(Z)-2-[[(2S)-2-[[(Z)-2-[[(2S)-2-[[[(2S)-1-[(Z)-2-[[(2S)-2-(dimethylamino)-1-hydroxypropylidene]amino]but-2-enoyl]pyrrolidin-2-yl]-hydroxymethylidene]amino]-1-hydroxypropylidene]amino]-1-hydroxybut-2-enylidene]amino]-1-hydroxy-3-phenylpropylidene]amino]-1-hydroxybut-2-enylidene]amino]-1-hydroxy-3-methylbutylidene]amino]-1-hydroxypropylidene]amino]pentanediimidic acid Chemical compound CC[C@H](C)[C@H](\N=C(/O)[C@@H](\N=C(/O)[C@H](Cc1ccccc1)\N=C(/O)[C@H](CCC(O)=N)\N=C(/O)[C@H](C)\N=C(/O)[C@@H](\N=C(/O)\C(=C\C)\N=C(/O)[C@H](Cc1ccccc1)\N=C(/O)\C(=C\C)\N=C(/O)[C@H](C)\N=C(/O)[C@@H]1CCCN1C(=O)\C(=C\C)\N=C(/O)[C@H](C)N(C)C)C(C)C)C(C)C)C(\O)=N\[C@@H](CCC(O)=N)C(\O)=N\C\C(O)=N\[C@@H](CO)C(\O)=N\C(=C\C)\C(\O)=N\[C@@H]([C@@H](C)CC)C(\O)=N\[C@H]1CS\C=C/N=C(O)\[C@@H](\N=C(O)/[C@H](CC(C)C)\N=C1\O)C(C)C MXABZXILAJGOTL-AUYMZICSSA-N 0.000 description 1
- BUSGWUFLNHIBPT-XYBORKQMSA-N (2e,4e,6e)-7-[(1r,5r,6s)-3-[[(2e,4e)-5-cyclohexylpenta-2,4-dienoyl]amino]-5-hydroxy-2-oxo-7-oxabicyclo[4.1.0]hept-3-en-5-yl]hepta-2,4,6-trienoic acid Chemical compound C([C@]([C@H]1O[C@H]1C1=O)(O)/C=C/C=C/C=C/C(=O)O)=C1NC(=O)\C=C\C=C\C1CCCCC1 BUSGWUFLNHIBPT-XYBORKQMSA-N 0.000 description 1
- LCADVYTXPLBAGB-AUQKUMLUSA-N (2e,4e,6z,8e,10e,14e)-13-hydroxy-n-(1-hydroxypropan-2-yl)-2,10,12,14,16-pentamethyl-18-phenyloctadeca-2,4,6,8,10,14-hexaenamide Chemical compound OCC(C)NC(=O)C(\C)=C\C=C\C=C/C=C/C(/C)=C/C(C)C(O)C(\C)=C\C(C)CCC1=CC=CC=C1 LCADVYTXPLBAGB-AUQKUMLUSA-N 0.000 description 1
- FKHUGQZRBPETJR-RXSRXONKSA-N (2r)-2-[[(4r)-4-[[(2s)-2-[[(2r)-2-[(3r,4r,5s,6r)-3-acetamido-2,5-dihydroxy-6-(hydroxymethyl)oxan-4-yl]oxypropanoyl]amino]propanoyl]amino]-5-amino-5-oxopentanoyl]amino]-6-(octadecanoylamino)hexanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(=O)NCCCC[C@H](C(O)=O)NC(=O)CC[C@H](C(N)=O)NC(=O)[C@H](C)NC(=O)[C@@H](C)O[C@H]1[C@H](O)[C@@H](CO)OC(O)[C@@H]1NC(C)=O FKHUGQZRBPETJR-RXSRXONKSA-N 0.000 description 1
- SWTGJCNCBUCXSS-ISUZDFFFSA-N (2r)-3,4-dihydroxy-2-[(4s)-2-phenyl-1,3-dioxolan-4-yl]-2h-furan-5-one Chemical compound OC1=C(O)C(=O)O[C@@H]1[C@H]1OC(C=2C=CC=CC=2)OC1 SWTGJCNCBUCXSS-ISUZDFFFSA-N 0.000 description 1
- RCGXNDQKCXNWLO-WLEIXIPESA-N (2r)-n-[(2s)-5-amino-1-[[(2r,3r)-1-[[(3s,6z,9s,12r,15r,18r,19s)-9-benzyl-15-[(2r)-butan-2-yl]-6-ethylidene-19-methyl-2,5,8,11,14,17-hexaoxo-3,12-di(propan-2-yl)-1-oxa-4,7,10,13,16-pentazacyclononadec-18-yl]amino]-3-methyl-1-oxopentan-2-yl]amino]-1-oxopent Chemical compound N([C@@H](CCCN)C(=O)N[C@H]([C@H](C)CC)C(=O)N[C@H]1C(N[C@@H](C(=O)N[C@@H](C(=O)N[C@@H](CC=2C=CC=CC=2)C(=O)NC(/C(=O)N[C@H](C(=O)O[C@H]1C)C(C)C)=C\C)C(C)C)[C@H](C)CC)=O)C(=O)[C@H]1CCCN1C(=O)[C@H](NC(=O)[C@@H](NC(=O)[C@@H](NC(=O)[C@H](NC(=O)CCCC(C)C)C(C)C)[C@@H](C)O)C(C)C)C(C)C RCGXNDQKCXNWLO-WLEIXIPESA-N 0.000 description 1
- PAYBYKKERMGTSS-MNCSTQPFSA-N (2r,3r,3as,9ar)-7-fluoro-2-(hydroxymethyl)-6-imino-2,3,3a,9a-tetrahydrofuro[1,2][1,3]oxazolo[3,4-a]pyrimidin-3-ol Chemical compound N=C1C(F)=CN2[C@@H]3O[C@H](CO)[C@@H](O)[C@@H]3OC2=N1 PAYBYKKERMGTSS-MNCSTQPFSA-N 0.000 description 1
- WDQLRUYAYXDIFW-RWKIJVEZSA-N (2r,3r,4s,5r,6r)-4-[(2s,3r,4s,5r,6r)-3,5-dihydroxy-4-[(2r,3r,4s,5s,6r)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy-6-[[(2r,3r,4s,5s,6r)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxymethyl]oxan-2-yl]oxy-6-(hydroxymethyl)oxane-2,3,5-triol Chemical compound O[C@@H]1[C@@H](CO)O[C@@H](O)[C@H](O)[C@H]1O[C@H]1[C@H](O)[C@@H](O[C@H]2[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O2)O)[C@H](O)[C@@H](CO[C@H]2[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O2)O)O1 WDQLRUYAYXDIFW-RWKIJVEZSA-N 0.000 description 1
- NOENHWMKHNSHGX-IZOOSHNJSA-N (2s)-1-[(2s)-2-[[(2s)-2-[[(2r)-2-[[(2r)-2-[[(2s)-2-[[(2r)-2-[[(2s)-2-[[(2r)-2-acetamido-3-naphthalen-2-ylpropanoyl]amino]-3-(4-chlorophenyl)propanoyl]amino]-3-pyridin-3-ylpropanoyl]amino]-3-hydroxypropanoyl]amino]-3-(4-hydroxyphenyl)propanoyl]amino]-6-(ca Chemical compound C([C@H](C(=O)N[C@H](CCCCNC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCNC(C)C)C(=O)N1[C@@H](CCC1)C(=O)N[C@H](C)C(N)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](CC=1C=NC=CC=1)NC(=O)[C@H](CC=1C=CC(Cl)=CC=1)NC(=O)[C@@H](CC=1C=C2C=CC=CC2=CC=1)NC(C)=O)C1=CC=C(O)C=C1 NOENHWMKHNSHGX-IZOOSHNJSA-N 0.000 description 1
- RDJGLLICXDHJDY-NSHDSACASA-N (2s)-2-(3-phenoxyphenyl)propanoic acid Chemical compound OC(=O)[C@@H](C)C1=CC=CC(OC=2C=CC=CC=2)=C1 RDJGLLICXDHJDY-NSHDSACASA-N 0.000 description 1
- ZZKNRXZVGOYGJT-VKHMYHEASA-N (2s)-2-[(2-phosphonoacetyl)amino]butanedioic acid Chemical compound OC(=O)C[C@@H](C(O)=O)NC(=O)CP(O)(O)=O ZZKNRXZVGOYGJT-VKHMYHEASA-N 0.000 description 1
- CUCSSYAUKKIDJV-FAXBSAIASA-N (2s)-2-[[(2r)-2-[[(2s)-2-[[(2r)-2-[[(2s)-2-amino-5-(diaminomethylideneamino)pentanoyl]amino]-3-(1h-indol-3-yl)propanoyl]-methylamino]-3-phenylpropanoyl]amino]-3-(1h-indol-3-yl)propanoyl]amino]-n-[(2s)-1-amino-4-methylsulfanyl-1-oxobutan-2-yl]-4-methylpent Chemical compound C([C@@H](C(=O)N[C@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCSC)C(N)=O)N(C)C(=O)[C@@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@@H](N)CCCN=C(N)N)C1=CC=CC=C1 CUCSSYAUKKIDJV-FAXBSAIASA-N 0.000 description 1
- ZUQBAQVRAURMCL-DOMZBBRYSA-N (2s)-2-[[4-[2-[(6r)-2-amino-4-oxo-5,6,7,8-tetrahydro-1h-pyrido[2,3-d]pyrimidin-6-yl]ethyl]benzoyl]amino]pentanedioic acid Chemical compound C([C@@H]1CC=2C(=O)N=C(NC=2NC1)N)CC1=CC=C(C(=O)N[C@@H](CCC(O)=O)C(O)=O)C=C1 ZUQBAQVRAURMCL-DOMZBBRYSA-N 0.000 description 1
- JRBXPUUAYKCCLQ-QMMMGPOBSA-N (2s)-2-amino-2-[3-hydroxy-4-(hydroxymethyl)phenyl]acetic acid Chemical compound OC(=O)[C@@H](N)C1=CC=C(CO)C(O)=C1 JRBXPUUAYKCCLQ-QMMMGPOBSA-N 0.000 description 1
- DWNBOPVKNPVNQG-LURJTMIESA-N (2s)-4-hydroxy-2-(propylamino)butanoic acid Chemical compound CCCN[C@H](C(O)=O)CCO DWNBOPVKNPVNQG-LURJTMIESA-N 0.000 description 1
- KYHUYMLIVQFXRI-XYUQHQMCSA-N (2s)-n-[(2r)-1-[[(3s,6s,8s,12s,13r,16s,17r,20s,23s)-13-[(2s)-butan-2-yl]-12-hydroxy-20-[(4-methoxyphenyl)methyl]-6,17,21-trimethyl-3-(2-methylpropyl)-2,5,7,10,15,19,22-heptaoxo-8-propan-2-yl-9,18-dioxa-1,4,14,21-tetrazabicyclo[21.3.0]hexacosan-16-yl]amino Chemical compound CN([C@H](CC(C)C)C(=O)N[C@@H]1C(=O)N[C@@H]([C@H](CC(=O)O[C@H](C(=O)[C@H](C)C(=O)N[C@@H](CC(C)C)C(=O)N2CCC[C@H]2C(=O)N(C)[C@@H](CC=2C=CC(OC)=CC=2)C(=O)O[C@@H]1C)C(C)C)O)[C@@H](C)CC)C(=O)[C@@H]1CCCN1C(=O)C(C)O KYHUYMLIVQFXRI-XYUQHQMCSA-N 0.000 description 1
- HJNZCKLMRAOTMA-BRBGIFQRSA-N (2s)-n-[(2s)-1-[[(2s)-1-[[(2s)-1-[[(2s)-1-[[(2r)-1-[[(2s)-1-[[(2s)-5-(diaminomethylideneamino)-1-[(2s)-2-(ethylcarbamoyl)pyrrolidin-1-yl]-1-oxopentan-2-yl]amino]-4-methyl-1-oxopentan-2-yl]amino]-3-(2-methyl-1h-indol-3-yl)-1-oxopropan-2-yl]amino]-3-(4-hydr Chemical compound CCNC(=O)[C@@H]1CCCN1C(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@H](CC=1N=CNC=1)NC(=O)[C@H]1NC(=O)CC1)CC1=C(C)NC2=CC=CC=C12 HJNZCKLMRAOTMA-BRBGIFQRSA-N 0.000 description 1
- NAALWFYYHHJEFQ-ZASNTINBSA-N (2s,5r,6r)-6-[[(2r)-2-[[6-[4-[bis(2-hydroxyethyl)sulfamoyl]phenyl]-2-oxo-1h-pyridine-3-carbonyl]amino]-2-(4-hydroxyphenyl)acetyl]amino]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid Chemical compound N([C@@H](C(=O)N[C@H]1[C@H]2SC([C@@H](N2C1=O)C(O)=O)(C)C)C=1C=CC(O)=CC=1)C(=O)C(C(N1)=O)=CC=C1C1=CC=C(S(=O)(=O)N(CCO)CCO)C=C1 NAALWFYYHHJEFQ-ZASNTINBSA-N 0.000 description 1
- VEEGZPWAAPPXRB-BJMVGYQFSA-N (3e)-3-(1h-imidazol-5-ylmethylidene)-1h-indol-2-one Chemical compound O=C1NC2=CC=CC=C2\C1=C/C1=CN=CN1 VEEGZPWAAPPXRB-BJMVGYQFSA-N 0.000 description 1
- PUDHBTGHUJUUFI-SCTWWAJVSA-N (4r,7s,10s,13r,16s,19r)-10-(4-aminobutyl)-n-[(2s,3r)-1-amino-3-hydroxy-1-oxobutan-2-yl]-19-[[(2r)-2-amino-3-naphthalen-2-ylpropanoyl]amino]-16-[(4-hydroxyphenyl)methyl]-13-(1h-indol-3-ylmethyl)-6,9,12,15,18-pentaoxo-7-propan-2-yl-1,2-dithia-5,8,11,14,17-p Chemical compound C([C@H]1C(=O)N[C@H](CC=2C3=CC=CC=C3NC=2)C(=O)N[C@@H](CCCCN)C(=O)N[C@H](C(N[C@@H](CSSC[C@@H](C(=O)N1)NC(=O)[C@H](N)CC=1C=C2C=CC=CC2=CC=1)C(=O)N[C@@H]([C@@H](C)O)C(N)=O)=O)C(C)C)C1=CC=C(O)C=C1 PUDHBTGHUJUUFI-SCTWWAJVSA-N 0.000 description 1
- YJGVMLPVUAXIQN-LGWHJFRWSA-N (5s,5ar,8ar,9r)-5-hydroxy-9-(3,4,5-trimethoxyphenyl)-5a,6,8a,9-tetrahydro-5h-[2]benzofuro[5,6-f][1,3]benzodioxol-8-one Chemical compound COC1=C(OC)C(OC)=CC([C@@H]2C3=CC=4OCOC=4C=C3[C@@H](O)[C@@H]3[C@@H]2C(OC3)=O)=C1 YJGVMLPVUAXIQN-LGWHJFRWSA-N 0.000 description 1
- WTSKMKRYHATLLL-UHFFFAOYSA-N (6-benzoyloxy-3-cyanopyridin-2-yl) 3-[3-(ethoxymethyl)-5-fluoro-2,6-dioxopyrimidine-1-carbonyl]benzoate Chemical compound O=C1N(COCC)C=C(F)C(=O)N1C(=O)C1=CC=CC(C(=O)OC=2C(=CC=C(OC(=O)C=3C=CC=CC=3)N=2)C#N)=C1 WTSKMKRYHATLLL-UHFFFAOYSA-N 0.000 description 1
- LKBBOPGQDRPCDS-YAOXHJNESA-N (7s,9r,10r)-7-[(2r,4s,5s,6s)-4-amino-5-hydroxy-6-methyloxan-2-yl]oxy-9-ethyl-4,6,9,10,11-pentahydroxy-8,10-dihydro-7h-tetracene-5,12-dione Chemical compound O([C@H]1C[C@]([C@@H](C2=C(O)C=3C(=O)C4=CC=CC(O)=C4C(=O)C=3C(O)=C21)O)(O)CC)[C@H]1C[C@H](N)[C@H](O)[C@H](C)O1 LKBBOPGQDRPCDS-YAOXHJNESA-N 0.000 description 1
- MWWSFMDVAYGXBV-FGBSZODSSA-N (7s,9s)-7-[(2r,4s,5r,6s)-4-amino-5-hydroxy-6-methyloxan-2-yl]oxy-6,9,11-trihydroxy-9-(2-hydroxyacetyl)-4-methoxy-8,10-dihydro-7h-tetracene-5,12-dione;hydron;chloride Chemical compound Cl.O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(=O)CO)[C@H]1C[C@H](N)[C@@H](O)[C@H](C)O1 MWWSFMDVAYGXBV-FGBSZODSSA-N 0.000 description 1
- GYPCWHHQAVLMKO-XXKQIVDLSA-N (7s,9s)-7-[(2r,4s,5s,6s)-4-amino-5-hydroxy-6-methyloxan-2-yl]oxy-6,9,11-trihydroxy-9-[(e)-n-[(1-hydroxy-2,2,6,6-tetramethylpiperidin-4-ylidene)amino]-c-methylcarbonimidoyl]-4-methoxy-8,10-dihydro-7h-tetracene-5,12-dione;hydrochloride Chemical group Cl.O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(\C)=N\N=C1CC(C)(C)N(O)C(C)(C)C1)[C@H]1C[C@H](N)[C@H](O)[C@H](C)O1 GYPCWHHQAVLMKO-XXKQIVDLSA-N 0.000 description 1
- RCFNNLSZHVHCEK-YGCMNLPTSA-N (7s,9s)-7-[(2s,4r,6s)-4-amino-6-methyloxan-2-yl]oxy-6,9,11-trihydroxy-9-(2-hydroxyacetyl)-4-methoxy-8,10-dihydro-7h-tetracene-5,12-dione;hydrochloride Chemical compound Cl.O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(=O)CO)[C@H]1C[C@H](N)C[C@H](C)O1 RCFNNLSZHVHCEK-YGCMNLPTSA-N 0.000 description 1
- FPVKHBSQESCIEP-UHFFFAOYSA-N (8S)-3-(2-deoxy-beta-D-erythro-pentofuranosyl)-3,6,7,8-tetrahydroimidazo[4,5-d][1,3]diazepin-8-ol Natural products C1C(O)C(CO)OC1N1C(NC=NCC2O)=C2N=C1 FPVKHBSQESCIEP-UHFFFAOYSA-N 0.000 description 1
- IEXUMDBQLIVNHZ-YOUGDJEHSA-N (8s,11r,13r,14s,17s)-11-[4-(dimethylamino)phenyl]-17-hydroxy-17-(3-hydroxypropyl)-13-methyl-1,2,6,7,8,11,12,14,15,16-decahydrocyclopenta[a]phenanthren-3-one Chemical compound C1=CC(N(C)C)=CC=C1[C@@H]1C2=C3CCC(=O)C=C3CC[C@H]2[C@H](CC[C@]2(O)CCCO)[C@@]2(C)C1 IEXUMDBQLIVNHZ-YOUGDJEHSA-N 0.000 description 1
- ALSTYHKOOCGGFT-KTKRTIGZSA-N (9Z)-octadecen-1-ol Chemical compound CCCCCCCC\C=C/CCCCCCCCO ALSTYHKOOCGGFT-KTKRTIGZSA-N 0.000 description 1
- PMJHHCWVYXUKFD-SNAWJCMRSA-N (E)-1,3-pentadiene Chemical group C\C=C\C=C PMJHHCWVYXUKFD-SNAWJCMRSA-N 0.000 description 1
- FDKXTQMXEQVLRF-ZHACJKMWSA-N (E)-dacarbazine Chemical compound CN(C)\N=N\c1[nH]cnc1C(N)=O FDKXTQMXEQVLRF-ZHACJKMWSA-N 0.000 description 1
- 239000001707 (E,7R,11R)-3,7,11,15-tetramethylhexadec-2-en-1-ol Substances 0.000 description 1
- NSMXQKNUPPXBRG-SECBINFHSA-N (R)-lisofylline Chemical compound O=C1N(CCCC[C@H](O)C)C(=O)N(C)C2=C1N(C)C=N2 NSMXQKNUPPXBRG-SECBINFHSA-N 0.000 description 1
- SYTBZMRGLBWNTM-JTQLQIEISA-N (S)-flurbiprofen Chemical compound FC1=CC([C@@H](C(O)=O)C)=CC=C1C1=CC=CC=C1 SYTBZMRGLBWNTM-JTQLQIEISA-N 0.000 description 1
- YLRXAIKMLINXQY-ZDUSSCGKSA-O (S)-magnoflorine Chemical compound C1=C(OC)C(O)=C2C3=C(O)C(OC)=CC=C3C[C@@H]3[N+](C)(C)CCC1=C23 YLRXAIKMLINXQY-ZDUSSCGKSA-O 0.000 description 1
- 108091032973 (ribonucleotides)n+m Proteins 0.000 description 1
- 102000040650 (ribonucleotides)n+m Human genes 0.000 description 1
- NWUYHJFMYQTDRP-UHFFFAOYSA-N 1,2-bis(ethenyl)benzene;1-ethenyl-2-ethylbenzene;styrene Chemical compound C=CC1=CC=CC=C1.CCC1=CC=CC=C1C=C.C=CC1=CC=CC=C1C=C NWUYHJFMYQTDRP-UHFFFAOYSA-N 0.000 description 1
- FFGSXKJJVBXWCY-UHFFFAOYSA-N 1,4-bis[2-(2-hydroxyethylamino)ethylamino]anthracene-9,10-dione Chemical compound O=C1C2=CC=CC=C2C(=O)C2=C1C(NCCNCCO)=CC=C2NCCNCCO FFGSXKJJVBXWCY-UHFFFAOYSA-N 0.000 description 1
- OUPZKGBUJRBPGC-HLTSFMKQSA-N 1,5-bis[[(2r)-oxiran-2-yl]methyl]-3-[[(2s)-oxiran-2-yl]methyl]-1,3,5-triazinane-2,4,6-trione Chemical compound O=C1N(C[C@H]2OC2)C(=O)N(C[C@H]2OC2)C(=O)N1C[C@H]1CO1 OUPZKGBUJRBPGC-HLTSFMKQSA-N 0.000 description 1
- UOAFGUOASVSLPK-UHFFFAOYSA-N 1-(2-chloroethyl)-3-(2,2-dimethylpropyl)-1-nitrosourea Chemical compound CC(C)(C)CNC(=O)N(N=O)CCCl UOAFGUOASVSLPK-UHFFFAOYSA-N 0.000 description 1
- YQYBWJPESSJLTK-HXFLIBJXSA-N 1-(2-chloroethyl)-3-[(2r,3s,4r,6s)-3-hydroxy-2-(hydroxymethyl)-6-methoxyoxan-4-yl]-1-nitrosourea Chemical compound CO[C@@H]1C[C@@H](NC(=O)N(CCCl)N=O)[C@H](O)[C@@H](CO)O1 YQYBWJPESSJLTK-HXFLIBJXSA-N 0.000 description 1
- RCLLNBVPCJDIPX-UHFFFAOYSA-N 1-(2-chloroethyl)-3-[2-(dimethylsulfamoyl)ethyl]-1-nitrosourea Chemical compound CN(C)S(=O)(=O)CCNC(=O)N(N=O)CCCl RCLLNBVPCJDIPX-UHFFFAOYSA-N 0.000 description 1
- JQJSFAJISYZPER-UHFFFAOYSA-N 1-(4-chlorophenyl)-3-(2,3-dihydro-1h-inden-5-ylsulfonyl)urea Chemical compound C1=CC(Cl)=CC=C1NC(=O)NS(=O)(=O)C1=CC=C(CCC2)C2=C1 JQJSFAJISYZPER-UHFFFAOYSA-N 0.000 description 1
- SNYUHPPZINRDSG-UHFFFAOYSA-N 1-(oxiran-2-ylmethyl)-4-[1-(oxiran-2-ylmethyl)piperidin-4-yl]piperidine Chemical compound C1CC(C2CCN(CC3OC3)CC2)CCN1CC1CO1 SNYUHPPZINRDSG-UHFFFAOYSA-N 0.000 description 1
- GJNXBNATEDXMAK-RXXWGPEVSA-N 1-[(2s)-2-[[(2s)-2-[[(2r)-2-[[(2r)-2-[[(2r)-2-[[(2s)-2-[[(2s)-2-[(2-acetamido-3-naphthalen-2-ylpropanoyl)amino]-3-(4-chlorophenyl)propanoyl]amino]-3-pyridin-3-ylpropanoyl]amino]-3-hydroxypropanoyl]amino]-3-(4-hydroxyphenyl)propanoyl]amino]-6-[bis(ethylami Chemical compound C([C@H](C(=O)N[C@H](CCCCN=C(NCC)NCC)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCN=C(NCC)NCC)C(=O)N1C(CCC1)C(=O)N[C@H](C)C(N)=O)NC(=O)[C@@H](CO)NC(=O)[C@H](CC=1C=NC=CC=1)NC(=O)[C@H](CC=1C=CC(Cl)=CC=1)NC(=O)C(CC=1C=C2C=CC=CC2=CC=1)NC(C)=O)C1=CC=C(O)C=C1 GJNXBNATEDXMAK-RXXWGPEVSA-N 0.000 description 1
- ZKFNOUUKULVDOB-UHFFFAOYSA-N 1-amino-1-phenylmethyl phosphonic acid Chemical compound OP(=O)(O)C(N)C1=CC=CC=C1 ZKFNOUUKULVDOB-UHFFFAOYSA-N 0.000 description 1
- UKNVCOILWOLTLJ-UHFFFAOYSA-N 10-(3-aminopropylimino)-6,8-dihydroxy-14-[2-(2-hydroxyethylamino)ethyl]-14,15-diazatetracyclo[7.6.1.02,7.013,16]hexadeca-1,4,6,8,11,13(16)-hexaen-3-one Chemical compound C1=CC(=NCCCN)C2=C(C3=C(C=CC(=O)C3=C4C2=C1N(N4)CCNCCO)O)O UKNVCOILWOLTLJ-UHFFFAOYSA-N 0.000 description 1
- WJFKNYWRSNBZNX-UHFFFAOYSA-N 10H-phenothiazine Chemical compound C1=CC=C2NC3=CC=CC=C3SC2=C1 WJFKNYWRSNBZNX-UHFFFAOYSA-N 0.000 description 1
- OVSKIKFHRZPJSS-UHFFFAOYSA-N 2,4-D Chemical class OC(=O)COC1=CC=C(Cl)C=C1Cl OVSKIKFHRZPJSS-UHFFFAOYSA-N 0.000 description 1
- XMGJGSKRRWXOIF-UHFFFAOYSA-N 2-(azepan-1-yl)ethyl 2-cyclohexyl-2-thiophen-3-ylacetate;2-hydroxypropane-1,2,3-tricarboxylic acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O.C1CCCCC1C(C1=CSC=C1)C(=O)OCCN1CCCCCC1 XMGJGSKRRWXOIF-UHFFFAOYSA-N 0.000 description 1
- FCAJYRVEBULFKS-UHFFFAOYSA-N 2-(oxolan-2-yl)ethanol Chemical compound OCCC1CCCO1 FCAJYRVEBULFKS-UHFFFAOYSA-N 0.000 description 1
- PDWUPXJEEYOOTR-UHFFFAOYSA-N 2-[(3-iodophenyl)methyl]guanidine Chemical compound NC(=N)NCC1=CC=CC(I)=C1 PDWUPXJEEYOOTR-UHFFFAOYSA-N 0.000 description 1
- KPRFMAZESAKTEJ-UHFFFAOYSA-N 2-[1-amino-4-[2,5-dioxo-4-(1-phenylethyl)pyrrolidin-3-yl]-1-oxobutan-2-yl]-5-carbamoylheptanedioic acid;azane Chemical compound [NH4+].[NH4+].C=1C=CC=CC=1C(C)C1C(CCC(C(CCC(CC([O-])=O)C(N)=O)C([O-])=O)C(N)=O)C(=O)NC1=O KPRFMAZESAKTEJ-UHFFFAOYSA-N 0.000 description 1
- XXVLKDRPHSFIIB-UHFFFAOYSA-N 2-[2-(dimethylamino)ethyl]-5-nitrobenzo[de]isoquinoline-1,3-dione Chemical compound [O-][N+](=O)C1=CC(C(N(CCN(C)C)C2=O)=O)=C3C2=CC=CC3=C1 XXVLKDRPHSFIIB-UHFFFAOYSA-N 0.000 description 1
- MHXVDXXARZCVRK-WCWDXBQESA-N 2-[2-[4-[(e)-3,3,3-trifluoro-1,2-diphenylprop-1-enyl]phenoxy]ethylamino]ethanol Chemical compound C1=CC(OCCNCCO)=CC=C1C(\C=1C=CC=CC=1)=C(C(F)(F)F)/C1=CC=CC=C1 MHXVDXXARZCVRK-WCWDXBQESA-N 0.000 description 1
- JKMHFZQWWAIEOD-UHFFFAOYSA-N 2-[4-(2-hydroxyethyl)piperazin-1-yl]ethanesulfonic acid Chemical compound OCC[NH+]1CCN(CCS([O-])(=O)=O)CC1 JKMHFZQWWAIEOD-UHFFFAOYSA-N 0.000 description 1
- PXJJOGITBQXZEQ-JTHROIFXSA-M 2-[4-[(z)-1,2-diphenylbut-1-enyl]phenoxy]ethyl-trimethylazanium;iodide Chemical compound [I-].C=1C=CC=CC=1C(/CC)=C(C=1C=CC(OCC[N+](C)(C)C)=CC=1)/C1=CC=CC=C1 PXJJOGITBQXZEQ-JTHROIFXSA-M 0.000 description 1
- HYHJFNXFVPGMBI-UHFFFAOYSA-N 2-[[2-chloroethyl(nitroso)carbamoyl]-methylamino]acetamide Chemical compound NC(=O)CN(C)C(=O)N(CCCl)N=O HYHJFNXFVPGMBI-UHFFFAOYSA-N 0.000 description 1
- VDCRFBBZFHHYGT-IOSLPCCCSA-N 2-amino-9-[(2r,3r,4s,5r)-3,4-dihydroxy-5-(hydroxymethyl)oxolan-2-yl]-7-prop-2-enyl-3h-purine-6,8-dione Chemical compound O=C1N(CC=C)C=2C(=O)NC(N)=NC=2N1[C@@H]1O[C@H](CO)[C@@H](O)[C@H]1O VDCRFBBZFHHYGT-IOSLPCCCSA-N 0.000 description 1
- NIXVOFULDIFBLB-QVRNUERCSA-N 2-amino-9-[(2r,3r,4s,5r)-3,4-dihydroxy-5-(hydroxymethyl)oxolan-2-yl]purine-6-sulfinamide Chemical compound C12=NC(N)=NC(S(N)=O)=C2N=CN1[C@@H]1O[C@H](CO)[C@@H](O)[C@H]1O NIXVOFULDIFBLB-QVRNUERCSA-N 0.000 description 1
- GNIDUJTWLLFYKT-UHFFFAOYSA-N 2-hydroxy-2-[[5-hydroxy-4-(hydroxymethyl)-6-methylpyridin-3-yl]methoxy]acetic acid;[3-nitrooxy-2,2-bis(nitrooxymethyl)propyl] nitrate Chemical compound CC1=NC=C(COC(O)C(O)=O)C(CO)=C1O.[O-][N+](=O)OCC(CO[N+]([O-])=O)(CO[N+]([O-])=O)CO[N+]([O-])=O GNIDUJTWLLFYKT-UHFFFAOYSA-N 0.000 description 1
- DSWLRNLRVBAVFC-UHFFFAOYSA-N 2-methylsulfinyl-1-pyridin-2-ylethanone Chemical compound CS(=O)CC(=O)C1=CC=CC=N1 DSWLRNLRVBAVFC-UHFFFAOYSA-N 0.000 description 1
- CTRPRMNBTVRDFH-UHFFFAOYSA-N 2-n-methyl-1,3,5-triazine-2,4,6-triamine Chemical compound CNC1=NC(N)=NC(N)=N1 CTRPRMNBTVRDFH-UHFFFAOYSA-N 0.000 description 1
- NDMPLJNOPCLANR-UHFFFAOYSA-N 3,4-dihydroxy-15-(4-hydroxy-18-methoxycarbonyl-5,18-seco-ibogamin-18-yl)-16-methoxy-1-methyl-6,7-didehydro-aspidospermidine-3-carboxylic acid methyl ester Natural products C1C(CC)(O)CC(CC2(C(=O)OC)C=3C(=CC4=C(C56C(C(C(O)C7(CC)C=CCN(C67)CC5)(O)C(=O)OC)N4C)C=3)OC)CN1CCC1=C2NC2=CC=CC=C12 NDMPLJNOPCLANR-UHFFFAOYSA-N 0.000 description 1
- GRLUHXSUZYFZCW-UHFFFAOYSA-N 3-(8,8-diethyl-2-aza-8-germaspiro[4.5]decan-2-yl)-n,n-dimethylpropan-1-amine;dihydrochloride Chemical compound Cl.Cl.C1C[Ge](CC)(CC)CCC11CN(CCCN(C)C)CC1 GRLUHXSUZYFZCW-UHFFFAOYSA-N 0.000 description 1
- QGJZLNKBHJESQX-UHFFFAOYSA-N 3-Epi-Betulin-Saeure Natural products C1CC(O)C(C)(C)C2CCC3(C)C4(C)CCC5(C(O)=O)CCC(C(=C)C)C5C4CCC3C21C QGJZLNKBHJESQX-UHFFFAOYSA-N 0.000 description 1
- WUIABRMSWOKTOF-OYALTWQYSA-O 3-[[2-[2-[2-[[(2s,3r)-2-[[(2s,3s,4r)-4-[[(2s,3r)-2-[[6-amino-2-[(1s)-3-amino-1-[[(2s)-2,3-diamino-3-oxopropyl]amino]-3-oxopropyl]-5-methylpyrimidine-4-carbonyl]amino]-3-[(2r,3s,4s,5s,6s)-3-[(2r,3s,4s,5r,6r)-4-carbamoyloxy-3,5-dihydroxy-6-(hydroxymethyl)ox Chemical compound OS(O)(=O)=O.N([C@H](C(=O)N[C@H](C)[C@@H](O)[C@H](C)C(=O)N[C@@H]([C@H](O)C)C(=O)NCCC=1SC=C(N=1)C=1SC=C(N=1)C(=O)NCCC[S+](C)C)[C@@H](O[C@H]1[C@H]([C@@H](O)[C@H](O)[C@H](CO)O1)O[C@@H]1[C@H]([C@@H](OC(N)=O)[C@H](O)[C@@H](CO)O1)O)C=1N=CNC=1)C(=O)C1=NC([C@H](CC(N)=O)NC[C@H](N)C(N)=O)=NC(N)=C1C WUIABRMSWOKTOF-OYALTWQYSA-O 0.000 description 1
- WELIVEBWRWAGOM-UHFFFAOYSA-N 3-amino-n-[2-[2-(3-aminopropanoylamino)ethyldisulfanyl]ethyl]propanamide Chemical compound NCCC(=O)NCCSSCCNC(=O)CCN WELIVEBWRWAGOM-UHFFFAOYSA-N 0.000 description 1
- KFIRODWJCYBBHY-UHFFFAOYSA-N 3-nitrophthalic acid Chemical compound OC(=O)C1=CC=CC([N+]([O-])=O)=C1C(O)=O KFIRODWJCYBBHY-UHFFFAOYSA-N 0.000 description 1
- CLOUCVRNYSHRCF-UHFFFAOYSA-N 3beta-Hydroxy-20(29)-Lupen-3,27-oic acid Natural products C1CC(O)C(C)(C)C2CCC3(C)C4(C(O)=O)CCC5(C)CCC(C(=C)C)C5C4CCC3C21C CLOUCVRNYSHRCF-UHFFFAOYSA-N 0.000 description 1
- AOJJSUZBOXZQNB-VTZDEGQISA-N 4'-epidoxorubicin Chemical compound O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(=O)CO)[C@H]1C[C@H](N)[C@@H](O)[C@H](C)O1 AOJJSUZBOXZQNB-VTZDEGQISA-N 0.000 description 1
- LIETVYHJBSLSSW-UHFFFAOYSA-N 4,6,9-trihydroxy-8-methyl-3,4-dihydro-2h-anthracen-1-one Chemical compound OC1CCC(=O)C2=C1C=C1C=C(O)C=C(C)C1=C2O LIETVYHJBSLSSW-UHFFFAOYSA-N 0.000 description 1
- HLLSOEKIMZEGFV-UHFFFAOYSA-N 4-(dibutylsulfamoyl)benzoic acid Chemical compound CCCCN(CCCC)S(=O)(=O)C1=CC=C(C(O)=O)C=C1 HLLSOEKIMZEGFV-UHFFFAOYSA-N 0.000 description 1
- HQFSNUYUXXPVKL-UHFFFAOYSA-N 4-[(4-fluorophenyl)methyl]-2-[1-(2-phenylethyl)azepan-4-yl]phthalazin-1-one Chemical compound C1=CC(F)=CC=C1CC(C1=CC=CC=C1C1=O)=NN1C1CCN(CCC=2C=CC=CC=2)CCC1 HQFSNUYUXXPVKL-UHFFFAOYSA-N 0.000 description 1
- SGOOQMRIPALTEL-UHFFFAOYSA-N 4-hydroxy-N,1-dimethyl-2-oxo-N-phenyl-3-quinolinecarboxamide Chemical compound OC=1C2=CC=CC=C2N(C)C(=O)C=1C(=O)N(C)C1=CC=CC=C1 SGOOQMRIPALTEL-UHFFFAOYSA-N 0.000 description 1
- NSUDGNLOXMLAEB-UHFFFAOYSA-N 5-(2-formyl-3-hydroxyphenoxy)pentanoic acid Chemical compound OC(=O)CCCCOC1=CC=CC(O)=C1C=O NSUDGNLOXMLAEB-UHFFFAOYSA-N 0.000 description 1
- PXLPCZJACKUXGP-UHFFFAOYSA-N 5-(3,4-dichlorophenyl)-6-ethylpyrimidine-2,4-diamine Chemical compound CCC1=NC(N)=NC(N)=C1C1=CC=C(Cl)C(Cl)=C1 PXLPCZJACKUXGP-UHFFFAOYSA-N 0.000 description 1
- APNRZHLOPQFNMR-WEIUTZTHSA-N 5-[(e)-5-[(1s)-2,2-dimethyl-6-methylidenecyclohexyl]-3-methylpent-2-enyl]phenazin-1-one Chemical compound C12=CC=CC=C2N=C(C(C=CC=2)=O)C=2N1C\C=C(/C)CC[C@@H]1C(=C)CCCC1(C)C APNRZHLOPQFNMR-WEIUTZTHSA-N 0.000 description 1
- IDPUKCWIGUEADI-UHFFFAOYSA-N 5-[bis(2-chloroethyl)amino]uracil Chemical compound ClCCN(CCCl)C1=CNC(=O)NC1=O IDPUKCWIGUEADI-UHFFFAOYSA-N 0.000 description 1
- NMUSYJAQQFHJEW-KVTDHHQDSA-N 5-azacytidine Chemical compound O=C1N=C(N)N=CN1[C@H]1[C@H](O)[C@H](O)[C@@H](CO)O1 NMUSYJAQQFHJEW-KVTDHHQDSA-N 0.000 description 1
- DQOGWKZQQBYYMW-LQGIGNHCSA-N 5-methyl-6-[(3,4,5-trimethoxyanilino)methyl]quinazoline-2,4-diamine;(2s,3s,4s,5r,6s)-3,4,5,6-tetrahydroxyoxane-2-carboxylic acid Chemical compound O[C@H]1O[C@H](C(O)=O)[C@@H](O)[C@H](O)[C@H]1O.COC1=C(OC)C(OC)=CC(NCC=2C(=C3C(N)=NC(N)=NC3=CC=2)C)=C1 DQOGWKZQQBYYMW-LQGIGNHCSA-N 0.000 description 1
- PXBZKHOQHTVCSQ-QZTJIDSGSA-N 5-nitro-2-[(2r)-1-[2-[[(2r)-2-(5-nitro-1,3-dioxobenzo[de]isoquinolin-2-yl)propyl]amino]ethylamino]propan-2-yl]benzo[de]isoquinoline-1,3-dione Chemical compound [O-][N+](=O)C1=CC(C(N([C@@H](CNCCNC[C@@H](C)N2C(C=3C=C(C=C4C=CC=C(C=34)C2=O)[N+]([O-])=O)=O)C)C2=O)=O)=C3C2=CC=CC3=C1 PXBZKHOQHTVCSQ-QZTJIDSGSA-N 0.000 description 1
- MMRCWWRFYLZGAE-ZBZRSYSASA-N 533u947v6q Chemical compound O([C@]12[C@H](OC(C)=O)[C@]3(CC)C=CCN4CC[C@@]5([C@H]34)[C@H]1N(C)C1=C5C=C(C(=C1)OC)[C@]1(C(=O)OC)C3=C(C4=CC=CC=C4N3)CCN3C[C@H](C1)C[C@@](C3)(O)CC)C(=O)N(CCCl)C2=O MMRCWWRFYLZGAE-ZBZRSYSASA-N 0.000 description 1
- ATCGGEJZONJOCL-UHFFFAOYSA-N 6-(2,5-dichlorophenyl)-1,3,5-triazine-2,4-diamine Chemical compound NC1=NC(N)=NC(C=2C(=CC=C(Cl)C=2)Cl)=N1 ATCGGEJZONJOCL-UHFFFAOYSA-N 0.000 description 1
- VJXSSYDSOJBUAV-UHFFFAOYSA-N 6-(2,5-dimethoxy-benzyl)-5-methyl-pyrido[2,3-d]pyrimidine-2,4-diamine Chemical compound COC1=CC=C(OC)C(CC=2C(=C3C(N)=NC(N)=NC3=NC=2)C)=C1 VJXSSYDSOJBUAV-UHFFFAOYSA-N 0.000 description 1
- OTSZCHORPMQCBZ-UHFFFAOYSA-N 6-[(3-chlorophenyl)-imidazol-1-ylmethyl]-1h-benzimidazole;hydron;chloride Chemical compound Cl.ClC1=CC=CC(C(C=2C=C3NC=NC3=CC=2)N2C=NC=C2)=C1 OTSZCHORPMQCBZ-UHFFFAOYSA-N 0.000 description 1
- LJIRBXZDQGQUOO-KVTDHHQDSA-N 6-amino-3-[(2r,3r,4s,5r)-3,4-dihydroxy-5-(hydroxymethyl)oxolan-2-yl]-1,4-dihydro-1,3,5-triazin-2-one Chemical compound C1NC(N)=NC(=O)N1[C@H]1[C@H](O)[C@H](O)[C@@H](CO)O1 LJIRBXZDQGQUOO-KVTDHHQDSA-N 0.000 description 1
- STQGQHZAVUOBTE-UHFFFAOYSA-N 7-Cyan-hept-2t-en-4,6-diinsaeure Natural products C1=2C(O)=C3C(=O)C=4C(OC)=CC=CC=4C(=O)C3=C(O)C=2CC(O)(C(C)=O)CC1OC1CC(N)C(O)C(C)O1 STQGQHZAVUOBTE-UHFFFAOYSA-N 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- 208000030507 AIDS Diseases 0.000 description 1
- 244000235603 Acacia catechu Species 0.000 description 1
- 244000215068 Acacia senegal Species 0.000 description 1
- 235000006491 Acacia senegal Nutrition 0.000 description 1
- DLFVBJFMPXGRIB-UHFFFAOYSA-N Acetamide Chemical compound CC(N)=O DLFVBJFMPXGRIB-UHFFFAOYSA-N 0.000 description 1
- RZVAJINKPMORJF-UHFFFAOYSA-N Acetaminophen Chemical compound CC(=O)NC1=CC=C(O)C=C1 RZVAJINKPMORJF-UHFFFAOYSA-N 0.000 description 1
- WFDIJRYMOXRFFG-UHFFFAOYSA-N Acetic anhydride Chemical compound CC(=O)OC(C)=O WFDIJRYMOXRFFG-UHFFFAOYSA-N 0.000 description 1
- 208000024893 Acute lymphoblastic leukemia Diseases 0.000 description 1
- 208000014697 Acute lymphocytic leukaemia Diseases 0.000 description 1
- 206010000871 Acute monocytic leukaemia Diseases 0.000 description 1
- 206010048998 Acute phase reaction Diseases 0.000 description 1
- 229910002016 Aerosil® 200 Inorganic materials 0.000 description 1
- 101710153593 Albumin A Proteins 0.000 description 1
- 239000005995 Aluminium silicate Substances 0.000 description 1
- 201000003076 Angiosarcoma Diseases 0.000 description 1
- 235000006226 Areca catechu Nutrition 0.000 description 1
- MJINRRBEMOLJAK-DCAQKATOSA-N Arg-Lys-Asp Chemical compound OC(=O)C[C@@H](C(O)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@@H](N)CCCN=C(N)N MJINRRBEMOLJAK-DCAQKATOSA-N 0.000 description 1
- BFYIZQONLCFLEV-DAELLWKTSA-N Aromasine Chemical compound O=C1C=C[C@]2(C)[C@H]3CC[C@](C)(C(CC4)=O)[C@@H]4[C@@H]3CC(=C)C2=C1 BFYIZQONLCFLEV-DAELLWKTSA-N 0.000 description 1
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 1
- 108010024976 Asparaginase Proteins 0.000 description 1
- 102000015790 Asparaginase Human genes 0.000 description 1
- 108700032558 Aspergillus restrictus MITF Proteins 0.000 description 1
- 206010003571 Astrocytoma Diseases 0.000 description 1
- 206010003591 Ataxia Diseases 0.000 description 1
- 241001263178 Auriparus Species 0.000 description 1
- YOZSEGPJAXTSFZ-ZETCQYMHSA-N Azatyrosine Chemical compound OC(=O)[C@@H](N)CC1=CC=C(O)C=N1 YOZSEGPJAXTSFZ-ZETCQYMHSA-N 0.000 description 1
- NOWKCMXCCJGMRR-UHFFFAOYSA-N Aziridine Chemical compound C1CN1 NOWKCMXCCJGMRR-UHFFFAOYSA-N 0.000 description 1
- 208000004736 B-Cell Leukemia Diseases 0.000 description 1
- VGGGPCQERPFHOB-MCIONIFRSA-N Bestatin Chemical compound CC(C)C[C@H](C(O)=O)NC(=O)[C@@H](O)[C@H](N)CC1=CC=CC=C1 VGGGPCQERPFHOB-MCIONIFRSA-N 0.000 description 1
- DIZWSDNSTNAYHK-XGWVBXMLSA-N Betulinic acid Natural products CC(=C)[C@@H]1C[C@H]([C@H]2CC[C@]3(C)[C@H](CC[C@@H]4[C@@]5(C)CC[C@H](O)C(C)(C)[C@@H]5CC[C@@]34C)[C@@H]12)C(=O)O DIZWSDNSTNAYHK-XGWVBXMLSA-N 0.000 description 1
- 206010005003 Bladder cancer Diseases 0.000 description 1
- 241000283690 Bos taurus Species 0.000 description 1
- 206010006187 Breast cancer Diseases 0.000 description 1
- 208000026310 Breast neoplasm Diseases 0.000 description 1
- COVZYZSDYWQREU-UHFFFAOYSA-N Busulfan Chemical compound CS(=O)(=O)OCCCCOS(C)(=O)=O COVZYZSDYWQREU-UHFFFAOYSA-N 0.000 description 1
- GAGWJHPBXLXJQN-UORFTKCHSA-N Capecitabine Chemical compound C1=C(F)C(NC(=O)OCCCCC)=NC(=O)N1[C@H]1[C@H](O)[C@H](O)[C@@H](C)O1 GAGWJHPBXLXJQN-UORFTKCHSA-N 0.000 description 1
- GAGWJHPBXLXJQN-UHFFFAOYSA-N Capecitabine Natural products C1=C(F)C(NC(=O)OCCCCC)=NC(=O)N1C1C(O)C(O)C(C)O1 GAGWJHPBXLXJQN-UHFFFAOYSA-N 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 1
- OKTJSMMVPCPJKN-NJFSPNSNSA-N Carbon-14 Chemical compound [14C] OKTJSMMVPCPJKN-NJFSPNSNSA-N 0.000 description 1
- 201000009030 Carcinoma Diseases 0.000 description 1
- 208000024172 Cardiovascular disease Diseases 0.000 description 1
- DLGOEMSEDOSKAD-UHFFFAOYSA-N Carmustine Chemical compound ClCCNC(=O)N(N=O)CCCl DLGOEMSEDOSKAD-UHFFFAOYSA-N 0.000 description 1
- 102000014914 Carrier Proteins Human genes 0.000 description 1
- JDVVGAQPNNXQDW-WCMLQCRESA-N Castanospermine Natural products O[C@H]1[C@@H](O)[C@H]2[C@@H](O)CCN2C[C@H]1O JDVVGAQPNNXQDW-WCMLQCRESA-N 0.000 description 1
- JDVVGAQPNNXQDW-TVNFTVLESA-N Castinospermine Chemical compound C1[C@H](O)[C@@H](O)[C@H](O)[C@H]2[C@@H](O)CCN21 JDVVGAQPNNXQDW-TVNFTVLESA-N 0.000 description 1
- 108090000994 Catalytic RNA Proteins 0.000 description 1
- 102000053642 Catalytic RNA Human genes 0.000 description 1
- 241000218645 Cedrus Species 0.000 description 1
- 101150015280 Cel gene Proteins 0.000 description 1
- 108010067225 Cell Adhesion Molecules Proteins 0.000 description 1
- 102000016289 Cell Adhesion Molecules Human genes 0.000 description 1
- 102000000844 Cell Surface Receptors Human genes 0.000 description 1
- 108010001857 Cell Surface Receptors Proteins 0.000 description 1
- 206010008342 Cervix carcinoma Diseases 0.000 description 1
- 108010012236 Chemokines Proteins 0.000 description 1
- 102000019034 Chemokines Human genes 0.000 description 1
- JWBOIMRXGHLCPP-UHFFFAOYSA-N Chloditan Chemical compound C=1C=CC=C(Cl)C=1C(C(Cl)Cl)C1=CC=C(Cl)C=C1 JWBOIMRXGHLCPP-UHFFFAOYSA-N 0.000 description 1
- 102000003914 Cholinesterases Human genes 0.000 description 1
- 108090000322 Cholinesterases Proteins 0.000 description 1
- 241000251730 Chondrichthyes Species 0.000 description 1
- 208000005243 Chondrosarcoma Diseases 0.000 description 1
- 201000009047 Chordoma Diseases 0.000 description 1
- 208000006332 Choriocarcinoma Diseases 0.000 description 1
- 208000001333 Colorectal Neoplasms Diseases 0.000 description 1
- HVXBOLULGPECHP-WAYWQWQTSA-N Combretastatin A4 Chemical compound C1=C(O)C(OC)=CC=C1\C=C/C1=CC(OC)=C(OC)C(OC)=C1 HVXBOLULGPECHP-WAYWQWQTSA-N 0.000 description 1
- DFDTZECTHJFPHE-UHFFFAOYSA-N Crambescidin 816 Natural products C1CC=CC(CC)OC11NC(N23)=NC4(OC(C)CCC4)C(C(=O)OCCCCCCCCCCCCCCCC(=O)N(CCCN)CC(O)CCN)C3(O)CCC2C1 DFDTZECTHJFPHE-UHFFFAOYSA-N 0.000 description 1
- 208000009798 Craniopharyngioma Diseases 0.000 description 1
- 241000195493 Cryptophyta Species 0.000 description 1
- LUEYTMPPCOCKBX-UHFFFAOYSA-N Curacin A Natural products C=CCC(OC)CCC(C)=CC=CCCC=CC1CSC(C2C(C2)C)=N1 LUEYTMPPCOCKBX-UHFFFAOYSA-N 0.000 description 1
- LUEYTMPPCOCKBX-KWYHTCOPSA-N Curacin A Chemical compound C=CC[C@H](OC)CC\C(C)=C\C=C\CC\C=C/[C@@H]1CSC([C@H]2[C@H](C2)C)=N1 LUEYTMPPCOCKBX-KWYHTCOPSA-N 0.000 description 1
- CMSMOCZEIVJLDB-UHFFFAOYSA-N Cyclophosphamide Chemical compound ClCCN(CCCl)P1(=O)NCCCO1 CMSMOCZEIVJLDB-UHFFFAOYSA-N 0.000 description 1
- 108010092160 Dactinomycin Proteins 0.000 description 1
- WEAHRLBPCANXCN-UHFFFAOYSA-N Daunomycin Natural products CCC1(O)CC(OC2CC(N)C(O)C(C)O2)c3cc4C(=O)c5c(OC)cccc5C(=O)c4c(O)c3C1 WEAHRLBPCANXCN-UHFFFAOYSA-N 0.000 description 1
- GJKXGJCSJWBJEZ-XRSSZCMZSA-N Deslorelin Chemical compound CCNC(=O)[C@@H]1CCCN1C(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@H](CC=1NC=NC=1)NC(=O)[C@H]1NC(=O)CC1)CC1=CNC2=CC=CC=C12 GJKXGJCSJWBJEZ-XRSSZCMZSA-N 0.000 description 1
- 101100135868 Dictyostelium discoideum pde3 gene Proteins 0.000 description 1
- 101100189582 Dictyostelium discoideum pdeD gene Proteins 0.000 description 1
- 101100351286 Dictyostelium discoideum pdeE gene Proteins 0.000 description 1
- 101100135859 Dictyostelium discoideum regA gene Proteins 0.000 description 1
- MWWSFMDVAYGXBV-RUELKSSGSA-N Doxorubicin hydrochloride Chemical compound Cl.O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(=O)CO)[C@H]1C[C@H](N)[C@H](O)[C@H](C)O1 MWWSFMDVAYGXBV-RUELKSSGSA-N 0.000 description 1
- CYQFCXCEBYINGO-DLBZAZTESA-N Dronabinol Natural products C1=C(C)CC[C@H]2C(C)(C)OC3=CC(CCCCC)=CC(O)=C3[C@H]21 CYQFCXCEBYINGO-DLBZAZTESA-N 0.000 description 1
- 101001117089 Drosophila melanogaster Calcium/calmodulin-dependent 3',5'-cyclic nucleotide phosphodiesterase 1 Proteins 0.000 description 1
- 101100229963 Drosophila melanogaster grau gene Proteins 0.000 description 1
- 208000000059 Dyspnea Diseases 0.000 description 1
- 206010013975 Dyspnoeas Diseases 0.000 description 1
- 239000004278 EU approved seasoning Substances 0.000 description 1
- DYEFUKCXAQOFHX-UHFFFAOYSA-N Ebselen Chemical compound [se]1C2=CC=CC=C2C(=O)N1C1=CC=CC=C1 DYEFUKCXAQOFHX-UHFFFAOYSA-N 0.000 description 1
- 201000009051 Embryonal Carcinoma Diseases 0.000 description 1
- NBEALWAVEGMZQY-UHFFFAOYSA-N Enpromate Chemical compound C=1C=CC=CC=1C(C#C)(C=1C=CC=CC=1)OC(=O)NC1CCCCC1 NBEALWAVEGMZQY-UHFFFAOYSA-N 0.000 description 1
- 206010014967 Ependymoma Diseases 0.000 description 1
- HTIJFSOGRVMCQR-UHFFFAOYSA-N Epirubicin Natural products COc1cccc2C(=O)c3c(O)c4CC(O)(CC(OC5CC(N)C(=O)C(C)O5)c4c(O)c3C(=O)c12)C(=O)CO HTIJFSOGRVMCQR-UHFFFAOYSA-N 0.000 description 1
- VAPSMQAHNAZRKC-PQWRYPMOSA-N Epristeride Chemical compound C1C=C2C=C(C(O)=O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H](C(=O)NC(C)(C)C)[C@@]1(C)CC2 VAPSMQAHNAZRKC-PQWRYPMOSA-N 0.000 description 1
- 208000036566 Erythroleukaemia Diseases 0.000 description 1
- 241000588724 Escherichia coli Species 0.000 description 1
- KGWDUNBJIMUFAP-KVVVOXFISA-N Ethanolamine Oleate Chemical compound NCCO.CCCCCCCC\C=C/CCCCCCCC(O)=O KGWDUNBJIMUFAP-KVVVOXFISA-N 0.000 description 1
- DBVJJBKOTRCVKF-UHFFFAOYSA-N Etidronic acid Chemical compound OP(=O)(O)C(O)(C)P(O)(O)=O DBVJJBKOTRCVKF-UHFFFAOYSA-N 0.000 description 1
- ITIONVBQFUNVJV-UHFFFAOYSA-N Etomidoline Chemical compound C12=CC=CC=C2C(=O)N(CC)C1NC(C=C1)=CC=C1OCCN1CCCCC1 ITIONVBQFUNVJV-UHFFFAOYSA-N 0.000 description 1
- 208000006168 Ewing Sarcoma Diseases 0.000 description 1
- 241000282326 Felis catus Species 0.000 description 1
- 102100024785 Fibroblast growth factor 2 Human genes 0.000 description 1
- 108090000379 Fibroblast growth factor 2 Proteins 0.000 description 1
- 201000008808 Fibrosarcoma Diseases 0.000 description 1
- 229920001917 Ficoll Polymers 0.000 description 1
- 108010029961 Filgrastim Proteins 0.000 description 1
- GHASVSINZRGABV-UHFFFAOYSA-N Fluorouracil Chemical compound FC1=CNC(=O)NC1=O GHASVSINZRGABV-UHFFFAOYSA-N 0.000 description 1
- PGTKVMVZBBZCKQ-UHFFFAOYSA-N Fulvene Chemical compound C=C1C=CC=C1 PGTKVMVZBBZCKQ-UHFFFAOYSA-N 0.000 description 1
- 206010059024 Gastrointestinal toxicity Diseases 0.000 description 1
- 108010010803 Gelatin Proteins 0.000 description 1
- 102000013382 Gelatinases Human genes 0.000 description 1
- 108010026132 Gelatinases Proteins 0.000 description 1
- 208000032612 Glial tumor Diseases 0.000 description 1
- 206010018338 Glioma Diseases 0.000 description 1
- DHMQDGOQFOQNFH-UHFFFAOYSA-N Glycine Chemical compound NCC(O)=O DHMQDGOQFOQNFH-UHFFFAOYSA-N 0.000 description 1
- 244000068988 Glycine max Species 0.000 description 1
- 235000010469 Glycine max Nutrition 0.000 description 1
- 108010015899 Glycopeptides Proteins 0.000 description 1
- 102000002068 Glycopeptides Human genes 0.000 description 1
- 102100039620 Granulocyte-macrophage colony-stimulating factor Human genes 0.000 description 1
- 229920002907 Guar gum Polymers 0.000 description 1
- 239000007995 HEPES buffer Substances 0.000 description 1
- 239000012981 Hank's balanced salt solution Substances 0.000 description 1
- 206010019233 Headaches Diseases 0.000 description 1
- 208000001258 Hemangiosarcoma Diseases 0.000 description 1
- 206010019851 Hepatotoxicity Diseases 0.000 description 1
- 101000917858 Homo sapiens Low affinity immunoglobulin gamma Fc region receptor III-A Proteins 0.000 description 1
- 101000917839 Homo sapiens Low affinity immunoglobulin gamma Fc region receptor III-B Proteins 0.000 description 1
- 101000582950 Homo sapiens Platelet factor 4 Proteins 0.000 description 1
- 101000807541 Homo sapiens Ubiquitin carboxyl-terminal hydrolase 24 Proteins 0.000 description 1
- 241000598436 Human T-cell lymphotropic virus Species 0.000 description 1
- AVXURJPOCDRRFD-UHFFFAOYSA-N Hydroxylamine Chemical class ON AVXURJPOCDRRFD-UHFFFAOYSA-N 0.000 description 1
- VSNHCAURESNICA-UHFFFAOYSA-N Hydroxyurea Chemical compound NC(=O)NO VSNHCAURESNICA-UHFFFAOYSA-N 0.000 description 1
- MPBVHIBUJCELCL-UHFFFAOYSA-N Ibandronate Chemical compound CCCCCN(C)CCC(O)(P(O)(O)=O)P(O)(O)=O MPBVHIBUJCELCL-UHFFFAOYSA-N 0.000 description 1
- HEFNNWSXXWATRW-UHFFFAOYSA-N Ibuprofen Chemical compound CC(C)CC1=CC=C(C(C)C(O)=O)C=C1 HEFNNWSXXWATRW-UHFFFAOYSA-N 0.000 description 1
- XDXDZDZNSLXDNA-UHFFFAOYSA-N Idarubicin Natural products C1C(N)C(O)C(C)OC1OC1C2=C(O)C(C(=O)C3=CC=CC=C3C3=O)=C3C(O)=C2CC(O)(C(C)=O)C1 XDXDZDZNSLXDNA-UHFFFAOYSA-N 0.000 description 1
- 244000188472 Ilex paraguariensis Species 0.000 description 1
- 108700022013 Insecta cecropin B Proteins 0.000 description 1
- 102100040018 Interferon alpha-2 Human genes 0.000 description 1
- 108010079944 Interferon-alpha2b Proteins 0.000 description 1
- 102000013462 Interleukin-12 Human genes 0.000 description 1
- 108010065805 Interleukin-12 Proteins 0.000 description 1
- 244000017020 Ipomoea batatas Species 0.000 description 1
- 235000002678 Ipomoea batatas Nutrition 0.000 description 1
- 108010044467 Isoenzymes Proteins 0.000 description 1
- 208000007766 Kaposi sarcoma Diseases 0.000 description 1
- 102100040441 Keratin, type I cytoskeletal 16 Human genes 0.000 description 1
- 101710183400 Keratin, type I cytoskeletal 16 Proteins 0.000 description 1
- 201000003129 Kidney Papillary Necrosis Diseases 0.000 description 1
- 239000005909 Kieselgur Substances 0.000 description 1
- 108010092694 L-Selectin Proteins 0.000 description 1
- ODKSFYDXXFIFQN-BYPYZUCNSA-N L-arginine Chemical compound OC(=O)[C@@H](N)CCCN=C(N)N ODKSFYDXXFIFQN-BYPYZUCNSA-N 0.000 description 1
- 239000004395 L-leucine Substances 0.000 description 1
- 108010043135 L-methionine gamma-lyase Proteins 0.000 description 1
- FBOZXECLQNJBKD-ZDUSSCGKSA-N L-methotrexate Chemical compound C=1N=C2N=C(N)N=C(N)C2=NC=1CN(C)C1=CC=C(C(=O)N[C@@H](CCC(O)=O)C(O)=O)C=C1 FBOZXECLQNJBKD-ZDUSSCGKSA-N 0.000 description 1
- 102000016551 L-selectin Human genes 0.000 description 1
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 1
- ZHTRILQJTPJGNK-FYBAATNNSA-N Leinamycin Chemical compound N([C@@H](C=1SC=C(N=1)\C=C/C=C/C(=O)[C@H](O)/C=C(C)/CC1)C)C(=O)C[C@@]21S(=O)SC(=O)[C@]2(C)O ZHTRILQJTPJGNK-FYBAATNNSA-N 0.000 description 1
- ZHTRILQJTPJGNK-UHFFFAOYSA-N Leinamycin Natural products C1CC(C)=CC(O)C(=O)C=CC=CC(N=2)=CSC=2C(C)NC(=O)CC21S(=O)SC(=O)C2(C)O ZHTRILQJTPJGNK-UHFFFAOYSA-N 0.000 description 1
- 208000018142 Leiomyosarcoma Diseases 0.000 description 1
- 108010062867 Lenograstim Proteins 0.000 description 1
- 229920001491 Lentinan Polymers 0.000 description 1
- LMVRPBWWHMVLPC-KBPJCXPTSA-N Leptolstatin Natural products CC(CC=CC(=CC(C)C(=O)C(C)C(O)C(C)CC(=CCO)C)C)C=C(C)/C=C/C1CC=CC(=O)O1 LMVRPBWWHMVLPC-KBPJCXPTSA-N 0.000 description 1
- LPGWZGMPDKDHEP-HLTPFJCJSA-N Leurosine Chemical compound C([C@]1([C@@H]2O1)CC)N(CCC=1C3=CC=CC=C3NC=11)C[C@H]2C[C@]1(C(=O)OC)C1=CC([C@]23[C@H]([C@@]([C@H](OC(C)=O)[C@]4(CC)C=CCN([C@H]34)CC2)(O)C(=O)OC)N2C)=C2C=C1OC LPGWZGMPDKDHEP-HLTPFJCJSA-N 0.000 description 1
- LPGWZGMPDKDHEP-GKWAKPNHSA-N Leurosine Natural products O=C(O[C@H]1[C@](O)(C(=O)OC)[C@@H]2N(C)c3c(cc(c(OC)c3)[C@]3(C(=O)OC)c4[nH]c5c(c4CCN4C[C@]6(CC)O[C@@H]6[C@H](C3)C4)cccc5)[C@@]32[C@H]2[C@@]1(CC)C=CCN2CC3)C LPGWZGMPDKDHEP-GKWAKPNHSA-N 0.000 description 1
- HLFSDGLLUJUHTE-SNVBAGLBSA-N Levamisole Chemical compound C1([C@H]2CN3CCSC3=N2)=CC=CC=C1 HLFSDGLLUJUHTE-SNVBAGLBSA-N 0.000 description 1
- GQYIWUVLTXOXAJ-UHFFFAOYSA-N Lomustine Chemical compound ClCCN(N=O)C(=O)NC1CCCCC1 GQYIWUVLTXOXAJ-UHFFFAOYSA-N 0.000 description 1
- 102100029185 Low affinity immunoglobulin gamma Fc region receptor III-B Human genes 0.000 description 1
- 206010058467 Lung neoplasm malignant Diseases 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- KYEAXNAYHSCLMT-CVVGWEDFSA-N Magnoflorine Natural products C[C@H]1OC=C2[C@@H]3[C@@H]1CN4CCc5c([nH]c6ccccc56)[C@@H]4[C@@H]3OC2=O KYEAXNAYHSCLMT-CVVGWEDFSA-N 0.000 description 1
- 240000003183 Manihot esculenta Species 0.000 description 1
- 235000016735 Manihot esculenta subsp esculenta Nutrition 0.000 description 1
- BLOFGONIVNXZME-UHFFFAOYSA-N Mannostatin A Natural products CSC1C(N)C(O)C(O)C1O BLOFGONIVNXZME-UHFFFAOYSA-N 0.000 description 1
- 206010026851 Marrow hyperplasia Diseases 0.000 description 1
- WSTYNZDAOAEEKG-UHFFFAOYSA-N Mayol Natural products CC1=C(O)C(=O)C=C2C(CCC3(C4CC(C(CC4(CCC33C)C)=O)C)C)(C)C3=CC=C21 WSTYNZDAOAEEKG-UHFFFAOYSA-N 0.000 description 1
- 229930126263 Maytansine Natural products 0.000 description 1
- WSTYNZDAOAEEKG-QSPBTJQRSA-N Maytenin Natural products CC1=C(O)C(=O)C=C2[C@@](CC[C@@]3([C@@H]4C[C@H](C(C[C@@]4(CC[C@]33C)C)=O)C)C)(C)C3=CC=C21 WSTYNZDAOAEEKG-QSPBTJQRSA-N 0.000 description 1
- 208000007054 Medullary Carcinoma Diseases 0.000 description 1
- 208000000172 Medulloblastoma Diseases 0.000 description 1
- ZRVUJXDFFKFLMG-UHFFFAOYSA-N Meloxicam Chemical compound OC=1C2=CC=CC=C2S(=O)(=O)N(C)C=1C(=O)NC1=NC=C(C)S1 ZRVUJXDFFKFLMG-UHFFFAOYSA-N 0.000 description 1
- 206010027406 Mesothelioma Diseases 0.000 description 1
- 241000361919 Metaphire sieboldi Species 0.000 description 1
- PCZOHLXUXFIOCF-UHFFFAOYSA-N Monacolin X Natural products C12C(OC(=O)C(C)CC)CC(C)C=C2C=CC(C)C1CCC1CC(O)CC(=O)O1 PCZOHLXUXFIOCF-UHFFFAOYSA-N 0.000 description 1
- 208000035489 Monocytic Acute Leukemia Diseases 0.000 description 1
- 101000686985 Mouse mammary tumor virus (strain C3H) Protein PR73 Proteins 0.000 description 1
- 241000282339 Mustela Species 0.000 description 1
- 101000909851 Mycobacterium tuberculosis (strain ATCC 25618 / H37Rv) cAMP/cGMP dual specificity phosphodiesterase Rv0805 Proteins 0.000 description 1
- 208000033761 Myelogenous Chronic BCR-ABL Positive Leukemia Diseases 0.000 description 1
- 208000033776 Myeloid Acute Leukemia Diseases 0.000 description 1
- 201000007224 Myeloproliferative neoplasm Diseases 0.000 description 1
- NWIBSHFKIJFRCO-WUDYKRTCSA-N Mytomycin Chemical class C1N2C(C(C(C)=C(N)C3=O)=O)=C3[C@@H](COC(N)=O)[C@@]2(OC)[C@@H]2[C@H]1N2 NWIBSHFKIJFRCO-WUDYKRTCSA-N 0.000 description 1
- USVMJSALORZVDV-SDBHATRESA-N N(6)-(Delta(2)-isopentenyl)adenosine Chemical compound C1=NC=2C(NCC=C(C)C)=NC=NC=2N1[C@@H]1O[C@H](CO)[C@@H](O)[C@H]1O USVMJSALORZVDV-SDBHATRESA-N 0.000 description 1
- FXHOOIRPVKKKFG-UHFFFAOYSA-N N,N-Dimethylacetamide Chemical compound CN(C)C(C)=O FXHOOIRPVKKKFG-UHFFFAOYSA-N 0.000 description 1
- WUKZPHOXUVCQOR-UHFFFAOYSA-N N-(1-azabicyclo[2.2.2]octan-3-yl)-6-chloro-4-methyl-3-oxo-1,4-benzoxazine-8-carboxamide Chemical compound C1N(CC2)CCC2C1NC(=O)C1=CC(Cl)=CC2=C1OCC(=O)N2C WUKZPHOXUVCQOR-UHFFFAOYSA-N 0.000 description 1
- 102000003945 NF-kappa B Human genes 0.000 description 1
- 108010057466 NF-kappa B Proteins 0.000 description 1
- 108010021717 Nafarelin Proteins 0.000 description 1
- CMWTZPSULFXXJA-UHFFFAOYSA-N Naproxen Natural products C1=C(C(C)C(O)=O)C=CC2=CC(OC)=CC=C21 CMWTZPSULFXXJA-UHFFFAOYSA-N 0.000 description 1
- 102400000058 Neuregulin-1 Human genes 0.000 description 1
- 108090000556 Neuregulin-1 Proteins 0.000 description 1
- 206010029260 Neuroblastoma Diseases 0.000 description 1
- BUSGWUFLNHIBPT-UHFFFAOYSA-N Nisamycin Natural products O=C1C2OC2C(C=CC=CC=CC(=O)O)(O)C=C1NC(=O)C=CC=CC1CCCCC1 BUSGWUFLNHIBPT-UHFFFAOYSA-N 0.000 description 1
- GQPLMRYTRLFLPF-UHFFFAOYSA-N Nitrous Oxide Chemical compound [O-][N+]#N GQPLMRYTRLFLPF-UHFFFAOYSA-N 0.000 description 1
- KGTDRFCXGRULNK-UHFFFAOYSA-N Nogalamycin Natural products COC1C(OC)(C)C(OC)C(C)OC1OC1C2=C(O)C(C(=O)C3=C(O)C=C4C5(C)OC(C(C(C5O)N(C)C)O)OC4=C3C3=O)=C3C=C2C(C(=O)OC)C(C)(O)C1 KGTDRFCXGRULNK-UHFFFAOYSA-N 0.000 description 1
- 239000004677 Nylon Substances 0.000 description 1
- 229960005524 O6-benzylguanine Drugs 0.000 description 1
- KRWMERLEINMZFT-UHFFFAOYSA-N O6-benzylguanine Chemical compound C=12NC=NC2=NC(N)=NC=1OCC1=CC=CC=C1 KRWMERLEINMZFT-UHFFFAOYSA-N 0.000 description 1
- 201000010133 Oligodendroglioma Diseases 0.000 description 1
- VTAZRSXSBIHBMH-UHFFFAOYSA-N Ophiocordin Natural products OC1=CC(C(=O)O)=CC(O)=C1C(=O)C1=C(O)C=CC=C1C(=O)NC1C(OC(=O)C=2C=CC(O)=CC=2)CCCNC1 VTAZRSXSBIHBMH-UHFFFAOYSA-N 0.000 description 1
- 206010033128 Ovarian cancer Diseases 0.000 description 1
- 206010061535 Ovarian neoplasm Diseases 0.000 description 1
- LKBBOPGQDRPCDS-UHFFFAOYSA-N Oxaunomycin Natural products C12=C(O)C=3C(=O)C4=C(O)C=CC=C4C(=O)C=3C(O)=C2C(O)C(CC)(O)CC1OC1CC(N)C(O)C(C)O1 LKBBOPGQDRPCDS-UHFFFAOYSA-N 0.000 description 1
- 108010035766 P-Selectin Proteins 0.000 description 1
- 102000008212 P-Selectin Human genes 0.000 description 1
- 101150098694 PDE5A gene Proteins 0.000 description 1
- VYOQBYCIIJYKJA-UHFFFAOYSA-N Palauamine Natural products C1N2C(=O)C3=CC=CN3C3N=C(N)NC32C2C1C(CN)C(Cl)C12NC(N)=NC1O VYOQBYCIIJYKJA-UHFFFAOYSA-N 0.000 description 1
- 208000027089 Parkinsonian disease Diseases 0.000 description 1
- 206010034010 Parkinsonism Diseases 0.000 description 1
- 108010057150 Peplomycin Proteins 0.000 description 1
- 102000035195 Peptidases Human genes 0.000 description 1
- APNRZHLOPQFNMR-UHFFFAOYSA-N Phenazinomycin Natural products C12=CC=CC=C2N=C(C(C=CC=2)=O)C=2N1CC=C(C)CCC1C(=C)CCCC1(C)C APNRZHLOPQFNMR-UHFFFAOYSA-N 0.000 description 1
- 102000004160 Phosphoric Monoester Hydrolases Human genes 0.000 description 1
- 108090000608 Phosphoric Monoester Hydrolases Proteins 0.000 description 1
- BLUHKGOSFDHHGX-UHFFFAOYSA-N Phytol Natural products CC(C)CCCC(C)CCCC(C)CCCC(C)C=CO BLUHKGOSFDHHGX-UHFFFAOYSA-N 0.000 description 1
- 208000007641 Pinealoma Diseases 0.000 description 1
- KMSKQZKKOZQFFG-HSUXVGOQSA-N Pirarubicin Chemical compound O([C@H]1[C@@H](N)C[C@@H](O[C@H]1C)O[C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(=O)CO)[C@H]1CCCCO1 KMSKQZKKOZQFFG-HSUXVGOQSA-N 0.000 description 1
- 102000001938 Plasminogen Activators Human genes 0.000 description 1
- 108010001014 Plasminogen Activators Proteins 0.000 description 1
- 101100082606 Plasmodium falciparum (isolate 3D7) PDEbeta gene Proteins 0.000 description 1
- 102100030304 Platelet factor 4 Human genes 0.000 description 1
- 208000006664 Precursor Cell Lymphoblastic Leukemia-Lymphoma Diseases 0.000 description 1
- HFVNWDWLWUCIHC-GUPDPFMOSA-N Prednimustine Chemical compound O=C([C@@]1(O)CC[C@H]2[C@H]3[C@@H]([C@]4(C=CC(=O)C=C4CC3)C)[C@@H](O)C[C@@]21C)COC(=O)CCCC1=CC=C(N(CCCl)CCCl)C=C1 HFVNWDWLWUCIHC-GUPDPFMOSA-N 0.000 description 1
- 208000033826 Promyelocytic Acute Leukemia Diseases 0.000 description 1
- 239000004365 Protease Substances 0.000 description 1
- 229940079156 Proteasome inhibitor Drugs 0.000 description 1
- 102000002727 Protein Tyrosine Phosphatase Human genes 0.000 description 1
- PICZCWCKOLHDOJ-UHFFFAOYSA-N Pseudoaxinellin Natural products N1C(=O)C2CCCN2C(=O)C(CC(N)=O)NC(=O)C(C(C)C)NC(=O)C2CCCN2C(=O)C(C(C)C)NC(=O)C(C(C)C)NC(=O)C1CC1=CC=CC=C1 PICZCWCKOLHDOJ-UHFFFAOYSA-N 0.000 description 1
- 229940123827 Purine nucleoside phosphorylase inhibitor Drugs 0.000 description 1
- XESARGFCSKSFID-UHFFFAOYSA-N Pyrazofurin Natural products OC1=C(C(=O)N)NN=C1C1C(O)C(O)C(CO)O1 XESARGFCSKSFID-UHFFFAOYSA-N 0.000 description 1
- AHHFEZNOXOZZQA-ZEBDFXRSSA-N Ranimustine Chemical compound CO[C@H]1O[C@H](CNC(=O)N(CCCl)N=O)[C@@H](O)[C@H](O)[C@H]1O AHHFEZNOXOZZQA-ZEBDFXRSSA-N 0.000 description 1
- 102000003901 Ras GTPase-activating proteins Human genes 0.000 description 1
- 108090000231 Ras GTPase-activating proteins Proteins 0.000 description 1
- 229920000297 Rayon Polymers 0.000 description 1
- 208000006265 Renal cell carcinoma Diseases 0.000 description 1
- 201000000582 Retinoblastoma Diseases 0.000 description 1
- 102100037486 Reverse transcriptase/ribonuclease H Human genes 0.000 description 1
- NCDNCNXCDXHOMX-UHFFFAOYSA-N Ritonavir Natural products C=1C=CC=CC=1CC(NC(=O)OCC=1SC=NC=1)C(O)CC(CC=1C=CC=CC=1)NC(=O)C(C(C)C)NC(=O)N(C)CC1=CSC(C(C)C)=N1 NCDNCNXCDXHOMX-UHFFFAOYSA-N 0.000 description 1
- 241000283984 Rodentia Species 0.000 description 1
- 239000006146 Roswell Park Memorial Institute medium Substances 0.000 description 1
- GCPUVEMWOWMALU-UHFFFAOYSA-N Rubiginone B1 Natural products C1C(C)CC(O)C2=C1C=CC1=C2C(=O)C(C=CC=C2OC)=C2C1=O GCPUVEMWOWMALU-UHFFFAOYSA-N 0.000 description 1
- 108010005173 SERPIN-B5 Proteins 0.000 description 1
- QCHFTSOMWOSFHM-UHFFFAOYSA-N SJ000285536 Natural products C1OC(=O)C(CC)C1CC1=CN=CN1C QCHFTSOMWOSFHM-UHFFFAOYSA-N 0.000 description 1
- 235000014680 Saccharomyces cerevisiae Nutrition 0.000 description 1
- 101100135860 Saccharomyces cerevisiae (strain ATCC 204508 / S288c) PDE2 gene Proteins 0.000 description 1
- 206010039491 Sarcoma Diseases 0.000 description 1
- 241000287219 Serinus canaria Species 0.000 description 1
- 102100030333 Serpin B5 Human genes 0.000 description 1
- BQCADISMDOOEFD-UHFFFAOYSA-N Silver Chemical compound [Ag] BQCADISMDOOEFD-UHFFFAOYSA-N 0.000 description 1
- 229920000519 Sizofiran Polymers 0.000 description 1
- 206010041067 Small cell lung cancer Diseases 0.000 description 1
- OCOKWVBYZHBHLU-UHFFFAOYSA-N Sobuzoxane Chemical compound C1C(=O)N(COC(=O)OCC(C)C)C(=O)CN1CCN1CC(=O)N(COC(=O)OCC(C)C)C(=O)C1 OCOKWVBYZHBHLU-UHFFFAOYSA-N 0.000 description 1
- 239000004141 Sodium laurylsulphate Substances 0.000 description 1
- 244000061456 Solanum tuberosum Species 0.000 description 1
- 235000002595 Solanum tuberosum Nutrition 0.000 description 1
- 206010041349 Somnolence Diseases 0.000 description 1
- NINIDFKCEFEMDL-AKLPVKDBSA-N Sulfur-35 Chemical compound [35S] NINIDFKCEFEMDL-AKLPVKDBSA-N 0.000 description 1
- CYQFCXCEBYINGO-UHFFFAOYSA-N THC Natural products C1=C(C)CCC2C(C)(C)OC3=CC(CCCCC)=CC(O)=C3C21 CYQFCXCEBYINGO-UHFFFAOYSA-N 0.000 description 1
- 229940123582 Telomerase inhibitor Drugs 0.000 description 1
- 208000031320 Teratogenesis Diseases 0.000 description 1
- HNZBNQYXWOLKBA-UHFFFAOYSA-N Tetrahydrofarnesol Natural products CC(C)CCCC(C)CCCC(C)=CCO HNZBNQYXWOLKBA-UHFFFAOYSA-N 0.000 description 1
- WXZSUBHBYQYTNM-UHFFFAOYSA-N Tetrazomine Natural products C1=CC=2CC(N34)C(N5C)C(CO)CC5C4OCC3C=2C(OC)=C1NC(=O)C1NCCCC1O WXZSUBHBYQYTNM-UHFFFAOYSA-N 0.000 description 1
- UPGGKUQISSWRJJ-XLTUSUNSSA-N Thiocoraline Chemical compound O=C([C@H]1CSSC[C@@H](N(C(=O)CNC2=O)C)C(=O)N(C)[C@@H](C(SC[C@@H](C(=O)NCC(=O)N1C)NC(=O)C=1C(=CC3=CC=CC=C3N=1)O)=O)CSC)N(C)[C@H](CSC)C(=O)SC[C@@H]2NC(=O)C1=NC2=CC=CC=C2C=C1O UPGGKUQISSWRJJ-XLTUSUNSSA-N 0.000 description 1
- 102000036693 Thrombopoietin Human genes 0.000 description 1
- 108010041111 Thrombopoietin Proteins 0.000 description 1
- 108010078233 Thymalfasin Proteins 0.000 description 1
- 102400000800 Thymosin alpha-1 Human genes 0.000 description 1
- IVTVGDXNLFLDRM-HNNXBMFYSA-N Tomudex Chemical compound C=1C=C2NC(C)=NC(=O)C2=CC=1CN(C)C1=CC=C(C(=O)N[C@@H](CCC(O)=O)C(O)=O)S1 IVTVGDXNLFLDRM-HNNXBMFYSA-N 0.000 description 1
- 101710183280 Topoisomerase Proteins 0.000 description 1
- 229920001615 Tragacanth Polymers 0.000 description 1
- 239000007983 Tris buffer Substances 0.000 description 1
- YZCKVEUIGOORGS-NJFSPNSNSA-N Tritium Chemical compound [3H] YZCKVEUIGOORGS-NJFSPNSNSA-N 0.000 description 1
- 206010048302 Tubulointerstitial nephritis Diseases 0.000 description 1
- GBOGMAARMMDZGR-UHFFFAOYSA-N UNPD149280 Natural products N1C(=O)C23OC(=O)C=CC(O)CCCC(C)CC=CC3C(O)C(=C)C(C)C2C1CC1=CC=CC=C1 GBOGMAARMMDZGR-UHFFFAOYSA-N 0.000 description 1
- 102100037176 Ubiquitin carboxyl-terminal hydrolase 24 Human genes 0.000 description 1
- 208000025865 Ulcer Diseases 0.000 description 1
- 208000007097 Urinary Bladder Neoplasms Diseases 0.000 description 1
- 102000003990 Urokinase-type plasminogen activator Human genes 0.000 description 1
- 108090000435 Urokinase-type plasminogen activator Proteins 0.000 description 1
- 208000006105 Uterine Cervical Neoplasms Diseases 0.000 description 1
- MALFODICFSIXPO-UHFFFAOYSA-N Veratramin Natural products C=1C=C2C3CC=C4CC(O)CCC4(C)C3CC2=C(C)C=1C(C)C1NCC(C)CC1O MALFODICFSIXPO-UHFFFAOYSA-N 0.000 description 1
- JXLYSJRDGCGARV-WWYNWVTFSA-N Vinblastine Natural products O=C(O[C@H]1[C@](O)(C(=O)OC)[C@@H]2N(C)c3c(cc(c(OC)c3)[C@]3(C(=O)OC)c4[nH]c5c(c4CCN4C[C@](O)(CC)C[C@H](C3)C4)cccc5)[C@@]32[C@H]2[C@@]1(CC)C=CCN2CC3)C JXLYSJRDGCGARV-WWYNWVTFSA-N 0.000 description 1
- 229940122803 Vinca alkaloid Drugs 0.000 description 1
- MHGVSUAAUXQULX-UHFFFAOYSA-N Vinepidine Natural products CCC1CC2CN(CCC3C(=Nc4ccccc34)C(C2)(C(=O)OC)c5cc6c(cc5OC)N(C=O)C7C(O)(C(OC(=O)C)C8(CC)C=CCN9CCC67C89)C(=O)OC)C1 MHGVSUAAUXQULX-UHFFFAOYSA-N 0.000 description 1
- 208000008383 Wilms tumor Diseases 0.000 description 1
- MHDDZDPNIDVLNK-ZGIWMXSJSA-N Zanoterone Chemical compound C1C2=NN(S(C)(=O)=O)C=C2C[C@]2(C)[C@H]3CC[C@](C)([C@](CC4)(O)C#C)[C@@H]4[C@@H]3CC[C@H]21 MHDDZDPNIDVLNK-ZGIWMXSJSA-N 0.000 description 1
- OGQICQVSFDPSEI-UHFFFAOYSA-N Zorac Chemical compound N1=CC(C(=O)OCC)=CC=C1C#CC1=CC=C(SCCC2(C)C)C2=C1 OGQICQVSFDPSEI-UHFFFAOYSA-N 0.000 description 1
- ZZWKZQDOSJAGGF-VRSYWUPDSA-N [(1s,2e,7s,10e,12r,13r,15s)-12-hydroxy-7-methyl-9-oxo-8-oxabicyclo[11.3.0]hexadeca-2,10-dien-15-yl] 2-(dimethylamino)acetate Chemical compound O[C@@H]1\C=C\C(=O)O[C@@H](C)CCC\C=C\[C@@H]2C[C@H](OC(=O)CN(C)C)C[C@H]21 ZZWKZQDOSJAGGF-VRSYWUPDSA-N 0.000 description 1
- VUPBDWQPEOWRQP-RTUCOMKBSA-N [(2R,3S,4S,5R,6R)-2-[(2R,3S,4S,5S,6S)-2-[(1S,2S)-3-[[(2R,3S)-5-[[(2S,3R)-1-[[2-[4-[4-[[4-amino-6-[3-(4-aminobutylamino)propylamino]-6-oxohexyl]carbamoyl]-1,3-thiazol-2-yl]-1,3-thiazol-2-yl]-1-[(2S,3R,4R,5S,6S)-5-amino-3,4-dihydroxy-6-methyloxan-2-yl]oxy-2-hydroxyethyl]amino]-3-hydroxy-1-oxobutan-2-yl]amino]-3-hydroxy-5-oxopentan-2-yl]amino]-2-[[6-amino-2-[(1S)-3-amino-1-[[(2S)-2,3-diamino-3-oxopropyl]amino]-3-oxopropyl]-5-methylpyrimidine-4-carbonyl]amino]-1-(1H-imidazol-5-yl)-3-oxopropoxy]-4,5-dihydroxy-6-(hydroxymethyl)oxan-3-yl]oxy-3,5-dihydroxy-6-(hydroxymethyl)oxan-4-yl] carbamate Chemical compound C[C@@H](O)[C@H](NC(=O)C[C@H](O)[C@@H](C)NC(=O)[C@@H](NC(=O)c1nc(nc(N)c1C)[C@H](CC(N)=O)NC[C@H](N)C(N)=O)[C@H](O[C@@H]1O[C@@H](CO)[C@@H](O)[C@H](O)[C@@H]1O[C@H]1O[C@H](CO)[C@@H](O)[C@H](OC(N)=O)[C@@H]1O)c1cnc[nH]1)C(=O)NC(O[C@@H]1O[C@@H](C)[C@@H](N)[C@@H](O)[C@H]1O)C(O)c1nc(cs1)-c1nc(cs1)C(=O)NCCCC(N)CC(=O)NCCCNCCCCN VUPBDWQPEOWRQP-RTUCOMKBSA-N 0.000 description 1
- ZHHIHQFAUZZMTG-BSVJBJGJSA-N [(2r,3s,4s,5r,6r)-2-[(2r,3s,4s,5s,6s)-2-[(1r,2s)-2-[[6-amino-2-[(1s)-3-amino-1-[[(2s)-2,3-diamino-3-oxopropyl]amino]-3-oxopropyl]-5-methylpyrimidine-4-carbonyl]amino]-3-[[(2r,3s,4s)-3-hydroxy-5-[[(2s,3r)-3-hydroxy-1-oxo-1-[2-[4-[4-[3-[[(1s)-1-phenylethyl] Chemical compound OS(O)(=O)=O.N([C@H](C(=O)N[C@H](C)[C@@H](O)[C@H](C)C(=O)N[C@@H]([C@H](O)C)C(=O)NCCC=1SC=C(N=1)C=1SC=C(N=1)C(=O)NCCCN[C@@H](C)C=1C=CC=CC=1)[C@@H](O[C@H]1[C@H]([C@@H](O)[C@H](O)[C@H](CO)O1)O[C@@H]1[C@H]([C@@H](OC(N)=O)[C@H](O)[C@@H](CO)O1)O)C=1NC=NC=1)C(=O)C1=NC([C@H](CC(N)=O)NC[C@H](N)C(N)=O)=NC(N)=C1C ZHHIHQFAUZZMTG-BSVJBJGJSA-N 0.000 description 1
- SPKNARKFCOPTSY-HYPCCMLGSA-N [(2s,3r)-2-(3-methyloxiran-2-yl)-6-oxo-2,3-dihydropyran-3-yl] acetate Chemical compound CC1OC1[C@@H]1[C@H](OC(C)=O)C=CC(=O)O1 SPKNARKFCOPTSY-HYPCCMLGSA-N 0.000 description 1
- IVCRCPJOLWECJU-XQVQQVTHSA-N [(7r,8r,9s,10r,13s,14s,17s)-7,13-dimethyl-3-oxo-2,6,7,8,9,10,11,12,14,15,16,17-dodecahydro-1h-cyclopenta[a]phenanthren-17-yl] acetate Chemical compound C1C[C@]2(C)[C@@H](OC(C)=O)CC[C@H]2[C@@H]2[C@H](C)CC3=CC(=O)CC[C@@H]3[C@H]21 IVCRCPJOLWECJU-XQVQQVTHSA-N 0.000 description 1
- IFJUINDAXYAPTO-UUBSBJJBSA-N [(8r,9s,13s,14s,17s)-17-[2-[4-[4-[bis(2-chloroethyl)amino]phenyl]butanoyloxy]acetyl]oxy-13-methyl-6,7,8,9,11,12,14,15,16,17-decahydrocyclopenta[a]phenanthren-3-yl] benzoate Chemical compound C([C@@H]1[C@@H](C2=CC=3)CC[C@]4([C@H]1CC[C@@H]4OC(=O)COC(=O)CCCC=1C=CC(=CC=1)N(CCCl)CCCl)C)CC2=CC=3OC(=O)C1=CC=CC=C1 IFJUINDAXYAPTO-UUBSBJJBSA-N 0.000 description 1
- JJULHOZRTCDZOH-JGJFOBQESA-N [1-[[[(2r,3s,4s,5r)-5-(4-amino-2-oxopyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-hydroxyphosphoryl]oxy-3-octadecylsulfanylpropan-2-yl] hexadecanoate Chemical compound O[C@H]1[C@H](O)[C@@H](COP(O)(=O)OP(O)(=O)OCC(CSCCCCCCCCCCCCCCCCCC)OC(=O)CCCCCCCCCCCCCCC)O[C@H]1N1C(=O)N=C(N)C=C1 JJULHOZRTCDZOH-JGJFOBQESA-N 0.000 description 1
- PNDPGZBMCMUPRI-XXSWNUTMSA-N [125I][125I] Chemical compound [125I][125I] PNDPGZBMCMUPRI-XXSWNUTMSA-N 0.000 description 1
- NAFFDQVVNWTDJD-UHFFFAOYSA-L [4-(azanidylmethyl)oxan-4-yl]methylazanide;cyclobutane-1,1-dicarboxylate;platinum(4+) Chemical compound [Pt+4].[NH-]CC1(C[NH-])CCOCC1.[O-]C(=O)C1(C([O-])=O)CCC1 NAFFDQVVNWTDJD-UHFFFAOYSA-L 0.000 description 1
- GDXWHFPKFUYWBE-UHFFFAOYSA-N [F].Cl Chemical compound [F].Cl GDXWHFPKFUYWBE-UHFFFAOYSA-N 0.000 description 1
- HXVHPXMKVOBYTJ-UHFFFAOYSA-N [Gd].N1=CC=CC2=CC=CC=C12 Chemical compound [Gd].N1=CC=CC2=CC=CC=C12 HXVHPXMKVOBYTJ-UHFFFAOYSA-N 0.000 description 1
- BHXPEPFWKYNSKH-UHFFFAOYSA-N [Lu].N1=CC=CC2=CC=CC=C12 Chemical compound [Lu].N1=CC=CC2=CC=CC=C12 BHXPEPFWKYNSKH-UHFFFAOYSA-N 0.000 description 1
- JURAJLFHWXNPHG-UHFFFAOYSA-N [acetyl(methylcarbamoyl)amino] n-methylcarbamate Chemical compound CNC(=O)ON(C(C)=O)C(=O)NC JURAJLFHWXNPHG-UHFFFAOYSA-N 0.000 description 1
- GZOSMCIZMLWJML-VJLLXTKPSA-N abiraterone Chemical compound C([C@H]1[C@H]2[C@@H]([C@]3(CC[C@H](O)CC3=CC2)C)CC[C@@]11C)C=C1C1=CC=CN=C1 GZOSMCIZMLWJML-VJLLXTKPSA-N 0.000 description 1
- 229960000853 abiraterone Drugs 0.000 description 1
- JXLYSJRDGCGARV-KSNABSRWSA-N ac1l29ym Chemical compound C([C@H](C[C@]1(C(=O)OC)C=2C(=CC3=C([C@]45[C@H]([C@@]([C@H](OC(C)=O)[C@]6(CC)C=CCN([C@H]56)CC4)(O)C(=O)OC)N3C)C=2)OC)C[C@](C2)(O)CC)N2CCC2=C1NC1=CC=CC=C21 JXLYSJRDGCGARV-KSNABSRWSA-N 0.000 description 1
- RUGAHXUZHWYHNG-NLGNTGLNSA-N acetic acid;(4r,7s,10s,13r,16s,19r)-10-(4-aminobutyl)-n-[(2s,3r)-1-amino-3-hydroxy-1-oxobutan-2-yl]-19-[[(2r)-2-amino-3-naphthalen-2-ylpropanoyl]amino]-16-[(4-hydroxyphenyl)methyl]-13-(1h-indol-3-ylmethyl)-6,9,12,15,18-pentaoxo-7-propan-2-yl-1,2-dithia-5, Chemical compound CC(O)=O.CC(O)=O.CC(O)=O.CC(O)=O.CC(O)=O.C([C@H]1C(=O)N[C@H](CC=2C3=CC=CC=C3NC=2)C(=O)N[C@@H](CCCCN)C(=O)N[C@H](C(N[C@@H](CSSC[C@@H](C(=O)N1)NC(=O)[C@H](N)CC=1C=C2C=CC=CC2=CC=1)C(=O)N[C@@H]([C@@H](C)O)C(N)=O)=O)C(C)C)C1=CC=C(O)C=C1.C([C@H]1C(=O)N[C@H](CC=2C3=CC=CC=C3NC=2)C(=O)N[C@@H](CCCCN)C(=O)N[C@H](C(N[C@@H](CSSC[C@@H](C(=O)N1)NC(=O)[C@H](N)CC=1C=C2C=CC=CC2=CC=1)C(=O)N[C@@H]([C@@H](C)O)C(N)=O)=O)C(C)C)C1=CC=C(O)C=C1 RUGAHXUZHWYHNG-NLGNTGLNSA-N 0.000 description 1
- QAWIHIJWNYOLBE-OKKQSCSOSA-N acivicin Chemical compound OC(=O)[C@@H](N)[C@@H]1CC(Cl)=NO1 QAWIHIJWNYOLBE-OKKQSCSOSA-N 0.000 description 1
- 229950003478 acodazole Drugs 0.000 description 1
- 208000004064 acoustic neuroma Diseases 0.000 description 1
- 229950000616 acronine Drugs 0.000 description 1
- RJURFGZVJUQBHK-IIXSONLDSA-N actinomycin D Chemical compound C[C@H]1OC(=O)[C@H](C(C)C)N(C)C(=O)CN(C)C(=O)[C@@H]2CCCN2C(=O)[C@@H](C(C)C)NC(=O)[C@H]1NC(=O)C1=C(N)C(=O)C(C)=C2OC(C(C)=CC=C3C(=O)N[C@@H]4C(=O)N[C@@H](C(N5CCC[C@H]5C(=O)N(C)CC(=O)N(C)[C@@H](C(C)C)C(=O)O[C@@H]4C)=O)C(C)C)=C3N=C21 RJURFGZVJUQBHK-IIXSONLDSA-N 0.000 description 1
- 230000004913 activation Effects 0.000 description 1
- 239000004480 active ingredient Substances 0.000 description 1
- 239000008186 active pharmaceutical agent Substances 0.000 description 1
- 125000002252 acyl group Chemical group 0.000 description 1
- 208000009956 adenocarcinoma Diseases 0.000 description 1
- 102000019997 adhesion receptor Human genes 0.000 description 1
- 108010013985 adhesion receptor Proteins 0.000 description 1
- 239000003470 adrenal cortex hormone Substances 0.000 description 1
- 230000001476 alcoholic effect Effects 0.000 description 1
- 229940060516 alferon n Drugs 0.000 description 1
- 239000003513 alkali Substances 0.000 description 1
- 229930013930 alkaloid Natural products 0.000 description 1
- 125000003545 alkoxy group Chemical group 0.000 description 1
- 125000005257 alkyl acyl group Chemical group 0.000 description 1
- 150000003973 alkyl amines Chemical class 0.000 description 1
- 125000005907 alkyl ester group Chemical group 0.000 description 1
- 125000005360 alkyl sulfoxide group Chemical group 0.000 description 1
- 229940100198 alkylating agent Drugs 0.000 description 1
- 239000002168 alkylating agent Substances 0.000 description 1
- BOTWFXYSPFMFNR-OALUTQOASA-N all-rac-phytol Natural products CC(C)CCC[C@H](C)CCC[C@H](C)CCCC(C)=CCO BOTWFXYSPFMFNR-OALUTQOASA-N 0.000 description 1
- SHGAZHPCJJPHSC-YCNIQYBTSA-N all-trans-retinoic acid Chemical compound OC(=O)\C=C(/C)\C=C\C=C(/C)\C=C\C1=C(C)CCCC1(C)C SHGAZHPCJJPHSC-YCNIQYBTSA-N 0.000 description 1
- 150000001370 alpha-amino acid derivatives Chemical class 0.000 description 1
- 235000008206 alpha-amino acids Nutrition 0.000 description 1
- 239000004411 aluminium Substances 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- 239000005030 aluminium foil Substances 0.000 description 1
- 235000012211 aluminium silicate Nutrition 0.000 description 1
- 229950010949 ambamustine Drugs 0.000 description 1
- 229950011363 ametantrone Drugs 0.000 description 1
- JKOQGQFVAUAYPM-UHFFFAOYSA-N amifostine Chemical compound NCCCNCCSP(O)(O)=O JKOQGQFVAUAYPM-UHFFFAOYSA-N 0.000 description 1
- 229960001097 amifostine Drugs 0.000 description 1
- 229960003437 aminoglutethimide Drugs 0.000 description 1
- ROBVIMPUHSLWNV-UHFFFAOYSA-N aminoglutethimide Chemical compound C=1C=C(N)C=CC=1C1(CC)CCC(=O)NC1=O ROBVIMPUHSLWNV-UHFFFAOYSA-N 0.000 description 1
- 229960002550 amrubicin Drugs 0.000 description 1
- VJZITPJGSQKZMX-XDPRQOKASA-N amrubicin Chemical compound O([C@H]1C[C@](CC2=C(O)C=3C(=O)C4=CC=CC=C4C(=O)C=3C(O)=C21)(N)C(=O)C)[C@H]1C[C@H](O)[C@H](O)CO1 VJZITPJGSQKZMX-XDPRQOKASA-N 0.000 description 1
- 229960001694 anagrelide Drugs 0.000 description 1
- OTBXOEAOVRKTNQ-UHFFFAOYSA-N anagrelide Chemical compound N1=C2NC(=O)CN2CC2=C(Cl)C(Cl)=CC=C21 OTBXOEAOVRKTNQ-UHFFFAOYSA-N 0.000 description 1
- 239000004037 angiogenesis inhibitor Substances 0.000 description 1
- 229940121369 angiogenesis inhibitor Drugs 0.000 description 1
- 125000002490 anilino group Chemical group [H]N(*)C1=C([H])C([H])=C([H])C([H])=C1[H] 0.000 description 1
- 239000006053 animal diet Substances 0.000 description 1
- 238000005571 anion exchange chromatography Methods 0.000 description 1
- 230000008485 antagonism Effects 0.000 description 1
- 108010070670 antarelix Proteins 0.000 description 1
- VGQOVCHZGQWAOI-YQRHFANHSA-N anthramycin Chemical compound N1[C@H](O)[C@@H]2CC(\C=C\C(N)=O)=CN2C(=O)C2=CC=C(C)C(O)=C12 VGQOVCHZGQWAOI-YQRHFANHSA-N 0.000 description 1
- PYKYMHQGRFAEBM-UHFFFAOYSA-N anthraquinone Natural products CCC(=O)c1c(O)c2C(=O)C3C(C=CC=C3O)C(=O)c2cc1CC(=O)OC PYKYMHQGRFAEBM-UHFFFAOYSA-N 0.000 description 1
- 150000004056 anthraquinones Chemical class 0.000 description 1
- 238000011861 anti-inflammatory therapy Methods 0.000 description 1
- 239000000051 antiandrogen Substances 0.000 description 1
- 102000025171 antigen binding proteins Human genes 0.000 description 1
- 108091000831 antigen binding proteins Proteins 0.000 description 1
- 239000002246 antineoplastic agent Substances 0.000 description 1
- 230000006502 antiplatelets effects Effects 0.000 description 1
- 239000000074 antisense oligonucleotide Substances 0.000 description 1
- 238000012230 antisense oligonucleotides Methods 0.000 description 1
- 229950006345 antramycin Drugs 0.000 description 1
- NOFOAYPPHIUXJR-APNQCZIXSA-N aphidicolin Chemical compound C1[C@@]23[C@@]4(C)CC[C@@H](O)[C@@](C)(CO)[C@@H]4CC[C@H]3C[C@H]1[C@](CO)(O)CC2 NOFOAYPPHIUXJR-APNQCZIXSA-N 0.000 description 1
- 230000006907 apoptotic process Effects 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- YZXBAPSDXZZRGB-DOFZRALJSA-N arachidonic acid Chemical group CCCCC\C=C/C\C=C/C\C=C/C\C=C/CCCC(O)=O YZXBAPSDXZZRGB-DOFZRALJSA-N 0.000 description 1
- 108010055530 arginyl-tryptophyl-N-methylphenylalanyl-tryptophyl-leucyl-methioninamide Proteins 0.000 description 1
- 229960003272 asparaginase Drugs 0.000 description 1
- DCXYFEDJOCDNAF-UHFFFAOYSA-M asparaginate Chemical compound [O-]C(=O)C(N)CC(N)=O DCXYFEDJOCDNAF-UHFFFAOYSA-M 0.000 description 1
- 239000000305 astragalus gummifer gum Substances 0.000 description 1
- 229950006933 atrimustine Drugs 0.000 description 1
- 108010093161 axinastatin 1 Proteins 0.000 description 1
- PICZCWCKOLHDOJ-GHTSNYPWSA-N axinastatin 1 Chemical compound C([C@H]1C(=O)N[C@H](C(=O)N[C@H](C(=O)N2CCC[C@H]2C(=O)N[C@H](C(N[C@@H](CC(N)=O)C(=O)N2CCC[C@H]2C(=O)N1)=O)C(C)C)C(C)C)C(C)C)C1=CC=CC=C1 PICZCWCKOLHDOJ-GHTSNYPWSA-N 0.000 description 1
- 108010093000 axinastatin 2 Proteins 0.000 description 1
- OXNAATCTZCSVKR-AVGVIDKOSA-N axinastatin 2 Chemical compound C([C@H]1C(=O)N[C@H](C(=O)N[C@H](C(N2CCC[C@H]2C(=O)N[C@@H](C(=O)N[C@@H](CC(N)=O)C(=O)N2CCC[C@H]2C(=O)N1)C(C)C)=O)CC(C)C)C(C)C)C1=CC=CC=C1 OXNAATCTZCSVKR-AVGVIDKOSA-N 0.000 description 1
- UZCPCRPHNVHKKP-UHFFFAOYSA-N axinastatin 2 Natural products CC(C)CC1NC(=O)C2CCCN2C(=O)C(NC(=O)C(CC(=O)N)NC(=O)C3CCCN3C(=O)C(Cc4ccccc4)NC(=O)C(NC1=O)C(C)C)C(C)C UZCPCRPHNVHKKP-UHFFFAOYSA-N 0.000 description 1
- 108010092978 axinastatin 3 Proteins 0.000 description 1
- ANLDPEXRVVIABH-WUUSPZRJSA-N axinastatin 3 Chemical compound C([C@H]1C(=O)N[C@H](C(N[C@@H](CC(C)C)C(=O)N2CCC[C@H]2C(=O)N[C@@H](C(=O)N[C@@H](CC(N)=O)C(=O)N2CCC[C@H]2C(=O)N1)C(C)C)=O)[C@@H](C)CC)C1=CC=CC=C1 ANLDPEXRVVIABH-WUUSPZRJSA-N 0.000 description 1
- RTGMQVUKARGBNM-UHFFFAOYSA-N axinastatin 3 Natural products CCC(C)C1NC(=O)C(CC(C)C)NC(=O)C2CCCN2C(=O)C(NC(=O)C(CC(=O)N)NC(=O)C3CCCN3C(=O)C(Cc4ccccc4)NC1=O)C(C)C RTGMQVUKARGBNM-UHFFFAOYSA-N 0.000 description 1
- 229960002756 azacitidine Drugs 0.000 description 1
- VUSKAKFAMMEXDX-WBUYAQKGSA-N azalomycin Chemical compound O([C@@H]1C[C@@]([C@H]([C@H](C)[C@]23[C@H](C)CC[C@]1(C)[C@@H]2C(=O)CC3)O)(C)CC)C(=O)CSC1=NNC(N)=N1 VUSKAKFAMMEXDX-WBUYAQKGSA-N 0.000 description 1
- 229950009711 azalomycin Drugs 0.000 description 1
- 229930191236 azalomycin Natural products 0.000 description 1
- OPWOOOGFNULJAQ-UHFFFAOYSA-L azane;cyclopentanamine;2-hydroxybutanedioate;platinum(2+) Chemical compound N.[Pt+2].NC1CCCC1.[O-]C(=O)C(O)CC([O-])=O OPWOOOGFNULJAQ-UHFFFAOYSA-L 0.000 description 1
- VSRXQHXAPYXROS-UHFFFAOYSA-N azanide;cyclobutane-1,1-dicarboxylic acid;platinum(2+) Chemical compound [NH2-].[NH2-].[Pt+2].OC(=O)C1(C(O)=O)CCC1 VSRXQHXAPYXROS-UHFFFAOYSA-N 0.000 description 1
- 229950005951 azasetron Drugs 0.000 description 1
- HRXVDDOKERXBEY-UHFFFAOYSA-N azatepa Chemical compound C1CN1P(=O)(N1CC1)N(CC)C1=NN=CS1 HRXVDDOKERXBEY-UHFFFAOYSA-N 0.000 description 1
- 229950002182 azatepa Drugs 0.000 description 1
- 229950004295 azotomycin Drugs 0.000 description 1
- 150000004200 baccatin III derivatives Chemical class 0.000 description 1
- XYUFCXJZFZPEJD-PGRDOPGGSA-N balanol Chemical compound OC(=O)C1=CC=CC(O)=C1C(=O)C1=C(O)C=C(C(=O)O[C@H]2[C@H](CNCCC2)NC(=O)C=2C=CC(O)=CC=2)C=C1O XYUFCXJZFZPEJD-PGRDOPGGSA-N 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 229950005567 benzodepa Drugs 0.000 description 1
- MMIMIFULGMZVPO-UHFFFAOYSA-N benzyl 3-bromo-2,6-dinitro-5-phenylmethoxybenzoate Chemical compound [O-][N+](=O)C1=C(C(=O)OCC=2C=CC=CC=2)C([N+](=O)[O-])=C(Br)C=C1OCC1=CC=CC=C1 MMIMIFULGMZVPO-UHFFFAOYSA-N 0.000 description 1
- VFIUCBTYGKMLCM-UHFFFAOYSA-N benzyl n-[bis(aziridin-1-yl)phosphoryl]carbamate Chemical compound C=1C=CC=CC=1COC(=O)NP(=O)(N1CC1)N1CC1 VFIUCBTYGKMLCM-UHFFFAOYSA-N 0.000 description 1
- QGJZLNKBHJESQX-FZFNOLFKSA-N betulinic acid Chemical compound C1C[C@H](O)C(C)(C)[C@@H]2CC[C@@]3(C)[C@]4(C)CC[C@@]5(C(O)=O)CC[C@@H](C(=C)C)[C@@H]5[C@H]4CC[C@@H]3[C@]21C QGJZLNKBHJESQX-FZFNOLFKSA-N 0.000 description 1
- 108091008324 binding proteins Proteins 0.000 description 1
- 230000003570 biosynthesizing effect Effects 0.000 description 1
- 229950002370 bisnafide Drugs 0.000 description 1
- 230000000740 bleeding effect Effects 0.000 description 1
- 230000023555 blood coagulation Effects 0.000 description 1
- 230000036765 blood level Effects 0.000 description 1
- 210000001772 blood platelet Anatomy 0.000 description 1
- 238000010241 blood sampling Methods 0.000 description 1
- 210000004204 blood vessel Anatomy 0.000 description 1
- 230000037396 body weight Effects 0.000 description 1
- 238000009835 boiling Methods 0.000 description 1
- PZOHOALJQOFNTB-UHFFFAOYSA-M brequinar sodium Chemical compound [Na+].N1=C2C=CC(F)=CC2=C(C([O-])=O)C(C)=C1C(C=C1)=CC=C1C1=CC=CC=C1F PZOHOALJQOFNTB-UHFFFAOYSA-M 0.000 description 1
- 208000003362 bronchogenic carcinoma Diseases 0.000 description 1
- 229950002361 budotitane Drugs 0.000 description 1
- 102100029175 cGMP-specific 3',5'-cyclic phosphodiesterase Human genes 0.000 description 1
- 229960002882 calcipotriol Drugs 0.000 description 1
- LWQQLNNNIPYSNX-UROSTWAQSA-N calcipotriol Chemical compound C1([C@H](O)/C=C/[C@@H](C)[C@@H]2[C@]3(CCCC(/[C@@H]3CC2)=C\C=C\2C([C@@H](O)C[C@H](O)C/2)=C)C)CC1 LWQQLNNNIPYSNX-UROSTWAQSA-N 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- CJZGTCYPCWQAJB-UHFFFAOYSA-L calcium stearate Chemical compound [Ca+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O CJZGTCYPCWQAJB-UHFFFAOYSA-L 0.000 description 1
- 235000013539 calcium stearate Nutrition 0.000 description 1
- 239000008116 calcium stearate Substances 0.000 description 1
- 238000004364 calculation method Methods 0.000 description 1
- 244000309466 calf Species 0.000 description 1
- LSUTUUOITDQYNO-UHFFFAOYSA-N calphostin C Chemical compound C=12C3=C4C(CC(C)OC(=O)C=5C=CC=CC=5)=C(OC)C(O)=C(C(C=C5OC)=O)C4=C5C=1C(OC)=CC(=O)C2=C(O)C(OC)=C3CC(C)OC(=O)OC1=CC=C(O)C=C1 LSUTUUOITDQYNO-UHFFFAOYSA-N 0.000 description 1
- IVFYLRMMHVYGJH-PVPPCFLZSA-N calusterone Chemical compound C1C[C@]2(C)[C@](O)(C)CC[C@H]2[C@@H]2[C@@H](C)CC3=CC(=O)CC[C@]3(C)[C@H]21 IVFYLRMMHVYGJH-PVPPCFLZSA-N 0.000 description 1
- 229950009823 calusterone Drugs 0.000 description 1
- VSJKWCGYPAHWDS-FQEVSTJZSA-N camptothecin Chemical class C1=CC=C2C=C(CN3C4=CC5=C(C3=O)COC(=O)[C@]5(O)CC)C4=NC2=C1 VSJKWCGYPAHWDS-FQEVSTJZSA-N 0.000 description 1
- 239000012830 cancer therapeutic Substances 0.000 description 1
- 229940022399 cancer vaccine Drugs 0.000 description 1
- 230000003327 cancerostatic effect Effects 0.000 description 1
- 229960004117 capecitabine Drugs 0.000 description 1
- 229950009338 caracemide Drugs 0.000 description 1
- 239000004202 carbamide Substances 0.000 description 1
- 229950005155 carbetimer Drugs 0.000 description 1
- 235000011089 carbon dioxide Nutrition 0.000 description 1
- 238000001460 carbon-13 nuclear magnetic resonance spectrum Methods 0.000 description 1
- 229960004562 carboplatin Drugs 0.000 description 1
- WNRZHQBJSXRYJK-UHFFFAOYSA-N carboxyamidotriazole Chemical compound NC1=C(C(=O)N)N=NN1CC(C=C1Cl)=CC(Cl)=C1C(=O)C1=CC=C(Cl)C=C1 WNRZHQBJSXRYJK-UHFFFAOYSA-N 0.000 description 1
- 230000000747 cardiac effect Effects 0.000 description 1
- 238000012754 cardiac puncture Methods 0.000 description 1
- 230000003130 cardiopathic effect Effects 0.000 description 1
- XREUEWVEMYWFFA-CSKJXFQVSA-N carminomycin Chemical compound C1[C@H](N)[C@H](O)[C@H](C)O[C@H]1O[C@@H]1C2=C(O)C(C(=O)C3=C(O)C=CC=C3C3=O)=C3C(O)=C2C[C@@](O)(C(C)=O)C1 XREUEWVEMYWFFA-CSKJXFQVSA-N 0.000 description 1
- 229960005243 carmustine Drugs 0.000 description 1
- 210000000845 cartilage Anatomy 0.000 description 1
- 239000005018 casein Substances 0.000 description 1
- BECPQYXYKAMYBN-UHFFFAOYSA-N casein, tech. Chemical compound NCCCCC(C(O)=O)N=C(O)C(CC(O)=O)N=C(O)C(CCC(O)=N)N=C(O)C(CC(C)C)N=C(O)C(CCC(O)=O)N=C(O)C(CC(O)=O)N=C(O)C(CCC(O)=O)N=C(O)C(C(C)O)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=O)N=C(O)C(CCC(O)=O)N=C(O)C(COP(O)(O)=O)N=C(O)C(CCC(O)=N)N=C(O)C(N)CC1=CC=CC=C1 BECPQYXYKAMYBN-UHFFFAOYSA-N 0.000 description 1
- 235000021240 caseins Nutrition 0.000 description 1
- 230000020411 cell activation Effects 0.000 description 1
- 230000021164 cell adhesion Effects 0.000 description 1
- 230000007248 cellular mechanism Effects 0.000 description 1
- 230000036755 cellular response Effects 0.000 description 1
- 239000001913 cellulose Substances 0.000 description 1
- 201000010881 cervical cancer Diseases 0.000 description 1
- 229960004485 cetiedil citrate Drugs 0.000 description 1
- SBNPWPIBESPSIF-MHWMIDJBSA-N cetrorelix Chemical compound C([C@@H](C(=O)N[C@H](CCCNC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N1[C@@H](CCC1)C(=O)N[C@H](C)C(N)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](CC=1C=NC=CC=1)NC(=O)[C@@H](CC=1C=CC(Cl)=CC=1)NC(=O)[C@@H](CC=1C=C2C=CC=CC2=CC=1)NC(C)=O)C1=CC=C(O)C=C1 SBNPWPIBESPSIF-MHWMIDJBSA-N 0.000 description 1
- 108700008462 cetrorelix Proteins 0.000 description 1
- 229960003230 cetrorelix Drugs 0.000 description 1
- HZCWPKGYTCJSEB-UHFFFAOYSA-N chembl118841 Chemical compound C12=CC(OC)=CC=C2NC2=C([N+]([O-])=O)C=CC3=C2C1=NN3CCCN(C)C HZCWPKGYTCJSEB-UHFFFAOYSA-N 0.000 description 1
- NQGMIPUYCWIEAW-OVCLIPMQSA-N chembl1834105 Chemical compound O/N=C/C1=C(SC)C(OC)=CC(C=2N=CC=CC=2)=N1 NQGMIPUYCWIEAW-OVCLIPMQSA-N 0.000 description 1
- UKTAZPQNNNJVKR-KJGYPYNMSA-N chembl2368925 Chemical compound C1=CC=C2C(C(O[C@@H]3C[C@@H]4C[C@H]5C[C@@H](N4CC5=O)C3)=O)=CNC2=C1 UKTAZPQNNNJVKR-KJGYPYNMSA-N 0.000 description 1
- DCKFXSZUWVWFEU-JECTWPLRSA-N chembl499423 Chemical compound O1[C@@H](CC)CCCC[C@]11NC(N23)=N[C@]4(O[C@H](C)CCC4)[C@@H](C(=O)OCCCCCCCCCCCCCCCC(=O)N(CCCN)C[C@@H](O)CCN)[C@@]3(O)CC[C@H]2C1 DCKFXSZUWVWFEU-JECTWPLRSA-N 0.000 description 1
- 239000002975 chemoattractant Substances 0.000 description 1
- 150000004035 chlorins Chemical class 0.000 description 1
- 208000006990 cholangiocarcinoma Diseases 0.000 description 1
- 229940048961 cholinesterase Drugs 0.000 description 1
- 230000006020 chronic inflammation Effects 0.000 description 1
- 231100000850 chronic interstitial nephritis Toxicity 0.000 description 1
- 208000024207 chronic leukemia Diseases 0.000 description 1
- 208000032852 chronic lymphocytic leukemia Diseases 0.000 description 1
- LZFOPEXOUVTGJS-UHFFFAOYSA-N cis-sinapyl alcohol Natural products COC1=CC(C=CCO)=CC(OC)=C1O LZFOPEXOUVTGJS-UHFFFAOYSA-N 0.000 description 1
- JKNIRLKHOOMGOJ-UHFFFAOYSA-N cladochrome D Natural products COC1=C(CC(C)OC(=O)Oc2ccc(O)cc2)c3c4C(=C(OC)C(=O)c5c(O)cc(OC)c(c45)c6c(OC)cc(O)c(C1=O)c36)CC(C)OC(=O)c7ccc(O)cc7 JKNIRLKHOOMGOJ-UHFFFAOYSA-N 0.000 description 1
- SRJYZPCBWDVSGO-UHFFFAOYSA-N cladochrome E Natural products COC1=CC(O)=C(C(C(OC)=C(CC(C)OC(=O)OC=2C=CC(O)=CC=2)C2=3)=O)C2=C1C1=C(OC)C=C(O)C(C(C=2OC)=O)=C1C=3C=2CC(C)OC(=O)C1=CC=CC=C1 SRJYZPCBWDVSGO-UHFFFAOYSA-N 0.000 description 1
- AGOYDEPGAOXOCK-KCBOHYOISA-N clarithromycin Chemical compound O([C@@H]1[C@@H](C)C(=O)O[C@@H]([C@@]([C@H](O)[C@@H](C)C(=O)[C@H](C)C[C@](C)([C@H](O[C@H]2[C@@H]([C@H](C[C@@H](C)O2)N(C)C)O)[C@H]1C)OC)(C)O)CC)[C@H]1C[C@@](C)(OC)[C@@H](O)[C@H](C)O1 AGOYDEPGAOXOCK-KCBOHYOISA-N 0.000 description 1
- 229960002626 clarithromycin Drugs 0.000 description 1
- 239000004927 clay Substances 0.000 description 1
- GKIRPKYJQBWNGO-OCEACIFDSA-N clomifene Chemical class C1=CC(OCCN(CC)CC)=CC=C1C(\C=1C=CC=CC=1)=C(\Cl)C1=CC=CC=C1 GKIRPKYJQBWNGO-OCEACIFDSA-N 0.000 description 1
- VNFPBHJOKIVQEB-UHFFFAOYSA-N clotrimazole Chemical compound ClC1=CC=CC=C1C(N1C=NC=C1)(C=1C=CC=CC=1)C1=CC=CC=C1 VNFPBHJOKIVQEB-UHFFFAOYSA-N 0.000 description 1
- 229960004022 clotrimazole Drugs 0.000 description 1
- 229930193484 collismycin Natural products 0.000 description 1
- 208000029742 colonic neoplasm Diseases 0.000 description 1
- 201000010989 colorectal carcinoma Diseases 0.000 description 1
- 210000002777 columnar cell Anatomy 0.000 description 1
- 229960005537 combretastatin A-4 Drugs 0.000 description 1
- HVXBOLULGPECHP-UHFFFAOYSA-N combretastatin A4 Natural products C1=C(O)C(OC)=CC=C1C=CC1=CC(OC)=C(OC)C(OC)=C1 HVXBOLULGPECHP-UHFFFAOYSA-N 0.000 description 1
- 150000004814 combretastatins Chemical class 0.000 description 1
- 230000000052 comparative effect Effects 0.000 description 1
- 230000002860 competitive effect Effects 0.000 description 1
- GLESHRYLRAOJPS-DHCFDGJBSA-N conagenin Chemical compound C[C@@H](O)[C@H](C)[C@@H](O)C(=O)N[C@@](C)(CO)C(O)=O GLESHRYLRAOJPS-DHCFDGJBSA-N 0.000 description 1
- 239000012141 concentrate Substances 0.000 description 1
- 235000008504 concentrate Nutrition 0.000 description 1
- 230000001143 conditioned effect Effects 0.000 description 1
- 235000009508 confectionery Nutrition 0.000 description 1
- OMFXVFTZEKFJBZ-HJTSIMOOSA-N corticosterone Chemical compound O=C1CC[C@]2(C)[C@H]3[C@@H](O)C[C@](C)([C@H](CC4)C(=O)CO)[C@@H]4[C@@H]3CCC2=C1 OMFXVFTZEKFJBZ-HJTSIMOOSA-N 0.000 description 1
- COFJBSXICYYSKG-OAUVCNBTSA-N cph2u7dndy Chemical compound OS(O)(=O)=O.C([C@H](C[C@]1(C(=O)OC)C=2C(=CC3=C([C@]45[C@H]([C@@]([C@H](O)[C@]6(CC)C=CCN([C@H]56)CC4)(O)C(N)=O)N3C)C=2)OC)C[C@@](C2)(O)CC)N2CCC2=C1NC1=CC=CC=C21 COFJBSXICYYSKG-OAUVCNBTSA-N 0.000 description 1
- 239000012043 crude product Substances 0.000 description 1
- XCIXKGXIYUWCLL-UHFFFAOYSA-N cyclopentanol Chemical compound OC1CCCC1 XCIXKGXIYUWCLL-UHFFFAOYSA-N 0.000 description 1
- 108010041566 cypemycin Proteins 0.000 description 1
- 208000002445 cystadenocarcinoma Diseases 0.000 description 1
- GBOGMAARMMDZGR-JREHFAHYSA-N cytochalasin B Natural products C[C@H]1CCC[C@@H](O)C=CC(=O)O[C@@]23[C@H](C=CC1)[C@H](O)C(=C)[C@@H](C)[C@@H]2[C@H](Cc4ccccc4)NC3=O GBOGMAARMMDZGR-JREHFAHYSA-N 0.000 description 1
- GBOGMAARMMDZGR-TYHYBEHESA-N cytochalasin B Chemical compound C([C@H]1[C@@H]2[C@@H](C([C@@H](O)[C@@H]3/C=C/C[C@H](C)CCC[C@@H](O)/C=C/C(=O)O[C@@]23C(=O)N1)=C)C)C1=CC=CC=C1 GBOGMAARMMDZGR-TYHYBEHESA-N 0.000 description 1
- 230000009089 cytolysis Effects 0.000 description 1
- 229960000640 dactinomycin Drugs 0.000 description 1
- 238000013480 data collection Methods 0.000 description 1
- STQGQHZAVUOBTE-VGBVRHCVSA-N daunorubicin Chemical compound O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(C)=O)[C@H]1C[C@H](N)[C@H](O)[C@H](C)O1 STQGQHZAVUOBTE-VGBVRHCVSA-N 0.000 description 1
- 229960003109 daunorubicin hydrochloride Drugs 0.000 description 1
- 238000006356 dehydrogenation reaction Methods 0.000 description 1
- 125000000422 delta-lactone group Chemical group 0.000 description 1
- CYQFCXCEBYINGO-IAGOWNOFSA-N delta1-THC Chemical compound C1=C(C)CC[C@H]2C(C)(C)OC3=CC(CCCCC)=CC(O)=C3[C@@H]21 CYQFCXCEBYINGO-IAGOWNOFSA-N 0.000 description 1
- 238000000432 density-gradient centrifugation Methods 0.000 description 1
- 230000001419 dependent effect Effects 0.000 description 1
- 238000013461 design Methods 0.000 description 1
- 108700025485 deslorelin Proteins 0.000 description 1
- 229960005408 deslorelin Drugs 0.000 description 1
- 230000001066 destructive effect Effects 0.000 description 1
- 229960003957 dexamethasone Drugs 0.000 description 1
- UREBDLICKHMUKA-CXSFZGCWSA-N dexamethasone Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@]2(F)[C@@H]1[C@@H]1C[C@@H](C)[C@@](C(=O)CO)(O)[C@@]1(C)C[C@@H]2O UREBDLICKHMUKA-CXSFZGCWSA-N 0.000 description 1
- 229960000605 dexrazoxane Drugs 0.000 description 1
- SGTNSNPWRIOYBX-HHHXNRCGSA-N dexverapamil Chemical compound C1=C(OC)C(OC)=CC=C1CCN(C)CCC[C@@](C#N)(C(C)C)C1=CC=C(OC)C(OC)=C1 SGTNSNPWRIOYBX-HHHXNRCGSA-N 0.000 description 1
- 229950005878 dexverapamil Drugs 0.000 description 1
- 238000003745 diagnosis Methods 0.000 description 1
- 229960001259 diclofenac Drugs 0.000 description 1
- DCOPUUMXTXDBNB-UHFFFAOYSA-N diclofenac Chemical compound OC(=O)CC1=CC=CC=C1NC1=C(Cl)C=CC=C1Cl DCOPUUMXTXDBNB-UHFFFAOYSA-N 0.000 description 1
- GJQPMPFPNINLKP-UHFFFAOYSA-N diclofenamide Chemical compound NS(=O)(=O)C1=CC(Cl)=C(Cl)C(S(N)(=O)=O)=C1 GJQPMPFPNINLKP-UHFFFAOYSA-N 0.000 description 1
- 229960005081 diclofenamide Drugs 0.000 description 1
- 229930189582 didemnin Natural products 0.000 description 1
- HPNMFZURTQLUMO-UHFFFAOYSA-O diethylammonium Chemical compound CC[NH2+]CC HPNMFZURTQLUMO-UHFFFAOYSA-O 0.000 description 1
- PZXJOHSZQAEJFE-UHFFFAOYSA-N dihydrobetulinic acid Natural products C1CC(O)C(C)(C)C2CCC3(C)C4(C)CCC5(C(O)=O)CCC(C(C)C)C5C4CCC3C21C PZXJOHSZQAEJFE-UHFFFAOYSA-N 0.000 description 1
- 229910001873 dinitrogen Inorganic materials 0.000 description 1
- 239000001177 diphosphate Substances 0.000 description 1
- XPPKVPWEQAFLFU-UHFFFAOYSA-J diphosphate(4-) Chemical compound [O-]P([O-])(=O)OP([O-])([O-])=O XPPKVPWEQAFLFU-UHFFFAOYSA-J 0.000 description 1
- 235000011180 diphosphates Nutrition 0.000 description 1
- 230000006806 disease prevention Effects 0.000 description 1
- CZLKTMHQYXYHOO-QTNFYWBSSA-L disodium;(2s)-2-[(2-phosphonatoacetyl)amino]butanedioic acid Chemical compound [Na+].[Na+].OC(=O)C[C@@H](C(O)=O)NC(=O)CP([O-])([O-])=O CZLKTMHQYXYHOO-QTNFYWBSSA-L 0.000 description 1
- SVJSWELRJWVPQD-KJWOGLQMSA-L disodium;(2s)-2-[[4-[2-[(6r)-2-amino-4-oxo-5,6,7,8-tetrahydro-1h-pyrido[2,3-d]pyrimidin-6-yl]ethyl]benzoyl]amino]pentanedioate Chemical compound [Na+].[Na+].C([C@@H]1CC=2C(=O)N=C(NC=2NC1)N)CC1=CC=C(C(=O)N[C@@H](CCC([O-])=O)C([O-])=O)C=C1 SVJSWELRJWVPQD-KJWOGLQMSA-L 0.000 description 1
- 239000006185 dispersion Substances 0.000 description 1
- 238000006073 displacement reaction Methods 0.000 description 1
- 238000004821 distillation Methods 0.000 description 1
- 208000002173 dizziness Diseases 0.000 description 1
- POULHZVOKOAJMA-UHFFFAOYSA-N dodecanoic acid Chemical compound CCCCCCCCCCCC(O)=O POULHZVOKOAJMA-UHFFFAOYSA-N 0.000 description 1
- 229960003413 dolasetron Drugs 0.000 description 1
- ZWAOHEXOSAUJHY-ZIYNGMLESA-N doxifluridine Chemical compound O[C@@H]1[C@H](O)[C@@H](C)O[C@H]1N1C(=O)NC(=O)C(F)=C1 ZWAOHEXOSAUJHY-ZIYNGMLESA-N 0.000 description 1
- 229950005454 doxifluridine Drugs 0.000 description 1
- 229960002918 doxorubicin hydrochloride Drugs 0.000 description 1
- NOTIQUSPUUHHEH-UXOVVSIBSA-N dromostanolone propionate Chemical compound C([C@@H]1CC2)C(=O)[C@H](C)C[C@]1(C)[C@@H]1[C@@H]2[C@@H]2CC[C@H](OC(=O)CC)[C@@]2(C)CC1 NOTIQUSPUUHHEH-UXOVVSIBSA-N 0.000 description 1
- 229960004242 dronabinol Drugs 0.000 description 1
- 239000003937 drug carrier Substances 0.000 description 1
- 238000007876 drug discovery Methods 0.000 description 1
- 230000008406 drug-drug interaction Effects 0.000 description 1
- 230000009977 dual effect Effects 0.000 description 1
- 229960005501 duocarmycin Drugs 0.000 description 1
- VQNATVDKACXKTF-XELLLNAOSA-N duocarmycin Chemical compound COC1=C(OC)C(OC)=C2NC(C(=O)N3C4=CC(=O)C5=C([C@@]64C[C@@H]6C3)C=C(N5)C(=O)OC)=CC2=C1 VQNATVDKACXKTF-XELLLNAOSA-N 0.000 description 1
- 229930184221 duocarmycin Natural products 0.000 description 1
- 230000008482 dysregulation Effects 0.000 description 1
- 229950010033 ebselen Drugs 0.000 description 1
- FSIRXIHZBIXHKT-MHTVFEQDSA-N edatrexate Chemical compound C=1N=C2N=C(N)N=C(N)C2=NC=1CC(CC)C1=CC=C(C(=O)N[C@@H](CCC(O)=O)C(O)=O)C=C1 FSIRXIHZBIXHKT-MHTVFEQDSA-N 0.000 description 1
- 229950006700 edatrexate Drugs 0.000 description 1
- 229960001776 edrecolomab Drugs 0.000 description 1
- 229960002759 eflornithine Drugs 0.000 description 1
- 238000002003 electron diffraction Methods 0.000 description 1
- MGQRRMONVLMKJL-KWJIQSIXSA-N elsamitrucin Chemical compound O1[C@H](C)[C@H](O)[C@H](OC)[C@@H](N)[C@H]1O[C@@H]1[C@](O)(C)[C@@H](O)[C@@H](C)O[C@H]1OC1=CC=CC2=C(O)C(C(O3)=O)=C4C5=C3C=CC(C)=C5C(=O)OC4=C12 MGQRRMONVLMKJL-KWJIQSIXSA-N 0.000 description 1
- 229950002339 elsamitrucin Drugs 0.000 description 1
- 229950005450 emitefur Drugs 0.000 description 1
- 239000003995 emulsifying agent Substances 0.000 description 1
- 210000002889 endothelial cell Anatomy 0.000 description 1
- 230000003511 endothelial effect Effects 0.000 description 1
- 229950010625 enloplatin Drugs 0.000 description 1
- 229950001022 enpromate Drugs 0.000 description 1
- 238000006911 enzymatic reaction Methods 0.000 description 1
- 239000002532 enzyme inhibitor Substances 0.000 description 1
- 229940125532 enzyme inhibitor Drugs 0.000 description 1
- YJGVMLPVUAXIQN-UHFFFAOYSA-N epipodophyllotoxin Natural products COC1=C(OC)C(OC)=CC(C2C3=CC=4OCOC=4C=C3C(O)C3C2C(OC3)=O)=C1 YJGVMLPVUAXIQN-UHFFFAOYSA-N 0.000 description 1
- 229960001904 epirubicin Drugs 0.000 description 1
- 229960003265 epirubicin hydrochloride Drugs 0.000 description 1
- 230000003628 erosive effect Effects 0.000 description 1
- 229960001842 estramustine Drugs 0.000 description 1
- 229960001766 estramustine phosphate sodium Drugs 0.000 description 1
- IIUMCNJTGSMNRO-VVSKJQCTSA-L estramustine sodium phosphate Chemical compound [Na+].[Na+].ClCCN(CCCl)C(=O)OC1=CC=C2[C@H]3CC[C@](C)([C@H](CC4)OP([O-])([O-])=O)[C@@H]4[C@@H]3CCC2=C1 IIUMCNJTGSMNRO-VVSKJQCTSA-L 0.000 description 1
- 229940011871 estrogen Drugs 0.000 description 1
- 239000000262 estrogen Substances 0.000 description 1
- 230000001076 estrogenic effect Effects 0.000 description 1
- CKHUMILJZKSHJU-UHFFFAOYSA-N ethane;tin Chemical compound CC[Sn] CKHUMILJZKSHJU-UHFFFAOYSA-N 0.000 description 1
- HYSIJEPDMLSIQJ-UHFFFAOYSA-N ethanolate;1-phenylbutane-1,3-dione;titanium(4+) Chemical compound [Ti+4].CC[O-].CC[O-].CC(=O)[CH-]C(=O)C1=CC=CC=C1.CC(=O)[CH-]C(=O)C1=CC=CC=C1 HYSIJEPDMLSIQJ-UHFFFAOYSA-N 0.000 description 1
- XPGDODOEEWLHOI-GSDHBNRESA-N ethyl (2s)-2-[[(2s)-2-[[(2s)-2-amino-3-(4-fluorophenyl)propanoyl]amino]-3-[3-[bis(2-chloroethyl)amino]phenyl]propanoyl]amino]-4-methylsulfanylbutanoate Chemical compound C([C@@H](C(=O)N[C@@H](CCSC)C(=O)OCC)NC(=O)[C@@H](N)CC=1C=CC(F)=CC=1)C1=CC=CC(N(CCCl)CCCl)=C1 XPGDODOEEWLHOI-GSDHBNRESA-N 0.000 description 1
- HZQPPNNARUQMJA-IMIWJGOWSA-N ethyl n-[4-[[(2r,4r)-2-(2,4-dichlorophenyl)-2-(imidazol-1-ylmethyl)-1,3-dioxolan-4-yl]methylsulfanyl]phenyl]carbamate;hydrochloride Chemical compound Cl.C1=CC(NC(=O)OCC)=CC=C1SC[C@@H]1O[C@@](CN2C=NC=C2)(C=2C(=CC(Cl)=CC=2)Cl)OC1 HZQPPNNARUQMJA-IMIWJGOWSA-N 0.000 description 1
- VBQUDDWATQWCPP-UHFFFAOYSA-N ethylsulfonylbenzene Chemical class CCS(=O)(=O)C1=CC=CC=C1 VBQUDDWATQWCPP-UHFFFAOYSA-N 0.000 description 1
- 229960005293 etodolac Drugs 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 229960000255 exemestane Drugs 0.000 description 1
- 208000021045 exocrine pancreatic carcinoma Diseases 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- 229960001419 fenoprofen Drugs 0.000 description 1
- 210000002950 fibroblast Anatomy 0.000 description 1
- 229960004177 filgrastim Drugs 0.000 description 1
- 239000000945 filler Substances 0.000 description 1
- 238000011049 filling Methods 0.000 description 1
- 239000000706 filtrate Substances 0.000 description 1
- 239000013020 final formulation Substances 0.000 description 1
- DBEPLOCGEIEOCV-WSBQPABSSA-N finasteride Chemical compound N([C@@H]1CC2)C(=O)C=C[C@]1(C)[C@@H]1[C@@H]2[C@@H]2CC[C@H](C(=O)NC(C)(C)C)[C@@]2(C)CC1 DBEPLOCGEIEOCV-WSBQPABSSA-N 0.000 description 1
- 229960004039 finasteride Drugs 0.000 description 1
- 229950006000 flezelastine Drugs 0.000 description 1
- 235000013312 flour Nutrition 0.000 description 1
- 229960000961 floxuridine Drugs 0.000 description 1
- ODKNJVUHOIMIIZ-RRKCRQDMSA-N floxuridine Chemical compound C1[C@H](O)[C@@H](CO)O[C@H]1N1C(=O)NC(=O)C(F)=C1 ODKNJVUHOIMIIZ-RRKCRQDMSA-N 0.000 description 1
- 229960000390 fludarabine Drugs 0.000 description 1
- 229960002390 flurbiprofen Drugs 0.000 description 1
- 229950005682 flurocitabine Drugs 0.000 description 1
- 229940064302 folacin Drugs 0.000 description 1
- 235000019152 folic acid Nutrition 0.000 description 1
- 239000011724 folic acid Substances 0.000 description 1
- 229950004217 forfenimex Drugs 0.000 description 1
- 229960004421 formestane Drugs 0.000 description 1
- OSVMTWJCGUFAOD-KZQROQTASA-N formestane Chemical compound O=C1CC[C@]2(C)[C@H]3CC[C@](C)(C(CC4)=O)[C@@H]4[C@@H]3CCC2=C1O OSVMTWJCGUFAOD-KZQROQTASA-N 0.000 description 1
- 229960004783 fotemustine Drugs 0.000 description 1
- YAKWPXVTIGTRJH-UHFFFAOYSA-N fotemustine Chemical compound CCOP(=O)(OCC)C(C)NC(=O)N(CCCl)N=O YAKWPXVTIGTRJH-UHFFFAOYSA-N 0.000 description 1
- 230000006870 function Effects 0.000 description 1
- 230000004927 fusion Effects 0.000 description 1
- 229940044658 gallium nitrate Drugs 0.000 description 1
- 108700032141 ganirelix Proteins 0.000 description 1
- 231100000414 gastrointestinal toxicity Toxicity 0.000 description 1
- 238000001641 gel filtration chromatography Methods 0.000 description 1
- 239000008273 gelatin Substances 0.000 description 1
- 229920000159 gelatin Polymers 0.000 description 1
- 239000007903 gelatin capsule Substances 0.000 description 1
- 235000019322 gelatine Nutrition 0.000 description 1
- 235000011852 gelatine desserts Nutrition 0.000 description 1
- 229960005144 gemcitabine hydrochloride Drugs 0.000 description 1
- 238000001415 gene therapy Methods 0.000 description 1
- 230000002068 genetic effect Effects 0.000 description 1
- 239000012362 glacial acetic acid Substances 0.000 description 1
- 230000009477 glass transition Effects 0.000 description 1
- 235000011187 glycerol Nutrition 0.000 description 1
- 235000013905 glycine and its sodium salt Nutrition 0.000 description 1
- 239000003979 granulating agent Substances 0.000 description 1
- 238000000892 gravimetry Methods 0.000 description 1
- 239000000665 guar gum Substances 0.000 description 1
- 235000010417 guar gum Nutrition 0.000 description 1
- 229960002154 guar gum Drugs 0.000 description 1
- 201000009277 hairy cell leukemia Diseases 0.000 description 1
- 235000015220 hamburgers Nutrition 0.000 description 1
- 231100000869 headache Toxicity 0.000 description 1
- 201000002222 hemangioblastoma Diseases 0.000 description 1
- 210000003677 hemocyte Anatomy 0.000 description 1
- 229940000351 hemocyte Drugs 0.000 description 1
- 231100000304 hepatotoxicity Toxicity 0.000 description 1
- 230000007686 hepatotoxicity Effects 0.000 description 1
- 125000000623 heterocyclic group Chemical class 0.000 description 1
- 210000005260 human cell Anatomy 0.000 description 1
- 230000036571 hydration Effects 0.000 description 1
- 238000006703 hydration reaction Methods 0.000 description 1
- 229960001330 hydroxycarbamide Drugs 0.000 description 1
- 235000010979 hydroxypropyl methyl cellulose Nutrition 0.000 description 1
- 229920003088 hydroxypropyl methyl cellulose Polymers 0.000 description 1
- BTXNYTINYBABQR-UHFFFAOYSA-N hypericin Chemical compound C12=C(O)C=C(O)C(C(C=3C(O)=CC(C)=C4C=33)=O)=C2C3=C2C3=C4C(C)=CC(O)=C3C(=O)C3=C(O)C=C(O)C1=C32 BTXNYTINYBABQR-UHFFFAOYSA-N 0.000 description 1
- 229940005608 hypericin Drugs 0.000 description 1
- PHOKTTKFQUYZPI-UHFFFAOYSA-N hypericin Natural products Cc1cc(O)c2c3C(=O)C(=Cc4c(O)c5c(O)cc(O)c6c7C(=O)C(=Cc8c(C)c1c2c(c78)c(c34)c56)O)O PHOKTTKFQUYZPI-UHFFFAOYSA-N 0.000 description 1
- 229960005236 ibandronic acid Drugs 0.000 description 1
- 229960001680 ibuprofen Drugs 0.000 description 1
- 229960000908 idarubicin Drugs 0.000 description 1
- 229960001176 idarubicin hydrochloride Drugs 0.000 description 1
- TZBDEVBNMSLVKT-UHFFFAOYSA-N idramantone Chemical compound C1C(C2)CC3CC1(O)CC2C3=O TZBDEVBNMSLVKT-UHFFFAOYSA-N 0.000 description 1
- 229950009926 idramantone Drugs 0.000 description 1
- NITYDPDXAAFEIT-DYVFJYSZSA-N ilomastat Chemical compound C1=CC=C2C(C[C@@H](C(=O)NC)NC(=O)[C@H](CC(C)C)CC(=O)NO)=CNC2=C1 NITYDPDXAAFEIT-DYVFJYSZSA-N 0.000 description 1
- 229960003696 ilomastat Drugs 0.000 description 1
- 230000002519 immonomodulatory effect Effects 0.000 description 1
- 238000003018 immunoassay Methods 0.000 description 1
- 238000002991 immunohistochemical analysis Methods 0.000 description 1
- 239000002955 immunomodulating agent Substances 0.000 description 1
- 229940121354 immunomodulator Drugs 0.000 description 1
- 230000002584 immunomodulator Effects 0.000 description 1
- 229960001438 immunostimulant agent Drugs 0.000 description 1
- 239000003022 immunostimulating agent Substances 0.000 description 1
- 230000008676 import Effects 0.000 description 1
- 230000002779 inactivation Effects 0.000 description 1
- 229960000905 indomethacin Drugs 0.000 description 1
- 230000006698 induction Effects 0.000 description 1
- 230000008595 infiltration Effects 0.000 description 1
- 238000001764 infiltration Methods 0.000 description 1
- 239000000976 ink Substances 0.000 description 1
- 230000002452 interceptive effect Effects 0.000 description 1
- 108010006088 interferon alfa-n1 Proteins 0.000 description 1
- 229960004061 interferon alfa-n1 Drugs 0.000 description 1
- 230000000968 intestinal effect Effects 0.000 description 1
- 238000007912 intraperitoneal administration Methods 0.000 description 1
- 239000011630 iodine Substances 0.000 description 1
- 229910052740 iodine Inorganic materials 0.000 description 1
- 229940044173 iodine-125 Drugs 0.000 description 1
- 239000003456 ion exchange resin Substances 0.000 description 1
- 229920003303 ion-exchange polymer Polymers 0.000 description 1
- 229960000779 irinotecan hydrochloride Drugs 0.000 description 1
- GURKHSYORGJETM-WAQYZQTGSA-N irinotecan hydrochloride (anhydrous) Chemical compound Cl.C1=C2C(CC)=C3CN(C(C4=C([C@@](C(=O)OC4)(O)CC)C=4)=O)C=4C3=NC2=CC=C1OC(=O)N(CC1)CCC1N1CCCCC1 GURKHSYORGJETM-WAQYZQTGSA-N 0.000 description 1
- 229910052742 iron Inorganic materials 0.000 description 1
- 229940075525 iron chelating agent Drugs 0.000 description 1
- 239000000797 iron chelating agent Substances 0.000 description 1
- 229950000855 iroplact Drugs 0.000 description 1
- 229950010984 irsogladine Drugs 0.000 description 1
- XUGNVMKQXJXZCD-UHFFFAOYSA-N isopropyl palmitate Chemical compound CCCCCCCCCCCCCCCC(=O)OC(C)C XUGNVMKQXJXZCD-UHFFFAOYSA-N 0.000 description 1
- GQWYWHOHRVVHAP-DHKPLNAMSA-N jaspamide Chemical compound C1([C@@H]2NC(=O)[C@@H](CC=3C4=CC=CC=C4NC=3Br)N(C)C(=O)[C@H](C)NC(=O)[C@@H](C)C/C(C)=C/[C@H](C)C[C@@H](OC(=O)C2)C)=CC=C(O)C=C1 GQWYWHOHRVVHAP-DHKPLNAMSA-N 0.000 description 1
- 108010052440 jasplakinolide Proteins 0.000 description 1
- GQWYWHOHRVVHAP-UHFFFAOYSA-N jasplakinolide Natural products C1C(=O)OC(C)CC(C)C=C(C)CC(C)C(=O)NC(C)C(=O)N(C)C(CC=2C3=CC=CC=C3NC=2Br)C(=O)NC1C1=CC=C(O)C=C1 GQWYWHOHRVVHAP-UHFFFAOYSA-N 0.000 description 1
- 108010091711 kahalalide F Proteins 0.000 description 1
- NLYAJNPCOHFWQQ-UHFFFAOYSA-N kaolin Chemical compound O.O.O=[Al]O[Si](=O)O[Si](=O)O[Al]=O NLYAJNPCOHFWQQ-UHFFFAOYSA-N 0.000 description 1
- 229960004752 ketorolac Drugs 0.000 description 1
- OZWKMVRBQXNZKK-UHFFFAOYSA-N ketorolac Chemical compound OC(=O)C1CCN2C1=CC=C2C(=O)C1=CC=CC=C1 OZWKMVRBQXNZKK-UHFFFAOYSA-N 0.000 description 1
- 208000022013 kidney Wilms tumor Diseases 0.000 description 1
- 239000008101 lactose Substances 0.000 description 1
- 229960002437 lanreotide Drugs 0.000 description 1
- 229960001739 lanreotide acetate Drugs 0.000 description 1
- 229940070765 laurate Drugs 0.000 description 1
- 238000002386 leaching Methods 0.000 description 1
- 229960002618 lenograstim Drugs 0.000 description 1
- 229940115286 lentinan Drugs 0.000 description 1
- 229960003136 leucine Drugs 0.000 description 1
- 208000032839 leukemia Diseases 0.000 description 1
- 210000000265 leukocyte Anatomy 0.000 description 1
- 150000002617 leukotrienes Chemical class 0.000 description 1
- 229960001614 levamisole Drugs 0.000 description 1
- UGFHIPBXIWJXNA-UHFFFAOYSA-N liarozole Chemical compound ClC1=CC=CC(C(C=2C=C3NC=NC3=CC=2)N2C=NC=C2)=C1 UGFHIPBXIWJXNA-UHFFFAOYSA-N 0.000 description 1
- 229950007056 liarozole Drugs 0.000 description 1
- 239000003446 ligand Substances 0.000 description 1
- 229940059904 light mineral oil Drugs 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 150000002632 lipids Chemical class 0.000 description 1
- 206010024627 liposarcoma Diseases 0.000 description 1
- 239000002502 liposome Substances 0.000 description 1
- 239000004973 liquid crystal related substance Substances 0.000 description 1
- 108010020270 lissoclinamide 7 Proteins 0.000 description 1
- RBBBWKUBQVARPL-SWQMWMPHSA-N lissoclinamide 7 Chemical compound C([C@H]1C(=O)N2CCC[C@H]2C2=N[C@@H]([C@H](O2)C)C(=O)N[C@@H](C=2SC[C@H](N=2)C(=O)N[C@H](CC=2C=CC=CC=2)C=2SC[C@H](N=2)C(=O)N1)C(C)C)C1=CC=CC=C1 RBBBWKUBQVARPL-SWQMWMPHSA-N 0.000 description 1
- RBBBWKUBQVARPL-UHFFFAOYSA-N lissoclinamide 7 Natural products N1C(=O)C(N=2)CSC=2C(CC=2C=CC=CC=2)NC(=O)C(N=2)CSC=2C(C(C)C)NC(=O)C(C(O2)C)N=C2C2CCCN2C(=O)C1CC1=CC=CC=C1 RBBBWKUBQVARPL-UHFFFAOYSA-N 0.000 description 1
- 201000007270 liver cancer Diseases 0.000 description 1
- 208000014018 liver neoplasm Diseases 0.000 description 1
- 229950000909 lometrexol Drugs 0.000 description 1
- 229960002247 lomustine Drugs 0.000 description 1
- 229960003538 lonidamine Drugs 0.000 description 1
- WDRYRZXSPDWGEB-UHFFFAOYSA-N lonidamine Chemical compound C12=CC=CC=C2C(C(=O)O)=NN1CC1=CC=C(Cl)C=C1Cl WDRYRZXSPDWGEB-UHFFFAOYSA-N 0.000 description 1
- YROQEQPFUCPDCP-UHFFFAOYSA-N losoxantrone Chemical compound OCCNCCN1N=C2C3=CC=CC(O)=C3C(=O)C3=C2C1=CC=C3NCCNCCO YROQEQPFUCPDCP-UHFFFAOYSA-N 0.000 description 1
- 229950008745 losoxantrone Drugs 0.000 description 1
- XDMHALQMTPSGEA-UHFFFAOYSA-N losoxantrone hydrochloride Chemical compound Cl.Cl.OCCNCCN1N=C2C3=CC=CC(O)=C3C(=O)C3=C2C1=CC=C3NCCNCCO XDMHALQMTPSGEA-UHFFFAOYSA-N 0.000 description 1
- PCZOHLXUXFIOCF-BXMDZJJMSA-N lovastatin Chemical compound C([C@H]1[C@@H](C)C=CC2=C[C@H](C)C[C@@H]([C@H]12)OC(=O)[C@@H](C)CC)C[C@@H]1C[C@@H](O)CC(=O)O1 PCZOHLXUXFIOCF-BXMDZJJMSA-N 0.000 description 1
- 229960004844 lovastatin Drugs 0.000 description 1
- QLJODMDSTUBWDW-UHFFFAOYSA-N lovastatin hydroxy acid Natural products C1=CC(C)C(CCC(O)CC(O)CC(O)=O)C2C(OC(=O)C(C)CC)CC(C)C=C21 QLJODMDSTUBWDW-UHFFFAOYSA-N 0.000 description 1
- 201000005202 lung cancer Diseases 0.000 description 1
- 208000020816 lung neoplasm Diseases 0.000 description 1
- RVFGKBWWUQOIOU-NDEPHWFRSA-N lurtotecan Chemical compound O=C([C@]1(O)CC)OCC(C(N2CC3=4)=O)=C1C=C2C3=NC1=CC=2OCCOC=2C=C1C=4CN1CCN(C)CC1 RVFGKBWWUQOIOU-NDEPHWFRSA-N 0.000 description 1
- 230000001926 lymphatic effect Effects 0.000 description 1
- 208000003747 lymphoid leukemia Diseases 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 229950001474 maitansine Drugs 0.000 description 1
- 208000024714 major depressive disease Diseases 0.000 description 1
- 238000007726 management method Methods 0.000 description 1
- MQXVYODZCMMZEM-ZYUZMQFOSA-N mannomustine Chemical compound ClCCNC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CNCCCl MQXVYODZCMMZEM-ZYUZMQFOSA-N 0.000 description 1
- 229950008612 mannomustine Drugs 0.000 description 1
- BLOFGONIVNXZME-YDMGZANHSA-N mannostatin A Chemical compound CS[C@@H]1[C@@H](N)[C@@H](O)[C@@H](O)[C@H]1O BLOFGONIVNXZME-YDMGZANHSA-N 0.000 description 1
- 229950008959 marimastat Drugs 0.000 description 1
- OCSMOTCMPXTDND-OUAUKWLOSA-N marimastat Chemical compound CNC(=O)[C@H](C(C)(C)C)NC(=O)[C@H](CC(C)C)[C@H](O)C(=O)NO OCSMOTCMPXTDND-OUAUKWLOSA-N 0.000 description 1
- 239000011159 matrix material Substances 0.000 description 1
- 230000035800 maturation Effects 0.000 description 1
- WKPWGQKGSOKKOO-RSFHAFMBSA-N maytansine Chemical compound CO[C@@H]([C@@]1(O)C[C@](OC(=O)N1)([C@H]([C@@H]1O[C@@]1(C)[C@@H](OC(=O)[C@H](C)N(C)C(C)=O)CC(=O)N1C)C)[H])\C=C\C=C(C)\CC2=CC(OC)=C(Cl)C1=C2 WKPWGQKGSOKKOO-RSFHAFMBSA-N 0.000 description 1
- 229960004961 mechlorethamine Drugs 0.000 description 1
- 230000007721 medicinal effect Effects 0.000 description 1
- 210000005074 megakaryoblast Anatomy 0.000 description 1
- MIKKOBKEXMRYFQ-WZTVWXICSA-N meglumine amidotrizoate Chemical compound C[NH2+]C[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO.CC(=O)NC1=C(I)C(NC(C)=O)=C(I)C(C([O-])=O)=C1I MIKKOBKEXMRYFQ-WZTVWXICSA-N 0.000 description 1
- 229960003846 melengestrol acetate Drugs 0.000 description 1
- 229960001929 meloxicam Drugs 0.000 description 1
- SGDBTWWWUNNDEQ-LBPRGKRZSA-N melphalan Chemical compound OC(=O)[C@@H](N)CC1=CC=C(N(CCCl)CCCl)C=C1 SGDBTWWWUNNDEQ-LBPRGKRZSA-N 0.000 description 1
- 229960001924 melphalan Drugs 0.000 description 1
- 206010027191 meningioma Diseases 0.000 description 1
- GLVAUDGFNGKCSF-UHFFFAOYSA-N mercaptopurine Chemical compound S=C1NC=NC2=C1NC=N2 GLVAUDGFNGKCSF-UHFFFAOYSA-N 0.000 description 1
- 229960001428 mercaptopurine Drugs 0.000 description 1
- 230000004060 metabolic process Effects 0.000 description 1
- 108700025096 meterelin Proteins 0.000 description 1
- 229960000485 methotrexate Drugs 0.000 description 1
- BKBBTCORRZMASO-ZOWNYOTGSA-M methotrexate monosodium Chemical compound [Na+].C=1N=C2N=C(N)N=C(N)C2=NC=1CN(C)C1=CC=C(C(=O)N[C@@H](CCC(O)=O)C([O-])=O)C=C1 BKBBTCORRZMASO-ZOWNYOTGSA-M 0.000 description 1
- 229960003058 methotrexate sodium Drugs 0.000 description 1
- 229920000609 methyl cellulose Polymers 0.000 description 1
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 1
- 239000001923 methylcellulose Substances 0.000 description 1
- TTWJBBZEZQICBI-UHFFFAOYSA-N metoclopramide Chemical compound CCN(CC)CCNC(=O)C1=CC(Cl)=C(N)C=C1OC TTWJBBZEZQICBI-UHFFFAOYSA-N 0.000 description 1
- 229960004503 metoclopramide Drugs 0.000 description 1
- QTFKTBRIGWJQQL-UHFFFAOYSA-N meturedepa Chemical compound C1C(C)(C)N1P(=O)(NC(=O)OCC)N1CC1(C)C QTFKTBRIGWJQQL-UHFFFAOYSA-N 0.000 description 1
- 229950009847 meturedepa Drugs 0.000 description 1
- HPNSFSBZBAHARI-UHFFFAOYSA-N micophenolic acid Natural products OC1=C(CC=C(C)CCC(O)=O)C(OC)=C(C)C2=C1C(=O)OC2 HPNSFSBZBAHARI-UHFFFAOYSA-N 0.000 description 1
- 244000005700 microbiome Species 0.000 description 1
- 239000004005 microsphere Substances 0.000 description 1
- 210000000110 microvilli Anatomy 0.000 description 1
- 229960003248 mifepristone Drugs 0.000 description 1
- VKHAHZOOUSRJNA-GCNJZUOMSA-N mifepristone Chemical compound C1([C@@H]2C3=C4CCC(=O)C=C4CC[C@H]3[C@@H]3CC[C@@]([C@]3(C2)C)(O)C#CC)=CC=C(N(C)C)C=C1 VKHAHZOOUSRJNA-GCNJZUOMSA-N 0.000 description 1
- 238000003801 milling Methods 0.000 description 1
- 229960003775 miltefosine Drugs 0.000 description 1
- PQLXHQMOHUQAKB-UHFFFAOYSA-N miltefosine Chemical compound CCCCCCCCCCCCCCCCOP([O-])(=O)OCC[N+](C)(C)C PQLXHQMOHUQAKB-UHFFFAOYSA-N 0.000 description 1
- 229950008541 mirimostim Drugs 0.000 description 1
- CFCUWKMKBJTWLW-BKHRDMLASA-N mithramycin Chemical compound O([C@@H]1C[C@@H](O[C@H](C)[C@H]1O)OC=1C=C2C=C3C[C@H]([C@@H](C(=O)C3=C(O)C2=C(O)C=1C)O[C@@H]1O[C@H](C)[C@@H](O)[C@H](O[C@@H]2O[C@H](C)[C@H](O)[C@H](O[C@@H]3O[C@H](C)[C@@H](O)[C@@](C)(O)C3)C2)C1)[C@H](OC)C(=O)[C@@H](O)[C@@H](C)O)[C@H]1C[C@@H](O)[C@H](O)[C@@H](C)O1 CFCUWKMKBJTWLW-BKHRDMLASA-N 0.000 description 1
- DRCJGCOYHLTVNR-ZUIZSQJWSA-N mitindomide Chemical compound C1=C[C@@H]2[C@@H]3[C@H]4C(=O)NC(=O)[C@H]4[C@@H]3[C@H]1[C@@H]1C(=O)NC(=O)[C@H]21 DRCJGCOYHLTVNR-ZUIZSQJWSA-N 0.000 description 1
- 229950000911 mitogillin Drugs 0.000 description 1
- 229960003539 mitoguazone Drugs 0.000 description 1
- MXWHMTNPTTVWDM-NXOFHUPFSA-N mitoguazone Chemical compound NC(N)=N\N=C(/C)\C=N\N=C(N)N MXWHMTNPTTVWDM-NXOFHUPFSA-N 0.000 description 1
- VFKZTMPDYBFSTM-GUCUJZIJSA-N mitolactol Chemical compound BrC[C@H](O)[C@@H](O)[C@@H](O)[C@H](O)CBr VFKZTMPDYBFSTM-GUCUJZIJSA-N 0.000 description 1
- 229950010913 mitolactol Drugs 0.000 description 1
- 229950005715 mitosper Drugs 0.000 description 1
- 229960000350 mitotane Drugs 0.000 description 1
- 229960001156 mitoxantrone Drugs 0.000 description 1
- KKZJGLLVHKMTCM-UHFFFAOYSA-N mitoxantrone Chemical compound O=C1C2=C(O)C=CC(O)=C2C(=O)C2=C1C(NCCNCCO)=CC=C2NCCNCCO KKZJGLLVHKMTCM-UHFFFAOYSA-N 0.000 description 1
- ZAHQPTJLOCWVPG-UHFFFAOYSA-N mitoxantrone dihydrochloride Chemical compound Cl.Cl.O=C1C2=C(O)C=CC(O)=C2C(=O)C2=C1C(NCCNCCO)=CC=C2NCCNCCO ZAHQPTJLOCWVPG-UHFFFAOYSA-N 0.000 description 1
- 238000012544 monitoring process Methods 0.000 description 1
- 229940035032 monophosphoryl lipid a Drugs 0.000 description 1
- FOYWNSCCNCUEPU-UHFFFAOYSA-N mopidamol Chemical compound C12=NC(N(CCO)CCO)=NC=C2N=C(N(CCO)CCO)N=C1N1CCCCC1 FOYWNSCCNCUEPU-UHFFFAOYSA-N 0.000 description 1
- 229950010718 mopidamol Drugs 0.000 description 1
- 230000001002 morphogenetic effect Effects 0.000 description 1
- 239000002324 mouth wash Substances 0.000 description 1
- 229940051866 mouthwash Drugs 0.000 description 1
- 229940087004 mustargen Drugs 0.000 description 1
- 229960000951 mycophenolic acid Drugs 0.000 description 1
- HPNSFSBZBAHARI-RUDMXATFSA-N mycophenolic acid Chemical compound OC1=C(C\C=C(/C)CCC(O)=O)C(OC)=C(C)C2=C1C(=O)OC2 HPNSFSBZBAHARI-RUDMXATFSA-N 0.000 description 1
- 208000001611 myxosarcoma Diseases 0.000 description 1
- CRJGESKKUOMBCT-PMACEKPBSA-N n-[(2s,3s)-1,3-dihydroxyoctadecan-2-yl]acetamide Chemical compound CCCCCCCCCCCCCCC[C@H](O)[C@H](CO)NC(C)=O CRJGESKKUOMBCT-PMACEKPBSA-N 0.000 description 1
- NKFHKYQGZDAKMX-PPRKPIOESA-N n-[(e)-1-[(2s,4s)-4-[(2r,4s,5s,6s)-4-amino-5-hydroxy-6-methyloxan-2-yl]oxy-2,5,12-trihydroxy-7-methoxy-6,11-dioxo-3,4-dihydro-1h-tetracen-2-yl]ethylideneamino]benzamide;hydrochloride Chemical compound Cl.O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(\C)=N\NC(=O)C=1C=CC=CC=1)[C@H]1C[C@H](N)[C@H](O)[C@H](C)O1 NKFHKYQGZDAKMX-PPRKPIOESA-N 0.000 description 1
- ARKYUICTMUZVEW-UHFFFAOYSA-N n-[5-[[5-[(3-amino-3-iminopropyl)carbamoyl]-1-methylpyrrol-3-yl]carbamoyl]-1-methylpyrrol-3-yl]-4-[[4-[bis(2-chloroethyl)amino]benzoyl]amino]-1-methylpyrrole-2-carboxamide Chemical compound C1=C(C(=O)NCCC(N)=N)N(C)C=C1NC(=O)C1=CC(NC(=O)C=2N(C=C(NC(=O)C=3C=CC(=CC=3)N(CCCl)CCCl)C=2)C)=CN1C ARKYUICTMUZVEW-UHFFFAOYSA-N 0.000 description 1
- WRINSSLBPNLASA-FOCLMDBBSA-N n-methyl-n-[(e)-(n-methylanilino)diazenyl]aniline Chemical compound C=1C=CC=CC=1N(C)\N=N\N(C)C1=CC=CC=C1 WRINSSLBPNLASA-FOCLMDBBSA-N 0.000 description 1
- XJXLRNPVSQADMZ-UHFFFAOYSA-N n-methyl-n-[4-[(7-methyl-3h-imidazo[4,5-f]quinolin-9-yl)amino]phenyl]acetamide Chemical compound C1=CC(N(C(C)=O)C)=CC=C1NC1=CC(C)=NC2=CC=C(NC=N3)C3=C12 XJXLRNPVSQADMZ-UHFFFAOYSA-N 0.000 description 1
- RWHUEXWOYVBUCI-ITQXDASVSA-N nafarelin Chemical compound C([C@@H](C(=O)N[C@H](CC=1C=C2C=CC=CC2=CC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N1[C@@H](CCC1)C(=O)NCC(N)=O)NC(=O)[C@H](CO)NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@H](CC=1NC=NC=1)NC(=O)[C@H]1NC(=O)CC1)C1=CC=C(O)C=C1 RWHUEXWOYVBUCI-ITQXDASVSA-N 0.000 description 1
- 229960002333 nafarelin Drugs 0.000 description 1
- JZGDNMXSOCDEFQ-UHFFFAOYSA-N napavin Chemical compound C1C(CC)(O)CC(C2)CN1CCC(C1=CC=CC=C1N1)=C1C2(C(=O)OC)C(C(=C1)OC)=CC2=C1N(C)C1C2(C23)CCN3CC=CC2(CC)C(O)C1(O)C(=O)NCCNC1=CC=C(N=[N+]=[N-])C=C1[N+]([O-])=O JZGDNMXSOCDEFQ-UHFFFAOYSA-N 0.000 description 1
- 229960002009 naproxen Drugs 0.000 description 1
- CMWTZPSULFXXJA-VIFPVBQESA-N naproxen Chemical compound C1=C([C@H](C)C(O)=O)C=CC2=CC(OC)=CC=C21 CMWTZPSULFXXJA-VIFPVBQESA-N 0.000 description 1
- 229940097496 nasal spray Drugs 0.000 description 1
- 239000007922 nasal spray Substances 0.000 description 1
- CTMCWCONSULRHO-UHQPFXKFSA-N nemorubicin Chemical compound C1CO[C@H](OC)CN1[C@@H]1[C@H](O)[C@H](C)O[C@@H](O[C@@H]2C3=C(O)C=4C(=O)C5=C(OC)C=CC=C5C(=O)C=4C(O)=C3C[C@](O)(C2)C(=O)CO)C1 CTMCWCONSULRHO-UHQPFXKFSA-N 0.000 description 1
- 229950010159 nemorubicin Drugs 0.000 description 1
- QZGIWPZCWHMVQL-UIYAJPBUSA-N neocarzinostatin chromophore Chemical compound O1[C@H](C)[C@H](O)[C@H](O)[C@@H](NC)[C@H]1O[C@@H]1C/2=C/C#C[C@H]3O[C@@]3([C@@H]3OC(=O)OC3)C#CC\2=C[C@H]1OC(=O)C1=C(O)C=CC2=C(C)C=C(OC)C=C12 QZGIWPZCWHMVQL-UIYAJPBUSA-N 0.000 description 1
- 208000025189 neoplasm of testis Diseases 0.000 description 1
- MQYXUWHLBZFQQO-UHFFFAOYSA-N nepehinol Natural products C1CC(O)C(C)(C)C2CCC3(C)C4(C)CCC5(C)CCC(C(=C)C)C5C4CCC3C21C MQYXUWHLBZFQQO-UHFFFAOYSA-N 0.000 description 1
- 201000008026 nephroblastoma Diseases 0.000 description 1
- 231100000417 nephrotoxicity Toxicity 0.000 description 1
- PUUSSSIBPPTKTP-UHFFFAOYSA-N neridronic acid Chemical compound NCCCCCC(O)(P(O)(O)=O)P(O)(O)=O PUUSSSIBPPTKTP-UHFFFAOYSA-N 0.000 description 1
- 229950010733 neridronic acid Drugs 0.000 description 1
- 230000007935 neutral effect Effects 0.000 description 1
- 238000001683 neutron diffraction Methods 0.000 description 1
- 239000012299 nitrogen atmosphere Substances 0.000 description 1
- OSTGTTZJOCZWJG-UHFFFAOYSA-N nitrosourea Chemical compound NC(=O)N=NO OSTGTTZJOCZWJG-UHFFFAOYSA-N 0.000 description 1
- KGTDRFCXGRULNK-JYOBTZKQSA-N nogalamycin Chemical compound CO[C@@H]1[C@@](OC)(C)[C@@H](OC)[C@H](C)O[C@H]1O[C@@H]1C2=C(O)C(C(=O)C3=C(O)C=C4[C@@]5(C)O[C@H]([C@H]([C@@H]([C@H]5O)N(C)C)O)OC4=C3C3=O)=C3C=C2[C@@H](C(=O)OC)[C@@](C)(O)C1 KGTDRFCXGRULNK-JYOBTZKQSA-N 0.000 description 1
- 229950009266 nogalamycin Drugs 0.000 description 1
- 229920001778 nylon Polymers 0.000 description 1
- XMLQWXUVTXCDDL-UHFFFAOYSA-N oleyl alcohol Natural products CCCCCCC=CCCCCCCCCCCO XMLQWXUVTXCDDL-UHFFFAOYSA-N 0.000 description 1
- 229940055577 oleyl alcohol Drugs 0.000 description 1
- 229950011093 onapristone Drugs 0.000 description 1
- 229940054534 ophthalmic solution Drugs 0.000 description 1
- 239000002997 ophthalmic solution Substances 0.000 description 1
- 229940041672 oral gel Drugs 0.000 description 1
- ZLLOIFNEEWYATC-XMUHMHRVSA-N osaterone Chemical compound C1=C(Cl)C2=CC(=O)OC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@@](C(=O)C)(O)[C@@]1(C)CC2 ZLLOIFNEEWYATC-XMUHMHRVSA-N 0.000 description 1
- 229950006466 osaterone Drugs 0.000 description 1
- 201000008968 osteosarcoma Diseases 0.000 description 1
- 229960001756 oxaliplatin Drugs 0.000 description 1
- DWAFYCQODLXJNR-BNTLRKBRSA-L oxaliplatin Chemical compound O1C(=O)C(=O)O[Pt]11N[C@@H]2CCCC[C@H]2N1 DWAFYCQODLXJNR-BNTLRKBRSA-L 0.000 description 1
- 229950000370 oxisuran Drugs 0.000 description 1
- TWNQGVIAIRXVLR-UHFFFAOYSA-N oxo(oxoalumanyloxy)alumane Chemical compound O=[Al]O[Al]=O TWNQGVIAIRXVLR-UHFFFAOYSA-N 0.000 description 1
- 239000006174 pH buffer Substances 0.000 description 1
- VYOQBYCIIJYKJA-VORKOXQSSA-N palau'amine Chemical compound N([C@@]12[C@@H](Cl)[C@@H]([C@@H]3[C@@H]2[C@]24N=C(N)N[C@H]2N2C=CC=C2C(=O)N4C3)CN)C(N)=N[C@H]1O VYOQBYCIIJYKJA-VORKOXQSSA-N 0.000 description 1
- 125000000913 palmityl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- WRUUGTRCQOWXEG-UHFFFAOYSA-N pamidronate Chemical compound NCCC(O)(P(O)(O)=O)P(O)(O)=O WRUUGTRCQOWXEG-UHFFFAOYSA-N 0.000 description 1
- 229960003978 pamidronic acid Drugs 0.000 description 1
- 229950003440 panomifene Drugs 0.000 description 1
- 229960005489 paracetamol Drugs 0.000 description 1
- 239000006072 paste Substances 0.000 description 1
- 238000009928 pasteurization Methods 0.000 description 1
- 101150037969 pde-6 gene Proteins 0.000 description 1
- DOHVAKFYAHLCJP-UHFFFAOYSA-N peldesine Chemical compound C1=2NC(N)=NC(=O)C=2NC=C1CC1=CC=CN=C1 DOHVAKFYAHLCJP-UHFFFAOYSA-N 0.000 description 1
- 229950000039 peldesine Drugs 0.000 description 1
- 229950006960 peliomycin Drugs 0.000 description 1
- 230000000149 penetrating effect Effects 0.000 description 1
- VOKSWYLNZZRQPF-GDIGMMSISA-N pentazocine Chemical compound C1C2=CC=C(O)C=C2[C@@]2(C)[C@@H](C)[C@@H]1N(CC=C(C)C)CC2 VOKSWYLNZZRQPF-GDIGMMSISA-N 0.000 description 1
- 229960005301 pentazocine Drugs 0.000 description 1
- 229960002340 pentostatin Drugs 0.000 description 1
- FPVKHBSQESCIEP-JQCXWYLXSA-N pentostatin Chemical compound C1[C@H](O)[C@@H](CO)O[C@H]1N1C(N=CNC[C@H]2O)=C2N=C1 FPVKHBSQESCIEP-JQCXWYLXSA-N 0.000 description 1
- 229950003180 peplomycin Drugs 0.000 description 1
- 108010091748 peptide A Proteins 0.000 description 1
- WTWWXOGTJWMJHI-UHFFFAOYSA-N perflubron Chemical compound FC(F)(F)C(F)(F)C(F)(F)C(F)(F)C(F)(F)C(F)(F)C(F)(F)C(F)(F)Br WTWWXOGTJWMJHI-UHFFFAOYSA-N 0.000 description 1
- 210000005259 peripheral blood Anatomy 0.000 description 1
- 239000011886 peripheral blood Substances 0.000 description 1
- 239000000546 pharmaceutical excipient Substances 0.000 description 1
- 230000003285 pharmacodynamic effect Effects 0.000 description 1
- 239000002831 pharmacologic agent Substances 0.000 description 1
- LCADVYTXPLBAGB-GNCBHIOISA-N phenalamide A1 Natural products CC(CO)NC(=O)C(=CC=CC=C/C=C/C(=C/C(C)C(O)C(=CC(C)CCc1ccccc1)C)/C)C LCADVYTXPLBAGB-GNCBHIOISA-N 0.000 description 1
- 150000004713 phosphodiesters Chemical class 0.000 description 1
- 238000000554 physical therapy Methods 0.000 description 1
- 230000004962 physiological condition Effects 0.000 description 1
- 230000035790 physiological processes and functions Effects 0.000 description 1
- BOTWFXYSPFMFNR-PYDDKJGSSA-N phytol Chemical compound CC(C)CCC[C@@H](C)CCC[C@@H](C)CCC\C(C)=C\CO BOTWFXYSPFMFNR-PYDDKJGSSA-N 0.000 description 1
- 229960001416 pilocarpine Drugs 0.000 description 1
- 208000024724 pineal body neoplasm Diseases 0.000 description 1
- 201000004123 pineal gland cancer Diseases 0.000 description 1
- PMJHHCWVYXUKFD-UHFFFAOYSA-N piperylene Natural products CC=CC=C PMJHHCWVYXUKFD-UHFFFAOYSA-N 0.000 description 1
- NJBFOOCLYDNZJN-UHFFFAOYSA-N pipobroman Chemical compound BrCCC(=O)N1CCN(C(=O)CCBr)CC1 NJBFOOCLYDNZJN-UHFFFAOYSA-N 0.000 description 1
- 229960000952 pipobroman Drugs 0.000 description 1
- NUKCGLDCWQXYOQ-UHFFFAOYSA-N piposulfan Chemical compound CS(=O)(=O)OCCC(=O)N1CCN(C(=O)CCOS(C)(=O)=O)CC1 NUKCGLDCWQXYOQ-UHFFFAOYSA-N 0.000 description 1
- 229950001100 piposulfan Drugs 0.000 description 1
- 229960001221 pirarubicin Drugs 0.000 description 1
- XESARGFCSKSFID-FLLFQEBCSA-N pirazofurin Chemical compound OC1=C(C(=O)N)NN=C1[C@H]1[C@H](O)[C@H](O)[C@@H](CO)O1 XESARGFCSKSFID-FLLFQEBCSA-N 0.000 description 1
- 229950001030 piritrexim Drugs 0.000 description 1
- 229950001746 piroxantrone Drugs 0.000 description 1
- QYSPLQLAKJAUJT-UHFFFAOYSA-N piroxicam Chemical compound OC=1C2=CC=CC=C2S(=O)(=O)N(C)C=1C(=O)NC1=CC=CC=N1 QYSPLQLAKJAUJT-UHFFFAOYSA-N 0.000 description 1
- 229960002702 piroxicam Drugs 0.000 description 1
- 210000002381 plasma Anatomy 0.000 description 1
- 229940127126 plasminogen activator Drugs 0.000 description 1
- 239000011505 plaster Substances 0.000 description 1
- 239000004033 plastic Substances 0.000 description 1
- 229920003023 plastic Polymers 0.000 description 1
- 150000003057 platinum Chemical class 0.000 description 1
- 229960003171 plicamycin Drugs 0.000 description 1
- JKPDEYAOCSQBSZ-OEUJLIAZSA-N plomestane Chemical compound O=C1CC[C@]2(CC#C)[C@H]3CC[C@](C)(C(CC4)=O)[C@@H]4[C@@H]3CCC2=C1 JKPDEYAOCSQBSZ-OEUJLIAZSA-N 0.000 description 1
- 229950004541 plomestane Drugs 0.000 description 1
- 229960000540 polacrilin potassium Drugs 0.000 description 1
- 229920000191 poly(N-vinyl pyrrolidone) Polymers 0.000 description 1
- 229920000768 polyamine Polymers 0.000 description 1
- 239000008389 polyethoxylated castor oil Substances 0.000 description 1
- 239000000244 polyoxyethylene sorbitan monooleate Substances 0.000 description 1
- 229940068968 polysorbate 80 Drugs 0.000 description 1
- 235000013809 polyvinylpolypyrrolidone Nutrition 0.000 description 1
- 229920000523 polyvinylpolypyrrolidone Polymers 0.000 description 1
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 1
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 1
- 150000004032 porphyrins Chemical class 0.000 description 1
- WVWZXTJUCNEUAE-UHFFFAOYSA-M potassium;1,2-bis(ethenyl)benzene;2-methylprop-2-enoate Chemical compound [K+].CC(=C)C([O-])=O.C=CC1=CC=CC=C1C=C WVWZXTJUCNEUAE-UHFFFAOYSA-M 0.000 description 1
- 229920001592 potato starch Polymers 0.000 description 1
- 239000002244 precipitate Substances 0.000 description 1
- 238000001556 precipitation Methods 0.000 description 1
- 229960004694 prednimustine Drugs 0.000 description 1
- 229960004618 prednisone Drugs 0.000 description 1
- XOFYZVNMUHMLCC-ZPOLXVRWSA-N prednisone Chemical compound O=C1C=C[C@]2(C)[C@H]3C(=O)C[C@](C)([C@@](CC4)(O)C(=O)CO)[C@@H]4[C@@H]3CCC2=C1 XOFYZVNMUHMLCC-ZPOLXVRWSA-N 0.000 description 1
- 238000003825 pressing Methods 0.000 description 1
- 150000003141 primary amines Chemical class 0.000 description 1
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 1
- 238000007639 printing Methods 0.000 description 1
- 229960001586 procarbazine hydrochloride Drugs 0.000 description 1
- 229960003387 progesterone Drugs 0.000 description 1
- 239000000186 progesterone Substances 0.000 description 1
- 230000001737 promoting effect Effects 0.000 description 1
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 150000003180 prostaglandins Chemical class 0.000 description 1
- 102000017953 prostanoid receptors Human genes 0.000 description 1
- 108050007059 prostanoid receptors Proteins 0.000 description 1
- 235000019833 protease Nutrition 0.000 description 1
- 235000019419 proteases Nutrition 0.000 description 1
- 239000003207 proteasome inhibitor Substances 0.000 description 1
- 239000003528 protein farnesyltransferase inhibitor Substances 0.000 description 1
- 108020000494 protein-tyrosine phosphatase Proteins 0.000 description 1
- 108090000623 proteins and genes Proteins 0.000 description 1
- 230000017854 proteolysis Effects 0.000 description 1
- 238000000425 proton nuclear magnetic resonance spectrum Methods 0.000 description 1
- SSKVDVBQSWQEGJ-UHFFFAOYSA-N pseudohypericin Natural products C12=C(O)C=C(O)C(C(C=3C(O)=CC(O)=C4C=33)=O)=C2C3=C2C3=C4C(C)=CC(O)=C3C(=O)C3=C(O)C=C(O)C1=C32 SSKVDVBQSWQEGJ-UHFFFAOYSA-N 0.000 description 1
- 238000010926 purge Methods 0.000 description 1
- 239000000784 purine nucleoside phosphorylase inhibitor Substances 0.000 description 1
- 150000003212 purines Chemical class 0.000 description 1
- 229950010131 puromycin Drugs 0.000 description 1
- MKSVFGKWZLUTTO-FZFAUISWSA-N puromycin dihydrochloride Chemical compound Cl.Cl.C1=CC(OC)=CC=C1C[C@H](N)C(=O)N[C@H]1[C@@H](O)[C@H](N2C3=NC=NC(=C3N=C2)N(C)C)O[C@@H]1CO MKSVFGKWZLUTTO-FZFAUISWSA-N 0.000 description 1
- JEXVQSWXXUJEMA-UHFFFAOYSA-N pyrazol-3-one Chemical class O=C1C=CN=N1 JEXVQSWXXUJEMA-UHFFFAOYSA-N 0.000 description 1
- 150000003230 pyrimidines Chemical class 0.000 description 1
- HNJBEVLQSNELDL-UHFFFAOYSA-N pyrrolidin-2-one Chemical compound O=C1CCCN1 HNJBEVLQSNELDL-UHFFFAOYSA-N 0.000 description 1
- 238000004445 quantitative analysis Methods 0.000 description 1
- 238000010791 quenching Methods 0.000 description 1
- 230000000171 quenching effect Effects 0.000 description 1
- 230000005855 radiation Effects 0.000 description 1
- 229960002185 ranimustine Drugs 0.000 description 1
- 239000011541 reaction mixture Substances 0.000 description 1
- 230000000384 rearing effect Effects 0.000 description 1
- 239000002464 receptor antagonist Substances 0.000 description 1
- 229940044551 receptor antagonist Drugs 0.000 description 1
- 102000005962 receptors Human genes 0.000 description 1
- 108020003175 receptors Proteins 0.000 description 1
- 238000011084 recovery Methods 0.000 description 1
- 229950002225 retelliptine Drugs 0.000 description 1
- 210000001525 retina Anatomy 0.000 description 1
- 201000009410 rhabdomyosarcoma Diseases 0.000 description 1
- WUAPFZMCVAUBPE-IGMARMGPSA-N rhenium-186 Chemical compound [186Re] WUAPFZMCVAUBPE-IGMARMGPSA-N 0.000 description 1
- 108091092562 ribozyme Proteins 0.000 description 1
- 230000000630 rising effect Effects 0.000 description 1
- NCDNCNXCDXHOMX-XGKFQTDJSA-N ritonavir Chemical compound N([C@@H](C(C)C)C(=O)N[C@H](C[C@H](O)[C@H](CC=1C=CC=CC=1)NC(=O)OCC=1SC=NC=1)CC=1C=CC=CC=1)C(=O)N(C)CC1=CSC(C(C)C)=N1 NCDNCNXCDXHOMX-XGKFQTDJSA-N 0.000 description 1
- 229960000311 ritonavir Drugs 0.000 description 1
- 238000009490 roller compaction Methods 0.000 description 1
- 108700033545 romurtide Proteins 0.000 description 1
- 229950003733 romurtide Drugs 0.000 description 1
- 229960003522 roquinimex Drugs 0.000 description 1
- YGSDEFSMJLZEOE-UHFFFAOYSA-M salicylate Chemical compound OC1=CC=CC=C1C([O-])=O YGSDEFSMJLZEOE-UHFFFAOYSA-M 0.000 description 1
- 229960001860 salicylate Drugs 0.000 description 1
- 238000005070 sampling Methods 0.000 description 1
- 229930192242 sarcophytol Natural products 0.000 description 1
- 229960002530 sargramostim Drugs 0.000 description 1
- 108010038379 sargramostim Proteins 0.000 description 1
- 238000006748 scratching Methods 0.000 description 1
- 230000002393 scratching effect Effects 0.000 description 1
- 238000007789 sealing Methods 0.000 description 1
- 201000008407 sebaceous adenocarcinoma Diseases 0.000 description 1
- 201000007321 sebaceous carcinoma Diseases 0.000 description 1
- 150000003335 secondary amines Chemical class 0.000 description 1
- 208000012201 sexual and gender identity disease Diseases 0.000 description 1
- 208000015891 sexual disease Diseases 0.000 description 1
- 208000013220 shortness of breath Diseases 0.000 description 1
- RMAQACBXLXPBSY-UHFFFAOYSA-N silicic acid Chemical compound O[Si](O)(O)O RMAQACBXLXPBSY-UHFFFAOYSA-N 0.000 description 1
- 235000012239 silicon dioxide Nutrition 0.000 description 1
- 229910052709 silver Inorganic materials 0.000 description 1
- 239000004332 silver Substances 0.000 description 1
- 229950009089 simtrazene Drugs 0.000 description 1
- 238000004088 simulation Methods 0.000 description 1
- 229950001403 sizofiran Drugs 0.000 description 1
- 208000000587 small cell lung carcinoma Diseases 0.000 description 1
- 239000003998 snake venom Substances 0.000 description 1
- 229950010372 sobuzoxane Drugs 0.000 description 1
- 229940006198 sodium phenylacetate Drugs 0.000 description 1
- 229940080313 sodium starch Drugs 0.000 description 1
- JSMHQMIPUOPQLR-UHFFFAOYSA-M sodium;dioctadecyl phosphate Chemical compound [Na+].CCCCCCCCCCCCCCCCCCOP([O-])(=O)OCCCCCCCCCCCCCCCCCC JSMHQMIPUOPQLR-UHFFFAOYSA-M 0.000 description 1
- 239000007901 soft capsule Substances 0.000 description 1
- 239000007905 soft elastic gelatin capsule Substances 0.000 description 1
- 239000008279 sol Substances 0.000 description 1
- 239000012439 solid excipient Substances 0.000 description 1
- 239000007790 solid phase Substances 0.000 description 1
- 238000003746 solid phase reaction Methods 0.000 description 1
- 239000012265 solid product Substances 0.000 description 1
- 238000010671 solid-state reaction Methods 0.000 description 1
- 239000012453 solvate Substances 0.000 description 1
- 229950004225 sonermin Drugs 0.000 description 1
- 235000011076 sorbitan monostearate Nutrition 0.000 description 1
- 239000001587 sorbitan monostearate Substances 0.000 description 1
- 229940035048 sorbitan monostearate Drugs 0.000 description 1
- 229950004796 sparfosic acid Drugs 0.000 description 1
- 229950009641 sparsomycin Drugs 0.000 description 1
- XKLZIVIOZDNKEQ-CLQLPEFOSA-N sparsomycin Chemical compound CSC[S@](=O)C[C@H](CO)NC(=O)\C=C\C1=C(C)NC(=O)NC1=O XKLZIVIOZDNKEQ-CLQLPEFOSA-N 0.000 description 1
- XKLZIVIOZDNKEQ-UHFFFAOYSA-N sparsomycin Natural products CSCS(=O)CC(CO)NC(=O)C=CC1=C(C)NC(=O)NC1=O XKLZIVIOZDNKEQ-UHFFFAOYSA-N 0.000 description 1
- 241000894007 species Species 0.000 description 1
- YBZRLMLGUBIIDN-NZSGCTDASA-N spicamycin Chemical compound O1[C@@H](C(O)CO)[C@H](NC(=O)CNC(=O)CCCCCCCCCCCCC(C)C)[C@@H](O)[C@@H](O)[C@H]1NC1=NC=NC2=C1N=CN2 YBZRLMLGUBIIDN-NZSGCTDASA-N 0.000 description 1
- YBZRLMLGUBIIDN-UHFFFAOYSA-N spicamycin Natural products O1C(C(O)CO)C(NC(=O)CNC(=O)CCCCCCCCCCCCC(C)C)C(O)C(O)C1NC1=NC=NC2=C1NC=N2 YBZRLMLGUBIIDN-UHFFFAOYSA-N 0.000 description 1
- 210000000952 spleen Anatomy 0.000 description 1
- 239000007921 spray Substances 0.000 description 1
- 206010041823 squamous cell carcinoma Diseases 0.000 description 1
- 238000010186 staining Methods 0.000 description 1
- 230000003068 static effect Effects 0.000 description 1
- 210000000130 stem cell Anatomy 0.000 description 1
- 230000024642 stem cell division Effects 0.000 description 1
- 239000008174 sterile solution Substances 0.000 description 1
- 150000003431 steroids Chemical class 0.000 description 1
- 239000012258 stirred mixture Substances 0.000 description 1
- UCSJYZPVAKXKNQ-HZYVHMACSA-N streptomycin Natural products CN[C@H]1[C@H](O)[C@@H](O)[C@H](CO)O[C@H]1O[C@@H]1[C@](C=O)(O)[C@H](C)O[C@H]1O[C@@H]1[C@@H](NC(N)=N)[C@H](O)[C@@H](NC(N)=N)[C@H](O)[C@H]1O UCSJYZPVAKXKNQ-HZYVHMACSA-N 0.000 description 1
- 229960005322 streptomycin Drugs 0.000 description 1
- QTENRWWVYAAPBI-YCRXJPFRSA-N streptomycin sulfate Chemical compound OS(O)(=O)=O.OS(O)(=O)=O.OS(O)(=O)=O.CN[C@H]1[C@H](O)[C@@H](O)[C@H](CO)O[C@H]1O[C@@H]1[C@](C=O)(O)[C@H](C)O[C@H]1O[C@@H]1[C@@H](N=C(N)N)[C@H](O)[C@@H](N=C(N)N)[C@H](O)[C@H]1O.CN[C@H]1[C@H](O)[C@@H](O)[C@H](CO)O[C@H]1O[C@@H]1[C@](C=O)(O)[C@H](C)O[C@H]1O[C@@H]1[C@@H](N=C(N)N)[C@H](O)[C@@H](N=C(N)N)[C@H](O)[C@H]1O QTENRWWVYAAPBI-YCRXJPFRSA-N 0.000 description 1
- 229960001052 streptozocin Drugs 0.000 description 1
- ZSJLQEPLLKMAKR-GKHCUFPYSA-N streptozocin Chemical compound O=NN(C)C(=O)N[C@H]1[C@@H](O)O[C@H](CO)[C@@H](O)[C@@H]1O ZSJLQEPLLKMAKR-GKHCUFPYSA-N 0.000 description 1
- 125000001174 sulfone group Chemical group 0.000 description 1
- 150000003457 sulfones Chemical class 0.000 description 1
- 229960000894 sulindac Drugs 0.000 description 1
- MLKXDPUZXIRXEP-MFOYZWKCSA-N sulindac Chemical compound CC1=C(CC(O)=O)C2=CC(F)=CC=C2\C1=C/C1=CC=C(S(C)=O)C=C1 MLKXDPUZXIRXEP-MFOYZWKCSA-N 0.000 description 1
- 229950007841 sulofenur Drugs 0.000 description 1
- 235000020238 sunflower seed Nutrition 0.000 description 1
- 231100000617 superantigen Toxicity 0.000 description 1
- 239000000829 suppository Substances 0.000 description 1
- FIAFUQMPZJWCLV-UHFFFAOYSA-N suramin Chemical compound OS(=O)(=O)C1=CC(S(O)(=O)=O)=C2C(NC(=O)C3=CC=C(C(=C3)NC(=O)C=3C=C(NC(=O)NC=4C=C(C=CC=4)C(=O)NC=4C(=CC=C(C=4)C(=O)NC=4C5=C(C=C(C=C5C(=CC=4)S(O)(=O)=O)S(O)(=O)=O)S(O)(=O)=O)C)C=CC=3)C)=CC=C(S(O)(=O)=O)C2=C1 FIAFUQMPZJWCLV-UHFFFAOYSA-N 0.000 description 1
- 229960005314 suramin Drugs 0.000 description 1
- 238000004441 surface measurement Methods 0.000 description 1
- 238000013268 sustained release Methods 0.000 description 1
- 239000012730 sustained-release form Substances 0.000 description 1
- 206010042863 synovial sarcoma Diseases 0.000 description 1
- 239000006188 syrup Substances 0.000 description 1
- 235000020357 syrup Nutrition 0.000 description 1
- 108700003774 talisomycin Proteins 0.000 description 1
- 229950002687 talisomycin Drugs 0.000 description 1
- 108010021891 tallimustine Proteins 0.000 description 1
- 229950005667 tallimustine Drugs 0.000 description 1
- 239000008399 tap water Substances 0.000 description 1
- 235000020679 tap water Nutrition 0.000 description 1
- 229960000565 tazarotene Drugs 0.000 description 1
- 239000003277 telomerase inhibitor Substances 0.000 description 1
- 229950008703 teroxirone Drugs 0.000 description 1
- 229960005353 testolactone Drugs 0.000 description 1
- BPEWUONYVDABNZ-DZBHQSCQSA-N testolactone Chemical compound O=C1C=C[C@]2(C)[C@H]3CC[C@](C)(OC(=O)CC4)[C@@H]4[C@@H]3CCC2=C1 BPEWUONYVDABNZ-DZBHQSCQSA-N 0.000 description 1
- WXZSUBHBYQYTNM-WMDJANBXSA-N tetrazomine Chemical compound C=1([C@@H]2CO[C@@H]3[C@H]4C[C@@H](CO)[C@H](N4C)[C@@H](N23)CC=1C=C1)C(OC)=C1NC(=O)C1NCCC[C@H]1O WXZSUBHBYQYTNM-WMDJANBXSA-N 0.000 description 1
- 238000002411 thermogravimetry Methods 0.000 description 1
- RTKIYNMVFMVABJ-UHFFFAOYSA-L thimerosal Chemical compound [Na+].CC[Hg]SC1=CC=CC=C1C([O-])=O RTKIYNMVFMVABJ-UHFFFAOYSA-L 0.000 description 1
- 108010062880 thiocoraline Proteins 0.000 description 1
- UPGGKUQISSWRJJ-UHFFFAOYSA-N thiocoraline Natural products CN1C(=O)CNC(=O)C(NC(=O)C=2C(=CC3=CC=CC=C3N=2)O)CSC(=O)C(CSC)N(C)C(=O)C(N(C(=O)CNC2=O)C)CSSCC1C(=O)N(C)C(CSC)C(=O)SCC2NC(=O)C1=NC2=CC=CC=C2C=C1O UPGGKUQISSWRJJ-UHFFFAOYSA-N 0.000 description 1
- 206010043554 thrombocytopenia Diseases 0.000 description 1
- NZVYCXVTEHPMHE-ZSUJOUNUSA-N thymalfasin Chemical compound CC(=O)N[C@@H](CO)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](C)C(=O)N[C@@H](C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](C)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(N)=O)C(O)=O NZVYCXVTEHPMHE-ZSUJOUNUSA-N 0.000 description 1
- 229960004231 thymalfasin Drugs 0.000 description 1
- 108010013515 thymopoietin receptor Proteins 0.000 description 1
- 229950010183 thymotrinan Drugs 0.000 description 1
- 229960003723 tiazofurine Drugs 0.000 description 1
- FVRDYQYEVDDKCR-DBRKOABJSA-N tiazofurine Chemical compound NC(=O)C1=CSC([C@H]2[C@@H]([C@H](O)[C@@H](CO)O2)O)=N1 FVRDYQYEVDDKCR-DBRKOABJSA-N 0.000 description 1
- 229940098465 tincture Drugs 0.000 description 1
- WSTYNZDAOAEEKG-GWJSGULQSA-N tingenone Chemical compound CC1=C(O)C(=O)C=C2[C@@](CC[C@]3([C@@H]4C[C@H](C(C[C@@]4(CC[C@@]33C)C)=O)C)C)(C)C3=CC=C21 WSTYNZDAOAEEKG-GWJSGULQSA-N 0.000 description 1
- 229960003087 tioguanine Drugs 0.000 description 1
- 208000037816 tissue injury Diseases 0.000 description 1
- 229960001017 tolmetin Drugs 0.000 description 1
- UPSPUYADGBWSHF-UHFFFAOYSA-N tolmetin Chemical compound C1=CC(C)=CC=C1C(=O)C1=CC=C(CC(O)=O)N1C UPSPUYADGBWSHF-UHFFFAOYSA-N 0.000 description 1
- 230000000699 topical effect Effects 0.000 description 1
- ONYVJPZNVCOAFF-UHFFFAOYSA-N topsentin Natural products Oc1ccc2cc([nH]c2c1)C(=O)c3ncc([nH]3)c4c[nH]c5ccccc45 ONYVJPZNVCOAFF-UHFFFAOYSA-N 0.000 description 1
- 231100000331 toxic Toxicity 0.000 description 1
- 230000002588 toxic effect Effects 0.000 description 1
- 239000003053 toxin Substances 0.000 description 1
- 231100000765 toxin Toxicity 0.000 description 1
- 230000009466 transformation Effects 0.000 description 1
- 238000000844 transformation Methods 0.000 description 1
- 229960001727 tretinoin Drugs 0.000 description 1
- AYNNSCRYTDRFCP-UHFFFAOYSA-N triazene Chemical compound NN=N AYNNSCRYTDRFCP-UHFFFAOYSA-N 0.000 description 1
- 229960000538 trimetrexate glucuronate Drugs 0.000 description 1
- LENZDBCJOHFCAS-UHFFFAOYSA-N tris Chemical compound OCC(N)(CO)CO LENZDBCJOHFCAS-UHFFFAOYSA-N 0.000 description 1
- 229910052722 tritium Inorganic materials 0.000 description 1
- ZNRGQMMCGHDTEI-ITGUQSILSA-N tropisetron Chemical compound C1=CC=C2C(C(=O)O[C@H]3C[C@H]4CC[C@@H](C3)N4C)=CNC2=C1 ZNRGQMMCGHDTEI-ITGUQSILSA-N 0.000 description 1
- 229960003688 tropisetron Drugs 0.000 description 1
- 210000004881 tumor cell Anatomy 0.000 description 1
- 229940046728 tumor necrosis factor alpha inhibitor Drugs 0.000 description 1
- 239000002452 tumor necrosis factor alpha inhibitor Substances 0.000 description 1
- WMPQMBUXZHMEFZ-YJPJVVPASA-N turosteride Chemical compound CN([C@@H]1CC2)C(=O)CC[C@]1(C)[C@@H]1[C@@H]2[C@@H]2CC[C@H](C(=O)N(C(C)C)C(=O)NC(C)C)[C@@]2(C)CC1 WMPQMBUXZHMEFZ-YJPJVVPASA-N 0.000 description 1
- 229950007816 turosteride Drugs 0.000 description 1
- 239000005483 tyrosine kinase inhibitor Substances 0.000 description 1
- 150000004917 tyrosine kinase inhibitor derivatives Chemical class 0.000 description 1
- 229950009811 ubenimex Drugs 0.000 description 1
- 231100000397 ulcer Toxicity 0.000 description 1
- 210000003606 umbilical vein Anatomy 0.000 description 1
- 241001430294 unidentified retrovirus Species 0.000 description 1
- 229960001055 uracil mustard Drugs 0.000 description 1
- SPDZFJLQFWSJGA-UHFFFAOYSA-N uredepa Chemical compound C1CN1P(=O)(NC(=O)OCC)N1CC1 SPDZFJLQFWSJGA-UHFFFAOYSA-N 0.000 description 1
- 229950006929 uredepa Drugs 0.000 description 1
- AUFUWRKPQLGTGF-FMKGYKFTSA-N uridine triacetate Chemical compound CC(=O)O[C@@H]1[C@H](OC(C)=O)[C@@H](COC(=O)C)O[C@H]1N1C(=O)NC(=O)C=C1 AUFUWRKPQLGTGF-FMKGYKFTSA-N 0.000 description 1
- 201000005112 urinary bladder cancer Diseases 0.000 description 1
- 210000002700 urine Anatomy 0.000 description 1
- 229960005356 urokinase Drugs 0.000 description 1
- 238000001291 vacuum drying Methods 0.000 description 1
- 229940111506 vaniqa Drugs 0.000 description 1
- 230000002227 vasoactive effect Effects 0.000 description 1
- 229950008261 velaresol Drugs 0.000 description 1
- 229960003048 vinblastine Drugs 0.000 description 1
- KDQAABAKXDWYSZ-JKDPCDLQSA-N vincaleukoblastine sulfate Chemical compound OS(O)(=O)=O.C([N@]1C[C@@H](C[C@]2(C(=O)OC)C=3C(=CC4=C([C@]56[C@H]([C@@]([C@H](OC(C)=O)[C@]7(CC)C=CCN([C@H]67)CC5)(O)C(=O)OC)N4C)C=3)OC)C[C@@](C1)(O)CC)CC1=C2NC2=CC=CC=C12 KDQAABAKXDWYSZ-JKDPCDLQSA-N 0.000 description 1
- AQTQHPDCURKLKT-JKDPCDLQSA-N vincristine sulfate Chemical compound OS(O)(=O)=O.C([C@@H](C[C@]1(C(=O)OC)C=2C(=CC3=C([C@]45[C@H]([C@@]([C@H](OC(C)=O)[C@]6(CC)C=CCN([C@H]56)CC4)(O)C(=O)OC)N3C=O)C=2)OC)C[C@@](C2)(O)CC)N2CCC2=C1NC1=CC=CC=C21 AQTQHPDCURKLKT-JKDPCDLQSA-N 0.000 description 1
- 229960004355 vindesine Drugs 0.000 description 1
- UGGWPQSBPIFKDZ-KOTLKJBCSA-N vindesine Chemical compound C([C@@H](C[C@]1(C(=O)OC)C=2C(=CC3=C([C@]45[C@H]([C@@]([C@H](O)[C@]6(CC)C=CCN([C@H]56)CC4)(O)C(N)=O)N3C)C=2)OC)C[C@@](C2)(O)CC)N2CCC2=C1N=C1[C]2C=CC=C1 UGGWPQSBPIFKDZ-KOTLKJBCSA-N 0.000 description 1
- KLFUUCHXSFIPMH-YBFGSCICSA-N vinepidine Chemical compound C([C@@H](C[C@]1(C(=O)OC)C=2C(=CC3=C([C@]45[C@H]([C@@]([C@H](OC(C)=O)[C@]6(CC)C=CCN([C@H]56)CC4)(O)C(=O)OC)N3C=O)C=2)OC)C[C@@H](C2)CC)N2CCC2=C1NC1=CC=CC=C21 KLFUUCHXSFIPMH-YBFGSCICSA-N 0.000 description 1
- 229950001270 vinepidine Drugs 0.000 description 1
- BOELCLFVOBIXIF-MAKKXPIGSA-N vinfosiltine Chemical compound C([C@@H](C[C@@](O)(CC)C1)C[C@@]2(C3=C(OC)C=C4N(C)[C@H]5[C@@]([C@@H]([C@]6(CC)C=CCN7CC[C@]5([C@H]67)C4=C3)O)(O)C(=O)N[C@@H](P(=O)(OCC)OCC)C(C)C)C(=O)OC)N1CCC1=C2NC2=CC=CC=C12 BOELCLFVOBIXIF-MAKKXPIGSA-N 0.000 description 1
- 229950001739 vinfosiltine Drugs 0.000 description 1
- YNSIUGHLISOIRQ-SWSODSCOSA-N vinglycinate Chemical compound C([C@@H](C[C@]1(C(=O)OC)C=2C(=CC3=C([C@]45[C@H]([C@@]([C@H](OC(=O)CN(C)C)[C@]6(CC)C=CCN([C@H]56)CC4)(O)C(=O)OC)N3C)C=2)OC)C[C@@](C2)(O)CC)N2CCC2=C1NC1=CC=CC=C21 YNSIUGHLISOIRQ-SWSODSCOSA-N 0.000 description 1
- 229950008883 vinglycinate Drugs 0.000 description 1
- 229950009832 vinleurosine Drugs 0.000 description 1
- 229960002066 vinorelbine Drugs 0.000 description 1
- GBABOYUKABKIAF-GHYRFKGUSA-N vinorelbine Chemical compound C1N(CC=2C3=CC=CC=C3NC=22)CC(CC)=C[C@H]1C[C@]2(C(=O)OC)C1=CC([C@]23[C@H]([C@]([C@H](OC(C)=O)[C@]4(CC)C=CCN([C@H]34)CC2)(O)C(=O)OC)N2C)=C2C=C1OC GBABOYUKABKIAF-GHYRFKGUSA-N 0.000 description 1
- 229960002166 vinorelbine tartrate Drugs 0.000 description 1
- GBABOYUKABKIAF-IWWDSPBFSA-N vinorelbinetartrate Chemical compound C1N(CC=2C3=CC=CC=C3NC=22)CC(CC)=C[C@H]1C[C@]2(C(=O)OC)C1=CC(C23[C@H]([C@@]([C@H](OC(C)=O)[C@]4(CC)C=CCN([C@H]34)CC2)(O)C(=O)OC)N2C)=C2C=C1OC GBABOYUKABKIAF-IWWDSPBFSA-N 0.000 description 1
- 229950003670 vinrosidine Drugs 0.000 description 1
- 229950005839 vinzolidine Drugs 0.000 description 1
- 239000011782 vitamin Substances 0.000 description 1
- 235000013343 vitamin Nutrition 0.000 description 1
- 229940088594 vitamin Drugs 0.000 description 1
- 229930003231 vitamin Natural products 0.000 description 1
- 150000003722 vitamin derivatives Chemical class 0.000 description 1
- 238000005303 weighing Methods 0.000 description 1
- 229950005561 zanoterone Drugs 0.000 description 1
- 229950009268 zinostatin Drugs 0.000 description 1
- FYQZGCBXYVWXSP-STTFAQHVSA-N zinostatin stimalamer Chemical compound O1[C@H](C)[C@H](O)[C@H](O)[C@@H](NC)[C@H]1OC1C/2=C/C#C[C@H]3O[C@@]3([C@H]3OC(=O)OC3)C#CC\2=C[C@H]1OC(=O)C1=C(C)C=CC2=C(C)C=C(OC)C=C12 FYQZGCBXYVWXSP-STTFAQHVSA-N 0.000 description 1
- 229950009233 zinostatin stimalamer Drugs 0.000 description 1
- 150000003952 β-lactams Chemical class 0.000 description 1
Images
Landscapes
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
The invention discloses a solid form containing (+)-2-[1-(3-oxethyl-4-methoxyphenyl)-2-methylsulfonylethyl]-4-acetylaminoisoindoline-1,3-dione, a composition containing the solid form, a method for preparing the solid form and an application method thereof. The method comprises a treatment and/or prevention method capable of improving disease symptoms by lowering TNF-alpha level or inhibiting PDE4.
Description
1. invention field
The present invention is to provide and comprise (+)-2-[1-(3-oxyethyl group-4-p-methoxy-phenyl)-2-methyl sulphonyl ethyl]-4-acetylamino isoindoline-1, the solid form of 3-diketone, comprise the compsn of this solid form, the method for preparing the method for this solid form and use their treatment various diseases and/or illness.
2. background of invention
Tumor necrosis factor alpha (TNF-α) is a kind of cytokine that the immunostimulation deposits yields is replied and mainly discharged by MNP that is directed against.TNF-α can strengthen most of cell physiological process, for example breaks up, raises, propagation and proteolytic degradation.When low-level, TNF-α creates antagonism and infects the provide protection of reagent, tumour and tissue injury.But TNF-α also plays a role in numerous disease.When the patient is used, TNF-α can cause or aggravates inflammation, heating, cardiovascular effect, hemorrhage, blood coagulation and with acute infection and shock state during being seen similar acute phase reaction.Numerous disease and medical conditions all relate to enhanced or uncontrolled TNF-α produces, for example, and cancer, for example solid tumor and haematogenous tumour; Heart trouble, for example congestive heart failure; And viral, heredity, inflammatory, supersensitivity and autoimmune disease.
Adenosine 3', 5'-encircle single phosphoric acid (cAMP) and also in numerous disease and illness, play a role, such as but not limited to asthma and inflammation, and other illness (Lowe and Cheng, Drugs of the Future, 17 (9), 799-807,1992).Show that cAMP rising in inflammatory leukocytes can suppress the release subsequently that their activation and inflammatory mediator comprise TNF-α and NF-κ B.The cAMP level raises and also can cause airway smooth muscle lax.
The main cell mechanism of believing the cAMP inactivation is that cAMP is decomposed (Beavo and Reitsnyder, Trends in Pharm., 11,150-155,1990) by one type of isozyme that is called cyclic nucleotide phosphodiesterase (PDE).Existing 11 known PDE families.For example have realized that, suppress PDE IV type and discharge and airway smooth muscle lax among both all effective (Verghese etc., JPharm.Exper.Therapeut., 272 (3), 1313-1320,1995) especially suppressing inflammatory mediator.Therefore, the compound that suppresses PDE4 (PDEIV) is inflammation-inhibiting and help airway smooth muscle lax specifically, makes unwanted spinoff simultaneously, and for example cardiovascular or antiplatelet effect minimizes.The PDE4 suppressor factor that uses at present lacks selective action under acceptable therapeutic dose.
Cancer is a kind ofly to have destructive disease especially, and the ill risk of cancer and diffusion relate to the increase of TNF-α blood level.Usually, in the individuality of health, cancer cells can not be survived in the recycle system, and one of them reason is that blood vessel has played the effect that tumour cell exosmoses that hinders.But, shown the external adhesion that has increased cancer cells and endothelium significantly of being increased in of cytokine levels.A kind of explanation be cytokine for example TNF-α stimulated the biosynthesizing and the expression of the cell surface receptor that is called ELAM-1 (endothelial leukocyte adhesion molecule).ELAM-1 is the member of the Ca-dependent cell adhesion receptor family of the known LEC-CAM of being called, and this family comprises LECAM-1 and GMP-140.During Inflammatory response, the ELAM-1 on the endotheliocyte plays a role as leukocytic " homing receptor ".Recently, show the adherent increase (Rice etc., 1989, Science 246:1303-1306) of ELAM-1 mediation colon cancer cell and the endothelium of handling with cytokine on the endotheliocyte.
Inflammatory diseases for example sacroiliitis, relevant arhritis conditions (for example osteo-arthritis and rheumatoid arthritis), inflammatory bowel (for example segmental enteritis and ulcerative colitis), septicemia, psoriatic, atopic dermatitis, contact dermatitis, chronic obstructive pulmonary disease and chronic inflammation tuberculosis also is a popular difficult and complicated illness widely.TNF-α brings into play main effect in Inflammatory response, and in the animal model of inflammatory diseases, uses its antagonist and hindered chronic and acute reaction.
Viral, heredity, struvite, supersensitivity and autoimmune disease relates to enhanced or uncontrolled TNF-α produces.The instance of such disease includes but not limited to: HIV; Hepatitis; Adult respiratory distress syndrome; Bone resorption disease; Chronic obstructive pulmonary disease; Chronic pulmonary inflammation disease; Asthma; Dermatitis; Cystic fibrosis; Septic shock; Septicemia; Endotoxin shock; The haemodynamics shock; Septicemia syndrome; Postischemic reperfusion damage; Meningitis; Psoriatic; Fibrotic disease; Emaciation; Transplant rejection; Autoimmune disease; Rheumatoid spondylitis; Arhritis conditions, for example rheumatoid arthritis and osteo-arthritis; Osteoporosis; Segmental enteritis; Ulcerative colitis; Inflammatory bowel; Multiple sclerosis; Systemic lupus erythematosus; ENL in the leprosy; Radiation injury; Asthma; And hyperoxic alveolar injury.Tracey etc., 1987, Nature 330:662-664 and Hinshaw etc., 1990, Circ.Shock30:279-292 (endotoxin shock); Dezube etc., 1990, Lancet, 335:662 (emaciation); Millar etc., 1989, Lancet 2:712-714 and Ferrai-Baliviera etc., 1989, Arch.Surg.124:1400-1405 (adult respiratory distress syndrome); Bertolini etc., 1986, Nature 319:516-518, Johnson etc.; 1989, Endocrinology 124:1424-1427, Holler etc., 1990; Blood75:1011-1016 and Grau etc., 1989, N.Engl.J.Med.320:1586-1591 (bone resorption disease); Pignet etc., 1990, Nature, 344:245-247, Bissonnette etc., 1989, Inflammation13:329-339 and Baughman etc., 1990, J.Lab.Clin.Med.115:36-42 (chronic pulmonary inflammation disease); Elliot etc., 1995, Int.J.Pharmac.17:141-145 (rheumatoid arthritis); Von Dullemen etc., 1995, Gastroenterology, 109:129-135 (segmental enteritis); Duh etc., 1989, Proc.Nat.Acad.Sci.86:5974-5978, Poll etc., 1990, Proc.Nat.Acad.Sci.87:782-785; Monto etc., 1990, Blood 79:2670, Clouse etc., 1989, J.Immunol.142; 431-438, Poll etc., 1992, AIDS Res.Hum.Retrovirus, 191-197; PoIi etc. 1990, Proc.Natl.Acad.Sci.87:782-784, Folks etc., 1989, PNAS 86:2365-2368 (opportunistic infection that HIV and HIV cause).
The medical compounds that some cytokine comprises the active of TNF-α or suppress its generation can be blocked and favourable therapeutical agent can be become.Shown that many micromolecular inhibitors have the ability of treatment or the prevention inflammatory diseases relevant with TNF-α (summary referring to Lowe, 1998Exp.Opin.Ther.Patents 8:1309-1332).One type of such molecule is at U.S. Patent number 6,020, the substituted styroyl sulfone of describing in 358.
The variation of considering solid form possibly influence various physics and chemical property; And these character may produce benefit or drawback to processing, preparation, stability and bioavailability and other important medicinal property, so the preparation of the solid form of medical compounds and selection are complicated.The potential medical solid comprises crystalline solid and unformed solid.Unformed solid characteristic is the structural order that lacks long distance, and the characteristic of crystalline solid is a structural cycle property.The required classification of medical solid depends on concrete application; Sometimes based on the for example unformed solid of enhanced dissulution feature selection; And crystalline solid possibly be fit to because of character such as physics or chemicalstability needs (referring to, for example, S.R.Vippagunta etc.; Adv.Drug.Deliv.Rev., (2001) 48:3-26; L.Yu, Adv.Drug.Deliv.Rev., (2001) 48:27-42).
No matter be crystallization or unformed, the potential solid form of medical compounds comprises single component and multicomponent solid.The single component solid is made up of medical compounds basically, does not contain other compound.Mutation can appear potentially in the single component crystalline material, polymorphism for example, in polymorphism the certain drugs compound exist multiple three-dimensional arrangement (referring to, for example, S.R.Byrn etc.,
Solid State Chemistry of Drugs, (1999) SSCI, West Lafayette).Ritonavir, a kind of hiv protease suppressor factor that is configured to soft gelatin capsule, example stressed the importance of research polymorphic form.After this product was gone into operation about 2 years; In preparation a kind of unanticipated of new polymorphic form than indissoluble to deposition force product to be recalled from market; Up to developing a kind of preparation with consistency (referring to S.R.Chemburkar etc.; Org.Process Res.Dev., (2000) 4:413-417).
The variety that can occur other in the potential solid form of medical compounds, for example, multicomponent solid possibility.The crystalline solid that comprises two or more ionic speciess can be known as salt (referring to for example,
Handbook of Pharmaceutical Salts:Properties, Selection and Use, P.H.Stahl and C.G.Wermuth edit, and (2002), Wiley, Weinheim).For example can comprise for the multicomponent solid additional type that medical compounds or its salt provide other character to improve potentially hydrate, solvolyte, eutectic and inclusion compound etc. (referring to for example, S.R.Byrn etc.,
Solid State Chemistry of Drugs, (1999) SSCI, West Lafayette).In addition, the multicomponent crystal formation can be easy to become polymorphic potentially, and wherein specified multicomponent compsn can multiple three-dimensional crystals be arranged existence.Solid form be prepared in exploitation safety, effectively, be extremely important in the stable and salable medical compounds.
The present invention is to provide that to meet chemical name be (+)-2-[1-(3-oxyethyl group-4-p-methoxy-phenyl)-2-methyl sulphonyl ethyl]-4-acetylamino isoindoline-1; The embodiment of the demand of the solid form of the compound of 3-diketone (" compd A "); This compound is disclosed in the U. S. application submitted on March 19th, 2003 number 10/392; The U.S. Provisional Application series number 60/366 that 195 (as U.S. Patent number 6,962,940 are authorized to) and on March 20th, 2002 submit to; Submitted on January 7th, 515 and 2003 60/438,450 in.
3. summary of the invention
The present invention relates to utilize the enantiomorph of substituted styroyl sulphones and the method for pharmaceutically acceptable solvolyte, hydrate, eutectic, inclusion compound, prodrug and polymorphic form treatment disease and illness thereof, and the method that in Mammals, reduces cytokine and precursor level thereof.The present invention also relates to pharmaceutical composition, it contains 2-[1-(3-oxyethyl group-4-p-methoxy-phenyl)-2-methyl sulphonyl ethyl]-4-acetylamino isoindoline-1, (+) enantiomorph of 3-diketone and pharmaceutically acceptable carrier.The invention further relates to 2-[1-(3-oxyethyl group-4-p-methoxy-phenyl)-2-methyl sulphonyl the ethyl]-4-acetylamino isoindoline-1 that does not contain its (-) enantiomorph basically, (+) enantiomorph of 3-diketone.
The present invention relates to 2-[1-(3-oxyethyl group-4-p-methoxy-phenyl)-2-methyl sulphonyl ethyl]-4-acetylamino isoindoline-1, (+) enantiomorph of 3-diketone particularly.Believe this compound and its racemoid 2-[1-(3-oxyethyl group-4-p-methoxy-phenyl)-2-methyl sulphonyl ethyl]-4-acetylamino isoindoline-1, the 3-diketone is compared the effectiveness and other benefit with increase.
The present invention includes 2-[1-(3-oxyethyl group-4-p-methoxy-phenyl)-2-methyl sulphonyl ethyl]-4-acetylamino isoindoline-1, (+) enantiomorph of 3-diketone is used for can producing the disease improved or the purposes of illness through suppressing TNF-α in Mammals treatment or prevention.In certain embodiments, this treatment comprises minimizing or avoids undesirable action.This illness includes but not limited to cancer, includes but not limited to: the cancer of head, Tiroidina, neck, eye, skin, mouth, larynx, esophagus, chest, bone, blood, marrow, lung, colon, sigmoid colon, rectum, stomach, prostate gland, breast, ovary, kidney, liver, pancreas, brain, intestines, heart, suprarenal gland, subcutis, lymphoglandula, heart and combination thereof.Treatable by this method concrete cancer has multiple myeloma, malignant melanoma, glioblastoma, white blood disease and solid tumor.
The present invention also comprises 2-[1-(3-oxyethyl group-4-p-methoxy-phenyl)-2-methyl sulphonyl ethyl]-4-acetylamino isoindoline-1; (+) enantiomorph of 3-diketone includes but not limited to: the purposes in the septic shock of congestive heart failure, myocardosis, wet lung, endotaxin mediate, acute viral myocarditis, heart heteroplastic transplantation repulsion and the myocardial infarction at treatment or preventing heart disease.
The present invention also comprises use 2-[1-(3-oxyethyl group-4-p-methoxy-phenyl)-2-methyl sulphonyl ethyl]-4-acetylamino isoindoline-1, and (+) enantiomorph of 3-diketone is treated can be through suppressing disease or the illness that PDE4 improves.For example, compound of the present invention and compsn can be used for treatment or prevent viral, heredity, struvite, supersensitivity and autoimmune disease.The instance of such disease includes but not limited to: HIV; Hepatitis; Adult respiratory distress syndrome; Bone resorption disease; Chronic obstructive pulmonary disease; Chronic pulmonary inflammation disease; Dermatitis; Inflammatory skin diseases, atopic dermatitis, cystic fibrosis; Septic shock; Septicemia; Endotoxin shock; The haemodynamics shock; Septicemia syndrome; Postischemic reperfusion damage; Meningitis; Psoriatic; Fibrotic disease; Emaciation; Transplant rejection comprises graft versus host disease; Autoimmune disease; Rheumatoid spondylitis; Arhritis conditions, for example rheumatoid arthritis and osteo-arthritis; Osteoporosis; Segmental enteritis; Ulcerative colitis; Inflammatory bowel; Multiple sclerosis; Systemic lupus erythematosus; ENL in the leprosy (ENL); Radiation injury; Asthma; And hyperoxic alveolar injury.
In yet another embodiment; 2-[1-(3-oxyethyl group-4-p-methoxy-phenyl)-2-methyl sulphonyl ethyl]-4-acetylamino isoindoline-1; Pure (+) enantiomorph of the stereoisomerism of 3-diketone also is useful in the symptom of treatment or prophylaxis of microbial infection or infected by microbes, includes but not limited to the opportunistic infection that infectation of bacteria, fungi infestation, malaria, mycobacterial infections and HIV cause.
The present invention further comprises pharmaceutical composition and single unit dosage; It comprises 2-[1-(3-oxyethyl group-4-p-methoxy-phenyl)-2-methyl sulphonyl ethyl]-4-acetylamino isoindoline-1, (+) enantiomorph of 3-diketone and pharmaceutically acceptable polymorphic form, prodrug, hydrate, inclusion compound and solvolyte.
In a separate embodiments, the present invention includes 2-[1-(3-oxyethyl group-4-p-methoxy-phenyl)-2-methyl sulphonyl ethyl]-4-acetylamino isoindoline-1, (+) enantiomorph of 3-diketone.
In a further embodiment; The present invention includes preparation 2-[1-(3-oxyethyl group-4-p-methoxy-phenyl)-2-methyl sulphonyl ethyl]-4-acetylamino isoindoline-1; The method of (+) enantiomorph that the stereoisomerism of 3-diketone is pure, it comprises makes 1-(3-oxyethyl group-4-methoxyl group-phenyl)-2-methane sulfonyl-ethamine contact with chiral amino acid, and makes the product and the N-(1 of the first step; 3-dioxy-1,3-dihydro-isobenzofuran-4-yl)-the ethanamide contact.In the embodiment that is correlated with, the present invention includes the chirality salt of 1-(3-oxyethyl group-4-methoxyl group-phenyl)-2-methane sulfonyl-ethamine.
Embodiment of the present invention provide solid form, and it comprises chemical name is (+)-2-[1-(3-oxyethyl group-4-p-methoxy-phenyl)-2-methyl sulphonyl ethyl]-4-acetylamino isoindoline-1, the compound of 3-diketone (" compd A ").Compd A can according to any method that it will be apparent to those skilled in the art, be included in the synthetic or acquisition of the method for describing among the following embodiment based on the instruction of this paper.Compd A also may be in accordance with the method for describing in the U.S. Patent number 6,962,940 of authorizing on November 8th, 2005 and prepares, and its its full text is incorporated this paper by reference into.
In certain embodiments, solid form is the single component crystal formation of compd A.In certain embodiments, solid form is the multicomponent crystal formation, includes but not limited to eutectic and/or the solvolyte (comprising hydrate) of inclusion compound A.In other embodiments, solid form is the single component amorphous forms of compd A.In other embodiments, solid form is the multicomponent amorphous forms.Do not hope to be subject to any particular theory; Some novel solid form provided by the invention has specific favourable physics and/or chemical property; Make it to for example preparing, process, prepare and/or storing useful; Simultaneously also has particularly advantageous biological property, for example like bioavailability and/or biological activity.
In specific embodiments, solid form provided by the invention comprises the solid form of inclusion compound A, includes but not limited to single component and the multicomponent solid form of inclusion compound A.In certain embodiments, solid form provided by the invention comprises polymorphic form, solvolyte (comprising hydrate) and the eutectic of inclusion compound A.Certain embodiments of the present invention provide preparation, separation and/or have characterized the method for solid form provided by the invention.
Solid form provided by the invention can be used as active pharmaceutical ingredient and is used for being prepared in the preparation that the patient uses.Therefore, embodiment of the present invention comprise the purposes of these solid forms as final medicament prodn.Some embodiment provides useful and character that have improvement in the final formulation of preparation, for example preparations such as the compatible character of flow of powder character, compacting character, compressing tablet character, stable character and vehicle, processes, prepares and/or store the solid form of the needed character of final medicament prodn.Certain embodiments of the present invention provide pharmaceutical composition; It contains single component crystal formation, multicomponent crystal formation, single component amorphous forms and/or the multicomponent amorphous forms of inclusion compound A, and pharmaceutically acceptable thinner, vehicle or carrier.Solid form provided by the invention can be used for for example treating, prevents or controls disease provided by the present invention and illness with final medicament prodn.
Certain embodiments of the present invention provide uses solid form provided by the present invention in Mammals, to treat, prevent or control and can produce the disease improved or the method for illness through suppressing TNF-α, for example: HIV; Hepatitis; Adult respiratory distress syndrome; Bone resorption disease; Chronic obstructive pulmonary disease; Chronic pulmonary inflammation disease; Asthma; Dermatitis; Cystic fibrosis; Septic shock; Septicemia; Endotoxin shock; The haemodynamics shock; Septicemia syndrome; Postischemic reperfusion damage; Meningitis; Psoriatic; Fibrotic disease; Emaciation; Transplant rejection; Autoimmune disease; Rheumatoid spondylitis; Arhritis conditions, for example psoriatic arthritis, rheumatoid arthritis and osteo-arthritis; Osteoporosis; Segmental enteritis; Ulcerative colitis; Inflammatory bowel; Multiple sclerosis; Systemic lupus erythematosus; Lupus erythematosus,cutaneous; Sarcoidosis of lung; ENL in the leprosy; Radiation injury; Asthma; And hyperoxic alveolar injury.This illness further includes but not limited to cancer, includes but not limited to: the cancer of head, Tiroidina, neck, eye, skin, mouth, larynx, esophagus, chest, bone, blood, marrow, lung, colon, sigmoid colon, rectum, stomach, prostate gland, breast, ovary, kidney, liver, pancreas, brain, intestines, heart, suprarenal gland, subcutis, lymphoglandula, heart and combination thereof.Treatable by this method concrete cancer has multiple myeloma, malignant melanoma, glioblastoma, white blood disease and solid tumor.In certain embodiments, use the method for solid form provided by the present invention to comprise minimizing or avoid some undesirable action.
Certain embodiments of the present invention provide the method for in cardiopathic treatment or prevention, using solid form provided by the present invention, include but not limited to: the septic shock of congestive heart failure, myocardosis, wet lung, endotaxin mediate, acute viral myocarditis, heart heteroplastic transplantation repulsion and myocardial infarction.
Certain embodiments of the present invention provide use solid form provided by the present invention to treat can be through suppressing the disease that PDE4 improves or the method for illness.For example, solid form provided by the present invention can be used for treatment or prevents viral, heredity, struvite, supersensitivity and autoimmune disease.The instance of this disease includes but not limited to: HIV; Hepatitis; Adult respiratory distress syndrome; Bone resorption disease; Chronic obstructive pulmonary disease; Chronic pulmonary inflammation disease; Dermatitis; Inflammatory skin diseases; Atopic dermatitis; Cystic fibrosis; Septic shock; Septicemia; Endotoxin shock; The haemodynamics shock; Septicemia syndrome; Postischemic reperfusion damage; Meningitis; Psoriatic; Fibrotic disease; Emaciation; Transplant rejection comprises graft versus host disease; Autoimmune disease; Rheumatoid spondylitis; Arhritis conditions, for example rheumatoid arthritis and osteo-arthritis; Osteoporosis; Segmental enteritis; Ulcerative colitis; Inflammatory bowel; Multiple sclerosis; Systemic lupus erythematosus; ENL in the leprosy (ENL); Radiation injury; Asthma; And hyperoxic alveolar injury.
Certain embodiments of the present invention provide the method for in the symptom of treatment or prophylaxis of microbial infection or infected by microbes, using solid form provided by the present invention, include but not limited to the opportunistic infection that infectation of bacteria, fungi infestation, malaria, mycobacterial infections and HIV cause.
Specific embodiments of the present invention provides the method for in treatment of diseases or prevention, using solid form provided by the present invention, and these diseases comprise: psoriatic, psoriatic arthritis, rheumatoid arthritis, chronic skin sarcoid, giant cell arteritis, parkinsonism, prurigo nodularis, lichen planus, complicacy aphthosis, Behcet's disease, lupus, hepatitis, uveitis, sjogren syndrome, depression (comprising major depression), interstitial cystitis, vulvodynia, prostatitis, osteo-arthritis, diffuse large B cell lymphoma, polymyositis, dermatomyositis, inclusion body myositis, EOA, interstitial cystitis, hepatitis, endometriosis, radiculopathy and pyoderma gangraenosum.
Certain embodiments of the present invention provide pharmaceutical composition and the single unit dosage that comprises one or more solid forms provided by the present invention.
3.1 brief description of the drawings
Fig. 1 provides exemplary x-ray powdery diffractometry (" the XRPD ") figure of the form A of compd A.
Fig. 2 provides typical dsc (" the DSC ") figure of the form A of compd A.
Fig. 3 provides typical thermogravimetic analysis (TGA) (" the TGA ") figure of the form A of compd A.
Fig. 4 provides exemplary dynamic Gas Phase Adsorption (" the DVS ") figure of the form A of compd A.
Fig. 5 provides the typical XRPD figure of the form B of compd A.
Fig. 6 provides the typical DSC figure of the form B of compd A.
Fig. 7 provides the typical TGA figure of the form B of compd A.
Fig. 8 provides the typical DVS figure of the form B of compd A.
Fig. 9 provides the typical XRPD figure of the form A of compd A.
Figure 10 provides the typical DSC figure of the form A of compd A.
Figure 11 provides the typical TGA figure of the form A of compd A.
Figure 12 provides the typical DVS figure of the form A of compd A.
Figure 13 provides the typical XRPD figure of the form D of compd A.
Figure 14 provides the typical DSC figure of the form D of compd A.
Figure 15 provides the typical TGA figure of the form D of compd A.
Figure 16 provides the typical DVS figure of the form D of compd A.
Figure 17 provides the typical XRPD figure of the form E of compd A.
Figure 18 provides the typical DSC figure of the form E of compd A.
Figure 19 provides the typical TGA figure of the form E of compd A.
Figure 20 provides the typical DVS figure of the form E of compd A.
Figure 21 provides the typical XRPD figure of the form D of compd A.
Figure 22 provides the typical DSC figure of the form D of compd A.
Figure 23 provides the typical TGA figure of the form D of compd A.
Figure 24 provides the typical DVS figure of the form D of compd A.
Figure 25 provides the typical XRPD of the form G of compd A.
Figure 26 provides the typical DSC figure of the form G of compd A.
Figure 27 provides the typical TGA figure of the form G of compd A.
Figure 28 provides the typical DVS figure of the form G of compd A.
Figure 29 has shown 2-[1-(3-oxyethyl group-4-p-methoxy-phenyl)-2-methyl sulphonyl ethyl]-4-acetylamino isoindoline-1, the preparation of (+) enantiomorph of 3-diketone.
Figure 30 has shown that compd A is to the effect of LPS inductive neutrophilia in the lung of clear-headed ferret.
Figure 31 has shown in the clinical study of assessment compd A in the patient who suffers from the severe psoriasis in plaques, in the percentage change of the 29th day whole 15 individual mesocuticle thickness.
Figure 32 has shown in the clinical study of assessment compd A in the patient who suffers from the severe psoriasis in plaques, the variation of average iNOS (standardized hARP) in the biopsy specimen of the 29th day infringement skin.
Figure 33 shown in the patient who suffers from the severe psoriasis in plaques in the clinical study of assessment compd A, and mark from the percentage change of baseline with severity index (PASI) in total psoriatic zone in the 29th day valuable patient.
3.2
Definition
The term " compd A " that the present invention uses refers to (+)-2-[1-(3-oxyethyl group-4-p-methoxy-phenyl)-2-methyl sulphonyl the ethyl]-4-acetylamino isoindoline-1 of enantiomer-pure; The 3-diketone; When post is 150mm * 4.6mm Ultron chirality ES-OVS chirality HPLC post (Agilent Technology), elutriant is the 15:85 ethanol of pH 3.5: 20mM KH
2PO
4, and the observation wavelength is when being 240nm, it separated from this HPLC post at about 25.4 minutes.Compd A
1H NMR spectrum is following basically: δ (CDCl
3); 1.47 (t, 3H); 2.26 (s, 3H); 2.87 (s, 3H); 3.68-3.75 (dd, 1H); 3.85 (s, 3H); 4.07-4.15 (q, 2H); 4.51-4.61 (dd, 1H); 5.84-5.90 (dd, 1H); 6.82-8.77 (m, 6H); 9.46 (s, 1H).Compd A
13C NMR spectrum is following basically: δ (DMSO-d
6); 14.66; 24.92; 41.61; 48.53; 54.46; 55.91; 64.51; 111.44; 112.40; 115.10; 118.20; 120.28; 124.94; 129.22; 131.02; 136.09; 137.60; 148.62; 149.74; 167.46; 169.14; 169.48.Be dissolved in compd A in the methyl alcohol to (+) direction Plane of rotation polarized light.
Not being subject to theory, believing that compd A is S-{2-[1-(3-oxyethyl group-4-p-methoxy-phenyl)-2-methyl sulphonyl ethyl]-4-acetylamino isoindoline-1, the 3-diketone, it has following structure:
The term " patient " that the present invention uses refers to Mammals, particularly people.
The term " pharmacy acceptable salt " that the present invention uses refers to by pharmaceutically acceptable non-toxic acid or alkali, comprises the salt of inorganic bronsted lowry acids and bases bronsted lowry and organic bronsted lowry acids and bases bronsted lowry preparation.
Only if point out in addition, the term " prodrug " that the present invention uses refers to the verivate of compound, and it can hydrolysis, oxidation or otherwise react so that this compound to be provided down at biotic condition (external or body in).But the instance of prodrug includes but not limited to the part that comprises biological hydrolysis of compd A, but but but but but but the for example verivate and the metabolite of the phosphoric acid salt analogue of the uride of the carbonic ether biological hydrolysis of the carbamate biological hydrolysis of the ester biological hydrolysis of the acid amides biological hydrolysis of biological hydrolysis and biological hydrolysis.Prodrug can use the method preparation of knowing usually, Burger ' s Medicinal Chemistry and Drug Discovery for example, 172-178, those methods of 949-982 (Manfred E.Wolff edit, the 5th edition 1995) description.
Only if point out in addition; Term " but acid amides of biological hydrolysis ", " but ester of biological hydrolysis ", " but carbamate of biological hydrolysis ", " but carbonic ether of biological hydrolysis ", " but uride of biological hydrolysis ", " but phosphoric acid salt of biological hydrolysis " that the present invention uses refers to acid amides, ester, carbamate, carbonic ether, uride or the phosphoric acid salt of compound respectively; It is 1 years old) biological activity of interfering compound not; But can give this compound advantageous feature in vivo, that for example absorbs, acts on continues or effect initial; Or 2) not bioactive, but convert bioactive compounds in vivo to.But the instance of the ester of biological hydrolysis includes but not limited to: lower alkyl esters, alkoxyl group acyloxyate, alkyl acyl aminoalkyl ester and cholinesterase.But the instance of the acid amides of biological hydrolysis includes but not limited to low alkyl group acid amides, alpha-amino acid amides, alkoxyl group acyl group acid amides and alkylamino alkyl-carbonyl acid amides.But the instance of the carbamate of biological hydrolysis includes but not limited to: low-grade alkylamine, substituted quadrol, amino acid, hydroxyalkyl amine, heterocycle and aromatic heterocycle amine and polyetheramine.
Only if point out in addition, the term " stereoisomerism is pure " that the present invention uses refers to a kind of steric isomer of compsn inclusion compound and does not contain other steric isomer of this compound basically.For example, the pure compsn of stereoisomerism that has the compound of a chiral centre is incited somebody to action the opposite enantiomorph that will not contain this compound basically.The pure compsn of stereoisomerism with compound of two chiral centres will not contain other diastereomer of this compound basically.The typical pure compound of stereoisomerism comprises greater than a kind of steric isomer of this compound of about 80% weight with less than other steric isomer of this compound of about 20% weight; More preferably greater than a kind of steric isomer of this compound of about 90% weight with less than other steric isomer of this compound of about 10% weight; Even more preferably greater than a kind of steric isomer of this compound of about 95% weight with less than other steric isomer of this compound of about 5% weight with most preferably greater than a kind of steric isomer of this compound of about 97% weight with less than other steric isomer of this compound of about 3% weight.
Only if point out in addition, the term " enantiomer-pure " that the present invention uses refers to have the pure compsn of stereoisomerism of the compound of a chiral centre.
The term " undesirable action " that the present invention uses includes but not limited to stomach and intestine, kidney and hepatotoxicity, oligoleukocythemia, and the bleeding time that causes owing to for example thrombopenia increases, and gestation prolongs; Feel sick, vomiting, drowsiness; Weakness, dizzy, teratogenesis; Extrapyramidal sign is cathisophobiaed, and cardiac toxic comprises that cardiovascular disorder, inflammation, male sexual disorder and serum liver enzyme level raise.Term " gastrointestinal toxicity " includes but not limited to gastroenteritic ulcer and erosion.Term " Toxicity of Kidney " includes but not limited to situation for example papillary necrosis and chronic interstitial nephritis.
Only if point out in addition, the phrase that the present invention uses " reduce or avoid undesirable action " refers to reduce the severity that one or more the present invention lock the undesirable action of justice.
Should be noted that if exist inconsistently between structure of describing and the title that this structure is provided, then be as the criterion with the structure of describing.In addition, the stereochemistry of if structure or structure division for example thick line of no use or dotted line represent that then this structure or structure division should be understood to include its all steric isomers.
Unless otherwise, the term " solid form " and the relational language of the present invention's use refer to not to be mainly to be in liquid state or gasiform physical form.Unless otherwise, term " solid form " that the present invention uses and relational language refer to not to be the physical form that mainly is in liquid state or gasiform inclusion compound A when being used to refer to compd A in the present invention.Solid form can be crystallization, unformed or its mixture.In specific embodiments, solid form can be a liquid crystals." single component " solid form of inclusion compound A is made up of compd A basically." multicomponent " solid form of inclusion compound A is included in the one or more extra kind of the significant quantity in the solid form, for example ion and/or molecule.For example, in specific embodiments, the crystallization multicomponent solid form of inclusion compound A further comprises one or more kinds of position clocklike that are bonded in non-covalently in lattice.The multicomponent solid form of inclusion compound A comprises eutectic, solvolyte (for example hydrate) and the inclusion compound of compd A.In specific embodiments, term " solid form of inclusion compound A " and relational language comprise single component and the multicomponent solid form of inclusion compound A.In specific embodiments, " solid form of inclusion compound A " and relational language comprise crystal formation, the inclusion compound A of inclusion compound A amorphous forms, and composition thereof.
Unless otherwise; The relational language that the term " crystallization " that the present invention uses and the present invention use; When being used for describing compound, material, modification, material, composition or product; Unless otherwise, refer to that it is crystallization that this compound, material, modification, material, composition or product are measured basic through X-ray diffraction.Referring to, for example,
Remington:The Science and Practice of Pharmacy, the 21st edition, Lippincott, Williams and Wilkins, Baltimore, MD (2005);
The United States Pharmacopeia, the 23rd edition, 1843-1844 (1995).
Unless otherwise, the relational language of term " crystal formation ", " crystallized form " and this paper that the present invention uses refers to the crystalline solid form.Crystal formation comprises single component crystal formation and multicomponent crystal formation, and includes but not limited to: polymorphic form, solvolyte, hydrate and/or other molecular complex.In certain embodiments, the crystal formation of material can not contain amorphous forms and/or other crystal formation basically.In certain embodiments, the crystal formation of material can comprise one or more amorphous forms and/or other crystal formation of about 1%, 2%, 3%, 4%, 5%, 6%, 7%, 8%, 9%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45% or 50% weight.In certain embodiments, the crystal formation of material can be a physics and/or chemical pure.In certain embodiments, the crystal formation of material can be about 99%, 98%, 97%, 96%, 95%, 94%, 93%, 92%, 91% or 90% physics and/or chemical pure.
Unless otherwise, the relational language of term " polymorphic form ", " polymorphic forms " and this paper of the present invention's use refers to basic two or more crystal formations of being made up of same molecular and/or ion.The same with different crystal formations, as molecule and/or ion is arranged in lattice or the result of conformation, different polymorphic forms can have different physical propertiess, for example like temperature of fusion, melting heat, solubleness, dissulution character and/or vibrational spectrum.The difference of physical properties can influence drug parameters, for example package stability, compressibility and density (very important in prescription and product prepn) and dissolution rate (important factor in the bioavailability).The difference of stability can by chemically reactive variation (variable color quickly when for example different oxidation makes formulation ratio comprises another kind of polymorphic form when comprising a kind of polymorphic form) or metataxis (for example; Because the preferred polymorphic form of kinetics changes into the more stable polymorphic form of thermodynamics; Tablet bursts apart in storage) or both (for example, a kind of tablet of polymorphic form is easier to decompose in high humidity) cause.As the result of solubleness/dissulution difference, under extreme case, some solid state transformations possibly cause rendeing a service disappearance, perhaps, are in that another is extreme, cause toxicity.In addition, the work in-process physical properties possibly be important (for example, a kind of polymorphic form possibly form solvolyte more easily, maybe possibly be not easy to filter and wash out impurity, and particle shape and size-grade distribution possibly be different between polymorphic form).
Unless otherwise, the term " solvolyte " that uses of the present invention and " solvation " crystal formation that comprises solvent that refers to material.Term " hydrate " and " hydration " refer to the wherein aqueous solvolyte of solvent package." polymorphic form of solvolyte " refers to that there is multiple crystal formation in specific solvolyte compsn.Similarly, " polymorphic form of hydrate " refers to that there is multiple crystal formation in specific hydrate compositions.The term " desolvated solvolyte " that the present invention uses refers to can be through removing the crystal formation of the material that solvent prepares from solvolyte.
Unless otherwise, the relational language that the term " unformed " that the present invention uses, " amorphous forms " and the present invention use refers to that it is not crystallization basically that material, composition or the product referred to are measured through X-ray diffraction.Particularly, unordered solid form described in term " amorphous forms ", promptly lacks the solid form of the order of crystallization of long distance.In certain embodiments, the amorphous forms of material can not contain other amorphous forms and/or crystal formation basically.In other embodiments, the amorphous forms of material can comprise one or more other amorphous forms and/or crystal formations that are lower than about 1%, 2%, 3%, 4%, 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45% or 50% weight.In certain embodiments, the amorphous forms of material can be a physics and/or chemical pure.In certain embodiments, the amorphous forms of material is about 99%, 98%, 97%, 96%, 95%, 94%, 93%, 92%, 91% or 90% physics and/or chemical pure.
The technology that supplies to characterize crystal formation and amorphous forms includes but not limited to: thermogravimetic analysis (TGA) (TGA); Dsc (DSC); X-ray powder diffraction (XRPD); Single crystal method of X-ray diffractometry; Vibrational spectrum for example infrared (IR) and Raman spectrum; Solid-state and solution nucleus magnetic resonance (NMR) spectrum; Opticmicroscope; Hot platform opticmicroscope; Sem (SEM); Electron crystallography and quantitative analysis; Sreen analysis (PSA); Surface-area is analyzed; Solubleness is measured; Dissulution is measured; Ultimate analysis and Karl Fischer analyze.Distinctive unit cell parameters can be used one or more technical measurements, such as but not limited to X-ray diffraction and neutron diffraction, comprises single crystal diffraction and powdery diffractometry.The technology that can be used for the analysed for powder diffraction data comprises the profile refine, for example Rietveld refine, and it can be used for for example comprising above analyzing single relevant diffraction peak in a kind of sample of solid phase.Other method that can be used for the analysed for powder diffraction data comprises the structure cell retrieval, and it allows those skilled in the art to measure unit cell parameters from the sample that comprises crystalline powder.
Unless otherwise; The term " about " that the present invention uses is with " approximately " and numerical value or the value scope that characterizes the particular solid form is provided; For example actual temp or TR; For example as describe DSC or TGA incident heat, for example comprise melt, the actual temp or the TR of dehydration, desolvation or glass transition incident; Quality change is for example as the quality change of the function of temperature or humidity; The content of solvent or water, unit is for example quality or per-cent; Or the peak position, for example the peak position in analyzing through IR or Raman spectrum or XRPD links to each other when using, and refers to that this value or value scope can depart from those of ordinary skills and think reasonable range, but still describes this particular solid form.For example; In specific embodiments; Unless otherwise; The term " about " of in this context, using and " approximately ", refer to this numerical value or value scope can the value of statement or value scope 25%, 20%, 15%, 10%, 9%, 8%, 7%, 6%, 5%, 4%, 3%, 2%, 1.5%, 1%, 0.5% or 0.25% in variation.
Unless otherwise; The specific crystal formation that comprises " pure basically " (for example not containing other solid form and/or other chemical cpd basically) that the present invention uses or the sample of amorphous forms; In specific embodiments, comprise one or more other solid forms and/or other chemical cpd that weight ratio is lower than about 25%, 20%, 15%, 10%, 9%, 8%, 7%, 6%, 5%, 4%, 3%, 2%, 1%, 0.75%, 0.5%, 0.25% or 0.1% per-cent.
Unless otherwise; " not containing basically " one or more other solid forms that the present invention uses and/or the sample or the compsn of other chemical cpd; Refer to that said composition comprises; In specific embodiments, weight ratio is lower than one or more other solid forms and/or other chemical cpd of about 25%, 20%, 15%, 10%, 9%, 8%, 7%, 6%, 5%, 4%, 3%, 2%, 1%, 0.75%, 0.5%, 0.25% or 0.1% per-cent.
Unless otherwise, the term " treatment " that uses of the present invention refers to eradicate or improves one or more symptoms that disease or illness or this disease or illness are correlated with.In certain embodiments, this term points to the patient who suffers from such disease or illness and uses one or more preventions or therapeutical agent so that the diffusion of this disease or illness or deterioration minimize.In some embodiments, this term refers to behind the paresthesia epilepsy of specified disease, follows or do not follow other extra promoting agent, uses compound provided by the present invention.
Unless otherwise, the term " prevention " of the present invention's use refers to outbreak, recurrence or the diffusion of preventing disease or illness or its one or more symptoms.In certain embodiments, this term refers to before paresthesia epilepsy, particularly to having the patient of disease provided by the present invention or illness risk, follows or do not follow other extra active compound, uses compound provided by the present invention or treats with it.This term comprises the symptom that suppresses or alleviate specified disease.In certain embodiments, the patient who especially has a family history of disease is the candidate of prevention scheme.In addition, the patient who has a symptomatic recurrence history also is the potential candidate of prevention.Just in this point, term " prevention " can exchange with term " prophylactic treatment " and use.
Unless otherwise, the term " control " of the present invention's use refers to prevent or slow down progress, diffusion or the deterioration of disease or illness or its one or more symptoms.In most cases, the patient can not make disease or illness cure from the beneficial effect that prevents and/or treats agent and obtain.Just in this point, term " control " comprises the patient that treatment once suffered from specified disease, to attempt to prevent or minimize the recurrence of disease.
Unless otherwise, " the treatment significant quantity " of the compound that the present invention uses is in disease or treatment of conditions or control, to be enough to provide the treatment benefit, perhaps postpones or minimize the amount of one or more relevant symptoms of disease or illness.The treatment significant quantity of compound refers to the amount of therapeutical agent, and it is united separately or with other treatment, in disease or treatment of conditions or control, the treatment benefit is provided.Term " treatment significant quantity " can comprise improvement totally to be treated, reduces or avoid symptom or the cause of disease or illness, or strengthen the amount of the result of treatment of another kind of therapeutical agent.
Unless otherwise, " the prevention significant quantity " of the compound of the present invention's use is the amount that is enough to preventing disease or illness or prevents its recurrence.The prevention significant quantity of compound refers to the amount of therapeutical agent, and it is united separately or with other medicaments, and the prevention benefit is provided in the prevention of disease.Term " prevention significant quantity " can comprise the amount that improvement totally prevented or strengthened the preventive effect of another kind of preventive.
The term " compsn " that the present invention uses will comprise and contain the product of specifying composition (if point out, containing with specified amount), and any directly or indirectly by the product of specifying composition to produce with the specified amount combination." pharmaceutically acceptable " is meant that thinner, vehicle or carrier must be compatible with other composition of preparation, and harmless to its recipient.
4. detailed description of the Invention
The present invention relates to the pure compd A of stereoisomerism; It is 2-[1-(3-oxyethyl group-4-p-methoxy-phenyl)-2-methyl sulphonyl ethyl]-4-acetylamino isoindoline-1; (+) enantiomorph of 3-diketone; Basically do not contain its (-) enantiomorph, and the novel method and the compsn that comprises it of using the solid form of pure compd A of stereoisomerism and/or inclusion compound A.For example, the present invention includes interior use of external and body of compd A, and compd A is joined in the pharmaceutical composition and single unit dosage useful in various diseases and treatment of conditions and prevention.Can be well known in the art through disease and the illness that reduction TNF-alpha levels or inhibition PDE4 improve, and description is arranged in the present invention.Concrete grammar of the present invention reduces or has avoided as the relevant undesirable action of the compound of TNF-alpha inhibitor.Other concrete grammar of the present invention reduces or has avoided using racemic 2-[1-(3-oxyethyl group-4-p-methoxy-phenyl)-2-methyl sulphonyl ethyl]-4-acetylamino isoindoline-1, the undesirable action that the 3-diketone is relevant.
Concrete grammar of the present invention comprises treatment or preventing disease and illness, includes but not limited to the method for solid tumor, haematogenous tumour and inflammatory diseases.
The medicine of the present invention and the formulation of inclusion compound A or its pharmaceutically acceptable polymorphic form, prodrug, inclusion compound, solvolyte or hydrate (wherein specific embodiments comprises the solid form like inclusion compound A described in the invention) can be used in the method for the invention.
Be not subject to theory, believe compd A, comprise the solid form of inclusion compound A, can suppress TNF-α and produce.Therefore; First embodiment of the present invention relates to and suppresses the method that TNF-α produces, and it comprises makes the cell that shows abnormal T NF-α generation contact with pure compd A or its pharmaceutically acceptable prodrug, metabolite, polymorphic form, solvolyte, hydrate or the inclusion compound (wherein specific embodiments comprises the solid form like inclusion compound A described in the invention) of the stereoisomerism of significant quantity.In a specific embodiments; The present invention relates to suppress the method that TNF-α produces, it comprises makes the mammalian cell that shows abnormal T NF-α generation contact with pure compd A or its pharmaceutically acceptable prodrug, metabolite, polymorphic form, solvolyte, hydrate or the inclusion compound (wherein specific embodiments comprises the solid form like inclusion compound A described in the invention) of the stereoisomerism of significant quantity.
The present invention also relates in the patient treatment, prevention or control can be through reducing the method for the illness that the TNF-alpha levels improves, and it comprises pure compd A or its pharmaceutically acceptable prodrug, metabolite, polymorphic form, solvolyte, hydrate or the inclusion compound (wherein specific embodiments comprises the solid form like inclusion compound A described in the invention) of stereoisomerism to patient's administering therapeutic of this treatment of needs or prevention or prevention significant quantity.In specific embodiments, in Mammals, can produce disease or the illness improved and include but not limited to: HIV through inhibition TNF-α; Hepatitis; Adult respiratory distress syndrome; Bone resorption disease; Chronic obstructive pulmonary disease; Chronic pulmonary inflammation disease; Asthma; Dermatitis; Cystic fibrosis; Septic shock; Septicemia; Endotoxin shock; The haemodynamics shock; Septicemia syndrome; Postischemic reperfusion damage; Meningitis; Psoriatic; Fibrotic disease; Emaciation; Transplant rejection; Autoimmune disease; Rheumatoid spondylitis; Arhritis conditions, for example psoriatic arthritis, rheumatoid arthritis and osteo-arthritis; Osteoporosis; Segmental enteritis; Ulcerative colitis; Inflammatory bowel; Multiple sclerosis; Systemic lupus erythematosus; Lupus erythematosus,cutaneous; Sarcoidosis of lung; ENL in the leprosy (ENL); Radiation injury; Asthma; And hyperoxic alveolar injury.This illness further includes but not limited to cancer, includes but not limited to: the cancer of head, Tiroidina, neck, eye, skin, mouth, larynx, esophagus, chest, bone, blood, marrow, lung, colon, sigmoid colon, rectum, stomach, prostate gland, breast, ovary, kidney, liver, pancreas, brain, intestines, heart, suprarenal gland, subcutis, lymphoglandula, heart and combination thereof.Treatable by this method concrete cancer has multiple myeloma, malignant melanoma, glioblastoma, white blood disease and solid tumor.
Further embodiment of the present invention relates to treatment or preventing cancer in the patient; Include but not limited to solid tumor, haematogenous tumour, white blood disease; The method of multiple myeloma particularly, it comprises compd A or its pharmaceutically acceptable prodrug, metabolite, polymorphic form, solvolyte, hydrate or the inclusion compound (wherein specific embodiments comprise solid form like described in the invention inclusion compound A) pure to the stereoisomerism of patient's administering therapeutic significant quantity of this treatment of needs or prevention; Particularly, wherein patient is a Mammals.
In another embodiment; The present invention relates to suppress the method for PDE4, it comprises that the PDE4 that makes in the cell (for example mammalian cell) contacts with pure compd A or its pharmaceutically acceptable prodrug, metabolite, polymorphic form, solvolyte, hydrate or the inclusion compound (wherein specific embodiments comprises the solid form like inclusion compound A described in the invention) of the stereoisomerism of significant quantity.
Further embodiment of the present invention relates in the patient treatment or prevention can be through suppressing the disease that PDE4 improves or the method for illness, and it comprises pure compd A or its pharmaceutically acceptable prodrug, metabolite, polymorphic form, solvolyte, hydrate or the inclusion compound (wherein specific embodiments comprises the solid form like inclusion compound A described in the invention) of stereoisomerism to patient's administering therapeutic of this treatment of needs or prevention or prevention significant quantity.Can include but not limited to asthma, inflammation (for example by pouring into the inflammation that causes again), chronic or acute occlusion property tuberculosis, chronic or acute pulmonary inflammation disease, inflammatory bowel, segmental enteritis, Behcet's disease or colitis through the illness that inhibition PDE4 improves.
In another embodiment; The present invention relates to control the method for the cAMP level in the cell, it comprises contacts the pure compd A of the cell and the stereoisomerism of significant quantity or its pharmaceutically acceptable prodrug, metabolite, polymorphic form, solvolyte, hydrate or inclusion compound (wherein specific embodiments comprises the solid form like inclusion compound A described in the invention).The term that the present invention uses " control cAMP level " comprises prevention or reduces adenosine 3'; 5'-encircle single phosphoric acid (cAMP) in cell rate of decomposition or increase the adenosine 3' that exists in the cell; 5'-encircles the amount of single phosphoric acid, is preferably mammalian cell, more preferably is the human cell.In concrete grammar, with not with similar cell that compound of the present invention contacts in speed compare, the cAMP rate of decomposition has reduced about 10,25,50,100,200 or 500%.
Further embodiment of the present invention relates in the patient method of treatment or prevention depression, asthma, inflammation (for example contact dermatitis, atopic dermatitis, psoriatic, rheumatoid arthritis, osteo-arthritis, inflammatory skin diseases, the inflammation that caused by perfusion again), chronic or acute occlusion property tuberculosis, chronic or pulmonary inflammation disease, inflammatory bowel, segmental enteritis, Behcet's disease or colitis, and it comprises patient's administering therapeutic of this treatment of needs or prevention or prevents pure compd A or its pharmaceutically acceptable prodrug, metabolite, polymorphic form, solvolyte, hydrate or the inclusion compound (wherein specific embodiments comprises the solid form like inclusion compound A described in the invention) of stereoisomerism of significant quantity; Particularly, wherein the patient is a Mammals.
A separate embodiments of the present invention comprises the method for treatment or prevention myeloproliferative disorder syndrome (MDS), and it comprises pure compd A or its pharmaceutically acceptable solvolyte, hydrate, steric isomer, inclusion compound or the prodrug (wherein specific embodiments comprises the solid form like inclusion compound A described in the invention) of stereoisomerism to patient's administering therapeutic of this treatment of needs or prevention or prevention significant quantity.MDS refers to a different set of hemopoietic stem cell illness.The characteristic of MDS is form and the maturation (abnormal marrow hyperplasia) that cell marrow has infringement, peripheral blood hemocytopenia and proceed to the variable risk of acute leukemia, and it is produced by invalid hemocyte and causes.Referring to
The Merck Manual953 (the 17th editions, 1999) and List etc., 1990, J.Clin.Oncol.8:1424.
A separate embodiments of the present invention comprises the method for treatment or prevention myeloproliferative diseases (MPD), and it comprises pure compd A or its pharmaceutically acceptable solvolyte, hydrate, steric isomer, inclusion compound or the prodrug (wherein specific embodiments comprises the solid form like inclusion compound A described in the invention) of stereoisomerism to patient's administering therapeutic of this treatment of needs or prevention or prevention significant quantity.Myeloproliferative diseases (MPD) refers to that a stack features is the unusual illness of hemopoietic stem cell clone.Referring to for example,
Current Medical Diagnosis & Treatment, 499 pages (the 37th edition, editors such as Tierney, Appleton & Lange, 1998).
The present invention comprises that also treatment, prevention or pain management include but not limited to the method for complex region property pain syndrome, and it comprises pure compd A or its pharmaceutically acceptable solvolyte, hydrate, steric isomer, inclusion compound or the prodrug (wherein specific embodiments comprises the solid form like inclusion compound A described in the invention) of stereoisomerism to patient's administering therapeutic of this treatment of needs, prevention or control or prevention significant quantity.In one specific embodiment, be applied in be intended to reduce or avoid before the operation or Physiotherapy of symptom of complex region property pain syndrome, during or in the patient, carry out afterwards.
In concrete grammar of the present invention, the compd A that stereoisomerism is pure or its pharmaceutically acceptable polymorphic form, prodrug, solvolyte, hydrate or inclusion compound (wherein specific embodiments comprises the solid form like inclusion compound A described in the invention) are used with at least a additional therapeutic agent is auxiliary.The embodiment of additional therapeutic agent includes but not limited to: anticarcinogen, antiphlogiston, antihistaminic agent and Decongestant.
4.1.
The solid form of inclusion compound A
Certain embodiments of the present invention provide solid form, and it comprises the compd A with top chemical structure that shows.Racemic 2-[1-(3-oxyethyl group-4-p-methoxy-phenyl)-2-methyl sulphonyl ethyl]-4-acetylamino isoindoline-1,3-diketone use the method in the U.S. Patent number 6,020,358 to prepare easily, and it incorporates this paper by reference into.Compd A is 2-[1-(3-oxyethyl group-4-p-methoxy-phenyl)-2-methyl sulphonyl ethyl]-4-acetylamino isoindoline-1; (+) enantiomorph of 3-diketone; It can comprise U.S. Patent number 6,962 according to any method preparation that it will be apparent to those skilled in the art; The method of describing in 940, it incorporates this paper by reference into.
The solid form of inclusion compound A comprises single component and multicomponent form, comprises crystal formation and amorphous forms, and includes but not limited to polymorphic form, solvolyte, hydrate, eutectic and inclusion compound.Specific embodiments of the present invention provides the single component of compd A unformed solid form.Specific embodiments of the present invention provides the single component crystalline solid forms of compd A.Specific embodiments of the present invention provides the multicomponent amorphous forms of inclusion compound A.Specific embodiments of the present invention provides the multicomponent crystalline solid forms of inclusion compound A.Multicomponent solid form provided by the invention comprises the solid form that can use term salt, eutectic, hydrate, solvolyte, inclusion compound and/or polymorphic form to describe, also comprises the solid form that can use one or more these term descriptions.
The solid form of inclusion compound A can comprise the method described among the following embodiment or technology preparation known in the art through method described herein, comprise that heating, cooling, lyophilize, freeze-drying, fusing quenching, solvent evaporation fast, slowly solvent evaporation, solvent recrystallization, anti-solvent add, the slurry recrystallization, by melting crystal, desolvation, restricted clearance for example recrystallization in nanoporous or the kapillary, surface or template for example recrystallization on the polymkeric substance, additive for example the eutectic ligand molecule in the presence of recrystallization, desolvation, dehydration, cool off, slowly cool off, be exposed to that solvent and/or water, drying comprise for example vacuum-drying, vapor diffusion, distillation, grinding (comprising for example cold grinding, the grinding of solvent drippage or liquid assisted milling), microwave induced deposition, ultrasonic degradation induced precipitation, induced with laser precipitates and precipitated by supercutical fluid fast.The granularity of the solid form that obtains can change (for example nano level is to the millimeter level); This can through for example change crystallization condition for example crystallization rate and/or recrystallisation solvent system control, or through granularity reduce that technology is for example ground, milled, micronization or ultrasonic degradation control.
Although do not hope to be subject to any particular theory, some solid form is characterised in that the physical properties that is fit to medicine and therapeutic dosage forms is for example stable, solubleness and dissolution rate.In addition; Although do not hope to be subject to any particular theory; Some solid form be characterised in that influence particular procedure (for example output, filtration, washing, drying, mill, mixing, compressing tablet, flowability, dissulution, preparation and freeze-drying) physical properties (for example density, compressibility, hardness, form, dissociate, viscosity, solubleness, moisture absorption, electrical property, thermal behavior, solid state reaction property, physical stability and chemicalstability), this makes some solid form be fit to the preparation solid dosage.Such character can be used like this paper and describe and concrete analysis chemical technology known in the art mensuration, comprises solid-state analytical technology (for example X-ray diffraction, microscope, spectrum and heat are analyzed).
Certain embodiments of the present invention provide the compsn that comprises one or more solid forms.Some embodiment provides the compsn of one or more solid forms and the combination of other activeconstituents.Some embodiment provides in disease and treatment of conditions, prevention or control and has used these method for compositions, includes but not limited to disease provided by the invention and illness.
Except that the solid form of inclusion compound A, the present invention also provides the solid form of the prodrug of inclusion compound A.
Solid form provided by the invention also can comprise non-natural atom isotope part in the one or more atoms place in compd A.For example, compound can come radio-labeled with ri, for example as tritium (
3H), iodine-125 (
125I), Sulphur-35 (
35S) or carbon-14 (
14C).Radiolabeled compound useful as therapeutics for example cancer therapeutic agent, research reagent for example combines the for example interior developer of body of test reagent and diagnostic reagent.No matter whether all isotropic substance mutation of compd A be radioactive, all is comprised in the scope of embodiment provided by the invention.
4.1.1.
The form A of compd A
Certain embodiments of the present invention provide the form A crystal formation of compd A.In certain embodiments, the form A of compd A can be obtained by all kinds of SOLVENTS, includes but not limited to comprise the solvent system of acetone, ethanol and composition thereof.In certain embodiments, form A can use quick crystallisation by cooling process to obtain.
In certain embodiments, the form A of compd A can characterize through the X-ray powder diffraction analysis.The typical XRPD figure of the form A of compd A provides in Fig. 1.In certain embodiments, the characteristic of the form A of compd A is the XRPD peak that is arranged in one, two, three, four, five, six, seven, eight, nine, ten, 11 or 12 position of following apparent position: 8.1,14.4,15.2,17.4,18.4,19.2,20.5,22.8,23.2,23.6,24.5,25.1 degree, 2 θ.In certain embodiments, the characteristic of the form A of compd A be with Fig. 1 in the XRPD figure that is complementary of the figure that shows.In certain embodiments, the characteristic of the form A of compd A is to have the XRPD figure that the peak among 1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24 or 25 peak and the canonical form A provided by the present invention figure is complementary.
In certain embodiments, the form A of compd A can characterize through the heat analysis.The typical DSC figure of the form A of compd A shows in Fig. 2.In certain embodiments, the characteristic of form A is the DSC figure that comprises the heat absorption incident of the about 145 ° of C of starting temperature.In certain embodiments, the characteristic of form A is the DSC figure that further comprises the heat absorption incident of the about 155 ° of C of starting temperature.The typical TGA figure of the form A of compd A shows in Fig. 3.In certain embodiments, the characteristic of form A is when being heated to about 140 ° of C from about 25 ° of C, comprises to be lower than the about 1% of sample total mass, the TGA figure of for example about 0.05% mass loss.In certain embodiments, the form A of compd A does not comprise a large amount of water or other solvent in lattice.In certain embodiments, form A is a solvation not.In certain embodiments, form A is anhydrous.
In certain embodiments, the form A of compd A can characterize through the water adsorption analysis.Typical water adsorption isothermal chart shows in Fig. 4.In certain embodiments, when relative humidity (" RH ") is increased to about 95%RH from about 0%, form A shows and is lower than the about 1% of sample initial mass, for example about 0.4% quality change.In certain embodiments, when RH rolls back about 0%RH, by the quality disappearance of absorption acquisition.Therefore, in certain embodiments, form A is nonhygroscopic basically.In certain embodiments, the XRPD figure of form A material was constant basically after the adsorption/desorption Fufen was analysed.In certain embodiments, form A is stable for humidity.
In certain embodiments, the form A of compd A can characterize through its stability features.In certain embodiments, form A material is stable, and for example when being exposed to high temperature, when being exposed to high humidity, when being exposed to one or more solvents, and/or under pressure, its XRPD figure keeps constant basically.In certain embodiments, for example, be exposed to about 40 ° of C environment and the about four stars after date of about 75%RH environment, form A is stable.In certain embodiments, be exposed to one or more solvent systems that comprise for example ethanol, water and/or heptane at least about the four stars after date at about 40 ° of C, form A is stable.In certain embodiments, when being exposed to the solvent four stars during phase that includes but not limited to toluene, form A changes into the form A of compd A.In certain embodiments, under the pressure of the about 2000psi of pressure about one minute, form A was stable.
In certain embodiments, the form A of compd A can characterize through grain size analysis.In certain embodiments, the characteristic of form A is a white powder.In certain embodiments, the sample of form A comprises the particle with lamellar morphology.In certain embodiments, the sample of form A comprises D
90Value is lower than the particle of about 18 μ m.(the D that the present invention uses
90Value, representative is 90 through the percentile of the size-grade distribution of linear measure; Promptly 90% particle has the length of this value or shorter).
Certain embodiments of the present invention provide the form A of pure basically compd A.Certain embodiments of the present invention provide the form A of compd A, and the solid form that it does not contain other inclusion compound A basically for example comprises, like form B, C, D, E, F, G and/or the unformed solid form of inclusion compound A provided by the invention.The form A that certain embodiments of the present invention provide is the mixture of the solid form of inclusion compound A, comprises for example comprising following one or more mixture: like form B, C, D, E, F, G and the unformed solid form of inclusion compound A provided by the invention.
4.1.2.
The form B of compd A
Certain embodiments of the present invention provide the form B crystal formation of compd A.In certain embodiments; The form B of compd A can be obtained by all kinds of SOLVENTS, the solvent system that includes but not limited to comprise 2-propyl alcohol, acetone, acetonitrile, ethanol, ETHYLE ACETATE, heptane, methyl alcohol, methyl ethyl ketone, methyl tert-butyl ether, methylene dichloride, propyl carbinol, n-butyl acetate, THF, toluene, water and comprise its two or more mixture.For example, in certain embodiments, form B can be through from comprising 1:1 ethanol: crystallization obtains the solvent system of water, and for example through being included in about 25 ° of C evaporation 1:1 ethanol: the solvent system of water, then the process of unpack format B obtains.For example; In certain embodiments; Form B can be through from comprising 1:1 acetone: crystallization obtains the alcoholic acid solvent system, and for example through being included in about 25 ° of C at 1:1 acetone: the solid form of pulp inclusion compound A is about 2 days in the ethanol, and then the process of unpack format B obtains.
In certain embodiments, the form B of compd A can characterize through the X-ray powder diffraction analysis.The typical XRPD figure of the form B of compd A provides in Fig. 5.In certain embodiments, the characteristic of the form B of compd A is the XRPD peak that is arranged in one, two, three, four, five, six, seven, eight, nine, ten, 11 or 12 position of following apparent position: 10.1,12.4,13.5,15.7,16.3,18.1,20.7,22.5,24.7,26.2,26.9,29.1 degree, 2 θ.In certain embodiments, the characteristic of the form B of compd A be with Fig. 5 in the XRPD figure that is complementary of the figure that shows.In certain embodiments, the characteristic of the form B of compd A is to have the XRPD figure that the peak among 1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24 or 25 peak and the canonical form B provided by the invention figure is complementary.
In certain embodiments, the form B of compd A can characterize through the heat analysis.The typical DSC figure of the form B of compd A shows in Fig. 6.In certain embodiments, the characteristic of form B is the DSC figure that comprises the heat absorption incident of the about 154 ° of C of starting temperature.The typical TGA figure of the form B of compd A shows in Fig. 7.In certain embodiments, the characteristic of form B is when being heated to about 140 ° of C from about 25 ° of C, comprises to be lower than the about 1% of sample total mass, the TGA figure of for example about 0.25% mass loss.In certain embodiments, the form B of compd A does not comprise a large amount of water or other solvent in lattice.In certain embodiments, form B is anhydrous.In certain embodiments, form B is a solvation not.
In certain embodiments, the form B of compd A can characterize through the water adsorption analysis.Typical water adsorption isothermal chart shows in Fig. 8.In certain embodiments, when RH is increased to about 95%RH from about 0%, form B shows and is lower than the about 1% of sample initial mass, for example about 0.6% quality change.In certain embodiments, when RH rolls back about 0%RH, by the quality disappearance of absorption acquisition.In certain embodiments, form B is nonhygroscopic basically.In certain embodiments, the XRPD figure of form B material was constant basically after the adsorption/desorption Fufen was analysed.In certain embodiments, form B is stable for humidity.
In certain embodiments, the form B of compd A can characterize through its stability features.In certain embodiments, form B material is stable, and for example when being exposed to high temperature, when being exposed to high humidity, when being exposed to one or more solvents, and/or under pressure, its XRPD figure keeps constant basically.In certain embodiments, for example, be exposed to about 40 ° of C environment and the about four stars after date of about 75%RH environment, form B is stable.In certain embodiments, about 40 ° of C be exposed to comprise for example ethanol, water or heptane solvent system at least about the four stars after date, form B is stable.In certain embodiments, be exposed to the about four stars of solvent system during the phase that comprises toluene for example, form B changes into the form A of compd A.In certain embodiments, after under the pressure of the about 2000psi of pressure about one minute, form B is stable.
In certain embodiments, the form B of compd A can characterize through grain size analysis.In certain embodiments, the characteristic of form B is a white powder.In certain embodiments, the sample of form B comprises the particle with sheet-like form.In certain embodiments, the sample of form B comprises the particle that the D90 value is lower than about 12 μ m.
Certain embodiments of the present invention provide the form B of pure basically compd A.Certain embodiments of the present invention provide the form B of compd A, and the solid form that it does not contain other inclusion compound A basically for example comprises, like form A, C, D, E, F, G and/or the unformed solid form of inclusion compound A provided by the invention.The form B that certain embodiments of the present invention provide is the mixture of the solid form of inclusion compound A, comprises for example comprising following one or more mixture: like form A, C, D, E, F, G and the unformed solid form of inclusion compound A provided by the present invention.
4.1.3.
The form A of compd A
Certain embodiments of the present invention provide the form A crystal formation of compd A.In certain embodiments; The form A of compd A can be obtained by all kinds of SOLVENTS system, the solvent system that includes but not limited to comprise acetone, acetonitrile, ethanol, heptane, methyl alcohol, methyl ethyl ketone, THF, toluene, water and comprise its two or more mixture.For example, in certain embodiments, form A can obtain through crystallization from the solvent system that comprises toluene, for example uses toluene as anti-solvent through comprising, then the process of unpack format C obtains.
In certain embodiments, the form A of compd A can characterize through the X-ray powder diffraction analysis.The typical XRPD figure of the form A of compd A provides in Fig. 9.In certain embodiments, the characteristic of the form A of compd A is the XRPD peak that is arranged in one, two, three, four, five, six, seven, eight, nine, ten, 11 or 12 position of following apparent position: 7.5,11.3,15.3,16.4,17.8,21.4,22.6,23.5,24.8,25.5,26.4,27.6 degree, 2 θ.In certain embodiments, the characteristic of the form A of compd A be with Fig. 9 in the XRPD figure that is complementary of the figure that shows.In certain embodiments, the characteristic of the form A of compd A is to have the XRPD figure that the peak among 1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24 or 25 peak and the canonical form C provided by the present invention figure is complementary.
In certain embodiments, the form A of compd A can characterize through the heat analysis.The typical DSC figure of the form A of compd A shows in Figure 10.In certain embodiments, the characteristic of form A is the DSC figure that comprises the heat absorption incident of the about 138 ° of C of starting temperature.In certain embodiments, distinctive form A DSC figure further comprises one or more additional events, for example like the heat absorption incident of the about 166 ° of C of starting temperature.The typical TGA figure of the form A of compd A shows in Figure 11.In certain embodiments, the characteristic of form A is when being heated to about 140 ° of C from about 25 ° of C, comprises to be lower than the about 10% of sample total mass, the TGA figure of for example about 5.9% mass loss.In certain embodiments, like what show through for example TG-IR analysis, TGA mass loss incident comprises the loss of solvent toluene.In certain embodiments, the form A of compd A is a solvation.In certain embodiments, form A is the toluene solvant thing.In certain embodiments, the every mole compound A of the lattice of form A comprises about three moles of toluene.
In certain embodiments, the form A of compd A can characterize through the water adsorption analysis.Typical water adsorption isothermal chart shows in Figure 12.In certain embodiments, when RH is increased to about 95%RH from about 0%, form A shows and is lower than the about 1% of sample initial mass, for example about 0.5% quality change.In certain embodiments, when RH rolls back about 0%RH, by the quality disappearance of absorption acquisition.In certain embodiments, form A is nonhygroscopic basically.In certain embodiments, the XRPD figure of form A material was constant basically after the adsorption/desorption Fufen was analysed.In certain embodiments, form A is stable for humidity.
In certain embodiments, the form A of compd A can characterize through its stability features.In certain embodiments, the form A material is stable, and for example when being exposed to high temperature, when being exposed to high humidity, when being exposed to one or more solvents, and/or under pressure, its XRPD figure keeps constant basically.In certain embodiments, for example, be exposed to about 40 ° of C environment and the about four stars after date of about 75%RH environment, form A is stable.In certain embodiments, about 40 ° of C be exposed to comprise for example ethanol, water, heptane or toluene solvent system at least about the four stars after date, form A is stable.In certain embodiments, after under the pressure of the about 2000psi of pressure about one minute, form A is stable.
In certain embodiments, the form A of compd A can characterize through grain size analysis.In certain embodiments, the characteristic of form A is a white powder.In certain embodiments, the sample of form A comprises the particle with lamellar morphology.In certain embodiments, the sample of form A comprises D
90Value is lower than the particle of about 12 μ m.
Certain embodiments of the present invention provide the form A of pure basically compd A.Certain embodiments of the present invention provide the form A of compd A, and the solid form that it does not contain other inclusion compound A basically for example comprises, like form A, B, D, E, F, G and/or the unformed solid form of inclusion compound A provided by the present invention.The form A that certain embodiments of the present invention provide is the mixture of the solid form of inclusion compound A, comprises for example comprising following one or more mixture: like form A, B, D, E, F, G and the unformed solid form of inclusion compound A provided by the present invention.
4.1.4.
The form D of compd A
Certain embodiments of the present invention provide the form D crystal formation of compd A.In certain embodiments, the form D of compd A can be obtained by all kinds of SOLVENTS, includes but not limited to comprise the solvent system of methylene dichloride.For example, in certain embodiments, form D can obtain through crystallization from the solvent system that comprises methylene dichloride, and for example through comprising the evaporation methylene dichloride, then the process of unpack format D obtains.
In certain embodiments, the form D of compd A can characterize through the X-ray powder diffraction analysis.The typical XRPD figure of the form D of compd A provides in Figure 13.In certain embodiments, the characteristic of the form D of compd A is the XRPD peak that is arranged in one, two, three, four, five, six, seven, eight, nine, ten, 11 or 12 position of following apparent position: 7.5,9.6,11.3,13.9,16.3,17.7,20.5,23.2,24.6,25.2,26.0,28.8 degree, 2 θ.In certain embodiments, the characteristic of the form D of compd A be with Figure 13 in the XRPD figure that is complementary of the figure that shows.In certain embodiments, the characteristic of the form D of compd A is to have the XRPD figure that the peak among 1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24 or 25 peak and the canonical form D provided by the present invention figure is complementary.
In certain embodiments, the form D of compd A can characterize through the heat analysis.The typical DSC figure of the form D of compd A shows in Figure 14.In certain embodiments, the characteristic of form D is the DSC figure that comprises the heat absorption incident of the about 100 ° of C of starting temperature.The typical TGA figure of the form D of compd A shows in Figure 15.In certain embodiments, the characteristic of form D is when being heated to about 110 ° of C from about 25 ° of C, comprises to be lower than the about 10% of sample total mass, the TGA figure of for example about 6.5% mass loss.In certain embodiments, like what show through for example TG-IR analysis, TGA mass loss incident comprises the loss of methylene chloride (being methylene chloride).In certain embodiments, the form D of compd A is a solvation.In certain embodiments, form D is the dichloromethane solvent thing.In certain embodiments, the every mole compound A of the lattice of form D comprises about 2.5 moles of methylene dichloride.
In certain embodiments, the form D of compd A can characterize through the water adsorption analysis.Typical water adsorption isothermal chart shows in Figure 16.In certain embodiments, when RH is increased to about 95%RH from about 0%, form D shows and is lower than the about 3% of sample initial mass, for example about 1.5% quality change.In certain embodiments, when RH rolls back about 0%RH, by the quality disappearance of absorption acquisition.Therefore, in certain embodiments, form D is slight moisture absorption.In certain embodiments, the XRPD figure of form D material was constant basically after the adsorption/desorption Fufen was analysed.In certain embodiments, form D is stable for humidity.
In certain embodiments, the form D of compd A can characterize through its stability features.In certain embodiments, form D material is stable, and for example its XRPD figure keeps constant basically under pressure.For example, in certain embodiments, after under the pressure of the about 2000psi of pressure about one minute, form D is stable.In certain embodiments, be exposed to about 40 ° of C environment and the about four stars after date of about 75%RH environment, form D is stable, although in certain embodiments, the peak intensity that the XRPD of form D figure obtains descends.In certain embodiments, this decline of XRPD peak intensity is because formed the amorphous material of inclusion compound A.In certain embodiments, be exposed to the about four stars of solvent system during the phase that comprises for example heptane, ethanol and/or water at about 40 ° of C, form D changes into the form B of compd A.In certain embodiments, be exposed to the about four stars of solvent system during the phase that comprises toluene at about 40 ° of C, form D changes into the form A of compd A.
In certain embodiments, the form D of compd A can characterize through grain size analysis.In certain embodiments, the characteristic of form D is a white powder.In certain embodiments, the sample of form D comprises the particle with sheet-like form.In certain embodiments, the sample of form D comprises D
90Value is lower than the particle of about 18 μ m.
Certain embodiments of the present invention provide the form D of pure basically compd A.Certain embodiments of the present invention provide the form D of compd A, and the solid form that it does not contain other inclusion compound A basically for example comprises as form A, B, C, E, F, G and/or the unformed solid form of inclusion compound A provided by the present invention.The form D that certain embodiments of the present invention provide is the mixture of the solid form of inclusion compound A, comprises for example comprising following one or more mixture: like form A, B, C, E, F, G and the unformed solid form of inclusion compound A provided by the present invention.
4.1.5.
The form E of compd A
Certain embodiments of the present invention provide the form E crystal formation of compd A.In certain embodiments, the form E of compd A can be obtained by all kinds of SOLVENTS, the solvent system that includes but not limited to comprise acetone, acetonitrile, heptane, methylene dichloride and comprise its two or more mixture.For example, in certain embodiments, form E can obtain through crystallization from the solvent system that comprises acetonitrile, and for example through comprising the evaporation acetonitrile, then the process of unpack format E obtains.
In certain embodiments, the form E of compd A can characterize through the X-ray powder diffraction analysis.The typical XRPD figure of the form E of compd A provides in Figure 17.In certain embodiments, the characteristic of the form E of compd A is the XRPD peak that is arranged in one, two, three, four, five, six, seven, eight, nine, ten, 11 or 12 position of following apparent position: 7.6,9.2,11.4,15.5,16.5,17.9,19.6,20.5,21.6,22.8,23.8,26.6 degree, 2 θ.In certain embodiments, the characteristic of the form E of compd A be with Figure 17 in the XRPD figure that is complementary of the figure that shows.In certain embodiments, the characteristic of the form E of compd A is to have the XRPD figure that the peak among 1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24 or 25 peak and the canonical form E provided by the present invention figure is complementary.
In certain embodiments, the form E of compd A can characterize through the heat analysis.The typical DSC figure of the form E of compd A shows in Figure 18.In certain embodiments, the characteristic of form E is the DSC figure that comprises the heat absorption incident of the about 95 ° of C of starting temperature.The typical TGA figure of the form E of compd A shows in Figure 19.In certain embodiments, the characteristic of form E is when being heated to about 110 ° of C from about 25 ° of C, comprises to be lower than the about 8% of sample total mass, the TGA figure of for example about 4.0% mass loss.In certain embodiments, like what show through for example TG-IR analysis, TGA mass loss incident comprises the loss of solvent acetonitrile.In certain embodiments, the form E of compd A is a solvation.In certain embodiments, form E is an acetonitrile solvate.In certain embodiments, the every mole compound A of the lattice of form E comprises about 2.5 moles of acetonitriles.
In certain embodiments, the form E of compd A can characterize through the water adsorption analysis.Typical water adsorption isothermal chart shows in Figure 20.In certain embodiments, when RH is increased to about 95%RH from about 0%, form E shows and is lower than the about 10% of sample initial mass, for example about 5.1% quality change.In certain embodiments, when RH rolls back about 0%RH, by the quality disappearance of absorption acquisition.In certain embodiments, form E is moisture absorption.In certain embodiments, the XRPD figure of form E material was constant basically after the adsorption/desorption Fufen was analysed.In certain embodiments, form E is stable for humidity.
In certain embodiments, the form E of compd A can characterize through its stability features.In certain embodiments, form E material is stable, and for example its XRPD figure keeps constant basically under pressure.For example, in certain embodiments, after under the pressure of the about 2000psi of pressure about one minute, form E is stable.
In certain embodiments, the form E of compd A can characterize through grain size analysis.In certain embodiments, the characteristic of form E is a white powder.In certain embodiments, the sample of form E comprises the particle with sheet-like form.In certain embodiments, the sample of form E comprises D
90Value is lower than the particle of about 18 μ m.
Certain embodiments of the present invention provide the form E of pure basically compd A.Certain embodiments of the present invention provide the form E of compd A, and the solid form that it does not contain other inclusion compound A basically for example comprises as form A, B, C, D, F, G and/or the unformed solid form of inclusion compound A provided by the present invention.The form E that certain embodiments of the present invention provide is the mixture of the solid form of inclusion compound A, comprises for example comprising following one or more mixture: like form A, B, C, D, F, G and the unformed solid form of inclusion compound A provided by the present invention.
4.1.6.
The form D of compd A
Certain embodiments of the present invention provide the form D crystal formation of compd A.In certain embodiments, the form D of compd A can be obtained by all kinds of SOLVENTS, the solvent system that includes but not limited to comprise acetone, ethanol, water and comprise its two or more mixture.For example; In certain embodiments; Form D can obtain through crystallization from the solvent system that comprises ethanol and/or water, and for example through comprising that the solid form that makes inclusion compound A contacts with the solvent system that comprises ethanol and/or water, then the process of unpack format F obtains.
In certain embodiments, the form D of compd A can characterize through the X-ray powder diffraction analysis.The typical XRPD figure of the form D of compd A provides in Figure 21.In certain embodiments, the characteristic of the form D of compd A is the XRPD peak that is arranged in one, two, three, four, five, six, seven, eight, nine, ten, 11 or 12 position of following apparent position: 8.1,8.6,15.6,17.3,19.3,21.4,22.8,24.6,25.4,25.9,26.6,27.7 degree, 2 θ.In certain embodiments, the characteristic of the form D of compd A be with Figure 21 in the XRPD figure that is complementary of the figure that shows.In certain embodiments, the characteristic of the form D of compd A is to have the XRPD figure that the peak among 1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24 or 25 peak and the canonical form F provided by the present invention figure is complementary.
In certain embodiments, the form D of compd A can characterize through the heat analysis.The typical DSC figure of the form D of compd A shows in Figure 22.In certain embodiments, the characteristic of form D is the DSC figure that comprises the heat absorption incident of the about 145 ° of C of starting temperature.The typical TGA figure of the form D of compd A shows in Figure 23.In certain embodiments, the characteristic of form D is when being heated to about 180 ° of C from about 25 ° of C, comprises to be lower than the about 1% of sample total mass, the TGA figure of for example about 0.1% mass loss.In certain embodiments, the form D of compd A does not comprise a large amount of water or other solvent in lattice.In certain embodiments, form D is a solvation not.In certain embodiments, form D is anhydrous.
In certain embodiments, the form D of compd A can characterize through the water adsorption analysis.Typical water adsorption isothermal chart shows in Figure 24.In certain embodiments, when RH is increased to about 95%RH from about 0%, form D shows and is lower than the about 1% of sample initial mass, for example about 0.2% quality change.In certain embodiments, when RH rolls back about 0%RH, by the quality disappearance of absorption acquisition.In certain embodiments, form D is nonhygroscopic basically.In certain embodiments, the XRPD figure of form D material was constant basically after the adsorption/desorption Fufen was analysed.In certain embodiments, form D is stable for humidity.
In certain embodiments, the form D of compd A can characterize through its stability features.In certain embodiments, the form D material is stable, and for example its XRPD figure keeps constant basically under pressure.For example, in certain embodiments, after under the pressure of the about 2000psi of pressure about one minute, form D is stable.In certain embodiments, about 25 ° of C be exposed to comprise for example ethanol, acetone or its mixture solvent system approximately two days later, form D is stable.
In certain embodiments, the form D of compd A can characterize through grain size analysis.In certain embodiments, the characteristic of form D is a white powder.In certain embodiments, the sample of form D comprises the particle with sheet-like form.In certain embodiments, the sample of form D comprises D
90Value is lower than the particle of about 18 μ m.
Certain embodiments of the present invention provide the form D of pure basically compd A.Certain embodiments of the present invention provide the form D of compd A, and the solid form that it does not contain other inclusion compound A basically for example comprises as form A, B, C, D, E, G and/or the unformed solid form of inclusion compound A provided by the present invention.The form D that certain embodiments of the present invention provide is the mixture of the solid form of inclusion compound A, comprises for example comprising following one or more mixture: like form A, B, C, D, E, G and the unformed solid form of inclusion compound A provided by the present invention.
4.1.7.
The form G of compd A
Certain embodiments of the present invention provide the form G crystal formation of compd A.In certain embodiments, the form G of compd A can be obtained by all kinds of SOLVENTS, includes but not limited to comprise the solvent system of ETHYLE ACETATE.For example; In certain embodiments; Form G can obtain through crystallization from the solvent system that comprises ETHYLE ACETATE, and for example through comprising that the solid form with inclusion compound A contacts with the solvent system that comprises ETHYLE ACETATE, then the process of unpack format G obtains.
In certain embodiments, the form G of compd A can characterize through the X-ray powder diffraction analysis.The typical XRPD figure of the form G of compd A provides in Figure 25.In certain embodiments, the characteristic of the form G of compd A is the XRPD peak that is arranged in one, two, three, four, five, six, seven, eight, nine, ten, 11 or 12 position of following apparent position: 7.9,9.5,11.7,15.7,16.8,18.1,19.7,21.8,22.8,25.1,25.8,26.7 degree, 2 θ.In certain embodiments, the characteristic of the form G of compd A be with Figure 25 in the XRPD figure that is complementary of the figure that shows.In certain embodiments, the characteristic of the form G of compd A is to have the XRPD figure that the peak among 1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24 or 25 peak and the canonical form G provided by the present invention figure is complementary.
In certain embodiments, the form G of compd A can characterize through the heat analysis.The typical DSC figure of the form G of compd A shows in Figure 26.In certain embodiments, the characteristic of form G is the DSC figure that comprises the heat absorption incident of the about 109 ° of C of starting temperature.The typical TGA figure of the form G of compd A shows in Figure 27.In certain embodiments, the characteristic of form G is when being heated to about 110 ° of C from about 25 ° of C, comprises to be lower than the about 8% of sample total mass, the TGA figure of for example about 3.8% mass loss.In certain embodiments, like what show through for example TG-IR analysis, TGA mass loss incident comprises the loss of solvent ethyl acetate.In certain embodiments, the form G of compd A is a solvation.In certain embodiments, form G is the ethyl acetate solvent thing.In certain embodiments, the every mole compound A of the lattice of form G comprises about three mole of acetic acid ethyl esters.
In certain embodiments, the form G of compd A can characterize through the water adsorption analysis.Typical water adsorption isothermal chart shows in Figure 28.In certain embodiments, when RH is increased to about 95%RH from about 0%, form G shows and is lower than the about 1% of sample initial mass, for example about 0.4% quality change.In certain embodiments, when RH rolls back about 0%RH, by the quality disappearance of absorption acquisition.In certain embodiments, form G is nonhygroscopic basically.In certain embodiments, the XRPD figure of form G material was constant basically after the adsorption/desorption Fufen was analysed.In certain embodiments, form G is stable for humidity.
In certain embodiments, the form G of compd A can characterize through its stability features.In certain embodiments, form G material is stable, and for example its XRPD figure keeps constant basically under pressure.For example, in certain embodiments, after under the pressure of the about 2000psi of pressure about one minute, form D is stable.In certain embodiments, be exposed to the solvent system in the time of about two days that comprises for example ethanol, acetone or its mixture at about 25 ° of C, form G changes into form B.
In certain embodiments, the form G of compd A can characterize through grain size analysis.In certain embodiments, the characteristic of form G is a white powder.In certain embodiments, the sample of form G comprises the particle with sheet-like form.In certain embodiments, the sample of form G comprises D
90Value is lower than the particle of about 18 μ m.
Certain embodiments of the present invention provide the form G of pure basically compd A.Certain embodiments of the present invention provide the form G of compd A, and the solid form that it does not contain other inclusion compound A basically for example comprises as form A, B, C, D, E, F and/or the unformed solid form of inclusion compound A provided by the present invention.The form G that certain embodiments of the present invention provide is the mixture of the solid form of inclusion compound A, comprises for example comprising following one or more mixture: like form A, B, C, D, E, F and the unformed solid form of inclusion compound A provided by the present invention.
4.2.
Treat-ment
Present invention resides among the patient treatment, prevention and control can be through reducing the disease that the TNF-alpha levels improves or the method for illness; It comprises the solid form to one or more inclusion compounds A of patient's administering therapeutic of this treatment of needs, prevention or control or prevention significant quantity; For example as, like the form A of compd A provided by the present invention, the form B of compd A, the form A of compd A, the form D of compd A, the form E of compd A, the form D of compd A, the form G of compd A or the amorphous forms of compd A.
Can include but not limited to through the illness that inhibition TNF-α improves: heart trouble, the septic shock of for example congestive heart failure, myocardosis, wet lung, endotaxin mediate, acute viral myocarditis, heart heteroplastic transplantation are repelled and myocardial infarction; Solid tumor includes but not limited to sarcoma, cancer, fibrosarcoma, myxosarcoma, liposarcoma, chondrosarcoma, osteogenic sarcoma, chordoma, angiosarcoma, endotheliosarcoma, lymphangiosarcoma, lymphangioendothelial sarcoma, synovioma, mesothelioma, Ewing's tumour, leiomyosarcoma, rhabdosarcoma, colorectal carcinoma, carcinoma of the pancreas, mammary cancer, ovarian cancer, prostate cancer, squamous cell carcinoma, rodent cancer, gland cancer, syringocarcinoma, sebaceous carcinoma, papillary carcinoma, papillary carcinoma, cystadenocarcinoma, medullary carcinoma, bronchogenic carcinoma, renal cell carcinoma, liver cancer, cholangiocarcinoma, choriocarcinoma, spermocytoma, embryonal carcinoma, the nephroblastoma, cervical cancer, tumor of testis, lung cancer, small cell lung cancer, bladder cancer, epithelial cancer, glioma, astrocytoma, medulloblastoma, craniopharyngioma, ependymoma, Kaposi's sarcoma, pinealoma, hemangioblastoma, acoustic tumor, oligodendroglioma, meningioma, melanoma, neuroblastoma and retinoblastoma; And haematogenous tumour; Include but not limited to acute lymphoblastic leukemia " ALL ", acute lymphocytoblast property B cell leukemia, acute lymphocytoblast property T HTLV, acute myeloblastic leukemia " AML ", acute promyelocytic leukemia " APL ", acute monoblastic leukemia, Di Guglielmo syndrome property white blood disease, acute megakaryoblast property white blood disease, acute grain monocytic leukemia, acute nonlymphocytic leukemia, acute nondifferentiated leukemia, chronic myelocytic leukemia " CML ", lymphocytic leukemia " CLL ", hairy cell leukemia, multiple myeloma and acute and chronic leukemia, for example lymphocytoblast property, marrow property, lymphatic and myelocytic leukemia.
Concrete grammar of the present invention further comprises uses extra therapeutical agent (being the therapeutical agent outside the compd A).The embodiment of additional therapeutic agent includes but not limited to anticarcinogen, such as but not limited to: alkylating agent, mustargen, ethyleneimine, methyl melamine, AS, nitrosourea, triazene, folacin, pyrimidine analogue, purine analogue, vinca alkaloids, epipodophyllotoxin, microbiotic, topoisomerase enzyme inhibitor and anti-cancer vaccine.
Concrete additional therapeutic agent includes but not limited to: U 42126; Aclarubicin; The hydrochloric acid acodazole; Acronine; U 73975; RIL-2; Altretamine; Duazomycin C; The acetic acid ametantrone; Aminoglutethimide; Amsacrine; Anastrozole; Antramycin; Asparaginase; Asperline; Azacitidine; Azatepa; Azotomycin; BB-94; Benzodepa; Bicalutamide; Bisantrene hydrochloride; Two methylsulfonic acid Bisnafides; U 77779; BLEOMYCIN SULPHATE; Brequinar sodium; U-54461; Busulfan; Sanarnycin; Calusterone; Caracemide; Carbetimer; Carboplatin; Carmustine; NSC-180024; U 80244; SPC 101210; TV; U 12241; Cis-platinum; CldAdo; The methylsulfonic acid crisnatol; Endoxan; Cytosine arabinoside; Dicarbazine; Dactinomycin; Daunorubicin hydrochloride; NSC 127716; U 78938; Dezaguanine; The methylsulfonic acid Dezaguanine; NSC-182986; Docetaxel; Dx; Doxorubicin hydrochloride; Droloxifene; K-21060E; NSC-12198; Duazomycin; Edatrexate; Vaniqa; Elsamitrucin; Enloplatin; Enpromate; NSC 56308; Epirubicin hydrochloride; R 55104; IMI 58; Estramustine; Estramustine phosphate sodium; SR-2508; VP; The phosphoric acid VP; Etoprine; CGS-16949A; NSC 281272; HPR; Floxuridine; NSC-328002; Fluracil; Flurocitabine; GR 63178X; Phosphotrienin sodium; Gemcitabine; Gemcitabine hydrochloride; Hydroxyurea; Idarubicin hydrochloride; Ifosfamide; Thio ALP; Interleukin-II (the interleukin-II or the rIL2 that comprise reorganization); Intederon Alpha-2a; Interferon Alpha-2b; Interferon alfa-n1; Alferon N; Interferon beta-Ia; Interferon-gamma-Ib; NSC 256927; Irinotecan hydrochloride; Lanreotide acetate; Letrozole; TAP-144; Liarozole hydrochloride; Lometrexol sodium; Lomustine; Losoxantrone hydrochloride; Masoprocol; Maytenin; Mustine hydrochlcride; Magace; Melengestrol acetate; Melphalan; Menogaril; Mercaptopurine; Methotrexate; Methotrexate sodium; U-197; Meturedepa; NSC 284356; Rice holder jinx; NSC 77471; Mitogillin; NSC-B 2992; MTC; Mitosper; Mitotane; NSC-301739; Mycophenolic Acid; R 17934; Nogalamycin; Ormaplatin; Oxisuran; Taxol; Pegaspargase; Peliomycin; Pentamustine; Peplomycin Sulfate; Perfosfamide; Pipobroman; Piposulfan; The hydrochloric acid piroxantrone; Plicamycin; Plomestane; Porfimer sodium; Porfiromycin; Prednimustine; Procarbazine hydrochloride; Puromycin; Puromycin hydrochloride; Pyrazofurin; SQ 22558; Rogletimide; SPC 100270; The hydrochloric acid SPC 100270; Semustine; Simtrazene; Sparfosate sodium; Sparsomycin; NSC-192965; Spiromustine; Spiral shell platinum; Streptonigrin; Streptozocin; Sulofenur; Talisomycin; Tecogalan sodium; Tegafur; Teloxandrone hydrochloride; Temoporfin; Teniposide; Teroxirone; Testolactone; ITG; Tioguanine; Plug is for group; Tiazofurine; Win-59075; FC-1157a; Trestolone acetate; The phosphoric acid triciribine; Trimetrexate; The trimetrexate glucuronate; Triptorelin; Tubulozole hydrochloride; Uramustine; Uredepa; Vapreotide; Visudyne; Vinblastine sulphate; Vincristine sulphate; Vindesine; LY-099094; The sulfuric acid vinepidine; The sulfuric acid vinglycinate; The sulfuric acid vinleurosine; Vinorelbine tartrate; The sulfuric acid vinrosidine; The sulfuric acid vinzolidine; Vorozole; Zeniplatin; Zinostatin; NSC-164011.Other anticarcinogen includes but not limited to: 20-shows-1,25 dihydroxy vitamin d3s; 5-ethinyluracil; Abiraterone; Aclarubicin; The acyl group fulvene; The gland cyclopentanol; U 73975; RIL-2; Full TK antagonist; Altretamine; Ambamustine; 2,4 dichlorphenoxyacetic acids; Amifostine; Amino-laevulic acid; Amrubicin; Amsacrine; Anagrelide; Anastrozole; Rographolide; Angiogenesis inhibitor; Antagonist D; Antagonist G; Antarelix; Anti-dorsalization morphogenetic proteins-1; Androgen antagonist, prostate cancer; Estrogen antagonist; Antineoplaston; Antisense oligonucleotide; The glycocoll NSC-234714; The apoptogene regulator; Apoptosis regulator; Apurinic acid; Ara-CDP-DL-PTBA; The l-arginine desaminase; 9-[2-methoxyl group-4-(methyl sulphonyl is amino) phenyl amino]-N, 5-dimethyl--4-acridine methane amide; SH 489; Atrimustine; Axinastatin 1; Axinastatin2; Axinastatin 3; Azasetron; Azalomycin; Azatyrosine; Baccatin III derivative; Barlan alcohol (balanol); BB-94; The BCR/ABL antagonist; The benzo chlorins; Benzoyl-star spore rhzomorph; The beta-lactam verivate; β-alethine; Beta clarithromycin (betaclamycin) B; Betulinic acid; The bFGF suppressor factor; Bicalutamide; Bisantrene; Two '-aziridino spermine; Bisnafide; Two cetiedil citrate A; U 77779; Bu Fulei (breflate); U-54461; Budotitane; BSA; Calcipotriol; Calphostin C; Camptothecin derivative; Canary pox IL-2; Capecitabine; Methane amide-amino-triazole; NSC 609974; CaRest M3; CARN 700; Be derived from the suppressor factor of cartilage; U 80244; Casein SU11752 (ICOS); Castanospermine; Cecropin B; Cetrorelix; Chlorins; The nefrosulfin quinoxaline; ZK 96480; The cis porphyrin; CldAdo; The Clomiphene analogue; Clotrimazole; Kao Lisi mycin (collismycin) A; Kao Lisi mycin B; Combretastatin A4; The combretastatin analogue; Conagenin; Crambescidin 816; Crisnatol; From beads algal rim peptide 8; From beads algal rim peptide A verivate; Curacin A; Encircle penta anthraquinone; Ring platinum (cycloplatam); But match mycin (cypemycin); Cytosine arabinoside octadecyl sodium phosphate; The lysis factor; Hexestryl diphosphate; Dacliximab; NSC 127716; The cyclodepsipeptide of dehydrogenation (dehydrodidemnin) B; Deslorelin; DEXAMETHASONE BP98; Right ifosfamide; Dexrazoxane; Dexverapamil; NSC-182986; Cyclodepsipeptide (didemnin) B; 3,4-dihydroxyl benzo hydroxamic acid (didox); Diethylammonium removes the first spermine; Dihydro-5-azacytidine; Dihydro is safe plain, 9-; Two oxamycins; The xenyl spiromustine; Docetaxel; V-1326; Dolasetron; Doxifluridine; Droloxifene; Dronabinol; Many Ka-7038s (duocarmycin) SA; Ebselen; NSC 609224; Ro 14-5243; Edrecolomab; Eflornithine; Elemenum Emulsion; Emitefur; Epirubicin; SKF-105657; The estramustine analogue; Estrogen agonist; Estrogen antagonist; SR-2508; The phosphoric acid VP; FCE-24304; Fadrozole; NSC 281272; HPR; Filgrastim; Finasteride; Husband's degree of evening up; Flezelastine; The fluoranthene sterone; Fludarabine; Hydrochloric acid fluorine daunomycin; Forfenimex; Formestane; Phosphotrienin; Fotemustine; De Kesafei quinoline gadolinium; Gallium nitrate; Ro 09-1390; RS 26306; The gelatinase suppressor factor; Gemcitabine; The gsh suppressor factor; Thionamic acid 1,7 heptane two basic esters; Heregulin; HMBA; Hypericin; Ibandronic acid; Idarubicin; CB-7432; Idramantone; Thio ALP; Ilomastat; The imidazo dihydroketoacridine; S-26308; The immunostimulant polypeptide; The IGF-1R suppressor factor; The Interferon, rabbit agonist; Interferon, rabbit; Interleukin-; M-iodobenzylguanidine; The iodine Dx; Sweet potato alcohol, 4-; Iroplact; Irsogladine; Foreign country's lattice azoles (isobengazole); Isohomohalicondrin B; U 98079A; Jasplakinolide; Kahalalide F; Nitrilotriacetic sheet spiral shell element-N; Lanreotide; That mycin of thunder (leinamycin); Lenograstim; The sulfuric acid lentinan; Leptolstatin; Letrozole; LIF; The white corpuscle IFN-; Leuprorelin+oestrogenic hormon+progesterone; Leuprorelin; LEVAMISOLE HCL; Liarozole; Linear polyamine analogue; Lipophilic two glycopeptides; Lipophilic platinic compound; Lissoclinamide 7; Lip river platinum; The earthworm propylhomoserin; Lometrexol; Lonidamine; Losoxantrone; Lovastatin; RWJ 21757; OSI 211; De Kesafei quinoline lutetium; 1-((R)-5-hydroxyl hexyl) Theobromine; The cracking polypeptide; Maitansine; Mannostatin A; Marimastat; Masoprocol; Maspin; The stromlysin suppressor factor; NMPI; Menogaril; Mei Balong; Meterelin; Methioninase; Metoclopramide; The MIF suppressor factor; Mifepristone; Miltefosine; Mirimostim; The double-stranded RNA of mispairing; Mitoguazone; Mitolactol; Mitomycin analogs; NSC 300288; Mitotoxin fibroblast growth factor-saporin; Mitoxantrone; Ro 40-8757; Sch-39300; Monoclonal antibody, pregnancy urine extract; Monophosphoryl lipid A+mycobacterium cell walls sk; Mopidamol; The agent of multi-medicine resistance gene inhibition; Treatment based on many tumor inhibitors 1; The Mannomustine carcinostatic agent; Indian Ocean sponge B; The mycobacterium cell wall extracts; Myriaporone; N-ethanoyl Goe 1734; The N-substituted benzamide; Nafarelin; Nagrestip; Nx+pentazocine; Napavin; The naphthalene terpinum; Neu-up 100; S 254; Nemorubicin; Neridronic acid; Proteinase, kidney brush border neutral; RU-23908; Silt mycin (nisamycin); The nitrogen protoxide regulator; The nitroxide inhibitor; Nitrullyn; O6-benzyl guanine; Sostatin; Okicenone; Oligonucleotide; Onapristone; Ondansetron; Ondansetron; Diclofenamide; Oral cytokine induction agent; Ormaplatin; Osaterone; Oxaliplatin; Ao Kesa mycin (oxaunomycin); Taxol; Paclitaxel analogs; D51-7059; Palau amine; The palmityl nitragin; Pamidronic acid; Panoxatriol; Panomifene; The secondary bacterium iron of hexa-coordinate catechu amine iron chelating agent is plain; SR 95225; Pegaspargase; Peldesine; Piperylene gathers sodium sulfate; Pentostatin; Pan arrest azoles (pentrozole); PFOB; Perfosfamide; Sinapyl alcohol; Azophenlyene mycin (phenazinomycin); Toluylic acid; Inhibitors of phosphatases; Dissolve the chain bacterium; The hydrochloric acid pilocarpine; Pirarubicin; Piritrexim; Placetin A; Placetin B; The suppressor factor of plasminogen activator; Platinum complexes; Platinic compound; Platinum-three amine compound; Porfimer sodium; Porfiromycin; Prednisone; Propyl group two-dihydroketoacridine; Prostaglandin(PG) J2; Proteasome inhibitor; Immunomodulator based on albumin A; Inhibitors of protein kinase C; Inhibitors of protein kinase C, little algae; Inhibitors of protein tyrosine phosphatase; Purine nucleoside phosphorylase inhibitor; Purpurin; Pyrazoloacridine; Oxyphorase myocoril T 46155 conjugate; The raf antagonist; ZD-1694; Ranimustine; The ras farnesyl protein transferase inhibitor; The ras suppressor factor; The ras-GAP suppressor factor; The demethyl retelliptine; Etidronic acid rhenium Re 186; Nitragin; Ribozyme; RII dimension A amine; Rogletimide; Rohitukine; Romurtide; Roquinimex; Rubiginone B1; Ruboxyl; SPC 100270; Saintopin; SarCNU; Sa Ke phytol (sarcophytol) A; Sargramostim; The Sdi1 dummy; Semustine; Be derived from old and feeble suppressor factor 1; Positive MODN; Signal transduction inhibitor; Signal transduction modulators; Single chain antigen binding protein; Sizofiran; Sobuzoxane; Sodium borocaptate; Sodium phenylacetate; Solverol; SM-binding protein; Sonermin; Sparfosic acid; But this visits mycin (spicamycin) D; Spiromustine; The spleen pentapeptide; CD-spiro ketal precursor delta-lactone 1; Shark amine; Stem cell inhibitors; The stem cell division suppressor factor; Stipiamide; Stromlysin suppressor factor sulfinosine; Strong vasoactive peptide antagonists of imitating; Suradista; Suramin; Sphaerophysine; The synthetic TGSS C3; Tallimustine; The tamoxifen methiodide; TCNU; Tazarotene; Tecogalan sodium; Tegafur; Tellurapyrylium; Telomerase inhibitor; Temoporfin; TM; Teniposide; Ten oxidation tetrachloros; Four nitrogen amine (tetrazomine); Thalictrine; Thiocoraline; TSF; Thrombopoietin is imitated thing; Thymosin-Alpha1; The thymopoietin receptor stimulant; Thymotrinan; TTH; Ethyl tin is C.I. Natural Red 8 just; Win-59075; Dichloro titanium alkene; Marine alkaloids topsentin; Toremifene; All-round STEMCELLFACTOR; TI; Tretinoin; Triacetyl uridine; Triciribine; Trimetrexate; Triptorelin; Tropisetron; Turosteride; Tyrosine kinase inhibitor; Tyrphostin tyrphostins; The UBC suppressor factor; Ubenimex; Be derived from the GIF of urogenital sinus; The urokinase receptor antagonist; Vapreotide; Variolin B; Carrier system, the red corpuscle gene therapy; Velaresol; Veratramin(e); Verdins; Visudyne; Vinorelbine; Vinfosiltine; Wei Taxin (vitaxin); Vorozole; Zanoterone; Zeniplatin; Zilascorb; And Zinostatin stimalamer.
The embodiment of this paper further is included among the patient treatment or prevention can be through suppressing the disease that PDE4 improves or the method for illness, and it comprises that the patient to this treatment of needs or prevention uses the solid form of one or more inclusion compounds A.Can include but not limited to asthma, inflammation, chronic or acute occlusion property tuberculosis, chronic or acute pulmonary inflammation disease, inflammatory bowel, segmental enteritis, Behcet's disease, colitis, ulcerative colitis and sacroiliitis or the inflammation that causes by perfusion again through suppressing illness that PDE4 improves.In a preferred embodiment, wait to treat or the disease or the illness of preventing are chronic obstructive pulmonary diseases.
Concrete grammar of the present invention can comprise uses extra therapeutical agent, such as but not limited to antiphlogiston, antihistaminic agent and Decongestant.The instance of such additional therapeutic agent includes but not limited to: antihistaminic agent includes but not limited to thanomin, quadrol, piperazine and thiodiphenylamine; Antiphlogiston; Nonsteroid anti-inflammatory drugs (NSAIDS), include but not limited to Frosst), salicylate, PARACETAMOL BP98, indomethacin, sulindac, R-ETODOLAC, fragrant that ester, tolmetin, ketorolac, diclofenac, Ibuprofen BP/EP, Naproxen Base, fenoprofen, KP, FLURBIPROFEN USP24, Taisho), piroxicam, meloxicam, pyrazolone derivative; And steroid, include but not limited to reflunomide and adrenocortical steroid.
Concrete grammar of the present invention is avoided or has been reduced the relevant drug drug interaction of the medicament (comprising racemic substituted phenylethyl sulfone) that in such treatment of conditions, uses and other undesirable action.Be not subject to any theory; The solid form of some inclusion compound A can be with respect to racemic 2-[1-(3-oxyethyl group-4-p-methoxy-phenyl)-2-methyl sulphonyl ethyl]-4-acetylamino isoindoline-1; The 3-diketone comprises that its solid form further provides the result of treatment or the therapeutic index of overall improvement.
As stated, the solid form of some inclusion compound A can be used for treatment or prevention various diseases and illness.In the acute or chronic control of disease or illness, the prevention of given activity composition of the present invention or the amount of therapeutic dose can change with the character of disease or illness and the route of administration of severity and activeconstituents.Perhaps, dosage also has administration frequency, also will change according to age, body weight and the reaction of individual patient.Suitable dosage regimen can be considered these factors and selection easily by those skilled in the art.In general, to the described illness of this present invention, dosage every day of recommendation be every day about 1mg to about 1,000mg gave as single dosage once-a-day in one day, gave preferably as the dosage that separates.More specifically, every day, dosage was to give for twice separate doses every day of equal portions.Especially, every day dosage range can for every day about 5mg to about 500mg, more especially at every day about 10mg and about 200mg.Especially, every day, dosage can be used with the formulation of 5mg, 10mg, 15mg, 20mg, 25mg, 50mg or 100mg.In control during the patient, treatment should be from beginning than low dosage, maybe for about 1mg to about 25mg, and depend on patient's W-response, if be necessary, be increased to every day about 200mg and arrive approximately 1,000mg is as single dose or the dosage that separates.Perhaps, every day, dosage was from 0.01mg/kg to 100mg/kg.
In some cases, it possibly be necessary outside the disclosed dosage range of the present invention, using activeconstituents, and this will be obvious to those of ordinary skills.It should be noted that in addition clinicist or treatment doctor combine the reaction of individual patient, will how and when understand interrupt, adjustment or stopped treatment.
The phrase " treatment significant quantity " that the present invention uses, " prevention significant quantity " and " treatment or prevention significant quantity " comprise above-mentioned dosage and administration frequency timetable.Different treatment significant quantities can be applied to various disease and illness, and this is that those of ordinary skills will understand easily.Similarly; Be enough to treat or prevent such illness; But be not enough to cause or be enough to and reduce and racemic 2-[1-(3-oxyethyl group-4-p-methoxy-phenyl)-2-methyl sulphonyl ethyl]-4-acetylamino isoindoline-1; The amount of the undesirable action that the 3-diketone is relevant is also included within above-mentioned dosage and the administration frequency timetable.
4.3.
Pharmaceutical composition
This paper provides the pharmaceutical composition and the single unit dosage of the solid form that comprises one or more inclusion compounds A.The present invention also provides the pharmaceutical composition of the solid form that comprises one or more inclusion compounds A and the preparation method of single unit dosage.For example; In certain embodiments, comprise solid form provided by the present invention or use the separately formulation of solid form provided by the present invention preparation can be suitable for oral, mucous membrane (comprising rectum, nose or vagina), parenteral (comprise subcutaneous, intramuscular, inject, intra-arterial or intravenously), hypogloeeis, transdermal, oral cavity or topical application.
In certain embodiments, pharmaceutical composition provided by the present invention and formulation contain the solid form of one or more inclusion compounds A.Certain embodiments of the present invention provide the solid form that contains inclusion compound A; For example as; Like the pharmaceutical composition and the formulation of form A, B, C, D, E, F, G or the unformed solid form of inclusion compound A provided by the present invention, wherein the solid form of inclusion compound A is pure basically.Certain embodiments of the present invention provide the solid form that contains inclusion compound A; For example; Pharmaceutical composition and formulation like form A, B, C, D, E, F, G or the unformed solid form of inclusion compound A provided by the present invention; Other solid form that it does not contain inclusion compound A basically for example comprises, like form A, B, C, D, E, F, G and/or the unformed solid form of inclusion compound A provided by the present invention.Certain embodiments of the present invention provide the pharmaceutical composition and the formulation of the mixture of the solid form that contains inclusion compound A, comprise for example comprising following one or more mixture: like form A, B, C, D, E, F and the unformed solid form of inclusion compound A provided by the present invention.Pharmaceutical composition provided by the present invention and formulation also comprise one or more pharmaceutically acceptable vehicle, diluent or carrier usually.
The concrete pharmaceutical composition that is comprised by this embodiment contains solid form and at least a additional therapeutic agent of one or more inclusion compounds A.The instance of additional therapeutic agent includes but not limited to anticarcinogen and anti-inflammatory therapy agent, include but not limited to provided by the present invention those.
That single unit dosage of the present invention is suitable for is oral, mucous membrane (for example nose, hypogloeeis, vagina, oral cavity or rectum), parenteral (for example subcutaneous, intravenously, inject, intramuscular or intra-arterial) or transdermal administration patient.The instance of formulation includes but not limited to: tablet; The capsule sheet; Capsule is soft elastic gelatin capsule for example; Cachet; Lozenge; Lozenge; Dispersion agent; Suppository; Ointment; Paste (poultice); Paste; Powder; Dressing; Emulsifiable paste; Plaster; Solution; Paster; Aerosol (for example nasal spray or inhalation); Gel; Be fit to liquid dosage form oral to the patient or that mucous membrane is used, comprise suspension-s (the for example suspension-s of water or on-aqueous liquid, O/w emulsion or water-in-oil liquid emulsion), solution and elixir; Be fit to liquid dosage form to patient's parenteral administration; And sterile solid (for example crystallization or amorphous solid), it can be by reprovision to provide the liquid dosage form that is fit to patient's parenteral administration.
The compsn of formulation of the present invention, shape and type will depend on its purposes usually and change.For example, compare with the formulation of the chronic treatment that is used for same disease, the formulation that is used for the emergency treatment of inflammation or associated conditions can comprise more substantial one or more activeconstituentss.Similarly, compare with the oral dosage form that is used to treat same disease or illness, parenteral dosage forms can comprise one or more activeconstituentss of less amount.The variation each other of these of the concrete formulation that the present invention is included and other modes will be conspicuous to those skilled in the art.Referring to, for example
Remington's Pharmaceutical Sciences, the 18th edition, Mack Publishing, Easton PA (1990).
Typical pharmaceutical composition and formulation comprise one or more vehicle.Suitable vehicle is that the pharmacy field technician knows, and the limiting examples of suitable vehicle provides in the present invention.Concrete vehicle whether is fit to join pharmaceutical composition or formulation depends on multiple factor well known in the art, includes but not limited to formulation is applied to patient's mode.For example, oral dosage form for example tablet can comprise the vehicle that is not suitable in parenteral dosage forms, using.The suitability of concrete vehicle also can depend on the concrete activeconstituents in the formulation.
Lactose-free compsn of the present invention can comprise vehicle well known in the art and that in for example USP (USP) SP (XXI)/NF (XVI), list.In general, lactose-free compsn comprises activeconstituents, tackiness agent/weighting agent and the lubricant of pharmaceutically compatible and pharmaceutically acceptable amount.Preferred lactose-free formulation comprises activeconstituents, Microcrystalline Cellulose, pregelatinized Starch and Magnesium Stearate.
Because water can promote the degraded of some compounds, so the present invention further comprises anhydrous pharmaceutical composition and the formulation that comprises activeconstituents.For example, in order to confirm for example quality guaranteed period or preparation characteristics such as stability in time, add water (for example 5%) and be the means of the simulation long storage that pharmaceutical field accepts extensively.Referring to, for example, Jens T.Carstensen,
Drug Stability:Principles & Practice, the 2nd edition, Marcel Dekker, NY, NY, 1995,379-80 page or leaf.In fact, water and heat can quicken the decomposition of some compounds.Therefore, water can be significant to the effect of preparation, because in preparation, processing, packing, storage, the transportation of preparation with run into moisture and/or moisture through regular meeting between the usage period.
Anhydrous pharmaceutical composition of the present invention and formulation can be used the composition of anhydrous or low moisture content, under low moisture or low, prepare.Can substantive the contact be arranged with moisture and/or moisture if be expected at preparation, packing and/or lay up period, then comprising lactose and at least a, to contain the pharmaceutical composition and the formulation of activeconstituents of primary amine or secondary amine preferably anhydrous.
Anhydrous pharmaceutical composition should be produced and store to such an extent that its no aqueous nature is held.Therefore, anhydrous compsn preferably uses the known material packing that prevents to be exposed to water, makes them can be contained in the suitable prescription test kit.The instance of suitable packing includes but not limited to seal aluminium foil, plastics, unit-dose container (for example bottle), Blister Package and band packing.
The present invention further comprises pharmaceutical composition and formulation, and it comprises one or more compounds that reduces the activeconstituents rate of decomposition.Such compound is known as " stablizer " in this article, and it includes but not limited to inhibitor for example xitix, pH buffer reagent or salt buffer agent.
The same with the amount and the type of vehicle, the amount of the activeconstituents in the formulation can depend on various factors with particular type and be different, such as but not limited to the approach of using to the patient.But, exemplary dosage form provided by the present invention every day about 1mg to about 1, in the 000mg scope, give in the morning as single dosage once-a-day, but in one day, give preferably as the dosage that separates.More especially, dosage every day every day is with the separate doses administered twice of equal portions.Especially, every day dosage range can for every day about 5mg to about 500mg, more especially for every day about 10mg arrive about 200mg.In control during the patient, treatment can be from beginning than low dosage, maybe for about 1mg to about 25mg, and depend on patient's W-response, if be necessary, be increased to every day about 200mg and arrive approximately 1,000mg is single dose or the dosage that separates.
4.3.1.
Oral dosage form
Be suitable for Orally administered pharmaceutical composition of the present invention and can be used as discrete formulation existence, such as but not limited to tablet (for example masticable tablet), capsule sheet, capsule and liquid (for example seasoning syrup).Such formulation comprises the activeconstituents of predetermined amount, and the preparation of the pharmaceutical methods that can know by one of skill in the art.Generally referring to
Remington's Pharmaceutical Sciences, the 18th edition, Mack Publishing, Easton PA (1990).
Through activeconstituents and at least a vehicle are prepared exemplary oral dosage form of the present invention according to conventional pharmaceutical hybrid technology thorough mixing.According to using required dosage form, vehicle can have various broad form.For example, the vehicle that is applicable to liquid oral or aerosol dosage forms includes but not limited to water, terepthaloyl moietie, oil, alcohol, seasonings, sanitas and tinting material.The instance that is applicable to the vehicle of solid oral dosage form (for example powder, tablet, capsule and capsule sheet) includes but not limited to starch, sugar, Microcrystalline Cellulose, thinner, granulating agent, lubricant, tackiness agent and disintegrating agent.
Because use conveniently, use the tablet and the capsule of solid excipient to represent best oral dosage unit form.If desired, tablet can be through the water-based or the non-aqueous technology coatings of standard.Such formulation can be through any pharmaceutical methods preparation.In general, through activeconstituents and liquid vehicle, fine solid carrier or both are evenly mixed to come pharmaceutical compositions and formulation fully,, process required shape to product if be necessary then.
For example, can be through compacting or the molded tablet for preparing.Can randomly prepare the tablet of compacting through compacting in suitable machine with the activeconstituents that is in free-flowing form (for example powder or particle) of mixed with excipients.Can prepare molded tablet through molded mixture in suitable machine by the wetting powder compounds of inert liquid diluent.
The instance of the vehicle that can in oral dosage form of the present invention, use includes but not limited to tackiness agent, weighting agent, disintegrating agent and lubricant.The tackiness agent that is adapted at using in pharmaceutical composition and the formulation includes but not limited to: W-Gum; Potato starch; Or other starch; Gelatin; Natural and synthetic glue is Sudan Gum-arabic for example; Sodiun alginate; Lalgine; Other alginate; The powder tragacanth gum; Guar gum; Mierocrystalline cellulose and verivate thereof (TKK 021 for example; FM; ECG-505; Xylo-Mucine); Vinylpyrrolidone polymer; Methylcellulose gum; Pregelatinized Starch; HPMC (for example 2208; 2906; No. 2910); Microcrystalline Cellulose; And composition thereof.
The instance of the weighting agent that is adapted at using in disclosed pharmaceutical composition of the present invention and the formulation includes but not limited to: talcum, lime carbonate (for example particle or powder), Microcrystalline Cellulose, cellulose powder, VISOSE, kaolin, N.F,USP MANNITOL, silicic acid, Sorbitol Powder, starch, pregelatinized Starch, and composition thereof.Tackiness agent in the pharmaceutical composition of the present invention or weighting agent exist to about 99% weight with about 50 of pharmaceutical composition or formulation usually.
The suitable form of Microcrystalline Cellulose includes but not limited to as AVICEL-PH-101
TM, AVICEL-PH-103
TM, AVICEL RC-581
TM, AVICEL-PH-105
TMThe material that (can be from FMC Corporation, American Viscose Division, Avicel Sales, Marcus Hook, PA obtains) sells, and composition thereof.Concrete tackiness agent is as AVICEL RC-581
TMThe Microcrystalline Cellulose of selling and the mixture of Xylo-Mucine.Suitable anhydrous or low-moisture vehicle or additive comprise AVICEL-PH-103
TMWith starch 1500LM
TM
The tablet of disintegration when in compsn of the present invention, using disintegrating agent to be exposed to aqueous environments to be provided at.The tablet that comprises too much disintegrating agent maybe disintegration in storage, may not be and comprise the tablet of very few disintegrating agent with required speed disintegration, or can not disintegration under required condition.Therefore, should use the disintegrating agent of capacity to form solid oral dosage form of the present invention, this amount is exceeded few only, can not change the release of activeconstituents nocuously.The amount of the disintegrating agent that uses changes based on the type of preparation, and is those of ordinary skills' identifications easily.Typical pharmaceutical composition comprises about 0.5 disintegrating agent to about 15% weight, particularly about 1 disintegrating agent to about 5% weight.
The disintegrating agent that can in pharmaceutical composition of the present invention and formulation, use includes but not limited to: agar, Lalgine, lime carbonate, Microcrystalline Cellulose, cross-linked carboxymethyl cellulose sodium, PVPP, Polacrilin potassium, sodium starch glycollate, potato or tapioca(flour), pregelatinized Starch, other starch, clay, other algins, other Mierocrystalline celluloses, glue, and composition thereof.
The lubricant that can in pharmaceutical composition of the present invention and formulation, use includes but not limited to: calcium stearate, Magnesium Stearate, MO, light mineral oil, glycerine, Sorbitol Powder, N.F,USP MANNITOL, polyoxyethylene glycol, other terepthaloyl moietie, Triple Pressed Stearic Acid, sodium lauryl sulphate, talcum, Wecobee M (for example peanut oil, Oleum Gossypii semen, sunflower seed oil, til, sweet oil, Semen Maydis oil and soya-bean oil), Zinic stearas, OE, Laurate ethyl, agar, and composition thereof.Extra lubricant comprises for example syloid silica gel (AEROSIL200
TM, W.R.Grace company, Baltimore, MD preparation), the solidified gas colloidal sol (TX sells for Degussa company, Piano) of synthetic silica, CAB-O-SIL
TM(Cabot company, Boston, the silica product that the high heat that MA sells is processed), and composition thereof.If use fully, lubricant uses to account for the pharmaceutical composition that comprises it or the amount that is less than about 1% weight of formulation usually.
4.3.2.
The time-delay release dosage form
Can use the solid form of inclusion compound A provided by the present invention through the delivery apparatus that controlled-release device or those of ordinary skills know.Instance includes but not limited at U.S. Patent number 3,845 770,3,916,899,3,536,809,3,598; 123, and 4,008,719,5,674,533,5,059; 595,5,591,767,5,120,548,5,073,543,5; Those that describe in 639,476,5,354,556 and 5,733,566, it incorporates this paper separately by reference into.Can use such formulation to come one or more activeconstituentss are provided slowly or sustained release, it uses for example Vltra tears, other polymeric matrixs, gel, permeable film, osmosis system, multiple coatings, particulate, liposome, microsphere or its of different ratios to make up the release characteristic that provides required.Can easily select the known suitable controlled release preparation of those of ordinary skills (comprise described in the invention those), use with activeconstituents of the present invention being used for.Therefore, the present invention includes and be suitable for Orally administered single unit dosage, such as but not limited to the tablet, capsule, soft capsule and the capsule sheet that are fit to controlled release.
The medicament prodn of all controlled releases all has common objective, promptly improves the pharmacological agent that can reach with respect to its non-controlled release equivalent.Ideally, in therapeutic treatment, use the controlled release preparation of optimum design to be characterised in that the medicine that uses minimum cures or control illness in the shortest time.The advantage of controlled release preparation comprises the pharmaceutical activity of prolongation, the administration frequency of reduction and the patient's compliance that increases.In addition, controlled release preparation can be used to time or other characteristics of influence onset, the blood concentration of medicine for example, and therefore can influence the generation of spinoff (for example undesirable action).
Most of controlled release preparations are designed to discharge a certain amount of medicine (activeconstituents) at first, and it promptly produces required result of treatment, and progressively and continuously discharges the medicine of other amounts, in the time period that prolongs, to keep the treatment or the preventive effect of this level.In order to keep this constant level of medicine in vivo, medicine must discharge with given pace from formulation, and this speed will be replaced the medication amount of discharging by metabolism with in the body.The controlled release of activeconstituents can include but not limited to pH, temperature, enzyme, water or other physiological conditions or compound through various conditioned stimulus.
4.3.3.
Parenteral dosage forms
Parenteral dosage forms can be used to the patient by all means, includes but not limited to subcutaneous, intravenously (comprise and injecting), intramuscular and intra-arterial.Because using of parenteral dosage forms got around the natural defence of patient to pollutent usually, so they are preferably aseptic or can before the patient uses, sterilized.The instance of parenteral dosage forms includes but not limited to injection solution, is suitable in pharmaceutically acceptable vector, dissolving or suspending for power-product, injection suspension-s and the emulsion of injection.
The suitable vector that can be used to provide parenteral dosage forms of the present invention is well known to those skilled in the art.Instance includes but not limited to: USP water for injection; The water-based vector is such as but not limited to sodium chloride injection, ringer's injection, Vadex injection liquid, Vadex and sodium chloride injection and lactic acid salt ringer's injection; Be prone to water blended vector such as but not limited to ethanol, polyoxyethylene glycol and W 166; And non-aqueous vector is such as but not limited to Semen Maydis oil, Oleum Gossypii semen, peanut oil, til, OE, Isopropyl myristate and phenylamino benzoic acid methyl esters.
The compound that also can in parenteral dosage forms of the present invention, add the solubleness that improves disclosed one or more activeconstituentss of the present invention.
4.3.4.
Transdermal, part and mucous membrane formulation
Transdermal of the present invention, part and mucous membrane formulation include but not limited to ophthalmic solution, sprays, aerosol, emulsifiable paste, washing lotion, ointment, gel, solution, emulsion, suspension-s or other form well known by persons skilled in the art.Referring to, for example
Remington's Pharmaceutical Sciences, the 16th and 18 edition, Mack Publishing, Easton PA (1980&1990); With
Introduction to Pharmaceutical Dosage Forms, the 4th edition, Lea & Febiger, Philadelphia (1985).The formulation that is fit to the intraoral mucous membrane tissue of treatment can be configured to mouth wash shua or oral gel.In addition, the transdermal formulation comprises " storage type " or " matrix type " paster, and they can be applied to skin, and adheres to one section specified time, to allow the activeconstituents infiltration of aequum.
The material that suitable vehicle (for example carrier and thinner) and other can be used to provide transdermal, part and mucous membrane formulation that the present invention comprises is that the pharmacy field technician knows, and depends on the concrete tissue that given pharmaceutical composition or formulation will be applied to.Consider this fact; Typical vehicle includes but not limited to water, acetone, ethanol, terepthaloyl moietie, Ucar 35,1; 3-butyleneglycol, Isopropyl myristate, Wickenol 111, MO, and composition thereof, to form nontoxic and pharmaceutically acceptable washing lotion, tincture, emulsifiable paste, emulsion, gel or ointment.If desired.Can also in pharmaceutical composition and formulation, add wetting Agent for Printing Inks or wetting agent.The instance of such supplementary component is well known in the art.Referring to, for example
Remington's Pharmaceutical Sciences, the 16th and 18 edition, Mack Publishing, Easton PA (1980&1990).
Depend on concrete tissue to be treated, can be before, simultaneously or use extra composition afterwards with activeconstituents of the present invention treatment.For example, penetration enhancers can be used for auxiliary to organizing delivering active ingredients.Suitable penetration enhancers includes but not limited to: acetone; Various alcohol are ethanol, oleyl alcohol and tetrahydrofurfuryl carbinol for example; Alkyl sulfoxide is DMSO 99.8MIN. for example; N,N-DIMETHYLACETAMIDE; N; Polyoxyethylene glycol; Pyrrolidone is Vinylpyrrolidone polymer for example; The Kollidon of different grades (Vinylpyrrolidone polymer, polyvidone); Urea; With various water solubles or insoluble sugar ester, for example Tween 80
TM(polysorbate80) and Span 60
TM(sorbitan monostearate).
Also can regulate the pH value of pharmaceutical composition or formulation, the perhaps pH value of pharmaceutical composition or the applied tissue of formulation is to improve sending of one or more activeconstituentss.Similarly, polarity, its ionic strength or the tension force that can regulate solvent carrier are sent with improvement.Compound for example stearate also be introduced in pharmaceutical composition or the formulation, with wetting ability or the lipotropy that advantageously changes one or more activeconstituentss, sends thereby improve.In this, stearate can serve as lipid vector, emulsifying agent or the tensio-active agent of preparation and send and improve or penetration enhancers.The different solid forms that comprise activeconstituents can be used to further regulate the character of the compsn that obtains.
4.3.5.
Test kit
The present invention comprises test kit, and it can simplify an amount of activeconstituents using the patient when being used by the medical practitioner.
Typical agents box of the present invention comprises the compd A of unit dosage or second activeconstituents of its pharmaceutically acceptable solid form or prodrug and unit dosage.The instance of second activeconstituents includes but not limited to those that the present invention is listed.
Test kit of the present invention can further comprise the device that is used for using activeconstituents.The instance of such device includes but not limited to syringe, dropping liquid bag, paster and sucker.
Test kit of the present invention can further comprise the pharmaceutically acceptable vector that can be used for using one or more activeconstituentss.For example; If activeconstituents provides with the solid form that must prepare again for parenteral administration; Test kit can comprise the suitable vector of a sealed vessel so, and activeconstituents can dissolve to form the sterile solution of the no particulate that is fit to parenteral administration therein.The embodiment of pharmaceutically acceptable vector includes but not limited to: USP water for injection; The water-based vector is such as but not limited to sodium chloride injection, ringer's injection, Vadex injection liquid, Vadex and sodium chloride injection and lactic acid salt ringer's injection; Be prone to water blended vector such as but not limited to ethanol, polyoxyethylene glycol and W 166; And non-aqueous vector is such as but not limited to Semen Maydis oil, Oleum Gossypii semen, peanut oil, til, OE, Isopropyl myristate and phenylamino benzoic acid methyl esters.
5.
Embodiment
The application is with U.S. Patent number 6,962, and the full text of 940 (mandates on November 8th, 2005) is incorporated this paper by reference into, comprises the embodiment that wherein provides.
5.1.
Embodiment 1:2-[1-(3-oxyethyl group-4-p-methoxy-phenyl)-2-methyl sulphonyl ethyl]-4-acetylamino isoindoline-1,3-diketone synthetic
(1.0g is 3.7mmol) with 3-kharophen Tetra hydro Phthalic anhydride (751mg, 3.66mmol) the heating 15h under refluxing of the stirred solution in acetic acid (20mL) with 1-(3-oxyethyl group-4-p-methoxy-phenyl)-2-methyl sulphonyl ethamine.Remove solvent in a vacuum, output oil.The oil that obtains is yellow solid (1.0g, productive rate 59%): mp, 144 ° of C through chromatogram output product;
1H NMR (CDCl
3) δ: 1.47 (t, J=7.0Hz, 3H, CH
3), 2.26 (s, 3H, CH
3), 2.88 (s, 3H, CH
3), 3.75 (dd, J=4.4,14.3Hz, 1H, CH), 3.85 (s, 3H, CH3), 4.11 (q, J=7Hz, 2H, CH
2), 5.87 (dd, J=4.3,10.5Hz, 1H, NCH), 6.82-6.86 (m, 1H, Ar), 7.09-7.11 (m, 2H, Ar), 7.47 (d, J=7Hz, 1H, Ar), 7.64 (t, J=8Hz, 1H, Ar), 8.74 (d, J=8Hz, 1H, Ar), 9.49 (br s, 1H, NH);
13C NMR (CDCl
3) δ: 14.61,24.85,41.54,48.44,54.34,55.85,64.43,111.37,112.34,115.04,118.11,120.21,124.85,129.17,130.96,136.01,137.52,148.54,149.65,167.38,169.09,169.40; C
22H
24NO
7The analytical calculation value of S: C, 57.38; H, 5.25; N, 6.08.Observed value: C, 57.31; H, 5.34; N, 5.83.
5.2.
Embodiment 2: (+) 2-[1-(3-oxyethyl group-4-p-methoxy-phenyl)-2-methyl sulphonyl ethyl]-4-acetylamino isoindoline-1,3-diketone synthetic
The preparation of 3-aminophthalic acid
With 10%Pd/C (2.5g), 3-nitrophthalic acid (75.0g, 355mmol) and ethanol (1.5L) under nitrogen atmosphere, install in the 2.5L Parr hydrogenator.Filling hydrogen reaches 55psi in reaction vessel.Mixture vibration 13 hours, keep simultaneously hydrogen pressure 50 and 55psi between.Release hydrogen is with purging with nitrogen gas mixture 3 times.Suspension-s filters through bed of diatomaceous earth, and uses washed with methanol.Filtrating concentrates in a vacuum.The solid that obtains is pulp again in ether, and passes through isolated by vacuum filtration.The solid dried in vacuum provides 54g (productive rate 84%) the 3-aminophthalic acid to constant weight, is yellow product.
1H-NMR(DMSO-d6)δ:3.17(s,2H),6.67(d,1H),6.82(d,1H),7.17(t,1H),8-10(br,s,2H);
13C-NMR(DMSO-d6)δ:112.00,115.32,118.20,131.28,135.86,148.82,169.15,170.09。
The preparation of 3-kharophen Tetra hydro Phthalic anhydride
In the 1L 3-neck round-bottomed flask that has been equipped with mechanical stirrer, TM and condensing surface, pack into the 3-aminophthalic acid (108g, 596mmol) and diacetyl oxide (550mL).Reaction mixture reflux 3 hours is cooled to about 25 ° of C, and further is cooled to 0-5 ° of C, keeps other 1 hour.Collect crystalline solid through vacuum filtration, and wash with ether.Solid product at room temperature dried in vacuum obtains 75g (productive rate 61%) 3-kharophen Tetra hydro Phthalic anhydride to constant weight, is white product.
1H-NMR(CDCl
3)δ:2.21(s,3H),7.76(d,1H),7.94(t,1H),8.42(d,1H),9.84(s,1H)。
2-(the heavily dissolving of 3-oxyethyl group-4-p-methoxy-phenyl-1-(methyl sulphonyl)-second-2-base amine
2-(3-oxyethyl group-4-p-methoxy-phenyl)-1-(methyl sulphonyl)-second-2-base amine (137.0g packs in the 3L 3-neck round-bottomed flask that has been equipped with mechanical stirrer, TM and condensing surface; 500mmol), N-ethanoyl-L-leucine (52g, 300mmol) and methyl alcohol (1.0L).The slurry reflux that stirs 1 hour.Let stirred mixture be cooled to room temperature, and at room temperature continue to stir other 3 hours.Cross filter pulp, and wash with methyl alcohol (250L).Air-dry solid, at room temperature dried in vacuum obtains 109.5g (productive rate 98%) crude product (85.8%ee) to constant weight then.Rough solid (55.0g) and methyl alcohol (440mL) were refluxed 1 hour, be cooled to room temperature, and at room temperature stirred other 3 hours.Cross filter pulp, with methyl alcohol (200mL) washing leaching cake.Air-dry solid, then 30 ° of C dried in vacuum to constant weight, output 49.6g (recovery 90%) (S)-2-(3-oxyethyl group-4-p-methoxy-phenyl)-1-(methyl sulphonyl)-second-2-base amine-N-ethanoyl-L-leucine salt (98.4%ee).Chirality HPLC (1/99EtOH/20mM KH
2PO
4PH 7.0, Ultron chirality ES-OVS, and from Agilent Technologies, 150mm * 4.6mm, the 0.5mL/ branch, 240nm): 18.4 minutes (S-isomer, 99.2%), 25.5 minutes (R-isomer, 0.8%).
The preparation of compd A
500mL 3-neck round-bottomed flask is equipped with mechanical stirrer, TM and condensing surface.(S)-2-(3-oxyethyl group-4-p-methoxy-phenyl)-1-(methyl sulphonyl)-second-2-base amine N-ethanoyl-L-leucine salt (25g packs in reaction vessel; 56mmol; 98%ee), 3-kharophen Tetra hydro Phthalic anhydride (12.1g, 58.8mmol) and Glacial acetic acid min. 99.5 (250mL).Mixture refluxes and spends the night, and is cooled to < 50 ° of C then.Remove solvent in a vacuum, resistates dissolves in ETHYLE ACETATE.The solution with water that obtains (250mL * 2), saturated NaHCO
3The aqueous solution (250mL * 2) and salt solution (250mL * 2) washing, and use dried over sodium sulfate.Evaporating solvent in a vacuum, resistates is with the dual solvent recrystallization that contains ethanol (150mL) and acetone (75mL).Solid passes through isolated by vacuum filtration, and washs with ethanol (100mL * 2).Product to constant weight, provides 19.4g (productive rate 75%) S-{2-[1-(3-oxyethyl group-4-p-methoxy-phenyl)-2-methyl sulphonyl ethyl]-4-kharophen isoindoline-1, the 3-diketone 60 ° of C dried in vacuum }, 98%ee.Chirality HPLC (15/85EtOH/20mM KH
2PO
4PH 5, Ultron chirality ES-OVS, and from Agilent Technology, 150mm * 4.6mm, the 0.4mL/ branch, 240nm): 25.4 minutes (S-isomer, 98.7%), 29.5 minutes (R-isomer, 1.2%).
1H-NMR(CDCl
3)δ:1.47(t,3H),2.26(s,3H),2.87(s,3H),3.68-3.75(dd,1H),3.85(s,3H),4.07-4.15(q,2H),4.51-4.61(dd,1H),5.84-5.90(dd,1H),6.82-8.77(m,6H),9.46(s,1H);
13C-NMR(DMSO-d6)δ:14.66,24.92,41.61,48.53,54.46,55.91,64.51,111.44,112.40,115.10,118.20,120.28,124.94,129.22,131.02,136.09,137.60,148.62,149.74,167.46,169.14,169.48。
Figure 29 provides the reaction scheme of the preparation of (+) enantiomorph that shows compd A.
5.3.
Embodiment 3:TNF-α suppresses
LPS inductive TNF-α test in people's whole blood
Except the whole blood that uses fresh extraction substitutes the PBMC, adopt the LPS inductive TNF-α test among the human PBMC as mentioned below equally to measure the ability that compound suppresses people's whole blood LPS inductive TNF-α generation basically.(Muller etc., 1999, Bioorg.&Med.Chem.Lett., 9:1625-1630.) to compd A, people's whole blood LPS inductive TNF-α IC
50=294nM.
LPS inductive serum TNF in the mouse-α suppresses
Compound according to the previous method of describing (Corral etc., 1996, MoI.Med. 2:506-515), tests in this animal model.To compd A, LPS inductive serum TNF-α suppresses (ED in the mouse
50, mg/kg, oral)=0.05.
LPS inductive TNF-α produces
LPS (LPS) is that it has induced many pro-inflammatory cytokines to comprise the generation of TNF-α by the Gram-negative bacteria intracellular toxin produced of intestinal bacteria (E.coli) for example.In PMBC (PBMC), be derived from monocyte as the TNF-α that replying of LPS produced, it has formed about 5-20% of total PBMC.The ability that test compounds inhibition human PBMC's LPS inductive TNF-α produces as previous description (Muller etc., 1996, J.Med.Chem., 39:3238).PBMC from normal donor passes through Ficoll Hypaque (Pharmacia, Piscataway, NJ, USA) Density Gradient Centrifugation acquisition.Cell is replenishing 10%AB ± human serum (Gemini Bio-products; Woodland; CA, USA), RPMI (Life Technologies, the Grand Island of 2mM L-glutaminate, 100U/ml penicillium mould and 100 μ g/ml Streptomycin sulphates (Life Technologies); NY, USA) the middle cultivation.
With PBMC (2 * 10
5Individual cell) be inoculated into 96 holes flat Costar tissue culturing plate (Corning, NY, USA) in, three parts of every formulas.Cell compound lack or in the presence of with 100ng/ml LPS (Sigma, St.Louis, MO, USA) stimulation.(NJ USA) is dissolved among the DMSO (Sigma) for Celgene Corp., Warren, and further dilution in substratum immediately before use with compound.Whole DMSO concentration in all samples is 0.25%.Stimulated last hour at LPS, compound is added in the cell.Cell at 37 ° of C at 5%CO
2Under cultivated 18-20 hour, collect supernatant then, with substratum dilution, and through ELISA (Endogen, Boston, MA, USA) test TNF-alpha levels.To compd A, LPS inductive TNF-α IC
50=77nM.
The beta induced TNF-α of IL-1 produces
During the process of inflammatory diseases, TNF-α often produces to be stimulated by cytokine IL-1 β, and is not stimulated by the LPS of bacterial origin.The ability that test compounds inhibition human PBMC's the beta induced TNF-α of IL-1 produces as above-mentioned LPS inductive TNF-α produces, difference is that PBMC is white corpuscle unit (Sera-Tec Biologicals, North Brunswick from the source; NJ; USA) pass through with Ficoll-Paque Plus (Amersham Pharmacia, Piscataway, NJ; USA) centrifugal and isolating, and with 3 * 10
5Individual cells/well is inoculated into the RPMI-1640 substratum (BioWhittaker that comprises 10% heat-inactivated foetal calf serum (Hyclone), 2mM L-glutaminate, 100U/ml penicillium mould and 100mg/ml Streptomycin sulphate (perfect medium) in the 96 hole tissue culturing plates; Walkersville, Maryland, USA) in; With 10,2,0.4,0.08,0.016,0.0032,0.00064 and 0 μ M compound; Two parts of every formulas, whole DMSO concentration is 0.1%, in the incubator of 37 ° of C in humidity at 5%CO
2Following pre-treatment one hour, the people IL-1 β (Endogen) with the 50ng/ml reorganization stimulated 18 hours then.To compd A, the TNF-α IC that IL-is beta induced
50=83nM.
5.4.
Embodiment 4:PDE selectivity
PDE1,2,3,5 and 6 enzyme tests
Compound to the specificity of PDE4 through in single concentration (10 μ M) down to ox PDE1, people PDE2, PDE3 with from PDE5 (Hidaka and Asano, 1976, the Biochem.Biophys.Acta of human blood platelets; 429:485, and Nicholsen etc., 1991; Trends Pharmaco.Sci. is 12:19) and from PDE6 (Baehr etc., 1979 of the outer section of bovine retina columnar cell; J.Biol.Chem., 254:11669 and Gillespie etc.; 1989, MoI.Pharm., 36:773) test is assessed.The result enumerates in table 1.
The PDE7 enzyme test
PDE7 is cAMP selectivity PDE, mainly in T cell and Skelettmuskel, expresses.The cytokine of T cell source for example IL-2 and IFN-γ can suppress to regulate potentially through PDE7.As PDE7 such as the previous description through anion-exchange chromatography from Hut78 human T-cell purifying (Bloom and Beavo, 1996, Proc.Natl.Acad.Sci.USA, 93:14188-14192).As in the table 1 PDE4 being described, compound tests to the PDE7 that in the presence of 10nM cAMP, prepares.
5.5.
Embodiment 5:PDE4 suppresses
PDE4 (being derived from the U937 cell) enzyme test
PDE4 enzyme such as previous description through gel filtration chromatography from U937 person monocytic cell purifying (Muller etc., 1998, Bioorg.&Med.Chem.Lett.8:2669-2674).At 30 ° of C at 50mM Tris HClpH 7.5,5mM MgCl
2, 1 μ M cAMP, 10nM [
3H]-carried out the phosphodiester enzyme reaction 30 minutes among the CAMP, through boiling termination, handle with the 1mg/ml snake venom, use AG-IXS ion exchange resin (BioRad) to separate like (Muller etc., 1998, Bioorg.& Med.Chem.Lett.8:2669-2674) as describing.Reaction consumes less than 15% available substrates.The result enumerates in table 1.
Table 1.PDE specificity
* compd B is compd A (-) enantiomorph.
5.6.
Embodiment 6: human T-cell's test
SEB inductive IL-2 and IFN-γ produce
SEB (SEB) is the superantigen that is derived from gram-positive microorganism streptococcus aureus (Staphylococcus aureus).The physiological stimulation that the SEB specificity facilitates to the T cell of expressing specific T cells acceptor V β chain.White corpuscle unit separates human PBMC's (the T cell by about 50% is formed) as stated from the source, and with 3 * 10
5Individual cells/well is inoculated in the perfect medium in the 96 hole tissue culturing plates, with 10,2,0.4,0.08,0.016,0.0032,0.00064 and 0 μ M compound, two parts of every formulas, whole DMSO concentration is 0.1%, in the incubator of 37 ° of C in humidity at 5%CO
2Following pre-treatment 1 hour, (MO USA) stimulated 18 hours for Sigma Chemical Co., St.Louis to use 100ng/ml SEB then.(MN USA) measures IL-2 and IFN-γ level for R&D Systems, Minneapolis through ELISA.To compd A, IL-2IC
50=291nM.To compd A, IFN-γ IC
50=46nM.
5.7.
Embodiment 7:cAMP lifting test
PGE
2Inductive cAMP promotes
PGE
2(PGE
2) with monocyte, T cell and other white corpuscle on the prostanoid receptors bind, and therefore promoted intracellular cAMP level, thereby caused the inhibition of cell response.PGE
2With the collaborative cAMP of the lifting level in these cell types that is combined in of PDE4 suppressor factor, and at PGE
2Exist down, the lifting of the cAMP that the PDE4 suppressor factor causes in PBMC is directly proportional with the inhibition activity of this PDE4 suppressor factor.In the human PBMC, intracellular cAMP measures as follows: PBMC separates as stated, and with 1 * 10
6The every hole of individual cell is inoculated among the RPMI-1640 in 96 orifice plates.Cell in the DMSO of final concentration 2%, with 100,10,1,0.1,0.01 and 0 μ M compound, two parts of every formulas, in the incubator of 37 ° of C in humidity at 5%CO
2Following pre-treatment one hour.Cell is used PGE then
2(10 μ M) (Sigma) stimulated 1 hour.Cell is used the HCl cracking, final concentration 0.1N, and with the inhibition phosphodiesterase activity, and plate is freezing at-20 ° of C.The cAMP that produces uses cAMP (low pH) immunoassay kit (R&D Systems) to measure.For racemoid, PBMC cAMP EC
50Be 3.09 μ M.To compd A, PBMC cAMP EC
50Be 1.58 μ M.
CAMP in people's neutrophilic granulocyte promotes following the measurement.Through centrifugal, from source white corpuscle (Sera-Tec Biologicals), remove PBMC with Ficoll-Paque Plus (Amersham Pharmacia).Red corpuscle/the polymorphonuclear cell that obtains (PMN) group is resuspended in Hank's balanced salt solution (BioWhittaker), and mixes with 3%DextranT-500 (Amersham Pharmacia) in isopyknic 0.9% salt solution.Make erythroprecipitin 20 minutes, and removed PMN, and 4 ° of C at 120rpm centrifugal 8 minutes.The cracking 30 seconds in 0.2% cold salt solution of remaining red corpuscle adds equal-volume 1.6% salt solution, lets cellular-restoring etc. ooze.PMN was 4 ° of C at 1200rpm centrifugal 8 minutes, and is resuspended in RPMI-1640 then, and as above to the lifting of the said test of PBMC cAMP.Through (CA USA) goes up the execution flow cytometry, finds that PMN is about 74%CD18/CD11b for Becton Dickinson, San Jose at FACSCalibur
+, 71%CD16
+CD9
+Neutrophilic granulocyte.The result shows in table 2.
FMLF inductive LTB4 produces
N-formyl radical-methionine(Met)-Ile-Phe (fMLF) is the polypeptide of bacterial origin; Its activate neutrophilic granulocyte quick degranulationization, move, adhere on the endotheliocyte and discharge leukotrienes LTB4, it is arachidonic acid metabolite and self is the neutrophilic granulocyte chemoattractant.As discussed previously (Hatzelmann and Schudt, 2001, J.Pharm.Exp.Ther 297:267-279) and below doing changes, and test compounds hinders the ability that fMLF inductive neutrophilic granulocyte LTB4 produces.Neutrophilic granulocyte such as above-mentioned separation, resuspended in the phosphate buffered saline buffer of calcic or magnesium (Bio Whittaker) not, this damping fluid comprises 10mM HEPES, and pH is 7.2, and with concentration 1.7 * 10
6Individual cells/well is inoculated in the 96 hole tissue culturing plates.Cell is at 37 ° of C 5%CO
2With 50 μ M Thiomersalate (Sigma)/1mM CaCl
2/ 1mM MgCl
2Handled 15 minutes, and used 1000,200,40,8,1.6,0.32,0.064 and the 0nM compound then, whole DMSO concentration is 0.01%, and two parts of every formulas were handled 10 minutes.Neutrophilic granulocyte stimulated 30 minutes with 1 μ M fMLF, then through adding methyl alcohol (final concentration 20%) cracking, and in dry ice/isopropanol is bathed freezing 10 minutes.Split product is stored at-70 ° of C, up to measuring LTB4 content through competitive LTB4ELISA (R&D Systems).The result shows in table 2.
Zymosan inductive IL-8 produces
The yeast saccharomyces cerevisiae of zymosan A or heat killed (Saccharomyces cerevisiae) combines with the lip-deep adhesion molecule Mac-1 of neutrophilic granulocyte and causes phagolysis, cell activation and IL-8 to produce.That zymosan inductive IL-8 produces is as discussed previously (Au etc., 1998, Brit.J.Pharm. 123:1260-1266) measures and does following change.People's neutrophilic granulocyte is purifying as stated, with 3 * 10
5Individual cells/well is inoculated in the perfect medium in the 96 hole tissue culturing plates, with 10,2,0.4,0.08,0.016,0.0032,0.00064 and 0 μ M compound, and two parts of every formulas, whole DMSO concentration is 0.1%, at 37 ° of C 5%CO
2Handled 1 hour.Neutrophilic granulocyte uses the zymosan A (Sigma) that boiled without opsonification with 2.5 * 10 then
5Individual particulate/hole stimulated 18 hours.The results supernatant, and through ELISA (R&D Systems) test I L-8.The result shows in table 2.
FMLF inductive CD18/CD11b expresses
The CD18/CD11b of neutrophilic granulocyte (Mac-1) expresses (Derian etc., 1995, J.Immunol., 154:308-317) measurement, and change below doing as discussed previously.Neutrophilic granulocyte separates as stated, then in perfect medium with 1 * 10
6Individual cell/ml is resuspended, with 10,1,0.1,0.01 and 0 μ M compound, and two parts of every formulas, whole DMSO concentration is 0.1%, at 37 ° of C 5%CO
2Pre-treatment 10 minutes.Cell stimulated 30 minutes with 30nM fMLF then, was cooled to 4 ° of C then.Cell is with rabbit igg (Jackson ImmunoResearch Labs, West Grove, PA, USA) (10 μ g/1x10
6Individual cell) handles,,, and on FACSCalibur, pass through flow cytometry with CD18-FITC and CD11b-PE (Becton Dickinson) dyeing with blocking-up Fc acceptor.CD18/CD11b when from all samples, deducting stimulation and not existing expresses (mean fluorecence), suppresses curve and calculates IC obtaining
50Value.The result shows in table 2.
The fMLF inductive is to the adhesion of HUVEC
Human umbilical vein endothelial cells (HUVEC) as discussed previously (Derian etc., 1995, J.Immunol. 154:308-317) as the adherent substrate of neutrophilic granulocyte, and changes below doing.(Cedar Knolls, NJ USA) obtain the HUVEC cell, and neutrophilic granulocyte cytochalasin B of no use is handled by Anthrogenesis.Cell was with 10,1,0.1,0.01,0.001 and 0 μ M compound treatment 10 minutes, and whole DMSO concentration is 0.1%, two parts of every formulas; Stimulated 30 minutes with 500nM fMLF, and with after the PBS washed twice, (VT USA) goes up measurement fluorescence for Bio-Tek Instruments, Winooski to read the plate device at FLX800.The result shows in table 2.
Table 2. test-results
| People's neutrophilic granulocyte test (all values is in nM) | Racemic compound | Compd A |
| PGE 2Inductive cAMP EC 50 | 12589 | ?4570 |
| FMLF inductive LTB4IC 50 | 20.1 | ?2.48 |
[0283]
| Zymosan inductive IL-8IC 50 | ND | 94 |
| FMLF inductive CD18 expresses IC 50 | ND | 390 |
| FMLF inductive CD11b expresses IC 50 | ND | 74 |
| The fMLF inductive is to the adhesion IC of HUVEC 50 | ND | 150 |
5.8.
Embodiment 8: water solubility
Equilibrium solubility is measured in the buffered soln of pH 7.4.The damping fluid of pH 7.4 passes through with 10NNaOH 0.07M NaH
2PO
4The pH regulator to 7.4 of solution prepares.The ionic strength of solution is 0.15.Will be at least the 1mg powder combine with the 1ml damping fluid, to prepare>mixture of 1mg/ml.The vibration of these samples>2 hours, and be allowed to condition at hold over night under the room temperature.Then through filtering sample with 0.45 saturated μ m nylon syringe strainer of sample earlier.Twice of filtrating serial sampling.Preparation standard in 50% methyl alcohol is through the HPLC filtrates tested.The water solubility of compd A is bigger 3.5 times than racemic mixture.The solubleness of measuring: compd A=0.012mg/mL; Racemic mixture=0.0034mg/mL.
5.9.
Embodiment 9:LPS inductive lung neutrophilia ferret model
Use clear-headed ferret model to investigate the anti-inflammatory of administered through oral (p.o.) PDE4 suppressor factor when approach is used, emetic and behavior effect.Through these experiments, can measure the therapeutic index (TI) of various PDE4 suppressor factor.TI calculates divided by anti-inflammatory dosage (causing the dosage that 50% of LPS inductive neutrophilia suppresses) through the threshold dose that causes emetic outbreak and behavior change.
Animal rearing
Male ferret (Mustela Pulorius Euro, heavy 1-2kg).Ferret is by Bury Green Farm or Misay Consultancy supply.After transportation, let animal in storing chamber, adapt to one period that is no less than 7 days.Food comprises that the SDS diet C ball shape food and the per week that infinitely give give three times Whiskers
TMThe cat food.Water is the animal level tap water through pasteurization, and change every day.
Give PDE4 suppressor factor
Oral (p.o.) uses the PDE4 suppressor factor, and predose is 1-10g/kg, but for whether definite TI is 10 or higher, rises to 30mg/kg subsequently, and/or to use than low dosage, with the minimum dose of confirming to cause that 50% of neutrophilia suppresses.The ferret overnight fasting, but let it freely drink water.Use is insinuated into the 15cm administration pin the esophagus from the larynx rear portion, to animal orally give vector or PDE4 suppressor factor.After the administration, animal turns back in the storage cages that organic glass door has been installed so that observe, and lets it freely drink water.After the administration, constantly observe animal and write down any vomiting or behavior change.Behind the oral administration 60 to 90 minutes, let animal diet followed.
Be exposed to LPS
Behind orally give compound or the contrast vector 30 minutes, ferret is placed in the organic Glass Containers of sealing, and be exposed to LPS aerosol (100 μ g/ml) 10 minutes.(DeVilbiss USA) generates the LPS aerosol, and imports in the synthetic glass exposure chamber through atomizer.After 10 minutes exposure period, animal returns storage cages, lets it freely drink water, and lets its free diet in later period.Behind the oral administration, observation continues at least 2.5 hours time, and writes down emetic outbreak and behavior change.
Bronchoalveolar lavage
LPS exposes back six hours, through the excessive vetanarcol kill animals of using of intraperitoneal.In tracheae, insert PA tube then, with (twice in phosphate buffered saline buffer (PBS) the lavation lungs of 10 units/ml) of 20ml heparinization.
Blood sampling/tissue displacement
Through through the chest cardiac puncture, remove terminal blood sample this (10ml).Blood is 2, and 500rpm rotation 15 minutes is removed blood plasma and stored at-20 ° of C.Remove brain and freezing equally, supply the compounds content analysis at-20 ° of C.
Cell counting
Bronchoalveolar lavage (BAL) sample is 1, centrifugal 5 minutes of 500rpm.Remove supernatant, the cell mass that obtains is resuspended in 1ml PBS.The cell smear for preparing resuspended liquid, and with the dyeing of Leishmans staining, to allow the cell divide counting.Use remaining resuspended sample to make total cell count.Measure the neutrophilic granulocyte total amount among the BAL thus.
The parameter of measuring
1.LPS the inhibition per-cent of inductive lung neutrophilia.
2. vomiting of emetic Fa Zuo – counting and the amount of retching.
3. the following behavior effect of behavior change-attention: hydrostomia, shortness of breath, oral cavity scratching, calm posture, ataxia, arched back and the walking that falls back.Any behavior change all uses severity grading (slightly, moderate or severe) to come half to quantize.
4. calculate TI, for the maximum dose level that does not cause emetic outbreak found divided by find 50% or suppress the lowest dose level of lung neutrophilia more.
In the lung of clear-headed ferret, compd A is explained in Figure 30 the effect of LPS inductive neutrophilia.
Vomiting and behavior change
Behind the PDE4 oral administration, observe ferret at least two hours, and write down emetic outbreak (vomiting and retch) and behavior change.
In the relevant pretreated ferret of vector (acetone/cremophor/ zero(ppm) water) of oral administration, do not observe emetic outbreak (retching or vomiting).In the animal (7/22) of sub-fraction control treatment, see slight behavior change (lick lip and fall back walking).
Compd A (0.1-3mg/kg, oral) does not cause emetic outbreak (retching and vomiting).Observe some behavior changes (calm posture, lick lip and fall back walking), and classify as slight.At 10mg/g, in 2/6 ferret, observe some retch but not obviously vomiting, follow hydrostomia and behavior change (being assessed as slight or moderate).At the maximum dose level (30mg/kg) of test, in 3/4 animal, observe moderate to significant emesis, follow tangible behavior change.These data are summarized in table 3.
Table 3. ferret that regains consciousness: emetic outbreak and behavior change behind the Orally administered compd A
Observe animal after administration three hours.The animal number that numeral in the parenthesis responds.Animal number range from 4 to 22 in every group.
Therapeutic index is calculated
By these experiments, through using the threshold dose of inducing emetic outbreak divided by the ED that suppresses lung neutrophilia
50Value is every kind of compound determination therapeutic index (TI).TI calculates general introduction in table 4.The TI that compd A has is 12, does not cause emetic outbreak at the anti-inflammatory dosage of 1mg/kg.
Inhibition (the ED of table 4. pair LPS inductive lung neutrophilia
50) and the therapeutic index general introduction of inducing the effective dose of vomiting and being derived from these values
5.10. embodiment 10: the biological activity of compd A in suffering from severe psoriasis in plaques patient
Compd A is the new oral medicament of the generation of downward modulation pro-inflammatory cytokine in people's cell model.Compd A has shown the generation that reduces TNF-α, IL-12 and IFN-γ, and promotes the generation of IL-10.Psoriatic is strong relevant with the dysregulation of cytokine and chemokine, and this permission is carried out potential treatment with immunomodulatory compounds.This 2 phase, open-label, the single armed pilot study is designed the biological activity of coming assessment compd A in severe psoriasis in plaques patient.Carry out the extra assessment of clinical effectiveness, with the potential curative effect of assessment compd A in treatment severe psoriasis in plaques.
Orally administered compd A, every day, 20mg totally 29 days, followed the tracks of the phase with extra 28 days to the observation of patient safety.In baseline, the 15th day with obtained the skin penetrating biopsy specimen (6mm) of target patch on the 29th day.Get harmless skin biopsy at baseline equally.The main pharmacodynamics terminal point is the 29th day epidermal thickness percentage change from baseline.Carry out the epidermis skin thickness by blind examiner and measure and immunohistochemical analysis, with assessment CD11c, CD83, K16, ICAM-1, HLA-DR and fillagrin.Analyze biopsy specimen through RT-PCR, analytic target is: TNF-α, p40-IL12/IL23, IL-10, IFN-γ, IP10, IL-2, IL-8, iNOS, p19-IL23, K16, CD83 and hARP.During 29 days treatment phases of research, carry out PASI, PGA and BSA measurement and study clinical efficacy.Adverse events report, clinical labororatory's assessment, health check-up, ECG and life sign measurement assessment security.Amounting to 19 patients participates in: 15 patients have a complete set of biopsy estimated, and 17 patients have complete curative effect assessment.
This research mesocuticle variation in thickness assessment is main terminal point.19 patients participate in the research, and wherein 15 at baseline with had a complete set of biopsy estimated on the 29th day.In 19 individualities 17 are at baseline and measured the clinical efficacy parameter on the 29th day.At baseline with had in the 29th day among 15 patients that can estimate biopsy, there are eight (53.3%) to show that the epidermis skin thickness has descended 20%.Baseline and the 29th day, have in whole 15 individualities that can estimate biopsy, the average decline of epidermal thickness is 20.5% (p=0.015).Figure 31 has shown the variation that has in the individuality that can estimate biopsy from 29 days epidermal thicknesses of baseline to the.
The crucial struvite mark of assessment comprises epidermis and corium T cell, CD83+ and CD11c cell in biopsy specimen.8 patients' that respond result shows 42.56% and 28.79% (decline of >=20% epidermal thickness) that descended respectively of respondent's mesocuticle and corium T cell.Respondent's mesocuticle and corium CD83+ cell are respectively 32.50% and 25.86% from the average decline of baseline.In respondent's epidermis and corium, the CD11c cell has descended 40.16% and 18.50%.Table 5 has been enumerated the decline of the struvite mark of crucial skin biopsy among respondent and the nonresponder.In addition, a patient who has unusual K16 at baseline had normal K16 at the 29th day.Three patients that have unusual ICAM-1 at baseline had normal ICAM-1 at the 29th day.Two patients with unusual HLA-DR had normal HLA-DR at the 29th day, and three patients that have unusual fillagrin at baseline had normal fillagrin at the 29th day.
The decline per-cent of the 29th day crucial struvite mark of table 5.
| Cell | ? | Epidermis | Corium |
| The T cell | The respondent | -42.56% | -28.79% |
| ? | The nonresponder | +8.74% | -17.34% |
| ?CD83+ | The respondent | -32.50% | -25.86% |
| ? | The nonresponder | -16.31% | +0.46% |
| ?CD11c | The respondent | -40.16% | -18.50% |
| ? | The nonresponder | -2.54% | -21.19% |
Biopsy specimen comprises through the mRNA genetic expression that RT-PCR assesses crucial struvite mark: TNF α, p40-IL12/IL23, IL-10, IFN γ, IP10, IL-2, IL-8, iNOS, p19-IL23, K16 and CD83.After 29 days, the mRNA of iNOS expresses and has reduced by 66.5% (p=0.025) in the infringement skin with the compd A treatment.Reduction and increase that the mRNA of other struvite mark expresses have shown overall improvement tendency.Figure 32 illustrates iNOS change of Expression during the research.
In the individuality of 19 participation, accomplished 29 days treatment phases and had the assessment of complete clinical curative effect for 17 altogether.In the individuality of 19 participation, at the 29th day, have 14 (73.7%) to show that its PASI improves, 3 (15.8%) among these patients have shown that its total psoriatic zone marks from baseline with severity index (PASI)>50% decline.Figure 33 shows that the PASI scoring is from the percentage change of baseline among the 29th day valuable patient.In addition; After compd A treatment 29 days; Among 17 valuable patients, there are 9 (52.9%) to show the improvement of static doctor's total evaluation (sPGA), and have 10 (58.8%) to show that its psoriatic body surface area (BSA) descends from baseline among 17 valuable patients.During treatment and tracking phase, assess security through monitoring adverse events, ECGs, lab investigation, health check-up and vital sign.Report is not dead, has no the patient to withdraw from too early because of adverse events yet.Modally comprise headache (26.3%) and nauseating (15.8%) with the relevant adverse events of treatment.
In this clinical study, once a day oral administration of compound A 20mg totally 29 days in the individuality of suffering from the severe psoriasis in plaques, be safe.Descend along with there being 8 (53.3%) to reach 20% of epidermal thickness at the 29th day in 15 individualities, reach main terminal point.In skin biopsy, notice the decline of crucial struvite mark, comprise T cell, CD83+ and the CD11c cell of corium and epidermis.RT-PCR analyzes demonstration, and iNOS mRNA has significantly descended 66.5% in the 29th day skin biopsy on statistics.After 29 days, notice male clinical efficacy signal with the compd A treatment.At the 29th day, the patient's of participation 73.7% showed the improvement of its psoriatic symptom, and these patients' 15.8% show that its PASI scoring is from baseline>50% decline.At the 29th day, the patient's of participation 47.4% showed the improvement of its sPGA, and 52.6% its psoriatic body surface area (BSA) of demonstration of the patient who participates in descends from baseline.
5.11. embodiment 11: prove the curative effect of compd A in suffering from the psoriatic individuality of moderate to severe and the 2 phases research of security
This 2 phase, polycentric, at random, double blinding, placebo, parallel-group, dosage comparative studies assessed curative effect and the security of compd A in the individuality of suffering from moderate to severe psoriasis in plaques, these individualities are candidates of whole body therapeutic.
This research comprises the treatment phase in 12 week, then is that the phase is followed the tracks of in the observation in 4 weeks.Amount to 260 individualities and accept compd A 20mg secondary every day, compd A 20mg at random once a day or placebo, totally 12 weeks.The main terminal point of this research is, with the individuality of compd A treatment the 12nd the week/treat its psoriatic zone and severity index scoring reach 75% decline (" PASI-75 ") based on baseline visit per-cent for the last time.Last treatment is defined in the last PASI assessment of accomplishing during the treatment phase in 12 week.
The 12nd the week/last treatment, with placebo (10%; P=0.023) relatively, significantly the individuality (24%) with the treatment of 20mg secondary every day of higher proportion has reached PASI-75.To accepting the individuality of 20mg secondary every day or placebo, the 12nd the week/last treatment, the ratio that reaches PASI-50 is respectively 57% and 23%; And the ratio that reaches PASI-90 is respectively 14% and 6%.The 12nd the week/last treatment, the PASI of the individuality of 20mg secondary every day and placebo has reached 52% and 17% average decline respectively from baseline.The individuality of accepting compd A continues to improve in time, descends at the highest average percent that shows the PASI scoring the 12nd week.Generally speaking, in all three treatment groups, the adverse events characteristic is similar.Most adverse events of report are slight.In this research, not report and the relevant serious adverse events of research medicine.In 20mg secondary every day group, during observing the tracking phase, there is not the aggravation of individual experience psoriatic.
In this clinical study, compd A is presented in the individuality of suffering from moderate to severe psoriasis in plaques and is tolerated well, and is safe.After 12 weeks treatment, PASI reaches the clinical activity that 50%, the 75% and 90% individual ratio improved has been explained compd A.
5.12.
Embodiment 12: the solid form screening study
5.12.1.
Experimental technique
solubility studies.Compd A sample (about 100mg) through weighing is handled with about 2mL test solvent.The solvent that uses is SILVER REAGENT or HPLC level.The mixture that obtains stirred 24 hours at about 25 ° of C at least.When through being observed visually all solids when dissolving, calculate the solubleness of estimating.Estimate solubleness through these experiments based on the solvent TV that is used for obtaining solution.Owing to use the speed of a large amount of solvents or stripping slow, so actual solubility maybe be greater than those calculated values.If there is not stripping in experimental session, then solubleness is measured through gravimetry.The filtrating of known volume is evaporated to drying, and measures the weight of resistates.
The research of
solution evaporation.Solubleness to compd A is wherein carried out solution evaporation, for example acetone, acetonitrile, methylene dichloride and THF greater than the solvent of about 50mg/mL.Through about 25 ° of C or about 50 ° of C in nitrogen in the opening bottle slow evaporating solvent, obtain solid sample.
balance studies.Balance test is carried out through in about 2mL test solvent, adding excessive compd A.The mixture that obtains stirred 24 hours at about 25 ° of C or about 50 ° of C at least.When reaching balance, remove saturated solution, and let it respectively in about 25 ° of C or about 50 ° of C slowly evaporation in the opening bottle in nitrogen.The slurry that is obtained by balance is filtered, and at air drying.
The research of
crystallisation by cooling.Carry out crystallisation by cooling research.Solid is dissolved in solvent at the about 65 ° of C of temperature that raise, and let it be cooled to about 25 ° of C.To there be the crystalline sample to be placed in the refrigerator (about 0-5 ° C) at about 25 ° of C.Solid separates through decant, and is allowed to condition at air drying.
The research of
solvent/anti-solvent deposition.Deposition is carried out through the solvent/anti-solvent combination.Solid compd A therein has in the solvent of relative high-dissolvability and dissolves, and the solvent (being anti-solvent) that then compd A of selecting is had relative low solubility therein adds in the solution.In some solvent/anti-solvent systems, deposition forms immediately.If deposition does not occur immediately, the mixture that obtains is cooled off in refrigerator (about 0-5 ° C), up to forming deposition.Deposition is separated through decant then, and is allowed to condition at air drying.
change research.The change experiment is carried out through the slurry of preparation solid form in saturated solvent.Slurry stirred 2 days at about 25 ° of C at least.Through filtering, remove saturated solution, and at the air drying solid.
pressure investigation.Pressure test was carried out through pressing down sample with Carver Mini C press in the power of 2000psi at least one minute.Then through the XRPD analyzing samples.
Study on Hygroscopicity.The water absorbability of various solid forms uses the surface measurement DVS of system instrument to study.Usually the sample size between will about 10-50mg is loaded in the DVS instrument sample disk, and sample is analyzed on DVS robotization adsorption analysis appearance at about 25 ° of C.Relative humidity is increased to about 95%RH by about 0%, interval about 10%.Relative humidity descends in a similar manner then, attaches circulation to accomplish complete adsorption/desorption.Experimental session is with the fixed time interval recording quality.
5.12.2.
Characterizing method
Usually through X-ray powder diffraction (XRPD) analysis as at the sample that generates described in the solid form screening.XRPD is at Thermo ARL X ' TRA
TMImplement on the X-ray powder diffraction meter, use Cu K α radiation,
Apparatus preparation thin burnt X-ray tube.The voltage of x ray generator and amperes are separately positioned on 45kV and 40mA.Disperse sheet and be arranged on 4mm and 2mm, and measured sheet is arranged on 0.5mm and 0.2mm.The radioactive rays of diffraction detect through peltier refrigerative Si (Li) solid-state detector.Usually carry out θ-2 θ continuous sweep with 2.40 °/minute (0.5 second/0.02 ° per steps) from 1.5 ° of 2 θ to 40 ° of 2 θ.Use agglomerating aluminum oxide standard to check the peak position.In general, the measuring basis through about ± 0.2 ° of 2 θ is measured expection XRPD peak position independent variation.In general, understand like this area, if the characteristic peak of the characteristic peak of first figure and second figure is positioned at roughly the same position, these two XRPD scheme to match each other so.Understand like this area; Confirm whether two XRPD figure matees or two XRPD figure in independent peak whether mate, possibly need to consider independent variable and variation, the variation of XRPD data collection parameters and/or the variation of XRPD data processing etc. of parameter such as but not limited to preferred direction, impurity, percent crystallinity, granularity, the setting of diffractometer instrument mutually.Confirm whether two figure mate and can carry out through naked eyes and/or Computer Analysis.Use the instance of the XRPD figure of these methods and parameter collection and analysis to provide in the present invention, for example like Fig. 1, Fig. 5, Fig. 9, Figure 13, Figure 17, Figure 21 and Figure 25.
Dsc (DSC) is analyzed at TA instrument Q 1000
TMLast execution.About 5mg sample is placed in the DSC dish of taring, and accurately writes down sample weight.Usually, sample is heated to the about 200 ° of C of final temperature with about 10 ° of C/ branch speed from about 25 ° of C in nitrogen.Usually, incident heat is registered as the starting temperature of reckoning.Use these methods and parameter collection and the thermographic instance of analysis DSC to provide in the present invention, for example like Fig. 2, Fig. 6, Figure 10, Figure 14, Figure 18, Figure 22 and Figure 26.
Thermogravimetric analysis (TGA) is at TA instrument Q500
TMLast execution.The calibration of use caoxalate.About 10mg sample is placed on the dish, accurately weighs, and in the TGA stove of packing into.Sample is heated to the about 200 ° of C of final temperature with about 10 ° of C/ branch speed from about 25 ° of C in nitrogen.Use these methods and parameter collection and the thermographic instance of analysis TGA to provide in the present invention, for example like Fig. 3, Fig. 7, Figure 11, Figure 15, Figure 19, Figure 23 and Figure 27.
Solvation solvent through the TG-IR experimental identification with quantitatively, use to have connected the spectrophotometric TA instrument of Thermo Nicolet AEM Fourier conversion IR Q500
TMTGA.Usually, the about 20-50mg of sample size is weighed in the aluminium dish, and is heated to about 200 ° of C.The TGA run duration, steam flows to cell through hot delivery conduit.The temperature of delivery conduit and cell all is arranged on about 225 ° of C.The 10 second time of every repetition, collect IR spectrum.Volatile matter is through the storehouse identification of search Aldrich gas-phase spectrum, and the steam that exists storehouse matching result demonstration to be discerned.
The form of sample and sreen analysis use the Olympus microscope to carry out.Instrument is used the USP standard calibration.Use Image Plus-Material Plus software to measure the D90 value.The representative of D90 value is 90 through the percentile of the size-grade distribution of linear measure; Promptly 90% particle has the length of this value or shorter.
5.12.3.
Solid form screening study result
The solid form of the inclusion compound A that during the solid form screening study, prepares comprises form A, B, C, D, E, F, G and amorphous forms.Form A, B, C, D, E, F and G typical XRPD figure, DSC figure, TGA figure and DVS figure separately is provided as Fig. 1-Figure 28 in the present invention.
solubility studies.The form B that measures compd A is in the approximate solubility of about 25 ° of C in all kinds of SOLVENTS.The result shows in table 6.Discovery form B the most solvable in acetone, acetonitrile, methylene dichloride, methyl ethyl ketone and THF (greater than about 50mg/mL) then is in ETHYLE ACETATE (about 30.15mg/mL).Find that also form B has low solubility in several solvents, comprise propyl carbinol, heptane, 2-propyl alcohol, toluene and water (being lower than about 1mg/mL).
Solution evaporation researchThe result of the solution evaporation research of carrying out at about 25 ° of C and about 50 ° of C summarizes in table 7.
Balance studiesThe result of the balance studies that carries out at about 25 ° of C and about 50 ° of C summarizes in table 8.
The research of
crystallisation by cooling.The result of crystallisation by cooling research summarizes in table 9.Crystallisation by cooling research output crystalline material from many solvents comprises acetone, acetonitrile, n-butyl acetate, ETHYLE ACETATE, methyl alcohol, methylene dichloride, methyl ethyl ketone (MEK) and THF (THF).The crystalline material that obtains characterizes through XRPD, DSC and TGA usually.
The research of
solvent/anti-solvent deposition.The result of solvent/anti-solvent deposition research summarizes in table 10.When with heptane, water and toluene when about 40 ° of C add in the solution of form B in THF, form deposition immediately.When with heptane, methyl tert-butyl ether (MTBE), toluene and water respectively when about 25 ° of C add in the solution of form B in acetonitrile, form settled solution or mixture.After the stirred overnight, obtain crystalline material from MTBE/ acetonitrile, water/acetonitrile and toluene/acetonitrile.But, there is not crystallization to occur to heptane/acetonitrile mixture.When with water when about 50 ° of C add in the solution of form B in methyl alcohol, form deposition immediately, and when with heptane and toluene respectively when about 50 ° of C add in the solution of form B in methyl alcohol, formation settled solution or mixture.After the stirred overnight, from toluene and methanol and heptane/methyl alcohol, obtain crystalline material.When with toluene when about 25 ° of C add in the solution of form B in methylene dichloride, form deposition immediately, and when with MTBE when about 25 ° of C add in the solution of form B in methylene dichloride, the acquisition settled solution.After the stirred overnight, from the MTBE/ methylene dichloride, obtain crystalline material.But, in the time of in heptane being added to the solution of form B in methylene dichloride, do not have crystallization to occur.When with heptane when about 50 ° of C add in the solution of form B in MEK, form deposition immediately, and when with MTBE and toluene respectively when about 50 ° of C add in the solution of form B in MEK, the acquisition settled solution.After the stirred overnight, from MTBE/MEK and toluene/MEK, obtain crystalline material.When with heptane when about 50 ° of C add in the solution of form B in n-butyl acetate, form deposition immediately, and when with MTBE and toluene respectively when about 50 ° of C add in the solution of form B in MEK, the acquisition settled solution.After the stirred overnight, from MTBE/ n-butyl acetate and toluene/n-butyl acetate, obtain crystalline material.When with water and toluene respectively when about 40 ° of C add in the solution of form B in acetone, form deposition immediately, and when with ethanol and 2-propyl alcohol respectively when about 40 ° of C add in the solution of form B in acetone, the acquisition settled solution.After the stirred overnight, from ethanol/acetone and 2-propyl alcohol/acetone, obtain crystalline material.The crystalline material that obtains is identified through XRPD, DSC, TGA.
stability study.The result of stability study summarizes in table 11.Through solid sample being exposed to the four stars phase under 40 ° of C/75%RH stressed conditions, the stability of research form A, B, C and D.In addition, form A, B, C and the D stability in different solvents is through studying in 40 ° of C balance four stars phase in different solvents.Cross filter pulp then, and at air drying.Solid sample by stability experiment obtains is passed through XRPD and dsc analysis.
Change researchThe result of change research summarizes in table 12.
Pressure investigationForm A, B, C, D, E, F and G to compd A carry out pressure test.Like what arrive, find that each form be studied all is basic physically stable through the XRPD analysis and observation.
Study on Hygroscopicity.Form A, B, C, D, E, F and G are carried out water absorbability (water adsorption/desorption) research.After the complete adsorption/desorption of experience attached circulation in the DVS system, each solid sample was analyzed through XRPD.XRPD result points out that as the result that DVS analyzes, none lives through substantial solid-state conversion in the form of analysis.
The solubility studies of table 6. form B
| Solvent system | Approximate solubility (mg/ml) |
| Acetone | >;50 |
| Acetonitrile | >;50 |
| Propyl carbinol | >;0.72 |
| N-butyl acetate | 9.75 |
| Absolute ethyl alcohol | 1.38 |
| ETHYLE ACETATE | 30.15 |
| Heptane | 0.41 |
| Methylene dichloride | >;50 |
| Methyl ethyl ketone | >;50 |
| Methyl alcohol | 4.05 |
| Methyl tert-butyl ether | 1.17 |
| The 2-propyl alcohol | 0.81 |
| THF | >;50 |
| Toluene | 0.90 |
| Water | 0.69 |
| Ethanol: water (1:1) | 2.86 |
The research of table 7. solution evaporation
Table 8. balance studies
The research of table 9. crystallisation by cooling
| Initial form | Solvent system | XRPD analyzes | The DSC incident heat |
| B | Acetone | Form E | ? |
| B | Acetonitrile | Form E | 95.42°C |
| B | N-butyl acetate | Form B | ? |
| B | ETHYLE ACETATE | Form B | ? |
| B | Methylene dichloride | Form D | 100.90°C |
| B | Methyl alcohol | Form B | ? |
| B | Methyl ethyl ketone | Form B | ? |
| B | THF | Form H | ? |
The research of table 10. solvent/anti-solvent deposition
*Abbreviation: MEK=methyl ethyl ketone; DCM=methylene dichloride (being methylene chloride); The MtBE=methyl tert-butyl ether
Table 11. stability study
Table 12. change research
5.13.
Embodiment 13:200MG dose capsule
Table 13 has been explained the batch prescription and the single dose prescription of the single dose unit of the solid form (promptly about 40% weight ratio) that in the 0# capsule, comprises 200mg inclusion compound A.
The capsular prescription of table 13.200mg
Make pregelatinized W-Gum (SPRESS
TMB-820) and the compd A composition through 710 μ m sieve, in the diffusion mixer that the band baffle plate of packing into then inserts, mixed 15 minutes.Make Magnesium Stearate pass through 210 μ m sieve, add in the diffusion mixer.Use Dosator type capsule filler mixture to be packed in the 0# capsule into 500mg/ capsule (production lot 8400 capsules) then.
5.14.
Embodiment 14:100MG oral dosage form
Table 14 has been explained batch prescription and single dose unit's prescription of the solid form that comprises 100mg inclusion compound A.
The prescription of table 14.100mg tablet
Make Microcrystalline Cellulose, cross-linked carboxymethyl cellulose sodium and compd A composition through #30 mesh sieve (about 430 μ are to about 655 μ).Make Pluronic
(by JRH Biosciences; Lenexa company, KS preparation) tensio-active agent is through #20 mesh sieve (about 457 μ are to about 1041 μ).Rolled in the stirrer in Pluronic
tensio-active agent and 0.5kg cross-linked carboxymethyl cellulose sodium 16 quarts of bivalves of packing into, mixed about 5 minutes.Then mixture is transferred to 3 cubic feet of bivalves and roll in the stirrer, add Microcrystalline Cellulose, mixed about 5 minutes.Add the solid form of inclusion compound A, mixed other 25 minutes.This pre-composition has the roller compaction machine of beater grinder through discharging place, and retracts in the stirrer that rolls.Remaining cross-linked carboxymethyl cellulose sodium and Magnesium Stearate join in the stirrer that rolls, and mix about 3 minutes.Final mixture is compressed to 250mg/ sheet (200,000 of production lots) on Rotarytabletpress.
Although the present invention is described specific embodiments, those skilled in the art will be obviously can not deviate from the claim defined purport of the present invention and scope and carry out various changes and modification.Such modification also drops in the appended claim scope.
Claims (11)
2. solid form as claimed in claim 1, its dsc figure comprises the heat absorption incident of the about 154 ° of C of starting temperature.
3. solid form as claimed in claim 1, when being heated to about 140 ° of C from about 25 ° of C, its thermogravimetic analysis (TGA) figure comprises and is lower than about 1% mass loss.
4. solid form as claimed in claim 1, when being heated to about 140 ° of C from about 25 ° of C, its thermogravimetic analysis (TGA) figure comprises and is lower than about 0.25% mass loss.
5. solid form as claimed in claim 1, when experience when about 0% relative humidity is increased to about 95% relative humidity, its demonstration is lower than about 1% quality to be increased.
6. solid form as claimed in claim 1, when experience when about 0% relative humidity is increased to about 95% relative humidity, its demonstration is lower than about 0.6% quality to be increased.
7. solid form as claimed in claim 4, when being exposed to about 4 weeks of about 40 ° of C and about 75% relative humidity, it is stable.
8. the pharmaceutical composition that comprises each described solid form among the claim 1-7.
Among the claim 1-7 each described solid form be used for treating in preparation can be through suppressing the purposes that TNF-α produces the medicine of the disease improved or illness, wherein said disease or illness are selected from psoriatic, psoriatic arthritis, rheumatoid arthritis, chronic skin sarcoid, giant cell arteritis, Parkinson's disease, prurigo nodularis, lichen planus, complicacy aphthosis, Behcet's disease, lupus, hepatitis, uveitis, sjogren syndrome, depression, interstitial cystitis, vulvodynia, prostatitis, osteo-arthritis, diffuse large B cell lymphoma, polymyositis, dermatomyositis, inclusion body myositis, EOA, interstitial cystitis, hepatitis, endometriosis, radiculopathy and pyoderma gangraenosum.
10. each described solid form is used for treating the purposes of medicine that can be through suppressing disease that PDE4 improves or illness in preparation among the claim 1-7, and wherein said disease or illness are selected from: HIV; Hepatitis; Adult respiratory distress syndrome; Bone resorption disease; Chronic obstructive pulmonary disease; Chronic pulmonary inflammation disease; Dermatitis; Inflammatory skin diseases, atopic dermatitis, cystic fibrosis; Septic shock; Septicemia; Endotoxin shock; The haemodynamics shock; Septicemia syndrome; Postischemic reperfusion damage; Meningitis; Psoriatic; Fibrotic disease; Emaciation; Transplant rejection; Graft versus host disease; Autoimmune disease; Rheumatoid spondylitis; Arhritis conditions; Rheumatoid arthritis; Osteo-arthritis; Osteoporosis; Segmental enteritis; Ulcerative colitis; Inflammatory bowel; Multiple sclerosis; Systemic lupus erythematosus; ENL in the leprosy; Radiation injury; Asthma; And hyperoxic alveolar injury.
11. each described solid form is used for treating the purposes of the medicine of cancer in preparation among the claim 1-7, wherein said cancer is selected from multiple myeloma, malignant melanoma, glioblastoma, white blood disease and solid tumor.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2012101389499A CN102702070A (en) | 2008-03-27 | 2008-03-27 | Solid form containing (+)-2-[1-(3-oxethyl-4-methoxyphenyl)-2-methylsulfonylethyl]-4-acetylaminoisoindoline-1,3-dione, composition and application thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2012101389499A CN102702070A (en) | 2008-03-27 | 2008-03-27 | Solid form containing (+)-2-[1-(3-oxethyl-4-methoxyphenyl)-2-methylsulfonylethyl]-4-acetylaminoisoindoline-1,3-dione, composition and application thereof |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN2008801294626A Division CN102046167A (en) | 2008-03-27 | 2008-03-27 | Contains (+)-2-[1-(3-ethoxy-4-methoxyphenyl)-2-methylsulfonylethyl]-4-acetylaminoisoindoline-1,3- Solid forms of diketones, compositions thereof and uses thereof |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN102702070A true CN102702070A (en) | 2012-10-03 |
Family
ID=46895183
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN2012101389499A Pending CN102702070A (en) | 2008-03-27 | 2008-03-27 | Solid form containing (+)-2-[1-(3-oxethyl-4-methoxyphenyl)-2-methylsulfonylethyl]-4-acetylaminoisoindoline-1,3-dione, composition and application thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN102702070A (en) |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN105294534A (en) * | 2014-07-15 | 2016-02-03 | 上海优拓医药科技有限公司 | Industrial method for preparing apremilast and intermediate thereof |
| WO2017084597A1 (en) | 2015-11-19 | 2017-05-26 | 常州爱诺新睿医药技术有限公司 | Amorphous form of apremilast, preparation method therefor, and application thereof |
| CN111821297A (en) * | 2019-04-16 | 2020-10-27 | 天津合美医药科技有限公司 | Use of isoindoline derivatives for the treatment of immunoglobulin E (IgE) -mediated diseases |
| CN112245403A (en) * | 2020-10-23 | 2021-01-22 | 杭州朱养心药业有限公司 | Phosphodiesterase-4 inhibitor and oral solid composition thereof |
| WO2023105286A1 (en) * | 2021-12-06 | 2023-06-15 | My Personal Therapeutics Ltd | A combination treatment for cancer |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1652772A (en) * | 2002-03-20 | 2005-08-10 | 细胞基因公司 | (+)-2-[1-(3-ethoxy-4-methoxyphenyl)-2-methylsulfonylethyl]-4-acetylaminoisoindoline-1,3-dione: Methods of use and compositions thereof |
-
2008
- 2008-03-27 CN CN2012101389499A patent/CN102702070A/en active Pending
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1652772A (en) * | 2002-03-20 | 2005-08-10 | 细胞基因公司 | (+)-2-[1-(3-ethoxy-4-methoxyphenyl)-2-methylsulfonylethyl]-4-acetylaminoisoindoline-1,3-dione: Methods of use and compositions thereof |
| US20080027123A1 (en) * | 2002-03-20 | 2008-01-31 | Celgene Corporation | (+)-2-[1-(3-Ethoxy-4methoxyphenyl)-2-methylsulfonylethyl]-4-acetylaminoisoindoline-1,3,-dione:methods of using and compositions thereof |
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN105294534A (en) * | 2014-07-15 | 2016-02-03 | 上海优拓医药科技有限公司 | Industrial method for preparing apremilast and intermediate thereof |
| CN105294534B (en) * | 2014-07-15 | 2020-04-10 | 上海优拓医药科技有限公司 | Industrialized method for preparing aplidine and intermediate thereof |
| WO2017084597A1 (en) | 2015-11-19 | 2017-05-26 | 常州爱诺新睿医药技术有限公司 | Amorphous form of apremilast, preparation method therefor, and application thereof |
| CN111821297A (en) * | 2019-04-16 | 2020-10-27 | 天津合美医药科技有限公司 | Use of isoindoline derivatives for the treatment of immunoglobulin E (IgE) -mediated diseases |
| CN112245403A (en) * | 2020-10-23 | 2021-01-22 | 杭州朱养心药业有限公司 | Phosphodiesterase-4 inhibitor and oral solid composition thereof |
| CN112245403B (en) * | 2020-10-23 | 2022-04-22 | 杭州朱养心药业有限公司 | Phosphodiesterase-4 inhibitor and oral solid composition thereof |
| WO2023105286A1 (en) * | 2021-12-06 | 2023-06-15 | My Personal Therapeutics Ltd | A combination treatment for cancer |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US11040024B2 (en) | Methods of using (+)-2-[1-(3-ethoxy-4-methoxyphenyl)-2-methylsulfonylethyl]-4-acetylaminoisoindoline-1,3-dione | |
| US9433606B2 (en) | Solid forms comprising (+)-2-[1-(3-ethoxy-4-methoxyphenyl)-2-methylsulfonylethyl]-4-acetylaminoisoindoline-1,3-dione, compositions thereof, and uses thereof | |
| AU2008353468B2 (en) | Solid forms comprising (+)-2- [1-(3-Ethoxy-4-methoxyphenyl)-2-methylsulfonylethyl] -4-acetylaminoisoindoline-1,3-dione, compositions thereof, and uses thereof | |
| CN102702070A (en) | Solid form containing (+)-2-[1-(3-oxethyl-4-methoxyphenyl)-2-methylsulfonylethyl]-4-acetylaminoisoindoline-1,3-dione, composition and application thereof | |
| CN102898349A (en) | Solid containing compound, a composition containing the solid and use of the solid | |
| AU2014265073A1 (en) | Solid forms comprising (+)-2-[1-(3-ethoxy-4-methoxyphenyl)-2-methylsulfonylethyl]-4-acetylaminoisoindoline-1,3-dione, compositions thereof, and uses thereof | |
| AU2007231843A1 (en) | Tooth Whitening Products |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| RJ01 | Rejection of invention patent application after publication |
Application publication date: 20121003 |
|
| RJ01 | Rejection of invention patent application after publication |