CN102657626A - Medicinal composite tablet of pioglitazone medicine - Google Patents
Medicinal composite tablet of pioglitazone medicine Download PDFInfo
- Publication number
- CN102657626A CN102657626A CN2012101605784A CN201210160578A CN102657626A CN 102657626 A CN102657626 A CN 102657626A CN 2012101605784 A CN2012101605784 A CN 2012101605784A CN 201210160578 A CN201210160578 A CN 201210160578A CN 102657626 A CN102657626 A CN 102657626A
- Authority
- CN
- China
- Prior art keywords
- pioglitazone hydrochloride
- medicine
- pioglitazone
- emulsifying
- emulsifying agent
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 239000003814 drug Substances 0.000 title claims abstract description 65
- HYAFETHFCAUJAY-UHFFFAOYSA-N pioglitazone Chemical compound N1=CC(CC)=CC=C1CCOC(C=C1)=CC=C1CC1C(=O)NC(=O)S1 HYAFETHFCAUJAY-UHFFFAOYSA-N 0.000 title abstract description 18
- 229960005095 pioglitazone Drugs 0.000 title abstract description 9
- 239000002131 composite material Substances 0.000 title abstract 3
- 239000003995 emulsifying agent Substances 0.000 claims abstract description 28
- WTJKGGKOPKCXLL-RRHRGVEJSA-N phosphatidylcholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCCCCCCC=CCCCCCCCC WTJKGGKOPKCXLL-RRHRGVEJSA-N 0.000 claims abstract description 26
- GHUUBYQTCDQWRA-UHFFFAOYSA-N Pioglitazone hydrochloride Chemical compound Cl.N1=CC(CC)=CC=C1CCOC(C=C1)=CC=C1CC1C(=O)NC(=O)S1 GHUUBYQTCDQWRA-UHFFFAOYSA-N 0.000 claims description 82
- 229960002827 pioglitazone hydrochloride Drugs 0.000 claims description 82
- 229940079593 drug Drugs 0.000 claims description 27
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims description 19
- 238000012377 drug delivery Methods 0.000 claims description 18
- 239000000203 mixture Substances 0.000 claims description 13
- 235000010482 polyoxyethylene sorbitan monooleate Nutrition 0.000 claims description 12
- 229920000053 polysorbate 80 Polymers 0.000 claims description 12
- 239000003921 oil Substances 0.000 claims description 11
- 235000019198 oils Nutrition 0.000 claims description 11
- -1 polyoxyethylene Polymers 0.000 claims description 10
- 239000007787 solid Substances 0.000 claims description 8
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 claims description 6
- 239000007788 liquid Substances 0.000 claims description 6
- DCXXMTOCNZCJGO-UHFFFAOYSA-N tristearoylglycerol Chemical compound CCCCCCCCCCCCCCCCCC(=O)OCC(OC(=O)CCCCCCCCCCCCCCCCC)COC(=O)CCCCCCCCCCCCCCCCC DCXXMTOCNZCJGO-UHFFFAOYSA-N 0.000 claims description 6
- ZEMPKEQAKRGZGQ-AAKVHIHISA-N 2,3-bis[[(z)-12-hydroxyoctadec-9-enoyl]oxy]propyl (z)-12-hydroxyoctadec-9-enoate Chemical compound CCCCCCC(O)C\C=C/CCCCCCCC(=O)OCC(OC(=O)CCCCCCC\C=C/CC(O)CCCCCC)COC(=O)CCCCCCC\C=C/CC(O)CCCCCC ZEMPKEQAKRGZGQ-AAKVHIHISA-N 0.000 claims description 5
- 239000011347 resin Substances 0.000 claims description 5
- 229920005989 resin Polymers 0.000 claims description 5
- 229920003171 Poly (ethylene oxide) Polymers 0.000 claims description 4
- GHVNFZFCNZKVNT-UHFFFAOYSA-N decanoic acid Chemical compound CCCCCCCCCC(O)=O GHVNFZFCNZKVNT-UHFFFAOYSA-N 0.000 claims description 4
- ZQPPMHVWECSIRJ-KTKRTIGZSA-N oleic acid Chemical compound CCCCCCCC\C=C/CCCCCCCC(O)=O ZQPPMHVWECSIRJ-KTKRTIGZSA-N 0.000 claims description 4
- 229920000168 Microcrystalline cellulose Polymers 0.000 claims description 3
- 239000008108 microcrystalline cellulose Substances 0.000 claims description 3
- 235000019813 microcrystalline cellulose Nutrition 0.000 claims description 3
- 229940016286 microcrystalline cellulose Drugs 0.000 claims description 3
- OYHQOLUKZRVURQ-NTGFUMLPSA-N (9Z,12Z)-9,10,12,13-tetratritiooctadeca-9,12-dienoic acid Chemical compound C(CCCCCCC\C(=C(/C\C(=C(/CCCCC)\[3H])\[3H])\[3H])\[3H])(=O)O OYHQOLUKZRVURQ-NTGFUMLPSA-N 0.000 claims description 2
- WRIDQFICGBMAFQ-UHFFFAOYSA-N (E)-8-Octadecenoic acid Natural products CCCCCCCCCC=CCCCCCCC(O)=O WRIDQFICGBMAFQ-UHFFFAOYSA-N 0.000 claims description 2
- LQJBNNIYVWPHFW-UHFFFAOYSA-N 20:1omega9c fatty acid Natural products CCCCCCCCCCC=CCCCCCCCC(O)=O LQJBNNIYVWPHFW-UHFFFAOYSA-N 0.000 claims description 2
- HIQIXEFWDLTDED-UHFFFAOYSA-N 4-hydroxy-1-piperidin-4-ylpyrrolidin-2-one Chemical compound O=C1CC(O)CN1C1CCNCC1 HIQIXEFWDLTDED-UHFFFAOYSA-N 0.000 claims description 2
- QSBYPNXLFMSGKH-UHFFFAOYSA-N 9-Heptadecensaeure Natural products CCCCCCCC=CCCCCCCCC(O)=O QSBYPNXLFMSGKH-UHFFFAOYSA-N 0.000 claims description 2
- 239000004925 Acrylic resin Substances 0.000 claims description 2
- 229910002012 Aerosil® Inorganic materials 0.000 claims description 2
- 239000005632 Capric acid (CAS 334-48-5) Substances 0.000 claims description 2
- 229920000623 Cellulose acetate phthalate Polymers 0.000 claims description 2
- 239000001856 Ethyl cellulose Substances 0.000 claims description 2
- ZZSNKZQZMQGXPY-UHFFFAOYSA-N Ethyl cellulose Chemical compound CCOCC1OC(OC)C(OCC)C(OCC)C1OC1C(O)C(O)C(OC)C(CO)O1 ZZSNKZQZMQGXPY-UHFFFAOYSA-N 0.000 claims description 2
- 108010010803 Gelatin Proteins 0.000 claims description 2
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 claims description 2
- 239000005642 Oleic acid Substances 0.000 claims description 2
- ZQPPMHVWECSIRJ-UHFFFAOYSA-N Oleic acid Natural products CCCCCCCCC=CCCCCCCCC(O)=O ZQPPMHVWECSIRJ-UHFFFAOYSA-N 0.000 claims description 2
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 claims description 2
- DTOSIQBPPRVQHS-PDBXOOCHSA-N alpha-linolenic acid Chemical compound CC\C=C/C\C=C/C\C=C/CCCCCCCC(O)=O DTOSIQBPPRVQHS-PDBXOOCHSA-N 0.000 claims description 2
- 235000020661 alpha-linolenic acid Nutrition 0.000 claims description 2
- CDQSJQSWAWPGKG-UHFFFAOYSA-N butane-1,1-diol Chemical compound CCCC(O)O CDQSJQSWAWPGKG-UHFFFAOYSA-N 0.000 claims description 2
- 229920002301 cellulose acetate Polymers 0.000 claims description 2
- 229940081734 cellulose acetate phthalate Drugs 0.000 claims description 2
- 150000002148 esters Chemical class 0.000 claims description 2
- 235000019325 ethyl cellulose Nutrition 0.000 claims description 2
- 229920001249 ethyl cellulose Polymers 0.000 claims description 2
- FMMOOAYVCKXGMF-MURFETPASA-N ethyl linoleate Chemical compound CCCCC\C=C/C\C=C/CCCCCCCC(=O)OCC FMMOOAYVCKXGMF-MURFETPASA-N 0.000 claims description 2
- 229940031016 ethyl linoleate Drugs 0.000 claims description 2
- 239000008273 gelatin Substances 0.000 claims description 2
- 229920000159 gelatin Polymers 0.000 claims description 2
- 235000019322 gelatine Nutrition 0.000 claims description 2
- 235000011852 gelatine desserts Nutrition 0.000 claims description 2
- 125000005456 glyceride group Chemical group 0.000 claims description 2
- 229920003132 hydroxypropyl methylcellulose phthalate Polymers 0.000 claims description 2
- 229940031704 hydroxypropyl methylcellulose phthalate Drugs 0.000 claims description 2
- QXJSBBXBKPUZAA-UHFFFAOYSA-N isooleic acid Natural products CCCCCCCC=CCCCCCCCCC(O)=O QXJSBBXBKPUZAA-UHFFFAOYSA-N 0.000 claims description 2
- 229940074928 isopropyl myristate Drugs 0.000 claims description 2
- 239000008101 lactose Substances 0.000 claims description 2
- FMMOOAYVCKXGMF-UHFFFAOYSA-N linoleic acid ethyl ester Natural products CCCCCC=CCC=CCCCCCCCC(=O)OCC FMMOOAYVCKXGMF-UHFFFAOYSA-N 0.000 claims description 2
- 229960004488 linolenic acid Drugs 0.000 claims description 2
- KQQKGWQCNNTQJW-UHFFFAOYSA-N linolenic acid Natural products CC=CCCC=CCC=CCCCCCCCC(O)=O KQQKGWQCNNTQJW-UHFFFAOYSA-N 0.000 claims description 2
- 150000004667 medium chain fatty acids Chemical class 0.000 claims description 2
- WIBFFTLQMKKBLZ-SEYXRHQNSA-N n-butyl oleate Chemical compound CCCCCCCC\C=C/CCCCCCCC(=O)OCCCC WIBFFTLQMKKBLZ-SEYXRHQNSA-N 0.000 claims description 2
- 229940049964 oleate Drugs 0.000 claims description 2
- 239000004006 olive oil Substances 0.000 claims description 2
- 235000008390 olive oil Nutrition 0.000 claims description 2
- 210000000582 semen Anatomy 0.000 claims description 2
- 229920002379 silicone rubber Polymers 0.000 claims description 2
- 239000004945 silicone rubber Substances 0.000 claims description 2
- 239000003549 soybean oil Substances 0.000 claims description 2
- 235000012424 soybean oil Nutrition 0.000 claims description 2
- 229940014259 gelatin Drugs 0.000 claims 1
- 210000003734 kidney Anatomy 0.000 abstract description 33
- 210000004185 liver Anatomy 0.000 abstract description 32
- 230000000694 effects Effects 0.000 abstract description 20
- 238000002360 preparation method Methods 0.000 abstract description 14
- 238000011282 treatment Methods 0.000 abstract description 11
- 206010012601 diabetes mellitus Diseases 0.000 abstract description 9
- 206010020772 Hypertension Diseases 0.000 abstract description 7
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 abstract description 7
- 201000010099 disease Diseases 0.000 abstract description 6
- 238000004090 dissolution Methods 0.000 abstract description 5
- 208000007530 Essential hypertension Diseases 0.000 abstract description 4
- 238000010521 absorption reaction Methods 0.000 abstract description 4
- 206010005003 Bladder cancer Diseases 0.000 abstract description 2
- 208000002249 Diabetes Complications Diseases 0.000 abstract description 2
- 206010012655 Diabetic complications Diseases 0.000 abstract description 2
- 208000007097 Urinary Bladder Neoplasms Diseases 0.000 abstract description 2
- 201000005112 urinary bladder cancer Diseases 0.000 abstract description 2
- 230000006735 deficit Effects 0.000 abstract 1
- 230000036267 drug metabolism Effects 0.000 abstract 1
- 230000014759 maintenance of location Effects 0.000 abstract 1
- NOESYZHRGYRDHS-UHFFFAOYSA-N insulin Chemical compound N1C(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(NC(=O)CN)C(C)CC)CSSCC(C(NC(CO)C(=O)NC(CC(C)C)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CCC(N)=O)C(=O)NC(CC(C)C)C(=O)NC(CCC(O)=O)C(=O)NC(CC(N)=O)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CSSCC(NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2C=CC(O)=CC=2)NC(=O)C(CC(C)C)NC(=O)C(C)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2NC=NC=2)NC(=O)C(CO)NC(=O)CNC2=O)C(=O)NCC(=O)NC(CCC(O)=O)C(=O)NC(CCCNC(N)=N)C(=O)NCC(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC(O)=CC=3)C(=O)NC(C(C)O)C(=O)N3C(CCC3)C(=O)NC(CCCCN)C(=O)NC(C)C(O)=O)C(=O)NC(CC(N)=O)C(O)=O)=O)NC(=O)C(C(C)CC)NC(=O)C(CO)NC(=O)C(C(C)O)NC(=O)C1CSSCC2NC(=O)C(CC(C)C)NC(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CC(N)=O)NC(=O)C(NC(=O)C(N)CC=1C=CC=CC=1)C(C)C)CC1=CN=CN1 NOESYZHRGYRDHS-UHFFFAOYSA-N 0.000 description 18
- 239000008280 blood Substances 0.000 description 12
- 210000004369 blood Anatomy 0.000 description 12
- 210000001519 tissue Anatomy 0.000 description 12
- 238000000034 method Methods 0.000 description 10
- 238000011160 research Methods 0.000 description 10
- 239000000243 solution Substances 0.000 description 10
- 102000004877 Insulin Human genes 0.000 description 9
- 108090001061 Insulin Proteins 0.000 description 9
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 9
- 238000009826 distribution Methods 0.000 description 9
- 230000002496 gastric effect Effects 0.000 description 9
- 229940125396 insulin Drugs 0.000 description 9
- 238000010253 intravenous injection Methods 0.000 description 9
- 239000000463 material Substances 0.000 description 8
- 208000001072 type 2 diabetes mellitus Diseases 0.000 description 7
- 238000005303 weighing Methods 0.000 description 7
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 6
- 229920002125 Sokalan® Polymers 0.000 description 6
- HVYWMOMLDIMFJA-DPAQBDIFSA-N cholesterol Chemical compound C1C=C2C[C@@H](O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@H](C)CCCC(C)C)[C@@]1(C)CC2 HVYWMOMLDIMFJA-DPAQBDIFSA-N 0.000 description 6
- 239000000839 emulsion Substances 0.000 description 6
- 238000001802 infusion Methods 0.000 description 6
- 239000002245 particle Substances 0.000 description 6
- 241000699666 Mus <mouse, genus> Species 0.000 description 5
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 5
- 229960004756 ethanol Drugs 0.000 description 5
- 235000019197 fats Nutrition 0.000 description 5
- 210000002784 stomach Anatomy 0.000 description 5
- 208000007342 Diabetic Nephropathies Diseases 0.000 description 4
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 4
- 239000000337 buffer salt Substances 0.000 description 4
- 208000033679 diabetic kidney disease Diseases 0.000 description 4
- 238000010790 dilution Methods 0.000 description 4
- 239000012895 dilution Substances 0.000 description 4
- 230000001804 emulsifying effect Effects 0.000 description 4
- 239000008103 glucose Substances 0.000 description 4
- 230000003907 kidney function Effects 0.000 description 4
- 239000012086 standard solution Substances 0.000 description 4
- 239000011550 stock solution Substances 0.000 description 4
- 230000008685 targeting Effects 0.000 description 4
- 238000012360 testing method Methods 0.000 description 4
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 3
- 230000006378 damage Effects 0.000 description 3
- 238000002474 experimental method Methods 0.000 description 3
- 239000012530 fluid Substances 0.000 description 3
- 238000003304 gavage Methods 0.000 description 3
- 201000001421 hyperglycemia Diseases 0.000 description 3
- 238000002347 injection Methods 0.000 description 3
- 239000007924 injection Substances 0.000 description 3
- 235000013336 milk Nutrition 0.000 description 3
- 239000008267 milk Substances 0.000 description 3
- 210000004080 milk Anatomy 0.000 description 3
- 230000009467 reduction Effects 0.000 description 3
- 230000035945 sensitivity Effects 0.000 description 3
- 239000002689 soil Substances 0.000 description 3
- 230000001225 therapeutic effect Effects 0.000 description 3
- 230000001988 toxicity Effects 0.000 description 3
- 231100000419 toxicity Toxicity 0.000 description 3
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 3
- 102000009027 Albumins Human genes 0.000 description 2
- 108010088751 Albumins Proteins 0.000 description 2
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 2
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 2
- 206010013786 Dry skin Diseases 0.000 description 2
- 208000022461 Glomerular disease Diseases 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- 208000017170 Lipid metabolism disease Diseases 0.000 description 2
- 241000700159 Rattus Species 0.000 description 2
- 208000027418 Wounds and injury Diseases 0.000 description 2
- 239000012190 activator Substances 0.000 description 2
- 239000002671 adjuvant Substances 0.000 description 2
- 229940127003 anti-diabetic drug Drugs 0.000 description 2
- 239000003472 antidiabetic agent Substances 0.000 description 2
- 230000036772 blood pressure Effects 0.000 description 2
- 210000004204 blood vessel Anatomy 0.000 description 2
- 230000008859 change Effects 0.000 description 2
- 238000006243 chemical reaction Methods 0.000 description 2
- 235000012000 cholesterol Nutrition 0.000 description 2
- 208000019425 cirrhosis of liver Diseases 0.000 description 2
- 230000001276 controlling effect Effects 0.000 description 2
- 229960000935 dehydrated alcohol Drugs 0.000 description 2
- 238000003745 diagnosis Methods 0.000 description 2
- 238000007599 discharging Methods 0.000 description 2
- 230000000857 drug effect Effects 0.000 description 2
- 238000001035 drying Methods 0.000 description 2
- 238000009472 formulation Methods 0.000 description 2
- 230000006870 function Effects 0.000 description 2
- 210000004211 gastric acid Anatomy 0.000 description 2
- 239000000499 gel Substances 0.000 description 2
- 230000002218 hypoglycaemic effect Effects 0.000 description 2
- 230000005764 inhibitory process Effects 0.000 description 2
- 208000014674 injury Diseases 0.000 description 2
- 208000017169 kidney disease Diseases 0.000 description 2
- 150000002632 lipids Chemical class 0.000 description 2
- 230000007774 longterm Effects 0.000 description 2
- 208000010125 myocardial infarction Diseases 0.000 description 2
- 150000002978 peroxides Chemical class 0.000 description 2
- 230000008569 process Effects 0.000 description 2
- 230000001681 protective effect Effects 0.000 description 2
- 238000011084 recovery Methods 0.000 description 2
- 230000008439 repair process Effects 0.000 description 2
- 210000002966 serum Anatomy 0.000 description 2
- 239000007779 soft material Substances 0.000 description 2
- 229910001220 stainless steel Inorganic materials 0.000 description 2
- 239000010935 stainless steel Substances 0.000 description 2
- 239000000758 substrate Substances 0.000 description 2
- 239000006228 supernatant Substances 0.000 description 2
- 238000010998 test method Methods 0.000 description 2
- ZOBPZXTWZATXDG-UHFFFAOYSA-N 1,3-thiazolidine-2,4-dione Chemical compound O=C1CSC(=O)N1 ZOBPZXTWZATXDG-UHFFFAOYSA-N 0.000 description 1
- 102000005862 Angiotensin II Human genes 0.000 description 1
- 101800000733 Angiotensin-2 Proteins 0.000 description 1
- 206010003210 Arteriosclerosis Diseases 0.000 description 1
- 229940127291 Calcium channel antagonist Drugs 0.000 description 1
- 206010008111 Cerebral haemorrhage Diseases 0.000 description 1
- 206010013183 Dislocation of vertebra Diseases 0.000 description 1
- 208000032928 Dyslipidaemia Diseases 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- 208000004930 Fatty Liver Diseases 0.000 description 1
- 206010017711 Gangrene Diseases 0.000 description 1
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 1
- 102000001554 Hemoglobins Human genes 0.000 description 1
- 108010054147 Hemoglobins Proteins 0.000 description 1
- 206010019708 Hepatic steatosis Diseases 0.000 description 1
- 101000852815 Homo sapiens Insulin receptor Proteins 0.000 description 1
- 208000035150 Hypercholesterolemia Diseases 0.000 description 1
- 208000031226 Hyperlipidaemia Diseases 0.000 description 1
- 208000013016 Hypoglycemia Diseases 0.000 description 1
- CZGUSIXMZVURDU-JZXHSEFVSA-N Ile(5)-angiotensin II Chemical compound C([C@@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CC=1NC=NC=1)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CC=1C=CC=CC=1)C([O-])=O)NC(=O)[C@@H](NC(=O)[C@H](CCCNC(N)=[NH2+])NC(=O)[C@@H]([NH3+])CC([O-])=O)C(C)C)C1=CC=C(O)C=C1 CZGUSIXMZVURDU-JZXHSEFVSA-N 0.000 description 1
- 102100036721 Insulin receptor Human genes 0.000 description 1
- 206010054805 Macroangiopathy Diseases 0.000 description 1
- 206010027525 Microalbuminuria Diseases 0.000 description 1
- 241000699670 Mus sp. Species 0.000 description 1
- 206010029155 Nephropathy toxic Diseases 0.000 description 1
- 208000037273 Pathologic Processes Diseases 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- 208000022329 Protein metabolism disease Diseases 0.000 description 1
- 208000001647 Renal Insufficiency Diseases 0.000 description 1
- 206010038372 Renal arteriosclerosis Diseases 0.000 description 1
- 229940100389 Sulfonylurea Drugs 0.000 description 1
- 229940123464 Thiazolidinedione Drugs 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- 238000013019 agitation Methods 0.000 description 1
- 230000001476 alcoholic effect Effects 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 239000002333 angiotensin II receptor antagonist Substances 0.000 description 1
- 229940126317 angiotensin II receptor antagonist Drugs 0.000 description 1
- 229950006323 angiotensin ii Drugs 0.000 description 1
- 230000006907 apoptotic process Effects 0.000 description 1
- 208000011775 arteriosclerosis disease Diseases 0.000 description 1
- 210000000227 basophil cell of anterior lobe of hypophysis Anatomy 0.000 description 1
- 239000002876 beta blocker Substances 0.000 description 1
- 229940097320 beta blocking agent Drugs 0.000 description 1
- 230000017531 blood circulation Effects 0.000 description 1
- 210000004556 brain Anatomy 0.000 description 1
- 239000000872 buffer Substances 0.000 description 1
- 239000007853 buffer solution Substances 0.000 description 1
- 239000000480 calcium channel blocker Substances 0.000 description 1
- 208000020450 carbohydrate metabolism disease Diseases 0.000 description 1
- 150000001720 carbohydrates Chemical class 0.000 description 1
- 235000014633 carbohydrates Nutrition 0.000 description 1
- 230000007211 cardiovascular event Effects 0.000 description 1
- 210000004027 cell Anatomy 0.000 description 1
- 208000026106 cerebrovascular disease Diseases 0.000 description 1
- 239000000084 colloidal system Substances 0.000 description 1
- 230000001447 compensatory effect Effects 0.000 description 1
- 238000011443 conventional therapy Methods 0.000 description 1
- 230000001186 cumulative effect Effects 0.000 description 1
- 230000001934 delay Effects 0.000 description 1
- 238000001514 detection method Methods 0.000 description 1
- 238000009792 diffusion process Methods 0.000 description 1
- 208000035475 disorder Diseases 0.000 description 1
- 239000002270 dispersing agent Substances 0.000 description 1
- 239000012153 distilled water Substances 0.000 description 1
- 239000002552 dosage form Substances 0.000 description 1
- 230000002526 effect on cardiovascular system Effects 0.000 description 1
- 239000012636 effector Substances 0.000 description 1
- 230000003203 everyday effect Effects 0.000 description 1
- 230000029142 excretion Effects 0.000 description 1
- 208000010706 fatty liver disease Diseases 0.000 description 1
- 230000003849 fibrinolytic balance Effects 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- 210000004051 gastric juice Anatomy 0.000 description 1
- WIGIZIANZCJQQY-RUCARUNLSA-N glimepiride Chemical compound O=C1C(CC)=C(C)CN1C(=O)NCCC1=CC=C(S(=O)(=O)NC(=O)N[C@@H]2CC[C@@H](C)CC2)C=C1 WIGIZIANZCJQQY-RUCARUNLSA-N 0.000 description 1
- 229960004346 glimepiride Drugs 0.000 description 1
- 239000008187 granular material Substances 0.000 description 1
- 239000004519 grease Substances 0.000 description 1
- 210000002216 heart Anatomy 0.000 description 1
- 230000002440 hepatic effect Effects 0.000 description 1
- 238000004128 high performance liquid chromatography Methods 0.000 description 1
- 230000003516 hyperlipidaemic effect Effects 0.000 description 1
- 230000001631 hypertensive effect Effects 0.000 description 1
- 230000001077 hypotensive effect Effects 0.000 description 1
- 238000001727 in vivo Methods 0.000 description 1
- 208000017745 inborn carbohydrate metabolic disease Diseases 0.000 description 1
- 230000003914 insulin secretion Effects 0.000 description 1
- 210000004153 islets of langerhan Anatomy 0.000 description 1
- 201000006370 kidney failure Diseases 0.000 description 1
- 230000003902 lesion Effects 0.000 description 1
- 238000012417 linear regression Methods 0.000 description 1
- 210000005229 liver cell Anatomy 0.000 description 1
- 230000005923 long-lasting effect Effects 0.000 description 1
- 235000020905 low-protein-diet Nutrition 0.000 description 1
- 230000010534 mechanism of action Effects 0.000 description 1
- 230000004060 metabolic process Effects 0.000 description 1
- 239000012982 microporous membrane Substances 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 210000003205 muscle Anatomy 0.000 description 1
- 230000003387 muscular Effects 0.000 description 1
- 230000007694 nephrotoxicity Effects 0.000 description 1
- 231100000417 nephrotoxicity Toxicity 0.000 description 1
- 201000001119 neuropathy Diseases 0.000 description 1
- 230000007823 neuropathy Effects 0.000 description 1
- 230000007935 neutral effect Effects 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- 229940127017 oral antidiabetic Drugs 0.000 description 1
- 239000003538 oral antidiabetic agent Substances 0.000 description 1
- 210000000056 organ Anatomy 0.000 description 1
- 238000007427 paired t-test Methods 0.000 description 1
- 230000008506 pathogenesis Effects 0.000 description 1
- 230000009054 pathological process Effects 0.000 description 1
- 208000033808 peripheral neuropathy Diseases 0.000 description 1
- 210000002824 peroxisome Anatomy 0.000 description 1
- 229920000136 polysorbate Polymers 0.000 description 1
- 230000000291 postprandial effect Effects 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 239000000047 product Substances 0.000 description 1
- 238000004393 prognosis Methods 0.000 description 1
- 230000000770 proinflammatory effect Effects 0.000 description 1
- 230000035755 proliferation Effects 0.000 description 1
- 102000004169 proteins and genes Human genes 0.000 description 1
- 108090000623 proteins and genes Proteins 0.000 description 1
- 210000001187 pylorus Anatomy 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 102000005962 receptors Human genes 0.000 description 1
- 108020003175 receptors Proteins 0.000 description 1
- 239000013558 reference substance Substances 0.000 description 1
- 230000008929 regeneration Effects 0.000 description 1
- 238000011069 regeneration method Methods 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 230000002441 reversible effect Effects 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 239000007964 self emulsifier Substances 0.000 description 1
- 231100000240 steatosis hepatitis Toxicity 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 238000005728 strengthening Methods 0.000 description 1
- 238000010254 subcutaneous injection Methods 0.000 description 1
- 239000007929 subcutaneous injection Substances 0.000 description 1
- YROXIXLRRCOBKF-UHFFFAOYSA-N sulfonylurea Chemical class OC(=N)N=S(=O)=O YROXIXLRRCOBKF-UHFFFAOYSA-N 0.000 description 1
- 238000013268 sustained release Methods 0.000 description 1
- 239000012730 sustained-release form Substances 0.000 description 1
- 230000008961 swelling Effects 0.000 description 1
- 208000011580 syndromic disease Diseases 0.000 description 1
- 210000004026 tunica intima Anatomy 0.000 description 1
- 238000000825 ultraviolet detection Methods 0.000 description 1
- 230000002485 urinary effect Effects 0.000 description 1
- 210000002700 urine Anatomy 0.000 description 1
- 230000007923 virulence factor Effects 0.000 description 1
- 239000000304 virulence factor Substances 0.000 description 1
- 230000003820 β-cell dysfunction Effects 0.000 description 1
Images
Landscapes
- Medicinal Preparation (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
The invention discloses a medicinal composite tablet of a pioglitazone medicine. The medicinal composite tablet comprises a self-emulsifying medicine release part and a water-soluble carrier part, wherein the self-emulsifying medicine release part comprises pioglitazone, an oil phase, an emulsifier and a promotion emulsifier in the mass ratio of 1:(2-25):(1-10): (1-5). The self-emulsifying tablet can increase the total area of dissolution of the pioglitazone medicine. Compared with the traditional preparation, the medicine is relatively long in retention time in a human body and more facilitates the absorption of the human body. The self-emulsifying preparation provided by the invention can reduce the drug metabolism from kidneys, and the relapse of bladder cancer is reduced to a certain extent. In addition, phosphatidylcholine in the preparation also has the activity of repairing diabetic complications, such as liver impairment, caused by diseases, such as diabetic hypertension and primary hypertension, so that the treatment effect of the preparation is improved.
Description
Technical field
The present invention relates to a kind of medicine preparation, relate to a kind of pioglitazone hydrochloride composition tablet more specifically.
Technical background
Diabetes are to be the syndrome of basic biochemical character to continue hyperglycemia.Cause insulin to be enough to relatively or anything but and in various degree insulin resistant by a variety of causes, make carbohydrate, fat and protein metabolism disorder, clinical manifestation is diabetes.Along with the prolongation of the course of disease, blood capillary and macroangiopathy widely can appear, cause losing the sight of both eyes, renal failure, acromelic gangrene, myocardial infarction and cerebrovascular disease serious threat patient's life.Wherein type 2 diabetes mellitus accounts for 90%~95%, and the pathogenesis basis of type 2 diabetes mellitus is insulin resistant and β cell dysfunction.Discovering so-called " type 2 diabetes mellitus ", is under the effect of various paathogenic factors, forms through very long pathological process.Because the existence of virulence factor, normal blood constitutional balance is destroyed, and insulin is renderd a service and weakened relatively in the blood, through the startup of body internal feedback system, at first involves islets of langerhans, makes it long-term overwork and loses compensatory capacity.Be main feature clinically with the hyperglycemia; The secular blood sugar increasing of diabetics can quicken the beta Cell of islet apoptosis; Cause blood capillary, trunk and neuropathy widely, cause the functional lesion even the depletion of vitals such as the heart, brain, kidney, serious threat patient's life.Controlling needs strict blood sugar control, the protection target organ.The type 2 diabetes mellitus patient is tending towards more early, more being prone to take place cardiovascular complication.Except that hyperglycemia, dyslipidemia, the disorder of fibrinolytic balance all are the key factors that promotes that cardiovascular event takes place.Pioglitazone hydrochloride belongs to the thiazolidinedione oral antidiabetic drug, can improve cell to glucose and utilization through strengthening the sensitivity of tissue to insulin, and obvious the reduction on an empty stomach and post-prandial glycemia.Its mechanism of action is relevant with two receptors (ppary) of specificity peroxide activator enzyme paraphyte factor activator; Have the insulin of adjusting effector and transcribe and control the glucose generation; The effect of rotating and utilizing; Can increase the sensitivity of insulin, thereby play blood sugar reducing function liver, muscle insulin tissue.Glimepiride is a third generation sulfonylurea antidiabetic drug, has the inhibition hepatic glucose and synthesizes, promotes muscular tissue to the picked-up of periphery glucose and the effect of promotion insulin secretion.
But, discover that pioglitazone hydrochloride is dissolved in N, the N-dimethyl imide is slightly soluble in ethanol, is dissolved in acetone and acetonitrile hardly, water insoluble and ether, blood drug level maximum in human body is 30-50%, total bioavailability is 20-25%.Mainly be to improve the dissolution velocity of preparation to the research that improves biological pioglitazone hydrochloride bioavailability at present at human body.Chinese patent (publication number: CN101269040A) disclose a kind of pioglitazone hydrochloride sustained-release micropill and preparation method thereof; Utilize hydrophilic framework material and hydrophobicity framework material as substrate; Process dispersant with pioglitazone hydrochloride; Make medicine be molecule, colloid or microcrystalline state and be scattered in the substrate, the total surface area of medicine strengthens, and improves bioavailability.
Summary of the invention
The present invention is directed to the pioglitazone hydrochloride bad water-solubility for addressing the above problem; A kind of self emulsifying tablet is provided; From increasing medicine at the intravital time of staying of people, raising drug solubility; Three aspects such as total surface area that increase medicine solve the not high problem of pioglitazone hydrochloride bioavailability, make the drug effect cycle elongated; Self-emulsifier can increase the absorption of liver to medicine simultaneously, has certain targeting, reduces the injury of medicine to kidney; More helping medicine plays a role.Phosphatidylcholine not only can play the effect of emulsifying agent simultaneously, and simultaneously, phosphatidylcholine has the effect that cuts grease; Can promote blood circulation, improve serum lipids, remove peroxide; Make cholesterol and the reduction of neutral fat content in the blood; Reduce fat in the holdup time of blood vessel, promote the dissipation of atherosis speckle, prevent the tunica intima damage that causes by cholesterol.Take phosphatidylcholine hyperlipidemia and hypercholesterolemia are had obvious effect, thereby can prevent and treat arteriosclerosiss such as hypertension, myocardial infarction, cerebral hemorrhage.In addition, phosphatidylcholine not only can prevent fatty liver, can also promote liver cell regeneration, and simultaneously, phosphatidylcholine can reduce serum cholesterol content, prevents liver cirrhosis and helps liver and the recovery of renal function.This drug regimen is lowering blood pressure, in the time of diseases such as treatment diabetic hypertension, essential hypertension, can also repair the hepar damnification that causes because of diseases such as diabetic hypertension and essential hypertensions.
A kind of pioglitazone hydrochloride drug regimen tablet; Comprise self-emulsifying drug delivery part and water-soluble solid carrier part; Said self-emulsifying drug delivery partly comprises pioglitazone hydrochloride, oil phase, emulsifying agent and short emulsifying agent; Wherein, the mass ratio of pioglitazone hydrochloride, oil phase, emulsifying agent and short emulsifying agent is 1:2-25:1-10:1-5.
Comprised phosphatidylcholine in the said emulsifying agent.
The mixture that except phosphatidylcholine, also comprises any one or two kinds in ethyoxyl polyoxyethylene glyceride, polyoxyethylene oleate and the tween 80 of liquid or solid in the said emulsifying agent.
Said emulsifying agent is the mixture of phosphatidylcholine and tween 80.
The weight ratio of said phosphatidylcholine and tween 80 is 1-2:1-2.
The weight ratio of said phosphatidylcholine and tween 80 is 1:1.
Said water-soluble solid carrier portion is the combination of any one or two kinds in microcrystalline Cellulose (MCC), lactose, cellulose acetate, ethyl cellulose, polyacrylic resin, silicone rubber, gelatin, Hydroxypropyl Methyl Cellulose Phthalate (I-IPMCAS-LG), cellulose acetate phthalate ester (CAP), dichloromethane and aerosil, hydroxypropyl emthylcellulose (HPMC) and the Ka Baibo resin (carbopol).
Said water-soluble solid carrier partly is the combination of hydroxypropyl emthylcellulose and Ka Baibo resin.
Said oil phase is any one or the two or more mixture in Oleum Ricini, soybean oil, Oleum Arachidis hypogaeae semen, olive oil, butyl oleate, Ethyl linoleate, isopropyl myristate, medium-chain fatty acid triacylglycerol, sad, capric acid triacylglycerol, oleic acid, linoleic acid, the linolenic acid triacylglycerol.
Said short emulsifying agent is a kind of in ethanol or the butanediol.
Useful technique effect of the present invention is: adopt the self emulsifying tablet not only can increase the gross area of pioglitazone hydrochloride medicine dissolution, and the relative conventional formulation of time that medicine is detained human body had than leap ahead, more helped absorption of human body; Self-emulsifiable preparation provided by the invention can reduce the metabolism of medicine from kidney, has reduced the morbidity of bladder cancer to a certain extent; The intermediary phosphatidylcholine of preparation also has the activity that can also repair the diabetic complications such as hepar damnification that cause because of diseases such as diabetic hypertension and essential hypertensions in addition, has improved the curative effect of preparation according to the invention.
Description of drawings
The emulsion droplet particle diameter of Fig. 1 different formulations;
The release profiles of Fig. 2 pioglitazone hydrochloride tablet medicine.
The specific embodiment
The research of embodiment 1 pioglitazone hydrochloride tablet drug release behavior
Pioglitazone hydrochloride (Pioglitazone hydrochloride; PGH) be Thiazolidine ketone antidiabetic drug; Act on peroxisome proliferation activated receptor-(PPAR-), increase the sensitivity of Insulin receptor INSR, be mainly used in the treatment type 2 diabetes mellitus; Its drug effect and dosage are dependency, have the characteristics of administration once a day.PGH not only untoward reaction is less, and can not cause hypoglycemic reaction.With tween 80 and phosphatidylcholine is emulsifying agent, is framework material with hydroxypropyl emthylcellulose (HPMC) and Ka Baibo resin (carbopol), optimizes prescription and has prepared the self emulsifying hydrophilic gel slow releasing tablet that meets the 24h release request, to improve bioavailability.
Precision takes by weighing quantitative pioglitazone hydrochloride raw material and adds in the quantitative buffer salt solution, 37 ℃ of water-baths, and electronic stirring, rotating speed is 50r/min, oil droplet presents situation in solution of every separated 30min observation.The preliminary pH value of confirming slow releasing tablet.
The consumption of emulsifying agent is chosen tween 80 and the phosphatidylcholine and the buffer salt of different proportion, and pioglitazone hydrochloride is granulated jointly, and get quantitative (amount that is equivalent to a slice) granule and be dissolved in the quantitative distilled water, electromagnetic agitation, rotating speed is 50r/min.Get the emulsion smear, observe emulsion droplet particle diameter and quantity down in 1600 power microscopes.Confirm the consumption of emulsifying agent with this.
The consumption of framework material is chosen the HPMC and the carbopol of different proportion, with medicine and the abundant mix homogeneously of adjuvant on the basis that emulsifying agent and buffer salt are confirmed; With dehydrated alcohol system soft material, 24 order stainless steel sifts are granulated, 45 ℃ of dryings; 8.5mm the scrobicula stamping, hardness 7 ± 0.5kg.
Precision takes by weighing the about 20mg of pioglitazone hydrochloride, puts in the 100ml volumetric flask, adds simulated gastric fluid dissolving standardize solution, makes storing solution.Accurate this liquid 0.5,1,2,3 of drawing; 4,5ml adds simulated gastric fluid dilution standardize solution in the 10ml volumetric flask, and the 269nm place measures trap (A); Make linear regression with corresponding concentration (c), get standard curve equation: c (μ g/ml)=0.1671A+0.00175, r=0.9999.
According to 2010 editions appendix dissolution determination first methods of Chinese Pharmacopoeia, release medium (simulated gastric fluid) 900ml, 37 ± 0.5 ℃ of temperature, rotating speed is 50r/min; In 1,4,8,10; 12,16,20, the 24h 5ml (adding the equivalent medium simultaneously) that respectively takes a sample; Discharge liquid through filtering with microporous membrane, measure absorption value in the 260nm place, calculating cumulative discharges percentage rate.
The situation of pioglitazone hydrochloride in different medium observed
Discover that along with increasing of buffer pH value, the oil droplet time of occurrence in advance.When pH value is respectively 7.6,7.8,8.0 o'clock, the oil droplet time of occurrence was respectively 4.2,2.5,0.2h, and self-emulsifying drug delivery partly is transformed into the quantity showed increased of oily molecular state.But consider that the hypertensive patient needs the characteristics of long-term prescription, the prescription pH value again can not be too high, and therefore when pH=8.0, oil droplet 0.2h occurs, and meets requirement of experiment.
The emulsion droplet particle diameter is seen table 1 with shown in Figure 1, can know that Fg prescription particle diameter is minimum.
The composition of table 1 emulsifying agent
A, b, c, d: emulsifying agent/tablet (g/g) A, B, C: phosphatidylcholine/tween 80 (g/g)
Experiment showed, that the size of emulsion droplet particle diameter and the total amount and the ratio of the two of phosphatidylcholine and tween 80 all have relation, is not The more the better.When the total amount of emulsifying agent was 10%, gained prescription size ratio was less.Further optimize, the ratio of phosphatidylcholine and tween is transferred to 2:1 from 1:2 respectively, the result is with the prescription particle diameter minimum of 1:1.
On the basis of above-mentioned research, be aided with the hydrophilic blocker as framework material, consumption and ratio through the adjustment framework material obtain 9 prescriptions, and as shown in table 2, its release curve is shown in accompanying drawing 2.The blocker total amount is many more, and release is slow more.In certain scope, HPMC is many more, and release is slow more.With the Higuchi equation of statement Fick ' s first law and the single index equation of reaction diffusion and the comprehensive function of skeleton swelling the release parameter of 9 prescriptions is carried out regression treatment respectively, it is more reasonable that the result shows with the higuchi equation model.Try to achieve the release parameter T of each prescription according to regression equation
15, T
50, T
90As shown in table 3.Can be known by the result, contain blocker 30%, HPMC and carbopol are the prescription F of 1:3
5, discharging 15% dose needs about 1h, and discharging 50% dose needs about 8h, 23h release 90%.
The composition of table 2 framework material
The Higuish fit equation of 9 prescriptions is following:
F1:Q%=0.20l4t
1/2+0.1073,r=0.9469
F2:Q%=0.2179t
1/2-0.0025,r=0.9695
F3:Q%=0.2015t
1/2-0.0671,r=0.9873
F4:Q%=0.2106t
1/2+0.0257,r=0.9590
F5:Q%=0.2145t
1/2-0.0145,r=0.9709
F6:Q%=0.1904t
1/2-0.0957,r=0.9879
F7:Q%=0.1999t
1/2-0.0114,r=0.9671
F8:Q%=0.2062t
1/2-0.0404,r=0.9695
F9:Q%=0.1811t
1/2-0.0894,r=0.1862
The drug parameters that table 3 pioglitazone hydrochloride tablet discharges
Comprehensive above-mentioned research can be found after the drug oral, usually about Entogastric lingering 2h; In this section process, gastric juice can only see through tablet surface on a small quantity, fails to arrive label; Therefore tablet still can not form complete gel layer, and the gastric acid buffer salt with tablet surface of merely hitting does not change the label pH value.Take medicine behind the 2h, tablet arrives near the pylorus, and this position pH value has reached 5~6, and the buffer salt in the tablet is enough to overcome weak acidic medium, and remains on pH 8.0.In dosage form, add blocker, can slow down the change of gastric acid on the one hand, thereby guarantee the needed pH value of emulsifying the tablet pH value; Emulsion droplet is swum out of lentamente, to reach long lasting purpose from skeleton.
The preparation of embodiment 2 pioglitazone hydrochloride tablets
The mass ratio of pioglitazone hydrochloride, oil phase, emulsifying agent and short emulsifying agent is 1:2-25:1-10:0.2-5.
According to embodiment 1 described research conclusion, preparation pioglitazone hydrochloride tablet takes by weighing pioglitazone hydrochloride 15g, Oleum Ricini 30g, and phosphatidylcholine 10g, soil temperature-80 5g, ethanol 3g is prepared into the self-emulsifying drug delivery part; Take by weighing HPMC 2.72g, carbopol10.88g.With medicine and the abundant mix homogeneously of adjuvant, with dehydrated alcohol system soft material, 24 order stainless steel sifts are granulated, 45 ℃ of dryings, the stamping of 8.5mm scrobicula, hardness 7 ± 0.5kg.Obtain pioglitazone hydrochloride tablet medicine group I.
Adopt above-mentioned same procedure, take by weighing pioglitazone hydrochloride 15g, Oleum Ricini 45g, phosphatidylcholine 30g, soil temperature-80 30g, ethanol 15g, HPMC 8.1g, carbopol 24.3g.Obtain pioglitazone hydrochloride tablet medicine group II.
Adopt above-mentioned same procedure, take by weighing pioglitazone hydrochloride 15g, Oleum Ricini 375g, phosphatidylcholine 50g, soil temperature-80 100g, ethanol 75g, HPMC 82g, carbopol 164g.Obtain pioglitazone hydrochloride tablet medicine group III.
The tissue distribution research of embodiment 3 pioglitazone hydrochloride self-emulsifying drug deliveries part in liver, kidney
1. test material
Pioglitazone hydrochloride self-emulsifying drug delivery part (the drug group I for preparing according to the method for the embodiment of the invention 2; Adopt the normal saline dilution according to desired concn during test; Every bottle of hydrochloric pioglitazone 2.5g of 100ml), oral administration gavage pioglitazone hydrochloride solution (adopting 5% alcoholic solution that pioglitazone hydrochloride is configured to 6mg/ml solution).The SD rat (male, quality 200 ± 20g).
2. instrument and chromatographic condition
The Waters2695 high performance liquid chromatograph, the Waters2996 UV-detector; Symmetry shield C18 chromatographic column (4.6mm * 250mm, 5 μ m); Mobile phase acetonitrile: water (35:65); Flow velocity 0.5ml/min, 30 ℃ of column temperatures, ultraviolet detection wavelength 269nm, sample size are 20 μ l.
3. method and result
3.1 sample collecting and processing
Get 18 of rats, be divided into 2 groups at random, 9 every group, fasting 12h before the experiment; 60mg/kg according to dosage, one group of oral administration gavage pioglitazone hydrochloride solution, another group oral administration gavage pioglitazone hydrochloride self-emulsifying drug delivery part, after the administration in different time points; Get 3 mice cervical vertebra dislocation execution for each every group, get mouse liver and kidney, blot, prepare homogenate liquid with normal saline after precision is weighed with filter paper after the normal saline rinsing; Get equal serosity 0.1g, add methanol 0.5ml, vortex; Vortex vibration 1min, centrifugal 10min (12000r/min), supernatant dries up with nitrogen current; Residue adds 0.1ml methanol ultrasonic dissolution, and centrifugal 10min (12000r/min) gets supernatant and carries out the HPLC analysis.
3.2 standard solution preparation
Precision takes by weighing pioglitazone hydrochloride reference substance 11.36mg, adopts dissolve with methanol, and standardize solution is 10ml, and fully mixing is processed the 1.136mg/ml stock solution.
3.3 standard curve preparation
With an amount of equal serosity of blank liver, nephridial tissue the standard stock solution is diluted to series standard solution respectively; Measure by 2 following chromatographic conditions; The record peak area is an abscissa with the concentration C, and peak area A is a vertical coordinate; Get the standard curve of pioglitazone hydrochloride in liver, kidney respectively, as shown in table 4.
The standard curve of table 4 pioglitazone hydrochloride in liver and kidney
3.4 mouse stomach and injection back tissue distribution
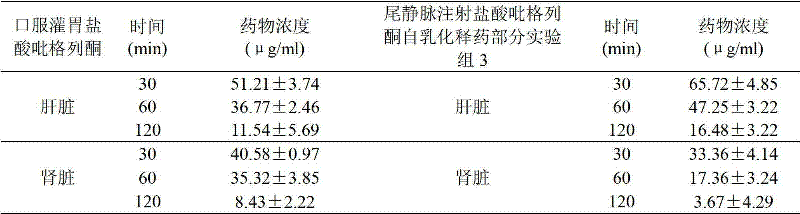
Behind dosage such as mouse stomach and intravenous injection (the 60 μ g/ml) pioglitazone hydrochloride, the distribution results of different time points medicine in liver and kidney is as shown in table 5.Can find out by table 5; The concentration of different time points medicine in liver all is higher than gastric infusion behind the intravenous injection pioglitazone hydrochloride fat milk, and the concentration in kidney all is lower than gastric infusion, explains that thus the pioglitazone hydrochloride self-emulsifying drug delivery partly has certain targeting property; Can make medicine more accumulate in liver after the intravenous injection; Reduce the drug level in the kidney, reach the raising therapeutic effect, reduce the effect of Toxicity of Kidney with this.
The concentration of pioglitazone hydrochloride in table 5 different time points liver and the kidney
The tissue distribution research 2 of embodiment 4 pioglitazone hydrochloride self-emulsifying drug deliveries part in liver, kidney
Hydrochloride for injection pioglitazone self-emulsifying drug delivery part ((the drug group II for preparing according to the method for the embodiment of the invention 2; Adopt the normal saline dilution according to desired concn during test; Every bottle of hydrochloric pioglitazone 2.5g of 100ml); Test method is identical with embodiment 3, investigates the tissue distribution research of pioglitazone hydrochloride self-emulsifying drug delivery part in liver, kidney.
With an amount of equal serosity of blank liver, nephridial tissue the standard stock solution is diluted to series standard solution respectively; Measure by 2 following chromatographic conditions; The record peak area is an abscissa with the concentration C, and peak area A is a vertical coordinate; Get the standard curve of pioglitazone hydrochloride in liver, kidney respectively, as shown in table 6.
The standard curve of table 6 pioglitazone hydrochloride in liver and kidney
Behind dosage such as mouse stomach and intravenous injection (the 60 μ g/ml) pioglitazone hydrochloride, the distribution results of different time points medicine in liver and kidney is as shown in table 7.Can find out by table 7; The concentration of different time points medicine in liver all is higher than gastric infusion behind the intravenous injection pioglitazone hydrochloride fat milk, and the concentration in kidney all is lower than gastric infusion, explains that thus the pioglitazone hydrochloride self-emulsifying drug delivery partly has certain targeting property; Can make medicine more accumulate in liver after the intravenous injection; Reduce the drug level in the kidney, reach the raising therapeutic effect, reduce the effect of Toxicity of Kidney with this.
The concentration of pioglitazone hydrochloride in table 7 different time points liver and the kidney
The tissue distribution research 3 of embodiment 5 pioglitazone hydrochloride self-emulsifying drug deliveries part in liver, kidney
Hydrochloride for injection pioglitazone self-emulsifying drug delivery part ((the drug group III for preparing according to the method for the embodiment of the invention 2; Adopt the normal saline dilution according to desired concn during test; Every bottle of hydrochloric pioglitazone 2.5g of 100ml); Test method is identical with embodiment 3, investigates the tissue distribution research of pioglitazone hydrochloride self-emulsifying drug delivery part in liver, kidney.
With an amount of equal serosity of blank liver, nephridial tissue the standard stock solution is diluted to series standard solution respectively; Measure by 2 following chromatographic conditions; The record peak area is an abscissa with the concentration C, and peak area A is a vertical coordinate; Get the standard curve of pioglitazone hydrochloride in liver, kidney respectively, as shown in table 8.
The standard curve of table 8 pioglitazone hydrochloride in liver and kidney
Behind dosage such as mouse stomach and intravenous injection (the 60 μ g/ml) pioglitazone hydrochloride, the distribution results of different time points medicine in liver and kidney is as shown in table 9.Can find out by table 9; The concentration of different time points medicine in liver all is higher than gastric infusion behind the intravenous injection pioglitazone hydrochloride fat milk, and the concentration in kidney all is lower than gastric infusion, explains that thus the pioglitazone hydrochloride self-emulsifying drug delivery partly has certain targeting property; Can make medicine more accumulate in liver after the intravenous injection; Reduce the drug level in the kidney, reach the raising therapeutic effect, reduce the effect of Toxicity of Kidney with this.
The concentration of pioglitazone hydrochloride in table 9 different time points liver and the kidney
Embodiment 6 pioglitazone hydrochloride tablets are investigated the activity of early diabetic nephropathy
Press WHO diagnosis of diabetes Standard Selection type 2 diabetes mellitus patient, 24h urinary albumin excretion rate (UAER) meets the III phase diagnostic criteria of diabetic nephropathy all at 20~200/min.All cases are all got rid of primary glomerulopathy, renal arteriosclerosis and the secondary glomerulopathy that uses nephrotoxicity medicine etc. to cause in the recent period.Be divided into matched group and drug group I, drug group II and drug group III at random.Matched group 31 examples, wherein male 16 examples, women 15 examples, 51~70 years old age, average (54.5 ± 8.5) year; Drug group I 31 examples, wherein male 17 examples, women 14 examples, 50~70 years old age, average (53.5 ± 10.5) year.Drug group II 31 examples, wherein male 16 examples, women 15 examples, 50~70 years old age, average (54.1 ± 10.8) year.Drug group III 31 examples, wherein male 15 examples, women 16 examples, 50~70 years old age, average (53.4 ± 10.4) year.There are no significant for 4 groups of sexes, age and course of disease differences.
All patients all give oral hypoglycemic thing blood sugar control; Part patient is the subcutaneous injection insulin simultaneously, gives the blood lipid-lowering medicine treatment simultaneously for merging hyperlipemic patients, and the complicated hypertension patient gives controlling blood pressure; The general calcium channel blocker, beta-blocker used; Low protein diet, every day albumin intake moon 1.0g/Kg.2 groups all give conventional therapy, and matched group is oral commercially available pioglitazone hydrochloride sheet simultaneously, and dosage is pioglitazone hydrochloride tablet 40mg/d.Pioglitazone hydrochloride tablet medicine group I, drug group II and drug group III that the oral embodiment of the invention 2 of drug group I, drug group II and drug group III provides; Dosage is pioglitazone hydrochloride 43mg/d; 2 groups of patients all treated 6 months, observed the variation of each parameter after 6 months.
Data represent with mean ± standard deviation, and curative effect is relatively used the paired t-test analysis before and after the treatment, and < 0.05 has significance for difference to P.2 groups of patients glycolated hemoglobin (HbAlc), Ccr, serum potassium concentration (cK) before and after treatment, blood lipid level is not statistically significant (P>0.05) relatively all, and ordinary circumstance is seen shown in the table 10; UAER compares before and after 2 groups of patient treatments, and is as shown in table 11.
Annotate: with compare P<0.05 before the treatment
Modern medicine study proves that sugared microalbuminuria is the index of diagnosis early diabetic nephropathy, also is the important indicator of judging prognosis and curative effect, and making urinaryalbumin reduce even recover normally is the key that delays and reverse DN.Albuminuretic control confirms that angiotensin ii receptor antagonist (ARB) can play a protective role to the renal function of early stage DN; This protective effect is to be independent of outside the hypotensive effect, raises relevant with the direct regulating action of blood vessel in the kidney, the pro-inflammatory that suppresses Angiotensin II, inhibition TGF-B.
Detection type 2 diabetes mellitus patient's UAER can be used as the diagnosing diabetes nephropathy and judges the index of its progress, and UAER obviously reduced after drug group I case-finding used pioglitazone hydrochloride sheet provided by the invention, and significant difference (P<0.05) is arranged.This result of study shows that the more traditional pioglitazone hydrochloride tablet of pioglitazone hydrochloride sheet provided by the invention not only has the urine protein effect of reduction and delay the diabetics renal function injury all more remarkable.Phosphatidylcholine can reduce serum cholesterol content, prevents liver cirrhosis and helps liver and the recovery of renal function; Have collaborative hypoglycemic activity with pioglitazone hydrochloride in vivo, strengthened the curative effect of product, take pioglitazone hydrochloride tablet provided by the invention diabetes early nephropathy patient's UAER is obviously descended, the kidney protection effect is arranged.Conventional this medicine that uses of patient can delay making progress of diabetic nephropathy.
Claims (10)
1. pioglitazone hydrochloride drug regimen tablet; It is characterized in that: comprise self-emulsifying drug delivery part and water-soluble solid carrier part; Said self-emulsifying drug delivery partly comprises pioglitazone hydrochloride, oil phase, emulsifying agent and short emulsifying agent; Wherein, the mass ratio of pioglitazone hydrochloride, oil phase, emulsifying agent and short emulsifying agent is 1:2-25:1-10:0.2-5.
2. pioglitazone hydrochloride tablet according to claim 1 is characterized in that: comprised phosphatidylcholine in the said emulsifying agent.
3. pioglitazone hydrochloride tablet according to claim 2 is characterized in that: the mixture that except phosphatidylcholine, also comprises any one or two kinds in ethyoxyl polyoxyethylene glyceride, polyoxyethylene oleate and the tween 80 of liquid or solid in the said emulsifying agent.
4. pioglitazone hydrochloride tablet according to claim 3 is characterized in that: said emulsifying agent is the mixture of phosphatidylcholine and tween 80.
5. pioglitazone hydrochloride tablet according to claim 4 is characterized in that: the weight ratio of said phosphatidylcholine and tween 80 is 1-2:1-2.
6. pioglitazone hydrochloride tablet according to claim 5 is characterized in that: the weight ratio of said phosphatidylcholine and tween 80 is 1:1.
7. pioglitazone hydrochloride tablet according to claim 1 is characterized in that: said water-soluble solid carrier portion is for dividing the combination of any one or two kinds in a microcrystalline Cellulose, lactose, cellulose acetate, ethyl cellulose, polyacrylic resin, silicone rubber, gelatin, Hydroxypropyl Methyl Cellulose Phthalate, cellulose acetate phthalate ester, dichloromethane and aerosil, hydroxypropyl emthylcellulose and the Ka Baibo resin.
8. pioglitazone hydrochloride tablet according to claim 7 is characterized in that: said water-soluble solid carrier partly is the combination of hydroxypropyl emthylcellulose and Ka Baibo resin.
9. pioglitazone hydrochloride tablet according to claim 1 is characterized in that: said oil phase is any one or the two or more mixture in Oleum Ricini, soybean oil, Oleum Arachidis hypogaeae semen, olive oil, butyl oleate, Ethyl linoleate, isopropyl myristate, medium-chain fatty acid triacylglycerol, sad, capric acid triacylglycerol, oleic acid, linoleic acid, the linolenic acid triacylglycerol.
10. pioglitazone hydrochloride tablet according to claim 1 is characterized in that: said short emulsifying agent is a kind of in ethanol or the butanediol.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN 201210160578 CN102657626B (en) | 2012-05-23 | 2012-05-23 | Medicinal composite tablet of pioglitazone medicine |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN 201210160578 CN102657626B (en) | 2012-05-23 | 2012-05-23 | Medicinal composite tablet of pioglitazone medicine |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN102657626A true CN102657626A (en) | 2012-09-12 |
| CN102657626B CN102657626B (en) | 2013-07-17 |
Family
ID=46767298
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN 201210160578 Active CN102657626B (en) | 2012-05-23 | 2012-05-23 | Medicinal composite tablet of pioglitazone medicine |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN102657626B (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN107823151A (en) * | 2017-11-06 | 2018-03-23 | 广州市桐晖药业有限公司 | A kind of Rates of Pioglitazone Hydrochloride Tablets Preparation |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2001037808A1 (en) * | 1999-11-23 | 2001-05-31 | Lipocine, Inc. | Solid carriers for improved delivery of active ingredients in pharmaceutical compositions |
| CN1709249A (en) * | 2005-07-04 | 2005-12-21 | 宛六一 | Pioglitazone soft capsule and its preparing method |
| WO2006135693A2 (en) * | 2005-06-10 | 2006-12-21 | Novartis Ag | Direct compression formulation of dpp-iv inhibitors and glitazones, and process |
-
2012
- 2012-05-23 CN CN 201210160578 patent/CN102657626B/en active Active
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2001037808A1 (en) * | 1999-11-23 | 2001-05-31 | Lipocine, Inc. | Solid carriers for improved delivery of active ingredients in pharmaceutical compositions |
| WO2006135693A2 (en) * | 2005-06-10 | 2006-12-21 | Novartis Ag | Direct compression formulation of dpp-iv inhibitors and glitazones, and process |
| CN1709249A (en) * | 2005-07-04 | 2005-12-21 | 宛六一 | Pioglitazone soft capsule and its preparing method |
Non-Patent Citations (1)
| Title |
|---|
| 冯浩等: "马来酸罗格列酮胃漂浮型缓释片的研究", 《中国药科大学学报》, vol. 33, no. 3, 31 December 2002 (2002-12-31), pages 196 - 199 * |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN107823151A (en) * | 2017-11-06 | 2018-03-23 | 广州市桐晖药业有限公司 | A kind of Rates of Pioglitazone Hydrochloride Tablets Preparation |
Also Published As
| Publication number | Publication date |
|---|---|
| CN102657626B (en) | 2013-07-17 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20240342096A1 (en) | Modified release composition of orlistat and acarbose for the treatment of obesity and related metabolic disorders | |
| JP6333855B2 (en) | Delayed release compositions containing biguanides | |
| JP2018172392A (en) | Modified release 1-[(3-hydroxy-adamant-1-ylamino)-acetyl]-pyrrolidine-2(s)-carbonitrile formulation | |
| JP2018087214A (en) | Compositions comprising statins, biguanides and further agents for reducing cardiometabolic risk | |
| CN102727894B (en) | A kind of pharmaceutical composition and application thereof for the treatment of diabetes and complication thereof | |
| EP4545072A2 (en) | Use of vibegron to treat overactive bladder symptoms in men with benign prostatic hyperplasia | |
| US20220370446A1 (en) | Methods For Reducing Liver Fat and For Treating Fatty Liver Disorders | |
| CN102657626B (en) | Medicinal composite tablet of pioglitazone medicine | |
| CN102462772B (en) | Application of extract of total glucosides of picrorhiza in preparing medicines for preventing and treating fatty liver | |
| CN103417971A (en) | Dipeptidyl peptidase inhibitor and vitamin-B pharmaceutical composition and application thereof | |
| CN103230594A (en) | Medicine composition of alpha-glucosidase inhibitor and vitamin B | |
| CN107823151A (en) | A kind of Rates of Pioglitazone Hydrochloride Tablets Preparation | |
| CN104208034B (en) | A kind of Glimepiride medicinal composition tablets, preparation method and applications | |
| CN106110310A (en) | A kind of compound for reducing blood suger containing pioglitazone and preparation method thereof | |
| CN104906114A (en) | Metformin-gliquidone compound sustained-release capsule and preparation method thereof | |
| KR102817359B1 (en) | Pharmaceutical Formulation for Oral Administration comprising Sodium-Glucose Cotransporter-2 Inhibitors and Angiotensin Ⅱ Receptor Blockers | |
| US20250367137A1 (en) | Phloroglucinol Formulations And Methods Of Use | |
| CN102349906B (en) | Atorvastatin calcium and nicotinic acid composition and preparation method thereof | |
| CN103599540B (en) | Containing glinides and the medical composition and its use of vitamin B group | |
| EP4620459A1 (en) | Metformin hydrochloride enteric-coated tablet | |
| CN102218062B (en) | Medicine composition for treating diabetes mellitus | |
| CN1969855B (en) | Pharmaceutical composition having target organ protection function and usage thereof | |
| Kavadi | Role of Sotagliflozin in Managing Heart Failure in Diabetes | |
| CN100346783C (en) | Sustained released malotilate preparation for hepa titis and cirrhosis | |
| AU2024279669A1 (en) | Sparsentan for use in a method of treating iga-mediated diseases |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| CP01 | Change in the name or title of a patent holder |
Address after: North District 401122 Chongqing City No. 1010 garden Jinyu Road Patentee after: Chongqing kangkere Pharmaceutical Co., Ltd Address before: North District 401122 Chongqing City No. 1010 garden Jinyu Road Patentee before: CHONGQING CONQUER PHARMACEUTICAL Co.,Ltd. |
|
| CP01 | Change in the name or title of a patent holder |