CN102603610B - 1,3-dihydro-1-oxo-2H-isoindole compound, its preparation method and use - Google Patents
1,3-dihydro-1-oxo-2H-isoindole compound, its preparation method and use Download PDFInfo
- Publication number
- CN102603610B CN102603610B CN201210038183.7A CN201210038183A CN102603610B CN 102603610 B CN102603610 B CN 102603610B CN 201210038183 A CN201210038183 A CN 201210038183A CN 102603610 B CN102603610 B CN 102603610B
- Authority
- CN
- China
- Prior art keywords
- dihydro
- acid
- reaction
- compound
- amino
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
Landscapes
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
本发明公开了1,3-二氢-1-氧-2H-异吲哚类化合物(I)及其药学上可接受的盐,还公开了该类化合物的制备方法,及其在制备预防或/和治疗各种因整合素αvβ3介导所引起的肿瘤转移、肿瘤生长、实体瘤生长、血管新生、视网膜病变、黄斑变性、骨质疏松、关节炎、平滑肌细胞移行、和动脉粥样硬化症状或疾病药物中的用途,。The present invention discloses 1,3-dihydro-1-oxo-2H-isoindole compounds (I) and pharmaceutically acceptable salts thereof, as well as methods for preparing the compounds and their use in preparing drugs for preventing and/or treating various symptoms or diseases of tumor metastasis, tumor growth, solid tumor growth, angiogenesis, retinopathy, macular degeneration, osteoporosis, arthritis, smooth muscle cell migration, and atherosclerosis mediated by integrin α v β 3. .
Description
技术领域 technical field
本发明属药物化学领域,涉及一类1,3-二氢-1-氧-2H-异吲哚类化合物(I)及其药学上可接受的盐、其制备方法、药物组合物和在制备预防或/和治疗各种因整合素αvβ3介导所引起的症状或疾病药物中的用途,包括但不限于肿瘤转移、肿瘤生长、实体瘤生长、血管新生、视网膜病变、黄斑变性、骨质疏松、关节炎、平滑肌细胞移行、和动脉粥样硬化等。 The invention belongs to the field of medicinal chemistry, and relates to a class of 1,3-dihydro-1-oxo-2H-isoindole compounds (I) and pharmaceutically acceptable salts thereof, their preparation methods, pharmaceutical compositions and preparation methods. Prevention or/and treatment of various symptoms or diseases mediated by integrin α v β 3 , including but not limited to tumor metastasis, tumor growth, solid tumor growth, angiogenesis, retinopathy, macular degeneration, Osteoporosis, arthritis, smooth muscle cell migration, and atherosclerosis, etc.
背景技术 Background technique
血管新生(angiogenesis)是指从已存在的血管床中产生新生血管系统的过程,在正常生理情况下,血管生成被严格控制于某些短暂的、特定的生理过程,如生殖、发育过程和伤口愈合过程等,而存续的血管生成是某些病变,如:肿瘤组织的恶性生长、视网膜病变、黄斑变性、关节炎、骨质疏松等的特性。研究表明,肿瘤诱导的血管新生与肿瘤细胞的增殖、侵润和转移密切相关,新生血管不仅为肿瘤细胞的恶性生长提供充足的营养物质,而且还为肿瘤细胞的远位转移提供了通路,因此,抑制肿瘤诱导的血管新生,减少或阻断肿瘤组织的血液供应而将肿瘤细胞“饿死”,是防治肿瘤转移的有效途径。该饥饿疗法与传统的细胞毒类抗肿瘤药物相比,由于抗血管新生药物是通过减少或阻断肿瘤组织的血液供应而使肿瘤细胞增殖周期延长,促进肿瘤细胞坏死而起作用,而不是直接杀死肿瘤细胞,因此,这类药物对正常细胞无明显毒副作用,表现出较好的肿瘤靶向性;并且该疗法可克服传统化疗药物治疗过程中的抗药性问题,而抗药性是目前化疗治疗手段失败的重要原因之一。 Angiogenesis refers to the process of generating a new vascular system from an existing vascular bed. Under normal physiological conditions, angiogenesis is strictly controlled by certain transient, specific physiological processes, such as reproduction, developmental processes, and wounds. Healing process, etc., while persistent angiogenesis is characteristic of certain diseases, such as: malignant growth of tumor tissue, retinopathy, macular degeneration, arthritis, osteoporosis, etc. Studies have shown that tumor-induced angiogenesis is closely related to the proliferation, invasion and metastasis of tumor cells. New blood vessels not only provide sufficient nutrients for the malignant growth of tumor cells, but also provide a pathway for distant metastasis of tumor cells. Therefore, , inhibit tumor-induced angiogenesis, reduce or block the blood supply of tumor tissue and "starve" tumor cells to death, which is an effective way to prevent and treat tumor metastasis. Compared with traditional cytotoxic antitumor drugs, this starvation therapy works because antiangiogenesis drugs prolong the proliferation cycle of tumor cells and promote tumor cell necrosis by reducing or blocking the blood supply of tumor tissues, rather than directly Kill tumor cells, therefore, this type of drug has no obvious toxic and side effects on normal cells, and shows better tumor targeting; and this therapy can overcome the problem of drug resistance in the treatment process of traditional chemotherapy drugs, and drug resistance is a common problem in current chemotherapy. One of the important reasons for treatment failure. the
最近研究表明,肿瘤诱导的血管新生和肿瘤的转移与整合素(Integrins)的作用密切相关。整合素属细胞表面粘附分子家族,由α和β两个亚基通过非共价键连接而成的异源二聚体跨膜糖蛋白。迄今为止共发现了20多种整合素亚型,不同的亚型具有不同的细胞特性与粘附特性,它们通过介导细胞与细胞、细胞与细胞外基质(extracellular matrix,ECM)的粘附以及参与细胞的信号转导,而对细胞的粘附、增殖、转移、凋亡等起重要调控作用,其中整合素αvβ3在肿瘤生长、局部浸润、转移,特别是肿瘤诱导的血管生成等过程中起重要作用。 Recent studies have shown that tumor-induced angiogenesis and tumor metastasis are closely related to the function of integrins. Integrins belong to the family of cell surface adhesion molecules, which are heterodimeric transmembrane glycoproteins composed of α and β subunits connected by non-covalent bonds. So far, more than 20 integrin subtypes have been discovered, and different subtypes have different cell characteristics and adhesion characteristics. They mediate cell-cell, cell-extracellular matrix (ECM) adhesion and Participate in cell signal transduction, and play an important regulatory role in cell adhesion, proliferation, metastasis, apoptosis, etc. Among them, integrin α v β 3 plays an important role in tumor growth, local invasion, metastasis, especially tumor-induced angiogenesis, etc. important role in the process.
整合素αvβ3是由αv和β3两个亚基组成,分为胞外区、跨膜区和胞内区三部分,其中αv亚基有1018个氨基酸残基,分子量125kD,β3亚基有762个氨基酸残基,分子量105kD;对整合素αvβ3胞外区及其与配体复合物的晶体结构研究表明,其胞外区由12个结构域组成,其中α与β亚基的N末端部分组装成一个卵形的“头”,从“头”延伸出两条几 乎平行的“尾巴”;配体的结合发生在α和β亚基的接触区,配体结合区主要包括α亚基的β-propeller结构域和β亚基的βA结构域,并且需二价阳离子的参与,阳离子在配体结合中不仅与配体的酸性基团作用介导配体与αvβ3的结合,还能稳定αvβ3的配体结合表面。整合素αvβ3的内源性配基为细胞外基质成分(如:纤连蛋白(fibronectin)、层连蛋白(laminin)、透明质粘连蛋白(vitronectin)和血小板反应素(thromobospondin)等)中含精氨酰-甘氨酰-天冬氨酰(Arg-Gly-Asp,RGD,1)三肽的序列,并对细胞外基质成分的识别具有选择性,当αvβ3与基质成分中的RGD结合后,通过丝裂原激活的蛋白激酶途径使血管内皮细胞增殖、分化和迁移,促进管腔形成而成为新生血管。实验证明,使用外源性含RGD的序列肽或类似物,可抑制肿瘤细胞的粘附与迁移、诱导肿瘤细胞凋亡和抑制肿瘤血管新生,最终导致肿瘤生长受到抑制,甚至肿瘤消退。由于整合素αvβ3在正常的血管内皮细胞和绝大多数正常器官系统上很少表达,只是在受到血管生长因子(angiogenic growth factors)刺激下激活的血管内皮细胞、激活的破骨细胞和侵袭性的肿瘤细胞上高表达;并且新生血管内皮细胞属于遗传稳定的非恶性细胞,其表面的αvβ3受体没发生突变,在与抑制剂作用过程中不会产生耐药性问题;另外,αvβ3受体位于细胞表面,直接与血液接触,药物易于到达靶点;因此,整合素αvβ3是设计高效、高选择性的抗血管新生和抗肿瘤转移药物较理想的靶标。 Integrin α v β 3 is composed of α v and β 3 subunits, which are divided into three parts: extracellular region, transmembrane region and intracellular region, among which α v subunit has 1018 amino acid residues and a molecular weight of 125kD. The β 3 subunit has 762 amino acid residues and a molecular weight of 105kD; the study on the crystal structure of the extracellular region of integrin α v β 3 and its complex with the ligand shows that the extracellular region consists of 12 domains, of which α Assembled with the N-terminal part of the β subunit to form an oval "head", from which two almost parallel "tails"extend; the binding of the ligand occurs in the contact area between the α and β subunits, and the ligand The binding region mainly includes the β-propeller domain of the α subunit and the β A domain of the β subunit, and requires the participation of divalent cations. The cations not only interact with the acidic groups of the ligands to mediate ligand binding. Binding to α v β 3 can also stabilize the ligand-binding surface of α v β 3 . The endogenous ligands of integrin α v β 3 are extracellular matrix components (such as: fibronectin, laminin, vitaminectin, thrombospondin, etc.) Contains the sequence of arginyl-glycyl-aspartyl (Arg-Gly-Asp, RGD, 1) tripeptide, and is selective for the recognition of extracellular matrix components, when α v β 3 and matrix components After the combination of RGD, vascular endothelial cells proliferate, differentiate and migrate through the mitogen-activated protein kinase pathway, and promote the formation of tube lumen to become new blood vessels. Experiments have shown that the use of exogenous RGD-containing sequence peptides or analogs can inhibit the adhesion and migration of tumor cells, induce tumor cell apoptosis and inhibit tumor angiogenesis, and finally lead to inhibition of tumor growth and even tumor regression. Since integrin α v β 3 is rarely expressed in normal vascular endothelial cells and most normal organ systems, only activated vascular endothelial cells, activated osteoclasts and Invasive tumor cells are highly expressed; and neovascular endothelial cells are genetically stable non-malignant cells, and the α v β 3 receptors on their surface have not undergone mutations, and there will be no drug resistance problems in the process of interacting with inhibitors; In addition, the α v β 3 receptor is located on the cell surface, directly in contact with the blood, and the drug is easy to reach the target; therefore, integrin α v β 3 is ideal for the design of highly efficient and selective anti-angiogenesis and anti-tumor metastasis drugs target.
寻找整合素αvβ3抑制剂,对于发现抗血管新生和抗肿瘤转移药物具有重要意义,还能用于治疗因新生血管过度增生所导致的肿瘤生长、实体瘤生长、视网膜病变、黄斑变性、骨质疏松、关节炎、平滑肌细胞移行、和动脉粥样硬化等常见病症。 Finding integrin α v β 3 inhibitors is of great significance for the discovery of anti-angiogenesis and anti-tumor metastasis drugs, and can also be used to treat tumor growth, solid tumor growth, retinopathy, macular degeneration, Common conditions such as osteoporosis, arthritis, smooth muscle cell migration, and atherosclerosis.
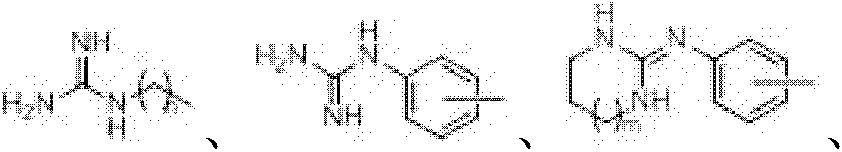
目前,以整合素αvβ3为作用靶点的抗血管新生和抗肿瘤转移药物尚处于临床或临床前试验阶段,这些抑制剂可分为三类,即:单克隆抗体、多肽和环肽及其模拟物、非肽类小分子。Vitaxin和Vitaxin II为Eli Lilly公司开发的αvβ3的单克隆抗体,能够与整合素αvβ3特异性结合,抑制肿瘤生长和血管新生,目前正进行II期临床试验,用来治疗前列腺癌、转移的黑色素瘤、晚期结肠癌以及风湿性关节炎。研究发现,使用外源性含RGD序列的线性肽或聚合肽,如:RGDS、GRGDS、poly-RGDS等可阻止bFGF、TNF-α和人各种肿瘤片 段诱导的血管新生,而对已存在的血管无影响。但这些肽类分子柔性强,导致对整合素αvβ3受体的选择性及亲和力都较差。环肽由于其构象被束缚在一定范围内,因此对不同的整合素具有较好的选择性。环肽cyclo(RGDfV)(Cyclo(-Arg-Gly-Asp-D-Phe-Val-),2)是一种高活性和高选择性的整合素αvβ3抑制剂,其体外抑制αvβ3与其配体蛋白结合的IC50为2.5nmol/L;在此基础上Merck公司研究人员将cyclo(RGDfV)肽链骨架甲基化,得到更高活性的多肽模拟物cyclo(RGDf-N(Me)V)(3,通用名Cilengitide),其IC50为0.58nmol/L,能够拮抗整合素αvβ3介导的血管内皮细胞与细胞外基质的相互作用,抑制黑色素瘤的生长和血管新生,并能够诱导在vitronectin上生长的脑肿瘤细胞的凋亡,目前Cliengitide正在进行II/III期临床试验,用于治疗肺癌、胰腺癌、乳腺癌、黑色素瘤、肾脏肿瘤以及结肠癌。由于抗体和肽类药物存在体内降解、半衰期短、生物利用度低需静脉给药、大量制备困难等不足,因此,寻找和发现高效、高选择性可口服给药的非肽类整合素αvβ3抑制剂是目前国内外抗血管新生和抗肿瘤转移药物研究的重要领域之一。 At present, anti-angiogenesis and anti-metastasis drugs targeting integrin α v β 3 are still in clinical or preclinical trials. These inhibitors can be divided into three categories, namely: monoclonal antibodies, peptides and cyclic peptides And its mimics, non-peptide small molecules. Vitaxin and Vitaxin II are α v β 3 monoclonal antibodies developed by Eli Lilly, which can specifically bind to integrin α v β 3 , inhibit tumor growth and angiogenesis, and are currently undergoing phase II clinical trials for the treatment of prostate carcinoma, metastatic melanoma, advanced colon cancer, and rheumatoid arthritis. Studies have found that the use of exogenous linear peptides or polymeric peptides containing RGD sequences, such as: RGDS, GRGDS, poly-RGDS, etc., can prevent angiogenesis induced by bFGF, TNF-α and various human tumor fragments, while the existing Blood vessels are unaffected. However, these peptide molecules are flexible, resulting in poor selectivity and affinity for integrin α v β 3 receptors. Cyclic peptides have better selectivity for different integrins due to their conformational constraints. The cyclic peptide cyclo(RGDfV) (Cyclo(-Arg-Gly-Asp-D-Phe-Val-), 2) is a highly active and selective integrin α v β 3 inhibitor, which inhibits α v The IC 50 of β3 binding to its ligand protein is 2.5nmol/L; on this basis, Merck researchers methylated the peptide chain backbone of cyclo(RGDfV) to obtain a more active polypeptide mimic cyclo(RGDf-N( Me)V)(3, common name Cilengitide), with an IC 50 of 0.58nmol/L, can antagonize the interaction between vascular endothelial cells and extracellular matrix mediated by integrin α v β 3 , inhibit the growth of melanoma and angiogenesis Neonatal and capable of inducing apoptosis in brain tumor cells grown on vitronectin, Clengitide is currently undergoing Phase II/III clinical trials for the treatment of lung, pancreatic, breast, melanoma, kidney and colon cancers. Due to the shortcomings of antibodies and peptide drugs such as in vivo degradation, short half-life, low bioavailability requiring intravenous administration, and difficulty in large-scale preparation, it is necessary to find and discover non-peptide integrin α v with high efficiency and high selectivity that can be administered orally β 3 inhibitors are one of the important fields of anti-angiogenesis and anti-tumor metastasis drug research at home and abroad.
发明内容 Contents of the invention
本发明的目的在于公开一类具有1,3-二氢-1-氧-2H-异吲哚类化学结构(I)的整合素αvβ3抑制剂或其药学上可接受的盐。 The purpose of the present invention is to disclose a class of integrin α v β 3 inhibitors with 1,3-dihydro-1-oxo-2H-isoindole chemical structure (I) or pharmaceutically acceptable salts thereof.
本发明的另一目的在于公开该1,3-二氢-1-氧-2H-异吲哚类化合物(I)的制备方法。 Another object of the present invention is to disclose the preparation method of the 1,3-dihydro-1-oxo-2H-isoindole compound (I). the
本发明的又一目的在于公开包含1,3-二氢-1-氧-2H-异吲哚类化合物(I)或其药学上可接受的盐的药物组合物。 Another object of the present invention is to disclose a pharmaceutical composition comprising 1,3-dihydro-1-oxo-2H-isoindole compound (I) or a pharmaceutically acceptable salt thereof. the
本发明的再一目的在于公开1,3-二氢-1-氧-2H-异吲哚类化合物(I)或其药学上可接受的盐在制备预防或/和治疗各种因整合素αvβ3介导所引起的症状或疾病药物中的用途,包括但不限于肿瘤转移、肿瘤生长、实体瘤生长、血管新生、视网膜病变、黄斑变性、骨质疏松、关节炎、平滑肌细胞移行、和动脉粥样硬化等。 Another object of the present invention is to disclose 1,3-dihydro-1-oxo-2H-isoindole compounds (I) or pharmaceutically acceptable salts thereof in the preparation of prevention or/and treatment of various factors integrin α v β 3 mediates the use of drugs in causing symptoms or diseases, including but not limited to tumor metastasis, tumor growth, solid tumor growth, angiogenesis, retinopathy, macular degeneration, osteoporosis, arthritis, smooth muscle cell migration, and atherosclerosis etc.
本发明提供的1,3-二氢-1-氧-2H-异吲哚类化合物(I)的化学结构通式为: 1,3-dihydro-1-oxygen-2H-isoindole compound (I) provided by the invention has a general chemical structure formula:
式中:R1表示H、苯基、2-呋喃基、2-噻吩基、3-噻吩基、3-吡啶基、4-吡啶基、3,4-亚甲基二氧基苯基、2,3-亚甲基二氧基苯基、或 R3、R4、R5各自独立地表示H、C1~C6烷基、C1~C6烷氧基、羟基、卤素、氨 基、硝基、氰基、羧基、CF3、NR7R8,R6表示H、C1~C6烷基、C1~C6烷氧基、卤素、硝基、氰基或CF3,R7、R8各自独立地表示C1~C6烷基,R3、R4、R5、R6可在苯环的任意可能位置,R3、R4、R5可以相同,也可以不同;R2表示 n表示1-3、m表示0-2,R9表示C1~C6烷基、卤素、羟基或CF3。 In the formula : R represents H, phenyl, 2-furyl, 2-thienyl, 3-thienyl, 3-pyridyl, 4-pyridyl, 3,4-methylenedioxyphenyl, 2 ,3-methylenedioxyphenyl, or R 3 , R 4 , and R 5 each independently represent H, C 1 -C 6 alkyl, C 1 -C 6 alkoxy, hydroxyl, halogen, amino, nitro, cyano, carboxyl, CF 3 , NR 7 R 8 and R 6 represent H, C 1 to C 6 alkyl, C 1 to C 6 alkoxy, halogen, nitro, cyano or CF 3 , R 7 and R 8 each independently represent C 1 to C 6 Alkyl, R 3 , R 4 , R 5 , R 6 can be in any possible position of the benzene ring, R 3 , R 4 , R 5 can be the same or different; R 2 means n represents 1-3, m represents 0-2, R 9 represents C 1 -C 6 alkyl, halogen, hydroxyl or CF 3 .
对于本发明所公开的1,3-二氢-1-氧-2H-异吲哚类化合物(I)分子中存在手性碳时,化合物(I)为消旋体或光学活性体。 When there is a chiral carbon in the molecule of the 1,3-dihydro-1-oxo-2H-isoindole compound (I) disclosed in the present invention, the compound (I) is a racemic body or an optically active body. the
本发明所公开的1,3-二氢-1-氧-2H-异吲哚类化合物(I)可通过以下方法制备得到: The 1,3-dihydro-1-oxo-2H-isoindole compound (I) disclosed in the present invention can be prepared by the following method:
式中:R1和R2的定义与化学结构通式(I)相同; In the formula: R 1 and R 2 are defined as the same as the chemical structure general formula (I);
以2-甲酰基-5-硝基苯甲酸(4)和β-丙氨酸甲酯类衍生物(5)为起始原料,在合适溶剂中经还原、环合得3-(6-硝基-1-氧异吲哚-2-基)丙酸甲酯(6),所得中间体(6)用还原剂将硝基还原为氨基,得3-(6-氨基-1-氧异吲哚-2-基)丙酸甲酯(7),然后与含胍基的羧酸类化合物(8)在缩合剂存在条件下缩合,得1,3-二氢-1-氧-2H-异吲哚甲酯类化合物(9),再经碱水解、酸中和,即得1,3-二氢-1-氧-2H-异吲哚类化合物(I)。 Using 2-formyl-5-nitrobenzoic acid (4) and β-alanine methyl ester derivatives (5) as starting materials, 3-(6-nitrobenzoic acid) is obtained through reduction and cyclization in a suitable solvent Base-1-oxoisoindol-2-yl) methyl propionate (6), the resulting intermediate (6) uses a reducing agent to reduce the nitro group to amino to obtain 3-(6-amino-1-oxoisoindol Indol-2-yl) methyl propionate (7), and then condensed with guanidine-containing carboxylic acid compound (8) in the presence of a condensing agent to obtain 1,3-dihydro-1-oxo-2H-iso Indole methyl ester compound (9) is hydrolyzed with alkali and acid neutralized to obtain 1,3-dihydro-1-oxo-2H-isoindole compound (I). the
上述方法各反应步骤具体描述如下: Each reaction step of the above-mentioned method is specifically described as follows:
A)以2-甲酰基-5-硝基苯甲酸(4)和β-丙氨酸甲酯类衍生物(5)为起始原料,在合适溶剂中经还原、环合得3-(6-硝基-1-氧异吲哚-2-基)丙酸甲酯(6);其中,反应所用溶剂为:C1-8脂肪醇、N,N-二甲基甲酰胺、醚类(如:乙醚、异丙醚、甲基叔丁基醚、四氢呋喃、乙二醇二甲醚等)、C1-6脂肪酸、C1-6脂肪酸与C1-6脂肪醇所形成酯、卤代烃(如:二氯甲烷、氯仿、1,2-二氯乙烷等)、芳香烃或取代芳香烃(如:苯、甲苯、二甲苯、氯苯、邻二氯苯 等)、或乙腈,优选为甲醇、乙醇、乙酸、乙酸乙酯、乙醚、二氯甲烷或四氢呋喃;所用还原剂为:KBH4、NaBH4、LiBH4、NaBH3CN,优选还原剂为NaBH4、KBH4;2-甲酰基-5-硝基苯甲酸(4)、β-丙氨酸甲酯类衍生物(5)和还原剂的摩尔投料比为1.0∶1.0~5.0:0.5~5.0,优选摩尔投料比为1.0∶1.1~3.0∶1.0~3.0;反应温度为-20℃~120℃,优选为0℃~100℃;反应时间为30分钟~72小时,优选为1~15小时。 A) With 2-formyl-5-nitrobenzoic acid (4) and β-alanine methyl ester derivatives (5) as starting materials, 3-(6) is obtained through reduction and cyclization in a suitable solvent -Nitro-1-oxoisoindol-2-yl)methyl propionate (6); wherein, the solvent used for the reaction is: C 1-8 fatty alcohol, N,N-dimethylformamide, ethers ( Such as: ether, isopropyl ether, methyl tert-butyl ether, tetrahydrofuran, ethylene glycol dimethyl ether, etc.), C 1-6 fatty acids, esters of C 1-6 fatty acids and C 1-6 fatty alcohols, halogenated Hydrocarbons (such as dichloromethane, chloroform, 1,2-dichloroethane, etc.), aromatic hydrocarbons or substituted aromatic hydrocarbons (such as: benzene, toluene, xylene, chlorobenzene, o-dichlorobenzene, etc.), or acetonitrile, Preferably methanol, ethanol, acetic acid, ethyl acetate, ether, dichloromethane or tetrahydrofuran; the reducing agent used is: KBH 4 , NaBH 4 , LiBH 4 , NaBH 3 CN, the preferred reducing agent is NaBH 4 , KBH 4 ; 2- The molar feeding ratio of formyl-5-nitrobenzoic acid (4), β-alanine methyl ester derivatives (5) and reducing agent is 1.0: 1.0~5.0: 0.5~5.0, preferably the molar feeding ratio is 1.0 : 1.1 to 3.0: 1.0 to 3.0; the reaction temperature is -20°C to 120°C, preferably 0°C to 100°C; the reaction time is 30 minutes to 72 hours, preferably 1 to 15 hours.
由步骤A)得到的3-(6-硝基-1-氧异吲哚-2-基)丙酸甲酯(6)在合适溶剂中,经化学还原或催化氢化还原,将硝基还原为氨基,得3-(6-氨基-1-氧异吲哚-2-基)丙酸甲酯(7);其中,用化学还原法时,反应所用溶剂为:C1~C6脂肪醇、四氢呋喃、乙醚、C1-6脂肪酸、C1-6脂肪酸与C1-6脂肪醇所形成酯、苯、甲苯、1,4-二氧六环、乙二醇二甲醚、二氯甲烷、氯仿、1,2-二氯乙烷、浓度为1%-36%的盐酸水溶液、浓度为1%-30%的氯化铵水溶液、或上述溶剂的混合物,优选为甲醇、乙醇、乙酸、乙酸乙酯、乙醚、二氯甲烷或四氢呋喃;所用还原剂为:锌粉、铁粉或二氯化锡;化合物(6)与还原剂的摩尔投料比为1.0∶1.0~10.0,优选摩尔投料比为1.0∶1.5~5.0;反应温度为0℃~150℃,优选反应温度为室温~溶剂回流温度;还原反应时间为0.5~72小时,优选反应时间为1~12小时; The 3-(6-nitro-1-oxoisoindol-2-yl) methyl propionate (6) obtained in step A) is in a suitable solvent, through chemical reduction or catalytic hydrogenation reduction, the nitro group is reduced to Amino, to get 3-(6-amino-1-oxoisoindol-2-yl) methyl propionate (7); Wherein, when using the chemical reduction method, the solvent used for the reaction is: C 1 ~ C 6 fatty alcohol, Tetrahydrofuran, ether, C 1-6 fatty acid, ester of C 1-6 fatty acid and C 1-6 fatty alcohol, benzene, toluene, 1,4 - dioxane, ethylene glycol dimethyl ether, dichloromethane, Chloroform, 1,2-dichloroethane, hydrochloric acid aqueous solution with a concentration of 1%-36%, ammonium chloride aqueous solution with a concentration of 1%-30%, or a mixture of the above solvents, preferably methanol, ethanol, acetic acid, acetic acid Ethyl ester, ether, dichloromethane or tetrahydrofuran; The reducing agent used is: zinc powder, iron powder or tin dichloride; The molar feed ratio of compound (6) and reducing agent is 1.0: 1.0~10.0, preferably molar feed ratio is 1.0:1.5~5.0; the reaction temperature is 0°C~150°C, preferably the reaction temperature is room temperature~solvent reflux temperature; the reduction reaction time is 0.5~72 hours, preferably the reaction time is 1~12 hours;
采用催化氢化还原时,反应所用溶剂为:C1-8脂肪醇、乙醚、异丙醚、甲基叔丁基醚、四氢呋喃、乙二醇二甲醚、C1-6脂肪酸、C1-6脂肪酸与C1-6脂肪醇所形成酯、苯、甲苯、二甲苯或乙腈,优选为甲醇、乙醇、乙酸或乙酸乙酯;所用催化剂为:1%~30%Pd-C、1%~30%Pd(OH)2-C、Raney-Ni,优选催化剂为:Raney-Ni、5%~20%Pd-C、5%~20%Pd(OH)2-C;化合物(6)与催化剂的质量比为1.0∶0.01~1.0;反应压力为常压~10.0MPa,优选为常压~2.0MPa;反应温度为室温~150℃,优选为室温~80℃;反应时间为1~48小时,优选为1~24小时。 When using catalytic hydrogenation reduction, the solvents used in the reaction are: C 1-8 fatty alcohol, diethyl ether, isopropyl ether, methyl tert-butyl ether, tetrahydrofuran, ethylene glycol dimethyl ether, C 1-6 fatty acid, C 1-6 Esters formed by fatty acids and C 1-6 fatty alcohols, benzene, toluene, xylene or acetonitrile, preferably methanol, ethanol, acetic acid or ethyl acetate; the catalysts used are: 1% to 30% Pd-C, 1% to 30 %Pd(OH) 2 -C, Raney-Ni, the preferred catalyst is: Raney-Ni, 5% to 20% Pd-C, 5% to 20% Pd(OH) 2 -C; compound (6) and catalyst The mass ratio is 1.0:0.01~1.0; the reaction pressure is normal pressure~10.0MPa, preferably normal pressure~2.0MPa; the reaction temperature is room temperature~150°C, preferably room temperature~80°C; the reaction time is 1~48 hours, preferably 1 to 24 hours.
由步骤B)得到的3-(6-氨基-1-氧异吲哚-2-基)丙酸甲酯(7)在合适溶剂中与含胍基的羧酸类化合物(8)在缩合剂存在条件下缩合,得1,3-二氢-1-氧-2H-异吲哚甲酯类化合物(9);其中,反应所用溶剂为:吡啶、N,N-二甲基甲酰胺、C3-8脂肪酮、醚类(如:乙醚、异丙醚、甲基叔丁基醚、四氢呋喃、乙二醇二甲醚等)、C1-6脂肪酸与C1-6脂肪醇所形成酯、卤代烃(如:二氯甲烷、氯仿、1,2-二氯乙烷等)、芳香烃或取代芳香烃(如:苯、甲苯、二甲苯、氯苯、邻二氯苯等)或乙腈,反应可在上述单一溶剂中进行,也可在上述混合溶剂中进行,混合溶剂体积比为1∶0.1~10,优选溶剂为吡啶、四氢呋喃、N,N-二甲基甲酰胺、丙酮、氯苯或乙腈;所用缩合剂为:氯甲酸C1-8脂肪醇酯类化合物(如:氯甲酸乙酯、氯甲酸叔丁酯、氯甲酸苄酯等)、N-乙氧羰基-2-乙氧基-1,2-二氢喹啉(EEDQ)、碳二亚胺类 化合物(如:二环己基碳二亚胺(简称为DCC)、1-乙基-3-(3-二甲胺丙基)碳二亚胺盐酸盐(简称为EDCI))、氰基磷酸二乙酯(DEPC)、2-氯-4,6-二甲氧基-1,3,5-三嗪(简称为CDMT)、氯化4-(4,6-二甲氧基-1,3,5-三嗪-2-基)-4-甲基吗啉盐(简称为DMTMM),其中,DMTMM中的阴离子为氯、溴、高氯酸根、氟硼酸根、甲烷磺酸基、苯磺酸基、对甲苯磺酸基、樟脑磺酸基、氨基磺酸基,优选缩合剂为:氯甲酸乙酯、二环己基碳二亚胺(DCC)、EDCI、DMTMM;化合物(7):化合物(8):缩合剂的摩尔投料比为1.0∶1.0~4.5∶1.0~4.5,优选摩尔投料比为1.0∶1.0~2.0∶1.0~2.0;缩合反应温度为0~130℃,优选反应温度为0~50℃;缩合反应时间为30分钟~72小时,优选反应时间为2~48小时。 3-(6-amino-1-oxoisoindol-2-yl) methyl propionate (7) obtained by step B) is in a condensing agent with a guanidine-containing carboxylic acid compound (8) in a suitable solvent Condensation under existing conditions, 1,3-dihydro-1-oxo-2H-isoindole methyl ester compound (9); wherein, the solvent used for the reaction is: pyridine, N, N-dimethylformamide, C 3-8 Fatty ketones, ethers (such as diethyl ether, isopropyl ether, methyl tert-butyl ether, tetrahydrofuran, ethylene glycol dimethyl ether, etc.), esters formed by C 1-6 fatty acids and C 1-6 fatty alcohols , halogenated hydrocarbons (such as: methylene chloride, chloroform, 1,2-dichloroethane, etc.), aromatic hydrocarbons or substituted aromatic hydrocarbons (such as: benzene, toluene, xylene, chlorobenzene, o-dichlorobenzene, etc.) or Acetonitrile, reaction can be carried out in above-mentioned single solvent, also can carry out in above-mentioned mixed solvent, mixed solvent volume ratio is 1: 0.1~10, preferred solvent is pyridine, tetrahydrofuran, N,N-dimethylformamide, acetone, Chlorobenzene or acetonitrile; the condensing agent used is: chloroformic acid C 1-8 fatty alcohol esters (such as: ethyl chloroformate, tert-butyl chloroformate, benzyl chloroformate, etc.), N-ethoxycarbonyl-2- Ethoxy-1,2-dihydroquinoline (EEDQ), carbodiimide compounds (such as: dicyclohexylcarbodiimide (abbreviated as DCC), 1-ethyl-3-(3-dimethyl Aminopropyl) carbodiimide hydrochloride (abbreviated as EDCI)), diethyl cyanophosphate (DEPC), 2-chloro-4,6-dimethoxy-1,3,5-triazine ( CDMT for short), 4-(4,6-dimethoxy-1,3,5-triazin-2-yl)-4-methylmorpholinium chloride (DMTMM for short), wherein, in DMTMM The anions are chlorine, bromine, perchlorate, fluoroborate, methanesulfonic acid, benzenesulfonic acid, p-toluenesulfonic acid, camphorsulfonic acid, aminosulfonic acid, and the preferred condensing agent is: ethyl chloroformate , dicyclohexylcarbodiimide (DCC), EDCI, DMTMM; Compound (7): compound (8): the molar feed ratio of condensing agent is 1.0: 1.0~4.5: 1.0~4.5, and preferred molar feed ratio is 1.0: 1.0~2.0:1.0~2.0; the condensation reaction temperature is 0~130°C, preferably the reaction temperature is 0~50°C; the condensation reaction time is 30 minutes~72 hours, preferably the reaction time is 2~48 hours.
由步骤C)得到的1,3-二氢-1-氧-2H-异吲哚甲酯类化合物(9)经碱水解、酸中和,得1,3-二氢-1-氧-2H-异吲哚类化合物(I),所得(I)再按照常规方法与相应的酸成盐,即得化合物(I)的盐;其中,水解所用碱为碱金属或碱土金属氢氧化物、碱金属或碱土金属碳酸盐、碱金属或碱土金属碳酸氢盐,优选碱为:氢氧化锂、碳酸钠;碱与化合物(9)的摩尔投料比为1.0~5.0∶1.0,优选摩尔投料比为1.1~2.5∶1.0;水解反应温度为室温~150℃,优选反应温度为室温反应;水解时间为0.5~24小时,优选反应时间为1~7小时。 The 1,3-dihydro-1-oxo-2H-isoindole methyl ester compound (9) obtained in step C) is subjected to alkali hydrolysis and acid neutralization to obtain 1,3-dihydro-1-oxo-2H - Isoindole compound (I), the obtained (I) is salted with the corresponding acid according to the conventional method to obtain the salt of the compound (I); wherein, the alkali used for hydrolysis is alkali metal or alkaline earth metal hydroxide, alkali Metal or alkaline earth metal carbonate, alkali metal or alkaline earth metal bicarbonate, preferred alkali is: lithium hydroxide, sodium carbonate; The molar feed ratio of alkali and compound (9) is 1.0~5.0: 1.0, preferred molar feed ratio is 1.1~2.5:1.0; the hydrolysis reaction temperature is room temperature~150°C, preferably the reaction temperature is room temperature reaction; the hydrolysis time is 0.5~24 hours, preferably the reaction time is 1~7 hours. the
本发明所公开的1,3-二氢-1-氧-2H-异吲哚类化合物(I)可与任何合适的酸通过药学上常规的成盐方法得到其药物学上可接受的盐,所述的酸为盐酸、氢溴酸、硝酸、硫酸、磷酸、甲酸、三氟乙酸、乙酸、丙酸、草酸、苯甲酸、马来酸、富马酸、琥珀酸、酒石酸、扁桃酸、柠檬酸、甲基磺酸、乙基磺酸、樟脑磺酸、苯磺酸或对甲苯磺酸。 The 1,3-dihydro-1-oxo-2H-isoindole compound (I) disclosed in the present invention can be combined with any suitable acid to obtain its pharmaceutically acceptable salt through a pharmaceutically conventional salt-forming method, Described acid is hydrochloric acid, hydrobromic acid, nitric acid, sulfuric acid, phosphoric acid, formic acid, trifluoroacetic acid, acetic acid, propionic acid, oxalic acid, benzoic acid, maleic acid, fumaric acid, succinic acid, tartaric acid, mandelic acid, lemon acid, methanesulfonic acid, ethylsulfonic acid, camphorsulfonic acid, benzenesulfonic acid or p-toluenesulfonic acid. the
本发明的起始原料——2-甲酰基-5-硝基苯甲酸(4)和β-丙氨酸甲酯类衍生物(5)可用本领域常见的技术制得,包括但不局限于以下文献中所公开的方法:1.R.L.Shriner,F.J.Wolf.Org.Syn.Coll.Vo1.3(1955)737;2.V.T.Abaev,A.S.Dmitriev,A.V.Gutnov J.Heterocyclic Chem.,43(2006)1195;3.C.Y.K.Tana,D.F.Weaver.Tetrahedron58(2002)7449。 The starting materials of the present invention—2-formyl-5-nitrobenzoic acid (4) and β-alanine methyl ester derivatives (5) can be prepared by common techniques in the art, including but not limited to Methods disclosed in the following documents: 1.R.L.Shriner, F.J.Wolf.Org.Syn.Coll.Vo1.3 (1955) 737; 2.V.T.Abaev, A.S.Dmitriev, A.V.Gutnov J.Heterocyclic Chem., 43(2006) 1195; 3. C.Y.K. Tana, D.F. Weaver. Tetrahedron 58 (2002) 7449. the
本发明所公开的药物组合物包括治疗有效量的一种或多种1,3-二氢-1-氧-2H-异吲哚类化合物(I)或其药学上可接受的盐,该药物组合物可进一步含有一种或多种药学上可接受的载体或赋形剂。所述“治疗有效量”是指引起研究者或医生所针对的组织、系统或动物的生物或医药反应的药物或药剂的量;所述“组合物”是指通过将一种以上物质或组份混和而成的产品;所述“药学上可接受的载体”是指药学上可接受的物质、组合物或载体,如:液体或固体填充剂、稀释剂、赋形剂、溶剂或包囊物质,它们携带或转运某种化学物质。 The pharmaceutical composition disclosed in the present invention includes one or more 1,3-dihydro-1-oxo-2H-isoindole compounds (I) or their pharmaceutically acceptable salts in a therapeutically effective amount. The composition may further contain one or more pharmaceutically acceptable carriers or excipients. The "therapeutically effective amount" refers to the amount of the drug or agent that causes the biological or medical response of the tissue, system or animal targeted by the researcher or doctor; the "composition" refers to the combination of more than one substance or The mixed product; the "pharmaceutically acceptable carrier" refers to a pharmaceutically acceptable substance, composition or carrier, such as: liquid or solid filler, diluent, excipient, solvent or encapsulation Substances that carry or transport a chemical substance. the
本发明所提出的1,3-二氢-1-氧-2H-异吲哚类化合物(I)或其药学上可接受的盐进行了如下的生物活性筛选: The 1,3-dihydro-1-oxo-2H-isoindole compound (I) or pharmaceutically acceptable salt thereof proposed by the present invention has been screened for the following biological activity:
(1)固相受体筛选 (1) Solid-phase receptor screening
筛选在96孔培养板中进行。整合素αvβ3蛋白用Coating Buffer溶液(Tris-HCl20mM、NaCl150mM、CaCl21mM和NaN30.02%)配成浓度为4μg/mL,加入96孔板中,每孔100μL,4℃包被过夜,阴性对照用2%BSA同法包被,弃去上清液,每孔加入100μL的2%BSA,37℃封闭1小时后用Binding Buffer溶液(Tris-HCl50mM、CaCl22mM、NaN30.02%和BSA1mg/mL(0.1%))清洗。将待筛选化合物用Binding Buffer溶液稀释成不同浓度,取用生物素标记的Bio-Fibronectin溶液(8μg/mL)与不同浓度的待筛选化合物混匀,加入96孔板中,每孔100μL,阴性对照组和模型组加入等量Binding Buffer溶液,37℃孵育3小时,用Binding Buffer溶液清洗后,每孔加入100μL的HRP-链霉亲和素(1∶500),37℃孵育1小时,用Binding Buffer溶液清洗三遍,每孔加入100μL的3,3’,5,5’-四甲基联苯胺原液(TMB),室温孵育10分钟,然后加入1.0M硫酸溶液(每孔100μL)终止反应,在酶标仪上测定各孔OD值(波长为450nm),计算待筛选化合物与整合素αvβ3蛋白的结合抑制率,抑制率(%)=[1-(受试品OD值-阴性组OD)/(模型组OD-阴性组OD))×100。 Screening was performed in 96-well culture plates. Integrin α v β 3 protein was prepared with Coating Buffer solution (Tris-HCl 20mM, NaCl 150mM, CaCl 2 1mM and NaN 3 0.02%) to a concentration of 4 μg/mL, added to a 96-well plate, 100 μL per well, and coated overnight at 4°C , the negative control was coated with 2% BSA in the same way, the supernatant was discarded, and 100 μL of 2% BSA was added to each well, and after blocking for 1 hour at 37 ° C, the Binding Buffer solution (Tris-HCl 50 mM, CaCl 2 2 mM, NaN 3 0.02% and BSA 1 mg/mL (0.1%)). Dilute the compound to be screened with Binding Buffer solution to different concentrations, take biotin-labeled Bio-Fibronectin solution (8 μg/mL) and mix with different concentrations of the compound to be screened, add to 96-well plate, 100 μL per well, negative control Add the same amount of Binding Buffer solution to the model group and the model group, incubate at 37°C for 3 hours, wash with Binding Buffer solution, add 100 μL of HRP-streptavidin (1:500) to each well, incubate at 37°C for 1 hour, and wash with Binding Buffer solution. Buffer solution was washed three times, and 100 μL of 3,3',5,5'-tetramethylbenzidine stock solution (TMB) was added to each well, incubated at room temperature for 10 minutes, and then 1.0 M sulfuric acid solution (100 μL per well) was added to stop the reaction. Measure the OD value of each hole on a microplate reader (wavelength is 450nm), calculate the binding inhibition rate of the compound to be screened and the integrin α v β 3 protein, inhibition rate (%)=[1-(test article OD value-negative Group OD)/(model group OD-negative group OD))×100.
利用上述方法,测定了目标物对整合素αvβ3蛋白的亲和力,结果显示本发明所公开的1,3-二氢-1-氧-2H-异吲哚类化合物(I)或其药学上可接受的盐与整合素αvβ3蛋白的结合抑制IC50在1.8nM~2000nM,而阳性对照药cyclo(RGDfV)的IC50为10.0nM。 Using the above method, the affinity of the target substance to the integrin α v β 3 protein was determined, and the results showed that the 1,3-dihydro-1-oxo-2H-isoindole compound (I) or its pharmaceutical The IC 50 of the binding inhibition IC 50 of acceptable salts and integrin α v β 3 protein is 1.8nM-2000nM, while the IC 50 of the positive control drug cyclo (RGDfV) is 10.0nM.
(2)细胞粘附抑制作用的测定 (2) Determination of cell adhesion inhibition
96孔培养板用2.0μg/mL的Fibronectin包被过夜,对照孔用1%BSA包被,弃去上清液,每孔加入100μL的1%BSA,室温封闭1小时。将待筛选化合物用0.01M的PBS溶液配成不同浓度,加入浓度为5×105/ml的M21细胞悬液中,阳性对照用cyclo(RGDfV),空白对照用0.01M的PBS溶液代替,37℃孵育30分钟,然后将M21细胞悬液加入上述预先封闭的96孔培养板中,每孔100μL,37℃孵育1小时,轻轻吸出孔中的细胞悬液,用预温的PBS洗3次,除去未粘附的细胞,每孔加入100μL的10%戊二醛固定30分钟,用去离子水洗涤至戊二醛彻底洗净,置37℃烘箱彻底干燥,加入0.1%的结晶紫对细胞进行染色,振摇30分钟,用蒸馏水将多余的结晶紫洗净,置37℃烘箱彻底干燥,加入10%的乙酸对细胞吸收的结晶紫进行提取,1小时后在酶标仪上测定OD值(波长为595nm),计算细胞粘附抑制率,即可得出目标物对整合素αvβ3的抑制活性。利用上述方法,测定了部分目标物对M21细胞的粘附抑制活性,其结果见实施例10。 The 96-well culture plate was coated with 2.0 μg/mL Fibronectin overnight, and the control well was coated with 1% BSA, the supernatant was discarded, 100 μL of 1% BSA was added to each well, and blocked for 1 hour at room temperature. The compound to be screened was made into different concentrations with 0.01M PBS solution, and added to the M21 cell suspension with a concentration of 5×10 5 /ml. The positive control was replaced by cyclo (RGDfV), and the blank control was replaced by 0.01M PBS solution. 37 Incubate at ℃ for 30 minutes, then add the M21 cell suspension to the above-mentioned pre-blocked 96-well culture plate, 100 μL per well, incubate at 37℃ for 1 hour, gently suck out the cell suspension in the well, wash 3 times with pre-warmed PBS , remove non-adherent cells, add 100 μL of 10% glutaraldehyde to each well to fix for 30 minutes, wash with deionized water until the glutaraldehyde is thoroughly washed, dry thoroughly in an oven at 37°C, add 0.1% crystal violet to the cells For staining, shake for 30 minutes, wash the excess crystal violet with distilled water, dry it thoroughly in an oven at 37°C, add 10% acetic acid to extract the crystal violet absorbed by the cells, and measure the OD value on a microplate reader after 1 hour (wavelength is 595nm), calculate the cell adhesion inhibition rate, and then the inhibitory activity of the target substance on integrin α v β 3 can be obtained. Using the above method, the adhesion inhibitory activity of some target substances on M21 cells was measured, and the results are shown in Example 10.
(3)目标物细胞毒活性的测定 (3) Determination of target cytotoxic activity
为了排除目标物对M21细胞的抗细胞粘附活性是由细胞毒因素所造成的,为此,我们测试 了目标物对人肝癌细胞HepG2、人肺腺癌细胞A549、小鼠黑色素瘤细胞B16、人黑色素瘤细胞M21及小鼠结肠癌细胞C26的抑制活性。将肿瘤细胞分别消化计数,并接种于96孔板(5000个细胞/孔),24小时后以pH7.4的PBS液洗涤2遍,加入含不同浓度的药物,培养48小时,洗涤后,用0.4%磺酰罗丹明(SRB)溶液室温下染色15分钟,1%冰醋酸洗3遍,空气中将染色的细胞干燥,以10mM的Tris-HCl溶解,并用Dynatech MR7000仪测540nm波长的吸光度值,计算药物对肿瘤细胞生长的抑制率。 In order to rule out that the anti-cell adhesion activity of the target substance on M21 cells is caused by cytotoxic factors, we tested the target substance against human liver cancer cell HepG2, human lung adenocarcinoma cell A549, mouse melanoma cell B16, Inhibitory activity of human melanoma cell line M21 and mouse colon cancer cell line C26. Tumor cells were digested and counted separately, and seeded in 96-well plate (5000 cells/well). After 24 hours, they were washed twice with PBS solution of pH 7.4. Drugs with different concentrations were added and cultured for 48 hours. After washing, they were washed with Stain with 0.4% sulforhodamine (SRB) solution at room temperature for 15 minutes, wash 3 times with 1% glacial acetic acid, dry the stained cells in air, dissolve with 10mM Tris-HCl, and measure the absorbance value at 540nm wavelength with Dynatech MR7000 instrument , to calculate the inhibitory rate of the drug on tumor cell growth. the
试验结果表明,本发明所公开的1,3-二氢-1-氧-2H-异吲哚类化合物(I)或其药学上可接受的盐对这些肿瘤细胞抑制的IC50均大于80μg/mL,说明此类化合物的细胞毒较小。 The test results show that the 1,3-dihydro-1-oxo-2H-isoindole compounds (I) disclosed in the present invention or their pharmaceutically acceptable salts have an IC 50 of inhibition of these tumor cells greater than 80 μg/ mL, indicating that the cytotoxicity of such compounds is small.
本发明所公开的1,3-二氢-1-氧-2H-异吲哚类化合物(I)或其药学上可接受的盐具有显著的整合素αvβ3抑制活性,可作为整合素αvβ3抑制剂用于预防或/和治疗各种因新生血管过度增生所引起的症状或疾病药物中的用途,包括但不限于肿瘤转移、肿瘤生长、实体瘤生长、血管新生、视网膜病变、黄斑变性、骨质疏松、关节炎、平滑肌细胞移行和动脉粥样硬化等。 The 1,3-dihydro-1-oxo-2H-isoindole compound (I) or a pharmaceutically acceptable salt thereof disclosed in the present invention has significant integrin α v β 3 inhibitory activity and can be used as an integrin Use of α v β 3 inhibitors in preventing or/and treating various symptoms or diseases caused by excessive neovascular proliferation, including but not limited to tumor metastasis, tumor growth, solid tumor growth, angiogenesis, retinopathy , macular degeneration, osteoporosis, arthritis, smooth muscle cell migration and atherosclerosis, etc.
具体实施方式 Detailed ways
通过下面的实施例可对本发明进行进一步的描述,然而,本发明的范围并不限于下述实施例。本领域的专业人员能够理解,在不背离本发明的精神和范围的前提下,可以对本发明进行各种变化和修饰。 The present invention can be further described by the following examples, however, the scope of the present invention is not limited to the following examples. Those skilled in the art can understand that various changes and modifications can be made in the present invention without departing from the spirit and scope of the present invention. the
实施例1 Example 1
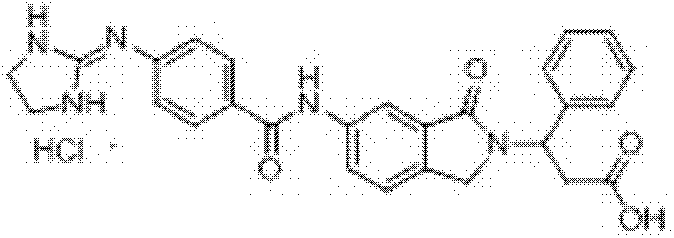
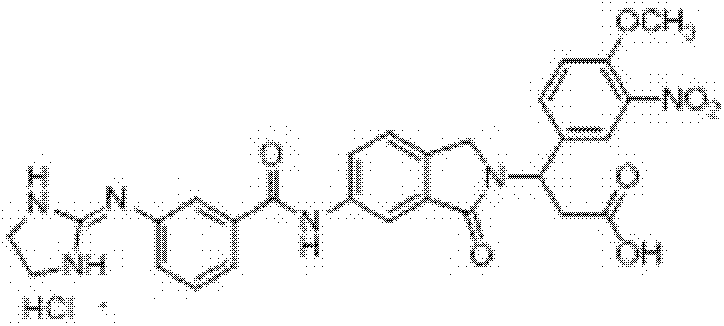
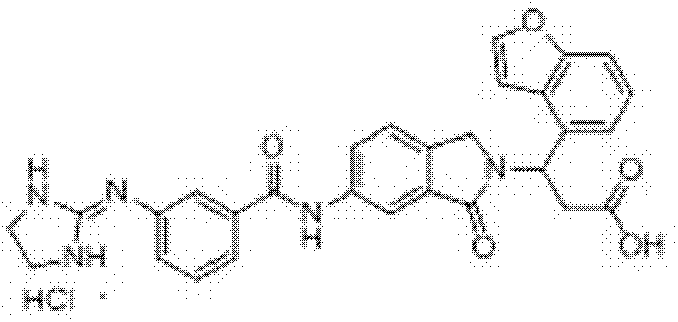
3-(6-(3-胍基苯甲酰胺基)-1-氧异吲哚-2-基)-3-苯基丙酸盐酸盐(I-1)和三氟乙酸盐的制备 Preparation of 3-(6-(3-guanidinobenzamido)-1-oxoisoindol-2-yl)-3-phenylpropion hydrochloride (I-1) and trifluoroacetate
步骤(a):在反应瓶中加入2-甲酰基-5-硝基苯甲酸(4)10.0mmol、β-苯基-β-丙氨酸甲酯12.0mmol和甲醇80ml,室温搅拌反应2.0h,置冰水浴冷却下缓慢加入NaBH410.0mmol,保温搅拌反应(反应进程用TLC监测),反应结束后,减压蒸除溶剂,残余物中加入醋酸20ml,80℃保温反应40min,向反应瓶中加入去离子水100ml,过滤析出的固体,滤饼用碳酸钠水溶液洗涤,干燥,即得3-(6-硝基-1-氧异吲哚-2-基)-3-苯基丙酸甲酯,收率90.0%,ESI-MS(m/z,+Q):341.0[M+H]+; Step (a): Add 10.0mmol of 2-formyl-5-nitrobenzoic acid (4), 12.0mmol of β-phenyl-β-alanine methyl ester and 80ml of methanol into the reaction flask, and stir at room temperature for 2.0h , slowly add NaBH 4 10.0mmol under cooling in an ice-water bath, keep stirring and react (the reaction process is monitored by TLC), after the reaction is completed, evaporate the solvent under reduced pressure, add 20ml of acetic acid to the residue, keep the reaction at 80°C for 40min, and pour into the reaction flask Add 100ml of deionized water to the mixture, filter the precipitated solid, wash the filter cake with aqueous sodium carbonate solution, and dry to obtain 3-(6-nitro-1-oxoisoindol-2-yl)-3-phenylpropionic acid Methyl ester, yield 90.0%, ESI-MS (m/z, +Q): 341.0[M+H] + ;
步骤(b):在反应瓶中加入3-(6-硝基-1-氧异吲哚-2-基)-3-苯基丙酸甲酯10.0mmol和甲醇200ml,搅拌均匀后,加入10%Pd/C0.5g,用氢气置换反应瓶中空气后,于常温常压下通氢还原反应12h(反应进程用TLC监测,石油醚/乙酸乙酯=1∶2v/v),反应结束后,过滤,滤 液减压蒸除溶剂,残余物经重结晶或柱层析纯化,即得3-(6-氨基-1-氧异吲哚-2-基)-3-苯基丙酸甲酯,收率99.0%,ESI-MS(m/z,+Q):311.1[M+H]+; Step (b): Add 10.0mmol of 3-(6-nitro-1-oxoisoindol-2-yl)-3-phenylpropionic acid methyl ester and 200ml of methanol into the reaction flask, stir well, then add 10 %Pd/C0.5g, after replacing the air in the reaction bottle with hydrogen, carry out hydrogen reduction reaction at normal temperature and pressure for 12h (reaction progress is monitored by TLC, petroleum ether/ethyl acetate=1:2v/v), after the reaction , filtered, the filtrate was evaporated to remove the solvent under reduced pressure, and the residue was purified by recrystallization or column chromatography to obtain 3-(6-amino-1-oxoisoindol-2-yl)-3-phenylpropionic acid methyl ester , yield 99.0%, ESI-MS (m/z, +Q): 311.1[M+H] + ;
步骤(c):在反应瓶中加入3-(6-氨基-1-氧异吲哚-2-基)-3-苯基丙酸甲酯1.0mmol、间胍基苯甲酸盐酸盐1.2mmol和吡啶25ml,室温搅拌至固体全溶后,加入1-乙基-3-(3-二甲胺丙基)碳二亚胺盐酸盐1.5mmol,室温搅拌24~48h(反应进程用TLC监测,氯仿/甲醇=5∶1v/v),反应结束后,过滤,滤液减压蒸除溶剂,残余物经重结晶或柱层析纯化,即得3-(6-(3-胍基苯甲酰胺基)-1-氧异吲哚-2-基)-3-苯基丙酸甲酯盐酸盐,收率80.5%,ESI-MS(m/z,+Q):472.1[M-Cl]+; Step (c): Add 1.0 mmol of 3-(6-amino-1-oxoisoindol-2-yl)-3-phenylpropionic acid methyl ester, 1.2 m-guanidinobenzoic acid hydrochloride in the reaction flask mmol and pyridine 25ml, stirred at room temperature until the solid was completely dissolved, then added 1.5mmol of 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride, stirred at room temperature for 24-48h (the reaction process was determined by TLC monitoring, chloroform/methanol=5:1v/v), after the reaction finishes, filter, and the filtrate decompresses to remove solvent, and the residue is purified by recrystallization or column chromatography to obtain 3-(6-(3-guanidinobenzene Formamido)-1-oxoisoindol-2-yl)-3-phenylpropionic acid methyl ester hydrochloride, yield 80.5%, ESI-MS (m/z, +Q): 472.1 [M- Cl] + ;
步骤(d):在反应瓶中加入3-(6-(3-胍基苯甲酰胺基)-1-氧异吲哚-2-基)-3-苯基丙酸甲酯盐酸盐0.5mmol、甲醇10ml、四氢呋喃5ml和去离子水5ml,室温搅拌均匀后加入氢氧化锂2.0mmol、室温搅拌反应1.5h(反应进程用TLC监测,氯仿/甲醇/HOAc=3/1/0.5v/v),反应结束后,用10%盐酸水溶液调节溶液pH至1-2,减压蒸除溶剂,残余物经重结晶或柱层析纯化,即得3-(6-(3-胍基苯甲酰胺基)-1-氧异吲哚-2-基)-3-苯基丙酸盐酸盐,收率65.0%,1H NMR(400MHz,DMSO-d6)δ:12.45(brs,1H,COOH)10.64(s,1H,CONH),10.52(brs,1H,HCl),8.21(d,1H,J=1.6Hz,Ar-H),7.95(dd,1H,J1=1.6Hz,J2=8.4Hz,Ar-H),7.89(d,1H,J=8.0Hz,Ar-H),7.86(s,1H,Ar-H),7.69(brs,4H,NH2C(NH)NH),7.59(d,1H,J=8.0Hz,Ar-H),7.59(t,1H,J=8.0Hz,Ar-H),7.53(d,1H,J=8.0Hz,Ar-H),7.43(d,2H,J=8.0Hz,Ar-H),7.41-7.27(m,5H,Ar-H),5.76(t,1H,J=8.0Hz,CH),4.51(d,1H,J=17.6Hz,ArCH2),4.14(d,1H,J=17.6Hz,ArCH2),3.18-3.05(m,2H,CH2COOH);ESI-MS(m/z):458.10[M-Cl]+; Step (d): add 3-(6-(3-guanidinobenzamido)-1-oxoisoindol-2-yl)-3-phenylpropanoic acid methyl ester hydrochloride 0.5 mmol, 10ml of methanol, 5ml of tetrahydrofuran and 5ml of deionized water, stir well at room temperature, add lithium hydroxide 2.0mmol, stir at room temperature for 1.5h (the reaction process is monitored by TLC, chloroform/methanol/HOAc=3/1/0.5v/v ), after the reaction, adjust the pH of the solution to 1-2 with 10% hydrochloric acid aqueous solution, evaporate the solvent under reduced pressure, and the residue is purified by recrystallization or column chromatography to obtain 3-(6-(3-guanidinobenzyl Amino)-1-oxoisoindol-2-yl)-3-phenylpropionium hydrochloride, yield 65.0%, 1 H NMR (400MHz, DMSO-d 6 )δ: 12.45 (brs, 1H, COOH) 10.64 (s, 1H, CONH), 10.52 (brs, 1H, HCl), 8.21 (d, 1H, J = 1.6Hz, Ar-H), 7.95 (dd, 1H, J1 = 1.6Hz, J2 =8.4Hz, Ar-H), 7.89(d, 1H, J=8.0Hz, Ar-H), 7.86(s, 1H, Ar-H), 7.69(brs, 4H, NH 2 C(NH)NH) , 7.59(d, 1H, J=8.0Hz, Ar-H), 7.59(t, 1H, J=8.0Hz, Ar-H), 7.53(d, 1H, J=8.0Hz, Ar-H), 7.43 (d, 2H, J=8.0Hz, Ar-H), 7.41-7.27(m, 5H, Ar-H), 5.76(t, 1H, J=8.0Hz, CH), 4.51(d, 1H, J= 17.6Hz, ArCH 2 ), 4.14 (d, 1H, J=17.6Hz, ArCH 2 ), 3.18-3.05 (m, 2H, CH 2 COOH); ESI-MS (m/z): 458.10 [M-Cl] + ;
在上述步骤(d)中,水解结束后用三氟乙酸调节溶液pH至1-2,减压蒸除溶剂,残余物经重结晶或柱层析纯化,即得3-(6-(3-胍基苯甲酰胺基)-1-氧异吲哚-2-基)-3-苯基丙酸三氟乙酸盐,收率60.0%。 In the above step (d), after the hydrolysis is completed, the pH of the solution is adjusted to 1-2 with trifluoroacetic acid, the solvent is evaporated under reduced pressure, and the residue is purified by recrystallization or column chromatography to obtain 3-(6-(3- Guanidinobenzamido)-1-oxoisoindol-2-yl)-3-phenylpropionic acid trifluoroacetate, yield 60.0%. the
实施例2 Example 2
3-(6-(4-胍基苯甲酰胺基)-1-氧异吲哚-2-基)-3-苯基丙酸盐酸盐(I-2)的制备 Preparation of 3-(6-(4-guanidinobenzamido)-1-oxoisoindol-2-yl)-3-phenylpropion hydrochloride (I-2)
操作过程同实施例1,只是将步骤(c)中的间胍基苯甲酸盐酸盐用对胍基苯甲酸盐酸盐替代,缩合剂1-乙基-3-(3-二甲胺丙基)碳二亚胺盐酸盐用DCC替代,得3-(6-(4-胍基苯甲酰胺基)-1-氧异吲哚-2-基)-3-苯基丙酸盐酸盐,收率74.0%,1H NMR(400MHz,DMSO-d6)δ:12.42(brs,1H,COOH),11.01(brs,1H,HCl),10.64(s,1H,CONH),8.26(d,1H,J=1.6Hz,Ar-H), 8.11(d,2H,J=8.4Hz,Ar-H),7.98(dd,1H,J1=1.6Hz,J2=8.0Hz,Ar-H),7.97(brs,4H,NH2C(NH)NH),7.52(d,1H,J=8.0Hz,Ar-H),7.33(d,2H,J=8.4Hz,Ar-H),7.40-7.27(m,5H,Ar-H),5.77(t,1H,J=8.0Hz,CH),4.52(d,1H,J=17.6Hz,ArCH2),4.14(d,1H,J=17.6Hz,ArCH2),3.13-3.02(m,2H,CH2COOH);ESI-MS(m/z):458.15[M-Cl]+。 The operation process is the same as in Example 1, except that the m-guanidinobenzoic acid hydrochloride in the step (c) is replaced with p-guanidinobenzoic acid hydrochloride, and the condensing agent 1-ethyl-3-(3-dimethyl Aminopropyl) carbodiimide hydrochloride is replaced by DCC to obtain 3-(6-(4-guanidinobenzamido)-1-oxoisoindol-2-yl)-3-phenylpropionic acid Hydrochloride, yield 74.0%, 1 H NMR (400MHz, DMSO-d 6 ) δ: 12.42 (brs, 1H, COOH), 11.01 (brs, 1H, HCl), 10.64 (s, 1H, CONH), 8.26 (d, 1H, J = 1.6Hz, Ar-H), 8.11 (d, 2H, J = 8.4Hz, Ar-H), 7.98 (dd, 1H, J 1 = 1.6Hz, J 2 = 8.0Hz, Ar -H), 7.97 (brs, 4H, NH 2 C(NH)NH), 7.52 (d, 1H, J=8.0Hz, Ar-H), 7.33 (d, 2H, J=8.4Hz, Ar-H) , 7.40-7.27(m, 5H, Ar-H), 5.77(t, 1H, J=8.0Hz, CH), 4.52(d, 1H, J=17.6Hz, ArCH 2 ), 4.14(d, 1H, J =17.6 Hz, ArCH 2 ), 3.13-3.02 (m, 2H, CH 2 COOH); ESI-MS (m/z): 458.15 [M-Cl] + .
实施例3 Example 3
3-[6-[3-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]-3-苯基丙酸盐酸盐(I-3)的制备 3-[6-[3-[(4,5-Dihydro-1H-imidazol-2-yl)amino]benzamido]-1-oxoisoindol-2-yl]-3-phenylpropane Preparation of hydrochloride (I-3)
操作过程同实施例1,只是将步骤(c)中的间胍基苯甲酸盐酸盐用3-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酸盐酸盐替代,缩合剂1-乙基-3-(3-二甲胺丙基)碳二亚胺盐酸盐用DCC替代,得3-[6-[3-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]-3-苯基丙酸盐酸盐,收率85.0%,1H NMR(400MHz,DMSO-d6)δ:12.44(brs,1H,COOH),10.89(s,1H,HCl),10.78(s,1H,CONH),8.58(s,2H,NHCNH),8.25(d,1H,J=1.2Hz,Ar-H),7.95(dd,1H,J1=1.2Hz,J2=8.4Hz,Ar-H),7.88-7.86(m,2H,Ar-H),7.59(t,1H,J=8.0Hz,Ar-H),7.53(d,1H,J=8.0Hz,Ar-H),7.45(d,2H,J=8.0Hz,Ar-H),7.41-7.28(m,5H,Ar-H),5.76(t,1H,J=8.0Hz,CH),4.50(d,1H,J=17.6Hz,ArCH2),4.12(d,1H,J=17.6Hz,ArCH2),3.69(s,4H,CH2CH2),3.23-3.09(m,2H,CH2COOH);ESI-MS(m/z):484.20[M-Cl]+。 The operation process is the same as in Example 1, except that the m-guanidinobenzoic acid hydrochloride in the step (c) is 3-[(4,5-dihydro-1H-imidazol-2-yl)amino]benzoic acid salt Salt replacement, the condensing agent 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide hydrochloride is replaced by DCC, and 3-[6-[3-[(4,5-dihydro -1H-imidazol-2-yl)amino]benzamido]-1-oxoisoindol-2-yl]-3-phenylpropion hydrochloride, yield 85.0%, 1 H NMR (400MHz, DMSO-d 6 ) δ: 12.44 (brs, 1H, COOH), 10.89 (s, 1H, HCl), 10.78 (s, 1H, CONH), 8.58 (s, 2H, NHCNH), 8.25 (d, 1H, J =1.2Hz, Ar-H), 7.95(dd, 1H, J 1 =1.2Hz, J 2 =8.4Hz, Ar-H), 7.88-7.86(m, 2H, Ar-H), 7.59(t, 1H , J=8.0Hz, Ar-H), 7.53(d, 1H, J=8.0Hz, Ar-H), 7.45(d, 2H, J=8.0Hz, Ar-H), 7.41-7.28(m, 5H , Ar-H), 5.76 (t, 1H, J=8.0Hz, CH), 4.50 (d, 1H, J=17.6Hz, ArCH 2 ), 4.12 (d, 1H, J=17.6Hz, ArCH 2 ), 3.69 (s, 4H, CH2CH2 ), 3.23-3.09 (m, 2H, CH2COOH ); ESI-MS (m/z ) : 484.20 [M-Cl] + .
实施例4 Example 4
3-[6-[4-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]-3-苯基丙酸盐酸盐(I-4)的制备 3-[6-[4-[(4,5-Dihydro-1H-imidazol-2-yl)amino]benzamido]-1-oxoisoindol-2-yl]-3-phenylpropane Preparation of hydrochloride (I-4)
操作过程同实施例1,只是将步骤(c)中的间胍基苯甲酸盐酸盐用4-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酸盐酸盐替代,缩合剂1-乙基-3-(3-二甲胺丙基)碳二亚胺盐酸盐用DCC替代,得3-[6-[4-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]-3-苯基丙酸盐酸盐,收率78.0%,1H NMR(400MHz,DMSO-d6)δ:12.10(brs,1H,COOH),11.00(s,1H,HCl),10.51(s,1H,CONH),8.75(brs,2H,NHCNH),8.22(d,1H,J=1.6Hz,Ar-H),8.07(d,2H,J=8.8Hz,Ar-H),7.94(dd,1H,J1=1.2Hz,J2=8.4Hz,Ar-H),7.54(d,1H,J=8.4Hz,Ar-H),7.33(d,2H,J=8.8Hz,Ar-H),7.40-7.28(m,5H,Ar-H),5.76(t,1H,J=8.0Hz,CH),4.51(d,1H,J=17.6Hz,ArCH2),4.13(d, 1H,J=17.6Hz,ArCH2),3.69(s,4H,CH2CH2),3.20-3.07(m,2H,CH2COOH);ESI-MS(m/z):484.20[M-Cl]+。 The operation process is the same as in Example 1, except that the m-guanidinobenzoic acid hydrochloride in the step (c) is replaced with 4-[(4,5-dihydro-1H-imidazol-2-yl)amino]benzoic acid salt Salt replacement, the condensing agent 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide hydrochloride is replaced by DCC, and 3-[6-[4-[(4,5-dihydro -1H-imidazol-2-yl)amino]benzamido]-1-oxoisoindol-2-yl]-3-phenylpropion hydrochloride, yield 78.0%, 1 H NMR (400MHz, DMSO-d 6 ) δ: 12.10 (brs, 1H, COOH), 11.00 (s, 1H, HCl), 10.51 (s, 1H, CONH), 8.75 (brs, 2H, NHCNH), 8.22 (d, 1H, J = 1.6Hz, Ar-H), 8.07 (d, 2H, J = 8.8Hz, Ar-H), 7.94 (dd, 1H, J 1 = 1.2Hz, J 2 = 8.4Hz, Ar-H), 7.54 ( d, 1H, J=8.4Hz, Ar-H), 7.33(d, 2H, J=8.8Hz, Ar-H), 7.40-7.28(m, 5H, Ar-H), 5.76(t, 1H, J =8.0Hz, CH), 4.51 (d, 1H, J=17.6Hz, ArCH 2 ), 4.13 (d, 1H, J=17.6Hz, ArCH 2 ), 3.69 (s, 4H, CH 2 CH 2 ), 3.20 -3.07 (m, 2H, CH2COOH ); ESI-MS (m/z): 484.20 [M-Cl] + .
实施例5 Example 5
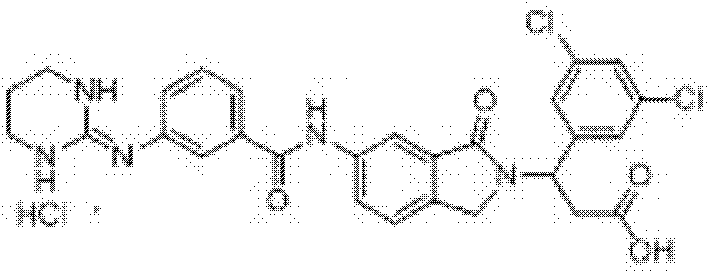
3-(3,5-二氯苯基)-3-[6-(3-胍基苯甲酰胺基)-1-氧异吲哚-2-基]丙酸盐酸盐(I-5)的制备 3-(3,5-Dichlorophenyl)-3-[6-(3-guanidinobenzamido)-1-oxoisoindol-2-yl]propion hydrochloride (I-5) preparation of
步骤(a):操作过程同实施例1步骤(a),只是将β-苯基-β-丙氨酸甲酯用β-(3,5-二氯苯基)-β-丙氨酸甲酯替代,得3-(3,5-二氯苯基)-3-(6-硝基-1-氧异吲哚-2-基)丙酸甲酯,收率82.0%,ESI-MS(m/z,+Q):409.0[M+H]+; Step (a): The operation process is the same as step (a) of Example 1, except that β-phenyl-β-alanine methyl ester is replaced with β-(3,5-dichlorophenyl)-β-alanine methyl ester Ester substitution, 3-(3,5-dichlorophenyl)-3-(6-nitro-1-oxoisoindol-2-yl) methyl propionate, yield 82.0%, ESI-MS ( m/z,+Q): 409.0[M+H] + ;
步骤(b):将3-(3,5-二氯苯基)-3-(6-硝基-1-氧异吲哚-2-基)丙酸甲酯10.0mmol溶于四氢呋喃150ml中,加入乙酸25ml和锌粉160.0mmol,加热回流4h(反应进程用TLC监测,石油醚/乙酸乙酯=1∶2v/v),反应结束后,冷却至室温,用饱和碳酸氢钠水溶液调节溶液pH至碱性,过滤,滤液用3×70ml CH2Cl2萃取,合并有机层并用饱和食盐水洗涤,有机层经无水NaS04干燥,减压蒸除溶剂,残余物经重结晶或柱层析纯化,即得3-(3,5-二氯苯基)-3-(6-氨基-1-氧异吲哚-2-基)丙酸甲酯,收率96.0%,ESI-MS(m/z,+Q):365.1[M+H]+; Step (b): Dissolve 10.0 mmol of methyl 3-(3,5-dichlorophenyl)-3-(6-nitro-1-oxoisoindol-2-yl)propionate in 150 ml of tetrahydrofuran, Add 25ml of acetic acid and 160.0mmol of zinc powder, heat to reflux for 4h (the reaction process is monitored by TLC, petroleum ether/ethyl acetate=1:2v/v), after the reaction is completed, cool to room temperature, and adjust the pH of the solution with saturated aqueous sodium bicarbonate to alkaline, filter, the filtrate was extracted with 3×70ml CH 2 Cl 2 , the organic layers were combined and washed with saturated brine, the organic layer was dried over anhydrous NaSO 4 , the solvent was evaporated under reduced pressure, and the residue was subjected to recrystallization or column chromatography Purify to obtain 3-(3,5-dichlorophenyl)-3-(6-amino-1-oxoisoindol-2-yl)propionic acid methyl ester with a yield of 96.0%, ESI-MS (m /z,+Q): 365.1[M+H] + ;
步骤(c):操作过程同实施例1步骤(c),只是将3-(6-氨基-1-氧异吲哚-2-基)-3-苯基丙酸甲酯用3-(3,5-二氯苯基)-3-(6-氨基-1-氧异吲哚-2-基)丙酸甲酯替代,得3-(3,5-二氯苯基)-3-[6-(3-胍基苯甲酰胺基)-1-氧异吲哚-2-基]丙酸甲酯盐酸盐,收率80.0%,ESI-MS(m/z,+Q):540.0[M-Cl]+; Step (c): The operation process is the same as that of Example 1 step (c), except that 3-(6-amino-1-oxoisoindol-2-yl)-3-phenylpropionic acid methyl ester is used with 3-(3 , 5-dichlorophenyl)-3-(6-amino-1-oxoisoindol-2-yl) methyl propionate instead, 3-(3,5-dichlorophenyl)-3-[ 6-(3-guanidinobenzamido)-1-oxoisoindol-2-yl]propionic acid methyl ester hydrochloride, yield 80.0%, ESI-MS (m/z, +Q): 540.0 [M-Cl] + ;
步骤(d):操作过程同实施例1步骤(d),只是将3-(6-(3-胍基苯甲酰胺基)-1-氧异吲哚-2-基)-3-苯基丙酸甲酯盐酸盐用3-(3,5-二氯苯基)-3-[6-(3-胍基苯甲酰胺基)-1-氧异吲哚-2-基]丙酸甲酯盐酸盐替代,得3-(3,5-二氯苯基)-3-[6-(3-胍基苯甲酰胺基)-1-氧异吲哚-2-基]丙酸盐酸盐,收率93.0%,1H NMR(400MHz,DMSO-d6)δ:12.53(brs,1H,COOH),10.62(s,1H,HCl),10.12(s,1H,CONH),8.23(d,1H,J=1.2Hz,Ar-H),7.96(dd,1H,J1=2.0Hz,J2=8.4Hz,Ar-H),7.91(d,1H,J=8.0Hz,Ar-H),7.87(s,1H,Ar-H),7.63(brs,4H,NH2C(NH)NH),7.59(d,1H,J=8.0Hz,Ar-H),7.57(d,1H,J=2.0Hz,Ar-H),7.56(d,1H,J=8.4Hz,Ar-H),7.48(d,2H,J=1.6Hz,Ar-H),7.45(d,1H,J=8.0Hz,Ar-H),5.65(t,1H,J=7.6Hz,CH),4.52(d,1H,J=17.6Hz,ArCH2),4.30(d,1H,J=17.6Hz,ArCH2),3.27-3.18(m,2H,CH2COOH);ESI-MS(m/z):526.15[M-Cl]+。 Step (d): The operation process is the same as step (d) of Example 1, except that 3-(6-(3-guanidinobenzamido)-1-oxoisoindol-2-yl)-3-phenyl Methyl propionate hydrochloride with 3-(3,5-dichlorophenyl)-3-[6-(3-guanidinobenzamido)-1-oxoisoindol-2-yl]propionic acid Substitution of methyl ester hydrochloride to obtain 3-(3,5-dichlorophenyl)-3-[6-(3-guanidinobenzamido)-1-oxoisoindol-2-yl]propionic acid Hydrochloride, yield 93.0%, 1 H NMR (400MHz, DMSO-d 6 ) δ: 12.53 (brs, 1H, COOH), 10.62 (s, 1H, HCl), 10.12 (s, 1H, CONH), 8.23 (d, 1H, J = 1.2Hz, Ar-H), 7.96 (dd, 1H, J 1 = 2.0Hz, J 2 = 8.4Hz, Ar-H), 7.91 (d, 1H, J = 8.0Hz, Ar -H), 7.87(s, 1H, Ar-H), 7.63(brs, 4H, NH 2 C(NH)NH), 7.59(d, 1H, J=8.0Hz, Ar-H), 7.57(d, 1H, J=2.0Hz, Ar-H), 7.56(d, 1H, J=8.4Hz, Ar-H), 7.48(d, 2H, J=1.6Hz, Ar-H), 7.45(d, 1H, J=8.0Hz, Ar-H), 5.65(t, 1H, J=7.6Hz, CH), 4.52(d, 1H, J=17.6Hz, ArCH2 ), 4.30(d, 1H, J=17.6Hz, ArCH 2 ), 3.27-3.18 (m, 2H, CH 2 COOH); ESI-MS (m/z): 526.15 [M-Cl] + .
实施例6 Example 6
3-(3,5-二氯苯基)-3-[6-(4-胍基苯甲酰胺基)-1-氧异吲哚-2-基]丙酸盐酸盐(I-6)的制备 3-(3,5-Dichlorophenyl)-3-[6-(4-guanidinobenzamido)-1-oxoisoindol-2-yl]propion hydrochloride (I-6) preparation of
操作过程同实施例5,只是将步骤(c)中的间胍基苯甲酸盐酸盐用对胍基苯甲酸盐酸盐替代,缩合剂1-乙基-3-(3-二甲胺丙基)碳二亚胺盐酸盐用DCC替代,得3-(3,5-二氯苯基)-3-[6-(4-胍基苯甲酰胺基)-1-氧异吲哚-2-基]丙酸盐酸盐,收率97.0%,1H NMR(400MHz,DMSO-d6)δ:12.51(brs,1H,COOH),10.53(s,1H,HCl),10.32(brs,1H,CONH),8.24(d,1H,J=1.6Hz,Ar-H),8.08(d,2H,J=8.8Hz,Ar-H),7.96(dd,1H,J1=2.0Hz,J2=8.4Hz,Ar-H),7.74(brs,4H,NH2C(NH)NH),7.57(d,1H,J=2.0Hz,Ar-H),7.55(d,1H,J=8.4Hz,Ar-H),7.48(d,2H,J=1.6Hz,Ar-H),7.38(d,2H,J=8.8Hz,Ar-H),5.65(t,1H,J=7.6Hz,CH),4.52(d,1H,J=17.6Hz,ArCH2),4.30(d,1H,J=17.6Hz,ArCH2),3.29-3.27(m,2H,CH2COOH);ESI-MS(m/z):526.10[M-Cl]+。 The operation process is the same as in Example 5, except that the m-guanidinobenzoic acid hydrochloride in the step (c) is replaced with p-guanidinobenzoic acid hydrochloride, and the condensing agent 1-ethyl-3-(3-dimethyl Aminopropyl) carbodiimide hydrochloride is replaced by DCC to obtain 3-(3,5-dichlorophenyl)-3-[6-(4-guanidinobenzamido)-1-oxoisoind Indol-2-yl]propion hydrochloride, yield 97.0%, 1 H NMR (400MHz, DMSO-d 6 ) δ: 12.51 (brs, 1H, COOH), 10.53 (s, 1H, HCl), 10.32 ( brs, 1H, CONH), 8.24 (d, 1H, J = 1.6Hz, Ar-H), 8.08 (d, 2H, J = 8.8Hz, Ar-H), 7.96 (dd, 1H, J 1 = 2.0Hz , J 2 =8.4Hz, Ar-H), 7.74 (brs, 4H, NH 2 C(NH)NH), 7.57 (d, 1H, J=2.0Hz, Ar-H), 7.55 (d, 1H, J =8.4Hz, Ar-H), 7.48(d, 2H, J=1.6Hz, Ar-H), 7.38(d, 2H, J=8.8Hz, Ar-H), 5.65(t, 1H, J=7.6 Hz, CH), 4.52 (d, 1H, J=17.6Hz, ArCH 2 ), 4.30 (d, 1H, J=17.6Hz, ArCH 2 ), 3.29-3.27 (m, 2H, CH 2 COOH); ESI- MS (m/z): 526.10 [M-Cl] + .
实施例7 Example 7
3-(3,5-二氯苯基)-3-[6-[3-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]丙酸盐酸盐(I-7)的制备 3-(3,5-dichlorophenyl)-3-[6-[3-[(4,5-dihydro-1H-imidazol-2-yl)amino]benzamido]-1-oxoiso Preparation of indol-2-yl] propionate hydrochloride (I-7)
操作过程同实施例5,只是将步骤(c)中的间胍基苯甲酸盐酸盐用3-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酸盐酸盐替代,缩合剂1-乙基-3-(3-二甲胺丙基)碳二亚胺盐酸盐用DCC替代,得3-(3,5-二氯苯基)-3-[6-[3-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]丙酸盐酸盐,收率98.0%,1H NMR(400MHz,DMSO-d6)δ:12.53(brs,1H,COOH),10.90(s,1H,HCl)10.81(s,1H,CONH),8.60(brs,2H,NHCNH),8.26(d,1H,J=2.0Hz,Ar-H),8.01(dd,1H,J1=2.0Hz,J2=8.4Hz,Ar-H),7.88(d,1H,J=8.0Hz,Ar-H),7.87(s,1H,Ar-H),7.61(d,1H,J=8.0Hz,Ar-H),7.57(d,1H,J=8.4Hz,Ar-H),7.56(d,1H,J=2.0Hz,Ar-H),7.48(d,2H,J=2.0Hz,Ar-H),7.45(d,1H,J=8.0Hz,Ar-H),5.65(t,1H,J=7.6Hz,CH),4.52(d,1H,J=17.6Hz,ArCH2),4.30(d,1H,J=17.6Hz,ArCH2),3.70(s,4H,CH2CH2),3.30-3.18(m,2H,CH2COOH);ESI-MS(m/z):552.20[M-Cl]+。 The operating process is the same as in Example 5, except that the m-guanidinobenzoic acid hydrochloride in the step (c) is replaced with 3-[(4,5-dihydro-1H-imidazol-2-yl)amino]benzoic acid salt Salt replacement, condensing agent 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide hydrochloride is replaced by DCC, and 3-(3,5-dichlorophenyl)-3-[ 6-[3-[(4,5-Dihydro-1H-imidazol-2-yl)amino]benzamido]-1-oxoisoindol-2-yl]propion hydrochloride, yield 98.0 %, 1 H NMR (400MHz, DMSO-d 6 ) δ: 12.53 (brs, 1H, COOH), 10.90 (s, 1H, HCl), 10.81 (s, 1H, CONH), 8.60 (brs, 2H, NHCNH), 8.26(d, 1H, J=2.0Hz, Ar-H), 8.01(dd, 1H, J1 =2.0Hz, J2 =8.4Hz, Ar-H), 7.88(d, 1H, J=8.0Hz, Ar-H), 7.87(s, 1H, Ar-H), 7.61(d, 1H, J=8.0Hz, Ar-H), 7.57(d, 1H, J=8.4Hz, Ar-H), 7.56( d, 1H, J=2.0Hz, Ar-H), 7.48(d, 2H, J=2.0Hz, Ar-H), 7.45(d, 1H, J=8.0Hz, Ar-H), 5.65(t, 1H, J=7.6Hz, CH), 4.52(d, 1H, J=17.6Hz, ArCH2 ), 4.30(d, 1H, J=17.6Hz, ArCH2 ) , 3.70(s, 4H, CH2CH2 ), 3.30-3.18 (m, 2H, CH 2 COOH); ESI-MS (m/z): 552.20 [M-Cl] + .
实施例8 Example 8
3-(3,5-二氯苯基)-3-[6-[4-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]丙酸盐酸盐(I-8)的制备 3-(3,5-dichlorophenyl)-3-[6-[4-[(4,5-dihydro-1H-imidazol-2-yl)amino]benzamido]-1-oxoiso Preparation of indol-2-yl] propionate hydrochloride (I-8)
操作过程同实施例5,只是将步骤(c)中的间胍基苯甲酸盐酸盐用4-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酸盐酸盐替代,缩合剂1-乙基-3-(3-二甲胺丙基)碳二亚胺盐酸盐用DCC替代,得3-(3,5-二氯苯基)-3-[6-[4-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]丙酸盐酸盐,收率90.0%,1H NMR(400MHz,DMSO-d6)δ:12.49(brs,1H,COOH),11.86(brs,1H,HCl),10.58(s,1H,CONH),8.85(brs,2H,NHCNH),8.25(d,1H,J=2.4Hz,Ar-H),8.08(d,2H,J=8.8Hz,Ar-H),7.97(dd,1H,J1=1.2Hz,J2=8.0Hz,Ar-H),7.56(d,1H,J=1.2Hz,Ar-H),7.55(d,1H,J=8.0Hz,Ar-H),7.47(d,2H,J=2.4Hz,Ar-H),7.37(d,2H,J=8.8Hz,Ar-H),5.66(t,1H,J=7.6Hz,CH),4.53(d,1H,J=17.6Hz,ArCH2),4.31(d,1H,J=17.6Hz,ArCH2),3.69(s,4H,CH2CH2),3.24-3.13(m,2H,CH2COOH);ESI-MS(m/z):552.15[M-Cl]+。 The operation process is the same as in Example 5, except that the m-guanidinobenzoic acid hydrochloride in the step (c) is replaced with 4-[(4,5-dihydro-1H-imidazol-2-yl)amino]benzoic acid salt Salt replacement, condensing agent 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide hydrochloride is replaced by DCC, and 3-(3,5-dichlorophenyl)-3-[ 6-[4-[(4,5-Dihydro-1H-imidazol-2-yl)amino]benzamido]-1-oxoisoindol-2-yl]propion hydrochloride, yield 90.0 %, 1 H NMR (400MHz, DMSO-d 6 ) δ: 12.49 (brs, 1H, COOH), 11.86 (brs, 1H, HCl), 10.58 (s, 1H, CONH), 8.85 (brs, 2H, NHCNH) , 8.25(d, 1H, J=2.4Hz, Ar-H), 8.08(d, 2H, J=8.8Hz, Ar-H), 7.97(dd, 1H, J1 =1.2Hz, J2 =8.0Hz , Ar-H), 7.56(d, 1H, J=1.2Hz, Ar-H), 7.55(d, 1H, J=8.0Hz, Ar-H), 7.47(d, 2H, J=2.4Hz, Ar -H), 7.37 (d, 2H, J=8.8Hz, Ar-H), 5.66 (t, 1H, J=7.6Hz, CH), 4.53 (d, 1H, J=17.6Hz, ArCH 2 ), 4.31 (d, 1H, J=17.6Hz, ArCH 2 ), 3.69 (s, 4H, CH 2 CH 2 ), 3.24-3.13 (m, 2H, CH 2 COOH); ESI-MS (m/z): 552.15[ M-Cl] + .
实施例9 Example 9
采用实施例1~8类似的合成方法,只是将相应的反应底物进行替换,即可得到相应的1,3-二氢-1-氧-2H-异吲哚类化合物(I),其结构式如下: Adopt the similar synthetic method of embodiment 1~8, just replace corresponding reaction substrate, can obtain corresponding 1,3-dihydro-1-oxo-2H-isoindole compound (I), its structural formula as follows:
3-[6-(2-胍基乙酰胺基)-1-氧异吲哚-2-基]丙酸盐酸盐(I-9) 3-[6-(2-guanidinoacetamido)-1-oxoisoindol-2-yl]propion hydrochloride (I-9)
收率40.5%,ESI-MS(m/z):320.10[M-Cl]+; Yield 40.5%, ESI-MS (m/z): 320.10[M-Cl] + ;
3-[6-(3-胍基丙酰胺基)-1-氧异吲哚-2-基]丙酸盐酸盐(I-10) 3-[6-(3-guanidinopropionamido)-1-oxoisoindol-2-yl]propion hydrochloride (I-10)
收率36.0%,ESI-MS(m/z):334.20[M-Cl]+; Yield 36.0%, ESI-MS (m/z): 334.20[M-Cl] + ;
3-[1-氧-6-[3-[(1,4,5,6-四氢嘧啶-2-基)氨基]苯甲酰胺基]异吲哚-2-基]-3-苯基丙酸盐酸盐(I-11) 3-[1-Oxo-6-[3-[(1,4,5,6-tetrahydropyrimidin-2-yl)amino]benzamido]isoindol-2-yl]-3-phenyl Propion hydrochloride (I-11)
收率79.5%,ESI-MS(m/z):498.20[M-Cl]+; Yield 79.5%, ESI-MS (m/z): 498.20[M-Cl] + ;
3-[1-氧-6-[4-[(1,4,5,6-四氢嘧啶-2-基)氨基]苯甲酰胺基]异吲哚-2-基]-3-苯基丙酸盐酸盐(I- 12) 3-[1-Oxo-6-[4-[(1,4,5,6-tetrahydropyrimidin-2-yl)amino]benzamido]isoindol-2-yl]-3-phenyl Propion hydrochloride (I-12)
收率77.0%,ESI-MS(m/z):498.10[M-Cl]+; Yield 77.0%, ESI-MS (m/z): 498.10[M-Cl] + ;
3-(3,5-二氯苯基)-3-[1-氧-6-[3-[(1,4,5,6-四氢嘧啶-2-基)氨基]苯甲酰胺基]异吲哚-2-基]丙酸盐酸盐(I-13) 3-(3,5-dichlorophenyl)-3-[1-oxo-6-[3-[(1,4,5,6-tetrahydropyrimidin-2-yl)amino]benzamido] Isoindol-2-yl]propion hydrochloride (I-13)
收率87.5%,ESI-MS(m/z):566.10[M-Cl]+; Yield 87.5%, ESI-MS (m/z): 566.10[M-Cl] + ;
3-(3,5-二氯苯基)-3-[1-氧-6-[4-[(1,4,5,6-四氢嘧啶-2-基)氨基]苯甲酰胺基]异吲哚-2-基]丙酸盐酸盐(I-14) 3-(3,5-dichlorophenyl)-3-[1-oxo-6-[4-[(1,4,5,6-tetrahydropyrimidin-2-yl)amino]benzamido] Isoindol-2-yl]propion hydrochloride (I-14)
收率90.0%,ESI-MS(m/z):566.05[M-Cl]+; Yield 90.0%, ESI-MS (m/z): 566.05[M-Cl] + ;
3-(3,5-二氯苯基)-3-[1-氧-6-[3-[(5-氟-1,4,5,6-四氢嘧啶-2-基)氨基]苯甲酰胺基]异吲哚-2-基]丙酸盐酸盐(I-15) 3-(3,5-dichlorophenyl)-3-[1-oxo-6-[3-[(5-fluoro-1,4,5,6-tetrahydropyrimidin-2-yl)amino]benzene Formamido]isoindol-2-yl]propion hydrochloride (I-15)
收率85.6%,ESI-MS(m/z):584.10[M-Cl]+; Yield 85.6%, ESI-MS (m/z): 584.10[M-Cl] + ;
3-(3,5-二氯苯基)-3-[1-氧-6-[3-[(5-甲基-1,4,5,6-四氢嘧啶-2-基)氨基]苯甲酰胺基]异吲哚-2-基]丙酸盐酸盐(I-16) 3-(3,5-dichlorophenyl)-3-[1-oxo-6-[3-[(5-methyl-1,4,5,6-tetrahydropyrimidin-2-yl)amino] Benzamido]isoindol-2-yl]propion hydrochloride (I-16)
收率87.3%,ESI-MS(m/z):580.20[M-Cl]+; Yield 87.3%, ESI-MS (m/z): 580.20[M-Cl] + ;
3-(3,5-二氯苯基)-3-[1-氧-6-[3-[(5-三氟甲基-1,4,5,6-四氢嘧啶-2-基)氨基]苯甲酰胺基]异吲哚-2- 基]丙酸盐酸盐(I-17) 3-(3,5-dichlorophenyl)-3-[1-oxo-6-[3-[(5-trifluoromethyl-1,4,5,6-tetrahydropyrimidin-2-yl) Amino]benzamido]isoindol-2-yl]propion hydrochloride (I-17)
收率93.5%,ESI-MS(m/z):634.10[M-Cl]+; Yield 93.5%, ESI-MS (m/z): 634.10[M-Cl] + ;
3-(3,5-二氯苯基)-3-[1-氧-6-[3-[(5-羟基-1,4,5,6-四氢嘧啶-2-基)氨基]苯甲酰胺基]异吲哚-2-基]丙酸盐酸盐(I-18) 3-(3,5-dichlorophenyl)-3-[1-oxo-6-[3-[(5-hydroxy-1,4,5,6-tetrahydropyrimidin-2-yl)amino]benzene Formamido]isoindol-2-yl]propion hydrochloride (I-18)
收率80.5%,ESI-MS(m/z):582.15[M-Cl]+; Yield 80.5%, ESI-MS (m/z): 582.15[M-Cl] + ;
3-(4-甲氧基苯基)-3-[6-[3-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]丙酸盐酸盐(I-19) 3-(4-methoxyphenyl)-3-[6-[3-[(4,5-dihydro-1H-imidazol-2-yl)amino]benzamido]-1-oxoisoindo Indol-2-yl]propion hydrochloride (I-19)
收率80.5%,ESI-MS(m/z):514.20[M-C1]+; Yield 80.5%, ESI-MS (m/z): 514.20[M-C1] + ;
3-(4-甲氧基苯基)-3-[1-氧-6-[3-[(1,4,5,6-四氢嘧啶-2-基)氨基]苯甲酰胺基]异吲哚-2-基]丙酸盐酸盐(I-20) 3-(4-methoxyphenyl)-3-[1-oxo-6-[3-[(1,4,5,6-tetrahydropyrimidin-2-yl)amino]benzamido]iso Indol-2-yl]propion hydrochloride (I-20)
收率77.3%,ESI-MS(m/z):528.20[M-Cl]+; Yield 77.3%, ESI-MS (m/z): 528.20[M-Cl] + ;
3-(4-甲氧基苯基)-3-[6-[4-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]丙酸盐酸盐(I-21) 3-(4-methoxyphenyl)-3-[6-[4-[(4,5-dihydro-1H-imidazol-2-yl)amino]benzamido]-1-oxoisoindo Indol-2-yl]propion hydrochloride (I-21)
收率79.5%,ESI-MS(m/z):514.15[M-Cl]+; Yield 79.5%, ESI-MS (m/z): 514.15[M-Cl] + ;
3-(4-甲氧基苯基)-3-[1-氧-6-[3-[(5-氟-1,4,5,6-四氢嘧啶-2-基)氨基]苯甲酰胺基]异吲哚-2-基]丙酸盐酸盐(I-22) 3-(4-methoxyphenyl)-3-[1-oxo-6-[3-[(5-fluoro-1,4,5,6-tetrahydropyrimidin-2-yl)amino]benzidine Amino]isoindol-2-yl]propion hydrochloride (I-22)
收率85.0%,ESI-MS(m/z):546.30[M-Cl]+; Yield 85.0%, ESI-MS (m/z): 546.30[M-Cl] + ;
3-(4-溴苯基)-3-[6-[3-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]丙酸盐酸盐(I-23) 3-(4-Bromophenyl)-3-[6-[3-[(4,5-dihydro-1H-imidazol-2-yl)amino]benzamido]-1-oxoisoindole- 2-yl] propionate hydrochloride (I-23)
收率88.5%,ESI-MS(m/z):562.20[M-Cl]+; Yield 88.5%, ESI-MS (m/z): 562.20[M-Cl] + ;
3-(4-溴苯基)-3-[1-氧-6-[3-[(1,4,5,6-四氢嘧啶-2-基)氨基]苯甲酰胺基]异吲哚-2-基]丙酸盐酸盐(I-24) 3-(4-Bromophenyl)-3-[1-oxo-6-[3-[(1,4,5,6-tetrahydropyrimidin-2-yl)amino]benzamido]isoindole -2-yl] propionate hydrochloride (I-24)
收率90.0%,ESI-MS(m/z):576.15[M-Cl]+; Yield 90.0%, ESI-MS (m/z): 576.15[M-Cl] + ;
3-(4-溴苯基)-3-[1-氧-6-[3-[(5-三氟甲基-1,4,5,6-四氢嘧啶-2-基)氨基]苯甲酰胺基]异吲哚-2-基]丙酸盐酸盐(I-25) 3-(4-Bromophenyl)-3-[1-oxo-6-[3-[(5-trifluoromethyl-1,4,5,6-tetrahydropyrimidin-2-yl)amino]benzene Formamido]isoindol-2-yl]propion hydrochloride (I-25)
收率82.3%,ESI-MS(m/z):644.10[M-Cl]+; Yield 82.3%, ESI-MS (m/z): 644.10[M-Cl] + ;
3-(3-硝基苯基)-3-[6-[3-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]丙酸盐酸盐(I-26) 3-(3-nitrophenyl)-3-[6-[3-[(4,5-dihydro-1H-imidazol-2-yl)amino]benzamido]-1-oxoisoindole -2-yl] propionate hydrochloride (I-26)
收率75.5%,ESI-MS(m/z):529.20[M-Cl]+; Yield 75.5%, ESI-MS (m/z): 529.20[M-Cl] + ;
3-(3-硝基苯基)-3-[1-氧-6-[3-[(1,4,5,6-四氢嘧啶-2-基)氨基]苯甲酰胺基]异吲哚-2-基]丙酸盐酸盐(I-27) 3-(3-nitrophenyl)-3-[1-oxo-6-[3-[(1,4,5,6-tetrahydropyrimidin-2-yl)amino]benzamido]isoindo Indol-2-yl]propion hydrochloride (I-27)
收率73.2%,ESI-MS(m/z):543.25[M-Cl]+; Yield 73.2%, ESI-MS (m/z): 543.25[M-Cl] + ;
3-(4-甲氧基-3-硝基苯基)-3-[6-[3-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]丙酸盐酸盐(I-28) 3-(4-methoxy-3-nitrophenyl)-3-[6-[3-[(4,5-dihydro-1H-imidazol-2-yl)amino]benzamido]- 1-Oxoisoindol-2-yl]propion hydrochloride (I-28)
收率83.2%,ESI-MS(m/z):559.20[M-Cl]+; Yield 83.2%, ESI-MS (m/z): 559.20[M-Cl] + ;
3-(4-甲氧基-3-硝基苯基)-3-[1-氧-6-[3-[(1,4,5,6-四氢嘧啶-2-基)氨基]苯甲酰胺基]异吲哚-2-基]丙酸盐酸盐(I-29) 3-(4-methoxy-3-nitrophenyl)-3-[1-oxo-6-[3-[(1,4,5,6-tetrahydropyrimidin-2-yl)amino]benzene Formamido]isoindol-2-yl]propion hydrochloride (I-29)
收率75.3%,ESI-MS(m/z):573.15[M-Cl]+; Yield 75.3%, ESI-MS (m/z): 573.15[M-Cl] + ;
3-(4-二甲胺基-3-硝基苯基)-3-[6-[3-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]丙酸盐酸盐(I-30) 3-(4-Dimethylamino-3-nitrophenyl)-3-[6-[3-[(4,5-dihydro-1H-imidazol-2-yl)amino]benzamido] -1-Oxoisoindol-2-yl]propion hydrochloride (I-30)
收率70.0%,ESI-MS(m/z):572.25[M-Cl]+; Yield 70.0%, ESI-MS (m/z): 572.25[M-Cl] + ;
3-(4-二甲胺基-3-硝基苯基)-3-[6-[4-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]丙酸盐酸盐(I-31) 3-(4-Dimethylamino-3-nitrophenyl)-3-[6-[4-[(4,5-dihydro-1H-imidazol-2-yl)amino]benzamido] -1-Oxoisoindol-2-yl]propion hydrochloride (I-31)
收率68.5%,ESI-MS(m/z):572.20[M-Cl]+; Yield 68.5%, ESI-MS (m/z): 572.20[M-Cl] + ;
3-(4-氯-3-硝基苯基)-3-[6-[3-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]丙酸盐酸盐(I-32) 3-(4-chloro-3-nitrophenyl)-3-[6-[3-[(4,5-dihydro-1H-imidazol-2-yl)amino]benzamido]-1- Oxyisoindol-2-yl]propion hydrochloride (I-32)
收率82.0%,ESI-MS(m/z):563.15[M-Cl]+; Yield 82.0%, ESI-MS (m/z): 563.15[M-Cl] + ;
3-(4-氯-3-硝基苯基)-3-[6-[4-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]丙酸盐酸盐(I-33) 3-(4-chloro-3-nitrophenyl)-3-[6-[4-[(4,5-dihydro-1H-imidazol-2-yl)amino]benzamido]-1- Oxyisoindol-2-yl]propion hydrochloride (I-33)
收率75.5%,ESI-MS(m/z):563.15[M-Cl]+; Yield 75.5%, ESI-MS (m/z): 563.15[M-Cl] + ;
3-(3,4-二氯苯基)-3-[6-[3-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]丙酸盐酸盐(I-34) 3-(3,4-dichlorophenyl)-3-[6-[3-[(4,5-dihydro-1H-imidazol-2-yl)amino]benzamido]-1-oxoiso Indol-2-yl]propion hydrochloride (I-34)
收率86.0%,ESI-MS(m/z):552.10[M-Cl]+; Yield 86.0%, ESI-MS (m/z): 552.10[M-Cl] + ;
3-(3,4-二氯苯基)-3-[6-[4-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]丙酸盐酸盐(I-35) 3-(3,4-dichlorophenyl)-3-[6-[4-[(4,5-dihydro-1H-imidazol-2-yl)amino]benzamido]-1-oxoiso Indol-2-yl]propion hydrochloride (I-35)
收率82.0%,ESI-MS(m/z):552.20[M-Cl]+; Yield 82.0%, ESI-MS (m/z): 552.20[M-Cl] + ;
3-(3,4-二甲氧基苯基)-3-[6-[3-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]丙酸盐酸盐(I-36) 3-(3,4-dimethoxyphenyl)-3-[6-[3-[(4,5-dihydro-1H-imidazol-2-yl)amino]benzamido]-1- Oxyisoindol-2-yl]propion hydrochloride (I-36)
收率75.0%,ESI-MS(m/z):544.30[M-Cl]+; Yield 75.0%, ESI-MS (m/z): 544.30[M-Cl] + ;
3-(3,4-二甲氧基苯基)-3-[6-[4-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]丙酸盐酸盐(I-37) 3-(3,4-dimethoxyphenyl)-3-[6-[4-[(4,5-dihydro-1H-imidazol-2-yl)amino]benzamido]-1- Oxyisoindol-2-yl]propion hydrochloride (I-37)
收率72.9%,ESI-MS(m/z):544.20[M-Cl]+; Yield 72.9%, ESI-MS (m/z): 544.20[M-Cl] + ;
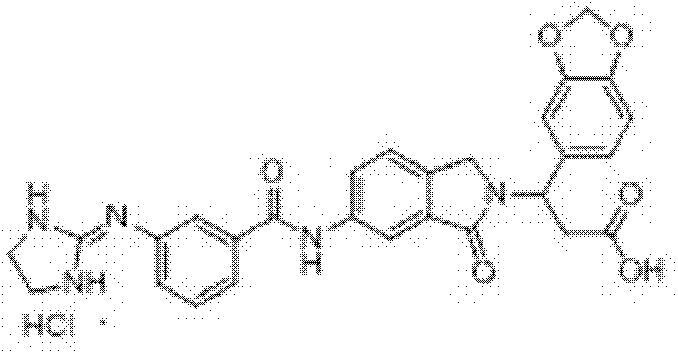
3-(苯并[d][1,3]二噁-5-基)-3-[6-[3-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]丙酸盐酸盐(I-38) 3-(Benzo[d][1,3]diox-5-yl)-3-[6-[3-[(4,5-dihydro-1H-imidazol-2-yl)amino]benzyl Amino]-1-oxoisoindol-2-yl]propion hydrochloride (I-38)
收率77.5%,ESI-MS(m/z):528.20[M-Cl]+; Yield 77.5%, ESI-MS (m/z): 528.20[M-Cl] + ;
3-(苯并[d][1,3]二噁-5-基)-3-[6-[4-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]丙酸盐酸盐(I-39) 3-(Benzo[d][1,3]diox-5-yl)-3-[6-[4-[(4,5-dihydro-1H-imidazol-2-yl)amino]benzyl Amino]-1-oxoisoindol-2-yl]propion hydrochloride (I-39)
收率69.5%,ESI-MS(m/z):528.10[M-Cl]+; Yield 69.5%, ESI-MS (m/z): 528.10[M-Cl] + ;
3-(苯并[d][1,3]二噁-4-基)-3-[6-[3-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]丙酸盐酸盐(I-40) 3-(Benzo[d][1,3]diox-4-yl)-3-[6-[3-[(4,5-dihydro-1H-imidazol-2-yl)amino]benzyl Amino]-1-oxoisoindol-2-yl]propion hydrochloride (I-40)
收率65.6%,ESI-MS(m/z):528.05[M-Cl]+; Yield 65.6%, ESI-MS (m/z): 528.05[M-Cl] + ;
3-(苯并[d][1,3]二噁-4-基)-3-[6-[4-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]丙酸盐酸盐(I-41) 3-(Benzo[d][1,3]diox-4-yl)-3-[6-[4-[(4,5-dihydro-1H-imidazol-2-yl)amino]benzyl Amino]-1-oxoisoindol-2-yl]propion hydrochloride (I-41)
收率63.0%,ESI-MS(m/z):528.20[M-Cl]+; Yield 63.0%, ESI-MS (m/z): 528.20[M-Cl] + ;
3-(2-呋喃基)-3-[6-[3-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]丙酸盐酸盐(I-42) 3-(2-furyl)-3-[6-[3-[(4,5-dihydro-1H-imidazol-2-yl)amino]benzamido]-1-oxoisoindole-2 -yl] propionate hydrochloride (I-42)
收率55.0%,ESI-MS(m/z):474.15[M-Cl]+; Yield 55.0%, ESI-MS (m/z): 474.15[M-Cl] + ;
3-(2-噻吩基)-3-[6-[3-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]丙酸盐酸盐(I-43) 3-(2-thienyl)-3-[6-[3-[(4,5-dihydro-1H-imidazol-2-yl)amino]benzamido]-1-oxoisoindole-2 -yl] propionate hydrochloride (I-43)
收率72.0%,ESI-MS(m/z):490.20[M-Cl]+; Yield 72.0%, ESI-MS (m/z): 490.20[M-Cl] + ;
3-(3-噻吩基)-3-[6-[3-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]丙酸盐酸盐(I-44) 3-(3-thienyl)-3-[6-[3-[(4,5-dihydro-1H-imidazol-2-yl)amino]benzamido]-1-oxoisoindole-2 -yl] propionate hydrochloride (I-44)
收率70.3%,ESI-MS(m/z):490.15[M-Cl]+; Yield 70.3%, ESI-MS (m/z): 490.15[M-Cl] + ;
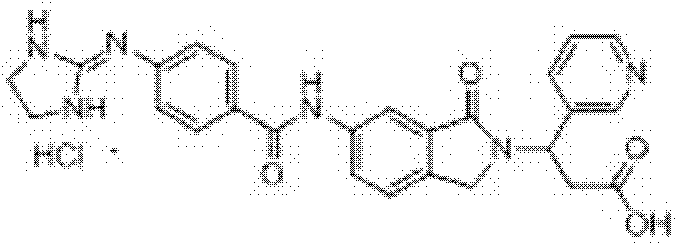
3-(吡啶-3-基)-3-[6-[3-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]丙酸盐酸 盐(I-45) 3-(pyridin-3-yl)-3-[6-[3-[(4,5-dihydro-1H-imidazol-2-yl)amino]benzamido]-1-oxoisoindole- 2-yl] propionate hydrochloride (I-45)
收率62.5%,ESI-MS(m/z):485.20[M-Cl]+; Yield 62.5%, ESI-MS (m/z): 485.20[M-Cl] + ;
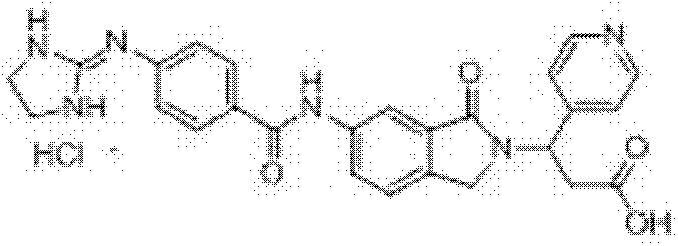
3-(吡啶-3-基)-3-[6-[4-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]丙酸盐酸盐(I-46) 3-(pyridin-3-yl)-3-[6-[4-[(4,5-dihydro-1H-imidazol-2-yl)amino]benzamido]-1-oxoisoindole- 2-yl] propionate hydrochloride (I-46)
收率60.0%,ESI-MS(m/z):485.20[M-Cl]+; Yield 60.0%, ESI-MS (m/z): 485.20[M-Cl] + ;
3-(吡啶-4-基)-3-[6-[3-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]丙酸盐酸盐(I-47) 3-(pyridin-4-yl)-3-[6-[3-[(4,5-dihydro-1H-imidazol-2-yl)amino]benzamido]-1-oxoisoindole- 2-yl] propionate hydrochloride (I-47)
收率65.5%,ESI-MS(m/z):485.25[M-Cl]+; Yield 65.5%, ESI-MS (m/z): 485.25[M-Cl] + ;
3-(吡啶-4-基)-3-[6-[4-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]丙酸盐酸盐(I-48) 3-(pyridin-4-yl)-3-[6-[4-[(4,5-dihydro-1H-imidazol-2-yl)amino]benzamido]-1-oxoisoindole- 2-yl] propionate hydrochloride (I-48)
收率57.0%,ESI-MS(m/z):485.20[M-Cl]+; Yield 57.0%, ESI-MS (m/z): 485.20[M-Cl] + ;
3-(苯并呋喃-4-基)-3-[6-[3-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]丙酸盐酸盐(I-49) 3-(benzofuran-4-yl)-3-[6-[3-[(4,5-dihydro-1H-imidazol-2-yl)amino]benzamido]-1-oxoisoind Indol-2-yl]propion hydrochloride (I-49)
收率85.5%,ESI-MS(m/z):524.25[M-Cl]+; Yield 85.5%, ESI-MS (m/z): 524.25[M-Cl] + ;
3-(苯并呋喃-4-基)-3-[6-[4-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]丙酸 盐酸盐(I-50) 3-(benzofuran-4-yl)-3-[6-[4-[(4,5-dihydro-1H-imidazol-2-yl)amino]benzamido]-1-oxoisoind Indol-2-yl]propionic acid hydrochloride (I-50)
收率87.0%,ESI-MS(m/z):524.20[M-Cl]+; Yield 87.0%, ESI-MS (m/z): 524.20[M-Cl] + ;
3-(7-氯-苯并呋喃-4-基)-3-[6-[3-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]丙酸盐酸盐(I-51) 3-(7-Chloro-benzofuran-4-yl)-3-[6-[3-[(4,5-dihydro-1H-imidazol-2-yl)amino]benzamido]-1 -Oxyisoindol-2-yl]propion hydrochloride (I-51)
收率90.0%,ESI-MS(m/z):558.20[M-Cl]+; Yield 90.0%, ESI-MS (m/z): 558.20[M-Cl] + ;
3-(7-氯-苯并呋喃-4-基)-3-[6-[4-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]丙酸盐酸盐(I-52) 3-(7-Chloro-benzofuran-4-yl)-3-[6-[4-[(4,5-dihydro-1H-imidazol-2-yl)amino]benzamido]-1 -Oxyisoindol-2-yl]propion hydrochloride (I-52)
收率93.2%,ESI-MS(m/z):558.10[M-Cl]+; Yield 93.2%, ESI-MS (m/z): 558.10[M-Cl] + ;
3-(2,3-二氢苯并呋喃-4-基)-3-[6-[3-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]丙酸盐酸盐(I-53) 3-(2,3-Dihydrobenzofuran-4-yl)-3-[6-[3-[(4,5-dihydro-1H-imidazol-2-yl)amino]benzamido] -1-Oxoisoindol-2-yl]propion hydrochloride (I-53)
收率87.5%,ESI-MS(m/z):526.20[M-Cl]+; Yield 87.5%, ESI-MS (m/z): 526.20[M-Cl] + ;
3-(2,3-二氢苯并呋喃-4-基)-3-[6-[4-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]丙酸盐酸盐(I-54) 3-(2,3-Dihydrobenzofuran-4-yl)-3-[6-[4-[(4,5-dihydro-1H-imidazol-2-yl)amino]benzamido] -1-Oxoisoindol-2-yl]propion hydrochloride (I-54)
收率85.6%,ESI-MS(m/z):526.20[M-Cl]+; Yield 85.6%, ESI-MS (m/z): 526.20[M-Cl] + ;
3-(2,3-二氢苯并呋喃-5-基)-3-[6-[3-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]丙酸盐酸盐(I-55) 3-(2,3-Dihydrobenzofuran-5-yl)-3-[6-[3-[(4,5-dihydro-1H-imidazol-2-yl)amino]benzamido] -1-Oxoisoindol-2-yl]propion hydrochloride (I-55)
收率80.8%,ESI-MS(m/z):526.25[M-Cl]+; Yield 80.8%, ESI-MS (m/z): 526.25[M-Cl] + ;
3-(2,3-二氢苯并呋喃-5-基)-3-[6-[4-[(4,5-二氢-1H-咪唑-2-基)氨基]苯甲酰胺基]-1-氧异吲哚-2-基]丙酸盐酸盐(I-56) 3-(2,3-Dihydrobenzofuran-5-yl)-3-[6-[4-[(4,5-dihydro-1H-imidazol-2-yl)amino]benzamido] -1-Oxoisoindol-2-yl]propion hydrochloride (I-56)
收率82.0%,ESI-MS(m/z):526.15[M-Cl]+。 Yield 82.0%, ESI-MS (m/z): 526.15 [M-Cl] + .
实施例10生物活性测试结果 Embodiment 10 biological activity test result
表一:部分目标物对黑色素瘤M21细胞在Fibronectin上粘附的抑制率(n=3) Table 1: Inhibition rate of some targets on the adhesion of melanoma M21 cells on Fibronectin (n=3)
结论:本发明所公开的1,3-二氢-1-氧-2H-异吲哚类化合物(I)或其药学上可接受的盐可显著抑制黑色素瘤细胞M21对fibronectin的粘附,部分目标化合物的抑制活性较阳性对照药物——cyclo(RGDfV)强。 Conclusion: The 1,3-dihydro-1-oxo-2H-isoindole compound (I) or its pharmaceutically acceptable salt disclosed in the present invention can significantly inhibit the adhesion of melanoma cell M21 to fibronectin, and some The inhibitory activity of the target compound was stronger than that of the positive control drug—cyclo(RGDfV). the
Claims (3)
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201210038183.7A CN102603610B (en) | 2012-02-21 | 2012-02-21 | 1,3-dihydro-1-oxo-2H-isoindole compound, its preparation method and use |
| CN201410270438.1A CN104045594B (en) | 2012-02-21 | 2012-02-21 | One class 1,3-dihydro-1-oxygen-2H-isoindoles compound and application thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201210038183.7A CN102603610B (en) | 2012-02-21 | 2012-02-21 | 1,3-dihydro-1-oxo-2H-isoindole compound, its preparation method and use |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201410270438.1A Division CN104045594B (en) | 2012-02-21 | 2012-02-21 | One class 1,3-dihydro-1-oxygen-2H-isoindoles compound and application thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN102603610A CN102603610A (en) | 2012-07-25 |
| CN102603610B true CN102603610B (en) | 2014-07-09 |
Family
ID=46521452
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201210038183.7A Expired - Fee Related CN102603610B (en) | 2012-02-21 | 2012-02-21 | 1,3-dihydro-1-oxo-2H-isoindole compound, its preparation method and use |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN102603610B (en) |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN103159667B (en) * | 2013-03-15 | 2015-04-01 | 四川大学 | 6-guanidyl-2H-isoindole compound as well as preparation method and application thereof |
| CN105294533A (en) * | 2015-12-02 | 2016-02-03 | 宋彤云 | Pharmaceutical composition for treating bone diseases |
| CN108530497A (en) * | 2018-04-09 | 2018-09-14 | 江南大学 | Glycoprotein amino acid and the oligosaccharides analogies prepared using glycoprotein amino acid |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA2331461C (en) * | 1998-05-11 | 2008-10-07 | Entremed, Inc. | Analogs of 2-phthalimidinoglutaric acid and their use as inhibitors of angiogenesis |
| CN1190420C (en) * | 2001-11-07 | 2005-02-23 | 四川大学 | Non-peptide neomatal vascularization inhibitor |
| US7405237B2 (en) * | 2004-07-28 | 2008-07-29 | Celgene Corporation | Isoindoline compounds and methods of their use |
-
2012
- 2012-02-21 CN CN201210038183.7A patent/CN102603610B/en not_active Expired - Fee Related
Also Published As
| Publication number | Publication date |
|---|---|
| CN102603610A (en) | 2012-07-25 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US11512087B2 (en) | BRK inhibitory compound | |
| CN107406426B (en) | Cyclic ureas as ROCK inhibitors | |
| TW448175B (en) | Cycloalkano-indole and-azaindole derivatives | |
| CN100436425C (en) | Fused imidazolium derivatives | |
| US8338565B2 (en) | Macrocyclic compounds for inhibition of tumor necrosis factor alpha | |
| CN110857293B (en) | Novel quinoline derivative inhibitor | |
| EP3752499B1 (en) | Inhibitors of trpc6 | |
| TW200808310A (en) | Amide derivative or salt thereof | |
| WO2018130124A1 (en) | Tricyclic compound as selective estrogen receptor down-regulator and use thereof | |
| CA2990564A1 (en) | Bicyclic heterocyclic amide derivative | |
| CN105683175A (en) | Bicyclic inhibitors | |
| KR20070026357A (en) | Indole derivatives and their use as kinase inhibitors, in particular Iv2 inhibitors | |
| CN106687453A (en) | Tetrahydroquinoline derivatives as bromodomain inhibitors | |
| CN102603610B (en) | 1,3-dihydro-1-oxo-2H-isoindole compound, its preparation method and use | |
| WO2018196747A1 (en) | Indoleamine 2,3-dioxygenase inhibitor and application | |
| CN106117182B (en) | Quinazoline-N-phenethyltetrahydroisoquinoline compound and its preparation method and application | |
| JP3936844B2 (en) | Integrin receptor ligand | |
| US10519105B2 (en) | KCNQ2-5 channel activator | |
| CN104045594B (en) | One class 1,3-dihydro-1-oxygen-2H-isoindoles compound and application thereof | |
| CN118271284A (en) | Estrogen receptor modulators and uses thereof | |
| CN112759587A (en) | 3- (dimethylamino methyl) piperidine-4-alcohol derivative and preparation method and pharmaceutical application thereof | |
| CN108117551B (en) | Substituted (1H-pyrazolo [3,4-b ] pyridine) urea compound and anti-tumor application thereof | |
| CN111978317A (en) | Imidazopyridine MNK1/MNK2 kinase inhibitor and preparation method and application thereof | |
| CN103159667B (en) | 6-guanidyl-2H-isoindole compound as well as preparation method and application thereof | |
| CN102336740B (en) | Novel imidazole compound and its application |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| CF01 | Termination of patent right due to non-payment of annual fee | ||
| CF01 | Termination of patent right due to non-payment of annual fee |
Granted publication date: 20140709 Termination date: 20160221 |