CN102552184A - Thrombopoietin peptide analogue freeze-dried preparation - Google Patents
Thrombopoietin peptide analogue freeze-dried preparation Download PDFInfo
- Publication number
- CN102552184A CN102552184A CN2012100345402A CN201210034540A CN102552184A CN 102552184 A CN102552184 A CN 102552184A CN 2012100345402 A CN2012100345402 A CN 2012100345402A CN 201210034540 A CN201210034540 A CN 201210034540A CN 102552184 A CN102552184 A CN 102552184A
- Authority
- CN
- China
- Prior art keywords
- thrombopoietin
- peptide
- buffer
- concentration
- intended
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 108010041111 Thrombopoietin Proteins 0.000 title claims abstract description 64
- 238000002360 preparation method Methods 0.000 title abstract description 19
- 102000036693 Thrombopoietin Human genes 0.000 claims abstract description 61
- DHMQDGOQFOQNFH-UHFFFAOYSA-N Glycine Chemical compound NCC(O)=O DHMQDGOQFOQNFH-UHFFFAOYSA-N 0.000 claims abstract description 50
- 239000004471 Glycine Substances 0.000 claims abstract description 25
- 108090000623 proteins and genes Proteins 0.000 claims abstract description 25
- 102000004169 proteins and genes Human genes 0.000 claims abstract description 24
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 claims abstract description 20
- 235000010355 mannitol Nutrition 0.000 claims abstract description 19
- 235000010482 polyoxyethylene sorbitan monooleate Nutrition 0.000 claims abstract description 17
- 229920000053 polysorbate 80 Polymers 0.000 claims abstract description 17
- 108090000765 processed proteins & peptides Proteins 0.000 claims description 58
- 239000000872 buffer Substances 0.000 claims description 25
- 239000007974 sodium acetate buffer Substances 0.000 claims description 24
- BHZOKUMUHVTPBX-UHFFFAOYSA-M sodium acetic acid acetate Chemical compound [Na+].CC(O)=O.CC([O-])=O BHZOKUMUHVTPBX-UHFFFAOYSA-M 0.000 claims description 22
- 229930195725 Mannitol Natural products 0.000 claims description 17
- 239000000594 mannitol Substances 0.000 claims description 17
- 239000000203 mixture Substances 0.000 claims description 17
- 238000009472 formulation Methods 0.000 claims description 16
- 239000008363 phosphate buffer Substances 0.000 claims description 8
- 239000000243 solution Substances 0.000 claims description 5
- 239000000546 pharmaceutical excipient Substances 0.000 claims description 3
- 239000007981 phosphate-citrate buffer Substances 0.000 claims description 2
- 239000000463 material Substances 0.000 abstract description 5
- 239000007788 liquid Substances 0.000 abstract description 2
- 230000003139 buffering effect Effects 0.000 abstract 1
- 238000009776 industrial production Methods 0.000 abstract 1
- 239000000523 sample Substances 0.000 description 22
- 235000018102 proteins Nutrition 0.000 description 21
- 239000002671 adjuvant Substances 0.000 description 15
- 238000000151 deposition Methods 0.000 description 12
- 239000003814 drug Substances 0.000 description 12
- 239000011550 stock solution Substances 0.000 description 10
- 230000008021 deposition Effects 0.000 description 9
- 239000000843 powder Substances 0.000 description 8
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 7
- 229930006000 Sucrose Natural products 0.000 description 7
- 238000001962 electrophoresis Methods 0.000 description 7
- 239000005720 sucrose Substances 0.000 description 7
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 5
- 208000031981 Thrombocytopenic Idiopathic Purpura Diseases 0.000 description 5
- 201000003710 autoimmune thrombocytopenic purpura Diseases 0.000 description 5
- 230000000694 effects Effects 0.000 description 5
- 238000004519 manufacturing process Methods 0.000 description 5
- 238000000034 method Methods 0.000 description 5
- 238000011160 research Methods 0.000 description 5
- 238000005070 sampling Methods 0.000 description 5
- 238000002415 sodium dodecyl sulfate polyacrylamide gel electrophoresis Methods 0.000 description 5
- 238000012360 testing method Methods 0.000 description 5
- 206010021245 Idiopathic thrombocytopenic purpura Diseases 0.000 description 4
- 239000007853 buffer solution Substances 0.000 description 4
- 238000002474 experimental method Methods 0.000 description 4
- 229920001184 polypeptide Polymers 0.000 description 4
- 102000004196 processed proteins & peptides Human genes 0.000 description 4
- 238000004007 reversed phase HPLC Methods 0.000 description 4
- 150000001413 amino acids Chemical group 0.000 description 3
- 238000001514 detection method Methods 0.000 description 3
- 229940079593 drug Drugs 0.000 description 3
- 238000005516 engineering process Methods 0.000 description 3
- 230000007774 longterm Effects 0.000 description 3
- 238000010911 splenectomy Methods 0.000 description 3
- 238000013112 stability test Methods 0.000 description 3
- KRKNYBCHXYNGOX-UHFFFAOYSA-K Citrate Chemical compound [O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O KRKNYBCHXYNGOX-UHFFFAOYSA-K 0.000 description 2
- 108060003951 Immunoglobulin Proteins 0.000 description 2
- 206010028980 Neoplasm Diseases 0.000 description 2
- 230000001133 acceleration Effects 0.000 description 2
- 235000001014 amino acid Nutrition 0.000 description 2
- 238000004458 analytical method Methods 0.000 description 2
- 201000011510 cancer Diseases 0.000 description 2
- 230000008859 change Effects 0.000 description 2
- 239000003795 chemical substances by application Substances 0.000 description 2
- 238000000855 fermentation Methods 0.000 description 2
- 230000004151 fermentation Effects 0.000 description 2
- 238000006206 glycosylation reaction Methods 0.000 description 2
- 230000036541 health Effects 0.000 description 2
- 102000018358 immunoglobulin Human genes 0.000 description 2
- 238000002347 injection Methods 0.000 description 2
- 239000007924 injection Substances 0.000 description 2
- 238000011835 investigation Methods 0.000 description 2
- 210000001501 megacaryocyte Anatomy 0.000 description 2
- 210000003593 megakaryocyte Anatomy 0.000 description 2
- 230000006320 pegylation Effects 0.000 description 2
- 238000004321 preservation Methods 0.000 description 2
- 230000008569 process Effects 0.000 description 2
- 239000012488 sample solution Substances 0.000 description 2
- 238000005303 weighing Methods 0.000 description 2
- QKNYBSVHEMOAJP-UHFFFAOYSA-N 2-amino-2-(hydroxymethyl)propane-1,3-diol;hydron;chloride Chemical compound Cl.OCC(N)(CO)CO QKNYBSVHEMOAJP-UHFFFAOYSA-N 0.000 description 1
- JGSARLDLIJGVTE-UHFFFAOYSA-N 3,3-dimethyl-7-oxo-6-[(2-phenylacetyl)amino]-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid Chemical compound O=C1N2C(C(O)=O)C(C)(C)SC2C1NC(=O)CC1=CC=CC=C1 JGSARLDLIJGVTE-UHFFFAOYSA-N 0.000 description 1
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 1
- 102000009027 Albumins Human genes 0.000 description 1
- 108010088751 Albumins Proteins 0.000 description 1
- 239000004475 Arginine Substances 0.000 description 1
- 244000025254 Cannabis sativa Species 0.000 description 1
- 206010008111 Cerebral haemorrhage Diseases 0.000 description 1
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 1
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 1
- 206010018276 Gingival bleeding Diseases 0.000 description 1
- 208000024283 Gingival haemorrhages Diseases 0.000 description 1
- 102000003886 Glycoproteins Human genes 0.000 description 1
- 108090000288 Glycoproteins Proteins 0.000 description 1
- 208000032843 Hemorrhage Diseases 0.000 description 1
- 102000008100 Human Serum Albumin Human genes 0.000 description 1
- 108091006905 Human Serum Albumin Proteins 0.000 description 1
- 208000028622 Immune thrombocytopenia Diseases 0.000 description 1
- ONIBWKKTOPOVIA-BYPYZUCNSA-N L-Proline Chemical compound OC(=O)[C@@H]1CCCN1 ONIBWKKTOPOVIA-BYPYZUCNSA-N 0.000 description 1
- QNAYBMKLOCPYGJ-REOHCLBHSA-N L-alanine Chemical compound C[C@H](N)C(O)=O QNAYBMKLOCPYGJ-REOHCLBHSA-N 0.000 description 1
- ODKSFYDXXFIFQN-BYPYZUCNSA-P L-argininium(2+) Chemical compound NC(=[NH2+])NCCC[C@H]([NH3+])C(O)=O ODKSFYDXXFIFQN-BYPYZUCNSA-P 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- ONIBWKKTOPOVIA-UHFFFAOYSA-N Proline Natural products OC(=O)C1CCCN1 ONIBWKKTOPOVIA-UHFFFAOYSA-N 0.000 description 1
- 206010037549 Purpura Diseases 0.000 description 1
- 241001672981 Purpura Species 0.000 description 1
- 102000007056 Recombinant Fusion Proteins Human genes 0.000 description 1
- 108010008281 Recombinant Fusion Proteins Proteins 0.000 description 1
- 239000012505 Superdex™ Substances 0.000 description 1
- 125000000218 acetic acid group Chemical group C(C)(=O)* 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 230000004913 activation Effects 0.000 description 1
- 235000004279 alanine Nutrition 0.000 description 1
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 description 1
- 238000003556 assay Methods 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 230000006472 autoimmune response Effects 0.000 description 1
- 230000001580 bacterial effect Effects 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 239000003124 biologic agent Substances 0.000 description 1
- 230000004071 biological effect Effects 0.000 description 1
- 235000019846 buffering salt Nutrition 0.000 description 1
- 150000001720 carbohydrates Chemical class 0.000 description 1
- 238000010523 cascade reaction Methods 0.000 description 1
- 210000004027 cell Anatomy 0.000 description 1
- 238000002512 chemotherapy Methods 0.000 description 1
- 230000001684 chronic effect Effects 0.000 description 1
- 235000009508 confectionery Nutrition 0.000 description 1
- 230000001276 controlling effect Effects 0.000 description 1
- 239000003246 corticosteroid Substances 0.000 description 1
- 230000006378 damage Effects 0.000 description 1
- 238000011033 desalting Methods 0.000 description 1
- 238000013461 design Methods 0.000 description 1
- 230000004069 differentiation Effects 0.000 description 1
- 238000010790 dilution Methods 0.000 description 1
- 239000012895 dilution Substances 0.000 description 1
- 239000002552 dosage form Substances 0.000 description 1
- 230000007613 environmental effect Effects 0.000 description 1
- 208000001780 epistaxis Diseases 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- 238000004108 freeze drying Methods 0.000 description 1
- 210000001035 gastrointestinal tract Anatomy 0.000 description 1
- 230000013595 glycosylation Effects 0.000 description 1
- 239000003102 growth factor Substances 0.000 description 1
- 238000004128 high performance liquid chromatography Methods 0.000 description 1
- 208000026278 immune system disease Diseases 0.000 description 1
- 230000036039 immunity Effects 0.000 description 1
- 229960003444 immunosuppressant agent Drugs 0.000 description 1
- 230000001861 immunosuppressant effect Effects 0.000 description 1
- 239000003018 immunosuppressive agent Substances 0.000 description 1
- 239000012535 impurity Substances 0.000 description 1
- 230000036512 infertility Effects 0.000 description 1
- 208000020658 intracerebral hemorrhage Diseases 0.000 description 1
- 238000005304 joining Methods 0.000 description 1
- 210000004185 liver Anatomy 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 230000010534 mechanism of action Effects 0.000 description 1
- 239000012528 membrane Substances 0.000 description 1
- 230000003647 oxidation Effects 0.000 description 1
- 238000007254 oxidation reaction Methods 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- 238000011020 pilot scale process Methods 0.000 description 1
- 239000000902 placebo Substances 0.000 description 1
- 229940068196 placebo Drugs 0.000 description 1
- 238000006116 polymerization reaction Methods 0.000 description 1
- 229920005862 polyol Polymers 0.000 description 1
- 150000003077 polyols Chemical class 0.000 description 1
- 230000009465 prokaryotic expression Effects 0.000 description 1
- 239000003223 protective agent Substances 0.000 description 1
- 239000012460 protein solution Substances 0.000 description 1
- 238000000746 purification Methods 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 238000004153 renaturation Methods 0.000 description 1
- 238000012552 review Methods 0.000 description 1
- 229960004641 rituximab Drugs 0.000 description 1
- 238000012216 screening Methods 0.000 description 1
- HEMHJVSKTPXQMS-UHFFFAOYSA-M sodium hydroxide Inorganic materials [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 239000000600 sorbitol Substances 0.000 description 1
- 238000007619 statistical method Methods 0.000 description 1
- 238000003860 storage Methods 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 239000004094 surface-active agent Substances 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 206010043554 thrombocytopenia Diseases 0.000 description 1
- 201000003067 thrombocytopenia due to platelet alloimmunization Diseases 0.000 description 1
- 230000003582 thrombocytopenic effect Effects 0.000 description 1
- 230000001988 toxicity Effects 0.000 description 1
- 231100000419 toxicity Toxicity 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 1
- 239000008215 water for injection Substances 0.000 description 1
Landscapes
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
Abstract
The invention provides a thrombopoietin peptide analogue freeze-dried preparation. The freeze-dried preparation is obtained by dissolving thrombopoietin and medicinal auxiliary materials into a medicinal buffering liquid of which the pH is 5-6.0, wherein the protein concentration of the thrombopoietin is 0.1-1 mg/ml; the medicinal auxiliary materials include mannite, glycine and tween 80; the mass concentration of the mannite is 2-8 percent; the mass concentration of the glycine is 1-5 percent; and the volume concentration of the tween 80 is 0.001-0.01 percent. The freeze-dried preparation containing a thrombopoietin peptide analogue has the advantages of high efficiency, easiness, convenience, low cost and suitability for industrial production.
Description
Technical field
The present invention relates to a kind of freeze-dried powder that thrombopoietin is intended peptide that contains, and corresponding preparation technology.
Background technology
Chronic idiopathic thrombocytopenic purpura (ITP) is a kind of self property immune disease; Cause by causing thrombocytopenic a kind of autoimmune response; It is characterized in that platelet count reduces, its patient faces hemorrhage excessive risk, often causes the little angiorrbagia of patient; Symptom is green grass or young crops, epistaxis and the gingival hemorrhage of becoming silted up, and mortality gastrointestinal tract and intracerebral hemorrhage can appear in severe patient.At present the main means of treatment ITP are to use immunosuppressant class medicine, like corticosteroid, quiet notes immunoglobulin, anti-D immunoglobulin and Genentech Company products Rituximab, then are the row splenectomys to serious debilitating patient's last ditch.Wherein most of medicines had not experienced the investigation of placebo clinical trial at random, though all effective to different patient crowds, also can produce serious toxicity simultaneously, its mechanism of action is to stop platelet destruction, but not promotes platelet to produce.
Thrombopoietin (TPO) is a kind of glycoprotein, mainly results from liver, in megalokaryocyte and hematoblastic generation, plays important regulatory role.Its with can activate the downstream signal cascade reaction after c-Mpl combines, thereby cause Megakaryocytic differentiation and increment, and be accompanied by platelet levels and raise.Short platelet generated agent-glycosylation total length RhTPPO RHTPO (rhTPO) and Pegylation recombined human megakaryocyte growth and grew the factor (PEG-rHuMGDF) and comes out thereupon the 1st generation.It can promote that megalokaryocyte generates in health volunteer's body, and improves platelet counts, and has all obtained success in the research that is used for treating cancer patient, immunity thrombopoietin minimizing purpura patient and accepting the cancer patient of non-clear marrow property chemotherapy.Yet; The short platelet generation agent of the 1st generation may cause producing in the subject anti-TPO antibody; Thereby having the risk of bringing out thrombocytopenia, the exploitation of glycosylation total length RhTPPO RHTPO (rhTPO) and Pegylation recombined human megakaryocyte growth and the growth factor (PEG-rHuMGDF) is compelled to be stopped.After this research worker in polypeptide libraries, seek at random, the polypeptide of no dependency, attempt can stimulate the cell strain that depends on TPO under the situation that does not produce anti-TPO antibody; Polypeptide chain attempting screening is obtained is connected to various carrier molecules, to increase its half-life.Develop thus with thrombopoietin and intend the 2nd generation short platelet generation agent that peptide is representative.Thrombopoietin is intended the test of peptide in health volunteer and ITP patient and has all been obtained success, and does not also see the anti-TPO antibody of generation in the subject in the test.
It is a kind of novel recombinant protein that thrombopoietin is intended peptide, but specificity combines and activation thrombopoietin (TPO) receptor c-Mpl, thereby significantly improves platelet levels.Thrombopoietin is intended peptide and is contained two identical subunits, and each subunit is made up of an IgG1 Fc structural area and c-Mpl land covalent bond respectively, and does not have amino acid sequence homology with TPO, so can in medication person's body, not produce anti-TPO antibody.The production that thrombopoietin is intended peptide is to utilize technique for gene engineering to construct thrombopoietin to intend peptide prokaryotic expression bacterial strain; Adopt fermentation engineering to realize thrombopoietin plan peptide high density fermentation; Be purified into through protein renaturation and purification technique and meet other thrombopoietin of pharmaceutical grade and intend peptide stock solution, be mixed with preparation.Be mainly used in adult's immune thrombocytopenia of treatment splenectomy and non-splenectomy.
But the BA of pharmaceutical grade protein receives multiple Effect of Environmental, and multiple physics or chemical change possibly take place, phenomenons such as for example polymerization, oxidation and degraded, thereby loss of activity greatly.Therefore we have selected the dosage form, adjuvant, the production technology that are fit to, through controlling crucial process conditions, make system product kept very high active and stable.Pilot scale research shows that product manufacture is stable, and is simple to operate, and the cycle is short, and cost is low, is fit to industrialization production.
Summary of the invention
The objective of the invention is to preserve thrombopoietin plan peptide protein, and be suitable for the confused pin of lyophilizing that recombinant platelet generation element is intended peptide that contains of actual clinical use in order to provide a kind of ability to stablize.
For reaching goal of the invention, the technical scheme that the present invention adopts is:
A kind ofly contain the freeze-dried powder that thrombopoietin is intended peptide, said freeze-dried powder is intended in the buffer that peptide and pharmaceutical grade stability adjuvant be dissolved in pH5~6 by thrombopoietin and being obtained, and said thrombopoietin plan peptide protein concentration is 0.1~1mg/mL.
Thrombopoietin is intended peptide stock solution by my company's preparation, and its preparation method is an existing write up in 201110235281.5 at the number of patent application of my company's application, this no longer large fierce dog state.Said stable adjuvant can suitably add as required.Preferred stable adjuvant is mannitol, glycine and Tween 80 among the present invention, and preferred mannitol mass concentration is 2-8%, and more preferably the mannitol mass concentration is 5%.Preferred glycine mass concentration is l-5%, and more preferably the glycine mass concentration is 2.5%.Preferred Tween 80 volumetric concentration is 0.001%-0.01%, and more preferably warm 80 volumetric concentrations are 0.005%.
The protein concentration that said thrombopoietin is intended peptide is 0.1~1mg/mL.
Said buffer is phosphate buffer or citrate buffer solution or acetic acid one sodium-acetate buffer.Said buffer is preferably acetic acid one sodium-acetate buffer, and concentration is 5~50mmol/L.
Preferably; Said freeze-dried powder is to obtain in 5~50mmol/L acetic acid-sodium-acetate buffer by the concentration that thrombopoietin plan peptide and glycine are dissolved in pH5~6; It is 0.1~1mg/mL that said thrombopoietin is intended peptide protein concentration; Said mannitol mass concentration is 2-8%, and the glycine mass concentration is 1~4%, and the Tween 80 volumetric concentration is 0.001%-0.01%.
Preferred; Said freeze-dried powder is intended in acetic acid-sodium-acetate buffer that peptide protein and glycine be dissolved in pH5.5, concentration 20mmol/L by thrombopoietin and being obtained; It is 0.5mg/mL that said thrombopoietin is intended peptide protein concentration; Said mannitol mass concentration is 5%, and the glycine mass concentration is 2.5%, and the Tween 80 volumetric concentration is 0.005%.
The beneficial effect that contains thrombopoietin plan peptide freeze-dried powder pin of the present invention is mainly reflected in: provide a kind of ability the stable lyophilized formulations of preserving thrombopoietin plan peptide protein, and be suitable for industrialization production.
The specific embodiment
Contain the stability that thrombopoietin is intended the peptide medicament in order to improve; If having carried out deep research and discovery selects several kinds of adding preparations from proper supplementary material; And, then can effectively reach this purpose through selecting suitable pH and thrombopoietin to intend the protein concentration of peptide.
Intend in the peptide medicament stability study process at thrombopoietin, chosen the outward appearance detection, the SDS-PAGE electrophoresis detection, rp-hplc analysis, biological activity assays etc. are observed the situation of change that thrombopoietin is intended the peptide medicament as investigating index.
Below in conjunction with specific embodiment the present invention is described further, but protection scope of the present invention is not limited in this:
Embodiment 1: buffer system is intended the influence of stabilized peptide property to thrombopoietin
PH is a key factor that influences protein product stability in the injectable drug preparation.Through adding suitable buffering salt, keep preparation pH.As add phosphate, acetate, citrate etc.In order to investigate stability of formulation under the condition of different pH, carried out the influence experiment of condition of different pH to preparation stability by following condition:
Prepare acetic acid-sodium acetate buffer system respectively, citrate, phosphate buffer, 1ml/ bottle (protein concentration is 0.5mg/ml), pH is designed with 4.0,4.5, and 5.0,5.5,6.0,6.5,7.0,7.5,8.0 concentration are 20mmol/L, (seeing the following form 1).Buffer system through using SUPERDEX G-25 desalting column that thrombopoietin is intended peptide stock solution replaces to design team; And the thrombopoietin of above-mentioned different buffer systems is intended peptide solution be positioned over to deposit under 37 ℃ of conditions and carry out stability test, detect electrophoresis purity (seeing table 2) respectively at sampling in 7,14,28 and 60 days.Detection method; The non-reduced electrophoresis method of SDS-PAGE.
Table 1: the preparation of different buffer systems
| Buffer 1 | 20mmol/L acetic acid-sodium acetate buffer system, pH4.0. |
| Buffer 2 | 20mmol/L acetic acid-sodium acetate buffer system, pH4.5. |
| Buffer 3 | 20mmol/L acetic acid-sodium acetate buffer system, pH5.0. |
| Buffer 4 | 20mmol/L acetic acid-sodium acetate buffer system, pH5.5. |
| Buffer 5 | The 20mmol/L phosphate buffer, pH6.0. |
| Buffer 6 | The 20mmol/L phosphate buffer, pH6.5. |
| Buffer 7 | The 20mmol/L phosphate buffer, pH7.0. |
| Buffer 8 | The 20mmol/L phosphate buffer, pH7.5. |
Show 2:37 ℃ of condition and place the electrophoresis purity measurement result of TPO plan peptide under the different buffer systems in back
| Specific activity | 0 day | 7 days | 14 days | 21 days | 28 days | 60 days |
| Buffer1 | 98.4 | Separate out deposition | — | ? — | — | — |
| Buffer2 | 99.1 | ?99.0 | ?98.0 | Separate out deposition | — | — |
| Buffer3 | 98.2 | ?98.4 | ?98.1 | ?97.2 | ?95.7 | Separate out deposition |
| Buffer4 | 99.3 | ?99.4 | ?99.2 | ?97.1 | ?96.4 | Separate out deposition |
| Buffer5 | 98.5 | ?98.7 | ?98.8 | ?97.5 | ?96.7 | Separate out deposition |
| Buffer6 | 98.3 | ?95.3 | ?80.3 | Separate out deposition | — | — |
| Buffer7 | 99.5 | Separate out deposition | — | — | — | — |
| Buffer8 | 98.1 | Separate out deposition | — | — | — | ? ?— |
Conclusion: from the result shown in the last table, can find out; It is bigger that pH value is intended the influence of peptide stock solution purity to thrombopoietin, and stock solution is separated out deposition very soon under the pH4.0 condition, when pH5.0~6.0 scopes; It is more stable that thrombopoietin is intended peptide stock solution, so select the buffer system of pH5.5; The buffer capacity of acetic acid-sodium acetate buffer system is better under this pH value condition.Therefore, we confirm that metastable buffer system is acetic acid-sodium acetate buffer system of 20mmol/L pH5.5.
Embodiment 2: add different pharmaceutical adjuncts are intended stabilized peptide property to thrombopoietin influence
The protein freeze drying protectant is the emphasis of biological agent research always, also is that protein is as the problem of medicinal application to clinical preceding necessary solution.The adjuvant that we use protective agent commonly used in the present protein medicaments has albumin, saccharide, amino acids, surfactant etc.The adjuvant that the present invention has selected some to be suitable for human use screens.Though the human albumin has good protein protection effect, because its source is limited and have potential safety hazard, so do not investigate.
Be applicable to that polyol of the present invention can select mannitol and sorbitol, preferred mannitol is as excipient.
Be applicable to polypeptide of the present invention, aminoacid from following one group of material, to select and come: glycine, arginine, alanine and proline etc., the preferred glycine of the present invention.
In order to investigate the influence of different auxiliary material, different auxiliary material is screened by following condition to stability of formulation.The accessory formula that the present invention screened sees Table that percent concentration is a mass percent concentration in the 3(table).Take by weighing the adjuvant of aequum, or measure the dense adjuvant stock solution for preparing.Adjuvant solid or adjuvant stock solution are joined in the suitable buffer (acetic acid-sodium-acetate buffer of pH5.5); Add the high concentration thrombopoietin of joining then and intend peptide protein solution with suitable buffer (acetic acid-sodium-acetate buffer of pH5.5); Reuse lmol/1 HCL or 10%Na0H are adjusted to required pH value, and the acetic acid-sodium-acetate buffer, the protein concentration that contain 20mmol/L pH5.0 with the acquisition certain volume are the sample solution of 1mg/ml.The sample solution for preparing is divided into two batches, and every batch has 10 kinds of samples, each 10 bottles in every kind of sample, and the 0.5ml/ bottle carries out lyophilizing.A collection of as initial T=0 analysis, then in 4 ℃ of preservations.37 ℃ of preservations of another batch are investigated 4 months, do analyzing and testing every month.Analysis project is a sample appearance after the lyophilizing, and non-reduced electrophoresis of SDS-PAGE and RP-HPLC detect (result sees table 4).
Table 3: adjuvant is formed table
| Group | Adjuvant | Prescription concentration |
| l | Sucrose | 1% |
| 2 | Sucrose | 2.5% |
| 3 | Sucrose | 5% |
| 4 | Mannitol | 5% |
| 5 | Glycine l | 1% |
| 6 | Glycine 2 | 2.5% |
| 7 | Glycine 3 | 5% |
| 8 | Tween 80 | 0.005% |
| 9 | Mannitol+glycine+Tween 80 | 5%,2.5%,0.005% |
| 10 | Mannitol+sucrose+Tween 80 | 5%,2.5%,0.005% |
Below be different formulations stability experiment result:
The outward appearance of table 4 sample
| Group | Sample appearance |
| l | Against regulation |
| 2 | Up to specification |
| 3 | Up to specification |
| 4 | Up to specification |
| 5 | Against regulation |
| 6 | Up to specification |
| 7 | Up to specification |
| 8 | Against regulation |
| 9 | Up to specification |
| 10 | Up to specification |
Sample after the biological product lyophilizing should be the loose body of white shell shape with reference to the relevant regulations in three ones of the Chinese Pharmacopoeias, adds to redissolve rapidly behind the labelled amount water for injection to be clear liquid.Table 4 shows that group 2,3,4,6,7,9,10 sample appearance are up to specification.
(1) electrophoresis impurity detects degraded and aggressiveness
Table 5: sample was deposited 4 months under 4 ℃ of conditions, detected with SDS-PAGE every other month
| Group | 0 month | January | February | March | April |
| 2 | Do not have | Do not have | Do not have | Do not have | Do not have |
| 3 | Do not have | Do not have | Do not have | Do not have | Do not have |
| 4 | Do not have | Do not have | Do not have | Do not have | Do not have |
| 6 | Do not have | Do not have | Do not have | Do not have | Do not have |
| 7 | Do not have | Do not have | Do not have | Do not have | Do not have |
| 9 | Do not have | Do not have | Do not have | Do not have | Do not have |
| 10 | Do not have | Do not have | Do not have | Do not have | Do not have |
Table 6: sample was deposited 4 months under 37 ℃ of conditions, detected with SDS-PAGE every other month
| Group | 0 month | January | February | March | April |
| 2 | Do not have | Have | Have | Have | Have |
| 3 | Do not have | Have | Have | Have | Have |
| 4 | Do not have | Have | Have | Have | Have |
| 6 | Do not have | Have | Have | Have | Have |
| 7 | Do not have | Have | Have | Have | Have |
| 9 | Do not have | Do not have | Do not have | Do not have | Have |
| 10 | Do not have | Do not have | Do not have | Do not have | Have |
Brief summary: we can find out from last table, and sample is deposited not degraded and aggressiveness generation in 4 months under 4 ℃ of conditions; Sample is under 37 ℃ of acceleration environments, and it is more stable relatively only to contain 9 and 10 sample, until degraded and aggressiveness generation were just arranged 4 months the time.
(3) RP-HPLC detects
Table 7: sample was deposited 4 months under 4 ℃ of conditions, detected with RP-HPLC every other month
| Group | 0 month | January | February | March | April |
| 2 | 99.5 | 98.7 | 97.4 | 96.3 | 95.5 |
| 3 | 99.3 | 99.2 | 97.5 | 97.5 | 96.6 |
| 4 | 99.4 | 99.1 | 98.0 | 98.5 | 96.3 |
| 6 | 98.9 | 99.0 | 97.6 | 97.3 | 97.8 |
| 7 | 99.5 | 98.7 | 98.2 | 98.5 | 96.1 |
| 9 | 99.3 | 99.0 | 98.9 | 98.1 | 99.2 |
| 10 | 99.1 | 99.0 | 98.9 | 98.0 | 99.7 |
Table 8: sample was deposited 4 months under 37 ℃ of conditions, detected with RP-HPLC every other month
| Group | 0 month | January | February | March | April |
| 2 | 99.7 | 98.9 | 95.4 | 90.3 | 77.5 |
| 3 | 99.5 | 99.5 | 96.5 | 89.5 | 70.6 |
| 4 | 99.3 | 99.2 | 96.1 | 90.5 | 75.6 |
| 6 | 98.9 | 99.0 | 94.6 | 87.3 | 79.8 |
| 7 | 99.5 | 98.7 | 94.2 | 80.5 | 75.6 |
| 9 | 99.3 | 99.0 | 98.9 | 98.1 | 95.2 |
| 10 | 99.1 | 99.0 | 98.9 | 98.0 | 95.7 |
We can find out from last table: sample is deposited 4 months internal stabilities under 4 ℃ of conditions better, and big variation does not take place sample purity; And under 37 ℃ of acceleration environments, have only the sample purity of the sample of group 9 and group 10 that big variation does not take place, the purity of other sample all has bigger reduction.
(4) BA (tiring)
Show that according to above studies result the sample that contains glycine or sucrose is the most stable, therefore only to the assessment of tiring of the sample of group 9 and group 10.Sample is deposited 4 months under 4 ℃ and 37 ℃ of conditions after respectively sampling carry out BA and detect.
Table 9: different prescription samples carry out BA at 4 ℃ after depositing 4 months with 37 ℃ of conditions and detect
| Group | Storage temperature | % tires |
| 9 | 4℃ | 102.2 |
| 9 | 37℃ | 85.3 |
| 10 | 4℃ | 105.2 |
| 10 | 37℃ | 89.5 |
Brief summary: we can find out from table 9: the tiring all in acceptability limit of all preparations.
Therefore select glycine+mannitol+Tween 80 or sucrose+mannitol+Tween 80 to intend the adjuvant of peptide lyophilized formulations as thrombopoietin.But sucrose does not have other authentication code of injection stage at home, can not be as the adjuvant of injection medicine, so the final adjuvant of selecting glycine+mannitol+Tween 80 to intend the peptide lyophilized formulations as thrombopoietin.
Comprehensive embodiment 1~2, it is ideal that following medicament is formed: the thrombopoietin of 0.1-1mg/ml is intended peptide protein, preferably 0.1-1mg/ml; The glycine of suitable concn, preferred concentrations are 2.5% (weight ratio).Selected buffer has phosphate buffer, Tris-Hcl buffer, acetic acid-sodium-acetate buffer, acetic acid-sodium-acetate buffer preferably, and concentration is 5-100mmol/L, and most preferred concentration is 20 mmol/L, and pH is between 5.0-6.0, and optimum is 5.5.The Tween 80 of suitable concn, preferred 0.005% (volume ratio).
Embodiment 3: contain the preparation that thrombopoietin is intended the lyophilized formulations of peptide
Get 500 milliliters of thrombopoietins and intend peptide stock solution; Protein concentration is 2 mg/ml; And contain 20mmol acetic acid-sodium-acetate buffer (pH 5.5); Take by weighing the 25g glycine and join in the stock solution dissolving fully, adding an amount of 20mmol acetic acid-sodium-acetate buffer (pH 5.5) dilution at last, to make the preparation final volume be 1000ml, said preparation carried out aseptic filtration and be sub-packed in the cillin bottle with the filter membrane of micron O.22 behind the mix homogeneously.Final preparation consists of: it is 1 mg/ml that thrombopoietin is intended peptide, and the concentration of acetic acid-sodium-acetate buffer is 20mmol/L (pH 5.5), and sweet oxygen acid content is 2.5% (weight ratio), and the content of Tween 80 is 0.005% (volume ratio).
Embodiment 4: contain the investigation that thrombopoietin is intended the lyophilized formulations stability of peptide.
According to embodiment 3 disclosed methods, prepare following several kinds of optimizing prescriptions:
Prescription 1: contain 0.5mg/ and prop up thrombopoietin plan peptide protein; Acetic acid-sodium-acetate buffer pH5.5 buffer; 2.5% glycine, the freeze-dried powder of 2.5% mannitol.
Prescription 2: contain 0.25mg/ and prop up thrombopoietin plan peptide protein; Acetic acid-sodium-acetate buffer pH5.5 buffer; 2.5% glycine, the freeze-dried powder of 2.5% mannitol.
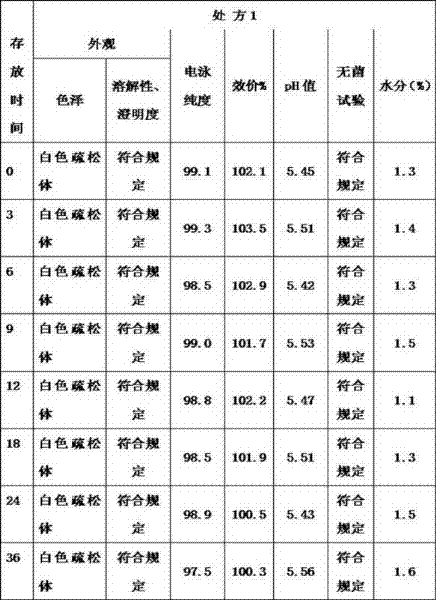
Solution according to above-mentioned 2 kinds of optimizing prescriptions preparation; Carry out long term test reference " three ones of Chinese Pharmacopoeias " appendix " medicine stability test guideline " respectively at depositing 36 months under the 2-8 ℃ of condition; Took a sample once in per 3 months, respectively at 0 month, 3 months, 6 months, 9 months, 12 months, sampling in 18 months, 24 months, 36 months detected; With result and comparison in 0 month, the result is carried out statistical analysis.Detect (outward appearance of finished product, dissolubility, clarity, BA, pH value, electrophoresis purity, sterility test and moisture) by stable high spot reviews project, with result and comparison in 0 month.The result sees table 10 and table 11
Table 10: thrombopoietin is intended peptide 0.5mg/ and is propped up the long-term stable experiment result
Table 11: recombinant platelet generates the plain peptide 0.25mg/ that intends and props up the long-term stable experiment result
Conclusion: prescription 1 and prescription 2 reference " three ones of Chinese Pharmacopoeias " appendix " medicine stability test guideline " are respectively at depositing 36 months under the 2-8 ℃ of condition; Sampling in per 3 months once; Respectively at 0 month, 3 months, 6 months, 9 months, 12 months; Sampling in 18 months, 24 months, 36 months detects, and each item index all meets the requirements, and effect duration can fix tentatively 3 years.
Claims (6)
1. a thrombopoietin is intended the peptide lyophilized formulations, it is characterized in that: said lyophilized formulations is dissolved in by thrombopoietin and pharmaceutic adjuvant in the medicinal buffer of pH5-6.0 and obtains, and said thrombopoietin plan peptide protein concentration is 0.1-1mg/ml; Said pharmaceutic adjuvant is mannitol, glycine and Tween 80, and the mannitol mass concentration is 2-8%, and the glycine mass concentration is 1-5%, and the Tween 80 volumetric concentration is 0.001%-0.01%.
2. a kind of thrombopoietin as claimed in claim 1 is intended the peptide lyophilized formulations, it is characterized in that: it is 0.5 mg/ml that said thrombopoietin is intended peptide protein concentration.
3. a kind of thrombopoietin as claimed in claim 1 is intended the peptide lyophilized formulations, and it is characterized in that: said mannitol mass concentration is 5%, and the glycine mass concentration is 2.5%, and the Tween 80 volumetric concentration is 0.005%.
4. intend the peptide lyophilized formulations like the arbitrary described a kind of thrombopoietin of claim 1-3, it is characterized in that: said buffer is phosphate buffer or citrate buffer solution or acetic acid-sodium-acetate buffer.
5. a kind of thrombopoietin as claimed in claim 4 is intended the peptide lyophilized formulations, and it is characterized in that: said buffer is acetic acid-sodium-acetate buffer, and concentration is 5~50mmol/L, preferred 20 mmol/L.
6. a kind of thrombopoietin as claimed in claim 5 is intended the peptide lyophilized formulations, and it is characterized in that: said buffer is acetic acid-sodium-acetate buffer, and concentration is 20 mmol/L.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2012100345402A CN102552184A (en) | 2012-02-16 | 2012-02-16 | Thrombopoietin peptide analogue freeze-dried preparation |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2012100345402A CN102552184A (en) | 2012-02-16 | 2012-02-16 | Thrombopoietin peptide analogue freeze-dried preparation |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN102552184A true CN102552184A (en) | 2012-07-11 |
Family
ID=46399652
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN2012100345402A Pending CN102552184A (en) | 2012-02-16 | 2012-02-16 | Thrombopoietin peptide analogue freeze-dried preparation |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN102552184A (en) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN110833514A (en) * | 2019-12-10 | 2020-02-25 | 广州赛莱拉干细胞科技股份有限公司 | Polypeptide freeze-dried powder and application thereof in preparation of anti-aging cosmetics |
| CN113527508A (en) * | 2020-04-17 | 2021-10-22 | 上海多米瑞生物技术有限公司 | A kind of preparation method of thrombopoietin peptidomimetic-Fc fusion protein |
| CN115429762A (en) * | 2022-10-13 | 2022-12-06 | 国药集团武汉血液制品有限公司 | Freeze-drying protective agent for high-concentration plasminogen preparation |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0841066A1 (en) * | 1995-06-08 | 1998-05-13 | Kirin Brewery Company, Ltd. | Stable freeze-dried composition containing tpo |
| CN1239480A (en) * | 1996-10-04 | 1999-12-22 | 安姆根有限公司 | Pharmaceutical compositions containing MPL ligands |
| CN101362797A (en) * | 2008-09-28 | 2009-02-11 | 华东师范大学 | A kind of thrombopoietin mimetic peptide, its preparation and application |
| EP2319928A1 (en) * | 1998-10-23 | 2011-05-11 | Kirin-Amgen, Inc. | Dimeric thrombopoietin peptide mimetics binding to MP1 receptor and having thrombopoietic activity |
-
2012
- 2012-02-16 CN CN2012100345402A patent/CN102552184A/en active Pending
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0841066A1 (en) * | 1995-06-08 | 1998-05-13 | Kirin Brewery Company, Ltd. | Stable freeze-dried composition containing tpo |
| CN1239480A (en) * | 1996-10-04 | 1999-12-22 | 安姆根有限公司 | Pharmaceutical compositions containing MPL ligands |
| EP2319928A1 (en) * | 1998-10-23 | 2011-05-11 | Kirin-Amgen, Inc. | Dimeric thrombopoietin peptide mimetics binding to MP1 receptor and having thrombopoietic activity |
| CN101362797A (en) * | 2008-09-28 | 2009-02-11 | 华东师范大学 | A kind of thrombopoietin mimetic peptide, its preparation and application |
Non-Patent Citations (1)
| Title |
|---|
| WEIWANG: "Lyophilization and development of solid protein pharmaceuticals", 《INTERNATIONAL JOURNAL OF PHARMACEUTICS》 * |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN110833514A (en) * | 2019-12-10 | 2020-02-25 | 广州赛莱拉干细胞科技股份有限公司 | Polypeptide freeze-dried powder and application thereof in preparation of anti-aging cosmetics |
| CN113527508A (en) * | 2020-04-17 | 2021-10-22 | 上海多米瑞生物技术有限公司 | A kind of preparation method of thrombopoietin peptidomimetic-Fc fusion protein |
| CN113527508B (en) * | 2020-04-17 | 2024-01-05 | 上海多米瑞生物技术有限公司 | Preparation method of thrombopoietin peptidomimetic-Fc fusion protein |
| CN115429762A (en) * | 2022-10-13 | 2022-12-06 | 国药集团武汉血液制品有限公司 | Freeze-drying protective agent for high-concentration plasminogen preparation |
| CN115429762B (en) * | 2022-10-13 | 2023-07-14 | 国药集团武汉血液制品有限公司 | Lyoprotectant for high concentration plasminogen preparations |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR101275949B1 (en) | Combinations and methods for subcutaneous administration of immune globulin and hyaluronidase | |
| EP3417875B1 (en) | Immunoglobulin formulation and method of preparation thereof | |
| ES2893861T3 (en) | Stable, aqueous antibody formulations | |
| EP2654784B1 (en) | Stable immunogenic compositions of staphylococcus aureus antigens | |
| EP2830658B1 (en) | Stable igg4 binding agent formulations | |
| JP2010174036A (en) | Complex of protein crystal and ionic polymer | |
| CN103599529A (en) | Multivalent pneumococcal polysaccharide-protein conjugate composition | |
| EP3639845B1 (en) | Il-15 protein complex pharmaceutical composition and uses thereof | |
| CN110787292B (en) | Programmed cell death receptor 1 antibody preparation and application thereof | |
| US20180207278A1 (en) | Liquid formulation comprising gm-csf neutralizing compound | |
| CN102552184A (en) | Thrombopoietin peptide analogue freeze-dried preparation | |
| CN102321168B (en) | Method for renaturing and purifying novel thrombopoietin | |
| CN101428035B (en) | Gemcitabine hydrochloride or gemcitabine composition | |
| CN101721700A (en) | Lyophilized preparation of anti-human Her2 antibody | |
| CN111375057A (en) | Pharmaceutical formulation comprising anti-Her 2 monoclonal antibody | |
| CN102614492A (en) | Liquid pharmaceutical composition containing echinocandin antifungal agent micafungin | |
| Ambrosino et al. | Effect of Haemophilus influenzae polysaccharide outer membrane protein complex conjugate vaccine on macrophages | |
| CN103505424B (en) | Preparation method for bortezomib for injection | |
| CN101716343A (en) | Freeze-drying preparation of monoclonal antibody | |
| Halford et al. | Hypersensitivity Rates in Pediatric Patients Receiving Intravenous or Intramuscular Asparaginase Agents. | |
| CN113082203B (en) | Liquid preparation of immunosuppressant monoclonal antibody | |
| CN101163716A (en) | Interleukin-6 polyethylene glycol conjugate and its preparing method and use | |
| CA2543484C (en) | Modulation of immune response to an immunogen with ctla-4 and tnfbp | |
| HK40119812A (en) | Taci-fc fusion protein liquid pharmaceutical preparation | |
| AU2006202688B2 (en) | Stable liquid formulations of antibodies |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C12 | Rejection of a patent application after its publication | ||
| RJ01 | Rejection of invention patent application after publication |
Application publication date: 20120711 |