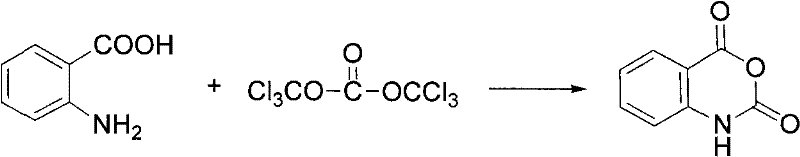CN102464592A - Acylbenzylamine compounds and their applications - Google Patents
Acylbenzylamine compounds and their applications Download PDFInfo
- Publication number
- CN102464592A CN102464592A CN2010105351330A CN201010535133A CN102464592A CN 102464592 A CN102464592 A CN 102464592A CN 2010105351330 A CN2010105351330 A CN 2010105351330A CN 201010535133 A CN201010535133 A CN 201010535133A CN 102464592 A CN102464592 A CN 102464592A
- Authority
- CN
- China
- Prior art keywords
- radical
- halo
- alkyl
- unsubstituted
- alkoxy
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 0 CCC1=CC=C[C@@]2C1=C2* Chemical compound CCC1=CC=C[C@@]2C1=C2* 0.000 description 10
- RWZYAGGXGHYGMB-UHFFFAOYSA-N Nc1ccccc1C(O)=O Chemical compound Nc1ccccc1C(O)=O RWZYAGGXGHYGMB-UHFFFAOYSA-N 0.000 description 1
- TXJUTRJFNRYTHH-UHFFFAOYSA-N O=C(c(cccc1)c1N1)OC1=O Chemical compound O=C(c(cccc1)c1N1)OC1=O TXJUTRJFNRYTHH-UHFFFAOYSA-N 0.000 description 1
Landscapes
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Pyridine Compounds (AREA)
Abstract
The invention discloses an acyl benzylamine compound with broad-spectrum bactericidal activity, which has a structure shown as a general formula :
Description
Technical Field
The invention belongs to the field of agricultural bactericides, and particularly relates to an acyl benzylamine compound and application thereof.
Background
The germs cause great damage to crops, especially to agricultural products such as grains, fibers and the like, such as cotton, rice, corn, wheat, soybean and the like, which can meet the basic requirements of human beings. Killing or inhibiting the growth of pathogens while avoiding or reducing damage to the crop is an effective way to improve agricultural production. There is therefore a continuing need to develop more effective novel fungicides. Acyl benzylamine compounds such as tolfenpyrad (US 5039693), tebufenpyrad (US 4950668), CN1927860 and the like are frequently used as insecticidal and acaricidal agents, and CN1919838 and WO2002083647 report that the compounds have insecticidal and bactericidal activity, but the use of acyl benzylamine compounds with the structural general formula as bactericides is not reported in documents.
Disclosure of Invention
In order to meet the agricultural requirements, the invention provides a novel acyl benzylamine compound and application thereof in preventing and controlling germs in agriculture.
The technical scheme of the invention is as follows: (the claims are partly amended and made together)
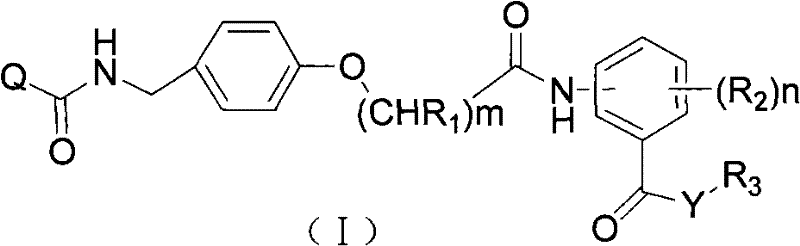
The invention provides an acyl benzylamine compound, which is shown as a general formula (I):
in the formula:
R1selected from hydrogen or C1-C12An alkyl group; m is an integer selected from 1 to 3;
R2selected from halogen, cyano, nitro, C1-C12Alkyl, halo C1-C12Alkyl radical, C1-C12Alkoxy, halo C1-C12Alkoxy radical, C1-C12Alkylthio radical, C1-C12Alkylsulfonyl or R5(ii) a n is an integer from 0 to 4;
y is selected from O,S or NR4;
R3、R4Can be the same or different and are respectively selected from hydrogen, cyano and C1-C12Alkyl, halo C1-C12Alkyl radical, C1-C12Alkoxy, halo C1-C12Alkoxy radical, C3-C6Cycloalkyl, cyano C1-C12Alkyl radical, C1-C12Alkylamino, halogeno C1-C12Alkylamino radical, C2-C6Dialkylamino radical, C2-C12Alkenyl, halo C2-C12Alkenyl radical, C2-C12Alkenyloxy, halogeno C2-C12Alkenyloxy radical, C2-C12Alkynyl, halo C2-C12Alkynyl, C2-C12Alkynyloxy, halo C2-C12Alkynyloxy, C1-C12Alkylthio, halo C1-C12Alkylthio radical, C1-C12Alkylsulfonyl radical, C1-C12Alkylcarbonyl, halo C1-C12Alkylcarbonyl, halo C1-C12Alkylsulfonyl or R5(ii) a Or,
when Y is selected from NR4N R4And R3Form a substituted or unsubstituted five-or six-membered ring, the substituents being selected from 1 to 2C1-C4An alkyl group;
R5selected from phenyl, benzyl or heteroaryl, unsubstituted or substituted with 1 to 4 groups independently selected from: halogen, nitro, cyano, C1-C12Alkyl, halo C1-C12Alkyl radical, C1-C12Alkoxy, halo C1-C12Alkoxy radical, C1-C12Alkylthio radical, C1-C12Alkylcarbonyl or C1-C12An alkoxycarbonyl group;
(CHR1) The connecting position of mCONH and a benzene ring is selected from 2, 3 or 4 positions;
q is selected from C unsubstituted or substituted by 1 to 41-C6Alkyl, cyclopropyl or cyclohexyl: cl, Br, F or C1-C4An alkyl group;
or selected from anilino or C, unsubstituted or substituted by 1-32-C4Alkenyl: cl, Br, F, C1-C4Alkyl radical, C3-C6Cycloalkyl radical, C1-C4Haloalkyl, C1-C4Alkoxy radical, C1-C4Haloalkoxy or C1-C4An alkoxycarbonyl group;
or selected from phenyl, phenoxyethyl, phenoxypropyl, pyridyl, pyrimidinyl, piperidinyl, piperazinyl, thiazolyl, isothiazolyl, thiadiazolyl, oxazolyl, isoxazolyl, oxadiazolyl, triazolyl, thienyl, furanyl, quinolinyl, indolyl, 3-pyrazolyl, 4-pyrazolyl or 4-pyrazolylmethyl, unsubstituted or substituted with 1-3 of the following groups: halogen, CN, NO2、C1-C4Alkyl radical, C3-C6Cycloalkyl radical, C1-C3Haloalkyl, C1-C3Alkoxyalkyl group, C1-C3Alkoxy radical, C1-C3Haloalkoxy, COR6、CO2R6、CON R6R7、NR6R7、NR7COR6、NR7CO2R6、SR7、SOR7、SO2R7、SO2NR6R7Or R7;
R6Is selected from H or C1-C4An alkyl group;
R7selected from H, C1-C4Alkyl radical, C1-C3Halogenated alkyl, substituted or unsubstituted phenyl, the substituent being selected from Cl, Br, F, CN, NO2、C1-C4Alkyl, CF3、OCH3、OCF3Or CO2CH3。
Preferred compounds of the invention are: in the general formula (I)
R1Selected from hydrogen or C1-C6An alkyl group; m is an integer selected from 1 to 3;
R2selected from halogen, cyano, nitro, C1-C6Alkyl, halo C1-C6Alkyl radical, C1-C6Alkoxy, halo C1-C6Alkoxy radical, C1-C6Alkylthio radical, C1-C6Alkylsulfonyl or R5(ii) a n is an integer from 0 to 4;
y is selected from O, S or NR4;
R3、R4Can be the same or different and are respectively selected from hydrogen, cyano and C1-C6Alkyl, halo C1-C6Alkyl radical, C1-C6Alkoxy, halo C1-C6Alkoxy radical, C3-C6Cycloalkyl, cyano C1-C6Alkyl radical, C1-C6Alkylamino, halogeno C1-C6Alkylamino radical, C2-C6Dialkylamino radical, C3-C6Alkenyl, halo C3-C6Alkenyl radical, C3-C6Alkenyloxy, halogeno C3-C6Alkenyloxy radical, C3-C6Alkynyl, halo C3-C6Alkynyl, C3-C6Alkynyloxy, C1-C6Alkylthio, halo C1-C6Alkylthio radical, C1-C6Alkylsulfonyl radical, C1-C6Alkylcarbonyl, halo C1-C6Alkylcarbonyl, halo C1-C6Alkylsulfonyl or R5(ii) a Or,
when Y is selected from NR4N R4And R3To form morpholine, piperazine, piperidine, pyrazole, pyrrole or triazole ring;
R5selected from phenyl, benzyl, pyridyl, pyrimidinyl, thiazolyl, isothiazolyl, oxazolyl, isoxazolyl, oxadiazolyl, thiadiazolyl, pyrazolyl, pyranyl, triazolyl or tetrazolyl, unsubstituted or substituted with 1 to 4 groups independently selected from: halogen, nitro, cyano, C1-C6Alkyl, halo C1-C6Alkyl radical, C1-C6Alkoxy, halo C1-C6Alkoxy radical, C1-C6Alkylthio radical, C1-C6Alkylcarbonyl or C1-C6An alkoxycarbonyl group;
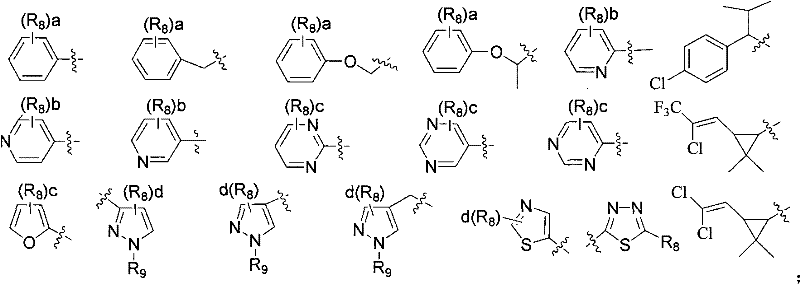
q is selected from one of the groups shown below:
R8selected from H, Cl, Br, F, CN, NO2、NH2、CH3、CH2CH3Tert-butyl, cyclopropyl, CF3、CH2CF3、OCH3、OCF3、OCH2CF3、SO2CH3、CO2CH3、C1-C3Alkylamino carbonyl, C2-C4Dialkylaminocarbonyl, unsubstituted or substituted phenyl, unsubstituted or substituted pyridyl, said substituents being selected from 1 to 3 of the following groups: cl, Br, F, CN, NO2、CH3、CH2CH3Tert-butyl, cyclopropyl, CHF2、CF3、CH2CF3、OCH3、OCHF2、OCF3、OCH2CF3Or SO2CH3;
R9Selected from H, CH3、CH2CH3Tert-butyl, CF3、CH2CF3Unsubstituted or substituted phenyl, unsubstituted or substituted pyridyl, said substituents being selected from 1 to 3 of the following groups: cl, Br, F, CN, NO2、CH3、CH2CH3Tert-butyl, cyclopropyl, CHF2、CF3、CH2CF3、OCH3、OCHF2、OCF3、OCH2CF3Or SO2CH3;
a is selected from an integer of 1 to 5; b is selected from an integer of 1 to 4; c is an integer selected from 1 to 3; d is selected from an integer from 1 to 2.
Further preferred compounds of the invention are: in the general formula (I)
R1Selected from hydrogen or C1-C3An alkyl group; m is an integer selected from 1 to 3;
R2selected from halogen, cyano, nitro, C1-C4Alkyl, halo C1-C4Alkyl radical, C1-C4Alkoxy, halo C1-C4Alkoxy radical, C1-C3Alkylthio radical, C1-C4Alkylsulfonyl or R5(ii) a n is an integer from 0 to 3;
y is selected from O, S or NR4;
R3、R4Can be the same or different and are respectively selected from hydrogen, cyano and C1-C6Alkyl, halo C1-C6Alkyl radical, C1-C6Alkoxy, halo C1-C6Alkoxy radical, C3-C6Cycloalkyl, cyano C1-C6Alkyl radical, C1-C6Alkylamino, halogeno C1-C6Alkylamino radical, C2-C6Dialkylamino radical, C3-C6Alkenyl, halo C3-C6Alkenyl radical, C3-C6Alkynyl, halo C3-C6Alkynyl, C1-C6Alkylthio, halo C1-C6Alkylthio radical, C1-C6Alkylsulfonyl radical, C1-C6Alkylcarbonyl, halo C1-C6Alkylcarbonyl, halo C1-C6Alkylsulfonyl or R5(ii) a Or,
when Y is selected from NR4N R4And R3To form a morpholine, piperazine, piperidine or pyrazole ring;
R5selected from phenyl, benzyl, pyridyl, pyrimidyl, thiazolyl, oxazolyl, oxadiazolyl, thiadiazolyl, pyrazolyl, triazolyl or tetrazolyl, unsubstituted or substituted with 1-4 groups independently selected from: halogen, nitro, cyano, C1-C4Alkyl, halo C1-C4Alkyl radical, C1-C4Alkoxy, halo C1-C4Alkoxy radical, C1-C4Alkylthio radical, C1-C4Alkylcarbonyl or C1-C4An alkoxycarbonyl group;
q is selected from one of the groups shown below:
R8selected from H, Cl, Br, F, CN, NO2、CH3、CH2CH3Tert-butyl, cyclopropyl, CF3、CH2CF3、OCH3、OCF3、OCH2CF3、SO2CH3、CO2CH3Unsubstituted or substituted phenyl, unsubstituted or substituted pyridyl, said substituents being selected from 1 to 3 of the following groups: cl, Br, F, CN, NO2、CH3、CH2CH3Tert-butyl, cyclopropyl, CHF2、CF3、CH2CF3、OCH3、OCHF2、OCF3、OCH2CF3Or SO2CH3;
R9Selected from H, CH3、CH2CH3Tert-butyl, unsubstituted or substituted phenyl, unsubstituted or substituted pyridyl, said substituents being selected from 1 to 3 of the following groups: cl, Br, F, CN, NO2、CH3、CH2CH3Tert-butyl, cyclopropyl, CF3、CH2CF3、OCH3、、OCF3Or SO2CH3;
a is selected from an integer of 1 to 5; b is selected from an integer of 1 to 4; c is an integer selected from 1 to 3; d is selected from an integer from 1 to 2.
Further preferred compounds of the invention are: in the general formula (I)
R1Selected from hydrogen or methyl; m is an integer selected from 1 to 3;
R2selected from fluorine, chlorine, bromine, iodine, cyano, nitro, C1-C4Alkyl or halo C1-C4An alkyl group; n is an integer from 0 to 3;
y is selected from O or NR4;
R3、R4Can be the same or different and are respectively selected from hydrogen, cyano and C1-C5Alkyl, halo C1-C5Alkyl radical, C1-C5Alkoxy, halo C1-C5Alkoxy radical, C3-C6Cycloalkyl, cyano C1-C5Alkyl radical, C1-C5Alkylamino, halogeno C1-C5Alkylamino radical, C2-C6Dialkylamino radical, C3-C5Alkenyl, halo C3-C5Alkenyl radical, C3-C5Alkynyl, halo C3-C5Alkynyl, C1-C5Alkylsulfonyl radical, C1-C5Alkylcarbonyl, halo C1-C5Alkylcarbonyl, halo C1-C5Alkylsulfonyl or R5(ii) a Or,
when Y is selected from NR4N R4And R3To form a morpholine, piperazine or piperidine ring;
R5selected from phenyl, benzyl, pyridyl, pyrimidyl, thiazolyl, oxazolyl or pyrazolyl, unsubstituted or substituted with 1-3 groups independently selected from: fluorine, chlorine, bromine, nitro, cyano, C1-C3Alkyl, halo C1-C3Alkyl radical, C1-C3Alkoxy or halo C1-C3An alkoxy group;
q is selected from one of the groups shown below:
R8selected from H, Cl, Br, F, CN, CH3、CH2CH3Tert-butyl, cyclopropyl, CF3、OCH3、OCF3、OCH2CF3、SO2CH3Unsubstituted or substituted phenyl, unsubstituted or substituted pyridyl, said substituents being selected from 1 to 3 of the following groups: cl, Br, F, CN, NO2、CH3、CH2CH3Tert-butyl, CHF2、CF3、OCH3、OCHF2、OCF3、OCH2CF3Or SO2CH3;
R9Selected from H, CH3、CH2CH3Tert-butyl, unsubstituted or substituted phenyl, unsubstituted or substituted pyridyl, said substituents being selected from 1 to 3 of the following groups: cl, Br, F, CN, NO2、CH3Or CF3;
a is selected from an integer of 1 to 4; b is an integer selected from 1 to 3; c is an integer selected from 1 to 2; d is selected from an integer from 1 to 2.
Still further preferred compounds of the invention are: in the general formula (I)
R1Selected from hydrogen or methyl; m is an integer selected from 1 to 2;
R2selected from fluorine, chlorine, bromine, iodine, cyano, nitro, C1-C3Alkyl or halo C1-C3An alkyl group; n is an integer from 0 to 2;
y is selected from O or NR4;
R3、R4Can be the same or different and are respectively selected from hydrogen and C1-C5Alkyl radical, C1-C5Alkoxy, cyano C1-C5Alkyl radical, C1-C5Alkylamino radical, C2-C6Dialkylamino radical, C3-C5Alkenyl radical, C3-C5Alkynyl, C1-C5Alkylsulfonyl radical, C1-C5Alkylcarbonyl or R5(ii) a Or,
when Y is selected from NR4N R4And R3To form a morpholine, piperazine or piperidine ring;
R5selected from phenyl, benzyl, pyridyl, pyrimidinyl, thiazolyl or pyrazolyl, unsubstituted or substituted with 1-2 groups independently selected from: fluorine, chlorine, bromine, nitro, cyano, C1-C3Alkyl, halo C1-C3Alkyl radical, C1-C3Alkoxy or halo C1-C3An alkoxy group;
q is selected from one of the groups shown below:
R8selected from H, Cl, Br, F, CN, CH3、CH2CH3Tert-butyl, CF3、OCH3、OCF3、SO2CH3Unsubstituted or substituted phenyl, unsubstituted or substituted pyridyl, said substituents being selected from 1 to 3 of the following groups: cl, Br, F, CN, NO2、CH3、CH2CH3Tert-butyl, CHF2、CF3、OCH3、OCHF2、OCF3Or SO2CH3;
R9Selected from H, CH3、CH2CH3Tert-butyl, unsubstituted or substituted phenyl, unsubstituted or substituted pyridyl, said substituents being selected from 1 to 3 of the following groups: cl, Br, F, CH3Or CF3;
a is selected from an integer of 1 to 3; b is an integer selected from 1 to 2; c is an integer selected from 1 to 2; d is selected from an integer from 1 to 2.
More preferred compounds of the invention are: in the general formula (I)
R1Selected from hydrogen or methyl; m is selected from 1;
R2selected from fluorine, chlorine, bromine or C1-C3An alkyl group; n is an integer from 0 to 2;
y is selected from O or NR4;
R3、R4May be the same or different and are each selected from hydrogen or C1-C3An alkyl group;
R5selected from phenyl, benzyl, pyridine or pyrimidinyl, unsubstituted or substituted with 1-2 groups independently selected from: fluorine, chlorine, bromine, nitro, cyano, C1-C3Alkyl, halo C1-C3Alkyl radical, C1-C3Alkoxy or halo C1-C3An alkoxy group;
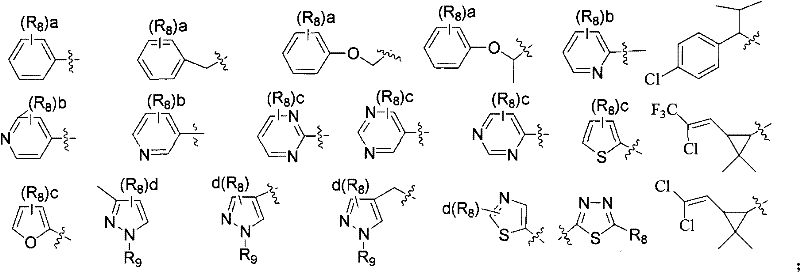
q is selected from one of the groups shown below:
R8selected from H, Cl, Br, F, CN, CH3、CH2CH3、CF3、OCH3、OCF3、SO2CH3Unsubstituted or substituted phenyl, unsubstituted or substituted pyridyl, said substituents being selected from 1 to 3 of the following groups: cl, Br, F, NO2、CH3、CH2CH3、CF3Or OCH3;
R9Selected from H, CH3Unsubstituted or substituted phenyl, unsubstituted or substituted pyridyl, said substituents being selected from 1 to 3 of the following groups: cl, Br, F or CH3。
In the definitions of the compounds of the general formula (I) given above, the terms used are generally defined as follows:
halogen: refers to fluorine, chlorine, bromine or iodine.
Alkyl groups: straight-chain or branched alkyl groups, such as methyl, ethyl, propyl, isopropyl or tert-butyl.
Halogenated alkyl groups: straight-chain or branched haloalkyl groups in which the hydrogen atoms may be partially or wholly replaced by halogen atoms, for example, haloalkyl groups such as chloromethyl, dichloromethyl, trichloromethyl, fluoromethyl, difluoromethyl or trifluoromethyl.
Alkoxy groups: straight-chain, branched-chain or cyclic alkoxy, for example methoxy, ethoxy, propoxy, isopropoxy, tert-butoxy or cyclopropoxy.
Haloalkoxy groups: straight-chain or branched haloalkoxy groups in which the hydrogen atoms may be partially or fully substituted by halogen atoms, for example, haloalkyl groups such as chloromethoxy, dichloromethoxy, trichloromethoxy, fluoromethoxy, difluoromethoxy or trifluoromethoxy groups.
Alkylthio group: straight or branched chain alkyl groups attached to the structure via a sulfur atom.
Haloalkylthio: straight-chain or branched alkylthio groups in which the hydrogen atoms may be partially or fully substituted by halogen atoms. For example, chloromethylthio, dichloromethylthio, trichloromethylthio, fluoromethylthio, difluoromethylthio, trifluoromethylthio, chlorofluoromethylthio and the like.
Alkenyl: straight-chain or branched and may have a double bond in any position, for example a vinyl or allyl group.
Alkynyl: straight or branched chain and may have a triple bond at any position, for example ethynyl or propargyl.
The heteroaryl group referred to in the present invention is a five-membered or six-membered ring aryl group containing 1 or more heteroatoms of N, O, S. For example, pyridyl, pyrimidyl, thiazolyl, isothiazolyl, oxazolyl, isoxazolyl, oxadiazolyl, thiadiazolyl, pyrazolyl, pyranyl, triazolyl or tetrazolyl groups and the like.
The technical scheme of the invention also comprises an intermediate phenylbenzylamine or a salt thereof for preparing the compound shown in the general formula (I), wherein the intermediate phenylbenzylamine or the salt thereof has a novel structure and is shown in the general formula (II):
in the formula:
R1selected from hydrogen or C1-C12An alkyl group; m is an integer selected from 1 to 3;
R2selected from halogen, cyano, nitro, C1-C12Alkyl, halo C1-C12Alkyl radical, C1-C12Alkoxy, halo C1-C12Alkoxy radical, C1-C12Alkylthio radical, C1-C12Alkylsulfonyl or R5(ii) a n is an integer from 0 to 4;
y is selected from O, S or NR4;
R3、R4Can be the same or different and are respectively selected from hydrogen, cyano and C1-C12Alkyl, halo C1-C12Alkyl radical, C1-C12Alkoxy, halo C1-C12Alkoxy radical, C3-C6Cycloalkyl, cyano C1-C12Alkyl radical, C1-C12Alkylamino, halogeno C1-C12Alkylamino radical, C2-C6Dialkylamino radical, C2-C12Alkenyl, halo C2-C12Alkenyl radical, C2-C12Alkenyloxy, halogeno C2-C12Alkenyloxy radical, C2-C12Alkynyl, halo C2-C12Alkynyl, C2-C12Alkynyloxy, halo C2-C12Alkynyloxy, C1-C12Alkylthio, halo C1-C12Alkylthio radical, C1-C12Alkylsulfonyl radical, C1-C12Alkylcarbonyl, halo C1-C12Alkylcarbonyl, halo C1-C12Alkylsulfonyl or R5(ii) a Or,
when Y is selected from NR4N R4And R3Form a substituted or unsubstituted five-or six-membered ring, the substituents being selected from 1 to 2C1-C4An alkyl group;
R5selected from phenyl, benzyl or heteroaryl, unsubstituted or substituted with 1 to 4 groups independently selected from: halogen, nitro, cyano, C1-C12Alkyl, halo C1-C12Alkyl radical, C1-C12Alkoxy, halo C1-C12Alkoxy radical, C1-C12Alkylthio radical, C1-C12Alkylcarbonyl or C1-C12An alkoxycarbonyl group;
(CHR1) The connection position of mCONH and a benzene ring is selected from 2, 3 or 4 positions.
The acid which can be salified with the phenylbenzylamine represented by the general formula (II) of the present invention includes: carboxylic acids such as acetic acid, propionic acid, butyric acid, oxalic acid, adipic acid, dodecanedioic acid, lauric acid, stearic acid, trifluoroacetic acid, fumaric acid, maleic acid, benzoic acid, or phthalic acid; sulfonic acids such as methanesulfonic acid, 1, 3-propanedisulfonic acid, p-toluenesulfonic acid or dodecylbenzenesulfonic acid; and inorganic acids such as hydrochloric acid, sulfuric acid, nitric acid, or carbonic acid.
(CHR) in the Compound of the general formula (I) of the present invention1) The connecting position of mCONH and the benzene ring can be selected from 2, 3 or 4 positions of the benzene ring.
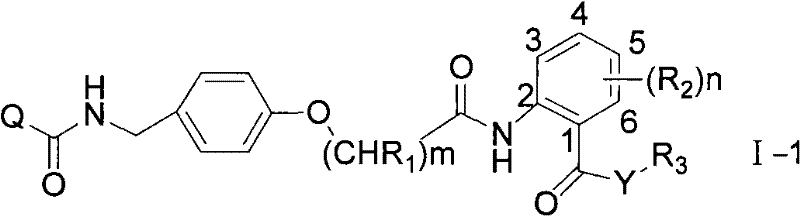
When attached to the 2-position of the phenyl ring, the structural formula is shown as I-1:
when the compound is connected with the 3-position of a benzene ring, the structural formula is shown as I-2;
when the structure is connected with the 4-position of the benzene ring, the structural formula is shown as I-3;
in the compounds of general formula (I) of the present invention, stereoisomers (different configurations are represented by R and S, respectively) may be formed due to the attachment of different groups or substituents to the chiral carbon or nitrogen. The compounds of the general formula (I) according to the invention therefore include the R or S isomer, as well as mixtures in any ratio.
The detailed structure of part of the group Q in the compound of the general formula I is shown in the table 1; when CHR is present1) When the connection position of mCONH and the benzene ring is 2 position, R in the benzene ring2Specific substituents are shown in table 2; when (CHR)1) When the connection position of mCONH and benzene ring is 3 position, R2Specific substituents are shown in table 3; when (CHR)1) When the connection position of mCONH and benzene ring is 4, R2See table 4 for specific substituents; Y-R3Specific substituents are shown in table 5; other substituents in the general formula (I) are, for example, R1、R3、R4、R5M and n are as defined above.
TABLE 1
TABLE 2
| R2 | R2 | R2 | R2 | R2 |
| 3-I | 3-Br | 6-OCH3 | 3-CH3-5-NO2 | 6-CH3-3,5-2Br |
| 3-CH3 | 4-Br | 5-OCH3 | 4-CH3-3-NO2 | 3-CF3O-4,6-2Cl |
| 4-CH3 | 5-Br | 3,5-2Cl | 4-CH3-5-NO2 | 4-CH3-5-NO2-3-Br |
| 5-CH3 | 6-Br | 3,5-2Br | 5-CH3-3-NO2 | 3-CN-4,6-2Cl |
| 6-CH3 | 5-I | 4-CH3-5-Br | 6-CH3-4-NO2 | 3-CN-4-CH3-6-Cl |
| 3-Cl | 5-F | 6-CH3-5-CN | 6-CH3-5-NO2 | 3-CN-4-CF3-6-Cl |
| 4-Cl | 6-F | 3,5,6-3Cl | 3-NO2-5-Cl | 4-CH3-5-CN-6-Cl |
| 5-Cl | 3-CN | 3-CH2CH3 | 3-NO2-5-Br | 4-CF3-5-CN-6-Cl |
| 6-Cl | 4-CN | 4-CH2CH3 | 5-NO2-3-Br | 3-CF3O-6-Cl |
| 3-CF3 | 5-CN | 5-CH2CH3 | 5-CH3-3-Br | 5-CN-3-Cl |
| 4-CF3 | 6-CN | 6-CH2CH3 | 6-CH3-5-Br | 5-CF3-3,6-2Cl |
| 5-CF3 | 3-NO2 | 5-CF3-3-Cl | 3-CH3-5-Br | 5-CF3-6-Cl |
| 6-CF3 | 5-NO2 | 5-CH3-3-Cl | 3-CF3-6-Cl | 3-CN-6-Cl |
| 6-I | 3-F | 4-F | 4-NO2 | 6-NO2 |
TABLE 3
| R2 | R2 | R2 | R2 | R2 |
| 2-I | 2-Br | 6-OCH3 | 4-CF3 | 6-CH3-4,5-2Br |
| 2-CH3 | 4-Br | 5-OCH3 | 4-CH3-6-NO2 | 2-NO2 |
| 4-CH3 | 5-Br | 2,5-2Cl | 6-CH3-4-NO2 | 5-CN-4,6-2Cl |
| 5-CH3 | 6-Br | 2,5-2Br | 5-CH3-5-Br | 6-CF3 |
| 6-CH3 | 5-I | 4-CH3-5-Br | 6-CH3-4Br | 5-CN-4-Cl |
| 2-Cl | 5-F | 6-CH3-5-CN | 4-CH3-5-Br | 5-CF3-6-Cl |
| 4-Cl | 6-F | 4,5,6-3Cl | 4-CF3-6-Cl | 2-CN-6-Cl |
| 5-Cl | 2-CN | 2-CH2CH3 | 5-CH3-2-Cl | 4-CH3-2-Cl |
| 6-Cl | 4-CN | 4-CH2CH3 | 4,5-2Cl | 5-CH3-4-Cl |
| 2-CF3 | 5-CN | 5-NO2 | 5-CF3 | 6-CN |
TABLE 4
| R2 | R2 | R2 | R2 | R2 |
| 2-F | 2-C(CH3)2 | 3-CH3 | 2,5-2CH3 | 2-Cl-5-F |
| 3-F | 3-C(CH3)2 | 2,3-2CN | 3,6-2CH3 | 2-Cl-5-Br |
| 5-F | 5-C(CH3)2 | 5,6-2CN | 5-CH3-2-Br | 5-Cl-2-Br |
| 6-F | 2-CF3 | 2,6-2CN | 5-CH3-2-Cl | 2,5-2Cl |
| 2-Cl | 3-CF3 | 3,6-2CN | 2,6-2Br | 5-CH3-3-F |
| 3-Cl | 5-CF3 | 2-F-5-Br | 3,6-2Br | 6-CH3-3-Cl |
| 5-Cl | 6-CF3 | 2-F-6-Cl | 2-CH3-5-F | 2-CH3-3-Br |
| 6-Cl | 2-OCH3 | 2-CN-5-Cl | 5-CH3-2-CN | 2-CH3-3-F |
| 2-Br | 3-OCH3 | 2-CN-5-Br | 2-CN | 2-CH3-3-Cl |
| 3-Br | 5-OCH3 | 5-CN-2-Cl | 3-CN | 2-CH3-5-F |
| 5-Br | 3-OCF3 | 5-CH3-2-F | 5-CN | 2-CH3-5-Cl |
| 6-Br | 5-OCF3 | 2,3-2Br | 6-CN | 3-CH3-2-Br |
| 2-I | 2,3-2F | 6-CH3 | 2-NO2 | 3-CH3-5-Cl |
| 3-I | 5,6-2F | 2-C2H5 | 3-NO2 | 3-CH3-5-Br |
| 5-I | 3,5-2F | 5,6-2Cl | 5-NO2 | 3-CH3-5-I |
| 6-NO2 | 2,3-2Cl | 2-CH3 | 6-I | 2-CH3-5-I |
| 3,6-2Cl | 2,6-2Cl | 5-CH3 | 3,5-2Cl |
TABLE 5
Some preferred compounds may be exemplified by the specific compounds listed in tables 6, 7, and 8, but are not intended to limit the present invention.
TABLE 6
Abbreviations Bn, Py and Ph in the above tables represent benzyl, pyridyl and phenyl, respectively, and Table 6R2In the column, "-" denotes no substituent, i.e.the corresponding position on the phenyl ring is hydrogen.
TABLE 7
The compound number 194-386 in Table 7 (omitted), and each of the other groups R in the table1、R2、Y-R3、R4And m and n are the same as those of compounds No. 1 to 193 in Table 6 in the order named.
TABLE 8
The compound in Table 8 (omitted) is 387-579, and the other groups R in the table1、R2、Y-R3、R4And m and n are the same as those of compounds No. 1 to 193 in Table 6 in the order named.
The compounds of general formula (I) according to the invention can be prepared according to the following method:
according to the above reaction equation, the acid chloride (III) and the phenylbenzylamine (II) are reacted in the presence or absence of a base, preferably using a solvent at 0 to 100 ℃, preferably at 0 to 25 ℃, for 30 minutes to 20 hours, preferably 0.5 to 10 hours. The solvent is a solvent having no direct influence on the reaction, and includes aromatic hydrocarbons such as benzene, toluene and xylene; ketones, such as acetone methyl ethyl ketone and methyl isobutyl ketone; halogenated hydrocarbons such as dichloromethane, chloroform and dichloroethane; lipids, such as methyl acetate and ethyl acetate; and polar solvents such as tetrahydrofuran, acetonitrile, dioxane, N-dimethylformamide, N-methylpyrrolidone, dimethylsulfoxide and pyridine.
The base includes metal hydrides such as sodium hydride; alkali metal hydroxides such as sodium hydroxide and potassium hydroxide; alkali metal carbonates such as sodium carbonate and potassium carbonate; organic amines such as pyridine and triethylamine.
In order to isolate the target compound of (I) after the reaction, in the case of using a solvent soluble in water, the solvent is distilled off under reduced pressure, water is added to the resulting residue, and the residue is purified by using an aromatic hydrocarbon insoluble in water, such as benzene, toluene and xylene; halogenated alkanes such as chloroform and dichloromethane; and esters such as ethyl acetate, and washing the extract with saturated aqueous sodium chloride solution, drying the obtained extract with a drying agent such as anhydrous magnesium sulfate or anhydrous sodium sulfate, and then removing the solvent under reduced pressure. When a water-insoluble solvent is used, water is added to the reaction mixture, separation is carried out with shaking, the resulting organic layer is washed with a saturated aqueous sodium chloride solution, dried over a drying agent such as anhydrous magnesium sulfate or anhydrous sodium sulfate, and then the solvent is removed under reduced pressure. The resulting residue was purified by recrystallization, washing and column chromatography to give the objective compound represented by the general formula (I).
As an intermediate for synthesizing the target compound represented by the general formula (I), an acid chloride represented by the general formula (III) was synthesized according to the method described in JP 4069379.
The benzyl benzylamine compound shown in the general formula (II) is a key intermediate for preparing the compound shown in the general formula (I), the structure of the benzyl benzylamine compound is not reported before, and the benzyl benzylamine compound can be prepared by the following method:
(1) preparation of phenyl benzylamine
Wherein R is1、R2、R3、R4、R5M and n have the same meanings as defined in the general formula (I).
Reacting the benzonitrile (IV) with hydrogen in the presence of a metal catalyst and aqueous ammonia, preferably using an organic solvent, at a temperature of 0 to 100 ℃, preferably 20 to 50 ℃; the reaction time is 30 minutes to 20 hours, preferably 0.5 to 10 hours.
Any organic solvent may be used as the organic solvent for the reaction as long as it has no direct influence on the reaction. The organic solvent includes alcohols such as methanol, ethanol, and isopropanol; aromatic hydrocarbons, such as benzene. Toluene and xylene; ketones such as acetone, methyl ethyl ketone and methyl isobutyl ketone; halogenated hydrocarbons such as chloroform and dichloromethane; water; esters, such as methyl acetate and ethyl acetate; or polar solvents such as tetrahydrofuran, acetonitrile, dioxane, N-dimethylformamide, N-methylpyrrolidone, dimethylsulfoxide and pyridine.
The metal catalyst includes raney nickel, palladium on carbon, and platinum oxide.
In order to isolate the target compound of formula (II) after the reaction, the metal catalyst is removed from the resulting reaction mixture by filtration, then the solvent is distilled off under reduced pressure, and the resultant is purified by recrystallization, evaporation from standing, washing and column chromatography to obtain a pure product.
The solvent may be used according to known methods such as US2001070671, j.am.chem.soc.1960, 82: 2953 benzonitrile (IV) is synthesized from the corresponding aminobenzoic acid derivative, halogen acid chloride and hydroxybenzonitrile.
(2) The salt of phenylbenzylamine can be synthesized by:
benzylamine (II) is preferably reacted with the acid using a solvent at-5 to 50 ℃, preferably at a temperature of 0 to 25 ℃, with particular reference to CN 1511142A.
The acid which can be salified with the phenylbenzylamine represented by the general formula (II) of the present invention includes: carboxylic acids such as acetic acid, propionic acid, butyric acid, oxalic acid, adipic acid, dodecanedioic acid, lauric acid, stearic acid, trifluoroacetic acid, fumaric acid, maleic acid, benzoic acid, or phthalic acid; sulfonic acids such as methanesulfonic acid, 1, 3-propanedisulfonic acid, p-toluenesulfonic acid or dodecylbenzenesulfonic acid; and inorganic acids such as hydrochloric acid, sulfuric acid, nitric acid or carbonic acid.
Any organic solvent may be used as the organic solvent for the reaction as long as it has no direct influence on the reaction. The organic solvent includes aromatic hydrocarbon such as benzene, toluene or xylene; ketones, such as acetone, methyl ethyl ketone or methyl isobutyl ketone; halogenated hydrocarbons such as chloroform or dichloromethane; water; esters, such as methyl acetate or ethyl acetate; and polar solvents such as tetrahydrofuran, acetonitrile, dioxane, N-dimethylformamide, N-methylpyrrolidone, dimethylsulfoxide, or pyridine.
The salt of phenyl benzylamine is obtained by conventional separation method.
The compound of the general formula (I) has high bactericidal activity on plant pathogenic bacteria, and can effectively control diseases caused by downy mildew, powdery mildew, rust disease and the like when being used as a bactericide. Therefore, the technical scheme of the invention also comprises the application of the compound with the general formula (I) in preventing and treating germs. The plant pathogenic bacteria include, for example, Magnaporthe grisea, powdery mildew, Rust disease and downy mildew, and especially have good activity on cucumber downy mildew, rice blast and corn rust. Therefore, the compounds can be used as active ingredients of bactericides in agricultural fields such as agriculture, horticulture and flower cultivation, and therefore the compounds of the general formula (I) of the present invention are preferably used for controlling phytopathogens in agricultural and other fields. The plant germs which can be controlled by the compounds of the present invention are not limited to the above.
Owing to their good properties, the abovementioned compounds can be used advantageously for protecting crops, livestock and breeding animals of agricultural and horticultural importance, as well as the environment in which humans are often exposed, against fungal damage.
The amount of the compound used to achieve the desired effect will vary depending on factors such as the compound used, the crop being protected, the type of pest, the extent of infection, the climatic conditions, the method of application, and the dosage form employed.
A dose of the compound of 10g to 1000 g per hectare provides adequate control.
A further object of the present invention relates to a method for controlling phytopathogenic fungi of crops of agricultural and horticultural importance and/or of domestic and livestock animals and/or of the environment frequented by humans by applying compounds of the general formula (I). In particular, the amount of compound used varies from 10 grams per hectare to 1000 grams per hectare.
For practical use in agriculture, it is often beneficial to use compositions containing one or more compounds of formula (I).
Another object of the invention relates to a bactericidal composition containing one or more compounds of the general formula (I) as active ingredients, wherein the weight percentage of the active ingredients in the composition is 0.1-99%. The compositions also contain an agriculturally acceptable carrier, and typically a surfactant. Therefore, the technical scheme of the invention also comprises the application of the composition as a bactericide in preventing and controlling germs in agriculture or other fields.
The composition can be used in the forms of dry powder, wettable powder, missible oil, microemulsion, paste, granules, solution, suspending agent and the like: the choice of the type of composition depends on the particular application.
The compositions are prepared in a known manner, for example by diluting or dissolving the active substance with a solvent medium and/or a solid diluent, optionally in the presence of a surfactant.
Useful solid diluents or carriers are, for example: silica, kaolin, bentonite, talc, diatomaceous earth, dolomite, calcium carbonate, magnesium oxide, chalk, clay, synthetic silicates, attapulgite, sepiolite and the like.
Besides water, usable liquid diluents also include, for example, aromatic organic solvents (xylene or a mixture of alkylbenzenes, chlorobenzene, etc.), paraffins (petroleum fractions), alcohols (methanol, propanol, butanol, octanol, glycerol), esters (ethyl acetate, isobutyl acetate, etc.), ketones (cyclohexanone, acetone, acetophenone, isophorone, ethyl amyl ketone, etc.), amides (N, N-dimethylformamide, N-methylpyrrolidone, etc.).
Useful surfactants are the sodium, calcium, triethylamine or triethanolamine salts of alkyl sulfonates, alkylaryl sulfonates, polyoxyethylene alkylphenols, polyoxyethylene esters of sorbitol, lignosulfonates, and the like.
The composition may further contain specific additives for specific purposes, for example, binders such as gum arabic, polyvinyl alcohol, polyvinyl pyrrolidone, etc.
The concentration of the active ingredient in the above composition may vary within wide limits depending on the active ingredient, the purpose of use, the environmental conditions and the type of formulation employed. The concentration of the active ingredient is generally in the range from 0.1 to 99%, preferably from 5 to 60%.
If desired, other active ingredients compatible with the compounds of formula (I) may be added to the composition, for example other acaricides/insecticides, fungicides, plant growth regulators, antibiotics, herbicides, fertilizers.
However, the form of the composition is not limited thereto, and one or two or more compounds in combination may be mixed as an active ingredient.
Several common formulation methods are exemplified below:
preparation of a suspending agent: the content of active components in the common formula is 5-35%. Adding water as medium, raw medicine, dispersant, suspending agent and anti-freezing agent into a sand mill, grinding, and making into suspension.
Preparing the aqueous emulsion: the raw medicine, the solvent and the emulsifier are added together to be dissolved into a uniform oil phase. Mixing water and antifreeze agent to obtain uniform water phase. And adding the water phase into the oil phase or adding the oil phase into the water phase under high-speed stirring to form the aqueous emulsion with good dispersibility. The content of the active component of the aqueous emulsion is generally 5 to 15 percent. To prepare a concentrated emulsion, the compounds of the invention can be dissolved in one or more solvents and emulsifiers added to enhance the dispersion of the compounds in water.
Preparing wettable powder: according to the formula requirement, the original medicine, various surfactants, solid diluents and the like are fully mixed and crushed by an ultrafine crusher to obtain a wettable powder product with a preset content (for example, 10-60%). To prepare wettable powders suitable for spraying, the compounds of the invention may be mixed with finely divided solid powders such as clays, inorganic silicates, carbonates and wetting, binding and/or dispersing agents.
Preparing water dispersible granules: mixing and crushing the original medicine, the powdery solid diluent, the wetting and spreading agent, the adhesive and the like, adding water, kneading, adding into a granulator with a certain specification of sieve for granulation, and then drying and sieving (according to the range of the sieve). Or adding the raw medicine, dispersant, disintegrant, wetting agent and solid diluent into a sand mill, grinding with water as a medium to prepare a suspending agent, and then carrying out spray drying granulation to prepare a granular product with the content of 20-30 percent.
Detailed Description
The following specific examples are intended to further illustrate the invention, but the invention is by no means limited to these examples.
Synthetic examples
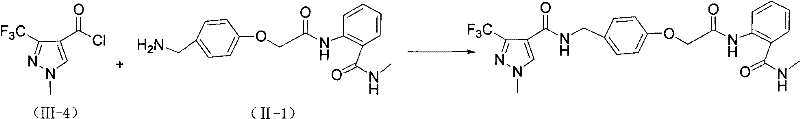
Example 1: preparation of intermediate II-1
1).
Adding 19.80g (0.067mol) of solid phosgene into 150ml of tetrahydrofuran solution containing 13.70g (0.1mol) of anthranilic acid in small portions under stirring at room temperature, finishing the addition within about 1.5 hours, continuing stirring at room temperature for 2-3 hours, after the completion of TLC monitoring reaction, evaporating the solvent under reduced pressure, adding the residue into 80ml of water, stirring until excessive phosgene is completely decomposed, filtering and washing by respectively 50ml of water and petroleum ether in sequence to obtain 14.00g of white solid, the yield is 86.0 percent, and the melting point is 239-240 ℃.
2).
Slowly dripping 40% methylamine water solution into 100ml acetonitrile solution containing 16.30g (0.1mol) benzoxazinone under stirring at room temperature until the solution becomes clear, dripping 80ml methylamine water solution, continuing to react for 0.5 hour at room temperature, after TLC monitoring reaction, decompressing and removing solvent, washing with water and petroleum ether 50ml respectively to obtain white solid 13.73g, yield 91.5%, melting point 78-79 ℃.
3).
Slowly dropwise adding 30ml of dichloromethane solution containing 12.40g (0.11mol) of chloroacetyl chloride into 50ml of dichloromethane solution containing 15.00g (0.1mol) of anthranilamide and 12.10g (0.12mol) of triethylamine under stirring at room temperature, continuously reacting at room temperature for 2-3 hours, monitoring the reaction by TLC to an end point, removing the solvent under reduced pressure, and sequentially washing by respectively 50ml of 10% dilute hydrochloric acid, saturated sodium bicarbonate solution, water and petroleum ether to obtain 18.90g of white solid, wherein the yield is 83.4%, and the melting point is 155-.
4).
22.65g (0.1mol) of o-chloroacetylaminobenzamide and 14.29g (0.12mol) of p-hydroxybenzonitrile are added into 80ml of butanone, 27.60g (0.2mol) of potassium carbonate is added, the mixture is heated to reflux under stirring, the reaction is carried out for 4 to 5 hours, TLC monitors the reaction until the end point, the solvent is evaporated under reduced pressure, 50ml of alkali, water and petroleum ether are sequentially used for washing to obtain most of target products, a small amount of residue can be obtained by column chromatography, 25.00g of white solid is obtained, and the yield is 81.0%.
5).
The mixture consisting of 3.09g (0.01mol) of the intermediate (IV-1), Raney nickel (1.0g), 10ml of 25% ammonia water and 50ml of ethanol is stirred and reacted for 3-4 hours at room temperature under hydrogen atmosphere, TLC monitors the reaction to the end point, Raney nickel is filtered out, the solvent is removed under reduced pressure to obtain viscous liquid (II-1), the viscous liquid is cooled and washed to obtain white solid, 2.16g of the white solid is obtained, the yield is 69.00%, and the melting point is 114-.
Example 2: preparation of Compound 23
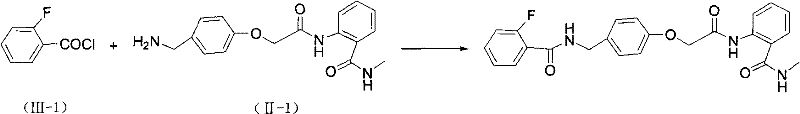
Adding 0.313g (0.001mol) of benzylamine (II-1) and 0.12g (0.0012mol) of triethylamine into 20ml of dichloromethane, dropwise adding 0.17g (0.0011mol) of 10ml of dichloromethane solution of (III-1) while stirring at room temperature, continuing stirring at room temperature for 1 hour after dropwise adding, monitoring the reaction by TLC until the end point, pouring the reaction mixture into 30ml of water, shaking to separate an organic layer, washing the organic layer by using 10% dilute hydrochloric acid, saturated sodium bicarbonate aqueous solution and saturated saline solution in turn, drying and desolventizing to obtain a product, washing the product by using ethyl acetate to obtain a white solid, and carrying out column chromatography to obtain 0.38g of a pure product, wherein the yield is 87.5%, and the melting point is 168-169 ℃.
Example 3: preparation of Compound 62
Adding 0.313g (0.001mol) of benzylamine (II-1) and 0.12g (0.0012mol) of triethylamine into 20ml of dichloromethane, dropwise adding 0.22g (0.0011mol) of 10ml of dichloromethane solution of (III-2) while stirring at room temperature, continuing stirring at room temperature for 1 hour after dropwise adding, monitoring the reaction by TLC until the end point, pouring the reaction mixture into 30ml of water, shaking to separate an organic layer, washing the organic layer by using 10% dilute hydrochloric acid, saturated sodium bicarbonate aqueous solution and saturated saline solution in sequence, washing by using 20ml of each organic layer, drying and desolventizing to obtain a product, washing by using ethyl acetate to obtain a white solid, and performing column chromatography to obtain 0.39g of a pure product, wherein the yield is 82.0%, and the melting point is 154-156 ℃.
Example 4: preparation of Compound 133
Adding 0.313g (0.001mol) of benzylamine (II-1) and 0.12g (0.0012mol) of triethylamine into 20ml of dichloromethane, dropwise adding 0.27g (0.0011mol) of (III-3) of 10ml of dichloromethane while stirring at room temperature, continuing stirring at room temperature for 1 hour after dropwise adding, monitoring the reaction by TLC until the end point, pouring the reaction mixture into 30ml of water, shaking to separate an organic layer, washing the organic layer by 10% of dilute hydrochloric acid, saturated sodium bicarbonate water solution and saturated saline water in sequence, drying and desolventizing to obtain a product, washing the product by ethyl acetate to obtain a white solid, and performing column chromatography to obtain 0.41g of a pure product, wherein the yield is 78.2%, and the melting point is 173-.
Example 5: preparation of Compound 150
Adding 0.313g (0.001mol) of benzylamine (II-1) and 0.12g (0.0012mol) of triethylamine into 20ml of dichloromethane, dropwise adding 10ml of dichloromethane solution containing 0.21g (0.0011mol) (III-4) while stirring at room temperature, continuing stirring at room temperature for 1 hour after dropwise adding, monitoring the reaction end point by TLC, pouring the reaction mixture into 30ml of water, shaking to separate an organic layer, washing the organic layer by 10% diluted hydrochloric acid, saturated sodium bicarbonate water solution and 50ml of saturated saline solution respectively, drying and desolventizing to obtain a product, washing by ethyl acetate to obtain a white solid, and performing column chromatography to obtain 0.41g of a pure product with the yield of 86.0%. Melting point 178-.
Other compounds of formula (I) may be prepared by the preparation methods provided herein.
Melting Point of some Compounds (melting Point instrument uncorrected) and Nuclear magnetic data: (1HNMR, 300MHz, internal standard TMS, solvent CDCl3) The following were used:
compound 1: melting point 147-. δ ppm 1.15-1.30(6H, m), 1.50-1.53(1H, d), 1.91-1.97(1H, m), 3.74(3H, s), 4.35-4.37(2H, d), 5.21(2H, s), 5.79(1H, s), 6.44-6.46(1H, d), 7.02-7.05(2H, m), 7.21-7.26(2H, m), 7.50-7.55(1H, m), 7.75-7.77(2H, m), 8.28-8.31(1H, m).
Compound 8: melting point 173-. Delta ppm 0.67-0.69(3H, d), 1.01-1.03(3H, d), 2.40(1H, m), 2.76-2.80(1H, d), 3.72(3H, s), 4.18-4.42(2H, m), 5.16(2H, s), 5.80(1H, s), 6.95-6.97(2H, m), 7.09-7.11(2H, m), 7.50(1H, m), 7.73-7.74(2H, m), 8.30(1H, m).
Compound 10: melting point 199-. Delta ppm 3.74(3H, s), 4.57-4.59(2H, d), 5.22(2H, s), 6.40(1H, s), 7.03-7.06(2H, m), 7.26-7.32(3H, m), 7.39-7.52(5H, m), 7.75-7.79(4H, m), 8.27(1H, m).
Compound 13: melting point 167-. δ ppm 3.72(3H, s), 4.59-4.61(2H, d), 5.18(2H, s), 6.51(1H, s), 7.03-7.06(2H, m), 7.27-7.39(5H, m), 7.48-7.53(1H, m), 7.64-7.76(2H, m), 8.27-8.29(1H, d).
Compound 23: melting point 168-. Delta ppm 3.74(3H, s), 4.62-4.63(2H, d), 5.18(2H, s), 7.04-7.14(4H, m), 7.25-7.34(4H, m), 7.46-7.54(2H, m), 7.73-7.77(2H, m), 8.13(1H, m), 8.29-8.31(1H, m).
Compound 27: melting point 187-. δ ppm 3.76(3H, s), 4.62(2H, d), 5.22(2H, s), 7.10(2H, m), 7.26-7.30(6H, m), 7.80(2H, m), 8.30(1H, d).
Compound 33: melting point 225-. Delta ppm 3.73(3H, s), 4.60-4.61(2H, d), 5.20(2H, s), 6.24(1H, s), 6.90-6.95(2H, m), 7.03-7.06(2H, m), 7.30-7.37(3H, m), 7.49-7.53(1H, m), 7.71-7.77(2H, m), 8.27-8.29(1H, d).
Compound 38: melting point 182-. δ ppm 2.38(3H, s), 3.71(3H, s), 4.56-4.57(2H, d), 5.17(2H, s), 6.43(1H, s), 7.01-7.04(2H, m), 7.19-7.31(5H, m), 7.48-7.53(1H, m), 7.66-7.76(4H, m), 8.27-8.29(1H, m).
Compound 44: melting point 163-. δ ppm 2.27(3H, s), 3.63(2H, s), 3.73(3H, s), 4.32-4.34(2H, d), 5.15(2H, s), 5.60(1H, s), 6.96-6.99(2H, m), 7.10-7.26(6H, m), 7.49-7.54(1H, m), 7.69-7.77(2H, m), 8.28-8.30(1H, m).
Compound 47: melting point 163-. δ ppm 2.38(3H, s), 3.73(3H, s), 4.58-4.59(2H, d), 5.18(2H, s), 7.02-7.05(2H, m), 7.26-7.32(4H, m), 7.49-7.61(3H, m), 7.69-7.77(2H, m), 8.28-8.30(1H, m).
Compound 55: melting point 200 ℃ and 201 ℃. δ ppm 3.73(3H, s), 4.56-4.58(2H, d), 5.19(2H, s), 6.10(1H, s), 7.03-7.06(2H, m), 7.27-7.32(2H, m), 7.51-7.55(4H, m), 7.72-7.74(4H, m), 8.30(1H, m).
Compound 59: melting point 118-. δ ppm 2.97-3.06(6H, d), 4.58-4.60(4H, m), 7.00-7.03(1H, m), 7.14-7.17(1H, m), 7.26-7.28(2H, m), 7.31-7.34(2H, m), 7.53-7.59(4H, m), 7.69-7.71(1H, m), 8.38(1H, m), 9.98(1H, s).
Compound 62: melting point 154-. Delta ppm 2.97-3.06(6H, d), 4.58-4.60(4H, m), 7.00-7.03(1H, m), 7.14-7.17(1H, m), 7.26-7.28(2H, m), 7.31-7.34(2H, m), 7.53-7.59(4H, m), 7.69-7.71(1H, m).
Compound 70: melting point 149-. Delta ppm 3.73(3H, s), 3.91-3.92(6H, m), 4.56-4.58(2H, d), 5.18(2H, s), 6.40(1H, s), 6.82-6.85(1H, m), 7.02-7.05(2H, m), 7.27-7.31(3H, m), 7.45-7.51(2H, m), 7.69-7.76(2H, m), 8.28-8.30(1H, d).
Compound 74: melting point 178-. δ ppm 3.74(3H, s), 3.87-3.88(9H, m), 4.57-4.59(2H, d), 5.18(2H, s), 7.01-7.06(4H, m), 7.26-7.32(2H, m), 7.70(1H, m), 7.71(2H, m), 8.30(1H, m).
Compound 79: melting point 205-. δ ppm 3.05(3H, s), 3.74(3H, s), 4.60-4.61(2H, d), 5.22(2H, s), 6.45(1H, s), 7.05-7.08(2H, m), 7.26-7.34(2H, m), 7.52-7.55(1H, m), 7.74-7.78(2H, m), 7.81-7.87(2H, m), 7.97(1H, s), 8.27-8.29(1H, d).
Compound 83: melting point 171-. Δ ppm 3.74(3H, s), 4.27(2H, m), 5.21(2H, s), 6.40(1H, s), 6.88(4H, m), 7.35-7.61(10H, m), 7.77(2H, m), 8.27(1H, m).
Compound 88: melting point 202-. δ ppm 3.72(3H, s), 3.87(3H, s), 4.61-4.63(2H, d), 4.17(2H, s), 6.20(1H, s), 7.02-7.09(3H, m), 7.27-7.28(1H, m), 7.31-7.37(2H, m), 7.44-7.48(1H, m), 7.71(2H, m), 8.26(1H, d).
Compound 95: melting point 168-. Delta ppm 3.74(3H, s), 4.49-4.51(2H, m), 4.55(2H, s), 5.18(2H, s), 6.82-6.84(1H, m), 7.03-7.06(3H, m), 7.20-7.27(4H, m), 7.38-7.39(1H, m), 7.52(1H, m), 7.70-7.77(2H, m), 8.29-8.31(1H, d).
Compound 101: melting point 194 ℃ and 195 ℃. Delta ppm of 3.74-3.77(3H, s), 4.60-4.62(2H, d), 5.22-5.24(2H, s), 6.80(1H, s), 7.05-7.08(2H, m), 7.27-7.36(3H, m), 7.52-7.55(1H, m), 7.74-7.77(2H, m), 8.10-8.14(1H, m), 8.27-8.30(1H, m), 8.44-8.46(1H, m).
Compound 105: melting point 206-. δ ppm 3.74(3H, s), 4.61(2H, d), 5.20(2H, s), 6.77(1H, s), 7.04-7.07(2H, m), 7.26-7.49(3H, m), 7.51-7.52(1H, m), 7.72-7.77(1H, m), 7.89-7.93(1H, m), 8.12-8.14(1H, m), 8.45-8.46(1H, m).
Compound 108: the melting point is 118-120 ℃. δ ppm 1.37-1.41(3H, t), 2.62(3H, s), 4.24-4.27(2H, q), 4.57-4.59(2H, d), 5.21(2H, s), 6.59(1H, s), 7.07-7.10(2H, m), 7.26-7.31(2H, m), 7.38-7.40(2H, m), 7.58(1H, m), 8.07-8.11(2H, m), 8.74-8.75(1H, m).
Compound 112: melting point 208-. Delta ppm 3.72(3H, s), 4.56-4.58(2H, d), 5.18(2H, s), 6.59(1H, s), 7.02-7.05(2H, m), 7.27-7.30(3H, m), 7.38-7.41(1H, m), 7.49-7.54(1H, m), 7.69-7.77(2H, m), 8.08-8.10(1H, m), 8.26-8.29(1H, m), 8.74-8.75(1H, s).
Compound 118: melting point 177-. Delta ppm 3.73(3H, s), 4.58-4.60(2H, d), 5.20(2H, s), 6.20(1H, s), 7.04-7.07(2H, m), 7.26-7.32(3H, m), 7.51-7.56(2H, m), 7.73-7.77(2H, m), 8.26-8.28(1H, d), 8.82(1H, s).
Compound 122: melting point 182-. Δ ppm 3.73(3H,), 4.53-4.54(2H, d), 5.19(2H, s), 6.10(1H, s), 7.03-7.06(2H, m), 7.28-7.30(3H, m), 7.52(1H, m), 7.73-7.78(2H, m), 8.30(1H, m), 8.57(1H, s).
Compound 129: melting point 157-. δ ppm 3.73(3H, s), 4.54-4.56(2H, d), 5.18(2H, s), 6.50(1H, s), 6.66(1H, s), 7.03-7.06(2H, m), 7.14-7.15(1H, m), 7.28-7.31(3H, m), 7.51(1H, m), 7.66-7.77(2H, m), 8.29-8.31(1H, m).
Compound 133: melting point 173-. δ ppm 2.71(3H, s), 3.73(3H, s), 4.53-4.54(2H, d), 5.18(2H, s), 6.43(1H, s), 7.03-7.06(2H, m), 7.26-7.29(2H, m), 7.50(1H, m), 7.70(2H, m), 8.30(1H, d).
Compound 138: melting point 214-. Delta ppm 3.62(3H, s), 3.80-4.00(2H, m), 5.28(2H, s), 7.14-7.24(4H, m), 7.54(3H, m), 7.64-7.67(3H, m), 7.81-7.82(3H, m), 8.14(1H, m), 8.50-8.52(1H, m).
Compound 142: melting point 170-. δ ppm 1.16-1.21(3H, t), 2.68-2.70(2H, q), 3.79(3H, s), 4.55-4.57(2H, m), 5.08(2H, s), 6.96-7.03(3H, m), 7.25-7.35(1H, m), 7.51(2H, m), 7.71-7.80(21H, m), 8.26(1H, m), 9.98(1H, s).
Compound 143: melting point 207-. δ ppm 1.16-1.21(3H, t), 2.59(3H, s), 2.70-2.73(2H, q), 3.84(3H, s), 4.47-4.49(2H, m), 4.63(2H, s), 6.99-7.02(2H, m), 7.14(1H, m), 7.30-7.33(2H, m), 7.52-7.58(1H, m), 7.70-7.76(2H, m), 8.04-8.08(1H, m), 8.72-8.78(1H, m).
Compound 144: melting point 129-. δ ppm 1.14-1.19(3H, t), 1.83-1.85(3H, d), 2.66-2.68(2H, q), 3.71(3H, s), 3.80(3H, s), 4.50-4.52(2H, d), 5.50-5.58(1H, m), 6.94-6.97(3H, m), 7.24-7.28(2H, m), 7.42-7.55(1H, m), 7.72-7.73(2H, m), 8.25-8.30(1H, m).
Compound 145: melting point 130-. δ ppm 1.34-1.38(3H, t), 2.29(3H, s), 2.68-2.71(2H, q), 3.81(3H, s), 4.30-4.33(2H, m), 4.56-4.58(3H, m), 4.66(2H, s), 6.99-7.02(4H, m), 7.22-7.26(2H, m), 7.33-7.35(1H, m), 7.80(1H, m), 9.92(1H, s).
Compound 146: melting point 113-. δ ppm 1.15-1.20(3H, t), 2.67-2.70(2H, q), 3.73(3H, s), 3.79(3H, s), 4.53-4.55(2H, d), 5.18(2H, s), 7.01-7.04(3H, m), 7.27-7.33(2H, m), 7.52(1H, m), 7.72-7.74(2H, m), 8.30(1H, m).
Compound 150: melting point 178-. Delta ppm 3.74(3H, s), 4.02-4.03(3H, s), 4.51-4.53(2H, d), 5.20(2H, s), 6.15(1H, s), 7.03-7.06(2H, d), 7.27-7.29(3H, m), 7.52(1H, m), 7.69-7.77(3H, m), 8.27-8.30(1H, m).
Compound 152: melting point 179-. Delta ppm 3.74(3H, s), 3.91(3H, s), 4.54-4.56(2H, d), 5.20(2H, s), 6.30(1H, s), 7.03-7.06(2H, d), 7.27-7.30(2H, m), 7.49-7.54(1H, m), 7.71-7.77(2H, m), 8.27-8.30(1H, m).
Compound 159: melting point 156-. δ ppm 1.58(3H, m), 3.94(3H, s), 4.30(2H, m), 4.40(2H, m), 5.19(2H, s), 6.80-7.20(6H, m), 7.30-7.40(3H, m), 7.42-7.58(3H, m), 7.70-7.80(1H, m), 7.82-8.00(1H, m).
Compound 165: melting point 137-. δ ppm 0.98-1.00(3H, m), 2.39(3H, s), 3.74(3H, s), 3.90(3H, s), 4.50-4.52(2H, d), 4.92(2H, s), 5.19(2H, s), 7.04-7.07(2H, m), 7.52-7.55(2H, m), 7.72-7.78(3H, m), 8.30(1H, m).
Compound 193: the melting point is 98-99 ℃. Delta ppm 3.91-3.93(8H, m), 3.99(6H, m), 4.62-4.64(4H, m), 6.30-6.35(2H, m), 7.21-7.27(4H, m), 7.38-7.41(3H, m).
Formulation examples (the amounts of the components added are in weight percent, and the active compound is metered in after being reduced to a hundred)
Example 6: 30% wettable powder
Compound 2330%
Sodium dodecyl sulfate 2%
Sodium lignosulfonate 3%
Naphthalene sulfonic acid formaldehyde condensate 5%
The light calcium carbonate is complemented to 100 percent
And (3) fully mixing the compound 23 and other components, and crushing by using an ultrafine crusher to obtain a 30% wettable powder product.
Example 7: 40% thick suspending agent
Compound 15240%
10 percent of ethylene glycol
Nonyl phenol polyglycol ether 6%
Sodium lignosulfonate 10%
1 percent of carboxymethyl cellulose
37% aqueous formaldehyde solution 0.2%
75% of silicone oil water emulsion 0.8%
The water is complemented to 100 percent
Compound 152 and the other components are mixed thoroughly to give a suspension concentrate, and the suspension concentrate is diluted with water to give a dilution of any desired concentration.
Example 8: 60% water dispersible granule
Compound 13860%
Sodium naphthalenesulfonate formaldehyde condensate 12%
Sodium N-methyl-N-oleoyl-taurate 8%
2 percent of polyvinylpyrrolidone
2 percent of carboxymethyl cellulose
Kaolin is complemented to 100%
Mixing and crushing the compound 138 and other components, adding water, kneading, adding into a granulator with a 10-100 mesh screen for granulation, and then drying and screening (according to the range of the screen).
Examples of measurement of biological Activity
Example 9: biological activity of greenhouse pot (cucumber downy mildew)
The test was performed using potted seedling assay. Selecting potted cucumber seedlings with regular and consistent growth, cutting off growth points, reserving two true leaves, carrying out leaf surface spraying treatment by using the compound according to the designed concentration, additionally arranging a blank control for spraying clear water, and repeating for 3 times. Inoculating cucumber downy mildew spore suspension on the next day after treatment, placing the inoculated cucumber downy mildew spore suspension in a climatic chamber (the temperature is 25 ℃ in the day, 20 ℃ in the night, and the relative humidity is 95-100%) for moisture preservation and culture, and placing the inoculated cucumber downy mildew spore suspension in a greenhouse (25 ℃ +/-1 ℃) for normal management after 24 hours. The control effect is investigated 7 days after inoculation, and the control effect is calculated according to disease indexes by grading according to the national standard of the people's republic of China, namely the pesticide field efficacy test criterion.
Some of the test results are as follows:
when the concentration of the liquid medicine is 400ppm, the control effect of the compounds 13, 23, 95, 138, 143 and 152 on cucumber downy mildew reaches 100 percent;
when the concentration of the liquid medicine is 100ppm, the control effect of the compounds 13, 23, 95, 138, 143 and 152 on cucumber downy mildew reaches 50 percent.
Example 10: greenhouse pot biological activity (wheat powdery mildew)
The test was performed using potted seedling assay. Selecting the wheat potted seedlings with regular and consistent growth in the 2-leaf stage, carrying out leaf surface spraying treatment by using the compound according to the designed concentration, additionally setting a blank control for spraying clear water, and repeating for 3 times. Inoculating the treated plants by a spore shaking method on the next day, and normally managing the inoculated plants in a greenhouse (25 +/-1 ℃). The disease classification refers to the national standard of the people's republic of China, namely the pesticide field efficacy test criterion, and the prevention and treatment effect is calculated according to the disease index after the medicine is used for investigation for seven days.
Some of the test results are as follows:
when the concentration of the liquid medicine is 400ppm, the control effect of the compound 62 on wheat powdery mildew reaches 100 percent;
example 11: greenhouse pot biological activity (corn rust)
The test was performed using potted seedling assay. The corn potted seedlings with 2-3 leaf stages which grow regularly are selected, the compound is used for carrying out leaf surface spraying treatment according to the designed concentration, a blank control for spraying clear water is additionally arranged, and the steps are repeated for 3 times. Inoculating the corn rust spore suspension on the next day after treatment, placing the inoculated corn rust spore suspension in a climatic chamber (the temperature is 25 ℃ in the day, the temperature is 20 ℃ in the night, and the relative humidity is 95-100%) for moisture preservation and culture, and placing the corn rust spore suspension in a greenhouse (25 ℃ +/-1 ℃) for normal management after 24 hours. The control effect is investigated 10 days after inoculation, and the control effect is calculated according to disease indexes by grading according to the national standard of the people's republic of China, namely the pesticide field efficacy test criterion.
Some of the test results are as follows:
when the concentration of the liquid medicine is 400ppm, the control effect of the compounds 62 and 193 on the corn rust disease reaches 100 percent.
Claims (10)
1. An acyl benzylamine compound, which has a structure shown in the general formula (I):
in the formula:
R1selected from hydrogen or C1-C12An alkyl group; m is an integer selected from 1 to 3;
R2selected from halogen, cyano, nitro, C1-C12Alkyl, halo C1-C12Alkyl radical, C1-C12Alkoxy, halo C1-C12Alkoxy radical, C1-C12Alkylthio radical, C1-C12Alkylsulfonyl or R5(ii) a n is an integer from 0 to 4;
y is selected from O, S or NR4;
R3、R4Can be the same or different and are respectively selected from hydrogen, cyano and C1-C12Alkyl, halo C1-C12Alkyl radical, C1-C12Alkoxy, halo C1-C12Alkoxy radical, C3-C6Cycloalkyl, cyano C1-C12Alkyl radical, C1-C12Alkylamino, halogeno C1-C12Alkylamino radical, C2-C6Dialkylamino radical, C2-C12Alkenyl, halo C2-C12Alkenyl radical, C2-C12Alkenyloxy, halogeno C2-C12Alkenyloxy radical, C2-C12Alkynyl, halo C2-C12Alkynyl, C2-C12Alkynyloxy, halo C2-C12Alkynyloxy, C1-C12Alkylthio, halo C1-C12Alkylthio radical, C1-C12Alkylsulfonyl radical, C1-C12Alkylcarbonyl, halo C1-C12Alkylcarbonyl, halo C1-C12Alkylsulfonyl or R5(ii) a Or,
when Y is selected from NR4N R4And R3Form a substituted or unsubstituted five-or six-membered ring, the substituents being selected from 1 to 2C1-C4An alkyl group;
R5selected from phenyl, benzyl or heteroaryl, unsubstituted or substituted with 1 to 4 groups independently selected from: halogen, nitro, cyano, C1-C12Alkyl, halo C1-C12Alkyl radical, C1-C12Alkoxy, halo C1-C12Alkoxy radical, C1-C12Alkylthio radical, C1-C12Alkylcarbonyl or C1-C12An alkoxycarbonyl group;
(CHR1) The connecting position of mCONH and a benzene ring is selected from 2, 3 or 4 positions;
q is selected from C unsubstituted or substituted by 1 to 41-C6Alkyl, cyclopropyl or cyclohexyl: cl, Br, F or C1-C4An alkyl group;
or selected from anilino or C, unsubstituted or substituted by 1-32-C4Alkenyl: cl, Br, F, C1-C4Alkyl radical, C3-C6Cycloalkyl radical, C1-C4Haloalkyl, C1-C4Alkoxy radical, C1-C4Haloalkoxy or C1-C4An alkoxycarbonyl group;
or selected from phenyl, phenoxyethyl, phenoxypropyl, pyridyl, pyrimidinyl, piperidinyl, piperazinyl, thiazolyl, isothiazolyl, thiadiazolyl, oxazolyl, isoxazolyl, oxadiazolyl, triazolyl, thienyl, furanyl, quinolinyl, indolyl, 3-pyrazolyl, 4-pyrazolyl or 4-pyrazolylmethyl, unsubstituted or substituted with 1-3 of the following groups: halogen, CN, NO2、C1-C4Alkyl radical, C3-C6Cycloalkyl radical, C1-C3Haloalkyl, C1-C3Alkoxyalkyl group, C1-C3Alkoxy radical, C1-C3Haloalkoxy, COR6、CO2R6、CON R6R7、NR6R7、NR7COR6、NR7CO2R6、SR7、SOR7、SO2R7、SO2NR6R7Or R7;
R6Is selected from H or C1-C4An alkyl group;
R7selected from H, C1-C4Alkyl radical, C1-C3Halogenated alkyl, substituted or unsubstituted phenyl, the substituent being selected from Cl, Br, F, CN, NO2、C1-C4Alkyl, CF3、OCH3、OCF3Or CO2CH3。
2. The acyl benzylamine compound according to claim 1, characterized in that: in the general formula (I)
R1Selected from hydrogen or C1-C6An alkyl group; m is an integer selected from 1 to 3;
R2selected from halogen, cyano, nitro, C1-C6Alkyl, halo C1-C6Alkyl radical, C1-C6Alkoxy, halo C1-C6Alkoxy radical, C1-C6Alkylthio radical, C1-C6Alkylsulfonyl or R5(ii) a n is an integer from 0 to 4;
y is selected from O, S or NR4;
R3、R4Can be the same or different and are respectively selected from hydrogen, cyano and C1-C6Alkyl, halo C1-C6Alkyl radical, C1-C6Alkoxy, halo C1-C6Alkoxy radical, C3-C6Cycloalkyl, cyano C1-C6Alkyl radical, C1-C6Alkylamino, halogeno C1-C6Alkylamino radical, C2-C6Dialkylamino radical, C3-C6Alkenyl, halo C3-C6Alkenyl radical, C3-C6Alkenyloxy, halogeno C3-C6Alkenyloxy radical, C3-C6Alkynyl, halo C3-C6Alkynyl, C3-C6Alkynyloxy, C1-C6Alkylthio, halo C1-C6Alkylthio radical, C1-C6Alkylsulfonyl radical, C1-C6Alkylcarbonyl, halo C1-C6Alkylcarbonyl, halo C1-C6Alkylsulfonyl or R5(ii) a Or,
when Y is selected from NR4N R4And R3Consisting of morpholine, piperazine, piperidine, pyrazole, pyrrole or triazole rings;
R5Selected from phenyl, benzyl, pyridyl, pyrimidinyl, thiazolyl, isothiazolyl, oxazolyl, isoxazolyl, oxadiazolyl, thiadiazolyl, pyrazolyl, pyranyl, triazolyl or tetrazolyl, unsubstituted or substituted with 1 to 4 groups independently selected from: halogen, nitro, cyano, C1-C6Alkyl, halo C1-C6Alkyl radical, C1-C6Alkoxy, halo C1-C6Alkoxy radical, C1-C6Alkylthio radical, C1-C6Alkylcarbonyl or C1-C6An alkoxycarbonyl group;
q is selected from one of the groups shown below:
R8selected from H, Cl, Br, F, CN, NO2、NH2、CH3、CH2CH3Tert-butyl, cyclopropyl, CF3、CH2CF3、OCH3、OCF3、OCH2CF3、SO2CH3、CO2CH3、C1-C3Alkylamino carbonyl, C2-C4Dialkylaminocarbonyl, unsubstituted or substituted phenyl, unsubstituted or substituted pyridyl, said substituents being selected from 1 to 3 of the following groups: cl, Br, F, CN, NO2、CH3、CH2CH3Tert-butyl, cyclopropyl, CHF2、CF3、CH2CF3、OCH3、OCHF2、OCF3、OCH2CF3Or SO2CH3;
R9Selected from H, CH3、CH2CH3Tert-butyl, CF3、CH2CF3Unsubstituted or substituted phenyl, unsubstituted or substituted pyridyl, said substituents being selected from 1 to 3 of the following groups: cl, Br, F, CN, NO2、CH3、CH2CH3Tert-butyl, cyclopropyl, CHF2、CF3、CH2CF3、OCH3、OCHF2、OCF3、OCH2CF3Or SO2CH3;
a is selected from an integer of 1 to 5; b is selected from an integer of 1 to 4; c is an integer selected from 1 to 3; d is selected from an integer from 1 to 2.
3. The compound of claim 2, wherein: in the general formula (I)
R1Selected from hydrogen or C1-C3An alkyl group; m is an integer selected from 1 to 3;
R2selected from halogen, cyano, nitro, C1-C4Alkyl, halo C1-C4Alkyl radical, C1-C4Alkoxy, halo C1-C4Alkoxy radical, C1-C3Alkylthio radical, C1-C4Alkylsulfonyl or R5(ii) a n is an integer selected from O to 3;
y is selected from O, S or NR4;
R3、R4Can be the same or different and are respectively selected from hydrogen, cyano and C1-C6Alkyl, halo C1-C6Alkyl radical, C1-C6Alkoxy, halo C1-C6Alkoxy radical, C3-C6Cycloalkyl, cyano C1-C6Alkyl radical, C1-C6Alkylamino, halogeno C1-C6Alkylamino radical, C2-C6Dialkylamino radical, C3-C6Alkenyl, halo C3-C6Alkenyl radical, C3-C6Alkynyl, halo C3-C6Alkynyl, C1-C6Alkylthio, halo C1-C6Alkylthio radical, C1-C6Alkylsulfonyl radical, C1-C6Alkylcarbonyl, halo C1-C6Alkylcarbonyl, halo C1-C6Alkylsulfonyl or R5(ii) a Or,
when Y is selected from NR4N R4And R3To form a morpholine, piperazine, piperidine or pyrazole ring;
R5selected from phenyl, benzyl, pyridyl, pyrimidyl, thiazolyl, oxazolyl, oxadiazolyl, thiadiazolyl, pyrazolyl, triazolyl or tetrazolyl, unsubstituted or substituted with 1-4 groups independently selected from: halogen, nitro, cyano, C1-C4Alkyl, halo C1-C4Alkyl radical, C1-C4Alkoxy, halo C1-C4Alkoxy radical, C1-C4Alkylthio radical, C1-C4Alkylcarbonyl or C1-C4An alkoxycarbonyl group;
q is selected from one of the groups shown below:
R8selected from H, Cl, Br, F, CN, NO2、CH3、CH2CH3Tert-butyl, cyclopropyl, CF3、CH2CF3、OCH3、OCF3、OCH2CF3、SO2CH3、CO2CH3Unsubstituted or substituted phenyl, unsubstituted or substituted pyridyl, said substituents being selected from 1 to 3 of the following groups: cl, Br, F, CN, NO2、CH3、CH2CH3Tert-butyl, cyclopropyl, CHF2、CF3、CH2CF3、OCH3、OCHF2、OCF3、OCH2CF3Or SO2CH3;
R9Selected from H, CH3、CH2CH3Tert-butyl, unsubstituted or substituted phenyl, unsubstituted or substituted pyridyl, said substituents being selected from 1 to 3 of the following groups: cl, Br, F, CN, NO2、CH3、CH2CH3Tert-butyl, cyclopropyl, CF3、CH2CF3、OCH3、、OCF3Or SO2CH3;
a is selected from an integer of 1 to 5; b is selected from an integer of 1 to 4; c is an integer selected from 1 to 3; d is selected from an integer from 1 to 2.
4. A compound according to claim 3, characterized in that: in the general formula (I)
R1Selected from hydrogen or methyl; m is an integer selected from 1 to 3;
R2selected from fluorine, chlorine, bromine, iodine, cyano, nitro, C1-C4Alkyl or halo C1-C4An alkyl group; n is an integer from 0 to 3;
y is selected from O or NR4;
R3、R4Can be the same or different and are respectively selected from hydrogen, cyano and C1-C5Alkyl, halo C1-C5Alkyl radical, C1-C5Alkoxy, halo C1-C5Alkoxy radical, C3-C6Cycloalkyl, cyano C1-C5Alkyl radical, C1-C5Alkylamino, halogeno C1-C5Alkylamino radical, C2-C6Dialkylamino radical, C3-C5Alkenyl, halo C3-C5Alkenyl radical, C3-C5Alkynyl, halo C3-C5Alkynyl, C1-C5Alkylsulfonyl radical, C1-C5Alkylcarbonyl, halo C1-C5Alkylcarbonyl, halo C1-C5Alkylsulfonyl or R5(ii) a Or,
when Y is selected from NR4N R4And R3To form a morpholine, piperazine or piperidine ring;
R5selected from phenyl, benzyl, pyridyl, pyrimidyl, thiazolyl, oxazolyl or pyrazolyl, unsubstituted or substituted with 1-3 groups independently selected from: fluorine, chlorine, bromine, nitro, cyano, C1-C3Alkyl, halo C1-C3Alkyl radical, C1-C3Alkoxy or halo C1-C3An alkoxy group;
q is selected from one of the groups shown below:
R8selected from H, Cl, Br, F, CN, CH3、CH2CH3Tert-butyl, cyclopropyl, CF3、OCH3、OCF3、OCH2CF3、SO2CH3Unsubstituted or substituted phenyl, unsubstituted or substituted pyridyl, said substituents being selected from 1 to 3 of the following groups: cl, Br, F, CN, NO2、CH3、CH2CH3Tert-butyl, CHF2、CF3、OCH3、OCHF2、OCF3、OCH2CF3Or SO2CH3;
R9Selected from H, CH3、CH2CH3Tert-butyl, unsubstituted or substituted phenyl, unsubstituted or substituted pyridyl, said substituents being selected from 1 to 3 of the following groups: cl, Br, F, CN, NO2、CH3Or CF3;
a is selected from an integer of 1 to 4; b is an integer selected from 1 to 3; c is an integer selected from 1 to 2; d is selected from an integer from 1 to 2.
5. The compound of claim 4, wherein: in the general formula (I)
R1Selected from hydrogen or methyl; m is an integer selected from 1 to 2;
R2selected from fluorine, chlorine, bromine, iodine, cyano, nitro, C1-C3Alkyl or halo C1-C3An alkyl group; n is an integer from 0 to 2;
y is selected from O or NR4;
R3、R4Can be the same or different and are respectively selected from hydrogen and C1-C5Alkyl radical, C1-C5Alkoxy, cyano C1-C5Alkyl radical, C1-C5Alkylamino radical, C2-C6Dialkylamino radical, C3-C5Alkenyl radical, C3-C5Alkynyl, C1-C5Alkylsulfonyl radical, C1-C5Alkylcarbonyl or R5(ii) a Or,
when Y is selected from NR4N R4And R3To form a morpholine, piperazine or piperidine ring;
R5selected from phenyl, benzyl, pyridyl, pyrimidinyl, thiazolyl or pyrazolyl, unsubstituted or substituted with 1-2 groups independently selected from: fluorine, chlorine, bromine, nitro, cyano, C1-C3Alkyl, halo C1-C3Alkyl radical, C1-C3Alkoxy or halo C1-C3An alkoxy group;
q is selected from one of the groups shown below:
R8selected from H, Cl, Br, F, CN, CH3、CH2CH3Tert-butyl, CF3、OCH3、OCF3、SO2CH3Unsubstituted or substituted phenyl, unsubstituted or substituted pyridyl, said substituents being selected from 1 to 3 of the following groups: cl, Br, F, CN, NO2、CH3、CH2CH3Tert-butyl, CHF2、CF3、OCH3、OCHF2、OCF3Or SO2CH3;
R9Selected from H, CH3、CH2CH3Tert-butyl, unsubstituted or substituted phenyl, unsubstituted or substituted pyridyl, said substituents being selected from 1 to 3 of the following groups: cl, Br, F, CH3Or CF3;
a is selected from an integer of 1 to 3; b is an integer selected from 1 to 2; c is an integer selected from 1 to 2; d is selected from an integer from 1 to 2.
6. The compound of claim 5, wherein: in the general formula (I)
R1Selected from hydrogen or methyl; m is selected from 1;
R2selected from fluorine, chlorine, bromine or C1-C3An alkyl group; n is an integer from 0 to 2;
y is selected from O or NR4;
R3、R4May be the same or different and are each selected from hydrogen or C1-C3An alkyl group;
R5selected from phenyl, benzyl, pyridine or pyrimidinyl, unsubstituted or substituted with 1-2 groups independently selected from: fluorine, chlorine, bromine, nitro, cyano, C1-C3Alkyl, halo C1-C3Alkyl radical, C1-C3Alkoxy or halo C1-C3An alkoxy group;
q is selected from one of the groups shown below:
R8selected from H, Cl, Br, F, CN, CH3、CH2CH3、CF3、OCH3、OCF3、SO2CH3Unsubstituted or substituted phenyl, unsubstituted or substituted pyridyl, said substituents being selected from 1 to 3 of the following groups: cl, Br, F, NO2、CH3、CH2CH3、CF3Or OCH3;
R9Selected from H, CH3Unsubstituted or substituted phenyl, unsubstituted or substituted pyridyl, said substituents being selected from 1 to 3 of the following groups: cl, Br, F or CH3。
7. An intermediate phenylbenzylamine or a salt thereof for the preparation of a compound of formula (I), as shown in formula (II):
in the formula:
R1selected from hydrogen or C1-C12An alkyl group; m is an integer selected from 1 to 3;
R2selected from halogen, cyano, nitro, C1-C12Alkyl, halo C1-C12Alkyl radical, C1-C12Alkoxy, halo C1-C12Alkoxy radical, C1-C12Alkylthio radical, C1-C12Alkylsulfonyl or R5(ii) a n is an integer from 0 to 4;
y is selected from O, S or NR4;
R3、R4Can be the same or different and are respectively selected from hydrogen, cyano and C1-C12Alkyl, halo C1-C12Alkyl radical, C1-C12Alkoxy, halo C1-C12Alkoxy radical, C3-C6Cycloalkyl, cyano C1-C12Alkyl radical, C1-C12Alkylamino, halogeno C1-C12Alkylamino radical, C2-C6Dialkylamino radical, C2-C12Alkenyl, halo C2-C12Alkenyl radical, C2-C12Alkenyloxy, halogeno C2-C12Alkenyloxy radical, C2-C12Alkynyl, halo C2-C12Alkynyl, C2-C12Alkynyloxy, halo C2-C12Alkynyloxy, C1-C12Alkylthio, halo C1-C12Alkylthio radical, C1-C12Alkylsulfonyl radical, C1-C12Alkylcarbonyl, halo C1-C12Alkylcarbonyl, halo C1-C12Alkylsulfonyl or R5(ii) a Or,
when Y is selected from NR4N R4And R3Form substituted or unsubstituted pentaA member or six-membered ring, said substituents being selected from 1-2C1-C4An alkyl group;
R5selected from phenyl, benzyl or heteroaryl, unsubstituted or substituted with 1 to 4 groups independently selected from: halogen, nitro, cyano, C1-C12Alkyl, halo C1-C12Alkyl radical, C1-C12Alkoxy, halo C1-C12Alkoxy radical, C1-C12Alkylthio radical, C1-C12Alkylcarbonyl or C1-C12An alkoxycarbonyl group;
(CHR1) The connection position of mCONH and a benzene ring is selected from 2, 3 or 4 positions.
8. Use of the acyl benzylamine compound of the general formula (I) according to claim 1 for controlling germs in agriculture or other fields.
9. A germicidal composition, characterized by: containing as active ingredient a compound of general formula (I) according to any one of claims 1 to 6, the composition containing from 0.1 to 99% by weight of active ingredient.
10. Use of a composition according to claim 9 for controlling pathogens in agriculture or other fields.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201010535133.0A CN102464592B (en) | 2010-11-03 | 2010-11-03 | Acyl benzylamine compound and use thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201010535133.0A CN102464592B (en) | 2010-11-03 | 2010-11-03 | Acyl benzylamine compound and use thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN102464592A true CN102464592A (en) | 2012-05-23 |
| CN102464592B CN102464592B (en) | 2014-09-03 |
Family
ID=46068755
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201010535133.0A Expired - Fee Related CN102464592B (en) | 2010-11-03 | 2010-11-03 | Acyl benzylamine compound and use thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN102464592B (en) |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN103980203A (en) * | 2014-05-20 | 2014-08-13 | 浙江金伯士药业有限公司 | New preparation method of carnosine |
| CN104672101A (en) * | 2013-11-26 | 2015-06-03 | 中国中化股份有限公司 | o-chloroacetylamino benzoyl methylamine preparation method |
| CN106973990A (en) * | 2016-01-19 | 2017-07-25 | 中国科学院上海有机化学研究所 | A kind of fumigating method for being used to prevent and treat grain entomophthora |
| EP3377486A4 (en) * | 2015-11-18 | 2019-09-04 | Monsanto Technology LLC | INSECTICIDAL COMPOSITIONS AND ASSOCIATED METHODS |
| CN116239500A (en) * | 2023-03-13 | 2023-06-09 | 中国农业大学 | A kind of highly active abscisic acid functional analog (I) and preparation method thereof |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1642963A (en) * | 2002-03-27 | 2005-07-20 | 辛根塔参与股份公司 | Siliconized phenylamide compounds useful as microbicides |
| WO2007060164A1 (en) * | 2005-11-22 | 2007-05-31 | Bayer Cropscience Sa | N-(1-alkyl-2-phenylethyl)-carboxamide derivatives and use thereof as fungicides |
-
2010
- 2010-11-03 CN CN201010535133.0A patent/CN102464592B/en not_active Expired - Fee Related
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1642963A (en) * | 2002-03-27 | 2005-07-20 | 辛根塔参与股份公司 | Siliconized phenylamide compounds useful as microbicides |
| WO2007060164A1 (en) * | 2005-11-22 | 2007-05-31 | Bayer Cropscience Sa | N-(1-alkyl-2-phenylethyl)-carboxamide derivatives and use thereof as fungicides |
Cited By (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN104672101A (en) * | 2013-11-26 | 2015-06-03 | 中国中化股份有限公司 | o-chloroacetylamino benzoyl methylamine preparation method |
| CN103980203A (en) * | 2014-05-20 | 2014-08-13 | 浙江金伯士药业有限公司 | New preparation method of carnosine |
| CN103980203B (en) * | 2014-05-20 | 2015-11-18 | 浙江金伯士药业有限公司 | A kind of novel preparation method of carnosine |
| EP3377486A4 (en) * | 2015-11-18 | 2019-09-04 | Monsanto Technology LLC | INSECTICIDAL COMPOSITIONS AND ASSOCIATED METHODS |
| US10827755B2 (en) | 2015-11-18 | 2020-11-10 | Monsanto Technology Llc | Insecticidal compositions and methods |
| CN106973990A (en) * | 2016-01-19 | 2017-07-25 | 中国科学院上海有机化学研究所 | A kind of fumigating method for being used to prevent and treat grain entomophthora |
| CN106973990B (en) * | 2016-01-19 | 2019-12-10 | 中国科学院上海有机化学研究所 | fumigation method for preventing and controlling entomophthora in grains |
| CN116239500A (en) * | 2023-03-13 | 2023-06-09 | 中国农业大学 | A kind of highly active abscisic acid functional analog (I) and preparation method thereof |
| CN116239500B (en) * | 2023-03-13 | 2024-07-02 | 中国农业大学 | High-activity abscisic acid functional analogue (I) and preparation method thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| CN102464592B (en) | 2014-09-03 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN103081916B (en) | Application of pyrazole amide compound as agricultural bactericide | |
| CN106536517B (en) | Isoxazoline-containing uracil compound and application thereof | |
| EP0532022B1 (en) | Acrylate compound, preparation process thereof and fungicide using the same | |
| CN101668748A (en) | Substituted pyrimidine ether compound and application thereof | |
| CN102464592B (en) | Acyl benzylamine compound and use thereof | |
| CN101875639A (en) | Substituted pyrimidine ether compounds and their applications | |
| CN102336744A (en) | Substituted triazoline ether ketone class compound and application thereof as bactericide as well as insecticide and acaricide | |
| CN117069673B (en) | Oxadiazole compound and use thereof | |
| CN102336742A (en) | Substituted triazolinone ether compounds and application thereof | |
| CN102827071B (en) | Benzonitrile-substitute compound containing aminobenzonitrile and application cthereof | |
| CN106543139B (en) | Triazolone compound and application thereof | |
| CN102464618B (en) | Pyrazolecarboxamide compound and application thereof | |
| CN113045561A (en) | Diarylamine derivatives as fungicides | |
| CN111454202B (en) | Heteroaryl formanilide compound containing pentafluorothio and preparation method and application thereof | |
| CN102827034B (en) | P-dicyanoaniline-containing compounds and applications thereof | |
| CN103288771B (en) | Isothiazole compound and use thereof as fungicide | |
| JPH09268169A (en) | Salicylic acid anilide derivative and controlling agent for plant disease injury containing the same derivative as active ingredient | |
| CN116903604A (en) | Oxadiazole compound and application thereof | |
| EP4003976B1 (en) | Picolinamide derivatives useful as agricultural fungicides | |
| CN102993189B (en) | Hydrazone-containing triazolinone compounds and uses thereof | |
| CN110964037B (en) | Pyrimidine-fused ring-containing compound and preparation method and application thereof | |
| CN102827033A (en) | Compound containing o-dicyanoaniline and application thereof | |
| CN111655677A (en) | Benzimidazole compounds as agrochemicals | |
| JP2003096046A (en) | Diamine derivatives, production method therefor, germicides containing the derivatives as active ingredient | |
| JPS6239574A (en) | 1-aryl-5-hydrazino-pyrazoles |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| C41 | Transfer of patent application or patent right or utility model | ||
| TR01 | Transfer of patent right |
Effective date of registration: 20160121 Address after: 110021 Liaodong Road, Tiexi District, Liaoning, No. 8-1, No. Patentee after: SHENYANG SINOCHEM AGROCHEMICALS R&D Co.,Ltd. Address before: 100031 Beijing, Xicheng District, the door of the revitalization of the main street, No. 28 Patentee before: SINOCHEM Corp. Patentee before: SHENYANG RESEARCH INSTITUTE OF CHEMICAL INDUSTRY Co.,Ltd. |
|
| CF01 | Termination of patent right due to non-payment of annual fee |
Granted publication date: 20140903 |
|
| CF01 | Termination of patent right due to non-payment of annual fee |