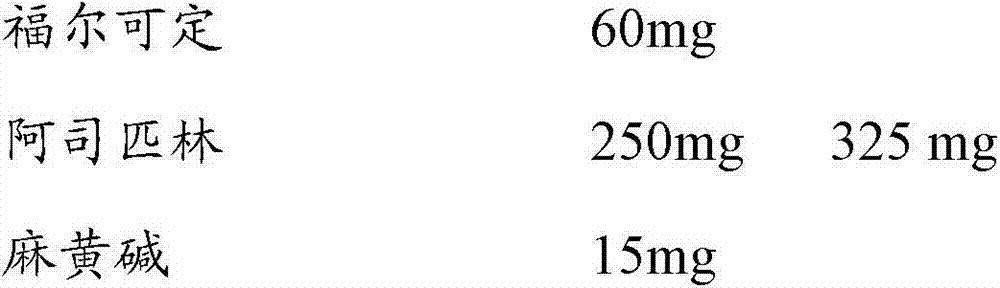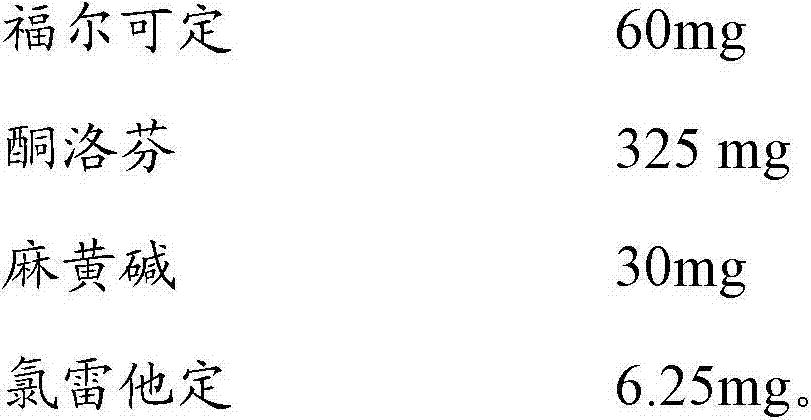CN102380101B - A compound medicinal preparation containing pholcodine for treating common cold - Google Patents
A compound medicinal preparation containing pholcodine for treating common cold Download PDFInfo
- Publication number
- CN102380101B CN102380101B CN201110331486.3A CN201110331486A CN102380101B CN 102380101 B CN102380101 B CN 102380101B CN 201110331486 A CN201110331486 A CN 201110331486A CN 102380101 B CN102380101 B CN 102380101B
- Authority
- CN
- China
- Prior art keywords
- preparation
- pholcodine
- pellets
- pack
- acetaminophen
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
Images
Landscapes
- Medicinal Preparation (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
Abstract
Description
本申请是2009年11月13日提交的申请号为200910222919.4、发明名称为“一种含福尔可定的用于治疗感冒的复方药物制剂”申请的分案申请。This application is a divisional application of the application number 200910222919.4 and the title of the invention "a compound pharmaceutical preparation containing pholcodine for treating colds" submitted on November 13, 2009.
技术领域technical field
本发明涉及药物制剂领域,特别涉及一种含福尔可定的用于治疗感冒的复方药物制剂。The invention relates to the field of pharmaceutical preparations, in particular to a compound pharmaceutical preparation containing pholcodine for treating colds.
背景技术Background technique
感冒,是由多种病毒引起的呼吸道常见病,有着较高的发病率。目前比较实用的治疗方法是通过解热镇痛药,减鼻充血剂,抗组胺药以及中枢镇咳药的联用来减轻感冒症状--由感冒引起的发热,头痛,四周四肢酸痛,喷嚏,流涕,鼻塞,咳嗽等。Influenza is a common respiratory disease caused by a variety of viruses, and has a high incidence rate. At present, the more practical treatment method is to relieve cold symptoms through the combination of antipyretic and analgesics, nasal decongestants, antihistamines and central cough suppressants--fever caused by colds, headache, sore limbs, sneezing , runny nose, stuffy nose, cough, etc.
中枢镇咳药镇咳作用强大,并有轻微的镇静镇痛的作用;解热镇痛药,在缓解疼痛的同时有退热功效;减充血剂,可以减轻鼻子堵塞症状并缓解咳嗽;抗组胺药,可以缓解感冒中的喷嚏等过敏症状,而且此类药有嗜睡的特点,若在夜晚服用含此类药物的制剂可以使患者有比较好的睡眠,能更好地缓解疲劳等症状。Central antitussives have strong antitussive effect and have mild sedative and analgesic effects; antipyretic and analgesic drugs have antipyretic effect while relieving pain; decongestants can relieve nasal congestion symptoms and relieve cough; Amine medicines can relieve allergic symptoms such as sneezing in colds, and these medicines have the characteristics of drowsiness. If the preparations containing such medicines are taken at night, patients can have better sleep and relieve fatigue and other symptoms better.
目前在市面上应用比较多的中枢镇咳药是磷酸可待因和氢溴酸右美沙芬:磷酸可待因的有着比较大的成瘾性,现市场上多次出现滥用的事故而受到国家食品药品监督管理局的严格控制;氢溴酸右美沙芬的镇咳效果好,但现在呈现的不良反应事例越来越多,而且也出现大量的滥用事件,美国食品药品监督管理局时刻关注右美沙芬的滥用情况,并发出不要滥用右美沙芬的警告。福尔可定也为中枢镇咳药,具有与磷酸可待因相似的中枢镇咳作用,也有镇静和镇痛作用,但成瘾性较磷酸可待因弱,新生儿与儿童耐受此药。且此药吸收快消除较慢,对减少服药次数,安全用药有积极作用。At present, the central antitussive drugs that are widely used in the market are codeine phosphate and dextromethorphan hydrobromide: codeine phosphate has a relatively high degree of addiction, and there have been many abuse accidents in the market and have been criticized by the state. Strict control by the Food and Drug Administration; dextromethorphan hydrobromide has a good antitussive effect, but now there are more and more cases of adverse reactions, and there are also a large number of abuse incidents. The U.S. Food and Drug Administration always pays attention to the right Abuse of dextromethorphan and warnings not to abuse dextromethorphan. Pholcodine is also a central antitussive drug, which has a central antitussive effect similar to codeine phosphate, and also has sedative and analgesic effects, but its addiction is weaker than codeine phosphate, and newborns and children tolerate this drug . Moreover, the drug is absorbed quickly and eliminated slowly, which has a positive effect on reducing the number of times of taking the drug and ensuring safe drug use.
专利CN200410040448.2“治疗无痰咳嗽的感冒药组合物及其加工方法”是国内目前唯一的一项有关福尔可定的专利,专利采用福尔可定、盐酸伪麻黄碱、马来酸氯苯那敏、95%药用乙醇及纯化水配制成药剂。该专利只能用于止咳平喘,而对于感冒引起的发热,头痛,四周四肢酸痛,喷嚏,流涕,鼻塞不能有效缓解。同时由于含有马来酸氯苯那敏,日用时产生很强的副作用。Patent CN200410040448.2 "Cold Medicine Composition for Treating Phlegmless Cough and Its Processing Method" is currently the only patent related to pholcodine in China. The patent uses pholcodine, pseudoephedrine hydrochloride, and chlorpheniramine maleate Sensitivity, 95% medicinal ethanol and purified water are prepared into medicaments. This patent can only be used for relieving cough and relieving asthma, but for fever caused by cold, headache, sore limbs, sneezing, runny nose, nasal congestion cannot be effectively relieved. Simultaneously owing to contain chlorpheniramine maleate, produce very strong side effect during daily use.
发明内容Contents of the invention
本发明的目的是提供一种以福尔可定为中枢镇咳药的复方制剂,所述制剂能有效而迅速的治疗感冒和流行性感冒。The object of the present invention is to provide a compound preparation using pholcodine as a central antitussive drug, which can effectively and rapidly treat colds and influenza.
本发明的治疗感冒和流行感冒症状的药物制剂,其特征在于,所述药物制剂包括:福尔可定,解热镇痛剂和减充血剂。The pharmaceutical preparation for treating cold and flu symptoms of the present invention is characterized in that the pharmaceutical preparation comprises: pholcodine, antipyretic analgesic and decongestant.
本发明的药物制剂中还含有抗组胺剂。Antihistamines are also included in the pharmaceutical preparations of the invention.
本发明的药物制剂,其单位剂量的制剂组成如下:Pharmaceutical preparation of the present invention, the preparation of its unit dose is composed as follows:
福尔可定 1-60mgPholcodine 1-60mg
解热镇痛剂 50-4000mgAntipyretic analgesic 50-4000mg
减充血剂 5-400mgDecongestants 5-400mg
或者or
本发明的复方制剂分为日用制剂和夜用制剂。The compound preparation of the present invention is divided into daily preparation and night preparation.
所述的日用制剂包括:福尔可定,解热镇痛剂,减充血剂。日用制剂用于缓解白天感冒和流感症状。The daily preparation includes: pholcodine, antipyretic analgesic, and decongestant. Daily formulation for the relief of cold and flu symptoms during the day.
所述的夜用制剂包括:福尔可定,解热镇痛剂,减充血剂和抗组胺剂。夜用制剂除了缓解感冒和流感症状外还可帮助改善睡眠。The nighttime preparations include: pholcodine, antipyretic analgesics, decongestants and antihistamines. The nighttime formulation can help improve sleep in addition to relieving cold and flu symptoms.
本发明的日用制剂,配方组成如下:Daily preparation of the present invention, formula is composed as follows:
福尔可定 1-60mgPholcodine 1-60mg
解热镇痛剂 50-4000mgAntipyretic analgesic 50-4000mg
减充血剂 5-400mgDecongestants 5-400mg
本发明的夜用制剂,配方组成如下:The night preparation of the present invention has a formula as follows:
其中所述的解热镇痛药选自对乙酰氨基酚、阿司匹林、布洛芬或酮洛芬。优选对乙酰氨基酚。Wherein said antipyretic and analgesic drugs are selected from paracetamol, aspirin, ibuprofen or ketoprofen. Acetaminophen is preferred.
所述的减充血剂选自:麻黄碱、伪麻黄碱或其药学上可接受的盐。优选盐酸伪麻黄碱。The decongestant is selected from: ephedrine, pseudoephedrine or pharmaceutically acceptable salts thereof. Pseudoephedrine hydrochloride is preferred.
所述的抗组胺剂选自:苯海拉明、氯苯那敏、曲吡那敏、异丙嗪、氯雷他定、西替利嗪或其药学上可接受的盐。优选盐酸苯海拉明。The antihistamine is selected from the group consisting of diphenhydramine, chlorpheniramine, tripyramine, promethazine, loratadine, cetirizine or pharmaceutically acceptable salts thereof. Diphenhydramine hydrochloride is preferred.
本发明优选的日用制剂,配方组成如下:The preferred daily preparation of the present invention, formula is as follows:
福尔可定 1-60mgPholcodine 1-60mg
乙酰氨基酚 50-4000mgAcetaminophen 50-4000mg
盐酸伪麻黄碱 5-400mg。Pseudoephedrine hydrochloride 5-400mg.
本发明优选的夜用制剂,配方组成如下:The preferred night-use preparation of the present invention has a formula as follows:
本发明进一步优选的日用制剂,配方组成如下:The further preferred daily preparation of the present invention, formula composition is as follows:
福尔可定 1-30mgPholcodine 1-30mg
乙酰氨基酚 50-750mgAcetaminophen 50-750mg
盐酸伪麻黄碱 10-300mg。Pseudoephedrine hydrochloride 10-300mg.
本发明进一步优选的夜用制剂,配方组成如下:The further preferred night-use preparation of the present invention, formula composition is as follows:
本发明特别优选的日用制剂,配方组成如下:The particularly preferred daily preparation of the present invention, formula composition is as follows:
福尔可定 1-15mgPholcodine 1-15mg
乙酰氨基酚 50-650mgAcetaminophen 50-650mg
盐酸伪麻黄碱 1-50mg。Pseudoephedrine hydrochloride 1-50mg.
本发明特别优选的夜用制剂,配方组成如下:The particularly preferred night-use preparation of the present invention, formula composition is as follows:
本发明最优选的复方制剂在实施例中。The most preferred compound preparation of the present invention is in the embodiment.
本发明的复方制剂的配方中可以加入一或多种药用添加剂。One or more pharmaceutical additives can be added into the formula of the compound preparation of the present invention.
所述药用添加剂包括包括但不限于稀释剂,填充剂、粘合剂,粘附剂、润滑剂、助流剂、崩解剂、表面活性剂、载体溶剂、增塑剂、稀释剂、缓冲剂、抑菌剂、防腐剂、着色剂、矫味剂、稳定剂和其它本领域中已知的药用添加剂。The pharmaceutical additives include but not limited to diluents, fillers, binders, adhesives, lubricants, glidants, disintegrants, surfactants, carrier solvents, plasticizers, diluents, buffers agents, antibacterial agents, preservatives, coloring agents, flavoring agents, stabilizers and other pharmaceutical additives known in the art.
润滑剂包括但不限于硬脂酸镁、硬脂酸钙、硬脂酸锌、粉末硬脂酸、单硬脂酸甘油酯、硬脂酸棕榈酸甘油酯、硅胶、硅酸镁、二氧化硅、二氧化钛、苯甲酸钠、十二烷基硫酸钠、硬脂酰富马酸钠、氮化植物油、滑石粉、聚乙二醇和矿物油。Lubricants include, but are not limited to, magnesium stearate, calcium stearate, zinc stearate, powdered stearic acid, glyceryl monostearate, glyceryl palmitostearate, silica gel, magnesium silicate, silicon dioxide , titanium dioxide, sodium benzoate, sodium lauryl sulfate, sodium stearyl fumarate, nitrogenated vegetable oil, talc, macrogol and mineral oil.
表面活性剂包括但不限于十二烷基硫酸钠、二辛基琥珀酸磺酸钠、三乙醇胺、聚氧乙烯脱水山梨醇;赋形剂,例如乳糖、甘露醇、葡萄糖、果糖、木糖、半乳糖、蔗糖、麦芽糖、木糖醇、山梨醇以及钾、钠和镁的氯化物、硫酸盐和磷酸盐;凝胶剂,例如胶质粘土;增稠剂,例如藻酸钠,泡腾混合物;和润湿剂,例如卵磷脂、聚山梨醇酯或十二烷基硫酸盐类。Surfactants include, but are not limited to, sodium lauryl sulfate, sodium dioctyl sulfosuccinate, triethanolamine, polyoxyethylene sorbitan; excipients such as lactose, mannitol, glucose, fructose, xylose, Galactose, sucrose, maltose, xylitol, sorbitol, and chlorides, sulfates, and phosphates of potassium, sodium, and magnesium; gelling agents, such as colloidal clay; thickening agents, such as sodium alginate, effervescent mixtures and wetting agents such as lecithin, polysorbates or lauryl sulfates.
着色剂可以用于改善外观,或者由于识别药用组合物。如食用色素。Colorants can be used to improve appearance, or to identify pharmaceutical compositions. Such as food coloring.
在其中药用组合物压制成固体剂型(例如片剂)的实施方案中,粘合剂可以有助于将各个成分粘结在一起。粘合剂包括但不限于糖类,例如蔗糖、乳糖和葡萄糖;复合糖浆;大豆多糖;明胶;聚维酮;纤维素衍生物,例如微晶纤维素、羟丙基甲基纤维素、羟丙基纤维素、乙基纤维素、乙基纤维素、羧甲基纤维素钠和甲基纤维素;丙烯酸和甲基丙烯酸共聚物;卡波姆;聚乙烯聚吡咯烷;聚乙二醇;药用糖浆;藻酸盐,例如藻酸和藻酸钠;胶类,例如阿拉伯树胶、瓜耳胶和阿拉伯胶;黄茂胶;糊精和麦芽糊精;奶的衍生物,例如乳清;淀粉类,例如预胶化淀粉和淀粉浆;氢化植物油;硅酸铝镁,以及其它本领域技术人员已知的常规粘合剂。典型的非限定性填充物包括糖、乳糖、明胶、淀粉和二氧化硅。In embodiments where the pharmaceutical composition is compressed into a solid dosage form such as a tablet, a binder can help to hold the ingredients together. Binders include, but are not limited to, sugars such as sucrose, lactose, and glucose; complex syrups; soybean polysaccharides; gelatin; povidone; cellulose derivatives such as microcrystalline cellulose, hydroxypropylmethylcellulose, hydroxypropyl Acrylic acid and methacrylic acid copolymers; Carbomer; Polyvinylpolypyrrolidine; Polyethylene glycol; Syrups; alginates, such as alginic acid and sodium alginate; gums, such as acacia, guar and acacia; tragacanth; dextrins and maltodextrins; milk derivatives, such as whey; starches Such as pregelatinized starch and starch slurry; hydrogenated vegetable oil; magnesium aluminum silicate, and other conventional binders known to those skilled in the art. Typical, non-limiting fillers include sugar, lactose, gelatin, starch and silicon dioxide.
助流剂可以改善非压制的固体剂型的流动性,可以提高定量投料的准确性。助流剂包括但不限于二氧化硅、硅胶、滑石粉、三硅酸镁、硬脂酸镁或钙、微粉纤维素、淀粉和磷酸三钙。Glidants can improve the fluidity of non-compressed solid dosage forms, and can improve the accuracy of quantitative feeding. Glidants include, but are not limited to, silicon dioxide, silica gel, talc, magnesium trisilicate, magnesium or calcium stearate, micronized cellulose, starch and tricalcium phosphate.
增塑剂包括但不限于疏水性和/或亲水性增塑剂,例如邻苯二甲酸二乙酯、邻苯二甲酸丁酯、癸二酸二乙酯、癸二酸二丁酯、柠檬酸三乙酯、柠檬酸乙酰三乙酯、檬酸乙酰三丁酯、丙二醇、蓖麻油、乙酸甘油酯、聚乙二醇、丙二醇、甘油和山梨醇。增塑剂特别适用于含有聚合物的药用组合物以及在软胶囊中的药用组合物和薄膜一包衣片剂。矫味剂改善适口性,特别适用于咀嚼片或液体剂型。矫味剂包括但不限于麦芽糖醇、香草醛、乙基香草醛、薄荷醇、柠檬酸、富马酸、乙基麦芽糖醇和酒石酸。甜味剂包括但不限于山梨醇、糖精、糖精钠、蔗糖、阿斯巴甜、果糖、甘露醇和转化糖。Plasticizers include, but are not limited to, hydrophobic and/or hydrophilic plasticizers such as diethyl phthalate, butyl phthalate, diethyl sebacate, dibutyl sebacate, lemon Triethyl Citrate, Acetyl Triethyl Citrate, Acetyl Tributyl Citrate, Propylene Glycol, Castor Oil, Glyceryl Acetate, Polyethylene Glycol, Propylene Glycol, Glycerin and Sorbitol. Plasticizers are particularly useful in pharmaceutical compositions containing polymers and pharmaceutical compositions in soft capsules and film-coated tablets. Flavoring agents improve palatability, especially for chewable tablet or liquid dosage forms. Flavoring agents include, but are not limited to, maltitol, vanillin, ethyl vanillin, menthol, citric acid, fumaric acid, ethyl maltitol, and tartaric acid. Sweeteners include, but are not limited to, sorbitol, saccharin, sodium saccharin, sucrose, aspartame, fructose, mannitol, and invert sugar.
防腐剂和/或稳定剂能够改善储存性,包括但不限于乙醇、苯甲酸钠、丁基化羟基甲苯、丁基化对羟基筒香醚和乙二胺四乙酸。Preservatives and/or stabilizers to improve shelf life include, but are not limited to, ethanol, sodium benzoate, butylated hydroxytoluene, butylated p-hydroxytoluene, and ethylenediaminetetraacetic acid.
崩解剂可以提高药用组合物的溶出速率。崩解剂包括但不限于藻酸盐(例如藻酸和藻酸钠)、羧甲基纤维素钙、羧甲基纤维素钠、胶体二氧化硅、交联羧甲基纤维素钠、交联聚维酮、聚乙烯聚吡咯烷、瓜尔胶、硅酸铝镁、甲基纤维素、微晶纤维素、波拉克林钾、粉末纤维素、淀粉、预胶化淀粉、羟乙酸淀粉钠。Disintegrants can increase the dissolution rate of pharmaceutical compositions. Disintegrants include, but are not limited to, alginates (such as alginic acid and sodium alginate), calcium carboxymethylcellulose, sodium carboxymethylcellulose, colloidal silicon dioxide, croscarmellose sodium, crosslinked Povidone, polyvinylpolypyrrolidine, guar gum, magnesium aluminum silicate, methylcellulose, microcrystalline cellulose, polacrilin potassium, powdered cellulose, starch, pregelatinized starch, sodium starch glycolate.
稀释剂能够增加剂型的体积,可以使得剂型更易于处理。用于固体剂型(例如片剂和胶囊)的典型的稀释剂包括但不限于乳糖、葡萄糖、蔗糖、纤维素、淀粉和磷酸钙;用于软胶囊的橄榄油和油酸乙酯;用于液体剂型(例如混悬液和乳剂)的水和植物油。其它适当的稀释剂包括但不限于蔗糖、葡聚糖、糊精、麦芽糊精、微晶纤维素、微粉纤维素、粉末纤维素、预胶化淀粉、磷酸钙二水合物、大豆多糖、明胶、二氧化硅、硫酸钙、碳酸钙、碳酸镁、氧化镁、山梨醇、甘露醇、高岭土、聚甲基丙烯酸酯、氯化钾、氯化钠和滑石粉。Diluents can increase the bulk of the dosage form, which can make the dosage form easier to handle. Typical diluents for solid dosage forms such as tablets and capsules include, but are not limited to, lactose, dextrose, sucrose, cellulose, starch, and calcium phosphate; olive oil and ethyl oleate for soft capsules; water and vegetable oils for dosage forms such as suspensions and emulsions. Other suitable diluents include, but are not limited to, sucrose, dextran, dextrin, maltodextrin, microcrystalline cellulose, micronized cellulose, powdered cellulose, pregelatinized starch, calcium phosphate dihydrate, soybean polysaccharide, gelatin , Silica, Calcium Sulfate, Calcium Carbonate, Magnesium Carbonate, Magnesium Oxide, Sorbitol, Mannitol, Kaolin, Polymethacrylate, Potassium Chloride, Sodium Chloride, and Talc.
在其中将复方制剂制成液体剂型的实施方案中,药用组合物可以包含一或多种溶剂。适当的溶剂包括但不限于水、醇类(例如乙醇和异丙醇)、植物油、聚乙二醇、丙二醇和甘油或其混合和组合。In embodiments wherein the combination formulation is presented as a liquid dosage form, the pharmaceutical composition may comprise one or more solvents. Suitable solvents include, but are not limited to, water, alcohols (eg, ethanol and isopropanol), vegetable oils, polyethylene glycol, propylene glycol, and glycerin, or mixtures and combinations thereof.
根据本发明,复方制剂可以含有缓冲剂。缓冲剂包括但不限于乳酸、柠檬酸、乙酸、乳酸钠、柠檬酸钠和乙酸钠。According to the present invention, the compound preparation may contain a buffering agent. Buffering agents include, but are not limited to, lactic acid, citric acid, acetic acid, sodium lactate, sodium citrate, and sodium acetate.
适用于缓释制剂的亲水性聚合物包括:一或多种天然或者部分或全部合成的亲水性胶类,例如阿拉伯胶、黄茂胶;改良的纤维素物质,例如甲基纤维素、羟甲基纤维素、羟丙基甲基纤维素、羟丙基纤维素、羟乙基纤维素、羧甲基纤维素;蛋白类似物,例如琼脂、果胶和藻酸盐类;其它亲水性聚合物例如卡波姆、明胶、酪蛋白、玉米蛋白、硅酸铝镁、多糖、改良淀粉衍生物和其它本领域技术人员已知的亲水性聚合物或此类聚合物的组合。Hydrophilic polymers suitable for sustained release formulations include: one or more natural or partially or fully synthetic hydrophilic gums, such as gum arabic, tragacanth; modified cellulosic substances, such as methylcellulose, Hydroxymethylcellulose, hydroxypropylmethylcellulose, hydroxypropylcellulose, hydroxyethylcellulose, carboxymethylcellulose; protein analogs such as agar, pectin, and alginates; other hydrophilic Hydrophilic polymers such as carbomer, gelatin, casein, zein, magnesium aluminum silicate, polysaccharides, modified starch derivatives and other hydrophilic polymers or combinations of such polymers known to those skilled in the art.
本发明的复方制剂可以制成任何可药用的剂型,此药用剂型适合阻滞配方并保证活性化合物以本发明方式释放。优选的剂型为:糖浆剂、口服液,颗粒剂,混悬剂,滴剂,片剂,胶囊,粉剂,咀嚼片,分散片,缓释片,缓释胶囊,微丸,凝胶制剂等。这些用药形式均经口服达到临床效果。The compound preparation of the present invention can be made into any pharmaceutically acceptable dosage form, which is suitable for retarding the formulation and ensuring the release of the active compound in the manner of the present invention. Preferred dosage forms are: syrup, oral liquid, granule, suspension, drop, tablet, capsule, powder, chewable tablet, dispersible tablet, sustained-release tablet, sustained-release capsule, pellets, gel preparation, etc. These medication forms all achieve clinical effects through oral administration.
本发明的另一目的在于提供本发明的复方制剂的制备方法。Another object of the present invention is to provide a preparation method of the compound preparation of the present invention.
本发明的复方制剂可制成口服制剂,如:口服液,糖浆,颗粒剂,混悬剂,滴剂,凝胶制剂等口服液体类制剂,其单位剂量为每1ml含药用组合物的量。口服液体类制剂采用溶解稀释法,然后利用适合的添加剂与方法将液体分装成例如糖浆剂。The compound preparation of the present invention can be made into oral preparations, such as: oral liquids, syrups, granules, suspensions, drops, gel preparations and other oral liquid preparations, and the unit dose is the amount of the pharmaceutical composition per 1ml . Oral liquid preparations are prepared by dissolving and diluting, and then the liquid is subpackaged into, for example, a syrup with suitable additives and methods.
本发明采用溶解稀释法制得的制剂,在成份混合时最好在升高的温度下进行,可以是25至100℃,优选40至60℃。各组份应维持在溶液中,以达成本发明的益处,且该溶液应在一定时间内,并在消费者应用时正常遇到的条件下维持稳定。例如,已经在室温下放置该溶液一定时间期间,仍维持澄清和稳定,没有活性成份的沉淀。此外,已经使该溶液接受交替的冷冻和室温条件,而活性成份并未形成结晶,且仍澄清和稳定。The preparations prepared by the method of dissolution and dilution according to the present invention are preferably carried out at an elevated temperature during the mixing of the ingredients, which may be from 25 to 100°C, preferably from 40 to 60°C. The components should remain in solution to achieve the benefits of the present invention, and the solution should be stable over time and under the conditions normally encountered in consumer use. For example, having stood at room temperature for a period of time, the solution remains clear and stable without precipitation of the active ingredient. Furthermore, the solution has been subjected to alternating freezing and room temperature conditions without crystallization of the active ingredient and remains clear and stable.
本发明的复方制剂也可以制成其他剂型的口服制剂,如:片剂,胶囊,咀嚼片,分散片,缓释片,缓释胶囊,微丸等口服固体制剂,其单位剂量为每片、粒、袋所含药用组合物的量。The compound preparation of the present invention can also be made into oral preparations of other dosage forms, such as: tablets, capsules, chewable tablets, dispersible tablets, sustained-release tablets, sustained-release capsules, and oral solid preparations such as micropills. The unit dose is each tablet, The amount of pharmaceutical composition contained in granules and bags.
口服固体制剂采用喷雾造料法和挤压造粒技术制造药物的方法特别有利,然后利用适合的添加剂与方法压制成例如片剂或分装成例如胶囊剂。在一个特别优选实施方案中,采用了挤出滚圆法制造药物制剂,其主要流程为配料、混合制粒、挤条成形、离心滚圆、筛分、干燥,在实施方案中通过专业设备达到目的,如湿法混合制粒机、挤条机、离心滚圆机等。Oral solid preparations are particularly advantageous in which the drug is manufactured by spraying and extrusion granulation techniques, and then compressed into, for example, tablets or subpackaged into, for example, capsules using suitable additives and methods. In a particularly preferred embodiment, the extrusion spheronization method is used to manufacture pharmaceutical preparations, the main processes of which are compounding, mixing and granulation, extrusion forming, centrifugal spheronization, sieving, and drying. In the embodiment, the purpose is achieved through professional equipment. Such as wet mixing granulator, extrusion machine, centrifugal spheronizer, etc.
挤出滚圆法为近年新近建立的一种生产方法。采用挤出滚圆法制造的药物为微丸,有别于一般的挤压法制造的药物颗粒,载药量高是其一大优势,国内外报道,采用包衣锅制丸法、流化床制丸法或其它方法制备的微丸载药量一般为20~50%,而采用挤出滚圆技术制备的微丸,载药量可达80%以上。并且可根据品种需求进行防潮避光的包衣处理。本领域技术人员可充公理解,为了生产具有所需特性的产物,于挤出滚圆法过程中可改变各种参数,例如原料粒度、水分、微丸粒径、挤出速度、滚圆速度、滚圆时间等。实施例部分提供根据本发明的制剂的多种实施例,其是利用挤出滚圆法制得的。The extrusion spheronization method is a newly established production method in recent years. The drug produced by the extrusion spheronization method is a pellet, which is different from the drug granules produced by the general extrusion method. The high drug loading is one of its major advantages. According to reports at home and abroad, the pellets made by the coating pot and the fluidized bed are used. The drug loading of pellets prepared by pelleting method or other methods is generally 20-50%, while the drug loading of pellets prepared by extrusion and spheronization technology can reach more than 80%. And it can be coated with moisture-proof and light-proof coating according to the needs of varieties. Those skilled in the art can fully understand that in order to produce products with desired characteristics, various parameters can be changed during the extrusion spheronization process, such as raw material particle size, moisture content, pellet size, extrusion speed, spheronization speed, spheronization time wait. The Examples section provides various examples of formulations according to the invention, which were prepared using extrusion spheronization.
上述参数将由所使用的专业设备及药物性质、生产环境而决定。实施例中,原料粒度可为50至200目,优选为100目。水分因包装材料的不同而决定,实施例中选用胶囊,因本品种盐酸苯海拉明吸湿严重,故将水分控制在3%以下。常选择合适的微丸粒径,提高微丸堆密度,满足装量要求,优选微丸粒径约为0.1mm至1.5mm,特别优选0.5mm。本领域技术人员可充分理解,挤出速度、滚圆速度、滚圆时间对药物质量影响很大,挤出速度可以是10至200rpm,优选55rpm。滚圆速度可以是100至2000rpm,优选600至800rpm,滚圆时间可以为0.5min至3min,优选2min。The above parameters will be determined by the professional equipment used, the nature of the drug, and the production environment. In an embodiment, the particle size of the raw material may be 50 to 200 mesh, preferably 100 mesh. Moisture is determined because of the difference of packing material, selects capsule for use in the embodiment, because this kind diphenhydramine hydrochloride is hygroscopic seriously, so moisture is controlled below 3%. Often select the appropriate particle size of the pellets to increase the bulk density of the pellets and meet the loading requirements. The preferred particle size of the pellets is about 0.1mm to 1.5mm, especially preferably 0.5mm. Those skilled in the art can fully understand that the extrusion speed, spheronization speed, and spheronization time have a great influence on the quality of the drug, and the extrusion speed can be 10 to 200 rpm, preferably 55 rpm. The spheronization speed can be 100 to 2000 rpm, preferably 600 to 800 rpm, and the spheronization time can be 0.5 min to 3 min, preferably 2 min.
本领域技术人员知道上述参数皆取决于特殊生产条件(生产环境,设备种类,物料性质,物料数量等),可能有必要加以修改,以使通过挤压法或挤出滚圆法制得的制剂提供持续、独立且稳定的释放,及上述贮存稳定性。Those skilled in the art know that the above-mentioned parameters all depend on special production conditions (production environment, equipment type, material properties, material quantity, etc.), and it may be necessary to modify them so that the preparations obtained by extrusion or extrusion spheronization provide continuous , independent and stable release, and the aforementioned storage stability.
本发明的复方制剂分为日用制剂和夜用制剂。The compound preparation of the present invention is divided into daily preparation and night preparation.
本发明的复方制剂可以单独日用制剂包装、单独夜用制剂包装及日用与夜用制剂混合包装。同样,本发明的复方制剂还可以包含或者伴有标记,它使得人们能够识别作为用于处方治疗产品的复方制剂。例如,为了区别日用与夜用制剂混合包装,可以赋予日用和夜用制剂不同颜色或立体几何构形等。The compound preparation of the present invention can be packaged individually for daily use, for night use alone or as a mixture of day and night preparations. Likewise, the combination preparations of the present invention may also contain or be accompanied by indicia that allow one to identify the combination preparations as products for the prescribed treatment. For example, in order to distinguish the mixed packaging of day-use and night-use preparations, the day-use and night-use preparations can be endowed with different colors or three-dimensional geometric configurations.
本发明的复方制剂能有效而迅速的治疗感冒和流行性感冒,如:感冒引起的发热、头痛、周身四肢酸痛、喷嚏、流涕、鼻塞、咳嗽等。The compound preparation of the invention can effectively and rapidly treat colds and influenza, such as: fever, headache, soreness in limbs, sneezing, runny nose, nasal congestion, coughing, etc. caused by colds.
本发明的复方制剂与现有技术相比,具有协同作用,治疗效果好,见效快,耐药性小,药用安全性高,副作用少,稳定性好,保值期长,携带方便,价格便宜,制备操作简单,适合大规模生产等特点。Compared with the prior art, the compound preparation of the present invention has synergistic effect, good therapeutic effect, quick effect, low drug resistance, high medicinal safety, few side effects, good stability, long value preservation period, convenient portability and low price , simple preparation and operation, suitable for large-scale production and the like.
附图说明Description of drawings
图1:实施例4中处方1日用制剂的测试曲线图Fig. 1: the test curve chart of prescription 1 daily use preparation in embodiment 4
图2:实施例4中处方2日用制剂的测试曲线图Fig. 2: the test graph of prescription 2 daily use preparations in embodiment 4
图3:实施例4中处方1夜用制剂的测试曲线图Fig. 3: the test curve graph of prescription 1 night use preparation in embodiment 4
图4:实施例4中处方2夜用制剂的测试曲线图Fig. 4: the test curve graph of prescription 2 night use preparations in embodiment 4
具体实施方式Detailed ways
通过以下实施例对本发明作一步说明,但不作为本发明的限制。The present invention is further illustrated by the following examples, but not as a limitation of the present invention.
实施例1利用喷雾造粒法制得的含有不同量的复方片剂Embodiment 1 utilizes the compound tablet that spray granulation method makes to contain different amounts
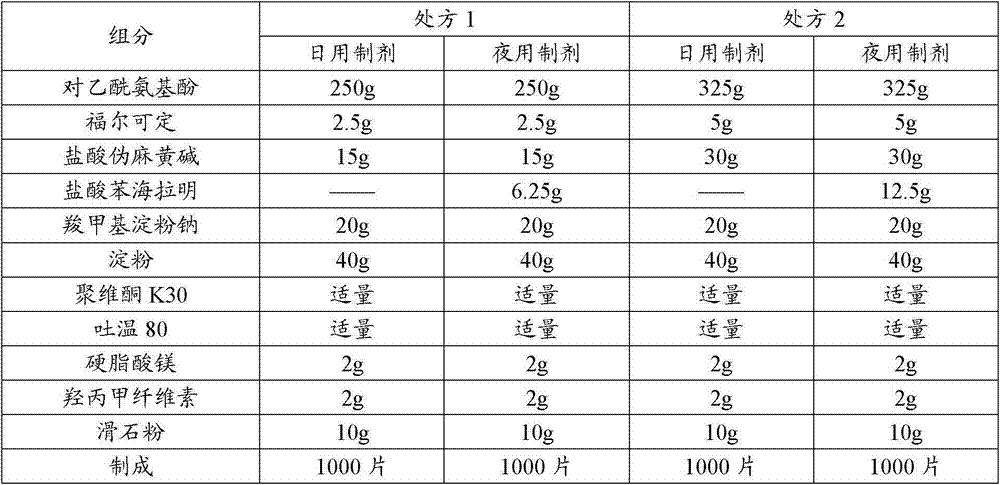
表1、本发明的复方片剂组成Table 1, compound tablet of the present invention form
制备方法Preparation
10万级洁净区内In a 100,000-class clean area
A将福尔可定,对乙酰氨基酚,盐酸伪麻黄碱、盐酸苯海拉明(夜用)过100目筛;A crosses 100 mesh sieves with pholcodine, paracetamol, pseudoephedrine hydrochloride, diphenhydramine hydrochloride (night use);
B领取少量淀粉配浆,加一半处方量吐温80搅拌,制成粘合剂;B receives a small amount of starch for mixing, adds half of the prescription amount of Tween 80 and stirs to make an adhesive;
C取处方量聚维酮加纯化水及吐温80搅拌,制成黏合剂;C takes the prescribed amount of povidone, adds purified water and Tween 80 and stirs to make an adhesive;
D将对乙酰氨基酚,盐酸伪麻黄碱、盐酸苯海拉明(夜用)及淀粉、羧甲基淀粉钠投入沸腾制粒机中预混合;D paracetamol, pseudoephedrine hydrochloride, diphenhydramine hydrochloride (night use) and starch, sodium carboxymethyl starch are put into premixed boiling granulator;
E先后将粘合剂和黏合剂用蠕动泵喷入沸腾制粒机中,喷完关毕蠕动泵。干燥,控制水分3%;E successively spray the adhesive and the adhesive into the boiling granulator with a peristaltic pump, and turn off the peristaltic pump after spraying. Dry, control moisture 3%;
D18目整粒,加入硬脂酸镁混合,压片即得。D18 mesh granules, mixed with magnesium stearate, compressed into tablets.
实施例2利用挤压造粒法制得的复方片剂Embodiment 2 utilizes the compound tablet that extrusion granulation method makes
表2、本发明的复方片剂。Table 2, compound tablet of the present invention.
制备方法Preparation
10万级洁净区内In a 100,000-class clean area
A将福尔可定,对乙酰氨基酚,盐酸伪麻黄碱、盐酸苯海拉明(夜用)过100目筛;A crosses 100 mesh sieves with pholcodine, paracetamol, pseudoephedrine hydrochloride, diphenhydramine hydrochloride (night use);
B称取处方用量的主药及微晶纤维素,预交化淀粉,PVPP,混合均匀;B takes the main ingredient and microcrystalline cellulose of prescription dosage, precrossed starch, PVPP, and mixes evenly;
C3%PVP溶液制软才,18#制粒,干燥,控制水分3%;C3% PVP solution for softening, 18# granulation, drying, moisture control 3%;
D18目整粒,加入硬脂酸镁混合,压片即得。D18 mesh granules, mixed with magnesium stearate, compressed into tablets.
实施例3制得的复方糖浆剂The compound syrup that embodiment 3 makes
表3、本发明的复方糖浆剂。Table 3, compound syrup of the present invention.
制备方法:Preparation:
在10万级洁净区内In a 100,000-class clean area
A540g蔗糖溶于200mL煮沸的水中制成糖浆,滤过;A540g sucrose is dissolved in 200mL boiled water to make syrup, filter;
B称取处方量福尔可定,溶于适量的乙醇中;B takes by weighing the prescribed amount of pholcodine, and dissolves it in an appropriate amount of ethanol;
C称取处方量对乙酰氨基酚、盐酸伪麻黄碱、盐酸苯海拉明(夜用)溶于50mL纯化水中;C takes prescription quantity paracetamol, pseudoephedrine hydrochloride, diphenhydramine hydrochloride (night use) and is dissolved in 50mL purified water;
D将A步骤、B步骤和C步骤所得溶液混合,纯化水加至1000mL,再加入山梨酸钾、人工香精、枸橼酸,过滤,得糖浆,检查,分装即可。D Mix the solution obtained in step A, step B and step C, add purified water to 1000mL, then add potassium sorbate, artificial essence, citric acid, filter to obtain syrup, check and pack.
实施例4利用挤出滚圆法制得的复方胶囊剂Embodiment 4 utilizes the compound capsule that extrusion spheronization method makes
表4、利用挤出滚圆法制得的本发明的复方胶囊剂。Table 4. Compound capsules of the present invention prepared by extrusion spheronization.
制备方法:Preparation:
在10万级洁净区内In a 100,000-class clean area
A将福尔可定,对乙酰氨基酚,盐酸伪麻黄碱、盐酸苯海拉明过100目筛;A Pass pholcodine, acetaminophen, pseudoephedrine hydrochloride and diphenhydramine hydrochloride through a 100-mesh sieve;
B将处方量的福尔可定、盐酸伪麻黄碱、盐酸苯海拉明(夜用)、微晶纤维素、羧甲淀粉钠混合均匀,按等量递增法与对乙酰氨基酚混合均匀。B Mix the prescribed amount of pholcodine, pseudoephedrine hydrochloride, diphenhydramine hydrochloride (night use), microcrystalline cellulose, and sodium carboxymethyl starch, and mix them evenly with acetaminophen in equal increments.
C将处方量的十二烷基硫酸钠溶于5%PVPk30溶液中制湿软材。C Dissolve the prescribed amount of sodium lauryl sulfate in 5% PVPk30 solution to make wet soft material.
D通过0.5mm筛网的挤出机以45rpm的速度挤出,挤出后迅速于抛丸机内抛圆成丸,再在流化床内于60~70℃干燥,筛取16~30目之间的微丸,测定水分,装胶囊,包装即成。D is extruded at a speed of 45rpm through an extruder with a 0.5mm screen. After extrusion, it is quickly cast into pellets in a shot blasting machine, and then dried in a fluidized bed at 60-70°C, and sieved to 16-30 mesh The pellets in between, measure the water content, pack into capsules, and pack.
实施例5实施例4制得的复方胶囊剂的释放模式The release pattern of the compound capsule that embodiment 5 embodiment 4 makes
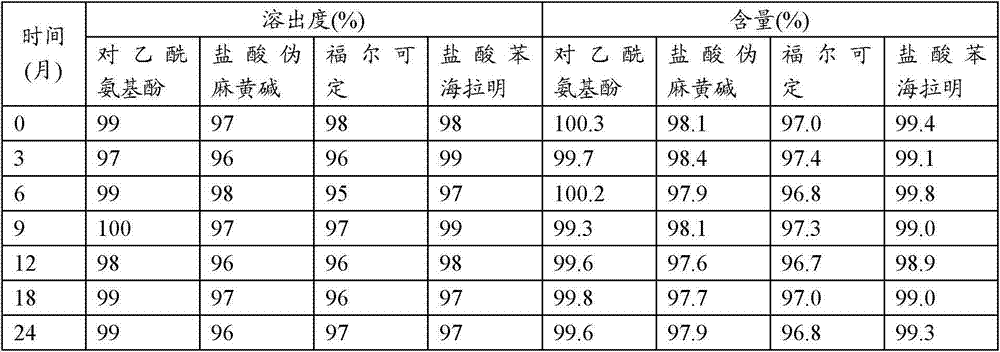
应用中国药典的药篮方法,于盐酸溶液为溶出介质,对乙酰氨基酚使用分光光度法,盐酸伪麻黄碱、福尔可定和盐酸苯海拉明使用高效液相法,历经45分钟测量活性化合物的释放。测试了实施例4中的处方1、处方2中的复方胶囊剂。图1、图2、图3、图4分别为测试曲线。Apply the drug basket method of the Chinese Pharmacopoeia, use hydrochloric acid solution as the dissolution medium, paracetamol use spectrophotometry, pseudoephedrine hydrochloride, pholcodine and diphenhydramine hydrochloride use high performance liquid phase method, after 45 minutes to measure the concentration of active compounds freed. Tested the compound capsule in prescription 1 in embodiment 4, prescription 2. Figure 1, Figure 2, Figure 3, and Figure 4 are the test curves respectively.
表5、为实施例4中的处方1所测示的历经45分钟不同组份的释放Table 5. The release of different components over 45 minutes as measured by prescription 1 in Example 4
表6、为实施例4中的处方2所测示的历经45分钟不同组份的释放Table 6. Release of different components over 45 minutes as measured for formulation 2 in Example 4
实施例6实施例4制得的复方胶囊剂的稳定性考察情况The stability investigation situation of the compound capsule that embodiment 6 embodiment 4 makes
应用中国药典的药篮方法,于盐酸溶液为溶出介质,对乙酰氨基酚使用分光光度法,盐酸伪麻黄碱、福尔可定和盐酸苯海拉明使用高效液相法,历经24个月测量活性化合物的释放和含量。测试了实施例4中的处方1、处方2中的复方胶囊剂。The drug basket method of the Chinese Pharmacopoeia was applied, with hydrochloric acid solution as the dissolution medium, paracetamol was used spectrophotometry, pseudoephedrine hydrochloride, pholcodine and diphenhydramine hydrochloride were used high performance liquid chromatography, and the active compounds were measured over 24 months release and content. Tested the compound capsule in prescription 1 in embodiment 4, prescription 2.
由下列表所示数值知道,不同含量的组份的释放维持相同且稳定。From the values shown in the table below, it is known that the release of components with different contents remains the same and stable.
表7、为实施例4中的处方1日用制剂所测示的历经24个月不同组份的释放和含量。Table 7. The release and content of different components measured over 24 months for the prescription 1 daily formulation in Example 4.
表8、为实施例4中的处方1夜用制剂所测示的历经24个月不同组份的释放和含量。Table 8. The release and content of different components measured over 24 months for the prescription 1-night formulation in Example 4.
表9、为实施例4中的处方2日用制剂所测示的历经24个月不同组份的释放和含量。Table 9. The release and content of different components measured over 24 months for the prescription 2-day formulation in Example 4.
表10、为实施例4中的处方1夜用制剂所测示的历经24个月不同组份的释放和含量。Table 10. The release and content of different components measured over 24 months for the prescription 1-night formulation in Example 4.
实施例7、本发明的日用,夜用制剂Embodiment 7, daily use of the present invention, night preparation
日用:Daily use:
福尔可定 1mgPholcodine 1mg
乙酰氨基酚 50mgAcetaminophen 50mg
盐酸伪麻黄碱 1mg。Pseudoephedrine hydrochloride 1mg.
夜用制剂,配方组成如下:Night use preparation, the formula composition is as follows:
实施例8、本发明的日用,夜用制剂Embodiment 8, daily use of the present invention, night preparation
日用:Daily use:
福尔可定 1mgPholcodine 1mg
乙酰氨基酚 50mgAcetaminophen 50mg
盐酸伪麻黄碱 1mg。Pseudoephedrine hydrochloride 1mg.
夜用制剂,配方组成如下:Night use preparation, the formula composition is as follows:
实施例9、本发明的日用,夜用制剂Embodiment 9, daily use of the present invention, night preparation
日用:Daily use:
福尔可定 1mgPholcodine 1mg
乙酰氨基酚 50mgAcetaminophen 50mg
盐酸伪麻黄碱 10mg。Pseudoephedrine hydrochloride 10mg.
夜用制剂,配方组成如下:Night use preparation, the formula composition is as follows:
实施例10、本发明的日用,夜用制剂
日用:Daily use:
福尔可定 30mgPholcodine 30mg
乙酰氨基酚 750mgAcetaminophen 750mg
盐酸伪麻黄碱 300mg。Pseudoephedrine hydrochloride 300mg.
夜用制剂,配方组成如下:Night use preparation, the formula composition is as follows:
实施例11、本发明的日用,夜用制剂Embodiment 11, daily use of the present invention, night preparation
日用:Daily use:
福尔可定 1mgPholcodine 1mg
乙酰氨基酚 50mgAcetaminophen 50mg
盐酸伪麻黄碱 5mg。Pseudoephedrine hydrochloride 5mg.
本发明优选的夜用制剂,配方组成如下:The preferred night-use preparation of the present invention has a formula as follows:
实施例10、本发明的日用,夜用制剂
日用:Daily use:
福尔可定 60mgPholcodine 60mg
乙酰氨基酚 4000mgAcetaminophen 4000mg
盐酸伪麻黄碱 400mg。Pseudoephedrine hydrochloride 400mg.
夜用制剂,配方组成如下:Night use preparation, the formula composition is as follows:
实施例11、本发明的日用,夜用制剂Embodiment 11, daily use of the present invention, night preparation
日用:Daily use:
福尔可定 60mgPholcodine 60mg
阿司匹林 250mg 325mgAspirin 250mg 325mg
麻黄碱 15mg 30
夜用制剂,配方组成如下:Night use preparation, the formula composition is as follows:
实施例12、本发明的日用,夜用制剂Embodiment 12, daily use of the present invention, night preparation
日用:Daily use:
福尔可定 60mgPholcodine 60mg
布洛芬 325mgIbuprofen 325mg
麻黄碱 30mgEphedrine 30mg
夜用制剂,配方组成如下:Night use preparation, the formula composition is as follows:
实施例13、本发明的日用,夜用制剂Embodiment 13, daily use of the present invention, night preparation
日用:Daily use:
福尔可定 60mgPholcodine 60mg
酮洛芬 250mgKetoprofen 250mg
麻黄碱 15mgEphedrine 15mg
夜用制剂,配方组成如下:Night use preparation, the formula composition is as follows:
实施例14、本发明的日用,夜用制剂Embodiment 14, daily use of the present invention, night use preparation
日用:Daily use:
福尔可定 60mgPholcodine 60mg
酮洛芬 325mgKetoprofen 325mg
麻黄碱 30mgEphedrine 30mg
夜用制剂,配方组成如下:Night use preparation, the formula composition is as follows:
实施例15、本发明的日用,夜用制剂Embodiment 15, daily use of the present invention, night preparation
日用:Daily use:
福尔可定 60mgPholcodine 60mg
酮洛芬 325mgKetoprofen 325mg
麻黄碱 30mgEphedrine 30mg
夜用制剂,配方组成如下:Night use preparation, the formula composition is as follows:
实施例16、本发明的日用,夜用制剂Embodiment 16, daily use of the present invention, night use preparation
日用:Daily use:
福尔可定 60mgPholcodine 60mg
酮洛芬 325mgKetoprofen 325mg
麻黄碱 30mgEphedrine 30mg
夜用制剂,配方组成如下:Night use preparation, the formula composition is as follows:
Claims (2)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201110331486.3A CN102380101B (en) | 2009-11-13 | 2009-11-13 | A compound medicinal preparation containing pholcodine for treating common cold |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201110331486.3A CN102380101B (en) | 2009-11-13 | 2009-11-13 | A compound medicinal preparation containing pholcodine for treating common cold |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN200910222919A Division CN101716344A (en) | 2009-11-13 | 2009-11-13 | Compound pharmaceutical preparation containing pholcodine for treating cold |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN102380101A CN102380101A (en) | 2012-03-21 |
| CN102380101B true CN102380101B (en) | 2014-03-05 |
Family
ID=45820196
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201110331486.3A Active CN102380101B (en) | 2009-11-13 | 2009-11-13 | A compound medicinal preparation containing pholcodine for treating common cold |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN102380101B (en) |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101065112A (en) * | 2004-06-02 | 2007-10-31 | 惠氏公司 | Multi-layer tablet comprising non-steroidal anti-inflammatory drugs, decongestants and non-sedating antihist amines |
-
2009
- 2009-11-13 CN CN201110331486.3A patent/CN102380101B/en active Active
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101065112A (en) * | 2004-06-02 | 2007-10-31 | 惠氏公司 | Multi-layer tablet comprising non-steroidal anti-inflammatory drugs, decongestants and non-sedating antihist amines |
Also Published As
| Publication number | Publication date |
|---|---|
| CN102380101A (en) | 2012-03-21 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| ES2237121T3 (en) | FAST DISGREGABLE SOLID PREPARATION. | |
| KR101752014B1 (en) | Orally disintegrating tablet compositions comprising combinations of high and low-dose drugs | |
| JPS63196511A (en) | Swellable pellets | |
| CN107028900A (en) | Rapid dispersion particle, oral disnitegration tablet and method | |
| JP2005527508A (en) | Rapid melting multiparticulate formulation for oral delivery | |
| CN104244930A (en) | Orally disintegrating tablet and its production method | |
| JP5956475B2 (en) | Orally disintegrating tablets containing bitter mask granules | |
| EP2385823A1 (en) | Dual release pharmaceutical suspension | |
| US6254891B1 (en) | Extended release acetaminophen | |
| WO2011043370A1 (en) | Coating film, and granules and tablets each utilizing same | |
| CN101716344A (en) | Compound pharmaceutical preparation containing pholcodine for treating cold | |
| JP6314206B2 (en) | Resin acid phenylephrine particles | |
| EP2642980A2 (en) | Pediatric formulation | |
| CN106822907B (en) | Two-phase release preparation containing racecadotril and preparation method thereof | |
| EP3431107A1 (en) | Pharmaceutical composition particles and orally disintegrating preparation including same | |
| CN109157527B (en) | Irbesartan capsule and preparation method thereof | |
| CN102380101B (en) | A compound medicinal preparation containing pholcodine for treating common cold | |
| CN100404025C (en) | Orally disintegrating tablet containing tramadol hydrochloride and acetaminophen and preparation method thereof | |
| JP4995099B2 (en) | Non-plastic medicine taste-masking system | |
| CN105902564B (en) | A kind of pharmaceutical composition and preparation method for treating hypertension | |
| JP7330948B2 (en) | Compositions containing suplatast tosylate | |
| JP2012072133A (en) | Small-sized sustained release formulation of ambroxol hydrochloride | |
| CN101766608B (en) | Compound pseudoephedrine hydrochloride sustained-release preparation and preparation method thereof | |
| CN102232934B (en) | Pulsatile pellet, pulsatile orally disintegrating tablet containing same, and preparation methods and applications thereof | |
| JP2020063202A (en) | Pharmaceutical composition containing caffeine and hyoscyamine and method for producing the same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |