CN102276799B - Quinoxaline unit-containing fluorene copolymer, and preparation method and application thereof - Google Patents
Quinoxaline unit-containing fluorene copolymer, and preparation method and application thereof Download PDFInfo
- Publication number
- CN102276799B CN102276799B CN201010196323.4A CN201010196323A CN102276799B CN 102276799 B CN102276799 B CN 102276799B CN 201010196323 A CN201010196323 A CN 201010196323A CN 102276799 B CN102276799 B CN 102276799B
- Authority
- CN
- China
- Prior art keywords
- preparation
- alkyl
- fluorene copolymer
- compd
- organic
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
- 238000002360 preparation method Methods 0.000 title claims abstract description 92
- NIHNNTQXNPWCJQ-UHFFFAOYSA-N o-biphenylenemethane Natural products C1=CC=C2CC3=CC=CC=C3C2=C1 NIHNNTQXNPWCJQ-UHFFFAOYSA-N 0.000 title claims abstract description 63
- 229920001577 copolymer Polymers 0.000 title claims abstract description 60
- 125000001567 quinoxalinyl group Chemical group N1=C(C=NC2=CC=CC=C12)* 0.000 title claims abstract description 59
- 238000006243 chemical reaction Methods 0.000 claims abstract description 74
- 125000003545 alkoxy group Chemical group 0.000 claims abstract description 9
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims abstract description 7
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 claims description 58
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 claims description 43
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 claims description 30
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 claims description 29
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 claims description 27
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 claims description 25
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 claims description 22
- 239000001301 oxygen Substances 0.000 claims description 22
- 229910052760 oxygen Inorganic materials 0.000 claims description 22
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 claims description 21
- AIGRXSNSLVJMEA-FQEVSTJZSA-N ethoxy-(4-nitrophenoxy)-phenyl-sulfanylidene-$l^{5}-phosphane Chemical compound O([P@@](=S)(OCC)C=1C=CC=CC=1)C1=CC=C([N+]([O-])=O)C=C1 AIGRXSNSLVJMEA-FQEVSTJZSA-N 0.000 claims description 21
- 239000000463 material Substances 0.000 claims description 21
- PCLIMKBDDGJMGD-UHFFFAOYSA-N N-bromosuccinimide Chemical compound BrN1C(=O)CCC1=O PCLIMKBDDGJMGD-UHFFFAOYSA-N 0.000 claims description 20
- 239000003960 organic solvent Substances 0.000 claims description 18
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 claims description 15
- -1 basic metal carbonate Chemical class 0.000 claims description 13
- 229960001701 chloroform Drugs 0.000 claims description 12
- 150000002220 fluorenes Chemical class 0.000 claims description 11
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 claims description 10
- 125000000217 alkyl group Chemical group 0.000 claims description 10
- 229920000642 polymer Polymers 0.000 claims description 10
- LVTJOONKWUXEFR-FZRMHRINSA-N protoneodioscin Natural products O(C[C@@H](CC[C@]1(O)[C@H](C)[C@@H]2[C@]3(C)[C@H]([C@H]4[C@@H]([C@]5(C)C(=CC4)C[C@@H](O[C@@H]4[C@H](O[C@H]6[C@@H](O)[C@@H](O)[C@@H](O)[C@H](C)O6)[C@@H](O)[C@H](O[C@H]6[C@@H](O)[C@@H](O)[C@@H](O)[C@H](C)O6)[C@H](CO)O4)CC5)CC3)C[C@@H]2O1)C)[C@H]1[C@H](O)[C@H](O)[C@H](O)[C@@H](CO)O1 LVTJOONKWUXEFR-FZRMHRINSA-N 0.000 claims description 10
- 229910052799 carbon Inorganic materials 0.000 claims description 9
- MZRVEZGGRBJDDB-UHFFFAOYSA-N N-Butyllithium Chemical compound [Li]CCCC MZRVEZGGRBJDDB-UHFFFAOYSA-N 0.000 claims description 8
- 229910052744 lithium Inorganic materials 0.000 claims description 8
- 229910052763 palladium Inorganic materials 0.000 claims description 8
- 239000003054 catalyst Substances 0.000 claims description 7
- 238000005401 electroluminescence Methods 0.000 claims description 7
- 229940073455 tetraethylammonium hydroxide Drugs 0.000 claims description 7
- LRGJRHZIDJQFCL-UHFFFAOYSA-M tetraethylazanium;hydroxide Chemical compound [OH-].CC[N+](CC)(CC)CC LRGJRHZIDJQFCL-UHFFFAOYSA-M 0.000 claims description 7
- 239000003513 alkali Substances 0.000 claims description 6
- 150000001875 compounds Chemical class 0.000 claims description 6
- OMFXVFTZEKFJBZ-HJTSIMOOSA-N corticosterone Chemical compound O=C1CC[C@]2(C)[C@H]3[C@@H](O)C[C@](C)([C@H](CC4)C(=O)CO)[C@@H]4[C@@H]3CCC2=C1 OMFXVFTZEKFJBZ-HJTSIMOOSA-N 0.000 claims description 6
- 230000005669 field effect Effects 0.000 claims description 6
- 239000003446 ligand Substances 0.000 claims description 6
- 239000000203 mixture Substances 0.000 claims description 6
- 125000000951 phenoxy group Chemical group [H]C1=C([H])C([H])=C(O*)C([H])=C1[H] 0.000 claims description 6
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical group CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 claims description 6
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 claims description 5
- VPMJBJSLTPBZLR-UHFFFAOYSA-N 3,6-dibromobenzene-1,2-diamine Chemical class NC1=C(N)C(Br)=CC=C1Br VPMJBJSLTPBZLR-UHFFFAOYSA-N 0.000 claims description 4
- 239000000376 reactant Substances 0.000 claims description 4
- UBJFKNSINUCEAL-UHFFFAOYSA-N lithium;2-methylpropane Chemical compound [Li+].C[C-](C)C UBJFKNSINUCEAL-UHFFFAOYSA-N 0.000 claims description 3
- 238000006386 neutralization reaction Methods 0.000 claims description 3
- 230000003287 optical effect Effects 0.000 claims description 3
- NHKJPPKXDNZFBJ-UHFFFAOYSA-N phenyllithium Chemical compound [Li]C1=CC=CC=C1 NHKJPPKXDNZFBJ-UHFFFAOYSA-N 0.000 claims description 3
- 238000003860 storage Methods 0.000 claims description 3
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 claims description 2
- FERIUCNNQQJTOY-UHFFFAOYSA-N Butyric acid Natural products CCCC(O)=O FERIUCNNQQJTOY-UHFFFAOYSA-N 0.000 claims description 2
- 150000008044 alkali metal hydroxides Chemical class 0.000 claims description 2
- 238000005893 bromination reaction Methods 0.000 claims description 2
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 claims description 2
- 229910052794 bromium Inorganic materials 0.000 claims description 2
- IHLVCKWPAMTVTG-UHFFFAOYSA-N lithium;carbanide Chemical compound [Li+].[CH3-] IHLVCKWPAMTVTG-UHFFFAOYSA-N 0.000 claims description 2
- 229910052728 basic metal Inorganic materials 0.000 claims 1
- 230000031709 bromination Effects 0.000 claims 1
- 150000008301 phosphite esters Chemical class 0.000 claims 1
- YTPLMLYBLZKORZ-UHFFFAOYSA-N Thiophene Chemical group C=1C=CSC=1 YTPLMLYBLZKORZ-UHFFFAOYSA-N 0.000 abstract description 45
- 238000004519 manufacturing process Methods 0.000 abstract description 5
- 238000002835 absorbance Methods 0.000 abstract 1
- KHUXNRRPPZOJPT-UHFFFAOYSA-N phenoxy radical Chemical group O=C1C=C[CH]C=C1 KHUXNRRPPZOJPT-UHFFFAOYSA-N 0.000 abstract 1
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 39
- 239000000047 product Substances 0.000 description 31
- 239000000243 solution Substances 0.000 description 29
- 238000000034 method Methods 0.000 description 28
- 230000008569 process Effects 0.000 description 22
- 229930192474 thiophene Natural products 0.000 description 20
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 18
- 239000002585 base Substances 0.000 description 18
- 229910052757 nitrogen Inorganic materials 0.000 description 17
- 238000003756 stirring Methods 0.000 description 17
- 238000000926 separation method Methods 0.000 description 16
- 238000004440 column chromatography Methods 0.000 description 15
- 238000001035 drying Methods 0.000 description 15
- 238000001840 matrix-assisted laser desorption--ionisation time-of-flight mass spectrometry Methods 0.000 description 15
- 230000004044 response Effects 0.000 description 15
- 238000010025 steaming Methods 0.000 description 15
- 239000012265 solid product Substances 0.000 description 14
- 238000000967 suction filtration Methods 0.000 description 13
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 12
- QARVLSVVCXYDNA-UHFFFAOYSA-N bromobenzene Chemical compound BrC1=CC=CC=C1 QARVLSVVCXYDNA-UHFFFAOYSA-N 0.000 description 12
- XSCHRSMBECNVNS-UHFFFAOYSA-N quinoxaline Chemical compound N1=CC=NC2=CC=CC=C21 XSCHRSMBECNVNS-UHFFFAOYSA-N 0.000 description 12
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 12
- 239000011248 coating agent Substances 0.000 description 10
- 238000000576 coating method Methods 0.000 description 10
- 239000007789 gas Substances 0.000 description 10
- 238000000605 extraction Methods 0.000 description 9
- 238000006069 Suzuki reaction reaction Methods 0.000 description 7
- 239000000126 substance Substances 0.000 description 7
- WRPMSBHZWIWJTC-UHFFFAOYSA-N C(CCCCCCCCCCCCCCCCCCC)C1=NC2=CC=CC=C2C=C1 Chemical compound C(CCCCCCCCCCCCCCCCCCC)C1=NC2=CC=CC=C2C=C1 WRPMSBHZWIWJTC-UHFFFAOYSA-N 0.000 description 6
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 6
- 239000007864 aqueous solution Substances 0.000 description 6
- KGBXLFKZBHKPEV-UHFFFAOYSA-N boric acid Chemical compound OB(O)O KGBXLFKZBHKPEV-UHFFFAOYSA-N 0.000 description 6
- 239000004327 boric acid Substances 0.000 description 6
- PIBWKRNGBLPSSY-UHFFFAOYSA-L palladium(II) chloride Chemical compound Cl[Pd]Cl PIBWKRNGBLPSSY-UHFFFAOYSA-L 0.000 description 6
- 238000004062 sedimentation Methods 0.000 description 6
- 239000004065 semiconductor Substances 0.000 description 6
- 230000003595 spectral effect Effects 0.000 description 6
- 238000004528 spin coating Methods 0.000 description 6
- 238000001291 vacuum drying Methods 0.000 description 6
- 229910001873 dinitrogen Inorganic materials 0.000 description 5
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 description 4
- 229940126062 Compound A Drugs 0.000 description 4
- 241000790917 Dioxys <bee> Species 0.000 description 4
- NLDMNSXOCDLTTB-UHFFFAOYSA-N Heterophylliin A Natural products O1C2COC(=O)C3=CC(O)=C(O)C(O)=C3C3=C(O)C(O)=C(O)C=C3C(=O)OC2C(OC(=O)C=2C=C(O)C(O)=C(O)C=2)C(O)C1OC(=O)C1=CC(O)=C(O)C(O)=C1 NLDMNSXOCDLTTB-UHFFFAOYSA-N 0.000 description 4
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical class [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 4
- SOHOYYHPFIUFDH-UHFFFAOYSA-M [Br-].[Zn+]C Chemical compound [Br-].[Zn+]C SOHOYYHPFIUFDH-UHFFFAOYSA-M 0.000 description 4
- 229910052782 aluminium Inorganic materials 0.000 description 4
- 239000000872 buffer Substances 0.000 description 4
- 230000005611 electricity Effects 0.000 description 4
- MRNHPUHPBOKKQT-UHFFFAOYSA-N indium;tin;hydrate Chemical compound O.[In].[Sn] MRNHPUHPBOKKQT-UHFFFAOYSA-N 0.000 description 4
- 238000009413 insulation Methods 0.000 description 4
- 229910052751 metal Inorganic materials 0.000 description 4
- 239000002184 metal Substances 0.000 description 4
- 0 *Cc1c[s]c2c1[s]cc2* Chemical compound *Cc1c[s]c2c1[s]cc2* 0.000 description 3
- KZPYGQFFRCFCPP-UHFFFAOYSA-N 1,1'-bis(diphenylphosphino)ferrocene Chemical compound [Fe+2].C1=CC=C[C-]1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=C[C-]1P(C=1C=CC=CC=1)C1=CC=CC=C1 KZPYGQFFRCFCPP-UHFFFAOYSA-N 0.000 description 3
- KBVDUUXRXJTAJC-UHFFFAOYSA-N 2,5-dibromothiophene Chemical compound BrC1=CC=C(Br)S1 KBVDUUXRXJTAJC-UHFFFAOYSA-N 0.000 description 3
- BZLVMXJERCGZMT-UHFFFAOYSA-N Methyl tert-butyl ether Chemical compound COC(C)(C)C BZLVMXJERCGZMT-UHFFFAOYSA-N 0.000 description 3
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 3
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 3
- SMWDFEZZVXVKRB-UHFFFAOYSA-N anhydrous quinoline Natural products N1=CC=CC2=CC=CC=C21 SMWDFEZZVXVKRB-UHFFFAOYSA-N 0.000 description 3
- 230000009286 beneficial effect Effects 0.000 description 3
- 230000005540 biological transmission Effects 0.000 description 3
- 238000001816 cooling Methods 0.000 description 3
- 238000005516 engineering process Methods 0.000 description 3
- 150000002500 ions Chemical class 0.000 description 3
- 238000012545 processing Methods 0.000 description 3
- 238000010926 purge Methods 0.000 description 3
- 238000001953 recrystallisation Methods 0.000 description 3
- 150000003839 salts Chemical class 0.000 description 3
- 238000007789 sealing Methods 0.000 description 3
- 229910052710 silicon Inorganic materials 0.000 description 3
- 239000010703 silicon Substances 0.000 description 3
- 238000001228 spectrum Methods 0.000 description 3
- 239000000758 substrate Substances 0.000 description 3
- 229920002554 vinyl polymer Polymers 0.000 description 3
- 238000005406 washing Methods 0.000 description 3
- MCEWYIDBDVPMES-UHFFFAOYSA-N [60]pcbm Chemical compound C123C(C4=C5C6=C7C8=C9C%10=C%11C%12=C%13C%14=C%15C%16=C%17C%18=C(C=%19C=%20C%18=C%18C%16=C%13C%13=C%11C9=C9C7=C(C=%20C9=C%13%18)C(C7=%19)=C96)C6=C%11C%17=C%15C%13=C%15C%14=C%12C%12=C%10C%10=C85)=C9C7=C6C2=C%11C%13=C2C%15=C%12C%10=C4C23C1(CCCC(=O)OC)C1=CC=CC=C1 MCEWYIDBDVPMES-UHFFFAOYSA-N 0.000 description 2
- 238000000862 absorption spectrum Methods 0.000 description 2
- 229910052786 argon Inorganic materials 0.000 description 2
- 230000008901 benefit Effects 0.000 description 2
- 125000002091 cationic group Chemical group 0.000 description 2
- 238000011161 development Methods 0.000 description 2
- 238000010790 dilution Methods 0.000 description 2
- 239000012895 dilution Substances 0.000 description 2
- 230000007613 environmental effect Effects 0.000 description 2
- 239000011521 glass Substances 0.000 description 2
- 239000012046 mixed solvent Substances 0.000 description 2
- 239000000178 monomer Substances 0.000 description 2
- 239000003921 oil Substances 0.000 description 2
- 230000009467 reduction Effects 0.000 description 2
- 230000001105 regulatory effect Effects 0.000 description 2
- 238000011160 research Methods 0.000 description 2
- 239000011734 sodium Substances 0.000 description 2
- 238000012546 transfer Methods 0.000 description 2
- 238000007738 vacuum evaporation Methods 0.000 description 2
- VNDYJBBGRKZCSX-UHFFFAOYSA-L zinc bromide Chemical compound Br[Zn]Br VNDYJBBGRKZCSX-UHFFFAOYSA-L 0.000 description 2
- QFZLOYZIBDWMLB-UHFFFAOYSA-N 3,6-dioctylthieno[3,2-b]thiophene Chemical compound CCCCCCCCC1=CSC2=C1SC=C2CCCCCCCC QFZLOYZIBDWMLB-UHFFFAOYSA-N 0.000 description 1
- LCSHATKNSDVDRY-UHFFFAOYSA-N 5,8-dibromoquinoline Chemical compound C1=CC=C2C(Br)=CC=C(Br)C2=N1 LCSHATKNSDVDRY-UHFFFAOYSA-N 0.000 description 1
- WJGYYOOPOPFPBB-UHFFFAOYSA-N BrC1=C(C(=NC2=CC=CC=C12)CCCCCCCC)CCCCCCCC Chemical compound BrC1=C(C(=NC2=CC=CC=C12)CCCCCCCC)CCCCCCCC WJGYYOOPOPFPBB-UHFFFAOYSA-N 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-L Carbonate Chemical compound [O-]C([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-L 0.000 description 1
- ZNZYKNKBJPZETN-WELNAUFTSA-N Dialdehyde 11678 Chemical compound N1C2=CC=CC=C2C2=C1[C@H](C[C@H](/C(=C/O)C(=O)OC)[C@@H](C=C)C=O)NCC2 ZNZYKNKBJPZETN-WELNAUFTSA-N 0.000 description 1
- 229920000144 PEDOT:PSS Polymers 0.000 description 1
- 239000004698 Polyethylene Substances 0.000 description 1
- 229910004298 SiO 2 Inorganic materials 0.000 description 1
- JEQMKASXJFBIQB-UHFFFAOYSA-M [Br-].CCCCCCCCCCCCCCCCCCCC[Zn+] Chemical compound [Br-].CCCCCCCCCCCCCCCCCCCC[Zn+] JEQMKASXJFBIQB-UHFFFAOYSA-M 0.000 description 1
- 239000004411 aluminium Substances 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- 238000013459 approach Methods 0.000 description 1
- 125000001204 arachidyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 230000008033 biological extinction Effects 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 229920000547 conjugated polymer Polymers 0.000 description 1
- 230000009849 deactivation Effects 0.000 description 1
- 230000007423 decrease Effects 0.000 description 1
- 230000007812 deficiency Effects 0.000 description 1
- 238000000151 deposition Methods 0.000 description 1
- 230000008021 deposition Effects 0.000 description 1
- 238000010586 diagram Methods 0.000 description 1
- 238000001704 evaporation Methods 0.000 description 1
- 230000008020 evaporation Effects 0.000 description 1
- 230000004907 flux Effects 0.000 description 1
- PCHJSUWPFVWCPO-UHFFFAOYSA-N gold Chemical compound [Au] PCHJSUWPFVWCPO-UHFFFAOYSA-N 0.000 description 1
- 229910052737 gold Inorganic materials 0.000 description 1
- 239000010931 gold Substances 0.000 description 1
- 238000005286 illumination Methods 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 238000011068 loading method Methods 0.000 description 1
- 239000011159 matrix material Substances 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 125000002347 octyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 230000005693 optoelectronics Effects 0.000 description 1
- 150000002894 organic compounds Chemical class 0.000 description 1
- 239000011368 organic material Substances 0.000 description 1
- 238000013086 organic photovoltaic Methods 0.000 description 1
- 229920001467 poly(styrenesulfonates) Polymers 0.000 description 1
- 229920000573 polyethylene Polymers 0.000 description 1
- 229940005642 polystyrene sulfonic acid Drugs 0.000 description 1
- 238000007639 printing Methods 0.000 description 1
- 230000005855 radiation Effects 0.000 description 1
- 230000035484 reaction time Effects 0.000 description 1
- 230000027756 respiratory electron transport chain Effects 0.000 description 1
- 230000035939 shock Effects 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- NMFKEMBATXKZSP-UHFFFAOYSA-N thieno[3,2-b]thiophene Chemical compound S1C=CC2=C1C=CS2.S1C=CC2=C1C=CS2 NMFKEMBATXKZSP-UHFFFAOYSA-N 0.000 description 1
- 230000009466 transformation Effects 0.000 description 1
- 230000007704 transition Effects 0.000 description 1
- 229910052721 tungsten Inorganic materials 0.000 description 1
- 238000004506 ultrasonic cleaning Methods 0.000 description 1
- 229940102001 zinc bromide Drugs 0.000 description 1
Images
Landscapes
- Polyoxymethylene Polymers And Polymers With Carbon-To-Carbon Bonds (AREA)
Abstract
The invention discloses a quinoxaline unit-containing fluorene copolymer with a molecular structural general formula shown as (I), and a preparation method and application thereof. In the formula, x plus y is equal to 1, x is not equal to 0 and y is not equal to 0; n is any integer of 1 to 200; R1 and R2 are H or C1 to C20 alkyl; R3 and R4 are C1 to C20 alkyl, alkoxy, phenyl or phenoxyl; and Ar1 is a thiophene unit-containing group. The quinoxaline unit-containing fluorene copolymer has high stability and solubility, high absorbance and wide light-absorbing range and improves photoelectric conversion efficiency. The preparation method is simple, high in yield, easy in operation and control and suitable for industrialized production.
Description
Technical field
The invention belongs to organic compound synthesis technical field, relate to specifically a kind of containing quinoxaline unit fluorene copolymer and its preparation method and application.
Background technology
Utilize cheap material to prepare low cost, dynamical solar cell is study hotspot and the difficult point in photovoltaic field always.The current silicon solar cell for ground, because complex manufacturing, cost are high, is restricted its application.In order to reduce costs, expand range of application, people are finding novel solar cell material always for a long time.Polymer solar battery receives much concern because the mode big area such as cheap, the quality of cost of material is light, flexibility, simple, the available coating of production technique, printing such as prepare at the advantage, if its energy conversion efficiency can be brought up to the level that approaches commodity silicon solar cell, its market outlook will be very huge.On SCIENCE, report conjugated polymers and C from N.S.Sariciftci in 1992 etc.
60between Photoinduced Electron transfer phenomena after, people have dropped into large quantity research aspect polymer solar battery, and have obtained development at full speed.At present, the energy conversion efficiency of polymer solar battery has reached 6.5%, but still much lower than the efficiency of conversion of inorganic solar cell, the main restricting factor that limiting performance improves has: the carrier mobility that organic semiconductor device is relatively low, the spectral response of device is not mated with solar radiation spectrum, the red light district of high photon flux be not used effectively and the electrode collection effciency of current carrier low etc.For polymer solar battery is on the actual application, the material of development of new, increases substantially its effciency of energy transfer and is still the top priority of this research field.
Summary of the invention
The object of the invention is to overcome the above-mentioned deficiency of prior art, provide a kind of containing quinoxaline unit fluorene copolymer, this multipolymer stability and solvability are good, absorbancy strong, extinction wide ranges, have improved its electricity conversion.
Another object of the present invention is to provide a kind of preparation method containing quinoxaline unit fluorene copolymer.
Further aim of the present invention be to provide a kind of containing quinoxaline unit fluorene copolymer at organic photoelectrical material, polymer solar battery, organic electroluminescence device, organic field effect tube, organic light storage device, organic non-linear optical properties or/and the application in organic laser apparatus.
In order to realize foregoing invention object, technical scheme of the present invention is as follows:
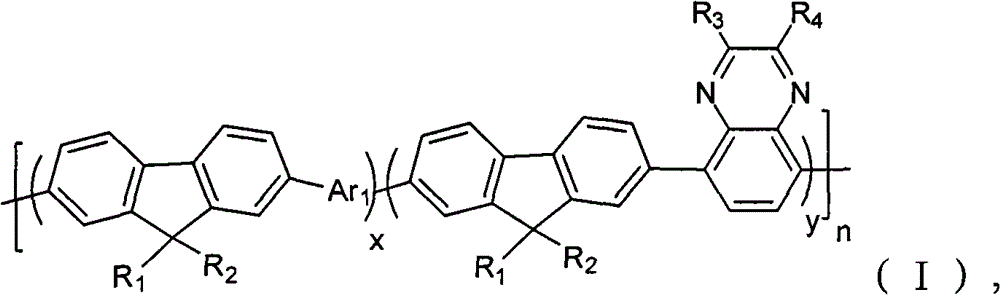
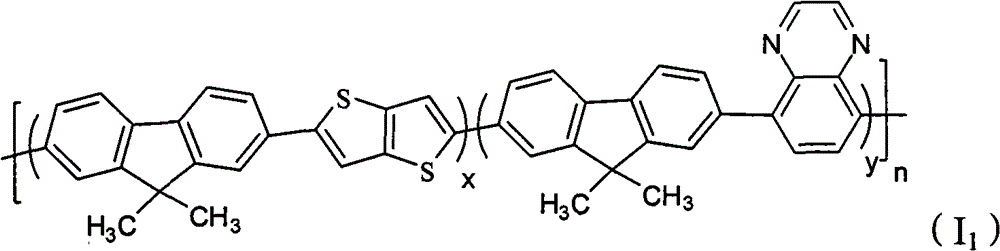
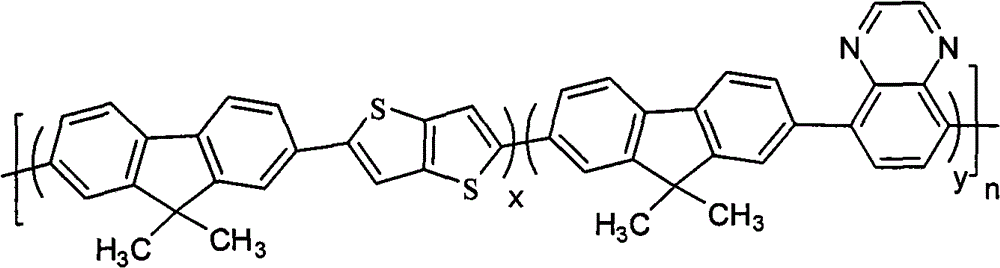
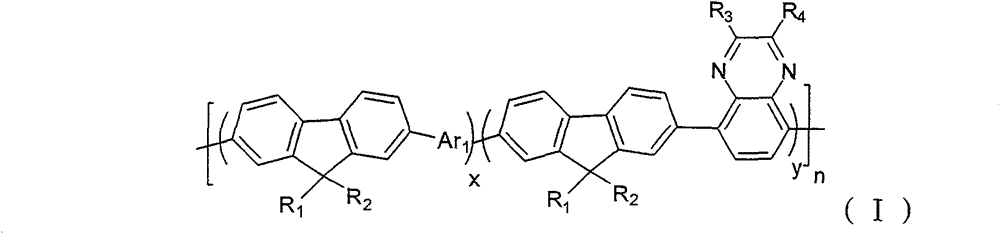
A kind of containing quinoxaline unit fluorene copolymer, its general formula of molecular structure is following (I):
In formula, x+y=1, and x ≠ 0, y ≠ 0; Arbitrary integer of n=1~200; R
1, R
2be selected from H or C
1~C
20alkyl; R
3, R
4be selected from C
1~C
20alkyl, alkoxyl group, phenyl or phenoxy group; Ar
1for containing thiophene unit group.
And, a kind of containing quinoxaline unit fluorene copolymer preparation method, comprise the steps:
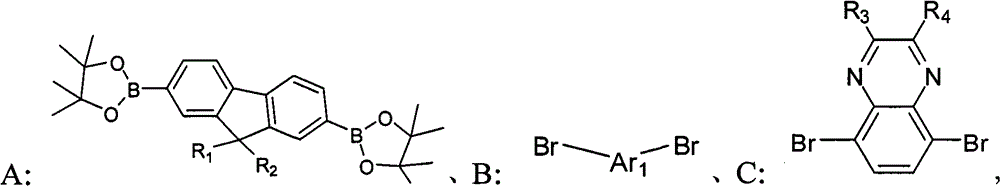
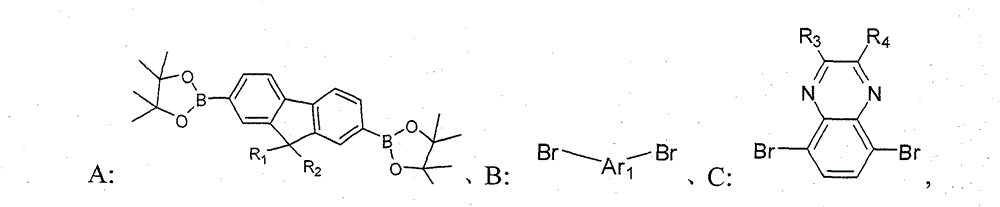
The compd A, B, the C that provide respectively following structural formula to represent,
Wherein, R
1, R
2be selected from H or C
1~C
20alkyl; R
3, R
4be selected from C
1~C
20alkyl, alkoxyl group, phenyl or phenoxy group; Ar
1for containing thiophene unit group;
Under the condition existing in anaerobic, alkaline environment catalyst neutralisation, organic solvent, compd A, B, C are carried out to Suzuki linked reaction, obtain following general structure for (I) represent containing quinoxaline unit fluorene copolymer,
In formula, x+y=1, and x ≠ 0, y ≠ 0; Arbitrary integer of n=1~200.
The present invention compared with prior art at least possesses following advantage:
1. fluorenes class monomeric compound A and thiophene-based monomeric compound B are two kinds of very excellent donor materials, and quinoxaline monomeric compound C is a kind of very excellent acceptor material, the multipolymer being made up of these three kinds of monomers can form a kind of very strong donor-receiver structure, simultaneously, increase the density of polymer backbone electronic cloud by introduce assorted (thick) toroidal molecule in copolymer chain, thereby improve on the one hand the stability of this multipolymer, reduce on the other hand this multipolymer band gap, expand light abstraction width, improve electricity conversion, this multipolymer absorption spectrum can in very large range be regulated, there is wider spectral response range, better to mate sun emmission spectrum,
2. the syntheti c route of synthetic fluorenes class monomeric compound A, thiophene-based monomeric compound B and quinoxaline monomeric compound C is fairly simple, easily improve solvability and the molecular weight of product by introducing alkyl or alkoxyl group, with realize can spin coating polymkeric substance or oligomer that can spin coating, be beneficial to film forming processing;
3. preparation method is simple, and productive rate is high, and reaction conditions gentleness, is easy to operate and control, and is suitable for suitability for industrialized production.
Accompanying drawing explanation
Fig. 1 is the schematic diagram containing quinoxaline unit fluorene copolymer general formula of molecular structure of the embodiment of the present invention;
Fig. 2 is the structural representation as the polymer solar cell device of active coating containing quinoxaline unit fluorene copolymer take the embodiment of the present invention;
Fig. 3 is the structural representation with the organic electroluminescence device containing quinoxaline unit fluorene copolymer of the embodiment of the present invention;
Fig. 4 is the structural representation as the organic field effect tube device of organic semiconductor layer containing quinoxaline unit fluorene copolymer take the embodiment of the present invention.
Embodiment
In order to make the technical problem to be solved in the present invention, technical scheme and beneficial effect clearer, below in conjunction with embodiment, the present invention is further elaborated.Should be appreciated that specific embodiment described herein, only in order to explain the present invention, is not intended to limit the present invention.
Refer to Fig. 1, show that the one of the embodiment of the present invention is containing quinoxaline unit fluorene copolymer general formula of molecular structure, it is following (I):
In formula, x+y=1, and x ≠ 0, y ≠ 0; Arbitrary integer of n=1~200; R
1, R
2be selected from H or C
1~C
20alkyl; R
3, R
4be selected from C
1~C
20alkyl, alkoxyl group, phenyl or phenoxy group; Ar
1for containing thiophene unit group.
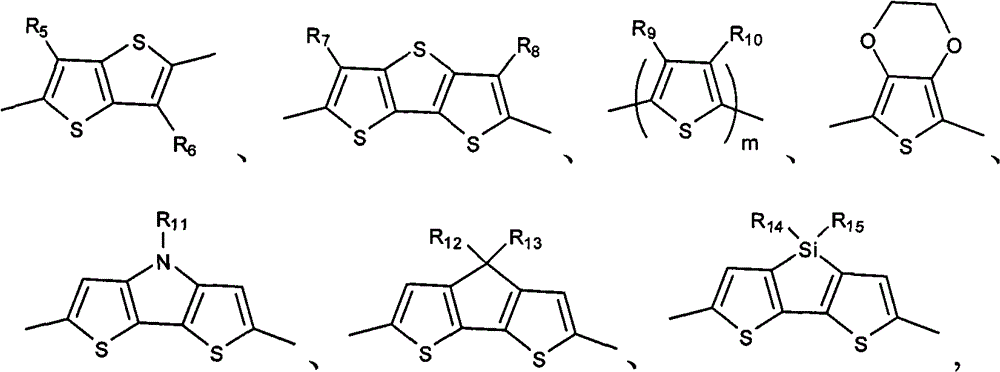
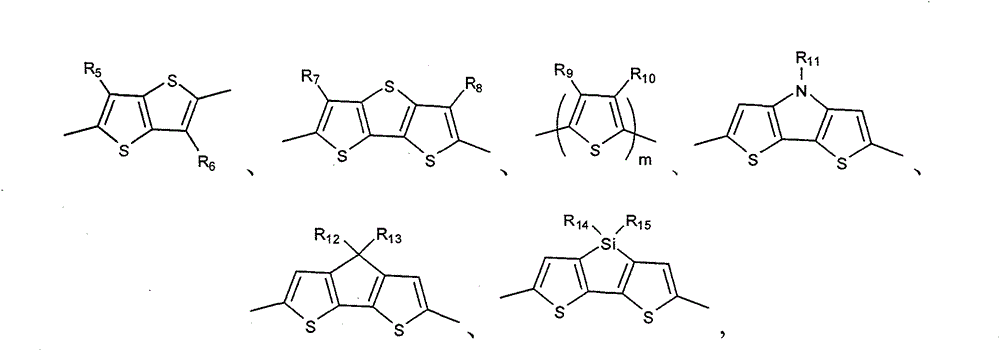
Above-mentioned Ar
1be preferably following one or more groups:
In formula, R
5, R
6, R
7, R
8, R
9, R
10be selected from H, C
1~C
20alkyl; R
11, R
12, R
13, R
14, R
15be selected from C
1~C
20alkyl; M is number of repeat unit, arbitrary integer of m=1~20.
What above-described embodiment provided contains the groups such as poly-fluorenes, thiophene, quinoxaline containing in quinoxaline unit fluorene copolymer molecule.Wherein, light stability and the excellent heat stability of poly-fluorenes and derivative thereof; Thiophene is five-membered ring structure, meets shock youngster rule, has moderate band gap, wider spectral response, thermostability and environmental stability preferably, the spectral response range that contributes to widen polymkeric substance; Because quinoxaline has good two dimensional structure, it is a body unit that is subject to that contains a five-ring and six-ring skeleton, and there is intramolecular charge and shift character, excellent electrochemical reduction character, has stronger electron-withdrawing power, therefore, it is often as be incorporated in photoelectric material to regulate bandwidth and reduction potential by body unit, quinoxaline unit also has stronger modifiability, can utilize easy method to introduce electron-donating group and electron-accepting group, regulates its electrophilic performance.
Fluorenes class monomeric compound A and thiophene-based monomeric compound B are two kinds of very excellent donor materials, and quinoxaline monomeric compound C is a kind of very excellent acceptor material, the multipolymer being made up of these three kinds of monomers can form a kind of very strong donor-receiver structure, simultaneously, increase the density of polymer backbone electronic cloud by introduce assorted (thick) toroidal molecule in copolymer chain, thereby improve on the one hand the stability of this multipolymer, reduce on the other hand this multipolymer band gap, expand light abstraction width, improve electricity conversion, this multipolymer absorption spectrum can in very large range be regulated, there is wider spectral response range, better to mate sun emmission spectrum, in the fields such as organic photovoltaic cell, have a good application prospect.
The syntheti c route of fluorenes class monomeric compound A, thiophene-based monomeric compound B and quinoxaline monomeric compound C is fairly simple, easily improve solvability and the molecular weight of product by introducing alkyl or alkoxyl group, with realize can spin coating polymkeric substance or oligomer that can spin coating, be beneficial to film forming processing.
And the embodiment of the present invention also provides this containing quinoxaline unit fluorene copolymer preparation method, comprises the steps:
1) compd A, B, the C that provide respectively following structural formula to represent,
Wherein, R
1, R
2be selected from H or C
1~C
20alkyl; R
3, R
4be selected from C
1~C
20alkyl, alkoxyl group, phenyl or phenoxy group; Ar
1for containing thiophene unit group;
2), under the condition existing in anaerobic, alkaline environment catalyst neutralisation, organic solvent, compd A, B, C are carried out to Suzuki linked reaction, obtain following general structure for (I) represent containing quinoxaline unit fluorene copolymer,
In formula, x+y=1, and x ≠ 0, y ≠ 0; Arbitrary integer of n=1~200,
The chemical equation of this Suzuki linked reaction is as follows:
In above-mentioned steps (2), catalyst loading is that described catalyst levels is 0.05%~20% of compd A mole dosage, this catalyzer is preferably the mixture of organic palladium or organic palladium and organophosphorus ligand, and wherein, organic palladium catalyzer is preferably Pd (PPh
3)
4, Pd
2(dba)
3or Pd (PPh
3)
2cl
2in at least one, organophosphorus ligand is preferably but be not limited only to P (o-Tol)
3, in the time that this catalyzer is the mixture of organic palladium and organophosphorus ligand, the mol ratio of organic palladium and organophosphorus ligand is preferably 1: 2~and 20; In above-mentioned steps (2), in the presence that there is no alkali, Suzuki coupling reaction is to be difficult to occur, not even reaction.In reaction, the power of alkali (negative ion) is not only depended in the impact of alkali, and to take into account cationic character, if too little transition state ylide (Pd) intermediate that is unfavorable for that generation is middle of positively charged ion, speed and efficiency too little when positively charged ion and conductively-closed reaction will significantly decline, suitably large cationic alkali, can accelerate the speed of Suzuki reaction, therefore, alkali is preferably at least one in alkali metal hydroxide, alkaline carbonate, tetraethyl ammonium hydroxide, as NaOH solution, Na
2cO
3solution, NaHCO
3solution or tetraethyl ammonium hydroxide solution etc., its addition is 1~15 times of compd A mole dosage.
Above-mentioned steps (2) must be reacted in oxygen-free environment, and this is because the catalyzer of Suzuki reaction is to be all afraid of oxygen, if the existence of aerobic in reaction system can cause catalyst deactivation, thereby causes Suzuki coupling reaction to carry out.Oxygen-free environment in reaction system can adopt vacuum or be full of rare gas element and realize, and is preferably full of rare gas element and realizes oxygen-free environment, and this rare gas element is the conventional rare gas element of the art, such as nitrogen, argon gas etc., preferred nitrogen.
In above-mentioned steps (2), the mol ratio of compd A, B, C tri-reactants is preferably 1.0: a: b, wherein, a, b should meet: a > 0, b > 0, a+b=1.0, organic solvent is preferably at least one in toluene, tetrahydrofuran (THF), trichloromethane or ethyl acetate, and its minimum interpolation should guarantee that this Suzuki coupling reaction can carry out smoothly; The temperature of this Suzuki reaction is preferably 65~120 ℃, and the reaction times is preferably 12~72 hours.
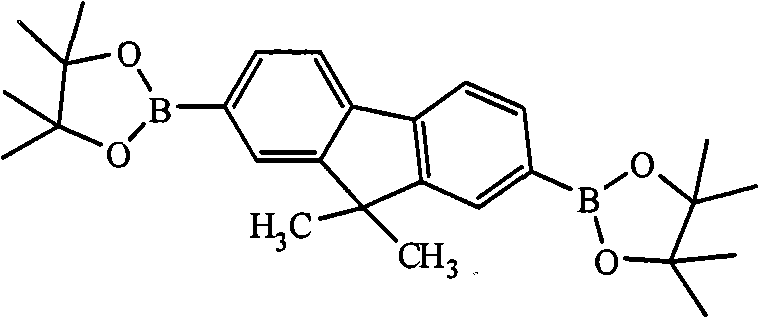
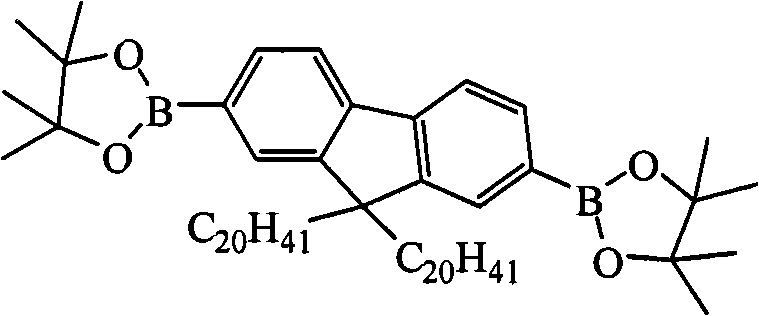
In above-mentioned steps (1), the preparation method of compd A is: in the system of anhydrous, oxygen-free environment and organic solvent, by bromo-2,7-bis-9, the alkyl derivative of 9-dialkyl group fluorenes, lithium and 2-isopropoxy-4,4,5,5-tetramethyl--1,3,2-bis-is assorted, and oxygen pentaborane reacts, and generates described compd A, and its chemical equation is as follows:
Preparation method's preferred version of above-claimed cpd A is: in the system of anhydrous, oxygen-free environment and organic solvent, first by 2,7-bis-is bromo-9, after 9-dialkyl group fluorenes reacts with the alkyl derivative of lithium, then adds 2-isopropoxy-4,4,5,5-tetramethyl--1,3,2-bis-is assorted, and oxygen pentaborane reacts, and generates described compd A.
In the preparation process of above-claimed cpd A, described 2,7-bis-is bromo-9, the alkyl derivative of 9-dialkyl group fluorenes, lithium and 2-isopropoxy-4, and 4,5,5-tetramethyl--1, the mol ratio of 3,2-, bis-assorted oxygen pentaborane three reactants is preferably 1: 2.0~and 4.0: 2.0~4.0; The alkyl derivative of lithium is preferably at least one in n-Butyl Lithium, tert-butyl lithium, lithium methide, phenyl lithium; Organic solvent is preferably at least one in tetrahydrofuran (THF), ether, methylene dichloride, trichloromethane or ethyl acetate; The temperature of reaction of the preparation process of compd A is preferably-100~-25 ℃, and the time is preferably 24~48 hours.
The preparation of above-claimed cpd A must be reacted in anhydrous and oxygen-free environment, and this is because there is water can cause the alkyl derivative of lithium, as butyllithium is decomposed, makes this reaction failure; Aerobic can cause 2-isopropoxy-4,4,5,5-tetramethyl--1, and 3,2-bis-is assorted, and oxygen pentaborane is oxidized, makes to react failure or productive rate greatly reduces.Oxygen-free environment can adopt vacuum or be full of rare gas element and realize, and is preferably full of rare gas element and realizes oxygen-free environment, and this rare gas element is the conventional rare gas element of the art, such as nitrogen, argon gas etc., preferred nitrogen.
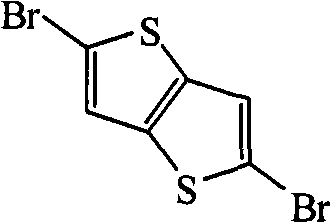
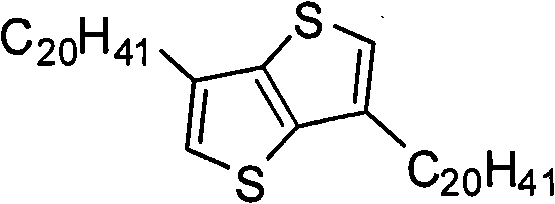
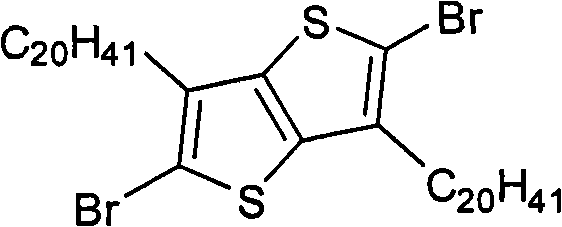
In above-mentioned steps (1), the preparation method of compd B is: under the condition existing at organic solvent, by the Ar not replaced by bromine
1compounds and N-bromosuccinimide carry out bromination reaction, generate described compd B, and its chemical equation is as follows:
Ar
1+NBS→Br-Ar
1-Br(B)
Wherein, described Ar
1the mol ratio of compounds and N-bromosuccinimide is preferably 1: 2.0~and 2.5; Described organic solvent is preferably N-DMF, tetrahydrofuran (THF), ether, methylene dichloride, trichloromethane, ethyl acetate or acetic acid; Temperature of reaction prepared by described compd B is-5~30 ℃, and the time is preferably 12~48 hours.
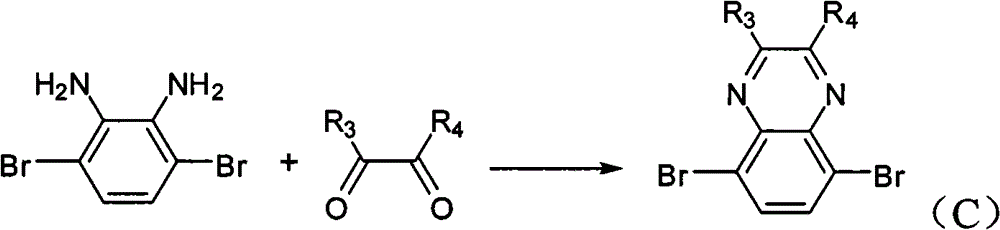
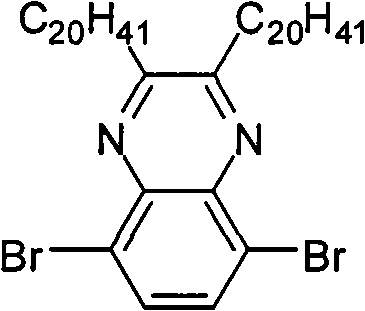
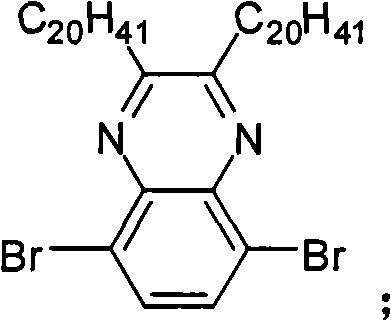
In above-mentioned steps (1), the preparation method of Compound C is: under the condition of catalyzer and organic solvent existence, by 3,6-dibromobenzene-1,2-diamines reacts with alkyl diketone, generates described Compound C, and its chemical equation is as follows:
Wherein, described 3,6-dibromobenzene-1, the mol ratio of 2-diamines and alkyl diketone is preferably 1: 1~and 2; Described catalyzer is preferably tosic acid (p-TSA), acetic acid, NH
4cl, its consumption is preferably 3,6-dibromobenzene-1,0.01~10 times of 2-diamines mole dosage; Described organic solvent trichloromethane, tetrahydrofuran (THF), ether, methylene dichloride or ethyl acetate; The temperature of reaction of the preparation process of described Compound C is preferably 20~40 ℃, and the time is preferably 12~48 hours.
Above-described embodiment only need mix each reactant under certain condition and carry out Suzuki coupling reaction and can obtain described final target product containing quinoxaline unit fluorene copolymer preparation method, without special equipment and environmental requirement, its preparation method technique is simple, productive rate is high, and mild condition, be easy to operate and control, be suitable for suitability for industrialized production.
Therefore, above-described embodiment can be at organic photoelectrical material, polymer solar battery, organic electroluminescence device, organic field effect tube, organic light storage device, organic non-linear optical properties or/and apply in organic laser apparatus containing quinoxaline unit fluorene copolymer.
Now in conjunction with the embodiments, the present invention is further elaborated.
Embodiment 1
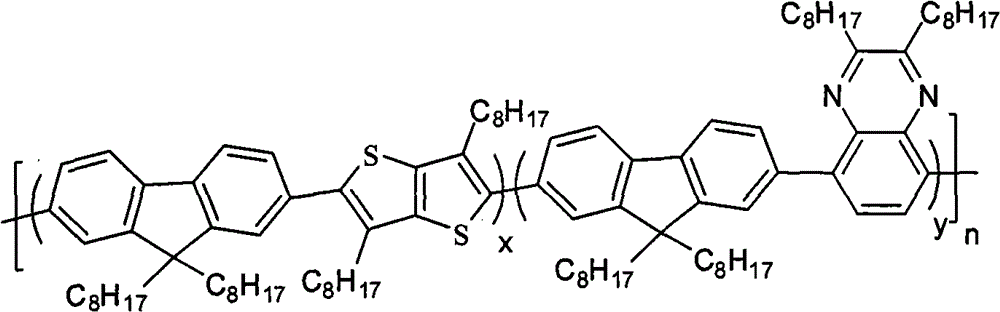
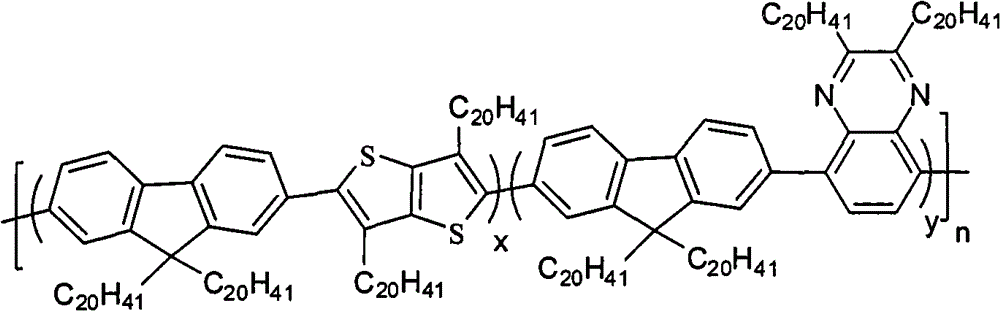
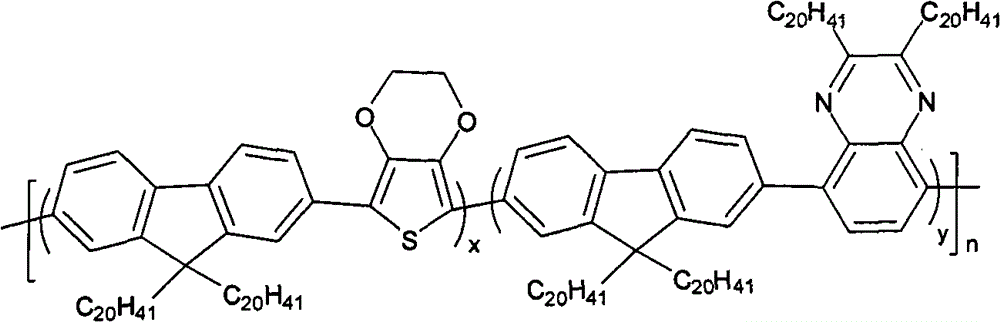
The multipolymer of the present embodiment 1 be following molecular structural formula represent containing quinoxaline unit fluorene copolymer I
1, its structural formula is as follows:
Its preparation process is as follows:
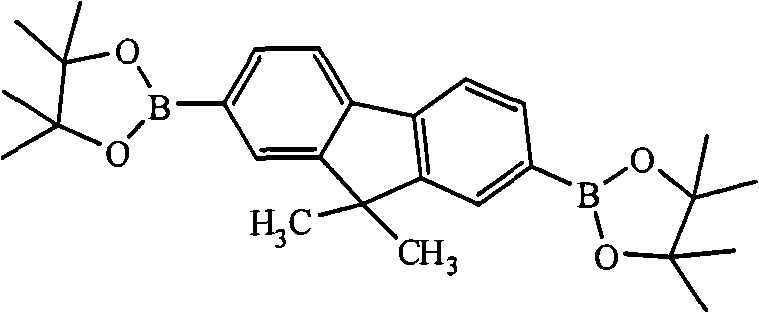
Two (4,4,5,5-tetramethyl--1, the assorted oxygen pentaborane bases of 3,2-bis-)-9 of (1) 2,7-, the preparation of 9-dimethyl fluorene, its structural formula is as follows:
Preparation detailed process is: at-100 ℃, anhydrous, under nitrogen protection condition, by 20.00mL, concentration is that the n-Butyl Lithium oil solution of 1.00M is added to and fills 2 of 3.52g, 7-bis-bromo-9, in the reaction flask of 9-dimethyl fluorene and 100mL tetrahydrofuran (THF), stir after 2 hours, slowly drip 4.17mL 2-isopropoxy-4, 4, 5, 5-tetramethyl--1, 3, the assorted oxygen pentaborane of 2-bis-, return to room temperature, continue to stir 24 hours, question response finishes, reaction solution is poured into water, extracted with diethyl ether, anhydrous magnesium sulfate drying, revolve steaming, column chromatography for separation obtains solid product, the MALDI-TOF-MS (m/z) of this product: 446.3 (M
+),
The preparation of (2) 2,5-dibromo thiophene [3,2-b] thiophthenes, its structural formula is as follows:
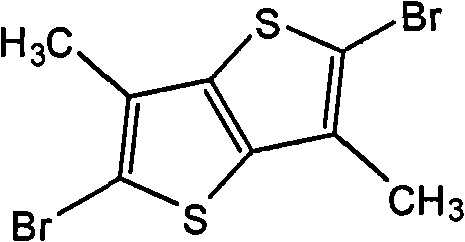
Preparation detailed process is: under-5 ℃ of conditions, the NBS of 3.56g is added in the reaction flask of the DMF that fills 1.40g thiophene [3,2-b] thiophthene and 100mL in batches, stirs 48 hours.Reaction finishes, and pours reaction solution in frozen water cancellation, dichloromethane extraction, and anhydrous magnesium sulfate drying, revolves steaming, and column chromatography for separation obtains solid product, the MALDI-TOF-MS (m/z) of this product: 298.0 (M
+);
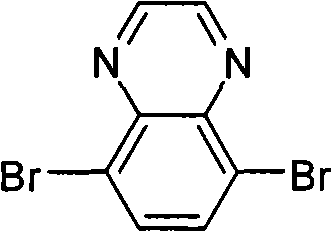
The preparation of (3) 5,8-dibromo quinoline woodss, its structural formula is as follows:
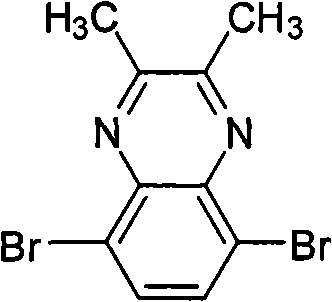
Preparation detailed process is: at 20 ℃, and by 2.66g 3,6-dibromobenzene-1,2-diamines is dissolved in 100mL chloroform, adds 0.58g oxalic dialdehyde and 0.17gp-TSA, reacts 24 hours.Reaction finishes, reaction solution poured in saturated sodium bicarbonate solution, and dichloromethane extraction, anhydrous magnesium sulfate drying, suction filtration, revolves steaming, and column chromatography for separation obtains product, the MALDI-TOF-MS (m/z) of this product: 287.9 (M
+);
(4) containing quinoxaline unit fluorene copolymer I
1, its structural formula is as follows:
Preparation detailed process is: under nitrogen protection; toward containing 0.446g 2,7-two (4,4; 5; 5-tetramethyl--1, the assorted oxygen pentaborane base of 3,2-bis-)-9; 2 of 9-dimethyl fluorene, 0.150g; 5 of 5-dibromo thiophene [3,2-b] thiophthene, 0.144g, the Pd (PPh of 8-dibromo quinoline woods, 0.101g
3)
4reaction flask in add the 6mL 20%Wt tetraethyl ammonium hydroxide aqueous solution and toluene (20mL), after nitrogen replacement 1 hour, be heated to 65 ℃, react after 72 hours, add 1.1mL bromobenzene reaction 12 hours, add 0.011g phenylo boric acid to continue reaction 12 hours, reaction finishes, methyl alcohol sedimentation three times again, suction filtration, vacuum-drying obtains solid product, the GPC:Mn=5673 of this product, PDI=1.5.
Embodiment 2
The multipolymer of the present embodiment 2 be following molecular structural formula represent containing quinoxaline unit fluorene copolymer I
2preparation, its structural formula is as follows:
Its preparation process is as follows:
Two (4,4,5,5-tetramethyl--1, the assorted oxygen pentaborane bases of 3,2-bis-)-9 of (1) 2,7-, the preparation of 9-dimethyl fluorene, its structural formula is as follows, and preparation process refers to embodiment 1.
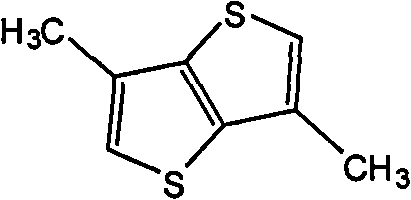
The preparation of (2) 3,6-dimethyl base thieno-[3,2-b] thiophene, its structural formula is as follows:
Preparation detailed process is: by 12.00g 3,6-bis-bromos-thieno-[3,2-b] thiophene and 132mg (1,1 '-bis-(diphenylphosphino) ferrocene) Palladous chloride (II) is added in the tubular vial of 100mL that stirring rod is housed, sealing, purging with nitrogen gas, adding 30mL tetrahydrofuran (THF) and 50mL methyl zinc bromide (is dissolved in methyl zinc bromide in tetrahydrofuran solution, its concentration is 1.0M), stirring at room temperature 30 minutes, heats in the microwave reactor at 140 ℃ 50 minutes.Cooling, with MTBE dilution, and use respectively rare HCl solution, salt water washing, anhydrous sodium sulfate drying, suction filtration, revolves steaming.Column chromatography for separation, recrystallization obtains product, the MALDI-TOF-MS (m/z) of this product: 168.3 (M
+);
(3) 2,5-bis-are bromo-3, and 6-thioxene is the preparation of [3,2-b] thiophene also, and its structural formula is as follows:
Preparation detailed process is: under 30 ℃ of conditions, the NBS of 16.00g is added in batches and fills 3 of 6.72g, 6-thioxene also in the reaction flask of [3,2-b] thiophene and 200mL ether, stirs 12 hours.Reaction finishes, and pours reaction solution in frozen water cancellation, dichloromethane extraction, and anhydrous magnesium sulfate drying, revolves steaming, and column chromatography for separation obtains solid product, the MALDI-TOF-MS (m/z) of this product: 326.1 (M
+).
(4) 5,8-bis-are bromo-2, the preparation of 3-dimethyl quinoline woods, and its structural formula is as follows:
Preparation detailed process is: at 40 ℃, and by 3 of 2.66g, 6-dibromobenzene-1,2-diamines is dissolved in 120mL chloroform, adds 2 of 1.72g, the p-TSA of 3-dimethyl diketone and 0.24g, react 22 hours, question response finishes, and reaction solution is poured in saturated sodium bicarbonate solution, dichloromethane extraction, anhydrous magnesium sulfate drying, suction filtration, revolves steaming, column chromatography for separation obtains product, the MALDI-TOF-MS (m/z) of this product: 316.0 (M
+).
(5) containing quinoxaline unit fluorene copolymer I
2, its structural formula is as follows:
Preparation detailed process is: under nitrogen protection, and toward containing 2 of 0.893g, 7-two (4; 4,5,5-tetramethyl--1; 3; the assorted oxygen pentaborane base of 2-bis-)-9,2 of 9-dimethyl fluorene, 0.326g, 5-bis-bromo-3; 6-thioxene also [3; 2-b] thiophene, 0.316g 5,8-bis-is bromo-2, the Pd (PPh of 3-dimethyl quinoline woods, 0.220g
3)
2cl
2reaction flask in add the NaOH aqueous solution and the trichloromethane (50mL) of 15mL 20%Wt, nitrogen replacement, after 1 hour, is heated to 100 ℃, reacts 48 hours.Add the reaction of 2.0mL bromobenzene after 13 hours, add 0.020g phenylo boric acid to continue reaction 13 hours, question response finishes, methyl alcohol sedimentation three times, and suction filtration, vacuum-drying obtains solid product, the GPC:Mn=19863 of this product, PDI=1.7.
Embodiment 3
The multipolymer of the present embodiment 3 be following molecular structural formula represent containing quinoxaline unit fluorene copolymer I
3preparation, its structural formula is as follows:
Its preparation process is as follows:
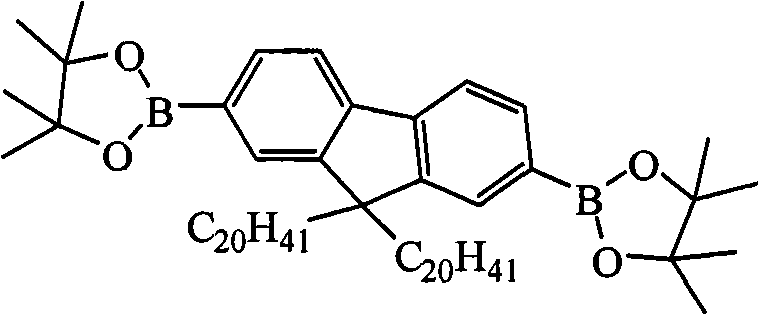
Two (4,4,5,5-tetramethyl--1, the assorted oxygen pentaborane bases of 3,2-bis-)-9 of (1) 2,7-, the preparation of 9-dioctyl fluorene, its structural formula is as follows:
Preparation detailed process is: under-78 ℃, condition of nitrogen gas, be added to and fill 2 of 11.00g with the tert-butyl lithium solution that syringe is 2.00M by 23.00mL, concentration, 7-bis-is bromo-9, in two mouthfuls of flasks of 9-dioctyl fluorene and 100.00mL ethyl acetate, stir and after 2 hours, slowly drip 9.80mL 2-isopropoxy-4,4,5,5-tetramethyl--1,3, the assorted oxygen pentaborane of 2-bis-, returns to room temperature, continues to stir 25 hours.Reaction finishes, reaction solution is poured into water, and extracted with diethyl ether, anhydrous magnesium sulfate drying, revolves steaming, and column chromatography for separation obtains solid product, the MALDI-TOF-MS (m/z) of this product: 642.6 (M
+);
The preparation of (2) 3,6-dioctyl thieno-[3,2-b] thiophene, its structural formula is as follows:
Preparation detailed process is: by 3 of 2.00g, 6-bis-bromos-thieno-[3,2-b] thiophene and 22mg (1,1 '-bis-(diphenylphosphino) ferrocene) Palladous chloride (II) is added in the tubular vial of 50mL that stirring rod is housed, sealing, purging with nitrogen gas, adding 15mL tetrahydrofuran (THF) and 19mL octyl group zinc bromide (is dissolved in methyl zinc bromide in tetrahydrofuran solution, its concentration is 1.0M), stirring at room temperature 15 minutes, heats in the microwave reactor at 150 ℃ 40 minutes.Cooling, with MTBE dilution, and use respectively rare HCl solution, salt water washing, anhydrous sodium sulfate drying, suction filtration, revolves steaming.Column chromatography for separation, recrystallization obtains product, the MALDI-TOF-MS (m/z) of this product: 364.7 (M
+);
(3) 2,5-bis-are bromo-3, the preparation of 6-dioctyl thieno-[3,2-b] thiophene, and its structural formula is as follows:
Preparation detailed process is: under ice bath, lucifuge condition, the NBS of 1.97g is added in batches and fills 2.02g 3,6-dioctyl thieno-[3,2-b] in thiophene and the ethyl acetate of 1: 1 by volume of 60mL and the mixed solvent reaction flask of methylene dichloride, stirring at room temperature 15 hours, question response finishes, pour reaction solution in frozen water cancellation, dichloromethane extraction, anhydrous magnesium sulfate drying, revolve steaming, column chromatography for separation obtains solid product, the MALDI-TOF-MS (m/z) of this product: 522.5 (M
+);
(4) 5,8-bis-are bromo-2, the preparation of 3-dioctyl quinoline woods, and its structural formula is as follows:
Preparation detailed process is: at 30 ℃, and by 3 of 2.66g, 6-dibromobenzene-1,2-diamines is dissolved in 120mL ether, adds 3.09g octadecane-9,10-diketone and 0.11g p-TSA, react 12 hours, question response finishes, and reaction solution is poured in saturated sodium bicarbonate solution, dichloromethane extraction, anhydrous magnesium sulfate drying, suction filtration, revolves steaming, column chromatography for separation obtains product, the MALDI-TOF-MS (m/z) of this product: 512.4 (M+);
(5) containing quinoxaline unit fluorene copolymer I
3, its structural formula is as follows:
Preparation detailed process is: under nitrogen protection, and toward containing 2 of 0.446g, 7-two (4; 4,5,5-tetramethyl--1; 3; the assorted oxygen pentaborane base of 2-bis-)-9,2 of 9-dioctyl fluorene, 0.261g, 5-bis-bromo-3; 6-dioctyl thieno-[3; 2-b] thiophene, 0.256g 5,8-bis-is bromo-2,3-dioctyl quinoline woods, 0.129gPd
2(dba)
3/ P (o-Tol)
3in the reaction flask of mixture, add toluene and the tetrahydrofuran (THF) mixed solvent (30mL) of the 10mL 20%Wt tetraethyl ammonium hydroxide aqueous solution and 1: 0.5 by volume, after nitrogen replacement 2 hours, be heated to 110 ℃, react 55 hours, add again 1.1mL bromobenzene reaction 14 hours, then add 0.011g phenylo boric acid to continue reaction 14 hours, reaction finishes, methyl alcohol sedimentation three times, suction filtration, vacuum-drying obtains solid product, the GPC:Mn=44520 of this product, PDI=1.5.
Embodiment 4
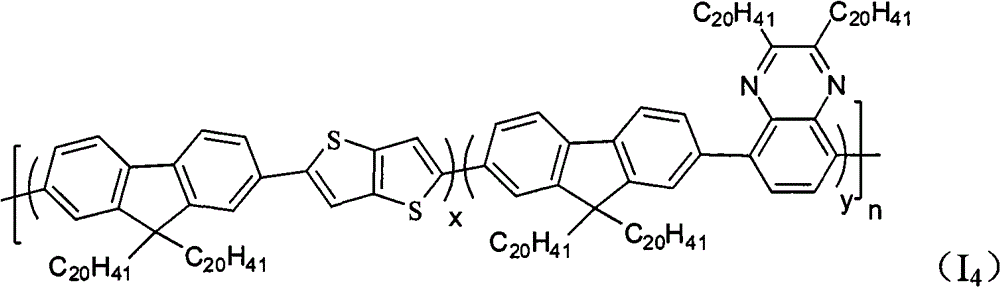
The multipolymer of the present embodiment 4 be following molecular structural formula represent containing quinoxaline unit fluorene copolymer I
4preparation, its structural formula is as follows:
Its preparation process is as follows:
Two (4,4,5,5-tetramethyl--1, the assorted oxygen pentaborane bases of 3,2-bis-)-9 of (1) 2,7-, the preparation of the two eicosyl fluorenes of 9-, its structural formula is as follows:
Preparation detailed process is: at-25 ℃, under condition of nitrogen gas, by 16.50mL, concentration is that the phenyl lithium oil solution of 2.00M is added to and fills 2 of 8.87g, 7-bis-bromo-9, in the reaction flask of the two eicosyl fluorenes of 9-and 150mL methylene dichloride, stir 2-isopropoxy-4 that slowly drip 5.50mL after 2 hours, 4, 5, 5-tetramethyl--1, 3, the assorted oxygen pentaborane of 2-bis-, return to room temperature, continue to stir 44 hours, question response finishes, reaction solution is poured into water, extracted with diethyl ether, anhydrous magnesium sulfate drying, revolve steaming, column chromatography for separation obtains solid product, the MALDI-TOF-MS (m/z) of this product: 979.2 (M
+),
The preparation of (2) 2,5-dibromo thiophene [3,2-b] thiophthenes, its structural formula is as follows, and preparation process refers to embodiment 1;
(3) 5,8-bis-are bromo-2, the preparation of the two icosyl quinoline woodss of 3-, and its structural formula is as follows:
Preparation detailed process is: at 25 ℃, and by 2.00g 3,6-dibromobenzene-1,2-diamines is dissolved in 80mL chloroform, adds 6.33g dotetracontane-21, and 22-diketone and 0.10g p-TSA react 15 hours.Reaction finishes, reaction solution poured in saturated sodium bicarbonate solution, and dichloromethane extraction, anhydrous magnesium sulfate drying, suction filtration, revolves steaming, and column chromatography for separation obtains product, the MALDI-TOF-MS (m/z) of this product: 849.0 (M
+);
(4) containing quinoxaline unit fluorene copolymer I
4, its structural formula is as follows:
Preparation detailed process is: under nitrogen protection, and toward containing 0.979g 2,7-two (4; 4; 5,5-tetramethyl--1,3; the assorted oxygen pentaborane base of 2-bis-)-9; the two eicosyl fluorenes of 9-, 0.149g 2,5-dibromo thiophene [3,2-b] thiophthene, 0.425g 5; 8-bis-is bromo-2, the two icosyl quinoline woodss of 3-, 0.145g Pd (PPh
3)
4reaction flask in add the Na of 10mL 20%W
2cO
3the aqueous solution and toluene (40mL), after 2 hours, be heated to 110 ℃ with nitrogen replacement, react 61 hours, add again the reaction of 1.0mL bromobenzene after 15 hours, add 0.010g phenylo boric acid to continue reaction 15 hours, question response finishes, methyl alcohol sedimentation three times, suction filtration, vacuum-drying obtains solid product, the GPC:Mn=62173 of this product, PDI=1.8;
Embodiment 5
The multipolymer of the present embodiment 5 be following molecular structural formula represent containing quinoxaline unit fluorene copolymer I
5preparation, its structural formula is as follows:
Its preparation process is as follows:
Two (4,4,5,5-tetramethyl--1, the assorted oxygen pentaborane bases of 3,2-bis-)-9 of (1) 2,7-, the preparation of the two eicosyl fluorenes of 9-, its structural formula is as follows, and preparation process refers to embodiment 1;
The preparation of two icosyl thieno-[3, the 2-b] thiophene of (2) 3,6-, its structural formula is as follows:
Preparation detailed process is: by 3 of 3.00g, 6-bis-bromos-thieno-[3, 2-b] thiophene and 34mg (1, 1 '-bis-(diphenylphosphino) ferrocene) Palladous chloride (II) is added in the tubular vial of 100mL that stirring rod is housed, sealing, adopt purging with nitrogen gas, adding the tetrahydrofuran (THF) of 40mL and the icosyl zinc bromide of 30mL (is dissolved in methyl zinc bromide in tetrahydrofuran solution, its concentration is 1.0M), stirring at room temperature 30 minutes, in microwave reactor at 150 ℃, heat 1 hour, cooling, dilute with MTBE, and use respectively rare HCl solution, salt water washing, anhydrous sodium sulfate drying, suction filtration, revolve steaming.Column chromatography for separation, recrystallization obtains product, the MALDI-TOF-MS (m/z) of this product: 701.3 (M
+);
(3) 2,5-bis-are bromo-3, the preparation of two icosyl thieno-[3, the 2-b] thiophene of 6-, and its structural formula is as follows:
Preparation detailed process is: under ice bath, lucifuge condition, 1.88g NBS is added in batches and fills 3.50g 3, in the reaction flask of two icosyl thieno-[3, the 2-b] thiophene of 6-and 50mL DMF, stirring at room temperature 22 hours, reaction finishes, and pours reaction solution into cancellation in frozen water, dichloromethane extraction, anhydrous magnesium sulfate drying, revolve steaming, column chromatography for separation obtains solid product, the MALDI-TOF-MS (m/z) of this product: 859.1 (M
+);
(4) 5,8-bis-are bromo-2, the preparation of the two icosyl quinoline woodss of 3-, and its structural formula is as follows, and preparation process refers to embodiment 1,
(5) containing quinoxaline unit fluorene copolymer I
5, its structural formula is as follows:
Preparation detailed process is: under nitrogen protection, and toward containing 2 of 0.979g, 7-two (4; 4,5,5-tetramethyl--1; 3; the assorted oxygen pentaborane base of 2-bis-)-9,2 of the two eicosyl fluorenes of 9-, 0.430g, 5-bis-bromo-3; the two icosyl thieno-s [3 of 6-; 2-b] thiophene, 0.425g 5,8-bis-is bromo-2, the two icosyl quinoline woodss of 3-, 0.145gPd (PPh
3)
4reaction flask in add the 8mL 20%Wt tetraethyl ammonium hydroxide aqueous solution and toluene (40mL).After nitrogen replacement 1 hour, be heated to 120 ℃, react 67 hours.Add the reaction of 1.0mL bromobenzene after 15 hours, add 0.010g phenylo boric acid to continue reaction 15 hours, question response finishes, methyl alcohol sedimentation three times, and suction filtration, vacuum-drying obtains solid product, the GPC:Mn=81092 of this product, PDI=1.9.
Embodiment 6
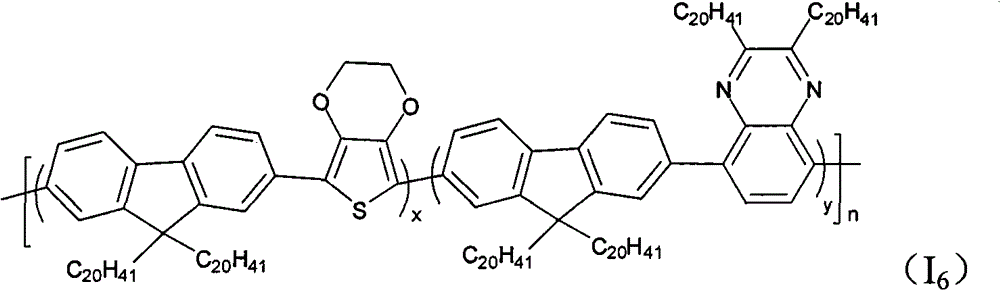
The multipolymer of the present embodiment 6 be following molecular structural formula represent containing quinoxaline unit fluorene copolymer I
6preparation, its structural formula is as follows:
Its preparation process is as follows:
Two (4,4,5,5-tetramethyl--1, the assorted oxygen pentaborane bases of 3,2-bis-)-9 of (1) 2,7-, the preparation of the two eicosyl fluorenes of 9-, its structural formula is as follows, and preparation process refers to embodiment 1;
(2) 2,5-bis-are bromo-3, the preparation of 4-vinyl dioxy thiophene:
Preparation detailed process is: under ice bath, lucifuge condition, 3.98g NBS is added in batches and fills 1.41g 3, and in the reaction flask of 4-vinyl dioxy thiophene and 40mL DMF, stirring at room temperature 24 hours.Reaction finishes, and pours reaction solution in frozen water cancellation, dichloromethane extraction, and anhydrous magnesium sulfate drying, revolves steaming, and column chromatography for separation obtains solid product, the MALDI-TOF-MS (m/z) of this product: 300.0 (M
+).
(3) 5,8-bis-are bromo-2, the preparation of the two icosyl quinoline woodss of 3-, and its structural formula is as follows, and preparation process refers to embodiment 1,
(4) containing quinoxaline unit fluorene copolymer I
6, its structural formula is as follows:
Preparation detailed process is: under nitrogen protection, and toward containing 2 of 0.979g, 7-two (4; 4; 5,5-tetramethyl--1,3; the assorted oxygen pentaborane base of 2-bis-)-9; 2 of the two eicosyl fluorenes of 9-, 0.225g, 5-bis-is bromo-3,5 of 4-vinyl dioxy thiophene, 0.425g; 8-bis-is bromo-2, the two icosyl quinoline woodss of 3-, 0.141g Pd (PPh
3)
4reaction flask in add the 10mL 20%Wt tetraethyl ammonium hydroxide aqueous solution and toluene (45mL), after nitrogen replacement 1 hour, be heated to 100 ℃, react 68 hours, add again the reaction of 1.5mL bromobenzene after 13 hours, add 0.010g phenylo boric acid to continue reaction 13 hours, question response finishes, methyl alcohol sedimentation three times, suction filtration, vacuum-drying obtains solid product, the GPC:Mn=57800 of this product, PDI=1.8.
Application Example 7
Apply in solar cell device as active coating containing quinoxaline unit fluorene copolymer using embodiment 1:
Refer to Fig. 2, adopt above-described embodiment 1 to contain the solar cell device of quinoxaline unit fluorene copolymer as active coating, it comprises the glass-base 11 stacking gradually, transparent anode 12, middle supplementary layer 13, active coating 14, negative electrode 15, middle supplementary layer 13 adopts polyethylene dioxy base thiophene: polystyrene-sulfonic acid matrix material (referred to as PEDOT:PSS), active coating 14 comprises electron donor material and electron acceptor material, prepared by electron donor material employing above-described embodiment 1 contains quinoxaline unit fluorene copolymer, electron acceptor material can be [6, 6] phenyl-C
61-methyl-butyrate (referred to as PCBM).Transparent anode 12 can adopt tin indium oxide (referred to as ITO), is preferably the tin indium oxide that square resistance is 10-20 Ω/.Negative electrode 15 can adopt aluminium electrode or double-metal layer electrode, such as Ca/Al or Ba/Al etc.Wherein, glass-base 11 can be used as bottom, when making, choose ito glass, and after ultrasonic cleaning, with oxygen-Plasma processing, supplementary layer 13 in the middle of applying on ito glass, then prepared by embodiment 1 be coated on after by blend containing quinoxaline unit fluorene copolymer and electron acceptor material in the middle of on supplementary layer 13, form active coating 14, and then by vacuum evaporation technology deposition cathode 15 on active coating 14, obtain above-mentioned solar cell device.In a preferred embodiment, the thickness of transparent anode 12, middle supplementary layer 13, active coating 14, double-metal layer Ca and Al layer be respectively 180,40,160,70nm.
As shown in Figure 2, under illumination, light transmission glass-base 11 and ITO electrode 12, prepared by the embodiment 1 in active coating 14 absorbs luminous energy containing quinoxaline unit fluorene copolymer, and produces exciton, these excitons move to the interface of electron donor(ED)/acceptor material again, and by transfer transport to electron acceptor material, as PCBM, realize the separation of electric charge, thereby form current carrier freely, i.e. electronics and hole freely.These freely electronics along electron acceptor material to metallic cathode transmission collected by negative electrode, hole is along electron donor material to ito anode transmission collected by anode freely, thereby forms photoelectric current and photovoltage, realizes opto-electronic conversion, when external load 16, can power to it.In this process, prepared by embodiment 1 contains quinoxaline unit fluorene copolymer because it has very wide spectral response range, can utilize more fully luminous energy, to obtain higher photoelectric transformation efficiency, increases the electricity generation ability of solar cell device.And this organic materials can also alleviate the quality of solar cell device, and can make by technology such as spin coatings, be convenient to large batch of preparation.
Application Example 8
That prepares with above-described embodiment 2 applies in organic electroluminescence device containing quinoxaline unit fluorene copolymer:
Refer to Fig. 3, show the organic electroluminescence device containing quinoxaline unit fluorene copolymer that adopts above-described embodiment 2 to prepare, it comprises the glass-base 21, transparent anode 22, luminescent layer 23, buffer layer 24, the negative electrode 25 that are cascading.Transparent anode 22 can adopt tin indium oxide (referred to as ITO), is preferably the tin indium oxide that square resistance is 10-20 Ω/.Luminescent layer 23 comprise prepared by above-described embodiment 2 containing quinoxaline unit fluorene copolymer.Buffer layer 24 can adopt LiF etc., but is not limited to this.Negative electrode 25 can be but be not limited to metal A l etc.Thereby in a specific embodiment, organic electroluminescence device representation is: prepared by ITO/ embodiment 2 contains quinoxaline unit fluorene copolymer/LiF/Al.Each layer can adopt existing method to form, and can be formed on ITO by spin coating technique containing quinoxaline unit fluorene copolymer and prepared by embodiment 2.On this luminescent layer, can adopt vacuum evaporation LiF buffer layer, on buffer layer, can adopt evaporation metal Al, as the negative electrode of device.
Application Example 9
That prepares with embodiment 3 applies in organic field effect tube containing quinoxaline unit fluorene copolymer:
Refer to Fig. 4, show the organic field effect tube containing quinoxaline unit fluorene copolymer that adopts above-described embodiment 3 to prepare, it comprises substrate 31, insulation layer 32, decorative layer 33, the organic semiconductor layer 34 being cascading and is located at source electrode 35 and the drain electrode 36 on organic semiconductor layer 34.Wherein, substrate 31 can be but be not limited to highly doped silicon chip (Si), and insulation layer 32 can be but be not limited to micro-nano (as 450nm) thick SiO
2.Organic semiconductor layer 34 adopt prepared by above-described embodiment 3 containing quinoxaline unit fluorene copolymer.Source electrode 35 and drain electrode 36 all can adopt but be not limited to gold.Decorative layer 33 can be but be not limited to octadecyl trichlorosilane alkane.Substrate 31, insulation layer 32, decorative layer 33 and source electrode 35 and drain electrode 36 all can adopt existing method to form.Organic semiconductor layer 34 can be prepared by above-described embodiment 3 being spun on the insulation layer 32 of being modified by decorative layer 33 containing quinoxaline unit fluorene copolymer.
The foregoing is only preferred embodiment of the present invention, not in order to limit the present invention, all any modifications of doing within the spirit and principles in the present invention, be equal to and replace and improvement etc., within all should being included in protection scope of the present invention.
Claims (9)
1. containing a quinoxaline unit fluorene copolymer, its general formula of molecular structure is following (I):
In formula, x+y=1, and x ≠ 0, y ≠ 0; Arbitrary integer of n=1~200; R
1, R
2be selected from H or C
1~C
20alkyl; R
3, R
4be selected from C
1~C
20alkyl, alkoxyl group, phenyl or phenoxy group; Described Ar
1for following one or more groups:
In formula, R
5, R
6, R
7, R
8, R
9, R
10be selected from H, C
1~C
20alkyl; R
11, R
12, R
13, R
14, R
15be selected from C
1~C
20alkyl; M is number of repeat unit, arbitrary integer of m=1~20.
2. containing a quinoxaline unit fluorene copolymer preparation method, comprise the steps:
The compd A, B, the C that provide respectively following structural formula to represent,
Wherein, R
1, R
2be selected from H or C
1~C
20alkyl; R
3, R
4be selected from C
1~C
20alkyl, alkoxyl group, phenyl or phenoxy group; Ar
1for following one or more groups:
In formula, R
5, R
6, R
7, R
8, R
9, R
10be selected from H, C
1~C
20alkyl; R
11, R
12, R
13, R
14, R
15be selected from C
1~C
20alkyl; M is number of repeat unit, arbitrary integer of m=1~20;
Under the condition existing in anaerobic, alkaline environment catalyst neutralisation, organic solvent, compd A, B, C are carried out to Suzuki linked reaction, obtain following general structure for (I) represent containing quinoxaline unit fluorene copolymer,
In formula, x+y=1, and x ≠ 0, y ≠ 0; Arbitrary integer of n=1~200.
3. according to claim 2 containing quinoxaline unit fluorene copolymer preparation method, it is characterized in that:
The preparation method of described compd A comprises the steps: in the system of anhydrous, oxygen-free environment and organic solvent, by 2,7-bis-bromo-9, the alkyl derivative of 9-dialkyl group fluorenes, lithium and 2-isopropoxy-4,4,5,5-tetramethyl--1,3,2-bis-is assorted, and oxygen pentaborane reacts, and generates described compd A;
Described 2,7-bis-is bromo-9, the alkyl derivative of 9-dialkyl group fluorenes, lithium and 2-isopropoxy-4, and 4,5,5-tetramethyl--1, the mol ratio of 3,2-, bis-assorted oxygen pentaborane three reactants is 1: 2.0~4.0: 2.0~4.0;
The alkyl derivative of described lithium is at least one in n-Butyl Lithium, tert-butyl lithium, lithium methide, phenyl lithium;
Described organic solvent is at least one in tetrahydrofuran (THF), ether, methylene dichloride, trichloromethane or ethyl acetate;
The temperature of reaction of the preparation process of described compd A is-100~-25 ℃.
4. according to claim 2 containing quinoxaline unit fluorene copolymer preparation method, it is characterized in that:
The preparation method of described compd B is: under the condition existing at organic solvent, by the Ar not replaced by bromine
1compounds enters bromination row with N-bromosuccinimide and reacts, and generates described compd B;
Described Ar
1the mol ratio of compounds and N-bromosuccinimide is 1: 2.0~2.5;
Described organic solvent is N-DMF, tetrahydrofuran (THF), ether, methylene dichloride, trichloromethane, ethyl acetate or acetic acid;
The temperature of reaction of the preparation process of described compd B is-5~30 ℃.
5. according to claim 2 containing quinoxaline unit fluorene copolymer preparation method, it is characterized in that:
The preparation method of described Compound C is: under the condition of catalyzer and organic solvent existence, by 3,6-dibromobenzene-1,2-diamines reacts with alkyl diketone, generates described Compound C;
Described 3,6-dibromobenzene-1, the mol ratio of 2-diamines and alkyl diketone is 1: 1~2;
Described catalyzer is tosic acid, acetic acid, NH
4cl etc., its consumption is 3,6-dibromobenzene-1,0.01~10 times of 2-diamines mole dosage;
Described organic solvent is trichloromethane, tetrahydrofuran (THF), ether, methylene dichloride or ethyl acetate;
The temperature of reaction of the preparation process of described Compound C is 20~40 ℃.
6. according to claim 2 containing quinoxaline unit fluorene copolymer preparation method, it is characterized in that: in described Suzuki linked reaction, the mol ratio of described compd A, B, C is 1.0: a: b, wherein, a > 0, b > 0, a+b=1.0.
According to described in claim 2 or 6 containing quinoxaline unit fluorene copolymer preparation method, it is characterized in that: in described Suzuki linked reaction,
Described catalyzer is the mixture of organic palladium catalyzer or organic palladium catalyzer and organophosphorus ligand, and its consumption is 0.05%~20% of compd A mole dosage;
Alkali in described alkaline environment is at least one in alkali metal hydroxide, basic metal carbonate, tetraethyl ammonium hydroxide, and its consumption is 1~15 times of compd A mole dosage;
Described organic solvent is at least one in toluene, tetrahydrofuran (THF), trichloromethane or ethyl acetate;
The temperature of described Suzuki linked reaction is 65~120 ℃, and the time is 12~72 hours.
8. according to claim 7 containing quinoxaline unit fluorene copolymer preparation method, it is characterized in that: described organic palladium catalyzer is Pd
2(dba)
3, Pd (PPh
3)
4, Pd (PPh
3)
2cl
2in at least one; Described organophosphite ligand body catalyst is P (o-Tol)
3.
According to claim 1 containing quinoxaline unit fluorene copolymer at organic photoelectrical material, polymer solar battery, organic electroluminescence device, organic field effect tube, organic light storage device, organic non-linear optical properties or/and the application in organic laser apparatus.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201010196323.4A CN102276799B (en) | 2010-06-09 | 2010-06-09 | Quinoxaline unit-containing fluorene copolymer, and preparation method and application thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201010196323.4A CN102276799B (en) | 2010-06-09 | 2010-06-09 | Quinoxaline unit-containing fluorene copolymer, and preparation method and application thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN102276799A CN102276799A (en) | 2011-12-14 |
| CN102276799B true CN102276799B (en) | 2014-05-28 |
Family
ID=45102571
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201010196323.4A Expired - Fee Related CN102276799B (en) | 2010-06-09 | 2010-06-09 | Quinoxaline unit-containing fluorene copolymer, and preparation method and application thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN102276799B (en) |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN104177590A (en) * | 2013-05-28 | 2014-12-03 | 海洋王照明科技股份有限公司 | Polymer of fluorene/oxadiazole group, preparation method and organic electroluminescent device thereof |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1337987A (en) * | 1999-02-04 | 2002-02-27 | 陶氏化学公司 | Fluorene copolymers and devices made therefrom |
| CN1487937A (en) * | 2001-01-24 | 2004-04-07 | ʾ | Monomers used to make polymers used in optical devices |
| CN1835986A (en) * | 2003-08-12 | 2006-09-20 | 科文有机半导体有限公司 | Conjugated polymers containing dihydrophenanthrene units and use thereof |
| CN101389634A (en) * | 2006-02-22 | 2009-03-18 | 住友化学株式会社 | Fluorine-containing compound, method for producing same, fluorine-containing polymer, organic thin film, and organic thin film element |
-
2010
- 2010-06-09 CN CN201010196323.4A patent/CN102276799B/en not_active Expired - Fee Related
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1337987A (en) * | 1999-02-04 | 2002-02-27 | 陶氏化学公司 | Fluorene copolymers and devices made therefrom |
| CN1487937A (en) * | 2001-01-24 | 2004-04-07 | ʾ | Monomers used to make polymers used in optical devices |
| CN1835986A (en) * | 2003-08-12 | 2006-09-20 | 科文有机半导体有限公司 | Conjugated polymers containing dihydrophenanthrene units and use thereof |
| CN101389634A (en) * | 2006-02-22 | 2009-03-18 | 住友化学株式会社 | Fluorine-containing compound, method for producing same, fluorine-containing polymer, organic thin film, and organic thin film element |
Non-Patent Citations (2)
| Title |
|---|
| "Synthesis and Properties of Fluorene-Based Polyheteroarylenes for Photovoltaic Devices";RAJA SHAHID ASHRAF, et al;《《Journal of Polymer Science: Part A: Polymer Chemistry》》;20061107;第44卷(第24期);第6952-3961页 * |
| RAJA SHAHID ASHRAF, et al."Synthesis and Properties of Fluorene-Based Polyheteroarylenes for Photovoltaic Devices".《《Journal of Polymer Science: Part A: Polymer Chemistry》》.2006,第44卷(第24期),第6952-3961页. |
Also Published As
| Publication number | Publication date |
|---|---|
| CN102276799A (en) | 2011-12-14 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2562197B1 (en) | Copolymer comprising anthracene and benzoselenadiazole, preparing method and uses thereof | |
| CN102295749B (en) | Fluorine copolymer containing thiophene and pyrrole-pyrrole units and preparation method as well as application thereof | |
| CN101787111B (en) | Fluorene copolymer and preparation method, application and polymer solar cell device thereof | |
| EP2530084B1 (en) | Copolymer containing fluorenylporphyrin-anthracene, preparation method and use thereof | |
| EP2927259B1 (en) | Benzodithiophene based copolymer containing thiophene pyrroledione units and preparing method and applications thereof | |
| CN102753599B (en) | Fluorene copolymer, method for preparation and use thereof | |
| CN102276802B (en) | Thiophene-quinoxaline unit containing fluorene copolymer as well as preparation method and application thereof | |
| CN102260191A (en) | Quinonoid fluorine semiconductor material as well as preparation method thereof and application thereof | |
| CN102453228B (en) | Organic semiconductor material containing fluorene, anthracene and benzodithiophene units, preparation method, and application thereof | |
| CN102206329B (en) | Dithiophene silole-quinoxalines conjugated polymer, and its preparation method and application | |
| CN102234365B (en) | Anthracene and benzothiadiazole copolymer, and preparation method and application thereof | |
| CN102477143B (en) | Fluorene-containing organic semiconductor material, and preparation method and application thereof | |
| CN102146151A (en) | Perylene tetracarboxylic dianiline conjugated polymer and preparation method and application thereof | |
| CN102146153A (en) | Perylene tetracarboxylic diimide-carbazole-dithienyldiazosulfide conjugated polymer as well as preparation method and application thereof | |
| CN102276799B (en) | Quinoxaline unit-containing fluorene copolymer, and preparation method and application thereof | |
| US20130172508A1 (en) | Fluorene-containing organic semiconductor material, preparation method and use thereof | |
| US20130225782A1 (en) | Organic semiconductor material, preparation methods and uses thereof | |
| CN102134307B (en) | Perylene diimide-fluorene-thiophene and (3, 4-b) pyrazine conjugated polymer and preparation method and application thereof | |
| US8481754B2 (en) | Quinoid silafluorene organic semiconductor material, preparation method and use thereof | |
| WO2011143806A1 (en) | Porphyrin copolymer containing quinoxaline unit, preparation method and uses thereof | |
| CN102260370B (en) | Benzothiophene unit-containing perylene diimide copolymer, and preparation method and application thereof | |
| CN102206327B (en) | Thieno (3,4-b) pyrazine contained fluorene copolymer, preparation method and application thereof | |
| CN102234366B (en) | Thiophene-containing perylene tetracarboxylic diimide copolymer, and preparation method and application thereof | |
| CN102206328B (en) | Porphyrin copolymer containing benzothiadiazole unit, its preparation method and application | |
| CN102206330B (en) | Bi-thiophene silole-containing conjugated polymer and preparation method and application thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| CF01 | Termination of patent right due to non-payment of annual fee | ||
| CF01 | Termination of patent right due to non-payment of annual fee |
Granted publication date: 20140528 Termination date: 20190609 |