CN102161980B - Method for culturing induced pluripotent stem cells by using human mesenchymal stem cells as trophoblast - Google Patents
Method for culturing induced pluripotent stem cells by using human mesenchymal stem cells as trophoblast Download PDFInfo
- Publication number
- CN102161980B CN102161980B CN 201110036898 CN201110036898A CN102161980B CN 102161980 B CN102161980 B CN 102161980B CN 201110036898 CN201110036898 CN 201110036898 CN 201110036898 A CN201110036898 A CN 201110036898A CN 102161980 B CN102161980 B CN 102161980B
- Authority
- CN
- China
- Prior art keywords
- stem cells
- human
- cells
- mesenchymal stem
- ipscs
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
Images
Landscapes
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
Abstract
Description
技术领域 technical field
本发明属于生物技术领域,涉及一种以人骨髓间充质干细胞作为滋养层培养诱导多能干细胞的方法。 The invention belongs to the field of biotechnology, and relates to a method for culturing induced pluripotent stem cells using human bone marrow mesenchymal stem cells as a trophoblast.
背景技术 Background technique
诱导多能干细胞(Induced Pluripotent Stem Cells, iPSCs)是近年来干细胞研究领域中具有里程碑意义的突破。其是通过异位表达几个与维持胚胎干细胞(Embryonic Stem Cells,ESCs)多潜能性相关的关键性核转录因子,使体细胞发生重编程,得到的一种类似ESCs的多潜能细胞,可以在体内外分化为三胚层的所有组织细胞。由体细胞重编程而来的iPSCs为获得与患者自身遗传背景一致的多能干细胞增加了一个新途径,且不再使用人类早期胚胎和卵母细胞,故伦理学的争论将随之平息,核移植技术缺乏卵母细胞和技术繁杂的窘境也得到了合理的解决。是组织工程和再生医学、疾病模型、药物筛选的理想种子细胞,因此具有广阔的临床应用背景。目前,研究者已在体外将人iPSCs向各种组织细胞诱导分化,如造血干/祖细胞、红细胞、T细胞、B细胞、心肌细胞、胰岛β细胞、肝脏细胞等;建立了多种遗传性疾病人iPSCs疾病模型,用于发病机制的研究,并在动物模型上尝试通过对有遗传缺陷的iPSCs进行基因改造,治疗遗传性疾病;建立多种肿瘤iPSCs疾病模型,用于发病机制的研究和药物筛选。 Induced pluripotent stem cells (Induced Pluripotent Stem Cells, iPSCs) are a milestone breakthrough in the field of stem cell research in recent years. It is a pluripotent cell similar to ESCs obtained by ectopically expressing several key nuclear transcription factors related to the maintenance of pluripotency of embryonic stem cells (Embryonic Stem Cells, ESCs). All tissue cells that differentiate into the three germ layers in vitro and in vivo. iPSCs reprogrammed from somatic cells adds a new way to obtain pluripotent stem cells consistent with the patient's own genetic background, and no longer uses human early embryos and oocytes, so the ethical debate will subside. The dilemma of lack of oocytes and complex techniques in transplantation techniques has also been reasonably resolved. It is an ideal seed cell for tissue engineering and regenerative medicine, disease models, and drug screening, so it has a broad clinical application background. At present, researchers have induced the differentiation of human iPSCs into various tissue cells in vitro, such as hematopoietic stem/progenitor cells, red blood cells, T cells, B cells, cardiomyocytes, pancreatic islet β cells, liver cells, etc.; Disease human iPSCs disease model for the study of pathogenesis, and try to treat genetic diseases by genetically modifying iPSCs with genetic defects in animal models; establish a variety of tumor iPSCs disease models for the study of pathogenesis and drug screening.
人iPSCs的体外培养需要细胞与细胞的相互作用、各种细胞因子、基质等来维持其自我更新和多潜能性。其经典培养模式是以小鼠胚胎成纤维细胞(Mouse Embryonic fibroblasts,MEF)为滋养层,用含bFGF、血清替代物(serum replacement,SR)的培养液培养。但是,人iPSCs进入临床应用前,去除培养体系中异源蛋白、细胞污染是必须解决的一个问题。因此,自人iPSCs出现以后,研究者就开始尝试改善其培养体系,在维持其自我更新和多潜能性的同时,减少异源蛋白、细胞的污染。如2010年,Christian等用永生化的人皮肤成纤维细胞作为滋养层,培养人iPSCs,发现这种人源化的滋养层可产生和维持人iPSCs的培养,但其培养时间较短(报道的培养代数为7代);同年,Kristiina等用一种成分明确的无异源蛋白的RegES培养液培养人iPSCs,发现其能长期(>80代)维持iPSCs的自我更新和多潜能性,但是并没有证明在重编程过程中,该培养液可使人体细胞重编程为iPSCs。Ruth等用另一种成分明确的mTeSRTM培养液悬浮培养人iPSCs,发现培养17代后,仍可维持其自我更新和多潜能性,但是该培养体系价格昂贵,且同样没有证明在重编程过程中,该培养液可使人体细胞重编程为iPSCs。因此,进一步开发研究人iPSCs人源化的培养体系,包括重编程过程和维持培养,以及对iPSCs生物学特性的影响,是人iPSCs走向临床应用的重要前提。 The in vitro culture of human iPSCs requires cell-cell interactions, various cytokines, substrates, etc. to maintain their self-renewal and pluripotency. The classic culture mode is to use mouse embryonic fibroblasts (Mouse Embryonic fibroblasts, MEF) as the trophoblast, cultured in the culture medium containing bFGF and serum replacement (serum replacement, SR). However, before human iPSCs enter clinical application, the removal of heterologous proteins and cell contamination in the culture system is a problem that must be solved. Therefore, since the emergence of human iPSCs, researchers have begun to try to improve their culture system, while maintaining their self-renewal and pluripotency, while reducing the contamination of heterologous proteins and cells. For example, in 2010, Christian et al. used immortalized human skin fibroblasts as a trophoblast to culture human iPSCs, and found that this humanized trophoblast could produce and maintain the culture of human iPSCs, but the culture time was short (reported In the same year, Kristiina et al. cultured human iPSCs with a well-defined RegES medium without heterologous proteins, and found that it could maintain the self-renewal and pluripotency of iPSCs for a long time (>80 passages), but it did not It has not been demonstrated that the medium can reprogram human cells into iPSCs during the reprogramming process. Ruth et al. used another well-defined mTeSRTM medium to culture human iPSCs in suspension, and found that after 17 generations of culture, they could still maintain their self-renewal and pluripotency, but the culture system was expensive, and it also did not prove that it was in the process of reprogramming. , the culture medium can reprogram human cells into iPSCs. Therefore, further development and study of the humanized culture system of human iPSCs, including the reprogramming process and maintenance culture, as well as the effect on the biological characteristics of iPSCs, is an important prerequisite for the clinical application of human iPSCs.
发明内容 Contents of the invention
本发明的目的是提供一种用人骨髓间充质干细胞(Mesenchymal Stem Cells,MSCs)作为滋养层培养人诱导多能干细胞(iPSCs)的方法,通过以下技术方案实现: The purpose of the present invention is to provide a method for culturing human induced pluripotent stem cells (iPSCs) using human bone marrow mesenchymal stem cells (Mesenchymal Stem Cells, MSCs) as a trophoblast, which is achieved through the following technical solutions:
(1)滋养层细胞的准备:用传代培养、低温冻存复苏后培养的第三~第五代的人骨髓间充质干细胞作为滋养层细胞。人骨髓间充质干细胞采用目前常用的Ficoll-paque密度梯度离心结合贴壁筛选分离培养获得。作为滋养层时,细胞接种到用0.1%gelatin处理后的六孔板上,细胞融合度达80%-90%(约2×104/cm2),并用10ug/ml的丝裂霉素C处理2小时灭活,灭活后的细胞24小时内使用; (1) Preparation of trophoblast cells: The third to fifth passage human bone marrow mesenchymal stem cells cultured after subculture, cryopreservation and recovery were used as trophoblast cells. Human bone marrow mesenchymal stem cells were isolated and cultured by the commonly used Ficoll-paque density gradient centrifugation combined with adherence screening. When used as a feeder layer, the cells were seeded on a six-well plate treated with 0.1% gelatin, and the cell confluence reached 80%-90% (about 2×10 4 /cm 2 ), and treated with 10ug/ml mitomycin C Treat for 2 hours to inactivate, and use the inactivated cells within 24 hours;
(2)人iPSCs的获得:取2-3岁儿童包皮成纤维细胞,用携带有Klf4、Sox2、Oct4和c-Myc转录因子的逆转录病毒转染两次。第二天在培养体系中加维生素C 25ug/ml,第五天加入VPA2 μM(共使用5天),第六天将转染后的成纤维细胞接种到滋养层细胞上,第七天,换用含bFGF、替代血清的人ESCs培养液。随后每天换液,直到出现类ESCs的克隆,挑选、扩增培养; (2) Obtaining human iPSCs: Foreskin fibroblasts from 2-3-year-old children were taken and transfected twice with retroviruses carrying Klf4, Sox2, Oct4 and c-Myc transcription factors. Add vitamin C 25ug/ml to the culture system on the second day, add VPA2 μM on the fifth day (used for 5 days in total), inoculate the transfected fibroblasts on the trophoblast cells on the sixth day, and replace the Human ESCs culture medium containing bFGF and replacing serum was used. Then change the medium every day until ESCs-like clones appear, select, expand and culture;
(3)人iPSCs的扩增培养:显微镜下挑选类ESCs的克隆,用IV型胶原酶消化5-10分钟后,轻轻吹散成小团块,接种到准备好的滋养层细胞上。每天换液。每7-8天传代一次。扩增后的细胞用机械法传代,即用5ml的玻璃滴管将细胞从滋养层上轻轻刮下,随后轻轻吹散成小团块,接种到准备好的滋养层细胞上; (3) Expansion culture of human iPSCs: ESCs-like clones were selected under a microscope, digested with type IV collagenase for 5-10 minutes, blown gently into small clumps, and inoculated on the prepared trophoblast cells. Change fluid daily. Subculture every 7-8 days. The amplified cells are passaged mechanically, that is, the cells are gently scraped off the trophoblast with a 5ml glass dropper, and then gently blown into small clumps, and inoculated on the prepared trophoblast cells;
(4)人iPSCs的生物学特性和多潜能性的维持:在以人骨髓间充质干细胞为滋养层上扩增培养14代以后,用RT-PCR法鉴定扩增培养后人iPSCs的ESCs特异基因表达情况;同时用EB形成法和免疫缺陷小鼠畸胎瘤形成法检测其在体内外是否可向三胚层分化。 (4) Biological characteristics and maintenance of pluripotency of human iPSCs: after 14 passages of expansion and culture on human bone marrow mesenchymal stem cells, RT-PCR method was used to identify the ESCs specificity of human iPSCs after expansion and culture. Gene expression; at the same time, the EB formation method and the immunodeficiency mouse teratoma formation method were used to detect whether it could differentiate into three germ layers in vitro and in vivo.
本发明提供了一种用人源化细胞即人骨髓间充质干细胞作为滋养层获得培养人iPSCs的方法,这种人源化的滋养层细胞可长期维持人iPSCs的生物学特征和多潜能性;由此获得的人iPSCs没有异源细胞的污染,为其临床应用提供了可能性。本发明使用人骨髓间充质干细胞代替传统方法中用小鼠胚胎成纤维细胞作为滋养层细胞,培养人诱导多能干细胞,减少了培养体系中异源细胞的污染,并证明了该培养体系可使人诱导多能干细胞在体外扩增(见实施例4),且维持了其生物学特性和多潜能性(见实施例5),为人诱导多能干细胞进入临床应用提供了可能。 The present invention provides a method for obtaining and culturing human iPSCs using humanized cells, that is, human bone marrow mesenchymal stem cells, as a trophoblast. The humanized trophoblast cells can maintain the biological characteristics and pluripotency of human iPSCs for a long time; The human iPSCs thus obtained are free from contamination by heterologous cells, which opens up the possibility of their clinical application. The present invention uses human bone marrow mesenchymal stem cells instead of using mouse embryonic fibroblasts as trophoblast cells in the traditional method to cultivate human induced pluripotent stem cells, which reduces the contamination of heterologous cells in the culture system, and proves that the culture system can Human induced pluripotent stem cells are expanded in vitro (see Example 4), and their biological characteristics and pluripotency are maintained (see Example 5), which provides the possibility for human induced pluripotent stem cells to enter clinical applications.
附图说明 Description of drawings
图1为技术路线图。 Figure 1 is the technology roadmap.
图2为第4代人骨髓间充质干细胞。A为灭活前;B为丝裂霉素C灭活后。 Figure 2 is the 4th passage of human bone marrow mesenchymal stem cells. A is before inactivation; B is after mitomycin C inactivation.
图3为用人骨髓间充质干细胞作为滋养层获得的人iPSCs。A为成纤维细胞转染21-25天后,出现的肉芽肿样克隆(标尺为100μm),B为扩增培养后形成的iPSCs克隆(标尺为100μm),C为克隆局部细胞形态(标尺为20μm)。 Figure 3 shows human iPSCs obtained using human bone marrow mesenchymal stem cells as a feeder layer. A is the granuloma-like colony that appeared 21-25 days after transfection of fibroblasts (the scale bar is 100 μm), B is the iPSCs clone formed after expansion and culture (the scale bar is 100 μm), and C is the local cell morphology of the clone (the scale bar is 20 μm ).
图4为第8-11代人iPSCs在以第4代人骨髓间充质干细胞为滋养层的培养体系中的生长曲线。
Figure 4 is the growth curve of human iPSCs at passage 8-11 in a culture system using human bone marrow mesenchymal stem cells at
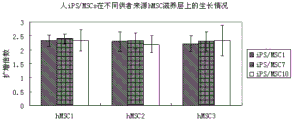
图5为第9、10代人iPSCs在不同供者来源第4代人骨髓间充质干细胞为滋养层的培养体系中的扩增倍数(hMSC1和hMSC2为女性供者来源,hMSC3为男性供者来源)。 Figure 5 shows the expansion multiples of human iPSCs at the 9th and 10th passages in the culture system of the 4th passage human bone marrow mesenchymal stem cells from different donor sources (hMSC1 and hMSC2 are from female donors, and hMSC3 is from male donors source).
图6为RT-PCR检测人iPS/MSC7在人骨髓间充质干细胞为滋养层的培养体系中扩增培养14代以后人ESCs特异基因的表达情况。 Figure 6 shows the expression of human ESCs-specific genes detected by RT-PCR after human iPS/MSC7 was amplified and cultured for 14 generations in a culture system in which human bone marrow mesenchymal stem cells were used as trophoblast.
图7为人iPS/MSC7在人骨髓间充质干细胞为滋养层的培养体系中扩增培养14代以后在体外形成EB向三胚层分化情况。A为EB培养8天后形成的悬浮EB(标尺为50μm);B-D为培养14天后EB贴壁分化成的各种类型细胞(标尺为100μm);E为PT-PCT检测EB分化后多潜能基因(SOX2和OCT4)和三胚层的标记基因(FOXA2和AFP为内胚层标记基因;MSX1和BRACHYURY为中胚层标记基因;MAP2和PAX6为外胚层标记基因)表达情况(U表示未分化,D表示分化后)。 Figure 7 shows the differentiation of human iPS/MSC7 from EB to three germ layers in vitro after being expanded and cultured for 14 passages in a culture system in which human bone marrow mesenchymal stem cells are trophoblasts. A is the suspension EB formed after EB cultured for 8 days (the scale is 50 μm); B-D is the various types of cells differentiated from the adherent EB after 14 days of culture (the scale is 100 μm); E is the PT-PCT detection of pluripotency genes after EB differentiation ( SOX2 and OCT4) and three germ layer marker genes (FOXA2 and AFP are endoderm marker genes; MSX1 and BRACHYURY are mesoderm marker genes; MAP2 and PAX6 are ectoderm marker genes) expression (U means undifferentiated, D means differentiated ).
图8为人iPS/MSC7在人骨髓间充质干细胞为滋养层的培养体系中扩增培养14代以后在NOD-SCID小鼠体内形成畸胎瘤情况。A为长出畸胎瘤的小鼠;B为畸胎瘤内形成的腺样组织(HE染色,标尺为20μm);C为畸胎瘤内形成的肌肉样组织(HE染色,标尺为20μm)。 Figure 8 shows the formation of teratomas in NOD-SCID mice after human iPS/MSC7 was expanded and cultured for 14 passages in a culture system in which human bone marrow mesenchymal stem cells were used as trophoblast. A is the mouse with teratoma; B is the adenoid tissue formed in the teratoma (HE staining, the scale bar is 20 μm); C is the muscle-like tissue formed in the teratoma (HE staining, the scale bar is 20 μm) .
具体实施方式 Detailed ways
本发明结合附图和实施例作进一步的说明。 The present invention will be further described in conjunction with drawings and embodiments.
实施例1:人骨髓间充质干细胞分离培养、冻存复苏 Example 1: Isolation and culture of human bone marrow mesenchymal stem cells, cryopreservation and recovery
(1) 人骨髓间充质干细胞分离培养 (1) Isolation and culture of human bone marrow mesenchymal stem cells
无菌条件下采集3份健康供者的骨髓,肝素抗凝,用Ficoll-paque(比重1.077)密度梯度离心收集单个核细胞,以5×105个细胞/cm2密度接种,培养液为含10%(V/V)胎牛血清和1%谷氨酰胺的低糖DMEM(LG-DMEM)培养液,置于370C、5%CO2培养箱培养。24小时后更换培养液,弃去非贴壁细胞,可见有梭形贴壁细胞。之后每3天换液1次,14天左右原代细胞贴壁融合达90%-95%,排列有明显方向性,成漩涡状、网状、辐射状(见图2A)。用0.25%胰蛋白酶-1 mmol EDTA消化,按1:3比例分瓶传代培养,并标记为P1细胞。传代培养过程中每3天换液一次,细胞达90%融合后消化传代,并标记为P2,以此类推。 The bone marrow of 3 healthy donors was collected under sterile conditions, anticoagulated with heparin, mononuclear cells were collected by density gradient centrifugation with Ficoll-paque (specific gravity 1.077), seeded at a density of 5×10 5 cells/cm 2 , and the culture medium contained 10% (V/V) fetal bovine serum and 1% glutamine low-glucose DMEM (LG-DMEM) culture solution, placed in a 37 0 C, 5% CO2 incubator for culture. After 24 hours, the culture medium was replaced, the non-adherent cells were discarded, and there were spindle-shaped adherent cells. After that, the medium was changed every 3 days, and the adhesion and fusion of the primary cells reached 90%-95% in about 14 days. They were digested with 0.25% trypsin-1 mmol EDTA, subcultured in bottles at a ratio of 1:3, and labeled as P1 cells. During the subculture process, the medium was changed every 3 days. After the cells reached 90% confluence, they were digested and passaged, and marked as P2, and so on.
(2)人骨髓间充质干细胞的冻存和复苏 (2) Cryopreservation and recovery of human bone marrow mesenchymal stem cells
冻存时,将处于对数生长期的间充质干细胞消化后,用冻存保护液(60% LG-DMEM、30% 胎牛血清、10% DMSO)重悬细胞,细胞终浓度为5×106/ml,分转入冻存管内,标记上细胞名称、代数、冻存时间和冻存者。立即将冻存管放入异丙醇冻存盒后,放入-800C,过夜后移入-1960C液氮罐保存。细胞复苏时,从液氮中取出细胞放入370C恒温水浴箱快速解冻,待冻存液解冻至小冰晶后,移入超净台。无菌条件下,将细胞悬液移入15ml离心管,逐滴加入预冷的培养液10ml,200g离心5min后,弃去上清。将细胞团块用适量预热培养液重悬培养。复苏存活细胞接种6h后完全贴壁,细胞形态逐渐变成梭形。细胞经过1-2天休眠期后开始迅速增殖。复苏后的细胞形态与冻存前基本一致。 During cryopreservation, digest the mesenchymal stem cells in the logarithmic growth phase, and resuspend the cells in cryopreservation solution (60% LG-DMEM, 30% fetal bovine serum, 10% DMSO), and the final concentration of cells is 5× 10 6 /ml, divided into cryopreservation tubes, marked with the cell name, generation number, cryopreservation time and cryopreserver. Immediately put the cryopreservation tube into the isopropanol freezing box, put it at -80 0 C, and transfer it to a -196 0 C liquid nitrogen tank for storage overnight. When the cells were resuscitated, the cells were taken out of the liquid nitrogen and placed in a 37 0 C constant temperature water bath to quickly thaw. Under sterile conditions, transfer the cell suspension into a 15ml centrifuge tube, add 10ml of pre-cooled culture solution dropwise, centrifuge at 200g for 5min, and discard the supernatant. Resuspend the cell mass with an appropriate amount of preheated culture medium. The revived surviving cells were completely attached to the wall 6 hours after inoculation, and the cell shape gradually became spindle-shaped. Cells began to proliferate rapidly after a rest period of 1-2 days. The cell morphology after recovery was basically the same as that before cryopreservation.
实施例2:人骨髓间充质干细胞作为滋养层细胞的准备 Example 2: Preparation of human bone marrow mesenchymal stem cells as trophoblast cells
取第3-5代人骨髓间充质干细胞作为滋养层的细胞。将6孔板预先用0.1%gelatin在370C孵育1h到过夜。人骨髓间充质干细胞以2×104/cm2密度接种到gelatin处理后的六孔板上,使第二天细胞融合度达80%-90%。用10ug/ml的丝裂霉素C处理2小时灭活,灭活后的细胞形态与灭活前基本一致(见图2)。灭活后的细胞24小时内使用。 Human bone marrow mesenchymal stem cells of the 3rd to 5th generation were used as the trophoblast cells. The 6-well plate was pre-incubated with 0.1% gelatin at 37 ° C for 1h to overnight. Human bone marrow mesenchymal stem cells were seeded on gelatin-treated six-well plates at a density of 2×10 4 /cm 2 , so that the cell confluency reached 80%-90% the next day. Treated with 10ug/ml mitomycin C for 2 hours to inactivate, the cell morphology after inactivation was basically the same as that before inactivation (see Figure 2). The inactivated cells were used within 24 hours.
实施例3:以人骨髓间充质干细胞为滋养层的人iPSCs的诱导 Example 3: Induction of human iPSCs using human bone marrow mesenchymal stem cells as trophoblast
取第三代儿童(2-3岁)包皮成纤维细胞,用携带有Klf4、Sox2、Oct4和c-Myc转录因子的逆转录病毒转染两次。转染结束后在培养体系中加维生素C 25ug/ml直到挑选克隆,第五天加入VPA2 μM(共使用5天),第六天将转染后的成纤维细胞接种到以人骨髓间充质干细胞滋养层细胞上,第七天换用含8ng/ml的bFGF、1%谷氨酰胺、1%非必需氨基酸、100μM和20%替代血清的人ESCs培养液。随后每天换液,直到出现类ESCs的克隆(见图3A),边界清楚、细胞致密。显微镜下逐个挑选、扩增培养。技术路线见图1 Foreskin fibroblasts from children of the third generation (2-3 years old) were transfected twice with retrovirus carrying Klf4, Sox2, Oct4 and c-Myc transcription factors. After transfection, vitamin C 25ug/ml was added to the culture system until clones were selected, VPA2 μM was added on the fifth day (5 days in total), and the transfected fibroblasts were inoculated into human bone marrow mesenchyme on the sixth day On the stem cell trophoblast cells, on the seventh day, replace with human ESCs culture medium containing 8ng/ml bFGF, 1% glutamine, 1% non-essential amino acids, 100μM and 20% serum replacement. Then change the medium every day until ESCs-like clones appear (see Figure 3A) with clear borders and dense cells. Select one by one under the microscope, amplify and cultivate. The technical roadmap is shown in Figure 1
实施例4:以人骨髓间充质干细胞为滋养层的人iPSCs的扩增培养 Example 4: Expansion and culture of human iPSCs using human bone marrow mesenchymal stem cells as trophoblast
(1)人iPSCs在以人骨髓间充质干细胞为滋养层的培养体系中扩增培养后的细胞形态 (1) Cell morphology of human iPSCs expanded and cultured in a culture system using human bone marrow mesenchymal stem cells as a feeder layer
显微镜下挑选类ESCs的克隆,用1mg/ml的IV型胶原酶消化5-10分钟后,轻轻吹散成小团块,接种到准备好的滋养层细胞上,用人ESCs培养液,在370C、5%CO2中培养。每天换液。iPSCs每7-8天传代一次。扩增后的细胞用机械法传代,即用5ml的玻璃滴管将细胞从滋养层上轻轻刮下,随后轻轻吹散成小团块,以1:2-1:3接种到准备好的滋养层细胞上。人iPSCs在以人骨髓间充质干细胞为滋养层的培养体系中扩增后形成典型的人ESCs样克隆:边界清楚,细胞致密,大部分细胞为圆形,核仁明显,边界为类成纤维样细胞(见图3B、C)。 Select ESCs-like clones under a microscope, digest them with 1mg/ml type IV collagenase for 5-10 minutes, blow them gently into small clumps, inoculate them on the prepared trophoblast cells, and use human ESCs culture medium at 37 0° C, 5% CO2 culture. Change fluid daily. iPSCs were passaged every 7-8 days. The amplified cells are mechanically passaged, that is, the cells are gently scraped off the feeder layer with a 5ml glass dropper, and then gently blown into small clumps, and inoculated at a ratio of 1:2-1:3 until ready on the trophoblast cells. Human iPSCs were expanded in a culture system using human bone marrow mesenchymal stem cells as a feeder layer to form typical human ESCs-like clones: clear boundaries, dense cells, most of the cells are round, with obvious nucleoli, and the border is fibroblast-like Like cells (see Figure 3B, C).
(2)人iPSCs在以人骨髓间充质干细胞为滋养层的培养体系中的生长动力学 (2) Growth kinetics of human iPSCs in a culture system using human bone marrow mesenchymal stem cells as feeder layer
将第8代的三株人iPSCs(iPS/MSC1、iPS/MSC7和iPS/MSC10)以5×104细胞/孔接种(以小团块接种)到铺有人骨髓间充质干细胞为滋养层的12孔板中,设三个复孔。培养7天后,将人iPSCs从滋养层上刮下,取部分计数,计算扩增倍数;同时取其中部分接种到新的铺有人骨髓间充质干细胞为滋养层的12孔板中,设三个复孔,继续培养7天,计数并计算扩增倍数。以此类推,共培养三代,计算扩增倍数。人iPSCs在以人骨髓间充质干细胞为滋养层的培养体系中可持续扩增,平均每代扩增2.32±0.05倍(见图4)。 Three lines of human iPSCs (iPS/MSC1, iPS/MSC7 and iPS/MSC10) at passage 8 were inoculated at 5×10 4 cells/well (inoculated in small clumps) into a culture medium lined with human bone marrow mesenchymal stem cells as feeder layer. In the 12-well plate, three replicate wells were set. After 7 days of culture, human iPSCs were scraped off from the trophoblast, a part was counted, and the expansion factor was calculated; at the same time, part of it was inoculated into a new 12-well plate covered with human bone marrow mesenchymal stem cells as a trophoblast, and three Duplicate wells were cultured for 7 days, counted and calculated the amplification factor. By analogy, three generations were co-cultured, and the amplification factor was calculated. Human iPSCs can be continuously expanded in a culture system using human bone marrow mesenchymal stem cells as a feeder layer, with an average expansion of 2.32±0.05 times per generation (see Figure 4).
(3)人iPSCs在不同供者来源的人骨髓间充质干细胞为滋养层的培养体系中的生长情况 (3) The growth of human iPSCs in the culture system of human bone marrow mesenchymal stem cells from different donors as trophoblast
将第9、10代的三株人iPSCs(iPS/MSC1、iPS/MSC7和iPS/MSC10)以5×104细胞/孔接种(以小团块接种)到铺有不同供者来源的人骨髓间充质干细胞(hMSC1、hMSC2、hMSC3,其中hMSC1和hMSC2为女性供者,hMSC3为男性供者)为滋养层的12孔板中,设三个复孔。培养7天后,将人iPSCs从滋养层上刮下,取部分计数,计算扩增倍数。不同供者来源的骨髓间充质干细胞为滋养层的培养体系对人iPSCs细胞的扩增没有显著差异(见图5)。
Three lines of human iPSCs (iPS/MSC1, iPS/MSC7 and iPS/MSC10) at
实施例5:人iPSCs在人骨髓间充质干细胞为滋养层的培养体系中扩增培养14代以后生物学特性和多潜能性的维持情况 Example 5: Maintenance of biological characteristics and pluripotency of human iPSCs after 14 generations of expansion and culture in a culture system in which human bone marrow mesenchymal stem cells are used as feeder layer
(1)人iPSCs表达人ESCs特异基因情况 (1) Human iPSCs express human ESCs-specific genes
收集在人骨髓间充质干细胞为滋养层的培养体系中扩增培养14代以后的人iPSC/MSC7细胞团块,用TRIZOL提取总RNA,随后进行RT-PCR,检测人ESCs特异基因的表达情况:包括内源性KLF4、SOX2、c-Myc、OCT4以及NANOG、DNMT3B、DPPA4、hTERT、NODAL、REX13、TDGF1等。同时以H1人ESCs和儿童包皮成纤维细胞为对照。检测结果显示这些基因在人iPSCs中的表达水平类似于H1人ESCs。 The human iPSC/MSC7 cell aggregates expanded and cultured in the culture system of human bone marrow mesenchymal stem cells for 14 passages were collected, and the total RNA was extracted with TRIZOL, followed by RT-PCR to detect the expression of human ESCs-specific genes : Including endogenous KLF4, SOX2, c-Myc, OCT4, NANOG, DNMT3B, DPPA4, hTERT, NODAL, REX13, TDGF1, etc. At the same time, H1 human ESCs and children's foreskin fibroblasts were used as controls. The detection results showed that the expression levels of these genes in human iPSCs were similar to H1 human ESCs.
(2)人iPSCs在体外形成EB向三胚层分化情况 (2) Human iPSCs formed EBs in vitro and differentiated into three germ layers
在人骨髓间充质干细胞为滋养层的培养体系中扩增培养14代以后的人iPSC/MSC7从滋养层上刮下,以细胞团块接种在超低黏附性6孔板中,培养液为不含bFGF的人ESCs培养液,在370C、5%CO2中培养,隔天换液;培养8天后,接种到0.1%gelatin处理后的6孔板中,继续用不含bFGF的人ESCs培养液,在370C、5%CO2中贴壁培养8天。实验结果显示扩增后的人iPSCs可在体外形成EB(见图7A),这些EB贴壁培养后,可分化成各种类型的细胞(见图7B-D)。收集这些分化的细胞,用RT-PCR检测三胚层标记性基因的表达情况,结果显示分化后的细胞三胚层标记基因(FOXA2和AFP为内胚层标记基因;MSX1和BRACHYURY为中胚层标记基因;MAP2和PAX6为外胚层标记基因)表达明显增加。表明在人骨髓间充质干细胞为滋养层的培养体系中扩增培养14代以后的人iPSCs仍维持了其多向分化潜能。 Human iPSC/MSC7 expanded and cultured for 14 passages in a culture system in which human bone marrow mesenchymal stem cells were used as a trophoblast were scraped off from the trophoblast, and inoculated in ultra-low adhesion 6-well plates as cell clumps, and the culture medium was Human ESCs culture medium without bFGF was cultured at 37 0 C, 5% CO 2 , and the medium was changed every other day; after 8 days of culture, inoculated into 6-well plates treated with 0.1% gelatin, and continued to be cultured with human ESCs without bFGF ESCs culture medium was adherently cultured at 37 ° C and 5% CO2 for 8 days. Experimental results show that the expanded human iPSCs can form EBs in vitro (see Figure 7A), and these EBs can differentiate into various types of cells after adherent culture (see Figure 7B-D). These differentiated cells were collected, and the expression of three germ layer marker genes were detected by RT-PCR. The results showed that the differentiated cells had three germ layer marker genes (FOXA2 and AFP were endoderm marker genes; MSX1 and BRACHYURY were mesoderm marker genes; MAP2 and PAX6 are ectoderm marker genes) expression was significantly increased. It shows that human iPSCs after 14 passages in the culture system in which human bone marrow mesenchymal stem cells are used as trophoblast still maintain their multilineage differentiation potential.
(3)人iPSCs在体内形成畸胎瘤向三胚层分化情况 (3) Human iPSCs formed teratomas in vivo and differentiated into three germ layers
将在人骨髓间充质干细胞为滋养层的培养体系中扩增培养14代以后的人iPSC/MSC7从滋养层上刮下,用不含钙镁离子的PBS缓冲液清洗两遍,用PBS将人iPSCs重悬,取200ul注射到NOD-SCID小鼠的后腿根部肌肉中,每个注射点的细胞数量为2-3×106。注射后观察成瘤情况。待畸胎瘤形成后,取出畸胎瘤行石蜡切片和HE染色, 观察其向三胚层分化情况。实验结果显示扩增后的人iPSCs可在免疫缺陷小鼠体内形成畸胎瘤(见图8A),HE染色后见瘤内有肌肉样、腺样组织形成(见图8B、C)。表示在人骨髓间充质干细胞为滋养层的培养体系中扩增培养14代以后的人iPSCs仍维持了其多向分化潜能。 The human iPSC/MSC7 expanded and cultured in the culture system of human bone marrow mesenchymal stem cells for 14 passages was scraped from the feeder layer, washed twice with PBS buffer without calcium and magnesium ions, and washed with PBS. Human iPSCs were resuspended, and 200ul was injected into the root muscle of the hind legs of NOD-SCID mice, and the number of cells at each injection point was 2-3×10 6 . Tumor formation was observed after injection. After the teratoma was formed, the teratoma was taken out for paraffin section and HE staining to observe its differentiation into three germ layers. The experimental results showed that the expanded human iPSCs could form teratomas in immunodeficient mice (see Figure 8A), and after HE staining, muscle-like and adenoid tissues were found in the tumor (see Figure 8B, C). It indicated that human iPSCs after 14 passages in the culture system in which human bone marrow mesenchymal stem cells were used as trophoblast still maintained their multidirectional differentiation potential.
Claims (2)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN 201110036898 CN102161980B (en) | 2011-02-12 | 2011-02-12 | Method for culturing induced pluripotent stem cells by using human mesenchymal stem cells as trophoblast |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN 201110036898 CN102161980B (en) | 2011-02-12 | 2011-02-12 | Method for culturing induced pluripotent stem cells by using human mesenchymal stem cells as trophoblast |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN102161980A CN102161980A (en) | 2011-08-24 |
| CN102161980B true CN102161980B (en) | 2013-07-31 |
Family
ID=44463434
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN 201110036898 Expired - Fee Related CN102161980B (en) | 2011-02-12 | 2011-02-12 | Method for culturing induced pluripotent stem cells by using human mesenchymal stem cells as trophoblast |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN102161980B (en) |
Families Citing this family (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102286532B (en) * | 2011-09-05 | 2012-10-17 | 浙江大学 | Method for obtaining inductive pluripotent stem cell |
| CN102965393B (en) * | 2012-11-06 | 2015-08-26 | 上海交通大学 | The preparation method and its usage of people's induced multi-potent stem cells |
| CN103589686B (en) * | 2013-11-06 | 2015-11-18 | 四川新生命干细胞科技股份有限公司 | A kind of human umbilical cord mesenchymal stem cells that uses is as the method for feeder layer cultivator induced multi-potent stem cells |
| CN104140951B (en) * | 2014-08-06 | 2016-08-24 | 深圳市科晖瑞生物医药有限公司 | A kind of method set up and cultivate people induced multi-potent stem cell |
| CN106381282B (en) * | 2016-10-12 | 2020-08-07 | 广东艾时代生物科技有限责任公司 | Passage method for induced pluripotent stem cells |
| CN107236703B (en) * | 2017-05-25 | 2021-06-01 | 佛山科学技术学院 | Feeder layer cell, culture method and application |
| CN109975264B (en) * | 2019-04-18 | 2022-04-08 | 安徽农业大学 | Method for researching gene function of lentivirus transfected blastocyst trophoblast cells |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101649305A (en) * | 2009-09-17 | 2010-02-17 | 中国医学科学院血液学研究所 | Method for amplifying megakaryocyte progenitor cell from human cord blood CD34<+> cell |
| WO2010099539A1 (en) * | 2009-02-27 | 2010-09-02 | Cellular Dynamics International, Inc. | Differentiation of pluripotent cells |
-
2011
- 2011-02-12 CN CN 201110036898 patent/CN102161980B/en not_active Expired - Fee Related
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2010099539A1 (en) * | 2009-02-27 | 2010-09-02 | Cellular Dynamics International, Inc. | Differentiation of pluripotent cells |
| CN101649305A (en) * | 2009-09-17 | 2010-02-17 | 中国医学科学院血液学研究所 | Method for amplifying megakaryocyte progenitor cell from human cord blood CD34<+> cell |
Non-Patent Citations (2)
| Title |
|---|
| Park Y.et al..The efficacy of human placenta as a source of the universal feeder in human and mouse pluripotent stem cell culture.《Cell Reprogram》.2010,第12卷(第3期),315-328. * |
| Raymond M.Anchan et al..Amniocytes can serve a dual function as a source of iPS cells and feeder layers.《Hum Mol Genet》.2010,962-974. * |
Also Published As
| Publication number | Publication date |
|---|---|
| CN102161980A (en) | 2011-08-24 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN102161980B (en) | Method for culturing induced pluripotent stem cells by using human mesenchymal stem cells as trophoblast | |
| Badenes et al. | Microcarrier-based platforms for in vitro expansion and differentiation of human pluripotent stem cells in bioreactor culture systems | |
| Eguchi | Cellular and molecular background of Wolffian lens regeneration | |
| CN110484506B (en) | Construction method and application of glioblastoma organoid model | |
| Ghasemi‐Dehkordi et al. | Comparison between the cultures of human induced pluripotent stem cells (hiPSCs) on feeder‐and serum‐free system (Matrigel matrix), MEF and HDF feeder cell lines | |
| CN114480259B (en) | Culture medium and induction method for inducing totipotent stem cells of mice | |
| WO2010143747A1 (en) | Method for production of artificial intestinal tract | |
| CN111235094B (en) | Method for differentiating human pluripotent stem cells into endoderm | |
| Kim et al. | Effective differentiation of induced pluripotent stem cells into dental cells | |
| CN105779395A (en) | Immortalized canine adipic mesenchymal stem cell line and constructing method thereof | |
| CN105420193B (en) | Differentiation medium and its use in preparing neural stem cells | |
| JP6421335B2 (en) | Method for culturing hepatic progenitor-like cells and culture thereof | |
| JP2019022509A (en) | Adhesion signature based method for isolating stem cells and cells derived from stem cells | |
| Vallabhaneni et al. | Suspension culture on microcarriers and as aggregates enables expansion and differentiation of pluripotent stem cells (PSCs) | |
| CN107937346B (en) | Method for culturing induced pluripotent stem cells by using human urine cells as feeder layer | |
| CN101497872A (en) | Method for inducing human embryo stem cell for directional differentiation into hepatocyte and special culture medium | |
| WO2010099643A1 (en) | Method for promoting somatic cells proliferation | |
| CN105154393A (en) | Method for differentiation of embryonic stem cells (ESC) into mesenchymal stem cells (MSC) | |
| Klimanskaya et al. | Embryonic stem cells | |
| Du et al. | Human iPS cell-derived fibroblast-like cells as feeder layers for iPS cell derivation and expansion | |
| CN102154195B (en) | In-vitro separation and preparation method of male germline stem cells of goat | |
| CN115261301A (en) | A kind of retinal pigment epithelial cell in vitro induction and culture method | |
| CN106337035A (en) | Technology for large-scale culture of fish fibroblast with application of bioreactors | |
| CN106367380B (en) | Cell co-culture method and the liver bud of liver bud can be prepared in vitro | |
| CN111321110A (en) | Method for differentiating human pluripotent stem cells into mesoderm |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| CF01 | Termination of patent right due to non-payment of annual fee |
Granted publication date: 20130731 Termination date: 20200212 |
|
| CF01 | Termination of patent right due to non-payment of annual fee |