CN102160855B - Dex-ibuprofen sustained release tablets and preparation method thereof - Google Patents
Dex-ibuprofen sustained release tablets and preparation method thereof Download PDFInfo
- Publication number
- CN102160855B CN102160855B CN2011100786082A CN201110078608A CN102160855B CN 102160855 B CN102160855 B CN 102160855B CN 2011100786082 A CN2011100786082 A CN 2011100786082A CN 201110078608 A CN201110078608 A CN 201110078608A CN 102160855 B CN102160855 B CN 102160855B
- Authority
- CN
- China
- Prior art keywords
- dex
- parts
- ibuprofen
- sustained release
- weight portion
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
- HEFNNWSXXWATRW-JTQLQIEISA-N dexibuprofen Chemical compound CC(C)CC1=CC=C([C@H](C)C(O)=O)C=C1 HEFNNWSXXWATRW-JTQLQIEISA-N 0.000 title claims abstract description 29
- 229960003428 dexibuprofen Drugs 0.000 title claims abstract description 20
- 239000007939 sustained release tablet Substances 0.000 title claims abstract description 19
- 238000002360 preparation method Methods 0.000 title claims abstract description 15
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 claims abstract description 22
- 235000019359 magnesium stearate Nutrition 0.000 claims abstract description 11
- 229920000168 Microcrystalline cellulose Polymers 0.000 claims abstract description 6
- 235000019813 microcrystalline cellulose Nutrition 0.000 claims abstract description 6
- 239000008108 microcrystalline cellulose Substances 0.000 claims abstract description 6
- 229940016286 microcrystalline cellulose Drugs 0.000 claims abstract description 6
- 239000001866 hydroxypropyl methyl cellulose Substances 0.000 claims abstract description 5
- 235000010979 hydroxypropyl methyl cellulose Nutrition 0.000 claims abstract description 5
- 229920003088 hydroxypropyl methyl cellulose Polymers 0.000 claims abstract description 5
- UFVKGYZPFZQRLF-UHFFFAOYSA-N hydroxypropyl methyl cellulose Chemical compound OC1C(O)C(OC)OC(CO)C1OC1C(O)C(O)C(OC2C(C(O)C(OC3C(C(O)C(O)C(CO)O3)O)C(CO)O2)O)C(CO)O1 UFVKGYZPFZQRLF-UHFFFAOYSA-N 0.000 claims abstract description 5
- 239000000203 mixture Substances 0.000 claims description 27
- 238000009472 formulation Methods 0.000 claims description 15
- 239000008187 granular material Substances 0.000 claims description 9
- 239000012729 immediate-release (IR) formulation Substances 0.000 claims description 9
- 239000001856 Ethyl cellulose Substances 0.000 claims description 6
- ZZSNKZQZMQGXPY-UHFFFAOYSA-N Ethyl cellulose Chemical compound CCOCC1OC(OC)C(OCC)C(OCC)C1OC1C(O)C(O)C(OC)C(CO)O1 ZZSNKZQZMQGXPY-UHFFFAOYSA-N 0.000 claims description 6
- 235000019325 ethyl cellulose Nutrition 0.000 claims description 6
- 229920001249 ethyl cellulose Polymers 0.000 claims description 6
- -1 hydroxypropyl Chemical group 0.000 claims description 6
- 239000003826 tablet Substances 0.000 claims description 6
- 206010013786 Dry skin Diseases 0.000 claims description 3
- 230000001476 alcoholic effect Effects 0.000 claims description 3
- 238000001035 drying Methods 0.000 claims description 3
- 239000007779 soft material Substances 0.000 claims description 3
- 238000000034 method Methods 0.000 claims description 2
- 239000003814 drug Substances 0.000 abstract description 5
- 229920002153 Hydroxypropyl cellulose Polymers 0.000 abstract description 3
- 229920002472 Starch Polymers 0.000 abstract description 3
- 239000001863 hydroxypropyl cellulose Substances 0.000 abstract description 3
- 235000010977 hydroxypropyl cellulose Nutrition 0.000 abstract description 3
- 238000001727 in vivo Methods 0.000 abstract description 3
- 239000008107 starch Substances 0.000 abstract description 3
- 235000019698 starch Nutrition 0.000 abstract description 3
- 238000013268 sustained release Methods 0.000 abstract 2
- 239000012730 sustained-release form Substances 0.000 abstract 2
- 210000001035 gastrointestinal tract Anatomy 0.000 abstract 1
- 230000007794 irritation Effects 0.000 abstract 1
- 239000002671 adjuvant Substances 0.000 description 4
- 208000002193 Pain Diseases 0.000 description 3
- 239000011230 binding agent Substances 0.000 description 3
- 230000001186 cumulative effect Effects 0.000 description 3
- 239000000463 material Substances 0.000 description 3
- HEFNNWSXXWATRW-UHFFFAOYSA-N Ibuprofen Chemical compound CC(C)CC1=CC=C(C(C)C(O)=O)C=C1 HEFNNWSXXWATRW-UHFFFAOYSA-N 0.000 description 2
- 238000010521 absorption reaction Methods 0.000 description 2
- 230000001154 acute effect Effects 0.000 description 2
- 239000002775 capsule Substances 0.000 description 2
- 239000003795 chemical substances by application Substances 0.000 description 2
- 201000010099 disease Diseases 0.000 description 2
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 2
- 229940079593 drug Drugs 0.000 description 2
- 230000000694 effects Effects 0.000 description 2
- 238000010579 first pass effect Methods 0.000 description 2
- 229960001680 ibuprofen Drugs 0.000 description 2
- 238000005057 refrigeration Methods 0.000 description 2
- 239000000829 suppository Substances 0.000 description 2
- 208000006820 Arthralgia Diseases 0.000 description 1
- 206010006811 Bursitis Diseases 0.000 description 1
- 206010013935 Dysmenorrhoea Diseases 0.000 description 1
- 201000005569 Gout Diseases 0.000 description 1
- 206010018634 Gouty Arthritis Diseases 0.000 description 1
- 206010019233 Headaches Diseases 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- 206010028391 Musculoskeletal Pain Diseases 0.000 description 1
- 208000000112 Myalgia Diseases 0.000 description 1
- 208000007613 Shoulder Pain Diseases 0.000 description 1
- 206010046914 Vaginal infection Diseases 0.000 description 1
- 201000008100 Vaginitis Diseases 0.000 description 1
- 235000009392 Vitis Nutrition 0.000 description 1
- 241000219095 Vitis Species 0.000 description 1
- 230000000202 analgesic effect Effects 0.000 description 1
- 230000003110 anti-inflammatory effect Effects 0.000 description 1
- 206010003246 arthritis Diseases 0.000 description 1
- 238000003556 assay Methods 0.000 description 1
- 230000003115 biocidal effect Effects 0.000 description 1
- 230000036760 body temperature Effects 0.000 description 1
- 230000002950 deficient Effects 0.000 description 1
- 239000002552 dosage form Substances 0.000 description 1
- 230000000857 drug effect Effects 0.000 description 1
- 230000002496 gastric effect Effects 0.000 description 1
- 231100000869 headache Toxicity 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- TUZRJGVLAFMQEK-LARVRRBISA-N hydron (2S)-2-[4-(2-methylpropyl)phenyl]propanoate (2S)-2-[4-(2-methylpropyl)phenyl]propanoic acid Chemical compound CC(C)CC1=CC=C([C@H](C)C(O)=O)C=C1.CC(C)CC1=CC=C([C@H](C)C(O)=O)C=C1 TUZRJGVLAFMQEK-LARVRRBISA-N 0.000 description 1
- 238000000338 in vitro Methods 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 239000011159 matrix material Substances 0.000 description 1
- 239000007896 modified release capsule Substances 0.000 description 1
- 201000008482 osteoarthritis Diseases 0.000 description 1
- 230000002688 persistence Effects 0.000 description 1
- 239000000546 pharmaceutical excipient Substances 0.000 description 1
- 150000003180 prostaglandins Chemical class 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 201000003068 rheumatic fever Diseases 0.000 description 1
- 206010039073 rheumatoid arthritis Diseases 0.000 description 1
- 238000012216 screening Methods 0.000 description 1
- 210000004872 soft tissue Anatomy 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 208000004371 toothache Diseases 0.000 description 1
Landscapes
- Medicinal Preparation (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
Abstract
The invention belongs to the technical field of pharmacy, and in particular relates to a prescription and preparation of dex-ibuprofen sustained release tablets. The dex-ibuprofen sustained release tablets are prepared by mixing a quick release part, a sustained release part and magnesium stearate, wherein the quick release part comprises the following components in part by weight: 40 to 80 parts of dex-ibuprofen, 31.0 to 240 parts of starch, 10.0 to 60 parts of hydroxypropylcellulose, and 1.0 to 5.0 parts of 3 mass percent of hydroxypropyl methylcellulose solution; the sustained release part comprises the following components in part by weight: 220 to 260 parts of dex-ibuprofen, 10 to 40 parts of microcrystalline cellulose, 40 to 130 parts of hydroxypropyl methyl cellulose and 2.0 to 8.0 parts of ethyecellulose; and the magnesium stearate accounts for 3.0 to 18.0 weight parts of the total weight. The dex-ibuprofen sustained release tablets prolong the release time of the medicine in vivo, are easily absorbed by a human body, reduce irritation to gastrointestinal tract and improve the bioavailability.
Description
Technical field
The invention belongs to the pharmaceutical technology field, be specifically related to a kind of prescription and preparation of Dex-ibuprofen sustained release tablets.
Background technology
Raw material: (S)-ibuprofen
Chemical name: S (+)-2 (4-isobutylphenyl) Propionic acid
English name: Dexibuprofen
Molecular formula: C
13H
18O
2
Molecular weight: 206.28
Structural formula:
(S)-ibuprofen is non-deep and remote body class antibiotic medicine, mainly is to suppress the synthetic of Cycloxygenase and prostaglandin and reach antiinflammatory, analgesic activity, and plays refrigeration function by the hypothalamus center of body temperature regulation.Has application value.
Multiplex in 1 clinically at present, alleviate the acute attack stage of the various chronic arthritiss such as rheumatoid arthritis, osteoarthritis, SpA, gouty arthritis, rheumatic arthritis or the arthralgia condition of illness of persistence, anosis effect because for the treatment of and the control course of disease; 2. treat non-arthrogenous various soft tissue rheumatism pain, such as shoulder pain, key vaginitis, bursitis, myalgia and the rear damaging pain of motion etc.; Acute light, moderate pain as; After the operation, after the wound, after old the damage, primary dysmenorrhea, toothache, headache etc.; 4. adult and child's heating there is refrigeration function.
At present, the (S)-ibuprofen of listing only has tablet, capsule and suppository, and the oral formulations such as tablet and capsule exist and absorb the shortcomings such as inhomogeneous, that first pass effect is strong, bioavailability is low, the decrement when being not easy to the special disease such as old people is taken.The subject matter of suppository is to use inconvenience, and its applicable crowd only is defined in pediatric patient.There is no any slow releasing preparation and be seen in report.
Summary of the invention
Dosage form for existing medicine absorbs the defectives such as inhomogeneous, that first pass effect is strong, bioavailability is low, the invention provides a kind of Dex-ibuprofen sustained release tablets, prolong drug release time in vivo, be conducive to absorption of human body, minimizing irritates gastrointestinal, improves bioavailability.
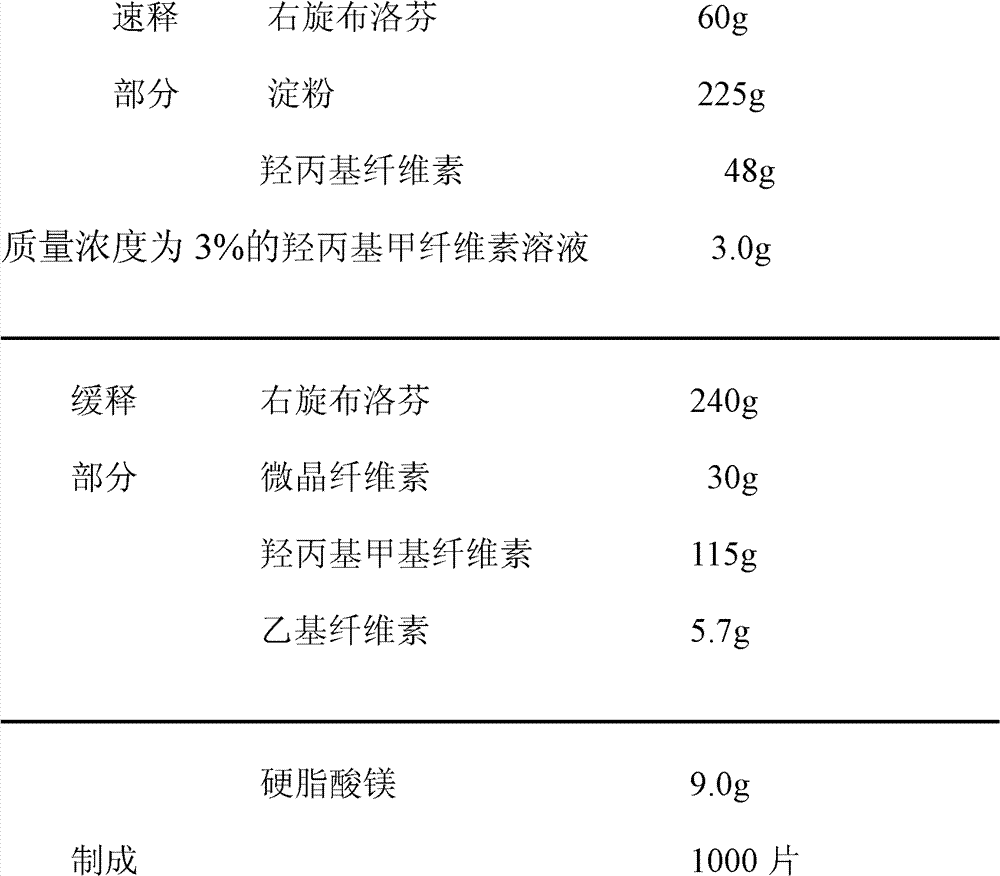
1, a kind of Dex-ibuprofen sustained release tablets of the present invention is comprised of immediate release section, slow-released part and magnesium stearate mixing;
The immediate release section prescription according to the weight portion formulation ratio that accounts for gross weight is:
The slow-released part prescription according to the weight portion formulation ratio that accounts for gross weight is:
The weight portion of the shared gross weight of described magnesium stearate is 3.0 parts-18.0 parts.
Dex-ibuprofen sustained release tablets optimum ratio of the present invention is as follows:
The immediate release section prescription according to the weight portion formulation ratio that accounts for gross weight is:
The slow-released part prescription according to the weight portion formulation ratio that accounts for gross weight is:
The weight portion of the shared gross weight of described magnesium stearate is 9.0 parts.
The method for preparing Dex-ibuprofen sustained release tablets of the present invention, step is:
(1) preparation of slow-released part
Get (S)-ibuprofen, microcrystalline Cellulose, hydroxypropyl emthylcellulose according to the described formulation ratio mixing of claim 1, sieve with 80 orders; Alcoholic solution with ethyl cellulose and hydroxypropyl methylcellulose mixtures is made soft material with said mixture; After secondary is crossed 30 mesh sieves; 40 ℃ of lower oven dry 12h;
(2) preparation of immediate release section
Mix according to the described formulation ratio of claim 1, cross 30 mesh sieve post-dryings;
(3) preparation of slow releasing tablet
After above two kinds of granules mixing, by 30 mesh sieve granulate; The magnesium stearate mixing that adds the described weight portion of claim 1; Last tabletting gets Dex-ibuprofen sustained release tablets.
Advantage of the present invention is: Dex-ibuprofen sustained release tablets provided by the invention, prolong drug release time in vivo be conducive to absorption of human body, improve bioavailability.Vitro release first hour was 15%---35%, and second hour is 30%---55%, and the 4th hour is 50%---75%, and the 7th hour is 65%---90%, and the tenth hour is more than 85%. the pharmacokinetics of dog oral administration shows:
The specific embodiment
The present invention is further illustrated according to specific embodiment for the below, but do not mean that restriction protection scope of the present invention.
One, prescription
Two, prescription foundation
1, (S)-ibuprofen dosage
With reference to the dosage of ibuprofen in the ibuprofen modified release capsule and decide.
2, the selection of adjuvant and consumption thereof
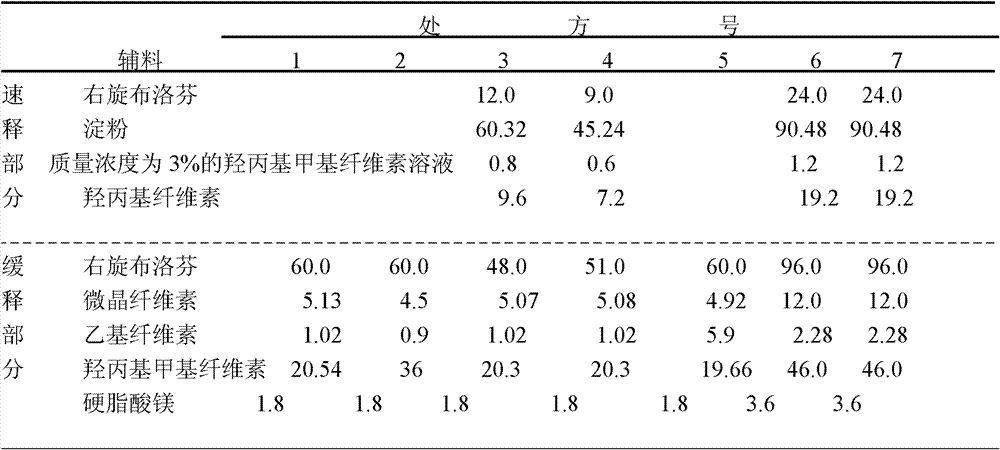
The guiding theory of this sustained-release tablet recipe design, it is very fast to be to make the (S)-ibuprofen onset to produce, and clinical effectiveness is stronger, and release continues longer.Therefore, each composition reference literature of will writing out a prescription is first made respectively rapid release and two kinds of granules of slow release; And then mix; Carry out at last tabletting.By prerun, 26 prescriptions of preliminary examination, screening different auxiliary material and consumption and pressure, take the cumulative in vitro release as index, selected suitable adjuvant, consumption and pressure.Now selecting 7 prescriptions is summarized as follows:
The composition (g) of table 1, seven different prescriptions
No. 6 and No. 7 different pressures of writing out a prescription together of notes *
1-5 number is 200 recipe quantity; 6, No. 7 is 400 recipe quantity.
The cumulative release degree (%) of table 2, seven different prescription slow releasing tablet
(assay method is seen data NO.7)
Comprehensive above-mentioned the selection result is chosen hydroxypropyl emthylcellulose and the ethyl cellulose of proper proportion macromolecular material and is made the purpose that matrix sustained release tablet can reach the slow release (S)-ibuprofen and prolong drug effect.The result of the cumulative release degree of eight prescriptions shows that No. eight prescription wherein is fit to and satisfies the clinical treatment requirement, is more excellent scheme.
Three, preparation technology
(1) preparation of slow-releasing granules
Get (S)-ibuprofen, microcrystalline Cellulose, hydroxypropyl emthylcellulose and mix according to above-mentioned formulation ratio, sieve with 80 orders; Alcoholic solution with ethyl cellulose and hydroxypropyl methylcellulose mixtures is made soft material with said mixture; After secondary is crossed 30 mesh sieves; 40 ℃ of lower oven dry 12h;
(2) preparation of immediate-release granules
Above-mentioned formulation ratio mixes, and crosses 30 mesh sieve post-dryings;
(3) preparation of slow releasing tablet
After above two kinds of granules mixing, by 30 mesh sieve granulate; Add the magnesium stearate mixing; Last tabletting gets Dex-ibuprofen sustained release tablets.
Four, the effect of adjuvant in prescription
The starch disintegrating agent;
The microcrystalline Cellulose disintegrating agent;
Ethyl cellulose blocker binding agent;
The hydroxypropyl cellulose binding agent;
Hydroxypropyl emthylcellulose binding agent blocker;
Magnesium;
Five, adjuvant source and quality standard
Starch: Fructus Vitis viniferae sugar refinery, Hunan, Chinese Pharmacopoeia;
Microcrystalline Cellulose: Shanghai pharmaceutic adjuvant factory, Q/WS-1133-82 difficult to understand;
Ethyl cellulose: Britain blocks happy Kanggong department, American Pharmacopeia;
Hydroxypropyl emthylcellulose: Britain blocks happy Kanggong department, American Pharmacopeia;
Hydroxypropyl cellulose: Shenyang No. 1 Pharmaceutical Factory, Chinese Pharmacopoeia.
Six, documents and materials
Luo Mingsheng etc., pharmaceutical necessities is complete works of, 1993.3, P648,743,806.
Claims (3)
1. a Dex-ibuprofen sustained release tablets is characterized in that, is mixed by immediate release section, slow-released part and magnesium stearate to form;
The immediate release section prescription according to the weight portion formulation ratio that accounts for gross weight is:
The slow-released part prescription according to the weight portion formulation ratio that accounts for gross weight is:
The weight portion of the shared gross weight of described magnesium stearate is 3.0 parts-18.0 parts.
2. described Dex-ibuprofen sustained release tablets according to claim 1 is characterized in that, the immediate release section prescription according to the weight portion formulation ratio that accounts for gross weight is:
The slow-released part prescription according to the weight portion formulation ratio that accounts for gross weight is:
The weight portion of the shared gross weight of described magnesium stearate is 9.0 parts.
3. prepare the method for the described Dex-ibuprofen sustained release tablets of claim 1, it is characterized in that, step is:
(1) preparation of slow-released part
Get (S)-ibuprofen, microcrystalline Cellulose, hydroxypropyl emthylcellulose according to the described formulation ratio mixing of claim 1, sieve with 80 orders; Alcoholic solution with ethyl cellulose and hydroxypropyl methylcellulose mixtures is made soft material with said mixture; After secondary is crossed 30 mesh sieves; 40 ℃ of lower oven dry 12h;
(2) preparation of immediate release section
Mix according to the described formulation ratio of claim 1, cross 30 mesh sieve post-dryings;
(3) preparation of slow releasing tablet
After above two kinds of granules mixing, by 30 mesh sieve granulate; The magnesium stearate mixing that adds the described weight portion of claim 1; Last tabletting gets Dex-ibuprofen sustained release tablets.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2011100786082A CN102160855B (en) | 2011-03-30 | 2011-03-30 | Dex-ibuprofen sustained release tablets and preparation method thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2011100786082A CN102160855B (en) | 2011-03-30 | 2011-03-30 | Dex-ibuprofen sustained release tablets and preparation method thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN102160855A CN102160855A (en) | 2011-08-24 |
| CN102160855B true CN102160855B (en) | 2013-01-23 |
Family
ID=44462481
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN2011100786082A Expired - Fee Related CN102160855B (en) | 2011-03-30 | 2011-03-30 | Dex-ibuprofen sustained release tablets and preparation method thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN102160855B (en) |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2013154512A1 (en) * | 2012-04-13 | 2013-10-17 | Mahmut Bilgic | Pharmaceutical formulations comprising dexibuprofen |
| CN103417505B (en) * | 2012-05-24 | 2016-05-11 | 中国科学院上海药物研究所 | There is huperzine controlled release preparation of two-phase drug release behavior and preparation method thereof |
| CA2924722A1 (en) * | 2013-09-27 | 2015-04-02 | Johnson & Johnson Consumer Inc. | Compression coated pulsatile release compositions |
| CN105935445B (en) * | 2016-03-28 | 2019-02-01 | 赤峰赛林泰药业有限公司 | Pharmaceutical composition and preparation method thereof containing 2- (- 4- isobutyl phenenyl) propionic acid dextrogyre |
| CN110037994B (en) * | 2019-05-24 | 2022-04-12 | 中国药科大学 | A kind of ibuprofen immediate-release sustained-release double-layer tablet and preparation method thereof |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1972666A (en) * | 2003-09-26 | 2007-05-30 | 阿尔扎公司 | OROS push-stick for controlled delivery of active agents |
| CN101073563A (en) * | 2007-02-07 | 2007-11-21 | 西安利君制药有限责任公司 | Chiral composition containing dextrothyroxine buprofenli and levomethadyl cysteliqin and its double slow-releasing tablet |
| CN101784262A (en) * | 2007-08-15 | 2010-07-21 | 麦克内尔-Ppc股份有限公司 | Immediate release and sustained release ibuprofen dosing regiment |
-
2011
- 2011-03-30 CN CN2011100786082A patent/CN102160855B/en not_active Expired - Fee Related
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1972666A (en) * | 2003-09-26 | 2007-05-30 | 阿尔扎公司 | OROS push-stick for controlled delivery of active agents |
| CN101073563A (en) * | 2007-02-07 | 2007-11-21 | 西安利君制药有限责任公司 | Chiral composition containing dextrothyroxine buprofenli and levomethadyl cysteliqin and its double slow-releasing tablet |
| CN101784262A (en) * | 2007-08-15 | 2010-07-21 | 麦克内尔-Ppc股份有限公司 | Immediate release and sustained release ibuprofen dosing regiment |
Non-Patent Citations (2)
| Title |
|---|
| 洪怡等.右旋布洛芬缓释片的制备及其释放度测定.《中国药师》.2006,第861-862页. * |
| 牟晓虹等.布洛芬缓释片的制备及工艺研究.《沈阳药科大学学报》.1996,第5-8页,第40页. * |
Also Published As
| Publication number | Publication date |
|---|---|
| CN102160855A (en) | 2011-08-24 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN102657629B (en) | Ticagrelor sustained-release tablet system and preparation method thereof | |
| CN102160855B (en) | Dex-ibuprofen sustained release tablets and preparation method thereof | |
| CN106924208A (en) | A kind of compound Dapagliflozin Metformin Extended-release Tablets and preparation method thereof | |
| CN104248767A (en) | Ibuprofen preparation and preparation method thereof | |
| CN101601670B (en) | Compound niclosamide parasite-expelling tablet used for pet and preparation method thereof | |
| CN102276447B (en) | Naproxen hydrate crystal, preparation method thereof and medicinal composition containing naproxen hydrate crystal and sumatriptan | |
| CN101342177B (en) | Lornoxicam double-layer sustained release tablets | |
| CN104771400A (en) | Oral pharmaceutical composition of diacerein and berberine, and applications thereof | |
| CN101658507B (en) | Glyceryl guaiacolate and pseudoephedrine compound sustained release preparation | |
| CN105250233B (en) | (S)-ibuprofen enteric-coated sustained-release tablet and preparation method thereof | |
| CN104367562A (en) | Pramipexole dihydrochloride slow-release tablets and preparation method thereof | |
| CN102429883A (en) | Fluoxetine hydrochloride oral cavity disintegrating medicinal composition | |
| CN100571700C (en) | Carbazochrome sodium sulfonate slow-released tablet and preparation method thereof | |
| CN101716162B (en) | Aminophylline slow-release capsules and preparation method thereof | |
| CN101579342A (en) | Desloratadine-contained patulin composition | |
| CN101518519A (en) | Naringin slow-release tablet and method for preparing same | |
| CN101422441A (en) | Nimesulide sustained-release tablet and preparation method thereof | |
| CN101822648A (en) | Preparation method and application of nano dobesilate calcium capsule or tablet | |
| CN103432596B (en) | Method for researching abirritation mechanism of Chinese herbal medicinal ingredients of Xinhuang tablets | |
| CN103156877B (en) | A kind of capecitabine fast release micropill and preparation method thereof | |
| CN101766608B (en) | Compound pseudoephedrine hydrochloride sustained-release preparation and preparation method thereof | |
| CN101756987A (en) | Compound sustained-release preparation of guaiacol olycerin ether, pseudoephedrine and dextromethorphan | |
| CN102225065B (en) | Parcetamol pseudoephedrine hydrochloride and dextromethorphan hydrochloride capsule and preparation method thereof | |
| CN101757001B (en) | Compound slow-release preparation of benorilate, pseudoephedrine and chlorphenamine | |
| CN102335153A (en) | Piperazine ferulate sustained-release tablet and its preparation method |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| CF01 | Termination of patent right due to non-payment of annual fee |
Granted publication date: 20130123 Termination date: 20180330 |
|
| CF01 | Termination of patent right due to non-payment of annual fee |