CN102093309A - 非布司他的新晶型及其制备方法 - Google Patents
非布司他的新晶型及其制备方法 Download PDFInfo
- Publication number
- CN102093309A CN102093309A CN 201110027282 CN201110027282A CN102093309A CN 102093309 A CN102093309 A CN 102093309A CN 201110027282 CN201110027282 CN 201110027282 CN 201110027282 A CN201110027282 A CN 201110027282A CN 102093309 A CN102093309 A CN 102093309A
- Authority
- CN
- China
- Prior art keywords
- febuxostat
- crystal form
- crystal
- crystal forms
- solvent
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 239000013078 crystal Substances 0.000 title claims abstract description 81
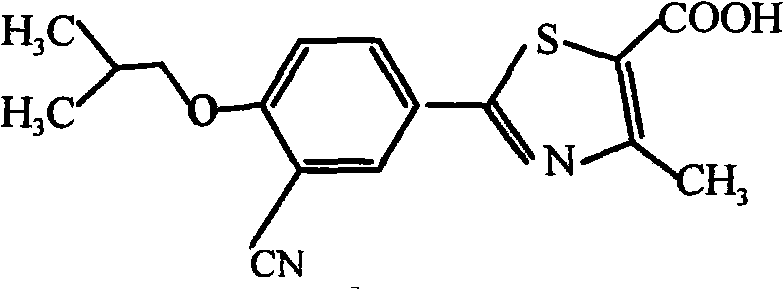
- BQSJTQLCZDPROO-UHFFFAOYSA-N febuxostat Chemical compound C1=C(C#N)C(OCC(C)C)=CC=C1C1=NC(C)=C(C(O)=O)S1 BQSJTQLCZDPROO-UHFFFAOYSA-N 0.000 title claims abstract description 57
- 229960005101 febuxostat Drugs 0.000 title claims abstract description 54
- 238000002360 preparation method Methods 0.000 title abstract description 20
- 238000010521 absorption reaction Methods 0.000 claims abstract description 18
- 239000008194 pharmaceutical composition Substances 0.000 claims abstract description 13
- 238000000634 powder X-ray diffraction Methods 0.000 claims abstract description 13
- 201000001431 Hyperuricemia Diseases 0.000 claims abstract description 7
- 201000010099 disease Diseases 0.000 claims abstract description 7
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 claims abstract description 7
- 239000003814 drug Substances 0.000 claims abstract description 5
- 238000004519 manufacturing process Methods 0.000 claims abstract description 5
- 229940079593 drug Drugs 0.000 claims abstract description 3
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical group CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 claims description 24
- 239000002904 solvent Substances 0.000 claims description 16
- 238000000034 method Methods 0.000 claims description 10
- 238000010438 heat treatment Methods 0.000 claims description 9
- 238000002329 infrared spectrum Methods 0.000 claims description 8
- 238000002425 crystallisation Methods 0.000 claims description 7
- 230000008025 crystallization Effects 0.000 claims description 7
- FVSKHRXBFJPNKK-UHFFFAOYSA-N propionitrile Chemical compound CCC#N FVSKHRXBFJPNKK-UHFFFAOYSA-N 0.000 claims description 6
- 238000000967 suction filtration Methods 0.000 claims description 3
- 239000002671 adjuvant Substances 0.000 claims description 2
- 125000000217 alkyl group Chemical group 0.000 claims description 2
- 125000001188 haloalkyl group Chemical group 0.000 claims description 2
- 239000002245 particle Substances 0.000 claims description 2
- 239000002244 precipitate Substances 0.000 claims description 2
- 239000003826 tablet Substances 0.000 description 15
- 230000009102 absorption Effects 0.000 description 13
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 12
- 239000000843 powder Substances 0.000 description 12
- 239000002775 capsule Substances 0.000 description 9
- 229920002472 Starch Polymers 0.000 description 8
- 239000008107 starch Substances 0.000 description 7
- 235000019698 starch Nutrition 0.000 description 7
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 6
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 6
- 238000000862 absorption spectrum Methods 0.000 description 6
- 235000019359 magnesium stearate Nutrition 0.000 description 6
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 6
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 6
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 6
- 229920000168 Microcrystalline cellulose Polymers 0.000 description 5
- 239000001866 hydroxypropyl methyl cellulose Substances 0.000 description 5
- 235000010979 hydroxypropyl methyl cellulose Nutrition 0.000 description 5
- 229920003088 hydroxypropyl methyl cellulose Polymers 0.000 description 5
- UFVKGYZPFZQRLF-UHFFFAOYSA-N hydroxypropyl methyl cellulose Chemical compound OC1C(O)C(OC)OC(CO)C1OC1C(O)C(O)C(OC2C(C(O)C(OC3C(C(O)C(O)C(CO)O3)O)C(CO)O2)O)C(CO)O1 UFVKGYZPFZQRLF-UHFFFAOYSA-N 0.000 description 5
- 239000008108 microcrystalline cellulose Substances 0.000 description 5
- 235000019813 microcrystalline cellulose Nutrition 0.000 description 5
- 229940016286 microcrystalline cellulose Drugs 0.000 description 5
- 239000000203 mixture Substances 0.000 description 5
- 238000003860 storage Methods 0.000 description 5
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 5
- LEHOTFFKMJEONL-UHFFFAOYSA-N Uric Acid Chemical compound N1C(=O)NC(=O)C2=C1NC(=O)N2 LEHOTFFKMJEONL-UHFFFAOYSA-N 0.000 description 4
- TVWHNULVHGKJHS-UHFFFAOYSA-N Uric acid Natural products N1C(=O)NC(=O)C2NC(=O)NC21 TVWHNULVHGKJHS-UHFFFAOYSA-N 0.000 description 4
- 239000008280 blood Substances 0.000 description 4
- 210000004369 blood Anatomy 0.000 description 4
- 239000003795 chemical substances by application Substances 0.000 description 4
- 239000011248 coating agent Substances 0.000 description 4
- 238000000576 coating method Methods 0.000 description 4
- 238000010586 diagram Methods 0.000 description 4
- 239000008187 granular material Substances 0.000 description 4
- 239000000314 lubricant Substances 0.000 description 4
- 238000005259 measurement Methods 0.000 description 4
- 239000000546 pharmaceutical excipient Substances 0.000 description 4
- 239000002994 raw material Substances 0.000 description 4
- 238000003756 stirring Methods 0.000 description 4
- 229940116269 uric acid Drugs 0.000 description 4
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 3
- 229920000881 Modified starch Polymers 0.000 description 3
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 3
- 238000002441 X-ray diffraction Methods 0.000 description 3
- 238000004458 analytical method Methods 0.000 description 3
- 239000011230 binding agent Substances 0.000 description 3
- 238000002474 experimental method Methods 0.000 description 3
- 239000000945 filler Substances 0.000 description 3
- 238000011835 investigation Methods 0.000 description 3
- 239000007787 solid Substances 0.000 description 3
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 2
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 2
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 2
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 2
- 201000005569 Gout Diseases 0.000 description 2
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 2
- 229930195725 Mannitol Natural products 0.000 description 2
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 2
- YKTSYUJCYHOUJP-UHFFFAOYSA-N [O--].[Al+3].[Al+3].[O-][Si]([O-])([O-])[O-] Chemical compound [O--].[Al+3].[Al+3].[O-][Si]([O-])([O-])[O-] YKTSYUJCYHOUJP-UHFFFAOYSA-N 0.000 description 2
- CDQSJQSWAWPGKG-UHFFFAOYSA-N butane-1,1-diol Chemical compound CCCC(O)O CDQSJQSWAWPGKG-UHFFFAOYSA-N 0.000 description 2
- 239000003086 colorant Substances 0.000 description 2
- 150000001875 compounds Chemical class 0.000 description 2
- 238000007796 conventional method Methods 0.000 description 2
- 238000007907 direct compression Methods 0.000 description 2
- 239000007884 disintegrant Substances 0.000 description 2
- 239000002552 dosage form Substances 0.000 description 2
- 235000019441 ethanol Nutrition 0.000 description 2
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 2
- -1 fatty acid esters Chemical class 0.000 description 2
- 238000011049 filling Methods 0.000 description 2
- 238000005469 granulation Methods 0.000 description 2
- 230000003179 granulation Effects 0.000 description 2
- IPCSVZSSVZVIGE-UHFFFAOYSA-N hexadecanoic acid Chemical compound CCCCCCCCCCCCCCCC(O)=O IPCSVZSSVZVIGE-UHFFFAOYSA-N 0.000 description 2
- 239000008101 lactose Substances 0.000 description 2
- 230000007774 longterm Effects 0.000 description 2
- 229940031703 low substituted hydroxypropyl cellulose Drugs 0.000 description 2
- 239000000594 mannitol Substances 0.000 description 2
- 235000010355 mannitol Nutrition 0.000 description 2
- 238000002844 melting Methods 0.000 description 2
- 230000008018 melting Effects 0.000 description 2
- LYRFLYHAGKPMFH-UHFFFAOYSA-N octadecanamide Chemical compound CCCCCCCCCCCCCCCCCC(N)=O LYRFLYHAGKPMFH-UHFFFAOYSA-N 0.000 description 2
- 239000003921 oil Substances 0.000 description 2
- 238000004321 preservation Methods 0.000 description 2
- 239000000243 solution Substances 0.000 description 2
- 239000000600 sorbitol Substances 0.000 description 2
- 235000010356 sorbitol Nutrition 0.000 description 2
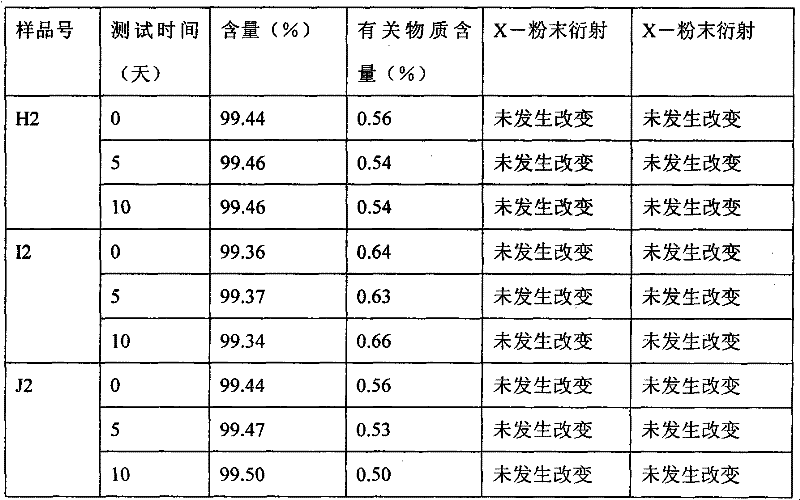
- 238000013112 stability test Methods 0.000 description 2
- 230000003068 static effect Effects 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- 239000007939 sustained release tablet Substances 0.000 description 2
- 238000005550 wet granulation Methods 0.000 description 2
- 239000000080 wetting agent Substances 0.000 description 2
- JNAYPRPPXRWGQO-UHFFFAOYSA-N 2-chloropropanenitrile Chemical compound CC(Cl)C#N JNAYPRPPXRWGQO-UHFFFAOYSA-N 0.000 description 1
- 229920000178 Acrylic resin Polymers 0.000 description 1
- 239000004925 Acrylic resin Substances 0.000 description 1
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 1
- 229920002261 Corn starch Polymers 0.000 description 1
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 1
- 229920003136 Eudragit® L polymer Polymers 0.000 description 1
- 229920002153 Hydroxypropyl cellulose Polymers 0.000 description 1
- 206010028980 Neoplasm Diseases 0.000 description 1
- 235000021314 Palmitic acid Nutrition 0.000 description 1
- 239000004372 Polyvinyl alcohol Substances 0.000 description 1
- 235000021355 Stearic acid Nutrition 0.000 description 1
- 229930006000 Sucrose Natural products 0.000 description 1
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 1
- TVXBFESIOXBWNM-UHFFFAOYSA-N Xylitol Natural products OCCC(O)C(O)C(O)CCO TVXBFESIOXBWNM-UHFFFAOYSA-N 0.000 description 1
- DPXJVFZANSGRMM-UHFFFAOYSA-N acetic acid;2,3,4,5,6-pentahydroxyhexanal;sodium Chemical compound [Na].CC(O)=O.OCC(O)C(O)C(O)C(O)C=O DPXJVFZANSGRMM-UHFFFAOYSA-N 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 239000006189 buccal tablet Substances 0.000 description 1
- KVNRLNFWIYMESJ-UHFFFAOYSA-N butyronitrile Chemical compound CCCC#N KVNRLNFWIYMESJ-UHFFFAOYSA-N 0.000 description 1
- CJZGTCYPCWQAJB-UHFFFAOYSA-L calcium stearate Chemical compound [Ca+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O CJZGTCYPCWQAJB-UHFFFAOYSA-L 0.000 description 1
- 239000008116 calcium stearate Substances 0.000 description 1
- 235000013539 calcium stearate Nutrition 0.000 description 1
- 201000011510 cancer Diseases 0.000 description 1
- 239000001768 carboxy methyl cellulose Substances 0.000 description 1
- 235000010980 cellulose Nutrition 0.000 description 1
- 229920002678 cellulose Polymers 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 238000002512 chemotherapy Methods 0.000 description 1
- 239000007910 chewable tablet Substances 0.000 description 1
- 239000000460 chlorine Substances 0.000 description 1
- 229910052801 chlorine Inorganic materials 0.000 description 1
- 239000007931 coated granule Substances 0.000 description 1
- 238000007906 compression Methods 0.000 description 1
- 230000006835 compression Effects 0.000 description 1
- 239000008120 corn starch Substances 0.000 description 1
- 229940099112 cornstarch Drugs 0.000 description 1
- 235000014113 dietary fatty acids Nutrition 0.000 description 1
- 239000003085 diluting agent Substances 0.000 description 1
- 239000008298 dragée Substances 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 239000007938 effervescent tablet Substances 0.000 description 1
- 238000005538 encapsulation Methods 0.000 description 1
- 239000000194 fatty acid Substances 0.000 description 1
- 229930195729 fatty acid Natural products 0.000 description 1
- 239000007941 film coated tablet Substances 0.000 description 1
- 239000000796 flavoring agent Substances 0.000 description 1
- 235000013355 food flavoring agent Nutrition 0.000 description 1
- 235000003599 food sweetener Nutrition 0.000 description 1
- 235000011187 glycerol Nutrition 0.000 description 1
- 239000001341 hydroxy propyl starch Substances 0.000 description 1
- 239000001863 hydroxypropyl cellulose Substances 0.000 description 1
- 235000010977 hydroxypropyl cellulose Nutrition 0.000 description 1
- 235000013828 hydroxypropyl starch Nutrition 0.000 description 1
- 239000012535 impurity Substances 0.000 description 1
- 238000002347 injection Methods 0.000 description 1
- 239000007924 injection Substances 0.000 description 1
- 239000011159 matrix material Substances 0.000 description 1
- 230000001404 mediated effect Effects 0.000 description 1
- HEBKCHPVOIAQTA-UHFFFAOYSA-N meso ribitol Natural products OCC(O)C(O)C(O)CO HEBKCHPVOIAQTA-UHFFFAOYSA-N 0.000 description 1
- NBTOZLQBSIZIKS-UHFFFAOYSA-N methoxide Chemical compound [O-]C NBTOZLQBSIZIKS-UHFFFAOYSA-N 0.000 description 1
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 1
- 239000012046 mixed solvent Substances 0.000 description 1
- WQEPLUUGTLDZJY-UHFFFAOYSA-N n-Pentadecanoic acid Natural products CCCCCCCCCCCCCCC(O)=O WQEPLUUGTLDZJY-UHFFFAOYSA-N 0.000 description 1
- 239000002547 new drug Substances 0.000 description 1
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 1
- OQCDKBAXFALNLD-UHFFFAOYSA-N octadecanoic acid Natural products CCCCCCCC(C)CCCCCCCCC(O)=O OQCDKBAXFALNLD-UHFFFAOYSA-N 0.000 description 1
- 239000008184 oral solid dosage form Substances 0.000 description 1
- 239000006191 orally-disintegrating tablet Substances 0.000 description 1
- 239000000825 pharmaceutical preparation Substances 0.000 description 1
- 239000004014 plasticizer Substances 0.000 description 1
- 229920002451 polyvinyl alcohol Polymers 0.000 description 1
- 238000001556 precipitation Methods 0.000 description 1
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 235000013772 propylene glycol Nutrition 0.000 description 1
- 238000001959 radiotherapy Methods 0.000 description 1
- 238000001953 recrystallisation Methods 0.000 description 1
- 239000000377 silicon dioxide Substances 0.000 description 1
- 235000012239 silicon dioxide Nutrition 0.000 description 1
- 239000002002 slurry Substances 0.000 description 1
- 235000019812 sodium carboxymethyl cellulose Nutrition 0.000 description 1
- 229920001027 sodium carboxymethylcellulose Polymers 0.000 description 1
- 229960002920 sorbitol Drugs 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 229940037312 stearamide Drugs 0.000 description 1
- 239000008117 stearic acid Substances 0.000 description 1
- 239000005720 sucrose Substances 0.000 description 1
- 235000000346 sugar Nutrition 0.000 description 1
- 239000007940 sugar coated tablet Substances 0.000 description 1
- 150000008163 sugars Chemical class 0.000 description 1
- 239000003765 sweetening agent Substances 0.000 description 1
- 239000000454 talc Substances 0.000 description 1
- 229910052623 talc Inorganic materials 0.000 description 1
- 239000006068 taste-masking agent Substances 0.000 description 1
- 238000011287 therapeutic dose Methods 0.000 description 1
- 239000000811 xylitol Substances 0.000 description 1
- 235000010447 xylitol Nutrition 0.000 description 1
- HEBKCHPVOIAQTA-SCDXWVJYSA-N xylitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)CO HEBKCHPVOIAQTA-SCDXWVJYSA-N 0.000 description 1
- 229960002675 xylitol Drugs 0.000 description 1
Images
Landscapes
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
Claims (8)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN 201110027282 CN102093309B (zh) | 2006-12-07 | 2006-12-07 | 非布司他的晶型及其制备方法 |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN 201110027282 CN102093309B (zh) | 2006-12-07 | 2006-12-07 | 非布司他的晶型及其制备方法 |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN2006100952630A Division CN1970547B (zh) | 2006-12-07 | 2006-12-07 | 非布司他的晶型及其制备方法 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN102093309A true CN102093309A (zh) | 2011-06-15 |
| CN102093309B CN102093309B (zh) | 2012-07-04 |
Family
ID=44126596
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN 201110027282 Active CN102093309B (zh) | 2006-12-07 | 2006-12-07 | 非布司他的晶型及其制备方法 |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN102093309B (zh) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8415481B2 (en) | 2009-06-10 | 2013-04-09 | Teva Pharmaceuticals Usa, Inc. | Crystalline form of febuxostat |
| CN103467412A (zh) * | 2013-09-30 | 2013-12-25 | 杭州朱养心药业有限公司 | 用于痛风的药物化合物 |
| WO2016091230A1 (en) | 2014-12-12 | 2016-06-16 | Zentiva, K.S. | Formulations containing a solid solution of febuxostat |
| CN114341120A (zh) * | 2019-08-21 | 2022-04-12 | 国立大学法人东京大学 | Abcc11抑制剂 |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE69122084T2 (de) * | 1990-11-30 | 1997-04-03 | Teijin Ltd., Osaka | 2-arylthiazolderivat sowie dieses enthaltendes arzneimittel |
| SK287928B6 (sk) * | 1998-06-19 | 2012-04-03 | Teijin Pharma Limited | Polymorphic modifications of 2-(3-cyano-4-isobutyloxyphenyl)-4- methyl-5-thiazole-carboxylic acid and processes for the preparation thereof |
-
2006
- 2006-12-07 CN CN 201110027282 patent/CN102093309B/zh active Active
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8415481B2 (en) | 2009-06-10 | 2013-04-09 | Teva Pharmaceuticals Usa, Inc. | Crystalline form of febuxostat |
| US8609856B2 (en) | 2009-06-10 | 2013-12-17 | Teva Pharmaceuticals Usa, Inc. | Crystalline forms of Febuxostat |
| US8742129B2 (en) | 2009-06-10 | 2014-06-03 | Teva Pharmaceutical Industries Ltd. | Crystalline forms of febuxostat |
| CN103467412A (zh) * | 2013-09-30 | 2013-12-25 | 杭州朱养心药业有限公司 | 用于痛风的药物化合物 |
| WO2016091230A1 (en) | 2014-12-12 | 2016-06-16 | Zentiva, K.S. | Formulations containing a solid solution of febuxostat |
| CN114341120A (zh) * | 2019-08-21 | 2022-04-12 | 国立大学法人东京大学 | Abcc11抑制剂 |
Also Published As
| Publication number | Publication date |
|---|---|
| CN102093309B (zh) | 2012-07-04 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN1970547B (zh) | 非布司他的晶型及其制备方法 | |
| ES2562843T3 (es) | Forma IV de clorhidrato de ivabradina | |
| CN104797566A (zh) | 沃替西汀氢溴酸盐的新结晶形式 | |
| CN102007138B (zh) | 吡唑衍生物的半富马酸盐 | |
| MX2008002541A (es) | Formas de cristal delta y epsilon de mesilato de imatinib. | |
| JPWO2004022538A1 (ja) | 経口用固形医薬用結晶およびそれを含む排尿障害治療用経口用固形医薬 | |
| CN110054624B (zh) | 盐酸小檗碱与咖啡酸共晶物及制备方法和其组合物与用途 | |
| JP2016509031A (ja) | トレラグリプチンの固形形式及びその製造方法と用途 | |
| JP6126040B2 (ja) | ニコサミド(Nicousamide)化合物の五つの晶型、その製法やその薬物組合と用途 | |
| WO2016155670A1 (zh) | 一种cdk抑制剂和mek抑制剂的共晶及其制备方法 | |
| CN102093309B (zh) | 非布司他的晶型及其制备方法 | |
| WO2016095650A1 (zh) | 盐酸优克那非的多晶型物及其制备方法、组合物和用途 | |
| CN106008277A (zh) | 一种新型二甲双胍盐酸盐及其制备方法 | |
| KR20180006441A (ko) | 요산 수송체 억제제의 나트륨 염 및 이의 결정성 형태 | |
| CN101993417A (zh) | 磷酸二甲啡烷的稳定新晶型 | |
| WO2010111951A1 (zh) | 普拉格雷氢溴酸盐的晶体 | |
| CN115141161A (zh) | 一种普拉克索昔萘酸盐的晶型a及其制备方法 | |
| CN115536654A (zh) | 小檗碱橙皮素盐晶型及其制备方法和其组合物与应用 | |
| CN109721557B (zh) | 来曲唑晶ii型固体物质及制备方法和其药物组合物与用途 | |
| CN120271529B (zh) | 一种非布司他-金雀花碱共晶及其制备方法和应用 | |
| JP2018510173A (ja) | トピロキソスタットの新規結晶形及びその製造方法 | |
| CN102093308B (zh) | 非布司他的晶型及其制备方法 | |
| CN113831252A (zh) | 咖啡酸文拉法辛及制备方法和其组合物与用途 | |
| CN102093387B (zh) | 4,5,6,7-四氢异噁唑并[5,4-c]吡啶-3-醇一水合物的晶型化合物 | |
| JP7671362B2 (ja) | 医薬組成物、その製造方法及び使用 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| TR01 | Transfer of patent right |
Effective date of registration: 20200720 Address after: 221001 south side of Dongshan, comprehensive zone, Jinshanqiao Development Zone, Xuzhou City, Jiangsu Province Co-patentee after: JIANGSU WANBANG BIOPHARMACEUTICAL GROUP CO.,LTD. Patentee after: Xuzhou Wanbang Jinqiao Pharmaceutical Co.,Ltd. Address before: 400061 No. 565, Tu Shan Road, Nan'an District, Chongqing Patentee before: CHONGQING PHARMACEUTICAL RESEARCH INSTITUTE Co.,Ltd. |
|
| TR01 | Transfer of patent right | ||
| CP03 | Change of name, title or address |
Address after: 221001 south side of Dongshan, comprehensive zone, Jinshanqiao Development Zone, Xuzhou City, Jiangsu Province Patentee after: Xuzhou Wanbang Jinqiao Pharmaceutical Co.,Ltd. Country or region after: China Patentee after: Fosun Wanbang (Jiangsu) Pharmaceutical Group Co.,Ltd. Address before: 221001 south side of Dongshan, comprehensive zone, Jinshanqiao Development Zone, Xuzhou City, Jiangsu Province Patentee before: Xuzhou Wanbang Jinqiao Pharmaceutical Co.,Ltd. Country or region before: China Patentee before: JIANGSU WANBANG BIOPHARMACEUTICAL GROUP CO.,LTD. |
|
| CP03 | Change of name, title or address |