CN102070526B - Method for synthesizing 3-aza-bicyclo[4.1.0]heptane-6-formic acid with protective group - Google Patents
Method for synthesizing 3-aza-bicyclo[4.1.0]heptane-6-formic acid with protective group Download PDFInfo
- Publication number
- CN102070526B CN102070526B CN2009102018600A CN200910201860A CN102070526B CN 102070526 B CN102070526 B CN 102070526B CN 2009102018600 A CN2009102018600 A CN 2009102018600A CN 200910201860 A CN200910201860 A CN 200910201860A CN 102070526 B CN102070526 B CN 102070526B
- Authority
- CN
- China
- Prior art keywords
- reaction
- aza
- bicyclo
- heptane
- formic acid
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
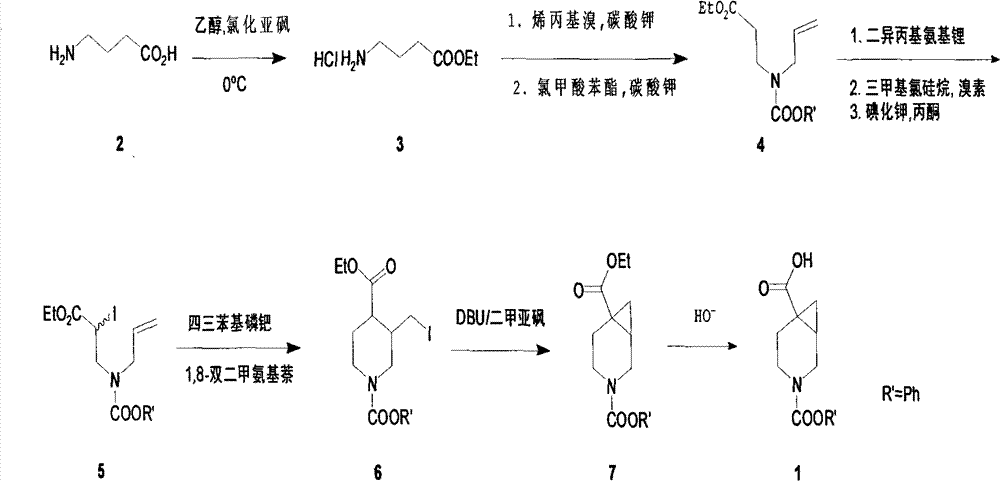
- 125000006239 protecting group Chemical group 0.000 title claims abstract description 11
- DIITVVANFKGDNX-UHFFFAOYSA-N 3-azabicyclo[4.1.0]heptane-6-carboxylic acid Chemical compound C1NCCC2(C(=O)O)C1C2 DIITVVANFKGDNX-UHFFFAOYSA-N 0.000 title abstract description 6
- 238000000034 method Methods 0.000 title abstract description 5
- 230000002194 synthesizing effect Effects 0.000 title 1
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims abstract description 31
- 238000006243 chemical reaction Methods 0.000 claims abstract description 29
- 239000002994 raw material Substances 0.000 claims abstract description 6
- 238000007363 ring formation reaction Methods 0.000 claims abstract description 5
- 238000005904 alkaline hydrolysis reaction Methods 0.000 claims abstract description 3
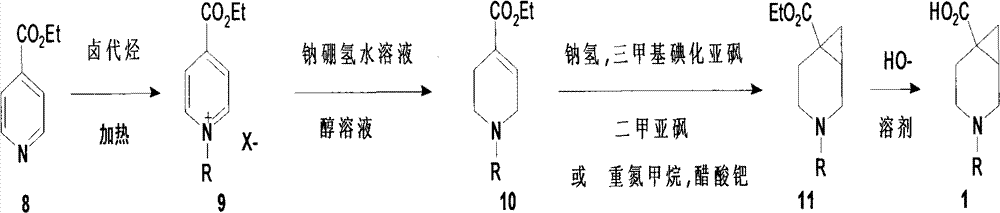
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 claims description 33
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 claims description 14
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 claims description 12
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 claims description 12
- BDAGIHXWWSANSR-UHFFFAOYSA-N formic acid Substances OC=O BDAGIHXWWSANSR-UHFFFAOYSA-N 0.000 claims description 10
- 229910000033 sodium borohydride Inorganic materials 0.000 claims description 9
- 239000012279 sodium borohydride Substances 0.000 claims description 9
- XBLVHTDFJBKJLG-UHFFFAOYSA-N Ethyl nicotinate Chemical compound CCOC(=O)C1=CC=CN=C1 XBLVHTDFJBKJLG-UHFFFAOYSA-N 0.000 claims description 8
- 238000010189 synthetic method Methods 0.000 claims description 8
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 claims description 7
- 238000004440 column chromatography Methods 0.000 claims description 7
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 claims description 6
- 238000006460 hydrolysis reaction Methods 0.000 claims description 6
- 239000003960 organic solvent Substances 0.000 claims description 6
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 claims description 6
- 239000003513 alkali Substances 0.000 claims description 5
- WMFOQBRAJBCJND-UHFFFAOYSA-M Lithium hydroxide Chemical compound [Li+].[OH-] WMFOQBRAJBCJND-UHFFFAOYSA-M 0.000 claims description 4
- 229940064982 ethylnicotinate Drugs 0.000 claims description 4
- 229910052763 palladium Inorganic materials 0.000 claims description 4
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 4
- MZFQJBMXUXJUHF-UHFFFAOYSA-N 4-azabicyclo[4.1.0]heptane Chemical compound C1CNCC2CC21 MZFQJBMXUXJUHF-UHFFFAOYSA-N 0.000 claims description 3
- YXHKONLOYHBTNS-UHFFFAOYSA-N Diazomethane Chemical compound C=[N+]=[N-] YXHKONLOYHBTNS-UHFFFAOYSA-N 0.000 claims description 3
- AGEZXYOZHKGVCM-UHFFFAOYSA-N benzyl bromide Chemical compound BrCC1=CC=CC=C1 AGEZXYOZHKGVCM-UHFFFAOYSA-N 0.000 claims description 3
- BPLKQGGAXWRFOE-UHFFFAOYSA-M trimethylsulfoxonium iodide Chemical compound [I-].C[S+](C)(C)=O BPLKQGGAXWRFOE-UHFFFAOYSA-M 0.000 claims description 3
- 125000002252 acyl group Chemical group 0.000 claims description 2
- 125000000217 alkyl group Chemical group 0.000 claims description 2
- 125000003368 amide group Chemical group 0.000 claims description 2
- 150000001412 amines Chemical group 0.000 claims description 2
- 125000004494 ethyl ester group Chemical group 0.000 claims description 2
- 239000012046 mixed solvent Substances 0.000 claims description 2
- 238000000926 separation method Methods 0.000 claims description 2
- 239000000758 substrate Substances 0.000 claims description 2
- PKAUVIXBZJUYRV-UHFFFAOYSA-N methane;hydroiodide Chemical compound C.I PKAUVIXBZJUYRV-UHFFFAOYSA-N 0.000 claims 1
- 239000012453 solvate Substances 0.000 claims 1
- 238000000746 purification Methods 0.000 abstract description 7
- 150000005826 halohydrocarbons Chemical class 0.000 abstract description 5
- -1 cyanoborohydride Chemical compound 0.000 abstract description 3
- 238000010992 reflux Methods 0.000 abstract description 3
- 238000005804 alkylation reaction Methods 0.000 abstract 1
- 238000011097 chromatography purification Methods 0.000 abstract 1
- MCRPKBUFXAKDKI-UHFFFAOYSA-N ethyl pyridine-4-carboxylate Chemical compound CCOC(=O)C1=CC=NC=C1 MCRPKBUFXAKDKI-UHFFFAOYSA-N 0.000 abstract 1
- 238000004519 manufacturing process Methods 0.000 abstract 1
- 125000001453 quaternary ammonium group Chemical group 0.000 abstract 1
- 238000003756 stirring Methods 0.000 description 10
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 9
- 239000000243 solution Substances 0.000 description 8
- QFLWZFQWSBQYPS-AWRAUJHKSA-N (3S)-3-[[(2S)-2-[[(2S)-2-[5-[(3aS,6aR)-2-oxo-1,3,3a,4,6,6a-hexahydrothieno[3,4-d]imidazol-4-yl]pentanoylamino]-3-methylbutanoyl]amino]-3-(4-hydroxyphenyl)propanoyl]amino]-4-[1-bis(4-chlorophenoxy)phosphorylbutylamino]-4-oxobutanoic acid Chemical compound CCCC(NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@@H](NC(=O)CCCCC1SC[C@@H]2NC(=O)N[C@H]12)C(C)C)P(=O)(Oc1ccc(Cl)cc1)Oc1ccc(Cl)cc1 QFLWZFQWSBQYPS-AWRAUJHKSA-N 0.000 description 5
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 5
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 4
- 150000001875 compounds Chemical class 0.000 description 4
- 239000002904 solvent Substances 0.000 description 4
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 3
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical class [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 3
- 239000007864 aqueous solution Substances 0.000 description 3
- 238000001035 drying Methods 0.000 description 3
- 239000007788 liquid Substances 0.000 description 3
- 239000011734 sodium Substances 0.000 description 3
- 239000007787 solid Substances 0.000 description 3
- 238000005406 washing Methods 0.000 description 3
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 2
- IMNFDUFMRHMDMM-UHFFFAOYSA-N N-Heptane Chemical compound CCCCCCC IMNFDUFMRHMDMM-UHFFFAOYSA-N 0.000 description 2
- 230000001476 alcoholic effect Effects 0.000 description 2
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 2
- 238000001514 detection method Methods 0.000 description 2
- 238000005516 engineering process Methods 0.000 description 2
- 238000001914 filtration Methods 0.000 description 2
- INQOMBQAUSQDDS-UHFFFAOYSA-N iodomethane Chemical compound IC INQOMBQAUSQDDS-UHFFFAOYSA-N 0.000 description 2
- 239000011259 mixed solution Substances 0.000 description 2
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 2
- 238000002360 preparation method Methods 0.000 description 2
- 238000010025 steaming Methods 0.000 description 2
- 125000003903 2-propenyl group Chemical group [H]C([*])([H])C([H])=C([H])[H] 0.000 description 1
- RMTDJEYZSNNPNM-UHFFFAOYSA-N 3-[(2-methylpropan-2-yl)oxycarbonyl]-3-azabicyclo[4.1.0]heptane-6-carboxylic acid Chemical compound C1N(C(=O)OC(C)(C)C)CCC2(C(O)=O)CC21 RMTDJEYZSNNPNM-UHFFFAOYSA-N 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 1
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- KCXMKQUNVWSEMD-UHFFFAOYSA-N benzyl chloride Chemical compound ClCC1=CC=CC=C1 KCXMKQUNVWSEMD-UHFFFAOYSA-N 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 239000003054 catalyst Substances 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 239000000460 chlorine Substances 0.000 description 1
- 229940125904 compound 1 Drugs 0.000 description 1
- 229940125773 compound 10 Drugs 0.000 description 1
- 239000012043 crude product Substances 0.000 description 1
- 230000006837 decompression Effects 0.000 description 1
- KPUNOVLMCQQCSK-UHFFFAOYSA-N diazomethane;ethoxyethane Chemical compound C=[N+]=[N-].CCOCC KPUNOVLMCQQCSK-UHFFFAOYSA-N 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 230000032050 esterification Effects 0.000 description 1
- 238000005886 esterification reaction Methods 0.000 description 1
- 238000003810 ethyl acetate extraction Methods 0.000 description 1
- PQVSTLUFSYVLTO-UHFFFAOYSA-N ethyl n-ethoxycarbonylcarbamate Chemical compound CCOC(=O)NC(=O)OCC PQVSTLUFSYVLTO-UHFFFAOYSA-N 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- 239000000706 filtrate Substances 0.000 description 1
- 229910052736 halogen Inorganic materials 0.000 description 1
- 230000002140 halogenating effect Effects 0.000 description 1
- 150000002367 halogens Chemical group 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- 150000002431 hydrogen Chemical class 0.000 description 1
- 238000005984 hydrogenation reaction Methods 0.000 description 1
- 239000012535 impurity Substances 0.000 description 1
- ZLVXBBHTMQJRSX-VMGNSXQWSA-N jdtic Chemical compound C1([C@]2(C)CCN(C[C@@H]2C)C[C@H](C(C)C)NC(=O)[C@@H]2NCC3=CC(O)=CC=C3C2)=CC=CC(O)=C1 ZLVXBBHTMQJRSX-VMGNSXQWSA-N 0.000 description 1
- GLXDVVHUTZTUQK-UHFFFAOYSA-M lithium hydroxide monohydrate Substances [Li+].O.[OH-] GLXDVVHUTZTUQK-UHFFFAOYSA-M 0.000 description 1
- 229940040692 lithium hydroxide monohydrate Drugs 0.000 description 1
- 239000000203 mixture Substances 0.000 description 1
- XONPDZSGENTBNJ-UHFFFAOYSA-N molecular hydrogen;sodium Chemical compound [Na].[H][H] XONPDZSGENTBNJ-UHFFFAOYSA-N 0.000 description 1
- AJUXDFHPVZQOGF-UHFFFAOYSA-N n,n-dimethyl-1-naphthylamine Chemical compound C1=CC=C2C(N(C)C)=CC=CC2=C1 AJUXDFHPVZQOGF-UHFFFAOYSA-N 0.000 description 1
- 230000007935 neutral effect Effects 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- QQVIHTHCMHWDBS-UHFFFAOYSA-N perisophthalic acid Natural products OC(=O)C1=CC=CC(C(O)=O)=C1 QQVIHTHCMHWDBS-UHFFFAOYSA-N 0.000 description 1
- 239000012450 pharmaceutical intermediate Substances 0.000 description 1
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 1
- 239000000047 product Substances 0.000 description 1
- 230000035484 reaction time Effects 0.000 description 1
- 238000011084 recovery Methods 0.000 description 1
- 238000005204 segregation Methods 0.000 description 1
- 238000010898 silica gel chromatography Methods 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
Classifications
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P20/00—Technologies relating to chemical industry
- Y02P20/50—Improvements relating to the production of bulk chemicals
- Y02P20/55—Design of synthesis routes, e.g. reducing the use of auxiliary or protecting groups
Landscapes
- Nitrogen Condensed Heterocyclic Rings (AREA)
Abstract
Description
Claims (4)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2009102018600A CN102070526B (en) | 2009-11-24 | 2009-11-24 | Method for synthesizing 3-aza-bicyclo[4.1.0]heptane-6-formic acid with protective group |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2009102018600A CN102070526B (en) | 2009-11-24 | 2009-11-24 | Method for synthesizing 3-aza-bicyclo[4.1.0]heptane-6-formic acid with protective group |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN102070526A CN102070526A (en) | 2011-05-25 |
| CN102070526B true CN102070526B (en) | 2013-12-25 |
Family
ID=44029308
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN2009102018600A Active CN102070526B (en) | 2009-11-24 | 2009-11-24 | Method for synthesizing 3-aza-bicyclo[4.1.0]heptane-6-formic acid with protective group |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN102070526B (en) |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN104860870A (en) * | 2014-02-26 | 2015-08-26 | 欧美嘉股份有限公司 | Preparation method of piperidines with different substituents |
| CN104628644A (en) * | 2015-01-23 | 2015-05-20 | 常州大学 | 3-azabicyclo [4,1,0] heptyl aldehydes and preparation method thereof |
| CN110092753B (en) * | 2018-01-31 | 2022-04-26 | 南京药石科技股份有限公司 | A kind of preparation method of synthesizing 3-azabicyclo[4.1.0]heptane-2-carboxylic acid and its hydrochloride |
| CN109879797A (en) * | 2019-01-10 | 2019-06-14 | 安徽昊帆生物有限公司 | N- benzyl-tetrahydropyridines and preparation method thereof |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1198156A (en) * | 1995-09-28 | 1998-11-04 | 科研制药株式会社 | The preparation method of 4-methylene piperidine |
-
2009
- 2009-11-24 CN CN2009102018600A patent/CN102070526B/en active Active
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1198156A (en) * | 1995-09-28 | 1998-11-04 | 科研制药株式会社 | The preparation method of 4-methylene piperidine |
Also Published As
| Publication number | Publication date |
|---|---|
| CN102070526A (en) | 2011-05-25 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN106966947B (en) | A kind of preparation method of vildagliptin | |
| CN106146459B (en) | Preparation method of bilastine | |
| CN102070526B (en) | Method for synthesizing 3-aza-bicyclo[4.1.0]heptane-6-formic acid with protective group | |
| CN103724261A (en) | Novel industrial production method for hydroxychloroquine sulfate | |
| CN101486753A (en) | Novel method for synthesizing finasteroid | |
| CN107778223B (en) | Preparation method of betrixaban maleate | |
| CN109563023A (en) | The method for preparing substituted biphenyl | |
| CN115594639A (en) | A kind of Tegrazan key intermediate synthetic method | |
| CN103204801A (en) | Synthesis method for N-Boc-3-piperidone | |
| CN104860872A (en) | Bis-(3R,4R)-1-benzyl-N,4-dimethyl piperidin-3-amine L-di-p-toluyl tartrate synthesis method | |
| CN112479993A (en) | Synthetic method applied to KRAS inhibitor drug heterocyclic intermediate | |
| CN104837817B (en) | Synthetic route for preparation of 3-amino-piperidine compounds | |
| CN115536593A (en) | Scalable production method of 4-hydroxy-N,N,2-trimethylbenzimidazole-6-carboxamide | |
| CN104193638A (en) | Method for preparing (S)-2',6'-dimethyl tyrosine and derivative of (S)-2',6'-dimethyl tyrosine, and derivative | |
| WO2025140300A1 (en) | Synthesis method for 5,6-dihydroxyindole and intermediate thereof | |
| CN103073496A (en) | Method for preparing dextromethorphan | |
| CN102351869B (en) | Preparation method of high-purity methylnaltrexone bromide | |
| CN106748966A (en) | A kind of synthetic method of Ramipril key intermediate | |
| CN111100042B (en) | Preparation method of 2-methoxy-5-sulfonamide benzoic acid | |
| CN113264885B (en) | Synthesis of tolvaptan degradation derivative | |
| CN101182304A (en) | The preparation method of N-alkylpyrrolidine | |
| CN104177301A (en) | Preparation method of dexrazoxane | |
| CN103360323B (en) | Preparation method of triclabendazole | |
| CN101555223A (en) | Pirlimycin intermediate and preparation method thereof | |
| CN114105848B (en) | Preparation method of cis-D-hydroxyproline derivative |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| C41 | Transfer of patent application or patent right or utility model | ||
| TR01 | Transfer of patent right |
Effective date of registration: 20170220 Address after: 200131 Shanghai City, Pudong New Area Waigaoqiao Free Trade Zone Foote Road No. 288 Patentee after: Shanghai Yaoming Kangde New Medicine Development Co., Ltd. Patentee after: Yaomingkangde New Medicine Development Co., Ltd., Wuxi Patentee after: Shanghai SynTheAll Pharmaceutical Co., Ltd. Address before: 200131 Shanghai City, Pudong New Area Waigaoqiao Free Trade Zone Foote Road No. 288 Patentee before: Shanghai Yaoming Kangde New Medicine Development Co., Ltd. Patentee before: Yaomingkangde New Medicine Development Co., Ltd., Wuxi |
|
| CP01 | Change in the name or title of a patent holder | ||
| CP01 | Change in the name or title of a patent holder |
Address after: 200131 Shanghai City, Pudong New Area Waigaoqiao Free Trade Zone Foote Road No. 288 Co-patentee after: Wuxi pharmatechs Limited by Share Ltd Patentee after: Shanghai Yaoming Kangde New Medicine Development Co., Ltd. Co-patentee after: Shanghai SynTheAll Pharmaceutical Co., Ltd. Address before: 200131 Shanghai City, Pudong New Area Waigaoqiao Free Trade Zone Foote Road No. 288 Co-patentee before: Yaomingkangde New Medicine Development Co., Ltd., Wuxi Patentee before: Shanghai Yaoming Kangde New Medicine Development Co., Ltd. Co-patentee before: Shanghai SynTheAll Pharmaceutical Co., Ltd. |