CN101984960A - Rizatriptan benzoate capsule and preparation method thereof - Google Patents
Rizatriptan benzoate capsule and preparation method thereof Download PDFInfo
- Publication number
- CN101984960A CN101984960A CN 201010525054 CN201010525054A CN101984960A CN 101984960 A CN101984960 A CN 101984960A CN 201010525054 CN201010525054 CN 201010525054 CN 201010525054 A CN201010525054 A CN 201010525054A CN 101984960 A CN101984960 A CN 101984960A
- Authority
- CN
- China
- Prior art keywords
- capsule
- lizakuputan benzoate
- preparation
- starch
- microcrystalline cellulose
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Landscapes
- Medicinal Preparation (AREA)
Abstract
The invention discloses a medicinal preparation rizatriptan benzoate capsule for treating migraine and a preparation method thereof. Every 10000 capsules are prepared from the following formulation constituents: 29-250g of rizatriptan benzoate, 20-400g of microcrystalline cellulose, 220-1100g of starch, 100-400ml of 10-20% polyvinyl pyrrolidone ethanol solution and 4-30g of magnesium stearate. The invention also provides a preparation method of the rizatriptan benzoate capsule. The capsule of the invention covers the foreign taste of the main medicine; release and absorption of the medicine are accelerated; medical particles in the capsule are not punched or molten, thereby overcoming some defects of tablets; after the capsule is taken orally, the medicine is directly dispersed in the gastrointestinal fluid and is moistened by the gastrointestinal fluid, thus the effective surface area is large; and the medicine has higher dissolving rate in the gastrointestinal fluid, can be better absorbed and has high bioavailability.
Description
Technical field
The present invention relates to a kind of medicine and preparation method thereof, be specifically related to Lizakuputan benzoate capsule and preparation method thereof.
Background technology
Lizakuputan benzoate, its chemical name is: N, N-dimethyl-2-[5-(1,2,4-triazol-1-yl methyl)-1H-indol-3-yl] the ethamine benzoate, molecular formula is C
15H
19N
5C
7H
6O
2, molecular weight is 391.47.Rizatriptan is to the 5-HT of human body
1BAnd 5-HT
1DReceptor has the affinity of height.To other 5-HT
1Receptor and 5-HT
7Receptor affinity is lower, to 5-HT
2, 5-HT
3, epinephrine, DA, histamine, choline or BZ receptor do not have obvious activity.The brain of expanding during the exciting migraine of rizatriptan is outer, the 5-HT on intracranial vessel and the nervi trigeminus tip
1B/1D, cause intracranial vessel to shrink, suppress the release and the transmission of neuropeptide in the nervi trigeminus pain path, and bring into play its treatment migraine effect.
According to present disclosed patent documentation, as can be seen, the research of para Toluic Acid's rizatriptan preparation at present mainly concentrates on the rizatriptan benzoate tablet, as the Chinese patent publication number is that the patent of invention of CN1868472 discloses a kind of tablets of Lizakuputan benzoate, the tablets of Lizakuputan benzoate of invention, prepared with conventional method by Lizakuputan benzoate and adjuvant, raw material and consumption that wherein per 1000 tablets of Lizakuputan benzoate adopt are: Lizakuputan benzoate 1.453g to 21.795g, lactose 30.0g to 150.0g, microcrystalline Cellulose 10.0g to 100.0g, 20% polyvinylpyrrolidone alcoholic solution 10 is to 200ml, amylum pregelatinisatum 10.0g to 100.0g, magnesium stearate 1.0g to 5.0g.
Summary of the invention
The object of the present invention is to provide the migrainous medicine Lizakuputan benzoate of a kind of quick curing capsule.
The present invention is achieved in that
A kind of Lizakuputan benzoate capsule is characterized in that the following constituent prescription of every preparation 10000 capsules needs:
Lizakuputan benzoate 29 is to 250g
Microcrystalline Cellulose 20 is to 400g
Starch 220 is to 1100g
10 to 20% polyvinylpyrrolidone alcoholic solution 100 are to 400ml
Magnesium stearate 4 is to 30g
The present invention also provides a kind of Lizakuputan benzoate capsular preparation method, comprises the steps:
Step 1: Lizakuputan benzoate, microcrystalline Cellulose, starch and magnesium stearate pulverize separately are crossed 100 mesh sieves, standby;
Step 2: take by weighing raw material Lizakuputan benzoate and microcrystalline cellulose excipients, starch and magnesium stearate according to the mentioned component prescription;
Step 3: preparation binding agent: polyvinylpyrrolidone is joined in 80 to 98% ethanol, stir and to make dissolving fully, make concentration and be 10 to 20% polyvinylpyrrolidone alcoholic solution, it is standby to make binding agent;
Step 4:, and, be prepared into soft material to wherein adding binding agent totally 100 to 400ml with Lizakuputan benzoate, microcrystalline Cellulose and the starch equivalent mix homogeneously that progressively increases;
Step 5: soft material is granulated with 20 mesh sieves;
Step 6: place baking oven dry wet granular, the control temperature was dried 1 hour at 55 to 75 ℃, crossed 18 mesh sieve granulate;
Step 7: in dried granule, add magnesium stearate, mix homogeneously;
Step 8: through after the inspection of semifinished product, filled capsules promptly makes Lizakuputan benzoate capsule of the present invention.
In the present invention, Lizakuputan benzoate is the principal agent in the present invention's prescription, and microcrystalline Cellulose is as disintegrating agent, and starch is as diluent and filler, and magnesium stearate is as lubricant, and the polyvinylpyrrolidone alcoholic solution is as binding agent.The present invention selects for use common starch as diluent and filler, and microcrystalline Cellulose is as disintegrating agent, and dissolution is good, and the granule of preparation is loose mobile strong, helps the quick performance of drug effect.Capsule of the present invention can be covered the abnormal flavour of principal agent; Can quicken the release and the absorption of medicine, drug particles in the capsule is not subjected to punching press or fusing, some defectives of tablet have been overcome, oral back medicine directly is scattered in the gastro-intestinal Fluid, medicine is by the gastro-intestinal Fluid moistening, and effective surface area is big, and the speed that medicine is dissolved in gastro-intestinal Fluid is bigger, absorb better, the bioavailability height is rapid-action.In addition, Lizakuputan benzoate capsule output height, the cost of the present invention's preparation are low; Divided dose accurately, carry with easy to use; Physical and chemical properties of drugs is more stable, storage period is longer.
Quality standard test method of the present invention:
(1) related substance
It is an amount of to get capsule 's content, adds mobile phase dissolving and dilution and makes the solution that contains 0.5mg among every 1ml, filters, and gets subsequent filtrate as need testing solution.Precision is measured in right amount, adds mobile phase and makes the solution that contains 5 μ g among every 1ml, solution in contrast.According to the chromatographic condition under the assay item, get contrast solution 20 μ l and inject chromatograph of liquid, regulate detection sensitivity, the peak height that makes the rizatriptan chromatographic peak is 20% of a full scale.Precision is measured need testing solution and each 20 μ l of contrast solution again, inject chromatograph of liquid respectively, the record chromatogram is to 3 times of rizatriptan peak retention time, in the need testing solution chromatogram as show impurity peaks (being about except the adjuvant peak before 0.44) with respect to the main peak retention time, measure each impurity peak area sum, must not be greater than contrast solution rizatriptan and two main peak areas of benzoic acid sum (1.0%).
(2) uniformity of dosage units
Get 1 of capsule, in content impouring 50ml measuring bottle, softgel shell is cleaned with the mobile phase gradation, and washing liquid is incorporated in the measuring bottle, it is an amount of to add mobile phase, after ultrasonic 10 minutes, puts and is chilled to room temperature, adds mobile phase and is diluted to scale, shake up, filter, get subsequent filtrate as need testing solution with the 0.8um filter membrane; According to the method under the assay item, white " it is an amount of that precision takes by weighing the Lizakuputan benzoate reference substance in addition " rises, and measures in accordance with the law, should (two appendix XE of Chinese Pharmacopoeia version in 2005) up to specification.
(3) dissolution
Getting this product, according to dissolution method (two appendix XC of Chinese Pharmacopoeia version in 2005 three therapeutic methods of traditional Chinese medicine), is solvent with 250ml hydrochloric acid solution (9 → 1000), rotating speed is that per minute 75 changes, and operation in accordance with the law is in the time of 30 minutes, sampling 10ml filters, and gets subsequent filtrate as need testing solution.It is an amount of that other gets the Lizakuputan benzoate reference substance, accurate claims surely, adds hydrochloric acid solution (9 → 1000) and dissolve and make the solution that contains 30 μ g among every 1ml, product solution in contrast.Get above-mentioned two kinds of solution, measure trap respectively at wavelength place, 280nm place according to spectrophotography (two appendix IV of Chinese Pharmacopoeia version in 2005 A).By the stripping quantity of every of the ratio calculation of the two trap, and result of calculation and 0.6880 multiplied each other, limit is 75% of a labelled amount, should be up to specification.
(4) assay
Measure according to high performance liquid chromatography (two appendix V of Chinese Pharmacopoeia version in 2005 D).
Chromatographic condition and system suitability test are filler with octadecylsilane chemically bonded silica, (get sodium heptanesulfonate 4.045g with acetonitrile-heptanesulfonic acid sodium solution, adding water dissolves in right amount, add phosphoric acid 4ml, add acetic acid 8ml, thin up is to 1000ml, regulates pH value to 3.0 with triethylamine, shakes up) (20: 80) be mobile phase; The detection wavelength is 280nm.Number of theoretical plate calculates by the rizatriptan peak should be not less than 1500.
Algoscopy is got 20 of this product, and accurate the title decides, with the content mixing, precision takes by weighing content an amount of (being equivalent to rizatriptan 25mg approximately), puts in the 50ml measuring bottle, and it is an amount of to add mobile phase, after ultrasonic 10 minutes, put and be chilled to room temperature, add mobile phase and be diluted to scale, shake up, filter with the 0.8um filter membrane, precision is measured subsequent filtrate 5ml, put in the 25ml measuring bottle, add mobile phase and be diluted to scale, shake up, as need testing solution; It is an amount of that precision takes by weighing the Lizakuputan benzoate reference substance in addition, makes the solution that contains rizatriptan 0.1mg among every 1ml with mobile phase, in contrast product solution.Get each 20 μ l of above-mentioned solution respectively, inject chromatograph of liquid, the record chromatogram., and result of calculation and 0.6880 multiplied each other promptly to get supply to contain C in the examination amount with calculated by peak area by external standard method
15H
19N
5Weight.
The specific embodiment
Embodiment 1:
A kind of Lizakuputan benzoate capsule (specification: the 5mg/ grain, by rizatriptan), the prescription that every preparation 10000 capsules need is as follows:
| Lizakuputan benzoate | 72.65g |
| Microcrystalline Cellulose | 200g |
| Starch | 670g |
| 15% polyvinylpyrrolidone alcoholic solution | 250ml |
| Magnesium stearate | 8g |
The capsular preparation method of Lizakuputan benzoate comprises the steps:
Step 1: Lizakuputan benzoate, microcrystalline Cellulose, starch and magnesium stearate pulverize separately are crossed 100 mesh sieves, standby;
Step 2: take by weighing raw material Lizakuputan benzoate and microcrystalline cellulose excipients, starch and magnesium stearate according to the mentioned component prescription;
Step 3: the preparation of binding agent: polyvinylpyrrolidone is joined in 80 to 98% ethanol, stir and to make dissolving fully, make concentration and be 15% polyvinylpyrrolidone alcoholic solution, standby;
Step 4: adopt the equivalent method of progressively increasing to make its mix homogeneously Lizakuputan benzoate, microcrystalline Cellulose and starch, and add binding agent 250ml altogether, be prepared into soft material to gradation wherein;
Step 5: soft material is granulated with 20 mesh sieves;
Step 6: place baking oven dry wet granular, the control temperature is dried 1 hour qualified back of moisture content and is crossed 18 mesh sieve granulate at 60 ℃;
Step 7: in dried granule, add the recipe quantity magnesium stearate and mix mix homogeneously;
Step 8: through after the inspection of semifinished product, filled capsules promptly makes Lizakuputan benzoate capsule of the present invention.
Embodiment 2:
A kind of Lizakuputan benzoate capsule (specification: the 5mg/ grain, by rizatriptan.), the prescription that every preparation 10000 capsules need is as follows:
| Lizakuputan benzoate | 72.65g |
| Microcrystalline Cellulose | 300g |
| Starch | 700g |
| 15% polyvinylpyrrolidone alcoholic solution | 300ml |
| Magnesium stearate | 10g |
The capsular preparation method of Lizakuputan benzoate comprises the steps:
Step 1: Lizakuputan benzoate, microcrystalline Cellulose, starch and magnesium stearate pulverize separately are crossed 100 mesh sieves, standby;
Step 2: take by weighing raw material Lizakuputan benzoate and microcrystalline cellulose excipients, starch and magnesium stearate according to the mentioned component prescription;
Step 3: preparation binding agent: polyvinylpyrrolidone 45g joined in 95% ethanol make into 300ml, stir and make dissolving fully, make concentration and be 15% polyvinylpyrrolidone alcoholic solution, standby;
Step 4: with Lizakuputan benzoate, microcrystalline Cellulose and starch, take equivalent to progressively increase after mixing method makes it mix all, add binding agent 300ml altogether, be prepared into suitable soft material;
Step 5: soft material is granulated with 20 mesh sieves;
Step 6:, cross 18 mesh sieve granulate with wet granular under 60 to 75 ℃ condition dry 1 hour;
Step 7: in dried granule, add the recipe quantity magnesium stearate and mix mix homogeneously;
Step 8: through after the inspection of semifinished product, carry out capsule and fill, carry out inner packing, promptly make Lizakuputan benzoate capsule of the present invention through after the assay was approved.
Embodiment 3:
A kind of Lizakuputan benzoate capsule (specification: the 10mg/ grain, by rizatriptan.), the prescription that every preparation 10000 capsules need is as follows:
| Lizakuputan benzoate | 145.3g |
| Microcrystalline Cellulose | 400g |
| Starch | 800g |
| 15% polyvinylpyrrolidone alcoholic solution | 500ml |
| Magnesium stearate | 8g |
The capsular preparation method of Lizakuputan benzoate comprises the steps:
Step 1: Lizakuputan benzoate, microcrystalline Cellulose, starch and magnesium stearate pulverize separately are crossed 100 mesh sieves, standby;
Step 2: take by weighing raw material Lizakuputan benzoate and microcrystalline cellulose excipients, starch and magnesium stearate according to the mentioned component prescription;
Step 3: preparation binding agent: polyvinylpyrrolidone 75g joined in 95% ethanol make into 500ml, stir and make dissolving fully, make concentration and be 15% polyvinylpyrrolidone alcoholic solution, standby;
Step 4: with Lizakuputan benzoate, microcrystalline Cellulose and starch, take equivalent to progressively increase after mixing method makes it mix all, add binding agent 500ml altogether, be prepared into suitable soft material;
Step 5: soft material is granulated with 20 mesh sieves;
Step 6:, cross 18 mesh sieve granulate with wet granular under 60 to 75 ℃ condition dry 1 hour;
Step 7: in dried granule, add recipe quantity magnesium stearate, mix homogeneously;
Step 8: through after the inspection of semifinished product, carry out capsule and fill, carry out inner packing, promptly make Lizakuputan benzoate capsule of the present invention through after the assay was approved.
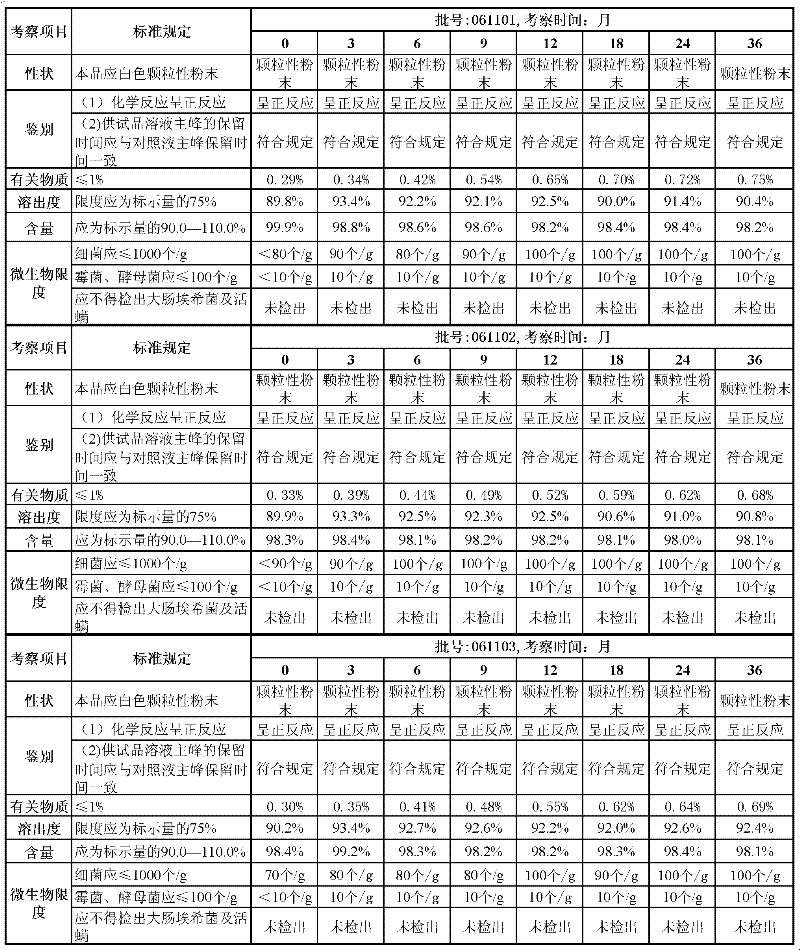
For essence of the present invention better is described, will illustrate that this capsule physicochemical property is stable, effect duration is long with the capsular long-term stable experiment of the prepared Lizakuputan benzoate of the present invention below.
Get three batches of the Lizakuputan benzoate capsules that embodiment one makes, lot number is respectively: 061101,061102,061103.
Reference substance Lizakuputan benzoate (press dry product and calculate, content is 99.8%), lot number: 200501, provide by Nat'l Pharmaceutical ﹠ Biological Products Control Institute.
Reagent acetonitrile (chromatographically pure): TEDIA; Sodium heptanesulfonate: the Tokyo changes into Industrial Co., Ltd; Phosphoric acid (AR): Chengdu chemical reagent factory; Acetic acid (AR): Chengdu chemical reagent factory; Triethylamine (AR): Chongqing, Shen, Chongqing chemical reagent factory; Redistilled water: self-control.
Instrument 756 ultraviolet-uisible spectrophotometers (limited company of Shanghai spectral instrument), the intelligent dissolution test instrument of ZRS-8G (Radio Factory of Tianjin Univ.); LC-10AT type high performance liquid chromatograph (day island proper Feng), SPD-10AVP type detector, N2000 two pass chromatographic work station; BP211D type electronic balance (German Sai Duolisi)
Investigation project: character, discriminating, dissolution, related substance, sign content, limit test of microbe.
Test basis: " national drug standards " (trying) YBH06812006.
Content of the test and condition
Plastic-aluminum aluminum packing, long-term stable experiment under the normal temperature condition (0 ℃ to 30 ℃ of temperature, relative humidity 60% ± 15%).
Long-term stable experiment the results are shown in Table 1.
Table 1 Lizakuputan benzoate capsule long-term stable experiment result:
Conclusion: place under the condition of 0 ℃ to 30 ℃ at normal temperatures of Lizakuputan benzoate capsule, relative humidity 60% ± 15%, respectively at getting the check that keeps sample 3rd month, 6 months, 9 months, 12 months, 18 months, 24 months, 36 the end of month, result and comparison in 0 month, removing related substance slightly increases, other every indexs have no significant change, and related substance is still in the scope of standard-required, and the long-term stable experiment result shows that the Lizakuputan benzoate capsule stores 36 months its steady qualities under normal temperature condition.
Claims (2)
1. Lizakuputan benzoate capsule is characterized in that the following constituent prescription of every preparation 10000 capsules needs:
Lizakuputan benzoate 29 is to 250g
Microcrystalline Cellulose 20 is to 400g
Starch 220 is to 1100g
10 to 20% polyvinylpyrrolidone alcoholic solution 100 are to 400ml
Magnesium stearate 4 is to 30g.
2. the capsular preparation method of Lizakuputan benzoate as claimed in claim 1 is characterized in that comprising the steps:
Step 1: Lizakuputan benzoate, microcrystalline Cellulose, starch and magnesium stearate pulverize separately are crossed 100 mesh sieves, standby;
Step 2: take by weighing raw material Lizakuputan benzoate and microcrystalline cellulose excipients, starch and magnesium stearate according to the mentioned component prescription;
Step 3: preparation binding agent: polyvinylpyrrolidone is joined in 80 to 98% ethanol, stir and to make dissolving fully, make concentration and be 10 to 20% polyvinylpyrrolidone alcoholic solution, it is standby to make binding agent;
Step 4: with Lizakuputan benzoate, microcrystalline Cellulose and starch mix homogeneously, and add binding agent 250ml altogether, be prepared into soft material to gradation wherein;
Step 5: soft material is granulated with 20 mesh sieves;
Step 6: wet granular placed under 55 to 75 ℃ of conditions dry, dry 1 hour, cross 18 mesh sieve granulate;
Step 7: in dried granule, add magnesium stearate, mix homogeneously;
Step 8: through after the inspection of semifinished product, filled capsules promptly makes Lizakuputan benzoate capsule of the present invention.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2010105250541A CN101984960B (en) | 2010-10-29 | 2010-10-29 | Rizatriptan benzoate capsule and preparation method thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2010105250541A CN101984960B (en) | 2010-10-29 | 2010-10-29 | Rizatriptan benzoate capsule and preparation method thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN101984960A true CN101984960A (en) | 2011-03-16 |
| CN101984960B CN101984960B (en) | 2011-10-26 |
Family
ID=43709392
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN2010105250541A Active CN101984960B (en) | 2010-10-29 | 2010-10-29 | Rizatriptan benzoate capsule and preparation method thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN101984960B (en) |
Cited By (59)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN106721084A (en) * | 2017-03-13 | 2017-05-31 | 山东大学 | A kind of measure herds special C32 spansule of feed intake and preparation method thereof |
| WO2020010196A1 (en) * | 2018-07-03 | 2020-01-09 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10583144B2 (en) | 2017-01-04 | 2020-03-10 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10653777B2 (en) | 2015-02-10 | 2020-05-19 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10688102B2 (en) | 2017-06-29 | 2020-06-23 | Axsome Therapeutics, Inc. | Combination treatment for migraine and other pain |
| US10688185B2 (en) | 2017-06-29 | 2020-06-23 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10695429B2 (en) | 2015-02-10 | 2020-06-30 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10695430B2 (en) | 2015-02-10 | 2020-06-30 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10702535B2 (en) | 2017-01-04 | 2020-07-07 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10702602B2 (en) | 2015-02-10 | 2020-07-07 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10722583B2 (en) | 2015-02-10 | 2020-07-28 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10729696B2 (en) | 2017-01-04 | 2020-08-04 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10729774B1 (en) | 2015-02-10 | 2020-08-04 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10729773B2 (en) | 2015-02-10 | 2020-08-04 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10729697B2 (en) | 2017-01-04 | 2020-08-04 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10758617B2 (en) | 2017-06-29 | 2020-09-01 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10758618B2 (en) | 2015-02-10 | 2020-09-01 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10780165B2 (en) | 2015-02-10 | 2020-09-22 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10780166B2 (en) | 2015-02-10 | 2020-09-22 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10799588B2 (en) | 2015-02-10 | 2020-10-13 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10821181B2 (en) | 2015-02-10 | 2020-11-03 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10821182B2 (en) | 2017-06-29 | 2020-11-03 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10894053B2 (en) | 2017-01-04 | 2021-01-19 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10918722B2 (en) | 2017-06-29 | 2021-02-16 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10933136B2 (en) | 2015-02-10 | 2021-03-02 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10933137B2 (en) | 2015-02-10 | 2021-03-02 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10940153B2 (en) | 2017-01-04 | 2021-03-09 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10987358B2 (en) | 2017-06-29 | 2021-04-27 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11013805B2 (en) | 2015-02-10 | 2021-05-25 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11013806B2 (en) | 2015-02-10 | 2021-05-25 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11045549B2 (en) | 2015-02-10 | 2021-06-29 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11077117B2 (en) | 2017-01-04 | 2021-08-03 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11110173B2 (en) | 2015-02-10 | 2021-09-07 | Axsome Therapeutics, Inc | Pharmaceutical compositions comprising meloxicam |
| US11185550B2 (en) | 2017-06-29 | 2021-11-30 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11207327B2 (en) | 2017-01-04 | 2021-12-28 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11219626B2 (en) | 2017-06-29 | 2022-01-11 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11266657B2 (en) | 2017-01-04 | 2022-03-08 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| CN114689758A (en) * | 2022-04-13 | 2022-07-01 | 南通市食品药品监督检验中心 | Concentration, extraction and determination method of sorbic acid and benzoic acid in gelatin empty capsules |
| US11433078B2 (en) | 2017-01-04 | 2022-09-06 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11433079B2 (en) | 2017-01-04 | 2022-09-06 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11471465B2 (en) | 2017-01-04 | 2022-10-18 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11510927B2 (en) | 2017-06-29 | 2022-11-29 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11602563B2 (en) | 2015-02-10 | 2023-03-14 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11607456B2 (en) | 2015-02-10 | 2023-03-21 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11617756B2 (en) | 2017-06-29 | 2023-04-04 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11617755B2 (en) | 2017-01-04 | 2023-04-04 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11617791B2 (en) | 2017-06-29 | 2023-04-04 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11738085B2 (en) | 2015-02-10 | 2023-08-29 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11759522B2 (en) | 2017-06-29 | 2023-09-19 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11801250B2 (en) | 2017-01-04 | 2023-10-31 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11806354B2 (en) | 2017-01-04 | 2023-11-07 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11865117B2 (en) | 2017-06-29 | 2024-01-09 | Axsome Therapeutics, Inc | Pharmaceutical compositions comprising meloxicam |
| US11998552B2 (en) | 2020-12-31 | 2024-06-04 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US12005118B2 (en) | 2022-05-19 | 2024-06-11 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US12128052B2 (en) | 2020-05-28 | 2024-10-29 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US12268693B2 (en) | 2020-04-06 | 2025-04-08 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising a tolfenamic acid |
| US12370196B2 (en) | 2020-12-31 | 2025-07-29 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US12485176B2 (en) | 2015-02-10 | 2025-12-02 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US12544383B1 (en) | 2025-03-10 | 2026-02-10 | Axsome Therapeutics, Inc. | Methods of treating migraines with a combination of a meloxicam and a rizatriptan |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1868472A (en) * | 2006-06-29 | 2006-11-29 | 朱光荣 | Tablets of Lizakuputan benzoate |
| CN101757623A (en) * | 2008-10-09 | 2010-06-30 | 北京德众万全药物技术开发有限公司 | 5-HT receptor agonist solid pharmaceutical composition |
-
2010
- 2010-10-29 CN CN2010105250541A patent/CN101984960B/en active Active
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1868472A (en) * | 2006-06-29 | 2006-11-29 | 朱光荣 | Tablets of Lizakuputan benzoate |
| CN101757623A (en) * | 2008-10-09 | 2010-06-30 | 北京德众万全药物技术开发有限公司 | 5-HT receptor agonist solid pharmaceutical composition |
Cited By (87)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11013805B2 (en) | 2015-02-10 | 2021-05-25 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US12485176B2 (en) | 2015-02-10 | 2025-12-02 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11738085B2 (en) | 2015-02-10 | 2023-08-29 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10653777B2 (en) | 2015-02-10 | 2020-05-19 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11607456B2 (en) | 2015-02-10 | 2023-03-21 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11602563B2 (en) | 2015-02-10 | 2023-03-14 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10695429B2 (en) | 2015-02-10 | 2020-06-30 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10695430B2 (en) | 2015-02-10 | 2020-06-30 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11369684B2 (en) | 2015-02-10 | 2022-06-28 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10702602B2 (en) | 2015-02-10 | 2020-07-07 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10722583B2 (en) | 2015-02-10 | 2020-07-28 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11357854B2 (en) | 2015-02-10 | 2022-06-14 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10729774B1 (en) | 2015-02-10 | 2020-08-04 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10729773B2 (en) | 2015-02-10 | 2020-08-04 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11285213B2 (en) | 2015-02-10 | 2022-03-29 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11285214B2 (en) | 2015-02-10 | 2022-03-29 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10758618B2 (en) | 2015-02-10 | 2020-09-01 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10780165B2 (en) | 2015-02-10 | 2020-09-22 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10780166B2 (en) | 2015-02-10 | 2020-09-22 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10799588B2 (en) | 2015-02-10 | 2020-10-13 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10821181B2 (en) | 2015-02-10 | 2020-11-03 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11129895B2 (en) | 2015-02-10 | 2021-09-28 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11123431B2 (en) | 2015-02-10 | 2021-09-21 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11110173B2 (en) | 2015-02-10 | 2021-09-07 | Axsome Therapeutics, Inc | Pharmaceutical compositions comprising meloxicam |
| US11045549B2 (en) | 2015-02-10 | 2021-06-29 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10933136B2 (en) | 2015-02-10 | 2021-03-02 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10933137B2 (en) | 2015-02-10 | 2021-03-02 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11013806B2 (en) | 2015-02-10 | 2021-05-25 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11266657B2 (en) | 2017-01-04 | 2022-03-08 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11207327B2 (en) | 2017-01-04 | 2021-12-28 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10940153B2 (en) | 2017-01-04 | 2021-03-09 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11826354B2 (en) | 2017-01-04 | 2023-11-28 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11806354B2 (en) | 2017-01-04 | 2023-11-07 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11077117B2 (en) | 2017-01-04 | 2021-08-03 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11801250B2 (en) | 2017-01-04 | 2023-10-31 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10894053B2 (en) | 2017-01-04 | 2021-01-19 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10583144B2 (en) | 2017-01-04 | 2020-03-10 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10702535B2 (en) | 2017-01-04 | 2020-07-07 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11628173B2 (en) | 2017-01-04 | 2023-04-18 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11426414B2 (en) | 2017-01-04 | 2022-08-30 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11207328B2 (en) | 2017-01-04 | 2021-12-28 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11617755B2 (en) | 2017-01-04 | 2023-04-04 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10729696B2 (en) | 2017-01-04 | 2020-08-04 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11471465B2 (en) | 2017-01-04 | 2022-10-18 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11433079B2 (en) | 2017-01-04 | 2022-09-06 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10729697B2 (en) | 2017-01-04 | 2020-08-04 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11433078B2 (en) | 2017-01-04 | 2022-09-06 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| CN106721084A (en) * | 2017-03-13 | 2017-05-31 | 山东大学 | A kind of measure herds special C32 spansule of feed intake and preparation method thereof |
| CN106721084B (en) * | 2017-03-13 | 2023-08-25 | 山东大学 | A special C32 slow-release capsule for measuring grazing feed intake and its preparation method |
| US11571428B2 (en) | 2017-06-29 | 2023-02-07 | Axsome Therapeutics, Inc | Pharmaceutical compositions comprising meloxicam |
| US11617791B2 (en) | 2017-06-29 | 2023-04-04 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11331323B2 (en) | 2017-06-29 | 2022-05-17 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11285215B2 (en) | 2017-06-29 | 2022-03-29 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10758617B2 (en) | 2017-06-29 | 2020-09-01 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11471464B2 (en) | 2017-06-29 | 2022-10-18 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11504429B2 (en) | 2017-06-29 | 2022-11-22 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11510927B2 (en) | 2017-06-29 | 2022-11-29 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10987358B2 (en) | 2017-06-29 | 2021-04-27 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10688185B2 (en) | 2017-06-29 | 2020-06-23 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10688102B2 (en) | 2017-06-29 | 2020-06-23 | Axsome Therapeutics, Inc. | Combination treatment for migraine and other pain |
| US11617756B2 (en) | 2017-06-29 | 2023-04-04 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11219626B2 (en) | 2017-06-29 | 2022-01-11 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US12472258B2 (en) | 2017-06-29 | 2025-11-18 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11185550B2 (en) | 2017-06-29 | 2021-11-30 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11135295B2 (en) | 2017-06-29 | 2021-10-05 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10821182B2 (en) | 2017-06-29 | 2020-11-03 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11759522B2 (en) | 2017-06-29 | 2023-09-19 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US10918722B2 (en) | 2017-06-29 | 2021-02-16 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US12478678B2 (en) | 2017-06-29 | 2025-11-25 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11826370B2 (en) | 2017-06-29 | 2023-11-28 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11020483B2 (en) | 2017-06-29 | 2021-06-01 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11865117B2 (en) | 2017-06-29 | 2024-01-09 | Axsome Therapeutics, Inc | Pharmaceutical compositions comprising meloxicam |
| US12472256B2 (en) | 2017-06-29 | 2025-11-18 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11944683B2 (en) | 2017-06-29 | 2024-04-02 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US12472255B2 (en) | 2017-06-29 | 2025-11-18 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US12472257B2 (en) | 2017-06-29 | 2025-11-18 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| CN112384198A (en) * | 2018-07-03 | 2021-02-19 | 艾克萨姆治疗公司 | Pharmaceutical composition comprising meloxicam |
| WO2020010196A1 (en) * | 2018-07-03 | 2020-01-09 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US12268693B2 (en) | 2020-04-06 | 2025-04-08 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising a tolfenamic acid |
| US12357640B2 (en) | 2020-05-28 | 2025-07-15 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US12128052B2 (en) | 2020-05-28 | 2024-10-29 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US12370196B2 (en) | 2020-12-31 | 2025-07-29 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US11998552B2 (en) | 2020-12-31 | 2024-06-04 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| CN114689758B (en) * | 2022-04-13 | 2024-02-20 | 南通市食品药品监督检验中心 | Method for concentrating, extracting and measuring sorbic acid and benzoic acid in gelatin hollow capsules |
| CN114689758A (en) * | 2022-04-13 | 2022-07-01 | 南通市食品药品监督检验中心 | Concentration, extraction and determination method of sorbic acid and benzoic acid in gelatin empty capsules |
| US12005118B2 (en) | 2022-05-19 | 2024-06-11 | Axsome Therapeutics, Inc. | Pharmaceutical compositions comprising meloxicam |
| US12544383B1 (en) | 2025-03-10 | 2026-02-10 | Axsome Therapeutics, Inc. | Methods of treating migraines with a combination of a meloxicam and a rizatriptan |
Also Published As
| Publication number | Publication date |
|---|---|
| CN101984960B (en) | 2011-10-26 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN101984960B (en) | Rizatriptan benzoate capsule and preparation method thereof | |
| CN102266307B (en) | Valsartan capsules and preparation method thereof | |
| CN103181923B (en) | Pharmaceutical preparation comprising Repaglinide and preparation method thereof | |
| CN104876947B (en) | Cefetamet Pivoxil Hydrochloride hydrate crystal and its dispersible tablet | |
| CN103463120B (en) | A kind of stable type compound vitamin B tablet and preparation method thereof | |
| AU2013346363A1 (en) | Solid dosage form comprising micronized cytisine | |
| CN102349877A (en) | Hydroxychloroquine sulfate capsule, hydroxychloroquine sulfate dispersible tablet and application thereof | |
| CN117054548A (en) | In-vitro dissolution detection method of candesartan cilexetil tablet | |
| KR20120130761A (en) | Solid pharmaceutical formulations of ramipril and amlodipine besylate, and their preparation | |
| CN102600149B (en) | Pharmaceutical composition for treating diabetes | |
| CN100496487C (en) | Mitiglinide preparation and preparing method | |
| CN104721158A (en) | Stable everolimus tablet | |
| CN100546572C (en) | Clindamycin palmitate soft capsules and preparation method thereof | |
| CN102716132A (en) | Compound amlodipine/valsartan/hydrochlorothiazide tablets and method for making the same | |
| CN103948564B (en) | A kind of lyrica capsule and preparation method thereof | |
| CN101342146A (en) | Preparation method of glimepiride tablet | |
| CN106551927A (en) | Pharmaceutical composition comprising vildagliptin and metformin hydrochloride and preparation method thereof | |
| CN108295035A (en) | Procaterol Hydrochloride piece and preparation method thereof | |
| Al Naimi et al. | Comparative in-vitro pharmaceutical evaluation of four brands of metronidazole tablets marketed in gulf region | |
| CN108379235B (en) | Tacrolimus sustained-release tablet composition capable of rapidly disintegrating | |
| CN101874792A (en) | Phloroglucinol oral disintegrating tablet and preparation method thereof | |
| CN101732261A (en) | Stable type compound pellet, preparation method thereof and capsule containing same | |
| CN103893185B (en) | A kind of valsartan and Hydrochlorothiade dispersible tablet and preparation method thereof | |
| CN101474167A (en) | Preparation method of estradiol capsule for vagina | |
| CN100560069C (en) | A kind of mitiglinide calcium preparation and detection method thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| C56 | Change in the name or address of the patentee | ||
| CP03 | Change of name, title or address |
Address after: 641000 No. 456, Anji street, Neijiang economic and Technological Development Zone, Sichuan Patentee after: Sichuan zitonggong pharmaceutical Limited by Share Ltd Address before: Neijiang City, Sichuan province 641001 city music town Vitex dam Patentee before: Sichuan Zitonggong Pharmaceutical Co., Ltd. |