CN101983098A - Systems and methods for processing CO2 - Google Patents
Systems and methods for processing CO2 Download PDFInfo
- Publication number
- CN101983098A CN101983098A CN2010800009519A CN201080000951A CN101983098A CN 101983098 A CN101983098 A CN 101983098A CN 2010800009519 A CN2010800009519 A CN 2010800009519A CN 201080000951 A CN201080000951 A CN 201080000951A CN 101983098 A CN101983098 A CN 101983098A
- Authority
- CN
- China
- Prior art keywords
- gas
- liquid
- solution
- water
- industrial
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Images
Classifications
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02A—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE
- Y02A50/00—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE in human health protection, e.g. against extreme weather
- Y02A50/20—Air quality improvement or preservation, e.g. vehicle emission control or emission reduction by using catalytic converters
Landscapes
- Treating Waste Gases (AREA)
- Gas Separation By Absorption (AREA)
Abstract
提供了降低二氧化碳及其他大气污染物的水平的系统和方法。还讨论了能够从气态废物物流中除去大量二氧化碳及其他大气污染物并且以存储稳定的形成埋存它们的经济可行的系统和方法。
Systems and methods for reducing the levels of carbon dioxide and other atmospheric pollutants are provided. Economically viable systems and methods for removing large quantities of carbon dioxide and other atmospheric pollutants from gaseous waste streams and storing them in a storage-stable form are also discussed.
Description
交叉引用cross reference
本申请要求以下申请的权益,其每个全盘引入本文作为参考。This application claims the benefit of the following applications, each of which is incorporated herein by reference in its entirety.
美国临时专利申请61/158,992,申请日2009年3月10日,题目为Liquid Absorption of Gaseous Pollutants Using a Flat Jet Reactor″;U.S. provisional patent application 61/158,992, dated March 10, 2009, titled "Liquid Absorption of Gaseous Pollutants Using a Flat Jet Reactor";
美国临时专利申请61/168,166,申请日2009年4月9日,题目为Apparatus,Systems,and Methods for Treating Industrial Waste Gases″;U.S. provisional patent application 61/168,166, filed April 9, 2009, entitled "Apparatus, Systems, and Methods for Treating Industrial Waste Gases";
美国临时专利申请61/178,475,申请日2009年5月14日,题目为″Apparatus,Systems,and Methods for Treating Industrial Waste Gases″;U.S. provisional patent application 61/178,475, dated May 14, 2009, titled "Apparatus, Systems, and Methods for Treating Industrial Waste Gases";
美国临时专利申请61/228,210,2009年7月24日,题目为″Apparatus,Systems,and Methods for Treating Industrial Waste Gases″;U.S. Provisional Patent Application 61/228,210, July 24, 2009, entitled "Apparatus, Systems, and Methods for Treating Industrial Waste Gases";
美国临时专利申请61/230,042,申请日2009年7月30日,题目为″Apparatus,Systems,and Methods for Treating Industrial Waste Gases″;U.S. provisional patent application 61/230,042, dated July 30, 2009, titled "Apparatus, Systems, and Methods for Treating Industrial Waste Gases";
美国临时专利申请61/239,429,2009年9月2日,题目为″Apparatus,Systems,and Methods for Treating Industrial Waste Gases″;U.S. Provisional Patent Application 61/239,429, September 2, 2009, entitled "Apparatus, Systems, and Methods for Treating Industrial Waste Gases";
美国临时专利申请61/178,360,2009年5月14日,题目为″Methodsand Apparatus for Contacting Gas and Liquid″;U.S. Provisional Patent Application 61/178,360, May 14, 2009, entitled "Methods and Apparatus for Contacting Gas and Liquid";
美国临时专利申请61/221,457,申请日2009年6月29日,题目为″Gas-Liquid-Solid Contactor and Precipitator:Apparatus and Methods″;U.S. provisional patent application 61/221,457, filed on June 29, 2009, entitled "Gas-Liquid-Solid Contactor and Precipitator: Apparatus and Methods";
美国临时专利申请61/221,631,申请日2009年6月30日,题目为″Gas,Liquid,Solid Contacting:Methods and Apparatus″;U.S. provisional patent application 61/221,631, filed on June 30, 2009, entitled "Gas, Liquid, Solid Contacting: Methods and Apparatus";
美国临时专利申请61/223,657,2009年7月7日,题目为″Gas,Liquid,Solid Contacting:Methods and Apparatus″;U.S. Provisional Patent Application 61/223,657, July 7, 2009, entitled "Gas, Liquid, Solid Contacting: Methods and Apparatus";
美国临时专利申请61/289,657,申请日2009年12月23日,题目为″Gas,Liquid,Solid Contacting:Methods and Apparatus″;U.S. provisional patent application 61/289,657, filed on December 23, 2009, entitled "Gas, Liquid, Solid Contacting: Methods and Apparatus";
美国临时专利申请61/306,412,申请日2010年2月19日,题目为“Apparatus,Systems,And Methods For Treating Industrial Waste Gases″;和U.S. Provisional Patent Application 61/306,412, filed February 19, 2010, and entitled "Apparatus, Systems, And Methods For Treating Industrial Waste Gases"; and
美国临时专利申请61/311,275,申请日2010年3月5日,题目为“Apparatus,Systems,And Methods For Treating Industrial Waste Gases.″U.S. Provisional Patent Application 61/311,275, filed March 5, 2010, entitled "Apparatus, Systems, And Methods For Treating Industrial Waste Gases."
背景background
二氧化碳和许多其它大气污染物(例如NOx,SOx,挥发性有机物(″VOCs″),和颗粒物)的最浓缩点源是产生能量的发电厂,特别地通过燃烧碳型燃料生产它们的电力的发电厂(例如火力发电厂)。考虑到预计全球能量需要增加,并且尽管非碳型能源的持续增长,但是预计二氧化碳及碳型燃料的其它燃烧产物的大气水平还会增加。因而,利用碳型燃料的发电厂特别地是针对降低二氧化碳及其它大气污染物的技术的用武之地。The most concentrated point sources of carbon dioxide and many other atmospheric pollutants (such as NOx, SOx, volatile organic compounds ("VOCs"), and particulate matter) are power plants that produce energy, particularly those that produce their electricity by burning carbon-based fuels factories (such as thermal power plants). Considering the projected increase in global energy demand, and despite the continued growth of non-carbon-based energy sources, atmospheric levels of carbon dioxide and other combustion products of carbon-based fuels are projected to increase. Thus, power plants utilizing carbon-based fuels are particularly useful for technologies aimed at reducing carbon dioxide and other atmospheric pollutants.
降低来自发电厂废物物流的二氧化碳及其它大气污染物的排放的企图已经产生了许多不同技术,其大部分需要非常大的能量输入以便解决与分离和浓缩扩散气体物质有关的能量。另外,目前的技术和有关设备是效率低的并且成本高的。因而,重要的是开发这样的经济可行的技术,其能够通过以稳定形式埋存(sequestering)二氧化碳及其它大气污染物或者将其转化为有用的商品产品而从气态废物物流中除去大量的二氧化碳及其它大气污染物。Attempts to reduce emissions of carbon dioxide and other atmospheric pollutants from power plant waste streams have resulted in many different technologies, most of which require very large energy inputs to account for the energy associated with separating and concentrating diffuse gaseous species. Additionally, current techniques and related equipment are inefficient and costly. Thus, it is important to develop economically viable technologies capable of removing large quantities of carbon dioxide and other atmospheric pollutants from gaseous waste streams by sequestering carbon dioxide and other atmospheric pollutants in a stable form or by converting them into useful commodity products. other air pollutants.
考虑到上述内容,对于有效且经济地埋存二氧化碳及其它大气污染物的系统和方法存在着重大需要。In view of the foregoing, there is a significant need for systems and methods to efficiently and economically sequester carbon dioxide and other atmospheric pollutants.
概要summary
在一些实施方案中,本发明提供用于将气体组分转移入液体的装置,其包括气体入口;被配置以使液体和气体接触的室;在室中第一位置的第一液体引入单元和在室中第二位置的第二液体引入单元,其中液体引入单元被配置以将液体引入室以便与气体接触;被配置以在液体已经接触气体后包含所述液体的储存器;用于液体的在其已经接触气体后的出口,其中入口、室、液体引入单元、储存器和出口可操作地连接;和以下特征中的至少一个:i)在室中的至少一个阵列的棚排,其中棚排被配置以当气体进入室时重新分布其流动,使得与气体在进入所述室时在与棚排相互作用前的流动相比,气体在室横截面的更大面积上轴向地沿室流动;ii)被配置以减少储存器中的泡沫的消泡设备;iii)每一液体引入单元至少一个泵,用于将液体泵送通过该引入单元;iv)配置液体引入单元,使得液体离开第一单元的流动方向不同于液体离开第二单元的流动方向;v)一个或多个节流孔机构(排泄阀),其被配置以将液体流动引导到液体引入单元中的至少一个、到气体入口中、或其组合;和vi)改变液体引入单元所覆盖的区域,其中液体引入单元包括产生喷雾的雾化单元,其中至少一个雾化单元被配置以产生角度不同于其它雾化单元的喷雾。在一些实施方案中,所述装置包括所述特征中的至少两个。在一些实施方案中,所述装置包括所述特征中的至少三个。在一些实施方案中,所述装置包括所述特征中的至少四个。在一些实施方案中,所述装置包括所述特征中的至少五个。在一些实施方案中,所述装置包括全部所述特征。在一些实施方案中,气体入口被配置以接受工业废气、压缩环境空气、压缩二氧化碳、超临界二氧化碳或其任何组合。在一些实施方案中,气体包括工业废气、已经先前与工业废气分离的二氧化碳、或其任何组合。在一些实施方案中,气体包括二氧化碳、氮氧化物、和硫氧化物中的一种或多种。在一些实施方案中,第一液体引入单元位于气体入口之上的最低水平处并且被取向为以基本上与气体流动方向顺流的方向引导液体流动进入所述室。在一些实施方案中,第二液体引入单元被取向为以基本上与气体流动方向逆流的方向引导液体流动进入所述室。在一些实施方案中,第一液体引入单元,第二液体引入单元或两者包括喷口。在一些实施方案中,喷口包括双流体喷口。在一些实施方案中,喷口包括喷射器-喷射喷口。在一些实施方案中,用变频机控制泵中的至少一个。在一些实施方案中,消泡设备包括位于储存器上的锥体。在一些实施方案中,消泡设备进一步包括朝锥体取向的液体喷雾器。在一些实施方案中,所述装置进一步包括液体再循环回路,其被配置以将液体从储存器引导到液体引入单元中的一个或多个。在一些实施方案中,该装置进一步包括在气体离开接触室前的除雾面(demisting level)。在一些实施方案中,除雾面(demisting level)包括波浪形除雾器,平喷喷雾器,湿式静电沉降器,填料床,或其任何组合。在一些实施方案中,提供给除雾面(demisting level)的液体包括与提供给液体引入单元的液体不同的溶液。在一些实施方案中,提供给每一液体引入单元的液体包括不同的溶液。在一些实施方案中,提供给除雾面(demisting level)的液体是清液。在一些实施方案中,提供给液体引入单元的液体包括浆液。在一些实施方案中,该装置进一步包括粉碎台,其被配置以接受来自储存器的浆液并且将加工过的浆液提供给液体引入单元。在一些实施方案中,再循环回路包括粉碎台。在一些实施方案中,储存器位于在接触室的底部的喷口之下。在一些实施方案中,该装置进一步包括可操作连接到接触室的沉淀槽。在一些实施方案中,沉淀槽包括温度控制器,用于添加pH调节剂的入口,搅拌器,用于晶体生长剂的入口,用于晶体播种剂的入口,用于沉降剂(settling agent)的入口,用于絮凝剂的入口,或其任何组合。在一些实施方案中,该装置进一步包括可操作连接到沉淀槽的沉淀出口。在一些实施方案中,沉淀出口收集固体沉淀物和上层清液溶液。在一些实施方案中,沉淀出口从上层清液溶液中分离固体沉淀物。在一些实施方案中,该装置进一步包括管道,而将固体沉淀物提供给建筑材料制造台。在一些实施方案中,气体入口被配置以接受来自工业设备的废气。在一些实施方案中,气体入口被配置以接受来自燃烧化石燃料的设备的烟道气。在一些实施方案中,气体入口被配置以接受来自燃烧化石燃料的设备的烟道气,进一步地其中烟道气在被提供至所述装置的气体入口前已经通过排放控制系统。在一些实施方案中,排放控制系统包括以下中的一种或多种:i)用于收集颗粒物的静电沉降器;ii)SOx控制技术;iii)NOx控制技术;iv)用于收集颗粒物的物理过滤技术;或v)汞控制技术。在一些实施方案中,雾化单元的喷雾器包括靠近接触室的壁的60°喷雾器和在接触室的内横截面中的90°的喷雾器。在一些实施方案中,气体沿棚排的流动是垂直的。在一些实施方案中,在所述室中,该装置进一步包括填充材料、塔盘、填料床或其任何组合。在一些实施方案中,在所述室中,该装置进一步包括至少一个膜或一个微孔膜。In some embodiments, the present invention provides a device for transferring a gas component into a liquid, comprising a gas inlet; a chamber configured to contact the liquid and the gas; a first liquid introduction unit at a first location in the chamber; and A second liquid introduction unit at a second position in the chamber, wherein the liquid introduction unit is configured to introduce liquid into the chamber so as to contact the gas; a reservoir configured to contain the liquid after the liquid has contacted the gas; a reservoir for the liquid an outlet after it has been exposed to gas, wherein the inlet, chamber, liquid introduction unit, reservoir and outlet are operably connected; and at least one of the following features: i) at least one array of rows in the chamber, wherein the The rows are configured to redistribute the flow of the gas as it enters the chamber such that the gas flows axially along the chamber over a greater area of the chamber cross-section than the flow of the gas upon entering the chamber prior to interacting with the rows. flow; ii) a defoaming device configured to reduce foam in the reservoir; iii) at least one pump per liquid introduction unit for pumping liquid through the introduction unit; iv) configuring the liquid introduction unit so that the liquid leaves The direction of flow of the first unit is different from the direction of flow of liquid exiting the second unit; v) one or more orifice mechanisms (drain valves) configured to direct the flow of liquid to at least one of the liquid introduction units, to In the gas inlet, or a combination thereof; and vi) changing the area covered by the liquid introduction unit, wherein the liquid introduction unit includes an atomization unit that produces a spray, wherein at least one atomization unit is configured to produce an angle different from that of the other atomization units spray. In some embodiments, the device includes at least two of the features. In some embodiments, the device includes at least three of the features. In some embodiments, the device includes at least four of the features. In some embodiments, the device includes at least five of the features. In some embodiments, the device includes all of the described features. In some embodiments, the gas inlet is configured to accept industrial waste gas, compressed ambient air, compressed carbon dioxide, supercritical carbon dioxide, or any combination thereof. In some embodiments, the gas includes industrial waste gas, carbon dioxide that has been previously separated from the industrial waste gas, or any combination thereof. In some embodiments, the gas includes one or more of carbon dioxide, nitrogen oxides, and sulfur oxides. In some embodiments, the first liquid introduction unit is located at the lowest level above the gas inlet and is oriented to direct liquid flow into the chamber in a direction substantially co-current to the direction of gas flow. In some embodiments, the second liquid introduction unit is oriented to direct liquid flow into the chamber in a direction substantially countercurrent to the direction of gas flow. In some embodiments, the first liquid introduction unit, the second liquid introduction unit, or both include a spout. In some embodiments, the jet comprises a two-fluid jet. In some embodiments, the jets comprise injector-jet jets. In some embodiments, at least one of the pumps is controlled with a variable frequency drive. In some embodiments, the defoaming device includes a cone located on the reservoir. In some embodiments, the defoaming device further includes a liquid sprayer oriented towards the cone. In some embodiments, the device further comprises a liquid recirculation loop configured to direct liquid from the reservoir to one or more of the liquid introduction units. In some embodiments, the apparatus further comprises a demisting level before the gas exits the contacting chamber. In some embodiments, the demisting level includes a wave demister, a flat spray nebulizer, a wet electrostatic precipitator, a packed bed, or any combination thereof. In some embodiments, the liquid provided to the demisting level comprises a different solution than the liquid provided to the liquid introduction unit. In some embodiments, the liquid provided to each liquid introduction unit comprises a different solution. In some embodiments, the liquid provided to the demisting level is clear liquid. In some embodiments, the liquid provided to the liquid introduction unit comprises a slurry. In some embodiments, the apparatus further includes a comminution station configured to accept the slurry from the reservoir and provide processed slurry to the liquid introduction unit. In some embodiments, the recirculation loop includes a comminution station. In some embodiments, the reservoir is located below the spout at the bottom of the contacting chamber. In some embodiments, the apparatus further includes a settling tank operatively connected to the contacting chamber. In some embodiments, the settling tank includes a temperature controller, an inlet for adding a pH adjuster, an agitator, an inlet for a crystal growth agent, an inlet for a crystal seeding agent, an inlet for a settling agent inlet, inlet for flocculant, or any combination thereof. In some embodiments, the device further includes a settling outlet operatively connected to the settling tank. In some embodiments, the precipitation outlet collects the solid precipitate and the supernatant solution. In some embodiments, the precipitation outlet separates solid precipitate from the supernatant solution. In some embodiments, the apparatus further includes a conduit to provide the solid sediment to the building material manufacturing station. In some embodiments, the gas inlet is configured to accept exhaust gas from an industrial facility. In some embodiments, the gas inlet is configured to accept flue gas from a fossil fuel fired facility. In some embodiments, the gas inlet is configured to accept flue gas from a fossil fuel fired facility, further wherein the flue gas has passed through an emission control system before being provided to the gas inlet of the device. In some embodiments, the emission control system includes one or more of: i) electrostatic precipitators for collecting particulate matter; ii) SOx control technology; iii) NOx control technology; iv) physical Filtration technology; or v) Mercury control technology. In some embodiments, the nebulizers of the nebulization unit comprise 60° nebulizers near the walls of the contacting chamber and 90° nebulizers in the inner cross-section of the contacting chamber. In some embodiments, the flow of gas along the rows is vertical. In some embodiments, in the chamber, the apparatus further comprises packing material, trays, packed beds, or any combination thereof. In some embodiments, in said chamber, the device further comprises at least one membrane or one microporous membrane.
在一些实施方案中,本发明提供一种装置,其包括吸收器,所述吸收器包括鼓泡塔;可操作地连接到吸收器的用于工业气体的入口,其中工业气体包括数量等于时均原始组成的CO2,SOx,NOx,重金属,非CO2酸性气和/或飞灰;用于吸收溶液的入口,其中吸收溶液包括碱性溶液,其包括盐水、颗粒物材料或在吸收浆液中的两者;用于排出气体的出口,所述气体特征为其相对于所述工业气体的所述原始组成而言贫乏CO2及SOx、NOx、重金属、非CO2酸性气体和/或飞灰中的至少一个,其中所述出口可操作地连接到所述吸收器;可操作地连接到吸收器的用于已接触工业气体的吸收溶液的出口;和可操作连接到该出口的加工台,其中加工台被配置以获得以下可售产品中的至少一种:包括CO2埋存组分的建筑材料、脱盐水、饮用水、包括CO2埋存组分的浆液或包括CO2埋存组分的溶液,其中鼓泡塔被配置以在吸收溶液中生产工业气体的气泡使得工业气体中的至少10wt%的二氧化碳被转移到吸收溶液。在一些实施方案中,本发明提供一种装置,其包括吸收器,所述吸收器包括喷布(sparging)容器;可操作地连接到吸收器的用于工业气体的入口,其中工业气体包括数量等于时均原始组成的CO2,SOx,NOx,重金属,非CO2酸性气和/或飞灰;用于吸收溶液的入口,其中吸收溶液包括碱性溶液,其包括盐水、颗粒物材料或在吸收浆液中的两者;用于排出气体的出口,所述气体特征为其相对于所述工业气体的所述原始组成而言贫乏CO2及SOx、NOx、重金属、非CO2酸性气体和/或飞灰中的至少一个,其中所述出口可操作地连接到所述吸收器;可操作地连接到吸收器的用于已接触工业气体的吸收溶液的出口;和可操作连接到该出口的加工台,其中加工台被配置以获得以下可售产品中的至少一种:包括CO2埋存组分的建筑材料、脱盐水、饮用水、包括CO2埋存组分的浆液或包括CO2埋存组分的溶液,其中喷布容器被配置以在吸收溶液中生产工业气体的气泡使得工业气体中的至少10wt%的二氧化碳被转移到吸收溶液。在一些实施方案中,本发明提供一种装置,所述装置包括吸收器,其包括喷雾塔;可操作地连接到吸收器的用于工业气体的入口,其中工业气体包括数量等于时均原始组成的CO2,SOx,NOx,重金属,非CO2酸性气和/或飞灰;用于吸收溶液的入口,其中吸收溶液包括碱性溶液、颗粒物材料或在吸收浆液中的两者;用于排出气体的出口,所述气体特征为其相对于所述工业气体的所述原始组成而言贫乏CO2及SOx、NOx、重金属、非CO2酸性气体和/或飞灰中的至少一个,其中所述出口可操作地连接到所述吸收器;可操作地连接到吸收器的用于已接触工业气体的吸收溶液的出口;和可操作连接到该出口的加工台,其中加工台被配置以获得以下可售产品中的至少一种:包括CO2埋存组分的建筑材料、脱盐水、饮用水、包括CO2埋存组分的浆液或包括CO2埋存组分的溶液,其中喷雾塔被配置以在工业气体中产生吸收溶液的物流、液滴或其组合使得工业气体中的至少10wt%的二氧化碳被转移到吸收溶液,进一步地其中喷雾塔被配置以在50-5,000加仑/分钟/1000实际立方英尺的液体流动速率与气体流动速率比值(L/G比)下操作。在一些实施方案中,本发明提供一种装置,所述装置包括吸收器,其包括喷雾塔、填充材料、填料床、塔盘、棚排或微孔膜中的至少一种;可操作地连接到吸收器的用于工业气体的入口,其中工业气体包括数量等于时均原始组成的CO2,SOx,NOx,重金属,非CO2酸性气和/或飞灰;用于吸收溶液的入口,其中吸收溶液包括碱性溶液、颗粒物材料或在吸收浆液中的两者;用于排出气体的出口,所述气体特征为其相对于所述工业气体的所述原始组成而言贫乏CO2及SOx、NOx、重金属、非CO2酸性气体和/或飞灰中的至少一个,其中所述出口可操作地连接到所述吸收器;可操作地连接到吸收器的用于已接触工业气体的吸收溶液的出口;和可操作连接到该出口的加工台,其中加工台被配置以获得以下可售产品中的至少一种:包括CO2埋存组分的建筑材料、脱盐水、饮用水、包括CO2埋存组分的浆液或包括CO2埋存组分的溶液,其中吸收器被配置以在工业气体中产生吸收溶液的物流、液滴或其组合使得工业气体中的至少10wt%的二氧化碳被转移到吸收溶液,并且进一步其中吸收器被配置以在50-5,000加仑/分/1000实际立方英尺的液体流动速率与气体流动速率比值(L/G比)操作。在一些实施方案中,本发明提供一种装置,所述装置包括吸收器,其包括喷雾塔、填充材料、填料床、塔盘、棚排或微孔膜中的至少一种;可操作地连接到吸收器的用于工业气体的入口,其中工业气体包括数量等于时均原始组成的CO2,SOx,NOx,重金属,非CO2酸性气和/或飞灰;用于吸收溶液的入口,其中吸收溶液包括碱性溶液、颗粒物材料或在吸收浆液中的两者,其中所述碱性溶液包括盐水、清液或其任何组合;用于排出气体的出口,所述气体特征为其相对于所述工业气体的所述原始组成而言贫乏CO2及SOx、NOx、重金属、非CO2酸性气体和/或飞灰中的至少一个,其中所述出口可操作地连接到所述吸收器;可操作地连接到吸收器的用于已接触工业气体的吸收溶液的出口;和可操作连接到该出口的加工台,其中加工台被配置以获得以下可售产品中的至少一种:包括CO2埋存组分的建筑材料、脱盐水、饮用水、包括CO2埋存组分的浆液或包括CO2埋存组分的溶液,其中吸收器被配置以在工业气体中产生吸收溶液的物流、液滴或其组合使得工业气体中的至少10wt%的二氧化碳被转移到吸收溶液。在一些实施方案中,本发明提供一种装置,所述装置包括吸收器,其包括喷雾塔、填充材料、填料床、塔盘、棚排或微孔膜中的至少一种;可操作地连接到吸收器的用于工业气体的入口,其中工业气体包括数量等于时均原始组成的CO2,SOx,NOx,重金属,非CO2酸性气和/或飞灰;用于吸收溶液的入口,其中吸收溶液包括碱性溶液、颗粒物材料或在吸收浆液中的两者,其中所述碱性溶液包括盐水;用于排出气体的出口,所述气体特征为其相对于所述工业气体的所述原始组成而言贫乏CO2及SOx、NOx、重金属、非CO2酸性气体和/或飞灰中的至少一个,其中所述出口可操作地连接到所述吸收器;可操作地连接到吸收器的用于已接触工业气体的吸收溶液的出口;和可操作连接到该出口的加工台,其中加工台被配置以获得可售产品,其中吸收器被配置以在工业气体中产生吸收溶液的物流、液滴或其组合使得工业气体中的至少10wt%的二氧化碳被转移到吸收溶液。在一些实施方案中,用于工业气体的入口被配置以接受工业废气,燃烧烟道气,水泥窑烟道气,压缩二氧化碳,超临界二氧化碳或其任何组合。在一些实施方案中,吸收溶液接触工业气体使得吸收溶液以液滴、溪流(rivulets)、液体柱、喷射喷雾、液膜、溶液等浮云(neutrally buoyantclouds of soluteion)或其任何组合的形式存在。在一些实施方案中,该装置进一步包括在接触室中的雾化组件,包括:压力雾化器(喷口)、旋转雾化器、空气辅助雾化器、空气喷射雾化器、超声雾化器、喷墨雾化器、MEMS雾化器、静电喷雾器、双流体雾化器、排出喷口或其任何组合。在一些实施方案中,吸收溶液中的盐水包括海水、碱性盐水、富阳离子的盐水、合成盐水、工业废水、工业废盐水或其任何组合。在一些实施方案中,该装置进一步包括再循环系统。在一些实施方案中,再循环系统包括管道和泵从而将已接触工业气体的吸收溶液从用于已接触工业气体的吸收溶液的出口、加工台或两者转移到用于吸收溶液的入口、雾化组件或其任何组合。在一些实施方案中,再循环系统包括管道和泵从而将CO2减少的气体从用于排出气体的出口转移到用于工业气体的入口、鼓泡塔、喷布容器或其任何组合。In some embodiments, the present invention provides an apparatus comprising an absorber comprising a bubble column; an inlet for an industrial gas operatively connected to the absorber, wherein the industrial gas comprises an amount equal to the time-average Original composition of CO2 , SOx, NOx, heavy metals, non- CO2 acid gases and/or fly ash; inlet for absorption solutions including alkaline solutions including brine, particulate material or in absorption slurries Both; outlets for exhaust gases characterized by being depleted in CO2 and SOx, NOx, heavy metals, non- CO2 acid gases and/or in fly ash relative to said original composition of said industrial gas At least one of, wherein the outlet is operatively connected to the absorber; an outlet for the absorbing solution that has been contacted with the industrial gas is operatively connected to the absorber; and a processing station is operatively connected to the outlet, wherein The processing station is configured to obtain at least one of the following salable products: building materials including a CO2 storage component, desalinated water, potable water, a slurry including a CO2 storage component, or a CO2 storage component A solution in which the bubble column is configured to produce bubbles of the industrial gas in the absorption solution such that at least 10 wt% of the carbon dioxide in the industrial gas is transferred to the absorption solution. In some embodiments, the present invention provides an apparatus comprising an absorber comprising a sparging vessel; an inlet for an industrial gas operatively connected to the absorber, wherein the industrial gas comprises a quantity CO 2 , SOx, NOx, heavy metals, non-CO 2 acid gases and/or fly ash equal to the time-averaged original composition; inlet for absorption solutions including alkaline solutions including brine, particulate material or both in the slurry; an outlet for exhaust gas characterized by its depletion in CO and SOx, NOx, heavy metals, non- CO acid gases and/or relative to the original composition of the industrial gas At least one of the fly ash, wherein the outlet is operatively connected to the absorber; an outlet for the absorbing solution that has been contacted with the industrial gas is operably connected to the absorber; and a process operatively connected to the outlet station, wherein the processing station is configured to obtain at least one of the following salable products: building materials including a CO2 sequestration component, desalinated water, drinking water, a slurry including a CO2 sequestration component, or a CO2 sequestration component A solution of a storage component, wherein the dispensing vessel is configured to generate bubbles of the industrial gas in the absorbing solution such that at least 10 wt% of the carbon dioxide in the industrial gas is transferred to the absorbing solution. In some embodiments, the present invention provides an apparatus comprising an absorber comprising a spray tower; an inlet for an industrial gas operatively connected to the absorber, wherein the industrial gas comprises an amount equal to the time-averaged original composition CO2 , SOx, NOx, heavy metals, non- CO2 acid gases, and/or fly ash; for inlet of absorption solution, where absorption solution includes alkaline solution, particulate material, or both in the absorption slurry; for discharge an outlet for a gas characterized by being depleted in CO and at least one of SOx, NOx, heavy metals, non- CO acid gases and/or fly ash relative to said original composition of said industrial gas, wherein said Said outlet is operatively connected to said absorber; an outlet for absorbing solution that has been contacted with industrial gas is operably connected to the absorber; and a processing station is operatively connected to the outlet, wherein the processing station is configured to obtain At least one of the following salable products: building materials including a CO2 sequestration component, desalinated water, drinking water, a slurry including a CO2 sequestration component, or a solution including a CO2 sequestration component, wherein the spray tower A stream, liquid droplets, or combination thereof configured to produce an absorbing solution in an industrial gas such that at least 10 wt% of the carbon dioxide in the industrial gas is transferred to the absorbing solution, further wherein the spray tower is configured to operate at Operates at a liquid flow rate to gas flow rate ratio (L/G ratio) of 1000 actual cubic feet. In some embodiments, the present invention provides an apparatus comprising an absorber comprising at least one of a spray tower, packing material, packed bed, tray, row, or microporous membrane; operably connected to Inlets to absorbers for industrial gases, where industrial gases include CO2 , SOx, NOx, heavy metals, non- CO2 acid gases, and/or fly ash in quantities equal to the time-averaged original composition; inlets for absorption solutions, where Absorbing solution comprising alkaline solution, particulate material or both in the absorbing slurry; outlet for venting gas characterized by being depleted in CO2 and SOx relative to said original composition of said industrial gas, At least one of NOx, heavy metals, non- CO2 acid gases and/or fly ash, wherein said outlet is operatively connected to said absorber; an absorption solution for exposed industrial gases operatively connected to absorber and a processing station operatively connected to the outlet, wherein the processing station is configured to obtain at least one of the following salable products: building materials including CO2 sequestration components, desalinated water, drinking water, including CO 2 a slurry of a sequestration component or a solution comprising a CO2 sequestration component, wherein the absorber is configured to produce a stream, droplets, or combination thereof of the absorbing solution in an industrial gas such that at least 10 wt% of the carbon dioxide in the industrial gas is absorbed Transfer to absorption solution, and further wherein the absorber is configured to operate at a liquid flow rate to gas flow rate ratio (L/G ratio) of 50-5,000 gallons per minute per 1000 actual cubic feet. In some embodiments, the present invention provides an apparatus comprising an absorber comprising at least one of a spray tower, packing material, packed bed, tray, row, or microporous membrane; operably connected to Inlets to absorbers for industrial gases, where industrial gases include CO2 , SOx, NOx, heavy metals, non- CO2 acid gases, and/or fly ash in quantities equal to the time-averaged original composition; inlets for absorption solutions, where The absorption solution includes an alkaline solution, particulate material, or both in the absorption slurry, wherein the alkaline solution includes saline, clear liquid, or any combination thereof; an outlet for exhausting gases characterized by their relative said raw composition of said industrial gas is depleted in CO and at least one of SOx, NOx, heavy metals, non- CO acid gases, and/or fly ash, wherein said outlet is operatively connected to said absorber; an outlet operatively connected to the absorber for the absorption solution that has been contacted with the industrial gas; and a processing station operably connected to the outlet, wherein the processing station is configured to obtain at least one of the following salable products: comprising CO A construction material of a sequestration component, desalinated water, drinking water, a slurry comprising a CO2 sequestration component or a solution comprising a CO2 sequestration component, wherein the absorber is configured to produce a stream of the absorbing solution in an industrial gas, The droplets or combination thereof cause at least 10 wt% of the carbon dioxide in the industrial gas to be transferred to the absorbing solution. In some embodiments, the present invention provides an apparatus comprising an absorber comprising at least one of a spray tower, packing material, packed bed, tray, row, or microporous membrane; operably connected to Inlets to absorbers for industrial gases, where industrial gases include CO2 , SOx, NOx, heavy metals, non- CO2 acid gases, and/or fly ash in quantities equal to the time-averaged original composition; inlets for absorption solutions, where an absorption solution comprising an alkaline solution, particulate material, or both in an absorption slurry, wherein the alkaline solution comprises brine; an outlet for exhausting gases characterized by their original Compositionally lean in CO and at least one of SOx, NOx, heavy metals, non- CO acid gases, and/or fly ash, wherein the outlet is operatively connected to the absorber; an outlet for the absorption solution that has been contacted with the industrial gas; and a processing station operably connected to the outlet, wherein the processing station is configured to obtain a salable product, wherein the absorber is configured to generate a stream of the absorption solution in the industrial gas, The droplets or combination thereof cause at least 10 wt% of the carbon dioxide in the industrial gas to be transferred to the absorbing solution. In some embodiments, the inlet for industrial gas is configured to accept industrial waste gas, combustion flue gas, cement kiln flue gas, compressed carbon dioxide, supercritical carbon dioxide, or any combination thereof. In some embodiments, the absorbing solution is exposed to the industrial gas such that the absorbing solution exists as liquid droplets, rivulets, liquid columns, jet sprays, liquid films, neutrally buoyant clouds of solution, or any combination thereof. In some embodiments, the device further comprises atomization components in the contacting chamber, including: pressure atomizers (orifices), rotary atomizers, air-assisted atomizers, air-jet atomizers, ultrasonic atomizers , an inkjet atomizer, a MEMS atomizer, an electrostatic atomizer, a two-fluid atomizer, a discharge nozzle, or any combination thereof. In some embodiments, the brine in the absorption solution includes seawater, alkaline brine, cation-rich brine, synthetic brine, industrial wastewater, industrial waste brine, or any combination thereof. In some embodiments, the apparatus further includes a recirculation system. In some embodiments, the recirculation system includes piping and pumps to transfer the industrial gas contacted absorption solution from the outlet for the exposed industrial gas absorption solution, the processing station, or both to the inlet for the absorption solution, the mist components or any combination thereof. In some embodiments, the recirculation system includes piping and pumps to transfer the CO2 - reduced gas from an outlet for vent gas to an inlet for industrial gas, a bubble column, a spray vessel, or any combination thereof.
在一些实施方案中,本发明提供可操作连接到发电厂的排放控制系统,其中发电厂产生能量和包括二氧化碳的工业废气,其中排放控制系统被配置以吸收来自废气的至少50%的二氧化碳并且被配置以使用小于30%的发电厂所产生的能量。在一些实施方案中,本发明提供可操作连接到发电厂的排放控制系统,其中发电厂产生能量和包括硫氧化物的工业废气,其中排放控制系统被配置以吸收来自废气的至少90%的硫氧化物并且被配置以使用小于30%的发电厂所产生的能量。在一些实施方案中,本发明提供可操作连接到发电厂的排放控制系统,其中发电厂产生能量和包括二氧化碳和硫氧化物的工业废气,其中排放控制系统被配置以吸收来自废气的至少50%的二氧化碳和至少80%的硫氧化物,并且其中所述排放控制系统进一步被配置以使用小于30%的发电厂所产生的能量。在一些实施方案中,排放控制系统被配置以接受来自发电厂的至少10%的工业废气。在一些实施方案中,排放控制系统被配置以接受来自被配置以产生苛性碱溶液(caustic solution)的电化学系统的碱性溶液。在一些实施方案中,电化学系统包括阳极、阴极和在阳极和阴极之间的至少一个离子选择膜。在一些实施方案中,电化学系统被配置以在阳极和阴极两端施加的2.8V或更小的电压下操作。在一些实施方案中,排放控制系统被配置以接受pH调节剂,其中pH调节剂包括工业废物、天然存在的pH调节剂、所产生的pH调节剂或其任何组合。在一些实施方案中,排放控制系统被配置以在5-5,000加仑/分/1000实际立方英尺每分钟的液体流动速率与气体流动速率比值(L/G)下操作。在一些实施方案中,系统被配置以在100-500加仑/分/1000实际立方英尺每分钟的液体流动速率与气体流动速率比值(L/G)下操作。In some embodiments, the present invention provides an emission control system operably connected to a power plant that produces energy and industrial exhaust gas comprising carbon dioxide, wherein the emission control system is configured to absorb at least 50% of the carbon dioxide from the exhaust gas and is Configured to use less than 30% of the energy produced by the power plant. In some embodiments, the present invention provides an emission control system operably connected to a power plant, wherein the power plant generates energy and an industrial waste gas comprising sulfur oxides, wherein the emission control system is configured to absorb at least 90% of the sulfur from the waste gas oxide and is configured to use less than 30% of the energy produced by the power plant. In some embodiments, the present invention provides an emission control system operably connected to a power plant, wherein the power plant produces energy and industrial exhaust gases comprising carbon dioxide and sulfur oxides, wherein the emission control system is configured to absorb at least 50% of the emissions from the exhaust gases carbon dioxide and at least 80 percent sulfur oxides, and wherein the emission control system is further configured to use less than 30 percent of the energy produced by the power plant. In some embodiments, the emission control system is configured to accept at least 10% of the industrial exhaust from the power plant. In some embodiments, the emission control system is configured to accept an alkaline solution from an electrochemical system configured to generate a caustic solution. In some embodiments, an electrochemical system includes an anode, a cathode, and at least one ion selective membrane between the anode and the cathode. In some embodiments, the electrochemical system is configured to operate at a voltage of 2.8 V or less applied across the anode and cathode. In some embodiments, the emission control system is configured to accept a pH adjusting agent, wherein the pH adjusting agent comprises industrial waste, naturally occurring pH adjusting agents, produced pH adjusting agents, or any combination thereof. In some embodiments, the emission control system is configured to operate at a liquid flow rate to gas flow rate ratio (L/G) of 5-5,000 gallons per minute per 1000 actual cubic feet per minute. In some embodiments, the system is configured to operate at a liquid flow rate to gas flow rate ratio (L/G) of 100-500 gallons per minute per 1000 actual cubic feet per minute.
在本文中提供了这样的系统,其包括用于产生包括沉淀产物(包括碳酸盐、碳酸氢盐或其组合)的排出物的沉淀反应器,该沉淀反应器可操作地连接到用于浓缩来自沉淀反应器排出物的沉淀产物的液-固分离装置。Provided herein is a system comprising a precipitation reactor for producing an effluent comprising precipitation products including carbonates, bicarbonates, or combinations thereof, the precipitation reactor being operatively connected to a Liquid-solid separation device for precipitation products from precipitation reactor effluent.
在液-固分离装置的一个版本中,液-固分离装置包括折流板(baffle),所述折流板的位置使得在操作中折流板使沉淀反应器排出物偏转,从而使得沉淀产物下降至液-固分离装置的下部区域并且上层清液上升并且离开液-固分离装置。在液-固装置的另一版本中,液-固分离装置包括螺旋形通道,其被配置以引导来自沉淀反应器的排出物在螺旋形通道中流动,使得基于尺寸与质量进行沉淀产物的浓缩并且产生上层清液。本文中所述的系统的液-固分离装置包括沉淀产物收集器,其能够收集50%-100%,75%-100%,或95%-100%的来自沉淀台的沉淀产物。另外,液-固分离装置能够加工100L/min-20,000L/min,5000L/min-20,000L/min,或10,000L/min-20,000L/min的来自沉淀台的排出物。In one version of the liquid-solid separation device, the liquid-solid separation device includes baffles positioned such that in operation the baffles deflect the precipitation reactor effluent so that the precipitated product Descends to the lower region of the liquid-solid separation device and the supernatant rises and leaves the liquid-solid separation device. In another version of the liquid-solid device, the liquid-solid separation device includes a helical channel configured to direct the effluent from the precipitation reactor to flow in the helical channel such that the concentration of the precipitated product is based on size and mass And a supernatant was produced. The liquid-solid separation device of the system described herein includes a precipitated product collector capable of collecting 50%-100%, 75%-100%, or 95%-100% of the precipitated product from the settling table. In addition, the liquid-solid separation device is capable of processing 100L/min-20,000L/min, 5000L/min-20,000L/min, or 10,000L/min-20,000L/min of the effluent from the settling table.
本文中所述的系统的沉淀反应器可以包括加料反应器和沉淀台。加料反应器能够从工业废气物流中除去CO2。此外,加料反应器可以能够从工业废气物流(steam)中除去SOx、NOx、重金属、颗粒物、VOCs或其组合中的一种或多种。加料反应器包括连接到水源的平喷喷口,其中平喷喷口适合于形成平喷物流,从而使包括CO2的气态废物物流与来自水源的水接触。包括CO2的气态废物物流是来自燃烧碳型燃料、煅烧材料或其组合的工业设备的废物物流。水源所提供的水可以包含碱土金属离子;在此情况下水源可以选自淡水微咸水、海水和盐水。沉淀台可操作地连接到pH-提高剂源。pH-提高剂可以包括灰、氧化物、氢氧化物或碳酸盐。沉淀台适合于生产包括碳酸盐、碳酸氢盐或其组合的沉淀产物。The precipitation reactors of the systems described herein may include a feed reactor and a precipitation table. Addition reactors are capable of removing CO2 from industrial waste gas streams. Additionally, the feed reactor may be capable of removing one or more of SOx, NOx, heavy metals, particulate matter, VOCs, or combinations thereof from the industrial waste gas stream (steam). The feed reactor includes a flat jet nozzle connected to a water source, wherein the flat jet nozzle is adapted to form a flat jet stream such that a gaseous waste stream including CO2 is contacted with water from the water source. Gaseous waste streams including CO2 are waste streams from industrial facilities burning carbon-based fuels, calcined materials, or combinations thereof. The water source The water provided may contain alkaline earth metal ions; in this case the water source may be selected from fresh brackish water, sea water and brine. The settling station is operatively connected to a source of pH-enhancing agent. pH-raising agents may include ash, oxides, hydroxides or carbonates. The precipitation station is suitable for producing precipitation products including carbonates, bicarbonates or combinations thereof.
本文中所述的系统可以进一步包括电化学电池。电化学电池可以被配置以除去来自加料台、沉淀台或加料和沉淀台的质子。The systems described herein can further include electrochemical cells. The electrochemical cell can be configured to remove protons from a feeding station, a settling station, or a feeding and settling station.
还提供了综合系统,其包括燃烧碳型燃料的发电厂而产生包括二氧化碳的废气物流,其可操作地连接到废气加工系统。废气加工系统包括用于产生包括沉淀产物(包括碳酸盐、碳酸氢盐或其组合)的排出物的沉淀反应器,其可操作地连接到用于浓缩来自沉淀反应器排出物的沉淀产物的液-固分离装置。在液-固分离装置的一个版本中,液-固分离装置包括折流板(baffle),所述折流板的位置使得在操作中折流板使沉淀反应器排出物偏转,从而使得沉淀产物下降至液-固分离装置的下部区域并且上层清液上升并且离开液-固分离装置。在液-固分离装置的另一版本中,液-固分离装置包括螺旋形通道,其被配置以引导来自沉淀反应器的排出物在螺旋形通道中流动,使得基于尺寸与质量进行沉淀产物的浓缩并且产生上层清液。废气物流进一步包括SOx、NOx、重金属、VOCs、颗粒物或其组合。Also provided is an integrated system comprising a power plant burning a carbon-based fuel to produce an exhaust gas stream comprising carbon dioxide operatively connected to an exhaust gas processing system. The exhaust gas processing system includes a precipitation reactor for producing an effluent comprising precipitation products, including carbonates, bicarbonates, or combinations thereof, operably connected to a precipitation reactor for concentrating the precipitation products from the precipitation reactor effluent Liquid-solid separation device. In one version of the liquid-solid separation device, the liquid-solid separation device includes baffles positioned such that in operation the baffles deflect the precipitation reactor effluent so that the precipitated product Descends to the lower region of the liquid-solid separation device and the supernatant rises and leaves the liquid-solid separation device. In another version of the liquid-solid separation device, the liquid-solid separation device includes a helical channel configured to direct the effluent from the precipitation reactor to flow in the helical channel such that the precipitation product is separated based on size and mass. Concentrate and yield a supernatant. The exhaust gas stream further includes SOx, NOx, heavy metals, VOCs, particulate matter, or combinations thereof.
还提供了这样的方法,其包括将部分或全部的包括二氧化碳的来自工业设备的气态废物物流转移到用于产生包括沉淀产物(包括碳酸盐、碳酸氢盐或其组合)的排出物的沉淀反应器;和在液-固分离装置中浓缩来自沉淀反应器排出物的沉淀产物。在液-固分离装置的一个版本中,排出物在液-固分离装置中倚靠折流板偏转,使得沉淀产物下降至液-固分离装置的下部区域并且上层清液上升并且离开液-固分离装置。在液-固分离装置的另一版本中,使得排出物在螺旋形通道中流动,使得基于尺寸与质量进行沉淀产物的浓缩和产生上层清液。Also provided is a method comprising diverting part or all of a gaseous waste stream comprising carbon dioxide from an industrial facility to a precipitation facility for producing an effluent comprising precipitation products including carbonates, bicarbonates, or combinations thereof a reactor; and concentrating the precipitation product from the precipitation reactor effluent in a liquid-solid separation device. In one version of the liquid-solid separation device, the effluent is deflected against baffles in the liquid-solid separation device so that the precipitated product descends into the lower region of the liquid-solid separation device and the supernatant rises and exits the liquid-solid separation device device. In another version of the liquid-solid separation device, the effluent is made to flow in a helical channel, allowing concentration of the precipitated product and generation of a supernatant based on size and mass.
可以使用前述权利要求中的任一项的任何系统,进行埋存二氧化碳的方法。The method of sequestering carbon dioxide may be carried out using any system of any one of the preceding claims.
附图说明Description of drawings
在所附权利要求中详细地阐述了本发明的新颖特征。参考以下阐述其中利用了本发明原理的例证性实施方案的详细说明和附图将获得本发明的特征和优点的更好的理解,在附图中:The novel features of the invention are set forth with particularity in the appended claims. A better understanding of the features and advantages of the present invention will be obtained by reference to the following detailed description and accompanying drawings which set forth illustrative embodiments in which the principles of the invention are utilized, in which:
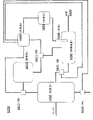
图1提供一种本发明的系统,其包括处理器,其中处理器被配置以加工各种包括二氧化碳的气体。Figure 1 provides a system of the present invention comprising a processor configured to process various gases including carbon dioxide.
图2提供一种本发明的系统,其包括处理器和处理系统,其中处理系统被配置以处理来自处理器的组合物。Figure 2 provides a system of the present invention comprising a processor and a processing system, wherein the processing system is configured to process a composition from the processor.
图3提供一种本发明的系统,其包括处理器和任选的处理系统,其中处理器包括接触器和反应器。Figure 3 provides a system of the present invention comprising a processor and an optional processing system, wherein the processor comprises a contactor and a reactor.
图4提供一种本发明的系统,其包括处理器和处理系统,其中来自处理系统的上层清液可以任选地被再循环到处理器。Figure 4 provides a system of the present invention comprising a processor and a treatment system, wherein the supernatant from the treatment system can optionally be recycled to the processor.
图5提供一种本发明的系统,其包括处理器、处理系统和电化学系统,其中来自处理系统的上层清液可以任选地被再循环到处理器、电化学系统或其组合。Figure 5 provides a system of the present invention comprising a processor, a treatment system, and an electrochemical system, wherein the supernatant from the treatment system can optionally be recycled to the processor, the electrochemical system, or a combination thereof.
图6A提供了根据本发明一个实施方案的CO2埋存方法的示意图。Figure 6A provides a schematic diagram of a CO2 sequestration method according to one embodiment of the present invention.
图6B提供了根据本发明一个实施方案的CO2埋存系统的示意图。Figure 6B provides a schematic diagram of a CO2 storage system according to one embodiment of the present invention.
图6C提供是该系统实施方案的示意图。Figure 6C provides a schematic diagram of an embodiment of this system.
图7提供了根据本发明的另一实施方案的CO2埋存系统的示意图。Figure 7 provides a schematic diagram of a CO2 storage system according to another embodiment of the present invention.
图8提供了用于电化学生产氢氧化物的低压装置的一个实施方案的简图。Figure 8 provides a schematic diagram of one embodiment of a low pressure apparatus for the electrochemical production of hydroxides.
图9提供了用于电化学生产氢氧化物的低压装置的另一实施方案的简图。Figure 9 provides a schematic diagram of another embodiment of a low pressure apparatus for the electrochemical production of hydroxides.
图10提供了用于电化学生产氢氧化物的低压装置的另一实施方案的简图。Figure 10 provides a schematic diagram of another embodiment of a low pressure apparatus for the electrochemical production of hydroxides.
图11是具有塔配置的用于接触固体材料、液体和气体的装置的实施方案的示意图。Figure 11 is a schematic diagram of an embodiment of an apparatus for contacting solid material, liquid and gas with a column configuration.
图12是用于接触固体材料、液体和气体的方法的示意图。Figure 12 is a schematic illustration of a method for contacting solid materials, liquids and gases.
图13是如在横截面纵向来看具有水平配置的用于接触固体材料、液体和气体的装置的实施方案的示意图。Figure 13 is a schematic illustration of an embodiment of a device for contacting solid material, liquid and gas with a horizontal configuration as viewed longitudinally in cross section.
图14是如在横截面端点来看具有水平配置的用于接触固体材料、液体和气体的装置的实施方案的示意图。Figure 14 is a schematic illustration of an embodiment of a device for contacting solid materials, liquids and gases having a horizontal configuration as viewed in cross-sectional endpoints.
图15是该装置的实施方案的示意图。Figure 15 is a schematic illustration of an embodiment of the device.
图16是整个装置的实施方案的示意图。Figure 16 is a schematic diagram of an embodiment of the entire device.
图17是显示棚排设计的装置的实施方案的一部分的示意图。Figure 17 is a schematic diagram showing a portion of an embodiment of an apparatus for row design.
图18是显示喷雾器的可能排布的装置的实施方案的示意图。Figure 18 is a schematic diagram of an embodiment of the device showing a possible arrangement of nebulizers.
图19是显示喷雾器的可能排布的装置的实施方案的示意图。Figure 19 is a schematic diagram of an embodiment of the device showing a possible arrangement of nebulizers.
图20是可用于装置和系统的一些实施方案的棚排配置的示意图。Figure 20 is a schematic illustration of a row configuration that may be used with some embodiments of the devices and systems.
图21A显示本发明的实施方案的侧视图,其中液滴和气体沿行在室周围的长路径,其中气体入口在室的顶端。Figure 21A shows a side view of an embodiment of the invention in which droplets and gas follow a long path around a chamber with the gas inlet at the top of the chamber.
图21B显示本发明的实施方案的自顶向下的剖视图,其中液滴和气体沿行在室周围的长路径,其中气体入口在室的顶端。Figure 21B shows a top-down cross-sectional view of an embodiment of the invention in which droplets and gas follow a long path around a chamber with the gas inlet at the top of the chamber.
图22A显示本发明的实施方案的侧视图,其中液滴和气体沿行在室周围的长路径,其中气体入口在室的底部。Figure 22A shows a side view of an embodiment of the invention in which droplets and gas follow a long path around a chamber with the gas inlet at the bottom of the chamber.
图22B显示本发明的实施方案的自顶向下剖视图,其中液滴和气体沿行在室周围的长路径,其中气体入口在室的底部。Figure 22B shows a top-down cross-sectional view of an embodiment of the invention in which the liquid droplets and gas follow a long path around the chamber with the gas inlet at the bottom of the chamber.
图23提供了气-液或气-液-固接触器的示意图。Figure 23 provides a schematic diagram of a gas-liquid or gas-liquid-solid contactor.
图24提供了图23的气-液或气-液-固接触器的另一视图的示意图。FIG. 24 provides a schematic diagram of another view of the gas-liquid or gas-liquid-solid contactor of FIG. 23 .
图25提供在线监控器的简图。Figure 25 provides a simplified diagram of the online monitor.
图26是显示纵向和水平取向的段的装置的实施方案的示意图。Figure 26 is a schematic diagram of an embodiment of a device showing longitudinally and horizontally oriented segments.
图27是显示纵向和水平取向的段的装置的实施方案的示意图。Figure 27 is a schematic diagram of an embodiment of a device showing longitudinally and horizontally oriented segments.
图28是显示纵向和水平取向的段的装置的实施方案的示意图,其中在使用(suing)泵的水平取向的段中相对于气体流动具有逆流溶液循环。Figure 28 is a schematic diagram of an embodiment of a device showing longitudinal and horizontal oriented sections with countercurrent solution circulation with respect to gas flow in the horizontally oriented sections suing pumps.
图29是装置和系统的实施方案的示意图,其中该系统是一系列装置。Figure 29 is a schematic illustration of an embodiment of a device and system, where the system is a series of devices.
图30是显示装置阵列的系统的实施方案的示意图。Figure 30 is a schematic diagram of an embodiment of a system showing an array of devices.
图31是显示装置阵列的系统的实施方案的示意图。Figure 31 is a schematic diagram of an embodiment of a system showing an array of devices.
图32是系统的实施方案的示意图。Figure 32 is a schematic diagram of an embodiment of the system.
图33提供了根据本发明的实施方案的集成以CO2埋存系统的发电厂的示意图。Figure 33 provides a schematic diagram of a power plant integrating a CO2 storage system according to an embodiment of the present invention.
图34提供了波特兰水泥厂的示意图。Figure 34 provides a schematic diagram of a Portland cement plant.
图35提供了根据本发明一个实施方案的与沉淀装置共处的水泥厂的示意图。Figure 35 provides a schematic diagram of a cement plant co-located with a settling unit according to one embodiment of the present invention.
图36提供了根据本发明一个实施方案的不需要开采的石灰石原料的水泥厂的示意图。Figure 36 provides a schematic diagram of a cement plant that does not require mined limestone raw material according to one embodiment of the present invention.
图37提供了根据本发明一个实施方案的系统的示意图。Figure 37 provides a schematic diagram of a system according to one embodiment of the invention.
图38、39和40提供了根据本发明一个实施方案的系统的示意图。Figures 38, 39 and 40 provide schematic illustrations of a system according to one embodiment of the invention.
图41和42提供了本发明的沉淀物的照片。Figures 41 and 42 provide photographs of precipitates of the present invention.
图43提供了本发明的无定形沉淀物的照片。Figure 43 provides a photograph of the amorphous precipitate of the present invention.
图44提供了在以下实施例部分中报道的CO2吸收实验的图表结果。Figure 44 provides graphical results from the CO2 absorption experiments reported in the Examples section below.
图45显示了本发明的实施方案,其中液滴的形成和液滴与所关心的气体的接触在不同的室中进行。Figure 45 shows an embodiment of the invention in which droplet formation and contact of the droplet with the gas of interest are performed in separate chambers.
图46显示了一种实施方案,其中液滴形成和气体接触在一个室中进行,该室被设计成使凝聚的液滴与形成液滴的液体分开。Figure 46 shows an embodiment in which droplet formation and gas contact are performed in a chamber designed to separate the coalescing droplets from the droplet-forming liquid.
图47是显示了使用去泡沫锥体和喷雾器的装置的实施方案的示意图。Figure 47 is a schematic diagram showing an embodiment of a device using a defoaming cone and a nebulizer.
详细说明Detailed description
本文中介绍了与有效地接触固体材料、液体和气体相关的系统、装置和方法。本文中介绍的实施方案代表了用于将气体引入到液体或浆液或两者中的方法和装置。在一些其中介绍了浆液的实施方案中,浆液包括液体和固体材料组分,使得在液体与气体接触的整个过程中存在固体材料。引入所指的是将气体溶解和/或吸收到液体中或者将气体吸附和/或化学吸附到液体的表面上。将气体引入液体是通过优化接触条件而实现的。在本文中实施方案中变化和优化的条件包括:液体的溶液化学;液体的表面积与体积之比;液体或浆液和气体(L/G)的流速比;和液体和气体之间的接触时间。将气体引入液体是令人期望的,这出于许多原因,其中一些在本文中的实施方案中被考虑,包括但不限于除去气体物流的组分(例如洗涤烟道气)和从在气体和液体间的反应中有效的沉淀材料(例如形成细固体颗粒物)。Systems, devices, and methods related to efficiently contacting solid materials, liquids, and gases are described herein. Embodiments presented herein represent methods and apparatus for introducing gases into liquids or slurries or both. In some embodiments where a slurry is introduced, the slurry includes liquid and solid material components such that the solid material is present throughout the contact of the liquid with the gas. Introducing means dissolving and/or absorbing a gas into a liquid or adsorbing and/or chemisorbing a gas onto the surface of a liquid. The introduction of gas into the liquid is achieved by optimizing the contact conditions. Conditions varied and optimized in embodiments herein include: solution chemistry of the liquid; surface area to volume ratio of the liquid; flow rate ratio of the liquid or slurry and gas (L/G); and contact time between the liquid and gas. Introducing a gas into a liquid is desirable for a number of reasons, some of which are contemplated in embodiments herein, including but not limited to removing components of the gas stream (e.g., scrubbing flue gas) and Effective in precipitating material in reactions between liquids (e.g. formation of fine solid particles).
本发明的方法、装置和系统可以利用如本文中进一步所描述的气-液或气-液-固技术。可以利用其中液体或浆液(即吸收溶液)被引入气体的接触方法,其中液体和/或浆液以液滴、物流或其组合的形式引入。可以利用其中气体被引入液体或浆液(即吸收溶液)的接触方法,其中在液体或浆液中气体产生气泡或泡沫。在任一种情况中,可以优化参数以便将二氧化碳和SOx、NOx、重金属、非CO2酸性气体或飞灰中的至少一种引入构成吸收溶液的液体或浆液。为促进这种引入,在在本发明的方法、装置和系统中可以使用结构特征如填料材料、填料床、塔盘、棚排或膜,包括微孔膜。一旦气体的期望组分已经通过溶液和气体之间的接触引入吸收溶液,被接触的溶液可通过适宜的手段进行处理,所述适宜的手段未使得从气体中除去的组分可释放到地球大气中。或者,被接触的溶液可以经受沉淀条件,使得形成固体沉淀物,并且这样的沉淀物和排出物液体可以被进一步处理以便回收可售产品(例如饮用水、建筑材料包括CO2-埋存组分)。The methods, devices and systems of the invention may utilize gas-liquid or gas-liquid-solid technology as further described herein. Contacting methods may be utilized wherein a liquid or slurry (ie, absorption solution) is introduced into the gas, wherein the liquid and/or slurry is introduced in the form of droplets, a stream, or a combination thereof. Contacting methods may be utilized in which a gas is introduced into a liquid or slurry (ie, an absorption solution) in which the gas generates bubbles or foam. In either case, parameters can be optimized to introduce carbon dioxide and at least one of SOx, NOx, heavy metals, non- CO2 acid gases, or fly ash into the liquid or slurry making up the absorption solution. To facilitate such incorporation, structural features such as packing materials, packed beds, trays, rows or membranes, including microporous membranes, may be used in the methods, devices and systems of the invention. Once the desired components of the gas have been introduced into the absorbing solution by contact between the solution and the gas, the contacted solution may be treated by suitable means that do not allow the components removed from the gas to be released into the Earth's atmosphere middle. Alternatively, the contacted solution may be subjected to precipitation conditions such that a solid precipitate is formed, and such precipitate and effluent liquid may be further processed for recovery of salable products (e.g. potable water, building materials including CO2 -storage components ).
本发明的方法、装置和系统可适用于在可操作地连接到发电厂的排放控制系统中使气体和吸收溶液接触,使得来自发电厂的烟道气(即工业废气)包含二氧化碳、SOx、NOx、重金属、非二氧化碳酸性气体和有时候飞灰。令人期望的是除去二氧化碳和SOx、NOx、重金属、非二氧化碳酸性气体和有时候飞灰中的至少一种,同时使用很少的发电厂产生的能量,如30%的发电厂产生的能量,来为排放控制系统提供能量。排放控制系统包括动作和组分如,但不限于,泵送和再循环吸收溶液,再生或再装填吸收溶液,循环烟道气,除去包括飞灰和沉淀物的颗粒物,和处理包含二氧化碳和SOx、NOx、重金属、非二氧化碳酸性气体和有时候飞灰(从烟道气中脱除的)中的至少一种的任何液体、固体或浆液。本发明的方法、装置和系统可以代替其它排放控制系统或者可以与在适当位置发电厂可能具有的现有的系统一起使用,然而,在一些实施方案中,未必必需具有单独的CO2和SOx排放控制系统。The method, apparatus and system of the present invention can be adapted for contacting gas and absorbing solution in an emission control system operably connected to a power plant such that flue gas (i.e. industrial waste gas) from the power plant contains carbon dioxide, SOx, NOx , heavy metals, non-carbon dioxide acid gases and sometimes fly ash. It is desirable to remove carbon dioxide and at least one of SOx, NOx, heavy metals, non-carbon dioxide acid gases, and sometimes fly ash while using very little, such as 30%, of the energy produced by a power plant, to power the emission control system. Emission control systems include actions and components such as, but not limited to, pumping and recirculating absorption solution, regenerating or recharging absorption solution, circulating flue gas, removing particulate matter including fly ash and sediment, and treating carbon dioxide and SOx Any liquid, solid or slurry of at least one of , NOx, heavy metals, non-carbon dioxide acid gases and sometimes fly ash (removed from flue gas). The methods, apparatus and systems of the present invention may replace other emission control systems or may be used with existing systems that power plants may have in place, however, in some embodiments it is not necessary to have separate CO2 and SOx emissions Control System.
一些实施方案利用了同时粉碎、减小粒径和混合浆液的固体组分和浆液的液体组分。一些实施方案包括与气体接触,而同时混合并且减小固体颗粒物的尺寸。粉碎,或减小粒径,可用来通过增加可能参与反应的固体的表面积以及通过在浆液每一次通过粉碎步骤使新的固体材料露出而改进浆液的固体组分的反应性。使用任何合适的减小固体颗粒物尺寸的装置,可以完成粉碎,这样的装置包括但不限于喷射磨、螺旋输送机、高剪切混合器、湿磨机、擦碎机、胶体磨或其任何组合。在一些实施方案中,在浆液与气体首次接触前,进行粉碎。在一些实施方案中,在再循环期间,在浆液与气体首次接触后,进行粉碎。在一些实施方案中,在管道中用螺旋输送机进行粉碎,同时在到达接触室前浆液还与气体接触。Some embodiments utilize simultaneous pulverization, particle size reduction, and mixing of the solid component of the slurry and the liquid component of the slurry. Some embodiments include contacting with a gas while mixing and reducing the size of the solid particulate matter. Comminution, or particle size reduction, can be used to improve the reactivity of the solid components of the slurry by increasing the surface area of solids that may participate in the reaction and by exposing new solid material with each pass of the slurry through the comminution step. Comminution may be accomplished using any suitable device for reducing the size of the solid particles, including but not limited to jet mills, screw conveyors, high shear mixers, wet mills, attritors, colloid mills, or any combination thereof . In some embodiments, pulverization occurs prior to the first contact of the slurry with the gas. In some embodiments, comminution occurs after the slurry is first contacted with the gas during recirculation. In some embodiments, comminution is performed in a pipeline with a screw conveyor while the slurry is also in contact with a gas before reaching the contact chamber.
一些实施方案利用高效气液接触方法或装置。高效气液接触方法或装置具有最优化的接触参数的附加优点,如:较高的表面积,借此气体引入液体可以进行;溶液化学有利于气体引入液体的动力学;在气体引入液体的同时,通过减慢流体流动或者增加液体和气体行程的路径长度,提供了足够的停留时间。Some embodiments utilize high-efficiency gas-liquid contacting methods or devices. High-efficiency gas-liquid contact methods or devices have additional advantages of optimized contact parameters, such as: higher surface area, whereby gas introduction into liquid can be carried out; solution chemistry is conducive to the kinetics of gas introduction into liquid; while gas is introduced into liquid, Sufficient residence time is provided by slowing fluid flow or increasing the path length of liquid and gas travel.
有效的气液接触方法和装置的用途之一是最优化沉淀反应。在气-液接触器中液体的溶液化学的变化影响了液体引入气体的能力。在一些实施方案中,影响液体(例如吸收溶液、接触混合物)的化学性质从而将气体更有效地引入。在一些实施方案中,液体(例如吸收溶液、接触混合物)的pH值允许同时脱除来自气体的CO2和SO2和/或SOx。在一些实施方案中,液体(例如吸收溶液、接触混合物)的化学性质不需要单独的(即,与除去CO2的方法或步骤不同的)SO2和/或SOx脱除步骤。在一些实施方案中,液体的pH值为pH 4至pH 13.5。在一些实施方案中,液体的pH值为pH 4至pH 11。在一些实施方案中,液体的pH值为pH 5至pH10。在一些实施方案中,液体的pH值为pH 5.5至pH 9.5。在一些实施方案中,液体的pH值受加入制备液滴的溶液的试剂的影响。改变液体的pH值的试剂包括但不局限于:天然存在的pH提高剂,微生物和真菌,合成化学pH提高剂,回收的人造废物物流,和电化学手段产生的碱性溶液。One of the uses of efficient gas-liquid contacting methods and apparatus is to optimize precipitation reactions. Changes in the solution chemistry of a liquid in a gas-liquid contactor affect the ability of the liquid to introduce gas. In some embodiments, the chemistry of the liquid (eg, absorption solution, contact mixture) is influenced to introduce the gas more efficiently. In some embodiments, the pH of the liquid (eg, absorption solution, contacting mixture) allows simultaneous removal of CO2 and SO2 and/or SOx from the gas. In some embodiments, the chemistry of the liquid (eg, absorption solution, contacting mixture) does not require a separate (ie, distinct from the method or step of removing CO 2 ) SO 2 and/or SOx removal steps. In some embodiments, the pH of the liquid is between
在更详细地描述本发明前,应将理解的是本发明不局限于本文中所述的具体实施方案,因而实施方案可以变化。同样应当理解,本文中使用的术语仅仅是为了描述具体实施方案的目的,并不意图用于进行限制,因为本发明的范围将仅仅由所附权利要求限制。除非另外定义,本文中使用的全部技术和科学术语具有与本发明所属领域的技术人员通常理解的相同的含义。Before the present invention is described in greater detail, it is to be understood that this invention is not limited to particular embodiments described herein, as such may vary. It is also to be understood that the terminology used herein is for the purpose of describing particular embodiments only and is not intended to be limiting, since the scope of the present invention will be limited only by the appended claims. Unless defined otherwise, all technical and scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art to which this invention belongs.
在提供数值范围的情况下,应当理解的是,在所述范围上下限之间的各个居中值,至所述下限单位十分之一,除非上下文明确相反指出的,以及在所述范围内的任何其它所述的或居间的值,包括在本发明中。这些较小范围的上下限可独立地包括在较小范围内并且也包括在本发明之内,受约束于在所述范围内任何具体排除的界限。在所述范围包括一个或者两个界限时,不包括那些被包括的界限之一或两者的范围也包括在本发明内。Where a range of values is provided, it is understood that each intervening value between the upper and lower limits of the stated range, to the tenth of the unit of the lower limit unless the context clearly dictates otherwise, and within the stated range Any other stated or intervening values are encompassed by the invention. The upper and lower limits of these smaller ranges may independently be included in the smaller ranges and are also encompassed within the invention, subject to any specifically excluded limit in the stated range. Where the stated range includes one or both of the limits, ranges excluding either or both of those included limits are also included in the invention.
某些范围在本文中以术语“大约”在数值之前的形式给出。术语“大约”用于本文中来提供其之后的精确数字以及该术语之后的数字附近或近似数字的文字支持。在确定一个数字是否在具体所述的数字附近或是否是近似,附近或近似的未描述数字可以是这样的数字,其在其所表示于其中的范围中提供了具体所述数字的基本上等同物。Certain ranges are given herein with the term "about" preceding the numerical value. The term "about" is used herein to provide literal support for the exact number that it precedes as well as a number near or approximately the number that the term precedes. In determining whether a number is around or approximate to a specifically stated number, an adjacent or approximately unrecited number may be a number which, in the range in which it is expressed, provides a substantial equivalent of the specifically stated number things.
本说明书中所述的全部出版物、专利和专利申请引入本文作为参考至相同的程度,如同各自单独的出版物、专利或专利申请具体地和单独地被指出来结合作为参考。此外,各个引用的出版物、专利或专利申请引入本文作为参考从而公开和描述了与所引用的出版物相关的主题。任何出版物的引用是为在申请日之前的其公开内容并且不应当被理解为允许本文中所述的本发明由于在先发明而无权预料这样的出版物。进一步,所提供的公开日可能不同于实际公开日,其可能需要单独证实。All publications, patents, and patent applications mentioned in this specification are herein incorporated by reference to the same extent as if each individual publication, patent, or patent application was specifically and individually indicated to be incorporated by reference. In addition, each cited publication, patent, or patent application is incorporated herein by reference to disclose and describe the subject matter to which the publication is cited. Citation of any publication is for its disclosure prior to the filing date and should not be construed as an admission that the inventions described herein are not entitled to such publication by virtue of prior invention. Further, the dates of publication provided may be different from the actual publication dates, which may need to be independently confirmed.
注意的是,如本文中使用的,和所附权利要求中使用的,单数形式“a”、“an”和“the”包括多个所述之项,除非上下文明确相反指出。进一步注意到是可以撰写权利要求来排除任何任选的元素。因而,这种声明意图是用作在权利要求元素列举方面使用这样的排他性术语如“仅仅”等,或者使用“否定”限制的在先基础。Note that as used herein, and in the appended claims, the singular forms "a", "an" and "the" include plural recited items unless the context clearly dictates otherwise. It is further noted that the claims may be drafted to exclude any optional elements. Accordingly, this statement is intended to be used as a prior basis for the use of such exclusive terminology, such as "merely" etc., or for the use of a "negative" limitation with respect to the recitation of claim elements.
当阅读本公开内容时,对于本领域技术人员显而易见的是本文中所述的和举例说明的每一个单独实施方案具有分立的组件和特征,其可以容易地与任何其它多个实施方案的特征分离和合并,而没有背离本发明的范围或精神。以所述事件的顺序或者以任何其它逻辑上可行的顺序,可以进行任何所述的方法。尽管任何类似或等同于本文中所述的那些的方法和材料也可用于实施或测试本发明,但是现在描述代表性的例证性的方法和材料。It will be apparent to those skilled in the art upon reading this disclosure that each individual embodiment described and illustrated herein has discrete components and features that can be readily separated from the features of any other multiple embodiments and incorporated without departing from the scope or spirit of the invention. Any described method may be performed in the order of events described or in any other logically feasible order. Although any methods and materials similar or equivalent to those described herein can also be used in the practice or testing of the present invention, representative illustrative methods and materials are now described.
本发明的方法和系统通过可以利用由以下化学反应所总结的过程:The methods and systems of the present invention may utilize a process summarized by the following chemical reactions:
在液、气或固相中含碳燃料源的燃烧形成了气态二氧化碳:Combustion of carbonaceous fuel sources in the liquid, gaseous or solid phase forms gaseous carbon dioxide:
C+O2(g)→CO2(g)C+O 2 (g)→CO 2 (g)
(2)二氧化碳源与水源的接触使二氧化碳成溶剂化物而得到二氧化碳的水溶液:(2) The contact between the carbon dioxide source and the water source makes carbon dioxide into a solvate to obtain an aqueous solution of carbon dioxide:
CO2(g) CO2(aq)CO 2 (g) CO 2 (aq)
(3)溶于水的二氧化碳形成与碳酸水溶液的平衡:(3) The formation of carbon dioxide dissolved in water and the equilibrium of carbonic acid aqueous solution:
CO2(aq)+H2O H2CO3(aq)CO 2 (aq)+H 2 O H 2 CO 3 (aq)
(4)碳酸是一种弱酸,其在两步中离解,其中平衡天平部分地由溶液的pH值确定,其中通常pHs低于8-9有利于碳酸氢盐形成和pHs高于9-10有利于碳酸盐形成。在第二步中,氢氧化物源可以被加入以便增加碱度:(4) Carbonic acid is a weak acid that dissociates in two steps, where the equilibrium balance is determined in part by the pH of the solution, where typically pHs below 8-9 favor bicarbonate formation and pHs above 9-10 favor conducive to the formation of carbonates. In a second step, a hydroxide source can be added to increase the alkalinity:
H2CO3+2H2O H3O+(aq)+HCO3 -(aq)H 2 CO 3 +2H 2 O H 3 O + (aq)+HCO 3 - (aq)
HCO3 -(aq)+OH-(aq) H2O+CO3 2-(aq)HCO 3 - (aq)+OH - (aq) H 2 O+CO 3 2- (aq)
来自IIA族的元素金属阳离子与碳酸盐阴离子的反应形成金属碳酸盐沉淀物:The reaction of an elemental metal cation from group IIA with a carbonate anion forms a metal carbonate precipitate:
mX(aq)+nCO3 2- Xm(CO3)n(s)mX(aq)+nCO 3 2- X m (CO 3 ) n (s)
其中X是能够与一个或多个碳酸盐基团化学结合的任何元素或元素组合,m和n是化学计量正整数。wherein X is any element or combination of elements capable of chemically binding to one or more carbonate groups, and m and n are stoichiometric positive integers.
在进一步描述主题发明中,首先详细地描述根据本发明的实施方案的CO2埋存方法。然后描述可用于实施本发明的方法的各种实施方案的系统,随后使用本发明的方法和系统可以生产的组合物。In further describing the subject invention, a CO2 sequestration method according to an embodiment of the present invention is first described in detail. Systems that can be used to practice various embodiments of the methods of the invention are then described, followed by compositions that can be produced using the methods and systems of the invention.
CO2埋存的方法Methods of CO2 storage
在一些实施方案中,本发明提供CO2埋存方法。在这样的实施方案中,一定量的CO2可以从环境中,例如地球大气或由工业设备产生的气态废物物流中被除去或埋存,以便一些或全部的CO2不再存在于其中除去CO2的环境中。例如,CO2埋存除去了来自燃料燃烧的CO2或者防止来自燃料燃烧的CO2释放到大气中。在一些实施方案中,被埋存的CO2的形式是一种包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的组合物。这样的组合物可以包括溶液,包括沉淀材料的浆液,或单独的或者与用于建筑材料中的或用作建筑材料的一种或多种另外的材料结合的沉淀材料。例如,本发明的组合物可以包括沉淀材料,其包括碳酸酯化合物(例如无定形碳酸钙、方解石、霰石、vaterite等)。因此,在一些实施方案中,根据本发明各方面的CO2埋存产生了组合物(例如包括碳酸酯化合物的沉淀材料),其中组合物中至少部分的碳源自于人类所用的燃料(例如化石燃料)。本发明的CO2-埋存方法由一定量的CO2生产了存储稳定的产物,使得于是其中产生产物的CO2被埋存在该产物中。存储稳定的CO2-埋存产物是存储稳定的组合物,其将一定量的CO2引入存储稳定的形式,如地上、水下或地下存储稳定的形式,使得CO2不再以大气中的气体的形式存在或者CO2对于大气中的气体来说不再可得。因而,根据本发明的方法的CO2的埋存的结果是防止CO2气体进入大气并且以使得CO2没有变成大气一部分的方式允许长期存储CO2。In some embodiments, the present invention provides methods for CO2 sequestration. In such an embodiment, an amount of CO2 may be removed or sequestered from the environment, such as the Earth's atmosphere or a gaseous waste stream produced by an industrial facility, so that some or all of the CO2 is no longer present in it. CO removal 2 environment. For example, CO2 sequestration removes CO2 from fuel combustion or prevents CO2 from fuel combustion from being released into the atmosphere. In some embodiments, the form of CO2 that is sequestered is a form that includes carbonate, bicarbonate, or a combination of carbonate and bicarbonate. Such compositions may include solutions, slurries including precipitation materials, or precipitation materials alone or in combination with one or more additional materials used in or as building materials. For example, compositions of the present invention may include precipitation materials that include carbonate compounds (eg, amorphous calcium carbonate, calcite, aragonite, vaterite, etc.). Thus, in some embodiments, CO2 sequestration according to aspects of the invention results in a composition (e.g., a precipitation material comprising carbonate compounds) wherein at least a portion of the carbon in the composition is derived from fuels used by humans (e.g., Fossil fuels). The CO 2 -storage method according to the invention produces a storage-stable product from an amount of CO 2 such that the CO 2 in which the product was generated is then sequestered in the product. A storage-stable CO 2 -storage product is a storage-stable composition that introduces an amount of CO 2 into a storage-stable form, such as aboveground, underwater, or underground, such that the CO 2 is no longer available in the atmosphere gaseous form or CO2 is no longer available to the gas in the atmosphere. Thus, the sequestration of CO 2 according to the method of the invention prevents CO 2 gas from entering the atmosphere and allows long-term storage of CO 2 in such a way that CO 2 does not become part of the atmosphere.
本发明的方法的实施方案包括小-、中性-或负性-碳足迹方法。本发明的碳中性方法包括具有可忽略的碳足迹或没有碳足迹的方法。在负性-碳足迹方法中,通过方法的实施而埋存(例如通过将CO2转化为碳酸盐)的CO2的重量大于通过方法的实施而产生(例如通过电力生产、碱生产等)的CO2的重量。在有些情况下,通过实践方法所埋存的CO2的重量超过通过实践方法所产生的CO2的重量达1至100%、如5至100%、包括10至95%、10至90%、10至80%、10至70%、10至60%、10至50%、10至40%、10至30%、10至20%、20至95%、20至90%、20至80%、20至70%、20至60%、20至50%、20至40%、20至30%、30至95%、30至90%、30至80%、30至70%、30至60%、30至50%、30至40%、40至95%、40至90%、40至80%、40至70%、40至60%、40至50%、50至95%、50至90%、50至80%、50至70%、50至60%、60至95%、60至90%、60至80%、60至70%、70至95%、70至90%、70至80%、80至95%、80至90%、和90至95%。在有些情况下,通过实践方法所埋存的CO2的重量超过通过实践方法所产生的CO2的重量达5%或更多、达10%或更多、达15%或更多、达20%或更多、达30%或更多、达40%或更多、达50%或更多、达60%或更多、达70%或更多、达80%或更多、达90%或以上或达95%或更多。Embodiments of the methods of the present invention include small-, neutral- or negative-carbon footprint methods. Carbon neutral methods of the present invention include methods with negligible or no carbon footprint. In a negative-carbon footprint approach, the weight of CO2 that is stored (e.g. by converting CO2 to carbonate) through the implementation of the process is greater than that produced by the implementation of the process (e.g. by electricity production, alkali production, etc.) weight of CO2 . In some cases, the CO2 stored by the practical method exceeds the CO2 produced by the practical method by 1 to 100%, such as 5 to 100%, including 10 to 95%, 10 to 90%, 10 to 80%, 10 to 70%, 10 to 60%, 10 to 50%, 10 to 40%, 10 to 30%, 10 to 20%, 20 to 95%, 20 to 90%, 20 to 80%, 20 to 70%, 20 to 60%, 20 to 50%, 20 to 40%, 20 to 30%, 30 to 95%, 30 to 90%, 30 to 80%, 30 to 70%, 30 to 60%, 30 to 50%, 30 to 40%, 40 to 95%, 40 to 90%, 40 to 80%, 40 to 70%, 40 to 60%, 40 to 50%, 50 to 95%, 50 to 90%, 50 to 80%, 50 to 70%, 50 to 60%, 60 to 95%, 60 to 90%, 60 to 80%, 60 to 70%, 70 to 95%, 70 to 90%, 70 to 80%, 80 to 95%, 80 to 90%, and 90 to 95%. In some cases, the weight of CO stored by the practical method exceeds the weight of the CO generated by the practical method by 5% or more, by 10% or more, by 15% or more, by 20% % or more, up to 30% or more, up to 40% or more, up to 50% or more, up to 60% or more, up to 70% or more, up to 80% or more, up to 90% or above or up to 95% or more.
参考图1的系统,本发明提供了处理二氧化碳源(130)并且产生包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的组合物的基于水性的方法,其中二氧化碳源包括一个或多个除二氧化碳之外的另外的组分。在这样的实施方案中,二氧化碳的工业源可以作为源头,质子-除去剂源(140)可以作为源头,并且每一个可以被提供给处理器110来被处理(即,经受适宜条件以便产生包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的组合物)。在一些实施方案中,处理二氧化碳的工业源包括在接触器(例如但不限于气-液接触器或气-液-固接触器)中接触质子-除去剂源以从初始水溶液或浆液产生二氧化碳-加料的组合物,该组合物可以是溶液或浆液。在一些实施方案中,包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的组合物可以在接触器中由二氧化碳-加料的溶液或浆液产生。在一些实施方案中,二氧化碳-加料的溶液或浆液可以被提供给反应器,在该反应器中,可以产生包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的组合物。在一些实施方案中,组合物在接触器和反应器中产生。例如,在一些实施方案中,接触器可以产生包括碳酸氢盐的初始组合物,反应器可以由初始组合物产生包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的组合物。在一些实施方案中,本发明的方法可以进一步包括提供(sourcing)二价阳离子源,如碱土金属(例如Ca2+、Mg2+)源。在这样的实施方案中,二价阳离子源可以被提供给质子-除去剂源或者直接地提供给处理器。倘若由质子-除去剂源、由二价阳离子源或者由上述源的组合提供足够的二价阳离子,包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的组合物可以包括可分离的沉淀材料(例如CaCO3、MgCO3或其组合物)。无论来自处理器的组合物是否包括可分离的沉淀材料,在制造建筑材料中可以直接地使用来自处理器的组合物(任选地使用极少的后处理)。在一些实施方案中,直接来自处理器的包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的组合物(任选地使用极少的后处理)可以被注入到地下位置,如美国临时专利申请61/232,401(2009年8月7日申请,该申请全盘引入本文作为参考)中所述。Referring to the system of FIG. 1 , the present invention provides an aqueous-based method for processing a carbon dioxide source (130) and producing a composition comprising carbonates, bicarbonates, or carbonates and bicarbonates, wherein the carbon dioxide source comprises one or more other components besides carbon dioxide. In such an embodiment, an industrial source of carbon dioxide can be sourced, a proton-removing agent source (140) can be sourced, and each can be provided to
参考图2-5的系统,本发明提供了处理二氧化碳源(130)并且产生包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的组合物的基于水性的方法,其中二氧化碳源包括一个或多个除二氧化碳之外的另外的组分。除产生如参考图1所述的组合物之外,本发明进一步提供了处理包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的组合物的方法。因而,在一些实施方案中,本发明提供了处理二氧化碳源(130)以产生包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的组合物并且处理所产生的组合物的基于水性的方法。无论本发明的处理器产生的组合物是否包括可分离的沉淀材料,组合物可以直接提供到本发明的处理系统进行处理(例如浓缩、过滤等)。在一些实施方案中,组合物可以从处理器的接触器、反应器或澄清槽直接提供到处理系统。例如,不包含可分离的沉淀材料的处理器-产生的组合物可以直接提供到处理系统来浓缩组合物和产生上层清液。在另一非限制性实例中,包括可分离的沉淀材料的处理器-产生的组合物可以直接提供到处理系统进行液-固分离。处理器-产生的组合物可以提供到许多处理系统子系统中的任一个,该子系统包括但不局限于脱水系统、过滤系统或与过滤系统结合的脱水系统,其中处理系统或其子系统,分离上层清液与组合物以生产浓缩组合物(例如浓缩组合物就碳酸盐、碳酸氢盐或碳酸盐和碳酸盐来说是更浓缩的)。Referring to the systems of Figures 2-5, the present invention provides an aqueous-based method for processing a carbon dioxide source (130) and producing a composition comprising carbonate, bicarbonate, or carbonate and bicarbonate, wherein the carbon dioxide source comprises a or more additional components other than carbon dioxide. In addition to producing compositions as described with reference to Figure 1, the present invention further provides methods of treating compositions comprising carbonates, bicarbonates, or carbonates and bicarbonates. Thus, in some embodiments, the present invention provides an aqueous-based method for treating a carbon dioxide source (130) to produce a composition comprising carbonate, bicarbonate, or carbonate and bicarbonate and treating the resulting composition. method. Whether or not the composition produced by the processor of the invention includes separable precipitation material, the composition can be provided directly to the treatment system of the invention for processing (eg, concentration, filtration, etc.). In some embodiments, the composition may be provided directly to the treatment system from the contactor, reactor, or clarifier of the processor. For example, a processor-produced composition that does not contain separable precipitation material can be provided directly to a processing system to concentrate the composition and produce a supernatant. In another non-limiting example, a processor-generated composition comprising separable precipitation material can be provided directly to a processing system for liquid-solid separation. Processor-produced compositions may be provided to any of a number of treatment system subsystems, including but not limited to dewatering systems, filtration systems, or dewatering systems in combination with filtration systems, wherein the treatment system or its subsystems, The supernatant is separated from the composition to produce a concentrated composition (eg, the concentrated composition is more concentrated with respect to carbonate, bicarbonate, or carbonate and carbonate).
参考图3的系统,在一些实施方案中,本发明提供用于加料溶液以来自工业废气物流的CO2的方法以产生包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的组合物。在这样的实施方案中,在加料溶液以CO2前,溶液的pH值可以为6.5-14.0。在一些实施方案中,在加料溶液以CO2前,溶液的pH值可以为至少6.5,7.0,7.5,8.0,8.5,9.0,9.5,10.0,10.5,11.0,11.5,12.0,12.5,13.0,13.5或14.0。使用任何适宜的方法,包括但不限于,使用质子-除去剂和用于进行质子除去的电化学方法,可以增加溶液的pH值。在一些实施方案中,在加料溶液以CO2前,可以使用质子-除去剂来增加溶液的pH值。这样的质子-除去剂包括但不局限于氢氧化物(例如NaOH,KOH)和碳酸盐(例如Na2CO3,K2CO3)。在一些实施方案中,使用氢氧化钠来增加溶液的pH值。因而,在一些实施方案中,本发明提供用于加料碱性溶液(例如pH值大于7.0)以来自工业废气物流的CO2的方法以产生包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的组合物。Referring to the system of FIG. 3 , in some embodiments, the present invention provides a method for feeding a solution with CO from an industrial waste gas stream to produce a composition comprising carbonate, bicarbonate, or carbonate and bicarbonate . In such an embodiment, the pH of the solution may be 6.5-14.0 prior to feeding the solution with CO2 . In some embodiments, the pH of the solution may be at least 6.5, 7.0, 7.5, 8.0, 8.5, 9.0, 9.5, 10.0, 10.5, 11.0, 11.5, 12.0, 12.5, 13.0, 13.5 prior to feeding the solution with CO or 14.0. The pH of the solution may be increased using any suitable method, including, but not limited to, the use of proton-removing agents and electrochemical methods for effecting proton removal. In some embodiments, a proton-removing agent may be used to increase the pH of the solution prior to feeding the solution with CO 2 . Such proton-removing agents include, but are not limited to, hydroxides (eg NaOH, KOH) and carbonates (eg Na2CO3 , K2CO3 ). In some embodiments, sodium hydroxide is used to increase the pH of the solution. Thus, in some embodiments, the present invention provides methods for feeding an alkaline solution (e.g., pH greater than 7.0) with CO from an industrial waste gas stream to produce Composition of hydrogen salts.
在一些实施方案中,由加料碱性溶液以来自工业废物源(即,包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的溶液)的CO2产生的组合物可以是浆液或基本上澄清的溶液(即,基本上没有沉淀材料,如至少95%或更多没有),这取决于在溶液加料以CO2时溶液中可得的阳离子。如这里所述,溶液加料以CO2时,溶液可以,在一些实施方案中,包括二价阳离子如Ca2+,Mg2+或其组合。在这样的实施方案中,所得的组合物可以包括形成浆液的二价阳离子的碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐(例如沉淀材料)。这样的浆液,例如,可以包括CaCO3,MgCO3或其组合。在一些实施方案中,溶液可以包括不充足的二价阳离子从而在溶液加料以CO2时形成包括二价阳离子的碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的浆液。在这样的实施方案中,在溶液加料以CO2时,所得的组合物可以包括在基本上澄清溶液(即,基本上没有沉淀材料,如至少95%或更多没有)中的碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐。在一些实施方案中,例如,在溶液加料以CO2时,一价阳离子如Na+,K+或其组合(任选地通过添加NaOH和/或KOH)可以存在于基本上澄清溶液中。由加料这样的溶液以CO2产生的组合物可以包括,例如,一价阳离子的碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐。In some embodiments, the composition produced by feeding an alkaline solution with CO from an industrial waste source (i.e., a solution comprising carbonate, bicarbonate, or carbonate and bicarbonate) can be a slurry or substantially A clear solution (ie, substantially free of precipitated material, such as at least 95% or more free), depending on the cations available in the solution when the solution is fed with CO2 . As described herein, when the solution is fed with CO2 , the solution may, in some embodiments, include divalent cations such as Ca2+, Mg2+ or combinations thereof. In such embodiments, the resulting composition may include carbonate, bicarbonate, or both carbonate and bicarbonate salts of divalent cations forming a slurry (eg, precipitation material). Such a slurry, for example, may include CaCO 3 , MgCO 3 or combinations thereof. In some embodiments, the solution may include insufficient divalent cations to form a slurry including carbonates, bicarbonates, or carbonates and bicarbonates of divalent cations when the solution is fed with CO 2 . In such embodiments, when the solution is fed with CO, the resulting composition may include carbonates, carbonates, Bicarbonate or carbonate and bicarbonate. In some embodiments, for example, monovalent cations such as Na+, K+ or combinations thereof (optionally by addition of NaOH and/or KOH) may be present in a substantially clear solution when the solution is fed with CO2 . Compositions resulting from feeding such solutions with CO2 may include, for example, carbonates, bicarbonates, or both carbonates and bicarbonates of monovalent cations.
因而,在一些实施方案中,本发明提供加料碱性溶液(例如pH大于7.0)以来自工业废气物流的CO2以产生包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的组合物的方法,其中组合物是基本上澄清的(即,基本上没有沉淀材料,如至少95%或更多没有)。基本上澄清的组合物可以随后接触二价阳离子源(例如Ca2+,Mg2+或其组合)以产生包括二价阳离子的碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的组合物,这形成浆液。如上,这样的浆液可以包括CaCO3,MgCO3或其组合,其可以如本文中所述进行处理。在非限制性实例中,包括NaOH(例如溶于缺乏显著的二价阳离子的淡水的NaOH)的碱性溶液可以在气-液接触器中接触来自工业废气物流的CO2以产生包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的组合物,其中组合物是基本上澄清的,这应归于缺乏沉淀材料,而后者又应归于缺乏显著的二价阳离子。取决于所添加的CO2(和补充NaOH,如果有的话)的量,基本上澄清的组合物可以包括NaOH,NaHCO3和/或Na2CO3。基本上澄清的组合物可以随后在气-液接触器外的反应器中接触二价阳离子源(例如,Ca2+,Mg2+,Sr2+等)以产生包括二价阳离子的碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的组合物(例如,沉淀材料),这形成浆液。因而,组合物可以包括CaCO3和/或MgCO3,组合物可以如本文中所述的进行处理。例如,组合物可以经受液-固分离并且固体被制成水泥,辅助的粘结材料,细集料,灰浆,粗集料,混凝土,火山灰水泥或其组合。Thus, in some embodiments, the present invention provides for feeding an alkaline solution (e.g., pH greater than 7.0) to CO from an industrial waste gas stream to produce a composition comprising carbonate, bicarbonate, or carbonate and bicarbonate The method, wherein the composition is substantially clear (ie, substantially free of precipitated material, such as at least 95% or more free). The substantially clear composition can then be contacted with a source of divalent cations (e.g., Ca2+, Mg2+, or combinations thereof) to produce a composition comprising carbonate, bicarbonate, or carbonate and bicarbonate of divalent cations, which form slurry. As above, such a slurry may include CaCO 3 , MgCO 3 or combinations thereof, which may be processed as described herein. In a non-limiting example, an alkaline solution comprising NaOH (such as NaOH dissolved in fresh water lacking significant divalent cations) can be contacted with CO from an industrial waste gas stream in a gas-liquid contactor to produce , bicarbonate, or a combination of carbonate and bicarbonate, wherein the composition is substantially clear due to the absence of precipitating material which in turn is due to the absence of significant divalent cations. Depending on the amount of CO2 (and make-up NaOH, if any) added, the substantially clear composition may include NaOH, NaHCO3 and/or Na2CO3 . The substantially clear composition can then be contacted with a source of divalent cations (e.g., Ca2+, Mg2+, Sr2+, etc.) in a reactor outside the gas-liquid contactor to produce carbonate, bicarbonate, or carbon containing divalent cations. A combination of acid salts and bicarbonates (eg, precipitation material), which forms a slurry. Thus, the composition may include CaCO3 and/or MgCO3 , and the composition may be processed as described herein. For example, the composition can undergo liquid-solid separation and the solids can be made into cement, secondary binding material, fine aggregate, mortar, coarse aggregate, concrete, pozzolan cement, or combinations thereof.
参考图4和5的系统,本发明还提供了处理二氧化碳源(130)并且产生包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的组合物的基于水性的方法,其中二氧化碳源包括一个或多个除二氧化碳之外的另外的组分,并且其中至少一部分处理系统上层清液被再循环。例如,在一些实施方案中,本发明提供了在处理器中处理废气物流包括CO2和,任选地,SOx、NOx、和/或Hg以产生被处理的废气物流(例如,纯净气体物流适用于释放到环境中),包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的组合物,和排出物的方法,其中至少一部分排出物被再循环到处理器。如图4和5所示,来自处理系统(其可以包括脱水系统和过滤系统)的上层清液可以以各种方式进行再循环。因而,在一些实施方案中,可以使用来自脱水系统、过滤系统或脱水系统和过滤系统的组合的上层清液的至少一部分来处理二氧化碳。上层清液可以被提供到二氧化碳-处理系统处理器。在这样的实施方案中,上层清液可以被提供到接触器(例如,气-液接触器,气-液-固接触器),反应器,接触器和反应器的组合,或用于处理二氧化碳的任何其它单元或单元的组合。另外,在一些实施方案中,至少一部分来自处理系统的上层清液可以被提供到洗涤系统。在这样的实施方案中,可以使用上层清液来洗涤本发明的组合物(例如,包括CaCO3,MgCO3或其组合的沉淀材料)。例如,可以使用上层清液来洗涤来自基于碳酸盐的沉淀材料的氯化物。参考图5,至少一部分处理系统上层清液可以被提供到电化学系统。因而,可以使用处理系统上层清液来产生质子-除去剂或者进行用于处理二氧化碳的质子除去。在一些实施方案中,至少一部分来自处理系统的上层清液可以被提供到不同的系统或工艺。例如,至少一部分处理系统上层清液可以被提供到脱盐装置或脱盐工艺使得处理系统上层清液,其在被用于处理二氧化碳后通常是比其它可用的进料(例如,海水,盐水等)更软化的(即,较低浓度的Ca2+和/或Mg2+),可以被脱盐用于饮用水。Referring to the systems of Figures 4 and 5, the present invention also provides an aqueous based method of processing a carbon dioxide source (130) and producing a composition comprising carbonate, bicarbonate, or carbonate and bicarbonate, wherein the carbon dioxide source comprises One or more additional components other than carbon dioxide, and wherein at least a portion of the treatment system supernatant is recycled. For example, in some embodiments, the present invention provides for treating a waste gas stream comprising CO and , optionally, SOx, NOx, and/or Hg in a processor to produce a treated waste gas stream (e.g., a pure gas stream suitable for for release into the environment), including carbonates, bicarbonates, or combinations of carbonates and bicarbonates, and effluents in which at least a portion of the effluents are recycled to the processor. As shown in Figures 4 and 5, the supernatant from the treatment system (which may include a dewatering system and a filtration system) can be recycled in various ways. Thus, in some embodiments, at least a portion of the supernatant from a dehydration system, a filtration system, or a combination of a dehydration system and a filtration system can be used to treat carbon dioxide. The supernatant can be provided to a carbon dioxide-treatment system processor. In such embodiments, the supernatant may be provided to a contactor (e.g., a gas-liquid contactor, a gas-liquid-solid contactor), a reactor, a combination of a contactor and a reactor, or used to treat carbon dioxide any other unit or combination of units. Additionally, in some embodiments, at least a portion of the supernatant from the treatment system can be provided to the wash system. In such embodiments, the supernatant may be used to wash compositions of the invention (eg, precipitation materials comprising CaCO 3 , MgCO 3 , or combinations thereof). For example, the supernatant can be used to wash chloride from the carbonate-based precipitation material. Referring to Figure 5, at least a portion of the treatment system supernatant may be provided to the electrochemical system. Thus, the treatment system supernatant can be used to generate proton-removing agents or to perform proton removal for the treatment of carbon dioxide. In some embodiments, at least a portion of the supernatant from the treatment system can be provided to a different system or process. For example, at least a portion of the treatment system supernatant may be provided to a desalination unit or desalination process such that the treatment system supernatant, which after being used to treat carbon dioxide, is generally more concentrated than other available feeds (e.g., seawater, brine, etc.) Softened (ie, lower concentrations of Ca2+ and/or Mg2+), can be desalinated for drinking water.
处理系统上层清液的再循环是有益的,因为再循环提供了可用资源的有效利用;周围环境的最小干扰;和降低的能量需要,所述降低的能量需要提供了本发明的系统和方法的更低的碳足迹。当本发明的二氧化碳-处理系统可操作地连接到工业设备(例如,燃烧化石燃料的发电厂如燃煤的发电厂)并且使用在工业设备处产生的电力,由处理系统上层清液的再循环提供的降低的能量需要提供了降低的能量需要。当表示为百分数时,给定的工艺、装置或系统的能量需要是相对于该工艺、装置或系统所连接的或者使用的发电厂的总输出,由该工艺、装置或系统所消耗的能量。没有为再循环配置的二氧化碳-处理系统(即,为单程法配置的二氧化碳处理系统)如图2中所示的那种可以具有至少10%的工业设备的能量需要,这可归因于连续地将碱度的新鲜源(例如,海水,盐水)泵入系统。在这样的实例中,100兆瓦发电厂(例如,燃煤的发电厂)将需要将10兆瓦的电力提供给二氧化碳-处理系统用于连续地将碱度的新鲜源泵入系统。相比之下,为再循环配置的系统如图4或图5中所示的那种可以具有小于10%,如小于8%,包括小于6%,例如,小于4%或者少于2%的工业设备的能量需要,该能量需要可能是可归因于泵送补给水和再循环上层清液。为再循环配置的二氧化碳-处理系统可以,当相比于为单程法设计的系统时,显示出至少2%,如至少5%,包括至少10%,例如,至少25%或至少50%的能量需要的下降。例如,如果为再循环配置的二氧化碳-处理系统消耗9兆瓦的电力来泵送补给水和再循环上层清液并且为单程法设计的二氧化碳-处理系统消耗可归因于泵送的10兆瓦,那么为再循环配置的二氧化碳-处理系统显示出10%的能量需要的下降。对于如图4和5中所示的那些的系统(即,为再循环配置的二氧化碳-处理系统)来说,可归因于泵送和再循环的能量需要的下降也可提供总能量需要的下降,特别地当相比于为单程法配置的二氧化碳-处理系统时。在一些实施方案中,再循环提供了二氧化碳-处理系统的总能量需要的下降,其中下降为至少2%,如至少4%,包括至少6%,例如至少8%或至少10%,当相比于为单程法配置的二氧化碳-处理系统的总能量需求时。例如,如果为再循环配置的二氧化碳-处理系统具有15%的能量需要并且为单程法设计的二氧化碳-处理系统具有20%能量需要,那么为再循环配置的二氧化碳-处理系统显示出5%的总能量需要的下降。例如,为再循环配置的二氧化碳-处理系统,其中再循环包括通过过滤单元如纳滤单元的过滤(例如,以便在渗余物中浓缩二价阳离子和降低在渗透物中的二价阳离子),可以具有至少2%,如至少4%,包括至少6%,例如至少8%或至少10%的总能量需要的下降,当相比于为单程法配置的二氧化碳-处理系统时。Recirculation of the supernatant of the treatment system is beneficial because the recirculation provides efficient use of available resources; minimal disturbance of the surrounding environment; and reduced energy requirements that provide the benefits of the systems and methods of the present invention. Lower carbon footprint. When the carbon dioxide-processing system of the present invention is operably connected to an industrial facility (e.g., a fossil fuel-fired power plant such as a coal-fired power plant) and uses electricity generated at the industrial facility, the recirculation of supernatant liquid from the treatment system The provided reduced energy requirement provides a reduced energy requirement. When expressed as a percentage, the energy requirement for a given process, plant or system is the energy consumed by the process, plant or system relative to the total output of the power plant to which the process, plant or system is connected or used. A carbon dioxide-processing system not configured for recirculation (i.e., a carbon dioxide processing system configured for a single-pass process) such as that shown in Figure 2 can have at least 10% of the energy requirements of an industrial plant, which can be attributed to continuous A fresh source of alkalinity (eg, seawater, brine) is pumped into the system. In such an example, a 100 MW power plant (eg, a coal-fired power plant) would need to provide 10 MW of power to the carbon dioxide-processing system for continuously pumping a fresh source of alkalinity into the system. In contrast, a system configured for recirculation such as that shown in Figure 4 or Figure 5 may have a Energy requirements of industrial plants, which may be attributable to pumping make-up water and recirculating supernatant. A carbon dioxide-processing system configured for recirculation may exhibit at least 2%, such as at least 5%, including at least 10%, for example, at least 25% or at least 50% energy when compared to a system designed for a single pass process drop in need. For example, if a CO2-treatment system configured for recirculation consumes 9 MW of electricity to pump the make-up water and recirculate the supernatant and a CO2-treatment system designed for the single-pass process consumes 10 MW of electricity attributable to pumping , then a carbon dioxide-treatment system configured for recirculation shows a 10% drop in energy requirements. For systems such as those shown in Figures 4 and 5 (i.e., carbon dioxide-processing systems configured for recirculation), the reduction in energy requirements attributable to pumping and recirculation can also provide a reduction in the total energy requirements. decline, especially when compared to carbon dioxide-processing systems configured for single-pass processes. In some embodiments, recycling provides a reduction in the total energy requirement of the carbon dioxide-processing system, wherein the reduction is at least 2%, such as at least 4%, including at least 6%, such as at least 8% or at least 10%, when compared to At the total energy demand of a carbon dioxide-treatment system configured for a single-pass process. For example, if a CO2-treatment system configured for recirculation has an energy requirement of 15% and a CO2-treatment system designed for a one-pass process has a 20% energy requirement, then a CO2-treatment system configured for recirculation shows an overall energy requirement of 5%. Decreased energy requirements. For example, a carbon dioxide-processing system configured for recirculation, wherein recirculation includes filtration through a filtration unit such as a nanofiltration unit (e.g., to concentrate divalent cations in the retentate and reduce divalent cations in the permeate), There may be a reduction in total energy requirement of at least 2%, such as at least 4%, including at least 6%, such as at least 8% or at least 10%, when compared to a carbon dioxide-processing system configured for a single pass process.
通过有效利用其它资源,本发明的二氧化碳-加工方法、装置和系统的能量需要可以被进一步降低。在一些实施方案中,通过有效利用来自工业源的热量,本发明的二氧化碳-加工系统的能量需要可以被进一步降低。在一些实施方案中,例如,来自二氧化碳的工业源的热量(例如,来自燃煤的发电厂的烟道气热量)可以被用于干燥包括沉淀材料(包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐)的组合物。在这样的实施方案中,喷雾干燥器可被用来喷雾干燥组合物。例如,通过热交换器可以利用低品位(例如,150-200℃)废热来蒸发喷雾干燥包括沉淀材料的组合物。另外,利用来自二氧化碳的工业源的热量干燥本发明的组合物允许同时冷却二氧化碳的工业源(例如,来自燃煤的发电厂的烟道气),这提高了二氧化碳的溶解,一种与温度成反比的过程。在一些实施方案中,本发明的二氧化碳-处理系统的能量需要可以进一步通过有效利用压力来降低。例如,在一些实施方案中,本发明的二氧化碳-处理系统配置以能量回收系统。这样的能量回收系统是已知的,例如,在脱盐领域中,并且通过压力交换来操作。在一些实施方案中,当捕获和处理70-90%的从工业设备(例如,燃煤的发电厂)排放的二氧化碳时,二氧化碳-处理系统的总能量需要可小于99.9%、90%、80%、70%、60%、50%、40%、30%、20%、15%、10%、5%或3%。例如,在一些实施方案中,当捕获和处理70-90%的从工业设备(例如,燃煤的发电厂)排放的二氧化碳,二氧化碳-处理系统的总能量需要可小于30%,如小于20%,包括小于15%,例如,小于10%,小于5%或小于3%。因而,为再循环、热交换和/或压力交换配置的本发明的二氧化碳-处理系统可以降低提供电力的工业设备的附加负载,同时保持二氧化碳处理能力。The energy requirements of the carbon dioxide-processing method, apparatus and system of the present invention can be further reduced by efficient use of other resources. In some embodiments, the energy requirements of the carbon dioxide-processing systems of the present invention can be further reduced by efficient use of heat from industrial sources. In some embodiments, for example, heat from an industrial source of carbon dioxide (e.g., flue gas heat from a coal-fired power plant) can be used to dry salts and bicarbonates). In such embodiments, a spray dryer may be used to spray dry the composition. For example, low grade (eg, 150-200° C.) waste heat can be utilized by a heat exchanger to evaporatively spray dry compositions including precipitation material. In addition, drying the compositions of the present invention using heat from an industrial source of carbon dioxide allows simultaneous cooling of an industrial source of carbon dioxide (e.g., flue gas from a coal-fired power plant), which enhances the dissolution of carbon dioxide, a temperature-dependent inverse process. In some embodiments, the energy requirements of the carbon dioxide-processing systems of the present invention can be further reduced through efficient use of pressure. For example, in some embodiments, the carbon dioxide-processing system of the present invention is configured with an energy recovery system. Such energy recovery systems are known, for example, in the field of desalination and operate by pressure exchange. In some embodiments, when capturing and processing 70-90% of the carbon dioxide emitted from industrial facilities (e.g., coal-fired power plants), the total energy requirement of the carbon dioxide-processing system can be less than 99.9%, 90%, 80% , 70%, 60%, 50%, 40%, 30%, 20%, 15%, 10%, 5%, or 3%. For example, in some embodiments, when capturing and treating 70-90% of the carbon dioxide emitted from industrial facilities (e.g., coal-fired power plants), the total energy requirement of the carbon dioxide-processing system may be less than 30%, such as less than 20% , including less than 15%, eg, less than 10%, less than 5% or less than 3%. Thus, a carbon dioxide-processing system of the present invention configured for recirculation, heat exchange, and/or pressure exchange can reduce the parasitic load on industrial equipment providing power while maintaining carbon dioxide processing capacity.
不可避免地,本文中所述的再循环及其它方法消耗水,因为水可能变成本发明的组合物的一部分(例如,沉淀材料包括,例如,无定形碳酸钙CaCO3·H2O;三水菱镁矿MgCO3·2H2O;等),可能通过干燥(例如,喷雾干燥)本发明的组合物而蒸发,或者在该方法的一些其它部分中损失。因而,可以提供补给水来计算为处理二氧化碳以产生本发明的组合物(例如,喷雾干燥的沉淀材料)而损失的水。例如,总计小于700,000加仑/日的补给水可以替换为从35兆瓦燃煤的发电厂的烟道气中产生例如喷雾干燥的沉淀材料而损失的水。仅仅需要补给水的方法可以被认为是零工艺水排放方法(即零液体废物方法)。在其中使用除补给水以外的另外的水的方法中,该水可以源自本文中所述的任何水源(例如,海水,盐水等)。在一些实施方案中,例如,水可以源自于发电厂冷却物流并且在闭环系统中返回到该物流。需要补给水和另外的工艺水的方法被认为是低工艺水排放方法,因为本发明的系统和方法被设计成有效地使用资源。Inevitably, the recycling and other methods described herein consume water as it may become part of the compositions of the present invention (e.g., precipitation materials include, e.g., amorphous calcium carbonate CaCO 3 ·H 2 O; three Hydromagnesite MgCO 3 ·2H 2 O; etc.), may be evaporated by drying (eg, spray drying) the composition of the invention, or lost in some other part of the process. Thus, make-up water can be provided to account for the water lost to process carbon dioxide to produce compositions of the invention (eg, spray-dried precipitation material). For example, a total of less than 700,000 gallons/day of make-up water could be replaced by water lost from producing, for example, spray-dried precipitation material from the flue gas of a 35 MW coal-fired power plant. A method requiring only make-up water may be considered a zero process water discharge method (ie, a zero liquid waste method). In methods where additional water is used in addition to make-up water, the water can be derived from any of the water sources described herein (eg, seawater, brine, etc.). In some embodiments, for example, water may be sourced from a power plant cooling stream and returned to this stream in a closed loop system. Processes that require make-up water and additional process water are considered low process water discharge processes because the systems and methods of the present invention are designed to use resources efficiently.
在一些实施方案中,本发明提供使一定体积的水溶液与二氧化碳源接触以产生包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的组合物,其中组合物是溶液或浆液。为产生包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的沉淀材料,本发明的方法包括使一定体积的含二阶阳离子的水溶液与CO2源接触并且使所得的溶液经受促进沉淀的条件。二价阳离子可以来自许多不同的二价阳离子源中的任一种,这取决于在特定位置的可用性。这样的源头包括工业废物,海水,盐水,硬水,岩石和矿物(例如,石灰,方镁石,包括金属硅酸盐的材料如蛇纹石和橄榄石),和任何其它适合的源头。In some embodiments, the present invention provides contacting a volume of an aqueous solution with a carbon dioxide source to produce a composition comprising carbonate, bicarbonate, or carbonate and bicarbonate, wherein the composition is a solution or a slurry. To produce precipitation material comprising carbonates, bicarbonates, or carbonates and bicarbonates, the method of the invention comprises contacting a volume of an aqueous solution containing secondary cations with a source of CO and subjecting the resulting solution to facilitated precipitation conditions of. The divalent cations can come from any of a number of different sources of divalent cations, depending on availability at a particular location. Such sources include industrial waste, seawater, brine, hard water, rocks and minerals (eg, lime, periclase, materials including metal silicates such as serpentine and olivine), and any other suitable source.
在一些位置中,来自各种工业方法(即,工业废物物流)的废物物流提供了适宜的二价阳离子源(以及质子-除去剂如金属氢氧化物)。这样的废物物流包括但不局限于矿业废物;灰(例如,煤灰如飞灰,炉底灰,锅炉渣);熔渣(例如铁熔渣,磷熔渣);水泥窑废物(例如,水泥窑灰尘);炼油厂/石化精炼厂废物(例如油田和沼气层盐水);煤层废物(例如采气盐水和煤层盐水);纸处理废物;水软化废物盐水(例如,离子交换排出物);硅加工废物;农业废物;金属抛光废物;高pH纺织废物;和碱渣。灰、水泥窑灰尘和熔渣,总起来说,金属氧化物的废物源头,其进一步描述于美国专利申请12/486,692(申请日为2009年6月17日,其全盘引入本文作为参考),可以与包括金属硅酸盐的材料以任何组合的形式使用,后者进一步描述于美国专利申请12/501,217(申请日为2009年7月10日,其也全盘引入本文作为参考)。为了实施本发明,可以混合搭配本文中所述的任何二价阳离子源。为了实施本发明,例如,包括金属硅酸盐(例如,硅酸镁矿物如橄榄石,蛇纹石,等等)的材料可以与本文中所述的任何二价阳离子源结合。In some locations, waste streams from various industrial processes (ie, industrial waste streams) provide suitable sources of divalent cations (as well as proton-removing agents such as metal hydroxides). Such waste streams include, but are not limited to, mining waste; ash (e.g., coal ash such as fly ash, bottom ash, boiler slag); slag (e.g., iron slag, phosphorus slag); cement kiln waste (e.g., cement kiln dust); refinery/petrochemical refinery waste (e.g. oil field and biogas layer brine); coal seam waste (e.g. gas extraction brine and coal seam brine); paper processing waste; water softening waste brine (e.g. ion exchange effluent); Processing waste; agricultural waste; metal polishing waste; high pH textile waste; Ash, cement kiln dust, and slag, in general, waste sources of metal oxides, which are further described in U.S. Patent Application 12/486,692 (filed June 17, 2009, which is incorporated herein by reference in its entirety), can be Use in any combination with materials including metallosilicates, which are further described in US Patent Application 12/501,217 (filed July 10, 2009, which is also incorporated herein by reference in its entirety). To practice the present invention, any of the sources of divalent cations described herein may be mixed and matched. To practice the invention, for example, materials including metal silicates (eg, magnesium silicate minerals such as olivine, serpentine, etc.) can be combined with any of the sources of divalent cations described herein.
在一些位置中,用于制备本发明的组合物(例如,包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的沉淀材料)的适宜二价阳离子源是水(例如,包括二价阳离子的水溶液如海水或盐水),后者可以变化,这取决于实施本发明的特定的位置。可使用的合适的二价阳离子的水溶液包括含一种或多种二价阳离子(例如,碱土金属阳离子如Ca2+和Mg2+)的溶液。在一些实施方案中,水性二价阳离子源包括碱土金属阳离子。在一些实施方案中,碱土金属阳离子包括钙,镁或其混合物。在一些实施方案中,二价阳离子的水溶液包括数量为50至50,000ppm,50至40,000ppm,50至20,000ppm,100至10,000ppm,200至5000ppm或400至1000ppm的钙。在一些实施方案中,二价阳离子的水溶液包括数量为50至40,000ppm,50至20,000ppm,100至10,000ppm,200至10,000ppm,500至5000ppm或500至2500ppm的镁。在一些实施方案中,在Ca2+和Mg2+都存在的情况下,在二价阳离子的水溶液中的Ca2+与Mg2+的比值(即,Ca2+∶Mg2+)为1∶1至1∶2.5;1∶2.5至1∶5;1∶5至1∶10;1∶10至1∶25;1∶25至1∶50;1∶50至1∶100;1∶100至1∶150;1∶150至1∶200;1∶200至1∶250;1∶250至1∶500;1∶500至1∶1000或其范围。例如,在一些实施方案中,在二价阳离子的水溶液中Ca2+与Mg2+的比值在1∶1和1∶10;1∶5和1∶25;1∶10和1∶50;1∶25和1∶100;1∶50和1∶500;或1∶100和1∶1000之间。在一些实施方案中,在二价阳离子的水溶液中Mg2+与Ca2+的比值(即,Mg2+∶Ca2+)为1∶1至1∶2.5;1∶2.5至1∶5;1∶5至1∶10;1∶10至1∶25;1∶25至1∶50;1∶50至1∶100;1∶100至1∶150;1∶150至1∶200;1∶200至1∶250;1∶250至1∶500;1∶500至1∶1000或其范围。例如,在一些实施方案中,在二价阳离子的水溶液中Mg2+与Ca2+的比值在1∶1和1∶10;1∶5和1∶25;1∶10和1∶50;1∶25和1∶100;1∶50和1∶500;或1∶100和1∶1000之间。In some locations, a suitable source of divalent cations for use in preparing compositions of the invention (e.g., comprising carbonate, bicarbonate, or precipitation material of carbonate and bicarbonate) is water (e.g., comprising divalent Aqueous solutions of cations such as seawater or brine), the latter can vary depending on the particular location in which the invention is practiced. Suitable aqueous solutions of divalent cations that may be used include solutions containing one or more divalent cations (eg, alkaline earth metal cations such as Ca2+ and Mg2+). In some embodiments, the aqueous source of divalent cations includes alkaline earth metal cations. In some embodiments, the alkaline earth metal cations include calcium, magnesium, or mixtures thereof. In some embodiments, the aqueous solution of divalent cations includes calcium in an amount of 50 to 50,000 ppm, 50 to 40,000 ppm, 50 to 20,000 ppm, 100 to 10,000 ppm, 200 to 5000 ppm, or 400 to 1000 ppm. In some embodiments, the aqueous solution of divalent cations includes magnesium in an amount of 50 to 40,000 ppm, 50 to 20,000 ppm, 100 to 10,000 ppm, 200 to 10,000 ppm, 500 to 5000 ppm, or 500 to 2500 ppm. In some embodiments, the ratio of Ca2+ to Mg2+ (i.e., Ca2+:Mg2+) in the aqueous solution of divalent cations in the presence of both Ca2+ and Mg2+ is 1:1 to 1:2.5; 1:2.5 to 1 :5; 1:5 to 1:10; 1:10 to 1:25; 1:25 to 1:50; 1:50 to 1:100; 1:100 to 1:150; 1:150 to 1:200 ; 1:200 to 1:250; 1:250 to 1:500; 1:500 to 1:1000 or a range thereof. For example, in some embodiments, the ratio of Ca2+ to Mg2+ in an aqueous solution of divalent cations is between 1:1 and 1:10; 1:5 and 1:25; 1:10 and 1:50; 1:25 and 1 :100; between 1:50 and 1:500; or between 1:100 and 1:1000. In some embodiments, the ratio of Mg2+ to Ca2+ (i.e., Mg2+:Ca2+) in the aqueous solution of divalent cations is 1:1 to 1:2.5; 1:2.5 to 1:5; 1:5 to 1:10; 1:10 to 1:25; 1:25 to 1:50; 1:50 to 1:100; 1:100 to 1:150; 1:150 to 1:200; 1:200 to 1:250; 1: 250 to 1:500; 1:500 to 1:1000 or the range thereof. For example, in some embodiments, the ratio of Mg2+ to Ca2+ in an aqueous solution of divalent cations is between 1:1 and 1:10; 1:5 and 1:25; 1:10 and 1:50; 1:25 and 1 :100; between 1:50 and 1:500; or between 1:100 and 1:1000.
由其制备本发明的组合物(例如,沉淀材料)的存在于二价阳离子源中的一种或多种组分可以被用于标识所使用的二价阳离子源。这些标识组分和其数量可以被称为“来源标识符”或“标记”。例如,如果二价阳离子源是海水,可以存在于本发明的组合物(例如,沉淀材料)中的来源标识符或标记包括但不局限于氯,钠,硫,钾,溴,硅,锶等。这样的元素可以以任何已知的化合价存在于组合物中。任何这样的来源标识符或标记可以以少量存在,例如,20,000ppm或更少,2000ppm或更少,200ppm或更少或20ppm或更少。在一些实施方案中,例如,标记是锶。在本发明的沉淀材料中,锶可以被引入到霰石晶格中,并且构成霰石晶格的10,000ppm或更少,在某些实施方案中,3至10,000ppm,如5至5000ppm,包括5至1000ppm,例如,5至500ppm或5至100ppm。来源标识符可以不同,这取决于用于产生本发明的组合物所使用的特定的二价阳离子源(例如,盐水)。在一些实施方案中,至少部分地由于二价阳离子源,本发明的组合物(例如,沉淀材料)的碳酸钙含量可以是25%w/w或更高,如40%w/w或更高,包括50%w/w或更高,例如,60%w/w或更高。在一些实施方案中,这样的组合物具有受由其产生组合物的二价阳离子源的影响的钙∶镁比值,并且因此反映了所述二价阳离子源。在一些实施方案中,钙∶镁摩尔比为10∶1至1∶5Ca∶Mg,如5∶1至1∶3Ca∶Mg。在一些实施方案中,组合物特征为具有来源标识碳酸盐∶氢氧化物化合物比值,其中该比值例如为100至1,10至1,或1至1。One or more components present in the source of divalent cations from which the compositions of the invention (eg, precipitation material) are prepared can be used to identify the source of divalent cations used. These identifying components and their quantities may be referred to as "source identifiers" or "markers". For example, if the source of divalent cations is seawater, source identifiers or labels that may be present in compositions of the invention (e.g., precipitation material) include, but are not limited to, chlorine, sodium, sulfur, potassium, bromine, silicon, strontium, etc. . Such elements may be present in the composition at any known valency. Any such source identifiers or indicia may be present in small amounts, for example, 20,000 ppm or less, 2000 ppm or less, 200 ppm or less or 20 ppm or less. In some embodiments, for example, the label is strontium. In the precipitation material of the present invention, strontium may be incorporated into the aragonite lattice and constitute 10,000 ppm or less of the aragonite lattice, in certain embodiments, 3 to 10,000 ppm, such as 5 to 5000 ppm, including 5 to 1000ppm, eg, 5 to 500ppm or 5 to 100ppm. The source identifier can vary depending on the particular source of divalent cations (eg, saline) used to produce the compositions of the invention. In some embodiments, due at least in part to the source of divalent cations, the calcium carbonate content of the compositions (e.g., precipitation materials) of the invention may be 25% w/w or higher, such as 40% w/w or higher , including 50% w/w or higher, eg, 60% w/w or higher. In some embodiments, such compositions have a calcium:magnesium ratio that is influenced by, and thus reflects, the source of divalent cations from which the composition is derived. In some embodiments, the calcium:magnesium molar ratio is 10:1 to 1:5 Ca:Mg, such as 5:1 to 1:3 Ca:Mg. In some embodiments, the composition is characterized as having a source-identifying carbonate:hydroxide compound ratio, wherein the ratio is, for example, 100 to 1, 10 to 1, or 1 to 1.
二价阳离子的水溶液可以包括源自淡水,微咸水,海水或盐水(例如,天然存在的盐水或人为的盐水如地热装置废水,脱盐装置废水,合成盐水,包括含溶解矿物的合成盐水),以及盐度大于淡水的其它水溶液(其中任一种可以是天然存在的或人为的)的二价阳离子。为了方便起见,在描述本发明中,淡水可以被认为具有小于0.5ppt(千分率)的盐度。微咸水可以包括比淡水更多的盐,但是没有像海水那样多的盐。微咸水可以被认为具有约0.5-约35ppt的盐度。海水可以是来自海、海洋或者盐度范围为约35-约50ppt的任何其它水体的水。盐水可以具有约50ppt或更多的盐度。因而,盐水可以是用盐饱和或几乎饱和的水。在一些实施方案中,二价阳离子源自其中的水源是富含矿物的(例如,富含钙的和/或富含镁的)淡水源。在一些实施方案中,二价阳离子源自其中的水源是天然存在的盐水源,其选自海,海洋,湖泊,沼泽,河口,泻湖,地表盐水,深盐水,碱湖,内海等等。在一些实施方案中,二价阳离子源自其中的水源是地表盐水。在一些实施方案中,二价阳离子源自其中的水源是地下盐水。在一些实施方案中,二价阳离子源自其中的水源是深盐水。在一些实施方案中,二价阳离子源自其中的水源是Ca-Mg-Na-(K)-Cl;Na-(Ca)-SO4-Cl;Mg-Na-(Ca)-SO4-Cl;Na-CO3-Cl;或Na-CO3-SO4-Cl盐水。在一些实施方案中,二价阳离子源自其中的水源是人为的盐水,其选自地热装置废水或脱盐废水。在一些实施方案中,水源还可包含以硼酸盐形式存在的硼,包括但不限于,尤其,BO3 3-、B2O5 4-、B3O7 5-和B4O9 6-。Aqueous solutions of divalent cations may include those derived from fresh water, brackish water, sea water, or brine (e.g., naturally occurring brines or anthropogenic brines such as geothermal plant effluents, desalination plant effluents, synthetic brines, including those containing dissolved minerals), and divalent cations of other aqueous solutions (either of which may be naturally occurring or anthropogenic) of greater salinity than fresh water. For convenience, in describing the present invention, fresh water may be considered to have a salinity of less than 0.5 ppt (parts per thousand). Brackish water can include more salt than fresh water, but not as much salt as seawater. Brackish water can be considered to have a salinity of about 0.5 to about 35 ppt. Seawater may be water from the sea, ocean, or any other body of water with a salinity in the range of about 35 to about 50 ppt. The brine may have a salinity of about 50 ppt or more. Thus, brine may be water saturated or nearly saturated with salt. In some embodiments, the water source from which the divalent cations are derived is a mineral-rich (eg, calcium-rich and/or magnesium-rich) freshwater source. In some embodiments, the water source from which the divalent cations are derived is a naturally occurring brine source selected from seas, oceans, lakes, swamps, estuaries, lagoons, surface brines, deep brines, soda lakes, inland seas, and the like. In some embodiments, the water source from which the divalent cations are derived is surface brine. In some embodiments, the water source from which the divalent cations are derived is subterranean brine. In some embodiments, the water source from which the divalent cations are derived is deep brine. In some embodiments, the water source from which the divalent cations are derived is Ca-Mg-Na-(K)-Cl; Na-(Ca) -SO4 -Cl; Mg-Na-(Ca) -SO4 -Cl ; Na-CO 3 -Cl; or Na-CO 3 -SO 4 -Cl brine. In some embodiments, the water source from which the divalent cations are derived is an anthropogenic brine selected from geothermal plant wastewater or desalination wastewater. In some embodiments, the water source may also contain boron in the form of borates, including but not limited to, inter alia, BO 3 3- , B 2 O 5 4- , B 3 O 7 5- , and B 4 O 9 6 - .
淡水常常是适宜的二价阳离子源(例如,碱土金属的阳离子如Ca2+和Mg2+)。可以使用许多合适的淡水源中任一种,包括从相对不含矿物的来源至相对富含矿物的来源的范围的淡水源。富含矿物的淡水源可能是天然存在的,包括许多硬水源、湖泊或内海中的任一种。一些富含矿物的淡水源如碱湖或内海(例如,土耳其的Lake Van)也提供了pH-改变剂的来源。富含矿物的淡水源也可是人为的。例如,矿物-贫乏的(软)水可以与二价阳离子源如碱土金属阳离子(例如,Ca2+,Mg2+,等等)接触以产生富含矿物的水,其适合于本文中所述的方法和系统。使用任何适宜的流程(例如,添加固体、悬浮液或溶液),二价阳离子或其前体(例如盐、矿物)可以被添加到淡水(或本文中所述的任何其它类型的水)。在一些实施方案中,将选自Ca2+和Mg2+的二价阳离子添加到淡水。在一些实施方案中,选自Na+和K+的一价阳离子被添加到淡水。在一些实施方案中,包括Ca2+的淡水结合以包括金属硅酸盐的材料,灰(例如,飞灰、炉底灰、锅炉渣),或其产物或加工形式,包括上述的组合,这形成了包括钙和镁阳离子的溶液。Fresh water is often a suitable source of divalent cations (eg, cations of alkaline earth metals such as Ca2+ and Mg2+). Any of a number of suitable freshwater sources may be used, including freshwater sources ranging from relatively mineral-free sources to relatively mineral-rich sources. Mineral-rich freshwater sources may be naturally occurring and include any of a number of hard water sources, lakes or inland seas. Some mineral-rich freshwater sources such as soda lakes or inland seas (for example, Lake Van in Turkey) also provide a source of pH-altering agents. Mineral-rich freshwater sources can also be anthropogenic. For example, mineral-poor (soft) water can be contacted with a source of divalent cations such as alkaline earth metal cations (e.g., Ca2+, Mg2+, etc.) to produce mineral-rich water, which is suitable for the methods and systems described herein . Divalent cations or precursors thereof (eg salts, minerals) may be added to fresh water (or any other type of water described herein) using any suitable procedure (eg, addition of solids, suspensions or solutions). In some embodiments, divalent cations selected from Ca2+ and Mg2+ are added to fresh water. In some embodiments, monovalent cations selected from Na+ and K+ are added to fresh water. In some embodiments, fresh water comprising Ca2+ is combined with materials comprising metal silicates, ash (e.g., fly ash, bottom ash, boiler slag), or products or processed forms thereof, including combinations thereof, which form A solution comprising calcium and magnesium cations.
因而,一些方法包括通过将一种或多种二价阳离子(例如,Ca2+、Mg2+、其组合,等等)添加到水源来制备二价阳离子源。镁阳离子的来源包括但不局限于氢氧化镁、氧化镁、等等。钙阳离子的来源包括但不局限于氢氧化钙、氧化钙、等等。可以使用这样的阳离子的天然存在的或人为的来源。这样的阳离子的天然存在的来源包括但不局限于镁铁质矿物(例如,橄榄石、蛇纹石、橄榄石、滑石,等等)诸如此类。在产生本发明的组合物前将附加的镁阳离子添加到水源(例如,海水)增加了产量(例如,沉淀材料的产量)以及影响了这样的组合物(例如,沉淀材料)的组成,这提供了通过利用矿物如但不限于橄榄石、蛇纹石和Mg(OH)2(水镁石)增加CO2埋存的方式。特定的阳离子(例如,Ca2+、Mg2+、其组合,等等)来源可以是天然存在的或人为的,并且相对于矿物来说可以是纯的或者不纯的(例如,由所关心的矿物及其它矿物和组分组成的组合物)。Thus, some methods include preparing a source of divalent cations by adding one or more divalent cations (eg, Ca2+, Mg2+, combinations thereof, etc.) to a water source. Sources of magnesium cations include, but are not limited to, magnesium hydroxide, magnesium oxide, and the like. Sources of calcium cations include, but are not limited to, calcium hydroxide, calcium oxide, and the like. Naturally occurring or man-made sources of such cations may be used. Naturally occurring sources of such cations include, but are not limited to, mafic minerals (eg, olivine, serpentine, olivine, talc, etc.) and the like. Adding additional magnesium cations to the water source (e.g., seawater) prior to producing the compositions of the present invention increases production (e.g., production of precipitation material) as well as affects the composition of such compositions (e.g., precipitation material), which provides A way to increase CO2 sequestration by utilizing minerals such as but not limited to olivine, serpentine and Mg(OH)2 (brucite). The source of a particular cation (e.g., Ca2+, Mg2+, combinations thereof, etc.) can be naturally occurring or anthropogenic, and can be pure or impure relative to the mineral (e.g., derived from the mineral of interest and other Compositions of minerals and components).
本发明的方法包括以足以在水中产生3或更高、例如、4或更高、如5或更高、例如6或更高、包括7或更高的镁与钙比值的方式,将镁阳离子源添加到初始的水。在某些实施方案中,水的期望的镁与钙阳离子比值为3-10,如4-8。任何适宜的镁阳离子源可以被添加到水中,而提供期望的镁与钙阳离子比值,其中所关心的特定的镁阳离子源包括但不局限于Mg(OH)2,蛇纹石,橄榄石,镁铁质矿物和超镁铁质矿物。添加到水的镁阳离子源的量可以变化,例如,这取决于具体的镁阳离子源和由其产生CO2-加料的水的初始的水。在某些实施方案中,添加到水中的镁阳离子的量为0.01至100.0克/升,如1至100克/升的水,包括5至100克/升的水,例如5至80克/升的水,包括5至50克/升的水。在某些实施方案中,添加到水中的镁阳离子的量足以产生硬度读数为0.06克/升或更多、如0.08克/升或更多、包括0.1克/升或更多的水,如由Metrohm滴定仪(Metrohm AG,瑞士)根据制造商的指导确定的。使用任何适宜的流程,例如搅拌、混合等,镁阳离子源可以结合以水。The method of the present invention comprises introducing magnesium cations in a manner sufficient to produce a magnesium to calcium ratio in water of 3 or higher, for example, 4 or higher, such as 5 or higher, for example 6 or higher, including 7 or higher source added to the initial water. In certain embodiments, the water has a desired magnesium to calcium cation ratio of 3-10, such as 4-8. Any suitable source of magnesium cations may be added to the water to provide the desired ratio of magnesium to calcium cations, where the particular source of magnesium cations of interest includes, but is not limited to, Mg(OH)2, serpentine, olivine, magnesium Iron and ultramafic minerals. The amount of source of magnesium cations added to the water can vary, eg, depending on the particular source of magnesium cations and the initial water from which the CO 2 -feed water is generated. In certain embodiments, the amount of magnesium cation added to the water is 0.01 to 100.0 g/L, such as 1 to 100 g/L of water, including 5 to 100 g/L of water, such as 5 to 80 g/L of water, including 5 to 50 g/l of water. In certain embodiments, the amount of magnesium cations added to the water is sufficient to produce water with a hardness reading of 0.06 g/L or more, such as 0.08 g/L or more, including 0.1 g/L or more, as determined by A Metrohm titrator (Metrohm AG, Switzerland) was determined according to the manufacturer's instructions. The source of magnesium cations can be combined with water using any suitable procedure, such as stirring, mixing, etc.
在其中镁、钙或镁和钙的组合的来源被添加到水的实施方案中,所述来源可以是固体形式的,例如大的、硬的并且常常结晶的颗粒或难于进入溶液的颗粒的结块的形式的。例如,水镁石形式的Mg(OH)2可以处于这样的形式,如许多可用于本发明的实施方案的矿物,如蛇纹石、橄榄石和其它硅酸镁矿物,以及水泥废物等。可以使用任何合适的方法将来自这样的来源的二价阳离子如镁阳离子引入适用于与碳酸盐反应而形成二价阳离子的碳酸盐的形式的水溶液中。通过降低粒径来增加表面积是一种这样的方法,其可以通过本领域众所周知的方法如球磨和喷射研磨来实现。喷射研磨具有破坏物质的许多晶体结构的进一步优点,这提高了溶解度。还考虑的是声化学,其中可以使用强烈的声处理以提高反应速率达期望的量,例如106倍或更高。颗粒,有或者没有减缩尺寸的,可以暴露于促进水溶液的条件,如暴露于酸如HCl、H2SO4等等;在一些实施方案中也可以使用弱酸或碱。参见例如,美国专利申请出版号2005/0022847;2004/0213705;2005/0018910;2008/0031801;和2007/0217981;欧洲专利申请EP1379469和EP1554031;和国际专利申请出版号WO 07/016271和WO 08/061305,其每个全盘引入本文作为参考。In embodiments where a source of magnesium, calcium, or a combination of magnesium and calcium is added to the water, the source may be in solid form, such as large, hard and often crystallized particles or a collection of particles that are difficult to get into solution. in block form. For example, Mg(OH) in the form of brucite may be in the form of many minerals useful in embodiments of the present invention, such as serpentine, olivine, and other magnesium silicate minerals, as well as cement waste, among others. Divalent cations from such sources, such as magnesium cations, may be introduced into an aqueous solution in the form of carbonate suitable for reaction with the carbonate to form the divalent cation using any suitable method. Increasing surface area by reducing particle size is one such method, which can be achieved by methods well known in the art such as ball milling and jet milling. Jet milling has the further advantage of destroying much of the crystalline structure of the substance, which increases solubility. Also contemplated is sonochemistry, where intense sonication can be used to increase reaction rates by a desired amount, eg 106 times or more. Particles, with or without size reduction, may be exposed to aqueous-promoting conditions, such as exposure to acids such as HCl, H2SO4 , etc.; mild acids or bases may also be used in some embodiments. See, eg, U.S. Patent Application Publication Nos. 2005/0022847; 2004/0213705; 2005/0018910; 2008/0031801; and 2007/0217981; European Patent Application Publication Nos. EP1379469 and EP1554031; 061305, each of which is incorporated herein by reference in its entirety.
在一些实施方案中,本发明的方法和系统使用蛇纹石作为矿物源。蛇纹石是天然存在的丰足的矿物并且可能通常由通式X2-3Si2O5(OH)4来描述,其中X选自以下:Mg、Ca、Fe2+、Fe3+、Ni、Al、Zn、和Mn,蛇纹石材料是主要由氢氧化镁和二氧化硅构成的非均质混合物。在本发明的一些实施方案中,蛇纹石不仅用作镁源,而且用作氢氧化物源。因此,在本发明的一些实施方案中,提供氢氧化物以便从水中除去质子和/或通过溶解蛇纹石来调节pH值;在这些实施方案中,酸溶解对于加速溶解来说不是理想的,并且使用其它方式,如喷射研磨和/或声处理。应当理解的是,在间歇或连续方法中,溶解蛇纹石或其它矿物的持续时间不是关键的,因为一旦所述方法以期望的规模开始,并且足够的时间已经过去达合适水平的溶解,已溶材料的连续物流可以无限地保持。因此,即使溶解到期望的水平花费数天、数周、数月或甚至数年,一旦所述方法已经达到已经发生期望的溶解时的第一时间点,其可以无限地保持。在已经发生期望的溶解时的时间点前,可以使用其它方法以便将一些或全部的镁和/或氢氧化物提供给所述方法。蛇纹石也是铁源,其是用于例如水泥(其中常常期望铁组分)的沉淀物的有用组分。In some embodiments, the methods and systems of the present invention use serpentine as the mineral source. Serpentine is a naturally occurring abundant mineral and may generally be described by the general formula X2-3Si2O5(OH)4, where X is selected from the group consisting of: Mg, Ca, Fe2 + , Fe3 + , Ni, Al, Zn, and Mn, the serpentine material is a heterogeneous mixture mainly composed of magnesium hydroxide and silica. In some embodiments of the present invention, serpentine is used not only as a magnesium source, but also as a hydroxide source. Thus, in some embodiments of the invention, hydroxide is provided to remove protons from water and/or to adjust pH by dissolving serpentine; in these embodiments acid dissolution is not ideal for accelerated dissolution, And using other means, such as jet milling and/or sonication. It should be understood that in a batch or continuous process, the duration of dissolution of the serpentine or other mineral is not critical, since once the process has started on the desired scale and sufficient time has elapsed to achieve a suitable level of dissolution, the A continuous flow of molten material can be maintained indefinitely. Thus, even if dissolution to the desired level takes days, weeks, months or even years, once the process has reached the first point in time when the desired dissolution has occurred, it can be maintained indefinitely. Other methods may be used to provide some or all of the magnesium and/or hydroxide to the method prior to the point in time when the desired dissolution has occurred. Serpentine is also a source of iron, which is a useful component for precipitates such as cement, where an iron component is often desired.
对本发明有用的硅酸盐型矿物的其它实例包括但不局限于橄榄石,天然的硅酸镁-铁((Mg,Fe)2SiO4),其可能通常由通式X2(SiO4)n来描述,其中X选自Mg、Ca、Fe2+、Fe3+、Ni、Al、Zn和Mn,和n=2或3;和硅酸钙,如钙硅石。矿物可以单独地或者彼此结合地使用,如美国专利申请(公开号)2009/0301352(公开于2009年12月10日)中所述,其全盘引入本文作为参考。另外,材料可以天然存在或者可以被制造。工业副产物的实例包括但不限于废水泥,富含钙的飞灰,和水泥窑灰尘(CKD),如美国专利申请(公开号)2010/0000444(公开于2010年1月7日)中所述,其全盘引入本文作为参考。Other examples of silicate-type minerals useful for the present invention include, but are not limited to, olivine, the natural magnesium-iron silicate ((Mg, Fe) 2SiO4 ), which may generally be represented by the general formula X2( SiO4 )n Described, wherein X is selected from Mg, Ca, Fe2+, Fe3+, Ni, Al, Zn and Mn, and n=2 or 3; and calcium silicate, such as wollastonite. Minerals may be used alone or in combination with each other as described in US Patent Application (Publication No.) 2009/0301352 (published December 10, 2009), which is incorporated herein by reference in its entirety. In addition, materials can occur naturally or can be manufactured. Examples of industrial by-products include, but are not limited to, waste cement, calcium-rich fly ash, and cement kiln dust (CKD), as disclosed in U.S. Patent Application (Publication No.) 2010/0000444 (published January 7, 2010). , which is incorporated herein by reference in its entirety.
在一些实施方案中,二价阳离子的水溶液可以由也正提供废气物流(例如,燃烧气体物流)的工业设备获得。例如,在水冷的工业设备,如海水-冷却的工业设备中,已经被工业设备用于冷却的水然后可以用作产生沉淀材料所用的水。如果期望的话,在进入本发明的沉淀系统前,水可以被冷却。这样的方法可以例如与直通式冷却系统一起使用。例如,城市或农业用水供给可以用作工业设备用的直通式冷却系统。来自工业设备的水然后可以用于产生沉淀材料,其中输出水具有降低的硬度和更大的纯度。In some embodiments, the aqueous solution of divalent cations can be obtained from an industrial facility that is also providing a waste gas stream (eg, a combustion gas stream). For example, in water-cooled industrial plants, such as seawater-cooled industrial plants, the water that has been used by the industrial plant for cooling can then be used as water for generating precipitation material. If desired, the water can be cooled before entering the precipitation system of the present invention. Such an approach may be used, for example, with a flow-through cooling system. For example, a municipal or agricultural water supply can be used as a flow-through cooling system for industrial equipment. The water from the industrial facility can then be used to produce precipitation material with the output water having reduced hardness and greater purity.
二价阳离子的水溶液可以进一步提供质子-除去剂,其可以表示为碱度或者含二阶阳离子的溶液将酸中和至碳酸盐或碳酸氢盐的等效点的能力。碱度(AT)可以由以下公式表示:Aqueous solutions of divalent cations may further provide proton-removing agents, which may be expressed as alkalinity or the ability of a solution containing divalent cations to neutralize acids to the equivalent point of carbonate or bicarbonate. Alkalinity (AT) can be expressed by the following formula:
AT=[HCO3 -]T+2[CO3 2-]T+[B(OH)4 -]T+[OH-]T+2[PO4 3-]T+[HPO4 2-]T+[SiO(OH)3 -]T-[H+]sws-[HSO4 -],A T =[HCO 3 - ] T +2[CO 3 2- ] T +[B(OH) 4 - ] T +[OH - ] T +2[PO 4 3- ] T +[HPO 4 2- ] T + [SiO(OH) 3 - ] T - [H + ] sws - [HSO 4 - ],
其中,“T”表示如测量的在溶液中的物质的总浓度。其它物质,取决于来源,也可以有助于碱度。在溶液中物质的总浓度与游离浓度不同,后者考虑了例如在海水中存在的显著量的离子对相互作用。根据该公式,水性二价阳离子源可以具有各种浓度的碳酸氢盐,碳酸盐,硼酸盐,氢氧化物,磷酸盐,磷酸氢盐和/或硅酸盐,后者可有助于水性二价阳离子源的碱度。任何类型的碱度适于本发明。例如,在一些实施方案中,高硼酸盐碱度的二价阳离子源适合于本发明。在这样的实施方案中,二价阳离子的来源可以包含以硼酸盐形式存在的硼,包括但不限于,尤其,BO3 3-、B2O5 4-、B3O7 5-和B4O9 6-。在这样的实施方案中,硼酸盐浓度可以超过溶液中任何其它物质的浓度,包括例如碳酸盐和/或碳酸氢盐。在一些实施方案中,二价阳离子的来源具有至少10,100,500,1000,1500,3000,5000或大于5000毫当量的碱度。例如,在一些实施方案中,二价阳离子的来源具有500至1000毫当量的碱度。where "T" represents the total concentration of the species in solution as measured. Other substances, depending on the source, can also contribute to alkalinity. The total concentration of species in solution differs from the free concentration, which takes into account the significant amount of ion-pair interactions that exist, for example, in seawater. According to this formula, the aqueous divalent cation source can have various concentrations of bicarbonate, carbonate, borate, hydroxide, phosphate, hydrogen phosphate and/or silicate, which can contribute to Alkalinity of an aqueous divalent cation source. Any type of alkalinity is suitable for the present invention. For example, in some embodiments, a source of divalent cations of high borate alkalinity is suitable for the present invention. In such embodiments, the source of divalent cations may comprise boron in the form of borates including, but not limited to, BO 3 3- , B 2 O 5 4- , B 3 O 7 5- , and B 4 O 9 6- . In such embodiments, the borate concentration may exceed the concentration of any other species in the solution, including, for example, carbonate and/or bicarbonate. In some embodiments, the source of divalent cations has an alkalinity of at least 10, 100, 500, 1000, 1500, 3000, 5000, or greater than 5000 milliequivalents. For example, in some embodiments, the source of divalent cations has a basicity of 500 to 1000 milliequivalents.
在本发明的一些方法中,在使水经受沉淀条件前,水(如盐水或富含矿物的水)没有接触CO2源。在这些方法中,水将具有一定量的与其有关的CO2,例如以碳酸氢盐离子的形式,后者已经从在实施所述方法前水所暴露到的环境中获得。使水经历本发明的沉淀条件导致这种CO2转化为存储稳定的沉淀物,并且因此埋存了CO2。当经受本发明的方法的水再次暴露于其自然环境如大气时,来自大气的更多的CO2将被水吸收,这导致来自大气的CO2的净除去并且将相应数量的CO2引入到存储稳定的产物中,其中富含矿物的淡水源可接触CO2源,例如,如下文更详细地描述的。这些方法的实施方案可以被看作埋存直接来自地球大气的CO2气体的方法。方法的实施方案有效地除去来自地球大气的CO2。例如,方法的实施方案被配置以按每加仑的盐水计以0.025M或更多、如0.05M或更多、包括0.1M或更多的速率从盐水中除去CO2。In some methods of the invention, water (such as brine or mineral-rich water) is not exposed to a source of CO2 prior to subjecting the water to precipitation conditions. In these methods, the water will have associated with it an amount of CO2 , for example in the form of bicarbonate ions, which have been obtained from the environment to which the water was exposed before carrying out the method. Subjecting the water to the precipitation conditions of the present invention results in the conversion of this CO 2 into a storage stable precipitate and thus sequesters the CO 2 . When the water subjected to the method of the present invention is again exposed to its natural environment such as the atmosphere, more CO from the atmosphere will be absorbed by the water, which results in a net removal of CO from the atmosphere and a corresponding amount of CO introduced into the In storage-stable products, a source of mineral-rich freshwater may be exposed to a source of CO 2 , for example, as described in more detail below. Embodiments of these methods can be viewed as methods for sequestering CO2 gas directly from the Earth's atmosphere. Embodiments of the method effectively remove CO2 from the Earth's atmosphere. For example, embodiments of the method are configured to remove CO2 from brine at a rate of 0.025M or more, such as 0.05M or more, including 0.1M or more per gallon of brine.
在一些实施方案中,本发明提供使一定体积的吸收溶液(例如水溶液)与二氧化碳源接触以产生包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的组合物,其中组合物是溶液或浆液。在一些实施方案中,所述溶液是包括含碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的沉淀材料的浆液。在一些实施方案中,沉淀材料是通过使在与二氧化碳源接触前、中或后使所述体积的水溶液经受沉淀条件而产生的。在水溶液中可能有足够的二氧化碳以产生显著量的碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐(例如,从盐水或海水);然而通常使用另外的二氧化碳。CO2的来源可以是任何适宜的CO2源。CO2的来源可以是气体、液体、固体(例如,干冰)、超临界流体或溶于液体的CO2。在一些实施方案中,CO2源是气体CO2源如废气物流。气体CO2源可以是基本上纯的CO2,或者,如下文更详细地描述的,包括一种或多种除CO2之外的组分,其中一种或多种组分包括一种或多种另外的气体如SOx(例如,SO,SO2,SO3),NOx(例如,NO,NO2),等等,非气体组分,或其组合。废物物流可以进一步包括VOC(挥发性有机化合物),金属(例如,汞,砷,镉,硒),和颗粒物质,包括固体的颗粒(例如,飞灰)或悬浮在气体中的液体。在一些实施方案中,气体CO2源可以是由工业设备的主动过程产生的废气物流(例如,排气)。工业设备的性质可以不同,工业设备包括但不限于发电厂,化学加工厂,机械加工厂,炼油厂,水泥厂,炼钢厂,和其它产生CO2作为燃料燃烧或另一加工步骤(例如,水泥厂的煅烧)的副产物的工业设备。在一些实施方案中,例如,气体CO2源可以是来自燃煤的发电厂的烟道气。In some embodiments, the invention provides for contacting a volume of an absorption solution (e.g., an aqueous solution) with a source of carbon dioxide to produce a composition comprising carbonate, bicarbonate, or carbonate and bicarbonate, wherein the composition is a solution or slurry. In some embodiments, the solution is a slurry comprising precipitation material comprising carbonates, bicarbonates, or carbonates and bicarbonates. In some embodiments, the precipitating material is produced by subjecting the volume of aqueous solution to precipitating conditions before, during, or after contacting with the carbon dioxide source. There may be sufficient carbon dioxide in an aqueous solution to generate significant amounts of carbonate, bicarbonate, or both (eg, from brine or seawater); however, additional carbon dioxide is typically used. The source of CO2 can be any suitable source of CO2 . The source of CO2 can be gas, liquid, solid (eg, dry ice), supercritical fluid, or CO2 dissolved in a liquid. In some embodiments, the CO2 source is a gaseous CO2 source such as a waste gas stream. The source of gaseous CO2 can be substantially pure CO2 , or, as described in more detail below, include one or more components other than CO2 , wherein one or more components include one or Various additional gases such as SOx (eg, SO, SO 2 , SO 3 ), NOx (eg, NO, NO 2 ), etc., non-gas components, or combinations thereof. The waste stream may further include VOCs (volatile organic compounds), metals (eg, mercury, arsenic, cadmium, selenium), and particulate matter, including particles of solids (eg, fly ash) or liquids suspended in a gas. In some embodiments, the source of gaseous CO2 can be an exhaust stream (eg, exhaust) produced by an active process of an industrial facility. The nature of industrial facilities can vary, and industrial facilities include, but are not limited to, power plants, chemical processing plants, mechanical processing plants, oil refineries, cement plants, steel plants, and others that generate CO2 for combustion as fuel or another process step (e.g., Industrial equipment for by-products of calcination in cement plants. In some embodiments, for example, the source of gaseous CO2 can be flue gas from a coal-fired power plant.
包括CO2的废气物流包括还原状态物流(例如,合成气,变换合成气,天然气,氢气等)和氧化状态物流(例如,由燃烧产生的烟道气)。可适用于本发明的特定的废气物流包括由燃烧产生的含氧的烟道气(例如,来自煤或另一碳型燃料,有很少的或者没有烟道气的预处理),涡轮增压锅炉产物气体,煤炭气化产物气体,预燃烧合成气体(例如,如在发电厂中在煤炭气化期间形成的那种),变换的煤炭气化产物气体,厌氧消化池产物气体,井口天然气物流,重整天然气或甲烷水合物等。来自任何适宜源的燃烧气体可以用于本发明的方法和系统中。在一些实施方案中,使用来自工业设备如发电厂、水泥厂和煤炭加工厂的燃烧后排出物烟囱的燃烧气体。Exhaust gas streams that include CO include streams in a reduced state (eg, syngas, shifted syngas, natural gas, hydrogen, etc.) and streams in an oxidized state (eg, flue gas resulting from combustion). Specific exhaust gas streams that may be suitable for use in the present invention include oxygenated flue gas produced by combustion (e.g., from coal or another carbon-based fuel with little or no flue gas pretreatment), turbocharged Boiler product gas, coal gasification product gas, pre-combustion synthesis gas (eg, as formed during coal gasification in power plants), shifted coal gasification product gas, anaerobic digester product gas, wellhead natural gas Logistics, reforming natural gas or methane hydrate, etc. Combustion gases from any suitable source may be used in the methods and systems of the present invention. In some embodiments, combustion gases from post-combustion effluent stacks of industrial facilities such as power plants, cement plants, and coal processing plants are used.
因此,废气物流可以由各种不同类型的工业设备产生。本发明的合适的废气物流包括由燃烧化石燃料(例如,煤炭,石油,天然气,丙烷,柴油)、生物质和/或天然存在的有机燃料储藏物(例如,焦油砂矿,重油,油页岩,等等)的人为的燃料产物的工业设备产生的废气物流。在一些实施方案中,适用于本发明的系统和方法的废气物流来源于燃煤的发电厂,如粉煤发电厂、超临界煤炭发电厂、大量燃煤发电厂、流化床煤炭发电厂。在一些实施方案中,废气物流来源于燃气或油的锅炉和蒸汽轮机发电厂,燃气或油的锅炉简单循环燃气轮机发电厂,或燃气或油的锅炉联合循环燃气轮机发电厂。在一些实施方案中,使用由燃烧合成气(即,通过有机物,例如,煤炭、生物质等的气化产生的气体)的发电厂产生的废气物流。在一些实施方案中,使用来自集成气化联合循环(IGCC)装置的废气物流。在一些实施方案中,根据本发明的系统和方法使用由余热回收蒸汽发生器(HRSG)装置产生的废气物流。Thus, waste gas streams can be produced by various different types of industrial equipment. Suitable waste gas streams of the present invention include those produced by burning fossil fuels (e.g., coal, oil, natural gas, propane, diesel), biomass, and/or naturally occurring organic fuel reserves (e.g., tar sands, heavy oil, oil shale). , etc.) anthropogenic fuel products of industrial plant waste streams. In some embodiments, exhaust gas streams suitable for use in the systems and methods of the present invention originate from coal fired power plants, such as pulverized coal power plants, supercritical coal power plants, bulk coal fired power plants, fluidized bed coal power plants. In some embodiments, the exhaust gas stream originates from a gas or oil boiler and steam turbine power plant, a gas or oil boiler simple cycle gas turbine power plant, or a gas or oil boiler combined cycle gas turbine power plant. In some embodiments, an exhaust gas stream produced by a power plant burning syngas (ie, gas produced by the gasification of organic matter, eg, coal, biomass, etc.) is used. In some embodiments, a waste gas stream from an integrated gasification combined cycle (IGCC) unit is used. In some embodiments, systems and methods in accordance with the present invention utilize an exhaust gas stream produced by a heat recovery steam generator (HRSG) unit.
包括CO2的废气物流也可由其它工业加工产生。由水泥厂产生的废气物流也适用于本发明的系统和方法。水泥厂废气物流包括来自湿法和干法装置的废气物流,所述装置可以使用竖窑或转窑,并且可以包括预煅烧炉。这些工业设备可以各自燃烧单种燃料,或者可以顺序地或同时地燃烧两种或更多种燃料。其它工业设备如熔炉和炼油厂也是包括二氧化碳的废气物流的有用的来源。Waste gas streams including CO2 may also result from other industrial processes. Waste gas streams produced by cement plants are also suitable for use in the systems and methods of the present invention. Cement plant off-gas streams include off-gas streams from wet and dry plants, which may use shaft kilns or rotary kilns, and may include precalciners. These industrial devices may each burn a single fuel, or may burn two or more fuels sequentially or simultaneously. Other industrial facilities such as furnaces and refineries are also useful sources of waste gas streams that include carbon dioxide.
以输送气态废物物流的任何适宜方式,气态废物物流可以由工业设备提供给本发明的CO2-处理系统。在一些实施方案中,废气物流由气体输送装置(例如,管道、管子等)提供,后者从工业设备的烟道或同功结构(例如,工业设备的烟道和烟囱)到CO2-处理系统的一个或多个位置。在这样的实施方案中,管线(例如,管道,管子,等等)可以连接到工业设备的烟道,使得通过烟道离开的气体被输送到CO2-处理系统的合适的位置(例如,处理器或其组件,如气-液接触器或气-液-固接触器)。取决于CO2-处理系统的特定的配置,工业设备上的气体输送装置的位置可以变换,例如,以便提供期望的温度的废气物流。因而,在一些实施方案中,其中期望温度为0℃至2000℃,如0℃至1800℃,包括60℃至700℃,例如,100℃至400℃的气态废物物流,在锅炉、燃气轮机、窑炉的出口点,或在提供期望的温度的发电厂的任一点可以获得烟道气。气体输送装置可以被配置以保持烟道气在露点(例如,125℃)以上的温度,以免冷凝和相关的复杂因素。可以采取其它步骤以降低冷凝及其它有害作用的不利影响,如使用不锈钢或氟烃(如聚(四氟乙烯))管线化的管道,使得管道不迅速地损坏。The gaseous waste stream may be provided to the CO2 -treatment system of the present invention by an industrial facility in any suitable manner for conveying the gaseous waste stream. In some embodiments, the waste gas stream is provided by a gas transport device (e.g., pipes, pipes, etc.) from an industrial plant flue or a similar structure (e.g., industrial plant flue and chimney) to the CO2 -processing One or more locations in the system. In such an embodiment, a line (e.g., pipe, pipe, etc.) may be connected to the flue of the industrial facility so that the gas exiting through the device or its components, such as gas-liquid contactor or gas-liquid-solid contactor). Depending on the particular configuration of the CO2 -processing system, the location of the gas delivery means on the industrial plant may be varied, for example, in order to provide a desired temperature of the off-gas stream. Thus, in some embodiments, where the desired temperature is in the range of 0°C to 2000°C, such as 0°C to 1800°C, including 60°C to 700°C, e.g. Flue gas can be obtained at the exit point of the furnace, or at any point in the power plant that provides the desired temperature. The gas delivery device may be configured to maintain the temperature of the flue gas above the dew point (eg, 125°C) to avoid condensation and related complications. Other steps can be taken to reduce the adverse effects of condensation and other deleterious effects, such as using stainless steel or fluorocarbon (eg poly(tetrafluoroethylene)) lined tubing so that the tubing does not quickly fail.
二氧化碳可以是废气物流中的主要的非空气源组分。在一些实施方案中,废气物流可以包括数量为200ppm至1,000,000ppm、如1000ppm至200,000ppm、包括2000ppm至200,000ppm、例如、2000ppm至180,000ppm或2000ppm至130,000ppm的二氧化碳。在一些实施方案中,废气物流可以包括数量为350ppm至400,000ppm的二氧化碳。这样数量的二氧化碳可以被认为是时间平均的数量。例如,在一些实施方案中,废气物流可以包括数量为40,000ppm(4%)至100,000ppm(10%)的二氧化碳,这取决于废气物流(例如,来自燃烧天然气的发电厂、熔炉、小型锅炉等的CO2)。例如,在一些实施方案中,废气物流可以包括数量为100,000ppm(10%)至150,000ppm(15%)的二氧化碳,这取决于废气物流(例如,来自燃煤的发电厂、燃油发电机、柴油发电机等的CO2)。例如,在一些实施方案中,废气物流可以包括数量为200,000ppm(20%)至400,000ppm(40%)的二氧化碳,这取决于废气物流(例如,来自水泥厂煅烧、化工厂等的CO2)。例如,在一些实施方案中,废气物流可以包括数量为900,000ppm(90%)至1,000,000ppm(100%)的二氧化碳,这取决于废气物流(例如,来自乙醇发酵罐的CO2,在炼油厂、氨装置、代用天燃气(SNG)装置处来自蒸汽重整的CO2、与酸性气体分离的CO2等)。废气物流中的CO2的浓度可以减少达10%或更多、20%或更多、30%或更多、40%或更多、50%或更多、60%或更多、70%或更多、80%或更多、90%或更多、95%或更多、99%或更多、99.9%或更多、或99.99%。换言之,至少10%、20%、30%、40%、50%、60%、70%、80%、90%、95%、99%、99.9%或99.99%的二氧化碳可以从废气物流中除去。Carbon dioxide can be the major non-air source component in the exhaust stream. In some embodiments, the exhaust gas stream may include carbon dioxide in an amount from 200 ppm to 1,000,000 ppm, such as from 1000 ppm to 200,000 ppm, including from 2000 ppm to 200,000 ppm, for example, from 2000 ppm to 180,000 ppm, or from 2000 ppm to 130,000 ppm. In some embodiments, the exhaust stream may include carbon dioxide in an amount from 350 ppm to 400,000 ppm. Such quantities of carbon dioxide can be considered as time-averaged quantities. For example, in some embodiments, the exhaust stream may include carbon dioxide in an amount of 40,000 ppm (4%) to 100,000 ppm (10%), depending on the exhaust stream (e.g., from a natural gas fired power plant, furnace, small boiler, etc. of CO 2 ). For example, in some embodiments, the exhaust stream may include carbon dioxide in amounts ranging from 100,000 ppm (10%) to 150,000 ppm (15%), depending on the exhaust stream (e.g., from a coal-fired power plant, oil-fired generator, diesel CO 2 for generators, etc.). For example, in some embodiments, the waste gas stream may include carbon dioxide in an amount from 200,000 ppm (20%) to 400,000 ppm (40%) depending on the waste gas stream (e.g., CO2 from cement plant calcination, chemical plant, etc.) . For example, in some embodiments, the waste gas stream may include carbon dioxide in amounts ranging from 900,000 ppm (90%) to 1,000,000 ppm (100%) depending on the waste gas stream (e.g., CO2 from ethanol fermenters, in refineries, Ammonia plants, CO2 from steam reforming at substitute natural gas (SNG) plants, CO2 from acid gas separation, etc.). The concentration of CO in the exhaust stream can be reduced by 10% or more, 20% or more, 30% or more, 40% or more, 50% or more, 60% or more, 70% or More, 80% or more, 90% or more, 95% or more, 99% or more, 99.9% or more, or 99.99%. In other words, at least 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 95%, 99%, 99.9%, or 99.99% of the carbon dioxide can be removed from the waste gas stream.
可以使用来自工业设备的一部分废气物流(即,非全部气态废物物流)来产生包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的组合物。在这种实施方案中,用于产生组合物的该部分的废气物流可以是75%或更少,如60%或更少,和包括50%和更少的废气物流。在其它实施方案中,由工业设备产生的整个废气物流的大多数(例如,80%或更多)用于产生组合物。在这种实施方案中,80%或更多、如90%或更多、包括95%或更多、直至100%的由所述来源产生的废气物流(例如,烟道气)可以用于产生本发明的组合物。A portion of the waste gas stream (ie, not the entire gaseous waste stream) from an industrial facility can be used to produce a composition comprising carbonate, bicarbonate, or carbonate and bicarbonate. In such embodiments, the waste gas stream used to generate the portion of the composition may be 75% or less, such as 60% or less, and includes 50% and less of the waste gas stream. In other embodiments, a majority (eg, 80% or more) of the entire waste gas stream produced by the industrial facility is used to produce the composition. In such embodiments, 80% or more, such as 90% or more, including 95% or more, up to 100% of the waste gas stream (e.g., flue gas) produced by the source may be used to generate Compositions of the invention.
在本发明的一些实施方案中,基本上100%的烟道气或者一部分烟道气(来自发电厂)中所含的CO2可以被埋存作为本发明的组合物(例如,包括一种或多种稳定的或亚稳定的无机物的沉淀材料)。这样的埋存可以在单个步骤中或者在多个步骤中进行,并且可以进一步包括其它用于埋存CO2的方法(例如,当烟道气中CO2的浓度降低时,用于除去气体中全部最初的CO2的能量消耗过高的更加能量集约的方法在除去气体中的最终CO2中可能变得可行)。因此,在一些实施方案中,进入发电厂的气体(常规的大气空气)可以包含一定浓度的CO2,其大于离开发电厂的烟道气中的CO2的浓度,所述烟道气已经由本发明的方法和系统处理。因此,在一些实施方案中,本发明的方法和系统包括这样的方法,其包括:提供气体(例如,大气空气)给发电厂,其中气体包括CO2;在发电厂中处理该气体(例如,通过燃烧化石燃料来消耗O2)以产生CO2,然后处理排气气体而除去CO2;和从发电厂释放气体,其中从发电厂释放的气体具有比提供给发电厂的气体更低的CO2含量。在一些实施方案中,从发电厂释放的气体包含比进入发电厂的气体少至少10%的CO2,或少至少20%的CO2,或少至少30%的CO2,或少至少40%的CO2,或少至少50%的CO2,或少至少60%的CO2,或少至少70%的CO2,或少至少80%的CO2,或少至少90%的CO2,或少至少95%的CO2,或少至少99%的CO2,或少至少99.5%的CO2,或少至少99.9%的CO2。在一些实施方案中,进入发电厂的气体是大气空气,离开发电厂的气体是经处理的烟道气。In some embodiments of the invention, substantially 100% of the CO contained in the flue gas or a portion of the flue gas (from a power plant) may be sequestered as a composition of the invention (e.g., comprising one or Precipitation material of various stable or metastable inorganic substances). Such sequestration can be performed in a single step or in multiple steps, and can further include other methods for sequestering CO2 (e.g., for removal of More energy-intensive methods may become feasible in removing final CO2 from the gas where the energy consumption of all initial CO2 is too high). Thus, in some embodiments, the gas entering the power plant (regular atmospheric air) may contain a concentration of CO2 that is greater than the concentration of CO2 in the flue gas leaving the power plant that has been produced by the present Inventive method and system process. Accordingly, in some embodiments, the methods and systems of the present invention include methods comprising: providing a gas (e.g., atmospheric air) to a power plant, wherein the gas includes CO 2 ; processing the gas in the power plant (e.g., Consuming O2 by burning fossil fuels to produce CO2 , then treating exhaust gas to remove CO2 ; and releasing gas from a power plant, where the gas released from the power plant has lower CO than the gas supplied to the power plant 2 content. In some embodiments, the gas released from the power plant contains at least 10% less CO2 , or at least 20% less CO2 , or at least 30% less CO2 , or at least 40% less CO2 than the gas entering the power plant CO 2 , or at least 50% less CO 2 , or at least 60% less CO 2 , or at least 70% less CO 2 , or at least 80% less CO 2 , or at least 90% less CO 2 , or At least 95% less CO2 , or at least 99% less CO2 , or at least 99.5% less CO2 , or at least 99.9% less CO2 . In some embodiments, the gas entering the power plant is atmospheric air and the gas leaving the power plant is treated flue gas.
尽管,来自工业设备的废气物流提供了相对浓缩的由化石燃料燃烧产生的CO2源和/或另外的组分,但是本发明的方法和系统也适用于除去来自较少浓缩的来源的燃烧气体组分(例如,大气空气),后者包含比例如烟道气浓度低得多的污染物。因此,在一些实施方案中,方法和系统包括通过产生本发明的组合物降低大气空气中的CO2和/或另外的组分的浓度。如同废气物流一样,一部分大气空气中的CO2的浓度可以降低达10%或更多、20%或更多、30%或更多、40%或更多、50%或更多、60%或更多、70%或更多、80%或更多、90%或更多、95%或更多、99%或更多、99.9%或更多、或99.99%。CO2的这样的降低可以以如本文中所述的收率,或者以更高或更低的收率实现,并且可以在一个处理步骤中或者在一系列处理步骤中实现。Although exhaust streams from industrial facilities provide relatively concentrated sources of CO and/or additional components produced by fossil fuel combustion, the methods and systems of the present invention are also applicable to the removal of combustion gases from less concentrated sources Components (eg, atmospheric air) that contain much lower concentrations of pollutants than, for example, flue gases. Accordingly, in some embodiments, methods and systems involve reducing the concentration of CO2 and/or additional components in atmospheric air by producing compositions of the invention. As with the exhaust stream, the concentration of CO2 in a portion of the atmospheric air can be reduced by 10% or more, 20% or more, 30% or more, 40% or more, 50% or more, 60% or more More, 70% or more, 80% or more, 90% or more, 95% or more, 99% or more, 99.9% or more, or 99.99%. Such reduction of CO2 can be achieved at the yields described herein, or at higher or lower yields, and can be achieved in one process step or in a series of process steps.
在某些实施方案中,氧化条件包括使气体物流经受过氧化氢(H2O2)或H2O2/CH3OH混合物。使用过氧化氢氧化含CO2的气体物流的系统和方法的示范性的说明可以见于美国专利5,670,122中,后者全盘引入本文作为参考。如′122专利所述,气体物流可以用过氧化氢处理达足够的时间以便氧化其中的组分,例如,从而将一氧化氮(NO)、三氧化硫(SO3)、轻质烃类(C1-C4)、一氧化碳(CO)和汞中的一种或多种转化为NO2、SO2、CO2和HgO。气体物流可以用过氧化氢或H2O2/CH3OH混合物处理,然后使气体物流与水接触(例如,含碱土金属离子的水,形式为平喷物流、喷雾或液滴、烟雾或其组合)。在一些实施方案中,一种回收的气体物流,其是在使气体物流与水(例如,含碱土金属的水)接触后回收的,用H2O2/CH3OH混合物处理并且再加工(即,第二次接触到水)。In certain embodiments, the oxidizing conditions include subjecting the gas stream to hydrogen peroxide (H 2 O 2 ) or a H 2 O 2 /CH 3 OH mixture. An exemplary description of a system and method for oxidizing a CO2 - containing gas stream using hydrogen peroxide can be found in US Patent No. 5,670,122, which is incorporated herein by reference in its entirety. As described in the '122 patent, the gas stream can be treated with hydrogen peroxide for a time sufficient to oxidize the components therein, for example, to convert nitrogen monoxide (NO), sulfur trioxide (SO 3 ), light hydrocarbons ( One or more of C1-C4), carbon monoxide (CO) and mercury are converted to NO2 , SO2 , CO2 and HgO. The gas stream can be treated with hydrogen peroxide or a H2O2 / CH3OH mixture , and the gas stream can then be contacted with water (e.g., water containing alkaline earth metal ions, in the form of flat jets, sprays or droplets, fumes or combination). In some embodiments, a recovered gas stream, which is recovered after contacting the gas stream with water (e.g., alkaline earth metal-containing water), is treated with a H 2 O 2 /CH 3 OH mixture and reprocessed ( i.e., second exposure to water).
过氧化氢或H2O2/CH3OH混合物的反应时间可以为0.01-5秒,例如0.1-2秒。NO2、SO2、CO2和HgO(及气体物流的其它组分)于是能够通过吸附到水例如含碱土金属离子的水中来除去。在某些实施方案中,气体加料的水然后经受沉淀条件而形成沉淀物,其包括来自气体物流的一种或多种化学组分(例如,NO2、SO2、CO2、HgO等)。因而,本发明提供了一种快速且有效的除去各式各样的气体源的化学组分的方法(例如,CO2、重要污染物和/或其它有毒的或环境有害的组分),使得这些组分没有以危险高浓度排放到大气中。例如,本发明可用于除去从锅炉、熔炉、焚烧炉、固定式发动机和其它与燃烧各种类型的燃料有关的系统散发的烟道气中的这些化合物。The reaction time of hydrogen peroxide or H 2 O 2 /CH 3 OH mixture may be 0.01-5 seconds, for example 0.1-2 seconds. NO2 , SO2 , CO2 and HgO (and other components of the gas stream) can then be removed by adsorption to water, eg water containing alkaline earth metal ions. In certain embodiments, the gas-fed water is then subjected to precipitation conditions to form a precipitate that includes one or more chemical components from the gas stream (eg, NO2 , SO2 , CO2 , HgO, etc.). Thus, the present invention provides a rapid and efficient method for removing chemical components (e.g., CO2 , important pollutants, and/or other toxic or environmentally harmful components) of a wide variety of gas sources such that These components are not emitted into the atmosphere in dangerously high concentrations. For example, the present invention may be used to remove these compounds from flue gases emitted from boilers, furnaces, incinerators, stationary engines and other systems involved in the combustion of various types of fuels.
与气体物流组合使用的过氧化氢和甲醇的总量,相对于组分总数的摩尔比,通常将是0.5-2.0,在大多数应用中,0.9-1.5。过氧化氢可以被喷射(例如,以水溶液的形式),浓度为1%-50%,例如,10-30%。过氧化氢也可以以H2O2溶液和甲醇的混合物的形式被喷射。使用H2O2和甲醇混合物是令人期望的,因为甲醇的成本非常低。甲醇与H2O2的比例应该尽可能高以便降低添加剂的成本,但是要满足排放要求。The total amount of hydrogen peroxide and methanol used in combination with the gas stream will generally be in a molar ratio of 0.5 to 2.0, in most applications 0.9 to 1.5, relative to the total number of components. Hydrogen peroxide may be sparged (eg, as an aqueous solution) at a concentration of 1%-50%, eg, 10-30%. Hydrogen peroxide can also be sparged as a mixture of H2O2 solution and methanol. Using a mixture of H2O2 and methanol is desirable because the cost of methanol is very low. The ratio of methanol to H2O2 should be as high as possible in order to reduce the cost of additives, but to meet emission requirements.
在本发明中使用过氧化氢具有许多优点。如果恰当存储的话,过氧化氢水溶液很稳定。使用过氧化氢没有产生任何环境问题,因为过氧化氢本身不是污染源,唯一的反应副产物是水和氧气。因此,在本发明中可以安全地使用过氧化氢。The use of hydrogen peroxide in the present invention has many advantages. Aqueous hydrogen peroxide solution is stable if stored properly. The use of hydrogen peroxide does not create any environmental concerns because hydrogen peroxide itself is not a source of pollution and the only by-products of the reaction are water and oxygen. Therefore, hydrogen peroxide can be safely used in the present invention.
与CO2源接触的水的pH值可以不同。在有些情况下,与CO2源接触的水的pH值是酸性的,使得pH是低于7、如6.5或更低、6或更低、5.5或更低、5或更低、4.5或更低或4或更低。在其它实施方案中,水的pH值可以是中性的至略碱性的,这是指水的pH值可以为pH 7至pH9,如pH 7至pH 8.5,包括pH 7.5至pH 8.5。The pH of the water in contact with the CO2 source can vary. In some cases, the pH of the water in contact with the CO2 source is acidic such that the pH is below 7, such as 6.5 or less, 6 or less, 5.5 or less, 5 or less, 4.5 or more Low or 4 or lower. In other embodiments, the pH of the water may be neutral to slightly alkaline, which means the pH of the water may be from
在有些情况下,当与CO2源如含二氧化碳的气体物流接触时,水如含碱土金属离子的水(包括碱性溶液或天然的盐碱水)是碱性的。在这一情况下,虽然是碱性的,但是水的pH值通常不足以引起存储稳定的二氧化碳埋存产物的沉淀。因而,pH可以是9.5或更低,如9.3或更低,包括9或更低。In some cases, water, such as alkaline earth metal ion-containing water (including alkaline solutions or naturally saline waters), is alkaline when contacted with a source of CO2 , such as a carbon dioxide-containing gas stream. In this case, although alkaline, the pH of the water is generally insufficient to cause precipitation of storage-stable carbon dioxide sequestration products. Thus, the pH may be 9.5 or lower, such as 9.3 or lower, including 9 or lower.
在有些情况下,在与含二氧化碳的气体物流接触时,如上所述的pH可以被维持在基本上定值,或者可以操控pH以便最大化CO2吸收同时最小化碱消耗或者除去质子的其它方式,如通过在某一pH开始并且随着CO2持续被引入,逐渐地使pH升高。在其中pH被维持在基本上恒定的实施方案中,其中“基本上恒定”是指在与二氧化碳接触的某些阶段中pH变化幅度是0.75或更少,如0.50或更少,包括0.25或更少,如0.10或更少。pH可以被维持在基本上定值,或者被操控以便最大化CO2吸收,但是防止氢氧化物沉淀,没有沉淀物,使用任何适宜的方法。在有些情况下,在水的CO2加料期间,pH被维持在基本上定值,或者在没有沉淀的情况下被操控来最大化CO2吸收,这是通过以提供基本上恒定的pH的方式添加足够量的碱至水中来实现的。有时候,令人期望的是控制pH以便在没有固体沉淀的情况下以及在没有从水(即吸收溶液)释放CO2的情况下最大化二氧化碳及来自气体物流的其它组分(例如SOx、NOx、重金属、其它酸性气体)的吸收。可以添加任何适宜的碱或碱的组合,包括但不限于氧化物和氢氧化物,如氢氧化镁,其中合适的碱的进一步的实例在下文综述。在其它情况中,pH可以被维持在基本上定值,或者被操控以便最大化CO2吸收,这是通过使用电化学流程,如如下所述的流程实现的,以便水的pH值用电化学方法维持在基本上定值。令人惊讶地,如实例IV中所示,已经发现,通过简单海水喷布,同时添加碱(除去质子),有可能吸收,例如,大于50%的包括20%的CO2的气体中所含的CO2。In some cases, the pH as described above may be maintained at a substantially constant value when contacted with the carbon dioxide-containing gas stream, or the pH may be manipulated to maximize CO2 absorption while minimizing base consumption or other means of removing protons , such as by starting at a certain pH and gradually raising the pH as CO2 continues to be introduced. In embodiments wherein the pH is maintained substantially constant, wherein "substantially constant" means that during certain stages of contact with carbon dioxide the pH varies by 0.75 or less, such as 0.50 or less, including 0.25 or more Less, like 0.10 or less. The pH can be maintained at a substantially constant value, or manipulated so as to maximize CO2 absorption, but prevent precipitation of hydroxides, no precipitate, using any suitable method. In some cases, during CO2 dosing of water, the pH is maintained at a substantially constant value, or is manipulated without precipitation to maximize CO2 absorption, in a manner that provides a substantially constant pH This is accomplished by adding a sufficient amount of alkali to the water. Sometimes it is desirable to control the pH to maximize carbon dioxide and other components from the gas stream (e.g. SOx, NOx , heavy metals, other acid gases) absorption. Any suitable base or combination of bases may be added, including but not limited to oxides and hydroxides such as magnesium hydroxide, with further examples of suitable bases being reviewed below. In other cases, the pH may be maintained at a substantially constant value, or manipulated to maximize CO2 absorption, by using an electrochemical process, such as the one described below, so that the pH of the water is measured electrochemically The method remains basically constant. Surprisingly, as shown in Example IV, it has been found that by simple seawater spraying, with simultaneous addition of alkali (removal of protons), it is possible to absorb, for example , greater than 50% of the of CO 2 .
在一些实施方案中,本发明的方法和系统能够吸收CO2的气体源如CO2工业源,例如来自发电厂的烟道气或来自水泥厂的废气中的5%或更多、10%或更多、15%或更多、20%或更多、25%或更多、30%或更多、35%或更多、40%或更多、45%或更多、50%或更多、55%或更多、60%或更多、65%或更多、70%或更多、75%或更多、80%或更多、85%或更多、90%或更多、95%或更多、或99%或更多的CO2。在一些实施方案中,本发明的方法和系统能够吸收CO2的气体源如CO2工业源例如来自发电厂的烟道气或来自水泥厂的废气中的50%或更多的CO2。In some embodiments, the methods and systems of the present invention are capable of absorbing 5% or more , 10%, or More, 15% or more, 20% or more, 25% or more, 30% or more, 35% or more, 40% or more, 45% or more, 50% or more , 55% or more, 60% or more, 65% or more, 70% or more, 75% or more, 80% or more, 85% or more, 90% or more, 95 % or more, or 99% or more CO2 . In some embodiments, the methods and systems of the present invention are capable of absorbing 50% or more of the CO2 in a gaseous source of CO2 such as an industrial source of CO2 such as flue gas from a power plant or waste gas from a cement plant.
在一些实施方案中,本发明的方法和系统能将大于20吨/小时的二氧化碳吸收到吸收溶液中,按72小时的连续操作的平均值计。在一些实施方案中,本发明的方法和系统能将大于40吨/小时的二氧化碳吸收到吸收溶液中。在一些实施方案中,本发明的方法和系统能将大于60吨/小时的二氧化碳吸收到吸收溶液中。在一些实施方案中,本发明的方法和系统能将大于70吨/小时的二氧化碳吸收到吸收溶液中。在一些实施方案中,本发明的方法和系统能将大于80吨/小时的二氧化碳吸收到吸收溶液中。在一些实施方案中,本发明的方法和系统能将大于90吨/小时的二氧化碳吸收到吸收溶液中。在一些实施方案中,本发明的方法和系统能将大于100吨/小时的二氧化碳吸收到吸收溶液中。在一些实施方案中,本发明的方法和系统能将大于110吨/小时的二氧化碳吸收到吸收溶液中。在一些实施方案中,本发明的方法和系统能将大于120吨/小时的二氧化碳吸收到吸收溶液中。在一些实施方案中,本发明的方法和系统能将大于130吨/小时的二氧化碳吸收到吸收溶液中。在一些实施方案中,本发明的方法和系统能将大于140吨/小时的二氧化碳吸收到吸收溶液中。在一些实施方案中,本发明的方法和系统能将大于150吨/小时的二氧化碳吸收到吸收溶液中。在一些实施方案中,本发明的方法和系统能将大于160吨/小时的二氧化碳吸收到吸收溶液中。在一些实施方案中,本发明的方法和系统能将大于170吨/小时的二氧化碳吸收到吸收溶液中。在一些实施方案中,本发明的方法和系统能将大于180吨/小时的二氧化碳吸收到吸收溶液中。在一些实施方案中,本发明的方法和系统能将大于190吨/小时的二氧化碳吸收到吸收溶液中。在一些实施方案中,本发明的方法和系统能将大于200吨/小时的二氧化碳吸收到吸收溶液中。In some embodiments, the methods and systems of the present invention are capable of absorbing greater than 20 tons/hour of carbon dioxide into the absorption solution, averaged over 72 hours of continuous operation. In some embodiments, the methods and systems of the present invention are capable of absorbing greater than 40 tons/hour of carbon dioxide into the absorption solution. In some embodiments, the methods and systems of the present invention are capable of absorbing greater than 60 tons/hour of carbon dioxide into the absorption solution. In some embodiments, the methods and systems of the present invention are capable of absorbing greater than 70 tons/hour of carbon dioxide into the absorption solution. In some embodiments, the methods and systems of the present invention are capable of absorbing greater than 80 tons/hour of carbon dioxide into the absorption solution. In some embodiments, the methods and systems of the present invention are capable of absorbing greater than 90 tons/hour of carbon dioxide into the absorption solution. In some embodiments, the methods and systems of the present invention are capable of absorbing greater than 100 tons/hour of carbon dioxide into the absorption solution. In some embodiments, the methods and systems of the present invention are capable of absorbing greater than 110 tons/hour of carbon dioxide into the absorption solution. In some embodiments, the methods and systems of the present invention are capable of absorbing greater than 120 tons/hour of carbon dioxide into the absorption solution. In some embodiments, the methods and systems of the present invention are capable of absorbing greater than 130 tons/hour of carbon dioxide into the absorption solution. In some embodiments, the methods and systems of the present invention are capable of absorbing greater than 140 tons/hour of carbon dioxide into the absorption solution. In some embodiments, the methods and systems of the present invention are capable of absorbing greater than 150 tons/hour of carbon dioxide into the absorption solution. In some embodiments, the methods and systems of the present invention are capable of absorbing greater than 160 tons/hour of carbon dioxide into the absorption solution. In some embodiments, the methods and systems of the present invention are capable of absorbing greater than 170 tons/hour of carbon dioxide into the absorption solution. In some embodiments, the methods and systems of the present invention are capable of absorbing greater than 180 tons/hour of carbon dioxide into the absorption solution. In some embodiments, the methods and systems of the present invention are capable of absorbing greater than 190 tons/hour of carbon dioxide into the absorption solution. In some embodiments, the methods and systems of the present invention are capable of absorbing greater than 200 tons/hour of carbon dioxide into the absorption solution.
在一些实施方案中,本发明提供使一定体积的水溶液与二氧化碳源接触以产生包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的组合物,其中组合物是溶液或浆液。使水溶液与二氧化碳源接触促进了CO2溶解到水溶液中,这产生了碳酸,一种与碳酸氢盐和碳酸盐平衡的物质。为了产生本发明的组合物(例如,包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的沉淀材料),在水溶液中从各种物质(例如碳酸、碳酸氢盐、水合氢离子等)中除去质子从而使得平衡朝着碳酸氢盐、碳酸盐或其间的某处移动。当质子被除去时,更多的CO2进入溶液中。在一些实施方案中,使用质子-除去剂和/或方法,同时使水溶液与CO2接触以提高CO2在反应单相中的吸收,其中pH可以保持恒定,增加或者甚至降低,随后快速除去质子(例如,通过添加碱),其在一些实施方案中可以引起沉淀材料的快速沉淀。通过任何适宜的方法,包括但不限于使用天然存在的质子-除去剂,使用微生物和真菌,使用合成的化学质子-除去剂,回收来自工业过程的废物物流,和使用电化学手段,质子可以从各种物质中除去(例如碳酸,碳酸氢盐,水合氢离子,等等)。In some embodiments, the present invention provides contacting a volume of an aqueous solution with a carbon dioxide source to produce a composition comprising carbonate, bicarbonate, or carbonate and bicarbonate, wherein the composition is a solution or a slurry. Contacting the aqueous solution with a source of carbon dioxide promotes the dissolution of CO2 into the aqueous solution, which produces carbonic acid, a species in equilibrium with bicarbonate and carbonate. To produce the compositions of the present invention (e.g., precipitation material comprising carbonates, bicarbonates, or carbonates and bicarbonates), various species (e.g., ) to shift the equilibrium toward bicarbonate, carbonate, or somewhere in between. When the protons are removed, more CO2 goes into solution. In some embodiments, a proton-removing agent and/or method is used while contacting an aqueous solution with CO to enhance CO uptake in a single phase of the reaction, where the pH can be held constant, increased, or even decreased, followed by rapid removal of protons (eg, by adding a base), which in some embodiments can cause rapid precipitation of the precipitation material. Protons can be removed from the Various substances are removed (such as carbonic acid, bicarbonate, hydronium ion, etc.).
天然存在的质子-除去剂包括在很宽泛的环境中存在的任何质子-除去剂,所述环境可以产生或具有碱性局部环境。一些实施方案提供了天然存在的质子-除去剂,包括当添加到溶液后产生碱性环境的矿物。这样的矿物包括但不局限于石灰(CaO);方镁石(MgO);氢氧化铁矿物(例如,针铁矿和褐铁矿);和火山灰。这样的矿物和包括这样的矿物的岩石的消化方法描述于2009年7月10日申请的美国专利申请No.12/501,217中,其全盘引入本文作为参考。一些实施方案提供使用天然存在的水体作为质子-除去剂源,该水体包括碳酸盐,硼酸盐,硫酸盐或硝酸盐碱度,或其一些组合。任何碱性盐水(例如,地表盐水,地下盐水,深盐水等)适用于本发明。在一些实施方案中,包括碳酸盐碱度的地表盐水提供了质子-除去剂源。在一些实施方案中,包括硼酸盐碱度的地表盐水提供了质子-除去剂源。在一些实施方案中,包括碳酸盐碱度的地下盐水提供了质子-除去剂源。在一些实施方案中,包括硼酸盐碱度的地下盐水提供了质子-除去剂源。在一些实施方案中,包括碳酸盐碱度的深盐水提供了质子-除去剂源。在一些实施方案中,包括硼酸盐碱度的深盐水提供了质子-除去剂源。天然碱性水体的实例包括但不局限于地表水源(例如碱湖如位于加利福尼亚的Mono湖)和地下水源(例如碱性含水层如位于加利福尼亚的Searles湖的深地质碱性含水层)。其它实施方案提供了使用来自干涸碱性水体的沉积物如沿非洲的东非大裂谷中的Natron湖的痂块。对于盐水和蒸发盐的另外的来源,参见2009年11月25日申请的美国临时专利申请No 61/264,564,其全盘引入本文作为参考。在一些实施方案中,在其正常代谢作用中分泌碱性分子或溶液的生物体用作质子-除去剂。这样的生物体的实例是产生碱性蛋白酶的真菌(例如,深海的真菌枯曲霉,最适pH为9)和产生碱性分子的细菌(例如,蓝细菌如鞘丝蓝细菌属,来自不列颠哥仑比亚省的Atlin湿地,其增加来自光合作用副产物的pH)。在一些实施方案中,使用生物体来产生质子-除去剂,其中生物体(例如,巴斯德氏芽胞杆菌,其将脲水解为氨)代谢污染物(例如脲)以产生质子-除去剂或包括质子-除去剂的溶液(例如,氨,氢氧化铵)。在一些实施方案中,生物体单独地由沉淀反应混合物中培养,其中质子-除去剂或包括质子-除去剂的溶液用于添加到沉淀反应混合物。在一些实施方案中,天然存在的或产生的酶与质子-除去剂组合使用来引起沉淀材料的沉淀。碳酸酐酶,其是一种由植物和动物产生的酶,在水溶液中加速了碳酸转化为碳酸氢盐。因而,碳酸酐酶可用于提高CO2的溶解并且加速沉淀材料的沉淀,如本文中更详细描述的。Naturally occurring proton-removing agents include any proton-removing agent that occurs in a wide variety of environments that may generate or have a basic local environment. Some embodiments provide naturally occurring proton-removing agents, including minerals that create a basic environment when added to a solution. Such minerals include, but are not limited to, lime (CaO); periclase (MgO); iron hydroxide minerals (eg, goethite and limonite); and pozzolans. Methods of digestion of such minerals and rocks comprising such minerals are described in US Patent Application No. 12/501,217, filed July 10, 2009, which is incorporated herein by reference in its entirety. Some embodiments provide for the use of a naturally occurring water body comprising carbonate, borate, sulfate, or nitrate alkalinity, or some combination thereof, as a source of proton-removing agent. Any alkaline brine (eg, surface brine, subterranean brine, deep brine, etc.) is suitable for use in the present invention. In some embodiments, surface brines including carbonate alkalinity provide a source of proton-removing agents. In some embodiments, surface brines including borate alkalinity provide a source of proton-removing agents. In some embodiments, subsurface brines including carbonate alkalinity provide a source of proton-removing agents. In some embodiments, subsurface brines including borate alkalinity provide a source of proton-removing agents. In some embodiments, deep brines including carbonate alkalinity provide a source of proton-removing agents. In some embodiments, deep brines including borate alkalinity provide a source of proton-removing agents. Examples of naturally alkaline water bodies include, but are not limited to, surface water sources (eg, alkaline lakes such as Lake Mono in California) and groundwater sources (eg, alkaline aquifers such as the deep geological alkaline aquifers of Lake Searles in California). Other embodiments provide for the use of sediment from dry alkaline water bodies such as the scabs of Lake Natron in the Great Rift Valley along Africa. For additional sources of brine and evaporated salt, see US Provisional Patent Application No. 61/264,564, filed November 25, 2009, which is incorporated herein by reference in its entirety. In some embodiments, organisms that secrete basic molecules or solutions as part of their normal metabolism are used as proton-removing agents. Examples of such organisms are fungi that produce alkaline proteases (e.g., the deep-sea fungus Aspergillus subtilis, with an optimum pH of 9) and bacteria that produce alkaline molecules (e.g., cyanobacteria such as the sp. Atlin wetlands in Colombia, which increases pH from photosynthesis by-products). In some embodiments, an organism is used to produce a proton-removing agent, wherein the organism (e.g., Bacillus pasteuria, which hydrolyzes urea to ammonia) metabolizes a pollutant (e.g., urea) to produce a proton-removing agent or Solutions including proton-removing agents (eg, ammonia, ammonium hydroxide). In some embodiments, the organism is cultured alone from the precipitation reaction mixture, wherein the proton-removing agent or a solution comprising the proton-removing agent is used to add to the precipitation reaction mixture. In some embodiments, naturally occurring or produced enzymes are used in combination with proton-removing agents to cause precipitation of precipitation material. Carbonic anhydrase, an enzyme produced by plants and animals, accelerates the conversion of carbonic acid to bicarbonate in aqueous solution. Thus, carbonic anhydrase can be used to increase the dissolution of CO2 and accelerate the precipitation of precipitation material, as described in more detail herein.
用于进行质子除去的化学试剂泛指合成的化学试剂,其大量生产并且是市售可得的。例如,用于除去质子的化学试剂包括但不局限于氢氧化物,有机碱,超强碱,氧化物,氨和碳酸盐。氢氧化物包括在溶液中提供氢氧化物阴离子的化学物种,包括,例如,氢氧化钠(NaOH),氢氧化钾(KOH),氢氧化钙(Ca(OH)2)或氢氧化镁(Mg(OH)2)。有机碱是含碳分子,其通常是含氮碱,包括伯胺如甲胺,仲胺如二异丙胺,叔胺如二异丙基乙胺,芳香族胺如苯胺,杂芳族化合物如吡啶,咪唑和苯并咪唑和其各种形式。在一些实施方案中,有机碱,其选自吡啶,甲胺,咪唑,苯并咪唑,组氨酸和磷腈,用于从各种物质(例如,碳酸,碳酸氢盐,水合氢离子等)中除去质子来制备本发明的组合物。在一些实施方案中,使用氨来将pH提高到足以制备本发明的组合物的水平。适于用作质子-除去剂的超强碱包括乙醇钠,氨基钠(NaNH2),氢化钠(NaH),丁基锂,二异丙基氨基化锂,二乙基氨基化锂和双(三甲基甲硅烷基)氨基化锂。例如包括氧化钙(CaO),氧化镁(MgO),氧化锶(SrO),氧化铍(BeO)和氧化钡(BaO)的氧化物也是合适的可使用的质子-除去剂。用于本发明的碳酸盐包括但不局限于碳酸钠。Chemicals used to perform proton removal generally refer to synthetic chemicals, which are produced in large quantities and are commercially available. For example, chemicals used to remove protons include, but are not limited to, hydroxides, organic bases, superbases, oxides, ammonia, and carbonates. Hydroxides include chemical species that provide hydroxide anions in solution, including, for example, sodium hydroxide (NaOH), potassium hydroxide (KOH), calcium hydroxide (Ca(OH)2) or magnesium hydroxide (Mg (OH)2). Organic bases are carbon-containing molecules that are usually nitrogen-containing bases and include primary amines such as methylamine, secondary amines such as diisopropylamine, tertiary amines such as diisopropylethylamine, aromatic amines such as aniline, and heteroaromatics such as pyridine , imidazole and benzimidazole and various forms thereof. In some embodiments, an organic base selected from the group consisting of pyridine, methylamine, imidazole, benzimidazole, histidine, and phosphazene is used to decompose various substances (e.g., carbonic acid, bicarbonate, hydronium ion, etc.) Compositions of the present invention are prepared by removing protons from . In some embodiments, ammonia is used to raise the pH to a level sufficient to prepare the compositions of the present invention. Superbases suitable for use as proton-removing agents include sodium ethoxide, sodium amide (NaNH 2 ), sodium hydride (NaH), butyllithium, lithium diisopropylamide, lithium diethylamide and bis( Lithium trimethylsilyl)amide. Oxides including, for example, calcium oxide (CaO), magnesium oxide (MgO), strontium oxide (SrO), beryllium oxide (BeO) and barium oxide (BaO) are also suitable usable proton-removing agents. Carbonates for use in the present invention include, but are not limited to, sodium carbonate.
除包括适用于本发明的阳离子(例如,Ca2+,Mg2+等)及其他合适的金属形式之外,来自各种工业方法的废物物流(即,工业废物物流)可以提供质子-除去剂。这样的废物物流包括但不局限于矿业废物;灰(例如,煤灰如飞灰,炉底灰,锅炉渣);熔渣(例如铁熔渣,磷熔渣);水泥窑废物(例如,水泥窑灰尘(CKD));炼油厂/石化精炼厂废物(例如油田和沼气层盐水);煤层废物(例如采气盐水和煤层盐水);纸处理废物;水软化废物盐水(例如,离子交换排出物);硅加工废物;农业废物;金属抛光废物;高pH纺织废物;和碱渣。矿业废物包括任何来自从地球提取金属或另一贵重或有用矿物的废物。在一些实施方案中,来自采矿的废物用于改变pH,其中废物选自来自Bayer铝提取方法的红泥;来自从海水中提取镁的废物(例如,Mg(OH)2如存在于加利福尼亚的Moss Landing中的那些);和来自包括浸出的采矿方法的废物。例如,红泥可用于改变pH,如以下文献中所述:2009年3月18日申请的美国临时专利申请No.61/161369,和2010年3月2日申请的题目为“Gas Stream Multi-Pollutants Control Systems And Methods”的PCT申请PCT/US10/25970和2010年3月2日申请的题目为“Gas Stream Multi-Pollutants ControlSystems And Methods”美国专利申请No.12/716,235,其以其全部内容引入本文作为参考。红泥,取决于加工条件和源材料(例如,铝矾土)可以包括Fe2O3,Al2O3,SiO2,Na2O,CaO,TiO2,K2O,MgO,CO2,S2O,MnO2,P2O5,所述物质的每一种以从极其丰富至较少丰富的顺序不严格地列出,并且所述物质的每一种用氧化物的形式表示,这为了方便起见。煤灰,水泥窑灰尘和熔渣,总起来说金属氧化物的废物来源,进一步描述于美国专利申请No.12/486692(申请日为2009年6月17日,公开号为US 2010-0000444A1,公开于2010年1月7日,题目为″Methods And Systems For Utilizing Waste Sources Of Metal Oxides.″),其公开内容全盘引入本文,它们可以单独地或者与其它质子-除去剂组合来使用从而提供用于本发明的质子-除去剂。农业废物,通过动物废物或过度的肥料使用,可以包含氢氧化钾(KOH)或氨(NH3)或两者。因而,农业废物在本发明的一些实施方案中可用作质子-除去剂。这种农业废物常常收集在池塘中,但是其也可渗流到含水层,其中其可以被获得和使用。Waste streams from various industrial processes (ie, industrial waste streams) can provide proton-removing agents, in addition to including cations (eg, Ca2+, Mg2+, etc.) and other suitable metal forms suitable for use in the present invention. Such waste streams include, but are not limited to, mining waste; ash (e.g., coal ash such as fly ash, bottom ash, boiler slag); slag (e.g., iron slag, phosphorus slag); cement kiln waste (e.g., cement kiln dust (CKD)); refinery/petrochemical refinery waste (e.g. oil field and biogas layer brine); coal seam waste (e.g. gas extraction brine and coal seam brine); paper processing waste; water softening waste brine (e.g. ion exchange effluent ); silicon processing waste; agricultural waste; metal polishing waste; high pH textile waste; and alkaline slag. Mining waste includes any waste that results from the extraction of a metal or another valuable or useful mineral from the earth. In some embodiments, waste from mining is used to alter the pH, wherein the waste is selected from red mud from Bayer's aluminum extraction process; waste from extraction of magnesium from seawater (e.g., Mg(OH)2 as found in Moss Landing); and waste from mining methods including leaching. For example, red mud can be used to alter pH as described in U.S. Provisional Patent Application No. 61/161369, filed March 18, 2009, and entitled "Gas Stream Multi- PCT Application PCT/US10/25970 for Pollutants Control Systems And Methods" and U.S. Patent Application No. 12/716,235, filed March 2, 2010, entitled "Gas Stream Multi-Pollutants Control Systems And Methods," which are incorporated in their entirety by This article serves as a reference. Red mud, depending on processing conditions and source material (for example, bauxite) can include Fe2O3 , Al2O3 , SiO2, Na2O , CaO, TiO2 , K2O, MgO, CO2 , S2O, MnO2 , P2O5, all Each of the species is listed in a loose order from most abundant to less abundant, and each of the species is represented as an oxide for convenience. Coal ash, cement kiln dust and slag, collectively waste sources of metal oxides, are further described in U.S. Patent Application No. 12/486692 (filed June 17, 2009, published as US 2010-0000444A1, Published on January 7, 2010, entitled "Methods And Systems For Utilizing Waste Sources Of Metal Oxides."), the disclosure of which is incorporated herein in its entirety, may be used alone or in combination with other proton-removing agents to provide in the proton-removing agent of the present invention. Agricultural waste, through animal waste or excessive fertilizer use, can contain potassium hydroxide (KOH) or ammonia (NH 3 ) or both. Thus, agricultural waste can be used as a proton-removing agent in some embodiments of the invention. This agricultural waste is often collected in ponds, but it can also percolate into aquifers where it can be harvested and used.
在本发明的一些实施方案中,灰可用于质子-除去剂,例如,为提高CO2-加料的水的pH值。灰可用作唯一的pH改变剂或者与一种或多种另外的pH改变剂一起使用。在某些实施方案中所考虑的是使用煤灰作为灰。如本发明中使用的煤灰是指在发电厂锅炉或燃煤熔炉(例如链式筛锅炉、旋风锅炉和流化床锅炉)中由燃烧粉碎的无烟煤,褐煤,烟煤或次烟煤产生的残余物。这样的煤灰包括飞灰,其是通过排气或烟道气来自熔炉所携带的细碎的煤灰;和炉底灰,其在熔炉底部以结块形式收集。In some embodiments of the invention, the ash can be used as a proton-removing agent, for example, to raise the pH of CO2 -fed water. Ash can be used as the sole pH altering agent or with one or more additional pH altering agents. Contemplated in certain embodiments is the use of coal ash as ash. Coal ash as used in the present invention refers to the residue resulting from the combustion of pulverized anthracite, lignite, bituminous or sub-bituminous coal in power plant boilers or coal-fired smelters such as chain screen boilers, cyclone boilers and fluidized bed boilers . Such coal ash includes fly ash, which is finely divided coal ash carried over from the furnace by exhaust or flue gas, and bottom ash, which collects in agglomerated form at the bottom of the furnace.
飞灰通常是高度非均质的,并且包括玻璃状颗粒与各种可识别的晶体相如石英、富铝红柱石和各种铁氧化物的混合物。所考虑的飞灰包括F型和C型飞灰。上述F型和C型飞灰是由CSA Standard A23.5和ASTM C618限定的。这些类别之间的主要差别是钙,二氧化硅,氧化铝的数量和灰中的铁含量。飞灰的化学性质主要受所燃烧的煤炭(即无烟煤、烟煤和褐煤)的化学内容物的影响。所考虑的飞灰包括大量的二氧化硅(二氧化硅,SiO2)(无定形和晶体两者)和石灰(氧化钙,CaO,氧化镁,MgO)。Fly ashes are generally highly heterogeneous and include a mixture of glassy particles with various identifiable crystalline phases such as quartz, mullite, and various iron oxides. Fly ashes considered include Type F and Type C fly ash. The aforementioned Types F and C fly ash are defined by CSA Standard A23.5 and ASTM C618. The main difference between these categories is the amount of calcium, silica, alumina and iron content in the ash. The chemical properties of fly ash are mainly influenced by the chemical content of the coal being burned (ie, anthracite, bituminous and lignite). Fly ashes considered include large amounts of silica (silica, SiO2 ) (both amorphous and crystalline) and lime (calcium oxide, CaO, magnesium oxide, MgO).
以下表3提供了各种类型的用于本发明实施方案的飞灰的化学成分。Table 3 below provides the chemical composition of various types of fly ash used in embodiments of the present invention.
表3.各种类型的飞灰的化学成分。Table 3. Chemical composition of various types of fly ash.
燃烧较硬的、长龄的无烟煤和烟煤一般地产生F类飞灰。F类飞灰本质上是火山灰质的,并且包含小于10%石灰(CaO)。由燃烧较年轻的褐煤或亚烟煤产生的飞灰,除具有火山灰质的性能以外,还具有一些自粘合性能。在水的存在下,C类飞灰将硬化并且随时间获得强度。C类飞灰通常包含大于20%石灰(CaO)。在C类飞灰中碱和硫酸盐(SO4)含量通常是较高的。Combustion of harder, older anthracite and bituminous coals generally produces Class F fly ash. Class F fly ash is pozzolanic in nature and contains less than 10% lime (CaO). Fly ash produced by burning younger lignite or sub-bituminous coals has some self-binding properties in addition to pozzolanic properties. In the presence of water, Class C fly ash will harden and gain strength over time. Class C fly ash typically contains greater than 20% lime (CaO). The alkali and sulfate (SO 4 ) content is generally higher in Class C fly ash.
飞灰材料固化,同时悬浮在排气气体中并且使用各种方法来收集,例如通过静电沉降器或过滤袋。因为颗粒固化,同时悬浮在排气气体中,飞灰颗粒通常是球状的并且尺寸为0.5μm-100μm。所考虑的飞灰包括其中至少80wt%包括小于45微米的颗粒的那些。在本发明的某些实施方案中,还考虑的是使用高度碱性的流化床燃烧器(FBC)飞灰。The fly ash material solidifies while suspended in the exhaust gas and is collected using various methods, such as by electrostatic precipitators or filter bags. Fly ash particles are generally spherical and have a size of 0.5 μm-100 μm as the particles solidify while being suspended in the exhaust gas. Fly ashes contemplated include those in which at least 80% by weight comprise particles smaller than 45 microns. In certain embodiments of the present invention, the use of highly alkaline fluidized bed combustor (FBC) fly ash is also contemplated.
在本发明实施方案中还考虑的是使用炉底灰。炉底灰是来自煤燃烧的在煤燃烧锅炉中以结块形式形成的。这样的燃烧锅炉可以是液态排渣锅炉或固态排渣锅炉。当在液态或固态排渣锅炉中产生时,炉底灰被水淬。猝灭获得了具有其中90%落入0.1mm-20mm粒度范围的尺寸的结块,其中炉底灰结块在该范围中具有广泛分布的结块尺寸。炉底灰的主要化学组分是二氧化硅和氧化铝,以及较少量的Fe,Ca,Mg,Mn,Na和K以及硫和碳的氧化物。Also contemplated in embodiments of the present invention is the use of bottom ash. Bottom ash from coal combustion is formed in the form of agglomerates in coal fired boilers. Such fired boilers may be liquid slagging boilers or solid slagging boilers. Bottom ash is water quenched when produced in liquid or solid slagging boilers. Quenching yielded agglomerates with a size where 90% fell within the 0.1 mm-20 mm particle size range, with bottom ash agglomerates having a broad distribution of agglomerate sizes within this range. The main chemical components of bottom ash are silica and alumina, with smaller amounts of Fe, Ca, Mg, Mn, Na and K, and oxides of sulfur and carbon.
在某些实施方案中还考虑的是使用火山灰作为灰。火山灰由小的火山碎屑(即,由火山喷发产生的粉碎的岩石和玻璃的屑,直径小于2毫米(0.079in))组成。Also contemplated in certain embodiments is the use of pozzolans as ash. Volcanic ash consists of small pyroclastics (ie, shavings of crushed rock and glass produced by volcanic eruptions, less than 2 millimeters (0.079 in) in diameter).
在本发明的一种实施方案中,水泥窑灰尘(CKD)被添加到反应容器,作为改变pH的手段。由其产生灰和/或CKD的燃料的性质和所述燃料的燃烧方式,将影响所得的灰和/或CKD的化学组成。因此,灰和/或CKD可用作调节pH的手段的一部分,或者唯一的手段,并且可以与特定的灰和/或CKD一起,基于灰和/或CKD的化学组成,使用各种其它组分。In one embodiment of the invention, cement kiln dust (CKD) is added to the reaction vessel as a means of changing the pH. The nature of the fuel from which the ash and/or CKD is produced and the manner in which the fuel is burned will affect the chemical composition of the resulting ash and/or CKD. Thus, ash and/or CKD may be used as part of, or the only means of adjusting pH, and various other components may be used along with a particular ash and/or CKD, based on the chemical composition of the ash and/or CKD .
在本发明的实施方案中,灰被添加到反应作为这些另外的反应物的一个来源,以产生碳酸盐矿物沉淀物,其包含一种或多种组分如无定形二氧化硅,结晶二氧化硅,硅酸钙,硅酸钙铝,或任何其它部分,其可以在碳酸盐矿物沉淀过程中由灰的反应产生。In an embodiment of the invention, ash is added to the reaction as a source of these additional reactants to produce a carbonate mineral precipitate comprising one or more components such as amorphous silica, crystalline di Silica, calcium silicate, calcium aluminum silicate, or any other moiety that may be produced by the reaction of ash during carbonate mineral precipitation.
用于本发明的灰可以与水接触以获得期望的pH改变,这使用任何适宜的流程,例如,通过将一定量的灰置于装水的反应器,其中添加灰的量足以提高pH至期望水平,通过使水流过一定量的灰,例如,以塔或床等的形式。The ash used in the present invention can be contacted with water to obtain the desired pH change using any suitable procedure, for example, by placing an amount of ash in a water-filled reactor where the amount of ash added is sufficient to raise the pH to the desired pH. Horizontally, by passing water through a volume of ash, for example, in the form of towers or beds, etc.
在某些实施方案中,其中pH没有被提高到12或更高的水平,用于方法中的飞灰,例如如下所述,未必溶解,而是相反将以颗粒物组合物的形式保持。这种未溶解的灰可以与其余的反应产物分离,例如,滤出,以便随后使用。或者,水可以流过一定量的灰,后者是以固定配置结构提供的,例如在塔或同功结构中,这提供了液体流过灰但是不允许灰固体随液体流出该结构。这种实施方案不需要分离未溶解的灰与产物液体。在其它实施方案中,其中pH超过12,灰溶解并且提供了火山灰质的产物,例如,如其它地方详细所述的。In certain embodiments, where the pH is not raised to a level of 12 or higher, the fly ash used in the process, such as described below, will not necessarily dissolve, but instead will remain in the form of a particulate composition. This undissolved ash can be separated from the rest of the reaction product, eg filtered off, for subsequent use. Alternatively, the water may flow through a volume of ash provided in a fixed arrangement, such as in a tower or co-function structure, which provides liquid flow through the ash but does not allow the ash solids to flow out of the structure with the liquid. This embodiment does not require separation of undissolved ash from product liquid. In other embodiments, where the pH exceeds 12, the ash dissolves and provides a pozzolanic product, eg, as described in detail elsewhere.
在本发明的实施方案中,其中灰在沉淀过程中被利用,首先,灰可以通过如静电沉降的手段从烟道气中除去,或者可以通过烟道气而被直接利用。在本发明的实施方案中使用灰可以提供反应物如氧化铝或二氧化硅,除了提高pH之外。In embodiments of the present invention, where the ash is utilized in the precipitation process, firstly, the ash can be removed from the flue gas by means such as electrostatic precipitation, or can be utilized directly through the flue gas. The use of ash in embodiments of the invention may provide reactants such as alumina or silica, in addition to raising the pH.
在本发明的某些实施方案中,熔渣用作pH改变剂,例如,以提高CO2加料的水的pH值。熔渣可用作唯一的pH改变剂或者与一种或多种另外的pH改变剂,如灰等一起使用。熔渣是由加工金属产生的,并且可以包含钙和镁氧化物以及铁,硅和铝化合物。在某些实施方案中,使用熔渣作为pH改变材料提供了通过引入反应性的硅和氧化铝至沉淀的产物的额外利益。所考虑的熔渣包括但不局限于来自生铁熔炼的高炉熔渣,来自钢的电弧或高炉加工的熔渣,铜熔渣,镍熔渣和磷熔渣。In certain embodiments of the invention, slag is used as a pH altering agent, eg, to increase the pH of CO fed water. Slag can be used as the sole pH changing agent or with one or more additional pH changing agents such as ash or the like. Slag is produced by working metal and can contain calcium and magnesium oxides as well as iron, silicon and aluminum compounds. In certain embodiments, the use of slag as a pH changing material provides additional benefits by introducing reactive silica and alumina to the precipitated product. Slags considered include, but are not limited to, blast furnace slags from pig iron smelting, slags from electric arc or blast furnace processing of steel, copper slags, nickel slags, and phosphorus slags.
通过从溶质(例如,碳酸或碳酸氢盐的脱质子化)或者从溶剂(例如水合氢离子或水的脱质子化)中除去质子,电化学方法提供了用于在溶液中从各种物质中除去质子的另一手段。例如,如果由CO2溶解产生质子匹配或超过从溶质分子中电化学质子除去的话,溶剂的脱质子化可以形成。在一些实施方案中,使用低压电化学方法来除去质子,例如,当CO2被溶解在沉淀反应混合物或沉淀反应混合物的前体溶液(即,可能或未必包含二价阳离子的溶液)中时。在一些实施方案中,溶于不包含二价阳离子的水溶液中的CO2通过低压电化学方法处理而从由CO2的溶解产生的碳酸,碳酸氢盐,水合氢离子,或任何物质或其组合中除去质子。低压电化学方法在2,1.9,1.8,1.7,或1.6V或更小,如1.5,1.4,1.3,1.2,1.1V或更小,如1V或更小,如0.9V或更小,0.8V或更小,0.7V或更小,0.6V或更小,0.5V或更小,0.4V或更小,0.3V或更小,0.2V或更小,或0.1V或更小的平均电压下操作。没有产生氯气的低压电化学方法便于用于本发明的系统和方法中。用于除去质子的没有产生氧气的低压电化学方法也便于用于本发明的系统和方法中。在一些实施方案中,在阳极低压方法不产生任何气体。在一些实施方案中,在阴极低压电化学方法产生氢气并且将其输送到阳极,其中氢气转化为质子。未产生氢气的电化学方法也可以是适宜的。在有些情况下,用于除去质子的电化学方法没有产生任何气体副产物。进行质子除去的电化学方法进一步描述于以下文献中:美国专利申请12/344,019(申请日为2008年12月24日);美国专利申请12/375,632(申请日为2008年12月23日);国际专利申请PCT/US08/088242(申请日为2008年12月23日);国际专利申请PCT/US09/32301(申请日为2009年1月28日);和国际专利申请PCT/US09/48511(申请日为2009年6月24日),其每个以其全部内容引入本文作为参考。By removing protons from solutes (e.g., deprotonation of carbonic acid or bicarbonate) or from solvents (e.g., deprotonation of hydronium ions or water), electrochemical methods provide a method for removing protons from various substances in solution. Another means of removing protons. For example, deprotonation of the solvent can form if the proton matching produced by CO2 dissolution exceeds or exceeds the electrochemical proton removal from the solute molecule. In some embodiments, low-voltage electrochemical methods are used to remove protons, for example, when CO is dissolved in the precipitation reaction mixture or a precursor solution (ie, a solution that may or may not contain divalent cations) of the precipitation reaction mixture. In some embodiments, CO dissolved in an aqueous solution that does not contain divalent cations is processed by low-pressure electrochemical methods to generate carbonic acid, bicarbonate, hydronium ions, or any species or combinations thereof that result from the dissolution of CO . remove protons. Low voltage electrochemical method at 2, 1.9, 1.8, 1.7, or 1.6V or less, such as 1.5, 1.4, 1.3, 1.2, 1.1V or less, such as 1V or less, such as 0.9V or less, 0.8V or less, 0.7V or less, 0.6V or less, 0.5V or less, 0.4V or less, 0.3V or less, 0.2V or less, or 0.1V or less under the average voltage operate. Low pressure electrochemical methods that do not generate chlorine gas are convenient for use in the systems and methods of the present invention. Low pressure electrochemical methods for removing protons that do not generate oxygen are also convenient for use in the systems and methods of the present invention. In some embodiments, no gas is produced at the anode low pressure process. In some embodiments, hydrogen gas is produced electrochemically at a low pressure at the cathode and delivered to the anode where it is converted to protons. Electrochemical methods that do not generate hydrogen may also be suitable. In some cases, the electrochemical methods used to remove protons did not produce any gaseous by-products. Electrochemical methods for proton removal are further described in: US Patent Application 12/344,019 (filed December 24, 2008); US Patent Application 12/375,632 (filed December 23, 2008); International Patent Application PCT/US08/088242 (dated December 23, 2008); International Patent Application PCT/US09/32301 (dated January 28, 2009); and International Patent Application PCT/US09/48511 ( filed June 24, 2009), each of which is incorporated herein by reference in its entirety.
或者,可以使用电化学方法产生碱性分子(例如,氢氧化物),例如,通过氯碱工艺或其变体(例如,低压变体)。电极(即,阴极和阳极)可以存在于包含水溶液或废气-加料的(例如,CO2-加料的)溶液的装置中,并且选择性阻隔物,如膜,可以分开电极。用于除去质子的电化学系统和方法可以产生副产物(例如,氢气),其可以被收集并且用于其它目的。可用于本发明的系统和方法的另外的电化学法包括但不局限于以下文献中所述的那些:美国临时专利申请61/081,299(申请日为2008年7月16日)和美国临时专利申请61/091,729,其公开内容引入本文作为参考。可以使用上述的质子-除去剂源和进行质子除去的方法的组合。Alternatively, alkaline molecules (eg, hydroxides) can be generated electrochemically, eg, by the chlor-alkali process or variations thereof (eg, low pressure variants). Electrodes (ie, cathode and anode) can be present in a device containing an aqueous or exhaust-fed (eg, CO2 -fed) solution, and a selective barrier, such as a membrane, can separate the electrodes. Electrochemical systems and methods for removing protons can produce by-products (eg, hydrogen gas), which can be collected and used for other purposes. Additional electrochemical methods that may be used in the systems and methods of the present invention include, but are not limited to, those described in U.S. Provisional Patent Application 61/081,299 (filed July 16, 2008) and U.S. Provisional Patent Application 61/091,729, the disclosure of which is incorporated herein by reference. Combinations of the proton-removing agent sources and methods of performing the proton removal described above may be used.
在其中使用电化学法除去质子和/或产生碱的实施方案中,通常还产生了酸物流如HCl物流,并且这种物流,单独地或酸的任何其它适宜的源,或其组合,可以被用于提高例如含镁的无机物,如橄榄石或蛇纹石,或钙源如水泥废物的溶解。溶解可以进一步通过声处理方法来提高,这可以产生局部化的极端温度和压力袋,提高反应速率达一百至超过一百万倍。这样的方法是本领域已知的。In embodiments where electrochemical methods are used to remove protons and/or generate bases, an acid stream, such as an HCl stream, is typically also produced, and this stream, alone or any other suitable source of acid, or combination thereof, can be For enhancing the dissolution of, for example, magnesium-containing inorganics such as olivine or serpentine, or calcium sources such as cement waste. Dissolution can be further enhanced by sonication methods, which can create pockets of localized extremes of temperature and pressure, increasing reaction rates by a factor of one hundred to over a million. Such methods are known in the art.
在一些实施方案中,本发明的方法允许大量镁和,有时候,钙被添加到在本发明的一些实施方案中使用的水中,这增加了在单个沉淀步骤中每单位的水可形成的沉淀物的量,当与从工业水源,例如,海水或其它盐水源溶解CO2的方法结合时,允许令人惊讶地高产率含碳酸盐的沉淀物。在一些实施方案中,本发明的方法包括通过对CO2已经从CO2的气源溶入其中的水进行沉淀步骤而从气源,例如,CO2的工业气源如来自发电厂的烟道气,或如来自水泥厂的废气中除去CO2的方法,其中沉淀步骤提供了如下数量的沉淀物:在单个沉淀步骤中10g/L或更多,在单个沉淀步骤中15g/L或更多,在单个沉淀步骤中20g/L或更多,在单个沉淀步骤中25g/L或更多,在单个沉淀步骤中30g/L或更多,在单个沉淀步骤中40g/L或更多,在单个沉淀步骤中50g/L或更多,在单个沉淀步骤中60g/L或更多,在单个沉淀步骤中70g/L或更多,在单个沉淀步骤中80g/L或更多,在单个沉淀步骤中90g/L或更多,在单个沉淀步骤中100g/L或更多,在单个沉淀步骤中125g/L或更多,或在单个沉淀步骤中150g/L或更多。In some embodiments, the methods of the invention allow large amounts of magnesium and, sometimes, calcium to be added to the water used in some embodiments of the invention, which increases the precipitate that can be formed per unit of water in a single precipitation step This amount, when combined with a method of dissolving CO2 from industrial water sources, eg, seawater or other brine sources, allows for surprisingly high yields of carbonate-containing precipitates. In some embodiments, the method of the invention comprises removing CO from a gas source, e.g., an industrial gas source such as a flue from a power plant, by subjecting the CO to water into which the CO has been dissolved from the gas source. gas, or as a method of removing CO2 from waste gas from a cement plant, where the precipitation step provides an amount of precipitate: 10 g/L or more in a single precipitation step, 15 g/L or more in a single precipitation step , 20 g/L or more in a single precipitation step, 25 g/L or more in a single precipitation step, 30 g/L or more in a single precipitation step, 40 g/L or more in a single precipitation step, in 50g/L or more in a single precipitation step, 60g/L or more in a single precipitation step, 70g/L or more in a single precipitation step, 80g/L or more in a single precipitation step, in a single precipitation step 90 g/L or more in a step, 100 g/L or more in a single precipitation step, 125 g/L or more in a single precipitation step, or 150 g/L or more in a single precipitation step.
在一些实施方案中,本发明的方法包括通过使CO2已经从CO2的气源(例如二氧化碳的工业源)溶入其中的水(例如海水,盐水,吸收溶液)经受沉淀条件而从气源,例如,CO2的工业气源如来自发电厂的烟道气,或如来自水泥厂的废气,除去CO2的方法,其中沉淀条件提供如下数量的沉淀物:在连续施加沉淀条件的72小时的时间段内平均136吨/小时至445吨/小时。In some embodiments, the method of the present invention comprises removing CO from a gas source by subjecting water (e.g., seawater, brine, absorption solution) into which CO has been dissolved from a gas source of CO (e.g., an industrial source of carbon dioxide) to precipitation conditions. , for example, an industrial gas source of CO2 such as flue gas from a power plant, or exhaust gas such as from a cement plant, a process for removing CO2 , wherein the precipitation conditions provide a deposit of the following amount: During 72 hours of continuous application of the precipitation conditions An average of 136 tons/hour to 445 tons/hour during the period of time.
在一些实施方案中,本发明的方法包括通过使CO2已经从CO2的气源(例如二氧化碳的工业源)溶入其中的水(例如海水,盐水,吸收溶液)经受沉淀条件而从气源,例如,CO2的工业气源如来自发电厂的烟道气,或如来自水泥厂的废气,除去CO2的方法,其中沉淀条件提供如下数量的沉淀物:在连续施加沉淀条件的72小时的时间段内平均2.6克的沉淀物/每升吸收溶液至26.11克的沉淀物/每升吸收溶液。在一些实施方案中,沉淀条件提供如下数量的沉淀物:在连续施加沉淀条件的72小时的时间段内平均5.2克的沉淀物/每升吸收溶液至26.11克的沉淀物/每升吸收溶液。在一些实施方案中,沉淀条件提供如下数量的沉淀物:在连续施加沉淀条件的72小时的时间段内平均7.83克的沉淀物/每升吸收溶液至26.11克的沉淀物/每升吸收溶液,如9.14至26.11,如10.44至26.11,如11.75至26.11,如13.05至26.11,如14.36至26.11,如15.66至26.11,如16.97至26.11,如18.27至26.11,如19.58至26.11,如20.88至26.11,如22.19至26.11,如23.5至26.11,如24.8至26.11克的沉淀物/每升吸收溶液。In some embodiments, the methods of the present invention comprise removing CO from a gas source by subjecting water (e.g., seawater, brine, absorption solution) into which CO has been dissolved from a gas source of CO (e.g., an industrial source of carbon dioxide) to precipitation conditions. , for example, an industrial gas source of CO2 such as flue gas from a power plant, or exhaust gas such as from a cement plant, a process for removing CO2 , wherein the precipitation conditions provide a deposit of the following amount: During 72 hours of continuous application of the precipitation conditions The time period averaged from 2.6 grams of sediment/liter of absorption solution to 26.11 grams of sediment/liter of absorption solution. In some embodiments, the precipitation conditions provide an amount of precipitate ranging from an average of 5.2 grams of precipitate per liter of absorption solution to 26.11 grams of precipitate per liter of absorption solution over a period of 72 hours of continuous application of the precipitation condition. In some embodiments, the precipitation conditions provide an amount of precipitate ranging from an average of 7.83 grams of precipitate per liter of absorption solution to 26.11 grams of precipitate per liter of absorption solution over a 72 hour period of continuous application of the precipitation conditions, Such as 9.14 to 26.11, such as 10.44 to 26.11, such as 11.75 to 26.11, such as 13.05 to 26.11, such as 14.36 to 26.11, such as 15.66 to 26.11, such as 16.97 to 26.11, such as 18.27 to 26.11, such as 19.58 to 26.11, such as 20.88 to 26. Such as 22.19 to 26.11, such as 23.5 to 26.11, such as 24.8 to 26.11 grams of precipitate per liter of absorption solution.
在一些实施方案中,沉淀物包括碳酸镁;在一些实施方案中沉淀物包括碳酸钙;在一些实施方案中,沉淀物包括碳酸镁和碳酸钙和/或碳酸镁/碳酸钙。在一些实施方案中,在单个沉淀步骤中产生的沉淀材料中的镁与钙的比值为至少0.5∶1,或至少1∶1,或至少2∶1,或至少3∶1,或至少4∶1,或至少5∶1,或至少6∶1,或至少7∶1,或至少8∶1,或至少9∶1,或至少10∶1。在一些实施方案中,在单个沉淀步骤中产生的沉淀材料中的镁与钙的比值为至少2∶1。在一些实施方案中,在单个沉淀步骤中产生的沉淀材料中的镁与钙的比值为至少4∶1。在一些实施方案中,在单个沉淀步骤中产生的沉淀材料中的镁与钙的比值为至少6∶1。在一些实施方案中,沉淀物包含碳酸钙和碳酸镁,并且包含允许至少一部分碳酸盐中的碳被追溯到化石燃料源头的组分。In some embodiments, the precipitate includes magnesium carbonate; in some embodiments, the precipitate includes calcium carbonate; in some embodiments, the precipitate includes magnesium carbonate and calcium carbonate and/or magnesium carbonate/calcium carbonate. In some embodiments, the ratio of magnesium to calcium in the precipitation material produced in a single precipitation step is at least 0.5:1, or at least 1:1, or at least 2:1, or at least 3:1, or at least 4:1. 1, or at least 5:1, or at least 6:1, or at least 7:1, or at least 8:1, or at least 9:1, or at least 10:1. In some embodiments, the ratio of magnesium to calcium in the precipitation material produced in a single precipitation step is at least 2:1. In some embodiments, the ratio of magnesium to calcium in the precipitation material produced in a single precipitation step is at least 4:1. In some embodiments, the ratio of magnesium to calcium in the precipitation material produced in a single precipitation step is at least 6:1. In some embodiments, the precipitate comprises calcium carbonate and magnesium carbonate, and comprises a component that allows at least a portion of the carbon in the carbonate to be traced back to a fossil fuel source.
在一些实施方案中,沉淀物包括碳酸镁;在一些实施方案中沉淀物包括碳酸钙;在一些实施方案中,沉淀物包括碳酸镁和碳酸钙和/或碳酸镁/碳酸钙。在一些实施方案中,在施加沉淀条件的72小时内平均产生的沉淀材料中的镁与钙的比值为至少0.5∶1,或至少1∶1,或至少2∶1,或至少3∶1,或至少4∶1,或至少5∶1,或至少6∶1,或至少7∶1,或至少8∶1,或至少9∶1,或至少10∶1。在一些实施方案中,在施加沉淀条件的72小时内平均产生的沉淀材料中的镁与钙的比值为至少2∶1。在一些实施方案中,在施加沉淀条件的72小时内平均产生的沉淀材料中的镁与钙的比值为至少4∶1。在一些实施方案中,在施加沉淀条件的72小时内平均产生的沉淀材料中的镁与钙的比值为至少6∶1。在一些实施方案中,沉淀物包含碳酸钙和碳酸镁,并且包含允许至少一部分碳酸盐中的碳被追溯到化石燃料源头的组分。In some embodiments, the precipitate includes magnesium carbonate; in some embodiments, the precipitate includes calcium carbonate; in some embodiments, the precipitate includes magnesium carbonate and calcium carbonate and/or magnesium carbonate/calcium carbonate. In some embodiments, the ratio of magnesium to calcium in the precipitation material produced on average within 72 hours of applying the precipitation conditions is at least 0.5:1, or at least 1:1, or at least 2:1, or at least 3:1, Or at least 4:1, or at least 5:1, or at least 6:1, or at least 7:1, or at least 8:1, or at least 9:1, or at least 10:1. In some embodiments, the ratio of magnesium to calcium in the precipitation material produced is, on average, at least 2:1 within 72 hours of applying the precipitation conditions. In some embodiments, the precipitation material produced has a ratio of magnesium to calcium of at least 4:1 on average within 72 hours of applying the precipitation conditions. In some embodiments, the precipitation material produced has a ratio of magnesium to calcium of at least 6:1 on average within 72 hours of applying the precipitation conditions. In some embodiments, the precipitate comprises calcium carbonate and magnesium carbonate, and comprises a component that allows at least a portion of the carbon in the carbonate to be traced back to a fossil fuel source.
如上所述,本发明的方法包括使水(其可能或未必在加料反应器中加料以CO2,如上所述)经受足以产生存储稳定的沉淀的二氧化碳埋存产物的沉淀条件。可以使用任何适宜的沉淀条件,所述条件导致产生了期望的埋存产物。As noted above, the method of the present invention involves subjecting water (which may or may not be fed with CO2 in a feed reactor, as described above) to precipitation conditions sufficient to produce a storage-stable precipitated carbon dioxide sequestration product. Any suitable precipitation conditions that result in the desired sequestered product may be used.
所关心的沉淀条件包括调节水的物理环境以产生期望的沉淀产物的那些。例如,水的温度可以被调节到适用于发生期望的产物沉淀的水平。在这样的实施方案中,水的温度可以被调节到0℃至90℃,如20℃至50℃和包括25℃至45℃的值。因而,尽管给定一组的沉淀条件可能具有范围为0℃至100℃的温度,但是在某些实施方案中可以调节温度以产生期望的沉淀物。使用任何适宜的流程,可以调节水的温度。在有些情况下,使用由低或零二氧化碳排放源,例如太阳能资源、风能资源、水电能资源、地热能资源,由烟道气的废热(其可以高达500℃,当需要加热时)等,产生的能量调节温度。Precipitation conditions of interest include those that adjust the physical environment of the water to produce the desired precipitation product. For example, the temperature of the water can be adjusted to a level suitable for the desired precipitation of the product to occur. In such embodiments, the temperature of the water may be adjusted to values from 0°C to 90°C, such as 20°C to 50°C and inclusive. Thus, while a given set of precipitation conditions may have temperatures ranging from 0°C to 100°C, in certain embodiments the temperature may be adjusted to produce the desired precipitate. The temperature of the water can be adjusted using any suitable procedure. In some cases, the use of low or zero carbon dioxide emission sources, such as solar energy resources, wind energy resources, hydroelectric energy resources, geothermal energy resources, waste heat from flue gases (which can be as high as 500 ° C when heating is required), etc., energy to regulate temperature.
尽管在给定的沉淀过程期间水的pH值可以为7至14,但是在有些情况下,pH值被提高到碱性水平,以便产生期望的沉淀产物。在这种实施方案中,pH值被提高到足以引起期望的CO2-埋存产物沉淀的水平,如上所述。因而,pH值可以被提高到9.5或更高,如10或更高,包括10.5或更高。在期望的情况下,pH值可以被提高到最小化(如果没有消除的话)沉淀期间产生CO2的水平。例如,pH值可以被提高到10或更高,如11或更高的值。在某些实施方案中,pH值被提高到7-11,如8-11,包括9-11,例如10-11。在该步骤中,pH值可以被提高到期望的碱性水平并且维持在期望的碱性水平,使得pH值被维持在恒定的碱性水平,或者pH值可以在两个或更多个不同的碱性水平之间转变或循环,根据期望的要求。Although the pH of the water may range from 7 to 14 during a given precipitation process, in some cases the pH is raised to alkaline levels in order to produce the desired precipitation product. In such embodiments, the pH is raised to a level sufficient to cause precipitation of the desired CO2 -storage product, as described above. Thus, the pH may be raised to 9.5 or higher, such as 10 or higher, including 10.5 or higher. Where desired, the pH can be raised to a level that minimizes, if not eliminates, CO2 production during precipitation. For example, the pH may be raised to a value of 10 or higher, such as 11 or higher. In certain embodiments, the pH is increased to 7-11, such as 8-11, including 9-11, such as 10-11. In this step, the pH can be raised to and maintained at the desired alkaline level such that the pH is maintained at a constant alkaline level, or the pH can be varied between two or more different Shift or cycle between alkaline levels, depending on desired requirements.
在正常的海水中,93%的溶解的CO2处于碳酸氢盐离子(HCO3-)的形式,6%处于碳酸盐离子(CO32-)的形式。当碳酸钙从正常的海水中沉淀时,释放CO2。在淡水中,高于pH 10.33,大于90%的碳酸盐处于碳酸盐离子的形式,在碳酸钙沉淀期间没有释放CO2。在海水中,这种转变发生在略低的pH下,接近于9.7的pH值。虽然在给定的沉淀过程期间,用于方法中的水的pH值可以为5至14,但是在某些实施方案中pH被提高到碱性水平以便驱使碳酸盐化合物以及其他化合物如化合物沉淀,根据期望而定。在这些实施方案的一些中,pH被提高到这样的水平,其最小化,如果没有消除的话,在沉淀期间CO2的产生,这引起溶解的CO2,例如碳酸盐和碳酸氢盐的形式的,被捕获在碳酸盐化合物沉淀物中。在这种实施方案中,pH可以被提高到9或更高,如10或更高,包括11或更高。In normal seawater, 93% of the dissolved CO2 is in the form of bicarbonate ions ( HCO3- ) and 6% is in the form of carbonate ions ( CO32- ). When calcium carbonate precipitates from normal seawater, CO2 is released. In fresh water, above pH 10.33, more than 90% of the carbonate is in the form of carbonate ions, and no CO2 is released during calcium carbonate precipitation. In seawater, this transition occurs at a slightly lower pH, closer to a pH of 9.7. While the pH of the water used in the process may range from 5 to 14 during a given precipitation process, in some embodiments the pH is raised to alkaline levels in order to drive the precipitation of carbonate compounds as well as other compounds such as , depending on expectations. In some of these embodiments, the pH is raised to a level that minimizes, if not eliminates, the production of CO2 during precipitation, which causes dissolved CO2 , for example in the form of carbonates and bicarbonates , are trapped in carbonate compound precipitates. In such embodiments, the pH may be raised to 9 or higher, such as 10 or higher, including 11 or higher.
如上所述,水源(例如,含碱土金属离子的水)的pH值是使用任何适宜方法提高的。在某些实施方案中,可以使用pH提高剂,其中这样的试剂的实例包括氧化物(氧化钙,氧化镁),氢氧化物(例如,氢氧化钾,氢氧化钠,水镁石(Mg(OH)2等),碳酸盐(例如,碳酸钠)等。As noted above, the pH of the water source (eg, alkaline earth metal ion-containing water) is raised using any suitable method. In certain embodiments, pH increasing agents may be used, where examples of such agents include oxides (calcium oxide, magnesium oxide), hydroxides (e.g., potassium hydroxide, sodium hydroxide, brucite (Mg( OH)2, etc.), carbonates (for example, sodium carbonate) and the like.
如上所指出,灰(或熔渣,在某些实施方案中)用于某些实施方案作为改变水的pH值至期望的水平的唯一方法。在其它实施方案中,与使用灰一起,用于一种或多种另外的pH改变流程。As noted above, ash (or slag, in some embodiments) is used in some embodiments as the only means of changing the pH of the water to a desired level. In other embodiments, ash is used along with one or more additional pH changing procedures.
备选地,或者连同使用pH提高剂(如上述的),水(例如,含碱土金属离子的水)源的pH值,通过电解水,使用电解或电化学流程,可以被提高到期望水平。所考虑的电化学流程包括但不局限于上述的那些以及以下文献中所述的那些:美国临时专利申请61/081,299,申请日2008年7月16日,和美国临时专利申请61/091,729,申请日2008年8月25日,其每个全盘引入本文作为参考。还考虑的是以下文献中描述电解方法:美国专利申请(公开号)2006/0185985,2006年8月24日出版,美国专利申请(公开号)2008/0248350,2008年10月9日出版,以及国际专利申请公开号WO 2008/018928,2008年2月14日出版,其每个全盘引入本文作为参考。Alternatively, or in conjunction with the use of a pH raising agent (as described above), the pH of a source of water (eg, alkaline earth metal ion-containing water) can be raised to a desired level by electrolysis of water, using electrolytic or electrochemical processes. Contemplated electrochemical processes include, but are not limited to, those described above as well as those described in U.S. Provisional Patent Application 61/081,299, filed July 16, 2008, and U.S. Provisional Patent Application 61/091,729, application dated August 25, 2008, each of which is incorporated herein by reference in its entirety. Also contemplated are the electrolytic processes described in U.S. Patent Application (Publication No.) 2006/0185985, published August 24, 2006, U.S. Patent Application (Publication No.) 2008/0248350, published October 9, 2008, and International Patent Application Publication No. WO 2008/018928, published February 14, 2008, each of which is incorporated herein by reference in its entirety.
其中期望的话,除了pH提高剂以外的添加剂也可被引入水中以便影响产生的沉淀物的性质。因而,方法的某些实施方案包括在水经受沉淀条件之前或期间在水中提供添加剂。某些碳酸钙多晶型物可能受惠于痕量的某些添加剂。例如,球霰石,一种高度不稳定的CaCO3的多晶型物,其以多种不同的形态沉淀并且迅速转化为方解石,可以通过在碳酸钙过饱和溶液中包括痕量的氯化镧形式的镧而以非常高的收率获得。除镧以外其他的所考虑的添加剂包括但不局限于过渡金属等。例如,添加亚铁或三价铁已知有利于形成无序的白云石(原白云石),其中否则的话它将不会形成。Where desired, additives other than the pH raising agent may also be introduced into the water in order to influence the properties of the precipitate produced. Thus, certain embodiments of the method include providing the additive in the water before or during the time the water is subjected to the precipitation conditions. Certain calcium carbonate polymorphs may benefit from trace amounts of certain additives. For example, vaterite, a highly unstable polymorph of CaCO3 that precipitates in a number of different forms and rapidly converts to calcite, can be formed by including trace amounts of lanthanum chloride in calcium carbonate supersaturated solutions Lanthanum was obtained in very high yields. Contemplated additives other than lanthanum include, but are not limited to, transition metals and the like. For example, addition of ferrous or ferric iron is known to favor the formation of disordered dolomite (protodolomite) where it would not otherwise form.
沉淀物的性质还可能受合适的主要离子比的选择的影响。主要离子比还具有多晶型物形成的很大的影响。例如,随着在水中镁∶钙比增加,霰石变成碳酸钙的有利的多晶型物,相比于低-镁方解石来说。在低镁∶钙比下,低-镁方解石是优选的多晶型物。The properties of the precipitate may also be influenced by the choice of an appropriate major ion ratio. The major ion ratio also has a strong influence on polymorph formation. For example, as the magnesium:calcium ratio in water increases, aragonite becomes a favorable polymorph of calcium carbonate compared to low-magnesium calcite. At low magnesium: calcium ratios, low-magnesium calcite is the preferred polymorph.
沉淀速度也对化合物相形成具有大的影响。最快速的沉淀可以通过用期望的相接晶溶液来实现。没有接晶,通过快速增加海水的pH值,这导致更多的无定形组分,可以获得快速的沉淀。当二氧化硅存在时,反应速率越快,越多的二氧化硅与碳酸盐沉淀物结合。pH越高,沉淀越快,沉淀物越是无定形的。Precipitation velocity also has a large influence on compound phase formation. The fastest precipitation can be achieved by using the desired crystallization solution. Without crystallization, rapid precipitation can be obtained by rapidly increasing the pH of seawater, which results in a more amorphous component. When silica is present, the faster the reaction rate, the more silica is bound to the carbonate precipitate. The higher the pH, the faster the precipitation and the more amorphous the precipitate.
因此,用于从水产生期望的沉淀物的一组沉淀条件包括,在某些实施方案中,水的温度,和pH,和在有些情况下在水中添加剂和离子物质的浓度。沉淀条件也可包括各种因素如混合速率,搅拌形式如超声,晶种、催化剂、膜或底物的存在。在一些实施方案中,沉淀条件包括过饱和条件,温度,pH,和/或浓度梯度,或者循环或变化这些参数中的任一种。用于制备本发明的碳酸盐化合物沉淀物的流程可以是间歇或连续流程。应当理解的是沉淀条件可以是不同的以在相比于间歇系统的连续流动系统中产生给定的沉淀物。Thus, a set of precipitation conditions for producing the desired precipitate from water includes, in certain embodiments, the temperature of the water, and the pH, and in some cases the concentration of additives and ionic species in the water. Precipitation conditions may also include factors such as rate of mixing, form of agitation such as sonication, presence of seeds, catalysts, membranes or substrates. In some embodiments, precipitation conditions include supersaturation conditions, temperature, pH, and/or concentration gradients, or cycling or varying any of these parameters. The process used to prepare the carbonate compound precipitate of the present invention may be a batch or continuous process. It should be understood that precipitation conditions may be different to produce a given precipitate in a continuous flow system as compared to a batch system.
在某些实施方案中,水(例如,含碱土金属离子的水)和CO2之间的接触可以使用任何适宜的流程来实现(例如喷枪,分段流动管式反应器)以便控制沉淀物颗粒的尺寸范围。在所要求保护的发明的方法和系统中,一种或多种添加剂可添加到含金属离子的水源,例如,絮凝剂,分散剂,表面活性剂,防垢剂,晶体生长阻滞剂,埋存剂等,以便控制沉淀物颗粒的尺寸范围。In certain embodiments, the contact between water (e.g., alkaline earth metal ion-containing water) and CO can be achieved using any suitable process (e.g., spray gun, staged flow tube reactor) to control sediment particles size range. In the method and system of the claimed invention, one or more additives may be added to the water source containing metal ions, for example, flocculants, dispersants, surfactants, antiscalants, crystal growth retardants, buried Preservatives, etc., in order to control the size range of sediment particles.
使用任何适宜途径,可以提高水的pH值。如本文中更详细描述的所考虑的途径包括但不局限于:使用pH提高剂,电化学法,使用天然碱性水,如来自碱湖的,等等。在有些情况下,可以使用pH-提高剂,其中这样的试剂的实例包括氧化物(如氧化钙,氧化镁,等等),氢氧化物(如氢氧化钠,氢氧化钾和氢氧化镁),碳酸盐(如碳酸钠)等。被添加到水的pH提高剂的量将取决于试剂的特定的性质和被改变的水的体积,并且将足以提高水的pH值至期望值。The pH of the water may be raised using any suitable means. Contemplated approaches as described in more detail herein include, but are not limited to: use of pH raising agents, electrochemical methods, use of naturally alkaline water, such as from soda lakes, and the like. In some cases, a pH-raising agent may be used, where examples of such agents include oxides (such as calcium oxide, magnesium oxide, etc.), hydroxides (such as sodium hydroxide, potassium hydroxide, and magnesium hydroxide) , carbonates (such as sodium carbonate), etc. The amount of pH raising agent added to the water will depend on the particular nature of the agent and the volume of water being altered, and will be sufficient to raise the pH of the water to the desired value.
以下描述了在本发明的实施方案中可使用的电化学法和系统。该方法和系统使用一种或多种离子选择膜(用于产生氢氧化物的低电压系统)。这些方法和系统进一步描述于以下文献中:国际专利申请PCT/US08/88242,申请日2008年12月23日,题目″Low-EnergyElectrochemical Hydroxide System and Method,″和国际专利申请PCT/US08/88246,申请日2008年12月23日,题目″Low-EnergyElectrochemical Proton Transfer System and Method,″,其每个全盘引入本文作为参考。Electrochemical methods and systems that may be used in embodiments of the invention are described below. The method and system use one or more ion selective membranes (low voltage systems for hydroxide generation). These methods and systems are further described in the following documents: International Patent Application PCT/US08/88242, filed December 23, 2008, titled "Low-Energy Electrochemical Hydroxide System and Method," and International Patent Application PCT/US08/88246, Application date on December 23, 2008, title "Low-Energy Electrochemical Proton Transfer System and Method," each of which is fully incorporated herein as a reference.
生产氢氧化物的低电压系统Low voltage system for the production of hydroxides
用于从水溶液除去质子/产生氢氧化物的第二组方法和系统涉及用电化学方法制备离子溶液的低能量方法,其利用率电化学电池中的离子交换膜。在一个实施方案中,该系统包括电化学系统,其中离子交换膜分隔了第一电解质和第二电解质,第一电解质接触阳极并且第二电解质接触阴极。在该系统中,在阳极和阴极两端施加电压时,在阴极形成氢氧离子,在阳极没有形成气体。A second group of methods and systems for proton removal/hydroxide generation from aqueous solutions involves low energy methods of electrochemically preparing ionic solutions utilizing ion exchange membranes in electrochemical cells. In one embodiment, the system includes an electrochemical system wherein the ion exchange membrane separates a first electrolyte and a second electrolyte, the first electrolyte contacts the anode and the second electrolyte contacts the cathode. In this system, when a voltage is applied across the anode and cathode, hydroxide ions are formed at the cathode and no gas is formed at the anode.
在另一实施方案中,该系统包括电化学系统,其包括第一电解电池,所述第一电解电池包括接触第一电解质的阳极和分隔第一电解质和第三电解质的阴离子交换膜;和第二电解电池包括接触阴极的第二电解质和分隔第一电解质和第三电解质的阳离子交换膜;其中在阳极和阴极两端施加电压时,在阴极形成氢氧离子,在阳极没有形成气体。In another embodiment, the system comprises an electrochemical system comprising a first electrolysis cell comprising an anode in contact with a first electrolyte and an anion exchange membrane separating the first electrolyte and a third electrolyte; and Two electrolytic cells comprising a second electrolyte in contact with the cathode and a cation exchange membrane separating the first and third electrolytes; wherein hydroxide ions are formed at the cathode and no gas is formed at the anode when a voltage is applied across the anode and cathode.
在一个实施方案中,该方法包括将离子交换膜放置在第一电解质和第二电解质之间,第一电解质接触阳极和第二电解质接触阴极;和通过在阳极和阴极两端施加电压使离子跨离子交换膜迁移而在阴极形成氢氧离子,而在阳极没有形成气体。In one embodiment, the method includes placing an ion exchange membrane between a first electrolyte and a second electrolyte, the first electrolyte contacting the anode and the second electrolyte contacting the cathode; The ion exchange membrane migrates to form hydroxide ions at the cathode without gas formation at the anode.
在另一实施方案中,该方法包括将第三电解质放置在阴离子交换膜和阳离子交换膜之间;将第一电解质放置在阴离子交换膜和阳极之间;和将第二电解质放置在阳离子交换膜和阴极之间;和通过将电压施加到阳极和阴极使离子跨越阳离子交换膜和阴离子交换膜迁移而在阴极形成氢氧离子,而在阳极没有形成气体。In another embodiment, the method includes placing a third electrolyte between the anion exchange membrane and the cation exchange membrane; placing the first electrolyte between the anion exchange membrane and the anode; and placing the second electrolyte between the cation exchange membrane and the cathode; and hydroxide ions are formed at the cathode by applying a voltage to the anode and cathode to cause ions to migrate across the cation exchange membrane and the anion exchange membrane without gas formation at the anode.
通过所述方法和系统,以低电压电化学方式,将来自一个溶液的离子物质转移到另一溶液,由此提供了用于各种应用的阴离子溶液,包括制备氢氧化钠溶液以备埋存二氧化碳,如本文中所述。在一个实施方案中,包括OH-的溶液从盐水中获得并且通过从包括碱土金属离子的盐溶液中沉淀钙和镁的碳酸盐和碳酸氢盐来用于埋存CO2。By the described methods and systems, low-voltage electrochemical transfer of ionic species from one solution to another provides anionic solutions for various applications, including the preparation of sodium hydroxide solutions for storage Carbon dioxide, as described herein. In one embodiment, a solution comprising OH- is obtained from brine and used for CO2 sequestration by precipitating calcium and magnesium carbonates and bicarbonates from a salt solution comprising alkaline earth metal ions.
在各种实施方案中的方法和系统涉及低电压电化学系统和方法,其用于在水溶液中产生氢氧化钠溶液,利用了一种或多种离子交换膜,其中,在阳极没有形成气体,并且其中在阴极形成氢氧离子。因此,在一些实施方案中,在电化学过程中形成氢氧离子,而没有形成氧气或氯气。在一些实施方案中,在电化学过程中形成氢氧离子,其中在阳极和阴极两端施加的电压小于2.8V,2.7V,2.5V,2.4V,2.3V,2.2V,2.1V,2.0V,1.9V,1.8V,1.7V,1.6V,1.5V,1.4V,1.3V,1.2V,1.1V,1.0V,0.9V,0.8V,0.7V,0.6V,0.5V,0.4V,0.3V,0.2V,或0.1V。在各种实施方案中,利用离子膜来分隔与阳极接触的盐水和与阴极接触的溶液如氯化钠。在阴极和阳极两端施加低电压时,溶液例如氢氧化钠溶液在阴极周围的溶液中形成;同时,包括氢氯酸的酸化溶液在阳极周围的溶液中形成。在各种实施方案中,气体如氯气或氧气没有在阳极形成。Methods and systems in various embodiments relate to low voltage electrochemical systems and methods for producing sodium hydroxide solutions in aqueous solutions utilizing one or more ion exchange membranes wherein no gas is formed at the anode, And wherein hydroxide ions are formed at the cathode. Thus, in some embodiments, hydroxide ions are formed during the electrochemical process, but no oxygen or chlorine gas is formed. In some embodiments, hydroxide ions are formed in an electrochemical process wherein the voltage applied across the anode and cathode is less than 2.8V, 2.7V, 2.5V, 2.4V, 2.3V, 2.2V, 2.1V, 2.0V , 1.9V, 1.8V, 1.7V, 1.6V, 1.5V, 1.4V, 1.3V, 1.2V, 1.1V, 1.0V, 0.9V, 0.8V, 0.7V, 0.6V, 0.5V, 0.4V, 0.3 V, 0.2V, or 0.1V. In various embodiments, an ionic membrane is utilized to separate the brine in contact with the anode from the solution, such as sodium chloride, in contact with the cathode. When a low voltage is applied across the cathode and anode, a solution such as sodium hydroxide solution is formed in the solution surrounding the cathode; at the same time, an acidified solution including hydrochloric acid is formed in the solution surrounding the anode. In various embodiments, gases such as chlorine or oxygen are not formed at the anode.
在各种实施方案中,氢氧化钠溶液可用于埋存CO2,如本文中所述,并且酸性溶液可用于溶解含钙和镁的无机物从而提供用于埋存CO2的钙和镁离子,同样如本文中所述。In various embodiments, a sodium hydroxide solution can be used to sequester CO2 , as described herein, and an acidic solution can be used to dissolve calcium and magnesium containing inorganics to provide calcium and magnesium ions for CO2 sequestration , also as described in this article.
转向图8-10,在各种实施方案中,系统可适应于间歇和连续过程,如本文中所述。参考图8-9,在一个实施方案中,该系统包括电化学电池,其中放置离子交换膜(802,824)来分隔第一电解质(804)和第二电解质(806),第一电解质接触阳极(808)并且第二电解质接触阴极(810)。如图8中举例说明的,使用了阴离子交换膜(802);在图9中,使用了阳离子交换膜(824)。Turning to Figures 8-10, in various embodiments, the system can be adapted for both batch and continuous processes, as described herein. Referring to Figures 8-9, in one embodiment, the system includes an electrochemical cell in which an ion exchange membrane (802, 824) is positioned to separate a first electrolyte (804) from a second electrolyte (806), the first electrolyte contacting the anode (808) and the second electrolyte contacts the cathode (810). As illustrated in Figure 8, an anion exchange membrane (802) is used; in Figure 9, a cation exchange membrane (824) is used.
在各种实施方案中,如图8和9中举例说明的,第一电解质(804)包括盐水溶液,后者包括海水、淡水、盐水或微咸水等;和第二电解质包括基本上氯化钠的溶液。在各种实施方案中,第二(806)电解质可以包括海水或氯化钠的浓缩溶液。在各种实施方案中,阴离子交换膜(802)和阳离子交换膜(824)包括常规的离子交换膜,其适用于在水溶液中至多100℃的工作温度下的酸性和/或碱性溶液中。如图8和9中举例说明的,第一和第二电解质接触阳极和阴极从而完成包括电压或电流调节器(812)的电路。根据需要,在系统中电流/电压调节器适合于增减阴极和阳极两端的电流或电压。In various embodiments, as illustrated in Figures 8 and 9, the first electrolyte (804) includes a brine solution, which includes seawater, freshwater, brine, or brackish water, etc.; and the second electrolyte includes substantially chlorinated sodium solution. In various embodiments, the second (806) electrolyte may include seawater or a concentrated solution of sodium chloride. In various embodiments, the anion exchange membrane (802) and cation exchange membrane (824) comprise conventional ion exchange membranes suitable for use in acidic and/or basic solutions at operating temperatures of up to 100°C in aqueous solution. As illustrated in Figures 8 and 9, the first and second electrolytes contact the anode and cathode to complete an electrical circuit including a voltage or current regulator (812). Current/voltage regulators are adapted to increase or decrease the current or voltage across the cathode and anode in the system as required.
参考图8和9,在各种实施方案中,电化学电池包括第一电解质入口孔(814),其可适应于将第一电解质(804)输入系统并且与阳极(808)接触。同样,电池包括第二电解质入口孔(816),其用于将第二电解质(806)输入系统并且与阴极(810)接触。另外,电池包括用于从电池排出第一电解质的出口孔(818),和用于从电池排出第二电解质的出口孔(820)。如本领域技术人员会理解的,入口和出口孔可适应于各种流动流程,包括间歇流动、半间歇流动或连续流动。在备选的实施方案中,该系统包括用于引导气体至阳极的管(822);在各种实施方案中,气体包括在阴极(810)处形成的氢气。Referring to Figures 8 and 9, in various embodiments, the electrochemical cell includes a first electrolyte inlet aperture (814), which can be adapted to feed the first electrolyte (804) into the system and contact the anode (808). Likewise, the cell includes a second electrolyte inlet port (816) for feeding the second electrolyte (806) into the system and in contact with the cathode (810). In addition, the cell includes an outlet aperture (818) for draining the first electrolyte from the cell, and an outlet aperture (820) for draining the second electrolyte from the cell. As will be appreciated by those skilled in the art, the inlet and outlet holes can accommodate various flow regimes, including intermittent, semi-intermittent, or continuous flow. In alternative embodiments, the system includes a tube (822) for directing gas to the anode; in various embodiments, the gas includes hydrogen formed at the cathode (810).
参考图8,其中利用了阴离子隔膜(802),当在阴极(810)和阳极(808)两端施加低电压时,在阴极(810)形成氢氧离子,并且在阳极(808)气体没有形成。进一步,其中第二电解质(806)包括氯化钠,氯离子从第二电解质(806)通过阴离子交换膜(802)迁移到第一电解质(804)中;在阳极(808)形成质子;和在阴极(810)形成氢气。如上所述,气体例如,氧气或氯气没有在阳极(808)形成。Referring to Figure 8, where an anion membrane (802) is utilized, when a low voltage is applied across the cathode (810) and anode (808), hydroxide ions are formed at the cathode (810) and no gas is formed at the anode (808) . Further, wherein the second electrolyte (806) includes sodium chloride, chloride ions migrate from the second electrolyte (806) to the first electrolyte (804) through the anion exchange membrane (802); form protons at the anode (808); and The cathode (810) forms hydrogen gas. As noted above, gases such as oxygen or chlorine are not formed at the anode (808).
参考图9,其中利用了阳离子隔膜(824),当在阴极(810)和阳极(808)两端施加低电压时,在阴极(810)形成氢氧离子,并且在阳极(808)气体没有形成。在各种实施方案中,阳离子交换膜(824)包括常规的离子交换膜,其适用于在水溶液中至多100℃的工作温度下的酸性和碱性溶液。如图9中举例说明的,第一和第二电解质接触阳极和阴极从而完成包括电压和/或电流调节器(812)的电路。根据需要,在系统中电压/电流调节器适合于增减阴极和阳极两端的电流或电压。在如图9中举例说明的系统中,其中第二电解质(804)包括氯化钠,钠离子从第一电解质(804)通过阳离子交换膜(824)迁移到第二电解质(806)中;在阳极(808)形成质子;和在阴极(810)形成氢气。如上所述,气体例如,氧气或氯气没有在阳极(808)形成。Referring to Figure 9, where a cationic membrane (824) is utilized, when a low voltage is applied across the cathode (810) and anode (808), hydroxide ions are formed at the cathode (810) and no gas is formed at the anode (808) . In various embodiments, the cation exchange membrane (824) comprises a conventional ion exchange membrane suitable for acidic and basic solutions in aqueous solution at operating temperatures up to 100°C. As illustrated in Figure 9, the first and second electrolytes contact the anode and cathode to complete the electrical circuit including voltage and/or current regulators (812). A voltage/current regulator is adapted to increase or decrease the current or voltage across the cathode and anode in the system as required. In a system as illustrated in Figure 9, wherein the second electrolyte (804) comprises sodium chloride, sodium ions migrate from the first electrolyte (804) through the cation exchange membrane (824) to the second electrolyte (806); in Protons are formed at the anode (808); and hydrogen is formed at the cathode (810). As noted above, gases such as oxygen or chlorine are not formed at the anode (808).
如本领域技术人员可以理解的,并且参考图8,在第二电解质(806)中,当氢氧离子从阳极(810)并且进入第二电解质(806),同时氯离子从第二电解质迁移出的时候,在第二电解质(806)中将形成氢氧化钠水溶液。因此,取决于系统上所施加的电压和通过系统的第二电解质(806)的流速,调节第二电解质的pH值。在一个实施方案中,当在阳极和阴极两端施加0.1V或更小、0.2V或更小、0.3V或更小、0.4V或更小、0.5V或更小、0.6V或更小、0.7V或更小、0.8V或更小、0.9V或更小、1.0V或更小、1.1V或更小、1.2V或更小、1.3V或更小、01.4V或更小、1.5V或更小、1.6V或更小、1.7V或更小、1.8V或更小、1.9V或更小或2.0V或更小的电势时,第二电解质溶液的pH值增加;在另一实施方案中,当在阳极和阴极两端施加0.1V至2.0V的电压时,第二电解质的pH值增加;在又一实施方案中,当在阳极和阴极两端施加0.1V至1V的电压时,第二电解质溶液的pH值增加。借助于电极两端的0.1V至0.8V;0.1V至0.7V;0.1V至0.6V;0.1V至0.5V;0.1V至0.4V;和0.1V至0.3V的电压,可实现相似的结果。根据该系统获得的示范性的结果总结在表2中。As can be understood by those skilled in the art, and with reference to FIG. , an aqueous sodium hydroxide solution will form in the second electrolyte (806). Thus, depending on the voltage applied to the system and the flow rate of the second electrolyte (806) through the system, the pH of the second electrolyte is adjusted. In one embodiment, when 0.1 V or less, 0.2 V or less, 0.3 V or less, 0.4 V or less, 0.5 V or less, 0.6 V or less, 0.7V or less, 0.8V or less, 0.9V or less, 1.0V or less, 1.1V or less, 1.2V or less, 1.3V or less, 01.4V or less, 1.5V or less, 1.6V or less, 1.7V or less, 1.8V or less, 1.9V or less, or 2.0V or less potential, the pH of the second electrolyte solution increases; in another implementation In one embodiment, when a voltage of 0.1V to 2.0V is applied across the anode and cathode, the pH of the second electrolyte increases; in yet another embodiment, when a voltage of 0.1V to 1V is applied across the anode and cathode , the pH of the second electrolyte solution increases. Similar results can be achieved with voltages of 0.1 V to 0.8 V; 0.1 V to 0.7 V; 0.1 V to 0.6 V; 0.1 V to 0.5 V; 0.1 V to 0.4 V; and 0.1 V to 0.3 V across the electrodes. Exemplary results obtained with this system are summarized in Table 2.
表2.低能量电化学方法和系统。Table 2. Low energy electrochemical methods and systems.
在本实例中,阳极和阴极都包括铂,第一和第二电解质包括氯化钠溶液。In this example, both the anode and cathode included platinum, and the first and second electrolytes included sodium chloride solution.
同样,参考图9,在第二电解质(806)中,当氢氧离子从阳极(810)进入溶液,同时钠离子从第一电解质迁移到第二电解质的时候,在第二电解质(806)中逐渐地将形成氢氧化钠水溶液。取决于系统上所施加的电压和通过系统的第二电解质的流速,将调节溶液的pH值。在一个实施方案中,当在阳极和阴极两端施加0.1V或更小、0.2V或更小、0.3V或更小、0.4V或更小、0.5V或更小、0.6V或更小、0.7V或更小、0.8V或更小、0.9V或更小、1.0V或更小、1.1V或更小、1.2V或更小、1.3V或更小、1.4V或更小、1.5V或更小、1.6V或更小、1.7V或更小、1.8V或更小、1.9V或更小或2.0V或更小的电压时,第二电解质溶液的pH值增加;在另一实施方案中,当在阳极和阴极两端施加0.1V至2.0V的电压时,第二电解质的pH值增加;在又一实施方案中,当在阳极和阴极两端施加0.1V至1V的电压时,第二电解质溶液的pH值增加。借助于电极两端的0.1V至0.8V;0.1V至0.7V;0.1V至0.6V;0.1V至0.5V;0.1V至0.4V;和0.1V至0.3V的电压,可实现相似的结果。在一个实施方案中,在阳极和阴极两端施加0.6V或更小的电压;在另一实施方案中,在阳极和阴极两端施加0.1V至0.6V或更小的电压;在又一实施方案中,在阳极和阴极两端施加0.1至1V或更小的电压。Likewise, referring to FIG. 9, in the second electrolyte (806), when hydroxide ions enter the solution from the anode (810) while sodium ions migrate from the first electrolyte to the second electrolyte, in the second electrolyte (806) Gradually an aqueous solution of sodium hydroxide will form. Depending on the voltage applied across the system and the flow rate of the second electrolyte through the system, the pH of the solution will be adjusted. In one embodiment, when 0.1 V or less, 0.2 V or less, 0.3 V or less, 0.4 V or less, 0.5 V or less, 0.6 V or less, 0.7V or less, 0.8V or less, 0.9V or less, 1.0V or less, 1.1V or less, 1.2V or less, 1.3V or less, 1.4V or less, 1.5V or less, 1.6V or less, 1.7V or less, 1.8V or less, 1.9V or less, or 2.0V or less voltage, the pH of the second electrolyte solution increases; in another implementation In one embodiment, when a voltage of 0.1V to 2.0V is applied across the anode and cathode, the pH of the second electrolyte increases; in yet another embodiment, when a voltage of 0.1V to 1V is applied across the anode and cathode , the pH of the second electrolyte solution increases. Similar results can be achieved with voltages of 0.1 V to 0.8 V; 0.1 V to 0.7 V; 0.1 V to 0.6 V; 0.1 V to 0.5 V; 0.1 V to 0.4 V; and 0.1 V to 0.3 V across the electrodes. In one embodiment, a voltage of 0.6V or less is applied across the anode and cathode; in another embodiment, a voltage of 0.1V to 0.6V or less is applied across the anode and cathode; in yet another embodiment In the scheme, a voltage of 0.1 to 1 V or less is applied across the anode and cathode.
在各种实施方案中并且参考图8-10,在阴极(810)形成的氢气被通到阳极(808),其中,在不受任何理论的束缚下,据信气体吸附和/或吸收到阳极中并且随后在阳极形成质子。因此,如可以理解的是,在质子在阳极形成并且例如如图8中的那样氯离子迁移到第一电解质(804)或者例如如图10中的那样,钠离子从第一电解质迁移出来的情况下,包括例如氢氯酸的酸性溶液在第一电解质(804)中获得。In various embodiments and with reference to Figures 8-10, hydrogen gas formed at the cathode (810) is passed to the anode (808), where, without being bound by any theory, it is believed that the gas adsorbs and/or absorbs to the anode and subsequently form protons at the anode. Thus, as can be appreciated, where protons are formed at the anode and chloride ions migrate to the first electrolyte (804) such as in FIG. 8 or sodium ions migrate out of the first electrolyte such as in FIG. Next, an acidic solution comprising eg hydrochloric acid is obtained in the first electrolyte (804).
在另一实施方案中,如图10中举例说明的,系统包括电化学电池,其包括接触第一电解质(804)的阳极(808)和分隔第一电解质和第三电解质(830)的阴离子交换膜(802);和第二电解电池,其包括接触阴极(880)的第二电解质(806)和分隔第一电解质和第三电解质的阳离子交换膜(824),其中在阳极和阴极两端施加电压时,在阴极形成氢离子而在阳极形成气体。如同图8和9的系统一样,图10的系统可适应于间歇和连续过程。In another embodiment, as illustrated in Figure 10, the system includes an electrochemical cell comprising an anode (808) in contact with a first electrolyte (804) and an anion exchange (830) separating the first and third electrolytes (830). a membrane (802); and a second electrolytic cell comprising a second electrolyte (806) contacting the cathode (880) and a cation exchange membrane (824) separating the first and third electrolytes, wherein applied across the anode and cathode When the voltage is high, hydrogen ions are formed at the cathode and gas is formed at the anode. Like the systems of Figures 8 and 9, the system of Figure 10 is adaptable to both batch and continuous processes.
在各种实施方案中,如图10中举例说明的,第一电解质(804)和第二电解质(806)包括盐水溶液,后者包括海水、淡水、盐水或微咸水等;和第二电解质包括基本上氯化钠的溶液。在各种实施方案中,第一(804)和第二(806)电解质可以包括海水。在图10中举例说明的实施方案中,第三电解质(830)包括基本上氯化钠溶液。In various embodiments, as illustrated in FIG. 10, the first electrolyte (804) and the second electrolyte (806) include a saline solution, the latter including seawater, fresh water, salt water, or brackish water, etc.; and the second electrolyte A solution consisting essentially of sodium chloride. In various embodiments, the first (804) and second (806) electrolytes can include seawater. In the embodiment illustrated in Figure 10, the third electrolyte (830) comprises a substantially sodium chloride solution.
在各种实施方案中,阴离子交换膜(802)包括任何合适的阴离子交换膜,其适用于在水溶液中至多100℃的工作温度下的酸性和碱性溶液。同样,阳离子交换膜(824)包括任何合适的阳离子交换膜,其适用于在水溶液中至多100℃的工作温度下的酸性和碱性溶液。In various embodiments, the anion exchange membrane (802) comprises any suitable anion exchange membrane suitable for use in acidic and basic solutions in aqueous solution at operating temperatures up to 100°C. Likewise, the cation exchange membrane (824) includes any suitable cation exchange membrane suitable for use in acidic and basic solutions in aqueous solution at operating temperatures up to 100°C.
如图10中举例说明的,在各种实施方案中,第一电解质(804)接触阳极(808),第二电解质(806)接触阴极(810)。接触阴离子和阳离子交换膜的第三电解质(830)完成包括电压或电流调节器(812)的电路。根据需要,在系统中电流/电压调节器适合于增减阴极和阳极两端的电流或电压。As illustrated in Figure 10, in various embodiments, the first electrolyte (804) contacts the anode (808) and the second electrolyte (806) contacts the cathode (810). A third electrolyte (830) in contact with the anion and cation exchange membranes completes the circuit including a voltage or current regulator (812). Current/voltage regulators are adapted to increase or decrease the current or voltage across the cathode and anode in the system as required.
参考图10,在各种实施方案中,电化学电池包括可适应于将第一电解质804输入系统的第一电解质入口孔(814);用于将第二电解质(806)输入系统的第二电解质入口孔(816);和用于将第三电解质输入系统的第三入口孔(826)。另外,电池包括用于排出第一电解质的出口孔(818);用于排出第二电解质的出口孔(820);和用于排出第三电解质的出口孔(828)。如本领域技术人员会理解的,入口和出口孔可适应于各种流动流程,包括间歇流动、半间歇流动或连续流动。在备选地实施方案中,该系统包括用于引导气体至阳极的管(822);在各种实施方案中,气体是在阴极(810)处形成的氢气。Referring to FIG. 10, in various embodiments, the electrochemical cell includes a first electrolyte inlet port (814) adaptable for introducing a
参考图10,当在阴极(810)和阳极(808)两端施加低电压时,在阴极(810)形成氢氧离子,并且在阳极(808)气体没有形成。进一步,在第三电解质(830)包括氯化钠的情况下,氯离子从第三电解质(830)通过阴离子交换膜(802)迁移到第一电解质(804)中;钠离子从第三电解质(830)迁移到第二电解质(806);质子在阳极形成;和氢气在阴极形成。如上所述,气体例如,氧气或氯气没有在阳极(808)形成。Referring to Figure 10, when a low voltage is applied across the cathode (810) and anode (808), hydroxide ions are formed at the cathode (810), and no gas is formed at the anode (808). Further, when the third electrolyte (830) includes sodium chloride, chloride ions migrate from the third electrolyte (830) to the first electrolyte (804) through the anion exchange membrane (802); sodium ions migrate from the third electrolyte (802) to the first electrolyte (804); 830) migrates to the second electrolyte (806); protons are formed at the anode; and hydrogen is formed at the cathode. As noted above, gases such as oxygen or chlorine are not formed at the anode (808).
如本领域技术人员可以理解的,并且参考图10,在第二电解质(806)中,当氢氧离子从阴极(810)进入溶液,同时氯离子从第三电解质迁移出的时候,逐渐地在第二电解质(806)中将形成氢氧化钠水溶液。取决于系统上所施加的电压和通过系统的第二电解质的流速,将调节溶液的pH值。在一个实施方案中,当在阳极和阴极两端施加0.1V或更小、0.2V或更小、0.3V或更小、0.4V或更小、0.5V或更小、0.6V或更小、0.7V或更小、0.8V或更小、0.9V或更小、1.0V或更小、1.1V或更小、1.2V或更小、1.3V或更小、01.4V或更小、1.5V或更小、1.6V或更小、1.7V或更小、1.8V或更小、1.9V或更小或2.0V或更小或更小的电压时,第二电解质溶液的pH值增加;在另一实施方案中,当在阳极和阴极两端施加0.1V至2.0V的电压时,第二电解质的pH值增加;在又一实施方案中,当在阳极和阴极两端施加0.1V至1V的电压时,第二电解质溶液的pH值增加。借助于电极两端的0.1V至0.8V;0.1V至0.7V;0.1V至0.6V;0.1V至0.5V;0.1V至0.4V;和0.1V至0.3V的电压,可实现相似的结果。在一个实施方案中,在阳极和阴极两端施加0.6V或更小的电压;在另一实施方案中,在阳极和阴极两端施加0.1V至0.6V或更小的电压;在又一实施方案中,在阳极和阴极两端施加0.1V至1V或更小的电压。As can be understood by those skilled in the art, and with reference to FIG. 10, in the second electrolyte (806), when hydroxide ions enter the solution from the cathode (810) while chloride ions migrate out of the third electrolyte, gradually in An aqueous sodium hydroxide solution will form in the second electrolyte (806). Depending on the voltage applied across the system and the flow rate of the second electrolyte through the system, the pH of the solution will be adjusted. In one embodiment, when 0.1 V or less, 0.2 V or less, 0.3 V or less, 0.4 V or less, 0.5 V or less, 0.6 V or less, 0.7V or less, 0.8V or less, 0.9V or less, 1.0V or less, 1.1V or less, 1.2V or less, 1.3V or less, 01.4V or less, 1.5V or less, 1.6V or less, 1.7V or less, 1.8V or less, 1.9V or less, or 2.0V or less or less, the pH value of the second electrolyte solution increases; In another embodiment, when a voltage of 0.1 V to 2.0 V is applied across the anode and cathode, the pH of the second electrolyte increases; in yet another embodiment, when a voltage of 0.1 V to 1 V is applied across the anode and cathode When the voltage is increased, the pH value of the second electrolyte solution increases. Similar results can be achieved with voltages of 0.1 V to 0.8 V; 0.1 V to 0.7 V; 0.1 V to 0.6 V; 0.1 V to 0.5 V; 0.1 V to 0.4 V; and 0.1 V to 0.3 V across the electrodes. In one embodiment, a voltage of 0.6V or less is applied across the anode and cathode; in another embodiment, a voltage of 0.1V to 0.6V or less is applied across the anode and cathode; in yet another embodiment In the scheme, a voltage of 0.1V to 1V or less is applied across the anode and cathode.
同样,参考图10,在第一电解质(804)中,当质子在阳极(808)形成并且进入溶液,同时氯离子从第三电解质迁移到第一电解质的时候,在第一电解质(804)中逐渐地将形成酸性溶液。取决于系统上所施加的电压和通过系统的第二电解质的流速,将调节溶液的pH值。在一个实施方案中,当在阳极和阴极两端施加0.1V或更小、0.2V或更小、0.3V或更小、0.4V或更小、0.5V或更小、0.6V或更小、0.7V或更小、0.8V或更小、0.9V或更小、1.0V或更小、1.1V或更小、1.2V或更小、1.3V或更小、1.4V或更小、1.5V或更小、1.6V或更小、1.7V或更小、1.8V或更小、1.9V或更小或2.0V或更小的电压时,第二电解质溶液的pH值增加;在另一实施方案中,当在阳极和阴极两端施加0.1V至2.0V的电压时,第二电解质的pH值增加;在又一实施方案中,当在阳极和阴极两端施加0.1V至1V的电压时,第二电解质溶液的pH值增加。借助于电极两端的0.1V至0.8V;0.1V至0.7V;0.1V至0.6V;0.1V至0.5V;0.1V至0.4V;和0.1V至0.3V的电压,可实现相似的结果。在一个实施方案中,在阳极和阴极两端施加0.6V或更小的电压;在另一实施方案中,在阳极和阴极两端施加0.1V至0.6V或更小的电压;在又一实施方案中,在阳极和阴极两端施加0.1V至1V或更小的电压,如表2中所示。Also, referring to Figure 10, in the first electrolyte (804), when protons are formed at the anode (808) and go into solution while chloride ions migrate from the third electrolyte to the first electrolyte, in the first electrolyte (804) Gradually an acidic solution will form. Depending on the voltage applied across the system and the flow rate of the second electrolyte through the system, the pH of the solution will be adjusted. In one embodiment, when 0.1 V or less, 0.2 V or less, 0.3 V or less, 0.4 V or less, 0.5 V or less, 0.6 V or less, 0.7V or less, 0.8V or less, 0.9V or less, 1.0V or less, 1.1V or less, 1.2V or less, 1.3V or less, 1.4V or less, 1.5V or less, 1.6V or less, 1.7V or less, 1.8V or less, 1.9V or less, or 2.0V or less voltage, the pH of the second electrolyte solution increases; in another implementation In one embodiment, when a voltage of 0.1V to 2.0V is applied across the anode and cathode, the pH of the second electrolyte increases; in yet another embodiment, when a voltage of 0.1V to 1V is applied across the anode and cathode , the pH of the second electrolyte solution increases. Similar results can be achieved with voltages of 0.1 V to 0.8 V; 0.1 V to 0.7 V; 0.1 V to 0.6 V; 0.1 V to 0.5 V; 0.1 V to 0.4 V; and 0.1 V to 0.3 V across the electrodes. In one embodiment, a voltage of 0.6V or less is applied across the anode and cathode; in another embodiment, a voltage of 0.1V to 0.6V or less is applied across the anode and cathode; in yet another embodiment In the scheme, a voltage of 0.1 V to 1 V or less was applied across the anode and cathode, as shown in Table 2.
如图10中举例说明的,在阴极(810)形成的氢气被通到阳极(808),其中,在不受任何理论的束缚下,据信氢气吸附和/或吸收到阳极中并且随后质子在阳极形成并且进入第一电解质(804)。此外,在如图8-10中举例说明的各种实施方案中,气体例如氧气或氯气没有在阳极(808)形成。因此,如可以理解的是,在在阳极形成质子并且氯气迁移到第一电解质的情况下,在第一电解质(804)中获得氢氯酸。As illustrated in Figure 10, the hydrogen gas formed at the cathode (810) is passed to the anode (808), where, without being bound by any theory, it is believed that the hydrogen gas is adsorbed and/or absorbed into the anode and subsequently the protons in An anode is formed and enters the first electrolyte (804). Furthermore, in various embodiments as illustrated in Figures 8-10, gases such as oxygen or chlorine are not formed at the anode (808). Thus, as can be appreciated, hydrochloric acid is obtained in the first electrolyte (804) with the formation of protons at the anode and the migration of chlorine gas to the first electrolyte.
如本领域技术人员可以理解的,并且参考图8,在第二电解质(806)中,当氢氧离子从阳极(810)并且进入第二电解质(806),同时氯离子从第二电解质迁移出的时候,在第二电解质(806)中将形成氢氧化钠水溶液。因此,取决于系统上所施加的电压和通过系统的第二电解质(806)的流速,调节第二电解质的pH值。在一个实施方案中,当在阳极和阴极两端施加0.1V或更小、0.2V或更小、0.3V或更小、0.4V或更小、0.5V或更小、0.6V或更小、0.7V或更小、0.8V或更小、0.9V或更小、1.0V或更小、1.1V或更小、1.2V或更小、1.3V或更小、1.4V或更小、1.5V或更小、1.6V或更小、1.7V或更小、1.8V或更小、1.9V或更小或2.0V或更小的电压时,第二电解质溶液的pH值增加;在另一实施方案中,当在阳极和阴极两端施加0.1V至2.0V的电压时,第二电解质的pH值增加;在又一实施方案中,当在阳极和阴极两端施加0.1V至1V的电压时,第二电解质溶液的pH值增加。借助于电极两端的0.1V至0.8V;0.1V至0.7V;0.1V至0.6V;0.1V至0.5V;0.1V至0.4V;和0.1V至0.3V的电压,可实现相似的结果。在一个实施方案中,当在阳极和阴极两端施加0.6V或更小的电压时,第二电解质溶液的pH值增加;在另一实施方案中,当在阳极和阴极两端施加0.1V至0.6V的电压时,第二电解质的pH值增加;在又一实施方案中,当在阳极和阴极两端施加0.1V至1V或更小的电压时,第二电解质溶液的pH值增加。As can be understood by those skilled in the art, and with reference to FIG. , an aqueous sodium hydroxide solution will form in the second electrolyte (806). Thus, depending on the voltage applied to the system and the flow rate of the second electrolyte (806) through the system, the pH of the second electrolyte is adjusted. In one embodiment, when 0.1 V or less, 0.2 V or less, 0.3 V or less, 0.4 V or less, 0.5 V or less, 0.6 V or less, 0.7V or less, 0.8V or less, 0.9V or less, 1.0V or less, 1.1V or less, 1.2V or less, 1.3V or less, 1.4V or less, 1.5V or less, 1.6V or less, 1.7V or less, 1.8V or less, 1.9V or less, or 2.0V or less voltage, the pH of the second electrolyte solution increases; in another implementation In one embodiment, when a voltage of 0.1V to 2.0V is applied across the anode and cathode, the pH of the second electrolyte increases; in yet another embodiment, when a voltage of 0.1V to 1V is applied across the anode and cathode , the pH of the second electrolyte solution increases. Similar results can be achieved with voltages of 0.1 V to 0.8 V; 0.1 V to 0.7 V; 0.1 V to 0.6 V; 0.1 V to 0.5 V; 0.1 V to 0.4 V; and 0.1 V to 0.3 V across the electrodes. In one embodiment, when a voltage of 0.6 V or less is applied across the anode and cathode, the pH of the second electrolyte solution increases; in another embodiment, when a voltage of 0.1 V to At a voltage of 0.6 V, the pH of the second electrolyte increases; in yet another embodiment, the pH of the second electrolyte solution increases when a voltage of 0.1 V to 1 V or less is applied across the anode and cathode.
任选地,通过将气体鼓泡到第二电解质溶液806中,如上所述,包括CO2的气体被溶解到第二电解质溶液中。在任选的步骤中,所得的第二电解质溶液用于沉淀碳酸盐和/或碳酸氢盐化合物如碳酸钙或碳酸镁和/或其碳酸氢盐,如本文中所述。沉淀的碳酸盐化合物可被用作水泥和建筑材料,如本文中所述。Optionally, by bubbling the gas into the
在另一任选的步骤中,利用酸化的第一电解质溶液804来溶解富含钙和/或镁的矿物,如镁铁矿物,包括蛇纹石或橄榄石,用作沉淀碳酸盐和碳酸氢盐的溶液,如本文中所述。在各种实施方案中,所得的溶液可被用作第二电解质溶液。同样地,在其中在第一电解质804中产生氢氯酸的实施方案中,代替酸化的第二电解质溶液,或者除酸化的第二电解质溶液以外,可以使用氢氯酸。In another optional step, the acidified
上述实施方案产生了富集碳酸氢盐离子和碳酸盐离子或其组合的电解质溶液以及酸化物流。酸化物流还可用于各种化学方法中。例如,可以使用酸化物流来溶解富钙和/或镁的矿物如蛇纹石和橄榄石从而产生用于储存器816(或832)的电解质溶液。这样的电解质溶液可以加料以碳酸氢盐离子并随后使其充分变碱性以便沉淀如本文中所述的碳酸盐化合物。The embodiments described above produce an electrolyte solution enriched in bicarbonate ions and carbonate ions, or a combination thereof, and an acidified stream. Acidified streams can also be used in various chemical processes. For example, an acidifying stream may be used to dissolve calcium and/or magnesium rich minerals such as serpentine and olivine to produce an electrolyte solution for reservoir 816 (or 832 ). Such an electrolyte solution may be dosed with bicarbonate ions and subsequently made basic enough to precipitate carbonate compounds as described herein.
在一些实施方案中,可以使用第一电化学过程而从溶液中除去质子来促进CO2吸收,而没有伴随产生氢氧化物,同时可以使用第二电化学过程来产生氢氧化物以便进一步除去质子而使平衡朝着碳酸盐移动并且引起碳酸盐的沉淀。两种过程可以具有不同的电压要求,例如,第一过程可需要比第二过程更低的电压,由此最小化用于该方法中的总电压。例如,第一过程可以是如美国专利申请12/344,019(2008年12月24日,其全盘引入本文作为参考)所述的双电极方法,在1.0V或更小,或0.9V或更小,或0.8V或更小,或0.7V或更小,或0.6V或更小,或0.5V或更小,或0.4V或更小,或0.3V或更小,或0.2V或更小,或0.1V或更小操作,而第二过程可以是如上所述的低压氢氧化物生产方法,在1.5V或更小,或1.4V或更小,或1.3V或更小,或1.2V或更小,或1.1V或更小,1.0V或更小,或0.9V或更小,或0.8V或更小,或0.7V或更小,或0.6V或更小,或0.5V或更小,或0.4V或更小,或0.3V或更小,或0.2V或更小,或0.1V或更小操作。例如,在一些实施方案中第一过程是在0.6V或更小操作的双电极方法,而第二过程是在1.2V或更小操作的低压氢氧化物生产方法。In some embodiments, a first electrochemical process can be used to remove protons from solution to facilitate CO2 absorption without concomitant generation of hydroxides, while a second electrochemical process can be used to generate hydroxides for further removal of protons Instead, the equilibrium shifts towards the carbonate and causes the precipitation of the carbonate. The two processes may have different voltage requirements, for example, the first process may require a lower voltage than the second process, thereby minimizing the total voltage used in the method. For example, the first process may be a two-electrode method as described in U.S. Patent Application 12/344,019 (December 24, 2008, which is incorporated herein by reference in its entirety), at 1.0 V or less, or 0.9 V or less, or 0.8V or less, or 0.7V or less, or 0.6V or less, or 0.5V or less, or 0.4V or less, or 0.3V or less, or 0.2V or less, or 0.1V or less operation, while the second process may be a low pressure hydroxide production method as described above, at 1.5V or less, or 1.4V or less, or 1.3V or less, or 1.2V or less Small, or 1.1V or less, 1.0V or less, or 0.9V or less, or 0.8V or less, or 0.7V or less, or 0.6V or less, or 0.5V or less, Or 0.4V or less, or 0.3V or less, or 0.2V or less, or 0.1V or less operation. For example, in some embodiments the first process is a two electrode process operating at 0.6V or less, while the second process is a low pressure hydroxide production process operating at 1.2V or less.
描述于以下文献中的电化学法也是所考虑的:美国专利申请(公开号)2006/0185985,公开日为2006年8月24日;美国专利申请(公开号)2008/0248350,公开日为2008年10月9日;国际专利申请(公开号)WO 2008/018928,公开日为2008年2月14日;和国际专利申请(公开号)WO 2009/086460,公开日为2009年7月7日,其每个全盘引入本文作为参考。The electrochemical methods described in the following documents are also considered: U.S. Patent Application (Publication No.) 2006/0185985, published August 24, 2006; U.S. Patent Application (Publication No.) 2008/0248350, published 2008 October 9, 2008; International Patent Application (publication number) WO 2008/018928, published on February 14, 2008; and International Patent Application (publication number) WO 2009/086460, published on July 7, 2009 , each of which is incorporated herein by reference in its entirety.
化学计量法指出生产待沉淀的碳酸酯以便埋存来自CO2源的CO2需要除去来自原始的碳酸的两个质子,所述初始碳酸是当CO2被溶解在水中时形成的(参见以上式1-5)。除去第一个质子产生了碳酸氢盐,除去第二个产生了碳酸盐,其可以沉淀,例如,以二阶阳离子的碳酸盐,如碳酸镁或碳酸钙的形式。除去两个质子需要一些方法或方法的组合,这些通常需要能量。例如,如果质子是通过添加氢氧化钠除去的,可再生的氢氧化钠源通常是氯碱(chloralkali)方法,其使用电化学法,需要至少2.8V和固定量的电子/每摩尔的氢氧化钠。该能量需要可以按照碳足迹来表示,即为提供推进该方法的能量而生产的碳的量。Stoichiometry dictates that producing the carbonate to be precipitated in order to sequester CO from a CO source requires the removal of two protons from the original carbonic acid formed when CO is dissolved in water (see Eq. 1-5). Removal of the first proton produces bicarbonate and removal of the second produces carbonate, which can precipitate, for example, in the form of a carbonate of a secondary cation, such as magnesium carbonate or calcium carbonate. Removing two protons requires some method or combination of methods, and these usually require energy. For example, if protons are removed by adding NaOH, the renewable NaOH source is usually the chloralkali method, which uses electrochemical methods and requires at least 2.8 V and a fixed amount of electrons per mole of NaOH . This energy requirement can be expressed in terms of a carbon footprint, ie the amount of carbon produced to provide the energy to drive the process.
表示质子除去的给定方法的碳足迹的适宜方法是从CO2源中取出的CO2的百分比。也就是说,除去质子所需要的能量可以根据产生该能量的常规发电方法的CO2排放量来表示,其又可以表示为从CO2源取出的CO2的百分比。为了方便起见,并且作为在本发明的这个方面中的定义,在所述方法中“所产生的CO2”将被认为是为除去两个质子而提供足够的能量在常规的煤/蒸汽发电厂中将产生的CO2。对于最近几年的这样的发电厂来说,数据是公众可得的,其显示了每产生总MWh的能量产生了数吨的CO2。参见,例如具有通过结合″http://carma.″和″org/api/″而产生的地址的网站。对于这里的定义的目的,将使用1吨CO2/MWh的值,这接近地相应于典型的燃煤的发电厂;例如2000年WA Parish工厂产生了18,200,000MWh的能量,同时产生大约19,500,000吨的CO2,并且目前产生了21,300,00MWh的能量,同时产生了20,900,000吨的CO2,这平均起来非常接近于所定义的1吨CO2/MWh,这将用于本发明中。这些数值因此能够用于计算为除去足够的质子所必需的CO2产量以便从气体物流中除去CO2,并且将其与所除去的CO2进行比较。例如,在利用氯碱方法的方法中,所述氯碱方法在2.8V下操作而提供碱,并且用于埋存来自煤/蒸汽发电厂的CO2,使用1吨CO2/1MWh比例,为供给能量以由氯碱方法产生碱而除去两个质子而通过发电厂产生的CO2的量,大大高于200%的通过除去两个质子和以稳定形式沉淀CO2所埋存的CO2的量。作为在本发明的这个方面中“所产生的CO2”的定义的进一步的条件,在全部“所产生的CO2”中没有包括例如应归于再利用该方法的副产物以便除去质子的能量负荷的降低的理论或实际的计算(例如,在氯碱方法情况下,在燃料电池中使用在该方法中所产生的氢气或通过直接燃烧以产生能量)。另外,没有考虑使用可再生能源由发电厂所供给的电力的理论或实际的补充,例如,产生很少二氧化碳或没有产生二氧化碳的能源,例如风能、太阳能、潮汐能、水电能等。如果除去质子的方法包括使用氢氧化物或其它碱,包括天然存在的或储备的碱,所产生的CO2的量将是基于通过其产生碱的方法可化学计量计算的数量,例如,对于工业产生的碱来说,通过其产生碱的标准氯碱方法或其它方法,和对于天然碱来说,用于碱的天然产生的最佳理论模型。A suitable way to express the carbon footprint of a given method of proton removal is as a percentage of CO2 withdrawn from the CO2 source. That is, the energy required to remove protons can be expressed in terms of CO2 emissions from conventional power generation methods that generate this energy, which in turn can be expressed as a percentage of CO2 withdrawn from the CO2 source. For convenience, and as a definition in this aspect of the invention, " CO2 produced" in the process will be considered to provide enough energy to remove two protons in a conventional coal/steam power plant CO 2 will be produced. For such power plants of the last few years, data are publicly available which show tons of CO2 are produced per total MWh of energy produced. See, eg, a website with an address generated by combining "http://carma." and "org/api/". For the purposes of the definitions here, a value of 1 tonne of CO 2 /MWh will be used, which corresponds closely to a typical coal-fired power plant; for example the WA Parish plant produced 18,200,000 MWh of energy in 2000 while producing approximately 19,500,000 tonnes of CO 2 , and currently produces 21,300,00 MWh of energy, while producing 20,900,000 tons of CO 2 , which on average is very close to the defined 1 ton of CO 2 /MWh, which will be used in this invention. These values can thus be used to calculate the CO2 production necessary to remove enough protons to remove CO2 from the gas stream and compare it to the CO2 removed. For example, in a process utilizing the chlor-alkali process, which operates at 2.8V to provide alkali and is used to sequester CO2 from a coal/steam power plant, using a ratio of 1 ton CO2 /1MWh for supplying energy The amount of CO2 produced by the power plant with the removal of two protons from the production of the base by the chlor-alkali process is much higher than 200% of the amount of CO2 sequestered by the removal of the two protons and the precipitation of the CO2 in a stable form. As a further proviso to the definition of " CO2 produced" in this aspect of the invention, in all " CO2 produced" no energy load is included, for example, attributable to reusing the by-products of the process in order to remove protons Theoretical or practical calculations of the reduction (for example, in the case of the chlor-alkali process, the use of the hydrogen produced in the process in a fuel cell or by direct combustion to generate energy). In addition, theoretical or practical supplementation of electricity supplied by power plants using renewable energy sources, such as energy sources that generate little or no carbon dioxide, such as wind energy, solar energy, tidal energy, hydroelectric energy, etc., is not considered. If the method of proton removal involves the use of hydroxide or other bases, including naturally occurring or stored bases, the amount of CO2 produced will be the amount stoichiometrically calculated based on the method by which the base was produced, e.g. for industrial For bases produced, the standard chlor-alkali method or other method by which bases are produced, and for trona, the best theoretical model for the natural production of bases.
在一些实施方案中使用“所产生的CO2”的这种定义,本发明包括由人类产生的CO2的气体源形成稳定的含CO2的沉淀物,其中沉淀物的形成使用了用于由其中溶解了部分或全部的CO2的气体源的CO2的水溶液除去质子的方法,并且其中通过除去质子的方法产生的CO2小于100,90,80,70,65,60,55,50,45,40,35,30,25,20,15,10,或5%的通过所述沉淀物的形成从CO2的气体源除去的CO2。在一些实施方案中,本发明包括由人类产生的CO2的气体源形成稳定的含CO2的沉淀物,其中沉淀物的形成使用了用于由其中溶解了部分或全部的CO2的气体源的CO2的水溶液除去质子的方法,并且其中通过除去质子的方法产生的CO2小于70%的通过沉淀物的形成从CO2的气体源除去的CO2。在一些实施方案中,本发明包括由人类产生的CO2的气体源形成稳定的含CO2的沉淀物,其中沉淀物的形成使用了用于由其中溶解了部分或全部的CO2的气体源的CO2的水溶液除去质子的方法,并且其中通过除去质子的方法产生的CO2小于50%的通过沉淀物的形成从CO2的气体源除去的CO2。在一些实施方案中,本发明包括由人类产生的CO2的气体源形成稳定的含CO2的沉淀物,其中沉淀物的形成使用了用于由其中溶解了部分或全部的CO2的气体源的CO2的水溶液除去质子的方法,并且其中通过除去质子的方法产生的CO2小于30%的通过沉淀物的形成从CO2的气体源除去的CO2。在一些实施方案中,除去质子的方法是这样的方法,如电化学法,如本文中所述,其除去质子而没有产生碱,例如,氢氧化物。在一些实施方案中,除去质子的方法是这样的方法,如电化学法,如本文中所述,其除去质子,通过产生碱,例如,氢氧化物。在一些实施方案中,除去质子的方法是以下方法的组合:一种方法,如电化学法,如本文中所述,其除去质子而没有产生碱,例如,氢氧化物,和一种方法,如电化学法,如本文中所述,其除去质子,通过产生碱,例如,氢氧化物。在一些实施方案中,质子除去的方法包括电化学法,直接(例如,直接除去质子)或间接(例如,产生氢氧化物)除去质子。在一些实施方案中,使用方法,例如,电化学法的组合,其中第一方法,例如电化学法,直接除去质子,和第二方法,例如,电化学法,间接除去质子(例如,通过产生氢氧化物)。Using this definition of " CO2 produced" in some embodiments, the invention includes the formation of stable CO2 -containing precipitates from gaseous sources of anthropogenic CO2 , wherein the precipitates are formed using A process for the deprotonation of an aqueous solution of CO2 in which part or all of the gaseous source of CO2 is dissolved, and wherein the CO2 produced by the process for deprotonation is less than 100, 90, 80, 70, 65, 60, 55, 50, 45, 40, 35, 30, 25, 20, 15, 10, or 5% of the CO2 removed from the CO2 gas source by the formation of the precipitate. In some embodiments, the invention includes forming a stable CO2 -containing precipitate from a gas source of anthropogenic CO2 , wherein the precipitate is formed using a gas source from which some or all of the CO2 is dissolved. A method for the deprotonation of an aqueous solution of CO2 , and wherein the CO2 produced by the deprotonation method is less than 70% of the CO2 removed from the CO2 gas source by the formation of precipitates. In some embodiments, the invention includes forming a stable CO2 -containing precipitate from a gas source of anthropogenic CO2 , wherein the precipitate is formed using a gas source from which some or all of the CO2 is dissolved. A method for the deprotonation of an aqueous solution of CO2 , and wherein the CO2 produced by the deprotonation method is less than 50% of the CO2 removed from the CO2 gas source by the formation of precipitates. In some embodiments, the invention includes forming a stable CO2 -containing precipitate from a gas source of anthropogenic CO2 , wherein the precipitate is formed using a gas source from which some or all of the CO2 is dissolved. A method for the deprotonation of an aqueous solution of CO2 , and wherein the CO2 produced by the deprotonation method is less than 30% of the CO2 removed from the CO2 gas source by the formation of precipitates. In some embodiments, the method of removing protons is a method, such as an electrochemical method, as described herein, that removes protons without generating a base, eg, hydroxide. In some embodiments, the method of removing protons is a method, such as an electrochemical method, as described herein, which removes protons by generating a base, eg, a hydroxide. In some embodiments, the method of removing protons is a combination of: a method, such as an electrochemical method, as described herein, which removes protons without generating a base, e.g., hydroxide, and a method, Such as electrochemical methods, as described herein, which remove protons by generating a base, eg, hydroxide. In some embodiments, the method of proton removal includes electrochemical methods, direct (eg, direct removal of protons) or indirect (eg, generation of hydroxides) proton removal. In some embodiments, a combination of methods, e.g., electrochemical methods, is used, wherein a first method, e.g., electrochemical method, removes protons directly, and a second method, e.g., electrochemical method, removes protons indirectly (e.g., by producing hydroxide).
在有些情况下,在没有添加二价金属离子源的情况下,在CO2加料(例如,如上所述)后期望的产物的沉淀发生。因而,在具有CO2的加料反应器中加料水后,水然后没有与二价金属离子源接触,如一种或多种二价金属离子盐,例如,氯化钙,氯化镁,海盐等等。In some cases, precipitation of the desired product occurred after CO2 addition (eg, as described above) without the addition of a source of divalent metal ions. Thus, after the water is fed in the feed reactor with CO , the water is then not in contact with a source of divalent metal ions, such as one or more divalent metal ion salts, eg, calcium chloride, magnesium chloride, sea salt, and the like.
在本发明的一种实施方案中,可以使用碳酸盐沉淀过程以便有选择地从溶液中沉淀碳酸钙材料,以便提供期望比例的镁与钙,随后是另外的CO2加料,和在一些实施方案中,另外的MgMg离子加料,以及最终的碳酸盐沉淀步骤。这种实施方案可用于利用浓缩水如脱盐盐水,其中阳离子含量足够高,使得添加更多的Mg离子是困难的。这种实施方案还可用于任何浓度的溶液,其中期望产生两个不同的产物,主要地碳酸钙材料,和然后碳酸镁占优势的材料。In one embodiment of the present invention, a carbonate precipitation process may be used to selectively precipitate calcium carbonate material from solution to provide the desired ratio of magnesium to calcium, followed by additional CO dosing, and in some implementations scheme, additional MgMg ion dosing, and a final carbonate precipitation step. This embodiment can be used to utilize concentrated water such as desalinated brine, where the cation content is high enough that adding more Mg ions is difficult. This embodiment can also be used for solutions of any concentration where it is desired to produce two distinct products, a predominantly calcium carbonate material, and then a magnesium carbonate predominant material.
由给定的沉淀反应的产物的收率可以不同,这取决于许多因素,包括所使用的特定类型的水,水是否补充以二价金属离子,所使用的特定的沉淀流程等等。在有些情况下,用于沉淀产品的沉淀物流程是高收率沉淀流程。在这些情况下,对于每升水,由单个沉淀反应产生的(这指的是水经受沉淀条件的单次,如增加pH到9.5或更高、如10或更高的值,如上文更详细地综述的)产物的量可以是5g或更多、如10g或更多、15g或更多、20g或更多、25g或更多、30g或更多、35g或更多、40g或更多、45g或更多、50g或更多、60g或更多、70g或更多、80g或更多、90g或更多、100g或更多、120g或更多、140g或更多、160g或更多、180g或更多、200g或更多的存储稳定的二氧化碳埋存产物。在有些情况下,对于每升水产生的产物的量是5至200g,如10至100g,包括20至100g。在以下情况中,其中在使水经受沉淀条件前,水的二价金属离子含量没有增补(例如其中水是海水并且海水没有补充一种或多种二价金属离子源),产物的收率可以是5至20g产物/1升的水,如5至10,例如,6至8,g产物/1升的水。在其它情况中,其中水补充以二价金属离子源,如镁和/或钙离子,产物的收率可以更高,2倍,3倍,5倍,10倍,20倍或更高,使得这样的方法的收率在一些实施方案中可以为,对于每升经受沉淀条件的水,10至200,如50至200包括100至200g产物。The yield of product from a given precipitation reaction can vary depending on many factors, including the particular type of water used, whether the water is supplemented with divalent metal ions, the particular precipitation protocol used, and the like. In some cases, the precipitation process used to precipitate the product is a high yield precipitation process. In these cases, for each liter of water, produced by a single precipitation reaction (this refers to a single time the water is subjected to precipitation conditions, such as increasing the pH to a value of 9.5 or higher, such as 10 or higher, as described in more detail above The amount of product reviewed) may be 5 g or more, such as 10 g or more, 15 g or more, 20 g or more, 25 g or more, 30 g or more, 35 g or more, 40 g or more, 45 g or more, 50g or more, 60g or more, 70g or more, 80g or more, 90g or more, 100g or more, 120g or more, 140g or more, 160g or more, 180g or more, 200 g or more of storage-stable carbon dioxide sequestration products. In some cases, the amount of product produced is 5 to 200 g, such as 10 to 100 g, including 20 to 100 g, per liter of water. In cases where the divalent metal ion content of the water is not supplemented prior to subjecting the water to precipitation conditions (e.g. where the water is seawater and the seawater is not supplemented with one or more sources of divalent metal ions), the yield of product can be It is 5 to 20 g product/1 liter of water, such as 5 to 10, eg, 6 to 8, g product/1 liter of water. In other cases, where the water is supplemented with a source of divalent metal ions, such as magnesium and/or calcium ions, the yield of product may be higher, 2-fold, 3-fold, 5-fold, 10-fold, 20-fold or higher, such that The yield of such a process may in some embodiments be from 10 to 200, such as from 50 to 200 including 100 to 200 g of product per liter of water subjected to the precipitation conditions.
由给定的沉淀反应的产物的收率可以不同,这取决于许多因素,包括所使用的特定类型的水,水是否补充以二价金属离子,所使用的特定的沉淀流程等等。在有些情况下,用于沉淀产品的沉淀物流程是高收率沉淀流程。在这一情况下,在平均72小时的连续操作下(即将沉淀条件施加到水或吸收溶液),由1升的水(即吸收溶液)产生的产物的量可以是,对于每升水来说,5g或更多、如10g或更多、15g或更多、20g或更多、25g或更多、30g或更多、35g或更多、40g或更多、45g或更多、50g或更多、60g或更多、70g或更多、80g或更多、90g或更多、100g或更多、120g或更多、140g或更多、160g或更多、180g或更多、200g或更多的存储稳定的二氧化碳埋存产物。在有些情况下,在平均72小时的连续操作下,对于每升水产生的产物的量是5至200g,如10至100g,包括20至100g。在以下情况中,其中在使水经受沉淀条件前,水的二价金属离子含量没有增补(例如其中水是海水并且海水没有补充一种或多种二价金属离子源),产物的收率可以是5至20g产物/1升的水,如5至10,例如,6至8,g产物/1升的水,在平均72小时的连续操作下。在其它情况中,其中水补充以二价金属离子源,如镁和/或钙离子,产物的收率可以更高,2倍,3倍,5倍,10倍,20倍或更高,使得这样的方法的收率在一些实施方案中可以为,对于每升经受沉淀条件的水,10至200,如50至200包括100至200g产物,在平均72小时的连续操作下。The yield of product from a given precipitation reaction can vary depending on many factors, including the particular type of water used, whether the water is supplemented with divalent metal ions, the particular precipitation protocol used, and the like. In some cases, the precipitation process used to precipitate the product is a high yield precipitation process. In this case, under an average of 72 hours of continuous operation (i.e. applying precipitation conditions to the water or the absorption solution), the amount of product produced from 1 liter of water (i.e. the absorption solution) may be, for each liter of water, 5g or more, such as 10g or more, 15g or more, 20g or more, 25g or more, 30g or more, 35g or more, 40g or more, 45g or more, 50g or more , 60g or more, 70g or more, 80g or more, 90g or more, 100g or more, 120g or more, 140g or more, 160g or more, 180g or more, 200g or more Storage-stable carbon dioxide sequestration products. In some cases, the amount of product produced per liter of water is 5 to 200 g, such as 10 to 100 g, including 20 to 100 g, under an average of 72 hours of continuous operation. In cases where the divalent metal ion content of the water is not supplemented prior to subjecting the water to precipitation conditions (e.g. where the water is seawater and the seawater is not supplemented with one or more sources of divalent metal ions), the yield of product can be From 5 to 20 g product/1 liter of water, such as 5 to 10, eg, 6 to 8, g product/1 liter of water, at an average of 72 hours of continuous operation. In other cases, where the water is supplemented with a source of divalent metal ions, such as magnesium and/or calcium ions, the yield of product may be higher, 2-fold, 3-fold, 5-fold, 10-fold, 20-fold or higher, such that The yield of such a process may in some embodiments range from 10 to 200, such as 50 to 200 including 100 to 200 g of product per liter of water subjected to the precipitation conditions, under an average of 72 hours of continuous operation.
在某些实施方案中,使用多步方法。在这些实施方案中,可以使用碳酸盐沉淀过程以便有选择地从溶液沉淀碳酸钙材料,随后CO2加料和随后的碳酸盐沉淀的另外的步骤。另外的CO2加料和碳酸盐沉淀的步骤有时候可以重复1、2、3、4、5、6、7、8、9、10或更多次,每一次循环沉淀另外数量的碳酸盐材料。有时候,最终的pH为pH 8至pH 10,如pH 9至pH 10,包括pH 9.5至pH 10,例如,pH 9.6至pH 9.8。In certain embodiments, a multi-step approach is used. In these embodiments, a carbonate precipitation process may be used to selectively precipitate calcium carbonate material from solution, followed by additional steps of CO dosing and subsequent carbonate precipitation. The steps of additional CO2 dosing and carbonate precipitation can sometimes be repeated 1, 2, 3, 4, 5, 6, 7, 8, 9, 10 or more times, each cycle precipitating an additional amount of carbonate Material. Sometimes the final pH is from
在某些实施方案中,可以使用两个或更多个反应器以便进行本文中所述的方法。在这些实施方案中,该方法可以包括第一反应器和第二反应器。在这些情况下,第一反应器用于使初始的水与镁离子源接触和用于用CO2加料初始的水,如上所述。水可以搅拌以便促进镁离子源的溶解和促进初始的水与CO2的接触。有时候,在CO2加料的水转移到第二反应器前,停止搅拌CO2加料的水,使得不溶解的固体可以靠重力沉降。CO2加料的水然后从第一反应器转移到第二反应器。在转移CO2加料的水至第二反应器后,可以进行碳酸盐沉淀的步骤,如本文中所述。In certain embodiments, two or more reactors may be used in order to carry out the methods described herein. In these embodiments, the process can include a first reactor and a second reactor. In these cases, the first reactor was used to contact the initial water with the source of magnesium ions and to feed the initial water with CO2 , as described above. The water may be stirred to facilitate dissolution of the source of magnesium ions and to facilitate initial water contact with CO2 . Occasionally, agitation of the CO2 - fed water was stopped before the CO2- fed water was transferred to the second reactor so that undissolved solids could settle by gravity. The CO fed water is then transferred from the first reactor to the second reactor. After transferring the CO2 - fed water to the second reactor, a carbonate precipitation step can be performed, as described herein.
在某些实施方案中,多步方法,如上所述,使用了两个或更多个反应器,如上所述,可用于进行本文中所述的方法。在这些实施方案中,第一反应器用于使初始的水与镁离子源接触和用于用CO2加料初始的水,如上所述。随后,CO2加料的水从第一反应器转移到第二反应器用于碳酸盐沉淀反应。在某些实施方案中,CO2加料和随后的碳酸盐沉淀的一个或多个另外的步骤可以在第二反应器中进行,如上所述。In certain embodiments, multi-step processes, as described above, using two or more reactors, as described above, can be used to carry out the methods described herein. In these embodiments, the first reactor is used to contact the initial water with the source of magnesium ions and to feed the initial water with CO , as described above. Subsequently, CO fed water was transferred from the first reactor to the second reactor for carbonate precipitation reaction. In certain embodiments, one or more additional steps of CO dosing and subsequent carbonate precipitation may be performed in a second reactor, as described above.
在某些实施方案中,可以使用有利于形成特定形态的碳酸盐化合物沉淀物的沉淀条件。例如,相比于形成结晶碳酸盐化合物沉淀物来说,可以使用有利于形成无定形碳酸盐化合物沉淀物的沉淀条件。在这些情况下,除使初始的水与镁离子源接触和用CO2加料初始的水外,如上所述,可以添加沉淀促进剂(facilitator)。在这些情况下,沉淀促进剂(facilitator)促进了在足以晶核形成但不足以晶体形成和生长的较低的pH下碳酸盐化合物沉淀物的形成。沉淀促进剂(facilitator)的实例包括但不局限于硫酸铝(Al2SO4)3。在某些实施方案中,所添加的沉淀促进剂(facilitator)的量为1ppm-1000ppm,如1ppm-500ppm,包括10ppm-200ppm,例如25ppm-75ppm。另外,在碳酸盐化合物沉淀形成期间,通过交替CO2加料和随后的碳酸盐沉淀,如上所述,水的pH值可以维持在6-8,如7-8。In certain embodiments, precipitation conditions may be used that favor the formation of a particular morphology of the carbonate compound precipitate. For example, precipitation conditions may be used that favor the formation of an amorphous carbonate compound precipitate over the formation of a crystalline carbonate compound precipitate. In these cases, in addition to contacting the initial water with a source of magnesium ions and dosing the initial water with CO 2 , a precipitation facilitator may be added as described above. In these cases, the precipitation facilitator promotes the formation of carbonate compound precipitates at lower pH sufficient for nucleation but insufficient for crystal formation and growth. Examples of precipitation facilitators include, but are not limited to, aluminum sulfate ( Al2SO4 )3. In certain embodiments, the amount of precipitation facilitator added is 1 ppm-1000 ppm, such as 1 ppm-500 ppm, including 10 ppm-200 ppm, such as 25 ppm-75 ppm. In addition, during the formation of carbonate compound precipitates, by alternating CO2 feeding and subsequent carbonate precipitation, as described above, the pH of the water can be maintained at 6-8, such as 7-8.
或者,在其它实施方案中,相比于形成无定形碳酸盐化合物沉淀物来说,可以使用有利于形成结晶碳酸盐化合物沉淀物的沉淀条件。Alternatively, in other embodiments, precipitation conditions may be used that favor the formation of a crystalline carbonate compound precipitate over the formation of an amorphous carbonate compound precipitate.
在下文中相对于本申请附图的描述提供了关于用于本发明的某些实施方案的特定的沉淀流程的更多细节。The description below with respect to the figures of the present application provides more details regarding the specific precipitation schemes used in certain embodiments of the present invention.
在由水产生沉淀物产物后,产生了组合物,其包括沉淀的产物和母液(即,从中产生沉淀的产物的其余液体)。这种组合物可以是沉淀物和母液的浆液。After producing the precipitated product from water, a composition is produced that includes the precipitated product and mother liquor (ie, the remaining liquid from which the precipitated product was produced). This composition may be a slurry of precipitate and mother liquor.
如上所述,在埋存二氧化碳中,在其产生后以一些方式处理沉淀的产物。短语“处理”是指产品被放置在存储位置或者用于在另一产物(即,制造或人造物品)中进一步的使用,其中至少它被存储在该其它产物中达该其它产物的预期的生存期。在有些情况下,这种处理步骤包括将上述浆液组合物运送到长期储存位置。存储位置可以是地上位置、地下位置或水下位置。在这些实施方案中,在将浆液置于存储位置后,浆液的上层清液组分可以自然地与沉淀物分离,例如通过蒸发,分散等。As noted above, in sequestering carbon dioxide, the precipitated product is handled in some way after its production. The phrase "processing" means that a product is placed in a storage location or for further use in another product (i.e., a manufactured or manufactured item) where at least it is stored in that other product for the expected life of that other product Expect. In some cases, this processing step includes transporting the aforementioned slurry composition to a long-term storage location. The storage location may be an aboveground location, an underground location or an underwater location. In these embodiments, the supernatant component of the slurry may be naturally separated from the precipitate, such as by evaporation, dispersion, etc., after the slurry is placed in the storage location.
在期望的情况下,所得的沉淀的产物可以与浆液的上层清液组分分离。使用许多适宜方法的任一种,可以实现沉淀的产物的分离。如在本文中进一步详述的,液-固分离器如Epuramat′s Extrem-Separator(″ExSep″)液-固分离器,Xerox PARC′s螺旋浓缩器,或Epuramat′s ExSep或Xerox PARC′s螺旋浓缩器中任何一个的变体可用于一些实施方案中。通过干燥沉淀的产物以产生干燥的沉淀产物,也可实现分离。所考虑的干燥流程包括从母液中过滤沉淀物以产生滤液并随后风干滤液。在滤液被风干的情况下,风干可以在-70至120℃的温度,根据期望。在有些情况下,干燥可以包括将浆液置于干燥位置,如尾矿池,使沉淀物的液体组分蒸发并且留下期望的干燥产品。还考虑的是冷冻干燥(即,冻干)流程,其中沉淀物被冷冻,降低周围压力并且添加足够的热量以使得材料中的冷冻水直接从冷冻沉淀相升华为气体。又一个所考虑的干燥流程是喷雾干燥,其中包含沉淀物的液体通过下述方式干燥:将其进料通过热气,例如,其中液体进料通过雾化器泵送到主干燥室中并且使热气以相对于雾化器方向顺流或逆流的形式通过。Where desired, the resulting precipitated product can be separated from the supernatant component of the slurry. Isolation of the precipitated product can be achieved using any of a number of suitable methods. As further detailed herein, a liquid-solid separator such as Epuramat's Extrem-Separator ("ExSep") liquid-solid separator, Xerox PARC's spiral concentrator, or Epuramat's ExSep or Xerox PARC's Variations of any of the spiral concentrators may be used in some embodiments. Isolation may also be achieved by drying the precipitated product to produce a dry precipitated product. The drying procedure considered involves filtering the precipitate from the mother liquor to produce a filtrate followed by air drying of the filtrate. In case the filtrate is air-dried, the air-drying can be at a temperature of -70 to 120 °C, as desired. In some cases, drying may involve placing the slurry in a dry location, such as a tailings pond, allowing the liquid components of the sediment to evaporate and leaving the desired dry product. Also contemplated is a freeze-drying (ie, lyophilization) procedure in which the precipitate is frozen, the ambient pressure is reduced and enough heat is added that the frozen water in the material sublimates directly from the frozen precipitate phase to gas. Yet another drying process considered is spray drying, in which the liquid containing the precipitate is dried by passing its feed through hot gas, for example, where the liquid feed is pumped through an atomizer into the main drying chamber and the hot gas Pass in either co-current or counter-current relative to the direction of the nebulizer.
在沉淀的产物与母液分离的情况下,所得的沉淀物可以以各种不同的方式处理,如下文进一步详细说明的。例如,可以使用沉淀物作为建筑材料的组分,如下文更详细论述的。或者,沉淀物可以被置于长期储存位置(有时在本领域中称为碳银行),其中所述位置可以是地上位置,地下位置或水下位置。关于所考虑的处理流程的更多细节在下文中提供。Where the precipitated product is separated from the mother liquor, the resulting precipitate can be treated in various ways, as explained in further detail below. For example, sediment can be used as a component of building materials, as discussed in more detail below. Alternatively, the sediment can be placed in a long-term storage location (sometimes referred to in the art as a carbon bank), where the location can be an aboveground location, an underground location, or an underwater location. More details on the considered processing flow are provided below.
所得的母液也可被处理,根据期望。例如,母液可以返回到水的来源,例如,海洋,或者到另一位置。在某些实施方案中,母液可以与CO2源接触,例如,如上所述,以便埋存其它的CO2。例如,在母液将返回到海洋的情况下,母液可以以足以增加存在于母液中的碳酸盐离子的浓度的方式与CO2的气体源接触。使用任何适宜的流程,如上述的那些,可以进行接触。在某些实施方案中,母液具有碱性pH,并且以足以降低pH至5-9,例如6-8.5,包括7.5-8.2的方式进行与CO2源接触。The resulting mother liquor can also be processed, as desired. For example, the mother liquor can be returned to the source of the water, eg, the ocean, or to another location. In certain embodiments, the mother liquor can be contacted with a source of CO2 , eg, as described above, to sequester additional CO2 . For example, where the mother liquor is to be returned to the ocean, the mother liquor may be contacted with a gas source of CO2 in a manner sufficient to increase the concentration of carbonate ions present in the mother liquor. Contacting can be performed using any suitable procedure, such as those described above. In certain embodiments, the mother liquor has an alkaline pH, and the contacting with the source of CO2 is performed in a manner sufficient to lower the pH to 5-9, such as 6-8.5, including 7.5-8.2.
本发明的方法可以在陆地或海进行,例如,在陆地位置,其中合适的水存在于该位置或者被运输到该位置,或者在海洋或其它含碱土金属的水体(其是天然存在的或人造的)中。在某些实施方案中,使用一种系统进行上述方法,其中这样的系统包括如下更详细描述的那些。The method of the present invention may be carried out on land or sea, for example, at a land location where suitable water is present or transported to the location, or in the ocean or other alkaline earth metal-containing water bodies (which are naturally occurring or man-made) of). In certain embodiments, the above methods are performed using a system, wherein such systems include those described in more detail below.
本申请的以上部分提供了本发明的方法的各个方面的综述。根据本发明的某些附图,现在更详细地进一步评述本发明的某些实施方案。The above section of the application provides an overview of various aspects of the methods of the invention. Certain embodiments of the invention will now be further reviewed in more detail, based on certain figures of the invention.
图6A提供了二氧化碳埋存方法的示意流程图,其可以在系统中实现,其中所述系统可以表示为单机装置或者另一种类型装置如发电装置、水泥生产装置等的集成部分。在图6A中,水10被输送到沉淀反应器20,例如,通过管道或其它适宜方式,并且经受碳酸盐矿物沉淀条件。图6A中举例说明的用于该方法的水是包括例如一种或多种碱土金属离子如Ca2+和Mg2+的水。在本发明的某些实施方案中,所考虑的水是包括如下数量的钙的水:50ppm-20,000ppm、如200ppm-5000ppm和包括400ppm-1000ppm。还考虑的水是包括如下数量的镁的水:50ppm-40,000ppm、如100ppm-10,000ppm和包括500ppm-2500ppm。在本发明的实施方案中,水(例如,含碱土金属离子的水)是盐水。如上所述,所考虑的盐水包括许多不同类型的除了淡水以外的水流体,如微咸水、海水和盐水(包括人造盐水,例如地热装置废水、脱盐废水等,以及如本文中所述的天然存在的盐水),以及其它盐度大于淡水的盐碱水盐碱水盐水。盐水是用盐饱和或几乎饱和的水并且具有50ppt(千分率)或更多的盐度。微咸水是比淡水成的水,但是没有像海水一样成,具有0.5-35ppt的盐度。海水可以是来自海或海洋的水并且具有约35-约50ppt的盐度。淡水是具有小于5ppt溶解盐的盐度的水。所考虑的盐水可以由天然存在的来源如海,海洋,湖泊,沼泽,河口,泻湖等或者人造来源获得,根据期望。Figure 6A provides a schematic flow diagram of a carbon dioxide sequestration method that can be implemented in a system, where the system can be represented as a stand-alone unit or as an integrated part of another type of unit such as a power generation unit, cement production unit, and the like. In Fig. 6A,
如上所述,所考虑的水还包括淡水。在某些实施方案中,用于本发明的水可以是富含矿物的,例如富含钙和/或镁的淡水源。在一些实施方案中,淡水,如富含钙的水,可以结合以溶液中的硅酸镁矿物,如橄榄石或蛇纹石,所述溶液变成酸性,由于添加来自碳酸的二氧化碳,所述碳酸溶解了硅酸镁,导致形成硅酸镁钙碳酸盐化合物。在某些实施方案中,水源可以是淡水,其中添加金属离子,例如,钠,钾,钙,镁等。金属离子可以添加到淡水源,使用任何适宜的流程,例如,以固体、水溶液、悬浮液等的形式。As mentioned above, water under consideration also includes fresh water. In certain embodiments, the water used in the present invention may be a mineral-rich, eg, calcium and/or magnesium-rich freshwater source. In some embodiments, fresh water, such as calcium-enriched water, can incorporate magnesium silicate minerals, such as olivine or serpentine, in a solution that becomes acidic due to the addition of carbon dioxide from carbonic acid, the The carbonic acid dissolves the magnesium silicate, resulting in the formation of calcium magnesium silicate carbonate compounds. In certain embodiments, the water source may be fresh water to which metal ions, eg, sodium, potassium, calcium, magnesium, etc., have been added. Metal ions may be added to the freshwater source using any suitable procedure, for example, in the form of a solid, aqueous solution, suspension, and the like.
在某些实施方案中,水可以获自工业设备,所述工业设备还提供着气态废物物流。例如,在水冷却的工业设备中,如海水冷却的工业设备,已经由工业设备使用的水然后可以被送到沉淀系统并且在沉淀反应中用作水。在期望的情况下,在进入沉淀反应器前,水可以被冷却。这样的方法可以例如与直通式冷却系统一起使用。例如,城市或农业用水供给可以用作工业设备用的直通式冷却系统。来自工业设备的水然后可以用于沉淀流程,其中输出水具有降低的硬度和更大的纯度。在期望的情况下,这样的系统可以被改变从而包括安全措施,例如,用于检测干扰(如添加毒物)并且与政府机构例如国土安全机构或其它机构配合。在这样的实施方案中可以使用另外的干扰或攻击保护措施。In certain embodiments, water may be obtained from an industrial facility that also provides a gaseous waste stream. For example, in water cooled industrial plants, such as seawater cooled industrial plants, the water that has been used by the industrial plant can then be sent to the precipitation system and used as water in the precipitation reaction. Where desired, the water can be cooled before entering the precipitation reactor. Such an approach may be used, for example, with a flow-through cooling system. For example, a municipal or agricultural water supply can be used as a flow-through cooling system for industrial equipment. The water from the industrial facility can then be used in the sedimentation process, where the output water has reduced hardness and greater purity. Where desired, such systems may be modified to include safety measures, for example, to detect tampering (such as adding poisons) and to cooperate with government agencies such as Homeland Security or other agencies. Additional interference or attack protection measures may be used in such embodiments.
如图6A所示,工业设备气态废物物流30在沉淀步骤20接触水以产生CO2加料的水(其可以发生在加料反应器中,在某些实施方案中)。水可以是吸收溶液,其中吸收溶液允许将二氧化碳和/或来自气体的其它组分引入吸收溶液中。吸收溶液可以是盐水,(例如海水和/或盐水),或碱性溶液。吸收溶液可以包括固体颗粒物并且可以描述为浆液。CO2加料的水是指已经与CO2气体接触的水,其中CO2分子已经结合以水分子从而产生了例如碳酸,碳酸氢盐和碳酸盐离子。在该步中加料水获得了水的“CO2含量”的增加,例如以碳酸、碳酸氢盐和碳酸盐离子的形式,并且伴随与水接触的废物物流的CO2的量的减少。在一些实施方案中,CO2加料的水是酸性的,具有6.0或更小,如4.0或更小,和包括3.0和更小的pH值。在某些实施方案中,由于该接触步骤,用于加料水的气体的CO2的量降低达85%或更多,如99%或更多,使得该方法除去50%或更多,如75%或更多,例如,85%或更多,包括99%或更多的原来存在于与水接触的气态废物物流中的CO2。所考虑的接触流程包括但不局限于:直接接触流程,例如,鼓泡气体通过水体积,顺流接触方式,即同向流动的气液相物流之间的接触,逆流接触,即反向流动的气液相物流之间的接触,等等。气体物流可以垂直地、水平地或者以某其它角度接触水源。As shown in Figure 6A, industrial facility
CO2可以与来自以下位置中的一个或多个的水源接触:水(例如,含碱土金属离子的水)的表面水平之下,之上,或之处。通过使用浸溃器,鼓泡器,流体文丘里反应器,喷布器,气体过滤器,喷雾器,塔盘,催化鼓泡塔反应器,吸出管型反应器或填料塔反应器等可以实现接触,这可能是适宜的。在期望的情况下,两个或更多个不同的CO2加料反应器(如塔或其它类型的反应器配置结构)可以使用,例如,串联或并联,如三个或更多个,四个或更多个,等等。在某些实施方案中,各种手段,例如,机械搅拌,电磁搅拌,旋转器,振荡器,振动器,鼓风机,超声破碎,用于搅拌或搅动反应溶液的,被用于增加CO2和水源之间的接触。The CO2 may be contacted with a water source from one or more of the following locations: below, above, or at the surface level of the water (eg, alkaline earth metal ion-containing water). Contact can be achieved by using dippers, bubblers, fluid venturi reactors, spargers, gas filters, sprayers, trays, catalytic bubble column reactors, suction tube reactors or packed column reactors, etc. , which may be appropriate. Where desired, two or more different CO2- feeding reactors (such as columns or other types of reactor configurations) can be used, for example, in series or in parallel, such as three or more, four or more, etc. In certain embodiments, various means, e.g., mechanical stirring, electromagnetic stirring, rotators, shakers, vibrators, blowers, sonication, for stirring or agitating the reaction solution, are used to increase CO and water sources contact between.
在图6A中所述的实施方案中,来自水源10的水(例如,包括碱土金属离子的水)首先加料以CO2以产生CO2加料的水,该CO2然后经受碳酸盐矿物沉淀条件。如图6A中所述,在沉淀步骤20,CO2气体物流30接触水。在沉淀步骤20,所提供的气体物流30接触合适的水以产生CO2加料的水。CO2加料的水是指已经与CO2气体接触的水,其中CO2分子已经结合以水分子从而产生了例如碳酸,碳酸氢盐和碳酸盐离子。在该步中加料水获得了水的“CO2含量”的增加,例如以碳酸、碳酸氢盐和碳酸盐离子的形式,并且伴随与水接触的废物物流的pCO2的减少。CO2加料的水可以是酸性的,具有6或更小,如5或更小,和包括4和更小的pH值。在一些实施方案中,CO2加料的水不是酸性的,例如具有7或更大的pH值,例如7-10,或7-9,或7.5-9.5,或8-10,或8-9.5,或8-9的pH值。在某些实施方案中,用于加料水的气体的CO2的浓度是10%或更高、25%或更高、包括50%或更高、如75%或更高的。In the embodiment depicted in FIG. 6A , water from water source 10 ( e.g. , water comprising alkaline earth metal ions) is first spiked with CO to produce CO - fed water, which is then subjected to carbonate mineral precipitation conditions . As depicted in Figure 6A, in the
CO2加料和碳酸盐矿物沉淀可以在系统的相同的或不同的反应器中进行。因而,根据本发明的某些实施方案,如在图6A中在步骤20处举例说明的,例如加料和沉淀可以在系统的相同反应器中进行。在本发明的其它实施方案中,这两步可以在单独的反应器中进行,使得水首先在加料反应器中加料以CO2并且然后在单独的反应器中,所得的CO2加料的水然后经受沉淀条件。可以使用其它反应器,例如,用期望的矿物加料水。 CO2 feeding and carbonate mineral precipitation can be performed in the same or different reactors of the system. Thus, according to certain embodiments of the invention, as illustrated at
水与CO2源的接触可以在水经受CO2沉淀条件之前和/或期间进行。因此,本发明的实施方案包括其中在使一定体积的水(例如,含碱土金属离子的水)经受矿物沉淀条件前使一定体积的水接触CO2源的方法。本发明的实施方案还包括其中在一定体积的水正在经受碳酸盐化合物沉淀条件时使一定体积的水接触CO2源的方法。本发明的实施方案包括其中在一定体积的水(例如,含碱土金属离子的水)经受碳酸盐化合物沉淀条件之前和在一定体积的水正在经受碳酸盐化合物沉淀条件时,使一定体积的水接触CO2源的方法。在一些实施方案中,相同的水可以循环不止一次,其中沉淀的第一循环主要地除去碳酸钙和碳酸镁矿物并且留下水,可以向其中添加金属离子,例如,碱土金属离子,并且其可以具有更多的通过其循环的CO2,这沉淀了更多的碳酸盐化合物。The contacting of the water with the CO2 source can occur before and/or during the water is subjected to the CO2 precipitation conditions. Accordingly, embodiments of the invention include methods wherein a volume of water (eg, alkaline earth metal ion-containing water) is contacted with a CO source prior to subjecting the volume of water to mineral precipitation conditions. Embodiments of the invention also include methods wherein a volume of water is contacted with a source of CO2 while the volume of water is being subjected to carbonate compound precipitation conditions. Embodiments of the present invention include wherein a volume of water (e.g., water containing alkaline earth metal ions) is subjected to carbonate compound precipitation conditions prior to and while the volume of water is being subjected to carbonate compound precipitation conditions. The method by which water contacts the CO2 source. In some embodiments, the same water can be cycled more than once, where the first cycle of precipitation primarily removes calcium and magnesium carbonate minerals and leaves water, to which metal ions, e.g., alkaline earth metal ions, can be added, and which can have The more CO2 that circulates through it, this precipitates more carbonate compounds.
无论当CO2接触水时,在有些情况下当CO2接触水时,水不是非常碱性,使得接触CO2的水可以具有10或更低,如9.5或更低,包括9或更低并且甚至8或更低的pH值。在一些实施方案中,接触CO2的水不是首先由电化学流程变成碱性的水。在一些实施方案中,接触CO2的水不是通过添加氢氧化物如氢氧化钠已经变成碱性的水。在一些实施方案中,水是如通过添加一定量的氧化物如氧化钙或氧化镁仅仅变成弱碱性的水。Regardless of when the CO2 contacts the water, in some cases when the CO2 contacts the water, the water is not very alkaline such that the water contacted by the CO2 can have a value of 10 or lower, such as 9.5 or lower, including 9 or lower and Even a pH of 8 or lower. In some embodiments, the water exposed to CO2 is not water that was first made alkaline by an electrochemical process. In some embodiments, the water exposed to CO2 is not water that has been made alkaline by the addition of hydroxides, such as sodium hydroxide. In some embodiments, the water is water that has only been made slightly alkaline, such as by adding an amount of an oxide such as calcium oxide or magnesium oxide.
如上所述,影响引入气体到液体中的效率的参数之一是液体的溶液化学。气体将具有不同的特定于溶液的溶解度,基于溶液的离子含量和pH值。例如,相比于结合到具有中性或酸性pH的溶液来说,二氧化碳气体更容易地结合到具有碱性pH的溶液,如NaOH的水溶液。在一些实施方案中,气体是工艺过程的烟道气。在一些实施方案中,该工艺过程包括化石燃料的燃烧。在一些实施方案中,气体包括CO2。在这些实施方案中,所述方法和设备可被调整以便最优化从气体中将CO2气体引入液体。在这样的实施方案中,提供了各种条件(例如溶液化学,表面积与体积比,液体流动速率与气体流动速率比值(L/G),停留时间),其容许至少10克的CO2被吸收,按每100ml的液体计。在一些实施方案中,提供了各种条件,其容许每100ml的液体至少20克的CO2被吸收,如每100ml的液体,至少30克,至少40克,至少50克,至少60克,至少70克,至少80克,至少90克,至少100克,或大于100克的CO2被吸收。在一些实施方案中,所述方法和装置提供了各种条件(例如溶液化学,表面体积比,停留时间),其中气体中的至少50%的CO2从气体中除去而进入液体或浆液中。在一些实施方案中,提供了各种条件,其允许气体中的至少60%的CO2被除去,如气体中的至少70%、至少80%、或至少90%的CO2被从气体中除去而进入液体或浆液中。在一些实施方案中,提供了各种条件,其允许气体中的至少95%的CO2被从气体中除去而进入液体或浆液中。在一些实施方案中,所述方法和装置提供了各种条件(例如溶液化学,表面体积比,液体流动速率与气体流动速率比值(L/G),停留时间),其中至少50%的气体组分(例如NOx,SOx,汞)从气体中除去而进入液体或浆液中。在一些实施方案中,提供了各种条件,其允许至少60%的气体组分被除去,如至少70%、至少80%、或至少90%的气体组分被从气体中除去而进入液体或浆液中。在一些实施方案中,提供了各种条件,其允许至少95%的气体组分被从气体中除去而进入液体或浆液中。As mentioned above, one of the parameters affecting the efficiency of introducing a gas into a liquid is the solution chemistry of the liquid. Gases will have different solution-specific solubilities, based on the ion content and pH of the solution. For example, carbon dioxide gas binds more readily to a solution with a basic pH, such as an aqueous solution of NaOH, than to a solution with a neutral or acidic pH. In some embodiments, the gas is the flue gas of a process. In some embodiments, the process includes the combustion of fossil fuels. In some embodiments, the gas includes CO2 . In these embodiments, the methods and apparatus can be tuned to optimize the introduction of CO2 gas from a gas to a liquid. In such embodiments, various conditions (e.g., solution chemistry, surface area to volume ratio, liquid flow rate to gas flow rate ratio (L/G), residence time) are provided that allow at least 10 grams of CO to be absorbed , calculated per 100ml of liquid. In some embodiments, conditions are provided which allow at least 20 grams of CO to be absorbed per 100 ml of liquid, such as at least 30 grams, at least 40 grams, at least 50 grams, at least 60 grams, at least 70 grams, at least 80 grams, at least 90 grams, at least 100 grams, or more than 100 grams of CO2 is absorbed. In some embodiments, the methods and devices provide conditions (eg, solution chemistry, surface volume ratio, residence time) where at least 50% of the CO in the gas is removed from the gas into a liquid or slurry. In some embodiments, conditions are provided that allow at least 60% of the CO in the gas to be removed, such as at least 70%, at least 80%, or at least 90% of the CO in the gas to be removed from the gas into liquids or slurries. In some embodiments, conditions are provided that allow at least 95% of the CO2 in the gas to be removed from the gas into a liquid or slurry. In some embodiments, the methods and devices provide conditions (e.g., solution chemistry, surface-to-volume ratio, ratio of liquid flow rate to gas flow rate (L/G), residence time) in which at least 50% of the gas group Particles (such as NOx, SOx, mercury) are removed from the gas into the liquid or slurry. In some embodiments, conditions are provided that allow at least 60% of the gaseous components to be removed, such as at least 70%, at least 80%, or at least 90% of the gaseous components to be removed from the gas into the liquid or in the slurry. In some embodiments, conditions are provided that allow at least 95% of the gas components to be removed from the gas into the liquid or slurry.
沉淀反应的优化是高效率的气液接触方法和装置的一个目标。在一些实施方案中,形成沉淀物,其包括碳酸盐和/或碳酸氢盐材料。在一些实施方案中形成二阶阳离子碳酸盐和/或碳酸氢盐材料的沉淀物。在一些实施方案中,形成碳酸钙和/或碳酸氢钙材料。在一些实施方案中,形成碳酸镁和/或碳酸氢镁材料。在一些实施方案中,形成碳酸钙和碳酸镁和/或碳酸氢钙和碳酸氢镁材料。在一些实施方案中,形成了钙和镁的碳酸盐和/或碳酸氢盐,例如白云石。Optimization of precipitation reactions is a goal of efficient gas-liquid contacting methods and devices. In some embodiments, a precipitate is formed that includes carbonate and/or bicarbonate material. In some embodiments, a precipitate of secondary cationic carbonate and/or bicarbonate material is formed. In some embodiments, calcium carbonate and/or calcium bicarbonate material is formed. In some embodiments, magnesium carbonate and/or magnesium bicarbonate material is formed. In some embodiments, calcium carbonate and magnesium carbonate and/or calcium bicarbonate and magnesium bicarbonate materials are formed. In some embodiments, carbonates and/or bicarbonates of calcium and magnesium, such as dolomite, are formed.
在一些本文中介绍的实施方案中,由浆液形成物流和/或液滴,并随后在方法和装置中与气体接触。用于产生液滴和/或物流的浆液可取决于在其中气-液接触器所处位置的可得物以及将接触液滴的气体和由气体和液滴之间的相互作用的期望的结果。可以构成浆液的液体组分的液体包括但不局限于:包含二价阳离子的pH为10或更大的水溶液;海水;微咸水;来自脱盐工艺的人造液体废物;天然存在的盐水;工业废盐水;合成盐水;添加阳离子的溶液;添加添加二氧化硅的溶液;或其任何组合。所使用的浆液的组合物包括但不局限于液体和工业废物颗粒物,液体和矿物颗粒物,液体和沉淀物或其组合。如上所述,可以调节溶液化学以便增加将气体引入液体或浆液。在一些实施方案中,提供了包括水溶液的液滴和/或物流。在一些实施方案中,提供了包括pH为4-11的水溶液的液滴和/或物流。一些实施方案提供了包括含离子的水溶液的液滴和/或物流。一些实施方案提供了包括含二价阳离子的水溶液的液滴和/或物流。一些实施方案提供了包括水溶液的液滴和/或物流,所述水溶液包括海水,溶解矿物质溶液,盐水或其任何组合。在一些实施方案中,提供了包括碱性溶液的液滴。在一些实施方案中,提供了包括碱金属氢氧化物的液滴和/或物流。在一些实施方案中,提供了包括NaOH或KOH或其组合的液滴和/或物流。一些实施方案提供了包括固体材料的液滴和/或物流。在一些实施方案中,液滴和/或物流包括由液滴与气体接触产生的沉淀材料。在一些实施方案中,液滴和/或物流包括工业废物如但不限于飞灰、熔渣、水泥窑灰尘、矿业废物或其任何组合的颗粒物。在一些实施方案中,液滴和/或物流包括沉淀材料和工业废物颗粒物。在一些实施方案中,提供了包括矿物颗粒物的液滴和/或物流。在一些实施方案中,提供了包括矿物颗粒物和工业废物颗粒物的液滴和/或物流。在一些实施方案中,提供了包括矿物颗粒物、工业废物颗粒物和沉淀材料的液滴和/或物流。In some of the embodiments described herein, streams and/or droplets are formed from slurries and subsequently contacted with gases in methods and apparatus. The slurry used to generate the droplets and/or streams may depend on the availability of the location in which the gas-liquid contactor is located and the gas that will contact the droplets and the desired outcome from the interaction between the gas and the droplets . Liquids that may make up the liquid component of the slurry include, but are not limited to: aqueous solutions containing divalent cations at a pH of 10 or greater; seawater; brackish water; man-made liquid wastes from desalination processes; naturally occurring brines; industrial waste A brine; a synthetic brine; a cation-added solution; a silica-added solution; or any combination thereof. The composition of the slurry used includes, but is not limited to, liquid and industrial waste particles, liquid and mineral particles, liquid and sediment, or combinations thereof. As noted above, the solution chemistry can be adjusted to increase the introduction of gas into the liquid or slurry. In some embodiments, droplets and/or streams comprising aqueous solutions are provided. In some embodiments, droplets and/or streams comprising an aqueous solution having a pH of 4-11 are provided. Some embodiments provide droplets and/or streams comprising ion-containing aqueous solutions. Some embodiments provide droplets and/or streams comprising divalent cation-containing aqueous solutions. Some embodiments provide droplets and/or streams comprising aqueous solutions including seawater, dissolved mineral solutions, brine, or any combination thereof. In some embodiments, droplets comprising a basic solution are provided. In some embodiments, droplets and/or streams comprising alkali metal hydroxides are provided. In some embodiments, droplets and/or streams comprising NaOH or KOH or combinations thereof are provided. Some embodiments provide droplets and/or streams comprising solid material. In some embodiments, the droplets and/or the stream include precipitation material resulting from contacting the droplets with the gas. In some embodiments, the droplets and/or streams include particulate matter of industrial waste such as, but not limited to, fly ash, slag, cement kiln dust, mining waste, or any combination thereof. In some embodiments, the droplets and/or streams include precipitation material and industrial waste particulates. In some embodiments, droplets and/or streams comprising mineral particulates are provided. In some embodiments, droplets and/or streams comprising mineral particulates and industrial waste particulates are provided. In some embodiments, droplets and/or streams comprising mineral particulates, industrial waste particulates, and precipitation materials are provided.
存在许多制造液滴的方法。使用蒸发,例如蒸发水来润湿某个区域,能够产生液滴的非常细的烟。用于产生液滴的非蒸发的技术包括但不局限于:压力雾化器(喷口)、旋转雾化器、空气辅助雾化器、空气喷射雾化器、超声雾化器、喷墨雾化器、MEMS雾化器、喷射器-喷射喷口和静电喷雾器。在描述液体和/或浆液液滴的实施方案中,可以使用液滴形成的任何方法。在一些实施方案中,利用包括超声换能器的系统制造液滴。在一些实施方案中,利用包括压力雾化器(喷口)、旋转雾化器、空气辅助雾化器、空气喷射雾化器或其任何组合的系统制造液滴。在一些实施方案中,提供产生液滴的系统或设备(即液体引入单元),其包括超声换能器。在一些实施方案中,提供产生液滴的系统或设备,其包括喷口。在一些实施方案中,提供产生液滴的系统或设备,其包括双流体喷口,喷射器-喷射喷口,或两者。在一些实施方案中,产生液滴的系统和/或设备具有喷口,其具有5微米或更小的开口。在一些实施方案中,产生液滴的系统和/或设备具有喷口,其具有10微米或更小的开口。在一些实施方案中,产生液滴的系统和/或设备具有喷口,其具有10微米或更大的开口。在一些实施方案中,产生液滴的系统和/或设备具有喷口,其具有25微米或更大、如50微米或更大、100微米或更大、250微米或更大、500微米或更大的开口。在一些实施方案中,产生液滴的系统和/或设备具有喷口,其具有1毫米或更大的开口。在一些实施方案中,提供产生液滴的系统和/或设备,其包括压力雾化器(喷口)、旋转雾化器、空气辅助雾化器、空气喷射雾化器或其任何组合。一些实施方案提供了能够接受浆液以产生液滴的系统和/或装置。在一些实施方案中,提供了产生液滴的系统和/或装置,其中液滴直径为50至1000微米(μm)。在一些实施方案中,提供了产生液滴的系统和/或装置,其中液滴平均直径大于1000微米(μm)。在一些实施方案中,提供了产生液滴的系统和/或装置,其中液滴平均直径大于500微米。在一些实施方案中,提供了产生液滴的系统和/或装置,其中液滴平均直径小于1微米。在一些实施方案中,提供了产生液滴的系统和/或装置,其中液滴的平均直径大于5微米,如大于10微米、大于20微米、大于50微米、大于100微米、大于200微米、大于300微米、或者大于400微米。在一些实施方案中,提供了产生液滴的系统和/或装置,其中液滴的平均直径为1至400微米。在一些实施方案中,提供了产生液滴的系统和/或装置,其中液滴的平均直径为1至500微米。在一些实施方案中,提供了产生等浮液滴的系统和/或装置。等浮液滴是在其中它们是等浮的环境中既不上升也不下沉的液滴,例如围绕液滴的气体。There are many methods of making droplets. Using evaporation, such as evaporating water to moisten an area, can produce a very fine smoke of liquid droplets. Non-evaporative techniques for producing droplets include, but are not limited to: pressure atomizers (orifices), rotary atomizers, air-assisted atomizers, air-jet atomizers, ultrasonic atomizers, inkjet atomization nebulizers, MEMS nebulizers, injectors-injection nozzles and electrostatic nebulizers. In embodiments describing liquid and/or slurry droplets, any method of droplet formation may be used. In some embodiments, droplets are produced using a system that includes an ultrasonic transducer. In some embodiments, droplets are produced using a system comprising a pressure atomizer (orifice), a rotary atomizer, an air-assisted atomizer, an air-jet atomizer, or any combination thereof. In some embodiments, a system or apparatus for generating liquid droplets (ie, a liquid introduction unit) comprising an ultrasonic transducer is provided. In some embodiments, a system or apparatus for generating droplets is provided that includes an orifice. In some embodiments, a system or apparatus for generating droplets is provided that includes a dual-fluid jet, an ejector-jet jet, or both. In some embodiments, a system and/or device for generating droplets has an orifice with an opening of 5 microns or less. In some embodiments, a system and/or device for generating droplets has an orifice with an opening of 10 microns or less. In some embodiments, a system and/or device for generating droplets has an orifice with an opening of 10 microns or greater. In some embodiments, the system and/or device for generating droplets has an orifice with a diameter of 25 microns or greater, such as 50 microns or greater, 100 microns or greater, 250 microns or greater, 500 microns or greater opening. In some embodiments, a system and/or device for generating droplets has an orifice with an opening of 1 mm or greater. In some embodiments, a system and/or apparatus for generating droplets comprising a pressure atomizer (orifice), a rotary atomizer, an air-assisted atomizer, an air-jet atomizer, or any combination thereof is provided. Some embodiments provide systems and/or devices capable of receiving a slurry to produce droplets. In some embodiments, systems and/or devices for generating liquid droplets are provided, wherein the liquid droplets are 50 to 1000 micrometers (μm) in diameter. In some embodiments, systems and/or devices for generating droplets are provided, wherein the droplets have an average diameter greater than 1000 micrometers (μm). In some embodiments, systems and/or devices for generating droplets are provided, wherein the droplets have an average diameter greater than 500 microns. In some embodiments, systems and/or devices for generating droplets are provided, wherein the droplets have an average diameter of less than 1 micron. In some embodiments, systems and/or devices for generating droplets are provided, wherein the droplets have an average diameter greater than 5 microns, such as greater than 10 microns, greater than 20 microns, greater than 50 microns, greater than 100 microns, greater than 200 microns, greater than 300 microns, or greater than 400 microns. In some embodiments, systems and/or devices for generating liquid droplets are provided, wherein the liquid droplets have an average diameter of 1 to 400 microns. In some embodiments, systems and/or devices for generating liquid droplets are provided, wherein the liquid droplets have an average diameter of 1 to 500 microns. In some embodiments, systems and/or devices for generating equi-buoyant droplets are provided. Isobuoyant droplets are droplets that neither rise nor sink in an environment in which they are buoyant, such as a gas surrounding the droplet.
也许可能的是通过改变使用的产生液滴的技术的类型最优化液滴所覆盖的装置的接触室或塔的横截面的面积。特别地,也许可能的是通过利用不同角度的喷雾器最优化液滴所覆盖的装置的横截面的面积。在一些实施方案中,靠近接触室的壁,使用60°的喷雾器,而在接触室的内横截面中使用90°的喷雾器。It may be possible to optimize the area of the cross-section of the device's contact chamber or column covered by the droplets by varying the type of droplet generation technique used. In particular, it may be possible to optimize the area of the cross-section of the device covered by the droplets by using sprayers of different angles. In some embodiments, a 60° nebulizer is used near the wall of the contacting chamber, while a 90° nebulizer is used in the inner cross-section of the contacting chamber.
与气体接触的液体的表面积与体积比是一个支配气体引入液体的效率的因素。在一些实施方案中,提供液滴和/或液体物流,其表面积与体积比(SA∶V)为12m2/升或更大。在一些实施方案中,提供液滴和/或液体物流,其表面积与体积比(SA∶V)为24m2/升或更大,如60m2/升或更大,80m2/升或更大,120m2/升或更大,600m2/升或更大,或6000m2/升或更大。在一些实施方案中,提供液滴,其平均直径大于500微米。在一些实施方案中,提供液滴,其平均直径小于1微米。在一些实施方案中,提供液滴,其平均直径大于5微米,如大于10微米、大于20微米、大于50微米、大于100微米、大于200微米、大于300微米、或者大于400微米。在一些实施方案中,提供液滴,其平均直径1至400微米。在一些实施方案中,提供液滴,其平均直径1至500微米。在一些实施方案中,提供液滴,其直径为50至1000微米(μm)。在一些实施方案中,提供液滴,其平均直径为100微米(μm)。在一些实施方案中,提供了产生和/或利用等浮液滴的方法。等浮液滴是在其中它们是等浮的环境中既不上升也不下沉的液滴,例如围绕液滴的气体。The surface area to volume ratio of the liquid in contact with the gas is a factor governing the efficiency with which the gas is introduced into the liquid. In some embodiments, liquid droplets and/or liquid streams are provided having a surface area to volume ratio (SA:V) of 12 m2/liter or greater. In some embodiments, liquid droplets and/or liquid streams are provided having a surface area to volume ratio (SA:V) of 24 m2/liter or greater, such as 60 m2/liter or greater, 80 m2/liter or greater, 120 m2/liter liter or greater, 600m2/liter or greater, or 6000m2/liter or greater. In some embodiments, droplets are provided having an average diameter greater than 500 microns. In some embodiments, droplets are provided having an average diameter of less than 1 micron. In some embodiments, droplets are provided having an average diameter greater than 5 microns, such as greater than 10 microns, greater than 20 microns, greater than 50 microns, greater than 100 microns, greater than 200 microns, greater than 300 microns, or greater than 400 microns. In some embodiments, droplets are provided having an average diameter of 1 to 400 microns. In some embodiments, droplets are provided having an average diameter of 1 to 500 microns. In some embodiments, droplets are provided that are 50 to 1000 micrometers (μm) in diameter. In some embodiments, droplets are provided having an average diameter of 100 micrometers (μm). In some embodiments, methods of generating and/or utilizing equibuantial droplets are provided. Isobuoyant droplets are droplets that neither rise nor sink in an environment in which they are buoyant, such as a gas surrounding the droplet.
在本发明的一些实施方案中,指出了特定的L/G比。L/G比是装置中的液体的流速与气体的流速的比值的数值。不束缚于理论学说,气体或气体的组分吸收到液体中与L/G比成正比。在该比值的一些运用中,L/G比是无量纲数,并且因而将表示相对于另一每单位时间体积流量的每单位时间体积流量,或者相对于另一每单位时间质量流量的每单位时间质量流量。在一些实施方案中,使用的L/G比为3-50。在一些实施方案中,使用的L/G比为3-20。在一些实施方案中,使用的L/G比是15。在一些实施方案中,使用的L/G比为50-4000。在一些实施方案中,使用的L/G比为100-1000。在一些实施方案中,使用的L/G比为200-400。在一些实施方案中,使用的L/G比是300。在一些实施方案中,装置和/或系统被配置以使用3-50的L/G比。在一些实施方案中,装置和/或系统被配置以使用3-20的L/G。在一些实施方案中,装置和/或系统被配置以使用15的L/G比。在一些实施方案中,装置和/或系统被配置以使用50-4000的L/G比。在一些实施方案中,装置和/或系统被配置以使用100-1000的L/G比。在一些实施方案中,装置和/或系统被配置以使用200-400的L/G比。在一些实施方案中,装置和/或系统被配置以使用300的L/G比。在某些L/G比的运用中,引用了单位以便标识在该比值中比较了体积或质量。在一些实施方案中,使用的L/G比为0-10,000加仑/分/1000实际立方英尺。在一些实施方案中,使用的L/G比为50-5,000加仑/分/1000实际立方英尺。在一些实施方案中,使用的L/G比为100-500加仑/分/1000实际立方英尺。在一些实施方案中,装置和/或系统被配置以在0-10,000加仑/分/1000实际立方英尺的液体流量与气体流量比(L/G比)下操作。在一些实施方案中,装置和/或系统被配置以在50-5,000加仑/分/1000实际立方英尺的液体流量与气体流量比(L/G比)下操作。在一些实施方案中,装置和/或系统被配置以在100-500加仑/分/1000实际立方英尺的液体流量与气体流量比(L/G比)下操作。In some embodiments of the invention, a specific L/G ratio is indicated. The L/G ratio is a numerical value of the ratio of the flow rate of liquid to the flow rate of gas in the device. Without being bound by theory, the absorption of a gas or a component of a gas into a liquid is directly proportional to the L/G ratio. In some uses of this ratio, the L/G ratio is a dimensionless number and will thus represent volume flow per unit of time relative to another volume flow per unit of time, or per unit of mass flow per unit of time relative to another Time mass flow. In some embodiments, an L/G ratio of 3-50 is used. In some embodiments, an L/G ratio of 3-20 is used. In some embodiments, a L/G ratio of 15 is used. In some embodiments, an L/G ratio of 50-4000 is used. In some embodiments, an L/G ratio of 100-1000 is used. In some embodiments, an L/G ratio of 200-400 is used. In some embodiments, a L/G ratio of 300 is used. In some embodiments, the device and/or system is configured to use an L/G ratio of 3-50. In some embodiments, the device and/or system is configured to use an L/G of 3-20. In some embodiments, the device and/or system is configured to use an L/G ratio of 15. In some embodiments, the device and/or system is configured to use an L/G ratio of 50-4000. In some embodiments, the device and/or system is configured to use an L/G ratio of 100-1000. In some embodiments, the device and/or system is configured to use an L/G ratio of 200-400. In some embodiments, the device and/or system is configured to use an L/G ratio of 300. In certain uses of the L/G ratio, units are quoted to indicate that volume or mass is being compared in the ratio. In some embodiments, a L/G ratio of 0-10,000 gallons per minute per 1000 actual cubic feet is used. In some embodiments, a L/G ratio of 50-5,000 gallons per minute per 1000 actual cubic feet is used. In some embodiments, a L/G ratio of 100-500 gallons per minute per 1000 actual cubic feet is used. In some embodiments, the device and/or system is configured to operate at a liquid flow to gas flow ratio (L/G ratio) of 0-10,000 gallons per minute per 1000 actual cubic feet. In some embodiments, the device and/or system is configured to operate at a liquid flow to gas flow ratio (L/G ratio) of 50-5,000 gallons per minute per 1000 actual cubic feet. In some embodiments, the device and/or system is configured to operate at a liquid flow to gas flow ratio (L/G ratio) of 100-500 gallons per minute per 1000 actual cubic feet.
存在着这样的结构特征,其可以被添加到其中液体或浆液和气体接触的室,其提高了气体引入液体或浆液。这样的结构特征意味着增加了气体当其流过液体或浆液时之间的相互作用,这是通过以下实现的:1)提供了一个或多个用于液体/气体系统达到平衡的区域,或者2)提供液体或浆液被气体夹带并且遵循更多旋转路径的机会,当液体或浆液通过室的空间时(例如,液体或浆液落下时)。在一些实施方案中,任一类型的结构特征用于接触吸收溶液,接触混合物,或浆液与气体。在一些实施方案中,装置或者系统包括任一类型的结构特征用于接触吸收溶液,接触混合物,或浆液与气体。在一些实施方案中,或者两种类型的结构特征的组合用于接触吸收溶液,接触混合物,或浆液与气体。在一些实施方案中,装置或系统包括两种类型的结构特征用于接触吸收溶液,接触混合物,或浆液与气体。There are structural features that can be added to the chamber where the liquid or slurry and gas are in contact that enhance the introduction of the gas into the liquid or slurry. Such structural features imply increased interaction between gases as they flow through liquids or slurries by: 1) providing one or more regions for the liquid/gas system to reach equilibrium, or 2) Provides an opportunity for the liquid or slurry to be entrained by the gas and to follow more rotational paths as the liquid or slurry passes through the space of the chamber (eg, as the liquid or slurry falls). In some embodiments, either type of structural feature is used to contact an absorption solution, a contact mixture, or a slurry and a gas. In some embodiments, the device or system includes any type of structural feature for contacting an absorption solution, contacting a mixture, or a slurry with a gas. In some embodiments, or a combination of both types of structural features are used for contacting absorption solutions, contacting mixtures, or slurries and gases. In some embodiments, a device or system includes two types of structural features for contacting an absorption solution, contacting a mixture, or a slurry and a gas.
在有限空间中增加液体和气体达到平衡的概率的结构特征包括填料材料和塔盘。填料材料可以是结构化或非结构化的,可以是陶瓷,塑料,或金属,球状的,蜂窝状的,带状的,鞍状的,环状的或随机形状形式。塔盘一般地是具有穿孔或开口的金属片,其允许气体和液体流过塔盘。制造填料材料和塔盘的材料基于塔或反应器中预期的气体、液体和产物的化学性质以及诸如物理耗损等考虑因素选择。利用填料材料或塔盘的塔或反应器可以被认为若干级的集合(即塔或反应器的长度),其中离开该级的气体与离开该级的液体平衡。理论级描述了塔的物理区段,其中本体蒸相和本体液相可以被近似为处于平衡,相对于当气体和液体穿过穿过塔时交换的物质的浓度来说。通过调节填料材料或塔盘的配置结构,可以调节体积效率。利用填料材料或塔盘的塔或反应器特征为在塔或反应器的总长度上高压降,这应归于与塔盘有关的液体压头或与填料材料有关的床压降。利用填料材料的塔通常不适于接触浆液与气体,但是填料材料已经被制成对于适应浆液来说是工业可行的。在一些实施方案中,塔盘,填料材料,或两者用于接触吸收溶液,接触混合物,或浆液与气体。在一些实施方案中,装置或系统包括填料材料,塔盘或其组合以接触吸收溶液,接触混合物,或浆液与气体。Structural features that increase the probability of liquid and gas reaching equilibrium in a confined space include packing materials and trays. Packing materials can be structured or unstructured, and can be ceramic, plastic, or metallic, in spherical, honeycomb, ribbon, saddle, ring, or random shape form. Trays are generally sheets of metal with perforations or openings that allow gases and liquids to flow through the trays. The materials from which the packing materials and trays are made are selected based on the chemical properties of the gases, liquids and products expected in the column or reactor, as well as considerations such as physical losses. A column or reactor utilizing packing material or trays can be considered as a collection of stages (ie the length of the column or reactor) where the gas leaving the stage is in equilibrium with the liquid leaving the stage. The theoretical level describes the physical section of the column in which the bulk vapor phase and bulk liquid phase can be approximated to be in equilibrium with respect to the concentrations of species exchanged as gas and liquid pass through the column. Volumetric efficiency can be adjusted by adjusting the packing material or the configuration of the trays. Columns or reactors utilizing packing materials or trays are characterized by a high pressure drop over the entire length of the column or reactor due to liquid head associated with the trays or bed pressure drop associated with the packing material. Columns utilizing packing materials are generally not suitable for contacting slurries and gases, but packing materials have been made commercially viable for accommodating slurries. In some embodiments, trays, packing material, or both are used to contact the absorbing solution, the contacting mixture, or the slurry with the gas. In some embodiments, the device or system includes packing material, trays, or a combination thereof to contact the absorbing solution, contact the mixture, or the slurry and the gas.
膜是另一种类型的结构特征,其可以包括在塔或反应器中以便促进在气体和液体之间的相互作用。在塔或反应器中使用的膜被用于增加液体和气体之间的界面面积以便最大化质量传递率。膜可由许多中空纤维组成。在这样的膜中,在每一个中空纤维孔的入口处出现气液界面。因此,这种类型的膜可被称为微孔膜。在一些实施方案中,使用一种或多种膜以便接触吸收溶液,接触混合物,或浆液与气体。在一些实施方案中,装置或系统包括一种或多种膜以便接触吸收溶液,接触混合物,或浆液与气体。在一些实施方案中,使用与填料材料和/或塔盘结合的一种或多种膜以便接触吸收溶液,接触混合物,或浆液与气体。在一些实施方案中,装置或系统包括与填料材料和/或塔盘结合的一种或多种膜以便接触吸收溶液,接触混合物,或浆液与气体。在一些实施方案中,使用一种或多种微孔膜以便接触吸收溶液,接触混合物,或浆液与气体。在一些实施方案中,装置或系统包括一种或多种微孔膜以便接触吸收溶液,接触混合物,或浆液与气体。在一些实施方案中,使用与填料材料和/或塔盘结合的一种或多种微孔膜以便接触吸收溶液,接触混合物,或浆液与气体。在一些实施方案中,装置或系统包括与填料材料和/或塔盘结合的一种或多种微孔膜以便接触吸收溶液,接触混合物,或浆液与气体。Membranes are another type of structural feature that may be included in a column or reactor to facilitate the interaction between gas and liquid. Membranes used in columns or reactors are used to increase the interfacial area between liquid and gas in order to maximize mass transfer rates. Membranes can be composed of many hollow fibers. In such membranes, a gas-liquid interface occurs at the entrance of each hollow fiber pore. Therefore, this type of membrane may be referred to as a microporous membrane. In some embodiments, one or more membranes are used for contacting the absorbing solution, the contacting mixture, or the slurry and the gas. In some embodiments, a device or system includes one or more membranes for contacting an absorbing solution, a contacting mixture, or a slurry and a gas. In some embodiments, one or more membranes are used in combination with packing material and/or trays for contacting the absorbing solution, the contacting mixture, or the slurry and the gas. In some embodiments, the device or system includes one or more membranes combined with packing material and/or trays for contacting the absorbing solution, contacting the mixture, or the slurry and the gas. In some embodiments, one or more microporous membranes are used for contacting the absorbing solution, the contacting mixture, or the slurry and the gas. In some embodiments, a device or system includes one or more microporous membranes for contacting an absorbing solution, a contacting mixture, or a slurry and a gas. In some embodiments, one or more microporous membranes are used in combination with packing material and/or trays for contacting the absorbing solution, the contacting mixture, or the slurry and the gas. In some embodiments, the device or system includes one or more microporous membranes combined with packing material and/or trays for contacting the absorbing solution, contacting the mixture, or the slurry and the gas.
结构特征,其在气体通过塔或室时允许液体或浆液变成夹带在气体中,同时促使气体和液体遵循更多旋转路径,包括棚排。棚排是具有更大的长比宽的材料板阵列,一般地为金属,在中间弯曲而形成倒置的“v”形态,其由环担载。一般地,设置棚排使得棚(shed)的位置在塔或反应器中与每一个台面(level)交替。因此,从棚落下的混合物(例如浆液)或液体将到达在其之下的棚顶附近,使得混合物或液体经塔或反应器的长度沿着更长的更多旋转路径向下,相比于其在没有棚排的情况下。棚排还影响了气体通过塔或反应器的流动。在棚顶和棚底之间的空间中,其之上是其中可能发生落下的混合物或液体夹带到气体中的地方。这种夹带增加了混合物或液体和气体必须在塔或反应器中相互作用的时间量。棚排与液体和浆液是相容的。休止角,其支配棚的宽窄,允许使用具有各种溶液的棚排并且用于适应固体颗粒物。可以调节棚的浸渍以便允许液体或浆液以允许在塔或反应器中与气体进行期望的相互作用的速度从棚顶滚下。棚排还允许在塔或反应器的高度上气体压力的较低下降,相比于填充以塔盘或填料材料的那些来说。在一些实施方案中,棚排存在于本发明的装置或系统中。在一些实施方案中,其中吸收溶液和气体接触的室包括棚排。在一些实施方案中,其中使接触混合物和气体接触的室包括棚排。在一些实施方案中,装置包括接触室,其包括棚排。在一些实施方案中,系统包括一种装置,所述装置包括棚排。在一些实施方案中,棚排包括同心圆的棚。在一些实施方案中,室包括沿室壁进入室中心的凸起。在这样的实施方案中,凸起类似于向下倾斜的棚的一半。Structural features that allow liquids or slurries to become entrained in the gas as the gas passes through the tower or chamber while forcing the gas and liquid to follow more rotational paths, including shed rows. A row is an array of sheets of material with greater length than width, typically metal, bent in the middle to form an inverted "v" configuration, carried by a ring. Generally, the rows of sheds are arranged such that the position of the sheds alternates with each level in the column or reactor. Thus, a mixture (such as a slurry) or liquid falling from the shed will reach near the roof of the shed below it, causing the mixture or liquid to follow a longer, more swirling path down the length of the column or reactor than It is in the case of no shed row. Rows also affect the flow of gas through the column or reactor. In the space between the roof and the bottom of the shed, above which is where entrainment of falling mixtures or liquids into the gas may occur. This entrainment increases the amount of time the mixture or liquid and gas must interact in the column or reactor. Rows are compatible with liquids and slurries. The angle of repose, which governs the width of the shed, allows the use of shed rows with various solutions and is used to accommodate solid particles. The impregnation of the shed can be adjusted to allow the liquid or slurry to roll off the roof at a velocity that allows the desired interaction with the gas in the column or reactor. Rows also allow for a lower drop in gas pressure over the height of the column or reactor compared to those packed with trays or packing material. In some embodiments, a row is present in a device or system of the invention. In some embodiments, the chamber in which the absorption solution contacts the gas comprises a row. In some embodiments, the chamber in which the contacting mixture and the gas are contacted comprises a row. In some embodiments, the apparatus includes a contacting chamber that includes rows. In some embodiments, a system includes an apparatus that includes a row. In some embodiments, the row of sheds comprises concentric circles of sheds. In some embodiments, the chamber includes a protrusion along the chamber wall into the center of the chamber. In such an embodiment, the protrusion resembles one half of a downwardly sloping shed.
存在着这样的实施方案,其中令人期望的是在与气体接触后,冷凝或凝聚液滴或细颗粒物材料。在这样的实施方案中,可以使用各种方法以引起液滴聚结,包括但不限于:温度变化,使用具有另外的表面积的元件,使用静电沉降器,或使用物流、板、或较大液滴的液体以便在接触气体后夹带液滴。在一些实施方案中,沉淀物在液滴中形成,同时液滴仍然与气体接触。在一些实施方案中,在沉淀物在液滴中形成同时仍然与气体接触的情况下,使用静电沉降器来收集和凝聚液滴,具有液滴中的沉淀物的电荷。在一些实施方案中,细颗粒物形成,并且使用静电沉降器收集这些颗粒。There are embodiments where it is desirable to condense or condense liquid droplets or fine particulate material after contact with the gas. In such embodiments, various methods can be used to induce coalescence of the droplets, including but not limited to: temperature changes, use of elements with additional surface area, use of electrostatic precipitators, or use of streams, plates, or larger liquids. Droplets of liquid to entrain droplets after contact with gas. In some embodiments, a precipitate forms in the droplets while the droplets are still in contact with the gas. In some embodiments, an electrostatic precipitator is used to collect and condense the droplets, having the charge of the precipitates in the droplets, with the precipitate forming in the droplets while still in contact with the gas. In some embodiments, fine particulate matter is formed, and electrostatic precipitators are used to collect these particles.
在一些实施方案中,必需使落下的液体或浆液重新定向,所述落下的液体或浆液以液滴、物流或板的形式沿装置高度下降,使得液体或浆液没有将过度的能量给予液体或浆液的储存器和产生泡沫。特别地,在一些实施方案中令人期望的是避免形成稳定泡沫层,其中稳定泡沫层可损害泵最佳工作的能力。可以使用结构元件来重新定向落下的液体或浆液。在一些实施方案中,倒置的锥体存在于装置中以便减轻起泡。除去泡沫层的传统方法是机械破碎构成泡沫层的气泡。在一些实施方案中,使用喷雾器来机械地破碎气泡并且减轻起泡。在一些实施方案中,使用倒置的锥体和喷雾器来减轻起泡。In some embodiments, it is necessary to redirect the falling liquid or slurry that descends the height of the device in the form of droplets, streams, or plates so that the liquid or slurry does not impart undue energy to the liquid or slurry reservoir and generate foam. In particular, it is desirable in some embodiments to avoid the formation of a stable foam layer that could impair the ability of the pump to work optimally. Structural elements may be used to redirect falling liquid or slurry. In some embodiments, inverted cones are present in the device to reduce foaming. The traditional method of removing the foam layer is to mechanically break up the air bubbles that make up the foam layer. In some embodiments, a nebulizer is used to mechanically break up air bubbles and reduce foaming. In some embodiments, an inverted cone and sprayer is used to reduce foaming.
在一些实施方案中,用于将气体组分转移(即结合)到液体中的本发明的装置包括气体入口,被配置以使液体和气体接触的室;在室中第一位置的第一液体引入单元和在室中第二位置的第二液体引入单元,用于接触气体;被配置以在液体已经接触气体后包含所述液体的储存器;用于液体的在其已经接触气体后的出口,和以下流动特征中的至少一个:i)在室中的至少一个阵列的棚排;ii)消泡设备;iii)每一液体引入单元至少一个泵,用于将液体泵送通过该引入单元;iv)配置液体引入单元,使得液体离开第一单元的流动方向不同于液体离开第二单元的流动方向;v)一个或多个节流孔机构排泄阀),其被配置以将液体流动引导到液体引入单元中的至少一个、到气体入口中、或其组合;和vi)改变液体引入单元所覆盖的区域。在这样的实施方案中,液体引入单元可以是喷雾器,液体柱,液体平喷射器,或其组合。在这样的实施方案中,棚排可以被配置以当气体进入室时重新分布其流动,使得在与棚排相互作用前,气体沿室轴向流动,从而与气体在进入室时流动相比,覆盖室横截面的更大面积。在一些实施方案中,消泡设备可以包括位于储存器以上的锥体(例如倒置的锥体)。在这样的实施方案中,也可使用面向锥体的喷雾器。在一些实施方案中,其中改变由液体引入单元覆盖的区域,可以使用变化角度的喷雾器。在一些实施方案中,用于将液体泵送过液体引入单元的泵是变频机泵。In some embodiments, a device of the invention for transferring (i.e. combining) a gas component into a liquid comprises a gas inlet, a chamber configured to contact the liquid and the gas; a first liquid at a first position in the chamber An introduction unit and a second liquid introduction unit at a second location in the chamber for contacting the gas; a reservoir configured to contain the liquid after it has contacted the gas; an outlet for the liquid after it has contacted the gas , and at least one of the following flow characteristics: i) at least one array of rows in the chamber; ii) defoaming equipment; iii) at least one pump per liquid introduction unit for pumping liquid through the introduction unit iv) configuring the liquid introduction unit so that the direction of flow of liquid leaving the first unit is different from the direction of flow of liquid leaving the second unit; v) one or more orifice mechanism drain valves) configured to direct the flow of liquid into at least one of the liquid introduction unit, into the gas inlet, or a combination thereof; and vi) changing the area covered by the liquid introduction unit. In such embodiments, the liquid introduction unit may be a sprayer, a liquid column, a liquid flat sparger, or a combination thereof. In such embodiments, the rows may be configured to redistribute the flow of the gas as it enters the chamber such that before interacting with the rows, the gas flows axially along the chamber, thereby Covers a larger area of the chamber cross-section. In some embodiments, the defoaming device can include a cone (eg, an inverted cone) positioned above the reservoir. In such embodiments, cone-facing nebulizers may also be used. In some embodiments, where the area covered by the liquid introduction unit is varied, varying angle sprayers may be used. In some embodiments, the pump used to pump the liquid through the liquid introduction unit is an inverter pump.
用于使气体物流和水接触的示范性的方法和系统使用接触气体物流至水的平喷物流。因此,在一些实施方案中,本发明提供用于分离二氧化碳和工业废物物流的装置,其包括气-液接触器,被配置以使液体,例如,水的平喷物流接触全部或者部分工业废物物流从而溶解CO2和,在一些实施方案中,工业废物物流的其他组分,并且进一步包括可操作地连接到气-液接触器用于例如通过蒸发、沉淀等产生包括在接触器中溶于水的全部或者部分CO2的固体组合物的单元。在一些实施方案中,固体组合物是包括碳酸氢盐的组合物。在一些实施方案中,固体组合物是包括碳酸盐的组合物。在一些实施方案中,固体组合物包括碳酸盐和碳酸氢盐。在一些实施方案中,固体组合物包括碳酸盐和/或碳酸氢盐以及一种或多种工业废气的其它组分,例如SOx或SOx衍生物,NOx或NOx衍生物,重金属或其衍生物,颗粒物,VOC或VOC衍生物,或其组合。Exemplary methods and systems for contacting a gas stream and water use a flat jet stream of contacting gas stream to water. Accordingly, in some embodiments, the present invention provides apparatus for separating carbon dioxide and an industrial waste stream comprising a gas-liquid contactor configured to contact all or a portion of the industrial waste stream with a flat jet stream of liquid, e.g., water thereby dissolving CO and, in some embodiments, other components of the industrial waste stream, and further comprising operably connected to a gas-liquid contactor for producing, for example, by evaporation, precipitation, etc. A unit of solid composition of all or part of CO2 . In some embodiments, the solid composition is a composition comprising bicarbonate. In some embodiments, the solid composition is a composition comprising carbonate. In some embodiments, the solid composition includes carbonates and bicarbonates. In some embodiments, the solid composition includes carbonates and/or bicarbonates and one or more other components of industrial waste gases, such as SOx or SOx derivatives, NOx or NOx derivatives, heavy metals or derivatives thereof , particulate matter, VOC or VOC derivatives, or combinations thereof.
美国专利7,379,487描述了示范性的平喷物流/气体接触系统,其公开内容在本文中引入作为参考。在一些实施方案中,接触包括CO2的气体物流包括使用两相反应器,如美国专利7,379,487中所述。平喷物流方法和系统的简要说明在下文给出。An exemplary flat jet flow/gas contacting system is described in US Patent 7,379,487, the disclosure of which is incorporated herein by reference. In some embodiments, contacting a gas stream comprising CO 2 includes using a two-phase reactor, as described in US Pat. No. 7,379,487. A brief description of the flat jet flow method and system is given below.
在许多气液接触系统中,气体传输至液相的速率是由液相传质系数k,界面表面积A和本体流体和气-液界面之间的浓度梯度ΔC控制的。气体吸收入液体中的速率的实际形式然后是:In many gas-liquid contact systems, the rate of gas transport to the liquid phase is controlled by the liquid phase mass transfer coefficient k, the interfacial surface area A, and the concentration gradient ΔC between the bulk fluid and the gas-liquid interface. The actual form of the rate of gas absorption into a liquid is then:
其中Ф是每一单位体积的反应器的气体吸收速率(摩尔/cm3·s),a是每一单位界面面积的平均吸收速率(摩尔/cm2·s)。a是每一单位体积的气液界面面积(cm2/cm3,或cm-1),p和pi分别是本体气体中的和在界面处的试剂气体的分压(巴),CL*是将与现有气相浓度,pi相平衡的液体侧浓度(摩尔/cm3),和CL(摩尔/cm3)是本体液体中溶解气体的平均浓度。kG和kL分别是气体侧和液体侧传质系数(cm/s)。Where Ф is the gas absorption rate per unit volume of the reactor (mol/cm3·s), and a is the average absorption rate per unit interface area (mol/cm2·s). a is the gas-liquid interface area (cm2/cm3, or cm-1) per unit volume, p and pi are respectively the partial pressure (bar) of the reagent gas in the bulk gas and at the interface, and CL* is to be compared with Existing gas phase concentration, pi phase equilibrium liquid side concentration (mol/cm3), and CL (mol/cm3) are the average concentrations of dissolved gases in the bulk liquid. kG and kL are the gas side and liquid side mass transfer coefficients (cm/s), respectively.
在许多气液反应系统中,CL*的溶解度是非常低的并且因此限制了浓度梯度的控制。因此,在设计有效的气-液流动反应器中考虑的主要参数是质量传递和界面表面积与反应器体积比,其亦称为比表面积。In many gas-liquid reaction systems, the solubility of CL* is very low and thus limits the control of concentration gradients. Therefore, the main parameters considered in designing an efficient gas-liquid flow reactor are mass transfer and the ratio of interfacial surface area to reactor volume, also known as specific surface area.
在某些实施方案中,本发明的平喷气液接触系统是气液接触系统,其使用了平喷的提高的比表面积从而改善了含CO2的气体物流与水(例如含碱土金属离子的水)之间的相互作用。在某些实施方案中,使用了含多个孔口的刚性喷口板,其产生了非常薄的平喷。平喷孔口在一种配置结构中具有V形室,其连接到水源,例如含碱土金属离子的水。平喷孔口可以具有一对相对的平面壁,其连接到V形室的顶部。平喷喷口可以具有锥形喷口,其连接到V形室的相对平面壁的相对端。在另一配置结构中,喷射孔口可以具有连接到液体源室的圆形孔口。平喷喷口可以具有V形的凹槽,与圆形孔口相交而产生椭圆形的孔口。平喷孔口可以是与含CO2的气体物流的入口源垂直,相对或平行取向的。平喷喷口的最小通道可以是大于600微米。喷口可以产生液体平喷,其具有为其厚度至少10倍、或至少8倍、至少6倍、或至少4倍的宽度。平喷可以被制得薄达10微米,例如,10-15微米,10-20微米,或10-40微米,并且分开达小于1.1,1.0,0.9,0.8,0.7,0.6,0.5,0.4,0.3,或0.2mm,例如小于1毫米,以便产生高填料喷射密度(β=0.010.01))和大比表面积,a=10-20cm-1。薄的喷射允许更多的水,例如,含碱土金属离子的水,经受含CO2的气体物流,相比于常规的接触器产生更高收率的反应产物(每单元液体质量流量),例如更多转移CO2和/或其它气体物流的组分,如SOx、NOx、重金属、颗粒物,VOC,和其衍生物和组合至液体,例如,水。In certain embodiments, the horizontal jet gas-liquid contacting system of the present invention is a gas-liquid contacting system that utilizes the increased specific surface area of the flat jet to improve the interaction of the CO2 -containing gas stream with water (e.g., alkaline earth metal ion-containing water). )interaction between. In certain embodiments, a rigid orifice plate with multiple orifices is used, which produces a very thin flat jet. The flat jet orifice has a V-shaped chamber in one configuration that is connected to a water source, such as water containing alkaline earth metal ions. The flat jet orifice may have a pair of opposing planar walls connected to the top of the V-shaped chamber. The flat jet nozzle may have a tapered nozzle connected to opposite ends of the opposite planar walls of the V-shaped chamber. In another arrangement, the spray orifice may have a circular orifice connected to the liquid source chamber. The flat jet nozzle may have a V-shaped groove that intersects a circular orifice to create an oval orifice. The flat jet orifice may be oriented perpendicular, opposite or parallel to the inlet source of the CO2 -containing gas stream. The smallest channel of a flat jet nozzle can be greater than 600 microns. The spout may produce a flat jet of liquid having a width that is at least 10 times, or at least 8 times, at least 6 times, or at least 4 times its thickness. Flat jets can be made as thin as 10 microns, for example, 10-15 microns, 10-20 microns, or 10-40 microns, and separated by less than 1.1, 1.0, 0.9, 0.8, 0.7, 0.6, 0.5, 0.4, 0.3 , or 0.2mm, such as less than 1 mm, in order to produce high filler injection density (β = 0.010.01)) and large specific surface area, a = 10-20cm-1. The thin jet allows more water, e.g., alkaline earth metal ion-containing water, to be subjected to the CO2 -containing gas stream, resulting in a higher yield of reaction product (per unit liquid mass flow rate) than conventional contactors, e.g. More transfer of CO2 and/or other components of gas streams, such as SOx, NOx, heavy metals, particulate matter, VOC, and their derivatives and combination to liquids, eg, water.
本发明一个实施方案将提供气液固接触器,其包括CO2加料反应器,具有多个薄的平喷物流,其密切且均匀间隔,具有高比表面积,具有均匀的喷射速度,空气动力学成型从而最小化液体喷射的气体流动干扰,没有盐堵塞和阻塞的孔口,以及在同向流动、反向流动和平行流动的气体工艺物流中操作。在一个实施方案中,喷射以与气体错流配置结构操作,例如,喷射从喷口垂直向下,而气体越过喷射水平地或近似水平地流动。One embodiment of the present invention will provide a gas-liquid-solid contactor comprising a CO2 fed reactor with multiple thin flat jet streams that are closely and evenly spaced, have a high specific surface area, have uniform jet velocity, aerodynamic Shaped to minimize gas flow disturbances for liquid jets, free from salt plugging and clogged orifices, and operate in co-flow, counter-flow and parallel-flow gas process streams. In one embodiment, the jet operates in a cross-flow configuration with the gas, eg, the jet is vertically downward from the jet while the gas flows horizontally or approximately horizontally across the jet.
使用本发明的平喷气液接触系统来使含CO2的气体物流与液体接触,例如水如含碱土金属离子的水。加料反应器包括室,其具有用于含CO2的气体物流的入口源和用于液体,例如水如含碱土金属离子的水的源头的平喷喷口。喷口可以具有众多孔口,其最小尺寸为长度大于200、300、400、500或600微米,例如长度大于600微米,并且产生高比表面积的薄的平喷。喷口可以具有一对平行相对的板,其具有连接到锥形喷口的第二端。喷口可以具有一对V形板,其连接到一对平行相对的板的第一端。液体,例如水如含碱土金属的水,孔口可以产生水的平喷,其宽度为其厚度的至少5、6、7、8、9、10、11、12、13、14或15倍,例如至少10倍。液体,例如水如含碱土金属的水的来源可以是任何适宜的来源(如上所述)。在某些实施方案中,水源包括具有用于水例如含碱土金属离子的水的输入端如来自海洋的管子或管道的储存器。在液体(例如水如含碱土金属离子的水)来源是海水的情况下,输入端与海水来源流体连通,如其中输入端是至陆地基系统的来自海洋水的管路或进料。The flat-spray gas-liquid contacting system of the present invention is used to contact a CO2 -containing gas stream with a liquid, such as water, such as water containing alkaline earth metal ions. The feed reactor comprises a chamber with an inlet source for a CO2 -containing gas stream and a flat jet orifice for a source of a liquid, eg water such as water containing alkaline earth metal ions. The jets may have a plurality of orifices with a minimum dimension of greater than 200, 300, 400, 500 or 600 microns in length, for example greater than 600 microns in length, and produce a thin flat jet of high specific surface area. The spout may have a pair of parallel opposing plates having a second end connected to the tapered spout. The spout may have a pair of V-shaped plates connected to first ends of a pair of parallel opposing plates. a liquid, such as water such as alkaline earth metal containing water, the orifice may produce a flat jet of water having a width at least 5, 6, 7, 8, 9, 10, 11, 12, 13, 14 or 15 times its thickness, For example at least 10 times. The source of liquid, for example water, such as alkaline earth metal-containing water, may be any suitable source (as described above). In certain embodiments, the water source comprises a reservoir having an input for water, eg, alkaline earth metal ion-containing water, such as a pipe or pipeline from the ocean. Where the source of liquid (eg, water such as alkaline earth metal ion-containing water) is seawater, the input is in fluid communication with the source of seawater, such as where the input is a line or feed from ocean water to a land-based system.
气液接触系统可以包括小部分的孔口阵列。孔口可以是交错的,使得喷射孔口分开达在一个方向中1.5-2.5cm,例如,1.7-2.3cm,例如1.9-2.1cm,或2cm,在另一方向中,1.5-2.5mm,例如,1.7-2.3mm,例如1.9-2.1mm,或2mm,和对角地0.5-1.5mm,例如,0.7-1.3mm,例如0.9-1.1mm,或1mm。孔口具有V形入口和圆锥形出口通道,以便形成喷射。入口和出口通道的交点产生了孔口。喷射长度与喷射宽度比值可以是5∶1至20∶1,或7∶1至15∶1,或8∶1至12∶1,或9∶1至11∶1,或10∶1,厚度为10-100微米,例如,10-20微米。A gas-liquid contacting system may include a small number of orifice arrays. The orifices may be staggered such that the jet orifices are separated by 1.5-2.5 cm, for example, 1.7-2.3 cm, such as 1.9-2.1 cm, or 2 cm in one direction, and 1.5-2.5 mm in the other direction, for example , 1.7-2.3mm, such as 1.9-2.1mm, or 2mm, and diagonally 0.5-1.5mm, such as 0.7-1.3mm, such as 0.9-1.1mm, or 1mm. The orifice has a V-shaped inlet and a conical outlet channel to form a jet. The intersection of the inlet and outlet channels creates the orifice. The ratio of spray length to spray width can be 5:1 to 20:1, or 7:1 to 15:1, or 8:1 to 12:1, or 9:1 to 11:1, or 10:1, with a thickness of 10-100 microns, eg, 10-20 microns.
本发明的加料反应器可以包括平喷气液接触系统以产生大比面积的水(例如,含碱土金属离子的水)。加料反应器具有含CO2的气体物流来源(气体反应物),其连接到反应器的歧管。在某些实施方案中,该歧管具有许多孔(开口),其允许CO2-气体物流喷射进入加料反应器。加料反应器也可以具有用于水(例如,水如含碱土金属离子的水)(“液体反应物”)的入口,其由至多个平喷喷口的管道系统连接,所述平喷喷口产生了水的平物流。水(例如,含碱土金属离子的水)的平物流与含CO2的气体物流相互作用以产生CO2加料的水组合物,其将经受沉淀条件。The feed reactor of the present invention may include a flat sparged air-liquid contacting system to produce large specific area water (eg, alkaline earth metal ion-containing water). The feed reactor has a source of CO 2 -containing gas stream (gas reactant), which is connected to the manifold of the reactor. In certain embodiments, the manifold has a number of holes (openings) that allow injection of the CO2 -gas stream into the feed reactor. The feed reactor may also have an inlet for water (e.g., water such as alkaline earth metal ion-containing water) (the "liquid reactant") connected by piping to a plurality of flat jet nozzles that produce Flat logistics of water. A flat stream of water (eg, alkaline earth metal ion-containing water) interacts with a CO2 -containing gas stream to produce a CO2 - fed water composition that will be subjected to precipitation conditions.
在一些实施方案中,加料反应器的平喷气液接触系统的喷口可以具有V形室,其在顶部连接到一对相对板的第一端。相对板的第二端连接到锥形喷口。水(例如,含碱土金属离子的水)流入V形室并且被强迫通过相对板之间的通道和离开喷口和产生平喷。取决于喷口面积,喷射流速和速度,喷射厚度为大约5至100微米和宽度为约1至5厘米。结果,宽度与厚度(之比)可以明显大于一个数量级。对于大约10m/s的喷射速度来说,平喷物流的长度可以是15厘米或更多。其中锥形喷口遇到相对的平面板的最窄通道是大于600微米的。这种喷口允许较大表面积的水(例如,含碱土金属离子的水),其显著地增加了CO2及其他组分(例如,SOx、NOx、颗粒物,重金属和/或VOCs)在...含CO2的气体物流和液膜之间的转移效率。进一步,由于大喷射表面积和小喷射厚度,这种喷口产生了非常大的比表面积,10-20cm-1,这能够实现水和气体物流之间的更高体积的接触。除增加水(例如,含碱土金属离子的水)的表面积之外,平喷射器具有600微米或更大的喉道大小。平喷射器的大喉道尺寸使得它们将阻塞变得不太可能。在一些实施方案中,其中阻塞不是太大问题,喉道尺寸可以较小,例如50微米或更大,100微米或更大,200微米或更大,300微米或更大,400微米或更大,或500微米或更大。该系统也可包括用于收集水(例如,含碱土金属离子的水)和CO2-贫乏的气体物流的组件,以便再利用。In some embodiments, the jet of the flat jet gas-liquid contacting system of the addition reactor may have a V-shaped chamber connected at the top to the first ends of a pair of opposing plates. The second end of the opposite plate is connected to the conical spout. Water (eg, water containing alkaline earth metal ions) flows into the V-shaped chamber and is forced through the channel between the opposing plates and exits the spout and creates a flat spray. Depending on the orifice area, jet flow rate and velocity, the jet thickness is about 5 to 100 microns and the width is about 1 to 5 centimeters. As a result, the (ratio) of width to thickness can be significantly greater than an order of magnitude. For a jet velocity of about 10 m/s, the length of the flat jet stream can be 15 cm or more. The narrowest channel where the conical orifice meets the opposing planar plate is greater than 600 microns. Such vents allow a large surface area of water (e.g., water containing alkaline earth metal ions), which significantly increases CO2 and other components (e.g., SOx, NOx, particulate matter, heavy metals, and/or VOCs) in the... Transfer efficiency between a CO2 -containing gas stream and a liquid film. Further, due to the large jet surface area and small jet thickness, this jet orifice produces a very large specific surface area, 10-20 cm-1, which enables higher volume contact between water and gas streams. In addition to increasing the surface area of water (eg, alkaline earth metal ion-containing water), flat injectors have throat sizes of 600 microns or greater. The large throat size of flat injectors makes it less likely that they will clog. In some embodiments, where clogging is not too much of an issue, the throat size can be smaller, such as 50 microns or greater, 100 microns or greater, 200 microns or greater, 300 microns or greater, 400 microns or greater , or 500 µm or larger. The system may also include components for collecting water (eg, alkaline earth metal ion-containing water) and CO2 -lean gas streams for reuse.
通过将气体导向以接触水的平喷物流,含CO2的气体物流接触水(例如,含碱土金属离子的水)。含CO2的气体物流可以以相对平喷物流的几乎任何方向流动,包括与平喷物流成直角,与平喷物流平行,与平喷物流逆平行,或与平喷物流成某一角度。The CO 2 -containing gas stream contacts water (eg, alkaline earth metal ion-containing water) by directing the gas to contact a flat jet stream of water. The CO2 -containing gas stream can flow in almost any direction relative to the flat jet stream, including at right angles to the flat jet stream, parallel to the flat jet stream, anti-parallel to the flat jet stream, or at an angle to the flat jet stream.
如上所述,来自工业设备30的气体可以被加工,然后用于加料水。例如,气体可以经受氧化条件以改善气体物流的组分的溶解度(例如,为将CO转化为CO2,NO转化为NO2,和SO2转化为SO3等)。Gas from
图11显示了本发明的装置的实施方案。包括液体和固体组分的浆液进入接触室,通过入口管道[100]至储存器[105],其还包含已经接触气体的浆液。在一些实施方案中,螺旋输送机提供了浆液与气体的粉碎和混合,当其进入室[110]时。在一些实施方案中,气体通过入口[110]进入室。在一些实施方案中,在液滴或物流产生系统的阵列中,存在着至少两个水平或段的液滴或物流产生[150和155]以及用于气体在室中向上移动的管道[160]。浆液运输系统[115,120,135,125,140,130]将浆液从储存器移动到液滴或物流产生系统以及在不同水平的液滴或物流产生中再循环浆液。粉碎系统[120,125,130]提供了浆液的固体组分的粒径减小,由此改善了固体在气体引入液体中的参与度。在一些实施方案中,粉碎系统是浆液运输系统的管道[115,145]中的螺旋输送机。在一些实施方案中,使用高效率的气-液接触器而从气体物流中除去更多的期望组分。没有固体的清液被提供至高效率的气-液接触器[165]并且非常细的液滴或者薄的液膜或其它高表面积/体积比手段被用于接触液体和气体。在一些实施方案中,需要冷凝器[170]以便使由高效率的气-液接触器产生的液滴和/或颗粒物落至储存器。在接触液体后的气体离开室[175]并且通过另一系统或者释放到空气中。接触后的液体,包括来自气体和液体之间的接触的沉淀材料以及极小量的浆液的固体组分离开储存器[180]并且被通到其它系统[185]如沉淀槽,脱水系统,和建筑制造系统。Figure 11 shows an embodiment of the device of the present invention. A slurry comprising liquid and solid components enters the contacting chamber through an inlet duct [100] to a reservoir [105] which also contains the slurry which has been contacted with gas. In some embodiments, a screw conveyor provides comminution and mixing of the slurry and gas as it enters the chamber [110]. In some embodiments, gas enters the chamber through inlet [110]. In some embodiments, in an array of droplet or stream generation systems, there are at least two levels or segments of droplet or stream generation [150 and 155] and conduits for gas movement up the chamber [160] . Slurry transport systems [115, 120, 135, 125, 140, 130] move slurry from storage to droplet or stream generation systems and recycle slurry at different levels of droplet or stream generation. Comminution systems [120, 125, 130] provide particle size reduction of the solid components of the slurry, thereby improving the participation of solids in the gas-introduced liquid. In some embodiments, the comminution system is a screw conveyor in the pipeline [115, 145] of the slurry transport system. In some embodiments, a high efficiency gas-liquid contactor is used to remove more of the desired component from the gas stream. The clear liquid free of solids is provided to a high efficiency gas-liquid contactor [165] and very fine droplets or thin liquid films or other high surface area/volume ratio means are used to contact liquid and gas. In some embodiments, a condenser [170] is required to allow the liquid droplets and/or particulates produced by the high efficiency gas-liquid contactor to fall to the reservoir. The gas after contact with the liquid leaves the chamber [175] and passes through another system or is released into the air. The contacted liquid, including the precipitation material from the contact between the gas and the liquid and the solid components of a very small amount of the slurry, leaves the storage [180] and is passed to other systems [185] such as settling tanks, dewatering systems, and Building manufacturing system.
图12显示了本发明的方法的实施方案的流程图。在大多数实施方案中提供了包括液体和固体组分的浆液[200]。浆液经历粉碎以减小固体组分的粒径,同样混合浆液[205]。由浆液形成液滴或物流[210],液滴或物流形式的液体被引入[215]到用于接触的室[220]。气体被引入接触室[225]。气体的来源可以是工业烟道气,如来自燃烧化石燃料如煤的那种。接触后,形成所接触的浆液并且在接触室[230]中在储存器中收集并且这种接触过的浆液的一部分在粉碎后被再循环[235]并且一部分被抽走用于固体和液体的分离[240]。固体被除去并且可被用于许多应用,包括但不限于建筑材料,作为土体稳定,油漆制造,和润滑剂制造。排出物液体[245]可以具有有利于用于高效率的气-液接触器[255]的溶液化学,所述高效率的气-液接触器[255]位于装置中以便最大化与气体的接触[250],当其通过较大的液滴和物流至接触的末级时。排出物液体还可能使用纳滤和反渗透[260]进行加工来提供矿物和化学品再生以及水,后者可能适合作为用于脱盐的进料水或适用于释放入环境[265]。Figure 12 shows a flow diagram of an embodiment of the method of the present invention. In most embodiments a slurry comprising liquid and solid components is provided [200]. The slurry undergoes crushing to reduce the particle size of the solid components, and the slurry is also mixed [205]. From the slurry, droplets or streams are formed [210] and the liquid in the form of droplets or streams is introduced [215] into a chamber for contacting [220]. The gas is introduced into the contact chamber [225]. The source of the gas may be industrial flue gas, such as that from burning fossil fuels such as coal. After contacting, a contacted slurry is formed and collected in a reservoir in the contacting chamber [230] and a part of this contacted slurry is recycled [235] after crushing and a part is pumped away for solid and liquid Separation [240]. The solids are removed and can be used in many applications including, but not limited to, construction materials, as soil stabilization, paint manufacturing, and lubricant manufacturing. The effluent liquid [245] may have a solution chemistry favorable for use in a high efficiency gas-liquid contactor [255] located in the plant to maximize contact with the gas [250], as it passes through larger droplets and streams to the final stage of contact. Effluent liquids may also be processed using nanofiltration and reverse osmosis [260] to provide mineral and chemical regeneration as well as water, which may be suitable as feed water for desalination or for release into the environment [265].
图13显示了本发明的装置的水平配置的实施方案。气体通过入口[300]进入室。包括液体和固体组分的浆液由储存器输送,其中浆液经受粉碎[330]并且通过液滴产生设备的阵列[350]进入接触室[310]以产生填充该室的液滴喷雾[320]。在一些实施方案中,存在着至少两段的液滴产生,其可操作地连接,使得气体沿所述段的长度移动并且在接触室的长度上变得贫乏气体组分。浆液运输系统[340,350,360,370]将浆液从接触室移动到液滴产生系统以及在不同段的液滴或物流产生中再循环浆液。浆液储存器[330]中的粉碎系统提供了浆液的固体组分的粒径减小,由此改善了固体在气体引入液体中的参与度。在接触液体后的气体离开室并且通过另一系统或者释放到空气中。接触后的液体,包括来自气体和液体之间的接触的沉淀材料以及极小量的浆液的固体组分离开接触室[370]并且被通到其它系统[380]如沉淀槽,脱水系统,和建筑制造系统。Figure 13 shows an embodiment of a horizontal configuration of the device of the present invention. Gas enters the chamber through the inlet [300]. A slurry comprising liquid and solid components is conveyed from a reservoir where the slurry is subjected to comminution [330] and passed through an array of droplet generating devices [350] into a contact chamber [310] to produce a spray of droplets [320] that fills the chamber. In some embodiments, there are at least two segments of droplet generation that are operatively connected such that the gas travels along the length of the segments and becomes depleted of gas components over the length of the contacting chamber. The slurry transport system [340, 350, 360, 370] moves the slurry from the contact chamber to the droplet generation system and recirculates the slurry in different stages of droplet or stream generation. The pulverization system in the slurry reservoir [330] provides particle size reduction of the solid components of the slurry, thereby improving the participation of solids in the gas-introduced liquid. The gas after contact with the liquid leaves the chamber and passes through another system or is released into the air. The contacted liquid, including the precipitation material from the contact between the gas and the liquid and the solid components of a very small amount of the slurry, exits the contact chamber [370] and is passed to other systems [380] such as settling tanks, dewatering systems, and Building manufacturing system.
图14显示了本发明的装置的水平配置的实施方案(来自端视图)。包括液体和固体组分的浆液由储存器输送,其中浆液经受粉碎[400]并且通过液滴产生设备的阵列[430]进入接触室[410]以产生填充该室的液滴喷雾[440]。气体通过入口进入室并且通过该室[450]。在一些实施方案中,存在着至少两段的液滴产生,其可操作地连接,使得气体沿所述段的长度移动并且在接触室的长度上变得贫乏气体组分。浆液运输系统[420,460,470]将浆液从接触室移动到液滴产生系统以及在不同段的液滴或物流产生中再循环浆液。浆液运输系统[460]中的粉碎系统提供了浆液的固体组分的粒径减小,由此改善了固体在气体引入液体中的参与度。在接触液体后的气体离开室并且通过另一系统或者释放到空气中。接触后的液体,包括来自气体和液体之间的接触的沉淀材料以及极小量的浆液的固体组分离开接触室并且被通到其它系统[480]如沉淀槽,脱水系统,和建筑制造系统。Figure 14 shows an embodiment of the device of the invention in a horizontal configuration (from an end view). A slurry comprising liquid and solid components is conveyed from a reservoir where the slurry is subjected to comminution [400] and passed through an array of droplet generating devices [430] into a contact chamber [410] to generate a spray of droplets [440] that fills the chamber. Gas enters the chamber through the inlet and passes through the chamber [450]. In some embodiments, there are at least two segments of droplet generation that are operatively connected such that the gas travels along the length of the segments and becomes depleted of gas components over the length of the contacting chamber. The slurry transport system [420, 460, 470] moves the slurry from the contact chamber to the droplet generation system and recirculates the slurry in the different stages of droplet or stream generation. The pulverization system in the slurry delivery system [460] provides particle size reduction of the solid components of the slurry, thereby improving the participation of solids in the gas-introduced liquid. The gas after contact with the liquid leaves the chamber and passes through another system or is released into the air. The contacted liquid, including the precipitated material from the contact between the gas and the liquid and the solid components of a very small amount of the slurry, exits the contact chamber and is passed to other systems [480] such as settling tanks, dewatering systems, and building fabrication systems .
图15显示了本发明的实施方案,其包括气体分布段,可变数目的接触段,和脱烟或最终的气体吸收段,其总是位于装置的顶部。气体在最下段,气体分布段,在收集的溶液以上,进入装置。气体向上流过装置并且离开装置的顶部。接触段的数目可以为1至多于一个,如2或3或更多。接触段的数目将是基于期望的最终产品的类型,所使用的吸收溶液或接触混合物,喷雾(或其他形式的液体物流),和任选地,所使用的棚排,来确定。临在气体离开装置前的段是除雾段。该段可以包括高效率的气体吸收系统,其例如使用清液的平膜或非常细的液滴,约小于100μm,如小于50μm直径液滴。在最终的段中,没有颗粒物的溶液将用于最大化吸收和/或液滴收集。用于除雾和最终的气体吸收段的溶液的化学性质可以不同于任何接触段中使用的溶液。在装置底部收集的溶液的再循环可以包括可在溶液中的任何固体的粉碎。再循环也可包括从溶液中分离任何可存在的固体并且将排出物返回到该装置,有或者没有处理以便补充溶液中的任何化学试剂不足。装置本质上可以是轻便的,使得整个装置包含在运输容器中,其可以通过铁路(火车),水路(驳船)和/或公路(卡车)来运输的。装置也可是模块化的,使得不同的段可以各自包含在运输容器中并且堆叠或以其它方式可操作地彼此连接。Figure 15 shows an embodiment of the invention comprising a gas distribution section, a variable number of contacting sections, and a defuming or final gas absorption section, always located at the top of the apparatus. The gas is in the lowest section, the gas distribution section, above the collected solution, into the unit. Gas flows up through the device and exits the top of the device. The number of contact segments may be 1 to more than one, such as 2 or 3 or more. The number of contacting stages will be determined based on the type of end product desired, the absorption solution or contacting mixture used, the spray (or other form of liquid stream), and optionally, the row used. The section just before the gas leaves the device is the demister section. This section may comprise a high efficiency gas absorption system, eg using a flat membrane of clear liquid or very fine droplets, about less than 100 μm, such as less than 50 μm diameter droplets. In the final stage, a particulate-free solution will be used to maximize absorption and/or droplet collection. The chemical nature of the solutions used in the demisting and final gas absorption stages may be different from the solutions used in any of the contacting stages. Recirculation of the solution collected at the bottom of the device may include comminution of any solids that may be in solution. Recycling may also include separating any solids that may be present from the solution and returning the effluent to the plant, with or without treatment to replenish any chemical deficits in the solution. The device may be portable in nature such that the entire device is contained in a transport container which may be transported by rail (train), water (barge) and/or road (truck). The device may also be modular such that different segments may each be contained within a shipping container and stacked or otherwise operably connected to each other.
图16和17是其中使用棚排的本发明的装置的实施方案的示意图。图16显示了整个装置,在装置的较低部分以上具有气体入口,其中所收集的溶液将被存储,然后取出至进一步的加工。中心段包括棚排,其配置结构表示在图17中。图16中显示的装置的上段包括喷雾器以便将吸收溶液或接触混合物引入装置和气体出口。Figures 16 and 17 are schematic illustrations of embodiments of the apparatus of the invention in which rows are used. Figure 16 shows the entire device, with a gas inlet above the lower part of the device, where the collected solution will be stored and then withdrawn for further processing. The central section includes rows of sheds, the configuration of which is shown in Figure 17. The upper section of the device shown in Figure 16 includes nebulizers to introduce the absorption solution or contact mixture into the device and a gas outlet.
图18是本发明的实施方案的示意图,其中装置包括喷雾器的阵列。显示了4个喷雾器;在垂直取向的装置中喷雾器全部向下导向。喷雾器可以以任何适宜方式实现,包括使用喷射器或喷射器-喷射喷口,双流体喷口,压力雾化器,旋转雾化器,空气辅助雾化器,空气喷射雾化器或超声雾化器或其任何组合。装置中气体的流动是向上的,以便全部喷雾器以逆流方式遇到气体流。气体在底部进入装置并且沿装置长度向上流动。装置可包括除雾段,其位于气体出口之前(未示)。吸收溶液或接触混合物可以是清液,或者其可以是浆液,包含固体组分如矿物,工业废物(例如飞灰,水泥窑灰尘)和/或沉淀材料,如果使用再循环的话。如果使用再循环,在吸收溶液中任何固体的粉碎可以发生。吸收溶液或接触混合物可以包括工业废盐水,天然存在的碱性盐水,海水,人工组成或合成盐水,或其任何组合。Figure 18 is a schematic illustration of an embodiment of the invention wherein the device comprises an array of nebulizers. 4 nebulizers are shown; the nebulizers are all directed downwards in the vertically oriented device. Nebulization can be accomplished in any suitable manner, including using an injector or injector-jet nozzle, a two-fluid nozzle, a pressure atomizer, a rotary atomizer, an air-assisted atomizer, an air-jet atomizer or an ultrasonic atomizer or any combination thereof. The flow of gas in the device is upward so that all nebulizers encounter the gas flow in a countercurrent fashion. Gas enters the device at the bottom and flows up the length of the device. The device may include a mist removal section before the gas outlet (not shown). The absorption solution or contacting mixture may be a clear liquid, or it may be a slurry, containing solid components such as minerals, industrial waste (eg fly ash, cement kiln dust) and/or precipitation material, if recycling is used. If recirculation is used, comminution of any solids in the absorption solution may occur. The absorption solution or contacting mixture may include industrial waste brine, naturally occurring alkaline brine, seawater, artificially composed or synthetic brine, or any combination thereof.
图18是本发明的实施方案的示意图,其中装置包括喷雾器的阵列。显示了4个喷雾器,两个中心喷雾器是任选的。顶端喷雾器是这样取向的,使得液体(即吸收溶液或接触混合物)相对于气体流动,逆流流动。最下层的喷雾器是这样取向的,使得液体与气体流动顺流流动(沿垂直取向的装置中心向上)。气体在底部进入装置并且沿装置长度向上流动。装置可以包括棚排,代替装置中心的两个任选的喷雾器。装置可包括除雾段,其位于气体出口之前(未示)。吸收溶液可以是清液,或者其可以是浆液,包含固体组分如矿物,工业废物(例如飞灰,水泥窑灰尘)和/或沉淀材料,如果使用再循环的话。如果使用再循环,在吸收溶液中任何固体的粉碎可以发生。Figure 18 is a schematic illustration of an embodiment of the invention wherein the device comprises an array of nebulizers. 4 sprayers are shown, the two center sprayers are optional. The tip sprayer is oriented such that the liquid (ie, absorbing solution or contact mixture) flows countercurrently to the gas flow. The lowermost nebulizers were oriented such that the liquid flowed co-currently with the gas flow (upward in the center of the vertically oriented device). Gas enters the device at the bottom and flows up the length of the device. The device may include rows of sheds in place of the two optional sprayers in the center of the device. The device may include a mist removal section before the gas outlet (not shown). The absorption solution may be a clear liquid, or it may be a slurry, containing solid components such as minerals, industrial waste (eg fly ash, cement kiln dust) and/or sedimentation material, if recycling is used. If recirculation is used, comminution of any solids in the absorption solution may occur.
图20显示了棚排配置结构的示意图。在左手侧显示了配置结构的自顶向下或平面视图。在右手侧显示了棚排配置结构的侧截面。最顶部配置结构是由环担载的圆柱形室或塔中的棚排的标准配置结构。棚是交错的,使得从上棚落下的液体将到达下面的棚的顶部附近。在侧面,其中不存在完整的棚,表示了向下倾斜的插入物。中间的配置结构是制成同心环的棚的配置结构。这些棚环由在棚下的线担载,从室或塔的一侧到另一侧。棚是交错的,此外,使得从上棚落下的液体将到达下面的棚的顶部附近。棚排的最下层的配置结构被配置使得代替全部的棚彼此平行地通过室或塔的整个高度排列,所有其他水平的棚相对于以上和以下的排90°取向。Figure 20 shows a schematic diagram of the shed row configuration. A top-down or plan view of the configuration is shown on the left-hand side. On the right-hand side a side section of the row configuration is shown. The topmost configuration is the standard configuration of rows of sheds in cylindrical chambers or towers carried by rings. The sheds are staggered so that liquid falling from the upper shed will reach near the top of the lower shed. On the sides, where no full shed exists, downward-sloping inserts are indicated. The configuration in the middle is the configuration of sheds made into concentric rings. These shed rings are carried by wires under the shed, from one side of the chamber or tower to the other. The sheds are staggered, moreover, so that liquid falling from the upper shed will reach near the top of the lower shed. The arrangement of the lowest level of the rows of sheds is configured such that instead of all sheds being aligned parallel to each other through the entire height of the chamber or tower, all other horizontal sheds are oriented at 90° relative to the rows above and below.
另一方面的高度有效的气液接触是液体和气体接触的时间,以便气体可以引入到液体中。在液体塔中的气泡或者由气体围绕的液滴的情况,这种接触时间还可称为停留时间。液滴在气体中的停留时间可以通过产生等浮液滴而增加,如上所述。在气体中液滴的停留时间还可以通过增加液滴在与气体接触时所沿着的路径长度而增加。在一些实施方案中,路径长度是由装置的配置结构提供的。一些实施方案利用了液滴在接触室中的对流或运动,从而提高了与气体接触的液滴的路径长度,和因此其停留时间。在一些实施方案中,通过连接室的管道提供了路径长度,其中由其中凝聚液滴的室产生了液滴。一些实施方案提供了液滴以沿长路径接触气体。在一些实施方案中,长路径是螺旋路径,如图21A-22B中的]]。在一些实施方案中,气体以渐开线进料的方式被引入装置[图21A:400,图21B:500,图22A:600,图22B:700],使得气体和与气体接触的液滴在装置的接触室中沿着螺旋状路径[图21A:410,图21B:510,图22A:610,图22B:710]。一些实施方案利用气体-入口的阵列[图21A:470,图21B:540,图22A:670,图22B:740]以提高液滴在气体中(例如在主接触室中)的停留时间。Another aspect of highly efficient gas-liquid contact is the time the liquid and gas are in contact so that the gas can be introduced into the liquid. In the case of gas bubbles in a liquid column or liquid droplets surrounded by gas, this contact time may also be referred to as residence time. The residence time of the droplets in the gas can be increased by creating equi-buoyant droplets, as described above. The residence time of a droplet in a gas can also be increased by increasing the length of the path that the droplet follows when it comes into contact with the gas. In some embodiments, the path length is provided by the configuration of the device. Some embodiments take advantage of the convection or motion of the droplets in the contacting chamber, thereby increasing the path length, and thus the residence time, of the droplets in contact with the gas. In some embodiments, the path length is provided by tubing connecting the chambers in which the droplets are generated from the chambers in which they condense. Some embodiments provide liquid droplets to contact gas along long paths. In some embodiments, the long path is a helical path, such as ]] in Figures 21A-22B. In some embodiments, the gas is introduced into the device as an involute feed [Fig. 21A: 400, Fig. 21B: 500, Fig. 22A: 600, Fig. 22B: 700] such that the gas and the liquid droplets in contact with the gas are The contact chamber of the device follows a helical path [Fig. 21A: 410, Fig. 21B: 510, Fig. 22A: 610, Fig. 22B: 710]. Some embodiments utilize an array of gas-inlets [Fig. 21A: 470, Fig. 21B: 540, Fig. 22A: 670, Fig. 22B: 740] to increase the residence time of the droplets in the gas (eg, in the main contacting chamber).
含二阶阳离子源的溶液和含CO2的气源可以可操作地连接到吸收器(例如,气-液接触器),CO2吸收器,如本文中所述。吸收器可以包括许多不同设计,包括,例如,图23A和23B的气-液接触器321。如图23B所示,气-液接触器可以配置以底部中的入口(310),通过其二价阳离子的水溶液可以引入气-液接触器。气-液接触器也可包括集管或歧管320(图23A,图23B),含CO2的气体(330i)可以引入其中。图23B提供了涡流(即,紊流混合)的示意图,其可以在含二阶阳离子的溶液中产生,此时含CO2的气体通过喷口322(例如,可调喷口)。尽管其未举例说明,气-液接触器可包括许多另外的可调喷口,沿着气液接触器的长度。另外的可调喷口可以用来保持涡流,特别地在如图23A中举例说明的系统中。气-液接触器也可被配置以具有圆锥形形状,该形状促进保持和/或提高在集管或歧管320中产生的涡流。在这样的实施方案中,另外的可调喷口可以仍然是期望的,特别地当期望提高混合时。A solution containing a source of secondary cations and a gas source containing CO can be operably connected to an absorber (eg, a gas-liquid contactor), a CO absorber, as described herein. The absorber can include many different designs including, for example, the gas-
气液接触器可以包括室,其包括多个可调喷口,其中喷口被配置以喷射包括二价阳离子的水溶液到含CO2的气体的(例如,来自燃煤的发电厂的烟道气)的气氛的室中。如图24所示,喷口410(例如,普通孔口压力雾化器等等)被配置以相抵于锯齿状表面420(即,包括许多凸块的表面)引导含二阶阳离子的水溶液使得含二阶阳离子的溶液被分散到水溶液的雾滴中,有效地增加进来的含二阶阳离子的溶液的表面积和最优化含二阶阳离子的溶液与含CO2的气体的相互作用。在这样的实施方案中,喷口被配置以在低压力操作,该低压操作最小化该方法的能量需要。在一些实施方案中,喷口在小于15psi,50psi,100psi,200psi,400psi,800psi,或1000psi的压力下操作。在一些实施方案中,喷口被配置以具有至少100微米,200微米,300微米,400微米,500微米,750微米,或1000微米的孔口尺寸。在这样的压力,喷口,与锯齿状的表面一起,能够产生含二阶阳离子的溶液的液滴,其中液滴具有小于0.5微米,1微米,10微米,15微米,30微米,60微米,125微米,250微米,500微米,1000微米,2000微米,或4000微米的平均直径。A gas-liquid contactor may include a chamber that includes a plurality of adjustable jets, wherein the jets are configured to inject an aqueous solution including divalent cations into a CO2 -containing gas (e.g., flue gas from a coal-fired power plant) atmosphere in the room. As shown in FIG. 24 , an orifice 410 (e.g., a common orifice pressure atomizer, etc.) is configured to direct an aqueous solution containing secondary cations against a serrated surface 420 (i.e., a surface comprising many bumps) such that the secondary cation-containing A solution of secondary cations is dispersed into droplets of aqueous solution, effectively increasing the surface area of the incoming secondary cation-containing solution and optimizing the interaction of the secondary cation-containing solution with the CO2- containing gas. In such embodiments, the jets are configured to operate at low pressures that minimize the energy requirements of the process. In some embodiments, the nozzle operates at a pressure of less than 15 psi, 50 psi, 100 psi, 200 psi, 400 psi, 800 psi, or 1000 psi. In some embodiments, the orifice is configured to have an orifice size of at least 100 microns, 200 microns, 300 microns, 400 microns, 500 microns, 750 microns, or 1000 microns. At such pressures, the orifice, together with the serrated surface, is capable of producing droplets of a solution containing secondary cations, where the droplets have diameters smaller than 0.5 micron, 1 micron, 10 micron, 15 micron, 30 micron, 60 micron, 125 Micron, 250 micron, 500 micron, 1000 micron, 2000 micron, or 4000 micron average diameter.
可以有效地增加气体和液体之间的接触时间的另一方法是将一个在另一个中再循环。在一些实施方案中,已经接触气体的液滴凝聚到液体溶液中,由其中产生液滴并且再次与气体接触。在一些实施方案中,已经接触液滴的气体通过气体-入口的阵列返回到接触室或装置[图21A:470,图21B:540,图22A:670,图22B:740]。在一些实施方案中,气体和液体都被再循环。在其中提供方法和装置以通过增加气体中液滴的路径长度而提高气体和液体之间的接触时间的实施方案中,液滴的尺寸可以为平均直径100微米至1毫米或更大,如平均直径200微米至1毫米、平均直径300微米至900微米、平均直径400微米至900微米、平均直径400微米至800微米、平均直径500微米至900微米、平均直径500微米至800微米、平均直径500微米至700微米、或者平均直径400微米至700微米。在一些实施方案中,提供了平均直径为500微米至大于1毫米的液滴。Another way that can effectively increase the contact time between gas and liquid is to recirculate one within the other. In some embodiments, droplets that have been exposed to a gas coalesce into a liquid solution from which droplets are generated and again contacted with a gas. In some embodiments, the gas that has contacted the droplets is returned to the contacting chamber or device through an array of gas-inlets [Figure 21A: 470, Figure 21B: 540, Figure 22A: 670, Figure 22B: 740]. In some embodiments, both gas and liquid are recycled. In embodiments where methods and apparatus are provided to increase the contact time between a gas and a liquid by increasing the path length of a droplet in a gas, the size of the droplets may range from 100 microns in average diameter to 1 mm or greater, such as the average 200 microns to 1 mm in diameter, 300 microns to 900 microns in average diameter, 400 microns to 900 microns in average diameter, 400 microns to 800 microns in average diameter, 500 microns to 900 microns in average diameter, 500 microns to 800 microns in average diameter, 500 microns in average diameter microns to 700 microns, or an average diameter of 400 microns to 700 microns. In some embodiments, droplets having an average diameter of 500 microns to greater than 1 mm are provided.
一些实施方案利用了在一个接触室中的延长的路径长度,再循环和高表面面积技术。在一些实施方案中,用于制造液滴的液体是浆液并且包含固体材料。在这样的实施方案中,具有减缩尺寸的固体组分的各相的粉碎混合和再循环一起使用。在一些实施方案中,装置包括多个液滴形成级,其中一些利用粉碎与再循环并且其中一些利用高表面面积技术,其中在暴露于气体前液体不包含固体材料。在一些实施方案中,该方法包括以使用粉碎的方式混合固体与液体;使固体和液体混合物,或浆液,与气体接触;和分离固体与接触后的液体和气体。在一些实施方案中,浆液被再循环并且与气体接触数次。在一些实施方案中,在浆液已经接触气体后与固体分离的液体被再循环。Some embodiments take advantage of extended path length, recirculation and high surface area techniques in one contacting chamber. In some embodiments, the liquid used to make the droplets is a slurry and contains solid material. In such embodiments comminutive mixing and recycling of the phases with size-reduced solid components are used together. In some embodiments, the apparatus includes multiple droplet formation stages, some of which utilize comminution and recycling and some of which utilize high surface area technology where the liquid does not contain solid material prior to exposure to the gas. In some embodiments, the method includes mixing a solid and a liquid using pulverization; contacting the solid and liquid mixture, or slurry, with a gas; and separating the solid from the contacted liquid and gas. In some embodiments, the slurry is recycled and contacted with gas several times. In some embodiments, the liquid separated from the solids after the slurry has been exposed to the gas is recycled.
在一些实施方案中,本发明的装置和系统被配置以结合大于20吨/小时的二氧化碳到吸收溶液中,在平均72小时的连续操作下。在一些实施方案中,本发明的装置和系统被配置以结合大于40吨/小时的二氧化碳到吸收溶液中。在一些实施方案中,本发明的装置和系统被配置以结合大于60吨/小时的二氧化碳到吸收溶液中。在一些实施方案中,本发明的装置和系统被配置以结合大于70吨/小时的二氧化碳到吸收溶液中。在一些实施方案中,本发明的装置和系统被配置以结合大于80吨/小时的二氧化碳到吸收溶液中。在一些实施方案中,本发明的装置和系统被配置以结合大于90吨/小时的二氧化碳到吸收溶液中。在一些实施方案中,本发明的装置和系统被配置以结合大于100吨/小时的二氧化碳到吸收溶液中。在一些实施方案中,本发明的装置和系统被配置以结合大于110吨/小时的二氧化碳到吸收溶液中。在一些实施方案中,本发明的装置和系统被配置以结合大于120吨/小时的二氧化碳到吸收溶液中。在一些实施方案中,本发明的装置和系统被配置以结合大于130吨/小时的二氧化碳到吸收溶液中。在一些实施方案中,本发明的装置和系统被配置以结合大于140吨/小时的二氧化碳到吸收溶液中。在一些实施方案中,本发明的装置和系统被配置以结合大于150吨/小时的二氧化碳到吸收溶液中。在一些实施方案中,本发明的装置和系统被配置以结合大于160吨/小时的二氧化碳到吸收溶液中。在一些实施方案中,本发明的装置和系统被配置以结合大于170吨/小时的二氧化碳到吸收溶液中。在一些实施方案中,本发明的装置和系统被配置以结合大于180吨/小时的二氧化碳到吸收溶液中。在一些实施方案中,本发明的装置和系统被配置以结合大于190吨/小时的二氧化碳到吸收溶液中。在一些实施方案中,本发明的装置和系统被配置以结合大于200吨/小时的二氧化碳到吸收溶液中。In some embodiments, the devices and systems of the present invention are configured to incorporate greater than 20 tons/hour of carbon dioxide into the absorption solution, under an average of 72 hours of continuous operation. In some embodiments, devices and systems of the present invention are configured to incorporate greater than 40 tons/hour of carbon dioxide into the absorption solution. In some embodiments, devices and systems of the present invention are configured to incorporate greater than 60 tons/hour of carbon dioxide into the absorption solution. In some embodiments, devices and systems of the present invention are configured to incorporate greater than 70 tons/hour of carbon dioxide into the absorption solution. In some embodiments, devices and systems of the present invention are configured to incorporate greater than 80 tons/hour of carbon dioxide into the absorption solution. In some embodiments, devices and systems of the present invention are configured to incorporate greater than 90 tons/hour of carbon dioxide into the absorption solution. In some embodiments, devices and systems of the present invention are configured to incorporate greater than 100 tons/hour of carbon dioxide into the absorption solution. In some embodiments, devices and systems of the present invention are configured to incorporate greater than 110 tons/hour of carbon dioxide into the absorption solution. In some embodiments, the devices and systems of the present invention are configured to incorporate greater than 120 tons/hour of carbon dioxide into the absorption solution. In some embodiments, the devices and systems of the present invention are configured to incorporate greater than 130 tons/hour of carbon dioxide into the absorption solution. In some embodiments, the devices and systems of the present invention are configured to incorporate greater than 140 tons/hour of carbon dioxide into the absorption solution. In some embodiments, devices and systems of the present invention are configured to incorporate greater than 150 tons/hour of carbon dioxide into the absorption solution. In some embodiments, devices and systems of the present invention are configured to incorporate greater than 160 tons/hour of carbon dioxide into the absorption solution. In some embodiments, the devices and systems of the present invention are configured to incorporate greater than 170 tons/hour of carbon dioxide into the absorption solution. In some embodiments, the devices and systems of the present invention are configured to incorporate greater than 180 tons/hour of carbon dioxide into the absorption solution. In some embodiments, the devices and systems of the present invention are configured to incorporate greater than 190 tons/hour of carbon dioxide into the absorption solution. In some embodiments, the devices and systems of the present invention are configured to incorporate greater than 200 tons/hour of carbon dioxide into the absorption solution.
在一些实施方案中,本发明的装置和系统包括用于从气体源,例如,工业CO2的气体源如来自发电厂的烟道气,或如来自水泥厂的废气除去CO2的装置和系统,其中该装置和系统被配置以使得CO2已经由CO2的气体源(例如二氧化碳的工业源)溶解到其中的水(例如海水,盐水,吸收溶液)经受沉淀条件,其中沉淀条件提供沉淀物,其数量为:2.6克的沉淀物/1升的吸收溶液至26.11克的沉淀物/1升的吸收溶液,在沉淀条件连续施加的平均72小时的时间上。在一些实施方案中,该装置和系统被配置以提供沉淀条件,其提供了沉淀物,,其数量为:5.2克的沉淀物/1升的吸收溶液至26.11克的沉淀物/1升的吸收溶液,在沉淀条件连续施加的平均72小时的时间上。在一些实施方案中,装置和系统被配置以提供沉淀条件,其提供沉淀物,其数量为:7.83克的沉淀物/1升的吸收溶液至26.11克的沉淀物/1升的吸收溶液,在沉淀条件连续施加的平均72小时的时间上,如9.14至26.11,如10.44至26.11,如11.75至26.11,如13.05至26.11,如14.36至26.11,如15.66至26.11,如16.97至26.11,如18.27至26.11,如19.58至26.11,如20.88至26.11,如22.19至26.11,如23.5至26.11,如24.8至26.11克的沉淀物/1升的吸收溶液。In some embodiments, the devices and systems of the present invention include devices and systems for removing CO from a gas source, for example, a gas source of industrial CO such as flue gas from a power plant, or exhaust gas such as from a cement plant , wherein the device and system are configured such that water (e.g. seawater, brine, absorption solution) into which CO2 has been dissolved by a gaseous source of CO2 (e.g. an industrial source of carbon dioxide) is subjected to precipitation conditions, wherein the precipitation conditions provide a precipitate , in amounts ranging from 2.6 g of precipitate/1 liter of absorption solution to 26.11 g of precipitate/1 liter of absorption solution, over an average period of 72 hours of continuous application of precipitation conditions. In some embodiments, the device and system are configured to provide precipitation conditions that provide a precipitate in an amount of: 5.2 grams of precipitate per liter of absorption solution to 26.11 grams of precipitate per liter of absorption solution, over an average period of 72 hours of continuous application of precipitation conditions. In some embodiments, the devices and systems are configured to provide precipitation conditions that provide precipitate in an amount of: 7.83 grams of precipitate per liter of absorption solution to 26.11 grams of precipitate per liter of absorption solution, at Precipitation conditions are applied continuously over an average period of 72 hours, such as 9.14 to 26.11, such as 10.44 to 26.11, such as 11.75 to 26.11, such as 13.05 to 26.11, such as 14.36 to 26.11, such as 15.66 to 26.11, such as 16.97 to 26.11, such as 18.27 to 26.11, such as 19.58 to 26.11, such as 20.88 to 26.11, such as 22.19 to 26.11, such as 23.5 to 26.11, such as 24.8 to 26.11 g of precipitate/1 liter of absorption solution.
碳酸盐矿物沉淀台20(即,反应器)可以包括许多不同的组件,如温度控制组件(例如,被配置以加热水至期望的温度),化学添加剂组件,例如,用于引入化学pH提高剂(如KOH,NaOH)到水中,电解组件,例如,阴极/阳极,等等,气体加料组件,增压组件(例如其中在加压条件下操作该流程,如50-800psi,或100-800psi,或400至800psi,或任何其他适合的压力范围,是期望的)等等,机械搅动和物理搅拌组件和用于再循环工业设备烟道气通过沉淀装置的组件。The carbonate mineral precipitation station 20 (i.e., the reactor) can include a number of different components, such as temperature control components (e.g., configured to heat the water to a desired temperature), chemical additive components, e.g., for introducing chemical pH raising reagents (such as KOH, NaOH) into water, electrolysis components, eg, cathode/anode, etc., gas feeding components, pressurization components (such as where the process is operated under pressurized conditions, such as 50-800psi, or 100-800psi , or 400 to 800 psi, or any other suitable pressure range, is desired), etc., mechanical agitation and physical agitation components and components for recirculating industrial plant flue gas through the settling device.
碳酸盐矿物沉淀台20可以包括许多不同的组件,其允许监控(例如,在线监控)一种或多种参数如内部反应器压力,pH,沉淀物颗粒物尺寸,金属-离子浓度,水溶液的电导率,水溶液的碱度,和pCO2。在碳酸盐沉淀方法期间的监控条件可以允许在沉淀过程期间进行校准调节。例如,可以进行校准调节以便增减碳酸盐化合物沉淀产量。在一些实施方案中,碳酸盐沉淀方法用可操作地连接到碳酸盐矿物沉淀台的在线监控装置来监控,在线监控装置包括稀释歧管,离子选择电极,伏特计,控制器,和稀释剂源。图25举例说明一个可能的在线监控器1600。操作上,控制器(1660)控制可变流量控制阀(1621),控制器-操作的可变流量控制阀允许沉淀台流出物进入稀释歧管(1630),一般地在通过过滤器(未示于图25中)后,以便除去碳酸盐矿物沉淀物。未稀释的沉淀台流出物然后测量离子浓度。为进行离子浓度测量,使用例如伏特计(1650),其连接到一个或多个电极(1640),对特定的离子(例如,Ca2+或Mg2+)具有选择性。如果测量的电压在被接受的电压范围以外(例如,例如在电压与离子浓度曲线的直线部分以外),在稀释歧管中的沉淀台流出物用稀释剂(1610)稀释。控制器,可操作地连接到伏特计和稀释剂,监控并且记录通过控制器-操作的可变的控制阀(1610)的稀释剂的输送。控制的稀释剂输送被减慢或停止,当电压读数落入被接受的范围内时。取决于在最初的沉淀台流出物中所考虑的离子的控制器-确定的离子浓度,通过可变流量控制阀1612(例如,如果离子浓度高的话)可以将稀释剂添加到沉淀台,或者沉淀台流出物可以从沉淀台通过阀1622排出(例如,如果离子浓度低的话),伴随着进行通过沉淀台入口的更浓缩的离子溶液的输送。The carbonate
可期望结合来自气体物流的一个或多个组分到吸收溶液中,如从工业烟道气中除去二氧化碳和任选地SOx、NOx、及其他非CO2酸性气体,而没有形成固体沉淀物。在此情况下,控制器,监控装置,和沉淀条件,在本文中其它地方详细描述的,可以被使用以便最优化至少一种气体组分引入吸收溶液并且继续防止一个或多个组分释放到地球大气中。如果引入气体物流的组分到吸收溶液中没有导致沉淀,所得的溶液可以进一步通过系统进行加工,以便回收可用的吸收溶液或者回收水,其是可饮用的或者适合于灌溉目的。或者,如果引入气体物流组分到吸收溶液中没有引起沉淀,那么所得的溶液可以被送到滞留设备,如,但不限于地下位置,包括地质构造如含水层。由将气体物流组分引入吸收溶液产生的这样的溶液可以被输送到滞留设备,使用任何便利方法,包括但不限于管道如管子或沟渠,油罐卡车,泵,通过铁路或驳船运输的罐子,或其组合。It may be desirable to incorporate one or more components from a gas stream into an absorption solution, such as removing carbon dioxide and optionally SOx, NOx, and other non- CO acid gases from industrial flue gases, without forming solid precipitates. In this case, controllers, monitoring devices, and precipitation conditions, described in detail elsewhere herein, may be used in order to optimize the introduction of at least one gas component into the absorption solution and continue to prevent the release of one or more components into the in the Earth's atmosphere. If the introduction of components of the gas stream into the absorption solution does not result in precipitation, the resulting solution can be processed further through the system to recover usable absorption solution or to recover water which is potable or suitable for irrigation purposes. Alternatively, if the introduction of gas stream components into the absorption solution does not cause precipitation, the resulting solution may be sent to a retention facility such as, but not limited to, an underground location, including geological formations such as aquifers. Such a solution resulting from the introduction of components of the gas stream into the absorption solution may be transported to the retention facility using any convenient method including, but not limited to, pipelines such as pipes or ditches, tank trucks, pumps, tanks transported by rail or barge, or a combination thereof.
如图6A中举例说明的,由在步骤20沉淀产生的沉淀产物可以与沉淀台流出物在步骤40处分离而产生分离的沉淀产物。由于新分离的沉淀产物可以在随后步骤中干燥,被分离的沉淀产物也可是“湿脱水沉淀物”。使用许多适宜途径,实现沉淀台流出物与沉淀产物的分离,包括排水(例如,沉淀产物的重力沉淀随后排水),倾析,过滤(例如,重力过滤,真空过滤,使用增压空气的过滤),离心,压榨,或其任何组合。在一些实施方案中,沉淀产物与沉淀台流出物的分离是通过相抵于折流板使沉淀台流出物流动来实现的,其中上层清液相抵于折流板偏转并且与沉淀产物颗粒分离,后者被收集在收集器中。在一些实施方案中,沉淀产物与沉淀台流出物的分离是通过使沉淀台流出物在螺旋通道中流动,分离沉淀产物颗粒并且由螺旋通道出口的阵列收集沉淀产物。机械地,至少一种液-固分离装置可操作地连接到沉淀台使得沉淀台流出物可以由沉淀台流动到液-固分离装置(例如,液-固分离装置包括折流板或螺旋通道)。沉淀台流出物可以直接流到液-固分离装置,或者流出物可以预处理,如下所详细描述。As illustrated in Figure 6A, the precipitated product resulting from precipitation at
对于任何上述分离途径的能量需要可以通过以下来实现:使方法适应于使用许多含能量的废物物流(例如,废热或废气物流),这些废物物流是由工业设备提供的;然后本领域技术人员应当理解的是需要较少能量的分离方法是令人期望的,在减小本发明的碳足迹方面。The energy requirements for any of the above separation pathways can be met by adapting the process to use a number of energy-containing waste streams (e.g., waste heat or waste gas streams) provided by industrial facilities; a person skilled in the art should then It is understood that separation methods requiring less energy are desirable in reducing the carbon footprint of the invention.
来自沉淀台流出物的沉淀产物的浓缩和分离可以连续或间歇地实现,使用描述于WO 2007/051640和CA02628270中的液-固分离方法和装置,其公开内容引入本文作为参考。在一些实施方案中,液-固分离装置包括容器,其具有漏斗状的段,在容器中排列而在纵向中延伸的沉淀台流出物管和通过用于引入通过沉淀台流出物管落下的沉淀台流出物流动的入口开口和在漏斗状段的下端形成的用于从容器除去分离的沉淀产物的除去开口向容器的开口,特征为在入口开口区域中排列的折流板,沉淀台流出物流动通过其得到偏转。液-固分离器如Epuramat′s Extrem-Separator(″ExSep″)液-固分离器或其变体可用于分离沉淀产物和沉淀台流出物的一些实施方案中。Concentration and separation of the precipitated product from the effluent of the settling table can be achieved continuously or intermittently, using the liquid-solid separation method and apparatus described in WO 2007/051640 and CA02628270, the disclosures of which are incorporated herein by reference. In some embodiments, the liquid-solid separation device includes a vessel having a funnel-shaped section, a settling table effluent pipe extending in the longitudinal direction arranged in the vessel and a channel for introducing sediment falling through the settling table effluent pipe. The inlet opening for the flow of the table effluent and the removal opening formed at the lower end of the funnel-shaped section for removing the separated precipitated product from the container The opening to the container is characterized by baffles arranged in the area of the inlet opening, the sedimentation table effluent flow Movement is deflected through it. Liquid-solid separators such as Epuramat's Extrem-Separator ("ExSep") liquid-solid separators or variations thereof may be used in some embodiments to separate the precipitation product and the precipitation table effluent.
为从水中分离沉淀产物,在重力的方向将沉淀台流出物引入浴中,其中在重力作用下沉淀产物颗粒下降并且从其底部被除去。沉淀产物颗粒的这种除去可以连续或间歇地进行。沉淀台流出物,当其引入浴中时,相抵于折流板流动,浴中的流动通过折流板而偏转。通过这种方法控制,在入口开口的区域中产生了水力-物理反应区,其中在重力方向中沉淀台流出物流动的至少显著的流动能被破坏。在垂直方向中使流入沉淀台流出物管的沉淀台流出物流动偏转有利于沉淀产物颗粒的分离,这应归于相比于水电密度差。当使沉淀台流出物偏转时,较重的沉淀产物颗粒具有更大的倾向从而在沉淀台流出物管的方向(即,在向下方向中)继续其运动轨迹,而水被偏转,并且与重的沉淀产物颗粒分离,上升。当相抵于折流板流动时(即在流过沉淀台流出物管的沉淀台流出物的流向中,在折流板下游离开沉淀台流出物管时以及主要地在其之后),基本上由偏转损失引起流动能的破坏。沉淀台流出物特别地通过下述的方式来偏转,即,沉淀产物颗粒(即,具有比水密度大的颗粒),其通常将在容器中下降,以基本上未拢动方式,在引入到所述浴期间,继续其起源于沉淀台流出物管的下降运动。偏转将不会具有这样的结果:具有高密度的沉淀产物颗粒,即,沉淀产物颗粒,在偏转期间具有施加到其上的向上速度的化合物。在偏转期间这样的速度分量应当仅仅施加到轻水上,使得由于偏转,在折流板处,水接收需要速度组分以便在浴中上升。To separate the precipitated product from water, the settling table effluent is introduced into the bath in the direction of gravity, where the precipitated product particles descend under the force of gravity and are removed from its bottom. This removal of precipitated product particles can be carried out continuously or intermittently. The settling table effluent, as it is introduced into the bath, flows against the baffles by which the flow in the bath is deflected. Controlled by this method, a hydro-physical reaction zone is created in the region of the inlet opening, in which at least significant flow energy of the settling table effluent flow in the direction of gravity is disrupted. Deflecting the flow of the sedimentation table effluent into the sedimentation table effluent pipe in the vertical direction facilitates the separation of the precipitation product particles due to the difference in electric density compared to the water. When the settling table effluent is deflected, the heavier settling product particles have a greater tendency to continue their trajectory in the direction of the settling table effluent pipe (i.e., in the downward direction), while the water is deflected, and with The heavy precipitated product particles separate and rise. When flowing against the baffles (i.e. in the flow direction of the settling table effluent flowing through the settling table effluent pipe, when leaving the settling table effluent pipe downstream of the baffles and mainly after it), basically by Deflection losses cause disruptions in kinetic energy. The settling table effluent is deflected in particular by the settling product particles (i.e., particles having a greater density than water), which would normally descend in the vessel, in a substantially undisturbed manner, upon introduction into the During the bath, the downward movement of the effluent pipe originating from the settling table continued. The deflection would not have the result of having a high density of precipitation product particles, ie the compound of the precipitation product particles having an upward velocity applied to them during the deflection. Such a velocity component should only be applied to the light water during deflection, so that due to the deflection, at the baffles, the water receives the required velocity component in order to rise in the bath.
或者,来自沉淀台流出物的沉淀产物的浓度和分离可以连续或间歇地实现,使用描述于US 2008/018331中的液-固分离方法和装置,其公开内容引入本文作为参考。在一些实施方案中,液-固分离装置包括入口,其操作以接收沉淀台流出物;通道,其操作以允许沉淀台流出物的流动,该通道处于螺旋配置结构中;分离手段,用于从沉淀台流出物分离沉淀产物;和至少一个用于沉淀产物贫乏的上层清液的出口。液-固分离器如Xerox PARC′s螺旋浓缩器,或其变体,可用于一些实施方案来分离沉淀产物与沉淀台流出物。Alternatively, the concentration and separation of precipitation products from the effluent of the precipitation table can be achieved continuously or intermittently, using the liquid-solid separation method and apparatus described in US 2008/018331, the disclosure of which is incorporated herein by reference. In some embodiments, the liquid-solid separation device comprises an inlet operative to receive the settling table effluent; a channel operative to allow flow of the settling table effluent, the channel being in a helical configuration; a precipitation stage effluent separation precipitation product; and at least one outlet for a precipitation product-depleted supernatant. Liquid-solid separators such as Xerox PARC's spiral concentrators, or variations thereof, may be used in some embodiments to separate the precipitation product from the precipitation table effluent.
基于沉淀产物颗粒(其被迫在螺旋通道中流动)的尺寸和质量分离,沉淀产物与沉淀台流出物分离。在螺旋段上,来自流体剪切的向内的横向压力场与向外的离心力竞争从而允许分离沉淀产物颗粒。在高速,离心力占优势,沉淀产物颗粒向外移动。在低速,横向压力占优势,沉淀产物颗粒向内移动。两个相对力的大小取决于流速,粒径,螺旋段的曲率半径,通道尺寸,和沉淀台流出物的粘度。在螺旋通道的末端,平行阵列的出口收集了沉淀产物的分离颗粒。对于任何粒径,所需的通道尺寸通过估计到达侧壁的转移时间来确定。该时间是流速,通道宽度,粘度,和曲率半径的函数。沉淀产物的较大的颗粒可以先于较小的颗粒到达通道壁,后者需要更多时间到达侧壁。因此,螺旋通道可以沿通道具有多出口。这种技术固有地在大尺寸范围(从亚毫米直至1微米)上是可伸缩的。The precipitation product is separated from the settling table effluent based on the size and mass separation of the precipitation product particles, which are forced to flow in the helical channel. On the helical section, the inward lateral pressure field from fluid shear competes with the outward centrifugal force allowing separation of the precipitated product particles. At high speeds, centrifugal force prevails and the precipitated product particles move outward. At low speeds, transverse pressure prevails and the precipitated product particles move inward. The magnitude of the two relative forces depends on the flow rate, particle size, radius of curvature of the helical section, channel dimensions, and viscosity of the sedimentation table effluent. At the end of the helical channel, a parallel array of outlets collects the separated particles of the precipitated product. For any particle size, the required channel size is determined by estimating the transfer time to the sidewall. This time is a function of flow rate, channel width, viscosity, and radius of curvature. Larger particles of precipitation product can reach the channel walls before smaller particles, which take more time to reach the side walls. Thus, a helical channel may have multiple outlets along the channel. This technique is inherently scalable over a large size range (from sub-millimeter down to 1 micron).
可能期望的是预处理(例如,粗过滤)沉淀台流出物而从流出物中除去沉淀产物的大尺寸颗粒,然后提供流出物给液-固分离装置,因为大尺寸的颗粒可妨碍液-固分离装置或方法。使用单个液-固分离装置可以实现来自沉淀台流出物的沉淀产物的分离。在一些实施方案中,可以使用2、3、4、5、或多于5个液-固分离装置的组合以便分离来自沉淀台流出物的沉淀产物。液-固分离器的组合可以串联、并联或者串并联组合使用,这取决于期望的处理量。在一些实施方案中,液-固分离装置或其组合能够以100L/min-2,000,000L/min,100L/min-1,000,000L/min,100L/min-500,000L/min,100L/min-250,000L/min,100L/min-100,000L/min,100L/min-50,000L/min,100L/min-25,000L/min,和100L/min-20,000L/min处理沉淀台流出物。在一些实施方案中,液-固分离装置或其组合能够以1000L/min-2,000,000L/min,5000L/min-2,000,000L/min,10,0000L/min-2,000,000L/min,20,000L/min-2,000,000L/min,25,000L/min-2,000,000L/min,50,000L/min-2,000,000L/min,100,000L/min-2,000,000L/min,250,000L/min-2,000,000L/min,500,000L/min-2,000,000L/min,和1,000,000L/min-2,000,000L/min处理沉淀台流出物。在一些实施方案中,液-固分离装置或其组合能够以1000L/min-20,000L/min,5000L/min-20,000L/min,10,000L/min-20,000L/min,1000L/min-10,000L/min,2000L/min-10,000L/min,3000L/min-10,000L/min,4000L/min-10,000L/min,5000L/min-10,000L/min,6000L/min-10,000L/min,7000L/min-10,000L/min,8000L/min-10,000L/min,9000L/min-10,000L/min,或9500L/min-10,000L/min处理沉淀台流出物。It may be desirable to pretreat (e.g., coarse filter) the settling table effluent to remove large-sized particles of the precipitated product from the effluent before providing the effluent to a liquid-solid separation device, since large-sized particles can interfere with liquid-solid separation. Separation device or method. Separation of the precipitation product from the precipitation table effluent can be achieved using a single liquid-solid separation device. In some embodiments, a combination of 2, 3, 4, 5, or more than 5 liquid-solid separation devices may be used to separate the precipitation product from the precipitation table effluent. Combinations of liquid-solid separators can be used in series, parallel or a combination of series and parallel, depending on the desired throughput. In some embodiments, the liquid-solid separation device or combination thereof can operate at 100L/min-2,000,000L/min, 100L/min-1,000,000L/min, 100L/min-500,000L/min, 100L/min-250,000L/min min, 100L/min-100,000L/min, 100L/min-50,000L/min, 100L/min-25,000L/min, and 100L/min-20,000L/min process the sedimentation table effluent. In some embodiments, the liquid-solid separation device or combination thereof can operate at 1000L/min-2,000,000L/min, 5000L/min-2,000,000L/min, 10,0000L/min-2,000,000L/min, 20,000L/min- 2,000,000L/min, 25,000L/min-2,000,000L/min, 50,000L/min-2,000,000L/min, 100,000L/min-2,000,000L/min, 250,000L/min-2,000,000L/min, 500,000L/min- 2,000,000L/min, and 1,000,000L/min-2,000,000L/min to treat sedimentation platform effluent. In some embodiments, the liquid-solid separation device or combination thereof can operate at 1000L/min-20,000L/min, 5000L/min-20,000L/min, 10,000L/min-20,000L/min, 1000L/min-10,000L /min, 2000L/min-10,000L/min, 3000L/min-10,000L/min, 4000L/min-10,000L/min, 5000L/min-10,000L/min, 6000L/min-10,000L/min, 7000L/min min-10,000L/min, 8000L/min-10,000L/min, 9000L/min-10,000L/min, or 9500L/min-10,000L/min to process the sedimentation platform effluent.
串联、并联或者串并联组合的液-固分离器的组合也可用于增加分离效率。另外,由一个或多个液-固分离装置产生的上层清液可以再循环通过液-固分离装置以提高分离效率。在一些实施方案中,从沉淀台流出物收集30%至100%,40%至100%,50%至100%,60%至100%,70%至100%,75%至100%,80%至100%,85%至100%,90%至100%,95%至100%,96%至100%,97%至100%,98%至100%,99%至100%的沉淀产物。取决于从沉淀台流出物除去的沉淀产物的量,上层清液可以被输送回到沉淀台或者提供到本发明的电解电池。在一些实施方案中,具有较高浓度的沉淀产物的上层清液被输送回到沉淀台用于沉淀产物颗粒的结块。在一些实施方案中,具有较高浓度的溶解的二价阳离子(例如,Ca2+或Mg2+)的上层清液被输送回到沉淀台作为二价阳离子源。在一些实施方案中,具有相对低浓度的沉淀产物和溶解的二价阳离子的上层清液被过滤而除去显著数量的剩余的二价阳离子并且提供给本发明的电解电池。Combinations of liquid-solid separators in series, parallel or a combination of series and parallel can also be used to increase separation efficiency. Additionally, the supernatant produced by one or more liquid-solid separation devices may be recycled through the liquid-solid separation device to increase separation efficiency. In some embodiments, 30% to 100%, 40% to 100%, 50% to 100%, 60% to 100%, 70% to 100%, 75% to 100%, 80% are collected from the settling table effluent to 100%, 85% to 100%, 90% to 100%, 95% to 100%, 96% to 100%, 97% to 100%, 98% to 100%, 99% to 100% of the precipitated product. Depending on the amount of precipitation product removed from the settling station effluent, the supernatant may be sent back to the settling station or provided to the electrolytic cell of the present invention. In some embodiments, the supernatant, which has a higher concentration of precipitation product, is conveyed back to the settling station for agglomeration of the precipitation product particles. In some embodiments, the supernatant with a higher concentration of dissolved divalent cations (eg, Ca2+ or Mg2+) is sent back to the settling station as a source of divalent cations. In some embodiments, the supernatant, which has relatively low concentrations of precipitated products and dissolved divalent cations, is filtered to remove significant amounts of remaining divalent cations and provided to the electrolytic cells of the present invention.
沉淀产物颗粒的这种除去可以连续或间歇地进行。This removal of precipitated product particles can be carried out continuously or intermittently.
在一些实施方案中,自沉淀台流出物中,没有分离或者仅仅部分分离了沉淀产物。在这样的实施方案中,流出物,包括一些(例如,在通过液固分离装置后)或者全部沉淀产物可以以许多不同的方式进行处理。在一些实施方案中,来自沉淀台的排出物,包括一些或全部的沉淀产物,被运输到陆地或水位置并且在该位置储藏。运输到海洋特别地在实施方案中是有用的,其中水源是海水。应将理解的是碳足迹,所使用的能量数量,和/或所产生的CO2的量,对于由工业废气埋存一定量的CO2来说,在方法中被最小化,其中对沉淀物没有进行进一步的除了处理之外的加工。In some embodiments, the precipitation product is not separated or only partially separated from the precipitation table effluent. In such embodiments, the effluent, including some (eg, after passing through the liquid-solid separation device) or all of the precipitated product, can be treated in a number of different ways. In some embodiments, the effluent from the settling station, including some or all of the settling products, is transported to a land or water location and stored there. Transport to the ocean is particularly useful in embodiments wherein the water source is seawater. It will be appreciated that the carbon footprint, the amount of energy used, and/or the amount of CO2 produced, for a certain amount of CO2 sequestered by industrial waste gas, is minimized in a method wherein the sediment No further processing other than treatment was performed.
在图6A中所举例说明的实施方案中,所得的脱水沉淀物然后被干燥以产生产物,如图6A的步骤60处举例说明的。通过空气干燥滤液可以实现干燥。在滤液被空气干燥的情况下,空气干燥可以在室温或高温下。在某些实施方案中,由工业设备气态废物物流提供了高温,如图7的步骤70处所举例说明的。在这些实施方案中,来自发电厂的气态废物物流(例如,烟道气)可以首先用于干燥步骤,其中气态废物物流可以具有30至700℃,如75至300℃的温度。气态废物物流可以直接与湿沉淀物在干燥阶段中接触,或者用于间接地在干燥阶段中加热气体(如空气)。通过使来自工业设备的气体输送器,例如,管道起源在合适的位置,例如,在HRSG中或烟道上某一距离的位置处,期望的温度可以被提供在气态废物物流中,如基于工业设备的排气气体和配置结构的细节来确定。在又一实施方案中,沉淀物被喷雾干燥来干燥沉淀物,其中含沉淀物的液体通过将其进料通过热气(如来自工业设备的气态废物物流)而被干燥,例如,其中液体进料被泵送通过雾化器进入主干燥室,而热气以相对于雾化器方向的顺流或逆流的形式通过。在某些实施方案中,干燥是通过冷冻干燥(即,冻干)来实现的,其中沉淀物被冷冻,降低周围压力并且添加足够的热量以使得材料中的冷冻水直接从冷冻沉淀相升华为气体。取决于系统的特定的干燥流程,干燥台可以包括过滤元件,冷冻干燥结构,喷雾干燥结构,等。In the embodiment illustrated in Figure 6A, the resulting dehydrated precipitate is then dried to produce a product, as illustrated at
在期望的情况下,如在图6A的任选的步骤50所举例说明的,在干燥前可以洗涤来自分离反应器40的脱水的沉淀物产物。沉淀物可以用淡水洗涤,例如,以便从脱水的沉淀物中除去盐(如NaCl)。所使用的洗涤水可以适宜地进行处理,例如,在尾矿池中处理它,等等。If desired, the dewatered precipitate product from the
在本发明的某些实施方案中,沉淀物可以被分离,洗涤和干燥,对于全部方法在相同的台中,或者对于全部方法,在不同的台中,或者任何其它可能的组合。例如,在一个实施方案中,在沉淀反应器20中可以发生沉淀和分离,但是干燥和洗涤在不同的反应器中发生。在又一实施方案中,沉淀、分离和干燥可以都在沉淀反应器20中发生,而洗涤在不同的反应器中进行。In certain embodiments of the invention, the precipitate can be separated, washed and dried, in the same station for all methods, or in different stations for all methods, or any other possible combination. For example, in one embodiment, precipitation and separation can occur in
在分离沉淀物和母液后,例如,如上所述,分离后的沉淀物可以进一步被处理,根据期望。在某些实施方案中,沉淀物然后可以被输送到某个位置以便长期存储,有效地埋存CO2。例如,沉淀物可以被输送并且置于长期储存位置,例如,在地上,地下,深海中等,根据期望。After separating the precipitate and mother liquor, eg, as described above, the separated precipitate can be further processed, as desired. In certain embodiments, the precipitate can then be transported to a location for long-term storage, effectively sequestering the CO2 . For example, sediment can be transported and placed in a long-term storage location, eg, above ground, underground, in deep sea, etc., as desired.
干燥产品可以以许多不同的方式处置。在某些实施方案中,沉淀物产物被输送至某一位置以便长期存储,有效地以稳定的沉淀的产物的形式,如以存储稳定的地上的CO2-埋存材料的形式埋存CO2。例如,沉淀物可以在邻近于工业设备和沉淀系统的长期储存位置存储。在其它实施方案中,沉淀物可以被输送并且置于长期储存位置,例如,在地上、地下等,根据期望,其中长期储存位置远离发电厂(这在其中在发电厂附近地产缺乏的实施方案中可能是令人期望的)。在其中沉淀物被输送至长期储存位置的这些实施方案中,其可以在空的运输交通工具(例如,驳船,车厢轮渡(train cars),卡车,等等)中进行输送,后者的使用以便将燃料或其它材料输送到工业设备和/或沉淀装置。如此,可以使用用于将燃料带到工业设备,将材料带到沉淀装置(例如,碱源)的运输交通工具来运输沉淀产物,由此埋存来自工业设备的CO2。Dried products can be disposed of in many different ways. In certain embodiments, the sediment product is transported to a location for long- term storage, effectively sequestering CO2 in the form of a stable precipitated product, such as in the form of a storage-stable aboveground CO2 -storage material . For example, sediment can be stored in long-term storage locations adjacent to industrial equipment and sedimentation systems. In other embodiments, the sediment can be transported and placed in a long-term storage location, e.g., above ground, underground, etc., as desired, where the long-term storage location is remote from the power plant (this is the case in embodiments where real estate near the power plant is scarce). may be desirable). In those embodiments where the sediment is transported to a long-term storage location, it may be transported in empty transport vehicles (e.g., barges, train cars, trucks, etc.), the latter being used for Conveying fuel or other materials to industrial equipment and/or settling plants. In this way, the precipitation product can be transported using the transport vehicle used to bring the fuel to the industrial facility and the material to the precipitation facility (eg, alkali source), thereby sequestering the CO2 from the industrial facility.
在某些实施方案中,在水下位置处理组合物。水下位置可以不同,这取决于特定的应用。虽然水下位置可以是内陆水下位置,例如,在湖泊(包括淡水湖)中,在某些实施方案中的兴趣是海洋或海水下位置。组合物仍然可以在母液中,而没有分离或者没有完全分离,或者组合物可以已经与母液分离。水下位置可以是浅的或深的。浅的位置是200英尺或更小的位置,如150英尺或更小,包括1000英尺或更小。深位置是200英尺或更大的那些,例如500英尺或更大,1000英尺或更大,2000英尺或更大,包括5000英尺或更大。In certain embodiments, the composition is treated in an underwater location. The underwater location can vary, depending on the particular application. While the underwater location may be an inland underwater location, for example, in a lake (including freshwater lakes), in certain embodiments the interest is a marine or seawater underwater location. The composition may still be in the mother liquor without separation or complete separation, or the composition may have been separated from the mother liquor. Underwater locations can be shallow or deep. A shallow location is a location of 200 feet or less, such as 150 feet or less, including 1000 feet or less. Deep locations are those that are 200 feet or greater, such as 500 feet or greater, 1000 feet or greater, 2000 feet or greater, including 5000 feet or greater.
在期望的情况下,由沉淀物和母液组成的组合物,在沉淀后和在处理前,可以存储一段时间。例如,组合物可以在1℃-40℃,如20℃-25℃,存储1至1000天或更长,如1-10天或更长。Where desired, the composition consisting of the precipitate and the mother liquor can be stored for a period of time after precipitation and before disposal. For example, the composition may be stored at 1°C-40°C, such as 20°C-25°C, for 1 to 1000 days or longer, such as 1-10 days or longer.
任何适宜的用于将组合物运输到处理位置的流程可被使用并且将必然地取决于沉淀反应器位置和处理位置相互关系来改变,其中处理位置是地上或地下的处理位置等等。在某些实施方案中,使用管道或者类似的浆液输送结构,其中这些途径可以包括有源泵,重力介导的流动,等等,根据期望。Any suitable procedure for transporting the composition to the processing location may be used and will necessarily vary depending on the precipitation reactor location and the processing location interrelationship, whether the processing location is an aboveground or underground processing location, and the like. In certain embodiments, pipes or similar slurry delivery structures are used, where these pathways may include active pumps, gravity mediated flow, etc., as desired.
虽然在某些实施方案中,沉淀物直接在处理位置处理而没有在沉淀后的进一步加工,但在其它实施方案中组合物可以在处理前被进一步加工。例如,在某些实施方案中,可以由组合物产生固体物理形状,其中所得的形状然后在所考虑的处理位置进行处理。这种实施方案的一个实例是在由碳酸盐化合物组合物产生人工礁结构的情况中,例如,通过将可流动的组合物置于合适的模具结构中并且随时间使组合物固化成期望的形状。所得的固体礁结构然后可以储存在合适的海洋位置,例如浅的水下位置,以产生人工礁,根据期望。While in certain embodiments the precipitate is processed directly at the processing site without further processing after precipitation, in other embodiments the composition may be further processed prior to processing. For example, in certain embodiments, a solid physical shape may be produced from the composition, wherein the resulting shape is then processed at the contemplated processing site. An example of such an embodiment is where an artificial reef structure is produced from a carbonate compound composition, for example, by placing a flowable composition in a suitable mold structure and allowing the composition to cure over time into the desired shape . The resulting solid reef structure can then be stored in a suitable marine location, such as a shallow underwater location, to create an artificial reef, as desired.
在某些实施方案中,通过本发明的方法产生的沉淀物通过将其用于制品中而处理。换言之,使用产物来制造人造物品,即制造物品。产物本身可以使用或者同一种或多种另外的材料相结合,使得其是制造物品的组分。所考虑的制造物品可以变化,其中所考虑的制造物品的实例包括建筑材料和非建筑材料如非粘结制造物品。所考虑的建筑材料包括混凝土的组分,如水泥,集料(细和粗的),辅助的粘结材料等。所考虑的建筑材料还包括预成形的建筑材料。In certain embodiments, the precipitate produced by the methods of the invention is treated by using it in an article of manufacture. In other words, using products to make man-made objects, ie making objects. The product may be used by itself or combined with one or more additional materials such that it is a component of an article of manufacture. Contemplated articles of manufacture may vary, with examples of contemplated articles of manufacture including construction materials and non-construction materials such as non-bonded articles of manufacture. The construction materials considered include concrete components such as cement, aggregates (fine and coarse), auxiliary binding materials, etc. Contemplated building materials also include preformed building materials.
在通过将产物结合在建筑材料中来处理产物的情况下,来自工业设备的气态废物物流的CO2被有效地埋存在人造环境中。在建筑材料中使用产物的实例包括其中产物用作某些类型的人造结构的建筑材料的情况,例如建筑(商业或住宅),公路,桥梁,大堤,水坝,和其它人造的结构等等。可以使用建筑材料作为这样的结构的结构或者非结构组分。在这样的实施方案中,沉淀装置可以与建筑产品工厂共处。 CO2 from gaseous waste streams from industrial facilities is effectively buried in man-made environments where the product is disposed of by incorporating it in building materials. Examples of the use of the product in building materials include cases where the product is used as a building material for certain types of man-made structures, such as buildings (commercial or residential), roads, bridges, levee, dams, and other man-made structures and the like. Building materials may be used as structural or non-structural components of such structures. In such an embodiment, the settling unit may be co-located with the building products factory.
在某些实施方案中,在随后使用前以某种方式精制(即,加工)沉淀的产物。如图6A的步骤80中举例说明的精制可以包括各种不同的流程。在某些实施方案中,产品受到机械精制,例如,磨碎,以便获得具有期望的物理性能的产品,例如,粒径等。在某些实施方案中,沉淀物与水硬水泥结合,例如,作为辅助粘结材料,作为沙、砂砾、作为集料等等。在某些实施方案中,一种或多种组分可以被添加到沉淀物,例如,其中沉淀物将被用作水泥,例如,一种或多种添加剂,沙,集料,辅助的粘结材料,等等以产生最终产品,例如,混凝土或者灰浆,90。In certain embodiments, the precipitated product is refined (ie, processed) in some manner prior to subsequent use. Refining as exemplified in
在某些实施方案中,碳酸盐化合物沉淀物被用于生产集料。这样的集料,它们的制造方法,和其用途描述于共同未决的美国专利申请(公开号)US 2010-0024686A1中,后者于2010年2月4日公开,其全盘引入本文作为参考。In certain embodiments, carbonate compound precipitates are used to produce aggregate. Such aggregates, their method of manufacture, and their use are described in co-pending United States Patent Application (Publication No.) US 2010-0024686A1, which was published on February 4, 2010, which is incorporated herein by reference in its entirety.
在某些实施方案中,碳酸盐化合物沉淀物用作水硬水泥的组分。术语“水硬水泥”以其常规含义使用,是指在与水结合后固化和硬化的组合物。通过本发明的水泥与水流体的组合产生的产品的固化和硬化源于水合物的产生,其是由水泥在与水反应时形成的,其中水合物基本上不溶于水。这样的碳酸盐化合物组分水硬水泥,它们的制造方法,和其用途描述于共同未决的美国专利申请(公开号)US 2009-0020044A1中,后者于2009年1月22日公开,其全盘引入本文作为参考。In certain embodiments, carbonate compound precipitates are used as components of hydraulic cement. The term "hydraulic cement" is used in its conventional sense to refer to a composition that cures and hardens after combining with water. The setting and hardening of the products produced by the combination of the cement of the present invention with an aqueous fluid results from the production of hydrates, which are formed by the cement upon reaction with water, wherein the hydrates are substantially insoluble in water. Such carbonate compound component hydraulic cements, their method of manufacture, and their use are described in co-pending U.S. Patent Application (Publication No.) US 2009-0020044A1, which was published on January 22, 2009, It is incorporated herein by reference in its entirety.
还考虑的是所成形的建筑材料。本发明的所成形的建筑材料可以变化很大。“所成形的”是指成型的,例如,模制的,浇铸的,切削的或者以其它方式产生的,成为人造结构所限定的物理形状,即,配置结构。所成形的建筑材料不同于无定形建筑材料,例如颗粒物(如粉末)组合物,后者不具有所限定的和稳定的形状,但反而符合其中装盛它们的容器,例如,袋子或者其它容器。例证性的所成形的建筑材料包括但不局限于:砖;板;管道;横梁;盆状物;柱状物;干式墙;等等。关于所成形的建筑材料的进一步的实例和细节包括美国专利申请NO12/571,398中所述的那些,该申请的申请日为2009年9月30日,其全盘引入本文作为参考。Also considered is the formed building material. The formed building materials of the present invention can vary widely. "Formed" means shaped, eg, molded, cast, cut or otherwise produced, into a defined physical shape of a man-made structure, ie, a configuration. Shaped building materials are distinguished from amorphous building materials, such as granular (eg, powder) compositions, which do not have a defined and stable shape, but instead conform to the container in which they are contained, eg, a bag or other container. Exemplary formed building materials include, but are not limited to: bricks; slabs; pipes; beams; pots; columns; drywall; Further examples and details regarding formed building materials include those described in US Patent Application No. 12/571,398, filed September 30, 2009, which is incorporated herein by reference in its entirety.
还考虑的是非粘结制造的物品,其包括本发明的产物作为组分。本发明的非粘结制造的物品可以变化很大。非粘结是指组合物不是水硬水泥。因而,组合物不是干燥的组合物,其当与固化流体结合时,如水,固化而产生稳定产品。例证性的组合物包括但不局限于:纸制品;聚合物产品;润滑剂;沥青制品;油漆;个人护理产品,如化妆品,牙膏,除臭剂,肥皂和洗发剂;人类可摄取的产品,包括液体和固体;农产品,比如土壤改良剂产品和动物进料;等等。关于非粘结制造的物品的进一步的实例和细节包括美国专利申请No 12/609,491中所述的那些,该申请的申请日为2009年10月30日,其全盘引入本文作为参考。Also contemplated are non-bonded manufactured articles comprising the products of the invention as components. The non-bonded manufactured articles of the present invention can vary widely. Non-bonding means that the composition is not hydraulic cement. Thus, the composition is not a dry composition, which when combined with a curing fluid, such as water, cures to produce a stable product. Exemplary compositions include, but are not limited to: paper products; polymer products; lubricants; asphalt products; paints; personal care products such as cosmetics, toothpaste, deodorants, soaps, and shampoos; human ingestible products , including liquids and solids; agricultural products, such as soil amendment products and animal feed; and more. Further examples and details regarding non-bonded manufactured articles include those described in U.S. Patent Application No. 12/609,491, filed October 30, 2009, which is incorporated herein by reference in its entirety.
所得的母液也可被处理,根据期望。例如,母液可以返回到水的来源,例如,海洋,或者到另一位置。在某些实施方案中,母液可以与CO2源接触,例如,如上所述,以便埋存其它的CO2。例如,在母液将返回到海洋的情况下,母液可以以足以增加存在于母液中的碳酸盐离子的浓度的方式与CO2的气体源接触。使用任何适宜的流程,如上述的那些,可以进行接触。在某些实施方案中,母液具有碱性pH,并且以足以降低pH至5-9,例如6-8.5,包括7.5-8.2的方式进行与CO2源接触。因此,所得的反应母液,例如,矿物碳酸盐贫乏的水,可使用任何适宜的流程进行处理。在某些实施方案中,其可以被送到尾矿池进行处理。在某些实施方案中,其可以在天然存在的水体,例如,海洋,海,湖泊或河流中处理。在某些实施方案中,其可以用作工业设备的冷却剂,例如,通过在沉淀系统和工业设备之间运转的作业线。在某些实施方案中,其可以用作灰水,脱盐用的水输入,和随后用作淡水,例如,在灌溉中,用于人类和动物消费,等等。因此,所考虑的是这样的配置结构,其中沉淀装置与脱盐装置共处,使得来自沉淀装置的输出水被用作脱盐装置的注入水。The resulting mother liquor can also be processed, as desired. For example, the mother liquor can be returned to the source of the water, eg, the ocean, or to another location. In certain embodiments, the mother liquor can be contacted with a source of CO2 , eg, as described above, to sequester additional CO2 . For example, where the mother liquor is to be returned to the ocean, the mother liquor may be contacted with a gas source of CO2 in a manner sufficient to increase the concentration of carbonate ions present in the mother liquor. Contacting can be performed using any suitable procedure, such as those described above. In certain embodiments, the mother liquor has an alkaline pH, and the contacting with the source of CO2 is performed in a manner sufficient to lower the pH to 5-9, such as 6-8.5, including 7.5-8.2. Thus, the resulting reaction mother liquor, eg, mineral carbonate-depleted water, may be treated using any suitable procedure. In some embodiments, it may be sent to tailings ponds for disposal. In certain embodiments, it can be processed in naturally occurring bodies of water, eg, oceans, seas, lakes or rivers. In certain embodiments, it can be used as a coolant for industrial equipment, for example, by a process line running between a sedimentation system and the industrial equipment. In certain embodiments, it can be used as gray water, water input for desalination, and subsequently used as fresh water, eg, in irrigation, for human and animal consumption, and the like. Therefore, contemplated are arrangements in which the precipitation unit is co-located with the desalination unit such that output water from the precipitation unit is used as the injection water for the desalination unit.
如上所述,在某些实施方案中,通过沉淀过程所产生的母液可以用于冷却提供CO2源的发电厂,例如,在一次通过性冷却系统中。在这样的实施方案中,在该方法中获得的热量然后可以反向循环到沉淀装置用于进一步使用,根据期望。在这样的实施方案中,原始的水源可以来自工业设备。这样的实施方案可以被改变而使用由工业设备提供的抽吸能力,例如,以提高总效率。As noted above, in certain embodiments, the mother liquor produced by the precipitation process can be used to cool a power plant that provides a source of CO 2 , for example, in a once-through cooling system. In such an embodiment, the heat obtained in the process can then be recycled back to the precipitation unit for further use, as desired. In such embodiments, the primary water source may be from an industrial facility. Such embodiments may be modified to use the suction capabilities provided by industrial equipment, for example, to increase overall efficiency.
在期望的情况下并且在产生CO2-埋存产品后,例如,如上所述,在产品中埋存的CO2的量被量化。“量化”是指确定已经埋存(即,固定)在CO2埋存产品中的CO2的量,例如,以数值的形式。确定可以是产品的绝对量化,在期望的情况下,或者其可以是近似量化,即,非精确的。在一些实施方案中,量化适于给出埋存的CO2的量的市场-可接受的量度。Where desired and after production of the CO 2 -storage product, eg, as described above, the amount of CO 2 stored in the product is quantified. "Quantitating" refers to determining the amount of CO2 that has been stored (ie, fixed) in a CO2 storage product, eg, in numerical form. The determination may be an absolute quantification of the product, where desired, or it may be an approximate quantification, ie imprecise. In some embodiments, quantification is suitable to give a market-accepted measure of the amount of CO 2 stored.
使用任何便利方法,CO2-埋存产品中CO2的量可以被量化。在某些实施方案中,通过组合物的实际测量,可以进行量化。在这些实施方案中,可以使用各种不同的方法。例如,测量组合物的质量或体积。在某些实施方案中,在沉淀物在母液中时,可以进行这样的测量。在这些情况下,可以使用另外的方法如X射线衍射来量化产品。在其它实施方案中,在沉淀物已经被洗涤和/或干燥后,进行测量。然后使用测量来量化在产品中埋存的CO2的量,例如,通过数学计算。例如,可以使用电量计来获得在沉淀的埋存产品中碳量的读数。可以使用这种电量计读数来确定沉淀物中的碳酸盐的量,其然后可以被转变为埋存的CO2,通过化学计量法,基于多个因素,如水的原始的金属离子含量,化学反应的限制试剂,反应的起始原料的理论收率,沉淀的产物的水化水等等。在一些实施方案中,污染物可以存在于产品中,产品纯度的其它测定,例如,元素分析,可能是确定所埋存的CO2的量所必需的。The amount of CO 2 in the CO 2 -storage product can be quantified using any convenient method. In certain embodiments, quantification can be performed by actual measurement of the composition. In these embodiments, a variety of different methods can be used. For example, measuring the mass or volume of a composition. In certain embodiments, such measurements can be performed while the precipitate is in the mother liquor. In these cases, additional methods such as X-ray diffraction can be used to quantify the product. In other embodiments, the measurement is performed after the precipitate has been washed and/or dried. The measurements are then used to quantify the amount of CO2 stored in the product, for example, by mathematical calculations. For example, a coulometer can be used to obtain a reading of the amount of carbon in the precipitated sequestered product. This coulometer reading can be used to determine the amount of carbonate in the sediment, which can then be converted to sequestered CO2 , by stoichiometry, based on factors such as the original metal ion content of the water, the chemical The limiting reagent for the reaction, the theoretical yield of the starting material for the reaction, the water of hydration for the precipitated product, etc. In some embodiments, contaminants may be present in the product and other determinations of product purity, eg, elemental analysis, may be necessary to determine the amount of CO2 being stored.
在其它实施方案中,使用同位素方法来确定产品的碳含量。化石燃料中的碳同位素的比值基本上不同于这样的同位素在地质源如石灰石中的这样的同位素的比值。因此,样品中的碳的来源或来源比值容易地通过质谱来阐明,所述质谱定量地测量同位素质量。因此即使石灰石集料用于混凝土(其将增加通过电量分析所确定的总碳量),使用同位素分析用的质谱将允许阐明可归因于来自化石燃料燃烧的捕获的CO2的碳的量。如此,沉淀物或甚至结合沉淀物的下游产物,例如,混凝土,中埋存的碳的量可以被确定,特别地其中用于制造沉淀物的CO2气体从化石燃料例如煤的燃烧获得。这种同位素方法的益处包括确定纯的沉淀物以及引入到另一产品中的沉淀物,以混凝土中的集料或沙的形式,等的碳含量的能力。In other embodiments, isotopic methods are used to determine the carbon content of the product. The ratios of carbon isotopes in fossil fuels differ substantially from the ratios of such isotopes in geological sources such as limestone. Thus, the source or source ratio of carbon in a sample is readily elucidated by mass spectrometry, which quantitatively measures isotopic masses. Thus even though limestone aggregates are used in concrete (which would add to the total carbon determined by coulometric analysis), the use of mass spectrometry for isotope analysis will allow elucidation of the amount of carbon attributable to captured CO2 from fossil fuel combustion. In this way, the amount of carbon sequestered in sediments or even downstream products associated with sediments, eg concrete, can be determined, particularly where the CO2 gas used to make the sediments is obtained from the combustion of fossil fuels such as coal. Benefits of this isotopic approach include the ability to determine the carbon content of pure precipitates as well as precipitates introduced into another product, in the form of aggregates or sand in concrete, and the like.
在其它实施方案中,通过进行埋存的CO2的量的理论测定,如通过计算埋存的CO2的量,可以进行量化。通过使用以上所述的方法的已知的收率,如其中收率是由早先的实验过程已知的,可以计算埋存的CO2的量。根据许多因素,可以改变已知的收率,包括以下中的一个或多个:气体(例如CO2)和水的输入,金属离子(例如,碱土金属离子)的浓度,pH,盐度,温度,气体物流的速率,所选择的方法的实施方案等等,如以上所述。可以使用,标准信息,例如,埋存的CO2的预定量/由给定的参考方法所产生的产品的量,容易地确定在给定方法中埋存的CO2的数量,其相同于或者大致近似于参考方法,例如,通过确定所产生的量并随后计算必需埋存其中的CO2的量。In other embodiments, quantification may be performed by making a theoretical determination of the amount of CO2 stored, such as by calculating the amount of CO2 stored. By using the known yield of the method described above, such as where the yield is known from an earlier experimental procedure, the amount of sequestered CO can be calculated. Known yields can vary depending on a number of factors, including one or more of the following: gas (e.g., CO2 ) and water input, concentration of metal ions (e.g., alkaline earth metal ions), pH, salinity, temperature , the rate of the gas stream, the selected method embodiment, etc., as described above. The amount of CO2 stored in a given process can be easily determined using, standard information, e.g., predetermined amount of CO2 stored/amount of product produced by a given reference process, which is the same as or Roughly approximating the reference method, for example, by determining the amount produced and then calculating the amount of CO2 that must be stored therein.
CO2埋存的系统 CO2 storage system
本发明的方面进一步包括系统,例如,加工装置或工厂,其用于例如通过实施如上所述的方法来埋存CO2。本发明的系统可以具有任何能够实施所考虑的特定的生产方法的配置结构。Aspects of the invention further include systems, eg, process plants or plants, for sequestering CO2 , eg, by implementing the methods described above. The system of the invention may have any configuration capable of implementing the particular production method under consideration.
在一些实施方案中,本发明提供用于处理二氧化碳的系统,如图1所示,其中该系统包括处理器(110),其被配置用于基于水性的方法来使用质子-除去剂源(140)加工来自二氧化碳源(130)的二氧化碳,和其中二氧化碳源包括一个或多个除二氧化碳之外的另外的组分。如图1所示,该系统可以进一步包括二价阳离子源(150),其可操作地连接到处理器。处理器可以包括接触器如气-液或气-液-固接触器,其中接触器被配置用于用二氧化碳加料水溶液或浆液以产生二氧化碳-加料的组合物,该组合物可以是溶液或浆液。在一些实施方案中,接触器被配置以从二氧化碳如由溶剂化或水合形式的二氧化碳(例如,碳酸,碳酸氢盐,碳酸盐)产生组合物,其中组合物包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐。在一些实施方案中,处理器可以进一步包括反应器,其被配置以从二氧化碳产生包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的组合物。在一些实施方案中,处理器可以进一步包括澄清槽,其被配置来沉降组合物,后者包括包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的沉淀材料。如图2所示,该系统可以进一步包括处理系统(例如,图2的处理系统120),其被配置以浓缩组合物,后者包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐,并且产生上层清液;然而,在一些实施方案中在没有进一步处理的情况下使用组合物。例如,在建筑材料的制造中,本发明的系统可以被配置以直接使用来自处理器的组合物(任选地使用极少的后处理)。在另一非限制性实例中,本发明的系统可以被配置以直接喷射来自处理器的组合物(任选地使用极少的后处理)到地下位置,如美国临时专利申请61/232,401(2009年8月7日申请,该申请全盘引入本文作为参考)中所述。二氧化碳源可以是任何各种二氧化碳的工业源,包括但不限于燃煤的发电厂和水泥厂。质子-除去剂源可以是任何各种质子-除去剂源,包括但不限于,质子-除去剂天然源和工业质子-除去剂源(包括工业废物源)。二价阳离子的来源可以是来自任何各种二价阳离子源,包括但不限于,海水,盐水和添加矿物的淡水。在这样的实施方案中,二价阳离子的来源可以可操作地连接到质子-除去剂源或直接连接到处理器。在一些实施方案中,二价阳离子的来源包括碱土金属的二价阳离子(例如,Ca2+,Mg2+)。In some embodiments, the present invention provides a system for processing carbon dioxide, as shown in FIG. ) processing carbon dioxide from a carbon dioxide source (130), and wherein the carbon dioxide source includes one or more additional components other than carbon dioxide. As shown in Figure 1, the system may further include a source of divalent cations (150) operatively connected to the processor. The processor may include a contactor such as a gas-liquid or gas-liquid-solid contactor, wherein the contactor is configured to feed an aqueous solution or slurry with carbon dioxide to produce a carbon dioxide-fed composition, which may be a solution or a slurry. In some embodiments, the contactor is configured to produce a composition from carbon dioxide, such as from solvated or hydrated forms of carbon dioxide (e.g., carbonic acid, bicarbonate, carbonate), wherein the composition includes carbonate, bicarbonate Or carbonates and bicarbonates. In some embodiments, the processor can further include a reactor configured to produce a composition comprising carbonate, bicarbonate, or carbonate and bicarbonate from carbon dioxide. In some embodiments, the processor may further include a clarifier configured to settle the composition comprising precipitation material including carbonates, bicarbonates, or carbonates and bicarbonates. As shown in FIG. 2, the system may further include a treatment system (e.g.,
本发明的系统如图1中所示的那种可以进一步包括处理系统。因而,在一些实施方案中,本发明提供用于处理二氧化碳的系统,如图2所示,其中该系统包括处理器(110)和处理系统(120),其被配置用于基于水性的方法来使用质子-除去剂源(140)加工来自二氧化碳源(130)的二氧化碳,和其中二氧化碳源包括一个或多个除二氧化碳之外的另外的组分。如同图1一样,图2的系统可以进一步包括二价阳离子源(150),其可操作地连接到处理器。处理器可以包括接触器如气-液或气-液-固接触器,其中接触器被配置用于用二氧化碳加料水溶液或浆液以产生二氧化碳-加料的组合物,该组合物可以是溶液或浆液。在一些实施方案中,接触器被配置以从二氧化碳如由溶剂化或水合形式的二氧化碳(例如,碳酸,碳酸氢盐,碳酸盐)产生组合物,其中组合物包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐。在一些实施方案中,处理器可以进一步包括反应器,其被配置以从二氧化碳产生包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的组合物。在一些实施方案中,处理器可以进一步包括澄清槽,其被配置来沉降组合物,后者包括包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的沉淀材料。处理系统可以包括脱水系统,其被配置以浓缩组合物,后者包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐。处理系统可以进一步包括过滤系统,其中过滤系统包括至少一个过滤单元,其被配置用于过滤来自脱水系统的上层清液,过滤来自处理器的组合物,或其组合。例如,在一些实施方案中,过滤系统包括一种或多种过滤单位,其选自微滤单元,超滤单元,纳滤单元,和反渗透单元。在一些实施方案中,二氧化碳处理系统包括纳滤单元,其被配置以增加渗余物中二价阳离子的浓度并且降低渗余物中二价阳离子的浓度。在这样的实施方案中,纳滤单元渗余物可以再循环到系统的处理器以便产生本发明的组合物。如图4所示,本发明的系统可以进一步被配置以再循环至少一部分来自处理系统的上层清液。The system of the present invention such as that shown in FIG. 1 may further include a processing system. Thus, in some embodiments, the present invention provides a system for processing carbon dioxide, as shown in Figure 2, wherein the system includes a processor (110) and a processing system (120) configured for an aqueous-based Carbon dioxide from a carbon dioxide source (130) is processed using a proton-removing agent source (140), and wherein the carbon dioxide source includes one or more additional components other than carbon dioxide. As with Figure 1, the system of Figure 2 may further include a source of divalent cations (150) operatively connected to the processor. The processor may include a contactor such as a gas-liquid or gas-liquid-solid contactor, wherein the contactor is configured to feed an aqueous solution or slurry with carbon dioxide to produce a carbon dioxide-fed composition, which may be a solution or a slurry. In some embodiments, the contactor is configured to produce a composition from carbon dioxide, such as from solvated or hydrated forms of carbon dioxide (e.g., carbonic acid, bicarbonate, carbonate), wherein the composition includes carbonate, bicarbonate Or carbonates and bicarbonates. In some embodiments, the processor can further include a reactor configured to produce a composition comprising carbonate, bicarbonate, or carbonate and bicarbonate from carbon dioxide. In some embodiments, the processor may further include a clarifier configured to settle the composition comprising precipitation material including carbonates, bicarbonates, or carbonates and bicarbonates. The treatment system may include a dehydration system configured to concentrate compositions including carbonates, bicarbonates, or carbonates and bicarbonates. The treatment system may further include a filtration system, wherein the filtration system includes at least one filtration unit configured to filter the supernatant from the dewatering system, filter the composition from the processor, or a combination thereof. For example, in some embodiments, the filtration system includes one or more filtration units selected from the group consisting of microfiltration units, ultrafiltration units, nanofiltration units, and reverse osmosis units. In some embodiments, the carbon dioxide treatment system includes a nanofiltration unit configured to increase the concentration of divalent cations in the retentate and decrease the concentration of divalent cations in the retentate. In such an embodiment, the nanofiltration unit retentate can be recycled to the processor of the system in order to produce the composition of the invention. As shown in Figure 4, the system of the present invention can be further configured to recycle at least a portion of the supernatant from the treatment system.
系统如图3中所示的那种可以进一步包括处理器(110),其包括接触器(112)(例如,气-液接触器,气-液-固接触器,等等)和反应器(114),其中处理器可操作地连接到含CO2气体源(130),质子-除去剂源(140),和二价阳离子源(150)。本发明的这样的系统被配置用于基于水性的处理来自二氧化碳源的二氧化碳,使用质子-除去剂的来源和二价阳离子的来源,其中二氧化碳源包括一个或多个除二氧化碳之外的另外的组分。接触器(112)可以可操作地连接到二氧化碳源(130)和质子-除去剂源(140)中的每一个,接触器可以被配置用于用二氧化碳加料水溶液或浆液以产生二氧化碳-加料的溶液或浆液。在一些实施方案中,接触器被配置以加料水溶液以二氧化碳以产生基本上澄清溶液(即,基本上没有沉淀材料,如至少95%或更多没有)。如图3所示,反应器(114)可以可操作地连接到接触器(112)和二价阳离子的来源(150),和反应器可以被配置以产生本发明的组合物,其中组合物是包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的溶液或浆液。在一些实施方案中,反应器被配置以接收来自处理器的碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的基本上澄清溶液并且产生包括沉淀材料的组合物(例如,二价阳离子的碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的浆液)。系统如图3中所示的可以任选地可操作地连接到处理系统,该处理系统可以包括液-固分离器(122)或一些其它脱水系统,其被配置以处理处理器-产生的组合物以产生上层清液和浓缩组合物(例如,相对碳酸盐和/或碳酸氢盐浓缩,和任何其它源于加工工业废气物流的辅助产物)。处理系统可以进一步包括过滤系统,其中过滤系统包括至少一个过滤单元,其被配置用于过滤来自脱水系统的上层清液,过滤来自处理器的组合物,或其组合。A system such as that shown in FIG. 3 may further include a processor (110) comprising a contactor (112) (e.g., a gas-liquid contactor, a gas-liquid-solid contactor, etc.) and a reactor ( 114), wherein the processor is operatively connected to a source of CO 2 -containing gas (130), a source of proton-removing agent (140), and a source of divalent cations (150). Such systems of the present invention are configured for aqueous based treatment of carbon dioxide from a carbon dioxide source using a source of proton-removing agents and a source of divalent cations, wherein the carbon dioxide source comprises one or more additional components other than carbon dioxide point. A contactor (112) can be operably connected to each of the carbon dioxide source (130) and the proton-removing agent source (140), and the contactor can be configured for feeding the aqueous solution or slurry with carbon dioxide to produce a carbon dioxide-fed solution or slurry. In some embodiments, the contactor is configured to feed the aqueous solution with carbon dioxide to produce a substantially clear solution (ie, substantially free of precipitation material, such as at least 95% or more free). As shown in Figure 3, the reactor (114) can be operably connected to the contactor (112) and the source (150) of divalent cations, and the reactor can be configured to produce the composition of the present invention, wherein the composition is Solutions or slurries of carbonates, bicarbonates or carbonates and bicarbonates are included. In some embodiments, the reactor is configured to receive carbonate, bicarbonate, or a substantially clear solution of carbonate and bicarbonate from the processor and produce a composition comprising precipitation material (e.g., divalent cation carbonate, bicarbonate or carbonate and bicarbonate slurry). The system as shown in Figure 3 may optionally be operably connected to a processing system which may include a liquid-solid separator (122) or some other dewatering system configured to treat the combined processor-generated to produce a supernatant and a concentrated composition (eg, concentrated relative to carbonate and/or bicarbonate, and any other by-products derived from processing industrial waste streams). The treatment system may further include a filtration system, wherein the filtration system includes at least one filtration unit configured to filter the supernatant from the dewatering system, filter the composition from the processor, or a combination thereof.
在一些实施方案中,本发明提供用于处理二氧化碳的系统,如图4所示,其中该系统包括处理器(110)和处理系统(120),其被配置用于基于水性的方法来使用质子-除去剂源(140)加工来自二氧化碳源(130)的二氧化碳,其中二氧化碳源包括一个或多个除二氧化碳之外的另外的组分,和进一步其中处理器和处理系统可操作地连接以便再循环至少一部分处理系统上层清液。这样的二氧化碳-处理系统的处理系统可以包括脱水系统和过滤系统。因而,脱水系统、过滤系统或脱水系统和过滤系统的组合可以被配置以提供至少一部分上层清液给处理器用于加工二氧化碳。尽管未示于图4中,处理系统也可被配置以提供至少一部分上层清液给洗涤系统,后者被配置以洗涤本发明的组合物,其中组合物包括沉淀材料(例如,CaCO3,MgCO3或其组合)。本发明的二氧化碳-处理系统的处理器可以被配置以接收以下中的处理系统上层清液:接触器(例如,气-液接触器,气-液-固接触器),反应器,接触器和反应器的组合,或者处理器中的任何其它单元或单位的组合。在一些实施方案中,二氧化碳-处理系统被配置以提供至少一部分上层清液给在二氧化碳处理系统以外的系统或工艺。例如,本发明的系统可以可操作地连接到脱盐装置,使得系统提供至少一部分处理系统上层清液给脱盐装置用于脱盐。In some embodiments, the present invention provides a system for processing carbon dioxide, as shown in Figure 4, wherein the system includes a processor (110) and a processing system (120) configured for an aqueous-based method using protons - The remover source (140) processes carbon dioxide from the carbon dioxide source (130), wherein the carbon dioxide source includes one or more additional components other than carbon dioxide, and further wherein the processor and treatment system are operatively connected for recycling At least a portion of the system supernatant is disposed of. The treatment systems of such carbon dioxide-treatment systems may include dehydration systems and filtration systems. Thus, the dehydration system, the filtration system, or a combination of the dehydration system and the filtration system may be configured to provide at least a portion of the supernatant to the processor for processing carbon dioxide. Although not shown in FIG. 4, the treatment system can also be configured to provide at least a portion of the supernatant to a washing system configured to wash the composition of the present invention, wherein the composition includes precipitation material (e.g., CaCO 3 , MgCO 3 or a combination thereof). The processor of the carbon dioxide-treatment system of the present invention can be configured to receive the treatment system supernatant in the following: contactors (e.g., gas-liquid contactors, gas-liquid-solid contactors), reactors, contactors, and A combination of reactors, or any other unit or combination of units in a processor. In some embodiments, the carbon dioxide-treatment system is configured to provide at least a portion of the supernatant to a system or process external to the carbon dioxide treatment system. For example, the system of the present invention can be operatively connected to a desalination unit such that the system provides at least a portion of the treatment system supernatant to the desalination unit for desalination.
在一些实施方案中,本发明提供用于处理二氧化碳的系统,如图5所示,其中该系统包括处理器(110)和处理系统(120),其被配置用于基于水性的方法来使用质子-除去剂源(140)加工来自二氧化碳源(130)的二氧化碳,其中二氧化碳源包括一个或多个除二氧化碳之外的另外的组分,其中系统进一步包括电化学系统(160),和进一步其中处理器,处理系统,和电化学系统可操作地连接以便再循环至少一部分处理系统上层清液。参考图4的处理系统如上所述,脱水系统、过滤系统或脱水系统和过滤系统的组合可以被配置以提供至少一部分处理系统上层清液给处理器用于加工二氧化碳。处理系统也可被配置以提供至少一部分处理系统上层清液给电化学系统,其中电化学系统可以被配置以产生质子-除去剂或进行质子除去。参考图4所述,处理系统也可被配置以提供至少一部分上层清液给洗涤系统,后者被配置以洗涤本发明的组合物,其中组合物包括沉淀材料(例如,CaCO3,MgCO3或其组合)。本发明的二氧化碳-处理系统的处理器可以被配置以接收以下中的处理系统上层清液或电化学系统物流:接触器(例如,气-液接触器,气-液-固接触器),反应器,接触器和反应器的组合,或者处理器中的任何其它单元或单位的组合。在一些实施方案中,二氧化碳-处理系统可以被配置以提供至少一部分上层清液给在二氧化碳处理系统以外的系统(例如,脱盐装置)或工艺(例如,脱盐)。In some embodiments, the present invention provides a system for processing carbon dioxide, as shown in FIG. 5, wherein the system includes a processor (110) and a processing system (120) configured for an aqueous based method using protons - the removal agent source (140) processes carbon dioxide from the carbon dioxide source (130), wherein the carbon dioxide source includes one or more additional components other than carbon dioxide, wherein the system further comprises an electrochemical system (160), and further wherein the processing The device, treatment system, and electrochemical system are operatively connected to recycle at least a portion of the treatment system supernatant. As described above with reference to the treatment system of FIG. 4 , the dewatering system, the filtration system, or a combination of the dehydration system and the filtration system can be configured to provide at least a portion of the treatment system supernatant to the processor for processing carbon dioxide. The treatment system can also be configured to provide at least a portion of the treatment system supernatant to an electrochemical system, where the electrochemical system can be configured to generate a proton-removing agent or perform proton removal. As described with reference to FIG. 4 , the treatment system can also be configured to provide at least a portion of the supernatant to a washing system configured to wash the composition of the present invention, wherein the composition includes precipitation material (e.g., CaCO 3 , MgCO 3 or its combination). The processor of the carbon dioxide-processing system of the present invention may be configured to receive a processing system supernatant or an electrochemical system stream from: a contactor (e.g., a gas-liquid contactor, a gas-liquid-solid contactor), a reaction reactor, combination of contactor and reactor, or any other unit or combination of units in a processor. In some embodiments, the carbon dioxide-processing system can be configured to provide at least a portion of the supernatant to a system (eg, desalination plant) or process (eg, desalination) outside the carbon dioxide processing system.
处理系统上层清液的再循环是有益的,因为再循环提供了可用资源的有效利用;周围环境的最小干扰;和降低的能量需要,所述降低的能量需要提供了本发明的系统和方法的更低的(例如,小的、中性的或负性的)碳足迹。当本发明的二氧化碳-处理系统可操作地连接到工业设备(例如,燃烧化石燃料的发电厂如燃煤的发电厂)并且使用在工业设备处产生的电力,由处理系统上层清液的再循环提供的降低的能量需要提供了降低的工业设备的附加负载。未配置用于再循环的二氧化碳-处理系统(即,配置用于单程法的二氧化碳处理系统)如图2中所示的那种可以具有至少10%的工业设备的附加负载,这可归因于连续地将碱度的新鲜源(例如,海水,盐水)泵入系统。在这样的实例中,100兆瓦发电厂(例如,燃煤的发电厂)将需要将10兆瓦的电力提供给二氧化碳-处理系统用于连续地将碱度的新鲜源泵入系统。相比之下,用于再循环配置的系统如图4或图5中所示的那种可以具有小于10%,如小于8%,包括小于6%,例如,小于4%或者少于2%的工业设备的附加负载,该附加负载可以是可归因于泵送补给水和再循环上层清液的。为再循环配置的二氧化碳-处理系统可以,当相比于为单程法设计的系统时,显示出至少2%,如至少5%,包括至少10%,例如,至少25%或至少50%的附加负载的降低。例如,如果为再循环配置的二氧化碳-处理系统消耗9兆瓦的电力来泵送补给水和再循环上层清液并且为单程法设计的二氧化碳-处理系统消耗可归因于泵送的10兆瓦,那么为再循环配置的二氧化碳-处理系统显示出10%的附加负载的下降。对于如图4和5中所示的那些的系统(即,为再循环配置的二氧化碳-处理系统)来说,可归因于泵送和再循环的附加负载的下降也可提供总附加负载的下降,特别地当相比于为单程法配置的二氧化碳-处理系统时。在一些实施方案中,再循环提供了二氧化碳-处理系统的总附加负载的下降,其中下降为至少2%,如至少4%,包括至少6%,例如至少8%或至少10%,当相比于为单程法配置的二氧化碳-处理系统的总附加负载时。例如,如果为再循环配置的二氧化碳-处理系统具有15%的附加负载并且为单程法设计的二氧化碳-处理系统具有20%附加负载,那么为再循环配置的二氧化碳-处理系统显示出5%的总附加负载的下降。例如,为再循环配置的二氧化碳-处理系统,其中再循环包括通过过滤单元如纳滤单元的过滤(例如,以便在渗余物中浓缩二价阳离子和降低在渗透物中的二价阳离子),可以具有至少2%,如至少4%,包括至少6%,例如至少8%或至少10%的总附加负载的下降,当相比于为单程法配置的二氧化碳-处理系统时。Recirculation of the supernatant of the treatment system is beneficial because the recirculation provides efficient use of available resources; minimal disturbance of the surrounding environment; and reduced energy requirements that provide the benefits of the systems and methods of the present invention. Lower (eg, small, neutral or negative) carbon footprint. When the carbon dioxide-processing system of the present invention is operably connected to an industrial facility (e.g., a fossil fuel-fired power plant such as a coal-fired power plant) and uses electricity generated at the industrial facility, the recirculation of supernatant liquid from the treatment system The reduced energy requirements provided provide for reduced parasitic loads on industrial equipment. A carbon dioxide-processing system not configured for recirculation (i.e., a carbon dioxide processing system configured for a one-pass process) such as that shown in Figure 2 may have an additional load of at least 10% of the industrial plant attributable to A fresh source of alkalinity (eg, seawater, brine) is continuously pumped into the system. In such an example, a 100 MW power plant (eg, a coal-fired power plant) would need to provide 10 MW of power to the carbon dioxide-processing system for continuously pumping a fresh source of alkalinity into the system. In contrast, a system for a recirculation configuration such as that shown in Figure 4 or Figure 5 may have less than 10%, such as less than 8%, including less than 6%, for example, less than 4% or less than 2% The additional load of the industrial equipment can be attributable to pumping make-up water and recirculating supernatant. A carbon dioxide-processing system configured for recirculation may, when compared to a system designed for a single pass process, exhibit an additional load reduction. For example, if a CO2-treatment system configured for recirculation consumes 9 MW of electricity to pump the make-up water and recirculate the supernatant and a CO2-treatment system designed for the single-pass process consumes 10 MW of electricity attributable to pumping , then a CO2-treatment system configured for recirculation shows a 10% drop in parasitic load. For systems such as those shown in Figures 4 and 5 (i.e., carbon dioxide-processing systems configured for recirculation), the drop in parasitic load attributable to pumping and recirculation also provides an estimate of the total parasitic load. decline, especially when compared to carbon dioxide-processing systems configured for single-pass processes. In some embodiments, recirculation provides a reduction in the total additional load of the carbon dioxide-processing system, wherein the reduction is at least 2%, such as at least 4%, including at least 6%, such as at least 8% or at least 10%, when compared to At the total additional load of the carbon dioxide-treatment system configured for the single-pass method. For example, if a CO2-treatment system configured for recirculation has an additional load of 15% and a CO2-treatment system designed for a one-pass process has an additional load of 20%, then a CO2-treatment system configured for recirculation shows an overall load of 5%. drop in additional load. For example, a carbon dioxide-processing system configured for recirculation, wherein recirculation includes filtration through a filtration unit such as a nanofiltration unit (e.g., to concentrate divalent cations in the retentate and reduce divalent cations in the permeate), There may be a reduction in total parasitic load of at least 2%, such as at least 4%, including at least 6%, such as at least 8% or at least 10%, when compared to a carbon dioxide-processing system configured for a single pass process.
图6B提供了根据本发明的实施方案的CO2-处理系统的示意图。如本文中所述,以及图6B中提供的,本发明的系统可以包括含CO2气体源(例如,工业废气物流如来自燃煤的发电厂的烟道气),碱度源(例如,质子-除去剂源),和二价阳离子源。含CO2的气体源,碱度源,和二价阳离子源可以各自可操作地连接到CO2处理器如本文中所述的吸收器,或者如图6B所示,含CO2的气体源可以可操作地连接到热交换(HX)干燥器,后者被配置以干燥沉淀材料,该干燥器又可操作地连接到CO2处理器。CO2处理器可以被配置以便气-液或气-液-固接触并且包括气-液接触器,气-液-固接触器,反应器,澄清槽,或其任何组合,进行如本文中所述的吸收。CO2处理器(例如,吸收器)可以被配置以提供包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的组合物(例如,溶液,浆液,等等)给本发明的处理系统,其,如提供于图6B中的实施方案所示,包括用于脱水,水处理,氯化物除去,干燥,和岩化的系统。脱水系统可以被配置以除去本体水,产生脱水的沉淀材料,其,取决于二价阳离子源和/或碱度源,可以包括氯化物。被配置以使用由水处理系统(例如,一种或多种过滤单元,其选自以下过滤单元:微滤、超滤、纳滤、反渗透、正向渗透过滤单元)提供的水,氯化物除去系统可以除去氯化物,和取决于最终产品,来自沉淀材料的其它盐。如图6B所述的系统可以进一步被配置以提供贫乏氯化物的沉淀材料以便在SCM干燥系统中随后干燥而提供辅助的粘结材料(SCM)。备选地,或者,另外,系统可以被配置以提供贫乏氯化物的沉淀材料给岩化系统,以便产生细集料粗集料。图6B还表明系统被配置以提供净化排气(例如,CO2-贫乏的废气物流(主要包括N2),淡水(例如,饮用水),盐水(例如,NaCl水溶液),和浓缩二价阳离子源,其包括由CO2处理留下的二价阳离子。Figure 6B provides a schematic diagram of a CO2 -processing system according to an embodiment of the invention. As described herein, and provided in Figure 6B, the system of the present invention can include a source of CO2 - containing gas (e.g., an industrial waste gas stream such as flue gas from a coal-fired power plant), a source of alkalinity (e.g., proton - a source of removing agent), and a source of divalent cations. The CO2 -containing gas source, the alkalinity source, and the divalent cation source can each be operably connected to a CO2 processor such as the absorber described herein, or as shown in Figure 6B, the CO2 -containing gas source can be is operatively connected to a heat exchange (HX) dryer configured to dry the precipitation material, which in turn is operatively connected to a CO2 processor. The CO processor can be configured for gas-liquid or gas-liquid-solid contacting and includes gas-liquid contactors, gas-liquid-solid contactors, reactors, clarifiers, or any combination thereof, to conduct as described herein the above-mentioned absorption. The CO processor (e.g., absorber) can be configured to provide a composition (e.g., solution, slurry, etc.) comprising carbonate, bicarbonate, or carbonate and bicarbonate to the treatment system of the present invention , which, as shown in the embodiment provided in Figure 6B, includes systems for dehydration, water treatment, chloride removal, drying, and lithification. The dehydration system may be configured to remove bulk water, resulting in dehydrated precipitation material, which, depending on the source of divalent cations and/or the source of alkalinity, may include chloride. configured to use water provided by a water treatment system (e.g., one or more filtration units selected from the group consisting of microfiltration, ultrafiltration, nanofiltration, reverse osmosis, forward osmosis filtration units), chloride The removal system can remove chlorides, and depending on the final product, other salts from the precipitation material. The system as depicted in FIG. 6B can be further configured to provide chloride-depleted precipitation material for subsequent drying in an SCM drying system to provide supplemental cementitious material (SCM). Alternatively, or in addition, the system may be configured to provide chloride-depleted precipitation material to the lithification system in order to produce fine aggregate coarse aggregate. Figure 6B also shows that the system is configured to provide purified exhaust (e.g., CO2 -poor exhaust gas stream (comprising mainly N2), fresh water (e.g., drinking water), salt water (e.g., NaCl aqueous solution), and a source of concentrated divalent cations , which includes divalent cations left over from CO2 treatment.
图6C提供了本发明的CO2处理器的更详细的描述。例如,在一些实施方案中,图6C提供了图6B的CO2处理器的更详细的描述。本发明的CO2处理器如图6B中所示的那种可以包括吸风风扇(ID风扇),其被配置以提供CO2工业源(例如,烟道气)给吸收器。吸收器可以进一步包括用于二价阳离子源和/或碱度源(例如,质子-除去剂源)的平衡箱。关于二价阳离子源,例如,二价阳离子源可以可操作地连接到平衡箱,后者被配置以调节潜在非连续流(例如,流动波动),该平衡箱可以可操作地连接到进料泵,后者被配置以将二价阳离子源泵送到吸收器。如图6C所示,CO2处理器可以进一步包括循环泵,其被配置以再循环来自吸收器底部的溶液或浆液至吸收器的一个或多个上一级。未再循环的溶液或浆液可以被提供到产品平衡箱,其连同可操作地连接的产品泵,被配置以提供调节流量的产品给本发明的脱水系统。该系统可以进一步被配置以中止再循环,将全部浆液的吸收溶液转移到产品平衡箱,用洗涤水洗涤吸收器。如同二价阳离子源和碱度源(例如,质子-除去剂源)一样,洗涤水可以由含洗涤水的水槽通过中间的泵被提供到吸收器。Figure 6C provides a more detailed description of the CO2 processor of the present invention. For example, in some embodiments, Figure 6C provides a more detailed description of the CO2 processor of Figure 6B. A CO2 processor of the present invention such as that shown in FIG. 6B may include an induction fan (ID fan) configured to provide an industrial source of CO2 (eg, flue gas) to the absorber. The absorber may further include a balance tank for a source of divalent cations and/or a source of alkalinity (eg, a source of proton-removing agent). With regard to the source of divalent cations, for example, the source of divalent cations can be operably connected to a balancing tank configured to accommodate potentially discontinuous flow (e.g., flow fluctuations), which can be operatively connected to a feed pump , the latter configured to pump a source of divalent cations to the absorber. As shown in Figure 6C, the CO2 processor may further include a circulation pump configured to recirculate the solution or slurry from the bottom of the absorber to one or more upper stages of the absorber. The non-recirculated solution or slurry can be provided to a product balance tank, which, along with an operably connected product pump, is configured to provide a regulated flow of product to the dewatering system of the present invention. The system can further be configured to stop the recirculation, transfer the entire slurry absorption solution to the product balance tank, and wash the absorber with wash water. As with the source of divalent cations and alkalinity (eg, proton-removing agent source), wash water can be supplied to the absorber from a tank containing wash water through an intermediate pump.
图7提供了根据本发明一个实施方案的系统的示意图。在图7中,系统100包括水源110。在某些实施方案中,水源110包括具有用于水(例如含碱土金属离子的水)的输入端如来自海洋等的管子或管道的结构。在由产生沉淀物的系统加工的水源是海水的情况下,输入端与海水源流体连通,例如,如其中输入端是来自海洋水至陆地基系统的管路或进料,或者在船的船体中的入口孔,例如,其中该系统属于船的一部分,例如,在海洋基系统中。Figure 7 provides a schematic diagram of a system according to one embodiment of the invention. In FIG. 7 ,
同样在图7中所示,是CO2源130。该系统还包括管,管道,或导管,其将CO2引导至系统100。用于本发明的方法中的气态废物物流可以以任何适宜方式由工业设备提供给沉淀位置,其将来自工业设备的气态废物物流输送给沉淀装置。在某些实施方案中,废物物流具有气体输送器,例如,管道,其从工业设备的位置,例如,工业设备的烟道到沉淀位置的一个或多个位置。气态废物物流的来源可以是相对于沉淀位置的远侧位置,使得气态废物物流的来源的位置距离沉淀位置达1英里或更大,如10英里或更大,包括100英里或更大。例如,气态废物物流可以从遥远的工业设备通过CO2气体运输系统,例如管道,运输到沉淀位置。工业设备产生的含CO2的气体可以或未必被加工,例如,除去其它组分,等,然后其到达沉淀位置(即,碳酸盐化合物沉淀装置)。在其它情况中,气态废物物流源邻近于沉淀位置,其中这样的情况可以包括其中沉淀位置集成以气态废物物流源的情况,如集成碳酸盐化合物沉淀反应器的发电厂。Also shown in FIG. 7 is a CO 2 source 130 . The system also includes tubes, conduits, or conduits that direct the CO 2 to the
在期望的情况下,在沉淀反应中可以使用一部分但是小于全部的来自工业设备的气态废物物流。在这些实施方案中,用于沉淀的气态废物物流的部分可以是75%或更少,如60%或更少和包括50%和更少。在其它实施方案中,在沉淀中使用基本上全部由工业设备产生的气态废物物流,例如基本上全部由工业设备产生的烟道气。在这些实施方案中,在沉淀期间可以使用由该来源产生的80%或更多,如90%或更多,包括95%或更多,直至100%的气态废物物流(例如,烟道气)。Where desired, a portion, but less than all, of the gaseous waste stream from the industrial plant may be used in the precipitation reaction. In these embodiments, the portion of the gaseous waste stream used for precipitation may be 75% or less, such as 60% or less and including 50% and less. In other embodiments, a substantially entirely gaseous waste stream produced by an industrial facility, such as substantially all flue gas produced by an industrial facility, is used in the precipitation. In these embodiments, 80% or more, such as 90% or more, including 95% or more, up to 100% of the gaseous waste stream (e.g., flue gas) produced by the source may be used during precipitation .
如上所指出,气态废物物流可以是从工业设备的烟道或同功结构中获得的气态废物物流。在这些实施方案中,作业线,例如,管道,连接到烟道以便气体通过作业线离开烟道并且被输送到沉淀系统的合适的位置(下文更详细地描述的)。取决于其中使用气态废物物流的沉淀系统的部分的特定的配置结构,从中获得气态废物物流的来源位置可以改变,例如,而提供具有合适的或期望的温度的废物物流。因而,在某些实施方案中,在具有0℃至1,800℃,如60℃至700℃的温度的气态废物物流是期望的情况下,烟道气在锅炉或燃气轮机,窑炉的出口点处,或者在通过发电厂或烟囱的任一点处,可以获得。在期望的情况下,烟道气被维持在露点以上的温度,例如,125℃,以免冷凝和有关的复杂情况。在这不可能的情况下,可以采取步骤来降低冷凝的不利影响,例如,使用不锈钢,氟烃(如聚(四氟乙烯))作业线的管道,用水稀释和pH控制,等等,如此,管道不迅速损坏。As noted above, the gaseous waste stream may be a gaseous waste stream obtained from a flue or co-function structure of an industrial facility. In these embodiments, a process line, eg, piping, is connected to the flue so that gas exits the flue through the process line and is transported to a suitable location in the precipitation system (described in more detail below). Depending on the particular configuration of the portion of the precipitation system in which the gaseous waste stream is used, the source location from which the gaseous waste stream is obtained may be changed, for example, to provide a waste stream having a suitable or desired temperature. Thus, in certain embodiments, where a gaseous waste stream having a temperature of 0°C to 1,800°C, such as 60°C to 700°C is desired, the flue gas is at the exit point of the boiler or gas turbine, kiln, Or at any point through a power plant or chimney, may be obtained. Where desired, the flue gas is maintained at a temperature above the dew point, eg, 125°C, to avoid condensation and related complications. Where this is not possible, steps can be taken to reduce the adverse effects of condensation, e.g., use of stainless steel, fluorocarbon (e.g. poly(tetrafluoroethylene)) line piping, dilution with water and pH control, etc., Pipes are not damaged quickly.
为提供效率,产生气态废物物流的工业设备可以与沉淀系统共处。“共处”是指工业设备和沉淀系统之间的距离为10至500码,如25至400码,包括30至350码。在期望的情况下,沉淀和工业设备可以被相互之间地配置以最小化温度损失和避免冷凝,以及最小化管道成本,例如,在沉淀装置位于工业设备的40码之内。To provide efficiencies, industrial equipment producing gaseous waste streams can be co-located with sedimentation systems. "Coexisting" means that the distance between the industrial equipment and the settling system is 10 to 500 yards, such as 25 to 400 yards, including 30 to 350 yards. Where desired, the settling and industrial equipment can be positioned relative to each other to minimize temperature loss and avoid condensation, as well as minimize piping costs, for example, where the settling unit is located within 40 yards of the industrial equipment.
还考虑的是在某些实施方案中,包括工业功能(如发电,水泥产生,等等)和本发明的沉淀系统的完全整合装置。在这样的整合装置中,常规的工业设备和沉淀系统,例如如下所述的,被改变以便提供期望的整合装置。改变包括但不局限于:协调烟囱,泵,控制,仪表设备,监控,使用装置能量,例如,蒸汽轮机能量来运转各部分的沉淀组件,例如,机械压力机,泵,压缩机,使用来自水泥和/或发电厂的从蒸汽中获得的热量或来自空气-空气换热器的热量,等等。Also contemplated are, in certain embodiments, fully integrated installations that include industrial functions (eg, power generation, cement production, etc.) and the precipitation system of the present invention. In such an integrated plant, conventional industrial equipment and precipitation systems, such as those described below, are modified to provide the desired integrated plant. Changes include, but are not limited to: coordination of chimneys, pumps, controls, instrumentation, monitoring, use of plant energy, e.g., steam turbine energy to run various parts of sedimentation components, e.g., mechanical presses, pumps, compressors, use of energy from cement And/or heat from steam or heat from air-to-air heat exchangers in power plants, etc.
在某些实施方案中,含CO2的气体物流可以被预先处理或预加工(例如,用H2O2处理),然后使其与水接触,例如含碱土金属的水(例如,在加料反应器中)。例证性的预处理或预加工步骤可以包括:温度调节(例如,加热或冷却),减压,挤压,引入另外的组分(例如,水合物促进剂气体),氧化各种组分而将其转化为更易于以稳定形式埋存的形式等等。在某些实施方案中,气态废物物流的预处理改善了含CO2的气体物流的组分吸收到水,例如含碱土金属的水。用于改善吸收的示范性的预处理包括使含CO2的气体物流经受氧化条件。In certain embodiments, the CO2 -containing gas stream can be pretreated or preprocessed (e.g., with H2O2 ) and then contacted with water, such as alkaline earth metal-containing water (e.g., in an addition reaction device). Exemplary pretreatment or preprocessing steps may include: temperature adjustment (e.g., heating or cooling), decompression, extrusion, introduction of additional components (e.g., hydrate promoter gas), oxidation of various components to It transforms into a form that is more easily sequestered in a stable form and so on. In certain embodiments, pretreatment of the gaseous waste stream improves absorption of components of the CO 2 -containing gas stream into water, such as alkaline earth metal-containing water. Exemplary pretreatments for improved absorption include subjecting the CO 2 -containing gas stream to oxidative conditions.
图7的水源110和CO2气体物流源130连接到沉淀反应器120中的CO2加料器。沉淀反应器120可以包括许多不同设计特征,如温度调节器(例如,被配置以加热水至期望的温度),化学添加剂组件,例如,用于引入化学pH提高剂(如氢氧化物,金属氧化物,或飞灰)到水中,电化学组件,例如,阴极/阳极,机械搅动和物理搅拌机构和再循环工业设备烟道气通过沉淀装置的组件。沉淀反应器120还可包含设计特点,其允许监控一种或多种参数如内部反应器压力,pH,沉淀物颗粒物尺寸,金属-离子浓度,水溶液的电导率,水溶液的碱度,和pCO2。这种反应器120可以以间歇过程或连续过程的方式操作。The
在一些实施方案中,接触器(例如气-液或气-液-固接触器)与系统相隔。在这样的系统中,在接触气体后吸收溶液被送到加工台。在一些实施方案中,在使气体和吸收溶液与气体的接触的任何产物接触后的吸收溶液被通到其它系统(即加工台),包括但不限于,沉淀槽,脱水系统和建筑制造系统。在一些实施方案中,在接触气体后的接触混合物,其包括液体组分,使接触混合物与气体接触的任何产物,和任选地并非沉淀物的固体组分,被通到其他系统,包括但不限于,沉淀槽,脱水系统和建筑制造系统。在一些实施方案中,在接触气体后的浆液,其包括液体组分,固体组分和使浆液与气体接触的任何产物,被通到其他系统,包括但不限于,沉淀槽,脱水系统和建筑制造系统。在一些实施方案中,吸收溶液的固体组分(如果有的话)和使吸收溶液与气体接触的产品与已经接触气体的吸收溶液分离。在一些实施方案中,接触混合物的固体组分(如果有的话)和使接触混合物与气体接触的产品与已经接触气体的接触混合物分离。在一些实施方案中,浆液的固体组分(如果有的话)和使浆液与气体接触的产品与已经接触气体的浆液分离。在一些实施方案中,分离固体和已经接触气体的吸收溶液,接触混合物,或浆液是通过筛、压机、离心机、喷雾干燥器、空气辅助方法、加热脱水方法或其任何组合来实现的。在一些实施方案中,在分离固体与已经接触气体的吸收溶液、接触混合物或浆液后剩余的流出物液体通过一种系统处理,所述系统包括但不局限于纳滤,反渗透,化学再生,脱盐,为再循环对溶液化学性质的调整,为释放的pH调整或其任何组合。在一些实施方案中,与已经接触气体的吸收溶液,接触混合物或浆液分离的固体被送到建筑材料生产系统。在一些实施方案中,装置是包含在运输容器中的便携式装置,使得其可以通过铁路(火车),水路(驳船)和/或公路(卡车)或其任何组合运输到任何期望的位置。在一些实施方案中,便携式仪表与系统分隔。在一些实施方案中,整个系统是包含在一个或多个运输容器中的便携式系统,其可以通过铁路(火车),水路(驳船)和/或公路(卡车)或其任何组合运输到任何期望的位置。In some embodiments, a contactor (eg, a gas-liquid or gas-liquid-solid contactor) is isolated from the system. In such systems, the absorbing solution is sent to the processing station after exposure to the gas. In some embodiments, the absorbing solution after contacting the gas and any products of contacting the absorbing solution with the gas is passed to other systems (ie, process stations), including, but not limited to, settling tanks, dehydration systems, and building fabrication systems. In some embodiments, the contacting mixture after contacting the gas, which includes liquid components, any products of contacting the contacting mixture with the gas, and optionally solid components other than precipitates, is passed to other systems, including but Not limited to, settling tanks, dewatering systems and building fabrication systems. In some embodiments, the gas-exposed slurry, which includes liquid components, solid components, and any products of contacting the slurry with the gas, is passed to other systems, including but not limited to, settling tanks, dewatering systems, and buildings manufacturing system. In some embodiments, the solid components of the absorption solution, if any, and the products of contacting the absorption solution with the gas are separated from the absorption solution that has been contacted with the gas. In some embodiments, the solid components (if any) of the contacting mixture and the products of contacting the contacting mixture with the gas are separated from the contacting mixture that has been contacted with the gas. In some embodiments, the solid components of the slurry, if any, and the products of contacting the slurry with the gas are separated from the slurry that has been contacted with the gas. In some embodiments, separation of the solids from the absorption solution that has been contacted with the gas, contacted mixture, or slurry is accomplished by sieves, presses, centrifuges, spray dryers, air-assisted methods, heat dehydration methods, or any combination thereof. In some embodiments, the effluent liquid remaining after separation of the solids from the gas-exposed absorption solution, contact mixture, or slurry is processed through a system including, but not limited to, nanofiltration, reverse osmosis, chemical regeneration, Desalination, adjustment of solution chemistry for recycle, pH adjustment for release, or any combination thereof. In some embodiments, the solids separated from the absorption solution, contact mixture or slurry that has been contacted with the gas are sent to the construction material production system. In some embodiments, the device is a portable device contained in a transport container such that it can be transported by rail (train), water (barge) and/or road (truck) or any combination thereof to any desired location. In some embodiments, the portable meter is separate from the system. In some embodiments, the entire system is a portable system contained in one or more transport containers, which can be transported by rail (train), waterway (barge) and/or road (truck) or any combination thereof to any desired destination. Location.
有时,为影响引入液体或浆液中的气体组分的量,气体需要接触液体或浆液比一次通过本发明的装置可能多的次数。在一些实施方案中,气体被再循环以便影响气体组分引入液体或浆液的量。可以使用多个装置来进行将气体组分引入液体或浆液,使得气体从一个装置通到一个或多个随后的装置。随后的装置可以利用不同的:液体或浆液;在装置的塔、室或反应器内的结构特征;液滴产生系统或装置;或具有与第一装置不同的总取向。在一些实施方案中,本发明的系统包括本发明的装置的阵列。在这样的实施方案中,阵列可以包括气体逐次通过其的装置,一个装置在另一个之后,或者阵列可以包括气体同时通过其的装置,使得该装置平行使用。在一些实施方案中,阵列包括若干排的多个装置。在一些实施方案中,气体同时地进入若干排队多个装置的第一装置中,然后流入随后的装置,使得有效地一系列装置平行工作。Sometimes, to affect the amount of the gas component introduced into the liquid or slurry, the gas needs to contact the liquid or slurry more times than is possible in one pass through the device of the invention. In some embodiments, the gas is recycled in order to affect the amount of gas components introduced into the liquid or slurry. The introduction of the gaseous components into the liquid or slurry may be performed using multiple devices, such that the gas passes from one device to one or more subsequent devices. Subsequent devices may utilize different: liquids or slurries; structural features within the towers, chambers, or reactors of the device; droplet generation systems or devices; or have a different general orientation than the first device. In some embodiments, systems of the invention include arrays of devices of the invention. In such embodiments, the array may include devices through which the gas is passed sequentially, one device after the other, or the array may include devices through which the gas is passed simultaneously such that the devices are used in parallel. In some embodiments, an array includes rows of multiple devices. In some embodiments, the gas enters simultaneously into a first device of several devices lined up and then flows into subsequent devices such that effectively a series of devices operate in parallel.
在一些实施方案中,本发明的系统设法最优化完成吸收所需要的马力和装置的物理足迹。在这样的实施方案中,系统包括用于使混合物(例如浆液或接触液体)与气体接触而除去一种或多种组分气体(例如CO2,SOx)的本发明的至少两个装置。第一装置是这样取向的,使得其长轴是水平的并且使得其置于离地面近。这种取向的目的在于这部分的吸收器将具有低液体压头要求,因此使得更易于泵送接触混合物(即吸收溶液)至装置的接触室的顶部,即需要较少能量。在装置的水平部分中接触混合物可以沿该部分的长度不同,或者其可以是相同的混合物。在装置的水平部分中的接触混合物可以是清液或浆液,其可以或未必被再循环。入口气体的流动和接触混合物的流动可以是顺流或逆流的,或者气体和溶液的流动可以在装置的水平长度中在级中变化。在水平取向的装置中的接触混合物可以在装置中进行再循环以便有效地引起相对于入口气体流动的接触混合物的逆向流动。系统中的第二装置是垂直取向的,使得其长度垂直于地面。垂直取向的装置可以是分级的(比不分级的更常见)并且接触混合物流动可以与入口气体的流动顺流或逆流。在气体出口前,吸收器的垂直段将具有除雾器。这段还可能将接触混合物再循环至垂直取向的装置中的各级或者至水平取向的装置。接触混合物还可能在若干下面的级中是清液或者浆液。浆液包括固体组分,其可以是矿物,工业废物(例如飞灰,水泥窑灰尘)和/或来自方法的固体沉淀物(在其中使用再循环的情况下)。如果接触混合物包括浆液,粉碎可以与再循环系统分隔。清液或浆液的液体组分可以是海水,天然存在的碱性盐水,工业废物盐水,脱盐流出物盐水,合成盐水,淡水,添加另外的二价阳离子的溶液,添加另外的碱度的溶液或其组合。In some embodiments, the system of the present invention seeks to optimize the horsepower required to absorb and the physical footprint of the device to accomplish this. In such embodiments, the system includes at least two devices of the invention for contacting a mixture (eg, slurry or contact liquid) with a gas to remove one or more component gases (eg, CO2 , SOx). The first device is oriented such that its long axis is horizontal and such that it is placed close to the ground. The purpose of this orientation is that this part of the absorber will have a low liquid head requirement, thus making it easier to pump the contact mixture (ie absorbing solution) to the top of the contact chamber of the device, ie requiring less energy. The contact mixture in a horizontal section of the device may be different along the length of that section, or it may be the same mixture. The contact mixture in the horizontal part of the plant may be a supernatant or a slurry, which may or may not be recycled. The flow of the inlet gas and the flow of the contacting mixture may be co-current or counter-current, or the flow of gas and solution may vary in stages over the horizontal length of the device. The contact mixture in the horizontally oriented apparatus may be recirculated within the apparatus to effectively induce a counter flow of the contact mixture relative to the inlet gas flow. The second device in the system is vertically oriented such that its length is perpendicular to the ground. Vertically oriented devices can be staged (more common than unstaged) and the contact mixture flow can be cocurrent or countercurrent to the flow of the inlet gas. The vertical section of the absorber will have demisters before the gas outlet. It is also possible for this section to recycle the contact mixture to stages in a vertically oriented plant or to a horizontally oriented plant. It is also possible that the contact mixture is a clear liquid or a slurry in several of the following stages. The slurry comprises solid components which may be minerals, industrial waste (eg fly ash, cement kiln dust) and/or solid sediment from the process (where recycling is used). If the contact mixture includes a slurry, comminution can be separated from the recirculation system. The liquid component of the serum or slurry may be seawater, naturally occurring alkaline brines, industrial waste brines, desalination effluent brines, synthetic brines, fresh water, solutions with added divalent cations, solutions with added alkalinity or its combination.
图26是本发明的实施方案的示意图,其中该系统包括两个装置,一个水平取向,离地面近,和第二个装置,垂直取向。图26(和同样地,图27和28)中所示的配置结构最小化了马力要求和系统的物理足迹要求。较低的,水平取向的装置需要较少的液体压头,并且因此需要较少的马力来操作。对于其物理足迹来说,较高的垂直取向的装置需要较少的面积。垂直取向的装置中的溶液可以再循环至水平取向的装置,仅仅在垂直取向的装置中再循环,在垂直取向的装置中部分地和在水平取向的装置中部分地再循环,或不进行再循环。垂直取向的装置可以具有除雾段,刚好在气体出口前,其中接受清液(即没有固体颗粒物)作为其吸入物。垂直取向的装置可以接受清液或者浆液作为其对于装置的主要部分的吸入物,在图中显示了3个喷雾器。包括两个装置的系统可以是便携式系统,使得该系统被包含在运输容器中,其可以通过铁路、水路和/或公路来运输。Figure 26 is a schematic diagram of an embodiment of the invention wherein the system includes two devices, one horizontally oriented, near the ground, and a second device, vertically oriented. The configuration shown in Figure 26 (and likewise, Figures 27 and 28) minimizes horsepower requirements and the physical footprint requirements of the system. A lower, horizontally oriented unit requires less hydraulic head, and thus requires less horsepower to operate. A taller vertically oriented device requires less area for its physical footprint. The solution in the vertically oriented device can be recycled to the horizontally oriented device, recycled only in the vertically oriented device, partially recycled in the vertically oriented device and partially recycled in the horizontally oriented device, or not recirculated cycle. A vertically oriented device may have a mist removal section, just before the gas outlet, which accepts clear liquid (ie, free of solid particulate matter) as its intake. The vertically oriented device can accept either serum or slurry as its intake to the main part of the device, 3 nebulizers are shown in the figure. A system comprising two devices may be a portable system such that the system is contained in a transport container which may be transported by rail, water and/or road.
图27是本发明的实施方案的示意图,类似于图26中所示的那种,其中该系统包括两个装置,一个水平取向,离地面近,和第二个装置,垂直取向。在图27中所示的实施方案中,第一装置中的气体的流动(其是水平取向的,离地面近)被强迫遵循旋转路径。气体所遵循的路径的性质产生了在第一(即,较低)装置中的气体和溶液之间的逆流和顺流接触的区域。如图26中,垂直取向的装置中的溶液可以再循环至水平取向的装置,仅仅在垂直取向的装置中再循环,在垂直取向的装置中部分地和在水平取向的装置中部分地再循环,或不进行再循环。垂直取向的装置可以具有除雾段,刚好在气体出口前,其中接受清液(即没有固体颗粒物)作为其吸入物。垂直取向的装置可以接受清液或者浆液作为其对于装置的主要部分的吸入物,在图中显示了3个喷雾器。包括两个装置的系统可以是便携式系统,使得该系统被包含在运输容器中,其可以通过铁路、水路和/或公路来运输。Figure 27 is a schematic illustration of an embodiment of the invention, similar to that shown in Figure 26, in which the system includes two devices, one horizontally oriented, near the ground, and a second device, vertically oriented. In the embodiment shown in Figure 27, the flow of gas in the first device (which is horizontally oriented, close to the ground) is forced to follow a rotational path. The nature of the path followed by the gas creates regions of countercurrent and cocurrent contact between the gas and the solution in the first (ie, lower) device. As in Figure 26, the solution in the vertically oriented device can be recycled to the horizontally oriented device, recycled only in the vertically oriented device, partially in the vertically oriented device and partially in the horizontally oriented device , or without recycling. A vertically oriented device may have a mist removal section, just before the gas outlet, which accepts clear liquid (ie, free of solid particulate matter) as its intake. The vertically oriented device can accept either serum or slurry as its intake to the main part of the device, 3 nebulizers are shown in the figure. A system comprising two devices may be a portable system such that the system is contained in a transport container which may be transported by rail, water and/or road.
图28是本发明的实施方案的示意图,类似于图26和27中所示的那种,其中该系统包括两个装置,一个水平取向,离地面近,和第二个装置,垂直取向。在图28中所示的实施方案中,在水平取向的装置中的溶液(例如吸收溶液,接触混合物)在该装置中被再循环,使得溶液最初在装置中离气体入口最远的点处进入装置。使用更接近于气体入口区域的泵,然后使溶液再循环。这种再循环有效地在装置中产生了整个流体流动和气体流动之间的逆向流动,然而装置的结构可以使气体流动旋转,使得相对于从喷雾器下降的溶液,气体在局部交替地顺流和逆流流动。如图26和27中,垂直取向的装置中的溶液可以再循环至水平取向的装置,仅仅在垂直取向的装置中再循环,在垂直取向的装置中部分地和在水平取向的装置中部分地再循环,或不进行再循环。垂直取向的装置可以具有除雾段,刚好在气体出口前,其中接受清液(即没有固体颗粒物)作为其吸入物。垂直取向的装置可以接受清液或者浆液作为其对于装置的主要部分的吸入物,在图中显示了3个喷雾器。包括两个装置的系统可以是便携式系统,使得该系统被包含在运输容器中,其可以通过铁路、水路和/或公路来运输。Figure 28 is a schematic illustration of an embodiment of the invention, similar to that shown in Figures 26 and 27, in which the system includes two devices, one horizontally oriented, near the ground, and a second device, vertically oriented. In the embodiment shown in Figure 28, the solution (e.g. absorbing solution, contacting mixture) in the device in a horizontal orientation is recirculated in the device such that the solution initially enters the device at the point furthest from the gas inlet. device. Use a pump closer to the gas inlet area and then recirculate the solution. This recirculation effectively creates a counterflow between the overall fluid flow and the gas flow in the device, however the structure of the device allows the gas flow to be rotated such that the gas is locally alternately co-current and countercurrent flow. As in Figures 26 and 27, the solution in the vertically oriented device can be recycled to the horizontally oriented device, recycled only in the vertically oriented device, partially in the vertically oriented device and partially in the horizontally oriented device Recirculation, or no recirculation. A vertically oriented device may have a mist removal section, just before the gas outlet, which accepts clear liquid (ie, free of solid particulate matter) as its intake. The vertically oriented device can accept either serum or slurry as its intake to the main part of the device, 3 nebulizers are shown in the figure. A system comprising two devices may be a portable system such that the system is contained in a transport container which may be transported by rail, water and/or road.
图29是本发明的实施方案的示意图,其中不同类型的装置串联使用。该装置是使用喷雾器的阵列或组合喷雾器和棚排的装置。该装置还具有相对于气体流动顺流和逆流流动的流体(即吸收溶液或接触混合物)。各自装置中的流体(即吸收溶液或接触混合物)可以是相同的或不同的,并且其也许在各自装置中再循环或者从一个装置至另一个装置,从而引起期望的气体引入(即吸收)和有时候沉淀。Figure 29 is a schematic illustration of an embodiment of the invention wherein different types of devices are used in series. The device is a device using an array of sprayers or a combination of sprayers and rows. The device also has a fluid (ie, absorbing solution or contacting mixture) flowing co-currently and counter-currently with respect to the gas flow. The fluid (i.e. absorbing solution or contacting mixture) in the respective units may be the same or different, and it may be recirculated within the respective units or from one unit to the other, resulting in the desired gas introduction (i.e. absorption) and Sometimes precipitation.
图30和31是显示了本发明系统的本发明的实施方案的图例,其中装置排列成排并且气体并联和串联地流入该装置。图30显示了一种配置结构,其中气体流入多于一个装置,并且在这些第一个装置中,气体和液体(例如吸收溶液,接触混合物)流动是顺流的,然后气体流入多于一个随后的装置,其中流动是逆流的。图31显示了一种配置结构,其中气体流入多于一个装置,其中在多个装置的第一个中,气体和液体流动是逆流的,然后在随后的多个装置中,流动是顺流的。在各自装置中,溶液或接触混合物可以在装置中再循环或者从一个装置到另一个装置,从而获得气体的组分(例如CO2,SOx)或沉淀物的期望的除去。Figures 30 and 31 are diagrams showing an embodiment of the invention of the system of the invention wherein the devices are arranged in rows and gases flow into the devices in parallel and in series. Figure 30 shows an arrangement in which the gas flows into more than one device, and in these first devices the gas and liquid (e.g. absorption solution, contact mixture) flow is co-current and the gas then flows into more than one subsequent device in which the flow is countercurrent. Figure 31 shows an arrangement where gas flows into more than one device, where the gas and liquid flow is countercurrent in the first of the multiple devices, then in subsequent multiple devices the flow is cocurrent . In the respective plant, the solution or the contact mixture can be recycled within the plant or from one plant to another in order to obtain the desired removal of components of the gas (eg CO 2 , SOx) or precipitates.
沉淀反应器120,进一步包括用于母液的输出运输工具。在一些实施方案中,输出运输工具可以被配置以将母液运输到尾矿池进行处理或者在天然存在的水体例如海洋、海、湖泊或河流中。在其它实施方案中,系统可以被配置以允许母液用作工业设备的冷却剂,通过在沉淀系统和工业设备之间运转的作业线。在某些实施方案中,沉淀装置可以与脱盐装置共处,使得来自沉淀装置的输出水被用作脱盐装置的注入水。系统可以包括运输工具(即,管道),其中输出水(例如,母液)可以直接泵送到脱盐装置中。The
图32显示了用于本发明的实施方案的管道系统和仪表简图。简图显示两个可能的来自发电厂的烟道气源。显示了烟道气,其进入接触室的底部(即项目标记的吸收器)。接触室具有在底部的出口导管以及至再循环系统的连接机构,包括泵和开关管。在接触室的中心是喷雾器,在室的顶端是除雾段(即除雾器),然后是气体出口。显示了用于除雾器段的喷淋水源,其与室中心段的水源分开。室中心的溶液源还具有到至少一个氢氧化钠源(例如电化学法,碱性盐水)的连接机构。浆液磨机也示于图32中。这些磨机可以是用于粉碎浆液的固体组分的位置,其中固体组分可以是沉淀材料,矿物或工业废物(例如飞灰,水泥窑灰尘)。在一些实施方案中,图32中所示的接触室或吸收器可以是便携式的,使得其适合于标准运输容器并且可以通过火车、驳船和/或卡车运输到任何设备,在期望的情况下。Figure 32 shows a schematic diagram of the plumbing and instrumentation used in an embodiment of the invention. Simplified diagram showing two possible sources of flue gas from a power plant. Flue gas is shown, which enters the bottom of the contact chamber (ie the item labeled absorber). The contact chamber has an outlet conduit at the bottom and connections to a recirculation system, including pumps and switch tubes. In the center of the contact chamber is the nebulizer, at the top of the chamber is the demister section (ie demister) and then the gas outlet. Shown is the spray water supply for the demister section, which is separate from the water supply for the chamber center section. The solution source in the center of the chamber also has a connection mechanism to at least one source of sodium hydroxide (eg electrochemical, alkaline brine). A slurry mill is also shown in Figure 32. These mills may be the location for comminuting the solid components of the slurry, where the solid components may be precipitation material, minerals or industrial waste (eg fly ash, cement kiln dust). In some embodiments, the contact chamber or absorber shown in Figure 32 can be portable such that it fits in a standard shipping container and can be transported by train, barge and/or truck to any facility, if desired.
图7中举例说明的系统进一步包括液固分离装置140,其用于分离沉淀碳酸盐矿物组合物和沉淀系统流出物。液-固分离装置可以通过以下方式实现沉淀产物与沉淀系统流出物的分离:排水(例如,沉淀产物的重力沉淀随后排水),倾析,过滤(例如,重力过滤,真空过滤,使用增压空气的过滤),离心,压榨,或其任何组合。在一些实施方案中,液-固分离装置包括折流板,沉淀台流出物相抵于其流动而进行沉淀产物和上层清液分离。在这样的实施方案中,液-固分离装置可以进一步包括用于收集沉淀产物的收集器。可用于一些实施方案中的液-固分离器的来源是Epuramat′s Extrem-Separator(“ExSep”)液-固分离器或其变体。在一些实施方案中,液-固分离装置包括螺旋通道,沉淀台流出物流入其中而进行沉淀产物和上层清液分离。在这样的实施方案中,液-固分离装置可以进一步包括用于收集沉淀产物的螺旋通道出口的阵列。可用于一些实施方案中的液-固分离器的来源是Xerox PARC′s螺旋浓缩器或其变体。至少一种液-固分离装置可操作地连接到沉淀台使得沉淀台流出物可以由沉淀台流动到液-固分离装置(例如,液-固分离装置包括折流板或螺旋通道)。如上所详述的,许多不同的液-固装置可以被结合在一起使用,以任何排列(例如,平行,串联或其组合),并且沉淀台流出物可以直接流动到液-固分离装置,或者流出物可以进行预处理。The system illustrated in Figure 7 further includes a liquid-
系统还包括洗涤台,150,其中来自分离台的本体脱水的沉淀物140被洗涤,例如,而从沉淀物中除去盐及其他溶质,然后在干燥台干燥。The system also includes a washing station, 150, wherein the bulk dewatered precipitate 140 from the separation station is washed, for example, to remove salts and other solutes from the precipitate, and then dried in a drying station.
系统进一步包括干燥台160,用于干燥由碳酸盐矿物沉淀台产生的沉淀碳酸盐矿物组合物。取决于系统的特定的干燥流程,干燥台可以包括过滤元件,冷冻干燥结构,喷雾干燥结构,等,如上更全面地描述。系统可以包括输送器,例如,管道,来自工业设备,其连接到干燥器,以便气态废物物流(即,工业设备烟道气)可以直接在干燥台中与湿沉淀物接触。The system further includes a drying
干燥的沉淀物可以经历进一步加工,例如,磨碎,研磨,在精制台180中,以便获得期望的物理性能。一种或多种组分可以添加到沉淀物,其中沉淀物用作建筑材料。The dried precipitate may undergo further processing, eg, grinding, milling, in a refining station 180 in order to obtain desired physical properties. One or more components can be added to the sediment, where the sediment is used as a building material.
系统进一步包括出口输送器,例如,运输带,浆液泵,其允许从以下一个或多个中除去沉淀物:反应器,干燥台,洗涤台或者精制台。可以以许多不同的方式处理沉淀反应的产物。沉淀物可被运输至长期储存位置,在空的运输交通工具中,例如,驳船,车厢轮渡(train cars),卡车等,其可以包括地上和地下存储设备。在其它实施方案中,沉淀物可在水下位置处理。可以使用任何适宜的用于将组合物运输到处理位置的流程。在某些实施方案中,可以使用管道或者类似的浆液输送结构,其中这些途径可以包括有源泵,重力介导的流动,等等。The system further comprises an outlet conveyor, for example, a conveyor belt, a slurry pump, which allows the sediment to be removed from one or more of: a reactor, a drying station, a washing station or a refining station. The product of the precipitation reaction can be handled in many different ways. Sediment can be transported to long-term storage locations in empty transport vehicles such as barges, train cars, trucks, etc., which can include above- and below-ground storage facilities. In other embodiments, the sediment may be disposed of in an underwater location. Any suitable process for transporting the composition to a processing location may be used. In certain embodiments, pipes or similar slurry delivery structures may be used, where such pathways may include active pumps, gravity mediated flow, and the like.
在某些实施方案中,系统将进一步包括用于由沉淀物制备建筑材料如水泥的工作台。所述工作台可以被配置以从沉淀物生产各种水泥、集料或粘结材料,例如,如共同未决的美国专利申请(公开号)2009/0020044的(2008年11月25日公开,其全盘引入本文作为参考)所述。In certain embodiments, the system will further include a station for preparing construction materials, such as cement, from the sediment. The workbench can be configured to produce various cements, aggregates, or cementitious materials from sediment, for example, as described in co-pending U.S. Patent Application (Publication No.) 2009/0020044 (published November 25, 2008, which is incorporated herein by reference in its entirety).
如上所指出,所述系统可以存在于陆地或海上。例如,系统可以是陆地基系统,其处于沿海地区,例如接近于海水源,或者甚至内陆位置,其中水由盐水源,例如,海洋通过管道输送到系统中。或者,所述系统可以是水基质系统,即,在水上或中存在的系统。这样的系统可以存在在船、海洋基平台等上,根据期望。在某些实施方案中,系统可以在任何合宜的位置与工业设备共处。沉淀装置可以是陆地基装置,其与陆地基工业设备共处,例如,在沿海地区中,如接近于水源(例如,海水)。还考虑的是内陆位置,其中水由水源(例如,工业设备,遥远湖泊,遥远海洋)直接通过管道输送到系统中。或者,沉淀装置可以存在于水上,例如,在驳船,船,海洋基平台等上,根据期望,例如,其中在工业设备附近的地产缺乏。在某些实施方案中,沉淀装置可以是可移动装置,使得其容易地与工业设备共处。As noted above, the system may exist on land or at sea. For example, the system may be a land-based system that is located in a coastal location, such as close to a seawater source, or even in an inland location where water is piped into the system from a saltwater source, such as the ocean. Alternatively, the system may be a water-based system, ie a system that exists on or in water. Such a system may exist on a ship, ocean-based platform, etc., as desired. In certain embodiments, the system can be co-located with industrial equipment at any convenient location. The settling unit may be a land-based unit that is co-located with land-based industrial equipment, eg, in coastal areas, such as close to a water source (eg, seawater). Also considered are inland locations where water is piped directly into the system from a source (eg, industrial facility, remote lake, remote ocean). Alternatively, the settling means may be present on water, eg, on a barge, boat, ocean-based platform, etc., as desired, eg, where real estate near industrial equipment is lacking. In certain embodiments, the settling unit may be a mobile unit such that it is easily co-located with industrial equipment.
与工业设备如发电厂共处的本发明的系统可以被配置以允许同步工业设备和沉淀装置的活动。在某些情况中,一种装置的活动未必相配于另一个的活动。例如,沉淀装置可能需要降低或停止其接受气态废物物流,但是工业设备可能需要保持操作。反之,情况可能出现,其中工业设备降低或停止操作而沉淀装置不是。为解决其中沉淀装置或工业设备可能需要降低或停止它的活动的情况,可以使用提供了共处装置之一连续操作而另一个降低或停止操作的设计特点,如上所详细描述的。例如,在某些实施方案中,本发明的系统可以在沿工业设备和沉淀装置之间的连接作业线的各点处包括鼓风机,风扇和/或压缩机,以便控制在连接工业设备至沉淀装置的管道中背压的出现。在某些实施方案中,气体存储设备可以存在于工业设备和沉淀装置之间。在期望的情况下,沉淀装置可以包括排放监控器以便评估由沉淀装置所产生的任何气态排放物,如由空气质量机构(Air Quality Agencies)要求的。The system of the present invention co-located with industrial equipment such as a power plant can be configured to allow synchronization of the activities of the industrial equipment and the settling plant. In some cases, the activity of one device may not necessarily match the activity of another. For example, a precipitation unit may need to be reduced or stopped from receiving a gaseous waste stream, but an industrial plant may need to remain in operation. Conversely, situations may arise where the industrial plant is reduced or out of operation while the settling plant is not. To address situations where a settling plant or industrial plant may need to reduce or cease its activity, design features that provide for continuous operation of one of the co-located devices while the other is reduced or ceased operation may be used, as detailed above. For example, in certain embodiments, the system of the present invention may include blowers, fans, and/or compressors at various points along the connection line between the industrial plant and the settling plant, in order to control the The occurrence of back pressure in the pipeline. In certain embodiments, a gas storage facility may exist between the industrial facility and the precipitation unit. Where desired, the settling unit may include emission monitors to assess any gaseous emissions produced by the settling unit, as required by Air Quality Agencies.
本发明的方面包括,在其中沉淀存储稳定的含CO2的产品的方法的一个或多个级处,使用含CO2的工业设备气态废物物流,例如,工业设备烟道气。因而,含CO2的工业设备气态废物物流用于沉淀方法中。在本发明的实施方案中,气态废物物流用于沉淀方法的一个或多个步骤,例如,在沉淀步骤中,例如,其中其用于加料水以CO2,或者在沉淀物干燥步骤期间,例如,其中沉淀的碳酸盐化合物被干燥等等。Aspects of the invention include the use of a CO2 -containing industrial facility gaseous waste stream, eg, industrial facility flue gas, at one or more stages of the process in which a stable CO2 -containing product is stored by precipitation. Thus, industrial plant gaseous waste streams containing CO2 are used in the precipitation process. In an embodiment of the invention, the gaseous waste stream is used in one or more steps of the precipitation process , e.g. , where the precipitated carbonate compounds are dried and so on.
在期望的情况下,来自工业设备的烟道气可以通过沉淀装置再循环直到全部吸附的剩余CO2接近100%,或者实现收益递减点,使得剩余的烟道气可以使用备选地流程来加工和/或释放到大气中。Where desired, the flue gas from the industrial plant can be recirculated through the precipitation unit until the total adsorbed remaining CO2
在一些实施方案中,本发明的装置和系统可以可操作地连接到发电厂,后者产生电力和工业废气(即烟道气)。在这样的实施方案中,本发明的装置和系统可以被考虑为从工业废气中除去某些组分的排放控制系统。在一些实施方案中,工业废气包括二氧化碳,SOx,NOx,重金属,非CO2酸性气体和飞灰。在一些实施方案中,本发明的装置和系统可以作为排放控制系统,其被配置以从工业废气除去二氧化碳。在一些实施方案中,本发明的装置和系统可以作为排放控制系统,其被配置以从工业废气中除去至少二氧化碳并且另外从工业废气中除去SOx。在一些实施方案中,通过包括本发明的装置和/或系统的排放控制系统除去二氧化碳和任选地SOx,同时利用最小量的由发电厂所产生的电力,例如小于30%的由发电厂所产生的电力。在一些实施方案中,本发明的排放控制系统可以实现利用小于30%的由发电厂产生的电力,包括使用或接受来自被配置以产生苛性碱溶液(caustic solution)的电化学系统的碱性溶液,特别地本文中进一步描述的低电压电化学系统类型的。在一些实施方案中,本发明的排放控制系统连接到发电厂并且被配置以吸收至少50%的来自废气的二氧化碳并且使用小于30%的发电厂所产生的能量。在一些实施方案中,本发明的排放控制系统连接到发电厂并且被配置以吸收至少90%的来自废气的硫氧化物(SOx)并且使用小于30%的发电厂所产生的能量。在一些实施方案中,本发明的排放控制系统连接到发电厂并且被配置以吸收来自废气的至少50%的二氧化碳和至少80%的硫氧化物(SOx)并且使用小于30%的发电厂所产生的能量。在一些实施方案中,本发明的装置和系统可以与现有的排放控制系统在燃烧化石燃料的工厂处在适当位置一起使用。在一些实施方案中,现有的排放控制系统可以包括或利用:用于收集颗粒物的静电沉降器,SOx控制技术,NOx控制技术,用于收集颗粒物的物理过滤技术,汞控制技术,连同其它控制措施。In some embodiments, the devices and systems of the present invention can be operatively connected to a power plant that produces electricity and industrial waste gases (ie, flue gases). In such embodiments, the devices and systems of the present invention may be considered emission control systems for the removal of certain components from industrial waste gases. In some embodiments, industrial waste gases include carbon dioxide, SOx, NOx, heavy metals, non- CO2 acid gases, and fly ash. In some embodiments, the devices and systems of the present invention may function as emission control systems configured to remove carbon dioxide from industrial waste gases. In some embodiments, the devices and systems of the present invention may serve as emission control systems configured to remove at least carbon dioxide and additionally remove SOx from industrial exhaust gases. In some embodiments, carbon dioxide and optionally SOx are removed by an emission control system comprising the devices and/or systems of the present invention while utilizing a minimal amount of electricity generated by the power plant, for example less than 30% of the electricity generated by the power plant generated electricity. In some embodiments, the emission control system of the present invention can be implemented to utilize less than 30% of the electricity generated by a power plant, including using or receiving alkaline solution from an electrochemical system configured to produce a caustic solution , in particular of the type of low voltage electrochemical system described further herein. In some embodiments, the emission control system of the present invention is connected to a power plant and is configured to absorb at least 50% of the carbon dioxide from the exhaust and use less than 30% of the energy produced by the power plant. In some embodiments, the emission control system of the present invention is coupled to a power plant and is configured to absorb at least 90% of sulfur oxides (SOx) from the exhaust and use less than 30% of the energy produced by the power plant. In some embodiments, the emission control system of the present invention is connected to a power plant and is configured to absorb at least 50% of the carbon dioxide and at least 80% of the sulfur oxides (SOx) from the exhaust and use less than 30% of that produced by the power plant energy of. In some embodiments, the devices and systems of the present invention may be used with existing emission control systems in place at fossil fuel burning plants. In some embodiments, existing emission control systems may include or utilize: electrostatic precipitators for particulate matter collection, SOx control technology, NOx control technology, physical filtration technology for particulate matter collection, mercury control technology, among other controls measure.
如上所述,本发明的沉淀系统可以与工业设备共处。这种系统的实例在图7中举例说明。在图7中,来自发电厂200的烟道气出口170用于沉淀反应器120作为CO2源130和干燥器160以及热源。在期望的情况下,使用背压控制来至少降低,如果没有消除的话,背压的出现,其可能起因于将一部分的,如果不是全部的话,工业设备气态废物物流引导到沉淀装置100。控制背压出现的任何适宜方式可以使用。在某些实施方案中,在沿工业设备和沉淀装置之间的连接作业线的某些点提供了鼓风机,风扇和/或压缩机。在某些实施方案中,安装鼓风机以便将烟道气吸入管道,其将烟道气送入沉淀装置。用于这些实施方案中的鼓风机可以是电力或机械驱动的鼓风机。在这些实施方案中,如果根本存在的话,背压被减小到5英寸或更小,如1英寸或更小的水平。在某些实施方案中,气体存储设备可以存在于工业设备和沉淀装置之间。当存在时,气体存储设备可以用作波动、停工和平稳系统,以便存在着均匀流动的烟道气至沉淀装置。As mentioned above, the precipitation system of the present invention can be co-located with industrial equipment. An example of such a system is illustrated in FIG. 7 . In Figure 7, the flue gas outlet 170 from the
本发明的方面包括同步工业设备和沉淀装置的活动。在某些情况中,一种装置的活动未必相配于另一个的活动。例如,沉淀装置可能需要降低或停止其接受气态废物物流,但是工业设备可能需要保持操作。反之,情况可能出现,其中工业设备降低或停止操作,然而沉淀装置不是。为解决这种情况,装置可以被配置以提供共处装置之一连续操作而另一个降低或停止操作,这是可以使用的。例如,为解决其中沉淀装置不得不降低或消除其从工业设备接受的气态废物物流的量的情况,可以配置系统,使得将废物物流输送至沉淀装置的鼓风机和管道以受控顺序关闭而最小化压力变化并且工业设备烟道用作排放气态废物物流的辅助烟囱。同样地,如果工业设备降低或消除其气态废物物流的生产,例如,工业设备全部或部分地派发掉,或者在某些预先同意的水平存在着工业设备输出的削减,那么系统可以被配置以允许沉淀装置继续操作,例如,通过提供备选地CO2源,通过在干燥器中提供备选地加热流程,等等。Aspects of the invention include synchronizing the activities of industrial equipment and settling plants. In some cases, the activity of one device may not necessarily match the activity of another. For example, a precipitation unit may need to be reduced or stopped from receiving a gaseous waste stream, but an industrial plant may need to remain in operation. Conversely, situations may arise where the industrial plant is reduced or out of operation, whereas the settling plant is not. To address this situation, it may be useful if the devices can be configured to provide one of the co-located devices with continuous operation while the other reduces or ceases operation. For example, to address situations in which a settling plant has to reduce or eliminate the amount of gaseous waste stream it receives from industrial equipment, the system can be configured so that blowers and pipes carrying the waste stream to the settling plant are shut down in a controlled sequence to minimize The pressure varies and the industrial plant flue acts as an auxiliary stack for discharging the gaseous waste stream. Likewise, if an industrial facility reduces or eliminates the production of its gaseous waste stream, for example, if the industrial facility is dispatched in whole or in part, or if there is a curtailment of the industrial facility's output at some pre-agreed level, then the system can be configured to allow The precipitation unit continues to operate, for example, by providing an alternative source of CO 2 , by providing an alternative heating process in the dryer, etc.
在期望的情况下,沉淀装置可以包括排放监控器来评估由沉淀装置产生的任何气态排放物并且向管理机构进行必要的报告,都是电子方式的(一般每隔15分钟),按日,按周,按月,按季度和按年。在某些实施方案中,在沉淀装置的气体处置是充分关闭的,使得来自沉淀装置的排放空气,其包含基本上全部来自工业设备的不用的烟道气,被导向烟囱,以便根据国家、省、州市或其它政治管辖区域的法定和规章的要求可以安装所需要的连续排放监控系统。Where desired, the settling plant may include emission monitors to assess any gaseous emissions produced by the settling plant and make necessary reports to regulatory agencies, all electronically (typically every 15 minutes), on a daily, monthly basis Weekly, monthly, quarterly and yearly. In certain embodiments, the gas handling at the settling plant is sufficiently closed such that the discharge air from the settling plant, which contains substantially all unused flue gas from the industrial plant, is directed to the chimney for Statutory and regulatory requirements of a state, state, or other political jurisdiction may install the required continuous emission monitoring system.
在某些实施方案中,由工业设备产生并且输送到沉淀装置的气态废物物流已经按空气质量机构(Air Quality Agencies)的要求进行处理,使得输送至沉淀装置的烟道气已经符合空气质量要求。在这些实施方案中,倘若沉淀装置停机,沉淀装置可能或未必在适当位置具有备选的处理系统。然而,如果被输送至的烟道气已经仅仅部分处理或者根本没有处理的话,沉淀装置可包括空气污染控制装置以便满足规章要求,或者寻找制定规章的当局以便短时间排放部分处理的烟道气。在其它实施方案中,烟道气被输送至沉淀装置用于全部加工。在这样的实施方案中,该系统可能包括防护设施,用于其中沉淀装置不能接受废物物流的情况,例如,通过确保在工业设备中安装的污染控制开启并且控制排放,如由空气质量机构(Air Quality Agencies)需要的。In certain embodiments, the gaseous waste stream produced by the industrial facility and delivered to the precipitation unit has been treated in accordance with Air Quality Agencies requirements such that the flue gas delivered to the precipitation unit already complies with air quality requirements. In these embodiments, the precipitation unit may or may not have an alternate treatment system in place in the event of a settling unit shutdown. However, if the flue gas being conveyed has been only partially treated or not treated at all, the precipitation plant may include air pollution control devices in order to meet regulatory requirements, or seek regulatory authority to discharge partially treated flue gas for a short period of time. In other embodiments, the flue gas is sent to a precipitation unit for all processing. In such an embodiment, the system may include containment for situations where the settling unit cannot accept the waste stream, for example, by ensuring that pollution controls installed in the industrial facility are on and control emissions, as provided by the Air Quality Agency (Air Quality Agencies).
与工业设备共处的沉淀装置可以存在于任何适宜位置,在陆地上或在水上。例如,沉淀装置可以是陆地基装置,其与陆地基工业设备共处,例如,在沿海地区中,如接近于海水源。还考虑的是内陆位置,其中水由水源(例如,工业设备,遥远湖泊,遥远海洋)直接通过管道输送到系统中。或者,沉淀装置可以存在于水上,例如,在驳船,船,海洋基平台等上,根据期望,例如,其中在工业设备附近的不动产缺乏。在某些实施方案中,沉淀装置可以是可移动装置,使得其容易地与工业设备共处。The settling unit co-located with the industrial plant may be located in any suitable location, on land or on water. For example, the settling unit may be a land-based unit that is co-located with land-based industrial equipment, for example, in coastal areas, such as close to seawater sources. Also considered are inland locations where water is piped directly into the system from a source (eg, industrial facility, remote lake, remote ocean). Alternatively, the settling device may be present on water, eg, on a barge, ship, ocean-based platform, etc., as desired, eg, where real estate near industrial equipment is lacking. In certain embodiments, the settling unit may be a mobile unit such that it is easily co-located with industrial equipment.
如上所指出,所考虑的是在某些实施方案中通过集成气化联合循环(IGCC)装置来产生废物物流。在这些类型的装置中,原始的燃料,例如,煤,生物质等首先经受气化过程以产生合成气,其然后变换,产生一定数量的CO2、CO和H2。气化流程的产物可以被输送到沉淀装置以便首先除去CO2,所得的CO2洗气产物被返回到发电厂用作燃料。在这样的实施方案中,来自发电厂的气化单元的作业线可以存在于发电厂和沉淀装置之间,第二返回作业线可以存在于沉淀装置和发电厂之间以便将吸气的合成气输送回到发电厂。As noted above, it is contemplated that in certain embodiments the waste stream will be generated by an integrated gasification combined cycle (IGCC) unit. In these types of installations, raw fuels such as coal, biomass, etc. are first subjected to a gasification process to produce syngas, which is then transformed, producing quantities of CO2 , CO and H2 . The product of the gasification process can be sent to a precipitation unit to remove CO 2 first, and the resulting CO 2 scrubbed product is returned to the power plant for use as fuel. In such an embodiment, a line from the gasification unit of the power plant may exist between the power plant and the settling unit, and a second return line may exist between the settling unit and the power plant to convert the aspirated syngas transported back to the power plant.
在某些实施方案中,共处的工业设备和沉淀装置(或集成装置)用另外的CO2减排方法来操作。例如,材料处理、交通工具和运土设备、机车可以被配置以使用生物燃料代替化石燃料。在这样的实施方案中,位置可以包括燃料罐以便存储生物燃料。In certain embodiments, the co-located industrial plant and precipitation plant (or integrated plant) are operated with additional CO2 abatement methods. For example, material handling, vehicles and earth-moving equipment, locomotives could be configured to use biofuels instead of fossil fuels. In such an embodiment, the location may include a fuel tank for storing biofuel.
除埋存CO2之外,本发明的实施方案还埋存了工业设备产生的气态废物物流的其它组分。例如,本发明的实施方案导致埋存可以存在于废物物流中的NOx,SOx,VOC,汞和颗粒物中的一个或多个的至少一部分,使得这些产品中的一个或多个被固定在固体沉淀物产品中。In addition to sequestering CO2 , embodiments of the present invention also sequester other components of gaseous waste streams generated by industrial facilities. For example, embodiments of the present invention result in the sequestration of at least a portion of one or more of NOx, SOx, VOC, mercury, and particulate matter that may be present in a waste stream such that one or more of these products are immobilized in solid deposits in the product.
在图7中,沉淀系统100与工业设备200共处。然而,沉淀系统100没有集成以工业设备200。在某些实施方案,因此还进一步关注的是一种集成的设备,其,除工业设备之外,包括发电,水处理(海水脱盐或富含矿物的淡水处理)和沉淀组件,如美国专利申请(公开号)2009/0001020中所述的,其公开日为2009年1月1日,其全盘引入本文作为参考。用于沉淀装置的水源可以衍生自水处理装置的废物物流。来自碳酸盐沉淀装置的所得的母液可以用作水处理装置的原料。所得的集成设备基本上使用燃料,矿物和未处理水作为输入物,并且输出能量,加工的工业产品,例如,水泥,清水,清洁空气,和碳埋存建筑材料。In FIG. 7 , a
图33提供了其中沉淀系统100集成以工业设备(在这种情况下烧煤电厂300)的实例。在发电厂300,煤310作为蒸汽锅炉315的燃料以产生蒸汽,其又运转涡轮(未示)以产生电力。蒸汽锅炉315还产生了炉底灰或锅炉渣325和烟道气320。烟道气320包含飞灰,CO2和硫酸盐。烟道气320和炉底灰325与来自水源330的水在反应器340中结合并且经受沉淀反应,如上所述。泵350促进来自反应器340的沉淀产品运输至喷雾干燥器360,其使用烟道气320以便喷雾干燥沉淀的产物,用于随后的处理,例如放置在填埋场中或者在建筑产品中使用。处理的烟道气370离开喷雾干燥器360并且然后在烟囱380中排出到大气。处理的烟道气370是其中与烟道气320相比飞灰,硫和CO2含量已经基本上降低,即使没有完全除去的话,的气体。作为图33中所示的系统的实例,CO2源可以是来自煤或其它燃料燃烧的烟道气,其接触一定体积的盐水,而很少或者没有烟道气的预处理。在这些实施方案中,使用燃料如高硫煤,次烟煤,褐煤等,其常常是廉价的并且被认为是低质量的,是实用的,这是由于所述方法除去SOx和其它污染物以及除去CO2的能力。这些燃料也可提供较高水平的辅助反应物如氧化铝和二氧化硅,在由烟道气携带的飞灰中,产生具有有益性能的改质的碳酸盐矿物沉淀物。Figure 33 provides an example where the
当与发电厂共处时,本发明的方法提供了大量的来自由发电厂产生的气态废物物流的CO2的埋存,而具有有限的能量需要。在有些情况下,所述方法提供了除去5%或更多,10%或更多,25%或更多,50%或更多,75%或更多的来自气态废物物流的CO2,而能量需要为50%或更小,如30%或更小,包括25%或更小。能量需要是操作二氧化碳埋存方法所需要的由发电厂所产生的能量的量。在有些情况下,上述水平的CO2除去是借助于20%或更小,15%或更小,10%或更小的能量需要获得的。When co-located with a power plant, the method of the invention provides for the sequestration of substantial amounts of CO2 from the gaseous waste stream produced by the power plant, with limited energy requirements. In some cases, the method provides removal of 5% or more, 10% or more, 25% or more, 50% or more, 75% or more of the CO2 from the gaseous waste stream, and The energy requirement is 50% or less, such as 30% or less, including 25% or less. The energy requirement is the amount of energy produced by the power plant required to operate the carbon dioxide storage process. In some cases, the above levels of CO2 removal are achieved with energy requirements of 20% or less, 15% or less, 10% or less.
可与本发明的沉淀装置共处的另一种类型的工业设备是水泥厂,如波特兰水泥生产装置。图34提供了示范性的波特兰水泥生产设备的示意图。在图34中,石灰石400以及页岩及其他添加剂410被研磨至合适的尺寸并且通过预锻烧炉500,其使用来自烟道气430的废热来预热混合物,利用来自窑炉510的废热来改善操作效率。预热的混合物进入窑炉510,其中其进一步通过燃烧煤炭420来加热。所得的渣块480被收集并且存储在料仓570中,其中其与添加剂571如石膏,石灰石等混合并且在球磨机580中磨碎至期望的尺寸。离开球磨机的产品是波特兰水泥490,其水泥料仓590中存储然后运输给顾客。Another type of industrial plant that can be co-located with the settling plant of the present invention is a cement plant, such as the Portland cement plant. Figure 34 provides a schematic diagram of an exemplary Portland cement production facility. In Figure 34,
来自窑炉510的烟道气430包含气体和颗粒物污染物。颗粒物污染物被称为窑炉粉尘440,并且通过静电沉降器或袋滤室520从烟道气中除去。如此除去的窑炉粉尘通常被送到填埋场600,可是偶而窑炉粉尘被循环到窑炉,或者作为辅助的粘结材料出售。然后烟道气通过风扇540吸入湿式洗涤器550,其中烟道气中的硫氧化物通过与生石灰浆液的反应被除去,产生亚硫酸钙(例如,石膏)浆液480,其通常在回收罐572中脱水并且在填埋场600中处理。烟道气430离开湿式洗涤器430并且通过烟囱560被释放到大气。如此释放的烟道气具有高浓度的CO2,这是通过煤炭的燃烧和通过氧化石灰石为波特兰水泥所要求的煅烧而释放的。
图35显示了根据本发明一个实施方案的示范性的共处的水泥厂和沉淀装置的示意图。该实例中的方法与图34中的相同,但是碳酸酯沉淀装置替换了图34的烟道气处理系统。一旦烟道气离开预锻烧炉500,其被风扇540吸入到反应器630,其中使用海水620和碱625,引发了沉淀反应。所得的浆液631通过泵640泵送到干燥台650,其中水651被排出并且干燥的粘结材料660被存储用于运输给顾客。与一部分的如果不是全部的被除去的污染物(包括汞,SOx,颗粒物,和CO2)一起,从烟囱670中排放烟道气430。Figure 35 shows a schematic diagram of an exemplary co-located cement plant and settling unit according to one embodiment of the present invention. The process in this example is the same as in Figure 34, but the flue gas treatment system of Figure 34 is replaced by a carbonate precipitation unit. Once the flue gas leaves the
图35显示了根据本发明一个实施方案的不需要石灰石采石场的示范性的水泥厂的示意图。在这种实施方案中,反应器630的产物可以采取相对纯的碳酸钙的形式,在其操作期间的部分时间内,以及建筑材料的其他形式,在其他部分时间内。在本实例中,而不是开采的石灰石,预锻烧炉500和窑炉510填装有混以相对纯的沉淀碳酸钙670的页岩及其他成分410的混合物。前述的以及结合作为参考的美国专利申请(公开号)2009/0020044,公开日2008年11月25日,详细描述了使用烟道气由海水沉淀霰石碳酸钙的流程。通过使用烟道气处理反应器的产物作为原料,水泥厂从海水中通过沉淀的产物汲取了其钙离子并且仅仅在第一短周期操作中需要开采的石灰石,直到足够的沉淀碳酸钙产生来用于加料窑炉。Figure 35 shows a schematic diagram of an exemplary cement plant that does not require a limestone quarry, according to one embodiment of the present invention. In such an embodiment, the product of
在本发明的一些实施方案中,吸收溶液接触二氧化碳的气体源以便在一个装置或系统中结合来自二氧化碳气体源的二氧化碳和也许一种或多种其它组分,使得不需要单独的排放控制系统或装置。在一些实施方案中,吸收溶液接触二氧化碳的气体源以便结合来自二氧化碳气体源的二氧化碳和也许一种或多种其它组分,并且所得的接触过的吸收溶液被处理而没有使溶液经受沉淀条件。In some embodiments of the invention, the absorption solution is contacted with a gas source of carbon dioxide to combine carbon dioxide and perhaps one or more other components from the source of carbon dioxide gas in one device or system such that a separate emission control system or device. In some embodiments, the absorption solution is contacted with a gas source of carbon dioxide to bind carbon dioxide and perhaps one or more other components from the carbon dioxide gas source, and the resulting contacted absorption solution is treated without subjecting the solution to precipitation conditions.
在本发明的实施方案中,在两级中进行碳酸盐沉淀。第一级有选择地沉淀碳酸钙,其然后可以用作水泥厂的原料,如图36中举例说明的。第二沉淀级可以产生许多不同的材料,包括水泥,集料,地上碳埋存材料等。In an embodiment of the invention, carbonate precipitation is carried out in two stages. The first stage selectively precipitates calcium carbonate, which can then be used as a raw material for a cement plant, as illustrated in FIG. 36 . The second precipitation stage can produce many different materials, including cement, aggregates, above-ground carbon storage materials, etc.
波特兰水泥是60-70质量%的CaO,其是通过加热CaCO3产生的,需要加热,对于所释放的每个分子的CaO,释放了一个分子的CO2。由于从燃料燃烧中释放的另外的CO2,来自沉淀装置的沉淀的CaCO3的输出将超过提供原料给水泥厂所需要的量。在这一情况下,沉淀装置的一部分的操作时间可致力于生产其它粘结材料660,如美国专利申请(公开号)2009/0020044中所述的那些,其公开日为2008年11月25日,其全盘引入本文作为参考。Portland cement is 60-70% by mass of CaO, which is produced by heating CaCO3 , requiring heating, for every molecule of CaO released, one molecule of CO2 is released. Due to the additional CO2 released from fuel combustion, the output of precipitated CaCO3 from the precipitation plant will exceed the amount required to provide raw material to the cement plant. In this case, a portion of the settling unit's operating time may be devoted to producing
如图36所示生产的波特兰水泥490是碳中性的,因为来自其制造过程的CO2被埋存到沉淀的碳酸盐矿物670和粘结材料660中。波特兰水泥490可以原样出售,或者与粘结材料660混和或相互研磨以产生混合水泥。Portland cement 490 produced as shown in FIG. 36 is carbon neutral in that CO 2 from its manufacturing process is sequestered into precipitated
所考虑的连续进料系统的实例描述在图37中。在图37中,系统1100包括水源(例如,来自海洋的管子从而提供海水)1101,其与反应器1110流体连通。还存在于反应器1110中的是Ca/Mg/OH离子源和催化剂1111,其已经以足量添加从而提高在反应器1110中存在的水中的Mg/Ca离子比至3或更多。反应器1110可以配置为填料床塔,并且配置由碳酸氢盐加料,如果期望的话。通过喷布器/鼓泡器1113,含CO2的气体,例如,烟道气1112与反应器1110中的水结合。Mg离子源和CO2与水在反应器1110中结合以产生CO2加料的酸性水,其在4.8-7.5的pH值下流出反应器1110。接下来,CO2加料的酸性水流过导管1120,其中其借助于以下实现循环:混合通过不同水平的碱度,例如,8.5和9.8,借助于使用各种CO2气体注射器1121,OH-调节器1123(如引入pH提高剂,电极,等等)和静止混合器1122,这些沿导管1120位于不同位置。通过导管1120的流速可以根据期望进行控制,例如,控制到1GPM-1,000,000GPM,如30GPM-100,000GPM和包括4,000GPM-60,000GPM。导管1120的长度可以改变,100ft至20,000ft,如1000ft至7000ft。在导管1120尾端,获得了浆液形式的产品1130,该浆液产品包括沉淀的CO2-埋存产品和母液。所得的浆液然后被转送到液固分离装置和澄清槽,如在1140处举例说明的。An example of a contemplated continuous feed system is depicted in FIG. 37 . In FIG. 37 , the
在某些实施方案中,可以使用两个或更多个反应器以便进行本文中所述的方法。使用两个反应器的实施方案的示意图示于图38,39和40中。在这种实施方案中,该方法可以包括第一反应器1210和第二反应器1220。在这些情况下,第一反应器1210用于使初始的水,例如新鲜的海水1230与镁离子源1240接触,并且用于加料初始的水以含CO2的气体,例如烟道气1250(其中这个步骤还称为碳酸氢盐加料)。烟道气1250可以与水在第一反应器1210中通过喷布器/鼓泡器1280接触。用搅拌器1260搅拌水从而促进镁离子源的溶解并且促进初始的水与含CO2的气体接触。有时候,在CO2加料的酸性水转移到第二反应器1220前,停止搅拌CO2加料的酸性水,使得不溶解的固体可以靠重力沉降。CO2加料的酸性水然后通过导管1270从第一反应器1210转移到第二反应器1220。In certain embodiments, two or more reactors may be used in order to carry out the methods described herein. Schematics of embodiments using two reactors are shown in FIGS. 38 , 39 and 40 . In such an embodiment, the process may include a first reactor 1210 and a second reactor 1220 . In these cases, a first reactor 1210 is used to contact initial water, such as fresh seawater 1230, with a source of magnesium ions 1240, and to feed the initial water to a CO2 -containing gas, such as flue gas 1250 (where This step is also known as bicarbonate addition). Flue gas 1250 may be contacted with water in first reactor 1210 via sparger/bubbler 1280 . The water is stirred with stirrer 1260 to facilitate dissolution of the source of magnesium ions and to facilitate initial contact of the water with the CO2- containing gas. Occasionally, agitation of the CO2 - fed sour water is stopped before the CO2- fed sour water is transferred to the second reactor 1220 so that undissolved solids can settle by gravity. The CO fed sour water is then transferred from the first reactor 1210 to the second reactor 1220 via
在转移CO2加料的酸性水至第二反应器1220后,可以进行碳酸盐沉淀的步骤。有时候,在第二反应器1220中使pH提高剂1290接触水而促进含碳酸盐的沉淀物的形成。可以用搅拌器1295搅拌第二反应器1220的内容物。在某些实施方案中,CO2加料和随后的碳酸盐沉淀的一个或多个另外的步骤可以在第二反应器中进行,如上所述。在这些情况下,另外的含CO2的气体,例如烟道气1255,通过喷布器/鼓泡器1285,与第二反应器520中的水接触。所得的浆液产品包括沉淀的CO2-埋存产品和母液,其然后转送到水/固体分离器或澄清槽,如上所述。After transferring the CO 2 -fed sour water to the second reactor 1220, a carbonate precipitation step may be performed. Occasionally, the formation of a carbonate-containing precipitate is promoted by contacting the pH-enhancing
组合物combination
本发明的组合物可以是溶液,固体或多相材料(例如,浆液),其包含碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐,任选地二价阳离子如Ca2+,Mg2+的,或其组合。通过本发明的方法生产的这样的组合物(例如,存储稳定的二氧化碳埋存产品如沉淀材料)中的碳的量可以变化。在一些实施方案中,组合物包括一定量的碳(如通过使用下文更详细描述的流程确定的,如同位素分析,例如,13c同位素分析):1%至15%(w/w),如5至15%(w/w),包括5至14%(w/w),5至13%(w/w),6至14%(w/w),6至12%(w/w)和7至12%(w/w),其中显著数量的碳可以是源于CO2来源的碳(如通过以下更详细描述的流程来确定)。在这样的实施方案中,10至100%,如50至100%,包括90至100%的存在于组合物(例如,存储稳定的二氧化碳埋存产品如沉淀材料)中的碳来自于CO2源(例如,包括二氧化碳的工业废气物流)。在有些情况下,存在于组合物中的可追溯至二氧化碳源的碳的量是50%或更多,60%或更多,70%或更多,80%或更多,90%或更多,95%或更多,99%或更多,包括100%。The compositions of the present invention may be solutions, solids or heterogeneous materials (e.g. slurries) comprising carbonates, bicarbonates or carbonates and bicarbonates, optionally of divalent cations such as Ca2+, Mg2+, or a combination thereof. The amount of carbon in such compositions (eg, storage-stable carbon dioxide sequestration products such as precipitation materials) produced by the methods of the invention can vary. In some embodiments, the composition includes an amount of carbon (as determined by using the procedure described in more detail below, such as isotopic analysis, e.g., 13c isotopic analysis): 1% to 15% (w/w), such as 5 to 15% (w/w), including 5 to 14% (w/w), 5 to 13% (w/w), 6 to 14% (w/w), 6 to 12% (w/w) and 7 to 12% (w/w), where a significant amount of carbon may be carbon originating from a CO2 source (as determined by the scheme described in more detail below). In such embodiments, 10 to 100%, such as 50 to 100%, including 90 to 100%, of the carbon present in the composition (e.g., a storage-stable carbon dioxide sequestration product such as a precipitation material) is derived from the CO source (eg, industrial waste streams including carbon dioxide). In some cases, the amount of carbon traceable to the source of carbon dioxide present in the composition is 50% or more, 60% or more, 70% or more, 80% or more, 90% or more , 95% or more, 99% or more, including 100%.
本发明的组合物(例如,包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的沉淀材料)可以存储50吨或更多的CO2,如100吨或更多的CO2,包括150吨或更多的CO2,例如200吨或更多的CO2,如250吨或更多的CO2,包括300吨或更多的CO2,如350吨或更多的CO2,包括400吨或更多的CO2,例如450吨或更多的CO2,如500吨或更多的CO2,包括550吨或更多的CO2,如600吨或更多的CO2,包括650吨或更多的CO2,例如700吨或更多的CO2,对于每1000吨的组合物。因此,在一些实施方案中,本发明的组合物(例如,包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的沉淀材料)包括5%或更多的CO2,如10%或更多的CO2,包括25%或更多的CO2,例如50%或更多的CO2,如75%或更多的CO2,包括90%或更多的CO2。这样的组合物,特别地本发明的沉淀材料可用于建筑环境。在一些实施方案中,可以使用组合物作为所制造的物品的组分,如建筑材料(例如,水泥组分、集料、混凝土或其组合)。组合物保持了存储稳定的CO2-埋存产品,因为使用制造的物品(如建筑材料)中的组合物没有导致埋存的CO2的再释放。在一些实施方案中,本发明的组合物(例如,包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的沉淀材料),当与波特兰水泥结合时,可以溶解并且与波特兰水泥的化合物结合而没有释放CO2。Compositions of the invention (e.g., precipitation materials comprising carbonates, bicarbonates, or carbonates and bicarbonates) can store 50 tons or more of CO 2 , such as 100 tons or more of CO 2 , including 150 tons or more of CO 2 , such as 200 tons or more of CO 2 , such as 250 tons or more of CO 2 , including 300 tons or more of CO 2 , such as 350 tons or more of CO 2 , including 400 tons or more of CO 2 , such as 450 tons or more of CO 2 , such as 500 tons or more of CO 2 , including 550 tons or more of CO 2 , such as 600 tons or more of CO 2 , including 650 tons or more of CO 2 , such as 700 tons or more of CO 2 , for every 1000 tons of the composition. Thus, in some embodiments, compositions of the invention (e.g., precipitation materials comprising carbonates, bicarbonates, or carbonates and bicarbonates) comprise 5% or more CO 2 , such as 10% or More CO2 , including 25% or more CO2 , such as 50% or more CO2 , such as 75% or more CO2 , including 90% or more CO2 . Such compositions, in particular the precipitation materials of the present invention, are useful in the built environment. In some embodiments, the compositions can be used as components of articles of manufacture, such as building materials (eg, cement components, aggregates, concrete, or combinations thereof). The composition maintains a storage-stable CO2 -storage product, since use of the composition in manufactured items, such as building materials, does not result in re-release of sequestered CO2 . In some embodiments, the compositions of the present invention (e.g., precipitation materials comprising carbonates, bicarbonates, or both carbonates and bicarbonates), when combined with Portland cement, can dissolve and interact with Portland cement. The compounds of the blue cement bind without releasing CO 2 .
用于将CO2转化为碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的条件可以导致一种或多种另外的组分和/或其辅助产物(即,由一种或多种另外的组分产生的产物),其中这样的另外的组分包括硫氧化物(SOx);氮氧化物(NOx);一氧化碳(CO);金属如锑(Sb),砷(As),钡(Ba),铍(Be),硼(B),镉(Cd),铬(Cr),钴(Co),铜(Cu),铅(Pb),锰(Mn),汞(Hg),钼(Mo),镍(Ni),镭(Ra),硒(Se),银(Ag),锶(Sr),铊(Tl),钒(V)和锌(Zn);颗粒物质;卤化物;有机物;有毒物质;放射性同位素等。在一些实施方案中,这样的一种或多种另外的组分和/或辅助产物可以是包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的溶液的一部分。在一些实施方案中,这样的一种或多种另外的组分和/或辅助产物可以是本发明的沉淀材料的一部分,通过沉淀一种或多种另外的组分和/或辅助产物以及碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐,通过在包括碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的沉淀材料中捕获一种或多种另外的组分和/或辅助产物,或者通过其某些组合。在一些实施方案中,这样的一种或多种另外的组分和/或辅助产物可以是包括上述溶液与沉淀材料的任何组合的浆液的一部分。The conditions used to convert CO to carbonate, bicarbonate, or carbonate and bicarbonate may result in one or more additional components and/or co-products thereof (i.e., produced by one or more products produced by additional components), where such additional components include sulfur oxides (SOx); nitrogen oxides (NOx); carbon monoxide (CO); metals such as antimony (Sb), arsenic (As), barium ( Ba), beryllium (Be), boron (B), cadmium (Cd), chromium (Cr), cobalt (Co), copper (Cu), lead (Pb), manganese (Mn), mercury (Hg), molybdenum ( Mo), nickel (Ni), radium (Ra), selenium (Se), silver (Ag), strontium (Sr), thallium (Tl), vanadium (V), and zinc (Zn); particulate matter; halides; organic matter ; Toxic substances; Radioactive isotopes, etc. In some embodiments, such one or more additional components and/or co-products may be part of a solution comprising carbonate, bicarbonate, or carbonate and bicarbonate. In some embodiments, such one or more additional components and/or by-products may be part of the precipitation material of the present invention by precipitating one or more additional components and/or by-products and carbon salt, bicarbonate or carbonate and bicarbonate by trapping one or more additional components and/or in the precipitation material comprising carbonate, bicarbonate or carbonate and bicarbonate Ancillary products, or by some combination thereof. In some embodiments, such one or more additional components and/or co-products may be part of a slurry comprising any combination of the solutions described above and precipitation material.
除碳酸盐和/或碳酸氢盐外,本发明的组合物可以包括硫酸盐,亚硫酸盐等。在一些实施方案中,组合物包括70-99.9%碳酸盐和/或碳酸氢盐以及0.05-30%硫酸盐和/或亚硫酸盐。例如,组合物可以包括至少50%、55%、60%、65%、70%、75%、80%、85%、90%、95%、或99.9%碳酸盐和/或碳酸氢盐。这样的组合物可以进一步包括至少0.05%、0.1%、0.5%、1.0%、5.0%、10%、15%、20%、25%或30%硫酸盐和/或亚硫酸盐。在一些实施方案中,本发明的组合物包括钙、镁或其组合的硫基化合物,任选地沉淀或捕获在由包括SOx(例如,SO2、SO3,等等)的废气物流产生的沉淀材料中。例如,镁和钙可以反应而分别形成MgSO4和CaSO4,以及其它的含镁和钙的化合物(例如,亚硫酸盐),这有效地从废气物流(例如,烟道气物流)中除去硫,而没有脱硫步骤如烟道气脱硫(″FGD″)。另外,可以形成包括CaSO4、MgSO4和相关化合物的组合物而没有另外释放CO2。在其中高含量的硫基化合物(例如,硫酸盐)存在的情况下,水溶液可以富集钙和/或镁,以便钙和/或镁可以用来在形成CaSO4、MgSO4和/或相关化合物之前、期间或之后形成碳酸盐化合物。在一些实施方案中,多种反应产物(例如,MgCO3、CaCO3、CaSO4、上述的混合物,等)在不同级收集,而在其它实施方案中单个反应产物(例如,包括碳酸盐、硫酸盐等的沉淀材料)被收集。In addition to carbonates and/or bicarbonates, the compositions of the present invention may include sulfates, sulfites, and the like. In some embodiments, the composition includes 70-99.9% carbonate and/or bicarbonate and 0.05-30% sulfate and/or sulfite. For example, the composition can include at least 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, or 99.9% carbonate and/or bicarbonate. Such compositions may further comprise at least 0.05%, 0.1%, 0.5%, 1.0%, 5.0%, 10%, 15%, 20%, 25% or 30% sulfate and/or sulfite. In some embodiments, the compositions of the present invention include sulfur-based compounds of calcium, magnesium, or combinations thereof, optionally precipitated or captured in exhaust gas streams that include SOx (e.g., SO2 , SO3 , etc.) in the sedimentation material. For example, magnesium and calcium can react to form MgSO4 and CaSO4 , respectively, and other magnesium- and calcium-containing compounds (e.g., sulfites), which effectively remove sulfur from waste gas streams (e.g., flue gas streams) , without a desulfurization step such as flue gas desulfurization ("FGD"). In addition, compositions including CaSO 4 , MgSO 4 and related compounds can be formed without additional release of CO 2 . In cases where high levels of sulfur-based compounds (e.g., sulfates) are present, the aqueous solution can be enriched in calcium and/or magnesium so that the calcium and/or magnesium can be used in the formation of CaSO 4 , MgSO 4 and/or related compounds Before, during or after formation of carbonate compounds. In some embodiments, multiple reaction products (e.g., MgCO 3 , CaCO 3 , CaSO 4 , mixtures of the above, etc.) are collected at different stages, while in other embodiments a single reaction product (e.g., including carbonates, Precipitated material such as sulfates) is collected.
本发明的组合物可以包括硝酸盐、亚硝酸盐和/或类似物。在一些实施方案中,本发明的组合物包括钙、镁或其组合的这样的氮基化合物,任选地沉淀或捕获在由包括NOx(例如,NO2,NO3,等等)的废气物流产生的沉淀材料中。例如,镁和钙可以反应而分别形成Mg(NO3)2和Ca(NO3)2,以及其它的含镁和钙的化合物(例如,硝酸盐),这有效地从废气物流(例如,烟道气物流)中除去氮,而没有选择性催化还原(″SCR″)步骤或非选择性催化还原(″NSCR″)步骤。另外,可以形成包括Ca(NO3)2、Mg(NO3)2和相关化合物的组合物而没有另外释放CO2。本发明的组合物可以进一步包括其它组分,如痕量金属(例如,汞)。使用汞作为痕量金属的非限制性实例,本发明的组合物可以包括元素汞(HgO)、含Hg2+的汞盐(例如,HgCl2、HgCO3,等等)、含Hg+的汞盐(例如,Hg2Cl2、Hg2CO3,等等)、含Hg2+的汞化合物(例如,HgO、有机汞化合物,等等)、含Hg+的汞化合物(例如,Hg2O、有机汞化合物,等等)、颗粒汞(Hg(p))等。在一些实施方案中,本发明的组合物包括这样的汞基化合物,任选地沉淀或捕获在由包括痕量金属如汞的废气物流产生的沉淀材料中。在一些实施方案中,组合物包括汞(或另一金属),其浓度为至少0.1、0.5、1、5、10、50、100、500、1,000、5,000、10,000ppb。汞可以反应而形成HgCO3或Hg2CO3以及其它含汞化合物(例如,氯化物、氧化物),这有效地从废气物流(例如,烟道气物流)除去汞而没有特异性的或非特异性的汞除去技术。另外,可以形成包括汞和和/或其它痕量金属的组合物而没有另外释放CO2。The compositions of the present invention may include nitrates, nitrites and/or the like. In some embodiments, the compositions of the present invention include such nitrogen-based compounds of calcium, magnesium, or combinations thereof, optionally precipitated or captured in exhaust gas streams that include NOx (e.g., NO 2 , NO 3 , etc.) in the resulting precipitated material. For example, magnesium and calcium can react to form Mg(NO 3 ) 2 and Ca(NO 3 ) 2 , respectively, and other magnesium- and calcium-containing compounds (eg, nitrates), which effectively Nitrogen is removed from the gas stream) without a selective catalytic reduction ("SCR") step or a non-selective catalytic reduction ("NSCR") step. Additionally, compositions including Ca( NO3 )2, Mg( NO3 )2, and related compounds can be formed without additional release of CO2 . The compositions of the present invention may further include other components, such as trace metals (eg, mercury). Using mercury as a non-limiting example of a trace metal, compositions of the present invention may include elemental mercury (HgO), Hg2+-containing mercury salts (e.g., HgCl2, HgCO3 , etc.), Hg+-containing mercury salts (e.g., Hg2Cl2, Hg2CO 3 , etc.), mercury compounds containing Hg2+ (for example, HgO, organic mercury compounds, etc.), mercury compounds containing Hg+ (for example, Hg2O, organic mercury compounds, etc.), particulate mercury (Hg( p)) etc. In some embodiments, the compositions of the present invention include mercury-based compounds that are optionally precipitated or trapped in precipitation material produced from exhaust gas streams that include trace amounts of metals such as mercury. In some embodiments, the composition includes mercury (or another metal) at a concentration of at least 0.1, 0.5, 1, 5, 10, 50, 100, 500, 1,000, 5,000, 10,000 ppb. Mercury can react to form HgCO3 or Hg2CO3 and other mercury-containing compounds (e.g., chlorides, oxides), which effectively removes mercury from waste gas streams (e.g., flue gas streams) without specific or nonspecific Mercury removal technology. Additionally, compositions including mercury and/or other trace metals can be formed without additional release of CO2 .
本发明的沉淀材料可以包括数个碳酸盐和/或数个碳酸盐矿物相,这些源于共沉淀,其中沉淀材料可以包括,例如,碳酸钙(例如,方解石)以及碳酸镁(例如,三水菱镁矿)。沉淀材料也可包括在单个矿物相中的单个碳酸盐,包括但不限于,碳酸钙(例如,方解石),碳酸镁(例如,三水菱镁矿),碳酸钙镁(例如,白云石)或铁碳铝硅酸盐(ferro-carbo-aluminosilicate)。由于不同的碳酸盐可以依次沉淀,沉淀材料可以是,取决于获得其的条件,在一个碳酸盐和/或一个矿物相中,相对富集的(例如,90%至95%)或显著富集的(例如,95%-99.9%),或者沉淀材料可以包括一定量的其它碳酸盐和/或其它矿物相(一个或多个),其中期望的矿物相是50-90%的沉淀材料。应将理解的是,在一些实施方案中,除碳酸盐外,沉淀材料可以包括一种或多种氢氧化物(例如,Ca(OH)2,Mg(OH)2)。还将理解的是存在于沉淀材料中的任何碳酸盐或氢氧化物可以是完全或部分无定形的。在一些实施方案中,碳酸盐和/或氢氧化物是完全无定形的。还将理解的是存在于沉淀材料中的任何碳酸盐或氢氧化物可以是完全或部分结晶的。在一些实施方案中,碳酸盐和/或氢氧化物是完全结晶的。The precipitation material of the present invention may include several carbonates and/or several carbonate mineral phases, which result from co-precipitation, wherein the precipitation material may include, for example, calcium carbonate (e.g., calcite) and magnesium carbonate (e.g., Trihydrate magnesite). Precipitation material may also include individual carbonates in a single mineral phase, including, but not limited to, calcium carbonate (e.g., calcite), magnesium carbonate (e.g., magnesite), calcium magnesium carbonate (e.g., dolomite) Or iron carbon aluminum silicate (ferro-carbo-aluminosilicate). Since different carbonates can precipitate sequentially, the precipitation material can be, depending on the conditions under which it was obtained, relatively enriched (e.g., 90% to 95%) or significantly enriched in a carbonate and/or a mineral phase. Enriched (e.g., 95%-99.9%), or the precipitated material may include an amount of other carbonate and/or other mineral phase(s), wherein the desired mineral phase is 50-90% precipitated Material. It will be appreciated that, in some embodiments, the precipitation material may include one or more hydroxides (eg, Ca(OH)2, Mg(OH)2) in addition to carbonate. It will also be understood that any carbonate or hydroxide present in the precipitation material may be fully or partially amorphous. In some embodiments, the carbonates and/or hydroxides are completely amorphous. It will also be understood that any carbonate or hydroxide present in the precipitation material may be fully or partially crystalline. In some embodiments, the carbonates and/or hydroxides are fully crystalline.
虽然由于原料的变化性,许多不同的含碳酸根的盐和化合物是可能的,但是包括碳酸镁、碳酸钙或其组合的沉淀材料是特别有用的。沉淀材料可以包括两个或更多个不同的碳酸盐化合物、三个或更多个不同的碳酸盐化合物、四个或更多个不同的碳酸盐化合物、五个或更多个不同的碳酸盐化合物等,包括非明显的、无定形碳酸盐化合物。本发明的沉淀材料可以包括具有分子式Xm(CO3)n的化合物,其中X是任何元素或元素的组合,其可以化学结合以碳酸根基团或其多倍体(multiple)并且m和n是化学计量的正整数。在一些实施方案中,X可以是碱土金属(元素周期表IIA族中的元素)或碱金属(元素周期表IA族中的元素)或者其某些组合。在一些实施方案中,沉淀材料包括白云石(CaMg(CO3)2),原白云石,碳钙镁石,(CaMg3(CO3)4),和/或水碳钙镁石(Ca2Mg11(CO3)13·H2O),其是包括钙和镁的碳酸盐矿物。在一些实施方案中,沉淀材料包括在一个或多个相中的碳酸钙,其选自方解石、霰石、球霰石或其组合。在一些实施方案中,沉淀材料包括水合式的碳酸钙(例如,Ca(CO3)·nH2O),其中在分子式中存在一个或多个结构水,其选自六水碳钙石(CaCO3·6H2O),无定形碳酸钙(CaCO3·nH2O),一水方解石(CaCO3·H2O),或其组合。在一些实施方案中,沉淀材料包括碳酸镁,其中碳酸镁不具有任何水合水。在一些实施方案中,沉淀材料包括碳酸镁,其中碳酸镁可以具有许多不同的水合水(例如,Mg(CO3)·nH2O),选自1、2、3、4或大于4个水合水。在一些实施方案中,沉淀材料包括1、2、3、4或大于4个不同的碳酸镁相,其中碳酸镁相在水合水数目上不同。例如,沉淀材料可以包括菱镁矿(MgCO3),水碳镁石(MgCO3·2H2O),三水碳镁石(MgCO3·3H2O),五水碳镁石(MgCO3·5H2O)和无定形碳酸镁。在一些实施方案中,沉淀材料包括碳酸镁,其包括氢氧化物和水合水,如水纤菱镁矿(MgCO3·Mg(OH)2·3H2O),水菱镁矿(Mg5(CO3)4(OH)2·3H2O)或其组合。因而,沉淀材料可以包括钙、镁或其组合的碳酸盐,处于全部或若干在本文中所列的水合的各种形式中。沉淀速率也可影响沉淀材料的性质,其中最快速的沉淀速率通过用期望相接晶溶液来获得。没有接晶,通过例如快速增加沉淀反应混合物的pH值,这导致更多的无定形组分,可以获得快速的沉淀。此外,pH越高,沉淀越快,该沉淀导致更多的无定形沉淀材料。While many different carbonate-containing salts and compounds are possible due to the variability of the feedstock, precipitation materials comprising magnesium carbonate, calcium carbonate, or combinations thereof are particularly useful. The precipitation material may comprise two or more different carbonate compounds, three or more different carbonate compounds, four or more different carbonate compounds, five or more different Carbonate compounds, etc., including non-distinct, amorphous carbonate compounds. The precipitation material of the present invention may comprise a compound having the molecular formula Xm(CO 3 )n, wherein X is any element or combination of elements which can be chemically bonded with a carbonate group or a multiple thereof and m and n are chemical A positive integer for the measurement. In some embodiments, X can be an alkaline earth metal (an element in Group IIA of the Periodic Table of the Elements) or an alkali metal (an element in Group IA of the Periodic Table of Elements), or some combination thereof. In some embodiments, the precipitation material includes dolomite (CaMg(CO 3 ) 2 ), orthodolomite, wortite, (CaMg3(CO 3 ) 4 ), and/or hydrocarbite (Ca2Mg11(CO 3 ) 13·H 2 O), which is a carbonate mineral including calcium and magnesium. In some embodiments, the precipitation material includes calcium carbonate in one or more phases selected from calcite, aragonite, vaterite, or combinations thereof. In some embodiments, the precipitation material comprises a hydrated form of calcium carbonate (e.g., Ca(CO 3 )·nH 2 O) in which one or more structural waters are present in the molecular formula selected from molarite (CaCO 3 ·6H 2 O), amorphous calcium carbonate (CaCO 3 ·nH 2 O), calcite monohydrate (CaCO 3 ·H 2 O), or combinations thereof. In some embodiments, the precipitation material includes magnesium carbonate, wherein the magnesium carbonate does not have any water of hydration. In some embodiments, the precipitation material includes magnesium carbonate, where the magnesium carbonate can have a number of different waters of hydration (e.g., Mg(CO 3 )·nH 2 O), selected from 1, 2, 3, 4, or more than 4 hydrated water. In some embodiments, the precipitation material includes 1, 2, 3, 4, or more than 4 different magnesium carbonate phases, wherein the magnesium carbonate phases differ in the number of waters of hydration. For example, the precipitation material may include magnesite (MgCO 3 ) , brucite (MgCO 3 . 5H 2 O) and amorphous magnesium carbonate. In some embodiments, the precipitation material includes magnesium carbonate, which includes hydroxide and water of hydration, such as hydromagnesite (MgCO 3 Mg(OH)2 3H 2 O), hydromagnesite (Mg5(CO 3 )4(OH)2· 3H2O ) or a combination thereof. Thus, the precipitation material may comprise carbonates of calcium, magnesium, or combinations thereof, in all or some of the hydrated forms listed herein. The precipitation rate can also affect the properties of the precipitated material, with the fastest precipitation rates being obtained with the desired contact crystallization solution. Without seeding, rapid precipitation can be obtained, for example, by rapidly increasing the pH of the precipitation reaction mixture, which results in a more amorphous component. Furthermore, the higher the pH, the faster the precipitation, which results in more amorphous precipitation material.
在有些情况下,沉淀材料中的碳酸钙化合物的按重量计的量可以超过沉淀材料中的碳酸镁化合物的按重量计的量。例如,沉淀材料中的碳酸钙化合物的按重量计的量可以超过沉淀材料中的碳酸镁化合物的按重量计的量达5%或更多,如10%或更多,15%或更多,20%或更多,25%或更多,30%或更多。在有些情况下,沉淀材料中的碳酸钙化合物与碳酸镁化合物的重量比为1.5-5至1,如2-4至1,包括2-3至1。在有些情况下,沉淀材料中的碳酸镁化合物的按重量计的量可以超过沉淀材料中的碳酸钙化合物的按重量计的量。例如,沉淀材料中的碳酸镁化合物的按重量计的量可以超过沉淀材料中的碳酸钙化合物的按重量计的量达5%或更多,如10%或更多,15%或更多,20%或更多,25%或更多,30%或更多。在有些情况下,沉淀材料中的碳酸镁化合物与碳酸钙化合物的重量比为1.5-5至1,如2-4至1,包括2-3至1。In some cases, the amount by weight of the calcium carbonate compound in the precipitation material may exceed the amount by weight of the magnesium carbonate compound in the precipitation material. For example, the amount by weight of the calcium carbonate compound in the precipitation material may exceed the amount by weight of the magnesium carbonate compound in the precipitation material by 5% or more, such as 10% or more, 15% or more, 20% or more, 25% or more, 30% or more. In some cases, the weight ratio of calcium carbonate compound to magnesium carbonate compound in the precipitation material is 1.5-5 to 1, such as 2-4 to 1, including 2-3 to 1. In some cases, the amount by weight of the magnesium carbonate compound in the precipitation material may exceed the amount by weight of the calcium carbonate compound in the precipitation material. For example, the amount by weight of the magnesium carbonate compound in the precipitation material may exceed the amount by weight of the calcium carbonate compound in the precipitation material by 5% or more, such as 10% or more, 15% or more, 20% or more, 25% or more, 30% or more. In some cases, the weight ratio of magnesium carbonate compound to calcium carbonate compound in the precipitation material is 1.5-5 to 1, such as 2-4 to 1, including 2-3 to 1.
通过本发明的方法生产的沉淀材料可以包括碳酸盐化合物,当与淡水组合时,其溶解初始的沉淀材料以产生包括碳酸盐化合物的淡水沉淀材料,相比于原始的沉淀材料的碳酸盐化合物,其在淡水中是更稳定的。(尽管当与淡水组合时,初始的沉淀材料的碳酸盐化合物可以溶解,产生了新的组合物。因此,在任何这样的反应中,CO2气体没有以显著的量释放,或者有时候,根本没有。)初始的沉淀材料的碳酸盐化合物可以是相比于它们在淡水中,在盐水中更稳定的化合物,使得碳酸盐化合物可以被看作在盐水中是亚稳定的。沉淀材料中的碳酸盐的量,如通过电量滴定测定的,可以是40%或更高、如70%或更高、包括80%或更高。The precipitation material produced by the method of the present invention may include carbonate compounds which, when combined with fresh water, dissolve the initial precipitation material to produce freshwater precipitation material comprising carbonate compounds, compared to the carbonic acid content of the original precipitation material. Salt compounds, which are more stable in fresh water. (Although when combined with fresh water, the carbonate compounds of the original precipitation material can be dissolved, resulting in a new composition. Therefore, in any such reaction, CO2 gas is not released in a significant amount, or sometimes, Not at all.) The carbonate compounds of the initial precipitation material may be more stable compounds in brine than they are in fresh water, so that the carbonate compounds may be considered metastable in brine. The amount of carbonate in the precipitation material, as determined by coulometric titration, may be 40% or higher, such as 70% or higher, including 80% or higher.
在沉淀期间调节主要离子比可影响沉淀材料的性质。主要离子比对多晶型物形成具有很大的影响。例如,随着在水中镁∶钙比增加,沉淀材料中,霰石变成碳酸钙的主要的多晶型物,相比于低-镁方解石来说。在低镁∶钙比下,低-镁方解石变成主要的多晶型物。在一些实施方案中,其中Ca2+和Mg2+都存在于水中,在水中Ca2+与Mg2+比(即,Ca2+∶Mg2+)为500∶1-1∶500,如100∶1-1∶100,如50∶1-1∶50,如20∶1-1∶20,如10∶1-1∶10。在一些实施方案中,其中Ca2+和Mg2+都存在于水中,在水中Ca2+与Mg2+比(即,Ca2+∶Mg2+)为5∶1-1∶1。在一些实施方案中,其中Ca2+和Mg2+都存在于水中,在水中Ca2+与Mg2+比(即,Ca2+∶Mg2+)是4∶1。在一些实施方案中,其中Ca2+和Mg2+都存在于水中,在水中Ca2+与Mg2+比(即,Ca2+∶Mg2+)为1∶1-1∶10。在一些实施方案中,其中Ca2+和Mg2+都存在于水中,在水中Mg2+与Ca2+比(即,Mg2+∶Ca2+,其是Ca2+∶Mg2+的倒数)为10∶1-1∶1,如5∶1-2∶1。在一些实施方案中,其中Ca2+和Mg2+都存在于水中,在水中Mg2+与Ca2+比(即,Mg2+∶Ca2+,其是Ca2+∶Mg2+的倒数)是4∶1。在一些实施方案中,其中Ca2+和Mg2+都存在,在沉淀材料中Ca2+与Mg2+的比(即,Ca2+∶Mg2+)为10∶1-1∶1;1∶1至1∶2.5;1∶2.5至1∶5;1∶5至1∶10;1∶10至1∶25;1∶25至1∶50;1∶50至1∶100;1∶100至1∶150;1∶150至1∶200;1∶200至1∶250;1∶250至1∶500;1∶500至1∶1000或其范围。例如,在一些实施方案中,沉淀材料中的Ca2+与Mg2+的比值在1∶1和1∶10;1∶5和1∶25;1∶10和1∶50;1∶25和1∶100;1∶50和1∶500;或1∶100和1∶1000之间。在一些实施方案中,沉淀材料中的Mg2+与Ca2+的比值(即,Mg2+∶Ca2+)在1∶1和1∶2.5;1∶2.5和1∶5;1∶5和1∶10;1∶10和1∶25;1∶25和1∶50;1∶50和1∶100;1∶100和1∶150;1∶150和1∶200;1∶200和1∶250;1∶250和1∶500;1∶500和1∶1000或其范围之间。例如,在一些实施方案中,沉淀材料中的Mg2+与Ca2+的比值在1∶1和1∶10;1∶5和1∶25;1∶10和1∶50;1∶25和1∶100;1∶50和1∶500;或1∶100和1∶1000之间。Adjusting the major ion ratio during precipitation can affect the properties of the precipitation material. The major ion ratio has a strong influence on polymorph formation. For example, as the magnesium:calcium ratio in water increases, aragonite becomes the predominant polymorph of calcium carbonate in precipitation material, compared to low-magnesium calcite. At low magnesium: calcium ratios, low-magnesium calcite becomes the predominant polymorph. In some embodiments, wherein both Ca2+ and Mg2+ are present in the water, the ratio of Ca2+ to Mg2+ (i.e., Ca2+:Mg2+) in the water is from 500:1 to 1:500, such as 100:1 to 1:100, such as 50:1 - 1:50, such as 20:1-1:20, such as 10:1-1:10. In some embodiments, wherein both Ca2+ and Mg2+ are present in the water, the ratio of Ca2+ to Mg2+ (ie, Ca2+:Mg2+) in the water is from 5:1 to 1:1. In some embodiments, wherein both Ca2+ and Mg2+ are present in the water, the ratio of Ca2+ to Mg2+ (ie, Ca2+:Mg2+) in the water is 4:1. In some embodiments, wherein both Ca2+ and Mg2+ are present in the water, the ratio of Ca2+ to Mg2+ (ie, Ca2+:Mg2+) in the water is from 1:1 to 1:10. In some embodiments, where both Ca2+ and Mg2+ are present in water, the ratio of Mg2+ to Ca2+ (i.e., Mg2+:Ca2+, which is the reciprocal of Ca2+:Mg2+) in the water is 10:1-1:1, such as 5:1- 2:1. In some embodiments, where both Ca2+ and Mg2+ are present in the water, the ratio of Mg2+ to Ca2+ (ie, Mg2+:Ca2+, which is the reciprocal of Ca2+:Mg2+) in the water is 4:1. In some embodiments, where both Ca2+ and Mg2+ are present, the ratio of Ca2+ to Mg2+ (i.e., Ca2+:Mg2+) in the precipitation material is from 10:1 to 1:1; from 1:1 to 1:2.5; from 1:2.5 to 1:5; 1:5 to 1:10; 1:10 to 1:25; 1:25 to 1:50; 1:50 to 1:100; 1:100 to 1:150; 1:150 to 1: 200; 1:200 to 1:250; 1:250 to 1:500; 1:500 to 1:1000 or the range thereof. For example, in some embodiments, the ratio of Ca2+ to Mg2+ in the precipitation material is between 1:1 and 1:10; 1:5 and 1:25; 1:10 and 1:50; 1:25 and 1:100; 1:50 and 1:500; or between 1:100 and 1:1000. In some embodiments, the ratio of Mg2+ to Ca2+ (i.e., Mg2+:Ca2+) in the precipitation material is between 1:1 and 1:2.5; 1:2.5 and 1:5; 1:5 and 1:10; 1:10 and 1:25; 1:25 and 1:50; 1:50 and 1:100; 1:100 and 1:150; 1:150 and 1:200; 1:200 and 1:250; 1:250 and 1 :500; between 1:500 and 1:1000 or the range thereof. For example, in some embodiments, the ratio of Mg2+ to Ca2+ in the precipitation material is between 1:1 and 1:10; 1:5 and 1:25; 1:10 and 1:50; 1:25 and 1:100; 1:50 and 1:500; or between 1:100 and 1:1000.
由于起始原料的变化性,包括除了钙或镁以外的平衡离子的含碳酸根的盐和化合物是可能的。例如,在一些实施方案中,本发明的组合物(例如,沉淀材料)包括霰石形式的碳酸钙。在这样的实施方案中,钙可以被许多不同的金属替代,包括但不限于锶,铅和锌,其每个,在一种形式中或者另一个,可以存在于一种或多种本发明的起始原料(例如,废气物流,质子-除去剂源,二价阳离子源,等等)中。组合物可以包括,例如,锶霰石,其是富含锶的霰石,或者组合物可以包括霰石和菱锶矿的混合物(例如,(Ca,Sr)CO3)。组合物可以包括,例如,铅霰石,其是富含铅的霰石,或者组合物可以包括霰石和白铅矿的混合物(例如,(Ca,Pb)CO3)。组合物可以包括,例如,锌霰石,其是富含Zn的霰石,或者组合物可以包括霰石和菱锌矿的混合物(例如,(Ca,Zn)CO3)。鉴于以上示范性实施方案,组合物(例如,沉淀材料)可以包括As,Ag,Ba,Be,Cd,Co,Cr,Cu,Hg,Mo,Ni,Pb,Se,Sb,Tl,V或Zn的碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐。例如,本发明的组合物可以包括Ag,Ba,Be,Cd,Co,Cu,Ni,Pb,Tl,Zn或其组合的碳酸盐。上述金属的碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐可以独立地形成(例如,菱锶矿)或者存在于镁和/或钙基质中(例如,锶霰石)。金属如As,Ag,Ba,Be,Cd,Co,Cr,Cu,Hg,Mo,Ni,Pb,Se,Sb,Tl,V和Zn可以由废气物流,质子-除去剂源,二价阳离子源或其组合提供。没有形成碳酸盐、碳酸氢盐或碳酸盐和碳酸氢盐的这样的来源(例如,废气物流,质子-除去剂源,二价阳离子源)中存在的金属及其他组分可以捕获在沉淀材料中或吸附在沉淀材料上。Due to the variability of the starting materials, carbonate-containing salts and compounds including counterions other than calcium or magnesium are possible. For example, in some embodiments, compositions (eg, precipitation materials) of the invention include calcium carbonate in the form of aragonite. In such embodiments, calcium may be replaced by a number of different metals, including but not limited to strontium, lead and zinc, each of which, in one form or another, may be present in one or more of the present invention In starting materials (eg, waste gas streams, sources of proton-removing agents, sources of divalent cations, etc.). The composition may include, for example, strontium aragonite, which is a strontium-rich aragonite, or the composition may include a mixture of aragonite and strontite (eg, (Ca,Sr)CO 3 ). The composition may include, for example, aragonite, which is a lead-rich aragonite, or the composition may include a mixture of aragonite and schenite (eg, (Ca,Pb)CO 3 ). The composition may include, for example, sauconite, which is a Zn-rich aragonite, or the composition may include a mixture of aragonite and smithsonite (eg, (Ca,Zn)CO 3 ). In view of the above exemplary embodiments, the composition (e.g., precipitation material) may include As, Ag, Ba, Be, Cd, Co, Cr, Cu, Hg, Mo, Ni, Pb, Se, Sb, Tl, V, or Zn carbonates, bicarbonates, or carbonates and bicarbonates. For example, compositions of the present invention may include carbonates of Ag, Ba, Be, Cd, Co, Cu, Ni, Pb, Tl, Zn, or combinations thereof. Carbonates, bicarbonates, or carbonates and bicarbonates of the above metals may be formed independently (eg, strontite) or present in a matrix of magnesium and/or calcium (eg, strontium aragonite). Metals such as As, Ag, Ba, Be, Cd, Co, Cr, Cu, Hg, Mo, Ni, Pb, Se, Sb, Tl, V and Zn can be produced from waste gas streams, proton-removing agent sources, divalent cation sources or a combination thereof. Metals and other components present in such sources (e.g., waste gas streams, sources of proton-removing agents, sources of divalent cations) that do not form carbonates, bicarbonates, or carbonates and bicarbonates can be trapped in the precipitate in the material or adsorbed on the precipitating material.
沉淀材料,其包括一种或多种源自工业CO2的合成碳酸盐,反映了工业CO2(来自燃烧化石燃料)源于其中的化石燃料(例如,煤,石油,天然气或烟道气)的相对碳同位素组成(δ13C)。相对碳同位素组成(δ13C)值,单位为‰(千分率)是碳的两种稳定同位素,即12C和13C,相对于化石化的箭石的标准物(PDB标准)的浓度的比值的量度。Precipitation material, which includes one or more synthetic carbonates derived from industrial CO 2 , reflecting the fossil fuel (for example, coal, oil, natural gas, or flue gas) from which industrial CO 2 (from burning fossil fuels) originated ) relative carbon isotopic composition (δ13C). The relative carbon isotopic composition (δ13C) value, in units of ‰ (parts per thousand), is a measure of the ratio of the concentrations of two stable isotopes of carbon, namely 12C and 13C, relative to the concentration of a fossilized arrowstone standard (PDB standard) .
δ13C‰=[(13C/12C样品-13C/12C PDB标准物)/(13C/12C PDB标准物)]x1000δ 13 C‰=[( 13 C/ 12 C sample - 13 C/ 12 C PDB standard)/( 13 C/ 12 C PDB standard)]x1000
因而,合成的含碳酸盐的沉淀材料的δ13C值用作CO2气体源的指纹。δ13C值可以由于不同来源(即化石燃料源)而变化,但是本发明的组合物的δ13C值通常,但不必然地,为-9‰至-35‰。在一些实施方案中,合成的含碳酸盐的沉淀材料的δ13C至是-1‰至-50‰,-5‰至-40‰,-5‰至-35‰,-7‰至-40‰,-7‰至-35‰,-9‰至-40‰,或-9‰至-35‰。在一些实施方案中,合成的含碳酸盐的沉淀材料的δ13C值小于(即,更负于)-3‰,-5‰,-6‰,-7‰,-8‰,-9‰,-10‰,-11‰,-12‰,-13‰,-14‰,-15‰,-16‰,-17‰,-18‰,-19‰,-20‰,-21‰,-22‰,-23‰,-24‰,-25‰,-26‰,-27‰,-28‰,-29‰,-30‰,-31‰,-32‰,-33‰,-34‰,-35‰,-36‰,-37‰,-38‰,-39‰,-40‰,-41‰,-42‰,-43‰,-44‰,或-45‰,其中δ13C值越负,合成的含碳酸盐的组合物越富集12C。任何合适的方法可以用于测量δ13C值,方法包括,但不限于,质谱或离轴积分腔输出光谱技术(离轴ICOS)。Thus, the δ13C value of the synthesized carbonate-containing precipitation material was used as a fingerprint of the CO2 gas source. δ13C values may vary due to different sources (ie, fossil fuel sources), but compositions of the invention typically, but not necessarily, have δ13C values in the range of -9‰ to -35‰. In some embodiments, the synthetic carbonate-containing precipitation material has a δ13C of -1‰ to -50‰, -5‰ to -40‰, -5‰ to -35‰, -7‰ to -40‰ , -7‰ to -35‰, -9‰ to -40‰, or -9‰ to -35‰. In some embodiments, the synthetic carbonate-containing precipitation material has a δ13C value less than (i.e., more negative than) -3‰, -5‰, -6‰, -7‰, -8‰, -9‰, -10‰, -11‰, -12‰, -13‰, -14‰, -15‰, -16‰, -17‰, -18‰, -19‰, -20‰, -21‰, -22 ‰, -23‰, -24‰, -25‰, -26‰, -27‰, -28‰, -29‰, -30‰, -31‰, -32‰, -33‰, -34‰, -35‰, -36‰, -37‰, -38‰, -39‰, -40‰, -41‰, -42‰, -43‰, -44‰, or -45‰, where the δ13C value is more negative , the synthesized carbonate-containing composition is more enriched in 12C. Any suitable method can be used to measure the δ13C value, including, but not limited to, mass spectrometry or off-axis integrating cavity output spectroscopy (off-axis ICOS).
除沉淀反应的含镁和钙的产物之外,使用本发明的方法和系统,包括硅、铝、铁和其它元素的化合物和材料也可被制备并且结合在沉淀材料中。这样的化合物在沉淀材料中的沉淀或者添加这样的化合物到沉淀材料中可能是被期望的以便改变包括源于该方法的沉淀材料的水泥的反应性,或者改变由其制备的固化水泥和混凝土的性能。包括金属硅酸盐的材料可以被添加到沉淀反应混合物作为这些组分的一种来源,以产生含碳酸盐的沉淀材料,其包含一种或多种组分,如无定形二氧化硅,无定形铝硅酸盐,结晶二氧化硅,硅酸钙,硅酸钙铝,等等。在一些实施方案中,沉淀材料包括碳酸盐(例如,碳酸钙,碳酸镁)和二氧化硅,其中碳酸盐∶二氧化硅比为1∶1-1∶1.5;1∶1.5-1∶2;1∶2-1∶2.5;1∶2.5-1∶3;1∶3-1∶3.5;1∶3.5-1∶4;1∶4-1∶4.5;1∶4.5-1∶5;1∶5-1∶7.5;1∶7.5-1∶10;1∶10-1∶15;1∶15-1∶20或其范围。例如,在一些实施方案中,沉淀材料包括碳酸盐和二氧化硅,其中碳酸盐∶二氧化硅比为1∶1-1∶5,1∶5-1∶10或1∶5-1∶20。在一些实施方案中,沉淀材料包括二氧化硅和碳酸盐(例如,碳酸钙,碳酸镁),其中二氧化硅∶碳酸盐比为1∶1-1∶1.5;1∶1.5-1∶2;1∶2-1∶2.5;1∶2.5-1∶3;1∶3-1∶3.5;1∶3.5-1∶4;1∶4-1∶4.5;1∶4.5-1∶5;1∶5-1∶7.5;1∶7.5-1∶10;1∶10-1∶15;1∶15-1∶20或其范围。例如,在一些实施方案中,沉淀材料包括二氧化硅和碳酸盐,其中二氧化硅∶碳酸盐比为1∶1-1∶5,1∶5-1∶10或1∶5-1∶20。概括地说,通过本发明的方法生产的沉淀材料包括硅基材料和至少一种碳酸盐相的混合物。概括地说,反应速率越快,越多二氧化硅结合以含碳酸盐的沉淀材料,前提是二氧化硅存在于沉淀反应混合物(即,前提是在包括金属硅酸盐的材料消化后没有除去二氧化硅)。In addition to the magnesium and calcium containing products of the precipitation reaction, compounds and materials including silicon, aluminum, iron and other elements can also be prepared and incorporated in the precipitation material using the methods and systems of the present invention. The precipitation of such compounds in the precipitation material or the addition of such compounds to the precipitation material may be desirable in order to alter the reactivity of the cement comprising the precipitation material resulting from the process, or to alter the properties of the cured cement and concrete produced therefrom. performance. Materials comprising metal silicates may be added to the precipitation reaction mixture as a source of these components to produce carbonate-containing precipitation material comprising one or more components such as amorphous silica, Amorphous aluminosilicate, crystalline silica, calcium silicate, calcium aluminum silicate, etc. In some embodiments, the precipitation material includes carbonates (e.g., calcium carbonate, magnesium carbonate) and silica, wherein the carbonate:silica ratio is 1:1-1:1.5; 1:1.5-1: 2; 1:2-1:2.5; 1:2.5-1:3; 1:3-1:3.5; 1:3.5-1:4; 1:4-1:4.5; 1:4.5-1:5; 1:5-1:7.5; 1:7.5-1:10; 1:10-1:15; 1:15-1:20 or the range thereof. For example, in some embodiments, the precipitation material includes carbonate and silica, wherein the carbonate:silica ratio is 1:1-1:5, 1:5-1:10, or 1:5-1 : 20. In some embodiments, the precipitation material comprises silica and carbonate (e.g., calcium carbonate, magnesium carbonate), wherein the silica:carbonate ratio is 1:1-1:1.5; 1:1.5-1: 2; 1:2-1:2.5; 1:2.5-1:3; 1:3-1:3.5; 1:3.5-1:4; 1:4-1:4.5; 1:4.5-1:5; 1:5-1:7.5; 1:7.5-1:10; 1:10-1:15; 1:15-1:20 or the range thereof. For example, in some embodiments, the precipitation material includes silica and carbonate, wherein the silica:carbonate ratio is 1:1-1:5, 1:5-1:10 or 1:5-1 : 20. In general terms, the precipitation material produced by the method of the present invention comprises a mixture of silicon-based material and at least one carbonate phase. In summary, the faster the reaction rate, the more silica is incorporated into the carbonate-containing precipitation material, provided that silica is present in the precipitation reaction mixture (i.e., provided that no remove silica).
沉淀材料可以处于存储稳定的形式(其可以简单地是空气干燥的沉淀材料),并且可以在地上在暴露条件下存储(即,对大气开放),而没有显著的,如果有的话,分解(或CO2损失)达延长的时间。在一些实施方案中,沉淀材料可以在暴露条件下是稳定的达1年或更长,5年或更长,10年或更长,25年或更长,50年或更长,100年或更长,250年或更长,1000年或更长,10,000年或更长,1,000,000年或更长,或甚至100,000,000年或更长。存储稳定形式的沉淀材料可以在各种不同的环境条件下是稳定的,例如温度-100℃至600℃和湿度0-100%,其中条件可以是无风、有风或暴风的。因为当在正常的雨水pH下在地上存储时,存储稳定形式的沉淀材料经历很小,如果有的话,的分解,分解的量,如果有的话,按从产物中的CO2气体释放来测量的,不超过5%每年,在某些实施方案中不会超过1%每年或0.001%每年。实际上,由本发明提供的沉淀材料,没有释放大于1%、5%或10%的其全部CO2,当暴露于温度和湿度的标准条件时,包括正常PH的降雨,达至少1、2、5、10或20年,或者达超过20年,例如,超过100年。在一些实施方案中,沉淀材料没有释放大于1%的其全部CO2,当暴露于温度和湿度的标准条件时,包括正常PH的降雨,达至少1年。在一些实施方案中,沉淀材料没有释放大于5%的其全部CO2,当暴露于温度和湿度的标准条件时,包括正常PH的降雨,达至少1年。在一些实施方案中,沉淀材料没有释放大于10%的其全部CO2,当暴露于温度和湿度的标准条件时,包括正常PH的降雨,达至少1年。在一些实施方案中,沉淀材料没有释放大于1%的其全部CO2,当暴露于温度和湿度的标准条件时,包括正常PH的降雨,达至少10年。在一些实施方案中,沉淀材料没有释放大于1%的其全部CO2,当暴露于温度和湿度的标准条件时,包括正常PH的降雨,达至少100年。在一些实施方案中,沉淀材料没有释放大于1%的其全部CO2,当暴露于温度和湿度的标准条件时,包括正常PH的降雨,达至少1000年。The precipitation material may be in a storage stable form (which may simply be air-dried precipitation material) and may be stored above ground under exposed conditions (i.e., open to the atmosphere) without significant, if any, decomposition ( or CO 2 loss) for extended periods of time. In some embodiments, the precipitation material can be stable under exposure conditions for 1 year or longer, 5 years or longer, 10 years or longer, 25 years or longer, 50 years or longer, 100 years or Longer, 250 years or more, 1,000 years or more, 10,000 years or more, 1,000,000 years or more, or even 100,000,000 years or more. Precipitation material in storage stable form may be stable under various environmental conditions, eg temperature -100°C to 600°C and humidity 0-100%, where conditions may be calm, windy or stormy. Because when stored above ground at normal rainwater pH, the stored stable form of the precipitated material undergoes little, if any, decomposition, and the amount of decomposition, if any, in terms of CO2 gas release from the product Measured, not to exceed 5% per annum, in certain embodiments not to exceed 1% per annum or 0.001% per annum. Indeed, the precipitation material provided by the present invention does not release more than 1%, 5% or 10% of its total CO2 when exposed to standard conditions of temperature and humidity, including rainfall at normal pH, up to at least 1, 2, 5, 10 or 20 years, or up to more than 20 years, for example, more than 100 years. In some embodiments, the precipitation material does not release greater than 1% of its total CO2 when exposed to standard conditions of temperature and humidity, including rainfall at normal pH, for at least 1 year. In some embodiments, the precipitation material does not release greater than 5% of its total CO2 when exposed to standard conditions of temperature and humidity, including rainfall at normal pH, for at least 1 year. In some embodiments, the precipitation material does not release greater than 10% of its total CO2 when exposed to standard conditions of temperature and humidity, including rainfall at normal pH, for at least 1 year. In some embodiments, the precipitation material has not released greater than 1% of its total CO2 when exposed to standard conditions of temperature and humidity, including rainfall at normal pH, for at least 10 years. In some embodiments, the precipitation material has not released greater than 1% of its total CO2 when exposed to standard conditions of temperature and humidity, including rainfall at normal pH, for at least 100 years. In some embodiments, the precipitation material has not released greater than 1% of its total CO2 when exposed to standard conditions of temperature and humidity, including rainfall at normal pH, for at least 1000 years.
可以使用任何合适的能够合理地预知这样的稳定性的代用标记或测试。例如,加速试验,包括高温条件和/或适度至更极端的pH条件,能够合理地表示在延长的时间上的稳定性。例如,取决于沉淀材料的预定用途和环境,沉淀材料的样品可以暴露于50、75、90、100、120或150℃在10%-50%相对湿度达1、2、5、25、50、100、200或500天,并且其碳的损失小于1%、2%、3%、4%、5%、10%、20%、30%或50%可以被认为是本发明的沉淀材料的稳定性达一段时间(例如,1、10、100、1000或大于1000年)的充足证据。Any suitable surrogate marker or test that is reasonably predictable of such stability may be used. For example, accelerated testing, including elevated temperature conditions and/or moderate to more extreme pH conditions, can reasonably indicate stability over extended periods of time. For example, depending on the intended use and environment of the precipitation material, a sample of the precipitation material may be exposed to 1, 2, 5, 25, 50, 100, 200 or 500 days, and its loss of carbon is less than 1%, 2%, 3%, 4%, 5%, 10%, 20%, 30% or 50% can be considered stable for the precipitation material of the present invention Sufficient evidence of persistence over a period of time (eg, 1, 10, 100, 1000, or greater than 1000 years).
许多合适的方法可以用于测试沉淀材料的稳定性,包括物理测试方法和化学测试方法,其中所述方法适于确定沉淀材料中的化合物类似于或等同于已知具有上述稳定性的天然存在的化合物(例如,石灰石)。通过任何合适的方法可以监控沉淀材料的CO2含量,一个这样的非限制性实例是电量分析。其他条件可以被调节,视情况而定,包括pH、压力、UV辐射等,这也取决于预期或可能的环境。应当理解的是可以使用任何适宜条件,其是本领域技术人员将合理地推断的,指出在所指出的时间内必需的稳定性。另外,如果被接受的化学知识指出沉淀材料将具有必需的稳定性达所指出的时间,这也可以被使用,除了实际测量之外或者代替实际测量。例如,某些碳酸盐化合物,其可能是本发明的沉淀材料的一部分(例如,以给定的多晶型物形式),可能是地质学上众所周知的并且已知经受了正常气候达数十年,数百年,或者甚至数千年,而没有明显的分解,并且因此具有必需的稳定性。A number of suitable methods can be used to test the stability of the precipitation material, including physical testing methods and chemical testing methods, where the methods are suitable for determining that the compounds in the precipitation material are similar or equivalent to naturally occurring compounds known to have the above-mentioned stability. compounds (eg, limestone). The CO2 content of the precipitation material can be monitored by any suitable method, one such non-limiting example is coulometric analysis. Other conditions may be adjusted as appropriate, including pH, pressure, UV radiation, etc., also depending on the expected or likely environment. It should be understood that any suitable conditions may be used, which a person skilled in the art would reasonably infer, given the necessary stability over the indicated times. Additionally, if accepted chemical knowledge indicates that the precipitation material will have the requisite stability for the time indicated, this may also be used in addition to or instead of actual measurements. For example, certain carbonate compounds, which may be part of the precipitation material of the present invention (e.g., in a given polymorphic form), may be geologically well known and known to have survived normal climates for tens of years, hundreds, or even thousands of years without appreciable decomposition, and thus have the requisite stability.
含碳酸盐的沉淀材料,其用来以在长时间(例如,地质时标)内稳定的形式埋存CO2,可以存储达长时间,如上所述。沉淀材料,如果需要获得某一比例的碳酸盐与二氧化硅,也可混合以硅基材料(例如,来自在包括金属硅酸盐的材料消化后的硅基材料;市售可得的SiO2;等等)来形成火山灰质材料。本发明的火山灰质材料是硅质或铝硅酸材料,其当与碱如氢氧化钙(Ca(OH)2)结合时,通过形成硅酸钙及其他粘结材料显示出粘结性能。含SiO2的材料如火山灰,飞灰,二氧化硅烟雾,高反应性偏高岭土和磨碎的粒状高炉炉渣等可以用于增强产生火山灰质材料的本发明的组合物。在一些实施方案中,本发明的火山灰质材料强化有0.5%至1.0%,1.0%至2.0%;2.0%至4.0%,4.0%至6.0%,6.0%至8.0%,8.0%至10.0%,10.0%至15.0%,15.0%至20.0%,20.0%至30.0%,30.0%至40.0%,40.0%至50.0%或其交叠范围,含SiO2的材料。这样的含SiO2的材料可以获自,例如,本发明的静电沉降器或织物过滤器。The carbonate-containing precipitation material used to sequester CO2 in a form that is stable over long periods of time (eg, geological timescales) can be stored for extended periods of time, as described above. The precipitation material, if desired to obtain a certain ratio of carbonate to silica, can also be mixed with a silicon-based material (e.g., from a silicon-based material after digestion of a material including metallosilicates; commercially available SiO 2 ; etc.) to form pozzolanic materials. The pozzolanic materials of the present invention are siliceous or aluminosilicate materials which, when combined with a base such as calcium hydroxide (Ca(OH)2), exhibit bonding properties by forming calcium silicates and other bonding materials. SiO2 -containing materials such as pozzolans, fly ash, silica fume, highly reactive metakaolin and ground granulated blast furnace slag, among others, can be used to enhance compositions of the present invention that produce pozzolanic materials. In some embodiments, the pozzolanic materials of the present invention are enhanced with 0.5% to 1.0%, 1.0% to 2.0%; 2.0% to 4.0%, 4.0% to 6.0%, 6.0% to 8.0%, 8.0% to 10.0%, 10.0% to 15.0%, 15.0% to 20.0%, 20.0% to 30.0%, 30.0% to 40.0%, 40.0% to 50.0%, or their overlapping ranges, SiO2 -containing materials. Such SiO2 -containing materials can be obtained, for example, from electrostatic precipitators or fabric filters of the present invention.
如上所指出,在一些实施方案中,沉淀材料包括亚稳定的碳酸盐化合物,其在盐水中比在淡水中更稳定,使得当与任何pH的淡水接触时,其溶解并且再沉淀为其它淡水稳定矿物。在某些实施方案中,碳酸盐化合物以小颗粒的形式存在,例如,粒度为0.1μm至100μm,1至100μm,10-100μm,50至100μm,如通过扫描电子显微术扫描电子显微术(SEM)测定的。在一些实施方案中,碳酸盐化合物的粒度为0.5至10μm,如由SEM测定的。在一些实施方案中,粒径显示出单峰分布。在一些实施方案中,粒度显示出双峰或多峰分布。在某些实施方案中,颗粒具有高表面积,例如,0.5至100m2/gm,0.5至50m2/gm,或0.5至2.m2/gm,如通过BET(Brauner,Emmit,&Teller)表面积分析测定的。在一些实施方案中,沉淀材料可以包括杆状晶体和/或非晶固体。杆状晶体可以在结构上不同,并且在某些实施方案中具有500至1、250至1或10至1的长度直径比。在某些实施方案中,晶体的长度为0.5μm至500μm,1μm至250μm或5μm至100μm。在其它实施方案中,产生了基本上完全非晶固体。As noted above, in some embodiments, the precipitation material includes metastable carbonate compounds that are more stable in salt water than in fresh water such that when contacted with fresh water at any pH, it dissolves and reprecipitates as other fresh water Stabilizing minerals. In certain embodiments, the carbonate compound is present in the form of small particles, e.g., a particle size of 0.1 μm to 100 μm, 1 to 100 μm, 10-100 μm, 50 to 100 μm, as detected by scanning electron microscopy. determined by SEM. In some embodiments, the carbonate compound has a particle size of 0.5 to 10 μm, as determined by SEM. In some embodiments, the particle sizes exhibit a monomodal distribution. In some embodiments, the particle size exhibits a bimodal or multimodal distribution. In certain embodiments, the particles have a high surface area, e.g., 0.5 to 100 m/gm, 0.5 to 50 m/gm, or 0.5 to 2.m/gm, as determined by BET (Brauner, Emmit, & Teller) surface area analysis. In some embodiments, the precipitation material can include rod-shaped crystals and/or amorphous solids. Rod crystals can vary in structure, and in certain embodiments have a length to diameter ratio of 500 to 1, 250 to 1, or 10 to 1. In certain embodiments, the crystals have a length of 0.5 μm to 500 μm, 1 μm to 250 μm, or 5 μm to 100 μm. In other embodiments, substantially completely amorphous solids are produced.
喷雾干燥的材料(例如,沉淀材料,硅基材料,火山灰质材料,等等),借助于喷雾干燥,可以具有一致的粒径(即,喷雾干燥的材料可以具有相对窄的粒度分布)。因而,在一些实施方案中,至少50%、60%、70%、80%、90%、95%、97%或99%的喷雾干燥的材料落入给定的平均粒径的±10微米、±20微米、±30微米、±40微米、±50微米、±75微米、±100微米或±250微米。在一些实施方案中,给定的平均粒径为5-500微米。在一些实施方案中,给定的平均粒径为5-250微米。在一些实施方案中,给定的平均粒径为5-100微米。在一些实施方案中,给定的平均粒径为5-50微米。在一些实施方案中,给定的平均粒径为5-25微米。例如,在一些实施方案中,至少70%的喷雾干燥的材料落入给定的平均粒径的±50微米,其中给定的平均粒径为5-500微米,如50-250微米或100-200微米。这样的喷雾干燥的材料可以用于制造本发明的水泥,细集料,灰浆,粗集料,混凝土和/或火山灰水泥;然而,本领域技术人员将认识到水泥,细集料,灰浆,粗集料,混凝土和/或火山灰水泥的制造不需要喷雾干燥的沉淀材料。空气干燥的沉淀材料,例如,也可用于制造本发明的水泥,细集料,灰浆,粗集料,混凝土和/或火山灰水泥。The spray-dried material (eg, precipitation material, silica-based material, pozzolanic material, etc.), can have a consistent particle size (ie, the spray-dried material can have a relatively narrow particle size distribution) by virtue of spray drying. Thus, in some embodiments, at least 50%, 60%, 70%, 80%, 90%, 95%, 97%, or 99% of the spray-dried material falls within ±10 microns, ±20 microns, ±30 microns, ±40 microns, ±50 microns, ±75 microns, ±100 microns or ±250 microns. In some embodiments, the given average particle size is 5-500 microns. In some embodiments, the given average particle size is 5-250 microns. In some embodiments, the given average particle size is 5-100 microns. In some embodiments, the given average particle size is 5-50 microns. In some embodiments, the given average particle size is 5-25 microns. For example, in some embodiments, at least 70% of the spray-dried material falls within ±50 microns of a given mean particle size, where the given mean particle size is 5-500 microns, such as 50-250 microns or 100- 200 microns. Such spray-dried materials can be used to make the cement, fine aggregate, mortar, coarse aggregate, concrete and/or pozzolan cement of the present invention; however, those skilled in the art will recognize that cement, fine aggregate, mortar, coarse The manufacture of aggregates, concrete and/or pozzolan cement does not require spray-dried settling material. Air-dried precipitation materials, for example, can also be used to make cements, fine aggregates, mortars, coarse aggregates, concrete and/or pozzolan cements of the present invention.
实施例Example
与上面的说明书相结合,将下面的实施例提供给本领域技术人员来完全的公开和说明如何制造和使用本发明。提出该实施例来提供据信是对于本发明的某些实施方案的最有用的和容易的理解程序和概念上的说明。同样,该实施例目的并非限制本发明人所认为的他们发明的范围,并且该实施例也不是提出全部的试验或者仅仅提出所进行的试验。已经进行了努力来确保所用的数值(例如用量,温度等等)的精度,但是应当会存在着一些试验误差和偏差。除非另有指示,份数是重量份,分子量是重均分子量,温度是摄氏度,压力处于或者接近于大气压。In conjunction with the above description, the following examples are provided to those skilled in the art to fully disclose and illustrate how to make and use the invention. This example is presented to provide what is believed to be the most useful and readily understood procedural and conceptual illustration of certain embodiments of the invention. Likewise, this example is not intended to limit the scope of what the inventors believe their invention to be, nor does this example set forth all of the tests or only those that were performed. Efforts have been made to ensure accuracy with respect to numbers used (eg, amounts, temperature, etc.), but some experimental errors and deviations should be accounted for. Unless indicated otherwise, parts are parts by weight, molecular weight is weight average molecular weight, temperature is in degrees Centigrade, and pressure is at or near atmospheric.
实施例I.P00099的沉淀Example I. Precipitation of P00099
A.P00099沉淀方法A.P00099 precipitation method
使用下面的方案来产生P00099沉淀物。将380L的过滤海水泵入到圆柱的聚乙烯60°锥形底部渐变的槽子中。该反应槽是一种开放系统,左边暴露于环境大气。使用顶部混合器持续搅拌该反应槽。在整个反应过程中持续监控pH,室温和水温。The following protocol was used to generate the P00099 precipitate. 380L of filtered seawater is pumped into
将25g颗粒状(Ca,Mg)O(也称作煅烧白云石或者煅烧的白云石)混入该海水中。将沉降到槽底的煅烧白云石手工从槽底通过顶部重新进行再循环,来促进反应物的充分混合和溶解。以相同的方式加入第二个25g的煅烧白云石,包括沉降反应物的手工再循环。当该水的pH达到9.2时,将10%的CO2(和90%的压缩空气)的气体混合物通过陶瓷气石缓慢的扩散到溶液中。当该溶液的pH下降到9.0时,将另外25g添加量的煅烧白云石加入到所述反应槽中,其引起pH重新上升。当该溶液的pH不断下降到9.0(或者以下)时,重复加入煅烧白云石,直到加入总量为225g为止。在每个煅烧白云石加入之间手工进行沉降的反应物的再循环。25 g of particulate (Ca,Mg)O (also known as calcined dolomite or calcined dolomite) were mixed into the seawater. Calcined dolomite that has settled to the bottom of the tank is manually recirculated from the bottom of the tank through the top to promote thorough mixing and dissolution of the reactants. A second 25 g of calcined dolomite was added in the same manner, including manual recirculation of the settled reactants. When the pH of the water reached 9.2, a gas mixture of 10% CO2 (and 90% compressed air) was slowly diffused into the solution through a ceramic airstone. When the pH of the solution dropped to 9.0, an additional 25 g addition of calcined dolomite was added to the reaction tank, which caused the pH to rise again. When the pH of the solution continued to drop to 9.0 (or below), the calcined dolomite was added repeatedly until the total amount added was 225 g. Recirculation of the settled reactants was performed manually between each calcined dolomite addition.
在最后加入煅烧白云石之后,停止气体通过该溶液的连续扩散。将该反应搅拌另外2小时。在此期间,pH持续升高。为了将pH保持在9.0-9.2,当pH升高到高于9.2时,将另外的气体扩散通过该反应,直到它达到9.0。在这2小时的期间内,还进行了4次沉降的反应物的手工再循环。After the final addition of calcined dolomite, the continuous diffusion of gas through the solution was stopped. The reaction was stirred for an additional 2 hours. During this period, the pH continued to rise. To maintain the pH at 9.0-9.2, when the pH rises above 9.2, additional gas is diffused through the reaction until it reaches 9.0. During this 2 hour period, 4 manual recirculations of the settled reactants were also performed.
在最后加入煅烧白云石之后2小时,停止搅拌、气体扩散和沉降的反应物的再循环。将该反应槽静置15小时(开放到大气中)。Two hours after the last addition of calcined dolomite, stirring, gas diffusion and recirculation of settled reactants were stopped. The reaction tank was left to stand for 15 hours (opened to the atmosphere).
在15小时的期间之后,使用潜水泵从该反应槽的顶部除去上澄液。将其余的混合物从槽底除去。将所收集的混合物沉降2小时。在沉降之后,滗析上澄液。将其余的浆体通过11μm孔径的滤纸在Büchner漏斗中进行真空过滤。将所收集的滤饼放入耐热玻璃盘中,并且在110℃焙烤24小时。After a period of 15 hours, the supernatant was removed from the top of the reaction tank using a submersible pump. The rest of the mixture was removed from the bottom of the tank. The collected mixture was allowed to settle for 2 hours. After settling, the supernatant was decanted. The rest of the slurry was vacuum filtered in a Büchner funnel through 11 μm pore size filter paper. The collected filter cake was put into a heat-resistant glass dish and baked at 110° C. for 24 hours.
将该干燥的产物在球磨机中研磨,并且通过一系列的筛网进行尺寸分级,来产生P00099沉淀物。The dried product was ground in a ball mill and size fractionated through a series of screens to produce a P00099 precipitate.
B.材料分析B. Material Analysis
在所收集的不同的筛分中,仅仅使用这样的筛分,其含有保留在38μm开口筛上,并且通过75μm开口筛的粒子。Of the different sieves collected, only those containing particles retained on the 38 μm opening sieve and passed through the 75 μm opening sieve were used.
1.化学特性1. Chemical properties
使用XRF来分析用于共混的P00099沉淀物的元素组成。在下表4中报告了用于在该共混物中所用的QUIKRETETM I/II型波特兰水泥以及用于P00099沉淀物的主要元素的结果。XRF was used to analyze the elemental composition of the P00099 precipitates used for blending. The results for the QUIKRETE™ Type I/II Portland cement used in the blend and for the major elements of the P00099 precipitate are reported in Table 4 below.
表4.在这种共混物中所用的I/II型波特兰水泥和P00099-002的XRF分析。Table 4. XRF analysis of Type I/II Portland cement and P00099-002 used in this blend.
该沉淀物的XRD分析表明存在着霰石和镁方解石(组成接近于Mg0.1Ca0.9CO3)和少量的氢氧镁石和岩盐(表5)。该P00099沉淀物的FT-IR分析证实存在着霰石,方解石和氢氧镁石。XRD analysis of the precipitate showed the presence of aragonite and magnesium calcite (composition close to Mg 0.1 Ca 0.9 CO 3 ) and small amounts of bruxite and halite (Table 5). FT-IR analysis of the P00099 precipitate confirmed the presence of aragonite, calcite and borschite.
表5.该沉淀物的XRD分析。Table 5. XRD analysis of the precipitate.
通过电量分析所测量的总无机碳含量与这样的值相当的符合,该值来自于结合有XRF元素组成的XRD Rietveld估计的组成。表6提供P00099的库仑分析,与来自XRD/XRF数据的%C进行比较。The total inorganic carbon content measured by coulometric analysis is in good agreement with the values derived from the XRD Rietveld estimated composition combined with the XRF elemental composition. Table 6 provides the coulometric analysis of P00099 compared to %C from XRD/XRF data.
表6.P00099的库仑分析,与来自XRD/XRF数据的%C进行比较。Table 6. Coulomb analysis of P00099 compared to %C from XRD/XRF data.
2.物理特性2. Physical properties
在该沉淀物上的SEM观察证实了占优势的霰石(针状)以及粒子聚集体的尺寸。所测定的波特兰水泥和P00099沉淀物的BET比表面积(“SSA”)在表7中给出。SEM observations on the precipitate confirmed the predominance of aragonite (acicular) and the size of particle aggregates. The measured BET specific surface areas ("SSA") of Portland cement and P00099 precipitates are given in Table 7.
表7.波特兰水泥和P00099沉淀物的BET比表面积(“SSA”)。Table 7. BET specific surface area ("SSA") of Portland cement and P00099 precipitates.
在2min的用于分离聚集粒子的预超声波分散之后,测定了粒度分布。The particle size distribution was determined after 2 min of pre-ultrasonic dispersion for separating aggregated particles.
实施例II.使用粉煤灰作为碱源Example II. Use of Fly Ash as Alkali Source
A.方法A. method
使用磁搅拌棒将500mol的海水(初始pH=8.01)在玻璃烧杯中连续搅拌。持续监控该反应的pH和温度。将F等级的粉煤灰(~10%CaO)作为粉末增量加入,使得pH在加入之间保持平衡。500 mol of seawater (initial pH = 8.01) was continuously stirred in a glass beaker using a magnetic stirring bar. The pH and temperature of the reaction were continuously monitored. Class F fly ash (-10% CaO) was added as a powder increment such that the pH was balanced between additions.
B.结果和观察:B. Results and Observations:
在加入5.00g粉煤灰之后,pH达到9.00。After adding 5.00 g of fly ash, the pH reached 9.00.
34.14g-->pH9.5034.14g-->pH9.50
168.89g-->pH9.76168.89g-->pH9.76
219.47g-->pH10.94219.47g-->pH10.94
254.13g-->pH11.20254.13g-->pH11.20
300.87g-->pH11.28300.87g-->pH11.28
(所列出的粉煤灰量是累积的总量,即,在试验时间点中所加入的总量。)(The amount of fly ash listed is the cumulative total, ie, the total amount added during the test time points.)
需要比蒸馏水多出很多的粉煤灰来提高海水的pH。初始pH升高(8-9)需要比进一步升高少得多的粉煤灰。在大部分的反应时间内pH相当稳定的保持在9.7左右。在~10之后pH的升高速率加快。同样要注意的是当加入粉煤灰时,pH的初始降低。这种pH的降低通过氢氧化钙的作用而得以快速克服。来自该反应的真空干燥的浆体的SEM图像表明一些粉煤灰球已经部分溶解。其余的球体似乎还埋入在可能似水泥的材料中。Much more fly ash than distilled water is required to raise the pH of seawater. The initial pH increase (8-9) required much less fly ash than further increases. The pH remained fairly constant at around 9.7 for most of the reaction time. The rate of increase in pH accelerated after ~10. Also of note is the initial drop in pH when fly ash is added. This pH drop is quickly overcome by the action of calcium hydroxide. SEM images of the vacuum-dried slurry from this reaction indicated that some fly ash spheres had partially dissolved. The rest of the spheres appear to be embedded in what may be a cement-like material.
C.结论c. conclusion
在淡(蒸馏)水中,已经发现少量的F等级粉煤灰(<1g/L)立即将pH从7(中性)升高到~11。升高pH所必需的该小的量很可能归因于蒸馏水的无缓冲性能。海水是被碳酸盐高度缓冲的,并因此它消耗了大量的粉煤灰来将pH升高到类似的程度。In dilute (distilled) water, small amounts of Class F fly ash (<1 g/L) have been found to immediately raise the pH from 7 (neutral) to ~11. This small amount necessary to raise the pH is likely due to the unbuffered properties of distilled water. Seawater is highly buffered by carbonates, and thus it consumes large amounts of fly ash to raise the pH to a similar degree.
实施例III.高产率的生产Example III. High Yield Production
A.方法1
将20%CO2/80%空气的气体混合物喷入1L海水中,直到达到pH<5。一旦达到,将1.0g的Mg(OH)2加入到该1L的碳酸/海水溶液中。将该20/80气体混合物持续喷布20分钟,来保证Mg(OH)2和气体的最大溶解。在溶解之后,停止喷布,加入2M的NaOH,直到达到pH为9.8。重新开始20/80气体的喷布,直到达到pH8.5。继续2M NaOH和相反加入20/80气体来将pH保持在8.5-9.8,直到加入了总共200ml的2MNaOH。观察到6.91g的产量,并且具有10.6%碳(~80%碳酸盐)的电量计读数。A gas mixture of 20% CO2 /80% air was sparged into 1 L of seawater until a pH<5 was reached. Once reached, 1.0 g of Mg(OH) 2 was added to the 1 L of carbonic acid/seawater solution. The 20/80 gas mixture was sprayed for 20 minutes to ensure maximum dissolution of Mg(OH) 2 and gas. After dissolution, spraying was stopped and 2M NaOH was added until a pH of 9.8 was reached. The 20/80 gas spray was restarted until pH 8.5 was reached. Continue 2M NaOH and reverse 20/80 gas addition to maintain the pH at 8.5-9.8 until a total of 200ml of 2M NaOH has been added. A yield of 6.91 g was observed with a coulometer reading of 10.6% carbon (-80% carbonate).
B.方法2B. Method 2
将20%CO2/80%空气的气体混合物喷入1L海水中,直到达到pH<5。一旦达到,将2.69g的Mg(OH)2加入到该1L的碳酸/海水溶液中。将该20/80气体混合物持续喷布20分钟,来保证Mg(OH)2和气体的最大溶解。在溶解之后,停止喷布,加入2M的NaOH,直到达到pH为9.8。重新开始20/80气体的喷布,直到达到pH8.5。继续2M NaOH和相反加入20/80气体来将pH保持在8.5-9.8,直到加入了总共200ml的2MNaOH。观察到10.24g的产量,并且具有9.7%碳(~75%碳酸盐)的电量计读数。A gas mixture of 20% CO2 /80% air was sparged into 1 L of seawater until a pH<5 was reached. Once reached, 2.69 g of Mg(OH) 2 was added to the 1 L of carbonic acid/seawater solution. The 20/80 gas mixture was sprayed for 20 minutes to ensure maximum dissolution of Mg(OH) 2 and gas. After dissolution, spraying was stopped and 2M NaOH was added until a pH of 9.8 was reached. The 20/80 gas spray was restarted until pH 8.5 was reached. Continue 2M NaOH and reverse 20/80 gas addition to maintain the pH at 8.5-9.8 until a total of 200ml of 2M NaOH has been added. A yield of 10.24 g was observed with a coulometer reading of 9.7% carbon (-75% carbonate).
该沉淀的碳酸盐的SEM,EDS和X射线衍射表明存在着无定形的和结晶的Ca和Mg碳酸盐,以及存在着Ca/Mg碳酸盐。沉淀物的图片提供在图41和42中。SEM, EDS and X-ray diffraction of the precipitated carbonates indicated the presence of amorphous and crystalline Ca and Mg carbonates, as well as the presence of Ca/Mg carbonates. Pictures of the precipitate are provided in Figures 41 and 42.
C.方法3C. Method 3
将CO2喷入1L海水,直到达到pH7或者更低。加入0-5.0g Mg离子补充物(称作“Moss Mag”,并且获自Calera Corporation′s Moss Landing地点,其是Kaiser Aluminum&Chemical Corporation and NationalRefractorie in Moss Landing California的以前的地点,这里该补充物是在该地点的尾料池中存在的富含Mg的废产物),同时混合和连续喷布CO2。加入0.175ppm的Al2(SO4)3。连续的喷布CO2和加入碱,同时将pH保持在7到8,终止于pH为7。停止喷布CO2,并加入碱,直到达到pH为9.0到10.4。如图43所示,上面的反应条件有利于形成无定形碳酸盐化合物沉淀。所形成的无定形沉淀产物容易喷雾干燥来产生干燥产物。Spray CO2 into 1L of seawater until pH7 or lower is reached. Add 0-5.0 g of Mg ion supplement (called "Moss Mag" and obtained from Calera Corporation's Moss Landing site, former site of Kaiser Aluminum & Chemical Corporation and National Refractorie in Moss Landing California, where the supplement is at Mg-rich waste products present in the tailings pond at the site) while mixing and continuously sparging CO2 . 0.175 ppm of Al 2 (SO 4 ) 3 was added. Continuous sparging of CO2 and addition of base while maintaining the pH at 7 to 8, terminated at
D.方法4
如图38,39和40所示,在某些实施方案中,使用多步骤,多反应器方法来进行此处所公开的方法。在第一反应器中,将获自加利福尼亚州Moss Landing地点的镁离子源(下文称作Moss Mag)放入使用碳酸的溶液中,并且搅拌。将该第一反应器中的海水的pH在Moss Mag溶解过程中保持在pH为7.0或者更低。在某些实施方案中,将1.0g的50-150μmMoss Mag溶解在每1L海水中成为溶液。6.2-6.6的pH或者>0.08g/l的硬度读数表明适量的Moss Mag溶解在该溶液中。将CO2源例如烟道气喷入第一反应器的水中。在整个反应中所消耗的大约40-50%的总烟道气在这个步骤中溶解到了该海水中。喷布烟道气,直到pH不再响应烟道气的溶解,这花费了大约30-60分钟。停止搅拌,来使得未反应的MossMag,沙子或者其他大粒子重力沉降,然后将带有CO2的酸性水从第一反应器转移到第二反应器。As shown in Figures 38, 39 and 40, in certain embodiments, a multi-step, multi-reactor approach is used to carry out the methods disclosed herein. In the first reactor, a source of magnesium ions obtained from the Moss Landing, California site (hereinafter referred to as Moss Mag) was placed in the solution using carbonic acid and stirred. The pH of the seawater in the first reactor was maintained at pH 7.0 or lower during the Moss Mag dissolution process. In certain embodiments, 1.0 g of 50-150 μm Moss Mag is dissolved per 1 L of seawater to form a solution. A pH of 6.2-6.6 or a hardness reading of >0.08 g/l indicates that the appropriate amount of Moss Mag is dissolved in the solution. A source of CO2 such as flue gas is sparged into the water in the first reactor. About 40-50% of the total flue gas consumed in the whole reaction is dissolved into the seawater in this step. The flue gas was sprayed until the pH no longer responded to the dissolution of the flue gas, which took approximately 30-60 minutes. Stirring is stopped to allow unreacted MossMag, sand or other large particles to settle by gravity, then the acidic water with CO2 is transferred from the first reactor to the second reactor.
然后将带有CO2的酸性水从第一反应器转移到第二反应器。该第二反应器用于产生成核位置和晶体生长二者。在该溶液从第一反应器转移到第二反应器之后,进行下面的步骤:The acidic water with CO2 is then transferred from the first reactor to the second reactor. This second reactor is used both to create nucleation sites and for crystal growth. After the solution was transferred from the first reactor to the second reactor, the following steps were carried out:
1.加入50%的NaOH,直到达到pH为9.5。例如对于1000加仑的反应来说,使用计量泵加入20-25kg的50%NaOH,该泵能够泵送5-25ml/s的50%NaOH。在达到pH为9.5之后,停止加入50%NaOH。1. Add 50% NaOH until a pH of 9.5 is reached. For example for a 1000 gallon reaction, 20-25 kg of 50% NaOH is added using a metering pump capable of pumping 5-25 ml/s of 50% NaOH. After reaching a pH of 9.5, the addition of 50% NaOH was stopped.
2.将包括20%CO2/80%压缩空气的混合物的CO2源喷入第二反应器,直到达到pH8.5。在达到pH为8.5之后,停止CO2的喷布。2. A CO2 source comprising a mixture of 20% CO2 /80% compressed air was sparged into the second reactor until pH 8.5 was reached. After reaching a pH of 8.5, the CO sparging was stopped.
3.进行加入50%NaOH到反应器中来升高pH和喷布CO2来降低pH这样的交替步骤。在交替加入50%NaOH和喷布CO2的过程中将pH保持在8.5-9.8。持续的交替计量加入50%NaOH和喷布CO2,直到向反应器中加入了总共90kg(即,在该步骤中的65-70kg+在第一步骤中的20-25kg)的50%NaOH。3. Alternate steps of adding 50% NaOH to the reactor to raise the pH and sparging CO2 to lower the pH. The pH was maintained at 8.5–9.8 during the alternating addition of 50% NaOH and sparging of CO2 . The alternating metering of 50% NaOH and sparging of CO2 was continued until a total of 90 kg (ie 65-70 kg in this step + 20-25 kg in the first step) of 50% NaOH had been added to the reactor.
4.在最后加入50%NaOH的之后,最终的pH是9.6-9.8。4. After the final addition of 50% NaOH, the final pH is 9.6-9.8.
5.停止搅拌,将沉淀物重力沉降一整夜,然后进行水/固体分离。可选择的,在停止搅拌之后,使得沉淀物重力沉降15分钟,然后进行加速的水/固体分离。将沉淀物保持在温度低于50℃。5. Stirring was stopped, and the precipitate was gravity settled overnight, followed by water/solid separation. Optionally, after cessation of agitation, the precipitate was allowed to settle by gravity for 15 minutes, followed by accelerated water/solid separation. The precipitate was kept at a temperature below 50°C.
所形成的产率是30-50lbs的沉淀物/1000加仑反应器,并且取决于沉淀之前的Mg离子溶解和总硬度。The resulting yield is 30-50 lbs of precipitate/1000 gallon reactor and depends on Mg ion dissolution and total hardness prior to precipitation.
实施例IV.CO2吸收Example IV. CO2 Absorption
A.方法1
在该实施例中,描述了试验室规模的二氧化碳吸收。将4.00L海水进行磁搅拌,同时将100%CO2重喷通过该溶液19分钟,这里pH达到最小值4.89。在2分钟内向该溶液中加入32.00g的喷射研磨的Mg(OH)2。同时,在Mg(OH)2溶解时,连续加入CO2总共18分钟,来将pH保持在7.90到8.00。接着,在5分钟内加入100.00mL的2M NaOH,同时通过加入CO2来将pH保持在8.00到8.10。为了促进沉淀,在5分钟内加入275mL的2M NaOH,将所形成的溶液搅拌另外52分钟。将该浆体真空过滤,并且在50℃的烘箱中干燥22小时,来回收19.5g钙和镁碳酸盐(分别是初级霰石和水碳镁石)/1L的初始海水溶液。In this example, laboratory scale carbon dioxide absorption is described. 4.00 L of seawater was magnetically stirred while 100% CO2 was resprayed through the solution for 19 minutes, where the pH reached a minimum of 4.89. To this solution was added 32.00 g of jet milled Mg(OH) 2 over 2 minutes. At the same time, while the Mg(OH) 2 was dissolved, CO 2 was continuously added for a total of 18 minutes to maintain the pH at 7.90 to 8.00. Next, 100.00 mL of 2M NaOH was added over 5 minutes while maintaining the pH at 8.00 to 8.10 by adding CO 2 . To facilitate precipitation, 275 mL of 2M NaOH was added over 5 minutes and the resulting solution was stirred for an additional 52 minutes. The slurry was vacuum filtered and dried in an oven at 50° C. for 22 hours to recover 19.5 g of calcium and magnesium carbonates (primary aragonite and bischite, respectively) per 1 L of initial seawater solution.
B.方法2B. Method 2
在该实施例中,描述了试验室规模的二氧化碳吸收。将100加仑的锥形底部的塑料反应容器填充100加仑(380L)的海水,其在整个方法过程中用顶部搅拌器(便携式混合器w/轴杆,2-4”SS螺旋桨叶片(1-推,1-拉),和安装框架)进行搅拌。第一步骤是用浓缩为20%CO2和80%压缩空气的CO2喷布该溶液,流量为25scfh。通过溶液pH的稳定来确定均衡点。第二步骤是加入2.70g/L的Mg(OH)2(1.02kg),并且进行重混合。为了进一步促进Mg(OH)2的溶解,将CO2喷布通过该溶液。第三步骤是加入50wt%NaOH溶液,直到达到pH9.8,随后将喷布另外的CO2来将pH降低到8.0。重复后面这两个加入50wt%NaOH来使得pH为9.8和喷布CO2来降低pH到8.0的步骤,直到已经向溶液中加入总共16.0kg的50wt%NaOH,这里使用最后加入的NaOH来达到pH10.0。从该溶液中分离和收集沉淀物,产率为10.24g/L的碳酸钙和碳酸镁水合物。In this example, laboratory scale carbon dioxide absorption is described. A 100 gallon conical bottom plastic reaction vessel was filled with 100 gallons (380 L) of seawater which was used throughout the process with an overhead agitator (portable mixer w/shaft, 2-4" SS propeller blades (1-push , 1-pull), and mounting frame) for stirring. The first step is to spray the solution with CO concentrated to 20% CO 2 and 80% compressed air at a flow rate of 25 scfh. The equilibrium point is determined by the stabilization of the solution pH The second step is to add 2.70g/L Mg(OH) 2 (1.02kg) and remix. To further promote the dissolution of Mg(OH) 2 CO is sprayed through the solution. The third step is Add 50 wt% NaOH solution until pH 9.8 is reached, followed by sparging additional CO to lower the pH to 8.0. Repeat the latter two adding 50 wt% NaOH to bring the pH to 9.8 and sparging CO to lower the pH to 8.0 steps until a total of 16.0 kg of 50 wt% NaOH has been added to the solution, where the last added NaOH was used to achieve pH 10.0. The precipitate was separated and collected from this solution with a yield of 10.24 g/L of calcium carbonate and magnesium carbonate hydrate.
C.方法3C. Method 3
在该实施例中,描述了试验室规模的二氧化碳吸收。将100加仑的锥形底部的塑料反应容器填充100加仑(380L)的海水,其在整个方法过程中用顶部搅拌器进行搅拌。第一步骤是用浓缩为20体积%的CO2喷布该溶液,流量为100scfm(标准立方英尺/分钟)。当容器顶部空间中的CO2浓度接近于入口气体时,确定均衡点。在这个步骤中所计算的CO2吸收是可理解的低的。第二步骤是缓慢加入379g的Mg(OH)2来避免pH的急剧升高,这将有利于不期望的碳酸盐沉淀。为了进一步促进Mg(OH)2的溶解,将CO2喷布通过该溶液,使最终的pH为6.3。最后的步骤是连续捕集溶液中的CO2。在3.5小时的时间内,加入4.9kg的NaOH来将pH平衡在7.9,同时喷布和反应CO2来形成碳酸氢盐离子。在这个步骤中所计算的CO2吸收是68%-70%。结果提供在图44中,其表示了pH的变化和CO2吸收(瞬间的和累积的)。在pH图中的点1的人造现象(Artifacts)是来自于除去pH探针来加入Mg(OH)2。In this example, laboratory scale carbon dioxide absorption is described. A 100 gallon conical bottom plastic reaction vessel was filled with 100 gallons (380 L) of seawater which was stirred with an overhead stirrer throughout the process. The first step is to spray the solution with CO2 concentrated to 20% by volume at a flow rate of 100 scfm (standard cubic feet per minute). Determine the equilibrium point when the CO2 concentration in the vessel headspace is close to that of the inlet gas. The calculated CO absorption at this step is understandably low. The second step was to slowly add 379 g of Mg(OH) 2 to avoid a sharp increase in pH, which would favor undesired carbonate precipitation. To further facilitate the dissolution of Mg(OH) 2 , CO2 was sparged through the solution, resulting in a final pH of 6.3. The final step is the continuous capture of CO2 in solution. Over a period of 3.5 hours, 4.9 kg of NaOH was added to balance the pH at 7.9 while spraying and reacting CO2 to form bicarbonate ions. The calculated CO2 absorption at this step is 68%-70%. The results are presented in Figure 44, which shows the change in pH and CO2 uptake (instantaneous and cumulative). Artifacts at
D.方法4
在该实施例中,描述了工业规模的二氧化碳吸收。将1000加仑的反应容器填充900加仑(3400L)的海水,其在整个方法过程中进行搅拌。第一步骤是用3.3kg Mg(OH)2填加该溶液,这增加了pH和镁含量二者。接着,喷布10体积%的CO2,并且通过连续加入高到30kg的NaOH来将pH保持在7.9。这些步骤共持续5-6小时。最后加入38kg NaOH来提高pH,以使形成和沉淀碳酸盐。该步骤持续10-20分钟。将该溶液搅拌1小时多,来进一步沉淀。使该反应沉降一整夜。滗析该溶液,并且通过压滤或者真空过滤来回收固体产物。此外,在滗析方法之后该溶液可以冲洗;由此加入水,并将样品压滤。可选择的,水是在初始真空过滤之后加入的,搅拌和重新过滤。最后将该产物喷雾干燥。整体产率是5-7g/L的初始溶液。In this example, industrial scale carbon dioxide absorption is described. The 1000 gallon reaction vessel was filled with 900 gallons (3400 L) of seawater, which was stirred throughout the process. The first step was to top up the solution with 3.3 kg Mg(OH) 2 , which increased both the pH and the magnesium content. Next, 10% by volume of CO2 was sprayed and the pH was maintained at 7.9 by continuous addition of up to 30 kg of NaOH. These steps lasted a total of 5-6 hours. Finally 38 kg NaOH was added to raise the pH to allow carbonate formation and precipitation. This step lasts 10-20 minutes. The solution was stirred for 1 more hour to allow further precipitation. The reaction was allowed to settle overnight. The solution was decanted and the solid product was recovered by press or vacuum filtration. In addition, the solution can be rinsed after the decanting process; water is thus added, and the sample is press filtered. Optionally, water is added after the initial vacuum filtration, agitation and refiltration. Finally the product is spray dried. The overall yield was 5-7 g/L of initial solution.
实施例V.铁镁质矿物在HCl中的高产率溶解Example V. High Yield Dissolution of Mafic Minerals in HCl
在该实施例中,描述了溶解橄榄石,随后用于沉淀CO2。使用10%HCl(475.66g)溶液在50℃溶解橄榄石(10.01g,粒度~5.8μm)。在该溶液搅拌10小时,并且放置9小时来提供0.2491mol/L的Mg2+(水溶液)浓度之后,将它真空热过滤来回收404.52g的滤出液。在1小时的期间内,使用15.01g的NaOH(固体)和5.23g NaOH(水溶液)(50wt%的溶液)来中和该溶液。同时,将100%CO2重喷布通过该混合物,来提供最终的pH8.9,这里形成了沉淀物。真空过滤该浆体,并且在50℃干燥17小时来产生19.26g,其包含MgCO3·H2O,NaCl,Fe基化合物和Si基化合物。In this example, the dissolution of olivine followed by the precipitation of CO2 is described. Olivine (10.01 g, particle size ~5.8 μm) was dissolved using a 10% HCl (475.66 g) solution at 50°C. After the solution was stirred for 10 hours and left for 9 hours to provide a Mg2 + (aq) concentration of 0.2491 mol/L, it was vacuum filtered hot to recover 404.52 g of filtrate. The solution was neutralized with 15.01 g of NaOH (solid) and 5.23 g of NaOH (aq) (50 wt% solution) over a period of 1 hour. Simultaneously, 100% CO2 was re-sprayed through the mixture to provide a final pH of 8.9, where a precipitate formed. The slurry was vacuum filtered and dried at 50°C for 17 hours to yield 19.26 g containing MgCO 3 ·H 2 O, NaCl, Fe-based compounds and Si-based compounds.
实施例VI.电化学Example VI. Electrochemistry
按照本发明的双电极系统所达到的示例性结果汇总在下表8中。Exemplary results achieved with a two-electrode system according to the invention are summarized in Table 8 below.
表8.低能电化学双电极方法和系统。Table 8. Low energy electrochemical two-electrode methods and systems.
在该实施例中,描述了用于对海水进行脱质子化的电化学系统,该系统已经填充了CO2。所用的电池是由两个被钯箔分开的1L室组成。第一室用CO2填充,直到达到pH4.994。牺牲锡阳极置入该第一室中,并且将该锡电极和钯隔膜保持在100nA/cm2的恒电流控制下,其代表了0.30V的电压。第二室由锡电极和溶解在海水中的SnCl2组成。第二室中的钯隔膜和锡电极在这里保持在0.15V。该系统运行30分钟,并且如表8所示,该系统表现出第一电解质中的pH升高,和第二电解质中的pH降低。In this example, an electrochemical system for the deprotonation of seawater, which has been filled with CO 2 , is described. The cell used consisted of two 1 L compartments separated by a palladium foil. The first chamber was filled with CO2 until pH 4.994 was reached. A sacrificial tin anode was placed in the first chamber, and the tin electrode and palladium diaphragm were kept under constant current control of 100 nA/cm 2 , which represented a voltage of 0.30V. The second chamber consists of tin electrodes and SnCl2 dissolved in seawater. The palladium diaphragm and tin electrodes in the second chamber were kept at 0.15V here. The system was run for 30 minutes, and as shown in Table 8, the system exhibited an increase in pH in the first electrolyte, and a decrease in pH in the second electrolyte.
按照该离子隔膜系统所达到的示例性结果汇总在下表9中。Exemplary results achieved with this ionic membrane system are summarized in Table 9 below.
表9.低能电化学离子交换系统和方法。Table 9. Low energy electrochemical ion exchange systems and methods.
在该实施例中,描述了用于在低的运行电压,利用位于阳极和阴极之间的离子交换隔膜来生产NaOH和HCl的电化学电池。所用的电池由被阴离子交换隔膜(PC-SA-250-250(PCT GmbH of Germany))所隔开的两个250mL室组成。在两个室中使用0.5M的NaCl的18MΩ水溶液。该阳极和阴极都是由10cmx5cm,45目Pt丝网制成。阳极室具有在Pt电极下喷布的H2气体,并且将这两个电极保持在0.6V和1.0V的偏压2000秒。如表9所示,两个测试实现了在阴极室中pH的明显升高,和阳极室中pH的下降。In this example, an electrochemical cell is described for the production of NaOH and HCl at low operating voltages using an ion exchange membrane located between the anode and cathode. The cell used consisted of two 250 mL compartments separated by an anion exchange membrane (PC-SA-250-250 (PCT GmbH of Germany)). 0.5 M NaCl in 18 MΩ in water was used in both chambers. Both the anode and cathode were made of 10cmx5cm, 45 mesh Pt screen. The anode chamber had H gas sparged under the Pt electrodes, and these two electrodes were kept at a bias of 0.6 V and 1.0 V for 2000 s. As shown in Table 9, both tests achieved a significant increase in pH in the cathodic compartment, and a decrease in pH in the anodic compartment.
实施例VII.液-固分离Example VII. Liquid-Solid Separation
A.方法1
在该预言的实施例中,描述了实验室规模的从沉淀位置排出物中分离沉淀产物。沉淀产物浆体是如上所述的实施例IV来制备的。In this prophetic example, a laboratory-scale separation of precipitation products from precipitation site effluents is described. A precipitation product slurry was prepared as described above in Example IV.
包含沉淀产物的浆体是在反应容器中生产的(参见实施例IV),其在这个实施例中称作沉淀位置。在形成沉淀产物浆体之后,将该浆体作为沉淀位置排出物提供到液-固分离设备。使用沉淀位置排出物管来将该浆体提供给液-固分离设备,并且引导浆体靠着挡板流动,通过其来偏转沉淀位置排出物的流动。较重的沉淀产物粒子继续它们沿着沉淀位置排出物管(即,在重力方向上)的向下移动路径达到收集器,而上澄液偏转,与沉淀产物粒子分离,并且通过该液-固分离设备的上部离开。将所形成的沉淀产物从收集器中除去,并且干燥来产生碳酸钙和碳酸镁水合物。A slurry containing the precipitation product was produced in a reaction vessel (see Example IV), which is referred to as the precipitation site in this example. After the precipitation product slurry is formed, the slurry is provided to a liquid-solid separation device as a precipitation site discharge. The settling site effluent pipe is used to provide the slurry to the liquid-solid separation device and direct the slurry to flow against a baffle through which to deflect the flow of the settling site effluent. The heavier precipitation product particles continue their downward travel path along the settling site discharge pipe (i.e., in the direction of gravity) to the collector while the supernatant is deflected, separated from the settling product particles, and passed through the liquid-solid The upper part of the separation device leaves. The precipitated product formed is removed from the collector and dried to produce calcium carbonate and magnesium carbonate hydrate.
B.方法2B. Method 2
在该预言的实施例中,描述了实验室规模的从沉淀位置排出物中分离沉淀产物。沉淀产物浆体是如上所述的实施例IV来制备的。In this prophetic example, a laboratory-scale separation of precipitation products from precipitation site effluents is described. A precipitation product slurry was prepared as described above in Example IV.
包含沉淀产物的浆体是在反应容器中生产的(参见实施例IV),其在这个实施例中称作沉淀位置。在形成沉淀产物浆体之后,将该浆体作为沉淀位置排出物提供到液-固分离设备,其中使得该浆体在螺旋形通道中流动。在该螺旋形通道的末端,平行排列的出口收集了分离的沉淀产物粒子。将所形成的沉淀产物从收集器中除去,并且干燥来产生碳酸钙和碳酸镁水合物。A slurry containing the precipitation product was produced in a reaction vessel (see Example IV), which is referred to as the precipitation site in this example. After the precipitation product slurry is formed, the slurry is provided as a precipitation site discharge to a liquid-solid separation device, wherein the slurry is allowed to flow in a helical channel. At the end of the helical channel, parallel-arranged outlets collect the separated precipitation product particles. The precipitated product formed is removed from the collector and dried to produce calcium carbonate and magnesium carbonate hydrate.
实例1。该实例表示了在一个室中使用超声波雾化器来产生具有高的表面积体积比的液滴,其然后在另一个室内与二氧化碳接触。该实施例的系统类似于图45。Example 1. This example shows the use of an ultrasonic nebulizer in one chamber to generate droplets with a high surface area to volume ratio, which are then contacted with carbon dioxide in another chamber. The system of this embodiment is similar to FIG. 45 .
该第一系统使用了市售的超声波雾化器[图45:200],其由10个转换器组成,其每小时能够雾化大约4L的水。使用4英寸(10.16cm)的内嵌式风扇[位于220]将液滴[215]薄雾移动到混合室[245]中。该气体然后从混合室[240]再循环,并且返回到转换器室[220]中,同时纯净的CO2连续流过该系统[225]。将碳酸氢钠饱和溶液(其是用108g的干NaOH粒重新配制的)用作雾化的苛性碱源。将该溶液倾倒到包围着转换器[205]的室中。在几个时间间隔(5min,10min,20min)中对在混合室[245]中所收集的薄雾的pH进行测量。已经发现该薄雾的pH总是低于8,这里溶液的初始pH高于13.5。在使用换能器、风扇和CO2气体运行20分钟之后,碳酸氢钠和碳酸钠的沉淀物填充了该转换器室。This first system used a commercially available ultrasonic nebulizer [Figure 45: 200] consisting of 10 transducers capable of nebulizing approximately 4 L of water per hour. A 4 inch (10.16 cm) inline fan [at 220] was used to move the mist of droplets [215] into the mixing chamber [245]. This gas is then recirculated from the mixing chamber [240] and back into the converter chamber [220] while pure CO2 flows continuously through the system [225]. A saturated solution of sodium bicarbonate, reconstituted with 108 g of dry NaOH pellets, was used as the source of caustic for nebulization. This solution is poured into the chamber surrounding the converter [205]. The pH of the mist collected in the mixing chamber [245] was measured at several time intervals (5min, 10min, 20min). It was found that the pH of the mist was always below 8, where the initial pH of the solution was above 13.5. After 20 minutes of operation with the transducer, fan and CO gas, a precipitate of sodium bicarbonate and sodium carbonate filled the transducer chamber.
实例2。该实例使用了一个室来产生液滴,将该液滴与气体接触,并且沉淀固体材料。该实施例的设备类似于图46。Example 2. This example uses a chamber to generate a droplet, contact the droplet with a gas, and precipitate solid material. The apparatus of this embodiment is similar to FIG. 46 .
该设备是由于具有下面尺寸的盒子制成:6英尺(182.88cm)高,4(121.92cm)英尺宽和6英寸(15.24cm)深。这里有距离顶部2英尺(60.96cm)的搁架(其包围着市售的10转换器超声波单元[图46:300])和风扇(其使得气体以圆形样式进行循环)。该槽子的底部是圆锥形的,其允许沉淀的浆体[350]流到存储槽[360]。搁架[310]填充有2.7N的氢氧化钠溶液,并且通过再循环泵从存储槽[340]进行连续的填充。将纯净的CO2[370]泵送通过该室,并且在几个小时之后,在该室的壁上形成了碳酸氢钠和碳酸钠沉淀物。The device is made as a box with the following dimensions: 6 feet (182.88 cm) high, 4 (121.92 cm) wide and 6 inches (15.24 cm) deep. There are shelves 2 feet (60.96 cm) from the top (which surround a commercially available 10-transducer ultrasound unit [Fig. 46: 300]) and fans (which circulate the gas in a circular pattern). The bottom of the tank is conical, which allows the settled slurry [350] to flow to the storage tank [360]. The rack [310] is filled with 2.7N sodium hydroxide solution and is continuously filled from the storage tank [340] by a recirculation pump. Pure CO2 [370] was pumped through the chamber, and after several hours, sodium bicarbonate and sodium carbonate precipitates formed on the walls of the chamber.
实例3。该实例证实了使用再循环和碱性化学品溶液来吸收二氧化碳气体。Example 3. This example demonstrates the use of recirculation and alkaline chemical solutions to absorb carbon dioxide gas.
该设备使用纯净的CO2作为气体,并且是由6英寸(15.24cm)直径、1.5(45.72cm)英尺长的水平管组成。低压泵将溶液再循环到喷口,该喷口产生了这样的液滴,其的液滴尺寸远大于由超声波雾化器所产生的液滴。该管填充有2.75N的氢氧化钠(NaOH),开启泵来将氢氧化钠雾化到富含CO2的室中。在1小时和40分钟之后,所形成的溶液包含了大量的pH8.2的沉淀物,这表明碳酸氢钠和碳酸钠已经因为用CO2过饱和该溶液而发生了沉淀。The device uses pure CO2 as the gas and consists of
实例4。该实例证实了使用高的表面积体积比的自来水液滴来将二氧化碳气体混入到该液滴中,以使得该液滴在第一个与气体接触的5分钟时间内近乎用CO2完全饱和。Example 4. This example demonstrates the use of a high surface to volume tap water droplet to incorporate carbon dioxide gas into the droplet such that the droplet is nearly fully saturated with CO2 within the first 5 minutes of contact with the gas.
该设备是由8英寸(20.32cm)直径、48英寸(121.92cm)长的管组成,其具有四个沿着底部等距离隔开的超声波转换器。该吸收器是使用初始pH为7.8的普通自来水来进行测试的。在5分钟之后收集通过转换器所收集的薄雾,并且进行测试。该大体积自来水的pH是6.3,并且所收集的薄雾的pH是5.44。所收集的薄雾然后在单独的喷布系统中用更多的CO2喷布30分钟,并且已经发现能够达到的最小pH是5.39,在第一个5分钟内接近于完全饱和。The device consists of an 8 inch (20.32 cm) diameter, 48 inch (121.92 cm) long tube with four ultrasonic transducers equally spaced along the bottom. The absorber was tested using normal tap water with an initial pH of 7.8. The mist collected by the converter was collected after 5 minutes and tested. The pH of the bulk tap water was 6.3 and the pH of the collected mist was 5.44. The collected mist was then sprayed with more CO2 for 30 minutes in a separate spraying system, and it has been found that the minimum pH that can be achieved is 5.39, which is close to full saturation within the first 5 minutes.
实例5。为了处理来自于200兆瓦特的发电厂的烟道气,将它吸收到100百万加仑/天(MGD)的盐水中,其中75MGD是具有500mEq碱度的碱性盐水,25MGD是钙浓度为25000ppm的硬盐水。当使用美国煤(即,该煤不是褐煤)时,该发电厂产生了200吨/小时的二氧化碳排放。二氧化碳捕集率是90%,这表明180吨/hr的二氧化碳混入到了包含上面所列的盐水的吸收溶液中。一旦碳酸钙与盐水分离,并且干燥,并且假定连续的运行该处理装置(即,排放控制系统),则每小时形成了408.6吨的碳酸钙,其等于每天大约9800吨的产物。这表明产生了0.196磅产物/加仑盐水,其等于23.5g产物/L盐水。当降低捕集率,例如从90%降低到45%时,捕集了90吨/hr的二氧化碳,并因此形成了204.3吨/hr的碳酸钙,其等于0.098lb/加仑(11.75g/l)。当增加该发电厂的尺寸时,例如从200兆瓦增加到400兆瓦时,在90%的捕集率时,所捕集的二氧化碳的量是360吨/hr,并因此每小时形成了817.2吨的碳酸钙,这对应于大约19600吨产物/天(24小时),假定在稳态连续运行。Example 5. To treat flue gas from a 200 MW power plant, absorb it into 100 million gallons per day (MGD) of brine, where 75MGD is an alkaline brine with an alkalinity of 500mEq and 25MGD is a calcium concentration of 25,000ppm of hard salt water. When using US coal (ie, the coal is not lignite), the power plant produces 200 tons/hour of carbon dioxide emissions. The carbon dioxide capture rate was 90%, which means that 180 tons/hr of carbon dioxide was mixed into the absorption solution containing the brine listed above. Once the calcium carbonate is separated from the brine and dried, and assuming continuous operation of the treatment plant (ie, emission control system), 408.6 tons of calcium carbonate are formed per hour, which equates to approximately 9800 tons of product per day. This indicates that 0.196 lbs of product/gallon of brine was produced, which equates to 23.5 g of product/L of brine. When the capture rate is reduced, for example from 90% to 45%, 90 tons/hr of carbon dioxide is captured and thus 204.3 tons/hr of calcium carbonate is formed, which equals 0.098 lb/gal (11.75 g/l) . When increasing the size of the power plant, for example from 200 MW to 400 MWh, at 90% capture rate, the amount of carbon dioxide captured is 360 tons/hr and thus forms 817.2 tons of calcium carbonate, which corresponds to approximately 19600 tons of product/day (24 hours), assuming continuous operation at steady state.
实例6。如此处所述,已经开发了用于从发电厂烟道气中捕集二氧化碳和硫氧化物的工艺。该工艺是一种两部方法,其使用碱/高碱度材料加上钙和/或其他二价阳离子来捕集和将二氧化碳和硫氧化物转化成为固体碳酸盐和硫酸盐。这些固体然后可以转化成为终端产物,用于销售或者处置。这种方案取消了对于分离和压缩所捕集的二氧化碳来用于地质存储的需要。使用示范装置来确定从发电厂烟道气中除去二氧化碳所需的商业规模加工和能量。该示范装置从烟道气的滑流中除去二氧化碳,该滑流是通过邻近的天然气点火的组合的循环发电厂所产生的。能够在该示范装置中加工的烟道气的设计流量是大约20000标准立方英尺/分钟(“scfm”),其等价于在产生大约40兆瓦(“MW”)功率的天然气点火的组合循环发电厂中所产生的流量。这种烟道气流量等价于来自大约10MW功率的燃煤设备的烟道气流量。还在试验工厂中研究了在更小规模的煤烟道气上的吸收,目的是建立另外一个邻近于该煤点火的发电厂的示范装置,来评估存在于煤点火的烟道气中的更高浓度的二氧化碳和硫氧化物的回收和转化率。一些亮点包括:Example 6. As described here, processes have been developed for capturing carbon dioxide and sulfur oxides from power plant flue gases. The process is a two-step process that uses alkaline/overbasic materials plus calcium and/or other divalent cations to capture and convert carbon dioxide and sulfur oxides to solid carbonate and sulfate. These solids can then be converted into end products for sale or disposal. This approach eliminates the need to separate and compress captured carbon dioxide for geological storage. A demonstration plant was used to determine the commercial-scale processing and energy required to remove carbon dioxide from power plant flue gases. The demonstration plant removes carbon dioxide from a slip stream of flue gas produced by an adjacent natural gas fired combined cycle power plant. The design flow rate of flue gas capable of being processed in this demonstration plant is approximately 20,000 standard cubic feet per minute ("scfm"), which is equivalent to a natural gas fired combined cycle generating approximately 40 megawatts ("MW") of power. Flow generated in a power plant. This flue gas flow is equivalent to that from a coal-fired facility of approximately 10 MW of power. Absorption on smaller-scale coal-fired flue gases was also studied in a pilot plant with the aim of establishing another demonstration plant adjacent to the coal-fired power plant to evaluate the more Recovery and conversion of high concentrations of carbon dioxide and sulfur oxides. Some highlights include:
●在一种试验中,持续运行了典型的两小时的稳态期,使用苛性碱和氯化钙作为模拟盐水的材料,并且烟道气流量是12000scfm,这实现了最小80%的二氧化碳除去率。二氧化碳除去率是大约86%。功率消耗是521千瓦(“kW”),其等价于所产生功率的8.6%,同时产生了所加工的烟道气流量(基于由等价的煤点火的烟道气流动所产生的功率)。●In one test, running continuously for a typical two-hour steady-state period, using caustic and calcium chloride as materials to simulate brine, and a flue gas flow rate of 12,000 scfm, this achieved a minimum CO2 removal of 80% . The carbon dioxide removal rate was about 86%. Power consumption is 521 kilowatts ("kW"), which is equivalent to 8.6% of the power produced while producing the flue gas flow processed (based on the power produced by an equivalent coal-fired flue gas flow) .
●该示范装置仪器和控制机构允许获得这样的数据,该数据是量化所获得的二氧化碳的除去量和吸收器构造所需的内部功率消耗以及目的是除去二氧化碳的运行条件和功率消耗所需的。- The demonstration plant instrumentation and control mechanism allow to obtain the data required to quantify the removal of carbon dioxide obtained and the internal power consumption required for the configuration of the absorber as well as the operating conditions and power consumption for the purpose of carbon dioxide removal.
●与从初始具有4%的二氧化碳的天然气点火的燃气涡轮烟道气中除去80%的二氧化碳(降低到小于1%)相比,从初始具有15%二氧化碳的煤点火的烟道气中除去80%的二氧化碳(降低到3%的二氧化碳)应当更容易。80% CO2 removal from coal fired flue gas initially with 15% CO2 compared to 80% CO2 removal from natural gas fired gas turbine flue gas initially with 4% CO2 (down to less than 1%) % CO2 (down to 3% CO2) should be easier.
●该示范装置和支持装置在测试装置部件和运行条件方面已经设计得足够灵活,来运行选择和然后确定或者随后改进有希望的内部构造和运行条件,这最终导致产生了最佳的结果。• The demonstration plant and support plant have been designed to be flexible enough in terms of test plant components and operating conditions to run options and then determine or subsequently improve promising internal configurations and operating conditions which ultimately lead to optimal results.
●该示范装置的规模足够大,这样可以观察和联系任何的大规模特有的问题。该示范装置还具有在洗涤器液体制备领域中足够的灵活性,来允许测试打算用于商业上的盐水和碱源的综合方案。• The demonstration plant is large enough to observe and relate any large-scale specific issues. The demonstration unit also has sufficient flexibility in the area of scrubber liquid preparation to allow testing of integrated solutions for brine and alkali sources intended for commercial use.
示范装置方法说明和设计Demonstration plant method description and design
下面的是对于设计用于目前安装和运行的示范装置的方法的说明。示范装置坐落的位置包括在二十世纪四十年代所用的从海水中回收镁的设备,并且该实验位置在该位置上使用了一些现有的大的地埋桶槽容量来用于该示范装置。该方法的一大部分是吸收器柱(“吸收器”),其洗涤烟道气,通过将二氧化碳吸收到洗涤液浆中来将它从烟道气中除去。该洗涤性液体包含一种或多种二价阳离子金属例如钙,其或者是溶解的,或者是作为细微分散的悬浮固体。该洗涤性液体还包含高pH碱源例如氢氧化钠。离开该天然气点火的发电厂的烟道气具有大约175-200的华氏温度(“°F”)。将该烟道气从天然气点火的发电厂通过36英寸的未绝缘的碳钢管传输到示范装置。将两个天然气点火的发电厂烟囱接上分接头,以使得可以从任何一个烟囱或者同时从二者中获取烟道气流。该天然气点火的发电厂允许示范装置用仅仅12小时或者更低的时间来获取高到24000scfm的烟道气。这等价于在天然气点火的发电厂中产生大约4.6MW的功率所产生的烟道气。该烟道气在大约70-110°F的温度进入到吸收器中,这取决于环境温度,和烟道气通过该管的流量。烟道气管在地下从天然气点火的发电厂通向示范装置。规定在两侧(即,天然气点火的发电厂侧和示范装置侧二者)从所述管道中的冷却的烟道气中收集任何的冷凝物。将在天然气点火的发电厂侧所收集的冷凝物送回到该天然气点火发电厂。将在示范装置侧所收集的冷凝物送到用于该示范装置的淡水存储中。The following is a description of the methodology designed for the demonstration plant currently installed and operating. The site where the demonstration unit is located includes equipment used in the 1940s to recover magnesium from seawater, and the experimental site uses some of the existing large inground tank capacity at the site for the demonstration unit . A large part of the process is the absorber column ("absorber"), which scrubs the flue gas, removing carbon dioxide from the flue gas by absorbing it into a scrubbing slurry. The detergent liquid comprises one or more divalent cationic metals, such as calcium, either dissolved or as finely divided suspended solids. The scrubbing liquid also contains a high pH alkali source such as sodium hydroxide. The flue gas leaving the natural gas fired power plant has a temperature of approximately 175-200 degrees Fahrenheit ("°F"). The flue gas was transported from a natural gas fired power plant to the demonstration unit through 36 inch uninsulated carbon steel pipe. Two natural gas fired power plant stacks are tapped so that the flue gas flow can be taken from either stack or both. This natural gas fired power plant allows the demonstration plant to capture flue gas up to 24,000 scfm in as little as 12 hours or less. This is equivalent to the flue gas produced in a natural gas fired power plant producing approximately 4.6 MW of power. The flue gas enters the absorber at a temperature of about 70-110°F, depending on the ambient temperature, and the flow rate of the flue gas through the tube. The flue gas pipe runs underground from the natural gas fired power plant to the demonstration unit. Provision was made to collect any condensate from the cooled flue gas in the duct on both sides (ie, both the natural gas fired power plant side and the demonstration plant side). Condensate collected on the natural gas fired power plant side is sent back to the natural gas fired power plant. The condensate collected on the demonstration unit side was sent to fresh water storage for the demonstration unit.
该示范装置已经被设计来达到(作为商业运行的目标)80%的获自天然气点火发电厂的烟道气中的二氧化碳除去率,同时将所需的功率消耗限制到不大于通过所加工的烟道气体积的生产所代表的功率输出的8%。由于需要过量的空气,因此天然气点火的燃气涡轮例如在天然气点火发电厂中所用的这些涡轮产生了比煤点火的蒸气发电更多的基于所产生的每单位发电功率的烟道气。因此,对于煤点火的效用率发电所用的烟道气二氧化碳除去来说,基于每MW的发电所必须加工的烟道气的量典型的可以小于天然气点火的组合循环发电。因为煤包含了比天然气更高的基于每单位重量燃料的碳,并且每单位功率输出产生了较少的烟道气,因此在用于煤点火发电的吸收器入口处的二氧化碳的烟道气浓度将高于天然气点火发电厂烟道气所用的二氧化碳浓度。更高的吸收器入口二氧化碳浓度将促进任何设定百分率的二氧化碳从烟道气中的除去。The demonstration plant has been designed to achieve (as a target for commercial operation) 80% carbon dioxide removal in flue gas obtained from natural gas fired power plants, while limiting the required power consumption to no greater than The production of air volume represents 8% of the power output. Due to the excess air required, natural gas fired gas turbines such as those used in natural gas fired power plants produce more flue gas per unit of power produced than coal fired steam power. Thus, the amount of flue gas that must be processed on a per MW basis for generation of electricity for flue gas carbon dioxide removal for coal-fired utility power generation can typically be less than for natural gas-fired combined cycle power generation. Because coal contains more carbon than natural gas on a per unit weight basis of fuel and produces less flue gas per unit power output, the flue gas concentration of carbon dioxide at the inlet of an absorber for coal-fired power generation Will be higher than the CO2 concentration used in the flue gas of natural gas fired power plants. A higher absorber inlet carbon dioxide concentration will facilitate the removal of any given percentage of carbon dioxide from the flue gas.
天然气点火发电厂烟道气典型的包含3.9-4.2体积%的二氧化碳,并且基本没有硫氧化物。假定煤点火的发电厂所用的“碳浓度”是0.9公吨所产生的二氧化碳/兆瓦小时(“MWh”)的发电,并且烟道气组成具有15体积%的二氧化碳(大约6%过量的空气,并且在90°F是对水饱和的)。在计算中不考虑除去二氧化硫的需要,因此不需要从天然气点火的发电厂烟道气中除去。计算了在产生10MW的同时,理论上煤点火的发电厂所产生的烟道气的总量。该烟道气的量是20000scfm,因此该示范装置设计为加工20000scfm。当加工20000scfm时,该示范装置的功率消耗目标因此保持为低于800kW(10MW的8%)。Natural gas fired power plant flue gas typically contains 3.9-4.2 vol% carbon dioxide and is substantially free of sulfur oxides. Assume a "carbon concentration" for a coal-fired power plant of 0.9 metric tons of carbon dioxide produced per megawatt-hour ("MWh") of power generation, and a flue gas composition with 15% carbon dioxide by volume (approximately 6% excess air, and is water saturated at 90°F). The need to remove sulfur dioxide is not considered in the calculations and therefore does not need to be removed from natural gas fired power plant flue gases. The total amount of flue gas produced by a theoretical coal-fired power plant while producing 10 MW was calculated. The flue gas volume is 20000 scfm, therefore the demonstration plant is designed to process 20000 scfm. The power consumption target for this demonstration plant is thus kept below 800 kW (8% of 10 MW) when processing 20000 scfm.
使用强制通风的离心式风扇将烟道气从天然气点火的发电厂推出,并且推动它穿过示范装置。该风扇使用变频驱动(“VFD”)来控制烟道气的流量。在该风扇上游提供通风孔来在来自天然气点火的发电厂的管道上逐渐提供吸力。当在管道中形成来自天然气点火的发电厂的流动时,关闭该通风孔。从天然气点火的发电厂烟囱到该上游通风孔的管道长度是大约2400英尺。在风扇和吸收器之间是大约另外300英尺的管道,总管道长度是2718英尺。因为大部分市售的吸收器是尽可能近的连接到烟道气烟囱上的,因此风扇功率(其包括在示范装置的功率消耗中,用于将烟道气移动通过大部分的该管线)不作为其他设备的功率消耗的一部分来计算。仅仅用于该吸收器所需的功率还可以由沿着该吸收器的压力降低和烟道气流量来计算。沿着该吸收器的压力降低是通过在烟道气入口(吸收器排放到大气中)的压力计来测量的。流向吸收器和流出吸收器的烟道气是使用插入到紊流中的热丝风速计来测量的。A forced-draft centrifugal fan is used to push flue gas from a natural gas-fired power plant and push it through the demonstration unit. The fan uses a variable frequency drive ("VFD") to control the flow of flue gas. Vents are provided upstream of the fan to gradually provide suction on the ducts from the natural gas fired power plant. This vent is closed when a flow from a natural gas fired power plant develops in the pipeline. The pipe length from the natural gas fired power plant stack to the upstream vent is approximately 2400 feet. Between the fan and the absorber is approximately another 300 feet of pipe for a total pipe length of 2718 feet. Since most commercially available absorbers are connected as close as possible to the flue gas stack, the fan power (which is included in the power consumption of the demonstration unit for moving the flue gas through most of this line) Not calculated as part of the power consumption of other devices. The power required just for the absorber can also be calculated from the pressure drop and flue gas flow along the absorber. The pressure drop along the absorber was measured by a pressure gauge at the flue gas inlet (absorber discharge to atmosphere). Flue gas flow to and from the absorber was measured using a hot wire anemometer inserted into the turbulent flow.
在该示范装置中,目的是使用天然存在的硬的和碱性盐水的代表性化学品,其被用于商业的操作中。这些包括但不限于氯化钙,氢氧化钙,氢氧化钠,氯化钠,碳酸钠和硼酸钠。发电厂粉煤灰(其是阳离子和碱二者的来源)也可以用于所需的洗涤性试剂的一部分。目前,氢氧化钙和/或氯化钙被用作该二价阳离子源,氢氧化钠和/或氢氧化钙用作碱源,并且使用淡水或者海水来形成示范装置吸收器中所用的吸收性液体。氢氧化钙是粉煤灰或者水泥窑粉尘中代表性的水合氧化钙(“CaO”),并且已经被用于启动和初始运行该示范装置。氯化钙被用于模拟期望用于商业操作中的盐水中的钙硬度。它的目的是膨胀所用的化学品,来包括碳酸钠和在蓄水池表面下所发现的其他化合物。这些化学品将进行测试来确定它们的提升脱水固体(来自从该吸收器回收的纯浆体)中期望的性能来制造副产物的能力。In this demonstration plant, the aim was to use a representative chemistry of naturally occurring hard and alkaline brines, which are used in commercial operations. These include but are not limited to calcium chloride, calcium hydroxide, sodium hydroxide, sodium chloride, sodium carbonate and sodium borate. Power plant fly ash (which is a source of both cations and alkali) can also be used as part of the required scrubbing reagents. Currently, calcium hydroxide and/or calcium chloride are used as the source of this divalent cation, sodium hydroxide and/or calcium hydroxide are used as the source of alkalinity, and either fresh water or sea water are used to form the absorbent used in the absorber of the demonstration plant. liquid. Calcium hydroxide is representative of hydrated calcium oxide ("CaO") in fly ash or cement kiln dust, and has been used for start-up and initial operation of the demonstration plant. Calcium chloride was used to simulate the calcium hardness in brine expected to be used in commercial operations. Its purpose is to expand the chemicals used to include sodium carbonate and other compounds found beneath the surface of the reservoir. These chemicals will be tested to determine their ability to enhance the desired properties in the dewatered solids (from the pure slurry recovered from the absorber) to produce by-products.
为了启动和试车该示范装置,使用了氢氧化钙。将固体氢氧化钙(93.5%纯度,补足量主要是碳酸钙)通过卡车送到示范装置位置。将该氢氧化钙与淡水和/或海水在120000加仑容量的碱混合池中进行混合,该混合池是所述位置现有的室外开放式槽子。在随后的时间内设置加入上澄液(来自Epuramat固-水分离器(“Epuramat”)的水相,其在下面描述)的能力。海水是从现有的海港泵来供给的,该泵向大约1000000加仑的海水存储槽提供海水。在用于减少产生污垢的风险之前,将该存储的海水泵送通过沙子过滤器来除去微粒和有机固体。将碱混合池中的液体通过涡轮机进行再循环,其将固体氢氧化钙与该池子中的浆体进行合并。使用搅拌器和泵来保持该浆体的良好混合。然后将该浆体泵送到碱混合槽。该碱混合槽是在所述设备处的现有的室外开放式槽子,容量为140000加仑。使用搅拌器来保持该槽子中的内容物的良好混合。在该碱混合槽中,可以加入液体(目前是过滤的海水,但是最后还有上澄液)来将氢氧化钙浆体的重量含量保持为大约6重量%的氢氧化钙固体。该浆体固体含量是通过从槽子中取样,并且测量总固体来进行监控的。将该浆体从碱混合槽泵送到10000加仑容量的碱冲击槽。For start-up and commissioning of the demonstration plant, calcium hydroxide was used. Solid calcium hydroxide (93.5% purity, make up mainly calcium carbonate) was delivered by truck to the demonstration plant location. The calcium hydroxide is mixed with fresh and/or sea water in a 120,000 gallon capacity caustic mixing tank which is an existing outdoor open tank at the site. The ability to add supernatant (the aqueous phase from the Epuramat solid-water separator ("Epuramat"), which is described below) is set at a later time. Seawater is supplied from an existing harbor pump which supplies seawater to approximately 1,000,000 gallon seawater storage tanks. This stored seawater is pumped through sand filters to remove particulates and organic solids before being used to reduce the risk of fouling. The liquid in the alkali mixing tank is recirculated through a turbine which combines solid calcium hydroxide with the slurry in the tank. Agitators and pumps are used to keep the slurry well mixed. The slurry is then pumped to a caustic mix tank. The caustic mixing tank was an existing outdoor open tank at the facility with a capacity of 140,000 gallons. A stirrer was used to keep the contents of the tank well mixed. In the caustic mixing tank, liquid (currently filtered seawater, but eventually supernatant) can be added to maintain the weight content of the calcium hydroxide slurry at approximately 6% by weight calcium hydroxide solids. The slurry solids content was monitored by taking samples from the tank and measuring the total solids. The slurry was pumped from the caustic mixing tank to a 10,000 gallon capacity caustic shock tank.
当使用时,液体氯化钙溶液用卡车输送的。碱制备系统还用于该阳离子源加工中。将氯化钙溶液存储在碱混合池中。它然后在该碱混合槽中稀释。氯化钙和水同时泵送入该碱混合槽中。稀释的氯化钙然后泵送到碱冲击槽。该稀释的氯化钙泵送到浆体供料管道的集管中,用于适当的/所选择程度的吸收器。When used, the liquid calcium chloride solution is delivered by truck. Base production systems are also used in this cation source processing. Store the calcium chloride solution in an alkali mixing tank. It is then diluted in the alkali mixing tank. Calcium chloride and water are simultaneously pumped into the alkali mixing tank. The diluted calcium chloride is then pumped to the alkali shock tank. This diluted calcium chloride is pumped into the header of the slurry feed line for the appropriate/selected level of absorber.
稀氢氧化钠从两个苛性碱稀释槽中的一个供给到碱冲击槽,来提供用于吸收二氧化碳的碱源。氢氧化钠稀溶液是通过将50重量%的氢氧化钠溶液(其存储在两个10000加仑容量的槽中)与淡水在该苛性碱稀释槽中的一个中进行合并来制造的。将该碱冲击槽中的内容物通过吸收器供料泵来泵送到吸收器。稀释的氢氧化钠也可以直接泵送到六个将浆体供给到吸收器的集管中的任何一个中。Dilute sodium hydroxide is fed from one of the two caustic dilution tanks to the caustic shock tank to provide a source of alkali for carbon dioxide absorption. Dilute sodium hydroxide solution was produced by combining 50% by weight sodium hydroxide solution (stored in two 10,000 gallon capacity tanks) with fresh water in one of the caustic dilution tanks. The contents of the alkali shock tank were pumped to the absorber by the absorber feed pump. Diluted sodium hydroxide can also be pumped directly into any of the six headers that feed the slurry to the absorber.
在运行操作之间,将碱混合池中的内容物送到碱混合槽,并且将该碱混合池用淡水或者海水冲洗。将冲洗物送入T1浆体存储槽,该槽目前用于存储纯吸收器产物浆体。因为碱混合槽的内容物可以由与空气喷雾接触来吸收二氧化碳,因此在操作之间还将碱混合槽的内容物直接或者通过吸收器清空到T1浆体存储槽中。Between running operations, the contents of the caustic mixing tank are sent to the caustic mixing tank and the caustic mixing tank is flushed with fresh water or sea water. The flush is sent to the T1 slurry storage tank, which is currently used to store pure absorber product slurry. The contents of the caustic mix tank were also emptied directly or via an absorber into the T1 slurry storage tank between operations because the contents of the caustic mix tank could absorb carbon dioxide from contact with the air spray.
烟道气进入低于洗涤级,并且高于液体收集池的吸收器中。该烟道气向上流动通过吸收器,通过吸收器中的六个水平或者级(从底部水平向上顺序编号),在此新的和再循环的洗涤性液体可以注入,并且可以安装内部件(使用连接到该吸收器内径上的外缘支撑环)。洗涤性液体在重力作用下沿着吸收器从它的注入点向下流动。在向上通过洗涤级之后,该烟道气然后穿过液体去雾器(一种蒸气-液体分离器,用于通过冲击除去所携带的液体),并且流出该吸收器顶部到大气中。不需要重新加热该烟道气。在吸收器之后,允许处于吸收器出口温度的烟道气排放到环境中。对烟道气进行气体取样来测量二氧化碳浓度是在去雾器之上和即将进入吸收器之前进行的。该二氧化碳测量的常规方法是通过连续排放监控系统(“CEMS”)来进行的,该系统使用了两种不同的分析装置。ThermoEnvironmental Model60i被用作主要测量,Servomex 1440D气体分析仪用作后备和用作多方面求证。还可以手工采集气体样品用于实验室分析。The flue gas enters the absorber below the scrubbing stage and above the liquid collection tank. The flue gas flows upwards through the absorber through six levels or stages (numbered sequentially from the bottom level up) in the absorber where fresh and recycled scrubbing liquid can be injected and internals installed (using rim support ring attached to the inside diameter of the absorber). The scrubbing liquid flows down the absorber from its injection point under the force of gravity. After passing up the scrubbing stage, the flue gas then passes through a liquid demister (a vapor-liquid separator used to remove entrained liquid by impingement) and exits the top of the absorber to the atmosphere. There is no need to reheat the flue gas. After the absorber, the flue gas at the absorber outlet temperature is allowed to vent to the environment. Gas sampling of the flue gas to measure the carbon dioxide concentration is performed above the demister and immediately before entering the absorber. The conventional method for this carbon dioxide measurement is through a Continuous Emissions Monitoring System ("CEMS"), which uses two different analytical devices. ThermoEnvironmental Model60i is used as the main measurement, and Servomex 1440D gas analyzer is used as backup and for various verification. Gas samples can also be collected manually for laboratory analysis.
吸收器内部件包含处于第一水平的棚排。该棚排(倒置的角铁)沿着吸收器的直径伸出,与烟道气的流动垂直,并且连接到吸收器壳的支撑环上。每个吸收器水平具有5个集管,其垂直于烟道气流动,其可以用于连接喷口。测量沿着喷口的压力降低。将来自洗涤物的液体聚积到液体收集池中。液体从吸收器底部通过三个平行的再循环泵再循环到一个或多个级中。为了避免液体在吸收器中聚集,将净化流(它的流动受控于液体池水平)从该再循环洗涤器液体中除去,并且送入吸收器产物冲击槽。使用吸收器产物转移泵来将产物浆体移动到Epuramat容器,在这里浆体从大约5-7重量%固体到20-40重量%固体进行脱水,形成液体上澄液和稠化的浆体。该Epuramat设计来接收泵送到容器顶部的吸入流,在这里该浆体然后在重力作用下沿着中心供料管向下流动,并且经由朝向所述装置底部布置的可调整的扩散器/分离器离开进入到环向区域中。该扩散器设计来引起从紊流到层流的转变,由此将固体材料在剪切梯度下分离,形成上澄液和稠化的浆体流。The absorber inners comprise rows at a first level. The rows (inverted angle irons) protrude along the diameter of the absorber, perpendicular to the flow of the flue gas, and are connected to the support ring of the absorber shell. Each absorber level has 5 headers, perpendicular to the flue gas flow, which can be used to connect jets. Measure the pressure drop along the orifice. Accumulate liquid from laundry into liquid collection sump. Liquid is recirculated from the bottom of the absorber to one or more stages through three parallel recirculation pumps. To avoid liquid accumulation in the absorber, purge stream (whose flow is controlled at the liquid sump level) is removed from the recirculating scrubber liquid and sent to the absorber product shock tank. The absorber product transfer pump is used to move the product slurry to an Epuramat vessel where the slurry is dewatered from about 5-7 wt% solids to 20-40 wt% solids to form a liquid supernatant and a thickened slurry. The Epuramat is designed to receive a suction flow pumped to the top of the vessel, where the slurry then flows under gravity down a central feed tube and via an adjustable diffuser/separator positioned towards the bottom of the unit. The device leaves and enters the loop area. The diffuser is designed to induce a transition from turbulent to laminar flow whereby solid materials are separated under a shear gradient to form a clear liquor and a thickened slurry flow.
安装该Epuramat,并且现在进行试车。现在,将吸收器产物冲击槽中的内容物送到室外的T1浆体开放式存储槽中,该槽的容量是2500000加仑,并且被用于通过重力作用从液体中分离浆体固体。一旦Epuramat运行,则将来自该Epuramat的脱水浆体送到T1浆体存储槽,并且将液体上澄液送到上澄液冲击槽。该上澄液用泵从上澄液冲击槽送出到2500000加仑的室外T4开放式上澄液存储槽中。将该上澄液再循环到碱混合池,来降低加工水消耗和利用未反应的碱。Install the Epuramat, and now for a test run. The contents of the absorber product shock tank are now sent to an outdoor T1 slurry open storage tank which has a capacity of 2,500,000 gallons and is used to separate the slurry solids from the liquid by gravity. Once the Epuramat is running, the dewatered slurry from the Epuramat is sent to the T1 slurry storage tank and the liquid sump is sent to the sump shock tank. The supernatant is pumped from the supernatant impact tank to the outdoor T4 open supernatant storage tank of 2,500,000 gallons. This supernatant is recycled to the caustic mixing tank to reduce process water consumption and utilize unreacted caustic.
目前,为了控制在T1浆体存储槽中的量,对来自沉降的吸收器净化浆体的在所述槽顶部的上澄液的pH进行检查,并且允许排出。在该方法中的液体的再循环可以减少或者可以消除对于从该方法中排出任何液体的需要。如果pH过高,并且需要调整,则通过使得二氧化碳鼓泡通过该液体来加入二氧化碳,来使得该液体是较低碱性的(较低的pH),并且适于排放。一些聚积在Tl浆体存储槽中的固体被用于制造和评估来自位于该位置上的其他装置中的脱水固体的产物。一旦Epuramat运行,则来自示范装置的过量的上澄液可以从T4槽排放到海湾中(并且根据pH调整的需要加入二氧化碳)。Currently, to control the amount in the T1 slurry storage tank, the pH of the supernatant at the top of the tank from the settled absorber purge slurry is checked and allowed to drain. Recirculation of liquid in the process may reduce or may eliminate the need to drain any liquid from the process. If the pH is too high and needs to be adjusted, carbon dioxide is added by bubbling carbon dioxide through the liquid so that the liquid is less alkaline (lower pH) and suitable for discharge. Some of the solids accumulated in the T1 slurry storage tank were used to make and evaluate products from dewatered solids in other units located at this location. Once the Epuramat is running, excess supernatant from the demonstration unit can be discharged from the T4 tank into the bay (and carbon dioxide added as needed for pH adjustment).
除了用Epuramat(其设计来接收全部的吸收器冲洗流)测试吸收器浆体脱水之外,另外的目的是在更小的规模上用厂商提供的试验工厂来测试4-6个其他的浆体脱水系统。流向这些试验工厂的浆体是从吸收器产物冲击槽中提供的。In addition to testing absorber slurry dewatering with the Epuramat (which is designed to receive the full absorber flush flow), the additional purpose was to test 4-6 other slurries on a smaller scale with a pilot plant provided by the manufacturer dehydration system. Slurry flow to these pilot plants was provided from the absorber product impingement tank.
在脱水之后,目的是测试用于对稠化的浆体进行另外的脱水和加工的装置,来评价由该吸收器固体能够生产什么样的最终产物。After dewatering, the aim was to test the device for additional dewatering and processing of the thickened slurry to assess what end product could be produced from the absorber solids.
该示范装置中全部的主要泵都使用了VFD。液体流量是通过泵速和通过磁性流量计来测量的。向吸收器的水平4,5和6的流量是用磁性流量计来测量的。目前,这里没有安装用于水平1,2和3的磁性流量计;当需要从水平1,2或者3进行测量时,从其他水平来重新安装该其他的水平流量计。用于流向水平1-3的流量计将在接下来的几周进行安装。所测量的沿着喷口的压力降低和用于该喷口设计的理论流量也被用来通过将该理论流量与实际流量进行比较,来监控喷口堵塞。All of the main pumps in this demonstration plant used VFDs. Liquid flow is measured by pump speed and by magnetic flow meters. Flows to
为了关闭吸收器的运行(或者为了将它“冲净”),停止来自天然气点火的发电厂的烟道气的流动,停止新鲜的洗涤性溶液的流动,和停止洗涤性溶液的再循环。然后,将来自喷水存储槽的淡水通过位于薄雾消除器下面的喷口泵入吸收器顶部。该喷水也可以根据需要导入到该薄雾消除器上面的喷口中来清洁它。To shut down the operation of the absorber (or to "flush it out"), the flow of flue gas from the natural gas fired power plant is stopped, the flow of fresh scrubbing solution is stopped, and the recirculation of the scrubbing solution is stopped. Fresh water from the spray storage tank is then pumped into the top of the absorber through a spout located below the mist eliminator. The water spray can also be directed into the nozzle above the mist eliminator to clean it as desired.
示范装置设计和运行Demonstration plant design and operation
为了评估和优化二氧化碳吸收和功率消耗,提供了下面的工程设计参数:To evaluate and optimize CO2 absorption and power consumption, the following engineering design parameters are provided:
●液体注入点的位置和数目●Location and number of liquid injection points
●液体喷型和液滴尺寸范围和喷口压力降低●Liquid spray pattern and droplet size range and nozzle pressure reduction
●相对于气体流量的液体喷雾量●Liquid spray volume relative to gas flow rate
●用于混合和质量转移的内部件● Internals for mixing and mass transfer
●在液体喷雾中的气体驻留时间●Gas residence time in liquid spray
供给到该方法的碱浓度也可以分别在苛性碱稀释槽和供料液体存储槽通过加水调整。受到任何现有设计在泵送或者烟道气风扇能力方面的限制,使用和改进这些参数来进行试验的能力应当允许确定朝着实现二氧化碳除去率和功率消耗目标的“最佳的”装置排列和运行条件。在测试内部件的同时,吸收器的尺寸(直径)足以避免壁效应和足以观察(然后解决)任何的与可能发生的差的烟道气或者洗涤性液体分布有关的问题。在测试继续时手工操作一些阀门(主要用作开/关阀来建立和引导不同构造的洗涤器液体流动)来提高操作性和保持性。The alkali concentration fed to the process can also be adjusted by adding water in the caustic dilution tank and feed liquid storage tank respectively. Subject to the limitations of any existing design in terms of pumping or flue gas fan capabilities, the ability to experiment with and modify these parameters should allow determination of the "best" plant arrangement and Operating conditions. While testing the internals, the size (diameter) of the absorber was sufficient to avoid wall effects and to observe (and then resolve) any problems related to poor flue gas or scrubbing liquid distribution that might occur. Some valves (mainly used as on/off valves to establish and direct flow of scrubber fluid in different configurations) were manually operated while the test continued to improve operability and holdover.
如上所述,平行的使用两个连续的烟道气二氧化碳和氧气浓度监控装置来测量作为运行条件和装置构造的函数的吸收器二氧化碳除去率。这些仪器可以通过切换到环境大气供料来进行在线检查,并且在每天的运行开始时候用气体标准物进行校正。As mentioned above, two continuous flue gas carbon dioxide and oxygen concentration monitoring devices were used in parallel to measure absorber carbon dioxide removal as a function of operating conditions and device configuration. These instruments can be checked online by switching to ambient air supply and calibrated with gas standards at the beginning of each day's operation.
除了CEMS数据之外,其他记录数据包括但不限于pH,气体流量,液体流量,压力,温度和固体百分含量。对于气体,液体和浆体的手工取样来说,遵循用于链条控制和数据处理的方法论。In addition to CEMS data, other recorded data includes, but is not limited to, pH, gas flow, liquid flow, pressure, temperature, and percent solids. For manual sampling of gases, liquids and slurries follow the methodology for chain control and data processing.
为了监控功率消耗,监控向全部的泵和烟道气风扇的VFD驱动的功率输入。还监控了用于正式功率消耗的电计量读数。在VFD功率使用总量和电计量读数之间的差压归因于灯,搅拌器,仪表,控制系统和其他示范装置负载。大量的这些负载将倾向于在整个规模的设备上进行降低,相对于表1中所列出的主要的发动机负载。因此,电计量读数是作为保守值来使用的。另外,烟道气引风(“ID”)风扇所需的功率预期将在整个规模的设备上成比例的相对降低,假定吸收装置接近的连接到该工厂的烟道气烟囱上。To monitor power consumption, the power input to the VFD drives of all pumps and flue gas fans is monitored. Electricity meter readings for official power consumption were also monitored. The difference between the total VFD power usage and the electricity meter reading is due to lamps, agitators, meters, control systems and other demonstrative device loads. A large number of these loads will tend to be reduced across the scale of the plant relative to the major engine loads listed in Table 1. Therefore, electricity meter readings are used as conservative values. In addition, the power required for the flue gas induced draft ("ID") fan is expected to be proportional to the relative reduction in the overall size of the plant, given the close connection of the absorption unit to the flue gas stack of the plant.
表1.示范装置主要功率负载Table 1. Main power load of the demonstration device
注:负载来自2010年2月23日,运行在12000scfm,并且完全在再循环泵上流动。NOTE: Load is from 2/23/2010, running at 12000scfm, and flowing entirely on the recirculation pump.
在建立起用于运行的烟道气流量和液体流量之后,将该示范装置置于稳态运行中(在液体流动加仑/分钟或者烟道气acfm中没有波动)。稳态通常是在小于1小时内建立起来的。一旦获得稳态,正式开始数据收集。典型的,在获得数据1小时或者2小时之后,改变运行条件,以使得能够测试不同组的运行条件。包含几个运行的测试时间典型的是12-20小时。After the flue gas flow and liquid flow for operation were established, the demonstration plant was placed in steady state operation (no fluctuations in liquid flow gallons per minute or flue gas acfm). Steady state is usually established in less than 1 hour. Once steady state is obtained, data collection begins formally. Typically, 1 hour or 2 hours after the data was acquired, the operating conditions were changed so that a different set of operating conditions could be tested. Typical test time covering several runs is 12-20 hours.
测试数据Test Data
已经收集了至少两个运行周期的全部数据。这两个运行周期的差别是新鲜盐水和氢氧化钠(苛性)供料的含量(流量)。两种运行周期都满足最小80%的二氧化碳除去率。但是,具有更高含量的新鲜盐水和苛性供料的运行周期高于目标的8%的功率消耗。具有较低含量的新鲜盐水和苛性供料的运行周期接近于目标的8%的功率消耗(在所用的烟道气流量为521kW,等价于8.6%)。这个运行使用氯化钙作为阳离子材料,氢氧化钠作为碱材料,并且烟道气流量是12000scfm。该吸收器再循环泵是以全部能力来运行的。二氧化碳除去率是大约86%。仍然评价了运行参数。Full data has been collected for at least two run cycles. The difference between these two run cycles is the fresh brine and sodium hydroxide (caustic) feed content (flow rate). Both run cycles met a minimum carbon dioxide removal rate of 80%. However, the run cycle with higher content of fresh brine and caustic feed was higher than the
如果在理论上将烟道气流量升高到20000scfm的设计流量,同时保持其他具体的运行条件,则整个所用的功率将预期会稍微升高(大约44kW),这归因于所需的烟道气风扇功率的升高(大约175%以上,因为风扇功率是与烟道气流量的平方成比例的),但是功率消耗值将降低(从8.6%降低到大约5.6%),因为另外的烟道气代表了67%以上的煤点火的发电。二氧化碳除去率预期会降低,这归因于基于每单位气体降低的可利用的洗涤性液体单位。实际的二氧化碳除去率将需要试验来确定。If the flue gas flow is raised theoretically to the design flow of 20,000 scfm while maintaining other specific operating conditions, the overall power used would be expected to be slightly higher (approximately 44kW) due to the required flue The air fan power will increase (about 175% or more, because the fan power is proportional to the square of the flue gas flow), but the power consumption value will be reduced (from 8.6% to about 5.6%), because the additional flue Gas represents more than 67% of coal-fired electricity generation. The carbon dioxide removal rate is expected to decrease due to the decreased available scrubbing liquid units on a per unit gas basis. Actual carbon dioxide removal rates will need to be determined experimentally.
观察observe
已经观察到该吸收器中的泡沫化。该泡沫化通过在液体池中安装冲击板来控制。Foaming in the absorber has been observed. This foaming is controlled by installing impingement plates in the liquid pool.
当氯化钙(“CaCl2”)用作阳离子源来除去二氧化碳(“CO2”),和氢氧化钠(“NaOH”)用作高pH碱源时,氯化钠(NaCl)是与水(“H2O”)和碳酸钙(“CaCO3”)一起形成的,其的形成为:Sodium chloride (" CaCl2 ") is compatible with water when calcium chloride ("CaCl2") is used as a source of cations to remove carbon dioxide (" CO2 "), and sodium hydroxide ("NaOH") is used as a source of high pH alkalinity ("H 2 O") and calcium carbonate ("CaCO 3 "), which are formed by:
CaCl2+CO2+2NaOH-->CaCO3+2NaCl+H2OCaCl 2 +CO 2 +2NaOH-->CaCO 3 +2NaCl+H 2 O
该碳酸钙是作为固体,通过洗涤器液体(即,在Epuramat或者其他装置中)的机械脱水来除去的。为了避免来自上澄液再循环的氯化钠的积聚,必须将它从系统中净化。因为氯化钠是高溶解性的,因此它可以在机械脱水步骤中,作为带有脱水固体的残留液体,用水来除去。已经进行了考虑,向这种方法中加入执行冲洗步骤和/或通过在干燥过程中的粒度分离。已经确定了氯化钠形成了非常小的蒸发盐晶体,其可以在喷雾干燥机中从更大的粒子中除去。The calcium carbonate is removed as a solid by mechanical dewatering of the scrubber liquid (ie in an Epuramat or other device). In order to avoid the accumulation of sodium chloride from the supernatant recycle, it must be purged from the system. Because sodium chloride is highly soluble, it can be removed with water during the mechanical dehydration step as residual liquid with dehydrated solids. It has already been considered to add to this method the execution of a washing step and/or separation by particle size during the drying process. It has been determined that sodium chloride forms very small evaporated salt crystals which can be removed from the larger particles in the spray dryer.
二氧化碳除去是在低于典型的发电厂烟囱气体温度的温度进行的。二氧化碳的吸收是通过较低的温度来提高的。该较低的温度还降低了必须加工的体积气体流量。它的目的是在示范装置测试的大致的温度范围内运行市售的吸收器。它的目的是将来自发电厂烟囱气体冷却的焓用于干燥脱水固体的加工中。该加工的烟道气在一些实施方案中可以重新加热来提供另外的浮力来垂直的分散烟道气(例如,避免由于冷凝形成的地面雾)。重新加热能力可以利用现有的发电厂中的硫氧化物洗涤系统。Carbon dioxide removal is performed at temperatures below typical power plant stack gas temperatures. Carbon dioxide absorption is enhanced by lower temperatures. This lower temperature also reduces the volumetric gas flow that must be processed. It is intended to operate commercially available absorbers over the approximate temperature range tested by the demonstration unit. Its purpose is to use the enthalpy from the cooling of power plant stack gases for use in the processing of dry dewatered solids. The processed flue gas may in some embodiments be reheated to provide additional buoyancy to vertically disperse the flue gas (eg, to avoid ground fog due to condensation). The reheat capability can utilize SOx scrubber systems in existing power plants.
二氧化碳能够吸收到洗涤性液体中的速率是有限的质量转移。基于试验室和试验工厂数据,已经断定主要的阻力是沿着液滴的液体侧边界层获得二氧化碳(来自烟道气)。因此,能够吸收的二氧化碳的速率和量是与液体(或者浆体)滴的表面积成比例的。更大的表面积提供了更多的吸收路径,特别在吸收速率不变时更是如此。小滴表面积的量是小滴尺寸(越小的小滴具有每单位体积更大的表面)和小滴的数目或者所述的液体体积的函数。在实际的术语中,所用的液体量表达为gpm(浆体)/1000scfm烟道气(“L/G”比)。如果全部的其他运行参数保持不变,则更高的L/G将除去比更低的L/G更多的二氧化碳。所除去的二氧化碳的量或者百分比还可以通过提高烟道气在吸收器中的驻留时间(暴露于液滴的时间)来提高。每个这些选项具有相关的成本。增加L/G提高了泵送另外的液体浆体所需的功率。降低浆体小滴尺寸也提高了用于该方法的功率消耗,因为需要更多的能量来产生更小的小滴(和产生用于浆体的更小的固体粒子)。提高吸收器驻留时间增加了吸收器的尺寸(和成本)。测试了不同构造的驻留时间,喷雾设计,液体流量和内部件,来建立用于商业规模的吸收器的设计参数。The rate at which carbon dioxide can be absorbed into the scrubbing liquid is limited by mass transfer. Based on laboratory and pilot plant data, it has been concluded that the primary drag is the acquisition of carbon dioxide (from the flue gas) along the liquid side boundary layer of the droplet. Thus, the rate and amount of carbon dioxide that can be absorbed is proportional to the surface area of the liquid (or slurry) droplet. A larger surface area provides more paths for absorption, especially at a constant absorption rate. The amount of droplet surface area is a function of droplet size (smaller droplets have more surface per unit volume) and the number of droplets or the volume of the liquid in question. In practical terms, the amount of liquid used is expressed as gpm (slurry)/1000 scfm flue gas ("L/G" ratio). A higher L/G will remove more carbon dioxide than a lower L/G if all other operating parameters are held constant. The amount or percentage of carbon dioxide removed can also be increased by increasing the residence time (time exposed to liquid droplets) of the flue gas in the absorber. Each of these options has an associated cost. Increasing L/G increases the power required to pump additional liquid slurries. Reducing the slurry droplet size also increases the power consumption for the process, since more energy is required to produce smaller droplets (and to produce smaller solid particles for the slurry). Increasing the absorber residence time increases the size (and cost) of the absorber. Different configurations of residence time, spray design, liquid flow and internals were tested to establish design parameters for a commercial scale absorber.
二氧化碳的质量转移还取决于烟道气和液体中的二氧化碳的相对浓度。因为所吸收的二氧化碳气体在洗涤性液体中反应来形成了碳酸钙固体,因此可以假定该液体中的二氧化碳浓度总是非常低的,接近于零。因此,烟道气中更高浓度的二氧化碳将提高二氧化碳向该液体中的转移速率。因此,与从初始具有4%的二氧化碳的天然气点火的燃气涡轮烟道气中除去80%的二氧化碳(降低到小于1%)相比,从初始具有15%二氧化碳的煤点火的烟道气中除去80%的二氧化碳(降低到3%的二氧化碳)更容易。这个现象证实了这样的假定,即,应当能够以小于从煤点火的烟道气中除去80%的二氧化碳实际所消耗的功率量的功率,来从等价于高到10MW的煤点火的发电厂烟道气的天然气点火的发电厂烟道气体积中除去80%的二氧化碳。The mass transfer of carbon dioxide also depends on the relative concentrations of carbon dioxide in the flue gas and liquid. Since the absorbed carbon dioxide gas reacts in the scrubbing liquid to form calcium carbonate solids, it can be assumed that the concentration of carbon dioxide in this liquid is always very low, close to zero. Therefore, a higher concentration of carbon dioxide in the flue gas will increase the transfer rate of carbon dioxide into the liquid. Thus, removing 80% of the carbon dioxide (down to less than 1%) from a natural gas fired gas turbine flue gas initially having 4% carbon dioxide removes 80% CO2 (down to 3% CO2) is easier. This phenomenon confirms the assumption that it should be possible to generate electricity from a coal-fired power plant equivalent to as high as 10 MW with less than the amount of power actually expended to remove 80% of Flue gas Natural gas fired power plant flue gas volume removes 80% of the carbon dioxide.
已经连续进行了使用上述的煤点火的试验工厂的测试。基于在该试验工厂中的在煤烟道气上的吸收方法的多污染物质测试,该方法:Pilot plant tests using the above described coal fires have been continuously carried out. Based on multi-pollutant testing of the absorption method on coal flue gas in this pilot plant, the method:
●使用US EPA参考方法除去了大部分痕量的金属排放,达到了无检出的水平;●Using the US EPA reference method to remove most trace metal emissions, reaching the level of no detection;
●以大于(“>”)80%的除去效率来捕集汞(取决于点火的煤类型);- capture mercury with a removal efficiency greater than (">") 80% (depending on the type of coal fired);
●通过吸收器来捕集高含量的酸性气体。所观察的捕集率是>99%的SO2;>88%的SO3和>81%的HCl;和● High content of acid gas is captured by absorber. The observed capture efficiencies were >99% SO2 ; >88% SO3 and >81% HCl; and
●在来自脱水的上澄液中全部所检查的痕量元素低于水排放限定(US:NPDES)。• All trace elements examined in the supernatant from dewatering were below water discharge limits (US: NPDES).
在该方法按比例放大到煤点火的工厂上时能够实现这些相同的结果。These same results can be achieved when the method is scaled up to a coal fired plant.
虽然已经在不同的实施方案方面对本发明进行了描述,并且虽然这些实施方案已经相当详细的进行了描述,但是本发明人的目的并非限定或者以任何方式将本发明的范围限定到这样的细节。对本领域技术人员来说,很显然可以对本发明进行不同的改变和改进,而不脱离本发明的主旨和范围。前述内容仅仅是能够使用的变量的例子,并且另外的优点和改进将对于本领域技术人员来说是显而易见的。因此,本发明在它的更宽的方面因此不局限于所示和所述的具体的细节,代表性的实施方案和示例性的实施例。因此,可以不拘泥于这样的细节,而不脱离本发明的主旨或者范围。因此,应当理解此处所提出的详细说明和附图目的并非限制本发明的宽度,其应当仅仅从下面的权利要求和它们适当解释的法律等同意义来推断。While the invention has been described in terms of various embodiments, and while these embodiments have been described in considerable detail, it is not the intention of the inventors to limit or in any way limit the scope of the invention to such detail. It is obvious to those skilled in the art that various changes and improvements can be made to the present invention without departing from the spirit and scope of the present invention. The foregoing are merely examples of variables that can be used and additional advantages and modifications will readily appear to those skilled in the art. Therefore, the invention in its broader aspects is therefore not limited to the specific details, representative embodiments and exemplary examples shown and described. Therefore, departures may be made from such details without departing from the spirit or scope of the invention. Accordingly, it should be understood that the detailed description and drawings presented herein are not intended to limit the breadth of the present invention, which should be inferred only from the following claims and their legal equivalents when properly construed.
Claims (64)
Applications Claiming Priority (24)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US15899209P | 2009-03-10 | 2009-03-10 | |
| US61/158992 | 2009-03-10 | ||
| US16816609P | 2009-04-09 | 2009-04-09 | |
| US61/168166 | 2009-04-09 | ||
| US17836009P | 2009-05-14 | 2009-05-14 | |
| US17847509P | 2009-05-14 | 2009-05-14 | |
| US61/178475 | 2009-05-14 | ||
| US61/178360 | 2009-05-14 | ||
| US22145709P | 2009-06-29 | 2009-06-29 | |
| US61/221457 | 2009-06-29 | ||
| US22163109P | 2009-06-30 | 2009-06-30 | |
| US61/221631 | 2009-06-30 | ||
| US22365709P | 2009-07-07 | 2009-07-07 | |
| US61/223657 | 2009-07-07 | ||
| US22821009P | 2009-07-24 | 2009-07-24 | |
| US61/228210 | 2009-07-24 | ||
| US23004209P | 2009-07-30 | 2009-07-30 | |
| US61/230042 | 2009-07-30 | ||
| US23942909P | 2009-09-02 | 2009-09-02 | |
| US61/239429 | 2009-09-02 | ||
| US61/289657 | 2009-12-23 | ||
| US61/306412 | 2010-02-19 | ||
| US61/311275 | 2010-03-05 | ||
| PCT/US2010/026880 WO2010104989A1 (en) | 2009-03-10 | 2010-03-10 | Systems and methods for processing co2 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN101983098A true CN101983098A (en) | 2011-03-02 |
Family
ID=43619886
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN2010800009519A Pending CN101983098A (en) | 2009-03-10 | 2010-03-10 | Systems and methods for processing CO2 |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN101983098A (en) |
Cited By (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN103316582A (en) * | 2012-03-22 | 2013-09-25 | 中国科学院城市环境研究所 | Carbon fixation method through biological enzyme, and apparatus |
| CN104936674A (en) * | 2013-01-25 | 2015-09-23 | 埃克森美孚上游研究公司 | Contacting a gas stream with a liquid stream |
| CN106468648A (en) * | 2015-08-19 | 2017-03-01 | 财团法人工业技术研究院 | Micro-particle detector and method for manufacturing screening element |
| CN109943313A (en) * | 2019-04-23 | 2019-06-28 | 中国石油大学(华东) | A kind of supercritical carbon dioxide microemulsion and fly ash particle composite dispersion preparation equipment and method |
| CN110198776A (en) * | 2016-12-01 | 2019-09-03 | 外界环境公司 | Carbon dioxide capture device and method |
| CN112275237A (en) * | 2020-09-08 | 2021-01-29 | 中国科学院广州能源研究所 | A CO2 hydrate method concentration system and method |
| CN112448413A (en) * | 2020-11-16 | 2021-03-05 | 成都精智艺科技有限责任公司 | Distributed energy supply system and method with near-zero carbon emission |
| CN114713016A (en) * | 2022-04-25 | 2022-07-08 | 西安热工研究院有限公司 | A distributed energy-driven air carbon capture device and method |
| CN116196741A (en) * | 2023-04-27 | 2023-06-02 | 中国科学院西北生态环境资源研究院 | Apparatus and method for forming carbon dioxide hydrate by ball milling |
| CN118815556A (en) * | 2024-07-11 | 2024-10-22 | 华能吉林发电有限公司 | Supercritical Carbon Dioxide Isothermal Compression System |
-
2010
- 2010-03-10 CN CN2010800009519A patent/CN101983098A/en active Pending
Cited By (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN103316582A (en) * | 2012-03-22 | 2013-09-25 | 中国科学院城市环境研究所 | Carbon fixation method through biological enzyme, and apparatus |
| CN104936674A (en) * | 2013-01-25 | 2015-09-23 | 埃克森美孚上游研究公司 | Contacting a gas stream with a liquid stream |
| CN104936674B (en) * | 2013-01-25 | 2016-11-09 | 埃克森美孚上游研究公司 | Gas streams contacts with liquid stream |
| US10458893B2 (en) | 2015-08-19 | 2019-10-29 | Industrial Technology Research Institute | Miniaturized particulate matter detector and manufacturing method of a filter |
| CN106468648A (en) * | 2015-08-19 | 2017-03-01 | 财团法人工业技术研究院 | Micro-particle detector and method for manufacturing screening element |
| CN110198776A (en) * | 2016-12-01 | 2019-09-03 | 外界环境公司 | Carbon dioxide capture device and method |
| US11779878B2 (en) | 2016-12-01 | 2023-10-10 | Enviro Ambient Corporation | Carbon dioxide capture device and method |
| CN109943313A (en) * | 2019-04-23 | 2019-06-28 | 中国石油大学(华东) | A kind of supercritical carbon dioxide microemulsion and fly ash particle composite dispersion preparation equipment and method |
| CN109943313B (en) * | 2019-04-23 | 2021-03-12 | 中国石油大学(华东) | A kind of supercritical carbon dioxide microemulsion and fly ash particle composite dispersion preparation equipment and method |
| CN112275237A (en) * | 2020-09-08 | 2021-01-29 | 中国科学院广州能源研究所 | A CO2 hydrate method concentration system and method |
| CN112448413A (en) * | 2020-11-16 | 2021-03-05 | 成都精智艺科技有限责任公司 | Distributed energy supply system and method with near-zero carbon emission |
| CN114713016A (en) * | 2022-04-25 | 2022-07-08 | 西安热工研究院有限公司 | A distributed energy-driven air carbon capture device and method |
| CN116196741A (en) * | 2023-04-27 | 2023-06-02 | 中国科学院西北生态环境资源研究院 | Apparatus and method for forming carbon dioxide hydrate by ball milling |
| CN118815556A (en) * | 2024-07-11 | 2024-10-22 | 华能吉林发电有限公司 | Supercritical Carbon Dioxide Isothermal Compression System |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US8137444B2 (en) | Systems and methods for processing CO2 | |
| CN101687648B (en) | Methods of sequestering CO2 | |
| US20120211421A1 (en) | Systems and methods for processing co2 | |
| CN101983098A (en) | Systems and methods for processing CO2 | |
| US20110277670A1 (en) | Systems and methods for processing co2 | |
| CA2695006A1 (en) | Systems and methods for processing co2 | |
| CN101883736B (en) | Utilize the method and system of waste sources of metal oxides | |
| WO2010132863A1 (en) | Systems and methods for processing co2 | |
| US20120291675A1 (en) | Methods and products utilizing magnesium oxide for carbon dioxide sequestration | |
| US20120244053A1 (en) | Staged absorber system and method | |
| US20230285895A1 (en) | Gaseous CO2 Capture Systems for Improving Capture Performance, and Methods of Use Thereof | |
| US20100150802A1 (en) | Processing co2 utilizing a recirculating solution | |
| US20100239467A1 (en) | Methods and systems for utilizing waste sources of metal oxides | |
| CN101977842A (en) | Airflow multi-pollutant control system and method | |
| HK40113443A (en) | Gaseous co2 capture systems for improving capture performace, and methods of use thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C02 | Deemed withdrawal of patent application after publication (patent law 2001) | ||
| WD01 | Invention patent application deemed withdrawn after publication |
Application publication date: 20110302 |