CN101919851B - Medicinal composition for treating type 2 diabetes of mammalian and human - Google Patents
Medicinal composition for treating type 2 diabetes of mammalian and human Download PDFInfo
- Publication number
- CN101919851B CN101919851B CN2009101476090A CN200910147609A CN101919851B CN 101919851 B CN101919851 B CN 101919851B CN 2009101476090 A CN2009101476090 A CN 2009101476090A CN 200910147609 A CN200910147609 A CN 200910147609A CN 101919851 B CN101919851 B CN 101919851B
- Authority
- CN
- China
- Prior art keywords
- pharmaceutical composition
- pharmaceutically acceptable
- acceptable salt
- pioglitazone
- rosiglitazone
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D209/00—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D209/02—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom condensed with one carbocyclic ring
- C07D209/52—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom condensed with one carbocyclic ring condensed with a ring other than six-membered
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/40—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil
- A61K31/403—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil condensed with carbocyclic rings, e.g. carbazole
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
- A61K31/4427—Non condensed pyridines; Hydrogenated derivatives thereof containing further heterocyclic ring systems
- A61K31/4436—Non condensed pyridines; Hydrogenated derivatives thereof containing further heterocyclic ring systems containing a heterocyclic ring having sulfur as a ring hetero atom
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
- A61K31/4427—Non condensed pyridines; Hydrogenated derivatives thereof containing further heterocyclic ring systems
- A61K31/4439—Non condensed pyridines; Hydrogenated derivatives thereof containing further heterocyclic ring systems containing a five-membered ring with nitrogen as a ring hetero atom, e.g. omeprazole
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings
- C07D403/12—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings linked by a chain containing hetero atoms as chain links
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Diabetes (AREA)
- Epidemiology (AREA)
- Emergency Medicine (AREA)
- Endocrinology (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Hematology (AREA)
- Obesity (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
Abstract
The invention discloses a medicinal composition for treating type 2 diabetes of mammalian and human, in particular to a medicinal composition comprising (3aS,5s,6aR)-5-(2-((2S,4S)-2-cyano-group-4-fluorine-pyrrolidine-1-base)-2-oxygenethylamino group)-N,N,5-trimethyl-hexahydro cyclopentyl [C] pyrrole-2(1H)- carbamoyl or pharmaceutically acceptable salt thereof and Rosiglitazone or Pioglitazone or pharmaceutically acceptable salt thereof which are combined at fixed dose. The invention also discloses a method for preparing the medicine composition and an application of the medicinal composition for treating the type 2 diabetes of mammalian and human.
Description
Technical field
The present invention relates to treat the pharmaceutical composition that mammal comprises people's type 2 diabetes mellitus; Particularly contain rosiglitazone or pioglitazone or their pharmaceutically acceptable salts and (3aS; 5s; 6aR)-5-(2-((2S, 4S)-2-cyanic acid-4-fluoro-pyrrolidine-1-yl)-2-oxygen ethylamino)-N, N; The pharmaceutical composition of the fixed dosage combination of 5-trimethyl-six hydrogen ring penta [C] pyrroles-2 (1H)-carbamyl or its pharmaceutically acceptable salt prepares the method for said pharmaceutical composition and the purposes that said medicine composite for curing mammal comprises people's type 2 diabetes mellitus.
Background technology
People's type 2 diabetes mellitus is by relating to the chronic and gradual disease that insulin resistant and the complicated pathophysiology that slackens the dual endocrine defects of insulin secretion cause.The treatment of people's type 2 diabetes mellitus generally begins with taking exercise from diet, carries out oral antidiabetic thing monotherapy subsequently.For many patients, this scheme can not be during long-term treatment abundant blood sugar control, this just causes need carrying out conjoint therapy in the several years after diagnosis.Yet two kinds or more kinds of oral antidiabetic things altogether-prescription can cause therapeutic scheme complicated and difficulty for the many patients that use.Two kinds or more kinds of anti-diabetic reagent are merged into single preparation to be provided and can not increase the possible mode of sending conjoint therapy that the patient treats the system complexity every day.
Dipeptidyl peptidase-IV (DPP-IV) inhibitor representes that one type of exploitation is used for treating or improving the novel agent that the blood glucose of suffering from the type 2 diabetes mellitus patient generates control.Medicine being used to treat people's type 2 diabetes mellitus in the pre-Clinical has MK-0431, row spit of fland, Victor (LAF-237), saxagliptin (BMS-47718), P93/01 (Prosidon), SYR322 (Takeda), GSK823093, Roche0730699, TS021 (Taisho), E3024 (Eisai) and PHX-1149 (Phenomix).Such as, have been found that row spit of fland, Victor is administered orally to human type 2 diabetes mellitus patient can reduce empty stomach glucose relevant with significantly reduced HbAIC level and glucose deviation after meal.Can be referring to the following discloses thing about the summary of using DDP-IV treatment type 2 diabetes mellitus: people such as (1) H.-U.Demuth; " Type 2diabetes-Theraphy with dipeptidylpeptidase IV inhibitors "; People such as Biochim.Biophvs.Acta.1751:33-44 (2005) and (2) K.Augustyns; " Inhibitors of proline-specific dipeptidylpeptidases:DPP4 inhibitors as a novel approach for the treatment of Type 2diabetes "; Expert Opin.Ther.Patants, 15:1387-1407 (2005).
Some DPP-IV inhibitor are disclosed (US5462928, US5543396, WO9515309, WO2003004498, WO2003082817, WO2004032836, WO2004085661) at present; Wherein the DPP-IV inhibitor HAF-237 of Novartis company generation has shown that good DPP-IV suppresses activity and selectivity, and in listing in 2007.
LAF-237
For with the PPAR antidiabetic, particularly lattice row ketone be applied to the maturity-onset diabetes patient (body weight: 50kg), for example, every day, dosage was generally 0.01 to 1000mg, preferred 0.1 to 500mg.This dosage is used once and arrive several times every day.Especially, when pioglitazone hydrochloride was used as insulin sensitizers, every day, the dosage of pioglitazone hydrochloride normally 7.5 arrived 60mg, preferred 15 to 45mg.When rosiglitazone (perhaps maleate) was used as insulin sensitizers, every day, the dosage of rosiglitazone normally 1 arrived 12mg, preferred 2 to 12mg.
Lattice row ketone is preferably pioglitazone, pioglitazone hydrochloride or rosiglitazone (perhaps its maleate), preferred especially pioglitazone hydrochloride.
The pharmaceutical dosage form of the combination of antidiabetic drug and thiazolidine diketone derivative has been proposed in this area.For example, EP00749751 has instructed the pharmaceutical composition that comprises insulin sensitivity enhancer (it is a thiazolidinedione compound) and the combination of other antidiabetics.More particularly; It is pioglitazone that EP00749751 instructs preferred insulin sensitivity enhancer; It can make up with other antidiabetics such as rosiglitazone, phenformin or buformin, and these medicines can combine (mixing and/or coating) conventional excipients to discharge taste masked to be provided or to continue.Another instance of the combination of antidiabetic drug and thiazolidine diketone derivative is a U.S. Patent number 6; 011; 049, this patent has been instructed the slow release form, like the single medicine compositions that contains pioglitazone or troglitazone and rosiglitazone or pioglitazone of osmotic pumps or skin patch form.Other combinations of antidiabetic drug and thiazolidine diketone derivative can be seen U.S. Patent number 6,524,621; 6,475,521; 6,451,342 and 6,153,632 with PCT patent application WO01/3594, they are introduced this paper as a reference.
Summary of the invention
Have following structural formula (3aS, 5s, 6aR)-(5-trimethyl-six hydrogen ring penta [C] pyrroles-2 (1H)-carbamyl is a compd A to 5-for 2-((2S, 4S)-2-cyanic acid-4-fluoro-pyrrolidine-1-yl)-2-oxygen ethylamino)-N, N.
Compd A
The active time ratio LAF-237 length that suppresses of compd A or its salt pair DPP-IV, inhibition strength is bigger than LAF-237.Pass through experimentation; The inventor is surprised to find: the compound recipe drug effect of compd A or its salt and rosiglitazone or pioglitazone or their salt is higher than the independent medication of arbitrary composition significantly; Especially the proportioning when rosiglitazone or its salt and compd A or its salt is 1: 6.25-1: 75, and the proportioning of pioglitazone or its salt and compd A or its salt is 1: 3-1: 50 o'clock better effects if.Therefore, the compositions that compd A or its salt and rosiglitazone or its salt are formed, the compositions that compd A or its salt and pioglitazone or its salt are formed is significant clinically.
The invention provides the pharmaceutical composition of the fixed dosage combination of compd A or its pharmaceutically acceptable salt and rosiglitazone or pioglitazone or their pharmaceutically acceptable salts.It is to discharge immediately or slowly discharge that pharmaceutical composition of the present invention provides two kinds of active pharmaceutical ingredients.Pharmaceutical composition of the present invention is a tablet form, and film coated tablets particularly, also can be for example capsules etc. of other peroral dosage forms.
The dosage form that relates in one aspect to the fixed dosage combination that is used for medical science administration compd A or its pharmaceutically acceptable salt and rosiglitazone or pioglitazone or their pharmaceutically acceptable salts of the present invention.Said dosage form can be powder or solid form, and comprises tablet, capsule, pouch or the like.Concrete solid dosage forms relates to the tablet of the fixed dosage combination that contains compd A or its pharmaceutically acceptable salt and rosiglitazone or pioglitazone or its pharmaceutically acceptable salt.
The present invention also provides the method for pharmaceutical composition for preparing the fixed dosage combination of compd A or its salt and rosiglitazone or pioglitazone or their salt through drying or wet treatment method.The dry process method comprises that dry method compression and dry granulation and wet treatment method comprise wet granulation.
Another aspect of the present invention provides pharmaceutical composition of the present invention to comprise that the treatment mammal purposes of people's type 2 diabetes mellitus, this purposes comprise that the main body that needs said treatment treats the pharmaceutical composition of the present invention of effective dose.
In specific embodiments of the present invention, pharmaceutical composition comprises (1) compd A or its pharmaceutically acceptable salt, is first kind of active pharmaceutical ingredient; (2) rosiglitazone or pioglitazone or their salt are second kind of active pharmaceutical ingredient; (3) lubricant or fluidizer.In the specific embodiments of the present invention aspect this, pharmaceutical composition can also contain one or more excipient, and said excipient is selected from one or more binding agents (bonding agent); One or more diluent; One or more surfactants or wetting agent; One or more disintegrating agents; With one or more antioxidants.
The salt of compd A pharmaceutically acceptable salt, rosiglitazone or pioglitazone includes but not limited to, tosilate, phosphate, hydrochlorate, sulfate, nitrate, hydrobromate, mesylate, maleate, tartrate, succinate, acetate, trifluoroacetate, fumarate, citrate, citrate, benzene sulfonate, benzoate, naphthalene sulfonate, lactate or malate.
It is 25 milligrams~300 milligrams that the day for human beings of integrating with compd A or its salt in the pharmaceutical composition of the present invention is used dosage.It is 50 milligrams~200 milligrams that the day for human beings of preferred compound A or its salt is used dosage.It is 25,50,75,100,150,200,250 and 300 milligrams of compd As or its salt that the discrete day for human beings is used dosage.
It is 1 milligram~12 milligrams that the day for human beings of integrating with rosiglitazone or its salt in the fixed dosage of the present invention combination is used dosage; It is 1,2,4,8,10 and 12 milligram that the discrete day for human beings is used dosage; It is 7.5 milligrams~60 milligrams that the day for human beings of pioglitazone or its salt is used dosage, and it is 7.5,15,30,45 and 60 milligrams that the discrete day for human beings is used dosage.The daily dose form of these of rosiglitazone or pioglitazone or their salt is shown in the daily dosage that the approval of China and/or the U.S. is used for commercially available treatment people type 2 diabetes mellitus.
In fixed dosage combination of the present invention, compd A or its salt and rosiglitazone or pioglitazone or their the salt day for human beings are following with the specific embodiments of dosage:
Compd A or its salt (mg) 25,50,75,100,150,200,250,300
Rosiglitazone or its salt (mg) 1,2,4,8,12
Pioglitazone or its salt (mg) 7.5,15,30,45,60
Can be the compositions of any treatment effective dose of any treatment effective dose of compd A or its salt and rosiglitazone or pioglitazone or their salt, for example: 50+2,50+4,100+2; 100+4 or 50+7.5,50+15,50+30,50+45; 75+7.5,75+15,100+30,100+45 or the like.
Two kinds of active pharmaceutical ingredients in the pharmaceutical composition of the present invention can have four kinds of releasing patterns:
Composition: compd A or its salt rosiglitazone or pioglitazone or they
Salt
Discharge immediately immediately and discharge
Discharge slowly immediately and discharge
Slowly discharge immediately and discharge
Slowly discharge slowly and discharge
Pharmaceutical composition of the present invention prepares through wet method or dry process method.In one embodiment, pharmaceutical composition prepares through wet treatment method.At an apoplexy due to endogenous wind of this embodiment, pharmaceutical composition prepares through the wet type prilling process.In carrying out wet granulation, can use high shear granulation or fluid bed granulation.In one embodiment, using fluid bed granulation to have makes tablet have the more advantage of high radial strength.
Can the pharmaceutical composition that obtain through dry method or wet treatment method be compressed into tablet, encapsulate or be metered in the pouch.
Pharmaceutical composition contains one or more lubricants or fluidizer.The instance of lubricant comprises magnesium stearate, calcium stearate, stearic acid, sodium stearyl fumarate, castor oil hydrogenated or its mixture.Preferred lubricant is magnesium stearate or sodium stearyl fumarate or its mixture.The instance of fluidizer comprises silica sol, calcium phosphate, magnesium silicate and Talcum.
Optional one or more binding agents that contains of pharmaceutical composition of the present invention.The embodiment of binding agent comprises hydroxypropyl cellulose (HPC), hydroxypropyl emthylcellulose (HMPC), hydroxyethyl-cellulose, starch 1500, polyvinylpyrrolidone (polyvinyl pyrrolidone) and copolymerization alkene pyrrone.Preferred adhesive is a polyvinylpyrrolidone.
Pharmaceutical composition of the present invention can also be chosen wantonly and contain one or more diluent.The instance of diluent comprises mannitol, sorbitol, biphosphate calcium dihydrate, microcrystalline Cellulose and efflorescence cellulose.Preferable absorbent is a microcrystalline Cellulose.Microcrystalline Cellulose can be obtained from several suppliers, comprises Avicel PH 101, Avicel PH 102, Avicel PH 103, Avicel PH 105 and Avicel PH 200 that FMC Corporation makes.
Pharmaceutical composition of the present invention can also be chosen wantonly and contain disintegrating agent.Disintegrating agent can be a kind of in several modified starches, modified cellulose polymer or the polycarboxylic acids, such as crosslinked Carboxymethyl cellulose sodium, PRIMOGEL, polacrilin potassium and calcium carboxymethylcellulose (CMCCalcium).In one embodiment, disintegrating agent is a croscarmellose sodium.Croscarmellose sodium NF type A obtains with trade name " Ac-di-sol " on market.
Pharmaceutical composition of the present invention can also be chosen wantonly and contain one or more surfactants or wetting agent.Surfactant can be anion, cation or neutral surface active agent.Anion surfactant comprise sodium lauryl sulfate, dodecyl sodium sulfate, oleyl sodium sulfate and with stearate and the blended sodium laurate of Talcum.Cationic surfactant comprises benzalkonium chloride and alkyl trimethyl ammonium bromide.The neutral surface active agent comprises glycerol list olein, polyoxyethylene sorbitan fatty acid ester, polyvinyl alcohol and anhydro sorbitol fat.The embodiment of wetting agent comprises poloxamer, polyoxyethylene alkyl ether, castor oil derivatives and polyoxyethylene 8 stearate fat.
Can choose wantonly antioxidant is joined in the preparation, thereby give its chemical stability.Antioxidant is selected from the extract of alpha-tocopherol, Gamma-Tocopherol, Delta-Tocopherol, tocopherol enrichment natural origin, L-ascorbic acid and its sodium or calcium salt, anti-bad blood acyl cetylate, propyl gallate, gallate monooctyl ester, gallate dodecyl ester, Yoshinox BHT (BHT) and butylated hydroxyanisol (BHA).In one embodiment, antioxidant is BHT or BHA.
The preferred dosage form of drug composition of the present invention is the tablet through the compression method preparation.Said tablet can contain titanium dioxide and/or other coloring agent, such as ferrum oxide, dyestuff and Se Dian with filming such as the mixture of hydroxypropyl cellulose and hydroxypropyl emthylcellulose in this mixture; The mixture of polyvinyl alcohol (PVA) and Polyethylene Glycol (PEG) contains titanium dioxide and/or other coloring agent, such as ferrum oxide, dyestuff and Se Dian; Perhaps any other suitable instant-free applies agent.Coating provides taste masked and other stability to final tablet.Commercially available
for the preparation mixture of powders that film and provide for Colorcon
At last, if desired, can add sweeting agent and/or fumet.
Rosiglitazone of the present invention or pioglitazone or their salt both can be that to discharge immediately also can be slow release, and compd A or its salt can be to discharge immediately, also can be slow release.
Pharmaceutical tablet composition of the present invention can also contain one or more and other be selected from the preparation composition in the known excipient of multiple field of pharmaceutical preparations.According to performance,, can separately or unite and select kind composition arbitrarily based on they known application in the preparation tablet composition to the expectation of drug regimen.Said composition includes but not limited to diluent, compression aid, fluidizer, disintegrating agent, lubricant, spice, fumet, sweeting agent and antiseptic.
Term " tablet " intention in this use comprises all shapes and big or small compression pharmaceutical dosage formulation, no matter whether applies.The material that can be used to apply comprises hyprolose, hydroxypropyl emthylcellulose, titanium dioxide, Talcum, sweeting agent, coloring agent and fumet.
The present invention also provides the fixed dosage composition of medicine combination treatment mammal a kind of of the present invention of the main body treatment effective dose of the said treatments of administered through oral administration needs to comprise the method for people's type 2 diabetes mellitus.In one embodiment, it is human needing the main body of said treatment.In another embodiment, pharmaceutical composition is the form of tablet, also can be Capsule form.
Contain pharmaceutical composition (QD), every day twice (BID) or three (TID) administrations every day once a day of fixed dosage combination.
The synthetic method of The compounds of this invention A
(3aS; 5s; 6aR)-5-(2-((2S, 4S)-2-cyanic acid-4-fluoro-pyrrolidine-1-yl)-2-oxygen ethylamino)-N, N; The synthetic method of 5-trimethyl-six hydrogen ring penta [C] pyrroles-2 (1H)-carbamyl (compd A) is according to the described methods preparation of PCT/CN2008/071014 embodiment 18, therefore with this disclosure document as a reference.
The specific embodiment
Following examples have further described and have explained embodiment within the scope of the present invention.Embodiment and Test Example only are that for the purpose of illustration purpose provides, and are not intended it is regarded as limitation of the present invention, and possibly there is the multiple variant that does not deviate from spirit and scope of the invention in it.
Embodiment 1, compd A tosilate and rosiglitazone maleate compound tablet
Write out a prescription every and contain:
Compd A tosilate 50mg
Rosiglitazone maleate 4mg
Microcrystalline Cellulose 10mg
2% starch slurry is an amount of
Magnesium stearate 0.5mg
Method for preparing: with the compd A tosilate, rosiglitazone maleate, the microcrystalline Cellulose mixing is with 2% starch slurry wet granulation.Drying adds magnesium stearate, the mixing tabletting.
Similarly; Through regulating compd A or its salt in the compound recipe; The consumption of rosiglitazone or its salt prepares the compound recipe of two kinds of active component that contain different proportion, and for example the ratio of rosiglitazone or its salt and compd A or its salt is 1: 6.25,1: 12.5,1: 25,1: 50,1: 75 or the like.
Embodiment 2, compd A tosilate and pioglitazone hydrochloride compound tablet
Write out a prescription every and contain:
Compd A tosilate 50mg
Pioglitazone hydrochloride 15mg
Microcrystalline Cellulose 10mg
2% starch slurry is an amount of
Magnesium stearate 0.5mg
Method for preparing: with the compd A tosilate, pioglitazone hydrochloride, the microcrystalline Cellulose mixing is with 2% starch slurry wet granulation.Drying adds magnesium stearate, the mixing tabletting.
Similarly, through regulating compd A or its salt in the compound recipe, the consumption of pioglitazone or its salt; The compound recipe for preparing two kinds of active component that contain different proportion, for example the ratio of pioglitazone or its salt and compd A or its salt is 7.5: 25,7.5: 50; 7.5: 75; 7.5: 100,7.5: 150,7.5: 375 or the like.
Test Example 1: the external activity of compd A, LAF-237 and selectivity research
Method:
Before using, the DPP-IV-Glo. that thaws cushions and equilibrates to room temperature; The frozen fluorescein detectable of buffering suspension DPP-IV-Glo. adds the slight mix homogeneously of ultra-pure water in substrate after, is processed the substrate of 1mM before using; The fluorescein detectable is put into amber bottle, add DPP-IV-Glo..The fluorescein detectable should be dissolved in 1 minute; With the DMSO dissolving institute chemical compound of surveying 50 times to final operation concentration, in each test tube 50 times of concentration of adding the chemical compound of surveying 2 μ L, adding 2 μ LDMSO in negative control and blank; In each test tube, add 46 μ L Tris buffer; In blank, add 48 μ L Tris buffer, in each test tube of negative control and test specimens, add 2 μ LDPP4 enzymes, vibration mixes and centrifuge tube.Material in the test tube is all transferred on the flat board of 96-hole, and mixing substrate and DPP-IV-Glo. ratio is 1: 49.Vibration is mixed to abundant mixing.At room temperature left standstill before the use 30-60 minute, and in each 96-hole plate well, added the mixed liquor of 50 μ L DPP-IV-Glo. and substrate, seal flat board, under 300-500rpm/30s, slowly mix material in 96 holes with oscillator plate with the envelope film.At room temperature cultivated 30 minutes to 3 hours, and detected the chemiluminescence count value at the multi-functional ELIASA of NOVOstar.
[table 1]
The result: compd A is superior to control drug LAF-237 to the inhibition activity of DPP4, and selectivity also is better than LAF-237.
Test Example 2: different proportion rosiglitazone and compd A pharmaceutical composition are to normal ICR mice
The research of oral glucose tolerance
Male ICR mouse is in fasting oral distilled water, different proportioning (rosiglitazone maleate: compd A tosilate=1: 6.25 after 6 hours; 1: 12.5; 1: 25,1: 50,1: 75) pharmaceutical composition of same dose (10mg/kg); Each is organized in administration oral glucose 2.5g/kg in the time of 30 minutes, does the oral glucose tolerance test.In getting blood in the time of 0,30,60,120 minute for the sugar back, measure serum level of glucose.
The serum glucose assay method;
Adopt glucose kit to measure the glucose content in the serum; Get 250 μ l enzyme working solutions, add 5 μ l serum, set up blank pipe (adding 5 μ l distilled waters) and standard pipe (adding 5 μ l glucoses mark liquid) simultaneously; Mixing; 37 ℃ of water-baths 20 minutes are with the zeroing of blank pipe, the colorimetric determination of OD505nm place.
Serum glucose level BG (nmol/l)=OD
Sample cell/ OD
Standard pipe* 5.55
Date processing and statistical analysis;
1, adopt mean SD and Student-t test that data are carried out statistical analysis
2, calculate blood glucose decline percentage rate and TG-AUC AUC when giving sugared back 30 minutes
[table 2]: the back influence to oral carbohydrate tolerance of normal ICR mice oral test pharmaceutical composition (10mg/kg) (mean SD, n=6)
*, P<0.05; Compare with the blank group;
*, P<0.01; Compare with the blank group;
* *, P<0.001; Compare with the blank group;
Test Example 3: different proportion pioglitazone and compd A pharmaceutical composition are to normal ICR mice
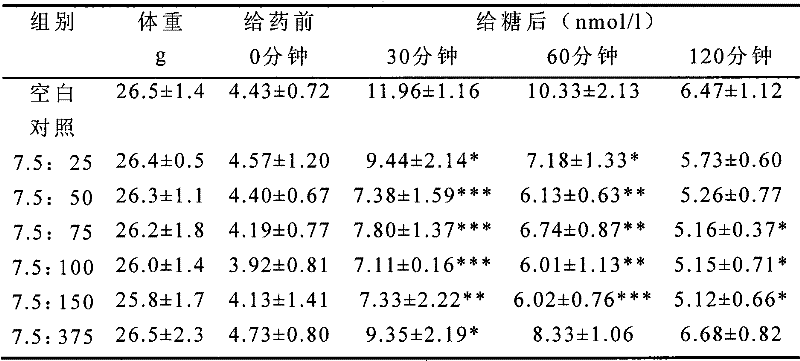
The research of oral glucose tolerance
Male ICR mouse is in fasting oral distilled water, different proportioning (pioglitazone hydrochlorate: compd A tosilate=7.5: 25 after 6 hours; 7.5: 50,7.5: 75,7.5: 100; 7.5: 150; 7.5: the 375) mixture of same dose (30mg/kg), each is organized in administration oral glucose 2.5g/kg in the time of 30 minutes, does the oral glucose tolerance test.In getting blood in the time of 0,30,60,120 minute for the sugar back, measure serum level of glucose.
The serum glucose assay method;
Adopt glucose kit to measure the glucose content in the serum; Get 250 μ l enzyme working solutions, add 5 μ l serum, set up blank pipe (adding 5 μ l distilled waters) and standard pipe (adding 5 μ l glucoses mark liquid) simultaneously; Mixing; 37 ℃ of water-baths 20 minutes are with the zeroing of blank pipe, the colorimetric determination of OD505nm place.
Serum glucose level BG (nmol/l)=OD sample cell/OD standard pipe * 5.55
Date processing and statistical analysis;
1, adopt mean SD and Student-t test that data are carried out statistical analysis
2, calculate blood glucose decline percentage rate and TG-AUC AUC when giving sugared back 30 minutes
[table 3]: the back influence to oral carbohydrate tolerance of normal ICR mice oral test compound recipe thing (30mg/kg) (mean SD, n=6)
*, P<0.05; Compare with the blank group;
*, P<0.01; Compare with the blank group;
* *, P<0.001; Compare with the blank group;
Test Example 4: compd A tosilate, LAF-237 respectively with the rosiglitazone maleic acid
Wistar fertilizer fat in heritability and that suffer from diabetes is taken in uniting of salt or pioglitazone hydrochlorate
Effect in the Mus
The fertile Mus of the male Wistar in 14~19 ages in week is divided into 8 groups; Every group 5~6, took distilled water, compd A tosilate, rosiglitazone maleate, pioglitazone hydrochlorate, LAF-237+ rosiglitazone maleate, LAF-237+ pioglitazone hydrochlorate, compd A tosilate+rosiglitazone maleate and compd A tosilate+pioglitazone hydrochlorate respectively 14 days.Get blood from the tail vein; Use a kind of commercially available reagent box (NC-ROPET; Nippon Chemiphar CO.) measuring plasma glucose and HbA1 respectively with enzyme process. the result is expressed as the meansigma methods ± standard deviation of every group (n=5-6) and with Dunnett ' s check analysis, in table 4, provides.Use 1% significance level.
[table 4]
| Dosage | Plasma glucose | Hemoglobin | |
| Matched group | 356±31 | 5.9±0.5 | |
| The compd A tosilate | 10mg/kg | 224±52 * | 5.9±0.3 |
| The rosiglitazone maleate | 2.5mg/kg | 327±45 | 6.2±0.6 |
| The pioglitazone hydrochlorate | 5mg/kg | 298±27 | 5.8±0.2 |
| LAF-237 | 10mg/kg | 284±31 | 6.1±0.4 |
| LAF-237+ rosiglitazone maleate | 10mg/kg+2.5mg/kg | 175±13 * | 4.7±0.5 * |
| LAF-237+ pioglitazone hydrochlorate | 10mg/kg+5mg/kg | 168±7 * | 4.6±0.5 * |
| Compd A tosilate+rosiglitazone maleate | 10mg/kg+2.5mg/kg | 125±21 * | 4.3±0.4 * |
| Compd A tosilate+pioglitazone hydrochlorate | 10mg/kg+5mg/kg | 112±17 * | 4.2±0.3 * |
*: P<0.01 is compared with matched group
The compd A tosilate is united with rosiglitazone maleate or pioglitazone hydrochlorate and is taken the concentration that has clearly reduced blood-glucose and hemoglobin in the table 4; Its intensity is individually dosed greater than compd A tosilate, rosiglitazone maleate, pioglitazone hydrochlorate, LAF-237's, and its intensity is greater than LAF-237 and rosiglitazone maleate or pioglitazone hydrochlorate drug combination.
Claims (11)
1. pharmaceutical composition of treating the mammal type 2 diabetes mellitus, said pharmaceutical composition contain rosiglitazone or pioglitazone or their pharmaceutically acceptable salts and (3aS, 5s of treating effective dose; 6aR)-5-(2-((2S, 4S)-2-cyanic acid-4-fluoro-pyrrolidine-1-yl)-2-oxygen ethylamino)-N, N; 5-trimethyl-six hydrogen ring penta [C] pyrroles-2 (1H)-carbamyl or its pharmaceutically acceptable salt, wherein rosiglitazone or its pharmaceutically acceptable salt and (3aS, 5s; 6aR)-5-(2-((2S, 4S)-2-cyanic acid-4-fluoro-pyrrolidine-1-yl)-2-oxygen ethylamino)-N, N; The weight ratio of 5-trimethyl-six hydrogen ring penta [C] pyrroles-2 (1H)-carbamyl or its pharmaceutically acceptable salt is 1: 6.25-1: 75, and pioglitazone or its pharmaceutically acceptable salt and (3aS, 5s; 6aR)-5-(2-((2S; 4S)-2-cyanic acid-4-fluoro-pyrrolidine-1-yl)-2-oxygen ethylamino)-N, N, the weight ratio of 5-trimethyl-six hydrogen ring penta [C] pyrroles-2 (1H)-carbamyl or its pharmaceutically acceptable salt is 1: 3-1: 50.
2. pharmaceutical composition according to claim 1, wherein said mammal is the people.
3. pharmaceutical composition according to claim 2; Wherein said (3aS; 5s, 6aR)-5-(2-((2S, 4S)-2-cyanic acid-4-fluoro-pyrrolidine-1-yl)-2-oxygen ethylamino)-N; N, people's consumption per day of 5-trimethyl-six hydrogen ring penta [C] pyrroles-2 (1H)-carbamyl or its pharmaceutically acceptable salt is 25-300mg.
4. pharmaceutical composition according to claim 2, people's consumption per day of wherein said rosiglitazone or its pharmaceutically acceptable salt is 1-12mg, people's consumption per day of said pioglitazone or its pharmaceutically acceptable salt is 7.5-60mg.
5. according to each described pharmaceutical composition of claim 1-4, wherein said pharmaceutical composition is tablet, hard capsule, soft capsule, oral solution, slow releasing agent, drop pill, electuary, granule or other peroral dosage forms.
6. according to each described pharmaceutical composition of claim 1-4; Wherein said (3aS; 5s, 6aR)-5-(2-((2S, 4S)-2-cyanic acid-4-fluoro-pyrrolidine-1-yl)-2-oxygen ethylamino)-N; N, 5-trimethyl-six hydrogen ring penta [C] pyrroles-2 (1H)-carbamyl or its pharmaceutically acceptable salt discharge immediately or slowly discharge.
7. according to each described pharmaceutical composition of claim 1-4, wherein said rosiglitazone or pioglitazone or their pharmaceutically acceptable salts are to discharge immediately or slowly discharge.
8. according to each described pharmaceutical composition of claim 1-4, wherein said pharmaceutical composition be with once-a-day, the Pharmaceutical composition of twice on the one or three administrations on the one.
9. according to each described pharmaceutical composition of claim 1-4, wherein said pharmaceutically acceptable salt is selected from tosilate, phosphate, hydrochlorate, sulfate, nitrate, hydrobromate, mesylate, maleate, tartrate, succinate, acetate, trifluoroacetate, fumarate, citrate, citrate, benzene sulfonate, benzoate, naphthalene sulfonate, lactate or malate.
10. like the purposes of each described pharmaceutical composition of claim 1-9 in the medicine of preparation treatment mammal diabetes.
11. purposes according to claim 10, wherein said mammal is the people.
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2009101476090A CN101919851B (en) | 2009-06-09 | 2009-06-09 | Medicinal composition for treating type 2 diabetes of mammalian and human |
| PCT/CN2010/072709 WO2010142182A1 (en) | 2009-06-09 | 2010-05-13 | Pharmaceutical composition for treatment of type 2 diabetes in mammals including human beings |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2009101476090A CN101919851B (en) | 2009-06-09 | 2009-06-09 | Medicinal composition for treating type 2 diabetes of mammalian and human |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN101919851A CN101919851A (en) | 2010-12-22 |
| CN101919851B true CN101919851B (en) | 2012-02-08 |
Family
ID=43308408
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN2009101476090A Expired - Fee Related CN101919851B (en) | 2009-06-09 | 2009-06-09 | Medicinal composition for treating type 2 diabetes of mammalian and human |
Country Status (2)
| Country | Link |
|---|---|
| CN (1) | CN101919851B (en) |
| WO (1) | WO2010142182A1 (en) |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102552140B (en) * | 2011-01-12 | 2013-05-29 | 北京人福军威医药技术开发有限公司 | Liquid composition of rosiglitazone |
| CN104030967B (en) * | 2013-03-05 | 2018-01-02 | 连云港恒运药业有限公司 | A kind of intermediate of DPP IV inhibitor, its preparation method and the method for DPP IV inhibitor is prepared by it |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101230058A (en) * | 2007-01-23 | 2008-07-30 | 上海恒瑞医药有限公司 | Bicycle aza alkyl derivative, preparation method and use in medicine thereof |
| CN101365432A (en) * | 2005-12-16 | 2009-02-11 | 默克公司 | Pharmaceutical composition of dipeptidyl peptidase-4 inhibitor in combination with metformin |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101230059B (en) * | 2007-01-23 | 2011-08-17 | 上海恒瑞医药有限公司 | Bicyclic aza-alkane derivatives, their preparation method and their use in medicine |
-
2009
- 2009-06-09 CN CN2009101476090A patent/CN101919851B/en not_active Expired - Fee Related
-
2010
- 2010-05-13 WO PCT/CN2010/072709 patent/WO2010142182A1/en not_active Ceased
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101365432A (en) * | 2005-12-16 | 2009-02-11 | 默克公司 | Pharmaceutical composition of dipeptidyl peptidase-4 inhibitor in combination with metformin |
| CN101230058A (en) * | 2007-01-23 | 2008-07-30 | 上海恒瑞医药有限公司 | Bicycle aza alkyl derivative, preparation method and use in medicine thereof |
Non-Patent Citations (1)
| Title |
|---|
| 李秀丽.2型糖尿病的治疗药物研究进展.《赤峰学院学报(自然科学版)》.2008,第24卷(第1期),第123-125页. * |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2010142182A1 (en) | 2010-12-16 |
| CN101919851A (en) | 2010-12-22 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5779566B2 (en) | Pharmaceutical composition comprising a combination of a dipeptidyl peptidase-4 inhibitor and metformin | |
| US20120059011A1 (en) | Pharmaceutical compositions of combinations of dipeptidyl peptidase-4 inhibitors with pioglitazone | |
| CN106924208A (en) | A kind of compound Dapagliflozin Metformin Extended-release Tablets and preparation method thereof | |
| CN106075451A (en) | Use SGLT2 inhibitor and combinations thereof thing that the treatment previously carried out with other antidiabetic drug is had in the patient of tolerance the method treating type ii diabetes | |
| CN101932241A (en) | Combination pharmaceutical composition of metformin and dipeptidyl peptidase-IV inhibitor | |
| CN107432869A (en) | Include net double-layer tablets of Metformin hydrochloride and En Gelie and preparation method thereof | |
| CN101919851B (en) | Medicinal composition for treating type 2 diabetes of mammalian and human | |
| CN101904840B (en) | Medicinal composition for treating type 2 diabetes of mammal including human | |
| US8476272B2 (en) | Pharmaceutical composition for treatment of type 2 diabetes | |
| CN101961336B (en) | Pharmaceutical composition for treating type 2 diabetes in mammals including human | |
| RU2533560C2 (en) | Pharmaceutical composition for treating type 2 diabetes | |
| CN109288836B (en) | Compound dihydralazine sulfate preparation as well as preparation method and application thereof | |
| TWI494313B (en) | Pharmaceutical composition for the treatment of type 2 diabetes in mammals including human beings | |
| CN106074553A (en) | Containing sitagliptin and the pharmaceutical composition of metformin | |
| CN104434856A (en) | Gastric floating glipizide controlled release tablet and preparation process thereof | |
| TWI484955B (en) | Pharmaceutical composition for the treatment of type 2 diabetes in mammals including human beings | |
| TWI462925B (en) | Pharmaceutical composition for the treatment of type 2 diabetes | |
| HK1147701A (en) | Pharmaceutical composition for the treatment of type 2 diabetes in mammals including man | |
| CN100455288C (en) | Formula of Reynoldazine hydrochloride prepn. | |
| HK1147700B (en) | Pharmaceutical composition for the treatment of type 2 diabetes in mammals including man | |
| TW201219384A (en) | Pharmaceutical composition for the treatment of type 2 diabetes in mammals including human beings | |
| HK1153143B (en) | Pharmaceutical composition for the treatment of type 2 diabetes in mammals including human beings |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| REG | Reference to a national code |
Ref country code: HK Ref legal event code: DE Ref document number: 1147701 Country of ref document: HK |
|
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| C17 | Cessation of patent right | ||
| CF01 | Termination of patent right due to non-payment of annual fee |
Granted publication date: 20120208 Termination date: 20120609 |
|
| REG | Reference to a national code |
Ref country code: HK Ref legal event code: WD Ref document number: 1147701 Country of ref document: HK |