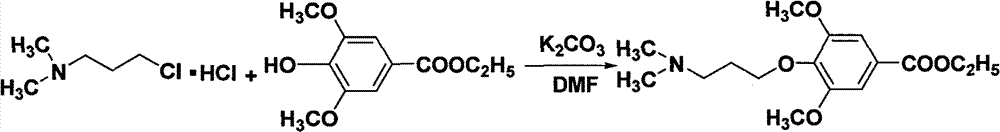CN101811974B - 4-(3-(dimethoxy) propoxy)-3, 5-dimethoxy benzoic acid, and preparation method and medical purpose thereof - Google Patents
4-(3-(dimethoxy) propoxy)-3, 5-dimethoxy benzoic acid, and preparation method and medical purpose thereof Download PDFInfo
- Publication number
- CN101811974B CN101811974B CN 201010151142 CN201010151142A CN101811974B CN 101811974 B CN101811974 B CN 101811974B CN 201010151142 CN201010151142 CN 201010151142 CN 201010151142 A CN201010151142 A CN 201010151142A CN 101811974 B CN101811974 B CN 101811974B
- Authority
- CN
- China
- Prior art keywords
- compound
- salt
- dimethoxy
- propoxy
- pharmacy acceptable
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 238000002360 preparation method Methods 0.000 title claims abstract description 6
- 150000001875 compounds Chemical class 0.000 claims abstract description 23
- 208000026106 cerebrovascular disease Diseases 0.000 claims abstract description 11
- 208000010110 spontaneous platelet aggregation Diseases 0.000 claims abstract description 10
- 208000024172 Cardiovascular disease Diseases 0.000 claims abstract description 9
- 230000002526 effect on cardiovascular system Effects 0.000 claims abstract description 8
- 150000003839 salts Chemical class 0.000 claims description 11
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 claims description 10
- 239000003814 drug Substances 0.000 claims description 5
- 239000008194 pharmaceutical composition Substances 0.000 claims description 4
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 claims description 4
- 208000006011 Stroke Diseases 0.000 claims description 3
- 239000003937 drug carrier Substances 0.000 claims description 3
- 208000010125 myocardial infarction Diseases 0.000 claims description 3
- BMFMQGXDDJALKQ-BYPYZUCNSA-N Argininic acid Chemical class NC(N)=NCCC[C@H](O)C(O)=O BMFMQGXDDJALKQ-BYPYZUCNSA-N 0.000 claims description 2
- KRKNYBCHXYNGOX-UHFFFAOYSA-K Citrate Chemical compound [O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O KRKNYBCHXYNGOX-UHFFFAOYSA-K 0.000 claims description 2
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 claims description 2
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 claims description 2
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 claims description 2
- WCUXLLCKKVVCTQ-UHFFFAOYSA-M Potassium chloride Chemical compound [Cl-].[K+] WCUXLLCKKVVCTQ-UHFFFAOYSA-M 0.000 claims description 2
- 159000000007 calcium salts Chemical class 0.000 claims description 2
- 159000000003 magnesium salts Chemical class 0.000 claims description 2
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 claims description 2
- 208000031225 myocardial ischemia Diseases 0.000 claims description 2
- 150000003016 phosphoric acids Chemical class 0.000 claims description 2
- 239000001103 potassium chloride Substances 0.000 claims description 2
- 235000011164 potassium chloride Nutrition 0.000 claims description 2
- 159000000000 sodium salts Chemical group 0.000 claims description 2
- 229940095064 tartrate Drugs 0.000 claims description 2
- 229950004288 tosilate Drugs 0.000 claims description 2
- 238000012360 testing method Methods 0.000 abstract description 4
- 230000000694 effects Effects 0.000 abstract description 3
- 230000000144 pharmacologic effect Effects 0.000 abstract description 2
- SHZKQBHERIJWAO-AATRIKPKSA-N ozagrel Chemical compound C1=CC(/C=C/C(=O)O)=CC=C1CN1C=NC=C1 SHZKQBHERIJWAO-AATRIKPKSA-N 0.000 description 12
- 229950003837 ozagrel Drugs 0.000 description 12
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 11
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 10
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 9
- 238000006243 chemical reaction Methods 0.000 description 8
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 6
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 6
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 6
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 5
- 239000003795 chemical substances by application Substances 0.000 description 5
- 201000010099 disease Diseases 0.000 description 5
- 239000007788 liquid Substances 0.000 description 5
- 241000283973 Oryctolagus cuniculus Species 0.000 description 4
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 4
- 210000001772 blood platelet Anatomy 0.000 description 4
- 230000002401 inhibitory effect Effects 0.000 description 4
- 239000011780 sodium chloride Substances 0.000 description 4
- 239000002904 solvent Substances 0.000 description 4
- 239000002775 capsule Substances 0.000 description 3
- 206010008118 cerebral infarction Diseases 0.000 description 3
- 230000006837 decompression Effects 0.000 description 3
- 229940079593 drug Drugs 0.000 description 3
- 238000001035 drying Methods 0.000 description 3
- 238000000034 method Methods 0.000 description 3
- 238000011084 recovery Methods 0.000 description 3
- FCMCDVRJVMDKAQ-UHFFFAOYSA-N 1-chloro-n,n-dimethylpropan-1-amine;hydrochloride Chemical compound Cl.CCC(Cl)N(C)C FCMCDVRJVMDKAQ-UHFFFAOYSA-N 0.000 description 2
- 238000001644 13C nuclear magnetic resonance spectroscopy Methods 0.000 description 2
- 238000005160 1H NMR spectroscopy Methods 0.000 description 2
- 206010008132 Cerebral thrombosis Diseases 0.000 description 2
- 201000001429 Intracranial Thrombosis Diseases 0.000 description 2
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 2
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 2
- 208000007536 Thrombosis Diseases 0.000 description 2
- 230000001154 acute effect Effects 0.000 description 2
- 238000001816 cooling Methods 0.000 description 2
- 208000029078 coronary artery disease Diseases 0.000 description 2
- XQGZSYKGWHUSDH-UHFFFAOYSA-N dazoxiben Chemical compound C1=CC(C(=O)O)=CC=C1OCCN1C=NC=C1 XQGZSYKGWHUSDH-UHFFFAOYSA-N 0.000 description 2
- 229950008000 dazoxiben Drugs 0.000 description 2
- 230000008021 deposition Effects 0.000 description 2
- 238000001514 detection method Methods 0.000 description 2
- 238000005516 engineering process Methods 0.000 description 2
- 235000019441 ethanol Nutrition 0.000 description 2
- WKUVKFZZCHINKG-UHFFFAOYSA-N ethyl 4-hydroxy-3,5-dimethoxybenzoate Chemical compound CCOC(=O)C1=CC(OC)=C(O)C(OC)=C1 WKUVKFZZCHINKG-UHFFFAOYSA-N 0.000 description 2
- 239000012065 filter cake Substances 0.000 description 2
- 238000010438 heat treatment Methods 0.000 description 2
- 238000006460 hydrolysis reaction Methods 0.000 description 2
- 230000000302 ischemic effect Effects 0.000 description 2
- 210000002381 plasma Anatomy 0.000 description 2
- 210000004623 platelet-rich plasma Anatomy 0.000 description 2
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Substances [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 2
- 235000015320 potassium carbonate Nutrition 0.000 description 2
- 238000001953 recrystallisation Methods 0.000 description 2
- 229910002027 silica gel Inorganic materials 0.000 description 2
- 239000000741 silica gel Substances 0.000 description 2
- 229960001866 silicon dioxide Drugs 0.000 description 2
- 239000000243 solution Substances 0.000 description 2
- 238000000967 suction filtration Methods 0.000 description 2
- -1 vitriol Chemical class 0.000 description 2
- 238000005406 washing Methods 0.000 description 2
- 238000005303 weighing Methods 0.000 description 2
- 0 CN(C)CCCOc(c(OC)c1)c(*)cc1C(O)=O Chemical compound CN(C)CCCOc(c(OC)c1)c(*)cc1C(O)=O 0.000 description 1
- 206010008190 Cerebrovascular accident Diseases 0.000 description 1
- 206010011091 Coronary artery thrombosis Diseases 0.000 description 1
- 208000005189 Embolism Diseases 0.000 description 1
- 208000005764 Peripheral Arterial Disease Diseases 0.000 description 1
- 208000030831 Peripheral arterial occlusive disease Diseases 0.000 description 1
- 229940124639 Selective inhibitor Drugs 0.000 description 1
- 208000001435 Thromboembolism Diseases 0.000 description 1
- 108010069102 Thromboxane-A synthase Proteins 0.000 description 1
- 208000007814 Unstable Angina Diseases 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 238000004220 aggregation Methods 0.000 description 1
- 230000002776 aggregation Effects 0.000 description 1
- 229910052783 alkali metal Inorganic materials 0.000 description 1
- 150000001340 alkali metals Chemical class 0.000 description 1
- 150000001413 amino acids Chemical class 0.000 description 1
- 230000000702 anti-platelet effect Effects 0.000 description 1
- 239000003146 anticoagulant agent Substances 0.000 description 1
- 210000001367 artery Anatomy 0.000 description 1
- 210000004369 blood Anatomy 0.000 description 1
- 239000008280 blood Substances 0.000 description 1
- 210000001715 carotid artery Anatomy 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 208000035850 clinical syndrome Diseases 0.000 description 1
- 238000013270 controlled release Methods 0.000 description 1
- 208000002528 coronary thrombosis Diseases 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- IZEKFCXSFNUWAM-UHFFFAOYSA-N dipyridamole Chemical compound C=12N=C(N(CCO)CCO)N=C(N3CCCCC3)C2=NC(N(CCO)CCO)=NC=1N1CCCCC1 IZEKFCXSFNUWAM-UHFFFAOYSA-N 0.000 description 1
- 238000004090 dissolution Methods 0.000 description 1
- 239000002552 dosage form Substances 0.000 description 1
- 239000003480 eluent Substances 0.000 description 1
- 238000003810 ethyl acetate extraction Methods 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 238000007710 freezing Methods 0.000 description 1
- 230000007062 hydrolysis Effects 0.000 description 1
- 238000011534 incubation Methods 0.000 description 1
- 230000001939 inductive effect Effects 0.000 description 1
- 239000003112 inhibitor Substances 0.000 description 1
- 238000002347 injection Methods 0.000 description 1
- 239000007924 injection Substances 0.000 description 1
- 229910052500 inorganic mineral Inorganic materials 0.000 description 1
- 229940079865 intestinal antiinfectives imidazole derivative Drugs 0.000 description 1
- 208000001286 intracranial vasospasm Diseases 0.000 description 1
- 206010025482 malaise Diseases 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 1
- 235000010755 mineral Nutrition 0.000 description 1
- 239000011707 mineral Substances 0.000 description 1
- 239000000203 mixture Substances 0.000 description 1
- 150000007524 organic acids Chemical group 0.000 description 1
- 230000003285 pharmacodynamic effect Effects 0.000 description 1
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 1
- 239000000047 product Substances 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 239000001509 sodium citrate Substances 0.000 description 1
- NLJMYIDDQXHKNR-UHFFFAOYSA-K sodium citrate Chemical compound O.O.[Na+].[Na+].[Na+].[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O NLJMYIDDQXHKNR-UHFFFAOYSA-K 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 239000000829 suppository Substances 0.000 description 1
- 230000001225 therapeutic effect Effects 0.000 description 1
- 210000003462 vein Anatomy 0.000 description 1
Landscapes
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
The invention relates to the field of medical chemistry, in particular to a preparation method and medical purpose of 4-(3-(dimethoxy) propoxy)-3, 5-dimethoxy benzoic acid (I). Proved by pharmacological tests, the compound (I) of the invention has curing effect on cardiovascular and cerebrovascular diseases caused by platelet aggregation.
Description
Technical field
The present invention relates to the pharmaceutical chemistry field, be specifically related to a 4-(3-(dimethylamino) propoxy-)-3,5-dimethoxybenzoic acid compound and salt thereof, it has therapeutic action to the cardiovascular and cerebrovascular diseases that platelet aggregation causes.
Background technology
Cardiovascular and cerebrovascular diseases is common disease, the frequently-occurring disease of serious harm human health, and along with the astogeny of social population, sickness rate rises day by day.According to statistics, the whole world has 1,600 ten thousand people to die from all kinds of cardiovascular and cerebrovascular diseases every year, is the No.1 killer who threatens human health.
Thromboembolism be cause cardiovascular and cerebrovascular diseases important factor it-; Coronary artery disease and corresponding ischemic complication thereof can cause the various clinical syndromes; Like apoplexy, myocardial infarction or peripheral arterial disease, the thrombus artery-clogging of main diseases because of in artery, forming exactly causes severe ischemic.With coronary artery thrombosis and cerebral thrombosis is that the thrombotic disease of core also has very high M & M in China, and therefore, anti-hemostasis suppository has also just become one of current the most popular research topic in cardiovascular disorder field.
Ozagrel and Dazoxiben are the TXA of high selectivity
2Synthetase inhibitors; Has very high antiplatelet aggregative activity (referring to Iizuka.K; Akahane.K; Momose.D, et al.Highyl selective inhibitors of thromboxanesynthetase.1.Imidazole Derivatives.J.Med.Chem.1981,24 (10): 1139-1148).Can suppress cerebral thrombosis and cerebral vasospasm.(referring to Song Bo, it is refined etc. that Jiang Congqing, behaviour pass, the clinical study of Sodium Ozagrel treatment unstable angina pectoris, Chinese omni-doctor's magazine, 2006,5 (4): 255-256 to be mainly used in acute cerebral infarction, coronary heart disease and anginal treatment clinically; Wu Jinying, the 42 routine clinical observations of Sodium Ozagrel treatment acute cerebral infarction).The structural formula of ozagrel and Dazoxiben is following:
Clinical needs are medicament for resisting platelet aggregation more effectively.
Summary of the invention
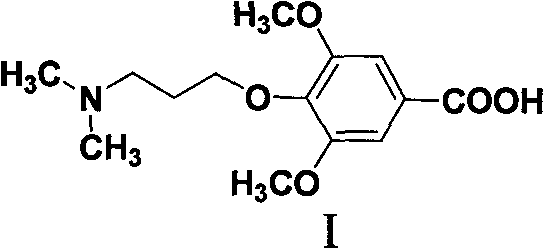
The invention discloses a 4-(3-(dimethylamino) propoxy-)-3,5-dimethoxybenzoic acid compound, I is following for its structural formula:
The present invention also comprises the pharmacy acceptable salt of formula I compound, and these salt are formula I compound and metal, earth alkali metal, amino acid, contain the salt that amino basic cpd, mineral acid or organic acid form.The preferably sodium salt of formula I compound, sylvite, calcium salt, magnesium salts, arginic acid salt, hydrochloride, vitriol, phosphoric acid salt, PHENRAMINE MALEATE, fumarate, citrate, mesylate, tosilate or tartrate.
Formula I compound of the present invention can be used following method preparation:
More preferred manufacturing procedure is following:
In three-necked bottle, add Ethyl syringate, N, N-dimethylamino chloropropane hydrochloride, salt of wormwood, N, dinethylformamide (DMF) is made solvent; Reaction 7h under 90 ℃, TLC detects to reaction complete basically, cools off; Filter, in filtrating, add entry, use ethyl acetate extraction then three times; The combined ethyl acetate layer is after water washing three times, through anhydrous sodium sulfate drying; Behind the reclaim under reduced pressure ETHYLE ACETATE lark liquid, through the silicagel column wash-out separate 4-(3-(dimethylamino) propoxy-)-3,5-dimethoxybenzoic acid ethyl ester weak yellow liquid.
In round-bottomed flask, add 4-(3-(dimethylamino) propoxy-)-3 successively, 5-dimethoxybenzoic acid ethyl ester, NaOH, ethanol, water, hydrolysis reaction 4h, TLC [developping agent: V (CHCl
3): V (methyl alcohol)=4: 1] detection reaction is complete basically, regulates about pH to 2 with concentrated hydrochloric acid, and decompression and solvent recovery is to doing; Add the 15mL absolute ethyl alcohol, heating in water bath 10min, suction filtration while hot; Cooling, the adularescent deposition is separated out, and filters; Filter cake is used the absolute ethyl alcohol recrystallization, gets 4-(3-(dimethylamino) propoxy-)-3,5-dimethoxybenzoic acid hydrochloride white crystal.
The invention also discloses a kind of pharmaceutical composition; Wherein contain the formula I compound and the pharmaceutically acceptable carrier of treating significant quantity, this pharmaceutical composition can also be pharmacy acceptable salt and the pharmaceutically acceptable carrier that contains the formula I compound of treating significant quantity.Said pharmaceutical composition can be a dosage form conventional on the technology of pharmaceutics such as conventional tablet or capsule, slow releasing tablet or capsule, controlled release tablet or capsule, oral liquid, injection.
Usually, when formula I compound of the present invention was used to treat, the human dosage range was 0.5mg~2000mg/ days.Also can be according to the difference and the disease severity of formulation, using dosage exceeds this scope.
Pharmacological testing proves that formula I compound or pharmaceutically acceptable salt thereof of the present invention has the effect of the cardiovascular and cerebrovascular diseases of treating or preventing to cause because of platelet aggregation, particularly treats diseases such as ischemic heart disease, myocardial infarction, Stroke, cerebral infarction disease.
Be the pharmacodynamics test and the result of formula I compound of the present invention below:
Medicine: positive drug: Sodium Ozagrel
Receive the reagent thing: formula I compound of the present invention
Dosage: Sodium Ozagrel, human dosage 120mg/60kg/d, rabbit consumption (120mg ÷ 60kg) * 3=6mg/kg.Sodium Ozagrel rabbit administration volume is 2mL/kg, and the administration concentration of rabbit is 3.00mg/mL, and volumetric molar concentration is 11.99mmol/L, volumetric molar concentrations such as given the test agent and Sodium Ozagrel.
Compound method: take by weighing Sodium Ozagrel 75mg, add 25mL saline water, final concentration 11.99mmol/L.Melbourne invention formula I compound such as take by weighing, add 15mL saline water earlier, ultrasonic dissolution adds NaOH solution 20~25 μ L of 1mol/L again, and solution is clarified fully, adds saline water again and is diluted to required volume.
Experimental technique:
To clean the level male rabbit (2.0~2.5kg) random packet, 6 every group, give compound (I) respectively, Sodium Ozagrel and saline water, administration volume are 2mL/kg.Anaesthetize after the auricular vein administration, the carotid artery intubate is got blood, 3.8% Sodium Citrate (1: 9) anti-freezing; With the centrifugal 10min of 800r/min, with the preparation platelet rich plasma (platelet-rich plasma, PRP); Afterwards again with the centrifugal 10min of 3000r/min; (platelet-poor plasma, PPP), the aggregation inducing agent is with ADP (final concentration 30 μ mol/L) for the preparation platelet poor plasma.Every pipe 270 μ l PRP incubation 5min add ADP then and induce gathering, measure the thrombocyte MA with LG-PABER-1 type platelet aggregation instrument.
Calculate the thrombocyte inhibiting rate by following formula: inhibiting rate (%)=[(blank group MA-administration group MA)/blank group MA] * 100%, the result is organized a t check, data are seen table.
| Group | Thrombocyte MA (%) | Assemble inhibiting rate (%) |
| Blank group Sodium Ozagrel formula I compound | 38.0±6.3 28.6±3.8 1.7±1.3 ** | 24.7 95.4 |
Table 1 shows: the platelet aggregation-against experimental result shows in the body of formula I compound of the present invention, and their platelet aggregation inhibitory activity is respectively 3.9 times of ozagrel positive drug, demonstrates excellent development and is worth.
Embodiment
Embodiment 1
4-(3-(dimethylamino) propoxy-)-3,5-dimethoxybenzoic acid hydrochloride synthetic
1,4-(3-(dimethylamino) propoxy-)-3,5-dimethoxybenzoic acid ethyl ester synthetic
Reaction formula
Reactions step
In three-necked bottle, add Ethyl syringate (3.0g, 0.013mol), N, N-dimethylamino chloropropane hydrochloride (2.5g, 0.016mol), salt of wormwood (4.1g; 0.030mol), DMF (50mL) reacts 7h down, TLC (developping agent: ETHYLE ACETATE) detect to reacting complete basically in 90 ℃; Cooling is crossed and is filtered filtrating, adds water 50mL, with ETHYLE ACETATE (3 * 50mL) extractions, combined ethyl acetate layer; Water (after 3 * 25mL) washings, through anhydrous sodium sulfate drying, the yellow liquid of reclaim under reduced pressure ETHYLE ACETATE; Separate through the silicagel column wash-out, ETHYLE ACETATE is eluent, collects product; Get 4-(3-(dimethylamino) propoxy-)-3,5-dimethoxybenzoic acid ethyl ester weak yellow liquid 3.1g, yield 75.1% behind the decompression and solvent recovery.
1H-NMR(CDCl
3,300MHz)δ:7.30(s,2H,Ar-H),4.38(q,J=7.2Hz,2H,CH
3 CH 2 O),4.10(t,J=6.3Hz,2H,CH
2CH
2 CH 2 O),3.90(s,6H,-OCH3),2.61(t,J=7.5Hz,2H,N
CH 2 CH
2CH
2O),2.33{s,6H,-N(CH
3)
2},1.98(m,2H,NCH
2 CH 2 CH
2O),4.38(t,J=7.2Hz,3H,
CH 3 CH
2O);
13C-NMR(CDCl
3,75.5MHz)δ:166.2,153.1,141.3,125.5,106.8,71.7,61.1,56.4,56.2,45.2,28.0,14.4;IR(KBr,cm
-1)υ:2953.9,2816.1,2788.0,2763.9,1704.3,1588.8,1501.3,1474.9,1415.1,1370.7,1331.2,1257.9,1234.7,1186.2,1130.8,1036.7,916.6,867.4,762.7,721.5;ESI-Mass(
+c)m/z?for?C
16H
25NO
5:312.06(M
++H).
2,4-(3-(dimethylamino) propoxy-)-3,5-dimethoxybenzoic acid hydrochloride synthetic
Reaction formula
Reactions step
In round-bottomed flask, add 4-(3-(dimethylamino) propoxy-)-3 successively, 5-dimethoxybenzoic acid ethyl ester (3.0g, 0.0096mol), NaOH (0.5g, 0.013mol), ethanol (15mL), pure water (10mL), hydrolysis 4h, TLC [developping agent: V (CHCl
3): V (methyl alcohol)=4: 1] detection reaction is complete basically, regulates about pH to 2 with 12mol/L hydrochloric acid, and decompression and solvent recovery is done to clean, adds the 15mL absolute ethyl alcohol; Heating in water bath 10min, suction filtration cools off while hot, and the adularescent deposition is separated out; Filter, filter cake is used the absolute ethyl alcohol recrystallization, and drying gets 4-(3-(dimethylamino) propoxy-)-3; 5-dimethoxybenzoic acid hydrochloride white solid 2.4g, yield 78.0%, m.p.249.6~251.0 ℃.
1H-NMR(D
2O,300MHz)δ:7.25(s,2H,Ar-H),4.10(s,2H,CH
2CH
2 CH 2 O),3.84(s,6H,-OCH
3),3.37(t,J=6.6Hz,2H,N
CH 2 CH
2CH
2O),2.90{s,6H,-N(CH
3)
2},2.10(m,2H,NCH
2 CH 2 CH
2O);
13C-NMR(D
2O,75.5MHz)δ:169.2,152.3,139.8,125.7,107.0,71.5,56.8,56.3,43.4,24.9;IR(KBr,cm
-1)υ:2966.7,2687.3,1699.3,1590.8,1504.7,1466.6,1415.1,1380.0,1317.1,1178.9,1129.6,1055.2,934.0,867.9,768.9;ESI-Mass(
+c)m/z?for?C
14H
21NO
5:(M
++H)284.10.
Claims (6)
1. the compound of structural formula (I) or its pharmacy acceptable salt:
2. the preparation method of the compound of claim 1 comprises:
3. the compound of claim 1 or its pharmacy acceptable salt, wherein pharmacy acceptable salt is sodium salt, sylvite, calcium salt, magnesium salts, arginic acid salt, hydrochloride, vitriol, phosphoric acid salt, PHENRAMINE MALEATE, fumarate, citrate, mesylate, tosilate or the tartrate of the compound of claim 1.
4. pharmaceutical composition wherein contains compound or its pharmacy acceptable salt and the pharmaceutically acceptable carrier of claim 1.
5. the compound of claim 1 or its pharmacy acceptable salt are used to prepare the purposes of the cardiovascular and cerebrovascular diseases medicament of treating or preventing to be caused by platelet aggregation.
6. the purposes of claim 5, the cardiovascular and cerebrovascular diseases that is wherein caused by platelet aggregation is ischemic heart disease, myocardial infarction or Stroke.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN 201010151142 CN101811974B (en) | 2010-04-15 | 2010-04-15 | 4-(3-(dimethoxy) propoxy)-3, 5-dimethoxy benzoic acid, and preparation method and medical purpose thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN 201010151142 CN101811974B (en) | 2010-04-15 | 2010-04-15 | 4-(3-(dimethoxy) propoxy)-3, 5-dimethoxy benzoic acid, and preparation method and medical purpose thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN101811974A CN101811974A (en) | 2010-08-25 |
| CN101811974B true CN101811974B (en) | 2012-12-12 |
Family
ID=42619362
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN 201010151142 Active CN101811974B (en) | 2010-04-15 | 2010-04-15 | 4-(3-(dimethoxy) propoxy)-3, 5-dimethoxy benzoic acid, and preparation method and medical purpose thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN101811974B (en) |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN113893242B (en) * | 2021-10-12 | 2023-10-13 | 广东宝康生物医药有限公司 | Use of ASH in myocardial protection |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4636500A (en) * | 1978-12-13 | 1987-01-13 | Pfizer Inc. | N-(phenoxyalkyl)imidazoles as selective inhibitors of the thromboxane synthetase enzyme and pharmaceutical compositions thereof |
| CN1376667A (en) * | 2002-04-09 | 2002-10-30 | 胥淑蓉 | Medical compound with antalgic, inflammation relieving and thrombocyte decoagulating actions |
-
2010
- 2010-04-15 CN CN 201010151142 patent/CN101811974B/en active Active
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4636500A (en) * | 1978-12-13 | 1987-01-13 | Pfizer Inc. | N-(phenoxyalkyl)imidazoles as selective inhibitors of the thromboxane synthetase enzyme and pharmaceutical compositions thereof |
| CN1376667A (en) * | 2002-04-09 | 2002-10-30 | 胥淑蓉 | Medical compound with antalgic, inflammation relieving and thrombocyte decoagulating actions |
Non-Patent Citations (1)
| Title |
|---|
| 周远鹏等.益母草碱、丁香酰胍醇及丁香酸氨基醇酯类化合物抗血小板聚集活性及其与结构的关系.《中国药学杂志》.1996,第31卷(第5期),271-274. * |
Also Published As
| Publication number | Publication date |
|---|---|
| CN101811974A (en) | 2010-08-25 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| TWI771272B (en) | Use of clemizole compounds for prevention and treatment of liver cancer | |
| DE69622532T2 (en) | GLYCOPROTEIN IIB / IIIA ANTAGONISTS | |
| CN101362702B (en) | Emodin derivates and application thereof in anti-cancer medicine preparation | |
| TWI774683B (en) | SOLID FORMS OF AN sGC STIMULATOR | |
| US20190255036A1 (en) | Compound and method for reducing neuropathic pain and depression | |
| EP2305260B1 (en) | Psychotropic agent and health food containing benzylisoquinoline derivative | |
| CN110234636B (en) | Compositions comprising methylphenidate prodrugs, methods of making and using the same | |
| CN105683152B (en) | Compositions for the treatment of hypertension and/or fibrosis | |
| CN116209442A (en) | Pyrido [4,3-b ] indole derivatives and their use as medicaments | |
| EP3546450A1 (en) | Piperidine-2,6-dione derivative and treatment for ulcerative colitis | |
| TW201737943A (en) | Methods of using FASN inhibitors | |
| CN104231013A (en) | Gastrodin ferulate compound and preparation method and application of gastrodin ferulate compound | |
| WO2019084300A1 (en) | Treatment of glioblastoma with fasn inhibitors | |
| CN103864760A (en) | Hydroxyfasudil compound | |
| CN101250183A (en) | Optical isomer of rebeprazole as well as preparation method and medical use thereof | |
| CN101851209B (en) | Imidazole ethyl vanillic acid ether, preparation method and medicinal application thereof | |
| CN101811974B (en) | 4-(3-(dimethoxy) propoxy)-3, 5-dimethoxy benzoic acid, and preparation method and medical purpose thereof | |
| AU2019254962B9 (en) | Isoindole derivatives | |
| CN105037180B (en) | Central analgesia noval chemical compound, the Preparation method and use of a kind of double action | |
| CN101812025B (en) | Pyrazine aromatic acid ester compound, preparation method and medical application | |
| CN102617448B (en) | 4-methoxymethyl-4- (N-propionyl) aniline piperidine compound, preparation method and application | |
| WO2022125614A1 (en) | Phosphonates as inhibitors of enpp1 and cdnp | |
| CN114853720A (en) | Novel flavane derivative in dragon's blood, preparation method, pharmaceutical composition and application thereof | |
| CN105829291B (en) | Compounds and pharmaceutical compositions related to pain | |
| JPH11302174A (en) | Cell adhesion inhibitor |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| CP01 | Change in the name or title of a patent holder | ||
| CP01 | Change in the name or title of a patent holder |
Address after: 230088 F8 Floor, Venture Center, Hefei High-tech Zone, Anhui Province Patentee after: Hefei Medical and Pharmaceutical Co., Ltd. Address before: 230088 F8 Floor, Venture Center, Hefei High-tech Zone, Anhui Province Patentee before: Hefei Yigong Medicine Co., Ltd. |