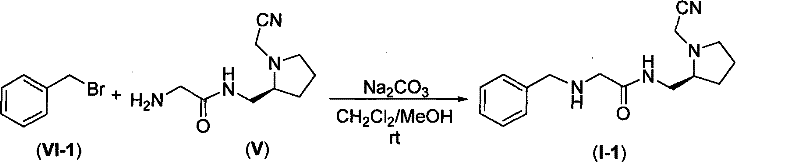CN101684088B - Cyanomethyl pyrrole derivative and preparation method and application thereof - Google Patents
Cyanomethyl pyrrole derivative and preparation method and application thereof Download PDFInfo
- Publication number
- CN101684088B CN101684088B CN2008101515014A CN200810151501A CN101684088B CN 101684088 B CN101684088 B CN 101684088B CN 2008101515014 A CN2008101515014 A CN 2008101515014A CN 200810151501 A CN200810151501 A CN 200810151501A CN 101684088 B CN101684088 B CN 101684088B
- Authority
- CN
- China
- Prior art keywords
- compound
- methyl
- amino
- acetyl
- cyanomethyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
- 238000002360 preparation method Methods 0.000 title abstract description 8
- MKVATVUOVMCHJJ-UHFFFAOYSA-N 2-(1h-pyrrol-2-yl)acetonitrile Chemical class N#CCC1=CC=CN1 MKVATVUOVMCHJJ-UHFFFAOYSA-N 0.000 title description 2
- 239000003814 drug Substances 0.000 claims abstract description 12
- 206010012601 diabetes mellitus Diseases 0.000 claims abstract description 10
- 239000000203 mixture Substances 0.000 claims abstract description 8
- 150000001875 compounds Chemical class 0.000 claims description 67
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 claims description 26
- -1 1, 2-dihydroquinolin-2-one-4-yl Chemical group 0.000 claims description 23
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 23
- 238000000034 method Methods 0.000 claims description 14
- 150000003839 salts Chemical class 0.000 claims description 13
- 239000007787 solid Substances 0.000 claims description 11
- 239000002904 solvent Substances 0.000 claims description 10
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 claims description 9
- DHMQDGOQFOQNFH-UHFFFAOYSA-N Glycine Chemical compound NCC(O)=O DHMQDGOQFOQNFH-UHFFFAOYSA-N 0.000 claims description 8
- 239000007788 liquid Substances 0.000 claims description 7
- FCSKOFQQCWLGMV-UHFFFAOYSA-N 5-{5-[2-chloro-4-(4,5-dihydro-1,3-oxazol-2-yl)phenoxy]pentyl}-3-methylisoxazole Chemical compound O1N=C(C)C=C1CCCCCOC1=CC=C(C=2OCCN=2)C=C1Cl FCSKOFQQCWLGMV-UHFFFAOYSA-N 0.000 claims description 6
- NLFBCYMMUAKCPC-KQQUZDAGSA-N ethyl (e)-3-[3-amino-2-cyano-1-[(e)-3-ethoxy-3-oxoprop-1-enyl]sulfanyl-3-oxoprop-1-enyl]sulfanylprop-2-enoate Chemical compound CCOC(=O)\C=C\SC(=C(C#N)C(N)=O)S\C=C\C(=O)OCC NLFBCYMMUAKCPC-KQQUZDAGSA-N 0.000 claims description 6
- 150000007529 inorganic bases Chemical class 0.000 claims description 6
- 239000008194 pharmaceutical composition Substances 0.000 claims description 6
- ONIBWKKTOPOVIA-BYPYZUCNSA-N L-Proline Chemical compound OC(=O)[C@@H]1CCCN1 ONIBWKKTOPOVIA-BYPYZUCNSA-N 0.000 claims description 5
- 239000003795 chemical substances by application Substances 0.000 claims description 5
- 238000002347 injection Methods 0.000 claims description 5
- 239000007924 injection Substances 0.000 claims description 5
- QARBMVPHQWIHKH-UHFFFAOYSA-N methanesulfonyl chloride Chemical compound CS(Cl)(=O)=O QARBMVPHQWIHKH-UHFFFAOYSA-N 0.000 claims description 5
- 125000006239 protecting group Chemical group 0.000 claims description 5
- 125000001255 4-fluorophenyl group Chemical group [H]C1=C([H])C(*)=C([H])C([H])=C1F 0.000 claims description 4
- FKLJPTJMIBLJAV-UHFFFAOYSA-N Compound IV Chemical compound O1N=C(C)C=C1CCCCCCCOC1=CC=C(C=2OCCN=2)C=C1 FKLJPTJMIBLJAV-UHFFFAOYSA-N 0.000 claims description 4
- 239000004471 Glycine Substances 0.000 claims description 4
- 229930182821 L-proline Natural products 0.000 claims description 4
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Substances [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 claims description 4
- 229910000027 potassium carbonate Inorganic materials 0.000 claims description 4
- 229960002429 proline Drugs 0.000 claims description 4
- 238000011282 treatment Methods 0.000 claims description 4
- REXUYBKPWIPONM-UHFFFAOYSA-N 2-bromoacetonitrile Chemical compound BrCC#N REXUYBKPWIPONM-UHFFFAOYSA-N 0.000 claims description 3
- 239000012359 Methanesulfonyl chloride Substances 0.000 claims description 3
- 125000002777 acetyl group Chemical group [H]C([H])([H])C(*)=O 0.000 claims description 3
- 125000000440 benzylamino group Chemical group [H]N(*)C([H])([H])C1=C([H])C([H])=C([H])C([H])=C1[H] 0.000 claims description 3
- 238000009472 formulation Methods 0.000 claims description 3
- 238000010438 heat treatment Methods 0.000 claims description 3
- 230000008569 process Effects 0.000 claims description 3
- 125000004178 (C1-C4) alkyl group Chemical group 0.000 claims description 2
- 125000004209 (C1-C8) alkyl group Chemical group 0.000 claims description 2
- 229910010084 LiAlH4 Inorganic materials 0.000 claims description 2
- 239000002253 acid Substances 0.000 claims description 2
- 238000010511 deprotection reaction Methods 0.000 claims description 2
- 239000012280 lithium aluminium hydride Substances 0.000 claims description 2
- 238000004519 manufacturing process Methods 0.000 claims description 2
- 150000007530 organic bases Chemical class 0.000 claims description 2
- PXIPVTKHYLBLMZ-UHFFFAOYSA-N sodium azide Substances [Na+].[N-]=[N+]=[N-] PXIPVTKHYLBLMZ-UHFFFAOYSA-N 0.000 claims description 2
- 239000007858 starting material Substances 0.000 claims description 2
- RWRDLPDLKQPQOW-UHFFFAOYSA-N tetrahydropyrrole Natural products C1CCNC1 RWRDLPDLKQPQOW-UHFFFAOYSA-N 0.000 claims 6
- HTSGKJQDMSTCGS-UHFFFAOYSA-N 1,4-bis(4-chlorophenyl)-2-(4-methylphenyl)sulfonylbutane-1,4-dione Chemical compound C1=CC(C)=CC=C1S(=O)(=O)C(C(=O)C=1C=CC(Cl)=CC=1)CC(=O)C1=CC=C(Cl)C=C1 HTSGKJQDMSTCGS-UHFFFAOYSA-N 0.000 claims 1
- VFRSADQPWYCXDG-LEUCUCNGSA-N ethyl (2s,5s)-5-methylpyrrolidine-2-carboxylate;2,2,2-trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F.CCOC(=O)[C@@H]1CC[C@H](C)N1 VFRSADQPWYCXDG-LEUCUCNGSA-N 0.000 claims 1
- 230000002194 synthesizing effect Effects 0.000 claims 1
- POCVKESNHZPAJL-UHFFFAOYSA-N 2-pyrrol-1-ylacetonitrile Chemical class N#CCN1C=CC=C1 POCVKESNHZPAJL-UHFFFAOYSA-N 0.000 abstract description 2
- KAESVJOAVNADME-UHFFFAOYSA-N Pyrrole Chemical compound C=1C=CNC=1 KAESVJOAVNADME-UHFFFAOYSA-N 0.000 description 23
- 239000010409 thin film Substances 0.000 description 13
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 12
- 102000016622 Dipeptidyl Peptidase 4 Human genes 0.000 description 9
- 101000930822 Giardia intestinalis Dipeptidyl-peptidase 4 Proteins 0.000 description 9
- 230000015572 biosynthetic process Effects 0.000 description 9
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 9
- 238000003786 synthesis reaction Methods 0.000 description 9
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 8
- 238000003756 stirring Methods 0.000 description 8
- 210000004369 blood Anatomy 0.000 description 7
- 239000008280 blood Substances 0.000 description 7
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 6
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 6
- HIMXGTXNXJYFGB-UHFFFAOYSA-N alloxan Chemical compound O=C1NC(=O)C(=O)C(=O)N1 HIMXGTXNXJYFGB-UHFFFAOYSA-N 0.000 description 6
- 238000006243 chemical reaction Methods 0.000 description 6
- 229940079593 drug Drugs 0.000 description 6
- 239000008187 granular material Substances 0.000 description 6
- NOESYZHRGYRDHS-UHFFFAOYSA-N insulin Chemical compound N1C(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(NC(=O)CN)C(C)CC)CSSCC(C(NC(CO)C(=O)NC(CC(C)C)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CCC(N)=O)C(=O)NC(CC(C)C)C(=O)NC(CCC(O)=O)C(=O)NC(CC(N)=O)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CSSCC(NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2C=CC(O)=CC=2)NC(=O)C(CC(C)C)NC(=O)C(C)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2NC=NC=2)NC(=O)C(CO)NC(=O)CNC2=O)C(=O)NCC(=O)NC(CCC(O)=O)C(=O)NC(CCCNC(N)=N)C(=O)NCC(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC(O)=CC=3)C(=O)NC(C(C)O)C(=O)N3C(CCC3)C(=O)NC(CCCCN)C(=O)NC(C)C(O)=O)C(=O)NC(CC(N)=O)C(O)=O)=O)NC(=O)C(C(C)CC)NC(=O)C(CO)NC(=O)C(C(C)O)NC(=O)C1CSSCC2NC(=O)C(CC(C)C)NC(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CC(N)=O)NC(=O)C(NC(=O)C(N)CC=1C=CC=CC=1)C(C)C)CC1=CN=CN1 NOESYZHRGYRDHS-UHFFFAOYSA-N 0.000 description 6
- 238000002156 mixing Methods 0.000 description 6
- 239000003921 oil Substances 0.000 description 6
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 6
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 6
- 239000000047 product Substances 0.000 description 6
- 238000007873 sieving Methods 0.000 description 6
- 229920000168 Microcrystalline cellulose Polymers 0.000 description 5
- 229920000881 Modified starch Polymers 0.000 description 5
- 229920002472 Starch Polymers 0.000 description 5
- 239000000706 filtrate Substances 0.000 description 5
- 239000008108 microcrystalline cellulose Substances 0.000 description 5
- 235000019813 microcrystalline cellulose Nutrition 0.000 description 5
- 229940016286 microcrystalline cellulose Drugs 0.000 description 5
- CDBYLPFSWZWCQE-UHFFFAOYSA-L sodium carbonate Substances [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 5
- 229910000029 sodium carbonate Inorganic materials 0.000 description 5
- 239000000243 solution Substances 0.000 description 5
- 239000008107 starch Substances 0.000 description 5
- 235000019698 starch Nutrition 0.000 description 5
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 5
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 4
- DTQVDTLACAAQTR-UHFFFAOYSA-N Trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 4
- DPXJVFZANSGRMM-UHFFFAOYSA-N acetic acid;2,3,4,5,6-pentahydroxyhexanal;sodium Chemical compound [Na].CC(O)=O.OCC(O)C(O)C(O)C(O)C=O DPXJVFZANSGRMM-UHFFFAOYSA-N 0.000 description 4
- 239000004480 active ingredient Substances 0.000 description 4
- 230000037396 body weight Effects 0.000 description 4
- 239000001768 carboxy methyl cellulose Substances 0.000 description 4
- FPAFDBFIGPHWGO-UHFFFAOYSA-N dioxosilane;oxomagnesium;hydrate Chemical compound O.[Mg]=O.[Mg]=O.[Mg]=O.O=[Si]=O.O=[Si]=O.O=[Si]=O.O=[Si]=O FPAFDBFIGPHWGO-UHFFFAOYSA-N 0.000 description 4
- 238000001035 drying Methods 0.000 description 4
- 238000001914 filtration Methods 0.000 description 4
- 239000008103 glucose Substances 0.000 description 4
- 239000003112 inhibitor Substances 0.000 description 4
- 235000019359 magnesium stearate Nutrition 0.000 description 4
- YMWUJEATGCHHMB-UHFFFAOYSA-N methylene chloride Substances ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 4
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 4
- 235000019812 sodium carboxymethyl cellulose Nutrition 0.000 description 4
- 229920001027 sodium carboxymethylcellulose Polymers 0.000 description 4
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 3
- 102000004877 Insulin Human genes 0.000 description 3
- 108090001061 Insulin Proteins 0.000 description 3
- 241000699670 Mus sp. Species 0.000 description 3
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- 239000002775 capsule Substances 0.000 description 3
- 239000003054 catalyst Substances 0.000 description 3
- 239000012230 colorless oil Substances 0.000 description 3
- 239000013078 crystal Substances 0.000 description 3
- 239000012153 distilled water Substances 0.000 description 3
- 230000002218 hypoglycaemic effect Effects 0.000 description 3
- 229940125396 insulin Drugs 0.000 description 3
- 239000000463 material Substances 0.000 description 3
- 239000000546 pharmaceutical excipient Substances 0.000 description 3
- 239000011734 sodium Substances 0.000 description 3
- 208000001072 type 2 diabetes mellitus Diseases 0.000 description 3
- 0 *C[C@@]1N(CC#N)CCC1 Chemical compound *C[C@@]1N(CC#N)CCC1 0.000 description 2
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 2
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 description 2
- 101800000224 Glucagon-like peptide 1 Proteins 0.000 description 2
- DTHNMHAUYICORS-KTKZVXAJSA-N Glucagon-like peptide 1 Chemical compound C([C@@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](C)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCCCN)C(=O)NCC(=O)N[C@@H](CCCNC(N)=N)C(N)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](C)NC(=O)[C@H](C)NC(=O)[C@H](CCC(N)=O)NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](CC=1C=CC=CC=1)NC(=O)[C@@H](NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H](N)CC=1N=CNC=1)[C@@H](C)O)[C@@H](C)O)C(C)C)C1=CC=CC=C1 DTHNMHAUYICORS-KTKZVXAJSA-N 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 2
- 102100040918 Pro-glucagon Human genes 0.000 description 2
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical class [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 2
- 229930006000 Sucrose Natural products 0.000 description 2
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 2
- 239000003472 antidiabetic agent Substances 0.000 description 2
- GZUXJHMPEANEGY-UHFFFAOYSA-N bromomethane Chemical compound BrC GZUXJHMPEANEGY-UHFFFAOYSA-N 0.000 description 2
- OSGAYBCDTDRGGQ-UHFFFAOYSA-L calcium sulfate Chemical compound [Ca+2].[O-]S([O-])(=O)=O OSGAYBCDTDRGGQ-UHFFFAOYSA-L 0.000 description 2
- 125000002057 carboxymethyl group Chemical group [H]OC(=O)C([H])([H])[*] 0.000 description 2
- 239000003153 chemical reaction reagent Substances 0.000 description 2
- 238000004440 column chromatography Methods 0.000 description 2
- 239000003085 diluting agent Substances 0.000 description 2
- 239000003937 drug carrier Substances 0.000 description 2
- 230000000694 effects Effects 0.000 description 2
- 239000000945 filler Substances 0.000 description 2
- 238000005984 hydrogenation reaction Methods 0.000 description 2
- BDAGIHXWWSANSR-UHFFFAOYSA-N methanoic acid Natural products OC=O BDAGIHXWWSANSR-UHFFFAOYSA-N 0.000 description 2
- 239000012074 organic phase Substances 0.000 description 2
- 239000008188 pellet Substances 0.000 description 2
- 239000011541 reaction mixture Substances 0.000 description 2
- 210000002966 serum Anatomy 0.000 description 2
- 159000000000 sodium salts Chemical class 0.000 description 2
- 239000005720 sucrose Substances 0.000 description 2
- 238000000967 suction filtration Methods 0.000 description 2
- 239000003826 tablet Substances 0.000 description 2
- 230000001225 therapeutic effect Effects 0.000 description 2
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 2
- 238000005406 washing Methods 0.000 description 2
- 230000004584 weight gain Effects 0.000 description 2
- 235000019786 weight gain Nutrition 0.000 description 2
- ZOBPZXTWZATXDG-UHFFFAOYSA-N 1,3-thiazolidine-2,4-dione Chemical compound O=C1CSC(=O)N1 ZOBPZXTWZATXDG-UHFFFAOYSA-N 0.000 description 1
- MCEYVJFKCOFDCS-UHFFFAOYSA-N 1-(bromomethyl)-2-chloro-3-methylbenzene Chemical compound CC1=CC=CC(CBr)=C1Cl MCEYVJFKCOFDCS-UHFFFAOYSA-N 0.000 description 1
- NVNPLEPBDPJYRZ-UHFFFAOYSA-N 1-(bromomethyl)-4-fluorobenzene Chemical compound FC1=CC=C(CBr)C=C1 NVNPLEPBDPJYRZ-UHFFFAOYSA-N 0.000 description 1
- OSWFIVFLDKOXQC-UHFFFAOYSA-N 4-(3-methoxyphenyl)aniline Chemical compound COC1=CC=CC(C=2C=CC(N)=CC=2)=C1 OSWFIVFLDKOXQC-UHFFFAOYSA-N 0.000 description 1
- BBAHJCUCNVVEQU-UHFFFAOYSA-N 4-(bromomethyl)-1h-quinolin-2-one Chemical compound C1=CC=C2C(CBr)=CC(=O)NC2=C1 BBAHJCUCNVVEQU-UHFFFAOYSA-N 0.000 description 1
- 206010067484 Adverse reaction Diseases 0.000 description 1
- 229940077274 Alpha glucosidase inhibitor Drugs 0.000 description 1
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 1
- 229920002785 Croscarmellose sodium Polymers 0.000 description 1
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 1
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 1
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 1
- 229920001353 Dextrin Polymers 0.000 description 1
- 239000004375 Dextrin Substances 0.000 description 1
- 229940124213 Dipeptidyl peptidase 4 (DPP IV) inhibitor Drugs 0.000 description 1
- 238000005033 Fourier transform infrared spectroscopy Methods 0.000 description 1
- 239000004366 Glucose oxidase Substances 0.000 description 1
- 108010015776 Glucose oxidase Proteins 0.000 description 1
- 206010019851 Hepatotoxicity Diseases 0.000 description 1
- 229920002153 Hydroxypropyl cellulose Polymers 0.000 description 1
- 208000013016 Hypoglycemia Diseases 0.000 description 1
- 238000004566 IR spectroscopy Methods 0.000 description 1
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 1
- 229910010082 LiAlH Inorganic materials 0.000 description 1
- 229930195725 Mannitol Natural products 0.000 description 1
- 241001465754 Metazoa Species 0.000 description 1
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 1
- 239000002202 Polyethylene glycol Substances 0.000 description 1
- OFOBLEOULBTSOW-UHFFFAOYSA-N Propanedioic acid Natural products OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 1
- 229940100389 Sulfonylurea Drugs 0.000 description 1
- 229940123464 Thiazolidinedione Drugs 0.000 description 1
- 235000011054 acetic acid Nutrition 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 239000000853 adhesive Substances 0.000 description 1
- 230000001070 adhesive effect Effects 0.000 description 1
- 230000006838 adverse reaction Effects 0.000 description 1
- 239000003888 alpha glucosidase inhibitor Substances 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- 229940024606 amino acid Drugs 0.000 description 1
- 150000001413 amino acids Chemical class 0.000 description 1
- 239000003708 ampul Substances 0.000 description 1
- 229940127003 anti-diabetic drug Drugs 0.000 description 1
- 230000003178 anti-diabetic effect Effects 0.000 description 1
- 229910000019 calcium carbonate Inorganic materials 0.000 description 1
- 235000010216 calcium carbonate Nutrition 0.000 description 1
- FUFJGUQYACFECW-UHFFFAOYSA-L calcium hydrogenphosphate Chemical compound [Ca+2].OP([O-])([O-])=O FUFJGUQYACFECW-UHFFFAOYSA-L 0.000 description 1
- 235000011132 calcium sulphate Nutrition 0.000 description 1
- 238000005119 centrifugation Methods 0.000 description 1
- 238000012512 characterization method Methods 0.000 description 1
- 235000015165 citric acid Nutrition 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- 239000001767 crosslinked sodium carboxy methyl cellulose Substances 0.000 description 1
- 235000010947 crosslinked sodium carboxy methyl cellulose Nutrition 0.000 description 1
- 235000019425 dextrin Nutrition 0.000 description 1
- 235000019700 dicalcium phosphate Nutrition 0.000 description 1
- 239000003603 dipeptidyl peptidase IV inhibitor Substances 0.000 description 1
- 239000007884 disintegrant Substances 0.000 description 1
- 239000007919 dispersible tablet Substances 0.000 description 1
- 230000000225 effect on diabetes Effects 0.000 description 1
- 238000001704 evaporation Methods 0.000 description 1
- 239000010408 film Substances 0.000 description 1
- 239000000796 flavoring agent Substances 0.000 description 1
- 235000013355 food flavoring agent Nutrition 0.000 description 1
- 235000019253 formic acid Nutrition 0.000 description 1
- 239000001530 fumaric acid Substances 0.000 description 1
- 235000011087 fumaric acid Nutrition 0.000 description 1
- 229940116332 glucose oxidase Drugs 0.000 description 1
- 235000019420 glucose oxidase Nutrition 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 230000007686 hepatotoxicity Effects 0.000 description 1
- 231100000304 hepatotoxicity Toxicity 0.000 description 1
- 239000001863 hydroxypropyl cellulose Substances 0.000 description 1
- 235000010977 hydroxypropyl cellulose Nutrition 0.000 description 1
- 239000001866 hydroxypropyl methyl cellulose Substances 0.000 description 1
- 235000010979 hydroxypropyl methyl cellulose Nutrition 0.000 description 1
- 229920003088 hydroxypropyl methyl cellulose Polymers 0.000 description 1
- UFVKGYZPFZQRLF-UHFFFAOYSA-N hydroxypropyl methyl cellulose Chemical compound OC1C(O)C(OC)OC(CO)C1OC1C(O)C(O)C(OC2C(C(O)C(OC3C(C(O)C(O)C(CO)O3)O)C(CO)O2)O)C(CO)O1 UFVKGYZPFZQRLF-UHFFFAOYSA-N 0.000 description 1
- 230000003345 hyperglycaemic effect Effects 0.000 description 1
- 238000001727 in vivo Methods 0.000 description 1
- 239000003978 infusion fluid Substances 0.000 description 1
- 230000002401 inhibitory effect Effects 0.000 description 1
- 230000005764 inhibitory process Effects 0.000 description 1
- 239000008101 lactose Substances 0.000 description 1
- 238000011866 long-term treatment Methods 0.000 description 1
- 229940031703 low substituted hydroxypropyl cellulose Drugs 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 239000008176 lyophilized powder Substances 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- 239000011976 maleic acid Substances 0.000 description 1
- 239000000594 mannitol Substances 0.000 description 1
- 235000010355 mannitol Nutrition 0.000 description 1
- XZWYZXLIPXDOLR-UHFFFAOYSA-N metformin Chemical compound CN(C)C(=N)NC(N)=N XZWYZXLIPXDOLR-UHFFFAOYSA-N 0.000 description 1
- 229960003105 metformin Drugs 0.000 description 1
- 229940102396 methyl bromide Drugs 0.000 description 1
- 229920000609 methyl cellulose Polymers 0.000 description 1
- 239000001923 methylcellulose Substances 0.000 description 1
- 235000010981 methylcellulose Nutrition 0.000 description 1
- 150000007522 mineralic acids Chemical class 0.000 description 1
- 238000010172 mouse model Methods 0.000 description 1
- 229910017604 nitric acid Inorganic materials 0.000 description 1
- 150000007524 organic acids Chemical class 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 230000008520 organization Effects 0.000 description 1
- 235000006408 oxalic acid Nutrition 0.000 description 1
- 238000004806 packaging method and process Methods 0.000 description 1
- 239000002504 physiological saline solution Substances 0.000 description 1
- 229920001223 polyethylene glycol Polymers 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- GCYXWQUSHADNBF-AAEALURTSA-N preproglucagon 78-108 Chemical compound C([C@@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](C)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCCCN)C(=O)NCC(=O)N[C@@H](CCCNC(N)=N)C(=O)NCC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](C)NC(=O)[C@H](C)NC(=O)[C@H](CCC(N)=O)NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](CC=1C=CC=CC=1)NC(=O)[C@@H](NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H](N)CC=1N=CNC=1)[C@@H](C)O)[C@@H](C)O)C(C)C)C1=CC=CC=C1 GCYXWQUSHADNBF-AAEALURTSA-N 0.000 description 1
- 239000003755 preservative agent Substances 0.000 description 1
- 230000004044 response Effects 0.000 description 1
- 230000028327 secretion Effects 0.000 description 1
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical class O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 1
- 239000007779 soft material Substances 0.000 description 1
- 239000000600 sorbitol Substances 0.000 description 1
- 235000010356 sorbitol Nutrition 0.000 description 1
- 230000001954 sterilising effect Effects 0.000 description 1
- YROXIXLRRCOBKF-UHFFFAOYSA-N sulfonylurea Chemical class OC(=N)N=S(=O)=O YROXIXLRRCOBKF-UHFFFAOYSA-N 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 125000005931 tert-butyloxycarbonyl group Chemical group [H]C([H])([H])C(OC(*)=O)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- WROMPOXWARCANT-UHFFFAOYSA-N tfa trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F.OC(=O)C(F)(F)F WROMPOXWARCANT-UHFFFAOYSA-N 0.000 description 1
- 229940124597 therapeutic agent Drugs 0.000 description 1
- 210000003462 vein Anatomy 0.000 description 1
Landscapes
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
The invention relates to the field of diabetes associated medicaments, in particular to an N-cyanomethyl pyrrole derivative with a general formula I, a preparation method, a medical composition containing the same and application thereof in preparing diabetes medicaments, wherein all groups are as defined in the specification.
Description
Technical Field
The invention relates to the field of diabetes-related medicines, in particular to a dipeptidyl peptidase-IV (DPP-IV) inhibitor containing an N-cyanomethylpyrrole structure and having a treatment effect on diabetes, a preparation method thereof and a pharmaceutical composition containing the DPP-IV inhibitor.
Background
According to the statistics of the world health organization, the diabetes patients around the world are about 1.7 hundred million, and about 90 percent of the diabetes patients are type II diabetes patients. The antidiabetic drugs currently used in clinic mainly include insulin, metformin, sulfonylurea drugs, and recently marketed thiazolidinedione drugs, α -glucosidase inhibitors, etc., which have good therapeutic effects, but have safety problems in long-term treatment, such as: it is easy to cause various problems such as hepatotoxicity and weight gain.
Dipeptidyl peptidase IV (DPP-IV) can effectively and rapidly degrade glucagon-like peptide 1 (GLP-1), wherein GLP-1 is one of the most effective stimulators for insulin production and secretion, so that the inhibition of DPP-IV can enhance the action of endogenous GLP-1, thereby increasing the level of insulin in blood (CN 200480017355.6). It has been medically established that DPP-IV Inhibitors are novel antidiabetic therapeutic agents (Deacon C.F., Holst J.J., Dipeptidyl Peptidase IV Inhibitors: A purifying New therapeutic Approach for the Management of Type 2 diabetes, the International journal of Biochemistry & Cell Biology, 2006, 38 (5-6): 831) 844). Clinical results show that the medicine has good hypoglycemic effect, and meanwhile, adverse reactions such as common weight gain, hypoglycemia and the like generated by other diabetes medicines are not found. The present invention has found novel DPP-IV inhibitors which are very effective in lowering plasma glucose levels, which compounds lay the foundation for the further discovery of drugs which can be used in the treatment of diabetes, particularly non-insulin dependent diabetes mellitus.
Disclosure of Invention
It is an object of the present invention to overcome the disadvantages and drawbacks of the prior art and to provide a compound having the general formula I and pharmaceutically acceptable salts thereof with good activity.
It is another object of the present invention to provide a process for the preparation of compounds having the general formula I and pharmaceutically acceptable salts thereof.
It is a further object of the present invention to provide pharmaceutical compositions containing a compound of formula I as an active ingredient, together with one or more pharmaceutically acceptable carriers, excipients or diluents, and their use in the treatment of diabetes.
The present disclosure will now be described in detail for the purpose of the invention.
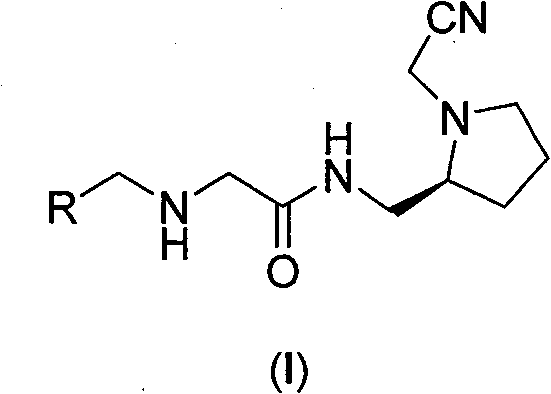
The compounds of the present invention having the general formula I have the following structural formula:
wherein,
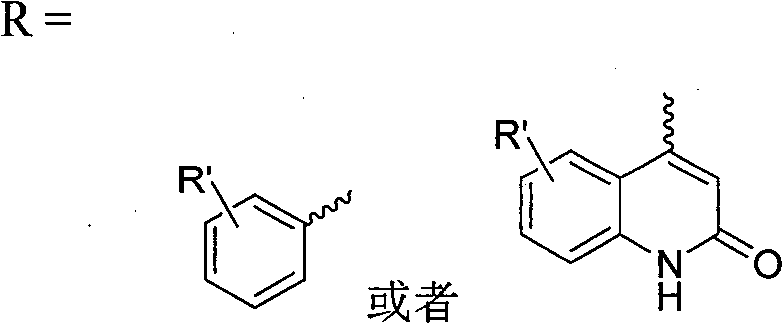
or
R’=H,R1,F,Cl,Br,I,NO2,NR2R3,COOR4,CONR2R3Or R4CO, and combinations thereof di-or tri-substituted, wherein R1Is C1-C8Alkyl of R2,R3And R4 is H or C1-C8Alkyl group of (1).
Preferred are compounds of the following general formula I or pharmaceutically acceptable salts thereof,
wherein,
R’=H,R1,F,Cl,Br,I,NO2,NR2R3,CONR2R3Or R4CO, and combinations thereof di-or tri-substituted, wherein R1Is C1-C4Alkyl of R2,R3And R4 is H or C1-C4Alkyl group of (1).
More preferred compounds of the invention having the general formula I are shown in the following table:
| (Code) | name of Compound |
| I-1 | (S) -2- { { [2- (benzylamino) acetyl]Amino } methyl } -1-cyanomethyl pyrrole |
| I-2 | (S) -1-cyanomethyl-2- { {2- { [ (4-fluorophenyl) methyl]Amino } acetyl } amino } methyl } pyrrole |
| I-3 | (S) -1-cyanomethyl-2- { {2- { [ (1, 2-dihydroquinolin-2-one-4-yl) methyl]Amino } acetyl } amino } methyl } pyrrole |
| I-4 | (S) -2- { {2- { [ (2-chloro-3-methylphenyl) methyl]Amino } acetyl } amino } methyl } -1-cyanomethyl pyrrole |
| I-5 | (S) -2- { {2- { [ (4-bromo-2-formyl-3-methylphenyl) methyl]Amino } acetyl } amino } methyl } -1-cyanomethyl pyrrole |
| I-6 | (S) -1-cyanomethyl-2- { {2- { [ (6-methyl-1, 2-dihydroquinolin-2-one-4-yl) methyl]Amino } acetyl } amino } methyl } pyrrole |
| I-7 | (S) -2- { { [2- (benzylamino) acetyl]Amino } methyl } -1-cyanomethyl pyrrole trifluoroacetate salt |
| I-8 | (S) -1-cyanomethyl-2- { {2- { [ (1, 2-dihydroquinolin-2-one-4-yl) methyl]Amino } acetyl } amino } methyl } pyrrole acetate salt |
The compound of the general formula I is synthesized by the following steps:
reacting the protected glycine PG-Gly-OH II with a compound III in the presence of a condensing agent to obtain a compound IV, wherein PG-Gly-OH II is protected glycine, and PG is a protecting group (such as Boc, Cbz and the like).
And deprotecting the compound IV by a deprotection method corresponding to the protecting group PG to obtain a compound V.
Compounds VI and V in inorganic bases, e.g. Na2CO3And K2CO3In the presence of a catalyst to obtain the compound I. The compound I reacts with acid HA at room temperature to obtain a compound I-s. Wherein R is as defined above.
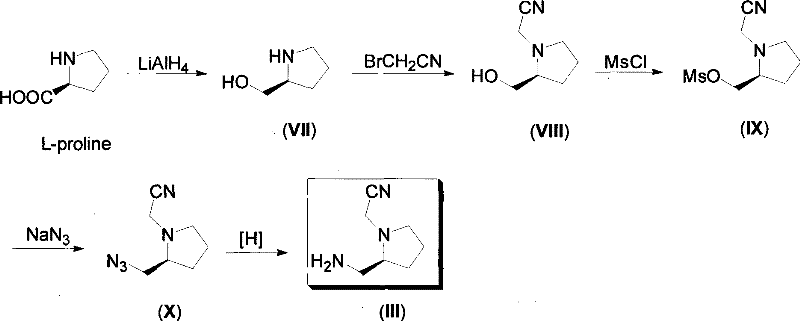
Wherein the compound III in the above route is synthesized by the following route:
the starting material is L-proline. L-proline is subjected to LiAlH4And reducing to obtain a compound VII. Compound VII with bromoacetonitrile in an inorganic base, e.g. Na2CO3And K2CO3And reacting in the presence of the catalyst to obtain the compound VIII. The compound VIII is reacted with methanesulfonyl chloride MsCl in the presence of an organic base such as triethylamine and the like to obtain a compound IX. Compounds IX and NaN3Heating and reflecting in an aprotic dipolar solvent to obtain a compound X. And catalytically hydrogenating the compound X at normal pressure to obtain a compound III.
The pharmaceutically acceptable salts of the compounds of formula I of the present invention include, but are not limited to, pharmaceutically acceptable salts formed with various inorganic acids, such as hydrochloric acid, sulfuric acid, nitric acid, phosphoric acid, and the like, or organic acids, such as formic acid, acetic acid, citric acid, oxalic acid, fumaric acid, maleic acid, amino acids, and the like.
The compound of formula I or the pharmaceutically acceptable salt thereof can be prepared into a pharmaceutical composition together with one or more pharmaceutically acceptable carriers, excipients or diluents. The pharmaceutical composition can be made into solid oral preparation, liquid oral preparation, injection, etc. The solid and liquid oral formulations comprise: tablet, dispersible tablet, sugar-coated preparation, granule, dry powder, capsule and solution. The injection comprises: small needle, large infusion solution, lyophilized powder for injection, etc.
The composition of the invention, the pharmaceutically or dietetically acceptable auxiliary materials are selected from: fillers, disintegrants, lubricants, glidants, effervescent agents, flavoring agents, preservatives, coating materials, or other excipients.
The composition of the invention, and the pharmaceutically or dietetically acceptable auxiliary materials. The filler is one or more of lactose, sucrose, dextrin, starch, pregelatinized starch, mannitol, sorbitol, calcium hydrogen phosphate, calcium sulfate, calcium carbonate, and microcrystalline cellulose; the adhesive comprises one or a combination of more of sucrose, starch, polyvidone, sodium carboxymethylcellulose, hydroxypropyl methylcellulose, hydroxypropyl cellulose, methylcellulose, polyethylene glycol, medicinal ethanol and water; the disintegrating agent comprises one or more of starch, cross-linked polyvidone, cross-linked sodium carboxymethyl cellulose, low-substituted hydroxypropyl cellulose, sodium carboxymethyl cellulose, and effervescent disintegrating agent.
The compound of the general formula I or the salt thereof has the DPP-IV inhibiting effect and can be used as an active ingredient for preparing a medicament for treating diabetes. The activity of the compound of the general formula I is verified by an in vivo hypoglycemic model.
The compounds of formula I of the present invention are effective over a relatively wide dosage range. For example, the daily dosage may be in the range of about 1mg to about 1000mg per person, divided into one or more administrations. The actual dosage of the compounds of formula I to be administered according to the invention can be determined by the physician in the light of the relevant circumstances. These include: the physical state of the subject, the route of administration, the age, body weight, individual response to the drug, severity of the symptoms, and the like.
Detailed Description
The present invention will be further described with reference to the following examples. It should be noted that the following examples are only for illustration and are not intended to limit the present invention. Variations of the teachings of the present invention may be made by those skilled in the art without departing from the scope of the claims of the present application.
Instruments and reagents unless otherwise specified, were commercially available analytical grade pure reagents and infrared spectroscopy was performed by a Fourier transform infrared spectrometer (KBr pellet or pure film method) of ThermoNicolet AVATAR FT370 USA.
Example 1
Synthesis of (S) -2- { {2- [ (t-butoxycarbonyl) amino ] acetyl } amino } methyl-1-cyanomethylpyrrole (IV-a)
A100 mL round-bottomed flask was charged with 1.75g (10mmol) of Boc-protected glycine II-a, dissolved in 20mL anhydrous THF, followed by addition of 2.06g (10mmol) of DCC, stirring at room temperature for 1 hour, followed by addition of 1.39g (10mmol) of Compound III, and the resulting system was stirred at room temperature overnight. The reaction mixture was filtered to remove solidsThe solvent was evaporated from the filtrate on a rotary evaporator to give a residue, which was subjected to column chromatography to give the product IV-a, 2.66g, yield 90%, as a colorless oily liquid. IR (thin film), 3321, 3217, 2228, 1690, 1684cm-1.
The compound II-a is one of the compounds II having the general formula, and in this case IV-a is also one of the compounds having the general formula IV. The protective group PG is then Boc (t-Butoxycarbonyl) as tert-butyloxycarbonyl.
Example 2
Synthesis of (S) -2- { [2- (amino) acetyl ] amino } methyl-1-cyanomethylpyrrole (V)
A100 mL reaction flask was charged with 2.96g (10mmol) of Compound IV-a as 20mL CH2Cl2Dissolve, add 1mL of trifluoroacetic acid (TFA) with stirring at room temperature, and then continue stirring at room temperature for 2 hours. The reaction system was poured with 100mL of 2% Na2CO3In solution, stir, use 50mL × 3 CH2Cl2Extracting, mixing the organic phases, washing with saturated saline solution, and drying (Na)2SO4) The solvent was evaporated on a rotary evaporator to give pure compound V as a colorless oily liquid, 1.90g, 97% yield IR (thin film), 3321, 3313, 2226, 1686cm-1.
Example 3
(S) -2- { { [2- (benzylamino) acetyl ] amino } methyl } -1-cyanomethylpyrrole (1-1)
A100 mL round-bottomed flask was charged with 1.71g (10mmol) of Compound VI-1, 1.96g (10mmol) of Compound V and 1.06g (10mmol) of solid Na2CO3To do soThen add 10mL MeOH and 10mL CH2Cl2. The system was stirred at room temperature overnight. The solid present in the reaction system was removed by filtration, the solvent was evaporated from the filtrate on a rotary evaporator, and the obtained residue was purified by column chromatography to give the product I-1 as a colorless oil, 2.55g, in 89% yield. IR (thin film), 3030, 3332, 3251, 2229, 1687cm-1.
Compound VI-1 is one of the compounds having the general formula VI.
Examples 4 to 7
The same procedure as in example 3, except that VI in the following table was used in place of VI-1 in example 3, and the other procedure was the same as in example 3, gave the compounds I-2 to I-5 in the following table.
| Example number | Yield/%) | VI | I |
| 4 | 90 | 4-Fluorobenzyl Bromide | I-2: (S) -1-cyanomethyl-2- { {2- { [ (4-fluorophenyl) methyl]Amino } acetyl } amino } methyl } pyrrole |
| 5 | 89 | (1, 2-dihydroquinolin-2-one-4-yl) methyl bromide | I-3: (S) -1-cyanomethyl-2- { {2- { [ (1, 2-dihydroquinolin-2-one-4-yl) methyl]Amino } acetyl } amino } methyl } pyrrole |
| 6 | 87 | 2-chloro-3-methylbenzyl bromide | I-4: (S) -2- { {2- { [ (2-chloro-3-methylphenyl) methyl]Amino } acetyl } amino } methyl } -1-cyanomethyl pyrrole |
| 7 | 90 | 4-bromo-2-formyl-3-methylphenylbenzyl bromide | I-5: (S) -2- { {2- { [ (4-bromo-2-formyl-3-methylphenyl) methyl]Amino } acetyl } amino } methyl } -1-cyanomethyl pyrrole |
| 8 | 88 | (6-methyl-1, 2- | I-6: (S) -1-cyanomethyl-2- { {2- { [ (6-methyl-1, 2-dihydro |
| Dihydroquinolin-2-one-4-yl) methyl bromide | Quinolin-2-one-4-yl) methyl]Amino } acetyl } amino } methyl } pyrrole |
The characterization data for compound I in the table above are as follows:
i-2: (S) -1-cyanomethyl-2- { {2- { [ (4-fluorophenyl) methyl]Amino } acetyl } amino } methyl } pyrrole, colourless oil, IR (thin film), 3033, 3335, 3253, 2229, 1688cm-1.
I-3: (S) -1-cyanomethyl-2- { {2- { [ (1, 2-dihydroquinolin-2-one-4-yl) methyl]Amino } acetyl } amino } methyl } pyrrole, colourless oil, IR (thin film), 3031, 3329, 3251, 2228, 1684, 1663cm-1.
I-4: (S) -2- { {2- { [ (2-chloro-3-methylphenyl) methyl]Amino } acetyl } amino } methyl } -1-cyanomethyl pyrrole, colorless oil, IR (thin film), 3035, 3322, 3253, 2229, 1682cm-1.
I-5: (S) -2- { {2- { [ (4-bromo-2-formyl-3-methylphenyl) methyl]Amino } acetyl } amino } methyl } -1-cyanomethyl pyrrole, colorless oil, IR (thin film), 3039, 3324, 3255, 2227, 1702, 1686cm-1.
I-6: (S) -1-cyanomethyl-2- { {2- { [ (6-methyl-1, 2-dihydroquinolin-2-one-4-yl) methyl]Amino } acetyl } amino } methyl } pyrrole, colourless oil, IR (thin film), 3039, 3324, 3255, 2227, 1702, 1686, 1655cm-1.
Example 9
Synthesis of (S) -2-hydroxymethylpyrrole (VII)
A500 mL round bottom flask was charged with 11.5g (0.1mol) of solid L-proline and 200mL of dry THF, followed by 7.6g (0.2mol) of solid LiAlH in portions with ice-cooling4And then stirred at room temperature overnight. Slowly dropwise adding 50mL of anhydrous methanol, stirring for half an hour, suction-filtering to remove solids, evaporating the solvent from the filtrate on a rotary evaporator to obtain an oil which is the compound VII, 8.6g, 85% yield IR (thin film), 3451, 3325cm-1.
Example 10
Synthesis of (S) -1-cyanomethyl-2-hydroxymethylpyrrole (VIII)
A250 mL round bottom flask was charged with 10.1g (0.1mol), 12.0g (0.1mol) and 10.6g (0.1mol) of solid Na2CO3Then, 100mL of anhydrous MeOH was added and the reaction was stirred at room temperature overnight. The solid was removed by suction filtration and the filtrate was freed of solvent on a rotary evaporator to give product VIII as a colourless oil, 12.0g, 0.86%. IR (thin film), 3454, 2229cm-1.
Example 11
Synthesis of (S) -1-cyanomethyl-2-methanesulfonyloxymethylpyrrole (IX)
14.0g (0.1mol) of Compound VIII was dissolved in 50mL of ethyl acetate, 11.1g (0.1mol) of triethylamine was added thereto, the mixture was stirred at room temperature, 11.5g (0.1mol) of methanesulfonyl chloride was slowly dropped, and after completion of the dropping, the reaction system was stirred at room temperature overnight. The reaction mixture was washed once with saturated brine in a separatory funnel and dried (Na)2SO4) The solvent was evaporated on a rotary evaporator to give the residue product IX as a colorless oily liquid, 20.3g, 93% yield. IR (thinfilm), 2228cm-1.
Example 12
Synthesis of (S) -2-azidomethyl-1-cyanomethylpyrrole (IX)
21.8g (0.1mol) of Compound IX are dissolved in 60mL of dry DMF, 19.5g (0.3mol) of solid NaN are added3The reaction was stirred at 80 ℃ overnight. The system was poured into 500mL of water and 70mL of 3 CH was used2Cl2Extracting, mixing the organic phases, washing with saturated saline solution, and drying (Na)2SO4) The solvent was evaporated on a rotary evaporator to give Compound X as a colorless oily liquid, 13.9g, 84% yield IR (thin film), 2241, 2226cm-1.
Example 13
Synthesis of (S) -2-aminomethyl-1-cyanomethylpyrrole (III)
16.5g (0.1mol) of Compound X were dissolved in 60mL of anhydrous methanol, and 1.0g of 5% Pd/C was added, followed by hydrogenation overnight according to a standard hydrogenation method. The catalyst was removed by suction filtration and the filtrate was freed of solvent on a rotary evaporator to give product III as a colourless oil, 11.8g, 85% yield. IR (thin film), 3323, 3312, 2229cm-1.
Example 14
Synthesis of (S) -2- { { [2- (benzylamino) acetyl ] amino } methyl } -1-cyanomethylpyrrole trifluoroacetate (I-7)
2.86g (10mmol) of Compound I-1 were dissolved in 20mL of dry ethyl acetate, followed by addition of 1.14g (10mmol) of trifluoroacetic acid and stirring at room temperature for 1 hour. The crystals were collected by filtration and dried to give the product I-7 as colorless crystals, 3.60g, 90% yield. IR (KBr), 3030, 2543, 2226, 1683cm-1。
Example 15
Synthesis of (S) -1-cyanomethyl-2- { {2- { [ (1, 2-dihydroquinolin-2-one-4-yl) methyl ] amino } acetyl } amino } methyl } pyrrole acetate (I-8)
The same operation as in example 14, except for replacing I-1 in example 14 with I-3 and replacing trifluoroacetic acid in example 14 with acetic acid, was carried out in the same manner as in example 14 to obtain compound I-8.
I-8: (S) -1-cyanomethyl-2- { {2- { [ (1, 2-dihydroquinolin-2-one-4-yl) methyl]Amino } acetyl } aminoMethyl pyrrole acetate, colorless crystals, 91% yield, IR (KBr), 3031, 2551, 2228, 1683, 1664cm-1。
Example 16
Dosage/tablet
Example 5 sample (I-3) 100mg
Microcrystalline cellulose 80mg
Pregelatinized starch 70mg
Polyvinylpyrrolidone 6mg
Carboxymethyl starch sodium salt 5mg
Magnesium stearate 2mg
Talcum powder 2mg
Sieving active ingredients, pregelatinized starch and microcrystalline cellulose, mixing, adding polyvinylpyrrolidone solution, mixing, making soft mass, sieving, making wet granule, drying at 50-60 deg.C, sieving carboxymethyl starch sodium salt, magnesium stearate and pulvis Talci, adding into the above granule, and tabletting.
Example 17
A pellet was obtained in the same manner as in example 16 except that the sample (I-1) obtained in example 3 was used in place of I-3 in example 16 in the procedure of example 16.
Example 18
Dosage/granule
EXAMPLE 6 sample (I-4) 50mg
Microcrystalline cellulose 30mg
Pregelatinized starch 20mg
Polyvinylpyrrolidone 3mg
Magnesium stearate 2mg
Talcum powder 1mg
Sieving active ingredients, pregelatinized starch and microcrystalline cellulose, mixing, adding polyvinylpyrrolidone solution, mixing, making soft material, sieving, making wet granule, drying at 50-60 deg.C, sieving magnesium stearate and pulvis Talci, adding into the above granule, and making into capsule.
Example 19
Referring to the procedure of example 18, a capsule was obtained in the same manner as in example 18 except that I-4 in example 18 was replaced with the sample (I-2) of example 4.
Example 20
The dosage is 50ml
Example 15 sample 50mg
Citric acid 100mg
Appropriate amount of NaOH (adjusting pH4.0-5.0)
50ml of distilled water
Adding distilled water and citric acid into distilled water, stirring for dissolving, adding sample, slightly heating for dissolving, adjusting pH to 4.0-5.0, adding 0.2 g of activated carbon, stirring at room temperature for 20 min, filtering, measuring solution concentration by central control, packaging at 5 ml per ampoule, and sterilizing at high temperature for 30 min to obtain injection.
Example 21
The samples were prepared as a 5mg/ml suspension in 1% sodium carboxymethylcellulose in a dose capacity of 0.4ml/20g body weight, corresponding to a 100mg/kg dose. Damekang is administered in a dose of about 80mg/kg, formulated with 1% sodium carboxymethylcellulose to a concentration of about 4mg/ml, and administered in a volume of about 0.4ml/20g of body weight. The alloxan is prepared into 15mg/ml with fresh physiological saline when being injected. 0.1ml/20g body weight, corresponding to 75 mg/kg.
Healthy ICR mice, each half of male and female, weigh 20-24 g, and meet the first-class standard. Animals were fasted for 16 hours and then injected tail vein with alloxan. After fasting for 6 hours after 48 hours, blood was taken from the retrobulbar venous plexus of mice by capillary, and serum was separated by centrifugation and the glucose content of serum was measured by glucose oxidase method. Selecting mice with blood glucose value higher than 300mg/dl, and dividing into 4 groups according to the measured blood glucose content, wherein the groups are respectively a model group, a positive group (80 mg/kg of Dameikang) group and an administration group. The administration is carried out for 3 days, fasting is carried out for 24 hours, blood is taken 1 hour after the last administration, and the blood sugar content is determined. The results are shown in the table
Influence on blood sugar content of mice model hyperglycemic by alloxan
Note: p <0.01 in comparison to model group
Claims (11)
3. A compound of general formula I as defined in claim 1, or a pharmaceutically acceptable salt thereof, selected from:
(S) -2- { { [2- (benzylamino) acetyl ] amino } methyl } -1-cyanomethylpyrrolidine
(S) -1-cyanomethyl-2- { {2- { [ (4-fluorophenyl) methyl ] amino } acetyl } amino } methyl } pyrrolidine
(S) -1-cyanomethyl-2- { {2- { [ (1, 2-dihydroquinolin-2-one-4-yl) methyl ] amino } acetyl } amino } methyl } pyrrolidine
(S) -1-cyanomethyl-2- { {2- { [ (6-methyl-1, 2-dihydroquinolin-2-one-4-yl) methyl ] amino } acetyl } amino } methyl } pyrrolidine
(S) -2- { { [2- (benzylamino) acetyl ] amino } methyl } -1-cyanomethylpyrrolidine trifluoroacetate salt
(S) -1-cyanomethyl-2- { {2- { [ (1, 2-dihydroquinolin-2-one-4-yl) methyl ] amino } acetyl } amino } methyl } pyrrolidine acetate.
4. A compound, or a pharmaceutically acceptable salt thereof, selected from: (S) -2- { {2- { [ (2-chloro-3-methylphenyl) methyl ] amino } acetyl } amino } methyl } -1-cyanomethyl-pyrrolidine
(S) -2- { {2- { [ (4-bromo-2-formyl-3-methylphenyl) methyl ] amino } acetyl } amino } methyl } -1-cyanomethylpyrrolidine.
5. A method of synthesizing a compound according to any one of claims 1 to 4, or a pharmaceutically acceptable salt thereof, comprising the steps of:
reacting compound II with compound III in the presence of a condensing agent to obtain compound IV, wherein compound IIPG-Gly-OH is protected glycine, and PG is protecting groups Boc and Cbz;
deprotecting the compound IV by a deprotection method corresponding to the protecting group PG to obtain a compound V;
reacting the compound VI with the compound V in the presence of inorganic base to obtain a compound I; the compound I reacts with acid HA at room temperature to obtain a compound I-s.
6. The process of claim 5, wherein compound VI and compound V are reacted in the presence of an inorganic base selected from Na to obtain compound I2CO3、K2CO3Any one of them.
7. The method of claim 5 or 6, wherein III is synthesized by the following route:
the starting material is L-proline; reducing the L-proline by LiAlH4 to obtain a compound VII; reacting the compound VII with bromoacetonitrile in the presence of inorganic base to obtain a compound VIII; reacting the compound VIII with methanesulfonyl chloride MsCl in the presence of organic base triethylamine to obtain a compound IX; compounds IX and NaN3Heating and reacting in an aprotic dipolar solvent to obtain a compound X; and catalytically hydrogenating the compound X at normal pressure to obtain a compound III.
8. The process of claim 7, wherein compound VII is reacted with bromoacetonitrile in the presence of an inorganic base selected from Na to obtain compound VIII2CO3、K2CO3Any one of them.
9. A compound according to any one of claims 1 to 4, or a pharmaceutically acceptable salt thereof, for use in the manufacture of a medicament for the treatment of diabetes.
10. A pharmaceutical composition comprising a compound according to any one of claims 1 to 4, or a pharmaceutically acceptable salt thereof, and a suitable carrier.
11. The pharmaceutical composition of claim 10, wherein the composition is a solid oral formulation, a liquid oral formulation or an injection.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2008101515014A CN101684088B (en) | 2008-09-22 | 2008-09-22 | Cyanomethyl pyrrole derivative and preparation method and application thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2008101515014A CN101684088B (en) | 2008-09-22 | 2008-09-22 | Cyanomethyl pyrrole derivative and preparation method and application thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN101684088A CN101684088A (en) | 2010-03-31 |
| CN101684088B true CN101684088B (en) | 2011-05-11 |
Family
ID=42047504
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN2008101515014A Expired - Fee Related CN101684088B (en) | 2008-09-22 | 2008-09-22 | Cyanomethyl pyrrole derivative and preparation method and application thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN101684088B (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2025158275A1 (en) | 2024-01-24 | 2025-07-31 | Pfizer Inc. | Combination therapy using glucose-dependent insulinotropic polypeptide receptor antagonist compounds and glp-1 receptor agonist compounds |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN108752255A (en) * | 2018-07-19 | 2018-11-06 | 重庆医科大学 | A kind of preparation method of pabishta and its key intermediate |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1377879A (en) * | 2001-03-28 | 2002-11-06 | 瑟维尔实验室 | New alpha-amino acid sulfuryl compounds, its preparing method and medicinal composition containing them |
| CN101005835A (en) * | 2004-06-08 | 2007-07-25 | 桑赛拉制药(瑞士)股份公司 | 1-[(3r)-amino-4-(2-fluoro-phenyl)-butyl]-pyrrolidine-(2r)-carboxylic acid-benzyl amine derivatives and related compounds as dipeptidyl-peptidase iv (dpp-iv) inhibitors for the treatment of type 2 diab |
| CN101087756A (en) * | 2004-07-23 | 2007-12-12 | 纽阿达有限责任公司 | Peptidase inhibitors |
-
2008
- 2008-09-22 CN CN2008101515014A patent/CN101684088B/en not_active Expired - Fee Related
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1377879A (en) * | 2001-03-28 | 2002-11-06 | 瑟维尔实验室 | New alpha-amino acid sulfuryl compounds, its preparing method and medicinal composition containing them |
| CN101005835A (en) * | 2004-06-08 | 2007-07-25 | 桑赛拉制药(瑞士)股份公司 | 1-[(3r)-amino-4-(2-fluoro-phenyl)-butyl]-pyrrolidine-(2r)-carboxylic acid-benzyl amine derivatives and related compounds as dipeptidyl-peptidase iv (dpp-iv) inhibitors for the treatment of type 2 diab |
| CN101087756A (en) * | 2004-07-23 | 2007-12-12 | 纽阿达有限责任公司 | Peptidase inhibitors |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2025158275A1 (en) | 2024-01-24 | 2025-07-31 | Pfizer Inc. | Combination therapy using glucose-dependent insulinotropic polypeptide receptor antagonist compounds and glp-1 receptor agonist compounds |
Also Published As
| Publication number | Publication date |
|---|---|
| CN101684088A (en) | 2010-03-31 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP4125896B1 (en) | Treatment of type 2 diabetes with 2-[(4-{6-[(4-cyano-2-fluorobenzyl)oxy]pyridin-2-yl} piperidin-1-yl)methyl]-1-[(2s)-oxetan-2-ylmethyl]-1h-benzimidazole-6-carboxylic acid or a pharmaceutically salt thereof | |
| CN101445527B (en) | Five-membered heteroaromatics tolylene glucoside and preparation method and use thereof | |
| CN101445492B (en) | Amidothiazole derivatives, preparation method and application thereof | |
| EP2530079A1 (en) | Phenyl c-glucoside derivatives, preparation methods and uses thereof | |
| CN101508712A (en) | Glucoside containing tetrazole structure, preparation method and application | |
| CN103626825B (en) | The glycogen phosphorylase inhibitors cholic acid derivative of target liver, its preparation method and medicinal use | |
| WO2008061456A1 (en) | The folacin-metformin compound and its manufacture | |
| EP4585593A1 (en) | Polymorph of glp-1r agonist compound, preparation method therefor, and use thereof | |
| CN103058972B (en) | Phenyl C-glucoside derivatives containing cyclohexane structure as well as preparation method and application thereof | |
| CN103626826B (en) | Containing the glycogen phosphorylase inhibitors cholic acid derivative of azo bond, its preparation method and medicinal use | |
| KR20230024976A (en) | Indene compounds, pharmaceutical compositions thereof, and therapeutic applications thereof | |
| CN113831336A (en) | Praziquantel and ferulic acid eutectic compound, preparation method, composition and application thereof | |
| CN101684103B (en) | Compound with 1,2,4-triazole structure and preparation method and application thereof | |
| CN101684088B (en) | Cyanomethyl pyrrole derivative and preparation method and application thereof | |
| CN102659813B (en) | 2-((2-(3-aminopiperidine-1)-4-oxythiophene (3, 2-d) pyrimidine-3(4H)-methyl) polymorphic benzonitrile, and preparation method and pharmacological applications thereof | |
| TWI608006B (en) | Novel crystalline form | |
| CN101445528B (en) | Sulpho-glucosan derivative and preparation method and application thereof | |
| CN101550112B (en) | 4,5-disubstituted thiazole derivative, preparation method and use thereof | |
| CN118420621A (en) | Sulfonamide polycyclic compound and preparation method and application thereof | |
| CN115073331A (en) | Metformin cinnamate monohydrate, preparation method, composition and application thereof | |
| CN114671839B (en) | Dapagliflozin solid form compound and preparation method and application thereof | |
| CN118420586A (en) | Dihydroquercetin and picolinic acid cocrystal, preparation method, pharmaceutical composition and use thereof | |
| CN102241644A (en) | Alpha-azyl-3-aryl propionamido thiazole derivative, preparation method and purpose thereof | |
| JP2019527708A (en) | Crystals of DPP-IV long-acting inhibitor and salts thereof | |
| CN109796455B (en) | Salt of aminopyrane derivative, crystal form of salt, preparation method and application of salt |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| CF01 | Termination of patent right due to non-payment of annual fee | ||
| CF01 | Termination of patent right due to non-payment of annual fee |
Granted publication date: 20110511 Termination date: 20160922 |