CN101560226B - Novel phosphorous compound, its production method and use - Google Patents
Novel phosphorous compound, its production method and use Download PDFInfo
- Publication number
- CN101560226B CN101560226B CN 200810094205 CN200810094205A CN101560226B CN 101560226 B CN101560226 B CN 101560226B CN 200810094205 CN200810094205 CN 200810094205 CN 200810094205 A CN200810094205 A CN 200810094205A CN 101560226 B CN101560226 B CN 101560226B
- Authority
- CN
- China
- Prior art keywords
- formula
- compound
- epoxy resin
- acid
- dopo
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
- 150000003018 phosphorus compounds Chemical class 0.000 title description 2
- 238000004519 manufacturing process Methods 0.000 title 1
- 150000001875 compounds Chemical class 0.000 claims abstract description 107
- 239000003822 epoxy resin Substances 0.000 claims abstract description 54
- 229920000647 polyepoxide Polymers 0.000 claims abstract description 54
- 238000000034 method Methods 0.000 claims abstract description 32
- RNFJDJUURJAICM-UHFFFAOYSA-N 2,2,4,4,6,6-hexaphenoxy-1,3,5-triaza-2$l^{5},4$l^{5},6$l^{5}-triphosphacyclohexa-1,3,5-triene Chemical compound N=1P(OC=2C=CC=CC=2)(OC=2C=CC=CC=2)=NP(OC=2C=CC=CC=2)(OC=2C=CC=CC=2)=NP=1(OC=1C=CC=CC=1)OC1=CC=CC=C1 RNFJDJUURJAICM-UHFFFAOYSA-N 0.000 claims abstract description 22
- 239000003063 flame retardant Substances 0.000 claims abstract description 22
- 239000004848 polyfunctional curative Substances 0.000 claims abstract description 18
- 229910052698 phosphorus Inorganic materials 0.000 claims abstract description 16
- 239000011574 phosphorus Substances 0.000 claims abstract description 14
- -1 phosphorus compound Chemical class 0.000 claims abstract description 9
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 35
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 claims description 32
- 239000003377 acid catalyst Substances 0.000 claims description 29
- GFUCMNMXYOVTDJ-UHFFFAOYSA-N 2,4-diamino-6-butan-2-ylphenol Chemical compound CCC(C)C1=CC(N)=CC(N)=C1O GFUCMNMXYOVTDJ-UHFFFAOYSA-N 0.000 claims description 22
- PAYRUJLWNCNPSJ-UHFFFAOYSA-N Aniline Chemical compound NC1=CC=CC=C1 PAYRUJLWNCNPSJ-UHFFFAOYSA-N 0.000 claims description 22
- 102100035915 D site-binding protein Human genes 0.000 claims description 22
- 101000873522 Homo sapiens D site-binding protein Proteins 0.000 claims description 22
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 18
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 claims description 16
- 239000003054 catalyst Substances 0.000 claims description 16
- RNVCVTLRINQCPJ-UHFFFAOYSA-N o-toluidine Chemical compound CC1=CC=CC=C1N RNVCVTLRINQCPJ-UHFFFAOYSA-N 0.000 claims description 13
- RXNYJUSEXLAVNQ-UHFFFAOYSA-N 4,4'-Dihydroxybenzophenone Chemical compound C1=CC(O)=CC=C1C(=O)C1=CC=C(O)C=C1 RXNYJUSEXLAVNQ-UHFFFAOYSA-N 0.000 claims description 12
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Phenol Chemical compound OC1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 claims description 12
- ZLSMCQSGRWNEGX-UHFFFAOYSA-N bis(4-aminophenyl)methanone Chemical compound C1=CC(N)=CC=C1C(=O)C1=CC=C(N)C=C1 ZLSMCQSGRWNEGX-UHFFFAOYSA-N 0.000 claims description 11
- NXXYKOUNUYWIHA-UHFFFAOYSA-N 2,6-Dimethylphenol Chemical compound CC1=CC=CC(C)=C1O NXXYKOUNUYWIHA-UHFFFAOYSA-N 0.000 claims description 10
- 125000000753 cycloalkyl group Chemical group 0.000 claims description 10
- 125000001188 haloalkyl group Chemical group 0.000 claims description 10
- 125000005843 halogen group Chemical group 0.000 claims description 10
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 claims description 8
- ZLUJJCPTVKQVDE-UHFFFAOYSA-N (4-aminophenyl)-(4-hydroxyphenyl)methanone Chemical compound C1=CC(N)=CC=C1C(=O)C1=CC=C(O)C=C1 ZLUJJCPTVKQVDE-UHFFFAOYSA-N 0.000 claims description 7
- UFFBMTHBGFGIHF-UHFFFAOYSA-N 2,6-dimethylaniline Chemical compound CC1=CC=CC(C)=C1N UFFBMTHBGFGIHF-UHFFFAOYSA-N 0.000 claims description 7
- QWVGKYWNOKOFNN-UHFFFAOYSA-N o-cresol Chemical compound CC1=CC=CC=C1O QWVGKYWNOKOFNN-UHFFFAOYSA-N 0.000 claims description 7
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 claims description 6
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 claims description 6
- 125000000951 phenoxy group Chemical group [H]C1=C([H])C([H])=C(O*)C([H])=C1[H] 0.000 claims description 6
- DTQVDTLACAAQTR-UHFFFAOYSA-N Trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 claims description 4
- 229910000147 aluminium phosphate Inorganic materials 0.000 claims description 4
- VSCWAEJMTAWNJL-UHFFFAOYSA-K aluminium trichloride Chemical compound Cl[Al](Cl)Cl VSCWAEJMTAWNJL-UHFFFAOYSA-K 0.000 claims description 4
- 239000004841 bisphenol A epoxy resin Substances 0.000 claims description 4
- 238000002156 mixing Methods 0.000 claims description 4
- 239000000203 mixture Substances 0.000 claims description 4
- 239000004843 novolac epoxy resin Substances 0.000 claims description 4
- ZMCHBSMFKQYNKA-UHFFFAOYSA-N 2-aminobenzenesulfonic acid Chemical compound NC1=CC=CC=C1S(O)(=O)=O ZMCHBSMFKQYNKA-UHFFFAOYSA-N 0.000 claims description 3
- CPELXLSAUQHCOX-UHFFFAOYSA-N Hydrogen bromide Chemical compound Br CPELXLSAUQHCOX-UHFFFAOYSA-N 0.000 claims description 3
- 239000002994 raw material Substances 0.000 claims description 3
- KRHYYFGTRYWZRS-UHFFFAOYSA-N Fluorane Chemical compound F KRHYYFGTRYWZRS-UHFFFAOYSA-N 0.000 claims description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 claims description 2
- 229910021578 Iron(III) chloride Inorganic materials 0.000 claims description 2
- 239000002841 Lewis acid Substances 0.000 claims description 2
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 claims description 2
- 239000002253 acid Substances 0.000 claims description 2
- WTEOIRVLGSZEPR-UHFFFAOYSA-N boron trifluoride Chemical compound FB(F)F WTEOIRVLGSZEPR-UHFFFAOYSA-N 0.000 claims description 2
- ZFXVRMSLJDYJCH-UHFFFAOYSA-N calcium magnesium Chemical compound [Mg].[Ca] ZFXVRMSLJDYJCH-UHFFFAOYSA-N 0.000 claims description 2
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 claims description 2
- 239000003153 chemical reaction reagent Substances 0.000 claims description 2
- XMBWDFGMSWQBCA-UHFFFAOYSA-N hydrogen iodide Chemical compound I XMBWDFGMSWQBCA-UHFFFAOYSA-N 0.000 claims description 2
- RBTARNINKXHZNM-UHFFFAOYSA-K iron trichloride Chemical compound Cl[Fe](Cl)Cl RBTARNINKXHZNM-UHFFFAOYSA-K 0.000 claims description 2
- 150000007517 lewis acids Chemical class 0.000 claims description 2
- 229910017604 nitric acid Inorganic materials 0.000 claims description 2
- 125000004430 oxygen atom Chemical group O* 0.000 claims description 2
- XJDNKRIXUMDJCW-UHFFFAOYSA-J titanium tetrachloride Chemical compound Cl[Ti](Cl)(Cl)Cl XJDNKRIXUMDJCW-UHFFFAOYSA-J 0.000 claims description 2
- DWSWCPPGLRSPIT-UHFFFAOYSA-N benzo[c][2,1]benzoxaphosphinin-6-ium 6-oxide Chemical compound C1=CC=C2[P+](=O)OC3=CC=CC=C3C2=C1 DWSWCPPGLRSPIT-UHFFFAOYSA-N 0.000 claims 8
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 claims 2
- HVBSAKJJOYLTQU-UHFFFAOYSA-N 4-aminobenzenesulfonic acid Chemical compound NC1=CC=C(S(O)(=O)=O)C=C1 HVBSAKJJOYLTQU-UHFFFAOYSA-N 0.000 claims 1
- 229910015900 BF3 Inorganic materials 0.000 claims 1
- ZOXJGFHDIHLPTG-UHFFFAOYSA-N Boron Chemical compound [B] ZOXJGFHDIHLPTG-UHFFFAOYSA-N 0.000 claims 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 claims 1
- 150000007513 acids Chemical class 0.000 claims 1
- 229910052796 boron Inorganic materials 0.000 claims 1
- 229910000042 hydrogen bromide Inorganic materials 0.000 claims 1
- 229910000041 hydrogen chloride Inorganic materials 0.000 claims 1
- IXCSERBJSXMMFS-UHFFFAOYSA-N hydrogen chloride Substances Cl.Cl IXCSERBJSXMMFS-UHFFFAOYSA-N 0.000 claims 1
- 229910000040 hydrogen fluoride Inorganic materials 0.000 claims 1
- 229910000043 hydrogen iodide Inorganic materials 0.000 claims 1
- 229940098779 methanesulfonic acid Drugs 0.000 claims 1
- DVECLMOWYVDJRM-UHFFFAOYSA-N pyridine-3-sulfonic acid Chemical compound OS(=O)(=O)C1=CC=CN=C1 DVECLMOWYVDJRM-UHFFFAOYSA-N 0.000 claims 1
- 229950000244 sulfanilic acid Drugs 0.000 claims 1
- FEONEKOZSGPOFN-UHFFFAOYSA-K tribromoiron Chemical compound Br[Fe](Br)Br FEONEKOZSGPOFN-UHFFFAOYSA-K 0.000 claims 1
- 238000002360 preparation method Methods 0.000 abstract description 2
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 48
- 238000006243 chemical reaction Methods 0.000 description 35
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 27
- 230000015572 biosynthetic process Effects 0.000 description 27
- 238000003786 synthesis reaction Methods 0.000 description 27
- 230000009477 glass transition Effects 0.000 description 25
- 238000001914 filtration Methods 0.000 description 24
- RAXXELZNTBOGNW-UHFFFAOYSA-N imidazole Natural products C1=CNC=N1 RAXXELZNTBOGNW-UHFFFAOYSA-N 0.000 description 24
- 239000000126 substance Substances 0.000 description 19
- LCFVJGUPQDGYKZ-UHFFFAOYSA-N Bisphenol A diglycidyl ether Chemical compound C=1C=C(OCC2OC2)C=CC=1C(C)(C)C(C=C1)=CC=C1OCC1CO1 LCFVJGUPQDGYKZ-UHFFFAOYSA-N 0.000 description 17
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 16
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 15
- 230000002378 acidificating effect Effects 0.000 description 14
- 238000006555 catalytic reaction Methods 0.000 description 14
- 239000012065 filter cake Substances 0.000 description 14
- 238000002844 melting Methods 0.000 description 14
- 230000008018 melting Effects 0.000 description 14
- 239000000047 product Substances 0.000 description 13
- 238000000926 separation method Methods 0.000 description 13
- 239000007787 solid Substances 0.000 description 13
- 238000003756 stirring Methods 0.000 description 11
- 229940126062 Compound A Drugs 0.000 description 10
- NLDMNSXOCDLTTB-UHFFFAOYSA-N Heterophylliin A Natural products O1C2COC(=O)C3=CC(O)=C(O)C(O)=C3C3=C(O)C(O)=C(O)C=C3C(=O)OC2C(OC(=O)C=2C=C(O)C(O)=C(O)C=2)C(O)C1OC(=O)C1=CC(O)=C(O)C(O)=C1 NLDMNSXOCDLTTB-UHFFFAOYSA-N 0.000 description 10
- 229920000642 polymer Polymers 0.000 description 10
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 9
- 238000012360 testing method Methods 0.000 description 9
- 239000008367 deionised water Substances 0.000 description 8
- 229910021641 deionized water Inorganic materials 0.000 description 8
- 238000003860 storage Methods 0.000 description 8
- 239000003795 chemical substances by application Substances 0.000 description 7
- LNEPOXFFQSENCJ-UHFFFAOYSA-N haloperidol Chemical compound C1CC(O)(C=2C=CC(Cl)=CC=2)CCN1CCCC(=O)C1=CC=C(F)C=C1 LNEPOXFFQSENCJ-UHFFFAOYSA-N 0.000 description 7
- 238000010438 heat treatment Methods 0.000 description 7
- VLGJMNYHUDQEMI-UHFFFAOYSA-N 2,6-dimethylaniline Chemical compound CC1=C(N)C(=CC=C1)C.NC=1C(=CC=CC1C)C VLGJMNYHUDQEMI-UHFFFAOYSA-N 0.000 description 6
- 229910052799 carbon Inorganic materials 0.000 description 6
- 238000005406 washing Methods 0.000 description 6
- 230000000694 effects Effects 0.000 description 5
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 4
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical group [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 4
- JNGZXGGOCLZBFB-IVCQMTBJSA-N compound E Chemical compound N([C@@H](C)C(=O)N[C@@H]1C(N(C)C2=CC=CC=C2C(C=2C=CC=CC=2)=N1)=O)C(=O)CC1=CC(F)=CC(F)=C1 JNGZXGGOCLZBFB-IVCQMTBJSA-N 0.000 description 4
- 238000004132 cross linking Methods 0.000 description 4
- 238000001394 phosphorus-31 nuclear magnetic resonance spectrum Methods 0.000 description 4
- 238000001556 precipitation Methods 0.000 description 4
- 238000001228 spectrum Methods 0.000 description 4
- 238000002485 combustion reaction Methods 0.000 description 3
- 238000001035 drying Methods 0.000 description 3
- FVIZARNDLVOMSU-UHFFFAOYSA-N ginsenoside K Natural products C1CC(C2(CCC3C(C)(C)C(O)CCC3(C)C2CC2O)C)(C)C2C1C(C)(CCC=C(C)C)OC1OC(CO)C(O)C(O)C1O FVIZARNDLVOMSU-UHFFFAOYSA-N 0.000 description 3
- 239000000463 material Substances 0.000 description 3
- LVTJOONKWUXEFR-FZRMHRINSA-N protoneodioscin Natural products O(C[C@@H](CC[C@]1(O)[C@H](C)[C@@H]2[C@]3(C)[C@H]([C@H]4[C@@H]([C@]5(C)C(=CC4)C[C@@H](O[C@@H]4[C@H](O[C@H]6[C@@H](O)[C@@H](O)[C@@H](O)[C@H](C)O6)[C@@H](O)[C@H](O[C@H]6[C@@H](O)[C@@H](O)[C@@H](O)[C@H](C)O6)[C@H](CO)O4)CC5)CC3)C[C@@H]2O1)C)[C@H]1[C@H](O)[C@H](O)[C@H](O)[C@@H](CO)O1 LVTJOONKWUXEFR-FZRMHRINSA-N 0.000 description 3
- BSYJHYLAMMJNRC-UHFFFAOYSA-N 2,4,4-trimethylpentan-2-ol Chemical compound CC(C)(C)CC(C)(C)O BSYJHYLAMMJNRC-UHFFFAOYSA-N 0.000 description 2
- TYTPPOBYRKMHAV-UHFFFAOYSA-N 2,6-dimethylphenol Chemical compound CC1=CC=CC(C)=C1O.CC1=CC=CC(C)=C1O TYTPPOBYRKMHAV-UHFFFAOYSA-N 0.000 description 2
- GXTKSTZCYSVOCA-UHFFFAOYSA-N 4-[bis(4-aminophenyl)-benzo[c][2,1]benzoxaphosphinin-6-ylmethyl]aniline Chemical compound C1=CC(N)=CC=C1C(C=1C=CC(N)=CC=1)(C=1C=CC(N)=CC=1)P1C2=CC=CC=C2C2=CC=CC=C2O1 GXTKSTZCYSVOCA-UHFFFAOYSA-N 0.000 description 2
- MQJKPEGWNLWLTK-UHFFFAOYSA-N Dapsone Chemical compound C1=CC(N)=CC=C1S(=O)(=O)C1=CC=C(N)C=C1 MQJKPEGWNLWLTK-UHFFFAOYSA-N 0.000 description 2
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 2
- FFBHFFJDDLITSX-UHFFFAOYSA-N benzyl N-[2-hydroxy-4-(3-oxomorpholin-4-yl)phenyl]carbamate Chemical compound OC1=C(NC(=O)OCC2=CC=CC=C2)C=CC(=C1)N1CCOCC1=O FFBHFFJDDLITSX-UHFFFAOYSA-N 0.000 description 2
- 150000001721 carbon Chemical group 0.000 description 2
- 229910052757 nitrogen Inorganic materials 0.000 description 2
- 229910052760 oxygen Inorganic materials 0.000 description 2
- 239000001301 oxygen Substances 0.000 description 2
- 125000004437 phosphorous atom Chemical group 0.000 description 2
- 239000004033 plastic Substances 0.000 description 2
- 125000001424 substituent group Chemical group 0.000 description 2
- 230000002194 synthesizing effect Effects 0.000 description 2
- KUCOHFSKRZZVRO-UHFFFAOYSA-N terephthalaldehyde Chemical compound O=CC1=CC=C(C=O)C=C1 KUCOHFSKRZZVRO-UHFFFAOYSA-N 0.000 description 2
- AKUPIABWVUYZPX-UHFFFAOYSA-N 1,2,3,4-tetrabromodibenzo-p-dioxin Chemical compound C1=CC=C2OC3=C(Br)C(Br)=C(Br)C(Br)=C3OC2=C1 AKUPIABWVUYZPX-UHFFFAOYSA-N 0.000 description 1
- QLPZYYOHERFPKO-UHFFFAOYSA-N 1,2,3,4-tetrabromodibenzofuran Chemical compound C1=CC=C2C3=C(Br)C(Br)=C(Br)C(Br)=C3OC2=C1 QLPZYYOHERFPKO-UHFFFAOYSA-N 0.000 description 1
- AZQWKYJCGOJGHM-UHFFFAOYSA-N 1,4-benzoquinone Chemical compound O=C1C=CC(=O)C=C1 AZQWKYJCGOJGHM-UHFFFAOYSA-N 0.000 description 1
- XQUPVDVFXZDTLT-UHFFFAOYSA-N 1-[4-[[4-(2,5-dioxopyrrol-1-yl)phenyl]methyl]phenyl]pyrrole-2,5-dione Chemical compound O=C1C=CC(=O)N1C(C=C1)=CC=C1CC1=CC=C(N2C(C=CC2=O)=O)C=C1 XQUPVDVFXZDTLT-UHFFFAOYSA-N 0.000 description 1
- JECYNCQXXKQDJN-UHFFFAOYSA-N 2-(2-methylhexan-2-yloxymethyl)oxirane Chemical compound CCCCC(C)(C)OCC1CO1 JECYNCQXXKQDJN-UHFFFAOYSA-N 0.000 description 1
- PKIRNMPSENTWJX-UHFFFAOYSA-N 4-aminobenzenesulfonic acid Chemical compound NC1=CC=C(S(O)(=O)=O)C=C1.NC1=CC=C(S(O)(=O)=O)C=C1 PKIRNMPSENTWJX-UHFFFAOYSA-N 0.000 description 1
- FYEHYMARPSSOBO-UHFFFAOYSA-N Aurin Chemical compound C1=CC(O)=CC=C1C(C=1C=CC(O)=CC=1)=C1C=CC(=O)C=C1 FYEHYMARPSSOBO-UHFFFAOYSA-N 0.000 description 1
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 1
- VXYXEUAMMZCHGJ-UHFFFAOYSA-N CC(C1)C(O)=CC=C1C(c(cc1)ccc1N)(c(cc1)ccc1N)P1(Oc(cccc2)c2-c2ccccc12)=O Chemical compound CC(C1)C(O)=CC=C1C(c(cc1)ccc1N)(c(cc1)ccc1N)P1(Oc(cccc2)c2-c2ccccc12)=O VXYXEUAMMZCHGJ-UHFFFAOYSA-N 0.000 description 1
- IAZDPXIOMUYVGZ-WFGJKAKNSA-N Dimethyl sulfoxide Chemical compound [2H]C([2H])([2H])S(=O)C([2H])([2H])[2H] IAZDPXIOMUYVGZ-WFGJKAKNSA-N 0.000 description 1
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 1
- GUGOZJZDBGQPEV-UHFFFAOYSA-N N1=CC(=CC=C1)S(=O)(=O)O.N1=CC(=CC=C1)S(=O)(=O)O Chemical compound N1=CC(=CC=C1)S(=O)(=O)O.N1=CC(=CC=C1)S(=O)(=O)O GUGOZJZDBGQPEV-UHFFFAOYSA-N 0.000 description 1
- 238000005481 NMR spectroscopy Methods 0.000 description 1
- ZLSOCDZUBPPZCK-UHFFFAOYSA-N P(=O)(=O)C1OC2=CC=CC=C2C=2C=CC=CC12 Chemical compound P(=O)(=O)C1OC2=CC=CC=C2C=2C=CC=CC12 ZLSOCDZUBPPZCK-UHFFFAOYSA-N 0.000 description 1
- OFOBLEOULBTSOW-UHFFFAOYSA-N Propanedioic acid Natural products OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- 230000004888 barrier function Effects 0.000 description 1
- RWCCWEUUXYIKHB-UHFFFAOYSA-N benzophenone Chemical compound C=1C=CC=CC=1C(=O)C1=CC=CC=C1 RWCCWEUUXYIKHB-UHFFFAOYSA-N 0.000 description 1
- 239000012965 benzophenone Substances 0.000 description 1
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 1
- 229910052794 bromium Inorganic materials 0.000 description 1
- 125000001246 bromo group Chemical group Br* 0.000 description 1
- VBRNLOQCBCPPHL-UHFFFAOYSA-N calmagite Chemical compound CC1=CC=C(O)C(N=NC=2C3=CC=CC=C3C(=CC=2O)S(O)(=O)=O)=C1 VBRNLOQCBCPPHL-UHFFFAOYSA-N 0.000 description 1
- 238000001460 carbon-13 nuclear magnetic resonance spectrum Methods 0.000 description 1
- 238000003763 carbonization Methods 0.000 description 1
- 239000003518 caustics Substances 0.000 description 1
- 239000000571 coke Substances 0.000 description 1
- 238000005336 cracking Methods 0.000 description 1
- 230000002950 deficient Effects 0.000 description 1
- 230000018044 dehydration Effects 0.000 description 1
- 238000006297 dehydration reaction Methods 0.000 description 1
- 238000010586 diagram Methods 0.000 description 1
- 150000002170 ethers Chemical class 0.000 description 1
- 229910052736 halogen Inorganic materials 0.000 description 1
- 150000002367 halogens Chemical class 0.000 description 1
- BHEPBYXIRTUNPN-UHFFFAOYSA-N hydridophosphorus(.) (triplet) Chemical compound [PH] BHEPBYXIRTUNPN-UHFFFAOYSA-N 0.000 description 1
- 229930195733 hydrocarbon Natural products 0.000 description 1
- 150000002430 hydrocarbons Chemical class 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 230000002401 inhibitory effect Effects 0.000 description 1
- GYCHYNMREWYSKH-UHFFFAOYSA-L iron(ii) bromide Chemical compound [Fe+2].[Br-].[Br-] GYCHYNMREWYSKH-UHFFFAOYSA-L 0.000 description 1
- 239000006166 lysate Substances 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- 239000011976 maleic acid Substances 0.000 description 1
- BCVXHSPFUWZLGQ-UHFFFAOYSA-N mecn acetonitrile Chemical compound CC#N.CC#N BCVXHSPFUWZLGQ-UHFFFAOYSA-N 0.000 description 1
- GXHMMDRXHUIUMN-UHFFFAOYSA-N methanesulfonic acid Chemical compound CS(O)(=O)=O.CS(O)(=O)=O GXHMMDRXHUIUMN-UHFFFAOYSA-N 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 125000000896 monocarboxylic acid group Chemical group 0.000 description 1
- 229920003986 novolac Polymers 0.000 description 1
- 238000005935 nucleophilic addition reaction Methods 0.000 description 1
- 150000002903 organophosphorus compounds Chemical class 0.000 description 1
- 239000005022 packaging material Substances 0.000 description 1
- 231100000614 poison Toxicity 0.000 description 1
- 229920003192 poly(bis maleimide) Polymers 0.000 description 1
- 239000002861 polymer material Substances 0.000 description 1
- 229920000137 polyphosphoric acid Polymers 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 239000002244 precipitate Substances 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 230000001681 protective effect Effects 0.000 description 1
- 239000000376 reactant Substances 0.000 description 1
- 229920005989 resin Polymers 0.000 description 1
- 239000011347 resin Substances 0.000 description 1
- CBXWGGFGZDVPNV-UHFFFAOYSA-N so4-so4 Chemical compound OS(O)(=O)=O.OS(O)(=O)=O CBXWGGFGZDVPNV-UHFFFAOYSA-N 0.000 description 1
- 239000000243 solution Substances 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 238000010183 spectrum analysis Methods 0.000 description 1
- 238000001308 synthesis method Methods 0.000 description 1
- 238000010189 synthetic method Methods 0.000 description 1
- 238000010998 test method Methods 0.000 description 1
- 239000003440 toxic substance Substances 0.000 description 1
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 1
- FAQYAMRNWDIXMY-UHFFFAOYSA-N trichloroborane Chemical compound ClB(Cl)Cl FAQYAMRNWDIXMY-UHFFFAOYSA-N 0.000 description 1
Images
Landscapes
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
Abstract
Description
技术领域 technical field
本发明揭示一种含磷化合物,其可作为环氧树脂硬化剂,并且使用于制备阻燃环氧树脂硬化物。The invention discloses a phosphorus-containing compound, which can be used as an epoxy resin hardener and used for preparing flame-retardant epoxy resin hardened products.
背景技术 Background technique
环氧树脂具有优良的电器性质,形状稳定性,耐高温,耐溶剂,低成本和高粘合性等优点,最适合做印刷电路板以及集成电路封装材料。然而,环氧树脂与一般塑料材料一样容易燃烧危害生命,因此全世界对于使用电子、信息用材料,其阻燃性有严格要求。含溴原子的环氧树脂特别适用于具有阻燃特性的电路板。然而,这些含溴环氧树脂在燃烧过程会释出四溴二苯并戴奥辛(tetrabromodibenzo-p-dioxin)与四溴二苯并呋喃(tetrabromodibenzofuran)等具有腐蚀性和毒性物质。除了含卤素的化合物,另一种阻燃的方法便是在塑料外再包覆不可燃的外层。这些阻燃化合物之中,有机磷化合物具有高度阻燃性,磷系阻燃剂在燃烧时会促使高分子材料先行脱水,使碳氢化合物的氢与空气中氧形成水,从而降低周遭环境的温度,使其低于燃烧温度而达到阻燃效果;另一方面在高温加热下会分解出磷酸促进高分子化合物的碳化,而形成不燃性焦炭层。另外磷酸在高温下会进一步脱水酯化形成聚磷酸覆盖于燃烧物的表面形成保护作用,阻止氧气进入高分子未燃烧的内层以及抑制挥发性裂解物的释放。导入磷元素的方法有两种,一种是合成含磷环氧树脂,另一种是合成含磷硬化剂。本发明是以合成新颖含磷硬化剂方式,使得环氧树脂硬化达到阻燃的效果。Epoxy resin has the advantages of excellent electrical properties, shape stability, high temperature resistance, solvent resistance, low cost and high adhesion, and is most suitable for printed circuit boards and integrated circuit packaging materials. However, epoxy resin is as easy to burn as general plastic materials and endangers life. Therefore, there are strict requirements for the flame retardancy of electronic and information materials used all over the world. Epoxy resins containing bromine atoms are especially suitable for circuit boards with flame retardant properties. However, these bromine-containing epoxy resins release corrosive and toxic substances such as tetrabromodibenzo-p-dioxin and tetrabromodibenzofuran during combustion. In addition to halogen-containing compounds, another flame-retardant method is to coat the plastic with a non-flammable outer layer. Among these flame retardant compounds, organophosphorus compounds are highly flame retardant. Phosphorous flame retardants will promote the dehydration of polymer materials when they burn, so that the hydrogen of hydrocarbons and oxygen in the air will form water, thereby reducing the temperature of the surrounding environment. The temperature is lower than the combustion temperature to achieve the flame retardant effect; on the other hand, under high temperature heating, phosphoric acid will be decomposed to promote the carbonization of polymer compounds, forming a non-combustible coke layer. In addition, phosphoric acid will be further dehydrated and esterified at high temperature to form polyphosphoric acid to cover the surface of the burning material to form a protective effect, preventing oxygen from entering the unburned inner layer of the polymer and inhibiting the release of volatile lysates. There are two ways to introduce phosphorus, one is to synthesize phosphorus-containing epoxy resin, and the other is to synthesize phosphorus-containing hardener. The invention uses the method of synthesizing a novel phosphorus-containing curing agent to harden the epoxy resin to achieve the effect of flame retardancy.
在含磷反应物中,具反应性的9,10-二氢-9-氧杂-10-磷菲-10-氧化物(9,10-dihydro-9-oxa-10-phosphaphenanthrene 10-oxide,DOPO)深受瞩目,因为具可与缺电子化合物如苯二酮(benzoquinone)、环氧乙烷(oxirane)、马来酸(maleic acid)、双马来酰亚胺(bismaleimide)、二氨基二苯甲酮(diaminobenzophenone)和对苯二甲醛(terephthaldicarboxaldehyde)等进行亲核加成反应。林等人在2005年分别揭露三官能硬化剂dopotriol[1]与dopo-ta[2]的合成方法与应用,成功的得到阻燃且具有高玻璃化转变温度的环氧树脂。然而,文献中合成dopotriol的原料蔷薇酸(rosolic acid)非常昂贵,在工业应用上不符合经济效益。本发明利用以较便宜的4,4′-二羟基二苯基甲酮(4,4′-dihydroxybenzophenone,DHBP)、DOPO与苯酚反应成功的合成化合物A(dopotriol),除此之外,并且更进一步揭露DHBP、4,4′-二氨基二苯基甲酮(4,4′-diamino benzophenone,DABP)与4-氨基-4′-羟基二苯基甲酮(4-amino-4′-hydroxy benzophenone,AHBP)衍生物的合成方法。Among phosphorus-containing reactants, reactive 9,10-dihydro-9-oxa-10-phosphaphenanthrene-10-oxide (9,10-dihydro-9-oxa-10-phosphaphenanthrene 10-oxide, DOPO) has attracted much attention because it can interact with electron-deficient compounds such as benzoquinone, oxirane, maleic acid, bismaleimide, diaminodi Benzophenone (diaminobenzophenone) and terephthaldicarboxaldehyde (terephthaldicarboxaldehyde) etc. undergo nucleophilic addition reaction. In 2005, Lin et al. disclosed the synthesis method and application of the trifunctional hardeners dopotriol[1] and dopo-ta[2], and successfully obtained a flame-retardant epoxy resin with a high glass transition temperature. However, rosolic acid, the raw material for synthesizing dopotriol in the literature, is very expensive, and it is not economical for industrial applications. The present invention utilizes the synthetic compound A (dopotriol) that successfully reacts with cheaper 4,4'-dihydroxybenzophenone (4,4'-dihydroxybenzophenone, DHBP), DOPO and phenol, in addition, and more Further expose DHBP, 4,4'-diaminobenzophenone (4,4'-diaminobenzophenone, DABP) and 4-amino-4'-hydroxybenzophenone (4-amino-4'-hydroxy The synthetic method of benzophenone, AHBP) derivative.
参考文献:references:
[1]C.H.Lin,S.X.Cai和C.H.Lin,“具有高玻璃转变化温度的阻燃性环氧树脂。II.使用新颖六官能固化剂:9,10-二氢-9-氧杂-10-磷菲10-基-三(4-氨基苯基)甲烷(Flame-Retardant Epoxy Resins with High Glass-Transition Temperatures.II.Using aNovel Hexafunctional Curing Agent:9,10-Dihydro-9-oxa-10-phosphaphenanthrene10-yl-tris(4-aminophenyl)methane,)”聚合物科学聚合物化学杂志(J.Polym.Sci.Polym.Chem.),2005,43,5971.[1] C.H.Lin, S.X.Cai, and C.H.Lin, "Flame-retardant epoxy resins with high glass transition temperature. II. Using a novel hexafunctional curing agent: 9,10-dihydro-9-oxa-10- Phosphaphenanthrene 10-yl-tri(4-aminophenyl)methane (Flame-Retardant Epoxy Resins with High Glass-Transition Temperatures.II.Using a Novel Hexafunctional Curing Agent: 9,10-Dihydro-9-oxa-10-phosphaphenanthrene10- yl-tris(4-aminophenyl)methane,)" Polymer Science Journal of Polymer Chemistry (J.Polym.Sci.Polym.Chem.), 2005, 43, 5971.
[2]S.X.Cai和C.H.Lin,“来自新颖三官能固化剂:Dopotriol具有高Tg的阻燃环氧树脂(Flame-Retardant Epoxy Resins With High Tg from a Novel Tri-functional CuringAgent:Dopotriol),”聚合物科学聚合物化学杂志,2005,43,2862。[2] S.X.Cai and C.H.Lin, "Flame-Retardant Epoxy Resins With High Tg from a Novel Tri-functional Curing Agent: Dopotriol" from a Novel Tri-functional Curing Agent: Dopotriol," Polymer Journal of Scientific Polymer Chemistry, 2005, 43, 2862.
发明内容 Contents of the invention
本发明目的提供一种新颖磷系化合物,其可作为环氧树脂硬化剂,也可进一步用于制备具有阻燃特性的环氧树脂。The object of the present invention is to provide a novel phosphorus compound, which can be used as a hardener for epoxy resin, and can also be further used to prepare epoxy resin with flame retardant properties.
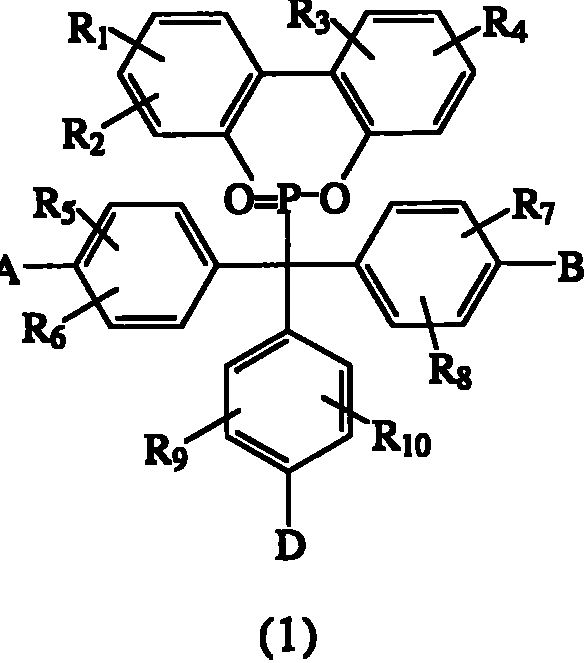
本发明揭示具有下列化学式的磷系化合物:The present invention discloses phosphorus compounds having the following chemical formula:
其中in
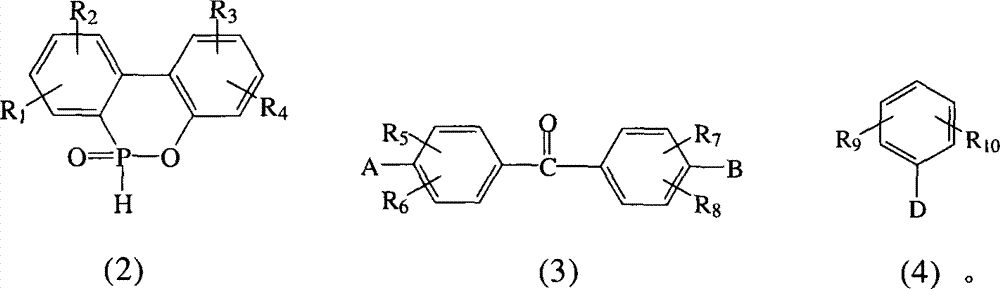
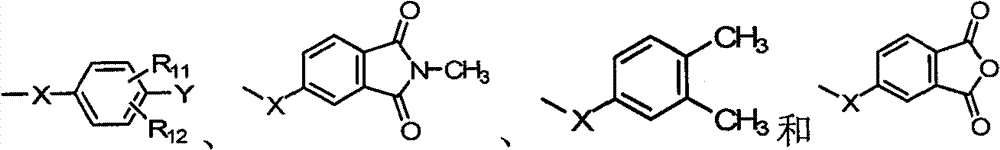
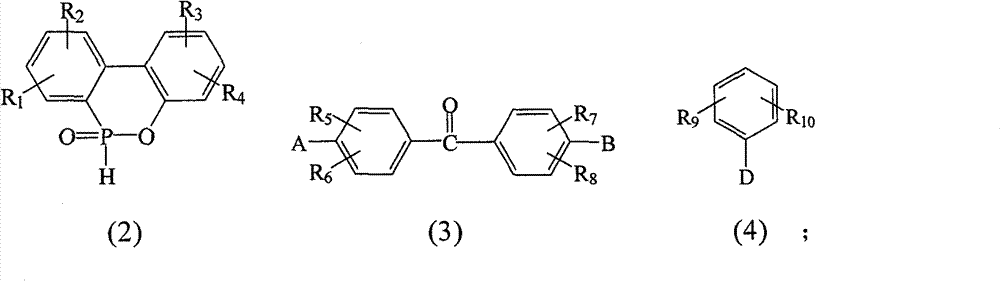
R1~R10各自独立选自氢原子、C1~C6烷基、C1~C6烷氧基、苯基、硝基、苯氧基、C1~C10卤烷基、C3~C7环烷基、-CF3、-OCF3和卤原子所组成的群组;R 1 to R 10 are each independently selected from a hydrogen atom, C 1 to C 6 alkyl, C 1 to C 6 alkoxy, phenyl, nitro, phenoxy, C 1 to C 10 haloalkyl, C 3 A group consisting of ~C 7 cycloalkyl, -CF 3 , -OCF 3 and halogen atoms;
A为-OH或-NH2;A is -OH or -NH 2 ;
B为-OH或-NH2;B is -OH or -NH 2 ;
D选自-OH、-NH2、C1~C6烷基、C1~C6烷氧基、C1~C10卤烷基、C3~C7环烷基、-CF3、-OCF3、卤原子、-NHR1、-NH(C=O)-R1、-NH(O=C-O)-R1、-NH(O=C-NH)-R1、 所组成的群组;D is selected from -OH, -NH 2 , C 1 ~C 6 alkyl, C 1 ~C 6 alkoxy, C 1 ~C 10 haloalkyl, C 3 ~C 7 cycloalkyl, -CF 3 , - OCF 3 , halogen atom, -NHR 1 , -NH(C=O)-R 1 , -NH(O=CO)-R 1 , -NH(O=C-NH)-R 1 , the group formed;
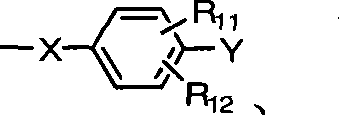
X为氧原子或-NH;X is an oxygen atom or -NH;
Y选自氢原子、-NO2、-OH、-NH2、-COOH、C1~C6烷基、C1~C6烷氧基、C3~C7环烷基、-CF3、-OCF3和卤原子所组成的群组;Y is selected from hydrogen atom, -NO 2 , -OH, -NH 2 , -COOH, C 1 ~C 6 alkyl, C 1 ~C 6 alkoxy, C 3 ~C 7 cycloalkyl, -CF 3 , - the group formed by OCF 3 and a halogen atom;
R11~R12各自独立选自氢原子、C1~C6烷基、C1~C6烷氧基、苯基、硝基、苯氧基、C1~C10卤烷基、C3~C7环烷基、-CF3、-OCF3和卤原子所组成的群组;R 11 to R 12 are each independently selected from a hydrogen atom, C 1 to C 6 alkyl, C 1 to C 6 alkoxy, phenyl, nitro, phenoxy, C 1 to C 10 haloalkyl, C 3 A group consisting of ~C 7 cycloalkyl, -CF 3 , -OCF 3 and halogen atoms;
其限制条件为:Its constraints are:
(1)当D为-OH,且A与B同时为-NH2时,(1) When D is -OH, and A and B are both -NH 2 ,
R1~R10中至少有一个为苯基、硝基或苯氧基;或At least one of R 1 to R 10 is phenyl, nitro or phenoxy; or
(2)当A、B与D同时为-OH时,(2) When A, B and D are -OH at the same time,
(i)R1~R10中至少有一个为C1~C10卤烷基、-CF3或-OCF3;或(i) At least one of R 1 to R 10 is C 1 to C 10 haloalkyl, -CF 3 or -OCF 3 ; or
(ii)(R1,R2),(R3,R4),(R5,R6),(R7,R8),(R9,R10)五组取代基组合中至少有一组与其它组的组合不同;或(ii) At least one of the five groups of substituent combinations (R 1 , R 2 ), (R 3 , R 4 ), (R 5 , R 6 ), (R7, R8), (R9, R10) is combined with other groups different combinations; or
(3)当D选自C1~C6烷基、C1~C6烷氧基、C1~C10卤烷基、C3~C7环烷基、-CF3、-OCF3和卤原子所组成的群组,且A与B同时为-NH2时,(3) When D is selected from C 1 ~C 6 alkyl, C 1 ~C 6 alkoxy, C 1 ~C 10 haloalkyl, C 3 ~C 7 cycloalkyl, -CF 3 , -OCF 3 and A group composed of halogen atoms, and when A and B are -NH 2 at the same time,
R1~R10中至少有一个为苯基、硝基或苯氧基;或At least one of R 1 to R 10 is phenyl, nitro or phenoxy; or
(4)当A、B与D同时为-NH2时,(4) When A, B and D are -NH 2 at the same time,
(i)R1~R10中至少有一个为苯基、硝基或苯氧基,且(R1,R2),(R3,R4),(R5,R6),(R7,R8),(R9,R10)五组取代基组合中至少有一组与其它组的组合不同;或(i) At least one of R 1 to R 10 is phenyl, nitro or phenoxy, and (R 1 , R 2 ), (R 3 , R 4 ), (R 5 , R 6 ), (R 7 , R 8 ), (R 9 , R 10 ) at least one of the five substituent combinations is different from the other combinations; or
(ii)R1~R10中至少有一个为苯基、硝基或苯氧基,且R1~R10中至少有一个为C1~C10卤烷基、-CF3、-OCF3。(ii) At least one of R 1 to R 10 is phenyl, nitro or phenoxy, and at least one of R 1 to R 10 is C 1 to C 10 haloalkyl, -CF 3 , -OCF 3 .
当上述式(1)化合物的A、B与D同时为-OH,R1~R9各为氢原子,R10为-CH3,所述式(1)化合物的结构式可为When A, B and D of the above-mentioned compound of formula (1) are -OH at the same time, each of R 1 to R 9 is a hydrogen atom, and R 10 is -CH 3 , the structural formula of the compound of formula (1) can be
其中一个具体实施例的方面为R10接在苯环位置3的碳上。An aspect of one specific embodiment is that R 10 is attached to the carbon at
当上述式(1)化合物的A、B与D同时为-OH,R1~R8各为氢原子,R9与R10为-CH3,所述式(1)化合物的结构式可为When A, B and D of the above-mentioned compound of formula (1) are -OH at the same time, R 1 to R 8 are each a hydrogen atom, and R 9 and R 10 are -CH 3 , the structural formula of the compound of formula (1) can be
其中一个具体实施例的方面为R9与R10接在苯环位置3与5的碳上。An aspect of one specific embodiment is that R 9 and R 10 are attached to carbons at
当上述式(1)化合物的A与B同时为-OH,D为-NH2,R1~R10各为氢原子,所述式(1)化合物的一个具体实施例的结构式可为When A and B of the above-mentioned compound of formula (1) are -OH at the same time, D is -NH 2 , R 1 to R 10 are each a hydrogen atom, the structural formula of a specific embodiment of the compound of formula (1) can be
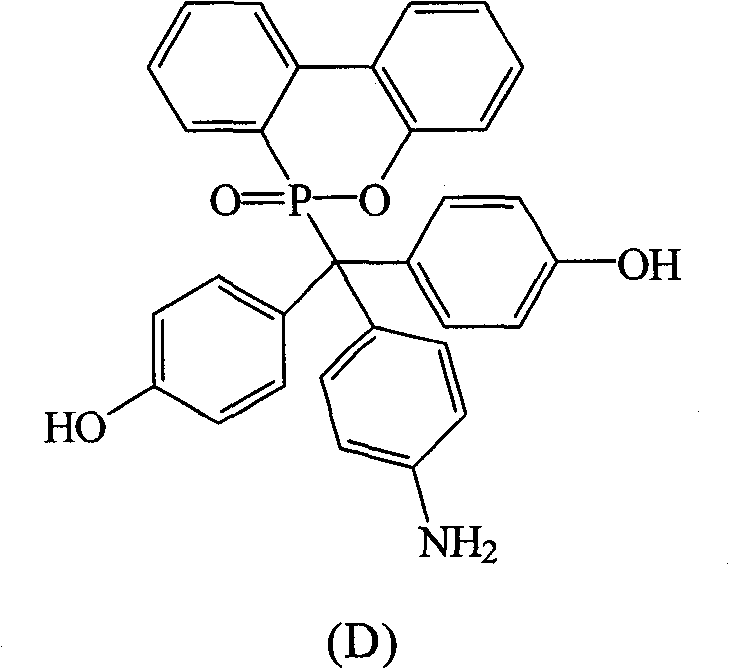
(D) 。(D) .
当上述式(1)化合物的A与B同时为-OH,D为-NH2,R1~R9各为氢原子,R10为-CH3,所述式(1)化合物的结构式可为When A and B of the above-mentioned compound of formula (1) are -OH at the same time, D is -NH 2 , R 1 to R 9 are hydrogen atoms, and R 10 is -CH 3 , the structural formula of the compound of formula (1) can be
其中一个具体实施例的方面为R10接在苯环位置3的碳上。An aspect of one specific embodiment is that R 10 is attached to the carbon at
当上述式(1)化合物的A与B同时为-OH,D为-NH2,R1~R8各为氢原子,R9与R10为-CH3,所述式(1)化合物的结构式可为When A and B of the above compound of formula (1) are -OH at the same time, D is -NH 2 , R 1 to R 8 are hydrogen atoms, R 9 and R 10 are -CH 3 , the compound of formula (1) The structural formula can be
其中一个具体实施例的方面为R9与R10接在苯环位置3与5的碳上。An aspect of one specific embodiment is that R 9 and R 10 are attached to carbons at
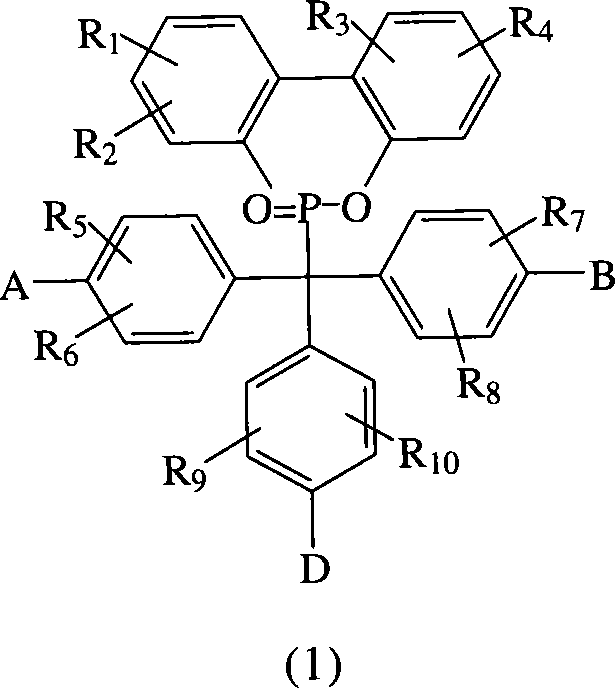
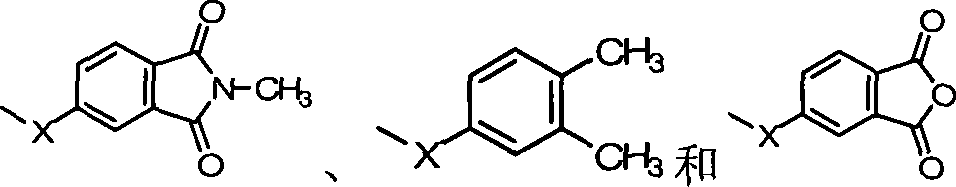
本发明提出一种制备如上述式(1)化合物的方法,包括将有机磷化物式(2)、式(3)化合物、式(4)化合物与酸催化剂进行反应,以生成式(1)的化合物,其中R1~R10、A、B和D如前定义;The present invention proposes a method for preparing compounds of formula (1), comprising reacting organophosphide compounds of formula (2), compounds of formula (3), and compounds of formula (4) with acid catalysts to generate compounds of formula (1) A compound, wherein R 1 to R 10 , A, B and D are as defined above;
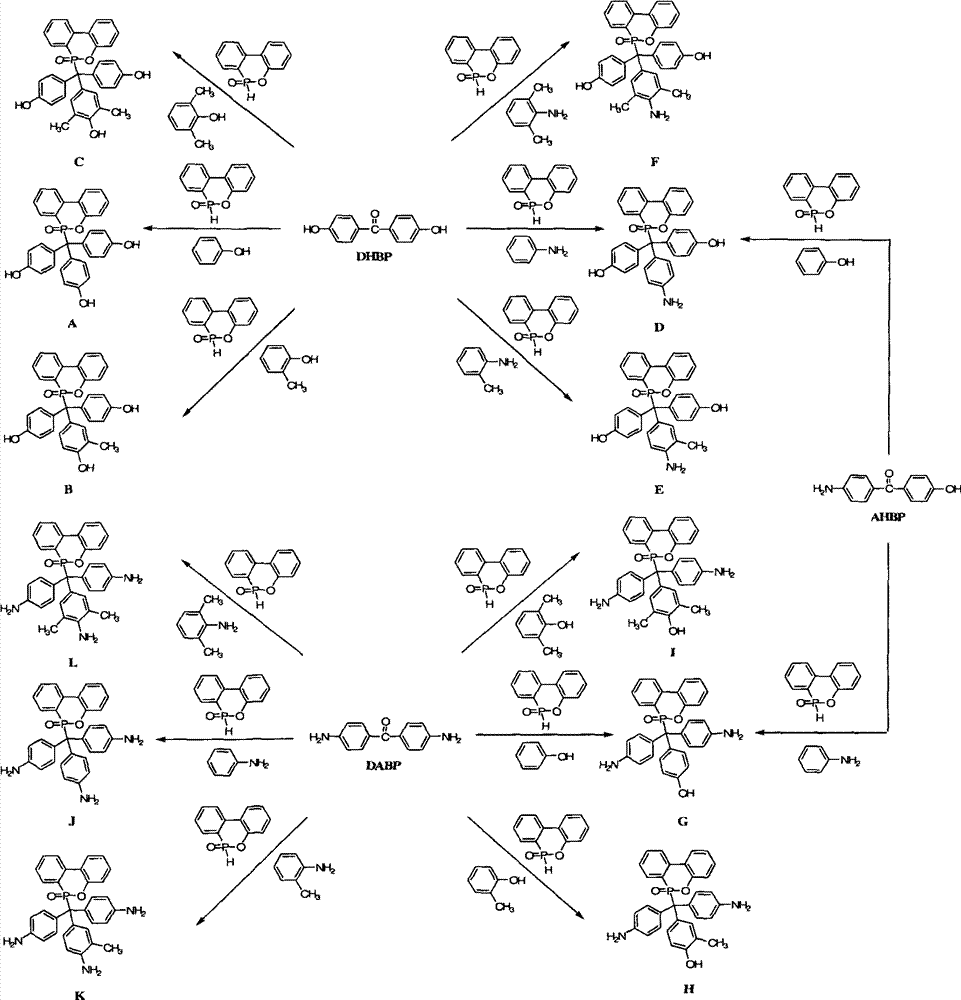
根据上述的方法,在本案的具体实施例中,当A、B与D同时为-OH时,R1~R10为氢原子,将式(2)9,10-二氢-9-氧杂-10-磷菲-10-氧化物(DOPO)、式(3)4,4′-二羟基二苯基甲酮(DHBP)、式(4)苯酚与酸催化剂进行反应,以生成式(A)化合物According to the above method, in the specific embodiment of this case, when A, B and D are -OH at the same time, R 1 ~ R 10 are hydrogen atoms, the formula (2) 9,10-dihydro-9-oxa -10-phosphophenanthrene-10-oxide (DOPO), formula (3) 4,4'-dihydroxybenzophenone (DHBP), formula (4) phenol react with acid catalyst to generate formula (A ) compound
根据上述的方法,在本案的具体实施例中,当A、B与D同时为-OH时,R1~R9为氢原子,R10为-CH3,将式(2)9,10-二氢-9-氧杂-10-磷菲-10-氧化物(DOPO)、式(3)4,4′-二羟基二苯基甲酮(DHBP)、式(4)2-甲酚与酸催化剂进行反应,以生成式(B)化合物。According to the above method, in the specific embodiment of this case, when A, B and D are -OH at the same time, R 1 ~ R 9 are hydrogen atoms, R 10 is -CH 3 , the formula (2)9,10- Dihydro-9-oxa-10-phosphophenanthrene-10-oxide (DOPO), formula (3) 4,4'-dihydroxybenzophenone (DHBP), formula (4) 2-cresol and The acid catalyst is reacted to form the compound of formula (B).
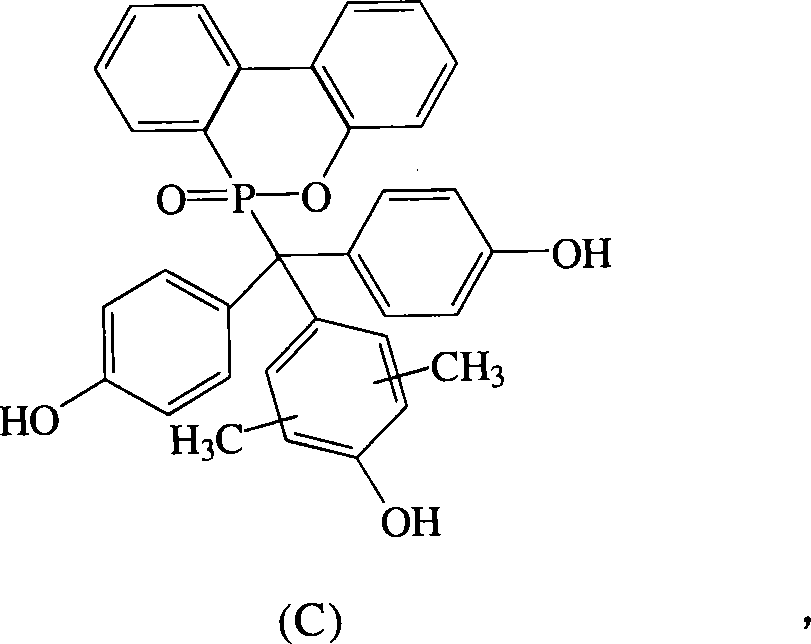
根据上述的方法,在本案的具体实施例中,当A、B与D同时为-OH时,R1~R8为氢原子,R9与R10为-CH3,将式(2)9,10-二氢-9-氧杂-10-磷菲-10-氧化物(DOPO)、式(3)4,4′-二羟基二苯基甲酮(DHBP)、式(4)2,6-二甲基苯酚与酸催化剂进行反应,以生成式(C)化合物。According to the above method, in the specific embodiment of this case, when A, B and D are -OH at the same time, R 1 ~ R 8 are hydrogen atoms, R 9 and R 10 are -CH 3 , the formula (2)9 , 10-dihydro-9-oxa-10-phosphophenanthrene-10-oxide (DOPO), formula (3) 4,4'-dihydroxybenzophenone (DHBP), formula (4) 2, 6-Dimethylphenol is reacted with an acid catalyst to produce a compound of formula (C).
根据上述的方法,在本案的具体实施例中,当A与B同时为-OH,D为-NH2时,R1~R10为氢原子,将式(2)9,10-二氢-9-氧杂-10-磷菲-10-氧化物(DOPO)、式(3)4,4′-二羟基二苯基甲酮(DHBP)、式(4)苯胺与酸催化剂进行反应,以生成式(D)化合物。According to the above method, in the specific embodiment of this case, when A and B are -OH at the same time, and D is -NH 2 , R 1 ~ R 10 are hydrogen atoms, the formula (2) 9,10-dihydro- 9-oxa-10-phosphophenanthrene-10-oxide compound (DOPO), formula (3) 4,4'-dihydroxybenzophenone (DHBP), formula (4) aniline and acid catalyst are reacted, with A compound of formula (D) is produced.
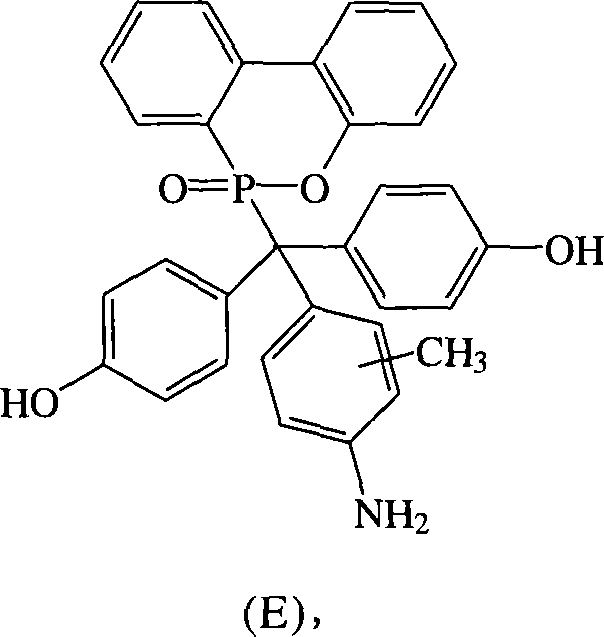
根据上述的方法,在本案的具体实施例中,当A与B同时为~OH,D为-NH2时,R1~R9为氢原子,R10为-CH3,将式(2)9,10-二氢-9-氧杂-10-磷菲-10-氧化物(DOPO)、式(3)4,4′-二羟基二苯基甲酮(DHBP)、式(4)邻-甲苯胺与酸催化剂进行反应,以生成式(E)化合物。According to the above method, in the specific embodiment of this case, when A and B are ~OH at the same time, and D is -NH 2 , R 1 ~R 9 are hydrogen atoms, R 10 is -CH 3 , the formula (2) 9,10-dihydro-9-oxa-10-phosphophenanthrene-10-oxide (DOPO), formula (3) 4,4'-dihydroxybenzophenone (DHBP), formula (4) o - Toluidine is reacted with an acid catalyst to form a compound of formula (E).
根据上述的方法,在本案的具体实施例中,当A与B同时为-OH,D为-NH2时,R1~R8为氢原子,R9与R10为-CH3,将式(2)9,10-二氢-9-氧杂-10-磷菲-10-氧化物(DOPO)、式(3)4,4′-二羟基二苯基甲酮(DHBP)、式(4)2,6-二甲基苯胺与酸催化剂进行反应,以生成式(F)化合物。According to the above method, in the specific embodiment of this case, when A and B are -OH at the same time, and D is -NH 2 , R 1 ~ R 8 are hydrogen atoms, R 9 and R 10 are -CH 3 , the formula (2) 9,10-dihydro-9-oxa-10-phosphophenanthrene-10-oxide (DOPO), formula (3) 4,4'-dihydroxybenzophenone (DHBP), formula ( 4) 2,6-dimethylaniline is reacted with an acid catalyst to produce a compound of formula (F).
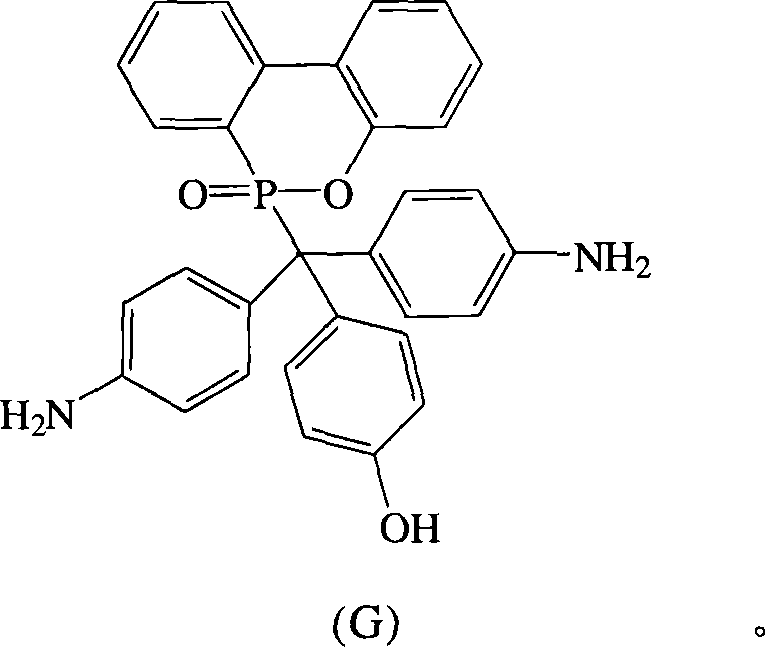
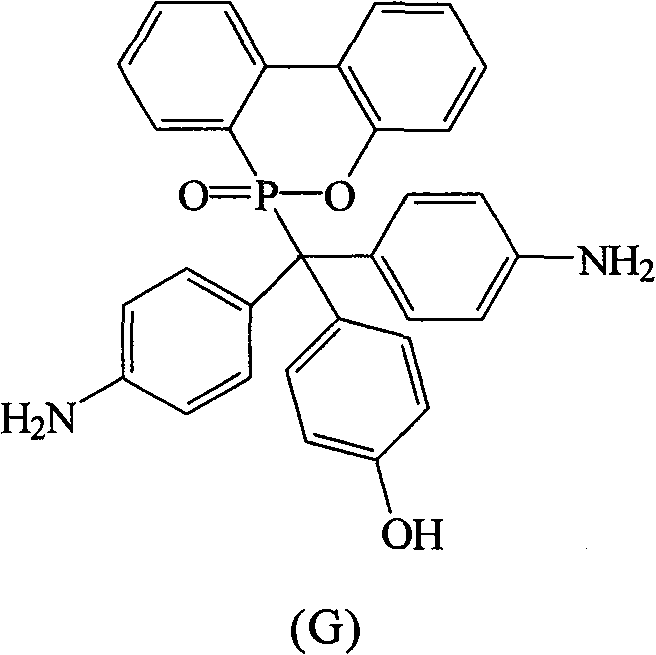
根据上述的方法,在本案的具体实施例中,当A与B同时为-NH2,D为-OH时,R1~R10为氢原子,将式(2)9,10-二氢-9-氧杂-10-磷菲-10-氧化物(DOPO)、式(3)4,4′-二氨基二苯基甲酮(DABP)、式(4)苯酚与酸催化剂进行反应,以生成式(G)化合物According to the above method, in the specific embodiment of this case, when A and B are -NH 2 at the same time, and D is -OH, R 1 ~ R 10 are hydrogen atoms, the formula (2) 9,10-dihydro- 9-oxa-10-phosphophenanthrene-10-oxide compound (DOPO), formula (3) 4,4'-diaminobenzophenone (DABP), formula (4) phenol react with acid catalyst, with Generate the compound of formula (G)
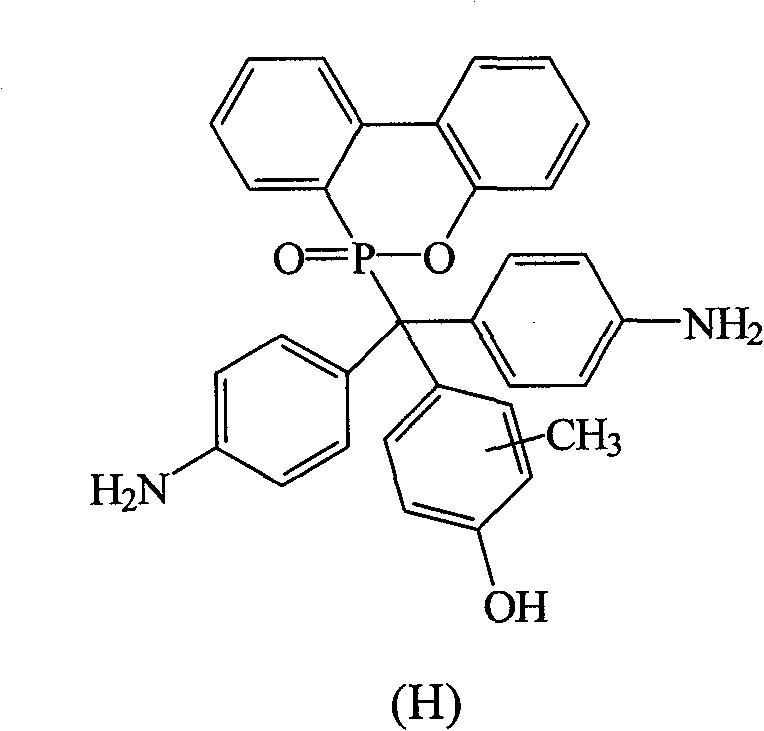
根据上述的方法,在本案的具体实施例中,当A与B同时为-NH2,D为-OH时,R1~R9为氢原子,R10为-CH3,将式(2)9,10-二氢-9-氧杂-10-磷菲-10-氧化物(DOPO)、式(3)4,4′-二氨基二苯基甲酮(DABP)、式(4)2-甲酚与酸催化剂进行反应,以生成式(H)化合物According to the above method, in the specific embodiment of this case, when A and B are both -NH 2 and D is -OH, R 1 to R 9 are hydrogen atoms, R 10 is -CH 3 , the formula (2) 9,10-dihydro-9-oxa-10-phosphophenanthrene-10-oxide (DOPO), formula (3) 4,4'-diaminobenzophenone (DABP), formula (4) 2 -cresol is reacted with an acid catalyst to generate a compound of formula (H)
其中一个具体实施例的方面为R10接在苯环位置3的碳上。An aspect of one specific embodiment is that R 10 is attached to the carbon at
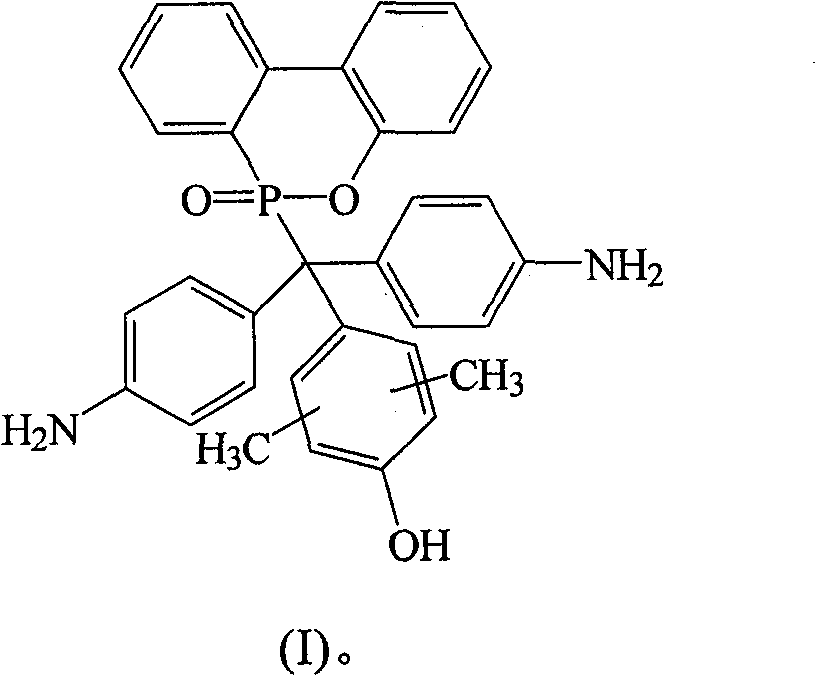
根据上述的方法,在本案的具体实施例中,当A与B同时为-NH2,D为-OH时,R1~R8为氢原子,R9与R10为-CH3,将式(2)9,10-二氢-9-氧杂-10-磷菲-10-氧化物(DOPO)、式(3)4,4′-二氨基二苯基甲酮(DABP)、式(4)2,6-二甲基苯酚与酸催化剂进行反应,以生成式(I)化合物According to the above method, in the specific embodiment of this case, when A and B are both -NH 2 and D is -OH, R 1 ~ R 8 are hydrogen atoms, R 9 and R 10 are -CH 3 , the formula (2) 9,10-dihydro-9-oxa-10-phosphophenanthrene-10-oxide (DOPO), formula (3) 4,4'-diaminobenzophenone (DABP), formula ( 4) 2,6-xylenol is reacted with an acid catalyst to generate a compound of formula (I)
其中一个具体实施例的方面为R9与R10接在苯环位置3与5的碳上。An aspect of one specific embodiment is that R 9 and R 10 are attached to carbons at
根据上述的方法,在本案的具体实施例中,当A、B与D同时为-NH2时,R1~R10为氢原子,将式(2)9,10-二氢-9-氧杂-10-磷菲-10-氧化物(DOPO)、式(3)4,4′-二氨基二苯基甲酮(DABP)、式(4)苯胺与酸催化剂进行反应,以生成式(J)化合物According to the above method, in the specific embodiment of this case, when A, B and D are -NH 2 at the same time, R 1 ~ R 10 are hydrogen atoms, the formula (2) 9,10-dihydro-9-oxo Hetero-10-phosphophenanthrene-10-oxide (DOPO), formula (3) 4,4'-diaminobenzophenone (DABP), formula (4) aniline and acid catalyst are reacted to generate formula ( J) compound
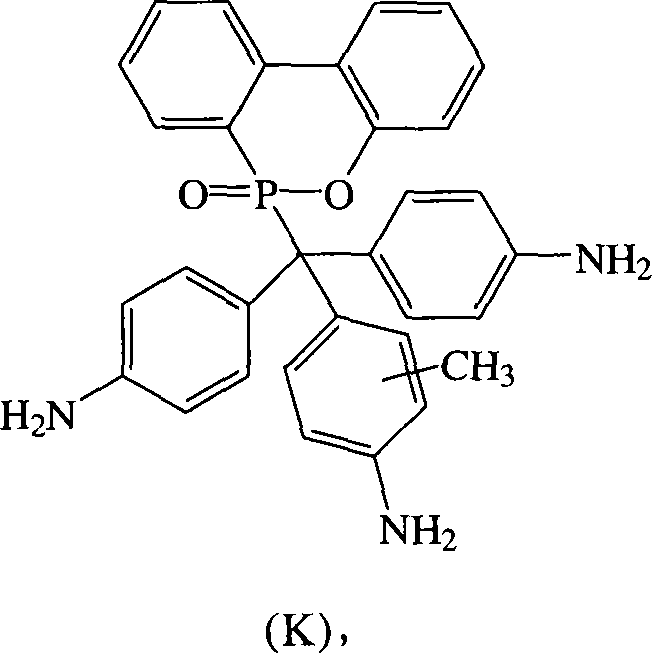
根据上述的方法,在本案的具体实施例中,当A、B与D同时为-NH2时,R1~R9为氢原子,R10为-CH3,将式(2)9,10-二氢-9-氧杂-10-磷菲-10-氧化物(DOPO)、式(3)4,4′-二氨基二苯基甲酮(DABP)、式(4)邻-甲苯胺与酸催化剂进行反应,以生成式(K)化合物According to the above method, in the specific embodiment of this case, when A, B and D are -NH 2 at the same time, R 1 to R 9 are hydrogen atoms, R 10 is -CH 3 , the formula (2)9,10 -Dihydro-9-oxa-10-phosphophenanthrene-10-oxide (DOPO), formula (3) 4,4'-diaminobenzophenone (DABP), formula (4) o-toluidine react with an acid catalyst to generate a compound of formula (K)
其中一个具体实施例的方面为R10接在苯环位置3的碳上。An aspect of one specific embodiment is that R 10 is attached to the carbon at
根据上述的方法,在本案的具体实施例中,当A、B与D同时为-NH2时,R1~R8为氢原子,R9与R10为-CH3,将式(2)9,10-二氢-9-氧杂-10-磷菲-10-氧化物(DOPO)、式(3)4,4′-二氨基二苯基甲酮(DABP)、式(4)2,6-二甲基苯胺与酸催化剂进行反应,以生成式(L)化合物According to the above method, in the specific embodiment of this case, when A, B and D are -NH 2 at the same time, R 1 to R 8 are hydrogen atoms, R 9 and R 10 are -CH 3 , the formula (2) 9,10-dihydro-9-oxa-10-phosphophenanthrene-10-oxide (DOPO), formula (3) 4,4'-diaminobenzophenone (DABP), formula (4) 2 , 6-dimethylaniline is reacted with an acid catalyst to generate a compound of formula (L)
其中一个具体实施例的方面为R9与R10接在苯环位置3与5的碳上。An aspect of one specific embodiment is that R 9 and R 10 are attached to carbons at
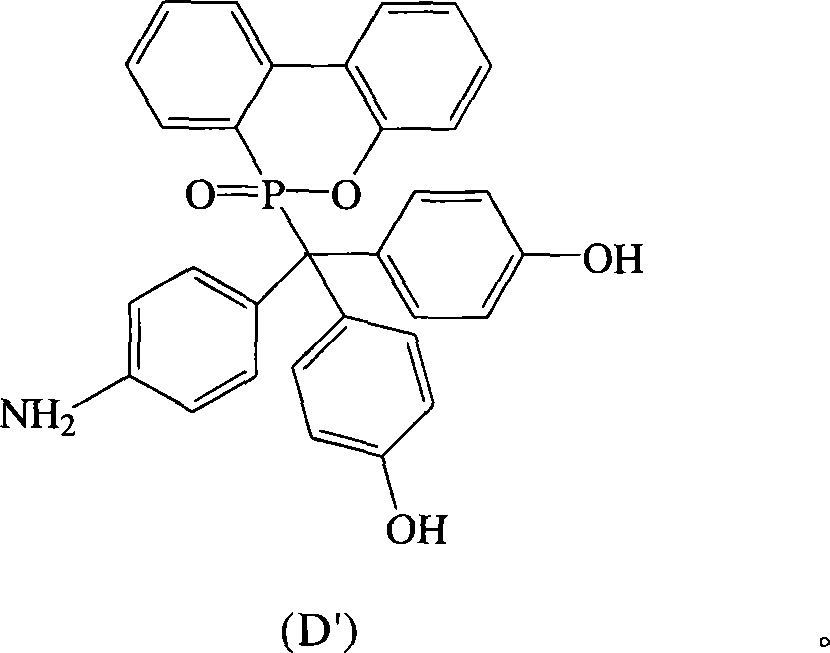
根据上述的方法,在本案的具体实施例中,当R1~R10为氢原子,A为-NH2,B与D同时为-OH时,将式(2)9,10-二氢-9-氧杂-10-磷菲-10-氧化物(DOPO)、式(3)4-氨基-4′-羟基二苯基甲酮(AHBP)、式(4)苯酚与酸催化剂进行反应,可生成式(D′)化合物According to the above method, in the specific embodiment of this case, when R 1 ~ R 10 are hydrogen atoms, A is -NH 2 , and B and D are both -OH, the formula (2) 9,10-dihydro- 9-oxa-10-phosphophenanthrene-10-oxide (DOPO), formula (3) 4-amino-4'-hydroxyl benzophenone (AHBP), formula (4) phenol react with acid catalyst, Can generate formula (D') compound
由于此结构中连接于磷原子的碳原子的化学键会旋转,所以所述式(D′)化合物可以式(D)化合物表示。Since the chemical bond of the carbon atom connected to the phosphorus atom in this structure is rotated, the compound of the formula (D') can be represented by the compound of the formula (D).
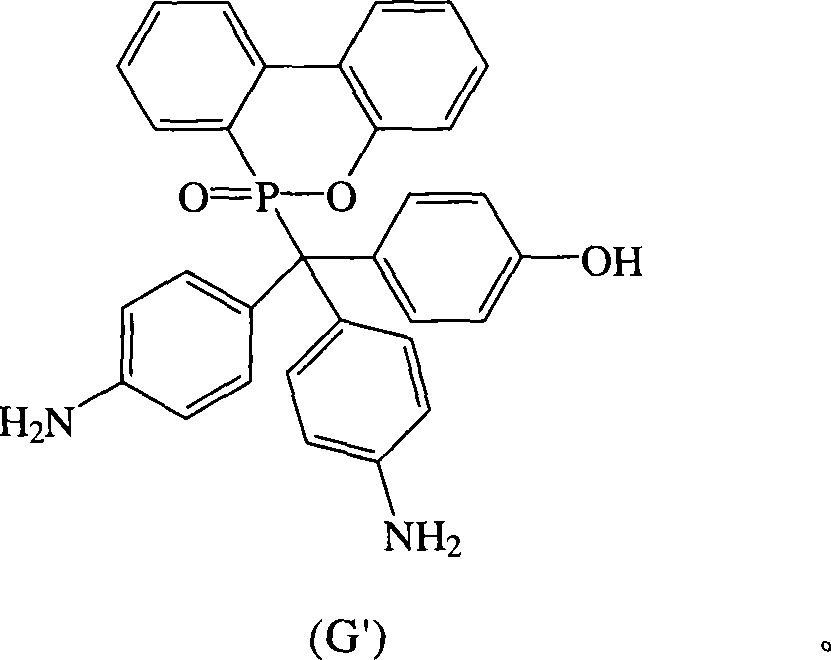
根据上述的方法,在本案的具体实施例中,当R1~R10为氢原子,A与D同时为-NH2,B为-OH时,将式(2)9,10-二氢-9-氧杂-10-磷菲-10-氧化物(DOPO)、式(3)4-氨基-4′-羟基二苯基甲酮(AHBP)、式(4)苯胺与酸催化剂进行反应,可生成式(G′)化合物According to the above method, in the specific embodiment of this case, when R 1 ~ R 10 are hydrogen atoms, A and D are both -NH 2 , and B is -OH, the formula (2) 9,10-dihydro- 9-oxa-10-phosphophenanthrene-10-oxide (DOPO), formula (3) 4-amino-4'-hydroxybenzophenone (AHBP), formula (4) aniline and acid catalyst react, Can produce formula (G') compound
同上述理由,由于此结构中连接于磷原子的碳原子的化学键会旋转,所以所述式(G′)化合物可以式(G)化合物表示。For the same reason as above, since the chemical bond of the carbon atom connected to the phosphorus atom in this structure is rotated, the compound of the formula (G') can be represented by the compound of the formula (G).
上述方法中使用的酸催化剂选自质子酸或路易斯酸所组成的群组。The acid catalyst used in the above method is selected from the group consisting of protic acid or Lewis acid.
上述方法中使用的酸催化剂选自于醋酸、对-甲基苯磺酸(p-Toluenesulfonic acid,PTSA)、甲基磺酸(Methanesulfonic acid)、钙镁试剂(Calmagite)、硫酸(Sulfuric acid)、2-氨基苯磺酸(Orthanilic acid)、3-吡啶磺酸(3-Pyridinesulfonic acid)、对氨基苯磺酸(Sulfanilic acid)、氯化氢(HCl)、溴化氢(HBr)、碘化氢(HI)、氟化氢(HF)、三氟乙酸(CF3COOH)、硝酸(HNO3)、磷酸(H3PO4)、氯化铝(AlCl3)、氟化硼(BF3)、溴化铁(FeBr3)、氯化铁(FeCl3)、氯化硼(BCl3)和氯化钛(TiCl4)所组成的群组。The acid catalyst used in the above method is selected from acetic acid, p-toluenesulfonic acid (p-Toluenesulfonic acid, PTSA), methanesulfonic acid (Methanesulfonic acid), calcium magnesium reagent (Calmagite), sulfuric acid (Sulfuric acid), 2-aminobenzenesulfonic acid (Orthanilic acid), 3-pyridinesulfonic acid (3-Pyridinesulfonic acid), p-aminobenzenesulfonic acid (Sulfanilic acid), hydrogen chloride (HCl), hydrogen bromide (HBr), hydrogen iodide (HI ), hydrogen fluoride (HF), trifluoroacetic acid (CF 3 COOH), nitric acid (HNO 3 ), phosphoric acid (H 3 PO 4 ), aluminum chloride (AlCl 3 ), boron fluoride (BF 3 ), iron bromide ( FeBr 3 ), ferric chloride (FeCl 3 ), boron chloride (BCl 3 ) and titanium chloride (TiCl 4 ).
上述方法中使用的酸催化剂用量为有机磷化物原料含量的0.1wt%~30wt%。The amount of the acid catalyst used in the above method is 0.1wt%-30wt% of the content of the organic phosphide raw material.
本发明揭示一种硬化剂,其为包含如式(1)的化合物或其混合物。The present invention discloses a curing agent, which comprises the compound of formula (1) or a mixture thereof.
本发明也揭示一种阻燃环氧树脂,其为包含环氧树脂与上述的硬化剂,其中环氧树脂可为双酚A环氧树脂(DGEBA)或磷甲基酚醛环氧树脂(CNE)。The present invention also discloses a flame-retardant epoxy resin, which comprises epoxy resin and the above-mentioned hardener, wherein the epoxy resin can be bisphenol A epoxy resin (DGEBA) or phosphorous methyl novolac epoxy resin (CNE) .
本发明也提出一种制备阻燃环氧树脂的方法,其包括将环氧树脂与上述的硬化剂以当量比例1∶0.1至1∶1均匀混合,接着进行固化后,可得到硬化的阻燃环氧树脂。The present invention also proposes a method for preparing a flame-retardant epoxy resin, which includes uniformly mixing the epoxy resin and the above-mentioned hardener in an equivalent ratio of 1:0.1 to 1:1, and then curing to obtain a hardened flame-retardant epoxy resin. epoxy resin.
在本案的制备阻燃环氧树脂的具体实施例中,使用的环氧树脂为双酚A环氧树脂(DGEBA)与磷甲基酚醛环氧树脂(CNE)。In the specific example of preparing the flame-retardant epoxy resin in this case, the epoxy resins used are bisphenol A epoxy resin (DGEBA) and phosphorus methyl novolac epoxy resin (CNE).
附图说明 Description of drawings
第1图为A的1H与13CNMR光谱图。Figure 1 is the 1 H and 13 CNMR spectrum of A.
第2图为A的31P NMR光谱图。Figure 2 is the 31 P NMR spectrum of A.
第3图为D的1H与13CNMR光谱图。Figure 3 is the 1 H and 13 CNMR spectrum of D.
第4图为D的31P NMR光谱图。Figure 4 is the 31 P NMR spectrum of D.
第5图为DGEBA系列硬化物的DSC扫描图。Figure 5 is the DSC scan of DGEBA series hardened products.
第6图为CNE系列硬化物的DSC扫描图。Figure 6 is a DSC scan of CNE series hardened products.
第7图为DGEBA系列的DMA曲线图。Figure 7 is the DMA curve of the DGEBA series.
第8图为CNE系列的DMA曲线图。Figure 8 is the DMA curve of the CNE series.
具体实施方式 Detailed ways
以下实施例将对本发明作进一步的说明,并非用以限制本发明的范围,任何所属领域的技术人员,在不违背本发明的精神下所得以达成的修饰和变化,均属本发明的范围。The following examples will further illustrate the present invention, and are not intended to limit the scope of the present invention. Any modifications and changes achieved by those skilled in the art without departing from the spirit of the present invention all belong to the scope of the present invention.
实施例Example
以上的相关发明的实施,可用流程1表示,并且我们将以下列的具体实施例说明。The implementation of the above related inventions can be represented by
流程1
实例1Example 1
化合物A的合成Synthesis of Compound A
化合物A是以DHBP,DOPO与苯酚在酸性催化剂催化下反应而得,其合成步骤如下:在附有温度指示装置的1升三颈反应器中,加入26.75克(0.125摩尔)的DHBP(4,4′-二羟基二苯甲酮)、27.00克(0.125摩尔)的DOPO(9,10-二氢-9-氧杂-10-磷菲10-氧化物)、100克的苯酚和1.10克的硫酸,升高反应温度达130℃,将反应系统温度维持在130℃,反应12小时后,滴入500毫升热水中搅拌析出,即为化合物A。Compound A is obtained by reacting DHBP, DOPO and phenol under the catalysis of an acidic catalyst. The synthesis steps are as follows: in a 1-liter three-necked reactor with a temperature indicating device, add 26.75 grams (0.125 moles) of DHBP (4, 4′-dihydroxybenzophenone), 27.00 grams (0.125 moles) of DOPO (9,10-dihydro-9-oxa-10-phosphophenanthrene 10-oxide), 100 grams of phenol and 1.10 grams of Sulfuric acid, raise the reaction temperature to 130°C, maintain the temperature of the reaction system at 130°C, react for 12 hours, drop into 500 ml of hot water and stir to precipitate, which is Compound A.
抽气过滤后,滤饼再用大量的热水洗涤,过滤分离,滤饼用真空烘箱在110℃下真空烘干,得到化合物A 54.50克,产率86%,熔点为294℃。After suction and filtration, the filter cake was washed with a large amount of hot water, separated by filtration, and dried in a vacuum oven at 110°C to obtain 54.50 g of Compound A with a yield of 86% and a melting point of 294°C.
化合物A的1H NMR与13CNMR光谱图,31P NMR光谱图分别被示于图1和图2,光谱分析的显示结果产物确实为化合物A。 The 1 H NMR and 13 CNMR spectra and 31 P NMR spectra of Compound A are shown in Figure 1 and Figure 2 respectively, and the results of spectral analysis show that the product is indeed Compound A.
实例2Example 2
化合物B的合成Synthesis of Compound B
化合物B是以DHBP,DOPO与2-甲酚在酸性催化剂催化下反应而得,其合成步骤如下:在附有温度指示装置的1升三颈反应器中,加入21.40克(0.1摩尔)的DHBP、21.60克(0.1摩尔)的DOPO、100克的2-甲酚和1.10克的硫酸,升高反应温度达130℃,将反应系统温度维持在130℃,反应12小时后,滴入100毫升甲苯中搅拌析出,得到橘红色的粘状物,移除甲苯后,利用乙醇将粘状物溶解后滴入去离子水中析出。Compound B is obtained by reacting DHBP, DOPO and 2-cresol under the catalysis of an acidic catalyst. The synthesis steps are as follows: In a 1-liter three-neck reactor with a temperature indicating device, add 21.40 grams (0.1 mole) of DHBP , 21.60 grams (0.1 moles) of DOPO, 100 grams of 2-cresol and 1.10 grams of sulfuric acid, raise the reaction temperature to 130 ° C, the temperature of the reaction system is maintained at 130 ° C, after 12 hours of reaction, drop 100 milliliters of toluene Stirring and precipitation in the medium, an orange-red sticky substance was obtained. After removing the toluene, the sticky substance was dissolved with ethanol and dropped into deionized water for precipitation.
过滤分离后,滤饼用真空烘箱在100℃下真空烘干,即得到橘红色固体的化合物B35.00克,产率67%,熔点为284℃。After separation by filtration, the filter cake was vacuum-dried in a vacuum oven at 100°C to obtain 35.00 g of Compound B as an orange-red solid with a yield of 67% and a melting point of 284°C.
实例3Example 3
化合物C的合成Synthesis of Compound C
化合物C是以DHBP,DOPO与2,6-二甲基苯酚(2,6-Dimethylphenol)在酸性催化剂催化下反应而得,其合成步骤如下:在附有温度指示装置的1升三颈反应器中,加入21.40克(0.1摩尔)的DHBP、21.60克(0.1摩尔)的DOPO、100克的2,6-二甲基苯酚和1.1克的硫酸,升高反应温度达130℃,将反应系统温度维持在130℃,反应12小时后,滴入100毫升甲苯中搅拌析出,得到橘红色的粘状物,移除甲苯后,利用乙醇将粘状物溶解后滴入去离子水中析出。Compound C is obtained by reacting DHBP, DOPO and 2,6-dimethylphenol (2,6-Dimethylphenol) under the catalysis of an acidic catalyst. In, add 21.40 grams (0.1 mole) of DHBP, 21.60 grams (0.1 mole) of DOPO, 100 grams of 2,6-dimethylphenol and 1.1 grams of sulfuric acid, increase the reaction temperature to 130 ° C, the reaction system temperature Maintained at 130°C, reacted for 12 hours, dropped into 100 ml of toluene and stirred to precipitate, and obtained an orange sticky substance. After removing the toluene, the sticky substance was dissolved with ethanol and dropped into deionized water to precipitate.
过滤分离后,滤饼用真空烘箱在100℃下真空烘干,即得到橘红色固体的化合物C40.00克,产率75%,熔点为291℃。After separation by filtration, the filter cake was vacuum-dried in a vacuum oven at 100°C to obtain 40.00 g of Compound C as an orange-red solid with a yield of 75% and a melting point of 291°C.
实例4Example 4
化合物D的合成Synthesis of Compound D
化合物D是以DHBP,DOPO与苯胺在酸性催化剂催化下反应而得,其合成步骤如下:在附有温度指示装置的1升三颈反应器中,加入21.40克(0.1摩尔)的DHBP、21.60克(0.1摩尔)的DOPO、100克的苯胺和4.30克的对-甲基苯磺酸(p-Toluenesulfonic Acid,PTSA),升高反应温度达130℃,将反应系统温度维持在130℃,反应12小时后,滴入100毫升乙腈(acetonitrile)中搅拌析出,得到黄色的固体,即为化合物D。Compound D is obtained by reacting DHBP, DOPO and aniline under the catalysis of an acidic catalyst. The synthesis steps are as follows: In a 1-liter three-necked reactor with a temperature indicating device, add 21.40 grams (0.1 moles) of DHBP, 21.60 grams (0.1 mole) of DOPO, 100 grams of aniline and 4.30 grams of p-toluenesulfonic acid (p-Toluenesulfonic Acid, PTSA), raising the reaction temperature up to 130 ° C, the reaction system temperature is maintained at 130 ° C,
过滤分离后,再利用乙腈加热洗涤,抽气过滤后,滤饼用真空烘箱在100℃下真空烘干,即得到白色固体的化合物D 34.00克,产率68%,熔点为300℃。After separation by filtration, heating and washing with acetonitrile, suction and filtration, the filter cake was vacuum-dried in a vacuum oven at 100°C to obtain 34.00 g of compound D as a white solid with a yield of 68% and a melting point of 300°C.
化合物D的1H NMR与13C NMR光谱图,31P NMR光谱图分别被示于图3与图4。 The 1 H NMR, 13 C NMR spectrum and 31 P NMR spectrum of compound D are shown in Fig. 3 and Fig. 4, respectively.
实例5Example 5
化合物E的合成Synthesis of Compound E
化合物E是以DHBP,DOPO与邻-甲苯胺在酸性催化剂催化下反应而得,其合成步骤如下:在附有温度指示装置的1升三颈反应器中,加入21.40克(0.1摩尔)的DHBP、21.60克(0.1摩尔)的DOPO、100克的邻-甲苯胺和4.30克的对-甲基苯磺酸(p-Toluenesulfonic Acid,PTSA),升高反应温度达130℃,将反应系统温度维持在130℃,反应12小时后,滴入100毫升甲苯中搅拌析出,得到褐色的粘状物,移除甲苯后,利用乙醇将粘状物溶解后滴入去离子水中析出。Compound E is obtained by reacting DHBP, DOPO and o-toluidine under the catalysis of an acidic catalyst. The synthesis steps are as follows: In a 1-liter three-neck reactor with a temperature indicating device, add 21.40 grams (0.1 mole) of DHBP , 21.60 grams (0.1 moles) of DOPO, 100 grams of o-toluidine and 4.30 grams of p-toluenesulfonic acid (p-Toluenesulfonic Acid, PTSA), raising the reaction temperature up to 130 ° C, the reaction system temperature is maintained After reacting at 130° C. for 12 hours, drop into 100 ml of toluene and stir to precipitate to obtain a brown sticky substance. After removing the toluene, dissolve the sticky substance with ethanol and drop into deionized water to precipitate.
过滤分离后,再利用乙腈加热洗涤,抽气过滤后,滤饼用真空烘箱在100℃下真空烘干,即得到粉紫色固体的化合物E 37.00克,产率72%,熔点为283℃。After separation by filtration, heating and washing with acetonitrile, suction and filtration, the filter cake was vacuum-dried in a vacuum oven at 100°C to obtain 37.00 g of Compound E as a pinkish-purple solid with a yield of 72% and a melting point of 283°C.
实例6Example 6
化合物F的合成Synthesis of Compound F
化合物F是以DHBP,DOPO与2,6-二甲基苯胺(2,6-Dimethylaniline)在酸性催化剂催化下反应而得,其合成步骤如下:在附有温度指示装置的1升三颈反应器中,加入21.40克(0.1摩尔)的DHBP、21.60克(0.1摩尔)的DOPO、100克的2,6-二甲基苯胺和4.30克的对-甲基苯磺酸(p-Toluenesulfonic Acid,PTSA),升高反应温度达130℃,将反应系统温度维持在130℃,反应12小时后,滴入100毫升甲苯中搅拌析出,得到褐色的粘状物,移除甲苯后,利用乙醇将粘状物溶解后滴入去离子水中析出。Compound F is obtained by reacting DHBP, DOPO and 2,6-dimethylaniline (2,6-Dimethylaniline) under the catalysis of an acidic catalyst. The synthesis steps are as follows: in a 1-liter three-neck reactor with a temperature indicating device In, add 21.40 grams (0.1 moles) of DHBP, 21.60 grams (0.1 moles) of DOPO, 100 grams of 2,6-dimethylaniline and 4.30 grams of p-toluenesulfonic acid (p-Toluenesulfonic Acid, PTSA ), raise the reaction temperature to 130°C, and maintain the temperature of the reaction system at 130°C. After 12 hours of reaction, drop into 100 ml of toluene and stir to precipitate to obtain a brown sticky substance. After removing the toluene, use ethanol to dissolve the sticky After the substance was dissolved, it was dropped into deionized water to precipitate.
过滤分离后,再利用乙腈加热洗涤,抽气过滤后,滤饼用真空烘箱在100℃下真空烘干,即得到淡黄色固体的化合物F 27.00克,产率50%,熔点为292℃。After separation by filtration, heating and washing with acetonitrile, suction and filtration, the filter cake was vacuum-dried in a vacuum oven at 100°C to obtain 27.00 g of Compound F as a pale yellow solid with a yield of 50% and a melting point of 292°C.
实例7Example 7
化合物G的合成Synthesis of Compound G
化合物G是以DABP,DOPO与苯酚在酸性催化剂催化下反应而得,其合成步骤如下:在附有温度指示装置的1升三颈反应器中,加入26.53克(0.125摩尔)的DABP、27.00克(0.125摩尔)的DOPO、25.00克的苯酚和1.10克的硫酸,升高反应温度达130℃,将反应系统温度维持在130℃,反应12小时后。滴入500毫升热水中搅拌,即可析出橘色的固体,即为化合物G。Compound G is obtained by reacting DABP, DOPO and phenol under the catalysis of an acidic catalyst. The synthesis steps are as follows: In a 1-liter three-neck reactor with a temperature indicating device, add 26.53 grams (0.125 moles) of DABP, 27.00 grams (0.125 moles) of DOPO, 25.00 grams of phenol and 1.10 grams of sulfuric acid, raise the reaction temperature to 130°C, maintain the temperature of the reaction system at 130°C, and react for 12 hours. Drop into 500 ml of hot water and stir to precipitate an orange solid, which is compound G.
过滤分离后,滤饼用大量的热水洗涤,抽气过滤后,滤饼用真空烘箱在110℃下真空烘干,得到化合物G 45.40克,产率72%,熔点为298℃。After filtration and separation, the filter cake was washed with a large amount of hot water, and after suction and filtration, the filter cake was vacuum-dried in a vacuum oven at 110°C to obtain 45.40 g of compound G with a yield of 72% and a melting point of 298°C.
实例8Example 8
化合物H的合成Synthesis of Compound H
化合物H是以DABP,DOPO与2-甲酚在酸性催化剂催化下反应而得,其合成步骤如下:在附有温度指示装置的1升三颈反应器中,加入26.53克(0.125摩尔)的DABP、27.00克(0.125摩尔)的DOPO、100克的2-甲酚和1.10克的硫酸,升高反应温度达130℃,将反应系统温度维持在130℃,反应12小时后,滴入100毫升甲苯中搅拌析出,得到褐色的粘状物,移除甲苯后,利用乙醇将粘状物溶解后滴入去离子水中析出。Compound H is obtained by reacting DABP, DOPO and 2-cresol under the catalysis of an acidic catalyst. The synthesis steps are as follows: In a 1-liter three-neck reactor with a temperature indicating device, add 26.53 grams (0.125 moles) of DABP , 27.00 grams (0.125 moles) of DOPO, 100 grams of 2-cresol and 1.10 grams of sulfuric acid, raising the reaction temperature to 130 ° C, the temperature of the reaction system is maintained at 130 ° C, after 12 hours of reaction, drop 100 milliliters of toluene Stirring and precipitation in medium, a brown sticky substance was obtained. After removing the toluene, the sticky substance was dissolved with ethanol and dropped into deionized water to precipitate.
过滤分离后,滤饼用真空烘箱在100℃下真空烘干,即得到粉红色固体的化合物H33.70克,产率65%,熔点为287℃。After separation by filtration, the filter cake was vacuum-dried in a vacuum oven at 100°C to obtain 3.70 g of compound H3 as a pink solid with a yield of 65% and a melting point of 287°C.
实例9Example 9
化合物I的合成Synthesis of Compound I
化合物I是以DABP,DOPO与2,6-二甲基苯酚(2,6-Dimethylphenol)在酸性催化剂催化下反应而得,其合成步骤如下:在附有温度指示装置的1升三颈反应器中,加入26.53克(0.125摩尔)的DABP、27.00克(0.125摩尔)的DOPO、100克的2,6-二甲基苯酚和1.10克的硫酸,升高反应温度达130℃,将反应系统温度维持在130℃,反应12小时后,滴入100毫升甲苯中搅拌析出,得到褐色的粘状物,移除甲苯后,利用乙醇将粘状物溶解后滴入去离子水中析出。Compound I is obtained by reacting DABP, DOPO and 2,6-dimethylphenol (2,6-Dimethylphenol) under the catalysis of an acidic catalyst. In, add 26.53 grams (0.125 moles) of DABP, 27.00 grams (0.125 moles) of DOPO, 100 grams of 2,6-dimethylphenol and 1.10 grams of sulfuric acid, raise the reaction temperature to 130 ° C, the reaction system temperature Maintained at 130°C, reacted for 12 hours, dropped into 100 ml of toluene and stirred to precipitate to obtain a brown sticky substance. After removing the toluene, the sticky substance was dissolved with ethanol and dropped into deionized water to precipitate.
过滤分离后,滤饼用真空烘箱在100℃下真空烘干,即得到紫色固体的化合物I 40.40克,产率76%,熔点为293℃。After separation by filtration, the filter cake was vacuum-dried in a vacuum oven at 100°C to obtain 40.40 g of Compound I as a purple solid with a yield of 76% and a melting point of 293°C.
实例10Example 10
化合物J的合成Synthesis of Compound J
化合物J是以DABP,DOPO与苯胺在酸性催化剂催化下反应而得,其合成步骤如下:在附有温度指示装置的1升三颈反应器中,加入26.53克(0.125摩尔)的DABP、27.00克(0.125摩尔)的DOPO、100.00克的苯胺和4.30克的PTSA,升高反应温度达130℃,将反应系统温度维持在130℃,反应12小时后,滴入100毫升乙腈中搅拌析出,得到粉紫色的固体,即为化合物J。Compound J is obtained by reacting DABP, DOPO and aniline under the catalysis of an acidic catalyst. The synthesis steps are as follows: In a 1-liter three-neck reactor with a temperature indicating device, add 26.53 grams (0.125 moles) of DABP, 27.00 grams (0.125 moles) of DOPO, 100.00 grams of aniline and 4.30 grams of PTSA, raising the reaction temperature to 130°C, maintaining the temperature of the reaction system at 130°C, and reacting for 12 hours, dropping it into 100 milliliters of acetonitrile and stirring to precipitate to obtain powder The purple solid is compound J.
过滤分离后,再利用乙腈加热洗涤,抽气过滤后,放入真空烘箱在100℃下烘干,得到化合物J 29.00克,产率57%,熔点为330℃。After separation by filtration, heating and washing with acetonitrile, suction and filtration, drying in a vacuum oven at 100°C to obtain 29.00 g of Compound J with a yield of 57% and a melting point of 330°C.
实例11Example 11
化合物K的合成Synthesis of Compound K
化合物K是以DABP,DOPO与邻-甲苯胺在酸性催化剂催化下反应而得,其合成步骤如下:在附有温度指示装置的1升三颈反应器中,加入26.53克(0.125摩尔)的DABP、27.00克(0.125摩尔)的DOPO、100.00克的邻-甲苯胺和4.30克的PTSA,升高反应温度达130℃,将反应系统温度维持在130℃,反应12小时后,滴入100毫升甲苯中搅拌析出,得到褐色的粘状物,移除甲苯后,利用乙醇将粘状物溶解后滴入去离子水中析出。Compound K is obtained by reacting DABP, DOPO and o-toluidine under the catalysis of an acidic catalyst. The synthesis steps are as follows: In a 1-liter three-neck reactor with a temperature indicating device, add 26.53 grams (0.125 moles) of DABP , 27.00 grams (0.125 moles) of DOPO, 100.00 grams of o-toluidine and 4.30 grams of PTSA, raising the reaction temperature to 130 ° C, the temperature of the reaction system was maintained at 130 ° C, after 12 hours of reaction, drop 100 milliliters of toluene Stirring and precipitation in medium, a brown sticky substance was obtained. After removing the toluene, the sticky substance was dissolved with ethanol and dropped into deionized water to precipitate.
过滤分离后,再利用乙腈加热洗涤,抽气过滤后,放入真空烘箱在100℃下烘干,得到化合物K 32.00克,产率62%,熔点为323℃。After separation by filtration, heating and washing with acetonitrile, suction and filtration, drying in a vacuum oven at 100°C to obtain 32.00 g of Compound K with a yield of 62% and a melting point of 323°C.
实例12Example 12
化合物L的合成Synthesis of Compound L
化合物L是以DABP,DOPO与2,6-二甲基苯胺在酸性催化剂催化下反应而得,其合成步骤如下:在附有温度指示装置的1升三颈反应器中,加入26.53克(0.1摩尔)的DABP、21.60克(0.1摩尔)的DOPO、100.00克的2,6-二甲基苯胺和4.30克的PTSA,升高反应温度达130℃,将反应系统温度维持在130℃,反应12小时后,滴入100毫升甲苯中搅拌析出,得到褐色的粘状物,移除甲苯后,利用乙醇将粘状物溶解后滴入去离子水中析出。Compound L is obtained by reacting DABP, DOPO and 2,6-dimethylaniline under the catalysis of an acidic catalyst. The synthesis steps are as follows: in a 1-liter three-neck reactor with a temperature indicating device, add 26.53 grams (0.1 moles) of DABP, 21.60 grams (0.1 moles) of DOPO, 100.00 grams of 2,6-dimethylaniline and 4.30 grams of PTSA, raising the reaction temperature to 130°C, maintaining the temperature of the reaction system at 130°C, and reacting 12 Hours later, it was dropped into 100 ml of toluene and stirred to precipitate to obtain a brown sticky substance. After removing the toluene, the sticky substance was dissolved with ethanol and dropped into deionized water to precipitate.
过滤分离后,再利用乙腈加热洗涤,抽气过滤后,放入真空烘箱在100℃下烘干,得到化合物L 27.60克,产率52%,熔点为329℃。After separation by filtration, heating and washing with acetonitrile, suction and filtration, drying in a vacuum oven at 100°C to obtain 27.60 g of compound L with a yield of 52% and a melting point of 329°C.
实例13Example 13
化合物D的合成Synthesis of Compound D
化合物D也可以AHBP,DOPO与苯酚在酸性催化剂催化下反应而得,其合成步骤如下:在附有温度指示装置的1升三颈反应器中,加入21.30克(0.1摩尔)的AHBP、21.60克(0.1摩尔)的DOPO、100.00克的苯酚和0.86克的硫酸,加入1升反应器中,升高反应温度达130℃,将反应系统温度维持在130℃,反应12小时后,滴入100毫升乙腈中搅拌析出,即可析出黄色的固体,即为化合物D。Compound D can also be obtained by reacting AHBP, DOPO and phenol under the catalysis of an acidic catalyst. The synthesis steps are as follows: In a 1-liter three-neck reactor with a temperature indicating device, add 21.30 grams (0.1 mole) of AHBP, 21.60 grams (0.1 mole) of DOPO, 100.00 g of phenol and 0.86 g of sulfuric acid were added to a 1-liter reactor, and the reaction temperature was increased to 130° C., and the temperature of the reaction system was maintained at 130° C. After 12 hours of reaction, 100 ml of Stir and precipitate in acetonitrile to precipitate a yellow solid, which is compound D.
抽气过滤后,滤饼用乙腈加热洗涤,过滤分离后,滤饼用真空烘箱在100℃下真空烘干,即得到白色固体的化合物D 30.00克,产率60%,熔点为300℃。After suction and filtration, the filter cake was heated and washed with acetonitrile. After filtration and separation, the filter cake was vacuum-dried in a vacuum oven at 100°C to obtain 30.00 g of compound D as a white solid with a yield of 60% and a melting point of 300°C.
产物在DMSO-D6溶液中的1H与13CNMR图谱,与第1图所示完全符合,显示结果为化合物D结构无误。The 1 H and 13 CNMR spectra of the product in DMSO-D6 solution are completely consistent with those shown in Figure 1, showing that the structure of compound D is correct.
实例14Example 14
化合物G的合成Synthesis of Compound G
化合物G也可以AHBP,DOPO与苯胺在酸性催化剂催化下反应而得,其合成步骤如下:在附有温度指示装置的1升三颈反应器中,加入21.30克(0.1摩尔)的AHBP、21.60克(0.1摩尔)的DOPO、25.00克的苯胺和4.32克的H2SO4,加入1升反应器中,升高反应温度达130℃,将反应系统温度维持在130℃,反应12小时后,滴入500毫升热水中搅拌,即可析出橘色的固体,即为化合物G。Compound G can also be obtained by reacting AHBP, DOPO and aniline under the catalysis of an acidic catalyst. The synthesis steps are as follows: In a 1-liter three-neck reactor with a temperature indicating device, add 21.30 g (0.1 mole) of AHBP, 21.60 g (0.1 mol) of DOPO, 25.00 g of aniline and 4.32 g of H 2 SO 4 were added to a 1-liter reactor, and the reaction temperature was increased to 130° C., and the temperature of the reaction system was maintained at 130° C. After 12 hours of reaction, dropwise Stir in 500 ml of hot water to precipitate an orange solid, which is compound G.
过滤分离后,滤饼用大量的热水洗涤,抽气过滤后,滤饼用真空烘箱在110℃下真空烘干,得到化合物G 41.62克,产率66%,熔点为298℃。After filtration and separation, the filter cake was washed with a large amount of hot water, and after suction and filtration, the filter cake was vacuum-dried in a vacuum oven at 110°C to obtain 41.62 g of compound G with a yield of 66% and a melting point of 298°C.
实例15Example 15
阻燃环氧树脂的制备与分析Preparation and Analysis of Flame Retardant Epoxy Resin
将上述合成例的化合物A、化合物B和化合物E,作为环氧树脂双酚A二环氧甘油醚(DGEBA)与磷甲基酚醛环氧树脂(CNE)的硬化剂,以当量比例1∶1均匀混合,进行固化得到硬化的阻燃树脂,并通过下列分析试验比较其效果。样品代号如表1所示:With compound A, compound B and compound E of above-mentioned synthesis example, as the curing agent of epoxy resin bisphenol A diglycidyl ether (DGEBA) and phosphorus methyl novolak epoxy resin (CNE), with equivalent ratio 1: 1 Mix evenly and cure to obtain a hardened flame retardant resin, and compare its effects through the following analysis tests. Sample codes are shown in Table 1:
表1:硬化物代号Table 1: Hardened product code
热性质分析Thermal Properties Analysis
图5为DGEBA与不同硬化剂均匀混合硬化后其玻璃化转变温度的叠图,图6为CNE与不同硬化剂均匀混合硬化后其玻璃化转变温度叠图,表2与表3为上述的环氧树脂混合物在不同硬化温度下利用DSC所测得玻璃化转变温度。我们可以发现环氧树脂固化物的玻璃化转变温度比较依序为:B-DGEBA>J-DGEBA>D-DGEBA(I)>J-DGEBA(I)>A-DGEBA(I),所述结果显示添加硬化促进剂(咪唑)对环氧树脂固化物的玻璃转化温度影响不大。此外,利用化合物D与化合物J作为硬化剂的环氧树脂固化物有较高的玻璃化转变温度,这是由于化合物D与化合物J提供较多的交联点,可增加交联密度,使其具有较高的玻璃化转变温度。Figure 5 is the overlay of the glass transition temperature of DGEBA after uniform mixing and hardening with different hardeners, and Figure 6 is the overlay of glass transition temperature after uniform mixing and hardening of CNE and different hardeners, Table 2 and Table 3 are the above-mentioned rings The glass transition temperature of the epoxy resin mixture was measured by DSC at different curing temperatures. We can find that the glass transition temperature of cured epoxy resin is compared in order: B-DGEBA>J-DGEBA>D-DGEBA(I)>J-DGEBA(I)>A-DGEBA(I), the result It shows that the addition of hardening accelerator (imidazole) has little effect on the glass transition temperature of cured epoxy resin. In addition, the cured epoxy resin using Compound D and Compound J as hardeners has a higher glass transition temperature, which is because Compound D and Compound J provide more crosslinking points, which can increase the crosslinking density, making it Has a higher glass transition temperature.
另外,环氧树脂固化物的玻璃化转变温度比较依序为:D-CNE>D-CNE(I)>J-CNE>J-CNE(I)。分析结果显示是否有添加硬化促进剂影响不大。D系列环氧树脂固化物的玻璃化转变温度较J系列环氧树脂固化物高,这是由于化合物J交联过程中会受立体障碍影响,阻碍交联结构的延伸而使玻璃化转变温度较低。表2结果显示,A与J系列在220℃的硬化温度下有最高的玻璃化转变温度,在240℃的硬化温度下会产生些微裂解现象使玻璃化转变温度下降。D-DGEBA在180℃的硬化温度下有最高的玻璃化转变温度,D-DGEBA(I)由于添加硬化促进剂的关系,在140℃的硬化温度下即可达到最高的玻璃化转变温度。从分析结果显示,D系列的硬化速度比A与J系列快,在较低的硬化温度下即可达到最高的玻璃化转变温度。表3也揭示除了D-CNE(I)在220℃的硬化温度下即可达到最高的玻璃化转变温度,其它皆在240℃的硬化温度下得到最高的玻璃化转变温度。In addition, the glass transition temperature of the cured epoxy resin is compared in order: D-CNE>D-CNE(I)>J-CNE>J-CNE(I). The analysis results show that whether the addition of hardening accelerator has little effect. The glass transition temperature of D series epoxy resin cured products is higher than that of J series epoxy resin cured products. This is because compound J will be affected by steric barriers during the crosslinking process, which hinders the extension of the crosslinked structure and makes the glass transition temperature higher. Low. The results in Table 2 show that the A and J series have the highest glass transition temperature at a hardening temperature of 220°C, and a slight cracking phenomenon will occur at a hardening temperature of 240°C to lower the glass transition temperature. D-DGEBA has the highest glass transition temperature at a hardening temperature of 180°C, and D-DGEBA(I) can reach the highest glass transition temperature at a hardening temperature of 140°C due to the addition of a hardening accelerator. The analysis results show that the hardening speed of the D series is faster than that of the A and J series, and the highest glass transition temperature can be reached at a lower hardening temperature. Table 3 also reveals that except for D-CNE (I) which can achieve the highest glass transition temperature at the hardening temperature of 220° C., all others have the highest glass transition temperature at the hardening temperature of 240° C.
表2:不同硬化温度的DSC玻璃化转变温度Table 2: DSC glass transition temperature for different hardening temperatures
a:添加0.2wt%(以环氧树脂为基础)硬化促进剂(咪唑)a: Add 0.2wt% (based on epoxy resin) hardening accelerator (imidazole)
b:氮气下DSC测试值b: DSC test value under nitrogen
c:数据来自文献[1]c: data from literature [1]
d:数据来自文献[2]d: data from literature [2]
表3:不同硬化温度的DSC玻璃化转变温度Table 3: DSC glass transition temperature for different hardening temperatures
a:添加0.2wt%(以环氧树脂为基础)硬化促进剂(咪唑)a: Add 0.2wt% (based on epoxy resin) hardening accelerator (imidazole)
b:氮气下DSC测试值b: DSC test value under nitrogen
动态机械分析Dynamic Mechanical Analysis
图7与图8分别为DGEBA系列和CNE系列的DMA分析图,并将DMA数据整理于表4与表5。分析结果显示DGEBA系列环氧树脂固化物的存储模量比较依序为:J-DGEBA(I)>D-DGEBA(I)>J-DGEBA>D-DGEBA>A-DGEBA(I);CNE系列的环氧树脂固化物的存储模量比较依序:J-CNE(I)>J-CNE>D-CNE>D-CNE(I)。除了D-CNE与D-CNE(I)的存储模量差异不大以外,其它有添加硬化促进剂的环氧树脂固化物有较高的存储模量。此外,以化合物J为硬化剂的环氧树脂固化物的存储模量最大,以化合物D为硬化剂的环氧树脂固化物的存储模量次之,以化合物A为硬化剂的环氧树脂固化物的存储模量最小,推测是由于化合物A与化合物D具有OH基,在与环氧树脂交联硬化后变成较柔软的醚,所以使得所述环氧树脂固化物的存储模量较低;分析tanδ值也会得到相同的结果,因为有较柔软的交联结构相对分子间比较容易移动所以tanδ高度值比较大。由tan δ所测量到的玻璃化转变温度,结果与DSC所测量的值相差不大。Figure 7 and Figure 8 are the DMA analysis diagrams of DGEBA series and CNE series respectively, and the DMA data are organized in Table 4 and Table 5. The analysis results show that the storage modulus of DGEBA series epoxy resin cured products is compared in order: J-DGEBA(I)>D-DGEBA(I)>J-DGEBA>D-DGEBA>A-DGEBA(I); CNE series The storage modulus of cured epoxy resin is compared in order: J-CNE(I)>J-CNE>D-CNE>D-CNE(I). Except that the storage modulus of D-CNE and D-CNE(I) has little difference, other cured epoxy resins with hardening accelerators have higher storage modulus. In addition, the storage modulus of the epoxy resin cured product using compound J as the hardener is the largest, followed by the storage modulus of the epoxy resin cured product using compound D as the hardener, and the epoxy resin cured product using compound A as the hardener The storage modulus of the compound is the smallest, presumably because compound A and compound D have OH groups, which become softer ethers after cross-linking and hardening with epoxy resin, so the storage modulus of the cured epoxy resin is lower ; Analyzing the tanδ value will also get the same result, because the softer cross-linked structure is relatively easy to move between molecules, so the tanδ height value is relatively large. The glass transition temperature measured by tan δ is not much different from the value measured by DSC.
表4:DGEBA系列动态机械分析结果Table 4: DGEBA series dynamic mechanical analysis results
a:tanδ曲线的波峰高度a: peak height of tanδ curve
b:到达玻璃化转变温度时的tanδ值b: tanδ value when reaching the glass transition temperature
表5:CNE系列动态机械分析结果Table 5: Dynamic Mechanical Analysis Results of CNE Series
a:tanδ曲线的波峰高度a: peak height of tanδ curve
b:到达玻璃化转变温度时的tanδ值b: tanδ value when reaching the glass transition temperature
UL-94测试UL-94 test
UL-94是一种测试阻燃性的方法,聚合物样品须经过两次各历时10秒的燃烧过程。第一次燃烧后,将火焰移开并记录聚合物自行熄灭所需的时间(t1)。如果聚合物在测试期间有滴落现象(dripping),待样品冷却后,进行第二次燃烧,并记录聚合物自行熄灭所需的时间(t2)和滴落现象。如果t1+t2少于10秒且无滴落现象,那么根据阻燃性的工业标准(industrial standard for flame-retardancy)归类为V0等级,如果t1+t2为10~30秒那么属V1等级。UL-94 is a method of testing flame retardancy in which a polymer sample is subjected to two burns of 10 seconds each. After the first burn, the flame was removed and the time (t 1 ) required for the polymer to extinguish itself was recorded. If the polymer has dripping during the test, after the sample is cooled, carry out the second combustion, and record the time (t 2 ) and dripping required for the polymer to extinguish itself. If t 1 + t 2 is less than 10 seconds and there is no dripping phenomenon, it is classified as V0 according to the industrial standard for flame-retardancy. If t 1 + t 2 is 10 to 30 seconds, then Belongs to V1 level.
将两种硬化剂化合物D与DDS(4,4′-二氨基二苯基砜)分别与DGEBA和CNE配置磷含量为1%、1.5%和1.8%,在150℃熔化均匀搅拌后,倒入铝制模具中,模具置于循环烘箱中阶段升温最高到220℃硬化,将硬化完成的试片做UL-94阻燃测试。表6为UL-94测试结果,环氧树脂固化物的阻燃性随磷含量增加而提高,磷含量1.5%可达到V0等级。Prepare the two hardener compounds D and DDS (4,4'-diaminodiphenyl sulfone) with DGEBA and CNE respectively, with phosphorus content of 1%, 1.5% and 1.8%, melt at 150°C and stir evenly, pour into In the aluminum mold, the mold is placed in a circulating oven and the temperature is raised up to 220°C to harden, and the hardened test piece is tested for UL-94 flame retardancy. Table 6 shows the test results of UL-94. The flame retardancy of cured epoxy resin increases with the increase of phosphorus content, and the phosphorus content of 1.5% can reach the V0 level.
表6:UL-94测试结果Table 6: UL-94 test results
a:UL-94测试结果平均值a: Average value of UL-94 test results
以上权利要求书是用以界定本发明的合理保护范围。但是应了解,技术人员基于本发明的揭示所可达成的种种显而易见的改进,也应归属本发明合理的保护范围。The above claims are used to define the reasonable protection scope of the present invention. However, it should be understood that various obvious improvements achieved by the skilled person based on the disclosure of the present invention should also belong to the reasonable protection scope of the present invention.
Claims (13)
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN 200810094205 CN101560226B (en) | 2008-04-18 | 2008-04-18 | Novel phosphorous compound, its production method and use |
| HK10103843.1A HK1135402B (en) | 2010-04-21 | Novel phosphorus-containing compounds and their preparing process and use |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN 200810094205 CN101560226B (en) | 2008-04-18 | 2008-04-18 | Novel phosphorous compound, its production method and use |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN101560226A CN101560226A (en) | 2009-10-21 |
| CN101560226B true CN101560226B (en) | 2013-01-09 |
Family
ID=41219225
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN 200810094205 Expired - Fee Related CN101560226B (en) | 2008-04-18 | 2008-04-18 | Novel phosphorous compound, its production method and use |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN101560226B (en) |
Families Citing this family (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102127205B (en) * | 2010-01-13 | 2013-11-06 | 长春人造树脂厂股份有限公司 | Phosphorus-containing epoxy resin semi-cured material and preparation method thereof |
| CN103119049A (en) * | 2010-09-22 | 2013-05-22 | 宇部兴产株式会社 | Organic phosphorus compound and method for producing same |
| CN104370966B (en) * | 2014-10-07 | 2017-03-29 | 北京理工大学 | A kind of metallo-organic compound and preparation method thereof |
| CN106905561B (en) * | 2017-03-02 | 2019-08-23 | 张家港市山牧新材料技术开发有限公司 | A kind of fluorine-containing DOPO derivative flame retardant and preparation method thereof |
| CN109265674B (en) * | 2017-07-18 | 2021-02-26 | 臻鼎科技股份有限公司 | Modified polyphenyl ether high-molecular polymer, high-molecular film and corresponding preparation method |
| CN111777642B (en) * | 2020-09-04 | 2020-12-11 | 中国科学院宁波材料技术与工程研究所 | Halogen-free flame retardant unsaturated polyester composition, polyester product, preparation method and application thereof |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1612244A1 (en) * | 2003-03-25 | 2006-01-04 | Sanko Co., Ltd. | Flame resistant synthetic resin composition |
| US20080045688A1 (en) * | 2006-08-17 | 2008-02-21 | National Chunghsing University | Processes of synthesizing aromatic amine-based benzoxazine resins |
-
2008
- 2008-04-18 CN CN 200810094205 patent/CN101560226B/en not_active Expired - Fee Related
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1612244A1 (en) * | 2003-03-25 | 2006-01-04 | Sanko Co., Ltd. | Flame resistant synthetic resin composition |
| US20080045688A1 (en) * | 2006-08-17 | 2008-02-21 | National Chunghsing University | Processes of synthesizing aromatic amine-based benzoxazine resins |
Non-Patent Citations (1)
| Title |
|---|
| Lin, Ching Hsuan et al..Flame-retardant epoxy resins with high glass-transition temperatures. II.Using a novel hexafunctional curing agent:9,10-dihydro-9-oxa-10-phosphaphenanthrene 10-yl-tris(4-aminophenyl)methane.《Journal of Polymer Science, Part A: Polymer Chemistry》.2005,第43卷(第23期),5971-5986. * |
Also Published As
| Publication number | Publication date |
|---|---|
| HK1135402A1 (en) | 2010-06-04 |
| CN101560226A (en) | 2009-10-21 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| TWI397533B (en) | Novel phosphorus-containing compounds and their preparing process and use | |
| TWI432445B (en) | Monofunctional, bifunctional, and multifunctional phosphinated phenols and their derivatives and preparation method thereof | |
| CN101560226B (en) | Novel phosphorous compound, its production method and use | |
| CN101880395B (en) | Polymer type phosphorous flame retardant containing DOPO and preparation method thereof | |
| Ho et al. | Thermal degradation kinetics and flame retardancy of phosphorus-containing dicyclopentadiene epoxy resins | |
| CN113234103A (en) | Phosphazene flame retardant and preparation method and application thereof | |
| TWI460186B (en) | Phosphinated amino and phenolic-containing multifunctional epoxy curing agents and their preparation and use | |
| CN103421192B (en) | Phosphorus-containing benzoxazine resin and composition | |
| CN105218789A (en) | The flame retardant curing agent of phosphorous and P-C bond structure, its preparation method and the fire retarding epoxide resin being raw material with it | |
| Wang et al. | Preparation, mechanical properties, and thermal degradation of flame retarded epoxy resins with an organophosphorus oligomer | |
| TW200842138A (en) | Flame retardant crosslink agent and epoxy resin compositions free of halogen and phosphor | |
| TWI417298B (en) | Multifunctional phosphorus-containing compounds as flame-retardancy curing agent and their preparing process and use | |
| TWI310401B (en) | ||
| KR101492996B1 (en) | Phosphorous-containing copolymer resin and flame-retardant composition using the same | |
| TWI443182B (en) | Manufacturing method of flame retardant and phosphorous-modified hardener | |
| CN113214454B (en) | Transparent halogen-free flame-retardant epoxy resin, material and preparation method thereof | |
| HK1135402B (en) | Novel phosphorus-containing compounds and their preparing process and use | |
| CN109836615B (en) | Diepoxy functional urushiol-based phosphorus-containing flame retardant, preparation method and application thereof | |
| TWI465457B (en) | Amino-containing multifunctional epoxy curing agents and their derivatives, preparation and use | |
| CN113582939B (en) | Secondary amino benzoxazine monomer and application thereof | |
| TWI522396B (en) | Phosphinated multifunctional phenol polymer and its epoxy thermosets with high glass transition temperature | |
| TWI494297B (en) | Styrenic phosphinated phenol, epoxy thermoset and manufacturing method thereof | |
| TWI439466B (en) | New multifunctional phosphorus-containing phenols and their derivatives and preparation method thereof | |
| CN120665107A (en) | Preparation method and application of P-N synergistic halogen-free flame retardant curing agent | |
| CN117069917A (en) | Preparation method of low dielectric loss and high-efficiency flame-retardant epoxy resin |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| REG | Reference to a national code |
Ref country code: HK Ref legal event code: DE Ref document number: 1135402 Country of ref document: HK |
|
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| REG | Reference to a national code |
Ref country code: HK Ref legal event code: GR Ref document number: 1135402 Country of ref document: HK |
|
| CF01 | Termination of patent right due to non-payment of annual fee |
Granted publication date: 20130109 Termination date: 20180418 |
|
| CF01 | Termination of patent right due to non-payment of annual fee |