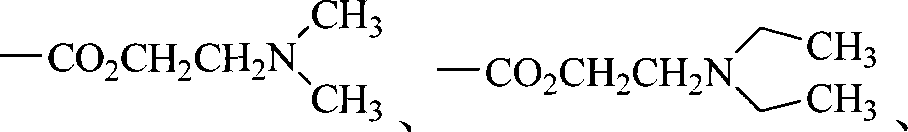CN101497602B - Anthranilic acid compound and use thereof - Google Patents
Anthranilic acid compound and use thereof Download PDFInfo
- Publication number
- CN101497602B CN101497602B CN2008100571021A CN200810057102A CN101497602B CN 101497602 B CN101497602 B CN 101497602B CN 2008100571021 A CN2008100571021 A CN 2008100571021A CN 200810057102 A CN200810057102 A CN 200810057102A CN 101497602 B CN101497602 B CN 101497602B
- Authority
- CN
- China
- Prior art keywords
- compound
- general formula
- con
- conhch
- compounds
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
- VWSLLSXLURJCDF-UHFFFAOYSA-N CC1=NCCN1 Chemical compound CC1=NCCN1 VWSLLSXLURJCDF-UHFFFAOYSA-N 0.000 description 1
- GUXJXWKCUUWCLX-UHFFFAOYSA-N CC1=NCCO1 Chemical compound CC1=NCCO1 GUXJXWKCUUWCLX-UHFFFAOYSA-N 0.000 description 1
- JUIQOABNSLTJSW-UHFFFAOYSA-N CC1=NCCS1 Chemical compound CC1=NCCS1 JUIQOABNSLTJSW-UHFFFAOYSA-N 0.000 description 1
- LXBGSDVWAMZHDD-UHFFFAOYSA-N Cc1ncc[nH]1 Chemical compound Cc1ncc[nH]1 LXBGSDVWAMZHDD-UHFFFAOYSA-N 0.000 description 1
- ZCHCHJQEWYIJDQ-UHFFFAOYSA-N Cc1ncc[o]1 Chemical compound Cc1ncc[o]1 ZCHCHJQEWYIJDQ-UHFFFAOYSA-N 0.000 description 1
- VZWOXDYRBDIHMA-UHFFFAOYSA-N Cc1ncc[s]1 Chemical compound Cc1ncc[s]1 VZWOXDYRBDIHMA-UHFFFAOYSA-N 0.000 description 1
Landscapes
- Plural Heterocyclic Compounds (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
Abstract
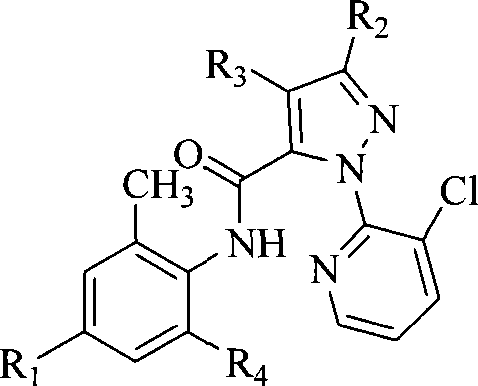
本发明公开了一种邻氨基苯甲酸类化合物,结构如通式I所示:式中各取代基的定义见说明书。本发明的邻氨基苯甲酸类化合物具有广谱杀虫活性,特别是对甜菜夜蛾、小菜蛾的活性更好,在较低的剂量下就可以获得很好的防治效果。The invention discloses an anthranilic acid compound, the structure of which is shown in the general formula I: For the definition of each substituent in the formula, see the description. The anthranilic acid compound of the present invention has broad-spectrum insecticidal activity, especially better activity on beet armyworm and diamondback moth, and can obtain good control effect at a lower dose.
Description
技术领域technical field
本发明属于杀虫剂领域,具体地涉及一种邻氨基苯甲酸类化合物、及N-氧化物、盐和组合物,以及使用它们在农业或其他领域中作为杀虫剂的应用。The invention belongs to the field of insecticides, and in particular relates to an anthranilic acid compound, N-oxide, salt and composition, and their application as insecticides in agriculture or other fields.
背景技术Background technique
无脊椎动物害虫的防治在实现高种植效率中极为重要。无脊椎动物害虫对生长和贮存的农作物的损害会引起生产率的显著降低,并因此导致消费者的花费增加。用于这些目的的许多产品是可购买的,但是仍然需要更有效、低成本、低毒型、对环境安全或具有不同作用方式的新化合物。邻氨基苯甲酸类化合物(鱼尼丁受体类)是近几年开发的防治无脊椎动物害虫的有效杀虫剂。Control of invertebrate pests is extremely important in achieving high planting efficiency. Damage to growing and stored crops by invertebrate pests can cause significant reductions in productivity and, consequently, increased costs to consumers. Many products are commercially available for these purposes, but there is still a need for new compounds that are more effective, low cost, less toxic, environmentally safe or have a different mode of action. Anthranilic acid compounds (ryanodine receptors) are effective insecticides developed in recent years to control invertebrate pests.
专利WO03015519中公开了如下具有杀虫活性的化合物:Patent WO03015519 discloses the following compounds with insecticidal activity:
专利WO2004033468中公开了如下具有杀虫活性的化合物:Patent WO2004033468 discloses the following compounds with insecticidal activity:
专利WO2004067528中公开了如下具有杀虫活性的化合物:Patent WO2004067528 discloses the following compounds with insecticidal activity:
以上专利(申请)中所有公开的化合物虽与本发明化合物有相似之处,但结构仍存在显著的不同。Although all the compounds disclosed in the above patents (applications) have similarities with the compounds of the present invention, there are still significant differences in their structures.
发明内容Contents of the invention
本发明的目的在于提供一种在很小的剂量下就可以控制多种病虫害的邻氨基苯甲酸类化合物,其N-氧化物或其盐,它可应用于在农学和非农学环境中防治无脊椎动物害虫。The object of the present invention is to provide a kind of anthranilic acid compound that just can control various diseases and insect pests under very small dosage, its N-oxide compound or its salt, it can be applied in the prevention and treatment of pest in agronomy and non-agronomy environment. Vertebrate pests.
本发明的技术方案如下:Technical scheme of the present invention is as follows:
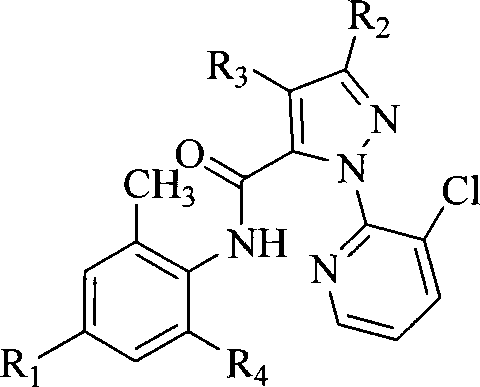
本发明提供一种邻氨基苯甲酸类化合物,如通式I所示:The present invention provides a kind of anthranilic acid compound, as shown in general formula I:
式中:In the formula:
R1选自氢、卤素、氰基、硝基、甲硫基、甲磺酰基、甲氧基羰基、甲胺基羰基、未取代的或被以下基团取代的氨基:甲基、二甲基、乙酰基或甲磺酰基; R is selected from hydrogen, halogen, cyano, nitro, methylthio, methylsulfonyl, methoxycarbonyl, methylaminocarbonyl, Amino group which is unsubstituted or substituted by: methyl, dimethyl, acetyl or methylsulfonyl;
R2选自卤素;R 2 is selected from halogen;
R3选自氢、卤素、氰基、硝基或氨基; R is selected from hydrogen, halogen, cyano, nitro or amino;
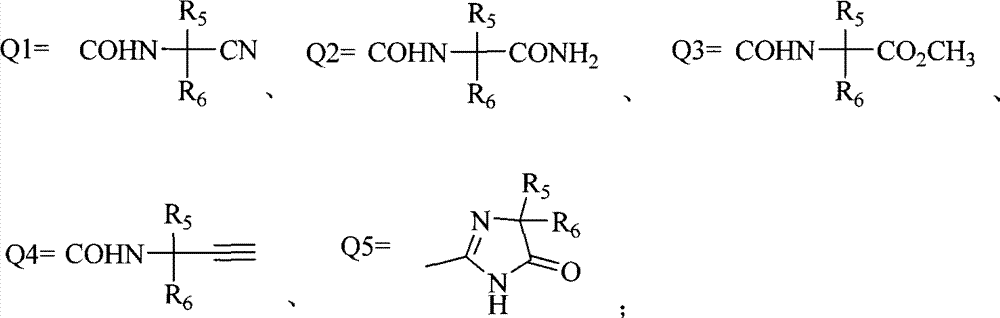
R4选自氰基、-CONHCH2CH2Cl、-CO2R、-CO2CH2CH2N(R)2、 -CONHCH(OR)2、-CONHCH(CH3)CH2CO2Et、-CONHCH(CH3)CH2CN、-COCH2COR、 或者Q1-Q5基团之一;R4 is selected from cyano, -CONHCH 2 CH 2 Cl, -CO 2 R, -CO 2 CH 2 CH 2 N(R) 2 , -CONHCH(OR) 2 , -CONHCH(CH 3 )CH 2 CO 2 Et, -CONHCH(CH 3 )CH 2 CN, -COCH 2 COR, Or one of the Q1-Q5 groups;
其中:in:
R选自氢、C1-C6烷基、卤代C1-C6烷基或苯基;R is selected from hydrogen, C 1 -C 6 alkyl, halogenated C 1 -C 6 alkyl or phenyl;
R5和R6可相同或不同,分别选自氢、卤素、氰基、C1-C6烷基、C2-C6烯基、C2-C6炔基、卤代C1-C6烷基、苯基、苄基、噻吩基、呋喃基或吡啶基。R 5 and R 6 may be the same or different, and are selected from hydrogen, halogen, cyano, C 1 -C 6 alkyl, C 2 -C 6 alkenyl, C 2 -C 6 alkynyl, halogenated C 1 -C 6 Alkyl, phenyl, benzyl, thienyl, furyl or pyridyl.
本发明中较为优选的化合物为:通式I中More preferred compounds in the present invention are: in general formula I
R1选自氢、Cl、Br、I、氰基、硝基、甲硫基、甲磺酰基、甲氧基羰基、甲胺基羰基、未取代的或被以下基团取代的氨基:甲基、二甲基、乙酰基或甲磺酰基; R1 is selected from hydrogen, Cl, Br, I, cyano, nitro, methylthio, methylsulfonyl, methoxycarbonyl, methylaminocarbonyl, amino unsubstituted or substituted by: methyl , dimethyl, acetyl or methylsulfonyl;
R2选自Cl或Br;R 2 is selected from Cl or Br;
R3选自氢、Cl、Br、氰基、硝基或氨基; R is selected from hydrogen, Cl, Br, cyano, nitro or amino;
R4选自氰基、-CONHCH2CH2Cl、-CO2CH3、 -CONHCH(OCH3)2、-COCH2COCH3、-CONHCH(CH3)CH2CO2Et、-CONHCH(CH3)CH2CN、-COCH2CN、 或者Q1-Q5基团之一;R4 is selected from cyano, -CONHCH 2 CH 2 Cl, -CO 2 CH 3 , -CONHCH(OCH 3 ) 2 , -COCH 2 COCH 3 , -CONHCH(CH 3 )CH 2 CO 2 Et, -CONHCH(CH 3 )CH 2 CN, -COCH 2 CN, Or one of the Q1-Q5 groups;
其中:in:
R5和R6可相同或不同,分别选自氢、卤素、氰基、C1-C6烷基、C2-C6烯基、C2-C6炔基、卤代C1-C6烷基、苯基、苄基、噻吩基、呋喃基或吡啶基。R 5 and R 6 may be the same or different, and are selected from hydrogen, halogen, cyano, C 1 -C 6 alkyl, C 2 -C 6 alkenyl, C 2 -C 6 alkynyl, halogenated C 1 -C 6 Alkyl, phenyl, benzyl, thienyl, furyl or pyridyl.
本发明进一步优选的化合物为:通式I中Further preferred compounds of the present invention are: in general formula I
R1选自氢、Cl或氰基; R is selected from hydrogen, Cl or cyano;
R2选自Cl或Br;R 2 is selected from Cl or Br;
R3选自氢、Cl、硝基或氨基; R is selected from hydrogen, Cl, nitro or amino;
R4选自氰基、-CONHCH2CH2Cl、-CO2CH3、 -COCH2COCH3、-CONHCH(CH3)CH2CO2Et、-CONHCH(CH3)CH2CN、-COCH2CN、 或者Q1-Q5基团之一;R4 is selected from cyano, -CONHCH 2 CH 2 Cl, -CO 2 CH 3 , -COCH 2 COCH 3 , -CONHCH(CH 3 )CH 2 CO 2 Et, -CONHCH(CH 3 )CH 2 CN, -COCH 2 CN, Or one of the Q1-Q5 groups;
其中:in:
R5和R6可相同或不同,分别选自氢、C1-C6烷基、苯基或苄基。R 5 and R 6 may be the same or different, and are respectively selected from hydrogen, C 1 -C 6 alkyl, phenyl or benzyl.
本发明更进一步优选的化合物为:通式I中The further preferred compound of the present invention is: in general formula I
R1选自氢、Cl或氰基; R is selected from hydrogen, Cl or cyano;
R2选自Cl或Br;R 2 is selected from Cl or Br;
R3选自氢、Cl、硝基或氨基; R is selected from hydrogen, Cl, nitro or amino;
R4选自氰基、-CONHCH2CH2Cl、-CO2CH3、-CONHCH(OCH3)2或Q1;R4 is selected from cyano, -CONHCH 2 CH 2 Cl, -CO 2 CH 3 , - CONHCH (OCH 3 ) 2 or Q1;
其中:R5和R6可相同或不同,分别选自C1-C3烷基。Wherein: R 5 and R 6 may be the same or different, and are respectively selected from C 1 -C 3 alkyl groups.
上面给出的通式I化合物的定义中,汇集所用术语一般定义如下:In the definitions of the compounds of general formula I given above, the collectively used terms are generally defined as follows:
未取代表示所有取代基都为氢。Unsubstituted means that all substituents are hydrogen.
取代的胺基中取代基的数目可为1~2。The number of substituents in the substituted amino group may be 1-2.
卤:指氟、氯、溴或碘。Halogen: refers to fluorine, chlorine, bromine or iodine.
烷基:直链或支链烷基,例如甲基、乙基、丙基、异丙基或叔丁基。Alkyl: straight-chain or branched-chain alkyl such as methyl, ethyl, propyl, isopropyl or tert-butyl.
卤代烷基:直链或支链烷基,在这些烷基上的氢原子可部分或全部被卤原子所取代,例如,卤代烷基诸如氯甲基、二氯甲基、三氯甲基、氟甲基、二氟甲基或三氟甲基。Haloalkyl: Straight chain or branched chain alkyl in which hydrogen atoms may be partially or completely replaced by halogen atoms, for example, haloalkyl such as chloromethyl, dichloromethyl, trichloromethyl, fluoromethyl group, difluoromethyl or trifluoromethyl.
烯基:直链或支链并可在任何位置上存在有双键,例如乙烯基或烯丙基。Alkenyl: Straight or branched chain with double bonds at any position, such as vinyl or allyl.
炔基:直链或支链并可在任何位置上存在有三键,例如乙炔基或炔丙基。Alkynyl: Straight or branched chain with a triple bond at any position, eg ethynyl or propargyl.
本发明的化合物可以作为一种或多种立体异构体存在。本领域技术人员会理解,当一种立体异构体相对于其它立体异构体而言浓度大时,或者当其与其它立体异构体分离时,可能更有活性和/或可能表现出有利的作用。另外,本领域技术人员知道如何分离、富集和/或选择性地制备所述立体异构体。因此,本发明包括选自通式I、其N-氧化物或其盐的化合物。本发明的化合物可以作为立体异构体的混合物、单个立体异构体或者作为旋光形式存在。The compounds of the invention may exist as one or more stereoisomers. Those skilled in the art will appreciate that one stereoisomer may be more active and/or may appear advantageous when present in greater concentrations relative to the other stereoisomers, or when isolated from the other stereoisomers. role. In addition, those skilled in the art know how to separate, enrich and/or selectively prepare said stereoisomers. Accordingly, the present invention includes compounds selected from the group consisting of general formula I, N-oxides or salts thereof. The compounds of the present invention may exist as mixtures of stereoisomers, individual stereoisomers or as optically active forms.
本发明的化合物的盐包括与无机或有机酸,如氢溴酸、盐酸、硝酸、磷酸、硫酸、乙酸、丁酸、富马酸、乳酸、马来酸、丙二酸、草酸、丙酸、水杨酸、酒石酸、4-甲苯磺酸或戊酸加成盐。在本发明的组合物和方法中,本发明的化合物的盐优选适于本文描述的农学和/或非农学用途。Salts of the compounds of the present invention include those with inorganic or organic acids such as hydrobromic acid, hydrochloric acid, nitric acid, phosphoric acid, sulfuric acid, acetic acid, butyric acid, fumaric acid, lactic acid, maleic acid, malonic acid, oxalic acid, propionic acid, Salicylic acid, tartaric acid, 4-toluenesulfonic acid or valeric acid addition salts. In the compositions and methods of the invention, the salts of the compounds of the invention are preferably suitable for the agronomic and/or non-agronomic uses described herein.
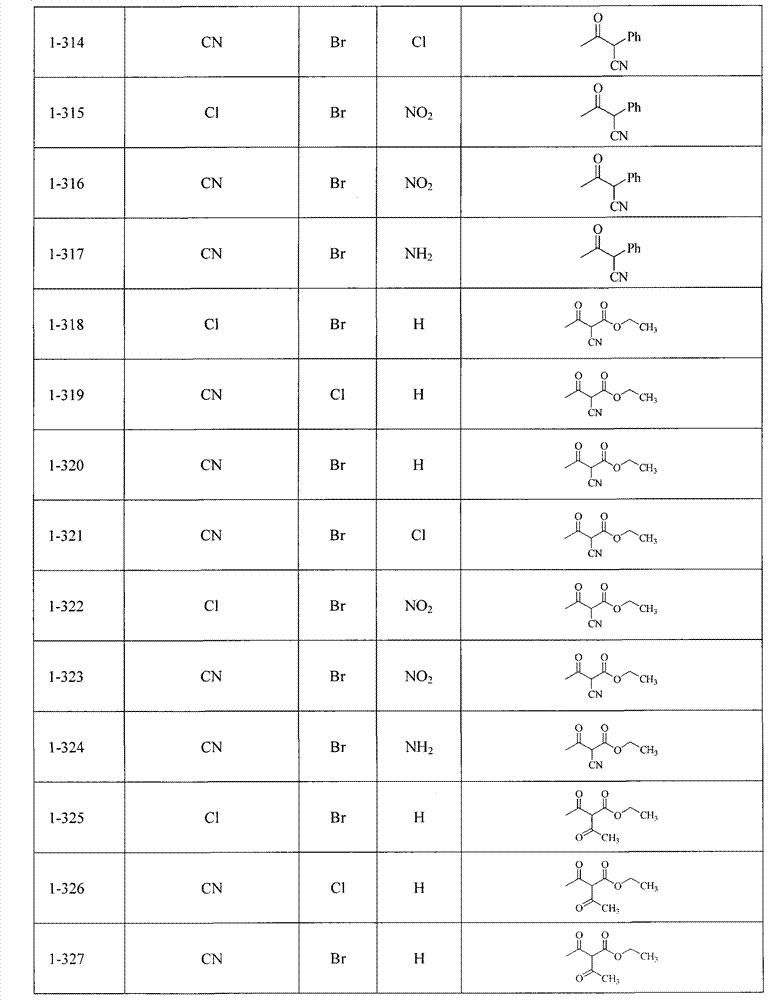
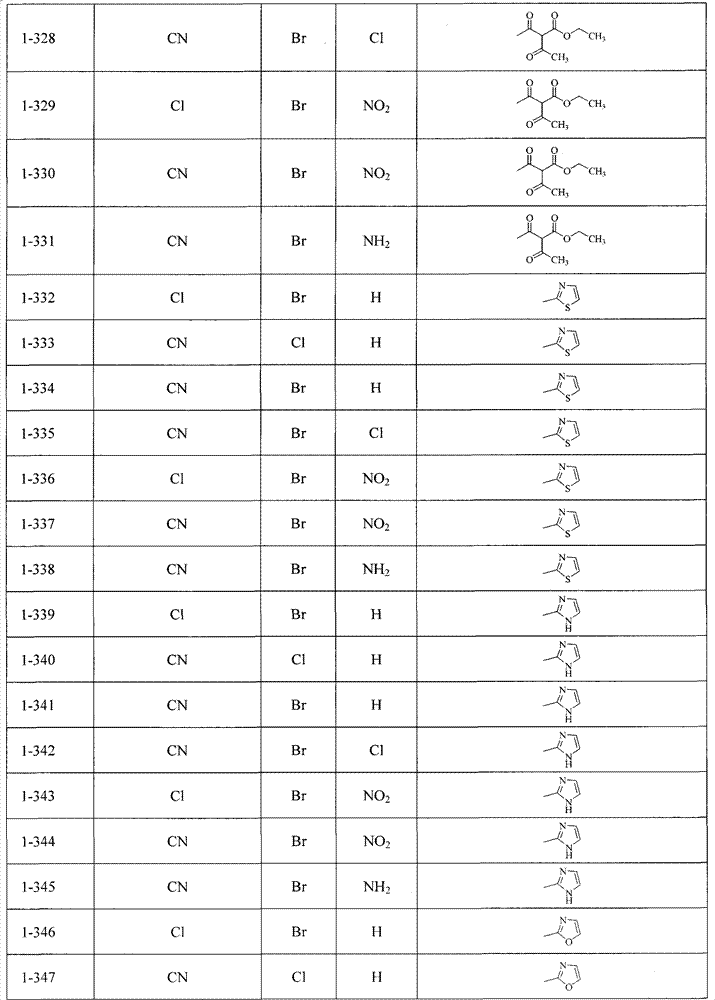
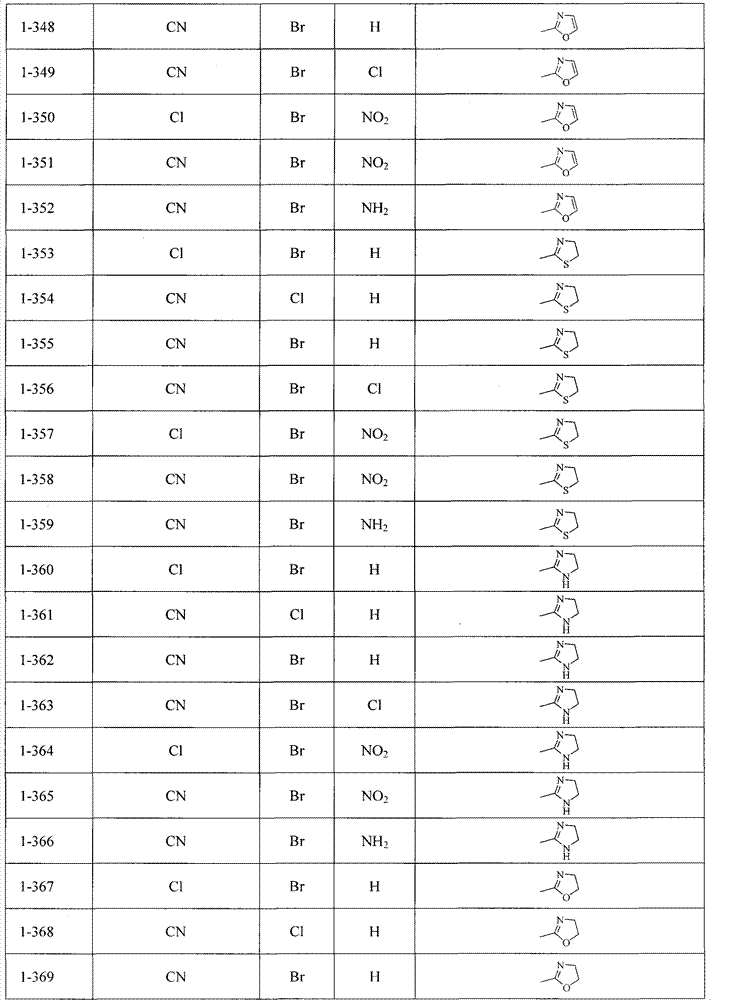
可以用下面表1-表2列出的化合物来说明本发明,但并不限定本发明。The compounds listed in Table 1-Table 2 below can be used to illustrate the present invention, but not to limit the present invention.
当R4不为Q1-Q5时, When R4 is not Q1-Q5,
表1Table 1
当R4为Q1-Q5之一时, When R 4 is one of Q 1 -Q 5 ,
表2Table 2
本发明的通式I化合物可以按照以下方法制备:The compound of general formula I of the present invention can be prepared according to the following methods:
方法一:method one:
通式I化合物可由通式II所示的恶嗪酮类化合物与取代的胺、醇反应制得:The compound of general formula I can be prepared by reacting oxazinone compounds shown in general formula II with substituted amines and alcohols:
方法二:Method Two:
通式I化合物可由通式IV所示的茚红酸酐类化合物与取代的胺、醇反应制得的苯胺中间体III,再与吡唑酰氯缩合制得,其中吡唑酰氯可由相应的羧酸(如通式VI所示)通过常规方法制得,如与氯化亚砜反应:The compound of general formula I can be prepared by the aniline intermediate III prepared by reacting indene acid anhydride compounds shown in general formula IV with substituted amines and alcohols, and then condensed with pyrazole acid chloride, wherein pyrazole acid chloride can be prepared by corresponding carboxylic acid ( As shown in general formula VI) is prepared by conventional methods, such as reacting with thionyl chloride:
通式IVIV中间体可由通式V所示的取代的邻氨基苯甲酸中间体与固体光气反应制得:The intermediate of general formula IVIV can be prepared by reacting the substituted anthranilic acid intermediate shown in general formula V with solid phosgene:
通式中各基团的定义同前。The definition of each group in the general formula is the same as before.
反应在溶剂中进行,适宜的选自如乙腈、四氢呋喃、乙醚、二氯甲烷、氯仿、乙酸乙酯、二氧六环、甲苯等。The reaction is carried out in a solvent, suitably selected from acetonitrile, tetrahydrofuran, diethyl ether, dichloromethane, chloroform, ethyl acetate, dioxane, toluene and the like.
反应温度在室温至溶剂沸点温度之间,通常为20~100℃。The reaction temperature is between room temperature and the boiling point of the solvent, usually 20-100°C.
反应时间为30分钟至20小时,通常1~10小时。The reaction time is 30 minutes to 20 hours, usually 1 to 10 hours.
上述制备方法中所涉及的原料中间体及来源如下:The raw material intermediate involved in the above-mentioned preparation method and source are as follows:
通式II为恶嗪酮类化合物,相关综述见Biorganic and Medicinal Chemistry 2000,8,2095-2103.和J.Heterocyclic Chemistry 1999,36,563-588,其制备方法参照WO03015519、WO2005118552方法。The general formula II is an oxazinone compound. For related reviews, see Biorganic and Medicinal Chemistry 2000, 8, 2095-2103. and J. Heterocyclic Chemistry 1999, 36, 563-588. The preparation method refers to the methods of WO03015519 and WO2005118552.
通式V所示的邻氨基苯甲酸类化合物可由芳胺经两步制得,参照如下文献:OrganicSyntheses,Coll.Vo1.10,p.23(2004);Vol.79,p.196(2002);Adv.Heterocycl.Chem.1975,18,1-58;Journal of the Brazilian Chemical Society 2001,12(3),273-324;Angew.Chem.Int.Ed.Engl.1980,19,222-223。The anthranilic acid compound shown in general formula V can be prepared by aromatic amine through two steps, refer to following document: Organic Syntheses, Coll.Vo1.10, p.23 (2004); Vol.79, p.196 (2002) ; Adv.Heterocycl.Chem.1975, 18, 1-58; Journal of the Brazilian Chemical Society 2001, 12(3), 273-324;
通式VI所示的羧酸化合物的制备方法参照WO2006062978、WO03015519。The preparation method of the carboxylic acid compound represented by the general formula VI refers to WO2006062978 and WO03015519.
取代的胺、醇部分有市售,也可按照常规方法自制。Substituted amines and alcohols are commercially available, and can also be made by conventional methods.
本发明的通式I化合物对农业、民用和动物技术领域中有害昆虫的成虫、幼虫和卵都显示出高杀虫活性。因此,本发明还包括通式I化合物在农业或其他领域中作为杀虫剂的应用。包括使该无脊椎动物害虫或该害虫所处的农学或非农学环境与生物学有效量的通式I化合物接触。The compound of general formula I of the present invention shows high insecticidal activity against adults, larvae and eggs of harmful insects in agricultural, civil and animal technology fields. Therefore, the present invention also includes the use of the compound of general formula I as a pesticide in agriculture or other fields. It includes contacting the invertebrate pest or the agronomic or non-agronomic environment in which the pest lives with a biologically effective amount of a compound of general formula I.
尤其是,通式I化合物对下列科和目的重要品种有活性:鳞翅目(贪夜蛾属、实夜蛾属、禾草螟属、苹果皮小卷蛾等)。例如对欧洲玉米螟、甘蔗螟、苹果小卷蛾、苹果食心虫、舞毒蛾等十分有效,特别是对小菜蛾活性更好,在很低的剂量下就可以获得很好的效果。In particular, the compounds of the general formula I are active against important species of the following families and orders: Lepidoptera (Spodoptera, Helicoptera, Grass moth, Apple-skin tortilla, etc.). For example, it is very effective against European corn borer, sugarcane borer, apple tortrix, apple borer, gypsy moth, etc., especially against diamondback moth, and can obtain good results at a very low dose.
同时,通式I化合物对许多有益的昆虫和螨虫、哺乳动物、鱼、鸟具有低毒性,而且没有植物毒性。At the same time, the compound of general formula I has low toxicity to many beneficial insects and mites, mammals, fish, birds, and has no phytotoxicity.
由于其积极的特性,上述化合物可有利地用于保护农业和园艺业重要的作物、家畜和种畜,以及人类常去的环境免于有害昆虫和真菌的伤害。Due to their positive properties, the above-mentioned compounds can be advantageously used for protecting agriculturally and horticulturally important crops, livestock and breeding stock, and environments frequented by humans from harmful insects and fungi.
为获得理想效果,化合物的用量因各种因素而改变,例如所用化合物、预保护的作物、有害生物的类型、感染程度、气候条件、施药方法、采用的剂型。The amount of compound used to achieve the desired effect will vary depending on factors such as compound used, crop to be protected, type of pest, degree of infestation, climatic conditions, method of application, formulation employed.
每公顷10克-5公斤的化合物剂量能提供充分的防治。Doses of 10 g-5 kg of compound per hectare provide adequate control.
本发明的另一目的还涉及通过施用通式I化合物,防治农业和园艺业重要的作物和/或家畜和种畜和/或人类常去的环境中的昆虫和/或植物致病性真菌的方法。尤其是,化合物的用量在每公顷10克-5公斤内变化。Another object of the present invention also relates to a method for controlling insects and/or phytopathogenic fungi in agriculturally and horticulturally important crops and/or livestock and breeding stock and/or in environments frequented by humans by applying compounds of general formula I . In particular, the amount of compound used varies from 10 g to 5 kg per hectare.
为了实际应用于农业,使用含一种或多种通式I化合物的组合物通常是有益的。For practical use in agriculture it is often advantageous to use compositions comprising one or more compounds of general formula I.
本发明还涉及用于防治无脊椎动物害虫的组合物,其包含生物学有效量的通式I化合物和至少一种选自表面活性剂、固体稀释剂和液体稀释剂,以及可以含有任选的有效量的至少一种附加生物学活性化合物。The present invention also relates to a composition for controlling invertebrate pests, which comprises a biologically effective amount of a compound of general formula I and at least one selected from surfactants, solid diluents and liquid diluents, and may optionally contain An effective amount of at least one additional biologically active compound.
本发明还涉及一种组合物在农业或其他领域中作为杀虫剂的应用,包括使该无脊椎动物害虫或该害虫所处的农学或非农学环境与生物学有效量的组合物接触,所述组合物包含生物学有效量的通式I化合物和至少一种选自表面活性剂、固体稀释剂或液体稀释剂的附加组分,所述组合物还可以任选进一步包含有效量的至少一种附加生物学活性化合物。The present invention also relates to the use of a composition as a pesticide in agriculture or other fields, comprising contacting the invertebrate pest or the agronomic or non-agronomic environment in which the pest is in contact with a biologically effective amount of the composition, wherein Said composition comprises the compound of general formula I of biologically effective amount and at least one additional component selected from surfactant, solid diluent or liquid diluent, and said composition can also optionally further comprise effective amount of at least one additional biologically active compounds.
组合物的使用形式可以是干粉、可湿性粉剂、乳油、微乳剂、糊剂、颗粒剂、溶液、悬浮剂等:组合物类型的选择取决于具体的应用。The composition can be used in the form of dry powder, wettable powder, emulsifiable concentrate, microemulsion, paste, granule, solution, suspension, etc.: the choice of composition type depends on the specific application.
组合物是以已知方法制备的,例如任选在表面活性剂的存在下,通过用溶剂介质和/或固体稀释剂稀释或溶解活性物质。The compositions are prepared in known manner, for example by diluting or dissolving the active substance with a solvent medium and/or a solid diluent, optionally in the presence of surfactants.
可用的固体稀释剂或载体是例如:二氧化硅、高岭土、膨润土、滑石、硅藻土、白云石、碳酸钙、氧化镁、白垩、粘土、合成硅酸盐、硅镁土、海泡石等。Usable solid diluents or carriers are, for example: silica, kaolin, bentonite, talc, diatomaceous earth, dolomite, calcium carbonate, magnesium oxide, chalk, clay, synthetic silicates, attapulgite, sepiolite, etc. .
除水以外,可用的液体稀释剂还包括如芳族有机溶剂(二甲苯或烷基苯的混合物、氯苯等),石蜡(石油馏分),醇类(甲醇、丙醇、丁醇、辛醇、甘油),酯类(乙酸乙酯、乙酸异丁酯等),酮类(环己酮、丙酮、苯乙酮、异佛尔酮、乙基戊基酮等),酰胺类(N,N-二甲基甲酰胺、N-甲基吡咯烷酮等)。In addition to water, usable liquid diluents include aromatic organic solvents (mixtures of xylene or alkylbenzenes, chlorobenzene, etc.), paraffins (petroleum fractions), alcohols (methanol, propanol, butanol, octanol , glycerin), esters (ethyl acetate, isobutyl acetate, etc.), ketones (cyclohexanone, acetone, acetophenone, isophorone, ethyl amyl ketone, etc.), amides (N, N -dimethylformamide, N-methylpyrrolidone, etc.).
可用的表面活性剂是烷基磺酸盐、烷基芳基磺酸盐、聚氧乙烯烷基酚、山梨醇的聚氧乙烯酯、木质素磺酸盐等的钠、钙、三乙基胺或三乙醇胺盐。Surfactants that can be used are sodium, calcium, triethylamine of alkylsulfonates, alkylarylsulfonates, polyoxyethylene alkylphenols, polyoxyethylene esters of sorbitol, lignosulfonates, etc. or triethanolamine salt.
组合物还可含特殊的添加剂用于特定的目的,例如含有粘合剂如阿拉伯胶、聚乙烯醇、聚乙烯吡咯烷酮等。The composition may also contain special additives for specific purposes, such as binders such as gum arabic, polyvinyl alcohol, polyvinylpyrrolidone and the like.
上述组合物中活性成分的浓度可根据活性成分、使用目的、环境条件和采用的制剂类型而在宽范围内改变。通常,活性成分的浓度范围是1-90%,优选5-60%。The concentration of the active ingredient in the above compositions may vary within wide limits depending on the active ingredient, purpose of use, environmental conditions and type of formulation employed. Usually, the concentration range of the active ingredient is 1-90%, preferably 5-60%.
如果需要,可以向组合物中添加能与通式I化合物兼容的其他活性成分,例如其他杀螨剂/杀虫剂、杀真菌剂、植物生长调节剂、抗生素、除草剂、肥料。If desired, other active ingredients compatible with the compounds of general formula I, such as other acaricides/insecticides, fungicides, plant growth regulators, antibiotics, herbicides, fertilizers, can be added to the composition.
几种剂型的配制方法举例如下:The preparation methods of several dosage forms are exemplified as follows:
悬浮剂的配制:常用配方中活性组分含量为5%-35%。以水为介质,将原药、分散剂、助悬剂和抗冻剂等加入砂磨机中,进行研磨,制成悬浮剂。Suspension preparation: the content of active ingredients in common formulations is 5%-35%. Using water as the medium, add the original drug, dispersant, suspending agent and antifreeze into a sand mill for grinding to make a suspension.
水乳剂的配制:将原药、溶剂和乳化剂加在一起,使溶解成均匀油相。将水、抗冻剂等混合一起,成为均一水相。在高速搅拌下,将水相加入到油相或将油相加入到水相,形成分散性良好的水乳剂。本发明的水乳剂活性组分含量一般为5%-15%。为制备浓乳剂,本发明的化合物可溶解于一种或数种混合溶剂,再加入乳化剂来增强化合物在水中的分散效果。Preparation of emulsion in water: Add the original drug, solvent and emulsifier together to dissolve into a uniform oil phase. Mix water, antifreeze, etc. together to form a homogeneous water phase. Under high-speed stirring, add the water phase to the oil phase or add the oil phase to the water phase to form a water emulsion with good dispersibility. The active component content of the aqueous emulsion of the present invention is generally 5%-15%. To prepare a concentrated emulsion, the compound of the present invention can be dissolved in one or several mixed solvents, and then an emulsifier is added to enhance the dispersion effect of the compound in water.
可湿性粉剂的配制:按配方要求,将原药、各种表面活性剂及固体稀释剂等充分混合,经超细粉碎机粉碎后,即得到预定含量(例如10%-60%)的可湿性粉剂产品。为制备适于喷洒用的可湿性粉剂,本发明的化合物可以和研细的固体粉末如粘土、无机硅酸盐、碳酸盐以及润湿剂、粘合剂和/或分散剂组成混合物。Preparation of wettable powder: according to the formula requirements, the original drug, various surfactants and solid diluents are fully mixed, and after being pulverized by a superfine pulverizer, a wettability powder with a predetermined content (such as 10%-60%) can be obtained. Powder products. To prepare wettable powders suitable for spraying, the compounds according to the invention can be mixed with finely divided solid powders such as clays, inorganic silicates, carbonates and wetting, binding and/or dispersing agents.
水分散性粒剂的配制:将原药和粉状固体稀释剂、润湿展着剂及粘合剂等进行混合粉碎,再加水捏合后,加入装有一定规格筛网的造粒机中进行造粒,然后再经干燥、筛分(按筛网范围)。也可将原药、分散剂、崩解剂和润湿剂及固体稀释剂加入砂磨机中,以水为介质研磨,制成悬浮剂,然后进行喷雾干燥造粒,通常配制含量为20%-30%颗粒状产品。Preparation of water-dispersible granules: mix and pulverize the original drug, powdery solid diluent, wetting and spreading agent, and binder, add water and knead, then add to a granulator equipped with a certain size screen. Granulate, then dry and sieve (according to the size of the sieve). The original drug, dispersant, disintegrating agent, wetting agent and solid diluent can also be added to a sand mill, ground with water as a medium to make a suspension, and then spray-dried and granulated, usually with a preparation content of 20% -30% granular product.
具体实施方式Detailed ways
以下具体实施例用来进一步说明本发明,但本发明绝非限于这些例子。The following specific examples are used to further illustrate the present invention, but the present invention is by no means limited to these examples.
合成实施例Synthetic example
实例1:化合物1-152的制备Example 1: Preparation of Compound 1-152
取0.5克II-I于50毫升反应瓶中,加入25毫升四氢呋喃,搅拌下加入1克HOCH2CH2N(CH3)2,升温回流反应1小时。TLC监测反应完毕后,减压脱溶后,向反应瓶中倒入50毫升饱和食盐水中,用60毫升乙酸乙酯分三次进行萃取,干燥,脱溶,柱层析得产品0.31克,即化合物1-152,收率51.8%。熔点80-82℃。Take 0.5 g of II-I in a 50 ml reaction flask, add 25 ml of tetrahydrofuran, add 1 g of HOCH 2 CH 2 N(CH 3 ) 2 under stirring, and raise the temperature to reflux for 1 hour. After TLC monitors the completion of the reaction, after desolvation under reduced pressure, pour 50 milliliters of saturated saline into the reaction flask, extract three times with 60 milliliters of ethyl acetate, dry, and desolventize, and column chromatography gives 0.31 grams of the product, namely the compound 1-152, yield 51.8%. The melting point is 80-82°C.
核磁数据(1HNMR,300MHz,内标TMS,溶剂CDCl3)如下:NMR data ( 1 HNMR, 300MHz, internal standard TMS, solvent CDCl 3 ) are as follows:
化合物1-152:δppm 2.18(3H,s),2.41(6H,s),2.79(2H,t),4.45(2H,t),7.19(1H,s),7.39(2H,m),7.80-7.88(2H,m),8.45(1H,q),10.14(1H,s)。Compound 1-152: δppm 2.18 (3H, s), 2.41 (6H, s), 2.79 (2H, t), 4.45 (2H, t), 7.19 (1H, s), 7.39 (2H, m), 7.80- 7.88 (2H, m), 8.45 (1H, q), 10.14 (1H, s).
实例2:化合物2-2的制备Example 2: Preparation of compound 2-2
取1克IV-1于100毫升反应瓶,加入50毫升二氧六环及中间体H2NC(CH3)2CN,回流反应3小时。TLC监测反应完毕后,减压脱溶后,向反应瓶中倒入50毫升饱和食盐水,用90毫升乙酸乙酯分三次进行萃取,干燥,脱溶,柱层析得III-1中间体0.63克。Take 1 g of IV-1 in a 100 ml reaction bottle, add 50 ml of dioxane and intermediate H 2 NC(CH 3 ) 2 CN, and react under reflux for 3 hours. After the completion of the reaction as monitored by TLC, after desolvation under reduced pressure, 50 ml of saturated saline was poured into the reaction flask, extracted three times with 90 ml of ethyl acetate, dried, and desolvated, and column chromatography gave III-1 intermediate 0.63 gram.
将制得的中间体III-1溶于60毫升四氢呋喃,加入1毫升三乙胺,搅拌下滴加1.02克酰氯,室温反应4小时,TLC监测反应完毕后,处理,柱层析得产品0.45克,即化合物2-2,收率31%。熔点170-172℃。The prepared intermediate III-1 was dissolved in 60 ml of tetrahydrofuran, 1 ml of triethylamine was added, 1.02 g of acid chloride was added dropwise under stirring, and the reaction was carried out at room temperature for 4 hours. After the reaction was monitored by TLC, 0.45 g of the product was obtained by column chromatography , namely compound 2-2, the yield is 31%. The melting point is 170-172°C.
核磁数据(1HNMR,300MHz,内标TMS,溶剂CDCl3)如下:NMR data ( 1 HNMR, 300MHz, internal standard TMS, solvent CDCl 3 ) are as follows:
化合物2-2:δppm 1.71(6H,s),2.10(3H,s),3.95(1H,s),6.75(1H,t),6.89(1H,s),7.16-7.24(2H,m),7.66-7.73(2H,m),8.38(1H,q)。Compound 2-2: δppm 1.71(6H, s), 2.10(3H, s), 3.95(1H, s), 6.75(1H, t), 6.89(1H, s), 7.16-7.24(2H, m), 7.66-7.73 (2H, m), 8.38 (1H, q).
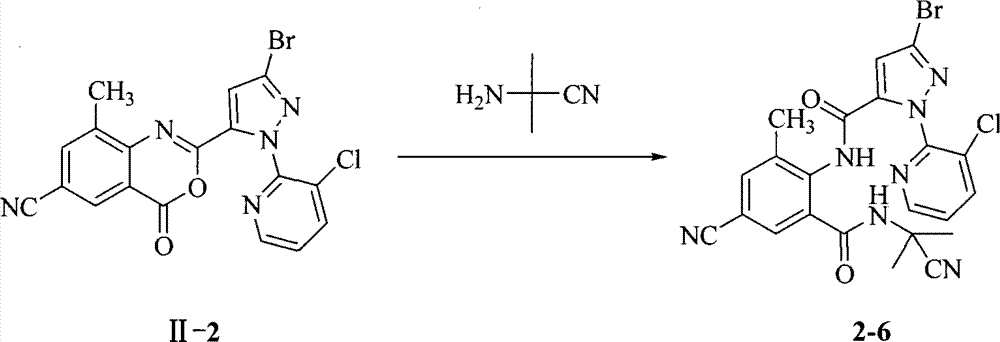
实例3:化合物2-6的制备Example 3: Preparation of Compound 2-6
取0.5克II-2于50毫升反应瓶中,加入25毫升乙腈,搅拌下加入0.8克H2NC(CH3)2CN,升温回流反应3小时。TLC监测反应完毕后,减压脱溶后,向反应瓶中倒入50毫升饱和食盐水,用90毫升乙酸乙酯分三次进行萃取,干燥,脱溶,柱层析得产品0.27克,即化合物2-6,收率45.3%。熔点140-142℃。Take 0.5 g of II-2 in a 50 ml reaction flask, add 25 ml of acetonitrile, add 0.8 g of H 2 NC(CH 3 ) 2 CN while stirring, and heat up to reflux for 3 hours. After TLC monitors the completion of the reaction, after desolvation under reduced pressure, pour 50 milliliters of saturated saline into the reaction flask, extract three times with 90 milliliters of ethyl acetate, dry, and desolventize, and obtain 0.27 grams of product by column chromatography, namely compound 2-6, yield 45.3%. The melting point is 140-142°C.
核磁数据(1HNMR,300MHz,内标TMS,溶剂CDCl3)如下:NMR data ( 1 HNMR, 300MHz, internal standard TMS, solvent CDCl 3 ) are as follows:
化合物2-6:δppm 1.95(6H,s),2.65(3H,s),6.85(1H,s),7.28(1H,q),7.87(1H,s),7.92(1H,d),8.30(1H,q),8.48(1H,s)。Compound 2-6: δppm 1.95 (6H, s), 2.65 (3H, s), 6.85 (1H, s), 7.28 (1H, q), 7.87 (1H, s), 7.92 (1H, d), 8.30 ( 1H,q), 8.48(1H,s).
通式I的其他化合物可以用本发明提供的制备方法制得。Other compounds of general formula I can be prepared by the preparation methods provided by the present invention.
部分通式I化合物的熔点和核磁数据(1HNMR,300MHz,内标TMS,溶剂CDCl3)如下:The melting points and NMR data ( 1 HNMR, 300 MHz, internal standard TMS, solvent CDCl 3 ) of some compounds of general formula I are as follows:
化合物1-102:熔点118-120℃。δppm 2.23(3H,s),3.92(3H,s),7.06(1H,s),7.19(1H,m),7.38(2H,m),7.84(2H,m),8.47(1H,q),10.19(1H,s)。Compound 1-102: melting point 118-120°C. δppm 2.23(3H, s), 3.92(3H, s), 7.06(1H, s), 7.19(1H, m), 7.38(2H, m), 7.84(2H, m), 8.47(1H, q), 10.19 (1H, s).
化合物1-104:熔点170-172℃。δppm 2.24(3H,s),3.74(3H,s),7.33(1H,s),7.51-7.59(3H,m),8.07(1H,q),8.47(1H,q),10.25(1H,s)。Compound 1-104: melting point 170-172°C. δppm 2.24(3H, s), 3.74(3H, s), 7.33(1H, s), 7.51-7.59(3H, m), 8.07(1H, q), 8.47(1H, q), 10.25(1H, s ).
化合物1-205:熔点60-62℃。δppm 1.12(6H,t),2.21(3H,s),2.68(4H,q),2.88(2H,t),4.41(2H,t),7.13(1H,s),7.39(2H,m),7.80(1H,d),7.86(1H,q),8.46(1H,q),10.17(1H,s)。Compound 1-205: melting point 60-62°C. δppm 1.12(6H, t), 2.21(3H, s), 2.68(4H, q), 2.88(2H, t), 4.41(2H, t), 7.13(1H, s), 7.39(2H, m), 7.80 (1H, d), 7.86 (1H, q), 8.46 (1H, q), 10.17 (1H, s).
化合物1-218:熔点206-207℃。δppm 2.17(3H,s),3.67(2H,m),3.72(2H,m),6.55(1H,s),7.07(1H,s),7.34(1H,s),7.36(1H,s),7.37(1H,q),7.84(1H,d),8.45(1H,d),9.89(1H,s)。Compound 1-218: melting point 206-207°C. δppm 2.17(3H, s), 3.67(2H, m), 3.72(2H, m), 6.55(1H, s), 7.07(1H, s), 7.34(1H, s), 7.36(1H, s), 7.37 (1H, q), 7.84 (1H, d), 8.45 (1H, d), 9.89 (1H, s).
化合物1-220:熔点240-242℃。δppm 2.26(3H,s),3.73(2H,m),3.79(2H,m),6.58(1H,s),7.02(1H,s),7.41(1H,q),7.36(1H,s),7.64(2H,m),7.86(1H,d),8.43(1H,d),10.41(1H,s)。Compound 1-220: melting point 240-242°C. δppm 2.26(3H, s), 3.73(2H, m), 3.79(2H, m), 6.58(1H, s), 7.02(1H, s), 7.41(1H, q), 7.36(1H, s), 7.64 (2H, m), 7.86 (1H, d), 8.43 (1H, d), 10.41 (1H, s).
化合物1-264:熔点208-209℃。δppm 2.19(3H,s),3.41(6H,s),3.55(2H,m),4.43(1H,t),6.28(1H,t),7.07(1H,s),7.26(2H,m),7.38(1H,q),7.85(1H,q),8.46(1H,q),10.08(1H,s)。Compound 1-264: melting point 208-209°C. δppm 2.19(3H, s), 3.41(6H, s), 3.55(2H, m), 4.43(1H, t), 6.28(1H, t), 7.07(1H, s), 7.26(2H, m), 7.38(1H,q), 7.85(1H,q), 8.46(1H,q), 10.08(1H,s).
化合物1-266:熔点182-183℃。δppm 2.22(3H,t),3.42(6H,s),3.58(2H,m),4.43(1H,t),6.33(1H,t),7.07(1H,s),7.22(2H,m),7.36(1H,q),7.80(1H,q),8.46(1H,q),10.20(1H,s)。Compound 1-266: melting point 182-183°C. δppm 2.22(3H, t), 3.42(6H, s), 3.58(2H, m), 4.43(1H, t), 6.33(1H, t), 7.07(1H, s), 7.22(2H, m), 7.36(1H,q), 7.80(1H,q), 8.46(1H,q), 10.20(1H,s).
化合物2-4:熔点190-192℃。δppm 1.70(6H,s),2.10(3H,s),3.95(1H,s),6.87(1H,s),7.21-7.25(2H,m),7.70-7.75(2H,m),8.39(1H,d)。Compound 2-4: melting point 190-192°C. δppm 1.70(6H, s), 2.10(3H, s), 3.95(1H, s), 6.87(1H, s), 7.21-7.25(2H, m), 7.70-7.75(2H, m), 8.39(1H , d).
制剂实施例(各组分加入量均为重量百分含量,活性化合物折百后计量加入)Formulation example (the addition amount of each component is the weight percent content, and the active compound is metered in after 100 percent)
实施例4:30%可湿性粉剂Example 4: 30% WP
化合物1-152 30%Compound 1-152 30%
十二烷基硫酸钠 2%Sodium Lauryl Sulfate 2%
木质素磺酸钠 3%Sodium lignosulfonate 3%
萘磺酸甲醛缩合物 5%Naphthalenesulfonic acid formaldehyde condensate 5%
轻质碳酸钙 补足至100%Light Calcium Carbonate Top up to 100%
将化合物1-152、各种表面活性剂及固体稀释剂等充分混合,经超细粉碎机粉碎后,即得到30%的可湿性粉剂产品。Compound 1-152, various surfactants and solid diluents are fully mixed, and pulverized by an ultrafine pulverizer to obtain a 30% wettable powder product.
实施例5:20%悬浮剂Embodiment 5: 20% suspending agent
化合物1-218 20%Compound 1-218 20%
甲基萘磺酸甲醛缩合物 3%Methylnaphthalenesulfonic acid formaldehyde condensate 3%
农乳0201B 2%Agricultural Milk 0201B 2%
农乳700# 1%Farm milk 700# 1%
黄原胶 0.2%Xanthan Gum 0.2%
乙二醇 5%Ethylene Glycol 5%
水 补足至100%Water up to 100%
以水为介质,将化合物1-218、分散剂、助悬剂和抗冻剂等加入砂磨机中,进行研磨,制成悬浮剂。Using water as the medium, add compound 1-218, dispersant, suspending agent and antifreeze into a sand mill and grind to prepare a suspension.
实施例6:60%水分散性粒剂Example 6: 60% water dispersible granules
化合物1-220 60%Compound 1-220 60%
萘磺酸钠甲醛缩合物 12%Sodium naphthalenesulfonate formaldehyde condensate 12%
N-甲基-油酰基-牛磺酸钠 8%Sodium N-methyl-oleoyl-taurate 8%
聚乙烯吡咯烷酮 2%Polyvinylpyrrolidone 2%
羧甲基纤维素 2%Carboxymethylcellulose 2%
高岭土 补足至100%Kaolin topped up to 100%
将化合物1-220和粉状固体稀释剂、润湿展着剂及粘合剂等进行混合粉碎,再加水捏合后,加入10-100目筛网的造粒机中进行造粒,然后再经干燥、筛分(按筛网范围)。Compound 1-220 is mixed with powdery solid diluent, wetting and spreading agent, binder, etc., mixed and pulverized, added water and kneaded, then added to a granulator with a 10-100 mesh screen for granulation, and then passed through Dry and sieve (according to the sieve range).
实施例7:10%水乳剂Example 7: 10% water emulsion
化合物1-264 10%Compound 1-264 10%
聚氧乙烯(n20)苯乙基Polyoxyethylene (n 20 ) phenylethyl
酚基醚油酸酯 8%Phenyl ether oleate 8%
十二烷基苯磺酸钙 16%Calcium dodecylbenzenesulfonate 16%
胡椒基丁醚 15%Piperonyl butoxide 15%
环己酮 10%Cyclohexanone 10%
山梨醇 5%Sorbitol 5%
水 补足至100%Water up to 100%
将化合物1-264、溶剂和乳化剂加在一起,使溶解成均匀油相。将水、抗冻剂等混合一起,成为均一水相。在高速搅拌下,将水相与油相混合,形成分散性良好的水乳剂。Compound 1-264, solvent and emulsifier were added together and allowed to dissolve into a homogeneous oil phase. Mix water, antifreeze, etc. together to form a homogeneous water phase. Under high-speed stirring, mix the water phase and the oil phase to form a water emulsion with good dispersibility.
实施例8:25%悬浮剂Example 8: 25% suspending agent
化合物2-6 25%Compound 2-6 25%
农乳0201B 2.5%Agricultural Milk 0201B 2.5%
农乳1601 1%Farm Milk 1601 1%
萘磺酸甲醛缩合物 2%Naphthalenesulfonic acid formaldehyde condensate 2%
白碳黑 0.1%White carbon black 0.1%
乙二醇 5%Ethylene Glycol 5%
水 补足至100%Water to make up to 100%
以水为介质,将化合物2-6、分散剂、助悬剂和抗冻剂等加入砂磨机中,进行研磨,制成悬浮剂。Using water as the medium, add compound 2-6, dispersant, suspending agent and antifreeze into a sand mill and grind to prepare a suspension.
实施例9:20%可湿性粉剂Example 9: 20% WP
化合物2-4 20%Compound 2-4 20%
亚甲基萘磺酸钠 6%Sodium Methylene Naphthalene Sulfonate 6%
萘磺酸甲醛缩合物 3%Naphthalenesulfonic acid formaldehyde condensate 3%
十二烷基硫酸钠 2%Sodium Lauryl Sulfate 2%
白碳黑 5%White carbon black 5%
农乳600# 0.5%Farm Milk 600 # 0.5%
轻质碳酸钙 补足至100%Light Calcium Carbonate Top up to 100%
将化合物2-4、各种表面活性剂及固体稀释剂等充分混合,经超细粉碎机粉碎后,即得到20%的可湿性粉剂产品。Compounds 2-4, various surfactants and solid diluents are fully mixed, and pulverized by an ultra-fine pulverizer to obtain a 20% wettable powder product.
生物活性测定实施例Examples of biological activity assays
实例10杀虫杀螨活性测定Example 10 Determination of insecticidal and acaricidal activity
用本发明化合物对几种昆虫和螨类进行了杀虫活性测定试验。测定的方法如下:Insecticidal activity tests were carried out on several insects and acarids using the compounds of the present invention. The method of determination is as follows:
待测化合物用丙酮/甲醇(1∶1)的混合溶剂溶解后,用含有0.1%吐温80的水稀释至所需的浓度。The compound to be tested was dissolved in a mixed solvent of acetone/methanol (1:1), and then diluted to the desired concentration with water containing 0.1% Tween 80.
以小菜蛾、甜菜夜蛾为靶标,采用airbrush喷雾法和浸液法(淡色库蚊幼虫)进行杀虫活性测定,airbrush喷雾处理的压力为10psi(约合0.7kg/cm2),喷液量为0.5毫升。处理后2-3日调查靶标的死亡率。With Plutella xylostella and beet armyworm as targets, the airbrush spray method and liquid immersion method (Culex larvae) were used to measure the insecticidal activity. for 0.5 ml. Mortality of the target was investigated 2-3 days after treatment.
部分测试结果如下:Some test results are as follows:
药液浓度为600ppm时,化合物1-102、1-104、1-152、1-205对小菜蛾和甜菜夜蛾死亡率达100%;When the liquid concentration is 600ppm, the mortality rate of compounds 1-102, 1-104, 1-152, 1-205 to diamondback moth and beet armyworm is 100%;
药液浓度为20ppm时,化合物2-2、2-4、2-6对甜菜夜蛾死亡率达100%;When the concentration of the drug solution is 20ppm, the mortality rate of compounds 2-2, 2-4, and 2-6 to beet armyworm reaches 100%;
药液浓度为1ppm时,化合物1-218、1-220、1-264、1-266对甜菜夜蛾死亡率达100%。When the concentration of the drug solution is 1 ppm, the mortality of the compounds 1-218, 1-220, 1-264 and 1-266 against the beet armyworm reaches 100%.
Claims (5)
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2008100571021A CN101497602B (en) | 2008-01-30 | 2008-01-30 | Anthranilic acid compound and use thereof |
| PCT/CN2008/070832 WO2008134970A1 (en) | 2007-04-30 | 2008-04-28 | Anthranilamide compounds and the use thereof |
| PCT/CN2008/070830 WO2008134969A1 (en) | 2007-04-30 | 2008-04-28 | Benzamide compounds and applications thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2008100571021A CN101497602B (en) | 2008-01-30 | 2008-01-30 | Anthranilic acid compound and use thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN101497602A CN101497602A (en) | 2009-08-05 |
| CN101497602B true CN101497602B (en) | 2012-11-07 |
Family
ID=40944891
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN2008100571021A Expired - Fee Related CN101497602B (en) | 2007-04-30 | 2008-01-30 | Anthranilic acid compound and use thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN101497602B (en) |
Families Citing this family (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102020633B (en) * | 2009-09-21 | 2013-08-07 | 中国中化股份有限公司 | Method for preparing 1-(3,5- dichloropyridine-2-yl)-pyrazolecarboxamide compounds |
| CN102020634B (en) * | 2009-09-21 | 2013-05-22 | 中国中化股份有限公司 | Preparation method of N-(w-cyanoalkyl) benzamide compounds |
| CN101845043A (en) * | 2010-01-15 | 2010-09-29 | 江苏省农药研究所股份有限公司 | Ortho heterocycle formanilide compound and synthesis method and application thereof |
| CN101747325A (en) * | 2010-01-15 | 2010-06-23 | 江苏省农药研究所股份有限公司 | Neighboring Heterocyclic formanilide compound and synthesis method and application thereof |
| CN102060841B (en) * | 2010-11-26 | 2014-04-02 | 贵州大学 | Acylhydrazone containing heterocyclic amide structure, oxime ester compound and application thereof |
| CN102391248B (en) * | 2011-09-29 | 2016-05-11 | 杭州宇龙化工有限公司 | O-aminobenzonitrile compounds and method for making thereof and purposes |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1541063A (en) * | 2001-08-13 | 2004-10-27 | ��Ļ���Ű˾ | Method for controlling special pests using anthranilamide compounds |
| CN1713819A (en) * | 2001-09-21 | 2005-12-28 | 杜邦公司 | Anthranilamide arthropodicide treatment |
| CN1829707A (en) * | 2003-01-28 | 2006-09-06 | 杜邦公司 | Cyanoanthranilide Insecticide |
| CN101298451A (en) * | 2007-04-30 | 2008-11-05 | 中国中化集团公司 | Benzamide compounds and their applications |
-
2008
- 2008-01-30 CN CN2008100571021A patent/CN101497602B/en not_active Expired - Fee Related
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1541063A (en) * | 2001-08-13 | 2004-10-27 | ��Ļ���Ű˾ | Method for controlling special pests using anthranilamide compounds |
| CN1713819A (en) * | 2001-09-21 | 2005-12-28 | 杜邦公司 | Anthranilamide arthropodicide treatment |
| CN1829707A (en) * | 2003-01-28 | 2006-09-06 | 杜邦公司 | Cyanoanthranilide Insecticide |
| CN101298451A (en) * | 2007-04-30 | 2008-11-05 | 中国中化集团公司 | Benzamide compounds and their applications |
Also Published As
| Publication number | Publication date |
|---|---|
| CN101497602A (en) | 2009-08-05 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| ES2347521T3 (en) | DERIVATIVES OF FENACILAMINE, PRODUCTION OF THE SAME AND PEST CONTROLLERS CONTAINING THE DERIVATIVES. | |
| AU2020343788B2 (en) | Piperonylic acid derivative and application thereof | |
| CN101497602B (en) | Anthranilic acid compound and use thereof | |
| CN102119144B (en) | The preparation method of amide derivatives | |
| CN101311170B (en) | Substituted pyrimidine ether compounds and their applications | |
| CA2612507A1 (en) | Amide derivative, insecticide containing the same and method for application thereof as insecticide | |
| HU229576B1 (en) | Acylsulfamoyl benzoic acid amides, plant protection agents containing said acylsulfamoyl benzoic acid amides, and method for producing the same | |
| BRPI0917556B1 (en) | AMIDA DERIVATIVE, PEST CONTROL AGENT CONTAINING AMIDE DERIVATIVE AND PEST CONTROL METHOD | |
| UA123706C2 (en) | Pyridazinone-type compound or salt thereof, and herbicide comprising same | |
| EP4293015A1 (en) | Benzimidazole compound or salt thereof, preparation method therefor and use thereof, and insecticide and acaricide and use thereof | |
| CN103201266B (en) | Aryloxydihalopropene ether compounds and their applications | |
| US20070135497A1 (en) | Biphenyl derivative or its salt, and pesticide containing it as an active ingredient | |
| CN103539681A (en) | Substituted diphenylamine compounds and application thereof | |
| ES2256874T3 (en) | DERIVATIVES OF PIRIDONA AND HERBICIDES. | |
| US5807806A (en) | Benzoylpyrazole derivatives having specific substituents and herbicide | |
| HU207832B (en) | Herbicide compositions containing substituted bicycloheptane-dion derivatives and process for producing these compounds | |
| UA82509C2 (en) | Herbicidal composition comprising 3-phenyluracil, and using thereof in method to control undesirable vegetation | |
| EP4119545B1 (en) | Pyrazole carboxylate compound and use thereof | |
| DE3418167A1 (en) | Use of amides for improving the crop plant compatibility of herbicidally active heteroaryloxyacetamides | |
| AU2018257296B2 (en) | N-(4-pyridyl) nicotinamide compound or salt thereof | |
| TW201234966A (en) | Benzyloxypyrinidine derivatives, pesticide for agro-horticulture having said derivatives and method of using same | |
| JPH11302276A (en) | Sulfamoyl triazol derivative and fungicide composition for agricultural and horticultural use | |
| CN101203485A (en) | Amide derivatives, insecticides containing the same and methods of use thereof | |
| AU2020363066B2 (en) | Alkene-containing amide compound and use thereof | |
| EA012140B1 (en) | Herbicidal compositions based on 3-phenyluracils and 3-sulfonylisoxazolines |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C41 | Transfer of patent application or patent right or utility model | ||
| COR | Change of bibliographic data |
Free format text: CORRECT: ADDRESS; FROM: 100045 F6-F12/F, KAICHEN WORLD TRADE CENTER, NO.28, FUXINGMEN INNER STREET, BEIJING TO: 100031 NO.28, FUXINGMEN INNER STREET, XICHENG DISTRICT, BEIJING |
|
| TA01 | Transfer of patent application right |
Effective date of registration: 20100613 Address after: 100031 Beijing, Xicheng District, the door of the revitalization of the main street, No. 28 Applicant after: SINOCHEM Corp. Co-applicant after: SHENYANG RESEARCH INSTITUTE OF CHEMICAL INDUSTRY Co.,Ltd. Address before: 100045 Beijing City 28 fuxingmennei Avenue Chemsunny World Trade Center F6-F12 Applicant before: Sinochem Corp. Co-applicant before: SHENYANG RESEARCH INSTITUTE OF CHEMICAL INDUSTRY Co.,Ltd. |
|
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| C41 | Transfer of patent application or patent right or utility model | ||
| TR01 | Transfer of patent right |
Effective date of registration: 20160122 Address after: 110021 Liaodong Road, Tiexi District, Liaoning, No. 8-1, No. Patentee after: SHENYANG SINOCHEM AGROCHEMICALS R&D Co.,Ltd. Address before: 100031 Beijing, Xicheng District, the door of the revitalization of the main street, No. 28 Patentee before: SINOCHEM Corp. Patentee before: SHENYANG RESEARCH INSTITUTE OF CHEMICAL INDUSTRY Co.,Ltd. |
|
| CF01 | Termination of patent right due to non-payment of annual fee | ||
| CF01 | Termination of patent right due to non-payment of annual fee |
Granted publication date: 20121107 |