CN101432251A - 4-arylfluorene compound and organic light-emitting device using same - Google Patents
4-arylfluorene compound and organic light-emitting device using same Download PDFInfo
- Publication number
- CN101432251A CN101432251A CNA2007800152248A CN200780015224A CN101432251A CN 101432251 A CN101432251 A CN 101432251A CN A2007800152248 A CNA2007800152248 A CN A2007800152248A CN 200780015224 A CN200780015224 A CN 200780015224A CN 101432251 A CN101432251 A CN 101432251A
- Authority
- CN
- China
- Prior art keywords
- unsubstituted
- compound
- replacement
- organic
- general formula
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 150000001875 compounds Chemical class 0.000 title abstract description 78
- 150000002894 organic compounds Chemical class 0.000 claims abstract description 15
- NIHNNTQXNPWCJQ-UHFFFAOYSA-N o-biphenylenemethane Natural products C1=CC=C2CC3=CC=CC=C3C2=C1 NIHNNTQXNPWCJQ-UHFFFAOYSA-N 0.000 claims description 27
- GVEPBJHOBDJJJI-UHFFFAOYSA-N fluoranthene Chemical compound C1=CC(C2=CC=CC=C22)=C3C2=CC=CC3=C1 GVEPBJHOBDJJJI-UHFFFAOYSA-N 0.000 claims description 13
- 125000003983 fluorenyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3CC12)* 0.000 claims description 10
- 125000000217 alkyl group Chemical group 0.000 claims description 8
- 125000003710 aryl alkyl group Chemical group 0.000 claims description 8
- 125000003118 aryl group Chemical group 0.000 claims description 8
- 125000003545 alkoxy group Chemical group 0.000 claims description 6
- 125000000623 heterocyclic group Chemical group 0.000 claims description 6
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 5
- 125000005843 halogen group Chemical group 0.000 claims description 5
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 claims description 5
- 125000005428 anthryl group Chemical group [H]C1=C([H])C([H])=C2C([H])=C3C(*)=C([H])C([H])=C([H])C3=C([H])C2=C1[H] 0.000 claims description 4
- 150000004646 arylidenes Chemical group 0.000 claims description 3
- 125000001624 naphthyl group Chemical group 0.000 claims description 3
- 125000000843 phenylene group Chemical group C1(=C(C=CC=C1)*)* 0.000 claims description 2
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 51
- 239000010410 layer Substances 0.000 description 50
- 239000000758 substrate Substances 0.000 description 27
- 239000010408 film Substances 0.000 description 24
- 239000000203 mixture Substances 0.000 description 19
- -1 fluorene compound Chemical class 0.000 description 18
- 239000000463 material Substances 0.000 description 17
- 238000000034 method Methods 0.000 description 17
- 230000005540 biological transmission Effects 0.000 description 14
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 10
- 239000000243 solution Substances 0.000 description 10
- IMNFDUFMRHMDMM-UHFFFAOYSA-N N-Heptane Chemical compound CCCCCCC IMNFDUFMRHMDMM-UHFFFAOYSA-N 0.000 description 8
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 8
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 8
- 238000006243 chemical reaction Methods 0.000 description 8
- 239000012044 organic layer Substances 0.000 description 8
- 238000001771 vacuum deposition Methods 0.000 description 8
- 239000013078 crystal Substances 0.000 description 7
- 230000027756 respiratory electron transport chain Effects 0.000 description 7
- 238000005160 1H NMR spectroscopy Methods 0.000 description 6
- 238000001035 drying Methods 0.000 description 6
- 239000002904 solvent Substances 0.000 description 6
- 230000005525 hole transport Effects 0.000 description 5
- 238000002347 injection Methods 0.000 description 5
- 239000007924 injection Substances 0.000 description 5
- PQXKHYXIUOZZFA-UHFFFAOYSA-M lithium fluoride Chemical compound [Li+].[F-] PQXKHYXIUOZZFA-UHFFFAOYSA-M 0.000 description 5
- 239000012299 nitrogen atmosphere Substances 0.000 description 5
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N silicon dioxide Inorganic materials O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 5
- 238000003756 stirring Methods 0.000 description 5
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 4
- 239000004411 aluminium Substances 0.000 description 4
- 229910052782 aluminium Inorganic materials 0.000 description 4
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 4
- 239000007864 aqueous solution Substances 0.000 description 4
- 238000000151 deposition Methods 0.000 description 4
- 238000000605 extraction Methods 0.000 description 4
- 238000010438 heat treatment Methods 0.000 description 4
- MRNHPUHPBOKKQT-UHFFFAOYSA-N indium;tin;hydrate Chemical compound O.[In].[Sn] MRNHPUHPBOKKQT-UHFFFAOYSA-N 0.000 description 4
- 238000001819 mass spectrum Methods 0.000 description 4
- UQPUONNXJVWHRM-UHFFFAOYSA-N palladium;triphenylphosphane Chemical compound [Pd].C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 UQPUONNXJVWHRM-UHFFFAOYSA-N 0.000 description 4
- 229910002027 silica gel Inorganic materials 0.000 description 4
- 239000000741 silica gel Substances 0.000 description 4
- 229960001866 silicon dioxide Drugs 0.000 description 4
- 229910000029 sodium carbonate Inorganic materials 0.000 description 4
- 235000017550 sodium carbonate Nutrition 0.000 description 4
- 229910052938 sodium sulfate Inorganic materials 0.000 description 4
- 235000011152 sodium sulphate Nutrition 0.000 description 4
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 4
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 3
- 230000008021 deposition Effects 0.000 description 3
- 238000005137 deposition process Methods 0.000 description 3
- 230000006866 deterioration Effects 0.000 description 3
- 238000010790 dilution Methods 0.000 description 3
- 239000012895 dilution Substances 0.000 description 3
- 239000011521 glass Substances 0.000 description 3
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 3
- 238000001228 spectrum Methods 0.000 description 3
- 230000002194 synthesizing effect Effects 0.000 description 3
- 230000007704 transition Effects 0.000 description 3
- 238000001291 vacuum drying Methods 0.000 description 3
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 2
- 239000004925 Acrylic resin Substances 0.000 description 2
- 229920000178 Acrylic resin Polymers 0.000 description 2
- 229910001148 Al-Li alloy Inorganic materials 0.000 description 2
- NLXLAEXVIDQMFP-UHFFFAOYSA-N Ammonia chloride Chemical compound [NH4+].[Cl-] NLXLAEXVIDQMFP-UHFFFAOYSA-N 0.000 description 2
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 2
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 2
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 2
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 2
- CBENFWSGALASAD-UHFFFAOYSA-N Ozone Chemical compound [O-][O+]=O CBENFWSGALASAD-UHFFFAOYSA-N 0.000 description 2
- ABRVLXLNVJHDRQ-UHFFFAOYSA-N [2-pyridin-3-yl-6-(trifluoromethyl)pyridin-4-yl]methanamine Chemical compound FC(C1=CC(=CC(=N1)C=1C=NC=CC=1)CN)(F)F ABRVLXLNVJHDRQ-UHFFFAOYSA-N 0.000 description 2
- 238000010521 absorption reaction Methods 0.000 description 2
- 239000000853 adhesive Substances 0.000 description 2
- 230000001070 adhesive effect Effects 0.000 description 2
- 239000012298 atmosphere Substances 0.000 description 2
- CUFNKYGDVFVPHO-UHFFFAOYSA-N azulene Chemical compound C1=CC=CC2=CC=CC2=C1 CUFNKYGDVFVPHO-UHFFFAOYSA-N 0.000 description 2
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 2
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 2
- 229910052794 bromium Inorganic materials 0.000 description 2
- 229910052801 chlorine Inorganic materials 0.000 description 2
- 239000000460 chlorine Substances 0.000 description 2
- 230000000052 comparative effect Effects 0.000 description 2
- 238000000113 differential scanning calorimetry Methods 0.000 description 2
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 2
- 229910052731 fluorine Inorganic materials 0.000 description 2
- 239000011737 fluorine Substances 0.000 description 2
- 229910052740 iodine Inorganic materials 0.000 description 2
- 239000011630 iodine Substances 0.000 description 2
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 2
- 125000005561 phenanthryl group Chemical group 0.000 description 2
- 125000000951 phenoxy group Chemical group [H]C1=C([H])C([H])=C(O*)C([H])=C1[H] 0.000 description 2
- 238000002360 preparation method Methods 0.000 description 2
- 125000002572 propoxy group Chemical group [*]OC([H])([H])C(C([H])([H])[H])([H])[H] 0.000 description 2
- 125000004076 pyridyl group Chemical group 0.000 description 2
- 238000004544 sputter deposition Methods 0.000 description 2
- 125000001424 substituent group Chemical group 0.000 description 2
- 125000001544 thienyl group Chemical group 0.000 description 2
- 238000004506 ultrasonic cleaning Methods 0.000 description 2
- YJTKZCDBKVTVBY-UHFFFAOYSA-N 1,3-Diphenylbenzene Chemical group C1=CC=CC=C1C1=CC=CC(C=2C=CC=CC=2)=C1 YJTKZCDBKVTVBY-UHFFFAOYSA-N 0.000 description 1
- YBYIRNPNPLQARY-UHFFFAOYSA-N 1H-indene Natural products C1=CC=C2CC=CC2=C1 YBYIRNPNPLQARY-UHFFFAOYSA-N 0.000 description 1
- DFZYPLLGAQIQTD-UHFFFAOYSA-N 2,7-ditert-butyl-9h-fluorene Chemical compound CC(C)(C)C1=CC=C2C3=CC=C(C(C)(C)C)C=C3CC2=C1 DFZYPLLGAQIQTD-UHFFFAOYSA-N 0.000 description 1
- MZSAMHOCTRNOIZ-UHFFFAOYSA-N 3-[4-(aminomethyl)-6-(trifluoromethyl)pyridin-2-yl]oxy-N-phenylaniline Chemical compound NCC1=CC(=NC(=C1)C(F)(F)F)OC=1C=C(NC2=CC=CC=C2)C=CC=1 MZSAMHOCTRNOIZ-UHFFFAOYSA-N 0.000 description 1
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 1
- DGEZNRSVGBDHLK-UHFFFAOYSA-N [1,10]phenanthroline Chemical compound C1=CN=C2C3=NC=CC=C3C=CC2=C1 DGEZNRSVGBDHLK-UHFFFAOYSA-N 0.000 description 1
- FCVHBUFELUXTLR-UHFFFAOYSA-N [Li].[AlH3] Chemical compound [Li].[AlH3] FCVHBUFELUXTLR-UHFFFAOYSA-N 0.000 description 1
- 125000001389 aceanthrenyl group Chemical group C1(CC2=CC=CC3=CC4=CC=CC=C4C1=C23)* 0.000 description 1
- 125000004062 acenaphthenyl group Chemical group C1(CC2=CC=CC3=CC=CC1=C23)* 0.000 description 1
- 125000003670 adamantan-2-yl group Chemical group [H]C1([H])C(C2([H])[H])([H])C([H])([H])C3([H])C([*])([H])C1([H])C([H])([H])C2([H])C3([H])[H] 0.000 description 1
- 230000032683 aging Effects 0.000 description 1
- 235000019270 ammonium chloride Nutrition 0.000 description 1
- 125000005605 benzo group Chemical group 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 230000000903 blocking effect Effects 0.000 description 1
- 229910000085 borane Inorganic materials 0.000 description 1
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 239000000919 ceramic Substances 0.000 description 1
- 238000002425 crystallisation Methods 0.000 description 1
- 230000008025 crystallization Effects 0.000 description 1
- 230000018109 developmental process Effects 0.000 description 1
- AAXGWYDSLJUQLN-UHFFFAOYSA-N diphenyl(propyl)phosphane Chemical compound C=1C=CC=CC=1P(CCC)C1=CC=CC=C1 AAXGWYDSLJUQLN-UHFFFAOYSA-N 0.000 description 1
- 239000002019 doping agent Substances 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 150000002219 fluoranthenes Chemical class 0.000 description 1
- 230000005283 ground state Effects 0.000 description 1
- 238000004770 highest occupied molecular orbital Methods 0.000 description 1
- 125000003427 indacenyl group Chemical group 0.000 description 1
- 125000003454 indenyl group Chemical group C1(C=CC2=CC=CC=C12)* 0.000 description 1
- 238000009413 insulation Methods 0.000 description 1
- 230000002452 interceptive effect Effects 0.000 description 1
- 238000004768 lowest unoccupied molecular orbital Methods 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- QMMRZOWCJAIUJA-UHFFFAOYSA-L nickel dichloride Chemical compound Cl[Ni]Cl QMMRZOWCJAIUJA-UHFFFAOYSA-L 0.000 description 1
- 125000002347 octyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- WCPAKWJPBJAGKN-UHFFFAOYSA-N oxadiazole Chemical compound C1=CON=N1 WCPAKWJPBJAGKN-UHFFFAOYSA-N 0.000 description 1
- 125000002971 oxazolyl group Chemical group 0.000 description 1
- 125000003933 pentacenyl group Chemical group C1(=CC=CC2=CC3=CC4=CC5=CC=CC=C5C=C4C=C3C=C12)* 0.000 description 1
- GUVXZFRDPCKWEM-UHFFFAOYSA-N pentalene Chemical compound C1=CC2=CC=CC2=C1 GUVXZFRDPCKWEM-UHFFFAOYSA-N 0.000 description 1
- 125000002080 perylenyl group Chemical group C1(=CC=C2C=CC=C3C4=CC=CC5=CC=CC(C1=C23)=C45)* 0.000 description 1
- CSHWQDPOILHKBI-UHFFFAOYSA-N peryrene Natural products C1=CC(C2=CC=CC=3C2=C2C=CC=3)=C3C2=CC=CC3=C1 CSHWQDPOILHKBI-UHFFFAOYSA-N 0.000 description 1
- 125000001828 phenalenyl group Chemical group C1(C=CC2=CC=CC3=CC=CC1=C23)* 0.000 description 1
- 239000004033 plastic Substances 0.000 description 1
- 229920003023 plastic Polymers 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000001725 pyrenyl group Chemical group 0.000 description 1
- 239000010453 quartz Substances 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 238000001953 recrystallisation Methods 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 238000004062 sedimentation Methods 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 125000001935 tetracenyl group Chemical group C1(=CC=CC2=CC3=CC4=CC=CC=C4C=C3C=C12)* 0.000 description 1
- 125000001113 thiadiazolyl group Chemical group 0.000 description 1
- 125000000335 thiazolyl group Chemical group 0.000 description 1
- 239000010409 thin film Substances 0.000 description 1
- UORVGPXVDQYIDP-UHFFFAOYSA-N trihydridoboron Substances B UORVGPXVDQYIDP-UHFFFAOYSA-N 0.000 description 1
Images
Landscapes
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Electroluminescent Light Sources (AREA)
Abstract
The present invention provides a high-performance organic light-emitting device, and a novel organic compound for use in the device. The novel compound of the present invention is a 4-arylfluorene compound having the following general formula (1). The organic light-emitting device of the present invention is an organic light-emitting device including: a pair of electrodes comprising an anode and a cathode; and an organic compound layer interposed between the pair of electrodes, wherein the organic compound layer contains the 4-arylfluorene compound.
Description
Technical field
The present invention relates to the organic luminescent device of novel 4-aryl fluorene compound and this compound of use.
Background technology
Organic luminescent device comprises: anode; Negative electrode; With the film that contains fluorescence organic compound or phosphorescence organic compound, this film is clamped between this anode and this negative electrode.Inject electronics and hole from each electrode to this film.
In addition, this organic luminescent device produces the exciton of this fluorescence organic compound or this phosphorescence organic compound.The light that this devices use is sent when this exciton returns its ground state.
The nearest development of organic luminescent device is remarkable.Have and to prepare thin, the lightweight organic light-emitting device characteristic that under low impressed voltage, has high brightness, various emission wavelength and high-speed response.Therefore, these characteristics hint that this organic luminescent device can use in multiple use.
But conventional organic luminescent device need have more high brightness or more the light output of high conversion efficiency.In addition, this organic luminescent device still relates to many problems aspect weather resistance, for example since life-time service cause through the time change and owing to for example contain the atmospheric deterioration of aerobic or moisture.
In addition, when attempting this device application, must send blue light, green glow and ruddiness with good purity of color in full-color display etc.But, relate to luminous problem and still do not solved fully.
In addition, use the material and the organic light-emitting device example of fluorene compound to be disclosed in Japanese Patent Application Publication No.H11-288783, H11-185960, H11-204262,2002-154993,2004-043349 and 2005-239650.But disclosed device has low luminous efficiency and inadequate endurance life in these applications.In addition, these the application in some for endurance life without any explanation.
Summary of the invention
The object of the present invention is to provide novel 4-aryl fluorene compound.
Another object of the present invention is to provide and use this 4-aryl fluorene compound and have extreme efficiency and the organic luminescent device of the light of high brightness output.Another object of the present invention is to provide organic luminescent device with high weather resistance.
Another object of the present invention is to provide the organic luminescent device that can easily prepare with low relatively cost.
According to the present invention, provide 4-aryl fluorene compound by following general formula (I) expression:
Wherein: n represents the integer of 0-10; When n represented 0, Ar represented the Direct Bonding between fluorenyl and the fluoranthene base; When n represented the integer of 1-10, Ar represented to replace or the inferior aralkyl of unsubstituted divalent alkyl, replacement or unsubstituted divalence, replacement or unsubstituted divalence arylidene or replacement or unsubstituted divalent heterocycle; When n represents the integer of 1-10, can be same to each other or different to each other between the Ar; R
1And R
2Represent replacement or unsubstituted alkyl, replacement or unsubstituted aralkyl, replacement or unsubstituted alkoxyl group, replacement or unsubstituted aryl, replacement or unsubstituted heterocyclic, replacement or unsubstituted amino, cyano group or halogen group separately, and R
1And R
2Can be same to each other or different to each other; X and y represent the integer of 0-9 separately; When x or y represent integer more than 2, R
1Between or R
2Between can be same to each other or different to each other perhaps R
1Between or R
2Between each other bonding to form ring.
4-aryl fluorene compound by general formula (I) expression of the present invention has good film-forming properties, and sends the blue light with excellent color purity.In addition, use the organic luminescent device of this 4-aryl fluorene compound hanging down under the impressed voltage with high efficiency light-emitting.
With reference to the explanation of accompanying drawing to exemplary, it is clear that further aspect of the present invention will become by following.
Description of drawings
Fig. 1 is the sectional view of organic light-emitting device example among expression the present invention.
Fig. 2 is the sectional view of another example of organic light-emitting device among expression the present invention.
Fig. 3 is the sectional view of another example of organic light-emitting device among expression the present invention.
Fig. 4 is the sectional view of another example of organic light-emitting device among expression the present invention.
Fig. 5 is the sectional view of another example of organic light-emitting device among expression the present invention.
Fig. 6 is an exemplary compounds 4
1HNMR figure.
Fig. 7 is an exemplary compounds 22
1HNMR figure.
Fig. 8 is an exemplary compounds 5
1HNMR figure.
Embodiment
According to the present invention, provide 4-aryl fluorene compound by following general formula (I) expression:
Wherein, n represents the integer of 0-10; When n represented 0, Ar represented the Direct Bonding between fluorenyl and the fluoranthene base; When n represented the integer of 1-10, Ar represented to replace or the inferior aralkyl of unsubstituted divalent alkyl, replacement or unsubstituted divalence, replacement or unsubstituted divalence arylidene or replacement or unsubstituted divalent heterocycle; When n represents the integer of 1-10, can be same to each other or different to each other between the Ar; R
1And R
2Represent replacement or unsubstituted alkyl, replacement or unsubstituted aralkyl, replacement or unsubstituted alkoxyl group, replacement or unsubstituted aryl, replacement or unsubstituted heterocyclic, replacement or unsubstituted amino, cyano group or halogen group separately, and R
1And R
2Can be same to each other or different to each other; X and y represent the integer of 0-9 separately; When x or y represent integer more than 2, R
1Between or R
2Between can be same to each other or different to each other perhaps R
1Between or R
2Between each other bonding to form ring.
In addition, according to the present invention, provide 4-aryl fluorene compound, wherein Ar represents to replace or unsubstituted phenylene in general formula (I).
In addition, according to the present invention, provide 4-aryl fluorene compound, wherein n represents 0 in general formula (I).
In addition, according to the present invention, provide 4-aryl fluorene compound, wherein Ar represents to replace or unsubstituted naphthyl in general formula (I).
In addition, according to the present invention, provide 4-aryl fluorene compound, wherein Ar represents to replace or unsubstituted anthryl in general formula (I).
In addition, according to the present invention, provide organic luminescent device, it comprises: the pair of electrodes with anode and negative electrode; And be clamped in organic compound layer between this counter electrode, wherein this organic compound layer contains any of above-mentioned 4-aryl fluorene compound.
In addition, according to the present invention, provide organic luminescent device, wherein this organic compound layer is a luminescent layer.
The substituent specific examples of compound is as described below in the general formula (I).
The example of alkyl comprises methyl, ethyl, n-propyl, sec.-propyl, normal-butyl, the tertiary butyl, sec-butyl, octyl group, 1-adamantyl, 2-adamantyl etc.
The example of aralkyl comprises benzyl and styroyl.
The example of alkoxyl group (alkoxy group) comprises methoxyl group, oxyethyl group, propoxy-and phenoxy group.
The example of aryl comprise phenyl, naphthyl, pentalene base, indenyl, Azulene base, anthryl, pyrenyl, indacenyl base, acenaphthenyl, phenanthryl, phenalenyl base, fluoranthene base, acephenanthyl base, aceanthrenyl, benzo [9,10] phenanthryl,
Base, naphthacenyl, perylene base, pentacenyl, xenyl, terphenyl and fluorenyl.
The example of heterocyclic radical comprises thienyl, pyrryl, pyridyl, oxazolyl, oxadiazole base, thiazolyl, thiadiazolyl group, terthienyl base, carbazyl, acridyl, phenanthroline (phenanthroyl) base etc.
The example of halogen atom comprises fluorine, chlorine, bromine, iodine etc.
Amino example comprises dimethylamino, diethylamino, dibenzyl amino, diphenyl amino, two (tolyl) amino, two (p-methoxy-phenyl) amino etc.
The substituent example that above-mentioned substituting group can have comprises: alkyl is methyl, ethyl and propyl group for example; Aralkyl is benzyl and styroyl for example; Aryl is phenyl and xenyl for example; Heterocyclic radical is thienyl, pyrryl and pyridyl for example; Amino is dimethylamino, diethylamino, dibenzyl amino, diphenyl amino, two (tolyl) amino and two (p-methoxy-phenyl) amino for example; Alkoxyl group is methoxyl group, oxyethyl group, propoxy-and phenoxy group for example; Cyano group; Cyano group; With halogen atom for example fluorine, chlorine, bromine and iodine.
4-aryl fluorene compound by general formula (I) expression can be used as the organic light-emitting device material.
In this device, can be used in each layer in hole transmission layer, electron transfer layer and the luminescent layer by the 4-aryl fluorene compound of general formula (I) expression.The result can access has high-luminous-efficiency and long-life device.
In addition, when the 4-aryl fluorene compound of general formula (I) expression is used for luminescent layer, have high color purity by using this compound according to any of various patterns, can accessing, high-luminous-efficiency and long-life device.
For example, this compound can be used separately in luminescent layer or uses as the material of main part of each in doping agent (object) material, fluorescent material and the phosphor material.The result can access have high color purity, high-luminous-efficiency and long-life device.
In the 4-aryl fluorene compound of general formula (I) expression, replace with the 4-position of aryl with this fluorenyl.This replacement makes the whole molecule of this compound to design with nonplanar structure.Therefore the result can provide the molecule with high amorphism, and can access and have high thermal stability and long-life device.In addition, reduce the emission of molecular association by the on-plane surface molecular structure of the 4-aryl fluorene compound of general formula (I) expression, so this compound is useful as the blue light-emitting material with the light that suppresses to produce with long wavelength owing to this association.In addition, substituting group is introduced this fluoranthene base the good green light or the material that glows can be provided.
In addition, the Ar base that substitutes in the 4-aryl fluorene compound shown in the general formula (I) with Direct Bonding or phenyl broadens the band gap of the whole molecule of this compound, and this compound becomes useful as the blue light-emitting material thus.In addition, with the fused polycycle group for example anthryl or fluorenyl substitute this Ar base the band gap of the whole molecule of this compound narrowed down, this compound becomes useful as the green light or the material that glows thus.
In addition, the fluorenyl or the fluoranthene base that are replaced by any other aryl can provide the device with high-luminous-efficiency, and reason is that these groups have high-luminous-efficiency.
By substituting group being incorporated in this fluorenyl or the fluoranthene base, can easily adjust the HOMO/LUMO energy level of the 4-aryl fluorene compound of general formula (I) expression.
Therefore, considering that the current carrier between hole and the electronics injects equilibrated simultaneously, can easily design the molecule of this compound.
Result as based on the molecular designing of aforesaid discussion has finished the present invention.
Below the present invention is described in more detail.
Specific examples by the compound of above-mentioned general formula (I) expression is as follows.But the present invention is not limited to these examples.
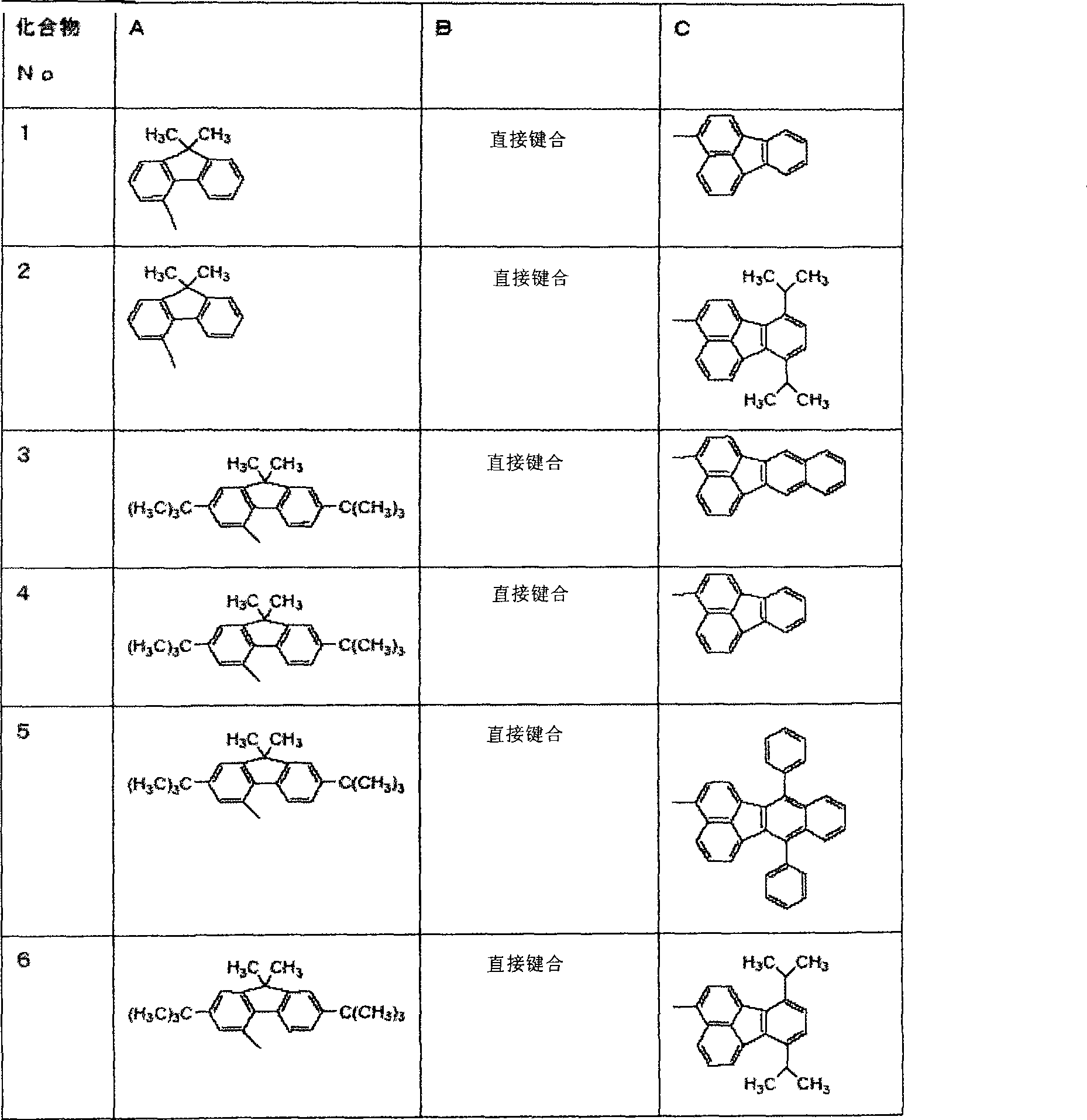
In the table 1, will be used for compound of the present invention and be expressed as A-B-C, and B represents the position of A and C bonding.That is expression exemplary compounds 9 as follows.Although letter b is shown in the fluorenyl of being represented by A, the 4-position of the group of being represented by A is replaced by aryl (B).In order to be more readily understood, in each and table 1 of following formula, the 4-position at fluorenyl A in A shows bonding end.
Table 1
Next, organic luminescent device of the present invention is elaborated.
Organic luminescent device of the present invention comprises: the pair of electrodes with anode and negative electrode; Contain one or more layers of organic compound separately, this one or more layers be clamped between this counter electrode, wherein this contains the compound that one deck at least in one or more layers of organic compound contains at least a general formula (I) expression separately.
Fig. 1,2,3,4 and 5 represents organic light-emitting device preferred embodiment of the present invention separately.
Fig. 1 is the sectional view of expression organic light-emitting device example of the present invention.As shown in fig. 1, this organic luminescent device has wherein anode 2, luminescent layer 3 and negative electrode 4 is provided at structure on the substrate 1 in proper order by this.The compound that itself has hole transport performance, electronic transmission performance and luminescent properties or will have each performance at this luminescent layer is used under the situation of this luminescent layer, and this luminescent device is useful as used herein.
Fig. 2 is the sectional view of expression another example of organic light-emitting device of the present invention.As shown in Figure 2, this organic luminescent device has wherein anode 2, hole transmission layer 5, electron transfer layer 6 and negative electrode 4 is provided at structure on the substrate 1 in proper order by this.Be used for each layer will having one of hole transport performance and electronic transmission performance or both materials, and with luminophore with do not have under the situation that the hole transport ability material of luminescent properties or electron-transporting combinations of substances use, this luminophore is useful.In this case, this luminescent layer is formed by this hole transmission layer 5 or this electron transfer layer 6.
Fig. 3 is the sectional view of expression another example of organic light-emitting device of the present invention.As shown in Figure 3, this organic luminescent device has wherein anode 2, hole transmission layer 5, luminescent layer 3, electron transfer layer 6 and negative electrode 4 is provided at structure on the substrate 1 in proper order by this.This organic luminescent device has isolating carrier transport function and lighting function.Use the device that wherein will have the compound appropriate combination of hole transport performance, electronic transmission performance or luminescent properties separately, can make thus and select the degree of freedom of employed material to significantly improve.In addition, can use all cpds, the diversity of glow color is improved with different emission wavelengths.In addition, in this luminescent layer 3 that provides by the central authorities that each current carrier or exciton are captured in efficiently at this device, luminous efficiency is improved.
Fig. 4 is the sectional view of expression another example of organic light-emitting device of the present invention.Fig. 4 has the structure shown in Fig. 3, and difference is to insert hole injection layer 7 in anode 2 sides.This structure is for improving binding property between this anode 2 and this hole transmission layer 5 or effective for improving the hole injection efficiency, and this is effective for reducing the voltage that this device is added.
Fig. 5 is the sectional view of expression another example of organic light-emitting device of the present invention.Fig. 5 has the structure shown in Fig. 3, and difference is between this luminescent layer 3 and this electron transfer layer 6 to insert blocking hole or the exciton transition layer (hole/exciton barrier-layer 8) to these negative electrode 4 sides.In this structure, the compound that will have high ionization potential is used for this hole/exciton barrier-layer 8, and this structure is effective for improving luminous efficiency.
Fig. 1,2,3,4 and 5 represents basic device architecture separately, and uses the organic light-emitting device structure of compound of the present invention to be not limited to the structure shown in Fig. 1,2,3,4 and 5.For example, organic luminescent device of the present invention can have any in the various layer structure, and these various layer structures comprise: the structure that insulation layer is provided at the interface between electrode and organic layer wherein; The structure of bonding coat or interfering layer wherein is provided; Wherein hole transmission layer comprises the two-layer structure that ionization potential is different.
The compound of employed general formula (I) expression can use with any form shown in Fig. 1,2,3,4 and 5 among the present invention.
Specifically, form the organic layer that uses compound of the present invention, cause crystallization etc. hardly by for example vacuum deposition method or solution coat method, and the ageing stability excellence.
In the present invention, especially with the composition of the compound shown in the general formula (I) as luminescent layer.Compound with the invention described above such as known lower molecular weight class or polymer class hole transport compound, luminophor, electric transmission compound can be used as required.
There is no particular restriction for employed substrate among the present invention, but the example comprises: opaque substrate is metal substrate or ceramic substrate for example; With transparency carrier for example glass substrate, quartz base plate or plastics sheet substrate.
In addition, this substrate can have colour filter film, fluorescence color conversion colour filter film, dielectric reflections film etc. and is used to control glow color.In addition, thin film transistor (TFT) can be on substrate, prepared, device of the present invention can be prepared then to be connected with TFT.
About the light emission direction of device, this device can have bottom-emission structure (from the luminous structure of substrate-side) or top lighting structure (from the luminous structure of the opposition side of substrate).
Below, come the present invention is more specifically illustrated with reference to following embodiment, but the present invention is not limited to these embodiment.
<embodiment 1 〉
Synthesizing of exemplary compounds 4
(a) midbody compound 1-1's is synthetic
By to as raw-material 2,7-di-tert-butyl-fluorene (Sigma-AldrichCorporation) carry out dimethylization, can prepare midbody compound 1-1.
(b) exemplary compounds 4 is synthetic
0.66g (1.70mmol) midbody compound 1-1,0.656g (2.00mmol) compound 1-2,120ml toluene and 20ml ethanol are packed in the 200-ml three-necked flask, and at room temperature under nitrogen atmosphere, stir this mixture.In this whipping process, splash into by 10g yellow soda ash is dissolved in the aqueous solution for preparing in the 100ml water to this mixture, then 0.20g (0.170mmol) four (triphenyl phosphine) palladium (0) is added in this mixture.Resultant is heated to 77 ℃, stirred then 5 hours.After the reaction, carry out drying with toluene extraction organic layer and with anhydrous sodium sulphate.After this, resultant is made with extra care, obtained 0.518g exemplary compounds 4 (yellow-white crystal) (yield 60.1%) thus with silicagel column (the Mixed Expansion solvent that contains heptane and toluene).
Mass spectrum confirms that the M+ of this compound is 506.3.
In addition,
1HNMR measures the structure (Fig. 6) of having identified exemplary compounds 4.In addition, differential scanning calorimetry (DSC) confirms that the fusing point of this compound is that 287 ℃ and second-order transition temperature are 122 ℃.
In addition, this compound is at dilution with toluene solution (1 * 10
-5Mol/l) emmission spectrum in is as follows: observe at 460nm and have the luminous by force of peak.
<embodiment 2 〉
Synthesizing of exemplary compounds 22
(a) midbody compound 1-3's is synthetic
With 12.0g (31.5mmol) midbody compound 1-1,1.70g (3.15mmol) [1, two (diphenylphosphino) propane of 3-] dichloro nickel, 120ml toluene and 20ml triethylamine pack in the 200-ml three-necked flask, and at room temperature stir this mixture under nitrogen atmosphere.In this whipping process, in this mixture, splash into 13.7ml (94.5mmol) tetramethyl-tetramethyl ethylene ketone borine.With this mixture heating up to 80 ℃, stirred then 8 hours.After the reaction, with the reaction soln cool to room temperature, in this reaction soln, add the aqueous ammonium chloride solution of the 1N of 100ml, and this mixing solutions was stirred 30 minutes.Carry out drying with toluene extraction organic layer and with anhydrous sodium sulphate.After this, resultant is made with extra care, obtained 9.34g midbody compound 1-3 (white crystal) (yield 72.0%) thus with silicagel column (the Mixed Expansion solvent that contains heptane and toluene).
(b) exemplary compounds 1-5's is synthetic
1.68g (5.02mmol) midbody compound 1-4,2.31g (5.52mmol) midbody compound 1-3,80ml toluene and 40ml ethanol are packed in the 200-ml three-necked flask, and at room temperature under nitrogen atmosphere, stir this mixture.In this whipping process, splash into by 1g yellow soda ash is dissolved in the aqueous solution for preparing in the 100ml water to this mixture, then 0.579g (0.502mmol) four (triphenyl phosphine) palladium (0) is added in this mixture.With this mixture heating up to 65 ℃, stirred then 5 hours.After the reaction, carry out drying with toluene extraction organic layer and with anhydrous sodium sulphate.After this, resultant is made with extra care, obtained 1.84g midbody compound 1-5 (yellow-white crystal) (yield 65.1%) thus with silicagel column (the Mixed Expansion solvent that contains heptane and toluene).
Mass spectrum confirms that the M+ of this compound is 561.
(c) exemplary compounds 22 is synthetic
1.10g (1.96mmol) midbody compound 1-5,0.709g (2.16mmol) compound 1-2,80ml toluene and 40ml ethanol are packed in the 200-ml three-necked flask, and at room temperature under nitrogen atmosphere, stir this mixture.In this whipping process, in this mixture, splash into by 1.41g yellow soda ash is dissolved in the aqueous solution for preparing in the 100ml water, then 0.227g (0.196mmol) four (triphenyl phosphine) palladium (0) is added in this mixture.With this mixture heating up to 77 ℃, stirred then 5 hours.After the reaction, carry out drying with toluene extraction organic layer and with anhydrous sodium sulphate.After this, resultant is made with extra care, obtained 0.920g exemplary compounds 22 (yellow-white crystal) (yield 69%) thus with silicagel column (the Mixed Expansion solvent that contains heptane and toluene).
Mass spectrum confirms that the M+ of this compound is 715.
In addition,
1HNMR measures the structure (Fig. 7) of having identified exemplary compounds 22.In addition, differential scanning calorimetry (DSC) confirms that the fusing point of this compound is that 287 ℃ and second-order transition temperature are 122 ℃.
In addition, as this compound at dilution with toluene solution (1 * 10
-5Mol/l) emmission spectrum in is observed at 469nm and is had the luminous by force of peak.
<embodiment 3 〉
Synthesizing of exemplary compounds 5
0.39g (1.00mmol) midbody compound 1-1,0.530g (1.00mmol) compound 1-6,10ml toluene and 50ml ethanol are packed in the 100-ml three-necked flask, and at room temperature under nitrogen atmosphere, stir this mixture.In this whipping process, splash into by 10g yellow soda ash is dissolved in the aqueous solution for preparing in the 10ml water to this mixture, then 0.06g (0.05mmol) four (triphenyl phosphine) palladium (0) is added in this mixture.With this mixture heating up to 77 ℃, stirred then 5 hours.Behind resultant cool to room temperature, the crystal of filtering-depositing, and the coarse crystal that generates is carried out recrystallization with toluene and ethanol.Obtain 0.120g exemplary compounds 5 (yellowish white crystal) (yield 16.9%) thus.
Mass spectrum confirms that the M+ of this compound is 708.4.
In addition,
1HNMR measures the structure (Fig. 8) of having identified exemplary compounds 5.
In addition, as this compound at dilution with toluene solution (1 * 10
-5Mol/l) emmission spectrum in is observed at 460nm and is had the luminous by force of peak.
<embodiment 4 〉
Method by the following stated prepares the organic luminescent device with structure shown in Fig. 3.
On glass substrate as substrate 1, form tin indium oxide (ITO) by sputtering method with film as anode 2 with 120nm thickness, and with products therefrom as the transparent conductivity support substrate.The gained substrate is carried out ultrasonic cleaning in acetone and Virahol (IPA) successively.Then this substrate is cleaned in ebullient IPA and carry out drying.This substrate is further carried out the UV/ ozone clean to be used as the transparent conductivity support substrate.
Preparation contains the chloroformic solution of the compound of being represented by following structural formula 3-1 of 0.1 weight % as hole mobile material.
On above-mentioned ITO electrode, at first the rotating speed with 500rpm keeps 10 seconds then with 1 with this drips of solution, and the rotating speed of 000rpm kept 1 minute, to form film by being spin-coated on the ITO electrode.Then, the substrate that will have film on ITO is placed in 80 ℃ of vacuum drying ovens and dry 10 minutes, fully to remove the solvent in this film thus.The hole transmission layer 5 of Xing Chenging has the thickness of 15nm like this.
Next, will on this hole transmission layer 5, provide the luminescent layer 3 of thickness 40nm as the above-mentioned exemplary compounds No.4 of first compound with as following structural formula 3-2 (with the weight ratio 10:90) codeposition of second compound.In this codeposition process, vacuum tightness is 1.0 * 10
-4Pa and rate of film build are second 0.2nm/ second~0.3nm/.
In addition, by using 2,9-[2-(9,9 '-the dimethyl fluorenyl)]-1, the vacuum deposition method of 10-phenanthroline forms the electron transfer layer 6 with 30nm thickness.In this vacuum deposition process, vacuum tightness is 1.0 * 10
-4Pa and rate of film build are second 0.2nm/ second~0.3nm/.
Next, on the organic layer that obtains, form aluminium lithium (AlLi) by vacuum deposition method with 0.5nm thickness, and by vacuum deposition method further form thereon have 150nm thickness the aluminium film as electron injection electrode (negative electrode 4), prepare organic luminescent device thus.Vacuum tightness is 1.0 * 10 in the deposition process
-4Pa.According to the condition that forms, the lithium fluoride rate of film build is 0.05nm/ second, and the aluminium rate of film build is second 1.0nm/ second~1.2nm/.
In dry air atmosphere, cover the organic EL device obtained and seal the device deterioration that causes with the absorption that prevents owing to moisture on it with the acrylic resin adhesive material with cover plate.
Adding under the voltage of 4V with the device of this A1 electrode (negative electrode 4) as negative pole as anodal to this ITO electrode (anode 2) that has that obtains like this, observing luminosity is 450cd/m
2And luminous efficiency is the blue-light-emitting of 2.81m/W.
<embodiment 5 〉
Method by the following stated prepares the organic luminescent device with structure shown in Fig. 4.
On glass substrate as substrate 1, by sputtering method form have 120nm thickness tin indium oxide (ITO) film as anode 2, and with products therefrom as the transparent conductivity support substrate.The gained substrate is carried out ultrasonic cleaning in acetone and Virahol (IPA) successively.Then this substrate is cleaned in ebullient IPA and in 120 ℃ vacuum drying oven, carry out drying.This substrate is further carried out the UV/ ozone clean to be used as the transparent conductivity support substrate.
Preparation contains the chloroformic solution of the compound of being represented by following structural formula 3-3 of 0.1 weight % as hole mobile material.
This drips of solution on above-mentioned ITO electrode, and is at first kept 10 seconds then with 1 with the rotating speed of 500rpm, and the rotating speed of 000rpm kept 40 seconds, formed film by being spin-coated on this ITO electrode.Then, products therefrom is placed in 80 ℃ of vacuum drying ovens and dry 10 minutes, fully to remove the solvent in this film thus.The hole transmission layer 7 of Xing Chenging has the thickness of 15nm like this.
Next, on this hole injection layer 7 further the compound represented by following structural formula 3-4 of vapour deposition so that the hole transmission layer 5 with 15nm thickness to be provided.
And then, will as the above-mentioned exemplary compounds 22 of first compound and as compound (with the weight ratio 5:95) codeposition of representing by said structure formula 3-2 of second compound on products therefrom so that the luminescent layer 3 with 30nm thickness to be provided.This layer forms under the following conditions, and this condition comprises: the vacuum tightness during vapour deposition is 1.0 * 10
-4Film sedimentation rate when Pa and vapour deposition is second 0.1nm/ second~0.2nm/.
In addition, have 2 of 30nm thickness by vacuum deposition method formation, 9-[2-(9,9 '-the dimethyl fluorenyl)]-1, the electron transfer layer 6 of 10-phenanthroline.In the deposition process, vacuum tightness is 1.0 * 10
-4Pa and rate of film build are second 0.1nm/ second~0.2nm/.
Next, on the organic layer that obtains like this, form lithium fluoride (LiF) film by vacuum deposition method with 0.5nm thickness, and by vacuum deposition method further form thereon have 150nm thickness the aluminium film as electron injection electrode (negative electrode 4), prepare organic luminescent device thus.Vacuum tightness is 1.0 * 10 in the deposition process
-4Pa.According to the condition that forms, the lithium fluoride rate of film build is 0.01nm/ second, and the aluminium rate of film build is second 1.0nm/ second~1.2nm/.
Under dry air atmosphere, cover the organic EL device obtained and seal the device deterioration that causes with the absorption that prevents owing to moisture on it with the acrylic resin adhesive material with cover plate.
Adding under the voltage of 4V with the device of this A1 electrode (negative electrode 4) as negative pole as anodal to this ITO electrode (anode 2) that has that obtains like this, observing luminosity is 104cd/m
2And luminous efficiency is the blue-light-emitting of 4.91m/W.
<comparative example 2 〉
With the mode fabricate devices identical with embodiment 4, difference is to use comparative compound 4-1 shown below to replace exemplary compounds 4, and with embodiment 4 in identical mode this device is estimated.The result does not have to observe luminous from exemplary compounds 4-1.
Although describe the present invention with reference to exemplary, should understand the present invention and be not limited to disclosed exemplary.The scope of following claim should give change and equivalent structure and the function of the most wide in range explanation to comprise that all are such.
The application requires in the right of the Japanese patent application No.2006-310380 of the Japanese patent application No.2006-123784 of submission on April 27th, 2006 and submission on November 16th, 2006, and these documents are introduced as reference is whole at this.
Claims (7)
1. by the 4-aryl fluorene compound of following general formula (I) expression:
Wherein: n represents the integer of 0-10;
When n represented 0, Ar represented the Direct Bonding between fluorenyl and the fluoranthene base;
When n represented the integer of 1-10, Ar represented to replace or the inferior aralkyl of unsubstituted divalent alkyl, replacement or unsubstituted divalence, replacement or unsubstituted divalence arylidene or replacement or unsubstituted divalent heterocycle;
When n represents the integer of 1-10, can be same to each other or different to each other between the Ar;
R
1And R
2Represent replacement or unsubstituted alkyl, replacement or unsubstituted aralkyl, replacement or unsubstituted alkoxyl group, replacement or unsubstituted aryl, replacement or unsubstituted heterocyclic, replacement or unsubstituted amino, cyano group or halogen group separately, and R
1And R
2Can be same to each other or different to each other;
X and y represent the integer of 0-9 separately; With
When x or y represent integer more than 2, R
1Between or R
2Between can be same to each other or different to each other perhaps R
1Between or R
2Between each other bonding to form ring.
2. 4-aryl fluorene compound according to claim 1, wherein, the Ar in the general formula (I) represents to replace or unsubstituted phenylene.
3. 4-aryl fluorene compound according to claim 1, wherein, the n in the general formula (I) represents 0.
4. 4-aryl fluorene compound according to claim 1, wherein, the Ar in the general formula (I) represents to replace or unsubstituted naphthyl.
5. 4-aryl fluorene compound according to claim 1, wherein, the Ar in the general formula (I) represents to replace or unsubstituted anthryl.
6. organic luminescent device, it comprises:
The pair of electrodes that comprises anode and negative electrode; With
Be clamped in the organic compound layer between this counter electrode,
Wherein this organic compound layer contains 4-aryl fluorene compound according to claim 1.
7. organic luminescent device according to claim 6, wherein, this organic compound layer is a luminescent layer.
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2006123784 | 2006-04-27 | ||
| JP123784/2006 | 2006-04-27 | ||
| JP310380/2006 | 2006-11-16 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN101432251A true CN101432251A (en) | 2009-05-13 |
Family
ID=40647020
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CNA2007800152248A Pending CN101432251A (en) | 2006-04-27 | 2007-04-12 | 4-arylfluorene compound and organic light-emitting device using same |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN101432251A (en) |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102898350A (en) * | 2011-07-25 | 2013-01-30 | 海洋王照明科技股份有限公司 | Anthracene derivative organic semiconductor material, preparation method and applications thereof |
| CN103319535A (en) * | 2012-03-19 | 2013-09-25 | 海洋王照明科技股份有限公司 | Organic semiconductor material, preparation method and electroluminescent device |
| CN103319531A (en) * | 2012-03-19 | 2013-09-25 | 海洋王照明科技股份有限公司 | Organic semiconductor material, preparation method and electroluminescent device |
| CN103319532A (en) * | 2012-03-19 | 2013-09-25 | 海洋王照明科技股份有限公司 | Organic semiconductor material, preparation method and electroluminescent device |
| CN101595080B (en) * | 2006-11-15 | 2013-12-18 | 出光兴产株式会社 | Fluoranthene compound, organic electroluminescent element using fluoranthene compound, and solution containing organic electroluminescent material |
| CN103468243A (en) * | 2012-06-06 | 2013-12-25 | 广东阿格蕾雅光电材料有限公司 | Organic electronic materials and organic electroluminescent device |
| CN104342126A (en) * | 2013-11-11 | 2015-02-11 | 北京阿格蕾雅科技发展有限公司 | Organic electroluminescent material and organic electroluminescent device |
| CN105492413A (en) * | 2013-09-06 | 2016-04-13 | 出光兴产株式会社 | Anthracene derivative and organic electroluminescent device using the anthracene derivative |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2004043349A (en) * | 2002-07-11 | 2004-02-12 | Mitsui Chemicals Inc | Hydrocarbon compound, material for organic electroluminescent element and organic electroluminescent element |
| CN1571763A (en) * | 2002-08-27 | 2005-01-26 | 佳能株式会社 | Fluorene compound and organic lumnescent device using the same |
| US20050023697A1 (en) * | 2000-01-18 | 2005-02-03 | Micron Technology, Inc. | Methods for making integrated-circuit wiring from copper, silver, gold, and other metals |
-
2007
- 2007-04-12 CN CNA2007800152248A patent/CN101432251A/en active Pending
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20050023697A1 (en) * | 2000-01-18 | 2005-02-03 | Micron Technology, Inc. | Methods for making integrated-circuit wiring from copper, silver, gold, and other metals |
| JP2004043349A (en) * | 2002-07-11 | 2004-02-12 | Mitsui Chemicals Inc | Hydrocarbon compound, material for organic electroluminescent element and organic electroluminescent element |
| CN1571763A (en) * | 2002-08-27 | 2005-01-26 | 佳能株式会社 | Fluorene compound and organic lumnescent device using the same |
Cited By (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101595080B (en) * | 2006-11-15 | 2013-12-18 | 出光兴产株式会社 | Fluoranthene compound, organic electroluminescent element using fluoranthene compound, and solution containing organic electroluminescent material |
| CN102898350A (en) * | 2011-07-25 | 2013-01-30 | 海洋王照明科技股份有限公司 | Anthracene derivative organic semiconductor material, preparation method and applications thereof |
| CN102898350B (en) * | 2011-07-25 | 2014-12-10 | 海洋王照明科技股份有限公司 | Anthracene derivative organic semiconductor material, preparation method and applications thereof |
| CN103319535A (en) * | 2012-03-19 | 2013-09-25 | 海洋王照明科技股份有限公司 | Organic semiconductor material, preparation method and electroluminescent device |
| CN103319531A (en) * | 2012-03-19 | 2013-09-25 | 海洋王照明科技股份有限公司 | Organic semiconductor material, preparation method and electroluminescent device |
| CN103319532A (en) * | 2012-03-19 | 2013-09-25 | 海洋王照明科技股份有限公司 | Organic semiconductor material, preparation method and electroluminescent device |
| CN103468243A (en) * | 2012-06-06 | 2013-12-25 | 广东阿格蕾雅光电材料有限公司 | Organic electronic materials and organic electroluminescent device |
| CN103468243B (en) * | 2012-06-06 | 2015-08-19 | 广东阿格蕾雅光电材料有限公司 | Organic electronic material and organic electroluminescence device |
| CN105492413A (en) * | 2013-09-06 | 2016-04-13 | 出光兴产株式会社 | Anthracene derivative and organic electroluminescent device using the anthracene derivative |
| CN105492413B (en) * | 2013-09-06 | 2019-05-14 | 出光兴产株式会社 | Anthracene derivative and organic electroluminescent element using the same |
| CN104342126A (en) * | 2013-11-11 | 2015-02-11 | 北京阿格蕾雅科技发展有限公司 | Organic electroluminescent material and organic electroluminescent device |
| WO2015067155A1 (en) * | 2013-11-11 | 2015-05-14 | 北京阿格蕾雅科技发展有限公司 | Organic electroluminescent material and organic electroluminescent device |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR102201097B1 (en) | Novel organic compounds for organic light-emitting diode and organic light-emitting diode including the same | |
| KR101823704B1 (en) | Aminoanthracene derivative and organic electroluminescent element formed using same | |
| JP2023535546A (en) | Heterocyclic compound, organic light-emitting device containing the same, and composition for organic layer of organic light-emitting device | |
| KR102156562B1 (en) | Asymmetric pyrene derivatives comprising aryl amine group and organic light-emitting diode including the same | |
| KR101996651B1 (en) | Fluorene derivatives and organic light-emitting diode including the same | |
| KR101897045B1 (en) | Organic metal compounds and organic light emitting diodes comprising the same | |
| JP7737719B2 (en) | Heterocyclic compound, organic light-emitting device containing the same, composition for organic layer of organic light-emitting device, and method for manufacturing organic light-emitting device | |
| KR20120117675A (en) | Pyrene derivative compounds and organic light-emitting diode including the same | |
| KR20090008411A (en) | 4-arylfluorene compound and organic light emitting device using the same | |
| KR20150093995A (en) | Compound for organic electronic element, organic electronic element using the same, and an electronic device thereof | |
| CN101341115A (en) | 4-aminofluorene compounds and organic light-emitting devices | |
| KR102051910B1 (en) | Deuteriated organometallic complex and organic light-emitting diode including the same | |
| CN109694328B (en) | Triarylamine compound, preparation method thereof and application thereof in organic electroluminescent device | |
| CN101432251A (en) | 4-arylfluorene compound and organic light-emitting device using same | |
| CN109694378B (en) | Polyheteroaromatic compound and organic electroluminescent device using same | |
| KR102298235B1 (en) | Heterocyclic compound, organic light emitting device comprising same, composition for organic layer of organic light emitting device and manufacturing method of organic light emitting device | |
| KR102319692B1 (en) | Heterocyclic compound, organic light emitting device comprising same and composition for organic layer of organic light emitting device | |
| KR102170538B1 (en) | Composition for an organic electronic device and organic electronic device using the same | |
| CN107602582A (en) | Organic compound and organic electroluminescent device using the same | |
| CN106800526A (en) | A kind of electroluminescent organic material and its application with cyclic diketones as core | |
| CN113292488B (en) | Cyano-containing aza-benzene compound, application thereof and organic electroluminescent device containing compound | |
| KR102193837B1 (en) | Asymmetric pyrene derivatives comprising amine group including pyridinyl group and organic light-emitting diode including the same | |
| KR20120112257A (en) | New compounds and organic light-emitting diode including the same | |
| KR20160090242A (en) | Novel antracene derivatives for organic light-emitting diode and organic light-emitting diode including the same | |
| KR20210038500A (en) | Asymmetric pyrene derivatives comprising amine group including pyridinyl group and organic light-emitting diode including the same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C02 | Deemed withdrawal of patent application after publication (patent law 2001) | ||
| WD01 | Invention patent application deemed withdrawn after publication |
Application publication date: 20090513 |