CN101406191B - A fungicidal composition - Google Patents
A fungicidal composition Download PDFInfo
- Publication number
- CN101406191B CN101406191B CN2007101756285A CN200710175628A CN101406191B CN 101406191 B CN101406191 B CN 101406191B CN 2007101756285 A CN2007101756285 A CN 2007101756285A CN 200710175628 A CN200710175628 A CN 200710175628A CN 101406191 B CN101406191 B CN 101406191B
- Authority
- CN
- China
- Prior art keywords
- composition
- component
- active compound
- compounds
- methyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
Landscapes
- Agricultural Chemicals And Associated Chemicals (AREA)
Abstract
Description
技术领域technical field
本发明属于农用杀菌剂领域,具体地涉及一种杀真菌组合物。The invention belongs to the field of agricultural fungicides, in particular to a fungicide composition.
背景技术Background technique
中国发明专利CN1657524A以及PCT申请WO2005080344A1中报道了一种取代唑类化合物,该类化合物对于病原真菌很好的防治效果,对谷物、水稻、果树、蔬菜等作物上的多种植物病害例如对稻瘟病、稻曲病、水稻纹枯病、油菜菌核病、西红柿叶霉病、西红柿灰霉病、苹果轮纹病、苹果树腐烂病、瓜类炭疽病、黄瓜枯萎病、黄瓜霜霉病、黄瓜白粉病、棉花黄萎病、棉花枯萎病、小麦白粉病等真菌病害均具有较好的防治作用。A substituted azole compound is reported in Chinese invention patent CN1657524A and PCT application WO2005080344A1. This type of compound has a good control effect on pathogenic fungi, and it is effective against various plant diseases on crops such as grains, rice, fruit trees, and vegetables, such as rice blast. , rice smut, rice sheath blight, rape sclerotinia, tomato leaf mold, tomato gray mold, apple ring spot, apple tree rot, melon anthracnose, cucumber wilt, cucumber downy mildew, cucumber Fungal diseases such as powdery mildew, cotton verticillium wilt, cotton fusarium wilt, and wheat powdery mildew all have good control effects.
该类化合物为线粒体呼吸抑制剂。由于抑制线粒体的电子传递作用位点非常单一,病菌容易对药剂产生适应性的变异,可能致使药剂的防效降低甚至无效,此类药剂在欧洲已有抗药性的相关报道。实际的农业经验也已经表明,长期重复且专一施用一种活性化合物来防治有害真菌在很多情况下会导致真菌菌株的快速选择性,所述菌株对特定活性化合物发展出天生或适应的抗性,导致不能再使用所述活性化合物来有效防治这些真菌。因此人们对这类问题非常关注。These compounds are inhibitors of mitochondrial respiration. Since the electron transfer site that inhibits mitochondria is very single, the bacteria are prone to adaptive mutations to the drug, which may reduce or even ineffective the control effect of the drug. There have been reports of resistance to such drugs in Europe. Practical agricultural experience has also shown that long-term repeated and exclusive application of an active compound for the control of harmful fungi leads in many cases to a rapid selection of fungal strains which develop an innate or adapted resistance to the specific active compound , so that the active compounds can no longer be used effectively against these fungi. So people pay a lot of attention to this kind of problem.
发明内容Contents of the invention
为了降低抗性真菌菌株选择性的危险性,通常使用不同活性化合物的混合物来防治有害真菌。通过将具有不同作用机理的化合物进行组合,可以更加有效的防治影响作物生长的有害真菌,延长混合物中各组分的使用寿命。本发明旨在提供一种含有不同活性化合物且具有协同增效作用的杀真菌组合物,以达到有效处理抗性和有效防治植物病原真菌的目的。In order to reduce the risk of selection of resistant fungal strains, mixtures of different active compounds are generally used for controlling harmful fungi. By combining compounds with different action mechanisms, it is possible to more effectively control harmful fungi that affect crop growth and prolong the service life of each component in the mixture. The present invention aims to provide a fungicidal composition containing different active compounds and having a synergistic effect, so as to achieve the purpose of effectively treating resistance and effectively controlling plant pathogenic fungi.
本发明的技术方案如下:Technical scheme of the present invention is as follows:
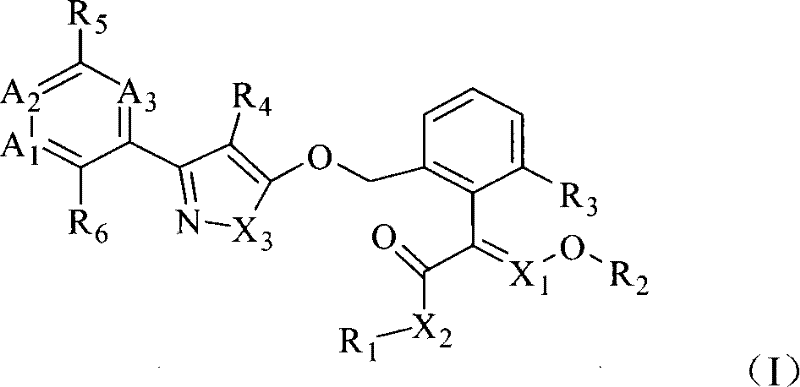
本发明提供了一种杀真菌组合物,包含协同增效的如下化合物作为活性组分:组分A选自如通式(I)所示的取代唑类化合物或其盐:The present invention provides a fungicidal composition comprising the synergistic following compounds as active components: Component A is selected from substituted azole compounds or salts thereof as shown in general formula (I):
式中:In the formula:
X1选自CH或N,X2选自O、S或NR7,X3选自O、S或NR8;X 1 is selected from CH or N, X 2 is selected from O, S or NR 7 , X 3 is selected from O, S or NR 8 ;
A1选自N或CR9,A2选自N或CR10,A3选自N或CR11,其中A1、A2、A3同时选自N的个数小于、等于1;A 1 is selected from N or CR 9 , A 2 is selected from N or CR 10 , A 3 is selected from N or CR 11 , wherein the number of A 1 , A 2 , and A 3 selected from N at the same time is less than or equal to 1;
R1、R2可相同或不同,分别选自氢、C1-C12烷基或卤代C1-C12烷基;R 1 and R 2 may be the same or different, and are respectively selected from hydrogen, C 1 -C 12 alkyl or halogenated C 1 -C 12 alkyl;
R3选自氢、卤原子、C1-C12烷基、卤代C1-C12烷基或C1-C12烷氧基;R 3 is selected from hydrogen, halogen atom, C 1 -C 12 alkyl, halogenated C 1 -C 12 alkyl or C 1 -C 12 alkoxy;
R7选自氢或C1-C12烷基;R 7 is selected from hydrogen or C 1 -C 12 alkyl;
R8选自氢、C1-C12烷基、卤代C1-C12烷基、C1-C12烷氧基羰基或C1-C12烷氧基羰基C1-C12烷基;R 8 is selected from hydrogen, C 1 -C 12 alkyl, halogenated C 1 -C 12 alkyl, C 1 -C 12 alkoxycarbonyl or C 1 -C 12 alkoxycarbonyl C 1 -C 12 alkyl ;
R4、R5、R6、R9、R10、R11可相同或不同,分别选自氢、卤原子、硝基、氰基、CONH2、CH2CONH2、CH2CN、C1-C12烷基、卤代C1-C12烷基、C1-C12烷氧基、卤代C1-C12烷氧基、C1-C12烷硫基、C1-C12烷磺酰基、C1-C12烷基羰基、C1-C12烷氧基C1-C12烷基、C1-C12烷氧基羰基、C1-C12烷氧基羰基C1-C12烷基、C1-C12卤代烷氧基C1-C12烷基、可任意取代的下列基团:胺基C1-C12烷基、芳基、杂芳基、芳氧基、芳基C1-C12烷基、芳C1-C12烷基氧基、杂芳基C1-C12烷基或杂芳基C1-C12烷氧基;及其立体异构体。R 4 , R 5 , R 6 , R 9 , R 10 , and R 11 may be the same or different, and are respectively selected from hydrogen, halogen, nitro, cyano, CONH 2 , CH 2 CONH 2 , CH 2 CN, and C 1 -C 12 alkyl, halogenated C 1 -C 12 alkyl, C 1 -C 12 alkoxy, halogenated C 1 -C 12 alkoxy, C 1 -C 12 alkylthio, C 1 -C 12 Alkylsulfonyl, C 1 -C 12 Alkylcarbonyl, C 1 -C 12 Alkoxy C 1 -C 12 Alkyl, C 1 -C 12 Alkoxycarbonyl, C 1 -C 12 Alkoxycarbonyl C 1 -C 12 alkyl, C 1 -C 12 haloalkoxy C 1 -C 12 alkyl, the following groups that can be optionally substituted: amino C 1 -C 12 alkyl, aryl, heteroaryl, aryloxy , aryl C 1 -C 12 alkyl, aryl C 1 -C 12 alkyloxy, heteroaryl C 1 -C 12 alkyl or heteroaryl C 1 -C 12 alkoxy; and stereoisomerism thereof body.
组分B选自具有杀真菌活性的人工合成的或者来源于天然产物的如下所述六类化合物、以及它们的农业上可接受的异构体或其盐:Component B is selected from the following six types of compounds that are artificially synthesized or derived from natural products with fungicidal activity, and their agriculturally acceptable isomers or salts thereof:
三唑类化合物(B1),例如氟环唑、三唑酮、恶醚唑、烯唑醇、腈菌唑、戊唑醇、丙环唑、丙硫菌唑、氧环唑、糠菌唑、环丙唑醇、苯醚甲环唑、高效烯唑醇、腈苯唑、氟硅唑、粉唑醇、己唑醇、亚胺唑、种菌唑、叶菌唑、戊菌唑、硅氟唑、四氟醚唑、三唑醇、灭菌唑、联苯三唑醇、三环唑。Triazole compounds (B 1 ), such as econazole, triadimefon, oxaconazole, diniconazole, myclobutanil, tebuconazole, propiconazole, prothioconazole, oxyconazole, furconazole , ciproconazole, difenoconazole, diniconazole, nibconazole, flusilazole, powderconazole, hexaconazole, imidazole, speciesconazole, metconazole, penconazole, silicon Fluconazole, Tetrafluconazole, Triaconazole, Fenconazole, Bifentriazole, Tricyclazole.
硫代氨基甲酸盐类化合物(B2),例如代森锰锌、代森锰、代森锌、丙森锌、代森铵、福美双、福美锌。Thiocarbamate compounds (B 2 ), such as mancozeb, maneb, zinc, propineb, thiram, thiram, and thiram.
甲氧基丙烯酸酯类化合物(B3),例如嘧菌酯、氟嘧菌酯、肟菌酯、啶氧菌酯、唑菌胺酯、醚菌胺、醚菌酯、苯氧菌胺、肟醚菌胺、烯肟菌酯(SYP-Z071)、烯肟菌胺(SYP-1620)、ZJ0712、UBF-307、KZ165。Methoxyacrylate compounds (B 3 ), such as azoxystrobin, fluoxastrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, kysstrobin, kysstrobin, fenoxystrobin, oxime Kysoxastrobin, Enoxastrobin (SYP-Z071), Enoxastrobin (SYP-1620), ZJ0712, UBF-307, KZ165.
氨基甲酸酯类化合物(B4),例如乙霉威、异丙菌胺、苯噻菌胺、valiphenal(IR5885)、霜霉威、磺菌威、pyribencarb、meptyldinocap。Carbamate compounds (B 4 ), such as Ditofencarb, Iprocarbam, Benthiocarb, valiphenal (IR5885), propamocarb, sulfacarb, pyribencarb, meptyldinocap.
抗生素类化合物(B5),例如灭瘟素、春雷霉素、myxothiazol、多抗霉素、多氧霉素、pseudomycin、PSF-D、有效霉素、井冈霉素、链霉素。Antibiotic compounds (B 5 ), such as blasticidin, kasugamycin, myxothiazol, polyoxin, polyoxin, pseudomycin, PSF-D, validamycin, jinggangmycin, streptomycin.
酰胺类化合物(B6),例如氟吗啉、烯酰吗啉、噻氟菌胺、环丙酰菌胺、环酰菌胺、氰菌胺、isotianil、硅噻菌胺、呋吡菌胺、苯酰菌胺、甲霜灵、呋霜灵、苯霜灵、双氯氰菌胺、磺菌胺。Amide compounds (B 6 ), such as flumorph, dimethomorph, thiafluzamide, cyproamid, fenhexamid, fenhexamid, isotianil, silthiocarb, furamet, Benalaxyl, metalaxyl, furalaxyl, benalaxyl, diclofenamide, sulfanil.
A、B两组分之间的重量比为1∶200~200∶1。The weight ratio between the two components A and B is 1:200˜200:1.
本发明的杀真菌组合物中较优选的技术方案为:The preferred technical scheme in the fungicidal composition of the present invention is:
组分A选自通式(I)所示的取代唑类化合物或其盐:Component A is selected from substituted azole compounds represented by general formula (I) or salts thereof:
式中:In the formula:
X1选自CH,X2选自O,X3选自NCH3;X 1 is selected from CH, X 2 is selected from O, X 3 is selected from NCH 3 ;
A1、A3分别选自CH,A2选自C-Cl;A 1 and A 3 are respectively selected from CH, and A 2 is selected from C-Cl;
R1、R2分别选自CH3,R3选自氢,R4、R5、R6分别选自氢;结构如下:R 1 and R 2 are respectively selected from CH 3 , R 3 is selected from hydrogen, R 4 , R 5 and R 6 are respectively selected from hydrogen; the structure is as follows:
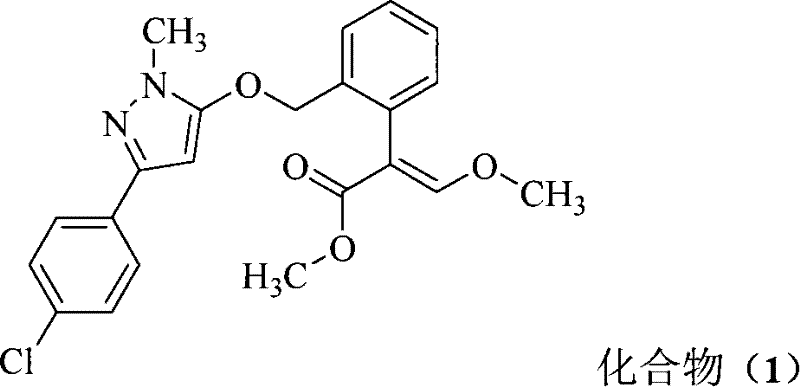
化合物(1)的中文通用名为唑菌酯,化学名称为:The Chinese common name of compound (1) is pyraclostrobin, and its chemical name is:
(E)-2-(2-((3-(4-氯苯基)-1-甲基-1H-吡唑-5-基氧基)甲基)苯基)-3-甲氧基丙烯酸甲酯(E)-2-(2-((3-(4-chlorophenyl)-1-methyl-1H-pyrazol-5-yloxy)methyl)phenyl)-3-methoxyacrylic acid methyl ester
[(E)-methyl 2-(2-((3-(4-chlorophenyl)-1-methyl-1H-pyrazol-5-yloxy)methyl)phenyl)-3-methoxyacrylate]。[(E)-methyl 2-(2-((3-(4-chlorophenyl)-1-methyl-1H-pyrazol-5-yloxy)methyl)phenyl)-3-methoxyacrylate].
组分B的较优选化合物选自:More preferred compounds of component B are selected from:
三唑类化合物(B1)包括:(2)氟环唑(epoxiconazole)、(3)三唑酮(triadimefon)、(4)恶醚唑(difenoconazole)、(5)烯唑醇(diniconazole)、(6)腈菌唑(myclobutanil)、(7)丙环唑(propiconazole)或(8)戊唑醇(tebuconazole);Triazole compounds (B 1 ) include: (2) epoxiconazole, (3) triadimefon, (4) difenoconazole, (5) diniconazole, (6) myclobutanil, (7) propiconazole or (8) tebuconazole;
硫代氨基甲酸盐类化合物(B2)包括:(9)代森锰锌(mancozeb)、(10)代森锰(maneb)、(11)代森锌(zineb)、(12)丙森锌(propineb)、(13)代森铵(amobam)、(14)福美双(thiram)或(15)福美锌(ziram);Thiocarbamate compounds (B 2 ) include: (9) Mancozeb (mancozeb), (10) Maneb (maneb), (11) Zinc (zineb), (12) Propineb (propineb), (13) amobam, (14) thiram or (15) ziram;
甲氧基丙烯酸酯类化合物(B3)包括:(16)嘧菌酯(azoxystrobin)、(17)肟菌酯(trifloxystrobin)、(18)啶氧菌酯(picoxystrobin)、(19)唑菌胺酯(pyraclostrobin)或(20)醚菌胺(dimoxystrobin);Methoxyacrylate compounds (B 3 ) include: (16) azoxystrobin, (17) trifloxystrobin, (18) picoxystrobin, (19) pyraclostrobin pyraclostrobin or (20) dimoxystrobin;
氨基甲酸酯类化合物(B4)包括:(21)乙霉威(diethofencarb)、(22)异丙菌胺(iprovalicarb)、(23)苯噻菌胺(benthiavalicarb-isopropyl)、(24)磺菌威(methasulfocarb)或(25)霜霉威(propamocarb);Carbamate compounds (B 4 ) include: (21) diethofencarb, (22) iprovalicarb, (23) benthiavalicarb-isopropyl, (24) sulfonium Granville (methasulfocarb) or (25) Propamocarb (propamocarb);
抗生素类化合物(B5)包括:(26)灭瘟素(blasticidin-S)、(27)春雷霉素(kasugamycin)、(28)多抗霉素(polyoxins)或(29)多氧霉素(polyoxorim)。Antibiotic compounds (B 5 ) include: (26) blasticidin-S, (27) kasugamycin, (28) polyoxins or (29) polyoxin ( polyoxorim).
酰胺类化合物(B6)包括:(30)氟吗啉(flumorph)、(31)烯酰吗啉(Dimethomorph)。Amide compounds (B 6 ) include: (30) flumorph, (31) dimethomorph.
上述B组分化合物是多种作物病害的常用防治药剂,在长期的使用过程中表现出了良好的防治效果,其详细叙述见“The Pesticide Manual”(12th ed UK:BCPC,2000)、“The PesticideManual”(13th ed UK:BCPC,2003)。The above-mentioned component B compounds are commonly used control agents for various crop diseases, and have shown good control effects during long-term use. For details, see "The Pesticide Manual" (12th ed UK: BCPC, 2000), "The Pesticide Manual" Pesticide Manual" (13th ed UK: BCPC, 2003).
组合物中A、B两组分较优选的重量比为1∶100~100∶1;进一步优选为1∶50~50∶1。The preferred weight ratio of A and B components in the composition is 1:100-100:1; more preferably 1:50-50:1.
更进一步优选的技术方案为:A further preferred technical solution is:
组分A选自(E)-2-(2-((3-(4-氯苯基)-1-甲基-1H-吡唑-5-基氧基)甲基)苯基)-3-甲氧基丙烯酸甲酯;组分B选自氟环唑、三唑酮、恶醚唑、烯唑醇、腈菌唑、丙环唑或戊唑醇;组合物中A、B两组分之间的重量比为1∶40~40∶1。Component A is selected from (E)-2-(2-((3-(4-chlorophenyl)-1-methyl-1H-pyrazol-5-yloxy)methyl)phenyl)-3 - Methyl methoxyacrylate; component B is selected from epoconazole, triadimefon, oxaconazole, diniconazole, myclobutanil, propiconazole or tebuconazole; two components A and B in the composition The weight ratio between them is 1:40~40:1.
或者,组分A选自(E)-2-(2-((3-(4-氯苯基)-1-甲基-1H-吡唑-5-基氧基)甲基)苯基)-3-甲氧基丙烯酸甲酯;组分B选自代森锰锌、代森锰、代森锌、丙森锌、代森铵、福美双或福美锌;组合物中A、B两组分之间的重量比为1∶20~20∶1。Alternatively, component A is selected from (E)-2-(2-((3-(4-chlorophenyl)-1-methyl-1H-pyrazol-5-yloxy)methyl)phenyl) - Methyl 3-methoxyacrylate; Component B is selected from mancozeb, maneb, zinc zinc, propineb, zinc thiram, thiram or zinc thiram; two groups of A and B in the composition The weight ratio between the parts is 1:20~20:1.
或者,组分A选自(E)-2-(2-((3-(4-氯苯基)-1-甲基-1H-吡唑-5-基氧基)甲基)苯基)-3-甲氧基丙烯酸甲酯;组分B选自嘧菌酯、肟菌酯、啶氧菌酯、唑菌胺酯或醚菌胺;组合物中A、B两组分之间的重量比为1∶20~20∶1。Alternatively, component A is selected from (E)-2-(2-((3-(4-chlorophenyl)-1-methyl-1H-pyrazol-5-yloxy)methyl)phenyl) - Methyl 3-methoxyacrylate; component B is selected from azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin or kyrestrobin; the weight between the two components of A and B in the composition The ratio is 1:20 to 20:1.
或者,组分A选自(E)-2-(2-((3-(4-氯苯基)-1-甲基-1H-吡唑-5-基氧基)甲基)苯基)-3-甲氧基丙烯酸甲酯;组分B选自乙霉威、异丙菌胺、苯噻菌胺、磺菌威或霜霉威;组合物中A、B两组分之间的重量比为1∶20~20∶1。Alternatively, component A is selected from (E)-2-(2-((3-(4-chlorophenyl)-1-methyl-1H-pyrazol-5-yloxy)methyl)phenyl) - Methyl 3-methoxyacrylate; Component B is selected from Ditofencarb, Iprocarbam, Benthiocarb, Sulfurcarb or Propamocarb; the weight between A and B in the composition The ratio is 1:20 to 20:1.
或者,组分A选自(E)-2-(2-((3-(4-氯苯基)-1-甲基-1H-吡唑-5-基氧基)甲基)苯基)-3-甲氧基丙烯酸甲酯;组分B选自灭瘟素、春雷霉素、多抗霉素或多氧霉素;组合物中A、B两组分之间的重量比为1∶20~20∶1。Alternatively, component A is selected from (E)-2-(2-((3-(4-chlorophenyl)-1-methyl-1H-pyrazol-5-yloxy)methyl)phenyl) -Methyl 3-methoxyacrylate; Component B is selected from blasticidin, kasugamycin, polyoxin or polyoxin; the weight ratio between A and B in the composition is 1: 20~20:1.
或者,组分A选自(E)-2-(2-((3-(4-氯苯基)-1-甲基-1H-吡唑-5-基氧基)甲基)苯基)-3-甲氧基丙烯酸甲酯;组分B选自氟吗啉或烯酰吗啉;组合物中A、B两组分之间的重量比为1∶20~20∶1。Alternatively, component A is selected from (E)-2-(2-((3-(4-chlorophenyl)-1-methyl-1H-pyrazol-5-yloxy)methyl)phenyl) - Methyl 3-methoxyacrylate; component B is selected from flumorph or dimethomorph; the weight ratio between A and B in the composition is 1:20 to 20:1.
现已发现,本发明的杀真菌组合物具有非常明显的协同增效作用,对农作物病害的防治效果有显著提高,同时能扩大杀菌谱、延缓病原物抗药性的发生。因此本发明的技术方案包括了防治植物病害方面的应用。It has been found that the fungicidal composition of the present invention has a very obvious synergistic effect, significantly improves the control effect on crop diseases, and can expand the bactericidal spectrum and delay the occurrence of drug resistance of pathogens. Therefore, the technical solution of the present invention includes the application in the prevention and treatment of plant diseases.
本发明组合物适合于防治多种有害真菌,尤其是禾谷类、蔬菜、果树、观赏植物、草坪及热带作物的多种病害,以及多种仓储病害。The composition of the invention is suitable for preventing and controlling various harmful fungi, especially various diseases of cereals, vegetables, fruit trees, ornamental plants, lawns and tropical crops, and various storage diseases.
当制备该组合物时,优选使用纯的活性化合物(1)和至少一种B组分化合物,可以向其中加入其它对有害病原物或其它害虫有效的活性物质,或具有除草作用以及生长调节作用的活性化合物或肥料。无论以任何形式使用该组合物,即化合物(1)和至少一种B组分化合物单独或联合施用或化合物(1)和至少一种B组分化合物的混合物的施用,均呈现显著的防治病害范围增大和对某些病害的防效提高,包括对子囊菌、担子菌、半知菌等引起的多种植物病害。组合物对防治各种作物如谷物、水稻、蔬菜、果树、花卉的真菌病害,以及用于拌种保护作物尤为重要。特别适合防治下列植物真菌病害:黄瓜白粉病、黄瓜霜霉病、黄瓜灰霉病、黄瓜黑星病、香瓜白粉病、大豆锈病、西红柿灰霉病、西红柿白粉病、西瓜炭疽病、水稻稻瘟病、水稻稻曲病、小麦白粉病、小麦锈病、梨黑星病、花生叶斑病、草莓白粉病、香蕉叶斑病等。When preparing the composition, it is preferred to use the pure active compound (1) and at least one component B compound, to which other active substances effective against harmful pathogens or other pests, or having a herbicidal and growth-regulating action can be added active compounds or fertilizers. Regardless of the use of the composition in any form, i.e. the application of compound (1) and at least one component B compound alone or in combination or the application of a mixture of compound (1) and at least one component B compound, significant disease control is exhibited Increased range and improved control effect on certain diseases, including various plant diseases caused by Ascomycetes, Basidiomycetes, and Deuteromycetes. The composition is particularly important for preventing and controlling fungal diseases of various crops such as grains, rice, vegetables, fruit trees and flowers, and for protecting crops by seed dressing. Especially suitable for the control of the following plant fungal diseases: cucumber powdery mildew, cucumber downy mildew, cucumber gray mold, cucumber scab, melon powdery mildew, soybean rust, tomato gray mold, tomato powdery mildew, watermelon anthracnose, rice blast , Rice smut, wheat powdery mildew, wheat rust, pear scab, peanut leaf spot, strawberry powdery mildew, banana leaf spot, etc.
根据农作物病害的发生程度,本发明组合物的使用浓度在农作物种植区域为1~1500g/hm2,优选15~400g/hm2。这里化合物(1)的使用浓度为0.02~400g/hm2,优选5~250g/hm2。相应的,化合物(B1)~(B5)的使用浓度为0.02~1000g/hm2,优选15~600g/hm2。According to the occurrence degree of crop diseases, the use concentration of the composition of the present invention is 1-1500 g/hm 2 , preferably 15-400 g/hm 2 in the crop planting area. The concentration of compound (1) used here is 0.02-400 g/hm 2 , preferably 5-250 g/hm 2 . Correspondingly, the use concentration of compounds (B 1 )-(B 5 ) is 0.02-1000 g/hm 2 , preferably 15-600 g/hm 2 .
处理种子时,组合物的使用浓度通常为1~1000g/kg,优选1~800g/kg。When treating seeds, the use concentration of the composition is usually 1-1000 g/kg, preferably 1-800 g/kg.
在防治植物病害的过程中,本发明的组合物的施用时期,可以是植物播种之前或之后,也可以在植物出苗之前或之后对种子、植株或土壤喷雾或散粉等方式而进行。In the process of preventing and controlling plant diseases, the application period of the composition of the present invention can be before or after plant sowing, and can also be carried out by spraying or loose powdering on seeds, plants or soil before or after plant emergence.
可将本发明的杀真菌组合物配制为可直接喷雾溶液、粉末、悬浮液、高度浓缩的水性、油性或其它悬浮液、分散体、乳液、油分散体、糊、粉剂、撒播用混合物或颗粒,且可通过喷雾、雾化、撒粉、撒播、或倾倒等方式施用。施用形式取决于特定目的;在各种情况下,应确保本发明组合物精细且均匀分布。The fungicidal compositions of the present invention can be formulated as direct sprayable solutions, powders, suspensions, highly concentrated aqueous, oily or other suspensions, dispersions, emulsions, oil dispersions, pastes, powders, mixtures or granules for broadcasting , and can be applied by spraying, atomizing, dusting, spreading, or pouring. The application form depends on the particular purpose; in each case, it should ensure a fine and even distribution of the composition according to the invention.
将本发明的杀真菌组合物以合适的剂型、使用适合的剂量防治有害真菌、其栖息地或要防治它们所侵染的植物体、种子、土壤、区域、材料或空间。化合物的施用可在有害真菌侵染之前或之后进行,重点应在侵染之前或侵染初期进行施用。The fungicidal composition of the present invention is used in suitable formulations and in suitable doses to control harmful fungi, their habitats or the plants, seeds, soil, areas, materials or spaces they infeste. The application of the compound can be carried out before or after the harmful fungus infestation, and the emphasis should be on the application before the infestation or the initial stage of the infestation.
可以用已知方法配制本发明杀真菌组合物的制剂。本发明的组合物中至少包含两种载体,载体可以是固体或液体,通常用于配制杀菌剂组合物的任何载体均能使用。组合物中至少一种载体是表面活性剂,例如可以是乳化剂、分散剂或湿润剂等。表面活性剂均为市售,其选择是本领域技术人员所已知的,例如,十二烷基硫酸钠、仲烷基硫酸钠、十二烷基苯磺酸钠、聚氧乙烯脂肪酸脂、聚氧乙烯脂肪醇醚或聚氧乙烯脂肪氨等。The formulation of the fungicidal composition of the present invention can be formulated by known methods. The composition of the present invention contains at least two kinds of carriers, and the carriers can be solid or liquid, and any carrier usually used for preparing fungicide compositions can be used. At least one carrier in the composition is a surfactant, such as an emulsifier, dispersant or wetting agent. Surfactant is all commercially available, and its selection is known to those skilled in the art, for example, sodium lauryl sulfate, secondary alkyl sodium sulfate, sodium dodecylbenzenesulfonate, polyoxyethylene fatty acid ester, Polyoxyethylene fatty alcohol ether or polyoxyethylene fatty ammonia, etc.
本发明组合物可以配制成乳油、悬浮剂、(水)乳剂、可湿性粉剂、(水分散)粒剂等,这些制剂可由通用的方法制备。组合物中,A、B组分的有效成分含量之和即活性组分的重量含量为0.1~95%,优选0.1~80%。The composition of the present invention can be formulated into emulsifiable concentrates, suspension concentrates, (water) emulsions, wettable powders, (water dispersible) granules, etc., and these formulations can be prepared by common methods. In the composition, the sum of the contents of active ingredients in components A and B, that is, the weight content of the active ingredients, is 0.1-95%, preferably 0.1-80%.
本发明的组合物为活性物质所配制成的几种常用剂型的实例(具体应用并非仅限于此)列举如下:Composition of the present invention is the example (concrete application is not limited to this) of several commonly used dosage forms that active substance is formulated as follows:
一、用水稀释的产品1. Products diluted with water
1.10%水乳剂(EW)1.10% emulsion in water (EW)
活性化合物10%,聚乙烯醇0.8%,烷基芳基聚氧乙烯聚氧丙烯醚8.5%,农乳2201 16%,二甲基甲酰胺11%,乙二醇5%,水补足至100%。Active compound 10%, polyvinyl alcohol 0.8%, alkylaryl polyoxyethylene polyoxypropylene ether 8.5%, Nongru 2201 16%, dimethylformamide 11%, ethylene glycol 5%, water to 100% .
2.20%乳油(EC)2.20% emulsifiable concentrate (EC)
活性化合物20%,农乳500# 10%,农乳OX-635 5%,甲醇10%,二甲苯补足至100%。Active compound 20%, Nongru 500# 10%, Nongru OX-635 5%, methanol 10%, xylene supplemented to 100%.
3.40%悬浮剂(SC)3.40% suspension concentrate (SC)
活性化合物40%,木质素磺酸钙5%,白碳黑0.3%,乙二醇4%,消泡剂少许,水补足至100%。40% active compound, 5% calcium lignosulfonate, 0.3% white carbon black, 4% ethylene glycol, a little defoamer, and water to make up to 100%.
4.50%水分散粒剂(WG)4.50% water dispersible granules (WG)
活性化合物50%,甲基萘磺酸钠甲醛缩合物12%,环氧聚醚5%,可溶性淀粉15%,石膏补足至100%。Active compound 50%, sodium methylnaphthalene sulfonate formaldehyde condensate 12%, epoxy polyether 5%, soluble starch 15%, gypsum supplemented to 100%.
5.70%可湿性粉剂(WP)5.70% wettable powder (WP)
活性化合物70%,十二烷基硫酸钠1.5%,羧甲基纤维素1%,木质素磺酸钠10%,轻质碳酸钙补足至100%。Active compound 70%, sodium lauryl sulfate 1.5%, carboxymethylcellulose 1%, sodium lignosulfonate 10%, light calcium carbonate make up to 100%.
二、不经稀释即可使用的产品2. Products that can be used without dilution
6.5%粉剂(DP)6.5% powder (DP)
将5份重活性化合物细碎研磨并与95份重的细碎高岭土充分混合得到粉剂。A powder is obtained by grinding 5 parts by weight of the active compound finely and mixing intimately with 95 parts by weight of finely divided kaolin.
7.0.5%颗粒剂(GR)7.0.5% Granules (GR)
将0.5份重活性化合物细碎研磨并结合99.5份重的载体,现行方法是挤出、喷雾干燥或流化床,得到不经稀释即可使用的颗粒。0.5 parts by weight of active compound are finely ground and combined with 99.5 parts by weight of carrier, conventionally by extrusion, spray drying or fluidized bed, to obtain granules which are ready to use without dilution.
本发明描述的产物以成品制剂形式提供,即组合物中各物质已经混合。同时,组合物的成分也可以单独制剂的形式提供,使用前在桶(罐)中直接混合。本发明的浓缩物通常与水混合得到所需活性物质的浓度,借助喷雾、雾化、撒粉、撒播或浇注来使用。使用形式完全取决于所要达到的目的,在各种情况下确保本发明活性化合物的最佳可能分布。The products described in the present invention are provided in the form of finished formulations, ie the substances of the composition have been mixed. At the same time, the ingredients of the composition can also be provided in the form of separate preparations, which are directly mixed in barrels (cans) before use. The concentrates according to the invention are usually mixed with water to obtain the desired concentration of active substance and applied by spraying, atomizing, dusting, broadcasting or casting. The use forms depend entirely on the purposes to be achieved, ensuring in each case the best possible distribution of the active compounds according to the invention.
含水使用形式可以通过加入水由乳液浓缩物、糊或可湿性粉末(可喷雾粉末、油分散体)制备。为制备乳液、糊或油分散体,可借助湿润剂、增粘剂、分散剂或乳化剂将该物质直接或溶于油或溶剂中后在水中均化。或者,可以制备适于用水稀释且由活性物质、湿润剂、增粘剂、分散剂或乳化剂、溶剂或油组成的浓缩物。Aqueous use forms can be prepared from emulsion concentrates, pastes or wettable powders (sprayable powders, oil dispersions) by adding water. To prepare emulsions, pastes or oil dispersions, the substances are homogenized in water as such or dissolved in an oil or solvent with the aid of wetting agents, tackifiers, dispersants or emulsifiers. Alternatively, concentrates suitable for dilution with water and consisting of active substance, wetting agent, tackifier, dispersant or emulsifier, solvent or oil may be prepared.
各种类型的油、湿润剂、辅助剂、除草剂、杀真菌剂、其它农药或杀菌剂都可加入活性化合物中,若合适,恰在紧邻使用前加入(桶混合)。这些试剂通常与本发明混合物以1∶30~30∶1的重量比混合。Oils of various types, wetting agents, adjuvants, herbicides, fungicides, other pesticides or bactericides can be added to the active compounds, if appropriate, just before use (tank mix). These agents are usually mixed with the mixture of the invention in a weight ratio of 1:30 to 30:1.
具体实施方式Detailed ways
本发明的杀真菌组合物对有害真菌的协同增效作用可通过下列实例说明,但本发明绝非仅限于此。The synergistic effect of the fungicidal composition of the present invention on harmful fungi can be illustrated by the following examples, but the present invention is by no means limited thereto.
将单独或联合的活性化合物制备成含有0.25%(重量)活性化合物的丙酮或DMSO储液。将1%(重量)的乳化剂UniperolEL(BASF公司产品)加入该储液中,并用水将该活性化合物或混合物稀释至以下实例中所需浓度。Active compounds, individually or in combination, were prepared as stock solutions in acetone or DMSO containing 0.25% by weight active compound. 1% (weight) of emulsifier Uniperol EL (product of BASF Corporation) was added to the stock solution, and the active compound or mixture was diluted with water to the desired concentration in the following examples.
通过测定叶面积侵染百分数进行评价。将这些百分数转化成效力。使用Abbot公式按照如下公式计算效力(W):Evaluation was carried out by determining the percent infection of the leaf area. These percentages were converted to efficacy. Potency (W) was calculated using Abbot's formula as follows:
W=(1-α/β)×100W=(1-α/β)×100
α处理植物的真菌侵染百分数;The percentage of fungal infestation of α-treated plants;
β未处理(空白对照)植物的真菌侵染百分数。Percent fungal infection of β-untreated (blank) plants.
效力为“0”表示处理植物的侵染水平与未处理对照植物的侵染水平相同;效力为“100”表示处理植物未受侵染。An efficacy of "0" indicates that the treated plants have the same level of infection as the untreated control plants; an efficacy of "100" indicates that the treated plants are not infected.
活性化合物混合物的预期效力使用Colby公式[R.S.Colby,杂草(Weeds)15,20-22(1967)]确定并与观察到的效力比较。The expected potency of a mixture of active compounds is determined using the Colby formula [R.S. Colby, Weeds 15, 20-22 (1967)] and compared to the observed potency.
Colby公式:E=x+y-xy/100Colby's formula: E=x+y-xy/100
E:使用浓度分别为a和b的活性化合物A和B的混合物时的预期效力,以未处理对照的%表示。E: Expected efficacy in % of untreated control when using a mixture of active compounds A and B at concentrations a and b respectively.
x使用浓度为a的活性化合物A时的效力,以未处理对照的%表示;x Efficacy at concentration a of active compound A expressed as % of untreated control;
y使用浓度为b的活性化合物B时的效力,以未处理对照的%表示。y Efficacy in % of untreated control when active compound B was used at concentration b.
下列实施例中,所使用的作物幼苗均以盆栽的方式在温室中种植培养。待幼苗生长至三叶期,挑选生长整齐一致的健康幼苗用于生测试验。各表中所列A、B两组分活性化合物的混合比为重量比。In the following examples, the crop seedlings used were all planted and cultivated in a greenhouse in a potted manner. When the seedlings grow to the three-leaf stage, healthy seedlings that grow neatly and uniformly are selected for the bioassay test. The mixing ratios of A and B two-component active compounds listed in each table are weight ratios.
应用实施例1 防治小麦白粉病试验Application Example 1 Test of Controlling Wheat Powdery Mildew
将栽培品种为“辽春10号”的盆栽小麦幼苗叶子用活性化合物浓度如下所述的含水悬浮液喷雾至滴流点。第二天,用白粉病菌(Blumeria graminis)的孢子含水悬浮液接种植物。然后将测试植物置于22~24℃和相对大气湿度为75%气候室中调节7天。然后目测测定叶子上病菌侵染的发展程度。Leaves of potted wheat seedlings of the cultivar "Liaochun No. 10" are sprayed to the trickle point with an aqueous suspension at the concentration of active compound indicated below. The next day, the plants are inoculated with an aqueous spore suspension of the powdery mildew fungus ( Blumeria graminis ). The test plants are then conditioned for 7 days in a climate chamber at 22-24° C. and a relative atmospheric humidity of 75%. The degree of development of the fungal infection on the leaves was then determined visually.
表1单独的活性化合物Table 1 Individual Active Compounds
表2本发明组合物Table 2 Compositions of the present invention
“*”:使用Colby公式计算的效力。" * ": Potency calculated using Colby's formula.
应用实施例2 防治黄瓜白粉病试验Application Example 2 Control of Cucumber Powdery Mildew Test
将栽培品种为“山东密刺”的盆栽黄瓜幼苗叶子用活性化合物浓度如下所述的含水悬浮液喷雾至滴流点。第二天,用白粉病菌(Erysiphe cucurbitacearum)的孢子含水悬浮液接种植物。然后将测试植物置于22~24℃和相对大气湿度为75%气候室中调节7天。然后目测测定叶子上病菌侵染的发展程度。Leaves of potted cucumber seedlings of the cultivar "Shandong Mici" are sprayed to the trickle point with an aqueous suspension having the concentration of active compound indicated below. The next day, plants are inoculated with an aqueous spore suspension of Erysiphe cucurbitacearum . The test plants are then conditioned for 7 days in a climate chamber at 22-24° C. and a relative atmospheric humidity of 75%. The degree of development of the fungal infection on the leaves was then determined visually.
表3单独的活性化合物Table 3 Individual Active Compounds
表4本发明组合物Table 4 Compositions of the present invention
“*”:使用Colby公式计算的效力。" * ": Potency calculated using Colby's formula.
应用实施例3 防治小麦白粉病试验Application Example 3 Test of Controlling Wheat Powdery Mildew
将栽培品种为“辽春10号”的盆栽小麦幼苗叶子用活性化合物浓度如下所述的含水悬浮液喷雾至滴流点。第二天,用白粉病菌(Blumeria graminis)的孢子含水悬浮液接种植物。然后将测试植物置于22~24℃和相对大气湿度为75~80%气候室中调节8天。然后目测测定叶子上病菌侵染的发展程度。Leaves of potted wheat seedlings of the cultivar "Liaochun No. 10" are sprayed to the trickle point with an aqueous suspension at the concentration of active compound indicated below. The next day, the plants are inoculated with an aqueous spore suspension of the powdery mildew fungus ( Blumeria graminis ). The test plants are then conditioned for 8 days in a climate chamber at 22-24° C. and a relative atmospheric humidity of 75-80%. The degree of development of the fungal infection on the leaves was then determined visually.
表5单独的活性化合物Table 5 Individual Active Compounds
表6本发明组合物Table 6 Compositions of the present invention
“*”:使用Colby公式计算的效力。" * ": Potency calculated using Colby's formula.
应用实施例4 防治黄瓜霜霉病试验Application Example 4 Control of Cucumber Downy Mildew Test
将栽培品种为“山东密刺”的盆栽黄瓜幼苗叶子用活性化合物浓度如下所述的含水悬浮液喷雾至滴流点。第二天,用霜霉病菌(Pseudoperonospora cubensis)的孢子含水悬浮液接种植物。然后将测试植物置于22~24℃和相对大气湿度为90%气候室中调节7天。然后目测测定叶子上病菌侵染的发展程度。Leaves of potted cucumber seedlings of the cultivar "Shandong Mici" are sprayed to the trickle point with an aqueous suspension having the concentration of active compound indicated below. The next day, the plants are inoculated with an aqueous spore suspension of Pseudoperonospora cubensis . The test plants are then conditioned for 7 days in a climate chamber at 22-24° C. and a relative atmospheric humidity of 90%. The degree of development of the fungal infection on the leaves was then determined visually.
表7单独的活性化合物Table 7 Individual Active Compounds
表8本发明的组合物Table 8 Compositions of the present invention
“*”:使用Colby公式计算的效力。" * ": Potency calculated using Colby's formula.
应用实施例5 防治小麦赤霉病试验Application Example 5 Test of Controlling Wheat Scab
将栽培品种为“辽春十号”的盆栽小麦麦穗用活性化合物浓度如下所述的含水悬浮液喷雾至滴流点。第二天,用赤霉病菌(Fusarium graminearum)的孢子含水悬浮液接种植物。然后将测试植物置于22~24℃和相对大气湿度为90%气候室中调节14天。然后肉眼测定麦穗上病菌侵染的发展程度。Potted wheat ears of the cultivar "Liaochun No. 10" are sprayed to the trickle point with an aqueous suspension at the concentration of active compound indicated below. The next day, plants are inoculated with an aqueous spore suspension of Fusarium graminearum. The test plants are then conditioned for 14 days in a climate chamber at 22-24° C. and a relative atmospheric humidity of 90%. The degree of development of the fungal infection on the ears was then determined visually.
表9单独的活性化合物Table 9 Individual Active Compounds
表10本发明的组合物Table 10 Compositions of the invention
“*”:使用Colby公式计算的效力。" * ": Potency calculated using Colby's formula.
应用实施例6 防治黄瓜灰霉病试验Application Example 6 Control of Cucumber Gray Mold Test
将栽培品种为“山东密刺”的盆栽黄瓜幼苗叶子用活性化合物浓度如下所述的含水悬浮液喷雾至滴流点。第二天,用灰霉病菌(Botrytis cinerea)的孢子含水悬浮液接种植物。然后将测试植物置于22~24℃和相对大气湿度为95~99%气候室中调节7天。然后肉眼测定叶子上病菌侵染的发展程度。Leaves of potted cucumber seedlings of the cultivar "Shandong Mici" are sprayed to the trickle point with an aqueous suspension having the concentration of active compound indicated below. The next day, plants are inoculated with an aqueous spore suspension of Botrytis cinerea. The test plants are then conditioned for 7 days in a climate chamber at 22-24° C. and a relative atmospheric humidity of 95-99%. The degree of development of the fungal infection on the leaves was then determined visually.
表11单独的活性化合物Table 11 Individual Active Compounds
表12本发明的组合物Table 12 Compositions of the invention
“*”:使用Colby公式计算的效力。" * ": Potency calculated using Colby's formula.
应用实施例7 防治水稻稻瘟病试验Application Example 7 Test of Controlling Rice Blast
将栽培品种为“越光”的盆栽水稻幼苗叶子用活性化合物浓度如下所述的含水悬浮液喷雾至滴流点。第二天,用稻瘟病菌(Pyricularia grisea)的孢子含水悬浮液接种植物。然后将测试植物置于24~28℃和相对大气湿度为95~99%气候室中调节5天。然后肉眼测定叶子上病菌侵染的发展程度。Leaves of potted rice seedlings of the cultivar "Koshihikari" are sprayed to the point of dripping with an aqueous suspension at the concentration of active compound indicated below. The next day, the plants were inoculated with an aqueous spore suspension of Pyricularia grisea. The test plants are then conditioned for 5 days in a climate chamber at 24-28° C. and a relative atmospheric humidity of 95-99%. The degree of development of the fungal infection on the leaves was then determined visually.
表29单独的活性化合物Table 29 Individual Active Compounds
表30本发明的组合物Table 30 Compositions of the invention
“*”:使用Colby公式计算的效力。" * ": Potency calculated using Colby's formula.
应用实施例8 防治黄瓜霜霉病试验Application Example 8 Control of Cucumber Downy Mildew Test
将栽培品种为“长春密刺”的盆栽黄瓜幼苗叶子用活性化合物浓度如下所述的含水悬浮液喷雾至滴流点。第二天,用霜霉病菌(Pseudoperonospora cubensis)的孢子含水悬浮液接种植物。然后将测试植物置于22~24℃和相对大气湿度为90%气候室中调节7天。然后目测测定叶子上病菌侵染的发展程度。Leaves of potted cucumber seedlings of the cultivar "Perigo Mici" are sprayed to the point of dripping with an aqueous suspension at the concentration of active compound indicated below. The next day, the plants are inoculated with an aqueous spore suspension of Pseudoperonospora cubensis . The test plants are then conditioned for 7 days in a climate chamber at 22-24° C. and a relative atmospheric humidity of 90%. The degree of development of the fungal infection on the leaves was then determined visually.
表31单独的活性化合物Table 31 Individual Active Compounds
表32本发明的组合物Table 32 Compositions of the invention
“*”:使用Colby公式计算的效力。" * ": Potency calculated using Colby's formula.
Claims (4)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2007101756285A CN101406191B (en) | 2007-10-09 | 2007-10-09 | A fungicidal composition |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2007101756285A CN101406191B (en) | 2007-10-09 | 2007-10-09 | A fungicidal composition |
Related Child Applications (5)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN 201110410209 Division CN102422837B (en) | 2007-10-09 | 2007-10-09 | Antifungal composition |
| CN 201210012335 Division CN102578100B (en) | 2007-10-09 | 2007-10-09 | Antifungal composition |
| CN 201210012456 Division CN102578125B (en) | 2007-10-09 | 2007-10-09 | Fungicidal composition |
| CN 201210012332 Division CN102578130B (en) | 2007-10-09 | 2007-10-09 | A fungicidal composition |
| CN 201110409917 Division CN102511490B (en) | 2007-10-09 | 2007-10-09 | A fungicidal composition |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN101406191A CN101406191A (en) | 2009-04-15 |
| CN101406191B true CN101406191B (en) | 2012-05-09 |
Family
ID=40569579
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN2007101756285A Expired - Fee Related CN101406191B (en) | 2007-10-09 | 2007-10-09 | A fungicidal composition |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN101406191B (en) |
Families Citing this family (22)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN104170837B (en) * | 2009-12-23 | 2016-06-29 | 沈阳中化农药化工研发有限公司 | A kind of Fungicidal composition containing pyraoxystrobin |
| CN103053606B (en) * | 2009-12-23 | 2014-08-06 | 中国中化股份有限公司 | Antifungal composition containing pyraoxystrobin |
| CN102106355B (en) * | 2009-12-23 | 2013-02-27 | 中国中化股份有限公司 | A kind of fungicidal composition containing pyraclostrobin |
| JP2011201853A (en) * | 2010-03-03 | 2011-10-13 | Sumitomo Chemical Co Ltd | Plant disease controlling composition and method for controlling plant disease |
| CN103503895B (en) * | 2012-06-19 | 2015-10-21 | 中国中化股份有限公司 | A kind of Fungicidal composition containing pyrazole compound and triazole class compounds |
| CN103651540A (en) * | 2012-09-14 | 2014-03-26 | 陕西美邦农药有限公司 | Bactericidal composition containing dimoxystrobin |
| CN103053579A (en) * | 2012-09-21 | 2013-04-24 | 吉林省八达农药有限公司 | Agricultural germicidal composition containing coumoxystrobin and iprodione |
| CN103704237B (en) * | 2012-09-28 | 2016-05-04 | 沈阳中化农药化工研发有限公司 | A kind of fungicidal composition containing pyrazole compound and imidazole compound |
| CN103719097A (en) * | 2012-10-16 | 2014-04-16 | 海利尔药业集团股份有限公司 | Sterilization composition containing pyraoxystrobin and azoxystrobin |
| CN103202301A (en) * | 2012-11-02 | 2013-07-17 | 海利尔药业集团股份有限公司 | Bactericidal composition containing pyraoxystrobin and mepanipyrim |
| CN103039460A (en) * | 2012-11-12 | 2013-04-17 | 海利尔药业集团股份有限公司 | Sterilization composition containing pyraoxystrobin and trifloxystrobin |
| CN103053561B (en) * | 2013-02-04 | 2015-06-17 | 陕西农心作物科技有限公司 | Benthiavalicarb isopropyl containing bactericidal composition |
| CN105532696B (en) * | 2013-11-19 | 2018-07-03 | 沈阳中化农药化工研发有限公司 | Fungicidal composition and its application |
| CN103563947B (en) * | 2013-11-19 | 2015-08-19 | 中国中化股份有限公司 | A kind of Fungicidal composition and application thereof |
| CN114557361A (en) | 2013-11-26 | 2022-05-31 | Upl 有限公司 | Method for controlling rust |
| CN104186485A (en) * | 2014-09-22 | 2014-12-10 | 江苏省绿盾植保农药实验有限公司 | Microbicide composition for preventing and controlling wheat powdery mildew and application of composition |
| CN104336036B (en) * | 2014-10-14 | 2017-04-12 | 沈阳中化农药化工研发有限公司 | Fungicidal composition and application thereof |
| CN105766945B (en) * | 2014-12-23 | 2018-08-31 | 沈阳中化农药化工研发有限公司 | A kind of Fungicidal composition and its application |
| CN106256204A (en) * | 2015-06-17 | 2016-12-28 | 沈阳中化农药化工研发有限公司 | A kind of insecticidal and bactericidal composition |
| EP4555862A3 (en) | 2017-03-07 | 2025-10-15 | UPL Ltd | Fungicidal combinations |
| CN106804601A (en) * | 2017-03-16 | 2017-06-09 | 安徽省农业科学院植物保护与农产品质量安全研究所 | A kind of bactericidal composition containing pyraoxystrobin and probenazole and application thereof |
| CN112471162B (en) * | 2020-12-02 | 2023-06-30 | 青岛海利尔生物科技有限公司 | Sterilization composition containing tetramycin and pyraclostrobin |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1657524A (en) * | 2004-02-20 | 2005-08-24 | 沈阳化工研究院 | Substituted azole compounds and their preparation and application |
-
2007
- 2007-10-09 CN CN2007101756285A patent/CN101406191B/en not_active Expired - Fee Related
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1657524A (en) * | 2004-02-20 | 2005-08-24 | 沈阳化工研究院 | Substituted azole compounds and their preparation and application |
| CN1906171A (en) * | 2004-02-20 | 2007-01-31 | 沈阳化工研究院 | Substituted azole compounds and their preparation and application |
Also Published As
| Publication number | Publication date |
|---|---|
| CN101406191A (en) | 2009-04-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN101406191B (en) | A fungicidal composition | |
| CN109169678B (en) | A kind of bactericide and its composition | |
| CN102106349B (en) | Antifungal composition containing tebuconazole and pyrimidine compound | |
| CN102106355B (en) | A kind of fungicidal composition containing pyraclostrobin | |
| JP2008525349A (en) | A fungicide mixture containing enestrobrin and at least one active substance selected from the group of azoles | |
| CN101953352B (en) | Antifungal composition containing pyraclostrobin and pyrimidine compounds | |
| CN101953351B (en) | Fungicide containing pyrametostrobin | |
| CN102578130B (en) | A fungicidal composition | |
| CN102578100B (en) | Antifungal composition | |
| CN102422837A (en) | Antifungal composition | |
| CN102578125A (en) | Fungicidal composition | |
| CN103053606B (en) | Antifungal composition containing pyraoxystrobin | |
| CN102511490A (en) | Fungus killing composition | |
| CN103053563B (en) | Antifungal composition containing pyraoxystrobin | |
| CN103053582B (en) | Antifungal composition containing pyraoxystrobin | |
| CN103503895A (en) | Antifungal composition containing pyrazole compound and triazole compound | |
| CN104170837B (en) | A kind of Fungicidal composition containing pyraoxystrobin |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| ASS | Succession or assignment of patent right |
Owner name: SHENYANG RESEARCH INSTITUTE OF CHEMICAL INDUSTRY C Free format text: FORMER OWNER: SHENYANG RESEARCH INSTITUTE OF CHEMICAL INDUSTRY |
|
| C41 | Transfer of patent application or patent right or utility model | ||
| COR | Change of bibliographic data |
Free format text: CORRECT: ADDRESS; FROM: 100045 ZHONGHUA BUILDING, NO.A2, FUXINGMEN OUTER STREET, BEIJING TO: 100031 NO.28, FUXINGMEN INNER STREET, XICHENG DISTRICT, BEIJING |
|
| TA01 | Transfer of patent application right |
Effective date of registration: 20100617 Address after: 100031 Beijing, Xicheng District, the door of the revitalization of the main street, No. 28 Applicant after: SINOCHEM Corp. Co-applicant after: SHENYANG RESEARCH INSTITUTE OF CHEMICAL INDUSTRY Co.,Ltd. Address before: 100045. Zhonghua building, No. A2, Fuxing street, Beijing Applicant before: Sinochem Corp. Co-applicant before: Shenyang Research Institute of Chemical Industry |
|
| C53 | Correction of patent of invention or patent application | ||
| CB03 | Change of inventor or designer information |
Inventor after: Si Naiguo Inventor after: Chen Liang Inventor after: Liu Changling Inventor after: Li Miao Inventor after: Zhang Guosheng Inventor after: Li Keke Inventor after: Yuan Jing Inventor before: Si Naiguo Inventor before: Chen Liang Inventor before: Liu Changling Inventor before: Li Miao Inventor before: Zhang Guosheng Inventor before: Li Keke Inventor before: Yuan Jing |
|
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| C41 | Transfer of patent application or patent right or utility model | ||
| TR01 | Transfer of patent right |
Effective date of registration: 20160128 Address after: 110021 Liaodong Road, Tiexi District, Liaoning, No. 8-1, No. Patentee after: SHENYANG SINOCHEM AGROCHEMICALS R&D Co.,Ltd. Address before: 100031 Beijing, Xicheng District, the door of the revitalization of the main street, No. 28 Patentee before: SINOCHEM Corp. Patentee before: SHENYANG RESEARCH INSTITUTE OF CHEMICAL INDUSTRY Co.,Ltd. |
|
| CF01 | Termination of patent right due to non-payment of annual fee | ||
| CF01 | Termination of patent right due to non-payment of annual fee |
Granted publication date: 20120509 |