CN101340916A - External preparation with improved temporal stability of steroid - Google Patents
External preparation with improved temporal stability of steroid Download PDFInfo
- Publication number
- CN101340916A CN101340916A CNA2006800477616A CN200680047761A CN101340916A CN 101340916 A CN101340916 A CN 101340916A CN A2006800477616 A CNA2006800477616 A CN A2006800477616A CN 200680047761 A CN200680047761 A CN 200680047761A CN 101340916 A CN101340916 A CN 101340916A
- Authority
- CN
- China
- Prior art keywords
- external preparation
- steroid
- preparation
- mass
- present
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0014—Skin, i.e. galenical aspects of topical compositions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/56—Compounds containing cyclopenta[a]hydrophenanthrene ring systems; Derivatives thereof, e.g. steroids
- A61K31/57—Compounds containing cyclopenta[a]hydrophenanthrene ring systems; Derivatives thereof, e.g. steroids substituted in position 17 beta by a chain of two carbon atoms, e.g. pregnane or progesterone
- A61K31/573—Compounds containing cyclopenta[a]hydrophenanthrene ring systems; Derivatives thereof, e.g. steroids substituted in position 17 beta by a chain of two carbon atoms, e.g. pregnane or progesterone substituted in position 21, e.g. cortisone, dexamethasone, prednisone or aldosterone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/08—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing oxygen, e.g. ethers, acetals, ketones, quinones, aldehydes, peroxides
- A61K47/10—Alcohols; Phenols; Salts thereof, e.g. glycerol; Polyethylene glycols [PEG]; Poloxamers; PEG/POE alkyl ethers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/08—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing oxygen, e.g. ethers, acetals, ketones, quinones, aldehydes, peroxides
- A61K47/12—Carboxylic acids; Salts or anhydrides thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/70—Web, sheet or filament bases ; Films; Fibres of the matrix type containing drug
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- General Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Organic Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Dermatology (AREA)
- Pain & Pain Management (AREA)
- Rheumatology (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Medicinal Preparation (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
技术领域 technical field
本发明涉及在含有类固醇的外用制剂中使类固醇的经时稳定性得到改善、使用感良好的外用制剂。更详细而言,涉及通过在含有类固醇的外用制剂中进一步含有苯甲酸酯类局麻剂和羟基羧酸,使类固醇的经时稳定性得到改善、使用感良好的外用制剂。The present invention relates to an external preparation containing a steroid, which improves the temporal stability of a steroid and provides a good usability. More specifically, it relates to an external preparation containing a steroid with improved stability over time by further including a benzoate-based local anesthetic and a hydroxycarboxylic acid in the external preparation containing a steroid and having a good usability.
背景技术 Background technique
一直以来,具有类固醇骨架的化合物广泛用作药品的有效成分。例如,在由虫叮咬伴随的炎症、皮炎等炎症性皮肤疾病的治疗中使用含有具抗炎作用的肾上腺皮质类固醇的外用制剂。用于上述外用制剂的肾上腺皮质类固醇的例子有氢化可的松或地塞米松等。另外,含有类固醇的外用制剂已知有:例如包含戊酸醋酸泼尼松龙和抗组胺药的组合的皮肤外用剂(专利文献1)等。但是,在以往所提供的含有类固醇的外用制剂中,类固醇的经时稳定性未必充分。Conventionally, compounds having a steroid skeleton have been widely used as active ingredients of pharmaceuticals. For example, topical preparations containing adrenal corticosteroids having anti-inflammatory effects are used in the treatment of inflammatory skin diseases such as inflammation and dermatitis associated with insect bites. Examples of adrenal corticosteroids used in the above external preparations include hydrocortisone, dexamethasone and the like. In addition, steroid-containing external preparations include, for example, skin external preparations containing a combination of prednisolone acetate valerate and an antihistamine (Patent Document 1). However, in the steroid-containing external preparations provided conventionally, the stability of the steroid over time is not necessarily sufficient.
作为有关改善类固醇在制剂中的稳定性的先进技术,已知例如有:含有类固醇、甘油和极性油的皮肤外用剂(专利文献2);含有类固醇的泥敷剂,在其基质中含有0.005~0.05重量%的类固醇作为有效成分、含有15重量%以下的克罗米通和表面活性剂作为稳定化剂,上述克罗米通的混合量为上述类固醇的混合量的200~3000重量倍,并且上述表面活性剂的混合量为上述克罗米通的混合量的1/20~1/5重量倍(专利文献3);以及含有类固醇类抗炎药和其稳定化所需的有效量的丙烯酸聚合物的无水外用剂组合物(专利文献4)。但是若混合极性油,则极性油和水有时会分离。另外,克罗米通有时会引起皮肤刺激,导致使用感恶化,因此并不优选大量混合。另外,在无水外用剂组合物中,例如由于不含水,有时制剂的延展性差等、使用感不佳。As an advanced technology related to improving the stability of steroids in preparations, there are known, for example: skin external preparations containing steroids, glycerin and polar oils (patent document 2); steroid-containing poultices containing 0.005 ~ 0.05% by weight of steroid as an active ingredient, crotamiton and a surfactant containing 15% by weight or less of crotamiton as a stabilizer, the mixing amount of crotamiton is 200 to 3000 times by weight of the mixing amount of the above-mentioned steroid, And the mixing amount of the above-mentioned surfactant is 1/20~1/5 weight times of the mixing amount of the above-mentioned crotamiton (patent document 3); An anhydrous external preparation composition of an acrylic polymer (Patent Document 4). However, when a polar oil is mixed, the polar oil and water may separate. In addition, crotamiton may cause skin irritation and worsen the feeling of use, so it is not preferable to mix it in a large amount. In addition, in an anhydrous external preparation composition, for example, since it does not contain water, the spreadability of the preparation may be poor, and the feeling in use may be poor.
专利文献1:日本特开2002-356430号公报Patent Document 1: Japanese Patent Laid-Open No. 2002-356430
专利文献2:日本特开2001-247463号公报Patent Document 2: Japanese Patent Laid-Open No. 2001-247463
专利文献3:日本特开2000-26299号公报Patent Document 3: Japanese Patent Laid-Open No. 2000-26299
专利文献4:日本特开2001-233772号公报Patent Document 4: Japanese Patent Laid-Open No. 2001-233772
发明内容 Contents of the invention
发明所要解决的问题The problem to be solved by the invention
如上所述,在含有类固醇的外用制剂中,迄今为止还未发现类固醇的经时稳定性充分改善、且使用感良好的外用制剂。As described above, among external preparations containing steroids, no external preparations have been found that have sufficiently improved the stability of steroids over time and have good usability.
本发明的目的在于提供在含有类固醇的外用制剂中类固醇的经时稳定性得到改善、使用感良好的外用制剂。An object of the present invention is to provide an external preparation containing a steroid, which has improved stability over time of a steroid and has a good usability.
解决问题的方法way of solving the problem
因此,本发明人等为了解决上述问题进行了深入研究,发现:通过向外用制剂中进一步混合苯甲酸酯类局麻剂和羟基羧酸,可得到外用制剂中类固醇的经时稳定性显著改善、使用感良好的外用制剂,从而完成了本发明。Therefore, the inventors of the present invention conducted intensive studies to solve the above problems, and found that by further mixing a benzoate local anesthetic and a hydroxycarboxylic acid in the external preparation, the stability of the steroid in the external preparation can be significantly improved over time, The present invention has been accomplished by providing an external preparation with a good feeling of use.
即,本发明如下。That is, the present invention is as follows.
(1)外用制剂,该外用制剂是在含有类固醇的外用制剂中进一步含有苯甲酸酯类局麻剂和羟基羧酸。(1) An external preparation further comprising a benzoate-based local anesthetic and a hydroxycarboxylic acid in a steroid-containing external preparation.
(2)(1)的外用制剂,其中类固醇为酯系类固醇。(2) The external preparation of (1), wherein the steroid is an ester steroid.
(3)(1)或(2)的外用制剂,其中类固醇为醋酸地塞米松。(3) The external preparation of (1) or (2), wherein the steroid is dexamethasone acetate.
(4)(1)~(3)的外用制剂,其中苯甲酸酯类局麻剂为利多卡因。(4) The external preparation of (1) to (3), wherein the benzoate local anesthetic is lidocaine.
(5)(1)~(4)的外用制剂,其中羟基羧酸为枸橼酸或苹果酸。(5) The external preparation of (1) to (4), wherein the hydroxycarboxylic acid is citric acid or malic acid.
(6)(1)~(5)的外用制剂,其中进一步含有抗组胺药。(6) The external preparation of (1) to (5), which further contains an antihistamine.
(7)(6)的外用制剂,其中抗组胺药为盐酸苯海拉明。(7) The external preparation of (6), wherein the antihistamine is diphenhydramine hydrochloride.
(8)(1)~(7)的外用制剂,其中进一步含有低级醇。(8) The external preparation of (1) to (7), which further contains a lower alcohol.
(9)(1)~(8)的外用制剂,该外用制剂为液体制剂、霜剂或凝胶剂。(9) The external preparation of (1) to (8), which is a liquid preparation, a cream or a gel.
实施发明的最佳方式The best way to practice the invention
在本发明中,类固醇并不限于具抗炎作用的类固醇(肾上腺皮质类固醇),只要能够被苯甲酸酯类局麻剂和羟基羧酸稳定化,包含能够混合到外用制剂中的所有类固醇,本发明中优选肾上腺皮质类固醇。这种类固醇的具体例子有:氢化可的松、安西奈德、泼尼松龙、甲泼尼龙、二氟可龙、地塞米松、倍他米松、二氟拉松、二氟泼尼酯、曲安西龙、曲安奈德、哈西奈德、氟米松、布地奈德(budesonide)、莫米松、氟轻松、伏草隆(fluometuron)、氟氢缩松、阿氯米松、氯倍他索、地泼罗酮、倍氯米松、氯倍他松以及它们的酯、缩酮、缩醛和半缩醛衍生物等,但并不限于这些。In the present invention, steroids are not limited to steroids with anti-inflammatory effects (adrenal corticosteroids), and include all steroids that can be mixed into external preparations as long as they can be stabilized by benzoate local anesthetics and hydroxycarboxylic acids. Adrenocorticosteroids are preferred in the invention. Specific examples of such steroids are: hydrocortisone, amcinonide, prednisolone, methylprednisolone, difluocorolone, dexamethasone, betamethasone, diflurasone, difluprednate, Triamcinolone, triamcinolone acetonide, halcinonide, flumethasone, budesonide, mometasone, fluocinolone, fluometuron, fludrocetone, aclomethasone, clobetasol, Predone, beclomethasone, clobetasone and their esters, ketals, acetals and hemiacetal derivatives, etc., but not limited to these.
在本发明中,与有机酸形成酯的酯系类固醇具体优选如:醋酸地塞米松、醋酸泼尼松龙、醋酸氢化可的松、戊酸倍他米松、戊酸醋酸泼尼松龙等;特别优选醋酸地塞米松。在本发明中,类固醇可以单独使用,也可以将两种以上组合使用。In the present invention, ester steroids that form esters with organic acids are specifically preferred such as: dexamethasone acetate, prednisolone acetate, hydrocortisone acetate, betamethasone valerate, prednisolone acetate valerate, etc.; Dexamethasone acetate is particularly preferred. In the present invention, steroids may be used alone or in combination of two or more.
本发明的外用制剂具有以下有利的效果:即使对于类固醇中特别容易发生水解、稳定性低的酯系类固醇也充分改善了其经时稳定性。The external preparation of the present invention has the advantageous effect of sufficiently improving the stability over time even for ester steroids which are particularly prone to hydrolysis and have low stability among steroids.
在本发明中,对外用制剂总量中的类固醇的混合量没有特别限定,从类固醇的药理效果和安全性的角度考虑,通常希望为0.001~1%质量,优选为0.005~0.5%质量,特别优选为0.01~0.5%质量。In the present invention, the blending amount of the steroid in the total amount of the external preparation is not particularly limited, but from the viewpoint of the pharmacological effect and safety of the steroid, it is generally desirable to be 0.001 to 1% by mass, preferably 0.005 to 0.5% by mass, especially Preferably it is 0.01 to 0.5% by mass.
本发明中的苯甲酸酯类局麻剂是具有麻醉作用的公知成分,但有关象本发明一样提高外用制剂中类固醇的经时稳定性迄今为止还是未知的。在本发明中,苯甲酸酯类局麻剂的具体例子有:利多卡因、盐酸利多卡因、盐酸地布卡因、盐酸普鲁卡因等,但并不限于这些。另外,即使是现在未知的成分,但只要能够同时稳定羟基羧酸和类固醇,即可使用。本发明中特别优选利多卡因。在本发明中,苯甲酸酯类局麻剂可以单独使用,也可以将两种以上组合使用。The benzoate-based local anesthetic in the present invention is a known component having an anesthetic effect, but it has not been known so far to improve the temporal stability of steroids in external preparations as in the present invention. In the present invention, specific examples of benzoate local anesthetics include: lidocaine, lidocaine hydrochloride, dibucaine hydrochloride, procaine hydrochloride, etc., but are not limited to these. In addition, even unknown components can be used as long as they can stabilize both hydroxycarboxylic acid and steroid. Lidocaine is particularly preferred in the present invention. In the present invention, the benzoate local anesthetics may be used alone or in combination of two or more.
在本发明中,对外用制剂总量中的苯甲酸酯类局麻剂的混合量没有特别限定,从类固醇的稳定化和麻醉作用的角度考虑,通常希望为0.01~4%质量,优选为0.05~2%质量,特别优选为0.1~1%质量。In the present invention, the mixing amount of the benzoate local anesthetic in the total amount of the external preparation is not particularly limited, but it is generally desirable to be 0.01 to 4% by mass, preferably 0.05% by mass, from the viewpoint of stabilization of the steroid and anesthesia. ~2% by mass, particularly preferably 0.1-1% by mass.
在本发明中,羟基羧酸表示在有机化合物的1分子内具有羧基和羟基的物质。本发明中,羟基羧酸可以是其药理学上可接受的盐。盐的例子有:钠盐、钾盐、钙盐等。羟基羧酸的具体例子有:乳酸、枸橼酸、苹果酸等,但并不限于这些。本发明中特别优选枸橼酸或苹果酸。在本发明中有机羧酸可以单独使用,也可以将两种以上组合使用。In the present invention, a hydroxycarboxylic acid means a substance having a carboxyl group and a hydroxyl group in one molecule of an organic compound. In the present invention, the hydroxycarboxylic acid may be a pharmacologically acceptable salt thereof. Examples of salts include sodium salts, potassium salts, calcium salts and the like. Specific examples of hydroxycarboxylic acids include, but are not limited to, lactic acid, citric acid, and malic acid. Citric acid or malic acid is particularly preferred in the present invention. In the present invention, organic carboxylic acids may be used alone or in combination of two or more.
在本发明中,对外用制剂总量中的羟基羧酸的混合量没有特别限定,从类固醇的稳定化角度考虑,通常希望为0.001~2%质量,优选为0.01~1%质量,特别优选为0.1~0.5%质量。In the present invention, the blending amount of hydroxycarboxylic acid in the total amount of the external preparation is not particularly limited, but it is usually desirably 0.001 to 2% by mass, preferably 0.01 to 1% by mass, and particularly preferably 0.1 to 0.5% by mass.
对本发明的外用制剂总量中的类固醇、苯甲酸酯类局麻剂、羟基羧酸的混合量的相对比例没有特别限定,特别是从类固醇的经时稳定性的角度考虑,以类固醇为1时,苯甲酸酯类局麻剂优选为0.01~4000,更优选为0.1~400,特别优选为0.2~100;羟基羧酸优选为0.001~2000,更优选为0.02~200,特别优选为0.2~50。The relative proportions of the steroid, benzoate local anesthetic, and hydroxycarboxylic acid in the total amount of the external preparation of the present invention are not particularly limited, especially from the viewpoint of the stability of the steroid over time, when the steroid is 1 , Benzoate local anesthetics are preferably 0.01-4000, more preferably 0.1-400, especially preferably 0.2-100; hydroxycarboxylic acids are preferably 0.001-2000, more preferably 0.02-200, especially preferably 0.2-50 .
在本发明中,对外用制剂的剂型没有特别限定,例如可以是含有水的外用制剂等谋求类固醇的稳定性的剂型,具体例子有:液体制剂、霜剂、凝胶剂、软膏剂、贴剂等。本发明中,从制剂的使用感方面考虑,特别优选液体制剂、霜剂或凝胶剂。In the present invention, the dosage form of the external preparation is not particularly limited. For example, it may be a dosage form that seeks the stability of steroids, such as an external preparation containing water. Specific examples include: liquid preparation, cream, gel, ointment, patch wait. In the present invention, liquid formulations, creams, or gels are particularly preferred from the viewpoint of the usability of the formulations.
推测本发明的外用制剂中类固醇的稳定性主要源自抑制类固醇、特别是酯系类固醇的水解。因此,在本发明的外用制剂的适合实施方案中,相对于外用制剂总量,水的含量优选0.1~99%质量、更优选5~95%质量、特别优选10~90%质量。It is presumed that the stability of steroids in the external preparation of the present invention is mainly derived from the inhibition of hydrolysis of steroids, especially ester-based steroids. Therefore, in a suitable embodiment of the external preparation of the present invention, the content of water is preferably 0.1 to 99% by mass, more preferably 5 to 95% by mass, particularly preferably 10 to 90% by mass relative to the total amount of the external preparation.
在本发明中,外用制剂的pH通常为3~8,优选为4~7的范围内。例如已知醋酸地塞米松在外用制剂的pH低于3或高于8时会不稳定。从稳定性的角度考虑,对醋酸地塞米松而言更优选的pH为5~6.5。另外,当外用制剂的pH低于3或高于8时,有时会对皮肤产生刺激。In this invention, pH of an external preparation is 3-8 normally, Preferably it exists in the range of 4-7. For example, dexamethasone acetate is known to be unstable when the pH of topical formulations is below 3 or above 8. From the viewpoint of stability, the more preferable pH for dexamethasone acetate is 5-6.5. In addition, when the pH of the preparation for external use is lower than 3 or higher than 8, skin irritation may sometimes occur.
本发明的外用制剂中,可以进一步混合抗组胺药。抗组胺药是具有止痒作用的公知成分,通过向本发明的外用制剂中混合抗组胺药,可得到速效性的止痒作用。抗组胺药的具体例子有:盐酸苯海拉明、苯海拉明、扑尔敏、盐酸异西喷地等,但并不限于这些。上述抗组胺药可以单独使用,也可以将两种以上组合使用。本发明中特别优选苯海拉明、盐酸苯海拉明。In the external preparation of the present invention, an antihistamine may be further mixed. An antihistamine is a known component having an antipruritic effect, and by mixing the antihistamine in the external preparation of the present invention, an immediate antipruritic effect can be obtained. Specific examples of antihistamines include, but are not limited to, diphenhydramine hydrochloride, diphenhydramine, chlorpheniramine, isopendine hydrochloride, and the like. The above-mentioned antihistamines may be used alone or in combination of two or more. In the present invention, diphenhydramine and diphenhydramine hydrochloride are particularly preferred.
在本发明中,对外用制剂总量中的抗组胺药的混合量没有特别限定,从止痒作用的角度考虑,通常希望为0.1~4%质量,优选为0.5~3%质量,特别优选为0.75~2%质量。In the present invention, the compounding amount of the antihistamine in the total amount of the external preparation is not particularly limited, but it is generally desirable to be 0.1 to 4% by mass, preferably 0.5 to 3% by mass, and particularly preferably It is 0.75 to 2% by mass.
本发明的外用制剂中,可以进一步混合低级醇。通过向本发明的外用制剂中混合低级醇,提高了类固醇的皮肤渗透性。另外,利用气化热使低级醇挥散,同时赋予冷却感。低级醇的例子有:乙醇、正丙醇、异丙醇等,本发明中优选乙醇和异丙醇。In the external preparation of the present invention, a lower alcohol may be further mixed. By mixing lower alcohols in the external preparation of the present invention, the skin permeability of the steroid is improved. In addition, the lower alcohol is volatilized by the heat of vaporization, and a cooling feeling is provided. Examples of lower alcohols include ethanol, n-propanol, and isopropanol, among which ethanol and isopropanol are preferred in the present invention.
对低级醇的混合量没有特别限定,通常为0.1~70%质量,优选为5~60%质量,特别优选为10~50%质量。若低级醇的混合量为0.1%质量以下,则无法给予皮肤充分的冷却感;若多于70%质量,则对皮肤的刺激性有时会变强。The compounding quantity of a lower alcohol is not specifically limited, Usually, it is 0.1-70 mass %, Preferably it is 5-60 mass %, Especially preferably, it is 10-50 mass %. If the blending amount of the lower alcohol is less than 0.1% by mass, sufficient cooling sensation cannot be given to the skin, and if it is more than 70% by mass, the irritation to the skin may become stronger.
在本发明的外用制剂中,可以进一步含有除类固醇、抗组胺药、苯甲酸酯类局麻剂以外的有效成分、保湿剂、保存剂、稳定化剂和润湿剂等。The external preparation of the present invention may further contain active ingredients other than steroids, antihistamines, and benzoate local anesthetics, moisturizers, preservatives, stabilizers, and wetting agents.
作为除类固醇、抗组胺药、苯甲酸酯类局麻剂以外的有效成分,可以列举出:甘草酸的钾盐或铵盐等抗炎药;抗坏血酸或抗坏血酸衍生物等美白剂等。Examples of active ingredients other than steroids, antihistamines, and benzoate local anesthetics include anti-inflammatory agents such as potassium or ammonium glycyrrhizic acid; whitening agents such as ascorbic acid or ascorbic acid derivatives, and the like.
保湿剂的例子有:纤维素衍生物类、黄原胶、羧甲基纤维素钠、果胶、透明质酸、海藻酸钠、鹿角菜胶等。Examples of humectants include cellulose derivatives, xanthan gum, sodium carboxymethylcellulose, pectin, hyaluronic acid, sodium alginate, carrageenan, and the like.
保存剂的例子有:对羟基苯甲酸酯类、苯扎氯铵等。Examples of preservatives include parabens, benzalkonium chloride, and the like.
稳定化剂的例子有:亚硫酸钠、亚硫酸氢钠、二丁基羟基甲苯、丁基羟基茴香醚、依地酸等。Examples of stabilizers include sodium sulfite, sodium bisulfite, dibutylhydroxytoluene, butylhydroxyanisole, edetic acid and the like.
润湿剂的例子有:丙二醇、1,3-丁二醇、异丙二醇、聚乙二醇等多元醇。Examples of wetting agents include polyhydric alcohols such as propylene glycol, 1,3-butanediol, isopropylene glycol, and polyethylene glycol.
本发明的外用制剂可以按照本领域技术人员所公知的方法容易地进行制备。例如,当制备液体制剂作为外用制剂时,通过将各成分分别溶解在适当的溶剂中后混合,即可制备本发明的外用制剂。The external preparation of the present invention can be easily prepared according to methods known to those skilled in the art. For example, when preparing a liquid preparation as an external preparation, the external preparation of the present invention can be prepared by dissolving the respective components in an appropriate solvent and then mixing them.
本发明的外用制剂用于类固醇或类固醇和抗组胺药所适用的疾病或症状,例如用于出现发痒、发红等诸症状的皮肤疾病部位的治疗和症状缓解。使用时将适量的本发明的外用制剂涂在患处。The external preparation of the present invention is used for diseases or symptoms to which steroids or steroids and antihistamines are applicable, for example, it is used for the treatment and symptom relief of skin disease sites with symptoms such as itching and redness. When in use, an appropriate amount of the external preparation of the present invention is applied to the affected part.
实施例Example
以下使用实施例来详细说明本发明,但本发明并不受限于此。The present invention will be described in detail below using examples, but the present invention is not limited thereto.
〔实施例1〕[Example 1]
将0.025g醋酸地塞米松和0.5g利多卡因加入到45g乙醇中溶解,制成乙醇溶液。接着,将2g盐酸苯海拉明和0.25g枸橼酸加入到30g纯净水中搅拌,制成水溶液。之后,将上述水溶液和乙醇溶液混合,加入纯净水使总量为100mL,得到实施例1的液体制剂(pH6.0)。Add 0.025g of dexamethasone acetate and 0.5g of lidocaine into 45g of ethanol and dissolve to prepare an ethanol solution. Next, 2 g of diphenhydramine hydrochloride and 0.25 g of citric acid were added to 30 g of purified water and stirred to prepare an aqueous solution. Thereafter, the above-mentioned aqueous solution and ethanol solution were mixed, and purified water was added to make the total amount 100 mL, whereby the liquid preparation (pH 6.0) of Example 1 was obtained.
〔比较例1〕[Comparative Example 1]
制备在实施例1的制剂中没有混合利多卡因的乙醇溶液,并和按照与实施例1相同的方法制备的水溶液混合,搅拌。之后添加枸橼酸钠使pH为6.0。向该溶液中加入纯净水使总量为100mL,得到比较例1的液体制剂。An ethanol solution in which lidocaine was not mixed in the preparation of Example 1 was prepared, mixed with the aqueous solution prepared in the same manner as in Example 1, and stirred. Sodium citrate was then added to bring the pH to 6.0. Purified water was added to this solution so that the total amount would be 100 mL, whereby the liquid preparation of Comparative Example 1 was obtained.
〔比较例2〕[Comparative Example 2]
制备在实施例1的制剂中加入磷酸以代替枸橼酸的水溶液,并和按照与实施例1相同的方法得到的乙醇溶液混合。向混合液中加入磷酸氢二钠使pH为6.0。向该溶液中加入纯净水使总量为100mL,得到比较例2的液体制剂。An aqueous solution in which phosphoric acid was added instead of citric acid to the formulation of Example 1 was prepared and mixed with the ethanol solution obtained in the same manner as in Example 1. Disodium hydrogen phosphate was added to the mixture to bring the pH to 6.0. Purified water was added to this solution so that the total amount would be 100 mL, whereby the liquid formulation of Comparative Example 2 was obtained.
〔试验例1〕[Test Example 1]
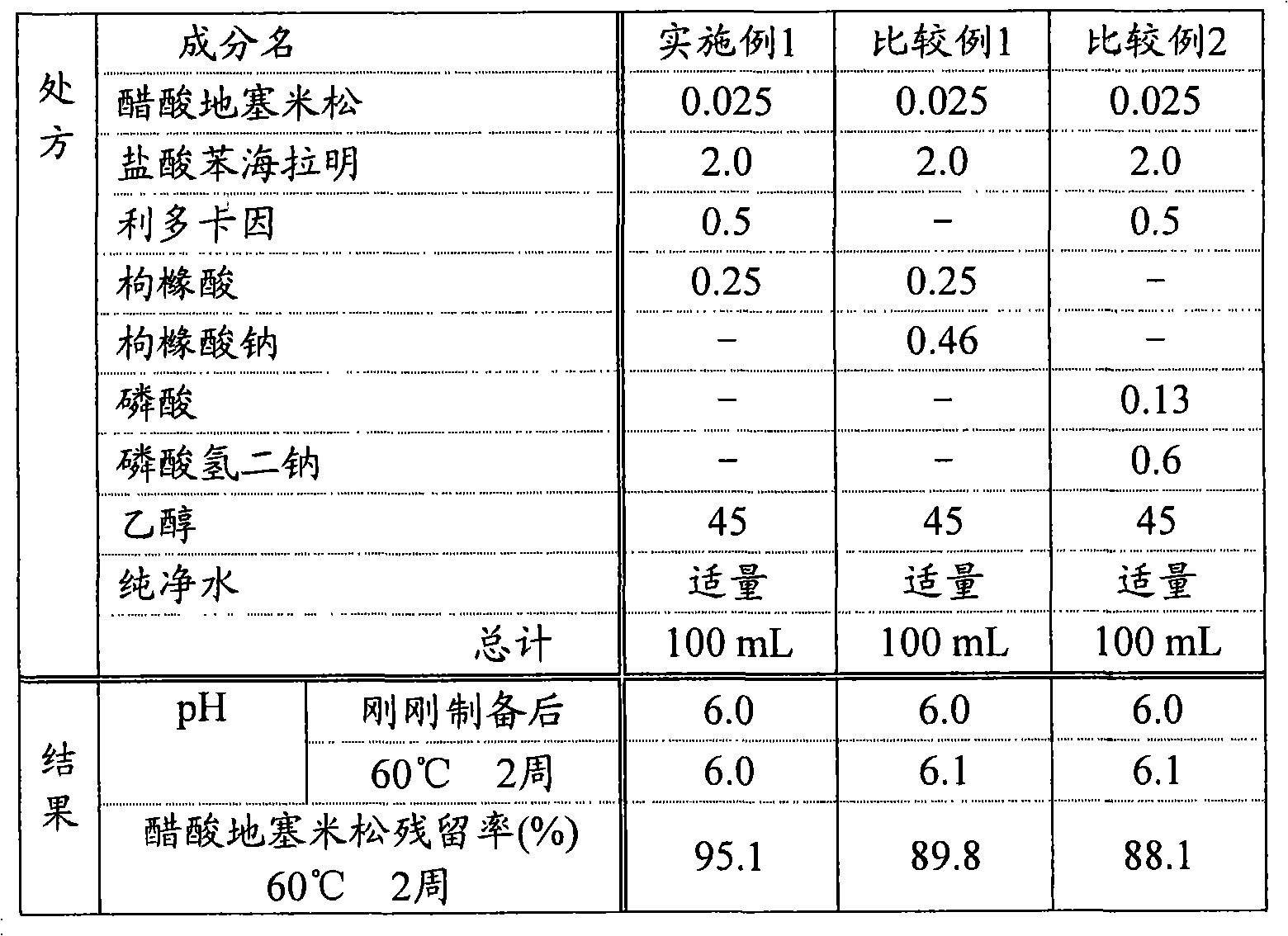
测定实施例1、比较例1和比较例2的样品在刚刚制备后和60℃下保存2周后的pH。另外,通过利用HPLC分离、测定在样品刚刚制备后和60℃下保存2周后的醋酸地塞米松的含量,算出60℃下保存2周后的醋酸地塞米松的残留率。结果如表1所示。The pH of the samples of Example 1, Comparative Example 1, and Comparative Example 2 was measured immediately after preparation and after storage at 60° C. for 2 weeks. In addition, the residual ratio of dexamethasone acetate after storage at 60° C. for 2 weeks was calculated by separating and measuring the content of dexamethasone acetate immediately after sample preparation and after storage at 60° C. for 2 weeks by HPLC. The results are shown in Table 1.
表1Table 1
在实施例1、不含苯甲酸酯类局麻剂-利多卡因的比较例1和不含作为羟基羧酸-枸橼酸的比较例2中,60℃下保存2周后的pH与各制剂刚刚制备后的pH相同。就60℃下保存2周后的醋酸地塞米松的残留率(60℃下保存2周后的含量/刚刚制备后的含量×100)而言,比较例1和比较例2中低于90%,而实施例1中为约95%,醋酸地塞米松的稳定性显著提高。另外,实施例1的制剂具有速干性,显示良好的使用感。In Example 1, Comparative Example 1 that does not contain benzoate local anesthetic-lidocaine, and Comparative Example 2 that does not contain citric acid as hydroxycarboxylic acid-citric acid, the pH after storage at 60°C for 2 weeks is different from that of each The pH of the formulation was the same immediately after preparation. The residual rate of dexamethasone acetate after storage at 60°C for 2 weeks (content after storage at 60°C for 2 weeks/content immediately after preparation×100) was less than 90% in Comparative Example 1 and Comparative Example 2 , and about 95% in Example 1, the stability of dexamethasone acetate is significantly improved. In addition, the preparation of Example 1 has quick-drying properties and exhibits a good feeling of use.
〔实施例2〕[Example 2]
将0.025g醋酸地塞米松和0.5g利多卡因加入到45g乙醇中溶解,制成乙醇溶液。接着,将2g盐酸苯海拉明和0.32g苹果酸加入到30g纯净水中搅拌,制成水溶液。之后将水溶液和乙醇溶液混合,加入纯净水使总量为100mL,得到实施例2的液体制剂(pH6.06)。在60℃下保存10天后pH为6.09。醋酸地塞米松的残留率(60℃下保存10天后的含量/刚刚制备后的含量×100)为96.6%,醋酸地塞米松的稳定性显著提高。应说明的是,在醋酸地塞米松的含量测定中,与试验例1同样使用HPLC。Add 0.025g of dexamethasone acetate and 0.5g of lidocaine into 45g of ethanol and dissolve to prepare an ethanol solution. Next, 2 g of diphenhydramine hydrochloride and 0.32 g of malic acid were added to 30 g of purified water and stirred to prepare an aqueous solution. Thereafter, the aqueous solution and the ethanol solution were mixed, and purified water was added to make the total amount 100 mL, thereby obtaining the liquid preparation (pH 6.06) of Example 2. The pH was 6.09 after storage at 60°C for 10 days. The residual rate of dexamethasone acetate (content after storage at 60° C. for 10 days/content just after preparation×100) was 96.6%, and the stability of dexamethasone acetate was significantly improved. In addition, in the content measurement of dexamethasone acetate, HPLC was used similarly to Test Example 1.
〔制备例1〕(液体制剂)[Preparation Example 1] (liquid preparation)
醋酸地塞米松 0.025%w/vDexamethasone acetate 0.025%w/v
盐酸苯海拉明 2.0%w/vDiphenhydramine Hydrochloride 2.0% w/v
利多卡因 0.5%w/vLidocaine 0.5% w/v
l-薄荷醇 3.0%w/vl-Menthol 3.0% w/v
dl-樟脑 2.0%w/vdl-camphor 2.0% w/v
枸橼酸 0.3%w/vCitric Acid 0.3%w/v
乙醇 30%w/vEthanol 30% w/v
纯净水 适量Appropriate amount of purified water
总计 100mLTotal 100mL
将醋酸地塞米松、l-薄荷醇、dl-樟脑和利多卡因加入到乙醇中溶解,制成乙醇溶液。另外,将盐酸苯海拉明和枸橼酸加入到纯净水中搅拌,制成水溶液。之后,将上述水溶液和乙醇溶液混合,得到外用液体制剂。Add dexamethasone acetate, l-menthol, dl-camphor and lidocaine into ethanol and dissolve to prepare ethanol solution. Alternatively, add diphenhydramine hydrochloride and citric acid to purified water and stir to prepare an aqueous solution. After that, the above-mentioned aqueous solution and ethanol solution were mixed to obtain a liquid preparation for external use.
〔制备例2〕(液体制剂)[Preparation Example 2] (liquid preparation)
醋酸泼尼松龙 0.5%w/vPrednisolone Acetate 0.5% w/v
利多卡因 0.5%w/vLidocaine 0.5% w/v
枸橼酸 0.3%w/vCitric acid 0.3%w/v
乙醇 30%w/vEthanol 30% w/v
纯净水 适量Purified water Appropriate amount
总计100mLTotal 100mL
将醋酸泼尼松龙和利多卡因加入到乙醇中溶解,制成乙醇溶液。将枸橼酸加入到纯净水中搅拌,制成水溶液。之后,将上述水溶液和乙醇溶液混合,得到外用液体制剂。Add prednisolone acetate and lidocaine into ethanol and dissolve to prepare ethanol solution. Add citric acid to purified water and stir to make an aqueous solution. After that, the above-mentioned aqueous solution and ethanol solution were mixed to obtain a liquid preparation for external use.
〔制备例3〕(凝胶剂)[Preparation Example 3] (Gel)
醋酸氢化可的松 0.5%质量Hydrocortisone acetate 0.5% by mass
盐酸苯海拉明 1.0%质量Diphenhydramine hydrochloride 1.0% by mass
利多卡因 0.5%质量Lidocaine 0.5% by mass
枸橼酸 0.3%质量Citric acid 0.3% by mass
乙醇 30%质量Ethanol 30% by mass
聚羧乙烯 1.2%质量Carbopol 1.2% by mass
二异丙醇胺 适量(调节pH)Diisopropanolamine Appropriate amount (adjust pH)
纯净水 适量Purified water Appropriate amount
总计100gTotal 100g
将盐酸苯海拉明、枸橼酸溶解在水中,之后加入聚羧乙烯,制成凝胶相。使用另外的容器,将醋酸氢化可的松、利多卡因溶解在乙醇中,制成乙醇相。将凝胶相和乙醇相混合,加入二异丙醇胺,调节pH至6,得到外用凝胶剂。Diphenhydramine hydrochloride and citric acid are dissolved in water, and then polycarboxyethylene is added to form a gel phase. Using a separate container, dissolve hydrocortisone acetate and lidocaine in ethanol to prepare an ethanol phase. Mix the gel phase and the ethanol phase, add diisopropanolamine, adjust the pH to 6, and obtain the gel for external use.
〔制备例4〕(液体制剂)[Preparation Example 4] (liquid preparation)
醋酸地塞米松 0.025%w/vDexamethasone acetate 0.025%w/v
盐酸地布卡因 0.5%w/vDibucaine Hydrochloride 0.5%w/v
利多卡因 0.5%w/vLidocaine 0.5% w/v
枸橼酸 0.3%w/vCitric Acid 0.3%w/v
异丙醇 30%w/vIsopropyl Alcohol 30% w/v
纯净水 适量Appropriate amount of purified water
总计100mLTotal 100mL
将醋酸地塞米松和利多卡因加入到异丙醇中溶解,制成异丙醇溶液。另外,将盐酸地布卡因和枸橼酸加入到纯净水中搅拌,制成水溶液。之后,将上述水溶液和异丙醇溶液混合,得到外用液体制剂。Add dexamethasone acetate and lidocaine to isopropanol and dissolve to make an isopropanol solution. In addition, dibucaine hydrochloride and citric acid were added to purified water and stirred to prepare an aqueous solution. After that, the above-mentioned aqueous solution and isopropanol solution were mixed to obtain a liquid preparation for external use.
〔制备例5〕(液体制剂)[Preparation Example 5] (liquid preparation)
醋酸地塞米松 0.025%w/vDexamethasone acetate 0.025%w/v
盐酸苯海拉明 2.0%w/vDiphenhydramine Hydrochloride 2.0% w/v
利多卡因 0.5%w/vLidocaine 0.5% w/v
枸橼酸 0.3%w/vCitric Acid 0.3%w/v
丙二醇 5.0%w/vPropylene Glycol 5.0%w/v
聚乙二醇400 10%w/vMacrogol 400 10% w/v
纯净水 适量Appropriate amount of purified water
总计100mLTotal 100mL
向丙二醇和聚乙二醇400的混合液中加入醋酸地塞米松和利多卡因,加热溶解,制成多元醇溶液。另外,将盐酸苯海拉明和枸橼酸加入到纯净水中搅拌,制成水溶液。之后,将上述水溶液和多元醇溶液混合,得到外用液体制剂。Add dexamethasone acetate and lidocaine to the mixed solution of propylene glycol and polyethylene glycol 400, heat and dissolve to prepare a polyol solution. Alternatively, diphenhydramine hydrochloride and citric acid were added to purified water and stirred to prepare an aqueous solution. After that, the above-mentioned aqueous solution and polyol solution are mixed to obtain a liquid preparation for external use.
〔制备例6〕(霜剂)[Preparation Example 6] (Cream)
戊酸倍他米松 0.012%质量Betamethasone valerate 0.012% by mass
盐酸苯海拉明 2.0%质量Diphenhydramine hydrochloride 2.0% by mass
盐酸利多卡因 0.5%质量Lidocaine hydrochloride 0.5% by mass
枸橼酸 0.1%质量Citric acid 0.1% by mass
枸橼酸钠 适量(调节pH)Sodium citrate Appropriate amount (to adjust pH)
己二酸二异丙酯 3.0%质量Diisopropyl adipate 3.0% by mass
中链脂肪酸三甘油酯 10.0%质量Medium-chain fatty acid triglycerides 10.0% by mass
吐温20 5.0%质量Tween 20 5.0% by mass
吐温60 2.0%质量Tween 60 2.0% by mass
丙二醇 10.0%质量Propylene glycol 10.0% by mass
1,3-丁二醇 5.0%质量1,3-Butanediol 5.0% by mass
聚乙二醇400 10%质量Macrogol 400 10% by mass
纯净水 适量Purified water Appropriate amount
总计100gTotal 100g
向中链脂肪酸三甘油酯中加入戊酸倍他米松、己二酸二异丙酯、吐温20、吐温60,加热至75℃,溶解。接下来,将盐酸苯海拉明、盐酸利多卡因、丙二醇、1,3-丁二醇、聚乙二醇400和枸橼酸溶解在水中,搅拌该水溶液和之前的溶解液直至均匀乳化,之后加入枸橼酸钠,调节pH至5.5,得到外用霜剂。Add betamethasone valerate, diisopropyl adipate, Tween 20, and Tween 60 to the medium-chain fatty acid triglyceride, heat to 75°C, and dissolve. Next, dissolve diphenhydramine hydrochloride, lidocaine hydrochloride, propylene glycol, 1,3-butanediol, polyethylene glycol 400, and citric acid in water, and stir the aqueous solution and the previous solution until uniformly emulsified, Then sodium citrate was added to adjust the pH to 5.5 to obtain a cream for external use.
产业实用性Industrial applicability
本发明提供在含有类固醇的外用制剂中、使类固醇的经时稳定性得到改善、使用感良好的外用制剂,本发明可用于医药领域等。The present invention provides an external preparation containing a steroid, which improves the temporal stability of a steroid and provides a good feeling of use. The present invention can be used in the field of medicine and the like.
Claims (9)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP369412/2005 | 2005-12-22 | ||
| JP2005369412 | 2005-12-22 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN101340916A true CN101340916A (en) | 2009-01-07 |
Family
ID=38188698
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CNA2006800477616A Pending CN101340916A (en) | 2005-12-22 | 2006-12-21 | External preparation with improved temporal stability of steroid |
Country Status (4)
| Country | Link |
|---|---|
| JP (1) | JP5111117B2 (en) |
| KR (1) | KR101121529B1 (en) |
| CN (1) | CN101340916A (en) |
| WO (1) | WO2007072923A1 (en) |
Cited By (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN104127397A (en) * | 2014-08-19 | 2014-11-05 | 董颖颖 | Medicine and method for treating acute and chronic allergic and congestive skin diseases |
| EP2658554A4 (en) * | 2010-12-29 | 2015-01-07 | Strategic Science & Tech Llc | Systems and methods for treatment of allergies and other indications |
| US9050365B2 (en) | 2004-04-19 | 2015-06-09 | Strategic Science & Technologies, Llc | Transdermal delivery of beneficial substances effected by a hostile biophysical environment |
| US9072659B2 (en) | 2009-06-24 | 2015-07-07 | Strategic Science & Technologies, Llc | Topical composition containing naproxen |
| US9155701B2 (en) | 2009-06-24 | 2015-10-13 | Strategic Science & Technologies, Llc | Delivery of ibuprofen and other compounds |
| US9226909B2 (en) | 2004-04-19 | 2016-01-05 | Strategic Science & Technologies, Llc | Beneficial effects of increasing local blood flow |
| US9463158B2 (en) | 2009-06-24 | 2016-10-11 | Strategic Science & Technologies, Llc | Treatment of erectile dysfunction and other indications |
| CN110051621A (en) * | 2018-01-18 | 2019-07-26 | 湖北舒邦药业有限公司 | A kind of compound dexamethasone acetate antiperspirant cream compositions and preparation method thereof |
| CN112716886A (en) * | 2020-12-03 | 2021-04-30 | 国药集团三益药业(芜湖)有限公司 | Dexamethasone acetate emulsifiable paste and preparation method thereof |
| CN113398101A (en) * | 2021-07-30 | 2021-09-17 | 温州医科大学附属眼视光医院 | Compound lidocaine gel patch |
| US11684624B2 (en) | 2009-06-24 | 2023-06-27 | Strategic Science & Technologies, Llc | Treatment of erectile dysfunction and other indications |
| US12138268B2 (en) | 2009-06-24 | 2024-11-12 | Strategic Science & Technologies, Llc | Treatment of erectile dysfunction and other indications |
Families Citing this family (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP5191121B2 (en) * | 2006-12-15 | 2013-04-24 | 東洋カプセル株式会社 | Diphenhydramine-containing pharmaceutical composition with improved solubility |
| WO2012158405A2 (en) * | 2011-05-16 | 2012-11-22 | Pearlman Dale L | Compositions and methods for the treatment of skin diseases |
| JP5849550B2 (en) * | 2011-09-08 | 2016-01-27 | 大正製薬株式会社 | External preparation containing steroidal anti-inflammatory drug |
| JP5909941B2 (en) * | 2011-09-08 | 2016-04-27 | 大正製薬株式会社 | External preparation containing steroidal anti-inflammatory drug |
| JP5879837B2 (en) * | 2011-09-08 | 2016-03-08 | 大正製薬株式会社 | External preparation containing steroidal anti-inflammatory drug |
| RU2486901C1 (en) * | 2012-03-23 | 2013-07-10 | Сергей Владимирович Сирак | Polycomponent adhesive ointment for treating erosive lesions of oral mucosa accompanying pemphigus |
| JP7133309B2 (en) * | 2016-12-29 | 2022-09-08 | 小林製薬株式会社 | Emulsified cream formulation |
| KR102444113B1 (en) * | 2018-11-14 | 2022-09-19 | 에이브이엠 바이오테크놀로지, 엘엘씨 | Stable glucocorticoid formulations |
| GB2607584B (en) * | 2021-06-03 | 2024-07-17 | Stichting Medische Kliniek Velsen | Composition and method of treatment |
| CN117338698B (en) * | 2023-11-02 | 2025-01-28 | 中国牧工商集团有限公司 | A kind of compound lidocaine gel and preparation method thereof |
| CN118130654A (en) * | 2024-03-06 | 2024-06-04 | 中国牧工商集团有限公司 | A method for analyzing related substances and BHT content in compound lidocaine prednisolone gel |
Family Cites Families (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH0236572B2 (en) * | 1984-11-13 | 1990-08-17 | Hokuriku Pharmaceutical | SUTEROIDOO177MONOESUTERUGANJUKURIIMUZAI |
| JPS61167614A (en) * | 1985-01-22 | 1986-07-29 | Mitsubishi Yuka Yakuhin Kk | Steroic-containing ointment |
| AU618517B2 (en) * | 1986-12-23 | 1992-01-02 | Eugene J. Van Scott | Additives enhancing topical actions of therapeutic agents |
| JP2860748B2 (en) * | 1993-12-24 | 1999-02-24 | エスエス製薬株式会社 | Stable hydrocortisone butyrate-containing cream |
| JP3545162B2 (en) * | 1997-03-05 | 2004-07-21 | 株式会社資生堂 | Oral composition |
| JP5307309B2 (en) * | 1999-09-03 | 2013-10-02 | ゼリア新薬工業株式会社 | External preparation containing prednisolone valerate acetate and basic local anesthetic |
| JP2004131472A (en) * | 2002-08-09 | 2004-04-30 | Taisho Pharmaceut Co Ltd | Hemorrhoid treatment ointment |
| JP4632687B2 (en) * | 2003-04-25 | 2011-02-16 | ロート製薬株式会社 | Nasal drops |
| JP2005206523A (en) * | 2004-01-23 | 2005-08-04 | Sankyo Co Ltd | Antipruritic agent for external use |
| JP4824336B2 (en) * | 2004-05-07 | 2011-11-30 | ロート製薬株式会社 | Topical skin preparation |
| JP4824337B2 (en) * | 2004-05-07 | 2011-11-30 | ロート製薬株式会社 | Topical skin preparation |
-
2006
- 2006-12-21 KR KR1020087017461A patent/KR101121529B1/en not_active Expired - Fee Related
- 2006-12-21 CN CNA2006800477616A patent/CN101340916A/en active Pending
- 2006-12-21 JP JP2007551158A patent/JP5111117B2/en active Active
- 2006-12-21 WO PCT/JP2006/325541 patent/WO2007072923A1/en not_active Ceased
Cited By (25)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9050365B2 (en) | 2004-04-19 | 2015-06-09 | Strategic Science & Technologies, Llc | Transdermal delivery of beneficial substances effected by a hostile biophysical environment |
| US9226909B2 (en) | 2004-04-19 | 2016-01-05 | Strategic Science & Technologies, Llc | Beneficial effects of increasing local blood flow |
| US10682357B2 (en) | 2009-06-24 | 2020-06-16 | Strategic Science & Technologies, Llc | Treatment of erectile dysfunction and other indications |
| US10172865B2 (en) | 2009-06-24 | 2019-01-08 | Strategic Science & Technologies, Llc | Treatment of erectile dysfunction and other indications |
| US9155701B2 (en) | 2009-06-24 | 2015-10-13 | Strategic Science & Technologies, Llc | Delivery of ibuprofen and other compounds |
| US9161915B2 (en) | 2009-06-24 | 2015-10-20 | Strategic Science & Technologies, Llc | Delivery of ibuprofen and other compounds |
| US11684624B2 (en) | 2009-06-24 | 2023-06-27 | Strategic Science & Technologies, Llc | Treatment of erectile dysfunction and other indications |
| US10898489B2 (en) | 2009-06-24 | 2021-01-26 | Strategic Science & Technologies, Llc | Treatment of erectile dysfunction and other indications |
| US12138268B2 (en) | 2009-06-24 | 2024-11-12 | Strategic Science & Technologies, Llc | Treatment of erectile dysfunction and other indications |
| US9737543B2 (en) | 2009-06-24 | 2017-08-22 | Strategic Science & Technologies, Llc | Treatment of erectile dysfunction and other indications |
| US9675619B2 (en) | 2009-06-24 | 2017-06-13 | Strategic Science & Technologies, Llc | Treatment of erectile dysfunction and other indications |
| US9492458B2 (en) | 2009-06-24 | 2016-11-15 | Strategic Science & Technologies, Llc | Treatment of erectile dysfunction and other indications |
| US9072659B2 (en) | 2009-06-24 | 2015-07-07 | Strategic Science & Technologies, Llc | Topical composition containing naproxen |
| US9463158B2 (en) | 2009-06-24 | 2016-10-11 | Strategic Science & Technologies, Llc | Treatment of erectile dysfunction and other indications |
| US9457092B2 (en) | 2009-06-24 | 2016-10-04 | Strategic Science & Technologies, Llc | Delivery of ibuprofen and other compounds |
| US9289495B2 (en) | 2010-12-29 | 2016-03-22 | Strategic Science & Technologies, Llc | Systems and methods for treatment of allergies and other indications |
| US9833456B2 (en) | 2010-12-29 | 2017-12-05 | Strategic Science & Technologies, Llc | Treatment of erectile dysfunction and other indications |
| US9498482B2 (en) | 2010-12-29 | 2016-11-22 | Strategic Science & Technologies, Llc | Treatment of erectile dysfunction and other indications |
| EP2658554A4 (en) * | 2010-12-29 | 2015-01-07 | Strategic Science & Tech Llc | Systems and methods for treatment of allergies and other indications |
| CN104127397B (en) * | 2014-08-19 | 2016-04-27 | 董颖颖 | One treats acute and chronic anaphylaxis and congested dermopathic medicine and method |
| CN104127397A (en) * | 2014-08-19 | 2014-11-05 | 董颖颖 | Medicine and method for treating acute and chronic allergic and congestive skin diseases |
| CN110051621A (en) * | 2018-01-18 | 2019-07-26 | 湖北舒邦药业有限公司 | A kind of compound dexamethasone acetate antiperspirant cream compositions and preparation method thereof |
| CN112716886A (en) * | 2020-12-03 | 2021-04-30 | 国药集团三益药业(芜湖)有限公司 | Dexamethasone acetate emulsifiable paste and preparation method thereof |
| CN112716886B (en) * | 2020-12-03 | 2022-07-08 | 国药集团三益药业(芜湖)有限公司 | Dexamethasone acetate emulsifiable paste and preparation method thereof |
| CN113398101A (en) * | 2021-07-30 | 2021-09-17 | 温州医科大学附属眼视光医院 | Compound lidocaine gel patch |
Also Published As
| Publication number | Publication date |
|---|---|
| KR101121529B1 (en) | 2012-02-28 |
| JP5111117B2 (en) | 2012-12-26 |
| JPWO2007072923A1 (en) | 2009-06-04 |
| KR20080080387A (en) | 2008-09-03 |
| WO2007072923A1 (en) | 2007-06-28 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5111117B2 (en) | External preparations with improved steroid stability over time | |
| CA2908571C (en) | Compositions of pharmaceutical actives containing diethylene glycol monoethyl ether or other alkyl derivatives | |
| JP4451061B2 (en) | Topical composition containing at least one vitamin D or one vitamin D analog and at least one corticosteroid | |
| US4552872A (en) | Penetrating topical pharmaceutical compositions containing corticosteroids | |
| US11179465B2 (en) | Topical compositions comprising a corticosteroid | |
| NO329486B1 (en) | Pharmaceutical compositions comprising vitamin D, a corticosteroid and a solvent component, and use thereof | |
| JP2001526210A (en) | Pharmaceutical compositions comprising micelles containing only one lipophilic carbohydrate corticosteroid and a surfactant | |
| CN101505725A (en) | Stable pharmacologically active compositions including vitamin D-containing and corticosteroid compounds with low pH compatibility | |
| CN116265017A (en) | Pharmaceutical composition comprising benvimod and corticosteroid | |
| US20250127711A1 (en) | Composition and method for treating fungal skin conditions and inflammation | |
| US6271219B2 (en) | Topical formulations comprising skin penetration agents and the use thereof | |
| EP1214055B1 (en) | Pharmaceutical gel containing active substances | |
| WO2009084020A2 (en) | Topical composition comprising halobetasol and salicylic acid | |
| CN105658202B (en) | Topical anti-inflammatory pharmaceutical composition in the form of zileuton cream | |
| JPS6218526B2 (en) | ||
| JP3029964B2 (en) | Steroid dissolving agent and external solution mainly containing steroids | |
| US20090233891A1 (en) | Pharmaceutical lotion comprising fluticasone propionate | |
| WO2016157112A1 (en) | Topical spray compositions of mometasone furoate | |
| CN111743853A (en) | A kind of external use pharmaceutical composition of near-neutral hydrocortisone butyrate | |
| HK1124771A (en) | Preparation for external use having improved temporal stability of steroid | |
| CN103127139B (en) | Difluprednate topical external preparation | |
| CN102309756A (en) | Compound medicament containing phosphodiesterase 4 inhibitor and glucocorticoid and used for treating dermatosis | |
| JP2005206523A (en) | Antipruritic agent for external use | |
| JP7106852B2 (en) | Suspension-type topical solution | |
| BR112012005895B1 (en) | COMBINATION AND COMPOSITION CONTAINING AN ANTIMICROBIAL AGENT, A GLUCOCORTICOID AND AN ANTIFUNGAL AGENT |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| REG | Reference to a national code |
Ref country code: HK Ref legal event code: DE Ref document number: 1124771 Country of ref document: HK |
|
| C02 | Deemed withdrawal of patent application after publication (patent law 2001) | ||
| WD01 | Invention patent application deemed withdrawn after publication |
Open date: 20090107 |
|
| REG | Reference to a national code |
Ref country code: HK Ref legal event code: WD Ref document number: 1124771 Country of ref document: HK |