CN101309706A - Drug release control composition and drug-releasing medical device - Google Patents
Drug release control composition and drug-releasing medical device Download PDFInfo
- Publication number
- CN101309706A CN101309706A CNA2006800430183A CN200680043018A CN101309706A CN 101309706 A CN101309706 A CN 101309706A CN A2006800430183 A CNA2006800430183 A CN A2006800430183A CN 200680043018 A CN200680043018 A CN 200680043018A CN 101309706 A CN101309706 A CN 101309706A
- Authority
- CN
- China
- Prior art keywords
- drug
- release
- medicament
- stent
- support
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Landscapes
- Medicinal Preparation (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
Abstract
Description
技术领域 technical field
本发明涉及药剂释放控制组合物等,尤其涉及对医疗器具等赋予药剂释放功能的药剂释放控制组合物及保持其的药剂释放性医疗器具,特别是支架(Stent)。The present invention relates to a controlled drug release composition and the like, and more particularly to a controlled drug release composition imparting a drug release function to a medical device and the like, and a drug releasing medical device holding it, particularly a stent.
背景技术 Background technique
近年,为了能够有效发挥药效,不仅对新药甚至对已知药物大力开发其制剂及给药技术。例如有用特殊的覆膜对药剂进行涂覆,使其过一定的时间后才释放药效组分的制剂技术。还有以药物传递系统(DDS)概念为基础,利用以脂质体为首的纳米球(nano sphere)、微胶囊的制剂技术得以广泛的研究。并且,这些DDS所追求的功能有,释放控制性、目标导向性、摄取·给药容易性和增强效果·降低副作用等。In recent years, in order to effectively exert the drug effect, not only new drugs but also known drugs have been vigorously developed for their formulations and drug delivery techniques. For example, there is a preparation technology in which a special film is used to coat the medicament so that the effective components are released after a certain period of time. In addition, based on the concept of drug delivery system (DDS), formulation technology using nanospheres and microcapsules including liposomes has been extensively studied. In addition, the functions pursued by these DDS include release controllability, target orientation, ease of ingestion and administration, enhancement of effect and reduction of side effects, and the like.
至今为止,作为利用于DDS的药剂缓释材料,一直被深入研究的有聚乳酸、乳酸/乙醇酸共聚物等聚合物基体(matrix)材料(专利文献1和2,非专利文献1)。但是,若仅仅向这些生物降解性聚合物中混合药剂,则在所传递处通常是无法获取期待的药剂释放速度的。因为在这种聚合物基体中,药剂扩散移动的速度非常慢并且不易从其游离(非专利文献1)。为了解决所述问题,已研发了通过将聚合物基体制成多孔体,或制成颗粒化以增加接触面积,进而确保或增加药剂释放量的技术,并且正处于实用化的过程中。对于上述聚合物基体,极为重要的是制成多孔体时对孔径的控制,并且需要高度的条件设定,所以不可避免地导致了制造成本的上升。So far, polymer matrix materials such as polylactic acid and lactic acid/glycolic acid copolymer have been intensively studied as drug sustained release materials used in DDS (Patent Documents 1 and 2, Non-Patent Document 1). However, simply mixing a drug with these biodegradable polymers generally fails to obtain the desired release rate of the drug at the delivery site. Because in such a polymer matrix, the speed of drug diffusion movement is very slow and is not easily dissociated therefrom (Non-Patent Document 1). In order to solve the above-mentioned problems, techniques for ensuring or increasing the drug release amount by making the polymer matrix porous or granulated to increase the contact area have been developed and are in the process of practical use. For the above-mentioned polymer matrix, it is extremely important to control the pore diameter when making a porous body, and a high degree of condition setting is required, which inevitably leads to an increase in manufacturing cost.
另一方面,随着医工学的进步,已开发出主要以诊断和治疗为目的,通过在活体内外结合、埋入或留置一些医疗用具、器件或装置等而达到所期望的目的的技术。而对于如导管、支架、人造血管等医疗用具上直接使用如上述的聚合物基体技术,多数人基本上持否定态度。这是由于,很难通过涂覆法在这些医疗器具的表面上形成多孔体,并且在该领域中,考虑到活体排异反应,反而需要平滑平面。On the other hand, with the advancement of medical engineering, technologies have been developed mainly for the purpose of diagnosis and treatment, and achieve the desired purpose by combining, embedding or indwelling some medical appliances, devices or devices inside and outside the living body. However, most people basically hold a negative attitude towards the direct use of the above-mentioned polymer matrix technology on medical devices such as catheters, stents, and artificial blood vessels. This is because it is difficult to form a porous body on the surface of these medical devices by a coating method, and in this field, a smooth plane is instead required in consideration of rejection in a living body.
适用于活体内的医疗器具之一的支架是用于治疗心脏冠动脉闭塞症的。即留置在血管内的支架能够补充切开部分的同时,防止血管的收缩,有效降低心脏冠动脉闭塞症患者的复发概率。The stent, which is one of the medical devices applicable to the living body, is used to treat coronary artery occlusive disease. That is, the indwelling stent in the blood vessel can supplement the cut part while preventing the contraction of the blood vessel, effectively reducing the recurrence probability of patients with coronary artery occlusive disease.
至今为止,已公开有各种关于治疗心脏冠动脉闭塞症为首的动脉血管闭塞症用的支架的材料、形状、以及手术方法。但是,以前的材料仍然无法完全避免再狭窄、再闭塞的危险,进而导致使用支架的血管成形术的适用范围变小。因此,医疗现场非常需要一种再狭窄、再闭塞危险少的支架。So far, various materials, shapes, and operation methods of stents for treating coronary artery occlusive disease including coronary artery occlusive disease have been disclosed. However, the previous materials still cannot completely avoid the risk of restenosis and reocclusion, which leads to the narrowing of the scope of application of angioplasty using stents. Therefore, a stent with less risk of restenosis and reocclusion is highly desired in the medical field.
并且,将抗癌剂、免疫抑制剂、抗生剂、抗凝固剂等,与各种高分子材料进行组合而成的药剂释放型支架也一直在研发中(专利文献2)。但是,此类药剂释放型支架中,对释放药剂的时机、其释放速度、释放量以及释放时间,根据要求进行设定,在现实上并不容易。例如,有可能出现,在留置后的初期发生猝发性的释放而无法实现持续的、缓释的释放,或者搭载药剂的方式有问题,导致从留置于体内的支架脱落的情况。In addition, drug-releasing stents that combine anticancer agents, immunosuppressants, antibiotics, anticoagulants, etc., with various polymer materials are also being developed (Patent Document 2). However, in such a drug-releasing stent, it is actually not easy to set the timing, release speed, release amount, and release time of the drug according to requirements. For example, sudden release may occur in the early stage after indwelling, and sustained and sustained release may not be achieved, or there may be a problem with the method of loading the drug, which may cause the stent to fall off from the indwelling stent.
作为上述抗凝血剂,已知的有,作为抗凝血酶药的阿加曲班(Argatroban)和作为抗血小板药的盐酸沙格雷酯(SarpogrelateHydrochloride)。专利文献3和4所公开的导管(catheter),是通过缓释阿加曲班而具有抗血栓性的医疗器具。现状是,至今为止,还没有将抗凝血酶药的阿加曲班或盐酸沙格雷酯用于支架并对其效果进行具体验证的例子,对于该药剂可使支架表现抗凝固性的必要释放速度,则完全不为人知。Argatroban, which is an antithrombin drug, and Sarpogrelate Hydrochloride, which is an antiplatelet drug, are known as the anticoagulant. Catheters disclosed in Patent Documents 3 and 4 are medical devices having antithrombotic properties by sustained release of argatroban. The current situation is that, until now, there have been no cases where antithrombin drugs such as argatroban or sargrelate hydrochloride were used in stents and their effects were specifically verified, and the release of the drugs necessary for the stent to exhibit anticoagulant properties has not been confirmed. Speed is completely unknown.
专利文献1:日本特开平9-151136号公报Patent Document 1: Japanese Patent Application Laid-Open No. 9-151136
专利文献2:日本特开平9-255590号公报Patent Document 2: Japanese Patent Application Laid-Open No. 9-255590
专利文献3:日本特开平6-292711号公报Patent Document 3: Japanese Patent Application Laid-Open No. 6-292711
专利文献4:日本特开平6-292718号公报Patent Document 4: Japanese Patent Application Laid-Open No. 6-292718
非专利文献1:【高分子加工】第45卷、第5号、222页、第6号、270页,1996年Non-Patent Document 1: [Polymer Processing] Vol. 45, No. 5, Page 222, No. 6, Page 270, 1996
非专利文献2:“Drug-Eluting Stent”,医学书院,2003年Non-Patent Document 2: "Drug-Eluting Stent", Academy of Medicine, 2003
发明内容 Contents of the invention
鉴于上述状况和问题,本发明人经过专心研究完成了本发明。即,发现了通过添加某种脂溶性低分子化合物,可加快药剂释放,进而完成了本发明。本发明提供可加快药剂释放且能够长期稳定地连续释放药剂的组合物,以及适用该组合物的药剂释放性医疗器具。In view of the above circumstances and problems, the present inventors have completed the present invention through intensive studies. That is, they discovered that the release of a drug can be accelerated by adding a certain fat-soluble low-molecular-weight compound, and completed the present invention. The present invention provides a composition capable of accelerating the drug release and continuously releasing the drug stably for a long period of time, and a drug-releasing medical device using the composition.
并且,本发明人进一步对上述的药剂的搭载形态是否也在很大程度上影响释放速度和连续释放时间,进行了专心研究。其结果发现,作为搭载该药剂的高分子材料,与药剂和高分子的相溶性对于在一定时间内缓释上述药剂具有重要的作用,优选搭载于非晶性高分子的事实,进而完成了本发明。Furthermore, the present inventors have conducted intensive studies on whether the above-mentioned loading form of the drug also greatly affects the release rate and continuous release time. As a result, it was found that as a polymer material carrying the drug, the compatibility with the drug and the polymer plays an important role in the sustained release of the drug for a certain period of time, and the fact that it is preferably carried on an amorphous polymer, and further completed this paper invention.
还发现通过将支架基材制成多孔体并将搭载了药剂的高分子材料保持在其孔内,能够使该药剂缓释一定时间。It has also been found that by making the stent base material porous and holding the polymer material loaded with the drug in the pores, the drug can be sustained-released for a certain period of time.
本发明的目的为,提供一种将含有抗凝血剂的高分子材料涂覆于支架或搭载于多孔性支架基材,能够使该药剂持续地缓释一定时间的药剂缓释性支架。The object of the present invention is to provide a drug sustained-release stent that can continuously release the drug for a certain period of time by coating a polymer material containing an anticoagulant on the stent or loading it on a porous stent substrate.
本发明的药剂释放控制组合物的特征为,其含有,100重量份的可溶于有机溶剂且非水溶性的有机高分子材料、5~60重量份的脂溶性低分子释放助剂、1~70重量份的药剂。The drug release controlled composition of the present invention is characterized in that it contains 100 parts by weight of an organic solvent-soluble and water-insoluble organic polymer material, 5-60 parts by weight of a fat-soluble low-molecular-weight release aid, 1- 70 parts by weight of the medicament.
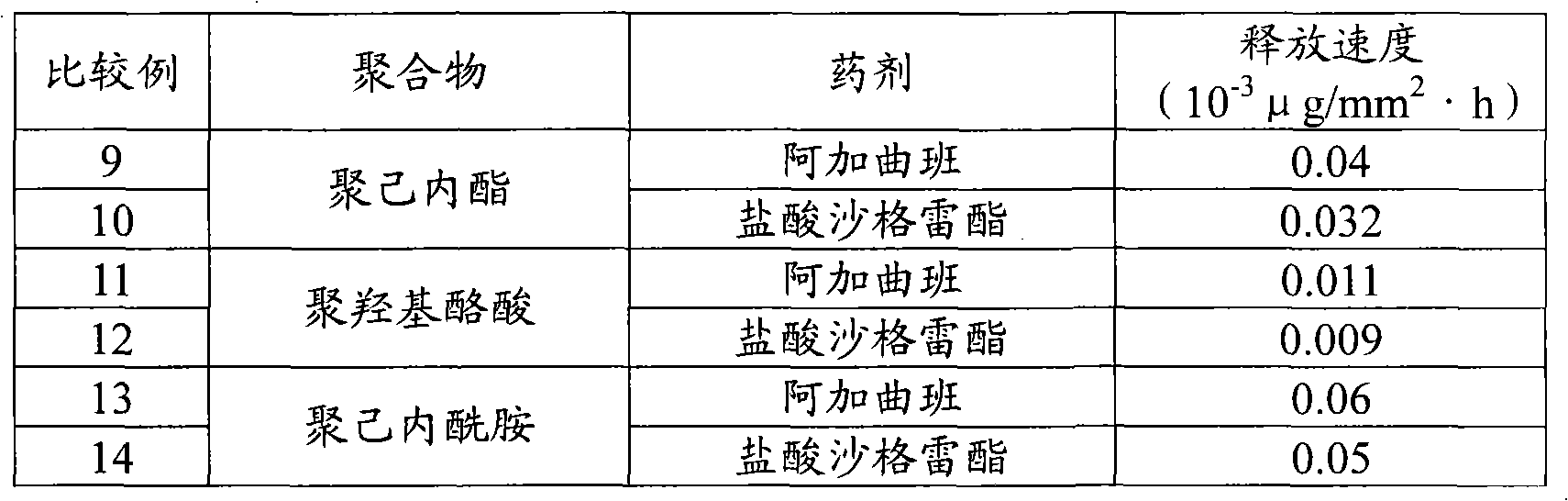
所述有机高分子材料优选具有生物降解性或活体适合性、或同时具备两者。生物降解性材料优选为脂肪族聚酯或脂肪族聚碳酸酯。具体的有聚乳酸、乳酸/乙醇酸共聚物、聚己内酯、聚羟基酪酸等。The organic polymer material preferably has biodegradability or biocompatibility, or both. The biodegradable material is preferably aliphatic polyester or aliphatic polycarbonate. Specifically, there are polylactic acid, lactic acid/glycolic acid copolymer, polycaprolactone, polyhydroxybutyric acid, and the like.
所述释放助剂为羧酸酯或甘油的单酯或二酯。优选为选自柠檬酸、酒石酸、苹果酸中的有机酸的酯、或甘油的单乙酸乙酯或二乙酸乙酯。The release aids are carboxylic acid esters or mono- or di-esters of glycerol. Preferred are esters of organic acids selected from citric acid, tartaric acid, malic acid, or ethyl monoacetate or diacetate of glycerol.
上述药剂为医药品,优选为抗凝固剂、抗癌剂或免疫抑制剂。The aforementioned agents are pharmaceuticals, preferably anticoagulants, anticancer agents or immunosuppressants.
上述组合物还可以含有细胞粘合性物质或医疗器具表面的内皮化促进物质。The above-mentioned composition may further contain a cell-adhesive substance or an endothelialization-promoting substance on the surface of a medical device.
本发明的药剂释放性医疗器具的特征为,保持上述组合物。The drug-releasing medical device of the present invention is characterized by holding the above composition.
优选为,在其表面形成有上述组合物的层。Preferably, a layer of the above composition is formed on the surface.
上述医疗器具优选为接触于活体的、或结合或被留置于活体内的医疗器具,具体的有支架、导管、线夹、器官代替用医疗器具、胶囊检测器或人造器官。The aforementioned medical device is preferably a medical device that is in contact with a living body, or is combined with or placed in a living body, and specifically includes a stent, a catheter, a wire clip, a medical device for organ replacement, a capsule detector or an artificial organ.
本发明的支架是用于治疗冠动脉狭窄的支架,其特征为,从其表面缓释阿加曲班(抗凝血酶药)或盐酸沙格雷酯(抗血小板药)或上述两种药剂。The stent of the present invention is a stent for treating coronary artery stenosis, and is characterized in that argatroban (antithrombin drug) or sargrelate hydrochloride (antiplatelet drug) or the above two agents are slowly released from its surface.
上述药剂的释放速度优选为,在留置该支架后的21天,阿加曲班和盐酸沙格雷酯皆为1×10-3μg/mm2·h~1μg/mm2·h。The release rate of the above-mentioned drugs is preferably 1×10 −3 μg/mm 2 ·h to 1 μg/mm 2 ·h for both argatroban and sargrelate hydrochloride 21 days after the stent is placed.
另外的特征为,涂覆在构成上述支架的金属表面的高分子材料中,或在多孔性支架基材中,搭载有缓释用的上述药剂。Another feature is that the above-mentioned drug for sustained release is carried on the polymer material coated on the metal surface constituting the above-mentioned stent, or on the porous stent substrate.
涂覆于上述支架表面的高分子材料优选为非晶性。The polymer material coated on the surface of the stent is preferably amorphous.
涂覆于上述支架表面的高分子材料优选为非晶性的生物降解性高分子材料。The polymer material coated on the surface of the stent is preferably an amorphous biodegradable polymer material.
优选的上述高分子材料有,具有生物降解性的聚乳酸或乳酸·乙醇酸共聚物。A preferable polymer material mentioned above is biodegradable polylactic acid or lactic acid-glycolic acid copolymer.
上述高分子材料优选还含有促进所搭载药剂的释放的助剂。The above-mentioned polymer material preferably further contains an auxiliary agent that promotes the release of the loaded drug.
上述药剂释放的助剂优选为酒石酸酯或苹果酸酯、或者是甘油的单酯或二酯。The auxiliary agents for the above drug release are preferably tartrates or malates, or mono- or di-glycerin esters.
上述构成支架的金属表面也可以为多孔体,缓释用的上述药剂被搭载于多孔体中。其孔径优选为0.01nm~300nm。The metal surface constituting the above-mentioned stent may be a porous body, and the above-mentioned drug for sustained release is carried on the porous body. The pore diameter thereof is preferably 0.01 nm to 300 nm.
本发明的药剂释放控制组合物因含有脂溶性低分子的释放助剂,可加快所含药剂在体内的释放。保持该组合物的医疗器具被传递或留置到规定的体内部位或体表面部位时,将释放其药剂,并且能够调整其时机、其释放速度、释放量以及释放时间。对所使用的药剂、医疗器具无特别限定。因此,本发明的药剂释放控制组合物能够对各种的医疗器具赋予药剂释放功能。The controlled drug release composition of the present invention can accelerate the release of the contained drug in the body because it contains a fat-soluble low-molecular release aid. When the medical device holding the composition is delivered or indwelled to a predetermined body part or body surface part, the drug will be released, and the timing, release speed, release amount and release time can be adjusted. The medicines and medical devices used are not particularly limited. Therefore, the drug release controlled composition of the present invention can impart a drug release function to various medical devices.
本发明的药剂释放型支架,因搭载阿加曲班、盐酸沙格雷酯的非晶性高分子材料与这些合成抗凝固药剂的相溶性良好,所以从留置于血管内的支架不易发生该药剂猝发性释放,进而能够将该药剂以所需的释放速度持续释放。In the drug-releasing stent of the present invention, since the amorphous polymer material loaded with argatroban and sargrelate hydrochloride has good compatibility with these synthetic anticoagulant drugs, drug burst is less likely to occur from the stent indwelling in the blood vessel. Sexual release, which in turn enables sustained release of the agent at a desired release rate.
通过添加用于促进抗凝固药剂释放的释放助剂,能够提高该药剂的释放速度,从留置支架的初期开始就释放可表现抗凝固剂药效的充分量。因此,本发明的药剂释放型支架通过支架的结构和抗凝固剂两者的作用,可有效防止动脉的再狭窄、再闭塞。By adding a release aid for accelerating the release of the anticoagulant agent, the release rate of the agent can be increased, and a sufficient amount to express the drug effect of the anticoagulant agent can be released from the initial stage of the indwelling stent. Therefore, the drug-releasing stent of the present invention can effectively prevent restenosis and reocclusion of arteries through the action of both the structure of the stent and the anticoagulant.
另外,通过对多孔性支架的孔径进行控制,可使该药剂以所需的释放速度持续被释放。In addition, by controlling the pore size of the porous scaffold, the drug can be continuously released at a desired release rate.
本发明中,“医疗器具”是指包括“医疗用具”,即广义上的应用于医疗领域的器具。In the present invention, "medical appliance" refers to "medical appliance", that is, an appliance used in the medical field in a broad sense.
“缓释性”是指,在制剂技术中,将药效组分缓慢释放的性质,是为了通过制剂设计防止药物的初期猝发,使药效长期持续。“生物降解性”是指在活体内以较快的速度被异化代谢,进而分解消失的特质。“活体适应性”是指对活体具有亲和性,不易被活体排异进而引起排除反应的、对活体无活性的倾向。"Sustained-release" refers to the property of slowly releasing the active ingredients in the preparation technology, in order to prevent the initial burst of the drug through the design of the preparation and make the drug effect last for a long time. "Biodegradability" refers to the characteristics of being metabolized at a relatively fast rate in the living body, and then decomposing and disappearing. "In vivo adaptability" refers to a tendency to have affinity for living organisms, not easily rejected by living organisms and cause rejection reactions, and inactive towards living organisms.
本说明书所述的“搭载”是指,在高分子基体或多孔体中,将药剂分子分散或制成纳米到亚微米的凝聚块进行分散。本说明书中,抗凝固药剂有时被记载为抗凝固剂或抗凝血剂。The term "carrying" in this specification refers to dispersing drug molecules or forming nanometer to submicron agglomerates in a polymer matrix or a porous body. In this specification, an anticoagulant agent may be described as an anticoagulant or an anticoagulant.
药剂释放控制组合物Drug release controlled composition
本发明的药剂释放控制组合物的特征为,其含有,The drug release controlled composition of the present invention is characterized in that it contains,
100重量份的可溶于有机溶剂且非水溶性的有机高分子材料、5~60重量份的脂溶性低分子释放助剂、和1~70重量份的药剂。100 parts by weight of an organic solvent-soluble and non-water-soluble organic polymer material, 5-60 parts by weight of a fat-soluble low-molecular release aid, and 1-70 parts by weight of a drug.
在此,“药剂释放控制”是指,对将药剂释放于体内规定部位的时机和其速度、释放量以及持续时间等进行的调整,并不只局限于缓释性。Here, "drug release control" refers to the adjustment of the timing, speed, release amount, duration, etc. of releasing a drug to a predetermined part of the body, and is not limited to sustained release.
下面,对本组合物所含的各组分进行说明。Next, each component contained in this composition is demonstrated.
·有机高分子材料·Organic polymer materials
留置于活体内的规定部位,作为用于搭载传递到目的部位的药剂的载体,使用可溶于有机溶剂且非水溶性的有机高分子材料。It is placed at a predetermined site in the living body, and an organic polymer material that is soluble in an organic solvent and insoluble in water is used as a carrier for loading and delivering a drug to the target site.
考虑到上述材料如后述地被使用在活体内外,从活体安全性的观点出发,这种有机高分子材料优选具有生物降解性或活体适应性,或者同时具备两者。Considering that the above-mentioned materials are used in vivo and in vitro as described later, such an organic polymer material preferably has biodegradability, biocompatibility, or both from the viewpoint of in vivo safety.
适合所述要求的高分子材料,尤其优选的是没有生理活性,并具有生物降解性的聚合物。生物降解性的聚合物有羟基碳酸均聚物、羟基碳酸低聚物或它们的混合物。例如,羟基碳酸、羟基碳酸低聚物的具体例子有聚乳酸、聚乙醇酸、乳酸-乙醇酸共聚物、聚丙交酯、聚(丙交酯-乙醇酸酯)、聚(乙二醇-丙交酯)、聚(乙醇酸-己内酯)、聚羟基丁酸酯、聚羟基异丁酸酯、聚戊内酯、聚γ-羟基戊酸、聚(羟基丁酸酯-羟基戊菊酯)、聚异丁基氰基丙烯酸酯、聚烷基氰基丙烯酸脂、聚丁二酸乙二醇酯等。还可以是聚乙酰氨基葡糖、壳聚糖、明胶、纤维素乙酸酯-对苯二酸酯等。Polymer materials suitable for the above requirements are especially preferably biodegradable polymers without physiological activity. Biodegradable polymers are hydroxycarbonate homopolymers, hydroxycarbonate oligomers or mixtures thereof. For example, specific examples of hydroxycarbonic acid and hydroxycarbonic acid oligomers include polylactic acid, polyglycolic acid, lactic acid-glycolic acid copolymer, polylactide, poly(lactide-glycolate), poly(ethylene glycol-propylene lactide), poly(glycolic acid-caprolactone), polyhydroxybutyrate, polyhydroxyisobutyrate, polyvalerolactone, poly-gamma-hydroxyvaleric acid, poly(hydroxybutyrate-hydroxyvalerate ), polyisobutyl cyanoacrylate, polyalkyl cyanoacrylate, polyethylene succinate, etc. It may also be polyacetylglucosamine, chitosan, gelatin, cellulose acetate-terephthalate and the like.
其中,作为本发明的材料尤其优选的共聚物有,脂肪族聚酯(如聚羟基脂肪酸酯)、脂肪族聚碳酸酯(如聚烯化碳酸酯)、或者聚己内酯等。具体的有乳酸/乙醇酸共聚物、聚乳酸、聚乙醇酸、聚苹果酸及它们的共聚物、乳酸-己内酯共聚物、聚羟基酪酸。这些聚合物可以是单独聚合物、也可以是共聚物、或它们的混合物、或者是其盐。本发明所使用的活体适合性高分子聚合物或生物降解性高分子聚合物比较容易获取,也可通过一般的合成法容易地合成。Among them, aliphatic polyester (such as polyhydroxyalkanoate), aliphatic polycarbonate (such as polyalkylene carbonate), or polycaprolactone are particularly preferable copolymers as materials of the present invention. Specifically, there are lactic acid/glycolic acid copolymers, polylactic acid, polyglycolic acid, polymalic acid and their copolymers, lactic acid-caprolactone copolymers, and polyhydroxybutyric acid. These polymers may be single polymers, copolymers, mixtures thereof, or salts thereof. The biocompatible polymer or biodegradable polymer used in the present invention is relatively easy to obtain and can be easily synthesized by a general synthesis method.
上述的聚乳酸、脂肪族聚酯、脂肪族碳酸酯可溶解于芳香族类有机溶剂(苯、甲苯、二甲苯等)、或卤类有机溶剂(二氯甲烷、氯仿、四氯化碳,1,1,2-三氯乙烷等),属于非水溶性聚合物。当药剂溶解于这些溶剂中时,可以直接使用。实际上,有很多药剂都是脂溶性,可溶于有机溶剂。相对于此,使用构成盐的药剂,例如盐酸沙格雷酯、萘莫司他、阿加曲班等时,其不溶于上述有机溶剂。因此,作为代替溶剂可使用如六氟异丙醇、三氟乙醇等的氟类醇等有机溶剂。The above-mentioned polylactic acid, aliphatic polyester, and aliphatic carbonate can be dissolved in aromatic organic solvents (benzene, toluene, xylene, etc.), or halogen organic solvents (methylene chloride, chloroform, carbon tetrachloride, 1 , 1,2-trichloroethane, etc.), are water-insoluble polymers. When the agent is dissolved in these solvents, it can be used directly. In fact, many agents are fat-soluble and soluble in organic solvents. On the other hand, when using agents that constitute salts, such as sargrelate hydrochloride, nafamostat, argatroban, etc., they are insoluble in the above-mentioned organic solvents. Therefore, an organic solvent such as a fluoroalcohol such as hexafluoroisopropanol or trifluoroethanol can be used instead of the solvent.
·释放助剂· Release aids
在药物传递系统中,通过向基材的高分子材料添加柠檬酸三丁酯、甘油或长链脂肪酸酯,可提高某种药剂的缓释速度(柠檬酸三丁酯、甘油的添加:Journal of Biomedical Mate rials Research,vol.13,497-507(1979),长链脂肪酸酯的添加:Journal of Controlled Releasevol.58,133-141,(1999))。In drug delivery systems, by adding tributyl citrate, glycerin, or long-chain fatty acid esters to the polymer material of the substrate, the sustained release rate of a certain drug can be increased (addition of tributyl citrate, glycerin: Journal of Biomedical Mate rials Research, vol.13, 497-507(1979), Addition of long-chain fatty acid esters: Journal of Controlled Release vol.58, 133-141, (1999)).
若仅向作为有机高分子材料的上述聚乳酸、乳酸/乙醇酸共聚物等聚合物中混合药剂,则在所传递的部位无法得到期望的药剂释放速度。本发明的机理是通过添加某种脂溶性低分子化合物,可加快药剂从溶剂挥发而固化的组合物中释放。本发明的组合物中,这种加快药剂释放的助剂与作为单体的有机高分子材料以及药剂一同被添加,进而发挥其效果。即,本发明的药剂释放控制组合物,不仅仅是起到缓释药剂的作用,还可调整药剂在体内规定部位上被释放的时机和其速度、释放量以及持续时间等。If a drug is simply mixed with polymers such as polylactic acid and lactic acid/glycolic acid copolymers as organic polymer materials, the desired release rate of the drug cannot be obtained at the delivery site. The mechanism of the invention is that by adding some fat-soluble low-molecular compound, the release of the medicament from the solvent volatilized and solidified composition can be accelerated. In the composition of the present invention, the auxiliary agent for accelerating drug release is added together with the organic polymer material as a monomer and the drug, and then exerts its effect. That is, the controlled drug release composition of the present invention not only functions to sustain the release of the drug, but also adjusts the timing, speed, release amount, and duration of drug release at a predetermined site in the body.
作为用于药剂释放控制组合物的低分子脂溶性释放助剂,优选从药剂释放效果以及安全性观点进行选择。安全性方面优选的是助剂自身对活体的毒性底,并且在活体内几乎完全被代谢,或完全不积累并且不被代谢而直接被排泄到体外的物质。适合这些要求的化合物有,脂肪族羧酸酯、或分子内具有羟基的酯化合物。例如有,分子内具有羟基的脂肪族羧酸酯、或者以甘油等多元醇为基底的酯。具体优选的有醋酸、丙酸等的碳数2~6的羧酸酯等,更优选选自柠檬酸、酒石酸、苹果酸中的有机酸的酯、或者甘油等多元醇的二酯、单酯。The low-molecular fat-soluble release aid used in the drug release controlled composition is preferably selected from the viewpoint of drug release effect and safety. In terms of safety, it is preferable that the auxiliary agent itself has low toxicity to the living body, and is almost completely metabolized in the living body, or does not accumulate at all and is not metabolized, but is directly excreted outside the body. Compounds suitable for these requirements include aliphatic carboxylic acid esters or ester compounds having a hydroxyl group in the molecule. For example, there are aliphatic carboxylic acid esters having a hydroxyl group in the molecule, or esters based on polyhydric alcohols such as glycerin. Specifically, preferred are carboxylic acid esters with 2 to 6 carbon atoms such as acetic acid and propionic acid, etc., more preferably esters of organic acids selected from citric acid, tartaric acid, and malic acid, or diesters and monoesters of polyalcohols such as glycerin. .
这些酯类的烷基长链优选为碳数1~12,更有选为1~6。其中甲基、乙基、丙基、丁基等因容易获取,并且与药剂和上述有机高分子材料的相溶性的观点出发优选。The long chain alkyl of these esters preferably has 1-12 carbon atoms, more preferably 1-6 carbon atoms. Among them, methyl group, ethyl group, propyl group, butyl group and the like are preferable from the viewpoint of easy availability and compatibility with the drug and the aforementioned organic polymer material.
优选的释放助剂例如有酒石酸二甲酯、酒石酸二乙酯、酒石酸二丙酯、酒石酸单甲酯、酒石酸单乙酯、酒石酸单丙酯等酒石酸的二酯类或酒石酸的半酯类;苹果酸二甲酯、苹果酸二乙酯、苹果酸二丙酯、苹果酸单甲酯、苹果酸单乙酯、苹果酸单丙酯等酒石酸的单酯类或苹果酸的二酯类;柠檬酸二甲酯、柠檬酸二乙酯、柠檬酸二丙酯、柠檬酸单甲酯、柠檬酸单乙酯、柠檬酸单丙酯、柠檬酸单丁酯等的柠檬酸的二酯类或柠檬酸的单酯类;或者甘油的部分醋酸酯(例如甘油单醋酸酯、甘油二醋酸酯等)。Preferred release aids are, for example, diesters of tartaric acid such as dimethyl tartrate, diethyl tartrate, dipropyl tartrate, monomethyl tartrate, monoethyl tartrate, monopropyl tartrate or half esters of tartaric acid; Dimethyl malate, diethyl malate, dipropyl malate, monomethyl malate, monoethyl malate, monopropyl malate and other monoesters of tartaric acid or diesters of malic acid; citric acid Citric acid diesters or citric acid such as dimethyl citrate, diethyl citrate, dipropyl citrate, monomethyl citrate, monoethyl citrate, monopropyl citrate, monobutyl citrate, etc. monoesters; or partial acetates of glycerol (such as glycerol monoacetate, glycerol diacetate, etc.).
释放助剂的添加量为,相对于上述有机高分子材料100重量份,释放助剂添加5~60重量份,更优选为10~40重量份。在上述范围时,可维持组合物的物理性质和聚合物的机械强度的同时,能够控制药剂释放速度等。例如,涂覆在医疗器具上时不发生涂覆层脱离等问题,且能够以适当的速度释放药剂。The amount of the release aid added is 5 to 60 parts by weight, more preferably 10 to 40 parts by weight, based on 100 parts by weight of the above-mentioned organic polymer material. Within the above range, the drug release rate and the like can be controlled while maintaining the physical properties of the composition and the mechanical strength of the polymer. For example, when coated on a medical device, problems such as detachment of the coating layer do not occur, and the drug can be released at an appropriate speed.
·药剂·Pharmacy
被含于本发明的药剂释放控制组合物当中,作为释放控制对象的药剂组分,通常是以医药品和医药部外品为首的药物。但是,根据其用途、目的,药剂除医药品之外还可以是化妆品、农药等。The drug component to be controlled for release contained in the controlled drug release composition of the present invention is generally a drug including pharmaceuticals and quasi-drugs. However, depending on the use and purpose, the pharmaceuticals may be cosmetics, agricultural chemicals, etc. in addition to pharmaceuticals.
对对象药剂无特别限定,对于溶解上述有机高分子材料的有机溶剂可溶解即可。因此,根据所要治疗的效果、药效而适当选择,适当的任意的生理活性药物都可以成为本发明的对象。而且,并不局限于一种药物,可以以多个药物共存的形式使用。例如,在胃溃疡、结核、感冒等的治疗中所采用的并用两种、三种或四种药物的疗法,同时使用多个药物,通过组合而确保相乘效果和相辅作用。The target drug is not particularly limited, as long as it is soluble in the organic solvent in which the above-mentioned organic polymer material is dissolved. Therefore, appropriate arbitrary physiologically active drugs can be selected according to the desired therapeutic effects and drug effects, and can be the object of the present invention. Furthermore, it is not limited to one kind of medicine, and it can be used in the form that a plurality of medicines coexist. For example, in combination therapy of two, three or four drugs used in the treatment of gastric ulcer, tuberculosis, cold, etc., a plurality of drugs are used at the same time, and synergistic and synergistic effects are ensured by combination.
药物的具体例子有抗凝血剂(例如合成抗凝血剂、抗血小板药、抗凝血酶药)、止血剂、血管新生抑制剂、血管辅强剂、防止血管再狭窄的康增生药物、抗血栓症药物或擦伤治疗药剂等。Specific examples of drugs include anticoagulants (such as synthetic anticoagulants, antiplatelet drugs, antithrombin drugs), hemostatic agents, angiogenesis inhibitors, vascular strengthening agents, proliferative drugs for preventing restenosis of blood vessels, Antithrombotic drugs or anti-abrasion agents, etc.
还有抗癌剂、免疫抑制剂、解热镇痛剂、抗炎症剂、镇咳祛痰剂、抗溃疡剂、镇静剂、筋弛缓剂、抗抑郁药、抗癫痫药、抗结核剂、抗心律失常药、血管扩张剂、强心剂、抗过敏剂、降压利尿剂、糖尿病治疗剂、荷尔蒙剂、生理活性肽剂、麻药拮抗剂、骨吸收抑制剂、抗风湿药物、避孕剂、利肝剂、健胃消化剂、整肠剂、维他命剂、疫苗剂、便秘治疗剂、疮治疗剂、各种酶制剂、抗原虫剂、干扰素诱生剂、驱虫剂、外用杀菌消毒剂、寄生虫性皮肤病药、造影剂等。There are also anticancer agents, immunosuppressants, antipyretic analgesics, anti-inflammatory agents, antitussive and expectorant agents, antiulcer agents, sedatives, muscle relaxants, antidepressants, antiepileptic drugs, antituberculosis agents, antiarrhythmic agents Abnormal drugs, vasodilators, cardiotonic agents, antiallergic agents, antihypertensive diuretics, diabetes therapeutic agents, hormone agents, physiologically active peptide agents, anesthetic antagonists, bone resorption inhibitors, antirheumatic drugs, contraceptives, liver agents, Stomach and digestive agent, intestinal regulator, vitamin agent, vaccine agent, constipation treatment agent, sore treatment agent, various enzyme preparations, antiprotozoal agent, interferon inducer, insect repellant, external bactericidal disinfectant, parasitic agent Dermatological drugs, contrast media, etc.
更具体的可适用的药物如下所述,但本发明并不局限于这些例子。药物除了其自身之外还可以是盐或衍生物的形式。More specific applicable drugs are described below, but the present invention is not limited to these examples. The drug may be in the form of a salt or a derivative other than itself.
抗凝血剂有肝素钠、柠檬酸钠等。作为低分子合成抗凝固剂,抗凝血酶药的阿加曲班、和抗血小板药的盐酸沙格雷酯表现血液适合性。作为血管新生抑制剂有烟曲霉素(Fumagillin)、烟曲霉醇衍生物、新生抑制类固醇,作为止血剂有凝血酶、促凝血酶原激酶、乙酰甲萘醌、亚硫酸氢钠甲萘醌、氨甲环酸、ε-氨基已酸、肾上腺色素单氨基胍甲基磺酸盐(adrenochrome monoaminoguanidine methane sulfonate)、卡络磺钠等。Anticoagulants include sodium heparin and sodium citrate. As a low-molecular synthetic anticoagulant, argatroban, an antithrombin drug, and sargrel hydrochloride, an antiplatelet drug, exhibit blood suitability. Angiogenesis inhibitors include fumagillin, fumagillin derivatives, and angiogenesis-inhibiting steroids, and hemostatic agents include thrombin, thromboplastin, acetylmenadione, sodium bisulfite menadione, Tranexamic acid, ε-aminocaproic acid, adrenochrome monoaminoguanidine methane sulfonate, sodium carbosulfonate, etc.
抗肿瘤剂有甲氨蝶呤(Methotrexate)、放线菌素D、丝裂霉素C(mitomycinC MMC)、盐酸博来霉素、盐酸道诺霉素、硫硫酸长春碱、硫酸醛基长春碱、阿霉素(Adriamycin)、新制癌菌素(Neocarzinostatin)、氟尿嘧啶、阿糖胞苷、云芝多糖K(Krestin)、血性链球菌制剂(picibanil)、香菇多糖、贝他定(Bestatin)、左旋咪唑、氨甲酰氰丙啶、甘草甜素、顺泊(Cisplastin)、紫杉醇(Paclitaxel)等。Antitumor agents include methotrexate (Methotrexate), actinomycin D, mitomycin C (mitomycinC MMC), bleomycin hydrochloride, daunorubicin hydrochloride, vinblastine sulfate, vinblastine sulfate , Doxorubicin (Adriamycin), Neocarzinostatin (Neocarzinostatin), Fluorouracil, Cytarabine, Coriolus polysaccharide K (Krestin), Blood Streptococcus preparation (picibanil), Lentinan, Betadine (Bestatin), L- Imidazole, carbamide, glycyrrhizin, Cisplastin, Paclitaxel, etc.
免疫抑制剂有雷帕霉素、环孢菌、他克莫司、甲氨蝶呤、硫唑嘌呤、环磷酰胺、肾上腺皮质激素(德萨美松(Dexamethasone)等)、咪唑立宾(Mizoribine)等。Immunosuppressants include rapamycin, cyclosporine, tacrolimus, methotrexate, azathioprine, cyclophosphamide, adrenal corticosteroids (Dexamethasone, etc.), mizoribine (Mizoribine) )wait.
抗生物质有盐酸四环素、盐酸土霉素、盐酸多西环、氢吡四环素、链霉素、新生霉素、新霉素、红霉素、黏杆菌素、洁霉素、盐霉素、尼日利亚菌素(nigericin)、卡那霉素(kanamycin)、吉他霉素(kitasamycin)、泰乐菌素、盐酸呋喃它酮、万古霉素、壮观霉素、瑞斯托霉素、Soymacin、阿米卡星、新霉素(fradiomycin)、西梭霉素、庆大霉素、卡内多霉素(kanendomycin)、盐酸地贝卡星(Dibekacin)、利维霉素、妥布霉素、氨苄青霉素、阿莫西林、替卡西林、哌拉西林、比噻孢菌素(Cefaloridine)、头孢菌素、头孢磺啶(Cefsulodin)、头孢替安(Cefotiam)、头孢甲肟、先锋美他醇(Cefmetazole)、头孢唑啉(cefazolin)、头孢噻肟、头孢哌酮、头孢唑肟、Moxolactam、磺酰胺菌素(sulfazecin)、氨曲南、Thienamycin、甲硝唑(metronidazole)、克拉霉素等。Antibiotics include tetracycline hydrochloride, oxytetracycline hydrochloride, doxycycline hydrochloride, hydropyridine, streptomycin, novobiocin, neomycin, erythromycin, colistin, lindamycin, salinomycin, nigeria Nigericin, kanamycin, kitasamycin, tylosin, furaltadone hydrochloride, vancomycin, spectinomycin, ristocetin, Soymacin, amikacin , neomycin (fradiomycin), sisomycin, gentamicin, kanendomycin (kanendomycin), dibekacin hydrochloride (Dibekacin), rivamycin, tobramycin, ampicillin, a Moxicillin, Ticarcillin, Piperacillin, Cefaloridine, Cephalosporin, Cefsulodin, Cefotiam, Cefmenoxime, Cefmetazole, Cefazolin, cefotaxime, cefoperazone, ceftizoxime, Moxolactam, sulfazecin, aztreonam, Thienamycin, metronidazole, clarithromycin, etc.
解热镇痛消炎剂有水杨酸钠、斯尔比林、双氯芬酸钠、氟灭酸钠、吲哚美辛钠、氢氯酸吗啡、盐酸哌替啶、氧化吗啡、酒石酸左啡诺等。Antipyretic, analgesic and anti-inflammatory agents include sodium salicylate, sulpyrine, diclofenac sodium, flufenamic acid sodium, indomethacin sodium, morphine hydrochloride, pethidine hydrochloride, oxymorphine, levorphanol tartrate, etc.
镇咳祛痰剂有盐酸麻黄碱、盐酸甲基麻黄碱、盐酸那可丁、磷酸可待因、磷酸双氢可待因、盐酸氯苯达诺、Aroclamide Hydrochloride、盐酸匹考哌林、盐酸氯哌丁、异丙基肾上腺素、盐酸普罗托醇、硫酸沙丁胺醇、硫酸特布他林(terbutaline sulfate)等。Antitussive and expectorant agents include ephedrine hydrochloride, methylephedrine hydrochloride, narcodine hydrochloride, codeine phosphate, dihydrocodeine phosphate, clophenedanol hydrochloride, aroclamide hydrochloride, picoperine hydrochloride, chlorochloride hydrochloride Peridine, isoproterenol, protopol hydrochloride, albuterol sulfate, terbutaline sulfate, etc.
抗溃疡剂有盐酸组氨酸、甲氧氯普胺等,镇静剂有丙氯拉嗪、盐酸氯丙嗪、甲哌氟丙嗪、硫酸阿托品、溴化甲基天冬氨酸等,筋弛缓剂有溴化双哌雄双酯、氯化筒箭毒碱、甲磺酸普立地诺等,抗抑郁药有丙咪嗪、氯米帕明、诺昔替林、硫酸苯乙基肼等,抗癫痫药有盐酸氯氮卓、乙酰唑胺钠、苯妥英钠、乙琥胺等。Anti-ulcer agents include histidine hydrochloride, metoclopramide, etc., sedatives include prochlorperazine, chlorpromazine hydrochloride, flupromazine, atropine sulfate, brominated methylaspartic acid, etc., tendon relaxants There are bipisteronide bromide, myostatin chloride, primidinol mesylate, etc. Antidepressants include imipramine, clomipramine, noxitriptyline, phenethylhydrazine sulfate, etc. Epilepsy drugs include chlordiazepoxide hydrochloride, acetazolamide sodium, phenytoin sodium, and ethosuximide.
糖尿病治疗剂有盐酸苯乙双胍、格列嘧啶钠、格列吡嗪等,抗结核剂有对氨基水杨酸钠、盐酸乙胺丁醇、异烟肼,抗心律失常剂有盐酸普萘洛尔、盐酸阿普洛尔、盐酸布非洛尔、心得平盐酸盐(oxprenololhydrochloride)等,血管扩张剂有盐酸硫氮卓酮、盐酸奥昔非君、盐酸妥拉唑林、克冠二胺、硫酸巴美生等,强心剂有氨茶碱、Theophyllol、盐酸依替福林、Trans-pi-oxocamphor等。抗过敏剂有马来酸氯苯那敏、盐酸甲氧那明、盐酸苯海拉明、盐酸曲吡那敏、盐酸甲地嗪、盐酸氯咪唑、盐酸甲氧那明、盐酸双苯比拉林等,降压利尿剂有五吡咯烷、六甲溴铵、盐酸美加明、盐酸乙肼莱哒嗪、盐酸可乐定等。Diabetes treatment agents include phenformin hydrochloride, glipizidine sodium, glipizide, etc., anti-tuberculosis agents include sodium p-aminosalicylate, ethambutol hydrochloride, isoniazid, and antiarrhythmic agents include propranol hydrochloride Diltiazem hydrochloride, alprenolol hydrochloride, bufemolol hydrochloride, oxprenolol hydrochloride, etc., vasodilators include diltiazem hydrochloride, oxifadrine hydrochloride, tolazoline hydrochloride, keguandiamine , Permexan Sulfate, etc. Cardiotonic agents include aminophylline, Theophyllol, etiforine hydrochloride, Trans-pi-oxocamphor, etc. Antiallergic agents include chlorpheniramine maleate, methoxyphenamine hydrochloride, diphenhydramine hydrochloride, tripyramine hydrochloride, medizine hydrochloride, clemizole hydrochloride, methoxyphenamine hydrochloride, dipheniramine hydrochloride Lin et al. Antihypertensive diuretics include pentapyrrolidine, hexamethylammonium bromide, mecamylamine hydrochloride, hydrazine hydrochloride, clonidine hydrochloride, etc.
荷尔蒙剂有泼尼松龙磷酸钠、泼尼松琥珀酸、地塞米松硫酸钠、倍他米松磷酸钠、醋酸己烯雌酚、磷酸己烷雌酚、甲硫咪唑等。Hormone agents include prednisolone sodium phosphate, prednisone succinic acid, dexamethasone sodium sulfate, betamethasone sodium phosphate, diethylstilbestrol acetate, hexestrol phosphate, and methimazole.
麻药拮抗剂有盐酸烯丙吗啡、盐酸纳洛酮、酒石酸莱瓦洛芬等,骨吸收抑制剂有(含硫烷基)氨基甲烯基磺酸等。Narcotic antagonists include alomorphine hydrochloride, naloxone hydrochloride, levalprofen tartrate, etc., and bone resorption inhibitors include (sulfur-containing alkyl) aminomethanesulfonic acid, etc.
作为生理活性肽类,只要具有生理活性则寡肽或聚肽都可以,并无特别限定。优选分子量约为200~80,000。具体例子有黄体形成荷尔蒙释放荷尔蒙或其衍生物,胰岛素、生长抑素或其衍生物,成长荷尔蒙,催乳素,肾上腺皮质刺激素,甲状腺刺激激素,黑素细胞刺激素、副甲状腺荷尔蒙、加压素(vasopressin)、催产素(Oxytocin)、降血钙素、胰高血糖素、人胃泌素、醋酸胰泌素、胆囊收缩素、促胰酶素、血管紧张素、脑啡肽、蛋白质合成刺激肽、人绒毛膜促性腺激素、人胎盘生成素、黄体生成素、促卵泡激素、各型的干扰素、白细胞介素、内啡肽、京都啡肽(kyotorphin)、血清促吞噬素(Tuftsin)、胸腺生成素、胸腺肽、胸腺刺激素(thymostimulin)、胸腺因子、肿瘤坏死因子,集落刺激因子、神经生长因子、神经肽P物质、舒血管素、胃动素、强啡肽、蛙皮素、黑蛙素、舒缓激肽、门冬酰胺酶、尿激酶、氯化溶菌酶、多粘菌素(Polymyxin B)、粘菌素、短杆菌肽、杆菌肽、红细胞生成素、血小板衍生生长因子,生长荷尔蒙的释放因子,上皮生长因子等。The physiologically active peptides are not particularly limited as long as they are oligopeptides or polymeric peptides, as long as they have physiological activity. Preferably the molecular weight is about 200 to 80,000. Specific examples are luteinizing hormone-releasing hormone or its derivatives, insulin, somatostatin or its derivatives, growth hormone, prolactin, cortico-stimulating hormone, thyroid-stimulating hormone, melanocyte-stimulating hormone, parathyroid hormone, vasopressor vasopressin, oxytocin, calcitonin, glucagon, human gastrin, secretin acetate, cholecystokinin, trypsin, angiotensin, enkephalin, protein synthesis Stimulating peptide, human chorionic gonadotropin, human placental hormone, luteinizing hormone, follicle-stimulating hormone, various types of interferon, interleukin, endorphin, kyotorphin, serum tuftsin (Tuftsin) ), thymopoietin, thymosin, thymostimulin (thymostimulin), thymus factor, tumor necrosis factor, colony-stimulating factor, nerve growth factor, neuropeptide substance P, vasodilator, motilin, dynorphin, bombesin , melanin, bradykinin, asparaginase, urokinase, lysozyme chloride, polymyxin B, colistin, gramicidin, bacitracin, erythropoietin, platelet-derived growth factor , growth hormone release factor, epithelial growth factor, etc.
造影剂有碘类X射线造影剂(碘克沙醇、碘帕醇、伊索显等)、MRI造影剂(钆化合物)、超音波造影剂(Echovist,Levovis),近红外荧光造影剂(吲哚菁绿类化合物)等。Contrast agents include iodine-based X-ray contrast agents (iodixanol, iopamidol, Aesophan, etc.), MRI contrast agents (gadolinium compounds), ultrasound contrast agents (Echovist, Levovis), near-infrared fluorescent contrast agents (ind Docyanine green compounds), etc.
医疗用药之外,还可以是各种化妆品(雪花膏、乳液、润肤膏、染睫毛油、护发剂、美白剂等)、农药(抗菌剂、除草剂、杀虫剂等,具体如日本专利特开平7-330629号公报)、植物生长激素、植物荷尔蒙、昆虫荷尔蒙等的药物。In addition to medical drugs, it can also be various cosmetics (creams, lotions, body creams, mascara, hair conditioners, whitening agents, etc.), pesticides (antibacterial agents, herbicides, insecticides, etc., specifically as in Japanese patents Japanese Patent Laid-Open Publication No. 7-330629), plant growth hormones, plant hormones, insect hormones, etc.
药剂的释放速度是根据血中或组织中的表现药效的最低浓度设定,因此应个药分别地分析,同样地,将释放时间设定成多少也应考虑患者个人的信息、病情、治疗目的、处理内容等。因此,药剂的添加量是无法划一决定的,考虑到药效和成本,通常是相对于100重量份的上述有机高分子材料,在1~150重量份,优选为5~70重量份,更有选为10~60重量份的范围内进行添加。在上述范围内时,可将药剂的溶解性和副作用控制为最低的同时,能够表现最大的药效。The release rate of the drug is set according to the lowest concentration of the drug in the blood or tissue, so each drug should be analyzed separately. Similarly, the release time should also be set in consideration of the patient's personal information, condition, and treatment. Purpose, processing content, etc. Therefore, the addition amount of the medicament cannot be uniformly determined. Considering the efficacy and cost, it is usually 1 to 150 parts by weight, preferably 5 to 70 parts by weight, and more It is selected and added within the range of 10 to 60 parts by weight. When it is within the above range, the solubility and side effects of the drug can be controlled to the minimum, and at the same time, the maximum drug effect can be exhibited.
·其它添加物·Other additives
本发明的药剂释放控制组合物为含有上述有机高分子材料、药剂和释放助剂,并适用于下述医疗器具的组合物。根据需要本组合物还可以含有细胞粘连性物质、或医疗器具表面的内皮化促进物质。The drug release controlled composition of the present invention is a composition containing the above-mentioned organic polymer material, a drug, and a release aid, and is suitable for use in the following medical devices. If necessary, the composition may contain a cell-adhesive substance or an endothelialization-promoting substance on the surface of a medical device.
作为细胞粘连性物质例如有胶原蛋白、纤维粘连蛋白、玻璃粘连蛋白、层粘连蛋白等。Examples of cell-adhesive substances include collagen, fibronectin, vitronectin, and laminin.
内皮化促进物质为,在适用于下述医疗器具尤其是使用在血管系中的后述支架上时,促进在留置后的较早阶段使内皮细胞游走、固定、增生于支架表面的物质。此类内皮化促进物质有细胞粘连性的低聚肽等。The endothelialization-promoting substance is a substance that promotes the migration, fixation, and proliferation of endothelial cells on the surface of the stent at an early stage after indwelling when it is applied to the following medical devices, especially the stent described later in the vascular system. Such endothelialization-promoting substances include cell-adhesive oligopeptides and the like.
覆盖血管内膜最内层的内皮细胞不仅具有覆盖血管内壁的作用,还发挥抗血栓、修复等血管·血流的平常性维持,血管新生、各种因子和调节物质的产生分泌等多种功能。血管内皮细胞不仅涉及对血管内壁损伤的治疗过程,还涉及所谓的血管新生,不管何种情况都经过如下过程,即发生向损伤部位的蛋白质、巨噬细胞等活体组分的移动、游走、固定,接着发生平滑肌细胞、内皮细胞的游走、固定、增生。The endothelial cells that cover the innermost layer of the vascular intima not only have the function of covering the inner wall of the blood vessel, but also perform various functions such as antithrombotic, repair, normal maintenance of blood vessels and blood flow, angiogenesis, production and secretion of various factors and regulatory substances, etc. . Vascular endothelial cells are not only involved in the healing process of damage to the inner wall of blood vessels, but also involved in the so-called angiogenesis. Regardless of the situation, the process of moving, migrating, migrating, and Fixation, followed by migration, fixation and proliferation of smooth muscle cells and endothelial cells.
若注意这类内皮细胞的举动,则可以发现要想避免再狭窄、再闭塞的主要原因,即活体对支架的排异,需要在处理后较早的阶段使血管内细胞游走、固定、增生在支架表面。若发生这种内皮化,在支架上迅速形成模拟血管内壁的状态,则该支架不易被活体排异,免疫·异物排除功能将不会启动。即,不易发生引起炎症反应的单球、巨噬细胞向支架留置部位的游走。为了促进支架表面的内皮化,上述组合物也可含有内皮化促进物质。If you pay attention to the behavior of these endothelial cells, you can find that in order to avoid the main cause of restenosis and reocclusion, that is, the rejection of the living body to the stent, it is necessary to allow the cells in the blood vessel to migrate, fix, and proliferate at an early stage after treatment. on the support surface. If such endothelialization occurs, the stent will quickly form a state that simulates the inner wall of the blood vessel, and the stent will not be easily rejected by the living body, and the immune and foreign body exclusion functions will not be activated. That is, migration of monocytes and macrophages that cause inflammatory reactions to the stent placement site is less likely to occur. In order to promote the endothelialization of the surface of the stent, the composition may contain an endothelialization promoting substance.
并且,根据需要还可以含有在制剂技术上通常使用的粘接剂、可溶化剂、乳化剂、稳定剂等。用于制剂的辅助剂、添加剂的选择和其含量可根据上述有机高分子材料和药剂、适用本组合物的医疗器具而适当决定。In addition, binders, solubilizers, emulsifiers, stabilizers, etc. that are generally used in formulation technology may be contained as needed. The selection and content of adjuvants and additives used in formulations can be appropriately determined according to the above-mentioned organic polymer materials and pharmaceuticals, and medical devices to which the present composition is applied.
药剂释放性医疗器具Drug releasing medical device
随着医工学的进步,已经有主要以诊断和治疗为目的,通过在活体内外结合、埋入或留置某种医疗用具、器件或装置等而达到所期望的目的的技术。本发明的药剂释放性医疗器具涉及这种技术,是保持上述组合物将其与活体接触、或结合或留置于活体内的医疗器具。对保持上述组合物的药剂释医疗器具无特别限定。适用上述组合物的器具的对象通常为在医疗领域使用的医疗器具,而在实际中是根据医疗现场的需要性而确定。With the advancement of medical engineering, there have been technologies that are mainly for the purpose of diagnosis and treatment, and achieve the desired purpose by combining, embedding or indwelling certain medical appliances, devices or devices inside and outside the living body. The drug-releasing medical device of the present invention relates to such technology, and is a medical device that holds the above-mentioned composition in contact with a living body, binds it, or indwells it in a living body. There are no particular limitations on the drug-releasing medical device holding the above-mentioned composition. The devices to which the above-mentioned composition is applied are usually medical devices used in the medical field, but are actually determined according to the needs of the medical field.
在此所述的医疗器具包括所谓的“医疗用具”。具体例子有体内-体外连接用的各种导管、点滴组,完全用于体内的支架、线夹、缝合器(stapler)、止血具、缝合线、骨折固定具、起搏器、器官代替用医疗用具(人造血管、人造气管、人造阀、人造晶体、人造骨、人造关节等)、人造器官(人造皮肤、人造乳房、人造肺、人造心脏等)等,用于体表附近的创伤包覆材料、隐形眼镜、嵌体、人造齿根、牙冠、植牙床、修复用的合成树脂、牙科用CTR材料等。还包括生物感知器(如胶囊型探知器的胶囊内视镜)、埋入型放射线放射源等。The medical devices described herein include so-called "medical devices". Specific examples include various catheters and drip sets for internal and external connections, stents, wire clips, staplers, hemostats, sutures, fracture fixation devices, pacemakers, and medical devices for organ replacement. Appliances (artificial blood vessels, artificial trachea, artificial valves, artificial crystals, artificial bones, artificial joints, etc.), artificial organs (artificial skin, artificial breasts, artificial lungs, artificial hearts, etc.), etc., used for wound dressing materials near the body surface , contact lenses, inlays, artificial tooth roots, crowns, implant beds, synthetic resins for restoration, CTR materials for dentistry, etc. It also includes biosensors (such as capsule endoscopes for capsule detectors), embedded radiation sources, and the like.
本发明的药剂释放性医疗器具保持本发明的药剂释放控制组合物,凭此将该药剂释放于体内规定部位。即,当药剂释放性医疗器具被结合、或留置于规定的体内部为或体表面部位时,释放所保持的药剂,而其时机和释放速度、释放量和释放时间已被调节。保持的形态因医疗器具的种类、用途等而变化,例如可以是通过涂布及喷雾形成的涂覆,以及向孔内的内包、抱合、结合、粘接、固定,以及搭载本发明组合物的药物传递用薄膜或带状物的缠绕等各种适用形态,并无特别限定。最简便的方法为,在医疗器具的表面形成所述组合物的层,因其表面变成机械性表面,所以适用范围广泛。The drug-releasing medical device of the present invention holds the drug release controlled composition of the present invention, whereby the drug is released at a predetermined site in the body. That is, when the drug-releasing medical device is combined or left in a predetermined body interior or body surface site, the retained drug is released, and the timing, release speed, release amount, and release time are adjusted. The form of retention varies depending on the type, use, etc. of the medical device. For example, coating by coating and spraying, and inclusion, entanglement, bonding, adhesion, and fixation into holes, and loading of the composition of the present invention may be possible. There are no particular limitations on various applicable forms such as winding of a film or tape for drug delivery. The easiest method is to form a layer of the composition on the surface of a medical device, and since the surface becomes a mechanical surface, it can be applied in a wide range of fields.
将本发明的药剂释放控制组合物保持于目的医疗器具的方法有,进行向该组合物溶液浸泡医疗器具后去除溶剂,或将该溶液喷到医疗器具的表面之后去除溶剂,或将该溶液涂布到医疗器具之后去除溶剂等之后,将药剂释放控制组合物以层状接合、固定于医疗器具的表面。对医疗器具实施这种涂覆时,涂覆层的厚度为1~数千nm,优选数十~数百nm的范围内。越薄包层的剥离越少。若不是涂覆而是将本发明的组合物以单体埋入医疗器具时,其厚度不会有问题,还可以选择板状、球状、棒状等的任意形式。The method of holding the drug release controlled composition of the present invention on the intended medical device includes removing the solvent after soaking the medical device in the composition solution, spraying the solution on the surface of the medical device and then removing the solvent, or applying the solution to the medical device. After the solvent and the like are removed after application to the medical device, the composition for controlling drug release is bonded and fixed to the surface of the medical device in a layered form. When such a coating is applied to a medical device, the thickness of the coating layer is in the range of 1 to several thousand nm, preferably tens to hundreds of nm. The thinner the cladding, the less the peeling. When the composition of the present invention is embedded in a medical device as a single body instead of coating, there is no problem with its thickness, and any form such as a plate shape, a spherical shape, or a rod shape can be selected.
作为本发明的药剂释放性医疗器具,根据每个医疗器具,其适用的形态不同,所以在此并不一一全部例举,作为代表例子有,包括支架在外科领域上的应用或包括嵌体(inlay)在牙科治疗中的应用。优选为支架、导管、线夹、胶囊检测器、器官替代用的医疗器具、或人造器官。As the drug-releasing medical device of the present invention, the applicable form varies depending on each medical device, so not all of them are listed here. Representative examples include the application of stents in the field of surgery or inlays. (inlay) application in dental treatment. Preferably, it is a stent, a catheter, a wire clip, a capsule detector, a medical device for organ replacement, or an artificial organ.
具体实施方式 Detailed ways
作为优选适用本发明的药剂释放控制组合物的医疗器具可例举支架。因此,下面作为本发明具体应用的例子,对向支架赋予药剂缓释性方式和这种支架进行说明。A stent is preferably exemplified as a medical device to which the drug release controlled composition of the present invention is applied. Therefore, as an example of the specific application of the present invention, a method of imparting drug sustained release to a stent and such a stent will be described below.
作为心肌梗塞主因之一的心脏冠动脉闭塞症等的治疗方法,大多采用血管成形术。该方法主要是通过球囊扩张而确保血管流路,以及利用激光切除的血管成形术,已有多个良好的治疗成绩。另一方面,处理后发生血管的再狭窄、再闭塞的比率高达40~50%,这也正是该术法的问题点。Angioplasty is often used as a treatment method for coronary artery occlusive disease, which is one of the main causes of myocardial infarction. This method mainly uses balloon dilation to secure blood vessel flow, and angioplasty using laser ablation, and has many good therapeutic results. On the other hand, the rate of restenosis and reocclusion of blood vessels after treatment is as high as 40 to 50%, which is also the problem of this technique.
对于再狭窄、再闭塞等物理问题,已尝试了给药、重新插入和扩张球囊导管、或者激光处理等,但都不能算是根本性的解决方法,也对患者带来极大的痛苦和压力。针对于此,使用了留置血管内部的支架。支架能够补充切开部分的同时防止血管的收缩,有效降低动脉闭塞症患者的复发概率。For physical problems such as restenosis and reocclusion, drug administration, reinsertion and expansion of balloon catheters, or laser treatment have been tried, but none of them can be regarded as fundamental solutions, and they also bring great pain and pressure to patients . For this purpose, a stent placed inside the blood vessel is used. The stent can supplement the cut part while preventing the constriction of blood vessels, effectively reducing the recurrence probability of patients with arterial occlusive disease.
血管用支架是由金属材料或高分子材料构成的管状的小器件医疗器具。用其处理闭塞血管的典型的处理方法的代表例子如下:经由插入到血管内腔的球囊导管将血管用支架留置到血管闭塞部位内。接着,通过膨胀球囊将该支架的直径非可逆地扩大,或留置在动脉血管中后用磁气诱导方式加热等某种方法使支架自己扩张,进而确保血管的开通性。通过这种方法来长期维持良好的血流。A stent for a blood vessel is a tubular small device medical device made of a metal material or a polymer material. A representative example of a typical treatment method for treating an occluded blood vessel is as follows: a stent for a blood vessel is placed in a vascular occlusion site via a balloon catheter inserted into the lumen of the blood vessel. Next, expand the diameter of the stent irreversibly by inflating a balloon, or indwell in the arterial vessel and heat it by magnetic induction to expand the stent itself, thereby ensuring the patency of the blood vessel. This method is used to maintain good blood flow in the long term.
至今为止,已开发有各种关于治疗心脏冠动脉闭塞症为首的动脉血管闭塞症用的支架的材料、形状、以及手术方法。但是,因现有的材料仍然无法完全避免再狭窄、再闭塞的危险,进而成为使用支架进行血管成形术的瓶颈。因此,医疗现场非常需要再狭窄、再闭塞的危险少的支架。作为上述药剂释放性医疗器具的优选实施方式应用于支架时,药剂可使用抗凝固剂、抗癌剂、免疫抑制剂等。向支架赋予药剂释放功能尤其是赋予药剂缓释性的方法可任选,(1)在支架表面适用(涂覆、埋入等)含药剂的组合物的方法,(2)将搭载释放或缓释用的药剂,必要时还搭载释放助剂的载体,涂覆到支架表面的方法。在(1)的方式中,优选使用含有有机高分子材料和药剂的上述药剂释放控制组合物。在(2)的方法中,覆盖支架表面的高分子材料中搭载有缓释用的药剂等。So far, various materials, shapes, and surgical methods of stents for treating coronary artery occlusive disease including coronary artery occlusive disease have been developed. However, the existing materials still cannot completely avoid the risk of restenosis and reocclusion, which has become the bottleneck of angioplasty using stents. Therefore, a stent with less risk of restenosis and reocclusion is strongly required in the medical field. When a preferred embodiment of the drug-releasing medical device described above is applied to a stent, anticoagulants, anticancer agents, immunosuppressants, and the like can be used as drugs. The method of imparting drug release function to the stent, especially the sustained release of the drug, is optional, (1) the method of applying (coating, embedding, etc.) a composition containing the drug on the surface of the stent, (2) loading the release or sustained release A method in which the drug to be released and, if necessary, a carrier carrying a release aid is coated on the surface of the stent. In the aspect (1), it is preferable to use the above-mentioned controlled drug release composition containing an organic polymer material and a drug. In the method (2), the polymer material covering the surface of the stent is loaded with a drug for sustained release or the like.
具体的合成抗凝固剂优选为盐酸沙格雷酯和阿加曲班。含这种药剂的组合物涂覆在支架表面作为包膜层。该药剂以所需的释放速度从这种药剂释放性支架的表面向血中或血管壁持续被释放。本发明的组合物因药剂释放速度高,所以从留置支架的初期开始以能够表现抗凝固剂药效的充分量进行释放。Specific synthetic anticoagulants are preferably sargrelate hydrochloride and argatroban. A composition containing this agent is coated on the surface of the stent as a coating. The drug is continuously released from the surface of the drug-releasing stent to the blood or the blood vessel wall at a desired release rate. Since the composition of the present invention has a high drug release rate, it is released in a sufficient amount to express the drug effect of the anticoagulant from the initial stage of the indwelling stent.
支架只要具有上述的特征,就可采用任意的结构、形态、材质、尺寸或状态,本发明的各种适用、应用对本领域人员来说比较容易。因此,上述支架适用于所有的以防止脉管(血管、淋巴管、胆管、尿管、气管等)的再狭窄、再闭塞为目的的场合。As long as the stent has the above-mentioned features, it can adopt any structure, shape, material, size or state, and various applications and applications of the present invention are relatively easy for those skilled in the art. Therefore, the above-mentioned stent is suitable for all occasions where the aim is to prevent restenosis and reocclusion of blood vessels (blood vessels, lymphatic vessels, bile ducts, urinary tracts, trachea, etc.).
本发明中的支架的特征为,从表面缓释阿加曲班(抗凝血酶药)或盐酸沙格雷酯(抗血小板药)或者上述俩者的药剂。优选为向涂覆在构成该支架的金属表面的高分子材料中,或者多孔性支架基材上搭载上述缓释药剂。本发明的支架优选使用于冠动脉狭窄的治疗。The stent of the present invention is characterized in that argatroban (antithrombin drug) or sargrelate hydrochloride (antiplatelet drug) or both of the above-mentioned agents are slowly released from the surface. Preferably, the above-mentioned sustained-release agent is carried on a polymer material coated on the metal surface constituting the stent, or on a porous stent substrate. The stent of the present invention is preferably used in the treatment of coronary artery stenosis.
支架bracket
本发明的支架的材质、构造,除了实施下述的表面处理之外,事实上可以是任意设计的支架。这表明在保持各种支架的特征及功能的同时,通过本发明还可以事先防止上述再狭窄、再闭塞的发生。The material and structure of the stent of the present invention may be a stent of any design except that the surface treatment described below is carried out. This shows that the present invention can prevent the above-mentioned restenosis and reocclusion in advance while maintaining the characteristics and functions of various stents.
支架可以是插入血管内前后不发生形状变化的,也可以是球囊扩张型、自己扩张型、及其组合。本发明的支架所使用的材料只要是具有能够实现其设计的物理性的材料,可使用任何材料。具体的有一直被使用的金属材料如不锈钢、钴/铬合金、钽、钛、钨、铂、钴及它们的合金等。The stent may not change in shape before and after being inserted into the blood vessel, or may be balloon-expandable, self-expandable, or a combination thereof. Any material can be used as the material used for the stent of the present invention as long as it has physical properties that can realize its design. Specifically, there are metal materials that have been used such as stainless steel, cobalt/chrome alloy, tantalum, titanium, tungsten, platinum, cobalt and their alloys.
使用金属以外的材料时,如后述,使用适合本发明目的的,即该材料能够搭载抗凝固剂。适合所述要求的材料有PET(聚对苯二甲酸乙二酯)、PBT(聚对苯二甲酸丁二酯)、聚碳酸酯、聚乙烯、聚丙烯、聚乙缩醛、聚苯乙烯等。生物降解性高分子优选为聚乳酸、聚乙醇酸、聚苹果酸及它们的聚合物、聚己内酯等的聚羟酯类。When a material other than metal is used, as will be described later, it is suitable for the purpose of the present invention, that is, the material can carry an anticoagulation agent. Materials suitable for the stated requirements include PET (polyethylene terephthalate), PBT (polybutylene terephthalate), polycarbonate, polyethylene, polypropylene, polyacetal, polystyrene, etc. . The biodegradable polymer is preferably polyhydroxyesters such as polylactic acid, polyglycolic acid, polymalic acid and their polymers, and polycaprolactone.
本发明的支架优选使用金属材料,其形状可以是筒状、波纹状、具有弯曲处的结构、网状、钢丝状的固体成型物,只要留置在血管中后不发生对强度和血管壁的损伤的问题,就可以采取各种各样的形状。The metal material is preferably used for the stent of the present invention, and its shape can be cylindrical, corrugated, structure with bends, mesh, steel wire-like solid molding, as long as it does not damage the strength and blood vessel wall after being placed in the blood vessel Questions can take a variety of shapes.
抗血凝固药剂Anticoagulants
从本发明的支架表面至少释放阿加曲班(抗凝血酶药)和盐酸沙格雷酯(抗血小板药)中的一种药剂。为此,阿加曲班或盐酸沙格雷酯或者两者的合成抗凝固药剂被搭载于,涂覆在构成该支架的金属表面的高分子材料中。At least one of argatroban (antithrombin drug) and sargrelate hydrochloride (antiplatelet drug) is released from the surface of the stent of the present invention. For this purpose, synthetic anticoagulant agents of argatroban or sargrelate hydrochloride or both are carried in, and coated on, a polymer material constituting the metal surface of the stent.
作为抑制血液凝固的药剂之一,本发明所使用的抗凝血酶药的阿加曲班为,具有下式所表示的化学结构的精氨酸衍生物类合成抗凝血酶药。阿加曲班的3支链结构通过与凝血酶的活性部位立体结合而高度抑制凝血酶的主要作用,即生成纤维朊的作用、因子XIII的活化引起的纤维朊的稳定化作用、血小板凝聚作用,从而起到抗凝血酶药作用。如上述,因直接作用于凝血酶,所以其对个人的差别比肝素(heparin)还低还确实,作用表现也迅速。并且没有天然阻止物,分子量也小,所以对结合了纤维朊的凝血酶也具有作用,可确实地阻止血栓的形成。而且,对肝素无法预防的、高剪断应力下所形成的白血栓也具有抑制作用。Argatroban, which is an antithrombin drug used in the present invention, is one of drugs that inhibit blood coagulation, and is an arginine derivative-based synthetic antithrombin drug having a chemical structure represented by the following formula. The three-branched structure of argatroban highly inhibits the main effects of thrombin by sterically binding to the active site of thrombin, namely, the generation of fibrin, the stabilization of fibrin caused by the activation of factor XIII, and the aggregation of platelets , thereby acting as an antithrombin drug. As mentioned above, since it acts directly on thrombin, it is more reliable than heparin in terms of individual differences, and its action is also expressed quickly. And there is no natural inhibitor, and the molecular weight is small, so it also has an effect on thrombin bound to fibrin, and can reliably prevent the formation of thrombus. Moreover, it also has an inhibitory effect on white thrombus formed under high shear stress which cannot be prevented by heparin.
【式1】【Formula 1】
本发明所使用的另一血液凝固抑制药剂的盐酸沙格雷酯具有抑制血小板活化的作用,其作用机理推测如下:Sargrelate hydrochloride, another blood coagulation inhibitor used in the present invention, has the effect of inhibiting platelet activation, and its mechanism of action is speculated as follows:
粘接凝聚在血管内皮障碍部位上的活性化血小板所释放的5-羟色胺(Serotonin)(5-HT)具有各种药理作用,通过血小板膜上和血管平滑肌细胞膜上的5-HT2受体,增强障碍部位上血小板的凝聚,并且收缩障碍部位血管的同时,增生血管平滑肌,导致末梢循环障碍。安步乐克(Anplag)通过与5-HT2进行选择性的嵌段,而表现抑制血小板凝聚、尤其抑制由于5-羟色胺而增强的血小板凝聚的作用、以及抑制血管收缩的作用。因此盐酸沙格雷酯对包括慢性动脉闭塞症的典型在内的各种血栓典型呈有效性。The 5-hydroxytryptamine (Serotonin) (5-HT) released by the activated platelets that bind and condense on the vascular endothelial barrier has various pharmacological effects. Enhance the aggregation of platelets at the site of the disorder, and shrink the blood vessels at the site of the disorder, while proliferating the smooth muscle of the blood vessels, resulting in peripheral circulation disorders. Anplag exhibits the effect of inhibiting platelet aggregation, especially the effect of inhibiting platelet aggregation enhanced by 5-hydroxytryptamine, and the effect of inhibiting vasoconstriction through selective block with 5-HT 2 . Therefore, sargrelate hydrochloride is effective for various types of thrombosis including those of chronic arterial occlusive disease.
【式2】[Formula 2]
这两种药剂尤其对初期血栓的抑制极为有效,在临床上作为抗凝固疗法以口服剂、或静脉给药的形式得到广泛应用。另一方面,在留置支架后的几个月的短期间内,预防留置部位上的血栓形成导致的冠动脉的闭塞是一个大课题。因此,在血管中设置本发明的支架时,通过从支架缓释这些抗凝固药剂而能够预防血栓形成导致的闭塞。其结果,在留置支架的部位上,可有效抑制血管的再狭窄、再闭塞。These two agents are extremely effective especially in the inhibition of initial thrombus, and are widely used clinically as anticoagulant therapy in the form of oral or intravenous administration. On the other hand, prevention of coronary artery occlusion due to thrombus formation at the indwelling site within a short period of several months after the indwelling stent is a major issue. Therefore, when the stent of the present invention is placed in a blood vessel, it is possible to prevent occlusion due to thrombus formation by slowly releasing these anticoagulant agents from the stent. As a result, restenosis and reocclusion of blood vessels can be effectively suppressed at the site where the stent is placed.
作为缓释阿加曲班的赋予抗血栓性的医疗器具,如上所述有特开平6-292711号公报和特开平6-292718号公报所公开的导管。前者记载了向热塑性高分子材料熔融混炼阿加曲班,成型为导管(catheter tube)的技术,后者记载了将导管浸泡在溶解有阿加曲班的有机溶剂而向该管中浸透阿加曲班的方法。这些技术中,导管基材基本上要使用具有良好机械强度和成型性的材料,可使用的优选材料有链段化尼龙、链段化聚氨酯、链段化聚酯等的晶体性热塑性弹性体材料。As medical devices for imparting antithrombotic properties for sustained release of argatroban, there are catheters disclosed in JP-A-6-292711 and JP-A-6-292718 as described above. The former describes the technology of melting and kneading argatroban into a thermoplastic polymer material to form a catheter (catheter tube), and the latter describes soaking the catheter in an organic solvent in which argatroban is dissolved and soaking the tube with argatroban. The method of adding tunes. In these technologies, the base material of the catheter basically needs to use a material with good mechanical strength and formability, and the preferred materials that can be used include crystalline thermoplastic elastomer materials such as segmented nylon, segmented polyurethane, and segmented polyester. .
·支架中的药剂搭载·Pharmaceutical loading in the stent
本发明的药剂释放型支架搭载有上述抗血液凝固药剂,当其留置于规定的血管内部时,所搭载的药剂在一定时间内被释放。将含有该药剂的上述共聚物向支架上搭载的方法可以是各种适用形态,无特别限定,但优选采用可调整释放时机和释放速度、释放的量和时间的搭载形态。例如有,在构成支架的金属表面上利用雷射钻孔或等离子蚀刻等设置微孔并封入药剂的方法;使用多孔质金属或多孔质无机材料形成支架并向该多孔质部位封入药剂的方法;在构成支架的金属表面上形成含该药剂的高分子层的方法;直接用含药剂的高分子材料制作支架本身的方法;将搭载有药剂的药物传递用薄膜或带状物进行缠绕等方法。其中最简便的是在构成支架的金属表面上形成含该药剂的高分子层的方法。该方法因可以利用现有的支架技术,而且支架表面直接变成功能性表面,所以适用范围广泛。The drug-releasing stent of the present invention carries the above-mentioned anticoagulant drug, and when it is indwelled in a predetermined blood vessel, the carried drug is released within a certain period of time. The method of loading the above-mentioned copolymer containing the drug on the stent can be applied in various forms and is not particularly limited, but it is preferable to adopt a loading form in which the release timing, release rate, release amount, and time can be adjusted. For example, there is a method of providing micropores on the metal surface constituting the stent by laser drilling or plasma etching, and enclosing a drug; a method of forming a stent using a porous metal or a porous inorganic material, and enclosing the drug in the porous part; The method of forming a polymer layer containing the drug on the metal surface constituting the stent; the method of directly using the polymer material containing the drug to make the stent itself; the method of winding the film or ribbon for drug delivery loaded with the drug. Among them, the most convenient method is to form a polymer layer containing the drug on the metal surface constituting the stent. This method has a wide range of applications because it can utilize existing scaffold technology and the surface of the scaffold can be directly transformed into a functional surface.
当上述构成支架的金属表面为多孔体时,将缓释用的上述药剂分散于高分子中后再搭载于该多孔体的孔部分。通过控制多孔性支架基材的孔径并将搭载有药剂的高分子材料保持在该孔中,而能够以所需的释放速度持续释放该药剂。上述多孔性支架基材中的孔径优选为0.01nm~300nm,更优选为0.1nm~100nm。When the metal surface constituting the stent is a porous body, the drug for sustained release is dispersed in a polymer and loaded on the pores of the porous body. By controlling the pore size of the porous stent substrate and holding the drug-loaded polymer material in the pores, the drug can be continuously released at a desired release rate. The pore diameter in the above-mentioned porous stent base material is preferably 0.01 nm to 300 nm, more preferably 0.1 nm to 100 nm.
用于支架涂覆的搭载用高分子材料Mounting polymer materials for stent coating
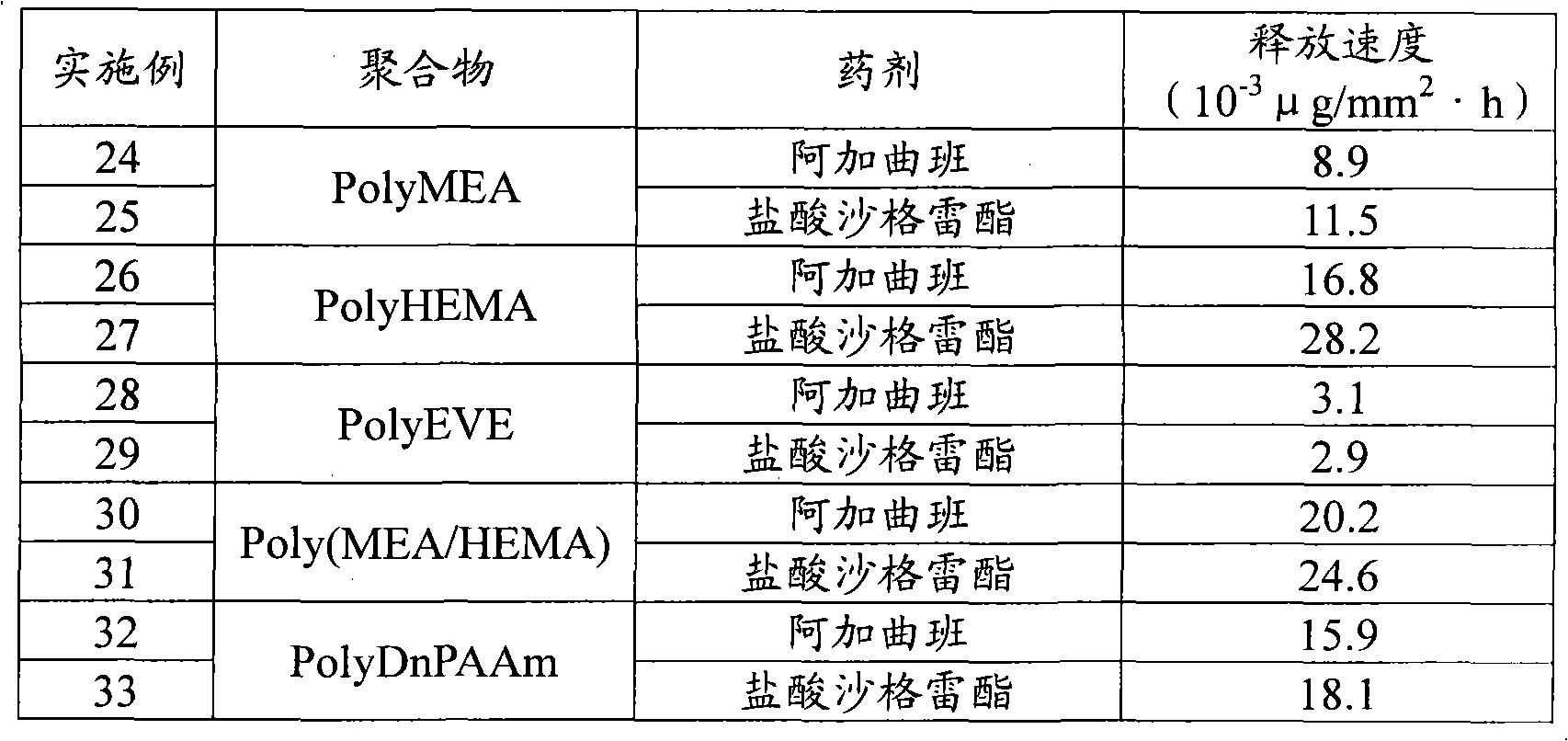
本发明人发现,作为搭载该药剂的高分子材料,药剂和高分子的相溶性对于在一定时间内缓释上述药剂具有重要的作用,尤其是搭载于非晶性高分子更为理想的事实。而且,优选玻璃转化点为体温37℃以下的材料。当将玻璃转化点为37℃以下的材料留置于血管内时,该高分子处于玻璃转化点以上,主链的分子运动性增大,从而促进释放药剂。使用晶体性高分子时,高分子晶体相和药剂相发生明显的相分离,引起药剂相表面偏析的现象。因此会导致药剂一次全部释放、即所谓的猝发释放,其后释放将会大幅度下降。The present inventors found that, as a polymer material carrying the drug, the compatibility between the drug and the polymer plays an important role in the sustained release of the drug over a certain period of time, especially the fact that it is more desirable to carry the drug on an amorphous polymer. Furthermore, a material having a glass transition point of 37° C. or lower in body temperature is preferable. When a material with a glass transition point of 37° C. or lower is placed in the blood vessel, the polymer is above the glass transition point, and the molecular mobility of the main chain increases, thereby promoting the release of the drug. When a crystalline polymer is used, the polymer crystal phase and the drug phase are clearly phase-separated, causing surface segregation of the drug phase. This results in a complete release of the medicament at one time, a so-called burst release, after which the release will be greatly reduced.
另一方面,本发明所用的抗凝固药剂,如其化学式所示具有碱性基团或离子性基团,所以不仅具有亲水性,而且亲油性也比较高,对水的溶解性相当低,而对醇的溶解性高。因此其与疏水性极高的聚烯烃等高分子材料的相容性低,同晶体性高分子材料一样,估计这些材料也会发生由相分离造成的猝发释放和其后溶出速度的急剧降低。例如在(甲基)丙烯酸酯类的高分子材料中,作为其酯残基优选为碳数4以下即甲酯、乙酯、丙酯、丁酯,或者具有能表现亲水性的羟基、甲氧基、环氧乙烷醚基(-(CH2CH2O)-)的烷酯。On the other hand, the anticoagulant agent used in the present invention has a basic group or an ionic group as shown in its chemical formula, so it not only has hydrophilicity, but also has relatively high lipophilicity, and has a relatively low solubility in water, and High solubility in alcohol. Therefore, it has low compatibility with high-molecular materials such as highly hydrophobic polyolefins. Like crystalline polymer materials, it is expected that these materials will also undergo sudden release due to phase separation and a sharp decrease in dissolution rate thereafter. For example, in (meth)acrylic ester polymer materials, the ester residue is preferably methyl ester, ethyl ester, propyl ester, butyl ester with 4 or less carbon atoms, or a hydroxyl group, methyl ester, etc. that can express hydrophilicity. Oxygen, alkyl esters of oxirane ether groups (-(CH 2 CH 2 O)-).
因此作为本发明的涂覆用高分子材料,优选的非晶性高分子材料有聚甲基丙烯酸丁酯、聚甲基丙烯酸乙酯、聚甲基丙烯酸丙酯、聚甲基丙烯酸羟基乙酯等聚甲基丙烯酸烷酯、聚甲基丙烯酸(羟基烷基)酯以及它们的共聚物;聚丙烯酸丁酯、聚丙烯酸乙酯、聚丙烯酸丙酯、聚丙烯酸甲氧基乙酯等聚丙烯酸烷酯以及它们的共聚物;聚碳酸丁烯酯、聚碳酸乙烯酯等脂肪族聚碳酸酯及其共聚物;醋酸乙烯酯、乙烯基吡咯烷酮、部分皂化聚乙烯醇、聚乙烯醚等聚乙烯基化合物及其共聚物;乳酸、乙醇酸为一个组分的生物降解高分子、DL-乳酸-乙醇酸共聚物等,但并不局限于此。Therefore, as the polymer material for coating of the present invention, preferred amorphous polymer materials include polybutyl methacrylate, polyethyl methacrylate, polypropyl methacrylate, polyhydroxyethyl methacrylate, etc. Polyalkyl methacrylate, poly(hydroxyalkyl) methacrylate and their copolymers; polyalkyl acrylate such as polybutyl acrylate, polyethyl acrylate, polypropyl acrylate, polymethoxyethyl acrylate, etc. And their copolymers; aliphatic polycarbonates such as polybutylene carbonate and polyvinyl carbonate and their copolymers; vinyl acetate, vinylpyrrolidone, partially saponified polyvinyl alcohol, polyvinyl ether and other polyvinyl compounds and Copolymers thereof; biodegradable polymers with lactic acid and glycolic acid as a component, DL-lactic acid-glycolic acid copolymer, etc., but not limited thereto.
上述非晶性高分子不同于晶体性高分子,有优良的有机溶剂溶解性,对支架进行涂覆时多个有机溶剂都能成为适用对象,从而增加了技术上的方便性。The above-mentioned amorphous polymers are different from crystalline polymers in that they have excellent solubility in organic solvents, and many organic solvents can be used when coating the stent, thereby increasing the technical convenience.
释放助剂release aid
如上所述,向形成DDS基体的高分子材料添加促进释放用的释放助剂(即“释放助剂”)则可提高药剂缓释速度,对于支架也不例外。因此,在本发明的支架中,应用上述药剂时,当与所述高分子材料组合也不能获得所需释放速度时,通过使用释放助剂而能够获得所希望的释放速度。尤其该高分子材料为具有比体温高的玻璃转化点时更有效,当聚乳酸、乳酸-乙醇酸共聚物等生物降解性高分子材料时,添加释放助剂则使玻璃转化点变低,因此有效。作为该助剂基本上都是脂溶性,但优选为表现一定程度的水溶性的低分子物质。其原因来自对于高分子和药剂两者的相容性。缺乏亲水性的长链脂肪族酯等因缺乏与该药剂的相容性而不优选。而如甘油的水溶性及亲油性都很低的低分子化合物因与高分子材料以及该药剂的相容性低而不优选。本发明优选的释放助剂有选自柠檬酸、酒石酸、苹果酸中的有机酸的酯、或甘油的二酯和单酯(如单乙酸乙酯或二乙酸乙酯等)。其具体例子有如上面所示的酯。As mentioned above, adding a release aid (ie, "release aid") to the polymer material forming the DDS matrix can increase the sustained release rate of the drug, and the stent is no exception. Therefore, in the stent of the present invention, when the above drug is used and the desired release rate cannot be obtained in combination with the polymer material, the desired release rate can be obtained by using a release aid. Especially when the polymer material has a higher glass transition point than body temperature, it is more effective. When polylactic acid, lactic acid-glycolic acid copolymer and other biodegradable polymer materials are used, the glass transition point will be lowered by adding a release aid, so efficient. These additives are basically all fat-soluble, but are preferably low-molecular substances showing some degree of water solubility. The reason for this comes from compatibility with both polymers and pharmaceuticals. Long-chain aliphatic esters lacking in hydrophilicity and the like are not preferable due to lack of compatibility with the agent. On the other hand, low-molecular-weight compounds such as glycerin, which have low water solubility and lipophilicity, are not preferred because of low compatibility with polymer materials and the drug. Preferred release aids of the present invention are esters of organic acids selected from citric acid, tartaric acid, malic acid, or di- and mono-esters of glycerol (such as ethyl monoacetate or ethyl diacetate, etc.). Specific examples thereof include the esters shown above.
这些添加剂可单独使用也可以组合两种以上使用。添加量可以根据药剂的释放速度适当设置,但优选是高分子材料重量的约5~60wt%,更优选10~60wt%的范围内。在该范围内可获取良好的添加效果的同时涂覆层具有充分的机械强度,且涂覆层不易从支架脱落。These additives may be used alone or in combination of two or more. The amount to be added can be appropriately set according to the release rate of the drug, but it is preferably in the range of about 5 to 60 wt%, more preferably 10 to 60 wt%, based on the weight of the polymer material. Within this range, the coating layer has sufficient mechanical strength while obtaining a good additive effect, and the coating layer is not easy to fall off from the stent.
含药剂涂覆层Chemical coating layer
在支架表面形成含有药剂的聚合物层,即含有药剂的上述高分子层的方法有,将包括药剂、高分子材料、必要时包括释放助剂等的其他添加剂溶解在它们可共同溶解的溶剂而获取的溶液,涂布到支架表面的涂布方法;将支架浸泡在该溶液之后取出进行干燥的浸泡方法;将该溶液喷雾到支架表面进而在支架上形成涂层的喷雾涂布法等。其中能够确实进行涂覆的方法为浸泡法,通过该方法可简单地对支架内面、外面的两方进行涂覆。尤其对与血液接触的支架内面进行确实的涂覆处理,则会获取关于赋予抗血栓性,以及降低动脉血管的再闭塞的充分的性能。A method of forming a drug-containing polymer layer, that is, the above-mentioned polymer layer containing a drug, on the surface of a stent is to dissolve other additives including a drug, a polymer material, and a release aid if necessary, in a solvent in which they can be dissolved together. The obtained solution is applied to the surface of the stent; the soaking method is used to soak the stent in the solution and then taken out to dry; the spray coating method is used to spray the solution onto the surface of the stent to form a coating on the stent, etc. Among them, the dipping method is a reliable method for coating, and by this method, both the inner surface and the outer surface of the stent can be easily coated. In particular, by properly coating the inner surface of the stent in contact with blood, sufficient performance can be obtained for imparting antithrombotic properties and reducing reocclusion of arterial vessels.
所形成的涂覆层厚度优选在0.05μm~30μm之间。在该范围内时,能够搭载足够的药剂量,可确保目标时间内的药剂释放,并且,对于伴随心跳而发生的支架变形,具有良好的追踪性,不易出现涂覆层的龟裂和脱落。The thickness of the formed coating layer is preferably between 0.05 μm and 30 μm. Within this range, a sufficient amount of drug can be loaded, and the release of the drug within the target time can be ensured. Moreover, it has good tracking performance for the deformation of the stent accompanying the heartbeat, and the coating layer is less prone to cracking and falling off.
药剂搭载量Drug load
本发明中的支架的药剂搭载量是由药剂的释放速度和所需释放持续时间所决定。猝发性的短时间大量释放使药剂在多时间内枯竭,因此必须要避免这种情况的发生。从预防初期血栓形成的观点出发,释放持续时间优选为数星期~数月期间的持续释放。因此药剂的释放速度为,在将该支架留置后的3周(21天)后的时点上,阿加曲班或盐酸沙格雷酯中的任意一种药剂的溶出时间优选是1×10-3μg/mm2·h~1μg/mm2·h,更优选为1×10-3μg/mm2·h~0.5μg/mm2·h。处于该范围内时,可长期持续抗凝固活性,因而优选。The drug load of the stent in the present invention is determined by the drug release rate and the required release duration. The sudden short-term large-scale release causes the agent to be exhausted over a long period of time, so this situation must be avoided. From the viewpoint of preventing initial thrombus formation, the release duration is preferably sustained release over several weeks to several months. Therefore, the release rate of the medicament is that at the time point after 3 weeks (21 days) after the stent is indwelled, the dissolution time of any one of the medicaments in argatroban or sargrelate hydrochloride is preferably 1×10 − 3 μg/mm 2 ·h to 1 μg/mm 2 ·h, more preferably 1×10 -3 μg/mm 2 ·h to 0.5 μg/mm 2 ·h. When it is within this range, the anticoagulant activity can be sustained for a long period of time, which is preferable.
药剂的释放速度的上限只要不超过毒性量就无特别限定,但由于搭载于支架的药剂搭载量有限,若考虑数百μg为最大量,并需要最低持续释放40天左右的情况,可以推测出实际上的最大速度约为1μg/mm2·h。例如,阿加曲班的从导管溶出的速度约为1.0×10-4~1.0×10-1μg/cm2·分钟,优选为2.5×10-4~7.0×10-3μg/cm2·分钟(人造器官,14(2)、p679~682(1985))。从阿加曲班和盐酸沙格雷酯的血中的有效药效浓度有关的临床认识和上述认识综合考虑,本发明中的阿加曲班和盐酸沙格雷酯的释放速度在上述文献“人造器官”所记载的范围内则比较适当。但是,上述公开内容是针对短期留置为前提的导管的释放速度,而本发明是永久留置,因此必须最低在留置后的3个星期中预防血栓的形成。过了该时间段后,随着内皮细胞等的再生,血栓形成的危险会大幅下降。因此维持刚刚留置后的释放速度之外,留置后3个星期左右时间后的释放速度的保持极为重要。The upper limit of the release rate of the drug is not particularly limited as long as it does not exceed the toxic amount. However, since the amount of the drug loaded on the stent is limited, considering that the maximum amount is several hundred μg and the minimum sustained release is required for about 40 days, it can be inferred that The actual maximum velocity is about 1 μg/mm 2 ·h. For example, the dissolution rate of argatroban from the catheter is about 1.0×10 -4 to 1.0×10 -1 μg/cm 2 ·min, preferably 2.5×10 -4 to 7.0×10 -3 μg/cm 2 · Minutes (Artificial Organs, 14(2), p679-682(1985)). Considering the clinical cognitions related to the effective concentration of argatroban and sargrelate hydrochloride in blood and the above-mentioned cognitions, the release speed of argatroban and sargrelate hydrochloride in the present invention is described in the above-mentioned document "artificial organ "The recorded range is more appropriate. However, the above disclosure is aimed at the release rate of the catheter on the premise of short-term indwelling, but the present invention is for permanent indwelling, so it is necessary to prevent the formation of thrombus within at least 3 weeks after indwelling. After this period of time has elapsed, the risk of thrombus formation is greatly reduced as endothelial cells and the like are regenerated. Therefore, in addition to maintaining the release rate immediately after indwelling, it is extremely important to maintain the release rate after about 3 weeks after indwelling.
实施例Example
通过以下的实施例进一步具体说明本发明,但本发明并不局限于这些实施例。实施例所使用的材料、使用量、浓度、处理时间、处理温度等数值条件、处理方法等仅仅是本发明范围内的优选例子。The present invention is further specifically described by the following examples, but the present invention is not limited to these examples. Numerical conditions such as materials, usage amounts, concentrations, treatment time, treatment temperature, and treatment methods used in the examples are only preferred examples within the scope of the present invention.
[实施例1~3、比较例1~3][Examples 1-3, Comparative Examples 1-3]
如表1所示,将90mg聚乳酸和乳酸/乙醇酸共聚物、作为释放助剂的柠檬酸三乙酯10mg、及抗血小板药的盐酸沙格雷酯10mg,溶解在1mL的六氟异丙醇中,在直径41mm的玻璃皿上流延,进行风干获取药剂载体。将其浸泡在100mL的pH7.4的磷酸缓冲液中,定期采样缓冲液,测定作为盐酸沙格雷酯的特性吸收带的270nm的吸光值(Abs),从而对该药剂的溶出量进行跟踪。溶出开始到3周后的吸光值如表1所示。As shown in Table 1, dissolve 90 mg of polylactic acid and lactic acid/glycolic acid copolymer, 10 mg of triethyl citrate as a release aid, and 10 mg of sarcogrelate hydrochloride as an antiplatelet drug in 1 mL of hexafluoroisopropanol , cast on a glass dish with a diameter of 41 mm, and air-dry to obtain a drug carrier. Soak it in 100 mL of pH 7.4 phosphate buffer, sample the buffer regularly, and measure the absorbance (Abs) at 270 nm as the characteristic absorption band of sarcogrelate hydrochloride, thereby tracking the amount of dissolution of the drug. Table 1 shows the absorbance values from the start of dissolution to 3 weeks after.
作为比较例1~3,除了不添加释放助剂之外,其他与实施例相同的条件进行了同样的溶出试验。As Comparative Examples 1 to 3, the same dissolution test was performed under the same conditions as in the Examples except that no release aid was added.
[表1][Table 1]
[实施例4~6、比较例4][Examples 4 to 6, Comparative Example 4]
将90mg的乳酸/乙醇酸(50/50)共聚物、作为释放助剂的烷基酒石酸二酯10mg、及抗血小板药的盐酸沙格雷酯10mg,溶解在1mL的六氟异丙醇中,在直径41mm的玻璃皿上流延,经风干获取药剂载体。将其浸泡在100mL的pH7.4的磷酸缓冲液中,定期采样缓冲液,测定作为盐酸沙格雷酯的特性吸收带的270nm的吸光值,从而对该药剂的溶出量进行跟踪。溶出开始到3周后的吸光值如表2所示。Dissolve 90 mg of lactic acid/glycolic acid (50/50) copolymer, 10 mg of alkyl tartrate diester as a release aid, and 10 mg of sarcogrelate hydrochloride as an antiplatelet drug in 1 mL of hexafluoroisopropanol. Cast on a glass dish with a diameter of 41mm, and air-dry to obtain the drug carrier. Soak it in 100 mL of pH 7.4 phosphate buffer, sample the buffer regularly, and measure the absorbance at 270 nm, which is the characteristic absorption band of sarcogrelate hydrochloride, so as to track the amount of dissolution of the drug. Table 2 shows the absorbance values from the start of the dissolution to 3 weeks later.
[表2][Table 2]
[实施例7~10、比较例5、6][Examples 7 to 10, Comparative Examples 5 and 6]
如表3所示,将90mg聚乳酸和乳酸/乙醇酸共聚物、作为释放助剂的酒石酸二乙酯或柠檬酸三乙酯10mg、及抗凝血酶药的阿加曲班10mg,溶解在1mL的六氟异丙醇中,在直径41mm的玻璃皿上流延,经风干获取药剂载体。将其浸泡在100mL的pH7.4的磷酸缓冲液中,定期采样缓冲液,测定作为阿加曲班的特性吸收带的330nm的吸光值,从而对该药剂的溶出量进行跟踪。溶出开始到3周后的吸光值如表3所示。As shown in Table 3, 90 mg of polylactic acid and lactic acid/glycolic acid copolymer, 10 mg of diethyl tartrate or triethyl citrate as a release aid, and 10 mg of argatroban as an antithrombin drug were dissolved in Cast in 1 mL of hexafluoroisopropanol on a glass dish with a diameter of 41 mm, and air-dry to obtain the drug carrier. It was soaked in 100 mL of pH 7.4 phosphate buffer solution, the buffer solution was regularly sampled, and the absorbance value at 330 nm, which is the characteristic absorption band of argatroban, was measured to track the dissolution amount of the drug. Table 3 shows the absorbance values from the start of the dissolution to 3 weeks afterward.
作为比较例5、6,除不添加释放助剂之外,其他与实施例相同的条件进行了同样的溶出试验。As Comparative Examples 5 and 6, the same dissolution test was performed under the same conditions as in the Examples except that no release aid was added.
[表3][table 3]
[实施例11~16、比较例7、8][Examples 11 to 16, Comparative Examples 7 and 8]
如表4所示,将90mg聚乳酸和乳酸/乙醇酸共聚物、作为释放助剂的酒石酸二乙酯10~30mg、及抗凝血酶药的阿加曲班10mg,溶解在1mL的六氟异丙醇中,在直径18mm的SUS316L皿上流延,经风干获取药剂载体。将其浸泡在50mL的pH7.4的磷酸缓冲液中,定期采样缓冲液,测定作为阿加曲班的特性吸收带的330nm的吸光值,从而对该药剂的溶出量进行跟踪。溶出开始到7天后的吸光值如表4所示。As shown in Table 4, 90 mg of polylactic acid and lactic acid/glycolic acid copolymer, 10 to 30 mg of diethyl tartrate as a release aid, and 10 mg of argatroban as an antithrombin drug were dissolved in 1 mL of hexafluoro In isopropanol, cast on a SUS316L dish with a diameter of 18mm, and air-dry to obtain the drug carrier. It was soaked in 50 mL of pH 7.4 phosphate buffer solution, the buffer solution was regularly sampled, and the absorbance value at 330 nm, which is the characteristic absorption band of argatroban, was measured to track the dissolution amount of the drug. Table 4 shows the absorbance values from the start of dissolution to 7 days later.
作为比较例7、8,除不添加释放助剂之外,其他与实施例相同的条件进行了同样的溶出试验。As Comparative Examples 7 and 8, the same dissolution test was carried out under the same conditions as in Examples except that no release aid was added.
[表4][Table 4]
[实施例17~19][Example 17-19]
如表5所示,将90mg聚乳酸、作为释放助剂的酒石酸二乙酯30mg、及以表5所示的量称取抗凝血酶药的阿加曲班,溶解在1mL的六氟异丙醇中,将600μL的该溶液在直径18mm的SUS316L皿上流延,经风干获取药剂载体。将其浸泡在50mL的pH7.4的磷酸缓冲液中,定期采样缓冲液,测定作为阿加曲班的特性吸收带的330nm的吸光值,从而对该药剂的溶出量进行跟踪。溶出开始到2周后的吸光值如表5所示。As shown in Table 5, 90 mg of polylactic acid, 30 mg of diethyl tartrate as a release aid, and argatroban as an antithrombin drug in the amount shown in Table 5 were dissolved in 1 mL of hexafluoroiso In propanol, 600 μL of this solution was cast on a SUS316L dish with a diameter of 18 mm, and air-dried to obtain a drug carrier. It was soaked in 50 mL of pH 7.4 phosphate buffer solution, the buffer solution was regularly sampled, and the absorbance value at 330 nm, which is the characteristic absorption band of argatroban, was measured to track the dissolution amount of the drug. Table 5 shows the absorbance values from the start of dissolution to 2 weeks later.
[表5][table 5]
[实施例20、21][Example 20, 21]
将90mg聚乳酸、作为释放助剂的酒石酸二乙酯或苹果酸二乙酯30mg、及抗凝血酶药的阿加曲班30mg,溶解在1mL的六氟异丙醇中,将600μL的该溶液在直径18mm的SUS316L皿上流延,经风干获取药剂载体。将其浸泡在50mL的pH7.4的磷酸缓冲液中,定期采样缓冲液,测定作为阿加曲班的特性吸收带的330nm的吸光值,从而对该药剂的溶出量进行跟踪。溶出开始到2周后的吸光值如表6所示。Dissolve 90 mg of polylactic acid, 30 mg of diethyl tartrate or diethyl malate as a release aid, and 30 mg of argatroban as an antithrombin drug in 1 mL of hexafluoroisopropanol, and dissolve 600 μL of the The solution was cast on a SUS316L dish with a diameter of 18mm, and air-dried to obtain the drug carrier. It was soaked in 50 mL of pH 7.4 phosphate buffer solution, the buffer solution was regularly sampled, and the absorbance value at 330 nm, which is the characteristic absorption band of argatroban, was measured to track the dissolution amount of the drug. Table 6 shows the absorbance values from the start of dissolution to 2 weeks after.
[表6][Table 6]
[实施例22、23][Example 22, 23]
将90mg聚乳酸、作为释放助剂甘油的单醋酸酯的甘油单醋酸酯20mg、或甘油的二醋酸酯的甘油二醋酸酯20mg、及抗凝血酶药的阿加曲班20mg,溶解在1mL的六氟异丙醇中,将600μL的该溶液在直径18mm的SUS316L皿上流延,经风干获取药剂载体。将其浸泡在50mL的pH7.4的磷酸缓冲液中,定期采样缓冲液,测定作为阿加曲班的特性吸收带的330nm的吸光值,从而对该药剂的溶出量进行跟踪。溶出开始到3周后的吸光值如表7所示。Dissolve 90 mg of polylactic acid, 20 mg of glycerol monoacetate, or 20 mg of glycerin diacetate as a release aid, and 20 mg of argatroban, an antithrombin drug, in 1 mL In hexafluoroisopropanol, 600 μL of the solution was cast on a SUS316L dish with a diameter of 18 mm, and air-dried to obtain a drug carrier. It was soaked in 50 mL of pH 7.4 phosphate buffer solution, the buffer solution was regularly sampled, and the absorbance value at 330 nm, which is the characteristic absorption band of argatroban, was measured to track the dissolution amount of the drug. Table 7 shows the absorbance values from the start of the dissolution to 3 weeks later.
[表7][Table 7]
以下实施例中的释放速度是这样定义的。将药剂载体在37℃下,在pH7.4的磷酸缓冲液(PBS)中持续浸泡21天,观察该期间内的PBS吸光值变化。从第20天的吸光值和第21天的吸光值的差求出该24小时中的溶剂溶出量,除以24小时和体表面积而得的值为释放速度(单位:μg/(h·mm2))。支架的表面积是通过用显微镜观察求出其厚度和形状展开图,根据这些求出面积的。The release rate in the following examples is defined as such. The drug carrier was continuously soaked in phosphate buffer solution (PBS) of pH 7.4 at 37° C. for 21 days, and the change of PBS absorbance value during this period was observed. Calculate the amount of solvent dissolved in the 24 hours from the difference between the absorbance value on the 20th day and the absorbance value on the 21st day, and divide it by 24 hours and the body surface area to obtain the release rate (unit: μg/(h·mm 2 )). The surface area of the stent was obtained by observing the thickness and the developed shape of the stent under a microscope, and the area was obtained from these.
[实施例24~33][Example 24-33]
将15mg阿加曲班或盐酸沙格雷酯、和表8所示的非晶性聚合物50mg溶解在0.6mL的甲醇,在直径16mm的SUS皿上进行展开,经风干·真空干燥而获得药剂载体。将载体浸泡在50mL的pH7.4磷酸缓冲液中,定期采样缓冲液,对阿加曲班在其特性吸收带330nm上测定吸光值,对盐酸沙格雷酯在270nm上测定吸光值,进而测定溶出量,求得释放速度。其结果如表8所示。Dissolve 15 mg of argatroban or sargrelate hydrochloride and 50 mg of the amorphous polymer shown in Table 8 in 0.6 mL of methanol, develop on a SUS dish with a diameter of 16 mm, and air-dry or vacuum-dry to obtain a drug carrier . Soak the carrier in 50mL of pH7.4 phosphate buffer, sample the buffer regularly, measure the absorbance of argatroban at its characteristic absorption band at 330nm, and measure the absorbance at 270nm of sarcogrelate hydrochloride, and then measure the dissolution amount to obtain the release rate. The results are shown in Table 8.
[表8][Table 8]
PolyMEA:聚(丙烯酸-2-甲氧基乙酯);PolyHEMA:聚(甲基丙烯酸-2-羟基二乙酯);PolyEVE:聚乙基乙烯醚;Poly(MEA/HEMA):烯酸-2-甲氧基乙酯/甲基丙烯酸-2-羟基二乙酯的共聚物;PolyDnPAAm:聚(N,N-二-n-丙基丙烯酸酯)PolyMEA: poly(2-methoxyethyl acrylate); PolyHEMA: poly(2-hydroxydiethyl methacrylate); PolyEVE: polyethyl vinyl ether; Poly(MEA/HEMA): enoic acid-2 - Copolymer of methoxyethyl ester/2-hydroxydiethyl methacrylate; PolyDnPAAm: poly(N,N-di-n-propyl acrylate)
[比较例9~14][Comparative examples 9 to 14]
除了用晶体性的聚己内酯、聚羟基酪酸、己内酰胺代替实施例24所示的非晶性聚合物之外,其它与实施例24相同地求得了阿加曲班、盐酸沙格雷酯的释放速度。其结果如表9所示。The release of argatroban and sarcogrelate hydrochloride was obtained in the same manner as in Example 24, except that crystalline polycaprolactone, polyhydroxybutyric acid, and caprolactam were used instead of the amorphous polymer shown in Example 24. speed. The results are shown in Table 9.
[表9][Table 9]
[实施例34~40,比较例15~17][Examples 34-40, Comparative Examples 15-17]
向表10所示的具有非晶性的(DL)聚乳酸、和(DL)聚乳酸/乙醇酸共聚物50mg中、添加释放助剂15mg、阿加曲班或盐酸沙格雷酯15mg,溶解在0.5mL的六氟异丙醇中,在直径16mm的SUS皿上进行流延,经风干·真空干燥而获得药剂载体。将其浸泡在50mL的pH7.4磷酸缓冲液中,定期采样缓冲液,对阿加曲班在其特性吸收带330nm上测定吸光值,对盐酸沙格雷酯测定270nm处的吸光值,进而测定溶出量,求得释放速度。作为比较例,除了使用晶体性聚(L)乳酸、和(L)乳酸·乙醇酸共聚物(50∶50)之外,在与实施例34~40相同的条件下进行了同样的溶出试验。这些实施例及比较例的结果如表10所示。To 50 mg of amorphous (DL) polylactic acid and (DL) polylactic acid/glycolic acid copolymer shown in Table 10, 15 mg of a release aid, 15 mg of argatroban or sarcogrelate hydrochloride were added, and dissolved in In 0.5 mL of hexafluoroisopropanol, it was cast on a SUS dish with a diameter of 16 mm, and air-dried or vacuum-dried to obtain a drug carrier. Soak it in 50mL of pH7.4 phosphate buffer solution, sample the buffer solution regularly, measure the absorbance value at 330nm of its characteristic absorption band for argatroban, measure the absorbance value at 270nm for sarcogrelate hydrochloride, and then measure the dissolution amount to obtain the release velocity. As a comparative example, the same dissolution test was performed under the same conditions as in Examples 34 to 40 except for using crystalline poly(L) lactic acid and (L) lactic acid-glycolic acid copolymer (50:50). Table 10 shows the results of these Examples and Comparative Examples.
[表10][Table 10]
[实施例41][Example 41]
(支架留置试验)(Stent retention test)
将24mg的阿加曲班、24mg的盐酸沙格雷酯、24mg的酒石酸二乙酯、80mg的(DL)乳酸/乙醇酸共聚物(50∶50),溶解在10mL的六氟异丙醇中制成了涂覆液。将Co-Cr合金的冠状动脉用支架(直径1.55φ,长度17.4mm)浸泡在该涂覆液中,通过浸泡涂覆法在支架表面进行了0.6mg的涂覆。将3个经过涂覆的支架和3个未进行涂覆的裸金属支架分别留置到12个月龄的3头小猪的冠动脉中,一个月后杀死,对支架的开存状态进行了评价。涂覆药剂的支架与未进行涂覆的支架(裸金属支架)相比,3头的开存状态都良好,证实了阿加曲班和盐酸沙格雷酯的狭窄抑制效果。Dissolve 24 mg of argatroban, 24 mg of sargrelate hydrochloride, 24 mg of diethyl tartrate, and 80 mg of (DL) lactic acid/glycolic acid copolymer (50:50) in 10 mL of hexafluoroisopropanol to prepare became a coating liquid. A Co-Cr alloy coronary stent (diameter 1.55φ, length 17.4 mm) was immersed in the coating solution, and 0.6 mg was coated on the surface of the stent by the dip coating method. Three coated stents and three uncoated bare metal stents were indwelled in the coronary arteries of three piglets aged 12 months, and they were killed one month later. evaluate. The drug-coated stents were all in better condition than the non-coated stents (bare metal stents), confirming the stenosis inhibitory effect of argatroban and sargrelate hydrochloride.
Claims (20)
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP331503/2005 | 2005-11-16 | ||
| JP2005331503 | 2005-11-16 | ||
| JP130786/2006 | 2006-05-09 |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201010204153XA Division CN101869514B (en) | 2005-11-16 | 2006-11-15 | Controlled drug-release composition and drug-releasable medical device |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN101309706A true CN101309706A (en) | 2008-11-19 |
Family
ID=40125751
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CNA2006800430183A Pending CN101309706A (en) | 2005-11-16 | 2006-11-15 | Drug release control composition and drug-releasing medical device |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN101309706A (en) |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2010140163A3 (en) * | 2009-06-02 | 2011-01-27 | Concept Medical Inc. | Non-implantable medical device coated with encapsulated nanoparticles containing therapeutic agents |
| CN103893832A (en) * | 2014-02-20 | 2014-07-02 | 威海洁瑞医用制品有限公司 | Polylactic acid venous indwelling needle guide pipe |
| CN104784749A (en) * | 2015-02-04 | 2015-07-22 | 云南民族大学 | Wound protection liquid and preparation method thereof |
| CN111494723A (en) * | 2020-04-22 | 2020-08-07 | 苏州大学附属第一医院 | Preparation method of micro-nano fiber for promoting nerve regeneration through micro-environment responsive immune regulation |
| WO2021022799A1 (en) * | 2019-08-02 | 2021-02-11 | 上海心玮医疗科技股份有限公司 | Drugcoated balloon controllable in drug metabolism and preparation method therefor |
-
2006
- 2006-11-15 CN CNA2006800430183A patent/CN101309706A/en active Pending
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2010140163A3 (en) * | 2009-06-02 | 2011-01-27 | Concept Medical Inc. | Non-implantable medical device coated with encapsulated nanoparticles containing therapeutic agents |
| CN103893832A (en) * | 2014-02-20 | 2014-07-02 | 威海洁瑞医用制品有限公司 | Polylactic acid venous indwelling needle guide pipe |
| CN103893832B (en) * | 2014-02-20 | 2015-10-14 | 威海洁瑞医用制品有限公司 | A kind of polylactic acid vein indwelling needle catheter |
| CN104784749A (en) * | 2015-02-04 | 2015-07-22 | 云南民族大学 | Wound protection liquid and preparation method thereof |
| WO2021022799A1 (en) * | 2019-08-02 | 2021-02-11 | 上海心玮医疗科技股份有限公司 | Drugcoated balloon controllable in drug metabolism and preparation method therefor |
| CN111494723A (en) * | 2020-04-22 | 2020-08-07 | 苏州大学附属第一医院 | Preparation method of micro-nano fiber for promoting nerve regeneration through micro-environment responsive immune regulation |
| CN111494723B (en) * | 2020-04-22 | 2021-10-12 | 苏州大学附属第一医院 | A kind of preparation method of micro-environment responsive immune regulation to promote nerve regeneration micro-nanofibers |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN101869514B (en) | Controlled drug-release composition and drug-releasable medical device | |
| JP4987232B2 (en) | Medical devices using new polymers | |
| ES2223774T3 (en) | MEDICAL DEVICE. | |
| US7985415B2 (en) | Medical devices employing novel polymers | |
| JP5443336B2 (en) | Stent with biodegradable layer | |
| RU2400256C2 (en) | Capturing of endothelial precursor-cells by medication-eluting implanted medical device | |
| EP2471497A1 (en) | Medical device for placement into a lumen and manufacturing method thereof | |
| ES2383374T3 (en) | PEC Polymer Coated Devices | |
| KR20040005936A (en) | Sustained release drug delivery system containing codrugs | |
| TW200306826A (en) | Drug delivery systems for the prevention and treatment of vascular diseases | |
| WO2006034467A2 (en) | Biocompatible hydrogel compositions | |
| JPH09510231A (en) | Complexes of nitric oxide and cardiovascular amines as dual-acting cardiovascular agents | |
| JP2007153896A (en) | Compound of nanoparticle and/or microparticle for treating vascular disease based on topical injection | |
| KR100455343B1 (en) | Covering composition for drug releasing stent and drug releasing stent manufactured using same | |
| WO2009082014A1 (en) | Drug delivery system | |
| US12290615B2 (en) | Drug coated balloon and preparation method thereof | |
| US20060193893A1 (en) | Medical devices | |
| CN107865868A (en) | The new application of Amlexanox | |
| KR101925442B1 (en) | Macrophage Targeting Nanoparticles, compositions for coating medical equipment containing the same, and Medical equipment for anti-inflammation | |
| US20090098187A1 (en) | Composition And Its Use For The Manufacture Of A Medicament For Treating, Prophylactically Treating, Preventing Cancer And/Or Infections In The Urinary Tract | |
| CN101239216A (en) | Novel sacculus dilating catheter | |
| WO2007119423A1 (en) | Substance to be placed in the living body | |
| CN101309706A (en) | Drug release control composition and drug-releasing medical device | |
| JP2002320629A (en) | Medical care material to be embedded in vivo and medical care instrument | |
| WO2004017939A1 (en) | Medical instrument to be implanted in the body |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C12 | Rejection of a patent application after its publication | ||
| RJ01 | Rejection of invention patent application after publication |
Open date: 20081119 |