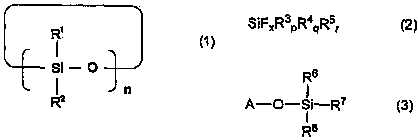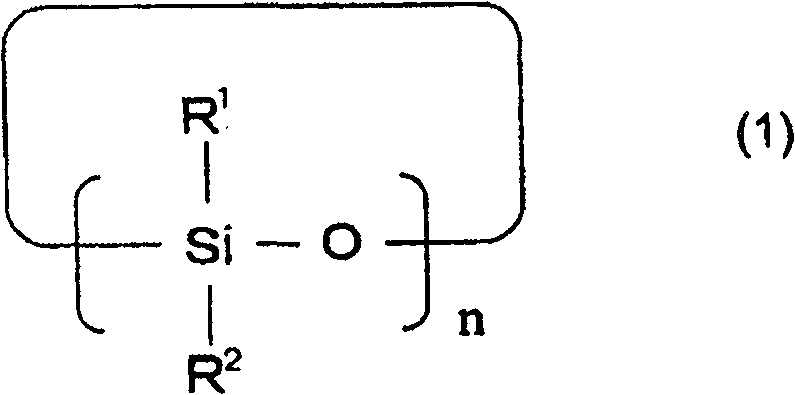CN101292389B - Lithium secondary battery and nonaqueous electrolyte used therein - Google Patents
Lithium secondary battery and nonaqueous electrolyte used therein Download PDFInfo
- Publication number
- CN101292389B CN101292389B CN2006800390896A CN200680039089A CN101292389B CN 101292389 B CN101292389 B CN 101292389B CN 2006800390896 A CN2006800390896 A CN 2006800390896A CN 200680039089 A CN200680039089 A CN 200680039089A CN 101292389 B CN101292389 B CN 101292389B
- Authority
- CN
- China
- Prior art keywords
- positive electrode
- active material
- electrode active
- secondary battery
- lithium secondary
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Images
Classifications
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/052—Li-accumulators
- H01M10/0525—Rocking-chair batteries, i.e. batteries with lithium insertion or intercalation in both electrodes; Lithium-ion batteries
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/056—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes
- H01M10/0564—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes the electrolyte being constituted of organic materials only
- H01M10/0566—Liquid materials
- H01M10/0567—Liquid materials characterised by the additives
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/056—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes
- H01M10/0564—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes the electrolyte being constituted of organic materials only
- H01M10/0566—Liquid materials
- H01M10/0569—Liquid materials characterised by the solvents
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/056—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes
- H01M10/0564—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes the electrolyte being constituted of organic materials only
- H01M10/0566—Liquid materials
- H01M10/0568—Liquid materials characterised by the solutes
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/058—Construction or manufacture
- H01M10/0587—Construction or manufacture of accumulators having only wound construction elements, i.e. wound positive electrodes, wound negative electrodes and wound separators
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M2004/021—Physical characteristics, e.g. porosity, surface area
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M2220/00—Batteries for particular applications
- H01M2220/20—Batteries in motive systems, e.g. vehicle, ship, plane
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M2300/00—Electrolytes
- H01M2300/0017—Non-aqueous electrolytes
- H01M2300/0025—Organic electrolyte
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M2300/00—Electrolytes
- H01M2300/0017—Non-aqueous electrolytes
- H01M2300/0025—Organic electrolyte
- H01M2300/0028—Organic electrolyte characterised by the solvent
- H01M2300/0037—Mixture of solvents
- H01M2300/004—Three solvents
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/13—Electrodes for accumulators with non-aqueous electrolyte, e.g. for lithium-accumulators; Processes of manufacture thereof
- H01M4/131—Electrodes based on mixed oxides or hydroxides, or on mixtures of oxides or hydroxides, e.g. LiCoOx
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/13—Electrodes for accumulators with non-aqueous electrolyte, e.g. for lithium-accumulators; Processes of manufacture thereof
- H01M4/133—Electrodes based on carbonaceous material, e.g. graphite-intercalation compounds or CFx
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/13—Electrodes for accumulators with non-aqueous electrolyte, e.g. for lithium-accumulators; Processes of manufacture thereof
- H01M4/134—Electrodes based on metals, Si or alloys
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/362—Composites
- H01M4/364—Composites as mixtures
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/58—Selection of substances as active materials, active masses, active liquids of inorganic compounds other than oxides or hydroxides, e.g. sulfides, selenides, tellurides, halogenides or LiCoFy; of polyanionic structures, e.g. phosphates, silicates or borates
- H01M4/583—Carbonaceous material, e.g. graphite-intercalation compounds or CFx
- H01M4/587—Carbonaceous material, e.g. graphite-intercalation compounds or CFx for inserting or intercalating light metals
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/10—Energy storage using batteries
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P70/00—Climate change mitigation technologies in the production process for final industrial or consumer products
- Y02P70/50—Manufacturing or production processes characterised by the final manufactured product
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Manufacturing & Machinery (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Electrochemistry (AREA)
- General Chemical & Material Sciences (AREA)
- General Physics & Mathematics (AREA)
- Inorganic Chemistry (AREA)
- Condensed Matter Physics & Semiconductors (AREA)
- Physics & Mathematics (AREA)
- Materials Engineering (AREA)
- Secondary Cells (AREA)
- Battery Electrode And Active Subsutance (AREA)
Abstract
Description
技术领域technical field
本发明涉及锂二次电池以及其中使用的非水电解液,更详细地说,涉及具有含有特定成分的锂二次电池用非水电解液、和具有特定组成和物性的能吸留和放出锂的正极和负极,并且特别是低温放电特性优异且容量高、长寿命、高输出的锂二次电池以及其中使用的非水电解液。The present invention relates to a lithium secondary battery and a non-aqueous electrolytic solution used therein, more specifically, to a non-aqueous electrolytic solution for a lithium secondary battery containing specific components, and a non-aqueous electrolytic solution having a specific composition and physical properties capable of occluding and releasing lithium. positive and negative electrodes, and particularly lithium secondary batteries excellent in low-temperature discharge characteristics and high in capacity, long life, and high output, and nonaqueous electrolytic solutions used therein.
背景技术Background technique
近年来,伴随着电子设备的小型化,对二次电池的高容量化的要求不断提高,能量密度比镍镉电池和镍氢电池高的锂二次电池受到关注。In recent years, along with the miniaturization of electronic equipment, the demand for higher capacity of secondary batteries has been increasing, and lithium secondary batteries with higher energy density than nickel-cadmium batteries and nickel-hydrogen batteries have attracted attention.
锂二次电池由于是高容量的二次电池,因此可用在各种用途中,但其主要使用在手机等比较小型的电池中,期待今后在汽车用途等的大型电池中扩大其应用。在大型电池中特别是对输出有要求,只是将以往的小型电池单纯变大在性能上并不充分。另外,提出了各种用于提高输出的电池材料的改良方法(例如,参照专利文献1~25、非专利文献1等)。可是,仍然不能得到足够的输出,要求进一步改善。Since lithium secondary batteries are high-capacity secondary batteries, they can be used in various applications, but they are mainly used in relatively small batteries such as mobile phones, and their application to large batteries such as automobiles is expected to expand in the future. In particular, output is required for large batteries, but simply enlarging conventional small batteries is not sufficient in terms of performance. In addition, various methods of improving battery materials for increasing output have been proposed (for example, refer to Patent Documents 1 to 25, Non-Patent Document 1, etc.). However, sufficient output cannot be obtained yet, and further improvement is demanded.
专利文献1:特开2005-071749号公报Patent Document 1: JP-A-2005-071749
专利文献2:特开2005-123180号公报Patent Document 2: JP-A-2005-123180
专利文献3:特开2001-206722号公报Patent Document 3: JP-A-2001-206722
专利文献4:特开2003-267732号公报Patent Document 4: JP-A-2003-267732
专利文献5:特开2001-015108号公报Patent Document 5: JP-A-2001-015108
专利文献6:WO2003/34518号公报Patent Document 6: WO2003/34518 Publication
专利文献7:特开平11-067270号公报Patent Document 7: JP-A-11-067270
专利文献8:特开昭61-168512号公报Patent Document 8: JP-A-61-168512
专利文献9:特开平6-275263号公报Patent Document 9: JP-A-6-275263
专利文献10:特开2000-340232号公报Patent Document 10: JP-A-2000-340232
专利文献11:特开2005-235397号公报Patent Document 11: JP-A-2005-235397
专利文献12:特开平11-031509号公报Patent Document 12: Japanese Unexamined Patent Publication No. 11-031509
专利文献13:特开平3-055770号公报Patent Document 13: JP-A-3-055770
专利文献14:特开2004-071458号公报Patent Document 14: JP-A-2004-071458
专利文献15:特开2004-087459号公报Patent Document 15: JP-A-2004-087459
专利文献16:特开平10-270074号公报Patent Document 16: Japanese Unexamined Patent Application Publication No. H10-270074
专利文献17:特开2002-075440号公报Patent Document 17: JP-A-2002-075440
专利文献18:特开平10-270075号公报Patent Document 18: Japanese Unexamined Patent Application Publication No. H10-270075
专利文献19:特开平8-045545号公报Patent Document 19: JP-A-8-045545
专利文献20:特开2001-006729号公报Patent Document 20: JP-A-2001-006729
专利文献21:特开平10-050342号公报Patent Document 21: Japanese Unexamined Patent Publication No. 10-050342
专利文献22:特开平9-106835号公报Patent Document 22: Japanese Unexamined Patent Publication No. 9-106835
专利文献23:特开2000-058116号公报Patent Document 23: JP-A-2000-058116
专利文献24:特开2001-015158号公报Patent Document 24: JP-A-2001-015158
专利文献25:特开2005-306619号公报Patent Document 25: JP-A-2005-306619
非专利文献1:J.Electrochem.soc.,145,L1(1998)Non-Patent Document 1: J. Electrochem.soc., 145, L1 (1998)
发明内容Contents of the invention
发明要解决的课题The problem to be solved by the invention
本发明就是鉴于这样的背景技术而作成的,其课题在于提供一种在制成更大型时也是高容量、长寿命、且具有高输出的锂二次电池。The present invention has been made in view of such background technology, and an object of the present invention is to provide a lithium secondary battery with high capacity, long life, and high output even when it is made larger.
解决课题的方法Solution to the problem
本发明人等对上述课题进行了深入研究,结果发现,通过使用具有特定的组成和物性的正极和负极、以及含有从特定的组中选择的化合物的非水电解液,可得到高容量、长寿命、且高输出的锂二次电池,从而完成了本发明。The inventors of the present invention conducted intensive studies on the above-mentioned subject, and as a result found that a high-capacity, long-term The present invention has been accomplished by providing a long-life and high-output lithium secondary battery.
即,本发明提供一种锂二次电池,该锂二次电池至少包含:在正极和负极间夹着微多孔膜隔板而构成的电极组、以及在非水溶剂中含有锂盐而形成的非水电解液,并且将它们装在电池外壳中,其中,That is, the present invention provides a lithium secondary battery comprising at least: an electrode group formed by sandwiching a porous membrane separator between a positive electrode and a negative electrode; non-aqueous electrolytes, and they are housed in battery casings, wherein,
所述正极和所述负极分别在集电体上形成活性物质层,该活性物质层含有能够吸留和放出锂离子的活性物质;The positive electrode and the negative electrode respectively form an active material layer on the current collector, and the active material layer contains an active material capable of absorbing and releasing lithium ions;
该非水电解液含有选自下述物质中的至少一种化合物,其在全部非水电解液中的含量为10ppm以上,所述物质包括:The non-aqueous electrolyte contains at least one compound selected from the following substances, and its content in all non-aqueous electrolytes is more than 10ppm, and the substances include:
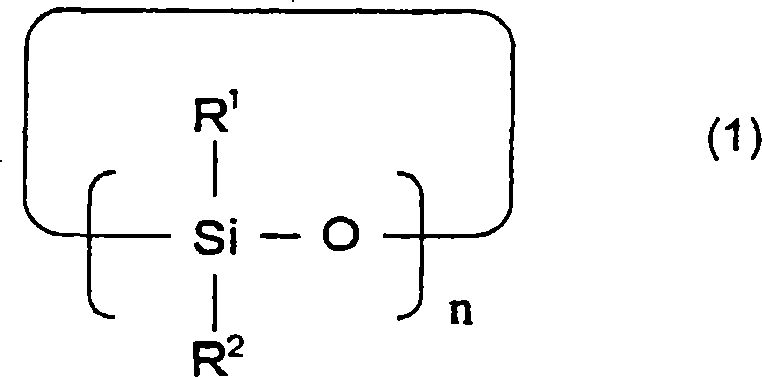
下述通式(1)表示的环状硅氧烷化合物、A cyclic siloxane compound represented by the following general formula (1),
[化学式1][chemical formula 1]
[通式(1)中,R1和R2可以相同也可以不同,表示碳原子数1~12的有机基团,n表示3~10的整数。][In the general formula (1), R 1 and R 2 may be the same or different, and represent an organic group with 1 to 12 carbon atoms, and n represents an integer of 3 to 10. ]
下述通式(2)表示的氟硅烷化合物、A fluorosilane compound represented by the following general formula (2),
[化学式2][chemical formula 2]
SiFxR3 pR4 qR5 r (2)SiF x R 3 p R 4 q R 5 r (2)
[通式(2)中,R3~R5可以相同也可以不同,表示碳原子数1~12的有机基团,x表示1~3的整数,p、q和r分别表示0~3的整数,且1≤p+q+r≤3。][In the general formula (2), R 3 to R 5 may be the same or different, and represent an organic group with 1 to 12 carbon atoms, x represents an integer of 1 to 3, and p, q and r represent 0 to 3, respectively. Integer, and 1≤p+q+r≤3. ]
下述通式(3)表示的化合物、A compound represented by the following general formula (3),
[化学式3][chemical formula 3]
[通式(3)中,R6~R8可以相同也可以不同,表示碳原子数1~12的有机基团,A表示由H、C、N、O、F、S、Si和/或P构成的基团。],以及[In the general formula (3), R 6 to R 8 may be the same or different, and represent an organic group with 1 to 12 carbon atoms, and A represents an organic group composed of H, C, N, O, F, S, Si and/or The group formed by P. ],as well as
分子内具有S-F键的化合物、硝酸盐、亚硝酸盐、单氟磷酸盐、二氟磷酸盐、乙酸盐和丙酸盐;Compounds with S-F bonds in the molecule, nitrates, nitrites, monofluorophosphates, difluorophosphates, acetates and propionates;
并且,所述正极是选自下述正极[1]~正极[5]中的任意一种正极:And, the positive electrode is any positive electrode selected from the following positive electrode [1] to positive electrode [5]:
正极[1]:包含正极活性物质的正极,该正极活性物质含有锰;Positive electrode [1]: a positive electrode comprising a positive electrode active material containing manganese;
正极[2]:包含正极活性物质的正极,该正极活性物质具有下述组成式(4)表示的组成,Positive electrode [2]: a positive electrode comprising a positive electrode active material having a composition represented by the following composition formula (4),
LixNi(1-y-z)CoyMzO2 组成式(4)Li x Ni (1-yz) Co y M z O 2 composition formula (4)
[组成式(4)中,M表示选自Mn、Al、Fe、Ti、Mg、Cr、Ga、Cu、Zn和Nb中的至少一种元素,x表示满足0<x≤1.2的数,y表示满足0.05≤y≤0.5的数,z表示满足0.01≤z≤0.5的数。];[In the composition formula (4), M represents at least one element selected from Mn, Al, Fe, Ti, Mg, Cr, Ga, Cu, Zn and Nb, x represents a number satisfying 0<x≤1.2, and y represents a number satisfying 0.05≤y≤0.5, and z represents a number satisfying 0.01≤z≤0.5. ];
正极[3]:包含选自下述(a)~(d)中的任一种正极活性物质的正极,Positive electrode [3]: a positive electrode comprising any positive electrode active material selected from the following (a) to (d),
(a)BET比表面积为0.4m2/g~2m2/g的正极活性物质(a) A positive electrode active material having a BET specific surface area of 0.4m 2 /g to 2m 2 /g
(b)平均一次粒径为0.1μm~2μm的正极活性物质(b) Positive active material having an average primary particle size of 0.1 μm to 2 μm
(c)中值粒径d50为1μm~20μm的正极活性物质(c) A positive electrode active material having a median diameter d50 of 1 μm to 20 μm
(d)振实密度为1.3g/cm3~2.7g/cm3的正极活性物质;(d) a positive electrode active material with a tap density of 1.3g/cm 3 to 2.7g/cm 3 ;
正极[4]:满足选自下述(e)~(f)中的任意一个条件的正极,Positive electrode [4]: A positive electrode that satisfies any one of the following conditions (e) to (f),
(e)在集电体上形成含有正极活性物质、导电材料和粘合剂的正极活性物质层而制作的正极,其中,正极活性物质层中的导电材料的含量为6质量%~20质量%的范围(e) A positive electrode made by forming a positive electrode active material layer containing a positive electrode active material, a conductive material and a binder on a current collector, wherein the content of the conductive material in the positive electrode active material layer is 6% by mass to 20% by mass range
(f)在集电体上形成含有正极活性物质和粘合剂的正极活性物质层而制作的正极,其中,正极活性物质层的密度为1.7g/cm3~3.5g/cm3的范围(f) A positive electrode produced by forming a positive electrode active material layer containing a positive electrode active material and a binder on a current collector, wherein the density of the positive electrode active material layer is in the range of 1.7 g/cm 3 to 3.5 g/cm 3
(g)在集电体上形成含有正极活性物质和粘合剂的正极活性物质层而制作的正极,其中,正极活性物质层和集电体的厚度之比(注入非水电解液之前的一侧活性物质层的厚度)/(集电体的厚度)的值为1.6~20;(g) a positive electrode made by forming a positive electrode active material layer containing a positive electrode active material and a binding agent on the current collector, wherein the ratio of the thickness of the positive electrode active material layer to the current collector (one period before injecting the nonaqueous electrolyte solution) The thickness of the side active material layer)/(the thickness of the current collector) is 1.6 to 20;
正极[5]:含有组成不同的2种以上正极活性物质的正极。Positive electrode [5]: A positive electrode containing two or more positive electrode active materials with different compositions.
另外,本发明提供一种锂二次电池,该锂二次电池至少包含:在正极和负极间夹着微多孔膜隔板而构成的电极组、以及在非水溶剂中含有锂盐而形成的非水电解液,并且将它们装在电池外壳中,其中,In addition, the present invention provides a lithium secondary battery comprising at least: an electrode group formed by sandwiching a porous film separator between a positive electrode and a negative electrode; non-aqueous electrolytes, and they are housed in battery casings, wherein,
所述正极和所述负极分别在集电体上形成活性物质层,该活性物质层含有能够吸留和放出锂离子的活性物质;The positive electrode and the negative electrode respectively form an active material layer on the current collector, and the active material layer contains an active material capable of absorbing and releasing lithium ions;
该非水电解液含有选自下述物质中的至少一种化合物,并且其在全部非水电解液中的含量为10ppm以上,所述物质包括:上述通式(1)表示的环状硅氧烷化合物、上述通式(2)表示的氟硅烷化合物、上述通式(3)表示的化合物、分子内具有S-F键的化合物、硝酸盐、亚硝酸盐、单氟磷酸盐、二氟磷酸盐、乙酸盐和丙酸盐;The non-aqueous electrolytic solution contains at least one compound selected from the following substances, and its content in all non-aqueous electrolytic solutions is more than 10 ppm, and the substance includes: the cyclic silicon oxide represented by the above general formula (1) alkane compounds, fluorosilane compounds represented by the above general formula (2), compounds represented by the above general formula (3), compounds having an S-F bond in the molecule, nitrates, nitrites, monofluorophosphates, difluorophosphates, Acetate and propionate;
并且,所述负极是选自下述负极[1]~负极[10]中的任意一种负极:And, the negative electrode is any negative electrode selected from the following negative electrodes [1] to negative electrodes [10]:
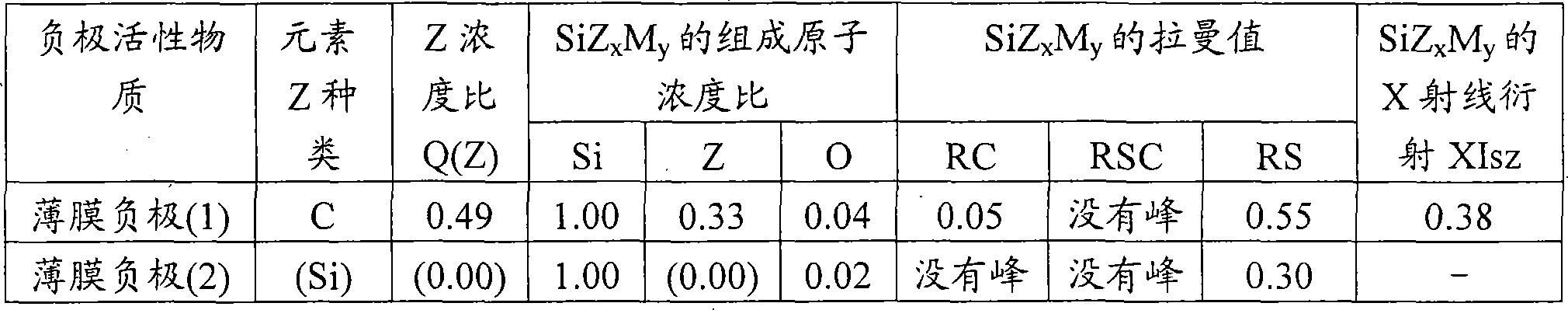
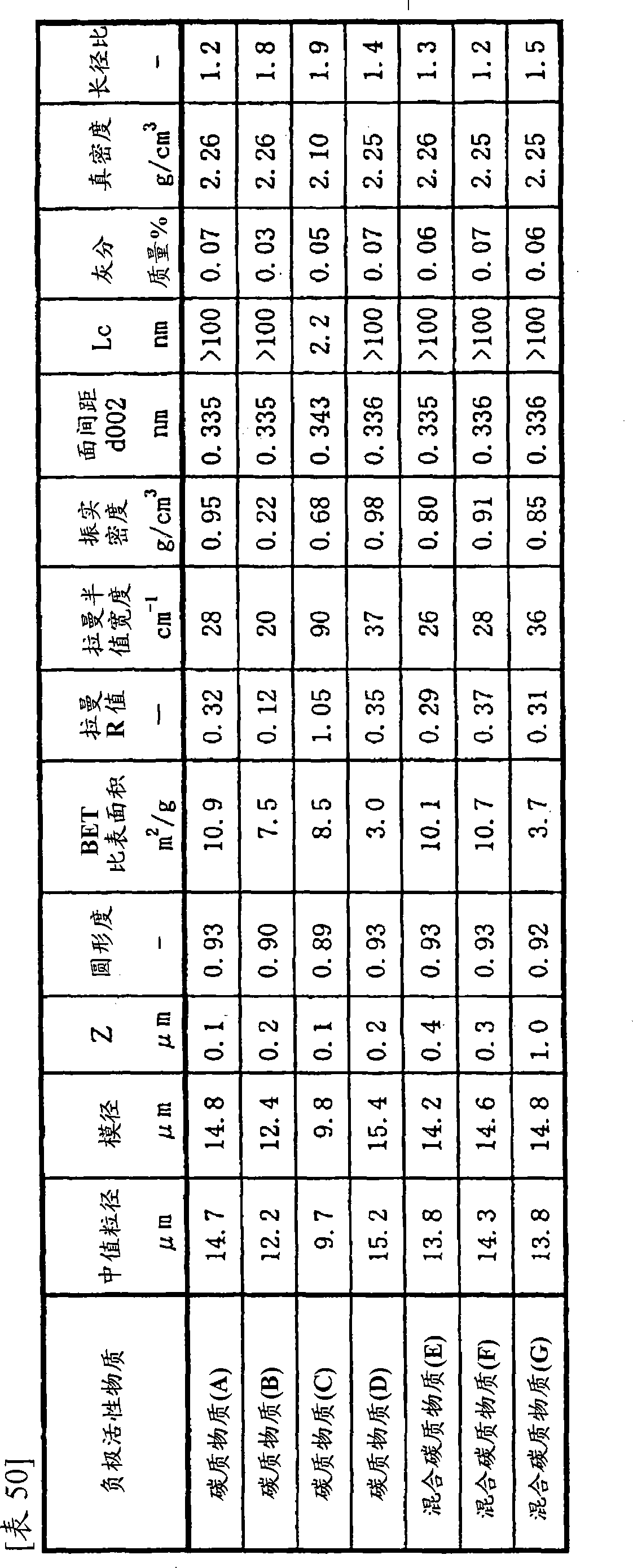
负极[1]:含有2种以上结晶性不同的碳质物质作为负极活性物质的负极;Negative electrode [1]: a negative electrode containing two or more carbonaceous substances with different crystallinity as the negative electrode active material;
负极[2]:含有无定形碳质作为负极活性物质的负极,所述无定形碳质通过广角X射线衍射法测定的(002)面的面间距(d002)为0.337nm以上,微晶大小(Lc)为80nm以下,采用氩离子激光拉曼光谱法测定的以1360cm-1的峰强度相对于1580cm-1的峰强度之比定义的拉曼R值为0.2以上;Negative pole [2]: the negative pole that contains amorphous carbonaceous as negative electrode active material, and the interplanar spacing (d002) of (002) plane of described amorphous carbonaceous by wide-angle X-ray diffractometry is more than 0.337nm, crystallite size ( Lc) is less than 80nm, and the Raman R value defined by the ratio of the peak intensity at 1360cm -1 to the peak intensity at 1580cm -1 measured by argon ion laser Raman spectroscopy is 0.2 or more;
负极[3]:含有金属氧化物作为负极活性物质的负极,所述金属氧化物含有能够吸留和放出锂的钛;Negative electrode [3]: a negative electrode containing a metal oxide as the negative electrode active material, and the metal oxide contains titanium capable of occluding and releasing lithium;
负极[4]:含有碳质物质作为负极活性物质的负极,所述碳质物质的圆形度为0.85以上,表面官能团量O/C值为0~0.01;Negative electrode [4]: a negative electrode containing carbonaceous material as the negative electrode active material, the circularity of the carbonaceous material is above 0.85, and the O/C value of the surface functional group is 0-0.01;
负极[5]:含有异取向性碳复合物作为负极活性物质的负极,所述异取向性碳复合物含有2种以上取向性不同的碳质物质;Negative electrode [5]: a negative electrode containing a hetero-orientation carbon composite as the anode active material, and the hetero-orientation carbon composite contains two or more carbonaceous substances with different orientations;
负极[6]:含有石墨质碳粒子作为负极活性物质的负极,所述石墨质碳粒子的圆形度为0.85以上,通过广角X射线衍射法测定的(002)面的面间距(d002)低于0.337nm,采用氩离子激光拉曼光谱法测定的以1360cm-1的峰强度相对于1580cm-1的峰强度之比定义的拉曼R值为0.12~0.8;Negative electrode [6]: a negative electrode containing graphitic carbon particles as the negative electrode active material, the circularity of the graphitic carbon particles is 0.85 or more, and the interplanar distance (d002) of the (002) plane measured by wide-angle X-ray diffraction method is low At 0.337nm, the Raman R value defined by the ratio of the peak intensity at 1360cm -1 to the peak intensity at 1580cm -1 measured by argon ion laser Raman spectroscopy is 0.12-0.8;
负极[7]:含有下述的包含多元素的负极活性物质(C’)作为负极活性物质的负极,所述包含多元素的负极活性物质(C’)含有选自Al、Pb、Zn、Sn、Bi、In、Mg、Ga、Cd、Ag、Si、B、Au、Pt、Pd和Sb的锂吸留金属(A’)和/或锂吸留合金(B’)中的至少一种,并且含有C和/或N作为元素Z;Negative electrode [7]: a negative electrode containing the following multi-element-containing negative electrode active material (C') as the negative electrode active material, the multi-element-containing negative electrode active material (C') containing , Bi, In, Mg, Ga, Cd, Ag, Si, B, Au, Pt, Pd, and Sb lithium storage metal (A') and/or lithium storage alloy (B'), and contains C and/or N as element Z;
负极[8]:含有性质不同的2种以上的负极活性物质作为负极活性物质的负极;Negative electrode [8]: a negative electrode containing two or more negative electrode active materials with different properties as the negative electrode active material;
负极[9]:含有振实密度为0.1g/cm3以上,并且采用水银孔隙率计测定的相当于直径0.01μm~1μm范围的粒子的微孔容积为0.01mL/g以上的负极活性物质的负极;Negative electrode [9]: Containing a negative electrode active material with a tap density of 0.1 g/cm 3 or more and a pore volume equivalent to particles in the range of 0.01 μm to 1 μm in diameter measured by a mercury porosimeter of 0.01 mL/g or more negative electrode;
负极[10]:充电到该负极的标称容量的60%时,由负极的对置电池产生的反应电阻为500Ω以下的负极。Negative pole [10]: when charged to 60% of the nominal capacity of the negative pole, the reaction resistance produced by the opposite battery of the negative pole is a negative pole below 500Ω.
另外,本发明提供一种锂二次电池,该锂二次电池至少包含:将正极和负极夹着微多孔膜隔板而构成的电极组、以及在非水溶剂中含有锂盐而形成的非水电解液,并且将它们装在电池外壳中,其中,In addition, the present invention provides a lithium secondary battery comprising at least: an electrode group formed by sandwiching a positive electrode and a negative electrode with a porous membrane separator; and a non-aqueous solvent containing a lithium salt. water electrolyte, and they are contained in the battery casing, wherein,
所述正极和所述负极分别在集电体上形成活性物质层,该活性物质层含有能够吸留和放出锂离子的活性物质;The positive electrode and the negative electrode respectively form an active material layer on the current collector, and the active material layer contains an active material capable of absorbing and releasing lithium ions;
该非水电解液含有选自下述物质中的至少一种化合物,并且其在全部非水电解液中的含量为10ppm以上,所述物质包括:上述通式(1)表示的环状硅氧烷化合物、上述通式(2)表示的氟硅烷化合物、上述通式(3)表示的化合物、分子内具有S-F键的化合物、硝酸盐、亚硝酸盐、单氟磷酸盐、二氟磷酸盐、乙酸盐和丙酸盐;The non-aqueous electrolytic solution contains at least one compound selected from the following substances, and its content in all non-aqueous electrolytic solutions is more than 10 ppm, and the substance includes: the cyclic silicon oxide represented by the above general formula (1) alkane compounds, fluorosilane compounds represented by the above general formula (2), compounds represented by the above general formula (3), compounds having an S-F bond in the molecule, nitrates, nitrites, monofluorophosphates, difluorophosphates, Acetate and propionate;
并且,所述非水电解液是满足选自下述电解液[1]~电解液[9]中的任一条件的电解液:And, the non-aqueous electrolyte is an electrolyte that satisfies any condition selected from the following electrolyte [1] to electrolyte [9]:
电解液[1]:构成电解液的非水溶剂是至少含有碳酸亚乙酯的混合溶剂,并且碳酸亚乙酯相对于非水溶剂总量的比例为1容量%~25容量%;Electrolyte [1]: The nonaqueous solvent constituting the electrolyte is a mixed solvent containing at least ethylene carbonate, and the ratio of ethylene carbonate to the total amount of nonaqueous solvent is 1% by volume to 25% by volume;
电解液[2]:构成电解液的非水溶剂含有至少一种不对称链状碳酸酯,并且该不对称链状碳酸酯在全部非水溶剂中所占的含有比例为5体积%~90体积%;Electrolyte [2]: The nonaqueous solvent constituting the electrolyte contains at least one asymmetric chain carbonate, and the proportion of the asymmetric chain carbonate in all nonaqueous solvents is 5% to 90% by volume %;
电解液[3]:构成电解液的非水溶剂含有至少一种链状羧酸酯;Electrolyte [3]: the non-aqueous solvent constituting the electrolyte contains at least one chain carboxylate;
电解液[4]:构成电解液的非水溶剂含有闪点为70℃以上的溶剂,其含量为全部非水溶剂的60容量%以上;Electrolyte [4]: The non-aqueous solvent constituting the electrolyte contains a solvent with a flash point above 70°C, and its content is above 60% by volume of all non-aqueous solvents;
电解液[5]:含有LiN(CnF2n+1SO2)2(式中,n为1~4的整数)和/或二(草酸根合)硼酸锂作为构成电解液的锂盐;Electrolyte [5]: containing LiN(C n F 2n+1 SO 2 ) 2 (wherein, n is an integer of 1 to 4) and/or lithium bis(oxalato)borate as the lithium salt constituting the electrolyte;
电解液[6]:构成电解液的锂盐是含氟锂盐,并且在全部非水电解液中含有10ppm~300ppm的氟化氢(HF);Electrolyte [6]: The lithium salt constituting the electrolyte is a fluorine-containing lithium salt, and contains 10 ppm to 300 ppm of hydrogen fluoride (HF) in all non-aqueous electrolytes;
电解液[7]:在电解液中含有碳酸亚乙烯酯,该碳酸亚乙烯酯的含量为电解液总质量的0.001质量%~3质量%的范围;Electrolyte [7]: the electrolyte contains vinylene carbonate, and the content of the vinylene carbonate is in the range of 0.001% by mass to 3% by mass of the total mass of the electrolyte;
电解液[8]:在电解液中还含有选自下述通式(4)表示的化合物、含有氮和/或硫的杂环化合物、环状羧酸酯、含氟环状碳酸酯中的至少一种化合物,其在全部非水电解液中的含量为0.001质量%~5质量%的范围,Electrolyte [8]: The electrolyte also contains compounds selected from the following general formula (4), heterocyclic compounds containing nitrogen and/or sulfur, cyclic carboxylates, and fluorine-containing cyclic carbonates. at least one compound, the content of which is in the range of 0.001% by mass to 5% by mass in the entire non-aqueous electrolyte solution,
[化学式4][chemical formula 4]
[通式(4)中,R9~R12可以相同也可以不同,表示由选自H、C、N、O、F、S和P中的至少一种元素构成的基团。];[In general formula (4), R 9 to R 12 may be the same or different, and represent a group composed of at least one element selected from H, C, N, O, F, S, and P. ];
电解液[9]:在电解液中,还含有过充电防止剂。Electrolyte solution [9]: The electrolyte solution also contains an overcharge preventing agent.
在上述二次电池中,特别优选具有选自下述(1)~(3)中任一种性质的二次电池,Among the above-mentioned secondary batteries, a secondary battery having any one of properties selected from the following (1) to (3) is particularly preferred,
(1)正极的电极面积总和相对于二次电池的外壳的表面积的面积比为20倍以上;(1) The area ratio of the total electrode area of the positive electrode to the surface area of the case of the secondary battery is 20 times or more;
(2)二次电池的直流电阻成分为20毫欧(mΩ)以下;(2) The DC resistance component of the secondary battery is below 20 milliohms (mΩ);
(3)装在二次电池的一个电池外壳中的电池要素所具有的电容为3安培小时(Ah)以上。(3) The battery element housed in one battery case of the secondary battery has a capacitance of 3 ampere hours (Ah) or more.
另外,本发明提供一种锂二次电池,该锂二次电池至少包含:将正极和负极夹着微多孔膜隔板而构成的电极组、以及在非水溶剂中含有锂盐而形成的非水电解液,并且将它们装在电池外壳中,其中,In addition, the present invention provides a lithium secondary battery comprising at least: an electrode group formed by sandwiching a positive electrode and a negative electrode with a porous membrane separator; and a non-aqueous solvent containing a lithium salt. water electrolyte, and they are contained in the battery casing, wherein,
所述正极和所述负极分别在集电体上形成活性物质层,该活性物质层含有能够吸留和放出锂离子的活性物质;The positive electrode and the negative electrode respectively form an active material layer on the current collector, and the active material layer contains an active material capable of absorbing and releasing lithium ions;
该非水电解液含有选自下述物质中的至少一种化合物,并且其在全部非水电解液中的含量为10ppm以上,所述物质包括:上述通式(1)表示的环状硅氧烷化合物、上述通式(2)表示的氟硅烷化合物、上述通式(3)表示的化合物、分子内具有S-F键的化合物、硝酸盐、亚硝酸盐、单氟磷酸盐、二氟磷酸盐、乙酸盐和丙酸盐;The non-aqueous electrolytic solution contains at least one compound selected from the following substances, and its content in all non-aqueous electrolytic solutions is more than 10 ppm, and the substance includes: the cyclic silicon oxide represented by the above general formula (1) alkane compounds, fluorosilane compounds represented by the above general formula (2), compounds represented by the above general formula (3), compounds having an S-F bond in the molecule, nitrates, nitrites, monofluorophosphates, difluorophosphates, Acetate and propionate;
并且,该二次电池具有选自下述结构[1]~结构[6]中的任一种性质:And, the secondary battery has any one of properties selected from the following structure [1] to structure [6]:
结构[1]:所述正极的电极面积总和相对于所述二次电池的外壳的表面积的面积比为20倍以上;Structure [1]: the area ratio of the sum of the electrode areas of the positive electrode to the surface area of the case of the secondary battery is 20 times or more;
结构[2]:所述二次电池的直流电阻成分为20毫欧(mΩ)以下;Structure [2]: The DC resistance component of the secondary battery is below 20 milliohms (mΩ);
结构[3]:装在所述二次电池的一个电池外壳中的电池要素所具有的电容为3安培小时(Ah)以上;Structure [3]: The battery element housed in one battery case of the secondary battery has a capacitance of 3 ampere hours (Ah) or more;
结构[4]:所述二次电池的正极和负极的集电体分别由金属材料制成,并且所述集电体的金属材料和用于将电流取出到外部的导电体通过槽焊、高频焊接或超声波焊接中的任一种方法进行焊接;Structure [4]: The current collectors of the positive and negative electrodes of the secondary battery are respectively made of metal materials, and the metal materials of the current collectors and the conductors for taking the current to the outside are welded by slot welding, high Welding by any method of frequency welding or ultrasonic welding;
结构[5]:所述二次电池的外壳是铝或铝合金;Structure [5]: the casing of the secondary battery is made of aluminum or aluminum alloy;
结构[6]:所述二次电池的形成上述电池外壳的外壳材料是如下的外壳材料:电池的内面侧的至少一部分包含使用热塑性树脂而形成的片材,在容纳所述电池组的同时,还可以通过使所述热塑性树脂层彼此热封而将所述电池组进行密封。Structure [6]: The case material forming the above-mentioned battery case of the secondary battery is a case material in which at least a part of the inner surface side of the battery includes a sheet formed using a thermoplastic resin, and while housing the battery pack, The battery pack can also be sealed by heat-sealing the thermoplastic resin layers to each other.
另外,本发明还提供一种二次电池用非水电解液,该非水电解液至少包含非水溶剂和锂盐,其中,In addition, the present invention also provides a non-aqueous electrolytic solution for a secondary battery, the non-aqueous electrolytic solution at least includes a non-aqueous solvent and a lithium salt, wherein,
该非水电解液含有选自下述物质中的至少一种化合物,并且其在全部非水电解液中的含量为10ppm以上,所述物质包括:上述通式(1)表示的环状硅氧烷化合物、上述通式(2)表示的氟硅烷化合物、上述通式(3)表示的化合物、分子内具有S-F键的化合物、硝酸盐、亚硝酸盐、单氟磷酸盐、二氟磷酸盐、乙酸盐和丙酸盐;The non-aqueous electrolytic solution contains at least one compound selected from the following substances, and its content in all non-aqueous electrolytic solutions is more than 10 ppm, and the substance includes: the cyclic silicon oxide represented by the above general formula (1) alkane compounds, fluorosilane compounds represented by the above general formula (2), compounds represented by the above general formula (3), compounds having an S-F bond in the molecule, nitrates, nitrites, monofluorophosphates, difluorophosphates, Acetate and propionate;
并且,所述非水电解液满足选自下述电解液[1]~电解液[9]中的任一条件:And, the non-aqueous electrolytic solution satisfies any condition selected from the following electrolytic solutions [1] to electrolytic solutions [9]:
电解液[1]:构成电解液的非水溶剂是至少含有碳酸亚乙酯的混合溶剂,并且碳酸亚乙酯相对于非水溶剂总量的比例为1容量%~25容量%;Electrolyte [1]: The nonaqueous solvent constituting the electrolyte is a mixed solvent containing at least ethylene carbonate, and the ratio of ethylene carbonate to the total amount of nonaqueous solvent is 1% by volume to 25% by volume;
电解液[2]:构成电解液的非水溶剂含有至少一种不对称链状碳酸酯,并且该不对称链状碳酸酯在全部非水溶剂中所占的含有比例为5体积%~90体积%;Electrolyte [2]: The nonaqueous solvent constituting the electrolyte contains at least one asymmetric chain carbonate, and the proportion of the asymmetric chain carbonate in all nonaqueous solvents is 5% to 90% by volume %;
电解液[3]:构成电解液的非水溶剂含有至少一种链状羧酸酯;Electrolyte [3]: the non-aqueous solvent constituting the electrolyte contains at least one chain carboxylate;
电解液[4]:构成电解液的非水溶剂含有闪点为70℃以上的溶剂,并且其含量为全部非水溶剂的60容量%以上;Electrolyte [4]: The non-aqueous solvent constituting the electrolyte contains a solvent with a flash point of 70°C or higher, and its content is more than 60% by volume of the total non-aqueous solvent;
电解液[5]:含有LiN(CnF2n+1SO2)2(式中,n为1~4的整数)和/或二(草酸根合)硼酸锂作为构成电解液的锂盐;Electrolyte [5]: containing LiN(C n F 2n+1 SO 2 ) 2 (wherein, n is an integer of 1 to 4) and/or lithium bis(oxalato)borate as the lithium salt constituting the electrolyte;
电解液[6]:构成电解液的锂盐是含氟锂盐,并且在全部非水电解液中含有10ppm~300ppm的氟化氢(HF);Electrolyte [6]: The lithium salt constituting the electrolyte is a fluorine-containing lithium salt, and contains 10 ppm to 300 ppm of hydrogen fluoride (HF) in all non-aqueous electrolytes;
电解液[7]:在电解液中含有碳酸亚乙烯酯,并且该碳酸亚乙烯酯的含量为电解液总质量的0.001质量%~3质量%的范围;Electrolyte [7]: the electrolyte contains vinylene carbonate, and the content of the vinylene carbonate is in the range of 0.001% by mass to 3% by mass of the total mass of the electrolyte;
电解液[8]:在电解液中还含有选自上述通式(4)表示的化合物、含有氮和/或硫的杂环化合物、环状羧酸酯、含氟环状碳酸酯中的至少一种化合物,并且其在全部非水电解液中的含量为0.001质量%~5质量%的范围;Electrolyte solution [8]: the electrolyte solution also contains at least A compound whose content in the entire non-aqueous electrolyte is in the range of 0.001% by mass to 5% by mass;
电解液[9]:在电解液中,还含有过充电防止剂。Electrolyte solution [9]: The electrolyte solution also contains an overcharge preventing agent.
发明的效果The effect of the invention
根据本发明,可以提供特别是低温放电特性优异的锂二次电池,通过后面叙述的发明的各种方式,可发挥例如如下所述的效果。According to the present invention, it is possible to provide a lithium secondary battery particularly excellent in low-temperature discharge characteristics, and various aspects of the invention to be described later can exhibit, for example, the following effects.
正极[1]:Positive pole [1]:
根据本发明,可以以更便宜的材料得到长寿命、高输出,因此可以提供特别适用于例如汽车用途等的大型电池的锂二次电池。According to the present invention, since a long life and high output can be obtained with less expensive materials, it is possible to provide a lithium secondary battery particularly suitable for large batteries such as automobiles.
正极[2]:Positive electrode [2]:
根据本发明,可以提供高容量且具有长寿命、高输出的锂二次电池,还可以提供特别适用于例如汽车用途等的大型电池的锂二次电池。According to the present invention, it is possible to provide a high-capacity, long-life, and high-output lithium secondary battery, and also provide a lithium secondary battery particularly suitable for large batteries such as automobiles.
正极[3]:Positive electrode [3]:
根据本发明,可以得到长寿命、高输出的锂二次电池,因此可以提供特别适用于例如汽车用途等的大型电池的锂二次电池。According to the present invention, a long-life and high-output lithium secondary battery can be obtained, and therefore, a lithium secondary battery particularly suitable for large batteries such as automobile applications can be provided.
正极[4]:Positive pole [4]:
根据本发明,可以得到高输出的锂二次电池,因此可以提供特别适用于例如汽车用途等的大型电池的锂二次电池。According to the present invention, since a high-output lithium secondary battery can be obtained, it is possible to provide a lithium secondary battery particularly suitable for a large battery such as an automobile.
正极[5]:Positive electrode [5]:
根据本发明,可以得到高容量、长寿命、高输出,并且抑制了因反复进行电池的充放电而引起的电池容量和输出的降低(反复充放电特性(循环特性)优异)的锂二次电池,因此可以提供特别适用于例如汽车用途等的大型电池的锂二次电池。According to the present invention, it is possible to obtain a lithium secondary battery with high capacity, long life, high output, and suppression of reduction in battery capacity and output due to repeated charge and discharge of the battery (excellent in repeated charge and discharge characteristics (cycle characteristics)) , and therefore it is possible to provide a lithium secondary battery which is particularly suitable for a large battery such as an automobile application.
负极[1]:Negative pole [1]:
根据本发明,可以提供特别适合作为大型电池的锂二次电池,该锂二次电池可以保持循环特性提高的效果,同时可以在循环初期到末期都保持高的输出特性,即使在进行充放电循环后的劣化之后,也可以保持大功率下的输出特性。According to the present invention, it is possible to provide a lithium secondary battery particularly suitable as a large-scale battery, which can maintain the effect of improving the cycle characteristics, and can maintain high output characteristics from the beginning to the end of the cycle at the same time, even after charging and discharging cycles. After the final degradation, the output characteristics under high power can also be maintained.
负极[2]:Negative pole [2]:
根据本发明,可以提供短时间高电流密度充放电特性良好的锂二次电池。According to the present invention, it is possible to provide a lithium secondary battery having excellent short-time high current density charge and discharge characteristics.
负极[3]:Negative pole [3]:
根据本发明,可以提供输出电阻小,并可以有效利用能量的锂二次电池。According to the present invention, it is possible to provide a lithium secondary battery that has a small output resistance and can utilize energy efficiently.
负极[4]:Negative pole [4]:
根据本发明,可以提供在低充电深度下的高温保存耐性得到提高的锂二次电池。According to the present invention, a lithium secondary battery having improved high-temperature storage resistance at a low depth of charge can be provided.
负极[5]:Negative electrode [5]:
根据本发明,可以提供在低充电深度下的反复长时间充放电时也可以保持良好性能的锂二次电池。According to the present invention, it is possible to provide a lithium secondary battery capable of maintaining good performance even when charging and discharging are repeated for a long time at a low depth of charge.
负极[6]:Negative electrode [6]:
根据本发明,可以提供低温时从低输出状态恢复输出快的锂二次电池。According to the present invention, it is possible to provide a lithium secondary battery that recovers output quickly from a low output state at low temperature.
负极[7]:Negative electrode [7]:
根据本发明,可以提供在制成更大型时容量也大,并且充电接受性良好的锂二次电池。According to the present invention, it is possible to provide a lithium secondary battery having a large capacity even when it is made larger and having good charge acceptance.
负极[8]:Negative electrode [8]:
根据本发明,可以提供循环特性和低温输出均良好的锂二次电池。According to the present invention, a lithium secondary battery having excellent cycle characteristics and low-temperature output can be provided.
负极[9][10]:Negative pole [9] [10]:
根据本发明,可以提供在将电池制成更大型时循环保持率也大,并可实现良好的电池寿命,即使在充放电循环试验后也可以实现高输出,或者两者兼备的锂二次电池。According to the present invention, it is possible to provide a lithium secondary battery that has a large cycle retention rate when the battery is made larger, and can achieve a good battery life, and can achieve high output even after a charge-discharge cycle test, or both. .
电解液[1]:Electrolyte[1]:
根据本发明,可以大幅提高非水电解质二次电池的低温特性,更详细地,可以不使循环特性和保存特性恶化而提高低温特性。According to the present invention, the low-temperature characteristics of the non-aqueous electrolyte secondary battery can be greatly improved, and more specifically, the low-temperature characteristics can be improved without deteriorating the cycle characteristics and storage characteristics.
电解液[2]:Electrolyte [2]:
根据本发明,可以提供循环特性和低温特性同时大幅提高的二次电池用非水电解液、以及使用该非水电解液的上述性能优异的二次电池。According to the present invention, it is possible to provide a non-aqueous electrolytic solution for a secondary battery in which both cycle characteristics and low-temperature characteristics are greatly improved, and a secondary battery using the non-aqueous electrolytic solution excellent in the above-mentioned performances.
电解液[3]:Electrolyte [3]:
根据本发明,可以提供低温输出特性得到大幅提高的二次电池用非水电解液以及二次电池。According to the present invention, it is possible to provide a non-aqueous electrolytic solution for a secondary battery and a secondary battery in which low-temperature output characteristics are greatly improved.
电解液[4]:Electrolyte [4]:
根据本发明,可以提供如下的二次电池用非水电解液以及二次电池:尽管该二次电池用非水电解液是由于溶剂的蒸发而使盐析出、或者闪点降低、在高温下内压容易上升等在含有大量低粘度溶剂时产生的问题少的电解液组成,但仍可以保持高的低温特性和输出特性。According to the present invention, the following non-aqueous electrolytic solution for secondary battery and secondary battery can be provided: although the non-aqueous electrolytic solution for secondary battery is due to the evaporation of solvent, salt is precipitated, or the flash point is lowered, and the internal The composition of the electrolyte solution has few problems when a large amount of low-viscosity solvent is contained, such as that the pressure is easy to rise, but it can still maintain high low-temperature characteristics and output characteristics.
电解液[5]:Electrolyte [5]:
根据本发明,可以提供输出特性大幅提高,并且高温保存特性和循环特性优异的二次电池用非水电解液以及二次电池。According to the present invention, it is possible to provide a non-aqueous electrolytic solution for a secondary battery and a secondary battery that have greatly improved output characteristics and are excellent in high-temperature storage characteristics and cycle characteristics.
电解液[6]:Electrolyte [6]:
根据本发明,可以提供高温保存特性和循环特性优异,并且输出特性大幅提高的二次电池用非水电解液。According to the present invention, it is possible to provide a non-aqueous electrolytic solution for secondary batteries that is excellent in high-temperature storage characteristics and cycle characteristics, and that has greatly improved output characteristics.
电解液[7]:Electrolyte [7]:
根据本发明,可以提供循环特性和低温特性同时大幅提高的二次电池。According to the present invention, it is possible to provide a secondary battery in which both cycle characteristics and low-temperature characteristics are greatly improved.
电解液[8]:Electrolyte [8]:
根据本发明,可以提供低温放电特性大幅提高,并且高温保存特性和循环特性优异的二次电池用非水电解液以及二次电池。According to the present invention, it is possible to provide a non-aqueous electrolytic solution for a secondary battery and a secondary battery that have greatly improved low-temperature discharge characteristics and are excellent in high-temperature storage characteristics and cycle characteristics.
电解液[9]:Electrolyte [9]:
根据本发明,可以提供同时满足高的速率特性和过充电时的安全性的二次电池。According to the present invention, it is possible to provide a secondary battery that satisfies both high rate characteristics and safety during overcharge.
结构[1]~[5]:Structure [1]~[5]:
根据本发明,可以得到高容量、长寿命、高输出,并且在过充电时安全性也高的锂二次电池,因此,可以提供特别适用于例如汽车用途等的大型电池的锂二次电池。According to the present invention, a lithium secondary battery with high capacity, long life, high output, and high safety during overcharge can be obtained. Therefore, a lithium secondary battery particularly suitable for large batteries such as automobiles can be provided.
结构[6]:Structure [6]:
根据本发明,可以得到高容量、长寿命、高输出,并且产生气体少,即使在过充电时安全性也高的锂二次电池,因此,可以提供特别适用于例如汽车用途等的大型电池的锂二次电池。According to the present invention, a high-capacity, long-life, high-output lithium secondary battery with low gas generation and high safety even when overcharged can be obtained. Therefore, it is possible to provide a lithium secondary battery that is particularly suitable for, for example, a large battery such as an automobile application. Lithium secondary battery.
具体实施方式Detailed ways
下面详细说明本发明的实施方式,但是下面记载的构成要件的说明是本发明的实施方式的一个例子(代表例),只要不超出其要点,则本发明并不限定于这些具体的内容。Embodiments of the present invention will be described in detail below, but the description of the constituent elements described below is an example (representative example) of embodiments of the present invention, and the present invention is not limited to these specific contents unless the gist thereof is exceeded.
本发明的锂二次电池至少包含:将正极和负极夹着微多孔膜隔板而构成的电极组、以及在非水溶剂中含有锂盐而形成的非水电解液,并且将它们装在电池外壳中,其中,The lithium secondary battery of the present invention at least includes: an electrode group formed by sandwiching a porous membrane separator between the positive electrode and the negative electrode, and a non-aqueous electrolytic solution formed by containing lithium salt in a non-aqueous solvent, and they are contained in the battery enclosure, in which,
所述正极和所述负极分别在集电体上形成活性物质层,该活性物质层含有能够吸留和放出锂离子的活性物质;The positive electrode and the negative electrode respectively form an active material layer on the current collector, and the active material layer contains an active material capable of absorbing and releasing lithium ions;
该非水电解液含有选自下述物质中的至少一种化合物(以下将它们简称为“特定化合物”),并且其在全部非水电解液中的含量为10ppm以上,所述物质包括:上述通式(1)表示的环状硅氧烷化合物、上述通式(2)表示的氟硅烷化合物、上述通式(3)表示的化合物、分子内具有S-F键的化合物、硝酸盐、亚硝酸盐、单氟磷酸盐、二氟磷酸盐、乙酸盐和丙酸盐;The non-aqueous electrolytic solution contains at least one compound selected from the following substances (hereinafter referred to as "specific compounds"), and its content in all non-aqueous electrolytic solutions is more than 10ppm, and the substances include: the above-mentioned Cyclic siloxane compounds represented by the general formula (1), fluorosilane compounds represented by the above general formula (2), compounds represented by the above general formula (3), compounds having an S-F bond in the molecule, nitrates, nitrites , monofluorophosphate, difluorophosphate, acetate and propionate;
并且,所述正极、负极、电解液或电池结构满足特定的条件。Also, the positive electrode, negative electrode, electrolyte solution or battery structure satisfies specific conditions.
下面,对本发明的锂二次电池更详细地进行说明。Next, the lithium secondary battery of the present invention will be described in more detail.
<正极><positive pole>
下面对本发明的锂二次电池中使用的正极进行说明。Next, the positive electrode used in the lithium secondary battery of the present invention will be described.
本发明中使用的正极只要是形成含有能够吸留和放出锂离子的活性物质的活性物质层的正极即可,没有特别限制,该正极优选选自下述正极[1]~正极[5]中的任意一种正极:The positive electrode used in the present invention is not particularly limited as long as it forms an active material layer containing an active material capable of occluding and releasing lithium ions. The positive electrode is preferably selected from the following positive electrodes [1] to positive electrodes [5]. Any positive pole of :
正极[1]:包含正极活性物质的正极,该正极活性物质含有锰;Positive electrode [1]: a positive electrode comprising a positive electrode active material containing manganese;
正极[2]:包含正极活性物质的正极,该正极活性物质具有下述组成式(4)表示的组成,Positive electrode [2]: a positive electrode comprising a positive electrode active material having a composition represented by the following composition formula (4),
LixNi(1-y-z)CoyMzO2 组成式(4)Li x Ni (1-yz) Co y M z O 2 composition formula (4)
[组成式(4)中,M表示选自Mn、Al、Fe、Ti、Mg、Cr、Ga、Cu、Zn和Nb中的至少一种元素,x表示满足0<x≤1.2的数,y表示满足0.05<y≤0.5的数,z表示满足0.01≤z≤0.5的数。];[In the composition formula (4), M represents at least one element selected from Mn, Al, Fe, Ti, Mg, Cr, Ga, Cu, Zn and Nb, x represents a number satisfying 0<x≤1.2, and y represents a number satisfying 0.05<y≤0.5, and z represents a number satisfying 0.01≤z≤0.5. ];
正极[3]:包含选自下述(a)~(d)中的任一种正极活性物质的正极,Positive electrode [3]: a positive electrode comprising any positive electrode active material selected from the following (a) to (d),
(a)BET比表面积为0.4m2/g~2m2/g的正极活性物质(a) A positive electrode active material having a BET specific surface area of 0.4m 2 /g to 2m 2 /g
(b)平均一次粒径为0.1μm~2μm的正极活性物质(b) Positive active material having an average primary particle size of 0.1 μm to 2 μm
(c)中值粒径d50为1μm~20μm的正极活性物质(c) A positive electrode active material having a median diameter d50 of 1 μm to 20 μm
(d)振实密度为1.3g/cm3~2.7g/cm3的正极活性物质;(d) a positive electrode active material with a tap density of 1.3g/cm 3 to 2.7g/cm 3 ;
正极[4]:满足选自下述(e)~(f)中的任意一个条件的正极,Positive electrode [4]: A positive electrode that satisfies any one of the following conditions (e) to (f),
(e)该正极是在集电体上形成含有正极活性物质、导电材料和粘合剂的正极活性物质层而制作的正极,其中,正极活性物质层中的导电材料的含量为6质量%~20质量%的范围(e) The positive electrode is a positive electrode made by forming a positive electrode active material layer containing a positive electrode active material, a conductive material and a binding agent on a current collector, wherein the content of the conductive material in the positive electrode active material layer is 6% by mass to 20% by mass range
(f)该正极是在集电体上形成含有正极活性物质和粘合剂的正极活性物质层而制作的正极,其中,正极活性物质层的密度为1.7g/cm3~3.5g/cm3的范围(f) The positive electrode is a positive electrode produced by forming a positive electrode active material layer containing a positive electrode active material and a binder on a current collector, wherein the density of the positive electrode active material layer is 1.7 g/cm 3 to 3.5 g/cm 3 range
(g)该正极是在集电体上形成含有正极活性物质和粘合剂的正极活性物质层而制作的正极,其中,正极活性物质层和集电体的厚度之比(注入非水电解液之前的一侧活性物质层的厚度)/(集电体的厚度)的值为1.6~20的范围;(g) The positive electrode is a positive electrode made by forming a positive electrode active material layer containing a positive electrode active material and a binder on the current collector, wherein the ratio of the thickness of the positive electrode active material layer to the current collector (injection of the non-aqueous electrolyte The value of the thickness of the active material layer on one side)/(the thickness of the current collector) is in the range of 1.6 to 20;
正极[5]:含有组成不同的2种以上正极活性物质的正极。Positive electrode [5]: A positive electrode containing two or more positive electrode active materials with different compositions.
下面,首先对本发明的锂二次电池中通常使用的正极进行说明。Next, first, a positive electrode generally used in the lithium secondary battery of the present invention will be described.
[正极活性物质][Positive electrode active material]
下面对正极中通常使用的正极活性物质进行说明。The positive electrode active material generally used in the positive electrode will be described below.
[[组成]][[composition]]
作为正极活性物质,只要是能够以电化学方式吸留和放出锂离子的物质即可,没有特别限制。优选含有锂和至少一种过渡金属的物质,例如可列举锂-过渡金属复合氧化物、含有锂的过渡金属磷酸化合物。The positive electrode active material is not particularly limited as long as it can electrochemically occlude and release lithium ions. A substance containing lithium and at least one transition metal is preferable, and examples thereof include lithium-transition metal composite oxides and lithium-containing transition metal phosphate compounds.
作为锂-过渡金属复合氧化物的过渡金属,优选V、Ti、Cr、Mn、Fe、Co、Ni、Cu等,作为具体例子,可列举LiCoO2等锂-钴复合氧化物;LiNiO2等锂-镍复合氧化物;LiMnO2、LiMn2O4、Li2MnO3等锂-锰复合氧化物;用Al、Ti、V、Cr、Mn、Fe、Co、Li、Ni、Cu、Zn、Mg、Ga、Zr、Si等其他金属来替代形成这些锂-过渡金属复合氧化物主体的过渡金属原子的一部分而得到的物质等。作为替代而得到的物质的具体例子,例如可列举LiNi0.5Mn0.5O2、LiNi0.85Co0.10Al0.05O2、LiNi0.33Co0.33Mn0.33O2、LiMn1.8Al0.2O4、LiMn1.5Ni0.5O4等。As the transition metal of the lithium-transition metal composite oxide, V, Ti, Cr, Mn, Fe, Co, Ni, Cu, etc. are preferable, and specific examples include lithium-cobalt composite oxides such as LiCoO 2 and lithium-cobalt composite oxides such as LiNiO 2 . - Nickel composite oxides; LiMnO 2 , LiMn 2 O 4 , Li 2 MnO 3 and other lithium-manganese composite oxides; Al, Ti, V, Cr, Mn, Fe, Co, Li, Ni, Cu, Zn, Mg , Ga, Zr, Si, and other metals instead of a part of the transition metal atoms forming the main body of these lithium-transition metal composite oxides. Specific examples of alternative substances include LiNi 0.5 Mn 0.5 O 2 , LiNi 0.85 Co 0.10 Al 0.05 O 2 , LiNi 0.33 Co 0.33 Mn 0.33 O 2 , LiMn 1.8 Al 0.2 O 4 , LiMn 1.5 Ni 0.5 O 4 etc.
作为含有锂的过渡金属磷酸化合物的过渡金属,优选V、Ti、Cr、Mn、Fe、Co、Ni、Cu等,作为具体例子,例如可列举LiFePO4、Li3Fe2(PO4)3、LiFeP2O7等磷酸铁类;LiCoPO4等磷酸钴类;用Al、Ti、V、Cr、Mn、Fe、Co、Li、Ni、Cu、Zn、Mg、Ga、Zr、Nb、Si等其他金属来替代形成这些含有锂的过渡金属磷酸化合物主体的过渡金属原子的一部分而得到的物质等。The transition metal of the lithium-containing transition metal phosphate compound is preferably V, Ti, Cr, Mn, Fe, Co, Ni, Cu, etc., and specific examples include LiFePO 4 , Li 3 Fe 2 (PO 4 ) 3 , Iron phosphate such as LiFeP 2 O 7 ; Cobalt phosphate such as LiCoPO 4 ; Al, Ti, V, Cr, Mn, Fe, Co, Li, Ni, Cu, Zn, Mg, Ga, Zr, Nb, Si and others A metal is used instead of a part of the transition metal atoms forming the main body of these transition metal phosphate compounds containing lithium, and the like.
[[表面包覆]][[Cladding]]
另外,优选在这些正极活性物质的表面附着有与芯的正极活性物质组成不同的物质。作为附着在表面的物质(以下,简称为“表面附着物质”),例如可列举氧化铝、氧化硅、氧化钛、氧化锆、氧化镁、氧化钙、氧化硼、氧化锑、氧化铋等氧化物;硫酸锂、硫酸钠、硫酸钾、硫酸镁、硫酸钙、硫酸铝等硫酸盐;碳酸锂、碳酸钙、碳酸镁等碳酸盐等。In addition, it is preferable that a substance having a composition different from that of the positive electrode active material of the core adheres to the surface of these positive electrode active materials. Examples of substances adhering to the surface (hereinafter simply referred to as "surface adhering substances") include oxides such as aluminum oxide, silicon oxide, titanium oxide, zirconium oxide, magnesium oxide, calcium oxide, boron oxide, antimony oxide, and bismuth oxide. ; Lithium sulfate, sodium sulfate, potassium sulfate, magnesium sulfate, calcium sulfate, aluminum sulfate and other sulfates; lithium carbonate, calcium carbonate, magnesium carbonate and other carbonates.
这些表面附着物质可以通过下述方法附着在正极活性物质表面,所述方法包括:例如,使表面附着物质溶解或悬浮在溶剂中,再含浸添加在正极活性物质中并进行干燥的方法;使表面附着物质前体溶解或悬浮在溶剂中,再含浸添加在正极活性物质中,然后通过加热等使之反应的方法;将表面附着物质添加到正极活性物质前体中,同时进行烧结的方法等。These surface attachment substances can be attached to the surface of the positive electrode active material by the following methods, which include: for example, dissolving or suspending the surface attachment substances in a solvent, then impregnating them into the positive electrode active material and drying them; making the surface A method in which the attachment substance precursor is dissolved or suspended in a solvent, impregnated and added to the positive electrode active material, and then reacted by heating or the like; a method in which the surface attachment substance is added to the positive electrode active material precursor and simultaneously sintered, etc.
作为表面附着物质的量,相对于正极活性物质以质量计,其下限优选为0.1ppm以上,更优选为1ppm以上,进一步优选为10ppm以上;其上限优选为20%以下,更优选为10%以下,进一步优选为5%以下。由表面附着物质可以抑制非水电解液在正极活性物质表面的氧化反应,从而可以提高电池寿命,但其附着量过少时,不能充分表现出其效果,而过多时,由于妨碍锂离子的出入,因此有时阻抗增加。As the amount of surface-attached substances, its lower limit is preferably more than 0.1ppm, more preferably more than 1ppm, more preferably more than 10ppm with respect to the positive electrode active material by mass; its upper limit is preferably less than 20%, more preferably less than 10% , and more preferably 5% or less. The oxidation reaction of the non-aqueous electrolyte on the surface of the positive electrode active material can be inhibited by the surface attachment substance, thereby improving the battery life, but when the amount of attachment is too small, the effect cannot be fully exhibited, and when it is too much, due to hindering the entry and exit of lithium ions, Therefore, the impedance sometimes increases.
[[形状]][[shape]]
本发明中的正极活性物质的粒子形状可使用以往使用的块状、多面体状、球状、椭圆球状、板状、针状、柱状等,其中,优选一次粒子凝聚而形成二次粒子,且该二次粒子的形状为球状或椭圆球状。通常,电化学元件随着其充放电,电极中的活性物质膨胀收缩,因此容易发生由于该应力引起的活性物质的破坏或导电通路切断等劣化。因此,与只是一次粒子的单一粒子活性物质相比,优选一次粒子凝聚而形成二次粒子的情况,因为形成二次粒子可以缓和膨胀收缩的应力,从而防止劣化。另外,与板状等轴取向性的粒子相比,优选球状或椭圆球状的粒子,因为球状或椭圆球状的粒子在电极成型时的取向少,充放电时的电极的膨胀收缩也少,并且在制作电极时与导电剂的混合时,也容易均匀混合。The particle shape of the positive electrode active material in the present invention can use block shape, polyhedron shape, spherical shape, ellipsoidal shape, plate shape, needle shape, columnar shape etc. used in the past, among them, preferred primary particle aggregates and forms secondary particle, and the two The shape of the secondary particles is spherical or ellipsoidal. In general, the active material in the electrode expands and shrinks as the electrochemical element is charged and discharged, and thus degradation of the active material due to this stress, such as destruction of the active material or interruption of the conductive path, tends to occur. Therefore, it is preferable to agglomerate primary particles to form secondary particles rather than single-particle active material with only primary particles, because the formation of secondary particles can relieve the stress of expansion and contraction, thereby preventing deterioration. In addition, spherical or ellipsoidal particles are preferred over plate-like equiaxially oriented particles, because spherical or ellipsoidal particles have less orientation during electrode molding and less expansion and contraction of the electrode during charging and discharging, and in It is also easy to mix uniformly with the conductive agent when making electrodes.
[[振实密度]][[Tap Density]]
正极活性物质的振实密度通常为1.3g/cm3以上,优选为1.5g/cm3以上,更优选为1.6g/cm3以上,最优选为1.7g/cm3以上。正极活性物质的振实密度如果低于上述下限,则在形成正极活性物质层时,需要的分散介质量增加,同时导电材料或粘合剂的必要量增加,正极活性物质在正极活性物质层中的填充率受到制约,电池容量有时受到制约。通过使用振实密度高的复合氧化物粉末,可以形成高密度的正极活性物质层。一般来说,振实密度越大越优选,没有特别的上限,但如果振实密度太大,则正极活性物质层内的以非水电解液为介质的锂离子的扩散成为决定速度的因素,负本发明的锂二次电池的负极[7]的负极活性物质中使用的元素Z外在锂吸留物质(F)中,吸留金属(A)和/或锂吸留合金(B’)与碳质物质(E’)的质量比通常为20/80以上,优选为50/50以上,更优选为80/20以上,特别优选为90/10以上,且优选为99.9/0.1以下,更优选为99/1以下,特别优选为98/2以下的范围。如果超过上述范围,则有时不能获得具有碳质物质(E’)的效果,而如果低于上述范围,则每单位质量的容量增加的效果有时变小。优选吸留金属(A)和/或锂吸留合金(B’)相对于全部元素Z外在锂吸留物质(F)为20质量%以上。The tap density of the positive electrode active material is usually 1.3 g/cm 3 or higher, preferably 1.5 g/cm 3 or higher, more preferably 1.6 g/cm 3 or higher, and most preferably 1.7 g/cm 3 or higher. If the tap density of the positive electrode active material is lower than the above-mentioned lower limit, then when forming the positive electrode active material layer, the amount of dispersion medium required increases, and the necessary amount of conductive material or binder increases simultaneously, and the positive electrode active material is in the positive electrode active material layer. The filling rate of the battery is restricted, and the battery capacity is sometimes restricted. By using a composite oxide powder with a high tap density, a high-density positive electrode active material layer can be formed. Generally speaking, the larger the tap density is, the more preferable, there is no special upper limit, but if the tap density is too large, the diffusion of lithium ions in the positive electrode active material layer with the non-aqueous electrolyte as the medium becomes a factor determining the speed, and the negative The element Z used in the negative electrode active material of the negative electrode [7] of the lithium secondary battery of the present invention is contained in the lithium occlusion material (F), and the occlusion metal (A) and/or lithium occlusion alloy (B') and The mass ratio of the carbonaceous substance (E') is usually 20/80 or more, preferably 50/50 or more, more preferably 80/20 or more, particularly preferably 90/10 or more, and preferably 99.9/0.1 or less, more preferably 99/1 or less, particularly preferably 98/2 or less. If it exceeds the above range, the effect of having the carbonaceous substance (E') may not be obtained, and if it is below the above range, the effect of increasing the capacity per unit mass may become small. Preferably, the occlusion metal (A) and/or lithium occlusion alloy (B′) is 20% by mass or more relative to the total element Z external lithium occlusion substance (F).
荷特性有时容易降低,因此,振实密度通常为2.9g/cm3以下,优选为2.7g/cm3以下,更优选为2.5g/cm3以下。Since the charge characteristics tend to decrease in some cases, the tap density is usually 2.9 g/cm 3 or less, preferably 2.7 g/cm 3 or less, more preferably 2.5 g/cm 3 or less.
在本发明中,振实密度如下定义:使试料通过孔径300μm的筛子落到20cm3的振实容器(tapping cell)中,充满容器容积之后,使用粉末密度测定器(例如,Seishin(セイシン)企业社制造的Tap densor(タツプデンサ一)),进行1000次冲程长度10mm的振动,将由此时的体积和试料的重量求出的密度作为振实密度。In the present invention, the tap density is defined as follows: the sample is dropped into a 20 cm 3 tapping container (tapping cell) through a sieve with an aperture of 300 μm. Tap densor manufactured by the company was vibrated 1000 times with a stroke length of 10 mm, and the density obtained from the volume at that time and the weight of the sample was taken as the tap density.
[[中值粒径d50]][[Median particle size d 50 ]]
正极活性物质的粒子的中值粒径d50(一次粒子凝聚而形成二次粒子时,为二次粒子直径)通常为0.1μm以上,优选为0.5μm以上,更优选为1μm以上,最优选为3μm以上,其上限通常为20μm以下,优选为18μm以下,更优选为16μm以下,最优选为15μm以下。如果中值粒径d50低于上述下限,则有时不能得到高振实密度的产品,而如果超过上限,则由于粒子内的锂的扩散需要时间,因此有时会产生电池性能降低,或者在制成电池的正极、也就是将活性物质和导电剂或粘合剂等在溶剂中浆料化再涂布成薄膜状时,产生条纹等问题。这里,通过混合2种以上具有不同的中值粒径d50的正极活性物质,可以进一步提高制作正极时的填充性。The median diameter d50 of the particles of the positive electrode active material (when the primary particles are aggregated to form secondary particles, the secondary particle diameter) is usually 0.1 μm or more, preferably 0.5 μm or more, more preferably 1 μm or more, and most preferably 0.1 μm or more. 3 μm or more, and its upper limit is usually 20 μm or less, preferably 18 μm or less, more preferably 16 μm or less, most preferably 15 μm or less. If the median particle diameter d50 is lower than the above-mentioned lower limit, a product with a high tap density may not be obtained, and if it exceeds the upper limit, since the diffusion of lithium in the particles takes time, the performance of the battery may sometimes decrease, or the resulting battery may be damaged. Streaks and other problems occur when the active material and conductive agent or binder are slurried in a solvent and then coated into a film. Here, by mixing two or more positive electrode active materials having different median diameters d 50 , the filling property at the time of producing the positive electrode can be further improved.
另外,本发明中的中值粒径d50可以利用公知的激光衍射/散射式粒度分布测定装置来测定。使用HORIBA公司制造的LA-920作为粒度分布计时,作为测定时使用的分散介质,使用0.1质量%六偏磷酸钠水溶液,进行5分钟超声波分散后,设定测定折射率1.24进行测定。In addition, the median diameter d50 in the present invention can be measured using a known laser diffraction/scattering type particle size distribution analyzer. LA-920 manufactured by HORIBA was used as a particle size distribution meter, and as a dispersion medium used in the measurement, 0.1 mass % sodium hexametaphosphate aqueous solution was used, ultrasonic dispersion was carried out for 5 minutes, and the measurement refractive index was set to 1.24 for measurement.
[[平均一次粒径]][[average primary particle size]]
一次粒子凝聚而形成二次粒子时,作为正极活性物质的平均一次粒径,通常为0.01μm以上,优选为0.05μm以上,更优选为0.08μm以上,最优选为0.1μm以上,且通常为3μm以下,优选为2μm以下,更优选为1μm以下,最优选为0.6μm以下。如果平均一次粒径超过上述上限,则难以形成球状的二次粒子,对粉末填充性带来不良影响,或者比表面积大幅降低,因此,输出特性等电池性能降低的可能性有时变高。相反,如果平均一次粒径低于上述下限,则通常由于结晶不完全,因此有时产生充放电的可逆性差等问题。When primary particles are aggregated to form secondary particles, the average primary particle diameter of the positive electrode active material is usually 0.01 μm or more, preferably 0.05 μm or more, more preferably 0.08 μm or more, most preferably 0.1 μm or more, and usually 3 μm or less, preferably 2 μm or less, more preferably 1 μm or less, and most preferably 0.6 μm or less. When the average primary particle size exceeds the above upper limit, it is difficult to form spherical secondary particles, which adversely affects the powder fillability, or the specific surface area is greatly reduced, and therefore battery performance such as output characteristics may decrease. On the contrary, if the average primary particle diameter is less than the above-mentioned lower limit, problems such as poor charge-discharge reversibility may occur due to incomplete crystallization.
另外,一次粒径可通过使用扫描电子显微镜(SEM)的观察来测定。具体地,通过如下方法求出:在倍率10000倍的照片中,对任意的50个一次粒子求出水平方向的直线上的由一次粒子的左右边界线产生的切片的最长值,并取其平均值。In addition, the primary particle size can be measured by observation using a scanning electron microscope (SEM). Specifically, it is obtained by the following method: in a photograph with a magnification of 10,000 times, for any 50 primary particles, the longest value of the slice generated by the left and right boundary lines of the primary particles on a straight line in the horizontal direction is obtained, and the maximum value is obtained. average value.
[[BET比表面积]][[BET specific surface area]]
本发明的提供给锂二次电池的正极活性物质的BET比表面积通常为0.2m2/g以上,优选为0.3m2/g以上,更优选为0.4m2/g以上,其上限通常为4.0m2/g以下,优选为2.5m2/g以下,更优选为1.5m2/g以下。如果BET比表面积比该范围小,则电池性能容易降低,而如果BET比表面积比该范围大,则振实密度难以提高,在形成正极活性物质时的涂布性上有时容易产生问题。The BET specific surface area of the positive electrode active material provided for the lithium secondary battery of the present invention is usually 0.2 m 2 /g or more, preferably 0.3 m 2 /g or more, more preferably 0.4 m 2 /g or more, and the upper limit is usually 4.0 m 2 /g or less, preferably 2.5 m 2 /g or less, more preferably 1.5 m 2 /g or less. If the BET specific surface area is smaller than this range, the battery performance is likely to decrease, and if the BET specific surface area is larger than this range, it is difficult to increase the tap density, and problems may easily occur in coating properties when forming a positive electrode active material.
BET比表面积定义为如下的值:使用表面积计(例如,大仓理研制造的全自动表面积测定装置),在氮流通下在150℃对试料进行30分钟预干燥,然后使用正确调整为氮相对于大气压的相对压力值为0.3的氮氦混合气体,通过采用气体流动法进行的氮吸附BET 1点法而测定的值。The BET specific surface area is defined as the following value: Using a surface area meter (for example, a fully automatic surface area measuring device manufactured by Okura Riken), the sample is pre-dried at 150°C for 30 minutes under nitrogen flow, and then adjusted to the nitrogen relative The value measured by the nitrogen adsorption BET 1-point method using the gas flow method for a nitrogen-helium mixed gas with a relative pressure value of 0.3 at atmospheric pressure.
[[制造法]][[Manufacturing method]]
作为正极活性物质的制造法,使用作为无机化合物的制造法的通常的方法。特别是在制作球状或椭圆球状的活性物质时,可考虑各种方法,例如可列举如下方法:将过渡金属硝酸盐、过渡金属硫酸盐等过渡金属原料物质和根据需要使用的其他元素的原料物质溶解或粉碎分散在水等溶剂中,边搅拌边调节pH,制作并获得球状的前体,根据需要将其干燥后,加入LiOH、Li2CO3、LiNO3等Li源,在高温下烧结而得到活性物质的方法;将过渡金属硝酸盐、过渡金属硫酸盐、过渡金属氢氧化物、过渡金属氧化物等过渡金属原料物质和根据需要使用的其他元素的原料物质溶解或粉碎分散在水等溶剂中,再通过喷雾干燥器等将其干燥成型,制成球状或椭圆球状的前体,然后在其中加入LiOH、Li2CO3、LiNO3等Li源,在高温下烧结而得到活性物质的方法;以及将过渡金属硝酸盐、过渡金属硫酸盐、过渡金属氢氧化物、过渡金属氧化物等过渡金属原料物质和LiOH、Li2CO3、LiNO3等Li源以及根据需要使用的其他元素的原料物质溶解或粉碎分散在水等溶剂中,再通过喷雾干燥器等将其干燥成型,制成球状或椭圆球状的前体,然后在高温下将其烧结而得到活性物质的方法等。As a method for producing the positive electrode active material, a common method for producing an inorganic compound is used. In particular, when producing a spherical or ellipsoidal active material, various methods can be considered, for example, the following method can be cited: a transition metal raw material such as a transition metal nitrate, a transition metal sulfate, and a raw material of other elements used as needed Dissolve or pulverize and disperse in solvents such as water, adjust the pH while stirring, make and obtain spherical precursors, dry them as needed, add Li sources such as LiOH, Li 2 CO 3 , LiNO 3 , and sinter at high temperature to form A method of obtaining an active material; dissolving or pulverizing raw materials of transition metals such as transition metal nitrates, transition metal sulfates, transition metal hydroxides, transition metal oxides, and other elements used as needed in a solvent such as water , and then dry it by spray dryer to form a spherical or ellipsoidal precursor, and then add Li sources such as LiOH, Li 2 CO 3 , LiNO 3 to it, and sinter at a high temperature to obtain an active material. ; and transition metal raw materials such as transition metal nitrates, transition metal sulfates, transition metal hydroxides, transition metal oxides and Li sources such as LiOH, Li 2 CO 3 , LiNO 3 and other elements used as required A method in which the substance is dissolved or pulverized and dispersed in a solvent such as water, and then dried and shaped by a spray dryer to form a spherical or ellipsoidal precursor, and then sintered at a high temperature to obtain an active material.
[正极的结构][Structure of positive electrode]
下面,对本发明中使用的正极的结构进行叙述。Next, the structure of the positive electrode used in the present invention will be described.
[[电极结构和制作法]][[Electrode structure and fabrication method]]
本发明的锂二次电池中使用的正极是在集电体上形成含有正极活性物质和粘合剂的正极活性物质层而制作的。使用正极活性物质的正极的制造可以按照通常的方法进行。即,将正极活性物质和粘合剂、以及根据需要使用的导电材料和增稠剂等干式混合,制成片状,再将得到的片状物压粘在正极集电体上,或者将这些材料溶解或分散在液体介质中,制成浆料,再将该浆料涂布在正极集电体上并进行干燥,由此可以在集电体上形成正极活性物质层,从而可以得到正极。The positive electrode used in the lithium secondary battery of the present invention is produced by forming a positive electrode active material layer containing a positive electrode active material and a binder on a current collector. The production of the positive electrode using the positive electrode active material can be performed according to a usual method. That is, the positive electrode active material and the binder, as well as the conductive material and thickener used as required, are dry mixed, made into a sheet, and then the obtained sheet is pressed and bonded on the positive electrode collector, or the These materials are dissolved or dispersed in a liquid medium to form a slurry, and then the slurry is coated on the positive electrode collector and dried, thereby forming a positive electrode active material layer on the current collector, thereby obtaining a positive electrode. .
在本发明中,正极活性物质层中的正极活性物质的含量通常为10质量%以上,优选为30质量%以上,特别优选为50质量%以上。另外,其上限通常为99.9质量%以下,优选为99质量%以下。如果正极活性物质层中的正极活性物质粉末的含量很低,则电容有时变得不充分。相反如果含量过高,则正极的强度有时不足。另外,正极活性物质可以单独使用一种,也可以以任意的组合和比例组合使用组成不同或粉末物性不同的2种以上的正极活性物质。In the present invention, the content of the positive electrode active material in the positive electrode active material layer is usually 10% by mass or more, preferably 30% by mass or more, particularly preferably 50% by mass or more. In addition, the upper limit thereof is usually 99.9% by mass or less, preferably 99% by mass or less. When the content of the positive electrode active material powder in the positive electrode active material layer is too low, the capacitance may become insufficient. Conversely, if the content is too high, the strength of the positive electrode may be insufficient. In addition, one type of positive electrode active material may be used alone, or two or more positive electrode active materials having different compositions or different powder properties may be used in combination in any combination and ratio.
[[压实]][[Compaction]]
为了提高正极活性物质的填充密度,通过涂布、干燥而得到的正极活性物质层优选利用手动压力机、辊压机等进行压实。正极活性物质层的密度的下限优选为1g/cm3以上,更优选为1.5g/cm3以上,进一步优选为2g/cm3以上,其上限优选为4g/cm3以下,更优选为3.5g/cm3以下,进一步优选为3g/cm3以下的范围。如果超过该范围,则非水电解液向集电体/活性物质界面附近的渗透性降低,特别是在高电流密度下的充放电特性有时降低。另外,如果低于该范围,则活性物质间的导电性降低,电池阻抗有时增大。In order to increase the packing density of the positive electrode active material, the positive electrode active material layer obtained by coating and drying is preferably compacted using a manual press, a roll press, or the like. The lower limit of the density of the positive electrode active material layer is preferably 1 g/cm or more, more preferably 1.5 g/cm or more, even more preferably 2 g/cm or more, and the upper limit is preferably 4 g/cm or less, more preferably 3.5 g /cm 3 or less, more preferably 3 g/cm 3 or less. If this range is exceeded, the permeability of the non-aqueous electrolyte solution to the vicinity of the current collector/active material interface decreases, and in particular, the charge-discharge characteristics at high current densities sometimes decrease. Moreover, if it is less than this range, the electrical conductivity between active materials will fall, and battery impedance may increase.
[[导电材料]][[Conductive Material]]
作为导电材料,可以任意使用公知的导电材料。作为具体的例子,可列举铜、镍等金属材料;天然石墨、人造石墨等石墨;乙炔黑等炭黑;针状焦等无定形碳等碳材料等。这些物质可以单独使用一种,也可以以任意的组合和比例组合使用2种以上。As the conductive material, known conductive materials can be used arbitrarily. Specific examples include metal materials such as copper and nickel; graphite such as natural graphite and artificial graphite; carbon black such as acetylene black; and carbon materials such as amorphous carbon such as needle coke. One of these substances may be used alone, or two or more of them may be used in any combination and ratio.
导电材料在正极活性物质层中的使用比例通常为0.01重量%以上,优选为0.1重量%以上,更优选为1重量%以上,其上限通常为50重量%以下,优选为30重量%以下,更优选为15重量%以下。如果含量低于该范围,则导电性有时不充分。反之,如果含量高于该范围,则电池容量有时降低。The use ratio of the conductive material in the positive electrode active material layer is usually more than 0.01% by weight, preferably more than 0.1% by weight, more preferably more than 1% by weight, and the upper limit is usually less than 50% by weight, preferably less than 30% by weight, more preferably Preferably it is 15% by weight or less. If the content is less than this range, the conductivity may be insufficient. On the contrary, if the content is higher than the range, the battery capacity may decrease.
[[粘合剂]][[Binder]]
作为在制造正极活性物质层时使用的粘合剂,没有特别限定,使用涂布法时,只要是可溶解或分散在电极制造时使用的液体介质中的材料即可,作为具体例子,可列举聚乙烯、聚丙烯、聚对苯二甲酸乙二醇酯、聚甲基丙烯酸甲酯、芳香族聚酰胺、纤维素、硝基纤维素等树脂类高分子;SBR(苯乙烯-丁二烯橡胶)、NBR(丙烯腈-丁二烯橡胶)、氟橡胶、异戊二烯橡胶、丁二烯橡胶、乙烯-丙烯橡胶等橡胶状高分子;苯乙烯-丁二烯-苯乙烯嵌段共聚物或其加氢物、EPDM(乙烯-丙烯-二烯烃三元共聚物)、苯乙烯-乙烯-丁二烯-乙烯共聚物、苯乙烯-异戊二烯-苯乙烯嵌段共聚物或其加氢物等热塑性弹性体状高分子;间规立构1,2-聚丁二烯、聚乙酸乙烯酯、乙烯-乙酸乙烯酯共聚物、丙烯-α-烯烃共聚物等软质树脂状高分子;聚偏氟乙烯(PVdF)、聚四氟乙烯、氟化聚偏氟乙烯、聚四氟乙烯-乙烯共聚物等氟类高分子;具有碱金属离子(特别是锂离子)的离子传导性的高分子组合物等。另外,这些物质可以单独使用一种,也可以以任意的组合和比例组合使用2种以上。The binder used in the production of the positive electrode active material layer is not particularly limited. When using the coating method, any material can be dissolved or dispersed in the liquid medium used in the production of the electrode. As a specific example, Polyethylene, polypropylene, polyethylene terephthalate, polymethyl methacrylate, aromatic polyamide, cellulose, nitrocellulose and other resin polymers; SBR (styrene-butadiene rubber ), NBR (acrylonitrile-butadiene rubber), fluororubber, isoprene rubber, butadiene rubber, ethylene-propylene rubber and other rubber-like polymers; styrene-butadiene-styrene block copolymer Or its hydrogenated product, EPDM (ethylene-propylene-diene terpolymer), styrene-ethylene-butadiene-ethylene copolymer, styrene-isoprene-styrene block copolymer or its addition Thermoplastic elastomeric polymers such as hydrogen compounds; soft resinous polymers such as syndiotactic 1,2-polybutadiene, polyvinyl acetate, ethylene-vinyl acetate copolymer, propylene-α-olefin copolymer, etc. ; Polyvinylidene fluoride (PVdF), polytetrafluoroethylene, fluorinated polyvinylidene fluoride, polytetrafluoroethylene-ethylene copolymer and other fluorine polymers; ion conductivity with alkali metal ions (especially lithium ions) Polymer composition, etc. In addition, these substances may be used individually by 1 type, and may be used combining 2 or more types by arbitrary combinations and ratios.
粘合剂在正极活性物质层中的比例通常为0.1质量%以上,优选为1质量%以上,更优选为3质量%以上,其上限通常为80质量%以下,优选为60质量%以下,更优选为40质量%以下,最优选10质量%以下。如果粘合剂的比例太低,则不能充分保持正极活性物质,正极的机械强度不足,循环特性等电池性能恶化。另一方面,如果粘合剂的比例太高,则有时会导致电池容量或导电性降低。The ratio of the binder in the positive electrode active material layer is usually 0.1% by mass or more, preferably 1% by mass or more, more preferably 3% by mass or more, and the upper limit is usually 80% by mass or less, preferably 60% by mass or less, more preferably Preferably it is 40% by mass or less, most preferably 10% by mass or less. If the ratio of the binder is too low, the positive electrode active material cannot be sufficiently retained, the mechanical strength of the positive electrode is insufficient, and battery performance such as cycle characteristics deteriorates. On the other hand, if the ratio of the binder is too high, it sometimes leads to a decrease in battery capacity or conductivity.
[[液体介质]][[liquid medium]]
作为用于形成浆料的液体介质,只要是能够溶解或分散正极活性物质、导电剂、粘合剂以及根据需要使用的增稠剂的溶剂,其种类就没有特别限制,可以使用水系溶剂和有机溶剂中的任一种。The liquid medium used to form the slurry is not particularly limited as long as it is a solvent capable of dissolving or dispersing the positive electrode active material, conductive agent, binder and, if necessary, a thickener. Aqueous solvents and organic solvents can be used. any of the solvents.
作为水系介质,例如可列举水、醇和水的混合溶剂等。作为有机溶剂,例如可列举己烷等脂肪族烃类;苯、甲苯、二甲苯、甲基萘等芳香族烃类;喹啉、吡啶等杂环化合物;丙酮、甲乙酮、环己酮等酮类;乙酸甲酯、丙烯酸甲酯等酯类;二亚乙基三胺、N,N-二甲基氨基丙胺等胺类;乙醚、环氧丙烷、四氢呋喃(THF)等醚类;N-甲基吡咯烷酮(NMP)、二甲基甲酰胺、二甲基乙酰胺等酰胺类;六甲基膦酰胺、二甲亚砜等非质子性极性溶剂等。As an aqueous medium, water, the mixed solvent of alcohol, and water, etc. are mentioned, for example. Examples of organic solvents include aliphatic hydrocarbons such as hexane; aromatic hydrocarbons such as benzene, toluene, xylene, and methylnaphthalene; heterocyclic compounds such as quinoline and pyridine; ketones such as acetone, methyl ethyl ketone, and cyclohexanone. ; Methyl acetate, methyl acrylate and other esters; Diethylenetriamine, N, N-dimethylaminopropylamine and other amines; Diethyl ether, propylene oxide, tetrahydrofuran (THF) and other ethers; N-methyl Amides such as pyrrolidone (NMP), dimethylformamide, and dimethylacetamide; aprotic polar solvents such as hexamethylphosphonamide and dimethyl sulfoxide, etc.
特别是在使用水系溶剂时,优选使用增稠剂和苯乙烯-丁二烯橡胶(SBR)等的胶乳来进行浆料化。增稠剂通常是用于调节浆料的粘度而使用的。作为增稠剂,没有特别限制,具体地,可列举羧甲基纤维素、甲基纤维素、羟甲基纤维素、乙基纤维素、聚乙烯醇、氧化淀粉、磷酸化淀粉、酪蛋白以及它们的盐等。它们可以单独使用一种,也可以以任意的组合和比例组合使用2种以上。另外,添加增稠剂时,增稠剂相对于活性物质的比例通常为0.1质量%以上,优选为0.5质量%以上,更优选为0.6质量%以上,其上限通常为5质量%以下,优选为3质量%以下,更优选为2质量%以下的范围。如果低于该范围,则涂布性有时显著降低。而如果超过该范围,则活性物质在正极活性物质层中所占的比例降低,有时会产生电池的容量降低的问题或正极活性物质间的阻抗增大的问题。In particular, when using an aqueous solvent, it is preferable to form a slurry using a thickener and a latex such as styrene-butadiene rubber (SBR). Thickeners are generally used to adjust the viscosity of slurry. The thickener is not particularly limited, and specifically, carboxymethylcellulose, methylcellulose, hydroxymethylcellulose, ethylcellulose, polyvinyl alcohol, oxidized starch, phosphorylated starch, casein and their salt etc. These may be used individually by 1 type, and may combine and use 2 or more types by arbitrary combinations and ratios. In addition, when adding a thickener, the ratio of the thickener to the active material is usually 0.1% by mass or more, preferably 0.5% by mass or more, more preferably 0.6% by mass or more, and the upper limit is usually 5% by mass or less, preferably 3% by mass or less, more preferably in the range of 2% by mass or less. If it is less than this range, coatability may fall remarkably. On the other hand, if this range is exceeded, the proportion of the active material in the positive electrode active material layer decreases, which may cause problems such as a decrease in battery capacity or an increase in impedance between positive electrode active materials.
[[集电体]][[Collector]]
作为正极集电体的材质,没有特别限制,可以任意地使用公知的材料。作为具体例子,可列举铝、不锈钢、镀镍、钛、钽等金属材料;碳布、碳纸等碳材料。其中,优选金属材料,特别优选铝。The material of the positive electrode current collector is not particularly limited, and known materials can be used arbitrarily. Specific examples include metal materials such as aluminum, stainless steel, nickel plating, titanium, and tantalum; and carbon materials such as carbon cloth and carbon paper. Among them, metal materials are preferable, and aluminum is particularly preferable.
作为集电体的形状,在金属材料的情况下,可列举金属箔、金属圆柱、金属线圈、金属板、金属薄膜、膨胀合金(エキスパンドメタル)、穿孔金属、发泡金属等;碳材料的情况下,可列举碳板、碳薄膜、碳圆柱等。这些当中,优选金属薄膜。另外,薄膜还可以适当地形成网状。薄膜的厚度是任意的,但通常为1μm以上,优选为3μm以上,更优选为5μm以上,并且通常为1mm以下,优选为100μm以下,更优选为50μm以下。薄膜如果比该范围薄,则有时作为集电体的必要的强度不足。相反,薄膜如果比该范围厚,则有时会损害操作性。As the shape of the current collector, in the case of metal materials, metal foil, metal cylinder, metal coil, metal plate, metal film, expanded alloy (Ekispand Metal), perforated metal, foamed metal, etc.; Below, carbon plates, carbon thin films, carbon cylinders, etc. are mentioned. Among these, metal thin films are preferable. In addition, the film can also be properly formed into a network. The thickness of the film is arbitrary, but it is usually 1 μm or more, preferably 3 μm or more, more preferably 5 μm or more, and usually 1 mm or less, preferably 100 μm or less, more preferably 50 μm or less. If the thin film is thinner than this range, the required strength as a current collector may be insufficient. On the contrary, when the film is thicker than this range, the handleability may be impaired.
集电体和正极活性物质层的厚度之比没有特别限定,(注入非水电解液之前的一侧的活性物质层厚度)/(集电体的厚度)优选为150以下,更优选为20以下,最优选为10以下,其下限优选为0.1以上,更优选为0.4以上,最优选为1以上的范围。如果超过该范围,则在高电流密度充放电时,集电体有时会因为焦耳热而产生发热。如果低于该范围,则有时集电体相对于正极活性物质的体积比增加,电池的容量减少。The ratio of the thickness of the current collector to the positive electrode active material layer is not particularly limited, and (the thickness of the active material layer on the side before injecting the nonaqueous electrolyte)/(thickness of the current collector) is preferably 150 or less, more preferably 20 or less , is most preferably 10 or less, and its lower limit is preferably 0.1 or more, more preferably 0.4 or more, and most preferably 1 or more. If this range is exceeded, the current collector may generate heat due to Joule heat during high current density charge and discharge. If it is less than this range, the volume ratio of the current collector to the positive electrode active material may increase and the capacity of the battery may decrease.
[[电极面积]][[Electrode Area]]
使用本发明的非水电解液时,从高输出以及提高高温时的稳定性的观点来看,优选正极活性物质层的面积比电池外壳的外表面积大。具体地,上述正极的电极面积总和相对于二次电池的外壳的表面积以面积比计,优选为15倍以上,更优选为40倍以上。所谓外壳的外表面积,在有底方形形状的情况下,是指除了端子的突起部分的填充有发电要素的外壳部分由长、宽和厚度的尺寸计算而求出的总面积。在有底圆筒形状的情况下,是将除了端子的突起部分的填充有发电要素的外壳部分近似为圆筒而得到的几何表面积。所谓正极的电极面积的总和,是与包含负极活性物质的复层材料层(合材)对置的正极复层材料层的几何表面积,在夹着集电体箔在两面形成正极复层材料层的结构中,是指分别计算各个面的面积的总和。When using the nonaqueous electrolytic solution of the present invention, it is preferable that the area of the positive electrode active material layer is larger than the outer surface area of the battery case from the viewpoint of high output and improved stability at high temperature. Specifically, the total electrode area of the above-mentioned positive electrode is preferably 15 times or more, more preferably 40 times or more, in terms of area ratio with respect to the surface area of the casing of the secondary battery. The outer surface area of the housing refers to the total area of the housing part filled with the power generating elements except the protruding part of the terminal calculated from the dimensions of length, width and thickness in the case of a square shape with a bottom. In the case of a bottomed cylindrical shape, it is a geometrical surface area obtained by approximating the housing portion filled with the power generating element except for the protruding portion of the terminal as a cylinder. The sum of the electrode areas of the so-called positive pole is combined with the composite material layer (composite material) that contains the negative active material. ) The geometric surface area of the positive electrode cladding material layer facing each other refers to the sum of the areas of the respective surfaces in the structure in which the positive electrode cladding material layer is formed on both sides of the current collector foil.
[[放电容量]][[Discharge capacity]]
使用本发明的二次电池用非水电解液时,装在1个二次电池的电池外壳中的电池要素所具有的电容(将电池由满充电状态放电至放电状态时的电容)如果为3安培小时(Ah)以上,则低温放电特性的提高效果变大,故优选。When using the non-aqueous electrolytic solution for secondary batteries of the present invention, if the capacitance (capacity when the battery is discharged from a fully charged state to a discharged state) of the battery elements contained in the battery case of one secondary battery is 3 If the ampere-hour (Ah) or more is greater, the effect of improving the low-temperature discharge characteristics becomes greater, which is preferable.
因此,正极板优选设计成在满充电时放电容量为3安培小时(Ah)以上且低于20Ah,更优选4Ah以上且低于10Ah。低于3Ah时,在取出大电流时,由电极反应阻抗引起的电压降低变大,功率效率有时变差。20Ah以上时,虽然电极反应阻抗变小,功率效率变好,但脉冲充放电时由电池内部发热引起的温度分布大,反复充放电的耐久性差,另外,在过充电或内部短路等异常时的剧烈发热时,其放热效率也变差,产生内压上升而使放气阀工作的现象(阀工作)、电池内容物剧烈地喷出到外面的现象(破裂)的几率有时升高。Therefore, the positive plate is preferably designed to have a discharge capacity of not less than 3 ampere hours (Ah) and less than 20 Ah, more preferably not less than 4 Ah and less than 10 Ah, when fully charged. If it is less than 3Ah, when a large current is taken out, the voltage drop due to the electrode reaction impedance becomes large, and the power efficiency may deteriorate. When it is above 20Ah, although the electrode reaction impedance becomes smaller and the power efficiency becomes better, the temperature distribution caused by the internal heating of the battery during pulse charging and discharging is large, and the durability of repeated charging and discharging is poor. When intense heat is generated, the heat release efficiency also deteriorates, and the internal pressure rises to cause the purge valve to operate (valve operation), and the possibility of the battery contents to be ejected violently to the outside (rupture) may increase.
[[正极板的厚度]][[Thickness of positive plate]]
正极板的厚度没有特别限定,但从高容量以及高输出的观点来看,减去芯材的金属箔厚度之后的复层材料层在集电体的一面上的厚度的下限优选为10μm以上,更优选为20μm,其上限优选为200μm以下,更优选为100μm以下。The thickness of the positive electrode plate is not particularly limited, but from the viewpoint of high capacity and high output, the lower limit of the thickness of the cladding material layer after subtracting the thickness of the metal foil of the core material on one side of the current collector is preferably 10 μm or more, More preferably, it is 20 μm, and the upper limit thereof is preferably 200 μm or less, more preferably 100 μm or less.
<正极[1]><positive pole[1]>
下面,对本发明的锂二次电池中使用的正极[1]“包含含有锰的正极活性物质的正极”进行说明。Next, the positive electrode [1] "positive electrode containing a positive electrode active material containing manganese" used in the lithium secondary battery of the present invention will be described.
[正极[1]的正极活性物质][Positive electrode active material of positive electrode [1]]
下面,对正极[1]中使用的正极活性物质进行说明。Next, the positive electrode active material used in the positive electrode [1] will be described.
[[组成]][[composition]]
作为正极活性物质,是含有能够以电化学方式吸留和放出锂离子的过渡金属的物质,作为过渡金属的至少一部分,在本发明中,使用含有锰的正极活性物质。优选进一步含有锂,更优选含有锂和锰的复合氧化物。The positive electrode active material contains a transition metal capable of electrochemically occluding and releasing lithium ions. In the present invention, a positive electrode active material containing manganese is used as at least a part of the transition metal. It is preferable to further contain lithium, and it is more preferable to contain a composite oxide of lithium and manganese.
上述含有锰的正极活性物质没有特别限定,但优选具有下述组成式(5)表示的组成的正极活性物质。The manganese-containing positive electrode active material is not particularly limited, but is preferably a positive electrode active material having a composition represented by the following composition formula (5).
LixMn(1-y)M1 yO2 组成式(5)Li x Mn (1-y) M 1 y O 2 composition formula (5)
[组成式(5)中,M1表示选自Ni、Co、Al、Fe、Ti、Mg、Cr、Ga、Cu、Zn和Nb中的至少一种元素,x表示满足0<x≤1.2的数,y表示满足0.05≤y≤0.8的数]。[In the composition formula (5), M1 represents at least one element selected from Ni, Co, Al, Fe, Ti, Mg, Cr, Ga, Cu, Zn and Nb, and x represents an element satisfying 0<x≤1.2 number, y represents the number satisfying 0.05≤y≤0.8].
在组成式(5)中,M1特别优选Ni、Co、Fe,另外,x特别优选0.2≤x≤1.15,y特别优选0.1≤y≤0.7。作为具有组成式(5)表示的组成的物质的具体例子,例如可列举LiMn0.5Ni0.5O2。In the composition formula (5), M 1 is particularly preferably Ni, Co, and Fe, and x is particularly preferably 0.2≤x≤1.15, and y is particularly preferably 0.1≤y≤0.7. As a specific example of the substance having the composition represented by composition formula (5), LiMn 0.5 Ni 0.5 O 2 is mentioned, for example.
另外,特别优选M1是Ni,且具有下述组成式(6)表示的组成的正极活性物质。In addition, a positive electrode active material in which M 1 is Ni and has a composition represented by the following composition formula (6) is particularly preferable.
LixMn(1-y-z)Ni1 yM2 zO2 组成式(6)Li x Mn (1-yz) Ni 1 y M 2 z O 2 composition formula (6)
[组成式(6)中,M2表示选自Co、Al、Fe、Ti、Mg、Cr、Ga、Cu、Zn和Nb中的至少一种元素,x表示满足0<x≤1.2的数,y表示满足0.05≤y≤0.8的数,z表示满足0.01≤z≤0.5的数]。[In the composition formula (6), M represents at least one element selected from Co, Al, Fe, Ti, Mg, Cr, Ga, Cu, Zn and Nb, and x represents a number satisfying 0<x≤1.2, y represents the number satisfying 0.05≤y≤0.8, and z represents the number satisfying 0.01≤z≤0.5].
在组成式(6)中,M2特别优选Co、Al、Fe、Mg,x特别优选0.2≤x≤1.15,y特别优选0.1≤y≤0.7,z特别优选0.1≤z≤0.7,y+z特别优选0.2≤y+z≤0.7。作为具有组成式(6)表示的组成的物质的具体例子,例如可列举LiMn0.33Ni0.33Co0.33O2等。In the composition formula (6), M2 is particularly preferably Co, Al, Fe, Mg, x is particularly preferably 0.2≤x≤1.15, y is particularly preferably 0.1≤y≤0.7, z is particularly preferably 0.1≤z≤0.7, y+z Particular preference is given to 0.2≦y+z≦0.7. Specific examples of the substance having a composition represented by the composition formula (6) include, for example, LiMn 0.33 Ni 0.33 Co 0.33 O 2 and the like.
作为含有锰的正极活性物质,优选具有下述组成式(7)表示的组成的正极活性物质。As the positive electrode active material containing manganese, a positive electrode active material having a composition represented by the following composition formula (7) is preferable.
LixMn(2-y)M3 yO4 组成式(7)Li x Mn (2-y) M 3 y O 4 composition formula (7)
[组成式(7)中,M3表示选自Ni、Co、Al、Fe、Ti、Mg、Cr、Ga、Cu、Zn和Nb中的至少一种元素,x表示满足0<x≤1.2的数,y表示满足0.05≤y≤0.8的数]。[In the composition formula (7), M represents at least one element selected from Ni, Co, Al, Fe, Ti, Mg, Cr, Ga, Cu, Zn and Nb, and x represents an element satisfying 0<x≤1.2 number, y represents the number satisfying 0.05≤y≤0.8].
在组成式(7)中,M3特别优选Ni、Co、Al、Mg,另外,x特别优选0.05≤x≤1.15,y特别优选0.1≤y≤0.7。作为具有组成式(7)表示的组成的物质的具体例子,例如可列举LiMn1.8Al0.2O4、LiMn1.5Ni0.5O4等。In the composition formula (7), M 3 is particularly preferably Ni, Co, Al, and Mg, and x is particularly preferably 0.05≤x≤1.15, and y is particularly preferably 0.1≤y≤0.7. Specific examples of the substance having the composition represented by the composition formula (7) include, for example, LiMn 1.8 Al 0.2 O 4 , LiMn 1.5 Ni 0.5 O 4 , and the like.
[[表面包覆]][[Cladding]]
另外,优选使用在该正极活性物质的表面附着有与上述含有锰的正极活性物质组成不同的物质的正极活性物质。表面附着物质的种类、附着方法、附着量等与上述相同。In addition, it is preferable to use a positive electrode active material having a composition different from that of the above-mentioned manganese-containing positive electrode active material adhered to the surface of the positive electrode active material. The type, method of adhesion, amount of adhesion, etc. of the surface adhesion substance are the same as above.
[[形状]][[shape]]
该正极活性物质的粒子形状与上述相同。The particle shape of the positive electrode active material is the same as above.
[[振实密度]][[Tap Density]]
该正极活性物质的振实密度与上述相同。The tap density of this positive electrode active material is the same as above.
[[中值粒径d50]][[Median particle size d 50 ]]
该正极活性物质的粒子的中值粒径d50(一次粒子凝聚而形成二次粒子时,为二次粒子直径)与上述相同。The median diameter d 50 (the diameter of the secondary particle when the primary particles are aggregated to form secondary particles) of the particles of the positive electrode active material is the same as above.
[[平均一次粒径]][[average primary particle size]]
一次粒子凝聚而形成二次粒子时,正极活性物质的平均一次粒径与上述相同。When primary particles are aggregated to form secondary particles, the average primary particle diameter of the positive electrode active material is the same as described above.
[[BET比表面积]][[BET specific surface area]]
正极活性物质的BET比表面积与上述相同。The BET specific surface area of the positive electrode active material is the same as above.
[[制造法]][[Manufacturing method]]
作为该正极活性物质的制造法,使用作为无机化合物的制造法的通常的方法。特别是在制作球状或椭圆球状的活性物质时,可考虑各种方法,例如可列举如下方法:将硝酸锰、硫酸锰等锰原料物质和根据需要使用的其他元素的原料物质溶解或粉碎分散在水等溶剂中,边搅拌边调节pH,制作并获得球状的前体,根据需要将其干燥后,加入LiOH、Li2CO3、LiNO3等Li源,在高温下烧结而得到活性物质的方法;将硝酸锰、硫酸锰、氧化锰、碱式氢氧化锰等锰原料物质和根据需要使用的其他元素的原料物质溶解或粉碎分散在水等溶剂中,再通过喷雾干燥器等将其干燥成型,制成球状或椭圆球状的前体,然后在其中加入LiOH、Li2CO3、LiNO3等Li源,在高温下烧结而得到活性物质的方法;以及将硝酸锰、硫酸锰、氧化锰、碱式氢氧化锰等锰原料物质和LiOH、Li2CO3、LiNO3等Li源以及根据需要使用的其他元素的原料物质溶解或粉碎分散在水等溶剂中,再通过喷雾干燥器等将其干燥成型,制成球状或椭圆球状的前体,然后在高温下将其烧结而得到活性物质的方法。As a method for producing the positive electrode active material, a common method for producing an inorganic compound is used. In particular, when producing a spherical or ellipsoidal active material, various methods can be considered. For example, the following method can be cited: dissolving or pulverizing raw materials of manganese such as manganese nitrate and manganese sulfate and other elements used as needed in In a solvent such as water, adjust the pH while stirring, prepare and obtain a spherical precursor, dry it if necessary, add a Li source such as LiOH, Li 2 CO 3 , LiNO 3 , and sinter at a high temperature to obtain an active material. Dissolve or pulverize manganese raw materials such as manganese nitrate, manganese sulfate, manganese oxide, basic manganese hydroxide, and other elements used as needed in solvents such as water, and then dry them into shape with a spray dryer, etc. , making a spherical or ellipsoidal precursor, then adding Li sources such as LiOH, Li 2 CO 3 , LiNO 3 to it, and sintering at a high temperature to obtain an active material; and manganese nitrate, manganese sulfate, manganese oxide, Manganese raw materials such as basic manganese hydroxide, Li sources such as LiOH, Li 2 CO 3 , and LiNO 3 , and raw materials of other elements used as needed are dissolved or pulverized in a solvent such as water, and then dried by a spray dryer or the like. It is a method of drying and forming a spherical or ellipsoidal precursor, and then sintering it at a high temperature to obtain an active material.
本发明中的正极活性物质可以单独使用该含有锰的正极活性物质,也可以以任意的组合和比例组合使用该含有锰的正极活性物质和一种或多种与该含有锰的正极活性物质组成不同或粉末物性不同的正极活性物质。作为此时的优选的组合,可列举含有锰的正极活性物质和LiNiO2或其部分Ni被其他过渡金属替代而得到的物质的组合;或者含有锰的正极活性物质和LiCoO2或其部分Co被其他过渡金属替代而得到的物质的组合。另外,作为特别优选的组合,还可列举组成式(5)~(7)表示的正极活性物质和LiNiO2或其部分Ni被其他过渡金属替代而得到的物质的组合;或者组成式(5)~(7)表示的正极活性物质和LiCoO2或其部分Co被其他过渡金属替代而得到的物质的组合。含有锰的正极活性物质,特别是组成式(5)~(7)表示的正极活性物质优选为全部正极活性物质的30质量%以上,更优选为50质量%以上。组成式(5)~(7)表示的正极活性物质的使用比例如果变少,则有时不会降低正极的成本。The positive electrode active material in the present invention can use this positive electrode active material containing manganese alone, also can use this positive electrode active material containing manganese and one or more with this positive electrode active material composition containing manganese in any combination and ratio Positive electrode active materials with different or different powder properties. As a preferred combination at this time, a combination of a positive electrode active material containing manganese and LiNiO 2 or part of Ni replaced by other transition metals; or a positive electrode active material containing manganese and LiCoO 2 or part of Co replaced by A combination of substances obtained by substitution of other transition metals. In addition, as a particularly preferred combination, the combination of the positive electrode active material represented by the composition formula (5) to (7) and LiNiO 2 or a part of Ni replaced by other transition metals; or the composition formula (5) A combination of the positive electrode active material represented by ~ (7) and LiCoO 2 or a part of Co replaced by other transition metals. The positive electrode active material containing manganese, especially the positive electrode active material represented by the composition formulas (5) to (7) is preferably 30% by mass or more, more preferably 50% by mass or more of the total positive electrode active material. If the ratio of the positive electrode active material represented by the composition formulas (5) to (7) is reduced, the cost of the positive electrode may not be reduced.
[正极[1]的正极的结构][Structure of the positive electrode of the positive electrode [1]]
下面,对正极[1]中使用的正极的结构进行说明。Next, the structure of the positive electrode used in the positive electrode [1] will be described.
正极[1]中的电极结构和制作方法、正极活性物质层的压实、导电材料、正极活性物质层的制造时使用的粘合材料、用于形成浆料的液体介质、集电体、电极面积、放电容量、正极板的厚度等与上述相同。Electrode structure and manufacturing method in positive electrode [1], compaction of positive electrode active material layer, conductive material, binder used in the production of positive electrode active material layer, liquid medium for forming slurry, current collector, electrode The area, discharge capacity, thickness of the positive electrode plate, etc. are the same as above.
<正极[2]><Positive pole [2]>
下面,对本发明的锂二次电池中使用的正极[2]“含有组成式(4)表示的具有特定组成的正极活性物质的正极”进行说明。Next, the positive electrode [2] "a positive electrode containing a positive electrode active material having a specific composition represented by the composition formula (4)" used in the lithium secondary battery of the present invention will be described.
[正极[2]的正极活性物质][Positive electrode active material of positive electrode [2]]
下面,对正极[2]中使用的正极活性物质进行说明。Next, the positive electrode active material used in the positive electrode [2] will be described.
[[组成]][[composition]]
作为正极活性物质,使用具有下述组成式(4)表示的组成的,含有能够以电化学方式吸留和放出锂离子的过渡金属的物质(下面,简称为“该正极活性物质”)。As the positive electrode active material, a material having a composition represented by the following composition formula (4) and containing a transition metal capable of electrochemically storing and releasing lithium ions (hereinafter, simply referred to as "the positive electrode active material") is used.
LixNi(1-y-z)CoyMzO2 组成式(4)Li x Ni (1-yz) Co y M z O 2 composition formula (4)
[组成式(4)中,M表示选自Mn、Al、Fe、Ti、Mg、Cr、Ga、Cu、Zn和Nb中的至少一种元素,x表示满足0<x≤1.2的数,y表示满足0.05≤y≤0.5的数,z表示满足0.01≤z≤0.5的数]。[In the composition formula (4), M represents at least one element selected from Mn, Al, Fe, Ti, Mg, Cr, Ga, Cu, Zn and Nb, x represents a number satisfying 0<x≤1.2, and y represents the number satisfying 0.05≤y≤0.5, and z represents the number satisfying 0.01≤z≤0.5].
在组成式(4)中,M优选为Mn、Al或Mg,另外,x优选0.2≤x≤1.15。另外,y优选0.08≤y≤0.4,特别优选0.1≤y≤0.3。另外,z优选0.02≤z≤0.4,特别优选0.03≤z≤0.3。In the composition formula (4), M is preferably Mn, Al or Mg, and x is preferably 0.2≤x≤1.15. In addition, y is preferably 0.08≤y≤0.4, particularly preferably 0.1≤y≤0.3. In addition, z is preferably 0.02≤z≤0.4, particularly preferably 0.03≤z≤0.3.
[[表面包覆]][[Cladding]]
另外,优选使用在该正极活性物质的表面附着有与该正极活性物质组成不同的物质的正极活性物质。表面附着物质的种类、附着方法、附着量等与上述相同。In addition, it is preferable to use a positive electrode active material having a composition different from that of the positive electrode active material adhered to the surface of the positive electrode active material. The type, method of adhesion, amount of adhesion, etc. of the surface adhesion substance are the same as above.
[[形状]][[shape]]
该正极活性物质的粒子形状与上述相同。The particle shape of the positive electrode active material is the same as above.
[[振实密度]][[Tap Density]]
该正极活性物质的振实密度与上述相同。The tap density of this positive electrode active material is the same as above.
[[中值粒径d50]][[Median particle size d 50 ]]
该正极活性物质的粒子的中值粒径d50(一次粒子凝聚而形成二次粒子时,为二次粒子直径)与上述相同。The median diameter d 50 (the diameter of the secondary particle when the primary particles are aggregated to form secondary particles) of the particles of the positive electrode active material is the same as above.
[[平均一次粒径]][[average primary particle size]]
一次粒子凝聚而形成二次粒子时,正极活性物质的平均一次粒径与上述相同。When primary particles are aggregated to form secondary particles, the average primary particle diameter of the positive electrode active material is the same as described above.
[[BET比表面积]][[BET specific surface area]]
正极活性物质的BET比表面积与上述相同。The BET specific surface area of the positive electrode active material is the same as above.
[[制造法]][[Manufacturing method]]
作为该正极活性物质的制造法,可使用作为无机化合物的制造法的通常的方法。特别是在制作球状或椭圆球状的活性物质时,可考虑各种方法,例如可列举如下方法:将硝酸镍、硫酸镍等镍原料物质和硝酸钴、硫酸钴等钴原料物质以及组成式(4)中的M的原料物质溶解或粉碎分散在水等溶剂中,边搅拌边调节pH,制作并获得球状的前体,根据需要将其干燥后,加入LiOH、Li2CO3、LiNO3等Li源,在高温下烧结而得到活性物质的方法;将硝酸镍、硫酸镍、氧化镍、氢氧化镍、碱式氢氧化镍等镍原料物质和硝酸钴、硫酸钴、氧化钴、氢氧化钴、碱式氢氧化钴等钴原料物质以及组成式(4)中的M的原料物质溶解或粉碎分散在水等溶剂中,再通过喷雾干燥器等将其干燥成型,制成球状或椭圆球状的前体,然后在其中加入LiOH、Li2CO3、LiNO3等Li源,在高温下烧结而得到活性物质的方法;以及将硝酸镍、硫酸镍、氧化镍、氢氧化镍、碱式氢氧化镍等镍原料物质和硝酸钴、硫酸钴、氧化钴、氢氧化钴、碱式氢氧化钴等钴原料物质以及组成式(4)中的M的原料物质与LiOH、Li2CO3、LiNO3等Li源溶解或粉碎分散在水等溶剂中,再通过喷雾干燥器等将其干燥成型,制成球状或椭圆球状的前体,然后在高温下将其烧结而得到活性物质的方法。As a method for producing the positive electrode active material, a common method for producing an inorganic compound can be used. In particular, when making a spherical or ellipsoidal active material, various methods can be considered, for example, the following method can be enumerated: nickel raw materials such as nickel nitrate and nickel sulfate, cobalt raw materials such as cobalt nitrate and cobalt sulfate, and the composition formula (4 ) in the M raw material is dissolved or pulverized and dispersed in water or other solvents, and the pH is adjusted while stirring to prepare and obtain a spherical precursor. After drying it as necessary, add LiOH, Li 2 CO 3 , LiNO 3 and other Li source, a method of sintering at high temperature to obtain an active material; nickel raw materials such as nickel nitrate, nickel sulfate, nickel oxide, nickel hydroxide, nickel hydroxide, and cobalt nitrate, cobalt sulfate, cobalt oxide, cobalt hydroxide, The cobalt raw material such as cobalt basic hydroxide and the raw material of M in the composition formula (4) are dissolved or pulverized and dispersed in a solvent such as water, and then dried and shaped by a spray dryer to form a spherical or ellipsoidal precursor. body, and then adding Li sources such as LiOH, Li 2 CO 3 , LiNO 3 , and sintering at high temperature to obtain active materials; and nickel nitrate, nickel sulfate, nickel oxide, nickel hydroxide, nickel hydroxide Nickel raw materials such as cobalt nitrate, cobalt sulfate, cobalt oxide, cobalt hydroxide, cobalt oxyhydroxide and other cobalt raw materials, and the raw materials of M in the composition formula (4) and LiOH, Li 2 CO 3 , LiNO 3 etc. Li source is dissolved or pulverized and dispersed in a solvent such as water, and then dried and shaped by a spray dryer to form a spherical or ellipsoidal precursor, which is then sintered at a high temperature to obtain an active material.
为了制造本发明中的正极,可以单独使用该正极活性物质(组成式(4)表示的正极活性物质和/或由上述表面附着物质包覆的组成式(4)表示的正极活性物质),也可以以任意的组合和比例组合使用该正极活性物质和一种以上与该正极活性物质组成不同的物质。作为此时的优选的组合,可列举该正极活性物质和LiMn2O4或其部分Mn被其他过渡金属替代而得到的物质的组合;或者该正极活性物质和LiCoO2或其部分Co被其他过渡金属替代而得到的物质的组合。In order to manufacture the positive electrode in the present invention, the positive electrode active material (the positive electrode active material represented by the composition formula (4) and/or the positive electrode active material represented by the composition formula (4) coated by the above-mentioned surface attachment substance) can be used alone, or The cathode active material and one or more substances different in composition from the cathode active material may be used in combination in any combination and ratio. As a preferred combination at this time, the combination of the positive active material and LiMn 2 O 4 or a part of Mn thereof is replaced by other transition metals; or the positive active material and LiCoO 2 or part of Co is replaced by other transition metals. Combination of substances obtained by substitution of metals.
这里,优选该正极活性物质为全部正极活性物质的30质量%以上,更优选为50质量%以上。该正极活性物质的使用比例如果变少,则有时电池容量变小。另外,将“该正极活性物质”和“该正极活性物质以外的正极活性物质”总称为“正极活性物质”。Here, the positive electrode active material is preferably 30% by mass or more, more preferably 50% by mass or more of the total positive electrode active material. When the usage ratio of the positive electrode active material is reduced, the battery capacity may be reduced. In addition, "the positive electrode active material" and "the positive electrode active material other than the positive electrode active material" are collectively referred to as "the positive electrode active material".
[正极[2]的正极的结构][Structure of the positive electrode of the positive electrode [2]]
下面,对正极[2]中使用的正极的结构进行叙述。Next, the structure of the positive electrode used in the positive electrode [2] will be described.
正极[2]中的电极结构和制作方法、正极活性物质层的压实、导电材料、正极活性物质层的制造时使用的粘合材料、用于形成浆料的液体介质、集电体、电极面积、放电容量、正极板的厚度等与上述相同。Electrode structure and manufacturing method in positive electrode [2], compaction of positive electrode active material layer, conductive material, binder used in the production of positive electrode active material layer, liquid medium for forming slurry, current collector, electrode The area, discharge capacity, thickness of the positive electrode plate, etc. are the same as above.
<正极[3]><Positive pole [3]>
下面,对本发明的锂二次电池中使用的正极[3]“包含选自下述(a)~(d)中的任一种正极活性物质的正极”进行说明。Next, the positive electrode [3] "a positive electrode containing any one positive electrode active material selected from the following (a) to (d)" used in the lithium secondary battery of the present invention will be described.
(a)BET比表面积为0.4m2/g~2m2/g的正极活性物质(a) A positive electrode active material having a BET specific surface area of 0.4m 2 /g to 2m 2 /g
(b)平均一次粒径为0.1μm~2μm的正极活性物质(b) Positive active material having an average primary particle size of 0.1 μm to 2 μm
(c)中值粒径d50为1μm~20μm的正极活性物质(c) A positive electrode active material having a median diameter d50 of 1 μm to 20 μm
(d)振实密度为1.3g/cm3~2.7g/cm3的正极活性物质。(d) A positive electrode active material having a tap density of 1.3 g/cm 3 to 2.7 g/cm 3 .
[正极[3]的正极活性物质][Positive electrode active material of positive electrode [3]]
下面,对正极[3]中使用的正极活性物质进行叙述。Next, the positive electrode active material used in the positive electrode [3] will be described.
[[组成]][[composition]]
作为正极活性物质,只要是能够以电化学方式吸留和放出锂离子的物质即可,没有特别限制。优选含有锂和至少一种过渡金属的物质,例如可列举锂-过渡金属复合氧化物、含有锂的过渡金属磷酸化合物。具体地,可使用具有与上述同样组成的物质。The positive electrode active material is not particularly limited as long as it can electrochemically occlude and release lithium ions. A substance containing lithium and at least one transition metal is preferable, and examples thereof include lithium-transition metal composite oxides and lithium-containing transition metal phosphate compounds. Specifically, those having the same composition as above can be used.
[[表面包覆]][[Cladding]]
优选该正极活性物质的表面附着有与芯的正极活性物质组成不同的物质的正极活性物质。表面附着物质的种类、附着方法、附着量等与上述相同。Preferably, a positive electrode active material having a composition different from that of the positive electrode active material of the core adheres to the surface of the positive electrode active material. The type, method of adhesion, amount of adhesion, etc. of the surface adhesion substance are the same as above.
[[形状]][[shape]]
该正极活性物质的粒子形状与上述相同。The particle shape of the positive electrode active material is the same as above.
[[BET比表面积]][[BET specific surface area]]
该正极活性物质的BET比表面积优选为0.4m2/g以上,更优选为0.5m2/g以上,进一步优选为0.6m2/g以上,其上限优选为2m2/g以下,更优选为1.8m2/g以下,进一步优选为1.5m2/g以下。如果BET比表面积比该范围小,则电池性能容易降低,而如果BET比表面积比该范围大,则振实密度难以提高,在形成正极活性物质时的涂布性上有时容易产生问题。The BET specific surface area of the positive electrode active material is preferably 0.4 m 2 /g or more, more preferably 0.5 m 2 /g or more, even more preferably 0.6 m 2 /g or more, the upper limit is preferably 2 m 2 /g or less, more preferably 1.8 m 2 /g or less, more preferably 1.5 m 2 /g or less. If the BET specific surface area is smaller than this range, the battery performance is likely to decrease, and if the BET specific surface area is larger than this range, it is difficult to increase the tap density, and problems may easily occur in coating properties when forming a positive electrode active material.
BET比表面积定义为如下的值:使用表面积计(例如,大仓理研制造的全自动表面积测定装置),在氮流通下在150℃对试料进行30分钟预干燥,然后使用正确调整为氮相对于大气压的相对压力值为0.3的氮氦混合气体,通过采用气体流动法进行的氮吸附BET 1点法而测定的值。The BET specific surface area is defined as the following value: Using a surface area meter (for example, a fully automatic surface area measuring device manufactured by Okura Riken), the sample is pre-dried at 150°C for 30 minutes under nitrogen flow, and then adjusted to the nitrogen relative The value measured by the nitrogen adsorption BET 1-point method using the gas flow method for a nitrogen-helium mixed gas with a relative pressure value of 0.3 at atmospheric pressure.
[[平均一次粒径]][[average primary particle size]]
一次粒子凝聚而形成二次粒子时,该正极活性物质的平均一次粒径优选为0.1μm以上,更优选为0.2μm以上,进一步优选为0.3μm以上,最优选为0.4μm以上,其上限优选为2μm以下,更优选为1.6μm以下,进一步优选为1.3μm以下,最优选为1μm以下。如果平均一次粒径超过上述上限,则难以形成球状的二次粒子,对粉末填充性带来不良影响,或者比表面积大幅降低,因此,输出特性等电池性能降低的可能性有时变高。相反,如果平均一次粒径低于上述下限,则通常由于结晶不完全,因此有时产生充放电的可逆性差等问题。When primary particles aggregate to form secondary particles, the average primary particle size of the positive electrode active material is preferably 0.1 μm or more, more preferably 0.2 μm or more, further preferably 0.3 μm or more, most preferably 0.4 μm or more, and the upper limit is preferably 2 μm or less, more preferably 1.6 μm or less, still more preferably 1.3 μm or less, most preferably 1 μm or less. When the average primary particle size exceeds the above upper limit, it is difficult to form spherical secondary particles, which adversely affects the powder fillability, or the specific surface area is greatly reduced, and therefore battery performance such as output characteristics may decrease. On the contrary, if the average primary particle diameter is less than the above-mentioned lower limit, problems such as poor charge-discharge reversibility may occur due to incomplete crystallization.
一次粒径可通过使用扫描电子显微镜(SEM)的观察来测定。具体地,通过如下方法求出:在倍率10000倍的照片中,对任意的50个一次粒子求出水平方向的直线上的由一次粒子的左右边界线产生的切片的最长值,并取其平均值。The primary particle size can be measured by observation using a scanning electron microscope (SEM). Specifically, it is obtained by the following method: in a photograph with a magnification of 10,000 times, for any 50 primary particles, the longest value of the slice generated by the left and right boundary lines of the primary particles on a straight line in the horizontal direction is obtained, and the maximum value is obtained. average value.
[[中值粒径d50]][[Median particle size d 50 ]]
该正极活性物质的粒子的中值粒径d50(一次粒子凝聚而形成二次粒子时,为二次粒子直径)优选为1μm以上,更优选为1.2μm以上,进一步优选为1.5μm以上,最优选为2μm以上,其上限优选为20μm以下,更优选为18μm以下,进一步优选为16μm以下,最优选为15μm以下。如果中值粒径d50低于上述下限,则有时不能得到高振实密度的产品,而如果超过上限,则由于粒子内的锂的扩散需要时间,因此有时会产生电池性能降低,或者在制成电池的正极、也就是将活性物质和导电剂或粘合剂等在溶剂中浆料化再涂布成薄膜状时,产生条纹等问题。本发明的正极活性物质为混合了2种以上具有不同的中值粒径d50的正极活性物质而得到的正极活性物质时,可以进一步提高制作正极时的填充性,是优选的。The median diameter d50 of the particles of the positive electrode active material (when the primary particles are aggregated to form secondary particles, the diameter of the secondary particles) is preferably 1 μm or more, more preferably 1.2 μm or more, even more preferably 1.5 μm or more, and most preferably 1 μm or more. It is preferably 2 μm or more, and the upper limit thereof is preferably 20 μm or less, more preferably 18 μm or less, still more preferably 16 μm or less, and most preferably 15 μm or less. If the median particle diameter d50 is lower than the above-mentioned lower limit, a product with a high tap density may not be obtained, and if it exceeds the upper limit, since the diffusion of lithium in the particles takes time, the performance of the battery may sometimes decrease, or the resulting battery may be damaged. Streaks and other problems occur when the active material and conductive agent or binder are slurried in a solvent and then coated into a film. When the positive electrode active material of the present invention is a positive electrode active material obtained by mixing two or more positive electrode active materials having different median particle diameters d50 , it can further improve the filling property when making a positive electrode, which is preferable.
另外,本发明中的中值粒径d50可以利用公知的激光衍射/散射式粒度分布测定装置来测定。使用HORIBA公司制造的LA-920作为粒度分布计时,作为测定时使用的分散介质,使用0.1质量%六偏磷酸钠水溶液,进行5分钟超声波分散后,设定测定折射率1.24来进行测定。In addition, the median diameter d50 in the present invention can be measured using a known laser diffraction/scattering type particle size distribution analyzer. LA-920 manufactured by HORIBA was used as a particle size distribution meter. As a dispersion medium used in the measurement, a 0.1% by mass sodium hexametaphosphate aqueous solution was used, ultrasonically dispersed for 5 minutes, and then measured by setting the measurement refractive index to 1.24.
[[振实密度]][[Tap Density]]
该正极活性物质的振实密度优选为1.3g/cm3以上,更优选为1.5g/cm3以上,进一步优选为1.6g/cm3以上,最优选为1.7g/cm3以上。正极活性物质的振实密度如果低于上述下限,则在形成正极活性物质层时,需要的分散介质量增加,同时导电材料或粘合剂的必要量增加,正极活性物质在正极活性物质层中的填充率受到制约,电池容量有时受到制约。通过使用振实密度高的复合氧化物粉末,可以形成高密度的正极活性物质层。一般来说,振实密度越大越优选,没有特别的上限,但如果振实密度太大,则正极活性物质层内的以电解液为介质的锂离子的扩散成为决定速度的因素,负荷特性有时容易降低,因此,振实密度的上限优选为2.7g/cm3以下,更优选为2.5g/cm3以下。The tap density of the positive electrode active material is preferably 1.3 g/cm 3 or higher, more preferably 1.5 g/cm 3 or higher, still more preferably 1.6 g/cm 3 or higher, most preferably 1.7 g/cm 3 or higher. If the tap density of the positive electrode active material is lower than the above-mentioned lower limit, then when forming the positive electrode active material layer, the amount of dispersion medium required increases, and the necessary amount of conductive material or binder increases simultaneously, and the positive electrode active material is in the positive electrode active material layer. The filling rate of the battery is restricted, and the battery capacity is sometimes restricted. By using a composite oxide powder with a high tap density, a high-density positive electrode active material layer can be formed. In general, the larger the tap density, the better, and there is no particular upper limit, but if the tap density is too large, the diffusion of lithium ions in the positive electrode active material layer with the electrolyte as a medium becomes a factor determining the speed, and the load characteristics sometimes Therefore, the upper limit of the tap density is preferably 2.7 g/cm 3 or less, more preferably 2.5 g/cm 3 or less.
在本发明中,振实密度如下定义:使试料通过孔径300μm的筛子落到20cm3的振实容器中,充满容器容积之后,使用粉末密度测定器(例如,seishin企业社制造的Tap densor),进行1000次冲程长度10mm的振动,将此时的容积密度作为振实密度。In the present invention, the tap density is defined as follows: the sample is dropped into a 20 cm 3 tapped container through a sieve with an aperture of 300 μm, and after the container volume is filled, a powder density measuring device (for example, Tap densor manufactured by Seishin Co., Ltd.) is used. , vibration with a stroke length of 10 mm was performed 1000 times, and the bulk density at this time was taken as the tap density.
从长寿命、高输出方面来看,本发明的锂二次电池中的正极活性物质特别优选同时满足上述的BET比表面积、平均一次粒径、中值粒径d50和振实密度中的2种以上物性。From the perspective of long life and high output, the positive electrode active material in the lithium secondary battery of the present invention preferably satisfies 2 of the above-mentioned BET specific surface area, average primary particle size, median particle size d50 and tap density at the same time. more than one physical property.
[[制造法]][[Manufacturing method]]
该正极活性物质的制造法与上述相同。The manufacturing method of this positive electrode active material is the same as above.
本发明的锂二次电池中的正极活性物质可以单独使用一种,也可以以任意的组合和比率组合2种以上组成不同或粉末物性不同的正极活性物质使用。另外,从提高寿命这点来看,本发明的正极优选含有具有上述任意一种物性的正极活性物质(该正极活性物质)和与该正极活性物质组成不同的正极活性物质的正极。The positive electrode active material in the lithium secondary battery of the present invention may be used alone, or may be used in combination of two or more positive electrode active materials having different compositions or different powder physical properties in any combination and ratio. In addition, from the viewpoint of improving life, the positive electrode of the present invention preferably contains a positive electrode active material (the positive electrode active material) having any of the above physical properties and a positive electrode active material having a composition different from the positive electrode active material.
[正极[3]的正极的结构][Structure of the positive electrode of the positive electrode [3]]
下面,对正极[3]中使用的正极的结构进行叙述。Next, the structure of the positive electrode used in the positive electrode [3] will be described.
正极[3]中的电极结构和制作方法、正极活性物质层的压实、导电材料、正极活性物质层的制造时使用的粘合材料、用于形成浆料的液体介质、集电体、电极面积、放电容量、正极板的厚度等与上述相同。Electrode structure and manufacturing method in positive electrode [3], compaction of positive electrode active material layer, conductive material, binder used in the production of positive electrode active material layer, liquid medium for forming slurry, current collector, electrode The area, discharge capacity, thickness of the positive electrode plate, etc. are the same as above.
<正极[4]><Positive pole [4]>
下面,对本发明的锂二次电池中使用的正极[4]“满足选自下述(e)~(f)中的任一条件的正极”进行说明。Next, the positive electrode [4] "a positive electrode satisfying any one of the following conditions (e) to (f)" used in the lithium secondary battery of the present invention will be described.
(e)在集电体上形成含有正极活性物质、导电材料和粘合剂的正极活性物质层而制作的正极,其中,正极活性物质层中的导电材料的含量为6质量%~20质量%的范围(方式1)(e) A positive electrode made by forming a positive electrode active material layer containing a positive electrode active material, a conductive material and a binder on a current collector, wherein the content of the conductive material in the positive electrode active material layer is 6% by mass to 20% by mass range (mode 1)
(f)在集电体上形成含有正极活性物质和粘合剂的正极活性物质层而制作的正极,其中,正极活性物质层的密度为1.7g/cm3~3.5g/cm3的范围(方式2)(f) a positive electrode made by forming a positive electrode active material layer containing a positive electrode active material and a binder on a current collector, wherein the density of the positive electrode active material layer is in the range of 1.7 g/cm 3 to 3.5 g/cm 3 ( way 2)
(g)在集电体上形成含有正极活性物质和粘合剂的正极活性物质层而制作的正极,其中,正极活性物质层和集电体的厚度之比(注入非水电解液之前的一侧活性物质层的厚度)/(集电体的厚度)的值为1.6~20(方式3)。(g) a positive electrode made by forming a positive electrode active material layer containing a positive electrode active material and a binding agent on the current collector, wherein the ratio of the thickness of the positive electrode active material layer to the current collector (one period before injecting the nonaqueous electrolyte solution) The value of (thickness of side active material layer)/(thickness of current collector) is 1.6 to 20 (form 3).
[正极[4]的正极活性物质][Positive electrode active material of positive electrode [4]]
下面,对正极[4]中使用的正极活性物质进行叙述。Next, the positive electrode active material used in the positive electrode [4] will be described.
作为正极活性物质,只要是能够以电化学方式吸留和放出锂离子的物质即可,没有特别限制。优选含有锂和至少一种过渡金属的物质,例如可列举锂-过渡金属复合氧化物、含有锂的过渡金属磷酸化合物。具体地,可使用具有与上述同样组成的物质。The positive electrode active material is not particularly limited as long as it can electrochemically occlude and release lithium ions. A substance containing lithium and at least one transition metal is preferable, and examples thereof include lithium-transition metal composite oxides and lithium-containing transition metal phosphate compounds. Specifically, those having the same composition as above can be used.
该正极活性物质的表面包覆、形状、振实密度、中值粒径d50、平均一次粒径、BET比表面积、制造法等与上述相同。The surface coating, shape, tap density, median particle size d 50 , average primary particle size, BET specific surface area, production method, etc. of the positive electrode active material are the same as above.
[正极[4]的正极的结构][Structure of the positive electrode of the positive electrode [4]]
下面,对正极[4]中使用的正极的结构进行叙述。Next, the structure of the positive electrode used in the positive electrode [4] will be described.
[[电极结构和制作法]][[Electrode structure and fabrication method]]
本发明的锂二次电池中的正极[4]是在集电体上形成含有正极活性物质和粘合剂的正极活性物质层而制作的。使用正极活性物质的正极的制造可按照通常的方法进行。即,至少将正极活性物质、粘合剂、根据需要使用的导电材料(在方式1中为必须含有成分)和根据需要使用的增稠剂等进行干式混合而制成片状,再将该片状材料压粘在正极集电体上,或者将这些材料溶解或分散在液体介质中,制成浆料,再将该浆料涂布在正极集电体上并进行干燥,由此可以在集电体上形成正极活性物质层,从而得到正极。The positive electrode [4] in the lithium secondary battery of the present invention is produced by forming a positive electrode active material layer containing a positive electrode active material and a binder on a current collector. The production of the positive electrode using the positive electrode active material can be performed according to a usual method. That is, at least the positive electrode active material, the binder, the conductive material used as needed (in the mode 1 is an essential component), and the thickener used as needed are dry-mixed to form a sheet, and then the The sheet-like material is pressure-bonded on the positive electrode collector, or these materials are dissolved or dispersed in a liquid medium to make a slurry, and then the slurry is coated on the positive electrode collector and dried, so that the A positive electrode active material layer is formed on the current collector to obtain a positive electrode.
正极活性物质层中的正极活性物质的含量优选为80质量%以上,更优选为82质量%以上,特别优选为84质量%以上。其上限优选为95质量%以下,更优选为93质量%以下。如果正极活性物质层中的正极活性物质的含量低,则有时电容变得不充分。相反,如果正极活性物质的含量太高,则正极的强度有时不充分。The content of the positive electrode active material in the positive electrode active material layer is preferably 80% by mass or more, more preferably 82% by mass or more, particularly preferably 84% by mass or more. The upper limit thereof is preferably 95% by mass or less, more preferably 93% by mass or less. When the content of the positive electrode active material in the positive electrode active material layer is low, the capacitance may become insufficient. Conversely, if the content of the positive electrode active material is too high, the strength of the positive electrode may not be sufficient.
[[导电材料]][[Conductive Material]]
作为导电材料,可以任意使用公知的导电材料。作为具体的例子,可列举与上述相同的导电材料。As the conductive material, known conductive materials can be used arbitrarily. Specific examples include the same conductive materials as those described above.
本发明的锂二次电池的正极中使用的导电材料在正极活性物质层中的含量如下:在方式1中必须为6质量%以上,优选为7质量%以上,更优选为8质量%以上,进一步优选为9质量%以上;另外,在方式2和方式3中没有特别限定,但优选为6质量%以上,更优选为7质量%以上,进一步优选为8质量%以上。正极活性物质层中的导电材料的含量如果太少的话,导电性不充分,有时得不到高输出。The content of the conductive material used in the positive electrode of the lithium secondary battery of the present invention in the positive electrode active material layer is as follows: in mode 1, it must be 6% by mass or more, preferably 7% by mass or more, more preferably 8% by mass or more, It is more preferably 9% by mass or more; and it is not particularly limited in Embodiment 2 and Embodiment 3, but is preferably 6% by mass or more, more preferably 7% by mass or more, and still more preferably 8% by mass or more. If the content of the conductive material in the positive electrode active material layer is too small, the conductivity may be insufficient, and high output may not be obtained.
另外,正极活性物质层中的导电材料的含量的上限如下:在方式1中必须为20质量%以下,优选为18质量%以下,特别优选为15质量%以下;另外,在方式2和方式3中没有特别限定,但优选为20质量%以下,更优选为18质量%以下,进一步优选为15质量%以下。如果其含量太多,则电池容量降低,有时得不到高输出。In addition, the upper limit of the content of the conductive material in the positive electrode active material layer is as follows: in mode 1, it must be 20 mass % or less, preferably 18 mass % or less, particularly preferably 15 mass % or less; in addition, in mode 2 and mode 3 is not particularly limited, but is preferably 20% by mass or less, more preferably 18% by mass or less, even more preferably 15% by mass or less. If its content is too large, the battery capacity will decrease, and high output may not be obtained.
[[粘合剂]][[Binder]]
作为在正极活性物质层的制造中使用的粘合剂,没有特别限定,使用涂布法时,只要是可溶解或分散在电极制造时使用的液体介质中的材料即可,作为具体例子,可列举与上述相同的物质。The binder used in the manufacture of the positive electrode active material layer is not particularly limited. When using the coating method, any material can be dissolved or dispersed in the liquid medium used in the manufacture of the electrode. As a specific example, The same substances as above are listed.
粘合剂在正极活性物质层中的比例通常为0.1质量%以上,优选为1质量%以上,更优选为3质量%以上,其上限通常为80质量%以下,优选为60质量%以下,更优选为40质量%以下,进一步优选为10质量%以下。如果粘合剂的比例太低,则有时不能充分保持正极活性物质,正极的机械强度不足,循环特性等电池性能恶化。另一方面,如果粘合剂的比例太高,则有时会导致电池容量或导电性降低。The ratio of the binder in the positive electrode active material layer is usually 0.1% by mass or more, preferably 1% by mass or more, more preferably 3% by mass or more, and the upper limit is usually 80% by mass or less, preferably 60% by mass or less, more preferably Preferably it is 40 mass % or less, More preferably, it is 10 mass % or less. If the ratio of the binder is too low, the positive electrode active material may not be sufficiently retained, the mechanical strength of the positive electrode may be insufficient, and battery performance such as cycle characteristics may deteriorate. On the other hand, if the ratio of the binder is too high, it sometimes leads to a decrease in battery capacity or conductivity.
[[用于形成浆料的液体介质]][[The liquid medium used to form the slurry]]
作为用于形成浆料的液体介质,只要是能够溶解或分散正极活性物质、导电剂、粘合剂以及根据需要使用的增稠剂的溶剂,其种类就没有特别限制,可以使用水系溶剂和有机溶剂中的任一种。作为具体例子,可列举与上述同样的物质。增粘材料的种类和添加量也与上述相同。The liquid medium used to form the slurry is not particularly limited as long as it is a solvent capable of dissolving or dispersing the positive electrode active material, conductive agent, binder and, if necessary, a thickener. Aqueous solvents and organic solvents can be used. any of the solvents. Specific examples include the same ones as above. The kind and addition amount of the tackifier are also the same as above.
[[压实]][[Compaction]]
为了提高正极活性物质的填充密度,通过涂布、干燥而得到的正极活性物质层优选利用手动压力机、辊压机等进行压实。正极活性物质层的密度的下限在方式2中必须为1.7g/cm3以上,优选为2.0g/cm3以上,特别优选为2.2g/cm3以上;另外,在方式1和方式3中没有特别限定,但优选为1.7g/cm3以上,更优选为2.0g/cm3以上,进一步优选为2.2g/cm3以上。如果密度低于该范围,则活性物质间的导电性降低,电池阻抗增大,有时不能得到高输出。In order to increase the packing density of the positive electrode active material, the positive electrode active material layer obtained by coating and drying is preferably compacted using a manual press, a roll press, or the like. The lower limit of the density of the positive electrode active material layer must be 1.7 g/cm or more in mode 2, preferably 2.0 g/cm or more, particularly preferably 2.2 g/cm or more; in addition, in mode 1 and mode 3, there is no Especially limited, it is preferably 1.7 g/cm 3 or more, more preferably 2.0 g/cm 3 or more, and still more preferably 2.2 g/cm 3 or more. If the density is lower than this range, the conductivity between the active materials will decrease, the battery impedance will increase, and high output may not be obtained.
正极活性物质层的密度的上限在方式2中必须为3.5g/cm3以下,优选为3.0g/cm3以下,特别优选为2.8g/cm3以下;另外,在方式1和方式3中没有特别限定,但优选为3.5g/cm3以下,更优选为3.0g/cm3以下,进一步优选为2.8g/cm3以下。如果密度高于该范围,则非水电解液向集电体/活性物质界面附近的渗透性降低,特别是在高电流密度下的充放电特性降低,有时不能得到高输出。The upper limit of the density of the positive electrode active material layer must be 3.5g/cm 3 or less in mode 2, preferably 3.0 g/cm 3 or less, particularly preferably 2.8 g/cm 3 or less; Especially limited, it is preferably 3.5 g/cm 3 or less, more preferably 3.0 g/cm 3 or less, and still more preferably 2.8 g/cm 3 or less. If the density is higher than this range, the permeability of the non-aqueous electrolyte solution to the vicinity of the current collector/active material interface decreases, especially the charge and discharge characteristics at high current density decrease, and high output may not be obtained.
[[集电体]][[Collector]]
作为正极集电体的材质,没有特别限制,与上述相同。另外,集电体的形状和薄膜的厚度也与上述相同。The material of the positive electrode current collector is not particularly limited, and is the same as above. In addition, the shape of the current collector and the thickness of the film are also the same as above.
对于正极活性物质层和集电体的厚度之比,(注入非水电解液之前的一侧的活性物质层厚度)/(集电体的厚度)的值在方式3中必须为20以下,优选为15以下,特别优选为10以下;另外,在方式1和方式2中没有特别限定,但优选为20以下,更优选为15以下,特别优选为10以下。如果超过该范围,则在高电流密度充放电时,有时集电体会因焦耳热而产生发热,正极受到损伤。For the ratio of the thickness of the positive electrode active material layer and the current collector, the value of (the thickness of the active material layer on the side before injecting the nonaqueous electrolyte)/(the thickness of the current collector) must be 20 or less in mode 3, preferably It is 15 or less, particularly preferably 10 or less; in addition, it is not particularly limited in Embodiment 1 and Embodiment 2, but it is preferably 20 or less, more preferably 15 or less, and particularly preferably 10 or less. If this range is exceeded, the current collector may generate heat due to Joule heat during high current density charge and discharge, and the positive electrode may be damaged.
对于(注入非水电解液之前的一侧的活性物质层厚度)/(集电体的厚度)的值的下限,在方式1中必须为1.6以上,优选为1.8以上,特别优选为2以上;另外,在方式1和方式2中没有特别限定,但优选为1.6以上,更优选为1.8以上,进一步优选为2以上。如果低于该范围,则由于集电体相对于正极活性物质的体积比增加,电池的容量减少,有时不能得到高输出。The lower limit of the value of (the thickness of the active material layer on the side before injecting the nonaqueous electrolyte)/(the thickness of the current collector) must be 1.6 or more in mode 1, preferably 1.8 or more, particularly preferably 2 or more; Moreover, although it does not specifically limit in the form 1 and the form 2, Preferably it is 1.6 or more, More preferably, it is 1.8 or more, More preferably, it is 2 or more. Below this range, since the volume ratio of the current collector to the positive electrode active material increases, the capacity of the battery decreases, and high output may not be obtained.
上述“导电材料在正极活性物质层中的含量”、“正极活性物质层的密度”、“正极活性物质层和集电体的厚度之比,即(注入非水电解液之前的一侧的活性物质层厚度)/(集电体的厚度)的值”如果组合各自的优选的范围,则可形成更优选的高输出的锂二次电池。The above-mentioned "content of conductive material in the positive electrode active material layer", "density of the positive electrode active material layer", "ratio of the thickness of the positive electrode active material layer and the current collector, that is (the activity of the side before injecting the non-aqueous electrolyte) A more preferable high-power lithium secondary battery can be formed by combining the respective preferable ranges of "substance layer thickness)/(current collector thickness)".
[[电极面积]][[Electrode Area]]
使用本发明的非水电解液时,从高输出以及提高高温时的稳定性的观点来看,优选正极活性物质层的面积比电池外壳的外表面积大,这与上述是相同的。When using the non-aqueous electrolytic solution of the present invention, from the viewpoint of high output and improved stability at high temperature, it is preferable that the area of the positive electrode active material layer is larger than the outer surface area of the battery case, which is the same as above.
[[放电容量]][[Discharge capacity]]
使用本发明的二次电池用非水电解液时,装在1个二次电池的电池外壳中的电池要素所具有的电容(将电池由满充电状态放电至放电状态时的电容)如果为3安培小时(Ah)以上,则输出特性的提高效果变大,故优选。When using the non-aqueous electrolytic solution for secondary batteries of the present invention, if the capacitance (capacity when the battery is discharged from a fully charged state to a discharged state) of the battery elements contained in the battery case of one secondary battery is 3 If the ampere-hour (Ah) or more is greater, the effect of improving the output characteristics becomes greater, which is preferable.
因此,正极板等的设计等也与上述相同。Therefore, the design and the like of the positive electrode plate and the like are also the same as described above.
[[正极板的厚度]][[Thickness of positive plate]]
正极板的厚度没有特别限定,与上述相同。The thickness of the positive electrode plate is not particularly limited, and is the same as above.
<正极[5]><Positive pole [5]>
下面,对本发明的锂二次电池中使用的正极[5]进行说明。Next, the positive electrode [5] used in the lithium secondary battery of the present invention will be described.
[正极[5]的正极活性物质][Positive electrode active material of positive electrode [5]]
下面,对正极[5]中使用的正极活性物质进行说明。Next, the positive electrode active material used in the positive electrode [5] will be described.
[[组成]][[composition]]
作为正极活性物质,只要是能够以电化学方式吸留和放出锂离子的物质即可,没有特别限制。优选含有锂和至少一种过渡金属的物质,例如可列举锂-过渡金属复合氧化物、含有锂的过渡金属磷酸化合物。The positive electrode active material is not particularly limited as long as it can electrochemically occlude and release lithium ions. A substance containing lithium and at least one transition metal is preferable, and examples thereof include lithium-transition metal composite oxides and lithium-containing transition metal phosphate compounds.
作为锂-过渡金属复合氧化物的过渡金属,优选V、Ti、Cr、Mn、Fe、Co、Ni、Cu等,作为具体例子,可列举LiCoO2等锂-钴复合氧化物;LiNiO2等锂-镍复合氧化物;LiMnO2、LiMn2O4、Li2MnO3等锂-锰复合氧化物;用Al、Ti、V、Cr、Mn、Fe、Co、Li、Ni、Cu、Zn、Mg、Ga、Zr、Si等其他金属来替代形成这些锂-过渡金属复合氧化物主体的过渡金属原子的一部分而得到的物质等。As the transition metal of the lithium-transition metal composite oxide, V, Ti, Cr, Mn, Fe, Co, Ni, Cu, etc. are preferable, and specific examples include lithium-cobalt composite oxides such as LiCoO 2 and lithium-cobalt composite oxides such as LiNiO 2 . - Nickel composite oxides; LiMnO 2 , LiMn 2 O 4 , Li 2 MnO 3 and other lithium-manganese composite oxides; Al, Ti, V, Cr, Mn, Fe, Co, Li, Ni, Cu, Zn, Mg , Ga, Zr, Si, and other metals instead of a part of the transition metal atoms forming the main body of these lithium-transition metal composite oxides.
上述锂-钴复合氧化物没有特别限定,但优选具有下述组成式(8)表示的组成的物质。The lithium-cobalt composite oxide is not particularly limited, but preferably has a composition represented by the following composition formula (8).
LixCo(1-y)M1 yO2 组成式(8)Li x Co (1-y) M 1 y O 2 composition formula (8)
[组成式(8)中,M1表示选自Ni、Mn、Al、Fe、Ti、Mg、Cr、Ga、Cu、Zn和Nb中的至少一种元素,x表示满足0<x≤1.2的数,y表示满足0.05≤y≤0.8的数]。[In the composition formula (8), M1 represents at least one element selected from Ni, Mn, Al, Fe, Ti, Mg, Cr, Ga, Cu, Zn and Nb, and x represents an element satisfying 0<x≤1.2 number, y represents the number satisfying 0.05≤y≤0.8].
在组成式(8)中,M1特别优选Ni、Mn、Al、Fe,另外,x特别优选0.2≤x≤1.15,y特别优选0.1≤y≤0.5。In the composition formula (8), M 1 is particularly preferably Ni, Mn, Al, and Fe, and x is particularly preferably 0.2≤x≤1.15, and y is particularly preferably 0.1≤y≤0.5.
上述锂-镍复合氧化物没有特别限定,但优选具有下述组成式(9)表示的组成的物质。The lithium-nickel composite oxide is not particularly limited, but preferably has a composition represented by the following composition formula (9).
LixNi(1-y)M2 yO2 组成式(9)Li x Ni (1-y) M 2 y O 2 composition formula (9)
[组成式(9)中,M2表示选自Co、Mn、Al、Fe、Ti、Mg、Cr、Ga、Cu、Zn和Nb中的至少一种元素,x表示满足0<x≤1.2的数,y表示满足0.05≤y≤0.8的数]。[In the composition formula (9), M represents at least one element selected from Co, Mn, Al, Fe, Ti, Mg, Cr, Ga, Cu, Zn and Nb, and x represents an element satisfying 0<x≤1.2 number, y represents the number satisfying 0.05≤y≤0.8].
在组成式(9)中,M2特别优选Co、Mn、Al、Fe,另外,x特别优选0.2≤x≤1.15,y特别优选0.1≤y≤0.5。In the composition formula (9), M2 is particularly preferably Co, Mn, Al, and Fe, and x is particularly preferably 0.2≤x≤1.15, and y is particularly preferably 0.1≤y≤0.5.
上述锂-锰复合氧化物没有特别限定,但优选具有下述组成式(10)表示的组成的物质。The lithium-manganese composite oxide is not particularly limited, but preferably has a composition represented by the following composition formula (10).
LixMn(1-y)M3 yO2 组成式(10)Li x Mn (1-y) M 3 y O 2 composition formula (10)
[组成式(10)中,M3表示选自Ni、Co、Al、Fe、Ti、Mg、Cr、Ga、Cu、Zn和Nb中的至少一种元素,x表示满足0<x≤1.2的数,y表示满足0.05≤y≤0.8的数]。[In the composition formula (10), M represents at least one element selected from Ni, Co, Al, Fe, Ti, Mg, Cr, Ga, Cu, Zn and Nb, and x represents an element satisfying 0<x≤1.2 number, y represents the number satisfying 0.05≤y≤0.8].
在组成式(10)中,M3特别优选Ni、Co、Fe,另外,x特别优选0.2≤x≤1.15,y特别优选0.1≤y≤0.7。In the composition formula (10), M 3 is particularly preferably Ni, Co, and Fe, and x is particularly preferably 0.2≤x≤1.15, and y is particularly preferably 0.1≤y≤0.7.
作为上述锂-锰复合氧化物,优选具有下述组成式(11)表示的组成的物质。As the above-mentioned lithium-manganese composite oxide, one having a composition represented by the following composition formula (11) is preferable.
LixMn(2-y)M4 yO4 组成式(11)Li x Mn (2-y) M 4 y O 4 composition formula (11)
[组成式(11)中,M4表示选自Ni、Co、Al、Fe、Ti、Mg、Cr、Ga、Cu、Zn和Nb中的至少一种元素,x表示满足0<x≤1.2的数,y表示满足0.05≤y≤0.8的数]。[In the composition formula (11), M represents at least one element selected from Ni, Co, Al, Fe, Ti, Mg, Cr, Ga, Cu, Zn and Nb, and x represents an element satisfying 0<x≤1.2 number, y represents the number satisfying 0.05≤y≤0.8].
在组成式(11)中,M4特别优选Ni、Co、Al、Mg,另外,x特别优选0.05≤x≤1.15,y特别优选0.1≤y≤0.7。In the composition formula (11), M 4 is particularly preferably Ni, Co, Al, and Mg, and x is particularly preferably 0.05≤x≤1.15, and y is particularly preferably 0.1≤y≤0.7.
作为上述锂-锰复合氧化物,优选具有下述组成式(12)表示的组成的物质。As the lithium-manganese composite oxide, one having a composition represented by the following composition formula (12) is preferable.
LixMn(1-y)M5 yO3 组成式(12)Li x Mn (1-y) M 5 y O 3 composition formula (12)
[组成式(12)中,M5表示选自Ni、Co、Al、Fe、Ti、Mg、Cr、Ga、Cu、Zn和Nb中的至少一种元素,x表示满足0<x≤2.4的数,y表示满足0.05≤y≤0.8的数]。[In the composition formula (12), M represents at least one element selected from Ni, Co, Al, Fe, Ti, Mg, Cr, Ga, Cu, Zn and Nb, and x represents an element satisfying 0<x≤2.4 number, y represents the number satisfying 0.05≤y≤0.8].
在组成式(12)中,M5特别优选Ni、Co、Al、Mg,另外,x特别优选0.1≤x≤2.3,y特别优选0.1≤y≤0.5。In the composition formula (12), M 5 is particularly preferably Ni, Co, Al, and Mg, and x is particularly preferably 0.1≤x≤2.3, and y is particularly preferably 0.1≤y≤0.5.
作为含有锂的过渡金属磷酸化合物的过渡金属,优选V、Ti、Cr、Mn、Fe、Co、Ni、Cu等,作为具体例子,例如可列举LiFePO4、Li3Fe2(PO4)3、LiFeP2O7等磷酸铁类;LiCoPO4等磷酸钴类;用Al、Ti、V、Cr、Mn、Fe、Co、Li、Ni、Cu、Zn、Mg、Ga、Zr、Nb、Si等其他金属来替代形成这些含有锂的过渡金属磷酸化合物的主体的过渡金属原子的一部分而得到的物质等。The transition metal of the lithium-containing transition metal phosphate compound is preferably V, Ti, Cr, Mn, Fe, Co, Ni, Cu, etc., and specific examples include LiFePO 4 , Li 3 Fe 2 (PO 4 ) 3 , Iron phosphate such as LiFeP 2 O 7 ; Cobalt phosphate such as LiCoPO 4 ; Al, Ti, V, Cr, Mn, Fe, Co, Li, Ni, Cu, Zn, Mg, Ga, Zr, Nb, Si and others Substitution of a part of the transition metal atoms forming the main body of these lithium-containing transition metal phosphate compounds with a metal, etc.
上述磷酸铁类没有特别限定,但优选具有下述组成式(13)表示的组成的物质。The above-mentioned iron phosphates are not particularly limited, but preferably have a composition represented by the following composition formula (13).
LixFe(1-y)M6 yPO4 组成式(13)Li x Fe (1-y) M 6 y PO 4 composition formula (13)
[组成式(13)中,M6表示选自Ni、Co、Mn、Al、Ti、Mg、Cr、Ga、Cu、Zn和Nb中的至少一种元素,x表示满足0<x≤1.2的数,y表示满足0.05≤y≤0.8的数]。[In the composition formula (13), M represents at least one element selected from Ni, Co, Mn, Al, Ti, Mg, Cr, Ga, Cu, Zn and Nb, and x represents an element satisfying 0<x≤1.2 number, y represents the number satisfying 0.05≤y≤0.8].
在组成式(13)中,M6特别优选Ni、Co、Mn、Al,另外,x特别优选0.2≤x≤1.15,y特别优选0.1≤y≤0.5。In the composition formula (13), M6 is particularly preferably Ni, Co, Mn, and Al, and x is particularly preferably 0.2≤x≤1.15, and y is particularly preferably 0.1≤y≤0.5.
本发明中的正极活性物质优选以任意的组合和比例组合使用上述正极活性物质中的组成不同的2种以上物质。由于各种正极活性物质各自所擅长的性能不同,因此正极活性物质优选根据目标电池的用途来组合需要的性能。一般来说,认为通过组合可使性能相加平均,但对于寿命来说,意外地有时能进一步得到寿命长的正极活性物质的效果,通过组合寿命比较良好的正极活性物质和寿命比较差但其他性能良好的正极活性物质,可以实现本发明的高输出、大容量、长寿命的锂二次电池。The positive electrode active material in the present invention is preferably used in combination of two or more materials having different compositions among the above positive electrode active materials in any combination and ratio. Since various positive electrode active materials have different performances, it is preferable to combine required performances of the positive electrode active material according to the intended use of the battery. In general, it is considered that the performance can be added and averaged by combination, but for life, the effect of a long-lived positive electrode active material can be further obtained unexpectedly, and by combining a positive electrode active material with a relatively good life and a relatively poor life but others. The positive electrode active material with good performance can realize the lithium secondary battery with high output, large capacity and long life of the present invention.
作为优选的组合的例子,可列举如下组合:As an example of a preferred combination, the following combinations can be enumerated:
组成式(8)表示的正极活性物质和组成式(10)表示的正极活性物质、The positive electrode active material represented by composition formula (8) and the positive electrode active material represented by composition formula (10),
组成式(8)表示的正极活性物质和组成式(11)表示的正极活性物质、The positive electrode active material represented by composition formula (8) and the positive electrode active material represented by composition formula (11),
组成式(8)表示的正极活性物质和组成式(12)表示的正极活性物质、The positive electrode active material represented by composition formula (8) and the positive electrode active material represented by composition formula (12),
组成式(8)表示的正极活性物质和组成式(13)表示的正极活性物质、The positive electrode active material represented by composition formula (8) and the positive electrode active material represented by composition formula (13),
组成式(9)表示的正极活性物质和组成式(10)表示的正极活性物质、The positive electrode active material represented by composition formula (9) and the positive electrode active material represented by composition formula (10),
组成式(9)表示的正极活性物质和组成式(11)表示的正极活性物质、The positive electrode active material represented by composition formula (9) and the positive electrode active material represented by composition formula (11),
组成式(9)表示的正极活性物质和组成式(12)表示的正极活性物质、The positive electrode active material represented by the composition formula (9) and the positive electrode active material represented by the composition formula (12),
组成式(9)表示的正极活性物质和组成式(13)表示的正极活性物质、The positive electrode active material represented by composition formula (9) and the positive electrode active material represented by composition formula (13),
组成式(10)表示的正极活性物质和组成式(11)表示的正极活性物质、The positive electrode active material represented by the composition formula (10) and the positive electrode active material represented by the composition formula (11),
组成式(10)表示的正极活性物质和组成式(12)表示的正极活性物质、The positive electrode active material represented by composition formula (10) and the positive electrode active material represented by composition formula (12),
组成式(10)表示的正极活性物质和组成式(13)表示的正极活性物质。A positive electrode active material represented by compositional formula (10) and a positive electrode active material represented by compositional formula (13).
特别优选的是,It is particularly preferred that
组成式(9)表示的正极活性物质和组成式(11)表示的正极活性物质、The positive electrode active material represented by composition formula (9) and the positive electrode active material represented by composition formula (11),
组成式(10)表示的正极活性物质和组成式(11)表示的正极活性物质。A positive electrode active material represented by compositional formula (10) and a positive electrode active material represented by compositional formula (11).
组合比例没有特别限定,但优选为10∶90~90∶10,更优选为20∶80~80∶20。The combination ratio is not particularly limited, but is preferably 10:90 to 90:10, more preferably 20:80 to 80:20.
本发明的锂二次电池中使用的正极中,含有组成不同的2种以上的正极活性物质,优选其中的至少一种正极活性物质的BET比表面积、平均一次粒径、中值粒径d50和/或振实密度(以下,简记为“下述物性”)在下述特定的范围内。组成不同的2种以上的正极活性物质中,也包含某种物性不在下述范围内的正极活性物质,但优选2种以上的正极活性物质的所有物质的某种物性均在下述范围内。“下述任一种物性在下述特定范围内的所有正极活性物质”相对于“正极中含有的所有正极活性物质”的含有比例依赖于其物性,没有特别限定,但优选为30质量%以上,更优选为50质量%以上,特别优选是其全部。优选正极所含有的2种以上的正极活性物质中的每一种,其下述物性中的任意一种以上的物性处于下述特定的范围内,更优选任意的2种以上物性处于下述特定的范围内,特别优选任意的3种以上物性处于下述特定的范围内,最优选下述所有物性均处于下述特定的范围内。In the positive electrode used in the lithium secondary battery of the present invention, there are more than two kinds of positive electrode active materials with different compositions , preferably the BET specific surface area, average primary particle size, and median particle size d of at least one positive electrode active material wherein And/or the tap density (hereinafter, abbreviated as "the following physical properties") is within the following specific range. Two or more positive electrode active materials having different compositions also include positive electrode active materials whose physical properties do not fall within the following ranges, but it is preferable that all of the two or more positive electrode active materials have certain physical properties within the following ranges. The content ratio of "all positive electrode active materials having any of the following physical properties within the following specific ranges" relative to "all positive electrode active materials contained in the positive electrode" depends on the physical properties and is not particularly limited, but is preferably 30% by mass or more, More preferably, it is 50 mass % or more, Especially preferably, it is all. Preferably, each of the two or more positive electrode active materials contained in the positive electrode has any one or more of the following physical properties within the following specific ranges, and more preferably any two or more physical properties are within the following specific ranges. It is particularly preferred that any three or more physical properties are within the following specific ranges, and most preferably all of the following physical properties are within the following specific ranges.
[[BET比表面积]][[BET specific surface area]]
该正极活性物质中的至少一种正极活性物质的BET比表面积优选为0.4m2/g以上,更优选为0.5m2/g以上,进一步优选为0.6m2/g以上,其上限为2m2/g以下,优选为1.8m2/g以下,更优选为1.5m2/g以下。如果BET比表面积比该范围小,则电池性能容易降低,而如果BET比表面积比该范围大,则振实密度难以提高,在形成正极活性物质时的涂布性上有时容易产生问题。The BET specific surface area of at least one positive electrode active material in the positive electrode active material is preferably 0.4 m 2 /g or more, more preferably 0.5 m 2 /g or more, still more preferably 0.6 m 2 /g or more, and the upper limit is 2 m 2 /g or less, preferably 1.8 m 2 /g or less, more preferably 1.5 m 2 /g or less. If the BET specific surface area is smaller than this range, the battery performance is likely to decrease, and if the BET specific surface area is larger than this range, it is difficult to increase the tap density, and problems may easily occur in coating properties when forming a positive electrode active material.
BET比表面积定义为如下的值:使用表面积计(例如,大仓理研制造的全自动表面积测定装置),在氮流通下在150℃对试料进行30分钟预干燥,然后使用正确调整为氮相对于大气压的相对压力值为0.3的氮氦混合气体,通过采用气体流动法进行的氮吸附BET 1点法而测定的值。The BET specific surface area is defined as the following value: Using a surface area meter (for example, a fully automatic surface area measuring device manufactured by Okura Riken), the sample is pre-dried at 150°C for 30 minutes under nitrogen flow, and then adjusted to the nitrogen relative The value measured by the nitrogen adsorption BET 1-point method using the gas flow method for a nitrogen-helium mixed gas with a relative pressure value of 0.3 at atmospheric pressure.
[[平均一次粒径]][[average primary particle size]]
作为该正极活性物质中的至少一种正极活性物质的平均一次粒径,优选为0.1μm以上,更优选为0.2μm以上,进一步优选为0.3μm以上,最优选为0.4μm以上,其上限优选为2μm以下,更优选为1.6μm以下,进一步优选为1.3μm以下,最优选为1μm以下。如果超过上述上限,则难以形成球状的二次粒子,对粉末填充性带来不良影响,或者比表面积大幅降低,因此,输出特性等电池性能降低的可能性有时变高。相反,如果低于上述下限,则通常由于结晶不完全,因此有时产生充放电的可逆性差等问题。As the average primary particle diameter of at least one positive electrode active material in the positive electrode active material, it is preferably 0.1 μm or more, more preferably 0.2 μm or more, further preferably 0.3 μm or more, most preferably 0.4 μm or more, and the upper limit is preferably 2 μm or less, more preferably 1.6 μm or less, still more preferably 1.3 μm or less, most preferably 1 μm or less. If the above upper limit is exceeded, it is difficult to form spherical secondary particles, which adversely affects the powder fillability, or the specific surface area is greatly reduced, and thus battery performance such as output characteristics may decrease in possibility. On the contrary, if it is less than the above-mentioned lower limit, problems such as poor reversibility of charging and discharging may arise due to incomplete crystallization.
另外,一次粒径可通过使用扫描电子显微镜(SEM)的观察来测定。具体地,通过如下方法求出:在倍率10000倍的照片中,对任意的50个一次粒子求出水平方向的直线上的由一次粒子的左右边界线产生的切片的最长值,并取其平均值。虽然存在一次粒子凝聚而形成二次粒子的情况,但在该情况下,也只对一次粒子进行测定。In addition, the primary particle size can be measured by observation using a scanning electron microscope (SEM). Specifically, it is obtained by the following method: in a photograph with a magnification of 10,000 times, for any 50 primary particles, the longest value of the slice generated by the left and right boundary lines of the primary particles on a straight line in the horizontal direction is obtained, and the maximum value is obtained. average value. Although primary particles may aggregate to form secondary particles, in this case, only primary particles are measured.
[[中值粒径d50]][[Median particle size d 50 ]]
该正极活性物质中的至少一种正极活性物质的粒子的中值粒径d50(一次粒子凝聚而形成二次粒子时,为二次粒子直径)优选为1μm以上,更优选为1.2μm以上,进一步优选为1.5μm以上,最优选为2μm以上,其上限优选为20μm以下,更优选为18μm以下,进一步优选为16μm以下,最优选为15μm以下。如果低于上述下限,则有时不能得到高振实密度的产品,而如果超过上限,则由于粒子内的锂的扩散需要时间,因此有时会产生电池性能降低,或者在制成电池的正极、也就是将活性物质和导电剂或粘合剂等在溶剂中浆料化再涂布成薄膜状时,产生条纹等问题。这里,通过混合2种以上具有不同的中值粒径d50的正极活性物质,可以进一步提高制作正极时的填充性。The median diameter d50 (the secondary particle diameter when the primary particles are aggregated to form secondary particles) of the particles of at least one positive electrode active material in the positive electrode active material is preferably 1 μm or more, more preferably 1.2 μm or more, More preferably 1.5 μm or more, most preferably 2 μm or more, the upper limit is preferably 20 μm or less, more preferably 18 μm or less, still more preferably 16 μm or less, most preferably 15 μm or less. If it is lower than the above-mentioned lower limit, sometimes a product with a high tap density cannot be obtained, and if it exceeds the upper limit, since the diffusion of lithium in the particles takes time, the performance of the battery may sometimes decrease, or the positive electrode of the battery, that is, the When active materials, conductive agents, binders, etc. are slurried in a solvent and then coated into a film, problems such as streaks occur. Here, by mixing two or more positive electrode active materials having different median diameters d 50 , the filling property at the time of producing the positive electrode can be further improved.
本发明中的中值粒径d50可以利用公知的激光衍射/散射式粒度分布测定装置来测定。使用HORIBA公司制造的LA-920作为粒度分布计时,作为测定时使用的分散介质,使用0.1质量%六偏磷酸钠水溶液,进行5分钟超声波分散后,设定测定折射率1.24进行测定。The median diameter d 50 in the present invention can be measured using a known laser diffraction/scattering particle size distribution analyzer. LA-920 manufactured by HORIBA was used as a particle size distribution meter, and as a dispersion medium used in the measurement, 0.1 mass % sodium hexametaphosphate aqueous solution was used, ultrasonic dispersion was carried out for 5 minutes, and the measurement refractive index was set to 1.24 for measurement.
[[[振实密度]]][[[Tap Density]]]
该正极活性物质中的至少一种正极活性物质的振实密度优选为1.3g/cm3以上,更优选为1.5g/cm3以上,进一步优选为1.6g/cm3以上,最优选为1.7g/cm3以上。正极活性物质的振实密度如果低于上述下限,则在形成正极活性物质层时,需要的分散介质量增加,同时导电材料或粘合剂的必要量增加,正极活性物质在正极活性物质层中的填充率受到制约,电池容量有时受到制约。通过使用振实密度高的复合氧化物粉末,可以形成高密度的正极活性物质层。一般来说,振实密度越大越优选,没有特别的上限,但如果振实密度太大,则正极活性物质层内的以非水电解液为介质的锂离子的扩散成为决定速度的因素,负荷特性有时容易降低,因此,振实密度的上限优选为2.7g/cm3以下,更优选为2.5g/cm3以下。The tap density of at least one positive active material in the positive active material is preferably 1.3 g/cm or more, more preferably 1.5 g/cm or more, further preferably 1.6 g/cm or more, most preferably 1.7 g /cm 3 or more. If the tap density of the positive electrode active material is lower than the above-mentioned lower limit, then when forming the positive electrode active material layer, the amount of dispersion medium required increases, and the necessary amount of conductive material or binder increases simultaneously, and the positive electrode active material is in the positive electrode active material layer. The filling rate of the battery is restricted, and the battery capacity is sometimes restricted. By using a composite oxide powder with a high tap density, a high-density positive electrode active material layer can be formed. Generally speaking, the larger the tap density is, the more preferable, there is no special upper limit, but if the tap density is too large, the diffusion of lithium ions in the positive electrode active material layer with the non-aqueous electrolyte as a medium becomes a factor determining the speed. Since the properties are likely to decrease in some cases, the upper limit of the tap density is preferably 2.7 g/cm 3 or less, more preferably 2.5 g/cm 3 or less.
在本发明中,振实密度如下定义:使试料通过孔径300μm的筛子落到20cm3的振实容器中,充满容器容积之后,使用粉末密度测定器(例如,seishin企业社制造的Tap densor),进行1000次冲程长度10mm的振动,将此时的容积密度作为振实密度。In the present invention, the tap density is defined as follows: the sample is dropped into a 20 cm 3 tapped container through a sieve with an aperture of 300 μm, and after the container volume is filled, a powder density measuring device (for example, Tap densor manufactured by Seishin Co., Ltd.) is used. , vibration with a stroke length of 10 mm was performed 1000 times, and the bulk density at this time was taken as the tap density.
[[表面包覆]][[Cladding]]
该正极活性物质中的至少一种正极活性物质优选使用其表面附着有与构成其正极活性物质主体的正极活性物质或芯的正极活性物质组成不同的物质(以下,简记为“表面附着物质”)。表面附着物质的种类、附着方法、附着量等与上述相同。At least one positive active material in the positive active material preferably uses a material (hereinafter referred to as "surface-attached material" for short) that is attached to its surface with a positive active material that constitutes the main body of the positive active material or a positive active material of the core. ). The type, method of adhesion, amount of adhesion, etc. of the surface adhesion substance are the same as above.
[[形状]][[shape]]
该正极活性物质中的至少一种正极活性物质的粒子形状可采用如以往使用那样的与上述相同的形状。The particle shape of at least one positive electrode active material among the positive electrode active materials may be the same shape as the above as conventionally used.
[[制造法]][[Manufacturing method]]
作为正极活性物质的制造法,可使用与上述同样的作为无机化合物的制造法的通常的方法。As a method for producing the positive electrode active material, a general method as a method for producing an inorganic compound similar to the above can be used.
[正极[5]的正极的结构][Structure of the positive electrode of the positive electrode [5]]
下面,对正极[5]中使用的正极的结构进行叙述。Next, the structure of the positive electrode used in the positive electrode [5] will be described.
在本发明中,电极结构和制作方法、正极活性物质层的压实、导电材料、正极活性物质层的制造时使用的粘合材料、用于形成浆料的液体介质、集电体、电极面积、放电容量、正极板的厚度等与上述相同。在制作正极时,正极活性物质可以事先混合2种以上使用,也可以在制作正极时同时加入来进行混合。In the present invention, electrode structure and production method, compaction of positive electrode active material layer, conductive material, binder material used in the manufacture of positive electrode active material layer, liquid medium for forming slurry, current collector, electrode area , discharge capacity, thickness of positive electrode plate, etc. are the same as above. When producing the positive electrode, two or more types of positive electrode active materials may be mixed beforehand, or may be added and mixed at the same time when producing the positive electrode.
<负极><negative electrode>
本发明的锂二次电池中使用的负极,只要是在集电体上形成含有能够吸留和放出锂离子的活性物质的活性物质层的负极即可,没有特别限制,但该负极优选选自下述负极[1]~负极[10]中的任意一种负极:The negative electrode used in the lithium secondary battery of the present invention is not particularly limited as long as an active material layer containing an active material capable of absorbing and releasing lithium ions is formed on the current collector, but the negative electrode is preferably selected from Any one of the following negative poles [1] to negative poles [10]:
负极[1]:含有2种以上结晶性不同的碳质物质作为负极活性物质的负极;Negative electrode [1]: a negative electrode containing two or more carbonaceous substances with different crystallinity as the negative electrode active material;
负极[2]:含有无定形碳质作为负极活性物质的负极,所述无定形碳质通过广角X射线衍射法测定的(002)面的面间距(d002)为0.337nm以上,微晶大小(Lc)为80nm以下,采用氩离子激光拉曼光谱法测定的以1360cm-1的峰强度相对于1580cm-1的峰强度之比定义的拉曼R值为0.2以上;Negative pole [2]: the negative pole that contains amorphous carbonaceous as negative electrode active material, and the interplanar spacing (d002) of (002) plane of described amorphous carbonaceous by wide-angle X-ray diffractometry is more than 0.337nm, crystallite size ( Lc) is less than 80nm, and the Raman R value defined by the ratio of the peak intensity at 1360cm -1 to the peak intensity at 1580cm -1 measured by argon ion laser Raman spectroscopy is 0.2 or more;
负极[3]:含有金属氧化物作为负极活性物质的负极,所述金属氧化物含有能够吸留和放出锂离子的钛;Negative electrode [3]: a negative electrode containing a metal oxide as the negative electrode active material, and the metal oxide contains titanium capable of absorbing and releasing lithium ions;
负极[4]:含有碳质物质作为负极活性物质的负极,所述碳质物质的圆形度为0.85以上,表面官能团量O/C值为0~0.01;Negative electrode [4]: a negative electrode containing carbonaceous material as the negative electrode active material, the circularity of the carbonaceous material is above 0.85, and the O/C value of the amount of surface functional groups is 0-0.01;
负极[5]:含有异取向性碳复合物作为负极活性物质的负极,所述异取向性碳复合物含有2种以上取向性不同的碳质物质;Negative electrode [5]: a negative electrode containing a hetero-orientation carbon composite as the anode active material, and the hetero-orientation carbon composite contains two or more carbonaceous substances with different orientations;
负极[6]:含有石墨质碳粒子作为负极活性物质的负极,所述石墨质碳粒子的圆形度为0.85以上,通过广角X射线衍射法测定的(002)面的面间距(d002)低于0.337nm,采用氩离子激光拉曼光谱法测定的以1360cm-1的峰强度相对于1580cm-1的峰强度之比定义的拉曼R值为0.12~0.8;Negative electrode [6]: a negative electrode containing graphitic carbon particles as the negative electrode active material, the circularity of the graphitic carbon particles is 0.85 or more, and the interplanar distance (d002) of the (002) plane measured by wide-angle X-ray diffraction method is low At 0.337nm, the Raman R value defined by the ratio of the peak intensity at 1360cm -1 to the peak intensity at 1580cm -1 measured by argon ion laser Raman spectroscopy is 0.12-0.8;
负极[7]:含有下述的包含多元素的负极活性物质(C’)作为负极活性物质的负极,所述包含多元素的负极活性物质(C’)含有选自Al、Pb、Zn、Sn、Bi、In、Mg、Ga、Cd、Ag、Si、B、Au、Pt、Pd和Sb的锂吸留金属(A’)和/或锂吸留合金(B’)中的至少一种,并且含有C和/或N作为元素Z;Negative electrode [7]: a negative electrode containing the following multi-element-containing negative electrode active material (C') as the negative electrode active material, the multi-element-containing negative electrode active material (C') containing , Bi, In, Mg, Ga, Cd, Ag, Si, B, Au, Pt, Pd, and Sb lithium storage metal (A') and/or lithium storage alloy (B'), and contains C and/or N as element Z;
负极[8]:含有性质不同的2种以上的负极活性物质作为负极活性物质的负极;Negative electrode [8]: a negative electrode containing two or more negative electrode active materials with different properties as the negative electrode active material;
负极[9]:含有振实密度为0.1g/cm3以上,并且采用水银孔隙率计测定的相当于直径0.01μm~1μm范围的粒子的微孔容积为0.01mL/g以上的负极活性物质的负极;Negative electrode [9]: Containing a negative electrode active material with a tap density of 0.1 g/cm 3 or more and a pore volume equivalent to particles in the range of 0.01 μm to 1 μm in diameter measured by a mercury porosimeter of 0.01 mL/g or more negative electrode;
负极[10]:充电到该负极的标称容量的60%时,由负极的对置电池产生的反应电阻为500Ω以下的负极。Negative pole [10]: when charged to 60% of the nominal capacity of the negative pole, the reaction resistance produced by the opposite battery of the negative pole is a negative pole below 500Ω.
下面,首先对本发明的锂二次电池中通常使用的负极进行说明。Next, first, a negative electrode generally used in the lithium secondary battery of the present invention will be described.
[负极活性物质][Negative electrode active material]
下面,对负极中通常使用的负极活性物质进行叙述。Next, negative electrode active materials commonly used in negative electrodes will be described.
[[组成]][[composition]]
作为负极活性物质,只要能够以电化学方式吸留和放出锂离子即可,没有特别的限制,可列举碳质材料、氧化锡或氧化硅等金属氧化物、金属复合氧化物、锂单质或锂铝合金等锂合金、Sn或Si等可以与锂形成合金的金属等。它们可以单独使用,也可以以任意的组合和比例组合使用2种以上。其中,从安全性观点来看,优选使用碳质材料或锂复合氧化物。The negative electrode active material is not particularly limited as long as it can electrochemically occlude and release lithium ions, and examples include carbonaceous materials, metal oxides such as tin oxide or silicon oxide, metal composite oxides, lithium simple substances, or lithium ions. Lithium alloys such as aluminum alloys, metals such as Sn and Si that can form alloys with lithium, and the like. These may be used individually, and may be used combining 2 or more types by arbitrary combinations and ratios. Among them, carbonaceous materials or lithium composite oxides are preferably used from the viewpoint of safety.
作为金属复合氧化物,只要可以吸留和放出锂即可,没有特别的限制,但在高电流密度充放电特性这一点上来看,优选含有钛和/或锂作为构成成分。The metal composite oxide is not particularly limited as long as it can occlude and release lithium, but it is preferable to contain titanium and/or lithium as a constituent in terms of high current density charge and discharge characteristics.
作为碳质材料,从初期不可逆容量、高电流密度充放电特性的平衡来看,优选以下碳质材料:As carbonaceous materials, from the balance of initial irreversible capacity and high current density charge and discharge characteristics, the following carbonaceous materials are preferred:
(1)天然石墨;(1) Natural graphite;
(2)人造碳质物质以及人造石墨物质;将碳质物质[例如,天然石墨、煤类焦炭、石油类焦炭、煤类沥青、石油类沥青或者将这些沥青氧化处理而得到的物质、针状焦、沥青焦炭、以及将它们部分石墨化而得到的碳材料;炉黑、乙炔黑、沥青类碳纤维等有机物的热分解产物、可碳化的有机物(例如,从软沥青到硬沥青的煤焦油沥青或者干馏液化油等煤类重油、常压残油、减压残油等直馏类重油;原油、石脑油等热分解时副产的乙烯焦油等分解类石油重油;以及苊烯、十环烯、蒽、菲等芳香族烃;吩嗪或吖啶等氮环化合物;噻吩、联二噻吩等硫环化合物;联苯、三联苯等聚苯;聚氯乙烯、聚乙烯醇、聚乙烯醇缩丁醛、它们的不溶解处理物、含氮的聚丙烯腈、聚吡咯等有机高分子;含硫的聚噻吩、聚苯乙烯等有机高分子;以纤维素、木质素、甘露聚糖、聚半乳糖醛酸、壳聚糖、蔗糖为代表的多糖类等天然高分子;聚苯硫醚、聚苯醚等热塑性树脂;糠醇树脂、酚醛树脂、酰亚胺树脂等热固性树脂)以及它们的碳化物;或者将可碳化的有机物溶解于苯、甲苯、二甲苯、喹啉、正己烷等低分子有机溶剂中而形成的溶液以及它们的碳化物]在400~3200℃范围内进行一次以上的热处理而形成的碳材料;(2) Artificial carbonaceous substances and artificial graphite substances; carbonaceous substances [such as natural graphite, coal coke, petroleum coke, coal pitch, petroleum pitch, or substances obtained by oxidation of these pitches, needle-shaped Coke, pitch coke, and carbon materials obtained by partially graphitizing them; thermal decomposition products of organic substances such as furnace black, acetylene black, and pitch-based carbon fibers, and carbonizable organic substances (for example, coal tar pitch from soft pitch to hard pitch) Or dry distillation liquefied oil and other coal-based heavy oil, atmospheric residual oil, vacuum residual oil and other straight-run heavy oil; crude oil, naphtha and other by-products of thermal decomposition of ethylene tar and other decomposed heavy oil; and acenaphthylene, decacyclic Aromatic hydrocarbons such as alkene, anthracene, and phenanthrene; nitrogen ring compounds such as phenazine or acridine; sulfur ring compounds such as thiophene and dithiophene; polyphenylenes such as biphenyl and terphenyl; polyvinyl chloride, polyvinyl alcohol, polyvinyl alcohol Butyral, their insoluble processed products, nitrogen-containing polyacrylonitrile, polypyrrole and other organic polymers; sulfur-containing polythiophene, polystyrene and other organic polymers; cellulose, lignin, mannan, Natural polymers such as polygalacturonic acid, chitosan, and sucrose; thermoplastic resins such as polyphenylene sulfide and polyphenylene ether; thermosetting resins such as furfuryl alcohol resins, phenolic resins, and imide resins) and their carbides; or solutions formed by dissolving carbonizable organic substances in low-molecular organic solvents such as benzene, toluene, xylene, quinoline, and n-hexane, and their carbides] in the range of 400 to 3200 ° C for more than one time Carbon materials formed by heat treatment;
(3)负极活性物质层由至少2种以上具有不同结晶性的碳质构成、和/或其不同结晶性的碳质具有相接触的界面的碳材料;(3) The negative electrode active material layer is composed of at least two carbonaceous materials with different crystallinity, and/or carbon materials with different crystallinity carbonaceous interfaces that have a contact interface;
(4)负极活性物质层由至少2种以上具有不同取向性的碳质构成、和/或其不同取向性的碳质具有相接触的界面的碳材料。(4) The negative electrode active material layer is composed of at least two kinds of carbonaceous materials with different orientations, and/or a carbon material in which the carbonaceous materials with different orientations have a contact interface.
[负极的结构、物性和制备方法][Structure, Physical Properties and Preparation Method of Negative Electrode]
关于碳材料的性质、包含碳材料的负极电极、电极化方法、集电体和锂二次电池,优选同时满足以下所示的(1)~(9)项中的任1项或多项。Regarding the properties of the carbon material, the negative electrode containing the carbon material, the electrode polarization method, the current collector and the lithium secondary battery, it is preferable to simultaneously satisfy any one or more of the following items (1) to (9).
(1)X射线参数(1) X-ray parameters
碳材料通过采用学振法的X射线衍射求得的晶格面(002)的d值(层间距离)优选为0.335nm以上,其上限通常为0.36nm以下,优选为0.35mm以下,更优选为0.345nm以下。另外,通过采用学振法的X射线衍射求得的碳材料的微晶尺寸(Lc)优选为1nm以上,更优选为1.5nm以上。The d value (interlayer distance) of the lattice plane (002) obtained by X-ray diffraction using the Gakujin method for the carbon material is preferably 0.335 nm or more, and its upper limit is usually 0.36 nm or less, preferably 0.35 mm or less, more preferably 0.345nm or less. In addition, the crystallite size (Lc) of the carbon material obtained by X-ray diffraction using the Gakushin method is preferably 1 nm or more, more preferably 1.5 nm or more.
(2)灰分(2) Ash content
碳质材料中所含的灰分相对于碳质材料的总质量优选为1质量%以下,更优选为0.5质量%以下,特别优选为0.1质量%以下,作为下限,优选为1ppm以上。如果超过上述范围,则充放电时由于与非水电解液的反应而引起的电池性能的劣化不能忽视。如果低于该范围,则在制造上需要较长时间和能量以及用于防止污染的设备,有时成本会上升。The ash contained in the carbonaceous material is preferably 1% by mass or less, more preferably 0.5% by mass or less, particularly preferably 0.1% by mass or less, and the lower limit is preferably 1 ppm or more based on the total mass of the carbonaceous material. If the above range is exceeded, the deterioration of battery performance due to the reaction with the non-aqueous electrolytic solution at the time of charging and discharging cannot be ignored. If it is less than this range, long time and energy are required for manufacturing and equipment for preventing pollution, and the cost may increase.
(3)体积基准平均粒径(3) Volume-based average particle size
碳质材料的体积基准平均粒径是通过激光衍射/散射法求得的体积基准的平均粒径(中值粒径),通常为1μm以上,优选为3μm以上,更优选为5μm以上,进一步优选为7μm以上。另外,其上限通常为100μm以下,优选为50μm以下,更优选为40μm以下,进一步优选为30μm以下,特别优选为25μm以下。如果低于上述范围,则不可逆容量增大,有时导致初期电池容量的损失。另外,如果超过上述范围,则通过涂布制作电极时,容易形成不均匀的涂布面,有时在电池制作工序上是不优选的。The volume-based average particle size of the carbonaceous material is the volume-based average particle size (median particle size) obtained by the laser diffraction/scattering method, and is usually 1 μm or more, preferably 3 μm or more, more preferably 5 μm or more, and even more preferably 7 μm or more. In addition, the upper limit thereof is usually 100 μm or less, preferably 50 μm or less, more preferably 40 μm or less, still more preferably 30 μm or less, particularly preferably 25 μm or less. If it is less than the above-mentioned range, the irreversible capacity may increase, resulting in loss of initial battery capacity. Moreover, if it exceeds the said range, when an electrode is produced by coating, a non-uniform coating surface will be easily formed, and it may be unpreferable in a battery production process.
在本发明中,体积基准平均粒径用中值粒径来定义,所述中值粒径通过以下方法测定:使碳粉末分散在作为表面活性剂的聚氧乙烯(20)山梨糖醇酐单月桂酸酯的0.2质量%水溶液(约10mL)中,使用激光衍射/散射式粒度分布计(例如,堀场制作所公司制造的LA-700)来测定。In the present invention, the volume-based average particle diameter is defined by a median particle diameter measured by dispersing carbon powder in polyoxyethylene (20) sorbitan as a surfactant. In a 0.2% by mass aqueous solution (about 10 mL) of monolaurate, it measures using a laser diffraction/scattering particle size distribution meter (for example, LA-700 manufactured by Horiba, Ltd.).
(4)拉曼R值、拉曼半值宽度(4) Raman R value, Raman half value width
使用氩离子激光拉曼光法测定的碳质材料的R值通常为0.01以上,优选为0.03以上,更优选为0.1以上,其上限为1.5以下,优选为1.2以下,更优选为1.0以下,进一步优选为0.5以下的范围。R值如果低于该范围,则粒子表面的结晶性过高,随着充放电Li进入层间的位点(サイト)有时变少。即,充电接受性有时降低。另外,涂布在集电体上之后,通过压制使负极高密度化时,结晶容易取向为与电极板平行的方向,有时导致负荷特性降低。另一方面,如果超过该范围,则粒子表面的结晶性降低,与非水电解液的反应性增加,有时导致效率降低或产生的气体增加。The R value of the carbonaceous material measured by the argon ion laser Raman optical method is usually 0.01 or more, preferably 0.03 or more, more preferably 0.1 or more, and its upper limit is 1.5 or less, preferably 1.2 or less, more preferably 1.0 or less, and further It is preferably in the range of 0.5 or less. If the R value is lower than this range, the crystallinity of the particle surface is too high, and the sites (sites) where Li enters the interlayer during charging and discharging may decrease. That is, charge acceptance may be lowered. In addition, when the anode is densified by pressing after coating on the current collector, the crystals tend to be oriented in a direction parallel to the electrode plate, which may result in a decrease in load characteristics. On the other hand, if it exceeds this range, the crystallinity of the particle surface decreases, the reactivity with the non-aqueous electrolyte solution increases, and the efficiency may decrease or the generated gas may increase.
另外,碳质材料在1580cm-1附近的拉曼半值宽度没有特别的限制,通常为10cm-1以上,优选为15cm-1以上,其上限通常为100cm-1以下,优选为80cm-1以下,更优选为60cm-1以下,进一步优选为40cm-1以下的范围。如果拉曼半值宽度低于该范围,则粒子表面的结晶性过高,随着充放电Li进入层间的位点(サイト)有时变少。即,充电接受性有时降低。另外,涂布在集电体上之后,通过压制使负极高密度化时,结晶容易取向为与电极板平行的方向,有时导致负荷特性降低。另一方面,如果高于该范围,则粒子表面的结晶性降低,与非水电解液的反应性增加,有时导致效率降低或产生的气体增加。In addition, the Raman half-value width of the carbonaceous material around 1580 cm -1 is not particularly limited, but it is usually 10 cm -1 or more, preferably 15 cm -1 or more, and its upper limit is usually 100 cm -1 or less, preferably 80 cm -1 or less , more preferably not more than 60 cm -1 , still more preferably not more than 40 cm -1 . If the Raman half-value width is below this range, the crystallinity of the particle surface is too high, and there may be fewer sites (sites) where Li enters the interlayer during charge and discharge. That is, charge acceptance may be lowered. In addition, when the anode is densified by pressing after coating on the current collector, the crystals tend to be oriented in a direction parallel to the electrode plate, which may result in a decrease in load characteristics. On the other hand, if it exceeds this range, the crystallinity of the particle surface decreases, the reactivity with the non-aqueous electrolyte solution increases, and the efficiency may decrease or the generated gas may increase.
拉曼光谱的测定如下进行:使用拉曼分光器(例如日本分光公司制造的拉曼分光器),使试样自然落下并填充在测定池(cell)内,对池内的试样表面照射氩离子激光,同时使池在与激光垂直的面内旋转。对于得到的拉曼光谱,测定1580cm-1附近的峰PA的强度IA和1360cm-1附近的峰PB的强度IB,计算其强度比R(R=IB/IA),将其定义为碳质材料的拉曼R值。另外,测定得到的拉曼光谱在1580cm-1附近的峰PA的半值宽度,将其定义为碳质材料的拉曼半值宽度。The measurement of the Raman spectrum is carried out as follows: Using a Raman spectrometer (for example, a Raman spectrometer manufactured by JASCO Corporation), the sample is naturally dropped and filled in a measurement cell, and argon ions are irradiated on the surface of the sample in the cell. laser while rotating the pool in a plane perpendicular to the laser. For the obtained Raman spectrum, the intensity I A of the peak PA near 1580 cm -1 and the intensity I B of the peak P B near 1360 cm -1 were measured , and the intensity ratio R (R= IB / IA ) was calculated. It is defined as the Raman R value of the carbonaceous material. In addition, the half-value width of the peak PA in the vicinity of 1580 cm −1 in the obtained Raman spectrum was measured, and this was defined as the Raman half-value width of the carbonaceous material.
另外,这里的拉曼测定条件如下。In addition, the Raman measurement conditions here are as follows.
·氩离子激光波长:514.5nmArgon ion laser wavelength: 514.5nm
·试样上的激光功率:15~25mW·Laser power on the sample: 15~25mW
·分辨率:10~20cm-1 ·Resolution: 10~20cm -1
·测定范围:1100cm-1~1730cm-1 · Measuring range: 1100cm -1 ~ 1730cm -1
·R值、半值宽度分析:本底(background)处理R value, half value width analysis: background (background) processing
·光滑(smoothing)处理:单纯平均、卷积5点(コンボリユシヨン5ポイント)Smoothing (smoothing) processing: simple average, convolution 5 points (コンボリユシヨン5ポイント)
(5)BET比表面积(5) BET specific surface area
使用BET法测定的本发明的碳质材料的比表面积通常为0.1m2/g以上,优选为0.7m2/g以上,更优选为1.0m2/g以上,进一步优选为1.5m2/g以上。其上限通常为100m2/g以下,优选为25m2/g以下,更优选为15m2/g以下,进一步优选为10m2/g以下。比表面积的值如果低于上述范围,则用作负极的情况下,在充电时锂的接受性变差,锂容易在电极表面析出。另一方面,如果高于上述范围,则用作负极材料时,与非水电解液的反应性增加,产生的气体容易变多,有时难以得到优选的电池。The specific surface area of the carbonaceous material of the present invention measured by the BET method is usually 0.1 m 2 /g or more, preferably 0.7 m 2 /g or more, more preferably 1.0 m 2 /g or more, and still more preferably 1.5 m 2 /g above. The upper limit thereof is usually 100 m 2 /g or less, preferably 25 m 2 /g or less, more preferably 15 m 2 /g or less, still more preferably 10 m 2 /g or less. If the value of the specific surface area is less than the above range, when it is used as a negative electrode, the acceptance of lithium at the time of charging will be deteriorated, and lithium will be easily deposited on the surface of the electrode. On the other hand, if it is higher than the above range, when used as a negative electrode material, the reactivity with the non-aqueous electrolytic solution increases, the gas generated tends to increase, and it may be difficult to obtain a preferable battery.
采用BET法测定的比表面积使用如下测定的值:使用表面积计(例如,大仓理研制造的全自动表面积测定装置),在氮气流通下,在350℃下对试样进行15分钟预干燥,然后,使用氮气相对于大气压的相对压力值准确调整为0.3的氮氦混合气体,通过采用气体流动法的氮吸附BET 1点法测定。The specific surface area measured by the BET method uses the value measured by pre-drying the sample at 350° C. for 15 minutes under nitrogen flow using a surface area meter (for example, a fully automatic surface area measuring device manufactured by Okura Riken), and then , using a nitrogen-helium mixed gas whose relative pressure value of nitrogen relative to the atmospheric pressure is accurately adjusted to 0.3, and measured by the nitrogen adsorption BET 1-point method using the gas flow method.
(6)微孔径分布(6) Micropore size distribution
本发明中使用的碳质材料的微孔径分布是通过水银孔隙率计(水银压入法)求得的相当于微孔直径为0.01μm~1μm的粒子内的空隙、由于粒子表面的高低不平造成的凹凸、粒子间的接触面等的量为0.01mL/g以上,优选为0.05mL/g以上,更优选为0.1mL/g以上,其上限为0.6mL/g以下,优选为0.4mL/g以下,更优选为0.3mL/g以下的范围。如果超过该范围,则有时在制造极板时需要大量的粘合剂。如果低于该范围,则高电流密度充放电特性降低,并且,有时不能得到充放电时的电极膨胀收缩的缓和效果。The pore size distribution of the carbonaceous material used in the present invention is obtained by mercury porosimetry (mercury intrusion method), which is equivalent to the voids in the particles with a pore diameter of 0.01 μm to 1 μm, which is caused by the unevenness of the particle surface. The amount of unevenness, the contact surface between particles, etc. is 0.01mL/g or more, preferably 0.05mL/g or more, more preferably 0.1mL/g or more, and the upper limit is 0.6mL/g or less, preferably 0.4mL/g or less, more preferably in the range of 0.3 mL/g or less. If it exceeds this range, a large amount of binder may be required when producing an electrode plate. If it is less than this range, the high current density charge-discharge characteristics will deteriorate, and the effect of alleviating electrode expansion and contraction during charge-discharge may not be obtained.
另外,相当于0.01μm~100μm范围的微孔径的总微孔容积优选为0.1mL/g以上,更优选为0.25mL/g以上,进一步优选为0.4mL/g以上,其上限为10mL/g以下,优选为5mL/g以下,更优选为2mL/g以下的范围。如果高于该范围,则有时在制造极板时需要大量的粘合剂。如果低于该范围,则在制造极板时有时不能得到增稠剂或粘结剂的分散效果。In addition, the total pore volume corresponding to the pore diameter in the range of 0.01 μm to 100 μm is preferably 0.1 mL/g or more, more preferably 0.25 mL/g or more, still more preferably 0.4 mL/g or more, and the upper limit thereof is 10 mL/g or less , preferably 5 mL/g or less, more preferably 2 mL/g or less. If it is higher than this range, a large amount of binder may be required when producing an electrode plate. If it is less than this range, the dispersion effect of the thickener or the binder may not be obtained at the time of manufacturing the electrode plate.
另外,平均微孔径优选为0.05μm以上,更优选为0.1μm以上,进一步优选为0.5μm以上,其上限为50μm以下,优选为20μm以下,更优选为10μm以下的范围。如果超过该范围,则有时需要大量的粘合剂。如果低于该范围,则有时高电流密度充放电特性降低。In addition, the average pore diameter is preferably 0.05 μm or more, more preferably 0.1 μm or more, even more preferably 0.5 μm or more, and the upper limit thereof is 50 μm or less, preferably 20 μm or less, more preferably 10 μm or less. If this range is exceeded, a large amount of adhesive is sometimes required. If it is less than this range, the high current density charge-discharge characteristics may deteriorate.
作为用作水银孔隙率计的装置,可以使用水银孔隙测定仪(autopore(オ一トポア)9520;micrometritics(マイクロメリテツクス)公司制造)。将约0.2g试样密封入粉末用容器中,在室温、真空下(50μmHg以下)进行10分钟脱气,进行前处理。接着,减压至4psia(约28kPa),导入水银,从4psia(约28kPa)阶段状升压至40000psia(约280MPa)后,再降压至25psia(约170kPa)。升压时的阶段数为80阶段以上,在各阶段中,在10秒的平衡时间后,测定水银压入量。使用Washburn式从这样得到的水银压入曲线计算出微孔径分布。另外,水银的表面张力(γ)为485dyne/cm,接触角(φ)为140°。平均微孔径使用累积微孔体积达到50%时的微孔径。As an apparatus used as a mercury porosimeter, a mercury porosimeter (autopore (Autopore) 9520; manufactured by Micrometrics (Micromeritex) Co., Ltd.) can be used. About 0.2 g of the sample was sealed in a container for powder, and degassed at room temperature under vacuum (50 μmHg or less) for 10 minutes to perform pretreatment. Next, depressurize to 4psia (about 28kPa), introduce mercury, increase the pressure from 4psia (about 28kPa) to 40000psia (about 280MPa) stepwise, and then lower the pressure to 25psia (about 170kPa). The number of stages during the pressurization was 80 or more, and the amount of mercury intrusion was measured after an equilibration time of 10 seconds in each stage. The micropore size distribution was calculated from the mercury intrusion curve thus obtained using the Washburn formula. In addition, the surface tension (γ) of mercury is 485 dyne/cm, and the contact angle (φ) is 140°. As the average pore diameter, the pore diameter at which the cumulative pore volume reaches 50% is used.
(7)圆形度(7) Circularity
使用圆形度作为碳质材料的球形的程度,其粒径为3~40μm范围的粒子的圆形度优选为0.1以上,特别优选为0.5以上,更优选为0.8以上,进一步优选为0.85以上,最优选为0.9以上。圆形度大时,高电流密度充放电特性提高,因此优选。Using circularity as the spherical degree of the carbonaceous material, the circularity of particles whose particle diameter is in the range of 3 to 40 μm is preferably 0.1 or more, particularly preferably 0.5 or more, more preferably 0.8 or more, and even more preferably 0.85 or more, Most preferably, it is 0.9 or more. When the circularity is large, the high current density charge and discharge characteristics are improved, which is preferable.
圆形度用下式定义,圆形度为1时,为理论真球。The circularity is defined by the following formula. When the circularity is 1, it is a theoretical true sphere.
圆形度=(具有与粒子投影形状相同面积的等效圆的周长)/(粒子投影形状的实际周长)Circularity = (perimeter of an equivalent circle having the same area as the particle projected shape)/(actual perimeter of the particle projected shape)
作为圆形度的值使用如下测定的值:使用例如流动式粒子图像分析装置(例如,Sysmex Industrial(シスメツクスインダストリアル)公司制造的FPIA),将约0.2g试样分散于作为表面活性剂的聚氧乙烯(20)山梨糖醇酐单月桂酸酯的0.2质量%水溶液(约50mL)中,以60W的输出功率照射1分钟28kHz的超声波后,指定0.6~400μm为检测范围,对粒径3~40μm范围的粒子进行测定。As the value of circularity, the value measured as follows is used: using, for example, a flow-type particle image analyzer (for example, FPIA manufactured by Sysmex Industrial (Sysmex Industrial) Co., Ltd.), about 0.2 g of the sample is dispersed in a surfactant as a surfactant. After irradiating a 0.2% by mass aqueous solution (approximately 50 mL) of polyoxyethylene (20) sorbitan monolaurate with 28 kHz ultrasonic waves at an output of 60 W for 1 minute, specify a detection range of 0.6 to 400 μm. Particles in the range of ~40μm are measured.
提高圆形度的方法没有特别的限制,但通过实施球形化处理(机械能处理)使之成为球形可以使制成电极体时粒子间空隙的形状整齐统一,故优选。作为球形化处理的例子,可以举出,通过施加剪切力、压缩力来机械地接近球形的方法,通过粘合剂或粒子自身具有的附着力将多个微粒造粒的机械/物理处理方法等。There is no particular limitation on the method of improving the circularity, but it is preferable to make the shape of the spheroid by performing the spheroidization treatment (mechanical energy treatment) because the shape of the gap between the particles can be uniformed when the electrode body is produced. Examples of spheroidization treatment include a method of mechanically approaching a spherical shape by applying a shear force or a compressive force, and a mechanical/physical treatment method of granulating a plurality of fine particles by the adhesive force possessed by a binder or the particles themselves. wait.
(8)真密度(8) True density
碳质材料的真密度通常为1.4g/cm3以上,优选为1.6g/cm3以上,更优选为1.8g/cm3以上,进一步优选为2.0g/cm3以上,其上限为2.26g/cm3以下。上限为石墨的理论值。如果低于该范围,则碳的结晶性过低,初期不可逆容量有时会增大。在本发明中,真密度定义为通过使用丁醇的液相置换法(比重计法)测定的值。The true density of the carbonaceous material is usually above 1.4 g/cm 3 , preferably above 1.6 g/cm 3 , more preferably above 1.8 g/cm 3 , even more preferably above 2.0 g/cm 3 , and its upper limit is 2.26 g/cm 3 or above. cm3 or less. The upper limit is the theoretical value of graphite. If it is less than this range, the crystallinity of carbon may be too low, and the initial irreversible capacity may increase. In the present invention, the true density is defined as a value measured by a liquid phase displacement method (pycnometer method) using butanol.
(9)振实密度(9) Tap density
碳质材料的振实密度通常为0.1g/cm3以上,优选为0.5g/cm3以上,更优选为0.7g/cm3以上,特别优选为1.0g/cm3以上。另外,其上限优选为2g/cm3以下,更优选为1.8g/cm3以下,特别优选为1.6g/cm3以下。振实密度如果低于该范围,则作为负极使用时,填充密度很难提高,有时不能得到高容量的电池。另一方面,如果超过该范围,则电极中的粒子间的空隙过少,难以确保粒子间的导电性,有时难以得到优选的电池特性。振实密度通过与正极中阐述的方法同样的方法来测定,并通过该方法定义。The tap density of the carbonaceous material is usually 0.1 g/cm 3 or more, preferably 0.5 g/cm 3 or more, more preferably 0.7 g/cm 3 or more, particularly preferably 1.0 g/cm 3 or more. In addition, the upper limit thereof is preferably 2 g/cm 3 or less, more preferably 1.8 g/cm 3 or less, particularly preferably 1.6 g/cm 3 or less. When the tap density is lower than this range, it is difficult to increase the packing density when used as a negative electrode, and a high-capacity battery may not be obtained. On the other hand, if it exceeds this range, the gaps between the particles in the electrode will be too small, making it difficult to ensure the conductivity between the particles, and it may be difficult to obtain preferable battery characteristics. The tap density is measured by the same method as described for the positive electrode, and is defined by this method.
(10)取向比(10) Orientation ratio
碳质材料的取向比通常为0.005以上,优选为0.01以上,更优选为0.015以上,其上限为理论值0.67以下的范围。如果低于该范围,则有时高密度充放电特性会降低。The orientation ratio of the carbonaceous material is usually not less than 0.005, preferably not less than 0.01, more preferably not less than 0.015, and the upper limit thereof is in the range of not more than a theoretical value of 0.67. If it is less than this range, the high-density charge-discharge characteristics may deteriorate.
取向比是在将试样加压成型后通过X射线衍射来进行测定。将0.47g试样填充到直径17mm的成型机中,以600kgf/cm2压缩得到成型体,使用粘土固定上述成型体,使其与测定用试样保持架的面为同一面,然后测定X射线衍射。从得到的碳的(110)衍射和(004)衍射的峰强度计算出以(110)衍射积分强度/(004)衍射积分强度表示的比值,将该比值定义为活性物质的取向比。The orientation ratio was measured by X-ray diffraction after press-molding the sample. Fill a 0.47g sample into a molding machine with a diameter of 17mm, compress it at 600kgf/ cm2 to obtain a molded body, fix the molded body with clay so that it is on the same side as the surface of the sample holder for measurement, and then measure the X-ray diffraction. The ratio represented by (110) diffraction integrated intensity/(004) diffraction integrated intensity was calculated from the obtained carbon (110) diffraction and (004) diffraction peak intensities, and this ratio was defined as the orientation ratio of the active material.
这里的X射线衍射测定条件如下。另外,“2θ”表示衍射角。The X-ray diffraction measurement conditions here are as follows. In addition, "2θ" represents a diffraction angle.
·靶:Cu(Kα射线)石墨单色光度仪Target: Cu (Kα ray) graphite monochromator
·狭缝:·Slit:
发散狭缝=0.5度,受光狭缝=0.15mm,散射狭缝=0.5度Divergence slit = 0.5 degrees, light receiving slit = 0.15mm, scattering slit = 0.5 degrees
·测定范围和步进角/测定时间・Measurement range and step angle/measurement time
(110)面:75度≤2θ≤80度1度/60秒(110) surface: 75 degrees ≤ 2θ ≤ 80 degrees 1 degree / 60 seconds
(004)面:52度≤2θ≤57度1度/60秒(004) surface: 52 degrees ≤ 2θ ≤ 57 degrees 1 degree / 60 seconds
(11)长径比(粉末)(11) aspect ratio (powder)
长径比理论上为1以上,上限为10以下,优选为8以下,更优选为5以下。如果超过上限,则制造极板时有时会产生条纹、无法得到均匀的涂布面,高电流密度充放电特性降低。The aspect ratio is theoretically 1 or more, and the upper limit is 10 or less, preferably 8 or less, more preferably 5 or less. When the upper limit is exceeded, streaks may be generated at the time of electrode plate production, a uniform coated surface may not be obtained, and high current density charge-discharge characteristics may be reduced.
另外,长径比用三维观察时碳质材料粒子的最长径A和与其垂直的最短径B的比值A/B来表示。碳粒子的观察通过可以放大观察的扫描型电子显微镜来进行。选择固定在厚度50微米以下的金属板的端面上的任意50个石墨粒子,旋转、倾斜固定有试样的载物台,分别测定这些粒子的A、B,求出A/B的平均值。In addition, the aspect ratio is represented by the ratio A/B of the longest diameter A of the carbonaceous material particle and the shortest diameter B perpendicular thereto when viewed three-dimensionally. Observation of carbon particles was performed with a scanning electron microscope capable of magnified observation. Select any 50 graphite particles fixed on the end face of a metal plate with a thickness of 50 μm or less, rotate and tilt the stage on which the sample is fixed, measure A and B of these particles, and calculate the average value of A/B.
(12)副材料混合(12) Sub-material mixing
所谓“副材料混合”是指负极电极中和/或负极活性物质中含有2种以上性质不同的碳质材料。这里所说的性质,是指X射线衍射参数、中值粒径、长径比、BET比表面积、取向比、拉曼R值、振实密度、真密度、微孔分布、圆形度、灰分量中的一个以上的特性。The so-called "mixture of auxiliary materials" means that two or more carbonaceous materials with different properties are contained in the negative electrode and/or the negative electrode active material. The properties mentioned here refer to X-ray diffraction parameters, median particle size, aspect ratio, BET specific surface area, orientation ratio, Raman R value, tap density, true density, pore distribution, circularity, gray More than one property in a component.
作为特别优选的实施方式,可以举出,体积基准粒度分布在以中值粒径为中心时左右不对称、含有2种以上拉曼R值不同的碳质材料、或X射线参数不同等。As particularly preferred embodiments, the volume-based particle size distribution is asymmetrical around the median diameter, contains two or more carbonaceous materials with different Raman R values, or has different X-ray parameters.
作为其效果的一个例子,可以列举,通过含有天然石墨、人造石墨等石墨;乙炔黑等炭黑;针状焦等无定形碳等碳质材料作为导电剂,可以降低电阻。它们可以单独使用,也可以以任意组合和任意比例组合使用2种以上。作为导电剂添加时,为0.1质量%以上,优选为0.5质量%以上,更优选为0.6质量%以上,其上限为45质量%以下,优选为40质量%的范围。如果低于该范围,则有时难以得到导电性提高的效果。如果高于上述范围,则有时会导致初期不可逆容量的增大。As an example of the effect, it is possible to reduce resistance by including carbonaceous materials such as graphite such as natural graphite and artificial graphite; carbon black such as acetylene black; and amorphous carbon such as needle coke as a conductive agent. These may be used individually, and may be used in combination of 2 or more types by arbitrary combinations and arbitrary ratios. When added as a conductive agent, it is 0.1% by mass or more, preferably 0.5% by mass or more, more preferably 0.6% by mass or more, and the upper limit is 45% by mass or less, preferably 40% by mass. If it is less than this range, it may be difficult to obtain the effect of improving electrical conductivity. If it exceeds the above-mentioned range, the initial irreversible capacity may be increased.
(13)电极制作(13) Electrode fabrication
制作电极可以采用通常的方法。例如,在负极活性物质中添加粘合剂、溶剂、根据需要添加的增稠剂、导电材料、填充材料等,制成浆料,将其涂布在集电体上,干燥后通过压制而形成电极。在非水电解液注入工序之前的阶段,电池的每面的负极活性物质的厚度通常为15μm以上,优选为20μm以上,更优选为30μm以上,其上限为150μm以下,优选为120μm以下,更优选为100μm以下。如果超过该范围,则非水电解液难以浸透到集电体界面附近,因此,高电流密度充放电特性降低。另外,如果低于该范围,则集电体相对于负极活性物质的体积比增加,电池容量有时会减少。另外,可以将负极活性物质进行辊成型制成片状电极,或通过压缩成型制成颗粒电极。A common method can be used to fabricate the electrodes. For example, a binder, solvent, thickener, conductive material, filler, etc. are added to the negative electrode active material to make a slurry, which is coated on the current collector, dried, and pressed to form electrode. In the stage before the non-aqueous electrolyte injection process, the thickness of the negative electrode active material on each side of the battery is usually more than 15 μm, preferably more than 20 μm, more preferably more than 30 μm, and the upper limit is 150 μm or less, preferably 120 μm or less, more preferably 100 μm or less. If it exceeds this range, the non-aqueous electrolytic solution becomes difficult to permeate to the vicinity of the current collector interface, so that the high current density charge and discharge characteristics are degraded. In addition, if it is less than this range, the volume ratio of the current collector to the negative electrode active material increases, and the battery capacity may decrease. In addition, the negative electrode active material can be roll-formed to form a sheet-like electrode, or can be formed into a granular electrode by compression-molding.
(14)集电体(14) Collector
作为集电体,可以任意地使用公知的集电体。作为负极的集电体,可以举出,铜、镍、不锈钢、镀镍钢等金属材料,其中,从加工容易和成本的观点来看,优选铜。集电体的形状在集电体为金属材料时,可列举金属箔、金属圆柱、金属线圈、金属板、金属薄膜、膨胀合金、穿孔金属、发泡金属等。其中,优选金属薄膜、更优选铜箔,进一步优选采用压延法制得的压延铜箔和采用电解法制得的电解铜箔,任何一个都可以作为集电体使用。铜箔的厚度比25μm薄时,可以使用比纯铜强度高的铜合金(磷青铜、钛铜、科森合金、Cu-Cr-Zr合金等)。另外,铝箔由于其比重轻,因此使用于集电体时,可以减少电池的重量,因此优选使用。As the current collector, known current collectors can be used arbitrarily. Examples of the current collector of the negative electrode include metal materials such as copper, nickel, stainless steel, and nickel-plated steel, among which copper is preferred from the viewpoints of ease of processing and cost. The shape of the current collector, when the current collector is a metal material, includes metal foil, metal cylinder, metal coil, metal plate, metal thin film, expanded alloy, perforated metal, foamed metal, and the like. Among them, metal thin films are preferred, copper foils are more preferred, rolled copper foils produced by calendering and electrolytic copper foils produced by electrolysis are further preferred, and any of them can be used as a current collector. When the thickness of the copper foil is thinner than 25 μm, a copper alloy (phosphor bronze, titanium copper, Corson alloy, Cu-Cr-Zr alloy, etc.) stronger than pure copper can be used. In addition, aluminum foil is preferably used because it can reduce the weight of a battery when used for a current collector because of its light specific gravity.
由采用压延法制作的铜箔制作的集电体中,由于铜结晶在压延方向排列,即使将负极紧密弯折或将负极弯折成锐角,都不易断裂,适用于小型圆筒状电池。电解铜箔如下制得:例如,在溶解有铜离子的非水电解液中浸渍金属制造的鼓,边使该鼓旋转边流通电流,由此使铜析出在鼓的表面,再将其剥离。还可以在上述压延铜箔的表面通过电解法析出铜。可以对铜箔的一面或两面进行粗糙化处理或表面处理(例如,厚度为几nm~1μm左右的铬酸盐处理、Ti等的底层处理等)。In the current collector made of copper foil produced by the rolling method, since the copper crystals are arranged in the rolling direction, even if the negative electrode is bent tightly or bent at an acute angle, it is not easy to break, and it is suitable for small cylindrical batteries. Electrodeposited copper foil is produced, for example, by immersing a metal drum in a non-aqueous electrolytic solution in which copper ions are dissolved, and passing an electric current while rotating the drum, thereby depositing copper on the surface of the drum and peeling it off. Copper can also be electrolytically deposited on the surface of the above-mentioned rolled copper foil. Roughening treatment or surface treatment (for example, chromate treatment with a thickness of about several nm to 1 μm, primer treatment such as Ti, etc.) may be performed on one or both sides of the copper foil.
对于集电体基板来讲,要求具有以下物性。The current collector substrate is required to have the following physical properties.
(1)平均表面粗糙度(Ra)(1) Average surface roughness (Ra)
按照JIS B 0601-1994记载的方法中规定的集电体基板的活性物质薄膜形成面的平均表面粗糙度(Ra)没有特别的限制,但通常为0.05μm以上,优选为0.1μm以上,特别优选为0.15μm以上,其上限通常为1.5μm以下,优选为1.3μm以下,特别优选为1.0μm以下。通过使集电体基板的平均表面粗糙度(Ra)在上述下限和上限之间的范围,可以期待良好的充放电循环特性。通过设定为上述下限值以上,与活性物质薄膜的界面面积变大,与活性物质的密合性提高。平均表面粗糙度(Ra)的上限值没有特别的限制,平均表面粗糙度(Ra)超过1.5μm时,难以得到作为电池合适厚度的箔,因此优选为1.5μm以下。According to the method described in JIS B 0601-1994, the average surface roughness (Ra) of the active material thin film formation surface of the current collector substrate is not particularly limited, but it is usually 0.05 μm or more, preferably 0.1 μm or more, particularly preferably It is 0.15 μm or more, and its upper limit is usually 1.5 μm or less, preferably 1.3 μm or less, particularly preferably 1.0 μm or less. When the average surface roughness (Ra) of the current collector substrate is in the range between the above-mentioned lower limit and the upper limit, favorable charge-discharge cycle characteristics can be expected. By setting it as more than the said lower limit, the interface area with an active material thin film will become large, and the adhesiveness with an active material will improve. The upper limit of the average surface roughness (Ra) is not particularly limited. If the average surface roughness (Ra) exceeds 1.5 μm, it will be difficult to obtain a foil with an appropriate thickness as a battery, so it is preferably 1.5 μm or less.
(2)拉伸强度(2) Tensile strength
集电体基板的拉伸强度没有特别的限制,但通常为100N/mm2以上,优选为250N/mm2以上,更优选为400N/mm2以上,特别优选为500N/mm2以上。所谓拉伸强度,是指试验片达到断裂时所需要的最大拉力除以试验片的截面积而得到的值。本发明中的拉伸强度,可采用与测定伸长率同样的装置和方法测定。如果是拉伸强度高的集电体基板,则可以抑制充电/放电时由于活性物质薄膜膨胀/收缩引起的集电体基板的龟裂,从而可以得到良好的循环特性。The tensile strength of the current collector substrate is not particularly limited, but is usually 100 N/mm 2 or higher, preferably 250 N/mm 2 or higher, more preferably 400 N/mm 2 or higher, particularly preferably 500 N/mm 2 or higher. The term "tensile strength" refers to a value obtained by dividing the maximum tensile force required for a test piece to break by the cross-sectional area of the test piece. The tensile strength in the present invention can be measured using the same apparatus and method as for measuring elongation. A current collector substrate with high tensile strength can suppress cracking of the current collector substrate due to expansion/shrinkage of the active material film during charging/discharging, thereby obtaining good cycle characteristics.
(3)0.2%耐力(3) 0.2% Stamina
集电体基板的0.2%耐力没有特别的限制,但通常为30N/mm2以上,优选为150N/mm2以上,特别优选为300N/mm2以上。所谓0.2%耐力,是指产生0.2%的塑性(永久)变形所需要的负荷大小,施加该大小的负荷后,除去负荷也会保持0.2的变形。本发明中的0.2%耐力可通过与测定伸长率相同的装置和方法来测定。如果是0.2%耐力高的集电体基板,则可以抑制充电/放电时由于活性物质薄膜膨胀/收缩引起的集电体基板的塑性变形,从而可以得到良好的循环特性。The 0.2% proof strength of the current collector substrate is not particularly limited, but is usually 30 N/mm 2 or more, preferably 150 N/mm 2 or more, particularly preferably 300 N/mm 2 or more. The so-called 0.2% endurance refers to the load required to produce 0.2% plastic (permanent) deformation. After applying the load of this size, the deformation of 0.2 will be maintained after removing the load. The 0.2% endurance in the present invention can be measured by the same apparatus and method as for measuring elongation. A current collector substrate with a high 0.2% endurance can suppress plastic deformation of the current collector substrate due to expansion/shrinkage of the active material film during charging/discharging, thereby obtaining good cycle characteristics.
金属薄膜的厚度是任意的,通常为1μm以上,优选为3μm以上,更优选为5μm以上。另外,其上限通常为1mm以下,优选为100μm以下,更优选为30μm以下。如果比1μm薄,则强度降低,有时涂布困难。另外,如果比100μm厚,则有时会使电极的形状发生卷曲等变形。另外,金属薄膜也可以是网状的。The thickness of the metal thin film is arbitrary, but it is usually 1 μm or more, preferably 3 μm or more, and more preferably 5 μm or more. In addition, the upper limit thereof is usually 1 mm or less, preferably 100 μm or less, more preferably 30 μm or less. If it is thinner than 1 μm, the strength will decrease and coating may be difficult. In addition, if it is thicker than 100 μm, the shape of the electrode may be deformed such as curling. In addition, the metal thin film may also be in the form of a mesh.
(15)集电体和活性物质层的厚度之比(15) The ratio of the thickness of the current collector to the active material layer
集电体和活性物质层的厚度之比没有特别的限制,(注入非水电解液之前的一侧活性物质层的厚度)/(集电体的厚度)优选为150以下,特别优选为20以下,更优选为10以下,其下限优选为0.1以上,更优选为0.4以下,进一步优选为1以上的范围。如果超过该范围,则高电流密度充放电时,集电体有时会因为焦耳热而产生发热。如果低于该范围,则有时集电体相对于负极活性物质的体积比增加,电池的容量减少。The ratio of the thickness of the current collector to the active material layer is not particularly limited, and (the thickness of the active material layer on one side before injecting the nonaqueous electrolyte)/(thickness of the current collector) is preferably 150 or less, particularly preferably 20 or less , more preferably 10 or less, the lower limit thereof is preferably 0.1 or more, more preferably 0.4 or less, and still more preferably 1 or more. If this range is exceeded, the current collector may generate heat due to Joule heat during high current density charge and discharge. If it is less than this range, the volume ratio of the current collector to the negative electrode active material may increase and the capacity of the battery may decrease.
(16)电极密度(16) Electrode density
将负极活性物质制成电极时的电极结构没有特别的限制,存在于集电体上的活性物质的密度优选为1.0g/cm3以上,更优选为1.2g/cm3以上,进一步优选为1.3g/cm3以上,其上限通常为2.0g/cm3以下,优选为1.9g/cm3以下,更优选为1.8g/cm3以下,进一步优选为1.7g/cm3以下的范围。如果超过该范围,则活性物质被破坏,导致初期不可逆容量增加、非水电解液向集电体/活性物质界面附近的浸透性降低,从而导致高电流密度充放电特性恶化。另外,如果低于该范围,则活性物质间的导电性下降,电池电阻增大,有时单位容积的电池容量降低。The electrode structure when the negative electrode active material is made into an electrode is not particularly limited, and the density of the active material present on the current collector is preferably 1.0 g/cm or more, more preferably 1.2 g/cm or more, and even more preferably 1.3 g/cm 3 or more, and the upper limit is usually 2.0 g/cm 3 or less, preferably 1.9 g/cm 3 or less, more preferably 1.8 g/cm 3 or less, still more preferably 1.7 g/cm 3 or less. If it exceeds this range, the active material will be destroyed, resulting in an increase in the initial irreversible capacity, and a reduction in the permeability of the non-aqueous electrolyte to the vicinity of the current collector/active material interface, resulting in deterioration of high current density charge and discharge characteristics. On the other hand, if it is less than this range, the conductivity between the active materials will decrease, the battery resistance will increase, and the battery capacity per unit volume may decrease.
(17)粘合剂(17) Adhesive
作为粘结活性物质的粘合剂,只要是对非水电解液或制造电极时使用的溶剂稳定的材料即可,没有特别的限制。具体地,可列举聚乙烯、聚丙烯、聚对苯二甲酸乙二醇酯、聚甲基丙烯酸甲酯、芳香族聚酰胺、纤维素、硝基纤维素等树脂类高分子;SBR(丁苯橡胶)、异戊二烯橡胶、丁二烯橡胶、氟橡胶、NBR(丙烯腈-丁二烯橡胶)、苯乙烯-丙烯橡胶等橡胶状高分子;苯乙烯-丁二烯-苯乙烯嵌段共聚物或其加氢物;EPDM(乙烯-丙烯-二烯三元共聚物)、苯乙烯-乙烯-丁二烯-苯乙烯共聚物、苯乙烯-异丁烯-苯乙烯嵌段共聚物或其加氢物等热塑性弹性体状高分子;间规立构1,2聚丁二烯、聚醋酸乙烯、乙烯-醋酸乙烯共聚物、丙烯-α-烯烃共聚物等软质树脂状高分子;聚偏氟乙烯、聚四氟乙烯、氟化聚偏氟乙烯、聚四氟乙烯-乙烯共聚物等氟类高分子;具有碱金属离子(特别是锂离子)的离子传导性的高分子组合物等。它们可以单独使用一种,也可以以任意组合和比例组合使用2种以上。The binder for binding the active material is not particularly limited as long as it is a material stable to the non-aqueous electrolyte solution or the solvent used in producing the electrode. Specifically, resin polymers such as polyethylene, polypropylene, polyethylene terephthalate, polymethyl methacrylate, aromatic polyamide, cellulose, and nitrocellulose; SBR (styrene-butadiene) rubber), isoprene rubber, butadiene rubber, fluororubber, NBR (acrylonitrile-butadiene rubber), styrene-propylene rubber and other rubber-like polymers; styrene-butadiene-styrene block Copolymer or its hydrogenated product; EPDM (ethylene-propylene-diene terpolymer), styrene-ethylene-butadiene-styrene copolymer, styrene-isobutylene-styrene block copolymer or its addition Thermoplastic elastomer-like polymers such as hydrogen compounds; soft resin-like polymers such as syndiotactic 1,2 polybutadiene, polyvinyl acetate, ethylene-vinyl acetate copolymer, propylene-α-olefin copolymer; polybias Fluorine polymers such as vinyl fluoride, polytetrafluoroethylene, fluorinated polyvinylidene fluoride, and polytetrafluoroethylene-ethylene copolymers; polymer compositions having ion conductivity with alkali metal ions (especially lithium ions), etc. These may be used individually by 1 type, and may combine and use 2 or more types by arbitrary combinations and ratios.
作为用于形成浆料的溶剂,只要是可以溶解或分散活性物质、粘合剂以及根据需要使用的增稠剂和导电剂的溶剂即可,其种类没有特别的限制,可以使用水系溶剂或有机类溶剂中的任一种。作为水系溶剂的例子,可列举水、醇等;作为有机类溶剂的例子,可列举N-甲基吡咯烷酮(NMP)、二甲基甲酰胺、二甲基乙酰胺、甲乙酮、环己酮、乙酸甲酯、丙烯酸甲酯、二乙基三胺、N,N-二甲基氨基丙胺、环氧丙烷、四氢呋喃(THF)、甲苯、丙酮、乙醚、二甲基乙酰胺、六甲基磷酰胺、二甲基硫醚、苯、二甲苯、喹啉、吡啶、甲基萘、己烷等。特别是在使用水系溶剂时,同时添加上述增稠剂和分散剂等,使用SBR等的胶乳进行浆料化。另外,它们可以单独使用一种,也可以以任意组合和比例组合使用2种以上。The solvent used to form the slurry is not particularly limited as long as it can dissolve or disperse the active material, binder, and optionally a thickener and conductive agent, and aqueous solvents or organic solvents can be used. any of the solvents. Examples of aqueous solvents include water, alcohol, etc.; examples of organic solvents include N-methylpyrrolidone (NMP), dimethylformamide, dimethylacetamide, methyl ethyl ketone, cyclohexanone, acetic acid Methyl ester, methyl acrylate, diethyltriamine, N,N-dimethylaminopropylamine, propylene oxide, tetrahydrofuran (THF), toluene, acetone, ether, dimethylacetamide, hexamethylphosphoramide, Dimethyl sulfide, benzene, xylene, quinoline, pyridine, methylnaphthalene, hexane, etc. In particular, when using a water-based solvent, add the above-mentioned thickener and dispersant at the same time, and form a slurry using a latex such as SBR. In addition, these may be used individually by 1 type, and may combine and use 2 or more types by arbitrary combinations and ratios.
粘合剂相对于活性物质的比例优选为0.1质量%以上,特别优选为0.5质量%以上,更优选为0.6质量%以上,其上限通常为20质量%以下,优选为15质量%以下,更优选为10质量%以下,进一步优选为8质量%以下的范围。如果超过该范围,则粘合剂的量中不会对电池容量带来帮助的粘合剂比例增加,有时导致电池容量降低。另外,如果低于上述范围,则有时导致负极电极的强度降低。特别是,主要成分中含有以SBR为代表的橡胶状高分子时,粘合剂相对于活性物质的比例通常为0.1质量%以上,优选为0.5质量%以上,更优选为0.6质量%以上,其上限通常为5质量%以下,优选为3质量%以下,更优选为2质量%以下的范围。另外,主要成分中含有以聚偏氟乙烯为代表的氟类高分子时,粘合剂相对于活性物质的比例通常为1质量%以上,优选为2质量%以上,更优选为3质量%以上,其上限通常为15质量%以下,优选为10质量%以下,更优选为8质量%以下。The ratio of the binder to the active material is preferably 0.1% by mass or more, particularly preferably 0.5% by mass or more, more preferably 0.6% by mass or more, and the upper limit thereof is usually 20% by mass or less, preferably 15% by mass or less, more preferably It is 10 mass % or less, More preferably, it is the range of 8 mass % or less. If this range is exceeded, the ratio of the binder that does not contribute to the battery capacity in the amount of the binder increases, resulting in a decrease in the battery capacity in some cases. Moreover, if it is less than the said range, the intensity|strength of a negative electrode may fall. In particular, when a rubber-like polymer represented by SBR is contained in the main component, the ratio of the binder to the active material is usually 0.1% by mass or more, preferably 0.5% by mass or more, more preferably 0.6% by mass or more. The upper limit is usually 5% by mass or less, preferably 3% by mass or less, more preferably 2% by mass or less. In addition, when a fluorine-based polymer typified by polyvinylidene fluoride is contained in the main component, the ratio of the binder to the active material is usually 1% by mass or more, preferably 2% by mass or more, more preferably 3% by mass or more , the upper limit thereof is usually 15% by mass or less, preferably 10% by mass or less, more preferably 8% by mass or less.
增稠剂通常是为了调节浆料的粘度而使用的。作为增稠剂,没有特别的限制,具体地,可列举羧甲基纤维素、甲基纤维素、羟甲基纤维素、乙基纤维素、聚乙烯醇、氧化淀粉、磷酸化淀粉、酪蛋白以及它们的盐等。它们可以单独使用一种,也可以以任意组合和比例组合使用2种以上。另外,在添加增稠剂时,增稠剂相对于活性物质的比例通常为0.1质量%以上,优选为0.5质量%以上,更优选为0.6质量%以上,其上限通常为5质量%以下,优选为3质量%以下,更优选为2质量%以下的范围。如果低于该范围,则有时涂布性显著降低。如果超过该范围,则负极活性物质层所占的活性物质的比例降低,有时产生电池容量降低的问题或负极活性物质之间的电阻增加的问题。Thickeners are generally used to adjust the viscosity of the slurry. The thickener is not particularly limited, and specifically, carboxymethyl cellulose, methyl cellulose, hydroxymethyl cellulose, ethyl cellulose, polyvinyl alcohol, oxidized starch, phosphorylated starch, casein and their salts, etc. These may be used individually by 1 type, and may combine and use 2 or more types by arbitrary combinations and ratios. In addition, when adding a thickener, the ratio of the thickener to the active material is usually 0.1% by mass or more, preferably 0.5% by mass or more, more preferably 0.6% by mass or more, and the upper limit is usually 5% by mass or less, preferably It is 3 mass % or less, More preferably, it is the range of 2 mass % or less. If it is less than this range, coatability may fall remarkably. If it exceeds this range, the ratio of the active material in the negative electrode active material layer decreases, which may cause a problem of a decrease in battery capacity or an increase in the resistance between the negative electrode active materials.
(18)极板取向比(18) plate orientation ratio
极板取向比优选为0.001以上,更优选为0.005以上,特别优选为0.01以上,上限为理论值,即0.67以下。如果低于该范围,则有时高密度充放电特性降低。The electrode plate orientation ratio is preferably 0.001 or more, more preferably 0.005 or more, particularly preferably 0.01 or more, and the upper limit is a theoretical value, ie, 0.67 or less. If it is less than this range, high-density charge-discharge characteristics may deteriorate.
极板取向比的测定如下进行。对压制成目标密度后的负极电极通过X射线衍射测定电极的活性物质取向比。具体方法没有特别限制,作为标准的方法,使用非对称皮尔森(ピアソン)VII作为分布(profile)函数,拟合通过X射线衍射得到的碳的(110)衍射和(004)衍射的峰,进行峰分离,分别计算出(110)衍射和(004)衍射的峰的积分强度。由得到的积分强度,计算出由(110)衍射积分强度/(004)衍射积分强度表示的比例。将该测定计算出的电极的活性物质取向比定义为极板取向比。The measurement of the electrode plate orientation ratio was performed as follows. The active material orientation ratio of the electrode was measured by X-ray diffraction with respect to the negative electrode pressed to the target density. The specific method is not particularly limited, but as a standard method, using asymmetric Pearson (Piason) VII as a distribution (profile) function, fitting the peaks of (110) diffraction and (004) diffraction of carbon obtained by X-ray diffraction, and performing The peaks were separated, and the integrated intensities of the (110) diffraction and (004) diffraction peaks were calculated respectively. From the obtained integrated intensity, the ratio represented by (110) diffraction integrated intensity/(004) diffraction integrated intensity was calculated. The active material orientation ratio of the electrode calculated by this measurement was defined as the plate orientation ratio.
这里的X射线衍射测定条件如下。另外,“2θ”表示衍射角。The X-ray diffraction measurement conditions here are as follows. In addition, "2θ" represents a diffraction angle.
·靶:Cu(Kα线)石墨单色光度仪Target: Cu (Kα line) graphite monochromator
·狭缝:发散狭缝=1度,受光狭缝=0.1mm,散射狭缝=1度· Slit: divergence slit = 1 degree, light receiving slit = 0.1mm, scattering slit = 1 degree
·测定范围和步进角/测定时间・Measurement range and step angle/measurement time
(110)面:76.5度≤2θ≤78.5度0.01度/3秒(110) surface: 76.5 degrees ≤ 2θ ≤ 78.5 degrees 0.01 degrees / 3 seconds
(004)面:53.5度≤2θ≤56.0度0.01度/3秒(004) plane: 53.5 degrees ≤ 2θ ≤ 56.0 degrees 0.01 degrees / 3 seconds
·试样制备:用0.1mm厚的双面胶带将电极固定在玻璃板上·Sample preparation: Fix the electrode on the glass plate with 0.1mm thick double-sided tape
(19)阻抗(19) Impedance
从放电状态充电到标称容量的60%时的负极阻抗优选为100Ω以下,更优选为50Ω以下,特别优选为20Ω以下,和/或双电层容量优选为1×10-6F以上,更优选为1×10-5F,特别优选为1×10-4F。该范围输出特性良好,故优选。The negative electrode impedance when charging to 60% of the nominal capacity from the discharged state is preferably 100Ω or less, more preferably 50Ω or less, particularly preferably 20Ω or less, and/or the electric double layer capacity is preferably 1× 10-6 F or more, more preferably It is preferably 1×10 -5 F, particularly preferably 1×10 -4 F. Since the output characteristics are good in this range, it is preferable.
负极电阻和双电层容量按照下述方法测定。待测定的锂二次电池使用如下的电池:用能够以5小时充电至标称容量的电流值进行充电后,维持20分钟不进行充放电的状态,接着,用能够以1小时将标称容量放电的电流值进行放电,此时的容量为标称容量的80%以上。对于上述放电状态的锂二次电池,通过能够以5小时充电至标称容量的电流值充电到标称容量的60%,立即将锂二次电池转移到氩气氛围下的球形容器内。在不放电或不短路的状态下迅速将该锂二次电池解体,取出负极,如果是双面涂布电极,那么将一面的电极活性物质剥离而不损伤另一面的电极活性物质,将2片负极电极冲裁成12.5mmφ,夹入隔板,使活性物质面没有错离地对置。在隔板和两负极之间滴加电池中使用的非水电解液60μL,进行粘合,保持不与外界接触的状态,使两负极的集电体导电,实施交流阻抗法。测定如下进行,在25℃的温度下,在10-2~105Hz的频率带测定复阻抗,将求得的call·call·plot的负极电阻成分的圆弧近似为半圆,求出表面电阻(R)和双电层容量(Cdl)。The negative electrode resistance and electric double layer capacity were measured by the following methods. The lithium secondary battery to be measured uses the following battery: After being charged with a current value that can be charged to the nominal capacity in 5 hours, it is maintained in a state where it is not charged and discharged for 20 minutes, and then, it is charged with a current value that can be charged to the nominal capacity in 1 hour. The discharge current value is discharged, and the capacity at this time is more than 80% of the nominal capacity. For the lithium secondary battery in the above discharge state, the lithium secondary battery was immediately transferred to a spherical container under an argon atmosphere by charging to 60% of the nominal capacity at a current value capable of charging to the nominal capacity in 5 hours. Quickly disassemble the lithium secondary battery in the state of no discharge or short circuit, and take out the negative electrode. If it is a double-sided coated electrode, peel off the electrode active material on one side without damaging the electrode active material on the other side. The negative electrode was punched out to 12.5 mmφ, and the separators were sandwiched so that the active material surfaces were opposed without being displaced. Between the separator and the two negative electrodes, 60 μL of the non-aqueous electrolyte solution used in the battery was added dropwise to bond them without contact with the outside world, and the current collectors of the two negative electrodes were made conductive, and an AC impedance method was performed. The measurement is carried out as follows. The complex impedance is measured at a temperature of 25°C in the frequency band of 10 -2 to 10 5 Hz, and the arc of the negative electrode resistance component of the obtained call·call·plot is approximated as a semicircle to obtain the surface resistance. (R) and double layer capacity (Cdl).
(20)负极板的面积和厚度(20) Area and thickness of the negative plate
负极板的面积没有特别的限制,通常设计成比对置的正极板稍大,使正极板不会露出到负极板外面。从反复充放电的循环寿命或抑制由于高温保存引起的劣化的观点来看,如果尽可能接近与正极相等的面积,那么可以进一步提高均匀且有效地工作的电极比例,从而特性得以提高,因此优选。特别是,以大电流使用时,该电极面积的设计是重要的。The area of the negative plate is not particularly limited, and it is generally designed to be slightly larger than the opposite positive plate so that the positive plate will not be exposed outside the negative plate. From the point of view of repeated charge-discharge cycle life or suppression of deterioration due to high-temperature storage, if the area is as close as possible to the same area as the positive electrode, the proportion of electrodes that work uniformly and efficiently can be further increased, and the characteristics can be improved. Therefore, it is preferable . In particular, when using a large current, the design of the electrode area is important.
负极板的厚度根据使用的正极板来设计,没有特别的限制,但减去芯材的金属箔厚度之后的复层材料层的厚度通常为15μm以上,优选为20μm以上,更优选为30μm以上,其上限通常为150μm以下,优选为120μm以下,更优选为100μm以下。The thickness of the negative plate is designed according to the positive plate used, and there is no particular limitation, but the thickness of the cladding material layer after subtracting the thickness of the metal foil of the core material is usually more than 15 μm, preferably more than 20 μm, more preferably more than 30 μm, The upper limit thereof is usually 150 μm or less, preferably 120 μm or less, more preferably 100 μm or less.
<负极[1]><negative pole[1]>
以下,对本发明的锂二次电池中使用的负极[1]“含有2种以上作为负极活性物质的结晶性不同的碳质物质的负极”进行说明。Hereinafter, the negative electrode [1] "negative electrode containing two or more carbonaceous materials having different crystallinity" as negative electrode active materials used in the lithium secondary battery of the present invention will be described.
[负极[1]的负极活性物质][Negative electrode active material of negative electrode [1]]
下面,说明负极[1]使用的负极活性物质。Next, the negative electrode active material used for the negative electrode [1] will be described.
[[构成]][[constitute]]
本发明的锂二次电池的负极[1]中使用的负极活性物质的特征在于含有2种以上结晶性不同的碳质物质。这里,“含有2种以上结晶性不同的碳质物质”表示负极中共存有结晶性不同的碳质物质,另外,其共存形态可以是以一个个单一粒子的形式存在并混合在一起的状态,或者是包含在一个二次粒子中,或者是上述二者的混合。另外,作为负极活性物质,优选含有包含2种以上结晶性不同的碳质物质的复合碳质物质,更优选在该复合碳质物质中还含有一种以上与该复合碳质物质在碳质的物性方面不同的碳质物质(碳质材料)作为副材料。The negative electrode active material used in the negative electrode [1] of the lithium secondary battery of the present invention is characterized by containing two or more carbonaceous substances having different crystallinity. Here, "containing two or more carbonaceous substances with different crystallinity" means that carbonaceous substances with different crystallinity coexist in the negative electrode. In addition, the coexistence form may be in the form of individual particles and mixed together. Either contained in a secondary particle, or a mixture of the two. In addition, as the negative electrode active material, it is preferable to contain a composite carbonaceous substance containing two or more carbonaceous substances with different crystallinity, and it is more preferable to also contain one or more carbonaceous substances in the composite carbonaceous substance in the composite carbonaceous substance. Carbonaceous substances (carbonaceous materials) different in physical properties are used as auxiliary materials.
这里所说的“包含在一个二次粒子中”表示结晶性不同的碳质物质被键合约束在一起的状态、物理约束的状态、通过静电约束而维持形状的状态等。这里所说的“物理约束”表示结晶性不同的一种碳质物质夹杂入另一种碳质物质中的状态、相互挂住的状态,所谓“静电约束”表示结晶不同的一种碳质物质通过静电能量附着于另一种碳质物质上的状态。另外,“通过键合约束的状态”表示氢键、共价键、离子键等化学键。Here, "included in one secondary particle" means a state in which carbonaceous substances having different crystallinity are bonded and constrained, a state in which they are physically constrained, a state in which their shape is maintained by electrostatic confinement, and the like. The term "physical restraint" here refers to the state in which one carbonaceous substance with different crystallinity is mixed into another carbonaceous substance, and the state in which they are intertwined, and the so-called "electrostatic restraint" refers to a carbonaceous substance with different crystallinity The state of attaching to another carbonaceous substance by electrostatic energy. In addition, "a state constrained by bonding" means a chemical bond such as a hydrogen bond, a covalent bond, or an ionic bond.
其中,成为芯的碳质物质的至少部分表面具有与通过键合形成的结晶性不同的覆盖层的界面的状态下,结晶性不同的碳质物质间的锂的移动阻抗小,故有利。该覆盖层可以通过与从外部供给的材料和/或它们的改性物的键合来形成,或者通过碳质物质的表面部的材料的改性来形成。这里,所谓覆盖,表示与碳质物质的表面的界面中的至少一部分具有化学键,并显示(1)覆盖全部表面的状态、(2)覆盖碳粒子局部的状态、(3)选择性地覆盖部分表面的状态、(4)存在于含有化学键的极小区域内的状态。另外,在界面上结晶性可以连续变化,也可以不连续变化。Among them, in the state where at least part of the surface of the carbonaceous material serving as the core has an interface with the clad layer having different crystallinity formed by bonding, the transfer resistance of lithium between the carbonaceous material with different crystallinity is small, which is advantageous. This covering layer can be formed by bonding with externally supplied materials and/or their modified products, or by modifying the material of the surface portion of the carbonaceous substance. Here, the so-called coverage means that at least a part of the interface with the surface of the carbonaceous material has a chemical bond, and it means (1) a state covering the entire surface, (2) a state covering a part of the carbon particle, (3) selectively covering a part The state of the surface, (4) the state that exists in an extremely small region containing chemical bonds. In addition, the crystallinity may change continuously or discontinuously at the interface.
复合碳质物质优选具有在粒子状碳质物质上覆盖有结晶性与该粒子状碳质物质不同的碳质物质而形成的界面、和/或在粒子状碳质物质上键合有结晶性与该粒子状碳质物质不同的碳质物质而形成的界面,且该界面的结晶性是连续和/或不连续变化的。“粒子状碳质物质”和“与该粒子状碳质物质结晶性不同的碳质物质”哪一个的结晶性高这一点没有特别的限定,但粒子状碳质物质的结晶性高时,可以实现本发明的上述效果,故优选。The composite carbonaceous material preferably has an interface formed by covering the particulate carbonaceous material with a carbonaceous material different in crystallinity from the particulate carbonaceous material, and/or the particulate carbonaceous material is bonded with a crystallinity and The particulate carbonaceous material is an interface formed by different carbonaceous materials, and the crystallinity of the interface changes continuously and/or discontinuously. Which of the "particulate carbonaceous material" and "the carbonaceous material different in crystallinity from the particulate carbonaceous material" has higher crystallinity is not particularly limited, but when the crystallinity of the particulate carbonaceous material is high, the Since the above-mentioned effects of the present invention are achieved, it is preferable.
这里,结晶性的差异通过采用X射线广角衍射法测定的(002)面的面间距(d002)的差异、Lc的差异、La的差异来判断,从表现出本发明的效果这点来看,优选结晶性的差异以(d002)计为0.0002nm以上,或者La为1nm以上,或者Lc为1nm以上。上述范围中,(d002)的差异优选为0.0005nm以上,更优选为0.001nm以上,进一步优选为0.003nm以上,其上限通常为0.03nm以下,优选为0.02nm以下的范围。如果低于该范围,由结晶性的差异引起的效果有时会变小。另一方面,如果超过上述范围,则结晶性低的部分的结晶性有变低的倾向,由此引起的不可逆容量可能会增加。另外,上述范围中,La或Lc的差异优选为2mm以上,更优选为5nm以上,进一步优选为10nm以上的范围。通常,石墨在100nm以上是不能定义的,因此不能指定上限。如果低于该范围,则结晶性的差异所引起的效果有时变小。Here, the difference in crystallinity is judged by the difference in the interplanar distance (d002) of the (002) plane measured by the X-ray wide-angle diffraction method, the difference in Lc, and the difference in La. From the point of view of expressing the effect of the present invention, Preferably, the difference in crystallinity in terms of (d002) is 0.0002 nm or more, or La is 1 nm or more, or Lc is 1 nm or more. In the above range, the difference in (d002) is preferably 0.0005 nm or more, more preferably 0.001 nm or more, still more preferably 0.003 nm or more, and the upper limit thereof is usually 0.03 nm or less, preferably 0.02 nm or less. If it is less than this range, the effect due to the difference in crystallinity may become small. On the other hand, if it exceeds the above-mentioned range, the crystallinity of the part with low crystallinity tends to decrease, and the irreversible capacity caused by this may increase. In addition, in the above-mentioned range, the difference in La or Lc is preferably 2 mm or more, more preferably 5 nm or more, and still more preferably 10 nm or more. Generally, graphite is not defined above 100nm, so no upper limit can be specified. If it is less than this range, the effect due to the difference in crystallinity may become small.
复合碳质物质是在粒子状碳质物质上覆盖和/或键合了结晶性与该粒子状碳质物质不同的碳质物质而成的,“粒子状碳质物质”和“与该粒子状碳质物质结晶性不同的碳质物质”只要任意一个是石墨类碳质物质,而另一个是低结晶性碳质物质即可,另外,优选“粒子状碳质物质”是石墨类碳质物质,而“与该粒子状碳质物质结晶性不同的碳质物质”是低结晶性碳质物质。A composite carbonaceous substance is formed by covering and/or bonding a carbonaceous substance different in crystallinity from the particulate carbonaceous substance on the particulate carbonaceous substance, and "particulate carbonaceous substance" and "with the granular carbonaceous substance" "Carbonaceous substances with different crystallinity of carbonaceous substances" as long as any one is graphite-like carbonaceous substances, and the other is low-crystalline carbonaceous substances. In addition, it is preferable that "particulate carbonaceous substances" be graphite-like carbonaceous substances , and "a carbonaceous substance different in crystallinity from the particulate carbonaceous substance" is a low-crystalline carbonaceous substance.
[[[粒子状碳质物质]]][[[Particulate carbonaceous matter]]]
作为粒子状碳质物质,优选含有天然石墨和/或人造石墨的石墨类碳质物质,或含有选自比它们结晶性稍低的(a)、(b)和(c)中的至少之一的碳质物质,As the particulate carbonaceous substance, it is preferable to contain natural graphite and/or a graphite-like carbonaceous substance containing artificial graphite, or to contain at least one selected from (a), (b) and (c) which are slightly lower in crystallinity than them. carbonaceous matter,
(a)选自煤类焦炭、石油类焦炭、炉黑、乙炔黑以及沥青类碳纤维中的有机物的热分解物;(a) Thermal decomposition products of organic matter selected from coal coke, petroleum coke, furnace black, acetylene black, and pitch-based carbon fibers;
(b)有机物气体的碳化物;(b) carbides of organic gases;
(c)将(a)或(b)的一部分或全部进行石墨化而得到的碳质物质。(c) A carbonaceous material obtained by graphitizing part or all of (a) or (b).
[[[[石墨类碳质物质]]]][[[[Graphite carbonaceous substances]]]]
粒子状碳质物质优选含有天然石墨和/或人造石墨的石墨类碳质物质。石墨类碳质物质是指采用X射线广角衍射法测定的(002)面的面间距(d002)显示不足0.340nm的结晶性高的各种碳质物质。The particulate carbonaceous material is preferably a graphitic carbonaceous material containing natural graphite and/or artificial graphite. The graphitic carbonaceous substance refers to various carbonaceous substances with high crystallinity whose interplanar distance (d002) of the (002) plane measured by X-ray wide-angle diffraction method is less than 0.340 nm.
作为石墨类碳质物质的具体例子,优选选自以下产品的粉末:天然石墨、人造石墨、或者它们的机械粉碎物、再热处理产品、膨胀石墨的再热处理产品、或者这些石墨的高纯度精制产品。作为上述人造石墨的具体例子,优选将一种以上的有机物质通常在2500℃~3200℃左右的烧结温度下进行石墨化,再通过适当的粉碎方法进行粉末化而得到的物质,所述有机物质选自:煤焦油沥青、煤类重油、常压残油、石油类重油、芳香族烃、含氮环状化合物、含硫环状化合物、聚苯、聚氯乙烯、聚乙烯醇、聚丙烯腈、聚乙烯醇缩丁醛、天然高分子、聚苯硫醚、聚苯醚、糠醇树脂、酚醛树脂、酰亚胺树脂等。As a specific example of graphite-based carbonaceous substances, powders selected from the following products are preferred: natural graphite, artificial graphite, or their mechanically pulverized products, reheated products, reheated products of expanded graphite, or high-purity refined products of these graphites . As a specific example of the above-mentioned artificial graphite, one or more organic substances are preferably graphitized at a sintering temperature of about 2500° C. to 3200° C., and then pulverized by an appropriate pulverization method. The organic substances Selected from: coal tar pitch, coal heavy oil, atmospheric residual oil, petroleum heavy oil, aromatic hydrocarbons, nitrogen-containing cyclic compounds, sulfur-containing cyclic compounds, polyphenylene, polyvinyl chloride, polyvinyl alcohol, polyacrylonitrile , polyvinyl butyral, natural polymer, polyphenylene sulfide, polyphenylene ether, furfuryl alcohol resin, phenolic resin, imide resin, etc.
(石墨类碳质物质的物性)(Physical properties of graphitic carbonaceous material)
石墨类碳质物质的性质优选同时满足下面所示的(1)~(11)中的任意一项或多项。The properties of the graphitic carbonaceous substance preferably simultaneously satisfy any one or more of (1) to (11) shown below.
(1)X射线参数(1) X-ray parameters
石墨类碳质物质通过采用学振法的X射线衍射求得的晶格面(002)的d值(层间距离)优选为0.335nm以上。另外,从定义来看,下限为不足0.340nm,优选为0.337nm以下。如果d值过大,则结晶性降低,有时会增加初期不可逆容量。另一方面,0.335为石墨的理论值。另外,通过采用学振法的X射线衍射求得的碳材料的微晶大小(Lc)通常为30nm以上,优选为50nm以上,更优选为100nm以上的范围。如果低于该范围,则结晶性降低,有可能增加初期不可逆容量。The graphitic carbonaceous material preferably has a d value (interlayer distance) of the lattice plane (002) obtained by X-ray diffraction using the Gakushin method of 0.335 nm or more. In addition, from the definition, the lower limit is less than 0.340 nm, preferably 0.337 nm or less. If the d value is too large, the crystallinity may decrease, which may increase the initial irreversible capacity. On the other hand, 0.335 is the theoretical value of graphite. In addition, the crystallite size (Lc) of the carbon material obtained by X-ray diffraction using the Gakushin method is usually in the range of 30 nm or more, preferably 50 nm or more, and more preferably 100 nm or more. If it is less than this range, the crystallinity may decrease and the initial irreversible capacity may increase.
(2)灰分(2) Ash content
石墨类碳质物质中所含的灰分相对于石墨类碳质物质的总质量优选为1质量%以下,更优选0.5质量%以下,特别优选0.1质量%以下,下限为1ppm以上。如果超过上述范围,则充放电时与电解液的反应而引起的电池性能的劣化不能忽视。如果低于该范围,则在制造上需要较长时间和能量以及用于防止污染的设备,有时成本会上升。The ash contained in the graphite-like carbonaceous material is preferably 1% by mass or less, more preferably 0.5% by mass or less, particularly preferably 0.1% by mass or less, with a lower limit of 1 ppm or more based on the total mass of the graphite-like carbonaceous material. If the above-mentioned range is exceeded, the degradation of battery performance due to the reaction with the electrolytic solution during charging and discharging cannot be ignored. If it is less than this range, long time and energy are required for manufacturing and equipment for preventing pollution, and the cost may increase.
(3)体积基准平均粒径(3) Volume-based average particle size
石墨类碳质物质的体积基准平均粒径是通过激光衍射/散射法求得的体积基准的平均粒径(中值粒径),通常为1μm以上,优选为3μm以上,更优选为5μm以上,进一步优选为7μm以上。另外,其上限通常为100μm以下,优选为50μm以下,更优选为40μm以下,进一步优选为30μm以下,特别优选为25μm以下。如果低于上述范围,则不可逆容量增大,有时导致初期电池容量的损失。另外,如果超过上述范围,则在制成电极极板时,容易形成不均匀的涂布面,有时在电池制作工序上是不优选的。The volume-based average particle size of the graphitic carbonaceous material is the volume-based average particle size (median particle size) obtained by the laser diffraction/scattering method, and is usually more than 1 μm, preferably more than 3 μm, more preferably more than 5 μm, More preferably, it is 7 μm or more. In addition, the upper limit thereof is usually 100 μm or less, preferably 50 μm or less, more preferably 40 μm or less, still more preferably 30 μm or less, particularly preferably 25 μm or less. If it is less than the above-mentioned range, the irreversible capacity may increase, resulting in loss of initial battery capacity. Moreover, if it exceeds the said range, it will become easy to form an uneven coating surface at the time of making an electrode pad, and it may be unpreferable in a battery manufacturing process.
在本发明中,体积基准平均粒径用中值粒径来定义,所述中值粒径通过以下方法测定:使碳粉末分散在作为表面活性剂的聚氧乙烯(20)山梨糖醇酐单月桂酸酯的0.2质量%水溶液(约1mL)中,使用激光衍射/散射式粒度分布计(例如,堀场制作所公司制造的LA-700)来测定。In the present invention, the volume-based average particle diameter is defined by a median particle diameter measured by dispersing carbon powder in polyoxyethylene (20) sorbitan as a surfactant. In a 0.2% by mass aqueous solution (about 1 mL) of monolaurate, it measures using a laser diffraction/scattering particle size distribution meter (for example, LA-700 manufactured by Horiba, Ltd.).
(4)拉曼R值、拉曼半值宽度(4) Raman R value, Raman half value width
使用氩离子激光拉曼光谱法测定的石墨类碳质物质的R值通常为0.01以上,优选为0.03以上,更优选为0.10以上,其上限通常为0.60以下,优选为0.40以下的范围。如果R值低于该范围,则粒子表面的结晶性过高,随着充放电Li进入层间的位点有时变少。即,充电接受性有时降低。另一方面,如果超过该范围,则粒子表面的结晶性降低,与电解液的反应性增加,有时导致效率降低或产生的气体增加。The R value of the graphitic carbonaceous material measured by argon ion laser Raman spectroscopy is usually 0.01 or more, preferably 0.03 or more, more preferably 0.10 or more, and its upper limit is usually 0.60 or less, preferably 0.40 or less. If the R value is lower than this range, the crystallinity of the particle surface is too high, and there may be fewer sites for Li to enter the interlayer due to charge and discharge. That is, charge acceptance may be lowered. On the other hand, if it exceeds this range, the crystallinity of the particle surface decreases, and the reactivity with the electrolyte solution increases, resulting in a decrease in efficiency or an increase in gas generation.
另外,石墨类碳质物质在1580cm-1附近的拉曼半值宽度没有特别的限制,通常为10cm-1以上,优选15cm-1以上,上限通常为60cm-1以下,优选为45cm-1以下,更优选为40cm-1以下的范围。如果拉曼半值宽度低于该范围,则粒子表面的结晶性过高,随着充放电Li进入层间的位点有时变少。即,充电接受性有时降低。另一方面,如果高于该范围,则粒子表面的结晶性降低,与电解液的反应性增加,有时导致效率降低或产生的气体增加。In addition, the Raman half-value width of graphitic carbonaceous material around 1580 cm -1 is not particularly limited, but it is usually 10 cm -1 or more, preferably 15 cm -1 or more, and the upper limit is usually 60 cm -1 or less, preferably 45 cm -1 or less , more preferably in the range of 40 cm -1 or less. If the Raman half-value width is below this range, the crystallinity of the particle surface may be too high, and there may be fewer sites for Li to enter the interlayer due to charge and discharge. That is, charge acceptance may be lowered. On the other hand, if it exceeds this range, the crystallinity of the particle surface will decrease, and the reactivity with the electrolytic solution will increase, which may result in a decrease in efficiency or an increase in generated gas.
拉曼光谱的测定如下进行:使用拉曼分光器(例如日本分光社制造的拉曼分光器),使试样自然落下并填充在测定池内,对池内的试样表面照射氩离子激光,同时,使池在与激光垂直的面内旋转。对于得到的拉曼光谱,测定1580cm-1附近的峰PA的强度IA和1360cm-1附近的峰PB的强度IB,计算其强度比R(R=IB/IA),将其定义为碳材料的拉曼R值。另外,测定得到的拉曼光谱在1580cm-1附近的峰PA的半值宽度,将其定义为碳材料的拉曼半值宽度。The measurement of the Raman spectrum is carried out as follows: Using a Raman spectrometer (such as a Raman spectrometer manufactured by JASCO Corporation), the sample is naturally dropped and filled in the measurement cell, and the surface of the sample in the cell is irradiated with an argon ion laser. At the same time, Rotate the cell in a plane perpendicular to the laser. For the obtained Raman spectrum, the intensity I A of the peak PA near 1580 cm -1 and the intensity I B of the peak P B near 1360 cm -1 were measured , and the intensity ratio R (R= IB / IA ) was calculated. It is defined as the Raman R value of the carbon material. In addition, the half-value width of the peak PA in the vicinity of 1580 cm −1 in the obtained Raman spectrum was measured, and this was defined as the Raman half-value width of the carbon material.
另外,这里的拉曼测定条件如下。In addition, the Raman measurement conditions here are as follows.
·氩离子激光波长:514.5nmArgon ion laser wavelength: 514.5nm
·试样上的激光功率:15~25mW·Laser power on the sample: 15~25mW
·分辨率:10~20cm-1 ·Resolution: 10~20cm -1
·测定范围:1100cm-1~1730cm-1 · Measuring range: 1100cm -1 ~ 1730cm -1
·R值、半值宽度分析:本底处理R value, half-value width analysis: background processing
·光滑处理:单纯平均、卷积5点·Smooth processing: simple average, convolution 5 points
(5)BET比表面积(5) BET specific surface area
使用BET法测定的本发明的石墨类碳质物质的比表面积通常为0.1m2/g以上,优选为0.7m2/g以上,更优选为1m2/g以上,进一步优选为1.5m2/g以上。其上限通常为100m2/g以下,优选为25m2/g以下,更优选为15m2/g以下,进一步优选为10m2/g以下。如果比表面积的值低于上述范围,则作为负极使用的情况下,在充电时,锂的接受性变差,锂容易在电极表面析出。另一方面,如果超过上述范围,则在作为负极材料使用时,与电解液的反应性增加,产生的气体变多,有时难以得到优选的电池。The specific surface area of the graphitic carbonaceous material of the present invention measured by the BET method is usually 0.1 m 2 /g or more, preferably 0.7 m 2 /g or more, more preferably 1 m 2 /g or more, still more preferably 1.5 m 2 /g or more. more than g. The upper limit thereof is usually 100 m 2 /g or less, preferably 25 m 2 /g or less, more preferably 15 m 2 /g or less, still more preferably 10 m 2 /g or less. If the value of the specific surface area is less than the above range, when used as a negative electrode, the acceptability of lithium will deteriorate during charging, and lithium will be easily deposited on the surface of the electrode. On the other hand, when it exceeds the above-mentioned range, when used as a negative electrode material, the reactivity with the electrolytic solution increases and gas generation increases, making it difficult to obtain a preferable battery in some cases.
BET比表面积定义为如下测定的值:使用表面积计(例如,大仓理研制造的全自动表面积测定装置),在氮气流通、350℃下对试样进行15分钟预干燥,然后,使用氮气相对于大气压的相对压力值准确调整为0.3的氮氦混合气体,通过采用气体流动法的氮吸附BET1点法测定。The BET specific surface area is defined as a value measured as follows: Using a surface area meter (for example, a fully automatic surface area measuring device manufactured by Okura Riken), the sample is pre-dried at 350° C. for 15 minutes under nitrogen flow, and then, using nitrogen gas relative to The relative pressure value of the atmospheric pressure is accurately adjusted to 0.3 nitrogen-helium mixed gas, and it is measured by the nitrogen adsorption BET 1-point method using the gas flow method.
(6)微孔分布(6) Micropore distribution
作为石墨类碳质物质,通过水银孔隙率计(水银压入法)求得的相当于直径为0.01μm~1μm的粒子内的空隙、由于粒子表面的高低不平造成的凹凸的量通常为0.01mL/g以上,优选为0.05mL/g以上,更优选为0.1mL/g以上,上限通常为0.6mL/g以下,优选为0.4mL/g以下,更优选为0.3mL/g以下的范围。如果超过该范围,则制造成极板时需要大量的粘合剂。另一方面,如果低于该范围,则高电流密度充放电特性降低,并且,有时不能得到充放电时的电极膨胀收缩的缓和效果。As a graphitic carbonaceous material, the amount corresponding to the voids in the particles with a diameter of 0.01 μm to 1 μm and the unevenness of the particle surface obtained by mercury porosimetry (mercury porosimetry) is usually 0.01 mL /g or more, preferably 0.05mL/g or more, more preferably 0.1mL/g or more, the upper limit is usually 0.6mL/g or less, preferably 0.4mL/g or less, more preferably 0.3mL/g or less. If it exceeds this range, a large amount of binder will be required to manufacture an electrode plate. On the other hand, if it is less than this range, the high current density charge-discharge characteristics deteriorate, and the effect of alleviating electrode expansion and contraction during charge-discharge may not be obtained.
另外,总微孔容积优选为0.1mL/g以上,更优选为0.25mL/g以上,其上限通常为10mL/g以下,优选为5mL/g以下,更优选为2mL/g以下的范围。如果超过该范围,则在制成基板时有时需要大量的粘合剂。如果低于该范围,则在制成极板时有时不能得到增稠剂或粘结剂的分散效果。In addition, the total pore volume is preferably 0.1 mL/g or more, more preferably 0.25 mL/g or more, and its upper limit is usually 10 mL/g or less, preferably 5 mL/g or less, more preferably 2 mL/g or less. If it exceeds this range, a large amount of adhesive may be required to form a substrate. If it is less than this range, the dispersion effect of the thickener or the binder may not be obtained when forming an electrode plate.
另外,平均微孔径优选为0.05μm以上,更优选为0.1μm以上,进一步优选为0.5μm以上,其上限通常为50μm以下,优选为20μm以下,更优选为10μm以下的范围。如果超过该范围,则有时需要大量的粘合剂。另一方面,如果低于该范围,则有时高电流密度充放电特性降低。In addition, the average pore diameter is preferably 0.05 μm or more, more preferably 0.1 μm or more, even more preferably 0.5 μm or more, and the upper limit thereof is usually 50 μm or less, preferably 20 μm or less, more preferably 10 μm or less. If this range is exceeded, a large amount of adhesive is sometimes required. On the other hand, if it is less than this range, the high current density charge-discharge characteristics may deteriorate.
作为用于水银孔隙率计的装置,可以使用水银孔隙率计(autopore9520;micrometritics公司制造)。称量约0.2g试样(负极材料),密封入粉末用容器中,在室温、真空下(50μmHg以下)进行10分钟脱气,实施前处理。接着,减压至4psia(约28kPa),导入水银,从4psia(约28kPa)阶段状升压至40000psia(约280MPa),然后降压至25psia(约170kPa)。升压时的阶段数为80阶段以上,在各阶段中,在10秒的平衡时间后,测定水银压入量。使用Washburn式从这样得到的水银压入曲线计算出微孔分布。另外,水银的表面张力(γ)为485dyne/cm,接触角(φ)为140°。平均微孔径使用累积微孔体积达到50%时的微孔径。As an apparatus used for the mercury porosimeter, a mercury porosimeter (autopore9520; manufactured by Micrometrics Inc.) can be used. About 0.2 g of the sample (negative electrode material) was weighed, sealed in a powder container, and degassed at room temperature under vacuum (50 μmHg or less) for 10 minutes to perform pretreatment. Next, the pressure is reduced to 4psia (about 28kPa), mercury is introduced, the pressure is raised from 4psia (about 28kPa) to 40000psia (about 280MPa) in steps, and then the pressure is lowered to 25psia (about 170kPa). The number of stages at the time of pressurization was 80 or more, and the amount of mercury intrusion was measured after a 10-second equilibration time in each stage. The pore distribution was calculated from the mercury intrusion curve thus obtained using the Washburn formula. In addition, the surface tension (γ) of mercury is 485 dyne/cm, and the contact angle (φ) is 140°. As the average pore diameter, the pore diameter at which the cumulative pore volume reaches 50% is used.
(7)圆形度(7) Circularity
使用圆形度作为石墨类碳质物质的球形的程度,石墨类碳质物质的粒径为3~40μm范围的粒子的圆形度优选为0.1以上,特别优选为0.5以上,更优选为0.8以上,进一步优选为0.85以上,最优选为0.9以上。圆形度大时,高电流密度充放电特性提高,因此优选。圆形度通过下式定义,圆形度为1时,成为理论真球。Using the degree of circularity as the degree of spherical shape of the graphite-like carbonaceous material, the circularity of the particles of the graphite-like carbonaceous material having a particle size in the range of 3 to 40 μm is preferably 0.1 or more, particularly preferably 0.5 or more, more preferably 0.8 or more , more preferably 0.85 or more, most preferably 0.9 or more. When the circularity is large, the high current density charge and discharge characteristics are improved, which is preferable. The circularity is defined by the following formula, and when the circularity is 1, it becomes a theoretical true sphere.
圆形度=(具有与粒子投影形状相同面积的等效圆的周长)/(粒子投影形状的实际周长)Circularity = (perimeter of an equivalent circle having the same area as the particle projected shape)/(actual perimeter of the particle projected shape)
作为圆形度的值使用如下测定的值:使用例如流动式粒子图像分析装置(例如,Sysmex Industrial公司制造的FPIA),将约0.2g试样分散于作为表面活性剂的聚氧乙烯(20)山梨糖醇酐单月桂酸酯的0.2质量%水溶液(约50mL)中,以60W的输出功率照射1分钟28kHz的超声波后,指定0.6~400μm为检测范围,对粒径3~40μm范围的粒子进行测定。As the value of circularity, the value measured as follows is used: using, for example, a flow type particle image analyzer (for example, FPIA manufactured by Sysmex Industrial Co., Ltd.), about 0.2 g of a sample is dispersed in polyoxyethylene (20) as a surfactant. In a 0.2% by mass aqueous solution of sorbitan monolaurate (approximately 50mL), after irradiating 28kHz ultrasonic waves with an output power of 60W for 1 minute, specify a detection range of 0.6 to 400μm, and perform a test on particles in the range of particle diameters from 3 to 40μm. Determination.
提高圆形度的方法没有特别限制,但通过实施球形化处理使之成为球形可以使制成电极体时粒子间空隙的形状整齐统一,故优选。作为球形化处理的例子,可以举出,通过施加剪切力、压缩力来机械地接近球形的方法;通过粘合剂或粒子自身具有的附着力将多个微粒造粒的机械/物理处理方法等。There is no particular limitation on the method of improving the circularity, but it is preferable to make the shape of the gap between the particles uniform when the electrode body is produced by performing a spheroidization treatment to make it spherical. Examples of spheroidization treatment include a method of mechanically approaching a spherical shape by applying a shearing force or a compressive force; a mechanical/physical treatment method of granulating a plurality of fine particles by means of a binder or the adhesive force of the particles themselves wait.
(8)真密度(8) True density
石墨类碳质物质的真密度通常为2g/cm3以上,优选为2.1g/cm3以上,更优选为2.2g/cm3以上,进一步优选为2.22g/cm3以上,其上限为2.26g/cm3以下。上限为石墨的理论值。如果低于该范围,则碳的结晶性过低,初期不可逆容量有时会增大。在本发明中,真密度定义为通过使用丁醇的液相置换法(比重计法)测定的值。The true density of the graphite-like carbonaceous material is usually above 2 g/cm 3 , preferably above 2.1 g/cm 3 , more preferably above 2.2 g/cm 3 , and even more preferably above 2.22 g/cm 3 , with an upper limit of 2.26 g /cm 3 or less. The upper limit is the theoretical value of graphite. If it is less than this range, the crystallinity of carbon may be too low, and the initial irreversible capacity may increase. In the present invention, the true density is defined as a value measured by a liquid phase displacement method (pycnometer method) using butanol.
(9)振实密度(9) Tap density
石墨类碳质物质的振实密度通常为0.1g/cm3以上,优选为0.5g/cm3以上,更优选为0.7g/cm3以上,特别优选为0.9g/cm3以上。另外,其上限优选为2g/cm3以下,更优选为1.8g/cm3以下,特别优选为1.6g/cm3以下。振实密度如果低于该范围,则在作为负极使用时,填充密度很难提高,有时不能得到高容量的电池。另一方面,如果高于该范围,则电极中的粒子间的空隙过少,难以确保粒子间的导电性,有时难以得到优选的电池特性。The tap density of the graphitic carbonaceous material is usually 0.1 g/cm 3 or higher, preferably 0.5 g/cm 3 or higher, more preferably 0.7 g/cm 3 or higher, particularly preferably 0.9 g/cm 3 or higher. In addition, the upper limit thereof is preferably 2 g/cm 3 or less, more preferably 1.8 g/cm 3 or less, particularly preferably 1.6 g/cm 3 or less. If the tap density is lower than this range, it is difficult to increase the packing density when used as a negative electrode, and a high-capacity battery may not be obtained. On the other hand, if it exceeds this range, the gaps between the particles in the electrode will be too small, making it difficult to ensure the conductivity between the particles, and it may be difficult to obtain preferable battery characteristics.
在本发明中,振实密度如下定义:使试样通过孔径300μm的筛子落在20cm3的振实容器内,直到试样充满容器的上端面后,使用粉末密度测定器(例如,seishin企业公司制造的Tap densor),进行1000次冲程长度10mm的振动,将此时的容积密度定义为振实密度。In the present invention, the tap density is defined as follows: the sample is passed through a sieve with an aperture of 300 μm and dropped in a 20 cm container for vibration until the sample is filled with the upper end surface of the container, and a powder density measuring device (for example, Seishin Enterprise Co., Ltd. manufactured Tap densor), vibrate 1000 times with a stroke length of 10mm, and define the bulk density at this time as the tap density.
(10)取向比(粉末)(10) Orientation ratio (powder)
石墨类碳质物质的取向比通常为0.005以上,优选为0.01以上,更优选为0.015以上,上限在理论上为0.67以下的范围。如果低于该范围,则有时高密度充放电特性降低。The orientation ratio of the graphitic carbonaceous material is usually 0.005 or more, preferably 0.01 or more, more preferably 0.015 or more, and the upper limit is theoretically 0.67 or less. If it is less than this range, high-density charge-discharge characteristics may deteriorate.
取向比通过X射线衍射测定。使用非对称皮尔森VII作为分布函数,拟合通过X射线衍射得到的碳的(110)衍射和(004)衍射的峰,进行峰分离,分别计算出(110)衍射和(004)衍射的峰的积分强度。由得到的积分强度计算出由(110)衍射积分强度/(004)衍射积分强度表示的比值,将该比值定义为活性物质取向比。The orientation ratio was measured by X-ray diffraction. Using asymmetric Pearson VII as a distribution function, fit the peaks of (110) diffraction and (004) diffraction of carbon obtained by X-ray diffraction, perform peak separation, and calculate the peaks of (110) diffraction and (004) diffraction respectively integral strength. A ratio represented by (110) diffraction integrated intensity/(004) diffraction integrated intensity was calculated from the obtained integrated intensity, and this ratio was defined as the active material orientation ratio.
这里的X射线衍射测定条件如下。另外,“2θ”表示衍射角。The X-ray diffraction measurement conditions here are as follows. In addition, "2θ" represents a diffraction angle.
·靶:Cu(Kα射线)石墨单色光度仪Target: Cu (Kα ray) graphite monochromator
·狭缝:·Slit:
发散狭缝=1度,受光狭缝=0.1mm,散射狭缝=1度Divergence slit = 1 degree, light receiving slit = 0.1mm, scattering slit = 1 degree
·测定范围和步进角/测定时间・Measurement range and step angle/measurement time
(110)面:76.5度≤2θ≤78.5度0.01度/3秒(110) surface: 76.5 degrees ≤ 2θ ≤ 78.5 degrees 0.01 degrees / 3 seconds
(004)面:53.5度≤2θ≤56.0度0.01度/3秒(004) plane: 53.5 degrees ≤ 2θ ≤ 56.0 degrees 0.01 degrees / 3 seconds
(11)长径比(粉末)(11) aspect ratio (powder)
石墨类碳质物质的长径比理论上为1以上,上限通常为10以下,优选为8以下,更优选为5以下。如果超过其上限,制成极板时会产生条纹、无法得到均匀的涂布面,高电流密度充放电特性有时降低。The aspect ratio of the graphitic carbonaceous material is theoretically 1 or more, and the upper limit is usually 10 or less, preferably 8 or less, more preferably 5 or less. If the upper limit is exceeded, streaks may be generated when forming an electrode plate, a uniform coating surface may not be obtained, and the high current density charge and discharge characteristics may deteriorate.
另外,长径比用三维观察时碳材料粒子的最长径A和与其垂直的最短径B的比值A/B来表示。碳粒子的观察通过可以放大观察的扫描型电子显微镜来进行。选择固定在厚度50μm以下的金属的端面上的任意50个石墨粒子,旋转、倾斜固定有试样的载物台,分别测定这些粒子的A、B,求出A/B的平均值。In addition, the aspect ratio is represented by the ratio A/B of the longest diameter A of the carbon material particle and the shortest diameter B perpendicular thereto when viewed three-dimensionally. Observation of carbon particles was performed with a scanning electron microscope capable of magnified observation. Select any 50 graphite particles fixed on the end face of the metal with a thickness of 50 μm or less, rotate and tilt the stage on which the sample is fixed, measure A and B of these particles, and calculate the average value of A/B.
[[[[低结晶性碳质物质]]]][[[[Low crystalline carbonaceous substances]]]]
低结晶性碳质物质是指通过X射线广角衍射法测定的(002)面的面间距(d002)为0.340nm以上的结晶性低的碳质物质。The low-crystalline carbonaceous substance refers to a carbonaceous substance with low crystallinity whose interplanar distance (d002) of the (002) plane measured by X-ray wide-angle diffractometry is 0.340 nm or more.
(低结晶性碳质物质的组成)(Composition of low crystalline carbonaceous substances)
“与粒子状碳质物质结晶不同的碳质物质”优选比粒子状碳质物质的结晶性低的低结晶性碳质物质。另外,特别优选以下的(d)或(e)的碳化物。The "carbonaceous substance different from the crystals of the particulate carbonaceous substance" is preferably a low-crystalline carbonaceous substance having lower crystallinity than the particulate carbonaceous substance. In addition, carbides of the following (d) or (e) are particularly preferred.
(d)选自煤类重油、直馏类重油、分解类石油重油、芳香族烃、N环化合物、S环化合物、聚苯、有机合成高分子、天然高分子、热塑性树脂和热固化树脂中的可碳化的有机物;(d) selected from coal-based heavy oil, straight-run heavy oil, decomposed petroleum-based heavy oil, aromatic hydrocarbons, N-ring compounds, S-ring compounds, polyphenylene, organic synthetic polymers, natural polymers, thermoplastic resins, and thermosetting resins carbonizable organic matter;
(e)将这些可碳化的有机物溶解于低分子有机溶剂中而得到的溶液。(e) A solution obtained by dissolving these carbonizable organic substances in a low-molecular organic solvent.
作为煤类重油,优选从软沥青到硬沥青的煤焦油沥青或者干馏液化油等;作为直馏类重油,优选常压残油、减压残油等;作为分解类石油重油,优选原油、石脑油等热分解时副产的乙烯焦油等;作为芳香族烃,优选苊烯、十环烯、蒽、菲等;作为N环化合物,优选吩嗪、吖啶等;作为S环化合物,优选噻吩、联二噻吩等;作为聚苯,优选联苯、三联苯等;作为有机高分子,优选聚氯乙烯、聚乙烯醇、聚乙烯醇缩丁醛、它们的不溶解处理物、聚丙烯腈、聚吡咯、聚噻吩、聚苯乙烯等;作为天然高分子,优选纤维素、木质素、甘露聚糖、聚半乳糖醛酸、壳聚糖、蔗糖等多糖类等;作为热塑性树脂,优选聚苯硫醚、聚苯醚等;作为热固性树脂,优选糠醇树脂、酚醛树脂、酰亚胺树脂等。As coal heavy oil, coal tar pitch from soft asphalt to hard asphalt or dry distillation liquefied oil, etc. are preferred; as straight run heavy oil, atmospheric residual oil, vacuum residual oil, etc. are preferred; as decomposed petroleum heavy oil, crude oil, petroleum Ethylene tar, etc., which are by-produced during thermal decomposition of naphtha, etc.; as aromatic hydrocarbons, preferably acenaphthylene, decacyclene, anthracene, phenanthrene, etc.; as N-ring compounds, preferably phenazine, acridine, etc.; as S-ring compounds, preferably Thiophene, bithiophene, etc.; as polyphenylene, biphenyl, terphenyl, etc. are preferred; as organic polymers, polyvinyl chloride, polyvinyl alcohol, polyvinyl butyral, their insoluble processed products, polyacrylonitrile , polypyrrole, polythiophene, polystyrene, etc.; as natural polymers, preferably polysaccharides such as cellulose, lignin, mannan, polygalacturonic acid, chitosan, sucrose, etc.; as thermoplastic resins, preferably Polyphenylene sulfide, polyphenylene ether, etc.; as the thermosetting resin, furfuryl alcohol resin, phenolic resin, imide resin, etc. are preferable.
“与粒子状碳质物质结晶性不同的碳质物质”优选上述“可碳化的有机物”的碳化物,另外,还优选将这些“可碳化的有机物”溶解在苯、甲苯、二甲苯、喹啉、正己烷等低分子有机溶剂中而得到溶液等的碳化物。另外,“与粒子状碳质物质结晶性不同的碳质物质”优选煤类焦炭、石油类焦炭的碳化物。"Carbonaceous substances different in crystallinity from particulate carbonaceous substances" are preferably carbides of the above-mentioned "carbonizable organic substances", and it is also preferable to dissolve these "carbonizable organic substances" in benzene, toluene, xylene, quinoline, etc. , n-hexane and other low-molecular organic solvents to obtain carbides such as solutions. In addition, the "carbonaceous substance different in crystallinity from the particulate carbonaceous substance" is preferably a carbide of coal-based coke or petroleum-based coke.
作为上述(d)或(e),特别优选液态。即,从与石墨类碳质物质部分生成界面的观点来看,优选在液相中进行碳化。As the above (d) or (e), a liquid state is particularly preferable. That is, from the viewpoint of partially forming an interface with the graphitic carbonaceous material, carbonization is preferably performed in the liquid phase.
(低结晶性碳质物质的物性)(Physical properties of low crystalline carbonaceous material)
作为低结晶性碳质物质的物性优选同时满足下示(1)~(5)中任何一项或多项。另外,可以单独使用一种显示这些物性的低结晶性碳质物质,也可以以任意组合和比例组合使用2种以上。The physical properties as the low-crystalline carbonaceous substance preferably satisfy any one or more of the following (1) to (5) at the same time. In addition, one kind of low-crystalline carbonaceous material exhibiting these physical properties may be used alone, or two or more kinds may be used in combination in any combination and ratio.
(1)X射线参数(1) X-ray parameters
对于低结晶性碳质物质部分,通过采用学振法进行的X射线衍射求得的晶格面(002面)的d值(层间距离)从定义上来讲为0.340nm以上,优选为0.340nm以上,特别优选为0.341nm以上。另外,其上限为0.380nm以下,特别优选为0.355nm以下,进一步优选为0.350以下。如果d值过大,则形成结晶性明显低的表面,有时会增加不可逆容量,而如果d值过小,则在表面设置低结晶性碳质物质而得到的充电接受性提高的效果变小,从而本发明的效果变小。另外,通过采用学振法的X射线衍射求得的碳材料的微晶大小(Lc)通常为1nm以上,优选为1.5nm以上的范围。如果低于该范围,则结晶性降低,有时会增加初期不可逆容量的增大。For the low-crystalline carbonaceous material part, the d value (interlayer distance) of the lattice plane (002 plane) obtained by X-ray diffraction by the Gakushin method is by definition 0.340 nm or more, preferably 0.340 nm or more, particularly preferably 0.341 nm or more. In addition, the upper limit thereof is 0.380 nm or less, particularly preferably 0.355 nm or less, further preferably 0.350 nm or less. If the d value is too large, a surface with significantly low crystallinity is formed, which may increase the irreversible capacity, and if the d value is too small, the effect of improving charge acceptance by providing a low-crystalline carbonaceous material on the surface becomes small, Thus, the effect of the present invention becomes small. In addition, the crystallite size (Lc) of the carbon material obtained by X-ray diffraction using the Gakushin method is usually 1 nm or more, preferably 1.5 nm or more. If it is less than this range, the crystallinity is lowered, and the increase of the initial irreversible capacity may be increased.
(2)灰分(2) Ash content
低结晶性碳质物质部分所含的灰分相对于复合碳质物质的总质量优选为1质量%以下,更优选为0.5质量%以下,特别优选为0.1质量%以下,作为下限,优选为1ppm以上。如果超过上述范围,则充放电时与电解液的反应而引起的电池性能的劣化不能忽视。而如果低于该范围,则在制造上需要较长时间和能量以及用于防止污染的设备,有时成本会上升。The ash contained in the low-crystalline carbonaceous material part is preferably 1% by mass or less, more preferably 0.5% by mass or less, and particularly preferably 0.1% by mass or less with respect to the total mass of the composite carbonaceous material. The lower limit is preferably 1 ppm or more . If the above-mentioned range is exceeded, the degradation of battery performance due to the reaction with the electrolytic solution during charging and discharging cannot be ignored. On the other hand, if it is less than this range, long time and energy are required for manufacturing and equipment for preventing pollution, and the cost may increase.
(3)拉曼R值、拉曼半值宽度(3) Raman R value, Raman half value width
使用氩离子激光拉曼光谱法测定的低结晶性碳质物质部分的R值通常为0.5以上,优选为0.7以上,更优选为0.9以上,其上限通常为1.5以下,优选为1.2以下的范围。如果R值低于该范围,则粒子表面的结晶性过高,随着充放电Li进入层间的位点有时变少。即,充电接受性有时降低。另外,涂布在集电体上之后,通过压制使负极高密度化时,结晶容易取向为与电极板平行的方向,有时导致负荷特性降低。另一方面,如果超过该范围,则粒子表面的结晶性降低,与电解液的反应性增加,有时导致效率降低或产生的气体增加。The R value of the low crystalline carbonaceous material portion measured by argon ion laser Raman spectroscopy is usually 0.5 or more, preferably 0.7 or more, more preferably 0.9 or more, and its upper limit is usually 1.5 or less, preferably 1.2 or less. If the R value is lower than this range, the crystallinity of the particle surface is too high, and there may be fewer sites for Li to enter the interlayer due to charge and discharge. That is, charge acceptance may be lowered. In addition, when the anode is densified by pressing after coating on the current collector, the crystals tend to be oriented in a direction parallel to the electrode plate, which may result in a decrease in load characteristics. On the other hand, if it exceeds this range, the crystallinity of the particle surface decreases, and the reactivity with the electrolyte solution increases, resulting in a decrease in efficiency or an increase in gas generation.
另外,低结晶性碳质物质部分在1580cm-1附近的拉曼半值宽度没有特别的限制,通常为40cm-1以上,优选为50cm-1以上,其上限通常为100cm-1以下,优选为90cm-1以下,更优选为80cm-1以下的范围。拉曼半值宽度如果低于该范围,则粒子表面的结晶性过高,随着充放电Li进入层间的位点有时变少。即,充电接受性有时降低。另外,涂布在集电体上之后,通过压制使负极高密度化时,结晶容易取向为与电极板平行的方向,有时导致负荷特性降低。另一方面,如果超过该范围,则粒子表面的结晶性降低,与电解液的反应性增加,有时导致效率降低或产生的气体增加。In addition, the Raman half-value width of the low-crystalline carbonaceous material part is not particularly limited in the vicinity of 1580 cm -1 , but it is usually 40 cm -1 or more, preferably 50 cm -1 or more, and its upper limit is usually 100 cm-1 or less, preferably 100 cm -1 or less. 90 cm -1 or less, more preferably 80 cm -1 or less. If the Raman half-value width is lower than this range, the crystallinity of the particle surface is too high, and there may be fewer sites for Li to enter the interlayer due to charge and discharge. That is, charge acceptance may be lowered. In addition, when the anode is densified by pressing after coating on the current collector, the crystals tend to be oriented in a direction parallel to the electrode plate, which may result in a decrease in load characteristics. On the other hand, if it exceeds this range, the crystallinity of the particle surface decreases, and the reactivity with the electrolyte solution increases, resulting in a decrease in efficiency or an increase in gas generation.
(4)真密度(4) True density
低结晶性碳质物质部分的真密度通常为1.4g/cm3以上,优选为1.5g/cm3以上,更优选为1.6g/cm3以上,进一步优选为1.7g/cm3以上,其上限通常为2.1g/cm3以下,优选为2g/cm3以下。如果超过该范围,则充电接受性可能受损。如果低于该范围,则碳的结晶性过低,初期不可逆容量有时会增大。The true density of the low-crystalline carbonaceous material part is usually 1.4 g/cm 3 or more, preferably 1.5 g/cm 3 or more, more preferably 1.6 g/cm 3 or more, and even more preferably 1.7 g/cm 3 or more, the upper limit of which is Usually 2.1 g/cm 3 or less, preferably 2 g/cm 3 or less. If this range is exceeded, charge acceptance may be impaired. If it is less than this range, the crystallinity of carbon may be too low, and the initial irreversible capacity may increase.
(5)取向比(粉末)(5) Orientation ratio (powder)
低结晶性碳质物质部分的取向比通常为0.005以上,优选为0.01以上,更优选为0.015以上,其上限为理论值0.67以下。如果低于该范围,则高密度充放电特性有时降低,不优选。取向比通过X射线衍射测定。使用非对称皮尔森VII作为分布函数,拟合碳通过X射线衍射得到的(110)衍射和(004)衍射的峰,进行峰分离,分别计算出(110)衍射和(004)衍射的峰的积分强度。由得到的积分强度计算出由(110)衍射积分强度/(004)衍射积分强度表示的比值,将该比值定义为活性物质取向比。The orientation ratio of the low-crystalline carbonaceous material portion is usually not less than 0.005, preferably not less than 0.01, more preferably not less than 0.015, and the upper limit thereof is not more than a theoretical value of 0.67. If it is less than this range, the high-density charge-discharge characteristics may deteriorate, which is not preferable. The orientation ratio was measured by X-ray diffraction. Use asymmetric Pearson VII as the distribution function, fit the peaks of (110) diffraction and (004) diffraction obtained by carbon through X-ray diffraction, carry out peak separation, and calculate the peaks of (110) diffraction and (004) diffraction respectively Integral strength. A ratio represented by (110) diffraction integrated intensity/(004) diffraction integrated intensity was calculated from the obtained integrated intensity, and this ratio was defined as the active material orientation ratio.
[[[复合碳质物质]]][[[Composite carbonaceous material]]]
本发明的锂二次电池中的负极[1]中使用的复合碳质物质优选含有“粒子状碳质物质”和“与该粒子状碳质物质结晶性不同的碳质物质”,此时,只要其中任何一个是石墨类碳质物质,而另一个为低结晶性碳质物质即可。另外,优选“粒子状碳质物质”为石墨类碳质物质,“与该粒子状碳质物质结晶性不同的碳质物质”为低结晶性碳质物质。The composite carbonaceous material used in the negative electrode [1] in the lithium secondary battery of the present invention preferably contains "particulate carbonaceous material" and "a carbonaceous material different in crystallinity from the particulate carbonaceous material". In this case, It is only necessary that any one of them is a graphitic carbonaceous substance and the other is a low-crystalline carbonaceous substance. In addition, it is preferable that the "particulate carbonaceous substance" is a graphitic carbonaceous substance, and the "carbonaceous substance different in crystallinity from the particulate carbonaceous substance" is a low-crystalline carbonaceous substance.
在复合碳质物质中,石墨类碳质物质和低结晶性碳质物质的质量比优选为50/50以上,更优选为80/20以上,特别优选为90/10以上,且优选为99.9/0.1以下,更优选为99/1以下,特别优选为98/2以下的范围。如果超过上述范围,则有时不能得到具有2种结晶性的碳质物质的效果,如果低于上述范围,初期不可逆容量表现出增加的倾向,有时在电池设计上存在问题。石墨类碳质物质相对于全部复合碳质物质为50质量%以上。In the composite carbonaceous material, the mass ratio of the graphite-like carbonaceous material and the low-crystalline carbonaceous material is preferably 50/50 or more, more preferably 80/20 or more, particularly preferably 90/10 or more, and preferably 99.9/20 or more. 0.1 or less, more preferably 99/1 or less, particularly preferably 98/2 or less. If it exceeds the above range, the effect of having two kinds of crystalline carbonaceous substances may not be obtained, and if it is below the above range, the initial irreversible capacity tends to increase, which may cause problems in battery design. The graphite-based carbonaceous substance is 50 mass % or more with respect to the whole composite carbonaceous substance.
(复合碳质物质的物性)(Physical properties of composite carbonaceous material)
作为复合碳质物质,优选同时满足下面所示的(1)~(11)中任何一项或多项。另外,可以单独使用一种显示这些物性的复合碳质物质,也可以以任意组合和比例组合使用2种以上。As the composite carbonaceous substance, it is preferable to satisfy any one or more of the following (1) to (11) at the same time. In addition, one kind of composite carbonaceous material exhibiting these physical properties may be used alone, or two or more kinds may be used in combination in any combination and ratio.
(1)X射线参数(1) X-ray parameters
复合碳质物质通过采用学振法的X射线衍射求得的晶格面(002)的d值(层间距离)优选为0.335nm以上,并且通常为0.350nm以下,优选为0.345nm以下,更优选为0.340nm以下。另外,通过采用学振法的X射线衍射求得的碳材料的微晶尺寸(Lc)通常为1.5nm以上,优选为3.0nm以上的范围。如果低于该范围,则结晶性降低,有可能增加初期不可逆容量的增大。The d value (interlayer distance) of the lattice plane (002) obtained by the X-ray diffraction of the composite carbonaceous material is preferably 0.335nm or more, and is usually 0.350nm or less, preferably 0.345nm or less, more Preferably it is 0.340 nm or less. In addition, the crystallite size (Lc) of the carbon material obtained by X-ray diffraction using the Gakushin method is usually 1.5 nm or more, preferably 3.0 nm or more. If it is less than this range, the crystallinity may decrease, which may increase the increase in the initial irreversible capacity.
(2)灰分(2) Ash content
复合碳质物质所含的灰分相对于复合碳质物质的总质量优选为1质量%以下,更优选为0.5质量%以下,特别优选为0.1质量%以下,作为其下限,优选为1ppm以上。如果超过上述范围,则充放电时与电解液的反应而引起的电池性能的劣化不能忽视。如果低于该范围,则在制造上需要较长时间和能量以及用于防止污染的设备,有时成本会上升。The ash contained in the composite carbonaceous material is preferably 1% by mass or less, more preferably 0.5% by mass or less, particularly preferably 0.1% by mass or less with respect to the total mass of the composite carbonaceous material, and the lower limit thereof is preferably 1 ppm or more. If the above-mentioned range is exceeded, the degradation of battery performance due to the reaction with the electrolytic solution during charging and discharging cannot be ignored. If it is less than this range, long time and energy are required for manufacturing and equipment for preventing pollution, and the cost may increase.
(3)体积基准平均粒径(3) Volume-based average particle size
复合碳质物质的体积基准平均粒径是通过激光衍射/散射法求得的体积基准的平均粒径(中值粒径),通常为1μm以上,优选为3μm以上,更优选为5μm以上,进一步优选为7μm以上。另外,其上限通常为100μm以下,优选为50μm以下,更优选为40μm以下,进一步优选为30μm以下,特别优选为25μm以下。如果低于上述范围,则不可逆容量增大,有时导致初期电池容量的损失。另外,如果超过上述范围,则在制成电极极板时容易形成不均匀的涂布面,有时在电池制作工序上是不优选的。The volume-based average particle size of the composite carbonaceous material is the volume-based average particle size (median particle size) obtained by the laser diffraction/scattering method, usually more than 1 μm, preferably more than 3 μm, more preferably more than 5 μm, and further It is preferably 7 μm or more. In addition, the upper limit thereof is usually 100 μm or less, preferably 50 μm or less, more preferably 40 μm or less, still more preferably 30 μm or less, particularly preferably 25 μm or less. If it is less than the above-mentioned range, the irreversible capacity may increase, resulting in loss of initial battery capacity. Moreover, if it exceeds the said range, it will become easy to form an uneven coating surface at the time of making an electrode plate, and it may be unpreferable in a battery manufacturing process.
(4)拉曼R值、拉曼半值宽(4) Raman R value, Raman half value width
使用氩离子激光拉曼光谱法测定的复合碳质物质的R值通常为0.03以上,优选为0.10以上,更优选为0.15以上,其上限通常为0.60以下,优选为0.50以下的范围。如果R值低于该范围,则粒子表面的结晶性过高,随着充放电Li进入层间的位点有时变少。即,充电接受性有时降低。另外,涂布在集电体上之后,通过压制使负极高密度化时,结晶容易取向为与电极板平行的方向,有时导致负荷特性降低。另一方面,如果超过该范围,则粒子表面的结晶性降低,与电解液的反应性增加,有时导致效率降低或产生的气体增加。The R value of the composite carbonaceous material measured by argon ion laser Raman spectroscopy is usually 0.03 or more, preferably 0.10 or more, more preferably 0.15 or more, and its upper limit is usually 0.60 or less, preferably 0.50 or less. If the R value is lower than this range, the crystallinity of the particle surface is too high, and there may be fewer sites for Li to enter the interlayer due to charge and discharge. That is, charge acceptance may be lowered. In addition, when the anode is densified by pressing after coating on the current collector, the crystals tend to be oriented in a direction parallel to the electrode plate, which may result in a decrease in load characteristics. On the other hand, if it exceeds this range, the crystallinity of the particle surface decreases, and the reactivity with the electrolyte solution increases, resulting in a decrease in efficiency or an increase in gas generation.
另外,复合碳质物质在1580cm-1附近的拉曼半值宽度没有特别的限制,通常为15cm-1以上,优选为20cm-1以上,其上限通常为70cm-1以下,优选为60cm-1以下,更优选为50cm-1以下的范围。如果拉曼半值宽度低于该范围,则粒子表面的结晶性过高,随着充放电Li进入层间的位点有时变少。即,充电接受性有时降低。另外,涂布在集电体上之后,通过压制使负极高密度化时,结晶容易取向为与电极板平行的方向,有时导致负荷特性降低。另一方面,如果超过该范围,则粒子表面的结晶性降低,与电解液的反应性增加,有时导致效率降低或产生的气体增加。In addition, the Raman half-value width of the composite carbonaceous material in the vicinity of 1580 cm -1 is not particularly limited, and is usually 15 cm -1 or more, preferably 20 cm -1 or more, and its upper limit is usually 70 cm -1 or less, preferably 60 cm -1 or less, more preferably in the range of 50 cm -1 or less. If the Raman half-value width is below this range, the crystallinity of the particle surface may be too high, and there may be fewer sites for Li to enter the interlayer due to charge and discharge. That is, charge acceptance may be lowered. In addition, when the anode is densified by pressing after coating on the current collector, the crystals tend to be oriented in a direction parallel to the electrode plate, which may result in a decrease in load characteristics. On the other hand, if it exceeds this range, the crystallinity of the particle surface decreases, and the reactivity with the electrolyte solution increases, resulting in a decrease in efficiency or an increase in gas generation.
(5)BET比表面积(5) BET specific surface area
使用BET法测定的本发明的复合碳质物质的比表面积通常为0.1m2/g以上,优选为0.7m2/g以上,更优选为1m2/g以上,进一步优选为1.5m2/g以上。其上限通常为100m2/g以下,优选为25m2/g以下,更优选为15m2/g以下,进一步优选为10m2/g以下。比表面积的值如果低于上述范围,则在作为负极材料使用的情况下,充电时锂的接受性变差,锂容易在电极表面析出。另一方面,如果超过上述范围,则作为负极材料使用时,与电解液的反应性增加,产生的气体变多,有时难以得到优选的电池。The specific surface area of the composite carbonaceous material of the present invention measured by the BET method is usually 0.1 m 2 /g or more, preferably 0.7 m 2 /g or more, more preferably 1 m 2 /g or more, and still more preferably 1.5 m 2 /g above. The upper limit thereof is usually 100 m 2 /g or less, preferably 25 m 2 /g or less, more preferably 15 m 2 /g or less, still more preferably 10 m 2 /g or less. If the value of the specific surface area is less than the above range, when used as a negative electrode material, the acceptance of lithium at the time of charging will be deteriorated, and lithium will be easily deposited on the surface of the electrode. On the other hand, if it exceeds the above-mentioned range, when used as a negative electrode material, the reactivity with the electrolytic solution increases and the gas generated increases, making it difficult to obtain a preferable battery.
(6)微孔分布(6) Micropore distribution
作为复合类碳质物质,通过水银孔隙率计(水银压入法)求得的相当于直径0.01μm~1μm的粒子内的空隙、由于粒子表面的高低不平造成的凹凸的量通常为0.01mL/g以上,优选为0.05mL/g以上,更优选为0.1mL/g以上,其上限通常为0.6mL/g以下,优选为0.4mL/g以下,更优选为0.3mL/g以下的范围。如果超过该范围,则制成极板时需要大量的粘合剂。如果低于该范围,则高电流密度充放电特性降低,并且,有时不能得到充放电时的电极膨胀收缩的缓和效果。As a composite carbonaceous material, the amount of voids in particles with a diameter of 0.01 μm to 1 μm and the amount of unevenness caused by particle surface is usually 0.01 mL/ g or more, preferably 0.05 mL/g or more, more preferably 0.1 mL/g or more, and the upper limit is usually 0.6 mL/g or less, preferably 0.4 mL/g or less, more preferably 0.3 mL/g or less. If it exceeds this range, a large amount of binder will be required to produce a pole plate. If it is less than this range, the high current density charge-discharge characteristics will deteriorate, and the effect of alleviating electrode expansion and contraction during charge-discharge may not be obtained.
另外,总微孔容积优选为0.1mL/g以上,更优选为0.25mL/g以上,其上限通常为10mL/g以下,优选为5mL/g以下,更优选为2mL/g以下的范围。如果超过该范围,则有时需要大量的粘合剂。而如果低于该范围,则在制成极板时有时不能得到增稠剂或粘结剂的分散效果。In addition, the total pore volume is preferably 0.1 mL/g or more, more preferably 0.25 mL/g or more, and its upper limit is usually 10 mL/g or less, preferably 5 mL/g or less, more preferably 2 mL/g or less. If this range is exceeded, a large amount of adhesive is sometimes required. On the other hand, if it is less than this range, the dispersion effect of the thickener or the binder may not be obtained when it is made into an electrode plate.
另外,平均微孔径优选为0.05μm以上,更优选为0.1μm以上,进一步优选为0.5μm以上,其上限通常为80μm以下,优选为50μm以下,更优选为20μm以下的范围。如果超过该范围,则有时需要大量的粘合剂。而如果低于该范围,则有时高电流密度充放电特性降低。In addition, the average pore diameter is preferably 0.05 μm or more, more preferably 0.1 μm or more, even more preferably 0.5 μm or more, and the upper limit thereof is usually 80 μm or less, preferably 50 μm or less, more preferably 20 μm or less. If this range is exceeded, a large amount of adhesive is sometimes required. On the other hand, if it is less than this range, the high current density charge and discharge characteristics may deteriorate.
(7)圆形度(7) Circularity
使用圆形度作为复合碳质物质的球形的程度,复合碳质物质的粒径为3~40μm范围的粒子的圆形度优选为0.85以上,更优选为0.87以上,进一步优选为0.9以上。圆形度大时,高电流密度充放电特性提高,因此优选。Using the circularity as the degree of sphericity of the composite carbonaceous material, the circularity of particles of the composite carbonaceous material having a particle size in the range of 3 to 40 μm is preferably 0.85 or more, more preferably 0.87 or more, and still more preferably 0.9 or more. When the circularity is large, the high current density charge and discharge characteristics are improved, which is preferable.
(8)真密度(8) True density
复合碳质物质的真密度通常为1.9g/cm3以上,优选为2g/cm3以上,更优选为2.1g/cm3以上,进一步优选为2.2g/cm3以上,其上限为2.26g/cm3以下。上限为石墨的理论值。如果低于该范围,则碳的结晶性过低,初期不可逆容量有时会增大。The true density of the composite carbonaceous material is usually above 1.9 g/cm 3 , preferably above 2 g/cm 3 , more preferably above 2.1 g/cm 3 , even more preferably above 2.2 g/cm 3 , and its upper limit is 2.26 g/cm 3 or above. cm3 or less. The upper limit is the theoretical value of graphite. If it is less than this range, the crystallinity of carbon may be too low, and the initial irreversible capacity may increase.
(9)振实密度(9) Tap density
复合碳质物质的振实密度通常为0.1g/cm3以上,优选为0.5g/cm3以上,更优选为0.7g/cm3以上,特别优选为1g/cm3以上。另外,其上限优选为2g/cm3以下,更优选为1.8g/cm3以下,特别优选为1.6g/cm3以下。如果振实密度低于该范围,则在作为负极使用时,填充密度很难提高,有时不能得到高容量的电池。另一方面,如果超过该范围,则电极中的粒子间的空隙过少,难以确保粒子间的导电性,有时难以得到优选的电池特性。振实密度通过与上述方法相同的方法进行测定,并根据该方法定义。The tap density of the composite carbonaceous material is usually 0.1 g/cm 3 or more, preferably 0.5 g/cm 3 or more, more preferably 0.7 g/cm 3 or more, particularly preferably 1 g/cm 3 or more. In addition, the upper limit thereof is preferably 2 g/cm 3 or less, more preferably 1.8 g/cm 3 or less, particularly preferably 1.6 g/cm 3 or less. When the tap density is lower than this range, it is difficult to increase the packing density when used as a negative electrode, and a high-capacity battery may not be obtained. On the other hand, if it exceeds this range, the gaps between the particles in the electrode will be too small, making it difficult to ensure the conductivity between the particles, and it may be difficult to obtain preferable battery characteristics. The tap density is measured by the same method as described above, and is defined according to the method.
(10)取向比(粉末)(10) Orientation ratio (powder)
复合碳质物质的取向比通常为0.005以上,优选为0.01以上,更优选为0.015以上,其上限在理论上为0.67以下的范围。如果低于该范围,则高密度充放电特性有时降低。The orientation ratio of the composite carbonaceous material is usually 0.005 or more, preferably 0.01 or more, more preferably 0.015 or more, and the upper limit is theoretically 0.67 or less. If it is less than this range, the high-density charge and discharge characteristics may deteriorate.
(11)长径比(粉末)(11) aspect ratio (powder)
复合碳质物质的长径比理论上为1以上,其上限通常为10以下,优选为8以下,更优选为5以下。如果超过其上限,制成极板时有时会产生条纹、无法得到均匀的涂布面,高电流密度充放电特性降低。The aspect ratio of the composite carbonaceous material is theoretically 1 or more, and its upper limit is usually 10 or less, preferably 8 or less, more preferably 5 or less. If the upper limit is exceeded, streaks may be generated when forming an electrode plate, a uniform coating surface may not be obtained, and the high current density charge-discharge characteristics may deteriorate.
(复合碳质物质的制造方法)(Manufacturing method of composite carbonaceous material)
这些复合碳质物质的制造方法没有特别的限制,可以举出以下所示的方法。The method for producing these composite carbonaceous materials is not particularly limited, and the methods shown below are exemplified.
石墨类碳质物质和低结晶性碳质物质的复合化可以采用以下方法:直接使用用于得到低结晶性碳质物质的碳前体,加热处理碳前体和石墨类碳质物质粉末的混合物,得到复合粉末的方法;将上述碳前体进行部分碳化,预先制备低结晶性碳质物质粉末,再与石墨类碳质物质粉末混合,加热处理以进行复合化的方法;预先制作上述低结晶性碳质物质粉末,混合石墨类碳质物质粉末、低结晶性碳质物质粉末和碳前体,加热进行复合化的方法等。另外,在后两种预先准备低结晶性碳质物质粉末的方法中,优选使用平均粒径为石墨类碳质物质的平均粒径的十分之一以下的低结晶性碳质物质粒子。另外,还可以采用以下方法:通过对预先制作的低结晶性碳质物质和石墨类碳质物质施加粉碎等力学能量,制成一种物质夹杂入另一物质中的结构,或者静电附着的结构的方法。The compounding of graphite-like carbonaceous substance and low-crystalline carbonaceous substance can adopt the following method: directly use the carbon precursor for obtaining low-crystalline carbonaceous substance, and heat-treat the mixture of carbon precursor and graphite-like carbonaceous substance powder , the method of obtaining composite powder; the above-mentioned carbon precursor is partially carbonized, and the low-crystalline carbonaceous substance powder is prepared in advance, and then mixed with the graphite-like carbonaceous substance powder, and the method for compounding by heat treatment; the above-mentioned low-crystalline carbonaceous substance powder is prepared in advance. Carbonaceous substance powder, a method of mixing graphite-like carbonaceous substance powder, low-crystalline carbonaceous substance powder and carbon precursor, heating and compounding, etc. In addition, in the latter two methods of preparing low-crystalline carbonaceous material powder in advance, it is preferable to use low-crystalline carbonaceous material particles whose average particle diameter is one-tenth or less of the average particle diameter of graphite-based carbonaceous material. In addition, the following method can also be used: by applying mechanical energy such as pulverization to prefabricated low-crystalline carbonaceous substances and graphite-like carbonaceous substances, a structure in which one substance is mixed into another substance, or a structure in which one substance is electrostatically attached Methods.
优选的是,对混合了石墨类碳质物质粒子和碳前体的混合物进行加热,得到中间物质,或者,对石墨类碳质物质粒子和低结晶性碳质物质粒子的混合物与碳前体混合而得到的混合物进行加热,得到中间物质,然后,碳化烧结,粉碎,由此最终得到在石墨类物质粒子中复合化有低结晶性碳质物质的复合碳质物质。Preferably, the mixture of the graphite-like carbonaceous material particles and the carbon precursor is heated to obtain an intermediate substance, or the mixture of the graphite-like carbonaceous material particles and the low-crystalline carbonaceous material particles is mixed with the carbon precursor The obtained mixture is heated to obtain an intermediate substance, which is then carbonized, sintered, and pulverized to finally obtain a composite carbonaceous substance in which a low-crystalline carbonaceous substance is composited in graphite-like substance particles.
用于得到复合碳质物质的制造工序分为以下4个工序。The manufacturing process for obtaining a composite carbonaceous material is divided into the following 4 processes.
第1工序:使用各种市售的混合机或混炼机将(石墨类碳质物质粒子或(石墨类碳质物质粒子和低结晶性碳质物质粒子的混合粒子))以及低结晶性碳质物质粒子的碳前体以及根据需要添加的溶剂混合,得到混合物。Step 1: Mix (graphite carbonaceous particles or (mixed particles of graphite carbonaceous particles and low-crystalline carbonaceous particles)) and low-crystalline carbon using various commercially available mixers or kneaders. The carbon precursor of the substance particle and, if necessary, a solvent are mixed to obtain a mixture.
第2工序:加热混合物,得到除去了溶剂以及由碳前体产生的挥发成分的中间物质。此时,根据需要,可以边搅拌上述混合物边进行。另外,即使残留有挥发成分,由于可在后续的第3工序中除去,所以也没问题。Second step: heating the mixture to obtain an intermediate substance from which the solvent and the volatile components generated from the carbon precursor have been removed. At this time, stirring the above-mentioned mixture can be performed as needed. Also, even if volatile components remain, they can be removed in the subsequent third step, so there is no problem.
第3工序:在氮气、二氧化碳、氩气等非活性气体氛围下,将上述混合物或中间物质加热到400℃~3200℃,得到石墨·低结晶性碳质物质复合物质。Step 3: In an inert gas atmosphere such as nitrogen, carbon dioxide, and argon, heat the above mixture or intermediate substance to 400° C. to 3200° C. to obtain a graphite-low crystalline carbonaceous material composite.
第4工序:根据需要对上述复合物质进行粉碎、破碎、分级处理等粉末加工。Step 4: If necessary, powder processing such as pulverization, crushing, and classification treatment is performed on the above-mentioned composite substance.
这些工序中,根据情况可以省略第2工序和第4工序,第4工序也可以在第3工序之前进行。但是,在第3工序之前实施第4工序时,根据需要,再次进行粉碎、破碎、分级等粉末加工,得到复合碳质物质。Among these steps, the second step and the fourth step may be omitted depending on circumstances, and the fourth step may be performed before the third step. However, when the fourth step is carried out before the third step, powder processing such as pulverization, crushing, and classification is performed again as necessary to obtain a composite carbonaceous material.
另外,作为第3工序的加热处理条件,热历程温度条件是重要的。该温度的下限根据碳前体的种类、该热历程而有所不同,但通常为400℃以上,优选为900℃以上。另一方面,上限温度可以达到基本不具有超越石墨类碳质物质粒子核的结晶结构的结构秩序的温度。因此,作为热处理的上限温度,通常为3200℃以下,优选为2000℃以下,更优选为1500℃以下。在这样的热处理条件下,根据目的可以任意设定升温速度、冷却速度、热处理时间等。另外,还可以在比较低的温度区域进行热处理后,升温到规定的温度。另外,本工序中使用的反应器可以是间歇式,也可以是连续式,另外,可以是一台也可以是多台。In addition, heat history temperature conditions are important as heat treatment conditions in the third step. The lower limit of the temperature varies depending on the type of carbon precursor and the heat history, but is usually 400°C or higher, preferably 900°C or higher. On the other hand, the upper limit temperature may reach a temperature at which there is substantially no structural order beyond the crystal structure of the graphite-like carbonaceous material particle nucleus. Therefore, the upper limit temperature of the heat treatment is usually not higher than 3200°C, preferably not higher than 2000°C, and more preferably not higher than 1500°C. Under such heat treatment conditions, the heating rate, cooling rate, heat treatment time, etc. can be set arbitrarily according to the purpose. In addition, after performing heat treatment in a relatively low temperature range, the temperature may be raised to a predetermined temperature. In addition, the reactor used in this step may be a batch type or a continuous type, and may be one or multiple.
[[副材料混合]][[Vice material mix]]
除上述复合碳质物质以外,在本发明的锂二次电池的负极活性物质中含有一种以上与上述复合碳质物质在碳质的物性方面不同的碳质物质(碳质材料),由此可以进一步谋求电池性能的提高。这里所说的“碳质的物性”,是指X射线衍射参数、中值粒径、长径比、BET比表面积、取向比、拉曼R值、振实密度、真密度、微孔分布、圆形度、灰分量的一个以上的特性。另外,作为优选的实施方式,可以举出,体积基准粒度分布以中值粒径为中心时左右不对称、含有2种以上拉曼R值不同的碳质材料、或X射线参数不同等。作为其效果的一个例子,可列举通过含有天然石墨、人造石墨等石墨;乙炔黑等炭黑;针状焦等无定形碳等碳材料作为副材料来降低电阻等。它们可以单独使用一种,也可以以任意组合和任意比例组合使用2种以上。作为副材料添加时,其添加量为0.1质量%以上,优选为0.5质量%以上,更优选为0.6质量%以上,其上限为80质量%以下,优选为50质量%以下,更优选为40质量%以下,进一步优选为30质量%以下的范围。如果低于该范围,则有时难以得到导电性提高的效果。如果超过上述范围,则有时会导致初期不可逆容量的增大。In addition to the above-mentioned composite carbonaceous substance, the negative electrode active material of the lithium secondary battery of the present invention contains one or more carbonaceous substances (carbonaceous materials) different from the above-mentioned composite carbonaceous substance in terms of carbonaceous physical properties, thereby Further improvement in battery performance can be achieved. The "carbonaceous physical properties" mentioned here refer to X-ray diffraction parameters, median particle size, aspect ratio, BET specific surface area, orientation ratio, Raman R value, tap density, true density, micropore distribution, One or more properties of circularity, gray content. In addition, as a preferred embodiment, the volume-based particle size distribution is asymmetrical around the median particle size, contains two or more carbonaceous materials with different Raman R values, or has different X-ray parameters. As an example of the effect, reduction of electrical resistance by including carbon materials such as graphite such as natural graphite and artificial graphite; carbon black such as acetylene black; and amorphous carbon such as needle coke as sub-materials can be cited. These may be used individually by 1 type, and may be used in combination of 2 or more types by arbitrary and arbitrary ratios. When added as an auxiliary material, the amount added is 0.1% by mass or more, preferably 0.5% by mass or more, more preferably 0.6% by mass or more, and the upper limit is 80% by mass or less, preferably 50% by mass or less, more preferably 40% by mass % or less, more preferably in the range of 30% by mass or less. If it is less than this range, it may be difficult to obtain the effect of improving electrical conductivity. If the above range is exceeded, the initial irreversible capacity may increase.
[制作负极[1]电极][Making the Negative [1] Electrode]
制造负极[1]可以采用通常的方法,可以与上述同样地形成负极[1]。集电体、集电体和活性物质层的厚度比、电极密度、粘合剂、极板取向比、阻抗等也与上述相同。A common method can be used to manufacture the negative electrode [1], and the negative electrode [1] can be formed in the same manner as above. The current collector, the thickness ratio between the current collector and the active material layer, electrode density, binder, plate orientation ratio, impedance, and the like are also the same as described above.
<负极[2]><Negative pole [2]>
下面,对本发明的锂二次电池中使用的负极[2]进行说明,该负极含有无定形碳质作为活性物质,所述无定形碳质采用广角X射线衍射法测定的(002)面的面间距(d002)为0.337nm以上,微晶大小(Lc)为80nm以下,在氩离子激光拉曼光谱法中以1360cm-1的峰强度相对于1580cm-1的峰强度的比值定义的拉曼R值为0.2以上。Next, the negative electrode [2] used in the lithium secondary battery of the present invention will be described. The negative electrode contains, as an active material, an amorphous carbonaceous substance whose (002) plane surface is measured by a wide-angle X-ray diffraction method. Raman R defined by the ratio of the peak intensity at 1360 cm -1 to the peak intensity at 1580 cm -1 in argon ion laser Raman spectroscopy with a spacing (d002) of 0.337 nm or more and a crystallite size (Lc) of 80 nm or less The value is 0.2 or more.
[负极[2]的负极活性物质][Negative electrode active material of the negative electrode [2]]
下面,对负极[2]中使用的负极活性物质进行说明。Next, the negative electrode active material used in the negative electrode [2] will be described.
本发明的锂二次电池的负极[2]中使用的负极活性物质是至少含有满足以下的(a)、(b)和(c)的无定形碳质。The negative electrode active material used in the negative electrode [2] of the lithium secondary battery of the present invention is an amorphous carbonaceous material containing at least the following (a), (b) and (c).
(a)采用广角X射线衍射法测定的(002)面的面间距(d002)为0.337nm以上;(a) The interplanar distance (d002) of the (002) plane measured by wide-angle X-ray diffraction method is 0.337nm or more;
(b)采用广角X射线衍射法测定的(002)面的微晶大小Lc为80nm以下;(b) The crystallite size Lc of the (002) plane measured by wide-angle X-ray diffraction method is below 80nm;
(c)在氩离子激光拉曼光谱法中以1360cm-1的峰强度相对于1580cm-1的峰强度的比值定义的拉曼R值(以下,有时简称为“拉曼R值”)为0.2以上。(c) The Raman R value defined by the ratio of the peak intensity at 1360 cm -1 to the peak intensity at 1580 cm -1 in argon ion laser Raman spectroscopy (hereinafter, sometimes simply referred to as "Raman R value") is 0.2 above.
本发明中使用的负极活性物质至少含有满足(a)、(b)和(c)的无定形碳质,这些无定形碳质在总负极活性物质中的含量优选为10质量%以上,更优选为50质量%以上,特别优选为100质量%,也就是所有活性物质都是无定形碳质。作为与无定形碳质组合使用的负极活性物质,没有特别的限定,可以使用公知的负极活性物质,例如,人造石墨、天然石墨等。The negative active material used in the present invention contains at least amorphous carbonaceous meeting (a), (b) and (c), the content of these amorphous carbonaceous in the total negative active material is preferably more than 10% by mass, more preferably It is 50% by mass or more, particularly preferably 100% by mass, that is, all active materials are amorphous carbonaceous. The negative electrode active material used in combination with amorphous carbon is not particularly limited, and known negative electrode active materials such as artificial graphite and natural graphite can be used.
[[面间距(d002)、Lc]][[Space between faces (d002), Lc]]
本发明的锂二次电池中作为负极[2]的负极活性物质使用的无定形碳质通过广角X射线衍射法测定的(002)面的面间距(d002)为0.337nm以上,优选为0.34nm以上。其上限通常为0.39nm以下,优选为0.38nm以下,更优选为0.37nm以下,进一步优选为0.36nm以下,特别优选为0.35nm以下。如果超过该范围,则结晶性显著降低,粒子间的导电性的降低不能忽视,有时难以得到短时间高电流密度充放电特性的提高效果。另一方面,如果低于该范围,则结晶性变得过高,有时难以得到短时间高电流密度充放电特性的提高效果。In the lithium secondary battery of the present invention, the interplanar distance (d002) of the (002) plane measured by the wide-angle X-ray diffraction method of the amorphous carbonaceous used as the negative electrode active material of the negative electrode [2] is 0.337 nm or more, preferably 0.34 nm above. The upper limit thereof is usually 0.39 nm or less, preferably 0.38 nm or less, more preferably 0.37 nm or less, still more preferably 0.36 nm or less, particularly preferably 0.35 nm or less. If it exceeds this range, the crystallinity will be remarkably reduced, and the reduction in the electrical conductivity between particles cannot be ignored, and it may be difficult to obtain the effect of improving the short-time high current density charge-discharge characteristics. On the other hand, if it is less than this range, the crystallinity becomes too high, and it may be difficult to obtain the effect of improving the short-time high current density charge-discharge characteristics.
本发明所说的采用广角X射线法测定的(002)面的面间距(d002)是指通过采用学振法的X射线衍射求得的晶格面(002)面的d值(层间距离)。The interplanar spacing (d002) of the (002) plane measured by the wide-angle X-ray method in the present invention refers to the d value (interlayer distance) of the lattice plane (002) plane obtained by X-ray diffraction using the Xuezhen method. ).
通过采用学振法测定的X射线衍射求得的无定形碳质的(002)面的微晶大小(Lc)为80nm以下,优选为35nm以下,更优选为20nm以下,进一步优选为10nm以下。其下限通常为0.1nm以上,优选为1nm以上。如果低于该范围,则结晶性显著降低,有时粒子间导电性的降低不能忽视,并且,有时难以得到短时间高电流密度充放电的提高效果。另一方面,如果超过该范围,则结晶性变得过高,有时难以得到短时间高电流密度充放电的提高效果。The crystallite size (Lc) of the (002) plane of the amorphous carbonaceous obtained by X-ray diffraction measured by the Gakushin method is 80 nm or less, preferably 35 nm or less, more preferably 20 nm or less, and even more preferably 10 nm or less. The lower limit thereof is usually 0.1 nm or more, preferably 1 nm or more. If it is less than this range, the crystallinity is remarkably reduced, and the reduction in the conductivity between particles may not be ignored, and it may be difficult to obtain the effect of improving charge and discharge at a high current density in a short time. On the other hand, if it exceeds this range, the crystallinity becomes too high, and it may be difficult to obtain the effect of improving short-time high current density charge and discharge.
[[拉曼R值]][[Raman R value]]
本发明的锂二次电池中作为负极[2]的负极活性物质使用的无定形碳质的拉曼R值必须为0.2以上,优选为0.5以上,特别优选为0.7以上,更优选为0.8以上。其上限通常为1.5以下,更优选为1.2以下。如果拉曼R值低于该范围,则粒子表面的结晶性过高,充电接受性降低,因此,有时难以得到短时间高电流密度充放电特性的提高效果。另一方面,如果超过该范围,则粒子表面的结晶性显著下降,因此,粒子间的接触电阻变大,有时难以得到短时间高电流密度充放电的提高效果。The Raman R value of the amorphous carbonaceous used as the negative electrode active material of the negative electrode [2] in the lithium secondary battery of the present invention must be 0.2 or more, preferably 0.5 or more, particularly preferably 0.7 or more, more preferably 0.8 or more. The upper limit thereof is usually 1.5 or less, more preferably 1.2 or less. If the Raman R value is lower than this range, the crystallinity of the particle surface is too high and the charge acceptance is lowered, so it may be difficult to obtain the effect of improving the short-time high current density charge-discharge characteristics. On the other hand, if it exceeds this range, the crystallinity of the surface of the particles will decrease significantly, so the contact resistance between the particles will increase, and it may be difficult to obtain the effect of improving charge and discharge at a high current density in a short time.
拉曼光谱的测定如下进行:使用拉曼分光器(例如,日本分光公司制造的拉曼分光器),使试样自然落入到测定容器内,进行试样填充,测定过程是,对容器内的试样表面照射氩离子激光,同时使该容器在与激光垂直的面内旋转。对于得到的拉曼光谱,测定1580cm-1的峰PA的强度IA和1360cm-1的峰PB的强度IB,计算其强度比R(R=IB/IA),将其定义为无定形碳质的拉曼R值。另外,测定得到的拉曼光谱在1580cm-1的峰PA的半值宽度,将其定义为无定形碳质的拉曼半值宽度。The measurement of the Raman spectrum is carried out as follows: using a Raman spectrometer (for example, a Raman spectrometer manufactured by JASCO Corporation), the sample is naturally dropped into the measurement container, and the sample is filled. The surface of the sample is irradiated with an argon ion laser while the container is rotated in a plane perpendicular to the laser. For the obtained Raman spectrum, measure the intensity I A of the peak PA at 1580 cm -1 and the intensity I B of the peak P B at 1360 cm -1 , calculate the intensity ratio R (R= IB / IA ), and define it as is the Raman R value of the amorphous carbonaceous. In addition, the half-value width of the peak PA at 1580 cm −1 in the obtained Raman spectrum was measured, and this was defined as the Raman half-value width of amorphous carbonaceous.
另外,这里的拉曼测定条件如下。In addition, the Raman measurement conditions here are as follows.
·氩离子激光波长:514.5nmArgon ion laser wavelength: 514.5nm
·试样上的激光功率:15~25mW·Laser power on the sample: 15~25mW
·分辨率:10~20cm-1 ·Resolution: 10~20cm -1
·测定范围:1100cm-1~1730cm-1 · Measuring range: 1100cm -1 ~ 1730cm -1
·拉曼R值、拉曼半值宽度分析:本底处理Raman R value, Raman half-value width analysis: background processing
·光滑处理:单纯平均、卷积5点·Smooth processing: simple average, convolution 5 points
另外,本发明的锂二次电池中作为负极[2]的负极活性物质使用的无定形碳质在1580cm-1的拉曼半值宽度没有特别的限制,通常为20cm-1以上,优选为25cm-1以上,另外,其上限通常为150cm-1以下,优选为140cm-1以下的范围。拉曼半值宽度如果低于该范围,则粒子表面的结晶性变得过高,充电接受性降低,因此,有时难以得到短时间高电流密度充放电的提高效果。另一方面,如果超过上述范围,则粒子表面的结晶性显著降低,因此,粒子间的接触电阻变大,有可能难以得到短时间高电流密度充放电的提高效果。但是,拉曼半值宽度有时不能通过峰形状来判断。In addition, in the lithium secondary battery of the present invention, the Raman half-value width of the amorphous carbonaceous used as the negative electrode active material of the negative electrode [2] is not particularly limited at 1580 cm -1 , and is usually more than 20 cm -1 , preferably 25 cm -1 or more, and the upper limit is usually 150 cm -1 or less, preferably 140 cm -1 or less. If the Raman half-value width is less than this range, the crystallinity of the particle surface becomes too high, and the charge acceptance decreases. Therefore, it may be difficult to obtain the effect of improving short-time high current density charge and discharge. On the other hand, if it exceeds the above-mentioned range, the crystallinity of the particle surface will be significantly reduced, so the contact resistance between particles will increase, and it may be difficult to obtain the effect of improving charge and discharge at a high current density in a short time. However, the Raman half-value width cannot sometimes be judged from the peak shape.
本发明使用的无定形碳质满足上述的面间距(d002)、微晶大小(Lc)以及拉曼R值的条件,但从电池的平衡这一点上来讲,更优选同时满足下述项目中的任意一项或多项条件。这些之中,特别优选同时满足真密度、H/C值、O/C值、振实密度、BET比表面积、0.01μm~1μm范围的微孔容积、灰分以及体积平均粒径中的任意一项或多项条件。The amorphous carbon used in the present invention satisfies the above-mentioned interplanar spacing (d002), crystallite size (Lc) and Raman R value conditions, but from the point of view of battery balance, it is more preferable to satisfy the following items at the same time Any one or more conditions. Among these, it is particularly preferable to simultaneously satisfy any one of true density, H/C value, O/C value, tap density, BET specific surface area, micropore volume in the range of 0.01 μm to 1 μm, ash content, and volume average particle diameter. or multiple conditions.
[[真密度]][[true density]]
无定形碳质的真密度通常为2.22g/cm3以下,优选为2.2g/cm3以下,更优选为2.1g/cm3以下,进一步优选为2.0g/cm3以下,其下限通常为1.4g/cm3以上,优选为1.5g/cm3以上,更优选为1.6g/cm3以上,进一步优选为1.7g/cm3以上,特别优选为1.8g/cm3以上的范围。如果低于该范围,则碳的结晶性过低,有时初期不可逆容量增大。如果超过该范围,则碳的结晶性过高,难以得到短时间高电流密度充放电的提高效果。The true density of amorphous carbonaceous is usually 2.22 g/cm 3 or less, preferably 2.2 g/cm 3 or less, more preferably 2.1 g/cm 3 or less, still more preferably 2.0 g/cm 3 or less, and its lower limit is usually 1.4 g/cm 3 or more, preferably 1.5 g/cm 3 or more, more preferably 1.6 g/cm 3 or more, still more preferably 1.7 g/cm 3 or more, particularly preferably 1.8 g/cm 3 or more. If it is less than this range, the crystallinity of carbon may be too low, and the initial irreversible capacity may increase. If it exceeds this range, the crystallinity of carbon will be too high, and it will be difficult to obtain the effect of improving charging and discharging at a high current density in a short time.
在本发明中,真密度定义为通过使用丁醇的液相置换法(比重计法)测定的值。In the present invention, the true density is defined as a value measured by a liquid phase displacement method (pycnometer method) using butanol.
[[O/C值]][[O/C value]]
无定形碳质的原子比量O/C值的上限通常为0.15以下,优选为0.1以下,更优选为0.05以下,进一步优选为0.03以下,其下限通常为0以上,优选为0.01以上。The upper limit of the atomic ratio O/C value of amorphous carbon is usually 0.15 or less, preferably 0.1 or less, more preferably 0.05 or less, further preferably 0.03 or less, and the lower limit is usually 0 or more, preferably 0.01 or more.
O/C值表示无定形碳质中存在的氧原子的摩尔浓度与碳原子的摩尔浓度的比例,是表示羧基、酚性羟基、羰基等官能团的存在量的指标。O/C值大的无定形碳质大多在碳微晶的端面等结合有含氧官能团。O/C值如果超过上述范围,则不可逆容量有时增大。The O/C value represents the ratio of the molar concentration of oxygen atoms present in amorphous carbon to the molar concentration of carbon atoms, and is an index representing the amount of functional groups such as carboxyl groups, phenolic hydroxyl groups, and carbonyl groups. Amorphous carbon with a large O/C value often has oxygen-containing functional groups bonded to the end faces of carbon crystallites. When the O/C value exceeds the above range, the irreversible capacity may increase.
[[H/C值]][[H/C value]]
无定形碳质的原子比量H/C值的上限通常为0.3以下,优选为0.15以下,更优选为0.1以下,进一步优选为0.08以下,其下限通常为0以上,优选为0.01以上。The upper limit of the atomic ratio H/C value of amorphous carbon is usually 0.3 or less, preferably 0.15 or less, more preferably 0.1 or less, further preferably 0.08 or less, and the lower limit is usually 0 or more, preferably 0.01 or more.
所谓H/C值,表示无定形碳质中存在的氢原子的摩尔浓度与碳原子的摩尔浓度的比例,是表示存在于无定形碳质的微晶端面上的氢的存在量的指标。H/C值大的无定形碳质材料,大多表示粒子面碳的微晶端面等多于碳的量。如果H/C值超过上述范围,则有时不可逆容量增大。The H/C value represents the ratio of the molar concentration of hydrogen atoms present in the amorphous carbonaceous substance to the molar concentration of carbon atoms, and is an index showing the amount of hydrogen present on the crystallite end faces of the amorphous carbonaceous substance. An amorphous carbonaceous material with a large H/C value often indicates that the amount of carbon such as crystallite end surfaces of carbon on the particle surface is greater than that of carbon. If the H/C value exceeds the above range, the irreversible capacity may increase.
本发明中所说的“O/C值”和“H/C值”通过下面所示的CHN元素分析求出。The "O/C value" and "H/C value" mentioned in the present invention are determined by CHN elemental analysis shown below.
[[CHN元素分析]][[CHN Elemental Analysis]]
将待测定的无定形碳质在120℃下减压干燥约15小时,然后放在干燥箱内的热板上于100℃下干燥1小时。接着,在氩气氛围中,将试样置于铝杯中,通过燃烧产生的二氧化碳气体的重量求出碳含量,另外,通过产生的水的重量求出氢含量,通过产生的二氧化氮的重量计算含氮量,由燃烧后残留的残渣的重量求出无机物含量。氧含量的值通过从总重量中减去碳含量、氢含量、氮含量、无机物含量而得出。由这些值计算摩尔数,采用得到的各个含量摩尔数通过下式求出O/C值、H/C值。The amorphous carbon to be measured was dried under reduced pressure at 120° C. for about 15 hours, and then dried on a hot plate in a drying oven at 100° C. for 1 hour. Next, in an argon atmosphere, the sample was placed in an aluminum cup, and the carbon content was obtained from the weight of carbon dioxide gas generated by combustion, and the hydrogen content was obtained from the weight of water generated, and the nitrogen dioxide gas generated was obtained by The nitrogen content was calculated by weight, and the inorganic substance content was obtained from the weight of the residue remaining after combustion. The value of the oxygen content is obtained by subtracting the carbon content, the hydrogen content, the nitrogen content, and the inorganic content from the total weight. The number of moles was calculated from these values, and the O/C value and the H/C value were obtained by the following formula using the obtained mole numbers of each content.
O/C值=含氧摩尔数/含碳摩尔数O/C value = moles of oxygen/moles of carbon
H/C值=含氢摩尔数/含碳摩尔数H/C value = moles of hydrogen/moles of carbon
[[振实密度]][[Tap Density]]
本发明的锂二次电池中作为负极[2]的活性物质使用的无定形碳质的振实密度优选为0.1g/cm3以上,更优选为0.2g/cm3以上,进一步优选为0.5g/cm3以上,特别优选为0.7g/cm3以上。另外,其上限优选为1.4g/cm3以下,更优选为1.2g/cm3以下,特别优选为1.1g/cm3以下。如果振实密度低于该范围,则作为负极使用时难以提高填充密度,粒子间的接触面积减少,因此粒子间的阻抗增加,有时会降低短时间高电流密度充放电特性。另一方面,如果超过该范围,则电极中的粒子间的空隙过少,非水电解液的流路会减少,因此有时会减少短时间高电流密度充放电特性。The tap density of the amorphous carbonaceous used as the active material of the negative electrode [2] in the lithium secondary battery of the present invention is preferably 0.1 g/cm 3 or more, more preferably 0.2 g/cm 3 or more, still more preferably 0.5 g /cm 3 or more, particularly preferably 0.7 g/cm 3 or more. In addition, the upper limit thereof is preferably 1.4 g/cm 3 or less, more preferably 1.2 g/cm 3 or less, particularly preferably 1.1 g/cm 3 or less. If the tap density is lower than this range, it will be difficult to increase the packing density when used as a negative electrode, and the contact area between particles will decrease, so the impedance between particles will increase, and the short-term high current density charge and discharge characteristics may be reduced. On the other hand, if it exceeds this range, the gaps between the particles in the electrode will be too small, and the flow path of the nonaqueous electrolyte solution will be reduced, so the short-time high current density charge and discharge characteristics may be reduced.
在本发明中,振实密度如下定义:使试样通过孔径300μm的筛子,使试样落入20cm3的振实容器中,到试样充满容器的上端面后,使用粉末密度测定器(例如,seishin企业公司制造的Tap densor),进行1000次冲程长度10mm的振动,将由此时的体积和试样的重量求出的密度作为振实密度。In the present invention, the tap density is defined as follows: the sample is passed through a sieve with an aperture of 300 μm, the sample is dropped into a 20 cm container for vibration, and after the sample is filled with the upper end of the container, a powder density measuring device (such as , Tap densor manufactured by Seishin Enterprise Co., Ltd.), vibration with a stroke length of 10 mm was performed 1000 times, and the density obtained from the volume at that time and the weight of the sample was taken as the tap density.
[[BET比表面积]][[BET specific surface area]]
本发明的锂二次电池中作为负极[2]的负极活性物质使用的无定形碳质通过BET法测定的比表面积优选为0.1m2/g以上,特别优选为0.5m2/g以上,更优选为0.7m2/g以上,进一步优选为1.5m2/g以上。其上限优选为100m2/g以下,特别优选为50m2/g以下,更优选为25m2/g以下,进一步优选为15m2/g以下。BET比表面积的值如果低于该范围,则在作为负极材料使用的情况下,在充电时锂的接受性容易变差,有时锂会在电极表面析出。另一方面,BET比表面积的值如果超过该范围,作为负极材料使用时,与非水电解液的反应性增加,产生的气体变多,有时得不到优选的电池。In the lithium secondary battery of the present invention, the specific surface area of the amorphous carbonaceous used as the negative electrode active material of the negative electrode [2] measured by the BET method is preferably 0.1 m 2 /g or more, particularly preferably 0.5 m 2 /g or more, and more preferably Preferably it is 0.7 m 2 /g or more, and more preferably 1.5 m 2 /g or more. The upper limit thereof is preferably 100 m 2 /g or less, particularly preferably 50 m 2 /g or less, more preferably 25 m 2 /g or less, and still more preferably 15 m 2 /g or less. If the value of the BET specific surface area is below this range, when used as a negative electrode material, lithium acceptance during charge tends to deteriorate, and lithium may be deposited on the electrode surface. On the other hand, if the value of the BET specific surface area exceeds this range, when used as a negative electrode material, the reactivity with the non-aqueous electrolyte solution increases, the gas generated increases, and a preferable battery may not be obtained.
BET比表面积定义为如下的值:使用表面积计(例如,大仓理研制造的全自动表面积测定装置),在氮气流通、350℃下对试样进行15分钟预干燥,然后,使用氮气相对于大气压的相对压力值准确调整为0.3的氮氦混合气体,通过采用气体流动法的氮吸附BET1点法测定的值。The BET specific surface area is defined as a value as follows: Using a surface area meter (for example, a fully automatic surface area measuring device manufactured by Okura Riken), the sample is pre-dried at 350° C. for 15 minutes under nitrogen flow, and then, using nitrogen gas relative to atmospheric pressure The relative pressure value is accurately adjusted to 0.3 of the nitrogen-helium mixed gas, the value measured by the nitrogen adsorption BET1 point method using the gas flow method.
[[体积平均粒径]][[Volume average particle size]]
本发明的锂二次电池中作为负极[2]的负极活性物质使用的无定形碳质的体积平均粒径定义为通过激光衍射/散射法求得的体积基准的平均粒径(中值粒径),通常为1μm以上,优选为3μm以上,更优选为5μm以上,进一步优选为7μm以上。另外,其上限通常为50μm以下,优选为40μm以下,更优选为30μm以下,进一步优选为25μm以下。如果低于上述范围,则不可逆容量增大,有时导致初期电池容量的损失。另外,如果超过上述范围,则制成电极的极板时,容易形成不均匀的涂布面,有时在电池制作工序上是不优选的。The volume average particle size of the amorphous carbonaceous used as the negative electrode active material of the negative electrode [2] in the lithium secondary battery of the present invention is defined as the average particle size (median particle size) of the volume basis obtained by the laser diffraction/scattering method ), usually 1 μm or more, preferably 3 μm or more, more preferably 5 μm or more, and even more preferably 7 μm or more. In addition, the upper limit thereof is usually 50 μm or less, preferably 40 μm or less, more preferably 30 μm or less, and still more preferably 25 μm or less. If it is less than the above-mentioned range, the irreversible capacity may increase, resulting in loss of initial battery capacity. In addition, when the above range is exceeded, an uneven coating surface is likely to be formed when forming an electrode plate, which may be unfavorable in the battery production process.
[[微孔容积]][[pore volume]]
本发明的锂二次电池中作为负极[2]的负极活性物质使用的无定形碳质的微孔容积,通过水银孔隙率计(水银压入法)求得的相当于直径0.01μm~1μm的粒子内的空隙、由于粒子表面的高低不平造成的凹凸的量为0.01mL/g以上,优选为0.05mL/g以上,更优选为0.1mL/g以上,其上限通常为0.6mL/g以下,优选为0.4mL/g以下,更优选为0.3mL/g以下的范围。如果超过该范围,则制造成极板时需要大量的粘合剂。而如果低于该范围,则短时间高电流密度充放电特性降低,并且,有时不能得到充放电时的电极膨胀收缩的缓和效果。The micropore volume of the amorphous carbonaceous used as the negative electrode active material of the negative electrode [2] in the lithium secondary battery of the present invention is obtained by a mercury porosimeter (mercury porosimetry) and is equivalent to a diameter of 0.01 μm to 1 μm. The amount of voids in the particles and the unevenness of the particle surface is 0.01 mL/g or more, preferably 0.05 mL/g or more, more preferably 0.1 mL/g or more, and the upper limit is usually 0.6 mL/g or less, The range is preferably 0.4 mL/g or less, more preferably 0.3 mL/g or less. If it exceeds this range, a large amount of binder will be required to manufacture an electrode plate. On the other hand, if it is less than this range, the short-time high current density charge-discharge characteristics will deteriorate, and the effect of alleviating electrode expansion and contraction during charge-discharge may not be obtained.
另外,总微孔容积优选0.1mL/g以上,更优选为0.25mL/g以上,其上限通常为10mL/g以下,优选为5mL/g以下,更优选为2mL/g以下的范围。如果超过该范围,则有时需要大量的粘合剂。如果低于该范围,则在制成极板时有时不能得到增稠剂或粘结剂的分散效果。In addition, the total pore volume is preferably 0.1 mL/g or more, more preferably 0.25 mL/g or more, and its upper limit is usually 10 mL/g or less, preferably 5 mL/g or less, more preferably 2 mL/g or less. If this range is exceeded, a large amount of adhesive is sometimes required. If it is less than this range, the dispersion effect of the thickener or the binder may not be obtained when forming an electrode plate.
另外,平均微孔径优选为0.05μm以上,更优选为0.1μm以上,进一步优选为0.5μm以上,其上限通常为50μm以下,优选为20μm以下,更优选为10μm以下的范围。如果超过该范围,则有时需要大量的粘合剂。另一方面,如果低于该范围,则有时短时间高电流密度充放电特性降低。In addition, the average pore diameter is preferably 0.05 μm or more, more preferably 0.1 μm or more, even more preferably 0.5 μm or more, and the upper limit thereof is usually 50 μm or less, preferably 20 μm or less, more preferably 10 μm or less. If this range is exceeded, a large amount of adhesive is sometimes required. On the other hand, if it is less than this range, the short-time high current density charge and discharge characteristics may deteriorate.
作为用于水银孔隙率计的装置,可以使用水银孔隙率计(autopore9520;micrometritics公司制造)。称量约0.2g试样(负极材料),密封入粉末用容器中,在室温、真空下(50μmHg以下)进行10分钟脱气,实施前处理。接着,减压至4psia(约28kPa),导入水银,从4psia(约28kPa)阶段状升压至40000psia(约280MPa)后,再降压至25psia(约170kPa)。升压时的阶段数为80阶段以上,在各阶段中,在10秒的平衡时间后,测定水银压入量。使用Washburn式从这样得到的水银压入曲线计算微孔分布。另外,水银的表面张力(γ)为485dyne/cm,接触角(φ)为140°。平均微孔径使用累积微孔体积达到50%时的微孔径。As an apparatus used for the mercury porosimeter, a mercury porosimeter (autopore9520; manufactured by Micrometrics Inc.) can be used. About 0.2 g of the sample (negative electrode material) was weighed, sealed in a powder container, and degassed at room temperature under vacuum (50 μmHg or less) for 10 minutes to perform pretreatment. Next, depressurize to 4psia (about 28kPa), introduce mercury, increase the pressure from 4psia (about 28kPa) to 40000psia (about 280MPa) stepwise, and then lower the pressure to 25psia (about 170kPa). The number of stages during the pressurization was 80 or more, and the amount of mercury intrusion was measured after an equilibration time of 10 seconds in each stage. The micropore distribution was calculated from the mercury intrusion curve thus obtained using the Washburn formula. In addition, the surface tension (γ) of mercury is 485 dyne/cm, and the contact angle (φ) is 140°. As the average pore diameter, the pore diameter at which the cumulative pore volume reaches 50% is used.
[[灰分]][[ash]]
碳质材料中所含的灰分相对于碳质材料的总质量优选为1质量%以下,更优选为0.5质量%以下,特别优选为0.1质量%以下,另外,作为下限,以质量计,优选为1ppm以上。如果超过上述范围,则充放电时由于与非水电解液的反应而引起的电池性能的劣化不能忽视。另一方面,如果低于该范围,则在制造上需要较长时间和能量以及用于防止污染的设备,有时成本会上升。The ash contained in the carbonaceous material is preferably 1% by mass or less, more preferably 0.5% by mass or less, and particularly preferably 0.1% by mass or less with respect to the total mass of the carbonaceous material. In addition, as the lower limit, it is preferably by mass. Above 1ppm. If the above range is exceeded, the deterioration of battery performance due to the reaction with the non-aqueous electrolytic solution at the time of charging and discharging cannot be ignored. On the other hand, if it is less than this range, long time and energy are required for manufacturing and equipment for preventing pollution, and the cost may increase.
[[圆形度]][[Circularity]]
本发明的锂二次电池中作为负极[2]的负极活性物质使用的无定形碳质的圆形度通常为0.1以上,优选为0.8以上,更优选为0.85以上,进一步优选为0.9以上。作为上限,圆形度为1时达到理论上的真球。如果低于该范围,则负极活性物质的填充性降低,粒子间的阻抗增大,有时短时间高电流密度充放电特性降低。The circularity of the amorphous carbonaceous used as the negative electrode active material of the negative electrode [2] in the lithium secondary battery of the present invention is usually 0.1 or more, preferably 0.8 or more, more preferably 0.85 or more, and even more preferably 0.9 or more. As an upper limit, a theoretical true sphere is achieved when the circularity is 1. If it is less than this range, the filling property of the negative electrode active material will decrease, the impedance between the particles will increase, and the short-time high current density charge and discharge characteristics may decrease.
本发明的圆形度以下式定义。The circularity in the present invention is defined by the following formula.
圆形度=(具有与粒子投影形状相同面积的等效圆的周长)/(粒子投影形状的实际周长)Circularity = (perimeter of an equivalent circle having the same area as the particle projected shape)/(actual perimeter of the particle projected shape)
作为圆形度的值使用如下的值:使用例如流动式粒子图像分析装置(例如,Sysmex Industrial公司制造的FPIA),将约0.2g试样分散于作为表面活性剂的聚氧乙烯(20)山梨糖醇酐单月桂酸酯的0.2质量%水溶液(约50mL)中,以60W的输出功率照射1分钟28kHz的超声波后,指定0.6~400μm为检测范围,对粒径3~40μm范围的粒子进行测定,并使用其测定的平均值。As the value of circularity, the following value is used: Using, for example, a flow type particle image analysis device (for example, FPIA manufactured by Sysmex Industrial Co., Ltd.), about 0.2 g of a sample is dispersed in polyoxyethylene (20) sorbitol as a surfactant. After irradiating a 0.2% by mass aqueous solution (approximately 50mL) of sugar-alcohol monolaurate with an output of 60W for 1 minute at 28kHz ultrasonic waves, specify a detection range of 0.6-400μm, and measure particles with a particle diameter of 3-40μm , and use its measured mean.
[[取向比]][[Orientation Ratio]]
本发明的锂二次电池中作为负极[2]的负极活性物质使用的无定形碳质的取向比通常为0.005以上,优选为0.01以上,更优选为0.015以上,其上限在理论上为0.67以下。如果低于该范围,则有时短时间高密度充放电特性降低。The orientation ratio of the amorphous carbonaceous used as the negative electrode active material of the negative electrode [2] in the lithium secondary battery of the present invention is usually 0.005 or more, preferably 0.01 or more, more preferably 0.015 or more, and its upper limit is theoretically 0.67 or less . If it is less than this range, short-time high-density charge and discharge characteristics may deteriorate.
取向比通过X射线衍射测定。使用非对称皮尔森VII作为分布函数,拟合通过X射线衍射得到的碳的(110)衍射和(004)衍射的峰,进行峰分离,分别计算出(110)衍射和(004)衍射的峰的积分强度。由得到的积分强度,计算出由(110)衍射积分强度/(004)衍射积分强度表示的比值,将该比值定义为活性物质取向比。The orientation ratio was measured by X-ray diffraction. Using asymmetric Pearson VII as a distribution function, fit the peaks of (110) diffraction and (004) diffraction of carbon obtained by X-ray diffraction, perform peak separation, and calculate the peaks of (110) diffraction and (004) diffraction respectively integral strength. From the obtained integrated intensity, a ratio represented by (110) diffraction integrated intensity/(004) diffraction integrated intensity was calculated, and this ratio was defined as the active material orientation ratio.
这里的X射线衍射测定条件如下。另外,“2θ”表示衍射角。The X-ray diffraction measurement conditions here are as follows. In addition, "2θ" represents a diffraction angle.
·靶:Cu(Kα射线)石墨单色光度仪Target: Cu (Kα ray) graphite monochromator
·狭缝:·Slit:
发散狭缝=1度,受光狭缝=0.1mm,散射狭缝=1度Divergence slit = 1 degree, light receiving slit = 0.1mm, scattering slit = 1 degree
·测定范围和步进角/测定时间・Measurement range and step angle/measurement time
(110)面:76.5度≤2θ≤78.5度0.01度/3秒(110) surface: 76.5 degrees ≤ 2θ ≤ 78.5 degrees 0.01 degrees / 3 seconds
(004)面:53.5度≤2θ≤56.0度0.01度/3秒(004) plane: 53.5 degrees ≤ 2θ ≤ 56.0 degrees 0.01 degrees / 3 seconds
[[长径比]][[Aspect Ratio]]
本发明的锂二次电池中作为负极[2]的负极活性物质使用的无定形碳质的长径比理论上为1以上,上限通常为10以下,优选为8以下,更优选为5以下。如果超过其上限,则在制成极板时有时会产生条纹、无法得到均匀的涂布面,短时间高电流密度充放电特性降低。The aspect ratio of the amorphous carbonaceous used as the negative electrode active material of the negative electrode [2] in the lithium secondary battery of the present invention is theoretically 1 or more, and the upper limit is usually 10 or less, preferably 8 or less, more preferably 5 or less. If the upper limit is exceeded, streaks may be generated when forming an electrode plate, a uniform coated surface may not be obtained, and the short-time high current density charge-discharge characteristics may deteriorate.
另外,长径比用三维观察时碳质材料粒子的最长径A和与其垂直的最短径B的比值A/B来表示。碳粒子的观察通过可以放大观察的扫描型电子显微镜来进行。任意选择固定在厚度为50μm以下的金属板的端面上的50个粒子,旋转、倾斜固定有试样的载物台,分别测定这些粒子的A、B,求出A/B的平均值。In addition, the aspect ratio is represented by the ratio A/B of the longest diameter A of the carbonaceous material particle and the shortest diameter B perpendicular thereto when viewed three-dimensionally. Observation of carbon particles was performed with a scanning electron microscope capable of magnified observation. Randomly select 50 particles fixed on the end face of a metal plate with a thickness of 50 μm or less, rotate and tilt the stage on which the sample is fixed, measure A and B of these particles, and calculate the average value of A/B.
另外,本发明的锂二次电池中作为负极[2]的负极活性物质使用中含有的无定形碳质优选选自以下(1)~(4)的无定形碳质。In addition, the amorphous carbonaceous substance contained in the lithium secondary battery of the present invention used as the negative electrode active material of the negative electrode [2] is preferably selected from the following amorphous carbonaceous substances (1) to (4).
(1)将选自煤类焦炭、石油类焦炭、炉黑、乙炔黑和沥青类碳纤维中的碳化物再进行热处理而得到的物质;(1) Substances obtained by heat-treating carbides selected from coal coke, petroleum coke, furnace black, acetylene black and pitch-based carbon fibers;
(2)将选自沥青原料、芳香族烃、N环化合物、S环化合物、聚苯、有机合成高分子、天然高分子、热塑性树脂和热固性树脂中的有机物和/或其热分解产物和/或进一步热处理而得到的物质;(2) organic matter selected from asphalt raw materials, aromatic hydrocarbons, N ring compounds, S ring compounds, polyphenylene, organic synthetic polymers, natural polymers, thermoplastic resins and thermosetting resins and/or thermal decomposition products thereof and/ or substances obtained by further heat treatment;
(3)将(2)的有机物溶解于低分子有机溶剂中而得到溶液的热分解物和/或进一步进行热处理而得到物质;(3) dissolving the organic matter of (2) in a low molecular organic solvent to obtain a thermal decomposition product of the solution and/or further heat-treating to obtain a substance;
(4)含有有机物的气体的碳化物。(4) Carbide of gas containing organic matter.
对于(2)来说,只要是可以碳化的物质即可,可以列举,沥青原料、苊烯、十环烯、蒽、菲等芳香族烃;吩嗪或吖啶等N环化合物;噻吩、联二噻吩等S环化合物;联苯、三联苯等聚苯;聚氯乙烯、聚乙烯醇、聚乙烯醇缩丁醛、它们的交联处理物、不溶解处理物、含氮的聚丙烯腈、聚吡咯等有机高分子;含硫的聚噻吩、聚苯乙烯等有机高分子;以纤维素、木质素、甘露聚糖、聚半乳糖醛酸、壳聚糖、蔗糖为代表的多糖类等天然高分子;聚苯硫醚、聚苯醚等热塑性树脂;糠醇树脂、酚醛树脂、酰亚胺树脂等热固性树脂;或者将它们溶解于苯、甲苯、二甲苯、喹啉、正己烷等低分子有机溶剂中而形成的溶液等有机物;含有这些有机物的可碳化的气体等。For (2), as long as it is a substance that can be carbonized, aromatic hydrocarbons such as pitch raw materials, acenaphthylene, decacyclene, anthracene, and phenanthrene; N-ring compounds such as phenazine or acridine; thiophene, bicycline, etc. S-ring compounds such as dithiophene; polyphenylenes such as biphenyl and terphenyl; polyvinyl chloride, polyvinyl alcohol, polyvinyl butyral, their cross-linked products, insoluble processed products, nitrogen-containing polyacrylonitrile, Polypyrrole and other organic polymers; sulfur-containing polythiophene, polystyrene and other organic polymers; polysaccharides represented by cellulose, lignin, mannan, polygalacturonic acid, chitosan, sucrose, etc. Natural polymers; thermoplastic resins such as polyphenylene sulfide and polyphenylene ether; thermosetting resins such as furfuryl alcohol resins, phenolic resins, and imide resins; Organic substances such as solutions formed in organic solvents; carbonizable gases containing these organic substances, etc.
这些当中,由于沥青原料的残炭率(残炭率)高,可以制作收率高的材料,故优选。另外,本说明书中,“沥青原料”是沥青和属于沥青的物质,是指可以通过适当的处理进行碳化或石墨化的物质。作为具体的沥青原料的例子,可以使用焦油、重油或沥青等。作为焦油的具体例子,可列举煤焦油、石油类焦油等。作为重油的具体例子,可以列举石油类重油的接触分解油、热分解油、常压残油、减压残油等。另外,作为沥青的具体例子,可列举煤焦油沥青、石油类沥青、合成沥青等。其中,煤焦油沥青的芳香性高,故优选。这些沥青原料可以单独使用任意一种,也可以以任意组合和比例组合使用2种以上。Among these, the pitch raw material is preferable because it can produce a high-yield material due to its high carbon residue ratio (carbon residue ratio). In addition, in this specification, a "pitch raw material" refers to pitch and a substance belonging to pitch, and refers to a substance that can be carbonized or graphitized by appropriate treatment. As an example of a specific pitch raw material, tar, heavy oil, pitch, etc. can be used. Specific examples of tar include coal tar, petroleum tar, and the like. Specific examples of the heavy oil include catalysis oil, pyrolysis oil, atmospheric residual oil, vacuum residual oil, and the like of petroleum heavy oil. In addition, specific examples of pitch include coal tar pitch, petroleum pitch, synthetic pitch, and the like. Among them, coal tar pitch is preferable because of its high aromaticity. Any one of these pitch raw materials may be used alone, or two or more of them may be used in combination in any desired ratio.
作为(3)的优选例子,可列举将上述(2)的有机物溶解于苯、甲苯、二甲苯、喹啉、正己烷等低分子有机溶剂中而得到溶液,再将该溶液的热分解物作为前体的碳化物。As a preferred example of (3), it is possible to enumerate a solution obtained by dissolving the above-mentioned organic substance in (2) in low-molecular organic solvents such as benzene, toluene, xylene, quinoline, and n-hexane, and then obtain a solution by thermally decomposing the solution as Precursor carbides.
关于(4),可列举甲烷、乙烷、丙烷、苯、乙炔、乙烯等烃化合物;一氧化碳等。Regarding (4), hydrocarbon compounds such as methane, ethane, propane, benzene, acetylene, and ethylene; carbon monoxide, and the like are exemplified.
优选进行交联处理。所谓交联处理,是为了将进行了交联处理的沥青原料等进行热处理得到的碳质材料进行难石墨化而进行的,通过实施这些处理,可以增加单位质量的充电容量。Preferably, a crosslinking treatment is performed. The so-called cross-linking treatment is performed to make the carbonaceous material obtained by heat-treating the cross-linked pitch raw material etc. difficult to graphitize, and the charge capacity per unit mass can be increased by carrying out these treatments.
作为交联处理的例子,可列举如下的处理方法:在二乙烯基苯、三乙烯基苯、邻苯二甲酸二烯丙酯、乙二醇二甲基丙烯酸酯、N,N-亚甲基双丙烯酰胺等乙烯基单体中使用作为自由基聚合引发剂的α,α’-偶氮二异丁腈(AIBN),过氧化苯甲酰(BPO)、过氧化月桂酰、氢过氧化异丙苯、氢过氧化叔丁基、过氧化氢等这样的交联处理;使用氧、臭氧、二氧化氮等氧化性气体,硫酸、硝酸、过氧化氢水溶液等氧化性液体而进行的氧化剂处理。作为交联处理的方法的一个例子,可以列举在使沥青原料的温度控制在50℃~400℃的状态下,与交联剂或氧化剂等混合再进行处理的方法。As an example of crosslinking treatment, the following treatment methods can be cited: in divinylbenzene, trivinylbenzene, diallyl phthalate, ethylene glycol dimethacrylate, N,N-methylene α,α'-azobisisobutyronitrile (AIBN), benzoyl peroxide (BPO), lauroyl peroxide, isohydroperoxide are used as radical polymerization initiators in vinyl monomers such as bisacrylamide Cross-linking treatment such as propylbenzene, tert-butyl hydroperoxide, hydrogen peroxide, etc.; oxidizing agent treatment using oxidizing gases such as oxygen, ozone, nitrogen dioxide, or oxidizing liquids such as sulfuric acid, nitric acid, and aqueous hydrogen peroxide . An example of a method of crosslinking treatment includes a method of treating the asphalt raw material after mixing with a crosslinking agent or an oxidizing agent while controlling the temperature of the pitch raw material at 50°C to 400°C.
[[Li-NMR移动]][[Li-NMR shift]]
如果在充电到满充电的状态下,对本发明的锂二次电池中所谓负极[2]的负极活性物质使用的无定形碳质进行7Li-NMR分析时,观察到了向基准物质LiCl的共振线的低磁场侧移动80~200ppm的主共振峰,那么为了增加每单位质量无定形碳质物质的容量,优选使用经过交联处理的无定形碳质物质。When the amorphous carbonaceous material used in the negative electrode active material of the so-called negative electrode [2] in the lithium secondary battery of the present invention is subjected to 7 Li-NMR analysis when it is charged to a fully charged state, a resonance line toward the reference material LiCl is observed. The main resonance peak shifted by 80 to 200ppm on the low magnetic field side of , so in order to increase the capacity per unit mass of the amorphous carbonaceous substance, it is preferable to use a cross-linked amorphous carbonaceous substance.
[[无定形碳质的制作方法]][[How to make amorphous carbon]]
上述的无定形碳质的制备方法只要在不超出本发明的要点的范围,则没有特别的限制,可以列举各种方法。在无定形碳质的制作中,必须经过1次热处理工序,但也可以将热处理分为2次以上,还优选在热处理的前后和/或热处理的中间阶段进行各种处理。所谓各种处理,可列举粉碎、分级、上述的交联处理等,粉碎、分级处理只要是固体状态则可以在热处理的前后、中间阶段的任意之一进行。交联处理优选在进行热处理前或中间阶段进行。通过进行这些处理,可以控制负极活性物质的比表面积,并且增加单位质量的容量。The method for producing the above-mentioned amorphous carbonaceous material is not particularly limited as long as it does not exceed the scope of the present invention, and various methods can be mentioned. In the production of amorphous carbon, one heat treatment step is necessary, but the heat treatment may be divided into two or more times, and it is also preferable to perform various treatments before and after the heat treatment and/or in the middle of the heat treatment. The various treatments include pulverization, classification, and the above-mentioned crosslinking treatment, etc., and the pulverization and classification treatment may be performed at any one of the before, after, and intermediate stages of the heat treatment as long as it is in a solid state. The crosslinking treatment is preferably performed before or during the heat treatment. By performing these treatments, the specific surface area of the negative electrode active material can be controlled, and the capacity per unit mass can be increased.
热处理前的粉碎中使用的装置没有特别的限制,例如,作为粗粉碎机,可以举出剪切式磨、颚式破碎机、冲击式压碎机、锥形压碎机等,作为中间粉碎机,可以举出辊式压碎机、锤式粉碎机等,作为微粉碎机,可以举出球磨、振动磨、销棒粉碎机、搅拌磨、喷射磨等。The device used in the pulverization before the heat treatment is not particularly limited, for example, as a coarse pulverizer, a shear mill, a jaw crusher, an impact crusher, a cone crusher, etc. can be mentioned, and as an intermediate pulverizer , include a roll crusher, a hammer mill, and the like, and examples of the fine pulverizer include a ball mill, a vibration mill, a pin mill, an agitator mill, and a jet mill.
作为原料的热处理中使用的装置,没有特别的限制,例如,可使用梭式炉(シヤトル炉)、隧道式炉(トンネル炉)、电炉、焦炭焙烧炉(リ一ドハンマ一炉)、回转炉、高压釜等反应器、coke(cokes制造的热处理槽)、直接通电炉等。处理原料时,根据需要可以进行搅拌。The apparatus used in the heat treatment of the raw material is not particularly limited, for example, a shuttle furnace (Shyatoll furnace), a tunnel furnace (Tonell furnace), an electric furnace, a coke roasting furnace (Ride Hanma furnace), a rotary kiln, Reactors such as autoclaves, cokes (heat treatment tanks manufactured by cokes), direct electric furnaces, etc. When processing raw materials, stirring can be performed as needed.
进行热处理时的温度条件没有特别的限制,通常为600℃以上,优选为900℃以上,另外,其上限通常为2500℃以下,优选为1300℃以下。温度条件低于上述范围时,结晶性变得过低,有可能增加不可逆容量。另一方面,如果超过其上限时,则结晶性变得过高,有可能使短时间高电流密度充放电特性降低。The temperature conditions for heat treatment are not particularly limited, but are usually 600°C or higher, preferably 900°C or higher, and the upper limit is usually 2500°C or lower, preferably 1300°C or lower. When the temperature condition is lower than the above range, the crystallinity becomes too low, which may increase the irreversible capacity. On the other hand, when the upper limit is exceeded, the crystallinity becomes too high, and there is a possibility that the short-time high current density charge and discharge characteristics may be degraded.
对于热处理后的无定形碳质,可以根据其块料或粒子的大小来进行粉碎或分级。粉碎使用的装置没有特别的限制,例如,作为粗粉碎机,可以举出剪切式磨、颚式破碎机、冲击式压碎机、锥形压碎机等,作为中间粉碎机,可以举出辊式压碎机、锤式粉碎机等,作为微粉碎机,可以举出,球磨、振动磨、销棒粉碎机、搅拌磨、喷射磨等。分级处理中使用的装置没有特别的限制,例如,在干式筛分的情况下,可以使用旋转式筛、摇动式筛、转动式筛(旋動式篩)、振动式筛等,在干式气流分级的情况下,可以使用重力式分级机、惯性力式分级机、离心力式分级机(分粒器、旋风分离器)等,另外,湿式筛分可以使用机械式湿式分级机、水力分级机、沉降分级机、离心式湿式分级机等。The heat-treated amorphous carbon can be pulverized or classified according to the size of its lumps or particles. The device used for pulverization is not particularly limited. For example, as a coarse pulverizer, shear mills, jaw crushers, impact crushers, cone crushers, etc. can be mentioned, and as intermediate pulverizers, there can be mentioned Roller crushers, hammer mills, etc., and examples of fine pulverizers include ball mills, vibration mills, pin mills, stirring mills, and jet mills. The device used in the classification process is not particularly limited. For example, in the case of dry sieving, a rotary sieve, a shaking sieve, a rotary sieve (rotary sieve), a vibrating sieve, etc. can be used. In the case of airflow classification, gravity classifiers, inertial force classifiers, centrifugal force classifiers (classifiers, cyclone separators), etc. can be used. In addition, mechanical wet classifiers and hydraulic classifiers can be used for wet sieving. , sedimentation classifier, centrifugal wet classifier, etc.
[制作负极[2]的电极][Making the electrode of the negative electrode [2]]
负极[2]的制造可以采用通常的方法,可以与上述同样地形成负极[2]。集电体、集电体和活性物质层的厚度比、电极密度、粘合剂、极板取向比、阻抗等也与上述相同。The negative electrode [2] can be produced by a common method, and the negative electrode [2] can be formed in the same manner as above. The current collector, the thickness ratio between the current collector and the active material layer, electrode density, binder, plate orientation ratio, impedance, and the like are also the same as described above.
<负极[3]><Negative pole [3]>
下面,对本发明的锂二次电池中使用的负极[3]进行说明,该负极含有金属氧化物作为负极活性物质,所述金属氧化物含有能够吸留和放出锂的钛。Next, the negative electrode [3] used in the lithium secondary battery of the present invention will be described. The negative electrode contains, as a negative electrode active material, a metal oxide containing titanium capable of occluding and releasing lithium.
[负极[3]的负极活性物质][Negative electrode active material of the negative electrode [3]]
下面,对负极[3]中使用的负极活性物质进行说明。Next, the negative electrode active material used in the negative electrode [3] will be described.
[[负极活性物质的构成]][[Composition of negative electrode active material]]
本发明的锂二次电池的负极[3]中使用的负极活性物质含有金属氧化物,所述金属氧化物含有能够吸留和放出锂的钛。在金属氧化物中,优选锂和钛的复合氧化物(以下,简称为“锂钛复合氧化物”),另外,该金属氧化物优选具有尖晶石结构的含钛金属氧化物。另外,在锂二次电池用负极活性物质中使用同时满足这些条件的金属氧化物,也就是含有具有尖晶石结构的锂钛复合氧化物时,可以大幅降低输出阻抗,故特别优选。The negative electrode active material used in the negative electrode [3] of the lithium secondary battery of the present invention contains a metal oxide containing titanium capable of occluding and releasing lithium. Among the metal oxides, a composite oxide of lithium and titanium (hereinafter, simply referred to as "lithium-titanium composite oxide") is preferable, and the metal oxide is preferably a titanium-containing metal oxide having a spinel structure. In addition, when a metal oxide satisfying these conditions is used in the negative electrode active material for a lithium secondary battery, that is, when a lithium-titanium composite oxide having a spinel structure is used, the output impedance can be greatly reduced, so it is particularly preferable.
另外,优选锂钛复合氧化物中的锂或钛被其他金属元素,例如选自Na、K、Co、Al、Fe、Ti、Mg、Cr、Ga、Cu、Zn和Nb中的至少一种元素置换。In addition, it is preferable that lithium or titanium in the lithium-titanium composite oxide is replaced by other metal elements, such as at least one element selected from Na, K, Co, Al, Fe, Ti, Mg, Cr, Ga, Cu, Zn, and Nb. replacement.
该金属氧化物是通式(1)表示的锂钛复合氧化物,从锂离子的掺杂/脱掺杂时的结构稳定来看,优选在通式(1)中,0.7≤x≤1.5、1.5≤y≤2.3、0≤z≤1.6。The metal oxide is a lithium-titanium composite oxide represented by the general formula (1), and from the viewpoint of structural stability during doping/dedoping of lithium ions, it is preferable that in the general formula (1), 0.7≤x≤1.5, 1.5≤y≤2.3, 0≤z≤1.6.
LixTiyMzO4 (1)Li x Ti y M z O 4 (1)
[通式(1)中,M表示选自Na、K、Co、Al、Fe、Ti、Mg、Cr、Ga、Cu、Zn和Nb中的至少一种元素]。[In general formula (1), M represents at least one element selected from Na, K, Co, Al, Fe, Ti, Mg, Cr, Ga, Cu, Zn and Nb].
上述通式(1)表示的组成中,以下所示结构由于电池性能的平衡良好,故特别优选。Among the compositions represented by the above-mentioned general formula (1), the structures shown below are particularly preferable because the balance of battery performance is good.
在通式(1)LixTiyMzO4中,In the general formula (1) Li x Ti y M z O 4 ,
(a)1.2≤x≤1.4、1.5≤y≤1.7、z=0(a) 1.2≤x≤1.4, 1.5≤y≤1.7, z=0
(b)0.9≤x≤1.1、1.9≤y≤2.1、z=0(b) 0.9≤x≤1.1, 1.9≤y≤2.1, z=0
(c)0.7≤x≤0.9、2.1≤y≤2.3、z=0(c) 0.7≤x≤0.9, 2.1≤y≤2.3, z=0
上述化合物的特别优选的代表的组成为:(a)为Li4/3Ti5/3O4、(b)为Li1Ti2O4、(c)为Li4/5Ti11/5O4。A particularly preferred representative composition of the above compound is: (a) Li 4/3 Ti 5/3 O 4 , (b) Li 1 Ti 2 O 4 , (c) Li 4/5 Ti 11/5 O 4 .
另外,对于Z≠0的结构,例如可列举Li4/3Ti4/3Al1/3O4作为优选的组成。In addition, regarding the structure of Z≠0, for example, Li 4/3 Ti 4/3 Al 1/3 O 4 can be cited as a preferable composition.
[[负极活性物质的物性、形状等]][[Physical properties, shape, etc. of the negative electrode active material]]
本发明的锂二次电池的负极[3]中使用的负极活性物质优选除了上述要件以外,还满足下述物性中的至少一种。另外,特别优选除了上述要件以外,同时满足下述物性中的至少2种以上。The negative electrode active material used in the negative electrode [3] of the lithium secondary battery of the present invention preferably satisfies at least one of the following physical properties in addition to the above requirements. In addition, it is particularly preferable to simultaneously satisfy at least two or more of the following physical properties in addition to the above requirements.
[[[BET比表面积]]][[[BET specific surface area]]]
本发明的锂二次电池中的负极[3]的负极活性物质使用的含有钛的金属氧化物通过BET法测定的比表面积优选为0.5m2/g以上,更优选为0.7m2/g以上,特别优选为1.0m2/g以上,进一步优选为1.5m2/g以上。其上限优选为200m2/g以下,更优选为100m2/g以下,特别优选为50m2/g以下,进一步优选为25m2/g以下。BET比表面积的值如果低于该范围,则在作为负极材料使用的情况下,与非水电解液接触的反应面积减少,输出阻抗有时增加。另一方面,BET比表面积的值如果超过该范围,则含有钛的金属氧化物的结晶表面或端面部分增加,并且由此还产生结晶的不均(歪),不可逆容量变得不能忽视,有时得不到优选的电池。The specific surface area of the titanium-containing metal oxide used for the negative electrode active material of the negative electrode [3] in the lithium secondary battery of the present invention as measured by the BET method is preferably 0.5 m 2 /g or more, more preferably 0.7 m 2 /g or more , particularly preferably at least 1.0 m 2 /g, more preferably at least 1.5 m 2 /g. The upper limit thereof is preferably 200 m 2 /g or less, more preferably 100 m 2 /g or less, particularly preferably 50 m 2 /g or less, further preferably 25 m 2 /g or less. If the value of the BET specific surface area is below this range, when used as a negative electrode material, the reaction area in contact with the non-aqueous electrolytic solution decreases, and the output impedance may increase. On the other hand, if the value of the BET specific surface area exceeds this range, the crystal surface or end face portion of the metal oxide containing titanium will increase, and thus crystal unevenness (distortion) will occur, and the irreversible capacity will become unnegligible, sometimes No preferred battery was obtained.
BET比表面积定义为如下的值:使用表面积计(例如,大仓理研制造的全自动表面积测定装置),在氮气流通、350℃下对试样进行15分钟预干燥,然后,使用氮气相对于大气压的相对压力值准确调整为0.3的氮氦混合气体,通过采用气体流动法的氮吸附BET1点法测定的值。The BET specific surface area is defined as a value as follows: Using a surface area meter (for example, a fully automatic surface area measuring device manufactured by Okura Riken), the sample is pre-dried at 350° C. for 15 minutes under nitrogen flow, and then, using nitrogen gas relative to atmospheric pressure The relative pressure value is accurately adjusted to 0.3 of the nitrogen-helium mixed gas, the value measured by the nitrogen adsorption BET1 point method using the gas flow method.
[[[体积平均粒径]]][[[Volume Average Particle Size]]]
本发明的锂二次电池中作为负极[3]的负极活性物质使用的含有钛的金属氧化物的体积平均粒径(一次粒子凝聚而形成二次粒子时,为二次粒径)定义为通过激光衍射/散射法求得的体积基准的平均粒径(中值粒径),优选为0.1μm以上,更优选为0.5μm以上,进一步优选为0.7μm以上。另外,其上限通常为50μm以下,优选为40μm以下,更优选为30μm以下,进一步优选为25μm以下。如果低于上述范围,则在制作电极时需要大量的粘合剂,结果有时电池容量降低。另外,如果超过上述范围,则制成电极的极板时,容易形成不均匀的涂布面,有时在电池制作工序上是不优选的。In the lithium secondary battery of the present invention, the volume average particle diameter of the metal oxide containing titanium used as the negative electrode active material of the negative electrode [3] (when the primary particles are aggregated to form secondary particles, it is the secondary particle diameter) is defined by The volume-based average particle size (median particle size) obtained by the laser diffraction/scattering method is preferably 0.1 μm or more, more preferably 0.5 μm or more, and still more preferably 0.7 μm or more. In addition, the upper limit thereof is usually 50 μm or less, preferably 40 μm or less, more preferably 30 μm or less, and still more preferably 25 μm or less. If it is less than the above-mentioned range, a large amount of binder is required when producing an electrode, and as a result, the battery capacity may decrease. In addition, when the above range is exceeded, an uneven coating surface is likely to be formed when forming an electrode plate, which may be unfavorable in the battery production process.
[[[平均一次粒径]]][[[Average primary particle size]]]
一次粒子凝聚而形成二次粒子时,作为用作本发明的锂二次电池的负极活性物质的含有钛的金属氧化物的平均一次粒径优选为0.01μm以上,更优选为0.05μm以上,进一步优选为0.1μm以上,最优选为0.2μm以上,其上限优选为2μm以下,更优选为1.6μm以下,进一步优选为1.3[μm以下,最优选为1μm以下。如果平均一次粒径超过上述上限,则难以形成球状的二次粒子,对粉末填充性带来不良影响,或者比表面积大幅降低,因此,输出特性等电池性能降低的可能性有时变高。相反,如果平均一次粒径低于上述下限,则通常由于结晶不完全,因此有时产生充放电的可逆性差等问题。When the primary particles are aggregated to form secondary particles, the average primary particle size of the titanium-containing metal oxide used as the negative electrode active material of the lithium secondary battery of the present invention is preferably 0.01 μm or more, more preferably 0.05 μm or more, and further preferably 0.05 μm or more. It is preferably 0.1 μm or more, most preferably 0.2 μm or more, and the upper limit thereof is preferably 2 μm or less, more preferably 1.6 μm or less, still more preferably 1.3 μm or less, and most preferably 1 μm or less. When the average primary particle size exceeds the above upper limit, it is difficult to form spherical secondary particles, which adversely affects the powder fillability, or the specific surface area is greatly reduced, and therefore battery performance such as output characteristics may decrease. On the contrary, if the average primary particle diameter is less than the above-mentioned lower limit, problems such as poor charge-discharge reversibility may occur due to incomplete crystallization.
一次粒径可通过使用扫描电子显微镜(SEM)的观察来测定。具体地,通过如下方法求出:在倍率10000~100000倍的照片中,对任意的50个一次粒子求出水平方向的直线上的由一次粒子的左右边界线产生的切片的最长值,并取其平均值。The primary particle size can be measured by observation using a scanning electron microscope (SEM). Specifically, it is obtained by the following method: in a photograph with a magnification of 10,000 to 100,000 times, for any 50 primary particles, the longest value of the slice generated by the left and right boundary lines of the primary particles on a straight line in the horizontal direction is obtained, and Take its average.
[[[形状]]][[[shape]]]
本发明的锂二次电池的负极[3]中使用的该含有钛的金属氧化物的粒子形状可使用以往使用的块状、多面体状、球状、椭圆球状、板状、针状、柱状等,其中,优选一次粒子凝聚而形成二次粒子,且该二次粒子的形状为球状或椭圆球状。通常,电化学元件随着其充放电,电极中的活性物质膨胀收缩,因此容易发生由于该应力引起的活性物质的破坏或导电通路切断等劣化。因此,与只是一次粒子的单一粒子活性物质相比,优选一次粒子凝聚而形成二次粒子的情况,因为形成二次粒子可以缓和膨胀收缩的应力,从而防止劣化。另外,与板状等轴取向性的粒子相比,优选球状或椭圆球状的粒子,因为球状或椭圆球状的粒子在电极成型时的取向少,充放电时的电极的膨胀收缩也少,并且在制作电极时与导电剂的混合时,也容易均匀混合。The particle shape of the titanium-containing metal oxide used in the negative electrode [3] of the lithium secondary battery of the present invention can be a block shape, a polyhedron shape, a spherical shape, an ellipsoidal shape, a plate shape, a needle shape, a columnar shape, etc., which have been used in the past, Among them, it is preferable that the primary particles are aggregated to form secondary particles, and the shape of the secondary particles is spherical or ellipsoidal. In general, the active material in the electrode expands and shrinks as the electrochemical element is charged and discharged, and thus degradation of the active material due to this stress, such as destruction of the active material or interruption of the conductive path, tends to occur. Therefore, it is preferable to agglomerate primary particles to form secondary particles rather than single-particle active material with only primary particles, because the formation of secondary particles can relieve the stress of expansion and contraction, thereby preventing deterioration. In addition, spherical or ellipsoidal particles are preferred over plate-like equiaxially oriented particles, because spherical or ellipsoidal particles have less orientation during electrode molding and less expansion and contraction of the electrode during charging and discharging, and in It is also easy to mix uniformly with the conductive agent when making electrodes.
[[[振实密度]]][[[Tap Density]]]
本发明的锂二次电池中的作为负极[3]的负极活性物质使用的含有钛的金属氧化物的振实密度优选为0.05g/cm3以上,更优选为0.1g/cm3以上,进一步优选为0.2g/cm3以上,特别优选为0.4g/cm3以上。另外,其上限优选为2.8g/cm3以下,更优选为2.4g/cm3以下,特别优选为2g/cm3以下。如果振实密度低于该范围,则作为负极使用时难以提高填充密度,并且粒子间的接触面积减少,因此粒子间的阻抗增加,有时会增加输出电阻。另一方面,如果超过该范围,则电极中的粒子间的空隙过少,非水电解液的流路会减少,因此有时会增加输出电阻。The tap density of the titanium-containing metal oxide used as the negative electrode active material of the negative electrode [3] in the lithium secondary battery of the present invention is preferably 0.05 g/cm 3 or more, more preferably 0.1 g/cm 3 or more, and furthermore Preferably it is 0.2 g/cm 3 or more, particularly preferably 0.4 g/cm 3 or more. In addition, the upper limit thereof is preferably 2.8 g/cm 3 or less, more preferably 2.4 g/cm 3 or less, particularly preferably 2 g/cm 3 or less. If the tap density is lower than this range, it will be difficult to increase the packing density when used as a negative electrode, and the contact area between particles will decrease, so the impedance between particles will increase, and the output resistance may increase. On the other hand, if it exceeds this range, the gaps between the particles in the electrode will be too small, and the flow path of the non-aqueous electrolyte solution will be reduced, which may increase the output resistance.
在本发明中,振实密度如下定义:使试样通过孔径300μm的筛子,使试样落入20cm3的振实容器中,到试样充满容器的上端面后,使用粉末密度测定器(例如,seishin企业公司制造的Tap densor),进行1000次冲程长度10mm的振动,将由此时的体积和试样的重量求出的密度作为振实密度。In the present invention, the tap density is defined as follows: the sample is passed through a sieve with an aperture of 300 μm, the sample is dropped into a 20 cm container for vibration, and after the sample is filled with the upper end of the container, a powder density measuring device (such as , Tap densor manufactured by Seishin Enterprise Co., Ltd.), vibration with a stroke length of 10 mm was performed 1000 times, and the density obtained from the volume at that time and the weight of the sample was taken as the tap density.
[[[圆形度]]][[[Circularity]]]
本发明的锂二次电池中作为负极[3]的负极活性物质使用的含有钛的金属氧化物的圆形度通常为0.10以上,优选为0.80以上,更优选为0.85以上,进一步优选为0.90以上。作为上限,圆形度为1时成为理论上的真球。如果低于该范围,则负极活性物质的填充性降低,粒子间的阻抗增大,有时短时间高电流密度充放电特性降低。The circularity of the titanium-containing metal oxide used as the negative electrode active material of the negative electrode [3] in the lithium secondary battery of the present invention is usually 0.10 or more, preferably 0.80 or more, more preferably 0.85 or more, and still more preferably 0.90 or more . As an upper limit, when the circularity is 1, it becomes a theoretical true sphere. If it is less than this range, the filling property of the negative electrode active material will decrease, the impedance between the particles will increase, and the short-time high current density charge and discharge characteristics may decrease.
本发明的圆形度以下式定义。The circularity in the present invention is defined by the following formula.
圆形度=(具有与粒子投影形状相同面积的等效圆的周长)/(粒子投影形状的实际周长)Circularity = (perimeter of an equivalent circle having the same area as the particle projected shape)/(actual perimeter of the particle projected shape)
作为圆形度的值使用如下的值:使用例如流动式粒子图像分析装置(例如,Sysmex Industrial公司制造的FPIA),将约0.2g试样分散于作为表面活性剂的聚氧乙烯(20)山梨糖醇酐单月桂酸酯的0.2质量%水溶液(约50mL)中,以60W的输出功率照射1分钟28kHz的超声波后,指定0.6~400μm为检测范围,对粒径3~40μm范围的粒子进行测定而得到的值。As the value of circularity, the following value is used: Using, for example, a flow type particle image analysis device (for example, FPIA manufactured by Sysmex Industrial Co., Ltd.), about 0.2 g of a sample is dispersed in polyoxyethylene (20) sorbitol as a surfactant. After irradiating a 0.2% by mass aqueous solution (approximately 50mL) of sugar-alcohol monolaurate with an output of 60W for 1 minute at 28kHz ultrasonic waves, specify a detection range of 0.6-400μm, and measure particles with a particle diameter of 3-40μm And get the value.
[[[长径比]]][[[Aspect Ratio]]]
本发明的锂二次电池中作为负极[3]的负极活性物质使用的含有钛的金属氧化物的长径比理论上为1以上,上限为5以下,优选为4以下,更优选为3以下,进一步优选为2以下。如果超过其上限,则在制成极板时有时会产生条纹、无法得到均匀的涂布面,短时间高电流密度充放电特性降低。In the lithium secondary battery of the present invention, the aspect ratio of the titanium-containing metal oxide used as the negative electrode active material of the negative electrode [3] is theoretically 1 or more, the upper limit is 5 or less, preferably 4 or less, more preferably 3 or less , and more preferably 2 or less. If the upper limit is exceeded, streaks may be generated when forming an electrode plate, a uniform coated surface may not be obtained, and the short-time high current density charge-discharge characteristics may deteriorate.
另外,将三维观察时粒子的最长径设定为A、并将与其垂直的最短径设定为B时,长径比用A/B来表示。粒子的观察通过可以放大观察的扫描型电子显微镜来进行。任意选择固定在厚度为50μm以下的金属板的端面上的50个石墨粒子,旋转、倾斜固定有试样的载物台,分别测定这些粒子的A、B,求出A/B的平均值。In addition, when A is the longest diameter of the particles in three-dimensional observation, and B is the shortest diameter perpendicular thereto, the aspect ratio is represented by A/B. Observation of particles is performed with a scanning electron microscope capable of magnified observation. Randomly select 50 graphite particles fixed on the end face of a metal plate with a thickness of 50 μm or less, rotate and tilt the stage on which the sample is fixed, measure A and B of these particles, and obtain the average value of A/B.
[[负极活性物质的制造法]][[Manufacturing method of negative electrode active material]]
作为本发明的锂二次电池中的负极[3]的负极活性物质的制造法,在不超过本发明要旨的范围内没有特别限制,可列举几种方法,可以使用作为无机化合物的制造法的通常的方法。例如可列举将氧化钛等钛原料物质、根据需要使用的其他元素的原料物质和LiOH、Li2CO3、LiNO3等Li源均匀混合,在高温下烧结而得到活性物质的方法。特别是在制作球状或椭圆球状的活性物质时,可考虑各种方法,例如可列举如下方法:将氧化钛等钛原料物质、根据需要使用的其他元素的原料物质溶解或粉碎分散在水等溶剂中,边搅拌边调节pH,制作并获得球状的前体,根据需要将其干燥后,加入LiOH、Li2CO3、LiNO3等Li源,在高温下烧结而得到活性物质的方法;将氧化钛等钛原料物质、根据需要使用的其他元素的原料物质溶解或粉碎分散在水等溶剂中,再通过喷雾干燥器等将其干燥成型,制成球状或椭圆球状的前体,然后在其中加入LiOH、Li2CO3、LiNO3等Li源,在高温下烧结而得到活性物质的方法;以及将氧化钛等钛原料物质和LiOH、Li2CO3、LiNO3等Li源以及根据需要使用的其他元素的原料物质溶解或粉碎分散在水等溶剂中,再通过喷雾干燥器等将其干燥成型,制成球状或椭圆球状的前体,然后在高温下将其烧结而得到活性物质的方法等。The method for producing the negative electrode active material of the negative electrode [3] in the lithium secondary battery of the present invention is not particularly limited within the scope not exceeding the gist of the present invention, and several methods can be cited, and the method for producing an inorganic compound can be used. the usual way. For example, a method of uniformly mixing a titanium raw material such as titanium oxide, raw materials of other elements used as needed, and a Li source such as LiOH, Li 2 CO 3 , LiNO 3 , and sintering at a high temperature to obtain an active material is mentioned. In particular, when producing a spherical or ellipsoidal active material, various methods are conceivable, for example, the method of dissolving or pulverizing a titanium raw material such as titanium oxide and, if necessary, raw materials of other elements in a solvent such as water In the process, adjusting the pH while stirring, making and obtaining spherical precursors, drying them as needed, adding Li sources such as LiOH, Li 2 CO 3 , LiNO 3 , and sintering at high temperatures to obtain active materials; Titanium raw materials such as titanium and raw materials of other elements used as needed are dissolved or pulverized in solvents such as water, and then dried and shaped by spray dryers to form spherical or ellipsoidal precursors, and then added Li sources such as LiOH, Li 2 CO 3 , LiNO 3 , and a method for obtaining an active material by sintering at a high temperature ; The raw materials of other elements are dissolved or pulverized and dispersed in a solvent such as water, and then dried and shaped by a spray dryer to form a spherical or ellipsoidal precursor, and then sintered at a high temperature to obtain an active material, etc. .
另外,在这些工序中,还可以在含有钛的金属氧化物结构中和/或以与含有钛的氧化物接触的形式存在除Ti以外的元素,例如Al、Mn、Ti、V、Cr、Mn、Fe、Co、Li、Ni、Cu、Zn、Mg、Ga、Zr、C、Si、Sn、Ag。通过含有这些元素,能够控制电池的工作电压、容量。In addition, during these steps, elements other than Ti, such as Al, Mn, Ti, V, Cr, Mn, may also be present in the structure of the titanium-containing metal oxide and/or in contact with the titanium-containing oxide. , Fe, Co, Li, Ni, Cu, Zn, Mg, Ga, Zr, C, Si, Sn, Ag. By containing these elements, the operating voltage and capacity of the battery can be controlled.
[制作负极[3]的电极][Making the electrode of the negative electrode [3]]
负极[3]的制造可以采用通常的方法,可以与上述同样地形成负极[3]。集电体、集电体和活性物质层的厚度比、电极密度、粘合剂、极板取向比、阻抗等也与上述相同。The negative electrode [3] can be produced by a common method, and the negative electrode [3] can be formed in the same manner as above. The current collector, the thickness ratio between the current collector and the active material layer, electrode density, binder, plate orientation ratio, impedance, and the like are also the same as described above.
<负极[4]><Negative pole [4]>
下面,对本发明的锂二次电池中使用的负极[4]进行说明,该负极含有碳质物质作为负极活性物质,所述碳质物质的圆形度为0.85以上、表面官能团量O/C值为0~0.01。Next, the negative electrode [4] used in the lithium secondary battery of the present invention will be described. The negative electrode contains a carbonaceous substance as the negative electrode active material, the circularity of the carbonaceous substance is 0.85 or more, the surface functional group content O/C value 0 to 0.01.
[负极[4]的负极活性物质][Negative electrode active material of negative electrode [4]]
下面,对负极[4]中使用的负极活性物质进行说明。Next, the negative electrode active material used in the negative electrode [4] will be described.
本发明的锂二次电池的负极[4]中使用的负极活性物质至少含有满足以下条件(a)和(b)的碳质物质。The negative electrode active material used in the negative electrode [4] of the lithium secondary battery of the present invention contains at least a carbonaceous material satisfying the following conditions (a) and (b).
(a)圆形度为0.85以上;(a) The circularity is above 0.85;
(b)表面官能团量O/C值为0~0.01。(b) O/C value of surface functional group amount is 0-0.01.
下面,对本发明中使用的碳质物质的详细情况进行说明。Next, details of the carbonaceous substance used in the present invention will be described.
[[圆形度]][[Circularity]]
碳质物质的圆形度通常为0.85以上,优选为0.87以上,更优选为0.89以上,特别优选为0.92以上。作为上限,圆形度为1时成为理论上的真球。如果低于该范围,则负极活性物质的填充性降低,负极电极的压实变得困难,压实时导致粒子的破坏,粒子内部在低充电深度下的高温保存耐性差的表面可能容易露出。The circularity of the carbonaceous material is usually not less than 0.85, preferably not less than 0.87, more preferably not less than 0.89, particularly preferably not less than 0.92. As an upper limit, when the circularity is 1, it becomes a theoretical true sphere. If it is lower than this range, the filling property of the negative electrode active material will decrease, the compaction of the negative electrode electrode will become difficult, and the particles will be destroyed during compaction, and the surface of the particle interior that is poor in high temperature storage resistance at a low charge depth may be easily exposed.
本发明中所说的圆形度以下式定义。The circularity referred to in the present invention is defined by the following formula.
圆形度=(具有与粒子投影形状相同面积的等效圆的周长)/(粒子投影形状的实际周长)Circularity = (perimeter of an equivalent circle having the same area as the particle projected shape)/(actual perimeter of the particle projected shape)
作为圆形度的值使用如下的值:使用例如流动式粒子图像分析装置(例如,Sysmex Industrial公司制造的FPIA),将约0.2g试样分散于作为表面活性剂的聚氧乙烯(20)山梨糖醇酐单月桂酸酯的0.2质量%水溶液(约50mL)中,以60W的输出功率照射1分钟28kHz的超声波后,指定0.6~400μm为检测范围,对粒径3~40μm范围的粒子进行测定而得到的值。As the value of circularity, the following value is used: Using, for example, a flow type particle image analysis device (for example, FPIA manufactured by Sysmex Industrial Co., Ltd.), about 0.2 g of a sample is dispersed in polyoxyethylene (20) sorbitol as a surfactant. After irradiating a 0.2% by mass aqueous solution (approximately 50mL) of sugar-alcohol monolaurate with an output of 60W for 1 minute at 28kHz ultrasonic waves, specify a detection range of 0.6-400μm, and measure particles with a particle diameter of 3-40μm And get the value.
[[O/C值]][[O/C value]]
碳质物质的表面官能团量O/C值必须为0~0.01。O/C值的上限优选为0.005以下,越接近于0越优选。The O/C value of the amount of surface functional groups of the carbonaceous substance must be 0-0.01. The upper limit of the O/C value is preferably 0.005 or less, and the closer to 0, the more preferable.
本发明中所说的O/C值是使用X射线光电分光法(XPS)测定的表面官能团量之比,表面官能团量O/C值如果超过上述范围,则粒子表面的官能团量增加,在存在特定化合物的条件下进行充电时,表面生成的SEI覆盖膜的稳定性不充分,有时导致低充电深度下的高温保存特性的降低。The O/C value mentioned in the present invention is the ratio of the amount of surface functional groups measured by X-ray photoelectric spectroscopy (XPS). If the O/C value of the amount of surface functional groups exceeds the above range, the amount of functional groups on the particle surface will increase. When charging is performed under the condition of a specific compound, the stability of the SEI coating film formed on the surface is insufficient, which may lead to a decrease in high-temperature storage characteristics at a low charge depth.
表面官能团量O/C值表示石墨材料等的表面中存在的氧原子的摩尔浓度与碳原子的摩尔浓度的比例,是表示羧基、酚性羟基、羰基等官能团在表面的存在量的指标。表面官能团量O/C值大的碳材料大多在粒子面碳的微晶端面等结合有表面含氧官能团。另外,作为石墨材料的表面官能团量O/C值,使用如下的值:在X射线光电分光法分析中,求出C1s和O1s的光谱的峰面积,由此计算出C和O的原子浓度比(O原子浓度/C原子浓度)。具体的测定程序没有特别限制,作为其实例,如下所述。The surface functional group amount O/C value represents the ratio of the molar concentration of oxygen atoms present on the surface of graphite materials and the like to the molar concentration of carbon atoms, and is an index representing the amount of functional groups such as carboxyl groups, phenolic hydroxyl groups, and carbonyl groups present on the surface. In many carbon materials having a large O/C value of surface functional groups, surface oxygen-containing functional groups are bonded to the crystallite end faces of the particle surface carbon. In addition, as the O/C value of the surface functional group amount of the graphite material, the following value is used: In the X-ray photoelectric spectrometry analysis, the peak area of the spectrum of C1s and O1s is obtained, and the atomic concentration ratio of C and O is calculated from this. (O atomic concentration/C atomic concentration). Specific measurement procedures are not particularly limited, and examples thereof are as follows.
即,使用X射线光电分光器(例如,ulvac-phi(アルバツク·フアイ)公司制造的ESCA)作为X射线光电分光法测定,将测定对象(这里为石墨材料)放置在试料台上并使表面平坦,以铝的Kα射线作为X射线源,通过多路复用测定来测定C1s(280~300eV)和O1s(525~545eV)的光谱。将得到的C1s的峰顶设定为284.3eV进行带电补偿,求出C1s和O1s的光谱的峰面积,再乘以装置感度系数,分别计算出C和O的表面原子浓度。计算出得到的该O和C的原子浓度比O/C(O原子浓度/C原子浓度),将其定义为石墨材料的表面官能团量O/C值。That is, using an X-ray photoelectric spectrometer (for example, ESCA manufactured by ulvac-phi (アルバツク·フアイ) company) as the X-ray photoelectric spectrometry measurement, the measurement object (here, graphite material) is placed on the sample stand and the surface It is flat, and the spectra of C1s (280-300eV) and O1s (525-545eV) are measured by multiplex measurement with aluminum Kα rays as the X-ray source. Set the obtained peak top of C1s to 284.3eV for charging compensation, calculate the peak areas of C1s and O1s spectra, and multiply by the device sensitivity coefficient to calculate the surface atomic concentrations of C and O, respectively. The calculated atomic concentration ratio O/C of O and C (O atomic concentration/C atomic concentration) is defined as the surface functional group amount O/C value of the graphite material.
本发明中使用的碳质物质满足上述的“圆形度”和“表面官能团量O/C值”的条件,但从电池的平衡这点来看,更优选进一步同时满足下述项目中的任一项或多项的条件。这些当中,优选同时满足振实密度、拉曼R值和体积平均粒径中的任一项或多项的条件。The carbonaceous substance used in the present invention satisfies the above-mentioned conditions of "circularity" and "surface functional group amount O/C value", but from the viewpoint of battery balance, it is more preferable to further satisfy any of the following items at the same time One or more conditions. Among these, it is preferable to simultaneously satisfy the conditions of any one or more of tap density, Raman R value, and volume average particle diameter.
[[振实密度]][[Tap Density]]
碳质物质的振实密度通常为0.55g/cm3以上,优选为0.7g/cm3以上,更优选为0.8g/cm3以上,特别优选为0.9g/cm3以上。另外,其上限优选为2g/cm3以下,更优选为1.8g/cm3以下,特别优选为1.6g/cm3以下。如果振实密度低于该范围,则作为负极使用时难以提高填充密度,有时不能得到高容量的电池。另一方面,如果超过该范围,则电极中的粒子间的空隙过少,难以确保粒子间的导电性,有时难以获得优选的电池特性。The tap density of the carbonaceous material is usually 0.55 g/cm 3 or more, preferably 0.7 g/cm 3 or more, more preferably 0.8 g/cm 3 or more, particularly preferably 0.9 g/cm 3 or more. In addition, the upper limit thereof is preferably 2 g/cm 3 or less, more preferably 1.8 g/cm 3 or less, particularly preferably 1.6 g/cm 3 or less. When the tap density is lower than this range, it is difficult to increase the packing density when used as a negative electrode, and a high-capacity battery may not be obtained. On the other hand, if it exceeds this range, the gaps between the particles in the electrode will be too small, making it difficult to ensure the conductivity between the particles, and it may be difficult to obtain preferable battery characteristics.
在本发明中,振实密度如下定义:使试样通过孔径300μm的筛子,使试样落入20cm3的振实容器中,到试样充满容器的上端面后,使用粉末密度测定器(例如,seishin企业公司制造的Tap densor),进行1000次冲程长度10mm的振动,由此时的体积和重量计算密度,将该值作为振实密度。In the present invention, the tap density is defined as follows: the sample is passed through a sieve with an aperture of 300 μm, the sample is dropped into a 20 cm container for vibration, and after the sample is filled with the upper end of the container, a powder density measuring device (such as , Tap densor manufactured by Seishin Enterprise Co., Ltd.) was vibrated 1000 times with a stroke length of 10 mm, and the density was calculated from the volume and weight at this time, and this value was taken as the tap density.
[[拉曼R值、半值宽度]][[Raman R value, half value width]]
使用氩离子激光拉曼光谱法测定的碳质物质的R值通常为0.001以上,优选为0.01以上,其上限通常为0.2以下,优选为0.18以下,更优选为0.15以下的范围。如果R值低于该范围,则粒子表面的结晶性过高,随着充放电Li进入层间的位点有时变少。即,充电接受性降低。另一方面,如果超过该范围,则粒子表面的结晶性降低,在存在特定化合物的条件下进行充电时,表面生成的SEI覆盖膜的稳定性不充分,有时导致低充电深度下的高温保存特性的降低。The R value of the carbonaceous material measured by argon ion laser Raman spectroscopy is usually 0.001 or more, preferably 0.01 or more, and its upper limit is usually 0.2 or less, preferably 0.18 or less, more preferably 0.15 or less. If the R value is lower than this range, the crystallinity of the particle surface is too high, and there may be fewer sites for Li to enter the interlayer due to charge and discharge. That is, the charge acceptance is lowered. On the other hand, if it exceeds this range, the crystallinity of the particle surface decreases, and when charging is performed in the presence of a specific compound, the stability of the SEI coating film formed on the surface is insufficient, which may lead to high-temperature storage characteristics at a low charge depth. decrease.
另外,碳质物质在1580cm-1附近的拉曼半值宽度没有特别的限制,通常为10cm-1以上,优选为15cm-1以上,上限通常为35cm-1以下,优选为30cm-1以下的范围。如果拉曼半值宽度低于该范围,则粒子表面的结晶性过高,随着充放电Li进入层间的位点有时变少。即,充电接受性有时降低。另一方面,如果高于该范围,则粒子表面的结晶性降低,在存在特定化合物的条件下进行充电时,表面生成的SEI覆盖膜的稳定性不充分,有时导致低充电深度下的高温保存特性的降低。In addition, the Raman half-value width of the carbonaceous material around 1580 cm -1 is not particularly limited, but it is usually 10 cm -1 or more, preferably 15 cm -1 or more, and the upper limit is usually 35 cm -1 or less, preferably 30 cm -1 or less. scope. If the Raman half-value width is below this range, the crystallinity of the particle surface may be too high, and there may be fewer sites for Li to enter the interlayer due to charge and discharge. That is, charge acceptance may be lowered. On the other hand, if it exceeds this range, the crystallinity of the particle surface decreases, and when charging is performed in the presence of a specific compound, the stability of the SEI coating film formed on the surface is insufficient, which may lead to high-temperature storage at a low charging depth. reduction in characteristics.
拉曼光谱的测定如下进行:使用拉曼分光(例如,日本分光公司制造的拉曼分光器),使试样自然落入到测定容器内,进行试样填充,测定过程是,对容器内的试样表面照射氩离子激光,同时使该容器在与激光垂直的面内旋转。对于得到的拉曼光谱,测定1580cm-1附近的峰PA的强度IA和1360cm-1附近的峰PB的强度IB,计算其强度比R(R=IB/IA),将其定义为碳质物质的拉曼R值。另外,测定得到的拉曼光谱在1580cm-1附近的峰PA的半值宽度,将其定义为碳质物质的拉曼半值宽度。The measurement of the Raman spectrum is carried out as follows: using a Raman spectrometer (for example, a Raman spectrometer manufactured by JASCO Corporation), the sample is naturally dropped into the measurement container, and the sample is filled. The surface of the sample is irradiated with an argon ion laser while rotating the container in a plane perpendicular to the laser. For the obtained Raman spectrum, the intensity I A of the peak PA near 1580 cm -1 and the intensity I B of the peak P B near 1360 cm -1 were measured , and the intensity ratio R (R= IB / IA ) was calculated. It is defined as the Raman R value of the carbonaceous material. In addition, the half-value width of the peak PA in the vicinity of 1580 cm −1 in the obtained Raman spectrum was measured, and this was defined as the Raman half-value width of the carbonaceous substance.
另外,这里的拉曼测定条件如下。In addition, the Raman measurement conditions here are as follows.
·氩离子激光波长:514.5nmArgon ion laser wavelength: 514.5nm
·试样上的激光功率:15~25mW·Laser power on the sample: 15~25mW
·分辨率:10~20cm-1 ·Resolution: 10~20cm -1
·测定范围:1100cm-1~1730cm-1 · Measuring range: 1100cm -1 ~ 1730cm -1
·R值、半值宽度分析:本底处理R value, half-value width analysis: background processing
·光滑处理:单纯平均、卷积5点·Smooth processing: simple average, convolution 5 points
[[体积基准平均粒径]][[Volume-Based Average Particle Size]]
碳质物质的体积基准平均粒径是通过激光衍射/散射法求得的体积基准的平均粒径(中值粒径),通常为1μm以上,优选为3μm以上,更优选为5μm以上,进一步优选为7μm以上。另外,其上限通常为50μm以下,优选为40μm以下,更优选为30μm以下,进一步优选为25μm以下。如果低于上述范围,则不可逆容量增大,有时导致初期的电池容量的损失。另外,如果超过上述范围,则制成电极的极板时,容易形成不均匀的涂布面,有时在电池制作工序上是不优选的。The volume-based average particle size of the carbonaceous material is the volume-based average particle size (median particle size) obtained by the laser diffraction/scattering method, and is usually 1 μm or more, preferably 3 μm or more, more preferably 5 μm or more, and even more preferably 7 μm or more. In addition, the upper limit thereof is usually 50 μm or less, preferably 40 μm or less, more preferably 30 μm or less, and still more preferably 25 μm or less. If it is less than the above-mentioned range, the irreversible capacity may increase, which may lead to loss of initial battery capacity. In addition, when the above range is exceeded, an uneven coating surface is likely to be formed when forming an electrode plate, which may be unfavorable in the battery production process.
另外,以体积基准的粒径计,其分布的90%粒径和10%粒径之比(d90/d10)为1.2以上,优选为1.5以上,更优选为1.7以上。上限为8以下,优选为5以下,更优选为4以下,进一步优选为3以下的范围。In addition, the ratio (d 90 /d 10 ) of the 90% particle size to the 10% particle size of the distribution is 1.2 or more, preferably 1.5 or more, and more preferably 1.7 or more in terms of volume-based particle size. The upper limit is 8 or less, preferably 5 or less, more preferably 4 or less, and still more preferably 3 or less.
在本发明中,体积基准平均粒径用中值粒径来定义,所述中值粒径通过以下方法测定:使碳粉末分散在作为表面活性剂的聚氧乙烯(20)山梨糖醇酐单月桂酸酯的0.2质量%水溶液(约1mL)中,使用激光衍射/散射式粒度分布计(例如,堀场制作所公司制造的LA-700)来测定。作为90%粒径和10%粒径之比(d90/d10),可以同样地测定体积基准90%粒径和10%粒径,并使用其比值(d90/d10)。In the present invention, the volume-based average particle diameter is defined by a median particle diameter measured by dispersing carbon powder in polyoxyethylene (20) sorbitan as a surfactant. In a 0.2% by mass aqueous solution (about 1 mL) of monolaurate, it measures using a laser diffraction/scattering particle size distribution meter (for example, LA-700 manufactured by Horiba, Ltd.). As the ratio (d 90 /d 10 ) of the 90% particle diameter to the 10% particle diameter, the volume-based 90% particle diameter and the 10% particle diameter can be measured in the same manner, and the ratio (d 90 /d 10 ) can be used.
[[X射线参数]][[X-Ray Parameters]]
碳质物质通过采用学振法的X射线衍射求得的晶格面(002)的d值(层间距离)优选为0.335nm以上。另外,上限为0.340nm以下,优选为0.337nm以下。如果d值过大,则结晶性降低,有时会增加初期不可逆容量。另一方面,0.335为石墨的理论值。另外,通过采用学振法的X射线衍射求得的碳材料的微晶大小(Lc)通常为30nm以上,优选为50nm以上,更优选为80nm以上的范围。如果低于该范围,则粒子的结晶性降低,有可能增加初期不可逆容量。The carbonaceous material preferably has a d value (interlayer distance) of the lattice plane (002) obtained by X-ray diffraction using the Gakushin method of 0.335 nm or more. In addition, the upper limit is 0.340 nm or less, preferably 0.337 nm or less. If the d value is too large, the crystallinity may decrease, which may increase the initial irreversible capacity. On the other hand, 0.335 is the theoretical value of graphite. In addition, the crystallite size (Lc) of the carbon material obtained by X-ray diffraction using the Gakushin method is usually in the range of 30 nm or more, preferably 50 nm or more, more preferably 80 nm or more. If it is less than this range, the crystallinity of the particles may decrease, which may increase the initial irreversible capacity.
[[灰分]][[ash]]
碳质物质中所含的灰分相对于碳质物质的总质量为1质量%以下,优选为0.5质量%以下,特别优选为0.1质量%以下,其下限为1ppm以上。如果超过上述范围,则充放电时与电解液的反应而引起的电池性能的劣化不能忽视。如果低于该范围,则在制造上需要较长时间和能量以及用于防止污染的设备,有时成本会上升。The ash contained in the carbonaceous material is 1% by mass or less, preferably 0.5% by mass or less, particularly preferably 0.1% by mass or less, with a lower limit of 1 ppm or more based on the total mass of the carbonaceous material. If the above-mentioned range is exceeded, the degradation of battery performance due to the reaction with the electrolytic solution during charging and discharging cannot be ignored. If it is less than this range, long time and energy are required for manufacturing and equipment for preventing pollution, and the cost may increase.
[[BET比表面积]][[BET specific surface area]]
使用BET法测定的碳质物质的比表面积通常为0.1m2/g以上,优选为0.7m2/g以上,更优选为1m2/g以上,进一步优选为1.5m2/g以上。其上限通常为100m2/g以下,优选为25m2/g以下,更优选为15m2/g以下,进一步优选为10m2/g以下。如果比表面积的值低于上述范围,则在作为负极材料使用的情况下,在充电时锂的接受性变差,锂容易在电极表面析出。另一方面,如果超过上述范围,则在作为负极材料使用时,与电解液的反应性增加,产生的气体变多,有时难以得到优选的电池。The specific surface area of the carbonaceous material measured by the BET method is usually at least 0.1 m 2 /g, preferably at least 0.7 m 2 /g, more preferably at least 1 m 2 /g, and still more preferably at least 1.5 m 2 /g. The upper limit thereof is usually 100 m 2 /g or less, preferably 25 m 2 /g or less, more preferably 15 m 2 /g or less, still more preferably 10 m 2 /g or less. If the value of the specific surface area is less than the above range, when used as a negative electrode material, the acceptance of lithium at the time of charging will be deteriorated, and lithium will be easily deposited on the electrode surface. On the other hand, when it exceeds the above-mentioned range, when used as a negative electrode material, the reactivity with the electrolytic solution increases and gas generation increases, making it difficult to obtain a preferable battery in some cases.
BET比表面积定义为如下测定的值:使用表面积计(例如,大仓理研制造的全自动表面积测定装置),在氮气流通、350℃下对试样进行15分钟预干燥,然后,使用氮气相对于大气压的相对压力值准确调整为0.3的氮氦混合气体,通过采用气体流动法的氮吸附BET1点法测定。The BET specific surface area is defined as a value measured as follows: Using a surface area meter (for example, a fully automatic surface area measuring device manufactured by Okura Riken), the sample is pre-dried at 350° C. for 15 minutes under nitrogen flow, and then, using nitrogen gas relative to The relative pressure value of the atmospheric pressure is accurately adjusted to 0.3 nitrogen-helium mixed gas, and it is measured by the nitrogen adsorption BET 1-point method using the gas flow method.
[[微孔分布]][[Pore Distribution]]
通过水银孔隙率计(水银压入法)求得的碳质物质的相当于直径0.01μm~1μm的粒子内的空隙、由于粒子表面的高低不平造成的凹凸的量通常为0.01mL/g以上,优选为0.05mL/g以上,更优选为0.1mL/g以上,上限通常为0.6mL/g以下,优选为0.4mL/g以下,更优选为0.3mL/g以下的范围。如果超过该范围,则制造成极板时有时需要大量的粘合剂。另一方面,如果低于该范围,则高电流密度充放电特性降低,并且,有时不能得到充放电时的电极膨胀收缩的缓和效果。The amount of voids in the carbonaceous material corresponding to particles with a diameter of 0.01 μm to 1 μm and the amount of unevenness caused by the surface of the particles measured by mercury porosimetry (mercury porosimetry) is usually 0.01 mL/g or more, It is preferably 0.05 mL/g or more, more preferably 0.1 mL/g or more, and the upper limit is usually 0.6 mL/g or less, preferably 0.4 mL/g or less, more preferably 0.3 mL/g or less. If it exceeds this range, a large amount of binder may be required when manufacturing an electrode plate. On the other hand, if it is less than this range, the high current density charge-discharge characteristics deteriorate, and the effect of alleviating electrode expansion and contraction during charge-discharge may not be obtained.
另外,总微孔容积优选为0.1mL/g以上,更优选为0.25mL/g以上,其上限通常为10mL/g以下,优选为5mL/g以下,更优选为2mL/g以下的范围。如果超过该范围,则在制成基板时有时需要大量的粘合剂。如果低于该范围,则在制成极板时有时不能得到增稠剂或粘结剂的分散效果。In addition, the total pore volume is preferably 0.1 mL/g or more, more preferably 0.25 mL/g or more, and its upper limit is usually 10 mL/g or less, preferably 5 mL/g or less, more preferably 2 mL/g or less. If it exceeds this range, a large amount of adhesive may be required to form a substrate. If it is less than this range, the dispersion effect of the thickener or the binder may not be obtained when forming an electrode plate.
另外,平均微孔径优选为0.05μm以上,更优选为0.1μm以上,进一步优选为0.5μm以上,其上限通常为50μm以下,优选为20μm以下,更优选为10μm以下的范围。如果超过该范围,则有时需要大量的粘合剂。另一方面,如果低于该范围,则有时高电流密度充放电特性降低。In addition, the average pore diameter is preferably 0.05 μm or more, more preferably 0.1 μm or more, even more preferably 0.5 μm or more, and the upper limit thereof is usually 50 μm or less, preferably 20 μm or less, more preferably 10 μm or less. If this range is exceeded, a large amount of adhesive is sometimes required. On the other hand, if it is less than this range, the high current density charge-discharge characteristics may deteriorate.
作为用于水银孔隙率计的装置,可以使用水银孔隙率计(autopore9520;micrometritics公司制造)。称量约0.2g试样(负极材料),密封入粉末用容器中,在室温、真空下(50μmHg以下)进行10分钟脱气,实施前处理。接着,减压至4psia(约28kPa),导入水银,从4psia(约28kPa)阶段状升压至40000psia(约280MPa),然后降压至25psia(约170kPa)。升压时的阶段数为80阶段以上,在各阶段中,在10秒的平衡时间后,测定水银压入量。使用Washburn式从这样得到的水银压入曲线计算出微孔分布。另外,水银的表面张力(γ)为485dyne/cm,接触角(φ)为140°。平均微孔径使用累积微孔体积达到50%时的微孔径。As an apparatus used for the mercury porosimeter, a mercury porosimeter (autopore9520; manufactured by Micrometrics Inc.) can be used. About 0.2 g of the sample (negative electrode material) was weighed, sealed in a powder container, and degassed at room temperature under vacuum (50 μmHg or less) for 10 minutes to perform pretreatment. Next, the pressure is reduced to 4psia (about 28kPa), mercury is introduced, the pressure is raised from 4psia (about 28kPa) to 40000psia (about 280MPa) in steps, and then the pressure is lowered to 25psia (about 170kPa). The number of stages during the pressurization was 80 or more, and the amount of mercury intrusion was measured after an equilibration time of 10 seconds in each stage. The micropore distribution was calculated from the mercury intrusion curve thus obtained using the Washburn formula. In addition, the surface tension (γ) of mercury is 485 dyne/cm, and the contact angle (φ) is 140°. As the average pore diameter, the pore diameter at which the cumulative pore volume reaches 50% is used.
[[真密度]][[true density]]
碳质物质的真密度通常为2g/cm3以上,优选为2.1g/cm3以上,更优选为2.2g/cm3以上,进一步优选为2.22g/cm3以上,其上限为2.26g/cm3以下。上限为石墨的理论值。如果低于该范围,则碳的结晶性过低,初期不可逆容量有时会增大。在本发明中,真密度定义为通过使用丁醇的液相置换法(比重计法)测定的值。The true density of the carbonaceous material is usually above 2 g/cm 3 , preferably above 2.1 g/cm 3 , more preferably above 2.2 g/cm 3 , and even more preferably above 2.22 g/cm 3 , with an upper limit of 2.26 g/cm 3 3 or less. The upper limit is the theoretical value of graphite. If it is less than this range, the crystallinity of carbon may be too low, and the initial irreversible capacity may increase. In the present invention, the true density is defined as a value measured by a liquid phase displacement method (pycnometer method) using butanol.
[[取向比]][[Orientation Ratio]]
碳质物质的取向比通常为0.005以上,优选为0.01以上,更优选为0.015以上,上限在理论上为0.67以下的范围。如果低于该范围,则有时高密度充放电特性降低。The orientation ratio of the carbonaceous material is usually 0.005 or more, preferably 0.01 or more, more preferably 0.015 or more, and the upper limit is theoretically 0.67 or less. If it is less than this range, high-density charge-discharge characteristics may deteriorate.
取向比通过X射线衍射测定。使用非对称皮尔森VII作为分布函数,拟合通过X射线衍射得到的碳的(110)衍射和(004)衍射的峰,进行峰分离,分别计算出(110)衍射和(004)衍射的峰的积分强度。由得到的积分强度计算出由(110)衍射积分强度/(004)衍射积分强度表示的比值,将该比值定义为活性物质取向比。The orientation ratio was measured by X-ray diffraction. Using asymmetric Pearson VII as a distribution function, fit the peaks of (110) diffraction and (004) diffraction of carbon obtained by X-ray diffraction, perform peak separation, and calculate the peaks of (110) diffraction and (004) diffraction respectively integral strength. A ratio represented by (110) diffraction integrated intensity/(004) diffraction integrated intensity was calculated from the obtained integrated intensity, and this ratio was defined as the active material orientation ratio.
这里的X射线衍射测定条件如下。另外,“2θ”表示衍射角。The X-ray diffraction measurement conditions here are as follows. In addition, "2θ" represents a diffraction angle.
·靶:Cu(Kα射线)石墨单色光度仪Target: Cu (Kα ray) graphite monochromator
·狭缝:发散狭缝=1度,受光狭缝=0.1mm,散射狭缝=1度· Slit: divergence slit = 1 degree, light receiving slit = 0.1mm, scattering slit = 1 degree
·测定范围和步进角/测定时间・Measurement range and step angle/measurement time
(110)面:76.5度≤2θ≤78.5度0.01度/3秒(110) surface: 76.5 degrees ≤ 2θ ≤ 78.5 degrees 0.01 degrees / 3 seconds
(004)面:53.5度≤2θ≤56.0度0.01度/3秒(004) plane: 53.5 degrees ≤ 2θ ≤ 56.0 degrees 0.01 degrees / 3 seconds
[[长径比]][[Aspect Ratio]]
碳质物质的长径比理论上为1以上,上限为10以下,优选为8以下,更优选为5以下。如果超过其上限,制成极板时会产生条纹、无法得到均匀的涂布面,高电流密度充放电特性有时降低。The aspect ratio of the carbonaceous material is theoretically 1 or more, and the upper limit is 10 or less, preferably 8 or less, more preferably 5 or less. If the upper limit is exceeded, streaks may be generated when forming an electrode plate, a uniform coating surface may not be obtained, and the high current density charge and discharge characteristics may deteriorate.
另外,长径比用三维观察时粒子的最长径A和与其垂直的最短径B的比值A/B来表示。粒子的观察通过可以放大观察的扫描型电子显微镜来进行。选择固定在厚度50微米以下的金属的端面上的任意50个石墨粒子,旋转、倾斜固定有试样的载物台,分别测定这些粒子的A、B,求出A/B的平均值。In addition, the aspect ratio is represented by the ratio A/B of the longest diameter A of a particle and the shortest diameter B perpendicular thereto in three-dimensional observation. Observation of particles is performed with a scanning electron microscope capable of magnified observation. Select any 50 graphite particles fixed on the end face of the metal with a thickness of 50 microns or less, rotate and tilt the stage on which the sample is fixed, measure A and B of these particles, and calculate the average value of A/B.
[[碳质物质的制造法及原料]][[Manufacturing method and raw material of carbonaceous material]]
本发明中使用的碳质物质可以是天然出产的物质,也可以是人工制造的物质,但优选来自于天然石墨的物质。另外,天然出产的物质或人工制造的物质也可以施加特定的处理。制造方法(也包含分选方法)没有特别限制,例如,可以是使用筛分或风力分级等分级方法分选具有上述特性的碳质物质而取得的物质。The carbonaceous substance used in the present invention may be either naturally occurring or artificial, but is preferably derived from natural graphite. In addition, naturally occurring substances or artificially produced substances may also be subject to specific treatments. The production method (including the sorting method) is not particularly limited, and for example, a carbonaceous material having the above-mentioned characteristics may be sorted using a classification method such as sieving or wind classification.
这些当中,从容易获得、前面的工序中加工容易这点来看,优选通过对天然出产的碳材料(天然石墨原料)进行热处理而得到的碳质物质。另外,从提高填充性等观点来看,优选如下获得的碳质物质:对天然出产的碳材料(天然石墨原料)或人工制造的碳材料施加力学能量处理而进行改性、球形化,再将得到的球形化碳质进行热处理。另外,从锂二次电池的性能的平衡等观点来看,特别优选如下制备的碳质物质:对天然石墨原料施加力学能量处理,并将得到的球形化天然石墨进行热处理。下面,有时将上述天然石墨原料等的热处理前的碳材料(原料)只称为“热处理前的原料”。Among these, carbonaceous substances obtained by heat-treating naturally occurring carbon materials (natural graphite raw materials) are preferable from the viewpoint of easy availability and easy processing in the preceding steps. In addition, from the viewpoint of improving filling properties, etc., carbonaceous substances obtained by applying mechanical energy treatment to naturally occurring carbon materials (natural graphite raw materials) or artificially produced carbon materials to modify and spheroidize, and then The obtained spheroidized carbonaceous is subjected to heat treatment. In addition, from the viewpoint of the balance of the performance of the lithium secondary battery, etc., carbonaceous materials prepared by subjecting natural graphite raw materials to mechanical energy treatment and heat-treating the obtained spheroidized natural graphite are particularly preferable. Hereinafter, the carbon material (raw material) before heat treatment, such as the above-mentioned natural graphite raw material, may be simply referred to as "raw material before heat treatment".
[[天然石墨原料]][[Natural graphite raw material]]
如上所述,作为碳质物质的原料,特别优选天然石墨。As described above, natural graphite is particularly preferable as the raw material of the carbonaceous substance.
天然石墨依照其性状,被分类为鳞片状石墨(Flake Graphite)、鳞状石墨(Crystalline(Vein)Graphite)、土壤石墨(Amorphous Graphite)(参见《粉粒体工艺技术集成》,(株)产业技术中心,昭和49年发行的石墨一项;以及“HANDBOOK OF CARBON,GRAPHITE,DIAMOND AND FULLERENES”,NoyesPublications发行)。石墨化度以鳞状石墨最高,为100%,接着是鳞片状石墨,为99.9%,而土壤石墨低至28%。天然石墨的品质主要根据产地和矿脉来确定。鳞片状石墨主要产自马达加斯加、中国、巴西、乌克兰、加拿大等;鳞状石墨主要产自斯里兰卡,土壤石墨的主产地为朝鲜半岛、中国、墨西哥等。这些天然石墨中,由于鳞片状石墨和鳞状石墨具有石墨化度高、杂质量少等优点,因此优选作为碳质物质的原料。Natural graphite is classified into flake graphite (Flake Graphite), scaly graphite (Crystalline (Vein) Graphite), and soil graphite (Amorphous Graphite) according to its properties (see "Powder Granular Process Technology Integration", Industrial Technology Co., Ltd. Center, Graphite item issued in Showa 49; and "HANDBOOK OF CARBON, GRAPHITE, DIAMOND AND FULLERENES", issued by NoyesPublications). The degree of graphitization is highest in flake graphite, which is 100%, followed by flake graphite, which is 99.9%, while soil graphite is as low as 28%. The quality of natural graphite is mainly determined by the place of origin and the vein. Flake graphite is mainly produced in Madagascar, China, Brazil, Ukraine, Canada, etc.; flake graphite is mainly produced in Sri Lanka, and soil graphite is mainly produced in the Korean Peninsula, China, Mexico, etc. Among these natural graphites, flaky graphite and flaky graphite are preferable as raw materials for carbonaceous substances because they have advantages such as a high degree of graphitization and a small amount of impurities.
[[[力学能量处理、球形化处理]]][[[Mechanical energy processing, spherical processing]]]
进行力学能量处理使处理前后的体积平均粒径为1以下。所谓“处理前后的体积平均粒径比”是处理后的体积平均粒径除以处理前的体积平均粒径而得到的值。在本发明中,为了制造热处理前的原料而进行的力学能量处理优选使处理前后的平均粒径比为1以下。The mechanical energy treatment is performed so that the volume average particle diameter before and after the treatment becomes 1 or less. The "volume average particle diameter ratio before and after treatment" is a value obtained by dividing the volume average particle diameter after treatment by the volume average particle diameter before treatment. In the present invention, it is preferable that the kinetic energy treatment performed to produce the raw material before the heat treatment is such that the average particle diameter ratio before and after the treatment is 1 or less.
力学能量处理是减小粒子尺寸以使粉末粒子处理前后的平均粒径比为1以下,同时控制粒子形状而进行的处理。在粉碎、分级、混合、造粒、表面改性、反应等可以在粒子设计中有效利用的工学单位操作中,力学能量处理属于粉碎处理。The mechanical energy treatment is a treatment performed to reduce the particle size so that the average particle diameter ratio before and after the powder particle treatment is 1 or less, and at the same time control the particle shape. Among engineering unit operations that can be effectively utilized in particle design, such as pulverization, classification, mixing, granulation, surface modification, and reaction, mechanical energy processing belongs to pulverization processing.
所谓粉碎,是指对物质施加力,使其大小减少以调节物质的粒径或粒度分布、填充性。粉碎处理依据对物质施加的力的种类、处理形态来进行分类。对物质施加的力大致分为如下4种:(1)敲打力(冲击力)、(2)压碎力(压缩力)、(3)磨碎力(磨碎力)、(4)削刮力(剪切力)。另一方面,处理形态大致分为如下2种:在粒子内部产生龟裂并使之传播的体积粉碎、以及切去粒子表面的表面粉碎。体积粉碎可采用冲击力、压缩力、剪切力来进行;表面粉碎可采用磨碎力、剪切力来进行。粉碎是将对这些物质施加的力的种类和处理形态进行各种组合的处理。其组合可以根据处理目的适当确定。The so-called pulverization refers to applying force to a substance to reduce its size to adjust the particle size, particle size distribution, and filling properties of the substance. Pulverization treatment is classified according to the type of force applied to the substance and the form of treatment. The force exerted on the substance is roughly divided into the following four types: (1) knocking force (impact force), (2) crushing force (compression force), (3) grinding force (grinding force), (4) scraping force force (shear force). On the other hand, processing forms are roughly classified into two types: volume crushing in which cracks are generated inside the particles and propagated, and surface crushing in which the particle surfaces are cut off. Volume crushing can be carried out by impact force, compression force and shear force; surface crushing can be carried out by grinding force and shear force. Pulverization is a process of various combinations of the type of force applied to these substances and the form of treatment. The combination thereof can be appropriately determined according to the purpose of treatment.
粉碎虽然也有使用爆破等化学的反应或体积膨胀来进行的情况,但通常使用粉碎机等机械装置来进行。作为本发明的原料的球形化碳质的制造中使用的粉碎处理优选最终表面处理所占的比例变高的处理,而不管有无体积粉碎。这是因为,去掉粒子表面粉碎的角以对粒子形状导入圆形是重要的。具体地,可以在进行一定程度的体积粉碎之后再进行表面处理,也可以几乎不进行体积粉碎而只进行表面处理,还可以同时进行体积粉碎和表面处理。优选最后进行表面粉碎,从粒子的表面去掉角的粉碎处理。Pulverization may be performed using a chemical reaction such as blasting or volume expansion, but it is usually performed using a mechanical device such as a pulverizer. The pulverization treatment used in the production of the spheroidized carbonaceous material that is the raw material of the present invention is preferably a treatment that increases the ratio of the final surface treatment regardless of the presence or absence of volume pulverization. This is because it is important to remove the pulverized corners of the particle surface to introduce a round shape to the particle shape. Specifically, the surface treatment may be performed after a certain volume pulverization is performed, only the surface treatment may be performed without volume pulverization, or the volume pulverization and surface treatment may be performed simultaneously. It is preferable to perform surface crushing at the end to remove corners from the surface of the particles.
进行力学能量处理的装置从能够进行上述的优选的处理的装置中选择。力学能量处理可以通过使用对上述物质施加的4种力中的一种以上的力来实现,但优选对主体反复进行包含粒子的相互作用的压缩、摩擦、剪切力等机械作用,从而对粒子赋予冲击力。因此,具体地,优选如下的装置:该装置在箱内部具有设置了多个叶片的转子,并通过该转子高速旋转而对导入到内部的碳材料赋予冲击压缩、摩擦、剪切力等机械的作用,从而边进行体积粉碎边进行表面处理。另外,更优选具有通过使碳质物质循环或对流而反复赋予机械作用的机构的装置。The device for mechanical energy processing is selected from devices capable of performing the above-mentioned preferable processing. The mechanical energy treatment can be realized by using more than one of the four kinds of forces applied to the above-mentioned substances, but it is preferable to repeatedly perform mechanical actions such as compression, friction, and shearing forces that include the interaction of particles on the body, so that the particles Gives impact. Therefore, specifically, a device that has a rotor provided with a plurality of blades inside the tank and that imparts mechanical forces such as impact compression, friction, and shearing force to the carbon material introduced into the inside by rotating the rotor at high speed is preferable. function, thereby carrying out surface treatment while performing volume crushing. In addition, a device having a mechanism for repeatedly imparting a mechanical action by circulating or convecting the carbonaceous material is more preferable.
作为优选的装置,可列举混合系统(奈良机械制作所公司制造)、Kryptron(クリプトロン)(Earth Technica(ア一ステクニカ)公司制造)、CF磨(宇部兴产公司制造)、机械熔融系统(hosokawamicron(ホソカワミクロン)公司制造)等。这些当中,优选奈良机械制作所公司制造的混合系统。使用该装置进行处理时,优选将旋转的转子的圆周速度设定为30~100m/秒,更优选设定为40~100m/秒,进一步优选设定为50~100m/秒。另外,处理可以只是仅仅使碳质物质通过,但优选在装置内循环或滞留30秒钟以上来进行处理,更优选在装置内循环或滞留1分钟以上来进行处理。As a preferable device, a mixing system (manufactured by Nara Machinery Manufacturing Co., Ltd.), Kryptron (cryptron) (manufactured by Earth Technica (Asteknica) Co., Ltd.), CF mill (manufactured by Ube Industrial Co., Ltd.), mechanical melting system (hosokawamicron) can be mentioned. (manufactured by Hosokawa Micron Co., Ltd.), etc. Among these, the mixing system manufactured by Nara Machinery Manufacturing Co., Ltd. is preferable. When using this apparatus for processing, the peripheral speed of the rotating rotor is preferably set at 30 to 100 m/sec, more preferably at 40 to 100 m/sec, and even more preferably at 50 to 100 m/sec. In addition, the treatment may be performed by merely passing the carbonaceous material, but it is preferably treated by circulating or staying in the device for 30 seconds or more, more preferably by circulating or staying in the device for 1 minute or more.
通过这样进行力学能量处理,碳粒子成为如下的粒子:整体上保持高结晶性,只是粒子的表面附近变得粗糙,倾斜且露出边缘面。这样,锂离子可以出入的面增加,即使在高电流密度下也具有高的容量。By performing the mechanical energy treatment in this way, the carbon particles become particles in which high crystallinity is maintained as a whole, but the vicinity of the surface of the particles becomes rough, and the edge surfaces are inclined and exposed. In this way, the surface where lithium ions can come and go increases, and it has a high capacity even at a high current density.
一般来说,鳞片状、鳞状、板状的碳材料的粒径越小,其填充性越有恶化的倾向。这可认为是因为如下原因:由于粒子通过粉碎而更加无定形化,或者粒子表面生成的“毛边”、“剥离”或“折曲”等突起状物增加,以及在粒子表面以某种程度的强度附着有更微细的无定形粒子等原因,这样,与邻接粒子之间的阻抗变大,使填充性恶化。In general, the smaller the particle size of the scale-like, scale-like, and plate-like carbon materials tends to be, the worse the fillability is. This is considered to be due to the following reasons: as the particles become more amorphous by pulverization, or protrusions such as "burrs", "peeling" or "bends" generated on the particle surface increase, and some degree of deformation occurs on the particle surface. The strength is caused by the adhesion of finer amorphous particles, etc., so that the resistance between adjacent particles becomes larger, and the filling property is deteriorated.
如果这些粒子的无定形性减少,粒子形状接近于球形,则即使粒径变小,填充性的减小也很少,在理论上,大粒径碳粉末、小粒径碳粉末都应该显示同等程度的振实密度。If the amorphousness of these particles is reduced and the particle shape is close to spherical, even if the particle size becomes smaller, the filling property will be reduced very little. In theory, both large particle size carbon powder and small particle size carbon powder should be Shows an equivalent degree of tap density.
[[[热处理前的原料的物性]]][[[Physical properties of raw materials before heat treatment]]]
热处理前的原料的物性优选同时满足下面所示的(1)~(11)中的任意一项或多项。另外,无形测定方法或定义均与上述碳质物质的情况相同。The physical properties of the raw material before heat treatment preferably satisfy any one or more of the following (1) to (11) at the same time. In addition, the intangible measurement method and definition are the same as in the case of the above-mentioned carbonaceous substance.
(1)X射线参数(1) X-ray parameters
热处理前的原料通过采用学振法的X射线衍射求得的晶格面(002)的d值(层间距离)优选为0.335nm以上。另外,其下限为不足0.340nm,优选为0.337nm以下。如果d值过大,则结晶性降低,有时会增加初期不可逆容量。另一方面,0.335为石墨的理论值。另外,通过采用学振法的X射线衍射求得的碳材料的微晶大小(Lc)通常为30nm以上,优选为50nm以上,更优选为80nm以上的范围。如果低于该范围,则结晶性降低,有可能增加初期不可逆容量。The d value (interlayer distance) of the lattice plane (002) obtained by X-ray diffraction using the Gakushin method of the raw material before heat treatment is preferably 0.335 nm or more. In addition, the lower limit thereof is less than 0.340 nm, preferably 0.337 nm or less. If the d value is too large, the crystallinity may decrease, which may increase the initial irreversible capacity. On the other hand, 0.335 is the theoretical value of graphite. In addition, the crystallite size (Lc) of the carbon material obtained by X-ray diffraction using the Gakushin method is usually in the range of 30 nm or more, preferably 50 nm or more, more preferably 80 nm or more. If it is less than this range, the crystallinity may decrease, which may increase the initial irreversible capacity.
(2)灰分(2) Ash content
热处理前的原料中所含的灰分相对于热处理前的原料的总质量通常为1质量%以下,优选为0.5质量%以下,特别优选为0.1质量%以下,其下限通常为1ppm以上。如果超过上述范围,则充放电时与电解液的反应而引起的电池性能的劣化不能忽视。如果低于该范围,则在制造上需要较长时间和能量以及用于防止污染的设备,有时成本会上升。The ash contained in the raw material before heat treatment is usually 1% by mass or less, preferably 0.5% by mass or less, particularly preferably 0.1% by mass or less, and the lower limit thereof is usually 1 ppm or more based on the total mass of the raw material before heat treatment. If the above-mentioned range is exceeded, the degradation of battery performance due to the reaction with the electrolytic solution during charging and discharging cannot be ignored. If it is less than this range, long time and energy are required for manufacturing and equipment for preventing pollution, and the cost may increase.
(3)体积基准平均粒径(3) Volume-based average particle size
热处理前的原料的体积基准平均粒径是通过激光衍射/散射法求得的体积基准的平均粒径(中值粒径),通常为1μm以上,优选为3μm以上,更优选为5μm以上,进一步优选为7μm以上。另外,其上限通常为50μm以下,优选为40μm以下,更优选为30μm以下,进一步优选为25μm以下。如果低于上述范围,则不可逆容量增大,有时导致初期电池容量的损失。另外,如果超过上述范围,则在制成电极极板时,容易形成不均匀的涂布面,有时在电池制作工序上是不优选的。The volume-based average particle diameter of the raw material before heat treatment is the volume-based average particle diameter (median particle diameter) obtained by the laser diffraction/scattering method, and is usually 1 μm or more, preferably 3 μm or more, more preferably 5 μm or more, and further It is preferably 7 μm or more. In addition, the upper limit thereof is usually 50 μm or less, preferably 40 μm or less, more preferably 30 μm or less, and still more preferably 25 μm or less. If it is less than the above-mentioned range, the irreversible capacity may increase, resulting in loss of initial battery capacity. Moreover, if it exceeds the said range, it will become easy to form an uneven coating surface at the time of making an electrode pad, and it may be unpreferable in a battery manufacturing process.
(4)拉曼R值、拉曼半值宽度(4) Raman R value, Raman half value width
使用氩离子激光拉曼光谱法测定的热处理前的原料的R值通常为0.10以上,优选为0.15以上,更优选为0.17以上,进一步优选为0.2以上,其上限通常为0.8以下,优选为0.6以下,更优选为0.4以下的范围。如果R值低于该范围,则有可能不进行粒子的球形化,有可能不能获得填充性提高的效果。另一方面,如果超过该范围,则粒子表面的结晶性降低,与电解液的反应性增加,有时导致效率降低或产生的气体增加。The R value of the raw material before heat treatment measured by argon ion laser Raman spectroscopy is usually 0.10 or more, preferably 0.15 or more, more preferably 0.17 or more, still more preferably 0.2 or more, and the upper limit thereof is usually 0.8 or less, preferably 0.6 or less , more preferably in the range of 0.4 or less. If the R value is lower than this range, the particles may not be spheroidized, and the effect of improving filling properties may not be obtained. On the other hand, if it exceeds this range, the crystallinity of the particle surface decreases, and the reactivity with the electrolyte solution increases, resulting in a decrease in efficiency or an increase in gas generation.
另外,热处理前的原料在1580cm-1附近的拉曼半值宽度没有特别的限制,通常为10cm-1以上,优选为15cm-1以上,上限通常为80cm-1以下,优选为60cm-1以下,更优选为45cm-1以下,进一步优选为40cm-1以下的范围。如果拉曼半值宽度低于该范围,则有可能不进行粒子的球形化,有可能不能获得填充性提高的效果。另一方面,如果高于该范围,则粒子表面的结晶性降低,与电解液的反应性增加,有时导致效率降低或产生的气体增加。In addition, the Raman half-value width near 1580 cm -1 of the raw material before heat treatment is not particularly limited, but it is usually 10 cm -1 or more, preferably 15 cm -1 or more, and the upper limit is usually 80 cm -1 or less, preferably 60 cm -1 or less , more preferably 45 cm -1 or less, still more preferably 40 cm -1 or less. If the Raman half-value width is less than this range, the particles may not be spheroidized, and the effect of improving filling properties may not be obtained. On the other hand, if it exceeds this range, the crystallinity of the particle surface will decrease, and the reactivity with the electrolytic solution will increase, which may result in a decrease in efficiency or an increase in generated gas.
(5)BET比表面积(5) BET specific surface area
使用BET法测定的热处理前的原料的比表面积通常为0.1m2/g以上,优选为0.7m2/g以上,更优选为1m2/g以上,进一步优选为1.5m2/g以上。其上限通常为100m2/g以下,优选为50m2/g以下,更优选为15m2/g以下,进一步优选为10m2/g以下。如果比表面积的值低于上述范围,则作为负极材料使用的情况下,在充电时锂的接受性变差,锂容易在电极表面析出。另一方面,如果超过上述范围,则经过热处理而使比表面积减少,产生必要以上的与电解液的反应,产生的气体变多,有时难以得到优选的电池。The specific surface area of the raw material before heat treatment measured by the BET method is usually 0.1 m 2 /g or more, preferably 0.7 m 2 /g or more, more preferably 1 m 2 /g or more, still more preferably 1.5 m 2 /g or more. The upper limit thereof is usually 100 m 2 /g or less, preferably 50 m 2 /g or less, more preferably 15 m 2 /g or less, still more preferably 10 m 2 /g or less. If the value of the specific surface area is less than the above range, when used as a negative electrode material, the acceptance of lithium at the time of charging will be deteriorated, and lithium will be easily deposited on the electrode surface. On the other hand, if it exceeds the above range, heat treatment will reduce the specific surface area, more than necessary reaction with the electrolytic solution will occur, and more gas will be generated, making it difficult to obtain a preferable battery.
(6)微孔分布(6) Micropore distribution
通过水银孔隙率计(水银压入法)求得的热处理前的原料的相当于直径为0.01μm~1μm的粒子内的空隙、由于粒子表面的高低不平造成的凹凸的量通常为0.01mL/g以上,优选为0.05mL/g以上,更优选为0.1mL/g以上,其上限通常为0.6mL/g以下,优选为0.4mL/g以下,更优选为0.3mL/g以下的范围。如果超过该范围,则制造成极板时需要大量的粘合剂。另一方面,如果低于该范围,则高电流密度充放电特性降低,并且,有时不能得到充放电时的电极膨胀收缩的缓和效果。The amount of voids in particles with a diameter of 0.01 μm to 1 μm and unevenness on the particle surface of the raw material before heat treatment measured by mercury porosimetry (mercury porosimetry) is usually 0.01 mL/g Above, preferably 0.05 mL/g or more, more preferably 0.1 mL/g or more, the upper limit is usually 0.6 mL/g or less, preferably 0.4 mL/g or less, more preferably 0.3 mL/g or less. If it exceeds this range, a large amount of binder will be required to manufacture an electrode plate. On the other hand, if it is less than this range, the high current density charge-discharge characteristics deteriorate, and the effect of alleviating electrode expansion and contraction during charge-discharge may not be obtained.
另外,总微孔容积优选为0.1mL/g以上,更优选为0.25mL/g以上,其上限通常为10mL/g以下,优选为5mL/g以下,更优选为2mL/g以下的范围。如果超过该范围,则在制成基板时有时需要大量的粘合剂。如果低于该范围,则在制成极板时有时不能得到增稠剂或粘结剂的分散效果。In addition, the total pore volume is preferably 0.1 mL/g or more, more preferably 0.25 mL/g or more, and its upper limit is usually 10 mL/g or less, preferably 5 mL/g or less, more preferably 2 mL/g or less. If it exceeds this range, a large amount of adhesive may be required to form a substrate. If it is less than this range, the dispersion effect of the thickener or the binder may not be obtained when forming an electrode plate.
另外,平均微孔径优选为0.05μm以上,更优选为0.1μm以上,进一步优选为0.5μm以上,其上限通常为50μm以下,优选为20μm以下,更优选为10μm以下的范围。如果超过该范围,则有时需要大量的粘合剂。另一方面,如果低于该范围,则有时高电流密度充放电特性降低。In addition, the average pore diameter is preferably 0.05 μm or more, more preferably 0.1 μm or more, even more preferably 0.5 μm or more, and the upper limit thereof is usually 50 μm or less, preferably 20 μm or less, more preferably 10 μm or less. If this range is exceeded, a large amount of adhesive is sometimes required. On the other hand, if it is less than this range, the high current density charge-discharge characteristics may deteriorate.
(7)圆形度(7) Circularity
使用圆形度作为热处理前的原料的球形的程度,热处理前的原料的粒径为3~40μm范围的粒子的圆形度优选为0.85以上,更优选为0.87以上,特别优选为0.90以上,进一步优选为0.92以上。圆形度大时,高电流密度充放电特性提高,因此优选。Using circularity as the degree of sphericity of the raw material before heat treatment, the circularity of particles in the range of 3 to 40 μm in particle diameter of the raw material before heat treatment is preferably 0.85 or more, more preferably 0.87 or more, particularly preferably 0.90 or more, and further Preferably it is 0.92 or more. When the circularity is large, the high current density charge and discharge characteristics are improved, which is preferable.
提高圆形度的方法没有特别限定,但优选上述的通过力学的能量实施球形化处理而制成球形,因为这样的话,在制成电极体时的粒子间空隙的形状整齐一致。The method of improving the circularity is not particularly limited, but it is preferable to perform the spheroidization treatment by the above-mentioned mechanical energy to form a spherical shape, because in this way, the shapes of the interparticle voids are uniform when the electrode body is produced.
(8)真密度(8) True density
热处理前的原料的真密度通常为2g/cm3以上,优选为2.1g/cm3以上,更优选为2.2g/cm3以上,进一步优选为2.22g/cm3以上,其上限为2.26g/cm3以下。上限为石墨的理论值。如果低于该范围,则碳的结晶性过低,初期不可逆容量有时会增大。The true density of the raw material before heat treatment is usually 2 g/cm 3 or more, preferably 2.1 g/cm 3 or more, more preferably 2.2 g/cm 3 or more, still more preferably 2.22 g/cm 3 or more, and the upper limit is 2.26 g/cm 3 or more. cm3 or less. The upper limit is the theoretical value of graphite. If it is less than this range, the crystallinity of carbon may be too low, and the initial irreversible capacity may increase.
(9)振实密度(9) Tap density
热处理前的原料的振实密度通常为0.55g/cm3以上,优选为0.7g/cm3以上,更优选为0.8g/cm3以上,特别优选为0.9g/cm3以上。另外,其上限优选为2g/cm3以下,更优选为1.8g/cm3以下,特别优选为1.6g/cm3以下。振实密度如果低于该范围,则在作为负极使用时,填充密度很难提高,有时不能得到高容量的电池。另一方面,如果高于该范围,则电极中的粒子间的空隙过少,难以确保粒子间的导电性,有时难以得到优选的电池特性。The tap density of the raw material before heat treatment is usually 0.55 g/cm 3 or more, preferably 0.7 g/cm 3 or more, more preferably 0.8 g/cm 3 or more, particularly preferably 0.9 g/cm 3 or more. In addition, the upper limit thereof is preferably 2 g/cm 3 or less, more preferably 1.8 g/cm 3 or less, particularly preferably 1.6 g/cm 3 or less. If the tap density is lower than this range, it is difficult to increase the packing density when used as a negative electrode, and a high-capacity battery may not be obtained. On the other hand, if it exceeds this range, the gaps between the particles in the electrode will be too small, making it difficult to ensure the conductivity between the particles, and it may be difficult to obtain preferable battery characteristics.
(10)取向比(10) Orientation ratio
热处理前的原料的取向比通常为0.005以上,优选为0.01以上,更优选为0.015以上,上限在理论上为0.67以下的范围。如果低于该范围,则有时高密度充放电特性降低。The orientation ratio of the raw material before heat treatment is usually 0.005 or more, preferably 0.01 or more, more preferably 0.015 or more, and the upper limit is theoretically 0.67 or less. If it is less than this range, high-density charge-discharge characteristics may deteriorate.
(11)长径比(11) aspect ratio
热处理前的原料的长径比理论上为1以上,上限通常为10以下,优选为8以下,更优选为5以下。如果超过其上限,制成极板时会产生条纹、无法得到均匀的涂布面,高电流密度充放电特性有时降低。The aspect ratio of the raw material before heat treatment is theoretically 1 or more, and the upper limit is usually 10 or less, preferably 8 or less, more preferably 5 or less. If the upper limit is exceeded, streaks may be generated when forming an electrode plate, a uniform coating surface may not be obtained, and the high current density charge and discharge characteristics may deteriorate.
[[[热处理温度]]][[[Heat treatment temperature]]]
作为热处理前的原料的热处理温度,通常为600℃以上,优选为1200℃以上,更优选为2000℃以上,进一步优选为2500℃以上,特别优选为2800℃以上的范围。其上限通常为3200℃以下,优选为3100℃以下的范围。温度条件如果低于该范围,则通过球形化处理等进行的散乱的石墨粒子表面的结晶修复不充分,有时拉曼R值和BET比表面积不会变小。另一方面,如果超过上述范围,则石墨的升华量有时容易变多。The heat treatment temperature of the raw material before heat treatment is usually 600°C or higher, preferably 1200°C or higher, more preferably 2000°C or higher, still more preferably 2500°C or higher, particularly preferably 2800°C or higher. The upper limit thereof is usually not higher than 3200°C, preferably not higher than 3100°C. If the temperature condition is lower than this range, the crystallization repair of the surface of scattered graphite particles by spheroidization treatment or the like is insufficient, and the Raman R value and the BET specific surface area may not be reduced. On the other hand, when it exceeds the said range, the amount of sublimation of graphite may become large easily.
[[[热处理方法]]][[[Heat treatment method]]]
热处理可以通过经过一次上述的温度范围来实现。将温度条件保持为上述范围的保持时间没有特别限制,但通常为比10秒钟长的时间,且为168小时以下。Heat treatment can be achieved by passing through the above-mentioned temperature range once. The holding time for keeping the temperature condition in the above range is not particularly limited, but is usually longer than 10 seconds and is 168 hours or less.
热处理通常在氮气等非活性气体氛围下或者在由原料石墨产生的气体而得到的非氧化性氛围下进行。但是,在包埋在焦粉(细小的沥青烧结碳)中的类型的炉中,有时在最初混合空气。在这样的情况下,也可以不必是完全非活性的气体氛围。The heat treatment is usually carried out in an inert gas atmosphere such as nitrogen gas or in a non-oxidizing atmosphere obtained from the gas generated from the raw material graphite. However, in the furnace of the type embedded in coke powder (fine pitch-sintered carbon), air is sometimes mixed initially. In such a case, a completely inert gas atmosphere may not be necessary.
作为热处理中使用的装置,没有特别的限制,例如,可使用梭式炉、隧道式炉、电炉、焦炭焙烧炉、回转炉、直接通电炉、阿切孙电炉、电阻加热炉、感应加热炉等。The apparatus used for the heat treatment is not particularly limited, and for example, a shuttle furnace, a tunnel furnace, an electric furnace, a coke roasting furnace, a rotary furnace, a direct electric furnace, an Acheson electric furnace, a resistance heating furnace, an induction heating furnace, etc. can be used. .
另外,除了上述各处理,还可以进行分级处理等各种处理。分级处理是为了得到目标粒径、除去粗粉和微粉而进行的处理。作为分级处理中使用的装置,没有特别的限制,例如,在干式筛分的情况下,可以使用旋转式筛、摇动式筛、转动式筛、振动式筛等;在干式气流分级的情况下,可以使用重力式分级机、惯性力式分级机、离心力式分级机(分粒器、旋风分离器)等;在湿式筛分的情况下,可以使用机械式湿式分级机、水力分级机、沉降分级机、离心式湿式分级机等。分级处理可以在热处理前进行,也可以在其他时间,例如热处理后进行。此外,还可以省略分级处理本身。但是,从石墨粉末负极材料的生产性的观点来看,优选在球形化处理刚结束后且在热处理之前进行分级处理。In addition, various processing such as classification processing may be performed in addition to the processing described above. Classification treatment is performed to obtain the target particle size and remove coarse powder and fine powder. As the device used in the classification process, there is no particular limitation. For example, in the case of dry sieving, rotary sieves, shaking sieves, rotary sieves, vibrating sieves, etc. can be used; in the case of dry airflow classification In the case of wet sieving, gravity classifiers, inertial force classifiers, centrifugal force classifiers (classifiers, cyclone separators), etc. can be used; in the case of wet sieving, mechanical wet classifiers, hydraulic classifiers, Sedimentation classifier, centrifugal wet classifier, etc. Grading treatment can be carried out before heat treatment, and can also be carried out at other times, such as after heat treatment. Furthermore, the classification process itself can also be omitted. However, from the viewpoint of productivity of the graphite powder negative electrode material, it is preferable to carry out the classification treatment immediately after the spheroidization treatment and before the heat treatment.
[[副材料混合]][[Vice material mix]]
除上述碳质物质以外,通过在本发明中使用的负极活性物质中含有一种以上与上述碳质物质在碳质的物性方面不同的碳质物质(碳质材料),可以进一步谋求电池性能的提高。这里所说的“碳质的物性”,是指X射线衍射参数、中值粒径、长径比、BET比表面积、取向比、拉曼R值、振实密度、真密度、微孔分布、圆形度、灰分量中的一种以上的特性。另外,作为优选的实施方式,可以举出,体积基准粒度分布以中值粒径为中心时左右不对称、含有2种以上拉曼R值不同的碳材料、或X射线参数不同等。作为其效果的一个例子,可列举通过含有天然石墨、人造石墨等石墨;乙炔黑等炭黑;针状焦等无定形碳等碳材料作为副材料来降低电阻等。它们可以单独使用一种,也可以以任意组合和任意比例组合使用2种以上。作为副材料添加时,其添加量通常为0.1质量%以上,优选为0.5质量%以上,更优选为0.6质量%以上,其上限通常为80质量%以下,优选为50质量%以下,更优选为40质量%以下,特别优选为30质量%以下的范围。如果低于该范围,则有时难以得到导电性提高的效果。如果超过上述范围,则有时会导致初期不可逆容量的增大。In addition to the above-mentioned carbonaceous substances, by containing one or more carbonaceous substances (carbonaceous materials) different from the above-mentioned carbonaceous substances in the physical properties of carbonaceous substances in the negative electrode active material used in the present invention, it is possible to further improve battery performance. improve. The "carbonaceous physical properties" mentioned here refer to X-ray diffraction parameters, median particle size, aspect ratio, BET specific surface area, orientation ratio, Raman R value, tap density, true density, micropore distribution, One or more properties among circularity and gray content. In addition, as a preferred embodiment, the volume-based particle size distribution is asymmetrical around the median particle size, contains two or more carbon materials with different Raman R values, or has different X-ray parameters. As an example of the effect, reduction of electrical resistance by including carbon materials such as graphite such as natural graphite and artificial graphite; carbon black such as acetylene black; and amorphous carbon such as needle coke as sub-materials can be cited. These may be used individually by 1 type, and may be used in combination of 2 or more types by arbitrary and arbitrary ratios. When added as an auxiliary material, its addition amount is usually 0.1 mass % or more, preferably 0.5 mass % or more, more preferably 0.6 mass % or more, and its upper limit is usually 80 mass % or less, preferably 50 mass % or less, more preferably 40% by mass or less, particularly preferably 30% by mass or less. If it is less than this range, it may be difficult to obtain the effect of improving electrical conductivity. If the above range is exceeded, the initial irreversible capacity may increase.
[制作负极[4]的电极][Making the electrode of the negative electrode [4]]
制造负极[4]可以采用通常的方法,可以与上述同样地形成负极[4]。集电体、集电体和活性物质层的厚度比、电极密度、粘合剂、极板取向比、阻抗等也与上述相同。A common method can be used to manufacture the negative electrode [4], and the negative electrode [4] can be formed in the same manner as above. The current collector, the thickness ratio between the current collector and the active material layer, electrode density, binder, plate orientation ratio, impedance, and the like are also the same as described above.
<负极[5]><Negative pole [5]>
下面,对本发明的锂二次电池中使用的负极[5]进行说明,该负极含有异取向性碳复合物作为活性物质,所述异取向性碳复合物含有2种以上取向性不同的碳质物质。Next, the negative electrode [5] used in the lithium secondary battery of the present invention will be described. The negative electrode contains, as an active material, a differently oriented carbon composite containing two or more types of carbon materials with different orientations. substance.
[负极[5]的负极活性物质][Negative electrode active material of negative electrode [5]]
下面,对负极[5]中使用的负极活性物质进行说明。Next, the negative electrode active material used in the negative electrode [5] will be described.
[[异取向性碳复合物的构成]][[Composition of Hetero-Oriented Carbon Composite]]
本发明的锂二次电池的负极[5]中使用的负极活性物质含有异取向性碳复合物,该异取向性碳复合物含有2种以上取向性不同的碳质物质。The negative electrode active material used in the negative electrode [5] of the lithium secondary battery of the present invention contains a differently oriented carbon composite containing two or more carbonaceous materials having different orientations.
这里所说的“取向性不同”,是指在用偏光显微镜观察粉末时,目视对比光学各向异性组织的各向异性单元的图案,即各向异性单元的大小、方向、数量等时,它们的大小、方向、数量等中的至少任意一个是不同的。例如可以举出,碳质1和碳质2中,一个具有向一个方向的结晶方向性,另一个具有无规的结晶方向性的情况;或者碳质1和碳质2分别都具有向一定方向的结晶方向性,但其方向不同的情况等。另外,碳质1和碳质2种的一个或两者不是单一的结晶,而是多个结晶的集合体时,将集合体的单元作为1个区域,对比其光学各向异性性组织的各向异性单元的集合图案。The "different orientation" mentioned here means that when the powder is observed with a polarizing microscope, the pattern of the anisotropic unit of the optical anisotropic structure is visually compared, that is, the size, direction, number, etc. of the anisotropic unit. At least any one of their size, direction, number, etc. is different. For example, among carbonaceous 1 and carbonaceous 2, one has crystal orientation in one direction and the other has random crystal orientation; or carbonaceous 1 and carbonaceous 2 each have crystal directionality, but the direction is different, etc. In addition, when one or both of carbonaceous 1 and carbonaceous 2 is not a single crystal but an aggregate of multiple crystals, the unit of the aggregate is regarded as one region, and the respective optical anisotropy structures are compared. An assemblage pattern of anisotropic units.
另外,各向异性碳复合物中的碳质1、碳质2共存的形态优选包含在1个二次粒子中。这里所说的“包含在1个二次粒子中”,是指取向性不同的碳质物质被物理地约束、附着的状态;通过静电的约束、附着而保持形状的状态;通过键合而被约束的状态等。这里所说的“物理的约束、附着”是指一种碳质物质夹杂在另一种碳质物质中、连在一起的状态;所谓“静电的约束、附着”是指一种碳质物质通过静电能量附着在另一种碳质物质中的状态。在该约束、附着的状态中,可以是上述的取向性不同的状态,也可以是原来的碳质物质为同样的物质。另外,所谓“通过键合而被约束的状态”是指氢键、共价键、离子键等化学键合。In addition, the form in which the carbonaceous 1 and the carbonaceous 2 coexist in the anisotropic carbon composite is preferably included in one secondary particle. The term "included in one secondary particle" here refers to a state in which carbonaceous substances with different orientations are physically restrained and attached; a state in which the shape is maintained by electrostatic restraint and adhesion; Constrained status, etc. The "physical constraint and attachment" mentioned here refers to the state that one carbonaceous substance is mixed in another carbonaceous substance and connected together; The state in which electrostatic energy is attached to another carbonaceous substance. In this constrained and adhered state, the aforementioned orientation may be different, or the original carbonaceous substance may be the same substance. In addition, the "state bound by bonding" refers to chemical bonds such as hydrogen bonds, covalent bonds, and ionic bonds.
其中,优选在一种碳质物质的至少部分表面上通过附着和/或键合而使另一种碳质物质具有取向性不同的界面的状态。具有界面与不具有界面的情况相比,在同一形状的粒子进行比较时,在充电时锂离子夹杂而引起的膨胀被分散到多方面中,从而可以防止电池劣化,从这点上看是优选的。Among them, it is preferable that another carbonaceous substance has an interface with a different orientation by adhesion and/or bonding on at least a part of the surface of one carbonaceous substance. Compared with the case of having an interface and not having an interface, when particles of the same shape are compared, the expansion caused by lithium ion inclusions during charging is dispersed in many areas, thereby preventing battery deterioration, which is preferable in this point of view. of.
该取向性不同的部分的形成可以通过与外部供给的材料和/或它们的改性物的键合而形成,或者可以通过碳质物质的表面部分的材料的改性而形成。其中,所谓覆盖,是在与碳质物质的表面的至少部分界面中具有化学键,从而显示:(1)覆盖整个表面的状态、(2)局部地覆盖碳质物质的状态、(3)选择性地覆盖部分表面的状态、(4)存在于包含化学键的极微小区域的状态。The portion having a different orientation may be formed by bonding with externally supplied materials and/or their modified products, or may be formed by modifying the material of the surface portion of the carbonaceous substance. Among them, the so-called coverage means having a chemical bond in at least part of the interface with the surface of the carbonaceous substance, thereby showing: (1) a state covering the entire surface, (2) a state of partially covering the carbonaceous substance, (3) selectivity (4) A state where a part of the surface is completely covered, and (4) a state where an extremely small region containing chemical bonds exists.
另外,在界面附近,碳质物质的取向性可以连续地变化,也可以不连续地变化。即,优选异取向性碳复合物具有取向性不同的碳质物质附着和/或键合而形成的界面,并且该界面的碳质物质的取向性不连续和/或连续地变化。In addition, in the vicinity of the interface, the orientation of the carbonaceous material may change continuously or discontinuously. That is, it is preferable that the hetero-oriented carbon composite has an interface formed by the attachment and/or bonding of carbonaceous substances with different orientations, and the orientation of the carbonaceous substances at this interface changes discontinuously and/or continuously.
另外,作为异取向性碳质复合物(A)的构成成分,只要具有结晶性即可,没有特别限定,但从单位质量的充电容量变高这点来看,优选取向性不同的碳质物质的一种以上为来自于天然石墨(D)的石墨类碳质物质(B)(下面,简称为“天然石墨类碳质物质(B)”)。In addition, the components of the hetero-oriented carbonaceous composite (A) are not particularly limited as long as they have crystallinity, but carbonaceous substances with different orientations are preferred from the viewpoint of higher charge capacity per unit mass. One or more of them are graphite-like carbonaceous substances (B) derived from natural graphite (D) (hereinafter, simply referred to as "natural graphite-like carbonaceous substances (B)").
另外,异取向性碳复合物中含有的天然石墨类碳质物质(B)相对于异取向性碳复合物的比例通常为5质量%以上,优选为20质量%以上,更优选为30质量%以上,进一步优选为40质量%以上,特别优选为50质量%以上的范围。其上限通常为99.9质量%以下,优选为99质量%以下,更优选为95质量%以下,进一步优选为90质量%以下的范围。如果低于该范围,则电极的压延时的荷重显著上升,有时会引起电极的剥离等。另一方面,如果超过该范围,则作为取向性不同的粒子的复合体的界面的粘结有时变弱。In addition, the ratio of the natural graphite-like carbonaceous material (B) contained in the heterooriented carbon composite to the heterooriented carbon composite is usually 5% by mass or more, preferably 20% by mass or more, more preferably 30% by mass Above, more preferably 40% by mass or more, particularly preferably 50% by mass or more. The upper limit thereof is usually 99.9% by mass or less, preferably 99% by mass or less, more preferably 95% by mass or less, and still more preferably 90% by mass or less. If it is less than this range, the load at the time of rolling of an electrode will increase remarkably, and peeling of an electrode, etc. may be caused. On the other hand, when this range is exceeded, the bonding at the interface of a composite of particles having different orientations may become weak.
另外,作为异取向性碳复合物的另外的构成成分,从异取向性碳复合物制备时的界面生成和界面的粘合性提高的观点来看,优选一种以上取向性不同的碳质物质为选自下面的(1)~(5)中的碳质物质(C)。In addition, as another constituent component of the differently oriented carbon composite, from the viewpoint of forming an interface during preparation of the differently oriented carbon composite and improving the adhesiveness of the interface, one or more carbonaceous substances having different orientations are preferable. It is a carbonaceous substance (C) selected from the following (1) to (5).
(1)从煤类焦炭、石油类焦炭、炉黑、乙炔黑和沥青类碳纤维中选择的碳化物;(1) Carbide selected from coal coke, petroleum coke, furnace black, acetylene black and pitch carbon fiber;
(2)将选自沥青原料、芳香族烃、N环化合物、S环化合物、聚苯、有机合成高分子、天然高分子、热塑性树脂和热固性树脂中的有机物和/或其热分解产物作为前体的碳化物;(2) Organic substances and/or thermal decomposition products selected from asphalt raw materials, aromatic hydrocarbons, N-ring compounds, S-ring compounds, polyphenylene, organic synthetic polymers, natural polymers, thermoplastic resins and thermosetting resins are used as precursors solid carbides;
(3)将(2)的有机物溶解在低分子有机溶剂中而得到溶液的热分解物作为前体的碳化物;(3) dissolving the organic substance of (2) in a low molecular weight organic solvent to obtain a carbide of the thermal decomposition product of the solution as a precursor;
(4)含有有机物的气体的碳化物;(4) Carbide of gas containing organic matter;
(5)(1)~(4)的石墨化物。(5) Graphites of (1) to (4).
对于(2)来说,只要是可以碳化的物质即可,没有特别限定,可以列举,沥青原料、苊烯、十环烯、蒽、菲等芳香族烃;吩嗪、吖啶等N环化合物;噻吩、联二噻吩等S环化合物;联苯、三联苯等聚苯;聚氯乙烯、聚乙烯醇、聚乙烯醇缩丁醛、它们的不溶解处理物、含氮的聚丙烯腈、聚吡咯等有机高分子;含硫的聚噻吩、聚苯乙烯等有机高分子;以纤维素、木质素、甘露聚糖、聚半乳糖醛酸、壳聚糖、蔗糖为代表的多糖类等天然高分子;聚苯硫醚、聚苯醚等热塑性树脂;糠醇树脂、酚醛树脂、酰亚胺树脂等热固性树脂;或者将它们溶解于苯、甲苯、二甲苯、喹啉、正己烷等低分子有机溶剂中而形成的溶液等有机物;可碳化的气体等。Regarding (2), it is not particularly limited as long as it is a carbonizable substance, and examples thereof include aromatic hydrocarbons such as pitch raw materials, acenaphthylene, decacyclene, anthracene, and phenanthrene; and N-ring compounds such as phenazine and acridine. ; S-ring compounds such as thiophene and dithiophene; polyphenylenes such as biphenyl and terphenyl; polyvinyl chloride, polyvinyl alcohol, polyvinyl butyral, their insoluble processed products, nitrogen-containing polyacrylonitrile, poly Organic polymers such as pyrrole; organic polymers such as sulfur-containing polythiophene and polystyrene; natural polysaccharides such as cellulose, lignin, mannan, polygalacturonic acid, chitosan, and sucrose Polymers; thermoplastic resins such as polyphenylene sulfide and polyphenylene ether; thermosetting resins such as furfuryl alcohol resins, phenolic resins, and imide resins; Organic substances such as solutions formed in solvents; carbonizable gases, etc.
这些当中,由于沥青原料的残炭率高,可以制作收率高的材料,故优选。另外,本说明书中,“沥青原料”是沥青和属于沥青的物质,是指可以通过适当的处理进行碳化和/或石墨化的物质。作为具体的沥青原料的例子,可以使用焦油、重油或沥青等。作为焦油的具体例子,可列举煤焦油、石油类焦油等。作为重油的具体例子,可以列举石油类重油的接触分解油、热分解油、常压残油、减压残油等。另外,作为沥青的具体例子,可列举煤焦油沥青、石油类沥青、合成沥青等。其中,煤焦油沥青的芳香性高,故优选。这些沥青原料可以单独使用任意一种,也可以以任意组合和比例组合使用2种以上。Among these, the asphalt raw material is preferable because it can produce a high-yield material due to its high carbon residue rate. In addition, in this specification, a "pitch raw material" refers to pitch and a substance belonging to pitch, and refers to a substance that can be carbonized and/or graphitized by appropriate treatment. As an example of a specific pitch raw material, tar, heavy oil, pitch, etc. can be used. Specific examples of tar include coal tar, petroleum tar, and the like. Specific examples of the heavy oil include catalysis oil, pyrolysis oil, atmospheric residual oil, vacuum residual oil, and the like of petroleum heavy oil. In addition, specific examples of pitch include coal tar pitch, petroleum pitch, synthetic pitch, and the like. Among them, coal tar pitch is preferable because of its high aromaticity. Any one of these pitch raw materials may be used alone, or two or more of them may be used in combination in any desired ratio.
作为(3)的优选例子,可列举将上述(2)的有机物溶解于苯、甲苯、二甲苯、喹啉、正己烷等低分子有机溶剂中而得到溶液,再将该溶液的热分解物作为前体的碳化物。As a preferred example of (3), it is possible to enumerate a solution obtained by dissolving the above-mentioned organic substance in (2) in low-molecular organic solvents such as benzene, toluene, xylene, quinoline, and n-hexane, and then obtain a solution by thermally decomposing the solution as Precursor carbides.
关于(4),可列举甲烷、乙烷、丙烷、苯、乙炔、乙烯等烃化合物;一氧化碳等。Regarding (4), hydrocarbon compounds such as methane, ethane, propane, benzene, acetylene, and ethylene; carbon monoxide, and the like are exemplified.
异取向性碳复合物中含有的碳质物质(C)的比例通常为0.1质量%以上,优选为1质量%以上,更优选为5质量%以上,进一步优选为10质量%以上的范围。其上限没有特别的限制,只要具有取向性不同的界面即可。如果低于该范围,则异取向性碳复合物中的界面的粘结有时变弱。另外,如果超过该范围,则由于含有碳质物质(C)引起的压制时的粒子的变形抑制效果减少,循环特性降低。The ratio of the carbonaceous substance (C) contained in the heterooriented carbon composite is usually 0.1% by mass or more, preferably 1% by mass or more, more preferably 5% by mass or more, and still more preferably 10% by mass or more. The upper limit is not particularly limited, as long as there are interfaces with different orientations. If it is less than this range, the bonding of the interface in the hetero-oriented carbon composite may become weak. In addition, if it exceeds this range, the effect of suppressing the deformation of the particles during pressing due to the carbonaceous substance (C) is reduced, and the cycle characteristics are reduced.
作为本发明中的锂二次电池中使用的负极活性物质的更优选的构成,从电极压延时的压制荷重的观点以及作为复合体的界面的粘合性的平衡的观点来看,优选异取向性碳复合物同时含有一种以上的天然石墨类碳质物质(B)和一种以上的碳质物质(C)。As a more preferable configuration of the negative electrode active material used in the lithium secondary battery in the present invention, from the viewpoint of the pressing load at the time of electrode rolling and the viewpoint of the balance of the adhesiveness of the interface as the composite, the hetero-orientation is preferable. The carbonaceous composite contains more than one natural graphite-like carbonaceous substance (B) and more than one carbonaceous substance (C).
由于与上述同样的理由,异取向性碳复合物中的天然石墨类碳质物质(B)和碳质物质(C)的质量比率(天然石墨类碳质物质(B)/碳质物质(C))通常为20/80以上,优选为40/60以上,更优选为60/40以上,进一步优选为70/30以上的范围。其上限为99.9/0.1以下,优选为99/1以下,更优选为95/5以下的范围。如果超过该范围(如果天然石墨类碳质物质(B)的比率过大),则由碳质物质(C)产生的界面的粘合性有降低的可能性。另一方面,如果低于该范围(如果天然石墨类碳质物质(B)的比率过小),则电极压延时的压制荷重显著变大,在压延时有时会剥离。For the same reason as above, the mass ratio of the natural graphite-like carbonaceous substance (B) and the carbonaceous substance (C) in the hetero-oriented carbon composite (natural graphite-like carbonaceous substance (B)/carbonaceous substance (C) )) is usually 20/80 or more, preferably 40/60 or more, more preferably 60/40 or more, and still more preferably 70/30 or more. The upper limit thereof is 99.9/0.1 or less, preferably 99/1 or less, more preferably 95/5 or less. If this range is exceeded (if the ratio of the natural graphite-like carbonaceous substance (B) is too large), the adhesion of the interface by the carbonaceous substance (C) may decrease. On the other hand, if it is below this range (if the ratio of the natural graphite-like carbonaceous substance (B) is too small), the pressing load at the time of electrode rolling becomes significantly large, and peeling may occur during rolling.
作为含有天然石墨类碳质物质(B)和碳质物质(C)的异取向性碳复合物,只要不超出本发明的主旨的范围,则可以采取任意的形态,下面示出一例。The heterooriented carbon composite containing natural graphite-like carbonaceous material (B) and carbonaceous material (C) can take any form as long as it does not exceed the scope of the present invention. An example is shown below.
(1)天然石墨类碳质物质(B)的整个表面或部分表面附着和/或覆盖和/或键合有碳质物质(C)的形态;(1) The entire surface or part of the surface of the natural graphite-like carbonaceous substance (B) is attached and/or covered and/or bonded with the carbonaceous substance (C);
(2)碳质物质(C)与天然石墨类碳质物质(B)的整个表面或部分表面粘结,2个以上的天然石墨类碳质物质(B)和/或碳质物质(C)复合化的形态;(2) The carbonaceous substance (C) is bonded to the entire surface or part of the surface of the natural graphite-like carbonaceous substance (B), and two or more natural graphite-like carbonaceous substances (B) and/or carbonaceous substances (C) Composite form;
(3)上述(1)和(2)以任意的比例混合的形态。(3) A form in which the above (1) and (2) are mixed in an arbitrary ratio.
另外,上述天然石墨类碳质物质(B)还可以与碳质物质(C)替换,另外,作为该异取向性碳复合物的具体的复合方式,可列举在作为核的粒子的表面附着和/或覆盖和/或键合有碳质物质的形态;多个作为核的粒子的表面附着和/或覆盖和/或键合的形态;或者作为核的粒子非平行地造粒的状态等。这里所说的非平行地进行造粒的状态,是指具有一定结晶性的粒子以向着随机的方向的状态被其他的碳质物质固定,从而成为具有显示异取向性的键合的状态。In addition, the above-mentioned natural graphite-like carbonaceous substance (B) can also be replaced with the carbonaceous substance (C). In addition, as a specific compounding method of the hetero-oriented carbon composite, it can be enumerated that it is attached to the surface of the particle as the nucleus and / or covered and / or bonded form of carbonaceous substances; the surface of a plurality of core particles attached and / or covered and / or bonded form; or the state of non-parallel granulation of core particles. The state of non-parallel granulation here refers to a state in which particles having a certain crystallinity are fixed by other carbonaceous substances in a state of being oriented in a random direction, and are bonded to exhibit hetero-orientation.
[[异取向性碳复合物的制备]][[Preparation of Hetero-Oriented Carbon Composite]]
对于异取向性碳复合物的制备,在后面的“异取向性碳复合物的制造方法1和制造方法2”中进行叙述,在制备异取向性碳复合物时,可以通过施加热处理来谋求提高结晶性,并可以增加每单位重量的容量。作为热处理温度,通常为400℃以上,优选为1000℃以上,更优选为2000℃以上,进一步优选为2400℃以上,特别优选为2800℃以上的范围。其上限通常为3400℃以下,优选为3200℃以下的范围。如果低于该范围,则不能充分提高结晶性,并有可能不能获得每单位重量的容量增加的效果。另一方面,如果超过该范围,则由于碳的升华导致的损失不能忽视,有可能引起收率的减少。For the preparation of differently oriented carbon composites, it is described in the following "Manufacturing method 1 and 2 of differently oriented carbon composites". When preparing differently oriented carbon composites, it can be improved by applying heat treatment. Crystalline and can increase capacity per unit weight. The heat treatment temperature is usually 400°C or higher, preferably 1000°C or higher, more preferably 2000°C or higher, still more preferably 2400°C or higher, particularly preferably 2800°C or higher. The upper limit thereof is usually not higher than 3400°C, preferably not higher than 3200°C. If it is less than this range, the crystallinity cannot be sufficiently improved, and there is a possibility that the capacity increase effect per unit weight cannot be obtained. On the other hand, if it exceeds this range, the loss due to the sublimation of carbon cannot be ignored, which may cause a decrease in yield.
[[异取向性碳复合物的性质]][[Properties of Hetero-Oriented Carbon Composite]]
对于关于异取向性碳复合物的性质,优选同时满足下面所示的(1)~(5)中的任意一项或多项。With respect to the properties of the heterooriented carbon composite, it is preferable to simultaneously satisfy any one or more of (1) to (5) shown below.
(1)圆形度(1) Circularity
使用圆形度作为异取向性碳复合物的球形的程度,异取向性碳复合物的粒径为3~40μm范围的粒子的圆形度通常为0.1以上,优选为0.5以上,更优选为0.8以上,进一步优选为0.85以上,最优选为0.9以上。圆形度大时,高电流密度充放电特性提高,因此优选。圆形度用下式定义,圆形度为1时,为理论真球。Using circularity as the degree of spherical shape of the heterooriented carbon composite, the circularity of the particles of the heterooriented carbon composite having a particle diameter in the range of 3 to 40 μm is usually 0.1 or more, preferably 0.5 or more, more preferably 0.8 or more, more preferably 0.85 or more, and most preferably 0.9 or more. When the circularity is large, the high current density charge and discharge characteristics are improved, which is preferable. The circularity is defined by the following formula. When the circularity is 1, it is a theoretical true sphere.
圆形度=(具有与粒子投影形状相同面积的等效圆的周长)/(粒子投影形状的实际周长)Circularity = (perimeter of an equivalent circle having the same area as the particle projected shape)/(actual perimeter of the particle projected shape)
作为圆形度的值使用如下测定的值:使用例如流动式粒子图像分析装置(例如,Sysmex Industrial公司制造的FPIA),将约0.2g试样分散于作为表面活性剂的聚氧乙烯(20)山梨糖醇酐单月桂酸酯的0.2质量%水溶液(约50mL)中,以60W的输出功率照射1分钟28kHz的超声波后,指定0.6~400μm为检测范围,对粒径3~40μm范围的粒子进行测定。As the value of circularity, the value measured as follows is used: using, for example, a flow type particle image analyzer (for example, FPIA manufactured by Sysmex Industrial Co., Ltd.), about 0.2 g of a sample is dispersed in polyoxyethylene (20) as a surfactant. In a 0.2% by mass aqueous solution of sorbitan monolaurate (approximately 50mL), after irradiating 28kHz ultrasonic waves with an output power of 60W for 1 minute, specify a detection range of 0.6 to 400μm, and perform a test on particles in the range of particle diameters from 3 to 40μm. Determination.
提高圆形度的方法没有特别的限制,但通过实施球形化处理使之成为球形可以使制成电极体时粒子间空隙的形状整齐统一,故优选。作为球形化处理的例子,可以举出,通过施加剪切力、压缩力来机械地接近球形的方法,通过粘合剂或粒子自身具有的附着力将多个微粒造粒的机械/物理处理方法等。There is no particular limitation on the method of improving the circularity, but it is preferable to make the shape of the gap between the particles uniform when the electrode body is produced by performing a spheroidization treatment to make it spherical. Examples of spheroidization treatment include a method of mechanically approaching a spherical shape by applying a shear force or a compressive force, and a mechanical/physical treatment method of granulating a plurality of fine particles by the adhesive force possessed by a binder or the particles themselves. wait.
(2)拉曼R值、拉曼半值宽度(2) Raman R value, Raman half value width
使用氩离子激光拉曼光谱法测定的异取向性碳复合物的拉曼R值通常为0.01以上,优选为0.02以上,更优选为0.04以上,其上限通常为0.35以下,优选为0.30以下,更优选为0.25以下的范围。如果拉曼R值低于该范围,则粒子表面的结晶性过高,随着充放电Li进入层间的位点有时变少。即,充电接受性有时降低。另一方面,如果超过该范围,则粒子表面的结晶性降低,与非水电解液的反应性增加,有时导致效率降低或产生的气体增加。The Raman R value of the heterooriented carbon composite measured by argon ion laser Raman spectroscopy is usually 0.01 or more, preferably 0.02 or more, more preferably 0.04 or more, and its upper limit is usually 0.35 or less, preferably 0.30 or less, more It is preferably in the range of 0.25 or less. If the Raman R value is lower than this range, the crystallinity of the particle surface may be too high, and there may be fewer sites for Li to enter the interlayer due to charge and discharge. That is, charge acceptance may be lowered. On the other hand, if it exceeds this range, the crystallinity of the particle surface decreases, the reactivity with the non-aqueous electrolyte solution increases, and the efficiency may decrease or the generated gas may increase.
另外,本发明的碳材料在1580cm-1附近的拉曼半值宽度没有特别的限制,但通常为5cm-1以上,优选为10cm-1以上,另外,其上限通常为40cm-1以下,优选为35cm-1以下,更优选为30cm-1以下的范围。如果拉曼半值宽度低于该范围,则粒子表面的结晶性过高,随着充放电Li进入层间的位点有时变少。即,充电接受性有时降低。另一方面,如果高于该范围,则粒子表面的结晶性降低,与非水电解液的反应性增加,有时导致效率降低或产生的气体增加。In addition, the Raman half-value width of the carbon material of the present invention is not particularly limited in the vicinity of 1580 cm -1 , but it is usually 5 cm -1 or more, preferably 10 cm -1 or more, and its upper limit is usually 40 cm -1 or less, preferably It is in the range of 35 cm -1 or less, more preferably 30 cm -1 or less. If the Raman half-value width is below this range, the crystallinity of the particle surface may be too high, and there may be fewer sites for Li to enter the interlayer due to charge and discharge. That is, charge acceptance may be lowered. On the other hand, if it exceeds this range, the crystallinity of the particle surface decreases, the reactivity with the non-aqueous electrolyte solution increases, and the efficiency may decrease or the generated gas may increase.
拉曼光谱的测定如下进行:使用拉曼分光器(例如日本分光社制造的拉曼分光器),使试样自然落下并填充在测定池内,对池内的试样表面照射氩离子激光,同时,使池在与激光垂直的面内旋转。对于得到的拉曼光谱,测定1580cm-1附近的峰PA的强度IA和1360cm-1附近的峰PB的强度IB,计算其强度比R(R=IB/IA),将其定义为碳材料的拉曼R值。另外,测定得到的拉曼光谱在1580cm-1附近的峰PA的半值宽度,将其定义为碳材料的拉曼半值宽度。The measurement of the Raman spectrum is carried out as follows: Using a Raman spectrometer (such as a Raman spectrometer manufactured by JASCO Corporation), the sample is naturally dropped and filled in the measurement cell, and the surface of the sample in the cell is irradiated with an argon ion laser. At the same time, Rotate the cell in a plane perpendicular to the laser. For the obtained Raman spectrum, the intensity I A of the peak PA near 1580 cm -1 and the intensity I B of the peak P B near 1360 cm -1 were measured , and the intensity ratio R (R= IB / IA ) was calculated. It is defined as the Raman R value of the carbon material. In addition, the half-value width of the peak PA in the vicinity of 1580 cm −1 in the obtained Raman spectrum was measured, and this was defined as the Raman half-value width of the carbon material.
另外,这里的拉曼测定条件如下。In addition, the Raman measurement conditions here are as follows.
·氩离子激光波长:514.5nmArgon ion laser wavelength: 514.5nm
·试样上的激光功率:15~25mW·Laser power on the sample: 15~25mW
·分辨率:10~20cm-1 ·Resolution: 10~20cm -1
·测定范围:1100cm-1~1730cm-1 · Measuring range: 1100cm -1 ~ 1730cm -1
·拉曼R值、拉曼半值宽度分析:本底处理Raman R value, Raman half-value width analysis: background processing
·光滑处理:单纯平均、卷积5点·Smooth processing: simple average, convolution 5 points
(3)振实密度(3) Tap density
异取向性碳复合物的振实密度通常为0.55g/cm3以上,优选为0.70g/cm3以上,更优选为0.9g/cm3以上,特别优选为1g/cm3以上。另外,优选为2.0g/cm3以下,更优选为1.8g/cm3以下,进一步优选为1.7g/cm3以下,特别优选为1.5g/cm3以下。振实密度如果低于该范围,则在作为负极使用时,填充密度很难提高,有时不能得到高容量的电池。另一方面,如果高于该范围,则电极中的粒子间的空隙过少,难以确保粒子间的导电性,有时难以得到优选的电池特性。The tap density of the heterooriented carbon composite is usually 0.55 g/cm 3 or more, preferably 0.70 g/cm 3 or more, more preferably 0.9 g/cm 3 or more, particularly preferably 1 g/cm 3 or more. In addition, it is preferably 2.0 g/cm 3 or less, more preferably 1.8 g/cm 3 or less, still more preferably 1.7 g/cm 3 or less, particularly preferably 1.5 g/cm 3 or less. If the tap density is lower than this range, it is difficult to increase the packing density when used as a negative electrode, and a high-capacity battery may not be obtained. On the other hand, if it exceeds this range, the gaps between the particles in the electrode will be too small, making it difficult to ensure the conductivity between the particles, and it may be difficult to obtain preferable battery characteristics.
在本发明中,振实密度如下定义:使试样通过孔径300μm的筛子落在20cm3的振实容器内,直到试样充满容器的上端面后,使用粉末密度测定器(例如,seishin企业公司制造的Tap densor)进行1000次冲程长度10mm的振动,将由此时的体积和试样的重量求得的密度定义为振实密度。In the present invention, the tap density is defined as follows: the sample is passed through a sieve with an aperture of 300 μm and dropped in a 20 cm container for vibration until the sample is filled with the upper end surface of the container, and a powder density measuring device (for example, Seishin Enterprise Co., Ltd. The manufactured Tap densor) was vibrated 1000 times with a stroke length of 10 mm, and the density obtained from the volume at that time and the weight of the sample was defined as the tap density.
(4)BET比表面积(4) BET specific surface area
使用BET法测定的异取向性碳复合物的比表面积通常为0.1m2/g以上,优选为0.7m2/g以上,更优选为1.0m2/g以上,进一步优选为1.2m2/g以上。其上限通常为100m2/g以下,优选为25m2/g以下,更优选为15m2/g以下,进一步优选为10m2/g以下。比表面积的值如果低于该范围,则作为负极材料使用时,在充电时锂的接受性容易变差,锂容易在电极表面析出。另一方面,如果高于上述范围,则用作负极材料时,与非水电解液的反应性增加,产生的气体容易变多,有时难以得到优选的电池。The specific surface area of the heterooriented carbon composite measured by the BET method is usually 0.1 m 2 /g or more, preferably 0.7 m 2 /g or more, more preferably 1.0 m 2 /g or more, and still more preferably 1.2 m 2 /g above. The upper limit thereof is usually 100 m 2 /g or less, preferably 25 m 2 /g or less, more preferably 15 m 2 /g or less, still more preferably 10 m 2 /g or less. When the value of the specific surface area is below this range, when used as a negative electrode material, lithium acceptance during charging tends to deteriorate, and lithium tends to be deposited on the electrode surface. On the other hand, if it is higher than the above range, when used as a negative electrode material, the reactivity with the non-aqueous electrolytic solution increases, the gas generated tends to increase, and it may be difficult to obtain a preferable battery.
BET比表面积定义为如下测定的值:使用表面积计(例如,大仓理研制造的全自动表面积测定装置),在氮气流通下,在350℃下对试样进行15分钟预干燥,然后,使用氮气相对于大气压的相对压力值准确调整为0.3的氮氦混合气体,通过采用气体流动法的氮吸附BET 1点法测定。The BET specific surface area is defined as a value measured by pre-drying the sample at 350° C. for 15 minutes under nitrogen flow using a surface area meter (for example, a fully automatic surface area measuring device manufactured by Okura Riken), and then, using nitrogen gas Nitrogen-helium mixed gas whose relative pressure value relative to the atmospheric pressure is accurately adjusted to 0.3 is measured by the nitrogen adsorption BET 1-point method using the gas flow method.
(5)体积基准平均粒径(5) Volume-based average particle size
异取向性碳复合物的体积基准平均粒径是通过激光衍射/散射法求得的体积基准的平均粒径(中值粒径),通常为1μm以上,优选为3μm以上,更优选为5μm以上,进一步优选为7μm以上。另外,其上限通常为100μm以下,优选为50μm以下,更优选为40μm以下,进一步优选为30μm以下,特别优选为25μm以下。如果低于上述范围,则不可逆容量增大,有时导致初期电池容量的损失。另外,如果超过上述范围,则在制成电极极板时,容易形成不均匀的涂布面,有时在电池制作工序上是不优选的。The volume-based average particle diameter of the heterooriented carbon composite is the volume-based average particle diameter (median particle diameter) obtained by the laser diffraction/scattering method, and is usually 1 μm or more, preferably 3 μm or more, more preferably 5 μm or more , and more preferably 7 μm or more. In addition, the upper limit thereof is usually 100 μm or less, preferably 50 μm or less, more preferably 40 μm or less, still more preferably 30 μm or less, particularly preferably 25 μm or less. If it is less than the above-mentioned range, the irreversible capacity may increase, resulting in loss of initial battery capacity. Moreover, if it exceeds the said range, it will become easy to form an uneven coating surface at the time of making an electrode pad, and it may be unpreferable in a battery manufacturing process.
在本发明中,体积基准平均粒径用中值粒径来定义,所述中值粒径通过以下方法测定:使碳粉末分散在作为表面活性剂的聚氧乙烯(20)山梨糖醇酐单月桂酸酯的0.2质量%水溶液(约1mL)中,使用激光衍射式粒度分布计(例如,堀场制作所公司制造的LA-700)来测定。In the present invention, the volume-based average particle diameter is defined by a median particle diameter measured by dispersing carbon powder in polyoxyethylene (20) sorbitan as a surfactant. In a 0.2% by mass aqueous solution (about 1 mL) of monolaurate, it measures using a laser diffraction particle size distribution meter (for example, LA-700 manufactured by Horiba, Ltd.).
从电池特性的平衡的观点来看,优选除了上述(1)~(5)项以外,还满足下述(6)~(11)项中的一项以上。From the viewpoint of the balance of battery characteristics, it is preferable to satisfy one or more of the following items (6) to (11) in addition to the above items (1) to (5).
(6)X射线参数(6) X-ray parameters
异取向性碳复合物通过采用学振法的X射线衍射求得的晶格面(002)的d值(层间距离)优选为0.335nm以上,且通常为0.340nm以下,优选为0.337nm以下。如果低于该范围,则结晶性降低,有可能会增加初期不可逆容量。另外,下限的0.335为石墨的理论值。另外,通过采用学振法的X射线衍射求得的碳材料的微晶大小(Lc)通常为30nm以上,优选为50nm以上,更优选为80nm以上的范围。如果低于该范围,则结晶性降低,有可能增加初期不可逆容量。The d value (interlayer distance) of the lattice plane (002) obtained by X-ray diffraction using the Gakushin method of the hetero-oriented carbon composite is preferably 0.335 nm or more, and usually 0.340 nm or less, preferably 0.337 nm or less . If it is less than this range, the crystallinity may decrease, which may increase the initial irreversible capacity. In addition, the lower limit of 0.335 is the theoretical value of graphite. In addition, the crystallite size (Lc) of the carbon material obtained by X-ray diffraction using the Gakushin method is usually in the range of 30 nm or more, preferably 50 nm or more, more preferably 80 nm or more. If it is less than this range, the crystallinity may decrease, which may increase the initial irreversible capacity.
(7)灰分(7) Ash content
异取向性碳复合物中所含的灰分相对于异取向性碳复合物的总质量通常为1质量%以下,优选为0.5质量%以下,特别优选为0.1质量%以下,其下限通常为1ppm以上。如果超过上述范围,则充放电时与非水电解液的反应而引起的电池性能的劣化不能忽视。如果低于该范围,则在制造上需要较长时间和能量以及用于防止污染的设备,有时成本会上升。The ash contained in the heterooriented carbon composite is usually 1% by mass or less, preferably 0.5% by mass or less, particularly preferably 0.1% by mass or less with respect to the total mass of the heterooriented carbon composite, and the lower limit thereof is usually 1 ppm or more . If the above-mentioned range is exceeded, the degradation of the battery performance due to the reaction with the non-aqueous electrolytic solution during charge and discharge cannot be ignored. If it is less than this range, long time and energy are required for manufacturing and equipment for preventing pollution, and the cost may increase.
(8)微孔分布(8) Micropore distribution
通过水银孔隙率计(水银压入法)求得的异取向性碳复合物的相当于直径0.01μm~1μm的粒子内的空隙、由于粒子表面的高低不平造成的凹凸的量通常为0.001mL/g以上,优选为0.002mL/g以上,其上限通常为0.6mL/g以下,优选为0.4mL/g以下,更优选为0.3mL/g以下的范围。如果超过该范围,则制造成极板时有时需要大量的粘合剂。另一方面,如果低于该范围,则高电流密度充放电特性降低,并且,有时不能得到充放电时的电极膨胀收缩的缓和效果。The amount of voids in particles with a diameter of 0.01 μm to 1 μm and unevenness on the surface of the particles of the heterooriented carbon composite obtained by mercury porosimetry (mercury porosimetry) is usually 0.001 mL/ g or more, preferably 0.002 mL/g or more, and the upper limit thereof is usually 0.6 mL/g or less, preferably 0.4 mL/g or less, more preferably 0.3 mL/g or less. If it exceeds this range, a large amount of binder may be required when manufacturing an electrode plate. On the other hand, if it is less than this range, the high current density charge-discharge characteristics deteriorate, and the effect of alleviating electrode expansion and contraction during charge-discharge may not be obtained.
另外,总微孔容积优选为0.1mL/g以上,更优选为0.25mL/g以上,其上限通常为10mL/g以下,优选为5mL/g以下,更优选为2mL/g以下的范围。如果超过该范围,则在制成基板时有时需要大量的粘合剂。如果低于该范围,则在制成极板时有时不能得到增稠剂或粘结剂的分散效果。In addition, the total pore volume is preferably 0.1 mL/g or more, more preferably 0.25 mL/g or more, and its upper limit is usually 10 mL/g or less, preferably 5 mL/g or less, more preferably 2 mL/g or less. If it exceeds this range, a large amount of adhesive may be required to form a substrate. If it is less than this range, the dispersion effect of the thickener or the binder may not be obtained when forming an electrode plate.
另外,平均微孔径优选为0.05μm以上,更优选为0.1μm以上,进一步优选为0.5μm以上,其上限通常为50μm以下,优选为20μm以下,更优选为10μm以下的范围。如果超过该范围,则有时需要大量的粘合剂。另一方面,如果低于该范围,则有时高电流密度充放电特性降低。In addition, the average pore diameter is preferably 0.05 μm or more, more preferably 0.1 μm or more, even more preferably 0.5 μm or more, and the upper limit thereof is usually 50 μm or less, preferably 20 μm or less, more preferably 10 μm or less. If this range is exceeded, a large amount of adhesive is sometimes required. On the other hand, if it is less than this range, the high current density charge-discharge characteristics may deteriorate.
作为用于水银孔隙率计的装置,可以使用水银孔隙率计(autopore9520;micrometritics公司制造)。称量约0.2g试样(负极材料),密封入粉末用容器中,在室温、真空下(50μmHg以下)进行10分钟脱气,实施前处理。接着,减压至4psia(约28kPa),导入水银,从4psia(约28kPa)阶段状升压至40000psia(约280MPa),然后降压至25psia(约170kPa)。升压时的阶段数为80阶段以上,在各阶段中,在10秒的平衡时间后,测定水银压入量。使用Washburn式从这样得到的水银压入曲线计算出微孔分布。另外,水银的表面张力(γ)为485dyne/cm,接触角(φ)为140°。平均微孔径使用累积微孔体积达到50%时的微孔径。As an apparatus used for the mercury porosimeter, a mercury porosimeter (autopore9520; manufactured by Micrometrics Inc.) can be used. About 0.2 g of the sample (negative electrode material) was weighed, sealed in a powder container, and degassed at room temperature under vacuum (50 μmHg or less) for 10 minutes to perform pretreatment. Next, the pressure is reduced to 4psia (about 28kPa), mercury is introduced, the pressure is raised from 4psia (about 28kPa) to 40000psia (about 280MPa) in steps, and then the pressure is lowered to 25psia (about 170kPa). The number of stages during the pressurization was 80 or more, and the amount of mercury intrusion was measured after an equilibration time of 10 seconds in each stage. The micropore distribution was calculated from the mercury intrusion curve thus obtained using the Washburn formula. In addition, the surface tension (γ) of mercury is 485 dyne/cm, and the contact angle (φ) is 140°. As the average pore diameter, the pore diameter at which the cumulative pore volume reaches 50% is used.
(9)真密度(9) True density
异取向性碳复合物的真密度通常为2.0g/cm3以上,优选为2.1g/cm3以上,更优选为2.2g/cm3以上,进一步优选为2.22g/cm3以上,其上限为2.26g/cm3以下。上限为石墨的理论值。如果低于该范围,则碳的结晶性过低,初期不可逆容量有时会增大。在本发明中,真密度定义为通过使用丁醇的液相置换法(比重计法)测定的值。The true density of the hetero-oriented carbon composite is usually 2.0 g/cm 3 or more, preferably 2.1 g/cm 3 or more, more preferably 2.2 g/cm 3 or more, still more preferably 2.22 g/cm 3 or more, and the upper limit is 2.26g/cm 3 or less. The upper limit is the theoretical value of graphite. If it is less than this range, the crystallinity of carbon may be too low, and the initial irreversible capacity may increase. In the present invention, the true density is defined as a value measured by a liquid phase displacement method (pycnometer method) using butanol.
(10)取向比(粉末)(10) Orientation ratio (powder)
异取向性碳复合物的取向比通常为0.005以上,优选为0.01以上,更优选为0.015以上,上限在理论上为0.67以下的范围。如果低于该范围,则有时高密度充放电特性降低。The orientation ratio of the hetero-oriented carbon composite is usually 0.005 or more, preferably 0.01 or more, more preferably 0.015 or more, and the upper limit is theoretically 0.67 or less. If it is less than this range, high-density charge-discharge characteristics may deteriorate.
取向比通过X射线衍射测定。使用非对称皮尔森VII作为分布函数,拟合通过X射线衍射得到的碳的(110)衍射和(004)衍射的峰,进行峰分离,分别计算出(110)衍射和(004)衍射的峰的积分强度。由得到的积分强度计算出由(110)衍射积分强度/(004)衍射积分强度表示的比值,将该比值定义为活性物质取向比。The orientation ratio was measured by X-ray diffraction. Using asymmetric Pearson VII as a distribution function, fit the peaks of (110) diffraction and (004) diffraction of carbon obtained by X-ray diffraction, perform peak separation, and calculate the peaks of (110) diffraction and (004) diffraction respectively integral strength. A ratio represented by (110) diffraction integrated intensity/(004) diffraction integrated intensity was calculated from the obtained integrated intensity, and this ratio was defined as the active material orientation ratio.
这里的X射线衍射测定条件如下。另外,“2θ”表示衍射角。The X-ray diffraction measurement conditions here are as follows. In addition, "2θ" represents a diffraction angle.
·靶:Cu(Kα射线)石墨单色光度仪Target: Cu (Kα ray) graphite monochromator
·狭缝:发散狭缝=1度,受光狭缝=0.1mm,散射狭缝=1度· Slit: divergence slit = 1 degree, light receiving slit = 0.1mm, scattering slit = 1 degree
·测定范围和步进角/测定时间・Measurement range and step angle/measurement time
(110)面:76.5度≤2θ≤78.5度0.01度/3秒(110) surface: 76.5 degrees ≤ 2θ ≤ 78.5 degrees 0.01 degrees / 3 seconds
(004)面:53.5度≤2θ≤56.0度0.01度/3秒(004) plane: 53.5 degrees ≤ 2θ ≤ 56.0 degrees 0.01 degrees / 3 seconds
(11)长径比(粉末)(11) aspect ratio (powder)
异取向性碳复合物的长径比理论上为1以上,上限为10以下,优选为8以下,更优选为5以下。如果超过其上限,制成极板时会产生条纹、无法得到均匀的涂布面,高电流密度充放电特性有时降低。The aspect ratio of the heterooriented carbon composite is theoretically 1 or more, and the upper limit is 10 or less, preferably 8 or less, more preferably 5 or less. If the upper limit is exceeded, streaks may be generated when forming an electrode plate, a uniform coating surface may not be obtained, and the high current density charge and discharge characteristics may deteriorate.
另外,长径比用三维观察时碳材料粒子的最长径A和与其垂直的最短径B的比值A/B来表示。粒子的观察通过可以放大观察的扫描型电子显微镜来进行。选择固定在厚度50微米以下的金属的端面上的任意50个石墨粒子,旋转、倾斜固定有试样的载物台,分别测定这些粒子的A、B,求出A/B的平均值。In addition, the aspect ratio is represented by the ratio A/B of the longest diameter A of the carbon material particle and the shortest diameter B perpendicular thereto when viewed three-dimensionally. Observation of particles is performed with a scanning electron microscope capable of magnified observation. Select any 50 graphite particles fixed on the end face of the metal with a thickness of 50 microns or less, rotate and tilt the stage on which the sample is fixed, measure A and B of these particles, and calculate the average value of A/B.
[[天然石墨类碳质物质(B)的原料]][[Raw material of natural graphite-like carbonaceous substance (B)]]
作为异取向性碳复合物中含有的天然石墨类碳质物质(B)的原料,通常可列举以采用X射线广角衍射法测定的(002)面的面间距(d002)显示0.340nm以下的结晶性高的天然石墨为原料的物质。具体地,优选选自如下材料的粉末:天然石墨或在其中加入机械粉碎物来提高圆形度而得到的产品、和/或将它们在100℃以上进行热处理而得到的产品、膨胀石墨的热处理产品、或者这些石墨的高纯度精制产品。As a raw material of the natural graphite-like carbonaceous substance (B) contained in the hetero-oriented carbon composite, generally, a crystal whose interplanar distance (d002) of the (002) plane measured by the X-ray wide-angle diffraction method is 0.340 nm or less is exemplified. It is made of high-strength natural graphite as a raw material. Specifically, powders selected from natural graphite or products obtained by adding mechanically pulverized materials to improve circularity, and/or products obtained by heat-treating them at 100° C. or higher, heat-treated expanded graphite products, or high-purity refined products of these graphites.
作为天然石墨类碳质物质(B)的前体的天然石墨(D),依照其性状,被分类为鳞片状石墨(Flake Graphite)、鳞状石墨(Crystalline(Vein)Graphite)、土壤石墨(Amorphous Graphite)(参见《粉粒体工艺技术集成》,((株)产业技术中心,昭和49年发行)的石墨一项;以及“HAND BOOK OF CARBON,GRAPHITE,DIAMOND AND FULLERENES”,(Noyes Publications发行))。石墨化度以鳞状石墨最高,为100%,接着是鳞片状石墨,为99.9%,而土壤石墨低至28%。作为天然石墨的鳞片状石墨产自马达加斯加、中国、巴西、乌克兰、加拿大等;鳞状石墨主要产自斯里兰卡。土壤石墨的主产地为朝鲜半岛、中国、墨西哥等。这些天然石墨中,土壤石墨不仅通常粒径较小,而且纯度低。相反,由于鳞片状石墨和鳞状石墨具有石墨化度或杂质量低等优点,因此可以在本发明中优选使用。Natural graphite (D), which is a precursor of natural graphite-like carbonaceous substances (B), is classified into flake graphite (Flake Graphite), scaly graphite (Crystalline (Vein) Graphite), soil graphite (Amorphous Graphite) (see "Powder Process Technology Integration", ((strain) Industrial Technology Center, issued in Showa 49) graphite item; and "HAND BOOK OF CARBON, GRAPHITE, DIAMOND AND FULLERENES", (issued by Noyes Publications) ). The degree of graphitization is highest in flake graphite, which is 100%, followed by flake graphite, which is 99.9%, while soil graphite is as low as 28%. Flake graphite as natural graphite is produced in Madagascar, China, Brazil, Ukraine, Canada, etc.; flake graphite is mainly produced in Sri Lanka. The main producing areas of soil graphite are the Korean Peninsula, China, Mexico, etc. Among these natural graphites, soil graphite not only usually has a smaller particle size, but also has low purity. In contrast, flaky graphite and flaky graphite can be preferably used in the present invention because they have advantages such as a low degree of graphitization or a low amount of impurities.
[[天然石墨类碳质物质(B)的制备]][[Preparation of natural graphitic carbonaceous material (B)]]
对于天然石墨类碳质物质(B)的制备,在后面的“异取向性碳复合物的制造方法1和制造方法2”中进行叙述。The preparation of the natural graphite-like carbonaceous material (B) will be described later in "Manufacturing Method 1 and Manufacturing Method 2 of Hetero-Oriented Carbon Composite".
[[天然石墨类碳质物质(B)的性质]][[Properties of Natural Graphite Carbonaceous Substance (B)]]
对于天然石墨类碳质物质(B),优选同时满足下面所示的(1)~(11)中任一项或多项。另外,各自的定义、测定方法等与异取向性碳复合物部分记载的定义、测定方法相同。For the natural graphite-like carbonaceous substance (B), it is preferable to simultaneously satisfy any one or more of (1) to (11) shown below. In addition, the definition, measurement method, etc. of each are the same as those described in the section of hetero-oriented carbon composite.
(1)X射线参数(1) X-ray parameters
天然石墨类碳质通过采用学振法的X射线衍射求得的晶格面(002)的d值(层间距离)优选为0.335nm以上,且通常为0.340nm以下,优选为0.337nm以下。如果低于该范围,则结晶性降低,有可能会增加初期不可逆容量。另外,0.335nm为石墨的理论值。另外,通过采用学振法的X射线衍射求得的碳材料的微晶大小(Lc)通常为30nm以上,优选为50nm以上,更优选为90nm以上的范围。如果低于该范围,则结晶性降低,有可能增加初期不可逆容量的增大。The d value (interlayer distance) of the lattice plane (002) obtained by X-ray diffraction using the Gakushin method of natural graphite-like carbonaceous is preferably 0.335 nm or more, and usually 0.340 nm or less, preferably 0.337 nm or less. If it is less than this range, the crystallinity may decrease, which may increase the initial irreversible capacity. In addition, 0.335 nm is a theoretical value of graphite. In addition, the crystallite size (Lc) of the carbon material obtained by X-ray diffraction using the Gakushin method is usually in the range of 30 nm or more, preferably 50 nm or more, more preferably 90 nm or more. If it is less than this range, the crystallinity may decrease, which may increase the increase in the initial irreversible capacity.
(2)灰分(2) Ash content
天然石墨类碳质中所含的灰分相对于天然石墨类碳质的总质量通常为1质量%以下,优选为0.5质量%以下,特别优选为0.1质量%以下,其下限通常为1ppm以上。如果超过上述范围,则充放电时与非水电解液的反应而引起的电池性能的劣化有时不能忽视。如果低于该范围,则在制造上需要较长时间和能量以及用于防止污染的设备,有时成本会上升。The ash contained in natural graphite-like carbonaceous is usually 1% by mass or less, preferably 0.5% by mass or less, particularly preferably 0.1% by mass or less, and the lower limit thereof is usually 1 ppm or more based on the total mass of natural graphite-like carbonaceous. When the above-mentioned range is exceeded, the deterioration of the battery performance due to the reaction with the non-aqueous electrolytic solution during charging and discharging may not be ignored. If it is less than this range, long time and energy are required for manufacturing and equipment for preventing pollution, and the cost may increase.
(3)体积基准平均粒径(3) Volume-based average particle size
天然石墨类碳质的体积基准平均粒径是通过激光衍射/散射法求得的体积基准的平均粒径(中值粒径),通常为1μm以上,优选为3μm以上,更优选为5μm以上,进一步优选为7μm以上。另外,其上限通常为50μm以下,优选为40μm以下,更优选为30μm以下,进一步优选为25μm以下。如果低于上述范围,则不可逆容量增大,有时导致初期电池容量的损失。另外,如果超过上述范围,则在制成电极极板时,容易形成不均匀的涂布面,有时在电池制作工序上是不优选的。The volume-based average particle size of natural graphite-like carbonaceous is the volume-based average particle size (median particle size) obtained by the laser diffraction/scattering method, and is usually 1 μm or more, preferably 3 μm or more, more preferably 5 μm or more, More preferably, it is 7 μm or more. In addition, the upper limit thereof is usually 50 μm or less, preferably 40 μm or less, more preferably 30 μm or less, and still more preferably 25 μm or less. If it is less than the above-mentioned range, the irreversible capacity may increase, resulting in loss of initial battery capacity. Moreover, if it exceeds the said range, it will become easy to form an uneven coating surface at the time of making an electrode pad, and it may be unpreferable in a battery manufacturing process.
(4)拉曼R值、拉曼半值宽度(4) Raman R value, Raman half value width
使用氩离子激光拉曼光谱法测定的天然石墨类碳质的拉曼R值通常为0.01以上,优选为0.02以上,更优选为0.04以上,其上限通常为0.35以下,优选为0.30以下,更优选为0.25以下的范围。如果拉曼R值低于该范围,则粒子表面的结晶性过高,随着充放电Li进入层间的位点有时变少。即,充电接受性有时降低。另一方面,如果超过该范围,则粒子表面的结晶性降低,与非水电解液的反应性增加,有时导致效率降低或产生的气体增加。The Raman R value of the natural graphitic carbonaceous measured by argon ion laser Raman spectroscopy is usually 0.01 or more, preferably 0.02 or more, more preferably 0.04 or more, and its upper limit is usually 0.35 or less, preferably 0.30 or less, more preferably It is in the range of 0.25 or less. If the Raman R value is lower than this range, the crystallinity of the particle surface may be too high, and there may be fewer sites for Li to enter the interlayer due to charge and discharge. That is, charge acceptance may be lowered. On the other hand, if it exceeds this range, the crystallinity of the particle surface decreases, the reactivity with the non-aqueous electrolyte solution increases, and the efficiency may decrease or the generated gas may increase.
另外,本发明的碳材料在1580cm-1附近的拉曼半值宽度没有特别的限制,通常为5cm-1以上,优选10cm-1以上,另外,其上限通常为40cm-1以下,优选为35cm-1以下,更优选为30cm-1以下的范围。如果拉曼半值宽度低于该范围,则粒子表面的结晶性过高,随着充放电Li进入层间的位点有时变少。即,充电接受性有时降低。另一方面,如果高于该范围,则粒子表面的结晶性降低,与非水电解液的反应性增加,有时导致效率降低或产生的气体增加。In addition, the Raman half-value width of the carbon material of the present invention is not particularly limited in the vicinity of 1580 cm -1 , but it is usually 5 cm -1 or more, preferably 10 cm -1 or more, and its upper limit is usually 40 cm -1 or less, preferably 35 cm-1 −1 or less, more preferably 30 cm −1 or less. If the Raman half-value width is below this range, the crystallinity of the particle surface may be too high, and there may be fewer sites for Li to enter the interlayer due to charge and discharge. That is, charge acceptance may be lowered. On the other hand, if it exceeds this range, the crystallinity of the particle surface decreases, the reactivity with the non-aqueous electrolyte solution increases, and the efficiency may decrease or the generated gas may increase.
(5)BET比表面积(5) BET specific surface area
使用BET法测定的天然石墨类碳质的比表面积通常为0.1m2/g以上,优选为0.7m2/g以上,更优选为1.0m2/g以上,进一步优选为1.5m2/g以上。其上限通常为100m2/g以下,优选为25m2/g以下,更优选为15m2/g以下,进一步优选为10m2/g以下。比表面积的值如果低于该范围,则作为负极材料使用时,在充电时锂的接受性变差,锂容易在电极表面析出。另一方面,如果高于上述范围,则用作负极材料时,与非水电解液的反应性增加,产生的气体容易变多,有时难以得到优选的电池。The specific surface area of natural graphite-like carbonaceous material measured by the BET method is usually 0.1 m 2 /g or more, preferably 0.7 m 2 /g or more, more preferably 1.0 m 2 /g or more, still more preferably 1.5 m 2 /g or more . The upper limit thereof is usually 100 m 2 /g or less, preferably 25 m 2 /g or less, more preferably 15 m 2 /g or less, still more preferably 10 m 2 /g or less. When the value of the specific surface area is lower than this range, when used as a negative electrode material, the acceptance of lithium during charging is deteriorated, and lithium is likely to be deposited on the surface of the electrode. On the other hand, if it is higher than the above range, when used as a negative electrode material, the reactivity with the non-aqueous electrolytic solution increases, the gas generated tends to increase, and it may be difficult to obtain a preferable battery.
(6)微孔分布(6) Micropore distribution
通过水银孔隙率计(水银压入法)求得的天然石墨类碳质的相当于直径0.01μm~1μm的粒子内的空隙、由于粒子表面的高低不平造成的凹凸的量通常为0.01mL/g以上,优选为0.05mL/g以上,更优选为0.1mL/g以上,其上限通常为0.6mL/g以下,优选为0.4mL/g以下,更优选为0.3mL/g以下的范围。如果超过该范围,则制造成极板时有时需要大量的粘合剂。如果低于该范围,则高电流密度充放电特性降低,并且,有时不能得到充放电时的电极膨胀收缩的缓和效果。The amount of voids in particles of natural graphite-like carbon equivalent to particles with a diameter of 0.01 μm to 1 μm, and the amount of unevenness due to particle surface roughness measured by mercury porosimetry (mercury intrusion method) is usually 0.01 mL/g Above, preferably 0.05 mL/g or more, more preferably 0.1 mL/g or more, the upper limit is usually 0.6 mL/g or less, preferably 0.4 mL/g or less, more preferably 0.3 mL/g or less. If it exceeds this range, a large amount of binder may be required when manufacturing an electrode plate. If it is less than this range, the high current density charge-discharge characteristics will deteriorate, and the effect of alleviating electrode expansion and contraction during charge-discharge may not be obtained.
另外,总微孔容积优选为0.1mL/g以上,更优选为0.25mL/g以上,其上限通常为10mL/g以下,优选为5mL/g以下,更优选为2mL/g以下的范围。如果超过该范围,则在制成基板时有时需要大量的粘合剂。如果低于该范围,则在制成极板时有时不能得到增稠剂或粘结剂的分散效果。In addition, the total pore volume is preferably 0.1 mL/g or more, more preferably 0.25 mL/g or more, and its upper limit is usually 10 mL/g or less, preferably 5 mL/g or less, more preferably 2 mL/g or less. If it exceeds this range, a large amount of adhesive may be required to form a substrate. If it is less than this range, the dispersion effect of the thickener or the binder may not be obtained when forming an electrode plate.
另外,平均微孔径优选为0.05μm以上,更优选为0.1μm以上,特别优选为0.5μm以上,其上限通常为50μm以下,优选为20μm以下,更优选为10μm以下的范围。如果超过该范围,则有时需要大量的粘合剂。如果低于该范围,则有时高电流密度充放电特性降低。In addition, the average pore diameter is preferably 0.05 μm or more, more preferably 0.1 μm or more, particularly preferably 0.5 μm or more, and the upper limit thereof is usually 50 μm or less, preferably 20 μm or less, more preferably 10 μm or less. If this range is exceeded, a large amount of adhesive is sometimes required. If it is less than this range, the high current density charge-discharge characteristics may deteriorate.
(7)圆形度(7) Circularity
使用圆形度作为天然石墨类碳材料的球形的程度,天然石墨类碳材料的粒径为3~40μm范围的粒子的圆形度通常为0.1以上,优选为0.5以上,更优选为0.8以上,进一步优选为0.85以上,最优选为0.9以上。圆形度大时,高电流密度充放电特性提高,因此优选。Using circularity as the spherical degree of the natural graphite-like carbon material, the circularity of the particles of the natural graphite-like carbon material with a particle diameter in the range of 3 to 40 μm is usually 0.1 or more, preferably 0.5 or more, more preferably 0.8 or more, More preferably, it is 0.85 or more, and most preferably, it is 0.9 or more. When the circularity is large, the high current density charge and discharge characteristics are improved, which is preferable.
(8)真密度(8) True density
天然石墨类碳质的真密度通常为2.0g/cm3以上,优选为2.1g/cm3以上,更优选为2.2g/cm3以上,进一步优选为2.22g/cm3以上,其上限为2.26g/cm3以下。上限为石墨的理论值。如果低于该范围,则碳的结晶性过低,初期不可逆容量有时会增大。The true density of natural graphite-like carbonaceous is usually above 2.0g/ cm3 , preferably above 2.1g/ cm3 , more preferably above 2.2g/ cm3 , even more preferably above 2.22g/ cm3 , and its upper limit is 2.26 g/ cm3 or less. The upper limit is the theoretical value of graphite. If it is less than this range, the crystallinity of carbon may be too low, and the initial irreversible capacity may increase.
(9)振实密度(9) Tap density
天然石墨类碳质的振实密度通常为0.1g/cm3以上,优选为0.5g/cm3以上,更优选为0.7g/cm3以上,特别优选为0.9g/cm3以上。另外,优选为2.0g/cm3以下,更优选为1.8g/cm3以下,特别优选为1.6g/cm3以下。振实密度如果低于该范围,则在作为负极使用时,填充密度很难提高,有时不能得到高容量的电池。另一方面,如果高于该范围,则电极中的粒子间的空隙过少,难以确保粒子间的导电性,有时难以得到优选的电池特性。The tap density of natural graphite-like carbonaceous is usually 0.1 g/cm 3 or more, preferably 0.5 g/cm 3 or more, more preferably 0.7 g/cm 3 or more, particularly preferably 0.9 g/cm 3 or more. In addition, it is preferably 2.0 g/cm 3 or less, more preferably 1.8 g/cm 3 or less, particularly preferably 1.6 g/cm 3 or less. If the tap density is lower than this range, it is difficult to increase the packing density when used as a negative electrode, and a high-capacity battery may not be obtained. On the other hand, if it exceeds this range, the gaps between the particles in the electrode will be too small, making it difficult to ensure the conductivity between the particles, and it may be difficult to obtain preferable battery characteristics.
(10)取向比(粉末)(10) Orientation ratio (powder)
天然石墨类碳质的取向比通常为0.005以上,优选为0.01以上,更优选为0.015以上,上限在理论上为0.67以下的范围。如果低于该范围,则有时高密度充放电特性降低。The orientation ratio of natural graphite-like carbonaceous is usually 0.005 or more, preferably 0.01 or more, more preferably 0.015 or more, and the upper limit is theoretically 0.67 or less. If it is less than this range, high-density charge-discharge characteristics may deteriorate.
(11)长径比(粉末)(11) aspect ratio (powder)
天然石墨类碳质的长径比理论上为1以上,上限通常为10以下,优选为8以下,更优选为5以下。如果超过其上限,制成极板时会产生条纹、无法得到均匀的涂布面,高电流密度充放电特性有时降低。The aspect ratio of natural graphite-like carbon is theoretically 1 or more, and the upper limit is usually 10 or less, preferably 8 or less, more preferably 5 or less. If the upper limit is exceeded, streaks may be generated when forming an electrode plate, a uniform coating surface may not be obtained, and the high current density charge and discharge characteristics may deteriorate.
[[碳质物质(C)的原料]][[Raw material of carbonaceous substance (C)]]
作为本发明的异取向性碳复合物中含有的碳质物质(C)的原料,只要是可以碳化的物质即可,没有特别限定,可以列举,沥青原料、苊烯、十环烯、蒽、菲等芳香族烃;吩嗪、吖啶等N环化合物;噻吩、联二噻吩等S环化合物;联苯、三联苯等聚苯;聚氯乙烯、聚乙烯醇、聚乙烯醇缩丁醛、它们的不溶解处理物、含氮的聚丙烯腈、聚吡咯等有机高分子;含硫的聚噻吩、聚苯乙烯等有机高分子;以纤维素、木质素、甘露聚糖、聚半乳糖醛酸、壳聚糖、蔗糖为代表的多糖类等天然高分子;聚苯硫醚、聚苯醚等热塑性树脂;糠醇树脂、酚醛树脂、酰亚胺树脂等热固性树脂;或者将它们溶解于苯、甲苯、二甲苯、喹啉、正己烷等低分子有机溶剂中而形成的溶液等有机物;可碳化的气体等。The raw material of the carbonaceous substance (C) contained in the heterooriented carbon composite of the present invention is not particularly limited as long as it can be carbonized, and examples thereof include pitch raw materials, acenaphthylene, decacyclene, anthracene, Aromatic hydrocarbons such as phenanthrene; N-ring compounds such as phenazine and acridine; S-ring compounds such as thiophene and dithiophene; polyphenylenes such as biphenyl and terphenyl; polyvinyl chloride, polyvinyl alcohol, polyvinyl butyral, Their insoluble processed products, nitrogen-containing polyacrylonitrile, polypyrrole and other organic polymers; sulfur-containing polythiophene, polystyrene and other organic polymers; cellulose, lignin, mannan, polygalacturaldehyde natural polymers such as polysaccharides represented by acid, chitosan, and sucrose; thermoplastic resins such as polyphenylene sulfide and polyphenylene ether; thermosetting resins such as furfuryl alcohol resins, phenolic resins, and imide resins; or dissolve them in benzene , toluene, xylene, quinoline, n-hexane and other low-molecular organic solvents and other organic substances; carbonizable gases, etc.
这些当中,由于沥青原料的残炭率高,可以制作收率高的材料,故优选。另外,本说明书中,“沥青原料”是沥青和属于沥青的物质,是指可以通过适当的处理进行碳化和/或石墨化的物质。作为具体的沥青原料的例子,可以使用焦油、重油或沥青等。作为焦油的具体例子,可列举煤焦油、石油类焦油等。作为重油的具体例子,可以列举石油类重油的接触分解油、热分解油、常压残油、减压残油等。另外,作为沥青的具体例子,可列举煤焦油沥青、石油类沥青、合成沥青等。其中,煤焦油沥青的芳香性高,故优选。这些沥青原料可以单独使用任意一种,也可以以任意组合和比例组合使用2种以上。Among these, the asphalt raw material is preferable because it can produce a high-yield material due to its high carbon residue rate. In addition, in this specification, a "pitch raw material" refers to pitch and a substance belonging to pitch, and refers to a substance that can be carbonized and/or graphitized by appropriate treatment. As an example of a specific pitch raw material, tar, heavy oil, pitch, etc. can be used. Specific examples of tar include coal tar, petroleum tar, and the like. Specific examples of the heavy oil include catalysis oil, pyrolysis oil, atmospheric residual oil, vacuum residual oil, and the like of petroleum heavy oil. In addition, specific examples of pitch include coal tar pitch, petroleum pitch, synthetic pitch, and the like. Among them, coal tar pitch is preferable because of its high aromaticity. Any one of these pitch raw materials may be used alone, or two or more of them may be used in combination in any desired ratio.
另外,上述喹啉不溶成分的沥青原料的含量没有特别限制,但通常使用处于30以下范围的沥青原料。所谓喹啉不溶成分,是煤焦油中微量含有的超微的碳粒子或极微小的油泥等,如果这些物质太多,则在石墨化过程中显著阻碍结晶性提高,从而导致石墨化之后的放电容量显著降低。另外,作为喹啉不溶成分的测定方法,例如可以使用JIS K2425中规定的方法。In addition, the content of the above-mentioned quinoline-insoluble pitch raw material is not particularly limited, but a pitch raw material in the range of 30 or less is usually used. The so-called quinoline insoluble components are ultrafine carbon particles or extremely fine sludge contained in a small amount of coal tar. If these substances are too large, the increase in crystallinity will be significantly hindered during the graphitization process, resulting in discharge after graphitization. The capacity is significantly reduced. In addition, as a method for measuring quinoline-insoluble components, for example, the method specified in JIS K2425 can be used.
另外,只要不妨碍本发明的效果,除了作为原料的上述沥青原料以外,还可以组合使用各种热固性树脂、热塑性树脂等。In addition, as long as the effect of the present invention is not hindered, various thermosetting resins, thermoplastic resins, and the like may be used in combination in addition to the above-mentioned pitch raw materials as raw materials.
[[碳质物质(C)的制备]][[Preparation of carbonaceous substance (C)]]
对于碳质物质(C)的制备,在后面的“异取向性碳复合物的制造方法1和制造方法2”中进行叙述。The preparation of the carbonaceous material (C) will be described later in "Manufacturing Method 1 and Manufacturing Method 2 of Hetero-Oriented Carbon Composite".
[[碳质物质(C)的性质]][[Properties of Carbonaceous Matter (C)]]
对于碳质物质(C),优选同时满足下面所示的(1)~(4)中的任意一项或多项。另外,它们的定义、测定方法等与异取向性碳复合物部分记载的定义、测定方法相同。The carbonaceous substance (C) preferably satisfies any one or more of the following (1) to (4) at the same time. In addition, their definitions, measurement methods, and the like are the same as those described in the section on heterooriented carbon composites.
(1)X射线参数(1) X-ray parameters
碳质物质(C)通过采用学振法的X射线衍射求得的晶格面(002)的d值(层间距离)优选为0.335nm以上,且通常为0.345nm以下,优选为0.340nm以下,更优选为0.337nm以下。另外,通过采用学振法的X射线衍射求得的碳材料的微晶大小(Lc)通常为5nm以上,优选为10nm以上,更优选为50nm以上,进一步优选为80nm以上的范围。如果低于该范围,则结晶性降低,有可能增加初期不可逆容量的增大。The d value (interlayer distance) of the lattice plane (002) of the carbonaceous substance (C) obtained by X-ray diffraction using the Gakushin method is preferably 0.335 nm or more, and usually 0.345 nm or less, preferably 0.340 nm or less , more preferably 0.337 nm or less. In addition, the crystallite size (Lc) of the carbon material obtained by X-ray diffraction using the Gakushin method is usually 5 nm or more, preferably 10 nm or more, more preferably 50 nm or more, and even more preferably 80 nm or more. If it is less than this range, the crystallinity may decrease, which may increase the increase in the initial irreversible capacity.
(2)灰分(2) Ash content
碳质物质(C)中所含的灰分相对于异取向性碳复合物的总质量通常为1质量%以下,优选为0.5质量%以下,特别优选为0.1质量%以下,其下限通常为1ppm以上。如果超过上述范围,则充放电时与非水电解液的反应而引起的电池性能的劣化有时不能忽视。如果低于该范围,则在制造上需要较长时间和能量以及用于防止污染的设备,有时成本会上升。The ash contained in the carbonaceous material (C) is usually 1% by mass or less, preferably 0.5% by mass or less, particularly preferably 0.1% by mass or less with respect to the total mass of the heterooriented carbon composite, and the lower limit thereof is usually 1 ppm or more . When the above-mentioned range is exceeded, the deterioration of the battery performance due to the reaction with the non-aqueous electrolytic solution during charging and discharging may not be ignored. If it is less than this range, long time and energy are required for manufacturing and equipment for preventing pollution, and the cost may increase.
(3)拉曼R值、拉曼半值宽度(3) Raman R value, Raman half value width
使用氩离子激光拉曼光谱法测定的碳质物质(C)的拉曼R值通常为0.01以上,优选为0.03以上,更优选为0.05以上,其上限通常为0.60以下,优选为0.30以下的范围。如果拉曼R值低于该范围,则粒子表面的结晶性过高,随着充放电Li进入层间的位点有时变少。即,充电接受性有时降低。另一方面,如果超过该范围,则粒子表面的结晶性降低,与非水电解液的反应性增加,有时导致效率降低或产生的气体增加。The Raman R value of the carbonaceous substance (C) measured by argon ion laser Raman spectroscopy is usually 0.01 or more, preferably 0.03 or more, more preferably 0.05 or more, and its upper limit is usually 0.60 or less, preferably 0.30 or less. . If the Raman R value is lower than this range, the crystallinity of the particle surface may be too high, and there may be fewer sites for Li to enter the interlayer due to charge and discharge. That is, charge acceptance may be lowered. On the other hand, if it exceeds this range, the crystallinity of the particle surface decreases, the reactivity with the non-aqueous electrolyte solution increases, and the efficiency may decrease or the generated gas may increase.
另外,本发明的碳质物质(C)在1580cm-1附近的拉曼半值宽度没有特别的限制,通常为5cm-1以上,优选10cm-1以上,另外,其上限通常为60cm-1以下,优选为45cm-1以下,更优选为30cm-1以下的范围。如果拉曼半值宽度低于该范围,则粒子表面的结晶性过高,随着充放电Li进入层间的位点有时变少。另一方面,如果高于该范围,则粒子表面的结晶性降低,与非水电解液的反应性增加,有时导致效率降低或产生的气体增加。In addition, the Raman half-value width of the carbonaceous material (C) of the present invention is not particularly limited in the vicinity of 1580 cm -1 , but is usually 5 cm -1 or more, preferably 10 cm -1 or more, and the upper limit is usually 60 cm -1 or less , preferably 45 cm -1 or less, more preferably 30 cm -1 or less. If the Raman half-value width is below this range, the crystallinity of the particle surface may be too high, and there may be fewer sites for Li to enter the interlayer due to charge and discharge. On the other hand, if it exceeds this range, the crystallinity of the particle surface decreases, the reactivity with the non-aqueous electrolyte solution increases, and the efficiency may decrease or the generated gas may increase.
(4)真密度(4) True density
碳质物质(C)的真密度通常为2.0g/cm3以上,优选为2.2g/cm3以上,更优选为2.22g/cm3以上,其上限为石墨的理论上限2.26g/cm3以下。如果低于该范围,则碳的结晶性过低,初期不可逆容量有时会增大。The true density of the carbonaceous substance (C) is usually 2.0 g/cm 3 or more, preferably 2.2 g/cm 3 or more, more preferably 2.22 g/cm 3 or more, and its upper limit is 2.26 g/cm 3 or less of the theoretical upper limit of graphite . If it is less than this range, the crystallinity of carbon may be too low, and the initial irreversible capacity may increase.
[负极[5]的异取向性碳复合物的制造方法][Manufacturing method of hetero-orientation carbon composite of negative electrode [5]]
作为制造方法,没有特别限定,只要不超出本发明的要旨的范围,就可以使用任何方法,优选的例子的概要如下面的(1)、(2)。The production method is not particularly limited, and any method can be used as long as it does not exceed the scope of the present invention, and the outlines of preferable examples are as follows (1) and (2).
(1)碳质物质(C)的初始原料在任何一个工序中其全部或一部分都形成液态的状态,并以该成为液态的状态与天然石墨(D)混合和/或进行捏合;(1) All or part of the initial raw material of the carbonaceous substance (C) is in a liquid state in any process, and mixed and/or kneaded with the natural graphite (D) in the liquid state;
(2)对该工序中得到的复合物进行脱挥发成分/烧结、石墨化,再进行粉碎、分级来调整粒度。在这些工序之间还可以经过一次以上的粉碎/分级工序。(2) Devolatilization, sintering, and graphitization are performed on the composite obtained in this step, followed by pulverization and classification to adjust the particle size. There may be one or more pulverization/classification steps between these steps.
下面示出上述概要的具体例。Specific examples of the above-mentioned outline are shown below.
[[异取向性碳复合物的制造方法1]][[Manufacturing Method 1 of Differently Oriented Carbon Composite]]
本发明中的异取向性碳复合物优选天然石墨类碳质物质(B)和取向性与天然石墨类碳质物质(B)不同的碳质物质(C)复合化,作为例子,可列举如下方法:在上述的天然石墨(D)中以规定的比例混合作为上述碳质物质(C)的前体的沥青原料、或对沥青原料等进行热处理并粉碎而得到的物质(下面,简称为“热处理石墨结晶前体”)、天然高分子等,进行热处理A后再进行粉碎,然后进行热处理B(烧结、石墨化)的方法等。另外,如果没有必要使中值粒径充分小,也可以不进行上述粉碎。The hetero-orientation carbon composite in the present invention is preferably composed of a natural graphite-like carbonaceous substance (B) and a carbonaceous substance (C) different in orientation from the natural graphite-like carbonaceous substance (B). As an example, the following Method: The above-mentioned natural graphite (D) is mixed with the above-mentioned pitch raw material which is a precursor of the above-mentioned carbonaceous substance (C), or a material obtained by heat-treating and pulverizing the pitch raw material (hereinafter, simply referred to as " Heat treatment of graphite crystal precursors"), natural polymers, etc., after heat treatment A, crushing, and then heat treatment B (sintering, graphitization) methods, etc. In addition, if it is not necessary to make the median particle diameter sufficiently small, the above pulverization may not be performed.
[[[热处理石墨结晶前体的制造]]][[[Manufacture of heat-treated graphite crystallization precursors]]]
事先对沥青原料实施热处理,得到热处理石墨结晶前体。将该事先的热处理称为沥青热处理。将该热处理石墨结晶前体粉碎后,与天然石墨(D)混合,然后进行热处理A,此时,其一部分或全部熔融,这里,可以通过事先的热处理(沥青热处理)来调整挥发成分的含量,由此来适当控制其熔融状态。另外,作为热处理石墨结晶前体中含有的挥发成分,通常可列举氢、苯、萘、蒽、芘等。Heat treatment is performed on the asphalt raw material in advance to obtain a heat-treated graphite crystallization precursor. This prior heat treatment is called asphalt heat treatment. After pulverizing the heat-treated graphite crystal precursor, mix it with natural graphite (D), and then perform heat treatment A. At this time, part or all of it melts. Here, the content of volatile components can be adjusted by prior heat treatment (pitch heat treatment), Thereby, its melting state is properly controlled. Moreover, hydrogen, benzene, naphthalene, anthracene, pyrene, etc. are usually mentioned as a volatile component contained in the heat-treated graphite crystal precursor.
沥青热处理时的温度条件没有特别限制,通常为300℃~550℃的范围。热处理的温度如果低于该范围,则挥发成分变多,因此有时难以在空气中安全地进行粉碎。另一方面,如果超过其上限,则在进行热处理A时,热处理石墨结晶前体的一部分或全部不会熔融,有时难以得到天然石墨类碳质物质(B)与热处理石墨结晶前体复合化的粒子(异取向性碳复合物)。进行沥青热处理时,在氮气等非活性气体氛围下或者由沥青原料产生的挥发成分氛围下进行。The temperature conditions during the asphalt heat treatment are not particularly limited, but are usually in the range of 300°C to 550°C. If the temperature of the heat treatment is lower than this range, volatile components will increase, so it may be difficult to pulverize safely in air. On the other hand, if the upper limit is exceeded, when heat treatment A is performed, part or all of the heat-treated graphite crystal precursor will not melt, and sometimes it is difficult to obtain the natural graphite-like carbonaceous material (B) and the heat-treated graphite crystal precursor. Particles (hetero-oriented carbon composites). When pitch heat treatment is performed, it is carried out in an atmosphere of an inert gas such as nitrogen or an atmosphere of volatile components generated from pitch raw materials.
作为沥青热处理时使用的装置,没有特别限制,例如可使用梭式炉、隧道式炉、电炉、高压釜等反应器、coka(コ一カ一)(cokes(コ一クス)制造的热处理槽)等。进行沥青热处理时,根据需要可以进行搅拌。There are no particular limitations on the equipment used for heat treatment of asphalt. For example, reactors such as shuttle furnaces, tunnel furnaces, electric furnaces, and autoclaves, and coka (cokes (cokes) heat treatment tanks) can be used. wait. During asphalt heat treatment, stirring can be performed as needed.
另外,作为热处理石墨结晶前体,优选使用挥发成分含有率通常为5质量%以上的物质。通过使用挥发成分含有率为该范围的石墨结晶前体,通过热处理A而使天然石墨类碳质物质(B)与碳质物质(C)复合化,从而可以得到具有上述规定物性的异取向性碳复合物。In addition, as the heat-treated graphite crystal precursor, it is preferable to use one having a volatile component content of usually 5% by mass or more. By using a graphite crystal precursor with a volatile component content within this range, the natural graphite-like carbonaceous substance (B) and the carbonaceous substance (C) are compounded by heat treatment A, thereby obtaining the hetero-orientation property having the above-mentioned predetermined physical properties. carbon composite.
首先,对于事先对沥青原料实施热处理,从而制造作为石墨结晶的前体的本体中间相(バルクメソフエ一ズ,bulk mesophase)(事先进行热处理的石墨结晶前体,下面简记为“热处理石墨结晶前体”)的方法进行说明。First of all, heat treatment is performed on the asphalt raw material in advance to produce a bulk mesophase (bulk mesofue one zu, bulk mesophase) as a precursor of graphite crystals (the graphite crystal precursors that have been heat-treated in advance are abbreviated as "heat-treated graphite crystal precursors" below. ”) method is described.
(热处理石墨结晶前体的挥发成分)(Volatile components of heat-treated graphite crystallization precursor)
通过沥青热处理而得到的石墨结晶前体的挥发成分没有特别限制,但通常为5质量%以上,优选为6质量%以上,另外,通常为20质量%以下,优选为15质量%以下。如果挥发成分低于上述范围,则由于挥发成分多,因此有时难以在空气中安全地进行粉碎,而如果超过其上限,则在进行热处理A时,石墨结晶前体的一部分或全部不会熔融,有时难以得到天然石墨类碳质物质(B)与热处理石墨结晶前体复合化的粒子(异取向性碳复合物)。另外,作为挥发成分的测定方法,例如使用JIS M8812中规定的方法。The volatile content of the graphite crystal precursor obtained by pitch heat treatment is not particularly limited, but is usually 5% by mass or more, preferably 6% by mass or more, and usually 20% by mass or less, preferably 15% by mass or less. If the volatile component is lower than the above-mentioned range, then because there are many volatile components, it is sometimes difficult to pulverize safely in the air, and if it exceeds the upper limit, then when heat treatment A is carried out, a part or all of the graphite crystal precursor will not be melted, It may be difficult to obtain particles (hetero-oriented carbon composites) in which natural graphite-like carbonaceous material (B) is composited with a heat-treated graphite crystal precursor. In addition, as a measurement method of volatile components, the method prescribed|regulated by JIS M8812 is used, for example.
(热处理石墨结晶前体的软化点)(Softening point of heat-treated graphite crystallization precursor)
通过沥青热处理而得到的石墨结晶前体的软化点没有特别限制,但通常为250℃以上,优选为300℃以上,更优选为370℃以上,另外,通常为470℃以下,优选为450℃以下,更优选为430℃以下的范围。如果低于下限,则热处理后的石墨结晶前体的碳化收率低,难以获得与天然石墨类碳质物质(B)的均匀的混合物,如果超过上限,则在进行热处理A时,石墨结晶前体的一部分或全部不会熔融,有时难以得到天然石墨类碳质物质(B)与热处理石墨结晶前体复合化的粒子(异取向性碳复合物)。The softening point of the graphite crystal precursor obtained by pitch heat treatment is not particularly limited, but is usually 250°C or higher, preferably 300°C or higher, more preferably 370°C or higher, and usually 470°C or lower, preferably 450°C or lower , more preferably in the range of 430°C or less. If it is lower than the lower limit, the carbonization yield of the graphite crystallization precursor after heat treatment is low, and it is difficult to obtain a uniform mixture with the natural graphite-like carbonaceous substance (B). Some or all of the body does not melt, and sometimes it is difficult to obtain particles (hetero-oriented carbon composites) in which the natural graphite-like carbonaceous material (B) and the heat-treated graphite crystal precursor are composited.
作为软化点,使用如下测定的值:使用热机械分析装置(例如,bruke-axs(ブルカ一·エイエツクス)株式会社制造的TMA4000),在氮流通下,升温速度10℃/分钟、针尖形状1mmφ、加重20gf的条件下,采用穿透法(ペネトレ一シヨン法)对用片剂成型器成型为1mm厚的试料进行测定。As the softening point, a value measured by using a thermomechanical analyzer (for example, TMA4000 manufactured by bruke-axs (Bruka-Eyex) Co., Ltd.) under nitrogen flow, a temperature increase rate of 10° C./min, a needle tip shape of 1 mmφ, Under the condition of increasing the weight of 20gf, the penetration method (Penetray-Sion method) was used to measure the sample molded to a thickness of 1mm by a tablet molding machine.
(热处理石墨结晶前体的粉碎)(Crushing of Heat-treated Graphite Crystal Precursor)
接着,将通过沥青热处理而得到的热处理石墨结晶前体进行粉碎。这是因为,通过热处理,将大单元且排列在同一方向上的热处理石墨结晶前体的结晶微细化,和/或均匀地进行天然石墨(D)和热处理石墨结晶前体的混合、复合化。Next, the heat-treated graphite crystal precursor obtained by heat-treating the pitch is pulverized. This is because, by heat treatment, the crystals of heat-treated graphite crystal precursors with large units arranged in the same direction are miniaturized, and/or the natural graphite (D) and heat-treated graphite crystal precursors are uniformly mixed and composited.
通过沥青热处理而得到的石墨结晶前体的粉碎没有特别限制,但粉碎后的热处理石墨结晶前体的粒度通常为1μm以上,优选为5μm以上,并且通常为10mm以下,优选为5mm以下,更优选为500μm以下,特别优选为200μm以下,进一步优选为50μm以下。上述粒度低于1μm时,在粉碎中或粉碎后由于热处理过的热处理石墨结晶前体的表面与空气接触而氧化,阻碍石墨化过程中的结晶性的提高,有时导致石墨化之后的放电容量的降低。另一方面,如果上述粒度超过10mm,则由于粉碎而带来的微细化效果减弱,结晶容易取向,碳质物质(C)容易取向,使用异取向性碳质复合物(A)的电极的活性物质取向比变低,难以抑制电极充电时的电极膨胀。和/或由于天然石墨(D)和热处理石墨结晶前体的粒径差变大,因此难以均匀混合,复合化有时容易变得不均匀。The pulverization of the graphite crystal precursor obtained by pitch heat treatment is not particularly limited, but the particle size of the heat-treated graphite crystal precursor after pulverization is usually 1 μm or more, preferably 5 μm or more, and usually 10 mm or less, preferably 5 mm or less, more preferably It is 500 μm or less, particularly preferably 200 μm or less, further preferably 50 μm or less. When the above-mentioned particle size is lower than 1 μm, the surface of the heat-treated graphite crystal precursor is oxidized due to contact with air during or after pulverization, which hinders the improvement of crystallinity in the graphitization process, and sometimes causes a decrease in the discharge capacity after graphitization. reduce. On the other hand, if the above-mentioned particle size exceeds 10 mm, the miniaturization effect due to pulverization will be weakened, the crystals will be easily oriented, and the carbonaceous substance (C) will be easily oriented, and the activity of the electrode using the different orientation carbonaceous composite (A) will be reduced. The material orientation ratio becomes low, and it becomes difficult to suppress electrode expansion during electrode charging. And/or since the difference in particle size between the natural graphite (D) and the heat-treated graphite crystal precursor becomes large, it is difficult to mix uniformly, and the compounding tends to become uneven in some cases.
用于粉碎的装置没有特别的限制,例如,作为粗粉碎机,可列举剪切式磨、颚式破碎机、冲击式压碎机、锥形压碎机等;作为中间粉碎机,可以举出辊式压碎机、锤式粉碎机等;作为微粉碎机,可以举出球磨、振动磨、销棒粉碎机、搅拌磨、喷射磨、涡轮磨(turbomill)等。The device used for pulverization is not particularly limited, for example, as a coarse pulverizer, shear mills, jaw crushers, impact crushers, cone crushers, etc. can be cited; as intermediate pulverizers, Roller crushers, hammer mills, and the like; examples of fine pulverizers include ball mills, vibration mills, pin mills, stirring mills, jet mills, and turbo mills.
[[[天然石墨(D)和热处理石墨结晶前体的热处理]]][[[Natural graphite (D) and thermal treatment of heat-treated graphite crystallization precursors]]]
将天然石墨(D)和热处理石墨结晶前体(碳质物质(C)的原料)以规定的比例混合,并进行热处理A、粉碎、热处理B(烧结、石墨化),由此制作异取向性碳复合物。Mix natural graphite (D) and heat-treated graphite crystal precursor (raw material of carbonaceous substance (C)) in a predetermined ratio, and perform heat treatment A, pulverization, and heat treatment B (sintering, graphitization), thereby producing hetero-orientation carbon composite.
(天然石墨(D)和热处理石墨结晶前体的混合)(mixture of natural graphite (D) and heat-treated graphite crystallization precursor)
在热处理A之前进行的天然石墨(D)和热处理石墨结晶前体的混合比例没有特别限制,天然石墨(D)相对于混合物的比例通常为20质量%以上,优选为30质量%以上,更优选为40质量%以上,另外,通常为95质量%以下,优选为90质量%以下。如果低于其下限,则由于异取向性碳复合物(A)中的碳质物质(C)的比例增加,在制成电极时难以提高填充密度,需要过大的压制荷重,有时不能获得将天然石墨类碳质物质(B)复合化的效果。如果超过其上限,则异取向性碳复合物(A)中的天然石墨类碳质物质(B)表面的露出增加,异取向性碳复合物(A)的比表面积有变大的可能性,在粉末物性上有时不优选。The mixing ratio of natural graphite (D) and the heat-treated graphite crystal precursor carried out before heat treatment A is not particularly limited, and the ratio of natural graphite (D) to the mixture is usually 20% by mass or more, preferably 30% by mass or more, more preferably 40% by mass or more, and usually 95% by mass or less, preferably 90% by mass or less. If it is lower than the lower limit, then due to the increase in the proportion of the carbonaceous substance (C) in the heterogeneous carbon composite (A), it is difficult to increase the packing density when the electrode is made, an excessive pressing load is required, and sometimes the Effect of compounding natural graphite-like carbonaceous material (B). If the upper limit is exceeded, the exposure of the surface of the natural graphite-like carbonaceous material (B) in the different-oriented carbon composite (A) increases, and the specific surface area of the different-oriented carbon composite (A) may become larger. It may not be preferable in terms of powder physical properties.
在将天然石墨(D)和调整为规定粒度的热处理石墨结晶前体进行混合时,对使用的装置没有特别限定,例如可列举V型混合机、W型混合机、容器可变型混合机、混炼机、滚筒混合器、剪切式混合器等。When mixing the natural graphite (D) and the heat-treated graphite crystal precursor adjusted to a predetermined particle size, the device used is not particularly limited, for example, a V-type mixer, a W-type mixer, a variable container type mixer, a mixer Mills, tumble mixers, shear mixers, etc.
(热处理A)(heat treatment A)
接着,对天然石墨(D)和热处理石墨结晶前体的混合物实施热处理A。这是因为,通过将粉碎后的热处理石墨结晶前体再熔融或熔合,天然石墨(D)和微细化后的热处理石墨结晶前体粒子以无取向状态接触而固定化。由此,天然石墨(D)和热处理石墨结晶前体的混合物不仅仅是粒子的混合物,也可以制成更均匀的复合化的混合物(下面适当称为“石墨复合混合物”)。Next, heat treatment A is performed on the mixture of natural graphite (D) and heat-treated graphite crystallization precursor. This is because the natural graphite (D) and the miniaturized heat-treated graphite crystal precursor particles are contacted and fixed in a non-oriented state by remelting or fusing the pulverized heat-treated graphite crystal precursor particles. Accordingly, the mixture of natural graphite (D) and heat-treated graphite crystal precursor can be not only a mixture of particles, but also a more uniform composite mixture (hereinafter appropriately referred to as "graphite composite mixture").
热处理A的温度条件没有特别限制,但通常为300℃以上,优选为400℃以上,更优选为450℃以上,另外,通常为650℃以下,优选为600℃以下。热处理A的温度如果低于上述范围,则在热处理A之后的材料中残存大量挥发成分,因此在烧结或石墨化工序时有可能引起粉末彼此之间的熔合,有时必须进行再粉碎。另一方面,如果超过上述范围,则再熔融的成分在粉碎时分离成针状,有时导致振实密度的降低。热处理A在氮气等非活性气体氛围下或者由粉碎而微细化的热处理石墨结晶前体产生的挥发成分氛围下进行。The temperature condition of the heat treatment A is not particularly limited, but is usually 300°C or higher, preferably 400°C or higher, more preferably 450°C or higher, and usually 650°C or lower, preferably 600°C or lower. If the temperature of heat treatment A is lower than the above range, a large amount of volatile components will remain in the material after heat treatment A, so the powders may be fused during the sintering or graphitization process, and regrinding may be necessary. On the other hand, if the above-mentioned range is exceeded, the remelted components will be separated into needles at the time of pulverization, which may lead to a decrease in the tap density. The heat treatment A is performed in an atmosphere of an inert gas such as nitrogen or an atmosphere of volatile components generated from the pulverized and miniaturized heat-treated graphite crystal precursor.
热处理A使用的装置没有特别限制,例如可使用梭式炉、隧道式炉、电炉等。The apparatus used for the heat treatment A is not particularly limited, and for example, a shuttle furnace, a tunnel furnace, an electric furnace, etc. can be used.
(热处理石墨结晶前体的粉碎和热处理A的代替处理)(Crushing of Heat-treated Graphite Crystal Precursor and Replacement Treatment of Heat-Treatment A)
可是,作为上述粉碎和热处理A的代替处理,可以是能够使热处理石墨结晶前体的组织微细化、无取向化的处理,例如,可以一边在热处理的石墨结晶前体熔融或软化的温度区域进行赋予机械能量的处理,一边与天然石墨(D)混合并进行热处理。However, as an alternative to the above pulverization and heat treatment A, it may be a treatment that can make the structure of the heat-treated graphite crystal precursor fine and non-oriented. For example, it may be performed in a temperature range where the heat-treated graphite crystal precursor melts or softens. In the process of imparting mechanical energy, it is mixed with natural graphite (D) and heat-treated.
作为该代替处理的热处理,没有特别限制,但通常在200℃以上,优选在250℃以上,且通常在450℃以下,优选在400℃以下进行。温度条件如果低于上述范围,则代替处理中的石墨结晶前体的熔融、软化不充分,有时难以进行与天然石墨(D)的复合化。另外,如果超过上述范围,则热处理容易急速进行,在粉碎时碳质热处理石墨结晶前体等的粒子分离成针状,有时容易导致振实密度的降低。The heat treatment as the replacement treatment is not particularly limited, but it is usually performed at 200°C or higher, preferably 250°C or higher, and usually 450°C or lower, preferably 400°C or lower. If the temperature condition is lower than the above-mentioned range, the melting and softening of the graphite crystal precursor during the substitution treatment may be insufficient, and it may be difficult to perform composite formation with natural graphite (D). In addition, if it exceeds the above range, the heat treatment is likely to proceed rapidly, and particles such as carbonaceous heat-treated graphite crystal precursors are separated into needles during pulverization, which may easily lead to a decrease in tap density.
该代替处理通常在氮气等非活性氛围下或者在空气等氧化性氛围下进行。但是,在氧化性氛围下进行处理时,在石墨化后有时难以得到高结晶性,因此有必要使氧导致的不熔化不要过度进行。具体地,使代替处理后的石墨结晶前体中的氧量通常为8质量%以下,优选为5质量%以下。This replacement treatment is usually performed in an inert atmosphere such as nitrogen or an oxidizing atmosphere such as air. However, when the treatment is performed in an oxidizing atmosphere, it may be difficult to obtain high crystallinity after graphitization, so it is necessary to prevent excessive infusion by oxygen. Specifically, the amount of oxygen in the graphite crystal precursor after the substitution treatment is usually 8% by mass or less, preferably 5% by mass or less.
另外,代替处理时使用的装置没有特别限制,例如可使用混合器、捏合机等。In addition, the device used for the replacement treatment is not particularly limited, and for example, a mixer, a kneader, or the like can be used.
(粉碎)(crushed)
接着,将进行了热处理A的石墨复合混合物进行粉碎。这是因为,通过热处理A而与天然石墨(D)复合化,以组织微细化、无取向化的状态熔融或熔合的石墨复合混合物的块通过粉碎而达到目标粒径。Next, the graphite composite mixture subjected to the heat treatment A is pulverized. This is because the lumps of the graphite composite mixture melted or fused in a finer structure and non-oriented state by heat treatment A to be composited with natural graphite (D) are pulverized to reach the target particle size.
粉碎后的石墨复合混合物的粒度没有特别限制,但通常为1μm以上,优选为3μm以上,更优选为5μm以上,进一步优选为7μm以上,另外,通常为50μm以下,优选为35以下,更优选为30μm以下。如果粒度低于上述范围,则作为异取向性碳质复合物(A)的振实密度变小,因此在制成电极时,难以提高活性物质的填充密度,难以获得高容量的电池。另一方面,如果超过上述范围,则在以异取向性碳质复合物(A)的形式进行涂布来制作电极时,有时容易产生涂布不均。The particle size of the pulverized graphite composite mixture is not particularly limited, but it is usually 1 μm or more, preferably 3 μm or more, more preferably 5 μm or more, further preferably 7 μm or more, and usually 50 μm or less, preferably 35 μm or less, more preferably Below 30μm. If the particle size is less than the above range, the tap density of the hetero-oriented carbonaceous composite (A) becomes small, so it is difficult to increase the filling density of the active material when it is made into an electrode, and it is difficult to obtain a high-capacity battery. On the other hand, if it exceeds the above range, when an electrode is produced by coating as the hetero-oriented carbonaceous composite (A), coating unevenness may easily occur.
对于用来粉碎的装置没有特别限制,例如,作为粗粉碎机,可列举颚式破碎机、冲击式压碎机、锥形压碎机等;作为中间粉碎机,可以举出辊式压碎机、锤式粉碎机等;作为微粉碎机,可以举出球磨、振动磨、销棒粉碎机、搅拌磨、喷射磨等。There is no particular limitation on the device used for pulverization. For example, as a coarse pulverizer, a jaw crusher, an impact crusher, a cone crusher, etc. can be mentioned; as an intermediate pulverizer, a roll crusher can be mentioned. , hammer mill, etc.; examples of fine pulverizers include ball mills, vibration mills, pin mills, stirring mills, jet mills, and the like.
(热处理B:烧结)(Heat treatment B: sintering)
热处理B是指烧结和石墨化。下面,从烧结开始说明。但是,也可以省略烧结。将通过粉碎而粉碎的石墨复合混合物进行烧结。这是因为,抑制石墨化时石墨复合混合物的熔合,并通过烧结而除去石墨复合混合物的挥发成分。Heat treatment B refers to sintering and graphitization. Next, the sintering will be described. However, sintering can also be omitted. The graphite composite mixture pulverized by pulverization is sintered. This is because fusion of the graphite composite mixture during graphitization is suppressed, and volatile components of the graphite composite mixture are removed by sintering.
进行烧结时的温度条件没有特别限制,但通常为600℃以上,优选为1000℃以上,并且上限通常为2400℃以下,优选为1300℃以下。温度条件如果低于上述范围,则在石墨化时有时容易引起石墨复合混合物粉末的熔合。另一方面,如果超过上述范围,则烧结设备需要费用,因此通常在上述温度条件的范围内进行。The temperature conditions for sintering are not particularly limited, but are usually 600°C or higher, preferably 1000°C or higher, and the upper limit is usually 2400°C or lower, preferably 1300°C or lower. If the temperature condition is lower than the above-mentioned range, fusion of graphite composite mixture powder may easily occur during graphitization. On the other hand, if the above-mentioned range is exceeded, the sintering equipment will require cost, so it is usually carried out within the range of the above-mentioned temperature conditions.
烧结在氮气等非活性氛围下或者在由再粉碎后的石墨复合混合物产生的气体形成的非氧化性氛围下进行。另外,为了简化制造工序,也可以不进行烧结工序而直接进行石墨化。The sintering is performed in an inert atmosphere such as nitrogen gas or a non-oxidizing atmosphere formed by gas generated from the re-pulverized graphite composite mixture. In addition, in order to simplify the production process, graphitization may be directly performed without performing the sintering process.
作为用于烧结的装置,没有特别限制,例如可使用梭式炉、隧道式炉、电炉、焦炭焙烧炉、回转炉等。The apparatus used for sintering is not particularly limited, and for example, a shuttle furnace, a tunnel furnace, an electric furnace, a coke roasting furnace, a rotary kiln, and the like can be used.
(热处理B:石墨化)(Heat treatment B: graphitization)
接着,对进行过烧结的石墨复合混合物实施石墨化。这是因为,增大电池评价中的放电容量,并提高结晶性。通过石墨化,可以得到异取向性碳质复合物(A)。Next, graphitization is performed on the sintered graphite composite mixture. This is because the discharge capacity in battery evaluation is increased and the crystallinity is improved. By graphitization, a hetero-oriented carbonaceous composite (A) can be obtained.
进行石墨化时的温度条件没有特别限制,但通常为2800℃以上,优选为2900℃以上,更优选为3000℃以上,并且通常为3400℃以下,优选为3200℃以下。如果超过上述范围,则有时电池的可逆容量变小,有时难以制作高容量的电池。另外,如果超过上述范围,则有时石墨的升华量容易变多。The temperature conditions for graphitization are not particularly limited, but are usually 2800°C or higher, preferably 2900°C or higher, more preferably 3000°C or higher, and usually 3400°C or lower, preferably 3200°C or lower. If the above-mentioned range is exceeded, the reversible capacity of the battery may decrease, and it may be difficult to manufacture a high-capacity battery. Moreover, when exceeding the said range, the amount of sublimation of graphite may become large easily.
石墨化在氩气等非活性氛围下或者在由烧结过的石墨复合混合物产生的气体形成的非氧化性氛围下进行。作为石墨化所使用的装置,没有特别限制,例如可列举直接通电炉、阿切孙电炉、作为间接通电式的电阻加热炉、感应加热炉等。Graphitization is carried out in an inert atmosphere such as argon gas or in a non-oxidizing atmosphere formed by the gas generated from the sintered graphite composite mixture. The apparatus used for graphitization is not particularly limited, and examples thereof include a direct electric furnace, an Acheson electric furnace, an indirect electric resistance heating furnace, an induction heating furnace, and the like.
另外,在石墨化处理时、或者其以前的工序,即从热处理到烧结的工序中,还可以在材料(天然石墨(D)、沥青原料或石墨结晶前体)中或表面添加Si、B等的石墨化催化剂。In addition, Si, B, etc. can also be added to the material (natural graphite (D), pitch raw material, or graphite crystal precursor) or on the surface during graphitization or in the previous process, that is, from heat treatment to sintering. graphitization catalyst.
(其他处理)(other processing)
另外,只要不妨碍本发明的效果,除了上述各处理以外,还可以进行分级处理等各种处理。分级处理是为了使石墨化处理后的粒度为目标粒径,并除去粗粉末和微粉末。In addition, as long as the effects of the present invention are not hindered, various processing such as classification processing may be performed in addition to the above-mentioned processing. The classification treatment is to make the particle size after the graphitization treatment the target particle size, and to remove coarse powder and fine powder.
作为用作分级处理的装置,没有特别限制,例如,在干式筛分的情况下,可以使用旋转式筛、摇动式筛、转动式筛、振动式筛等;在干式气流分级的情况下,可以使用重力式分级机、惯性力式分级机、离心力式分级机(分粒器、旋风分离器)等;湿式筛分可以使用机械式湿式分级机、水力分级机、沉降分级机、离心式湿式分级机等。As a device used for classification treatment, there is no particular limitation, for example, in the case of dry sieving, a rotary sieve, a shaking sieve, a rotary sieve, a vibrating sieve, etc. can be used; in the case of dry air classification , can use gravity classifier, inertial force classifier, centrifugal force classifier (classifier, cyclone separator), etc.; wet screening can use mechanical wet classifier, hydraulic classifier, sedimentation classifier, centrifugal wet classifier etc.
分级处理可以在热处理A后的粉碎之后立即继续进行,也可以在其他时间,例如粉碎后的烧结之后,或者石墨化之后进行。另外,还可以省略分级处理本身。但是,从降低异取向性碳质复合物(A)的BET比表面积这点,以及生产性这点来看,优选在热处理A后的粉碎之后立即继续进行。The classification treatment may be continued immediately after the pulverization after the heat treatment A, or may be performed at another time, such as after sintering after pulverization, or after graphitization. In addition, the classification process itself can also be omitted. However, from the viewpoint of reducing the BET specific surface area of the heterooriented carbonaceous composite (A) and the viewpoint of productivity, it is preferable to continue immediately after the pulverization after the heat treatment A.
(异取向性碳质复合物(A)的制造后的处理)(Processing after production of the hetero-oriented carbonaceous composite (A))
为了控制负极材料的BET比表面积、提高电极压制性、提高放电容量、以及廉价化等,还可以对以上述顺序制造的异取向性碳质复合物(A)添加另行制造的人造石墨粉末或天然石墨粉末并进行混合。In order to control the BET specific surface area of the negative electrode material, improve the electrode compactness, increase the discharge capacity, and reduce costs, etc., it is also possible to add artificial graphite powder or natural stone to the heterooriented carbonaceous composite (A) produced in the above order. Toner powder and mix.
[负极[5]的异取向性碳复合物的制造方法2][Manufacturing Method 2 of Different Orientation Carbon Composite of Negative Electrode [5]]
异取向性碳复合物可以按照下面的方法制造。本发明中的异取向性碳复合物优选天然石墨类碳质物质(B)和取向性与天然石墨类碳质物质(B)不同的碳质物质(C)进行复合化,作为例子,可列举如下方法:在上述天然石墨(D)中,将作为上述碳质物质(C)的前体的沥青原料经过“捏合(混合)”、“成型”、“烧结”、“石墨化”、“粉碎”的工序来制造等。但是,这些工序中,“成型”、“烧结”、“粉碎”可以省略和/或与其他工序同时实施。具体地,可以通过以下列举的制造方法等获得。A heterooriented carbon composite can be produced as follows. The hetero-orientation carbon composite in the present invention is preferably composed of a natural graphite-like carbonaceous substance (B) and a carbonaceous substance (C) different in orientation from the natural graphite-like carbonaceous substance (B). As an example, The following method: in the above-mentioned natural graphite (D), the pitch raw material as the precursor of the above-mentioned carbonaceous substance (C) is subjected to "kneading (mixing)", "forming", "sintering", "graphitization", "crushing "The process to manufacture and so on. However, among these steps, "molding", "sintering" and "crushing" may be omitted and/or performed simultaneously with other steps. Specifically, it can be obtained by the production methods listed below and the like.
[[捏合(混合)]][[kneading (mixing)]]
进行天然石墨(D)、沥青原料和根据希望添加的石墨化催化剂等原料的混合。此时,为了均匀混合,优选进行加热。由此,成为在天然石墨(D)和捏合温度下不熔融的原料中添加液态的沥青原料的状态。此时,可以在捏合机中加入所有原料同时进行捏合和升温,也可以在捏合机中装入沥青原料以外的成分并在搅拌状态下加热,温度上升到捏合温度后,再加入常温或硫化熔融状态的沥青原料。Mixing of raw materials such as natural graphite (D), a pitch raw material, and a graphitization catalyst added as desired is performed. At this time, it is preferable to heat for uniform mixing. Thereby, a liquid pitch raw material is added to the natural graphite (D) and the raw material that does not melt at the kneading temperature. At this time, you can add all the raw materials into the kneader and knead and heat up at the same time, or you can put ingredients other than asphalt raw materials into the kneader and heat them under stirring. state of asphalt raw material.
加热温度通常在沥青原料的软化点以上,优选在比软化点高10℃以上的温度,更优选在比软化点高20℃以上的温度范围进行。另外,上限通常为300℃以下,优选为250℃以下的范围。如果低于该范围,则沥青原料的粘度变高,混合有可能变得困难。另一方面,如果超过该范围,则由于挥发和缩聚,混合体系的粘度有时变得过高。The heating temperature is usually higher than the softening point of the pitch raw material, preferably at a temperature higher than the softening point by 10°C or higher, more preferably in a temperature range higher than the softening point by 20°C or higher. In addition, the upper limit is usually not more than 300°C, preferably not more than 250°C. If it is less than this range, the viscosity of the asphalt raw material will become high, and mixing may become difficult. On the other hand, if it exceeds this range, the viscosity of the mixed system may become too high due to volatilization and polycondensation.
混合机优选具有搅拌翼的机种,搅拌翼可以使用Z型、マチスケ一タ型这样的通用的搅拌翼。投入到混合机中的原料的量通常为混合机容积的10体积%以上,优选为15体积%以上,且为50体积%以下,优选为30体积%以下。混合时间必须为5分钟以上,最长为直到由于挥发成分的挥发而带来明显的粘度的变化的时间,通常为30~120分钟。混合机优选在混合之前预热到捏合温度。The mixer is preferably a model having stirring blades, and general-purpose stirring blades such as Z type and Machisket type can be used for the stirring blades. The amount of the raw material charged into the mixer is usually 10% by volume or more, preferably 15% by volume or more, and 50% by volume or less, preferably 30% by volume or less, based on the volume of the mixer. The mixing time must be 5 minutes or more, and the longest time until a significant change in viscosity due to volatilization of volatile components is brought about is usually 30 to 120 minutes. The mixer is preferably preheated to kneading temperature prior to mixing.
[[成型]][[forming]]
得到的混合物可以直接提供给用于除去挥发成分和碳化的脱挥发成分/烧结工序,但为了容易操作,优选在成型之后再提供给脱挥发成分/烧结工序。The obtained mixture may be directly supplied to a devolatilization/sintering process for removing volatile components and carbonization, but for ease of handling, it is preferably supplied to the devolatilization/sintering process after molding.
只要可以保持形状,则成型方法没有特别限制,可以采用挤出成型、模具成型、静水压成型等。其中,优选挤出成型或模具成型,因为挤出成型在成型体内粒子容易取向,而模具成型虽然粒子取向保持无规,但从生产性上来看,比静水压成型容易操作,另外,在混合时不会使无规取向的结构破坏而得到成型体。The molding method is not particularly limited as long as the shape can be maintained, and extrusion molding, die molding, hydrostatic molding, and the like can be used. Among them, extrusion molding or mold molding is preferred, because extrusion molding is easy to orient particles in the molding body, and mold molding is easier to operate than hydrostatic molding from the viewpoint of productivity, although the particle orientation remains random. A molded body can be obtained without destroying the structure of random orientation.
成型温度可以是室温(低温)、加热下(高温、沥青原料的软化点以上的温度)的任一种。在低温下成型时,为了提高成型性并获得成型体的均一性,希望将捏合后冷却的混合物预先粗粉碎至最大尺寸1mm以下。成型体的形状、大小没有特别限制,但在热成型时,如果成型体过大,则在成型之前进行均匀的预热需要时间,因此,通常优选最大尺寸为150cm左右以下的大小。The forming temperature may be any of room temperature (low temperature) and heating (high temperature, temperature above the softening point of the pitch raw material). When molding at low temperature, in order to improve the moldability and obtain the uniformity of the molded product, it is desirable to coarsely pulverize the mixture cooled after kneading to a maximum size of 1 mm or less. The shape and size of the molded body are not particularly limited, but in thermoforming, if the molded body is too large, it will take time to uniformly preheat before molding, so it is generally preferable to have a maximum size of about 150 cm or less.
成型压力的压力如果过高,则通过成型体的微孔的挥发成分的除去变得困难,并且不是真圆的天然石墨(D)进行取向,后续工序中的粉碎变得困难,因此成型压力的上限通常为3000kgf/cm2(294MPa)以下,优选为500kgf/cm2(49MPa)以下,更优选为10kgf/cm2(0.98MPa)以下。下限的压力没有特别限制,但优选设定为在脱挥发成分/烧结工序中可以保持成型体的形状的程度。If the pressure of the molding pressure is too high, it will be difficult to remove the volatile components passing through the micropores of the molded body, and the natural graphite (D) that is not perfectly round will be oriented, and the pulverization in the subsequent process will become difficult, so the molding pressure The upper limit is usually 3000 kgf/cm 2 (294 MPa) or less, preferably 500 kgf/cm 2 (49 MPa) or less, more preferably 10 kgf/cm 2 (0.98 MPa) or less. The lower limit pressure is not particularly limited, but is preferably set to such an extent that the shape of the compact can be maintained in the devolatilization/sintering process.
[[脱挥发成分/烧结]][[Devolatilization/Sintering]]
为了除去天然石墨(D)和沥青原料的挥发成分,并防止石墨化时的填充物的污染、填充物对成型体的粘合,对得到的成型体进行脱挥发成分/烧结。脱挥发成分/烧结通常在600℃以上、优选在650℃以上,且通常在1300℃以下、优选在1100℃以下的温度进行0.1小时~10小时。为了防止氧化,加热通常在氮、氩等非活性气体的流通下或者在间隙中填充有焦炭渣(breeze)、填充焦炭(Packing coke)等粒状碳材料的非氧化性氛围中进行。The obtained molded body was devolatilized/sintered in order to remove the volatile components of the natural graphite (D) and pitch raw materials, and to prevent contamination of the filler during graphitization and adhesion of the filler to the molded body. Devolatilization/sintering is performed at a temperature of usually 600°C or higher, preferably 650°C or higher, and usually 1300°C or lower, preferably 1100°C or lower, for 0.1 to 10 hours. In order to prevent oxidation, heating is usually carried out under the circulation of inert gas such as nitrogen and argon, or in a non-oxidizing atmosphere filled with granular carbon materials such as coke slag (breeze) and packed coke (Packing coke) in the gap.
脱挥发成分/烧结中使用的设备只要是电炉或煤气炉、电极材料用焦炭焙烧炉等可以在非氧化性氛围中烧结的设备即可,没有特别限定。为了除去挥发成分,加热时的升温速度优选为低速,通常以3~100℃/小时从低沸点成分开始挥发的200℃左右升温到只产生氢的700℃附近。The equipment used for devolatilization and sintering is not particularly limited as long as it can be sintered in a non-oxidizing atmosphere, such as an electric furnace, a gas furnace, and a coke roasting furnace for electrode materials. In order to remove volatile components, the temperature increase rate during heating is preferably low, usually at 3 to 100°C/hour from about 200°C where low boiling point components start to volatilize to around 700°C where only hydrogen is generated.
[[石墨化]][[Graphitization]]
通过脱挥发成分/烧结而得到的碳化物成型体接着在高温下加热进行石墨化。对于石墨化的条件,与制造方法1记载的相同。The carbide shaped body obtained by devolatilization/sintering is then graphitized by heating at high temperature. The conditions for graphitization are the same as those described in Production Method 1.
为了防止氧化,石墨化在氮、氩等非活性气体的流通下或者在间隙中填充有焦炭渣、填充焦炭等粒状碳材料的非氧化性氛围下进行。石墨化中使用的设备只要是电炉或煤气炉、电极材料用阿切孙电炉等可实现上述目的的设备即可,没有特别限定,升温速度、冷却速度、热处理时间等可以在使用的设备所允许的范围内任意设定。In order to prevent oxidation, graphitization is performed under the flow of an inert gas such as nitrogen or argon, or under a non-oxidizing atmosphere in which a granular carbon material such as coke slag or filled coke is filled in the gap. The equipment used in graphitization is not particularly limited as long as it is an electric furnace, a gas furnace, an Acheson electric furnace for electrode materials, etc. that can achieve the above purpose, and the heating rate, cooling rate, heat treatment time, etc. can be used within the limits of the equipment used. can be set arbitrarily within the range.
[[粉碎]][[Crush]]
这样得到的石墨化处理物通常是块状,难以作为负极活性物质使用,因此进行粉碎和/或大粒径状物质、小粒径物质的除去。石墨化处理物的粉碎方法没有特别限制,作为粉碎装置,机械磨碎的装置例如可列举球磨、锤式粉碎机、CF磨、细磨机、粉碎机(pulverize)、利用风力的粉碎装置例如喷射磨等。对于粗粉碎、中间粉碎,可以使用颚式破碎机、锤击式粉碎机、辊碾机等利用冲击力的粉碎方式。这里,粉碎时间可以是石墨化之前,也可以是石墨化之后。The graphitized product obtained in this way is usually in the form of a block and is difficult to use as a negative electrode active material. Therefore, pulverization and/or removal of large-diameter and small-diameter substances is performed. The pulverization method of the graphitized product is not particularly limited, and as the pulverization device, the device of mechanical pulverization includes, for example, a ball mill, a hammer mill, a CF mill, a fine mill, a pulverizer, and a pulverization device utilizing wind power, such as a jet Grinding etc. For coarse crushing and intermediate crushing, crushing methods using impact force such as jaw crushers, hammer mills, and roller mills can be used. Here, the pulverization time may be before graphitization or after graphitization.
[负极[5]的副材料混合][Mix of sub-materials of negative electrode [5]]
除上述异取向性碳复合物以外,通过在本发明的锂二次电池的负极活性物质中含有一种以上与上述异取向性碳复合物在碳质的物性方面不同的碳质物质(碳质材料),可以进一步谋求电池性能的提高。这里所说的“碳质的物性”,是指X射线衍射参数、中值粒径、长径比、BET比表面积、取向比、拉曼R值、振实密度、真密度、微孔分布、圆形度、灰分量的一种以上的特性。另外,作为优选的实施方式,可以举出,体积基准粒度分布以中值粒径为中心时左右不对称、含有2种以上拉曼R值不同的碳材料、或X射线参数不同等。作为其效果的一个例子,可列举通过含有天然石墨、人造石墨等石墨;乙炔黑等炭黑;针状焦等无定形碳等碳材料作为副材料来降低电阻等。它们可以单独使用一种,也可以以任意组合和任意比例组合使用2种以上。作为副材料添加时,其添加量通常为0.1质量%以上,优选为0.5质量%以上,更优选为0.6质量%以上,其上限通常为80质量%以下,优选为50质量%以下,更优选为40质量%以下,进一步优选为30质量%以下的范围。如果低于该范围,则有时难以得到导电性提高的效果。如果超过上述范围,则有时会导致初期不可逆容量的增大。In addition to the above-mentioned different orientation carbon composites, by containing one or more carbonaceous substances (carbonaceous materials) different from the above-mentioned different orientation carbon composites in the carbonaceous physical properties in the negative electrode active material of the lithium secondary battery of the present invention material), which can further improve the performance of the battery. The "carbonaceous physical properties" mentioned here refer to X-ray diffraction parameters, median particle size, aspect ratio, BET specific surface area, orientation ratio, Raman R value, tap density, true density, micropore distribution, More than one characteristic of circularity and gray content. In addition, as a preferred embodiment, the volume-based particle size distribution is asymmetrical around the median particle size, contains two or more carbon materials with different Raman R values, or has different X-ray parameters. As an example of the effect, reduction of electrical resistance by including carbon materials such as graphite such as natural graphite and artificial graphite; carbon black such as acetylene black; and amorphous carbon such as needle coke as sub-materials can be cited. These may be used individually by 1 type, and may be used in combination of 2 or more types by arbitrary and arbitrary ratios. When it is added as an auxiliary material, its addition amount is usually 0.1 mass % or more, preferably 0.5 mass % or more, more preferably 0.6 mass % or more, and its upper limit is usually 80 mass % or less, preferably 50 mass % or less, more preferably 40% by mass or less, more preferably in the range of 30% by mass or less. If it is less than this range, it may be difficult to obtain the effect of improving electrical conductivity. If the above range is exceeded, the initial irreversible capacity may increase.
[制作负极[5]的电极][Making the electrode of the negative electrode [5]]
制造负极可以采用通常的方法,可以与上述同样地形成负极[5]。集电体、集电体和活性物质层的厚度之比、电极密度、粘合剂、极板取向比、阻抗等也与上述相同。A common method can be used to manufacture the negative electrode, and the negative electrode can be formed in the same manner as above [5]. The current collector, the thickness ratio between the current collector and the active material layer, electrode density, binder, plate orientation ratio, impedance, and the like are also the same as those described above.
<负极[6]><Negative pole [6]>
下面,对本发明的锂二次电池中使用的负极[6]进行说明,该负极含有石墨质碳粒子作为负极活性物质,所述石墨质碳粒子的圆形度为0.85以上,通过广角X射线衍射法测定的(002)面的面间距(d002)低于0.337nm,采用氩离子激光拉曼光谱法测定的以1360cm-1的峰强度相对于1580cm-1的峰强度之比定义的拉曼R值为0.12~0.8。Next, the negative electrode [6] used in the lithium secondary battery of the present invention will be described. The negative electrode contains graphitic carbon particles as the negative electrode active material, and the circularity of the graphitic carbon particles is 0.85 or more. The interplanar distance (d002) of the (002) plane measured by the method is lower than 0.337nm, and the Raman R defined by the ratio of the peak intensity of 1360cm -1 to the peak intensity of 1580cm -1 measured by argon ion laser Raman spectroscopy The value is 0.12 to 0.8.
[负极[6]的负极活性物质][Negative electrode active material of negative electrode [6]]
下面,对负极[6]中使用的负极活性物质进行说明。Next, the negative electrode active material used in the negative electrode [6] will be described.
本发明的锂二次电池的负极[6]中使用的负极活性物质至少含有满足下面的(a)、(b)和(c)的石墨质碳粒子。The negative electrode active material used in the negative electrode [6] of the lithium secondary battery of the present invention contains at least graphitic carbon particles satisfying the following (a), (b) and (c).
(a)圆形度为0.85以上;(a) The circularity is above 0.85;
(b)广角X射线衍射法测定的(002)面的面间距(d002)低于0.337nm;(b) The interplanar distance (d002) of the (002) plane measured by wide-angle X-ray diffraction method is less than 0.337nm;
(c)采用氩离子激光拉曼光谱法测定的以1360cm-1的峰强度相对于1580cm-1的峰强度之比定义的拉曼R值(以下,有时只简记为“拉曼R值”)为0.12~0.8。(c) The Raman R value defined by the ratio of the peak intensity at 1360 cm -1 to the peak intensity at 1580 cm -1 measured by argon ion laser Raman spectroscopy (hereinafter, sometimes simply referred to as "Raman R value" ) is 0.12 to 0.8.
[[圆形度]][[Circularity]]
作为本发明的锂二次电池的负极活性物质使用的石墨质碳粒子的圆形度通常为0.85以上,优选为0.87以上,更优选为0.89以上,特别优选为0.92以上。作为上限,圆形度为1时,为理论真球。如果圆形度低于该范围,则负极活性物质的填充性降低,热传导性降低,因此有时防碍早期的输出恢复,特别是,由低温时的低输出状态的输出的恢复有时变慢。The circularity of the graphitic carbon particles used as the negative electrode active material of the lithium secondary battery of the present invention is usually 0.85 or more, preferably 0.87 or more, more preferably 0.89 or more, particularly preferably 0.92 or more. As an upper limit, when the circularity is 1, it is a theoretical true sphere. If the circularity is lower than this range, the filling property of the negative electrode active material is lowered, and the thermal conductivity is lowered. Therefore, early output recovery may be hindered. In particular, output recovery from a low output state at low temperature may be slow.
本发明中所说的圆形度用下式定义。The circularity referred to in the present invention is defined by the following formula.
圆形度=(具有与粒子投影形状相同面积的等效圆的周长)/(粒子投影形状的实际周长)Circularity = (perimeter of an equivalent circle having the same area as the particle projected shape)/(actual perimeter of the particle projected shape)
作为圆形度的值使用如下测定的值:使用流动式粒子图像分析装置(例如,Sysmex Industrial公司制造的FPIA),将约0.2g试样分散于作为表面活性剂的聚氧乙烯(20)山梨糖醇酐单月桂酸酯的0.2质量%水溶液(约50mL)中,以60W的输出功率照射1分钟28kHz的超声波后,指定0.6~400μm为检测范围,对粒径3~40μm范围的粒子进行测定。As the value of circularity, the value measured as follows is used: using a flow type particle image analyzer (for example, FPIA manufactured by Sysmex Industrial Co., Ltd.), about 0.2 g of a sample is dispersed in polyoxyethylene (20) sorbitol as a surfactant. After irradiating a 0.2% by mass aqueous solution (approximately 50mL) of sugar-alcohol monolaurate with an output of 60W for 1 minute at 28kHz ultrasonic waves, specify a detection range of 0.6-400μm, and measure particles with a particle diameter of 3-40μm .
[[面间距(d002)]][[Space between faces (d002)]]
作为本发明的锂二次电池中负极[6]的负极活性物质使用的石墨质碳粒子采用广角X射线衍射法测定的(002)面的面间距(d002)低于0.337nm,优选为0.336nm以下。作为下限,为石墨的理论值0.335。如果超过该范围,则结晶性降低,由电子引起的热传导性降低,早期的输出恢复特性有时降低,特别是,由低温时的低输出状态的输出的恢复有时变慢。The graphitic carbon particles used as the negative electrode active material of the negative electrode [6] in the lithium secondary battery of the present invention adopt the interplanar distance (d002) of the (002) plane measured by wide-angle X-ray diffraction method to be lower than 0.337nm, preferably 0.336nm the following. As the lower limit, the theoretical value of graphite is 0.335. If it exceeds this range, the crystallinity decreases, the thermal conductivity due to electrons decreases, and the early output recovery characteristics may decrease. In particular, the output recovery from the low output state at low temperature may be slow.
本发明中所说的采用广角X射线衍射法测定的(002)面的面间距(d002),是通过采用学振法的X射线衍射求得的晶格面(002面)的d值(层间距离)。The interplanar distance (d002) of the (002) plane measured by the wide-angle X-ray diffraction method mentioned in the present invention is the d value (layer distance).
另外,通过采用学振法的X射线衍射求得的石墨质碳材料的微晶大小(Lc)没有特别限定,但通常为10nm以上,优选为30nm以上,更优选为80nm以上的范围。如果低于该范围,则结晶性降低,由电子引起的热传导性降低,早期的输出恢复特性有时降低。In addition, the crystallite size (Lc) of the graphitic carbon material obtained by X-ray diffraction using the Gakushin method is not particularly limited, but is usually 10 nm or more, preferably 30 nm or more, more preferably 80 nm or more. If it is less than this range, the crystallinity is lowered, the thermal conductivity caused by electrons is lowered, and the early output recovery characteristics may be lowered.
[[拉曼R值]][[Raman R value]]
作为本发明的锂二次电池中负极[6]的负极活性物质使用的石墨质碳粒子的拉曼R值必须为0.12以上,优选为0.15以上,更优选为0.17以上,特别优选为0.2以上。其上限优选为0.8以下,更优选为0.6以下,特别优选为0.45以下。如果拉曼R值低于该范围,则粒子表面的结晶性过高,随着充放电位点的减少,输出有时降低。另一方面,如果超过该范围,则粒子表面的结晶性降低,由电子引起的热传导降低,输出的恢复特性有时降低。The Raman R value of the graphitic carbon particles used as the negative electrode active material of the negative electrode [6] in the lithium secondary battery of the present invention must be 0.12 or more, preferably 0.15 or more, more preferably 0.17 or more, particularly preferably 0.2 or more. The upper limit thereof is preferably 0.8 or less, more preferably 0.6 or less, particularly preferably 0.45 or less. If the Raman R value is lower than this range, the crystallinity of the particle surface is too high, and the output may decrease with the decrease of charge and discharge sites. On the other hand, if it exceeds this range, the crystallinity of the particle surface decreases, the heat conduction by electrons decreases, and the output recovery characteristics may decrease.
拉曼光谱的测定如下进行:使用拉曼分光器(例如日本分光社制造的拉曼分光器),使试样自然落下并填充在测定池内,对池内的试样表面照射氩离子激光,同时,使池在与激光垂直的面内旋转。对于得到的拉曼光谱,测定1580cm-1附近的峰PA的强度IA和1360cm-1附近的峰PB的强度IB,计算其强度比R(R=IB/IA),将其定义为石墨质碳材料的拉曼R值。另外,测定得到的拉曼光谱在1580cm-1附近的峰PA的半值宽度,将其定义为石墨质碳材料的拉曼半值宽度。The measurement of the Raman spectrum is carried out as follows: Using a Raman spectrometer (such as a Raman spectrometer manufactured by JASCO Corporation), the sample is naturally dropped and filled in the measurement cell, and the surface of the sample in the cell is irradiated with an argon ion laser. At the same time, Rotate the cell in a plane perpendicular to the laser. For the obtained Raman spectrum, the intensity I A of the peak PA near 1580 cm -1 and the intensity I B of the peak P B near 1360 cm -1 were measured , and the intensity ratio R (R= IB / IA ) was calculated. It is defined as the Raman R value of the graphitic carbon material. In addition, the half-value width of the peak PA in the vicinity of 1580 cm −1 in the obtained Raman spectrum was measured, and this was defined as the Raman half-value width of the graphitic carbon material.
另外,这里的拉曼测定条件如下。In addition, the Raman measurement conditions here are as follows.
·氩离子激光波长:514.5nmArgon ion laser wavelength: 514.5nm
·试样上的激光功率:15~25mW·Laser power on the sample: 15~25mW
·分辨率:10~20cm-1 ·Resolution: 10~20cm -1
·测定范围:1100cm-1~1730cm-1 · Measuring range: 1100cm -1 ~ 1730cm -1
·拉曼R值、半值宽度分析:本底处理Raman R value, half-value width analysis: background processing
·光滑处理:单纯平均、卷积5点·Smooth processing: simple average, convolution 5 points
作为本发明的锂二次电池中负极[6]的负极活性物质使用的石墨质碳粒子在1580cm-1附近的拉曼半值宽度没有特别的限制,但通常为10cm-1以上,优选为15cm-1以上,另外,其上限通常为60cm-1以下,优选为50cm-1以下,更优选为45cm-1以下的范围。如果拉曼半值宽度低于该范围,则粒子表面的结晶性过高,随着充放电位点的减少,输出有时降低。另一方面,如果超过该范围,则粒子表面的结晶性降低,由电子引起的热传导降低,输出的恢复特性有时降低。The Raman half-value width near 1580 cm of the graphitic carbon particles used as the negative electrode active material of the negative electrode [6] in the lithium secondary battery of the present invention is not particularly limited, but it is usually 10 cm or more, preferably 15 cm -1 or more, and the upper limit is usually 60 cm -1 or less, preferably 50 cm -1 or less, more preferably 45 cm -1 or less. If the Raman half-value width is below this range, the crystallinity of the particle surface is too high, and the output may decrease with the decrease of charge and discharge sites. On the other hand, if it exceeds this range, the crystallinity of the particle surface decreases, the heat conduction by electrons decreases, and the output recovery characteristics may decrease.
[[振实密度]][[Tap Density]]
作为本发明的锂二次电池中负极[6]的负极活性物质使用的石墨质碳粒子的振实密度通常为0.55g/cm3以上,优选为0.7g/cm3以上,更优选为0.8g/cm3以上,特别优选为1g/cm3以上。另外,上限优选为2g/cm3以下,更优选为1.8g/cm3以下,特别优选为1.6g/cm3以下。振实密度如果低于该范围,则在作为负极使用时,填充密度很难提高,粒子间的接触面积减少,因此热传导性有时降低。另一方面,如果超过该范围,则电极中的粒子间的空隙过少,非水电解液的流路减少,因此输出本身有时减少。The tap density of the graphitic carbon particles used as the negative electrode active material of the negative electrode [6] in the lithium secondary battery of the present invention is usually 0.55 g/cm 3 or more, preferably 0.7 g/cm 3 or more, more preferably 0.8 g /cm 3 or more, particularly preferably 1 g/cm 3 or more. In addition, the upper limit is preferably 2 g/cm 3 or less, more preferably 1.8 g/cm 3 or less, particularly preferably 1.6 g/cm 3 or less. When the tap density is lower than this range, when used as a negative electrode, it is difficult to increase the packing density, and the contact area between particles decreases, so thermal conductivity may decrease. On the other hand, if it exceeds this range, the gap between the particles in the electrode will be too small, and the flow path of the non-aqueous electrolyte solution will decrease, so the output itself may decrease.
在本发明中,振实密度如下定义:使试样通过孔径300μm的筛子落在20cm3的振实容器内,直到试样充满容器的上端面后,使用粉末密度测定器(例如,seishin企业公司制造的Tap densor)进行1000次冲程长度10mm的振动,将由此时的体积和试样的重量求得的密度定义为振实密度。In the present invention, the tap density is defined as follows: the sample is passed through a sieve with an aperture of 300 μm and dropped in a 20 cm container for vibration until the sample is filled with the upper end surface of the container, and a powder density measuring device (for example, Seishin Enterprise Co., Ltd. The manufactured Tap densor) was vibrated 1000 times with a stroke length of 10 mm, and the density obtained from the volume at that time and the weight of the sample was defined as the tap density.
[[BET比表面积]][[BET specific surface area]]
使用BET法测定的作为本发明的锂二次电池中负极[6]的负极活性物质使用的石墨质碳粒子的比表面积优选为0.1m2/g以上,更优选为0.7m2/g以上,特别优选为1m2/g以上,进一步优选为1.5m2/g以上。其上限优选为100m2/g以下,更优选为50m2/g以下,特别优选为25m2/g以下,进一步优选为15m2/g以下。BET比表面积的值如果低于该范围,则作为负极材料使用时,在充电时锂的接受性容易变差,锂有时容易在电极表面析出。另一方面,如果高于上述范围,则用作负极材料时,与非水电解液的反应性增加,产生的气体容易变多,有时难以得到优选的电池。The specific surface area of the graphitic carbon particles used as the negative electrode active material of the negative electrode [6] in the lithium secondary battery of the present invention measured by the BET method is preferably 0.1 m 2 /g or more, more preferably 0.7 m 2 /g or more, It is particularly preferably 1 m 2 /g or more, further preferably 1.5 m 2 /g or more. The upper limit thereof is preferably 100 m 2 /g or less, more preferably 50 m 2 /g or less, particularly preferably 25 m 2 /g or less, further preferably 15 m 2 /g or less. If the value of the BET specific surface area is less than this range, when used as a negative electrode material, the acceptance of lithium at the time of charge tends to deteriorate, and lithium tends to precipitate on the electrode surface in some cases. On the other hand, if it is higher than the above range, when used as a negative electrode material, the reactivity with the non-aqueous electrolytic solution increases, the gas generated tends to increase, and it may be difficult to obtain a preferable battery.
BET比表面积定义为如下测定的值:使用表面积计(例如,大仓理研制造的全自动表面积测定装置),在氮气流通下,在350℃下对试样进行15分钟预干燥,然后,使用氮气相对于大气压的相对压力值准确调整为0.3的氮氦混合气体,通过采用气体流动法的氮吸附BET 1点法测定。The BET specific surface area is defined as a value measured by pre-drying the sample at 350° C. for 15 minutes under nitrogen flow using a surface area meter (for example, a fully automatic surface area measuring device manufactured by Okura Riken), and then, using nitrogen gas Nitrogen-helium mixed gas whose relative pressure value relative to the atmospheric pressure is accurately adjusted to 0.3 is measured by the nitrogen adsorption BET 1-point method using the gas flow method.
[[体积平均粒径]][[Volume average particle size]]
作为本发明的锂二次电池中负极[6]的负极活性物质使用的石墨质碳粒子的体积平均粒径定义为通过激光衍射/散射法求得的体积基准的平均粒径(中值粒径),优选为1μm以上,更优选为3μm以上,特别优选为5μm以上,进一步优选为7μm以上。另外,其上限通常为50μm以下,优选为40μm以下,更优选为30μm以下,,特别优选为25μm以下。如果低于上述范围,则不可逆容量增大,有时导致初期电池容量的损失。另外,如果超过上述范围,则在制成电极极板时,容易形成不均匀的涂布面,有时在电池制作工序上是不优选的。The volume average particle diameter of the graphitic carbon particles used as the negative electrode active material of the negative electrode [6] in the lithium secondary battery of the present invention is defined as the average particle diameter (median particle diameter) of the volume basis obtained by the laser diffraction/scattering method ), preferably 1 μm or more, more preferably 3 μm or more, particularly preferably 5 μm or more, further preferably 7 μm or more. In addition, the upper limit thereof is usually 50 μm or less, preferably 40 μm or less, more preferably 30 μm or less, and particularly preferably 25 μm or less. If it is less than the above-mentioned range, the irreversible capacity may increase, resulting in loss of initial battery capacity. Moreover, if it exceeds the said range, it will become easy to form an uneven coating surface at the time of making an electrode pad, and it may be unpreferable in a battery manufacturing process.
[[微孔容积]][[pore volume]]
对于本发明的锂二次电池中负极[6]的负极活性物质使用的石墨质碳粒子的微孔容积,通过水银孔隙率计(水银压入法)求得的相当于直径0.01μm~1μm的粒子内的空隙、由于粒子表面的高低不平造成的凹凸的量通常为0.01mL/g以上,优选为0.05mL/g以上,更优选为0.1mL/g以上,其上限通常为0.6mL/g以下,优选为0.4mL/g以下,更优选为0.3mL/g以下的范围。如果超过该范围,则制造成极板时有时需要大量的粘合剂。如果低于该范围,则高电流密度充放电特性降低,并且,有时不能得到充放电时的电极膨胀收缩的缓和效果。For the micropore volume of the graphitic carbon particles used in the negative electrode active material of the negative electrode [6] in the lithium secondary battery of the present invention, the volume equivalent to 0.01 μm to 1 μm in diameter obtained by mercury porosimetry (mercury porosimetry) The amount of voids in the particles and the unevenness of the particle surface is usually 0.01 mL/g or more, preferably 0.05 mL/g or more, more preferably 0.1 mL/g or more, and the upper limit is usually 0.6 mL/g or less , preferably 0.4 mL/g or less, more preferably 0.3 mL/g or less. If it exceeds this range, a large amount of binder may be required when manufacturing an electrode plate. If it is less than this range, the high current density charge-discharge characteristics will deteriorate, and the effect of alleviating electrode expansion and contraction during charge-discharge may not be obtained.
另外,总微孔容积优选为0.1mL/g以上,更优选为0.25mL/g以上,其上限通常为10mL/g以下,优选为5mL/g以下,更优选为2mL/g以下的范围。如果超过该范围,则在制成基板时有时需要大量的粘合剂。如果低于该范围,则在制成极板时有时不能得到增稠剂或粘结剂的分散效果。In addition, the total pore volume is preferably 0.1 mL/g or more, more preferably 0.25 mL/g or more, and its upper limit is usually 10 mL/g or less, preferably 5 mL/g or less, more preferably 2 mL/g or less. If it exceeds this range, a large amount of adhesive may be required to form a substrate. If it is less than this range, the dispersion effect of the thickener or the binder may not be obtained when forming an electrode plate.
另外,平均微孔径优选为0.05μm以上,更优选为0.1μm以上,进一步优选为0.5μm以上,其上限通常为50μm以下,优选为20μm以下,更优选为10μm以下的范围。如果超过该范围,则有时需要大量的粘合剂。如果低于该范围,则有时高电流密度充放电特性降低。In addition, the average pore diameter is preferably 0.05 μm or more, more preferably 0.1 μm or more, even more preferably 0.5 μm or more, and the upper limit thereof is usually 50 μm or less, preferably 20 μm or less, more preferably 10 μm or less. If this range is exceeded, a large amount of adhesive is sometimes required. If it is less than this range, the high current density charge-discharge characteristics may deteriorate.
作为用于水银孔隙率计的装置,可以使用水银孔隙率计(autopore9520;micrometritics公司制造)。称量约0.2g试样(负极材料),密封入粉末用容器中,在室温、真空下(50μmHg以下)进行10分钟脱气,实施前处理。接着,减压至4psia(约28kPa),导入水银,从4psia(约28kPa)阶段状升压至40000psia(约280MPa),然后降压至25psia(约170kPa)。升压时的阶段数为80阶段以上,在各阶段中,在10秒的平衡时间后,测定水银压入量。使用Washburn式从这样得到的水银压入曲线计算出微孔分布。另外,水银的表面张力(γ)为485dyne/cm,接触角(φ)为140°。平均微孔径使用累积微孔体积达到50%时的微孔径。As an apparatus used for the mercury porosimeter, a mercury porosimeter (autopore9520; manufactured by Micrometrics Inc.) can be used. About 0.2 g of the sample (negative electrode material) was weighed, sealed in a powder container, and degassed at room temperature under vacuum (50 μmHg or less) for 10 minutes to perform pretreatment. Next, the pressure is reduced to 4psia (about 28kPa), mercury is introduced, the pressure is raised from 4psia (about 28kPa) to 40000psia (about 280MPa) in steps, and then the pressure is lowered to 25psia (about 170kPa). The number of stages during the pressurization was 80 or more, and the amount of mercury intrusion was measured after an equilibration time of 10 seconds in each stage. The micropore distribution was calculated from the mercury intrusion curve thus obtained using the Washburn formula. In addition, the surface tension (γ) of mercury is 485 dyne/cm, and the contact angle (φ) is 140°. As the average pore diameter, the pore diameter at which the cumulative pore volume reaches 50% is used.
[[灰分]][[ash]]
作为本发明的锂二次电池中负极[6]的负极活性物质使用的石墨质碳粒子的灰分相对于石墨质碳粒子的总质量优选为1质量%以下,特别优选为0.5质量%以下,更优选为0.1质量%以下。另外,其下限优选为1ppm以上。如果超过上述范围,则充放电时与非水电解液的反应而引起的电池性能的劣化有时不能忽视。另一方面,如果低于该范围,则在制造上需要较长时间和能量以及用于防止污染的设备,有时成本会上升。The ash content of the graphitic carbon particles used as the negative electrode active material of the negative electrode [6] in the lithium secondary battery of the present invention is preferably 1% by mass or less, particularly preferably 0.5% by mass or less, with respect to the total mass of the graphitic carbon particles. Preferably it is 0.1 mass % or less. In addition, the lower limit thereof is preferably 1 ppm or more. When the above-mentioned range is exceeded, the deterioration of the battery performance due to the reaction with the non-aqueous electrolytic solution during charging and discharging may not be ignored. On the other hand, if it is less than this range, long time and energy are required for manufacturing and equipment for preventing pollution, and the cost may increase.
石墨质碳粒子的真密度通常为2.0g/cm3以上,优选为2.1g/cm3以上,更优选为2.2g/cm3以上,进一步优选为2.22g/cm3以上,其上限为2.26g/cm3以下。上限为石墨的理论值。如果低于该范围,则碳的结晶性过低,初期不可逆容量有时会增大。在本发明中,真密度定义为通过使用丁醇的液相置换法(比重计法)测定的值。The true density of the graphitic carbon particles is usually 2.0 g/cm 3 or more, preferably 2.1 g/cm 3 or more, more preferably 2.2 g/cm 3 or more, still more preferably 2.22 g/cm 3 or more, and the upper limit is 2.26 g /cm 3 or less. The upper limit is the theoretical value of graphite. If it is less than this range, the crystallinity of carbon may be too low, and the initial irreversible capacity may increase. In the present invention, the true density is defined as a value measured by a liquid phase displacement method (pycnometer method) using butanol.
[[取向比]][[Orientation Ratio]]
作为本发明的锂二次电池中负极[6]的负极活性物质使用的石墨质碳粒子的取向比通常为0.005以上,优选为0.01以上,更优选为0.015以上,上限在理论上为0.67以下的范围。如果低于该范围,则有时高密度充放电特性降低。The orientation ratio of the graphitic carbon particles used as the negative electrode active material of the negative electrode [6] in the lithium secondary battery of the present invention is usually 0.005 or more, preferably 0.01 or more, more preferably 0.015 or more, and the upper limit is theoretically 0.67 or less. scope. If it is less than this range, high-density charge-discharge characteristics may deteriorate.
取向比通过X射线衍射测定。使用非对称皮尔森VII作为分布函数,拟合通过X射线衍射得到的碳的(110)衍射和(004)衍射的峰,进行峰分离,分别计算出(110)衍射和(004)衍射的峰的积分强度。由得到的积分强度计算出由(110)衍射积分强度/(004)衍射积分强度表示的比值,将该比值定义为活性物质取向比。The orientation ratio was measured by X-ray diffraction. Using asymmetric Pearson VII as a distribution function, fit the peaks of (110) diffraction and (004) diffraction of carbon obtained by X-ray diffraction, perform peak separation, and calculate the peaks of (110) diffraction and (004) diffraction respectively integral strength. A ratio represented by (110) diffraction integrated intensity/(004) diffraction integrated intensity was calculated from the obtained integrated intensity, and this ratio was defined as the active material orientation ratio.
这里的X射线衍射测定条件如下。另外,“2θ”表示衍射角。The X-ray diffraction measurement conditions here are as follows. In addition, "2θ" represents a diffraction angle.
·靶:Cu(Kα射线)石墨单色光度仪Target: Cu (Kα ray) graphite monochromator
·狭缝:发散狭缝=1度,受光狭缝=0.1mm,散射狭缝=1度· Slit: divergence slit = 1 degree, light receiving slit = 0.1mm, scattering slit = 1 degree
·测定范围和步进角/测定时间・Measurement range and step angle/measurement time
(110)面:76.5度≤2θ≤78.5度0.01度/3秒(110) surface: 76.5 degrees ≤ 2θ ≤ 78.5 degrees 0.01 degrees / 3 seconds
(004)面:53.5度≤2θ≤56.0度0.01度/3秒(004) plane: 53.5 degrees ≤ 2θ ≤ 56.0 degrees 0.01 degrees / 3 seconds
[[长径比]][[Aspect Ratio]]
作为本发明的锂二次电池中负极[6]的负极活性物质使用的石墨质碳粒子的长径比理论上为1以上,上限为10以下,优选为8以下,更优选为5以下。如果超过其上限,制成极板时会产生条纹、无法得到均匀的涂布面,高电流密度充放电特性有时降低。The aspect ratio of the graphitic carbon particles used as the negative electrode active material of the negative electrode [6] in the lithium secondary battery of the present invention is theoretically 1 or more, the upper limit is 10 or less, preferably 8 or less, more preferably 5 or less. If the upper limit is exceeded, streaks may be generated when forming an electrode plate, a uniform coating surface may not be obtained, and the high current density charge and discharge characteristics may deteriorate.
另外,长径比用三维观察时粒子的最长径A和与其垂直的最短径B的比值A/B来表示。粒子的观察通过可以放大观察的扫描型电子显微镜来进行。选择固定在厚度50微米以下的金属的端面上的任意50个石墨粒子,旋转、倾斜固定有试样的载物台,分别测定这些粒子的A、B,求出A/B的平均值。In addition, the aspect ratio is represented by the ratio A/B of the longest diameter A of a particle and the shortest diameter B perpendicular thereto in three-dimensional observation. Observation of particles is performed with a scanning electron microscope capable of magnified observation. Select any 50 graphite particles fixed on the end face of the metal with a thickness of 50 microns or less, rotate and tilt the stage on which the sample is fixed, measure A and B of these particles, and calculate the average value of A/B.
作为本发明的锂二次电池中负极[6]的负极活性物质使用的石墨质碳粒子可以是天然出产的物质,也可以是人工制造的物质,但优选石墨质碳粒子含有天然石墨。另外,天然出产的物质或人工制造的物质也可以施加特定的处理。另外,制造方法(也包含分选方法)没有特别限制,例如,可以使用筛分或风力分级等分级方法分选具有上述特性的石墨质碳粒子而取得。The graphitic carbon particles used as the negative electrode active material of the negative electrode [6] in the lithium secondary battery of the present invention may be naturally produced or artificially produced, but preferably the graphitic carbon particles contain natural graphite. In addition, naturally occurring substances or artificially produced substances may also be subject to specific treatments. In addition, the production method (including the sorting method) is not particularly limited, and for example, graphitic carbon particles having the above-mentioned characteristics can be obtained by sorting using a classification method such as sieving or wind classification.
这些当中,特别优选的石墨质碳粒子是对天然出产的碳质粒子或人工制造的碳质粒子施加力学能量处理进行改性而制造的。更优选作为力学能量处理的原料的碳质粒子含有天然石墨。Among these, particularly preferable graphitic carbon particles are produced by modifying naturally occurring carbonaceous particles or artificially produced carbonaceous particles by subjecting them to mechanical energy treatment. More preferably, the carbonaceous particles used as a raw material for mechanical energy treatment contain natural graphite.
[[力学能量处理]][[Mechanical Energy Processing]]
下面,对该力学能量处理进行说明。作为施加力学能量处理的对象的原料的碳质粒子,没有特别限定,是天然或人造的石墨类碳质粒子、作为石墨前体的碳质粒子等。这些原料的特性如下所示。Next, this mechanical energy processing will be described. The carbonaceous particles as the raw material to be subjected to the mechanical energy treatment are not particularly limited, and include natural or artificial graphite-based carbonaceous particles, carbonaceous particles that are precursors of graphite, and the like. The characteristics of these raw materials are shown below.
[[[作为力学能量处理的原料石墨类碳质粒子]]][[[Graphite-like carbonaceous particles as raw material for mechanical energy processing]]]
关于原料石墨类碳质粒子的性质,优选同时满足如下所示的(1)~(11)的任一项或多项。另外,物性测定方法或定义,均与上述石墨质碳粒子的情况相同。Regarding the properties of the raw material graphite-based carbonaceous particles, it is preferable to satisfy any one or more of the following (1) to (11) at the same time. In addition, the measurement methods and definitions of the physical properties are the same as in the case of the above-mentioned graphitic carbon particles.
(1)X射线参数(1) X-ray parameters
原料石墨类碳质粒子通过采用学振法的X射线衍射求得的晶格面(002)的d值(层间距离)优选为0.335nm以上,其上限通常为0.340nm以下,优选为0.337nm以下。另外,通过采用学振法的X射线衍射求得的石墨类碳质粒子的微晶大小(Lc)通常为30nm以上,优选为50nm以上,更优选为100nm以上的范围。如果低于该范围,则结晶性降低,有可能增加初期不可逆容量的增大。The d value (interlayer distance) of the lattice plane (002) obtained by the X-ray diffraction using the Gakushin method of the raw material graphite-based carbonaceous particles is preferably 0.335 nm or more, and the upper limit is usually 0.340 nm or less, preferably 0.337 nm the following. In addition, the crystallite size (Lc) of the graphitic carbonaceous particles obtained by X-ray diffraction using the Gakushin method is usually 30 nm or more, preferably 50 nm or more, and more preferably 100 nm or more. If it is less than this range, the crystallinity may decrease, which may increase the increase in the initial irreversible capacity.
(2)灰分(2) Ash content
原料石墨类碳质粒子中所含的灰分相对于石墨类碳质的总质量为1质量%以下,优选为0.5质量%以下,特别优选为0.1质量%以下,其下限通常为1ppm以上。如果超过上述范围,则充放电时与非水电解液的反应而引起的电池性能的劣化有时不能忽视。如果低于该范围,则在制造上需要较长时间和能量以及用于防止污染的设备,有时成本会上升。The ash contained in the raw graphite-like carbonaceous particles is 1% by mass or less, preferably 0.5% by mass or less, particularly preferably 0.1% by mass or less, and the lower limit is usually 1 ppm or more based on the total mass of the graphite-like carbonaceous particles. When the above-mentioned range is exceeded, the deterioration of the battery performance due to the reaction with the non-aqueous electrolytic solution during charging and discharging may not be ignored. If it is less than this range, long time and energy are required for manufacturing and equipment for preventing pollution, and the cost may increase.
(3)体积基准平均粒径(3) Volume-based average particle size
原料石墨类碳质粒子的体积基准平均粒径定义为通过激光衍射/散射法求得的体积基准的平均粒径(中值粒径),通常为1μm以上,优选为3μm以上,更优选为5μm以上,进一步优选为7μm以上。另外,其上限没有特别限制,通常为10mm以下,优选为1mm以下,更优选为500μm以下,进一步优选为100μm以下,特别优选为50μm以下。如果低于上述范围,则通过施加力学能量处理而使粒径变得过小,有时导致不可逆容量增大。另外,如果超过上述范围,则施加力学能量处理的装置难以有效运转,有时导致时间上的损失。The volume-based average particle size of the raw graphite-based carbonaceous particles is defined as the volume-based average particle size (median particle size) obtained by the laser diffraction/scattering method, and is usually 1 μm or more, preferably 3 μm or more, more preferably 5 μm or more, and more preferably 7 μm or more. The upper limit is not particularly limited, but is usually 10 mm or less, preferably 1 mm or less, more preferably 500 μm or less, further preferably 100 μm or less, particularly preferably 50 μm or less. If it is less than the above range, the particle diameter becomes too small by application of mechanical energy treatment, which may result in an increase in irreversible capacity. In addition, if it exceeds the above-mentioned range, it will be difficult for the device for applying mechanical energy treatment to operate effectively, which may result in a loss of time.
(4)拉曼R值、拉曼半值宽度(4) Raman R value, Raman half value width
使用氩离子激光拉曼光谱法测定的原料石墨类碳质粒子的拉曼R值通常为0.01以上,优选为0.03以上,更优选为0.1以上,其上限通常为0.6以下,优选为0.4以下。如果拉曼R值低于该范围,则粒子表面的结晶性过高,由于施加力学能量处理而引起的拉曼值的增加,以及由于结晶性的降低,随着充放电Li进入层间的位点有时变少,即,充电接受性有时降低。另一方面,如果超过该范围,则由于施加力学能量处理而使粒子表面的结晶性进一步降低,与非水电解液的反应性增加,有时导致效率降低或产生的气体增加。The Raman R value of the raw graphitic carbonaceous particles measured by argon ion laser Raman spectroscopy is usually 0.01 or more, preferably 0.03 or more, more preferably 0.1 or more, and its upper limit is usually 0.6 or less, preferably 0.4 or less. If the Raman R value is lower than this range, the crystallinity of the particle surface is too high, the Raman value increases due to the application of mechanical energy treatment, and due to the decrease in crystallinity, Li enters the interlayer sites with charge and discharge. The number of dots sometimes decreases, that is, the charge acceptance sometimes decreases. On the other hand, if it exceeds this range, the crystallinity of the particle surface will further decrease due to the mechanical energy treatment, and the reactivity with the non-aqueous electrolyte solution will increase, resulting in a reduction in efficiency or an increase in generated gas.
另外,1580cm-1的拉曼半值宽度没有特别的限制,通常为10cm-1以上,优选15cm-1以上,另外,其上限通常为50cm-1以下,优选为45cm-1以下,更优选为40cm-1以下的范围。如果拉曼半值宽度低于该范围,则粒子表面的结晶性过高,由于施加力学能量处理而引起的拉曼值的增加,以及由于结晶性的降低,随着充放电Li进入层间的位点有时变少,即,充电接受性有时降低。另一方面,如果高于该范围,则由于施加力学能量处理而使粒子表面的结晶性进一步降低,与非水电解液的反应性增加,有时导致效率降低或产生的气体增加。In addition, the Raman half-value width of 1580 cm -1 is not particularly limited, and is usually 10 cm -1 or more, preferably 15 cm -1 or more, and its upper limit is usually 50 cm -1 or less, preferably 45 cm -1 or less, more preferably The range below 40cm -1 . If the Raman half-value width is lower than this range, the crystallinity of the particle surface is too high, the Raman value increases due to the application of mechanical energy treatment, and due to the decrease in crystallinity, Li enters the interlayer due to charge and discharge. The number of sites may decrease, that is, the charge acceptance may decrease. On the other hand, if it is higher than this range, the crystallinity of the particle surface will further decrease due to the application of mechanical energy treatment, and the reactivity with the non-aqueous electrolyte solution will increase, resulting in a decrease in efficiency or an increase in generated gas.
(5)BET比表面积(5) BET specific surface area
使用BET法测定的原料石墨类碳质粒子的比表面积通常为0.05m2/g以上,优选为0.2m2/g以上,更优选为0.5m2/g以上,特别优选为1m2/g以上。其上限通常为50m2/g以下,优选为25m2/g以下,更优选为15m2/g以下,特别优选为10m2/g以下。BET比表面积的值如果低于该范围,则由于施加力学能量处理而使BET比表面积增加,在充电时锂的接受性容易变差,锂容易在电极表面析出。另一方面,如果高于上述范围,则由于施加力学能量处理而使BET比表面积进一步增加,用作负极活性物质时,与非水电解液的反应性增加,产生的气体容易变多,有时难以得到优选的电池。The specific surface area of the raw graphite-based carbonaceous particles measured by the BET method is usually 0.05 m 2 /g or more, preferably 0.2 m 2 /g or more, more preferably 0.5 m 2 /g or more, particularly preferably 1 m 2 /g or more . The upper limit thereof is usually 50 m 2 /g or less, preferably 25 m 2 /g or less, more preferably 15 m 2 /g or less, particularly preferably 10 m 2 /g or less. If the value of the BET specific surface area is lower than this range, the BET specific surface area increases due to the application of mechanical energy treatment, and the acceptance of lithium tends to deteriorate during charging, and lithium tends to be deposited on the electrode surface. On the other hand, if it is higher than the above-mentioned range, the BET specific surface area is further increased due to the application of mechanical energy treatment, and when used as a negative electrode active material, the reactivity with the non-aqueous electrolyte increases, and the gas generated tends to increase, sometimes difficult Get the best battery.
(7)圆形度(7) Circularity
使用圆形度作为原料石墨类碳质粒子的球形的程度,原料石墨类碳质粒子的粒径为3~40μm范围的粒子的圆形度优选为0.1以上,更优选为0.2以上,特别优选为0.4以上,进一步优选为0.5以上,最优选为0.6以上。如果低于该范围,则即使施加力学能量处理,也不会充分地球形化,高电流密度充放电特性有时降低。Using the degree of circularity as the degree of sphericity of the raw material graphite-like carbonaceous particles, the particle diameter of the raw material graphite-like carbonaceous particles is preferably 0.1 or more, more preferably 0.2 or more, and particularly preferably 0.1 or more. 0.4 or more, more preferably 0.5 or more, most preferably 0.6 or more. If it is less than this range, even if a mechanical energy treatment is applied, the terraformation will not be sufficient, and the high current density charge and discharge characteristics may deteriorate.
(8)真密度(8) True density
原料石墨类碳质粒子的真密度通常为2g/cm3以上,优选为2.1g/cm3以上,更优选为2.2g/cm3以上,进一步优选为2.22g/cm3以上,其上限为2.26g/cm3以下。上限为石墨的理论值。如果低于该范围,则碳的结晶性过低,初期不可逆容量有时会增大。The true density of the raw graphite-based carbonaceous particles is usually 2 g/cm or more, preferably 2.1 g/cm or more, more preferably 2.2 g/cm or more, further preferably 2.22 g/cm or more, and the upper limit is 2.26 g/ cm3 or less. The upper limit is the theoretical value of graphite. If it is less than this range, the crystallinity of carbon may be too low, and the initial irreversible capacity may increase.
(9)振实密度(9) Tap density
原料石墨类碳质粒子的振实密度通常为0.05g/cm3以上,优选为0.1g/cm3以上,更优选为0.2g/cm3以上,特别优选为0.5g/cm3以上。另外,优选为2g/cm3以下,更优选为1.8g/cm3以下,特别优选为1.6g/cm3以下。振实密度如果低于该范围,则即使施加力学能量处理,振实密度的提高也不充分,在作为负极活性物质使用时,填充密度很难提高,有时不能得到高容量的电池。另一方面,如果高于该范围,则施加力学能量处理时,振实密度进一步上升,制成电极后的电极中的粒子间的空隙过少,由于非水电解液的流路不足,高电流密度充放电特性有可能降低。石墨类碳质粒子的振实密度也采用与上述相同的方法测定并定义。The tap density of the raw graphite carbonaceous particles is usually 0.05 g/cm 3 or more, preferably 0.1 g/cm 3 or more, more preferably 0.2 g/cm 3 or more, particularly preferably 0.5 g/cm 3 or more. In addition, it is preferably 2 g/cm 3 or less, more preferably 1.8 g/cm 3 or less, particularly preferably 1.6 g/cm 3 or less. If the tap density is lower than this range, the tap density will not be sufficiently improved even if mechanical energy treatment is applied, and when used as a negative electrode active material, it will be difficult to increase the packing density, and a high-capacity battery may not be obtained. On the other hand, if it is higher than this range, when the mechanical energy is applied, the tap density will further increase, and the gap between the particles in the electrode after the electrode is made is too small, and the flow path of the non-aqueous electrolyte is insufficient, and high current Density charge and discharge characteristics may decrease. The tap density of graphite-based carbonaceous particles was also measured and defined by the same method as above.
(10)取向比(粉末)(10) Orientation ratio (powder)
原料石墨类碳质粒子的取向比通常为0.001以上,优选为0.005以上。上限在理论上为0.67以下。如果低于该范围,则即使施加力学能量处理,取向比的提高也不充分,有时高密度充放电特性降低。The orientation ratio of the raw graphite-based carbonaceous particles is usually 0.001 or more, preferably 0.005 or more. The upper limit is theoretically 0.67 or less. If it is less than this range, even if a mechanical energy process is applied, the improvement of an orientation ratio will not be sufficient, and high-density charge-discharge characteristics may fall.
(11)长径比(粉末)(11) aspect ratio (powder)
石墨类碳质粒子的长径比理论上为1以上,上限通常为10以下,优选为8以下,更优选为5以下。如果超过其上限,则即使施加力学能量处理,长径比也不能充分降低,制成极板时会产生条纹、无法得到均匀的涂布面,高电流密度充放电特性有时降低。The aspect ratio of the graphite-based carbonaceous particles is theoretically 1 or more, and the upper limit is usually 10 or less, preferably 8 or less, more preferably 5 or less. If the upper limit is exceeded, the aspect ratio cannot be sufficiently reduced even if a mechanical energy treatment is applied, and streaks may occur when forming an electrode plate, a uniform coated surface may not be obtained, and high current density charge-discharge characteristics may deteriorate.
上述的原料石墨类碳质粒子中,作为碳六角网面结构发达的高结晶性碳材料,可列举使六角网面以面取向明显生长的高取向石墨、和使高取向的石墨粒子集合在等方向上的各向同性高密度石墨。作为优选的高取向石墨,可列举斯里兰卡或马达加斯加出产的天然石墨、由熔融的铁以过饱和的碳的形式析出的所谓的集结石墨、部分高石墨化度的人造石墨等。Among the above-mentioned raw material graphite-based carbonaceous particles, examples of highly crystalline carbon materials in which the carbon hexagonal network structure is developed include highly oriented graphite in which the hexagonal network surface is clearly grown in a plane orientation, and highly oriented graphite particles assembled in direction of isotropic high-density graphite. Preferable highly oriented graphites include natural graphite produced in Sri Lanka or Madagascar, so-called aggregated graphite precipitated as supersaturated carbon from molten iron, artificial graphite with a partially high graphitization degree, and the like.
天然石墨依照其性状,被分类为鳞片状石墨(Flake Graphite)、鳞状石墨(Crystalline(Vein)Graphite)、土壤石墨(Amorphous Graphite)(参见《粉粒体工艺技术集成》,((株)产业技术中心,昭和49年发行)的石墨一项;以及“HANDBOOK OF CARBON,GRAPHITE,DIAMOND AND FULLERENES”,(NoyesPublications发行))。石墨化度以鳞状石墨最高,为100%,接着是鳞片状石墨,为99.9%,而土壤石墨低至28%。作为天然石墨的鳞片状石墨产自马达加斯加、中国、巴西、乌克兰、加拿大等;鳞状石墨主要产自斯里兰卡。土壤石墨的主产地为朝鲜半岛、中国、墨西哥等。这些天然石墨中,土壤石墨通常粒径小,且纯度低。与此相反,鳞片状石墨或鳞状石墨具有石墨化度高、杂质量少等优点,因此可以优选在本发明中使用。According to its properties, natural graphite is classified into flake graphite (Flake Graphite), scaly graphite (Crystalline (Vein) Graphite), soil graphite (Amorphous Graphite) (see "Powder Granular Process Technology Integration", ((Co., Ltd.) Industry Technology Center, Graphite Item, issued in Showa 49); and "HANDBOOK OF CARBON, GRAPHITE, DIAMOND AND FULLERENES", (issued by NoyesPublications)). The degree of graphitization is highest in flake graphite, which is 100%, followed by flake graphite, which is 99.9%, while soil graphite is as low as 28%. Flake graphite as natural graphite is produced in Madagascar, China, Brazil, Ukraine, Canada, etc.; flake graphite is mainly produced in Sri Lanka. The main producing areas of soil graphite are the Korean Peninsula, China, Mexico, etc. Among these natural graphites, soil graphite usually has a small particle size and low purity. In contrast, flaky graphite or flaky graphite has advantages such as a high degree of graphitization and a small amount of impurities, and thus can be preferably used in the present invention.
人造石墨可以在非氧化性氛围下通过对石油类重油、煤类重油、石油类焦炭或煤类焦炭在1500~3000℃或其该温度以上的温度下加热来制造。Artificial graphite can be produced by heating petroleum-based heavy oil, coal-based heavy oil, petroleum coke, or coal-based coke at a temperature of 1500 to 3000° C. or higher in a non-oxidizing atmosphere.
在本发明中,只要在进行力学能量处理和热处理之后显示高取向和高容量,则可以使用任意的人造石墨作为原料。另外,在上述人造石墨中,即使是石墨化不完全的材料,例如石墨前体,只要其可以通过进行力学能量处理而成为满足上述物性的石墨质碳粒子,则可以作为本发明的力学能量处理的原料使用。In the present invention, any artificial graphite may be used as a raw material as long as it exhibits high orientation and high capacity after mechanical energy treatment and heat treatment. In addition, in the above-mentioned artificial graphite, even if it is a material with incomplete graphitization, such as a graphite precursor, as long as it can be subjected to mechanical energy treatment to become a graphitic carbon particle satisfying the above physical properties, it can be used as the mechanical energy treatment of the present invention. raw materials used.
[[[力学能量处理的内容]]][[[Mechanical energy processing content]]]
对这些原料石墨类碳质粒子进行的力学能量处理可以减小粒径,使处理后与处理前的体积平均粒径之比为1以下,并且通过该处理而提高振实密度,通过该处理还可使拉曼R值达到1.1倍以上。The mechanical energy treatment of these raw graphite carbonaceous particles can reduce the particle size, so that the ratio of the volume average particle size after the treatment to that before the treatment is less than 1, and the tap density can be improved through the treatment, and the treatment can also It can make the Raman R value more than 1.1 times.
通过进行这样的力学能量处理,作为原料的石墨类碳质粒子等碳质粒子成为如下的粒子:整体上保持高结晶性,只是粒子的表面附近变得粗糙,倾斜且露出边缘面。这样,锂离子可以出入的面增加,即使在高电流密度下也具有高的容量。By performing such a mechanical energy treatment, carbonaceous particles such as graphite-based carbonaceous particles as a raw material become particles that maintain high crystallinity as a whole, but become rough near the surface of the particles, and slope and expose edge surfaces. In this way, the surface where lithium ions can come and go increases, and it has a high capacity even at a high current density.
在可以有效利用在粉碎、分级、混合、造粒、表面改性、反应等粒子设计中的工学上的基元操作中,本发明中的“力学能量处理”属于“粉碎处理”,但同时也包含通过对表面结构进行冲击、摩擦或压缩等而产生微细的结构缺陷这样的表面处理。Among the engineering primitive operations that can be effectively used in particle design such as crushing, classification, mixing, granulation, surface modification, and reaction, the "mechanical energy processing" in the present invention belongs to "crushing processing", but it is also Including surface treatment that produces fine structural defects by impacting, rubbing, or compressing the surface structure.
一般来说,所谓粉碎处理,是指对物质施加力,使其大小减少以调节物质的粒径或粒度分布、填充性。粉碎处理依据对物质施加的力的种类、处理形态来进行分类。对物质施加的力大致分为如下4种:(1)敲打力(冲击力)、(2)压碎力(压缩力)、(3)磨碎力(磨碎力)、削刮力(剪切力)。另一方面,处理形态大致分为如下2种:在粒子内部产生龟裂并使之传播的体积粉碎、以及切去粒子表面的表面粉碎。体积粉碎可采用冲击力、压缩力、剪切力来进行;表面粉碎可采用磨碎力、剪切力来进行。粉碎处理是将对这些物质施加的力的种类和处理形态进行各种组合的处理。其组合可以根据处理目的适当确定。粉碎处理虽然也有使用爆破等化学的反应或体积膨胀来进行的情况,但通常使用粉碎机等机械装置来进行。Generally speaking, the so-called crushing treatment refers to applying force to a substance to reduce its size to adjust the particle size, particle size distribution, and filling properties of the substance. Pulverization treatment is classified according to the type of force applied to the substance and the form of treatment. The force exerted on the substance is roughly divided into the following four types: (1) knocking force (impact force), (2) crushing force (compression force), (3) grinding force (grinding force), cutting force (shearing force) cutting force). On the other hand, processing forms are roughly classified into two types: volume crushing in which cracks are generated inside the particles and propagated, and surface crushing in which the particle surfaces are cut off. Volume crushing can be carried out by impact force, compression force and shear force; surface crushing can be carried out by grinding force and shear force. The pulverization treatment is a treatment of various combinations of the type of force applied to these substances and the treatment form. The combination thereof can be appropriately determined according to the purpose of treatment. The pulverization treatment may be performed using a chemical reaction such as blasting or volume expansion, but it is usually performed using a mechanical device such as a pulverizer.
本发明的石墨碳质粒子的制造中优选使用的力学能量处理优选最终包含表面处理得粒子表面部分的粉碎(表面粉碎)所占的比例变高的处理,而与有无体积粉碎无关。这是因为,粒子表面的粉碎对于去掉石墨碳质粒子等碳质粒子的角以使粒子形状变为圆形是重要的。具体地,可以在进行一定程度的体积粉碎之后再进行表面粉碎这样地来进行力学能量处理,也可以几乎不进行体积粉碎而只进行表面粉碎这样地来进行力学能量处理。还可以同时进行体积粉碎和表面粉碎这样地来进行力学能量处理。优选最后进行表面粉碎,从粒子的表面去掉角这样地来进行力学能量处理。The mechanical energy treatment preferably used in the production of the graphitic carbonaceous particles of the present invention preferably finally includes surface treatment so that the proportion of pulverization of the particle surface (surface pulverization) increases, regardless of the presence or absence of volume pulverization. This is because pulverization of the particle surface is important to remove the corners of carbonaceous particles such as graphite carbonaceous particles to make the particle shape into a circle. Specifically, the kinetic energy treatment may be performed such that surface pulverization is performed after performing a certain volume pulverization, or the dynamic energy treatment may be performed such that only surface pulverization is performed with little volume pulverization. It is also possible to perform mechanical energy processing by simultaneously performing volume crushing and surface crushing. It is preferable to carry out surface pulverization at the end, and to perform mechanical energy treatment such that corners are removed from the surface of the particles.
本发明的力学能量处理可以减小粒径,使处理后与处理前的体积平均粒径之比为1以下,并且通过该处理而提高振实密度,通过该处理还可使拉曼R值达到1.1倍以上。The mechanical energy treatment of the present invention can reduce the particle diameter, make the ratio of the volume average particle diameter after the treatment to be below 1, and improve the tap density through the treatment, and the Raman R value can also be reached by the treatment. More than 1.1 times.
所谓“处理前后的体积平均粒径之比”,是处理后的体积平均粒径除以处理前的体积平均粒径而得到的值。(处理后的体积平均粒径)/(处理前的体积平均粒径)的值为1以下,优选为0.95以下。如果实质上为1,则通过力学能量处理提高圆形度而得到的填充性提高的效果有时很小。另外,还可以通过减小粒子大小使得处理后与处理前的平均粒径之比为1以下来控制粒子形状。The "ratio of volume average particle diameter before and after treatment" is a value obtained by dividing the volume average particle diameter after treatment by the volume average particle diameter before treatment. The value of (volume average particle diameter after treatment)/(volume average particle diameter before treatment) is 1 or less, preferably 0.95 or less. If it is substantially 1, the effect of improving the fillability by improving the circularity by mechanical energy treatment may be small. In addition, the particle shape can also be controlled by reducing the particle size so that the ratio of the average particle diameter after treatment to that before treatment is 1 or less.
通过本发明中的力学能量处理可提高振实密度。提高振实密度是指如后所述的提高以圆形度为代表的球形化程度。因此,力学能量处理必须是这样的处理。(处理后的振实密度)/(处理前的振实密度)的值为1以上,优选为1.1以上。如果低于1,则圆形度提高所带来的填充性提高的效果有时很小。The tap density can be improved by the mechanical energy treatment in the present invention. Increasing the tap density means increasing the degree of spherification represented by circularity as described later. Therefore, the mechanical energy treatment must be such a treatment. The value of (tap density after treatment)/(tap density before treatment) is 1 or more, preferably 1.1 or more. If it is less than 1, the effect of improving the fillability by improving the circularity may be small.
通过本发明中的力学能量处理可使拉曼R值为1.1倍以上。提高拉曼R值是指如后所述的降低粒子表面附近的结晶性,力学能量处理必须是这样的处理。(处理后的拉曼R值)/(处理前的拉曼R值)的值为1.1以上,优选为1.4以上。如果低于1.1,则由于拉曼R值变化所引起的充电接受性提高的效果有时很小。Through the mechanical energy treatment in the present invention, the Raman R value can be made more than 1.1 times. Improving the Raman R value means reducing the crystallinity near the particle surface as described later, and mechanical energy treatment must be such a treatment. The value of (Raman R value after treatment)/(Raman R value before treatment) is 1.1 or more, preferably 1.4 or more. If it is less than 1.1, the effect of improving charge acceptance due to changes in the Raman R value may be small.
本发明的力学能量处理是使粒子变为圆形,从而使这些粒子的振实密度上升。为了提高粉末粒子的振实密度,已知填充可以进入到粒子和粒子之间形成的空隙的更小的粒子是优选的。因此,认为如果对石墨类碳质粒子等碳质粒子进行粉碎等处理以减小粒径,则可提高振实密度,但采用这样的方法来减小粒径,一般来说振实密度反而减少。作为其原因,认为是由于粉碎使得粒子形状变得更加无定形。The mechanical energy treatment of the present invention makes the particles into a circular shape, thereby increasing the tap density of these particles. In order to increase the tap density of the powder particles, smaller particles known to fill the interstices formed between the particles and the particles are preferred. Therefore, it is considered that if the carbonaceous particles such as graphite carbonaceous particles are pulverized to reduce the particle size, the tap density can be increased. However, if such a method is used to reduce the particle size, the tap density will generally decrease. . The reason for this is considered to be that the particle shape becomes more amorphous due to pulverization.
另一方面,与粉末粒子组中的一个粒子(关注的粒子)接触的粒子的个数(配位数n)越多,填充层的空隙所占的比例越是降低。即,作为对振实密度带来影响的因素,粒子的大小的比率和组成比,即粒径分布是重要的。但是,该研究只是以模型的球形粒子组来进行的,本发明中使用的处理前的石墨类碳质粒子等碳质粒子是鳞片状、鳞状、板状的,虽然尝试了仅仅采用通常的粉碎、分级等来控制粒径分布,从而提高振实密度,但不能产生那样的高填充状态。On the other hand, as the number of particles (coordination number n) in contact with one particle (particle of interest) in the powder particle group increases, the proportion of voids in the filled layer decreases. That is, as factors affecting the tap density, the size ratio and composition ratio of particles, that is, the particle size distribution, are important. However, this research is only carried out with the spherical particle group of the model, and the carbonaceous particles such as graphite-like carbonaceous particles before treatment used in the present invention are scale-shaped, scale-like, and plate-like. Grinding, classification, etc. are used to control the particle size distribution to increase the tap density, but such a high filling state cannot be produced.
一般来说,鳞片状、鳞状、板状的石墨类碳质粒子等碳质粒子的粒径越小,其振实密度越有降低的倾向。这可认为是因为如下原因:由于粒子通过粉碎而更加无定形化,并且粒子表面生成的“毛边”、“剥离”或“折曲”等突起状物增加,以及在粒子表面以某种程度的强度附着有更微细的无定形粒子等原因,这样,与邻接粒子之间的阻抗变大,使填充性恶化。In general, the smaller the particle size of carbonaceous particles such as scaly, scaly, and plate-shaped graphite-based carbonaceous particles, the lower the tap density tends to be. This is considered to be due to the following reasons: as the particles become more amorphous by pulverization, and protrusions such as "burrs", "peeling" or "bending" generated on the surface of the particles increase, and some degree of The strength is caused by the adhesion of finer amorphous particles, etc., so that the resistance between adjacent particles becomes larger, and the filling property is deteriorated.
如果这些粒子的无定形性减少,粒子形状接近于球形,则即使粒径变小,填充性的减小也变少,在理论上,大粒径粒子、小粒径粒子都应该显示同等程度的振实密度。If the amorphousness of these particles is reduced and the particle shape is close to spherical, even if the particle size becomes smaller, the reduction in filling property will be less. In theory, both large particle size and small particle size particles should show the same degree of Tapped density.
本发明人等通过研究确认了如下内容:真密度基本相等、平均粒径也基本相等的碳质或石墨质粒子,形状越是球形,振实密度越显示高的值。即,使粒子形状带用圆形,接近于球形是重要的。如果粒子形状接近球形,则粉末的填充性也同时明显提高。本发明中,由于上述理由,施加力学能量时的指标采用粉末的振实密度。处理后的粉粒体的填充性比处理前上升时,可认为是通过使用的处理方法而使粒子球形化而带来的结果。另外,本发明的方法中,进行处理使得粒径明显降低时所获得的碳材料的振实密度与采用通常的粉碎而获得的同等程度粒径的碳材料的振实密度相比,如果显示高值时,可认为是球形化的结果。The inventors of the present invention have confirmed through studies that carbonaceous or graphite particles having substantially the same true density and substantially the same average particle diameter exhibit higher tap densities as the shape is more spherical. That is, it is important to make the shape of the particles close to a sphere rather than a circle. If the particle shape is close to spherical, the filling property of the powder is also significantly improved at the same time. In the present invention, the tap density of the powder is used as an index when mechanical energy is applied for the above reasons. When the fillability of the treated powder or granule is higher than that before the treatment, it is considered that the particle is spheroidized by the treatment method used. In addition, in the method of the present invention, if the tap density of the carbon material obtained when the particle size is significantly reduced by processing is compared with the tap density of the carbon material with the same particle size obtained by ordinary pulverization, if it shows a high When the value is , it can be considered as the result of spherification.
作为粒子的结晶性和粒子表面的粗糙度,也就是结晶的边缘面存在量的指标,可以使用通过广角X射线衍射法测定的(002)面的面间距(d002)、微晶尺寸(Lc)和拉曼R值。一般来说,碳材料越是(002)面的面间距(d002)的值小、微晶尺寸(Lc)大的材料,其拉曼R值越小。即,石墨类碳质粒子等的全部碳质粒子几乎呈现同样的结晶状态。与此相反,本发明的石墨质碳粒子虽然(002)面的面间距(d002)的值小、微晶尺寸(Lc)大,但拉曼R值采取大的值。即,石墨质碳粒子的块虽然结晶性高,但表面附近(距离粒子表面级)的结晶性混乱,表现出边缘面的露出变多。As an indicator of the crystallinity of the particles and the roughness of the particle surface, that is, the amount of edge planes of the crystals, the interplanar distance (d002) of the (002) plane and the crystallite size (Lc) measured by the wide-angle X-ray diffraction method can be used. and Raman R value. In general, the smaller the value of the interplanar distance (d002) of the (002) plane and the larger the crystallite size (Lc) of the carbon material, the smaller the Raman R value. That is, all carbonaceous particles such as graphite-based carbonaceous particles exhibit almost the same crystal state. In contrast, the graphitic carbon particles of the present invention take a large Raman R value although the value of the interplanar distance (d002) of the (002) plane is small and the crystallite size (Lc) is large. That is, although the lumps of graphitic carbon particles have high crystallinity, they are near the surface (distance from the particle surface Grade) crystallinity is disordered, showing increased exposure of edge surfaces.
从提高填充性的观点来看,更优选通过本发明中的力学能量处理使圆形度达到1.02倍,特别优选使圆形度达到1.04倍。From the viewpoint of improving fillability, it is more preferable to increase the circularity to 1.02 times, and it is particularly preferred to increase the circularity to 1.04 times by the mechanical energy treatment in the present invention.
[[[力学能量处理所使用的装置]]][[[Device used in mechanical energy processing]]]
进行力学能量处理的装置从能够进行上述的优选的处理的装置中进行选择。本发明人等在进行研究时发现,虽然使用上述对物质施加的4种力的一种以上也可以实现,但优选如下的装置:以冲击力为主,并反复对粒子施加包括粒子的相互作用在内的压缩、摩擦、剪切力等机械作用。具体地,优选如下的装置:该装置在箱内部具有设置了多个叶片的转子,并通过该转子高速旋转而对导入到内部的碳质粒子赋予冲击压缩、摩擦、剪切力等机械的作用,从而边进行体积粉碎边进行表面粉碎。另外,更优选具有通过使碳质粒子循环或对流而反复赋予机械作用的机构的装置。箱内部的叶片的数量优选3片以上,特别优选5片以上。The device for performing mechanical energy processing is selected from devices capable of performing the above-mentioned preferable processing. The inventors of the present invention found that, although one or more of the above-mentioned four kinds of forces applied to the substance can be used, the following device is preferable: the impact force is the main force, and the particle is repeatedly applied to the particle, including the interaction. Mechanical effects such as compression, friction, and shear forces. Specifically, the following device is preferable: the device has a rotor provided with a plurality of blades inside the tank, and when the rotor rotates at a high speed, mechanical effects such as impact compression, friction, and shearing force are applied to the carbonaceous particles introduced into the inside. , so that surface pulverization is performed while volume pulverization is performed. In addition, a device having a mechanism for repeatedly imparting a mechanical action by circulating or convecting the carbonaceous particles is more preferable. The number of blades inside the box is preferably 3 or more, particularly preferably 5 or more.
作为满足这样的要件的优选的装置的一个例子,可列举(株)奈良机械制作所公司制造的混合系统。使用该装置进行处理时,优选将旋转的转子的圆周速度设定为30~100m/秒,更优选设定为40~100m/秒,进一步优选设定为50~100m/秒。另外,处理可以只是仅仅使碳质粒子通过,但优选在装置内循环或滞留30秒钟以上来进行处理,更优选在装置内循环或滞留1分钟以上来进行处理。As an example of a preferable apparatus which satisfies such requirements, the mixing system by Nara Machinery Manufacturing Co., Ltd. is mentioned. When using this apparatus for processing, the peripheral speed of the rotating rotor is preferably set at 30 to 100 m/sec, more preferably at 40 to 100 m/sec, and even more preferably at 50 to 100 m/sec. In addition, the treatment may be performed by simply passing the carbonaceous particles, but it is preferably treated by circulating or staying in the device for 30 seconds or more, more preferably by circulating or staying in the device for 1 minute or more.
作为原料的石墨类碳质粒子的真密度低于2.25,并且结晶性没有那么高时,优选在进行力学能量处理之后进一步进行提高结晶性的热处理。这样的热处理优选在2000℃以上进行,更优选在2500℃以上进行,进一步优选在2800℃以上进行。When the true density of the graphite-based carbonaceous particles used as a raw material is less than 2.25 and the crystallinity is not so high, it is preferable to further perform a heat treatment for improving crystallinity after performing the mechanical energy treatment. Such heat treatment is preferably performed at 2000°C or higher, more preferably at 2500°C or higher, and even more preferably at 2800°C or higher.
[制作负极[6]的电极][Making the electrode of the negative electrode [6]]
制造负极可以采用通常的方法,可以与上述同样地形成负极[6]。集电体、集电体和活性物质层的厚度之比、电极密度、粘合剂、极板取向比、阻抗等也与上述相同。A common method can be used to produce the negative electrode, and the negative electrode can be formed in the same manner as above [6]. The current collector, the thickness ratio between the current collector and the active material layer, electrode density, binder, plate orientation ratio, impedance, and the like are also the same as those described above.
<负极[7]><Negative [7]>
下面,对本发明的锂二次电池中使用的负极[7]进行说明,该负极含有包含多元素的负极活性物质(C’)作为负极活性物质,所述包含多元素的负极活性物质(C’)含有选自Al、Pb、Zn、Sn、Bi、In、Mg、Ga、Cd、Ag、Si、B、Au、Pt、Pd和Sb的锂吸留金属(A’)和/或锂吸留合金(B’)中的至少一种,并且含有C和/或N作为元素Z。Next, the negative electrode [7] used in the lithium secondary battery of the present invention will be described. The negative electrode contains a negative electrode active material (C') containing multiple elements as the negative electrode active material. ) containing a lithium occlusion metal (A') and/or a lithium occlusion selected from the group consisting of Al, Pb, Zn, Sn, Bi, In, Mg, Ga, Cd, Ag, Si, B, Au, Pt, Pd and Sb At least one of the alloys (B'), and contains C and/or N as the element Z.
[负极[7]的负极活性物质][Negative electrode active material of negative electrode [7]]
下面,对负极[7]中使用的负极活性物质进行说明。Next, the negative electrode active material used in the negative electrode [7] will be described.
[[组成]][[composition]]
本发明的锂二次电池的负极[7]中使用的负极活性物质的特征在于,至少含有能够吸留锂的金属(锂吸留金属(A’))和/或合金(锂吸留合金(B’)),并且含有C和/或N作为元素Z。The negative electrode active material used in the negative electrode [7] of the lithium secondary battery of the present invention is characterized in that it contains at least a metal (lithium storage metal (A')) and/or an alloy (lithium storage alloy ( B')), and contains C and/or N as element Z.
所谓锂吸留金属(A’),是指选自Al、Pb、Zn、Sn、Bi、In、Mg、Ga、Cd、Ag、Si、B、Au、Pt、Pd和Sb中的一种以上。其中,优选为Al、Si、Sn或Pb,更优选为Si或Sn,进一步优选为Si。作为优选Si的理由,可列举每单位质量的容量大。The so-called lithium storage metal (A') refers to more than one selected from Al, Pb, Zn, Sn, Bi, In, Mg, Ga, Cd, Ag, Si, B, Au, Pt, Pd and Sb . Among them, Al, Si, Sn, or Pb is preferable, Si or Sn is more preferable, and Si is still more preferable. The reason why Si is preferable is that the capacity per unit mass is large.
所谓锂吸留合金(B’),是指含有2种以上上述锂吸留金属(A’)的合金,或者在锂吸留金属(A’)中含有“锂吸留金属(A’)和C、N以外的元素”的合金。作为该“锂吸留金属(A’)和C、N以外的元素”,优选选自周期表4族、5族、6族、8族、9族、10族、11族、13族、16族中的一种或2种以上的元素,更优选为Ti、Zr、V、Cr、W、B、O、Co元素,进一步优选为Ti、Zr、W、O、Co元素。由于这些元素容易形成高熔点化合物,因此在控制反应性这点来看,可以优选使用。从容量的观点来看,形成合金的“锂吸留金属(A’)和C、N以外的元素”的含量优选相对于锂吸留合金(B’)为50摩尔%以下。The so-called lithium storage alloy (B') refers to an alloy containing two or more of the above lithium storage metals (A'), or the lithium storage metal (A') contains "lithium storage metal (A') and Alloys of elements other than C and N". The "lithium storage metal (A') and elements other than C and N" are preferably selected from groups 4, 5, 6, 8, 9, 10, 11, 13, and 16 of the periodic table. One or two or more elements in the group, more preferably Ti, Zr, V, Cr, W, B, O, Co elements, still more preferably Ti, Zr, W, O, Co elements. Since these elements tend to form high-melting compounds, they can be preferably used from the viewpoint of controlling reactivity. From the viewpoint of capacity, the content of the "lithium storage metal (A') and elements other than C and N" forming the alloy is preferably 50 mol% or less based on the lithium storage alloy (B').
另外,作为元素Z的C和/或N在包含多元素的负极活性物质(C’)中的含有方式,可列举如下方式:In addition, as the C and/or N of element Z in the negative electrode active material (C ') that contains multi-element, can enumerate following mode:
1.在锂吸留金属(A’)和/或锂吸留合金(B’)中含有的状态(内含元素Z的锂吸留物质(D’));1. State contained in lithium occlusion metal (A') and/or lithium occlusion alloy (B') (lithium occlusion substance (D') containing element Z);
2.存在于锂吸留金属(A’)和/或锂吸留合金(B’)的周围的状态(元素Z外在锂吸留物质(F));2. The state existing around the lithium occlusion metal (A') and/or the lithium occlusion alloy (B') (the element Z is external to the lithium occlusion substance (F));
3.上述1和2的状态复合的状态。3. A state in which the states of 1 and 2 above are combined.
作为内含元素Z的锂吸留物质(D’)的优选的方式,优选元素Z(C和/或N)以非平衡状态存在于锂吸留金属(A’)和/或锂吸留合金(B’)中,并且特别优选锂吸留金属(A’)是Si。As a preferred mode of the lithium storage material (D') containing the element Z, it is preferable that the element Z (C and/or N) exists in the lithium storage metal (A') and/or the lithium storage alloy in a non-equilibrium state Among (B'), and particularly preferably, the lithium-occluding metal (A') is Si.
作为元素Z外在锂吸留物质(F)的优选的方式,优选元素Z是C,且C可以表现出导电性的状态。As a preferred form of the element Z-external lithium storage material (F), it is preferable that the element Z is C, and that C can exhibit conductivity.
本发明中使用的作为负极活性物质的包含多元素的负极活性物质(C’),含有锂吸留金属(A’)和/或锂吸留合金(B’),并含有C和/或N为必须成分,优选进一步含有选自周期表4族、5族、6族、8族、9族、10族、11族、13族、16族中的一种或2种以上的元素。作为这样的元素,更优选为Ti、Zr、V、Cr、W、B、O、Co元素,进一步优选为Ti、Zr、W、O、Co元素。The multi-element-containing negative electrode active material (C') used as the negative electrode active material in the present invention contains a lithium storage metal (A') and/or a lithium storage alloy (B'), and contains C and/or N As an essential component, it is preferable to further contain one or two or more elements selected from Groups 4, 5, 6, 8, 9, 10, 11, 13, and 16 of the periodic table. Such elements are more preferably Ti, Zr, V, Cr, W, B, O, and Co elements, and still more preferably Ti, Zr, W, O, and Co elements.
1.内含元素Z的锂吸留物质(D’)1. Lithium occlusion material containing element Z (D’)
本发明的锂吸留金属(A’)中,由于Si容易获得效果,因此,作为内含元素Z的锂吸留物质(D’),优选以元素Z非平衡地存在于Si中的相的化合物为主成分的通式SiZxMy(式中,Z、M、x、y如下述条件(1)~(4)所示)表示的物质。In the lithium-occluding metal (A') of the present invention, since Si is easy to obtain the effect, as the lithium-occluding material (D') containing the element Z, it is preferable that the element Z exists in Si in a non-equilibrium phase. A compound represented by the general formula SiZ x M y (wherein, Z, M, x, and y are represented by the following conditions (1) to (4)) having the main component.
(1)元素Z是包括C和/或N的元素;(1) Element Z is an element including C and/or N;
(2)元素M是选自Si和元素Z以外的元素中的一种或2种以上;(2) The element M is one or two or more elements selected from Si and elements other than Z;
(3)x是使Z浓度比Q(Z)达到0.10~0.95的值,所述Z浓度比Q(Z)是相对于以最接近于Si的组成平衡存在的化合物SiaZp(式中,a、p为整数)的Z浓度(p/(a+p)),由式Q(Z)=[x/(1+x)]/[p/(a+p)]而算出的;(3) x is a value such that the Z concentration ratio Q(Z) relative to the compound SiaZp existing in equilibrium with the composition closest to Si is 0.10 to 0.95 (wherein, a, p is an integer) Z concentration (p/(a+p)), calculated by the formula Q(Z)=[x/(1+x)]/[p/(a+p)];
(4)y为0≤y≤0.50范围的数。(4) y is a number in the range of 0≤y≤0.50.
(关于SiZxMy)(about SiZ x M y )
((元素Z))((element Z))
SiZxMy中的元素Z是包括C和/或N的元素。另外,优选C和/或N作为内含的元素Z的理由如下:Element Z in SiZ x M y is an element including C and/or N. In addition, the reasons why C and/or N are preferred as the contained element Z are as follows:
(1)能够形成比Si熔点高的化合物;(1) Can form a compound with a higher melting point than Si;
(2)共价键半径比Si小;(2) The covalent bond radius is smaller than Si;
(3)在Si中的扩散系数小;(3) The diffusion coefficient in Si is small;
(4)即使与锂反应,其体积变化也小等。(4) Even if it reacts with lithium, its volume change is small.
具体地,元素C、N可形成SiC、Si3N4等熔点比Si高的平衡存在的化合物。并且,高熔点化合物通常生成的自由能为负,是非常稳定的化合物,因此从可以有效降低Si的活度,抑制与非水电解液的反应性的观点来看,优选C和/或N作为元素Z。Specifically, elements C and N can form equilibrium compounds such as SiC and Si 3 N 4 whose melting point is higher than that of Si. And, the free energy that the high-melting point compound usually generates is negative, is a very stable compound, so from the viewpoint of effectively reducing the activity of Si and suppressing the reactivity with the non-aqueous electrolytic solution, preferably C and/or N are used as Element Z.
另外,由于元素C、N比Si的共价键原子半径小,因此可认为难以形成在SiZxMy化合物中平衡存在的化合物,在高浓度下对于元素Z更均匀分布是有效的,虽然其详细情况不很清楚,但从可以有效降低Si的活度,抑制与非水电解液的反应性的观点来看,是优选的。In addition, since the atomic radius of the covalent bond of elements C and N is smaller than that of Si, it is considered that it is difficult to form a compound that exists in equilibrium in the SiZ x M y compound, and it is effective for a more uniform distribution of the element Z at a high concentration, although its The details are not clear, but it is preferable from the viewpoint of effectively reducing the activity of Si and suppressing the reactivity with the non-aqueous electrolytic solution.
另外,元素C、N在Si中的扩散系数小,因此元素C、N分散在Si中时,伴随着充放电产生的Si的凝聚或结晶化受到抑制,从抑制Si的微粉化或与非水电解液的反应的观点来看是优选的。另外,由于元素C、N即使与锂反应,其体积变化也小,因此不易对Si的导电通路切断带来影响,故优选。In addition, the diffusion coefficients of elements C and N in Si are small, so when elements C and N are dispersed in Si, the agglomeration or crystallization of Si accompanying charge and discharge is suppressed, from suppressing the micronization of Si or combining with non-aqueous It is preferable from the viewpoint of the reaction of the electrolytic solution. In addition, even if elements C and N react with lithium, the volume change is small, and therefore it is difficult to affect the interruption of the conductive path of Si, so it is preferable.
另外,如Cu、Ni等元素那样,Cu3Si、Ni2Si等能够平衡存在的化合物比Si低熔点时,不能使Si的活度有效降低并且难以抑制与非水电解液的反应性,并且Cu、Ni元素在Si中的扩散系数大,因此伴随着充放电进行Si的凝聚或结晶化,容易引起Si的微粉化,循环特性有时不会被改善。另外,在SiZxMy化合物中平衡存在的化合物成为主成分时,不能使Si的活度降低,并且不能抑制与非水电解液的反应性,因此存在循环特性恶化等情况。In addition, like elements such as Cu and Ni, Cu 3 Si, Ni 2 Si, etc., when the compound that can exist in equilibrium is lower than Si, the activity of Si cannot be effectively reduced and it is difficult to suppress the reactivity with the non-aqueous electrolyte, and Since Cu and Ni elements have a large diffusion coefficient in Si, aggregation or crystallization of Si proceeds along with charging and discharging, and micronization of Si tends to occur, and cycle characteristics may not be improved. In addition, when the compound existing in equilibrium in the SiZ x My compound becomes the main component, the activity of Si cannot be lowered, and the reactivity with the non-aqueous electrolyte cannot be suppressed, so the cycle characteristics may deteriorate.
((元素M))((element M))
SiZxMy中的元素M是从Si和元素Z以外的元素中选择的一种或2种以上元素,优选为选自周期表4族、5族、6族、8族、9族、10族、11族、13族、16族中的一种或2种以上的元素,由于容易形成高熔点化合物,从抑制反应性这点来看,更优选为Ti、Zr、V、Cr、W、B、O元素,进一步优选为Ti、Zr、W、O元素。The element M in SiZ x M y is one or two or more elements selected from elements other than Si and element Z, preferably selected from group 4, group 5, group 6, group 8, group 9, group 10 of the periodic table One or more elements of Group 11, Group 13, and Group 16 are more preferably Ti, Zr, V, Cr, W, B, O element, more preferably Ti, Zr, W, O element.
在SiZxMy的组成中,SiZxMy的x是使Z浓度比Q(Z)达到如下范围的值,所述范围是:通常为0.10以上、优选为0.15以上、更优选为0.30以上、特别优选为0.40以上,另外,上限通常为0.95以下、优选为0.85以下、更优选为0.75以下、进一步优选为0.65以下;所述Z浓度比Q(Z)是相对于以最接近于Si的组成平衡存在的化合物SiaZp(式中,a、p为整数)的Z浓度(p/(a+p)),由式Q(Z)=[x/(1+x)]/[p/(a+p)]而算出的。另外,所说的“以最接近于Si的组成平衡存在的化合物”,是指在SiaZp中,p/(a+p)的值取最低值而平衡存在的化合物SiaZp。In the composition of SiZ x My y , x in SiZ x My y is a value such that the Z concentration ratio Q(Z) is within a range of usually 0.10 or more, preferably 0.15 or more, more preferably 0.30 or more , especially preferably 0.40 or more, in addition, the upper limit is usually 0.95 or less, preferably 0.85 or less, more preferably 0.75 or less, more preferably 0.65 or less; the Z concentration ratio Q (Z) is relative to the closest Si The Z concentration (p/(a+p)) of the compound SiaZp (wherein, a, p are integers) that composition balance exists, by formula Q(Z)=[x/(1+x)]/[p/( a+p)] and calculated. In addition, the "compound that exists in equilibrium with a composition closest to Si" refers to the compound SiaZp that exists in equilibrium with the value of p/(a+p) taking the lowest value among SiaZp.
另外,本发明中的以最接近于Si的组成平衡存在的化合物SiaZp在Si和元素Z的相图(例如,ASM International公司出版的“Desk Handbooks PhaseDiagrams for Binary Alloys”)中有记载,在本发明中,相对于该SiaZp的Z浓度(p/(a+p))设定上述的Z浓度比Q(Z),并使用Z浓度比Q(Z)的数值范围来限定x的范围。In addition, the compound SiaZp existing in the composition equilibrium closest to Si in the present invention is recorded in the phase diagram of Si and element Z (for example, "Desk Handbooks Phase Diagrams for Binary Alloys" published by ASM International Corporation), and in the present invention Here, the aforementioned Z concentration ratio Q(Z) is set with respect to the Z concentration (p/(a+p)) of the SiaZp, and the range of x is defined using the numerical range of the Z concentration ratio Q(Z).
这里所说的所谓平衡存在的化合物,是在上述相图等中作为线图的顶点记载的化合物SiaZp(式中,a、p为整数)等定比化合物,例如,Z为C时,已知SiC是稳定的化合物,在本发明中,将该化合物作为平衡存在的化合物。因此,Z为C时,SiC相当于本发明的SiaZp。另外,例如Z为N时,虽然已知Si3N4是最稳定的化合物,但还已知Si2N3、SiN也作为定比化合物存在,在本发明中,将这些所有的化合物作为平衡存在的化合物。因此,Z为N时,SiN相当于本发明的SiaZp。The compound existing in equilibrium as mentioned here is a proportional compound such as the compound Si a Z p (wherein, a and p are integers) described as the apex of the line diagram in the above-mentioned phase diagram etc., for example, when Z is C , SiC is known to be a stable compound, and in the present invention, this compound is regarded as a compound existing in equilibrium. Therefore, when Z is C, SiC corresponds to Si a Z p of the present invention. In addition, for example, when Z is N, it is known that Si 3 N 4 is the most stable compound, but it is also known that Si 2 N 3 and SiN also exist as proportional compounds. In the present invention, all these compounds are considered to be balanced compounds present. Therefore, when Z is N, SiN corresponds to Si a Z p of the present invention.
另一方面,所谓非平衡存在的相的化合物,是指平衡存在的化合物以外的化合物。非平衡存在的化合物的情况下,不形成特定的定比化合物,从宏观上看Si原子和Z原子均匀分散。On the other hand, the compound in a non-equilibrium phase refers to a compound other than a compound in equilibrium. In the case of a non-equilibrium compound, no specific ratio compound is formed, and Si atoms and Z atoms are uniformly dispersed macroscopically.
Z浓度比Q(Z)如果低于该范围,则降低Si的活度的效果小,不能抑制与非水电解液的反应性,电极膨胀变大,有时不能得到优选的循环特性。另一方面,如果超过该范围,则形成平衡存在的稳定的化合物SiaZp等,即使增加元素Z,Si的活度也不降低,有时不能抑制与非水电解液的反应性。另外,由于SiaZp等的导电性低,因此形成这样的化合物时,活性物质的导电性恶化,锂的掺杂、脱掺杂变得困难,有时不能进行充放电。If the Z concentration ratio Q(Z) is lower than this range, the effect of lowering the activity of Si is small, the reactivity with the non-aqueous electrolyte cannot be suppressed, the electrode expansion becomes large, and preferable cycle characteristics may not be obtained. On the other hand, if it exceeds this range, a stable compound such as Si a Z p in equilibrium will be formed, and even if the element Z is increased, the activity of Si will not decrease, and the reactivity with the non-aqueous electrolyte may not be suppressed. In addition, since Si a Z p and the like have low conductivity, when such a compound is formed, the conductivity of the active material deteriorates, doping and dedoping of lithium become difficult, and charging and discharging may not be performed.
这里,Z浓度比Q(Z)为1时,意味着Si形成稳定的化合物SiaZp,不优选。另外,如果大幅超过该范围,则难以获得含有Si所带来的高容量化的效果,有时不能获得优选的电池特性。Here, when the Z concentration ratio Q(Z) is 1, it means that Si forms a stable compound Si a Z p , which is not preferable. On the other hand, when this range is greatly exceeded, it is difficult to obtain the effect of increasing the capacity by containing Si, and preferable battery characteristics may not be obtained.
另外,作为元素Z,在同时使用C和N元素时,求出相对于2种元素的每一个的SiaZp基准的元素Z浓度的Z浓度比Q(Z),并将其合计值看作是Z浓度比Q(Z)。In addition, when using C and N elements together as the element Z, the Z concentration ratio Q(Z) of the element Z concentration based on the Si a Z p standard for each of the two elements is obtained, and the total value is viewed as The operation is Z concentration ratio Q(Z).
SiZxMy中的y是满足0≤y≤0.5的实数。化合物SiZxMy中含有元素M,且y≠0时,化合物SiZxMy中的元素M的比例y通常为0.08以上,优选为0.10以上,另外,其上限通常为0.50以下,优选为0.40以下,更优选为0.30以下。如果y超过该范围,则Si的含量变少,有时难以得到高容量。实质上不含元素M时,元素M的比例y为y=0或y≒0。在本发明中,所谓y≒0是指在本发明涉及的负极活性物质的制造工序等中,不可避免地含有元素M(实质上不含M)的情况,例如,y不到0.08的情况。y in SiZ x M y is a real number satisfying 0≤y≤0.5. When the compound SiZ x M y contains the element M, and y≠0, the ratio y of the element M in the compound SiZ x M y is usually 0.08 or more, preferably 0.10 or more, and the upper limit is usually 0.50 or less, preferably 0.40 or less, more preferably 0.30 or less. When y exceeds this range, the Si content decreases and it may be difficult to obtain a high capacity. When the element M is substantially not contained, the ratio y of the element M is y=0 or y≒0. In the present invention, y≒0 refers to the case where the element M is unavoidably contained (substantially free of M) in the production process of the negative electrode active material related to the present invention, for example, the case where y is less than 0.08.
包含多元素的负极活性物质(C’)的组成可以按照通常的方法求出,例如,使用X射线光电分光器(例如,ulvac-phi公司制造的“ESCA”),使含有负极化合物一侧朝上放置在试料台上,并使其表面平坦,以铝的Kα射线作为X射线源,一边进行Ar溅射一边进行深度曲线(デプスプロファィル,depthprofile)测定,分别计算出Si、元素Z、元素M等的原子浓度。The composition of the negative electrode active material (C') that contains multiple elements can be obtained according to the usual method, for example, using an X-ray photoelectric spectrometer (for example, "ESCA" manufactured by ulvac-phi company), so that the side containing the negative electrode compound faces Place it on the sample table and make the surface flat, use aluminum Kα rays as the X-ray source, carry out Ar sputtering while performing depth profile (depth profile) measurement, respectively calculate Si, element Z, Atomic concentrations of elements M etc.
(SiCxOy的组成)(Composition of SiC x O y )
元素Z为C、且元素M为O时,在通式SiCxOy中,x通常为0.053以上,优选为0.08以上,更优选为0.15以上,特别优选为0.25以上,其上限通常为0.90以下,优选为0.75以下,更优选为0.60以下,特别优选为0.45以下。另外,y通常为0以上,优选为大于0的值,特别优选为0.08以上,更优选为0.10以上,另外,其上限通常为0.50以下,优选为0.40以下,特别优选为0.30以下。When the element Z is C and the element M is O, in the general formula SiC x O y , x is usually 0.053 or more, preferably 0.08 or more, more preferably 0.15 or more, particularly preferably 0.25 or more, and the upper limit thereof is usually 0.90 or less , preferably 0.75 or less, more preferably 0.60 or less, particularly preferably 0.45 or less. In addition, y is usually 0 or more, preferably a value greater than 0, particularly preferably 0.08 or more, more preferably 0.10 or more, and its upper limit is usually 0.50 or less, preferably 0.40 or less, particularly preferably 0.30 or less.
(内含元素Z的锂吸留物质(D’)中的元素Z在Si中的存在状态)(Existence state of element Z in Si in lithium occlusion material (D') containing element Z)
在本发明的负极化合物SiZxMy中,对于元素Z在Si中的存在状态,其X射线衍射测定的XIsz值没有特别限制,元素Z为C时,优选为1.2以下,更优选为0.7以下。元素Z为N时,优选为1.1以下,更优选为1.0以下。XIsz值为该范围以下,是指以元素Z非平衡地存在于Si中的相为主成分,而SiaZp等平衡存在的化合物不是主成分,不会产生下述的XIsz值超过上述范围的情况的问题,故优选。XIsz值超过上述范围时,即SiaZp等平衡存在的化合物的相为主成分(元素Z为C时是碳化硅、元素Z为N时是氮化硅)时,存在以下情况:不会使Si的活度降低,不能抑制与非水电解液的反应性,循环特性恶化的情况;由于SiaZp等的导电性低,活性物质薄膜的导电性恶化,锂的掺杂、脱掺杂变得困难,从而不能进行充放电的情况;或者每单位质量活性物质的放电容量变小的情况。XIsz值的下限值通常为0.00以上。In the negative electrode compound SiZ x M y of the present invention, the XIsz value measured by X-ray diffraction is not particularly limited for the state of the element Z in Si. When the element Z is C, it is preferably 1.2 or less, more preferably 0.7 or less . When the element Z is N, it is preferably 1.1 or less, more preferably 1.0 or less. When the XIsz value is below this range, it means that the phase in which the element Z exists non-equilibrium in Si is the main component, and the compound existing in equilibrium such as Si a Z p is not the main component, and the following XIsz value exceeding the above range will not occur. The problem of the situation, it is preferred. When the XIsz value exceeds the above range, that is, when the phase of the compound that exists in equilibrium such as Si a Z p is the main component (silicon carbide when the element Z is C, and silicon nitride when the element Z is N), there are the following cases: no The activity of Si is reduced, the reactivity with the non-aqueous electrolyte cannot be suppressed, and the cycle characteristics are deteriorated; due to the low conductivity of Si a Z p , etc., the conductivity of the active material film is deteriorated, and the doping and dedoping of lithium The case where the charge and discharge cannot be performed due to the impurity of impurities, or the case where the discharge capacity per unit mass of the active material becomes small. The lower limit of the XIsz value is usually 0.00 or more.
((X射线衍射测定方法))((X-ray diffraction measurement method))
通过X射线衍射测定的XIsz值例如可以将本发明的负极活性物质设置成照射面,并使用X射线衍射装置(例如,理学(リガク)公司制造的“X射线衍射装置”)来测定,对于测定条件,如后述的实施例中所示。The XIsz value measured by X-ray diffraction can be, for example, provided with the negative electrode active material of the present invention as an irradiation surface, and can be measured using an X-ray diffraction device (for example, "X-ray diffraction device" manufactured by Rigaku Corporation). Conditions are as shown in Examples described later.
另外,XIsz值的定义如下。In addition, the definition of the XIsz value is as follows.
(((元素Z为C时的XIsz值)))(((XIsz value when element Z is C)))
测定2θ为35.7度的峰强度Isz、2θ为28.4度的峰强度Is,计算出其强度比XIsz(XIsz=Isz/Is),将其定义为活性物质薄膜的XIsz。这里,2θ为35.7度的峰认为是来自于SiC的峰,28.4度的峰是来自于硅的峰,XIsz值为1.2以下是指几乎未检测出SiC。The peak intensity Isz at 2θ of 35.7 degrees and the peak intensity Is of 28.4 degrees at 2θ were measured, and the intensity ratio XIsz (XIsz=Isz/Is) was calculated and defined as XIsz of the active material thin film. Here, the peak with a 2θ of 35.7 degrees is considered to be a peak derived from SiC, the peak at 28.4 degrees is a peak derived from silicon, and the XIsz value of 1.2 or less means that almost no SiC was detected.
(((元素Z为N时的XIsz值)))(((XIsz value when element Z is N)))
测定2θ为70.2度的峰强度Isz、2θ为28.4度的峰强度Is,计算出其强度比XIsz(XIsz=Isz/Is),将其定义为活性物质薄膜的XIsz。这里,2θ为27.1度的峰认为是来自于Si3N4的峰,28.4度的峰是来自于硅的峰,XIsz值为1.1以下是指几乎未检测出Si3N4。The peak intensity Isz at 2θ of 70.2 degrees and the peak intensity Is of 28.4 degrees in 2θ were measured, and the intensity ratio XIsz (XIsz=Isz/Is) was calculated and defined as XIsz of the active material thin film. Here, the peak with a 2θ of 27.1 degrees is considered to be a peak derived from Si 3 N 4 , the peak at 28.4 degrees is a peak derived from silicon, and the XIsz value of 1.1 or less means that almost no Si 3 N 4 was detected.
(内含元素Z的锂吸留物质(D’)中的元素Z的分布状态)(Distribution state of element Z in lithium occlusion material (D') containing element Z)
本发明的SiZxMy中的元素Z例如以原子或分子、或者原子簇(cluster)等1μm以下水平的大小存在,元素Z的分布状态优选在SiZxMy中均匀分布,更优选从SiZxMy的中心部向表面方向使元素Z的浓度梯度变高地倾斜(后述的薄膜状负极材料的情况下,从与集电体的接触部分向薄膜表面使浓度梯度变高地倾斜;粉末状负极材料的情况下,从粒子中心部向薄膜表面使浓度梯度变高地倾斜)分布。在负极活性物质中,不均匀地局部存在元素Z的分布时,伴随着Si的充放电引起的膨胀/收缩集中发生在不存在元素Z的Si部分,因此,随着循环的进行,导电性有时恶化。元素Z的分散状态如后面所示,可通过EPMA等确认。The element Z in SiZ x My y of the present invention exists, for example, in the size of an atom or a molecule or an atomic cluster (cluster) of 1 μm or less, and the distribution state of the element Z is preferably uniformly distributed in SiZ x My , more preferably from SiZ The central part of x M y slopes toward the surface direction so that the concentration gradient of the element Z becomes higher (in the case of the film-shaped negative electrode material described later, the concentration gradient becomes higher from the contact part with the current collector to the surface of the film; powdery In the case of the negative electrode material, the concentration gradient is increased from the center of the particle to the surface of the thin film) and distributed obliquely. In the negative electrode active material, when the distribution of the element Z is unevenly localized, the expansion/contraction caused by the charge and discharge of Si occurs concentratedly in the Si part where the element Z does not exist, so as the cycle progresses, the conductivity sometimes decreases. deterioration. The dispersed state of the element Z is shown below, and can be confirmed by EPMA or the like.
(元素M的分布状态)(distribution state of element M)
本发明的SiZxMy中的元素M的分布状态没有特别限制,可以是均匀分布,也可以是不均匀分布。The distribution state of the element M in SiZ x M y of the present invention is not particularly limited, and may be uniformly distributed or non-uniformly distributed.
(拉曼RC值、拉曼RSC值、拉曼RS值)(Raman RC value, Raman RSC value, Raman RS value)
本发明中的内含元素Z的锂吸留物质(D’)通过拉曼光谱分析测定的拉曼RC值通常为0.0以上,其上限优选为2.0以下。如果拉曼RC值超过该范围,则难以获得含有Si所带来的高容量化的效果,难以得到优选的电池特性。特别是元素Z含有C时,关于本发明的负极活性物质SiZxMy的拉曼RC值,优选为2.0以下,更优选为1.0以下,特别优选为0.5以下。由于测定上的原因,拉曼RC值的下限值通常为0.0以上。The lithium occlusion substance (D') containing the element Z in the present invention has a Raman RC value measured by Raman spectroscopic analysis usually at least 0.0, and its upper limit is preferably at most 2.0. If the Raman RC value exceeds this range, it will be difficult to obtain the effect of increasing the capacity by containing Si, and it will be difficult to obtain preferable battery characteristics. Especially when the element Z contains C, the Raman RC value of the negative electrode active material SiZ x My y of the present invention is preferably 2.0 or less, more preferably 1.0 or less, particularly preferably 0.5 or less. For measurement reasons, the lower limit of the Raman RC value is usually 0.0 or more.
本发明中的内含元素Z的锂吸留物质(D’)通过拉曼光谱分析测定的拉曼RSC值通常为0.0以上,其上限优选为0.25以下。如果拉曼RSC值超过该范围,则导电性恶化,锂的掺杂、脱掺杂变得困难,有时不能进行充放电。另外,特别是元素Z含有C时,RSC值优选为0.25以下,更优选为0.20以下。由于测定上的原因,拉曼RSC值的下限值通常为0.0以上。The lithium occlusion substance (D') containing the element Z in the present invention has a Raman RSC value measured by Raman spectroscopic analysis usually at least 0.0, and the upper limit thereof is preferably at most 0.25. When the Raman RSC value exceeds this range, the conductivity deteriorates, doping and dedoping of lithium become difficult, and charge and discharge may not be performed. In addition, especially when the element Z contains C, the RSC value is preferably 0.25 or less, more preferably 0.20 or less. For measurement reasons, the lower limit of the Raman RSC value is usually 0.0 or more.
本发明中的内含元素Z的锂吸留物质(D’)通过拉曼光谱分析测定的拉曼RS值优选为0.40以上,更优选为0.50以上,其上限优选为1.00以下,更优选为0.90以下。如果拉曼RS值低于该范围,则循环特性有可能恶化。另一方面,如果超过该范围,则有可能不能进行充放电,不优选。特别是元素Z含有C时,RS值优选为0.40以上,更优选为0.50以上;其上限优选为0.75以下,优选为0.65以下。特别是元素Z含有N时,RS值优选为0.40以上,更优选为0.50以上;其上限优选为1.00以下,优选为0.90以下。The Raman RS value of the lithium occlusion substance (D') containing element Z in the present invention measured by Raman spectroscopic analysis is preferably 0.40 or more, more preferably 0.50 or more, and its upper limit is preferably 1.00 or less, more preferably 0.90 the following. If the Raman RS value is below this range, the cycle characteristics may deteriorate. On the other hand, if it exceeds this range, charge and discharge may not be performed, which is not preferable. Especially when the element Z contains C, the RS value is preferably 0.40 or more, more preferably 0.50 or more; the upper limit thereof is preferably 0.75 or less, preferably 0.65 or less. Especially when the element Z contains N, the RS value is preferably 0.40 or more, more preferably 0.50 or more; the upper limit thereof is preferably 1.00 or less, preferably 0.90 or less.
本发明中的通过拉曼光谱分析测定的拉曼RC值、拉曼RSC值、拉曼RS值由采用以下的拉曼测定方法的拉曼光谱分析求得,并分别如下定义。The Raman RC value, Raman RSC value, and Raman RS value measured by Raman spectroscopic analysis in the present invention are obtained by Raman spectroscopic analysis using the following Raman measurement method, and are defined as follows.
((拉曼测定方法))((Raman measurement method))
使用拉曼分光器(例如,日本分光公司制造的“拉曼分光器”),将本发明的非水电解质二次电池用负极安装在测定池中,对池内的试样表面照射氩离子激光并进行测定。通过对测定的拉曼光谱进行本底补偿,求出拉曼RC值、拉曼RSC值、拉曼RS值。另外,本底补偿如下进行:用直线连接峰的终始点,求出本底,再由峰强度减去该值。Using a Raman spectrometer (for example, "Raman spectrometer" manufactured by JASCO Corporation), the non-aqueous electrolyte secondary battery negative electrode of the present invention is installed in the measurement cell, and the sample surface in the cell is irradiated with argon ion laser and To measure. By performing background compensation on the measured Raman spectrum, the Raman RC value, Raman RSC value, and Raman RS value are obtained. In addition, the background compensation is carried out as follows: connect the end points of the peaks with a straight line to obtain the background, and then subtract this value from the peak intensity.
这里的拉曼测定条件如下,光滑处理为卷积15点的单纯平均。Here, the Raman measurement conditions are as follows, and the smoothing process is a simple average of 15 convolution points.
·氩离子激光波长:514.5nmArgon ion laser wavelength: 514.5nm
·试样上的激光功率:15~40mW·Laser power on the sample: 15~40mW
·分辨率:10~20cm-1 ·Resolution: 10~20cm -1
·测定范围:200cm-1~1900cm-1 · Measuring range: 200cm -1 ~ 1900cm -1
(((拉曼RC值)))(((Raman RC value)))
测定1300cm-1~1600cm-1附近出现的峰c的峰强度Ic、300cm-1~500cm-1附近出现的峰as的峰强度Ias,计算出其强度比RC(RC=Ic/Ias),将其定义为负极的拉曼RC值。这里,可认为峰c和峰as是分别来自于碳和硅的峰,因此,拉曼RC值反映碳的量,拉曼RC值为2.0以下是指几乎未检测出碳。Measure the peak intensity Ic of the peak c appearing around 1300cm -1 ~1600cm -1 , and the peak intensity Ias of the peak as appearing around 300cm -1 ~500cm -1 , and calculate the intensity ratio RC (RC=Ic/Ias), which is It is defined as the Raman RC value of the negative electrode. Here, peak c and peak as are considered to be peaks derived from carbon and silicon, respectively. Therefore, the Raman RC value reflects the amount of carbon, and the Raman RC value of 2.0 or less means that almost no carbon is detected.
(((拉曼RSC值)))(((Raman RSC value)))
测定650cm-1~850cm-1附近出现的峰sc的峰强度Isc、300cm-1~500cm-1附近出现的峰as的峰强度Ias,计算出其强度比RSC(RSC=Isc/Ias),将其定义为负极的拉曼RSC值。Measure the peak intensity Isc of the peak sc appearing around 650cm -1 ~850cm -1 , and the peak intensity Ias of the peak as appearing around 300cm -1 ~500cm -1 , and calculate the intensity ratio RSC (RSC=Isc/Ias), which is It is defined as the Raman RSC value of the negative electrode.
这里,可认为峰sc和峰as分别是来自于SiC和硅的峰,因此,拉曼RSC值反映SiC的量,拉曼RSC值为0.25以下是指几乎未检测出SiC。Here, the peak sc and the peak as are considered to be peaks derived from SiC and silicon, respectively. Therefore, the Raman RSC value reflects the amount of SiC, and the Raman RSC value of 0.25 or less means that almost no SiC is detected.
(((拉曼RS值)))(((Raman RS value)))
测定520cm-1的强度Is、300cm-1~500cm-1附近出现的峰as的峰强度Ias,计算出其强度比RS(RS=Is/Ias),将其定义为负极的拉曼RS值。拉曼RS值反映Si的状态。The intensity Is at 520 cm -1 and the peak intensity Ias of the peak as appearing around 300 cm -1 to 500 cm -1 were measured, and the intensity ratio RS (RS=Is/Ias) was calculated, which was defined as the Raman RS value of the negative electrode. The Raman RS value reflects the state of Si.
(IRsc值)(IRsc value)
对于进行充放电之后的本发明中使用的具有负极活性物质的负极,通过红外反射光分析测定的IRsc值优选为0.90以上,更优选为1.1以上,特别优选为1.2以上。IRsc如果低于该范围,则在循环中含有Si的负极和非水电解液反应,实质上能够进行充放电的活性物质量慢慢减少,有时难以得到优选的循环特性。IRsc值的上限值为3.0左右。另外,本发明中的负极的采用红外反射光分析测定的IRsc值由下面的利用红外分光光度计进行的红外反射光测定求得,并如下进行定义。The IRsc value of the negative electrode having the negative electrode active material used in the present invention after charging and discharging is preferably 0.90 or higher, more preferably 1.1 or higher, and particularly preferably 1.2 or higher. If the IRsc is lower than this range, the negative electrode containing Si reacts with the non-aqueous electrolyte solution during the cycle, and the amount of active material that can be substantially charged and discharged gradually decreases, and it may be difficult to obtain preferable cycle characteristics. The upper limit of the IRsc value is about 3.0. In addition, the IRsc value measured by the infrared reflected light analysis of the negative electrode in this invention is calculated|required by the following infrared reflected light measurement by the infrared spectrophotometer, and is defined as follows.
((利用红外分光光度计进行的红外反射光分析测定方法))((Infrared reflected light analysis method using infrared spectrophotometer))
将使用红外分光光度计(例如,热电子(サ一モエレクトロン,thermoelectron)公司制造的“Magna560”),进行充放电之后的锂二次电池的负极的活性物质面安装在测定池中,通过反射法进行测定。测定使用窗材料为金刚石制的反射测定用试样保持架(フオルダ一),在非活性氛围下进行。通过进行测定的红外线吸收光谱的本底补偿,求出IRsc值。另外,本底补偿如下进行:连接2000~4000cm-1范围的最小值,并将直线延长,求出本底,再由各强度减去该值。测定1600cm-1下的反射光强度Isc、1650cm-1下的反射光强度Iaco,计算出其强度比IRsc(IRsc=Isc/Iaco),将其定义为充放电之后的IRsc值。Using an infrared spectrophotometer (for example, "Magna560" manufactured by Thermoelectron (thermoelectron) Co., Ltd.), the active material surface of the negative electrode of the lithium secondary battery after charging and discharging is installed in the measurement cell, and the reflection method to measure. The measurement was carried out under an inert atmosphere using a sample holder for reflection measurement (Folder) made of diamond as a window material. The IRsc value was obtained by performing background compensation of the measured infrared absorption spectrum. In addition, the background compensation was carried out by connecting the minimum values in the range of 2000 to 4000 cm -1 and extending the straight line to obtain the background, and subtracting the value from each intensity. The reflected light intensity Isc at 1600cm -1 and the reflected light intensity Iaco at 1650cm -1 were measured, and the intensity ratio IRsc (IRsc=Isc/Iaco) was calculated, which was defined as the IRsc value after charging and discharging.
详细情况虽然还不明确,但认为Isc是来自于Si的皮膜,Iaco是来自于烷基碳酸锂的皮膜,因此,IRsc反映负极的皮膜(固体电解质界面:SEI)的状态和量比,IRsc的值为0.9以上意味着由来自于烷基碳酸锂的皮膜和来自于Si的皮膜构成。Although the details are not yet clear, it is considered that Isc is derived from the film of Si, and Iaco is derived from the film of alkyllithium carbonate. Therefore, IRsc reflects the state and quantity ratio of the film (solid electrolyte interface: SEI) of the negative electrode, and IRsc A value of 0.9 or more means that it is composed of a film derived from alkyllithium carbonate and a film derived from Si.
<作用、原理><Function, principle>
首先,对活度进行说明。一般来说,所谓活度是一种热力学浓度。对于包含物质量n1、n2...ni...的多成分体系,如果将成分i的化学势设定为μi、将纯物质的化学势设定为μi 0,则将以下式First, the activity will be described. In general, the so-called activity is a thermodynamic concentration. For a multi-component system containing substances n 1 , n 2 ...n i ..., if the chemical potential of component i is set as μ i and the chemical potential of pure substance is set as μ i 0 , then the the following formula
μi-μi 0=RTlogai μ i −μ i 0 =RTloga i
定义的ai称为活度。The defined a i is called the activity.
另外,将活度ai和浓度ci之比γi称为活度系数。In addition, the ratio γ i of the activity a i to the concentration c i is called the activity coefficient.
ai/ci=γi a i /c i =γ i
例如,将包含溶剂和溶质的某一体系作为热力学的溶液进行考虑时,活度系数是对应于将体系看作是理想溶液时的某一成分的化学势和将体系看作是真实溶液时的某一成分的真实的化学势之差的量。(1)在某一成分i为溶质的真实溶液的情况下,如果溶质的浓度变低,则体系接近于成分i为溶质的理想溶液,活度系数接近于1。相反,(2)在某一成分i为溶剂的真实溶液的情况下,如果溶剂的浓度变高,则体系接近于成分i为溶剂的理想溶液,活度系数接近于1。另外,真实溶液比理想溶液稳定时,成分i的化学势为γx<1。For example, when considering a system containing a solvent and a solute as a thermodynamic solution, the activity coefficient is the chemical potential corresponding to a certain component when the system is regarded as an ideal solution and when the system is regarded as a real solution. The amount of difference between the true chemical potentials of a component. (1) In the case of a real solution in which a certain component i is a solute, if the concentration of the solute becomes lower, the system is close to an ideal solution in which component i is a solute, and the activity coefficient is close to 1. On the contrary, (2) in the case of a real solution in which component i is a solvent, if the concentration of the solvent becomes higher, the system is close to an ideal solution in which component i is a solvent, and the activity coefficient is close to 1. In addition, when the real solution is more stable than the ideal solution, the chemical potential of component i is γx<1.
在本发明中,如果列举显示良好特性的Si为例子,成分i为Si,在被看作是溶剂的Si中通过含有被看作是溶质的元素Z,溶剂Si的活度ai降低,γi<1,含有元素Z的Si化合物(固溶体:看作是真实溶液)比Si(看作是理想溶液)稳定,其结果,可抑制与非水电解液的反应性。In the present invention, if Si showing good characteristics is cited as an example, the component i is Si, and by containing the element Z regarded as a solute in Si regarded as a solvent, the activity ai of the solvent Si decreases, and γ When i <1, the Si compound (solid solution: regarded as a real solution) containing the element Z is more stable than Si ( regarded as an ideal solution), and as a result, the reactivity with the non-aqueous electrolytic solution can be suppressed.
但是,如果形成Si和元素Z平衡存在的化合物SiaZp等,则不能有效降低Si的活度,因此,元素Z在Si中非平衡地存在是重要的。However, if a compound Si a Z p in which Si and the element Z exist in equilibrium is formed, the activity of Si cannot be effectively reduced, so it is important that the element Z exists in Si in a non-equilibrium manner.
[负极[7]的形态][Form of negative electrode [7]]
在本发明中,负极[7]中使用的负极活性物质的形态通常为薄膜状或粉末状。在本发明中,如后述的制造方法所记载,使用薄膜状活性物质的负极可通过在集电体上将活性物质层进行气相成膜而得到,粉末状活性物质的负极可以通过例如在集电体上涂布粉末状活性物质和粘合剂等而形成活性物质层。In the present invention, the form of the negative electrode active material used in the negative electrode [7] is usually film or powder. In the present invention, as described in the production method described later, the negative electrode using the thin film active material can be obtained by performing vapor phase film formation of the active material layer on the current collector, and the negative electrode of the powdery active material can be obtained by, for example, forming the active material layer on the current collector. A powdery active material, a binder, and the like are coated on the electrode to form an active material layer.
[[薄膜状活性物质]][[Film active substance]]
[[[结构]]][[[structure]]]
作为在集电体上成膜的薄膜状活性物质的结构,例如可列举柱状结构、层状结构等。Examples of the structure of the thin-film active material formed on the current collector include a columnar structure, a layered structure, and the like.
[[[膜厚]]][[[Film Thickness]]]
薄膜状活性物质的膜厚与使用该薄膜状活性物质的活性物质层的厚度相当,通常为1μm以上,优选为3μm以上,另外,其上限通常为30μm以下,优选为20μm以下,更优选为15μm以下。薄膜状活性物质的膜厚如果低于该范围,则使用该薄膜状活性物质的本发明的负极(以下,有时将使用薄膜状活性物质的负极称为“薄膜负极”)其每一枚的容量小,为了获得大容量的电池,需要大量的负极,因此,相应地,需要的正极、隔板、薄膜负极本身的集电体的总容积变大,每单位电池容积可以填充的负极活性物质实质上减少,难以增大电池容量。另一方面,如果超过该范围,则由于充放电引起的膨胀/收缩,薄膜状活性物质层有时从集电体基板上剥离,循环特性有可能恶化。The film thickness of the thin-film active material corresponds to the thickness of the active material layer using the thin-film active material, and is usually at least 1 μm, preferably at least 3 μm, and the upper limit thereof is usually 30 μm or less, preferably 20 μm or less, more preferably 15 μm the following. If the film thickness of the thin-film active material is lower than this range, the negative electrode of the present invention using the thin-film active material (hereinafter, the negative electrode using the thin-film active material may be referred to as "thin-film negative electrode") has a capacity of each Small, in order to obtain a large-capacity battery, a large number of negative electrodes are required. Therefore, correspondingly, the total volume of the current collector of the required positive electrode, separator, and thin-film negative electrode itself becomes larger, and the negative active material that can be filled per unit battery volume is essentially It is difficult to increase the battery capacity. On the other hand, if it exceeds this range, the thin-film active material layer may peel off from the current collector substrate due to expansion/shrinkage caused by charge and discharge, and the cycle characteristics may deteriorate.
[[粉末状活性物质]][[Powder Active Substance]]
[[[形状]]][[[shape]]]
作为粉末状活性物质的形状,例如可列举球形、多面体形、无定形等。Examples of the shape of the powdery active material include a spherical shape, a polyhedral shape, and an amorphous shape.
[[[体积基准平均粒径]]][[[Volume-Based Average Particle Size]]]
粉末状活性物质的体积基准平均粒径没有特别限制,通常为0.1μm以上,优选为1μm以上,更优选为3μm以上,另外,其上限通常为50μm以下,优选为30μm以下,更优选为25μm以下。如果粉末状活性物质的体积基准平均粒径低于上述范围,则由于粒径过小,难以获得粉末状活性物质间的导电通路或粉末状活性物质和后述的导电剂之间的导电通路,循环特性有时恶化。另一方面,如果超过上述范围,则在如后面所述的通过涂布在集电体上制造负极活性物质层时,有时会产生不均。The volume-based average particle diameter of the powdery active material is not particularly limited, but it is usually 0.1 μm or more, preferably 1 μm or more, more preferably 3 μm or more, and the upper limit thereof is usually 50 μm or less, preferably 30 μm or less, more preferably 25 μm or less . If the volume-based average particle size of the powdery active material is lower than the above-mentioned range, it is difficult to obtain a conductive path between the powdery active material or a conductive path between the powdery active material and a conductive agent described later because the particle size is too small, Cycle characteristics sometimes deteriorate. On the other hand, when the above-mentioned range is exceeded, unevenness may occur when the negative electrode active material layer is produced by coating on the current collector as described later.
另外,作为粉末状活性物质的体积基准平均粒径,使用如下测定的值:在测定对象中混合作为表面活性剂的聚氧乙烯(20)山梨糖醇酐单月桂酸酯的2体积%水溶液(约1mL)中,以离子交换水作为分散介质,使用激光衍射/散射式粒度分布计(例如,堀场制作所公司制造的“LA-920”),测定体积基准的平均粒径(中值粒径)。在后述的实施例中,通过该方法求出体积基准平均粒径。In addition, as the volume-based average particle diameter of the powdery active material, the value measured as follows: a 2 volume % aqueous solution of polyoxyethylene (20) sorbitan monolaurate ( About 1 mL), using ion-exchanged water as a dispersion medium, using a laser diffraction/scattering particle size distribution meter (for example, "LA-920" manufactured by Horiba Manufacturing Co., Ltd.), measure the average particle diameter (median particle size) based on volume path). In Examples described later, the volume-based average particle diameter was obtained by this method.
[[[BET比表面积]]][[[BET specific surface area]]]
粉末状活性物质的BET比表面积没有特别限制,但通常为0.1m2/g以上,优选为0.5m2/g以上,更优选为1.0m2/g以上,另外,通常为100m2/g以下,优选为30m2/g以下,更优选为15m2/g以下的范围。如果BET比表面积的值低于上述范围的下限,则在用于负极的情况下,在电池的充电时,锂的接受性容易变差,锂容易在电极表面析出,因此从安全上看不优选。另一方面,如果BET比表面积的值超过上述范围的上限,则在制成负极时,与非水电解液的反应性增加,产生的气体变多,有时难以得到优选的电池。The BET specific surface area of the powdery active material is not particularly limited, but is usually at least 0.1 m 2 /g, preferably at least 0.5 m 2 /g, more preferably at least 1.0 m 2 /g, and usually at most 100 m 2 /g , preferably not more than 30 m 2 /g, more preferably not more than 15 m 2 /g. If the value of the BET specific surface area is lower than the lower limit of the above-mentioned range, when it is used as a negative electrode, the acceptability of lithium is likely to deteriorate during battery charging, and lithium is likely to be deposited on the surface of the electrode, so it is not preferable in terms of safety. . On the other hand, if the value of the BET specific surface area exceeds the upper limit of the above-mentioned range, the reactivity with the non-aqueous electrolyte solution increases when the negative electrode is made, and a large amount of gas is generated, and it may be difficult to obtain a preferable battery.
另外,粉末状活性物质的BET比表面积使用如下测定的值:使用表面积计(例如,大仓理研制造的全自动表面积测定装置),在氮气流通、350℃下对粉末状活性物质进行15分钟预干燥,然后,使用氮气相对于大气压的相对压力值准确调整为0.3的氮氦混合气体,通过采用气体流动法的氮吸附BET1点法测定。In addition, the BET specific surface area of the powdery active material is a value measured by using a surface area meter (for example, a full-automatic surface area measuring device manufactured by Okura Riken) and pre-preparing the powdery active material at 350° C. for 15 minutes in nitrogen gas flow. After drying, it was measured by the nitrogen adsorption BET 1-point method using the gas flow method using a nitrogen-helium mixed gas in which the relative pressure value of nitrogen to the atmospheric pressure was accurately adjusted to 0.3.
[[[振实密度]]][[[Tap Density]]]
粉末状活性物质的振实密度没有特别限制,通常为0.2g/cm3以上,优选为0.3g/cm3以上,更优选为0.5g/cm3以上,另外,通常为3.5g/cm3以下,优选为2.5g/cm3以下的范围。如果振实密度低于该范围,则难以提高负极活性物质层的填充密度,有时不能得到高容量的电池。另一方面,如果超过该范围,则负极活性物质中的气孔量有时变少,有时难以得到优选的电池特性。The tap density of the powdery active material is not particularly limited, but is usually 0.2 g/cm 3 or more, preferably 0.3 g/cm 3 or more, more preferably 0.5 g/cm 3 or more, and usually 3.5 g/cm 3 or less , preferably in the range of 2.5 g/cm 3 or less. If the tap density is lower than this range, it will be difficult to increase the packing density of the negative electrode active material layer, and a high-capacity battery may not be obtained. On the other hand, if it exceeds this range, the amount of pores in the negative electrode active material may decrease, and it may be difficult to obtain preferable battery characteristics.
在本发明中,振实密度如下定义:使试样通过孔径300μm的筛子落在20cm3的振实容器内,直到粉末状活性物质充满容器的上端面后,使用粉末密度测定器(例如,seishin企业公司制造的Tap densor),进行1000次冲程长度10mm的振动,由此时的体积和试料的重量求出密度,将该密度定义为振实密度。In the present invention, the tap density is defined as follows: the sample is dropped in a 20cm container through a sieve with an aperture of 300 μm until the powdery active substance is filled with the upper end of the container, and the powder density measuring device (for example, seishin Tap densor (manufactured by the company) was vibrated 1000 times with a stroke length of 10 mm, and the density was obtained from the volume and the weight of the sample at this time, and this density was defined as the tap density.
2.元素Z外在锂吸留物质(F)2. Element Z external lithium occlusion substance (F)
所谓元素Z外在锂吸留物质(F),是指在负极中锂吸留金属(A’)和/或锂吸留合金(B’)与作为元素Z的C(碳)(碳质物质(E’))复合化而得到的物质。另外,这里所说的“复合化”表示锂吸留金属(A’)和/或锂吸留合金(B’)与碳质物质(E’)通过键合被约束的状态、被物理约束的状态、通过静电约束而保持形状的状态等。这里所说的“物理的约束”是指锂吸留金属(A’)和/或锂吸留合金(B’)夹杂在碳质物质(E’)中、连在一起的状态;所谓“静电的约束”是指锂吸留金属(A’)和/或锂吸留合金(B’)通过静电能量附着在碳质物质(E’)中的状态。另外,所谓“通过键合而被约束的状态”是指氢键、共价键、离子键等化学键合。The so-called external lithium storage material (F) of element Z refers to the lithium storage metal (A') and/or lithium storage alloy (B') and C (carbon) (carbonaceous substance) as element Z in the negative electrode. (E')) Compounded substance. In addition, the "composite" mentioned here means a state in which a lithium storage metal (A') and/or a lithium storage alloy (B') and a carbonaceous substance (E') are constrained by bonding, and a state in which they are physically constrained. state, a state in which shape is retained by electrostatic confinement, and the like. The "physical constraint" mentioned here refers to the state in which the lithium occlusion metal (A') and/or lithium occlusion alloy (B') are intermingled in the carbonaceous substance (E') and connected together; the so-called "static Constraint" refers to the state in which the lithium occlusion metal (A') and/or lithium occlusion alloy (B') is attached to the carbonaceous substance (E') by electrostatic energy. In addition, the "state bound by bonding" refers to chemical bonds such as hydrogen bonds, covalent bonds, and ionic bonds.
其中,从降低阻抗的观点看,锂吸留金属(A’)和/或锂吸留合金(B’)表面的至少一部分通过键合而具有与碳质物质(E’)的层的界面的状态是有利的。这里所说的覆盖,是在与碳质物质(E’)的表面的至少部分界面中具有化学键,并显示(1)覆盖整个表面的状态、(2)局部地覆盖碳质粒子的状态、(3)选择性地覆盖部分表面的状态、(4)存在于含有化学键的极微小区域的状态。Here, at least a part of the surface of the lithium storage metal (A') and/or the lithium storage alloy (B') has an interface with the layer of the carbonaceous substance (E') by bonding from the viewpoint of lowering the resistance. Status is favorable. The coverage mentioned here means that there is a chemical bond in at least part of the interface with the surface of the carbonaceous substance (E'), and it shows (1) a state covering the entire surface, (2) a state of partially covering carbonaceous particles, ( 3) The state of selectively covering a part of the surface, (4) The state of existing in an extremely small region containing chemical bonds.
另外,结晶性在界面上可以连续地变化,也可以不连续地进行变化。即,元素Z外在锂吸留物质(F)具有锂吸留金属(A’)和/或锂吸留合金(B’)被碳质物质(E’)覆盖和/或键合而形成的界面,该界面的结晶性优选不连续和/或连续地变化。In addition, the crystallinity may change continuously or discontinuously at the interface. That is, the external lithium storage material (F) of the element Z has a lithium storage metal (A') and/or a lithium storage alloy (B') covered and/or bonded by a carbonaceous substance (E') The interface, the crystallinity of which preferably changes discontinuously and/or continuously.
[负极[7]的碳质物质(E’)的性质][Properties of the carbonaceous material (E') of the negative electrode [7]]
[[碳质物质(E’)的组成]][[Composition of Carbonaceous Matter (E')]]
碳质物质(E’)特别优选下面所示的(a)或(b)的碳化物,还可以同时含有天然石墨、人造石墨等石墨质物质(G’)。由于石墨质(G)的结晶性非常高,因此一般来说,其导电性比石墨质(E)高,与石墨质(E)相比,由于提高导电性的效果高,因此,从提高导电性的观点看,优选与石墨质(E)同时存在。The carbonaceous substance (E') is particularly preferably a carbide of (a) or (b) shown below, and may also contain a graphite substance (G') such as natural graphite or artificial graphite. Since the crystallinity of graphite (G) is very high, generally speaking, its conductivity is higher than that of graphite (E). Compared with graphite (E), the effect of improving conductivity is high. From the viewpoint of properties, it is preferable to coexist with graphite (E).
(a)选自煤类重油、直馏类重油、分解类石油重油、芳香族烃、N环化合物、S环化合物、聚苯、有机合成高分子、天然高分子、热塑性树脂和热固化树脂中的可碳化的有机物;(a) selected from coal-based heavy oil, straight-run heavy oil, decomposed petroleum-based heavy oil, aromatic hydrocarbons, N-ring compounds, S-ring compounds, polyphenylene, organic synthetic polymers, natural polymers, thermoplastic resins, and thermosetting resins carbonizable organic matter;
(b)将这些可碳化的有机物溶解于低分子有机溶剂中而得到的溶液。(b) A solution obtained by dissolving these carbonizable organic substances in a low-molecular organic solvent.
这里,作为煤类重油,优选从软沥青到硬沥青的煤焦油沥青或者干馏液化油等;作为直馏类重油,优选常压残油、减压残油等;作为分解类石油重油,优选原油、石脑油等热分解时副产的乙烯焦油等;作为芳香族烃,优选苊烯、十环烯、蒽、菲等;作为N环化合物,优选吩嗪、吖啶等;作为S环化合物,优选噻吩、联二噻吩等;作为聚苯,优选联苯、三联苯等;作为有机高分子,优选聚氯乙烯、聚乙烯醇、聚乙烯醇缩丁醛、它们的不溶解处理物、聚丙烯腈、聚吡咯、聚噻吩、聚苯乙烯等;作为天然高分子,优选纤维素、木质素、甘露聚糖、聚半乳糖醛酸、壳聚糖、蔗糖等多糖类等;作为热塑性树脂,优选聚苯硫醚、聚苯醚等;作为热固性树脂,优选糠醇树脂、酚醛树脂、酰亚胺树脂等。Here, coal-based heavy oil is preferably coal tar pitch from soft asphalt to hard asphalt or dry distillation liquefied oil, etc.; straight-run heavy oil is preferably atmospheric residual oil, vacuum residual oil, etc.; decomposed petroleum heavy oil is preferably crude oil , naphtha and other by-products such as ethylene tar during thermal decomposition; as aromatic hydrocarbons, preferably acenaphthylene, decacyclene, anthracene, phenanthrene, etc.; as N-ring compounds, preferably phenazine, acridine, etc.; as S-ring compounds , preferably thiophene, dithiophene, etc.; as polyphenyl, preferably biphenyl, terphenyl, etc.; as organic polymers, preferably polyvinyl chloride, polyvinyl alcohol, polyvinyl butyral, their insoluble processed products, poly Acrylonitrile, polypyrrole, polythiophene, polystyrene, etc.; as natural polymers, preferably polysaccharides such as cellulose, lignin, mannan, polygalacturonic acid, chitosan, sucrose, etc.; as thermoplastic resins , preferably polyphenylene sulfide, polyphenylene ether, etc.; as the thermosetting resin, preferably furfuryl alcohol resin, phenolic resin, imide resin, etc.
碳质物质(E’)优选为上述的“可碳化的有机物”的碳化物,另外,还优选将这样的“可碳化的有机物”溶解在苯、甲苯、二甲苯、喹啉、正己烷等低分子有机溶剂中而得到的溶液的碳化物。The carbonaceous substance (E') is preferably a carbide of the above-mentioned "carbonizable organic matter". In addition, it is also preferable to dissolve such "carbonizable organic matter" in benzene, toluene, xylene, quinoline, n-hexane, etc. Molecular carbides obtained in solution in organic solvents.
作为上述(a)和(b),特别优选液态的。即,从生成与吸留金属(A)和/或锂吸留合金(B’)的界面的观点看,优选在液相中进行碳化。As the above (a) and (b), liquid ones are particularly preferred. That is, from the viewpoint of forming an interface with the storage metal (A) and/or the lithium storage alloy (B'), carbonization is preferably performed in the liquid phase.
[[碳质物质(E’)的物性]][[Physical properties of carbonaceous substance (E')]]
作为碳质物质(E’)的物性,优选同时满足下面所示的(1)~(3)中的任意一项或多项。另外,可以单独使用一种显示这样物性的碳质物质(E’),也可以以任意的组合和比例组合使用2种以上。As the physical properties of the carbonaceous substance (E'), it is preferable to simultaneously satisfy any one or more of the following (1) to (3). In addition, one kind of carbonaceous substance (E') exhibiting such physical properties may be used alone, or two or more kinds may be used in combination in any combination and ratio.
(1)X射线参数(1) X-ray parameters
对于碳质物质(E’)的物性,通过采用学振法进行的X射线衍射求得的晶格面(002面)的d值(层间距离)(下面,简记为“d002”)优选为0.38nm以下,特别优选为0.36nm以下,进一步优选为0.35nm以下。如果d值过大,则形成结晶性明显低的表面,阻抗增大,因此充电接受性提高的效果变小,从而本发明的效果变小。另外,对于下限,为石墨的理论值的0.335nm以上。Regarding the physical properties of the carbonaceous material (E'), the d value (interlayer distance) (hereinafter, abbreviated as "d002") of the lattice plane (002 plane) obtained by X-ray diffraction using the Gakushin method (hereinafter, abbreviated as "d002") is preferable It is 0.38 nm or less, particularly preferably 0.36 nm or less, further preferably 0.35 nm or less. If the d value is too large, a surface with significantly low crystallinity will be formed and the impedance will increase, so the effect of improving charge acceptance will be small, and the effect of the present invention will be small. In addition, the lower limit is 0.335 nm or more of the theoretical value of graphite.
另外,通过采用学振法的X射线衍射求得的碳材料的微晶大小(Lc)通常为1nm以上,优选为1.5nm以上的范围。如果低于该范围,则阻抗增大,充电接受性提高的效果有时变小。In addition, the crystallite size (Lc) of the carbon material obtained by X-ray diffraction using the Gakushin method is usually 1 nm or more, preferably 1.5 nm or more. If it is less than this range, the impedance increases, and the effect of improving charge acceptance may become small.
(2)拉曼R值、拉曼半值宽度(2) Raman R value, Raman half value width
使用氩离子激光拉曼光谱法测定的碳质物质(E’)部分的拉曼R值通常为0.2以上,优选为0.3以上,更优选为0.4以上,其上限通常为1.5以下,优选为1.2以下的范围。如果拉曼R值低于该范围,则粒子表面的结晶性过高,随着充放电Li进入层间的位点有时变少。即,充电接受性有时降低。另一方面,如果超过该范围,则粒子表面的结晶性降低,与非水电解液的反应性增加,有时导致效率降低或产生的气体增加。The Raman R value of the carbonaceous substance (E') portion measured by argon ion laser Raman spectroscopy is usually 0.2 or more, preferably 0.3 or more, more preferably 0.4 or more, and its upper limit is usually 1.5 or less, preferably 1.2 or less range. If the Raman R value is lower than this range, the crystallinity of the particle surface may be too high, and there may be fewer sites for Li to enter the interlayer due to charge and discharge. That is, charge acceptance may be lowered. On the other hand, if it exceeds this range, the crystallinity of the particle surface decreases, the reactivity with the non-aqueous electrolyte solution increases, and the efficiency may decrease or the generated gas may increase.
另外,碳质物质(E’)部分在1580cm-1附近的拉曼半值宽度没有特别的限制,通常为20cm-1以上,优选为30cm-1以上,另外,其上限通常为140cm-1以下,优选为100cm-1以下的范围。拉曼半值宽度如果低于该范围,则粒子表面的结晶性过高,随着充放电Li进入层间的位点有时变少。即,充电接受性有时降低。另一方面,如果超过该范围,则粒子表面的结晶性降低,与非水电解液的反应性增加,有时导致效率降低或产生的气体增加。In addition, the Raman half-value width of the carbonaceous material (E') portion near 1580 cm -1 is not particularly limited, but is usually 20 cm -1 or more, preferably 30 cm -1 or more, and its upper limit is usually 140 cm -1 or less , preferably in the range of 100 cm -1 or less. If the Raman half-value width is lower than this range, the crystallinity of the particle surface is too high, and there may be fewer sites for Li to enter the interlayer due to charge and discharge. That is, charge acceptance may be lowered. On the other hand, if it exceeds this range, the crystallinity of the particle surface decreases, the reactivity with the non-aqueous electrolyte solution increases, and the efficiency may decrease or the generated gas may increase.
(3)真密度(3) True density
碳质物质(E’)部分的真密度通常为1.4g/cm3以上,优选为1.5g/cm3以上,更优选为1.6g/cm3以上,进一步优选为1.7g/cm3以上,其上限为石墨的理论值2.26g/cm3以下。如果低于该范围,则碳的结晶性过低,初期不可逆容量有时会增大。The true density of the carbonaceous material (E') part is usually 1.4 g/cm 3 or more, preferably 1.5 g/cm 3 or more, more preferably 1.6 g/cm 3 or more, and even more preferably 1.7 g/cm 3 or more, which The upper limit is 2.26 g/cm 3 or less, the theoretical value of graphite. If it is less than this range, the crystallinity of carbon may be too low, and the initial irreversible capacity may increase.
本发明的锂二次电池的负极[7]的负极活性物质中使用的包含多元素的负极活性物质(C’),优选为吸留金属(A)和/或锂吸留合金(B’)与碳质物质(E’)复合化而得到的元素Z外在锂吸留物质(F),还优选进一步含有碳质物质(E’)以外的石墨质物质(G’)作为元素Z的碳(C)。The negative electrode active material (C') containing multiple elements used in the negative electrode active material of the negative electrode [7] of the lithium secondary battery of the present invention is preferably an occlusion metal (A) and/or a lithium occlusion alloy (B') The element Z exogenous lithium storage material (F) obtained by complexing with the carbonaceous material (E') is preferably carbon that further contains a graphite material (G') other than the carbonaceous material (E') as the element Z (C).
[[石墨质物质(G’)的组成和物性]][[Composition and properties of graphitic substance (G')]]
作为石墨质物质(G’)的组成的一例,可列举天然石墨、人造石墨和对它们实施粉碎等处理而得到的物质,作为石墨质物质(G’)的物性,优选同时满足下面所示的(1)~(3)中的任意一项或多项。另外,可以单独使用一种显示这样物性的石墨质物质(G’),也可以以任意的组合和比例组合使用2种以上。As an example of the composition of the graphitic substance (G'), natural graphite, artificial graphite, and those obtained by pulverizing them are mentioned, and the physical properties of the graphitic substance (G') preferably satisfy the following simultaneously. Any one or more of (1) to (3). In addition, one kind of graphitic substance (G') exhibiting such physical properties may be used alone, or two or more kinds may be used in combination in any combination and ratio.
(1)X射线参数(1) X-ray parameters
对于石墨质物质(G’)部分,通过采用学振法进行的X射线衍射求得的晶格面(002面)的d值(层间距离)为石墨的理论值0.335nm以上。其上限优选为0.340nm以下,更优选为0.338nm以下,特别优选为0.337nm以下。如果d值过大,则形成结晶性明显低的表面,阻抗增大,因此充电接受性提高的效果变小,从而本发明的效果变小。For the graphitic substance (G') portion, the d value (interlayer distance) of the lattice plane (002 plane) obtained by X-ray diffraction by the Gakushin method is 0.335 nm or more, the theoretical value of graphite. The upper limit thereof is preferably 0.340 nm or less, more preferably 0.338 nm or less, particularly preferably 0.337 nm or less. If the d value is too large, a surface with significantly low crystallinity will be formed and the impedance will increase, so the effect of improving charge acceptance will be small, and the effect of the present invention will be small.
另外,通过采用学振法的X射线衍射求得的石墨质物质(G’)的微晶大小(Lc)通常为10nm以上,优选为50nm以上,更优选为80nm以上的范围。如果低于该范围,则阻抗增大,因此充电接受性提高的效果有时变小。In addition, the crystallite size (Lc) of the graphitic substance (G') obtained by X-ray diffraction using the Gakushin method is usually 10 nm or more, preferably 50 nm or more, more preferably 80 nm or more. If it is less than this range, since the impedance increases, the effect of improving charge acceptance may become small.
(2)拉曼R值、拉曼半值宽度(2) Raman R value, Raman half value width
使用氩离子激光拉曼光谱法测定的石墨质物质(G’)部分的拉曼R值通常为0.01以上,优选为0.10以上,其上限通常为0.40以下,优选为0.35以下,更优选为0.25以下的范围。如果拉曼R值低于该范围,则粒子表面的结晶性过高,随着充放电Li进入层间的位点有时变少。即,充电接受性有时降低。另一方面,如果超过该范围,则粒子表面的结晶性降低,与非水电解液的反应性增加,有时导致效率降低或产生的气体增加。The Raman R value of the graphitic substance (G') portion measured by argon ion laser Raman spectroscopy is usually 0.01 or more, preferably 0.10 or more, and its upper limit is usually 0.40 or less, preferably 0.35 or less, more preferably 0.25 or less range. If the Raman R value is lower than this range, the crystallinity of the particle surface may be too high, and there may be fewer sites for Li to enter the interlayer due to charge and discharge. That is, charge acceptance may be lowered. On the other hand, if it exceeds this range, the crystallinity of the particle surface decreases, the reactivity with the non-aqueous electrolyte solution increases, and the efficiency may decrease or the generated gas may increase.
另外,石墨质物质(G’)部分在1580cm-1附近的拉曼半值宽度没有特别的限制,通常为10cm-1以上,优选为15cm-1以上,另外,其上限通常为50cm-1以下,优选为40cm-1以下的范围。拉曼半值宽度如果低于该范围,则粒子表面的结晶性过高,随着充放电Li进入层间的位点有时变少。即,充电接受性有时降低。另一方面,如果超过该范围,则粒子表面的结晶性降低,与非水电解液的反应性增加,有时导致效率降低或产生的气体增加。In addition, the Raman half-value width of the graphitic substance (G') portion near 1580 cm -1 is not particularly limited, but is usually 10 cm -1 or more, preferably 15 cm -1 or more, and its upper limit is usually 50 cm -1 or less , preferably in the range of 40 cm -1 or less. If the Raman half-value width is lower than this range, the crystallinity of the particle surface is too high, and there may be fewer sites for Li to enter the interlayer due to charge and discharge. That is, charge acceptance may be lowered. On the other hand, if it exceeds this range, the crystallinity of the particle surface decreases, the reactivity with the non-aqueous electrolyte solution increases, and the efficiency may decrease or the generated gas may increase.
(3)真密度(3) True density
石墨质物质(G’)部分的真密度通常为2.0g/cm3以上,优选为2.1g/cm3以上,更优选为2.2g/cm3以上,进一步优选为2.22g/cm3以上,其上限为石墨的理论值2.26g/cm3以下。如果低于该范围,则碳的结晶性过低,初期不可逆容量有时会增大。The true density of the graphitic substance (G') part is usually 2.0 g/cm 3 or more, preferably 2.1 g/cm 3 or more, more preferably 2.2 g/cm 3 or more, and even more preferably 2.22 g/cm 3 or more, which The upper limit is 2.26 g/cm 3 or less, the theoretical value of graphite. If it is less than this range, the crystallinity of carbon may be too low, and the initial irreversible capacity may increase.
本发明的锂二次电池的负极[7]的负极活性物质中使用的元素Z外在锂吸留物质(F)中,吸留金属(A)和/或锂吸留合金(B)与碳质物质(E)的质量比通常为20/80以上,优选为50/50以上,更优选为80/20以上,特别优选为90/10以上,且优选为99.9/0.1以下,更优选为99/1以下,特别优选为98/2以下的范围。如果超过上述范围,则有时不能获得具有碳质物质(E)的效果,而如果低于上述范围,则每单位质量的容量增加的效果有时变小。优选吸留金属(A)和/或锂吸留合金(B)相对于全部元素Z外在锂吸留物质(F)为20质量%以上。The element Z used in the negative electrode active material of the negative electrode [7] of the lithium secondary battery of the present invention is contained in the lithium occlusion substance (F), and the occlusion metal (A) and/or lithium occlusion alloy (B) and carbon The mass ratio of the substance (E) is usually 20/80 or more, preferably 50/50 or more, more preferably 80/20 or more, particularly preferably 90/10 or more, and preferably 99.9/0.1 or less, more preferably 99.9 /1 or less, particularly preferably 98/2 or less. If it exceeds the above range, the effect of having the carbonaceous substance (E) may not be obtained, and if it is below the above range, the effect of increasing the capacity per unit mass may become small. Preferably, the occlusion metal (A) and/or lithium occlusion alloy (B) is 20% by mass or more relative to the total element Z external lithium occlusion substance (F).
含有石墨质物质(G’)时,相对于碳质物质(E’)和石墨质物质(G’)的总量,石墨质物质(G’)优选为5质量%以上,更优选为20质量%以上,进一步优选为50质量%以上。另外,其上限优选为99质量%以下,更优选为95质量%以下。如果石墨质物质(G’)过多,则界面的键合变弱,有时难以获得导电性提高的效果,而如果石墨质物质(G’)过少,则有时难以获得含有石墨质物质(G’)而带来的导电性提高的效果。When the graphite substance (G') is contained, the graphite substance (G') is preferably 5% by mass or more, more preferably 20% by mass, based on the total amount of the carbonaceous substance (E') and the graphite substance (G'). % or more, more preferably 50% by mass or more. In addition, the upper limit thereof is preferably 99% by mass or less, more preferably 95% by mass or less. If there are too many graphitic substances (G'), the bonding at the interface becomes weak, and sometimes it is difficult to obtain the effect of improving the conductivity, and if there are too few graphitic substances (G'), it is sometimes difficult to obtain ') to improve the electrical conductivity.
在本发明中,元素Z外在锂吸留物质(F)的形态通常为薄膜状或粉末状。另外,在本发明中,使用了薄膜状活性物质的负极如后面的制造方法中所记载,可通过在集电体上将活性物质层进行气相成膜而得到,粉末状活性物质的负极可以通过例如在集电体上涂布粉末状活性物质和粘合剂等而形成活性物质层。In the present invention, the lithium storage substance (F) external to the element Z is usually in the form of film or powder. In addition, in the present invention, the negative electrode using the thin-film active material can be obtained by vapor-phase film-forming the active material layer on the current collector as described in the following manufacturing method, and the negative electrode of the powdery active material can be obtained by For example, an active material layer is formed by coating a powdery active material, a binder, and the like on a current collector.
元素Z外在锂吸留物质(F)为粉末状时的优选的粉末物性的范围与内含元素Z的锂吸留物质(D’)的优选的粉末物性的范围相同。The preferred ranges of powder physical properties when the lithium occlusion material (F) containing the element Z is in powder form are the same as the ranges of the preferred powder physical properties of the lithium occlusion material (D') containing the element Z.
[负极[7]的集电体][Current collector of negative electrode [7]]
(材质)(material)
作为集电体的材质,可列举铜、镍、不锈钢等,其中,优选容易加工成薄膜并且廉价的铜。铜箔有采用压延法制得的压延铜箔和采用电解法制得的电解铜箔,任何一个都可以作为集电体使用。另外,铜箔的厚度比25μm薄时,可以使用比纯铜强度高的铜合金(磷青铜、钛铜、科森合金、Cu-Cr-Zr合金等)。Examples of the material of the current collector include copper, nickel, stainless steel, and the like, and among them, copper, which is easy to process into a thin film and is inexpensive, is preferable. Copper foil includes rolled copper foil produced by calendering and electrolytic copper foil produced by electrolysis, and any of them can be used as a current collector. In addition, when the thickness of the copper foil is thinner than 25 μm, a copper alloy (phosphor bronze, titanium copper, Corson alloy, Cu-Cr-Zr alloy, etc.) that is stronger than pure copper can be used.
由采用压延法制作的铜箔制作的集电体中,由于铜结晶在压延方向排列,即使将负极紧密弯折或将负极弯折成锐角,都不易断裂,适用于小型圆筒状电池。电解铜箔如下制得:例如,在溶解有铜离子的非水电解液中浸渍金属制造的鼓,边使该鼓旋转边流通电流,由此使铜析出在鼓的表面,再将其剥离。还可以在上述压延铜箔的表面通过电解法析出铜。可以对铜箔的一面或两面进行粗糙化处理或表面处理(例如,厚度为几nm~1μm左右的铬酸盐处理、Ti等的底层处理等)。In the current collector made of copper foil produced by the rolling method, since the copper crystals are arranged in the rolling direction, even if the negative electrode is bent tightly or bent at an acute angle, it is not easy to break, and it is suitable for small cylindrical batteries. Electrodeposited copper foil is produced, for example, by immersing a metal drum in a non-aqueous electrolytic solution in which copper ions are dissolved, and passing an electric current while rotating the drum, thereby depositing copper on the surface of the drum and peeling it off. Copper can also be electrolytically deposited on the surface of the above-mentioned rolled copper foil. Roughening treatment or surface treatment (for example, chromate treatment with a thickness of about several nm to 1 μm, primer treatment such as Ti, etc.) may be performed on one or both sides of the copper foil.
(厚度)(thickness)
由铜箔等制成的集电体基板中,薄的可以制造薄的薄膜负极,从可以在相同收纳容积的电池容器内装入更大表面积的薄膜负极这点来看,是优选的,但如果太薄,则强度不足,在制造电池时的卷绕等时,担心铜箔切断,因此优选为10~70μm的厚度。在铜箔的两面形成活性物质层时,铜箔优选更薄的,但从避免伴随着充电放电产生的活性物质薄膜的膨胀/收缩而引起的龟裂的产生的观点来看,在该情况下,铜箔的更优选的厚度为8~35μm。另外,使用铜箔以外的金属箔作为集电体时,可以对应于各种金属箔使用优选的厚度,但大致为10~70μm左右的范围内。Among current collector substrates made of copper foil or the like, thinner thin film negative electrodes can be produced, and a thin film negative electrode with a larger surface area can be packed in a battery container with the same storage volume, which is preferable. However, if If it is too thin, the strength will be insufficient, and the copper foil may be cut during winding and the like during battery production, so the thickness is preferably 10 to 70 μm. When the active material layers are formed on both sides of the copper foil, the copper foil is preferably thinner, but from the viewpoint of avoiding the generation of cracks caused by the expansion/contraction of the active material film accompanying charge and discharge, in this case , the more preferable thickness of copper foil is 8-35 micrometers. In addition, when metal foil other than copper foil is used as the current collector, a preferable thickness can be used according to various metal foils, but it is generally within a range of about 10 to 70 μm.
(物性)(physical properties)
对于集电体基板来讲,还希望具有以下物性。The current collector substrate is also desired to have the following physical properties.
(1)平均表面粗糙度(Ra)(1) Average surface roughness (Ra)
以JIS B0601-1994记载的方法规定的集电体基板的活性物质薄膜形成面的平均表面粗糙度(Ra)没有特别的限制,但通常为0.01μm以上,优选为0.03μm以上,且通常为1.5μm以下,优选为1.3μm以下,特别优选为1.0μm以下。The average surface roughness (Ra) of the active material thin film formation surface of the current collector substrate specified by the method described in JIS B0601-1994 is not particularly limited, but is usually 0.01 μm or more, preferably 0.03 μm or more, and usually 1.5 μm or more. μm or less, preferably 1.3 μm or less, particularly preferably 1.0 μm or less.
通过使集电体基板的平均表面粗糙度(Ra)在上述下限和上限之间的范围内,可以期待良好的充放电循环特性。即,通过设定为上述下限值以上,与活性物质薄膜的界面面积变大,与活性物质薄膜的密合性提高。另外,平均表面粗糙度(Ra)的上限值没有特别的限制,平均表面粗糙度(Ra)超过1.5μm时,难以得到作为电池合适厚度的箔,因此优选为1.5μm以下。When the average surface roughness (Ra) of the current collector substrate is within the range between the above-mentioned lower limit and upper limit, good charge-discharge cycle characteristics can be expected. That is, by setting it as more than the said lower limit, the interface area with an active material thin film becomes large, and the adhesiveness with an active material thin film improves. In addition, the upper limit of the average surface roughness (Ra) is not particularly limited. If the average surface roughness (Ra) exceeds 1.5 μm, it is difficult to obtain a foil having an appropriate thickness as a battery, so it is preferably 1.5 μm or less.
(2)拉伸强度(2) Tensile strength
集电体基板的拉伸强度没有特别限制,通常为50N/mm2以上,优选为100N/mm2以上,更优选为150N/mm2以上。所谓拉伸强度,是指试验片达到断裂时所需要的最大拉力除以试验片的截面积而得到的值。本发明中的拉伸强度,可采用与测定伸长率同样的装置和方法测定。如果是拉伸强度高的集电体基板,则可以抑制充电/放电时由于活性物质薄膜膨胀/收缩引起的集电体基板的龟裂,从而可以得到良好的循环特性。The tensile strength of the current collector substrate is not particularly limited, but is usually 50 N/mm 2 or more, preferably 100 N/mm 2 or more, more preferably 150 N/mm 2 or more. The term "tensile strength" refers to a value obtained by dividing the maximum tensile force required for a test piece to break by the cross-sectional area of the test piece. The tensile strength in the present invention can be measured using the same apparatus and method as for measuring elongation. A current collector substrate with high tensile strength can suppress cracking of the current collector substrate due to expansion/shrinkage of the active material film during charging/discharging, thereby obtaining good cycle characteristics.
(3)0.2%耐力(3) 0.2% Stamina
集电体基板的0.2%耐力没有特别的限制,但通常为30N/mm2以上,优选为100N/mm2以上,特别优选为150N/mm2以上。所谓0.2%耐力,是指产生0.2%的塑性(永久)变形所需要的负荷大小,施加该大小的负荷后,除去负荷也会保持0.2%的变形。本发明中的0.2%耐力可通过与测定伸长率相同的装置和方法来测定。如果是0.2%耐力高的集电体,则可以抑制充电/放电时由于活性物质薄膜膨胀/收缩引起的集电体基板的塑性变形,从而可以得到良好的循环特性。The 0.2% proof strength of the current collector substrate is not particularly limited, but is usually 30 N/mm 2 or more, preferably 100 N/mm 2 or more, particularly preferably 150 N/mm 2 or more. The so-called 0.2% endurance refers to the load required to produce 0.2% plastic (permanent) deformation. After applying a load of this size, the load will also maintain 0.2% deformation. The 0.2% endurance in the present invention can be measured by the same apparatus and method as for measuring elongation. A current collector with a high 0.2% endurance can suppress plastic deformation of the current collector substrate due to expansion/shrinkage of the active material film during charging/discharging, thereby obtaining good cycle characteristics.
[负极[7]的包含多元素的负极活性物质(C’)的制造方法][Manufacturing method of negative electrode active material (C') containing multiple elements of negative electrode [7]]
本发明的包含多元素的负极活性物质(C’)(内含元素Z的锂吸留物质(D’)和元素Z外在锂吸留物质(F))的制造方法没有特别限制,例如,可通过下面列举的制造法来制造。The manufacturing method of the multi-element-containing negative electrode active material (C') of the present invention (lithium occlusion material (D') containing element Z and lithium occlusion material (F) external to element Z) is not particularly limited, for example, It can be produced by the production method listed below.
1.内含元素Z的锂吸留物质(D’)的制造方法1. Manufacturing method of lithium storage substance (D') containing element Z
<制造法1><Manufacturing method 1>
蒸镀源、溅射源或喷镀源使用如下物质中的任一种:Evaporation source, sputtering source or sputtering source use any of the following substances:
(i)Si、元素Z和元素M的组合物(其中,y=0或时为Si和元素Z的组合物);(i) Composition of Si, element Z and element M (wherein, y=0 or When it is the combination of Si and element Z);
(ii)Si、元素Z和元素M的混合物(其中,y=0或时为Si和元素Z的混合物);(ii) a mixture of Si, element Z and element M (wherein, y=0 or When it is a mixture of Si and element Z);
(iii)Si、元素Z和元素M各自的单质(各单质也可以是含有各元素的气体)(其中,y=0或时为Si和元素Z各自的单质);(iii) simple substances of Si, element Z and element M (each simple substance may also be a gas containing each element) (wherein, y=0 or When Si and element Z are the respective simple substances);
(iv)Si和元素Z的组合物或混合物与元素M的单质(也可以是含有M的气体);(iv) The composition or mixture of Si and element Z and the simple substance of element M (may also be a gas containing M);
(v)含有Si、元素Z和元素M的气体(其中,y=0或时为含有Si和元素Z的气体);(v) gas containing Si, element Z and element M (wherein, y=0 or When it is a gas containing Si and element Z);
(vi)Si单质、元素Z和元素M的组合物或混合物;(vi) Composition or mixture of Si simple substance, element Z and element M;
(vii)Si和元素M的组合物或混合物与元素Z的单质(也可以是含有M的气体),(vii) The composition or mixture of Si and element M and the simple substance of element Z (may also be a gas containing M),
并利用蒸镀法和/或溅射法以及喷镀法同时将Si、元素Z和元素M(其中,y=0或y≒0时为Si和元素Z)在上述集电体基板上形成1~30μm的厚度、优选为活性物质薄膜的膜厚一项中记载的厚度的膜。And utilize vapor deposition method and/or sputtering method and sputtering method to form Si, element Z and element M (wherein, when y=0 or y≒0, be Si and element Z) on above-mentioned current collector substrate to form 1 A thickness of ˜30 μm, preferably a film having a thickness described in the section of film thickness of the active material thin film.
(原料)(raw material)
作为蒸镀源、溅射源或喷镀(溶射)源(下面有时适当地称为“原料”)的Si单质原料,例如可以使用结晶性Si、无定形Si等。作为Z原料,可使用C、N元素。另外,元素Z只要是满足上述项目的元素即可,可以同时使用2种以上的多种元素。As an Si elemental raw material for a vapor deposition source, a sputtering source, or a sputtering (spraying) source (hereinafter, sometimes appropriately referred to as "raw material"), for example, crystalline Si, amorphous Si, or the like can be used. As the Z raw material, C and N elements can be used. In addition, as long as the element Z satisfies the above items, two or more kinds of elements may be used in combination.
原料中,作为(i)Si、元素Z和元素M的组合物(其中,y=0或y≒0时为Si和元素Z的组合物),可以使用组合了Si、元素Z和元素M,或者组合了Si和元素Z而形成的单一化合物,或者也可以制成多种化合物使用。另外,这些Si、Z原料、M原料的形态,例如可制成粉末状、颗粒状、小球状、块状、板状等使用。Among the raw materials, as (i) a composition of Si, element Z, and element M (wherein, when y=0 or y≒0, it is a composition of Si and element Z), a combination of Si, element Z, and element M can be used, Or a single compound formed by combining Si and element Z, or a plurality of compounds may be used. In addition, these Si, Z raw materials, and M raw materials can be used in the form of powder, granule, pellet, block, plate, etc., for example.
在通式SiZxMy中,y≠0且含有元素M时,元素M可以使用Si和元素Z以外的选自周期表2族、4族、8族、9族、10族、11族、13族、14族、15族和16族的元素中的一种或2种以上,优选使用Ti、Zr、W、O、Co元素,更优选使用O元素。In the general formula SiZ x M y , when y ≠ 0 and contains element M, the element M can be selected from group 2, group 4, group 8, group 9, group 10, group 11, One or two or more elements of Group 13, Group 14, Group 15, and Group 16 are preferably Ti, Zr, W, O, and Co elements, more preferably O elements.
(成膜法)(film forming method)
作为活性物质薄膜的形成方法,可列举气相成膜法,具体地,可列举蒸镀法(真空蒸镀法、CVD法、离子镀法)、溅射法、喷镀法(火焰喷涂法、等离子体喷涂法)等。还可以将溅射法和蒸镀法、溅射法和喷镀法进行组合来成膜。As a method for forming an active material thin film, a vapor phase film-forming method can be mentioned, and specifically, an evaporation method (vacuum evaporation method, CVD method, ion plating method), a sputtering method, a thermal spraying method (flame spraying method, plasma coating method) can be mentioned. body spraying method), etc. It is also possible to form a film by combining a sputtering method and a vapor deposition method, or a sputtering method and a thermal spraying method.
下面对负极活性物质薄膜的形成方法进行说明。Next, a method for forming the negative electrode active material thin film will be described.
A.溅射法A. Sputtering method
溅射法是在减压下利用等离子体使由含有上述原料的靶发出的活性物质材料与集电体基板碰撞、堆积而形成薄膜。如果采用溅射法,则形成的活性物质薄膜与集电体基板的界面状态良好,活性物质薄膜对于集电体的密合性也高。In the sputtering method, a thin film is formed by colliding and depositing an active material emitted from a target containing the above-mentioned raw material and a current collector substrate by using plasma under reduced pressure. According to the sputtering method, the interface state between the formed active material thin film and the current collector substrate is good, and the adhesiveness of the active material thin film to the current collector is also high.
作为对于靶的溅射电压的施加方法,可以使用直流电压、交流电压的任意一种,此时,对集电体基板实质上施加负的偏压,可以对来源于等离子体的离子的碰撞能量进行控制。开始形成薄膜前的腔内的极限真空度通常为0.1Pa以下以防止混入杂质。As a method of applying the sputtering voltage to the target, either a DC voltage or an AC voltage can be used. In this case, a negative bias voltage is substantially applied to the current collector substrate, and the collision energy of ions originating in the plasma can be reduced. Take control. The ultimate vacuum in the chamber before the start of film formation is usually 0.1 Pa or less to prevent contamination of impurities.
作为溅射气体,使用Ne、Ar、Kr、Xe等惰性气体。其中,从溅射效率等方面考虑,优选使用氩气。此外,化合物SiZxMy中的元素Z为N时,在上述惰性气体中同时存在微量的氮气在制造上是优选的。通常,溅射气体压力为0.05~70Pa左右。通过溅射法形成活性物质薄膜时的集电体基板可以通过水冷却或加热器等来控制温度。作为集电体基板的温度范围,通常为室温~900℃,但是优选为150℃以下。通过溅射法形成活性物质薄膜时的成膜速度通常为0.01~0.5μm/分钟。As the sputtering gas, inert gases such as Ne, Ar, Kr, and Xe are used. Among these, argon gas is preferably used from the viewpoint of sputtering efficiency and the like. In addition, when the element Z in the compound SiZ x My y is N, it is preferable in terms of production that a small amount of nitrogen gas coexists with the above-mentioned inert gas. Usually, the sputtering gas pressure is about 0.05 to 70 Pa. The temperature of the current collector substrate when the active material thin film is formed by the sputtering method can be controlled by water cooling or a heater. The temperature range of the current collector substrate is usually room temperature to 900°C, but is preferably 150°C or lower. The film forming rate when forming the active material thin film by the sputtering method is usually 0.01 to 0.5 μm/min.
而且,在形成活性物质薄膜前,可以通过反溅射或其它的等离子体处理等前处理来对集电体基板表面进行蚀刻。这样的前处理对于作为集电体基板的铜箔表面的污染物或氧化膜的除去、活性物质薄膜的密合性的提高是有效的。Furthermore, before forming the active material thin film, the surface of the current collector substrate may be etched by pretreatment such as reverse sputtering or other plasma treatment. Such pretreatment is effective for removing contaminants and oxide films on the surface of the copper foil serving as a current collector substrate, and for improving the adhesion of the active material thin film.
B.真空蒸镀法B. Vacuum evaporation method
真空蒸镀法是使作为活性物质的上述原料熔融、蒸发,从而堆积在集电体基板上的方法,一般来说,该方法具有与溅射法相比可以以更高的成膜速度形成薄膜的优点。从谋求缩短规定膜厚的活性物质薄膜的形成时间的观点来看,真空蒸镀法与溅射法相比,在制造成本方面可以有利地灵活应用。作为其具体的方法,可以举出感应加热法、电阻加热法、电子束加热蒸镀法等。感应加热法是在石墨等的蒸镀坩锅中通过感应电流使蒸镀材料加热熔融并蒸发而成膜;电阻加热法是通过通电的加热电流在蒸镀舟皿等中使蒸镀材料加热熔融并蒸发而成膜;电子束加热蒸镀是通过电子束使蒸镀材料加热熔融并蒸发而成膜。The vacuum evaporation method is a method of melting and evaporating the above-mentioned raw materials as active materials, and depositing them on the current collector substrate. Generally speaking, this method has the advantage of forming a thin film at a higher film formation rate than the sputtering method. advantage. From the viewpoint of shortening the time required to form an active material thin film having a predetermined film thickness, the vacuum vapor deposition method can be advantageously utilized in terms of production cost compared with the sputtering method. Specific methods thereof include an induction heating method, a resistance heating method, an electron beam heating vapor deposition method, and the like. The induction heating method is to heat and melt the evaporation material and evaporate it to form a film through an induction current in an evaporation crucible such as graphite; the resistance heating method is to heat and melt the evaporation material in an evaporation boat or the like through a heating current that is energized. and evaporated to form a film; electron beam heating evaporation is to heat, melt and evaporate the evaporation material to form a film through an electron beam.
作为真空蒸镀法的氛围气,一般使用真空。此外,化合物SiZxMy中的元素Z为N时,可以将微量的氮气与惰性气体一起导入并进行减压,在真空下同时形成SiZxMy。开始形成薄膜前的腔内的极限真空度通常为0.1Pa以下以防止混入杂质。As the atmosphere of the vacuum evaporation method, vacuum is generally used. In addition, when the element Z in the compound SiZ x My y is N, a trace amount of nitrogen gas can be introduced together with an inert gas and the pressure can be reduced to simultaneously form SiZ x My y under vacuum. The ultimate vacuum in the chamber before the start of film formation is usually 0.1 Pa or less to prevent contamination of impurities.
通过真空蒸镀法形成活性物质薄膜时的集电体基板可以通过加热器等控制温度。作为集电体基板的温度范围,通常为室温~900℃,但是优选为150℃以下。通过真空蒸镀法形成负极活性物质薄膜时的成膜速度通常为0.1~50μm/分钟。The temperature of the current collector substrate when the active material thin film is formed by the vacuum evaporation method can be controlled by a heater or the like. The temperature range of the current collector substrate is usually room temperature to 900°C, but is preferably 150°C or lower. The film formation rate when forming the negative electrode active material thin film by the vacuum deposition method is usually 0.1 to 50 μm/min.
另外,与溅射法的情况同样地,在集电体基板上堆积活性物质薄膜之前,可以通过用离子枪等进行离子照射来对集电体基板表面实施蚀刻处理。通过这样的蚀刻处理,可以进一步提高基板与活性物质薄膜的密合性。此外,形成薄膜时,通过使离子与集电体基板碰撞,可以进一步提高活性物质薄膜对于集电体基板的密合性。In addition, as in the case of the sputtering method, before depositing the active material thin film on the current collector substrate, the surface of the current collector substrate can be etched by ion irradiation with an ion gun or the like. Such etching can further improve the adhesion between the substrate and the active material thin film. In addition, when the thin film is formed, the adhesion of the active material thin film to the current collector substrate can be further improved by causing ions to collide with the current collector substrate.
C.CVD法C. CVD method
CVD法是通过气相化学反应使作为活性物质的上述原料堆积在集电体基板上。一般来说,CVD法具有如下特征:由于通过气体流入控制反应室内的化合物气体,因此可以高纯度地合成各种材料,作为其具体的方法,可以举出热CVD法、等离子体CVD法、光CVD法、cat-CVD法等。热CVD法是将蒸气压高的卤化合物的原料气体与载气或反应气体一起在1000℃左右导入到加热后的反应容器内,使其产生热化学反应而形成薄膜。等离子体CVD法是使用等离子体来替代热能的方法;光CVD是使用光能来替代热能的方法。cat-CVD法是催化化学气相沉积法,通过应用原料气体与加热催化剂的接触分解反应而形成薄膜。In the CVD method, the above-mentioned raw materials as active materials are deposited on the current collector substrate by a gas-phase chemical reaction. In general, the CVD method is characterized by the fact that various materials can be synthesized with high purity because the compound gas in the reaction chamber is controlled by the gas inflow. Specific methods include thermal CVD, plasma CVD, optical CVD method, cat-CVD method, etc. The thermal CVD method is to introduce the raw material gas of the halogen compound with high vapor pressure into the heated reaction vessel at about 1000°C together with the carrier gas or reaction gas, and make it produce a thermochemical reaction to form a thin film. Plasma CVD is a method of using plasma instead of thermal energy; photo CVD is a method of using light energy instead of thermal energy. The cat-CVD method is a catalytic chemical vapor deposition method that forms a thin film by applying a contact decomposition reaction of a raw material gas and a heated catalyst.
CVD法中使用的原料气体中,作为元素Si源为SiH4、SiCl4等;作为元素Z源为NH3、N2、BCl3、CH4、C2H6、C3H8等。Among the raw material gases used in the CVD method, the source of element Si is SiH 4 , SiCl 4 , etc.; the source of element Z is NH 3 , N 2 , BCl 3 , CH 4 , C 2 H 6 , C 3 H 8 , etc.
D.离子镀法D. Ion plating method
离子镀法是使作为活性物质的上述原料熔融、蒸发,在等离子体下将蒸发粒子离子化和激发,由此在集电体基板上牢固地成膜。具体来说,作为使原料熔融、蒸发的方法,可以举出感应加热法、电阻加热法、电子束加热蒸镀法等;作为离子化和激发的方法,可以举出活化反应蒸镀法、多阴极热电子照射法、高频激发法、HCD法、离子团束法(クラスタ一イオンビ一ム法)、多弧法等。另外,可以将蒸发上述原料的方法和离子化和激发的方法适当选择组合来进行。In the ion plating method, the above-mentioned raw material as an active material is melted and evaporated, and evaporated particles are ionized and excited under plasma, whereby a film is firmly formed on the current collector substrate. Specifically, methods for melting and evaporating raw materials include induction heating, resistance heating, and electron beam heating evaporation; methods for ionizing and exciting include activation reaction evaporation, poly Cathodic thermionic irradiation method, high-frequency excitation method, HCD method, ion beam method (klasta-ionbiim method), multi-arc method, etc. In addition, the method of evaporating the above-mentioned raw materials and the method of ionization and excitation can be appropriately selected and combined.
E.喷镀法E. Sputtering method
喷镀法是通过加热使作为活性物质的上述原料熔融或软化,形成微粒子状而加速,从而使粒子凝固、堆积在集电体基板上。作为其具体的方法,可以举出火焰喷镀法、电弧喷镀法、直流等离子体喷镀法、RF等离子体喷镀法、激光喷镀法等。In the sputtering method, the above-mentioned raw material as an active material is melted or softened by heating to form fine particles and accelerated, so that the particles are solidified and deposited on the current collector substrate. Specific methods thereof include a flame spraying method, an arc spraying method, a DC plasma spraying method, an RF plasma spraying method, a laser spraying method, and the like.
F.溅射法与蒸镀法的组合F. Combination of sputtering method and evaporation method
利用蒸镀法的高成膜速度的优点和溅射法的对集电体基板的强成膜密合性的优点,例如,通过溅射法形成第1薄膜层,然后通过蒸镀法高速形成第2薄膜层,由此可以形成与集电体基板的密合性优异的界面区域,同时以高成膜速度形成活性物质薄膜。利用这样的成膜方法的混合的组合方法,可以高效地制造充放电容量高且充放电循环特性优异的薄膜负极。Utilizing the advantages of high film-forming speed of the vapor deposition method and the strong film-forming adhesion to the current collector substrate of the sputtering method, for example, the first thin film layer is formed by the sputtering method and then formed at a high speed by the vapor deposition method The second thin film layer can thereby form an interface region with excellent adhesion with the current collector substrate, and at the same time form an active material thin film at a high film formation rate. A thin-film negative electrode having a high charge-discharge capacity and excellent charge-discharge cycle characteristics can be efficiently produced by such a mixed combination method of film-forming methods.
将溅射法与蒸镀法组合来形成活性物质薄膜优选保持减压氛围气而连续地进行。这是由于通过不暴露在大气下而连续地形成第1薄膜层和第2薄膜层,可以防止混入杂质。例如,优选使用如下的薄膜形成装置:在同一真空环境中,使集电体基板移动,同时依次进行溅射和蒸镀。The combination of sputtering and vapor deposition to form an active material thin film is preferably performed continuously while maintaining a reduced pressure atmosphere. This is because contamination of impurities can be prevented by continuously forming the first thin film layer and the second thin film layer without exposing them to the atmosphere. For example, it is preferable to use a thin film forming apparatus that sequentially performs sputtering and vapor deposition while moving the current collector substrate in the same vacuum environment.
在本发明中,通过这样的成膜法在集电体基板的两面上形成活性物质薄膜时,在集电体基板的一面上形成活性物质薄膜层(也可以是上述第1薄膜层和第2薄膜层的组合)和在集电体基板的另一面上形成活性物质薄膜层(也可以是上述第1薄膜层和第2薄膜层的组合),优选在保持减压氛围气的状态下连续进行。In the present invention, when the active material thin film is formed on both sides of the current collector substrate by such a film-forming method, the active material thin film layer (may also be the above-mentioned first thin film layer and second thin film layer) is formed on one side of the current collector substrate. Combination of thin film layers) and forming an active material thin film layer (also can be the combination of the first thin film layer and the second thin film layer) on the other side of the current collector substrate, preferably continuously under the state of maintaining a reduced pressure atmosphere .
<制造法2><Manufacturing method 2>
下面对通式SiZxMy中元素Z为C时的制造方法进行说明。The production method when the element Z in the general formula SiZ x M y is C will be described below.
蒸镀源、溅射源或喷镀源使用下述物质中的任一种:Evaporation sources, sputter sources or sputter sources use any of the following:
(i)Si、C和元素M的组合物(其中,y=0或y≒0时为Si和C的组合物);(i) the composition of Si, C and element M (wherein, when y=0 or y≒0, it is the composition of Si and C);
(ii)Si、C和元素M的混合物(其中,y=0或y≒0时为Si和C的混合物);(ii) A mixture of Si, C and element M (wherein, when y=0 or y≒0, it is a mixture of Si and C);
(iii)Si、C和元素M各自的单质(其中,y=0或y≒0时为Si和C各自的单质);(iii) simple substances of Si, C and element M (wherein, when y=0 or y≒0, they are simple substances of Si and C);
(iv)Si和C的组合物或混合物与元素M的单质(也可以为含有M的气体);(iv) The composition or mixture of Si and C and the simple substance of element M (may also be a gas containing M);
(v)含有Si、C和元素M的气体(其中,y=0或y≒0时为含有Si和C的气体);(v) Gas containing Si, C and element M (wherein, when y=0 or y≒0, it is a gas containing Si and C);
(vi)Si单质与C和元素M的组合物或混合物;(vi) Composition or mixture of Si elemental substance and C and element M;
(vii)Si和元素M的组合物或混合物与C单质,(vii) Compositions or mixtures of Si and element M with C alone,
并利用蒸镀法和/或溅射法以及喷镀法同时将Si、C和元素M(其中,y=0或y≒0时为Si和C)在上述集电体基板上形成1~30μm的厚度、优选为活性物质薄膜的膜厚项中记载的厚度的膜。And utilize vapor deposition method and/or sputtering method and sputtering method to form Si, C and element M (wherein, when y=0 or y≒0, be Si and C) on above-mentioned current collector substrate and form 1~30 μm The thickness is preferably a film with the thickness described in the item of film thickness of the active material thin film.
(原料)(raw material)
作为蒸镀源或溅射源(下面有时适当地称为“原料”)的Si原料,例如可以使用结晶性Si、无定形Si等。作为C原料,例如可以使用天然石墨、人造石墨等碳材料。作为M原料,通常为Si和元素Z以外的周期表2族、4族、8族、9族、10族、11族、13族、14族、15族和16族的元素,优选使用Ti、Zr、W、O、Co元素,特别优选使用O元素。As a Si raw material for a vapor deposition source or a sputtering source (hereinafter sometimes referred to as "raw material"), for example, crystalline Si, amorphous Si, or the like can be used. As the C raw material, for example, carbon materials such as natural graphite and artificial graphite can be used. As the M raw material, it is usually an element of Group 2, Group 4, Group 8, Group 9, Group 10, Group 11, Group 13, Group 14, Group 15, and Group 16 of the periodic table other than Si and the element Z. Preferably, Ti, Among Zr, W, O, and Co elements, O element is particularly preferably used.
原料中,作为(i)Si、C和元素M的组合物,可以使用Si、C和元素M组合而形成的单一化合物,或者也可以制成多种化合物使用。另外,这些Si、C、M原料的形态,例如可制成粉末状、颗粒状、小球状、块状、板状等使用。此外,元素M可以制成Si或C的氮化物、Si或C的氧化物使用,对于常温下作为气体存在的O等,在Si、C成膜中作为原料气体O等共存在制造上是优选的。Among the raw materials, as the composition of (i) Si, C, and element M, a single compound obtained by combining Si, C, and element M may be used, or a plurality of compounds may be used. In addition, the forms of these Si, C, and M raw materials can be used in, for example, powder, granule, pellet, block, plate, and the like. In addition, the element M can be used as a nitride of Si or C, or an oxide of Si or C. For O, etc. that exist as a gas at room temperature, it is preferable to coexist as a raw material gas O, etc. in Si and C film formation. of.
(成膜法)(film forming method)
使用与上述制造法1同样的成膜法。The same film-forming method as that of Production Method 1 above was used.
A.溅射法A. Sputtering method
作为溅射气体,使用Ne、Ar、Kr、Xe等惰性气体。其中,从溅射效率等方面考虑,优选使用氩气。此外,通式SiCxMy中的M元素为O时,在上述惰性气体中分别共存微量的氧气在制造上是优选的。通常,溅射气体压力为0.05~70Pa左右。As the sputtering gas, inert gases such as Ne, Ar, Kr, and Xe are used. Among these, argon gas is preferably used from the viewpoint of sputtering efficiency and the like. In addition, when the M element in the general formula SiC x My y is O, it is preferable in terms of production to coexist a small amount of oxygen in each of the above inert gases. Usually, the sputtering gas pressure is about 0.05 to 70 Pa.
B.真空蒸镀法B. Vacuum evaporation method
作为真空蒸镀法的氛围气,一般使用真空。此外,通式SiCxMy中的元素M为O时,可以分别将微量的氧气与惰性气体一起导入,同时进行减压,从而在真空下同时形成Si/C/M。As the atmosphere of the vacuum evaporation method, vacuum is generally used. In addition, when the element M in the general formula SiC x M y is O, a small amount of oxygen can be introduced together with an inert gas, and the pressure can be reduced simultaneously, thereby simultaneously forming Si/C/M under vacuum.
C.CVD法C. CVD method
CVD法使用的原料气体中,作为元素Si源为SiH4、SiCl4等;作为元素C源为CH4、C2H6、C3H8等。Among the raw material gases used in the CVD method, SiH 4 , SiCl 4 , etc. are used as the elemental Si source; CH 4 , C 2 H 6 , C 3 H 8 , etc. are used as the elemental C source.
<制造法3><Manufacturing method 3>
下面对通式SiZxMy中元素Z为C、元素M为O时的制造方法进行说明。Next, the production method when the element Z is C and the element M is O in the general formula SiZ x M y will be described.
蒸镀源、溅射源或喷镀源使用下述物质中的任一种:Evaporation sources, sputter sources or sputter sources use any of the following:
(I)Si和C的组合物;(1) the composition of Si and C;
(II)Si和C的混合物;(II) a mixture of Si and C;
(III)Si和C各自的单质,(III) the respective simple substances of Si and C,
或者or
(IV)含有Si和C的气体,(IV) gases containing Si and C,
在成膜气体中(在真空中成膜时为残留气体中)的氧浓度为0.0001~0.125%的氛围气下,利用蒸镀法和/或溅射法、以及喷镀法同时将Si和C在上述集电体基板上形成1~30μm的厚度、优选为活性物质薄膜的膜厚项中记载的厚度的膜。In an atmosphere where the oxygen concentration in the film-forming gas (in the residual gas during film formation in vacuum) is 0.0001 to 0.125%, Si and C are deposited simultaneously by vapor deposition and/or sputtering and sputtering A film having a thickness of 1 to 30 μm, preferably the thickness described in the section of film thickness of the active material thin film, is formed on the above-mentioned current collector substrate.
(原料)(raw material)
作为原料的蒸镀源、溅射源或喷镀源的Si原料,例如可以使用结晶性Si、无定形Si等。作为C原料,例如可以使用天然石墨、人造石墨等碳材料。作为成膜气体中的氧,将氧气等含有O元素的气体单独使用或与惰性气体组合使用。这些Si、C原料的形态,例如可制成粉末状、颗粒状、小球状、块状、板状等使用。此外,氧气作为原料气体在Si、C成膜中共存在制造上是优选的。As a Si raw material of a vapor deposition source, a sputtering source, or a thermal spraying source of a raw material, for example, crystalline Si, amorphous Si, or the like can be used. As the C raw material, for example, carbon materials such as natural graphite and artificial graphite can be used. As oxygen in the film-forming gas, a gas containing an O element such as oxygen is used alone or in combination with an inert gas. The forms of these Si and C raw materials can be used in powder form, granule form, pellet form, block form, plate form, etc., for example. In addition, oxygen is preferable as a raw material gas in the coexistence production of Si and C film formation.
(成膜法)(film forming method)
使用与上述制造法1同样的成膜法。The same film-forming method as that of Production Method 1 above was used.
(成膜时的氧浓度)(Oxygen concentration during film formation)
蒸镀和/或溅射、以及喷镀时在成膜气体中(真空中成膜时为残留气体中)的氧浓度通常为0.0001%以上,且通常为0.125%以下,优选为0.100%以下,更优选为0.020%以下。如果成膜气体中含有的氧浓度超过该范围,则Si/C/O薄膜中的元素O量增多,与非水电解液的反应性增大,有时导致充放电效率的降低。如果氧浓度过少,则有时不能形成Si/C/O薄膜。The oxygen concentration in the film-forming gas (residual gas during film-forming in vacuum) during vapor deposition and/or sputtering and sputtering is usually 0.0001% or more, and usually 0.125% or less, preferably 0.100% or less, More preferably, it is 0.020% or less. If the oxygen concentration contained in the film-forming gas exceeds this range, the amount of element O in the Si/C/O thin film will increase, and the reactivity with the non-aqueous electrolyte will increase, which may lead to a decrease in charge and discharge efficiency. If the oxygen concentration is too low, the Si/C/O thin film may not be formed.
而且,作为成膜气体中的氧浓度,例如,可通过使用四极滤质器(四極子フイルタ)分析成膜气体的质谱而得到。另外,使用共存有氧气的氩气作为成膜气体时,可通过氧分析计测定该氩气而求得。Furthermore, the oxygen concentration in the film-forming gas can be obtained, for example, by analyzing the mass spectrum of the film-forming gas using a quadrupole filter. In addition, when argon gas coexisting with oxygen gas is used as the film-forming gas, it can be obtained by measuring the argon gas with an oxygen analyzer.
<制造法4><Manufacturing method 4>
下面对通式SiZxMy中元素Z为N且y=0或y≒0时的制造方法进行说明。The production method when the element Z in the general formula SiZ x M y is N and y=0 or y≒0 will be described below.
蒸镀源、溅射源或喷镀源使用下述物质中的任一种:Evaporation sources, sputter sources or sputter sources use any of the following:
(I)Si单质;(1) Si simple substance;
(II)含有Si的组合物;(II) compositions containing Si;
(III)含有Si的混合物,(III) mixtures containing Si,
或or
(IV)含有Si的气体,(IV) Si-containing gas,
在成膜气体中(在真空中成膜时为残留气体中)的氮浓度为1~22%的氛围气下,利用蒸镀法和/或溅射法、以及喷镀法同时将Si和N在上述集电体基板上形成1~30μm的厚度、优选为活性物质薄膜的膜厚项中记载的厚度的膜。In the atmosphere where the nitrogen concentration in the film-forming gas (in the residual gas when forming a film in vacuum) is 1 to 22%, Si and N are simultaneously deposited by evaporation and/or sputtering and sputtering. A film having a thickness of 1 to 30 μm, preferably the thickness described in the section of film thickness of the active material thin film, is formed on the above-mentioned current collector substrate.
(原料)(raw material)
作为原料的蒸镀源、溅射源或喷镀源的Si单质原料,例如可以使用结晶性Si、无定形Si等。作为成膜气体中的N,将氮气等含N元素的气体单独使用或与惰性气体组合使用。这些Si等的形态,例如可制成粉末状、颗粒状、小球状、块状、板状等使用。另外,氮气作为原料气体在Si成膜中共存在制造上是优选的。As a raw material of Si simple substance as a vapor deposition source, a sputtering source, or a thermal spraying source of a raw material, for example, crystalline Si, amorphous Si, or the like can be used. As N in the film-forming gas, a gas containing an N element such as nitrogen is used alone or in combination with an inert gas. The form of these Si etc. can be used, for example in a powder form, a granular form, a pellet form, a lump form, a plate form, etc. In addition, nitrogen gas is preferable as a raw material gas in the coexistence production of Si film formation.
(成膜法)(film forming method)
使用与上述制造法1同样的成膜法。The same film-forming method as that of Production Method 1 above was used.
(成膜时的氮浓度)(Nitrogen concentration during film formation)
蒸镀和/或溅射、以及喷镀时在成膜气体中(真空中成膜时为残留气体中)的氮浓度通常为1%以上,且通常为22%以下,优选为15%以下,更优选为10%以下。如果成膜气体中含有的氮浓度超过该范围,则SiNx薄膜中的元素N量增多,产生不参与充放电的氮化硅,有时导致放电容量的降低。如果氮浓度过少,则有时不能形成含有N的SiNx薄膜且导致循环特性的降低。另外,作为成膜气体中的氮浓度,例如,可通过使用四极滤质器分析成膜气体的质谱而得到。The nitrogen concentration in the film-forming gas (residual gas during film-forming in vacuum) during vapor deposition and/or sputtering and sputtering is usually 1% or more, and usually 22% or less, preferably 15% or less, More preferably, it is 10% or less. If the nitrogen concentration contained in the film-forming gas exceeds this range, the amount of element N in the SiNx thin film will increase, and silicon nitride that does not participate in charge and discharge will be generated, resulting in a decrease in discharge capacity in some cases. If the nitrogen concentration is too low, the SiN x thin film containing N may not be formed, resulting in degradation of cycle characteristics. In addition, the nitrogen concentration in the film-forming gas can be obtained, for example, by analyzing the mass spectrum of the film-forming gas using a quadrupole mass filter.
2.元素Z外在锂吸留物质(F)的制法2. Preparation method of element Z external lithium occlusion substance (F)
<制造法5><Manufacturing method 5>
将锂吸留金属(A’)和/或锂吸留合金(B’)与碳质物质(E’)的组成项中所述的可碳化的有机物混合,并将有机物加热、分解,经过固相和/或液相和/或气相而进行碳化,形成碳质物质(E’),得到复合物。再对该粒子进行粉碎、分级,使体积基准平均粒径为适当的值。Mix the lithium storage metal (A') and/or the lithium storage alloy (B') with the carbonizable organic matter described in the composition item of the carbonaceous substance (E'), heat and decompose the organic matter, and solidify phase and/or liquid phase and/or gas phase to carbonize to form a carbonaceous substance (E') to obtain a composite. The particles are then pulverized and classified so that the volume-based average particle diameter becomes an appropriate value.
(原料)(raw material)
作为原料的锂吸留金属(A’)和/或锂吸留合金(B’)的体积基准平均粒径通常为100μm以下,优选为10μm以下,更优选为1μm以下,其下限为1nm以上的范围。如果超过上限,则难以缓和充电时的膨胀,循环保持率有时降低。另外,如果低于下限,则难以进行粉碎,有时导致时间上、经济上的损失。此外,作为碳质物质(E’)的原料如上所述,但是优选为在碳化时经过液相的物质。The volume-based average particle diameter of the lithium storage metal (A') and/or lithium storage alloy (B') as a raw material is usually 100 μm or less, preferably 10 μm or less, more preferably 1 μm or less, and the lower limit is 1 nm or more. scope. If the upper limit is exceeded, it will be difficult to relax swelling during charging, and the cycle retention may decrease. Moreover, if it is less than the lower limit, pulverization becomes difficult, and time and economic loss may be caused. In addition, although the raw material of the carbonaceous substance (E') is as described above, it is preferably a substance that passes through a liquid phase during carbonization.
<制造法6><Manufacturing method 6>
将锂吸留金属(A’)和/或锂吸留合金(B’)与石墨质(G)混合,再混合碳质物质(E’)的组成项中所述的可以碳化的有机物,并将有机物加热、分解,以液相进行碳化,形成碳质物质(E’),得到复合物。再对该粒子进行粉碎、分级,使体积基准平均粒径为适当的值。mixing the lithium storage metal (A') and/or the lithium storage alloy (B') with the graphite (G), and then mixing the carbonizable organic matter described in the composition item of the carbonaceous material (E'), and The organic matter is heated, decomposed, and carbonized in the liquid phase to form a carbonaceous substance (E') to obtain a composite. The particles are then pulverized and classified so that the volume-based average particle diameter becomes an appropriate value.
(原料)(raw material)
原料与<制造法5>相同。The raw materials are the same as in <Manufacturing method 5>.
<制造法7><Manufacturing method 7>
将锂吸留金属(A’)和/或锂吸留合金(B’)与石墨质(G)混合,再混合碳质物质(E’)的组成项中所述的可以碳化的有机物,并将有机物加热、分解,以固相进行碳化,经过相而形成碳质物质(E’),得到复合物。进一步对该粒子进行粉碎、分级,使体积基准平均粒径为适当的值。mixing the lithium storage metal (A') and/or the lithium storage alloy (B') with the graphite (G), and then mixing the carbonizable organic matter described in the composition item of the carbonaceous material (E'), and The organic matter is heated and decomposed, and carbonized in the solid phase, and the carbonaceous substance (E') is formed through the phase, and a composite is obtained. The particles are further pulverized and classified so that the volume-based average particle diameter becomes an appropriate value.
(原料)(raw material)
原料与<制造法5>相同。The raw materials are the same as in <Manufacturing method 5>.
<制造法8><Manufacturing method 8>
将锂吸留金属(A’)和/或锂吸留合金(B’)与石墨质(G)混合,再混合碳质物质(E’)的组成项中所述的可以碳化的有机物,并将有机物加热、分解,以气相进行碳化,经过相而形成碳质物质(E’),得到复合物。进一步对该粒子进行粉碎、分级,使体积基准平均粒径为适当的值。mixing the lithium storage metal (A') and/or the lithium storage alloy (B') with the graphite (G), and then mixing the carbonizable organic matter described in the composition item of the carbonaceous material (E'), and Organic matter is heated and decomposed, carbonized in the gas phase, and carbonaceous substance (E') is formed through the gas phase to obtain a composite. The particles are further pulverized and classified so that the volume-based average particle diameter becomes an appropriate value.
(原料)(raw material)
原料与<制造法5>相同。The raw materials are the same as in <Manufacturing method 5>.
[负极[7]的粉末状活性物质的电极化][Electric Polarization of Powdered Active Material of Negative Electrode [7]]
负极的制造可以按照常规方法进行,例如,如上所述,可以通过向负极活性物质中加入粘合剂、溶剂,根据需要加入增稠剂、导电材料、填充材料等而制成浆料,将其涂布在集电体上并进行干燥后,进行压制而形成。电池的电解液注入步骤之前的阶段中的每一面的负极活性物质层的厚度通常为5μm以上、优选为10μm以上、更优选为15μm以上,其上限为150μm以下、优选为120μm以下、更优选为100μm以下。如果超过该范围,由于非水电解液难以浸透至集电体界面附近,因此高电流密度充放电特性有时降低。此外,如果低于该范围,则集电体对于负极活性物质的体积比增加,电池的容量有时减少。此外,可以将负极活性物质辊成型而形成片电极,或者通过压缩成型而形成颗粒电极。The manufacture of negative pole can be carried out according to conventional method, for example, as mentioned above, can be made into slurry by adding binding agent, solvent in negative pole active material, adding thickener, conductive material, filling material etc. as required, its It is formed by pressing after coating and drying on the current collector. The thickness of the negative electrode active material layer on each side in the stage before the electrolyte solution injection step of the battery is usually 5 μm or more, preferably 10 μm or more, more preferably 15 μm or more, and its upper limit is 150 μm or less, preferably 120 μm or less, more preferably Below 100μm. If it exceeds this range, since the nonaqueous electrolyte solution becomes difficult to permeate to the vicinity of the current collector interface, the high current density charge and discharge characteristics may be deteriorated. In addition, if it is less than this range, the volume ratio of the current collector to the negative electrode active material increases, and the capacity of the battery may decrease. In addition, the negative electrode active material may be formed into a sheet electrode by roll molding, or a granular electrode may be formed by compression molding.
可以使用的粘合剂、增稠剂等与上述相同。Usable binders, thickeners, and the like are the same as above.
<负极[8]><negative pole[8]>
下面对本发明的锂二次电池中使用的负极[8]进行说明,所述负极[8]含有性质不同的2种以上负极活性物质作为负极活性物质。Next, the negative electrode [8] containing two or more negative electrode active materials having different properties as the negative electrode active material used in the lithium secondary battery of the present invention will be described.
[负极[8]的负极活性物质][Negative electrode active material of negative electrode [8]]
下面对负极[8]中使用的负极活性物质进行说明。Next, the negative electrode active material used in the negative electrode [8] will be described.
本发明的锂二次电池的负极[8]中使用的负极活性物质的特征在于,含有性质不同的2种以上的负极活性物质。The negative electrode active material used in the negative electrode [8] of the lithium secondary battery of the present invention is characterized by containing two or more negative electrode active materials having different properties.
其中所述的“性质不同”不仅是指以X射线衍射参数、中值粒径、长径比、BET比表面积、取向比、拉曼R值、振实密度、真密度、微孔分布、圆形度、灰分量等为代表的粉末形状和粉末物性不同,还包含“含有2种以上结晶性不同的碳质物质的复合碳质物质”、“含有2种以上取向性不同的碳质物质的异取向性碳复合物”等材料的构成不同,或“对负极活性物质实施热处理”、“对负极活性物质实施力学能量处理”等加工处理不同的情况。The "different properties" mentioned here not only refer to X-ray diffraction parameters, median particle size, aspect ratio, BET specific surface area, orientation ratio, Raman R value, tap density, true density, pore distribution, circular Shape, ash content, etc. are different in powder shape and powder physical properties, and also include "composite carbonaceous substances containing two or more carbonaceous substances with different crystallinity", "composite carbonaceous substances containing two or more carbonaceous substances with different orientations" The composition of materials such as "hetero-oriented carbon composites" is different, or the processing treatments such as "heat treatment of the negative electrode active material" and "mechanical energy treatment of the negative electrode active material" are different.
[形状、物性等的不同][differences such as shape, physical properties]
其中,本发明的锂二次电池的负极[8]中使用的负极活性物质通过含有体积基准平均粒径(中值粒径)不同的2种以上的负极活性物质,可以维持低温输出功率,同时可以提高循环特性。作为体积基准平均粒径(中值粒径)之差,通常存在1μm以上、优选存在2μm以上、更优选存在5μm以上的差。作为上限,通常为30μm以下、优选为25μm以下。若超过该范围,则中值粒径大的粒径有过大的趋势,因此在电极制造时有可能产生涂布面的拉丝等问题。另一方面,若低于该范围,则有可能难以表现出混合2种负极活性物质的效果。Wherein, the negative electrode active material used in the negative electrode [8] of the lithium secondary battery of the present invention can maintain the low temperature output power by containing more than two kinds of negative electrode active materials with different volume-based average particle diameters (median particle diameters), and at the same time Cycle characteristics can be improved. The difference in volume-based average particle diameter (median particle diameter) is usually 1 μm or more, preferably 2 μm or more, more preferably 5 μm or more. The upper limit is usually 30 μm or less, preferably 25 μm or less. If it exceeds this range, the particle diameter with a large median diameter tends to be too large, and therefore problems such as stringing of the coated surface may occur during electrode production. On the other hand, if it is less than this range, it may be difficult to express the effect of mixing two kinds of negative electrode active materials.
此外,对于本发明的锂二次电池的负极[8]中使用的负极活性物质,由于与上述相同的理由,体积基准粒度分布不均的材料也可以表现出良好的特性。所谓“体积基准粒度分布不均”,是指横轴为对数刻度时的体积基准粒度分布,以体积基准平均粒径(中值粒径)为中心时,不形成左右对称,作为非左右对称的程度,以下述式(1)表示的Z值通常为0.3μm以上、优选为0.5μm以上、更优选为1μm以上。若Z低于该值,则有可能难以得到通过粒度分布不均所带来的循环特性提高的效果。In addition, for the negative electrode active material used in the negative electrode [8] of the lithium secondary battery of the present invention, for the same reason as above, a material with a non-uniform volume-based particle size distribution can also exhibit good characteristics. The so-called "uneven volume-based particle size distribution" refers to the volume-based particle size distribution when the horizontal axis is a logarithmic scale. When the volume-based average particle size (median particle size) is the center, it does not form left-right symmetry. The Z value represented by the following formula (1) is usually 0.3 μm or more, preferably 0.5 μm or more, and more preferably 1 μm or more. If Z is lower than this value, it may be difficult to obtain the effect of improving cycle characteristics due to uneven particle size distribution.
Z=|(模径)-(中值粒径)| (1)Z=|(mode diameter)-(median particle diameter)| (1)
式(1)中,模径和中值粒径的单位都为“μm”,“||”表示绝对值。In formula (1), the unit of mode diameter and median particle diameter is "μm", and "||" represents an absolute value.
在本发明中,体积基准平均粒径(中值粒径)和模径定义为如下的值:使负极活性物质分散在作为表面活性剂的聚氧乙烯(20)山梨糖醇酐单月桂酸酯的0.2质量%水溶液(约1mL)中,使用激光衍射式粒度分布计(堀场制作所公司制造的LA-700)而测定的值。“中值粒径”通常也称为d50,指的是在体积基准下将粉末由某粒径分为两部分时较大侧和较小侧等量时的该粒径,“模径”指的是在体积基准的粒度分布中,表示分布的极大值的粒径,任意一个都只是堀场制作所公司制造的LA-700中称作“中值粒径”、“模径”的值,分别表示在装置上。In the present invention, the volume-based average particle diameter (median particle diameter) and the modulus diameter are defined as the following values: the negative electrode active material is dispersed in polyoxyethylene (20) sorbitan monolaurate as a surfactant The value measured using the laser diffraction particle size distribution meter (LA-700 by Horiba Co., Ltd.) in 0.2 mass % aqueous solution (about 1 mL). "Median particle size" is usually also called d 50 , which refers to the particle size when the powder is divided into two parts by a certain particle size under the volume basis, when the larger side and the smaller side are equal, and the "mode size" Refers to the particle size that represents the maximum value of the distribution in the volume-based particle size distribution, and any one of them is called "median particle size" and "mode size" in LA-700 manufactured by Horiba Co., Ltd. value, respectively, on the device.
此外,通过使负极活性物质的至少一种使用中值粒径为10μm以下的负极活性物质,可以维持低温输出功率,同时得到循环特性的提高效果,从这点上看是优选的。特别优选为8μm以下。中值粒径为10μm以下的负极活性物质相对于全部负极活性物质,特别优选在0.5~10质量%的范围内使用。In addition, it is preferable to use a negative electrode active material having a median particle size of 10 μm or less as at least one of the negative electrode active materials, since it is possible to maintain the low-temperature output and obtain an effect of improving cycle characteristics. Particularly preferably, it is 8 μm or less. The negative electrode active material having a median diameter of 10 μm or less is particularly preferably used in an amount of 0.5 to 10% by mass relative to the entire negative electrode active material.
另外,本发明的锂二次电池的负极[8]中使用的负极活性物质,通过含有采用氩离子激光拉曼光谱法测定的拉曼R值不同的2种以上的负极活性物质,可以维持循环特性,同时提高低温输出功率。作为拉曼R值之差,通常为0.1以上、优选为0.2以上、更优选为0.3以上,作为上限,通常为1.4以下、优选为1.3以下、更优选为1.2以下的范围。如果低于该范围,则有时难以得到由于拉曼R值的不同而带来的效果。另一方面,如果超过该范围,则由于拉曼R值高的部分,不可逆容量有时增加。In addition, the negative electrode active material used in the negative electrode [8] of the lithium secondary battery of the present invention can maintain cycle by containing two or more negative electrode active materials with different Raman R values measured by argon ion laser Raman spectroscopy. characteristics, while improving low temperature output power. The difference in Raman R value is usually 0.1 or more, preferably 0.2 or more, more preferably 0.3 or more, and the upper limit is usually 1.4 or less, preferably 1.3 or less, more preferably 1.2 or less. If it is less than this range, it may be difficult to obtain the effect due to the difference in Raman R value. On the other hand, if it exceeds this range, the irreversible capacity may increase due to a portion with a high Raman R value.
在本发明中,拉曼光谱的测定如下进行:使用拉曼分光器(例如,日本分光公司制造的拉曼分光器),使试样自然落入到测定容器内,进行试样填充,测定过程是,对容器内的试样表面照射氩离子激光,同时使该容器在与激光垂直的面内旋转。对于得到的拉曼光谱,测定1580cm-1的峰PA的强度IA和1360cm-1的峰PB的强度IB,计算其强度比R(R=IB/IA),将其定义为石墨质碳粒子的拉曼R值。另外,测定得到的拉曼光谱在1580cm-1的峰PA的半值宽度,将其定义为石墨质碳粒子的拉曼半值宽度。In the present invention, the measurement of the Raman spectrum is carried out as follows: using a Raman spectrometer (for example, a Raman spectrometer manufactured by JASCO Corporation), the sample is naturally dropped into the measurement container, the sample is filled, and the measurement process Yes, the sample surface in the container is irradiated with an argon ion laser while rotating the container in a plane perpendicular to the laser beam. For the obtained Raman spectrum, measure the intensity I A of the peak PA at 1580 cm -1 and the intensity I B of the peak P B at 1360 cm -1 , calculate the intensity ratio R (R= IB / IA ), and define it as is the Raman R value of the graphitic carbon particle. In addition, the half-value width of the peak PA at 1580 cm −1 in the obtained Raman spectrum was measured, and this was defined as the Raman half-value width of the graphitic carbon particles.
另外,这里的拉曼测定条件如下。In addition, the Raman measurement conditions here are as follows.
·氩离子激光波长:514.5nmArgon ion laser wavelength: 514.5nm
·试样上的激光功率:15~25mW·Laser power on the sample: 15~25mW
·分辨率:10~20cm-1 ·Resolution: 10~20cm -1
·测定范围:1100cm-1~1730cm-1 · Measuring range: 1100cm -1 ~ 1730cm -1
·拉曼R值、半值宽度分析:本底处理Raman R value, half-value width analysis: background processing
·光滑处理:单纯平均、卷积5点·Smooth processing: simple average, convolution 5 points
此外,作为本发明的锂二次电池的负极[8]中使用的负极活性物质所使用的负极活性物质,其1580cm-1的拉曼半值宽度没有特别限定,但是通常为10cm-1以上、优选为15cm-1以上,另外,其上限通常为150cm-1以下、优选为140cm-1以下的范围。如果拉曼半值宽度低于该范围,则粒子表面的结晶性过高,低温输出功率有时降低。另一方面,如果超过该范围,则由于粒子表面的结晶性降低,不可逆容量有时增加。In addition, the negative electrode active material used as the negative electrode active material used in the negative electrode [8] of the lithium secondary battery of the present invention has a Raman half-value width of 1580 cm It is preferably 15 cm -1 or more, and the upper limit thereof is usually 150 cm -1 or less, preferably 140 cm -1 or less. If the Raman half-value width is below this range, the crystallinity of the particle surface is too high, and the low-temperature output may decrease. On the other hand, if it exceeds this range, the irreversible capacity may increase due to the decrease in the crystallinity of the particle surface.
此外,作为本发明的锂二次电池的负极[8]中使用的负极活性物质所使用的负极活性物质,即使含有结晶性不同的2种以上的负极活性物质,也可以维持循环特性并提高低温输出功率。所谓结晶性,在这里是指碳的六角网面的叠层体的重复结构的厚度、间距等叠层结构。表示结晶性不同的具体的物性值没有特别限定,例如有面间距、微晶尺寸等,优选它们在本发明的锂二次电池中使用的2种以上的负极活性物质中不同。如果结晶性之差过小,则有时难以得到通过混合带来的效果。In addition, even if the negative electrode active material used as the negative electrode active material used in the negative electrode [8] of the lithium secondary battery of the present invention contains two or more negative electrode active materials with different crystallinity, the cycle characteristics can be maintained and the low temperature can be improved. Output Power. The term "crystallinity" here refers to a laminated structure such as the thickness and pitch of the repeating structure of the laminated body of carbon hexagonal network planes. The specific physical property values indicating the difference in crystallinity are not particularly limited, for example, interplanar spacing, crystallite size, etc., and these are preferably different among the two or more negative electrode active materials used in the lithium secondary battery of the present invention. When the difference in crystallinity is too small, it may be difficult to obtain the effect by mixing.
本发明的锂二次电池的负极[8]中使用的负极活性物质,通过含有采用广角X射线衍射法测定的(002)面的面间距(d002)不同的2种以上负极活性物质,可以维持循环特性并提高低温输出功率。作为面间距(d002)之差,通常为0.0005nm以上、优选为0.001nm以上、更优选为0.003nm以上、进一步优选为0.004nm以上,其上限通常为0.05nm以下、优选为0.04nm以下、更优选为0.03nm以下、进一步优选为0.02nm以下的范围。如果低于该范围,则有时难以得到由于结晶性不同而带来的效果。另一方面,如果超过该范围,则由于结晶性低的部分,不可逆容量有时增加。本发明中所说的采用广角X射线衍射法测定的(002)面的面间距(d002)是指通过采用学振法进行的X射线衍射求得的晶格面(002面)的d值(层间距离)。The negative electrode active material used in the negative electrode [8] of the lithium secondary battery of the present invention can maintain Cycle characteristics and improve low temperature output power. The difference between interplanar distances (d002) is usually at least 0.0005 nm, preferably at least 0.001 nm, more preferably at least 0.003 nm, and even more preferably at least 0.004 nm, and the upper limit thereof is usually at most 0.05 nm, preferably at most 0.04 nm, and more The range is preferably 0.03 nm or less, more preferably 0.02 nm or less. If it is less than this range, it may be difficult to obtain the effect due to the difference in crystallinity. On the other hand, if it exceeds this range, the irreversible capacity may increase due to the portion with low crystallinity. The interplanar spacing (d002) of the (002) plane measured by the wide-angle X-ray diffraction method mentioned in the present invention refers to the d value ( layer distance).
此外,本发明的锂二次电池的负极[8]中使用的负极活性物质,通过含有采用学振法以X射线衍射求得的微晶尺寸(Lc)不同的2种以上负极活性物质,可以维持循环特性并提高低温输出功率。通过学振法以X射线衍射求得的微晶尺寸(Lc)之差通常为1nm以上、优选为10nm以上、更优选为50nm以上。如果低于该范围,则有时难以得到由于微晶尺寸不同所带来的效果。In addition, the negative electrode active material used in the negative electrode [8] of the lithium secondary battery of the present invention can be obtained by containing two or more negative electrode active materials with different crystallite sizes (Lc) obtained by X-ray diffraction using the Gakushin method. Maintain cycle characteristics and increase low temperature output. The difference in crystallite size (Lc) obtained by X-ray diffraction by the Gakushin method is usually 1 nm or more, preferably 10 nm or more, more preferably 50 nm or more. If it is less than this range, it may be difficult to obtain the effect due to the difference in crystallite size.
此外,本发明的锂二次电池的负极[8]中使用的负极活性物质,通过含有真密度不同的2种以上负极活性物质,可以维持循环特性并提高低温输出功率。作为真密度之差,通常为0.03g/cm3以上、优选为0.05g/cm3以上、更优选为0.1g/cm3以上、进一步优选为0.2g/cm3以上,其上限通常为0.7g/cm3以下、优选为0.5g/cm3以下、更优选为0.4g/cm3以下的范围。如果低于该范围,则有时难以得到由于真密度不同而带来的效果。另一方面,如果超过该范围,则由于真密度低的部分,不可逆容量有时增加。In addition, the negative electrode active material used in the negative electrode [8] of the lithium secondary battery of the present invention can maintain cycle characteristics and improve low-temperature output power by containing two or more negative electrode active materials with different true densities. The difference in true density is usually 0.03 g/cm 3 or more, preferably 0.05 g/cm 3 or more, more preferably 0.1 g/cm 3 or more, still more preferably 0.2 g/cm 3 or more, and the upper limit is usually 0.7 g /cm 3 or less, preferably 0.5 g/cm 3 or less, more preferably 0.4 g/cm 3 or less. If it is less than this range, it may be difficult to obtain the effect due to the difference in true density. On the other hand, if this range is exceeded, the irreversible capacity may increase due to the low true density portion.
本发明中所称的真密度定义为通过使用丁醇的液相置换法(比重计法)测定的值。The true density referred to in the present invention is defined as a value measured by a liquid-phase displacement method (hydrometer method) using butanol.
此外,本发明中使用的负极活性物质通过含有圆形度不同的2种以上负极活性物质,也可以维持低温输出功率并提高循环特性。作为圆形度之差,通常为0.01以上、优选为0.02以上、更优选为0.03以上,其上限通常为0.3以下、优选为0.2以下、更优选为0.1以下的范围。如果低于该范围,则有时难以得到由于圆形度不同所带来的效果。另一方面,如果超过该范围,则由于圆形度低的部分,电极化时,有时产生拉丝等问题。In addition, when the negative electrode active material used in the present invention contains two or more negative electrode active materials having different circularities, it is possible to maintain low-temperature output and improve cycle characteristics. The difference in circularity is usually 0.01 or more, preferably 0.02 or more, more preferably 0.03 or more, and the upper limit is usually 0.3 or less, preferably 0.2 or less, more preferably 0.1 or less. If it is less than this range, it may be difficult to obtain the effect due to the difference in circularity. On the other hand, if it exceeds this range, problems such as stringing may occur at the time of electric polarization due to the portion with low circularity.
本发明中所说的圆形度用下式定义。The circularity referred to in the present invention is defined by the following formula.
圆形度=(具有与粒子投影形状相同面积的等效圆的周长)/(粒子投影形状的实际周长)Circularity = (perimeter of an equivalent circle having the same area as the particle projected shape)/(actual perimeter of the particle projected shape)
作为圆形度的值使用如下测定的值:使用流动式粒子图像分析装置(例如,Sysmex Industrial公司制造的FPIA),将约0.2g试样分散于作为表面活性剂的聚氧乙烯(20)山梨糖醇酐单月桂酸酯的0.2质量%水溶液(约50mL)中,以60W的输出功率照射1分钟28kHz的超声波后,指定0.6~400μm为检测范围,对粒径3~40μm范围的粒子进行测定。As the value of circularity, the value measured as follows is used: using a flow type particle image analyzer (for example, FPIA manufactured by Sysmex Industrial Co., Ltd.), about 0.2 g of a sample is dispersed in polyoxyethylene (20) sorbitol as a surfactant. After irradiating a 0.2% by mass aqueous solution (approximately 50mL) of sugar-alcohol monolaurate with an output of 60W for 1 minute at 28kHz ultrasonic waves, specify a detection range of 0.6-400μm, and measure particles with a particle diameter of 3-40μm .
此外,本发明的锂二次电池的负极[8]中使用的负极活性物质,通过含有振实密度不同的2种以上负极活性物质,可以维持低温输出功率并提高循环特性。作为振实密度之差,通常为0.1g/cm3以上、优选为0.2g/cm3以上、更优选为0.3g/cm3以上。如果低于该范围,则有时难以得到通过混合振实密度不同的材料而带来的效果。In addition, the negative electrode active material used in the negative electrode [8] of the lithium secondary battery of the present invention can maintain low-temperature output and improve cycle characteristics by containing two or more negative electrode active materials with different tap densities. The tap density difference is usually at least 0.1 g/cm 3 , preferably at least 0.2 g/cm 3 , and more preferably at least 0.3 g/cm 3 . If it is less than this range, it may be difficult to obtain the effect of mixing materials with different tap densities.
在本发明中,振实密度如下定义:使试样通过孔径300μm的筛子,使试样落入20cm3的振实容器中,到试样充满容器的上端面后,使用粉末密度测定器(例如,seishin企业公司制造的Tap densor),进行1000次冲程长度10mm的振动,由此时的体积和试料的重量计算密度,将该值作为振实密度。In the present invention, the tap density is defined as follows: the sample is passed through a sieve with an aperture of 300 μm, the sample is dropped into a 20 cm container for vibration, and after the sample is filled with the upper end of the container, a powder density measuring device (such as , Tap densor manufactured by Seishin Enterprise Co., Ltd.), vibration with a stroke length of 10 mm was performed 1000 times, and the density was calculated from the volume at this time and the weight of the sample, and this value was taken as the tap density.
此外,本发明的锂二次电池的负极[8]中使用的负极活性物质,通过含有BET比表面积不同的2种以上的负极活性物质,可以维持循环特性并提高低温输出功率。作为BET比表面积之差,通常为0.1m2/g以上、优选为0.5m2/g以上、更优选为1m2/g以上,其上限通常为20m2/g以下、优选为15m2/g以下、更优选为12m2/g以下的范围。如果低于该范围,则有时难以得到通过混合BET比表面积不同的材料所带来的效果。另一方面,如果超过该范围,则由于BET比表面积大的部分,不可逆容量有时增加。In addition, the negative electrode active material used in the negative electrode [8] of the lithium secondary battery of the present invention contains two or more negative electrode active materials with different BET specific surface areas, so that the cycle characteristics can be maintained and the low-temperature output can be improved. The difference in BET specific surface area is usually 0.1 m 2 /g or more, preferably 0.5 m 2 /g or more, more preferably 1 m 2 /g or more, and the upper limit is usually 20 m 2 /g or less, preferably 15 m 2 /g It is not more than 12 m 2 /g, and more preferably not more than 12 m 2 /g. If it is less than this range, it may be difficult to obtain the effect by mixing materials with different BET specific surface areas. On the other hand, if this range is exceeded, the irreversible capacity may increase due to the portion with a large BET specific surface area.
在本发明中,BET比表面积定义为如下的值:使用表面积计(大仓理研制造的全自动表面积测定装置),在氮流通下在350℃对试料进行15分钟预干燥,然后使用正确调整为氮相对于大气压的相对压力值为0.3的氮氦混合气体,通过采用气体流动法进行的氮吸附BET 1点法而测定的值。In the present invention, the BET specific surface area is defined as a value as follows: Using a surface area meter (automatic surface area measuring device manufactured by Okura Riken), the sample is pre-dried at 350° C. for 15 minutes under nitrogen flow, and then adjusted using It is a value measured by the nitrogen adsorption BET 1-point method using a gas flow method for a nitrogen-helium mixed gas whose relative pressure value of nitrogen to atmospheric pressure is 0.3.
作为本发明的锂二次电池的负极[8]中使用的2种以上不同的负极活性物质的混合比率,一种负极活性物质所占的比率相对于总量通常为0.1质量%以上、优选为1质量%以上、更优选为10质量%以上、进一步优选为20质量%以上,其上限通常为99.9质量%以下、优选为99质量%以下、更优选为90质量%以下、进一步优选为80质量%以下的范围。如果在该范围外,则有时难以得到通过含有不同的2种以上负极活性物质所带来的效果。As the mixing ratio of two or more different negative electrode active materials used in the negative electrode [8] of the lithium secondary battery of the present invention, the ratio of one negative electrode active material to the total amount is usually 0.1% by mass or more, preferably 1% by mass or more, more preferably 10% by mass or more, still more preferably 20% by mass or more, and the upper limit thereof is usually 99.9% by mass or less, preferably 99% by mass or less, more preferably 90% by mass or less, still more preferably 80% by mass % below the range. If it is outside this range, it may be difficult to obtain the effect by containing two or more different negative electrode active materials.
此外,其中不同的2种负极活性物质的至少一种中含有天然石墨和/或天然石墨的加工物,从性价比高方面看是优选的。In addition, natural graphite and/or a processed product of natural graphite are contained in at least one of the two different negative electrode active materials, which is preferable from the standpoint of high cost performance.
天然石墨依照其性状,被分类为鳞片状石墨(Flake Graphite)、鳞状石墨(Crystalline(Vein)Graphite)、土壤石墨(Amorphous Graphite)(参见《粉粒体工艺技术集成》,((株)产业技术中心,昭和49年发行)的石墨一项;以及“HANDBOOK OF CARBON,GRAPHITE,DIAMOND AND FULLERENES”,(NoyesPublications发行))。石墨化度以鳞状石墨最高,为100%,接着是鳞片状石墨,为99.9%,而土壤石墨低至28%。作为天然石墨的鳞片状石墨产自马达加斯加、中国、巴西、乌克兰、加拿大等;鳞状石墨主要产自斯里兰卡。土壤石墨的主产地为朝鲜半岛、中国、墨西哥等。这些天然石墨中,土壤石墨一般粒径小且纯度低。与此相反,鳞片状石墨、鳞状石墨由于具有石墨化度高、杂质量低等优点,可以在本发明中优选使用。According to its properties, natural graphite is classified into flake graphite (Flake Graphite), scaly graphite (Crystalline (Vein) Graphite), soil graphite (Amorphous Graphite) (see "Powder Granular Process Technology Integration", ((Co., Ltd.) Industry Technology Center, Graphite Item, issued in Showa 49); and "HANDBOOK OF CARBON, GRAPHITE, DIAMOND AND FULLERENES", (issued by NoyesPublications)). The degree of graphitization is highest in flake graphite, which is 100%, followed by flake graphite, which is 99.9%, while soil graphite is as low as 28%. Flake graphite as natural graphite is produced in Madagascar, China, Brazil, Ukraine, Canada, etc.; flake graphite is mainly produced in Sri Lanka. The main producing areas of soil graphite are the Korean Peninsula, China, Mexico, etc. Among these natural graphites, soil graphite generally has a small particle size and low purity. On the contrary, flaky graphite and flaky graphite can be preferably used in the present invention because of their advantages of high degree of graphitization and low amount of impurities.
[负极[8]处理的不同][difference in the negative electrode [8] handling]
本发明中使用的负极活性物质通过含有加工处理不同的2种以上的负极活性物质,可以维持循环特性并提高低温输出功率。作为天然石墨的加工方法,可以举出施加热处理的方法、施加力学能量处理的方法等。热处理的一个例子如下所述。The negative electrode active material used in the present invention can maintain cycle characteristics and improve low-temperature output by containing two or more different processed negative electrode active materials. Examples of the processing method of natural graphite include a method of applying heat treatment, a method of applying mechanical energy treatment, and the like. An example of heat treatment is described below.
[[热处理温度]][[heat treatment temperature]]
作为负极活性物质的热处理温度,通常为600℃以上、优选为1200℃以上、更优选为2000℃以上、进一步优选为2500℃以上、特别优选为2800℃以上。上限通常为3200℃以下、优选为3100℃以下。如果温度条件低于该范围,则天然石墨粒子的表面的结晶修复有时不充分。另一方面,如果超过上述范围,则石墨的升华量有时容易增多。本发明中使用的负极活性物质优选含有热处理温度不同的2种以上负极活性物质。The heat treatment temperature of the negative electrode active material is usually 600°C or higher, preferably 1200°C or higher, more preferably 2000°C or higher, further preferably 2500°C or higher, particularly preferably 2800°C or higher. The upper limit is usually 3200°C or lower, preferably 3100°C or lower. If the temperature condition is lower than this range, the crystallization restoration of the surface of the natural graphite particles may be insufficient. On the other hand, when it exceeds the said range, the amount of sublimation of graphite may increase easily. The negative electrode active material used in the present invention preferably contains two or more negative electrode active materials having different heat treatment temperatures.
[[热处理方法]][[Heat treatment method]]
热处理通过经过一次上述温度范围来实现。将温度条件保持在上述范围的保持时间没有特别限定,但通常为长于10秒钟的时间,且为168小时以下。Heat treatment is achieved by passing through the above temperature range once. The holding time for keeping the temperature condition within the above range is not particularly limited, but is usually longer than 10 seconds and not more than 168 hours.
热处理通常在氮气等非活性气体氛围下、或在由原料天然石墨产生的气体形成的非氧化性氛围下进行。但是,对于包埋在煤粉(细的沥青烧结碳)中类型的炉,最初有时混合有大气。这种情况下,可以不必完全非活性气体氛围化。作为热处理中使用的装置,没有特别限定,例如可以使用梭式炉、隧道式炉、电炉、焦炭焙烧炉、回转炉、直接通电炉、阿切孙电炉、电阻加热炉、感应加热炉等。本发明中使用的负极活性物质优选含有热处理方法不同的2种以上的负极活性物质。The heat treatment is usually carried out in an inert gas atmosphere such as nitrogen gas or a non-oxidizing atmosphere formed by gas generated from natural graphite as a raw material. However, for furnaces of the type embedded in pulverized coal (fine pitch-sintered carbon), the atmosphere is sometimes mixed initially. In this case, it is not necessary to completely inert gas atmosphere. The apparatus used for the heat treatment is not particularly limited, and for example, a shuttle furnace, a tunnel furnace, an electric furnace, a coke roasting furnace, a rotary furnace, a direct electric furnace, an Acheson electric furnace, a resistance heating furnace, an induction heating furnace, etc. can be used. The negative electrode active material used in the present invention preferably contains two or more negative electrode active materials having different heat treatment methods.
另外,除了上述各处理,还可以进行分级处理等各种处理。分级处理是为了得到目标粒径、除去粗粉和微粉而进行的处理。作为分级处理中使用的装置,没有特别的限制,例如,在干式筛分的情况下,可以使用旋转式筛、摇动式筛、转动式筛、振动式筛等;在干式气流分级的情况下,可以使用重力式分级机、惯性力式分级机、离心力式分级机(分粒器、旋风分离器)等;在湿式筛分的情况下,可以使用机械式湿式分级机、水力分级机、沉降分级机、离心式湿式分级机等。分级处理可以在热处理前进行,也可以在其他时间,例如热处理后进行。此外,还可以省略分级处理本身。本发明中使用的负极活性物质优选含有分级处理条件不同的2种以上的负极活性物质。In addition, various processing such as classification processing may be performed in addition to the processing described above. Classification treatment is performed to obtain the target particle size and remove coarse powder and fine powder. As the device used in the classification process, there is no particular limitation. For example, in the case of dry sieving, rotary sieves, shaking sieves, rotary sieves, vibrating sieves, etc. can be used; in the case of dry airflow classification In the case of wet sieving, gravity classifiers, inertial force classifiers, centrifugal force classifiers (classifiers, cyclone separators), etc. can be used; in the case of wet sieving, mechanical wet classifiers, hydraulic classifiers, Sedimentation classifier, centrifugal wet classifier, etc. Grading treatment can be carried out before heat treatment, and can also be carried out at other times, such as after heat treatment. Furthermore, the classification process itself can also be omitted. The negative electrode active material used in the present invention preferably contains two or more negative electrode active materials with different classification treatment conditions.
本发明中使用的负极活性物质通过含有后述的力学能量处理不同的2种以上的负极活性物质,可以维持循环特性并提高低温输出功率。力学能量处理的一个例子如下所述。The negative electrode active material used in the present invention can maintain cycle characteristics and improve low-temperature output by containing two or more negative electrode active materials different in mechanical energy treatment described later. An example of mechanical energy processing is described below.
[[力学能量处理]][[Mechanical Energy Processing]]
进行力学能量处理使处理前后的体积平均粒径比为1以下。所谓“处理前后的体积平均粒径比”是处理后的体积平均粒径除以处理前的体积平均粒径而得到的值。在本发明中,为了制造热处理前的原料而进行的力学能量处理优选使处理前后的平均粒径比为1以下。力学能量处理是减小粒子尺寸以使粉末粒子处理前后的平均粒径比为1以下,同时控制粒子形状而进行的处理。在粉碎、分级、混合、造粒、表面改性、反应等可以在粒子设计中有效利用的工学单位操作中,力学能量处理属于粉碎处理。The mechanical energy treatment is performed so that the volume average particle diameter ratio before and after the treatment becomes 1 or less. The "volume average particle diameter ratio before and after treatment" is a value obtained by dividing the volume average particle diameter after treatment by the volume average particle diameter before treatment. In the present invention, it is preferable that the kinetic energy treatment performed to produce the raw material before the heat treatment is such that the average particle diameter ratio before and after the treatment is 1 or less. The mechanical energy treatment is a treatment performed to reduce the particle size so that the average particle diameter ratio before and after the powder particle treatment is 1 or less, and at the same time control the particle shape. Among engineering unit operations that can be effectively utilized in particle design, such as pulverization, classification, mixing, granulation, surface modification, and reaction, mechanical energy processing belongs to pulverization processing.
所谓粉碎,是指对物质施加力,使其大小减少以调节物质的粒径或粒度分布、填充性。粉碎处理依据对物质施加的力的种类、处理形态来进行分类。对物质施加的力大致分为如下4种:(1)敲打力(冲击力)、(2)压碎力(压缩力)、(3)磨碎力(磨碎力)、(4)削刮力(剪切力)。另一方面,处理形态大致分为如下2种:在粒子内部产生龟裂并使之传播的体积粉碎、以及切去粒子表面的表面粉碎。体积粉碎可采用冲击力、压缩力、剪切力来进行;表面粉碎可采用磨碎力、剪切力来进行。粉碎是将对这些物质施加的力的种类和处理形态进行各种组合的处理。其组合可以根据处理目的适当确定。The so-called pulverization refers to applying force to a substance to reduce its size to adjust the particle size, particle size distribution, and filling properties of the substance. Pulverization treatment is classified according to the type of force applied to the substance and the form of treatment. The force exerted on the substance is roughly divided into the following four types: (1) knocking force (impact force), (2) crushing force (compression force), (3) grinding force (grinding force), (4) scraping force force (shear force). On the other hand, processing forms are roughly classified into two types: volume crushing in which cracks are generated inside the particles and propagated, and surface crushing in which the particle surfaces are cut off. Volume crushing can be carried out by impact force, compression force and shear force; surface crushing can be carried out by grinding force and shear force. Pulverization is a process of various combinations of the type of force applied to these substances and the form of treatment. The combination thereof can be appropriately determined according to the purpose of treatment.
粉碎虽然也有使用爆破等化学的反应或体积膨胀来进行的情况,但通常使用粉碎机等机械装置来进行。作为本发明的原料的球形化碳质的制造中使用的粉碎处理优选最终表面处理所占的比例变高的处理,而与有无体积粉碎无关。这是因为,去掉粒子表面粉碎的角以使粒子形状变为圆形是重要的。具体地,可以在进行一定程度的体积粉碎之后再进行表面处理,也可以几乎不进行体积粉碎而只进行表面处理,还可以同时进行体积粉碎和表面处理。优选最后进行表面粉碎,从粒子的表面去掉角的粉碎处理。本发明中使用的负极活性物质,通过含有上述表面处理的程度不同的2种以上的负极活性物质,可以维持循环特性并提高低温输出功率。Pulverization may be performed using a chemical reaction such as blasting or volume expansion, but it is usually performed using a mechanical device such as a pulverizer. The pulverization treatment used in the production of the spheroidized carbon as the raw material of the present invention is preferably a treatment in which the ratio of the final surface treatment increases, regardless of the presence or absence of volume pulverization. This is because it is important to remove the pulverized corners of the particle surface so that the particle shape becomes circular. Specifically, the surface treatment may be performed after a certain volume pulverization is performed, only the surface treatment may be performed without volume pulverization, or the volume pulverization and surface treatment may be performed simultaneously. It is preferable to perform surface crushing at the end to remove corners from the surface of the particles. When the negative electrode active material used in the present invention contains two or more negative electrode active materials having different degrees of surface treatment, cycle characteristics can be maintained and low-temperature output can be improved.
进行力学能量处理的装置从能够进行上述的优选的处理的装置中选择。力学能量处理可以通过使用对上述物质施加的4种力中的一种以上的力来实现,但优选以冲击力为主,并反复对粒子施加包括粒子的相互作用在内的压缩、摩擦、剪切力等机械作用。因此,具体地,优选如下的装置:该装置在箱内部具有设置了多个叶片的转子,并通过该转子高速旋转而对导入到内部的碳材料赋予冲击压缩、摩擦、剪切力等机械的作用,从而边进行体积粉碎边进行表面处理。另外,更优选具有通过使碳质物质循环或对流而反复赋予机械作用的机构的装置。The device for mechanical energy processing is selected from devices capable of performing the above-mentioned preferable processing. Mechanical energy processing can be achieved by using more than one of the four types of force applied to the above-mentioned substances, but it is preferably based on impact force, and repeatedly applies compression, friction, shearing, including particle interaction, to the particles. Mechanical effects such as shear force. Therefore, specifically, a device that has a rotor provided with a plurality of blades inside the tank and that imparts mechanical forces such as impact compression, friction, and shearing force to the carbon material introduced into the inside by rotating the rotor at high speed is preferable. function, thereby carrying out surface treatment while performing volume crushing. In addition, a device having a mechanism for repeatedly imparting a mechanical action by circulating or convecting the carbonaceous material is more preferable.
作为优选的装置,可列举混合系统(奈良机械制作所公司制造)、Kryptron(Earth Technica公司制造)、CF磨(宇部兴产公司制造)、机械熔融系统(hosokawamicron公司制造)等。这些当中,优选奈良机械制作所公司制造的混合系统。使用该装置进行处理时,优选将旋转的转子的圆周速度设定为30~100m/秒,更优选设定为40~100m/秒,进一步优选设定为50~100m/秒。另外,处理可以只是仅仅使碳质物质通过,但优选在装置内循环或滞留30秒钟以上来进行处理,更优选在装置内循环或滞留1分钟以上来进行处理。As a preferable apparatus, a mixing system (manufactured by Nara Machinery Co., Ltd.), Kryptron (manufactured by Earth Technica Co., Ltd.), a CF mill (manufactured by Ube Industries, Ltd.), a mechanical fusion system (manufactured by Hosokawa Micron Co., Ltd.) and the like are exemplified. Among these, the mixing system manufactured by Nara Machinery Manufacturing Co., Ltd. is preferable. When using this apparatus for processing, the peripheral speed of the rotating rotor is preferably set at 30 to 100 m/sec, more preferably at 40 to 100 m/sec, and even more preferably at 50 to 100 m/sec. In addition, the treatment may be performed by merely passing the carbonaceous material, but it is preferably treated by circulating or staying in the device for 30 seconds or more, more preferably by circulating or staying in the device for 1 minute or more.
通过这样进行力学能量处理,碳粒子成为如下的粒子:整体上保持高结晶性,只是粒子的表面附近变得粗糙,倾斜且露出边缘面。这样,锂离子可以出入的面增加,即使在高电流密度下也具有高的容量。By performing the mechanical energy treatment in this way, the carbon particles become particles in which high crystallinity is maintained as a whole, but the vicinity of the surface of the particles becomes rough, and the edge surfaces are inclined and exposed. In this way, the surface where lithium ions can come and go increases, and it has a high capacity even at a high current density.
一般来说,鳞片状、鳞状、板状的碳材料的粒径越小,其填充性越有恶化的倾向。这可认为是因为如下原因:由于粒子通过粉碎而更加无定形化,或者粒子表面生成的“毛边”、“剥离”或“折曲”等突起状物增加,以及在粒子表面以某种程度的强度附着有更微细的无定形粒子等原因,这样,与邻接粒子之间的阻抗变大,使填充性恶化。如果这些粒子的无定形性减少,粒子形状接近于球形,则即使粒径变小,填充性的减小也很少,在理论上,大粒径碳粉末、小粒径碳粉末都应该显示同等程度的振实密度。In general, the smaller the particle size of the scale-like, scale-like, and plate-like carbon materials tends to be, the worse the fillability is. This is considered to be due to the following reasons: as the particles become more amorphous by pulverization, or protrusions such as "burrs", "peeling" or "bends" generated on the particle surface increase, and some degree of deformation occurs on the particle surface. The strength is caused by the adhesion of finer amorphous particles, etc., so that the resistance between adjacent particles becomes larger, and the filling property is deteriorated. If the amorphousness of these particles is reduced and the particle shape is close to spherical, even if the particle size becomes smaller, the filling property will be reduced very little. In theory, both large particle size carbon powder and small particle size carbon powder should be Shows an equivalent degree of tap density.
作为这些天然石墨和/或天然石墨的加工物的比率,通常为0.1质量%以上、优选为1质量%以上、更优选为10质量%以上、进一步优选为20质量%以上,其上限通常为99.9质量%以下、优选为99质量%以下、更优选为90质量%以下、进一步优选为80质量%以下。如果低于该范围,则有时难以得到通过加入天然石墨和/或天然石墨的加工物而带来的性价比的提高。另一方面,如果超过该范围,则有时难以得到不同的负极活性物质所带来的效果的提高。The ratio of these natural graphite and/or processed products of natural graphite is usually 0.1% by mass or more, preferably 1% by mass or more, more preferably 10% by mass or more, still more preferably 20% by mass or more, and the upper limit is usually 99.9% by mass. Mass % or less, Preferably it is 99 mass % or less, More preferably, it is 90 mass % or less, More preferably, it is 80 mass % or less. If it is less than this range, it may be difficult to improve the cost performance by adding natural graphite and/or a processed product of natural graphite. On the other hand, if it exceeds this range, it may be difficult to obtain the improvement of the effect by a different negative electrode active material.
[[微孔容积等]][[pore volume, etc.]]
作为本发明的锂二次电池的负极活性物质使用的负极活性物质的细孔容积,通过水银孔隙率计(水银压入法)求得的由于相当于直径为0.01μm~1μm的粒子内的空隙、粒子表面的高低不平造成的凹凸的量(下面,简记为“微孔容积”)通常为0.01mL/g以上,优选为0.05mL/g以上,更优选为0.1mL/g以上,其上限通常为0.6mL/g以下,优选为0.4mL/g以下,更优选为0.3mL/g以下的范围。如果超过该范围,则制造成极板时需要大量的粘合剂。如果低于该范围,则高电流密度充放电特性降低,并且,有时不能得到充放电时的电极膨胀收缩的缓和效果。The pore volume of the negative electrode active material used as the negative electrode active material of the lithium secondary battery of the present invention is obtained by mercury porosimetry (mercury porosimetry) due to the voids in particles corresponding to a diameter of 0.01 μm to 1 μm. , The amount of unevenness on the surface of the particles (hereinafter referred to simply as "micropore volume") is usually more than 0.01mL/g, preferably more than 0.05mL/g, more preferably more than 0.1mL/g, the upper limit Usually it is 0.6 mL/g or less, preferably 0.4 mL/g or less, more preferably 0.3 mL/g or less. If it exceeds this range, a large amount of binder will be required to manufacture an electrode plate. If it is less than this range, the high current density charge-discharge characteristics will deteriorate, and the effect of alleviating electrode expansion and contraction during charge-discharge may not be obtained.
另外,总微孔容积优选为0.1mL/g以上,更优选为0.25mL/g以上,其上限通常为10mL/g以下,优选为5mL/g以下,更优选为2mL/g以下的范围。如果超过该范围,则在制成基板时有时需要大量的粘合剂。如果低于该范围,则在制成极板时有时不能得到增稠剂或粘结剂的分散效果。In addition, the total pore volume is preferably 0.1 mL/g or more, more preferably 0.25 mL/g or more, and its upper limit is usually 10 mL/g or less, preferably 5 mL/g or less, more preferably 2 mL/g or less. If it exceeds this range, a large amount of adhesive may be required to form a substrate. If it is less than this range, the dispersion effect of the thickener or the binder may not be obtained when forming an electrode plate.
另外,平均微孔径优选为0.05μm以上,更优选为0.1μm以上,进一步优选为0.5μm以上,其上限通常为50μm以下,优选为20μm以下,更优选为10μm以下的范围。如果超过该范围,则有时需要大量的粘合剂。如果低于该范围,则有时高电流密度充放电特性降低。In addition, the average pore diameter is preferably 0.05 μm or more, more preferably 0.1 μm or more, even more preferably 0.5 μm or more, and the upper limit thereof is usually 50 μm or less, preferably 20 μm or less, more preferably 10 μm or less. If this range is exceeded, a large amount of adhesive is sometimes required. If it is less than this range, the high current density charge-discharge characteristics may deteriorate.
作为用于水银孔隙率计的装置,可以使用水银孔隙率计(autopore9520;micrometritics公司制造)。称量0.2g左右的试样(负极材料),密封入粉末用容器中,在室温、真空下(50μmHg以下)进行10分钟脱气,实施前处理。接着,减压至4psia(约28kPa),导入水银,从4psia(约28kPa)阶段状升压至40000psia(约280MPa),然后降压至25psia(约170kPa)。升压时的阶段数为80阶段以上,在各阶段中,在10秒的平衡时间后,测定水银压入量。使用Washburn式从这样得到的水银压入曲线计算出微孔分布。另外,水银的表面张力(γ)为485dyne/cm,接触角(φ)为140°。平均微孔径使用累积微孔体积达到50%时的微孔径。As an apparatus used for the mercury porosimeter, a mercury porosimeter (autopore9520; manufactured by Micrometrics Inc.) can be used. About 0.2 g of a sample (negative electrode material) was weighed, sealed in a container for powder, and degassed at room temperature under vacuum (50 μmHg or less) for 10 minutes to perform pretreatment. Next, the pressure is reduced to 4psia (about 28kPa), mercury is introduced, the pressure is raised from 4psia (about 28kPa) to 40000psia (about 280MPa) in steps, and then the pressure is lowered to 25psia (about 170kPa). The number of stages during the pressurization was 80 or more, and the amount of mercury intrusion was measured after an equilibration time of 10 seconds in each stage. The micropore distribution was calculated from the mercury intrusion curve thus obtained using the Washburn formula. In addition, the surface tension (γ) of mercury is 485 dyne/cm, and the contact angle (φ) is 140°. As the average pore diameter, the pore diameter at which the cumulative pore volume reaches 50% is used.
[[灰分]][[ash]]
本发明的锂二次电池的负极活性物质的灰分相对于石墨质碳粒子的总质量优选为1质量%以下、更优选为0.5质量%以下、特别优选为0.1质量%以下。此外,其下限优选为1ppm以上。如果超过上述范围,则充放电时与非水电解液的反应而引起的电池性能的劣化不能忽视。另一方面,如果低于该范围,则在制造上需要较长时间和能量以及用于防止污染的设备,有时成本会上升。The ash content of the negative electrode active material of the lithium secondary battery of the present invention is preferably 1% by mass or less, more preferably 0.5% by mass or less, particularly preferably 0.1% by mass or less, based on the total mass of the graphitic carbon particles. In addition, the lower limit thereof is preferably 1 ppm or more. If the above-mentioned range is exceeded, the degradation of the battery performance due to the reaction with the non-aqueous electrolytic solution during charge and discharge cannot be ignored. On the other hand, if it is less than this range, long time and energy are required for manufacturing and equipment for preventing pollution, and the cost may increase.
[[取向比]][[Orientation Ratio]]
作为本发明的锂二次电池的负极活性物质使用的石墨质碳粒子的取向比通常为0.005以上、优选为0.01以上、更优选为0.015以上,上限理论上为0.67以下。如果低于该范围,则高密度充放电特性有时降低。The orientation ratio of the graphitic carbon particles used as the negative electrode active material of the lithium secondary battery of the present invention is usually 0.005 or more, preferably 0.01 or more, more preferably 0.015 or more, and the upper limit is theoretically 0.67 or less. If it is less than this range, the high-density charge and discharge characteristics may deteriorate.
取向比通过X射线衍射测定。使用非对称皮尔森VII作为分布函数,拟合通过X射线衍射得到的碳的(110)衍射和(004)衍射的峰,进行峰分离,分别计算出(110)衍射和(004)衍射的峰的积分强度。由得到的积分强度计算出由(110)衍射积分强度/(004)衍射积分强度表示的比值,将该比值定义为活性物质取向比。The orientation ratio was measured by X-ray diffraction. Using asymmetric Pearson VII as a distribution function, fit the peaks of (110) diffraction and (004) diffraction of carbon obtained by X-ray diffraction, perform peak separation, and calculate the peaks of (110) diffraction and (004) diffraction respectively integral strength. A ratio represented by (110) diffraction integrated intensity/(004) diffraction integrated intensity was calculated from the obtained integrated intensity, and this ratio was defined as the active material orientation ratio.
这里的X射线衍射测定条件如下。另外,“2θ”表示衍射角。The X-ray diffraction measurement conditions here are as follows. In addition, "2θ" represents a diffraction angle.
·靶:Cu(Kα射线)石墨单色光度仪Target: Cu (Kα ray) graphite monochromator
·狭缝:发散狭缝=1度,受光狭缝=0.1mm,散射狭缝=1度· Slit: divergence slit = 1 degree, light receiving slit = 0.1mm, scattering slit = 1 degree
·测定范围和步进角/测定时间・Measurement range and step angle/measurement time
(110)面:76.5度≤2θ≤78.5度0.01度/3秒(110) surface: 76.5 degrees ≤ 2θ ≤ 78.5 degrees 0.01 degrees / 3 seconds
(004)面:53.5度≤2θ≤56.0度0.01度/3秒(004) plane: 53.5 degrees ≤ 2θ ≤ 56.0 degrees 0.01 degrees / 3 seconds
[[长径比]][[Aspect Ratio]]
作为本发明的锂二次电池的负极活性物质使用的石墨质碳粒子的长径比理论上为1以上,上限为10以下,优选为8以下,更优选为5以下。如果超过其上限,制成极板时拉丝或无法得到均匀的涂布面,高电流密度充放电特性有时降低。The aspect ratio of the graphitic carbon particles used as the negative electrode active material of the lithium secondary battery of the present invention is theoretically 1 or more, the upper limit is 10 or less, preferably 8 or less, more preferably 5 or less. If the upper limit is exceeded, the electrode plate may be threaded or a uniformly coated surface may not be obtained, and the high current density charge and discharge characteristics may deteriorate.
另外,长径比用三维观察时粒子的最长径A和与其垂直的最短径B的比值A/B来表示。粒子的观察通过可以放大观察的扫描型电子显微镜来进行。选择固定在厚度50微米以下的金属的端面上的任意50个石墨粒子,旋转、倾斜固定有试样的载物台,分别测定这些粒子的A、B,求出A/B的平均值。In addition, the aspect ratio is represented by the ratio A/B of the longest diameter A of a particle and the shortest diameter B perpendicular thereto in three-dimensional observation. Observation of particles is performed with a scanning electron microscope capable of magnified observation. Select any 50 graphite particles fixed on the end face of the metal with a thickness of 50 microns or less, rotate and tilt the stage on which the sample is fixed, measure A and B of these particles, and calculate the average value of A/B.
本发明的具有负极[8]的锂二次电池只要含有性质不同的2种以上的负极活性物质即可,负极活性物质的种类没有特别限定。但是,对于面间距(d002)、微晶尺寸(Lc)、取向比、极板取向比等通过X射线衍射测定的性质;拉曼R值、拉曼半值宽度等与拉曼光谱相关的性质;以及灰分,由于上述数值以碳质物质为前提,因此上述数值之差适用于碳质物质。另一方面,对于中值粒径、模径、Z等与粒度分布相关的性质;BET比表面积;微孔容积、总微孔容积、平均微孔径等通过水银空隙率计测定的性质;真密度;圆形度;振实密度;以及长径比,上述数值不限于碳质物质,适用于所有可以用作负极活性物质的物质,上述数值之差也适用于所有物质。但是,优选具有上述2种性质的物质为碳质物质。此时,上述数值看作是表示碳质物质的性质的值,优选使用该性质不同的2种以上的碳质物质作为负极活性物质。The lithium secondary battery having the negative electrode [8] of the present invention only needs to contain two or more negative electrode active materials having different properties, and the type of negative electrode active material is not particularly limited. However, for properties measured by X-ray diffraction such as interplanar spacing (d002), crystallite size (Lc), orientation ratio, and plate orientation ratio; Raman R value, Raman half-value width, etc. ; and ash content, since the above values are premised on carbonaceous substances, the difference between the above values applies to carbonaceous substances. On the other hand, for properties related to particle size distribution such as median particle size, modulus diameter, Z, etc.; BET specific surface area; micropore volume, total micropore volume, average micropore diameter, etc., measured by mercury porosity meter; true density ; circularity; tap density; and aspect ratio, the above-mentioned values are not limited to carbonaceous materials, but are applicable to all materials that can be used as negative electrode active materials, and the difference between the above-mentioned values is also applicable to all materials. However, it is preferable that the substance having the above two properties is a carbonaceous substance. In this case, the above numerical values are regarded as values representing properties of the carbonaceous material, and it is preferable to use two or more types of carbonaceous materials having different properties as the negative electrode active material.
[负极[8]的2种以上负极活性物质的混合方法][Mixing method of two or more negative electrode active materials of the negative electrode [8]]
混合2种以上负极活性物质时,使用的装置没有特别限定,例如可举出V型混合机、W型混合机、容器可变型混合机、混炼机、滚筒式混合机、剪切混合机等。When mixing two or more negative electrode active materials, the device used is not particularly limited, and examples include V-type mixers, W-type mixers, variable container mixers, kneaders, tumble mixers, shear mixers, etc. .
[负极[8]的电极制造][Electrode manufacturing of negative electrode [8]]
负极[8]的制造可以通过常规方法进行,可以与上述同样地形成负极[8]。集电体、集电体与活性物质层的厚度比、电极密度、粘合剂、极板取向比、阻抗等也与上述相同。The manufacture of the negative electrode [8] can be performed by a conventional method, and the negative electrode [8] can be formed in the same manner as above. The current collector, the thickness ratio of the current collector to the active material layer, electrode density, binder, plate orientation ratio, impedance, and the like are also the same as described above.
<负极[9]和[10]><Negative electrodes [9] and [10]>
下面对本发明的锂二次电池中使用的负极[9](方式A)和负极[10](方式B)进行说明,所述负极[9]含有振实密度为0.1g/cm3以上,并且采用水银孔隙率计测定的相当于直径0.01μm~1μm范围的粒子的微孔容积为0.01mL/g以上的负极活性物质;所述负极[10]充电到该负极的标称容量的60%时,由负极的对置电池产生的反应电阻为500Ω以下。Next, the negative electrode [9] (form A) and the negative electrode [10] (form B) used in the lithium secondary battery of the present invention will be described. The negative electrode [9] contains a tap density of 0.1 g/cm 3 or more, and Negative electrode active materials with a micropore volume of 0.01 mL/g or more corresponding to particles in the range of 0.01 μm to 1 μm in diameter measured by a mercury porosimeter; when the negative electrode [10] is charged to 60% of the nominal capacity of the negative electrode , The reaction resistance generated by the opposite battery of the negative electrode is 500Ω or less.
《负极[9](方式A)》"Negative electrode [9] (mode A)"
本发明的方式A涉及一种锂二次电池,其中,对本发明中使用的负极用其中含有的负极活性物质的物性进行规定,并且该锂二次电池包括非水电解液和负极,所述非水电解液含有上述特定化合物,所述负极含有具有下述特定物性的负极活性物质。下面对本发明的方式A进行说明。Mode A of the present invention relates to a lithium secondary battery in which the physical properties of the negative electrode active material contained in the negative electrode used in the present invention are specified, and the lithium secondary battery includes a non-aqueous electrolytic solution and a negative electrode, the non-aqueous The aqueous electrolytic solution contains the above-mentioned specific compound, and the negative electrode contains a negative electrode active material having the following specific physical properties. Embodiment A of the present invention will be described below.
[负极[9]的负极活性物质][Negative electrode active material of the negative electrode [9]]
本发明的方式A中的负极活性物质在电化学上可以吸留、放出锂离子,并且还至少满足以下的(a)和(b)的要件。The negative electrode active material in aspect A of the present invention can electrochemically occlude and release lithium ions, and also satisfies at least the following requirements (a) and (b).
(a)振实密度为0.1g/cm3以上;(a) The tap density is above 0.1g/ cm3 ;
(b)采用水银孔隙率计测定的0.01μm~1μm的范围的微孔容积为0.01mL/g以上。(b) The pore volume in the range of 0.01 μm to 1 μm measured by a mercury porosimeter is 0.01 mL/g or more.
[[振实密度]][[Tap Density]]
本发明的锂二次电极的负极[9]中含有的负极活性物质的振实密度优选为0.1g/cm3以上,更优选为0.5g/cm3以上,进一步优选为0.7g/cm3以上,特别优选为0.9g/cm3以上。另外,其上限优选为2g/cm3以下,更优选为1.8g/cm3以下,特别优选为1.6g/cm3以下。如果振实密度低于该范围,则特别是不能实现高输出功率的效果。另一方面,如果超过该范围,则电极中的粒子间的空隙过少,非水电解液中的流路减少,有时输出功率本身减少。The tap density of the negative electrode active material contained in the negative electrode [9] of the lithium secondary electrode of the present invention is preferably 0.1 g/cm 3 or more, more preferably 0.5 g/cm 3 or more, even more preferably 0.7 g/cm 3 or more , particularly preferably 0.9 g/cm 3 or more. In addition, the upper limit thereof is preferably 2 g/cm 3 or less, more preferably 1.8 g/cm 3 or less, particularly preferably 1.6 g/cm 3 or less. If the tap density is lower than this range, especially the effect of high output cannot be achieved. On the other hand, if it exceeds this range, the gaps between the particles in the electrode will be too small, the flow path in the non-aqueous electrolyte solution will be reduced, and the output power itself may be reduced.
在本发明中,振实密度如下定义:使试样通过孔径300μm的筛子,使试样落入20cm3的振实容器中,到试样充满容器的上端面后,使用粉末密度测定器(例如,seishin企业公司制造的Tap densor),进行1000次冲程长度10mm的振动,由此时的体积和重量计算密度,将该值作为振实密度。In the present invention, the tap density is defined as follows: the sample is passed through a sieve with an aperture of 300 μm, the sample is dropped into a 20 cm container for vibration, and after the sample is filled with the upper end of the container, a powder density measuring device (such as , Tap densor manufactured by Seishin Enterprise Co., Ltd.) was vibrated 1000 times with a stroke length of 10 mm, and the density was calculated from the volume and weight at this time, and this value was taken as the tap density.
[[微孔容积]][[pore volume]]
本发明的锂二次电池的负极[9]中含有的负极活性物质的微孔容积,通过水银孔隙率计(水银压入法)求得的由于相当于直径为0.01μm~1μm的粒子内的空隙、粒子表面的高低不平造成的凹凸的量(下面,简记为“微孔容积”)为0.01mL/g以上,优选为0.05mL/g以上,更优选为0.1mL/g以上,其上限通常为0.6mL/g以下,优选为0.4mL/g以下,更优选为0.3mL/g以下的范围。如果超过该范围,则制造成极板时需要大量的粘合剂。另一方面,如果低于该范围,则不能实现长寿命、高输出功率。The micropore volume of the negative electrode active material contained in the negative electrode [9] of the lithium secondary battery of the present invention is obtained by the mercury porosimeter (mercury intrusion method) because it is equivalent to the volume in particles with a diameter of 0.01 μm to 1 μm. The amount of voids and irregularities on the particle surface (hereinafter, simply referred to as "micropore volume") is 0.01 mL/g or more, preferably 0.05 mL/g or more, more preferably 0.1 mL/g or more, and the upper limit Usually it is 0.6 mL/g or less, preferably 0.4 mL/g or less, more preferably 0.3 mL/g or less. If it exceeds this range, a large amount of binder will be required to manufacture an electrode plate. On the other hand, if it is below this range, long life and high output cannot be realized.
另外,总微孔容积优选为0.1mL/g以上,更优选为0.25mL/g以上,进一步优选为0.4mL/g以上,其上限通常为10mL/g以下,优选为5mL/g以下,更优选为2mL/g以下的范围。如果超过该范围,则在制成基板时有时需要大量的粘合剂。如果低于该范围,则在制成极板时有时不能得到增稠剂或粘结剂的分散效果。这里所说的“总微孔容积”是指在下述测定条件的全部范围内测定的微孔容积之和。In addition, the total pore volume is preferably 0.1 mL/g or more, more preferably 0.25 mL/g or more, and even more preferably 0.4 mL/g or more, and the upper limit thereof is usually 10 mL/g or less, preferably 5 mL/g or less, more preferably It is in the range of 2 mL/g or less. If it exceeds this range, a large amount of adhesive may be required to form a substrate. If it is less than this range, the dispersion effect of the thickener or the binder may not be obtained when forming an electrode plate. The "total pore volume" as used herein refers to the sum of the pore volumes measured in the entire range of the following measurement conditions.
另外,平均微孔径优选为0.05μm以上,更优选为0.1μm以上,进一步优选为0.5μm以上,其上限通常为50μm以下,优选为20μm以下,更优选为10μm以下的范围。如果超过该范围,则有时需要大量的粘合剂。如果低于该范围,则有时高电流密度充放电特性降低。In addition, the average pore diameter is preferably 0.05 μm or more, more preferably 0.1 μm or more, even more preferably 0.5 μm or more, and the upper limit thereof is usually 50 μm or less, preferably 20 μm or less, more preferably 10 μm or less. If this range is exceeded, a large amount of adhesive is sometimes required. If it is less than this range, the high current density charge-discharge characteristics may deteriorate.
作为用于水银孔隙率计的装置,可以使用水银孔隙率计(autopore9520;micrometritics公司制造)。称量0.2g左右的试样(负极材料),密封入粉末用容器中,在室温、真空下(50μmHg以下)进行10分钟脱气,实施前处理。接着,减压至4psia(约28kPa),导入水银,从4psia(约28kPa)阶段状升压至40000psia(约280MPa),然后降压至25psia(约170kPa)。升压时的阶段数为80阶段以上,在各阶段中,在10秒的平衡时间后,测定水银压入量。使用Washburn式从这样得到的水银压入曲线计算出微孔分布。另外,水银的表面张力(γ)为485dyne/cm,接触角(φ)为140°。平均微孔径使用累积微孔体积达到50%时的微孔径。As an apparatus used for the mercury porosimeter, a mercury porosimeter (autopore9520; manufactured by Micrometrics Inc.) can be used. About 0.2 g of a sample (negative electrode material) was weighed, sealed in a container for powder, and degassed at room temperature under vacuum (50 μmHg or less) for 10 minutes to perform pretreatment. Next, the pressure is reduced to 4psia (about 28kPa), mercury is introduced, the pressure is raised from 4psia (about 28kPa) to 40000psia (about 280MPa) in steps, and then the pressure is lowered to 25psia (about 170kPa). The number of stages during the pressurization was 80 or more, and the amount of mercury intrusion was measured after an equilibration time of 10 seconds in each stage. The micropore distribution was calculated from the mercury intrusion curve thus obtained using the Washburn formula. In addition, the surface tension (γ) of mercury is 485 dyne/cm, and the contact angle (φ) is 140°. As the average pore diameter, the pore diameter at which the cumulative pore volume reaches 50% is used.
本发明的方式A的锂二次电池,对于负极活性物质,只要满足上述要件(a)和(b)就可以发挥上述本发明的效果,充分发挥性能,进一步地,对于负极活性物质,优选同时满足下述物性的任意一项或多项。另外,对于负极,特别优选同时满足后述的方式B中的负极的物性或结构中的任意一项或多项。In the lithium secondary battery of mode A of the present invention, as long as the above-mentioned requirements (a) and (b) are satisfied for the negative electrode active material, the effect of the above-mentioned present invention can be exerted, and the performance can be fully exerted. Further, for the negative electrode active material, preferably simultaneously Satisfy any one or more of the following physical properties. In addition, it is particularly preferable for the negative electrode to simultaneously satisfy any one or more of the physical properties and structure of the negative electrode in Embodiment B described later.
[[BET比表面积]][[BET specific surface area]]
使用BET法测定的本发明的锂二次电池的负极[9]中含有的负极活性物质的比表面积优选为0.1m2/g以上,特别优选为0.7m2/g以上,更优选为1m2/g以上,进一步优选为1.5m2/g以上。其上限优选为100m2/g以下,特别优选为50m2/g以下,更优选为25m2/g以下,进一步优选为15m2/g以下。如果BET比表面积的值低于上述范围,则在作为负极材料使用的情况下,在充电时锂的接受性变差,锂容易在电极表面析出,有时不能得到高输出功率。另一方面,如果超过上述范围,则在作为负极材料使用时,与电解液的反应性增加,产生的气体变多,有时难以得到优选的电池。The specific surface area of the negative electrode active material contained in the negative electrode [9] of the lithium secondary battery of the present invention measured by the BET method is preferably 0.1 m 2 /g or more, particularly preferably 0.7 m 2 /g or more, more preferably 1 m 2 /g or more, more preferably 1.5 m 2 /g or more. The upper limit thereof is preferably 100 m 2 /g or less, particularly preferably 50 m 2 /g or less, more preferably 25 m 2 /g or less, and still more preferably 15 m 2 /g or less. If the value of the BET specific surface area is lower than the above range, when used as a negative electrode material, the acceptance of lithium at the time of charging will deteriorate, and lithium will be easily deposited on the electrode surface, and high output power may not be obtained. On the other hand, when it exceeds the above-mentioned range, when used as a negative electrode material, the reactivity with the electrolytic solution increases and gas generation increases, making it difficult to obtain a preferable battery in some cases.
BET比表面积定义为如下测定的值:使用表面积计(例如,大仓理研制造的全自动表面积测定装置),在氮气流通、350℃下对试样进行15分钟预干燥,然后,使用氮气相对于大气压的相对压力值准确调整为0.3的氮氦混合气体,通过采用气体流动法的氮吸附BET1点法测定。The BET specific surface area is defined as a value measured as follows: Using a surface area meter (for example, a fully automatic surface area measuring device manufactured by Okura Riken), the sample is pre-dried at 350° C. for 15 minutes under nitrogen flow, and then, using nitrogen gas relative to The relative pressure value of the atmospheric pressure is accurately adjusted to 0.3 nitrogen-helium mixed gas, and it is measured by the nitrogen adsorption BET 1-point method using the gas flow method.
[[体积平均粒径]][[Volume average particle size]]
本发明的锂二次电池的负极[9]中含有的负极活性物质的体积平均粒径定义为通过激光衍射/散射法求得的体积基准的平均粒径(中值粒径),优选为1μm以上,特别优选为3μm以上,更优选为5μm以上,进一步优选为7μm以上。另外,其上限通常为50μm以下,优选为40μm以下,更优选为30μm以下,进一步优选为25μm以下。如果低于上述范围,则不可逆容量增大,有时导致初期电池容量的损失。另外,如果超过上述范围,则在制成电极极板时,容易形成不均匀的涂布面,有时在电池制作工序上是不优选的。The volume average particle size of the negative electrode active material contained in the negative electrode [9] of the lithium secondary battery of the present invention is defined as the average particle size (median particle size) based on the volume obtained by the laser diffraction/scattering method, preferably 1 μm Above, especially preferably 3 μm or more, more preferably 5 μm or more, even more preferably 7 μm or more. In addition, the upper limit thereof is usually 50 μm or less, preferably 40 μm or less, more preferably 30 μm or less, and still more preferably 25 μm or less. If it is less than the above-mentioned range, the irreversible capacity may increase, resulting in loss of initial battery capacity. Moreover, if it exceeds the said range, it will become easy to form an uneven coating surface at the time of making an electrode pad, and it may be unpreferable in a battery manufacturing process.
在本发明中,体积基准平均粒径用中值粒径来定义,所述中值粒径通过以下方法测定:使碳粉末分散在作为表面活性剂的聚氧乙烯(20)山梨糖醇酐单月桂酸酯的0.2质量%水溶液(约1mL)中,使用激光衍射/散射式粒度分布计(例如,堀场制作所公司制造的LA-700)来测定。In the present invention, the volume-based average particle diameter is defined by a median particle diameter measured by dispersing carbon powder in polyoxyethylene (20) sorbitan as a surfactant. In a 0.2% by mass aqueous solution (about 1 mL) of monolaurate, it measures using a laser diffraction/scattering particle size distribution meter (for example, LA-700 manufactured by Horiba, Ltd.).
[[圆形度]][[Circularity]]
本发明的锂二次电极的负极[9]中含有的负极活性物质的圆形度通常为0.1以上,优选为0.5以上,更优选为0.8以上,特别优选为0.85以上,进一步优选为0.9以上。作为上限,圆形度为1时理论上形成真球。如果圆形度低于该范围,则电极化时有可能产生拉丝等问题。The circularity of the negative electrode active material contained in the negative electrode [9] of the lithium secondary electrode of the present invention is usually 0.1 or more, preferably 0.5 or more, more preferably 0.8 or more, particularly preferably 0.85 or more, and more preferably 0.9 or more. As an upper limit, when the circularity is 1, a true sphere is theoretically formed. If the circularity is lower than this range, problems such as stringing may occur during electric polarization.
本发明中所说的圆形度用下式定义。The circularity referred to in the present invention is defined by the following formula.
圆形度=(具有与粒子投影形状相同面积的等效圆的周长)/(粒子投影形状的实际周长)Circularity = (perimeter of an equivalent circle having the same area as the particle projected shape)/(actual perimeter of the particle projected shape)
作为圆形度的值使用如下测定的值:使用流动式粒子图像分析装置(例如,Sysmex Industrial公司制造的FPIA),将约0.2g试样分散于作为表面活性剂的聚氧乙烯(20)山梨糖醇酐单月桂酸酯的0.2质量%水溶液(约50mL)中,以60W的输出功率照射1分钟28kHz的超声波后,指定0.6~400μm为检测范围,对粒径3~40μm范围的粒子进行测定。As the value of circularity, the value measured as follows is used: using a flow type particle image analyzer (for example, FPIA manufactured by Sysmex Industrial Co., Ltd.), about 0.2 g of a sample is dispersed in polyoxyethylene (20) sorbitol as a surfactant. After irradiating a 0.2% by mass aqueous solution (approximately 50mL) of sugar-alcohol monolaurate with an output of 60W for 1 minute at 28kHz ultrasonic waves, specify a detection range of 0.6-400μm, and measure particles with a particle diameter of 3-40μm .
[[面间距(d002)]][[Space between faces (d002)]]
本发明的锂二次电池的负极[9]中含有的负极活性物质采用广角X射线衍射法测定的(002)面的面间距(d002)通常为0.38nm以下,优选为0.36nm以下,更优选为0.35nm以下,进一步优选为0.345nm以下,其下限为石墨的理论值0.335nm以上。如果超过该范围,则结晶性显著降低,不可逆容量有时增加。The interplanar distance (d002) of the (002) planes (d002) of the negative electrode active material contained in the negative electrode [9] of the lithium secondary battery of the present invention measured by wide-angle X-ray diffraction method is usually 0.38nm or less, preferably 0.36nm or less, more preferably It is 0.35 nm or less, more preferably 0.345 nm or less, and its lower limit is 0.335 nm or more, the theoretical value of graphite. If it exceeds this range, the crystallinity is significantly lowered, and the irreversible capacity may increase.
本发明中所说的通过广角X射线衍射法测定的(002)面的面间距(d002),是指通过学振法以X射线衍射求得的晶格面(002)面的d值(层间距离)。The interplanar spacing (d002) of the (002) plane measured by the wide-angle X-ray diffraction method in the present invention refers to the d value (layer distance).
[[微晶尺寸(Lc)]][[Crystalline Size (Lc)]]
此外,通过学振法以X射线衍射求得的负极活性物质的微晶尺寸(Lc)没有特别限定,通常为0.1nm以上,优选为0.5nm以上,更优选为1nm以上的范围。如果低于该范围,则结晶性显著降低,不可逆容量有时增加。In addition, the crystallite size (Lc) of the negative electrode active material obtained by X-ray diffraction by the Gakushin method is not particularly limited, but is usually 0.1 nm or more, preferably 0.5 nm or more, and more preferably 1 nm or more. If it is less than this range, the crystallinity will decrease remarkably, and the irreversible capacity may increase.
[[真密度]][[true density]]
本发明的锂二次电池的负极[9]中含有的负极活性物质的真密度通常为1.5g/cm3以上,优选为1.7g/cm3以上,更优选为1.8g/cm3以上,进一步优选为1.85g/cm3以上,其上限为2.26g/cm3以下。上限为石墨的理论值。如果低于该范围,则碳的结晶性过低,不可逆容量有时增加。在本发明中,真密度定义为通过使用丁醇的液相置换法(比重计法)测定的值。The true density of the negative electrode active material contained in the negative electrode [9] of the lithium secondary battery of the present invention is generally 1.5 g/cm 3 or more, preferably 1.7 g/cm 3 or more, more preferably 1.8 g/cm 3 or more, and further It is preferably 1.85 g/cm 3 or more, and the upper limit thereof is 2.26 g/cm 3 or less. The upper limit is the theoretical value of graphite. If it is less than this range, the crystallinity of carbon may be too low, and the irreversible capacity may increase. In the present invention, the true density is defined as a value measured by a liquid phase displacement method (pycnometer method) using butanol.
[[拉曼R值]][[Raman R value]]
本发明的锂二次电池的负极[9]中含有的负极活性物质的拉曼R值通常为0.01以上,优选为0.03以上,更优选为0.1以上。作为上限,优选为1.5以下,更优选为1.2以下,特别优选为0.5以下。如果拉曼R值低于该范围,则粒子表面的结晶性过高,随着充放电位点的减少,输出功率有时降低。另一方面,如果超过该范围,则粒子表面的结晶性降低,不可逆容量有时增加。The Raman R value of the negative electrode active material contained in the negative electrode [9] of the lithium secondary battery of the present invention is usually 0.01 or more, preferably 0.03 or more, more preferably 0.1 or more. The upper limit is preferably 1.5 or less, more preferably 1.2 or less, particularly preferably 0.5 or less. If the Raman R value is lower than this range, the crystallinity of the particle surface is too high, and the output power may decrease with the decrease of charge and discharge sites. On the other hand, if it exceeds this range, the crystallinity of the particle surface may decrease and the irreversible capacity may increase.
拉曼光谱的测定如下进行:使用拉曼分光器(例如日本分光社制造的拉曼分光器),使试样自然落下并填充在测定池内,对池内的试样表面照射氩离子激光,同时,使池在与激光垂直的面内旋转。对于得到的拉曼光谱,测定1580cm-1的峰PA的强度IA和1360cm-1的峰PB的强度IB,计算其强度比R(R=IB/IA),将其定义为负极活性物质的拉曼R值。另外,测定得到的拉曼光谱在1580cm-1附近的峰PA的半值宽度,将其定义为负极活性物质的拉曼半值宽度。The measurement of the Raman spectrum is carried out as follows: Using a Raman spectrometer (such as a Raman spectrometer manufactured by JASCO Corporation), the sample is naturally dropped and filled in the measurement cell, and the surface of the sample in the cell is irradiated with an argon ion laser. At the same time, Rotate the cell in a plane perpendicular to the laser. For the obtained Raman spectrum, measure the intensity I A of the peak PA at 1580 cm -1 and the intensity I B of the peak P B at 1360 cm -1 , calculate the intensity ratio R (R= IB / IA ), and define it as is the Raman R value of the negative electrode active material. In addition, the half-value width of the peak PA near 1580 cm −1 in the obtained Raman spectrum was measured, and this was defined as the Raman half-value width of the negative electrode active material.
另外,这里的拉曼测定条件如下。In addition, the Raman measurement conditions here are as follows.
·氩离子激光波长:514.5nmArgon ion laser wavelength: 514.5nm
·试样上的激光功率:15~25mW·Laser power on the sample: 15~25mW
·分辨率:10~20cm-1 ·Resolution: 10~20cm -1
·测定范围:1100cm-1~1730cm-1 · Measuring range: 1100cm -1 ~ 1730cm -1
·拉曼R值、拉曼半值宽度分析:本底处理Raman R value, Raman half-value width analysis: background processing
·光滑处理:单纯平均、卷积5点·Smooth processing: simple average, convolution 5 points
另外,本发明的锂二次电池的负极[9]中含有的负极活性物质在1580cm-1的拉曼半值宽度没有特别的限制,通常为10cm-1以上,优选为15cm-1以上,另外,其上限通常为150cm-1以下,优选为100cm-1以下,更优选为60cm-1以下的范围。如果拉曼半值宽度低于该范围,则粒子表面的结晶性过高,随着充放电位点的减少,输出功率有时降低。另一方面,如果高于该范围,则粒子表面的结晶性降低,不可逆容量有时增加。In addition, the Raman half-value width of the negative electrode active material contained in the negative electrode [9] of the lithium secondary battery of the present invention is not particularly limited at 1580 cm -1 , and is usually more than 10 cm -1 , preferably more than 15 cm -1 , and , the upper limit of which is usually 150 cm -1 or less, preferably 100 cm -1 or less, more preferably 60 cm -1 or less. If the Raman half-value width is below this range, the crystallinity of the surface of the particle is too high, and the output may decrease with the decrease of the charge and discharge sites. On the other hand, if it exceeds this range, the crystallinity of the particle surface may decrease, and the irreversible capacity may increase.
[[灰分]][[ash]]
本发明的锂二次电池的负极[9]中含有的负极活性物质的灰分优选为1质量%以下、更优选为0.5质量%以下、特别优选为0.1质量%以下。此外,作为下限,优选为1ppm以上。如果超过上述范围,则充放电时与非水电解液的反应而引起的电池性能的劣化不能忽视,循环保持率有时降低。另一方面,如果低于该范围,则在制造上需要较长时间和能量以及用于防止污染的设备,有时成本会上升。The ash content of the negative electrode active material contained in the negative electrode [9] of the lithium secondary battery of the present invention is preferably 1% by mass or less, more preferably 0.5% by mass or less, particularly preferably 0.1% by mass or less. In addition, the lower limit is preferably 1 ppm or more. If it exceeds the above-mentioned range, the degradation of battery performance due to the reaction with the non-aqueous electrolyte solution during charging and discharging cannot be ignored, and the cycle retention rate may decrease. On the other hand, if it is less than this range, long time and energy are required for manufacturing and equipment for preventing pollution, and the cost may increase.
[[长径比]][[Aspect Ratio]]
本发明的锂二次电池的负极[9]中含有的负极活性物质的的长径比理论上为1以上,作为上限,通常为10以下,优选为8以下,更优选为5以下。如果超过其上限,则制造负极时活性物质拉丝或无法得到均匀的涂布面,高电流密度充放电特性有时降低。The aspect ratio of the negative electrode active material contained in the negative electrode [9] of the lithium secondary battery of the present invention is theoretically 1 or more, and the upper limit is usually 10 or less, preferably 8 or less, more preferably 5 or less. If the upper limit is exceeded, the active material may become stringy or a uniform coating surface may not be obtained during the production of the negative electrode, and the high current density charge and discharge characteristics may be reduced.
另外,长径比用三维观察时粒子的最长径A和与其垂直的最短径B的比值A/B来表示。粒子的观察通过可以放大观察的扫描型电子显微镜来进行。选择固定在厚度50微米以下的金属的端面上的任意50个石墨粒子,旋转、倾斜固定有试样的载物台,分别测定这些粒子的A、B,求出A/B的平均值。In addition, the aspect ratio is represented by the ratio A/B of the longest diameter A of a particle and the shortest diameter B perpendicular thereto in three-dimensional observation. Observation of particles is performed with a scanning electron microscope capable of magnified observation. Select any 50 graphite particles fixed on the end face of the metal with a thickness of 50 microns or less, rotate and tilt the stage on which the sample is fixed, measure A and B of these particles, and calculate the average value of A/B.
[[取向比]][[Orientation Ratio]]
本发明的锂二次电池的负极[9]中含有的负极活性物质的取向比通常为0.005以上,优选为0.01以上,更优选为0.015以上,上限理论上为0.67以下。如果低于该范围,则高密度充放电特性有时降低。The orientation ratio of the negative electrode active material contained in the negative electrode [9] of the lithium secondary battery of the present invention is usually 0.005 or more, preferably 0.01 or more, more preferably 0.015 or more, and the upper limit is theoretically 0.67 or less. If it is less than this range, the high-density charge and discharge characteristics may deteriorate.
取向比通过X射线衍射测定。使用非对称皮尔森VII作为分布函数,拟合通过X射线衍射得到的碳的(110)衍射和(004)衍射的峰,进行峰分离,分别计算出(110)衍射和(004)衍射的峰的积分强度。由得到的积分强度计算出由(110)衍射积分强度/(004)衍射积分强度表示的比值,将该比值定义为负极活性物质取向比。The orientation ratio was measured by X-ray diffraction. Using asymmetric Pearson VII as a distribution function, fit the peaks of (110) diffraction and (004) diffraction of carbon obtained by X-ray diffraction, perform peak separation, and calculate the peaks of (110) diffraction and (004) diffraction respectively integral strength. A ratio represented by (110) diffraction integrated intensity/(004) diffraction integrated intensity was calculated from the obtained integrated intensity, and this ratio was defined as the negative electrode active material orientation ratio.
这里的X射线衍射测定条件如下。另外,“2θ”表示衍射角。The X-ray diffraction measurement conditions here are as follows. In addition, "2θ" represents a diffraction angle.
·靶:Cu(Kα射线)石墨单色光度仪Target: Cu (Kα ray) graphite monochromator
·狭缝:发散狭缝=1度,受光狭缝=0.1mm,散射狭缝=1度· Slit: divergence slit = 1 degree, light receiving slit = 0.1mm, scattering slit = 1 degree
·测定范围和步进角/测定时间・Measurement range and step angle/measurement time
(110)面:76.5度≤2θ≤78.5度0.01度/3秒(110) surface: 76.5 degrees ≤ 2θ ≤ 78.5 degrees 0.01 degrees / 3 seconds
(004)面:53.5度≤2θ≤56.0度0.01度/3秒(004) plane: 53.5 degrees ≤ 2θ ≤ 56.0 degrees 0.01 degrees / 3 seconds
[制造负极[9]的电极][Manufacture of electrode for negative electrode [9]]
负极的制造可以通过常规方法进行,可以与上述同样地形成负极[9]。集电体、集电体与活性物质层的厚度比、电极密度、粘合剂、极板取向比、阻抗等也与上述相同。The production of the negative electrode can be performed by a conventional method, and the negative electrode can be formed in the same manner as above [9]. The current collector, the thickness ratio of the current collector to the active material layer, electrode density, binder, plate orientation ratio, impedance, and the like are also the same as described above.
《对于负极[10](方式B)》"For the negative electrode [10] (mode B)"
本发明的负极[10](方式B)涉及一种锂二次电池,其中,对本发明中使用的负极用负极本身的物性和/或结构规定,该锂二次电池包括非水电解液和具有下述特定的物性和/或结构的负极,所述非水电解液含有上述的特定化合物。下面对本发明的负极[10](方式B)进行说明。The negative electrode [10] (form B) of the present invention relates to a lithium secondary battery, wherein, the negative electrode used in the present invention is specified for the physical properties and/or structure of the negative electrode itself, and the lithium secondary battery includes a non-aqueous electrolyte and has The following specific physical and/or structure of the negative electrode, the non-aqueous electrolyte contains the above-mentioned specific compounds. Next, the negative electrode [10] (form B) of the present invention will be described.
[负极[10]负极的物性、结构][Negative electrode [10] Physical properties and structure of the negative electrode]
以下对方式B的锂二次电池中使用的负极的物性和结构进行说明。负极在集电体上具有负极活性物质层。The physical properties and structure of the negative electrode used in the lithium secondary battery of Embodiment B will be described below. The negative electrode has a negative electrode active material layer on a current collector.
[[反应电阻]][[Reaction Resistance]]
本发明的方式B中使用的负极的对置阻抗(対向インピ一ダンス)测定中求得的反应电阻必须为500Ω以下,优选为100Ω以下、更优选为50Ω以下。下限没有特别限定。如果超过该范围,则制成锂二次电池时,循环试验之前和之后的输出功率特性降低。另外,有时会由于高电阻而产生循环保持率的降低。The reaction resistance obtained in the measurement of the opposite impedance (opposed impedance) of the negative electrode used in the aspect B of the present invention must be 500Ω or less, preferably 100Ω or less, more preferably 50Ω or less. The lower limit is not particularly limited. If it exceeds this range, when the lithium secondary battery is produced, the output characteristics before and after the cycle test will decrease. In addition, there may be a decrease in the cycle retention rate due to high electrical resistance.
下面说明对置阻抗的测定方法。一般在锂二次电池的阻抗测定中,对电池的正极端子和负极端子之间实施交流阻抗测定,但是若使用该方法则最终表示正极和负极混在一起的阻抗,不能明确地确定哪个正、负极电阻由哪个产生。因此,在本发明中,测定仅由负极的对置电池产生的反应电阻。下面叙述本发明中的反应电阻的测定方法。Next, the method of measuring the opposing impedance will be described. Generally, in the impedance measurement of lithium secondary batteries, the AC impedance is measured between the positive terminal and the negative terminal of the battery. However, if this method is used, the impedance of the positive and negative electrodes mixed together cannot be clearly determined which is positive and negative. Which produces the resistance. Therefore, in the present invention, the reaction resistance generated only by the counter battery of the negative electrode was measured. The method for measuring the reaction resistance in the present invention will be described below.
测定的锂二次电池使用如下的电池:以可以用5小时充电标称容量的电流值进行充电后,维持20分钟不充放电的状态,然后以可以用1小时放电标称容量的电流值进行放电,此时的容量为标称容量的80%以上。对于上述放电状态的锂二次电池,以可以用5小时充电标称容量的电流值充电至标称容量的60%,立即将锂二次电池转移至氩气氛围下的手套工作箱内。其中,将该锂二次电池在负极未放电或未短路的状态下快速地拆卸取出,如果是两面涂布电极,则不损害另一面电极活性物质地将一面的电极活性物质剥离,将2块负极电极冲孔成12.5mmφ,通过隔板使活性物质面不偏离地对置。向隔板和两负极间滴加电池中使用的非水电解液60μL并进行密封,保持不与外界接触的状态,在两负极的集电体上导电,实施交流阻抗法。测定是在温度25℃下、10-2~105Hz的频带下进行复阻抗测定,将求得的科利-科利曲线(コ一ル·コ一ル·プロツト)的负极电阻成分的圆弧近似为半圆,求得反应电阻(Rct)和双层容量(Cdl)。The measured lithium secondary battery uses the following battery: After charging with a current value that can charge the nominal capacity for 5 hours, maintain the state of not charging and discharging for 20 minutes, and then charge it with a current value that can discharge the nominal capacity for 1 hour. Discharge, the capacity at this time is more than 80% of the nominal capacity. For the lithium secondary battery in the above discharge state, charge it to 60% of the nominal capacity at a current value that can charge the nominal capacity for 5 hours, and immediately transfer the lithium secondary battery to a glove box under an argon atmosphere. Wherein, the lithium secondary battery is quickly disassembled and taken out in a state where the negative electrode is not discharged or short-circuited. If the electrode is coated on both sides, the electrode active material on one side is peeled off without damaging the electrode active material on the other side. The negative electrode was punched to a size of 12.5 mmφ, and the active material surface was opposed to each other without deviation through the separator. 60 μL of the non-aqueous electrolyte solution used in the battery was dripped between the separator and the two negative electrodes and sealed to keep it in a state of not being in contact with the outside world. Conduction was conducted on the current collectors of the two negative electrodes, and an AC impedance method was performed. The measurement was performed at a temperature of 25°C and a complex impedance measurement was performed in a frequency band of 10 -2 to 10 5 Hz, and the circle of the negative electrode resistance component of the obtained Coley-Cole curve (Colley-Cole plot) was calculated. The arc is approximated as a semicircle, and the reaction resistance (Rct) and double layer capacity (Cdl) are obtained.
本发明的方式B的锂二次电池中,对于负极,只要由负极的对置电池(对向セル)产生的反应电阻满足上述要件,就可以发挥上述本发明的效果,从而充分发挥性能,进一步地,优选同时满足下述负极的物性或结构的任意一项或多项。此外,对于负极活性物质,特别优选同时满足方式A项中所述的负极活性物质的物性的任意一项或多项。In the lithium secondary battery according to the aspect B of the present invention, as long as the reaction resistance generated by the opposite cell (opposite cell) of the negative electrode satisfies the above-mentioned requirements, the effect of the present invention can be exerted, so that the performance can be fully exhibited, and further Preferably, any one or more of the following physical properties or structures of the negative electrode are preferably satisfied at the same time. In addition, for the negative electrode active material, it is particularly preferable to simultaneously satisfy any one or more of the physical properties of the negative electrode active material described in item A of the aspect.
[[双层容量(Cdl)]][[Double Layer Capacity (Cdl)]]
本发明中使用的负极的对置阻抗测定中求得的双层容量(Cdl)优选为1×10-6F以上、特别优选为1×10-5F以上、更优选为3×10-5F以上。如果低于该范围,则由于可以反应的面积减少,结果输出功率有时降低。The double-layer capacity (Cdl) obtained by measuring the opposing impedance of the negative electrode used in the present invention is preferably 1×10 -6 F or higher, particularly preferably 1×10 -5 F or higher, more preferably 3×10 -5 F or above. If it is less than this range, since the area which can react decreases, output power may fall as a result.
[[粘合剂]][[Binder]]
作为将负极活性物质和集电体粘合的粘合剂,只要是对于非水电解液或电极制造时使用的溶剂稳定的材料即可,没有特别限制,可以使用的物质、粘合剂相对于全部负极活性物质层的比率等与上述相同。As the binder that binds the negative electrode active material and the current collector, as long as it is a stable material for the non-aqueous electrolyte or the solvent used in the manufacture of the electrode, there are no particular restrictions. The materials and binders that can be used are relatively The ratio etc. of all the negative electrode active material layers are the same as above.
[负极[10]电极制造][Negative electrode [10] electrode manufacturing]
负极[10]的制造可以通过常规方法进行,可以与上述同样地形成负极[10]。集电体、集电体与活性物质层的厚度比、电极密度、粘合剂、极板取向比、阻抗等也与上述相同。The manufacture of the negative electrode [10] can be performed by a conventional method, and the negative electrode [10] can be formed in the same manner as above. The current collector, the thickness ratio of the current collector to the active material layer, electrode density, binder, plate orientation ratio, impedance, and the like are also the same as described above.
<非水电解液><Non-aqueous electrolyte>
本发明中使用的非水电解液只要含有电解质(锂盐)及溶解该电解质的非水溶剂、特定化合物即可,没有特别限定,该非水电解液优选满足选自下述电解液[1]~电解液[9]中的任一条件的电解液:The non-aqueous electrolytic solution used in the present invention is not particularly limited as long as it contains an electrolyte (lithium salt) and a non-aqueous solvent and a specific compound for dissolving the electrolyte. The non-aqueous electrolytic solution preferably satisfies the following electrolytic solution [1] ~Electrolyte of any condition in the electrolyte [9]:
电解液[1]:构成电解液的非水溶剂是至少含有碳酸亚乙酯的混合溶剂,并且碳酸亚乙酯相对于非水溶剂总量的比例为1体积%~25体积%;Electrolyte [1]: The non-aqueous solvent constituting the electrolyte is a mixed solvent containing at least ethylene carbonate, and the ratio of ethylene carbonate to the total amount of non-aqueous solvent is 1% to 25% by volume;
电解液[2]:构成电解液的非水溶剂含有至少一种不对称链状碳酸酯,并且该不对称链状碳酸酯在全部非水溶剂中所占的含有比例为5体积%~90体积%;Electrolyte [2]: The nonaqueous solvent constituting the electrolyte contains at least one asymmetric chain carbonate, and the proportion of the asymmetric chain carbonate in all nonaqueous solvents is 5% to 90% by volume %;
电解液[3]:构成电解液的非水溶剂含有至少一种链状羧酸酯;Electrolyte [3]: the non-aqueous solvent constituting the electrolyte contains at least one chain carboxylate;
电解液[4]:构成电解液的非水溶剂含有闪点为70℃以上的溶剂,其含量为全部非水溶剂的60体积%以上;Electrolyte [4]: The non-aqueous solvent constituting the electrolyte contains a solvent with a flash point above 70°C, and its content is above 60% by volume of all non-aqueous solvents;
电解液[5]:含有LiN(CnF2n+1SO2)2(式中,n为1~4的整数)和/或二(草酸根合)硼酸锂作为构成电解液的锂盐;Electrolyte [5]: containing LiN(C n F 2n+1 SO 2 ) 2 (wherein, n is an integer of 1 to 4) and/or lithium bis(oxalato)borate as the lithium salt constituting the electrolyte;
电解液[6]:构成电解液的锂盐是含氟锂盐,并且在全部非水电解液中含有10ppm~300ppm的氟化氢(HF);Electrolyte [6]: The lithium salt constituting the electrolyte is a fluorine-containing lithium salt, and contains 10 ppm to 300 ppm of hydrogen fluoride (HF) in all non-aqueous electrolytes;
电解液[7]:在电解液中含有碳酸亚乙烯酯,该碳酸亚乙烯酯的含量为电解液总质量的0.001质量%~3质量%的范围;Electrolyte [7]: the electrolyte contains vinylene carbonate, and the content of the vinylene carbonate is in the range of 0.001% by mass to 3% by mass of the total mass of the electrolyte;
电解液[8]:在电解液中还含有选自上述通式(4)表示的化合物、含有氮和/或硫的杂环化合物、环状羧酸酯、含氟环状碳酸酯中的至少一种化合物,其在全部非水电解液中的含量为0.001质量%~5质量%的范围;Electrolyte solution [8]: the electrolyte solution also contains at least A compound whose content in the entire non-aqueous electrolyte is in the range of 0.001% by mass to 5% by mass;
电解液[9]:在电解液中,还含有过充电防止剂。Electrolyte solution [9]: The electrolyte solution also contains an overcharge preventing agent.
以下,首先对本发明的锂二次电池中通常使用的非水电解液进行说明。Hereinafter, first, non-aqueous electrolytic solutions generally used in the lithium secondary battery of the present invention will be described.
[锂盐][lithium salt]
作为电解质,只要是已知可以作为锂二次电池用非水电解液的电解质使用的锂盐则没有特别限定,例如可举出下述锂盐。The electrolyte is not particularly limited as long as it is a lithium salt known to be usable as an electrolyte of a nonaqueous electrolytic solution for lithium secondary batteries, and examples thereof include the following lithium salts.
无机锂盐:LiPF6、LiBF4、LiAsF6、LiSbF6等无机氟化物盐;LiClO4、LiBrO4、LiIO4等高卤酸盐;LiAlCl4等无机氯化物盐等。Inorganic lithium salts: LiPF 6 , LiBF 4 , LiAsF 6 , LiSbF 6 and other inorganic fluoride salts; LiClO 4 , LiBrO 4 , LiIO 4 and other perhalide salts; LiAlCl 4 and other inorganic chloride salts, etc.
含氟有机锂盐:LiCF3SO3等全氟链烷磺酸盐;LiN(CF3SO2)2、LiN(CF3CF2SO2)2、LiN(CF3SO2)(C4F9SO2)等全氟链烷磺酰亚胺盐;LiC(CF3SO2)3等全氟链烷磺酰甲基化盐,Li[PF5(CF2CF2CF3)]、Li[PF4(CF2CF2CF3)2]、Li[PF3(CF2CF2CF3)3]、Li[PF5(CF2CF2CF2CF3)]、Li[PF4(CF2CF2CF2CF3)2]、Li[PF3(CF2CF2CF2CF3)3]等氟烷基氟化磷酸盐等。Fluorinated organolithium salts: LiCF 3 SO 3 and other perfluoroalkane sulfonates; LiN(CF 3 SO 2 ) 2 , LiN(CF 3 CF 2 SO 2 ) 2 , LiN(CF 3 SO 2 )(C 4 F 9 SO 2 ) and other perfluoroalkanesulfonylimide salts; LiC(CF 3 SO 2 ) 3 and other perfluoroalkanesulfonylmethylation salts, Li[PF 5 (CF 2 CF 2 CF 3 )], Li [PF 4 (CF 2 CF 2 CF 3 ) 2 ], Li[PF 3 (CF 2 CF 2 CF 3 ) 3 ], Li[PF 5 (CF 2 CF 2 CF 2 CF 3 )], Li[PF 4 ( Fluoroalkyl fluorophosphates such as CF 2 CF 2 CF 2 CF 3 ) 2 ], Li[PF 3 (CF 2 CF 2 CF 2 CF 3 ) 3 ], etc.
草酸根合硼酸盐:二(草酸根合)硼酸锂、二氟草酸根合硼酸锂等。Oxalatoborates: lithium di(oxalato)borate, lithium difluorooxalatoborate, and the like.
它们可以单独使用一种,也可以以任意的组合和比率组合使用2种以上。其中,如果综合判断在非水溶剂中的溶解性、制成二次电池时的充放电特性、输出功率特性、循环特性等,则优选为LiPF6。These may be used individually by 1 type, and may use 2 or more types together in arbitrary combinations and ratios. Among them, LiPF 6 is preferable when the solubility in non-aqueous solvents, charge and discharge characteristics when used as a secondary battery, output characteristics, cycle characteristics, etc. are comprehensively judged.
组合使用2种以上时的优选的一个例子为组合使用LiPF6与LiBF4,此时,LiBF4占两者总计的比率优选为0.01质量%~20质量%,特别优选为0.1质量%~5质量%。A preferred example of using two or more types in combination is to use LiPF 6 and LiBF 4 in combination. In this case, the ratio of LiBF 4 to the total of both is preferably 0.01% by mass to 20% by mass, particularly preferably 0.1% by mass to 5% by mass. %.
此外,另外一个例子为组合使用无机氟化物盐与全氟链烷磺酰亚胺盐,此时,无机氟化物盐占两者总计的比率优选为70质量%~99质量%,更优选为80质量%~98质量%。该两者的组合使用具有抑制因高温保存引起的劣化的效果。In addition, another example is the combined use of an inorganic fluoride salt and a perfluoroalkanesulfonylimide salt. At this time, the ratio of the inorganic fluoride salt to the total of the two is preferably 70% by mass to 99% by mass, more preferably 80% by mass. % by mass to 98% by mass. The combined use of both has an effect of suppressing deterioration due to high-temperature storage.
非水电解液中的上述锂盐的浓度没有特别限制,但通常为0.5mol/L以上,优选为0.6mol/L以上,更优选为0.7mol/L以上。此外,其上限通常为2mol/L以下,优选为1.8mol/L以下,更优选为1.7mol/L以下。如果浓度过低,则非水电解液的电导率有时不充分,另一方面,如果浓度过高,则由于粘度升高,电导率有时降低,锂二次电池的性能有时降低。The concentration of the lithium salt in the non-aqueous electrolytic solution is not particularly limited, but is usually 0.5 mol/L or higher, preferably 0.6 mol/L or higher, more preferably 0.7 mol/L or higher. In addition, the upper limit thereof is usually 2 mol/L or less, preferably 1.8 mol/L or less, more preferably 1.7 mol/L or less. If the concentration is too low, the conductivity of the non-aqueous electrolytic solution may be insufficient. On the other hand, if the concentration is too high, the conductivity may decrease due to an increase in viscosity, and the performance of the lithium secondary battery may decrease.
[非水溶剂][Non-aqueous solvent]
作为非水溶剂,可以从以往的作为非水电解液的溶剂而提出的溶剂中适当选择使用。例如可以举出下述非水溶剂。As the non-aqueous solvent, it can be appropriately selected and used from those conventionally proposed as solvents for non-aqueous electrolytic solutions. For example, the following non-aqueous solvents are mentioned.
1)环状碳酸酯:1) Cyclic carbonate:
构成环状碳酸酯的亚烷基的碳原子数优选为2~6,特别优选为2~4。具体地说,例如可以举出碳酸亚乙酯、碳酸亚丙酯、碳酸亚丁酯等。其中,优选为碳酸亚乙酯、碳酸亚丙酯。The number of carbon atoms of the alkylene group constituting the cyclic carbonate is preferably 2-6, particularly preferably 2-4. Specifically, ethylene carbonate, propylene carbonate, butylene carbonate, etc. are mentioned, for example. Among them, ethylene carbonate and propylene carbonate are preferable.
2)链状碳酸酯2) Chain carbonate
作为链状碳酸酯,优选为碳酸二烷基酯,构成碳酸二烷基酯的烷基的碳原子数分别优选为1~5,特别优选为1~4。具体地说,例如可以举出碳酸二甲酯、碳酸二乙酯、碳酸二正丙酯、碳酸甲乙酯、碳酸甲基正丙基酯、碳酸乙基正丙基酯等碳酸二烷基酯。其中,优选为碳酸二甲酯、碳酸二乙酯、碳酸甲乙酯。As the chain carbonate, dialkyl carbonate is preferable, and the number of carbon atoms of the alkyl group constituting the dialkyl carbonate is preferably 1-5, particularly preferably 1-4. Specifically, for example, dialkyl carbonates such as dimethyl carbonate, diethyl carbonate, di-n-propyl carbonate, ethyl methyl carbonate, methyl-n-propyl carbonate, ethyl-n-propyl carbonate, etc. . Among them, dimethyl carbonate, diethyl carbonate, and ethyl methyl carbonate are preferable.
3)环状酯:3) Cyclic esters:
具体地说,例如可以举出γ-丁内酯、γ-戊内酯等。Specifically, γ-butyrolactone, γ-valerolactone, etc. are mentioned, for example.
4)链状酯:4) Chain esters:
具体地说,例如可以举出乙酸甲酯、乙酸乙酯、乙酸丙酯、丙酸甲酯等。Specifically, methyl acetate, ethyl acetate, propyl acetate, methyl propionate, etc. are mentioned, for example.
5)环状醚5) Cyclic ethers
具体地说,例如可以举出四氢呋喃、2-甲基四氢呋喃、四氢吡喃等。Specifically, tetrahydrofuran, 2-methyltetrahydrofuran, tetrahydropyran, etc. are mentioned, for example.
6)链状醚:6) Chain ethers:
具体地说,例如可以举出二甲氧基乙烷、二甲氧基甲烷等。Specifically, dimethoxyethane, dimethoxymethane, etc. are mentioned, for example.
7)含硫有机溶剂:7) Sulfur-containing organic solvents:
具体地说,例如可以举出环丁砜、二乙基砜等。Specifically, for example, sulfolane, diethylsulfone, etc. are mentioned.
它们可以单独使用,也可以组合使用2种以上,优选组合使用2种以上的化合物。例如,优选环状碳酸酯类或环状酯类等高介电常数的溶剂与链状碳酸酯类或链状酯类等低粘度溶剂组合使用。These may be used individually or in combination of 2 or more types, Preferably it is used in combination of 2 or more types. For example, it is preferable to use a solvent with a high dielectric constant such as cyclic carbonates or cyclic esters in combination with a low-viscosity solvent such as chain carbonates or chain esters.
非水溶剂的优选组合之一是以环状碳酸酯类和链状碳酸酯类为主体的组合。其中,环状碳酸酯类和链状碳酸酯类的总计在非水溶剂中占80容量%以上,优选85容量%以上,更优选90容量%以上。此外,相对于环状碳酸酯类和链状碳酸酯类的总计,环状碳酸酯类的容量为5%以上,优选为10%以上,更优选为15%以上,且通常为50%以下,优选为35%以下,更优选为30%以下。特别优选将碳酸酯类的总计在非水溶剂中所占的上述优选的容量范围、与环状碳酸酯类相对于环状和链状碳酸酯类的优选的上述容量范围组合。如果使用该非水溶剂的组合,则由于使用该组合而制造的电池的循环特性和高温保存特性(特别是高温保存后的残留容量和高负荷放电容量)的平衡变得优异,所以优选。One of the preferred combinations of non-aqueous solvents is a combination mainly composed of cyclic carbonates and chain carbonates. Among them, the total of cyclic carbonates and chain carbonates accounts for 80% by volume or more, preferably 85% by volume or more, more preferably 90% by volume or more in the non-aqueous solvent. In addition, the capacity of cyclic carbonates is 5% or more, preferably 10% or more, more preferably 15% or more, and usually 50% or less with respect to the total of cyclic carbonates and chain carbonates, Preferably it is 35% or less, More preferably, it is 30% or less. It is particularly preferred to combine the above-mentioned preferred volume ranges of the total carbonates in the non-aqueous solvent with the above-mentioned preferred volume ranges of cyclic carbonates with respect to cyclic and chain carbonates. Use of this combination of non-aqueous solvents is preferred because the balance between cycle characteristics and high-temperature storage characteristics (especially residual capacity after high-temperature storage and high-load discharge capacity) of a battery manufactured using the combination becomes excellent.
在该混合溶剂中含有选自锂盐和上述通式(1)所示的环状硅氧烷化合物、上述通式(2)所示的氟硅烷化合物、上述通式(3)所示的化合物、分子内具有S-F键的化合物、硝酸盐、亚硝酸盐、单氟磷酸盐、二氟磷酸盐、乙酸盐和丙酸盐中的至少一种化合物的非水电解液,由于使用该非水电解液制造的电池的循环特性和高温保存特性(特别是高温保存后的残留容量和高负荷放电容量)以及抑制气体产生的平衡变得优异,所以优选。The mixed solvent contains lithium salts and cyclic siloxane compounds represented by the above general formula (1), fluorosilane compounds represented by the above general formula (2), and compounds represented by the above general formula (3) , a compound having an S-F bond in the molecule, a non-aqueous electrolyte of at least one compound in nitrate, nitrite, monofluorophosphate, difluorophosphate, acetate and propionate, due to the use of the non-aqueous A battery made of an electrolyte solution is preferable because it is excellent in balance between cycle characteristics, high-temperature storage characteristics (particularly, residual capacity after high-temperature storage and high-load discharge capacity), and suppression of gas generation.
作为环状碳酸酯类和链状碳酸酯类的优选组合的具体例子,可以举出碳酸亚乙酯和碳酸二甲酯,碳酸亚乙酯和碳酸二乙酯,碳酸亚乙酯和碳酸甲乙酯,碳酸亚乙酯、碳酸二甲酯和碳酸二乙酯,碳酸亚乙酯、碳酸二甲酯和碳酸甲乙酯,碳酸亚乙酯、碳酸二乙酯和碳酸甲乙酯,碳酸亚乙酯、碳酸二甲酯、碳酸二乙酯和碳酸甲乙酯等。可以举出在这些碳酸亚乙酯和链状碳酸酯的组合中进一步加入了碳酸亚丙酯的组合作为优选的组合。含有碳酸亚丙酯时,碳酸亚乙酯和碳酸亚丙酯的容量比通常为99∶1~40∶60,优选为95∶5~50∶50。Specific examples of preferred combinations of cyclic carbonates and chain carbonates include ethylene carbonate and dimethyl carbonate, ethylene carbonate and diethyl carbonate, ethylene carbonate and methyl ethyl carbonate Ethylene carbonate, dimethyl carbonate and diethyl carbonate, ethylene carbonate, dimethyl carbonate and ethyl methyl carbonate, ethylene carbonate, diethyl carbonate and ethyl methyl carbonate, ethylene carbonate esters, dimethyl carbonate, diethyl carbonate and ethyl methyl carbonate, etc. A combination in which propylene carbonate is further added to these combinations of ethylene carbonate and chain carbonate is mentioned as a preferable combination. When propylene carbonate is contained, the capacity ratio of ethylene carbonate and propylene carbonate is usually 99:1 to 40:60, preferably 95:5 to 50:50.
进一步地,如果使碳酸亚丙酯占全部非水溶剂的量为0.1容量%以上、优选为1容量%以上、更优选为2容量%以上,且通常为10容量%以下、优选为8容量%以下、更优选为5容量%以下,则由于可以在维持碳酸亚乙酯和链状碳酸酯类的组合的特性的基础上,低温特性更优异,所以优选。Further, if the amount of propylene carbonate accounting for the total non-aqueous solvent is more than 0.1% by volume, preferably more than 1% by volume, more preferably more than 2% by volume, and usually less than 10% by volume, preferably 8% by volume It is less than, more preferably 5% by volume or less, since it is possible to maintain the characteristics of the combination of ethylene carbonate and chain carbonates, and the low-temperature characteristics are more excellent, so it is preferable.
其中,进一步优选含有非对称链状碳酸酯类,特别是含有碳酸亚乙酯、碳酸二甲酯和碳酸甲乙酯,碳酸亚乙酯、碳酸二乙酯和碳酸甲乙酯,碳酸亚乙酯、碳酸二甲酯、碳酸二乙酯和碳酸甲乙酯等碳酸亚乙酯、对称链状碳酸酯类和非对称链状碳酸酯类时,由于循环特性和大电流放电特性的平衡优异,所以优选。其中,优选非对称链状碳酸酯类为碳酸甲乙酯,此外,构成碳酸二烷基酯的烷基的碳原子数优选为1~2。Among them, it is further preferred to contain asymmetric chain carbonates, especially ethylene carbonate, dimethyl carbonate and ethyl methyl carbonate, ethylene carbonate, diethyl carbonate and ethyl methyl carbonate, ethylene carbonate , dimethyl carbonate, diethyl carbonate, ethyl methyl carbonate and other ethylene carbonate, symmetrical chain carbonates and asymmetric chain carbonates, because the balance between cycle characteristics and large current discharge characteristics is excellent, so preferred. Among them, the asymmetric chain carbonates are preferably ethyl methyl carbonate, and the number of carbon atoms of the alkyl group constituting the dialkyl carbonate is preferably 1-2.
优选的非水溶剂的其它例子是含有链状酯的非水溶剂。作为链状酯,特别优选为乙酸甲酯、乙酸乙酯等。链状酯在非水溶剂中所占的容量通常为5%以上、优选为8%以上、更优选为15%以上,且通常为50%以下、优选为35%以下、更优选为30%以下、进一步优选为25%以下。特别是,从提高电池的低温特性方面考虑,优选在上述环状碳酸酯类和链状碳酸酯类的混合溶剂中含有链状酯。Other examples of preferred non-aqueous solvents are non-aqueous solvents containing chain esters. As a chain ester, methyl acetate, ethyl acetate, etc. are especially preferable. The capacity of the chain ester in the non-aqueous solvent is usually 5% or more, preferably 8% or more, more preferably 15% or more, and usually 50% or less, preferably 35% or less, more preferably 30% or less , More preferably 25% or less. In particular, it is preferable to contain a chain ester in the mixed solvent of the above-mentioned cyclic carbonates and chain carbonates from the viewpoint of improving the low-temperature characteristics of the battery.
其它优选的非水溶剂的例子,为选自碳酸亚乙酯、碳酸亚丙酯、γ-丁内酯和γ-戊内酯中的一种有机溶剂,或含有选自该组中的2种以上有机溶剂的混合溶剂占全部溶剂的60容量%以上的非水溶剂。这样的混合溶剂优选闪点为50℃以上,其中,特别优选闪点为70℃以上。使用该溶剂的非水电解液即使在高温下使用,溶剂的蒸发或漏液也减少。其中,如果使用γ-丁内酯在非水溶剂中所占的量为60容量%以上的溶剂,碳酸亚乙酯和γ-丁内酯的总计在非水溶剂中占80容量%以上、优选为90容量%以上且碳酸亚乙酯和γ-丁内酯的容量比为5∶95~45∶55的溶剂,或碳酸亚乙酯和碳酸亚丙酯的总计在非水溶剂中占80容量%以上、优选为90容量%以上且碳酸亚乙酯和碳酸亚丙酯的容量比为30∶70~60∶40的溶剂,则一般循环特性和大电流放电特性等的平衡变得优异。Examples of other preferred non-aqueous solvents are an organic solvent selected from ethylene carbonate, propylene carbonate, γ-butyrolactone and γ-valerolactone, or containing 2 selected from this group A non-aqueous solvent in which the mixed solvent of the above organic solvents accounts for more than 60% by volume of the total solvents. Such a mixed solvent preferably has a flash point of 50°C or higher, and particularly preferably a flash point of 70°C or higher. Even if the nonaqueous electrolytic solution using this solvent is used at a high temperature, evaporation of the solvent and leakage are reduced. Wherein, if the amount of γ-butyrolactone in the non-aqueous solvent is 60% by volume or more, the total of ethylene carbonate and γ-butyrolactone accounts for 80% by volume or more in the non-aqueous solvent, preferably A solvent that is more than 90% by volume and has a volume ratio of ethylene carbonate to γ-butyrolactone of 5:95 to 45:55, or the total of ethylene carbonate and propylene carbonate accounts for 80% by volume in a non-aqueous solvent % or more, preferably 90% by volume or more, and a solvent in which the volume ratio of ethylene carbonate and propylene carbonate is 30:70 to 60:40, the balance between general cycle characteristics and large current discharge characteristics becomes excellent.
[特定化合物][specific compound]
本发明的非水电解液,如上所述,其特征在于,含有或添加选自通式(1)所示的环状硅氧烷化合物、通式(2)所示的氟硅烷化合物、通式(3)所示的化合物、分子内具有S-F键的化合物、硝酸盐、亚硝酸盐、单氟磷酸盐、二氟磷酸盐、乙酸盐和丙酸盐中的至少一种化合物(有时将它们简称为“特定化合物”)。The non-aqueous electrolytic solution of the present invention, as described above, is characterized in that it contains or adds a cyclic siloxane compound represented by the general formula (1), a fluorosilane compound represented by the general formula (2), a fluorosilane compound represented by the general formula Compounds shown in (3), compounds having an S-F bond in the molecule, at least one compound of nitrates, nitrites, monofluorophosphates, difluorophosphates, acetates, and propionates (sometimes they are referred to as "Specific Compound").
[[通式(1)所示的环状硅氧烷化合物]][[Cyclic siloxane compound represented by general formula (1)]]
通式(1)所示的环状硅氧烷化合物中的R1和R2为可以彼此相同或不同的碳原子数1~12的有机基团,作为R1和R2,可以举出甲基、乙基、正丙基、异丙基、丁基、异丁基、仲丁基、叔丁基等链状烷基;环己基、降冰片烷基等环状烷基;乙烯基、1-丙烯基、烯丙基、丁烯基、1,3-丁二烯基等链烯基;乙炔基、丙炔基、丁炔基等炔基;三氟甲基等卤代烷基;3-吡咯烷并丙基等具有饱和杂环基的烷基;可以具有烷基取代基的苯基等芳基;苯基甲基、苯基乙基等芳烷基;三甲基甲硅烷基等三烷基甲硅烷基;三甲基硅氧烷基等三烷基硅氧烷基等。R 1 and R 2 in the cyclic siloxane compound represented by the general formula (1) are organic groups having 1 to 12 carbon atoms which may be the same or different from each other, and examples of R 1 and R 2 include methyl Chain alkyl such as radical, ethyl, n-propyl, isopropyl, butyl, isobutyl, sec-butyl, tert-butyl; cyclic alkyl such as cyclohexyl, norbornyl; vinyl, 1 -alkenyl such as propenyl, allyl, butenyl, 1,3-butadienyl; alkynyl such as ethynyl, propynyl, butynyl; haloalkyl such as trifluoromethyl; 3-pyrrole Alkyl groups with saturated heterocyclic groups such as alkanopropyl groups; aryl groups such as phenyl groups that may have alkyl substituents; aralkyl groups such as phenylmethyl and phenylethyl groups; trialkyl groups such as trimethylsilyl groups silyl group; trimethylsiloxane group and other trialkylsiloxane groups, etc.
其中,碳原子数少的基团易表现出特性,优选为碳原子数1~6的有机基团。此外,链烯基由于作用于非水电解液或电极表面的涂膜而提高输出功率特性,芳基由于捕捉充放电时在电池内产生的自由基而具有提高电池整体性能的作用,所以优选。因此,作为R1和R2,特别优选为甲基、乙烯基或苯基。Among them, a group having a small number of carbon atoms tends to exhibit characteristics, and is preferably an organic group having 1 to 6 carbon atoms. In addition, the alkenyl group improves the output power characteristics by acting on the non-aqueous electrolyte or the coating film on the electrode surface, and the aryl group has the effect of improving the overall performance of the battery by capturing the free radicals generated in the battery during charge and discharge, so it is preferred. Therefore, R 1 and R 2 are particularly preferably methyl, vinyl or phenyl.
通式(1)中,n表示3~10的整数,优选为3~6的整数、特别优选为3或4。In General formula (1), n represents the integer of 3-10, Preferably it is an integer of 3-6, Especially preferably, it is 3 or 4.
作为通式(1)所示的环状硅氧烷化合物的例子,例如可以举出六甲基环三硅氧烷、六乙基环三硅氧烷、六苯基环三硅氧烷、1,3,5-三甲基-1,3,5-三乙烯基环三硅氧烷等环三硅氧烷;八甲基环四硅氧烷等环四硅氧烷;十甲基环五硅氧烷等环五硅氧烷等。其中,特别优选环三硅氧烷。Examples of the cyclic siloxane compound represented by the general formula (1) include hexamethylcyclotrisiloxane, hexaethylcyclotrisiloxane, hexaphenylcyclotrisiloxane, 1 , 3,5-trimethyl-1,3,5-trivinylcyclotrisiloxane and other cyclotrisiloxanes; octamethylcyclotetrasiloxane and other cyclotetrasiloxanes; decamethylcyclopentasiloxane and other Cyclopentasiloxane, such as siloxane, etc. Among them, cyclotrisiloxane is particularly preferable.
[[通式(2)所示的氟硅烷化合物]][[Fluorosilane compound represented by general formula (2)]]
通式(2)所示的氟硅烷化合物中的R3~R5为可以彼此相同或不同的碳原子数为1~12的有机基团,除了作为通式(1)中的R1和R2的例子举出的链状烷基、环状烷基、链烯基、炔基、卤代烷基、具有饱和杂环基的烷基、可以具有烷基的苯基等芳基、芳烷基、三烷基甲硅烷基、三烷基硅氧烷基之外,还可以举出乙氧羰基乙基等羰基;乙酰氧基、乙酰氧基甲基、三氟乙酰氧基等羧基;甲氧基、乙氧基、丙氧基、丁氧基、苯氧基、烯丙氧基等氧基;烯丙基氨基等氨基;苄基等。R 3 to R 5 in the fluorosilane compound represented by the general formula (2) are organic groups with 1 to 12 carbon atoms that may be the same or different from each other, except for R 1 and R in the general formula (1) Examples of 2 include chain alkyl, cyclic alkyl, alkenyl, alkynyl, haloalkyl, alkyl having a saturated heterocyclic group, aryl such as phenyl that may have an alkyl, aralkyl, In addition to trialkylsilyl and trialkylsiloxyl groups, carbonyl groups such as ethoxycarbonylethyl; carboxyl groups such as acetoxy, acetoxymethyl, and trifluoroacetoxy; methoxy , ethoxy, propoxy, butoxy, phenoxy, allyloxy and other oxygen groups; allylamino and other amino groups; benzyl and the like.
通式(2)中,x表示1~3的整数,p、q和r分别表示0~3的整数,且1≤p+q+r≤3。此外,必然地,x+p+q+r=4。In general formula (2), x represents the integer of 1-3, p, q, and r represent the integer of 0-3 respectively, and 1≤p+q+r≤3. Also, x+p+q+r=4 necessarily.
作为通式(2)所示的氟硅烷化合物的例子,可以举出三甲基氟硅烷、三乙基氟硅烷、三丙基氟硅烷、苯基二甲基氟硅烷、三苯基氟硅烷、乙烯基二甲基氟硅烷、乙烯基二乙基氟硅烷、乙烯基二苯基氟硅烷、三甲氧基氟硅烷、三乙氧基氟硅烷等单氟硅烷类,以及二甲基二氟硅烷、二乙基二氟硅烷、二乙烯基二氟硅烷、乙基乙烯基二氟硅烷等二氟硅烷类;甲基三氟硅烷、乙基三氟硅烷等三氟硅烷类。Examples of the fluorosilane compound represented by the general formula (2) include trimethylfluorosilane, triethylfluorosilane, tripropylfluorosilane, phenyldimethylfluorosilane, triphenylfluorosilane, Vinyldimethylfluorosilane, vinyldiethylfluorosilane, vinyldiphenylfluorosilane, trimethoxyfluorosilane, triethoxyfluorosilane and other monofluorosilanes, as well as dimethyldifluorosilane, Difluorosilanes such as diethyldifluorosilane, divinyldifluorosilane, and ethylvinyldifluorosilane; trifluorosilanes such as methyltrifluorosilane and ethyltrifluorosilane.
通式(2)所示的氟硅烷化合物如果沸点低,则由于挥发有时难以在非水电解液中含有规定的量。此外,含有在非水电解液中后,在由于充放电引起的电池的发热或外部环境为高温等条件下有可能挥发。因此,优选在1大气压下具有50℃以上沸点的化合物,其中特别优选具有60℃以上沸点的化合物。When the fluorosilane compound represented by the general formula (2) has a low boiling point, it may be difficult to contain a predetermined amount in the non-aqueous electrolytic solution due to volatilization. In addition, after being contained in the non-aqueous electrolytic solution, it may volatilize under conditions such as heat generation of the battery due to charge and discharge, or high temperature of the external environment. Therefore, compounds having a boiling point of 50°C or higher at 1 atmosphere are preferred, and compounds having a boiling point of 60°C or higher are particularly preferred.
此外,与通式(1)的化合物同样地,作为有机基团,碳原子数少的基团易表现出效果,碳原子数为1~6的链烯基作用于非水电解液或电极表面的涂膜而提高输出功率特性,芳基捕捉充放电时在电池内产生的自由基而具有提高电池整体性能的作用。因此,从该观点来看,作为有机基团,优选为甲基、乙烯基或苯基,作为化合物的例子,特别优选为三甲基氟硅烷、乙烯基二甲基氟硅烷、苯基二甲基氟硅烷、乙烯基二苯基氟硅烷等。In addition, like the compound of general formula (1), as an organic group, a group with a small number of carbon atoms is likely to exhibit an effect, and an alkenyl group with 1 to 6 carbon atoms acts on the surface of a non-aqueous electrolyte or an electrode. The coating film can improve the output power characteristics, and the aryl group can capture the free radicals generated in the battery during charging and discharging to improve the overall performance of the battery. Therefore, from this point of view, the organic group is preferably a methyl group, vinyl group, or phenyl group, and examples of the compound are particularly preferably trimethylfluorosilane, vinyldimethylfluorosilane, phenyldimethylsilane, or phenyldimethylsilane. Fluorosilane, vinyl diphenyl fluorosilane, etc.
[[通式(3)所示的化合物]][[Compound represented by general formula (3)]]
通式(3)所示的化合物中的R6~R8为可以彼此相同或不同的碳原子数1~12的有机基团,作为其例子,可以同样地举出作为通式(2)中的R3~R5的例子而举出的链状烷基、环状烷基、链烯基、炔基、卤代烷基、具有饱和杂环基的烷基、可以具有烷基的苯基等芳基、芳烷基、三烷基甲硅烷基、三烷基硅氧烷基、羰基、羧基、氧基、氨基、苄基等。R 6 to R 8 in the compound represented by the general formula (3) are organic groups having 1 to 12 carbon atoms which may be the same or different from each other, and examples thereof include the same as in the general formula (2) Examples of R 3 to R 5 include chain alkyl, cyclic alkyl, alkenyl, alkynyl, haloalkyl, alkyl with saturated heterocyclic group, aromatic group such as phenyl that may have alkyl, etc. group, aralkyl group, trialkylsilyl group, trialkylsiloxy group, carbonyl group, carboxyl group, oxy group, amino group, benzyl group, etc.
通式(3)所示的化合物中的A,只要是由H、C、N、O、F、S、Si和/或P构成的基团即可,没有特别限制,作为通式(3)中的与氧原子直接键合的元素,优选为C、S、Si或P。作为这些原子的存在方式,优选例如存在于链状烷基、环状烷基、链烯基、炔基、卤代烷基、羰基、磺酰基、三烷基甲硅烷基、磷酰基、氧膦基等中。A in the compound represented by the general formula (3) is not particularly limited as long as it is a group consisting of H, C, N, O, F, S, Si and/or P. As the general formula (3) The element directly bonded to the oxygen atom in is preferably C, S, Si or P. The presence of these atoms is preferably, for example, a chain alkyl group, a cyclic alkyl group, an alkenyl group, an alkynyl group, a haloalkyl group, a carbonyl group, a sulfonyl group, a trialkylsilyl group, a phosphoryl group, a phosphinyl group, and the like. middle.
此外,通式(3)所示的化合物的分子量优选为1000以下,其中特别优选为800以下,更优选为500以下。作为通式(3)所示的化合物的例子,可以举出六甲基二硅氧烷、1,3-二乙基四甲基二硅氧烷、六乙基二硅氧烷、八甲基三硅氧烷等硅氧烷化合物类;甲氧基三甲基硅烷、乙氧基三甲基硅烷等烷氧基硅烷类;过氧化双(三甲基甲硅烷基)等过氧化物类;乙酸三甲基甲硅烷基酯、乙酸三乙基甲硅烷基酯、丙酸三甲基甲硅烷基酯、甲基丙烯酸三甲基甲硅烷基酯、三氟乙酸三甲基甲硅烷基酯等羧酸酯类;甲磺酸三甲基甲硅烷基酯、乙磺酸三甲基甲硅烷基酯、甲磺酸三乙基甲硅烷基酯、氟甲磺酸三甲基甲硅烷基酯等磺酸酯类;双(三甲基甲硅烷基)硫酸酯等硫酸酯类;三(三甲基甲硅烷氧基)硼等硼酸酯类;三(三甲基甲硅烷基)磷酸酯、三(三甲基甲硅烷基)亚磷酸酯等磷酸或亚磷酸酯类等。In addition, the molecular weight of the compound represented by the general formula (3) is preferably 1000 or less, particularly preferably 800 or less, more preferably 500 or less. Examples of compounds represented by the general formula (3) include hexamethyldisiloxane, 1,3-diethyltetramethyldisiloxane, hexaethyldisiloxane, octamethyldisiloxane, Silicone compounds such as trisiloxane; alkoxysilanes such as methoxytrimethylsilane and ethoxytrimethylsilane; peroxides such as bis(trimethylsilyl) peroxide; Trimethylsilyl acetate, triethylsilyl acetate, trimethylsilyl propionate, trimethylsilyl methacrylate, trimethylsilyl trifluoroacetate, etc. Carboxylic acid esters; trimethylsilyl methanesulfonate, trimethylsilyl ethanesulfonate, triethylsilyl methanesulfonate, trimethylsilyl fluoromethanesulfonate, etc. Sulfonate esters; bis(trimethylsilyl)sulfate and other sulfate esters; tris(trimethylsilyloxy)boron and other borate esters; tris(trimethylsilyl)phosphate, tri Phosphoric acid such as (trimethylsilyl) phosphite or phosphite, etc.
其中,优选硅氧烷化合物类、磺酸酯类、硫酸酯类,特别优选磺酸酯类。作为硅氧烷化合物类,优选六甲基二硅氧烷;作为磺酸酯类,优选甲磺酸三甲基甲硅烷基酯;作为硫酸酯类,优选双(三甲基甲硅烷基)硫酸酯。Among them, silicone compounds, sulfonate esters, and sulfate esters are preferable, and sulfonate esters are particularly preferable. As the siloxane compound, hexamethyldisiloxane is preferred; as the sulfonate, trimethylsilyl methanesulfonate is preferred; as the sulfuric acid ester, bis(trimethylsilyl)sulfuric acid is preferred. ester.
[[分子内具有S-F键的化合物]][[Compounds with S-F bonds in the molecule]]
作为分子内具有S-F键的化合物,没有特别限定,但优选为磺酰氟类、氟磺酸酯类。The compound having an S-F bond in the molecule is not particularly limited, but sulfonyl fluorides and fluorosulfonate esters are preferable.
例如可以举出,甲磺酰氟、乙磺酰氟、甲烷双(磺酰氟)、乙烷-1,2-双(磺酰氟)、丙烷-1,3-双(磺酰氟)、丁烷-1,4-双(磺酰氟)、二氟甲烷双(磺酰氟)、1,1,2,2-四氟乙烷-1,2-双(磺酰氟)、1,1,2,2,3,3-六氟丙烷-1,3-双(磺酰氟)、氟磺酸甲酯、氟磺酸乙酯等。其中,优选甲磺酰氟、甲烷双(磺酰氟)或氟磺酸甲酯。For example, methanesulfonyl fluoride, ethanesulfonyl fluoride, methanebis(sulfonyl fluoride), ethane-1,2-bis(sulfonyl fluoride), propane-1,3-bis(sulfonyl fluoride), Butane-1,4-bis(sulfonyl fluoride), difluoromethane bis(sulfonyl fluoride), 1,1,2,2-tetrafluoroethane-1,2-bis(sulfonyl fluoride), 1, 1,2,2,3,3-hexafluoropropane-1,3-bis(sulfonyl fluoride), methyl fluorosulfonate, ethyl fluorosulfonate, etc. Among them, methanesulfonyl fluoride, methanebis(sulfonyl fluoride) or methyl fluorosulfonate is preferable.
[[硝酸盐、亚硝酸盐、单氟磷酸盐、二氟磷酸盐、乙酸盐、丙酸盐]][[Nitrate, Nitrite, Monofluorophosphate, Difluorophosphate, Acetate, Propionate]]
作为硝酸盐、亚硝酸盐、单氟磷酸盐、二氟磷酸盐、乙酸盐、丙酸盐的反阳离子,没有特别限定,除了Li、Na、K、Mg、Ca、Fe、Cu等金属元素外,还可以举出以NR9R10R11R12(式中,R9~R12各自独立地表示氢原子或碳原子数为1~12的有机基团)表示的铵、季铵。其中,作为R9~R12的碳原子数为1~12的有机基团,可以举出可以被卤原子取代的烷基、可以被卤原子取代的环烷基、可以被卤原子取代的芳基、含氮原子的杂环基等。作为R9~R12,分别优选为氢原子、烷基、环烷基、含氮原子的杂环基等。这些反阳离子中,从在锂二次电池中使用时的电池特性方面考虑,优选为锂、钠、钾、镁、钙或NR9R10R11R12,特别优选为锂。此外,其中,从输出功率提高率或循环特性方面考虑,优选为硝酸盐或二氟磷酸盐,特别优选为二氟磷酸锂。此外,这些化合物实质上可以直接使用在非水溶剂中合成而得到的化合物,也可以向非水溶剂中添加另外合成并实质上被分离的化合物。As the counter cation of nitrate, nitrite, monofluorophosphate, difluorophosphate, acetate, propionate, there is no particular limitation, except metal elements such as Li, Na, K, Mg, Ca, Fe, Cu, etc. In addition, ammonium and quaternary ammonium represented by NR 9 R 10 R 11 R 12 (in the formula, R 9 to R 12 each independently represent a hydrogen atom or an organic group having 1 to 12 carbon atoms) can also be mentioned. Among them, as the organic groups with 1 to 12 carbon atoms of R 9 to R 12 , there may be mentioned alkyl groups which may be substituted by halogen atoms, cycloalkyl groups which may be substituted by halogen atoms, aromatic groups which may be substituted by halogen atoms, group, heterocyclic group containing nitrogen atom, etc. R 9 to R 12 are each preferably a hydrogen atom, an alkyl group, a cycloalkyl group, a nitrogen atom-containing heterocyclic group, or the like. Among these counter cations, lithium, sodium, potassium, magnesium, calcium, or NR 9 R 10 R 11 R 12 are preferred from the viewpoint of battery characteristics when used in a lithium secondary battery, and lithium is particularly preferred. Among them, nitrates or difluorophosphates are preferable, and lithium difluorophosphate is particularly preferable from the standpoint of output power improvement rate and cycle characteristics. In addition, these compounds may be used substantially as they are synthesized in a non-aqueous solvent, or may be added to a non-aqueous solvent that is synthesized separately and substantially isolated.
特定化合物,即通式(1)所示的环状硅氧烷化合物、通式(2)所示的氟硅烷化合物、通式(3)所示的化合物、分子内具有S-F键的化合物、硝酸盐、亚硝酸盐、单氟磷酸盐、二氟磷酸盐、乙酸盐或丙酸盐可以单独使用一种,也可以以任意的组合和比例组合使用2种以上的化合物。此外,特定化合物中,即使是上述分别分类的化合物中,也可以单独使用一种,或者以任意的组合和比例组合使用2种以上的化合物。Specific compounds, namely cyclic siloxane compounds represented by general formula (1), fluorosilane compounds represented by general formula (2), compounds represented by general formula (3), compounds with S-F bonds in their molecules, nitric acid Salts, nitrites, monofluorophosphates, difluorophosphates, acetates, or propionates may be used alone, or two or more compounds may be used in combination in any combination and ratio. In addition, among the specific compounds, even among the above-mentioned separately classified compounds, one kind may be used alone, or two or more kinds of compounds may be used in combination in any combination and ratio.
非水电解液中的这些特定化合物的比率,相对于全部非水电解液,以总量计,必须为10ppm以上(0.001质量%以上),优选为0.01质量%以上,更优选为0.05质量%以上,进一步优选为0.1质量%以上。此外,上限优选为5质量%以下,更优选为4质量%以下,进一步优选为3质量%以下。如果特定化合物的浓度过低,则有时难以得到电池输出功率的提高效果或电池寿命的延长效果,另一方面,如果浓度过高,则有时导致充放电效率的降低。The ratio of these specific compounds in the nonaqueous electrolytic solution must be 10 ppm or more (0.001 mass % or more) in total with respect to the entire nonaqueous electrolytic solution, preferably 0.01 mass % or more, more preferably 0.05 mass % or more , and more preferably 0.1% by mass or more. In addition, the upper limit is preferably 5% by mass or less, more preferably 4% by mass or less, and still more preferably 3% by mass or less. If the concentration of the specific compound is too low, it may be difficult to improve the battery output or to extend the battery life. On the other hand, if the concentration is too high, the charge and discharge efficiency may decrease.
此外,这些特定化合物如果作为非水电解液实际供于二次电池制造中,则拆卸该电池再次取出非水电解液,其中的含量大多显著降低。因此,由从电池中抽出的非水电解液中可以至少检测出上述特定化合物也在本发明的范围内。本发明涉及的非水电解液可以通过在非水溶剂中溶解电解质锂盐、特定的化合物和根据需要的其它化合物来进行制备。制备非水电解液时,优选预先对各原料进行脱水,使水分含量通常为50ppm以下、优选为30ppm以下、特别优选为10ppm以下。In addition, when these specific compounds are actually used in the manufacture of secondary batteries as non-aqueous electrolytes, the contents of the non-aqueous electrolytes are often significantly reduced when the batteries are disassembled and taken out again. Therefore, it is within the scope of the present invention that at least the above-mentioned specific compound can be detected from the non-aqueous electrolytic solution extracted from the battery. The non-aqueous electrolytic solution related to the present invention can be prepared by dissolving an electrolyte lithium salt, a specific compound, and other compounds as needed in a non-aqueous solvent. When preparing a non-aqueous electrolyte, it is preferable to dehydrate each raw material in advance so that the moisture content is usually 50 ppm or less, preferably 30 ppm or less, particularly preferably 10 ppm or less.
[其它化合物][Other compounds]
本发明中的非水电解液在非水溶剂中含有作为电解质的锂盐和特定化合物作为必须成分,根据需要,在不损害本发明效果的范围内还可以含有任意量的其它化合物。作为该其它化合物,具体地说,可以举出如下物质,例如:The non-aqueous electrolytic solution in the present invention contains a lithium salt as an electrolyte and a specific compound as essential components in a non-aqueous solvent, and may contain any amount of other compounds as needed within the range not impairing the effect of the present invention. As the other compound, specifically, the following substances can be mentioned, for example:
(1)联苯、烷基联苯、三联苯、三联苯的部分氢化物、环己基苯、叔丁基苯、叔戊基苯、二苯基醚、二苯并呋喃等芳香族化合物;2-氟联苯、邻环己基氟苯、对环己基氟苯等上述芳香族化合物的部分氟化物;2,4-二氟苯甲醚、2,5-二氟苯甲醚、2,6-二氟苯甲醚、3,5-二氟苯甲醚等含氟苯甲醚化合物等过充电防止剂;(1) Aromatic compounds such as biphenyl, alkyl biphenyl, terphenyl, partially hydrogenated terphenyl, cyclohexylbenzene, tert-butylbenzene, tert-amylbenzene, diphenyl ether, and dibenzofuran; 2 - Partial fluorides of the above-mentioned aromatic compounds such as fluorobiphenyl, o-cyclohexylfluorobenzene, p-cyclohexylfluorobenzene; 2,4-difluoroanisole, 2,5-difluoroanisole, 2,6- Anti-overcharge agents such as difluoroanisole, 3,5-difluoroanisole and other fluorine-containing anisole compounds;
(2)碳酸亚乙烯酯、乙烯基碳酸亚乙酯、氟碳酸亚乙酯、三氟碳酸亚丙酯、琥珀酸酐、戊二酸酐、马来酸酐、柠康酸酐、戊烯二酸酐、衣康酸酐、环己烷二羧酸酐等负极涂膜形成剂;(2) Vinylene carbonate, vinyl ethylene carbonate, fluoroethylene carbonate, trifluoropropylene carbonate, succinic anhydride, glutaric anhydride, maleic anhydride, citraconic anhydride, glutaconic anhydride, itaconic Anode coating film forming agents such as acid anhydride and cyclohexanedicarboxylic anhydride;
(3)亚硫酸亚乙酯、亚硫酸亚丙酯、亚硫酸二甲酯、丙磺酸内酯、丁磺酸内酯、甲磺酸甲酯、二甲磺酸丁酯、甲苯磺酸甲酯、硫酸二甲酯、硫酸亚乙酯、环丁砜、二甲基砜、二乙基砜、二甲基亚砜、二乙基亚砜、四亚甲基亚砜、二苯硫醚、茴香硫醚、二苯基二硫醚、二吡啶二硫醚等正极保护剂等。(3) Ethylene sulfite, propylene sulfite, dimethyl sulfite, propane sultone, butane sultone, methyl methanesulfonate, butyl dimesylate, methyl toluenesulfonate Ester, dimethyl sulfate, ethylene sulfate, sulfolane, dimethyl sulfone, diethyl sulfone, dimethyl sulfoxide, diethyl sulfoxide, tetramethylene sulfoxide, diphenyl sulfide, anise sulfur Ether, diphenyl disulfide, dipyridine disulfide and other positive electrode protective agents.
作为过充电防止剂,优选联苯、烷基联苯、三联苯、三联苯的部分氢化物、环己基苯、叔丁基苯、叔戊基苯、二苯基醚、二苯并呋喃等芳香族化合物。它们可以组合使用2种以上。组合使用2种以上时,特别优选环己基苯或三联苯(或其部分氢化物)和叔丁基苯或叔戊基苯组合使用。As the anti-overcharge agent, biphenyl, alkylbiphenyl, terphenyl, partial hydrogenated product of terphenyl, cyclohexylbenzene, tert-butylbenzene, tert-amylbenzene, diphenyl ether, dibenzofuran and other aromatic compounds are preferable. Family compounds. These can be used in combination of 2 or more types. When two or more are used in combination, it is particularly preferable to use cyclohexylbenzene or terphenyl (or a partially hydrogenated product thereof) in combination with tert-butylbenzene or tert-amylbenzene.
作为负极涂膜形成剂,优选碳酸亚乙烯酯、乙烯基碳酸亚乙酯、氟碳酸亚乙酯、琥珀酸酐、马来酸酐。它们可以组合使用2种以上。组合使用2种以上时,优选为碳酸亚乙烯酯和乙烯基碳酸亚乙酯、氟碳酸亚乙酯、琥珀酸酐或马来酸酐。作为正极保护剂,优选为亚硫酸亚乙酯、亚硫酸亚丙酯、丙磺酸内酯、丁磺酸内酯、甲磺酸甲酯、甲磺酸丁酯。它们可以组合使用2种以上。此外,特别优选将负极涂膜形成剂和正极保护剂组合使用,以及将过充电防止剂、负极涂膜形成剂和正极保护剂组合使用。As the negative electrode coating film forming agent, vinylene carbonate, vinyl ethylene carbonate, fluoroethylene carbonate, succinic anhydride, and maleic anhydride are preferable. These can be used in combination of 2 or more types. When two or more are used in combination, vinylene carbonate, vinyl ethylene carbonate, fluoroethylene carbonate, succinic anhydride, or maleic anhydride are preferable. As the positive electrode protective agent, ethylene sulfite, propylene sulfite, propane sultone, butane sultone, methyl methanesulfonate, and butyl methanesulfonate are preferable. These can be used in combination of 2 or more types. In addition, it is particularly preferable to use a negative electrode coating film forming agent and a positive electrode protective agent in combination, and to use an overcharge preventing agent, a negative electrode coating film forming agent and a positive electrode protective agent in combination.
对非水电解液中的这些其它化合物的含有比率没有特别限定,相对于全部非水电解液,分别优选为0.01质量%以上,更优选为0.1质量%,进一步优选为0.2质量%,其上限优选为5质量%以下,更优选为3质量%,进一步优选为2质量%以下。通过添加这些化合物,可以在由于过充电而引起异常时抑制电池的破裂/起火,或使高温保存后的容量保持特性、循环特性提高。The content ratio of these other compounds in the nonaqueous electrolytic solution is not particularly limited, and is preferably 0.01% by mass or more, more preferably 0.1% by mass, and even more preferably 0.2% by mass, with respect to the entire nonaqueous electrolytic solution, and the upper limit is preferably It is 5 mass % or less, More preferably, it is 3 mass %, More preferably, it is 2 mass % or less. By adding these compounds, when an abnormality occurs due to overcharging, it is possible to suppress rupture and fire of the battery, and to improve capacity retention characteristics and cycle characteristics after high-temperature storage.
对于本发明的二次电池用非水电解液的制备方法,没有特别限定,可以在非水溶剂中根据常规方法溶解锂盐、特定化合物、根据需要的其它化合物来制备。The method for preparing the non-aqueous electrolyte solution for secondary batteries of the present invention is not particularly limited, and can be prepared by dissolving lithium salts, specific compounds, and other compounds as needed in a non-aqueous solvent according to a conventional method.
<电解液[1]><Electrolyte[1]>
在上述非水电解液中,优选构成电解液的非水溶剂为至少含有“碳酸亚乙酯”的混合溶剂,碳酸亚乙酯相对于非水溶剂总量的比率为1容量%~25容量%(电解液[1])。In the above-mentioned non-aqueous electrolytic solution, it is preferable that the non-aqueous solvent constituting the electrolytic solution is a mixed solvent containing at least "ethylene carbonate", and the ratio of ethylene carbonate to the total amount of non-aqueous solvent is 1% by volume to 25% by volume (Electrolyte[1]).
本发明(电解液[1])中的“锂盐”、“碳酸亚乙酯(EC)以外的非水溶剂”、“特定化合物”、“其它化合物”的种类及其含量、使用条件、非水电解液的制备方法等与上述相同。Types and contents of "lithium salts", "non-aqueous solvents other than ethylene carbonate (EC)", "specific compounds" and "other compounds" in the present invention (electrolyte solution [1]), use conditions, non-aqueous solvents, etc. The preparation method and the like of the aqueous electrolytic solution are the same as described above.
<电解液[2]><Electrolyte[2]>
在上述非水电解液中,优选构成电解液的非水溶剂含有“至少一种的非对称链状碳酸酯”且该非对称链状碳酸酯在全部非水溶剂中所占的含有比率为5体积%~90体积%(电解液[2])。In the above-mentioned non-aqueous electrolytic solution, it is preferable that the non-aqueous solvent constituting the electrolytic solution contains "at least one asymmetric chain carbonate" and the content ratio of the asymmetric chain carbonate in all non-aqueous solvents is 5 Volume %~90 volume % (electrolyte solution [2]).
本发明(电解液[2])中的“锂盐”、“非对称链状碳酸酯以外的非水溶剂”、“特定化合物”、“其它化合物”的种类及其含量、使用条件、非水电解液的制备方法等与上述相同。The type and content of "lithium salt", "nonaqueous solvent other than asymmetric chain carbonate", "specific compound" and "other compound" in the present invention (electrolyte solution [2]), use conditions, nonaqueous The preparation method and the like of the electrolytic solution are the same as described above.
本发明(电解液[2])中,在全部非水溶剂中含有5体积%~90体积%的至少一种的非对称链状碳酸酯。进一步地,全部非水溶剂中的至少一种的非对称链状碳酸酯的含量优选为8体积%以上、更优选为10体积%以上、进一步优选为15体积%以上、特别优选20体积%以上,此外上限优选为85体积%以下、更优选为70体积%以下、进一步优选为60体积%以下、特别优选为45体积%以下,由于兼具本发明的高的低温特性和循环特性等而优选。In the present invention (electrolyte solution [2]), at least one asymmetric chain carbonate is contained in 5% by volume to 90% by volume in the total non-aqueous solvent. Further, the content of at least one asymmetric chain carbonate in all nonaqueous solvents is preferably 8% by volume or more, more preferably 10% by volume or more, further preferably 15% by volume or more, particularly preferably 20% by volume or more In addition, the upper limit is preferably 85% by volume or less, more preferably 70% by volume or less, even more preferably 60% by volume or less, and particularly preferably 45% by volume or less. It is preferred due to the high low-temperature characteristics and cycle characteristics of the present invention. .
对于非对称链状碳酸酯,没有特别限定,但优选为非对称碳酸烷基酯,优选烷基的碳原子数为1~4。作为这样的非对称碳酸烷基酯的具体例子,例如可以举出,碳酸甲乙酯、碳酸甲基正丙基酯、碳酸乙基正丙基酯、碳酸甲基异丙基酯、碳酸乙基异丙基酯、碳酸甲基正丁基酯、碳酸乙基正丁基酯等。其中,优选为碳酸甲乙酯、碳酸甲基正丙基酯、碳酸乙基正丙基酯、碳酸甲基正丁基酯、碳酸乙基正丁基酯,更优选为碳酸甲乙酯、碳酸甲基正丙基酯、碳酸甲基正丁基酯,特别优选为碳酸甲乙酯。这些非对称链状碳酸酯可以混合使用2种以上。The asymmetric chain carbonate is not particularly limited, but is preferably an asymmetric alkyl carbonate, and preferably the alkyl group has 1 to 4 carbon atoms. Specific examples of such asymmetric alkyl carbonates include, for example, ethyl methyl carbonate, methyl n-propyl carbonate, ethyl n-propyl carbonate, methyl isopropyl carbonate, ethyl carbonate Isopropyl ester, methyl n-butyl carbonate, ethyl n-butyl carbonate, etc. Among them, preferably ethyl methyl carbonate, methyl n-propyl carbonate, ethyl n-propyl carbonate, methyl n-butyl carbonate, ethyl n-butyl carbonate, more preferably methyl ethyl carbonate, carbonic acid Methyl-n-propyl ester, methyl-n-butyl carbonate, particularly preferably methyl ethyl carbonate. These asymmetric chain carbonates may be used in combination of two or more.
如上所述,非水溶剂可以混合使用2种以上。特别是从二次电池的循环特性、保存特性提高等方面考虑,优选除了该非对称链状碳酸酯之外,进一步含有至少一种的环状碳酸酯。含有环状碳酸酯时,环状碳酸酯在全部非水溶剂中所占的比率通常为5体积%以上、优选为10体积%以上、更优选为15体积%以上,此外上限通常为50体积%以下、优选为35体积%以下、更优选为30体积%以下、进一步优选为25体积%以下。如果环状碳酸酯在全部非水溶剂中所占的比率过少,则有可能得不到二次电池的循环特性或保存特性的提高,另一方面,如果过多,则低温放电特性有时降低。As mentioned above, the nonaqueous solvent can mix and use 2 or more types. In particular, it is preferable to further contain at least one kind of cyclic carbonate in addition to the asymmetric chain carbonate from the viewpoint of improving the cycle characteristics and storage characteristics of the secondary battery. When a cyclic carbonate is contained, the ratio of the cyclic carbonate to the total non-aqueous solvent is usually 5% by volume or more, preferably 10% by volume or more, more preferably 15% by volume or more, and the upper limit is usually 50% by volume or less, preferably 35% by volume or less, more preferably 30% by volume or less, even more preferably 25% by volume or less. If the ratio of the cyclic carbonate in all non-aqueous solvents is too small, the cycle characteristics and storage characteristics of the secondary battery may not be improved. On the other hand, if it is too large, the low-temperature discharge characteristics may sometimes decrease. .
此外,从二次电池的循环特性或保存特性和低温放电特性的平衡的提高等方面考虑,优选除了该非对称链状碳酸酯之外,进一步含有至少一种的对称链状碳酸酯。含有对称链状碳酸酯时,对称链状碳酸酯在全部非水溶剂中所占的比率通常为5体积%以上、优选为10体积%以上、更优选为15体积%以上,此外,上限通常为80体积%以下、优选为70体积%以下、更优选为50体积%以下、进一步优选为40体积%以下。如果对称链状碳酸酯在全部非水溶剂中所占的比率过少,则有时得不到循环特性或保存特性和低温放电特性的平衡的提高,另一方面,如果过多,则有时得不到优异的循环特性。In addition, it is preferable to further contain at least one kind of symmetrical chain carbonate in addition to the asymmetric chain carbonate from the viewpoint of improving the balance between cycle characteristics, storage characteristics, and low-temperature discharge characteristics of the secondary battery. When a symmetrical chain carbonate is contained, the proportion of the symmetrical chain carbonate in the total non-aqueous solvent is usually 5% by volume or more, preferably 10% by volume or more, more preferably 15% by volume or more, and the upper limit is usually 80 volume % or less, Preferably it is 70 volume % or less, More preferably, it is 50 volume % or less, More preferably, it is 40 volume % or less. If the proportion of the symmetrical chain carbonate in all non-aqueous solvents is too small, sometimes the cycle characteristics or the balance between storage characteristics and low-temperature discharge characteristics cannot be obtained. On the other hand, if it is too large, sometimes it cannot be achieved to excellent cycle characteristics.
此外,优选非对称链状碳酸酯与除此之外的2种以上的非水溶剂的混合溶剂。即,优选包含该非对称链状碳酸酯的3成分以上的混合溶剂。作为非对称链状碳酸酯之外的2种以上的非水溶剂的混合溶剂,由于可以提高充放电特性、电池寿命等全部电池性能,因此优选将环状碳酸酯或环状酯等高介电常数溶剂和对称链状碳酸酯或链状酯等低粘度溶剂组合使用,特别优选环状碳酸酯和对称链状碳酸酯的组合。此时的混合比没有特别限定,但从提高循环特性、保存特性等方面考虑,优选相对于100体积份非对称链状碳酸酯,环状碳酸酯或环状酯等高介电常数溶剂为10~400体积份、对称链状碳酸酯或链状酯等低粘度溶剂为10~800体积份。In addition, a mixed solvent of an asymmetric chain carbonate and two or more other non-aqueous solvents is preferable. That is, a mixed solvent containing three or more components of the asymmetric chain carbonate is preferable. As a mixed solvent of two or more non-aqueous solvents other than asymmetric chain carbonates, since it can improve all battery performance such as charge and discharge characteristics, battery life, etc., it is preferable to use high dielectric strength such as cyclic carbonates or cyclic esters. A constant solvent and a low-viscosity solvent such as a symmetrical chain carbonate or a chain ester are used in combination, and a combination of a cyclic carbonate and a symmetrical chain carbonate is particularly preferable. The mixing ratio at this time is not particularly limited, but from the aspects of improving cycle characteristics, storage characteristics, etc., preferably relative to 100 parts by volume of asymmetric chain carbonates, high dielectric constant solvents such as cyclic carbonates or cyclic esters are 10 ~400 parts by volume, and low-viscosity solvents such as symmetrical chain carbonates or chain esters are 10 to 800 parts by volume.
非对称链状碳酸酯之外的2种以上的非水溶剂的优选组合之一是以环状碳酸酯和对称链状碳酸酯为主体的组合。作为环状碳酸酯和对称链状碳酸酯的优选组合的具体例子,可以举出碳酸亚乙酯和碳酸二甲酯、碳酸亚乙酯和碳酸二乙酯、碳酸亚乙酯和碳酸二甲酯以及碳酸二乙酯等。在全部非水溶剂中的含有比率没有特别限定,但优选非对称链状碳酸酯为8~80体积%、环状碳酸酯为10~35体积%、对称链状碳酸酯为10~70体积%。One of the preferred combinations of two or more types of non-aqueous solvents other than asymmetric chain carbonates is a combination mainly of cyclic carbonates and symmetrical chain carbonates. Specific examples of a preferable combination of cyclic carbonate and symmetrical chain carbonate include ethylene carbonate and dimethyl carbonate, ethylene carbonate and diethyl carbonate, ethylene carbonate and dimethyl carbonate and diethyl carbonate. The content ratio in all non-aqueous solvents is not particularly limited, but preferably 8 to 80% by volume of asymmetric chain carbonates, 10 to 35% by volume of cyclic carbonates, and 10 to 70% by volume of symmetrical chain carbonates. .
可以举出在这些碳酸亚乙酯和对称链状碳酸酯的组合中进一步加入碳酸亚丙酯的组合作为优选的组合。含有碳酸亚丙酯时,碳酸亚乙酯和碳酸亚丙酯的容量比优选为99∶1~40∶60,特别优选为95∶5~50∶50。A combination in which propylene carbonate is further added to these combinations of ethylene carbonate and symmetrical chain carbonate is mentioned as a preferable combination. When propylene carbonate is contained, the capacity ratio of ethylene carbonate to propylene carbonate is preferably 99:1 to 40:60, particularly preferably 95:5 to 50:50.
非对称链状碳酸酯之外的2种以上的非水溶剂的优选组合的另一种为含有链状酯的组合。特别是从电池的低温特性提高方面考虑,优选上述含有环状碳酸酯和链状酯的组合,作为链状酯,特别优选乙酸甲酯、乙酸乙酯等。链状酯在全部非水溶剂中所占的比率优选为5体积%以上、更优选为8体积%以上、特别优选为15体积%以上,其上限优选为50体积%以下、更优选为35体积%以下、进一步优选为30体积%以下、特别优选为25体积%以下。因此,此时在全部非水溶剂中的含有比率优选如下比例:非对称链状碳酸酯为8~85体积%、环状碳酸酯为10~35体积%、链状酯为5~50体积%。Another preferred combination of two or more nonaqueous solvents other than the asymmetric chain carbonate is a combination containing a chain ester. In particular, from the viewpoint of improving the low-temperature characteristics of the battery, a combination containing the above-mentioned cyclic carbonate and a chain ester is preferable, and as the chain ester, methyl acetate, ethyl acetate, and the like are particularly preferable. The proportion of the chain ester in the total non-aqueous solvent is preferably 5% by volume or more, more preferably 8% by volume or more, particularly preferably 15% by volume or more, and the upper limit thereof is preferably 50% by volume or less, more preferably 35% by volume % or less, more preferably 30 volume % or less, particularly preferably 25 volume % or less. Therefore, at this time, the content ratio in all non-aqueous solvents is preferably as follows: asymmetric chain carbonate is 8 to 85 volume%, cyclic carbonate is 10 to 35 volume%, and chain ester is 5 to 50 volume%. .
可以举出在这些环状碳酸酯和链状酯的组合中进一步加入对称链状碳酸酯的组合作为优选的组合。含有对称链状碳酸酯时,全部非水溶剂中的对称链状碳酸酯的含有比率优选为10~60体积%。A combination in which a symmetrical chain carbonate is further added to these combinations of cyclic carbonate and chain ester can be cited as a preferable combination. When a symmetrical chain carbonate is contained, the content ratio of the symmetrical chain carbonate in the entire non-aqueous solvent is preferably 10 to 60% by volume.
作为本发明的二次电池用非水电解液的非水溶剂的优选组合的具体例子,例如可以举出:碳酸甲乙酯和碳酸亚乙酯,碳酸甲基正丙基酯和碳酸亚乙酯,碳酸甲乙酯、碳酸亚乙酯和碳酸亚丙酯,碳酸甲基正丙基酯、碳酸亚乙酯和碳酸亚丙酯等非对称链状碳酸酯和环状碳酸酯的组合;碳酸甲乙酯、碳酸亚乙酯和碳酸二甲酯,碳酸甲乙酯、碳酸亚乙酯和碳酸二乙酯,碳酸甲乙酯、碳酸亚乙酯、碳酸二乙酯和碳酸二甲酯,碳酸甲基正丙基酯、碳酸亚乙酯和碳酸二甲酯,碳酸甲基正丙基酯、碳酸亚乙酯和碳酸二乙酯,碳酸甲基正丙基酯、碳酸亚乙酯、碳酸二乙酯和碳酸二甲酯等非对称链状碳酸酯、环状碳酸酯和对称链状碳酸酯的组合;碳酸甲乙酯、碳酸亚乙酯和乙酸甲酯,碳酸甲乙酯、碳酸亚乙酯和乙酸乙酯等非对称链状碳酸酯、环状碳酸酯和链状酯的组合;碳酸甲乙酯、碳酸亚乙酯、碳酸二甲酯和乙酸甲酯,碳酸甲乙酯、碳酸亚乙酯、碳酸二乙酯和乙酸甲酯等非对称链状碳酸酯、环状碳酸酯、对称链状碳酸酯和链状酯等的组合。As specific examples of the preferred combination of the non-aqueous solvent of the non-aqueous electrolytic solution for secondary batteries of the present invention, for example, ethyl methyl carbonate and ethylene carbonate, methyl n-propyl carbonate and ethylene carbonate , a combination of asymmetric chain carbonates and cyclic carbonates such as ethyl methyl carbonate, ethylene carbonate and propylene carbonate, methyl-n-propyl carbonate, ethylene carbonate and propylene carbonate; methyl carbonate Ethyl carbonate, ethylene carbonate and dimethyl carbonate, ethyl methyl carbonate, ethylene carbonate and diethyl carbonate, ethyl methyl carbonate, ethylene carbonate, diethyl carbonate and dimethyl carbonate, methyl carbonate N-propyl carbonate, ethylene carbonate and dimethyl carbonate, methyl-n-propyl carbonate, ethylene carbonate and diethyl carbonate, methyl-n-propyl carbonate, ethylene carbonate, diethyl carbonate Combinations of asymmetric chain carbonates, cyclic carbonates and symmetrical chain carbonates such as esters and dimethyl carbonate; ethyl methyl carbonate, ethylene carbonate and methyl acetate, ethyl methyl carbonate, ethylene carbonate Combinations of asymmetric chain carbonates, cyclic carbonates and chain esters such as ethyl acetate; ethyl methyl carbonate, ethylene carbonate, dimethyl carbonate and methyl acetate, ethyl methyl carbonate, ethylene carbonate Combinations of unsymmetrical chain carbonates, cyclic carbonates, symmetrical chain carbonates, and chain esters such as esters, diethyl carbonate, and methyl acetate.
在该混合溶剂中含有锂盐和相对于电解液总质量为10ppm以上的上述特定化合物例如二氟磷酸盐,该非对称链状碳酸酯在全部非水溶剂中所占的含有比率为5体积%~90体积%的非水电解液由于使用其制造的二次电池的循环特性、低温放电特性、高温保存特性(特别是高温保存后的残留容量和高负荷放电容量)以及抑制气体产生的平衡变得优异,因而优选。The mixed solvent contains lithium salt and the above-mentioned specific compound such as difluorophosphate in an amount of 10 ppm or more relative to the total mass of the electrolyte, and the content ratio of the asymmetric chain carbonate in all non-aqueous solvents is 5% by volume The non-aqueous electrolyte of ~90% by volume is due to the cycle characteristics, low-temperature discharge characteristics, high-temperature storage characteristics (especially the residual capacity after high-temperature storage and high-load discharge capacity) of the secondary battery manufactured using it, and the balance change of suppressing gas generation. It is excellent and therefore preferred.
<电解液[3]><Electrolyte[3]>
在上述非水电解液中,优选构成电解液的非水溶剂至少含有“一种以上的链状羧酸酯”(电解液[3])。Among the above-mentioned non-aqueous electrolytic solutions, it is preferable that the non-aqueous solvent constituting the electrolytic solution contains at least "one or more kinds of chain carboxylate" (electrolytic solution [3]).
本发明(电解液[3])中的“锂盐”、“链状羧酸酯以外的非水溶剂”、“特定化合物”、“其它化合物”的种类及其含量、使用条件、非水电解液的制备方法等与上述相同。Types and contents of "lithium salts", "non-aqueous solvents other than chain carboxylic acid esters", "specific compounds" and "other compounds" in the present invention (electrolyte solution [3]), use conditions, non-aqueous electrolysis The preparation method of the liquid and the like are the same as above.
作为本发明(电解液[3])中使用的链状羧酸酯,没有特别限定,优选包含羧基的碳在内的碳原子数为1~5的羧酸的碳原子数1~4的烷基酯。此外,对于上述羧酸的元数没有特别限定,优选为一元羧酸或二元羧酸。The chain carboxylate used in the present invention (electrolyte solution [3]) is not particularly limited, but is preferably an alkane having 1 to 4 carbon atoms of a carboxylic acid having 1 to 5 carbon atoms including the carbon of the carboxyl group. base ester. In addition, there is no particular limitation on the number of carboxylic acids mentioned above, but monocarboxylic acids or dicarboxylic acids are preferred.
其中,优选为甲酸酯、乙酸酯、丙酸酯、丁酸酯等脂肪酸酯类;各种二羧酸酯类等,特别优选乙酸酯或丙酸酯。Among them, fatty acid esters such as formate, acetate, propionate, and butyrate; various dicarboxylic acid esters, etc. are preferred, and acetate and propionate are particularly preferred.
具体地说,优选乙酸甲酯、乙酸乙酯、乙酸丙酯、丙酸甲酯或丙酸乙酯,特别优选乙酸甲酯、乙酸乙酯或丙酸甲酯。Specifically, methyl acetate, ethyl acetate, propyl acetate, methyl propionate or ethyl propionate is preferred, and methyl acetate, ethyl acetate or methyl propionate is particularly preferred.
此外,这些链状羧酸酯可以混合2种以上来使用。混合的组合没有特别限定,作为优选的组合,可以举出乙酸甲酯和乙酸乙酯,乙酸甲酯和丙酸甲酯,乙酸乙酯和丙酸甲酯,乙酸甲酯、乙酸乙酯和丙酸甲酯等。由此,可以根据目的调节输出功率特性和高温保存特性等的平衡。In addition, these chain carboxylic acid esters can be used in mixture of 2 or more types. The combination of mixing is not particularly limited, as a preferred combination, methyl acetate and ethyl acetate, methyl acetate and methyl propionate, ethyl acetate and methyl propionate, methyl acetate, ethyl acetate and propionate Acid methyl ester etc. Thus, the balance between output characteristics, high-temperature storage characteristics, and the like can be adjusted according to purposes.
使用上述链状羧酸酯和除此之外的非水溶剂的混合溶剂时,链状羧酸酯相对于该全部非水溶剂的含有比率优选为3体积%以上、更优选为5体积%以上、进一步优选为8体积%以上、特别优选为10体积%以上,此外,其上限优选为50体积%以下、更优选为35体积%以下、进一步优选为30体积%以下、特别优选为25体积%以下,由于兼具本发明的高的低温特性和循环特性等而优选。When using a mixed solvent of the chain carboxylate and other non-aqueous solvents, the content ratio of the chain carboxylate to the entire non-aqueous solvent is preferably 3% by volume or more, more preferably 5% by volume or more , more preferably 8% by volume or more, particularly preferably 10% by volume or more, and the upper limit is preferably 50% by volume or less, more preferably 35% by volume or less, further preferably 30% by volume or less, particularly preferably 25% by volume Hereinafter, it is preferable because it has both high low-temperature characteristics and cycle characteristics of the present invention.
虽然对于通过组合使用链状羧酸酯和上述特定化合物而大幅提高低温输出功率特性的理由还不明确,但是上述特定化合物即使不含有链状羧酸酯时也有可能具有某种程度的低温特性提高效果,认为这是由于对电极有某种作用,链状羧酸酯促进了该作用,换言之,认为是由于,通过存在在低温下流动性也高的链状羧酸酯,这些特定化合物浸透至电极板内部而没有浪费地发挥效果,或者链状羧酸酯传播上述特定化合物和电极的相互作用。Although the reason why the low-temperature output characteristics are greatly improved by combining the chain carboxylate and the above-mentioned specific compound is not clear, the above-mentioned specific compound may have some degree of improvement in low-temperature characteristics even when the chain carboxylate is not contained effect, which is considered to be due to a certain action on the electrode, which is promoted by the chain carboxylate, in other words, it is considered to be due to the presence of the chain carboxylate which is also high in fluidity at low temperatures, and these specific compounds penetrate into the The effect is exerted without waste inside the electrode plate, or the chain carboxylate propagates the interaction of the above-mentioned specific compound and the electrode.
<电解液[4]><Electrolyte [4]>
上述非水电解液中,优选构成电解液的非水溶剂含有全部非水溶剂的60容量%以上的闪点70℃以上的溶剂(电解液[4])。Among the non-aqueous electrolytic solutions, it is preferable that the non-aqueous solvent constituting the electrolytic solution contains a solvent having a flash point of 70° C. or higher at 60% by volume or more of the total non-aqueous solvent (electrolyte solution [4]).
本发明(电解液[4])中的“锂盐”、“闪点70℃以上的溶剂以外的非水溶剂”、“特定化合物”、“其它化合物”的种类及其含量、使用条件、非水电解液的制备方法等与上述相同。The types and contents of "lithium salts", "non-aqueous solvents other than solvents with a flash point of 70°C or higher", "specific compounds" and "other compounds" in the present invention (electrolyte solution [4]), use conditions, non-aqueous solvents, etc. The preparation method and the like of the aqueous electrolytic solution are the same as described above.
本发明中,非水溶剂以总量计,必须含有全部非水溶剂的60容量%以上的闪点70℃以上的溶剂。进一步地,优选以总量计含有全部非水溶剂的60容量%以上的闪点80℃以上的溶剂,特别优选以总量计含有全部非水溶剂的60容量%以上的闪点90℃以上的溶剂。In the present invention, the non-aqueous solvent must contain a solvent having a flash point of 70° C. or higher in an amount of at least 60% by volume of the total non-aqueous solvent. Further, it is preferable to contain 60% by volume or more of the total non-aqueous solvents and a solvent with a flash point of 80° C. or higher, and it is particularly preferred to contain 60% by volume or more of the total non-aqueous solvents and have a flash point of 90° C. or higher. solvent.
闪点70℃以上的溶剂没有特别限定,例如可以举出,碳酸亚乙酯、碳酸亚丙酯、碳酸亚丁酯、γ-丁内酯、γ-戊内酯等作为优选的溶剂,其中,特别优选碳酸亚乙酯、碳酸亚丙酯或γ-丁内酯。这些溶剂可以使用一种,也可以混合2种以上使用,其组合没有特别限定。The solvent having a flash point of 70° C. or higher is not particularly limited, and examples thereof include ethylene carbonate, propylene carbonate, butylene carbonate, γ-butyrolactone, γ-valerolactone, and the like as preferred solvents, and particularly Preference is given to ethylene carbonate, propylene carbonate or gamma-butyrolactone. These solvents may be used alone or in combination of two or more, and the combination is not particularly limited.
此外,从循环特性提高方面考虑,优选相对于全部非水溶剂含有10容量%以上的碳酸亚乙酯或碳酸亚丙酯的组成。另一方面,如果含有大量的碳酸亚乙酯,则由于低温特性降低,碳酸亚乙酯的含量优选为全部非水溶剂的70容量%以下、特别优选为60容量%以下。In addition, from the viewpoint of improving cycle characteristics, a composition containing 10% by volume or more of ethylene carbonate or propylene carbonate relative to the total non-aqueous solvent is preferable. On the other hand, if a large amount of ethylene carbonate is contained, the low-temperature characteristics will decrease, so the content of ethylene carbonate is preferably 70% by volume or less, particularly preferably 60% by volume or less, of the total non-aqueous solvent.
以总量计,含有全部非水溶剂的60容量%以上的闪点70℃以上的溶剂的非水溶剂的优选例子为:On a total basis, preferred examples of non-aqueous solvents containing solvents having a flash point of 70° C. or more of 60% by volume or more of the total non-aqueous solvents are:
(1)γ-丁内酯在全部非水溶剂中所占的量为60容量%以上的非水溶剂;(1) Non-aqueous solvents in which γ-butyrolactone accounts for more than 60% by volume in all non-aqueous solvents;
(2)碳酸亚乙酯和γ-丁内酯的总计在全部非水溶剂中占80容量%以上、优选占85容量%以上且碳酸亚乙酯和γ-丁内酯的容量比为5∶95~45∶55的非水溶剂;(2) The total of ethylene carbonate and γ-butyrolactone accounts for more than 80% by volume in all non-aqueous solvents, preferably accounts for more than 85% by volume and the capacity ratio of ethylene carbonate and γ-butyrolactone is 5: 95~45:55 non-aqueous solvent;
(3)碳酸亚乙酯和碳酸亚丙酯的总计在全部非水溶剂中占80容量%以上、优选占85容量%以上且碳酸亚乙酯和碳酸亚丙酯的容量比为30∶70~60∶40的非水溶剂等。(3) The sum of ethylene carbonate and propylene carbonate accounts for more than 80% by volume in all non-aqueous solvents, preferably accounts for more than 85% by volume and the capacity ratio of ethylene carbonate and propylene carbonate is 30: 70~ 60:40 non-aqueous solvent, etc.
如果使用这些非水溶剂,则特别是循环特性和大电流放电特性等的平衡变得优异。Use of these non-aqueous solvents provides excellent balance between cycle characteristics, high-current discharge characteristics, and the like.
此外,非水电解液优选闪点为40℃以上、更优选为50℃以上、进一步优选为60℃以上、特别优选为70℃以上。若非水电解液的闪点过低,则电池暴露在高温下时,有时担心电解液的引火。In addition, the non-aqueous electrolytic solution preferably has a flash point of 40°C or higher, more preferably 50°C or higher, still more preferably 60°C or higher, particularly preferably 70°C or higher. Unless the flash point of the aqueous electrolytic solution is too low, there may be a fear of ignition of the electrolytic solution when the battery is exposed to high temperature.
本发明的二次电池用非水电解液中使用的非水溶剂可以在闪点70℃以上的溶剂中进一步配合闪点小于70℃的非水溶剂成分(下面简称为“其它非水溶剂成分”)。这样的其它非水溶剂成分可以从以往的作为非水电解液的溶剂提出的溶剂中适当选择使用。例如可以举出下述非水溶剂。The non-aqueous solvent used in the non-aqueous electrolytic solution for secondary battery of the present invention can be further mixed with a non-aqueous solvent component (hereinafter referred to as "other non-aqueous solvent components" with a flash point of less than 70 ° C) in a solvent with a flash point of more than 70 ° C. ). Such other nonaqueous solvent components can be appropriately selected and used from solvents conventionally proposed as solvents for nonaqueous electrolytic solutions. For example, the following non-aqueous solvents are mentioned.
(1)链状碳酸酯(1) Chain carbonate
作为链状碳酸酯,优选为碳酸二烷基酯,构成碳酸二烷基酯的烷基的碳原子数分别优选为1~5,特别优选1~4。具体地说,例如可以举出碳酸二甲酯、碳酸二乙酯、碳酸二正丙酯、碳酸甲乙酯、碳酸甲基正丙基酯、碳酸乙基正丙基酯等碳酸二烷基酯。其中,优选碳酸二甲酯、碳酸二乙酯、碳酸甲乙酯。As the chain carbonate, dialkyl carbonate is preferable, and the number of carbon atoms of the alkyl group constituting the dialkyl carbonate is preferably 1-5, particularly preferably 1-4. Specifically, for example, dialkyl carbonates such as dimethyl carbonate, diethyl carbonate, di-n-propyl carbonate, ethyl methyl carbonate, methyl-n-propyl carbonate, ethyl-n-propyl carbonate, etc. . Among them, dimethyl carbonate, diethyl carbonate, and ethyl methyl carbonate are preferable.
(2)链状酯(2) chain ester
具体地说,例如可以举出乙酸甲酯、乙酸乙酯、乙酸丙酯、丙酸甲酯等。Specifically, methyl acetate, ethyl acetate, propyl acetate, methyl propionate, etc. are mentioned, for example.
(3)环状醚(3) Cyclic ether
具体地说,例如可以举出四氢呋喃、2-甲基四氢呋喃、四氢吡喃等。Specifically, tetrahydrofuran, 2-methyltetrahydrofuran, tetrahydropyran, etc. are mentioned, for example.
(4)链状醚(4) chain ether
具体地说,例如可以举出二甲氧基乙烷、二甲氧基甲烷等。Specifically, dimethoxyethane, dimethoxymethane, etc. are mentioned, for example.
其它非水溶剂成分可以一种与“闪点70℃以上的溶剂”组合使用,也可以组合2种以上其它非水溶剂成分使用。此时,特别是从循环特性和大电流放电特性方面考虑,优选将链状碳酸酯和/或链状酯与“闪点70℃以上的溶剂”组合使用。The other non-aqueous solvent components may be used in combination with the "solvent having a flash point of 70° C. or higher", or two or more other non-aqueous solvent components may be used in combination. In this case, it is preferable to use a chain carbonate and/or a chain ester in combination with a "solvent having a flash point of 70° C. or higher" particularly from the viewpoint of cycle characteristics and large-current discharge characteristics.
全部非水溶剂中的闪点为70℃以上的溶剂的含量必须为60容量%以上,优选为70容量%以上,更优选为75容量%以上,进一步优选为80容量%以上,特别优选为85容量%以上。此外,其上限优选为100容量%以下,特别优选为90容量%以下。如果闪点为70℃以上的溶剂的含量过少,则在高温下保存电池时有时得不到内压容易升高等期望的效果,另一方面,如果过多,则由于粘度升高而使电导率降低,锂二次电池的性能有时降低。The content of the solvent having a flash point of 70° C. or higher in all non-aqueous solvents must be 60% by volume or more, preferably 70% by volume or more, more preferably 75% by volume or more, further preferably 80% by volume or more, and particularly preferably 85% by volume. capacity % above. In addition, the upper limit thereof is preferably 100% by volume or less, particularly preferably 90% by volume or less. If the content of the solvent having a flash point of 70° C. or higher is too small, the desired effect of easily increasing the internal pressure may not be obtained when the battery is stored at a high temperature. The rate decreases, and the performance of the lithium secondary battery sometimes decreases.
虽然对于使用本发明的非水电解液(电解液[4])的非水电解质二次电池在使用某种量以上的闪点高的溶剂时输出功率特性优异的理由还不明确,但可认为是如下理由,但本发明应该不限于下述理由。即,认为上述特定化合物与电极作用,降低与锂离子的进出有关的反应电阻,从而提高输出功率特性。此外认为,碳酸亚乙酯、碳酸亚丙酯、碳酸亚丁酯、γ-丁内酯、γ-戊内酯等闪点高的溶剂与链状碳酸酯相比,介电常数高,存在某种程度以上的介电常数高的溶剂时,其效果变得显著。进一步地认为是由于,二次电池的1个电池外壳中收纳的电池要素所具有的电池容量为3安培小时(Ah)以上时和/或二次电池的直流电阻成分为10毫欧(Ω)以下时,直流电阻成分的贡献减少,与直流电阻成分的贡献大的电池相比,容易表现出该非水电解液本来的效果。Although the reason why the output power characteristics are excellent when a certain amount or more of a solvent with a high flash point is used for a nonaqueous electrolyte secondary battery using the nonaqueous electrolyte solution (electrolyte solution [4]) of the present invention is not clear, it is considered that The reasons are as follows, but the present invention should not be limited to the following reasons. That is, it is considered that the above-mentioned specific compound acts on the electrode to reduce the reaction resistance related to the entry and exit of lithium ions, thereby improving the output characteristics. In addition, it is considered that solvents with high flash points such as ethylene carbonate, propylene carbonate, butylene carbonate, γ-butyrolactone, and γ-valerolactone have higher dielectric constants than chain carbonates, and there is a certain The effect becomes remarkable in the case of a solvent with a high dielectric constant of more than a certain degree. It is further considered that the battery element contained in one battery case of the secondary battery has a battery capacity of 3 ampere hours (Ah) or more and/or the DC resistance component of the secondary battery is 10 milliohms (Ω) In the following cases, the contribution of the DC resistance component is reduced, and the original effect of the non-aqueous electrolytic solution is more likely to be exhibited compared with a battery with a large contribution of the DC resistance component.
<电解液[5]><Electrolyte [5]>
在上述非水电解液中,优选含有LiN(CnF2n+1SO2)2(式中,n为1~4的整数)和/或二(草酸根合)硼酸锂作为构成电解液的“锂盐”的电解液(电解液[5])。In the above-mentioned non-aqueous electrolytic solution, it is preferable to contain LiN(C n F 2n+1 SO 2 ) 2 (where n is an integer of 1 to 4) and/or bis(oxalato)lithium borate as constituents of the electrolytic solution. Electrolyte of "lithium salt" (electrolyte [5]).
本发明(电解液[5])中的“非水溶剂”、“特定化合物”、“其它化合物”的种类及其含量、使用条件、非水电解液的制备方法等与上述相同。The types and contents of "non-aqueous solvent", "specific compound", and "other compounds" in the present invention (electrolyte [5]), use conditions, preparation method of non-aqueous electrolytic solution, etc. are the same as above.
本发明的二次电池用非水电解液(电解液[5])至少在非水溶剂中溶解有锂盐,作为这样的锂盐,含有选自LiN(CnF2n+1SO2)2(式中,n为1~4的整数)和二(草酸根合)硼酸锂中的至少一种。它们可以单独使用一种,也可以以任意的组合和比率组合使用2种以上。The non-aqueous electrolytic solution for secondary batteries (electrolyte [5]) of the present invention has at least a lithium salt dissolved in a non-aqueous solvent, and such a lithium salt contains a lithium salt selected from LiN(C n F 2n+1 SO 2 ) 2 (wherein, n is an integer of 1 to 4) and at least one of lithium bis(oxalato)borate. These may be used individually by 1 type, and may use 2 or more types together in arbitrary combinations and ratios.
二次电池用非水电解液中,通过对于下述(a)组合使用10ppm以上的(b)上述“特定化合物”,可以提供输出功率特性大幅提高,且高温保存特性或循环特性优异的用于二次电池的非水电解液。In the non-aqueous electrolyte solution for secondary batteries, by using 10 ppm or more of (b) the above-mentioned "specific compound" in combination with the following (a), it is possible to provide a battery that has greatly improved output characteristics and excellent high-temperature storage characteristics or cycle characteristics. Non-aqueous electrolyte for secondary batteries.
(a)LiN(CnF2n+1SO2)2(式中,n为1~4的整数)和/或二(草酸根合)硼酸锂(a) LiN(C n F 2n+1 SO 2 ) 2 (where n is an integer of 1 to 4) and/or lithium bis(oxalato)borate
作为(a),没有特别限定,LiN(CF3SO2)2或二(草酸根合)硼酸锂由于可特别地发挥上述效果而优选。(a) is not particularly limited, but LiN(CF 3 SO 2 ) 2 or lithium bis(oxalato)borate is preferable because it can particularly exert the above-mentioned effects.
本发明的二次电池用非水电解液中,如上所述,作为锂盐,含有LiN(CnF2n+1SO2)2(式中,n为1~4的整数)和/或二(草酸根合)硼酸锂作为必须成分,但也可以组合使用除此之外的以往公知的锂盐(下面简称为“其它锂盐”)。In the non-aqueous electrolytic solution for secondary batteries of the present invention, as described above, LiN(C n F 2n+1 SO 2 ) 2 (where n is an integer of 1 to 4) and/or di Lithium (oxalato)borate is an essential component, but other conventionally known lithium salts (hereinafter simply referred to as "other lithium salts") may be used in combination.
作为其它锂盐,没有特别限定,例如可以举出下述锂盐。It does not specifically limit as another lithium salt, For example, the following lithium salt is mentioned.
无机锂盐:LiPF6、LiBF4、LiAsF6、LiSbF6等无机氟化物盐;LiClO4、LiBrO4、LiIO4等高卤酸盐;LiAlCl4等无机氯化物盐等。Inorganic lithium salts: LiPF 6 , LiBF 4 , LiAsF 6 , LiSbF 6 and other inorganic fluoride salts; LiClO 4 , LiBrO 4 , LiIO 4 and other perhalide salts; LiAlCl 4 and other inorganic chloride salts, etc.
含氟有机锂盐:LiCF3SO3等全氟链烷磺酸盐;LiC(CF3SO2)3等全氟链烷磺酰甲基化盐;Li[PF5(CF2CF2CF3)]、Li[PF4(CF2CF2CF3)2]、Li[PF3(CF2CF2CF3)3]、Li[PF5(CF2CF2CF2CF3)]、Li[PF4(CF2CF2CF2CF3)2]、Li[PF3(CF2CF2CF2CF3)3]等氟烷基氟化磷酸盐等。Fluorinated organic lithium salts: LiCF 3 SO 3 and other perfluoroalkane sulfonates; LiC(CF 3 SO 2 ) 3 and other perfluoroalkanesulfonylmethylation salts; Li[PF 5 (CF 2 CF 2 CF 3 )], Li[PF 4 (CF 2 CF 2 CF 3 ) 2 ], Li[PF 3 (CF 2 CF 2 CF 3 ) 3 ], Li[PF 5 (CF 2 CF 2 CF 2 CF 3 )], Li Fluoroalkyl fluorinated phosphates such as [PF 4 (CF 2 CF 2 CF 2 CF 3 ) 2 ], Li[PF 3 (CF 2 CF 2 CF 2 CF 3 ) 3 ], etc.
其它的草酸根合硼酸锂类:二氟草酸根合硼酸锂等。Other lithium oxalatoborates: lithium difluorooxalatoborate, etc.
它们可以单独使用一种,也可以以任意的组合和比率组合使用2种以上。这些“其它锂盐”中,如果综合判断在非水溶剂中的溶解性、用作二次电池时的充放电特性、输出功率特性、循环特性等,则优选LiPF6、LiBF4,特别优选LiPF6。These may be used individually by 1 type, and may use 2 or more types together in arbitrary combinations and ratios. Among these "other lithium salts", LiPF 6 and LiBF 4 are preferred, LiPF 6 is particularly preferred if the solubility in non-aqueous solvents, charge and discharge characteristics when used as a secondary battery, output power characteristics, and cycle characteristics are comprehensively judged. 6 .
非水电解液中的LiN(CnF2n+1SO2)2(式中,n为1~4的整数)和/或二(草酸根合)硼酸锂的浓度在将这些盐作为主要盐使用时,通常为0.3mol/L以上、优选为0.5mol/L以上、更优选为0.7mol/L以上,其上限通常为2mol/L以下、优选为1.8mol/L以下、更优选为1mol/L以下。此外,将LiPF6、LiBF4等其它锂盐作为主要盐使用时,LiN(CnF2n+1SO2)2(式中,n为1~4的整数)和/或二(草酸根合)硼酸锂的浓度为0.001mol/L以上、优选为0.01mol/L以上、更优选为0.03mol/L以上,此外,其上限通常为0.3mol/L以下、优选为0.2mol/L以下、更优选为0.1mol/L以下。这里所说的主要盐是指在非水电解液中浓度最大的锂盐。The concentration of LiN(C n F 2n+1 SO 2 ) 2 (wherein, n is an integer of 1 to 4) and/or bis(oxalato)lithium borate in the non-aqueous electrolyte is when these salts are used as the main salts. When used, it is usually more than 0.3 mol/L, preferably more than 0.5 mol/L, more preferably more than 0.7 mol/L, and its upper limit is usually less than 2 mol/L, preferably less than 1.8 mol/L, more preferably 1 mol/L Below L. In addition, when LiPF 6 , LiBF 4 and other lithium salts are used as the main salt, LiN(C n F 2n+1 SO 2 ) 2 (where n is an integer of 1 to 4) and/or bis(oxalato ) lithium borate concentration is more than 0.001mol/L, preferably more than 0.01mol/L, more preferably more than 0.03mol/L, in addition, its upper limit is usually less than 0.3mol/L, preferably less than 0.2mol/L, more Preferably it is 0.1 mol/L or less. The main salt mentioned here refers to the lithium salt with the largest concentration in the non-aqueous electrolyte.
非水电解液中的锂盐的总浓度,没有特别限定,但通常为0.5mol/L以上、优选为0.6mol/L以上、更优选为0.7mol/L以上。此外,其上限通常为2mol/L以下、优选为1.8mol/L以下、更优选为1.7mol/L以下。如果浓度过低,则非水电解液的电导率有时不充分,另一方面,如果浓度过高,则由于粘度升高,电导率有时降低,锂二次电池的性能有时降低。The total concentration of lithium salts in the non-aqueous electrolytic solution is not particularly limited, but is usually at least 0.5 mol/L, preferably at least 0.6 mol/L, and more preferably at least 0.7 mol/L. In addition, the upper limit thereof is usually 2 mol/L or less, preferably 1.8 mol/L or less, more preferably 1.7 mol/L or less. If the concentration is too low, the conductivity of the non-aqueous electrolytic solution may be insufficient. On the other hand, if the concentration is too high, the conductivity may decrease due to an increase in viscosity, and the performance of the lithium secondary battery may decrease.
组合使用2种以上锂盐时的优选的一个例子是将选自LiN(CnF2n+1SO2)2(式中,n为1~4的整数)和二(草酸根合)硼酸锂中的至少一种锂盐和LiPF6组合使用。通过组合使用,特别是可以提高输出功率特性、保存特性。此时的选自LiN(CnF2n+1SO2)2(式中,n为1~4的整数)和二(草酸根合)硼酸锂中的至少一种锂盐在非水电解液中的浓度如下:将这些锂盐作为主要盐使用时,优选为0.4mol/L以上、更优选为0.5mol/L以上、特别优选为0.6mol/L以上。其上限优选为1.8mol/L以下、更优选为1.5mol/L以下、特别优选为1.2mol/L以下。此时的LiPF6的浓度优选为0.001mol/L以上、更优选为0.01mol/L以上、特别优选为0.1mol/L以上。此外,其上限在LiPF6比选自LiN(CnF2n+1SO2)2(式中,n为1~4的整数)和二(草酸根合)硼酸锂中的至少一种锂盐的浓度低的范围内,优选为1mol/L以下、更优选为0.8mol/L以下、特别优选为0.3mol/L以下。A preferred example of using two or more lithium salts in combination is LiN(C n F 2n+1 SO 2 ) 2 (where n is an integer of 1 to 4) and lithium di(oxalato)borate At least one lithium salt and LiPF 6 are used in combination. By using them in combination, in particular, output characteristics and storage characteristics can be improved. At this time, at least one lithium salt selected from LiN(C n F 2n+1 SO 2 ) 2 (in the formula, n is an integer of 1 to 4) and lithium bis(oxalato)borate in the non-aqueous electrolyte solution The concentration in Li is as follows: when these lithium salts are used as the main salt, it is preferably 0.4 mol/L or more, more preferably 0.5 mol/L or more, particularly preferably 0.6 mol/L or more. The upper limit thereof is preferably 1.8 mol/L or less, more preferably 1.5 mol/L or less, particularly preferably 1.2 mol/L or less. The concentration of LiPF 6 at this time is preferably 0.001 mol/L or more, more preferably 0.01 mol/L or more, particularly preferably 0.1 mol/L or more. In addition, the upper limit is LiPF 6 to at least one lithium salt selected from LiN(C n F 2n+1 SO 2 ) 2 (where n is an integer from 1 to 4) and lithium bis(oxalato)borate. In the range where the concentration is low, it is preferably 1 mol/L or less, more preferably 0.8 mol/L or less, particularly preferably 0.3 mol/L or less.
将LiPF6作为主要盐使用时,选自LiN(CnF2n+1SO2)2(式中,n为1~4的整数)和二(草酸根合)硼酸锂中的至少一种锂盐的浓度优选为0.001mol/L以上、更优选为0.01mol/L以上、特别优选为0.03mol/L以上。另外,其上限优选为0.3mol/L以下、更优选为0.2mol/L以下、特别优选为0.1mol/L以下。此时的LiPF6的浓度优选为0.5mol/L以上、更优选为0.6mol/L以上、特别优选为0.7mol/L以上。此外,其上限优选为1.8mol/L以下、更优选为1.7mol/L以下、特别优选为1.5mol/L以下。When LiPF 6 is used as the main salt, at least one lithium selected from LiN(C n F 2n+1 SO 2 ) 2 (where n is an integer of 1 to 4) and lithium bis(oxalato)borate The concentration of the salt is preferably 0.001 mol/L or higher, more preferably 0.01 mol/L or higher, particularly preferably 0.03 mol/L or higher. In addition, the upper limit thereof is preferably 0.3 mol/L or less, more preferably 0.2 mol/L or less, particularly preferably 0.1 mol/L or less. The concentration of LiPF 6 at this time is preferably 0.5 mol/L or higher, more preferably 0.6 mol/L or higher, and particularly preferably 0.7 mol/L or higher. In addition, the upper limit thereof is preferably 1.8 mol/L or less, more preferably 1.7 mol/L or less, particularly preferably 1.5 mol/L or less.
另外,如果在该选自LiN(CnF2n+1SO2)2(式中,n为1~4的整数)和二(草酸根合)硼酸锂中的至少一种锂盐和LiPF6的组合中组合使用LiBF4,则由于具有抑制因高温保存引起的劣化的效果而优选。此时,LiBF4的浓度通常为0.001mol/L以上、优选为0.01mol/L以上、更优选为0.03mol/L以上,其上限通常为0.4mol/L以下、优选为0.15mol/L以下、更优选为0.1mol/L以下。In addition , if at least one lithium salt and LiPF 6 Use of LiBF 4 in combination is preferable since it has the effect of suppressing deterioration due to high-temperature storage. At this time, the concentration of LiBF is usually 0.001 mol/L or more, preferably 0.01 mol/L or more, more preferably 0.03 mol/L or more, and its upper limit is usually 0.4 mol/L or less, preferably 0.15 mol/L or less, More preferably, it is 0.1 mol/L or less.
含有LiN(CnF2n+1SO2)2(式中,n为1~4的整数)和/或二(草酸根合)硼酸锂和选自上述通式(1)所示的环状化合物、上述通式(2)所示的化合物、分子内具有通式(3)所示结构的链状化合物、分子内具有S-F键的化合物、硝酸盐、亚硝酸盐、单氟磷酸盐、二氟磷酸盐、乙酸盐、丙酸盐中的化合物中的至少一种化合物的非水电解液,由于使用该非水电解液制造的电池的循环特性和高温保存特性(特别是高温保存后的残留容量和高负荷放电容量)的平衡变得优异,所以优选。Containing LiN(C n F 2n+1 SO 2 ) 2 (wherein, n is an integer of 1 to 4) and/or bis(oxalato)lithium borate and cyclic Compounds, compounds represented by the above general formula (2), chain compounds with structures represented by general formula (3) in their molecules, compounds with SF bonds in their molecules, nitrates, nitrites, monofluorophosphates, di The non-aqueous electrolytic solution of at least one compound in the compound in fluorophosphate, acetate, propionate, due to the cycle characteristics and high-temperature storage characteristics (especially after high-temperature storage) of batteries manufactured using the non-aqueous electrolytic solution The balance between residual capacity and high-load discharge capacity) becomes excellent, so it is preferable.
虽然对于使用本发明的非水电解液的非水电解质二次电池的输出特性优异、且高温保存性或循环特性优异的理由还不明确,但可认为是由于上述特定化合物对电极有某种作用,降低与锂离子的进出有关的反应电阻,从而提高输出功率特性。其中推测,通过使用LiN(CnF2n+1SO2)2(式中,n为1~4的整数)和/或二(草酸根合)硼酸锂作为锂盐,这些锂盐在负极和正极表面上适度地进行反应,与其它的非水电解液成分形成来源于该锂盐的锂离子透过性优异的稳定的复合保护涂膜,通过该复合涂膜,抑制活性高的电极与非水电解液的多余的副反应,从而提高输出功率特性,同时高温保存特性和循环特性得到改善。Although it is not clear why the output characteristics of the nonaqueous electrolyte secondary battery using the nonaqueous electrolyte of the present invention are excellent, and the high-temperature storage stability or cycle characteristics are excellent, it is considered that the above-mentioned specific compound has a certain effect on the electrode. , Reduce the reaction resistance related to the entry and exit of lithium ions, thereby improving the output power characteristics. It is speculated that by using LiN(C n F 2n+1 SO 2 ) 2 (wherein, n is an integer of 1 to 4) and/or bis(oxalato)lithium borate as lithium salts, these lithium salts are in the negative electrode and Moderately react on the surface of the positive electrode, and form a stable composite protective coating film with excellent lithium ion permeability derived from the lithium salt with other non-aqueous electrolyte components. Unnecessary side reactions of the water electrolyte, thereby improving output power characteristics, while improving high-temperature storage characteristics and cycle characteristics.
<电解液[6]><Electrolyte [6]>
上述非水电解液中,优选构成电解液的锂盐为含氟锂盐,并且在全部非水电解液中含有10ppm~300ppm的氟化氢(HF)(电解液[6])。In the non-aqueous electrolytic solution, it is preferable that the lithium salt constituting the electrolytic solution is a fluorine-containing lithium salt, and that the entire non-aqueous electrolytic solution contains 10 ppm to 300 ppm of hydrogen fluoride (HF) (electrolyte solution [6]).
本发明(电解液[6])中的“非水溶剂”、“特定化合物”、“其它化合物”的种类及其含量、使用条件、非水电解液的制备方法等与上述相同。本发明(电解液[6])中,作为“含氟锂盐”,只要是已知可以用作锂二次电池用非水电解液的电解质的含氟锂盐即可,没有特别限定,例如可以使用上述锂盐。它们可以单独使用一种,也可以以任意的组合和比率组合使用2种以上。其中,从容易在后述醇类存在下生成氟化氢(HF)的观点来看,优选LiPF6、LiBF4等。此外,如果综合判断在非水溶剂中的溶解性、制成二次电池时的充放电特性、输出功率特性、循环特性等,则优选LiPF6。The types and contents of "non-aqueous solvent", "specific compound", and "other compounds" in the present invention (electrolyte [6]), use conditions, preparation method of non-aqueous electrolytic solution, etc. are the same as above. In the present invention (electrolyte solution [6]), the "fluorine-containing lithium salt" is not particularly limited as long as it is a known fluorine-containing lithium salt that can be used as an electrolyte for a non-aqueous electrolyte solution for a lithium secondary battery, for example The lithium salts mentioned above can be used. These may be used individually by 1 type, and may use 2 or more types together in arbitrary combinations and ratios. Among them, LiPF 6 , LiBF 4 and the like are preferable from the viewpoint of easily generating hydrogen fluoride (HF) in the presence of alcohols described later. In addition, LiPF 6 is preferable when the solubility in non-aqueous solvents, charge and discharge characteristics when used as a secondary battery, output power characteristics, cycle characteristics, etc. are comprehensively judged.
此外,电解液中除了含氟锂盐以外还可以混合使用不含氟的锂盐,作为例子,可以举出下述锂盐。In addition, in addition to the fluorine-containing lithium salt, a fluorine-free lithium salt may be mixed and used in the electrolytic solution, and examples thereof include the following lithium salts.
无机锂盐:LiClO4、LiBrO4、LiIO4等高卤酸盐;LiAlCl4等无机氯化物盐等。Inorganic lithium salts: LiClO 4 , LiBrO 4 , LiIO 4 and other perhalide salts; LiAlCl 4 and other inorganic chloride salts, etc.
草酸根合硼酸盐:二(草酸根合)硼酸锂等。Oxalatoborate: lithium bis(oxalato)borate, etc.
非水电解液中的上述锂盐的浓度没有特别限定,通常为0.5mol/L以上、优选为0.6mol/L以上、更优选为0.7mol/L以上。此外,其上限通常为2mol/L以下、优选为1.8mol/L以下、更优选为1.7mol/L以下。如果浓度过低,则非水电解液的电导率有时不充分,另一方面,如果浓度过高,则由于粘度升高,电导率有时降低,锂二次电池的性能有时降低。The concentration of the lithium salt in the non-aqueous electrolytic solution is not particularly limited, but is usually at least 0.5 mol/L, preferably at least 0.6 mol/L, and more preferably at least 0.7 mol/L. In addition, the upper limit thereof is usually 2 mol/L or less, preferably 1.8 mol/L or less, more preferably 1.7 mol/L or less. If the concentration is too low, the conductivity of the non-aqueous electrolytic solution may be insufficient. On the other hand, if the concentration is too high, the conductivity may decrease due to an increase in viscosity, and the performance of the lithium secondary battery may decrease.
非水电解液中的上述含氟锂盐的浓度没有特别限定,但通常为0.5mol/L以上、优选为0.6mol/L以上、更优选为0.7mol/L以上。此外,其上限通常为2mol/L以下、优选为1.8mol/L以下、更优选为1.7mol/L以下。如果浓度过低,则非水电解液的电导率有时不充分或氟化氢(HF)的生成有时不充分,另一方面,如果浓度过高,则由于粘度升高,电导率有时降低或氟化氢(HF)的生成有时过量进行,锂二次电池的性能有可能降低。The concentration of the fluorine-containing lithium salt in the non-aqueous electrolyte solution is not particularly limited, but is usually 0.5 mol/L or more, preferably 0.6 mol/L or more, more preferably 0.7 mol/L or more. In addition, the upper limit thereof is usually 2 mol/L or less, preferably 1.8 mol/L or less, more preferably 1.7 mol/L or less. If the concentration is too low, the conductivity of the non-aqueous electrolytic solution is sometimes insufficient or the generation of hydrogen fluoride (HF) is sometimes insufficient. On the other hand, if the concentration is too high, the conductivity is sometimes reduced or the hydrogen fluoride (HF) ) may be generated excessively, and the performance of the lithium secondary battery may decrease.
非水电解液中的含氟锂盐相对于全部锂盐的比率,相对于全部锂盐,优选含氟锂盐为50质量%以上、特别优选为70质量%以上。另外,特别优选混合的锂盐都为含氟锂盐。如果含氟锂盐的比率过低,则氟化氢的生成有时不充分。The ratio of the fluorine-containing lithium salt to the total lithium salt in the nonaqueous electrolyte is preferably 50% by mass or more, particularly preferably 70% by mass or more, based on the total lithium salt. In addition, it is particularly preferable that the mixed lithium salts are all fluorine-containing lithium salts. When the ratio of the fluorine-containing lithium salt is too low, the production of hydrogen fluoride may be insufficient.
锂盐可以单独使用一种,也以任意的组合和比率组合使用2种以上,组合使用2种以上锂盐时的优选的一个例子为LiPF6和LiBF4的组合使用,此时,LiBF4在两者总计中所占的比率优选为0.01质量%~20质量%,更优选为0.1质量%~5质量%。此外,其它优选的一个例子为组合使用无机氟化物盐和全氟链烷磺酰亚胺盐,此时,无机氟化物盐在两者总计中所占的比率优选为70质量%~99质量%,更优选为80质量%~98质量%。该两者的组合使用具有抑制因高温保存所导致的劣化的效果。One kind of lithium salt may be used alone, or two or more kinds may be used in combination in any combination and ratio. A preferred example when two or more kinds of lithium salts are used in combination is the combined use of LiPF6 and LiBF4 . At this time, LiBF4 is in the The ratio of the total of both is preferably 0.01% by mass to 20% by mass, more preferably 0.1% by mass to 5% by mass. Another preferred example is the combined use of an inorganic fluoride salt and a perfluoroalkanesulfonylimide salt. In this case, the ratio of the inorganic fluoride salt to the total of the two is preferably 70% by mass to 99% by mass. , and more preferably 80% by mass to 98% by mass. The combined use of both has an effect of suppressing deterioration due to high-temperature storage.
在上述非水溶剂中溶解有LiPF6等含氟锂盐而得到的非水电解液中,大多含有氟化氢(HF)。作为含有氟化氢(HF)的原因,除了来源于含氟锂盐中的杂质的氟化氢之外,还存在非水溶剂中的微量的水分或醇类和含氟锂盐反应而生成的氟化氢。上述专利文献16中,必须极力除去非水电解液中的氟化氢(HF),特别优选为15ppm以下,实施例中9ppm的非水电解液的循环特性为最优异的结果。Hydrogen fluoride (HF) is often contained in the non-aqueous electrolytic solution obtained by dissolving a fluorine-containing lithium salt such as LiPF 6 in the above-mentioned non-aqueous solvent. The reason for containing hydrogen fluoride (HF) is not only hydrogen fluoride derived from impurities in the fluorine-containing lithium salt, but also hydrogen fluoride produced by the reaction of a small amount of water in the non-aqueous solvent or alcohols and the fluorine-containing lithium salt. In the above-mentioned Patent Document 16, it is necessary to remove hydrogen fluoride (HF) in the non-aqueous electrolytic solution as much as possible, and it is particularly preferably 15 ppm or less. In the examples, the cycle characteristics of the non-aqueous electrolytic solution of 9 ppm are the most excellent results.
本发明(电解液[6])的情况下,作为氟化氢(HF)的含量,通常为10ppm以上、优选为12ppm以上、更优选为15ppm以上、特别优选为20ppm以上,且通常为300ppm以下、优选为250ppm以下、更优选为200ppm以下、特别优选为150ppm以下。如果含量过低,则输出功率提高的效果有时不充分,如果含量超过该范围,则有时对输出功率和循环特性有不良影响。In the case of the present invention (electrolyte solution [6]), the content of hydrogen fluoride (HF) is usually 10 ppm or more, preferably 12 ppm or more, more preferably 15 ppm or more, particularly preferably 20 ppm or more, and usually 300 ppm or less, preferably It is 250 ppm or less, more preferably 200 ppm or less, particularly preferably 150 ppm or less. If the content is too low, the effect of improving output power may not be sufficient, and if the content exceeds this range, output power and cycle characteristics may be adversely affected.
为了含有氟化氢(HF),可以向非水电解液或原料的非水溶剂中直接加入,或者利用水或醇类与含氟锂盐的反应在非水电解液内部产生,因此,可以使用向非水电解液中加入水或醇类、或者在原料的非水溶剂中预先以适当的浓度含有这些成分等的方法。此时,到反应结束有时需要一定的时间。换言之,在含有水或醇类的非水溶剂中溶解含氟锂盐,制备加入了特定化合物的电解液时,在水或醇类和含氟锂盐的反应结束前需要时间,但是将其用于电池制造时,可以不必等待反应结束。在本发明中,作为电池发挥功能时,存在特定浓度范围的氟化氢(HF)即可,也可以在电池内生成。在原料的非水溶剂中生成氟化氢(HF)时,也可以在作为原料使用的非水溶剂的一部分中生成氟化氢(HF),将其与不含有水或醇类的非水溶剂混合。In order to contain hydrogen fluoride (HF), it can be directly added to the non-aqueous electrolyte or the non-aqueous solvent of the raw material, or it can be generated inside the non-aqueous electrolyte by the reaction of water or alcohols and fluorine-containing lithium salt. Therefore, the non-aqueous electrolyte can be used A method in which water or alcohols are added to the aqueous electrolytic solution, or these components are contained in an appropriate concentration in advance in the non-aqueous solvent of the raw material. In this case, it may take a certain amount of time until the end of the reaction. In other words, when a fluorine-containing lithium salt is dissolved in a non-aqueous solvent containing water or alcohols to prepare an electrolytic solution to which a specific compound is added, it takes time until the reaction between water or alcohols and a fluorine-containing lithium salt is completed, but it is used When manufacturing batteries, there is no need to wait for the reaction to finish. In the present invention, to function as a battery, hydrogen fluoride (HF) in a specific concentration range may be present, and may be generated in the battery. When hydrogen fluoride (HF) is generated in the non-aqueous solvent of the raw material, hydrogen fluoride (HF) may be generated in a part of the non-aqueous solvent used as the raw material and mixed with a non-aqueous solvent not containing water or alcohols.
在非水溶剂中预先含有水或醇类时,根据使用的溶剂的纯度,有时从开始就含有比必要量多的水或醇类。此时,优选通过吸附处理、蒸馏、结晶析出等方法纯化非水溶剂,除去水或醇类而使用。可以直接使用除去水或醇类,残留规定量的水或醇类的非水溶剂,也可以配合水或醇类以在纯化的非水溶剂中为规定量来使用。When water or alcohols are previously contained in the non-aqueous solvent, depending on the purity of the solvent used, water or alcohols may be contained in a larger than necessary amount from the beginning. In this case, it is preferable to purify the nonaqueous solvent by methods such as adsorption treatment, distillation, and crystallization to remove water or alcohols before use. The non-aqueous solvent that removes water or alcohols and leaves a predetermined amount of water or alcohols may be used as it is, or it may be used in a purified non-aqueous solvent by mixing water or alcohols in a predetermined amount.
吸附处理若在液体的状态下则可以实施,可以通过在氧化铝、活性炭、硅胶、分子筛(商品名)4A和/或分子筛5A等非水溶剂中溶解或者通过不与非水溶剂反应的吸附剂进行纯化处理。此时,也可以分别纯化碳酸二甲酯这样的在常温下为液态的原料,而碳酸亚乙酯这样的在常温下为固体的原料也可以与其它的原料混合而形成液体,一起进行纯化处理。接触方法可以举出连续地浸渍非水溶剂的方法(下面称为连续法),或向非水溶剂中添加吸附剂、静置或搅拌的方法(下面称为间歇法)。连续法的情况下,接触时间以液时空速(LHSV)计优选为0.1~5/小时。此外,接触温度优选为10~60℃。间歇法的情况下,优选相对于非水溶剂添加0.1~30质量%的吸附剂,处理0.25小时~24小时。If the adsorption treatment can be carried out in a liquid state, it can be dissolved in a non-aqueous solvent such as alumina, activated carbon, silica gel, molecular sieve (trade name) 4A and/or molecular sieve 5A or by an adsorbent that does not react with a non-aqueous solvent. Carry out purification treatment. At this time, raw materials that are liquid at normal temperature, such as dimethyl carbonate, can also be purified separately, and raw materials that are solid at normal temperature, such as ethylene carbonate, can also be mixed with other raw materials to form a liquid, and they can be purified together. . Examples of the contact method include a method of continuously immersing the non-aqueous solvent (hereinafter referred to as a continuous method), or a method of adding an adsorbent to the non-aqueous solvent and leaving it to stand or stir (hereinafter referred to as a batch method). In the case of a continuous method, the contact time is preferably 0.1 to 5 hours per hour in terms of liquid hourly space velocity (LHSV). In addition, the contact temperature is preferably 10 to 60°C. In the case of the batch method, it is preferable to add 0.1 to 30% by mass of the adsorbent to the non-aqueous solvent, and to treat for 0.25 hours to 24 hours.
此外,碳酸亚乙酯等常温下为固体的原料也可以进行结晶析出处理。结晶析出可以使用乙腈、丙酮、甲苯等溶剂进行。In addition, raw materials that are solid at normal temperature, such as ethylene carbonate, can also be subjected to the crystallization treatment. Crystallization can be carried out using solvents such as acetonitrile, acetone, and toluene.
该纯化条件优选根据使用的原料的种类或纯度、目标的水或醇类的含量来适当调节。The purification conditions are preferably adjusted appropriately according to the type and purity of the raw material used, and the target water or alcohol content.
不添加氟化氢(HF)来制备本发明(电解液[6])的非水电解液时,在用于非水电解液的非水溶剂中含有水或醇类。即,添加水或醇类后使用或者不除去水或醇类来使用。优选含有醇类特别是一元或二元醇类。作为醇类,没有特别限定,烷基的种类或醇的元数也没有特别限定,具体地说,例如可以举出甲醇、乙醇、正丙醇、异丙醇、正丁醇、叔丁醇等一元醇类;乙二醇或丙二醇等二元醇类;甘油等三元醇类等作为优选的醇类。作为添加时特别优选的“水或醇类”,可以举出甲醇、乙醇、乙二醇、丙二醇等。When the nonaqueous electrolytic solution of the present invention (electrolyte solution [6]) is prepared without adding hydrogen fluoride (HF), water or alcohols are contained in the nonaqueous solvent used for the nonaqueous electrolytic solution. That is, it is used after adding water or alcohols or is used without removing water or alcohols. Alcohols are preferably contained, especially monohydric or dihydric alcohols. The alcohol is not particularly limited, nor is the type of the alkyl group or the number of alcohols, and specific examples include methanol, ethanol, n-propanol, isopropanol, n-butanol, tert-butanol, etc. Monohydric alcohols; dihydric alcohols such as ethylene glycol and propylene glycol; trihydric alcohols such as glycerin, and the like are preferable alcohols. Methanol, ethanol, ethylene glycol, propylene glycol, etc. are mentioned as "water or alcohols" which are especially preferable at the time of addition.
在使用的电解液用的非水溶剂中,使用因其制造过程等原因而混入的醇类从生产性、成本等方面考虑在工业上是优选的。特别优选在使用的优选的非水溶剂中含有甲醇、乙醇、乙二醇或丙二醇。In the non-aqueous solvent for the electrolytic solution to be used, it is industrially preferable to use alcohols mixed due to reasons such as the production process, etc., from the viewpoints of productivity, cost, and the like. Particular preference is given to using methanol, ethanol, ethylene glycol or propylene glycol in the preferred non-aqueous solvents used.
在用于本发明的非水电解液(电解液[6])的非水溶剂中,希望水或醇类的含量为3ppm以上、优选为10ppm以上、更优选为20ppm以上、进一步优选为30ppm以上,其上限为150ppm以下、优选为130ppm以下、更优选为120ppm以下、进一步优选为100ppm以下。如果非水溶剂中的水或醇类的含量过少,则有时不能充分得到作为本发明特征的高输出功率特性,如果过多,则循环特性或高温保存特性有时变差。In the non-aqueous solvent used in the non-aqueous electrolytic solution (electrolyte [6]) of the present invention, it is desirable that the content of water or alcohols is 3 ppm or more, preferably 10 ppm or more, more preferably 20 ppm or more, further preferably 30 ppm or more , the upper limit thereof is 150 ppm or less, preferably 130 ppm or less, more preferably 120 ppm or less, and still more preferably 100 ppm or less. If the content of water or alcohol in the non-aqueous solvent is too small, the high output characteristics characteristic of the present invention may not be sufficiently obtained, and if too large, the cycle characteristics and high-temperature storage characteristics may deteriorate.
其中,作为一元醇类,在非水溶剂中优选为5ppm以上、更优选为10ppm以上、进一步优选为15ppm以上,且优选为100ppm以下、更优选为80ppm以下、进一步优选为50ppm以下。此外,作为二元醇类,在非水溶剂中为3ppm以上、优选为10ppm以上、更优选为15ppm以上、进一步优选为20ppm以上,且优选为100ppm以下、更优选为90ppm以下、进一步优选为80ppm以下、特别优选为70ppm以下。此外,水在非水溶剂中为3ppm以上、优选为5ppm以上、更优选为10ppm以上,并且优选为100ppm以下、更优选为80ppm以下、进一步优选为70ppm以下。Among them, the monohydric alcohol is preferably 5 ppm or more, more preferably 10 ppm or more, even more preferably 15 ppm or more, and preferably 100 ppm or less, more preferably 80 ppm or less, and still more preferably 50 ppm or less in the non-aqueous solvent. In addition, as glycols, in the non-aqueous solvent, it is 3 ppm or more, preferably 10 ppm or more, more preferably 15 ppm or more, still more preferably 20 ppm or more, and is preferably 100 ppm or less, more preferably 90 ppm or less, still more preferably 80 ppm or less, particularly preferably 70 ppm or less. In addition, water in the non-aqueous solvent is 3 ppm or more, preferably 5 ppm or more, more preferably 10 ppm or more, and preferably 100 ppm or less, more preferably 80 ppm or less, still more preferably 70 ppm or less.
如果上述一定量的氟化氢(HF)和上述特定化合物在非水电解液中共存,则不会对循环特性带来不良影响,并可以提高锂二次电池的输出功率。If the above-mentioned certain amount of hydrogen fluoride (HF) and the above-mentioned specific compound coexist in the non-aqueous electrolyte solution, the output of the lithium secondary battery can be improved without adversely affecting the cycle characteristics.
在本发明的非水电解液中通过共存一定量的氟化氢(HF)和特定化合物不会对循环特性带来不良影响并提高输出功率的理由虽然还不明确,但可认为是如下理由。另外,本发明不限于下述作用原理。即,上述特定化合物有可能具有某种程度的输出功率提高效果,而不管氟化氢(HF)的含量如何。认为这是由于特定化合物对电池的电极有某种作用,降低了与锂离子的进出有关的反应电阻。其中,氟化氢(HF)有可能起到增强或传递该作用的功能。例如,特定化合物和氟化氢(HF)一起作用于电极或特定化合物作用于电极时氟化氢(HF)发挥媒介的功能。另外,认为这样赋予功能的氟化氢(HF)可以在电池内稳定地存在,从而不易带来循环特性的降低等不良影响。The reason why the output is improved without adversely affecting cycle characteristics by coexisting a certain amount of hydrogen fluoride (HF) and a specific compound in the non-aqueous electrolyte solution of the present invention is not clear, but it is considered to be as follows. In addition, the present invention is not limited to the principle of action described below. That is, the above-mentioned specific compound is likely to have an output improving effect to some extent regardless of the content of hydrogen fluoride (HF). This is considered to be because the specific compound acts on the electrodes of the battery to lower the reaction resistance related to the entry and exit of lithium ions. Among them, hydrogen fluoride (HF) may function to enhance or transmit this effect. For example, hydrogen fluoride (HF) functions as a mediator when a specific compound and hydrogen fluoride (HF) act together on the electrode or when the specific compound acts on the electrode. In addition, it is considered that the hydrogen fluoride (HF) provided with such a function can stably exist in the battery, so that it is unlikely to cause adverse effects such as a decrease in cycle characteristics.
<电解液[7]><Electrolyte [7]>
在上述非水电解液中,优选在电解液中含有碳酸亚乙烯酯,并且该碳酸亚乙烯酯的含量为电解液总质量的0.001质量%~3质量%的范围(电解液[7])。Among the non-aqueous electrolytic solutions, it is preferable that vinylene carbonate is contained in the electrolytic solution, and the content of the vinylene carbonate is in the range of 0.001% by mass to 3% by mass of the total mass of the electrolytic solution (electrolyte solution [7]).
本发明(电解液[7])中的“锂盐”、“非水溶剂”、“特定化合物”、“其它化合物”的种类及其含量、使用条件、非水电解液的制备方法等与上述相同。The types and contents of "lithium salt", "non-aqueous solvent", "specific compound" and "other compounds" in the present invention (electrolyte [7]), use conditions, preparation method of non-aqueous electrolytic solution, etc. are the same as those mentioned above same.
本发明的非水电解液,如上所述,其特征在于含有碳酸亚乙烯酯。在本发明中,碳酸亚乙烯酯在全部非水电解液中所占的比率通常为0.001质量%以上、优选为0.01质量%以上、更优选为0.1质量%以上。另外,通常为3质量%以下、优选为2.8质量%以下、更优选为2.5质量%以下。如果碳酸亚乙烯酯的浓度过低,则有时难以得到循环特性的改善效果,另一方面,如果浓度过高,则有时导致电池的低温特性降低。The non-aqueous electrolytic solution of the present invention is characterized by containing vinylene carbonate as described above. In the present invention, the ratio of vinylene carbonate to the entire non-aqueous electrolytic solution is usually 0.001% by mass or more, preferably 0.01% by mass or more, more preferably 0.1% by mass or more. Moreover, it is usually 3 mass % or less, Preferably it is 2.8 mass % or less, More preferably, it is 2.5 mass % or less. If the concentration of vinylene carbonate is too low, it may be difficult to obtain the effect of improving the cycle characteristics. On the other hand, if the concentration is too high, the low-temperature characteristics of the battery may be lowered.
非水电解液中的碳酸亚乙烯酯的含量相对于上述特定化合物的含量没有特别限定,以质量比计优选为0.01以上、更优选为0.1以上、特别优选为0.3以上,另外,其上限优选为300以下、更优选为100以下、特别优选为30以下。如果明显偏离该范围,则有时不能同时实现优异的循环特性和低温特性这样的目的。The content of vinylene carbonate in the non-aqueous electrolytic solution is not particularly limited with respect to the content of the above-mentioned specific compound. It is preferably 0.01 or more, more preferably 0.1 or more, and particularly preferably 0.3 or more in terms of mass ratio. In addition, the upper limit is preferably 300 or less, more preferably 100 or less, particularly preferably 30 or less. If it deviates significantly from this range, the goals of excellent cycle characteristics and low-temperature characteristics may not be achieved at the same time.
对于通过将碳酸亚乙烯酯与二氟磷酸盐等上述特定化合物组合使用,即使碳酸亚乙烯酯为少量,循环特性也提高的理由虽然还不明确,但认为其原因在于:二氟磷酸盐等特定化合物抑制了由于电池的充放电而在正极消耗的碳酸亚乙烯酯的量,碳酸亚乙烯酯可以没有浪费地在负极形成涂膜;以及通过二氟磷酸盐等特定化合物和碳酸亚乙烯酯混在一起,在负极形成的涂膜的质发生变化,形成薄、低电阻并且可以进一步大幅抑制电解质锂盐的分解的优质的涂膜。从而认为还可以实现低温特性的改善。The reason why cycle characteristics are improved even with a small amount of vinylene carbonate by using the above-mentioned specific compounds such as vinylene carbonate in combination with difluorophosphate is not clear, but it is considered that the reason is that specific compounds such as difluorophosphate The compound suppresses the amount of vinylene carbonate consumed at the positive electrode due to charge and discharge of the battery, and vinylene carbonate can form a coating film on the negative electrode without waste; and by mixing specific compounds such as difluorophosphate and vinylene carbonate , the quality of the coating film formed on the negative electrode changes, forming a thin, low-resistance high-quality coating film that can further significantly inhibit the decomposition of the electrolyte lithium salt. Thus, it is considered that improvement in low-temperature characteristics can also be achieved.
<电解液[8]><Electrolyte [8]>
在上述非水电解液中,优选在电解液中进一步含有选自上述通式(4)所示的化合物、含有氮和/或硫的杂环化合物、环状羧酸酯、含氟环状碳酸酯中的至少一种的化合物,且其在全部非水电解液中的含量范围为0.001质量%~5质量%(电解液[8])。In the above-mentioned non-aqueous electrolytic solution, it is preferable that the electrolytic solution further contains compounds selected from the above-mentioned general formula (4), heterocyclic compounds containing nitrogen and/or sulfur, cyclic carboxylic acid esters, fluorine-containing cyclic carbonic acids At least one compound in the ester, and its content in the whole non-aqueous electrolytic solution ranges from 0.001% by mass to 5% by mass (electrolyte solution [8]).
本发明(电解液[8])中的“锂盐”、“非水溶剂”、“特定化合物”、“其它化合物”的种类及其含量、使用条件、非水电解液的制备方法等与上述相同。The types and contents of "lithium salt", "non-aqueous solvent", "specific compound" and "other compounds" in the present invention (electrolyte [8]), use conditions, preparation method of non-aqueous electrolyte, etc. are the same as those mentioned above same.
本发明(电解液[8])的非水电解液中,进一步含有选自上述通式(4)所示的化合物、含有氮和/或硫的杂环化合物、环状羧酸酯、含氟环状碳酸酯中的至少一种的化合物(下面有时将它们简称为“特定化合物B”),且其在全部非水电解液中的含量范围为0.001质量%~5质量%。In the non-aqueous electrolytic solution of the present invention (electrolyte [8]), it further contains a compound selected from the above general formula (4), a heterocyclic compound containing nitrogen and/or sulfur, a cyclic carboxylate, a fluorine-containing At least one compound of cyclic carbonates (hereinafter sometimes referred to simply as "specific compound B"), and its content in the entire non-aqueous electrolytic solution ranges from 0.001% by mass to 5% by mass.
[[通式(4)所示的化合物]][[Compound represented by general formula (4)]]
上述通式(4)中,R9~R12表示可以彼此相同或不同的由选自H、C、N、O、F、S和P中的一种以上的元素构成的基团。In the above general formula (4), R 9 to R 12 represent groups composed of one or more elements selected from the group consisting of H, C, N, O, F, S, and P, which may be the same or different from each other.
作为这些原子的存在方式,具体地说,优选存在于氢原子、氟原子、烷基、环烷基、链烯基、炔基、卤代烷基、烷氧基、羰基、羰氧基、氧基羰基、氧基羰氧基、磺酰基、氧基磺酰基、磺酰氧基、磷酰基、氧膦基等中。此外,通式(4)所示化合物的分子量优选为500以下、其中更优选为300以下、进一步优选为200以下。As the presence of these atoms, specifically, hydrogen atoms, fluorine atoms, alkyl groups, cycloalkyl groups, alkenyl groups, alkynyl groups, haloalkyl groups, alkoxy groups, carbonyl groups, carbonyloxy groups, and oxycarbonyl groups are preferably present. , Oxycarbonyloxy, sulfonyl, oxysulfonyl, sulfonyloxy, phosphoryl, phosphinyl, etc. In addition, the molecular weight of the compound represented by the general formula (4) is preferably 500 or less, more preferably 300 or less, and even more preferably 200 or less.
作为通式(4)所示化合物的具体例子,可以举出乙烯基碳酸亚乙酯、二乙烯基碳酸亚乙酯、碳酸甲基乙烯基酯、碳酸乙基乙烯基酯、碳酸丙基乙烯基酯、碳酸二乙烯基酯、碳酸烯丙基甲基酯、碳酸烯丙基乙基酯、碳酸烯丙基丙基酯、碳酸二烯丙基酯等碳酸酯类;乙酸乙烯酯、丙酸乙烯酯、丙烯酸乙烯酯、丁烯酸乙烯酯、甲基丙烯酸乙烯酯、乙酸烯丙酯、丙酸烯丙酯、丙烯酸甲酯、丙烯酸乙酯、丙烯酸丙酯、甲基丙烯酸甲酯、甲基丙烯酸乙酯、甲基丙烯酸丙酯等酯类;二乙烯基砜、甲基乙烯基砜、乙基乙烯基砜、丙基乙烯基砜、二烯丙基砜、烯丙基甲基砜、烯丙基乙基砜、烯丙基丙基砜等砜类;二乙烯基亚硫酸酯、甲基乙烯基亚硫酸酯、乙基乙烯基亚硫酸酯、二烯丙基亚硫酸酯等亚硫酸酯类;乙烯基甲磺酸酯、乙烯基乙磺酸酯、烯丙基甲磺酸酯、烯丙基乙磺酸酯、甲基乙烯基磺酸酯、乙基乙烯基磺酸酯等磺酸酯类;二乙烯基硫酸酯、甲基乙烯基硫酸酯、乙基乙烯基硫酸酯、二烯丙基硫酸酯等硫酸酯类等。其中,特别优选乙烯基碳酸亚乙酯、二乙烯基碳酸亚乙酯、乙酸乙烯酯、丙酸乙烯酯、丙烯酸乙烯酯、二乙烯基砜、乙烯基甲磺酸酯等。Specific examples of the compound represented by the general formula (4) include vinyl ethylene carbonate, divinyl ethylene carbonate, methyl vinyl carbonate, ethyl vinyl carbonate, propyl vinyl carbonate, Ester, divinyl carbonate, allyl methyl carbonate, allyl ethyl carbonate, allyl propyl carbonate, diallyl carbonate and other carbonates; vinyl acetate, vinyl propionate ester, vinyl acrylate, vinyl crotonate, vinyl methacrylate, allyl acetate, allyl propionate, methyl acrylate, ethyl acrylate, propyl acrylate, methyl methacrylate, methacrylic acid Esters such as ethyl ester and propyl methacrylate; divinyl sulfone, methyl vinyl sulfone, ethyl vinyl sulfone, propyl vinyl sulfone, diallyl sulfone, allyl methyl sulfone, allyl Sulfones such as ethyl ethyl sulfone and allyl propyl sulfone; Sulfites such as divinyl sulfite, methyl vinyl sulfite, ethyl vinyl sulfite, diallyl sulfite ; Vinyl methanesulfonate, vinyl ethanesulfonate, allyl methanesulfonate, allyl ethanesulfonate, methyl vinylsulfonate, ethyl vinylsulfonate and other sulfonates Classes; divinyl sulfate, methyl vinyl sulfate, ethyl vinyl sulfate, diallyl sulfate and other sulfates, etc. Among them, vinyl ethylene carbonate, divinyl ethylene carbonate, vinyl acetate, vinyl propionate, vinyl acrylate, divinyl sulfone, vinyl methanesulfonate, and the like are particularly preferable.
[[含有氮和/或硫的杂环化合物]][[Heterocyclic compounds containing nitrogen and/or sulfur]]
作为含有氮和/或硫的杂环化合物,没有特别限定,例如可以举出1-甲基-2-吡咯烷酮、1,3-二甲基-2-吡咯烷酮、1,5-二甲基-2-吡咯烷酮、1-乙基-2-吡咯烷酮、1-环己基-2-吡咯烷酮等吡咯烷酮类;3-甲基-2-噁唑烷酮、3-乙基-2-噁唑烷酮、3-环己基-2-噁唑烷酮等噁唑烷酮类;1-甲基-2-哌啶酮、1-乙基-2-哌啶酮等哌啶酮类;1,3-二甲基-2-咪唑啉酮、1,3-二乙基-2-咪唑啉酮等咪唑啉酮类;环丁砜、2-甲基环丁砜、3-甲基环丁砜等环丁砜类;环丁烯砜;亚乙基亚硫酸酯、亚丙基亚硫酸酯等亚硫酸酯类;1,3-丙磺酸内酯、1-甲基-1,3-丙磺酸内酯、3-甲基-1,3-丙磺酸内酯、1,4-丁磺酸内酯、1,3-丙烯磺酸内酯、1,4-丁烯磺酸内酯等磺酸内酯类等。其中,特别优选1-甲基-2-吡咯烷酮、1-甲基-2-哌啶酮、1,3-丙磺酸内酯、1,4-丁磺酸内酯、1,3-丙烯磺酸内酯、1,4-丁烯磺酸内酯等。The heterocyclic compound containing nitrogen and/or sulfur is not particularly limited, and examples include 1-methyl-2-pyrrolidone, 1,3-dimethyl-2-pyrrolidone, 1,5-dimethyl-2 -pyrrolidone, 1-ethyl-2-pyrrolidone, 1-cyclohexyl-2-pyrrolidone and other pyrrolidones; 3-methyl-2-oxazolidone, 3-ethyl-2-oxazolidone, 3- Cyclohexyl-2-oxazolidinone and other oxazolidinones; 1-methyl-2-piperidone, 1-ethyl-2-piperidone and other piperidones; 1,3-dimethyl -2-imidazolinone, 1,3-diethyl-2-imidazolinone and other imidazolinones; sulfolane, 2-methylsulfolane, 3-methylsulfolane and other sulfolanes; sulfolene; ethylene Sulfites such as sulfite and propylene sulfite; 1,3-propane sultone, 1-methyl-1,3-propane sultone, 3-methyl-1,3 - sultones such as propane sultone, 1,4-butane sultone, 1,3-propene sultone, 1,4-butene sultone, etc. Among them, 1-methyl-2-pyrrolidone, 1-methyl-2-piperidone, 1,3-propane sultone, 1,4-butane sultone, 1,3-propene sulfonate are particularly preferred. Acid lactone, 1,4-butene sultone, etc.
[[环状羧酸酯]][[Cyclic carboxylate]]
作为环状羧酸酯,没有特别限定,例如可以举出γ-丁内酯、γ-戊内酯、γ-己内酯、γ-庚内酯、γ-辛内酯、γ-壬内酯、γ-癸内酯、γ-十一烷内酯、γ-十二烷内酯、α-甲基-γ-丁内酯、α-乙基-γ-丁内酯、α-丙基-γ-丁内酯、α-甲基-γ-戊内酯、α-乙基-γ-戊内酯、α,α-二甲基-γ-丁内酯、α,α-二甲基-γ-戊内酯、δ-戊内酯、δ-己内酯、δ-辛内酯、δ-壬内酯、δ-癸内酯、δ-十一烷内酯、δ-十二烷内酯等。其中,特别优选γ-丁内酯、γ-戊内酯等。The cyclic carboxylic acid ester is not particularly limited, and examples thereof include γ-butyrolactone, γ-valerolactone, γ-caprolactone, γ-enantholactone, γ-octanolactone, and γ-nonanolactone , γ-decalactone, γ-undecanolactone, γ-dodecyl lactone, α-methyl-γ-butyrolactone, α-ethyl-γ-butyrolactone, α-propyl- γ-butyrolactone, α-methyl-γ-valerolactone, α-ethyl-γ-valerolactone, α,α-dimethyl-γ-butyrolactone, α,α-dimethyl- γ-valerolactone, δ-valerolactone, δ-caprolactone, δ-octylactone, δ-nonanolide, δ-decalactone, δ-undecanolactone, δ-dodecyl lactone Esters etc. Among them, γ-butyrolactone, γ-valerolactone, and the like are particularly preferable.
[[含氟环状碳酸酯]][[Fluorinated cyclic carbonate]]
作为含氟环状碳酸酯,没有特别限定,可以举出氟碳酸亚乙酯、二氟碳酸亚乙酯、三氟碳酸亚乙酯、四氟碳酸亚乙酯、三氟碳酸亚丙酯等。其中,特别优选氟碳酸亚乙酯等。The fluorine-containing cyclic carbonate is not particularly limited, and examples thereof include fluoroethylene carbonate, difluoroethylene carbonate, trifluoroethylene carbonate, tetrafluoroethylene carbonate, and trifluoropropylene carbonate. Among them, fluoroethylene carbonate and the like are particularly preferable.
特定化合物B,即选自通式(4)所示的化合物、含有氮和/或硫的杂环化合物、环状羧酸酯、含氟环状碳酸酯中的至少一种的化合物可以单独使用一种,也可以以任意的组合和比率组合使用2种以上的化合物。此外,特定化合物B中,即使是上述分别分类的化合物,也可以单独使用一种或以任意的组合和比率组合使用2种以上的化合物。Specific compound B, that is, at least one compound selected from compounds represented by general formula (4), nitrogen- and/or sulfur-containing heterocyclic compounds, cyclic carboxylic acid esters, and fluorine-containing cyclic carbonates can be used alone One, or two or more compounds may be used in combination in any combination and ratio. In addition, in the specific compound B, even if it is a compound classified separately above, one kind may be used alone or two or more kinds of compounds may be used in combination in arbitrary combinations and ratios.
非水电解液中这些特定化合物B相对于全部非水电解液的含有比率以总量计通常为0.001质量%以上、更优选为0.05质量%以上、进一步优选为0.1质量%以上。此外,以总量计,其上限通常为5质量%以下、更优选为4质量%以下、进一步优选为3质量%以下。如果特定化合物B的浓度过低,则有时难以得到循环特性和保存特性的改善效果,另一方面,如果浓度过高,则有时导致充放电效率的降低。The content ratio of the specific compound B in the non-aqueous electrolyte solution to the entire non-aqueous electrolyte solution is usually 0.001% by mass or more, more preferably 0.05% by mass or more, and still more preferably 0.1% by mass or more. In addition, the upper limit thereof is usually 5% by mass or less, more preferably 4% by mass or less, and still more preferably 3% by mass or less, based on the total amount. If the concentration of the specific compound B is too low, it may be difficult to obtain the effect of improving cycle characteristics and storage characteristics. On the other hand, if the concentration is too high, the charge-discharge efficiency may decrease.
使用本发明的非水电解液的非水电解质二次电池的低温放电特性优异且高温保存特性或循环特性优异的理由虽然还不明确,但可认为是如下理由,但本发明不限于下述作用原理。即,推测其原因如下:特定化合物B在初期的充电时在负极上受到还原分解,在负极表面上形成来源于特定化合物B的稳定的涂膜,可以提高保存特性、循环特性。但是该涂膜在低温下电阻的增大显著,存在低温放电特性降低的问题。通过与特定化合物A共存,抑制特定化合物B的过量反应,即使在低温下也形成锂离子透过性优异的稳定复合保护涂膜,从而提高低温放电特性并改善高温保存特性或循环特性。Although the reason why the nonaqueous electrolyte secondary battery using the nonaqueous electrolytic solution of the present invention is excellent in low-temperature discharge characteristics and high-temperature storage characteristics or cycle characteristics is not clear, it is considered to be the following reason, but the present invention is not limited to the following effects principle. That is, it is presumed that the reason is that the specific compound B undergoes reductive decomposition on the negative electrode during initial charging, and forms a stable coating film derived from the specific compound B on the surface of the negative electrode, thereby improving storage characteristics and cycle characteristics. However, the resistance of this coating film increases significantly at low temperatures, and there is a problem that low-temperature discharge characteristics are lowered. By coexisting with specific compound A, excessive reaction of specific compound B is suppressed, and a stable composite protective coating film with excellent lithium ion permeability is formed even at low temperatures, thereby improving low-temperature discharge characteristics and improving high-temperature storage characteristics or cycle characteristics.
进一步地认为是由于,二次电池的1个电池外壳中收纳的电池要素所具有的电池容量为3安培小时(Ah)以上时和/或二次电池的直流电阻成分为10毫欧(Ω)以下时,直流电阻成分的贡献减少,与直流电阻成分的贡献大的电池相比,容易表现出该非水电解液本来的效果。It is further considered that the battery element contained in one battery case of the secondary battery has a battery capacity of 3 ampere hours (Ah) or more and/or the DC resistance component of the secondary battery is 10 milliohms (Ω) In the following cases, the contribution of the DC resistance component is reduced, and the original effect of the non-aqueous electrolytic solution is more likely to be exhibited compared with a battery with a large contribution of the DC resistance component.
<电解液[9]><Electrolyte [9]>
在上述非水电解液中,优选在电解液中进一步含有过充电防止剂(电解液[9])。In the above non-aqueous electrolytic solution, it is preferable to further contain an overcharge preventing agent (electrolytic solution [9]) in the electrolytic solution.
本发明(电解液[9])中的“锂盐”、“非水溶剂”、“特定化合物”、“其它化合物”的种类及其含量、使用条件、非水电解液的制备方法等与上述相同。The types and contents of "lithium salt", "non-aqueous solvent", "specific compound" and "other compounds" in the present invention (electrolyte [9]), use conditions, preparation method of non-aqueous electrolyte, etc. are the same as those mentioned above same.
本发明(电解液[9])的二次电池用非水电解液的特征在于,含有过充电防止剂。作为过充电防止剂,没有特别限定,但优选下述(1)、(2)或(3)所示的化合物等。The non-aqueous electrolytic solution for secondary batteries of the present invention (electrolyte solution [9]) is characterized by containing an overcharge preventing agent. Although it does not specifically limit as an overcharge inhibitor, The compound etc. which are shown by following (1), (2) or (3) are preferable.
(1)可以被烷基和/或氟原子取代的联苯、三联苯、二苯基醚或二苯并呋喃;(1) Biphenyls, terphenyls, diphenyl ethers or dibenzofurans which may be substituted by alkyl and/or fluorine atoms;
(2)三联苯的部分氢化物;(2) Partial hydrides of terphenyls;
(3)被叔烷基、环烷基、氟原子和/或甲氧基取代的苯。(3) Benzene substituted with a tertiary alkyl group, cycloalkyl group, fluorine atom and/or methoxy group.
作为(1)的化合物,没有特别限定,例如可以举出联苯、烷基联苯、三联苯等苯环连接化合物类;2-氟联苯等含氟苯环连接化合物类;二苯基醚等芳香族醚类;二苯并呋喃等芳香族杂环连接化合物类等。The compound of (1) is not particularly limited, and examples thereof include benzene ring linking compounds such as biphenyl, alkylbiphenyl, and terphenyl; fluorine-containing benzene ring linking compounds such as 2-fluorobiphenyl; diphenyl ether Aromatic ethers such as dibenzofuran; aromatic heterocyclic linking compounds such as dibenzofuran, etc.
作为(3)的化合物,没有特别限定,例如可以举出环己基苯、叔丁基苯、叔戊基苯等(环)烷基苯类;邻环己基氟苯、对环己基氟苯等氟原子取代苯类;2,4-二氟苯甲醚、2,5-二氟苯甲醚、2,6-二氟苯甲醚、3,5-二氟苯甲醚等含氟苯甲醚类等。The compound of (3) is not particularly limited, and examples thereof include (cyclo)alkylbenzenes such as cyclohexylbenzene, tert-butylbenzene, and tert-amylbenzene; Atom-substituted benzenes; 2,4-difluoroanisole, 2,5-difluoroanisole, 2,6-difluoroanisole, 3,5-difluoroanisole and other fluorine-containing anisole class etc.
作为优选的具体例子,可以举出联苯、烷基联苯、三联苯、三联苯的部分氢化物、环己基苯、叔丁基苯、叔戊基苯、二苯基醚、二苯并呋喃等芳香族化合物。这些化合物由于本发明中的高温保存后的速度特性改善效果增大,因而特别优选。Preferable specific examples include biphenyl, alkylbiphenyl, terphenyl, partially hydrogenated terphenyl, cyclohexylbenzene, tert-butylbenzene, tert-amylbenzene, diphenyl ether, dibenzofuran and other aromatic compounds. These compounds are particularly preferable because the effect of improving the speed characteristics after high-temperature storage in the present invention is increased.
过充电防止剂可以组合使用两种以上。组合使用两种以上时,特别优选将环己基苯或三联苯(或其部分氢化物)和叔丁基苯或叔戊基苯组合使用。The overcharge preventing agent can be used in combination of 2 or more types. When two or more are used in combination, it is particularly preferable to use cyclohexylbenzene or terphenyl (or a partially hydrogenated product thereof) and tert-butylbenzene or tert-amylbenzene in combination.
这里所说的三联苯的部分氢化物是指在三联苯的苯环的双键上部分地加成氢而得到的氢化物。三联苯的部分氢化物可以是单一化合物,也可以是含有多个化合物的混合物。例如,可以是具有不同的部分氢化率的2个以上三联苯的部分氢化物的混合物,也可以是部分氢化率相等的三联苯的部分氢化物。此外,可以为氢化的苯环的位置不同的氢化物的混合物,还可以是双键位置不同的混合物或含有结构异构体的混合物。The partial hydrogenated product of terphenyl referred to here refers to a hydrogenated product obtained by partially adding hydrogen to the double bond of the benzene ring of the terphenyl. The partial hydrogenated product of terphenyl may be a single compound or a mixture containing multiple compounds. For example, it may be a mixture of two or more partially hydrogenated terphenyls having different partial hydrogenation rates, or may be partially hydrogenated terphenyls having equal partial hydrogenation rates. In addition, the hydrogenated benzene ring may be a mixture of hydrides having different positions, a double bond position may be different, or a mixture containing structural isomers may be used.
三联苯的部分氢化率是指,将未在三联苯的苯环的双键上加成氢的部分氢化率作为0%、将三联苯的完全氢化物即在全部双键上加成氢时(1摩尔的三联苯中加成18摩尔的氢原子时)的部分氢化率作为100%进行计算而得到的值,混合物时为每摩尔平均的值。例如,在1摩尔的三联苯上加成2摩尔的氢原子时,部分氢化率为11.1%(=2/18)。The partial hydrogenation rate of terphenyl refers to the case where the partial hydrogenation rate at which hydrogen is not added to the double bond of the benzene ring of the terphenyl is 0%, and the complete hydrogenation product of the terphenyl, that is, when hydrogen is added to all the double bonds ( When 18 moles of hydrogen atoms are added to 1 mole of terphenyl), the partial hydrogenation rate is calculated as 100%, and in the case of a mixture, it is an average value per mole. For example, when 2 moles of hydrogen atoms are added to 1 mole of terphenyl, the partial hydrogenation rate is 11.1% (=2/18).
使用上述定义的部分氢化率时,本发明中使用的三联苯的部分氢化物的部分氢化率可以取超过0%且小于100%的值。三联苯的部分氢化物可以含有三联苯(部分氢化率0%)、间三联苯的完全氢化物(部分氢化率100%),混合物的摩尔平均部分氢化率优选取超过0%且小于100%的值。从电池的保存特性和在电解液中的溶解性方面考虑,三联苯的部分氢化率优选为30~70%、更优选为35~60%。此外,作为三联苯或三联苯的部分氢化物,没有特别限定,但特别优选为间三联苯或间三联苯的部分氢化物。When the partial hydrogenation rate defined above is used, the partial hydrogenation rate of the partially hydrogenated product of terphenyl used in the present invention can take a value exceeding 0% and less than 100%. The partial hydrogenation of terphenyl can contain the complete hydrogenation of terphenyl (partial hydrogenation rate 0%), m-terphenyl (partial hydrogenation rate 100%), and the molar average partial hydrogenation rate of the mixture is preferably more than 0% and less than 100%. value. The partial hydrogenation rate of terphenyl is preferably 30 to 70%, more preferably 35 to 60%, from the standpoint of storage properties of the battery and solubility in the electrolytic solution. Moreover, although it does not specifically limit as a terphenyl or the partially hydrogenated product of terphenyl, It is especially preferable that it is a partial hydrogenated product of m-terphenyl or m-terphenyl.
非水电解液中的这些过充电防止剂的比率分别通常为0.01质量%以上、优选为0.1质量%以上、特别优选为0.2质量%以上,其上限通常为5质量%以下、优选为3质量%以下、特别优选为2质量%以下。如果小于下限,则有时不能充分确保过充电时的安全性,如果超过上限,则有时即使与下述特定化合物混合也不能形成保存特性优异的电池。The ratios of these overcharge preventing agents in the non-aqueous electrolytic solution are usually 0.01% by mass or more, preferably 0.1% by mass or more, particularly preferably 0.2% by mass or more, and the upper limit is usually 5% by mass or less, preferably 3% by mass or less, particularly preferably 2% by mass or less. If it is less than the lower limit, it may not be possible to ensure sufficient safety during overcharge, and if it exceeds the upper limit, it may not be possible to form a battery with excellent storage characteristics even if it is mixed with the specific compound described below.
对于存在上述特定化合物时不易产生由于过充电添加剂所导致的保存后速率特性的变差的原因虽然还不明确,但是推测为如下原因。一般来说,过充电防止剂虽然可以在过充电时在正极表面上形成聚合涂膜而大幅提高电池的内部电阻,或者通过该聚合时产生的气体使电池罐内部的电流阻断装置工作,由此提高二次电池的安全性等,但是即使在充电状态电池的高温保存时也生成一部分该聚合涂膜,从而成为电池的放电容量降低或速率特性变差的原因。如果在其中存在本发明中指定的特定化合物,则化合物作用于正极表面,通过形成弱屏蔽,抑制通常的充电状态下过充电防止剂和正极活性物质的反应。但是,在电池过充电、正极成为过度活性的状态时,该弱屏蔽破坏或者正极和过充电防止剂的反应加速,超过通过屏蔽形成的阻碍,由此进行聚合反应,可以确保目的的过充电时安全性。The reason why the deterioration of the rate characteristic after storage due to the overcharge additive is less likely to occur in the presence of the above-mentioned specific compound is not clear, but it is presumed to be as follows. In general, the overcharge preventing agent can form a polymerized coating film on the surface of the positive electrode during overcharging to greatly increase the internal resistance of the battery, or the current blocking device inside the battery can can be operated by the gas generated during the polymerization. This improves the safety of the secondary battery, etc., but even when the battery is stored at a high temperature in a charged state, a part of the polymerized coating film is formed, which causes a decrease in the discharge capacity of the battery or deterioration of the rate characteristic. If the specific compound specified in the present invention exists therein, the compound acts on the surface of the positive electrode to suppress the reaction of the overcharge preventing agent and the positive electrode active material in a normal charged state by forming a weak barrier. However, when the battery is overcharged and the positive electrode is in an excessively active state, the weak barrier is destroyed or the reaction between the positive electrode and the overcharge preventive agent is accelerated, exceeding the barrier formed by the barrier, thereby proceeding to the polymerization reaction, and the purpose of overcharging can be ensured. safety.
<电池设计(电池结构)><Battery Design (Battery Structure)>
下面对本发明的锂二次电池的结构(电池结构)进行详细的说明。Next, the structure (battery structure) of the lithium secondary battery of the present invention will be described in detail.
本发明的可充放电的锂二次电池至少由可以吸留和放出锂离子的正极和负极、上述非水电解液、配置在正极和负极之间的微多孔膜隔板、集电端子以及外壳等构成。根据需要还可以在电池的内部和/或电池的外部安装保护元件。而且,本说明书中,有时将本发明的锂二次电池的结构(电池结构)的特征部分称为结构[1]~结构[6]。The rechargeable lithium secondary battery of the present invention at least consists of a positive electrode and a negative electrode capable of absorbing and releasing lithium ions, the above-mentioned non-aqueous electrolyte, a microporous membrane separator arranged between the positive electrode and the negative electrode, a collector terminal and a casing And so on. Protection elements can also be installed inside and/or outside the battery as required. In addition, in this specification, the characteristic part of the structure (battery structure) of the lithium secondary battery of this invention may be called structure [1] - structure [6].
[放电容量](结构[3])[Discharge capacity] (Structure[3])
本发明的锂二次电池中,如果二次电池的1个电池外壳中收纳的电池要素所具有的电容(将电池由满充电状态放电至放电状态时的电容)(有时将其简称为“电池容量”)为3安培小时(Ah)以上,则由于输出功率特性的提高效果增大而优选。因此,正极板优选设计成放电容量在满充电下为3安培小时(Ah)~20Ah,更优选为4Ah~10Ah(结构[3])。低于3Ah时,释放大电流时,由于电极反应电阻所导致的电压降低增大,功率利用系数有时变差。另一方面,大于20Ah时,虽然电极反应电阻减小,功率利用系数变好,但是由于脉冲充放电时的电池内部放热所形成的温度分布大,反复充放电的耐久性差,并且对于过充电或内部短路等异常时的急剧发热的放热效率也变差,内压上升,气体释放阀工作现象(阀工作)不停止,以至于发生电池内容物向外急剧喷出的现象(破裂)的几率有可能提高In the lithium secondary battery of the present invention, if the capacity of the battery element contained in one battery case of the secondary battery (the capacity when the battery is discharged from a fully charged state to a discharged state) (sometimes referred to as "battery" for short) The capacity ") is 3 ampere-hours (Ah) or more, since the effect of improving output power characteristics is increased, it is preferable. Therefore, the positive plate is preferably designed to have a discharge capacity of 3 ampere hours (Ah) to 20 Ah, more preferably 4 Ah to 10 Ah under full charge (structure [3]). When it is less than 3Ah, when a large current is released, the voltage drop due to the electrode reaction resistance increases, and the power utilization factor sometimes deteriorates. On the other hand, when it is greater than 20Ah, although the electrode reaction resistance decreases and the power utilization factor becomes better, the temperature distribution formed by the internal heat generation of the battery during pulse charge and discharge is large, the durability of repeated charge and discharge is poor, and the overcharge In case of abnormality such as internal short circuit or internal short circuit, the heat release efficiency of the sudden heat is also deteriorated, the internal pressure rises, and the operation phenomenon (valve operation) of the gas release valve does not stop, so that the probability of the phenomenon (rupture) that the battery contents are ejected rapidly outward It is possible to improve
[集电结构](结构[2]、结构[4])[Collector Structure] (Structure[2], Structure[4])
集电结构没有特别限定,但为了更有效地实现本发明的锂二次电池的输出功率特性的提高,需要形成降低配线部分或接合部分的电阻的结构。该内部电阻小时,可特别优异地发挥使用上述非水电解液的效果。The current collecting structure is not particularly limited, but in order to more effectively improve the output characteristics of the lithium secondary battery of the present invention, it is necessary to form a structure that reduces the resistance of the wiring portion or the junction portion. When this internal resistance is small, the effect of using the above-mentioned non-aqueous electrolytic solution can be exhibited particularly excellently.
电极组为后述叠层结构时,优选使用将各电极层的集电片(集電タブ)捆束连接在端子上而形成的结构。由于一块电极面积增大时,内部电阻增大,因此优选在电极内设置多个集电片来降低电阻。电极组为后述的卷绕结构时,在正极和负极上分别设置多个集电片,捆束在端子上,由此可以降低内部电阻。When the electrode group has a laminated structure described later, it is preferable to use a structure in which current collecting tabs of each electrode layer are bundled and connected to terminals. Since the internal resistance increases when the area of one electrode increases, it is preferable to provide a plurality of collector sheets in the electrode to reduce the resistance. When the electrode group has a wound structure described later, a plurality of current collector sheets are respectively provided on the positive electrode and the negative electrode, and bundled to the terminals, whereby the internal resistance can be reduced.
通过使上述集电结构最优化,可以尽可能地减小内部电阻。以大电流使用的电池中,用10kHz交流法测定的阻抗(下面简称为“直流电阻成分”)优选为20毫欧(mΩ)以下、更优选为10毫欧(mΩ)以下、进一步优选为5毫欧(mΩ)以下(结构[2])。另一方面,若直流电阻成分小于0.1毫欧,则虽然高输出功率特性得到提高,但是使用的集电结构材料所占的比率增加,电池容量有时减少。By optimizing the above current collecting structure, the internal resistance can be reduced as much as possible. In a battery used with a large current, the impedance measured by the 10 kHz AC method (hereinafter referred to simply as "DC resistance component") is preferably 20 milliohms (mΩ) or less, more preferably 10 milliohms (mΩ) or less, and even more preferably 5 milliohms (mΩ). Below milliohms (mΩ) (structure [2]). On the other hand, if the DC resistance component is less than 0.1 milliohm, the high output characteristics are improved, but the ratio of the current collecting structure material used increases, and the battery capacity may decrease.
阻抗的测定中,使用SOLAR(ソ一ラ一トロン)公司制造的电池测定装置1470和SOLAR公司制造的频率响应analyzer 1255B作为测定装置,对在5mV的偏压下施加10kHz的交流时的电阻进行测定,作为直流电阻成分。In the measurement of impedance, a battery measuring device 1470 manufactured by SOLAR (Solatron) Co., Ltd. and a frequency response analyzer 1255B manufactured by SOLAR Co., Ltd. were used as the measuring device, and the resistance when an alternating current of 10 kHz was applied under a bias voltage of 5 mV was measured. , as the DC resistance component.
本发明中的上述非水电解液对于降低与锂在电极活性物质中的进出相关的反应电阻有效果,从而可认为其成为可以实现优异的输出功率特性的主要原因。但是,通常的直流电阻成分大的电池中,受直流电阻成分阻碍,反应电阻降低的效果不能100%反映到输出功率特性中。通过使用直流电阻成分小的电池可以改善该问题,从而可以充分发挥本发明的效果。The above non-aqueous electrolytic solution in the present invention is effective in reducing the reaction resistance related to the entry and exit of lithium into and out of the electrode active material, and this is considered to be the main reason why excellent output characteristics can be realized. However, in a normal battery with a large DC resistance component, the effect of reducing the reaction resistance cannot be 100% reflected in the output characteristics due to the hindrance of the DC resistance component. This problem can be improved by using a battery with a small direct-current resistance component, and the effect of the present invention can be fully exhibited.
此外,从制造发挥非水电解液的效果并具有高输出功率特性的电池的观点考虑,特别优选同时满足该条件和上述的在二次电池的1个电池外壳中收纳的电池要素所具有的电容(电池由满充电状态放电至放电状态时的电容)(电池容量)为3安培小时(Ah)以上的条件。In addition, from the viewpoint of manufacturing a battery that exerts the effect of the non-aqueous electrolyte and has high output characteristics, it is particularly preferable to satisfy this condition and the above-mentioned capacitance of the battery element contained in one battery case of the secondary battery at the same time. (The capacity when the battery is discharged from the fully charged state to the discharged state) (battery capacity) is a condition of 3 ampere hours (Ah) or more.
上述集电片和端子的连接优选通过点焊接、高频焊接或超声波焊接的任意一种来接合(结构[4])。这些焊接方法以往是电阻小、简易的焊接方法,但是由于长时间使用,焊接部分与非水电解液中的杂质或副产物等反应而劣化,直流电阻成分增加。但是,使用含有特定化合物的上述非水电解液时,可以在焊接部分形成稳定的涂膜,此外,可抑制非水电解液在正极中的副反应,所以即使在长时间使用时,焊接部分的劣化也不易进行,不会增加直流电阻成分并可以维持高输出功率。The connection between the collector tab and the terminal is preferably joined by any one of spot welding, high-frequency welding, or ultrasonic welding (structure [4]). These soldering methods have traditionally been simple soldering methods with low resistance, but due to long-term use, the soldered part reacts with impurities or by-products in the non-aqueous electrolyte and deteriorates, increasing the DC resistance component. However, when using the above-mentioned non-aqueous electrolyte solution containing a specific compound, a stable coating film can be formed on the welded part, and in addition, the side reaction of the non-aqueous electrolyte solution in the positive electrode can be suppressed, so even when used for a long time, the welded part Deterioration is also difficult to progress, and high output power can be maintained without increasing the DC resistance component.
[电池外壳1](结构5)[Battery Case 1] (Structure 5)
电池外壳的材质只要是对使用的非水电解液稳定的物质即可,没有特别限定。具体地说,可使用镍镀钢板、不锈钢、铝或铝合金、镁合金等金属类,或树脂和铝箔的叠层膜(层压膜)作为优选的材质。从轻量化方面考虑,特别优选使用铝或铝合金的金属或层压膜。The material of the battery case is not particularly limited as long as it is stable to the non-aqueous electrolytic solution used. Specifically, metals such as nickel-plated steel sheets, stainless steel, aluminum or aluminum alloys, and magnesium alloys, or laminated films (laminated films) of resin and aluminum foil can be used as preferable materials. From the viewpoint of weight reduction, it is particularly preferable to use a metal or a laminated film of aluminum or an aluminum alloy.
在本发明中,使用上述非水电解液时,特别优选使用铝或铝合金的金属(结构[5])。铝或铝合金为轻量、成型性高的材料,但是由于作为电池外壳长时间的使用时,与非水电解液中的杂质或副产物等反应而劣化,若变差则有可能降低外壳的强度或开孔。使用含有特定化合物的上述非水电解液时,可以在铝表面或铝合金表面上形成稳定的涂膜,此外,可以抑制非水电解液在正极上的副反应,所以即使长时间使用,外壳的劣化也不易进行。In the present invention, when the above-mentioned non-aqueous electrolytic solution is used, it is particularly preferable to use aluminum or an aluminum alloy metal (structure [5]). Aluminum or aluminum alloy is a material with light weight and high formability, but when it is used as a battery case for a long time, it will deteriorate due to the reaction with impurities or by-products in the non-aqueous electrolyte solution. strength or opening. When using the above-mentioned non-aqueous electrolyte solution containing a specific compound, a stable coating film can be formed on the aluminum surface or the aluminum alloy surface. In addition, the side reaction of the non-aqueous electrolyte solution on the positive electrode can be suppressed, so even if it is used for a long time Deterioration is also not easy to proceed.
对于使用上述金属类的外壳,可以举出通过激光焊接、电阻焊接、超声波焊接将金属熔接而形成密封密闭结构的外壳,或通过树脂制垫圈使用上述金属类形成铆接结构的外壳。对于使用上述层压膜的外壳,可以举出通过将树脂层彼此热熔合形成密封密闭结构的外壳等。为了提高密封性,可以在上述树脂层之间存在与层压膜中使用的树脂不同的树脂。特别是通过集电端子将树脂层热熔合而形成密闭结构时,由于形成金属与树脂的接合,因此优选使用具有极性基团的树脂或导入了极性基团的改性树脂作为存在于树脂层之间的树脂。Cases using the above metals include cases in which metals are fused by laser welding, resistance welding, or ultrasonic welding to form a hermetically sealed structure, or cases in which the above metals are riveted with a resin gasket. Examples of the case using the above laminated film include a case in which a hermetically sealed structure is formed by thermally fusing resin layers to each other. In order to improve sealability, a resin different from the resin used in the laminated film may be present between the above-mentioned resin layers. Especially when the resin layer is thermally fused through the collector terminal to form a closed structure, since a metal-resin bond is formed, it is preferable to use a resin having a polar group or a modified resin introduced with a polar group as the resin present in the resin. resin between layers.
[电池外壳2](结构[6])[Battery Case 2] (Structure[6])
本发明中,形成电池外壳的外壳材料中,优选电池的内面侧的至少一部分包含使用热塑性树脂而形成的片,并将该电极组装入其中,同时将该热塑性树脂层热封,由此可以密封该电池组(结构[6])。In the present invention, in the case material forming the battery case, it is preferable that at least a part of the inner surface side of the battery includes a sheet formed using a thermoplastic resin, and the electrode assembly is incorporated therein, and at the same time, the thermoplastic resin layer is heat-sealed, whereby sealing can be achieved. The battery pack (structure [6]).
为了实现电池轻量化,结构[6]情况的电池外壳的材质为轻量、且对于使用的非水电解液稳定的材质,由于必须容易且确实地密封电极组,因此电池的内面侧的至少一部分必须含有使用热塑性树脂而形成的片。In order to reduce the weight of the battery, the material of the battery case in the case of structure [6] is lightweight and stable to the non-aqueous electrolyte used, and since the electrode group must be easily and reliably sealed, at least a part of the inner surface of the battery A sheet formed using a thermoplastic resin must be included.
结构[6]中,通过将热塑性树脂层热封来密封该电极组,其中,所谓“热封”是指在使热塑性树脂层彼此密合的基础上,设定该热塑性树脂的熔点以上的温度,将该热塑性树脂层彼此粘合。优选使用具有带状的发热部,并且进行加压的同时可以加热的密封机。此外,电池的内面侧的至少一部分使用热塑性树脂,其中,“至少一部分”是指在片的外周部分含有仅可以密封该电极组的部分的区域,可以仅在热封部分使用热塑性树脂。从片的制作工序的效率方面考虑,优选电池的内面侧的整个片表面被热塑性树脂层覆盖。In structure [6], the electrode group is sealed by heat-sealing the thermoplastic resin layer, wherein the so-called "heat-sealing" refers to setting the temperature above the melting point of the thermoplastic resin on the basis of making the thermoplastic resin layers adhere to each other. , bonding the thermoplastic resin layers to each other. It is preferable to use a sealing machine that has a band-shaped heat generating portion and can heat while pressurizing. In addition, thermoplastic resin is used for at least a part of the inner surface of the battery, where "at least a part" means that only the electrode group can be sealed in the outer peripheral part of the sheet, and thermoplastic resin can be used only for the heat-sealed part. From the viewpoint of the efficiency of the sheet production process, it is preferable that the entire sheet surface on the inner side of the battery is covered with a thermoplastic resin layer.
另外,在结构[6]中,后述的微多孔膜隔板具有通过加热堵塞孔的性质时,从过充电等异常时的安全性方面考虑,外壳材料优选在电池的内面侧的至少一部分含有使用具有比该微多孔膜隔板的孔的堵塞开始温度高的熔点的热塑性树脂而形成的片。即,在过充电等中产生异常发热时,电池温度升高,如果超过外壳材料的热塑性树脂的熔点,则存在电池外壳破裂或产生电解液泄漏以至于起火的情况,但是如果微多孔膜隔板具有通过加热堵塞孔的性质,则由于在产生电解液从外壳材料泄漏之前微多孔膜隔板的孔堵塞,可以抑制更多的发热,因此不至于破裂、起火,所以优选。其中,所谓熔点是指通过JIS K7121测定的熔化温度。In addition, in the structure [6], when the microporous membrane separator described later has the property of clogging pores by heating, it is preferable that the casing material contains A sheet formed using a thermoplastic resin having a melting point higher than the clogging initiation temperature of pores of the microporous membrane separator. That is, when abnormal heat generation occurs during overcharging, etc., the temperature of the battery rises, and if it exceeds the melting point of the thermoplastic resin of the case material, there is a case where the battery case ruptures or electrolyte leakage occurs to cause a fire, but if the microporous membrane separator It is preferable that the pores of the microporous membrane separator be blocked by heating before the leakage of the electrolyte from the casing material occurs, and further heat generation can be suppressed, so that rupture and ignition are prevented. Here, the melting point refers to the melting temperature measured by JIS K7121.
作为结构[6]中的热塑性树脂,没有特别限定,可优选列举聚乙烯、聚丙烯、改性聚烯烃、聚烯烃共聚物等聚烯烃类;聚对苯二甲酸乙二醇酯等聚酯类;尼龙等聚酰胺类等。热塑性树脂可以使用一种,也可以使用2种以上。The thermoplastic resin in the structure [6] is not particularly limited, but preferably polyolefins such as polyethylene, polypropylene, modified polyolefin, and polyolefin copolymer; polyesters such as polyethylene terephthalate ; Nylon and other polyamides, etc. One type of thermoplastic resin may be used, or two or more types may be used.
作为该结构[6]中的“外壳材料的至少一部分”,可以仅使用热塑性树脂,但是优选使用热塑性树脂和热固化性树脂、弹性体、金属材料、玻璃纤维、碳纤维等的复合材料。另外,还可以含有填料等填充材料。作为复合材料,特别优选热塑性树脂层与铝、铁、铜、镍、钛、钼、金等单质金属或不锈钢、哈斯特洛伊耐蚀高镍合金等合金的叠层片,进一步优选与加工性优异的铝金属的叠层片。即,外壳材料进一步优选至少包括具有铝层和热塑性树脂层叠层而得到的叠层片。这些金属或合金可以以金属等的箔的形式使用,也可以以金属蒸镀膜的形式使用。As "at least a part of the housing material" in the structure [6], only thermoplastic resin may be used, but composite materials such as thermoplastic resin and thermosetting resin, elastomer, metal material, glass fiber, carbon fiber, etc. are preferably used. In addition, fillers such as fillers may also be contained. As a composite material, a laminated sheet of a thermoplastic resin layer and a single metal such as aluminum, iron, copper, nickel, titanium, molybdenum, or gold, or an alloy such as stainless steel or Hastelloy, is more preferable and has excellent processability laminated sheets of aluminum metal. That is, it is more preferable that the casing material includes at least a laminated sheet having an aluminum layer and a thermoplastic resin layer laminated. These metals or alloys may be used as foils of metals or the like, or may be used as metal vapor-deposited films.
在结构[6]中,使用上述外壳材料的外壳中,可以举出通过将树脂层热熔合而形成密封密闭结构的外壳等。为了提高密封性,可以在上述树脂层之间存在与外壳材料中使用的树脂不同的树脂。特别是通过集电端子将树脂层热熔合而形成密闭结构时,由于形成金属与树脂的接合,因此优选使用具有极性基团的树脂或导入了极性基团的改性树脂作为存在于树脂层之间的树脂。In the configuration [6], examples of the case using the above-mentioned case material include a case in which a resin layer is thermally fused to form a hermetically sealed structure, and the like. In order to improve sealing performance, a resin different from the resin used in the case material may exist between the above-mentioned resin layers. Especially when the resin layer is thermally fused through the collector terminal to form a closed structure, since a metal-resin bond is formed, it is preferable to use a resin having a polar group or a modified resin introduced with a polar group as the resin present in the resin. resin between layers.
在结构[6]中,外壳材料的厚度没有特别限定,但优选为0.03mm以上、更优选为0.04mm以上、进一步优选为0.05mm以上。此外,其上限优选为0.5mm以下、更优选为0.3mm以下、进一步优选为0.2mm以下。如果外壳材料比该范围薄,则强度减小,有可时容易受到变形、破碎等。另一方面,如果外装材料比该范围厚,则由于外壳的质量增加,有时达不到电池轻量化的目的。In structure [6], the thickness of the casing material is not particularly limited, but is preferably 0.03 mm or more, more preferably 0.04 mm or more, and still more preferably 0.05 mm or more. In addition, the upper limit thereof is preferably 0.5 mm or less, more preferably 0.3 mm or less, and still more preferably 0.2 mm or less. If the casing material is thinner than this range, the strength will decrease, and it may be easily deformed, broken, etc. On the other hand, if the exterior material is thicker than this range, the weight reduction of the battery may not be achieved due to the increase in the mass of the case.
使用上述外壳材料的结构[6]的电池具有轻量、形状的自由度高的优点,另一方面,若电池内部产生气体,则内压升高,外壳材料有时容易变形。本发明中的锂二次电池中,非水电解液中的特定化合物吸附在正极活性物质表面,由此可抑制正极的副反应,并抑制气体成分的产生,因此即使使用上述外壳材料,也未表现出上述缺点,仅表现出上述外壳材料的上述优点,所以优选。The battery of the structure [6] using the above case material has the advantages of light weight and high degree of freedom in shape. On the other hand, if gas is generated inside the battery, the internal pressure increases and the case material may be easily deformed. In the lithium secondary battery in the present invention, the specific compound in the non-aqueous electrolyte is adsorbed on the surface of the positive electrode active material, thereby suppressing the side reaction of the positive electrode and suppressing the generation of gas components. Therefore, even if the above-mentioned casing material is used, there is no It exhibits the above-mentioned disadvantages and only exhibits the above-mentioned advantages of the above-mentioned shell material, so it is preferable.
[微多孔膜隔板][Microporous Membrane Separator]
本发明中使用的微多孔膜隔板只要是具有在电子上将两极间绝缘的规定的机械强度,离子透过度大,并兼备对与正极相接一侧的氧化性的耐性和对负极侧的还原性的耐性的隔板即可,没有不特别限定。作为具有该特性的微多孔膜隔板的材质,使用树脂、无机物、玻璃纤维等。As long as the microporous membrane separator used in the present invention has a predetermined mechanical strength to electronically insulate between the two electrodes, has a high ion permeability, and has both resistance to oxidation on the side contacting the positive electrode and resistance to the negative electrode side. The separator is not particularly limited as long as it is a reduction-resistant separator. As the material of the microporous membrane separator having this characteristic, resin, inorganic substance, glass fiber, etc. are used.
作为上述树脂,没有特别限定,例如可以举出烯烃类聚合物、氟类聚合物、纤维素类聚合物、聚酰亚胺、尼龙等。具体地说,优选从对非水电解液稳定、且保液性优异的材料中选择,优选使用以聚乙烯、聚丙烯等聚烯烃类为原料的多孔片或非织造布等。作为上述无机物,没有特别限定,例如可使用氧化铝、二氧化硅等氧化物类;氮化铝或氮化硅等氮化物类;硫酸钡、硫酸钙等硫酸盐类等。对于形状,可使用粒子状或纤维状的。It does not specifically limit as said resin, For example, an olefin polymer, a fluoropolymer, a cellulosic polymer, polyimide, nylon etc. are mentioned. Specifically, it is preferable to select from materials that are stable to non-aqueous electrolytes and have excellent liquid retention properties, and it is preferable to use porous sheets or nonwoven fabrics made of polyolefins such as polyethylene and polypropylene. The inorganic substance is not particularly limited, and examples thereof include oxides such as alumina and silica; nitrides such as aluminum nitride and silicon nitride; sulfates such as barium sulfate and calcium sulfate, and the like. For the shape, granular or fibrous ones can be used.
作为形态,优选为非织造布、织布、微多孔性膜等薄膜形状。薄膜形状中,优选使用孔径为0.01~1μm、厚度为5~50μm的薄膜。除了上述独立的薄膜形状以外,可以使用用树脂制的粘合剂将含有上述无机物粒子的复合多孔层形成在正极和/或负极的表层上的形态。作为这样的形态,例如可以举出在正极的两面上,使用氟树脂作为粘合剂形成90%粒径为1μm以下的氧化铝粒子而得到的多孔层。The form is preferably in the form of a film such as a nonwoven fabric, a woven fabric, or a microporous film. In the film shape, it is preferable to use a film having a pore diameter of 0.01 to 1 μm and a thickness of 5 to 50 μm. In addition to the aforementioned independent film shape, a form in which the composite porous layer containing the aforementioned inorganic particles is formed on the surface layer of the positive electrode and/or the negative electrode using a resin binder may be used. Such an aspect includes, for example, a porous layer in which 90% of alumina particles having a particle size of 1 μm or less are formed using a fluororesin as a binder on both surfaces of the positive electrode.
本发明中使用的微多孔膜隔板优选具有通过加热堵塞孔的性质的隔板。如果组合使用上述微多孔膜隔板、和作为外壳材料的在电池的内面侧的至少一部分使用具有比该微多孔膜隔板的孔堵塞开始温度高的熔点的热塑性树脂来形成的外壳材料,则在过充电等异常发热时,具有可以在电池外壳破裂或电解液泄漏之前停止发热的效果。此外,这里所说的“通过加热堵塞孔的性质”,是指形成了电解液可以在正负极之间移动的多孔层含有热塑性树脂,并且加热至该热塑性树脂的熔点附近时,该多孔层堵塞,电解液不能在正负极之间移动的性质。The microporous membrane separator used in the present invention preferably has a property of clogging pores by heating. If the above-mentioned microporous membrane separator is used in combination with an outer casing material formed using a thermoplastic resin having a melting point higher than the pore clogging initiation temperature of the microporous membrane separator at least part of the inner surface side of the battery as the outer casing material, then In the event of abnormal heat generation such as overcharging, it has the effect of stopping the heat generation before the battery case breaks or the electrolyte leaks. In addition, the "property of clogging pores by heating" mentioned here means that the porous layer that forms the electrolytic solution that can move between the positive and negative electrodes contains thermoplastic resin, and when heated to near the melting point of the thermoplastic resin, the porous layer will Blockage, the property that the electrolyte cannot move between the positive and negative electrodes.
[电池形状][battery shape]
对电池形状没有特别限定,可以举出有底筒型形状、有底方形形状、薄型形状、片状、纸状等。组装入系统或机器中时,为了提高容积效率提高收纳性,还可以是考虑到配置在电池周边的周边系统的收纳的马蹄形、梳型形状等异形。从有效地将电池内部的热量释放到外部方面考虑,优选具有至少一个比较平滑的大面积的面的方形形状。The shape of the battery is not particularly limited, and examples thereof include a cylindrical shape with a bottom, a square shape with a bottom, a thin shape, a sheet shape, and a paper shape. When assembled into a system or machine, in order to improve volumetric efficiency and storage capacity, it can also be a horseshoe shape, a comb shape, or other irregular shapes that consider the storage of peripheral systems arranged around the battery. From the viewpoint of effectively releasing heat inside the battery to the outside, a square shape having at least one relatively smooth large-area surface is preferable.
在有底筒型形状的电池中,由于相对于填充的发电元件的外表面积减小,因此优选形成有效地将充电或放电时由于内部电阻产生的焦耳热释放到外部的设计。此外,优选设计成提高导热性高的物质的填充比率、且减小内部的温度分布。In a battery with a bottomed cylindrical shape, since the outer surface area relative to the filled power generating element is reduced, it is preferable to form a design that efficiently releases Joule heat generated by internal resistance during charging or discharging to the outside. In addition, it is preferable to design so as to increase the filling ratio of a substance with high thermal conductivity and to reduce the internal temperature distribution.
有底方形形状中,优选最大面的面积S1(除去端子部的外形尺寸的宽与高的乘积、单位cm2)的2倍与电池外形的厚度T(单位cm)的比率(2×S1/T)的值为100以上,更优选为200以上。通过增大最大面,即使为高输出功率且大容量的电池,也可以提高循环性或高温保存等特性,同时可以提高异常发热时的放热效率,从而可以抑制形成“破裂”或“起火”的危险状态。In a square shape with a bottom, it is preferable that the ratio of the area S 1 of the largest surface (the product of the width and the height of the external dimension excluding the terminal portion, in cm 2 ) to the thickness T (in cm) of the battery external shape (2×S 1 /T) is 100 or more, more preferably 200 or more. By enlarging the maximum surface, it is possible to improve the characteristics such as cycleability and high-temperature storage even for high-output and large-capacity batteries, and at the same time, it is possible to improve the heat release efficiency when abnormal heat generation occurs, thereby suppressing the formation of "crack" or "fire". dangerous state.
[电极组][Electrode group]
电极组可以是在后述的正极板和负极板之间夹着后述的微多孔膜隔板而形成的叠层结构电极组,以及在后述的正极板和负极板之间夹着后述的微多孔膜隔板卷绕成螺旋状的卷绕结构电极组的任意一种。电极组的体积在不包括电池外壳的突起部的体积中所占的比率(下面简称为“电极组占有率”)优选为0.3~0.7,更优选为0.4~0.6。上述电极组占有率小于0.3时,电池容量变小,另外,如果超过0.7,则由于空隙空间减小,不能充分确保后述的集电结构所必需的体积,电池电阻有可能增加,此外,由于电池形成高温,部件膨胀或电解质的液体成分的蒸气压升高,内部压力升高,存在作为电池的反复充放电性能或高温保存等各种特性降低或进一步将内部压力释放于外部的气体释放阀工作的情况。The electrode group can be a laminated structure electrode group formed by sandwiching a microporous membrane separator described later between a positive electrode plate and a negative electrode plate described later, and an electrode group of a laminated structure formed by sandwiching a The microporous membrane separator is wound into any one of the spirally wound structure electrode groups. The ratio of the volume of the electrode group to the volume excluding the protrusion of the battery case (hereinafter simply referred to as "electrode group occupancy") is preferably 0.3 to 0.7, more preferably 0.4 to 0.6. When the above-mentioned electrode group occupancy ratio is less than 0.3, the battery capacity becomes small. In addition, if it exceeds 0.7, since the void space decreases, the volume necessary for the current collection structure described later cannot be sufficiently ensured, and the battery resistance may increase. In addition, due to When the battery becomes high temperature, parts expand or the vapor pressure of the liquid component of the electrolyte increases, the internal pressure increases, and there is a gas release valve that reduces various characteristics such as repeated charge and discharge performance of the battery or high temperature storage, or further releases the internal pressure to the outside. work situation.
此外,电极组为叠层结构时,正极和负极优选电极周围长度的总和L(单位cm)与电极面积S2(单位cm2)的2倍之比(L/(2×S2))为1以下、更优选为0.5以下、进一步优选为0.3以下。此外,其下限优选为0.02以上、更优选为0.03以上、进一步优选为0.05以上。叠层结构的情况下,由于与电极的周围接近的部分在因残留应力或切断引起的冲击下,电极膜的粘合性有可能变差,因此当L/(2×S2)超过上述范围时,输出功率特性有时降低。此外,若L/(2×S2)低于上述范围,则电池面积过大,有时并不实用。In addition, when the electrode group has a laminated structure, the ratio (L/(2×S 2 )) of the sum of the length L (unit cm) around the electrodes of the positive electrode and the negative electrode to twice the electrode area S 2 (unit cm 2 ) is: 1 or less, more preferably 0.5 or less, still more preferably 0.3 or less. In addition, the lower limit thereof is preferably 0.02 or more, more preferably 0.03 or more, and still more preferably 0.05 or more. In the case of a laminated structure, the adhesiveness of the electrode film may deteriorate under the impact of residual stress or cutting at the part close to the periphery of the electrode. Therefore, when L/(2×S 2 ) exceeds the above range , the output power characteristics sometimes degrade. In addition, when L/(2×S 2 ) is lower than the above-mentioned range, the battery area may become too large, which may not be practical.
此外,电极组为卷绕结构时,优选该正极的长度方向的长度对于宽度方向的长度的比率为15~200。若该比率小于上述范围,则存在电池外壳的有底筒型形状相对于底面积来说高度过大,平衡变差而不实用,或者正极活性物质层变厚而不能得到高输出功率的情况。此外,如果该比率低于上述范围,则存在电池外壳的有底筒型形状相对于底面积来说高度过小,平衡变差而不实用,或者集电体所占的比率增大,电池容量减小的情况。In addition, when the electrode group has a wound structure, it is preferable that the ratio of the length in the longitudinal direction of the positive electrode to the length in the width direction is 15-200. If the ratio is less than the above range, the height of the bottomed cylindrical shape of the battery case may be too high relative to the bottom area, and the balance may deteriorate to be impractical, or the positive electrode active material layer may become thick and high output may not be obtained. In addition, if the ratio is lower than the above range, the height of the bottomed cylindrical shape of the battery case is too small relative to the bottom area, and the balance becomes poor to be practical, or the ratio of the current collector increases, and the battery capacity reduced situation.
[正极电极面积](结构[1])[Positive Electrode Area] (Structure[1])
在本发明中,使用上述非水电解液时,从高输出功率且提高高温时的稳定性方面考虑,优选正极活性物质层的面积比电池外壳的外表面积大。具体地说,从输出功率的提高和通过集电体可以有效地将充放电带来的发热释放的观点考虑,优选上述正极的电极面积的总和相对于二次电池的外壳的表面积以面积比计为15倍以上,更优选为20倍以上(结构[1])。所谓外壳的外表面积,有底方形形状的情况下,是指由除了端子的突起部分的填充有发电要素的壳部分的由长、宽和厚度的尺寸计算求得的总面积。有底圆筒形状的情况下,是指除了端子的突起部分的填充有发电要素的壳部分近似为圆筒的几何表面积。所谓正极的电极面积的总和,是指与含有负极活性物质的复层材料层对置的正极复层材料层的几何表面积,在通过集电体箔在两面上形成正极复层材料层的结构中,是指分别计算出各个面的面积的总和。In the present invention, when the above-mentioned non-aqueous electrolytic solution is used, it is preferable that the area of the positive electrode active material layer is larger than the outer surface area of the battery case from the viewpoint of high output and improved stability at high temperature. Specifically, from the standpoint of improving the output power and effectively releasing the heat generated by charging and discharging through the current collector, it is preferable that the sum of the electrode areas of the above-mentioned positive electrode is calculated as an area ratio with respect to the surface area of the casing of the secondary battery. It is 15 times or more, more preferably 20 times or more (Structure [1]). The outer surface area of the casing refers to the total area calculated from the dimensions of length, width and thickness of the casing portion filled with the power generating element except the protruding portion of the terminal in the case of a square shape with a bottom. In the case of a bottomed cylindrical shape, it refers to a geometrical surface area in which the part of the case filled with the power generating elements except the protruding part of the terminal is approximately a cylinder. The sum of the electrode areas of the so-called positive electrode refers to the geometric surface area of the positive electrode composite layer material layer opposite to the composite layer material layer containing the negative electrode active material. In the structure where the positive electrode composite layer material layer is formed on both sides by the current collector foil , is to calculate the sum of the areas of each surface separately.
[保护元件][protection element]
作为上述保护元件,可以举出异常发热或过大电流流过时电阻增大的PTC(positive temperature coefficient,正温度系数)、温度熔断器、热敏电阻、异常发热时由于电池内部压力或内部温度的急剧升高而阻断电路中流过的电流的阀(电流阻断阀)等。上述保护元件优选选择在高电流的通常使用中不工作条件的元件,从高输出功率的观点考虑,更优选形成即使没有保护元件也不至于异常发热或热逃逸的设计。Examples of the above-mentioned protection elements include PTC (positive temperature coefficient, positive temperature coefficient) whose resistance increases when abnormal heat generation or excessive current flows, temperature fuses, thermistors, battery internal pressure or internal temperature when abnormal heat generation occurs. A valve (current blocking valve), etc., which rises sharply to block the current flowing in the circuit. The above-mentioned protection element is preferably an element that does not work in normal use with high current. From the viewpoint of high output power, it is more preferable to form a design that does not generate abnormal heat or thermal runaway even if there is no protection element.
[组合电池][Assembled battery]
将本发明的锂二次电池作为电源供于实际应用时,对于电源要求的电压大多为单电池电压以上,必须对应使用将单电池串联连接的升压装置等。因此,本发明的锂二次电池优选制成将其多个串联连接而成的组电池使用。通过形成组电池,降低连接部分的电阻,从而可以抑制作为组电池的输出功率降低。作为组电池,从电源电压方面考虑,优选串联连接5个以上。When the lithium secondary battery of the present invention is used as a power source in practical applications, the voltage required for the power source is often higher than the voltage of the single cell, and a booster device or the like that connects the cells in series must be used accordingly. Therefore, the lithium secondary battery of the present invention is preferably used as a battery pack in which a plurality of them are connected in series. By forming a battery pack, the resistance of the connecting portion is reduced, thereby suppressing a decrease in output power as a battery pack. As the battery pack, five or more batteries are preferably connected in series in terms of power supply voltage.
此外,连接多个制成组电池时,由于充放电带来的发热的影响增大,因此优选具有用于将电池保持在一定温度以下的冷却机构。作为电池温度,优选为10℃~40℃,优选水冷却或者通过空气冷却使用外气来进行冷却。In addition, when connecting a plurality of assembled batteries, since the influence of heat generated by charging and discharging increases, it is preferable to have a cooling mechanism for keeping the battery below a certain temperature. The temperature of the battery is preferably 10° C. to 40° C., and it is preferably cooled by water cooling or air cooling using outside air.
[用途][use]
本发明的锂二次电池以及将多个本发明的锂二次电池连接而成的组电池,由于具有高输出功率、长寿命、高安全性等,因此优选用于装载在车辆上,其功率至少供给车辆的驱动系统的用途中。The lithium secondary battery of the present invention and a battery pack formed by connecting a plurality of lithium secondary batteries of the present invention are preferably used for loading on vehicles due to their high output power, long life, high safety, etc., and their power At least for use in a drive system of a vehicle.
另外,认为上述特定化合物通过吸附在正极活性物质表面或金属材料表面,可以抑制与电解液等的副反应。认为通过该抑制在正极活性物质表面的副反应,气体产生少,即使使用片状的外壳材料也不易引起电池的变形或内压的升高,所以优选。此外认为,通过抑制正极活性物质表面的副反应,电池的寿命、输出功率、过充电时的安全性提高,这有利于在大型电池中的应用。此外还认为,通过抑制金属材料表面的副反应,在集电片和端子的连接中,即使使用电阻小、简易的焊接方法时,由于长期使用时直流电阻成分的增加得到抑制,因此有利于在大型电池中的应用。此外认为,降低正极的反应电阻对于在将电池形状或正极面积等设计成高输出功率的电池中进一步提高输出功率是有效的,所以优选。In addition, it is considered that the above-mentioned specific compound can suppress a side reaction with an electrolytic solution or the like by being adsorbed on the surface of the positive electrode active material or the surface of the metal material. It is considered that by suppressing side reactions on the surface of the positive electrode active material, less gas is generated, and even if a sheet-shaped casing material is used, deformation of the battery or increase in internal pressure is less likely to occur, which is preferable. In addition, it is considered that by suppressing the side reaction on the surface of the positive electrode active material, the battery life, output power, and safety during overcharge are improved, which is advantageous for application to large batteries. In addition, it is also believed that by suppressing the side reaction on the surface of the metal material, even when a simple welding method with low resistance is used in the connection of the current collector and the terminal, the increase in the DC resistance component during long-term use is suppressed, so it is beneficial to the connection between the current collector and the terminal. applications in large batteries. In addition, lowering the reaction resistance of the positive electrode is considered to be effective for further increasing the output in a battery designed for high output such as the shape of the battery or the area of the positive electrode, and is therefore preferable.
实施例Example
下面,举出实施例和比较例更具体地说明本发明,但只要不超出本发明的主旨,则本发明不受这些实施例的限定。Hereinafter, although an Example and a comparative example are given and this invention is demonstrated more concretely, this invention is not limited to these Examples unless the summary of this invention is exceeded.
正极[1][正极活性物质]Positive electrode[1][Positive electrode active material]
下面的实施例和比较例中使用的正极活性物质的种类和物性如下。The types and physical properties of positive electrode active materials used in the following Examples and Comparative Examples are as follows.
正极[1]表1Positive electrode [1] Table 1
[表1][Table 1]
正极[1]表1中,作为正极活性物质的物性,按照上述记载的方法,进行BET比表面积、平均一次粒径(用SEM测定)、中值粒径d50、振实密度的测定。Positive electrode [1] In Table 1, as the physical properties of the positive electrode active material, BET specific surface area, average primary particle size (measured by SEM), median particle size d 50 , and tap density were measured according to the method described above.
[正极活性物质A][Positive electrode active material A]
正极活性物质A是通过下面所示的方法合成的锂过渡金属复合氧化物,用组成式LiMn0.33Ni0.33Co0.33O2表示。以Mn∶Ni∶Co=1∶1∶1的摩尔比称量作为锰原料的Mn3O4、作为镍原料的NiO和作为钴原料的Co(OH)2,并向其中加入纯水制成浆料,边搅拌边使用循环式介质搅拌型湿式珠磨机将浆料中的固体成分湿式粉碎成中值粒径0.2μm。The positive electrode active material A is a lithium transition metal composite oxide synthesized by the method shown below, and is represented by the composition formula LiMn 0.33 Ni 0.33 Co 0.33 O 2 . Mn 3 O 4 as a manganese raw material, NiO as a nickel raw material, and Co(OH) 2 as a cobalt raw material are weighed at a molar ratio of Mn:Ni:Co=1:1:1, and pure water is added thereto to prepare For the slurry, wet pulverize the solid components in the slurry to a median particle size of 0.2 μm using a circulating medium agitation type wet bead mill while stirring.
通过喷雾干燥器将浆料进行喷雾干燥,得到仅仅包含锰原料、镍原料、钴原料的粒径约5μm的大致球状的造粒粒子。在得到的造粒粒子中添加中值粒径3μm的LiOH粉末,并使Li的摩尔数与Mn、Ni和Co的总摩尔数之比为1.05,用高速混合机进行混合,得到镍原料、钴原料、锰原料的造粒粒子和锂原料的混合粉末。在空气流通下、950℃对该混合粉末烧结12小时(升降温速度为5℃/分钟)后,粉碎,通过网眼45μm的筛子,得到正极活性物质A。The slurry was spray-dried by a spray dryer to obtain roughly spherical granulated particles having a particle diameter of about 5 μm and containing only manganese raw materials, nickel raw materials, and cobalt raw materials. LiOH powder with a median diameter of 3 μm was added to the obtained granulated particles, and the ratio of the number of moles of Li to the total number of moles of Mn, Ni, and Co was 1.05, and mixed with a high-speed mixer to obtain nickel raw materials, cobalt Raw material, granulated particles of manganese raw material and mixed powder of lithium raw material. The mixed powder was sintered at 950° C. for 12 hours under air circulation (the heating and cooling rate was 5° C./min), and then pulverized and passed through a sieve with a mesh size of 45 μm to obtain a positive electrode active material A.
[正极活性物质B][Positive electrode active material B]
正极活性物质B是与正极活性物质A同样地合成的锂过渡金属复合氧化物,用组成式LiMn0.33Ni0.33Co0.33O2表示,其不同之处为:变更喷雾干燥的条件,制成粒径约1μm的造粒粒子,并且烧结温度为930℃。The positive electrode active material B is a lithium transition metal composite oxide synthesized in the same way as the positive electrode active material A, and is represented by the composition formula LiMn 0.33 Ni 0.33 Co 0.33 O 2 , the difference is that the spray drying conditions are changed to make the particle size The granulated particles are about 1 μm, and the sintering temperature is 930°C.
[正极活性物质C][Positive electrode active material C]
正极活性物质C是按照下面所示的方法合成的在正极活性物质A的表面附着了硫化合物和锑化合物而形成的正极活性物质。即,将96.7重量份正极活性物质A在流动槽中搅拌,同时向其中喷雾1.3重量份硫酸锂(Li2SO4H2O)的水溶液成喷雾状。在得到的混合物中添加2.0重量份三氧化锑(Sb2O3,粒子中值粒径为0.8μm),充分混合。将该混合物转移到氧化铝容器中,在空气氛围下于680℃烧结2小时,得到正极活性物质C。The positive electrode active material C is a positive electrode active material in which a sulfur compound and an antimony compound are attached to the surface of the positive electrode active material A, synthesized by the method shown below. That is, while stirring 96.7 parts by weight of the positive electrode active material A in the flow cell, 1.3 parts by weight of an aqueous solution of lithium sulfate (Li 2 SO 4 H 2 O) was sprayed thereinto in a spray form. 2.0 parts by weight of antimony trioxide (Sb 2 O 3 , particle median diameter: 0.8 μm) was added to the obtained mixture, and mixed well. The mixture was transferred to an alumina container, and sintered at 680° C. for 2 hours in an air atmosphere to obtain a positive electrode active material C.
[正极活性物质D][Positive electrode active material D]
正极活性物质D是通过下面所示的方法合成的锂过渡金属复合氧化物,用组成式Li1.04Mn1.84Al0.12O4表示。以Li∶Mn∶Al=1.04∶1.84∶0.12的摩尔比称量作为锂原料的LiOH、作为锰原料的Mn2O3和作为铝原料的AlOOH,并向其中加入纯水制成浆料,边搅拌边使用循环式介质搅拌型湿式珠磨机将浆料中的固体成分湿式粉碎成中值粒径0.5μm。The positive electrode active material D is a lithium transition metal composite oxide synthesized by the method shown below, and is represented by the composition formula Li 1.04 Mn 1.84 Al 0.12 O 4 . LiOH as a lithium raw material, Mn2O3 as a manganese raw material, and AlOOH as an aluminum raw material were weighed at a molar ratio of Li:Mn:Al=1.04:1.84:0.12 , and pure water was added thereto to make a slurry, while While stirring, the solid content in the slurry was wet-milled to a median particle size of 0.5 μm using a circulating medium-stirred wet bead mill.
通过喷雾干燥器将浆料进行喷雾干燥,得到仅仅包含锂原料、锰原料、铝原料的粒径约10μm的大致球状的造粒粒子。将该造粒粒子在氮气流通、900℃下烧结3小时(升温速度为5℃/分钟)后,将流通气体由氮气变换成空气,再在900℃下烧结2小时(降温速度为1℃/分钟)。冷却到室温后,取出并粉碎,通过网眼45μm的筛子,得到正极活性物质D。The slurry was spray-dried by a spray dryer to obtain approximately spherical granulated particles having a particle diameter of about 10 μm and containing only the lithium raw material, the manganese raw material, and the aluminum raw material. The granulated particles were sintered at 900° C. for 3 hours in nitrogen flow (heating rate was 5° C./min), then the flow gas was changed from nitrogen to air, and then sintered at 900° C. for 2 hours (temperature drop rate was 1° C./min. minute). After cooling to room temperature, it was taken out and pulverized, and passed through a sieve with a mesh size of 45 μm to obtain a positive electrode active material D.
[正极活性物质E][Positive electrode active material E]
正极活性物质E是市售品(日本化学工业公司制造的钴酸锂),是用组成式Li1.03CoO2表示的锂钴氧化物。The positive electrode active material E is a commercial product (lithium cobalt oxide manufactured by Nippon Chemical Industry Co., Ltd.), and is lithium cobalt oxide represented by the composition formula Li 1.03 CoO 2 .
正极[1]实施例1Positive electrode [1] embodiment 1
《正极的制作》"Positive Production"
在N甲基吡咯烷酮溶剂中混合90质量%的作为正极活性物质的正极活性物质A、5质量%的作为导电材料的乙炔黑和5质量%作为粘合剂的聚偏氟乙烯(PVdF),制成浆料。将得到的浆料涂布在厚度15μm的铝箔两面,并干燥,用压制机压延成厚度为80μm,再将其切成具有宽100mm、长100mm大小的正极活性物质层和宽30mm的未涂布部分的形状,作为正极。正极活性物质的密度为2.35g/cm3,(一面的正极活性物质层的厚度)/(集电体的厚度)的值为2.2。Mix 90% by mass of positive electrode active material A as a positive electrode active material, 5% by mass of acetylene black as a conductive material, and 5% by mass of polyvinylidene fluoride (PVdF) as a binder in N-methylpyrrolidone solvent to prepare into slurry. The obtained slurry was coated on both sides of an aluminum foil with a thickness of 15 μm, dried, rolled into a thickness of 80 μm with a press, and then cut into a positive electrode active material layer with a width of 100 mm and a length of 100 mm and an uncoated layer with a width of 30 mm. The shape of the part, as the positive electrode. The density of the positive electrode active material was 2.35 g/cm 3 , and the value of (thickness of positive electrode active material layer on one side)/(thickness of current collector) was 2.2.
《负极的制作》"The Making of Negative Pole"
在98重量份人造石墨粉末KS-44(timcal(テイムカル)公司制造,商品名)中添加100重量份作为增稠剂的羧甲基纤维素钠的水性分散液(羧甲基纤维素钠的浓度为1质量%),2重量份作为粘合剂的苯乙烯-丁二烯橡胶的水性分散液(苯乙烯-丁二烯橡胶的浓度为50质量%),用分散器进行混合,制成浆料。将得到的浆料涂布在厚度10μm的铜箔两面,并干燥,用压制机压延成厚度为75μm,再将其切成具有宽104mm、长104mm大小的负极活性物质层和宽30mm的未涂布部分的形状,作为负极。In 98 parts by weight of artificial graphite powder KS-44 (manufactured by timcal (Timcal) company, trade name), add 100 parts by weight of aqueous dispersion of sodium carboxymethyl cellulose as a thickener (concentration of sodium carboxymethyl cellulose 1% by mass), 2 parts by weight of an aqueous dispersion of styrene-butadiene rubber as a binder (the concentration of styrene-butadiene rubber is 50% by mass), and mixed with a disperser to make a slurry material. The obtained slurry is coated on both sides of a copper foil with a thickness of 10 μm, dried, rolled into a thickness of 75 μm with a press machine, and then cut into a negative electrode active material layer with a width of 104 mm and a length of 104 mm and an uncoated layer with a width of 30 mm. The shape of the cloth part is used as the negative electrode.
《电解液的制作》"Making Electrolyte"
在干燥的氩氛围下,在纯化的碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的体积比3∶3∶4的混合溶剂中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。另外,含有0.3质量%的六甲基环三硅氧烷。Under a dry argon atmosphere, in a mixed solvent of purified ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) with a volume ratio of 3:3:4, at 1mol/L The concentration dissolves well-dried lithium hexafluorophosphate (LiPF 6 ). In addition, 0.3% by mass of hexamethylcyclotrisiloxane was contained.
《电池的制作》"Battery Making"
将32片正极和33片负极交替配置,各电极之间夹入多孔聚乙烯片隔板(厚度为25μm),进行叠层。此时,使正极活性物质面与负极活性物质面相对,并且不会露出负极活性物质面之外。将该正极和负极各自的未涂布部分焊接,制作集电片,将电极组密封在电池罐(外部尺寸:120×110×10mm)中。然后,在装有电极组的电池罐中注入20mL电解液,使电极充分浸透,密封而制作电池。正极的电极面积总和与电池的外壳表面积总和之比为20.6。32 positive electrodes and 33 negative electrodes were arranged alternately, and a porous polyethylene sheet separator (thickness: 25 μm) was sandwiched between the electrodes for lamination. At this time, the surface of the positive electrode active material faces the surface of the negative electrode active material without protruding from the surface of the negative electrode active material. The respective uncoated parts of the positive and negative electrodes were welded to produce current collector sheets, and the electrode group was sealed in a battery can (outer dimensions: 120×110×10 mm). Then, 20 mL of electrolytic solution was poured into the battery can containing the electrode group, the electrodes were sufficiently permeated, and the battery was sealed to produce a battery. The ratio of the sum of the electrode areas of the positive electrode to the sum of the surface areas of the battery case was 20.6.
《电池的评价》"Battery Evaluation"
(电池容量的测定方法)(Measuring method of battery capacity)
对未经充放电循环的新电池,在25℃、4.1V~3.0V的电压范围下,以0.2C的电流值(将1小时放电额定容量的电流值作为1C,所述额定容量取决于1小时率(one-hour-rate)的放电容量,下同)进行5个循环的初期充放电。此时的第5个循环的0.2C放电容量作为初期容量。电池评价的结果示于正极[1]表2。For a new battery that has not been charged and discharged, at 25°C and a voltage range of 4.1V to 3.0V, use a current value of 0.2C (the current value of the rated capacity of 1 hour discharge is taken as 1C, and the rated capacity depends on 1 One-hour-rate (one-hour-rate) discharge capacity, the same below) was performed for 5 cycles of initial charge and discharge. The 0.2C discharge capacity at the fifth cycle at this time was taken as the initial capacity. The results of the battery evaluation are shown in Table 2 of the positive electrode [1].
(初期输出功率的测定方法)(Measurement method of initial output power)
在25℃的环境下,通过0.2C的恒定电流进行150分钟充电,分别以0.1C、0.3C、1.0C、3.0C、10.0C放电10秒钟,测定第10秒的电压。将电流-电压直线和下限电压(3V)所包围的三角形的面积作为输出功率(W)。电池评价的结果示于正极[1]表2。In an environment of 25°C, charge was performed for 150 minutes with a constant current of 0.2C, and discharge was performed at 0.1C, 0.3C, 1.0C, 3.0C, and 10.0C for 10 seconds, respectively, and the voltage at the 10th second was measured. The area of the triangle surrounded by the current-voltage line and the lower limit voltage (3V) was taken as the output power (W). The results of the battery evaluation are shown in Table 2 of the positive electrode [1].
(循环试验(耐久后电池容量和耐久后输出功率的测定方法))(Cycle test (measuring method of battery capacity after endurance and output power after endurance))
在被视为锂二次电池的实际使用上限温度的60℃的高温环境下,进行循环试验。以2C恒定电流恒定电压法充电到充电上限电压4.1V后,以2C的恒定电流放电至放电终止电压3.0V,以此作为一个充放电循环,反复进行该循环,直至500循环。对于循环试验结束后的电池,在25℃的环境下以0.2C的电流值进行3个循环的充放电,以第3个循环的0.2C放电容量作为耐久后电池容量。另外,对于循环试验结束后的电池,进行输出功率测定,作为耐久后输出功率。电池评价的结果示于正极[1]表2。The cycle test was performed in a high-temperature environment of 60° C., which is regarded as the upper limit temperature for practical use of lithium secondary batteries. After charging to the charging upper limit voltage of 4.1V with 2C constant current and constant voltage method, discharge at a constant current of 2C to the end-of-discharge voltage of 3.0V, which is regarded as a charge-discharge cycle, and this cycle is repeated until 500 cycles. For the battery after the cycle test, three cycles of charging and discharging were performed at a current value of 0.2C in an environment of 25°C, and the 0.2C discharge capacity of the third cycle was used as the battery capacity after endurance. In addition, for the battery after the cycle test, the output power was measured, and it was set as the output power after durability. The results of the battery evaluation are shown in Table 2 of the positive electrode [1].
正极[1]实施例2Positive electrode [1] Example 2
使电解液含有0.3质量%的甲磺酸三甲基甲硅烷基酯来代替六甲基环三硅氧烷,除此以外,与正极[1]实施例1同样实施。电池评价的结果示于正极[1]表2。It carried out similarly to positive electrode [1] Example 1 except having made electrolytic solution contain 0.3 mass % of trimethylsilyl methanesulfonate instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 2 of the positive electrode [1].
正极[1]实施例3Positive electrode [1] Example 3
使电解液含有0.3质量%的苯基二甲基氟硅烷来代替六甲基环三硅氧烷,除此以外,与正极[1]实施例1同样实施。电池评价的结果示于正极[1]表2。It carried out similarly to positive electrode [1] Example 1 except having made electrolytic solution contain 0.3 mass % of phenyldimethylfluorosilanes instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 2 of the positive electrode [1].
正极[1]实施例4Positive electrode [1] Example 4
使电解液含有0.3质量%的二氟磷酸锂来代替六甲基环三硅氧烷,除此以外,与正极[1]实施例1同样实施。电池评价的结果示于正极[1]表2。The procedure was carried out in the same manner as in positive electrode [1] Example 1, except that the electrolytic solution contained 0.3% by mass of lithium difluorophosphate instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 2 of the positive electrode [1].
正极[1]比较例1Positive electrode [1] Comparative Example 1
除了电解液中不含六甲基环三硅氧烷以外,与正极[1]实施例1同样实施。电池评价的结果示于正极[1]表2。Except that hexamethylcyclotrisiloxane is not contained in the electrolytic solution, it is carried out in the same manner as the positive electrode [1] Example 1. The results of the battery evaluation are shown in Table 2 of the positive electrode [1].
正极[1]表2Positive electrode [1] Table 2
[表2][Table 2]
正极[1]实施例5Positive electrode [1] Example 5
除了使用正极活性物质A和正极活性物质B以2∶1的质量比充分混合而得到的正极活性物质作为正极活性物质,制成正极以外,与正极[1]实施例1同样实施。该混合的正极活性物质的BET比表面积为1.8m2/g,平均一次粒径为0.22μm,中值粒径d50为3.2μm,振实密度为1.5g/cm2。电池评价的结果示于正极[1]表3。Except using the positive electrode active material obtained by mixing the positive electrode active material A and the positive electrode active material B at a mass ratio of 2:1 as the positive electrode active material, the positive electrode [1] Example 1 was carried out in the same manner. The BET specific surface area of the mixed positive electrode active material was 1.8 m 2 /g, the average primary particle size was 0.22 μm, the median particle size d 50 was 3.2 μm, and the tap density was 1.5 g/cm 2 . The results of the battery evaluation are shown in Table 3 of the positive electrode [1].
正极[1]实施例6Positive electrode [1] Example 6
使电解液含有0.3质量%的甲磺酸三甲基甲硅烷基酯来代替六甲基环三硅氧烷,除此以外,与正极[1]实施例5同样实施。电池评价的结果示于正极[1]表3。It carried out similarly to positive electrode [1] Example 5 except having made electrolytic solution contain 0.3 mass % of trimethylsilyl methanesulfonate instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 3 of the positive electrode [1].
正极[1]实施例7Positive electrode [1] Example 7
使电解液含有0.3质量%的苯基二甲基氟硅烷来代替六甲基环三硅氧烷,除此以外,与正极[1]实施例5同样实施。电池评价的结果示于正极[1]表3。It carried out similarly to positive electrode [1] Example 5 except having made electrolytic solution contain 0.3 mass % of phenyldimethylfluorosilanes instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 3 of the positive electrode [1].
正极[1]实施例8Positive electrode [1] Example 8
使电解液含有0.3质量%的二氟磷酸锂来代替六甲基环三硅氧烷,除此以外,与正极[1]实施例5同样实施。电池评价的结果示于正极[1]表3。The same procedure was carried out as in Example 5 of the positive electrode [1] except that the electrolytic solution contained 0.3% by mass of lithium difluorophosphate instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 3 of the positive electrode [1].
正极[1]比较例2Positive electrode [1] Comparative example 2
除了电解液中不含六甲基环三硅氧烷以外,与正极[1]实施例5同样实施。电池评价的结果示于正极[1]表3。Except that the electrolytic solution does not contain hexamethylcyclotrisiloxane, the same implementation is carried out as in the positive electrode [1] Example 5. The results of the battery evaluation are shown in Table 3 of the positive electrode [1].
正极[1]表3Positive pole [1] Table 3
[表3][table 3]
正极[1]实施例9Positive electrode [1] Example 9
除了使用正极活性物质C作为正极活性物质,制成正极以外,与正极[1]实施例1同样实施。电池评价的结果示于正极[1]表4。Except using the positive electrode active material C as the positive electrode active material to prepare a positive electrode, it was carried out in the same manner as the positive electrode [1] Example 1. The results of the battery evaluation are shown in Table 4 of the positive electrode [1].
正极[1]实施例10Positive electrode [1] Example 10
使电解液含有0.3质量%的甲磺酸三甲基甲硅烷基酯来代替六甲基环三硅氧烷,除此以外,与正极[1]实施例9同样实施。电池评价的结果示于正极[1]表4。It carried out similarly to positive electrode [1] Example 9 except having made electrolytic solution contain 0.3 mass % of trimethylsilyl methanesulfonate instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 4 of the positive electrode [1].
正极[1]实施例11Positive electrode [1] Example 11
使电解液含有0.3质量%的苯基二甲基氟硅烷来代替六甲基环三硅氧烷,除此以外,与正极[1]实施例9同样实施。电池评价的结果示于正极[1]表4。The same procedure was carried out as in positive electrode [1] Example 9 except that the electrolytic solution contained 0.3% by mass of phenyldimethylfluorosilane instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 4 of the positive electrode [1].
正极[1]实施例12Positive electrode [1] Example 12
使电解液含有0.3质量%的二氟磷酸锂来代替六甲基环三硅氧烷,除此以外,与正极[1]实施例9同样实施。电池评价的结果示于正极[1]表4。The procedure was carried out in the same manner as in positive electrode [1] Example 9 except that the electrolytic solution contained 0.3% by mass of lithium difluorophosphate instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 4 of the positive electrode [1].
正极[1]比较例3Positive electrode [1] Comparative example 3
除了电解液中不含六甲基环三硅氧烷以外,与正极[1]实施例9同样实施。电池评价的结果示于正极[1]表4。Except that hexamethylcyclotrisiloxane is not contained in the electrolytic solution, the same implementation is carried out as in Example 9 of the positive electrode [1]. The results of the battery evaluation are shown in Table 4 of the positive electrode [1].
正极[1]表4Positive pole [1] Table 4
[表4][Table 4]
正极[1]实施例13Positive electrode [1] Example 13
使用正极活性物质D作为正极活性物质,制成正极,用压制机压延成厚度为108μm,并使用28片该正极和29片负极,除此之外,与正极[1]实施例1同样地制作电池。(正极一面的正极活性物质层厚度/正极集电体的厚度)为3.1,正极的电极面积的总和与电池的外壳表面积总和之比为18.1。容量测定的电压范围为3.0~4.2V,循环试验的上限电压为4.2V,除此以外,与正极[1]实施例1同样地评价电池。电池评价的结果示于正极[1]表5。Use the positive electrode active material D as the positive electrode active material to make the positive electrode, roll it into a thickness of 108 μm with a press machine, and use 28 pieces of the positive electrode and 29 negative electrodes, except that it is made in the same way as the positive electrode [1] Example 1 Battery. (The thickness of the positive electrode active material layer on the positive electrode side/the thickness of the positive electrode current collector) was 3.1, and the ratio of the total electrode area of the positive electrode to the total surface area of the battery case was 18.1. The battery was evaluated in the same manner as in Example 1 of the positive electrode [1], except that the voltage range of the capacity measurement was 3.0 to 4.2 V, and the upper limit voltage of the cycle test was 4.2 V. The results of the battery evaluation are shown in Table 5 of the positive electrode [1].
正极[1]实施例14Positive electrode [1] Example 14
使电解液含有0.3质量%的甲磺酸三甲基甲硅烷基酯来代替六甲基环三硅氧烷,除此以外,与正极[1]实施例13同样实施。电池评价的结果示于正极[1]表5。The procedure was carried out in the same manner as in positive electrode [1] Example 13 except that the electrolytic solution contained 0.3% by mass of trimethylsilyl methanesulfonate instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 5 of the positive electrode [1].
正极[1]实施例15Positive electrode [1] Example 15
使电解液含有0.3质量%的苯基二甲基氟硅烷来代替六甲基环三硅氧烷,除此以外,与正极[1]实施例13同样实施。电池评价的结果示于正极[1]表5。The procedure was carried out in the same manner as in positive electrode [1] Example 13 except that the electrolytic solution contained 0.3% by mass of phenyldimethylfluorosilane instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 5 of the positive electrode [1].
正极[1]实施例16Positive electrode [1] Example 16
使电解液含有0.3质量%的二氟磷酸锂来代替六甲基环三硅氧烷,除此以外,与正极[1]实施例13同样实施。电池评价的结果示于正极[1]表5。The procedure was carried out in the same manner as in positive electrode [1] Example 13 except that the electrolytic solution contained 0.3% by mass of lithium difluorophosphate instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 5 of the positive electrode [1].
正极[1]比较例4Positive electrode [1] Comparative example 4
除了电解液中不含六甲基环三硅氧烷以外,与正极[1]实施例13同样实施。电池评价的结果示于正极[1]表5。Except that hexamethylcyclotrisiloxane is not contained in the electrolytic solution, it is carried out in the same manner as the positive electrode [1] Example 13. The results of the battery evaluation are shown in Table 5 of the positive electrode [1].
正极[1]表5Positive pole [1] Table 5
[表5][table 5]
正极[1]实施例17Positive electrode [1] Example 17
使用正极活性物质D和正极活性物质E以2∶1的质量比充分混合而得到的正极活性物质作为正极活性物质,制成正极,用压制机压延成厚度为94μm,并使用29片该正极和30片负极,除此之外,与正极[1]实施例1同样实施。(正极一面的正极活性物质层厚度/正极集电体的厚度)为2.6,正极的电极面积的总和与电池的外壳表面积总和之比为18.7。另外,该混合的正极活性物质的BET比表面积为0.8m2/g,平均一次粒径为0.50μm,中值粒径d50为8.3μm,振实密度为2.5g/cm3。电池评价的结果示于正极[1]表6。The positive electrode active material obtained by fully mixing the positive electrode active material D and the positive electrode active material E with a mass ratio of 2:1 is used as the positive electrode active material to make the positive electrode, and it is rolled into a thickness of 94 μm with a pressing machine, and 29 pieces of the positive electrode and the positive electrode are used. Except for 30 negative electrodes, it was carried out in the same manner as the positive electrode [1] Example 1. (The thickness of the positive electrode active material layer on the positive electrode side/the thickness of the positive electrode current collector) was 2.6, and the ratio of the total electrode area of the positive electrode to the total surface area of the battery case was 18.7. In addition, the mixed positive electrode active material had a BET specific surface area of 0.8 m 2 /g, an average primary particle size of 0.50 μm, a median particle size d 50 of 8.3 μm, and a tap density of 2.5 g/cm 3 . The results of the battery evaluation are shown in Table 6 of the positive electrode [1].
正极[1]实施例18Positive electrode [1] Example 18
使电解液含有0.3质量%的甲磺酸三甲基甲硅烷基酯来代替六甲基环三硅氧烷,除此以外,与正极[1]实施例17同样实施。电池评价的结果示于正极[1]表6。The same procedure as in Example 17 of the positive electrode [1] was carried out except that the electrolytic solution contained 0.3% by mass of trimethylsilyl methanesulfonate instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 6 of the positive electrode [1].
正极[1]实施例19Positive electrode [1] Example 19
使电解液含有0.3质量%的苯基二甲基氟硅烷来代替六甲基环三硅氧烷,除此以外,与正极[1]实施例17同样实施。电池评价的结果示于正极[1]表6。The procedure was carried out in the same manner as in positive electrode [1] Example 17, except that the electrolytic solution contained 0.3% by mass of phenyldimethylfluorosilane instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 6 of the positive electrode [1].
正极[1]实施例20Positive electrode [1] Example 20
使电解液含有0.3质量%的二氟磷酸锂来代替六甲基环三硅氧烷,除此以外,与正极[1]实施例17同样实施。电池评价的结果示于正极[1]表6。The procedure was carried out in the same manner as in positive electrode [1] Example 17, except that the electrolytic solution contained 0.3% by mass of lithium difluorophosphate instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 6 of the positive electrode [1].
正极[1]比较例5Positive electrode [1] Comparative example 5
除了电解液中不含六甲基环三硅氧烷以外,与正极[1]实施例17同样实施。电池评价的结果示于正极[1]表6。Except that hexamethylcyclotrisiloxane is not contained in the electrolytic solution, the same implementation is carried out as in Example 17 of the positive electrode [1]. The results of the battery evaluation are shown in Table 6 of the positive electrode [1].
正极[1]表6Positive pole [1] Table 6
[表6][Table 6]
由正极[1]表2~正极[1]表6的结果可知,在任一个正极中,由于在电解液中都含有特定化合物,因此,输出功率、容量保持率得到提高,并且即使在循环试验后,也可以充分保持电池容量和输出功率。From the results in Table 2 of the positive electrode [1] to Table 6 of the positive electrode [1], it can be seen that in any positive electrode, since the electrolyte contains a specific compound, the output power and capacity retention rate are improved, and even after the cycle test , can also fully maintain the battery capacity and output power.
正极[2][正极活性物质]Positive electrode [2] [positive active material]
下面的实施例和比较例中使用的正极活性物质的种类和物性如下。The types and physical properties of positive electrode active materials used in the following Examples and Comparative Examples are as follows.
正极[2]表1Positive pole [2] Table 1
[表7][Table 7]
正极[2]表1中,作为正极活性物质的物性,按照上述记载的方法,进行BET比表面积、平均一次粒径(用SEM测定)、中值粒径d50、振实密度的测定。Positive electrode [2] In Table 1, as the physical properties of the positive electrode active material, BET specific surface area, average primary particle size (measured by SEM), median particle size d 50 , and tap density were measured according to the method described above.
[正极活性物质A][Positive electrode active material A]
正极活性物质A是通过下面所示的方法合成的锂过渡金属复合氧化物,用组成式Li1.05Ni0.80Co0.15Al0.05O2表示。以Ni∶Co∶Al=80∶15∶5的摩尔比称量作为镍原料的NiO、作为钴原料的Co(OH)2和作为Al原料的AlOOH,并向其中加入纯水制成浆料,边搅拌边使用循环式介质搅拌型湿式珠磨机将浆料中的固体成分湿式粉碎成中值粒径0.25μm。The positive electrode active material A is a lithium transition metal composite oxide synthesized by the method shown below, and is represented by the composition formula Li 1.05 Ni 0.80 Co 0.15 Al 0.05 O 2 . NiO as a nickel raw material, Co(OH) as a cobalt raw material, and AlOOH as an Al raw material were weighed at a molar ratio of Ni:Co:Al=80:15: 5 , and pure water was added thereto to make a slurry, While stirring, the solid content in the slurry was wet-pulverized to a median particle size of 0.25 μm using a circulating medium agitation type wet bead mill.
通过喷雾干燥器将浆料进行喷雾干燥,得到仅仅包含镍原料、钴原料、铝原料的粒径约10μm的大致球状的造粒粒子。在得到的造粒粒子中添加中值粒径3μm的LiOH粉末,并使Li的摩尔数与Ni、Co和Al的总摩尔数之比为1.05,用高速混合机进行混合,得到镍原料、钴原料、铝原料的造粒粒子和锂原料的混合粉末。在氧流通下、740℃对该混合粉末烧结6小时(升降温速度为5℃/分钟)后,粉碎,通过网眼45μm的筛子,得到正极活性物质A。The slurry was spray-dried with a spray dryer to obtain approximately spherical granulated particles having a particle diameter of about 10 μm and containing only nickel raw materials, cobalt raw materials, and aluminum raw materials. LiOH powder with a median diameter of 3 μm was added to the obtained granulated particles, and the ratio of the molar number of Li to the total molar number of Ni, Co and Al was 1.05, and mixed with a high-speed mixer to obtain nickel raw materials, cobalt Raw materials, granulated particles of aluminum raw materials and mixed powder of lithium raw materials. The mixed powder was sintered at 740° C. for 6 hours under oxygen flow (the heating and cooling rate was 5° C./min), and then pulverized and passed through a sieve with a mesh size of 45 μm to obtain a positive electrode active material A.
[正极活性物质B][Positive electrode active material B]
正极活性物质B是与正极活性物质A同样地合成的锂过渡金属复合氧化物,用组成式Li1.05Ni0.80Co0.15Al0.05O2表示,其不同之处为:变更喷雾干燥的条件,制成粒径约1μm的造粒粒子,并且烧结温度为720℃。The positive electrode active material B is a lithium transition metal composite oxide synthesized in the same way as the positive electrode active material A, and is represented by the composition formula Li 1.05 Ni 0.80 Co 0.15 Al 0.05 O 2 , and the difference is that the conditions of spray drying are changed to make Granulated particles with a particle size of about 1 μm, and a sintering temperature of 720°C.
[正极活性物质C][Positive electrode active material C]
正极活性物质C是按照下面所示的方法合成的在正极活性物质A的表面附着了硫化合物和锑化合物而形成的正极活性物质。即,将96.7重量份正极活性物质A在流动槽中搅拌,同时向其中喷雾1.3重量份硫酸锂(Li2SO4H2O)的水溶液成喷雾状。在得到的混合物中添加2.0重量份三氧化锑(Sb2O3,粒子中值粒径为0.8μm),充分混合。将该混合物转移到氧化铝容器中,在空气氛围下于680℃烧结2小时,得到正极活性物质C。The positive electrode active material C is a positive electrode active material in which a sulfur compound and an antimony compound are attached to the surface of the positive electrode active material A, synthesized by the method shown below. That is, while stirring 96.7 parts by weight of the positive electrode active material A in the flow cell, 1.3 parts by weight of an aqueous solution of lithium sulfate (Li 2 SO 4 H 2 O) was sprayed thereinto in a spray form. 2.0 parts by weight of antimony trioxide (Sb 2 O 3 , particle median diameter: 0.8 μm) was added to the obtained mixture, and mixed well. The mixture was transferred to an alumina container, and sintered at 680° C. for 2 hours in an air atmosphere to obtain a positive electrode active material C.
[正极活性物质D][Positive electrode active material D]
正极活性物质D是通过下面所示的方法合成的锂过渡金属复合氧化物,用组成式Li1.03Ni0.65Co0.20Mn0.15O2表示。以Li∶Co∶Mn=65∶20∶15的摩尔比称量作为镍原料的Ni(OH)2、作为钴原料的Co(OH)2和作为锰原料的Mn2O3,并向其中加入纯水制成浆料,边搅拌边使用循环式介质搅拌型湿式珠磨机将浆料中的固体成分湿式粉碎成中值粒径0.2μm。The positive electrode active material D is a lithium transition metal composite oxide synthesized by the method shown below, and is represented by the composition formula Li 1.03 Ni 0.65 Co 0.20 Mn 0.15 O 2 . Weigh Ni(OH) 2 as a nickel raw material, Co(OH) 2 as a cobalt raw material, and Mn 2 O 3 as a manganese raw material at a molar ratio of Li:Co:Mn=65:20:15, and add Pure water was used to make a slurry, and the solid content in the slurry was wet-milled to a median particle size of 0.2 μm using a circulating medium agitation type wet bead mill while stirring.
通过喷雾干燥器将浆料进行喷雾干燥,得到仅仅包含镍原料、钴原料、锰原料的粒径约12μm的大致球状的造粒粒子。在得到的造粒粒子中添加中值粒径3μm的LiOH粉末,并使Li的摩尔数与Ni、Co和Mn的总摩尔数之比为1.05,用高速混合机进行混合,得到镍原料、钴原料、锰原料的造粒粒子和锂原料的混合粉末。在空气流通下、950℃对该混合粉末烧结12小时(升降温速度为5℃/分钟)后,粉碎,通过网眼45μm的筛子,得到正极活性物质D。The slurry was spray-dried with a spray dryer to obtain approximately spherical granulated particles having a particle diameter of about 12 μm and containing only nickel raw materials, cobalt raw materials, and manganese raw materials. LiOH powder with a median diameter of 3 μm was added to the obtained granulated particles, and the ratio of the molar number of Li to the total molar number of Ni, Co and Mn was 1.05, and mixed with a high-speed mixer to obtain nickel raw materials, cobalt Raw material, granulated particles of manganese raw material and mixed powder of lithium raw material. The mixed powder was sintered at 950° C. for 12 hours under air circulation (the heating and cooling rate was 5° C./min), and then pulverized and passed through a sieve with a mesh size of 45 μm to obtain a positive electrode active material D.
[正极活性物质E][Positive electrode active material E]
正极活性物质E是通过下面所示的方法合成的锂过渡金属复合氧化物,用组成式Li1.04Mn1.84Al0.12O4表示。以Li∶Mn∶Al=1.04∶1.84∶0.12的摩尔比称量作为锂原料的LiOH、作为锰原料的Mn2O3和作为铝原料的AlOOH,并向其中加入纯水制成浆料,边搅拌边使用循环式介质搅拌型湿式珠磨机将浆料中的固体成分湿式粉碎成中值粒径0.5μm。The positive electrode active material E is a lithium transition metal composite oxide synthesized by the method shown below, and is represented by the composition formula Li 1.04 Mn 1.84 Al 0.12 O 4 . LiOH as a lithium raw material, Mn2O3 as a manganese raw material, and AlOOH as an aluminum raw material were weighed at a molar ratio of Li:Mn:Al=1.04:1.84:0.12 , and pure water was added thereto to make a slurry, while While stirring, the solid content in the slurry was wet-milled to a median particle size of 0.5 μm using a circulating medium-stirred wet bead mill.
通过喷雾干燥器将浆料进行喷雾干燥,得到仅仅包含锂原料、锰原料、铝原料的粒径约10μm的大致球状的造粒粒子。将该造粒粒子在氮气流通、900℃下烧结3小时(升温速度为5℃/分钟)后,将流通气体由氮气变换成空气,再在900℃下烧结2小时(降温速度为1℃/分钟)。冷却到室温后,取出并粉碎,通过网眼45μm的筛子,得到正极活性物质E。The slurry was spray-dried by a spray dryer to obtain approximately spherical granulated particles having a particle diameter of about 10 μm and containing only the lithium raw material, the manganese raw material, and the aluminum raw material. The granulated particles were sintered at 900° C. for 3 hours in nitrogen flow (heating rate was 5° C./min), then the flow gas was changed from nitrogen to air, and then sintered at 900° C. for 2 hours (temperature drop rate was 1° C./min. minute). After cooling to room temperature, it was taken out and pulverized, and passed through a sieve with a mesh size of 45 μm to obtain a positive electrode active material E.
正极[2]实施例1Positive electrode [2] Example 1
《正极的制作》"Positive Production"
在N甲基吡咯烷酮溶剂中混合90质量%的作为正极活性物质的正极活性物质A、5质量%的作为导电材料的乙炔黑和5质量%作为粘合剂的聚偏氟乙烯(PVdF),制成浆料。将得到的浆料涂布在厚度15μm的铝箔两面,并干燥,用压制机压延成厚度为66μm,再将其切成具有宽100mm、长100mm大小的正极活性物质层和宽30mm的未涂布部分的形状,作为正极。正极活性物质的密度为2.35g/cm3,(一面的正极活性物质层的厚度)/(集电体的厚度)的值为1.7。Mix 90% by mass of positive electrode active material A as a positive electrode active material, 5% by mass of acetylene black as a conductive material, and 5% by mass of polyvinylidene fluoride (PVdF) as a binder in N-methylpyrrolidone solvent to prepare into slurry. The obtained slurry was coated on both sides of an aluminum foil with a thickness of 15 μm, dried, rolled into a thickness of 66 μm with a press, and then cut into a positive electrode active material layer with a width of 100 mm and a length of 100 mm and an uncoated layer with a width of 30 mm. The shape of the part, as the positive electrode. The density of the positive electrode active material was 2.35 g/cm 3 , and the value of (thickness of positive electrode active material layer on one surface)/(thickness of current collector) was 1.7.
《负极的制作》"The Making of Negative Pole"
在98重量份人造石墨粉末KS-44(timcal公司制造,商品名)中添加100重量份作为增稠剂的羧甲基纤维素钠的水性分散液(羧甲基纤维素钠的浓度为1质量%),2重量份作为粘合剂的苯乙烯-丁二烯橡胶的水性分散液(苯乙烯-丁二烯橡胶的浓度为50质量%),用分散器进行混合,制成浆料。将得到的浆料涂布在厚度10μm的铜箔两面,并干燥,用压制机压延成厚度为75μm,再将其切成具有宽104mm、长104mm大小的负极活性物质层和宽30mm的未涂布部分的形状,作为负极。In 98 parts by weight of artificial graphite powder KS-44 (manufactured by timcal company, trade name), add 100 parts by weight of an aqueous dispersion of sodium carboxymethyl cellulose as a thickener (the concentration of sodium carboxymethyl cellulose is 1 mass %), 2 parts by weight of an aqueous dispersion of styrene-butadiene rubber as a binder (the concentration of styrene-butadiene rubber is 50% by mass), and mixed with a disperser to prepare a slurry. The obtained slurry is coated on both sides of a copper foil with a thickness of 10 μm, dried, rolled into a thickness of 75 μm with a press machine, and then cut into a negative electrode active material layer with a width of 104 mm and a length of 104 mm and an uncoated layer with a width of 30 mm. The shape of the cloth part is used as the negative electrode.
《电解液的制作》"Making Electrolyte"
在干燥的氩氛围下,在纯化的碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的体积比3∶3∶4的混合溶剂中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。另外,含有0.3质量%的六甲基环三硅氧烷。Under a dry argon atmosphere, in a mixed solvent of purified ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) with a volume ratio of 3:3:4, at 1mol/L The concentration dissolves well-dried lithium hexafluorophosphate (LiPF 6 ). In addition, 0.3% by mass of hexamethylcyclotrisiloxane was contained.
《电池的制作》"Battery Making"
将34片正极和35片负极交替配置,各电极之间夹入多孔聚乙烯片隔板(厚度为25μm),进行叠层。此时,使正极活性物质面与负极活性物质面相对,并且不会露出负极活性物质面之外。将该正极和负极各自的未涂布部分焊接,制作集电片,将电极组密封在电池罐(外部尺寸:120×110×10mm)中。然后,在装有电极组的电池罐中注入20mL电解液,使电极充分浸透,密封而制作电池。正极的电极面积总和与电池的外壳表面积总和之比为21.9。34 sheets of positive electrodes and 35 sheets of negative electrodes were alternately arranged, and a porous polyethylene sheet separator (thickness: 25 μm) was sandwiched between the electrodes to perform lamination. At this time, the surface of the positive electrode active material faces the surface of the negative electrode active material without protruding from the surface of the negative electrode active material. The respective uncoated parts of the positive and negative electrodes were welded to produce current collector sheets, and the electrode group was sealed in a battery can (outer dimensions: 120×110×10 mm). Then, 20 mL of electrolytic solution was poured into the battery can containing the electrode group, the electrodes were sufficiently permeated, and the battery was sealed to produce a battery. The ratio of the sum of the electrode areas of the positive electrode to the sum of the surface areas of the battery case was 21.9.
《电池的评价》"Battery Evaluation"
(电池容量的测定方法)(Measuring method of battery capacity)
对未经充放电循环的新电池,在25℃、4.1V~3.0V的电压范围下,以0.2C的电流值(将1小时放电额定容量的电流值作为1C,所述额定容量取决于1小时率(one-hour-rate)的放电容量,下同)进行5个循环的初期充放电。此时的第5个循环的0.2C放电容量作为初期容量。电池评价的结果示于正极[2]表2。For a new battery that has not been charged and discharged, at 25°C and a voltage range of 4.1V to 3.0V, use a current value of 0.2C (the current value of the rated capacity of 1 hour discharge is taken as 1C, and the rated capacity depends on 1 One-hour-rate (one-hour-rate) discharge capacity, the same below) was performed for 5 cycles of initial charge and discharge. The 0.2C discharge capacity at the fifth cycle at this time was taken as the initial capacity. The results of the battery evaluation are shown in Table 2 of the positive electrode [2].
(初期输出功率的测定方法)(Measurement method of initial output power)
在25℃的环境下,通过0.2C的恒定电流进行150分钟充电,分别以0.1C、0.3C、1.0C、3.0C、10.0C放电10秒钟,测定第10秒的电压。将电流-电压直线和下限电压(3V)所包围的三角形的面积作为输出功率(W)。电池评价的结果示于正极[2]表2。In an environment of 25°C, charge was performed for 150 minutes with a constant current of 0.2C, and discharge was performed at 0.1C, 0.3C, 1.0C, 3.0C, and 10.0C for 10 seconds, respectively, and the voltage at the 10th second was measured. The area of the triangle surrounded by the current-voltage line and the lower limit voltage (3V) was taken as the output power (W). The results of the battery evaluation are shown in Table 2 of the positive electrode [2].
(循环试验(耐久后电池容量和耐久后输出功率的测定方法))(Cycle test (measuring method of battery capacity after endurance and output power after endurance))
在被视为锂二次电池的实际使用上限温度的60℃的高温环境下,进行循环试验。以2C恒定电流恒定电压法充电到充电上限电压4.1V后,以2C的恒定电流放电至放电终止电压3.0V,以此作为一个充放电循环,反复进行该循环,直至500循环。对于循环试验结束后的电池,在25℃的环境下以0.2C的电流值进行3个循环的充放电,以第3个循环的0.2C放电容量作为耐久后电池容量。另外,对于循环试验结束后的电池,进行输出功率测定,作为耐久后输出功率。电池评价的结果示于正极[2]表2。The cycle test was performed in a high-temperature environment of 60° C., which is regarded as the upper limit temperature for practical use of lithium secondary batteries. After charging to the charging upper limit voltage of 4.1V with 2C constant current and constant voltage method, discharge at a constant current of 2C to the end-of-discharge voltage of 3.0V, which is regarded as a charge-discharge cycle, and this cycle is repeated until 500 cycles. For the battery after the cycle test, three cycles of charging and discharging were performed at a current value of 0.2C in an environment of 25°C, and the 0.2C discharge capacity of the third cycle was used as the battery capacity after endurance. In addition, for the battery after the cycle test, the output power was measured, and it was set as the output power after durability. The results of the battery evaluation are shown in Table 2 of the positive electrode [2].
正极[2]实施例2Positive electrode [2] Example 2
使电解液含有0.3质量%的甲磺酸三甲基甲硅烷基酯来代替六甲基环三硅氧烷,除此以外,与正极[2]实施例1同样实施。电池评价的结果示于正极[2]表2。It carried out similarly to positive electrode [2] Example 1 except having made electrolytic solution contain 0.3 mass % of trimethylsilyl methanesulfonate instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 2 of the positive electrode [2].
正极[2]实施例3Positive electrode [2] Example 3
使电解液含有0.3质量%的苯基二甲基氟硅烷来代替六甲基环三硅氧烷,除此以外,与正极[2]实施例1同样实施。电池评价的结果示于正极[2]表2。The procedure was carried out in the same manner as in positive electrode [2] Example 1 except that the electrolytic solution contained 0.3% by mass of phenyldimethylfluorosilane instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 2 of the positive electrode [2].
正极[2]实施例4Positive electrode [2] embodiment 4
使电解液含有0.3质量%的二氟磷酸锂来代替六甲基环三硅氧烷,除此以外,与正极[2]实施例1同样实施。电池评价的结果示于正极[2]表2。The procedure was carried out in the same manner as in positive electrode [2] Example 1 except that the electrolytic solution contained 0.3% by mass of lithium difluorophosphate instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 2 of the positive electrode [2].
正极[2]比较例1Positive electrode [2] Comparative example 1
除了电解液中不含六甲基环三硅氧烷以外,与正极[2]实施例1同样实施。电池评价的结果示于正极[2]表2。Except that hexamethylcyclotrisiloxane is not contained in the electrolytic solution, it is carried out in the same manner as the positive electrode [2] Example 1. The results of the battery evaluation are shown in Table 2 of the positive electrode [2].
正极[2]表2Positive pole [2] Table 2
[表8][Table 8]
正极[2]实施例5Positive electrode [2] Example 5
除了使用正极活性物质A和正极活性物质B以2∶1的质量比充分混合而得到的正极活性物质作为正极活性物质,制成正极以外,与正极[2]实施例1同样实施。该混合的正极活性物质的BET比表面积为1.4m2/g,平均一次粒径为0.2μm,中值粒径d50为6.3μm,振实密度为1.9g/cm2。电池评价的结果示于正极[2]表3。Except that positive electrode active material A and positive electrode active material B were fully mixed at a mass ratio of 2:1 as the positive electrode active material, the positive electrode was made in the same manner as positive electrode [2] Example 1. The BET specific surface area of the mixed positive electrode active material was 1.4 m 2 /g, the average primary particle size was 0.2 μm, the median particle size d 50 was 6.3 μm, and the tap density was 1.9 g/cm 2 . The results of the battery evaluation are shown in Table 3 of the positive electrode [2].
正极[2]实施例6Positive electrode [2] Embodiment 6
使电解液含有0.3质量%的甲磺酸三甲基甲硅烷基酯来代替六甲基环三硅氧烷,除此以外,与正极[2]实施例5同样实施。电池评价的结果示于正极[2]表3。The same procedure as in Example 5 of the positive electrode [2] was performed except that the electrolytic solution contained 0.3% by mass of trimethylsilyl methanesulfonate instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 3 of the positive electrode [2].
正极[2]实施例7Positive electrode [2] Example 7
使电解液含有0.3质量%的苯基二甲基氟硅烷来代替六甲基环三硅氧烷,除此以外,与正极[2]实施例5同样实施。电池评价的结果示于正极[2]表3。It carried out similarly to positive electrode [2] Example 5 except having made electrolytic solution contain 0.3 mass % of phenyldimethylfluorosilanes instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 3 of the positive electrode [2].
正极[2]实施例8Positive electrode [2] embodiment 8
使电解液含有0.3质量%的二氟磷酸锂来代替六甲基环三硅氧烷,除此以外,与正极[2]实施例5同样实施。电池评价的结果示于正极[2]表3。The same procedure was carried out as in Example 5 of the positive electrode [2] except that the electrolytic solution contained 0.3% by mass of lithium difluorophosphate instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 3 of the positive electrode [2].
正极[2]比较例2Positive electrode [2] Comparative example 2
除了电解液中不含六甲基环三硅氧烷以外,与正极[2]实施例5同样实施。电池评价的结果示于正极[2]表3。Except that the electrolytic solution does not contain hexamethylcyclotrisiloxane, it is carried out in the same manner as the positive electrode [2] Example 5. The results of the battery evaluation are shown in Table 3 of the positive electrode [2].
正极[2]表3Positive pole [2] Table 3
[表9][Table 9]
正极[2]实施例9Positive electrode [2] embodiment 9
除了使用正极活性物质C作为正极活性物质,制成正极以外,与正极[2]实施例1同样实施。电池评价的结果示于正极[2]表4。Except using the positive electrode active material C as the positive electrode active material to prepare a positive electrode, it was carried out in the same manner as the positive electrode [2] Example 1. The results of the battery evaluation are shown in Table 4 of the positive electrode [2].
正极[2]实施例10Positive electrode [2] Example 10
使电解液含有0.3质量%的甲磺酸三甲基甲硅烷基酯来代替六甲基环三硅氧烷,除此以外,与正极[2]实施例9同样实施。电池评价的结果示于正极[2]表4。The procedure was carried out in the same manner as in positive electrode [2] Example 9 except that the electrolytic solution contained 0.3% by mass of trimethylsilyl methanesulfonate instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 4 of the positive electrode [2].
正极[2]实施例11Positive electrode [2] Example 11
使电解液含有0.3质量%的苯基二甲基氟硅烷来代替六甲基环三硅氧烷,除此以外,与正极[2]实施例9同样实施。电池评价的结果示于正极[2]表4。The same procedure as in positive electrode [2] Example 9 was carried out except that the electrolytic solution contained 0.3% by mass of phenyldimethylfluorosilane instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 4 of the positive electrode [2].
正极[2]实施例12Positive electrode [2] Example 12
使电解液含有0.3质量%的二氟磷酸锂来代替六甲基环三硅氧烷,除此以外,与正极[2]实施例9同样实施。电池评价的结果示于正极[2]表4。The procedure was carried out in the same manner as in positive electrode [2] Example 9 except that the electrolytic solution contained 0.3% by mass of lithium difluorophosphate instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 4 of the positive electrode [2].
正极[2]比较例3Positive electrode [2] Comparative example 3
除了电解液中不含六甲基环三硅氧烷以外,与正极[2]实施例9同样实施。电池评价的结果示于正极[2]表4。Except that the electrolytic solution does not contain hexamethylcyclotrisiloxane, the same implementation is carried out as in the positive electrode [2] Example 9. The results of the battery evaluation are shown in Table 4 of the positive electrode [2].
正极[2]表4Positive pole [2] Table 4
[表10][Table 10]
正极[2]实施例13Positive electrode [2] Example 13
使用正极活性物质D作为正极活性物质,制成正极,用压制机压延成厚度为68μm,并使用34片该正极和35片负极,除此之外,与正极[2]实施例1同样地制作电池。(正极一面的正极活性物质层厚度/正极集电体的厚度)为1.8,正极的电极面积的总和与电池的外壳表面积总和之比为21.9。容量测定的电压范围为3.0~4.2V,循环试验的上限电压为4.2V,除此以外,与正极[2]实施例1同样地评价电池。电池评价的结果示于正极[2]表5。Use the positive active material D as the positive active material to make the positive pole, roll it into a thickness of 68 μm with a press, and use 34 pieces of the positive pole and 35 negative poles. In addition, it is made in the same way as the positive pole [2] Example 1 Battery. (The thickness of the positive electrode active material layer on the positive electrode side/the thickness of the positive electrode current collector) was 1.8, and the ratio of the total electrode area of the positive electrode to the total surface area of the battery case was 21.9. The battery was evaluated in the same manner as in Example 1 of the positive electrode [2], except that the voltage range of the capacity measurement was 3.0 to 4.2 V, and the upper limit voltage of the cycle test was 4.2 V. The results of the battery evaluation are shown in Table 5 of the positive electrode [2].
正极[2]实施例14Positive electrode [2] Example 14
使电解液含有0.3质量%的甲磺酸三甲基甲硅烷基酯来代替六甲基环三硅氧烷,除此以外,与正极[2]实施例13同样实施。电池评价的结果示于正极[2]表5。The same procedure as in Example 13 of the positive electrode [2] was performed except that the electrolytic solution contained 0.3% by mass of trimethylsilyl methanesulfonate instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 5 of the positive electrode [2].
正极[2]实施例15Positive electrode [2] Example 15
使电解液含有0.3质量%的苯基二甲基氟硅烷来代替六甲基环三硅氧烷,除此以外,与正极[2]实施例13同样实施。电池评价的结果示于正极[2]表5。The procedure was carried out in the same manner as in positive electrode [2] Example 13 except that the electrolytic solution contained 0.3% by mass of phenyldimethylfluorosilane instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 5 of the positive electrode [2].
正极[2]实施例16Positive electrode [2] Example 16
使电解液含有0.3质量%的二氟磷酸锂来代替六甲基环三硅氧烷,除此以外,与正极[2]实施例13同样实施。电池评价的结果示于正极[2]表5。The procedure was carried out in the same manner as in positive electrode [2] Example 13 except that the electrolytic solution contained 0.3% by mass of lithium difluorophosphate instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 5 of the positive electrode [2].
正极[2]比较例4Positive electrode [2] Comparative example 4
除了电解液中不含六甲基环三硅氧烷以外,与正极[2]实施例13同样实施。电池评价的结果示于正极[2]表5。Except that hexamethylcyclotrisiloxane is not contained in the electrolytic solution, the same implementation is carried out as in the positive electrode [2] Example 13. The results of the battery evaluation are shown in Table 5 of the positive electrode [2].
正极[2]表5Positive electrode [2] Table 5
[表11][Table 11]
正极[2]实施例17Positive electrode [2] Example 17
使用正极活性物质A和正极活性物质E以2∶1的质量比充分混合而得到的正极活性物质作为正极活性物质,制成正极,用压制机压延成厚度为76μm,并使用32片该正极和33片负极,除此之外,与正极[2]实施例1同样实施。(正极一面的正极活性物质层厚度/正极集电体的厚度)为2.0,正极的电极面积的总和与电池的外壳表面积总和之比为20.6。另外,该混合的正极活性物质的BET比表面积为0.7m2/g,平均一次粒径为0.6μm,中值粒径d50为8.7μm,振实密度为2.2g/cm3。电池评价的结果示于正极[2]表6。Use positive active material A and positive active material E to fully mix the positive active material that obtains with the mass ratio of 2: 1 as positive active material, make positive pole, be rolled into thickness with press machine and be 76 μ m, and use 32 this positive pole and Except for 33 negative electrodes, the same implementation was carried out as in Example 1 of the positive electrode [2]. (The thickness of the positive electrode active material layer on the positive electrode side/the thickness of the positive electrode current collector) was 2.0, and the ratio of the total electrode area of the positive electrode to the total surface area of the battery case was 20.6. In addition, the mixed positive electrode active material had a BET specific surface area of 0.7 m 2 /g, an average primary particle size of 0.6 μm, a median particle size d 50 of 8.7 μm, and a tap density of 2.2 g/cm 3 . The results of the battery evaluation are shown in Table 6 of the positive electrode [2].
正极[2]实施例18Positive electrode [2] Example 18
使电解液含有0.3质量%的甲磺酸三甲基甲硅烷基酯来代替六甲基环三硅氧烷,除此以外,与正极[2]实施例17同样实施。电池评价的结果示于正极[2]表6。It carried out similarly to positive electrode [2] Example 17 except having made electrolytic solution contain 0.3 mass % of trimethylsilyl methanesulfonate instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 6 of the positive electrode [2].
正极[2]实施例19Positive electrode [2] Example 19
使电解液含有0.3质量%的苯基二甲基氟硅烷来代替六甲基环三硅氧烷,除此以外,与正极[2]实施例17同样实施。电池评价的结果示于正极[2]表6。The same procedure as in positive electrode [2] Example 17 was carried out except that the electrolytic solution contained 0.3% by mass of phenyldimethylfluorosilane instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 6 of the positive electrode [2].
正极[2]实施例20Positive electrode [2] Example 20
使电解液含有0.3质量%的二氟磷酸锂来代替六甲基环三硅氧烷,除此以外,与正极[2]实施例17同样实施。电池评价的结果示于正极[2]表6。The procedure was carried out in the same manner as in positive electrode [2] Example 17, except that the electrolytic solution contained 0.3% by mass of lithium difluorophosphate instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 6 of the positive electrode [2].
正极[2]比较例5Positive electrode [2] Comparative example 5
除了电解液中不含六甲基环三硅氧烷以外,与正极[2]实施例17同样实施。电池评价的结果示于正极[2]表6。Except that the electrolytic solution does not contain hexamethylcyclotrisiloxane, the same implementation is carried out as in the positive electrode [2] Example 17. The results of the battery evaluation are shown in Table 6 of the positive electrode [2].
正极[2]表6Positive pole [2] Table 6
[表12][Table 12]
由正极[2]表2~正极[2]表6的结果可知,在任一个正极中,由于在电解液中都含有特定化合物,因此,输出功率、容量保持率得到提高,并且即使在循环试验后,也可以充分保持电池容量和输出功率。From the results in Table 2 of the positive electrode [2] to Table 6 of the positive electrode [2], it can be seen that in any positive electrode, since the electrolyte contains a specific compound, the output power and capacity retention rate are improved, and even after the cycle test , can also fully maintain the battery capacity and output power.
正极[3][正极活性物质]Positive electrode [3] [positive active material]
下面的实施例和比较例中使用的正极活性物质的种类和物性如下。The types and physical properties of positive electrode active materials used in the following Examples and Comparative Examples are as follows.
正极[3]表1Positive pole [3] Table 1
[表13][Table 13]
正极[3]表1中,作为正极活性物质的物性,按照上述记载的方法,进行BET比表面积、平均一次粒径(用SEM测定)、中值粒径d50、振实密度的测定。Positive electrode [3] In Table 1, as the physical properties of the positive electrode active material, BET specific surface area, average primary particle size (measured by SEM), median particle size d 50 , and tap density were measured according to the method described above.
[正极活性物质A][Positive electrode active material A]
正极活性物质A是通过下面所示的方法合成的锂过渡金属复合氧化物,用组成式Li1.05Ni0.8Co0.2O2表示。以Ni∶Co=80∶20的摩尔比称量作为镍原料的NiO和作为钴原料的Co(OH)2,并向其中加入纯水制成浆料,边搅拌边使用循环式介质搅拌型湿式珠磨机将浆料中的固体成分湿式粉碎成中值粒径0.25μm。The positive electrode active material A is a lithium transition metal composite oxide synthesized by the method shown below, and is represented by the composition formula Li 1.05 Ni 0.8 Co 0.2 O 2 . Weigh NiO as a nickel raw material and Co(OH) 2 as a cobalt raw material at a molar ratio of Ni:Co=80:20, and add pure water to them to make a slurry, and use a circulating medium stirring type wet type while stirring. The bead mill wet-milled the solid content in the slurry to a median particle size of 0.25 μm.
通过喷雾干燥器将浆料进行喷雾干燥,得到仅仅包含镍原料、钴原料的粒径约8μm的大致球状的造粒粒子。在得到的造粒粒子中添加中值粒径3μm的LiOH粉末,并使Li的摩尔数与Ni和Co的总摩尔数之比为1.05,用高速混合机进行混合,得到镍原料、钴原料的造粒粒子和锂原料的混合粉末。在氧流通下、740℃对该混合粉末烧结6小时(升降温速度为5℃/分钟)后,粉碎,通过网眼45μm的筛子,得到正极活性物质A。The slurry was spray-dried by a spray dryer to obtain approximately spherical granulated particles having a particle diameter of about 8 μm and containing only the nickel raw material and the cobalt raw material. LiOH powder with a median diameter of 3 μm was added to the obtained granulated particles, and the ratio of the molar number of Li to the total molar number of Ni and Co was 1.05, and mixed with a high-speed mixer to obtain nickel raw materials and cobalt raw materials. Mixed powder of granulated particles and lithium raw material. The mixed powder was sintered at 740° C. for 6 hours under oxygen flow (the heating and cooling rate was 5° C./min), and then pulverized and passed through a sieve with a mesh size of 45 μm to obtain a positive electrode active material A.
[正极活性物质B][Positive electrode active material B]
正极活性物质B是与正极活性物质A同样地合成的锂过渡金属复合氧化物,用组成式Li1.05Ni0.8Co0.2O2表示,其不同之处为:变更喷雾干燥的条件,制成粒径约1μm的造粒粒子,并且烧结温度为720℃。The positive electrode active material B is a lithium transition metal composite oxide synthesized in the same way as the positive electrode active material A, expressed by the composition formula Li 1.05 Ni 0.8 Co 0.2 O 2 , the difference is that the spray drying conditions are changed to make the particle size The granulated particles are about 1 μm, and the sintering temperature is 720°C.
[正极活性物质C][Positive electrode active material C]
正极活性物质C是按照下面所示的方法合成的在正极活性物质A的表面附着了硫化合物和锑化合物而形成的正极活性物质。即,将96.7重量份正极活性物质A在流动槽中搅拌,同时向其中喷雾1.3重量份硫酸锂(Li2SO4H2O)的水溶液成喷雾状。在得到的混合物中添加2.0重量份三氧化锑(Sb2O3,粒子中值粒径为0.8μm),充分混合。将该混合物转移到氧化铝容器中,在空气氛围下于680℃烧结2小时,得到正极活性物质C。The positive electrode active material C is a positive electrode active material in which a sulfur compound and an antimony compound are attached to the surface of the positive electrode active material A, synthesized by the method shown below. That is, while stirring 96.7 parts by weight of the positive electrode active material A in the flow cell, 1.3 parts by weight of an aqueous solution of lithium sulfate (Li 2 SO 4 H 2 O) was sprayed thereinto in a spray form. 2.0 parts by weight of antimony trioxide (Sb 2 O 3 , particle median diameter: 0.8 μm) was added to the obtained mixture, and mixed well. The mixture was transferred to an alumina container, and sintered at 680° C. for 2 hours in an air atmosphere to obtain a positive electrode active material C.
[正极活性物质D][Positive electrode active material D]
正极活性物质D是通过下面所示的方法合成的锂过渡金属复合氧化物,用组成式Li1.04Mn1.84Al0.12O4表示。以Li∶Mn∶Al=1.04∶1.84∶0.12的摩尔比称量作为锂原料的LiOH、作为锰原料的Mn2O3和作为铝原料的AlOOH,并向其中加入纯水制成浆料,边搅拌边使用循环式介质搅拌型湿式珠磨机将浆料中的固体成分湿式粉碎成中值粒径0.5μm。The positive electrode active material D is a lithium transition metal composite oxide synthesized by the method shown below, and is represented by the composition formula Li 1.04 Mn 1.84 Al 0.12 O 4 . LiOH as a lithium raw material, Mn2O3 as a manganese raw material, and AlOOH as an aluminum raw material were weighed at a molar ratio of Li:Mn:Al=1.04:1.84:0.12 , and pure water was added thereto to make a slurry, while While stirring, the solid content in the slurry was wet-milled to a median particle size of 0.5 μm using a circulating medium-stirred wet bead mill.
通过喷雾干燥器将浆料进行喷雾干燥,得到仅仅包含锂原料、锰原料、铝原料的粒径约10μm的大致球状的造粒粒子。将该造粒粒子在氮气流通、900℃下烧结3小时(升温速度为5℃/分钟)后,将流通气体由氮气变换成空气,再在900℃下烧结2小时(降温速度为1℃/分钟)。冷却到室温后,取出并粉碎,通过网眼45μm的筛子,得到正极活性物质D。The slurry was spray-dried by a spray dryer to obtain approximately spherical granulated particles having a particle diameter of about 10 μm and containing only the lithium raw material, the manganese raw material, and the aluminum raw material. The granulated particles were sintered at 900° C. for 3 hours in nitrogen flow (heating rate was 5° C./min), then the flow gas was changed from nitrogen to air, and then sintered at 900° C. for 2 hours (temperature drop rate was 1° C./min. minute). After cooling to room temperature, it was taken out and pulverized, and passed through a sieve with a mesh size of 45 μm to obtain a positive electrode active material D.
正极[3]实施例1Positive electrode [3] embodiment 1
《正极的制作》"Positive Production"
在N-甲基吡咯烷酮溶剂中混合90质量%的作为正极活性物质的正极活性物质A、5质量%的作为导电材料的乙炔黑和5质量%作为粘合剂的聚偏氟乙烯(PVdF),制成浆料。将得到的浆料涂布在厚度15μm的铝箔两面,并干燥,用压制机压延成厚度为65μm,再将其切成具有宽100mm、长100mm大小的正极活性物质层和宽30mm的未涂布部分的形状,制成正极。正极活性物质的密度为2.35g/cm3,(一面的正极活性物质层的厚度)/(集电体的厚度)的值为1.7。In N-methylpyrrolidone solvent, mix 90% by mass of positive electrode active material A as a positive electrode active material, 5% by mass of acetylene black as a conductive material and 5% by mass of polyvinylidene fluoride (PVdF) as a binder, Make slurry. The obtained slurry is coated on both sides of an aluminum foil with a thickness of 15 μm, dried, rolled into a thickness of 65 μm with a press, and then cut into a positive electrode active material layer with a width of 100 mm and a length of 100 mm and an uncoated layer with a width of 30 mm. The shape of the part is made into a positive electrode. The density of the positive electrode active material was 2.35 g/cm 3 , and the value of (thickness of positive electrode active material layer on one side)/(thickness of current collector) was 1.7.
《负极的制作》"The Making of Negative Pole"
在98重量份人造石墨粉末KS-44(timcal公司制造,商品名)中添加100重量份作为增稠剂的羧甲基纤维素钠的水性分散液(羧甲基纤维素钠的浓度为1质量%),2重量份作为粘合剂的苯乙烯-丁二烯橡胶的水性分散液(苯乙烯-丁二烯橡胶的浓度为50质量%),用分散器进行混合,制成浆料。将得到的浆料涂布在厚度10μm的铜箔两面,并干燥,用压制机压延成厚度为75μm,再将其切成具有宽104mm、长104mm大小的负极活性物质层和宽30mm的未涂布部分的形状,作为负极。In 98 parts by weight of artificial graphite powder KS-44 (manufactured by timcal company, trade name), add 100 parts by weight of an aqueous dispersion of sodium carboxymethyl cellulose as a thickener (the concentration of sodium carboxymethyl cellulose is 1 mass %), 2 parts by weight of an aqueous dispersion of styrene-butadiene rubber as a binder (the concentration of styrene-butadiene rubber is 50% by mass), and mixed with a disperser to prepare a slurry. The obtained slurry is coated on both sides of a copper foil with a thickness of 10 μm, dried, rolled into a thickness of 75 μm with a press machine, and then cut into a negative electrode active material layer with a width of 104 mm and a length of 104 mm and an uncoated layer with a width of 30 mm. The shape of the cloth part is used as the negative electrode.
《电解液的制作》"Making Electrolyte"
在干燥的氩氛围下,在纯化的碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的体积比3∶3∶4的混合溶剂中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。另外,含有0.3质量%的六甲基环三硅氧烷。Under a dry argon atmosphere, in a mixed solvent of purified ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) with a volume ratio of 3:3:4, at 1mol/L The concentration dissolves well-dried lithium hexafluorophosphate (LiPF 6 ). In addition, 0.3% by mass of hexamethylcyclotrisiloxane was contained.
《电池的制作》"Battery Making"
将34片正极和35片负极交替配置,各电极之间夹入多孔聚乙烯片隔板(厚度为25μm),进行叠层。此时,使正极活性物质面与负极活性物质面相对,并且不会露出负极活性物质面之外。将该正极和负极各自的未涂布部分焊接,制作集电片,将电极组密封在电池罐(外部尺寸:120×110×10mm)中。然后,在装有电极组的电池罐中注入20mL电解液,使电极充分浸透,密封而制作电池。正极的电极面积总和与电池的外壳表面积总和之比为21.9。34 sheets of positive electrodes and 35 sheets of negative electrodes were alternately arranged, and a porous polyethylene sheet separator (thickness: 25 μm) was sandwiched between the electrodes to perform lamination. At this time, the surface of the positive electrode active material faces the surface of the negative electrode active material without protruding from the surface of the negative electrode active material. The respective uncoated parts of the positive and negative electrodes were welded to produce current collector sheets, and the electrode group was sealed in a battery can (outer dimensions: 120×110×10 mm). Then, 20 mL of electrolytic solution was poured into the battery can containing the electrode group, the electrodes were sufficiently permeated, and the battery was sealed to produce a battery. The ratio of the sum of the electrode areas of the positive electrode to the sum of the surface areas of the battery case was 21.9.
《电池的评价》"Battery Evaluation"
(电池容量的测定方法)(Measuring method of battery capacity)
对未经充放电循环的新电池,在25℃、4.1V~3.0V的电压范围下,以0.2C的电流值(将1小时放电额定容量的电流值作为1C,所述额定容量取决于1小时率(one-hour-rate)的放电容量,下同)进行5个循环的初期充放电。此时的第5个循环的0.2C放电容量作为初期容量。电池评价的结果示于正极[3]表2。For a new battery that has not been charged and discharged, at 25°C and a voltage range of 4.1V to 3.0V, use a current value of 0.2C (the current value of the rated capacity of 1 hour discharge is taken as 1C, and the rated capacity depends on 1 One-hour-rate (one-hour-rate) discharge capacity, the same below) was performed for 5 cycles of initial charge and discharge. The 0.2C discharge capacity at the fifth cycle at this time was taken as the initial capacity. The results of the battery evaluation are shown in Table 2 of the positive electrode [3].
(初期输出功率的测定方法)(Measurement method of initial output power)
在25℃的环境下,通过0.2C的恒定电流进行150分钟充电,分别以0.1C、0.3C、1.0C、3.0C、10.0C放电10秒钟,测定第10秒的电压。将电流-电压直线和下限电压(3V)所包围的三角形的面积作为输出功率(W)。电池评价的结果示于正极[3]表2。In an environment of 25°C, charge was performed for 150 minutes with a constant current of 0.2C, and discharge was performed at 0.1C, 0.3C, 1.0C, 3.0C, and 10.0C for 10 seconds, respectively, and the voltage at the 10th second was measured. The area of the triangle surrounded by the current-voltage line and the lower limit voltage (3V) was taken as the output power (W). The results of the battery evaluation are shown in Table 2 of the positive electrode [3].
(循环试验(耐久后电池容量和耐久后输出功率的测定方法))(Cycle test (measuring method of battery capacity after endurance and output power after endurance))
在被视为锂二次电池的实际使用上限温度的60℃的高温环境下,进行循环试验。以2C恒定电流恒定电压法充电到充电上限电压4.1V后,以2C的恒定电流放电至放电终止电压3.0V,以此作为一个充放电循环,反复进行该循环,直至500循环。对于循环试验结束后的电池,在25℃的环境下以0.2C的电流值进行3个循环的充放电,以第3个循环的0.2C放电容量作为耐久后电池容量。另外,对于循环试验结束后的电池,进行输出功率测定,作为耐久后输出功率。电池评价的结果示于正极[3]表2。The cycle test was performed in a high-temperature environment of 60° C., which is regarded as the upper limit temperature for practical use of lithium secondary batteries. After charging to the charging upper limit voltage of 4.1V with 2C constant current and constant voltage method, discharge at a constant current of 2C to the end-of-discharge voltage of 3.0V, which is regarded as a charge-discharge cycle, and this cycle is repeated until 500 cycles. For the battery after the cycle test, three cycles of charging and discharging were performed at a current value of 0.2C in an environment of 25°C, and the 0.2C discharge capacity of the third cycle was used as the battery capacity after endurance. In addition, for the battery after the cycle test, the output power was measured, and it was set as the output power after durability. The results of the battery evaluation are shown in Table 2 of the positive electrode [3].
正极[3]实施例2Positive electrode [3] embodiment 2
使电解液含有0.3质量%的甲磺酸三甲基甲硅烷基酯来代替六甲基环三硅氧烷,除此以外,与正极[3]实施例1同样实施。电池评价的结果示于正极[3]表2。The same procedure as in Example 1 of the positive electrode [3] was performed except that the electrolytic solution contained 0.3% by mass of trimethylsilyl methanesulfonate instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 2 of the positive electrode [3].
正极[3]实施例3Positive electrode [3] Example 3
使电解液含有0.3质量%的苯基二甲基氟硅烷来代替六甲基环三硅氧烷,除此以外,与正极[3]实施例1同样实施。电池评价的结果示于正极[3]表2。It carried out similarly to positive electrode [3] Example 1 except having made electrolytic solution contain 0.3 mass % of phenyldimethylfluorosilanes instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 2 of the positive electrode [3].
正极[3]实施例4Positive electrode [3] embodiment 4
使电解液含有0.3质量%的二氟磷酸锂来代替六甲基环三硅氧烷,除此以外,与正极[3]实施例1同样实施。电池评价的结果示于正极[3]表2。The same procedure was carried out as in Example 1 of the positive electrode [3] except that the electrolytic solution contained 0.3% by mass of lithium difluorophosphate instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 2 of the positive electrode [3].
正极[3]比较例1Positive electrode [3] Comparative Example 1
除了电解液中不含六甲基环三硅氧烷以外,与正极[3]实施例1同样实施。电池评价的结果示于正极[3]表2。Except that the electrolytic solution does not contain hexamethylcyclotrisiloxane, it is carried out in the same manner as the positive electrode [3] Example 1. The results of the battery evaluation are shown in Table 2 of the positive electrode [3].
正极[3]表2Positive pole [3] Table 2
[表14][Table 14]
正极[3]实施例5Positive electrode [3] Example 5
除了使用正极活性物质A和正极活性物质B以2∶1的质量比充分混合而得到的正极活性物质作为正极活性物质,制成正极以外,与正极[3]实施例1同样实施。该混合的正极活性物质的BET比表面积为1.8m2/g,平均一次粒径为0.2μm,中值粒径d50为4.3μm,振实密度为1.8g/cm2。电池评价的结果示于正极[3]表3。Except using the positive electrode active material obtained by mixing the positive electrode active material A and the positive electrode active material B at a mass ratio of 2:1 as the positive electrode active material, the positive electrode [3] Example 1 was carried out in the same manner. The BET specific surface area of the mixed positive electrode active material was 1.8 m 2 /g, the average primary particle size was 0.2 μm, the median particle size d 50 was 4.3 μm, and the tap density was 1.8 g/cm 2 . The results of the battery evaluation are shown in Table 3 of the positive electrode [3].
正极[3]实施例6Positive electrode [3] embodiment 6
使电解液含有0.3质量%的甲磺酸三甲基甲硅烷基酯来代替六甲基环三硅氧烷,除此以外,与正极[3]实施例5同样实施。电池评价的结果示于正极[3]表3。It carried out similarly to positive electrode [3] Example 5 except having made electrolytic solution contain 0.3 mass % of trimethylsilyl methanesulfonate instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 3 of the positive electrode [3].
正极[3]实施例7Positive electrode [3] Example 7
使电解液含有0.3质量%的苯基二甲基氟硅烷来代替六甲基环三硅氧烷,除此以外,与正极[3]实施例5同样实施。电池评价的结果示于正极[3]表3。It carried out similarly to positive electrode [3] Example 5 except having made electrolytic solution contain 0.3 mass % of phenyldimethylfluorosilanes instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 3 of the positive electrode [3].
正极[3]实施例8Positive electrode [3] embodiment 8
使电解液含有0.3质量%的二氟磷酸锂来代替六甲基环三硅氧烷,除此以外,与正极[3]实施例5同样实施。电池评价的结果示于正极[3]表3。The procedure was carried out in the same manner as in positive electrode [3] Example 5, except that the electrolytic solution contained 0.3% by mass of lithium difluorophosphate instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 3 of the positive electrode [3].
正极[3]比较例2Positive electrode [3] Comparative example 2
除了电解液中不含六甲基环三硅氧烷以外,与正极[3]实施例5同样实施。电池评价的结果示于正极[3]表3。Except that hexamethylcyclotrisiloxane is not contained in the electrolytic solution, it is carried out in the same manner as the positive electrode [3] Example 5. The results of the battery evaluation are shown in Table 3 of the positive electrode [3].
正极[3]表3Positive pole [3] Table 3
[表15][Table 15]
正极[3]实施例9Positive electrode [3] embodiment 9
除了使用正极活性物质C作为正极活性物质,制成正极以外,与正极[3]实施例1同样实施。电池评价的结果示于正极[3]表4。Except using the positive electrode active material C as the positive electrode active material to prepare a positive electrode, it was carried out in the same manner as the positive electrode [3] Example 1. The results of the battery evaluation are shown in Table 4 of the positive electrode [3].
正极[3]实施例10Positive electrode [3] Example 10
使电解液含有0.3质量%的甲磺酸三甲基甲硅烷基酯来代替六甲基环三硅氧烷,除此以外,与正极[3]实施例9同样实施。电池评价的结果示于正极[3]表4。The procedure was carried out in the same manner as in positive electrode [3] Example 9 except that the electrolytic solution contained 0.3% by mass of trimethylsilyl methanesulfonate instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 4 of the positive electrode [3].
正极[3]实施例11Positive electrode [3] Example 11
使电解液含有0.3质量%的苯基二甲基氟硅烷来代替六甲基环三硅氧烷,除此以外,与正极[3]实施例9同样实施。电池评价的结果示于正极[3]表4。It carried out similarly to positive electrode [3] Example 9 except having made electrolytic solution contain 0.3 mass % of phenyldimethylfluorosilanes instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 4 of the positive electrode [3].
正极[3]实施例12Positive electrode [3] Example 12
使电解液含有0.3质量%的二氟磷酸锂来代替六甲基环三硅氧烷,除此以外,与正极[3]实施例9同样实施。电池评价的结果示于正极[3]表4。The procedure was carried out in the same manner as in positive electrode [3] Example 9 except that the electrolytic solution contained 0.3% by mass of lithium difluorophosphate instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 4 of the positive electrode [3].
正极[3]比较例3Positive electrode [3] Comparative example 3
除了电解液中不含六甲基环三硅氧烷以外,与正极[3]实施例9同样实施。电池评价的结果示于正极[3]表4。Except that the electrolytic solution does not contain hexamethylcyclotrisiloxane, the same implementation is carried out as in the positive electrode [3] Example 9. The results of the battery evaluation are shown in Table 4 of the positive electrode [3].
正极[3]表4Positive pole [3] Table 4
[表16][Table 16]
正极[3]实施例13Positive electrode [3] Example 13
使用正极活性物质A和正极活性物质D以2∶1的质量比充分混合而得到的正极活性物质作为正极活性物质,制成正极,用压制机压延成厚度为74μm,并使用32片该正极和33片负极,除此之外,与正极[3]实施例1同样实施。(正极一面的正极活性物质层厚度/正极集电体的厚度)为2.0,正极的电极面积的总和与电池的外壳表面积总和之比为20.6。该混合的正极活性物质的BET比表面积为1.2m2/g,平均一次粒径为0.6μm,中值粒径d50为6.7μm,振实密度为2.2g/cm2。电池评价的结果示于正极[3]表5。Use positive active material A and positive active material D to fully mix the positive active material that obtains with the mass ratio of 2: 1 as positive active material, make positive pole, be rolled into thickness with pressing machine and be 74 μ m, and use 32 this positive pole and Except for 33 negative electrodes, the same implementation was carried out as in Example 1 of the positive electrode [3]. (The thickness of the positive electrode active material layer on the positive electrode side/the thickness of the positive electrode current collector) was 2.0, and the ratio of the total electrode area of the positive electrode to the total surface area of the battery case was 20.6. The BET specific surface area of the mixed positive electrode active material was 1.2 m 2 /g, the average primary particle size was 0.6 μm, the median particle size d 50 was 6.7 μm, and the tap density was 2.2 g/cm 2 . The results of the battery evaluation are shown in Table 5 of the positive electrode [3].
正极[3]实施例14Positive electrode [3] Example 14
使电解液含有0.3质量%的甲磺酸三甲基甲硅烷基酯来代替六甲基环三硅氧烷,除此以外,与正极[3]实施例13同样实施。电池评价的结果示于正极[3]表5。It carried out similarly to positive electrode [3] Example 13 except having made electrolytic solution contain 0.3 mass % of trimethylsilyl methanesulfonate instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 5 of the positive electrode [3].
正极[3]实施例15Positive electrode [3] Example 15
使电解液含有0.3质量%的苯基二甲基氟硅烷来代替六甲基环三硅氧烷,除此以外,与正极[3]实施例13同样实施。电池评价的结果示于正极[3]表5。It carried out similarly to positive electrode [3] Example 13 except having made electrolytic solution contain 0.3 mass % of phenyldimethylfluorosilanes instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 5 of the positive electrode [3].
正极[3]实施例16Positive electrode [3] Example 16
使电解液含有0.3质量%的二氟磷酸锂来代替六甲基环三硅氧烷,除此以外,与正极[3]实施例13同样实施。电池评价的结果示于正极[3]表5。The procedure was carried out in the same manner as in positive electrode [3] Example 13 except that the electrolytic solution contained 0.3% by mass of lithium difluorophosphate instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 5 of the positive electrode [3].
正极[3]比较例4Positive electrode [3] Comparative example 4
除了电解液中不含六甲基环三硅氧烷以外,与正极[3]实施例13同样实施。电池评价的结果示于正极[3]表5。Except that hexamethylcyclotrisiloxane is not contained in the electrolytic solution, it is carried out in the same manner as the positive electrode [3] Example 13. The results of the battery evaluation are shown in Table 5 of the positive electrode [3].
正极[3]表5Positive pole [3] Table 5
[表17][Table 17]
由正极[3]表2~正极[3]表5的结果可知,在任一个正极中,由于在电解液中都含有特定化合物,因此,输出功率、容量保持率得到提高,并且即使在循环试验后,也可以充分保持电池容量和输出功率。From the results in Table 2 of positive electrode [3] to Table 5 of positive electrode [3], it can be seen that in any positive electrode, since specific compounds are contained in the electrolyte, the output power and capacity retention rate are improved, and even after the cycle test , can also fully maintain the battery capacity and output power.
正极[4][正极活性物质]Positive electrode [4] [positive electrode active material]
下面的实施例和比较例中使用的正极活性物质的种类和物性如下。The types and physical properties of positive electrode active materials used in the following Examples and Comparative Examples are as follows.
正极[4]表1Positive pole [4] Table 1
[表18]
正极[4]表1中,作为正极活性物质的物性,按照上述记载的方法,进行BET比表面积、平均一次粒径(用SEM测定)、中值粒径d50、振实密度的测定。Positive electrode [4] In Table 1, as the physical properties of the positive electrode active material, BET specific surface area, average primary particle size (measured by SEM), median particle size d 50 , and tap density were measured according to the method described above.
[正极活性物质A][Positive electrode active material A]
正极活性物质A是通过下面所示的方法合成的锂钴复合氧化物,用组成式LiCoO2表示。以Li∶Co=1∶1的摩尔比称量作为锂原料的LiOH和作为钴原料的Co(OH)2,并向其中加入纯水制成浆料,边搅拌边使用循环式介质搅拌型湿式珠磨机将浆料中的固体成分湿式粉碎成中值粒径0.2μm。The positive electrode active material A is a lithium-cobalt composite oxide synthesized by the method shown below and represented by the composition formula LiCoO 2 . Weigh LiOH as a lithium raw material and Co(OH) 2 as a cobalt raw material at a molar ratio of Li:Co=1:1, add pure water to them to make a slurry, and use a circulating medium stirring type wet type while stirring. The bead mill wet-milled the solid content in the slurry to a median particle size of 0.2 μm.
通过喷雾干燥器将浆料进行喷雾干燥,得到仅仅包含锂原料、钴原料的粒径约9μm的大致球状的造粒粒子。在空气流通下、880℃对该造粒粒子烧结6小时(升降温速度为5℃/分钟)。冷却到室温后,取出并粉碎,通过网眼45μm的筛子,得到正极活性物质A。The slurry was spray-dried by a spray dryer to obtain approximately spherical granulated particles having a particle diameter of about 9 μm and containing only the lithium raw material and the cobalt raw material. The granulated particles were sintered at 880° C. for 6 hours under air circulation (the heating and cooling rate was 5° C./minute). After cooling to room temperature, it was taken out and pulverized, and passed through a sieve with a mesh size of 45 μm to obtain a positive electrode active material A.
[正极活性物质B][Positive electrode active material B]
正极活性物质B是与正极活性物质A同样地合成的锂过渡金属复合氧化物,用组成式LiCoO2表示,其不同之处为:变更喷雾干燥的条件,制成粒径约1μm的造粒粒子,并且烧结温度为860℃。The positive electrode active material B is a lithium transition metal composite oxide synthesized in the same way as the positive electrode active material A, expressed by the composition formula LiCoO2 , the difference is that the spray drying conditions are changed to make granulated particles with a particle diameter of about 1 μm , and the sintering temperature is 860°C.
[正极活性物质C][Positive electrode active material C]
正极活性物质C是按照下面所示的方法合成的在正极活性物质A的表面附着了硫化合物和锑化合物而形成的正极活性物质。即,将96.7重量份正极活性物质A在流动槽中搅拌,同时向其中喷雾1.3重量份硫酸锂(Li2SO4H2O)的水溶液成喷雾状。在得到的混合物中添加2.0重量份三氧化锑(Sb2O3,粒子中值粒径为0.8μm),充分混合。将该混合物转移到氧化铝容器中,在空气氛围下于680℃烧结2小时,得到正极活性物质C。The positive electrode active material C is a positive electrode active material in which a sulfur compound and an antimony compound are attached to the surface of the positive electrode active material A, synthesized by the method shown below. That is, while stirring 96.7 parts by weight of the positive electrode active material A in the flow cell, 1.3 parts by weight of an aqueous solution of lithium sulfate (Li 2 SO 4 H 2 O) was sprayed thereinto in a spray form. 2.0 parts by weight of antimony trioxide (Sb 2 O 3 , particle median diameter: 0.8 μm) was added to the obtained mixture, and mixed well. The mixture was transferred to an alumina container, and sintered at 680° C. for 2 hours in an air atmosphere to obtain a positive electrode active material C.
[正极活性物质D][Positive electrode active material D]
正极活性物质D是通过下面所示的方法合成的锂过渡金属复合氧化物,用组成式Li1.04Mn1.84Al0.12O4表示。以Li∶Mn∶Al=1.04∶1.84∶0.12的摩尔比称量作为锂原料的LiOH、作为锰原料的Mn2O3和作为铝原料的AlOOH,并向其中加入纯水制成浆料,边搅拌边使用循环式介质搅拌型湿式珠磨机将浆料中的固体成分湿式粉碎成中值粒径0.5μm。The positive electrode active material D is a lithium transition metal composite oxide synthesized by the method shown below, and is represented by the composition formula Li 1.04 Mn 1.84 Al 0.12 O 4 . LiOH as a lithium raw material, Mn2O3 as a manganese raw material, and AlOOH as an aluminum raw material were weighed at a molar ratio of Li:Mn:Al=1.04:1.84:0.12 , and pure water was added thereto to make a slurry, while While stirring, the solid content in the slurry was wet-milled to a median particle size of 0.5 μm using a circulating medium-stirred wet bead mill.
通过喷雾干燥器将浆料进行喷雾干燥,得到仅仅包含锂原料、锰原料、铝原料的粒径约10μm的大致球状的造粒粒子。将该造粒粒子在氮气流通、900℃下烧结3小时(升温速度为5℃/分钟)后,将流通气体由氮气变换成空气,再在900℃下烧结2小时(降温速度为1℃/分钟)。冷却到室温后,取出并粉碎,通过网眼45μm的筛子,得到正极活性物质D。The slurry was spray-dried by a spray dryer to obtain approximately spherical granulated particles having a particle diameter of about 10 μm and containing only the lithium raw material, the manganese raw material, and the aluminum raw material. The granulated particles were sintered at 900° C. for 3 hours in nitrogen flow (heating rate was 5° C./min), then the flow gas was changed from nitrogen to air, and then sintered at 900° C. for 2 hours (temperature drop rate was 1° C./min. minute). After cooling to room temperature, it was taken out and pulverized, and passed through a sieve with a mesh size of 45 μm to obtain a positive electrode active material D.
正极[4]实施例1Positive pole [4] embodiment 1
《正极的制作》"Positive Production"
在N甲基吡咯烷酮溶剂中混合85质量%的作为正极活性物质的正极活性物质A、10质量%的作为导电材料的乙炔黑和5质量%作为粘合剂的聚偏氟乙烯(PVdF),制成浆料。将得到的浆料涂布在厚度15μm的铝箔两面,并干燥,用压制机压延成厚度为85μm,再将其切成具有宽100mm、长100mm大小的正极活性物质层和宽30mm的未涂布部分的形状,制成正极。正极活性物质的密度为2.35g/cm3,(一面的正极活性物质层的厚度)/(集电体的厚度)的值为2.3。Mix 85% by mass of positive electrode active material A as a positive electrode active material, 10% by mass of acetylene black as a conductive material, and 5% by mass of polyvinylidene fluoride (PVdF) as a binder in N-methylpyrrolidone solvent to prepare into slurry. The obtained slurry is coated on both sides of an aluminum foil with a thickness of 15 μm, dried, rolled into a thickness of 85 μm with a press, and then cut into a positive electrode active material layer with a width of 100 mm and a length of 100 mm and an uncoated layer with a width of 30 mm. The shape of the part is made into a positive electrode. The density of the positive electrode active material was 2.35 g/cm 3 , and the value of (thickness of positive electrode active material layer on one side)/(thickness of current collector) was 2.3.
《负极的制作》"The Making of Negative Pole"
在98重量份人造石墨粉末KS-44(timcal公司制造,商品名)中添加100重量份作为增稠剂的羧甲基纤维素钠的水性分散液(羧甲基纤维素钠的浓度为1质量%),2重量份作为粘合剂的苯乙烯-丁二烯橡胶的水性分散液(苯乙烯-丁二烯橡胶的浓度为50质量%),用分散器进行混合,制成浆料。将得到的浆料涂布在厚度10μm的铜箔两面,并干燥,用压制机压延成厚度为75μm,再将其切成具有宽104mm、长104mm大小的负极活性物质层和宽30mm的未涂布部分的形状,作为负极。In 98 parts by weight of artificial graphite powder KS-44 (manufactured by timcal company, trade name), add 100 parts by weight of an aqueous dispersion of sodium carboxymethyl cellulose as a thickener (the concentration of sodium carboxymethyl cellulose is 1 mass %), 2 parts by weight of an aqueous dispersion of styrene-butadiene rubber as a binder (the concentration of styrene-butadiene rubber is 50% by mass), and mixed with a disperser to prepare a slurry. The obtained slurry is coated on both sides of a copper foil with a thickness of 10 μm, dried, rolled into a thickness of 75 μm with a press machine, and then cut into a negative electrode active material layer with a width of 104 mm and a length of 104 mm and an uncoated layer with a width of 30 mm. The shape of the cloth part is used as the negative electrode.
《非水电解液的制作》"Preparation of Non-Aqueous Electrolyte"
在干燥的氩氛围下,在纯化的碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的3∶3∶4(体积比)的混合溶剂中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。另外,含有0.3质量%的六甲基环三硅氧烷。Under a dry argon atmosphere, in a 3:3:4 (volume ratio) mixed solvent of purified ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC), with 1mol /L concentration dissolves well-dried lithium hexafluorophosphate (LiPF 6 ). In addition, 0.3% by mass of hexamethylcyclotrisiloxane was contained.
《电池的制作》"Battery Making"
将31片正极和32片负极交替配置,各电极之间夹入多孔聚乙烯片隔板(厚度为25μm),进行叠层。此时,使正极活性物质面与负极活性物质面相对,并且不会露出负极活性物质面之外。将该正极和负极各自的未涂布部分焊接,制作集电片,将电极组密封在电池罐(外部尺寸:120×110×10mm)中。然后,在装有电极组的电池罐中注入20mL非水电解液,使电极充分浸透,密封而制作电池。正极的电极面积总和与电池的外壳表面积总和之比为20.0。31 positive electrodes and 32 negative electrodes were alternately arranged, and a porous polyethylene sheet separator (thickness: 25 μm) was sandwiched between the electrodes for lamination. At this time, the surface of the positive electrode active material faces the surface of the negative electrode active material without protruding from the surface of the negative electrode active material. The respective uncoated parts of the positive and negative electrodes were welded to produce current collector sheets, and the electrode group was sealed in a battery can (outer dimensions: 120×110×10 mm). Then, 20 mL of non-aqueous electrolytic solution was poured into the battery can containing the electrode group, the electrodes were sufficiently permeated, and the battery was sealed to produce a battery. The ratio of the sum of the electrode areas of the positive electrode to the sum of the surface areas of the case of the battery was 20.0.
《电池的评价》"Battery Evaluation"
(电池容量的测定方法)(Measuring method of battery capacity)
对未经充放电循环的新电池,在25℃、4.2V~3.0V的电压范围下,以0.2C的电流值(将1小时放电额定容量的电流值作为1C,所述额定容量取决于1小时率(one-hour-rate)的放电容量,下同)进行5个循环的初期充放电。此时的第5个循环的0.2C放电容量作为初期容量。电池评价的结果示于正极[4]表2。For a new battery that has not been charged and discharged, at 25°C and a voltage range of 4.2V to 3.0V, with a current value of 0.2C (the current value of the rated capacity of 1 hour discharge is taken as 1C, and the rated capacity depends on 1 One-hour-rate (one-hour-rate) discharge capacity, the same below) was performed for 5 cycles of initial charge and discharge. The 0.2C discharge capacity at the fifth cycle at this time was taken as the initial capacity. The results of the battery evaluation are shown in Table 2 of the positive electrode [4].
(初期输出功率的测定方法)(Measurement method of initial output power)
在25℃的环境下,通过0.2C的恒定电流进行150分钟充电,分别以0.1C、0.3C、1.0C、3.0C、10.0C放电10秒钟,测定第10秒的电压。将电流-电压直线和下限电压(3V)所包围的三角形的面积作为输出功率(W)。电池评价的结果示于正极[4]表2。In an environment of 25°C, charge was performed for 150 minutes with a constant current of 0.2C, and discharge was performed at 0.1C, 0.3C, 1.0C, 3.0C, and 10.0C for 10 seconds, respectively, and the voltage at the 10th second was measured. The area of the triangle surrounded by the current-voltage line and the lower limit voltage (3V) was taken as the output power (W). The results of the battery evaluation are shown in Table 2 of the positive electrode [4].
(循环试验(耐久后电池容量和耐久后输出功率的测定方法))(Cycle test (measuring method of battery capacity after endurance and output power after endurance))
在被视为锂二次电池的实际使用上限温度的60℃的高温环境下,进行循环试验。以2C恒定电流恒定电压法充电到充电上限电压4.2V后,以2C的恒定电流放电至放电终止电压3.0V,以此作为一个充放电循环,反复进行该循环,直至500循环。对于循环试验结束后的电池,在25℃的环境下以0.2C的电流值进行3个循环的充放电,以第3个循环的0.2C放电容量作为耐久后电池容量。另外,对于循环试验结束后的电池,进行输出功率测定,作为耐久后输出功率。电池评价的结果示于正极[4]表2。The cycle test was performed in a high-temperature environment of 60° C., which is regarded as the upper limit temperature for practical use of lithium secondary batteries. After charging to the charging upper limit voltage of 4.2V by 2C constant current and constant voltage method, discharge to the end-of-discharge voltage of 3.0V with a constant current of 2C, which is regarded as a charge-discharge cycle, and this cycle is repeated until 500 cycles. For the battery after the cycle test, three cycles of charging and discharging were performed at a current value of 0.2C in an environment of 25°C, and the 0.2C discharge capacity of the third cycle was used as the battery capacity after endurance. In addition, for the battery after the cycle test, the output power was measured, and it was set as the output power after durability. The results of the battery evaluation are shown in Table 2 of the positive electrode [4].
正极[4]实施例2Positive pole [4] embodiment 2
使非水电解液含有0.3质量%的甲磺酸三甲基甲硅烷基酯来代替六甲基环三硅氧烷,除此以外,与正极[4]实施例1同样实施。电池评价的结果示于正极[4]表2。The non-aqueous electrolytic solution was carried out in the same manner as in Example 1 of positive electrode [4] except that 0.3% by mass of trimethylsilyl methanesulfonate was included instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 2 of the positive electrode [4].
正极[4]实施例3Positive electrode [4] Example 3
使非水电解液含有0.3质量%的苯基二甲基氟硅烷来代替六甲基环三硅氧烷,除此以外,与正极[4]实施例1同样实施。电池评价的结果示于正极[4]表2。The non-aqueous electrolytic solution was carried out in the same manner as the positive electrode [4] Example 1 except that 0.3% by mass of phenyldimethylfluorosilane was contained instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 2 of the positive electrode [4].
正极[4]实施例4Positive electrode [4] embodiment 4
使非水电解液含有0.3质量%的二氟磷酸锂来代替六甲基环三硅氧烷,除此以外,与正极[4]实施例1同样实施。电池评价的结果示于正极[4]表2。The non-aqueous electrolytic solution was made to contain 0.3 mass % of lithium difluorophosphate instead of hexamethylcyclotrisiloxane, and it carried out similarly to positive electrode [4] Example 1. The results of the battery evaluation are shown in Table 2 of the positive electrode [4].
正极[4]比较例1Positive electrode [4] Comparative example 1
除了非水电解液中不含六甲基环三硅氧烷以外,与正极[4]实施例1同样实施。电池评价的结果示于正极[4]表2。Except that hexamethylcyclotrisiloxane is not contained in the non-aqueous electrolyte solution, it is carried out in the same manner as the positive electrode [4] Example 1. The results of the battery evaluation are shown in Table 2 of the positive electrode [4].
正极[4]表2Positive electrode [4] Table 2
[表19][Table 19]
正极[4]实施例5Positive electrode [4] embodiment 5
除了使用正极活性物质B作为正极活性物质,制成正极以外,与正极[4]实施例1同样实施。电池评价的结果示于正极[4]表3。Except for using the positive electrode active material B as the positive electrode active material to prepare a positive electrode, the same implementation was carried out as in the positive electrode [4] Example 1. The results of the battery evaluation are shown in Table 3 of the positive electrode [4].
正极[4]实施例6Positive electrode [4] embodiment 6
使非水电解液含有0.3质量%的甲磺酸三甲基甲硅烷基酯来代替六甲基环三硅氧烷,除此以外,与正极[4]实施例5同样实施。电池评价的结果示于正极[4]表3。The nonaqueous electrolytic solution was made to contain 0.3 mass % of trimethylsilyl methanesulfonate instead of hexamethylcyclotrisiloxane, and it carried out similarly to positive electrode [4] Example 5. The results of the battery evaluation are shown in Table 3 of the positive electrode [4].
正极[4]实施例7Positive electrode [4] embodiment 7
使非水电解液含有0.3质量%的苯基二甲基氟硅烷来代替六甲基环三硅氧烷,除此以外,与正极[4]实施例5同样实施。电池评价的结果示于正极[4]表3。The non-aqueous electrolytic solution was made to contain 0.3 mass % of phenyldimethylfluorosilane instead of hexamethylcyclotrisiloxane, and it carried out similarly to positive electrode [4] Example 5. The results of the battery evaluation are shown in Table 3 of the positive electrode [4].
正极[4]实施例8Positive electrode [4] embodiment 8
使非水电解液含有0.3质量%的二氟磷酸锂来代替六甲基环三硅氧烷,除此以外,与正极[4]实施例5同样实施。电池评价的结果示于正极[4]表3。The non-aqueous electrolytic solution was made to contain 0.3 mass % of lithium difluorophosphate instead of hexamethylcyclotrisiloxane, and it carried out similarly to positive electrode [4] Example 5. The results of the battery evaluation are shown in Table 3 of the positive electrode [4].
正极[4]比较例2Positive electrode [4] Comparative example 2
除了非水电解液中不含六甲基环三硅氧烷以外,与正极[4]实施例5同样实施。电池评价的结果示于正极[4]表3。Except that hexamethylcyclotrisiloxane is not contained in the non-aqueous electrolyte solution, it is carried out in the same manner as the positive electrode [4] Example 5. The results of the battery evaluation are shown in Table 3 of the positive electrode [4].
正极[4]表3Positive electrode [4] Table 3
[表20][Table 20]
正极[4]实施例9Positive electrode [4] embodiment 9
使用正极活性物质A和正极活性物质B以2∶1的质量比充分混合而得到的正极活性物质作为正极活性物质,制成正极,除此以外,与正极[4]实施例1同样实施。该混合的正极活性物质的BET比表面积为1.8m2/g,平均一次粒径为0.2μm,中值粒径d50为4.9μm,振实密度为1.8g/cm3。电池评价结果示于正极[4]表4。The positive electrode active material obtained by fully mixing the positive electrode active material A and the positive electrode active material B at a mass ratio of 2:1 was used as the positive electrode active material to make a positive electrode, except that it was carried out in the same manner as the positive electrode [4] Example 1. The mixed positive electrode active material had a BET specific surface area of 1.8 m 2 /g, an average primary particle size of 0.2 μm, a median particle size d 50 of 4.9 μm, and a tap density of 1.8 g/cm 3 . The battery evaluation results are shown in Table 4 of the positive electrode [4].
正极[4]实施例10Positive electrode [4] Example 10
使非水电解液含有0.3质量%的甲磺酸三甲基甲硅烷基酯来代替六甲基环三硅氧烷,除此以外,与正极[4]实施例9同样实施。电池评价的结果示于正极[4]表4。The non-aqueous electrolytic solution was carried out in the same manner as in positive electrode [4] Example 9 except that 0.3% by mass of trimethylsilyl methanesulfonate was included instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 4 of the positive electrode [4].
正极[4]实施例11Positive electrode [4] Example 11
使非水电解液含有0.3质量%的苯基二甲基氟硅烷来代替六甲基环三硅氧烷,除此以外,与正极[4]实施例9同样实施。电池评价的结果示于正极[4]表4。The non-aqueous electrolytic solution was made to contain 0.3 mass % of phenyldimethylfluorosilane instead of hexamethylcyclotrisiloxane, and it carried out similarly to positive electrode [4] Example 9. The results of the battery evaluation are shown in Table 4 of the positive electrode [4].
正极[4]实施例12Positive electrode [4] Example 12
使非水电解液含有0.3质量%的二氟磷酸锂来代替六甲基环三硅氧烷,除此以外,与正极[4]实施例9同样实施。电池评价的结果示于正极[4]表4。The non-aqueous electrolytic solution was made to contain 0.3 mass % of lithium difluorophosphate instead of hexamethylcyclotrisiloxane, and it carried out similarly to positive electrode [4] Example 9. The results of the battery evaluation are shown in Table 4 of the positive electrode [4].
正极[4]比较例3Positive electrode [4] Comparative Example 3
除了非水电解液中不含六甲基环三硅氧烷以外,与正极[4]实施例9同样实施。电池评价的结果示于正极[4]表4。Except that hexamethylcyclotrisiloxane is not contained in the non-aqueous electrolyte solution, it is carried out in the same manner as the positive electrode [4] Example 9. The results of the battery evaluation are shown in Table 4 of the positive electrode [4].
正极[4]表4Positive electrode [4] Table 4
[表21][Table 21]
正极[4]实施例13Positive electrode [4] Example 13
除了使用正极活性物质C作为正极活性物质,制成正极以外,与正极[4]实施例1同样实施。电池评价的结果示于正极[4]表5。Except for using the positive electrode active material C as the positive electrode active material to prepare a positive electrode, the same implementation was carried out as in the positive electrode [4] Example 1. The results of the battery evaluation are shown in Table 5 of the positive electrode [4].
正极[4]实施例14Positive electrode [4] Example 14
使非水电解液含有0.3质量%的甲磺酸三甲基甲硅烷基酯来代替六甲基环三硅氧烷,除此以外,与正极[4]实施例13同样实施。电池评价的结果示于正极[4]表5。The non-aqueous electrolytic solution was carried out in the same manner as in positive electrode [4] Example 13 except that 0.3% by mass of trimethylsilyl methanesulfonate was included instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 5 of the positive electrode [4].
正极[4]实施例15Positive electrode [4] Example 15
使非水电解液含有0.3质量%的苯基二甲基氟硅烷来代替六甲基环三硅氧烷,除此以外,与正极[4]实施例13同样实施。电池评价的结果示于正极[4]表5。The same procedure was carried out as in Example 13 of the positive electrode [4], except that the nonaqueous electrolyte contained 0.3% by mass of phenyldimethylfluorosilane instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 5 of the positive electrode [4].
正极[4]实施例16Positive electrode [4] Example 16
使非水电解液含有0.3质量%的二氟磷酸锂来代替六甲基环三硅氧烷,除此以外,与正极[4]实施例13同样实施。电池评价的结果示于正极[4]表5。The same procedure as in Example 13 of the positive electrode [4] was carried out except that the nonaqueous electrolytic solution contained 0.3% by mass of lithium difluorophosphate instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 5 of the positive electrode [4].
正极[4]比较例4Positive electrode [4] Comparative example 4
除了非水电解液中不含六甲基环三硅氧烷以外,与正极[4]实施例13同样实施。电池评价的结果示于正极[4]表5。Except not containing hexamethylcyclotrisiloxane in the non-aqueous electrolytic solution, the same implementation was carried out as in the positive electrode [4] Example 13. The results of the battery evaluation are shown in Table 5 of the positive electrode [4].
正极[4]表5Positive electrode [4] Table 5
[表22][Table 22]
正极[4]实施例17Positive electrode [4] Example 17
使用正极活性物质A和正极活性物质D以2∶1的质量比充分混合而得到的正极活性物质作为正极活性物质,制成正极,用压制机压延成厚度92μm,并使用30片该正极和31片负极,除此以外,与正极[4]实施例1同样实施。(正极一面的正极活性物质层厚度)/(正极集电体的厚度)为2.6,正极的电极面积总和与电池的外壳表面积总和之比为19.4。另外,该混合的正极活性物质的BET比表面积为1.1m2/g,平均一次粒径为0.6μm,中值粒径d50为7.3μm,振实密度为2.2g/cm3。电池评价结果示于正极[4]表6。Use positive active material A and positive active material D to fully mix the positive active material that obtains with the mass ratio of 2: 1 as positive active material, make positive pole, be rolled into thickness 92 μ m with pressing machine, and use 30 this positive pole and 31 The sheet negative electrode was carried out in the same manner as in Example 1 of the positive electrode [4] except for this. (Thickness of the positive electrode active material layer on the positive electrode side)/(Thickness of the positive electrode current collector) was 2.6, and the ratio of the total electrode area of the positive electrode to the total surface area of the battery case was 19.4. In addition, the mixed positive electrode active material had a BET specific surface area of 1.1 m 2 /g, an average primary particle size of 0.6 μm, a median particle size d 50 of 7.3 μm, and a tap density of 2.2 g/cm 3 . The battery evaluation results are shown in Table 6 of the positive electrode [4].
正极[4]实施例18Positive electrode [4] Example 18
使非水电解液含有0.3质量%的甲磺酸三甲基甲硅烷基酯来代替六甲基环三硅氧烷,除此以外,与正极[4]实施例17同样实施。电池评价的结果示于正极[4]表6。The non-aqueous electrolytic solution was carried out in the same manner as in positive electrode [4] Example 17, except that 0.3% by mass of trimethylsilyl methanesulfonate was included instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 6 of the positive electrode [4].
正极[4]实施例19Positive electrode [4] Example 19
使非水电解液含有0.3质量%的苯基二甲基氟硅烷来代替六甲基环三硅氧烷,除此以外,与正极[4]实施例17同样实施。电池评价的结果示于正极[4]表6。The non-aqueous electrolytic solution was made to contain 0.3 mass % of phenyldimethylfluorosilane instead of hexamethylcyclotrisiloxane, and it carried out similarly to positive electrode [4] Example 17. The results of the battery evaluation are shown in Table 6 of the positive electrode [4].
正极[4]实施例20Positive electrode [4] Example 20
使非水电解液含有0.3质量%的二氟磷酸锂来代替六甲基环三硅氧烷,除此以外,与正极[4]实施例17同样实施。电池评价的结果示于正极[4]表6。The same procedure as in Example 17 of the positive electrode [4] was carried out except that the nonaqueous electrolytic solution contained 0.3% by mass of lithium difluorophosphate instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 6 of the positive electrode [4].
正极[4]比较例5Positive electrode [4] Comparative Example 5
除了非水电解液中不含六甲基环三硅氧烷以外,与正极[4]实施例17同样实施。电池评价的结果示于正极[4]表6。Except that hexamethylcyclotrisiloxane is not contained in the non-aqueous electrolytic solution, the same implementation is carried out as in the positive electrode [4] Example 17. The results of the battery evaluation are shown in Table 6 of the positive electrode [4].
正极[4]表6Positive pole [4] Table 6
[表23][Table 23]
从正极[4]表2~正极[4]表6的结果可知,在任一个正极中,由于在非水电解液中都含有特定化合物,初期输出功率得到提高。另外,容量保持率得到提高,即使在循环试验后,也可以充分保持电池容量和输出功率。From the results in Table 2 of the positive electrode [4] to Table 6 of the positive electrode [4], it can be seen that in any positive electrode, the initial output power is improved due to the specific compound contained in the non-aqueous electrolyte. In addition, the capacity retention rate is improved, and the battery capacity and output power can be fully maintained even after a cycle test.
正极[5][正极活性物质]Positive electrode [5] [positive electrode active material]
下面的实施例和比较例中使用的正极活性物质的种类和物性如下。The types and physical properties of positive electrode active materials used in the following Examples and Comparative Examples are as follows.
正极[5]表1Positive electrode [5] Table 1
[表24][Table 24]
[2368]正极[5]表1中,作为正极活性物质的物性,按照上述记载的方法,进行BET比表面积、平均一次粒径(用SEM测定)、中值粒径d50、振实密度的测定。In positive pole [5] table 1, as the physical property of positive pole active material, according to the method described above, carry out BET specific surface area, average primary particle diameter (measure with SEM), median diameter d 50 , tap density Determination.
[正极活性物质A][Positive electrode active material A]
正极活性物质A是通过下面所示的方法合成的锂钴复合氧化物,用组成式LiCoO2表示。以Li∶Co=1∶1的摩尔比称量作为锂原料的LiOH和作为钴原料的Co(OH)2,并向其中加入纯水制成浆料,边搅拌边使用循环式介质搅拌型湿式珠磨机将浆料中的固体成分湿式粉碎成中值粒径0.2μm。The positive electrode active material A is a lithium-cobalt composite oxide synthesized by the method shown below and represented by the composition formula LiCoO 2 . Weigh LiOH as a lithium raw material and Co(OH) 2 as a cobalt raw material at a molar ratio of Li:Co=1:1, add pure water to them to make a slurry, and use a circulating medium stirring type wet type while stirring. The bead mill wet-milled the solid content in the slurry to a median particle size of 0.2 μm.
通过喷雾干燥器将浆料进行喷雾干燥,得到仅仅包含锂原料、钴原料的粒径约9μm的大致球状的造粒粒子。在空气流通下、880℃对该造粒粒子烧结6小时(升降温速度为5℃/分钟)。冷却到室温后,取出并粉碎,通过网眼45μm的筛子,得到正极活性物质A。The slurry was spray-dried by a spray dryer to obtain approximately spherical granulated particles having a particle diameter of about 9 μm and containing only the lithium raw material and the cobalt raw material. The granulated particles were sintered at 880° C. for 6 hours under air circulation (the heating and cooling rate was 5° C./minute). After cooling to room temperature, it was taken out and pulverized, and passed through a sieve with a mesh size of 45 μm to obtain a positive electrode active material A.
[正极活性物质B][Positive electrode active material B]
正极活性物质B是通过以下方法合成的锂过渡金属复合氧化物,以组成式Li1.05Ni0.80Co0.2O2表示。以Ni∶Co=80∶20的摩尔比称量作为镍原料的NiO,作为钴原料的Co(OH)2,并向其中加入纯水制成浆料,边搅拌边使用循环式介质搅拌型湿式珠磨机将浆料中的固体成分湿式粉碎成中值粒径0.25μm。The positive electrode active material B is a lithium transition metal composite oxide synthesized by the following method, represented by the composition formula Li 1.05 Ni 0.80 Co 0.2 O 2 . Weigh NiO as a nickel raw material and Co(OH) 2 as a cobalt raw material at a molar ratio of Ni:Co=80:20, and add pure water to them to make a slurry, and use a circulating medium stirring type wet type while stirring. The bead mill wet-milled the solid content in the slurry to a median particle size of 0.25 μm.
通过喷雾干燥器将浆料进行喷雾干燥,得到仅仅包含镍原料、钴原料的粒径约为8μm的大致球状的造粒粒子。在得到的造粒粒子中添加中值粒径为3μm的LiOH粉末,使得Li的摩尔数与Ni和Co的总摩尔数之比为1.05,用高速混合机进行混合,得到镍原料、钴原料的造粒粒子和锂原料的混合粉末。该混合粉末在氧流通下于740℃下烧结(升降温速度为5℃/分钟)6小时,然后进行粉碎,通过网眼45μm的筛子,得到正极活性物质B。The slurry was spray-dried by a spray dryer to obtain approximately spherical granulated particles having a particle diameter of about 8 μm and containing only the nickel raw material and the cobalt raw material. LiOH powder with a median diameter of 3 μm was added to the obtained granulated particles so that the ratio of the number of moles of Li to the total number of moles of Ni and Co was 1.05, and mixed with a high-speed mixer to obtain nickel raw materials and cobalt raw materials. Mixed powder of granulated particles and lithium raw material. The mixed powder was sintered at 740° C. for 6 hours under oxygen flow (the heating and cooling rate was 5° C./min), then pulverized, and passed through a sieve with a mesh size of 45 μm to obtain a positive electrode active material B.
[正极活性物质C][Positive electrode active material C]
正极活性物质C是通过以下方法合成的锂过渡金属复合氧化物,以组成式Li1.05Ni0.80Co0.15Al0.05O2表示。以Ni∶Co∶Al=80∶15∶5的摩尔比称量作为镍原料的NiO,作为钴原料的Co(OH)2以及作为铝原料的AlOOH,向其中加入纯水制成浆料,边搅拌边使用循环式介质搅拌型湿式珠磨机将浆料中的固体成分湿式粉碎成中值粒径0.25μm。The positive electrode active material C is a lithium transition metal composite oxide synthesized by the following method, represented by the composition formula Li 1.05 Ni 0.80 Co 0.15 Al 0.05 O 2 . NiO as a nickel raw material, Co(OH) as a cobalt raw material, and AlOOH as an aluminum raw material were weighed at a molar ratio of Ni:Co:Al=80:15:5, and pure water was added thereto to make a slurry, while While stirring, the solid content in the slurry was wet-milled to a median particle size of 0.25 μm using a circulating medium-stirred wet bead mill.
通过喷雾干燥器将浆料进行喷雾干燥,得到仅仅包含镍原料、钴原料、铝原料的粒径约10μm的大致球状的造粒粒子。在得到的造粒粒子中添加中值粒径3μm的LiOH粉末,使得Li的摩尔数相对于Ni、Co和Al的总摩尔数的比例为1.05,用高速混合机进行混合,得到镍原料、钴原料、铝原料的造粒粒子和锂原料的混合粉末。该混合粉末在氧流通下于740℃下烧结(升降温速度为5℃/分钟)6小时,然后进行粉碎,通过网眼45μm的筛子,得到正极活性物质C。The slurry was spray-dried with a spray dryer to obtain approximately spherical granulated particles having a particle diameter of about 10 μm and containing only nickel raw materials, cobalt raw materials, and aluminum raw materials. LiOH powder with a median diameter of 3 μm was added to the obtained granulated particles so that the ratio of the number of moles of Li to the total number of moles of Ni, Co, and Al was 1.05, and mixed with a high-speed mixer to obtain nickel raw materials, cobalt Raw materials, granulated particles of aluminum raw materials and mixed powder of lithium raw materials. The mixed powder was sintered at 740° C. for 6 hours under oxygen flow (the heating and cooling rate was 5° C./min), and then pulverized and passed through a sieve with a mesh size of 45 μm to obtain a positive electrode active material C.
[正极活性物质D][Positive electrode active material D]
正极活性物质C是按照下面所示的方法合成的在正极活性物质C的表面附着了硫化合物和锑化合物而形成的正极活性物质。即,将96.7重量份正极活性物质C在流动槽中搅拌,同时向其中喷雾1.3重量份硫酸锂(Li2SO4H2O)的水溶液成喷雾状。在得到的混合物中添加2.0重量份三氧化锑(Sb2O3,粒子中值粒径为0.8μm),充分混合。将该混合物转移到氧化铝容器中,在空气氛围下于680℃烧结2小时,得到正极活性物质D。The positive electrode active material C is a positive electrode active material in which a sulfur compound and an antimony compound are adhered to the surface of the positive electrode active material C by the method shown below. That is, while stirring 96.7 parts by weight of the positive electrode active material C in the flow cell, 1.3 parts by weight of an aqueous solution of lithium sulfate (Li 2 SO 4 H 2 O) was sprayed thereinto in a spray form. 2.0 parts by weight of antimony trioxide (Sb 2 O 3 , particle median diameter: 0.8 μm) was added to the obtained mixture, and mixed well. The mixture was transferred to an alumina container, and sintered at 680° C. for 2 hours in an air atmosphere to obtain a positive electrode active material D.
[正极活性物质E][Positive electrode active material E]
正极活性物质E是通过以下方法合成的锂过渡金属复合氧化物,以组成式LiMn0.33Ni0.33Co0.33O2表示。以Mn∶Ni∶Co=1∶1∶1的摩尔比称量作为锰原料的Mn3O4、作为镍原料的NiO以及作为钴原料的Co(OH)2,向其中加入纯水制成浆料,边搅拌边使用循环式介质搅拌型湿式珠磨机将浆料中的固体成分湿式粉碎成中值粒径0.2μm。The positive electrode active material E is a lithium transition metal composite oxide synthesized by the following method, represented by the composition formula LiMn 0.33 Ni 0.33 Co 0.33 O 2 . Weigh Mn 3 O 4 as a manganese raw material, NiO as a nickel raw material, and Co(OH) 2 as a cobalt raw material at a molar ratio of Mn:Ni:Co=1:1:1, and add pure water to make a slurry While stirring, the solid components in the slurry were wet pulverized into a median particle size of 0.2 μm using a circulating medium agitation wet bead mill.
通过喷雾干燥器将浆料进行喷雾干燥,得到仅仅包含锰原料、镍原料、钴原料的粒径约5μm的大致球状的造粒粒子。在得到的造粒粒子中添加中值粒径3μm的LiOH粉末,使得Li的摩尔数相对于Mn、Ni和Co的总摩尔数之比为1.05,用高速混合机进行混合,得到锰原料、镍原料、钴原料的造粒粒子和锂原料的混合粉末。该混合粉末在氧流通下于950℃下烧结(升降温速度为5℃/分钟)12小时,然后进行粉碎,通过网眼45μm的筛子,得到正极活性物质E。The slurry was spray-dried by a spray dryer to obtain roughly spherical granulated particles having a particle diameter of about 5 μm and containing only manganese raw materials, nickel raw materials, and cobalt raw materials. LiOH powder with a median diameter of 3 μm was added to the obtained granulated particles so that the ratio of the number of moles of Li to the total number of moles of Mn, Ni, and Co was 1.05, and mixed with a high-speed mixer to obtain manganese raw materials, nickel Raw material, granulated particles of cobalt raw material and mixed powder of lithium raw material. The mixed powder was sintered at 950° C. for 12 hours under oxygen flow (the heating and cooling rate was 5° C./min), and then pulverized and passed through a sieve with a mesh size of 45 μm to obtain a positive electrode active material E.
[正极活性物质F][Positive electrode active material F]
正极活性物质F是通过以下方法合成的锂过渡金属复合氧化物,以组成式Li1.04Mn1.84Al0.12O4表示。以Li∶Mn∶Al=1.04∶1.84∶0.12的摩尔比称量作为锂原料的LiOH、作为锰原料的Mn2O3和作为铝原料的AlOOH,向其中加入纯水制成浆料,边搅拌边使用循环式介质搅拌型湿式珠磨机将浆料中的固体成分湿式粉碎成中值粒径0.5μm。The positive electrode active material F is a lithium transition metal composite oxide synthesized by the following method, represented by the composition formula Li 1.04 Mn 1.84 Al 0.12 O 4 . Weigh LiOH as a lithium raw material, Mn2O3 as a manganese raw material, and AlOOH as an aluminum raw material at a molar ratio of Li:Mn:Al=1.04:1.84:0.12, add pure water to them to make a slurry, and stir The solid content in the slurry was wet pulverized to a median particle size of 0.5 μm while using a circulating medium agitation type wet bead mill.
通过喷雾干燥器将浆料进行喷雾干燥,得到仅仅包含锂原料、锰原料和铝原料的粒径约10μm的大致球状的造粒粒子。将得到的造粒粒子在氮气流通下于900℃下烧结(升降温速度为5℃/分钟)3小时,然后,将流通气体由氮气变换成空气,再在900℃下烧结(降温速度为1℃/分钟)2小时。冷却到室温后,取出并粉碎,通过网眼45μm的筛子,得到正极活性物质F。The slurry was spray-dried by a spray dryer to obtain approximately spherical granulated particles having a particle diameter of about 10 μm and containing only the lithium raw material, the manganese raw material and the aluminum raw material. The granulated particles obtained were sintered at 900°C under nitrogen flow (the rate of temperature rise and fall was 5°C/min) for 3 hours, then the flow gas was changed from nitrogen to air, and then sintered at 900°C (the rate of temperature drop was 1°C). °C/min) for 2 hours. After cooling to room temperature, it was taken out and pulverized, and passed through a sieve with a mesh size of 45 μm to obtain a positive electrode active material F.
正极[5]实施例1Positive pole [5] embodiment 1
《正极的制作》"Positive Production"
将正极活性物质A和负极活性物质B以1∶1的质量比充分混合,使用得到的正极活性物质制作正极。该混合后的正极活性物质的BET比表面积为1.2m2/g,平均一次粒径为0.8μm,中值粒径d50为6.5μm,振实密度为2.1g/cm3。The positive electrode active material A and the negative electrode active material B were fully mixed at a mass ratio of 1:1, and the obtained positive electrode active material was used to prepare a positive electrode. The mixed positive electrode active material had a BET specific surface area of 1.2 m 2 /g, an average primary particle size of 0.8 μm, a median particle size d 50 of 6.5 μm, and a tap density of 2.1 g/cm 3 .
在N甲基吡咯烷酮溶剂中混合90质量%上述的混合后的正极活性物质A、5质量%的作为导电材料的乙炔黑和5质量%作为粘合剂的聚偏氟乙烯(PVdF),制成浆料。将得到的浆料涂布在厚度15μm的铝箔两面,并干燥,用压制机压延成厚度为70μm,再将其切成具有宽100mm、长100mm大小的正极活性物质层和宽30mm的未涂布部分的形状,制成正极。正极活性物质的密度为2.35g/cm3,(一面的正极活性物质层的厚度)/(集电体的厚度)的值为1.8。In N methylpyrrolidone solvent, mix 90% by mass of the above-mentioned mixed positive electrode active material A, 5% by mass of acetylene black as a conductive material, and 5% by mass of polyvinylidene fluoride (PVdF) as a binder to form slurry. The obtained slurry was coated on both sides of an aluminum foil with a thickness of 15 μm, dried, rolled into a thickness of 70 μm with a press, and then cut into a positive electrode active material layer with a width of 100 mm and a length of 100 mm and an uncoated layer with a width of 30 mm. The shape of the part is made into a positive electrode. The density of the positive electrode active material was 2.35 g/cm 3 , and the value of (thickness of positive electrode active material layer on one side)/(thickness of current collector) was 1.8.
《负极的制作》"The Making of Negative Pole"
在98重量份人造石墨粉末KS-44(timcal公司制造,商品名)中添加100重量份作为增稠剂的羧甲基纤维素钠的水性分散液(羧甲基纤维素钠的浓度为1质量%),2重量份作为粘合剂的苯乙烯-丁二烯橡胶的水性分散液(苯乙烯-丁二烯橡胶的浓度为50质量%),用分散器进行混合,制成浆料。将得到的浆料涂布在厚度10μm的铜箔两面,并干燥,用压制机压延成厚度为75μm,再将其切成具有宽104mm、长104mm大小的负极活性物质层和宽30mm的未涂布部分的形状,作为负极。In 98 parts by weight of artificial graphite powder KS-44 (manufactured by timcal company, trade name), add 100 parts by weight of an aqueous dispersion of sodium carboxymethyl cellulose as a thickener (the concentration of sodium carboxymethyl cellulose is 1 mass %), 2 parts by weight of an aqueous dispersion of styrene-butadiene rubber as a binder (the concentration of styrene-butadiene rubber is 50% by mass), and mixed with a disperser to prepare a slurry. The obtained slurry is coated on both sides of a copper foil with a thickness of 10 μm, dried, rolled into a thickness of 75 μm with a press machine, and then cut into a negative electrode active material layer with a width of 104 mm and a length of 104 mm and an uncoated layer with a width of 30 mm. The shape of the cloth part is used as the negative electrode.
《非水电解液的制作》"Preparation of Non-Aqueous Electrolyte"
在干燥的氩氛围下,在纯化的碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的3∶3∶4(体积比)的混合溶剂中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。另外,含有0.3质量%的六甲基环三硅氧烷。Under a dry argon atmosphere, in a 3:3:4 (volume ratio) mixed solvent of purified ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC), with 1mol /L concentration dissolves well-dried lithium hexafluorophosphate (LiPF 6 ). In addition, 0.3% by mass of hexamethylcyclotrisiloxane was contained.
《电池的制作》"Battery Making"
将33片正极和34片负极交替配置,各电极之间夹入多孔聚乙烯片隔板(厚度为25μm),进行叠层。此时,使正极活性物质面与负极活性物质面相对,并且不会露出负极活性物质面之外。将该正极和负极各自的未涂布部分焊接,制作集电片,将电极组密封在电池罐(外部尺寸:120×110×10mm)中。然后,在装有电极组的电池罐中注入20mL非水电解液,使电极充分浸透,密封而制作电池。正极的电极面积总和与电池的外壳表面积总和之比为21.3。33 positive electrodes and 34 negative electrodes were alternately arranged, and a porous polyethylene sheet separator (thickness: 25 μm) was sandwiched between the electrodes for lamination. At this time, the surface of the positive electrode active material faces the surface of the negative electrode active material without protruding from the surface of the negative electrode active material. The respective uncoated parts of the positive and negative electrodes were welded to produce current collector sheets, and the electrode group was sealed in a battery can (outer dimensions: 120×110×10 mm). Then, 20 mL of non-aqueous electrolytic solution was poured into the battery can containing the electrode group, the electrodes were sufficiently permeated, and the battery was sealed to produce a battery. The ratio of the sum of the electrode areas of the positive electrode to the sum of the surface areas of the battery case was 21.3.
《电池的评价》"Battery Evaluation"
(电池容量的测定方法)(Measuring method of battery capacity)
对未经充放电循环的新电池,在25℃、4.2V~3.0V的电压范围下,以0.2C的电流值(将1小时放电额定容量的电流值作为1C,所述额定容量取决于1小时率(one-hour-rate)的放电容量,下同)进行5个循环的初期充放电。此时的第5个循环的0.2C放电容量作为初期容量。电池评价的结果示于正极[5]表2。For a new battery that has not been charged and discharged, at 25°C and a voltage range of 4.2V to 3.0V, with a current value of 0.2C (the current value of the rated capacity of 1 hour discharge is taken as 1C, and the rated capacity depends on 1 One-hour-rate (one-hour-rate) discharge capacity, the same below) was performed for 5 cycles of initial charge and discharge. The 0.2C discharge capacity at the fifth cycle at this time was taken as the initial capacity. The results of the battery evaluation are shown in Table 2 of the positive electrode [5].
(初期输出功率的测定方法)(Measurement method of initial output power)
在25℃的环境下,通过0.2C的恒定电流进行150分钟充电,分别以0.1C、0.3C、1.0C、3.0C、10.0C放电10秒钟,测定第10秒的电压。将电流-电压直线和下限电压(3V)所包围的三角形的面积作为输出功率(W)。电池评价的结果示于正极[5]表2。In an environment of 25°C, charge was performed for 150 minutes with a constant current of 0.2C, and discharge was performed at 0.1C, 0.3C, 1.0C, 3.0C, and 10.0C for 10 seconds, respectively, and the voltage at the 10th second was measured. The area of the triangle surrounded by the current-voltage line and the lower limit voltage (3V) was taken as the output power (W). The results of the battery evaluation are shown in Table 2 of the positive electrode [5].
(循环试验(耐久后电池容量和耐久后输出功率的测定方法))(Cycle test (measuring method of battery capacity after endurance and output power after endurance))
在被视为锂二次电池的实际使用上限温度的60℃的高温环境下,进行循环试验。以2C恒定电流恒定电压法充电到充电上限电压4.2V后,以2C的恒定电流放电至放电终止电压3.0V,以此作为一个充放电循环,反复进行该循环,直至500循环。对于循环试验结束后的电池,在25℃的环境下以0.2C的电流值进行3个循环的充放电,以第3个循环的0.2C放电容量作为耐久后电池容量。另外,对于循环试验结束后的电池,进行输出功率测定,作为耐久后输出功率。电池评价的结果示于正极[5]表2。The cycle test was performed in a high-temperature environment of 60° C., which is regarded as the upper limit temperature for practical use of lithium secondary batteries. After charging to the charging upper limit voltage of 4.2V by 2C constant current and constant voltage method, discharge to the end-of-discharge voltage of 3.0V with a constant current of 2C, which is regarded as a charge-discharge cycle, and this cycle is repeated until 500 cycles. For the battery after the cycle test, three cycles of charging and discharging were performed at a current value of 0.2C in an environment of 25°C, and the 0.2C discharge capacity of the third cycle was used as the battery capacity after endurance. In addition, for the battery after the cycle test, the output power was measured, and it was set as the output power after durability. The results of the battery evaluation are shown in Table 2 of the positive electrode [5].
正极[5]实施例2Positive pole [5] embodiment 2
使非水电解液含有0.3质量%的甲磺酸三甲基甲硅烷基酯来代替六甲基环三硅氧烷,除此以外,与正极[5]实施例1同样实施。电池评价的结果示于正极[5]表2。The non-aqueous electrolytic solution was carried out in the same manner as in Example 1 of the positive electrode [5] except that 0.3% by mass of trimethylsilyl methanesulfonate was contained instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 2 of the positive electrode [5].
正极[5]实施例3Positive electrode [5] Example 3
使非水电解液含有0.3质量%的苯基二甲基氟硅烷来代替六甲基环三硅氧烷,除此以外,与正极[5]实施例1同样实施。电池评价的结果示于正极[5]表2。The nonaqueous electrolytic solution was made to contain 0.3 mass % of phenyldimethylfluorosilane instead of hexamethylcyclotrisiloxane, and it carried out similarly to positive electrode [5] Example 1. The results of the battery evaluation are shown in Table 2 of the positive electrode [5].
正极[5]实施例4Positive electrode [5] embodiment 4
使非水电解液含有0.3质量%的二氟磷酸锂来代替六甲基环三硅氧烷,除此以外,与正极[5]实施例1同样实施。电池评价的结果示于正极[5]表2。The non-aqueous electrolytic solution was made to contain 0.3 mass % of lithium difluorophosphate instead of hexamethylcyclotrisiloxane, and it carried out similarly to positive electrode [5] Example 1. The results of the battery evaluation are shown in Table 2 of the positive electrode [5].
正极[5]比较例1Positive electrode [5] Comparative Example 1
除了非水电解液中不含六甲基环三硅氧烷以外,与正极[5]实施例1同样实施。电池评价的结果示于正极[5]表2。Except not containing hexamethylcyclotrisiloxane in the non-aqueous electrolytic solution, it was carried out in the same manner as the positive electrode [5] Example 1. The results of the battery evaluation are shown in Table 2 of the positive electrode [5].
正极[5]表2Positive electrode [5] Table 2
[表25][Table 25]
正极[5]实施例5Positive electrode [5] embodiment 5
使用正极活性物质A和正极活性物质E以1∶1的质量比充分混合而得到的正极活性物质作为正极活性物质,制成正极,用压制机压延成厚度78μm,并使用32片该正极和33片负极,除此以外,与正极[5]实施例1同样实施。(正极一面的正极活性物质层厚度)/(正极集电体的厚度)为2.1,正极的电极面积总和与电池的外壳表面积总和之比为20.6。该混合的正极活性物质的BET比表面积为1.2m2/g,平均一次粒径为0.8μm,中值粒径d50为5.7μm,振实密度为2.0g/cm3。电池评价结果示于正极[5]表3。The positive active material obtained by using the positive active material A and the positive active material E in a mass ratio of 1:1 is fully mixed as the positive active material to make a positive electrode, which is rolled into a thickness of 78 μm with a pressing machine, and 32 pieces of the positive electrode and 33 are used. The sheet negative electrode was carried out in the same manner as in Example 1 of the positive electrode [5] except for this. (Thickness of the positive electrode active material layer on the positive electrode side)/(Thickness of the positive electrode current collector) was 2.1, and the ratio of the total electrode area of the positive electrode to the total surface area of the battery case was 20.6. The mixed positive electrode active material had a BET specific surface area of 1.2 m 2 /g, an average primary particle size of 0.8 μm, a median particle size d 50 of 5.7 μm, and a tap density of 2.0 g/cm 3 . The battery evaluation results are shown in Table 3 of the positive electrode [5].
正极[5]实施例6Positive electrode [5] Embodiment 6
使非水电解液含有0.3质量%的甲磺酸三甲基甲硅烷基酯来代替六甲基环三硅氧烷,除此以外,与正极[5]实施例5同样实施。电池评价的结果示于正极[5]表3。The nonaqueous electrolytic solution was carried out in the same manner as in positive electrode [5] Example 5 except that 0.3% by mass of trimethylsilyl methanesulfonate was included instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 3 of the positive electrode [5].
正极[5]实施例7Positive electrode [5] embodiment 7
使非水电解液含有0.3质量%的苯基二甲基氟硅烷来代替六甲基环三硅氧烷,除此以外,与正极[5]实施例5同样实施。电池评价的结果示于正极[5]表3。The nonaqueous electrolytic solution was made to contain 0.3 mass % of phenyldimethylfluorosilane instead of hexamethylcyclotrisiloxane, and it carried out similarly to positive electrode [5] Example 5. The results of the battery evaluation are shown in Table 3 of the positive electrode [5].
正极[5]实施例8Positive electrode [5] embodiment 8
使非水电解液含有0.3质量%的二氟磷酸锂来代替六甲基环三硅氧烷,除此以外,与正极[5]实施例5同样实施。电池评价的结果示于正极[5]表3。The non-aqueous electrolytic solution was made to contain 0.3 mass % of lithium difluorophosphate instead of hexamethylcyclotrisiloxane, and it carried out similarly to positive electrode [5] Example 5. The results of the battery evaluation are shown in Table 3 of the positive electrode [5].
正极[5]比较例2Positive electrode [5] Comparative example 2
除了非水电解液中不含六甲基环三硅氧烷以外,与正极[5]实施例5同样实施。电池评价的结果示于正极[5]表3。Except that hexamethylcyclotrisiloxane is not contained in the non-aqueous electrolyte solution, it is carried out in the same manner as the positive electrode [5] Example 5. The results of the battery evaluation are shown in Table 3 of the positive electrode [5].
正极[5]表3Positive electrode [5] Table 3
[表26][Table 26]
正极[5]实施例9Positive electrode [5] embodiment 9
使用正极活性物质A和正极活性物质F以1∶1的质量比充分混合而得到的正极活性物质作为正极活性物质,制成正极,用压制机压延成厚度91μm,并使用30片该正极和31片负极,除此以外,与正极[5]实施例1同样实施。(正极一面的正极活性物质层厚度)/(正极集电体的厚度)为2.5,正极的电极面积总和与电池的外壳表面积总和之比为19.4。该混合的正极活性物质的BET比表面积为1.0m2/g,平均一次粒径为0.6μm,中值粒径d50为7.5μm,振实密度为2.2g/cm3。电池评价结果示于正极[5]表4。Use the positive active material obtained by fully mixing the positive active material A and the positive active material F with a mass ratio of 1:1 as the positive active material to make the positive pole, roll it into a thickness of 91 μm with a pressing machine, and use 30 pieces of the positive pole and 31 The sheet negative electrode was carried out in the same manner as in Example 1 of the positive electrode [5] except for this. (Thickness of the positive electrode active material layer on the positive electrode side)/(Thickness of the positive electrode current collector) was 2.5, and the ratio of the total electrode area of the positive electrode to the total surface area of the battery case was 19.4. The BET specific surface area of the mixed positive electrode active material was 1.0 m 2 /g, the average primary particle size was 0.6 μm, the median particle size d 50 was 7.5 μm, and the tap density was 2.2 g/cm 3 . The battery evaluation results are shown in Table 4 of the positive electrode [5].
正极[5]实施例10Positive electrode [5] Example 10
使非水电解液含有0.3质量%的甲磺酸三甲基甲硅烷基酯来代替六甲基环三硅氧烷,除此以外,与正极[5]实施例9同样实施。电池评价的结果示于正极[5]表4。The non-aqueous electrolytic solution was carried out in the same manner as in positive electrode [5] Example 9 except that 0.3% by mass of trimethylsilyl methanesulfonate was included instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 4 of the positive electrode [5].
正极[5]实施例11Positive electrode [5] Example 11
使非水电解液含有0.3质量%的苯基二甲基氟硅烷来代替六甲基环三硅氧烷,除此以外,与正极[5]实施例9同样实施。电池评价的结果示于正极[5]表4。The nonaqueous electrolytic solution was made to contain 0.3 mass % of phenyldimethylfluorosilane instead of hexamethylcyclotrisiloxane, and it carried out similarly to positive electrode [5] Example 9. The results of the battery evaluation are shown in Table 4 of the positive electrode [5].
正极[5]实施例12Positive electrode [5] Example 12
使非水电解液含有0.3质量%的二氟磷酸锂来代替六甲基环三硅氧烷,除此以外,与正极[5]实施例9同样实施。电池评价的结果示于正极[5]表4。The non-aqueous electrolytic solution was made to contain 0.3 mass % of lithium difluorophosphate instead of hexamethylcyclotrisiloxane, and it carried out similarly to positive electrode [5] Example 9. The results of the battery evaluation are shown in Table 4 of the positive electrode [5].
正极[5]比较例3Positive electrode [5] Comparative Example 3
除了非水电解液中不含六甲基环三硅氧烷以外,与正极[5]实施例9同样实施。电池评价的结果示于正极[5]表4。Except that hexamethylcyclotrisiloxane is not contained in the non-aqueous electrolyte solution, it is carried out in the same manner as the positive electrode [5] Example 9. The results of the battery evaluation are shown in Table 4 of the positive electrode [5].
正极[5]表4Positive electrode [5] Table 4
[表27][Table 27]
正极[5]实施例13Positive electrode [5] Example 13
使用正极活性物质C和正极活性物质E以1∶1的质量比充分混合而得到的正极活性物质作为正极活性物质,制成正极,用压制机压延成厚度72μm,使用33片该正极和34片负极,除此以外,与正极[5]实施例1同样实施。该混合后的正极活性物质的BET比表面积为0.9m2/g,平均一次粒径为0.7μm,中值粒径d50为6.7μm,振实密度为2.0g/cm3。(正极一面的正极活性物质层厚度)/(正极集电体的厚度)为1.9,正极的电极面积总和与电池的外壳表面积总和之比为21.3。除容量测定中的电压范围为3.0~4.1V、循环试验的上限电压为4.1V以外,与正极[5]实施例1同样地评价电池。电池评价结果示于正极[5]表5。Use the positive active material obtained by fully mixing the positive active material C and the positive active material E with a mass ratio of 1:1 as the positive active material to make the positive electrode, and roll it into a thickness of 72 μm with a press machine. Use 33 pieces of the positive electrode and 34 pieces The negative electrode was carried out in the same manner as in Example 1 of the positive electrode [5] except for this. The mixed positive electrode active material had a BET specific surface area of 0.9 m 2 /g, an average primary particle size of 0.7 μm, a median particle size d 50 of 6.7 μm, and a tap density of 2.0 g/cm 3 . (Thickness of the positive electrode active material layer on the positive electrode side)/(Thickness of the positive electrode current collector) was 1.9, and the ratio of the total electrode area of the positive electrode to the total surface area of the battery case was 21.3. The battery was evaluated in the same manner as in Example 1 of the positive electrode [5], except that the voltage range in the capacity measurement was 3.0 to 4.1 V, and the upper limit voltage in the cycle test was 4.1 V. The battery evaluation results are shown in Table 5 of the positive electrode [5].
正极[5]实施例14Positive electrode [5] Example 14
使非水电解液含有0.3质量%的甲磺酸三甲基甲硅烷基酯来代替六甲基环三硅氧烷,除此以外,与正极[5]实施例13同样实施。电池评价的结果示于正极[5]表5。The non-aqueous electrolytic solution was carried out in the same manner as in Example 13 of the positive electrode [5] except that 0.3% by mass of trimethylsilyl methanesulfonate was included instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 5 of the positive electrode [5].
正极[5]实施例15Positive electrode [5] Example 15
使非水电解液含有0.3质量%的苯基二甲基氟硅烷来代替六甲基环三硅氧烷,除此以外,与正极[5]实施例13同样实施。电池评价的结果示于正极[5]表5。The same procedure was carried out as in Example 13 of the positive electrode [5] except that the nonaqueous electrolytic solution contained 0.3% by mass of phenyldimethylfluorosilane instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 5 of the positive electrode [5].
正极[5]实施例16Positive electrode [5] Example 16
使非水电解液含有0.3质量%的二氟磷酸锂来代替六甲基环三硅氧烷,除此以外,与正极[5]实施例13同样实施。电池评价的结果示于正极[5]表5。The procedure was carried out in the same manner as in Example 13 of the positive electrode [5] except that the nonaqueous electrolytic solution contained 0.3% by mass of lithium difluorophosphate instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 5 of the positive electrode [5].
正极[5]比较例4Positive electrode [5] Comparative example 4
除了非水电解液中不含六甲基环三硅氧烷以外,与正极[5]实施例13同样实施。电池评价的结果示于正极[5]表5。Except not containing hexamethylcyclotrisiloxane in the non-aqueous electrolytic solution, the same implementation was carried out as in Example 13 of the positive electrode [5]. The results of the battery evaluation are shown in Table 5 of the positive electrode [5].
正极[5]表5Positive electrode [5] Table 5
[表28][Table 28]
[2453]正极[5]实施例17 [2453] positive electrode [5] embodiment 17
使用正极活性物质C和正极活性物质F以1∶1的质量比充分混合而得到的正极活性物质作为正极活性物质,制成正极,用压制机压延成厚度78μm,使用32片该正极和33片负极,除此以外,与正极[5]实施例1同样实施。(正极一面的正极活性物质层厚度)/(正极集电体的厚度)为2.1,正极的电极面积总和与电池的外壳表面积总和之比为20.6。该混合的正极活性物质的BET比表面积为0.8m2/g,平均一次粒径为0.6μm,中值粒径d50为8.5μm,振实密度为2.2g/cm3。电池评价结果示于正极[5]表6。The positive active material obtained by fully mixing the positive active material C and the positive active material F at a mass ratio of 1:1 is used as the positive active material to make the positive electrode, which is rolled into a thickness of 78 μm with a press machine, and 32 pieces of the positive electrode and 33 pieces are used. The negative electrode was carried out in the same manner as in Example 1 of the positive electrode [5] except for this. (Thickness of the positive electrode active material layer on the positive electrode side)/(Thickness of the positive electrode current collector) was 2.1, and the ratio of the total electrode area of the positive electrode to the total surface area of the battery case was 20.6. The BET specific surface area of the mixed positive electrode active material was 0.8 m 2 /g, the average primary particle size was 0.6 μm, the median particle size d 50 was 8.5 μm, and the tap density was 2.2 g/cm 3 . The battery evaluation results are shown in Table 6 of the positive electrode [5].
正极[5]实施例18Positive electrode [5] Example 18
使非水电解液含有0.3质量%的甲磺酸三甲基甲硅烷基酯来代替六甲基环三硅氧烷,除此以外,与正极[5]实施例17同样实施。电池评价的结果示于正极[5]表6。The non-aqueous electrolytic solution was carried out in the same manner as in Example 17 of the positive electrode [5] except that 0.3% by mass of trimethylsilyl methanesulfonate was included instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 6 of the positive electrode [5].
正极[5]实施例19Positive electrode [5] Example 19
使非水电解液含有0.3质量%的苯基二甲基氟硅烷来代替六甲基环三硅氧烷,除此以外,与正极[5]实施例17同样实施。电池评价的结果示于正极[5]表6。The non-aqueous electrolytic solution was made to contain 0.3 mass % of phenyldimethylfluorosilane instead of hexamethylcyclotrisiloxane, and it carried out similarly to positive electrode [5] Example 17. The results of the battery evaluation are shown in Table 6 of the positive electrode [5].
正极[5]实施例20Positive electrode [5] Example 20
使非水电解液含有0.3质量%的二氟磷酸锂来代替六甲基环三硅氧烷,除此以外,与正极[5]实施例17同样实施。电池评价的结果示于正极[5]表6。The same procedure as in Example 17 of the positive electrode [5] was carried out except that the nonaqueous electrolytic solution contained 0.3% by mass of lithium difluorophosphate instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 6 of the positive electrode [5].
正极[5]比较例5Positive electrode [5] Comparative Example 5
除了非水电解液中不含六甲基环三硅氧烷以外,与正极[5]实施例17同样实施。电池评价的结果示于正极[5]表6。Except that hexamethylcyclotrisiloxane is not contained in the non-aqueous electrolyte solution, the same implementation was carried out as in Example 17 of the positive electrode [5]. The results of the battery evaluation are shown in Table 6 of the positive electrode [5].
正极[5]表6Positive electrode [5] Table 6
[表29][Table 29]
[2466]正极[5]实施例21 [2466] positive electrode [5] embodiment 21
使用正极活性物质D和正极活性物质E以1∶1的质量比充分混合而得到的正极活性物质作为正极活性物质,制成正极,用压制机压延成厚度72μm,使用33片该正极和34片负极,除此以外,与正极[5]实施例1同样实施。该混合后的正极活性物质的BET比表面积为0.9m2/g,平均一次粒径为0.7μm,中值粒径d50为6.7μm,振实密度为2.0g/cm3。(正极一面的正极活性物质层厚度)/(正极集电体的厚度)为1.9,正极的电极面积总和与电池的外壳表面积总和之比为21.3。除容量测定中的电压范围为3.0~4.1V、循环试验的上限电压为4.1V以外,与正极[5]实施例1同样地评价电池。电池评价结果示于正极[5]表7。Use the positive active material obtained by fully mixing the positive active material D and the positive active material E with a mass ratio of 1:1 as the positive active material to make the positive electrode, and roll it into a thickness of 72 μm with a press machine. Use 33 pieces of the positive electrode and 34 pieces The negative electrode was carried out in the same manner as in Example 1 of the positive electrode [5] except for this. The mixed positive electrode active material had a BET specific surface area of 0.9 m 2 /g, an average primary particle size of 0.7 μm, a median particle size d 50 of 6.7 μm, and a tap density of 2.0 g/cm 3 . (Thickness of the positive electrode active material layer on the positive electrode side)/(Thickness of the positive electrode current collector) was 1.9, and the ratio of the total electrode area of the positive electrode to the total surface area of the battery case was 21.3. The battery was evaluated in the same manner as in Example 1 of the positive electrode [5], except that the voltage range in the capacity measurement was 3.0 to 4.1 V, and the upper limit voltage in the cycle test was 4.1 V. The battery evaluation results are shown in Table 7 of the positive electrode [5].
正极[5]实施例22Positive electrode [5] Example 22
使非水电解液含有0.3质量%的甲磺酸三甲基甲硅烷基酯来代替六甲基环三硅氧烷,除此以外,与正极[5]实施例17同样实施。电池评价的结果示于正极[5]表7。The non-aqueous electrolytic solution was made to contain 0.3 mass % of trimethylsilyl methanesulfonate instead of hexamethylcyclotrisiloxane, and it carried out similarly to positive electrode [5] Example 17. The results of the battery evaluation are shown in Table 7 of the positive electrode [5].
正极[5]实施例23Positive electrode [5] Example 23
使非水电解液含有0.3质量%的苯基二甲基氟硅烷来代替六甲基环三硅氧烷,除此以外,与正极[5]实施例17同样实施。电池评价的结果示于正极[5]表7。The non-aqueous electrolytic solution was made to contain 0.3 mass % of phenyldimethylfluorosilane instead of hexamethylcyclotrisiloxane, and it carried out similarly to positive electrode [5] Example 17. The results of the battery evaluation are shown in Table 7 of the positive electrode [5].
正极[5]实施例24Positive electrode [5] Example 24
使非水电解液含有0.3质量%的二氟磷酸锂来代替六甲基环三硅氧烷,除此以外,与正极[5]实施例17同样实施。电池评价的结果示于正极[5]表7。The same procedure as in Example 17 of the positive electrode [5] was carried out except that the nonaqueous electrolytic solution contained 0.3% by mass of lithium difluorophosphate instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 7 of the positive electrode [5].
正极[5]比较例6Positive electrode [5] Comparative example 6
除了非水电解液中不含六甲基环三硅氧烷以外,与正极[5]实施例17同样实施。电池评价的结果示于正极[5]表7。Except that hexamethylcyclotrisiloxane is not contained in the non-aqueous electrolyte solution, the same implementation was carried out as in Example 17 of the positive electrode [5]. The results of the battery evaluation are shown in Table 7 of the positive electrode [5].
正极[5]表7Positive electrode [5] Table 7
[表30][Table 30]
正极[5]实施例25Positive electrode [5] Example 25
使用正极活性物质E和正极活性物质F以1∶1的质量比充分混合而得到的正极活性物质作为正极活性物质,制成正极,用压制机压延成厚度86μm,使用31片该正极和32片负极,除此以外,与正极[5]实施例1同样实施。(正极一面的正极活性物质层厚度)/(正极集电体的厚度)为2.4,正极的电极面积总和与电池的外壳表面积总和之比为20.0。该混合后的正极活性物质的BET比表面积为1.1m2/g,平均一次粒径为0.6μm,中值粒径d50为6.2μm,振实密度为2.0g/cm3。电池评价结果示于正极[5]表8。Use the positive active material obtained by fully mixing the positive active material E and the positive active material F at a mass ratio of 1:1 as the positive active material to make a positive electrode, and roll it into a thickness of 86 μm with a press machine. Use 31 pieces of the positive electrode and 32 pieces The negative electrode was carried out in the same manner as in Example 1 of the positive electrode [5] except for this. (Thickness of the positive electrode active material layer on the positive electrode side)/(Thickness of the positive electrode current collector) was 2.4, and the ratio of the total electrode area of the positive electrode to the total surface area of the battery case was 20.0. The mixed positive electrode active material had a BET specific surface area of 1.1 m 2 /g, an average primary particle size of 0.6 μm, a median particle size d 50 of 6.2 μm, and a tap density of 2.0 g/cm 3 . The battery evaluation results are shown in Table 8 of the positive electrode [5].
正极[5]实施例26Positive electrode [5] Example 26
使非水电解液含有0.3质量%的甲磺酸三甲基甲硅烷基酯来代替六甲基环三硅氧烷,除此以外,与正极[5]实施例17同样实施。电池评价的结果示于正极[5]表8。The non-aqueous electrolytic solution was carried out in the same manner as in Example 17 of the positive electrode [5] except that 0.3% by mass of trimethylsilyl methanesulfonate was included instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 8 of the positive electrode [5].
正极[5]实施例27Positive electrode [5] Example 27
使非水电解液含有0.3质量%的苯基二甲基氟硅烷来代替六甲基环三硅氧烷,除此以外,与正极[5]实施例17同样实施。电池评价的结果示于正极[5]表8。The non-aqueous electrolytic solution was made to contain 0.3 mass % of phenyldimethylfluorosilane instead of hexamethylcyclotrisiloxane, and it carried out similarly to positive electrode [5] Example 17. The results of the battery evaluation are shown in Table 8 of the positive electrode [5].
正极[5]实施例28Positive electrode [5] Example 28
使非水电解液含有0.3质量%的二氟磷酸锂来代替六甲基环三硅氧烷,除此以外,与正极[5]实施例17同样实施。电池评价的结果示于正极[5]表8。The same procedure as in Example 17 of the positive electrode [5] was carried out except that the nonaqueous electrolytic solution contained 0.3% by mass of lithium difluorophosphate instead of hexamethylcyclotrisiloxane. The results of the battery evaluation are shown in Table 8 of the positive electrode [5].
正极[5]比较例7Positive electrode [5] Comparative example 7
除了非水电解液中不含六甲基环三硅氧烷以外,与正极[5]实施例17同样实施。电池评价的结果示于正极[5]表8。Except that hexamethylcyclotrisiloxane is not contained in the non-aqueous electrolyte solution, the same implementation was carried out as in Example 17 of the positive electrode [5]. The results of the battery evaluation are shown in Table 8 of the positive electrode [5].
正极[5]表8Positive pole [5] Table 8
[表31][Table 31]
从正极[5]表2~正极[5]表8的结果可知,在任一个正极中,由于在非水电解液中都含有特定化合物,因此,输出功率、容量保持率得到提高,并且即使在循环试验后,也可以充分保持电池容量和输出功率。From the results in Table 2 of the positive electrode [5] to Table 8 of the positive electrode [5], it can be seen that in any positive electrode, since the non-aqueous electrolyte contains a specific compound, the output power and capacity retention are improved, and even in the cycle After the test, the battery capacity and output power can also be fully maintained.
负极[1][负极活性物质的制作]Negative electrode[1][Production of negative electrode active material]
(负极活性物质的制作1)(Production of negative electrode active material 1)
为了防止作为粒子状碳质物质的市售的天然石墨粉末中混入粗大粒子,使用ASTM400目的筛子反复筛5次。将这样得到的负极材料作为碳质物质(A)。In order to prevent the commercially available natural graphite powder, which is a particulate carbonaceous material, from being mixed with coarse particles, the sieve was repeatedly sieved five times using an ASTM 400 mesh sieve. The negative electrode material thus obtained is referred to as a carbonaceous substance (A).
(负极活性物质的制作2)(Production of negative electrode active material 2)
在非活性气体中对石脑油热解时得到的石油类重油实施1300℃的碳化处理,然后,通过对烧结物进行分级处理得到碳质物质(B)。分级处理中,为了防止混入粗大粒子,使用ASTM400目的筛子反复筛5次。Petroleum heavy oil obtained during pyrolysis of naphtha was subjected to carbonization treatment at 1300° C. in an inert gas, and then the carbonaceous material (B) was obtained by classifying the sintered product. In the classification process, in order to prevent the mixing of coarse particles, the ASTM400 mesh sieve was used to repeatedly sieve 5 times.
(负极活性物质的制作3)(Production of negative electrode active material 3)
将95质量%的碳质物质(A)和5质量%的碳质物质(B)均匀混合,将混合物作为2种结晶性碳质物质混合物(C)。95% by mass of the carbonaceous substance (A) and 5% by mass of the carbonaceous substance (B) were uniformly mixed, and the mixture was made into a two-type crystalline carbonaceous substance mixture (C).
(负极活性物质的制作4)(Production of negative electrode active material 4)
在碳质物质(A)中混合石脑油热解时得到的石油类重油,在非活性气体中实施1300℃的碳化处理,然后,通过对烧结物进行分级处理得到碳质物质(D),所述复合碳质物质(D)是在碳质物质(A)粒子表面上包覆了具有不同结晶性的碳质物质的复合碳质物质。分级处理时,为了防止混入粗大粒子,使用ASTM400目的筛子反复筛5次。从残炭率可以确定,得到的负极活性物质粉末被相对于95重量份石墨为5重量的低结晶性碳质物质包覆。Petroleum heavy oil obtained during the pyrolysis of naphtha is mixed with the carbonaceous substance (A), carbonized at 1300° C. in an inert gas, and then the carbonaceous substance (D) is obtained by classifying the sintered product, The composite carbonaceous substance (D) is a composite carbonaceous substance in which carbonaceous substances having different crystallinity are coated on the surface of the carbonaceous substance (A) particle. During the classification process, in order to prevent the mixing of coarse particles, use an ASTM400 mesh sieve to repeatedly sieve 5 times. It was confirmed from the carbon residue rate that the obtained negative electrode active material powder was coated with 5 parts by weight of low-crystalline carbonaceous material relative to 95 parts by weight of graphite.
(负极活性物质的制作5)(Production of negative electrode active material 5)
除了减少负极活性物质的制作4中在石脑油热解时得到的石油类重油并混合以外,按照与负极活性物质的制作4同样的方法进行,得到复合碳质物质(E),其被相对于99重量份石墨为1重量的低结晶性碳质物质包覆。In addition to reducing the heavy petroleum oil obtained during the pyrolysis of naphtha in the making of the negative active material 4 and mixing, follow the same method as the making of the negative active material 4 to obtain a composite carbonaceous material (E), which is compared 99 parts by weight of graphite is coated with 1 weight of low-crystalline carbonaceous material.
(负极活性物质的制作6)(Production of negative electrode active material 6)
除了增加负极活性物质的制作4中在石脑油热解时得到的石油类重油并混合以外,按照与负极活性物质的制作4同样的方法进行,得到复合碳质物质(F),其被相对于90重量份石墨为10重量的低结晶性碳质物质包覆。In addition to increasing the petroleum heavy oil obtained during the pyrolysis of naphtha in the making of negative active material 4 and mixing, follow the same method as making 4 of negative active material to obtain composite carbonaceous material (F), which is compared 90 parts by weight of graphite is coated with 10 parts by weight of low-crystalline carbonaceous material.
(负极活性物质的制作7)(Production of negative electrode active material 7)
除了增加负极活性物质的制作4中在石脑油热解时得到的石油类重油并混合以外,按照与负极活性物质的制作4同样的方法进行,得到复合碳质物质(G),其被相对于70重量份石墨为30重量的低结晶性碳质物质包覆。In addition to increasing the petroleum heavy oil obtained during the pyrolysis of naphtha in the making of the negative active material 4 and mixing, follow the same method as the making of the negative active material 4 to obtain a composite carbonaceous material (G), which is compared 70 parts by weight of graphite is coated with 30 parts by weight of low-crystalline carbonaceous material.
(负极活性物质的制作8)(Production of negative electrode active material 8)
除了在负极活性物质的制作4中在非活性气体中实施3000℃的石墨化处理以外,按照与负极活性物质的制作4相同的方法进行,得到复合碳质物质(I),其被相对于95重量份石墨为5重量的低结晶性碳质物质包覆。In addition to implementing the graphitization treatment at 3000° C. in an inert gas in the making 4 of the negative electrode active material, the same method as the making 4 of the negative electrode active material was carried out to obtain a composite carbonaceous material (I), which was compared to 95 The parts by weight of graphite are coated with 5 parts by weight of low-crystalline carbonaceous material.
(负极活性物质的制作9)(Production of negative electrode active material 9)
在碳质物质(A)中混合苯酚-甲醛溶液,在非活性气体中实施1300℃的碳化处理,然后,通过对烧结物进行分级处理得到复合碳质物质粉末,所述复合碳质物质粉末是在石墨粒子表面上包覆了具有不同结晶性的碳质物质的复合碳质物质粉末。分级处理时,为了防止混入粗大粒子,使用ASTM400目的筛子反复筛5次,得到复合碳质物质(J)。从残炭率可以确定,得到的负极活性物质粉末被相对于95重量份石墨为5重量的低结晶性碳质物质包覆。Mix the phenol-formaldehyde solution in the carbonaceous substance (A), implement carbonization treatment at 1300° C. in an inert gas, and then obtain a composite carbonaceous substance powder by classifying the sintered material, and the composite carbonaceous substance powder is A composite carbonaceous substance powder in which carbonaceous substances with different crystallinity are coated on the surface of graphite particles. During the classification process, in order to prevent the mixing of coarse particles, the ASTM400 mesh sieve was used to sieve repeatedly 5 times to obtain the composite carbonaceous material (J). It was confirmed from the carbon residue rate that the obtained negative electrode active material powder was coated with 5 parts by weight of low-crystalline carbonaceous material relative to 95 parts by weight of graphite.
(负极活性物质的制作10)(Production of negative electrode active material 10)
在反应炉中于460℃下将不溶于喹啉的成分为0.05质量%以下的煤焦油沥青进行10小时热处理,使用粉碎机(seishin企业公司制造的orient mill(オリエントミル))将得到的块状碳质物质粉碎,再使用微粉碎机(matsubo(マツボ一)公司制造的涡轮式磨)进行微粉碎,微细化至中值粒径17μm。将该粒子装入金属制造的容器中,在箱形的电炉中、氮气流通下于540℃进行2小时热处理。得到的块状物质用粗粉碎机(吉田制作所制造的辊式压碎机(ロ一ルジヨ一クラツシヤ一))进行粉碎,再用微粉碎机(matsubo公司制造的涡轮式磨)进行微粉碎,将得到的粉末装入容器内,在氮气氛围下,用电炉在1300℃下烧结1小时。然后,对得到的烧结物进行分级处理,由此得到碳质物质(K)。分级处理时,为了防止混入粗大粒子,使用ASTM400目的筛子反复筛5次。Coal tar pitch having a quinoline-insoluble component of 0.05% by mass or less was heat-treated in a reaction furnace at 460°C for 10 hours, and the obtained block was crushed using a pulverizer (orient mill manufactured by Seishin Enterprise Co., Ltd.). The carbonaceous material was pulverized and then finely pulverized using a pulverizer (turbine mill manufactured by Matsubo Co., Ltd.) to a median particle size of 17 μm. The particles were placed in a metal container, and heat-treated at 540° C. for 2 hours in a box-shaped electric furnace under a flow of nitrogen gas. Obtained massive material was pulverized with a coarse pulverizer (roller crusher manufactured by Yoshida Seisakusho Co., Ltd.), and finely pulverized with a fine pulverizer (turbine mill manufactured by Matsubo Co., Ltd.). The obtained powder was put into a container, and sintered in an electric furnace at 1300° C. for 1 hour in a nitrogen atmosphere. Then, the obtained sintered product is subjected to classification treatment to obtain a carbonaceous material (K). During the classification process, in order to prevent the mixing of coarse particles, use an ASTM400 mesh sieve to repeatedly sieve 5 times.
(负极活性物质的制作11)(Production of negative electrode active material 11)
在碳质物质(K)中混合苯酚-甲醛溶液,在非活性气体中实施1300℃的碳化处理,然后,通过对烧结物进行分级处理得到复合碳质物质粉末,所述复合碳质物质粉末是在石墨粒子表面上包覆了具有不同结晶性的碳质物质的复合碳质物质粉末。分级处理时,为了防止混入粗大粒子,使用ASTM400目的筛子反复筛5次,得到复合碳质物质(L)。从残炭率可以确定,得到的负极活性物质粉末被相对于60重量份石墨为40重量的低结晶性碳质物质包覆。Mix the phenol-formaldehyde solution in the carbonaceous substance (K), implement carbonization treatment at 1300° C. in an inert gas, and then obtain a composite carbonaceous substance powder by classifying the sintered material, and the composite carbonaceous substance powder is A composite carbonaceous substance powder in which carbonaceous substances with different crystallinity are coated on the surface of graphite particles. During the classification process, in order to prevent mixing of coarse particles, the ASTM400 mesh sieve was used to repeatedly sieve 5 times to obtain the composite carbonaceous material (L). It was confirmed from the carbon residue rate that the obtained negative electrode active material powder was coated with 40 parts by weight of low-crystalline carbonaceous material relative to 60 parts by weight of graphite.
(负极活性物质的制作12)(Production of negative electrode active material 12)
在碳质物质(K)中混合石脑油热解时得到的石油类重油,在非活性气体中实施1300℃的碳化处理,然后,通过对烧结物进行分级处理得到复合碳质物质粉末,所述复合碳质物质粉末是在石墨粒子表面上包覆了具有不同结晶性的碳质物质的复合碳质物质粉末。分级处理时,为了防止混入粗大粒子,使用ASTM400目的筛子反复筛5次,得到复合碳质物质(M)。从残炭率可以确定,得到的负极活性物质粉末被相对于95重量份石墨为5重量的低结晶性碳质物质包覆。Petroleum heavy oil obtained during the pyrolysis of naphtha is mixed with the carbonaceous substance (K), carbonized at 1300°C in an inert gas, and then the composite carbonaceous substance powder is obtained by classifying the sintered product. The composite carbonaceous substance powder is a composite carbonaceous substance powder in which carbonaceous substances having different crystallinity are coated on the surface of graphite particles. During the classification process, in order to prevent the mixing of coarse particles, the ASTM400 mesh sieve was used to repeatedly sieve 5 times to obtain the composite carbonaceous material (M). It was confirmed from the carbon residue rate that the obtained negative electrode active material powder was coated with 5 parts by weight of low-crystalline carbonaceous material relative to 95 parts by weight of graphite.
(负极活性物质的制作13)(Production of negative electrode active material 13)
在反应炉中于460℃下将不溶于喹啉的成分为0.05质量%以下的煤焦油沥青进行10小时热处理,使用粉碎机(senshin企业公司制造的orient mill)将得到的块状碳质物质粉碎,再使用微粉碎机(matsubo公司制造的涡轮式磨)进行微粉碎,微细化至中值粒径17μm。将该粒子装入金属制造的容器中,在箱形的电炉中、氮气流通下于540℃进行2小时热处理。得到的块状物质用粗粉碎机(吉田制作所制造的辊式压碎机)进行粉碎,再用微粉碎机(matsubo公司制造的涡轮式磨)进行微粉碎,将得到的粉末装入容器内,在氮气氛围下,用电炉在1000℃下烧结1小时。然后,将烧结后的粉末转移到石墨坩埚中,用直接通电炉在非活性气体氛围中于3000℃下用5小时进行石墨化,得到碳质物质(N)。在碳质物质(N)中混合石脑油热解时得到的石油类重油,在非活性气体中实施900℃的碳化处理,然后,通过对烧结物进行分级处理得到碳质物质(O),所述复合碳质物质(O)是在石墨粒子表面上包覆了具有不同结晶性的碳质物质而得到的。分级处理时,为了防止混入粗大粒子,使用ASTM400目的筛子反复筛5次。从残炭率可以确定,得到的负极活性物质粉末被相对于95重量份石墨为5重量的低结晶性碳质物质包覆。Coal tar pitch with a quinoline-insoluble component of 0.05% by mass or less was heat-treated in a reaction furnace at 460°C for 10 hours, and the obtained massive carbonaceous material was pulverized using a pulverizer (orient mill manufactured by Senshin Enterprise Co., Ltd.) , and then finely pulverized using a pulverizer (turbine mill manufactured by Matsubo Co., Ltd.) to a median particle size of 17 μm. The particles were placed in a metal container, and heat-treated at 540° C. for 2 hours in a box-shaped electric furnace under a flow of nitrogen gas. The obtained lumpy material was pulverized with a coarse pulverizer (roller crusher manufactured by Yoshida Seisakusho), and then finely pulverized with a fine pulverizer (turbine mill manufactured by Matsubo Co., Ltd.), and the obtained powder was put into a container , under a nitrogen atmosphere, sintered in an electric furnace at 1000 ° C for 1 hour. Then, the sintered powder was transferred to a graphite crucible, and graphitized in an inert gas atmosphere at 3000° C. for 5 hours in a direct electric furnace to obtain a carbonaceous material (N). Petroleum-based heavy oil obtained during pyrolysis of naphtha is mixed with the carbonaceous substance (N), carbonized at 900°C in an inert gas, and then the carbonaceous substance (O) is obtained by classifying the sintered product, The composite carbonaceous substance (O) is obtained by coating the surface of graphite particles with carbonaceous substances having different crystallinity. During the classification process, in order to prevent the mixing of coarse particles, use an ASTM400 mesh sieve to repeatedly sieve 5 times. It was confirmed from the carbon residue rate that the obtained negative electrode active material powder was coated with 5 parts by weight of low-crystalline carbonaceous material relative to 95 parts by weight of graphite.
(负极活性物质的制作14)(Production of negative electrode active material 14)
除了使用鳞片状天然石墨来代替负极活性物质的制作4中的碳质物质(A)以外,按照与负极活性物质的制作4相同的方法进行,得到复合碳质物质(P),其被相对于95重量份石墨为5重量的低结晶性碳质物质包覆。Except using flaky natural graphite to replace the carbonaceous material (A) in the making 4 of the negative electrode active material, carry out according to the making 4 identical method with the negative electrode active material, obtain composite carbonaceous material (P), it is compared with 95 parts by weight of graphite is coated with 5 parts by weight of low-crystalline carbonaceous material.
(负极活性物质的制作15)(Production of negative electrode active material 15)
除了减少负极活性物质的制作14中在石脑油热解时得到的石油类重油并混合以外,按照与负极活性物质的制作4同样的方法进行,然后,将得到的粉末转移到石墨坩埚中,用直接通电炉在非活性气体氛围中于3000℃下用5小时进行石墨化。然后,通过对烧结物进行分级处理得到碳质物质(Q),所述复合碳质物质(Q)是在石墨粒子表面上包覆了具有不同结晶性的碳质物质而得到的。分级处理时,为了防止混入粗大粒子,使用ASTM400目的筛子反复筛5次。从残炭率可以确定,得到的负极活性物质粉末被相对于99重量份石墨为1重量的低结晶性碳质物质包覆。In addition to reducing the heavy petroleum oil obtained during naphtha pyrolysis in the making of negative electrode active material 14 and mixing, follow the same method as making 4 of negative electrode active material, then, the powder obtained is transferred in the graphite crucible, Graphitization was performed at 3000° C. for 5 hours in an inert gas atmosphere using a direct electric furnace. Then, the carbonaceous substance (Q) is obtained by classifying the sintered product, and the composite carbonaceous substance (Q) is obtained by coating the carbonaceous substance with different crystallinity on the surface of graphite particles. During the classification process, in order to prevent the mixing of coarse particles, use an ASTM400 mesh sieve to repeatedly sieve 5 times. It was confirmed from the carbon residue rate that the obtained negative electrode active material powder was coated with 1 weight part of low-crystalline carbonaceous material relative to 99 parts by weight of graphite.
(负极活性物质的制作16)(Production of negative electrode active material 16)
除了使用体积平均粒径为48μm的天然石墨来代替负极活性物质的制作4中的碳质物质(A)以外,按照与负极活性物质的制作4相同的方法进行,得到复合碳质物质(R),其被相对于95重量份石墨为5重量的低结晶性碳质物质包覆。In addition to using natural graphite with a volume average particle diameter of 48 μm to replace the carbonaceous material (A) in the production 4 of the negative active material, the method is the same as that in the production 4 of the negative active material to obtain a composite carbonaceous material (R). , which is coated with 5 wt. parts of low-crystalline carbonaceous material relative to 95 wt. parts of graphite.
(负极活性物质的制作17)(Production of negative electrode active material 17)
除了使用纯化程度低的残留有1%灰分的天然石墨来代替负极活性物质的制作4中的碳质物质(A)以外,按照与负极活性物质的制作4相同的方法进行,得到复合碳质物质(S),其被相对于95重量份石墨为5重量的低结晶性碳质物质包覆。Except using natural graphite with 1% ash remaining in a low degree of purification to replace the carbonaceous material (A) in the making of the negative active material 4, proceed according to the same method as the making of the negative active material 4 to obtain a composite carbonaceous material (S) which is coated with 5 weight parts of low-crystalline carbonaceous substances with respect to 95 weight parts of graphite.
得到的负极活性物质的物性示于负极[1]表1。物性的测定方法与上述相同。The physical properties of the obtained negative electrode active material are shown in Table 1 of the negative electrode [1]. The measurement method of the physical properties is the same as above.
负极[1]表1Negative electrode [1] Table 1
[表32][Table 32]
负极[1][电池的制作]Negative electrode[1][Battery production]
《正极的制作1》"Making of Positive Pole 1"
在N-甲基吡咯烷酮溶剂中混合90质量%的作为正极活性物质的钴酸锂(LiCoO2)、5质量%的作为导电材料的乙炔黑和5质量%作为粘合剂的聚偏氟乙烯(PVdF),制成浆料。将得到的浆料涂布在厚度15μm的铝箔两面,并干燥,用压制机压延成厚度80μm,再将其切成具有宽100mm、长100mm大小的正极活性物质层和宽30mm的未涂布部分的形状,作为正极。此时的正极活性物质的密度为2.35g/cm3。90% by mass of lithium cobaltate (LiCoO 2 ) as a positive electrode active material, 5% by mass of acetylene black as a conductive material, and 5% by mass of polyvinylidene fluoride ( PVdF), made into slurry. The obtained slurry was coated on both sides of an aluminum foil with a thickness of 15 μm, dried, rolled to a thickness of 80 μm with a press, and then cut into a positive electrode active material layer having a width of 100 mm and a length of 100 mm and an uncoated portion of a width of 30 mm. shape, as the positive electrode. The density of the positive electrode active material at this time was 2.35 g/cm 3 .
《正极的制作2》"Making of Positive Pole 2"
除了每一面涂布的活性物质的质量为正极的制作1的2倍以外,与正极的制作1同样地进行正极电极的制作。The positive electrode was produced in the same manner as in the positive electrode production 1, except that the mass of the active material coated on each surface was twice that of the positive electrode production 1.
《正极的制作3》"Making of Positive Pole 3"
在N-甲基吡咯烷酮溶剂中混合90质量%的作为正极活性物质的LiNi0.80Co0.15Al0.05O2、5质量%的作为导电材料的乙炔黑和5质量%作为粘合剂的聚偏氟乙烯(PVdF),制成浆料。将得到的浆料涂布在厚度15μm的铝箔两面,并干燥,用压制机压延成厚度65μm,再将其切成具有宽100mm、长100mm大小的正极活性物质层和宽30mm的未涂布部分的形状,作为正极。此时的正极活性物质的密度为2.35g/cm3。90% by mass of LiNi 0.80 Co 0.15 Al 0.05 O 2 as a positive electrode active material, 5% by mass of acetylene black as a conductive material, and 5% by mass of polyvinylidene fluoride as a binder were mixed in N-methylpyrrolidone solvent (PVdF), made into slurry. The obtained slurry was coated on both sides of an aluminum foil with a thickness of 15 μm, dried, and rolled to a thickness of 65 μm with a press, and then cut into a positive electrode active material layer with a width of 100 mm and a length of 100 mm and an uncoated part with a width of 30 mm. shape, as the positive electrode. The density of the positive electrode active material at this time was 2.35 g/cm 3 .
《负极的制作1》"Making of Negative Pole 1"
在98重量份负极活性物质中添加100重量份作为增稠剂的羧甲基纤维素钠的水性分散液(羧甲基纤维素钠的浓度为1质量%)、2重量份作为粘合剂的苯乙烯-丁二烯橡胶的水性分散液(苯乙烯-丁二烯橡胶的浓度为50质量%),用分散器进行混合,制成浆料。将得到的浆料涂布在厚度10μm的压延铜箔两面,并干燥,用压制机压延成厚度75μm,再将其切成具有宽104mm、长104mm大小的负极活性物质层和宽30mm的未涂布部分的形状,作为负极。此时的负极活性物质密度为1.35g/cm3。Add 100 parts by weight of an aqueous dispersion of sodium carboxymethylcellulose as a thickener (the concentration of sodium carboxymethylcellulose is 1% by mass) and 2 parts by weight of sodium carboxymethylcellulose as a binder in 98 parts by weight of the negative electrode active material. An aqueous dispersion of styrene-butadiene rubber (the concentration of styrene-butadiene rubber is 50% by mass) was mixed with a disperser to prepare a slurry. The obtained slurry is coated on both sides of a rolled copper foil with a thickness of 10 μm, dried, rolled into a thickness of 75 μm with a press, and then cut into a negative electrode active material layer with a width of 104 mm and a length of 104 mm and an uncoated layer with a width of 30 mm. The shape of the cloth part is used as the negative electrode. The negative electrode active material density at this time was 1.35 g/cm 3 .
《负极的制作2》"The Making of Negative Pole 2"
除了每一面涂布的活性物质的质量为负极的制作1中使用量的2倍以外,与负极的制作1同样地进行负极电极的制作。The preparation of the negative electrode was performed in the same manner as in the preparation 1 of the negative electrode, except that the mass of the active material coated on each surface was twice the amount used in the preparation 1 of the negative electrode.
《负极的制作3》"The Making of Negative Pole 3"
除了使负极的制作1中一侧的活性物质的密度为1.70g/cm3以外,与负极的制作1同样地进行负极电极的制作。A negative electrode was produced in the same manner as in negative electrode production 1 except that the density of the active material on one side in negative electrode production 1 was 1.70 g/cm 3 .
《负极的制作4》"The Making of Negative Pole 4"
在95重量份负极活性物质中添加100重量份作为增稠剂的羧甲基纤维素钠的水性分散液(羧甲基纤维素钠的浓度为1质量%)、8重量份作为粘合剂的苯乙烯-丁二烯橡胶的水性分散液(苯乙烯-丁二烯橡胶的浓度为50质量%),用分散器进行混合,制成浆料。将得到的浆料涂布在厚度10μm的铜箔两面,并干燥,用压制机压延成厚度75μm,再将其切成具有宽104mm、长104mm大小的负极活性物质层和宽30mm的未涂布部分的形状,作为负极。此时的活性物质密度为1.35g/cm3。Add 100 parts by weight of an aqueous dispersion of sodium carboxymethylcellulose as a thickener (the concentration of sodium carboxymethylcellulose is 1% by mass) and 8 parts by weight of sodium carboxymethylcellulose as a binder in 95 parts by weight of the negative electrode active material. An aqueous dispersion of styrene-butadiene rubber (the concentration of styrene-butadiene rubber is 50% by mass) was mixed with a disperser to prepare a slurry. The obtained slurry was coated on both sides of a copper foil with a thickness of 10 μm, dried, rolled into a thickness of 75 μm with a press, and then cut into a negative electrode active material layer with a width of 104 mm and a length of 104 mm and an uncoated layer with a width of 30 mm. The shape of the part, as the negative pole. The active material density at this time was 1.35 g/cm 3 .
《电解液的制作1》"Production of Electrolyte 1"
在干燥的氩氛围下,在碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的混合物(体积比3∶3∶4)中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。另外,还含有0.3质量%的二氟磷酸锂盐(LiPO2F2)。Under a dry argon atmosphere, in a mixture of ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) (volume ratio 3:3:4), at a concentration of 1mol/L Dissolve well-dried lithium hexafluorophosphate (LiPF 6 ). In addition, 0.3% by mass of lithium difluorophosphate (LiPO 2 F 2 ) was contained.
《电解液的制作2》"Making Electrolyte 2"
在干燥的氩氛围下,在碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的混合物(体积比3∶3∶4)中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。另外,还含有0.3质量%的甲磺酸三甲基甲硅烷基酯。Under a dry argon atmosphere, in a mixture of ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) (volume ratio 3:3:4), at a concentration of 1mol/L Dissolve well-dried lithium hexafluorophosphate (LiPF 6 ). In addition, 0.3% by mass of trimethylsilyl methanesulfonate was contained.
《电解液的制作3》"The Production of Electrolyte 3"
在干燥的氩氛围下,在碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的混合物(体积比3∶3∶4)中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。另外,还含有0.3质量%的六甲基环三硅氧烷。Under a dry argon atmosphere, in a mixture of ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) (volume ratio 3:3:4), at a concentration of 1mol/L Dissolve well-dried lithium hexafluorophosphate (LiPF 6 ). In addition, 0.3% by mass of hexamethylcyclotrisiloxane was contained.
《电解液的制作4》"The Production of Electrolyte 4"
在干燥的氩氛围下,在碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的混合物(体积比3∶3∶4)中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。Under a dry argon atmosphere, in a mixture of ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) (volume ratio 3:3:4), at a concentration of 1mol/L Dissolve well-dried lithium hexafluorophosphate (LiPF 6 ).
《电池的制作1》"Battery Making 1"
将32片正极和33片负极交替配置,各电极之间夹入多孔聚乙烯片隔板(厚度为25μm),进行叠层。此时,使正极活性物质面与负极活性物质面相对,并且不会露出负极活性物质面之外。将该正极和负极各自的未涂布部分焊接,制作集电片,将电极组密封在电池罐(外部尺寸:120×110×10mm)中。然后,在装有电极组的电池罐中注入20mL非水电解液,使电极充分浸透,密封而制作方形电池。该电池的额定放电容量约为6安培小时(Ah),以10kHz交流法测定的直流电阻成分约为5毫欧(mΩ)。32 positive electrodes and 33 negative electrodes were arranged alternately, and a porous polyethylene sheet separator (thickness: 25 μm) was sandwiched between the electrodes for lamination. At this time, the surface of the positive electrode active material faces the surface of the negative electrode active material without protruding from the surface of the negative electrode active material. The respective uncoated parts of the positive and negative electrodes were welded to produce current collector sheets, and the electrode group was sealed in a battery can (outer dimensions: 120×110×10 mm). Then, 20 mL of non-aqueous electrolytic solution was poured into the battery can containing the electrode group, the electrodes were fully soaked, and sealed to produce a square battery. The rated discharge capacity of the battery is about 6 ampere hours (Ah), and the DC resistance component measured by the 10 kHz alternating current method is about 5 milliohms (mΩ).
《电池的制作2》"Battery Making 2"
将16片正极和17片负极交替配置,各电极之间夹入多孔聚乙烯片隔板(厚度为25μm),进行叠层。此时,使正极活性物质面与负极活性物质面相对,并且不会露出负极活性物质面之外。将该正极和负极各自的未涂布部分焊接,制作集电片,将电极组密封在电池罐(外部尺寸:120×110×10mm)中。然后,在装有电极组的电池罐中注入20mL非水电解液,使电极充分浸透,密封而制作方形电池。该电池的额定放电容量约为6安培小时(Ah),以10kHz交流法测定的直流电阻成分约为7毫欧(mΩ)。16 sheets of positive electrodes and 17 sheets of negative electrodes were alternately arranged, and a porous polyethylene sheet separator (thickness: 25 μm) was sandwiched between the electrodes to perform lamination. At this time, the surface of the positive electrode active material faces the surface of the negative electrode active material without protruding from the surface of the negative electrode active material. The respective uncoated parts of the positive and negative electrodes were welded to produce current collector sheets, and the electrode group was sealed in a battery can (outer dimensions: 120×110×10 mm). Then, 20 mL of non-aqueous electrolytic solution was poured into the battery can containing the electrode group, the electrodes were fully soaked, and sealed to produce a square battery. The battery has a rated discharge capacity of about 6 ampere hours (Ah), and a DC resistance component of about 7 milliohms (mΩ) measured by an alternating current method at 10 kHz.
负极[1]实施例1Negative electrode [1] Example 1
使用以《负极的制作1》项的负极活性物质作为两种结晶性碳质物质混合物(C)而制成的负极、《正极的制作1》项中制作的正极和《电解液的制作1》项中制作的电解液,通过《电池的制作1》项中的方法制作电池。通过下述《电池的评价》项中所述的方法和上述测定方法对该电池进行测定。Use the negative electrode made by using the negative electrode active material in "Making of Negative Electrode 1" as a mixture of two crystalline carbonaceous substances (C), the positive electrode made in "Making of Positive Electrode 1" and "Making of Electrolyte 1" The electrolytic solution produced in the item is used, and the battery is produced by the method in the item of "Battery Production 1". The battery was measured by the method described in the following item "Evaluation of Battery" and the above-mentioned measurement method.
负极[1]实施例2Negative electrode [1] Example 2
除了负极[1]实施例1的《负极的制作1》项的负极活性物质使用复合碳质物质(D)以外,同样地制作电池,并进行《电池的评价》项记载的电池评价。Except that the composite carbonaceous material (D) was used as the negative electrode active material in the item "Preparation of Negative Electrode 1" of the negative electrode [1] Example 1, a battery was produced in the same manner, and the battery evaluation described in the item "Battery Evaluation" was performed.
负极[1]实施例3Negative electrode [1] Example 3
除了负极[1]实施例1的《负极的制作1》项的负极活性物质使用复合碳质物质(E)以外,同样地制作电池,并进行《电池的评价》项记载的电池评价。Except that the composite carbonaceous material (E) was used as the negative electrode active material in the item "Preparation of Negative Electrode 1" of the negative electrode [1] Example 1, a battery was produced in the same manner, and the battery evaluation described in the item "Battery Evaluation" was performed.
负极[1]实施例4Negative electrode [1] Example 4
除了负极[1]实施例1的《负极的制作1》项的负极活性物质使用复合碳质物质(F)以外,同样地制作电池,并进行《电池的评价》项记载的电池评价。Except that the composite carbonaceous material (F) was used as the negative electrode active material in the item "Preparation of Negative Electrode 1" of the negative electrode [1] Example 1, a battery was produced in the same manner, and the battery evaluation described in the item "Battery Evaluation" was performed.
负极[1]实施例5Negative electrode [1] Example 5
除了负极[1]实施例1的《负极的制作1》项的负极活性物质使用复合碳质物质(G)以外,同样地制作电池,并进行《电池的评价》项记载的电池评价。Except that the composite carbonaceous material (G) was used as the negative electrode active material in the item "Preparation of Negative Electrode 1" of the negative electrode [1] Example 1, a battery was produced in the same manner, and the battery evaluation described in the item "Battery Evaluation" was performed.
负极[1]实施例6Negative pole [1] embodiment 6
除了负极[1]实施例1的《负极的制作1》项的负极活性物质使用复合碳质物质(J)以外,同样地制作电池,并进行《电池的评价》项记载的电池评价。Except that the composite carbonaceous material (J) was used as the negative electrode active material in the item "Preparation of Negative Electrode 1" of the negative electrode [1] Example 1, a battery was produced in the same manner, and the battery evaluation described in the item "Battery Evaluation" was performed.
负极[1]实施例7Negative electrode [1] Example 7
除了负极[1]实施例1的《负极的制作1》项的负极活性物质使用复合碳质物质(I)以外,同样地制作电池,并进行《电池的评价》项记载的电池评价。Except that the composite carbonaceous material (I) was used as the negative electrode active material in the negative electrode [1] Example 1 of "Making of Negative Electrode 1", a battery was produced in the same manner, and the battery evaluation described in the item "Battery Evaluation" was carried out.
负极[1]实施例8Negative pole [1] embodiment 8
除了负极[1]实施例1的《负极的制作1》项的负极活性物质使用复合碳质物质(M)以外,同样地制作电池,并进行《电池的评价》项记载的电池评价。Except that the composite carbonaceous material (M) was used as the negative electrode active material in the item "Preparation of Negative Electrode 1" of the negative electrode [1] Example 1, a battery was produced in the same manner, and the battery evaluation described in the item "Battery Evaluation" was performed.
负极[1]实施例9Negative electrode [1] Example 9
除了负极[1]实施例1的《负极的制作1》项的负极活性物质使用复合碳质物质(R)以外,同样地制作电池,并进行《电池的评价》项记载的电池评价。Except that the composite carbonaceous material (R) was used as the negative electrode active material in the item "Preparation of Negative Electrode 1" of the negative electrode [1] Example 1, a battery was produced in the same manner, and the battery evaluation described in the item "Battery Evaluation" was performed.
负极[1]实施例10Negative electrode [1] Example 10
除了负极[1]实施例1的《负极的制作1》项的负极活性物质使用复合碳质物质(L)以外,同样地制作电池,并进行《电池的评价》项记载的电池评价。Except that the composite carbonaceous material (L) was used as the negative electrode active material in the item "Preparation of Negative Electrode 1" of the negative electrode [1] Example 1, a battery was produced in the same manner, and the battery evaluation described in the item "Battery Evaluation" was performed.
负极[1]实施例11Negative electrode [1] Example 11
除了负极[1]实施例1的《负极的制作1》项的负极活性物质使用复合碳质物质(S)以外,同样地制作电池,并进行《电池的评价》项记载的电池评价。Except that the composite carbonaceous material (S) was used as the negative electrode active material in the item "Preparation of Negative Electrode 1" of the negative electrode [1] Example 1, a battery was produced in the same manner, and the battery evaluation described in the item "Evaluation of the Battery" was performed.
负极[1]实施例12Negative electrode [1] Example 12
除了负极[1]实施例1的《负极的制作1》项的负极活性物质使用复合碳质物质(O)以外,同样地制作电池,并进行《电池的评价》项记载的电池评价。Except that the composite carbonaceous material (O) was used as the negative electrode active material in the item "Preparation of Negative Electrode 1" of the negative electrode [1] Example 1, a battery was produced in the same manner, and the battery evaluation described in the item "Evaluation of the Battery" was performed.
负极[1]实施例13Negative electrode [1] Example 13
除了负极[1]实施例1的《负极的制作1》项的负极活性物质使用复合碳质物质(P)以外,同样地制作电池,并进行《电池的评价》项记载的电池评价。Except that the composite carbonaceous material (P) was used as the negative electrode active material in the item "Preparation of Negative Electrode 1" of the negative electrode [1] Example 1, a battery was produced in the same manner, and the battery evaluation described in the item "Battery Evaluation" was performed.
负极[1]实施例14Negative electrode [1] Example 14
除了负极[1]实施例1的《负极的制作1》项的负极活性物质使用复合碳质物质(Q)以外,同样地制作电池,并进行《电池的评价》项记载的电池评价。Except that the composite carbonaceous material (Q) was used as the negative electrode active material in the item "Preparation of Negative Electrode 1" of the negative electrode [1] Example 1, a battery was produced in the same manner, and the battery evaluation described in the item "Battery Evaluation" was performed.
负极[1]实施例15Negative electrode [1] Example 15
使用以《负极的制作2》项的负极活性物质作为复合碳质物质(D)而制成的负极、《正极的制作2》项中制作的正极和《电解液的制作1》项中制作的电解液,通过《电池的制作2》项中的方法制作电池。此外,与负极[1]实施例1同样地进行电池的评价。Use the negative electrode made of the negative electrode active material in "Making of Negative Electrode 2" as the composite carbonaceous material (D), the positive electrode made in "Making of Positive Electrode 2" and the one made in "Making of Electrolyte 1" Electrolyte, make a battery by the method in "Battery Production 2". In addition, evaluation of the battery was carried out in the same manner as in negative electrode [1] Example 1.
负极[1]实施例16Negative electrode [1] Example 16
使用以《负极的制作3》项的负极活性物质作为复合碳质物质(D)而制成的负极、《正极的制作1》项中制作的正极和《电解液的制作1》项中制作的电解液,通过《电池的制作1》项中的方法制作电池。此外,与负极[1]实施例1同样地进行电池的评价。Use the negative electrode made of the negative electrode active material in "Making of Negative Electrode 3" as a composite carbonaceous material (D), the positive electrode made in "Making of Positive Electrode 1" and the one made in "Making of Electrolyte 1" Electrolyte, the battery is made by the method in item "Battery Production 1". In addition, evaluation of the battery was carried out in the same manner as in negative electrode [1] Example 1.
负极[1]实施例17Negative electrode [1] Example 17
使用以《负极的制作4》项的负极活性物质作为复合碳质物质(D)而制成的负极、《正极的制作1》项中制作的正极和《电解液的制作1》项中制作的电解液,通过《电池的制作1》项中的方法制作电池。此外,与负极[1]实施例1同样地进行电池的评价。Use the negative electrode made of the negative electrode active material in "Making of Negative Electrode 4" as a composite carbonaceous material (D), the positive electrode made in "Making of Positive Electrode 1" and the one made in "Making of Electrolyte 1" Electrolyte, the battery is made by the method in item "Battery Production 1". In addition, evaluation of the battery was carried out in the same manner as in negative electrode [1] Example 1.
负极[1]实施例1~负极[1]实施例17的评价结果示于负极[1]表2。The evaluation results of Negative Electrode [1] Example 1 to Negative Electrode [1] Example 17 are shown in Negative Electrode [1] Table 2.
负极[1]实施例18~34Negative electrode [1] Examples 18-34
除了将负极[1]实施例1~17的电解液更换成《电解液的制作2》项制作的电解液以外,以同样的方法进行电池的评价。负极[1]实施例18~负极[1]实施例34的评价结果示于负极[1]表3。The evaluation of the battery was performed in the same manner except that the electrolytic solution in Examples 1 to 17 of the negative electrode [1] was replaced with the electrolytic solution produced in the item "Preparation of Electrolyte Solution 2". The evaluation results of negative electrode [1] Example 18 to negative electrode [1] Example 34 are shown in Table 3 of negative electrode [1].
负极[1]实施例35~51Negative electrode [1] Examples 35-51
除了将负极[1]实施例1~17的电解液更换成《电解液的制作3》项制作的电解液以外,以同样的方法进行电池的评价。负极[1]实施例35~负极[1]实施例51的评价结果示于负极[1]表4。The evaluation of the battery was performed in the same manner except that the electrolytic solution in Examples 1 to 17 of the negative electrode [1] was replaced with the electrolytic solution produced in the item "Preparation of Electrolyte Solution 3". The evaluation results of negative electrode [1] Example 35 to negative electrode [1] Example 51 are shown in Table 4 of negative electrode [1].
负极[1]比较例1Negative electrode [1] Comparative Example 1
除了负极[1]实施例1的《负极的制作1》项的负极活性物质使用碳质物质(A)以外,同样地制作电池,并进行《电池的评价》项记载的电池评价。A battery was produced in the same manner as the negative electrode [1] except that the carbonaceous material (A) was used as the negative electrode active material in the item "Preparation of Negative Electrode 1" of Example 1, and the battery evaluation described in the item "Evaluation of Battery" was performed.
负极[1]比较例2Negative electrode [1] Comparative example 2
除了负极[1]比较例1的电解液使用《电解液的制作4》项制作的电解液以外,同样地制作电池,并进行《电池的评价》项记载的电池评价。Except for the negative electrode [1] and the electrolyte solution of Comparative Example 1, the electrolyte solution prepared in the item "Preparation of Electrolyte Solution 4" was used, and a battery was produced in the same manner, and the battery evaluation described in the item "Evaluation of Battery" was performed.
负极[1]比较例3Negative electrode [1] Comparative example 3
除了负极[1]实施例1的《负极的制作1》项的负极活性物质使用碳质物质(B)以外,同样地制作电池,并进行《电池的评价》项记载的电池评价。A battery was produced in the same manner as the negative electrode [1] except that the carbonaceous material (B) was used as the negative electrode active material in the item "Preparation of Negative Electrode 1" of Example 1, and the battery evaluation described in the item "Evaluation of Battery" was performed.
负极[1]比较例4Negative electrode [1] Comparative example 4
除了负极[1]比较例3的电解液使用《电解液的制作4》项制作的电解液以外,与负极[1]比较例3同样地制作电池,并进行《电池的评价》项记载的电池评价。Except that the electrolyte solution of the negative electrode [1] Comparative Example 3 was prepared in the item "Preparation of Electrolyte Solution 4", a battery was produced in the same manner as the negative electrode [1] Comparative Example 3, and the battery described in the item "Evaluation of the Battery" was performed. evaluate.
负极[1]比较例5Negative electrode [1] Comparative example 5
除了负极[1]实施例1的电解液使用《电解液的制作4》项制作的电解液以外,与负极[1]实施例1同样地制作电池,并进行《电池的评价》项记载的电池评价。Except that the electrolyte solution of the negative electrode [1] Example 1 uses the electrolyte solution produced in the item "Preparation of Electrolyte Solution 4", a battery is produced in the same manner as the negative electrode [1] Example 1, and the battery described in the item "Evaluation of the Battery" is carried out. evaluate.
负极[1]比较例6Negative electrode [1] Comparative Example 6
除了负极[1]实施例2的电解液使用《电解液的制作4》项制作的电解液以外,同样地制作电池,并进行《电池的评价》项记载的电池评价。Except for the negative electrode [1], the electrolyte solution of Example 2 was used, except that the electrolyte solution prepared in the item "Preparation of Electrolyte Solution 4" was used, and a battery was produced in the same manner, and the battery evaluation described in the item "Evaluation of Battery" was performed.
负极[1]比较例7Negative electrode [1] Comparative Example 7
除了负极[1]实施例1的《负极的制作1》项的负极活性物质使用碳质物质(K)以外,同样地制作电池,并进行《电池的评价》项记载的电池评价。A battery was produced in the same manner as the negative electrode [1] except that the carbonaceous material (K) was used as the negative electrode active material in the item "Preparation of Negative Electrode 1" of Example 1, and the battery evaluation described in the item "Evaluation of Battery" was performed.
负极[1]比较例8Negative electrode [1] Comparative example 8
除了负极[1]比较例7的电解液使用《电解液的制作4》项制作的电解液以外,同样地制作电池,并进行《电池的评价》项记载的电池评价。A battery was produced in the same manner except that the electrolyte solution prepared in the item "Preparation of Electrolyte Solution 4" was used for the electrolyte solution of the negative electrode [1] Comparative Example 7, and the battery evaluation described in the item "Evaluation of Battery" was performed.
负极[1]比较例9~11Negative electrode [1] Comparative Examples 9-11
除了将负极[1]比较例1、3、7的电解液更换成《电解液的制作2》项制作的电解液以外,以同样的方法进行电池的评价。The evaluation of the battery was performed in the same manner except that the electrolytic solution of the negative electrode [1] Comparative Examples 1, 3, and 7 was replaced with the electrolytic solution prepared in the item "Preparation of Electrolyte Solution 2".
负极[1]比较例12~14Negative electrode [1] Comparative examples 12-14
除了将负极[1]比较例1、3、7的电解液更换成《电解液的制作3》项制作的电解液以外,以同样的方法进行电池的评价。The evaluation of the battery was performed in the same manner except that the electrolytic solution of the negative electrode [1] Comparative Examples 1, 3, and 7 was replaced with the electrolytic solution prepared in the item "Preparation of Electrolyte Solution 3".
负极[1]比较例1~负极[1]比较例14的评价结果示于负极[1]表5。The evaluation results of Negative Electrode [1] Comparative Example 1 to Negative Electrode [1] Comparative Example 14 are shown in Negative Electrode [1] Table 5.
负极[1]实施例52Negative electrode [1] Example 52
使用以《负极的制作1》项的负极活性物质作为复合碳质物质(D)而制成的负极、《正极的制作1》项中制作的正极以及《电解液的制作1》项中制作的电解液,通过《电池的制作1》项中的方法制作电池,进行《电池的评价》项中记载的电池评价。Use the negative electrode made of the negative electrode active material in "Making of Negative Electrode 1" as the composite carbonaceous material (D), the positive electrode made in "Making of Positive Electrode 1" and the one made in "Making of Electrolyte 1" As for the electrolyte solution, a battery was produced by the method in the item "Production of Battery 1", and the battery evaluation described in the item "Evaluation of Battery" was performed.
负极[1]实施例53Negative electrode [1] Example 53
除了电解液使用《电解液的制作2》项制作的电解液以外,与实施例52同样地制作电池,并进行《电池的评价》项记载的电池评价。A battery was fabricated in the same manner as in Example 52, except that the electrolyte solution prepared in the item "Preparation of Electrolyte Solution 2" was used, and the battery evaluation described in the item "Evaluation of Battery" was performed.
负极[1]实施例54Negative electrode [1] Example 54
除了电解液使用《电解液的制作3》项制作的电解液以外,与实施例52同样地制作电池,并进行《电池的评价》项记载的电池评价。A battery was produced in the same manner as in Example 52, except that the electrolyte solution prepared in the item "Preparation of Electrolyte Solution 3" was used, and the battery evaluation described in the item "Evaluation of Battery" was performed.
负极[1]比较例15Negative electrode [1] Comparative Example 15
除了电解液使用《电解液的制作4》项制作的电解液以外,与实施例52同样地制作电池,并进行《电池的评价》项记载的电池评价。A battery was fabricated in the same manner as in Example 52, except that the electrolyte solution prepared in the item "Preparation of Electrolyte Solution 4" was used, and the battery evaluation described in the item "Evaluation of Battery" was performed.
实施例52~54和比较例15的评价结果示于负极[1]表6。The evaluation results of Examples 52 to 54 and Comparative Example 15 are shown in Table 6 of Negative Electrode [1].
负极[1]《电池的评价》Negative electrode [1] "Battery Evaluation"
(容量测定)(capacity measurement)
对未经充放电循环的电池,在25℃、4.2V~3.0V的电压范围下,以0.2C的电流值(将1小时放电额定容量的电流值作为1C,所述额定容量取决于1小时率(one-hour-rate)的放电容量,下同)进行5个循环的初期充放电。将此时的第5个循环的0.2C放电容量作为初期容量。然后进行下面所示的输出功率测定。For a battery that has not been charged and discharged, at 25°C and a voltage range of 4.2V to 3.0V, with a current value of 0.2C (the current value of the rated capacity of 1 hour discharge is taken as 1C, and the rated capacity depends on 1 hour Rate (one-hour-rate) discharge capacity, the same below) for 5 cycles of initial charge and discharge. The 0.2C discharge capacity at the fifth cycle at this time was taken as the initial capacity. Then perform the output power measurement shown below.
(输出功率测定)(Output power measurement)
在25℃的环境下,通过0.2C的恒定电流进行150分钟充电,分别以0.1C、0.3C、1.0C、3.0C、10.0C放电10秒钟,测定第10秒的电压。将电流-电压直线和下限电压(3V)所包围的三角形的面积作为输出功率(W)。In an environment of 25°C, charge was performed for 150 minutes with a constant current of 0.2C, and discharge was performed at 0.1C, 0.3C, 1.0C, 3.0C, and 10.0C for 10 seconds, respectively, and the voltage at the 10th second was measured. The area of the triangle surrounded by the current-voltage line and the lower limit voltage (3V) was taken as the output power (W).
(循环试验)(cycle test)
在被视为锂二次电池的实际使用上限温度的60℃的高温环境下,进行循环试验。以2C恒定电流恒定电压法充电到充电上限电压4.2V后,以2C的恒定电流放电至放电终止电压3.0V,以此作为一个充放电循环,反复进行该循环,直至500个循环。对于循环试验结束后的电池,在25℃的环境下进行3个循环的充放电,以第3个循环的0.2C放电容量作为循环后容量。由循环之前测定的初期容量和循环试验结束后测定的循环后容量通过下述计算式求出循环保持率。The cycle test was performed in a high-temperature environment of 60° C., which is regarded as the upper limit temperature for practical use of lithium secondary batteries. After charging to the charging upper limit voltage of 4.2V by 2C constant current and constant voltage method, discharge to the end-of-discharge voltage of 3.0V with a constant current of 2C, which is regarded as a charge-discharge cycle, and this cycle is repeated until 500 cycles. For the battery after the cycle test, 3 cycles of charging and discharging were performed in an environment of 25°C, and the 0.2C discharge capacity of the third cycle was used as the capacity after the cycle. The cycle retention rate was obtained from the initial capacity measured before the cycle and the post-cycle capacity measured after the cycle test was completed by the following calculation formula.
循环保持率(%)=100×循环后容量/初期容量Cycle retention (%) = 100 × capacity after cycle / initial capacity
对循环试验结束后的电池进行(输出功率测定)项记载的输出功率测定。此时,使用循环试验前的输出功率和循环试验结束后实施的输出功率计算下式所示的输出功率保持率。The output power measurement described in the item (Output Power Measurement) was performed on the battery after the cycle test. At this time, the output power retention rate shown by the following formula was calculated using the output power before the cycle test and the output power implemented after the cycle test was completed.
输出功率保持率(%)=100×循环试验结束后输出功率/循环前输出功率Output power retention (%) = 100 × output power after cycle test / output power before cycle
负极[1]表2中的阻抗Rct和双层容量Cdl是对输出功率有贡献的参数之一,阻抗Rct的值越小,或者双层容量Cdl的值越大,则输出功率越有改善的倾向。另外,“阻抗Rct”和“双层容量Cdl”通过说明阻抗的部分所记载的方法求出。The impedance Rct and the double-layer capacity Cdl in Table 2 of the negative electrode [1] are one of the parameters that contribute to the output power. The smaller the value of the impedance Rct, or the larger the value of the double-layer capacity Cdl, the more the output power will be improved. tendency. In addition, "impedance Rct" and "double layer capacitance Cdl" were obtained by the method described in the section describing impedance.
负极[1]表2Negative pole [1] Table 2
[表33][Table 33]
负极[1]表2中,电解液含有0.3质量%的《电解液的制作1》中制作的二氟磷酸锂盐(LiPO2F2)。Negative electrode [1] In Table 2, the electrolytic solution contained 0.3% by mass of lithium difluorophosphate (LiPO 2 F 2 ) prepared in <<Preparation of Electrolyte Solution 1>>.
负极[1]表3Negative pole [1] Table 3
[表34][Table 34]
负极[1]表3中,电解液含有0.3质量%的《电解液的制作2》中制作的甲磺酸三甲基甲硅烷基酯。Negative electrode [1] In Table 3, the electrolytic solution contained 0.3% by mass of trimethylsilyl methanesulfonate prepared in <<Preparation of Electrolyte Solution 2>>.
负极[1]表4Negative pole [1] Table 4
[表35][Table 35]
负极[1]表4中,电解液含有0.3质量%的《电解液的制作3》中制作的六甲基环三硅氧烷。Negative electrode [1] In Table 4, the electrolytic solution contained 0.3% by mass of hexamethylcyclotrisiloxane prepared in <<Preparation of Electrolyte Solution 3>>.
负极[1]表5Negative pole [1] Table 5
[表36][Table 36]
负极[1]表6Negative electrode [1] Table 6
表[36]
由负极[1]表2~6的结果可知,通过将含有二氟磷酸锂盐、甲磺酸三甲基甲硅烷基酯、六甲基环三硅氧烷的情况和使负极活性物质含有2种以上结晶性不同的碳质物质的情况进行组合,可以使循环后的输出功率保持率得到飞跃性的提高。From the results of Tables 2 to 6 of the negative electrode [1], it can be seen that by combining the situation of containing difluorophosphate lithium salt, trimethylsilyl methanesulfonate, and hexamethylcyclotrisiloxane and making the negative electrode active material contain 2 The combination of more than one carbonaceous material with different crystallinity can dramatically improve the power retention rate after cycling.
负极[2][负极活性物质的制作]Negative electrode [2] [Production of negative electrode active material]
(负极活性物质的制作1)(Production of negative electrode active material 1)
在反应炉中于460℃下将不溶于喹啉的成分为0.05质量%以下的煤焦油沥青进行10小时热处理,使用粉碎机(seishin企业公司制造的orient mill)将得到的块状碳质物质进行粉碎,再使用微粉碎机(matsubo公司制造的涡轮式磨)进行微粉碎,微细化至中值粒径18μm。将该粒子装入到金属制造的容器中,在箱形的电炉中,氮气流通下于540℃进行2小时热处理。用粗粉碎机(吉田制作所制造的辊式压碎机)将得到的块状物质粉碎,再用微粉碎机(matsubo公司制造的涡轮式磨)进行微粉碎,将得到的粉末装入容器内,在氮气氛围下,用电炉在1000℃下烧制1小时。然后,对得到的烧结物进行分级处理,由此得到无定形碳质(A)。分级处理时,为了防止混入粗大粒子,使用ASTM400目的筛子反复筛5次。Coal tar pitch with a quinoline-insoluble component of 0.05% by mass or less was heat-treated in a reaction furnace for 10 hours at 460° C., and the resulting lumpy carbonaceous material was crushed using a pulverizer (orient mill manufactured by Seishin Enterprise Co., Ltd.). After crushing, finely pulverize using a pulverizer (turbine mill manufactured by Matsubo Co., Ltd.), and refine it to a median particle size of 18 μm. The particles were placed in a metal container, and heat-treated at 540° C. for 2 hours in a box-shaped electric furnace under nitrogen gas flow. The resulting lumpy material was pulverized with a coarse pulverizer (roller crusher manufactured by Yoshida Seisakusho), and finely pulverized with a fine pulverizer (turbine mill manufactured by Matsubo Co., Ltd.), and the obtained powder was filled into a container , under a nitrogen atmosphere, fired in an electric furnace at 1000°C for 1 hour. Then, the obtained sintered product is subjected to classification treatment to obtain an amorphous carbonaceous material (A). During the classification process, in order to prevent the mixing of coarse particles, use an ASTM400 mesh sieve to repeatedly sieve 5 times.
(负极活性物质的制作2)(Production of negative electrode active material 2)
在反应炉中于460℃下将不溶于喹啉的成分为0.05质量%以下的煤焦油沥青进行10小时热处理,将得到的块状物质装入容器内,在氮气氛围下,用箱形电炉在1000℃下进行2小时热处理。然后,对得到的烧结物进行分级处理,由此得到无定形碳质(A)。使用粗粉碎机(吉田制作所制造的辊式压碎机)将得到的无定形块状物质粉碎,再使用微粉碎机(matsubo公司制造的涡轮式磨)进行微粉碎,得到无定形粉末。为了防止得到的粉末中混入粗大粒子,使用ASTM400目的筛子反复筛5次。将这样得到的负极活性物质作为无定形碳质物质(B)。In a reaction furnace at 460° C., coal tar pitch with a quinoline-insoluble component of 0.05% by mass or less was heat-treated for 10 hours. Heat treatment was performed at 1000°C for 2 hours. Then, the obtained sintered product is subjected to classification treatment to obtain an amorphous carbonaceous material (A). The obtained amorphous lump was pulverized using a coarse pulverizer (roll crusher manufactured by Yoshida Seisakusho), and finely pulverized using a fine pulverizer (turbine mill manufactured by Matsubo Co., Ltd.) to obtain an amorphous powder. In order to prevent coarse particles from being mixed into the obtained powder, the sieve was repeatedly sieved 5 times using an ASTM400 mesh sieve. The negative electrode active material thus obtained is referred to as an amorphous carbonaceous material (B).
(负极活性物质的制作3)(Production of negative electrode active material 3)
将(负极活性物质的制作2)中得到的无定形块状物质再转移到石墨坩埚中,使用直接通电炉,在非活性气体氛围下,在2200℃下热处理5小时,用粗粉碎机(吉田制作所制造的辊式压碎机)将得到的块状物质粉碎,再使用微粉碎机(matsubo公司制造的涡轮式磨)进行微粉碎,为了防止得到的粉末中混入粗大粒子,使用ASTM400目的筛子反复筛5次。将这样得到的负极活性物质作为无定形碳质物质(C)。The amorphous bulk material obtained in (making of negative electrode active material 2) is transferred in the graphite crucible again, use direct electric furnace, under inert gas atmosphere, heat treatment at 2200 ℃ for 5 hours, with coarse pulverizer (Yoshida Roller crusher manufactured by Seisakusho Co., Ltd.) pulverizes the obtained lumpy material, and then finely pulverizes it with a fine pulverizer (turbine mill manufactured by Matsubo Co., Ltd.). In order to prevent coarse particles from being mixed into the obtained powder, an ASTM400 mesh sieve is used. Sieve repeatedly 5 times. The negative electrode active material thus obtained is referred to as an amorphous carbonaceous material (C).
(负极活性物质的制作4)(Production of negative electrode active material 4)
将(负极活性物质的制作2)中得到无定形块状物质再转移到石墨坩埚中,使用直接通电炉,在非活性气体氛围下,在3000℃下进行5个小时石墨化,用粗粉碎机(吉田制作所制造的辊式压碎机)将得到的块状物质粉碎,再使用微粉碎机(matsubo公司制造的涡轮式磨)进行微粉碎,为了防止得到的粉末中混入粗大粒子,使用ASTM400目的筛子反复筛5次。将这样得到的负极活性物质作为无定形碳质物质(D)。Transfer the amorphous bulk material obtained in (production of negative electrode active material 2) to a graphite crucible, use a direct electric furnace, and carry out graphitization at 3000°C for 5 hours under an inert gas atmosphere, and use a coarse pulverizer (Roller crusher manufactured by Yoshida Seisakusho Co., Ltd.) crushed the obtained lumpy material, and then finely pulverized it using a pulverizer (turbine mill manufactured by Matsubo Co., Ltd.). In order to prevent coarse particles from being mixed into the obtained powder, ASTM400 was used The objective sieve was repeatedly sieved 5 times. The negative electrode active material thus obtained is referred to as an amorphous carbonaceous material (D).
(负极活性物质的制作5)(Production of negative electrode active material 5)
为了防止市售的鳞片状天然石墨粉末中混入粗大粒子,使用ASTM400目的筛子反复筛5次。将这样得到的负极活性物质作为无定形碳质物质(E)。In order to prevent the commercially available flaky natural graphite powder from being mixed with coarse particles, it was repeatedly sieved 5 times using an ASTM400 mesh sieve. The negative electrode active material thus obtained is referred to as an amorphous carbonaceous material (E).
通过上述方法对负极活性物质的制作1~5中得到的负极活性物质测定物性、形状等。结果示于负极[2]表1中。The physical properties, shape, etc. of the negative electrode active materials obtained in Preparations 1 to 5 of the negative electrode active materials were measured by the above-mentioned method. The results are shown in Table 1 of the negative electrode [2].
负极[2]表1Negative electrode [2] Table 1
[表37][Table 37]
负极[2][电池的制作]Negative electrode [2] [Battery production]
《正极的制作1》"Making of Positive Pole 1"
在N-甲基吡咯烷酮溶剂中混合90质量%的作为正极活性物质的钴酸锂(LiCoO2)、5质量%的作为导电材料的乙炔黑和5质量%作为粘合剂的聚偏氟乙烯(PVdF),制成浆料。将得到的浆料涂布在厚度15μm的铝箔两面,并干燥,用压制机压延成厚度80μm,再将其切成具有宽100mm、长100mm大小的活性物质层和宽30mm的未涂布部分的形状,作为正极。此时的正极活性物质的密度为2.35g/cm3。90% by mass of lithium cobaltate (LiCoO 2 ) as a positive electrode active material, 5% by mass of acetylene black as a conductive material, and 5% by mass of polyvinylidene fluoride ( PVdF), made into slurry. The obtained slurry was coated on both sides of an aluminum foil with a thickness of 15 μm, dried, rolled to a thickness of 80 μm with a press, and cut into an active material layer having a width of 100 mm and a length of 100 mm and an uncoated portion of a width of 30 mm. shape, as the positive electrode. The density of the positive electrode active material at this time was 2.35 g/cm 3 .
《负极的制作1》"Making of Negative Pole 1"
在98重量份负极活性物质中添加100重量份作为增稠剂的羧甲基纤维素钠的水性分散液(羧甲基纤维素钠的浓度为1质量%)、2重量份作为粘合剂的苯乙烯-丁二烯橡胶的水性分散液(苯乙烯-丁二烯橡胶的浓度为50质量%),用分散器进行混合,制成浆料。将得到的浆料涂布在厚度10μm的压延铜箔两面,并干燥,用压制机压延成厚度75μm,再将其切成具有宽104mm、长104mm大小的活性物质层和宽30mm的未涂布部分的形状,作为负极。此时的负极活性物质密度为1.35g/cm3。Add 100 parts by weight of an aqueous dispersion of sodium carboxymethylcellulose as a thickener (the concentration of sodium carboxymethylcellulose is 1% by mass) and 2 parts by weight of sodium carboxymethylcellulose as a binder in 98 parts by weight of the negative electrode active material. An aqueous dispersion of styrene-butadiene rubber (the concentration of styrene-butadiene rubber is 50% by mass) was mixed with a disperser to prepare a slurry. The obtained slurry was coated on both sides of a rolled copper foil with a thickness of 10 μm, dried, rolled to a thickness of 75 μm with a press, and then cut into an active material layer with a width of 104 mm and a length of 104 mm and an uncoated layer with a width of 30 mm. The shape of the part, as the negative pole. The negative electrode active material density at this time was 1.35 g/cm 3 .
《非水电解液的制作1》"Preparation of non-aqueous electrolyte 1"
在干燥的氩氛围下,在碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的混合物(体积比3∶3∶4)中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。另外,还含有0.3质量%的二氟磷酸锂盐(LiPO2F2)。Under a dry argon atmosphere, in a mixture of ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) (volume ratio 3:3:4), at a concentration of 1mol/L Dissolve well-dried lithium hexafluorophosphate (LiPF 6 ). In addition, 0.3% by mass of lithium difluorophosphate (LiPO 2 F 2 ) was contained.
《非水电解液的制作2》"The production of non-aqueous electrolyte 2"
在干燥的氩氛围下,在碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的混合物(体积比3∶3∶4)中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。另外,还含有0.3质量%的甲磺酸三甲基甲硅烷基酯。Under a dry argon atmosphere, in a mixture of ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) (volume ratio 3:3:4), at a concentration of 1mol/L Dissolve well-dried lithium hexafluorophosphate (LiPF 6 ). In addition, 0.3% by mass of trimethylsilyl methanesulfonate was contained.
《非水电解液的制作3》"The Production of Non-aqueous Electrolyte 3"
在干燥的氩氛围下,在碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的混合物(体积比3∶3∶4)中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。另外,还含有0.3质量%的六甲基环三硅氧烷。Under a dry argon atmosphere, in a mixture of ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) (volume ratio 3:3:4), at a concentration of 1mol/L Dissolve well-dried lithium hexafluorophosphate (LiPF 6 ). In addition, 0.3% by mass of hexamethylcyclotrisiloxane was contained.
《非水电解液的制作4》"The Production of Non-aqueous Electrolyte 4"
在干燥的氩氛围下,在碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的混合物(体积比3∶3∶4)中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。Under a dry argon atmosphere, in a mixture of ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) (volume ratio 3:3:4), at a concentration of 1mol/L Dissolve well-dried lithium hexafluorophosphate (LiPF 6 ).
《电池的制作1》"Battery Making 1"
将32片正极和33片负极交替配置,各电极之间夹入多孔聚乙烯片隔板(厚度为25μm),进行叠层。此时,使正极活性物质面与负极活性物质面相对,并且不会露出负极活性物质面之外。将该正极和负极各自的未涂布部分焊接,制作集电片,将电极组密封在电池罐(外部尺寸:120×110×10mm)中。然后,在装有电极组的电池罐中注入20mL非水电解液,使电极充分浸透,密封而制作方形电池。该电池的额定放电容量约为6安培小时(Ah),以10kHz交流法测定的直流电阻成分约为5毫欧(mΩ)。正极的电极面积总和相对于电池外壳表面积总和之比为20.6。32 positive electrodes and 33 negative electrodes were arranged alternately, and a porous polyethylene sheet separator (thickness: 25 μm) was sandwiched between the electrodes for lamination. At this time, the surface of the positive electrode active material faces the surface of the negative electrode active material without protruding from the surface of the negative electrode active material. The respective uncoated parts of the positive and negative electrodes were welded to produce current collector sheets, and the electrode group was sealed in a battery can (outer dimensions: 120×110×10 mm). Then, 20 mL of non-aqueous electrolytic solution was poured into the battery can containing the electrode group, the electrodes were fully soaked, and sealed to produce a square battery. The rated discharge capacity of the battery is about 6 ampere hours (Ah), and the DC resistance component measured by the 10 kHz alternating current method is about 5 milliohms (mΩ). The ratio of the total electrode area of the positive electrode to the total surface area of the battery case was 20.6.
负极[2]实施例1Negative electrode [2] Embodiment 1
使用以《负极的制作1》项的负极活性物质作为无定形碳质(A)制成的负极,《正极的制作1》项中制作的正极以及《电解液的制作1》项中制作的电解液,通过《电池的制作1》项中的方法制作电池。通过下述《电池的评价》项中所述的方法对该电池进行电池评价。结果示于负极[2]表2。Using the negative electrode active material in "Making of Negative Electrode 1" as the negative electrode made of amorphous carbonaceous (A), the positive electrode made in "Making of Positive Electrode 1" and the electrolysis made in "Making of Electrolyte 1" liquid, and make a battery by the method in "Battery Production 1". The battery was subjected to battery evaluation by the method described in the item "Evaluation of Battery" below. The results are shown in Table 2 of the negative electrode [2].
负极[2]实施例2Negative pole [2] embodiment 2
除了负极[2]实施例1的《负极的制作1》项的负极活性物质使用无定形碳质(B)以外,同样地制作电池,并进行《电池的评价》项记载的电池评价。结果示于负极[2]表2。Except for using amorphous carbonaceous (B) as the negative electrode active material in the item "Preparation of Negative Electrode 1" of the negative electrode [2] Example 1, a battery was produced in the same manner, and the battery evaluation described in the item "Battery Evaluation" was performed. The results are shown in Table 2 of the negative electrode [2].
负极[2]实施例3Negative electrode [2] Example 3
除了负极[2]实施例1的《负极的制作1》项的负极活性物质使用无定形碳质(C)以外,同样地制作电池,并进行《电池的评价》项记载的电池评价。结果示于负极[2]表2。Except for using amorphous carbonaceous (C) as the negative electrode active material in the item "Preparation of Negative Electrode 1" of the negative electrode [2] Example 1, a battery was produced in the same manner, and the battery evaluation described in the item "Battery Evaluation" was performed. The results are shown in Table 2 of the negative electrode [2].
负极[2]实施例4~6Negative pole [2] Embodiment 4~6
除了将负极[2]实施例1~2的非水电解液换成《非水电解液的制作2》项中制作的非水电解液以外,分别用同样的方法制作电池,并进行评价。结果示于负极[2]表2。Except that the non-aqueous electrolyte solution of Examples 1 to 2 of the negative electrode [2] was replaced with the non-aqueous electrolyte solution produced in the item "Preparation of Non-aqueous Electrolyte Solution 2", batteries were produced and evaluated in the same manner. The results are shown in Table 2 of the negative electrode [2].
负极[2]实施例7~9Negative electrode [2] Examples 7-9
除了将负极[2]实施例1~3的非水电解液换成《非水电解液的制作3》项中制作的非水电解液以外,分别用同样的方法制作电池,并进行评价。结果示于负极[2]表2。Except that the non-aqueous electrolyte solution of Examples 1 to 3 of the negative electrode [2] was replaced with the non-aqueous electrolyte solution produced in the item "Preparation of Non-aqueous Electrolyte Solution 3", batteries were produced and evaluated in the same manner. The results are shown in Table 2 of the negative electrode [2].
负极[2]比较例1~3Negative electrode [2] Comparative examples 1 to 3
除了将负极[2]比较例1~3的非水电解液换成《非水电解液的制作4》项中制作的非水电解液以外,分别用同样的方法制作电池,并进行评价。结果示于负极[2]表2。Batteries were produced and evaluated in the same manner, except that the non-aqueous electrolyte solutions of Comparative Examples 1 to 3 of the negative electrode [2] were replaced with the non-aqueous electrolyte solutions produced in the item "Preparation of Non-aqueous Electrolyte Solutions 4". The results are shown in Table 2 of the negative electrode [2].
负极[2]比较例4Negative electrode [2] Comparative example 4
除了负极[2]实施例1的《负极的制作1》项的负极活性物质使用石墨质碳质(D)以外,同样地制作电池,并进行《电池的评价》项记载的电池评价。结果示于负极[2]表2。Except for negative electrode [2] Example 1 "Preparation of Negative Electrode 1" negative electrode active material using graphite carbonaceous (D), similarly produced a battery, and carried out the battery evaluation described in "Battery Evaluation". The results are shown in Table 2 of the negative electrode [2].
负极[2]比较例5Negative electrode [2] Comparative Example 5
除了负极[2]实施例1的《负极的制作1》项的负极活性物质使用石墨质碳质物质(E)以外,同样地制作电池,并进行《电池的评价》项记载的电池评价。结果示于负极[2]表2。Except for using the graphite carbonaceous material (E) as the negative electrode active material in the item "Preparation of Negative Electrode 1" of the negative electrode [2] Example 1, a battery was produced in the same manner, and the battery evaluation described in the item "Battery Evaluation" was performed. The results are shown in Table 2 of the negative electrode [2].
负极[2]比较例6~7Negative electrode [2] Comparative examples 6-7
除了将负极[2]比较例4~5的非水电解液换成《非水电解液的制作2》项中制作的非水电解液以外,分别用同样的方法制作电池,并进行评价。结果示于负极[2]表2。Batteries were produced and evaluated in the same manner, except that the non-aqueous electrolyte solutions of Comparative Examples 4 to 5 of the negative electrode [2] were replaced with the non-aqueous electrolyte solutions produced in the item "Preparation of Non-aqueous Electrolyte Solutions 2". The results are shown in Table 2 of the negative electrode [2].
负极[2]比较例8~9Negative electrode [2] Comparative Examples 8-9
除了将负极[2]比较例4~5的非水电解液换成《非水电解液的制作3》项中制作的非水电解液以外,分别用同样的方法制作电池,并进行评价。结果示于负极[2]表2。Batteries were produced and evaluated in the same manner, except that the non-aqueous electrolyte solutions of Comparative Examples 4 to 5 of the negative electrode [2] were replaced with the non-aqueous electrolyte solutions produced in the item "Preparation of Non-aqueous Electrolyte Solutions 3". The results are shown in Table 2 of the negative electrode [2].
负极[2]比较例10~11Negative electrode [2] Comparative examples 10-11
除了将负极[2]比较例4~5的非水电解液换成《非水电解液的制作4》项中制作的非水电解液以外,分别用同样的方法制作电池,并进行评价。结果示于负极[2]表2。Batteries were produced and evaluated in the same manner, except that the non-aqueous electrolyte solutions of Comparative Examples 4 to 5 of the negative electrode [2] were replaced with the non-aqueous electrolyte solutions produced in the item "Preparation of Non-aqueous Electrolyte Solutions 4". The results are shown in Table 2 of the negative electrode [2].
负极[2]《电池的评价》Negative electrode [2] "Battery Evaluation"
(容量测定)(capacity measurement)
对未经充放电循环的新电池,在25℃、4.1V~3.0V的电压范围下进行5个循环的初期充放电(电压范围4.1V~3.0V)。此时的第5个循环的0.2(将1小时放电额定容量的电流值作为1C,所述额定容量取决于1小时率(one-hour-rate)的放电容量,下同)C放电容量作为初期容量。For a new battery that has not undergone charge-discharge cycles, 5 cycles of initial charge-discharge (voltage range 4.1V-3.0V) were performed at 25°C and a voltage range of 4.1V-3.0V. At this time, the 0.2 C discharge capacity of the fifth cycle (the current value of the 1-hour discharge rated capacity is taken as 1C, and the rated capacity depends on the discharge capacity of the 1-hour-rate (one-hour-rate), the same below) C discharge capacity is taken as the initial stage capacity.
(短时间高电流密度充放电特性试验)(Short time high current density charge and discharge characteristics test)
在室温环境25℃下,用0.2C的恒定电流对进行了容量测定后的电池进行150分钟充电。以此时的电压为中心,以10C的高负荷电流向充电方向、放电方向均通电约10秒钟,连续反复进行包含间歇时间在内的35秒一个循环的试验。在第10万个循环的时刻取出电池,以0.2C的电流放电至3V,通过与(容量测定)项同样的方法进行1循环,作为循环后容量。并且,短时间高电流密度充放电特性通过下式计算。The battery after the capacity measurement was charged for 150 minutes at a constant current of 0.2 C at a room temperature environment of 25° C. Taking the voltage at this time as the center, a high load current of 10C was applied to both the charging direction and the discharging direction for about 10 seconds, and the test was continuously repeated for a cycle of 35 seconds including the intermittent time. The battery was taken out at the time of the 100,000th cycle, discharged to 3V with a current of 0.2C, and one cycle was performed by the same method as in the item (capacity measurement), and the capacity after cycle was taken. Also, the short-time high current density charge and discharge characteristics were calculated by the following equation.
[短时间高电流密度充放电特性]=100×[循环后容量]/[初期容量][Short-time high current density charge-discharge characteristics] = 100×[Capacity after cycle]/[Initial capacity]
负极[2]表2Negative electrode [2] Table 2
[表38][Table 38]
由负极[2]表2的结果可知,通过含有二氟磷酸锂盐、甲磺酸三甲基甲硅烷基酯、六甲基环三硅氧烷,并且含有广角X射线衍射法测定的(002)面的面间距(d002)为0.337以上、Lc为80nm以下、且拉曼R值为0.2以上的无定形碳质作为负极活性物质,可以使短时间高电流密度充放电特性得以飞跃性地提高。From the results of negative pole [2] table 2, it can be seen that by containing lithium difluorophosphate, trimethylsilyl methanesulfonate, hexamethylcyclotrisiloxane, and containing (002 ) surface spacing (d002) of 0.337 or more, Lc of 80nm or less, and Raman R value of 0.2 or more amorphous carbon as the negative electrode active material can dramatically improve short-term high current density charge and discharge characteristics .
负极[3][负极活性物质的制作]Negative electrode [3] [Production of negative electrode active material]
(负极活性物质的制作1)(Production of negative electrode active material 1)
为了防止混入粗大粒子,使用ASTM400目的筛子将市售的体积平均粒径23μm的Li1.33Ti1.66O4反复筛5次,得到锂钛复合氧化物(A)。In order to prevent mixing of coarse particles, commercially available Li 1.33 Ti 1.66 O 4 with a volume average particle diameter of 23 μm was repeatedly sieved five times using an ASTM 400 mesh sieve to obtain a lithium-titanium composite oxide (A).
(负极活性物质的制作2)(Production of negative electrode active material 2)
为了防止混入粗大粒子,使用ASTM400目的筛子将市售的体积平均粒径1.0μm的Li1.33Ti1.66O4反复筛5次,得到锂钛复合氧化物(B)。In order to prevent mixing of coarse particles, commercially available Li 1.33 Ti 1.66 O 4 with a volume average particle diameter of 1.0 μm was repeatedly sieved five times using an ASTM 400 mesh sieve to obtain a lithium-titanium composite oxide (B).
(负极活性物质的制作3)(Production of negative electrode active material 3)
为了防止混入粗大粒子,使用ASTM400目的筛子将市售的体积平均粒径0.1μm的Li1.33Ti1.66O4反复筛5次,得到锂钛复合氧化物(C)。In order to prevent mixing of coarse particles, commercially available Li 1.33 Ti 1.66 O 4 with a volume average particle diameter of 0.1 μm was repeatedly sieved five times using an ASTM400 mesh sieve to obtain a lithium-titanium composite oxide (C).
(负极活性物质的制作4)(Production of negative electrode active material 4)
为了防止市售的鳞片状天然石墨粉末中混入粗大粒子,使用ASTM400目的筛子反复筛5次。将这样得到的负极活性物质作为石墨质碳质(D)。In order to prevent the commercially available flaky natural graphite powder from being mixed with coarse particles, it was repeatedly sieved 5 times using an ASTM400 mesh sieve. The negative electrode active material thus obtained is referred to as graphitic carbon (D).
负极活性物质的组成、结构、形状、物性等归纳示于负极[3]表1中。The composition, structure, shape, and physical properties of the negative electrode active material are summarized in Table 1 of the negative electrode [3].
负极[3]表1Negative pole [3] Table 1
[表39][Table 39]
负极[3][电池的制作]Negative electrode [3] [Battery production]
《正极的制作1》"Making of Positive Pole 1"
在N-甲基吡咯烷酮溶剂中混合90质量%的作为正极活性物质的钴酸锂(LiCoO2)、5质量%的作为导电材料的乙炔黑和5质量%作为粘合剂的聚偏氟乙烯(PVdF),制成浆料。将得到的浆料涂布在厚度15μm的铝箔两面,并干燥,用压制机压延成厚度80μm,再将其切成具有宽100mm、长100mm大小的活性物质层和宽30mm的未涂布部分的形状,作为正极。此时的正极的活性物质的密度为2.35g/cm3。90% by mass of lithium cobaltate (LiCoO 2 ) as a positive electrode active material, 5% by mass of acetylene black as a conductive material, and 5% by mass of polyvinylidene fluoride ( PVdF), made into slurry. The obtained slurry was coated on both sides of an aluminum foil with a thickness of 15 μm, dried, rolled to a thickness of 80 μm with a press, and cut into an active material layer having a width of 100 mm and a length of 100 mm and an uncoated portion of a width of 30 mm. shape, as the positive electrode. The density of the active material of the positive electrode at this time was 2.35 g/cm 3 .
《负极的制作1》"Making of Negative Pole 1"
在N-甲基吡咯烷酮溶剂中混合90质量%负极活性物质、5质量%的作为导电材料的乙炔黑和5质量%作为粘合剂的聚偏氟乙烯(PVdF),制成浆料。将得到的浆料涂布在厚度10μm的压延铜箔的一面,并干燥,用压制机压延成厚度90μm,再将其切成具有宽104mm、长104mm大小的活性物质层和宽30mm的未涂布部分的形状,作为负极。A slurry was prepared by mixing 90% by mass of a negative electrode active material, 5% by mass of acetylene black as a conductive material, and 5% by mass of polyvinylidene fluoride (PVdF) as a binder in an N-methylpyrrolidone solvent. The obtained slurry is coated on one side of a rolled copper foil with a thickness of 10 μm, dried, and rolled to a thickness of 90 μm with a press, and then cut into an active material layer with a width of 104 mm and a length of 104 mm and an uncoated layer with a width of 30 mm. The shape of the cloth part is used as the negative electrode.
《非水电解液的制作1》"Preparation of non-aqueous electrolyte 1"
在干燥的氩氛围下,在碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的混合物(体积比3∶3∶4)中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。另外,还含有0.3质量%的二氟磷酸锂盐(LiPO2F2)。Under a dry argon atmosphere, in a mixture of ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) (volume ratio 3:3:4), at a concentration of 1mol/L Dissolve well-dried lithium hexafluorophosphate (LiPF 6 ). In addition, 0.3% by mass of lithium difluorophosphate (LiPO 2 F 2 ) was contained.
《非水电解液的制作2》"The production of non-aqueous electrolyte 2"
在干燥的氩氛围下,在碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的混合物(体积比3∶3∶4)中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。另外,还含有0.3质量%的甲磺酸三甲基甲硅烷基酯。Under a dry argon atmosphere, in a mixture of ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) (volume ratio 3:3:4), at a concentration of 1mol/L Dissolve well-dried lithium hexafluorophosphate (LiPF 6 ). In addition, 0.3% by mass of trimethylsilyl methanesulfonate was contained.
《非水电解液的制作3》"The Production of Non-aqueous Electrolyte 3"
在干燥的氩氛围下,在碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的混合物(体积比3∶3∶4)中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。另外,还含有0.3质量%的六甲基环三硅氧烷。Under a dry argon atmosphere, in a mixture of ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) (volume ratio 3:3:4), at a concentration of 1mol/L Dissolve well-dried lithium hexafluorophosphate (LiPF 6 ). In addition, 0.3% by mass of hexamethylcyclotrisiloxane was contained.
《非水电解液的制作4》"The Production of Non-aqueous Electrolyte 4"
在干燥的氩氛围下,在碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的混合物(体积比3∶3∶4)中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。Under a dry argon atmosphere, in a mixture of ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) (volume ratio 3:3:4), at a concentration of 1mol/L Dissolve well-dried lithium hexafluorophosphate (LiPF 6 ).
(电池的制作1)(Battery production 1)
将负极和正极冲孔成12.5mmφ,在110℃下进行真空干燥后,转移到手套工作箱内,在氩氛围下将正极和负极夹着冲孔成14mmφ的聚乙烯隔板对置,并加入非水电解液的制作项中记载的非水电解液,制作2032型硬币电池(锂二次电池)。Punch the negative electrode and the positive electrode into 12.5mmφ, vacuum dry at 110°C, transfer to the glove box, and place the positive and negative electrodes opposite each other with a polyethylene separator punched into 14mmφ under the argon atmosphere, and add A 2032-type coin battery (lithium secondary battery) was produced using the nonaqueous electrolyte solution described in the section on preparation of the nonaqueous electrolyte solution.
负极[3]实施例1Negative pole [3] embodiment 1
使用《负极的制作1》项的负极活性物质为锂钛复合氧化物(A)而制作的负极、《正极的制作1》项中制作的正极以及《非水电解液的制作1》项中制作的电解液,通过《电池的制作1》项中的方法制作电池。对该电池进行下述《电池的评价》项中所述的电池评价。结果示于负极[3]表2。The negative electrode produced by using lithium-titanium composite oxide (A) as the negative electrode active material in the item "Production of Negative Electrode 1", the positive electrode produced in the item "Production of Positive Electrode 1", and the item produced in "Production of Non-aqueous Electrolyte 1" Electrolyte, make a battery by the method in "Battery Production 1". The batteries were subjected to the battery evaluations described in the item "Evaluation of Batteries" below. The results are shown in Table 2 of the negative electrode [3].
负极[3]实施例2Negative pole [3] embodiment 2
除了负极[3]实施例1的《负极的制作1》项的负极活性物质使用锂钛复合氧化物(B)以外,与负极[3]实施例1同样地制作电池,并进行《电池的评价》项记载的电池评价。结果示于负极[3]表2。Except that the negative electrode active material of negative electrode [3] Example 1 of "Making of Negative Electrode 1" uses lithium titanium composite oxide (B), a battery is produced in the same manner as in negative electrode [3] Example 1, and "Battery Evaluation" "Battery evaluation recorded in item. The results are shown in Table 2 of the negative electrode [3].
负极[3]实施例3Negative electrode [3] Example 3
除了负极[3]实施例1的《负极的制作1》项的负极活性物质使用锂钛复合氧化物(C)以外,与负极[3]实施例1同样地制作电池,并进行《电池的评价》项记载的电池评价。结果示于负极[3]表2。Except that the negative electrode active material of negative electrode [3] Example 1 of "Making of Negative Electrode 1" uses lithium-titanium composite oxide (C), a battery is produced in the same manner as negative electrode [3] Example 1, and "Battery Evaluation" "Battery evaluation recorded in item. The results are shown in Table 2 of the negative electrode [3].
负极[3]实施例4~6Negative pole [3] Embodiment 4~6
除了将负极[3]实施例1~3的非水电解液换成《非水电解液的制作2》项中制作的非水电解液以外,分别用同样的方法制作电池,并进行评价。结果示于负极[3]表2。Except that the non-aqueous electrolyte solution of Examples 1 to 3 of the negative electrode [3] was replaced with the non-aqueous electrolyte solution produced in the item "Preparation of Non-aqueous Electrolyte Solution 2", batteries were produced and evaluated in the same manner. The results are shown in Table 2 of the negative electrode [3].
负极[3]实施例7~9Negative electrode [3] Examples 7-9
除了将负极[3]实施例1~3的非水电解液换成《非水电解液的制作3》项中制作的非水电解液以外,分别用同样的方法制作电池,并进行评价。结果示于负极[3]表2。Except that the non-aqueous electrolyte solution of Examples 1 to 3 of the negative electrode [3] was replaced with the non-aqueous electrolyte solution produced in the item "Preparation of Non-aqueous Electrolyte Solution 3", batteries were produced and evaluated in the same manner. The results are shown in Table 2 of the negative electrode [3].
负极[3]比较例1~3Negative electrode [3] Comparative examples 1 to 3
除了将负极[3]实施例1~3的非水电解液换成《非水电解液的制作4》项中制作的非水电解液以外,分别用同样的方法制作电池,并进行评价。结果示于负极[3]表2。Except that the non-aqueous electrolyte solution of Examples 1 to 3 of the negative electrode [3] was replaced with the non-aqueous electrolyte solution produced in the item "Preparation of Non-aqueous Electrolyte Solution 4", batteries were produced and evaluated in the same manner. The results are shown in Table 2 of the negative electrode [3].
负极[3]比较例4Negative electrode [3] Comparative example 4
除了负极[3]实施例1的《负极的制作1》项的负极活性物质使用石墨质碳质(D)以外,与负极[3]实施例1同样地制作电池,并进行《电池的评价》项记载的电池评价。结果示于负极[3]表2。Except that the negative electrode active material of negative electrode [3] Example 1 of "Making of Negative Electrode 1" uses graphitic carbonaceous (D), a battery is produced in the same manner as negative electrode [3] Example 1, and "Battery Evaluation" is carried out Item recorded battery evaluation. The results are shown in Table 2 of the negative electrode [3].
负极[3]比较例5Negative electrode [3] Comparative example 5
除了将负极[3]比较例4的非水电解液换成《非水电解液的制作2》项中制作的非水电解液以外,分别用同样的方法制作电池,并进行评价。结果示于负极[3]表2。Except that the non-aqueous electrolyte solution of the negative electrode [3] Comparative Example 4 was replaced with the non-aqueous electrolyte solution produced in the item "Preparation of Non-aqueous Electrolyte Solution 2", batteries were produced and evaluated in the same manner. The results are shown in Table 2 of the negative electrode [3].
负极[3]比较例6Negative electrode [3] Comparative example 6
除了将负极[3]比较例4的非水电解液换成《非水电解液的制作3》项中制作的非水电解液以外,分别用同样的方法制作电池,并进行评价。结果示于负极[3]表2。Except that the non-aqueous electrolyte solution of the negative electrode [3] Comparative Example 4 was replaced with the non-aqueous electrolyte solution produced in the item "Preparation of Non-aqueous Electrolyte Solution 3", batteries were produced and evaluated in the same manner. The results are shown in Table 2 of the negative electrode [3].
负极[3]比较例7Negative electrode [3] Comparative example 7
除了将负极[3]比较例4的非水电解液换成《非水电解液的制作4》项中制作的非水电解液以外,分别用同样的方法制作电池,并进行评价。结果示于负极[3]表2。Except that the non-aqueous electrolyte solution in Comparative Example 4 of the negative electrode [3] was replaced with the non-aqueous electrolyte solution produced in the item "Preparation of Non-aqueous Electrolyte Solution 4", batteries were produced and evaluated in the same manner. The results are shown in Table 2 of the negative electrode [3].
负极[3]《电池的评价》Negative electrode [3] "Battery Evaluation"
(容量的测定)(measurement of capacity)
对未经充放电循环的新电池,以锂钛复合氧化物为175mAh/g、石墨质碳质为350mAh/g换算,由存在于铜箔上的活性物质量计算出电池容量。并且,以该电池容量为基准在25℃、2.7V~1.9V的电压范围下,以0.2C(将1小时放电额定容量的电流值作为1C,所述额定容量取决于1小时率(one-hour-rate)的放电容量,下同)对锂钛复合氧化物进行5个循环的初期充放电。同样地在25℃、4.1V~3.0V的电压范围下对石墨质碳质进行初期充放电。将此时的第5个循环的0.2C放电容量作为初期容量。For a new battery that has not been charged and discharged, the lithium-titanium composite oxide is 175mAh/g, and the graphite carbon is 350mAh/g, and the battery capacity is calculated from the amount of active material present on the copper foil. And, based on the battery capacity, at 25°C, in the voltage range of 2.7V to 1.9V, at 0.2C (the current value of the rated capacity of 1 hour discharge is taken as 1C, and the rated capacity depends on the rate of 1 hour (one- hour-rate) of the discharge capacity, the same below) for the initial charge and discharge of lithium-titanium composite oxide for 5 cycles. Similarly, the graphitic carbon was initially charged and discharged at 25° C. and in a voltage range of 4.1 V to 3.0 V. The 0.2C discharge capacity at the fifth cycle at this time was taken as the initial capacity.
(输出电阻的测定)(measurement of output resistance)
在25℃的环境下,通过0.2C的恒定电流进行150分钟充电,在-30℃的环境下分别以0.1C、0.3C、1.0C、3.0C、10.0C放电10秒钟,测定第10秒的电压。将电流-电压直线的斜率作为输出电阻(Ω),其结果示于负极[3]表2中。In an environment of 25°C, charge with a constant current of 0.2C for 150 minutes, and discharge at 0.1C, 0.3C, 1.0C, 3.0C, and 10.0C for 10 seconds in an environment of -30°C, and measure the 10th second voltage. The slope of the current-voltage line is taken as the output resistance (Ω), and the results are shown in Table 2 of the negative electrode [3].
负极[3]表2Negative electrode [3] Table 2
[表40][Table 40]
负极[3]表2中,“输出电阻减少率”是与不含特定化合物的对应的电池的输出电阻进行比较而得到的输出电阻的减少率(%)。In Table 2 of the negative electrode [3], the "output resistance reduction rate" is the reduction rate (%) of the output resistance compared with the output resistance of the corresponding battery not containing the specific compound.
由负极[3]表2的结果可知,通过使用含有二氟磷酸锂盐、甲磺酸三甲基甲硅烷基酯、六甲基环三硅氧烷,并且含有包含能够吸留、放出锂的钛的金属氧化物的负极活性物质,可以飞跃性地减少输出电阻。From the results in Table 2 of the negative electrode [3], it can be seen that by using lithium difluorophosphate, trimethylsilyl methanesulfonate, and hexamethylcyclotrisiloxane, and containing Titanium metal oxide negative electrode active material can dramatically reduce output resistance.
负极[4][负极活性物质的制作]Negative electrode [4] [Production of negative electrode active material]
(负极活性物质的制作1)(Production of negative electrode active material 1)
使用球形化处理装置(奈良机械制作所公司制造的混合系统),以转速6500rpm对高纯度化处理过的中值粒径约150μm的鳞片状天然石墨(灰分0.05重量%)进行5分钟球形化处理,再使用风力式分级机(seishin企业公司制造的OMC-100)除去45重量%微粉,得到球形化天然石墨(C)。Using a spheroidizing treatment device (mixing system manufactured by Nara Machinery Manufacturing Co., Ltd.), the highly purified scaly natural graphite (ash content 0.05% by weight) with a median diameter of about 150 μm was spheroidized at a rotation speed of 6500 rpm for 5 minutes. , 45% by weight of fine powder was removed using a wind-type classifier (OMC-100 manufactured by Seishin Enterprise Co., Ltd.) to obtain spheroidized natural graphite (C).
(负极活性物质的制作2)(Production of negative electrode active material 2)
将上述分级后的球形化天然石墨(C)的粉末装到石墨坩埚中,使用直接通电炉在非活性氛围下于3000℃热处理5小时,得到碳质物质(D)。The powder of the above classified spheroidized natural graphite (C) was charged into a graphite crucible, and heat-treated at 3000° C. for 5 hours in an inert atmosphere using a direct electric furnace to obtain a carbonaceous material (D).
(负极活性物质的制作3)(Production of negative electrode active material 3)
除了将(负极活性物质的制作2)中的热处理温度设定为2000℃以外,同样地得到碳质物质(E)。A carbonaceous material (E) was obtained in the same manner except that the heat treatment temperature in (Preparation 2 of the negative electrode active material) was set to 2000°C.
(负极活性物质的制作4)(Production of negative electrode active material 4)
除了将(负极活性物质的制作2)中的热处理温度设定为1600℃以外,同样地得到碳质物质(F)。A carbonaceous material (F) was obtained in the same manner except that the heat treatment temperature in (Preparation 2 of negative electrode active material) was set to 1600°C.
(负极活性物质的制作5)(Production of negative electrode active material 5)
除了将(负极活性物质的制作2)中的热处理温度设定为1200℃以外,同样地得到碳质物质(G)。A carbonaceous material (G) was obtained in the same manner except that the heat treatment temperature in (Preparation 2 of negative electrode active material) was set to 1200°C.
(负极活性物质的制作5)(Production of negative electrode active material 5)
将高纯度化处理过的中值粒径17μm、振实密度0.5g/cm3、BET比表面积6m2/g的鳞片状天然石墨(灰分0.1重量%)直接与(负极活性物质的制作2)同样地进行热处理而不进行球形化处理,得到热处理天然石墨(H)。Scale-like natural graphite (ash content 0.1% by weight) that has been highly purified with a median particle size of 17 μm, a tap density of 0.5 g/cm 3 , and a BET specific surface area of 6 m 2 /g is directly mixed with (production of negative electrode active material 2) In the same manner, heat treatment is performed without performing spheroidization treatment to obtain heat-treated natural graphite (H).
(负极活性物质的制作6)(Production of negative electrode active material 6)
将高纯度化处理过的中值粒径20μm、振实密度0.75g/cm3、BET比表面积3m2/g的天然石墨(灰分0.5重量%)直接与(负极活性物质的制作2)同样地进行热处理而不进行球形化处理,得到碳质物质(I)。The highly purified natural graphite (ash content 0.5% by weight) with a median particle size of 20 μm, a tap density of 0.75 g/cm 3 , and a BET specific surface area of 3 m 2 /g was directly carried out in the same manner as in (production of negative electrode active material 2) The heat treatment is performed without the spheroidization treatment to obtain the carbonaceous substance (I).
按照上述的方法,测定制作的球形化天然石墨(C)、碳质物质(D)、碳质物质(E)、碳质物质(F)、碳质物质(G)、热处理天然石墨(H)、碳质物质(I)的形状和物性。结果示于负极[4]表1。According to the above-mentioned method, the spheroidized natural graphite (C), carbonaceous substance (D), carbonaceous substance (E), carbonaceous substance (F), carbonaceous substance (G) and heat-treated natural graphite (H) were measured. , The shape and physical properties of the carbonaceous substance (I). The results are shown in Table 1 of the negative electrode [4].
负极[4]表1Negative pole [4] Table 1
负极[4][电池的制作]Negative electrode [4] [Battery production]
《正极的制作1》"Making of Positive Pole 1"
在N-甲基吡咯烷酮溶剂中混合90质量%的作为正极活性物质的钴酸锂(LiCoO2)、5质量%的作为导电材料的乙炔黑和5质量%作为粘合剂的聚偏氟乙烯(PVdF),制成浆料。将得到的浆料涂布在厚度15μm的铝箔两面,并干燥,用压制机压延成厚度80μm,再将其切成具有宽100mm、长100mm大小的活性物质层和宽30mm的未涂布部分的形状,作为正极。此时的正极的活性物质的密度为2.35g/cm3。90% by mass of lithium cobaltate (LiCoO 2 ) as a positive electrode active material, 5% by mass of acetylene black as a conductive material, and 5% by mass of polyvinylidene fluoride ( PVdF), made into slurry. The obtained slurry was coated on both sides of an aluminum foil with a thickness of 15 μm, dried, rolled to a thickness of 80 μm with a press, and cut into an active material layer having a width of 100 mm and a length of 100 mm and an uncoated portion of a width of 30 mm. shape, as the positive electrode. The density of the active material of the positive electrode at this time was 2.35 g/cm 3 .
《负极的制作1》"Making of Negative Pole 1"
在98重量份负极活性物质中添加100重量份作为增稠剂的羧甲基纤维素钠的水性分散液(羧甲基纤维素钠的浓度为1质量%)、2重量份作为粘合剂的苯乙烯-丁二烯橡胶的水性分散液(苯乙烯-丁二烯橡胶的浓度为50质量%),用分散器进行混合,制成浆料。将得到的浆料涂布在厚度10μm的压延铜箔两面,并干燥,用压制机压延成厚度75μm,再将其切成具有宽104mm、长104mm大小的活性物质层和宽30mm的未涂布部分的形状,作为负极。此时的负极的活性物质的密度为1.35g/cm3。Add 100 parts by weight of an aqueous dispersion of sodium carboxymethylcellulose as a thickener (the concentration of sodium carboxymethylcellulose is 1% by mass) and 2 parts by weight of sodium carboxymethylcellulose as a binder in 98 parts by weight of the negative electrode active material. An aqueous dispersion of styrene-butadiene rubber (the concentration of styrene-butadiene rubber is 50% by mass) was mixed with a disperser to prepare a slurry. The obtained slurry was coated on both sides of a rolled copper foil with a thickness of 10 μm, dried, rolled to a thickness of 75 μm with a press, and then cut into an active material layer with a width of 104 mm and a length of 104 mm and an uncoated layer with a width of 30 mm. The shape of the part, as the negative pole. The density of the active material of the negative electrode at this time was 1.35 g/cm 3 .
《非水电解液的制作1》"Preparation of non-aqueous electrolyte 1"
在干燥的氩氛围下,在碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的混合物(体积比3∶3∶4)中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。另外,还含有0.3质量%的二氟磷酸锂盐(LiPO2F2)。Under a dry argon atmosphere, in a mixture of ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) (volume ratio 3:3:4), at a concentration of 1mol/L Dissolve well-dried lithium hexafluorophosphate (LiPF 6 ). In addition, 0.3% by mass of lithium difluorophosphate (LiPO 2 F 2 ) was contained.
《非水电解液的制作2》"The production of non-aqueous electrolyte 2"
在干燥的氩氛围下,在碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的混合物(体积比3∶3∶4)中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。另外,还含有0.3质量%的甲磺酸三甲基甲硅烷基酯。Under a dry argon atmosphere, in a mixture of ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) (volume ratio 3:3:4), at a concentration of 1mol/L Dissolve well-dried lithium hexafluorophosphate (LiPF 6 ). In addition, 0.3% by mass of trimethylsilyl methanesulfonate was contained.
《非水电解液的制作3》"The Production of Non-aqueous Electrolyte 3"
在干燥的氩氛围下,在碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的混合物(体积比3∶3∶4)中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。另外,还含有0.3质量%的六甲基环三硅氧烷。Under a dry argon atmosphere, in a mixture of ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) (volume ratio 3:3:4), at a concentration of 1mol/L Dissolve well-dried lithium hexafluorophosphate (LiPF 6 ). In addition, 0.3% by mass of hexamethylcyclotrisiloxane was contained.
《非水电解液的制作4》"The Production of Non-aqueous Electrolyte 4"
在干燥的氩氛围下,在碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的混合物(体积比3∶3∶4)中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。Under a dry argon atmosphere, in a mixture of ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) (volume ratio 3:3:4), at a concentration of 1mol/L Dissolve well-dried lithium hexafluorophosphate (LiPF 6 ).
(电池的制作1)(Battery production 1)
将32片正极和33片负极交替配置,各电极之间夹入多孔聚乙烯片隔板(厚度为25μm),进行叠层。此时,使正极活性物质面与负极活性物质面相对,并且不会露出负极活性物质面之外。将该正极和负极各自的未涂布部分焊接,制作集电片,将电极组密封在电池罐(外部尺寸:120×110×10mm)中。然后,在装有电极组的电池罐中注入20mL非水电解液,使电极充分浸透,密封而制作方形电池。该电池的额定放电容量约为6安培小时(Ah),以10kHz交流法测定的直流电阻成分约为5毫欧(mΩ)。正极的电极面积总和相对于电池外壳表面积总和之比为20.6。32 positive electrodes and 33 negative electrodes were arranged alternately, and a porous polyethylene sheet separator (thickness: 25 μm) was sandwiched between the electrodes for lamination. At this time, the surface of the positive electrode active material faces the surface of the negative electrode active material without protruding from the surface of the negative electrode active material. The respective uncoated parts of the positive and negative electrodes were welded to produce current collector sheets, and the electrode group was sealed in a battery can (outer dimensions: 120×110×10 mm). Then, 20 mL of non-aqueous electrolytic solution was poured into the battery can containing the electrode group, the electrodes were fully soaked, and sealed to produce a square battery. The rated discharge capacity of the battery is about 6 ampere hours (Ah), and the DC resistance component measured by the 10 kHz alternating current method is about 5 milliohms (mΩ). The ratio of the total electrode area of the positive electrode to the total surface area of the battery case was 20.6.
负极[4]实施例1Negative pole [4] embodiment 1
使用《负极的制作1》项的负极活性物质为碳质物质(D)而制成的负极、《正极的制作1》项中制作的正极以及《非水电解液的制作1》项中制作的非水电解液,通过《电池的制作1》项中的方法制作电池。通过下述《电池的评价》项中所述的方法以及上述测定方法对该电池进行测定。结果示于负极[4]表2。Use the negative electrode made of carbonaceous material (D) as the negative electrode active material in the item "Making of Negative Electrode 1", the positive electrode made in the item of "Making of Positive Electrode 1", and the one made in the item of "Making of Nonaqueous Electrolyte 1" Non-aqueous electrolyte, the battery is made by the method in "Battery Production 1". The battery was measured by the method described in the following item "Evaluation of Battery" and the above-mentioned measurement method. The results are shown in Table 2 of the negative electrode [4].
负极[4]实施例2Negative pole [4] embodiment 2
除了负极[4]实施例1的《负极的制作1》项的负极活性物质使用碳质物质(E)以外,与负极[4]实施例1同样地制作电池,并进行《电池的评价》项记载的电池评价。结果示于负极[4]表2。Except that the carbonaceous material (E) is used as the negative electrode active material in the negative electrode [4] Example 1 of "Making of Negative Electrode 1", a battery is produced in the same manner as in negative electrode [4] Example 1, and the item "Battery Evaluation" is carried out. Documented battery evaluation. The results are shown in Table 2 of the negative electrode [4].
负极[4]实施例3Negative electrode [4] Example 3
除了负极[4]实施例1的《负极的制作1》项的负极活性物质使用碳质物质(F)以外,与负极[4]实施例1同样地制作电池,并进行《电池的评价》项记载的电池评价。结果示于负极[4]表2。Except that the carbonaceous material (F) is used as the negative electrode active material in the negative electrode [4] Example 1 of "Making of the Negative Electrode 1", a battery is produced in the same manner as in the negative electrode [4] Example 1, and the item "Evaluation of the Battery" is carried out. Documented battery evaluation. The results are shown in Table 2 of the negative electrode [4].
负极[4]实施例4Negative pole [4] embodiment 4
除了负极[4]实施例1的《负极的制作1》项的负极活性物质使用碳质物质(G)以外,与负极[4]实施例1同样地制作电池,并进行《电池的评价》项记载的电池评价。结果示于负极[4]表2。Except that the carbonaceous material (G) is used as the negative electrode active material in the negative electrode [4] Example 1 of "Making of the Negative Electrode 1", a battery is produced in the same manner as in the negative electrode [4] Example 1, and the item "Evaluation of the Battery" is carried out. Documented battery evaluation. The results are shown in Table 2 of the negative electrode [4].
负极[4]实施例5Negative pole [4] embodiment 5
除了负极[4]实施例1的《负极的制作1》项的负极活性物质使用碳质物质(I)以外,与负极[4]实施例1同样地制作电池,并进行《电池的评价》项记载的电池评价。结果示于负极[4]表2。Except that negative electrode [4] embodiment 1 " making of negative electrode 1 " item negative electrode active material uses carbonaceous material (I), make battery similarly with negative electrode [4] embodiment 1, and carry out " evaluation of battery " item Documented battery evaluation. The results are shown in Table 2 of the negative electrode [4].
负极[4]实施例6~10Negative pole [4] Embodiment 6~10
除了将负极[4]实施例1~5的非水电解液分别换成《非水电解液的制作2》项中制作的非水电解液以外,用同样的方法制作电池,并进行评价。结果示于表2(负极[4]表2)。Except that the non-aqueous electrolytes in Examples 1 to 5 of the negative electrode [4] were replaced with the non-aqueous electrolytes prepared in the item "Preparation of Non-aqueous Electrolyte 2", batteries were produced in the same way and evaluated. The results are shown in Table 2 (negative electrode [4] Table 2).
负极[4]实施例11~15Negative electrode [4] Examples 11-15
除了将负极[4]实施例1~5的非水电解液分别换成《非水电解液的制作3》项中制作的非水电解液以外,用同样的方法制作电池,并进行评价。结果示于负极[4]表2。Except that the non-aqueous electrolytic solutions in Examples 1 to 5 of the negative electrode [4] were replaced with the non-aqueous electrolytic solutions prepared in the item "Preparation of Non-aqueous Electrolyte 3", batteries were produced in the same way and evaluated. The results are shown in Table 2 of the negative electrode [4].
负极[4]比较例1Negative electrode [4] Comparative example 1
除了负极[4]实施例1的《负极的制作1》项的负极活性物质使用球形化天然石墨(C)以外,与负极[4]实施例1同样地制作电池,并进行《电池的评价》项记载的电池评价。结果示于负极[4]表2。Except that the negative electrode active material of negative electrode [4] Example 1 of "Making of Negative Electrode 1" uses spheroidized natural graphite (C), a battery is produced in the same manner as negative electrode [4] Example 1, and "Battery Evaluation" is carried out Item recorded battery evaluation. The results are shown in Table 2 of the negative electrode [4].
负极[4]比较例2Negative electrode [4] Comparative example 2
除了负极[4]比较例1的非水电解液使用《非水电解液的制作4》项中制作的非水电解液以外,与负极[4]比较例1同样地制作电池,并进行《电池的评价》项记载的电池评价。结果示于负极[4]表2。Except that the non-aqueous electrolytic solution of negative pole [4] comparative example 1 uses the non-aqueous electrolytic solution made in " making 4 of non-aqueous electrolytic solution ", make battery similarly with negative pole [4] comparative example 1, and carry out " battery The battery evaluation described in item "Evaluation". The results are shown in Table 2 of the negative electrode [4].
负极[4]比较例3Negative electrode [4] Comparative example 3
除了负极[4]实施例1的《负极的制作1》项的负极活性物质使用热处理天然石墨(H)以外,与负极[4]实施例1同样地制作电池,并进行《电池的评价》项记载的电池评价。结果示于负极[4]表2。Except that the negative electrode active material of negative electrode [4] Example 1 of "Making of Negative Electrode 1" uses heat-treated natural graphite (H), a battery is made in the same manner as negative electrode [4] Example 1, and the item "Battery Evaluation" is carried out Documented battery evaluation. The results are shown in Table 2 of the negative electrode [4].
负极[4]比较例4Negative electrode [4] Comparative example 4
除了负极[4]比较例3的非水电解液使用《非水电解液的制作4》项中制作的非水电解液以外,与负极[4]比较例3同样地制作电池,并进行《电池的评价》项记载的电池评价。结果示于负极[4]表2。Except that the non-aqueous electrolytic solution of negative pole [4] comparative example 3 uses the non-aqueous electrolytic solution made in " making 4 of non-aqueous electrolytic solution ", make battery similarly with negative pole [4] comparative example 3, and carry out " battery The battery evaluation described in item "Evaluation". The results are shown in Table 2 of the negative electrode [4].
负极[4]比较例5Negative electrode [4] Comparative example 5
除了负极[4]实施例5的非水电解液使用《非水电解液的制作4》项中制作的非水电解液以外,与负极[4]实施例5同样地制作电池,并进行《电池的评价》项记载的电池评价。结果示于负极[4]表2。Except that the non-aqueous electrolytic solution of negative pole [4] embodiment 5 uses the non-aqueous electrolytic solution made in " making 4 of non-aqueous electrolytic solution " item, make battery similarly with negative pole [4] embodiment 5, and carry out " battery The battery evaluation described in item "Evaluation". The results are shown in Table 2 of the negative electrode [4].
负极[4]比较例6Negative electrode [4] Comparative Example 6
除了负极[4]实施例1的非水电解液使用《非水电解液的制作4》项中制作的非水电解液以外,与负极[4]实施例1同样地制作电池,并进行《电池的评价》项记载的电池评价。结果示于负极[4]表2。Except that the nonaqueous electrolytic solution of negative pole [4] embodiment 1 uses the nonaqueous electrolytic solution made in " making 4 of nonaqueous electrolytic solution " item, make battery similarly with negative pole [4] embodiment 1, and carry out " battery The battery evaluation described in item "Evaluation". The results are shown in Table 2 of the negative electrode [4].
负极[4]比较例7、8Negative electrode [4] Comparative example 7, 8
除了将负极[4]比较例1、3的非水电解液换成《非水电解液的制作2》项中制作的非水电解液以外,分别按照同样的方法制作电池,并进行评价。结果示于负极[4]表2。Except that the non-aqueous electrolyte solution of the negative electrode [4] Comparative Examples 1 and 3 was replaced with the non-aqueous electrolyte solution prepared in the item "Preparation of Non-aqueous Electrolyte Solution 2", batteries were produced and evaluated in the same manner. The results are shown in Table 2 of the negative electrode [4].
负极[4]比较例9、10Negative electrode [4] Comparative example 9, 10
除了将负极[4]比较例1、3的非水电解液换成《非水电解液的制作3》项中制作的非水电解液以外,分别按照同样的方法制作电池,并进行评价。结果示于负极[4]表2。Except that the non-aqueous electrolyte solution of the negative electrode [4] Comparative Examples 1 and 3 was replaced with the non-aqueous electrolyte solution prepared in the item "Preparation of Non-aqueous Electrolyte Solution 3", batteries were produced and evaluated in the same manner. The results are shown in Table 2 of the negative electrode [4].
负极[4]表2Negative electrode [4] Table 2
负极[4]《电池的评价》Negative electrode [4] "Battery Evaluation"
(容量测定)(capacity measurement)
对未经充放电循环的电池,在25℃、4.2V~3.0V的电压范围下,以0.2C的电流值(将1小时放电额定容量的电流值作为1C,所述额定容量取决于1小时率(one-hour-rate)的放电容量,下同)进行5个循环的初期充放电。将此时的第5个循环的0.2C放电容量作为初期容量。For a battery that has not been charged and discharged, at 25°C and a voltage range of 4.2V to 3.0V, with a current value of 0.2C (the current value of the rated capacity of 1 hour discharge is taken as 1C, and the rated capacity depends on 1 hour Rate (one-hour-rate) discharge capacity, the same below) for 5 cycles of initial charge and discharge. The 0.2C discharge capacity at the fifth cycle at this time was taken as the initial capacity.
(保存试验)(preservation test)
在被视为锂二次电池的实际使用上限温度的60℃的高温环境下进行保存。以0.2C的恒定电流恒定电压法充电至容量达到容量测定时测定的初期容量的20%,且达到充电上限电压4.2V,然后在60℃的高温环境下保存1周。在25℃环境下将保存后的电池以0.2C放电至3V,进一步在与(容量测定)相同的条件下进行3个循环的充放电,将该第3个循环的0.2C放电容量作为低充电深度保存后容量。由保存试验之前测定的初期容量和保存试验结束后测定的低充电深度保存后容量按照下述计算式求出循环保持率。Stored in a high-temperature environment of 60° C., which is regarded as the upper limit temperature for practical use of lithium secondary batteries. Charge it with a constant current and constant voltage method at 0.2C until the capacity reaches 20% of the initial capacity measured during capacity measurement, and reach the charging upper limit voltage of 4.2V, and then store it in a high temperature environment of 60°C for 1 week. The stored battery was discharged at 0.2C to 3V in an environment of 25°C, and further charged and discharged for 3 cycles under the same conditions as (capacity measurement), and the 0.2C discharge capacity of the third cycle was regarded as low charge Capacity after deep save. The cycle retention rate was obtained from the initial capacity measured before the storage test and the capacity after storage at a low charge depth measured after the storage test was completed according to the following calculation formula.
低充电深度保存后恢复率(%)=100×低充电深度保存后容量/初期容量Recovery rate after storage at low charging depth (%) = 100×capacity after storage at low charging depth/initial capacity
由上述结果可知,通过含有二氟磷酸锂盐、甲磺酸三甲基甲硅烷基酯、六甲基环三硅氧烷,并制成含有负极活性物质的圆形度为0.85以上、表面官能基团量O/C值为0~0.01的碳质物质的负极,可以使低充电深度的保存试验后的低充电深度保存后恢复率得到飞跃性的提高。From the above results, it can be seen that by containing lithium difluorophosphate, trimethylsilyl methanesulfonate, and hexamethylcyclotrisiloxane, and making a negative electrode active material with a circularity of 0.85 or more and a surface functional The negative electrode of the carbonaceous material with the O/C value of the radical amount of 0-0.01 can dramatically improve the recovery rate after storage at the low depth of charge after the storage test at the low depth of charge.
负极[5][负极材料的制作]Negative electrode [5] [Production of negative electrode material]
[负极材料的制作1][Production of negative electrode material 1]
将喹啉不溶成分为0.05质量%以下的煤焦油沥青在反应炉中于460℃热处理10小时,得到软化点385℃的具有熔融性的块状的热处理石墨结晶前体。用中间粉碎机(seishin企业公司制造的orient mill)将得到的块状的热处理石墨结晶前体粉碎,再使用微粉碎机(matsubo公司制造的涡轮式磨)进行微粉碎,得到中值粒径17μm的微细化的石墨结晶前体粉末(E)。Coal tar pitch with a quinoline-insoluble content of 0.05% by mass or less was heat-treated in a reaction furnace at 460° C. for 10 hours to obtain a molten block-shaped heat-treated graphite crystal precursor with a softening point of 385° C. The obtained massive heat-treated graphite crystal precursor was pulverized with an intermediate pulverizer (orient mill manufactured by Seishin Enterprise Co., Ltd.), and then finely pulverized using a fine pulverizer (turbine mill manufactured by Matsubo Co., Ltd.) to obtain a median particle size of 17 μm. The micronized graphite crystal precursor powder (E).
在上述微细化的石墨结晶前体粉末(E)中,以相对于微细化石墨结晶前体粉末和天然石墨的总重量为50质量%的比例混合中值粒径为17μm、长径比为1.4、振实密度为1.0g/cm3、BET比表面积为6.5g/cm3、圆形度为0.92的天然石墨,得到混合粉末。In the above-mentioned miniaturized graphite crystal precursor powder (E), a median particle size of 17 μm and an aspect ratio of 1.4 were mixed in a ratio of 50% by mass relative to the total weight of the miniaturized graphite crystal precursor powder and natural graphite. , natural graphite with a tap density of 1.0 g/cm 3 , a BET specific surface area of 6.5 g/cm 3 , and a circularity of 0.92 to obtain a mixed powder.
将该热处理石墨结晶前体的混合粉末装入到金属制的容器中,在箱形的电炉中、氮气流通下于540℃进行2小时热处理A。在热处理A中,微细化的石墨结晶前体粉末熔融,成为与天然石墨均匀地复合化的热处理石墨结晶前体的混合物的块。The mixed powder of the heat-treated graphite crystal precursor was put into a metal container, and heat treatment A was performed at 540° C. for 2 hours in a box-shaped electric furnace under nitrogen flow. In heat treatment A, the finer graphite crystal precursor powder is melted to form a lump of a heat-treated graphite crystal precursor mixture uniformly composited with natural graphite.
用粗粉碎机(吉田制作所制造的辊式压碎机)将该固化后的热处理石墨结晶前体的混合物的块粉碎,再用微粉碎机(matsubo公司制造的涡轮式磨)进行微粉碎,得到中值粒径18.5μm的粉末。The block of the mixture of the solidified heat-treated graphite crystal precursor was pulverized with a coarse pulverizer (roller crusher manufactured by Yoshida Seisakusho), and finely pulverized with a fine pulverizer (turbine mill manufactured by Matsubo Co., Ltd.), A powder having a median particle size of 18.5 μm was obtained.
将得到的粉末装入容器内,在氮气氛围下,用电炉在1000℃下烧制1小时。烧制后,得到的粉末(热处理B之前的前体混合物(F))仍为粉末的形态,几乎未发现熔融、熔合。The obtained powder was put into a container, and fired in an electric furnace at 1000° C. for 1 hour in a nitrogen atmosphere. After firing, the obtained powder (precursor mixture (F) before heat treatment B) was still in the form of powder, and almost no melting or fusion was observed.
此外,将烧制后的粉末转移到石墨坩埚中,使用直接通电炉在非活性氛围下于3000℃石墨化5小时,为了防止混入粗大粒子,使用ASTM400目的筛子反复筛5次,得到异取向性碳复合物(G)。In addition, the fired powder was transferred to a graphite crucible, and graphitized at 3000°C for 5 hours in an inert atmosphere using a direct electric furnace. In order to prevent the mixing of coarse particles, the ASTM400 mesh sieve was used to repeatedly sieve 5 times to obtain different orientation. Carbon composite (G).
[负极材料的制作2][Production of negative electrode material 2]
将[负极材料的制作1]中使用的天然石墨和作为能够石墨化的粘合剂的软化点88℃的粘合剂沥青以100∶30的重量比混合,再投入到预先加热到128℃的具有素炼机型搅拌翼的捏合机中,混合20分钟。The natural graphite used in [Manufacture of Negative Electrode Material 1] and binder pitch as a graphitizable binder with a softening point of 88°C were mixed in a weight ratio of 100:30, and then put into a pre-heated to 128°C The mixture was mixed for 20 minutes in a kneader equipped with a masticator-type stirring blade.
将充分捏合后的混合物填充到预先预热到108℃的模压机的模具中,放置5分钟,在混合物的温度稳定时挤压柱塞,施加2kfg/cm2(0.20MPa)的压力来进行成型。保持该压力1分钟,然后停止驱动,压力降低结束后,取出天然石墨和石墨结晶前体粉末复合后的成型体。Fill the fully kneaded mixture into the mold of a molding machine preheated to 108°C, let it stand for 5 minutes, squeeze the plunger when the temperature of the mixture is stable, and apply a pressure of 2kfg/cm 2 (0.20MPa) for molding . The pressure was maintained for 1 minute, and then the drive was stopped. After the pressure reduction was completed, the composite molded body of natural graphite and graphite crystal precursor powder was taken out.
将得到的成型体装入到作为耐热容器的金属制耐火箱中,并在空隙中填充石墨质焦粉。用电炉经过48个小时从室温升温到1000℃,在1000℃下保持3小时,进行脱挥发成分、烧制。接着,将成型体放在石墨坩埚中,并在空隙中填充石墨质焦粉,使用直接通电炉在非活性氛围中于3000℃加热4小时,进行石墨化。The obtained molded body was put into a metal refractory box as a heat-resistant container, and graphite coke powder was filled in the void. The temperature was raised from room temperature to 1000° C. in an electric furnace over 48 hours, and kept at 1000° C. for 3 hours for devolatilization and firing. Next, the molded body was placed in a graphite crucible, and graphite coke powder was filled in the gap, and heated at 3000° C. for 4 hours in an inert atmosphere using a direct electric furnace to perform graphitization.
用颚式破碎机将得到的石墨质的成型体粗粉碎后,采用将粉碎叶片转速设定为4000转/分的碾磨机进行微粉碎。另外,在分级处理时,为了防止混入粗大粒子,使用ASTM400目的筛子反复筛5次,得到异取向性碳复合物(H)。The obtained graphite molded body was coarsely pulverized by a jaw crusher, and then finely pulverized by a mill with a pulverizing blade whose rotational speed was set to 4000 rpm. In addition, during the classification treatment, in order to prevent mixing of coarse particles, sieving was repeated 5 times using an ASTM 400-mesh sieve to obtain a hetero-oriented carbon composite (H).
[负极材料的制作3][Production of negative electrode material 3]
除了使[负极材料的制作2]的使用直接通电炉进行的热处理在2200℃下进行以外,按照与[负极材料的制作2]同样的方法得到异取向性碳复合物(I)。An anisotropic carbon composite (I) was obtained in the same manner as in [Preparation of Negative Electrode Material 2], except that the heat treatment in [Preparation of Negative Electrode Material 2] was performed at 2200° C. using a direct electric furnace.
[负极材料的制作4][Production of negative electrode material 4]
除了使[负极材料的制作2]的天然石墨为中值粒径为10μm、长径比为2.3、振实密度为0.64g/cm3、BET比表面积为9.5m2/g、圆形度为0.83的焦炭以外,与[负极材料的制作2]同样地,得到异取向性碳复合物(J)。Except that the natural graphite in [Production of Negative Electrode Material 2] has a median particle size of 10 μm, an aspect ratio of 2.3, a tap density of 0.64 g/cm 3 , a BET specific surface area of 9.5 m 2 /g, and a circularity of Except for the coke of 0.83, a hetero-oriented carbon composite (J) was obtained in the same manner as [Preparation of Negative Electrode Material 2].
[负极材料的制作5][Production of negative electrode material 5]
除了将[负极材料的制作4]中使用的焦炭、石墨化催化剂的碳化硅和作为能够石墨化的粘合剂的软化点88℃的粘合剂沥青以100∶10∶30的质量比混合以外,与[负极材料的制作2]同样地,得到异取向性碳复合物(K)。In addition to mixing the coke used in [Production of Negative Electrode Material 4], silicon carbide as a graphitization catalyst, and binder pitch as a graphitizable binder with a softening point of 88°C in a mass ratio of 100:10:30 , in the same manner as in [Preparation of Negative Electrode Material 2], a hetero-oriented carbon composite (K) was obtained.
[负极材料的制作6][Production of negative electrode material 6]
除了使[负极材料的制作2]的天然石墨为中值粒径为19.8μm、长径比为3.2、振实密度为0.47g/cm3、BET比表面积为5.9m2/g、圆形度为0.81的鳞片状天然石墨以外,与[负极材料的制作2]同样地,得到异取向性碳复合物(L)。In addition to making the natural graphite in [Production of Negative Electrode Material 2] have a median particle size of 19.8 μm, an aspect ratio of 3.2, a tap density of 0.47 g/cm 3 , a BET specific surface area of 5.9 m 2 /g, and a circularity A different orientation carbon composite (L) was obtained in the same manner as in [Preparation of Negative Electrode Material 2] except for flaky natural graphite having a value of 0.81.
[负极材料的制作7][Production of negative electrode material 7]
除了使[负极材料的制作2]的天然石墨为中值粒径为35μm、长径比为1.4、振实密度为1.02g/cm3、BET比表面积为3.9m2/g、圆形度为0.90的天然石墨以外,与[负极材料的制作2]同样地,得到异取向性碳复合物(M)。Except that the natural graphite in [Production of Negative Electrode Material 2] has a median particle size of 35 μm, an aspect ratio of 1.4, a tap density of 1.02 g/cm 3 , a BET specific surface area of 3.9 m 2 /g, and a circularity of Except for the natural graphite of 0.90, a different orientation carbon composite (M) was obtained in the same manner as in [Preparation of Negative Electrode Material 2].
[负极材料的制作8][Production of negative electrode material 8]
除了将[负极材料的制作2]的粉碎叶片转速设定为1500转/分以外,与[负极材料的制作2]同样地,得到异取向性碳复合物(N)。A hetero-oriented carbon composite (N) was obtained in the same manner as in [Preparation of Negative Electrode Material 2] except that the rotational speed of the pulverizing blade was set at 1500 rpm.
[负极材料的制作9][Production of negative electrode material 9]
除了使[负极材料的制作2]的天然石墨为中值粒径为6μm、长径比为1.5、振实密度为0.15g/cm3的天然石墨以外,与[负极材料的制作2]同样地,得到异取向性碳复合物(O)。Except that the natural graphite in [Preparation of Negative Electrode Material 2] was natural graphite with a median particle size of 6 μm, an aspect ratio of 1.5, and a tap density of 0.15 g/cm 3 , it was the same as in [Preparation of Negative Electrode Material 2] , to obtain hetero-orientation carbon composite (O).
[负极材料的制作10][Production of negative electrode material 10]
将[负极材料的制作1]中得到的石墨结晶前体粉末(E)装入到金属制的容器中,在箱形的电炉中、氮气流通下于540℃进行2小时热处理A。在热处理A中,石墨结晶前体粉末(E)熔融,成为块状。The graphite crystal precursor powder (E) obtained in [Preparation of Negative Electrode Material 1] was put into a metal container, and heat treatment A was performed at 540° C. for 2 hours in a box-shaped electric furnace under nitrogen flow. In the heat treatment A, the graphite crystal precursor powder (E) is melted into a lump.
用粗粉碎机(吉田制作所制造的辊式压碎机)将该固化后的热处理石墨结晶前体块粉碎,再用微粉碎机(matsubo公司制造的涡轮式磨)进行微粉碎,得到中值粒径18.5μm的粉末。The solidified heat-treated graphite crystallization precursor block was crushed with a coarse pulverizer (roller crusher manufactured by Yoshida Seisakusho), and finely pulverized with a fine pulverizer (turbine mill manufactured by Matsubo Corporation) to obtain a median Powder with a particle size of 18.5 μm.
将得到的粉末放入容器内,在氮气氛围下,用电炉在1000℃下烧制1小时。烧制后,得到的粉末仍为粉末的形态,几乎未发现熔融、熔合。The obtained powder was put into a container, and fired in an electric furnace at 1000° C. for 1 hour in a nitrogen atmosphere. After firing, the obtained powder was still in the form of a powder, and melting and fusion were hardly observed.
此外,将烧制后的粉末转移到石墨坩埚中,使用直接通电炉在非活性氛围下于3000℃石墨化5小时,为了防止混入粗大粒子,使用ASTM400目的筛子反复筛5次,得到碳质物质(P)。In addition, the fired powder was transferred to a graphite crucible, and graphitized at 3000°C for 5 hours in an inert atmosphere using a direct electric furnace. In order to prevent the mixing of coarse particles, the ASTM400 mesh sieve was used to repeatedly sieve 5 times to obtain carbonaceous materials. (P).
[负极材料的制作11][Production of negative electrode material 11]
将[负极材料的制作1]中使用的天然石墨装入到金属制的容器中,在箱形的电炉中、氮气流通下于540℃进行2小时热处理A。在热处理A之后,天然石墨几乎未发现熔融、熔合。将得到的粉末放入容器内,在氮气氛围下,用电炉在1000℃下烧制1小时。烧制后,得到的粉末仍为粉末的形态,几乎未发现熔融、熔合。The natural graphite used in [Preparation of Negative Electrode Material 1] was put into a metal container, and heat treatment A was performed at 540° C. for 2 hours in a box-shaped electric furnace under nitrogen flow. After heat treatment A, melting and fusion of natural graphite were hardly observed. The obtained powder was put into a container, and fired in an electric furnace at 1000° C. for 1 hour in a nitrogen atmosphere. After firing, the obtained powder was still in the form of a powder, and melting and fusion were hardly observed.
此外,将烧制后的粉末转移到石墨坩埚中,使用直接通电炉在非活性氛围下于3000℃石墨化5小时,为了防止混入粗大粒子,使用ASTM400目的筛子反复筛5次,得到碳质物质(Q)。In addition, the fired powder was transferred to a graphite crucible, and graphitized at 3000°C for 5 hours in an inert atmosphere using a direct electric furnace. In order to prevent the mixing of coarse particles, the ASTM400 mesh sieve was used to repeatedly sieve 5 times to obtain carbonaceous materials. (Q).
[负极材料的制作12][Production of negative electrode material 12]
将碳质物质(P)和碳质物质(Q)各50质量%进行混合,均匀混合后,得到碳质物质混合物(R)。The carbonaceous substance (P) and the carbonaceous substance (Q) were mixed at 50% by mass each, and after uniform mixing, a carbonaceous substance mixture (R) was obtained.
负极[5][电池的制作]Negative electrode [5] [Battery production]
《正极的制作1》"Making of Positive Pole 1"
在N-甲基吡咯烷酮溶剂中混合90质量%的作为正极活性物质的钴酸锂(LiCoO2)、5质量%的作为导电材料的乙炔黑和5质量%作为粘合剂的聚偏氟乙烯(PVdF),制成浆料。将得到的浆料涂布在厚度15μm的铝箔两面,并干燥,用压制机压延成厚度80μm,再将其切成具有宽100mm、长100mm大小的活性物质层和宽30mm的未涂布部分的形状,作为正极。此时的正极的活性物质的密度为2.35g/cm3。90% by mass of lithium cobaltate (LiCoO 2 ) as a positive electrode active material, 5% by mass of acetylene black as a conductive material, and 5% by mass of polyvinylidene fluoride ( PVdF), made into slurry. The obtained slurry was coated on both sides of an aluminum foil with a thickness of 15 μm, dried, rolled to a thickness of 80 μm with a press, and cut into an active material layer having a width of 100 mm and a length of 100 mm and an uncoated portion of a width of 30 mm. shape, as the positive electrode. The density of the active material of the positive electrode at this time was 2.35 g/cm 3 .
《负极的制作1》"Making of Negative Pole 1"
在98重量份负极活性物质中添加100重量份作为增稠剂的羧甲基纤维素钠的水性分散液(羧甲基纤维素钠的浓度为1质量%)、2重量份作为粘合剂的苯乙烯-丁二烯橡胶的水性分散液(苯乙烯-丁二烯橡胶的浓度为50质量%),用分散器进行混合,制成浆料。将得到的浆料涂布在厚度10μm的压延铜箔两面,并干燥,用压制机压延成厚度75μm,再将其切成具有宽104mm、长104mm大小的活性物质层和宽30mm的未涂布部分的形状,作为负极。此时的负极的活性物质密度为1.35g/cm3。Add 100 parts by weight of an aqueous dispersion of sodium carboxymethylcellulose as a thickener (the concentration of sodium carboxymethylcellulose is 1% by mass) and 2 parts by weight of sodium carboxymethylcellulose as a binder in 98 parts by weight of the negative electrode active material. An aqueous dispersion of styrene-butadiene rubber (the concentration of styrene-butadiene rubber is 50% by mass) was mixed with a disperser to prepare a slurry. The obtained slurry was coated on both sides of a rolled copper foil with a thickness of 10 μm, dried, rolled to a thickness of 75 μm with a press, and then cut into an active material layer with a width of 104 mm and a length of 104 mm and an uncoated layer with a width of 30 mm. The shape of the part, as the negative pole. The active material density of the negative electrode at this time was 1.35 g/cm 3 .
《非水电解液的制作1》"Preparation of non-aqueous electrolyte 1"
在干燥的氩氛围下,在碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的混合物(体积比3∶3∶4)中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。另外,还含有0.3质量%的二氟磷酸锂盐(LiPO2F2)。Under a dry argon atmosphere, in a mixture of ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) (volume ratio 3:3:4), at a concentration of 1mol/L Dissolve well-dried lithium hexafluorophosphate (LiPF 6 ). In addition, 0.3% by mass of lithium difluorophosphate (LiPO 2 F 2 ) was contained.
《非水电解液的制作2》"The production of non-aqueous electrolyte 2"
在干燥的氩氛围下,在碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的混合物(体积比3∶3∶4)中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。另外,还含有0.3质量%的甲磺酸三甲基甲硅烷基酯。Under a dry argon atmosphere, in a mixture of ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) (volume ratio 3:3:4), at a concentration of 1mol/L Dissolve well-dried lithium hexafluorophosphate (LiPF 6 ). In addition, 0.3% by mass of trimethylsilyl methanesulfonate was contained.
《非水电解液的制作3》"The Production of Non-aqueous Electrolyte 3"
在干燥的氩氛围下,在碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的混合物(体积比3∶3∶4)中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。另外,还含有0.3质量%的六甲基环三硅氧烷。Under a dry argon atmosphere, in a mixture of ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) (volume ratio 3:3:4), at a concentration of 1mol/L Dissolve well-dried lithium hexafluorophosphate (LiPF 6 ). In addition, 0.3% by mass of hexamethylcyclotrisiloxane was contained.
《非水电解液的制作4》"The Production of Non-aqueous Electrolyte 4"
在干燥的氩氛围下,在碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的混合物(体积比3∶3∶4)中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。Under a dry argon atmosphere, in a mixture of ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) (volume ratio 3:3:4), at a concentration of 1mol/L Dissolve well-dried lithium hexafluorophosphate (LiPF 6 ).
(电池的制作1)(Battery production 1)
将32片正极和33片负极交替配置,各电极之间夹入多孔聚乙烯片隔板(厚度为25μm),进行叠层。此时,使正极活性物质面与负极活性物质面相对,并且不会露出负极活性物质面之外。将该正极和负极各自的未涂布部分焊接,制作集电片,将电极组密封在电池罐(外部尺寸:120×110×10mm)中。然后,在装有电极组的电池罐中注入20mL非水电解液,使电极充分浸透,密封而制作方形电池。该电池的额定放电容量约为6安培小时(Ah),以10kHz交流法测定的直流电阻约为5毫欧(mΩ)。32 positive electrodes and 33 negative electrodes were arranged alternately, and a porous polyethylene sheet separator (thickness: 25 μm) was sandwiched between the electrodes for lamination. At this time, the surface of the positive electrode active material faces the surface of the negative electrode active material without protruding from the surface of the negative electrode active material. The respective uncoated parts of the positive and negative electrodes were welded to produce current collector sheets, and the electrode group was sealed in a battery can (outer dimensions: 120×110×10 mm). Then, 20 mL of non-aqueous electrolytic solution was poured into the battery can containing the electrode group, the electrodes were fully soaked, and sealed to produce a square battery. The battery has a rated discharge capacity of about 6 ampere hours (Ah), and a DC resistance of about 5 milliohms (mΩ) measured by an alternating current method at 10 kHz.
负极[5]实施例1Negative pole [5] embodiment 1
使用《负极的制作1》项的负极活性物质为异取向性碳复合物(G)而制成的负极、《正极的制作1》项中制作的正极以及《非水电解液的制作1》项中制作的非水电解液,通过《电池的制作1》项中的方法制作电池。通过下述《电池的评价》项中所述的方法以及上述测定方法对该电池进行测定。Negative electrode made by using hetero-orientation carbon composite (G) as the negative electrode active material in item "Preparation of Negative Electrode 1", positive electrode produced in item "Preparation of Positive Electrode 1", and item "Preparation of Nonaqueous Electrolyte 1" The non-aqueous electrolyte solution produced in , and the battery was produced by the method in the "Battery Production 1" item. The battery was measured by the method described in the following item "Evaluation of Battery" and the above-mentioned measurement method.
负极[5]实施例2Negative pole [5] embodiment 2
除了负极[5]实施例1的《负极的制作1》项的负极活性物质使用异取向性碳复合物(H)以外,与负极[5]实施例1同样地制作电池,并进行《电池的评价》项记载的电池评价。Except that negative electrode [5] embodiment 1 " making of negative electrode 1 " item negative electrode active material uses different orientation carbon compound (H), make battery similarly with negative electrode [5] embodiment 1, and carry out " battery Evaluation" item recorded in the battery evaluation.
负极[5]实施例3Negative electrode [5] Example 3
除了负极[5]实施例1的《负极的制作1》项的负极活性物质使用异取向性碳复合物(I)以外,与负极[5]实施例1同样地制作电池,并进行《电池的评价》项记载的电池评价。Except that negative electrode [5] embodiment 1 " making 1 of negative electrode " item negative electrode active material uses different orientation carbon compound (I), make battery similarly with negative electrode [5] embodiment 1, and carry out " battery Evaluation" item recorded in the battery evaluation.
负极[5]实施例4Negative pole [5] embodiment 4
除了负极[5]实施例1的《负极的制作1》项的负极活性物质使用异取向性碳复合物(J)以外,与负极[5]实施例1同样地制作电池,并进行《电池的评价》项记载的电池评价。Except that negative electrode [5] embodiment 1 " making of negative electrode 1 " item negative electrode active material uses different orientation carbon compound (J), make battery similarly with negative electrode [5] embodiment 1, and carry out " battery Evaluation" item recorded in the battery evaluation.
负极[5]实施例5Negative pole [5] embodiment 5
除了负极[5]实施例1的《负极的制作1》项的负极活性物质使用异取向性碳复合物(K)以外,与负极[5]实施例1同样地制作电池,并进行《电池的评价》项记载的电池评价。Except that negative electrode [5] embodiment 1 " making of negative electrode 1 " item negative electrode active material uses different orientation carbon compound (K), make battery similarly with negative electrode [5] embodiment 1, and carry out " battery Evaluation" item recorded in the battery evaluation.
负极[5]实施例6Negative electrode [5] Embodiment 6
除了负极[5]实施例1的《负极的制作1》项的负极活性物质使用异取向性碳复合物(L)以外,与负极[5]实施例1同样地制作电池,并进行《电池的评价》项记载的电池评价。Except negative electrode [5] embodiment 1 " making of negative electrode 1 " item negative electrode active material uses different orientation carbon compound (L), make battery similarly with negative electrode [5] embodiment 1, and carry out " battery Evaluation" item recorded in the battery evaluation.
负极[5]实施例7Negative electrode [5] Embodiment 7
除了负极[5]实施例1的《负极的制作1》项的负极活性物质使用异取向性碳复合物(M)以外,与负极[5]实施例1同样地制作电池,并进行《电池的评价》项记载的电池评价。Except that the negative electrode active material of the negative electrode [5] embodiment 1 of "making 1 of the negative electrode" item uses a different orientation carbon composite (M), the battery is made in the same way as the negative electrode [5] embodiment 1, and the "Battery Preparation" Evaluation" item recorded in the battery evaluation.
负极[5]实施例8Negative electrode [5] embodiment 8
除了负极[5]实施例1的《负极的制作1》项的负极活性物质使用异取向性碳复合物(N)以外,与负极[5]实施例1同样地制作电池,并进行《电池的评价》项记载的电池评价。Except negative electrode [5] embodiment 1 " making 1 of negative electrode " item negative electrode active material uses different orientation carbon compound (N), make battery similarly with negative electrode [5] embodiment 1, and carry out " battery Evaluation" item recorded in the battery evaluation.
负极[5]实施例9Negative pole [5] embodiment 9
除了负极[5]实施例1的《负极的制作1》项的负极活性物质使用异取向性碳复合物(O)以外,与负极[5]实施例1同样地制作电池,并进行《电池的评价》项记载的电池评价。Except that negative electrode [5] embodiment 1 " making of negative electrode 1 " item negative electrode active material uses different orientation carbon compound (O), make battery similarly with negative electrode [5] embodiment 1, and carry out " battery Evaluation" item recorded in the battery evaluation.
负极[5]实施例10~18Negative electrode [5] Examples 10-18
除了将负极[5]实施例1~9的非水电解液换成《非水电解液的制作2》项中制作的非水电解液以外,同样地制作电池,并按照同样的方法进行电池评价。Except that the non-aqueous electrolyte solution in Examples 1-9 of the negative electrode [5] was replaced with the non-aqueous electrolyte solution prepared in the item "Preparation of Non-aqueous Electrolyte Solution 2", the battery was made in the same way, and the battery evaluation was performed in the same way .
负极[5]实施例19~27Negative electrode [5] Examples 19-27
除了将负极[5]实施例1~9的非水电解液换成《非水电解液的制作3》项中制作的非水电解液以外,同样地制作电池,并按照同样的方法进行电池评价。Except that the non-aqueous electrolyte solution in Examples 1-9 of the negative electrode [5] was replaced with the non-aqueous electrolyte solution produced in the item "Production of Non-aqueous Electrolyte Solution 3", the battery was made in the same way, and the battery evaluation was performed in the same way .
负极[5]比较例1Negative electrode [5] Comparative Example 1
除了负极[5]实施例1的《负极的制作1》项的负极活性物质使用碳质物质(P)以外,与负极[5]实施例1同样地制作电池,并进行《电池的评价》项记载的电池评价。Except that the negative electrode active material of negative electrode [5] Example 1 of "Making of Negative Electrode 1" uses a carbonaceous substance (P), a battery is produced in the same manner as negative electrode [5] Example 1, and the item "Battery Evaluation" is carried out. Documented battery evaluation.
负极[5]比较例2Negative electrode [5] Comparative Example 2
除了将负极[5]比较例1的非水电解液换成《非水电解液的制作4》项中制作的非水电解液以外,与负极[5]比较例1同样地制作电池,并进行《电池的评价》项记载的电池评价。Except that the non-aqueous electrolyte solution of the negative electrode [5] comparative example 1 is changed into the non-aqueous electrolyte solution made in the "making 4 of the non-aqueous electrolyte solution", the battery is made in the same way as the negative electrode [5] comparative example 1, and carried out Battery evaluation described in "Evaluation of Batteries".
负极[5]比较例3Negative electrode [5] Comparative example 3
除了负极[5]实施例1的《负极的制作1》项的负极活性物质使用碳质物质(Q)以外,与负极[5]实施例1同样地制作电池,并进行《电池的评价》项记载的电池评价。Except that the carbonaceous material (Q) is used as the negative electrode active material in the negative electrode [5] Example 1 of "Making of Negative Electrode 1", a battery is produced in the same manner as in negative electrode [5] Example 1, and the item "Battery Evaluation" is carried out. Documented battery evaluation.
负极[5]比较例4Negative electrode [5] Comparative example 4
除了将负极[5]比较例3的非水电解液换成《非水电解液的制作4》项中制作的非水电解液以外,与负极[5]比较例3同样地制作电池,并进行《电池的评价》项记载的电池评价。Except that the non-aqueous electrolyte solution of the negative electrode [5] comparative example 3 is changed into the non-aqueous electrolyte solution made in the "making 4 of the non-aqueous electrolyte solution", the battery is made in the same way as the negative electrode [5] comparative example 3, and carried out Battery evaluation described in "Evaluation of Batteries".
负极[5]比较例5Negative electrode [5] Comparative Example 5
除了负极[5]实施例1的《负极的制作1》项的负极活性物质使用碳质物质混合物(R)以外,与负极[5]实施例1同样地制作电池,并进行《电池的评价》项记载的电池评价。Except that the carbonaceous material mixture (R) was used as the negative electrode active material in the negative electrode [5] Example 1 of "Making of the Negative Electrode 1", a battery was produced in the same manner as in the negative electrode [5] Example 1, and "Evaluation of the Battery" was carried out. Item recorded battery evaluation.
负极[5]比较例6Negative electrode [5] Comparative example 6
除了将负极[5]比较例5的非水电解液换成《非水电解液的制作4》项中制作的非水电解液以外,与负极[5]比较例5同样地制作电池,并进行《电池的评价》项记载的电池评价。Except changing the non-aqueous electrolyte solution of the negative electrode [5] comparative example 5 into the non-aqueous electrolyte solution made in the item "Making 4 of the non-aqueous electrolyte solution", the battery is made in the same way as the negative electrode [5] comparative example 5, and carried out Battery evaluation described in "Evaluation of Batteries".
负极[5]比较例7Negative electrode [5] Comparative Example 7
除了将负极[5]实施例1的非水电解液换成《非水电解液的制作4》项中制作的非水电解液以外,与负极[5]实施例1同样地制作电池,并进行《电池的评价》项记载的电池评价。Except that the non-aqueous electrolytic solution of the negative pole [5] embodiment 1 is changed into the non-aqueous electrolytic solution made in the "making 4 of the non-aqueous electrolytic solution", the battery is made in the same way as the negative pole [5] embodiment 1, and carried out Battery evaluation described in "Evaluation of Batteries".
负极[5]比较例8Negative electrode [5] Comparative example 8
除了将负极[5]实施例2的非水电解液换成《非水电解液的制作4》项中制作的非水电解液以外,与负极[5]实施例2同样地制作电池,并进行《电池的评价》项记载的电池评价。Except that the non-aqueous electrolytic solution of the negative pole [5] embodiment 2 is changed into the non-aqueous electrolytic solution made in the "making 4 of the non-aqueous electrolytic solution", the battery is made in the same way as the negative pole [5] embodiment 2, and carried out Battery evaluation described in "Evaluation of Batteries".
负极[5]比较例9Negative electrode [5] Comparative example 9
除了将负极[5]实施例4的非水电解液换成《非水电解液的制作4》项中制作的非水电解液以外,与负极[5]实施例4同样地制作电池,并进行《电池的评价》项记载的电池评价。Except that the non-aqueous electrolytic solution of the negative pole [5] embodiment 4 is changed into the non-aqueous electrolytic solution made in the "making 4 of the non-aqueous electrolytic solution", the battery is made in the same way as the negative pole [5] embodiment 4, and carried out Battery evaluation described in "Evaluation of Batteries".
负极[5]比较例10Negative electrode [5] Comparative example 10
除了将负极[5]实施例5的非水电解液换成《非水电解液的制作4》项中制作的非水电解液以外,与负极[5]实施例5同样地制作电池,并进行《电池的评价》项记载的电池评价。Except that the non-aqueous electrolytic solution of the negative pole [5] embodiment 5 is changed into the non-aqueous electrolytic solution made in the "making 4 of the non-aqueous electrolytic solution", the battery is made in the same way as the negative pole [5] embodiment 5, and carried out Battery evaluation described in "Evaluation of Batteries".
负极[5]比较例11~13Negative electrode [5] Comparative examples 11-13
除了将负极[5]比较例1、3、5的非水电解液换成《非水电解液的制作2》项中制作的非水电解液以外,同样地制作电池,并按照同样的方法进行电池评价。Except that the non-aqueous electrolyte solution of the negative electrode [5] comparative examples 1, 3, and 5 is replaced by the non-aqueous electrolyte solution produced in the item "Making of Non-aqueous Electrolyte Solution 2", the battery is made in the same way and carried out in the same way Battery evaluation.
负极[5]比较例14~16Negative electrode [5] Comparative examples 14-16
除了将负极[5]比较例1、3、5的非水电解液换成《非水电解液的制作3》项中制作的非水电解液以外,同样地制作电池,并按照同样的方法进行电池评价。Except that the non-aqueous electrolyte solution of the negative electrode [5] comparative examples 1, 3, and 5 is replaced by the non-aqueous electrolyte solution made in the "Making of Non-aqueous Electrolyte Solution 3", the battery is made in the same way and carried out in the same way. Battery evaluation.
负极[5]《电池的评价》Negative electrode [5] "Battery Evaluation"
(容量测定)(capacity measurement)
对未经充放电循环的电池,在25℃、4.2V~3.0V的电压范围下,以0.2C的电流值(将1小时放电额定容量的电流值作为1C,所述额定容量取决于1小时率(one-hour-rate)的放电容量,下同)进行5个循环的初期充放电。将此时的第5个循环的0.2C放电容量作为初期容量。接着进行下述所示的输出功率测定。For a battery that has not been charged and discharged, at 25°C and a voltage range of 4.2V to 3.0V, with a current value of 0.2C (the current value of the rated capacity of 1 hour discharge is taken as 1C, and the rated capacity depends on 1 hour Rate (one-hour-rate) discharge capacity, the same below) for 5 cycles of initial charge and discharge. The 0.2C discharge capacity at the fifth cycle at this time was taken as the initial capacity. Next, the output power measurement shown below was performed.
(低充电深度循环试验)(Low charge deep cycle test)
在被视为锂二次电池的实际使用上限温度的60℃的高温环境下进行循环试验。以2C的恒定电流恒定电压法充电至容量达到容量测定时测定的初期容量的20%,且达到充电上限电压4.2V,然后以2C的恒定电流放电至放电终止电压3.0V,将该充放电循环作为1个循环,并反复进行该循环直至500循环。The cycle test was performed in a high-temperature environment of 60° C., which is regarded as the practical upper limit temperature of the lithium secondary battery. Charge with 2C constant current and constant voltage method until the capacity reaches 20% of the initial capacity measured during capacity measurement, and reach the charging upper limit voltage of 4.2V, then discharge at a constant current of 2C to the end-of-discharge voltage of 3.0V, and cycle the charge and discharge As one cycle, this cycle was repeated up to 500 cycles.
对于循环试验结束后的电池,在25℃环境下进行3个循环的充放电,将该第3个循环的0.2C放电容量作为低充电深度循环后容量。由循环试验之前测定的初期容量和循环试验结束后测定的低充电深度循环后容量按照下述计算式求出循环保持率。For the battery after the cycle test, 3 cycles of charging and discharging were performed in an environment of 25°C, and the 0.2C discharge capacity of the third cycle was taken as the capacity after low-charging deep cycle. The cycle retention rate was obtained from the initial capacity measured before the cycle test and the capacity after low-charge deep cycle measured after the cycle test, according to the following calculation formula.
循环保持率(%)=100×低充电深度循环后容量/初期容量Cycle retention (%) = 100 × capacity after low charge deep cycle / initial capacity
负极[5]实施例和负极[5]比较例中使用的负极活性物质一览示于负极[5]表1,电池评价的结果示于负极[5]表2和负极[5]表3。由负极[5]表2和负极[5]表3的结果可知,通过含有二氟磷酸锂盐、甲磺酸三甲基甲硅烷基酯、六甲基环三硅氧烷,并组合作为负极活性物质的异取向性碳复合物,可以使低充电深度的循环试验后的容量保持率(循环保持率)得到飞跃性的提高。The list of negative electrode active materials used in the negative electrode [5] example and the negative electrode [5] comparative example is shown in the negative electrode [5] Table 1, and the results of the battery evaluation are shown in the negative electrode [5] Table 2 and the negative electrode [5] Table 3. From the results in Table 2 of the negative electrode [5] and Table 3 of the negative electrode [5], it can be seen that by containing lithium difluorophosphate, trimethylsilyl methanesulfonate, and hexamethylcyclotrisiloxane, and combining them as the negative electrode The hetero-oriented carbon composite of the active material can dramatically improve the capacity retention rate (cycle retention rate) after a cycle test at a low depth of charge.
负极[5]表1Negative electrode [5] Table 1
负极[5]表2Negative electrode [5] Table 2
[表44][Table 44]
负极[5]表3Negative electrode [5] Table 3
[表45][Table 45]
负极[6][负极活性物质的制作]Negative electrode[6][Production of negative electrode active material]
(负极活性物质的制作1)(Production of negative electrode active material 1)
为了防止在市售的天然石墨粉末(A)中混入粗大粒子,使用ASTM400目的筛子反复筛5次。将这样得到的负极活性物质作为碳质物质(B)。In order to prevent coarse particles from being mixed into the commercially available natural graphite powder (A), sieve was repeated 5 times using an ASTM400 mesh sieve. The negative electrode active material thus obtained is referred to as a carbonaceous material (B).
(负极活性物质的制作2)(Production of negative electrode active material 2)
用微粉碎机(matsubo公司制造的涡轮式磨)对市售的天然石墨粉末(C)(d002:0.336nm、Lc:100nm以上、拉曼R值:0.11、振实密度:0.46g/cm3、真密度:2.27g/cm3、体积平均粒径:28.7μm)进行处理,为了防止混入粗大粒子,使用ASTM400目的筛子反复筛5次,制备碳质物质(D)。Commercially available natural graphite powder (C) (d002: 0.336nm, Lc: 100nm or more, Raman R value: 0.11, tap density: 0.46g/cm 3 , true density: 2.27g/cm 3 , volume average particle diameter: 28.7μm), and in order to prevent mixing of coarse particles, use an ASTM400 mesh sieve to repeatedly sieve 5 times to prepare carbonaceous material (D).
(负极活性物质的制作3)(Production of negative electrode active material 3)
使用混合系统((株)奈良机械制作所制造的混合系统NHS-1型)以处理量90g、转子圆周速度60m/s、处理时间3分钟对天然石墨粉末(C)进行处理,由此进行球形化,另外,为了防止混入粗大粒子,使用ASTM400目的筛子反复筛5次。将这样得到的负极活性物质作为碳质物质(E)。通过反复进行该操作,确保了电池制作所必需的量。Natural graphite powder (C) is processed into a spherical shape by using a mixing system (NHS-1 type mixing system manufactured by Nara Machinery Co., Ltd.) with a processing capacity of 90 g, a rotor peripheral speed of 60 m/s, and a processing time of 3 minutes. In addition, in order to prevent the mixing of coarse particles, use an ASTM400 mesh sieve to repeatedly sieve 5 times. The negative electrode active material thus obtained is referred to as a carbonaceous material (E). By repeating this operation, the quantity necessary for battery production is secured.
(负极活性物质的制作4)(Production of negative electrode active material 4)
使用混合系统以处理量90g、转子圆周速度30m/s、处理时间1分钟的条件对市售的天然石墨粉末(F)(d002:0.336nm、Lc:100nm以上、拉曼R值:0.09、振实密度:0.57g/cm3、真密度:2.26g/cm3、体积平均粒径:85.4μm)进行球形化,另外,为了防止混入粗大粒子,使用ASTM400目的筛子反复筛5次。将这样得到的负极活性物质作为碳质物质(G)。通过反复进行该操作,确保了电池制作所必需的量。Commercially available natural graphite powder (F) (d002: 0.336nm, Lc: above 100nm, Raman R value: 0.09, vibration Bulk density: 0.57g/cm 3 , true density: 2.26g/cm 3 , volume average particle diameter: 85.4μm) were spheroidized, and in order to prevent mixing of coarse particles, sieve was repeated 5 times using an ASTM400 mesh sieve. The negative electrode active material thus obtained is referred to as a carbonaceous material (G). By repeating this operation, the quantity necessary for battery production is secured.
(负极活性物质的制作5)(Production of negative electrode active material 5)
将喹啉不溶成分为0.05质量%以下的煤焦油沥青在反应炉中于460℃热处理10小时,得到软化点385℃的具有熔融性的块状的碳化处理前体。将得到的块状的碳化处理前体装入到金属制的容器中,在箱形的电炉中、氮气流通下于1000℃进行2小时热处理。用粗粉碎机(吉田制作所制造的辊式压碎机)将得到的无定形的块粉碎,再用微粉碎机(matsubo公司制造的涡轮式磨)进行微粉碎,得到体积基准平均粒径18μm的无定形粉末。为了防止得到的粉末中混入粗大粒子,使用ASTM400目的筛子反复筛5次。将这样得到的负极活性物质作为碳质物质(H)。Coal tar pitch with a quinoline-insoluble content of 0.05% by mass or less was heat-treated in a reaction furnace at 460°C for 10 hours to obtain a meltable block-shaped carbonized precursor with a softening point of 385°C. The obtained bulk carbonized precursor was put into a metal container, and heat-treated at 1000° C. for 2 hours in a box-shaped electric furnace under nitrogen flow. The obtained amorphous block was pulverized with a coarse pulverizer (roller crusher manufactured by Yoshida Seisakusho), and finely pulverized with a fine pulverizer (turbine mill manufactured by Matsubo Co., Ltd.) to obtain a volume-based average particle size of 18 μm of amorphous powder. In order to prevent coarse particles from being mixed into the obtained powder, the sieve was repeatedly sieved 5 times using an ASTM400 mesh sieve. The negative electrode active material thus obtained is referred to as a carbonaceous material (H).
(负极活性物质的制作6)(Production of negative electrode active material 6)
将(负极活性物质的制作5)中得到的碳质物质(H)再转移到石墨坩埚中,使用直接通电炉在非活性氛围下于3000℃进行5小时石墨化,为了防止得到的粉末中混入粗大粒子,使用ASTM400目的筛子反复筛5次。将这里得到的负极活性物质作为碳质物质(I)。The carbonaceous material (H) obtained in (production of negative electrode active material 5) was transferred to a graphite crucible, and graphitized at 3000° C. for 5 hours in an inert atmosphere using a direct electric furnace. In order to prevent the obtained powder from being mixed with For coarse particles, use an ASTM400 mesh sieve to repeatedly sieve 5 times. The negative electrode active material obtained here is referred to as carbonaceous material (I).
(负极活性物质的制作7)(Production of negative electrode active material 7)
使用混合系统以处理量90g、转子圆周速度30m/s、处理时间1分钟的条件对(负极活性物质的制作6)中得到的碳质物质(I)进行球形化,另外,为了防止混入粗大粒子,使用ASTM400目的筛子反复筛5次。将这样得到的负极活性物质作为碳质物质(J)。通过反复进行该操作,确保了电池制作所必需的量。The carbonaceous material (I) obtained in (Production of Negative Electrode Active Material 6) was spheroidized using a mixing system with a treatment capacity of 90 g, a rotor peripheral speed of 30 m/s, and a treatment time of 1 minute. In addition, in order to prevent the mixing of coarse particles , use the ASTM400 mesh sieve to sieve repeatedly 5 times. The negative electrode active material thus obtained is referred to as a carbonaceous material (J). By repeating this operation, the quantity necessary for battery production is secured.
(负极活性物质的制作8)(Production of negative electrode active material 8)
将纯度比天然石墨粉末(A)低的天然石墨粉末(K)(d002:0.336nm、Lc:100nm以上、拉曼R值:0.10、振实密度:0.49g/cm3、真密度:2.27g/cm3、体积平均粒径:27.3μm、灰分0.5质量%)在与(负极活性物质的制作3)同样的条件下进行球形化和筛分,制备碳质物质(L)。通过反复进行该操作,确保了电池制作所必需的量。Natural graphite powder (K) (d002: 0.336nm, Lc: 100nm or more, Raman R value: 0.10, tap density: 0.49g/cm 3 , true density: 2.27g) lower in purity than natural graphite powder (A) /cm 3 , volume average particle diameter: 27.3 μm, ash content: 0.5% by mass) were spheroidized and sieved under the same conditions as (preparation of negative electrode active material 3) to prepare a carbonaceous material (L). By repeating this operation, the quantity necessary for battery production is secured.
(负极活性物质的制作9)(Production of negative electrode active material 9)
为了防止在市售的鳞片状天然石墨粉末(M)中混入粗大粒子,使用ASTM400目的筛子反复筛5次。将这样得到的负极活性物质作为碳质物质(N)。In order to prevent the commercially available flaky natural graphite powder (M) from being mixed with coarse particles, it was repeatedly sieved five times using an ASTM400 mesh sieve. The negative electrode active material thus obtained is referred to as a carbonaceous material (N).
按照上述的方法测定碳质物质(B)、(D)、(E)、(G)、(H)、(I)、(J)、(L)、(N)的形状和物性。结果示于负极[6]表1。The shape and physical properties of the carbonaceous substances (B), (D), (E), (G), (H), (I), (J), (L), and (N) were measured according to the methods described above. The results are shown in Table 1 of the negative electrode [6].
负极[6]表1Negative electrode [6] Table 1
负极[6][电池的制作]Negative electrode [6] [Battery production]
《正极的制作1》"Making of Positive Pole 1"
在N-甲基吡咯烷酮溶剂中混合90质量%作为正极活性物质的钴酸锂(LiCoO2)、5质量%的作为导电材料的乙炔黑和5质量%作为粘合剂的聚偏氟乙烯(PVdF),制成浆料。将得到的浆料涂布在厚度15μm的铝箔两面,并干燥,用压制机压延成厚度80μm,再将其切成具有宽100mm、长100mm大小的活性物质层和宽30mm的未涂布部分的形状,作为正极。此时的正极的活性物质的密度为2.35g/cm3。Mix 90% by mass of lithium cobaltate (LiCoO 2 ) as a positive electrode active material, 5% by mass of acetylene black as a conductive material, and 5% by mass of polyvinylidene fluoride (PVdF ) to make slurry. The obtained slurry was coated on both sides of an aluminum foil with a thickness of 15 μm, dried, rolled to a thickness of 80 μm with a press, and cut into an active material layer having a width of 100 mm and a length of 100 mm and an uncoated portion of a width of 30 mm. shape, as the positive electrode. The density of the active material of the positive electrode at this time was 2.35 g/cm 3 .
《负极的制作1》"Making of Negative Pole 1"
在98重量份负极活性物质中添加100重量份作为增稠剂的羧甲基纤维素钠的水性分散液(羧甲基纤维素钠的浓度为1质量%)、2重量份作为粘合剂的苯乙烯-丁二烯橡胶的水性分散液(苯乙烯-丁二烯橡胶的浓度为50质量%),用分散器进行混合,制成浆料。将得到的浆料涂布在厚度10μm的压延铜箔两面,并干燥,用压制机压延成厚度75μm,再将其切成具有宽104mm、长104mm大小的活性物质层和宽30mm的未涂布部分的形状,作为负极。此时的负极的活性物质的密度为1.35g/cm3。Add 100 parts by weight of an aqueous dispersion of sodium carboxymethylcellulose as a thickener (the concentration of sodium carboxymethylcellulose is 1% by mass) and 2 parts by weight of sodium carboxymethylcellulose as a binder in 98 parts by weight of the negative electrode active material. An aqueous dispersion of styrene-butadiene rubber (the concentration of styrene-butadiene rubber is 50% by mass) was mixed with a disperser to prepare a slurry. The obtained slurry was coated on both sides of a rolled copper foil with a thickness of 10 μm, dried, rolled to a thickness of 75 μm with a press, and then cut into an active material layer with a width of 104 mm and a length of 104 mm and an uncoated layer with a width of 30 mm. The shape of the part, as the negative pole. The density of the active material of the negative electrode at this time was 1.35 g/cm 3 .
《非水电解液的制作1》"Preparation of non-aqueous electrolyte 1"
在干燥的氩氛围下,在碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的混合物(体积比3∶3∶4)中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。另外,还含有0.3质量%的二氟磷酸锂盐(LiPO2F2)。Under a dry argon atmosphere, in a mixture of ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) (volume ratio 3:3:4), at a concentration of 1mol/L Dissolve well-dried lithium hexafluorophosphate (LiPF 6 ). In addition, 0.3% by mass of lithium difluorophosphate (LiPO 2 F 2 ) was contained.
《非水电解液的制作2》"The production of non-aqueous electrolyte 2"
在干燥的氩氛围下,在碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的混合物(体积比3∶3∶4)中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。另外,还含有0.3质量%的甲磺酸三甲基甲硅烷基酯。Under a dry argon atmosphere, in a mixture of ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) (volume ratio 3:3:4), at a concentration of 1mol/L Dissolve well-dried lithium hexafluorophosphate (LiPF 6 ). In addition, 0.3% by mass of trimethylsilyl methanesulfonate was contained.
《非水电解液的制作3》"The Production of Non-aqueous Electrolyte 3"
在干燥的氩氛围下,在碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的混合物(体积比3∶3∶4)中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。另外,还含有0.3质量%的六甲基环三硅氧烷。Under a dry argon atmosphere, in a mixture of ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) (volume ratio 3:3:4), at a concentration of 1mol/L Dissolve well-dried lithium hexafluorophosphate (LiPF 6 ). In addition, 0.3% by mass of hexamethylcyclotrisiloxane was contained.
《非水电解液的制作4》"The Production of Non-aqueous Electrolyte 4"
在干燥的氩氛围下,在碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的混合物(体积比3∶3∶4)中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。Under a dry argon atmosphere, in a mixture of ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) (volume ratio 3:3:4), at a concentration of 1mol/L Dissolve well-dried lithium hexafluorophosphate (LiPF 6 ).
《电池的制作1》"Battery Making 1"
将32片正极和33片负极交替配置,各电极之间夹入多孔聚乙烯片隔板(厚度为25μm),进行叠层。此时,使正极活性物质面与负极活性物质面相对,并且不会露出负极活性物质面之外。将该正极和负极各自的未涂布部分焊接,制作集电片,将电极组密封在电池罐(外部尺寸:120×110×10mm)中。然后,在装有电极组的电池罐中注入20mL非水电解液,使电极充分浸透,密封而制作方形电池。该电池的额定放电容量约为6安培小时(Ah),以10kHz交流法测定的直流电阻成分约为5毫欧(mΩ)。正极的电极面积的总和相对于电池的外壳表面积的总和之比为20.6。32 positive electrodes and 33 negative electrodes were arranged alternately, and a porous polyethylene sheet separator (thickness: 25 μm) was sandwiched between the electrodes for lamination. At this time, the surface of the positive electrode active material faces the surface of the negative electrode active material without protruding from the surface of the negative electrode active material. The respective uncoated parts of the positive and negative electrodes were welded to produce current collector sheets, and the electrode group was sealed in a battery can (outer dimensions: 120×110×10 mm). Then, 20 mL of non-aqueous electrolytic solution was poured into the battery can containing the electrode group, the electrodes were fully soaked, and sealed to produce a square battery. The rated discharge capacity of the battery is about 6 ampere hours (Ah), and the DC resistance component measured by the 10 kHz alternating current method is about 5 milliohms (mΩ). The ratio of the sum of the electrode areas of the positive electrode to the sum of the surface areas of the battery case was 20.6.
负极[6]实施例1Negative electrode [6] Embodiment 1
使用《负极的制作1》项的负极活性物质为碳质物质(D)而制成的负极、《正极的制作1》项中制作的正极以及《非水电解液的制作1》项中制作的非水电解液,通过《电池的制作1》项中的方法制作电池。通过下述《电池的评价》项中所述的方法以及上述测定方法对该电池进行测定。结果示于表2(负极[6]表2)。Use the negative electrode made of carbonaceous material (D) as the negative electrode active material in the item "Making of Negative Electrode 1", the positive electrode made in the item of "Making of Positive Electrode 1", and the one made in the item of "Making of Nonaqueous Electrolyte 1" Non-aqueous electrolyte, the battery is made by the method in "Battery Production 1". The battery was measured by the method described in the following item "Evaluation of Battery" and the above-mentioned measurement method. The results are shown in Table 2 (negative electrode [6] Table 2).
负极[6]实施例2Negative pole [6] embodiment 2
除了负极[6]实施例1的《负极的制作1》项中的负极活性物质使用碳质物质(E)以外,与负极[6]实施例1同样地制作电池,并进行《电池的评价》项记载的电池评价。结果示于负极[6]表2。Except that the carbonaceous material (E) is used as the negative electrode active material in the negative electrode [6] Example 1 of "Making of the Negative Electrode 1", a battery is produced in the same manner as in the negative electrode [6] Example 1, and "Evaluation of the Battery" is carried out. Item recorded battery evaluation. The results are shown in Table 2 of the negative electrode [6].
负极[6]实施例3Negative electrode [6] Example 3
除了负极[6]实施例1的《负极的制作1》项中的负极活性物质使用碳质物质(G)以外,与负极[6]实施例1同样地制作电池,并进行《电池的评价》项记载的电池评价。结果示于负极[6]表2。Except that the carbonaceous material (G) was used as the negative electrode active material in the negative electrode [6] Example 1 of "Making of the Negative Electrode 1", a battery was produced in the same manner as in the negative electrode [6] Example 1, and "Evaluation of the Battery" was carried out. Item recorded battery evaluation. The results are shown in Table 2 of the negative electrode [6].
负极[6]实施例4Negative pole [6] embodiment 4
除了负极[6]实施例1的《负极的制作1》项中的负极活性物质使用碳质物质(J)以外,与负极[6]实施例1同样地制作电池,并进行《电池的评价》项记载的电池评价。结果示于负极[6]表2。Except that the carbonaceous material (J) was used as the negative electrode active material in the negative electrode [6] Example 1 of "Making of the Negative Electrode 1", a battery was produced in the same manner as in the negative electrode [6] Example 1, and "Evaluation of the Battery" was carried out. Item recorded battery evaluation. The results are shown in Table 2 of the negative electrode [6].
负极[6]实施例5Negative pole [6] embodiment 5
除了负极[6]实施例1的《负极的制作1》项中的负极活性物质使用碳质物质(L)以外,与负极[6]实施例1同样地制作电池,并进行《电池的评价》项记载的电池评价。结果示于负极[6]表2。Except that the carbonaceous material (L) was used as the negative electrode active material in the negative electrode [6] Example 1 of "Making of the Negative Electrode 1", a battery was produced in the same manner as in the negative electrode [6] Example 1, and "Evaluation of the Battery" was carried out. Item recorded battery evaluation. The results are shown in Table 2 of the negative electrode [6].
负极[6]实施例6~10Negative pole [6] Embodiment 6~10
除了将负极[6]实施例1~5的非水电解液换成《非水电解液的制作2》项中制作的非水电解液以外,分别按照同样的方法制作电池,并进行评价。结果示于负极[6]表2。Except that the non-aqueous electrolyte solution of Examples 1 to 5 of the negative electrode [6] was replaced with the non-aqueous electrolyte solution produced in the item "Preparation of Non-aqueous Electrolyte Solution 2", batteries were produced and evaluated in the same manner. The results are shown in Table 2 of the negative electrode [6].
负极[6]实施例11~15Negative electrode [6] Examples 11-15
除了将负极[6]实施例1~5的非水电解液换成《非水电解液的制作3》项中制作的非水电解液以外,分别按照同样的方法制作电池,并进行评价。结果示于负极[6]表2。Except that the non-aqueous electrolyte solution of Examples 1 to 5 of the negative electrode [6] was replaced with the non-aqueous electrolyte solution produced in the item "Preparation of Non-aqueous Electrolyte Solution 3", batteries were produced and evaluated in the same manner. The results are shown in Table 2 of the negative electrode [6].
负极[6]比较例1Negative electrode [6] Comparative Example 1
除了负极[6]实施例1的《负极的制作1》项中的负极活性物质使用碳质物质(B)以外,与负极[6]实施例1同样地制作电池,并进行《电池的评价》项记载的电池评价。结果示于负极[6]表2。Except that the carbonaceous material (B) was used as the negative electrode active material in the negative electrode [6] Example 1 of "Making of the Negative Electrode 1", a battery was produced in the same manner as in the negative electrode [6] Example 1, and "Evaluation of the Battery" was carried out. Item recorded battery evaluation. The results are shown in Table 2 of the negative electrode [6].
负极[6]比较例2Negative electrode [6] Comparative Example 2
除了将负极[6]比较例1的非水电解液换成《非水电解液的制作4》项中制作的非水电解液以外,与负极[6]比较例1同样地制作电池,并进行《电池的评价》项记载的电池评价。结果示于负极[6]表2。Except changing the non-aqueous electrolyte solution of the negative electrode [6] comparative example 1 into the non-aqueous electrolyte solution made in the item "Making 4 of the non-aqueous electrolyte solution", the battery is made in the same way as the negative electrode [6] comparative example 1, and carried out Battery evaluation described in "Evaluation of Batteries". The results are shown in Table 2 of the negative electrode [6].
负极[6]比较例3Negative electrode [6] Comparative Example 3
除了将负极[6]实施例2的非水电解液换成《非水电解液的制作4》项中制作的非水电解液以外,与负极[6]实施例2同样地制作电池,并进行《电池的评价》项记载的电池评价。结果示于负极[6]表2。Except that the non-aqueous electrolytic solution of the negative pole [6] embodiment 2 is changed into the non-aqueous electrolytic solution made in the "making 4 of the non-aqueous electrolytic solution", the battery is made in the same way as the negative pole [6] embodiment 2, and carried out Battery evaluation described in "Evaluation of Batteries". The results are shown in Table 2 of the negative electrode [6].
负极[6]比较例4Negative electrode [6] Comparative example 4
除了将负极[6]实施例1的非水电解液换成《非水电解液的制作4》项中制作的非水电解液以外,与负极[6]实施例1同样地制作电池,并进行《电池的评价》项记载的电池评价。结果示于负极[6]表2。Except that the non-aqueous electrolytic solution of the negative pole [6] embodiment 1 is changed into the non-aqueous electrolytic solution made in the "making 4 of the non-aqueous electrolytic solution" item, the battery is made in the same way as the negative pole [6] embodiment 1, and carried out Battery evaluation described in "Evaluation of Batteries". The results are shown in Table 2 of the negative electrode [6].
负极[6]比较例5Negative electrode [6] Comparative Example 5
除了负极[6]实施例1的《负极的制作1》项中的负极活性物质使用碳质物质(H)以外,与负极[6]实施例1同样地制作电池,并进行《电池的评价》项记载的电池评价。结果示于负极[6]表2。Except that the negative electrode active material in the negative electrode [6] Example 1 of "Making of the Negative Electrode 1" uses a carbonaceous substance (H), a battery is produced in the same manner as in the negative electrode [6] Example 1, and "Evaluation of the Battery" is carried out. Item recorded battery evaluation. The results are shown in Table 2 of the negative electrode [6].
负极[6]比较例6Negative electrode [6] Comparative example 6
除了将负极[6]比较例5的非水电解液换成《非水电解液的制作4》项中制作的非水电解液以外,与负极[6]比较例5同样地制作电池,并进行《电池的评价》项记载的电池评价。结果示于负极[6]表2。Except that the non-aqueous electrolyte solution of the negative electrode [6] comparative example 5 is changed into the non-aqueous electrolyte solution made in the "making 4 of the non-aqueous electrolyte solution", the battery is made in the same way as the negative electrode [6] comparative example 5, and carried out Battery evaluation described in "Evaluation of Batteries". The results are shown in Table 2 of the negative electrode [6].
负极[6]比较例7Negative electrode [6] Comparative example 7
除了负极[6]实施例1的《负极的制作1》项中的负极活性物质使用碳质物质(N)以外,与负极[6]实施例1同样地制作电池,并进行《电池的评价》项记载的电池评价。结果示于负极[6]表2。Except that the carbonaceous material (N) is used as the negative electrode active material in the negative electrode [6] Example 1 of "Making of the Negative Electrode 1", a battery is produced in the same manner as in the negative electrode [6] Example 1, and "Evaluation of the Battery" is carried out. Item recorded battery evaluation. The results are shown in Table 2 of the negative electrode [6].
负极[6]比较例8Negative electrode [6] Comparative Example 8
除了将负极[6]比较例7的非水电解液换成《非水电解液的制作4》项中制作的非水电解液以外,与负极[6]比较例7同样地制作电池,并进行《电池的评价》项记载的电池评价。结果示于负极[6]表2。Except that the non-aqueous electrolytic solution of the negative pole [6] comparative example 7 is changed into the non-aqueous electrolytic solution made in the "making 4 of the non-aqueous electrolytic solution", the battery is made in the same way as the negative pole [6] comparative example 7, and carried out Battery evaluation described in "Evaluation of Batteries". The results are shown in Table 2 of the negative electrode [6].
负极[6]比较例9Negative electrode [6] Comparative example 9
除了负极[6]实施例1的《负极的制作1》项中的负极活性物质使用碳质物质(I)以外,与负极[6]实施例1同样地制作电池,并进行《电池的评价》项记载的电池评价。结果示于负极[6]表2。Except that the negative electrode active material in the "making of negative electrode 1" item of the negative electrode [6] embodiment 1 uses carbonaceous material (I), the battery is made in the same manner as the negative electrode [6] embodiment 1, and "evaluation of the battery" is carried out Item recorded battery evaluation. The results are shown in Table 2 of the negative electrode [6].
负极[6]比较例10Negative electrode [6] Comparative example 10
除了将负极[6]比较例9的非水电解液换成《非水电解液的制作4》项中制作的非水电解液以外,与负极[6]比较例9同样地制作电池,并进行《电池的评价》项记载的电池评价。结果示于负极[6]表2。Except that the non-aqueous electrolyte solution of the negative electrode [6] comparative example 9 is changed into the non-aqueous electrolyte solution made in the "making 4 of the non-aqueous electrolyte solution", the battery is made in the same way as the negative electrode [6] comparative example 9, and carried out Battery evaluation described in "Evaluation of Batteries". The results are shown in Table 2 of the negative electrode [6].
负极[6]比较例11~14Negative electrode [6] Comparative Examples 11-14
除了将负极[6]比较例1、5、7、9的非水电解液换成《非水电解液的制作2》项中制作的非水电解液以外,分别按照同样的方法制作电池,并进行评价。结果示于负极[6]表2。Except that the non-aqueous electrolytic solution of negative electrode [6] comparative examples 1, 5, 7, 9 is changed into the non-aqueous electrolytic solution made in the "making 2 of non-aqueous electrolytic solution", the battery is made in the same way respectively, and Make an evaluation. The results are shown in Table 2 of the negative electrode [6].
负极[6]比较例15~18Negative electrode [6] Comparative Examples 15-18
除了将负极[6]比较例1、5、7、9的非水电解液换成《非水电解液的制作3》项中制作的非水电解液以外,分别按照同样的方法制作电池,并进行评价。结果示于负极[6]表2。Except that the non-aqueous electrolytic solution of negative electrode [6] comparative examples 1, 5, 7, 9 is changed into the non-aqueous electrolytic solution made in the "making 3 of non-aqueous electrolytic solution", the battery is made in the same way respectively, and Make an evaluation. The results are shown in Table 2 of the negative electrode [6].
负极[6]《电池的评价》Negative electrode [6] "Battery Evaluation"
(容量测定)(capacity measurement)
对未经充放电循环的新电池,在25℃、4.1V~3.0V的电压范围进行5个循环的初期充放电(电压范围4.1V~3.0V)。将此时的第5个循环的0.2C(将1小时放电额定容量的电流值作为1C,所述额定容量取决于1小时率(one-hour-rate)的放电容量,下同)放电容量作为初期容量。For a new battery that has not undergone charge-discharge cycles, 5 cycles of initial charge-discharge (voltage range 4.1V-3.0V) were performed at 25°C in a voltage range of 4.1V-3.0V. The 0.2C discharge capacity of the 5th cycle at this time (the current value of the 1-hour discharge rated capacity is taken as 1C, and the rated capacity depends on the discharge capacity of the 1-hour-rate (one-hour-rate), the same below) discharge capacity as initial capacity.
(输出功率测定1)(Output power measurement 1)
在25℃的环境下,通过0.2C的恒定电流进行150分钟充电,在-30℃环境下静置3小时,然后分别以0.1C、0.3C、1.0C、3.0C、5.0C放电10秒钟,测定第10秒的电压。将电流-电压直线和下限电压(3V)围成的三角形的面积作为初期低温输出功率(W)。At 25°C, charge with a constant current of 0.2C for 150 minutes, stand at -30°C for 3 hours, and then discharge at 0.1C, 0.3C, 1.0C, 3.0C, and 5.0C for 10 seconds , Measure the voltage at the 10th second. The area of the triangle surrounded by the current-voltage line and the lower limit voltage (3V) is taken as the initial low temperature output power (W).
(输出功率测定2)(Output power measurement 2)
在输出功率测定1之后,实施1小时4.1V的低电压充电,然后将电池转移到25℃的环境下,15分钟之后,以0.1C、0.3C、1.0C、3.0C、5.0C放电10秒钟,测定第10秒的电压。将电流-电压直线和下限电压(3V)围成的三角形的面积作为温度上升时输出功率(W)。After output power measurement 1, implement low-voltage charging at 4.1V for 1 hour, then transfer the battery to an environment at 25°C, and after 15 minutes, discharge at 0.1C, 0.3C, 1.0C, 3.0C, and 5.0C for 10 seconds clock, measure the voltage at the 10th second. The area of the triangle surrounded by the current-voltage line and the lower limit voltage (3V) is taken as the output power (W) when the temperature rises.
由输出功率测定1和输出功率测定2的结果通过下述计算式计算出温度适应输出功率提高率(%)。From the results of the output measurement 1 and the output measurement 2, the temperature adaptation output improvement rate (%) was calculated by the following calculation formula.
温度适应输出功率提高率(%)Temperature adaptation output power increase rate (%)
=[(温度上升时输出功率(W)/初期低温输出功率(W))-1]×100=[(Output power when temperature rises (W)/Initial low temperature output power (W))-1]×100
负极[6]表2中的阻抗Rct和双层容量Cdl是对输出功率有贡献的参数之一,阻抗Rct的值越小,或者双层容量Cdl的值越大,则输出功率越有改善的倾向。另外,“阻抗Rct”和“双层容量Cdl”通过说明阻抗的部分所记载的方法求出。The impedance Rct and the double-layer capacity Cdl in Table 2 of the negative electrode [6] are one of the parameters that contribute to the output power. The smaller the value of the impedance Rct, or the larger the value of the double-layer capacity Cdl, the more the output power will be improved. tendency. In addition, "impedance Rct" and "double layer capacity Cdl" were obtained by the method described in the section describing impedance.
负极[6]表2Negative electrode [6] Table 2
[表47][Table 47]
由负极[6]表2的结果可知,通过含有二氟磷酸锂盐、甲磺酸三甲基甲硅烷基酯、六甲基环三硅氧烷,并且含有圆形度为0.85以上、广角X射线衍射法测定的(002)面的面间距(d002)低于0.337、且拉曼R值为0.12~0.8的石墨质碳粒子作为负极活性物质,可以使-30℃的低温状态下的输出功率随着温度上升的恢复得以飞跃性地加快。From the results in Table 2 of the negative electrode [6], it can be seen that by containing lithium difluorophosphate, trimethylsilyl methanesulfonate, and hexamethylcyclotrisiloxane, and containing a circularity of 0.85 or more, a wide-angle X The graphitic carbon particles whose interplanar distance (d002) of the (002) plane measured by the ray diffraction method is less than 0.337 and whose Raman R value is 0.12 to 0.8 are used as the negative electrode active material, which can increase the output power at a low temperature of -30°C. Recovery is dramatically accelerated with increasing temperature.
负极[7]<负极的制作>Negative electrode [7] <Production of negative electrode>
(负极的制作1)(Making of Negative Electrode 1)
使用Si和C的混合物(Si和C的面积比大致为100比9的圆板)作为靶材料,使用平均表面粗糙度(Ra)为0.2μm、拉伸强度为280N/mm2、0.2%耐力为220N/mm2、厚度为18μm的电解铜箔作为集电体基板,采用直流溅射装置(岛津制作所公司制造的“HSM-52”)进行45分钟活性物质薄膜的成膜,得到薄膜负极(1)。A mixture of Si and C (circular plate with an area ratio of Si and C of approximately 100:9) was used as the target material, and the average surface roughness (Ra) was 0.2 μm, the tensile strength was 280 N/mm 2 , and the tensile strength was 0.2%. Electrolytic copper foil with a thickness of 220 N/mm 2 and a thickness of 18 μm was used as a current collector substrate, and a DC sputtering device ("HSM-52" manufactured by Shimadzu Corporation) was used to form a thin film of active material for 45 minutes to obtain a thin film negative pole (1).
此时,集电体基板安装在水冷却的夹具上,保持在约25℃,将容器预先真空抽吸到4×10-4Pa,在容器内流通40sccm高纯度的氩气,调节主阀的开度成为1.6Pa的氛围后,在功率密度4.7W/cm2、沉积速度(成膜速度)约1.8nm/秒(0.108μm/分)下进行成膜。该溅射气体的氧浓度为0.0010%。另外,为了除去电解铜箔表面的氧化膜,在形成薄膜之前进行反溅射并进行基板表面的蚀刻。At this time, the current collector substrate was mounted on a water-cooled jig and kept at about 25°C. The container was pre-vacuum-pumped to 4× 10-4 Pa, and 40 sccm of high-purity argon gas was circulated in the container, and the main valve was adjusted. After the opening degree became an atmosphere of 1.6 Pa, film formation was performed at a power density of 4.7 W/cm 2 and a deposition rate (film formation rate) of about 1.8 nm/sec (0.108 μm/min). The oxygen concentration of the sputtering gas was 0.0010%. In addition, in order to remove the oxide film on the surface of the electrolytic copper foil, reverse sputtering and etching of the substrate surface are performed before thin film formation.
通过对得到的薄膜负极(1)的薄膜的截面进行扫描型电子显微镜(SEM)观察,成膜后的薄膜的膜厚为6μm(参照图1(a))。另外,按照下述方法用XPS进行薄膜的组成分析时,薄膜中含有元素C为24原子%,SiC中相对于元素C浓度的C浓度比Q(C)相当于0.49。另外,如果用原子浓度比来表示,则Si/C/O=1.00/0.33/0.04。另外,按照下述方法采用拉曼测定求出薄膜的拉曼值时,RC=0.05、未检测出RSC=sc的峰、RS=0.55。另外,按照下述方法进行薄膜的X射线衍射测定时,未检测出明确的SiC的峰,XIsz=0.38。结果示于负极[7]表1。Scanning electron microscope (SEM) observation of the cross-section of the thin film of the obtained thin film negative electrode (1) revealed that the film thickness of the formed thin film was 6 μm (see FIG. 1( a )). In addition, when the composition analysis of the thin film was performed by XPS according to the following method, the element C contained in the thin film was 24 atomic %, and the C concentration ratio Q(C) to the element C concentration in SiC corresponded to 0.49. In addition, when represented by an atomic concentration ratio, Si/C/O=1.00/0.33/0.04. In addition, when the Raman value of the thin film was obtained by Raman measurement according to the following method, RC=0.05, the peak of RSC=sc was not detected, and RS=0.55. In addition, when X-ray diffraction measurement of the thin film was performed according to the following method, no clear peak of SiC was detected, and XIsz=0.38. The results are shown in Table 1 of the negative electrode [7].
此外,按照下述方法采用电子探针显微分析仪(EPMA)测定薄膜中Si在膜厚方向的质量浓度分布时,如图1(b)所示,Si的最大值或最小值与平均值之差(绝对值)为25%以内,Si实质上从集电体上连续地成膜。另外,测定薄膜中的元素C的分布时,如图1(c)所示,元素C在Si薄膜中以1μm以下的大小均匀分布。In addition, when the electron probe microanalyzer (EPMA) is used to measure the mass concentration distribution of Si in the film thickness direction in the film according to the following method, as shown in Figure 1(b), the maximum or minimum value and the average value of Si If the difference (absolute value) is within 25%, Si is formed into a film substantially continuously from the current collector. In addition, when the distribution of element C in the thin film was measured, as shown in FIG. 1( c ), element C was uniformly distributed in the Si thin film with a size of 1 μm or less.
(负极的制作2)(Making of Negative Electrode 2)
除了靶材料使用Si以外,与(负极的制作1)同样地成膜活性物质薄膜,制作薄膜负极(2)。Except for using Si as the target material, an active material thin film was formed in the same manner as in (Preparation of Negative Electrode 1), and a thin-film negative electrode (2) was produced.
通过对得到的薄膜负极(2)的薄膜的截面进行扫描型电子显微镜(SEM)观察,成膜后的薄膜的膜厚为6μm。另外,进行薄膜的组成分析时,薄膜中不含元素C、N,用原子浓度比表示时,Si/O=1.00/0.02。另外,求出薄膜的拉曼值时,未检测出RC=c的峰、未检测出RSC=sc的峰、RS=0.30。结果示于负极[7]表1。Observation with a scanning electron microscope (SEM) of the cross section of the thin film of the obtained thin film negative electrode (2) revealed that the thickness of the thin film after film formation was 6 μm. In addition, when the composition analysis of the thin film is carried out, the elements C and N are not contained in the thin film, and Si/O=1.00/0.02 when represented by the atomic concentration ratio. In addition, when the Raman value of the thin film was obtained, the peak of RC=c was not detected, the peak of RSC=sc was not detected, and RS=0.30. The results are shown in Table 1 of the negative electrode [7].
<XPS测定><XPS measurement>
作为X射线光电分光法测定,使用X射线光电分光器(例如,ulvac-phi公司制造的“ESCA”),将薄膜负极放置在试料台上并使表面平坦,以铝的Kα射线作为X射线源,一边进行Ar溅射一边进行深度曲线测定。得到达到浓度一定的深度(例如200nm)下的Si2p(90~110eV)、C1s(280~300eV)和O1s(525~545eV)的谱图。将得到的C1s的峰顶设定为284.5eV进行带电补偿,求出Si2p、C1s和O1s的谱图的峰面积,再乘以装置感度系数,分别计算出Si、C和O的原子浓度。由得到的该Si、O和C的原子浓度计算出原子浓度比Si/C/O(Si原子浓度/C原子浓度/O原子浓度),将其定义为薄膜的组成值Si/C/O。As X-ray photoelectric spectrometry measurement, use an X-ray photoelectric spectrometer (for example, "ESCA" manufactured by ulvac-phi Co., Ltd.), place a thin-film negative electrode on a sample table and make the surface flat, and use Kα rays of aluminum as X-rays. Source, depth profile measurement was performed while performing Ar sputtering. Obtain the spectra of Si2p (90-110eV), C1s (280-300eV) and O1s (525-545eV) at a certain concentration depth (for example, 200nm). Set the peak top of the obtained C1s to 284.5eV to perform charge compensation, obtain the peak areas of the spectra of Si2p, C1s and O1s, and multiply them by the device sensitivity coefficient to calculate the atomic concentrations of Si, C and O, respectively. The atomic concentration ratio Si/C/O (Si atomic concentration/C atomic concentration/O atomic concentration) was calculated from the obtained atomic concentrations of Si, O, and C, and this was defined as the thin film composition value Si/C/O.
<拉曼测定><Raman measurement>
作为拉曼测定,使用拉曼分光器(例如日本分光社制造的“拉曼分光器”),将薄膜负极安装在测定池中,测定是对池内的试样表面照射氩离子激光来进行的。For Raman measurement, a Raman spectrometer (for example, "Raman spectrometer" manufactured by JASCO Corporation) is used, a thin-film negative electrode is installed in a measurement cell, and the measurement is performed by irradiating the surface of the sample in the cell with an argon ion laser.
另外,这里的拉曼测定条件如下。In addition, the Raman measurement conditions here are as follows.
·氩离子激光波长:514.5nmArgon ion laser wavelength: 514.5nm
·试样上的激光功率:15~40mW·Laser power on the sample: 15~40mW
·分辨率:10~20cm-1 ·Resolution: 10~20cm -1
·测定范围:200cm-1~1900cm-1 · Measuring range: 200cm -1 ~ 1900cm -1
·光滑处理:单纯平均、卷积15点·Smooth processing: simple average, convolution 15 points
<X射线衍射测定><X-ray Diffraction Measurement>
作为X射线衍射测定,使用理学公司制造的“RINT2000PC”,将薄膜负极安装在测定池中,使用out-of-plane法进行2θ=10~70度范围的测定。本底补偿是将2θ=15~20度附近和40~45度附近用直线进行连接而进行的。As the X-ray diffraction measurement, "RINT2000PC" manufactured by Rigaku Corporation was used, and the thin-film negative electrode was set in the measurement cell, and the measurement in the range of 2θ=10 to 70 degrees was performed using the out-of-plane method. The background compensation is performed by connecting the vicinity of 2θ=15 to 20 degrees and the vicinity of 40 to 45 degrees with a straight line.
<EPMA测定><EPMA measurement>
作为采用EPMA测定的膜厚方向的质量浓度分布或薄膜截面的分布分析,使用电子探针显微分析仪(JEOL公司制造的“JXA-8100”),不进行树脂包埋而对用切片机制成截面的薄膜负极进行从集电体到薄膜表面的元素分析。求出膜厚方向的质量浓度分布时,使用将测定的元素的总和换算成100%而得到的值,求出Si在膜厚方向的质量浓度分布。As the mass concentration distribution in the film thickness direction measured by EPMA or the distribution analysis of the film cross section, an electron probe microanalyzer ("JXA-8100" manufactured by JEOL Co., Ltd.) was used to make a microtome without resin embedding. Cross-sectional thin film anodes were subjected to elemental analysis from the current collector to the film surface. When obtaining the mass concentration distribution in the film thickness direction, the value obtained by converting the sum of the measured elements into 100% is used to obtain the mass concentration distribution of Si in the film thickness direction.
负极[7]表1Negative electrode [7] Table 1
[表48][Table 48]
负极[7]表1中,(Si)不相当于元素Z。Negative electrode [7] In Table 1, (Si) does not correspond to element Z.
负极[7]<正极的制作>Negative electrode [7] <Production of positive electrode>
(正极的制作1)(Making of positive electrode 1)
在N-甲基吡咯烷酮溶剂中混合90质量%作为正极活性物质的钴酸锂(LiCoO2)、5质量%的作为导电材料的乙炔黑和5质量%作为粘合剂的聚偏氟乙烯(PVdF),制成浆料。将得到的浆料涂布在厚度15μm的铝箔一个面上,并干燥,用压制机压延成厚度80μm。Mix 90% by mass of lithium cobaltate (LiCoO 2 ) as a positive electrode active material, 5% by mass of acetylene black as a conductive material, and 5% by mass of polyvinylidene fluoride (PVdF ) to make slurry. The obtained slurry was coated on one side of an aluminum foil having a thickness of 15 μm, dried, and rolled to a thickness of 80 μm with a press.
负极[7]<非水电解液的制作>Negative electrode [7] <Preparation of non-aqueous electrolyte>
(非水电解液的制作1)(Preparation of non-aqueous electrolyte solution 1)
在干燥的氩氛围下,在碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的混合物(体积比3∶3∶4)中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。另外,还含有0.3质量%的二氟磷酸锂盐(LiPO2F2)。Under a dry argon atmosphere, in a mixture of ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) (volume ratio 3:3:4), at a concentration of 1mol/L Dissolve well-dried lithium hexafluorophosphate (LiPF 6 ). In addition, 0.3% by mass of lithium difluorophosphate (LiPO 2 F 2 ) was contained.
(非水电解液的制作2)(Preparation of non-aqueous electrolyte solution 2)
在干燥的氩氛围下,在碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的混合物(体积比3∶3∶4)中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。另外,还含有0.3质量%的甲磺酸三甲基甲硅烷基酯。Under a dry argon atmosphere, in a mixture of ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) (volume ratio 3:3:4), at a concentration of 1mol/L Dissolve well-dried lithium hexafluorophosphate (LiPF 6 ). In addition, 0.3% by mass of trimethylsilyl methanesulfonate was contained.
(非水电解液的制作3)(Preparation of non-aqueous electrolyte solution 3)
在干燥的氩氛围下,在碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的混合物(体积比3∶3∶4)中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。另外,还含有0.3质量%的六甲基环三硅氧烷。Under a dry argon atmosphere, in a mixture of ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) (volume ratio 3:3:4), at a concentration of 1mol/L Dissolve well-dried lithium hexafluorophosphate (LiPF 6 ). In addition, 0.3% by mass of hexamethylcyclotrisiloxane was contained.
(非水电解液的制作4)(Preparation of non-aqueous electrolyte solution 4)
在干燥的氩氛围下,在碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的混合物(体积比3∶3∶4)中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。Under a dry argon atmosphere, in a mixture of ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) (volume ratio 3:3:4), at a concentration of 1mol/L Dissolve well-dried lithium hexafluorophosphate (LiPF 6 ).
负极[7]<锂二次电池的制作>Negative electrode [7] <Production of lithium secondary battery>
将薄膜负极和正极冲孔成10mmφ,在110℃下进行真空干燥后,转移到手套工作箱内,在氩氛围下通过冲孔成14mmφ的聚乙烯隔板将正极和负极对置,并加入非水电解液,制作2032型硬币电池(锂二次电池)。The negative and positive electrodes of the film were punched into 10mmφ, and after vacuum drying at 110°C, they were transferred to a glove box. Water electrolyte, making 2032 type coin battery (lithium secondary battery).
负极[7]实施例1Negative pole [7] embodiment 1
使用(负极的制作1)项中制作的负极、(正极的制作)项中制作的正极以及(非水电解液的制作1)项中制作的非水电解液,通过《锂二次电池的制作》项中的方法制作硬币电池。通过下述<电池评价>项中所述的方法求出该二次电池的电池特性。Using the negative electrode prepared in (Making of Negative Electrode 1), the positive electrode prepared in (Making of Positive Electrode) and the nonaqueous electrolyte prepared in (Making of Nonaqueous Electrolyte 1), pass the "Making of Lithium Secondary Battery" "The method in item makes coin battery. The battery characteristics of the secondary battery were determined by the method described in the following item <Battery Evaluation>.
负极[7]实施例2Negative pole [7] embodiment 2
除了将负极[7]实施例1的非水电解液换成(非水电解液的制作2)项中制作的非水电解液以外,与负极[7]实施例1同样地制作电池,并采用<电池的评价>项记载的方法进行电池评价。结果示于负极[7]表2。Except that the non-aqueous electrolytic solution of the negative pole [7] embodiment 1 is changed into the non-aqueous electrolytic solution made in the item (making 2 of the non-aqueous electrolytic solution), the battery is made in the same way as the negative pole [7] embodiment 1, and adopts The battery was evaluated by the method described in the item <Battery Evaluation>. The results are shown in Table 2 of the negative electrode [7].
负极[7]实施例3Negative electrode [7] Example 3
除了将负极[7]实施例1的非水电解液换成(非水电解液的制作3)项中制作的非水电解液以外,与负极[7]实施例1同样地制作电池,并采用<电池的评价>项记载的方法进行电池评价。结果示于负极[7]表2。Except that the non-aqueous electrolytic solution of the negative pole [7] embodiment 1 is changed into the non-aqueous electrolytic solution made in the item (making 3 of the non-aqueous electrolytic solution), the battery is made in the same way as the negative pole [7] embodiment 1, and adopts The battery was evaluated by the method described in the item <Battery Evaluation>. The results are shown in Table 2 of the negative electrode [7].
负极[7]比较例1Negative electrode [7] Comparative example 1
除了将负极[7]实施例1的非水电解液换成(非水电解液的制作4)项中制作的非水电解液以外,与负极[7]实施例1同样地制作电池,并采用<电池的评价>项记载的方法进行电池评价。结果示于负极[7]表2。Except that the non-aqueous electrolytic solution of the negative pole [7] embodiment 1 is changed into the non-aqueous electrolytic solution made in the item (making 4 of the non-aqueous electrolytic solution), the battery is made in the same way as the negative pole [7] embodiment 1, and adopts The battery was evaluated by the method described in the item <Battery Evaluation>. The results are shown in Table 2 of the negative electrode [7].
负极[7]比较例2Negative electrode [7] Comparative Example 2
除了将负极[7]实施例1的负极换成(负极的制作2)项中制作的电极以外,与负极[7]实施例1同样地制作电池,并采用<电池的评价>项记载的方法进行电池评价。结果示于负极[7]表2。Except that the negative electrode of negative electrode [7] Example 1 was replaced with the electrode produced in item (Making of Negative Electrode 2), a battery was produced in the same manner as negative electrode [7] Example 1, and the method described in the item <Battery Evaluation> was adopted. Perform battery evaluation. The results are shown in Table 2 of the negative electrode [7].
负极[7]比较例3Negative electrode [7] Comparative example 3
除了将负极[7]比较例2的非水电解液换成(非水电解液的制作2)项中制作的非水电解液以外,与负极[7]比较例2同样地制作电池,并采用<电池的评价>项记载的方法进行电池评价。结果示于负极[7]表2。Except that the non-aqueous electrolytic solution of the negative pole [7] comparative example 2 is changed into the non-aqueous electrolytic solution made in the item (making 2 of the non-aqueous electrolytic solution), the battery is made in the same way as the negative pole [7] comparative example 2, and adopts The battery was evaluated by the method described in the item <Battery Evaluation>. The results are shown in Table 2 of the negative electrode [7].
负极[7]比较例4Negative electrode [7] Comparative example 4
除了将负极[7]比较例2的非水电解液换成(非水电解液的制作3)项中制作的非水电解液以外,与负极[7]比较例2同样地制作电池,并采用<电池的评价>项记载的方法进行电池评价。结果示于负极[7]表2。Except that the non-aqueous electrolytic solution of the negative pole [7] comparative example 2 is changed into the non-aqueous electrolytic solution made in (making 3 of the non-aqueous electrolytic solution), the battery is made in the same way as the negative pole [7] comparative example 2, and adopts The battery was evaluated by the method described in the item <Battery Evaluation>. The results are shown in Table 2 of the negative electrode [7].
负极[7]比较例5Negative electrode [7] Comparative Example 5
除了将负极[7]比较例2的非水电解液换成(非水电解液的制作4)项中制作的非水电解液以外,与负极[7]比较例2同样地制作电池,并采用《电池的评价》项记载的方法进行电池评价。结果示于负极[7]表2。Except that the non-aqueous electrolytic solution of the negative pole [7] comparative example 2 is changed into the non-aqueous electrolytic solution made in the item (making 4 of the non-aqueous electrolytic solution), the battery is made in the same way as the negative pole [7] comparative example 2, and adopts The method described in "Battery Evaluation" item was used for battery evaluation. The results are shown in Table 2 of the negative electrode [7].
负极[7]<电池的评价>Negative electrode [7] <Evaluation of battery>
按照下述方法,对<锂二次电池的制作>项中制作的硬币电池进行放电容量的评价、充电接受性的测定。The evaluation of the discharge capacity and the measurement of the charge acceptance were performed on the coin cell produced in the section of <Preparation of Lithium Secondary Battery> according to the following methods.
《放电容量的评价》"Evaluation of Discharge Capacity"
以1.23mA/cm2的电流密度对锂对电极充电到4.2V,再以4.2V的恒定电压充电到电流值达到0.123mA,在负极中掺杂锂之后,以1.23mA/cm2的电流密度对锂对电极进行放电直到2.5V,将该充放电循环反复进行5次循环,将第3~5循环的放电的平均值作为放电容量。另外,作为单位质量的放电容量的情况下,活性物质质量可通过由负极质量减去冲孔成相同面积的铜箔的质量而求得,并按照下式计算。Charge the lithium counter electrode to 4.2V at a current density of 1.23mA/ cm2 , and then charge it at a constant voltage of 4.2V until the current value reaches 0.123mA. After doping the negative electrode with lithium, charge it at a current density of 1.23mA/ cm2 The lithium counter electrode was discharged to 2.5 V, and this charge-discharge cycle was repeated 5 times, and the average value of the discharges in the 3rd to 5th cycles was taken as the discharge capacity. In addition, in the case of the discharge capacity per unit mass, the mass of the active material can be obtained by subtracting the mass of copper foil punched into the same area from the mass of the negative electrode, and calculated according to the following formula.
放电容量(mAh/g)Discharge capacity (mAh/g)
=[第3~5循环的平均放电容量(mAh)]/[活性物质质量(g)]=[Average discharge capacity of the 3rd to 5th cycle (mAh)]/[Active material mass (g)]
活性物质质量(g)=负极质量(g)-相同面积的铜箔质量(g)Active material mass (g) = negative electrode mass (g) - copper foil mass (g) with the same area
《充电接受性的测定》"Determination of Charge Acceptance"
在25℃的环境下,将以0.2C(将1小时放电额定容量的电流值作为1C,所述额定容量取决于1小时率(one-hour-rate)的放电容量,下同)充电到4.2V时的容量作为0.2C充电容量,再以0.2C放电到2.5V,然后以1C充电到4.2V,将此时的容量作为1C充电容量。由得到的结果通过下述式子求出充电接受性。In an environment of 25°C, it will be charged to 4.2C at 0.2C (the current value of the rated capacity of 1 hour discharge is taken as 1C, and the rated capacity depends on the discharge capacity of 1 hour rate (one-hour-rate), the same below). The capacity at V is taken as the 0.2C charging capacity, and then discharged to 2.5V at 0.2C, and then charged to 4.2V at 1C, and the capacity at this time is taken as the 1C charging capacity. The charge acceptance was calculated|required from the obtained result by the following formula.
充电接受性(%)=100×[1C充电容量]/[0.2C充电容量]Charging acceptance (%) = 100×[1C charging capacity]/[0.2C charging capacity]
负极[7]表2Negative electrode [7] Table 2
[表49][Table 49]
由负极[7]表2的结果可知,在非水电解液中含有二氟磷酸锂盐、甲磺酸三甲基甲硅烷基酯或六甲基环三硅氧烷,并且使用包含多元素的负极活性物质(C’)作为负极活性物质的锂二次电池,可以使充电接受性得以飞跃性地提高,所述包含多元素的负极活性物质(C’)含有至少一种锂吸留金属(A’)和/或锂吸留合金(B’),并且含有选自C或N中的至少一种元素(元素Z)。From the results of the negative electrode [7] Table 2, it can be seen that lithium difluorophosphate, trimethylsilyl methanesulfonate or hexamethylcyclotrisiloxane are contained in the non-aqueous electrolyte, and using polyelement-containing The negative electrode active material (C') as the lithium secondary battery of the negative electrode active material can dramatically improve the charge acceptance, and the multi-element negative electrode active material (C') contains at least one lithium storage metal ( A') and/or lithium storage alloy (B'), and contains at least one element (element Z) selected from C or N.
负极[8][负极活性物质的制作]Negative electrode[8][Production of negative electrode active material]
(负极活性物质的制作1)(Production of negative electrode active material 1)
使用混合系统((株)奈良机械制作所制造的混合系统NHS-1型)以处理量90g、转子圆周速度60m/s、处理时间3分钟的条件对天然石墨粉末(d002:0.336nm、Lc:100nm以上、拉曼R值:0.11、振实密度:0.46g/cm3、真密度:2.27g/cm3、体积基准平均粒径:35.4μm)进行处理,由此进行球形化,另外,为了防止混入粗大粒子,使用ASTM400目的筛子反复筛5次。将这样得到的负极活性物质作为碳质物质(A)。通过反复进行该操作,确保了电池制作所必需的量。Use a mixing system (the mixing system NHS-1 type manufactured by Nara Machinery Manufacturing Co., Ltd.) to treat natural graphite powder (d002: 0.336nm, Lc: 100nm or more, Raman R value: 0.11, tap density: 0.46g/cm 3 , true density: 2.27g/cm 3 , volume-based average particle size: 35.4μm) to spheroidize, and to To prevent mixing of coarse particles, use an ASTM400 mesh sieve to sieve repeatedly 5 times. The negative electrode active material thus obtained is referred to as a carbonaceous material (A). By repeating this operation, the quantity necessary for battery production is secured.
(负极活性物质的制作2)(Production of negative electrode active material 2)
用风力分级装置对市售的鳞片状天然石墨粉末除去微粉,为了防止得到的粉末中混入粗大粒子,使用ASTM400目的筛子反复筛5次。将这样得到的负极活性物质作为碳质物质(B)。The commercially available flaky natural graphite powder was removed with a wind classifier, and in order to prevent the obtained powder from being mixed with coarse particles, the ASTM400 mesh sieve was used to repeatedly sieve 5 times. The negative electrode active material thus obtained is referred to as a carbonaceous material (B).
(负极活性物质的制作3)(Production of negative electrode active material 3)
将喹啉不溶成分为0.05质量%以下的煤焦油沥青在反应炉中于460℃热处理10小时,得到软化点385℃的具有熔融性的块状碳化处理前体。将得到的块状碳化处理前体装入到金属制的容器中,在箱形的电炉中、氮气流通下于1000℃进行2小时热处理。用粗粉碎机(吉田制作所制造的辊式压碎机)将得到的无定形的块粉碎,再用微粉碎机(matsubo公司制造的涡轮式磨)进行微粉碎,然后用风力分级装置除去微粉,为了防止得到的粉末中混入粗大粒子,使用ASTM400目的筛子反复筛5次,得到体积基准粒径为9μm的无定形粉末。将这样得到的负极活性物质作为碳质物质(C)。Coal tar pitch with a quinoline insoluble content of 0.05% by mass or less was heat-treated in a reaction furnace at 460°C for 10 hours to obtain a fusible bulk carbonized precursor with a softening point of 385°C. The obtained massive carbonized precursor was put into a metal container, and heat-treated at 1000° C. for 2 hours in a box-shaped electric furnace under a flow of nitrogen gas. The resulting amorphous lump was pulverized with a coarse pulverizer (roller crusher manufactured by Yoshida Seisakusho), finely pulverized with a fine pulverizer (turbine mill manufactured by Matsubo Co., Ltd.), and then fine powder was removed with a wind classifier , in order to prevent the obtained powder from being mixed with coarse particles, use an ASTM400 mesh sieve to repeatedly sieve 5 times to obtain an amorphous powder with a volume reference particle size of 9 μm. The negative electrode active material thus obtained is referred to as a carbonaceous material (C).
(负极活性物质的制作4)(Production of negative electrode active material 4)
在碳质物质(A)中混合石脑油热解时得到的石油类重油,在非活性气体中实施1300℃的碳化处理,然后,通过对烧结物进行分级处理得到碳质物质,所述碳质物质是在碳质物质(A)粒子表面上包覆了具有不同结晶性的碳质物质而得到的碳质物质。分级处理时,为了防止混入粗大粒子,使用ASTM400目的筛子反复筛5次,得到碳质物质(D)。从残炭率可以确定,得到的负极活性物质粉末被相对于95重量份石墨为5重量的来自于石油类重油的碳质包覆。Petroleum heavy oil obtained during the pyrolysis of naphtha is mixed with the carbonaceous substance (A), carbonized at 1300°C in an inert gas, and then the carbonaceous substance is obtained by classifying the sintered product. The carbonaceous substance is a carbonaceous substance obtained by coating the carbonaceous substance (A) particle surface with a carbonaceous substance having a different crystallinity. During the classification process, in order to prevent mixing of coarse particles, the ASTM400 mesh sieve was used to repeatedly sieve 5 times to obtain the carbonaceous material (D). It was confirmed from the carbon residue rate that the obtained negative electrode active material powder was coated with 5 parts by weight of carbon derived from petroleum heavy oil relative to 95 parts by weight of graphite.
(负极活性物质的制作5)(Production of negative electrode active material 5)
将80质量%碳质物质(A)和20质量%碳质物质(B)混合直至均匀,制成混合碳质物质(E)。80% by mass of the carbonaceous substance (A) and 20% by mass of the carbonaceous substance (B) were mixed until uniform to obtain a mixed carbonaceous substance (E).
(负极活性物质的制作6)(Production of negative electrode active material 6)
将95质量%碳质物质(A)和5质量%碳质物质(C)混合直至均匀,制成混合碳质物质(F)。95% by mass of the carbonaceous substance (A) and 5% by mass of the carbonaceous substance (C) were mixed until uniform to obtain a mixed carbonaceous substance (F).
(负极活性物质的制作7)(Production of negative electrode active material 7)
将80质量%碳质物质(D)和20质量%碳质物质(A)混合直至均匀,制成混合碳质物质(G)。80% by mass of the carbonaceous substance (D) and 20% by mass of the carbonaceous substance (A) were mixed until uniform to obtain a mixed carbonaceous substance (G).
用上述方法求出负极活性物质的制作1~7中制作的碳质物质(A)、(B)、(C)和混合碳质物质(E)、(F)、(G)的物性、形状等。结果归纳示于负极[8]表1中。The physical properties and shapes of the carbonaceous materials (A), (B), (C) and mixed carbonaceous materials (E), (F), (G) prepared in the production of negative electrode active materials 1 to 7 were obtained by the above method wait. The results are summarized in Table 1 of the negative electrode [8].
负极[8]表1Negative electrode [8] Table 1
负极[8][电池的制作]Negative electrode [8] [Battery production]
《正极的制作1》"Making of Positive Pole 1"
在N-甲基吡咯烷酮溶剂中混合90质量%作为正极活性物质的钴酸锂(LiCoO2)、5质量%的作为导电材料的乙炔黑和5质量%作为粘合剂的聚偏氟乙烯(PVdF),制成浆料。将得到的浆料涂布在厚度15μm的铝箔两面,并干燥,用压制机压延成厚度80μm,再将其切成具有宽100mm、长100mm大小的活性物质层和宽30mm的未涂布部分的形状,作为正极。此时的正极的活性物质的密度为2.35g/cm3。Mix 90% by mass of lithium cobaltate (LiCoO 2 ) as a positive electrode active material, 5% by mass of acetylene black as a conductive material, and 5% by mass of polyvinylidene fluoride (PVdF ) to make slurry. The obtained slurry was coated on both sides of an aluminum foil with a thickness of 15 μm, dried, rolled to a thickness of 80 μm with a press, and cut into an active material layer having a width of 100 mm and a length of 100 mm and an uncoated portion of a width of 30 mm. shape, as the positive electrode. The density of the active material of the positive electrode at this time was 2.35 g/cm 3 .
《负极的制作1》"Making of Negative Pole 1"
在98重量份负极活性物质中添加100重量份作为增稠剂的羧甲基纤维素钠的水性分散液(羧甲基纤维素钠的浓度为1质量%)、2重量份作为粘合剂的苯乙烯-丁二烯橡胶的水性分散液(苯乙烯-丁二烯橡胶的浓度为50质量%),用分散器进行混合,制成浆料。将得到的浆料涂布在厚度10μm的压延铜箔两面,并干燥,用压制机压延成厚度75μm,再将其切成具有宽104mm、长104mm大小的活性物质层和宽30mm的未涂布部分的形状,作为负极。此时的负极的活性物质的密度为1.35g/cm3。Add 100 parts by weight of an aqueous dispersion of sodium carboxymethylcellulose as a thickener (the concentration of sodium carboxymethylcellulose is 1% by mass) and 2 parts by weight of sodium carboxymethylcellulose as a binder in 98 parts by weight of the negative electrode active material. An aqueous dispersion of styrene-butadiene rubber (the concentration of styrene-butadiene rubber is 50% by mass) was mixed with a disperser to prepare a slurry. The obtained slurry was coated on both sides of a rolled copper foil with a thickness of 10 μm, dried, rolled to a thickness of 75 μm with a press, and then cut into an active material layer with a width of 104 mm and a length of 104 mm and an uncoated layer with a width of 30 mm. The shape of the part, as the negative pole. The density of the active material of the negative electrode at this time was 1.35 g/cm 3 .
《非水电解液的制作1》"Preparation of non-aqueous electrolyte 1"
在干燥的氩氛围下,在碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的混合物(体积比3∶3∶4)中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。另外,还含有0.3质量%的二氟磷酸锂盐(LiPO2F2)。Under a dry argon atmosphere, in a mixture of ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) (volume ratio 3:3:4), at a concentration of 1mol/L Dissolve well-dried lithium hexafluorophosphate (LiPF 6 ). In addition, 0.3% by mass of lithium difluorophosphate (LiPO 2 F 2 ) was contained.
《非水电解液的制作2》"The production of non-aqueous electrolyte 2"
在干燥的氩氛围下,在碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的混合物(体积比3∶3∶4)中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。另外,还含有0.3质量%的甲磺酸三甲基甲硅烷基酯。Under a dry argon atmosphere, in a mixture of ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) (volume ratio 3:3:4), at a concentration of 1mol/L Dissolve well-dried lithium hexafluorophosphate (LiPF 6 ). In addition, 0.3% by mass of trimethylsilyl methanesulfonate was contained.
《非水电解液的制作3》"The Production of Non-aqueous Electrolyte 3"
在干燥的氩氛围下,在碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的混合物(体积比3∶3∶4)中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。另外,还含有0.3质量%的六甲基环三硅氧烷。Under a dry argon atmosphere, in a mixture of ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) (volume ratio 3:3:4), at a concentration of 1mol/L Dissolve well-dried lithium hexafluorophosphate (LiPF 6 ). In addition, 0.3% by mass of hexamethylcyclotrisiloxane was contained.
《非水电解液的制作4》"The Production of Non-aqueous Electrolyte 4"
在干燥的氩氛围下,在碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的混合物(体积比3∶3∶4)中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。Under a dry argon atmosphere, in a mixture of ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) (volume ratio 3:3:4), at a concentration of 1mol/L Dissolve well-dried lithium hexafluorophosphate (LiPF 6 ).
《电池的制作1》"Battery Making 1"
将32片正极和33片负极交替配置,各电极之间夹入多孔聚乙烯片隔板(厚度为25μm),进行叠层。此时,使正极活性物质面与负极活性物质面相对,并且不会露出负极活性物质面之外。将该正极和负极各自的未涂布部分焊接,制作集电片,将电极组密封在电池罐(外部尺寸:120×110×10mm)中。然后,在装有电极组的电池罐中注入20mL非水电解液,使电极充分浸透,密封而制作方形电池。该电池的额定放电容量约为6安培小时(Ah),以10kHz交流法测定的直流电阻成分约为5毫欧(mΩ)。正极的电极面积的总和相对于电池的外壳表面积的总和之比为20.6。32 positive electrodes and 33 negative electrodes were arranged alternately, and a porous polyethylene sheet separator (thickness: 25 μm) was sandwiched between the electrodes for lamination. At this time, the surface of the positive electrode active material faces the surface of the negative electrode active material without protruding from the surface of the negative electrode active material. The respective uncoated parts of the positive and negative electrodes were welded to produce current collector sheets, and the electrode group was sealed in a battery can (outer dimensions: 120×110×10 mm). Then, 20 mL of non-aqueous electrolytic solution was poured into the battery can containing the electrode group, the electrodes were fully soaked, and sealed to produce a square battery. The rated discharge capacity of the battery is about 6 ampere hours (Ah), and the DC resistance component measured by the 10 kHz alternating current method is about 5 milliohms (mΩ). The ratio of the sum of the electrode areas of the positive electrode to the sum of the surface areas of the battery case was 20.6.
负极[8]实施例1Negative pole [8] embodiment 1
使用《负极的制作1》项的负极活性物质为混合碳质物质(E)而制作的负极、《正极的制作1》项中制作的正极以及《非水电解液的制作1》项中制作的非水电解液,通过《电池的制作1》项中的方法制作电池。对该电池进行下述《电池的评价》项中所述的电池评价。结果示于负极[8]表2。The negative electrode produced by using the negative electrode active material in "Making of Negative Electrode 1" as a mixed carbonaceous material (E), the positive electrode produced in "Making of Positive Electrode 1" and the one produced in "Making of Nonaqueous Electrolyte 1" Non-aqueous electrolyte, the battery is made by the method in "Battery Production 1". The batteries were subjected to the battery evaluations described in the item "Evaluation of Batteries" below. The results are shown in Table 2 of the negative electrode [8].
负极[8]实施例2Negative pole [8] embodiment 2
除了负极[8]实施例1的《负极的制作1》项的负极活性物质使用混合碳质物质(F)以外,与负极[8]实施例1同样地制作电池,并进行《电池的评价》项记载的电池评价。结果示于负极[8]表2。Except that the negative electrode active material of negative electrode [8] Example 1 of "Making of Negative Electrode 1" uses a mixed carbonaceous material (F), a battery is produced in the same manner as negative electrode [8] Example 1, and "Battery Evaluation" is carried out. Item recorded battery evaluation. The results are shown in Table 2 of the negative electrode [8].
负极[8]实施例3Negative electrode [8] Example 3
除了负极[8]实施例1的《负极的制作1》项的负极活性物质使用混合碳质物质(G)以外,与负极[8]实施例1同样地制作电池,并进行《电池的评价》项记载的电池评价。结果示于负极[8]表2。Except that the mixed carbonaceous material (G) was used as the negative electrode active material in the negative electrode [8] Example 1 of "Making of the Negative Electrode 1", a battery was produced in the same manner as in the negative electrode [8] Example 1, and "Evaluation of the Battery" was carried out. Item recorded battery evaluation. The results are shown in Table 2 of the negative electrode [8].
负极[8]实施例4~6Negative pole [8] Embodiment 4~6
除了将负极[8]实施例1~3的非水电解液换成《非水电解液的制作2》项中制作的非水电解液以外,分别按照同样的方法制作电池,并进行电池评价。结果示于负极[8]表2。Except that the non-aqueous electrolyte solution of Examples 1 to 3 of the negative electrode [8] was replaced with the non-aqueous electrolyte solution produced in the item "Preparation of Non-aqueous Electrolyte Solution 2", batteries were produced in the same way, and battery evaluation was performed. The results are shown in Table 2 of the negative electrode [8].
负极[8]实施例7~9Negative pole [8] Embodiment 7~9
除了将负极[8]实施例1~3的非水电解液换成《非水电解液的制作3》项中制作的非水电解液以外,分别按照同样的方法制作电池,并进行电池评价。结果示于负极[8]表2。Except that the non-aqueous electrolyte solution of Examples 1 to 3 of the negative electrode [8] was replaced with the non-aqueous electrolyte solution produced in the item "Preparation of Non-aqueous Electrolyte Solution 3", batteries were produced in the same way, and battery evaluation was performed. The results are shown in Table 2 of the negative electrode [8].
负极[8]比较例1Negative electrode [8] Comparative Example 1
除了将负极[8]实施例1的非水电解液换成《非水电解液的制作4》项中制作的非水电解液以外,与负极[8]实施例1同样地制作电池,并进行《电池的评价》项记载的电池评价。结果示于负极[8]表2。Except that the non-aqueous electrolytic solution of the negative pole [8] embodiment 1 is changed into the non-aqueous electrolytic solution made in the "making 4 of non-aqueous electrolytic solution" item, the battery is made in the same way as the negative pole [8] embodiment 1, and carried out Battery evaluation described in "Evaluation of Batteries". The results are shown in Table 2 of the negative electrode [8].
负极[8]比较例2Negative electrode [8] Comparative Example 2
除了将负极[8]实施例2的非水电解液换成《非水电解液的制作4》项中制作的非水电解液以外,与负极[8]实施例2同样地制作电池,并进行《电池的评价》项记载的电池评价。结果示于负极[8]表2。Except that the non-aqueous electrolytic solution of the negative pole [8] embodiment 2 is changed into the non-aqueous electrolytic solution made in the "making 4 of the non-aqueous electrolytic solution", the battery is made in the same way as the negative pole [8] embodiment 2, and carried out Battery evaluation described in "Evaluation of Batteries". The results are shown in Table 2 of the negative electrode [8].
负极[8]比较例3Negative electrode [8] Comparative Example 3
除了负极[8]比较例1的《负极的制作1》项的负极活性物质使用碳质物质(A)以外,与负极[8]比较例1同样地制作电池,并进行《电池的评价》项记载的电池评价。结果示于负极[8]表2。Except that the carbonaceous material (A) was used as the negative electrode active material in the negative electrode [8] Comparative Example 1 of "Preparation of Negative Electrode 1", a battery was produced in the same manner as in the negative electrode [8] Comparative Example 1, and the item "Evaluation of the Battery" was carried out. Documented battery evaluation. The results are shown in Table 2 of the negative electrode [8].
负极[8]比较例4Negative electrode [8] Comparative example 4
除了负极[8]比较例1的《负极的制作1》项的负极活性物质使用碳质物质(B)以外,与负极[8]比较例1同样地制作电池,并进行《电池的评价》项记载的电池评价。结果示于负极[8]表2。Except that the carbonaceous material (B) was used as the negative electrode active material in the negative electrode [8] Comparative Example 1 of "Preparation of the Negative Electrode 1", a battery was produced in the same manner as the negative electrode [8] Comparative Example 1, and the "Evaluation of the Battery" item was carried out. Documented battery evaluation. The results are shown in Table 2 of the negative electrode [8].
负极[8]比较例5Negative electrode [8] Comparative example 5
除了负极[8]比较例1的《负极的制作1》项的负极活性物质使用碳质物质(C)以外,与负极[8]比较例1同样地制作电池,并进行《电池的评价》项记载的电池评价。结果示于负极[8]表2。Except that the carbonaceous material (C) was used as the negative electrode active material in the negative electrode [8] Comparative Example 1 "Negative Electrode Production 1", a battery was produced in the same manner as the negative electrode [8] Comparative Example 1, and the "Battery Evaluation" item was carried out. Documented battery evaluation. The results are shown in Table 2 of the negative electrode [8].
负极[8]比较例6~8Negative electrode [8] Comparative examples 6-8
除了将负极[8]比较例3~5的非水电解液换成《非水电解液的制作1》项中制作的非水电解液以外,分别按照同样的方法制作电池,并进行评价。结果示于负极[8]表2。Batteries were produced and evaluated in the same manner, except that the non-aqueous electrolytes of Comparative Examples 3 to 5 of the negative electrode [8] were replaced with the non-aqueous electrolytes prepared in the item "Preparation of Non-aqueous Electrolyte 1". The results are shown in Table 2 of the negative electrode [8].
负极[8]比较例9~11Negative electrode [8] Comparative Examples 9-11
除了将负极[8]比较例3~5的非水电解液换成《非水电解液的制作2》项中制作的非水电解液以外,分别按照同样的方法制作电池,并进行评价。结果示于负极[8]表2。Batteries were produced and evaluated in the same manner, except that the non-aqueous electrolyte solutions of Comparative Examples 3 to 5 of the negative electrode [8] were replaced with the non-aqueous electrolyte solutions produced in the item "Preparation of Non-aqueous Electrolyte Solutions 2". The results are shown in Table 2 of the negative electrode [8].
负极[8]比较例12~14Negative electrode [8] Comparative Examples 12-14
除了将负极[8]比较例3~5的非水电解液换成《非水电解液的制作3》项中制作的非水电解液以外,分别按照同样的方法制作电池,并进行评价。结果示于负极[8]表2。Batteries were produced and evaluated in the same manner, except that the non-aqueous electrolyte solutions of Comparative Examples 3 to 5 of the negative electrode [8] were replaced with the non-aqueous electrolyte solutions produced in the item "Preparation of Non-aqueous Electrolyte Solutions 3". The results are shown in Table 2 of the negative electrode [8].
负极[8]《电池的评价》Negative electrode [8] "Battery Evaluation"
(容量测定)(capacity measurement)
对未经充放电循环的新电池,在25℃、4.1V~3.0V的电压范围进行5个循环的初期充放电(电压范围4.1V~3.0V)。将此时的第5个循环的0.2C(将1小时放电额定容量的电流值作为1C,所述额定容量取决于1小时率(one-hour-rate)的放电容量,下同)放电容量作为初期容量。For a new battery that has not undergone charge-discharge cycles, 5 cycles of initial charge-discharge (voltage range 4.1V-3.0V) were performed at 25°C in a voltage range of 4.1V-3.0V. The 0.2C discharge capacity of the 5th cycle at this time (the current value of the 1-hour discharge rated capacity is taken as 1C, and the rated capacity depends on the discharge capacity of the 1-hour-rate (one-hour-rate), the same below) discharge capacity as initial capacity.
(输出功率测定1)(Output power measurement 1)
在25℃的环境下,通过0.2C的恒定电流进行150分钟充电,在-30℃环境下静置3小时,然后分别以0.1C、0.3C、1.0C、3.0C、5.0C放电10秒钟,测定第10秒的电压。将电流-电压直线和下限电压(3V)围成的三角形的面积作为低温输出功率(W)。At 25°C, charge with a constant current of 0.2C for 150 minutes, stand at -30°C for 3 hours, and then discharge at 0.1C, 0.3C, 1.0C, 3.0C, and 5.0C for 10 seconds , Measure the voltage at the 10th second. The area of the triangle surrounded by the current-voltage line and the lower limit voltage (3V) is taken as the low-temperature output power (W).
(循环试验)(cycle test)
在被视为锂二次电池的实际使用上限温度的60℃的高温环境下进行循环试验。以2C的恒定电流恒定电压法充电至充电上限电压4.2V,然后以2C的恒定电流放电至放电终止电压3.0V,将该充放电循环作为1个循环,并反复进行该循环直至500循环。对于循环试验结束后的电池,在25℃环境下进行3个循环的充放电,将该第3个循环的0.2C放电容量作为循环后容量。由循环试验之前测定的初期容量和循环试验结束后测定的循环后容量按照下述计算式求出循环保持率。The cycle test was performed in a high-temperature environment of 60° C., which is regarded as the practical upper limit temperature of the lithium secondary battery. Charge to the charging upper limit voltage of 4.2V by the constant current and constant voltage method of 2C, and then discharge to the end-of-discharge voltage of 3.0V by the constant current of 2C. The charge-discharge cycle is regarded as one cycle, and this cycle is repeated until 500 cycles. For the battery after the cycle test, 3 cycles of charging and discharging were performed in an environment of 25° C., and the 0.2 C discharge capacity of the third cycle was taken as the capacity after cycle. The cycle retention rate was obtained from the initial capacity measured before the cycle test and the post-cycle capacity measured after the cycle test was completed according to the following calculation formula.
循环保持率(%)=100×循环后容量/初期容量Cycle retention (%) = 100 × capacity after cycle / initial capacity
负极[8]表2Negative electrode [8] Table 2
[表51][Table 51]
由负极[8]表2的结果可知,通过含有二氟磷酸锂盐、甲磺酸三甲基甲硅烷基酯、六甲基环三硅氧烷,并使用含有2种以上性质不同的负极活性物质的负极,循环特性和低温输出功率均良好。From the results in Table 2 of the negative electrode [8], it can be seen that by containing lithium difluorophosphate, trimethylsilyl methanesulfonate, and hexamethylcyclotrisiloxane, and using two or more negative electrodes with different properties The negative electrode of the material, cycle characteristics and low temperature output power are all good.
负极[9][10][负极活性物质的制作]Negative electrode[9][10][Production of negative electrode active material]
(负极活性物质的制作1)(Production of negative electrode active material 1)
使用球形化处理装置(奈良机械制作所制造的混合系统)以转速6500rpm对高纯度化处理过的中值粒径约150μm的鳞片状天然石墨(d002:0.336nm、Lc:100nm以上、拉曼R值:0.11、真密度:2.27g/cm3、灰分:0.05质量%)进行5分钟球形化处理,再使用风力式分级机(seishin企业公司制造的OMC-100)除去45质量%微粉,得到碳质物质(A)。Highly purified flaky natural graphite with a median diameter of about 150 μm (d002: 0.336 nm, Lc: 100 nm or more, Raman R value: 0.11, true density: 2.27g/cm 3 , ash content: 0.05% by mass) to spheroidize for 5 minutes, and then use an air classifier (OMC-100 manufactured by Seishin Enterprise Co., Ltd.) to remove 45% by mass of fine powder to obtain carbon Substance (A).
(负极活性物质的制作2)(Production of negative electrode active material 2)
将(负极活性物质的制作1)中制作的碳质物质(A)装入到石墨坩埚中,使用直接通电炉在非活性氛围下于3000℃进行5小时热处理,得到碳质物质(B)。The carbonaceous material (A) produced in (Production of Negative Electrode Active Material 1) was charged into a graphite crucible, and heat-treated at 3000° C. for 5 hours in an inert atmosphere using a direct electric furnace to obtain a carbonaceous material (B).
(负极活性物质的制作3)(Production of negative electrode active material 3)
在(负极活性物质的制作1)制作的碳质物质(A)中混合石脑油热解时得到的石油类重油,在非活性气体中实施1300℃的碳化处理,然后,通过对烧结物进行分级处理得到碳质物质,所述碳质物质是在碳质物质(A)粒子表面上包覆了具有不同结晶性的碳质物质而得到的碳质物质。分级处理时,为了防止混入粗大粒子,使用ASTM400目的筛子反复筛5次,得到碳质物质(C)。从残炭率可以确定,得到的负极活性物质粉末被相对于95重量份石墨为5重量的来自于石油类重油的碳质包覆。The petroleum-based heavy oil obtained during the pyrolysis of naphtha is mixed with the carbonaceous material (A) produced in (production of negative electrode active material 1), and carbonization treatment at 1300° C. is carried out in an inert gas, and then the sintered product is The classification process yields a carbonaceous substance in which carbonaceous substances having different crystallinity are coated on the surface of the carbonaceous substance (A) particles. During the classification process, in order to prevent mixing of coarse particles, the ASTM400 mesh sieve was used to repeatedly sieve 5 times to obtain carbonaceous material (C). It was confirmed from the carbon residue rate that the obtained negative electrode active material powder was coated with 5 parts by weight of carbon derived from petroleum heavy oil relative to 95 parts by weight of graphite.
(负极活性物质的制作4)(Production of negative electrode active material 4)
将喹啉不溶成分为0.05质量%以下的煤焦油沥青在反应炉中于460℃热处理10小时,得到软化点385℃的具有熔融性的块状碳化处理前体。将得到的块状碳化处理前体装入到金属制的容器中,在箱形的电炉中、氮气流通下于1000℃进行2小时热处理。用粗粉碎机(吉田制作所制造的辊式压碎机)将得到的无定形的块粉碎,再用微粉碎机(matsubo公司制造的涡轮式磨)进行微粉碎,得到体积基准平均粒径17μm的无定形粉末。为了防止得到的粉末中混入粗大粒子,使用ASTM400目的筛子反复筛5次。将这样得到的负极活性物质作为碳质物质(D)。Coal tar pitch with a quinoline insoluble content of 0.05% by mass or less was heat-treated in a reaction furnace at 460°C for 10 hours to obtain a fusible bulk carbonized precursor with a softening point of 385°C. The obtained massive carbonized precursor was put into a metal container, and heat-treated at 1000° C. for 2 hours in a box-shaped electric furnace under a flow of nitrogen gas. The obtained amorphous block was pulverized with a coarse pulverizer (roller crusher manufactured by Yoshida Seisakusho), and finely pulverized with a fine pulverizer (turbine mill manufactured by Matsubo Co., Ltd.) to obtain a volume-based average particle size of 17 μm of amorphous powder. In order to prevent coarse particles from being mixed into the obtained powder, the sieve was repeatedly sieved 5 times using an ASTM400 mesh sieve. The negative electrode active material thus obtained is referred to as a carbonaceous material (D).
用上述方法测定制作的碳质物质(A)、碳质物质(B)、碳质物质(C)、碳质物质(D)的形状和物性。结果示于负极[9][10]表1中。The shapes and physical properties of the produced carbonaceous substance (A), carbonaceous substance (B), carbonaceous substance (C) and carbonaceous substance (D) were measured by the method described above. The results are shown in Table 1 for negative electrodes [9][10].
负极[9][10]表1Negative electrode [9] [10] Table 1
[表52][Table 52]
负极[9][10][电池的制作]Negative electrode[9][10][Battery production]
《正极的制作1》"Making of Positive Pole 1"
在N-甲基吡咯烷酮溶剂中混合90质量%作为正极活性物质的钴酸锂(LiCoO2)、5质量%的作为导电材料的乙炔黑和5质量%作为粘合剂的聚偏氟乙烯(PVdF),制成浆料。将得到的浆料涂布在厚度15μm的铝箔两面,并干燥,用压制机压延成厚度80μm,再将其切成具有宽100mm、长100mm大小的正极活性物质层和宽30mm的未涂布部分的形状,作为正极。此时的正极的正极活性物质层的密度为2.35g/cm3。Mix 90% by mass of lithium cobaltate (LiCoO 2 ) as a positive electrode active material, 5% by mass of acetylene black as a conductive material, and 5% by mass of polyvinylidene fluoride (PVdF ) to make slurry. The obtained slurry was coated on both sides of an aluminum foil with a thickness of 15 μm, dried, rolled to a thickness of 80 μm with a press, and then cut into a positive electrode active material layer having a width of 100 mm and a length of 100 mm and an uncoated portion of a width of 30 mm. shape, as the positive electrode. The density of the positive electrode active material layer of the positive electrode at this time was 2.35 g/cm 3 .
《负极的制作1》"Making of Negative Pole 1"
在98重量份负极活性物质中添加100重量份作为增稠剂的羧甲基纤维素钠的水性分散液(羧甲基纤维素钠的浓度为1质量%)、2重量份作为粘合剂的苯乙烯-丁二烯橡胶的水性分散液(苯乙烯-丁二烯橡胶的浓度为50质量%),用分散器进行混合,制成浆料。将得到的浆料涂布在厚度10μm的压延铜箔两面,并干燥,用压制机压延成厚度75μm,再将其切成具有宽104mm、长104mm大小的负极活性物质层和宽30mm的未涂布部分的形状,作为负极。Add 100 parts by weight of an aqueous dispersion of sodium carboxymethylcellulose as a thickener (the concentration of sodium carboxymethylcellulose is 1% by mass) and 2 parts by weight of sodium carboxymethylcellulose as a binder in 98 parts by weight of the negative electrode active material. An aqueous dispersion of styrene-butadiene rubber (the concentration of styrene-butadiene rubber is 50% by mass) was mixed with a disperser to prepare a slurry. The obtained slurry is coated on both sides of a rolled copper foil with a thickness of 10 μm, dried, rolled into a thickness of 75 μm with a press, and then cut into a negative electrode active material layer with a width of 104 mm and a length of 104 mm and an uncoated layer with a width of 30 mm. The shape of the cloth part is used as the negative electrode.
此时的负极的负极活性物质层的密度为1.33~1.36g/cm3的范围(分别示于负极[9][10]表2的最右栏)。由上述可知,负极活性物质层中作为粘合剂的苯乙烯-丁二烯橡胶相对于整个负极活性物质层的含量为1质量%。另外,[(一面的负极活性物质层的厚度)/(集电体的厚度)]的值为75μm/10μm=7.5。The density of the negative electrode active material layer of the negative electrode at this time was in the range of 1.33 to 1.36 g/cm 3 (respectively shown in the rightmost column of Table 2 of negative electrodes [9] and [10]). From the above, it can be seen that the content of the styrene-butadiene rubber as a binder in the negative electrode active material layer is 1% by mass relative to the entire negative electrode active material layer. In addition, the value of [(thickness of negative electrode active material layer on one surface)/(thickness of current collector)] was 75 μm/10 μm=7.5.
《非水电解液的制作1》"Preparation of non-aqueous electrolyte 1"
在干燥的氩氛围下,在碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的混合物(体积比3∶3∶4)中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。另外,还含有0.3质量%的二氟磷酸锂盐(LiPO2F2)。Under a dry argon atmosphere, in a mixture of ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) (volume ratio 3:3:4), at a concentration of 1mol/L Dissolve well-dried lithium hexafluorophosphate (LiPF 6 ). In addition, 0.3% by mass of lithium difluorophosphate (LiPO 2 F 2 ) was contained.
《非水电解液的制作2》"The production of non-aqueous electrolyte 2"
在干燥的氩氛围下,在碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的混合物(体积比3∶3∶4)中,以1ol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。另外,还含有0.3质量%的甲磺酸三甲基甲硅烷基酯。Under a dry argon atmosphere, in a mixture of ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) (volume ratio 3:3:4), at a concentration of 1 mol/L Dissolve well-dried lithium hexafluorophosphate (LiPF 6 ). In addition, 0.3% by mass of trimethylsilyl methanesulfonate was contained.
《非水电解液的制作3》"The Production of Non-aqueous Electrolyte 3"
在干燥的氩氛围下,在碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的混合物(体积比3∶3∶4)中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。另外,还含有0.3质量%的六甲基环三硅氧烷。Under a dry argon atmosphere, in a mixture of ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) (volume ratio 3:3:4), at a concentration of 1mol/L Dissolve well-dried lithium hexafluorophosphate (LiPF 6 ). In addition, 0.3% by mass of hexamethylcyclotrisiloxane was contained.
《非水电解液的制作4》"The Production of Non-aqueous Electrolyte 4"
在干燥的氩氛围下,在碳酸亚乙酯(EC)、碳酸二甲酯(DMC)以及碳酸甲乙酯(EMC)的混合物(体积比3∶3∶4)中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。Under a dry argon atmosphere, in a mixture of ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) (volume ratio 3:3:4), at a concentration of 1mol/L Dissolve well-dried lithium hexafluorophosphate (LiPF 6 ).
《电池的制作1》"Battery Making 1"
将32片正极和33片负极交替配置,各电极之间夹入多孔聚乙烯片隔板(厚度为25μm),进行叠层。此时,使正极活性物质面与负极活性物质面相对,并且不会露出负极活性物质面之外。将该正极和负极各自的未涂布部分焊接,制作集电片,将电极组密封在电池罐(外部尺寸:120×110×10mm)中。然后,在装有电极组的电池罐中注入20mL非水电解液,使电极充分浸透,密封而制作方形电池。该电池的额定放电容量约为6安培小时(Ah),以10kHz交流法测定的直流电阻成分约为5毫欧(mΩ)。正极的电极面积的总和相对于电池的外壳表面积的总和之比为20.6。32 positive electrodes and 33 negative electrodes were arranged alternately, and a porous polyethylene sheet separator (thickness: 25 μm) was sandwiched between the electrodes for lamination. At this time, the surface of the positive electrode active material faces the surface of the negative electrode active material without protruding from the surface of the negative electrode active material. The uncoated parts of the positive and negative electrodes were welded to produce current collector sheets, and the electrode group was sealed in a battery can (outer dimensions: 120×110×10 mm). Then, 20 mL of non-aqueous electrolytic solution was poured into the battery can containing the electrode group, the electrodes were fully soaked, and sealed to produce a square battery. The rated discharge capacity of the battery is about 6 ampere hours (Ah), and the DC resistance component measured by the 10 kHz alternating current method is about 5 milliohms (mΩ). The ratio of the sum of the electrode areas of the positive electrode to the sum of the surface areas of the battery case was 20.6.
负极[9][10]实施例1Negative electrode [9] [10] Example 1
使用《负极的制作1》项的负极活性物质为碳质物质(A)而制作的负极、《正极的制作1》项中制作的正极以及《非水电解液的制作1》项中制作的非水电解液,通过《电池的制作1》项中的方法制作电池。下述《电池的评价》项中所述的方法对该电池进行测定。结果示于负极[9][10]表2。Use the negative electrode active material of " the making of negative electrode 1 " item to be the negative electrode made of carbonaceous material (A), the positive electrode made in the " making of positive electrode 1 " item and the nonaqueous electrolyte made in the item of " making 1 " The water electrolyte is used to make a battery by the method in "Battery Production 1". The battery was measured by the method described in the item "Evaluation of the battery" below. The results are shown in Table 2 of the negative electrode [9][10].
负极[9][10]实施例2Negative electrode [9] [10] Example 2
除了将负极[9][10]实施例1的非水电解液换成《非水电解液的制作2》项中制作的非水电解液以外,按照与负极[9][10]实施例1同样的方法制作电池,并同样地进行评价。结果示于负极[9][10]表2。Except that the non-aqueous electrolytic solution of the negative pole [9] [10] embodiment 1 is changed into the non-aqueous electrolytic solution made in the "making 2 of non-aqueous electrolytic solution" item, according to the negative pole [9] [10] embodiment 1 Batteries were produced in the same manner and evaluated in the same manner. The results are shown in Table 2 of the negative electrode [9][10].
负极[9][10]实施例3Negative electrode [9] [10] Example 3
除了将负极[9][10]实施例1的非水电解液换成《非水电解液的制作3》项中制作的非水电解液以外,按照与负极[9][10]实施例1同样的方法制作电池,并同样地进行评价。结果示于负极[9][10]表2。Except that the non-aqueous electrolytic solution of the negative pole [9] [10] embodiment 1 is changed into the non-aqueous electrolytic solution made in the "making 3 of non-aqueous electrolytic solution" item, according to the negative pole [9] [10] embodiment 1 Batteries were produced in the same manner and evaluated in the same manner. The results are shown in Table 2 of the negative electrode [9][10].
负极[9][10]比较例1Negative electrode [9] [10] Comparative Example 1
除了将负极[9][10]实施例1的非水电解液换成《非水电解液的制作4》项中制作的非水电解液以外,按照与负极[9][10]实施例1同样的方法制作电池,并同样地进行评价。结果示于负极[9][10]表2。Except that the non-aqueous electrolytic solution of the negative pole [9] [10] embodiment 1 is changed into the non-aqueous electrolytic solution made in the "making 4 of non-aqueous electrolytic solution" item, according to the negative pole [9] [10] embodiment 1 Batteries were produced in the same manner and evaluated in the same manner. The results are shown in Table 2 of the negative electrode [9][10].
负极[9][10]实施例4Negative electrode [9] [10] Example 4
除了负极[9][10]实施例1的《负极的制作1》项的负极活性物质使用碳质物质(B)以外,按照与负极[9][10]实施例1同样的方法制作电池,并同样地进行评价。结果示于负极[9][10]表2。Except that negative pole [9] [10] embodiment 1 " making of negative pole 1 " item negative pole active material uses carbonaceous material (B), according to the same method as negative pole [9] [10] embodiment 1, make battery, and evaluate in the same way. The results are shown in Table 2 of the negative electrode [9][10].
负极[9][10]实施例5Negative electrode [9] [10] Example 5
除了将负极[9][10]实施例4的非水电解液换成《非水电解液的制作2》项中制作的非水电解液以外,按照与负极[9][10]实施例4同样的方法制作电池,并同样地进行评价。结果示于负极[9][10]表2。Except that the non-aqueous electrolytic solution of the negative pole [9] [10] embodiment 4 is changed into the non-aqueous electrolytic solution made in the "making 2 of non-aqueous electrolytic solution" item, according to the negative pole [9] [10] embodiment 4 Batteries were produced in the same manner and evaluated in the same manner. The results are shown in Table 2 of the negative electrode [9][10].
负极[9][10]实施例6Negative electrode [9] [10] Example 6
除了将负极[9][10]实施例4的非水电解液换成《非水电解液的制作3》项中制作的非水电解液以外,按照与负极[9][10]实施例4同样的方法制作电池,并同样地进行评价。结果示于负极[9][10]表2。Except that the non-aqueous electrolytic solution of the negative pole [9] [10] embodiment 4 is changed into the non-aqueous electrolytic solution made in the "making 3 of non-aqueous electrolytic solution" item, according to the negative pole [9] [10] embodiment 4 Batteries were produced in the same manner and evaluated in the same manner. The results are shown in Table 2 of the negative electrode [9][10].
负极[9][10]比较例2Negative electrode [9] [10] Comparative example 2
除了将负极[9][10]实施例4的非水电解液换成《非水电解液的制作4》项中制作的非水电解液以外,按照与负极[9][10]实施例4同样的方法制作电池,并同样地进行评价。结果示于负极[9][10]表2。Except that the non-aqueous electrolytic solution of negative pole [9] [10] embodiment 4 is changed into the non-aqueous electrolytic solution made in " making 4 of non-aqueous electrolytic solution ", according to negative pole [9] [10] embodiment 4 Batteries were produced in the same manner and evaluated in the same manner. The results are shown in Table 2 of the negative electrode [9][10].
负极[9][10]实施例7Negative electrode [9] [10] Example 7
除了负极[9][10]实施例1的《负极的制作1》项的负极活性物质使用碳质物质(C)以外,按照与负极[9][10]实施例1同样的方法制作电池,并同样地进行评价。结果示于负极[9][10]表2。Except negative electrode [9] [10] embodiment 1 " making of negative electrode 1 " item negative electrode active material uses carbonaceous material (C), according to the same method as negative electrode [9] [10] embodiment 1, make battery, and evaluate in the same way. The results are shown in Table 2 of the negative electrode [9][10].
负极[9][10]实施例8Negative electrode [9] [10] Example 8
除了将负极[9][10]实施例7的非水电解液换成《非水电解液的制作2》项中制作的非水电解液以外,按照与负极[9][10]实施例7同样的方法制作电池,并同样地进行评价。结果示于负极[9][10]表2。Except that the non-aqueous electrolytic solution of the negative pole [9] [10] embodiment 7 is changed into the non-aqueous electrolytic solution made in the "making 2 of non-aqueous electrolytic solution" item, according to the negative pole [9] [10] embodiment 7 Batteries were produced in the same manner and evaluated in the same manner. The results are shown in Table 2 of the negative electrode [9][10].
负极[9][10]实施例9Negative electrode [9] [10] Example 9
除了将负极[9][10]实施例7的非水电解液换成《非水电解液的制作3》项中制作的非水电解液以外,按照与负极[9][10]实施例7同样的方法制作电池,并同样地进行评价。结果示于负极[9][10]表2。Except that the non-aqueous electrolytic solution of the negative pole [9] [10] embodiment 7 is changed into the non-aqueous electrolytic solution made in the "making 3 of non-aqueous electrolytic solution" item, according to the negative pole [9] [10] embodiment 7 Batteries were produced in the same manner and evaluated in the same manner. The results are shown in Table 2 of the negative electrode [9][10].
负极[9][10]比较例3Negative electrode [9] [10] Comparative example 3
除了将负极[9][10]实施例7的非水电解液换成《非水电解液的制作4》项中制作的非水电解液以外,按照与负极[9][10]实施例7同样的方法制作电池,并同样地进行评价。结果示于负极[9][10]表2。Except changing the non-aqueous electrolytic solution of negative pole [9] [10] embodiment 7 into the non-aqueous electrolytic solution made in " making 4 of non-aqueous electrolytic solution " item, according to negative pole [9] [10] embodiment 7 Batteries were produced in the same manner and evaluated in the same manner. The results are shown in Table 2 of the negative electrode [9][10].
负极[9][10]实施例10Negative electrode [9] [10] Example 10
除了负极[9][10]实施例1的《负极的制作1》项的负极活性物质使用碳质物质(D)以外,按照与负极[9][10]实施例1同样的方法制作电池,并同样地进行评价。结果示于负极[9][10]表2。Except negative electrode [9] [10] embodiment 1 " making of negative electrode 1 " item negative electrode active material uses carbonaceous material (D), according to the same method as negative electrode [9] [10] embodiment 1, make battery, and evaluate in the same way. The results are shown in Table 2 of the negative electrode [9][10].
负极[9][10]实施例11Negative electrode [9] [10] Example 11
除了将负极[9][10]实施例10的非水电解液换成《非水电解液的制作2》项中制作的非水电解液以外,按照与负极[9][10]实施例10同样的方法制作电池,并同样地进行评价。结果示于负极[9][10]表2。Except that the non-aqueous electrolytic solution of the negative pole [9] [10] embodiment 10 is changed into the non-aqueous electrolytic solution made in the "making of non-aqueous electrolytic solution 2" item, according to the negative pole [9] [10] embodiment 10 Batteries were produced in the same manner and evaluated in the same manner. The results are shown in Table 2 of the negative electrode [9][10].
负极[9][10]实施例12Negative electrode [9] [10] Example 12
除了将负极[9][10]实施例10的非水电解液换成《非水电解液的制作3》项中制作的非水电解液以外,按照与负极[9][10]实施例10同样的方法制作电池,并同样地进行评价。结果示于负极[9][10]表2。Except that the non-aqueous electrolytic solution of negative pole [9] [10] embodiment 10 is changed into the non-aqueous electrolytic solution made in " making 3 of non-aqueous electrolytic solution " item, according to negative pole [9] [10] embodiment 10 Batteries were produced in the same manner and evaluated in the same manner. The results are shown in Table 2 of the negative electrode [9][10].
负极[9][10]比较例4Negative electrode [9] [10] Comparative Example 4
除了将负极[9][10]实施例10的非水电解液换成《非水电解液的制作4》项中制作的非水电解液以外,按照与负极[9][10]实施例10同样的方法制作电池,并同样地进行评价。结果示于负极[9][10]表2。Except changing the non-aqueous electrolytic solution of negative pole [9] [10] embodiment 10 into the non-aqueous electrolytic solution made in " making 4 of non-aqueous electrolytic solution " item, according to negative pole [9] [10] embodiment 10 Batteries were produced in the same manner and evaluated in the same manner. The results are shown in Table 2 of the negative electrode [9][10].
负极[9][10]《电池的评价》Negative electrode[9][10] "Battery Evaluation"
(容量测定)(capacity measurement)
对未经充放电循环的电池,在25℃、4.2V~3.0V的电压范围下,以0.2C的电流值(将1小时放电额定容量的电流值作为1C,所述额定容量取决于1小时率(one-hour-rate)的放电容量,下同)进行5个循环的初期充放电。将此时的第5个循环的0.2C放电容量作为初期容量。接着进行下述所示的输出功率测定。For a battery that has not been charged and discharged, at 25°C and a voltage range of 4.2V to 3.0V, with a current value of 0.2C (the current value of the rated capacity of 1 hour discharge is taken as 1C, and the rated capacity depends on 1 hour Rate (one-hour-rate) discharge capacity, the same below) for 5 cycles of initial charge and discharge. The 0.2C discharge capacity at the fifth cycle at this time was taken as the initial capacity. Next, the output power measurement shown below was performed.
(输出功率测定)(Output power measurement)
在25℃的环境下,通过0.2C的恒定电流进行150分钟充电,分别以0.1C、0.3C、1.0C、3.0C、10.0C放电10秒钟,测定第10秒的电压。将电流-电压直线和下限电压(3V)围成的三角形的面积作为输出功率(W)。将循环试验前的输出功率作为“初期输出功率”。In an environment of 25°C, charge was performed for 150 minutes with a constant current of 0.2C, and discharge was performed at 0.1C, 0.3C, 1.0C, 3.0C, and 10.0C for 10 seconds, respectively, and the voltage at the 10th second was measured. The area of the triangle surrounded by the current-voltage line and the lower limit voltage (3V) is taken as the output power (W). Let the output power before the cycle test be the "initial output power".
(循环试验)(cycle test)
在被视为锂二次电池的实际使用上限温度的60℃的高温环境下进行循环试验。以2C的恒定电流恒定电压法充电至充电上限电压4.2V,然后以2C的恒定电流放电至放电终止电压3.0V,将该充放电循环作为1个循环,并反复进行该循环直至500循环。对于循环试验结束后的电池,在25℃环境下进行3个循环的充放电,将该第3个循环的0.2C放电容量作为循环后容量。由循环试验之前测定的初期容量和循环试验结束后测定的循环后容量按照下述计算式求出循环保持率。The cycle test was performed in a high-temperature environment of 60° C., which is regarded as the practical upper limit temperature of the lithium secondary battery. Charge to the charging upper limit voltage of 4.2V by the constant current and constant voltage method of 2C, and then discharge to the end-of-discharge voltage of 3.0V by the constant current of 2C. The charge-discharge cycle is regarded as one cycle, and this cycle is repeated until 500 cycles. For the battery after the cycle test, 3 cycles of charging and discharging were performed in an environment of 25° C., and the 0.2 C discharge capacity of the third cycle was taken as the capacity after cycle. The cycle retention rate was obtained from the initial capacity measured before the cycle test and the post-cycle capacity measured after the cycle test was completed according to the following calculation formula.
循环保持率(%)=100×循环后容量/初期容量Cycle retention (%) = 100 × capacity after cycle / initial capacity
负极[9][10]表2中的阻抗Rct和双层容量Cdl是对输出功率有贡献的参数之一,阻抗Rct的值越小,或者双层容量Cdl的值越大,则输出功率越有改善的倾向。另外,“阻抗Rct”和“双层容量Cdl”通过说明阻抗的部分所记载的方法求出。The impedance Rct and the double-layer capacity Cdl in Table 2 of the negative electrode [9][10] are one of the parameters that contribute to the output power. The smaller the value of the impedance Rct, or the larger the value of the double-layer capacity Cdl, the higher the output power. There is a tendency to improve. In addition, "impedance Rct" and "double layer capacitance Cdl" were obtained by the method described in the section describing impedance.
对于循环试验结束后的电池进行上述的(输出功率测定)项中所述的输出功率测定,并将得到的值作为“循环试验后输出功率”。上述负极[9][10]实施例和负极[9][10]比较例的锂二次电池的输出功率测定结果、容量测定结果、循环保持率、以及上述的对置阻抗测定中求出的反应电阻和双层容量的测定结果归纳示于负极[9][10]表2中。The output power measurement described in the above item (measurement of output power) was performed on the battery after the cycle test, and the obtained value was taken as "output power after cycle test". The output power measurement results, capacity measurement results, cycle retention ratio, and the above-mentioned counter impedance measurement of the lithium secondary batteries of the negative electrode [9] [10] examples and the negative electrode [9] [10] comparative examples. The measurement results of reaction resistance and double-layer capacity are summarized in Table 2 of the negative electrode [9][10].
负极[9][10]表2Negative pole [9] [10] Table 2
[表53][Table 53]
由负极[9][10]表2的结果可知,通过在非水电解液中含有二氟磷酸锂盐、甲磺酸三甲基甲硅烷基酯、六甲基环三硅氧烷等特定化合物,以及通过在负极中含有振实密度为0.1以上并且采用水银孔隙率计测定的0.01μm~1μm范围的微孔容积为0.01mL/g以上的负极活性物质,即使对于大型的电池,也可以实现高的循环后输出功率和高的循环保持率,从而可以提供具有高输出功率和长寿命两者的锂二次电池。From the results of the negative electrode [9][10] Table 2, it can be seen that by containing specific compounds such as lithium difluorophosphate, trimethylsilyl methanesulfonate, and hexamethylcyclotrisiloxane in the non-aqueous electrolyte , and by containing a negative electrode active material with a tap density of 0.1 or more and a pore volume of 0.01 mL/g or more in the range of 0.01 μm to 1 μm measured by a mercury porosimeter, even for large batteries, it can be realized High post-cycle output power and high cycle retention rate, so that a lithium secondary battery having both high output power and long life can be provided.
另外,通过在非水电解液中含有特定化合物,并且使负极的对置电池产生的反应电阻为500Ω以下,即使对于大型的电池,也可以实现高的循环后输出功率和高的循环保持率,从而兼备高输出功率和良好的寿命。In addition, by including a specific compound in the non-aqueous electrolyte solution and making the reaction resistance of the counter battery of the negative electrode less than 500Ω, even for a large battery, it is possible to achieve a high output after cycle and a high cycle retention rate, Thus both high output power and good life.
电解液[1]<二次电池的制作>Electrolyte solution [1] <Production of secondary battery>
[正极的制作][making of positive electrode]
在N-甲基吡咯烷酮溶剂中混合90质量%作为正极活性物质的钴酸锂(LiCoO2)、5质量%的作为导电材料的乙炔黑和5质量%作为粘合剂的聚偏氟乙烯(PVdF),制成浆料。将得到的浆料涂布在厚度15μm的铝箔两面,并干燥,用压制机压延成厚度80μm,再将其切成具有宽100mm、长100mm大小的活性物质层和宽30mm的未涂布部分的形状,作为正极。Mix 90% by mass of lithium cobaltate (LiCoO 2 ) as a positive electrode active material, 5% by mass of acetylene black as a conductive material, and 5% by mass of polyvinylidene fluoride (PVdF ) to make slurry. The obtained slurry was coated on both sides of an aluminum foil with a thickness of 15 μm, dried, rolled to a thickness of 80 μm with a press, and cut into an active material layer having a width of 100 mm and a length of 100 mm and an uncoated portion of a width of 30 mm. shape, as the positive electrode.
[负极的制作][Production of Negative Electrode]
在98重量份人造石墨粉末KS-44(timcal公司制造,商品名)中添加100重量份作为增稠剂的羧甲基纤维素钠的水性分散液(羧甲基纤维素钠的浓度为1质量%)、2重量份作为粘合剂的苯乙烯-丁二烯橡胶的水性分散液(苯乙烯-丁二烯橡胶的浓度为50质量%),用分散器进行混合,制成浆料。将得到的浆料涂布在厚度10μm的铜箔的两面,并干燥,用压制机压延成厚度为75μm,再将其切成具有宽104mm、长104mm大小的活性物质层和宽30mm的未涂布部分的形状,作为负极。In 98 parts by weight of artificial graphite powder KS-44 (manufactured by timcal company, trade name), add 100 parts by weight of an aqueous dispersion of sodium carboxymethyl cellulose as a thickener (the concentration of sodium carboxymethyl cellulose is 1 mass %), 2 parts by weight of an aqueous dispersion of styrene-butadiene rubber as a binder (the concentration of styrene-butadiene rubber is 50% by mass), and mixed with a disperser to prepare a slurry. The obtained slurry is coated on both sides of a copper foil with a thickness of 10 μm, dried, rolled into a thickness of 75 μm with a press, and then cut into an active material layer with a width of 104 mm and a length of 104 mm and an uncoated layer with a width of 30 mm. The shape of the cloth part is used as the negative electrode.
[电池的组装][assembling the battery]
将32片正极和33片负极交替配置,各电极之间夹入多孔聚乙烯片隔板(厚度为25μm),进行叠层。此时,使正极活性物质面与负极活性物质面相对,并且不会露出负极活性物质面之外。将该正极和负极各自的未涂布部分焊接,制作集电片,将电极组密封在电池罐(外部尺寸:120×110×10mm)中。然后,在装有电极组的电池罐中注入20mL非水电解液,使电极充分浸透,密封而制作方形电池。该电池的额定放电容量约为6安培小时(Ah),以10kHz交流法测定的直流电阻成分约为5毫欧。32 positive electrodes and 33 negative electrodes were arranged alternately, and a porous polyethylene sheet separator (thickness: 25 μm) was sandwiched between the electrodes for lamination. At this time, the surface of the positive electrode active material faces the surface of the negative electrode active material without protruding from the surface of the negative electrode active material. The respective uncoated parts of the positive and negative electrodes were welded to produce current collector sheets, and the electrode group was sealed in a battery can (outer dimensions: 120×110×10 mm). Then, 20 mL of non-aqueous electrolytic solution was poured into the battery can containing the electrode group, the electrodes were fully soaked, and sealed to produce a square battery. The battery has a rated discharge capacity of about 6 ampere hours (Ah) and a DC resistance component of about 5 milliohms as measured by the 10 kHz AC method.
[电池的评价][evaluation of the battery]
(初期容量的测定方法)(Measuring method of initial capacity)
对未经充放电循环的新电池,在25℃、4.1V~3.0V的电压范围进行5个循环的初期充放电(电压范围4.1V~3.0V)。将此时的第5个循环的0.2C(将1小时放电额定容量的电流值作为1C,所述额定容量取决于1小时率(one-hour-rate)的放电容量,下同)放电容量作为初期容量。For a new battery that has not undergone charge-discharge cycles, 5 cycles of initial charge-discharge (voltage range 4.1V-3.0V) were performed at 25°C in a voltage range of 4.1V-3.0V. The 0.2C discharge capacity of the 5th cycle at this time (the current value of the 1-hour discharge rated capacity is taken as 1C, and the rated capacity depends on the discharge capacity of the 1-hour-rate (one-hour-rate), the same below) discharge capacity as initial capacity.
(低温输出功率的测定方法)(Measurement method of low temperature output power)
在25℃的环境下,通过0.2C的恒定电流进行150分钟充电,在-30℃环境下分别以0.1C、0.3C、1.0C、3.0C、5.0C放电10秒钟,测定第10秒的电压。将电流-电压直线和下限电压(3V)围成的三角形的面积作为输出功率(W)。In an environment of 25°C, charge with a constant current of 0.2C for 150 minutes, and discharge at 0.1C, 0.3C, 1.0C, 3.0C, and 5.0C for 10 seconds in an environment of -30°C, and measure the 10th second Voltage. The area of the triangle surrounded by the current-voltage line and the lower limit voltage (3V) is taken as the output power (W).
电解液[1]实施例1Electrolyte [1] Example 1
在干燥氩氛围下,向碳酸亚乙酯(EC)、碳酸甲乙酯(EMC)的混合物(体积比15∶85)中以1mol/L加入六氟磷酸锂(LiPF6)并进行溶解,在该混合溶液中混合0.3质量%的量的六甲基环三硅氧烷,制备非水电解液。使用该非水电解液按照上述方法制作电池,并测定低温输出功率。结果如电解液[1]表1所示。Under a dry argon atmosphere, add lithium hexafluorophosphate (LiPF 6 ) at 1 mol/L to a mixture of ethylene carbonate (EC) and ethylmethyl carbonate (EMC) (volume ratio 15:85) and dissolve it. Hexamethylcyclotrisiloxane was mixed in an amount of 0.3% by mass to prepare a non-aqueous electrolytic solution. Using this non-aqueous electrolytic solution, a battery was fabricated according to the above-mentioned method, and the low-temperature output was measured. The results are shown in Table 1 of the electrolyte [1].
电解液[1]实施例2Electrolyte [1] Example 2
使用电解液[1]实施例1中以EC和EMC的体积比20∶80制备的非水电解液来制作电池,并测定低温输出功率。结果如电解液[1]表1所示。Use the non-aqueous electrolyte solution prepared in Example 1 of the electrolyte solution [1] with a volume ratio of EC and EMC of 20:80 to make a battery, and measure the low-temperature output power. The results are shown in Table 1 of the electrolyte [1].
电解液[1]实施例3Electrolyte [1] Example 3
使用电解液[1]实施例1中的非水溶剂变更为EC、碳酸二甲酯(DMC)、EMC的混合物(体积比15∶40∶45)而制备的非水电解液来制作电池,并测定低温输出功率。结果如电解液[1]表1所示。Use the non-aqueous electrolyte solution that the non-aqueous solvent in the embodiment 1 of electrolytic solution [1] is changed into the mixture (volume ratio 15:40:45) of EC, dimethyl carbonate (DMC), EMC and prepare battery, and Determination of low temperature output power. The results are shown in Table 1 of the electrolyte [1].
电解液[1]实施例4Electrolyte [1] Example 4
使用混合有苯基二甲基氟硅烷来替代电解液[1]实施例1中的六甲基环三硅氧烷而制备的非水电解液,制作电池,并测定低温输出功率。结果如电解液[1]表1所示。Using a non-aqueous electrolytic solution prepared by mixing phenyldimethylfluorosilane instead of hexamethylcyclotrisiloxane in the electrolytic solution [1] Example 1, a battery was produced, and the low-temperature output was measured. The results are shown in Table 1 of the electrolyte [1].
电解液[1]实施例5Electrolyte [1] Example 5
使用混合有六甲基二硅氧烷来替代电解液[1]实施例1中的六甲基环三硅氧烷而制备的非水电解液,制作电池,并测定低温输出功率。结果如电解液[1]表1所示。Using the non-aqueous electrolytic solution prepared by mixing hexamethyldisiloxane instead of the hexamethylcyclotrisiloxane in the electrolytic solution [1] Example 1, a battery was produced, and the low-temperature output was measured. The results are shown in Table 1 of the electrolyte [1].
电解液[1]实施例6Electrolyte [1] Example 6
使用混合有甲磺酸三甲基甲硅烷基酯来替代电解液[1]实施例1中的六甲基环三硅氧烷而制备的非水电解液,制作电池,并测定低温输出功率。结果如电解液[1]表1所示。Using a non-aqueous electrolytic solution prepared by mixing trimethylsilyl methanesulfonate instead of hexamethylcyclotrisiloxane in the electrolytic solution [1] Example 1, a battery was produced, and the low-temperature output was measured. The results are shown in Table 1 of the electrolyte [1].
电解液[1]实施例7Electrolyte [1] Example 7
使用混合有氟磺酸甲酯来替代电解液[1]实施例1中的六甲基环三硅氧烷而制备的非水电解液,制作电池,并测定低温输出功率。结果如电解液[1]表1所示。Using the non-aqueous electrolytic solution prepared by mixing methyl fluorosulfonate instead of hexamethylcyclotrisiloxane in the electrolytic solution [1] Example 1, a battery was produced, and the low-temperature output was measured. The results are shown in Table 1 of the electrolyte [1].
电解液[1]实施例8Electrolyte [1] Example 8
使用混合有硝酸锂来替代电解液[1]实施例1中的六甲基环三硅氧烷、并保存3天而得到的非水电解液来制作电池,并测定低温输出功率。结果如电解液[1]表1所示。A battery was produced using a non-aqueous electrolytic solution obtained by mixing lithium nitrate instead of hexamethylcyclotrisiloxane in the electrolytic solution [1] Example 1 and storing for 3 days, and the low-temperature output was measured. The results are shown in Table 1 of the electrolyte [1].
电解液[1]实施例9Electrolyte [1] Example 9
使用混合有根据Inorganic Nuclear Chemistry Letters(1969),5(7)的第581页~第582页中记载的方法制备的二氟磷酸锂来替代电解液[1]实施例1中的六甲基环三硅氧烷而制备的非水电解液作为非水电解液,制作电池,并测定低温输出功率。结果如电解液[1]表1所示。Use lithium difluorophosphate prepared according to the method described on pages 581 to 582 of Inorganic Nuclear Chemistry Letters (1969), 5 (7) to replace the hexamethyl ring in the electrolyte [1] Example 1 The non-aqueous electrolyte prepared by using trisiloxane was used as the non-aqueous electrolyte to make a battery and measure the low-temperature output power. The results are shown in Table 1 of the electrolyte [1].
电解液[1]实施例10Electrolyte [1] Example 10
电解液[1]实施例9中,使用将二氟磷酸锂的混合量变更为相对于非水溶剂和六氟磷酸锂的混合溶液为0.08质量%的量而得到的非水电解液来制作电池,并测定低温输出功率。结果如电解液[1]表1所示。Electrolyte solution [1] In Example 9, a battery was fabricated using a non-aqueous electrolyte solution obtained by changing the mixing amount of lithium difluorophosphate to an amount of 0.08% by mass relative to a mixed solution of a nonaqueous solvent and lithium hexafluorophosphate, and measured Low temperature output power. The results are shown in Table 1 of the electrolyte [1].
电解液[1]实施例11Electrolyte [1] Example 11
使用混合有乙酸锂来替代电解液[1]实施例1中的六甲基环三硅氧烷、并保存3天而得到的非水电解液,制作电池,并测定低温输出功率。结果如电解液[1]表1所示。Using the non-aqueous electrolytic solution obtained by mixing lithium acetate instead of hexamethylcyclotrisiloxane in the electrolytic solution [1] Example 1 and storing it for 3 days, a battery was produced, and the low-temperature output was measured. The results are shown in Table 1 of the electrolyte [1].
电解液[1]实施例12Electrolyte [1] Example 12
<二次电池的制作-2><Production of secondary battery-2>
[正极的制作][making of positive electrode]
在N-甲基吡咯烷酮溶剂中混合90质量%作为正极活性物质的钴酸锂(LiCoO2)、5质量%的作为导电材料的乙炔黑和5质量%作为粘合剂的聚偏氟乙烯(PVdF),制成浆料。将得到的浆料涂布在厚度20μm的铝箔两面,并干燥,用压制机压延成厚度80μm,再将其切成宽52mm、长830mm大小,作为正极。其中,表面背面都在长度方向上设置50mm的未涂布部分,活性物质层的长度为780mm。Mix 90% by mass of lithium cobaltate (LiCoO 2 ) as a positive electrode active material, 5% by mass of acetylene black as a conductive material, and 5% by mass of polyvinylidene fluoride (PVdF ) to make slurry. The obtained slurry was coated on both sides of an aluminum foil with a thickness of 20 μm, dried, and rolled to a thickness of 80 μm by a press machine, and then cut into a size of 52 mm in width and 830 mm in length as a positive electrode. Among them, an uncoated portion of 50 mm was provided in the longitudinal direction on both the front and back sides, and the length of the active material layer was 780 mm.
[负极的制作][Production of Negative Electrode]
在98重量份人造石墨粉末KS-44(timcal公司制造,商品名)中添加100重量份作为增稠剂的羧甲基纤维素钠的水性分散液(羧甲基纤维素钠的浓度为1质量%)、2重量份作为粘合剂的苯乙烯-丁二烯橡胶的水性分散液(苯乙烯-丁二烯橡胶的浓度为50质量%),用分散器进行混合,制成浆料。将得到的浆料均匀涂布在作为负极集电体的厚度18μm的铜箔的两面,干燥后,用压制机压延成厚度为85μm,再将其切成宽56mm、长850mm的大小,作为负极。其中,表面背面都在长度方向上设置30mm的未涂布部分。In 98 parts by weight of artificial graphite powder KS-44 (manufactured by timcal company, trade name), add 100 parts by weight of an aqueous dispersion of sodium carboxymethyl cellulose as a thickener (the concentration of sodium carboxymethyl cellulose is 1 mass %), 2 parts by weight of an aqueous dispersion of styrene-butadiene rubber as a binder (the concentration of styrene-butadiene rubber is 50% by mass), and mixed with a disperser to prepare a slurry. The obtained slurry was uniformly coated on both sides of a copper foil with a thickness of 18 μm as the negative electrode current collector, and after drying, it was rolled to a thickness of 85 μm by a press, and then cut into a size of 56 mm wide and 850 mm long, as the negative electrode . Among them, an uncoated portion of 30 mm was provided in the longitudinal direction on both the front and back sides.
[电解液的制作][Production of electrolyte solution]
制作与电解液[1]实施例1同样的非水电解液。The same non-aqueous electrolytic solution as in the electrolytic solution [1] Example 1 was prepared.
[电池的组装][assembling the battery]
将正极和负极与聚乙烯制的隔板一起重叠卷绕以使正极与负极不直接接触,制成电极体。使正极和负极的端子露出外部地装在电池罐中。The positive electrode and the negative electrode were stacked and wound together with a polyethylene separator so that the positive electrode and the negative electrode were not in direct contact, and an electrode body was produced. The positive and negative terminals were exposed in the battery can.
然后,向其中注入5mL后述的电解液后,进行铆接成型,制造18650型圆筒电池。该电池的额定放电容量约为0.7安培小时(Ah),用10kHz交流法测定的直流电阻约为35毫欧(mΩ)。此外,上述正极的电极面积的总和相对于该二次电池的外壳的表面积的面积比为19.4倍。对于上述电池,与电解液[1]实施例1同样地测定低温输出功率。结果如电解液[1]表1所示。Then, 5 mL of an electrolytic solution described later was injected thereinto, followed by riveting molding to manufacture a 18650-type cylindrical battery. The battery has a rated discharge capacity of about 0.7 ampere hours (Ah) and a DC resistance of about 35 milliohms (mΩ) measured by the 10 kHz AC method. In addition, the area ratio of the sum of the electrode areas of the positive electrode to the surface area of the case of the secondary battery was 19.4 times. For the above battery, the low-temperature output was measured in the same manner as in Example 1 of the electrolytic solution [1]. The results are shown in Table 1 of the electrolyte [1].
电解液[1]比较例1Electrolyte [1] Comparative Example 1
使用电解液[1]实施例1中不混合六甲基环三硅氧烷而制备的非水电解液来制作电池,并测定低温输出功率。结果如电解液[1]表1所示。A battery was fabricated using the non-aqueous electrolyte solution prepared without mixing hexamethylcyclotrisiloxane in Example 1 of the electrolyte solution [1], and the low-temperature output power was measured. The results are shown in Table 1 of the electrolyte [1].
电解液[1]比较例2Electrolyte [1] Comparative Example 2
使用电解液[1]实施例2中不混合六甲基环三硅氧烷而制备的非水电解液来制作电池,并测定低温输出功率。结果如电解液[1]表1所示。A battery was produced using the non-aqueous electrolyte solution prepared without mixing hexamethylcyclotrisiloxane in Example 2 of the electrolyte solution [1], and the low-temperature output power was measured. The results are shown in Table 1 of the electrolyte [1].
电解液[1]比较例3Electrolyte [1] Comparative Example 3
使用电解液[1]实施例3中不混合六甲基环三硅氧烷而制备的非水电解液来制作电池,并测定低温输出功率。结果如电解液[1]表1所示。A battery was produced using the non-aqueous electrolyte solution prepared without mixing hexamethylcyclotrisiloxane in Example 3 of the electrolyte solution [1], and the low-temperature output power was measured. The results are shown in Table 1 of the electrolyte [1].
电解液[1]比较例4Electrolyte [1] Comparative Example 4
在干燥氩氛围下,向碳酸亚乙酯和碳酸甲乙酯的混合物(体积比4∶6)中以1mol/L加入六氟磷酸锂(LiPF6)并进行溶解,再以相对于该混合溶液为0.3质量%的量混合六甲基环三硅氧烷来制备非水电解液。Under a dry argon atmosphere, add lithium hexafluorophosphate (LiPF 6 ) at 1 mol/L to a mixture of ethylene carbonate and ethyl methyl carbonate (volume ratio 4:6) and dissolve it, and then add 0.3 mass % of the amount of mixed hexamethylcyclotrisiloxane to prepare a non-aqueous electrolyte.
使用该非水电解液制作电池,并测定低温输出功率。结果如电解液[1]表1所示。A battery was produced using this non-aqueous electrolyte solution, and the low-temperature output was measured. The results are shown in Table 1 of the electrolyte [1].
电解液[1]比较例5Electrolyte [1] Comparative Example 5
使用混合有苯基二甲基氟硅烷来替代电解液[1]比较例4中的六甲基环三硅氧烷而制备的非水电解液,制作电池,并测定低温输出功率。结果如电解液[1]表1所示。Using a non-aqueous electrolytic solution prepared by mixing phenyldimethylfluorosilane instead of hexamethylcyclotrisiloxane in the electrolytic solution [1] Comparative Example 4, a battery was produced, and the low-temperature output was measured. The results are shown in Table 1 of the electrolyte [1].
电解液[1]比较例6Electrolyte [1] Comparative Example 6
使用混合有六甲基二硅氧烷来替代电解液[1]比较例4中的六甲基环三硅氧烷而制备的非水电解液,制作电池,并测定低温输出功率。结果如电解液[1]表1所示。Using a non-aqueous electrolytic solution prepared by mixing hexamethyldisiloxane instead of hexamethylcyclotrisiloxane in the electrolytic solution [1] Comparative Example 4, a battery was produced, and the low-temperature output was measured. The results are shown in Table 1 of the electrolyte [1].
电解液[1]比较例7Electrolyte [1] Comparative Example 7
使用混合有甲磺酸三甲基甲硅烷基酯来替代电解液[1]比较例4中的六甲基环三硅氧烷而制备的非水电解液,制作电池,并测定低温输出功率。结果如电解液[1]表1所示。Using a non-aqueous electrolytic solution prepared by mixing trimethylsilyl methanesulfonate instead of hexamethylcyclotrisiloxane in the electrolytic solution [1] Comparative Example 4, a battery was produced, and the low-temperature output was measured. The results are shown in Table 1 of the electrolyte [1].
电解液[1]比较例8Electrolyte [1] Comparative Example 8
使用混合有氟磺酸甲酯来替代电解液[1]比较例4中的六甲基环三硅氧烷而制备的非水电解液,制作电池,并测定低温输出功率。结果如电解液[1]表1所示。Using a nonaqueous electrolytic solution prepared by mixing methyl fluorosulfonate instead of hexamethylcyclotrisiloxane in the electrolytic solution [1] Comparative Example 4, a battery was produced, and the low-temperature output was measured. The results are shown in Table 1 of the electrolyte [1].
电解液[1]比较例9Electrolyte [1] Comparative Example 9
使用混合有硝酸锂来替代电解液[1]比较例4中的六甲基环三硅氧烷、并保存3天而得到的非水电解液,制作电池,并测定低温输出功率。结果如电解液[1]表1所示。A battery was manufactured using a non-aqueous electrolytic solution obtained by mixing lithium nitrate instead of hexamethylcyclotrisiloxane in the electrolytic solution [1] Comparative Example 4 and storing for 3 days, and the low-temperature output was measured. The results are shown in Table 1 of the electrolyte [1].
电解液[1]比较例10Electrolyte [1] Comparative Example 10
使用混合有二氟磷酸锂来替代电解液[1]比较例4中的六甲基环三硅氧烷而制备的非水电解液,制作电池,并测定低温输出功率。结果如电解液[1]表1所示。Using a non-aqueous electrolytic solution prepared by mixing lithium difluorophosphate instead of hexamethylcyclotrisiloxane in the electrolytic solution [1] Comparative Example 4, a battery was produced, and the low-temperature output was measured. The results are shown in Table 1 of the electrolyte [1].
电解液[1]比较例11Electrolyte [1] Comparative Example 11
使用混合有乙酸锂来替代电解液[1]比较例4中的六甲基环三硅氧烷、并保存3天而得到的非水电解液,制作电池,并测定低温输出功率。结果如电解液[1]表1所示。A battery was produced using a non-aqueous electrolytic solution obtained by mixing lithium acetate instead of hexamethylcyclotrisiloxane in the electrolytic solution [1] Comparative Example 4, and storing for 3 days, and the low-temperature output was measured. The results are shown in Table 1 of the electrolyte [1].
电解液[1]比较例12Electrolyte [1] Comparative Example 12
使用电解液[1]比较例4中不混合六甲基环三硅氧烷而制备的非水电解液,制作电池,并测定低温输出功率。结果如电解液[1]表1所示。Using the non-aqueous electrolytic solution prepared without mixing hexamethylcyclotrisiloxane in the electrolytic solution [1] Comparative Example 4, a battery was produced, and the low-temperature output was measured. The results are shown in Table 1 of the electrolyte [1].
电解液[1]比较例13Electrolyte [1] Comparative Example 13
电解液[1]实施例12中,使用与电解液[1]比较例1同样的非水电解液,制作电池,并测定低温输出功率。结果如电解液[1]表1所示。Electrolyte solution [1] In Example 12, a battery was fabricated using the same non-aqueous electrolyte solution as in Electrolyte Solution [1] Comparative Example 1, and the low-temperature output was measured. The results are shown in Table 1 of the electrolyte [1].
电解液[1]表1Electrolyte[1] Table 1
[表54][Table 54]
由电解液[1]表1可知,在非水电解液中含有特定量的EC和特定化合物的电解液[1]实施例1~11的锂二次电池,不仅与含有过量的EC、且不含有特定化合物的电解液[1]比较例12的锂二次电池相比,而且即使与虽然EC量在特定范围内但是不含有特定化合物的电解液[1]比较例1~3、以及虽然含有特定化合物但是EC过量的电解液[1]比较例4~11的锂二次电池相比,低温输出功率特性得到改善。此外,在非水电解液中含有特定量的EC和特定化合物的电解液[1]实施例12的锂二次电池,与虽然EC量在特定范围内但是不含有特定化合物的电解液[1]比较例13的锂二次电池相比,低温输出功率特性得到改善。It can be seen from Table 1 of the electrolyte [1] that the lithium secondary batteries of the electrolyte [1] Examples 1 to 11 containing a specific amount of EC and a specific compound in the non-aqueous electrolyte are not only compatible with the lithium secondary batteries containing an excessive amount of EC, but also not Compared with the lithium secondary battery of the electrolyte solution [1] Comparative Example 12 containing the specific compound, and even with the electrolyte solution [1] Comparative Examples 1 to 3 that did not contain the specific compound although the EC amount was within a specific range, and although it contained The electrolytic solution [1] in which the specific compound was excessive in EC had improved low-temperature output characteristics compared with the lithium secondary batteries of Comparative Examples 4 to 11. In addition, the lithium secondary battery of Example 12 in the electrolyte solution [1] containing a specific amount of EC and a specific compound in the non-aqueous electrolyte solution, and the electrolyte solution [1] which does not contain the specific compound although the amount of EC is within a specific range Compared with the lithium secondary battery of Comparative Example 13, the low-temperature output characteristics were improved.
此外,该效果不是两者的单纯叠加,通过满足两方面条件,效果明显增强。电解液[1]表1中记载了使用含有特定化合物(除此之外为同一组成)的非水电解液时相对于使用不含有特定化合物的非水电解液时的低温输出功率的上升率,与EC量过量的电解液[1]比较例4~11相比,电解液[1]实施例1~12中,该值大,本发明的效果大。In addition, the effect is not a simple superposition of the two, but the effect is significantly enhanced by satisfying two conditions. Electrolyte solution [1] Table 1 shows the rate of increase in low-temperature output when using a non-aqueous electrolyte solution containing a specific compound (except for the same composition) compared to when using a non-aqueous electrolyte solution that does not contain a specific compound. In the electrolytic solution [1] Examples 1 to 12, this value was larger than that of the electrolytic solution [1] Comparative Examples 4 to 11 in which the amount of EC was excessive, and the effect of the present invention was large.
此外,电解液[1]实施例12相对于电解液[1]比较例13的输出功率上升率为20.8%,而虽然使用同样的材料但是电池结构不同的电解液[1]实施例1相对于电解液[1]比较例1的输出功率上升率为26.4%,可知电池结构对本发明的非水电解液的效果有大的影响。即,对于高容量的电池、直流电阻小的电池,本发明的效果特别大。In addition, the increase rate of the output power of the electrolyte [1] Example 12 relative to the electrolyte [1] Comparative Example 13 was 20.8%, while the electrolyte [1] Example 1 with the same material but a different battery structure was compared to the The output increase rate of the electrolyte solution [1] Comparative Example 1 was 26.4%. It can be seen that the battery structure has a great influence on the effect of the non-aqueous electrolyte solution of the present invention. That is, the effect of the present invention is particularly large for a high-capacity battery or a battery with a small DC resistance.
进一步地,虽然表中未记载,但是使用EC含量小于1容量%的电解液时,与电解液[1]实施例1相比,初期容量稍微降低,常温下的输出功率特性或循环特性变差。Furthermore, although it is not described in the table, when an electrolyte solution with an EC content of less than 1% by volume is used, compared with the electrolyte solution [1] Example 1, the initial capacity is slightly lowered, and the output power characteristics or cycle characteristics at room temperature are deteriorated. .
如上所述,通过使用本发明的非水电解液,即具有下述特征的二次电池用非水电解液,可以发挥非常大的低温输出功率特性,所述二次电池用非水电解液的特征为:其为至少含有碳酸亚乙酯的混合溶剂,碳酸亚乙酯对于非水溶剂总量的比例为1容量%~25容量%,该非水电解液还含有选自下述物质中的至少一种化合物,并且其在全部非水电解液中的含量为10ppm以上,所述物质包括:通式(1)表示的环状硅氧烷化合物、通式(2)表示的氟硅烷化合物、通式(3)表示的化合物、分子内具有S-F键的化合物、硝酸盐、亚硝酸盐、单氟磷酸盐、二氟磷酸盐、乙酸盐和丙酸盐。As mentioned above, by using the non-aqueous electrolytic solution of the present invention, that is, a non-aqueous electrolytic solution for secondary batteries having the following characteristics, very large low-temperature output power characteristics can be brought into play. It is characterized by: it is a mixed solvent containing at least ethylene carbonate, the ratio of ethylene carbonate to the total amount of non-aqueous solvents is 1% to 25% by volume, and the non-aqueous electrolytic solution also contains At least one compound, and its content in all non-aqueous electrolytic solutions is 10ppm or more, said substances include: cyclic siloxane compounds represented by general formula (1), fluorosilane compounds represented by general formula (2), Compounds represented by the general formula (3), compounds having an S-F bond in the molecule, nitrates, nitrites, monofluorophosphates, difluorophosphates, acetates and propionates.
另外,与电解液[1]实施例12的电池结构相比,在电解液[1]实施例1的电池结构,即高容量的电池、直流电阻小的电池中,可以更显著地发挥该效果。In addition, compared with the battery structure of the electrolyte solution [1] Example 12, the battery structure of the electrolyte solution [1] Example 1, that is, a battery with a high capacity and a battery with a small DC resistance, can exert this effect more significantly .
电解液[2]<二次电池的制作-1>Electrolyte solution [2] <Production of secondary battery-1>
[正极的制作][making of positive electrode]
在N-甲基吡咯烷酮溶剂中混合90质量%作为正极活性物质的钴酸锂(LiCoO2)、5质量%的作为导电材料的乙炔黑和5质量%作为粘合剂的聚偏氟乙烯(PVdF),制成浆料。将得到的浆料涂布在厚度20μm的铝箔两面,并干燥,用压制机压延成厚度80μm,再将其切成宽52mm、长830mm大小,作为正极。其中,表面背面都在长度方向上设置50mm的未涂布部分,活性物质层的长度为780mm。Mix 90% by mass of lithium cobaltate (LiCoO 2 ) as a positive electrode active material, 5% by mass of acetylene black as a conductive material, and 5% by mass of polyvinylidene fluoride (PVdF ) to make slurry. The obtained slurry was coated on both sides of an aluminum foil with a thickness of 20 μm, dried, and rolled to a thickness of 80 μm by a press machine, and then cut into a size of 52 mm in width and 830 mm in length as a positive electrode. Among them, an uncoated portion of 50 mm was provided in the longitudinal direction on both the front and back sides, and the length of the active material layer was 780 mm.
[负极的制作][Production of Negative Electrode]
在98重量份人造石墨粉末KS-44(timcal公司制造,商品名)中添加100重量份作为增稠剂的羧甲基纤维素钠的水性分散液(羧甲基纤维素钠的浓度为1质量%)、2重量份作为粘合剂的苯乙烯-丁二烯橡胶的水性分散液(苯乙烯-丁二烯橡胶的浓度为50质量%),用分散器进行混合,制成浆料。将得到的浆料均匀涂布在作为负极集电体的厚度18μm的铜箔的两面,干燥后,用压制机压延成厚度为85μm,再将其切成宽56mm、长850mm的大小,作为负极。其中,表面背面都在长度方向上设置30mm的未涂布部分。In 98 parts by weight of artificial graphite powder KS-44 (manufactured by timcal company, trade name), add 100 parts by weight of an aqueous dispersion of sodium carboxymethyl cellulose as a thickener (the concentration of sodium carboxymethyl cellulose is 1 mass %), 2 parts by weight of an aqueous dispersion of styrene-butadiene rubber as a binder (the concentration of styrene-butadiene rubber is 50% by mass), and mixed with a disperser to prepare a slurry. The obtained slurry was uniformly coated on both sides of a copper foil with a thickness of 18 μm as the negative electrode current collector, and after drying, it was rolled to a thickness of 85 μm by a press, and then cut into a size of 56 mm wide and 850 mm long, as the negative electrode . Among them, an uncoated portion of 30 mm was provided in the longitudinal direction on both the front and back sides.
[电池的组装][assembling the battery]
将正极和负极与聚乙烯制的隔板一起重叠卷绕以使正极与负极不直接接触,制成电极体。使正极和负极的端子露出外部地装在电池罐中。然后,向其中注入5mL后述的电解液后,进行铆接成型,制造18650型圆筒电池。二次电池的1个电池外壳收纳的电池要素所具有的电容,即二次电池的额定放电容量约为0.7安培小时(Ah),用10kHz交流法测定的直流电阻成分约为35毫欧(mΩ)。The positive electrode and the negative electrode were stacked and wound together with a polyethylene separator so that the positive electrode and the negative electrode were not in direct contact, and an electrode body was produced. The positive and negative terminals were exposed in the battery can. Then, 5 mL of an electrolytic solution described later was injected thereinto, followed by riveting molding to manufacture a 18650-type cylindrical battery. The capacitance of the battery element contained in one battery case of the secondary battery, that is, the rated discharge capacity of the secondary battery is about 0.7 ampere hours (Ah), and the DC resistance component measured by the 10kHz alternating current method is about 35 milliohms (mΩ ).
[电池的评价][evaluation of the battery]
(初期充放电)(initial charge and discharge)
对制造的电池,在25℃下用0.2C的恒定电流恒定电压充电法充电至4.2V后,以0.2C的恒定电流放电至3.0V。将其进行5个循环使电池稳定。将此时的第5个循环的放电容量作为初期容量。而且,将1小时放电额定容量的电流值作为1C,所述额定容量取决于1小时率(one-hour-rate)的放电容量。The produced battery was charged to 4.2V at 25°C by a constant current constant voltage charging method of 0.2C, and then discharged to 3.0V by a constant current of 0.2C. This was performed for 5 cycles to stabilize the battery. The discharge capacity at the 5th cycle at this time was taken as the initial capacity. Also, let the current value of the 1-hour discharge rated capacity which depends on the 1-hour-rate discharge capacity be 1C.
(循环试验)(cycle test)
对实施了初期充放电的电池,在60℃下进行以1C的恒定电流恒定电压充电至4.2V后,再以1C的恒定电流放电至3.0V的充放电500个循环。将此时的第500个循环放电容量相对于第1个循环放电容量的比例作为循环保持率。The battery subjected to initial charge and discharge was charged to 4.2V at a constant current and constant voltage of 1C at 60°C, and then discharged to 3.0V at a constant current of 1C for 500 cycles. The ratio of the discharge capacity at the 500th cycle to the discharge capacity at the first cycle at this time was defined as the cycle retention ratio.
(低温试验)(low temperature test)
对实施了初期充放电的电池,在25℃下用0.2C的恒定电流恒定电压充电法充电至4.2V后,在-30℃下实施0.2C的恒定电流放电。将此时的放电容量作为初期低温容量,将初期低温容量相对于初期容量的比例作为初期低温放电率。The battery subjected to the initial charge and discharge was charged to 4.2V by a 0.2C constant current constant voltage charging method at 25°C, and then a 0.2C constant current discharge was performed at -30°C. The discharge capacity at this time was taken as the initial low-temperature capacity, and the ratio of the initial low-temperature capacity to the initial capacity was taken as the initial low-temperature discharge rate.
此外,对循环试验后的电池在25℃下用0.2C的恒定电流恒定电压充电法充电至4.2V后,以0.2C的恒定电流放电至3.0V。对其进行3个循环,将其第3个循环的放电容量作为循环后容量。然后,对同一电池在25℃下以0.2C的恒定电流恒定电压充电法充电至4.2V后,在-30℃下实施0.2C的恒定电流放电。将此时的放电容量作为循环后低温容量,将循环后低温容量相对于循环后容量的比例作为循环后低温放电率。In addition, the battery after the cycle test was charged to 4.2V at 25°C by a constant current constant voltage charging method of 0.2C, and then discharged to 3.0V by a constant current of 0.2C. Three cycles were performed on it, and the discharge capacity at the third cycle was taken as the capacity after cycle. Then, the same battery was charged to 4.2V at 25°C by a constant current constant voltage charging method of 0.2C, and then discharged at a constant current of 0.2C at -30°C. The discharge capacity at this time was taken as the post-cycle low-temperature capacity, and the ratio of the post-cycle low-temperature capacity to the post-cycle capacity was taken as the post-cycle low-temperature discharge rate.
电解液[2]实施例1Electrolyte [2] Example 1
在干燥氩氛围下,向碳酸亚乙酯(EC)、碳酸甲乙酯(EMC)和碳酸二乙酯(DEC)的混合物(体积比2∶4∶4)中以0.9mol/L加入六氟磷酸锂(LiPF6)并进行溶解,使该混合溶液中含有0.3质量%的根据Inorganic Nuclear ChemistryLetters(1969),5(7)的第581页~第582页中记载的方法制备的二氟磷酸锂,制备非水电解液。使用该非水电解液制造18650型圆筒电池,并测定循环保持率和低温放电率。结果如电解液[2]表1所示。而且,用离心分离机从循环后的低温容量测定后的电池中回收电解液,通过气相色谱(GC)对酯交换反应中生成的碳酸二甲酯(DMC)的量进行分析时,其量为电解液中的0.5质量%。Under a dry argon atmosphere, add lithium hexafluorophosphate (lithium hexafluorophosphate) at 0.9 mol/L to a mixture of ethylene carbonate (EC), ethylmethyl carbonate (EMC) and diethyl carbonate (DEC) (volume ratio 2:4:4). LiPF 6 ) was dissolved, and the mixed solution contained 0.3% by mass of lithium difluorophosphate prepared according to the method described on pages 581 to 582 of Inorganic Nuclear Chemistry Letters (1969), 5 (7), to prepare non- water electrolyte. A 18650-type cylindrical battery was manufactured using this non-aqueous electrolyte solution, and the cycle retention rate and low-temperature discharge rate were measured. The results are shown in Table 1 of the electrolyte [2]. Furthermore, when the electrolytic solution was recovered from the battery after the low-temperature capacity measurement after circulation by a centrifuge, and the amount of dimethyl carbonate (DMC) generated in the transesterification reaction was analyzed by gas chromatography (GC), the amount was 0.5% by mass in the electrolyte.
电解液[2]比较例1Electrolyte [2] Comparative Example 1
除了不含有二氟磷酸锂以外,与电解液[2]实施例1同样地制作18650型圆筒电池,并测定循环保持率和低温放电率。结果如电解液[2]表1所示。而且,用离心分离机从循环后的低温容量测定后的电池中回收电解液,通过气相色谱(GC)对酯交换反应中生成的碳酸二甲酯(DMC)的量进行分析时,其量为电解液中的9.7质量%。Except not containing lithium difluorophosphate, a 18650-type cylindrical battery was produced in the same manner as in the electrolyte solution [2] Example 1, and the cycle retention rate and low-temperature discharge rate were measured. The results are shown in Table 1 of the electrolyte [2]. Furthermore, when the electrolytic solution was recovered from the battery after the low-temperature capacity measurement after circulation by a centrifuge, and the amount of dimethyl carbonate (DMC) generated in the transesterification reaction was analyzed by gas chromatography (GC), the amount was 9.7% by mass in the electrolyte solution.
电解液[2]比较例2Electrolyte [2] Comparative Example 2
在干燥氩氛围下,向碳酸亚乙酯(EC)和碳酸二乙酯(DEC)的混合物(体积比2∶8)中以0.9mol/L加入六氟磷酸锂(LiPF6)并进行溶解,使该混合溶液中含有0.3质量%的二氟磷酸锂,制备非水电解液。使用该非水电解液制造18650型圆筒电池,并测定循环保持率和低温放电率。结果如电解液[2]表1所示。Under a dry argon atmosphere, lithium hexafluorophosphate (LiPF 6 ) was added at 0.9 mol/L to a mixture of ethylene carbonate (EC) and diethyl carbonate (DEC) (volume ratio 2:8) and dissolved, and the mixture The solution contained 0.3% by mass of lithium difluorophosphate to prepare a non-aqueous electrolytic solution. A 18650-type cylindrical battery was manufactured using this non-aqueous electrolyte solution, and the cycle retention rate and low-temperature discharge rate were measured. The results are shown in Table 1 of the electrolyte [2].
电解液[2]实施例2Electrolyte [2] Example 2
除了使用碳酸甲基正丙基酯来替代碳酸甲乙酯以外,与电解液[2]实施例1同样地制作18650型圆筒电池,并测定循环保持率和低温放电率。结果如电解液[2]表1所示。Except for using methyl-n-propyl carbonate instead of ethyl methyl carbonate, a 18650-type cylindrical battery was produced in the same manner as in the electrolyte solution [2] Example 1, and the cycle retention rate and low-temperature discharge rate were measured. The results are shown in Table 1 of the electrolyte [2].
电解液[2]比较例3Electrolyte [2] Comparative Example 3
除了不含有二氟磷酸锂以外,与电解液[2]实施例2同样地制作18650型圆筒电池,并测定循环保持率和低温放电率。结果如电解液[2]表1所示。Except not containing lithium difluorophosphate, a 18650-type cylindrical battery was produced in the same manner as in the electrolyte solution [2] Example 2, and the cycle retention rate and low-temperature discharge rate were measured. The results are shown in Table 1 of the electrolyte [2].
电解液[2]<二次电池的制作-2>Electrolyte solution [2] <Production of secondary battery-2>
<正极的制作><Production of positive electrode>
在N-甲基吡咯烷酮溶剂中混合90质量%作为正极活性物质的钴酸锂(LiCoO2)、5质量%的作为导电材料的乙炔黑和5质量%作为粘合剂的聚偏氟乙烯(PVdF),制成浆料。将得到的浆料涂布在厚度15μm的铝箔两面,并干燥,用压制机压延成厚度80μm,再将其切成具有宽100mm、长100mm大小的活性物质层和宽30mm的未涂布部分的形状,作为正极。Mix 90% by mass of lithium cobaltate (LiCoO 2 ) as a positive electrode active material, 5% by mass of acetylene black as a conductive material, and 5% by mass of polyvinylidene fluoride (PVdF ) to make slurry. The obtained slurry was coated on both sides of an aluminum foil with a thickness of 15 μm, dried, rolled to a thickness of 80 μm with a press, and cut into an active material layer having a width of 100 mm and a length of 100 mm and an uncoated portion of a width of 30 mm. shape, as the positive electrode.
<负极的制作><Production of Negative Electrode>
在98重量份人造石墨粉末KS-44(timcal公司制造,商品名)中添加100重量份作为增稠剂的羧甲基纤维素钠的水性分散液(羧甲基纤维素钠的浓度为1质量%),2重量份作为粘合剂的苯乙烯-丁二烯橡胶的水性分散液(苯乙烯-丁二烯橡胶的浓度为50质量%),用分散器进行混合,制成浆料。将得到的浆料涂布在厚度10μm的铜箔两面,并干燥,用压制机压延成厚度为75μm,再将其切成具有宽104mm、长104mm大小的活性物质层和宽30mm的未涂布部分的形状,作为负极。In 98 parts by weight of artificial graphite powder KS-44 (manufactured by timcal company, trade name), add 100 parts by weight of an aqueous dispersion of sodium carboxymethyl cellulose as a thickener (the concentration of sodium carboxymethyl cellulose is 1 mass %), 2 parts by weight of an aqueous dispersion of styrene-butadiene rubber as a binder (the concentration of styrene-butadiene rubber is 50% by mass), and mixed with a disperser to prepare a slurry. The obtained slurry was coated on both sides of a copper foil with a thickness of 10 μm, dried, rolled to a thickness of 75 μm with a press, and then cut into an active material layer with a width of 104 mm and a length of 104 mm and an uncoated layer with a width of 30 mm. The shape of the part, as the negative pole.
<电池的组装><Assembly of battery>
将32片正极和33片负极交替配置,各电极之间夹入多孔聚乙烯片隔板(厚度为25μm),进行叠层。此时,使正极活性物质面与负极活性物质面相对,并且不会露出负极活性物质面之外。将该正极和负极各自的未涂布部分焊接,制作集电片,将电极组密封在电池罐(外部尺寸:120×110×10mm)中。然后,在装有电极组的电池罐中注入20mL非水电解液,使电极充分浸透,密封而制作方形电池。二次电池的1个电池外壳中收纳的电池要素具有的电容,即二次电池的额定放电容量,约为6安培小时(Ah),以10kHz交流法测定的直流电阻成分约为5毫欧(mΩ)。32 positive electrodes and 33 negative electrodes were arranged alternately, and a porous polyethylene sheet separator (thickness: 25 μm) was sandwiched between the electrodes for lamination. At this time, the surface of the positive electrode active material faces the surface of the negative electrode active material without protruding from the surface of the negative electrode active material. The respective uncoated parts of the positive and negative electrodes were welded to produce current collector sheets, and the electrode group was sealed in a battery can (outer dimensions: 120×110×10 mm). Then, 20 mL of non-aqueous electrolytic solution was poured into the battery can containing the electrode group, the electrodes were fully soaked, and sealed to produce a square battery. The capacitance of the battery element contained in one battery case of the secondary battery, that is, the rated discharge capacity of the secondary battery, is about 6 ampere hours (Ah), and the DC resistance component measured by the 10 kHz alternating current method is about 5 milliohms ( mΩ).
电解液[2]实施例3Electrolyte [2] Example 3
使用电解液[2]实施例1中使用的电解液来制作方形电池,并采用与电解液[2]实施例1相同的方法实施循环试验、低温试验,并测定循环保持率和低温放电率。结果如电解液[2]表1所示。Use the electrolytic solution used in the electrolytic solution [2] Example 1 to make a square battery, and adopt the same method as the electrolytic solution [2] Example 1 to implement the cycle test and low temperature test, and measure the cycle retention rate and low temperature discharge rate. The results are shown in Table 1 of the electrolyte [2].
电解液[2]比较例4Electrolyte [2] Comparative Example 4
使用电解液[2]比较例1中使用的电解液来制作方形电池,实施循环试验、低温试验,并测定循环保持率和低温放电率。结果如电解液[2]表1所示。Using the electrolytic solution used in the electrolytic solution [2] Comparative Example 1, a prismatic battery was produced, a cycle test and a low temperature test were performed, and the cycle retention rate and low temperature discharge rate were measured. The results are shown in Table 1 of the electrolyte [2].
电解液[2]比较例5Electrolyte [2] Comparative Example 5
使用电解液[2]比较例2中使用的电解液来制作方形电池,实施循环试验、低温试验,并测定循环保持率和低温放电率。结果如电解液[2]表1所示。Using the electrolytic solution used in the electrolytic solution [2] Comparative Example 2, a prismatic battery was produced, a cycle test and a low temperature test were performed, and the cycle retention rate and low temperature discharge rate were measured. The results are shown in Table 1 of the electrolyte [2].
电解液[2]表1Electrolyte [2] Table 1
[表55][Table 55]
由电解液[2]表1可知,若对每个圆筒电池(电解液[2]实施例1、2,电解液[2]比较例1~3)、方形电池(电解液[2]实施例3,电解液[2]比较例4、5)进行比较,则非水电解液中同时含有不对称链状碳酸酯和二氟磷酸盐的电解液[2]实施例的锂二次电池,与不含有这些任意一种的电解液[2]比较例的锂二次电池相比,循环保持率、低温放电率都得到改善。From the electrolyte [2] Table 1, it can be seen that if each cylindrical battery (electrolyte [2] embodiment 1, 2, electrolyte [2] comparative example 1-3), square battery (electrolyte [2] Example 3, electrolytic solution [2] comparative example 4,5) compare, then contain the lithium secondary battery of the electrolytic solution [2] embodiment of asymmetric chain carbonate and difluorophosphate in the non-aqueous electrolytic solution simultaneously, Compared with the lithium secondary battery of the comparative example of the electrolytic solution [2] not containing any of these, both the cycle retention rate and the low-temperature discharge rate were improved.
如上所述,本实施例与本比较例的圆筒电池的额定放电容量小于3安培小时(Ah),直流电阻成分大于10毫欧(mΩ)。另一方面,本实施例与本比较例的方形电池的额定放电容量为3安培小时(Ah)以上,直流电阻成分为10毫欧(mΩ)以下。即,本实施例、本比较例的方形电池与圆筒电池相比,电阻小,电容大。而且,与电解液[2]实施例1相对于电解液[2]比较例1的低温特性提高程度相比,电解液[2]实施例3相对于电解液[2]比较例4的低温特性提高程度大,在电容大的二次电池或直流电阻小的二次电池中,本发明的效果更大。As mentioned above, the rated discharge capacity of the cylindrical batteries of this embodiment and this comparative example is less than 3 ampere hours (Ah), and the DC resistance component is greater than 10 milliohms (mΩ). On the other hand, the prismatic batteries of this example and this comparative example have a rated discharge capacity of 3 ampere hours (Ah) or more, and a DC resistance component of 10 milliohms (mΩ) or less. That is, the prismatic batteries of this example and this comparative example have lower resistance and higher capacitance than cylindrical batteries. Moreover, compared with the improvement degree of the low-temperature characteristics of the electrolyte [2] Example 1 relative to the electrolyte [2] Comparative Example 1, the low-temperature characteristics of the electrolyte [2] Example 3 relative to the electrolyte [2] Comparative Example 4 The degree of improvement is large, and the effect of the present invention is greater in a secondary battery with a large capacity or a secondary battery with a small DC resistance.
电解液[3]<二次电池的制造-1>Electrolyte solution [3] <Manufacture of secondary battery-1>
[正极的制造][production of positive electrode]
在N-甲基吡咯烷酮溶剂中混合90质量%作为正极活性物质的钴酸锂(LiCoO2)、5质量%的作为导电材料的乙炔黑和5质量%作为粘合剂的聚偏氟乙烯(PVdF),制成浆料。将得到的浆料涂布在厚度15μm的铝箔两面,并干燥,用压制机压延成厚度80μm,再将其切成具有宽100mm、长100mm大小的活性物质层和宽30mm的未涂布部分的形状,作为正极。Mix 90% by mass of lithium cobaltate (LiCoO 2 ) as a positive electrode active material, 5% by mass of acetylene black as a conductive material, and 5% by mass of polyvinylidene fluoride (PVdF ) to make slurry. The obtained slurry was coated on both sides of an aluminum foil with a thickness of 15 μm, dried, rolled to a thickness of 80 μm with a press, and cut into an active material layer having a width of 100 mm and a length of 100 mm and an uncoated portion of a width of 30 mm. shape, as the positive electrode.
[负极的制造][production of negative electrode]
在98重量份人造石墨粉末KS-44(timcal公司制造,商品名)中添加100重量份作为增稠剂的羧甲基纤维素钠的水性分散液(羧甲基纤维素钠的浓度为1质量%)、2重量份作为粘合剂的苯乙烯-丁二烯橡胶的水性分散液(苯乙烯-丁二烯橡胶的浓度为50质量%),用分散器进行混合,制成浆料。将得到的浆料涂布在厚度10μm的铜箔两面,并干燥,用压制机压延成厚度为75μm,再将其切成具有宽104mm、长104mm大小的活性物质层和宽30mm的未涂布部分的形状,作为负极。In 98 parts by weight of artificial graphite powder KS-44 (manufactured by timcal company, trade name), add 100 parts by weight of an aqueous dispersion of sodium carboxymethyl cellulose as a thickener (the concentration of sodium carboxymethyl cellulose is 1 mass %), 2 parts by weight of an aqueous dispersion of styrene-butadiene rubber as a binder (the concentration of styrene-butadiene rubber is 50% by mass), and mixed with a disperser to prepare a slurry. The obtained slurry was coated on both sides of a copper foil with a thickness of 10 μm, dried, rolled to a thickness of 75 μm with a press, and then cut into an active material layer with a width of 104 mm and a length of 104 mm and an uncoated layer with a width of 30 mm. The shape of the part, as the negative pole.
[电池的组装][assembling the battery]
将32片正极和33片负极交替配置,各电极之间夹入多孔聚乙烯片隔板(厚度为25μm),进行叠层。此时,使正极活性物质面与负极活性物质面相对,并且不会露出负极活性物质面之外。将该正极和负极各自的未涂布部分焊接,制作集电片,将电极组密封在电池罐(外部尺寸:120×110×10mm)中。32 positive electrodes and 33 negative electrodes were arranged alternately, and a porous polyethylene sheet separator (thickness: 25 μm) was sandwiched between the electrodes for lamination. At this time, the surface of the positive electrode active material faces the surface of the negative electrode active material without protruding from the surface of the negative electrode active material. The respective uncoated parts of the positive and negative electrodes were welded to produce current collector sheets, and the electrode group was sealed in a battery can (outer dimensions: 120×110×10 mm).
然后,在装有电极组的电池罐中注入20mL下述电解液[3]实施例和电解液[3]比较例的非水电解液,使电极充分浸透,密封而制作方形电池。该电池的额定放电容量约为6安培小时(Ah),以10kHz交流法测定的直流电阻成分约为5毫欧(mΩ)。此外,上述正极的电极面积的总和相对于该二次电池的外壳的表面积的面积比为20.65倍。Then, 20 mL of the non-aqueous electrolytes of the following electrolyte solution [3] example and electrolyte solution [3] comparative example were injected into the battery can containing the electrode group, the electrodes were fully soaked, and sealed to produce a square battery. The rated discharge capacity of the battery is about 6 ampere hours (Ah), and the DC resistance component measured by the 10 kHz alternating current method is about 5 milliohms (mΩ). In addition, the area ratio of the sum of the electrode areas of the positive electrode to the surface area of the case of the secondary battery was 20.65 times.
[电池的评价][evaluation of the battery]
(容量测定)(capacity measurement)
对未经充放电的新电池,在25℃、4.1V~3.0V的电压范围进行5个循环的初期充放电(电压范围4.1V~3.0V)。将此时的第5个循环的0.2C(将1小时放电额定容量的电流值作为1C,所述额定容量取决于1小时率(one-hour-rate)的放电容量,下同)放电容量作为初期容量。For a new battery that has not been charged and discharged, 5 cycles of initial charge and discharge (voltage range 4.1V to 3.0V) were performed at 25°C in a voltage range of 4.1V to 3.0V. The 0.2C discharge capacity of the 5th cycle at this time (the current value of the 1-hour discharge rated capacity is taken as 1C, and the rated capacity depends on the discharge capacity of the 1-hour-rate (one-hour-rate), the same below) discharge capacity as initial capacity.
(输出功率测定)(Output power measurement)
在25℃环境下以0.2C的恒定电流进行150分钟充电,在-30℃环境下分别以0.1C、0.3C、1.0C、3.0C、5.0C放电10秒,测定第10秒的电压。将由电流-电压直线与下限电压(3V)围成的三角形的面积作为输出功率(W)。Charge at a constant current of 0.2C for 150 minutes at 25°C, discharge at 0.1C, 0.3C, 1.0C, 3.0C, and 5.0C for 10 seconds at -30°C, and measure the voltage at the 10th second. The area of the triangle surrounded by the current-voltage straight line and the lower limit voltage (3V) is taken as the output power (W).
电解液[3]实施例1Electrolyte [3] Embodiment 1
在干燥氩氛围下,向碳酸亚乙酯(EC)、碳酸甲乙酯(EMC)和丙酸甲酯(MP)的混合物(体积比3∶6∶1)中以1mol/L加入六氟磷酸锂(LiPF6)并进行溶解,然后在该混合溶液中混合0.3质量%的量的六甲基环三硅氧烷,制备非水电解液。使用该非水电解液用上述方法制作电池,并测定低温输出功率。结果如电解液[3]表1所示。Under a dry argon atmosphere, lithium hexafluorophosphate (LiPF 6 ) and dissolving, and then mixing hexamethylcyclotrisiloxane in an amount of 0.3% by mass in the mixed solution to prepare a nonaqueous electrolytic solution. Using this non-aqueous electrolytic solution, a battery was produced by the method described above, and the low-temperature output was measured. The results are shown in Table 1 of the electrolyte [3].
电解液[3]实施例2Electrolyte [3] Embodiment 2
使用用等量的乙酸乙酯(EA)来替代电解液[3]实施例1中的丙酸甲酯而制备的非水电解液来制作电池,并测定低温输出功率。结果如电解液[3]表1所示。The non-aqueous electrolyte solution prepared by using an equal amount of ethyl acetate (EA) instead of methyl propionate in Example 1 of the electrolyte solution [3] was used to make a battery, and the low-temperature output power was measured. The results are shown in Table 1 of the electrolyte [3].
电解液[3]实施例3Electrolyte [3] Example 3
使用用等量的乙酸甲酯(MA)来替代电解液[3]实施例1中的丙酸甲酯而制备的非水电解液来制作电池,并测定低温输出功率。结果如电解液[3]表1所示。The non-aqueous electrolytic solution prepared by using an equal amount of methyl acetate (MA) instead of methyl propionate in Example 1 of the electrolytic solution [3] was used to make a battery, and the low-temperature output power was measured. The results are shown in Table 1 of the electrolyte [3].
电解液[3]实施例4Electrolyte [3] Example 4
在干燥氩氛围下,向碳酸亚乙酯(EC)、碳酸甲乙酯(EMC)、丙酸甲酯(MP)和乙酸甲酯(MA)的混合物(体积比30∶60∶5∶5)中以1mol/L加入六氟磷酸锂(LiPF6)并进行溶解,然后在该混合溶液中混合0.3质量%的量的六甲基环三硅氧烷,制备非水电解液。使用该非水电解液用上述方法制作电池,并测定低温输出功率。结果如电解液[3]表1所示。Under a dry argon atmosphere, a mixture of ethylene carbonate (EC), ethyl methyl carbonate (EMC), methyl propionate (MP) and methyl acetate (MA) (volume ratio 30:60:5:5) Lithium hexafluorophosphate (LiPF 6 ) was added and dissolved at 1 mol/L, and then 0.3% by mass of hexamethylcyclotrisiloxane was mixed in the mixed solution to prepare a non-aqueous electrolytic solution. Using this non-aqueous electrolytic solution, a battery was produced by the method described above, and the low-temperature output was measured. The results are shown in Table 1 of the electrolyte [3].
电解液[3]实施例5Electrolyte [3] Embodiment 5
使用电解液[3]实施例1中将碳酸亚乙酯(EC)、碳酸甲乙酯(EMC)和丙酸甲酯(MP)的混合物的体积比变更为3∶4∶3而制备的非水电解液,按照上述方法制作电池,并测定低温输出功率。结果如电解液[3]表1所示。Using the electrolyte solution [3] in Example 1, the volume ratio of the mixture of ethylene carbonate (EC), ethyl methyl carbonate (EMC) and methyl propionate (MP) was changed to 3:4:3. Water electrolyte, according to the above method to make the battery, and measure the low temperature output power. The results are shown in Table 1 of the electrolyte [3].
电解液[3]实施例6Electrolyte [3] Embodiment 6
在干燥氩氛围下,向碳酸亚乙酯(EC)和丙酸甲酯(MP)的混合物(体积比3∶7)中以1mol/L加入六氟磷酸锂(LiPF6)并进行溶解,然后在该混合溶液中混合0.3质量%的量的六甲基环三硅氧烷,制备非水电解液。使用该非水电解液用上述方法制作电池,并测定低温输出功率。结果如电解液[3]表1所示。Under a dry argon atmosphere, lithium hexafluorophosphate (LiPF 6 ) was added at 1 mol/L to a mixture of ethylene carbonate (EC) and methyl propionate (MP) (volume ratio 3:7) and dissolved, and then mixed Hexamethylcyclotrisiloxane was mixed in an amount of 0.3% by mass in the solution to prepare a non-aqueous electrolytic solution. Using this non-aqueous electrolytic solution, a battery was produced by the method described above, and the low-temperature output was measured. The results are shown in Table 1 of the electrolyte [3].
电解液[3]实施例7Electrolyte [3] Embodiment 7
使用混合等量的苯基二甲基氟硅烷来替代电解液[3]实施例1中的六甲基环三硅氧烷而得到的非水电解液作为非水电解液来制作电池,并测定低温输出功率。结果如电解液[3]表1所示。The non-aqueous electrolytic solution obtained by mixing an equal amount of phenyldimethylfluorosilane instead of the hexamethylcyclotrisiloxane in Example 1 of the electrolytic solution [3] is used as the non-aqueous electrolytic solution to make a battery, and measure Low temperature output power. The results are shown in Table 1 of the electrolyte [3].
电解液[3]实施例8Electrolyte [3] Embodiment 8
使用混合等量的六甲基环二硅氧烷来替代电解液[3]实施例1中的六甲基环三硅氧烷而得到的非水电解液作为非水电解液来制作电池,并测定低温输出功率。结果如电解液[3]表1所示。Use the hexamethylcyclotrisiloxane of mixing equal amount to replace the hexamethylcyclotrisiloxane in the electrolyte [3] embodiment 1 and obtain the non-aqueous electrolytic solution as the non-aqueous electrolytic solution to make the battery, and Determination of low temperature output power. The results are shown in Table 1 of the electrolyte [3].
电解液[3]实施例9Electrolyte [3] Embodiment 9
使用混合等量的甲磺酸三甲基甲硅烷基酯来替代电解液[3]实施例1中的六甲基环三硅氧烷而得到的非水电解液作为非水电解液来制作电池,并测定低温输出功率。结果如电解液[3]表1所示。Use the non-aqueous electrolytic solution obtained by mixing an equal amount of trimethylsilyl methanesulfonate to replace the hexamethylcyclotrisiloxane in the electrolytic solution [3] Example 1 as the non-aqueous electrolytic solution to make a battery , and determine the low temperature output power. The results are shown in Table 1 of the electrolyte [3].
电解液[3]实施例10Electrolyte [3] Embodiment 10
使用混合等量的氟磺酸甲酯来替代电解液[3]实施例1中的六甲基环三硅氧烷而得到的非水电解液作为非水电解液来制作电池,并测定低温输出功率。结果如电解液[3]表1所示。Use the non-aqueous electrolytic solution obtained by mixing an equal amount of methyl fluorosulfonate instead of hexamethylcyclotrisiloxane in the electrolytic solution [3] Example 1 as the non-aqueous electrolytic solution to make a battery, and measure the low-temperature output power. The results are shown in Table 1 of the electrolyte [3].
电解液[3]实施例11Electrolyte [3] Example 11
使用混合等量的硝酸锂来替代电解液[3]实施例1中的六甲基环三硅氧烷、并保存3天而得到的非水电解液作为非水电解液来制作电池,并测定低温输出功率。结果如电解液[3]表1所示。Use the same amount of lithium nitrate to replace the hexamethylcyclotrisiloxane in the electrolyte [3] embodiment 1, and store the non-aqueous electrolyte obtained for 3 days as the non-aqueous electrolyte to make a battery, and measure Low temperature output power. The results are shown in Table 1 of the electrolyte [3].
电解液[3]实施例12Electrolyte [3] Example 12
使用混合等量(相对于非水溶剂和六氟磷酸锂的混合溶液为0.3质量%的量)的根据Inorganic Nuclear Chemistry Letters(1969),5(7)的第581页~第582页中记载的方法制备的二氟磷酸锂来替代电解液[3]实施例1中的六甲基环三硅氧烷而制备的非水电解液作为非水电解液,制作电池,并测定低温输出功率。结果如电解液[3]表1所示。Prepared according to the method described in Inorganic Nuclear Chemistry Letters (1969), 5(7), pages 581 to 582, by mixing equal amounts (0.3% by mass relative to a mixed solution of a non-aqueous solvent and lithium hexafluorophosphate) Lithium difluorophosphate is used to replace the hexamethylcyclotrisiloxane in the electrolyte solution [3] and the non-aqueous electrolyte solution prepared in Example 1 is used as the non-aqueous electrolyte solution to make a battery and measure the low-temperature output power. The results are shown in Table 1 of the electrolyte [3].
电解液[3]实施例13Electrolyte [3] Example 13
使用电解液[3]实施例12中的二氟磷酸锂的混合量变更为相对于非水溶剂和六氟磷酸锂的混合溶液为0.08质量%的量而制备的非水电解液,制作电池,并测定低温输出功率。结果如电解液[3]表1所示。Using the non-aqueous electrolytic solution prepared by changing the mixed amount of lithium difluorophosphate in Example 12 to 0.08% by mass relative to the mixed solution of the non-aqueous solvent and lithium hexafluorophosphate, a battery was produced, and the low-temperature Output Power. The results are shown in Table 1 of the electrolyte [3].
电解液[3]实施例14Electrolyte [3] Example 14
<二次电池的制作-2><Production of secondary battery-2>
<正极的制作><Production of positive electrode>
在N-甲基吡咯烷酮溶剂中混合90质量%作为正极活性物质的钴酸锂(LiCoO2)、5质量%的作为导电材料的乙炔黑和5质量%作为粘合剂的聚偏氟乙烯(PVdF),制成浆料。将得到的浆料涂布在厚度20μm的铝箔两面,并干燥,用压制机压延成厚度80μm,再将其切成宽52mm、长830mm大小,作为正极。其中,表面背面都在长度方向上设置50mm的未涂布部分,活性物质层的长度为780mm。Mix 90% by mass of lithium cobaltate (LiCoO 2 ) as a positive electrode active material, 5% by mass of acetylene black as a conductive material, and 5% by mass of polyvinylidene fluoride (PVdF ) to make slurry. The obtained slurry was coated on both sides of an aluminum foil with a thickness of 20 μm, dried, and rolled to a thickness of 80 μm by a press machine, and then cut into a size of 52 mm in width and 830 mm in length as a positive electrode. Among them, an uncoated portion of 50 mm was provided in the longitudinal direction on both the front and back sides, and the length of the active material layer was 780 mm.
<负极的制作><Production of Negative Electrode>
在98重量份人造石墨粉末KS-44(timcal公司制造,商品名)中添加100重量份作为增稠剂的羧甲基纤维素钠的水性分散液(羧甲基纤维素钠的浓度为1质量%)、2重量份作为粘合剂的苯乙烯-丁二烯橡胶的水性分散液(苯乙烯-丁二烯橡胶的浓度为50质量%),用分散器进行混合,制成浆料。将得到的浆料均匀涂布在作为负极集电体的厚度18μm的铜箔的两面,干燥后,用压制机压延成厚度为85μm,再将其切成宽56mm、长850mm的大小,作为负极。其中,表面背面都在长度方向上设置30mm的未涂布部分。In 98 parts by weight of artificial graphite powder KS-44 (manufactured by timcal company, trade name), add 100 parts by weight of an aqueous dispersion of sodium carboxymethyl cellulose as a thickener (the concentration of sodium carboxymethyl cellulose is 1 mass %), 2 parts by weight of an aqueous dispersion of styrene-butadiene rubber as a binder (the concentration of styrene-butadiene rubber is 50% by mass), and mixed with a disperser to prepare a slurry. The obtained slurry was uniformly coated on both sides of a copper foil with a thickness of 18 μm as the negative electrode current collector, and after drying, it was rolled to a thickness of 85 μm by a press, and then cut into a size of 56 mm wide and 850 mm long, as the negative electrode . Among them, an uncoated portion of 30 mm was provided in the longitudinal direction on both the front and back sides.
[电解液的制备][Preparation of Electrolyte Solution]
制备与电解液[3]实施例1相同的非水电解液。Prepare the same non-aqueous electrolytic solution as in Example 1 of the electrolytic solution [3].
[电池的组装][assembling the battery]
将正极和负极与聚乙烯制的隔板一起重叠卷绕并使得正极与负极不直接接触,制成电极体。使正极和负极的端子露出外部地收纳在电池罐中。然后,向其中注入5mL后述的电解液后,进行铆接成型,制造18650型圆筒电池。该电池的额定放电容量约为0.7Ah,用10kHz交流法测定的直流电阻约为35毫欧(mΩ)。此外,上述正极的电极面积的总和相对于该二次电池的外壳的表面积的面积比为19.4倍。上述电池中,与电解液[3]实施例1同样地测定低温输出功率。结果如电解液[3]表1所示。The positive electrode and the negative electrode were overlapped and wound together with a polyethylene separator so that the positive electrode and the negative electrode were not in direct contact to form an electrode body. The positive and negative terminals are housed in the battery can with the terminals exposed to the outside. Then, 5 mL of an electrolytic solution described later was injected thereinto, followed by riveting molding to manufacture a 18650-type cylindrical battery. The rated discharge capacity of the battery is about 0.7 Ah, and the DC resistance measured by the 10 kHz AC method is about 35 milliohms (mΩ). In addition, the area ratio of the sum of the electrode areas of the positive electrode to the surface area of the case of the secondary battery was 19.4 times. In the above battery, the low-temperature output was measured in the same manner as in Example 1 of the electrolytic solution [3]. The results are shown in Table 1 of the electrolyte [3].
电解液[3]比较例1Electrolyte [3] Comparative Example 1
使用电解液[3]实施例1中不混合六甲基环三硅氧烷而制备的非水电解液,用上述方法制作电池,并测定低温输出功率。结果如电解液[3]表1所示。Using the non-aqueous electrolyte solution prepared without mixing hexamethylcyclotrisiloxane in Example 1 of the electrolyte solution [3], a battery was fabricated by the above method, and the low-temperature output power was measured. The results are shown in Table 1 of the electrolyte [3].
电解液[3]比较例2Electrolyte [3] Comparative Example 2
使用电解液[3]实施例2中不混合六甲基环三硅氧烷而制备的非水电解液,用上述方法制作电池,并测定低温输出功率。结果如电解液[3]表1所示。Using the non-aqueous electrolyte solution prepared without mixing hexamethylcyclotrisiloxane in Example 2 of the electrolyte solution [3], a battery was fabricated by the above method, and the low-temperature output power was measured. The results are shown in Table 1 of the electrolyte [3].
电解液[3]比较例3Electrolyte [3] Comparative Example 3
使用电解液[3]实施例3中不混合六甲基环三硅氧烷而制备的非水电解液,用上述方法制作电池,并测定低温输出功率。结果如电解液[3]表1所示。Using the non-aqueous electrolyte solution prepared without mixing hexamethylcyclotrisiloxane in Example 3 of the electrolyte solution [3], a battery was fabricated by the above method, and the low-temperature output power was measured. The results are shown in Table 1 of the electrolyte [3].
电解液[3]比较例4Electrolyte [3] Comparative Example 4
使用电解液[3]实施例4中不混合六甲基环三硅氧烷而制备的非水电解液,用上述方法制作电池,并测定低温输出功率。结果如电解液[3]表1所示。Using the non-aqueous electrolytic solution prepared without mixing hexamethylcyclotrisiloxane in the electrolytic solution [3] Example 4, a battery was fabricated by the above method, and the low-temperature output power was measured. The results are shown in Table 1 of the electrolyte [3].
电解液[3]比较例5Electrolyte [3] Comparative Example 5
使用电解液[3]实施例5中不混合六甲基环三硅氧烷而制备的非水电解液,用上述方法制作电池,并测定低温输出功率。结果如电解液[3]表1所示。Using the non-aqueous electrolyte solution prepared without mixing hexamethylcyclotrisiloxane in Example 5 of the electrolyte solution [3], a battery was fabricated by the above-mentioned method, and the low-temperature output power was measured. The results are shown in Table 1 of the electrolyte [3].
电解液[3]比较例6Electrolyte [3] Comparative Example 6
在干燥氩氛围下,向碳酸亚乙酯和碳酸甲乙酯的混合物(体积比3∶7)中以1mol/L加入六氟磷酸锂(LiPF6)并进行溶解,制备非水电解液。使用该非水电解液用上述方法制作电池,并测定低温输出功率。结果如电解液[3]表1所示。Under a dry argon atmosphere, lithium hexafluorophosphate (LiPF 6 ) was added at 1 mol/L to a mixture of ethylene carbonate and ethyl methyl carbonate (volume ratio 3:7) and dissolved to prepare a non-aqueous electrolyte. Using this non-aqueous electrolytic solution, a battery was produced by the method described above, and the low-temperature output was measured. The results are shown in Table 1 of the electrolyte [3].
电解液[3]比较例7Electrolyte [3] Comparative Example 7
在干燥氩氛围下,向碳酸亚乙酯和碳酸甲乙酯的混合物(体积比3∶7)中以1mol/L加入六氟磷酸锂(LiPF6)并进行溶解,然后在该混合溶液中混合0.3质量%的量的六甲基环三硅氧烷,制备非水电解液。使用该非水电解液用上述方法制作电池,并测定低温输出功率。结果如电解液[3]表1所示。In a dry argon atmosphere, lithium hexafluorophosphate (LiPF 6 ) was added at 1 mol/L to a mixture of ethylene carbonate and ethyl methyl carbonate (volume ratio 3:7) to dissolve it, and then mixed 0.3% by mass in the mixed solution amount of hexamethylcyclotrisiloxane to prepare a non-aqueous electrolyte. Using this non-aqueous electrolytic solution, a battery was produced by the method described above, and the low-temperature output was measured. The results are shown in Table 1 of the electrolyte [3].
电解液[3]比较例8Electrolyte [3] Comparative Example 8
使用混合等量的苯基二甲基氟硅烷来替代电解液[3]比较例7中的六甲基环三硅氧烷而得到的非水电解液作为非水电解液,用上述方法制作电池,并测定低温输出功率。结果如电解液[3]表1所示。Use the non-aqueous electrolyte obtained by mixing an equal amount of phenyldimethylfluorosilane instead of hexamethylcyclotrisiloxane in the electrolyte [3] Comparative Example 7 as the non-aqueous electrolyte, and make the battery by the above method , and determine the low temperature output power. The results are shown in Table 1 of the electrolyte [3].
电解液[3]比较例9Electrolyte [3] Comparative Example 9
使用混合等量的六甲基环二硅氧烷来替代电解液[3]比较例7中的六甲基环三硅氧烷而得到的非水电解液作为非水电解液,用上述方法制作电池,并测定低温输出功率。结果如电解液[3]表1所示。The non-aqueous electrolytic solution obtained by mixing an equal amount of hexamethylcyclotrisiloxane instead of the hexamethylcyclotrisiloxane in the electrolyte [3] Comparative Example 7 was made by the above-mentioned method battery, and determine the low temperature output power. The results are shown in Table 1 of the electrolyte [3].
电解液[3]比较例10Electrolyte [3] Comparative Example 10
使用混合等量的甲磺酸三甲基甲硅烷基酯来替代电解液[3]比较例7中的六甲基环三硅氧烷而得到的非水电解液作为非水电解液,用上述方法制作电池,并测定低温输出功率。结果如电解液[3]表1所示。The non-aqueous electrolytic solution obtained by mixing an equal amount of trimethylsilyl methanesulfonate to replace the hexamethylcyclotrisiloxane in the electrolytic solution [3] Comparative Example 7 is used as the non-aqueous electrolytic solution. Methods The battery was fabricated and the output power at low temperature was measured. The results are shown in Table 1 of the electrolyte [3].
电解液[3]比较例11Electrolyte [3] Comparative Example 11
使用混合等量的氟磺酸甲酯来替代电解液[3]比较例7中的六甲基环三硅氧烷而得到的非水电解液作为非水电解液,用上述方法制作电池,并测定低温输出功率。结果如电解液[3]表1所示。Use the non-aqueous electrolytic solution obtained as non-aqueous electrolytic solution to replace the hexamethylcyclotrisiloxane in electrolytic solution [3] comparative example 7 with the same amount of fluorosulfonic acid methyl ester, make battery with the above-mentioned method, and Determination of low temperature output power. The results are shown in Table 1 of the electrolyte [3].
电解液[3]比较例12Electrolyte [3] Comparative Example 12
使用混合等量的硝酸锂来替代电解液[3]比较例7中的六甲基环三硅氧烷、并保存3天而得到的非水电解液作为非水电解液,用上述方法制作电池,并测定低温输出功率。结果如电解液[3]表1所示。Use the same amount of lithium nitrate to replace the hexamethylcyclotrisiloxane in the electrolyte [3] Comparative Example 7, and store the non-aqueous electrolyte obtained for 3 days as the non-aqueous electrolyte, and make the battery by the above method , and determine the low temperature output power. The results are shown in Table 1 of the electrolyte [3].
电解液[3]比较例13Electrolyte [3] Comparative Example 13
使用混合等量的二氟磷酸锂来替代电解液[3]比较例7中的六甲基环三硅氧烷而得到的非水电解液作为非水电解液,用上述方法制作电池,并测定低温输出功率。结果如电解液[3]表1所示。Use the non-aqueous electrolyte obtained by mixing an equal amount of lithium difluorophosphate instead of hexamethylcyclotrisiloxane in Comparative Example 7 of the electrolyte [3] as the non-aqueous electrolyte, make a battery by the above method, and measure Low temperature output power. The results are shown in Table 1 of the electrolyte [3].
电解液[3]比较例14Electrolyte [3] Comparative Example 14
电解液[3]实施例14中,使用与电解液[3]比较例1同样地制备的非水电解液,用上述方法制作电池,并测定低温输出功率。结果如电解液[3]表1所示。Electrolyte solution [3] In Example 14, using the non-aqueous electrolyte solution prepared in the same manner as electrolyte solution [3] Comparative Example 1, a battery was produced by the above-mentioned method, and the low-temperature output was measured. The results are shown in Table 1 of the electrolyte [3].
电解液[3]表1Electrolyte [3] Table 1
[表56][Table 56]
由电解液[3]表1可知,在非水电解液中含有链状羧酸酯和特定化合物的电解液[3]实施例1~13的锂二次电池,不仅与不含其中任何一种的电解液[3]比较例6的锂二次电池相比,而且即使与不含特定化合物的电解液[3]比较例1~5、不含有链状羧酸酯的电解液[3]比较例7~13的锂二次电池相比,低温输出功率特性也得到改善。此外,该效果不是两者的单纯叠加,通过使用两者,效果明显增强。It can be seen from Table 1 of the electrolyte solution [3] that the lithium secondary batteries of the electrolyte solution [3] Examples 1 to 13 containing chain carboxylic acid esters and specific compounds in the non-aqueous electrolyte solution are not only compatible with those that do not contain any of them. Compared with the lithium secondary battery of the electrolyte [3] comparative example 6, and even compared with the electrolyte [3] comparative examples 1 to 5 that do not contain a specific compound, and the electrolyte [3] that does not contain a chain carboxylate Compared with the lithium secondary batteries of Examples 7 to 13, the low-temperature output characteristics were also improved. In addition, the effect is not a simple superposition of the two, but the effect is significantly enhanced by using both.
此外,电解液[3]实施例14相对于电解液[3]比较例14的输出功率上升率约为21%,而使用相同的材料、电池结构不同的电解液[3]实施例1对于电解液[3]比较例1的输出功率上升率约为29%,可知电池结构对本发明的非水电解液的效果大小有影响。In addition, the increase rate of the output power of the electrolyte [3] Example 14 relative to the electrolyte [3] Comparative Example 14 is about 21%, while using the same material and different battery structures Liquid [3] The output power increase rate of Comparative Example 1 is about 29%. It can be seen that the battery structure has an influence on the effect of the non-aqueous electrolyte of the present invention.
如上所述,通过使用本发明的非水电解液,即具有下述特征的二次电池用非水电解液,可以发挥非常大的低温输出功率特性,所述二次电池用非水电解液的特征是:含有至少一种以上链状羧酸酯,还含有选自下述物质中的至少一种化合物,并且其在全部非水电解液中的含量为10ppm以上,所述物质包括:通式(1)表示的环状硅氧烷化合物、通式(2)表示的氟硅烷化合物、通式(3)表示的化合物、分子内具有S-F键的化合物、硝酸盐、亚硝酸盐、单氟磷酸盐、二氟磷酸盐、乙酸盐和丙酸盐。As mentioned above, by using the non-aqueous electrolytic solution of the present invention, that is, a non-aqueous electrolytic solution for secondary batteries having the following characteristics, very large low-temperature output power characteristics can be brought into play. It is characterized by: containing at least one chain carboxylate, and at least one compound selected from the following substances, and its content in all non-aqueous electrolytes is more than 10ppm. The substances include: general formula Cyclic siloxane compounds represented by (1), fluorosilane compounds represented by general formula (2), compounds represented by general formula (3), compounds having an S-F bond in the molecule, nitrates, nitrites, monofluorophosphoric acid salt, difluorophosphate, acetate and propionate.
电解液[4]<二次电池的制作-1(方形电池的制造)>Electrolyte solution [4] <Production of secondary battery-1 (production of square battery)>
[正极的制作][making of positive electrode]
在N-甲基吡咯烷酮溶剂中混合90质量%作为正极活性物质的钴酸锂(LiCoO2)、5质量%的作为导电材料的乙炔黑和5质量%作为粘合剂的聚偏氟乙烯(PVdF),制成浆料。将得到的浆料涂布在厚度15μm的铝箔两面,并干燥,用压制机压延成厚度80μm,再将其切成具有宽100mm、长100mm大小的活性物质层和宽30mm的未涂布部分的形状,作为正极。Mix 90% by mass of lithium cobaltate (LiCoO 2 ) as a positive electrode active material, 5% by mass of acetylene black as a conductive material, and 5% by mass of polyvinylidene fluoride (PVdF ) to make slurry. The obtained slurry was coated on both sides of an aluminum foil with a thickness of 15 μm, dried, rolled to a thickness of 80 μm with a press, and cut into an active material layer having a width of 100 mm and a length of 100 mm and an uncoated portion of a width of 30 mm. shape, as the positive electrode.
[负极的制作][Production of Negative Electrode]
将2kg人造石墨粉末KS-44(timcal公司制造,商品名)和1kg石油类沥青混合,将得到的浆状的混合物在间歇式加热炉中于非活性氛围下升温2小时直至1100℃,在该温度下保持2小时。2kg of artificial graphite powder KS-44 (manufactured by timcal company, trade name) and 1kg of petroleum pitch were mixed, and the obtained slurry mixture was heated up to 1100° C. for 2 hours in a batch heating furnace under an inert atmosphere. Keep at temperature for 2 hours.
将其粉碎,通过振动式筛将粒径调整为18~22μm,最终得到以7质量%的无定形碳覆盖石墨表面的“无定形包覆石墨类碳质物质”。将本“无定形包覆石墨类碳质物质”作为负极活性物质,向98重量份“无定形包覆石墨类碳质物质”中添加100重量份作为增稠剂的羧甲基纤维素钠的水性分散液(羧甲基纤维素钠的浓度为1质量%)、2重量份作为粘合剂的苯乙烯-丁二烯橡胶的水性分散液(苯乙烯-丁二烯橡胶的浓度为50质量%),用分散器进行混合,制成浆料。将得到的浆料涂布在厚度10μm的铜箔两面,并干燥,用压制机压延成厚度为75μm,再将其切成具有宽104mm、长104mm大小的活性物质层和宽30mm的未涂布部分的形状,作为负极。This was pulverized, and the particle size was adjusted to 18 to 22 μm with a vibrating sieve to finally obtain an "amorphous-coated graphite-like carbonaceous material" in which the graphite surface was covered with 7% by mass of amorphous carbon. Using this "amorphous coated graphite-like carbonaceous material" as the negative active material, adding 100 parts by weight of sodium carboxymethylcellulose as a thickener to 98 parts by weight of the "amorphous coated graphite-based carbonaceous material" Aqueous dispersion (the concentration of sodium carboxymethylcellulose is 1% by mass), 2 parts by weight of an aqueous dispersion of styrene-butadiene rubber as a binder (the concentration of styrene-butadiene rubber is 50 mass %) %), mixed with a disperser to make a slurry. The obtained slurry was coated on both sides of a copper foil with a thickness of 10 μm, dried, rolled to a thickness of 75 μm with a press, and then cut into an active material layer with a width of 104 mm and a length of 104 mm and an uncoated layer with a width of 30 mm. The shape of the part, as the negative pole.
[电池的组装][assembling the battery]
将32片正极和33片负极交替配置,各电极之间夹入多孔聚乙烯片隔板(厚度为25μm),进行叠层。此时,使正极活性物质面与负极活性物质面相对,并且不会露出负极活性物质面之外。将该正极和负极各自的未涂布部分焊接,制作集电片,将电极组密封在带有排出阀的电池罐(外部尺寸:120×110×10mm)中。然后,在装有电极组的电池罐中注入20mL后述的非水电解液,使电极充分浸透,密封而制作方形电池。二次电池的1个电池外壳中收纳的电池要素所具有的电容,即该电池的额定放电容量约为6Ah,以10kHz交流法测定的直流电阻成分约为5毫欧。32 positive electrodes and 33 negative electrodes were arranged alternately, and a porous polyethylene sheet separator (thickness: 25 μm) was sandwiched between the electrodes for lamination. At this time, the surface of the positive electrode active material faces the surface of the negative electrode active material without protruding from the surface of the negative electrode active material. The respective uncoated portions of the positive and negative electrodes were welded to produce current collector sheets, and the electrode group was sealed in a battery can (outer dimensions: 120×110×10 mm) with a discharge valve. Then, 20 mL of a non-aqueous electrolytic solution described later was poured into the battery can containing the electrode group, the electrodes were sufficiently permeated, and sealed to produce a square battery. The capacitance of the battery element contained in one battery case of the secondary battery, that is, the rated discharge capacity of the battery is about 6Ah, and the DC resistance component measured by the 10kHz AC method is about 5 milliohms.
《电池的评价》"Battery Evaluation"
(容量测定)(capacity measurement)
对未经充放电循环的电池,在25℃、4.2V~3.0V的电压范围下,以0.2C的电流值(将1小时放电额定容量的电流值作为1C,所述额定容量取决于1小时率(one-hour-rate)的放电容量,下同)进行5个循环的初期充放电。将此时的第5个循环的0.2C放电容量作为初期容量。For a battery that has not been charged and discharged, at 25°C and a voltage range of 4.2V to 3.0V, with a current value of 0.2C (the current value of the rated capacity of 1 hour discharge is taken as 1C, and the rated capacity depends on 1 hour Rate (one-hour-rate) discharge capacity, the same below) for 5 cycles of initial charge and discharge. The 0.2C discharge capacity at the fifth cycle at this time was taken as the initial capacity.
(输出功率测定)(Output power measurement)
在25℃的环境下,通过0.2C的恒定电流进行150分钟充电,分别以0.1C、0.3C、1.0C、3.0C、10.0C放电10秒钟,测定第10秒的电压。将电流-电压直线和下限电压(3V)围成的三角形的面积作为输出功率(W)。In an environment of 25°C, charge was performed for 150 minutes with a constant current of 0.2C, and discharge was performed at 0.1C, 0.3C, 1.0C, 3.0C, and 10.0C for 10 seconds, respectively, and the voltage at the 10th second was measured. The area of the triangle surrounded by the current-voltage line and the lower limit voltage (3V) is taken as the output power (W).
电解液[4]实施例1Electrolyte [4] Embodiment 1
在干燥氩氛围下,向碳酸亚乙酯(EC:闪点143℃)、γ-丁内酯(GBL:闪点101℃)和碳酸二乙酯(DEC:闪点25℃)的混合物(容量比2∶7∶1)中加入0.3mol/L的LiPF6和0.7mol/L的LiBF4,并进行溶解,使该混合溶液中含有1质量%的碳酸亚乙烯酯、0.3质量%的六甲基环三硅氧烷,制备非水电解液。该非水电解液的闪点为61℃。使用该非水电解液用上述方法制作方形电池,并测定输出功率。结果如电解液[4]表1所示。Under a dry argon atmosphere, to a mixture (capacity 0.3 mol/L of LiPF 6 and 0.7 mol/L of LiBF 4 were added in a ratio of 2:7:1) and dissolved so that the mixed solution contained 1% by mass of vinylene carbonate, 0.3% by mass of hexamethanone Based cyclotrisiloxane, preparation of non-aqueous electrolyte. The flash point of this non-aqueous electrolytic solution was 61°C. Using this non-aqueous electrolytic solution, a rectangular battery was produced by the method described above, and the output was measured. The results are shown in Table 1 of the electrolyte [4].
电解液[4]实施例2Electrolyte [4] Embodiment 2
除了使用苯基二甲基氟硅烷来替代六甲基环三硅氧烷制成非水电解液以外,与电解液[4]实施例1同样地制作方形电池,并测定输出功率。结果如电解液[4]表1所示。Except for using phenyldimethylfluorosilane instead of hexamethylcyclotrisiloxane to prepare a nonaqueous electrolytic solution, a square battery was produced in the same manner as in Example 1 of the electrolytic solution [4], and the output power was measured. The results are shown in Table 1 of the electrolyte [4].
电解液[4]实施例3Electrolyte [4] Example 3
除了使用氟磺酸甲酯来替代六甲基环三硅氧烷制成非水电解液以外,与电解液[4]实施例1同样地制作方形电池,并测定输出功率。结果如电解液[4]表1所示。Except using methyl fluorosulfonate instead of hexamethylcyclotrisiloxane to prepare a non-aqueous electrolytic solution, a square battery was produced in the same manner as in Example 1 of the electrolytic solution [4], and the output power was measured. The results are shown in Table 1 of the electrolyte [4].
电解液[4]实施例4Electrolyte [4] Embodiment 4
除了使用根据Inorganic Nuclear Chemistry Letters(1969),5(7)的第581页~第582页中记载的方法制备的二氟磷酸锂来替代六甲基环三硅氧烷制成非水电解液以外,与电解液[4]实施例1同样地制作方形电池,并测定输出功率。结果如电解液[4]表1所示。In addition to using lithium difluorophosphate prepared according to the method described on pages 581 to 582 of Inorganic Nuclear Chemistry Letters (1969), 5(7) to replace hexamethylcyclotrisiloxane to make a non-aqueous electrolyte , A square battery was produced in the same manner as in Example 1 of the electrolyte solution [4], and the output power was measured. The results are shown in Table 1 of the electrolyte [4].
电解液[4]比较例1Electrolyte [4] Comparative Example 1
除了使非水电解液不含有六甲基环三硅氧烷以外,与电解液[4]实施例1同样地制作方形电池,并测定输出功率。结果如电解液[4]表1所示。Except that the non-aqueous electrolyte solution did not contain hexamethylcyclotrisiloxane, a square battery was produced in the same manner as in the electrolyte solution [4] Example 1, and the output was measured. The results are shown in Table 1 of the electrolyte [4].
电解液[4]<二次电池的制造-2(圆筒电池的制作)>Electrolyte solution [4] <Manufacture of secondary battery-2 (manufacture of cylindrical battery)>
[正极的制造][production of positive electrode]
在N-甲基吡咯烷酮溶剂中混合90质量%作为正极活性物质的钴酸锂(LiCoO2)、5质量%的作为导电材料的乙炔黑和5质量%作为粘合剂的聚偏氟乙烯(PVdF),制成浆料。将得到的浆料涂布在厚度20μm的铝箔两面,并干燥,用压制机压延成厚度80μm,再将其切成宽52mm、长830mm大小,作为正极。其中,表面背面都在长度方向上设置50mm的未涂布部分,活性物质层的长度为780mm。Mix 90% by mass of lithium cobaltate (LiCoO 2 ) as a positive electrode active material, 5% by mass of acetylene black as a conductive material, and 5% by mass of polyvinylidene fluoride (PVdF ) to make slurry. The obtained slurry was coated on both sides of an aluminum foil with a thickness of 20 μm, dried, and rolled to a thickness of 80 μm by a press machine, and then cut into a size of 52 mm in width and 830 mm in length as a positive electrode. Among them, an uncoated portion of 50 mm was provided in the longitudinal direction on both the front and back sides, and the length of the active material layer was 780 mm.
[负极的制造][production of negative electrode]
将2kg人造石墨粉末KS-44(timcal公司制造,商品名)和1kg石油类沥青混合,将得到的浆状的混合物在间歇式加热炉中于非活性氛围下升温2小时直至1100℃,在该温度下保持2小时。2kg of artificial graphite powder KS-44 (manufactured by timcal company, trade name) and 1kg of petroleum pitch were mixed, and the obtained slurry mixture was heated up to 1100° C. for 2 hours in a batch heating furnace under an inert atmosphere. Keep at temperature for 2 hours.
将其粉碎,通过振动式筛将粒径调整为18~22μm,最终得到以7质量%的无定形碳覆盖石墨表面的“无定形包覆石墨类碳质物质”。将本“无定形包覆石墨类碳质物质”作为负极活性物质,向98重量份“无定形包覆石墨类碳质物质”中添加100重量份作为增稠剂的羧甲基纤维素钠的水性分散液(羧甲基纤维素钠的浓度为1质量%)、2重量份作为粘合剂的苯乙烯-丁二烯橡胶的水性分散液(苯乙烯-丁二烯橡胶的浓度为50质量%),用分散器进行混合,制成浆料。将得到的浆料涂布在作为负极集电体的厚度18μm的铜箔的两面,并干燥,用压制机压延成厚度为85μm,再将其切成宽56mm、长850mm的大小,作为负极。其中,表面背面都在长度方向上设置30mm的未涂布部分。This was pulverized, and the particle size was adjusted to 18 to 22 μm with a vibrating sieve to finally obtain an "amorphous-coated graphite-like carbonaceous material" in which the graphite surface was covered with 7% by mass of amorphous carbon. Using this "amorphous coated graphite-like carbonaceous material" as the negative active material, adding 100 parts by weight of sodium carboxymethylcellulose as a thickener to 98 parts by weight of the "amorphous coated graphite-based carbonaceous material" Aqueous dispersion (the concentration of sodium carboxymethylcellulose is 1% by mass), 2 parts by weight of an aqueous dispersion of styrene-butadiene rubber as a binder (the concentration of styrene-butadiene rubber is 50 mass %) %), mixed with a disperser to make a slurry. The obtained slurry was coated on both sides of a copper foil with a thickness of 18 μm as a negative electrode current collector, dried, rolled to a thickness of 85 μm with a press, and then cut into a size of 56 mm in width and 850 mm in length as the negative electrode. Among them, an uncoated portion of 30 mm was provided in the longitudinal direction on both the front and back sides.
[电池的组装][assembling the battery]
将正极和负极与聚乙烯制的隔板一起重叠卷绕并使得正极与负极不直接接触,制成电极体。使正极和负极的端子露出外部地收纳在电池罐中。然后,向其中注入5mL后述的非水电解液后,进行铆接成型,制造18650型圆筒电池。二次电池的1个电池外壳中收纳的电池要素所具有的电容,即该电池的额定放电容量约为0.7安培小时(Ah),用10kHz交流法测定的直流电阻成分约为35毫欧(mΩ)。The positive electrode and the negative electrode were overlapped and wound together with a polyethylene separator so that the positive electrode and the negative electrode were not in direct contact to form an electrode body. The positive and negative terminals are housed in the battery can with the terminals exposed to the outside. Then, 5 mL of a non-aqueous electrolytic solution described later was injected thereinto, and then caulking was performed to manufacture a 18650-type cylindrical battery. The capacitance of the battery element contained in one battery case of the secondary battery, that is, the rated discharge capacity of the battery is about 0.7 ampere hours (Ah), and the DC resistance component measured by the 10kHz alternating current method is about 35 milliohms (mΩ ).
电解液[4]实施例5Electrolyte [4] Embodiment 5
使用电解液[4]实施例1中使用的非水电解液作为非水电解液,制作圆筒电池,并测定输出功率。结果如电解液[4]表1所示。The non-aqueous electrolytic solution used in the electrolytic solution [4] Example 1 was used as the non-aqueous electrolytic solution to manufacture a cylindrical battery, and the output power was measured. The results are shown in Table 1 of the electrolyte [4].
电解液[4]比较例2Electrolyte [4] Comparative Example 2
除了使用电解液[4]比较例1中使用的非水电解液作为非水电解液以外,与电解液[4]实施例5同样地制作圆筒电池,并测定输出功率。结果如电解液[4]表1所示。Except for using the non-aqueous electrolyte solution used in the electrolyte solution [4] Comparative Example 1 as the non-aqueous electrolyte solution, a cylindrical battery was produced in the same manner as in the electrolyte solution [4] Example 5, and the output was measured. The results are shown in Table 1 of the electrolyte [4].
电解液[4]表1Electrolyte [4] Table 1
[表57][Table 57]
由电解液[4]表1可知,若对每个方形电池(电解液[4]实施例1~4,电解液[4]比较例1)、圆筒电池(电解液[4]实施例5,电解液[4]比较例2)进行比较,则可知本实施例的锂二次电池的输出功率得到改善。From the electrolyte [4] Table 1, it can be seen that if each square battery (electrolyte [4] embodiment 1 to 4, electrolyte [4] comparative example 1), cylindrical battery (electrolyte [4] embodiment 5 , Electrolyte solution [4] Comparative Example 2) for comparison, it can be seen that the output power of the lithium secondary battery of this embodiment is improved.
如上所述,本实施例与本比较例的方形电池的额定放电容量为3安培小时(Ah)以上,直流电阻成分为10毫欧(mΩ)以下。另一方面,本实施例与本比较例的圆筒电池的额定放电容量小于3安培小时(Ah),直流电阻成分大于10毫欧(mΩ)。即,本实施例、本比较例的方形电池与圆筒电池相比,电阻小,电容大。而且,电解液[4]实施例1相对于电解液[4]比较例1的输出功率提高程度,比电解液[4]实施例5相对于电解液[4]比较例2的输出功率提高程度大,在电容大的二次电池或直流电阻小的二次电池中,本发明的效果更大。As described above, the rated discharge capacity of the prismatic batteries of this example and this comparative example is 3 ampere hours (Ah) or more, and the DC resistance component is 10 milliohms (mΩ) or less. On the other hand, the rated discharge capacity of the cylindrical batteries of this embodiment and this comparative example is less than 3 ampere hours (Ah), and the DC resistance component is greater than 10 milliohms (mΩ). That is, the prismatic batteries of this example and this comparative example have lower resistance and higher capacitance than cylindrical batteries. Moreover, the output power improvement degree of the electrolyte solution [4] Example 1 relative to the electrolyte solution [4] Comparative Example 1 is higher than the output power improvement degree of the electrolyte solution [4] Example 5 relative to the electrolyte solution [4] Comparative Example 2 Large, the effect of the present invention is greater in a secondary battery with a large capacitance or a secondary battery with a small DC resistance.
电解液[5]<二次电池的制作>Electrolyte [5] <Production of secondary battery>
[正极的制作][making of positive electrode]
在N-甲基吡咯烷酮溶剂中混合90质量%作为正极活性物质的钴酸锂(LiCoO2)、5质量%的作为导电材料的乙炔黑和5质量%作为粘合剂的聚偏氟乙烯(PVdF),制成浆料。将得到的浆料涂布在厚度15μm的铝箔两面,并干燥,用压制机压延成厚度80μm,再将其切成具有宽100mm、长100mm大小的活性物质层和宽30mm的未涂布部分的形状,作为正极。Mix 90% by mass of lithium cobaltate (LiCoO 2 ) as a positive electrode active material, 5% by mass of acetylene black as a conductive material, and 5% by mass of polyvinylidene fluoride (PVdF ) to make slurry. The obtained slurry was coated on both sides of an aluminum foil with a thickness of 15 μm, dried, rolled to a thickness of 80 μm with a press, and cut into an active material layer having a width of 100 mm and a length of 100 mm and an uncoated portion of a width of 30 mm. shape, as the positive electrode.
[负极的制作][Production of Negative Electrode]
在98重量份人造石墨粉末KS-44(timcal公司制造,商品名)中添加100重量份作为增稠剂的羧甲基纤维素钠的水性分散液(羧甲基纤维素钠的浓度为1质量%)、2重量份作为粘合剂的苯乙烯-丁二烯橡胶的水性分散液(苯乙烯-丁二烯橡胶的浓度为50质量%),用分散器进行混合,制成浆料。将得到的浆料涂布在厚度10μm的铜箔两面,并干燥,用压制机压延成厚度为75μm,再将其切成具有宽104mm、长104mm大小的活性物质层和宽30mm的未涂布部分的形状,作为负极。In 98 parts by weight of artificial graphite powder KS-44 (manufactured by timcal company, trade name), add 100 parts by weight of an aqueous dispersion of sodium carboxymethyl cellulose as a thickener (the concentration of sodium carboxymethyl cellulose is 1 mass %), 2 parts by weight of an aqueous dispersion of styrene-butadiene rubber as a binder (the concentration of styrene-butadiene rubber is 50% by mass), and mixed with a disperser to prepare a slurry. The obtained slurry was coated on both sides of a copper foil with a thickness of 10 μm, dried, rolled to a thickness of 75 μm with a press, and then cut into an active material layer with a width of 104 mm and a length of 104 mm and an uncoated layer with a width of 30 mm. The shape of the part, as the negative pole.
[电池的组装][assembling the battery]
将32片正极和33片负极交替配置,各电极之间夹入多孔聚乙烯片隔板(厚度为25μm),进行叠层。此时,使正极活性物质面与负极活性物质面相对,并且不会露出负极活性物质面之外。将该正极和负极各自的未涂布部分焊接,制作集电片,将电极组密封在带有排出阀的电池罐(外部尺寸:120×110×10mm)中。然后,在装有电极组的电池罐中注入20mL后述的非水电解液,使电极充分浸透,密封而制作方形电池。二次电池的1个电池外壳中收纳的电池要素所具有的电容,即该电池的额定放电容量约为6Ah,以10kHz交流法测定的直流电阻成分约为5毫欧(mΩ)。32 positive electrodes and 33 negative electrodes were arranged alternately, and a porous polyethylene sheet separator (thickness: 25 μm) was sandwiched between the electrodes for lamination. At this time, the surface of the positive electrode active material faces the surface of the negative electrode active material without protruding from the surface of the negative electrode active material. The respective uncoated portions of the positive and negative electrodes were welded to produce current collector sheets, and the electrode group was sealed in a battery can (outer dimensions: 120×110×10 mm) with a discharge valve. Then, 20 mL of a non-aqueous electrolytic solution described later was poured into the battery can containing the electrode group, the electrodes were sufficiently permeated, and sealed to produce a square battery. The capacitance of the battery element contained in one battery case of the secondary battery, that is, the rated discharge capacity of the battery is about 6Ah, and the DC resistance component measured by the 10kHz AC method is about 5 milliohms (mΩ).
[电池的评价][evaluation of the battery]
(容量测定)(capacity measurement)
对未经充放电循环的电池,在25℃、4.2V~3.0V的电压范围下,以0.2C的电流值(将1小时放电额定容量的电流值作为1C,所述额定容量取决于1小时率(one-hour-rate)的放电容量,下同)进行5个循环的初期充放电。将此时的第5个循环的0.2C放电容量作为初期容量。For a battery that has not been charged and discharged, at 25°C and a voltage range of 4.2V to 3.0V, with a current value of 0.2C (the current value of the rated capacity of 1 hour discharge is taken as 1C, and the rated capacity depends on 1 hour Rate (one-hour-rate) discharge capacity, the same below) for 5 cycles of initial charge and discharge. The 0.2C discharge capacity at the fifth cycle at this time was taken as the initial capacity.
(输出功率测定)(Output power measurement)
在25℃的环境下,通过0.2C的恒定电流进行150分钟充电,分别以0.1C、0.3C、1.0C、3.0C、10.0C放电10秒钟,测定第10秒的电压。将电流-电压直线和下限电压(3V)围成的三角形的面积作为输出功率(W)。In an environment of 25°C, charge was performed for 150 minutes with a constant current of 0.2C, and discharge was performed at 0.1C, 0.3C, 1.0C, 3.0C, and 10.0C for 10 seconds, respectively, and the voltage at the 10th second was measured. The area of the triangle surrounded by the current-voltage line and the lower limit voltage (3V) is taken as the output power (W).
(循环试验)(cycle test)
在被视为锂二次电池的实际使用上限温度的60℃的高温环境下进行循环试验。以2C的恒定电流恒定电压法充电至充电上限电压4.2V后,以2C的恒定电流放电至放电终止电压3.0V,将此充放电循环作为1个循环,重复该循环直到500次。The cycle test was performed in a high-temperature environment of 60° C., which is regarded as the practical upper limit temperature of the lithium secondary battery. After charging to the charging upper limit voltage of 4.2V by the constant current and constant voltage method of 2C, discharge to the end-of-discharge voltage of 3.0V by the constant current of 2C. This charge-discharge cycle is regarded as one cycle, and the cycle is repeated until 500 times.
(保存试验)(preservation test)
在60℃的高温环境下进行保存试验。将事先在25℃的环境下用恒定电流恒定电压法充电至充电上限电压4.2V的电池在60℃下保存1个月。The storage test was carried out in a high-temperature environment of 60°C. The battery previously charged to a charging upper limit voltage of 4.2 V by a constant current constant voltage method in an environment of 25° C. was stored at 60° C. for one month.
对容量测定后的电池、循环试验后的电池、保存试验后的电池进行输出功率测定,分别作为初期输出功率、循环后输出功率、保存后输出功率。The output power of the battery after the capacity measurement, the battery after the cycle test, and the battery after the storage test is measured, and they are respectively used as the initial output power, the output power after cycle, and the output power after storage.
电解液[5]实施例1Electrolyte [5] Embodiment 1
在干燥氩氛围下,向碳酸亚乙酯(EC)、碳酸甲乙酯(EMC)和碳酸二甲酯(DMC)的混合物(体积比2∶5∶3)中加入1mol/L的LiPF6和0.01mol/L的LiN(CF3SO2)2,并进行溶解,使该混合溶液中含有0.3质量%的六甲基环三硅氧烷,制备非水电解液。使用该非水电解液用上述方法制作电池,并测定初期输出功率、循环后输出功率和保存后输出功率。结果如电解液[5]表1所示。Under dry argon atmosphere, add 1 mol/L LiPF 6 and 0.01 mol/L of LiN(CF 3 SO 2 ) 2 was dissolved, and the mixed solution contained 0.3% by mass of hexamethylcyclotrisiloxane to prepare a non-aqueous electrolytic solution. Using this non-aqueous electrolytic solution, a battery was produced by the above-mentioned method, and the initial output power, the output power after cycling, and the output power after storage were measured. The results are shown in Table 1 of the electrolyte [5].
电解液[5]实施例2Electrolyte [5] Embodiment 2
除了使用苯基二甲基氟硅烷来替代六甲基环三硅氧烷制成非水电解液以外,与电解液[5]实施例1同样地制作电池,并测定初期输出功率、循环后输出功率和保存后输出功率。结果如电解液[5]表1所示。Except using phenyldimethylfluorosilane instead of hexamethylcyclotrisiloxane to make a non-aqueous electrolyte, a battery was made in the same manner as in Example 1 of the electrolyte [5], and the initial output power and output after cycling were measured. Power and output power after saving. The results are shown in Table 1 of the electrolyte [5].
电解液[5]实施例3Electrolyte [5] Example 3
除了使用氟磺酸甲酯来替代六甲基环三硅氧烷制成非水电解液以外,与电解液[5]实施例1同样地制作电池,并测定初期输出功率、循环后输出功率和保存后输出功率。结果如电解液[5]表1所示。Except using methyl fluorosulfonate to replace hexamethylcyclotrisiloxane to make non-aqueous electrolyte, make battery in the same way as electrolyte [5] embodiment 1, and measure initial output power, output power after cycle and Output power after saving. The results are shown in Table 1 of the electrolyte [5].
电解液[5]实施例4Electrolyte [5] Embodiment 4
除了使用根据Inorganic Nuclear Chemistry Letters(1969),5(7)的第581页~第582页中记载的方法制备的二氟磷酸锂来替代六甲基环三硅氧烷制成非水电解液以外,与电解液[5]实施例1同样地制作电池,并测定初期输出功率、循环后输出功率和保存后输出功率。结果如电解液[5]表1所示。In addition to using lithium difluorophosphate prepared according to the method described on pages 581 to 582 of Inorganic Nuclear Chemistry Letters (1969), 5(7) to replace hexamethylcyclotrisiloxane to make a non-aqueous electrolyte , The battery was produced in the same manner as the electrolyte solution [5] Example 1, and the initial output power, output power after cycle and output power after storage were measured. The results are shown in Table 1 of the electrolyte [5].
电解液[5]实施例5Electrolyte [5] Embodiment 5
除了使用二(草酸根合)硼酸锂(LiBOB)来替代LiN(CF3SO2)2制成非水电解液以外,与电解液[5]实施例1同样地制作电池,并测定初期输出功率、循环后输出功率和保存后输出功率。结果如电解液[5]表1所示。Except for using bis(oxalato)lithium borate (LiBOB) instead of LiN(CF 3 SO 2 ) 2 to make a non-aqueous electrolyte, a battery was produced in the same manner as in Example 1 of the electrolyte [5], and the initial output power was measured , output power after cycling and output power after storage. The results are shown in Table 1 of the electrolyte [5].
电解液[5]实施例6Electrolyte [5] Embodiment 6
除了使用二(草酸根合)硼酸锂(LiBOB)来替代LiN(CF3SO2)2制成非水电解液以外,与电解液[5]实施例4同样地制作电池,并测定初期输出功率、循环后输出功率和保存后输出功率。结果如电解液[5]表1所示。Except for using bis(oxalato)lithium borate (LiBOB) instead of LiN(CF 3 SO 2 ) 2 to make a non-aqueous electrolyte, a battery was produced in the same manner as in Example 4 of the electrolyte [5], and the initial output power was measured , output power after cycling and output power after storage. The results are shown in Table 1 of the electrolyte [5].
电解液[5]比较例1Electrolyte [5] Comparative Example 1
除了非水电解液中未溶解LiN(CF3SO2)2以外,与实施例1同样地制作电池,并测定初期输出功率、循环后输出功率和保存后输出功率。结果如电解液[5]表1所示。A battery was produced in the same manner as in Example 1 except that LiN(CF 3 SO 2 ) 2 was not dissolved in the non-aqueous electrolyte solution, and the initial output, output after cycles, and output after storage were measured. The results are shown in Table 1 of the electrolyte [5].
电解液[5]比较例2Electrolyte [5] Comparative Example 2
除了非水电解液中不含有六甲基环三硅氧烷以外,与电解液[5]实施例1同样地制作电池,并测定初期输出功率、循环后输出功率和保存后输出功率。结果如电解液[5]表1所示。Except not containing hexamethylcyclotrisiloxane in the nonaqueous electrolytic solution, a battery was produced in the same manner as in the electrolytic solution [5] Example 1, and the initial output power, output power after cycle, and output power after storage were measured. The results are shown in Table 1 of the electrolyte [5].
电解液[5]比较例3Electrolyte [5] Comparative Example 3
除了非水电解液中不含有LiN(CF3SO2)2和六甲基环三硅氧烷以外,与电解液[5]实施例1同样地制作电池,并测定初期输出功率、循环后输出功率和保存后输出功率。结果如电解液[5]表1所示。Except that LiN(CF 3 SO 2 ) 2 and hexamethylcyclotrisiloxane were not contained in the non-aqueous electrolyte solution, a battery was produced in the same manner as in Example 1 of the electrolyte solution [5], and the initial output power and output after cycling were measured. Power and output power after saving. The results are shown in Table 1 of the electrolyte [5].
电解液[5]实施例7Electrolyte [5] Embodiment 7
<二次电池的制造-2><Manufacture of secondary battery-2>
[正极的制造][production of positive electrode]
在N-甲基吡咯烷酮溶剂中混合90质量%作为正极活性物质的钴酸锂(LiCoO2)、5质量%的作为导电材料的乙炔黑和5质量%作为粘合剂的聚偏氟乙烯(PVdF),制成浆料。将得到的浆料涂布在厚度20μm的铝箔两面,并干燥,用压制机压延成厚度80μm,再将其切成宽52mm、长830mm大小,作为正极。其中,表面背面都在长度方向上设置50mm的未涂布部分,活性物质层的长度为780mm。Mix 90% by mass of lithium cobaltate (LiCoO 2 ) as a positive electrode active material, 5% by mass of acetylene black as a conductive material, and 5% by mass of polyvinylidene fluoride (PVdF ) to make slurry. The obtained slurry was coated on both sides of an aluminum foil with a thickness of 20 μm, dried, and rolled to a thickness of 80 μm by a press machine, and then cut into a size of 52 mm in width and 830 mm in length as a positive electrode. Among them, an uncoated portion of 50 mm was provided in the longitudinal direction on both the front and back sides, and the length of the active material layer was 780 mm.
[负极的制造][production of negative electrode]
在98重量份人造石墨粉末KS-44(timcal公司制造,商品名)中添加100重量份作为增稠剂的羧甲基纤维素钠的水性分散液(羧甲基纤维素钠的浓度为1质量%)、2重量份作为粘合剂的苯乙烯-丁二烯橡胶的水性分散液(苯乙烯-丁二烯橡胶的浓度为50质量%),用分散器进行混合,制成浆料。将得到的浆料均匀涂布在作为负极集电体的厚度18μm的铜箔的两面,干燥后,用压制机压延成厚度为85μm,再将其切成宽56mm、长850mm的大小,作为负极。其中,表面背面都在长度方向上设置30mm的未涂布部分。In 98 parts by weight of artificial graphite powder KS-44 (manufactured by timcal company, trade name), add 100 parts by weight of an aqueous dispersion of sodium carboxymethyl cellulose as a thickener (the concentration of sodium carboxymethyl cellulose is 1 mass %), 2 parts by weight of an aqueous dispersion of styrene-butadiene rubber as a binder (the concentration of styrene-butadiene rubber is 50% by mass), and mixed with a disperser to prepare a slurry. The obtained slurry was uniformly coated on both sides of a copper foil with a thickness of 18 μm as the negative electrode current collector, and after drying, it was rolled to a thickness of 85 μm by a press, and then cut into a size of 56 mm wide and 850 mm long, as the negative electrode . Among them, an uncoated portion of 30 mm was provided in the longitudinal direction on both the front and back sides.
[电池的组装][assembling the battery]
将正极和负极与聚乙烯制的隔板一起重叠卷绕并使得正极与负极不直接接触,制成电极体。使正极和负极的端子露出外部地收纳在电池罐中。然后,向其中注入5mL后述的非水电解液后,进行铆接成型,制造18650型圆筒电池。二次电池的1个电池外装收纳的电池要素具有的电容,即二次电池的额定放电容量约为0.7安培小时(Ah),用10kHz交流法测定的直流电阻成分约为35毫欧(mΩ)。The positive electrode and the negative electrode were overlapped and wound together with a polyethylene separator so that the positive electrode and the negative electrode were not in direct contact to form an electrode body. The positive and negative terminals are housed in the battery can with the terminals exposed to the outside. Then, 5 mL of a non-aqueous electrolytic solution described later was injected thereinto, and then caulking was performed to manufacture a 18650-type cylindrical battery. One battery of a secondary battery has the capacitance of the battery element contained in the exterior, that is, the rated discharge capacity of the secondary battery is about 0.7 ampere hours (Ah), and the DC resistance component measured by the 10 kHz alternating current method is about 35 milliohms (mΩ). .
使用电解液[5]实施例1中使用的非水电解液作为非水电解液来制造圆筒电池,并测定初期输出功率、循环后输出功率和保存后输出功率。结果如电解液[5]表1所示。The non-aqueous electrolytic solution used in the electrolytic solution [5] Example 1 was used as the non-aqueous electrolytic solution to manufacture a cylindrical battery, and the initial output power, output power after cycle and output power after storage were measured. The results are shown in Table 1 of the electrolyte [5].
电解液[5]实施例8Electrolyte [5] Embodiment 8
除了使用电解液[5]实施例2中使用的非水电解液作为非水电解液以外,与电解液[5]实施例7同样地制作圆筒电池,并测定初期输出功率、循环后输出功率和保存后输出功率。结果如电解液[5]表1所示。Except using the non-aqueous electrolytic solution used in the electrolytic solution [5] Example 2 as the non-aqueous electrolytic solution, a cylindrical battery was produced in the same manner as the electrolytic solution [5] Example 7, and the initial output power and the output power after cycling were measured. and output power after saving. The results are shown in Table 1 of the electrolyte [5].
电解液[5]实施例9Electrolyte [5] Embodiment 9
除了使用电解液[5]实施例3中使用的非水电解液作为非水电解液以外,与电解液[5]实施例7同样地制作圆筒电池,并测定初期输出功率、循环后输出功率和保存后输出功率。结果如电解液[5]表1所示。Except using the non-aqueous electrolytic solution used in the electrolytic solution [5] Example 3 as the non-aqueous electrolytic solution, a cylindrical battery was produced in the same manner as the electrolytic solution [5] Example 7, and the initial output power and the output power after the cycle were measured. and output power after saving. The results are shown in Table 1 of the electrolyte [5].
电解液[5]实施例10Electrolyte [5] Embodiment 10
除了使用电解液[5]实施例4中使用的非水电解液作为非水电解液以外,与电解液[5]实施例7同样地制作圆筒电池,并测定初期输出功率、循环后输出功率和保存后输出功率。结果如电解液[5]表1所示。Except using the non-aqueous electrolytic solution used in the electrolytic solution [5] Example 4 as the non-aqueous electrolytic solution, a cylindrical battery was produced in the same manner as the electrolytic solution [5] Example 7, and the initial output power and the output power after the cycle were measured. and output power after saving. The results are shown in Table 1 of the electrolyte [5].
电解液[5]实施例11Electrolyte [5] Embodiment 11
除了使用电解液[5]实施例5中使用的非水电解液作为非水电解液以外,与电解液[5]实施例7同样地制作圆筒电池,并测定初期输出功率、循环后输出功率和保存后输出功率。结果如电解液[5]表1所示。Except using the non-aqueous electrolytic solution used in the electrolytic solution [5] Example 5 as the non-aqueous electrolytic solution, a cylindrical battery was produced in the same manner as the electrolytic solution [5] Example 7, and the initial output power and the output power after the cycle were measured. and output power after saving. The results are shown in Table 1 of the electrolyte [5].
电解液[5]实施例12Electrolyte [5] Example 12
除了使用电解液[5]实施例6中使用的非水电解液作为非水电解液以外,与电解液[5]实施例7同样地制作圆筒电池,并测定初期输出功率、循环后输出功率和保存后输出功率。结果如电解液[5]表1所示。Except using the non-aqueous electrolytic solution used in the electrolytic solution [5] Example 6 as the non-aqueous electrolytic solution, a cylindrical battery was produced in the same manner as the electrolytic solution [5] Example 7, and the initial output power and the output power after the cycle were measured. and output power after saving. The results are shown in Table 1 of the electrolyte [5].
电解液[5]比较例4Electrolyte [5] Comparative Example 4
除了使用电解液[5]比较例1中使用的非水电解液作为非水电解液以外,与电解液[5]实施例7同样地制作圆筒电池,并测定初期输出功率、循环后输出功率和保存后输出功率。结果如电解液[5]表1所示。Except using the non-aqueous electrolytic solution used in the electrolytic solution [5] Comparative Example 1 as the non-aqueous electrolytic solution, a cylindrical battery was produced in the same manner as the electrolytic solution [5] Example 7, and the initial output power and the output power after cycling were measured. and output power after saving. The results are shown in Table 1 of the electrolyte [5].
电解液[5]比较例5Electrolyte [5] Comparative Example 5
除了使用电解液[5]比较例2中使用的非水电解液作为非水电解液以外,与电解液[5]实施例7同样地制作圆筒电池,并测定初期输出功率、循环后输出功率和保存后输出功率。结果如电解液[5]表1所示。Except using the non-aqueous electrolyte solution used in the electrolyte solution [5] Comparative Example 2 as the non-aqueous electrolyte solution, a cylindrical battery was produced in the same manner as the electrolyte solution [5] Example 7, and the initial output power and the output power after cycling were measured. and output power after saving. The results are shown in Table 1 of the electrolyte [5].
电解液[5]比较例6Electrolyte [5] Comparative Example 6
除了使用电解液[5]比较例3中使用的非水电解液作为非水电解液以外,与电解液[5]实施例7同样地制作圆筒电池,并测定初期输出功率、循环后输出功率和保存后输出功率。结果如电解液[5]表1所示。Except using the non-aqueous electrolytic solution used in the electrolytic solution [5] Comparative Example 3 as the non-aqueous electrolytic solution, a cylindrical battery was produced in the same manner as the electrolytic solution [5] Example 7, and the initial output power and the output power after cycling were measured. and output power after saving. The results are shown in Table 1 of the electrolyte [5].
电解液[5]表1Electrolyte [5] Table 1
[表58][Table 58]
由电解液[5]表1可知,若对每个方形电池(电解液[5]实施例1~6,电解液[5]比较例1~3)、圆筒电池(电解液[5]实施例7~12,电解液[5]比较例4~6)进行比较,则可知在非水电解液中同时含有LiN(CnF2n+1SO2)2(式中,n为1~4的整数)和/或二(草酸根合)硼酸锂与特定化合物的电解液[5]实施例的锂二次电池,与不含这些中任一种的电解液[5]比较例的锂二次电池相比,输出功率得到改善。From the electrolyte [5] Table 1, it can be seen that if each square battery (electrolyte [5] embodiment 1-6, electrolyte [5] comparative example 1-3), cylindrical battery (electrolyte [5] Examples 7-12, electrolyte [5] Comparative Examples 4-6) are compared, then it can be seen that LiN(C n F 2n+1 SO 2 ) 2 (where n is 1-4) is contained in the non-aqueous electrolyte Integer of ) and/or lithium bis(oxalato)borate and the electrolyte solution [5] of the specific compound The lithium secondary battery of the example, and the lithium secondary battery of the comparative example of the electrolyte solution [5] not containing any of these The output power is improved compared to the secondary battery.
如上所述,本实施例与本比较例的方形电池的额定放电容量为3安培小时(Ah)以上,直流电阻成分为10毫欧(mΩ)以下。另一方面,本实施例与本比较例的圆筒电池的额定放电容量小于3安培小时(Ah),直流电阻成分大于10毫欧(mΩ)。即,本实施例、本比较例的方形电池与圆筒电池相比,电阻小,电容大。而且,电解液[5]实施例1相对于电解液[5]比较例1的输出功率提高程度,比电解液[5]实施例7相对于电解液[5]比较例4的输出功率提高程度大。特别是在循环后的输出功率提高程度和保存后的输出功率提高程度中存在大的差异。由此可知,在电容大的二次电池或直流电阻小的二次电池中,本发明的效果更大。As described above, the rated discharge capacity of the prismatic batteries of this example and this comparative example is 3 ampere hours (Ah) or more, and the DC resistance component is 10 milliohms (mΩ) or less. On the other hand, the rated discharge capacity of the cylindrical batteries of this embodiment and this comparative example is less than 3 ampere hours (Ah), and the DC resistance component is greater than 10 milliohms (mΩ). That is, the prismatic batteries of this example and this comparative example have lower resistance and higher capacitance than cylindrical batteries. Moreover, the output power improvement degree of the electrolyte solution [5] Example 1 relative to the electrolyte solution [5] Comparative Example 1 is higher than the output power improvement degree of the electrolyte solution [5] Embodiment 7 relative to the electrolyte solution [5] Comparative Example 4 big. In particular, there is a large difference in the degree of output power increase after cycling and the degree of output power increase after storage. From this, it can be seen that the effect of the present invention is greater in a secondary battery with a large capacity or a secondary battery with a small DC resistance.
电解液[6]<非水溶剂的制备>Electrolyte solution [6] <Preparation of non-aqueous solvent>
用分子筛4A对市售的碳酸亚乙酯(EC)进行吸附处理(50℃,LHSV,1/小时)。另一方面,分别以回流比1、理论塔板数30塔板对碳酸二甲酯和碳酸甲乙酯充分进行精馏后,用分子筛4A进行吸附处理(25℃,LHSV,1/小时)。然后,以EC∶DMC∶EMC(体积比3∶3∶4)将它们混合,进一步用分子筛4A进行吸附处理(25℃,LHSV,1/小时),制备混合非水溶剂。此时,非水溶剂中,未检测出水或醇类。Commercially available ethylene carbonate (EC) was subjected to adsorption treatment (50° C., LHSV, 1/hour) with molecular sieve 4A. On the other hand, after fully rectifying dimethyl carbonate and ethyl methyl carbonate with a reflux ratio of 1 and a theoretical plate number of 30 plates, adsorption treatment was carried out with molecular sieve 4A (25° C., LHSV, 1/hour). Then, they were mixed with EC:DMC:EMC (volume ratio 3:3:4), and further subjected to adsorption treatment (25° C., LHSV, 1/hour) with molecular sieve 4A to prepare a mixed non-aqueous solvent. At this time, no water or alcohols were detected in the non-aqueous solvent.
电解液[6]<二次电池的制作>Electrolyte [6] <Production of secondary battery>
[正极的制作][making of positive electrode]
在N-甲基吡咯烷酮溶剂中混合90质量%作为正极活性物质的钴酸锂(LiCoO2)、5质量%的作为导电材料的乙炔黑和5质量%作为粘合剂的聚偏氟乙烯(PVdF),制成浆料。将得到的浆料涂布在厚度15μm的铝箔两面,并干燥,用压制机压延成厚度80μm,再将其切成具有宽100mm、长100mm大小的活性物质层和宽30mm的未涂布部分的形状,作为正极。Mix 90% by mass of lithium cobaltate (LiCoO 2 ) as a positive electrode active material, 5% by mass of acetylene black as a conductive material, and 5% by mass of polyvinylidene fluoride (PVdF ) to make slurry. The obtained slurry was coated on both sides of an aluminum foil with a thickness of 15 μm, dried, rolled to a thickness of 80 μm with a press, and cut into an active material layer having a width of 100 mm and a length of 100 mm and an uncoated portion of a width of 30 mm. shape, as the positive electrode.
[负极的制作][Production of Negative Electrode]
在98重量份人造石墨粉末KS-44(timcal公司制造,商品名)中添加100重量份作为增稠剂的羧甲基纤维素钠的水性分散液(羧甲基纤维素钠的浓度为1质量%)、2重量份作为粘合剂的苯乙烯-丁二烯橡胶的水性分散液(苯乙烯-丁二烯橡胶的浓度为50质量%),用分散器进行混合,制成浆料。将得到的浆料涂布在厚度10μm的铜箔两面,并干燥,用压制机压延成厚度为75μm,再将其切成具有宽104mm、长104mm大小的活性物质层和宽30mm的未涂布部分的形状,作为负极。In 98 parts by weight of artificial graphite powder KS-44 (manufactured by timcal company, trade name), add 100 parts by weight of an aqueous dispersion of sodium carboxymethyl cellulose as a thickener (the concentration of sodium carboxymethyl cellulose is 1 mass %), 2 parts by weight of an aqueous dispersion of styrene-butadiene rubber as a binder (the concentration of styrene-butadiene rubber is 50% by mass), and mixed with a disperser to prepare a slurry. The obtained slurry was coated on both sides of a copper foil with a thickness of 10 μm, dried, rolled to a thickness of 75 μm with a press, and then cut into an active material layer with a width of 104 mm and a length of 104 mm and an uncoated layer with a width of 30 mm. The shape of the part, as the negative pole.
[电池的组装][assembling the battery]
将32片正极和33片负极交替配置,各电极之间夹入多孔聚乙烯片隔板(厚度为25μm),进行叠层。此时,使正极活性物质面与负极活性物质面相对,并且不会露出负极活性物质面之外。将该正极和负极各自的未涂布部分焊接,制作集电片,将电极组密封在带有排气阀的电池罐(外部尺寸:120×110×10mm)中。然后,在装有电极组的电池罐中注入20mL非水电解液,使电极充分浸透,密封而制作方形电池。该电池的额定放电容量为高容量,约为6Ah,以10kHz交流法测定的直流电阻成分约为5毫欧。32 positive electrodes and 33 negative electrodes were arranged alternately, and a porous polyethylene sheet separator (thickness: 25 μm) was sandwiched between the electrodes for lamination. At this time, the surface of the positive electrode active material faces the surface of the negative electrode active material without protruding from the surface of the negative electrode active material. The respective uncoated parts of the positive and negative electrodes were welded to produce current collector sheets, and the electrode group was sealed in a battery can (outer dimensions: 120×110×10 mm) with a vent valve. Then, 20 mL of non-aqueous electrolytic solution was poured into the battery can containing the electrode group, the electrodes were fully soaked, and sealed to produce a square battery. The rated discharge capacity of this battery is a high capacity of about 6Ah, and the DC resistance component measured by the 10kHz AC method is about 5 milliohms.
[电池的评价][evaluation of the battery]
(容量测定)(capacity measurement)
对未经充放电循环的新电池,在25℃、4.2V~3.0V的电压范围下,以0.2C的电流值(将1小时放电额定容量的电流值作为1C,所述额定容量取决于1小时率(one-hour-rate)的放电容量,下同)进行5个循环的初期充放电。将此时的第5个循环的0.2C放电容量作为初期容量。For a new battery that has not been charged and discharged, at 25°C and a voltage range of 4.2V to 3.0V, with a current value of 0.2C (the current value of the rated capacity of 1 hour discharge is taken as 1C, and the rated capacity depends on 1 One-hour-rate (one-hour-rate) discharge capacity, the same below) was performed for 5 cycles of initial charge and discharge. The 0.2C discharge capacity at the fifth cycle at this time was taken as the initial capacity.
(输出功率测定)(Output power measurement)
在25℃的环境下,通过0.2C的恒定电流进行150分钟充电,分别以0.1C、0.3C、1.0C、3.0C、10.0C放电10秒钟,测定第10秒的电压。将电流-电压直线和下限电压(3V)围成的三角形的面积作为输出功率(W)。In an environment of 25°C, charge was performed for 150 minutes with a constant current of 0.2C, and discharge was performed at 0.1C, 0.3C, 1.0C, 3.0C, and 10.0C for 10 seconds, respectively, and the voltage at the 10th second was measured. The area of the triangle surrounded by the current-voltage line and the lower limit voltage (3V) is taken as the output power (W).
(循环试验)(cycle test)
在被看作锂二次电池的实际使用上限温度的60℃的高温环境下进行循环试验。以2C的恒定电流恒定电压法充电至充电上限电压4.2V后,以2C的恒定电流放电至放电终止电压3.0V,将此充放电循环作为1个循环,重复该循环直到500次。对于循环试验结束后的电池,在25℃环境下进行3个循环的充放电,将该第3个循环的0.2C放电容量作为循环后容量,将该循环后容量相对于初期容量的比例作为容量保持率。The cycle test was performed in a high-temperature environment of 60° C., which is regarded as the upper limit temperature for practical use of lithium secondary batteries. After charging to the charging upper limit voltage of 4.2V by the constant current and constant voltage method of 2C, discharge to the end-of-discharge voltage of 3.0V by the constant current of 2C. This charge-discharge cycle is regarded as one cycle, and the cycle is repeated until 500 times. For the battery after the cycle test, charge and discharge for 3 cycles at 25°C, the 0.2C discharge capacity of the third cycle is taken as the post-cycle capacity, and the ratio of the post-cycle capacity to the initial capacity is taken as the capacity retention rate.
电解液[6]实施例1Electrolyte [6] Embodiment 1
在干燥氩氛围下,向上述混合非水溶剂中混合10ppm量的甲醇,以0.8mol/L加入六氟磷酸锂(LiPF6),并进行溶解。1天后的溶液中的氟化氢(HF)量为12ppm,2周后再次测定时为14ppm。在该混合溶液(混合2周后)中混合0.3质量%的量的六甲基环三硅氧烷,制备非水电解液。使用该非水电解液用上述方法制作电池,并测定输出功率和容量保持率。结果如电解液[6]表1所示。In a dry argon atmosphere, 10 ppm of methanol was mixed into the mixed non-aqueous solvent, and lithium hexafluorophosphate (LiPF 6 ) was added and dissolved at 0.8 mol/L. The amount of hydrogen fluoride (HF) in the solution after 1 day was 12 ppm, and when measured again 2 weeks later, it was 14 ppm. Hexamethylcyclotrisiloxane was mixed in an amount of 0.3% by mass in this mixed solution (2 weeks after mixing) to prepare a non-aqueous electrolytic solution. Using this non-aqueous electrolytic solution, a battery was produced by the method described above, and the output power and capacity retention were measured. The results are shown in Table 1 of the electrolyte [6].
电解液[6]实施例2Electrolyte [6] Embodiment 2
除了在电解液[6]实施例1中将甲醇的混合量变更为相对于混合非水溶剂为20ppm的量以外,与电解液[6]实施例1同样地制备非水电解液,使用该非水电解液制作电池,并测定输出功率和容量保持率。结果如电解液[6]表1所示。另外,混合LiPF61天之后的混合溶液中的氟化氢(HF)量为16ppm,2周后再次测定时为19ppm。Except that in the electrolytic solution [6] Example 1, the mixing amount of methanol was changed to an amount of 20 ppm relative to the mixed non-aqueous solvent, a non-aqueous electrolytic solution was prepared in the same manner as the electrolytic solution [6] Example 1, and the non-aqueous electrolytic solution was used. The water electrolyte was used to make batteries, and the output power and capacity retention were measured. The results are shown in Table 1 of the electrolyte [6]. In addition, the amount of hydrogen fluoride (HF) in the mixed solution one day after mixing LiPF 6 was 16 ppm, and it was 19 ppm when measured again two weeks later.
电解液[6]实施例3Electrolyte [6] Embodiment 3
除了在电解液[6]实施例1中将甲醇的混合量变更为相对于混合非水溶剂为35ppm的量以外,与电解液[6]实施例1同样地制备非水电解液,使用该非水电解液制作电池,并测定输出功率和容量保持率。结果如电解液[6]表1所示。另外,混合LiPF61天之后的混合溶液中的氟化氢(HF)量为22ppm,2周后再次测定时为27ppm。Except that in the electrolytic solution [6] Example 1, the mixing amount of methanol was changed to an amount of 35 ppm relative to the mixed non-aqueous solvent, a non-aqueous electrolytic solution was prepared in the same manner as the electrolytic solution [6] Example 1, and the non-aqueous electrolytic solution was used. The water electrolyte was used to make batteries, and the output power and capacity retention were measured. The results are shown in Table 1 of the electrolyte [6]. In addition, the amount of hydrogen fluoride (HF) in the mixed solution one day after mixing LiPF 6 was 22 ppm, and it was 27 ppm when measured again two weeks later.
电解液[6]实施例4Electrolyte [6] Embodiment 4
除了在电解液[6]实施例1中相对于混合非水溶剂混合15ppm量的乙二醇(EG)来替代甲醇以外,与电解液[6]实施例1同样地制备非水电解液,使用该非水电解液制作电池,并测定输出功率和容量保持率。结果如电解液[6]表1所示。另外,混合LiPF61天之后的混合溶液中的氟化氢(HF)量为14ppm,2周后再次测定时为16ppm。Except in electrolytic solution [6] embodiment 1, the ethylene glycol (EG) of mixing 15ppm amount with respect to mixed nonaqueous solvent replaces methanol, prepare nonaqueous electrolytic solution in the same way as electrolytic solution [6] embodiment 1, use The non-aqueous electrolytic solution was used to manufacture a battery, and the output power and capacity retention were measured. The results are shown in Table 1 of the electrolyte [6]. In addition, the amount of hydrogen fluoride (HF) in the mixed solution one day after mixing LiPF 6 was 14 ppm, and it was 16 ppm when measured again two weeks later.
电解液[6]实施例5Electrolyte [6] Embodiment 5
除了在电解液[6]实施例1中相对于混合非水溶剂混合35ppm量的乙二醇来替代甲醇以外,与电解液[6]实施例1同样地制备非水电解液,使用该非水电解液制造电池,并测定输出功率和容量保持率。结果如电解液[6]表1所示。另外,混合LiPF61天之后的混合溶液中的氟化氢(HF)量为23ppm,2周后再次测定时为27ppm。Except in electrolyte solution [6] embodiment 1, mix the ethylene glycol of 35ppm amount with respect to mixed nonaqueous solvent to replace methanol, prepare nonaqueous electrolyte solution in the same way as electrolyte solution [6] embodiment 1, use this nonaqueous The electrolyte was used to manufacture batteries, and the output power and capacity retention were measured. The results are shown in Table 1 of the electrolyte [6]. In addition, the amount of hydrogen fluoride (HF) in the mixed solution one day after mixing LiPF 6 was 23 ppm, and it was 27 ppm when measured again two weeks later.
电解液[6]实施例6Electrolyte [6] Embodiment 6
除了在电解液[6]实施例1中相对于混合非水溶剂混合25ppm甲醇和25ppm乙二醇以外,与电解液[6]实施例1同样地制备非水电解液,使用该非水电解液制作电池,并测定输出功率和容量保持率。结果如电解液[6]表1所示。另外,混合LiPF61天之后的混合溶液中的氟化氢(HF)量为31ppm,2周后再次测定时为36ppm。Except mixing 25ppm methanol and 25ppm ethylene glycol with respect to the mixed nonaqueous solvent in the electrolyte solution [6] Example 1, prepare the nonaqueous electrolyte solution in the same manner as the electrolyte solution [6] Example 1, and use the nonaqueous electrolyte solution Batteries were manufactured, and output power and capacity retention were measured. The results are shown in Table 1 of the electrolyte [6]. In addition, the amount of hydrogen fluoride (HF) in the mixed solution 1 day after mixing LiPF 6 was 31 ppm, and it was 36 ppm when measured again 2 weeks later.
电解液[6]实施例7Electrolyte [6] Embodiment 7
除了在电解液[6]实施例1中相对于混合溶液混合0.3质量%的量的二氟磷酸锂(根据Inorganic Nuclear Chemistry Letters(1969),5(7)的第581页~第582页中记载的方法制备)来替代六甲基环三硅氧烷以外,与电解液[6]实施例1同样地制备非水电解液,使用该非水电解液制作电池,并测定输出功率和容量保持率定。结果如电解液[6]表1所示。In addition to mixing lithium difluorophosphate in an amount of 0.3% by mass with respect to the mixed solution in Example 1 of the electrolyte [6] (according to Inorganic Nuclear Chemistry Letters (1969), 5(7) 581-582 Prepared by the method) to replace hexamethylcyclotrisiloxane, prepare the non-aqueous electrolyte in the same manner as the electrolyte [6] Example 1, use the non-aqueous electrolyte to make a battery, and measure the output power and capacity retention Certainly. The results are shown in Table 1 of the electrolyte [6].
电解液[6]实施例8Electrolyte [6] Embodiment 8
除了在电解液[6]实施例3中相对于混合溶液混合0.3质量%的量的二氟磷酸锂来替代六甲基环三硅氧烷以外,与电解液[6]实施例3同样地制备非水电解液,使用该非水电解液制作电池,并测定输出功率和容量保持率。结果如电解液[6]表1所示。Prepared in the same manner as in Electrolytic Solution [6] Example 3, except that lithium difluorophosphate was mixed in an amount of 0.3% by mass with respect to the mixed solution in Electrolytic Solution [6] Example 3 instead of hexamethylcyclotrisiloxane A non-aqueous electrolytic solution, using the non-aqueous electrolytic solution to manufacture a battery, and measuring output power and capacity retention. The results are shown in Table 1 of the electrolyte [6].
电解液[6]实施例9Electrolyte [6] Embodiment 9
除了在电解液[6]实施例4中相对于混合溶液混合0.3质量%的量的二氟磷酸锂来替代六甲基环三硅氧烷以外,与电解液[6]实施例4同样地制备非水电解液,使用该非水电解液制作电池,并测定输出功率和容量保持率。结果如电解液[6]表1所示。Prepared in the same manner as in Electrolytic Solution [6] Example 4, except that lithium difluorophosphate was mixed in an amount of 0.3% by mass with respect to the mixed solution instead of hexamethylcyclotrisiloxane in Electrolytic Solution [6] Example 4 A non-aqueous electrolytic solution, using the non-aqueous electrolytic solution to manufacture a battery, and measuring output power and capacity retention. The results are shown in Table 1 of the electrolyte [6].
电解液[6]实施例10Electrolyte [6] Embodiment 10
除了在电解液[6]实施例6中相对于混合溶液混合0.3质量%的量的二氟磷酸锂来替代六甲基环三硅氧烷以外,与电解液[6]实施例6同样地制备非水电解液,使用该非水电解液制作电池,并测定输出功率和容量保持率。结果如电解液[6]表1所示。Prepared in the same manner as in Electrolytic Solution [6] Example 6, except that lithium difluorophosphate was mixed in an amount of 0.3% by mass with respect to the mixed solution in the Electrolytic Solution [6] Example 6 instead of hexamethylcyclotrisiloxane A non-aqueous electrolytic solution, using the non-aqueous electrolytic solution to manufacture a battery, and measuring output power and capacity retention. The results are shown in Table 1 of the electrolyte [6].
电解液[6]实施例11Electrolyte [6] Embodiment 11
除了在电解液[6]实施例6中相对于混合溶液混合0.3质量%的量的甲磺酸三甲基甲硅烷基酯来替代六甲基环三硅氧烷以外,与电解液[6]实施例6同样地制备非水电解液,使用该非水电解液制作电池,并测定输出功率和容量保持率。结果如电解液[6]表1所示。In addition to mixing trimethylsilyl methanesulfonate in an amount of 0.3% by mass with respect to the mixed solution in the electrolyte [6] Example 6 instead of hexamethylcyclotrisiloxane, the electrolyte [6] In Example 6, a non-aqueous electrolytic solution was prepared in the same manner, and a battery was fabricated using the non-aqueous electrolytic solution, and the output power and capacity retention were measured. The results are shown in Table 1 of the electrolyte [6].
电解液[6]实施例12Electrolyte [6] Example 12
<二次电池的制作-2><Production of secondary battery-2>
[正极的制作][making of positive electrode]
在N-甲基吡咯烷酮溶剂中混合90质量%作为正极活性物质的钴酸锂(LiCoO2)、5质量%的作为导电材料的乙炔黑和5质量%作为粘合剂的聚偏氟乙烯(PVdF),制成浆料。将得到的浆料涂布在厚度20μm的铝箔两面,并干燥,用压制机压延成厚度80μm,再将其切成宽52mm、长830mm大小,作为正极。其中,表面背面都在长度方向上设置50mm的未涂布部分,活性物质层的长度为780mm。Mix 90% by mass of lithium cobaltate (LiCoO 2 ) as a positive electrode active material, 5% by mass of acetylene black as a conductive material, and 5% by mass of polyvinylidene fluoride (PVdF ) to make slurry. The obtained slurry was coated on both sides of an aluminum foil with a thickness of 20 μm, dried, and rolled to a thickness of 80 μm by a press machine, and then cut into a size of 52 mm in width and 830 mm in length as a positive electrode. Among them, an uncoated portion of 50 mm was provided in the longitudinal direction on both the front and back sides, and the length of the active material layer was 780 mm.
[负极的制作][Production of Negative Electrode]
在98重量份人造石墨粉末KS-44(timcal公司制造,商品名)中添加100重量份作为增稠剂的羧甲基纤维素钠的水性分散液(羧甲基纤维素钠的浓度为1质量%)、2重量份作为粘合剂的苯乙烯-丁二烯橡胶的水性分散液(苯乙烯-丁二烯橡胶的浓度为50质量%),用分散器进行混合,制成浆料。将得到的浆料均匀涂布在作为负极集电体的厚度18μm的铜箔的两面,干燥后,用压制机压延成厚度为85μm,再将其切成宽56mm、长850mm的大小,作为负极。其中,表面背面都在长度方向上设置30mm的未涂布部分。In 98 parts by weight of artificial graphite powder KS-44 (manufactured by timcal company, trade name), add 100 parts by weight of an aqueous dispersion of sodium carboxymethyl cellulose as a thickener (the concentration of sodium carboxymethyl cellulose is 1 mass %), 2 parts by weight of an aqueous dispersion of styrene-butadiene rubber as a binder (the concentration of styrene-butadiene rubber is 50% by mass), and mixed with a disperser to prepare a slurry. The obtained slurry was uniformly coated on both sides of a copper foil with a thickness of 18 μm as the negative electrode current collector, and after drying, it was rolled to a thickness of 85 μm by a press, and then cut into a size of 56 mm wide and 850 mm long, as the negative electrode . Among them, an uncoated portion of 30 mm was provided in the longitudinal direction on both the front and back sides.
[非水电解液的制作][Production of non-aqueous electrolyte]
制作与电解液[6]实施例6同样的非水电解液。The same non-aqueous electrolytic solution as in the electrolytic solution [6] Example 6 was prepared.
[电池的组装][assembling the battery]
将正极和负极与聚乙烯制的隔板一起重叠卷绕并使得正极与负极不直接接触,制成电极体。使正极和负极的端子露出外部地收纳在电池罐中。然后,向其中注入5mL后述的非水电解液后,进行铆接成型,制造18650型圆筒电池。该电池的额定放电容量约为0.7安培小时(Ah),用10kHz交流法测定的直流电阻约为35毫欧(mΩ)。对于上述电池,与电解液[6]实施例6同样地测定输出功率和容量保持率。结果如电解液[6]表1所示。The positive electrode and the negative electrode were overlapped and wound together with a polyethylene separator so that the positive electrode and the negative electrode were not in direct contact to form an electrode body. The positive and negative terminals are housed in the battery can with the terminals exposed to the outside. Then, 5 mL of a non-aqueous electrolytic solution described later was injected thereinto, and then caulking was performed to manufacture a 18650-type cylindrical battery. The battery has a rated discharge capacity of about 0.7 ampere hours (Ah) and a DC resistance of about 35 milliohms (mΩ) measured by the 10 kHz AC method. For the above battery, the output power and capacity retention were measured in the same manner as in Example 6 of the electrolyte solution [6]. The results are shown in Table 1 of the electrolyte [6].
电解液[6]比较例1Electrolyte [6] Comparative Example 1
电解液[6]实施例3中,使用不混合六甲基环三硅氧烷而制备的非水电解液,制作电池,并测定输出功率和容量保持率。结果如电解液[6]表1所示。Electrolyte solution [6] In Example 3, a non-aqueous electrolyte solution prepared without mixing hexamethylcyclotrisiloxane was used to fabricate a battery, and the output power and capacity retention were measured. The results are shown in Table 1 of the electrolyte [6].
电解液[6]比较例2Electrolyte [6] Comparative Example 2
电解液[6]实施例5中,使用不混合六甲基环三硅氧烷而制备的非水电解液,制作电池,并测定输出功率和容量保持率。结果如电解液[6]表1所示。Electrolyte solution [6] In Example 5, a non-aqueous electrolyte solution prepared without mixing hexamethylcyclotrisiloxane was used to fabricate a battery, and the output power and capacity retention were measured. The results are shown in Table 1 of the electrolyte [6].
电解液[6]比较例3Electrolyte [6] Comparative Example 3
电解液[6]实施例6中,使用不混合六甲基环三硅氧烷而制备的非水电解液,制作电池,并测定输出功率和容量保持率。结果如电解液[6]表1所示。Electrolyte solution [6] In Example 6, a non-aqueous electrolyte solution prepared without mixing hexamethylcyclotrisiloxane was used to fabricate a battery, and the output power and capacity retention were measured. The results are shown in Table 1 of the electrolyte [6].
电解液[6]比较例4Electrolyte [6] Comparative Example 4
电解液[6]实施例1中,使用在混合非水溶剂中不混合甲醇而制备的非水电解液,制作电池,并测定输出功率和容量保持率。结果如电解液[6]表1所示。另外,混合LiPF61天之后的溶液中的氟化氢(HF)量为9ppm,2周后再次测定时仍为9ppm。Electrolyte solution [6] In Example 1, a nonaqueous electrolyte solution prepared without mixing methanol in a mixed nonaqueous solvent was used to fabricate a battery, and the output power and capacity retention were measured. The results are shown in Table 1 of the electrolyte [6]. In addition, the amount of hydrogen fluoride (HF) in the solution one day after mixing LiPF 6 was 9 ppm, and it was still 9 ppm when measured again 2 weeks later.
电解液[6]比较例5Electrolyte [6] Comparative Example 5
电解液[6]比较例4中,使用混合0.3质量%的二氟磷酸锂来替代六甲基环三硅氧烷而制备的非水电解液,制作电池,并测定输出功率和容量保持率。结果如电解液[6]表1所示。Electrolyte solution [6] In Comparative Example 4, a non-aqueous electrolyte solution prepared by mixing 0.3% by mass of lithium difluorophosphate instead of hexamethylcyclotrisiloxane was used to fabricate a battery, and the output power and capacity retention were measured. The results are shown in Table 1 of the electrolyte [6].
电解液[6]比较例6Electrolyte [6] Comparative Example 6
电解液[6]比较例4中,使用混合0.3质量%的甲磺酸三甲基甲硅烷基酯来替代六甲基环三硅氧烷而制备的非水电解液,制作电池,并测定输出功率和容量保持率。结果如电解液[6]表1所示。Electrolyte solution [6] In Comparative Example 4, a non-aqueous electrolyte solution prepared by mixing 0.3% by mass of trimethylsilyl methanesulfonate instead of hexamethylcyclotrisiloxane was used to manufacture a battery and measure the output Power and capacity retention. The results are shown in Table 1 of the electrolyte [6].
电解液[6]比较例7Electrolyte [6] Comparative Example 7
电解液[6]实施例1中,除了将甲醇的混合量变更为相对于混合非水溶剂为700ppm的量以外,与电解液[6]实施例1同样地制备非水电解液,使用该非水电解液制作电池,并测定输出功率和容量保持率。结果如电解液[6]表1所示。另外,混合LiPF61天之后的溶液中的氟化氢(HF)量为321ppm,2周后再次测定时为403ppm。In the electrolyte solution [6] Example 1, except that the mixing amount of methanol was changed to an amount of 700 ppm relative to the mixed non-aqueous solvent, a non-aqueous electrolyte solution was prepared in the same manner as the electrolyte solution [6] Example 1, and the non-aqueous electrolyte solution was used. The water electrolyte was used to make batteries, and the output power and capacity retention were measured. The results are shown in Table 1 of the electrolyte [6]. In addition, the amount of hydrogen fluoride (HF) in the solution one day after mixing LiPF 6 was 321 ppm, and it was 403 ppm when measured again two weeks later.
电解液[6]比较例8Electrolyte [6] Comparative Example 8
电解液[6]实施例1中,使用在混合非水溶剂中不混合甲醇,并且不混合六甲基环三硅氧烷而制备的非水电解液,制作电池,并测定输出功率和容量保持率。结果如电解液[6]表1所示。Electrolyte solution [6] In Example 1, use the non-aqueous electrolyte solution prepared without mixing methanol and hexamethylcyclotrisiloxane in the mixed non-aqueous solvent to make a battery, and measure the output power and capacity retention Rate. The results are shown in Table 1 of the electrolyte [6].
电解液[6]比较例9Electrolyte [6] Comparative Example 9
除了在电解液[6]实施例12中不混合六甲基环三硅氧烷以外,与电解液[6]实施例12同样地制备非水电解液,使用该非水电解液制作电池,并测定输出功率和容量保持率。结果如电解液[6]表1所示。Except not mixing hexamethylcyclotrisiloxane in the electrolyte solution [6] Example 12, prepare a non-aqueous electrolyte solution in the same manner as the electrolyte solution [6] Example 12, use the non-aqueous electrolyte solution to make a battery, and Determination of output power and capacity retention. The results are shown in Table 1 of the electrolyte [6].
电解液[6]表1Electrolyte [6] Table 1
[表59][Table 59]
由电解液[6]表1可知,含有更多的氟化氢(HF)的电解液[6]比较例1~3的锂二次电池与电解液[6]比较例8的锂二次电池相比,在输出功率特性、循环特性两方面逊色,但是在其中加有特定化合物的电解液[6]实施例1~11的锂二次电池,在输出功率特性、循环特性两方面,发现特性的提高。进一步地,在输出功率方面,电解液[6]实施例1~11的锂二次电池,与虽然含有特定化合物但是氟化氢(HF)含量少的电解液[6]比较例4~6的锂二次电池相比,具有更高的性能,通过存在氟化氢,得到强化通过特定化合物实现的输出功率提高效果的令人惊讶的结果。但是,含有过量的氟化氢(HF)的电解液[6]比较例7的锂二次电池中,得到特别是循环特性差的结果。From Table 1 of the electrolyte [6], it can be seen that the lithium secondary battery of the electrolyte [6] comparative examples 1-3 containing more hydrogen fluoride (HF) is compared with the lithium secondary battery of the electrolyte [6] comparative example 8 , In terms of output power characteristics and cycle characteristics, the electrolyte solution [6] Examples 1 to 11 are inferior to the electrolyte solution in which a specific compound is added. In the two aspects of output power characteristics and cycle characteristics, it is found that the characteristics are improved. . Furthermore, in terms of output power, the lithium secondary batteries of the electrolyte [6] Examples 1 to 11, and the lithium secondary batteries of the comparative examples 4 to 6 in the electrolyte [6] containing a specific compound but with a small amount of hydrogen fluoride (HF) Compared with the secondary battery, it has higher performance, and the surprising result of enhancing the output power improvement effect by the specific compound was obtained by the presence of hydrogen fluoride. However, in the lithium secondary battery of Comparative Example 7, which contained an excess amount of hydrogen fluoride (HF) in the electrolytic solution [6], particularly poor cycle characteristics were obtained.
此外,低容量、直流电阻高的电池结构的电解液[6]实施例12相对于电解液[6]比较例9的输出功率上升率仅约为19%,另一方面,高容量、直流电阻低的电池结构的电解液[6]实施例6相对于电解液[6]比较例3(都含有甲醇25ppm和乙二醇25ppm)的输出功率上升率约为29%,电解液[6]实施例5相对于电解液[6]比较例2(都含有乙二醇35ppm)的输出功率上升率约为26%,电解液[6]实施例3相对于电解液[6]比较例1(都含有甲醇35ppm)的输出功率上升率约为26%,较大。由此可知在高容量、直流电阻低的电池结构中,本发明的效果特别大。In addition, the output power increase rate of the electrolyte [6] Example 12 of the battery structure with low capacity and high DC resistance is only about 19% relative to the electrolyte [6] Comparative Example 9. On the other hand, the high capacity, DC resistance The output power increase rate of the electrolyte solution [6] embodiment 6 of the low battery structure is about 29% relative to the electrolyte solution [6] comparative example 3 (both containing methanol 25ppm and ethylene glycol 25ppm), and the electrolyte solution [6] implements The output power increase rate of example 5 is about 26% with respect to electrolyte solution [6] comparative example 2 (both containing ethylene glycol 35ppm), and electrolyte solution [6] embodiment 3 is relative to electrolyte solution [6] comparative example 1 (both Containing methanol 35ppm) the output power increase rate is about 26%, which is relatively large. From this, it can be seen that the effect of the present invention is particularly large in a battery structure with high capacity and low DC resistance.
如上所述,通过使用本发明的非水电解液,也就是在非水溶剂中混合含氟锂盐而形成的二次电池用非水电解液,循环特性不会变差,并可以得到大的输出功率特性,所述二次电池用非水电解液的特征是:上述非水电解液中的氟化氢(HF)为10ppm~300ppm,还含有选自下述物质中的至少一种化合物,并且其在全部非水电解液中的含量为10ppm以上,所述物质包括:通式(1)表示的环状硅氧烷化合物、通式(2)表示的氟硅烷化合物、通式(3)表示的化合物、分子内具有S-F键的化合物、硝酸盐、亚硝酸盐、单氟磷酸盐、二氟磷酸盐、乙酸盐和丙酸盐。As mentioned above, by using the non-aqueous electrolytic solution of the present invention, that is, the non-aqueous electrolytic solution for secondary batteries formed by mixing fluorine-containing lithium salt in the non-aqueous solvent, the cycle characteristics will not deteriorate, and a large battery life can be obtained. Output power characteristics, the non-aqueous electrolyte solution for secondary batteries is characterized by: the hydrogen fluoride (HF) in the above-mentioned non-aqueous electrolyte solution is 10 ppm to 300 ppm, and also contains at least one compound selected from the following substances, and its The content in all non-aqueous electrolyte solutions is more than 10ppm, and the substances include: cyclic siloxane compounds represented by general formula (1), fluorosilane compounds represented by general formula (2), fluorosilane compounds represented by general formula (3) Compounds, compounds with S-F bonds in their molecules, nitrates, nitrites, monofluorophosphates, difluorophosphates, acetates and propionates.
本实施例中,通过对纯化的非水溶剂加入醇类,可发挥效果,但是在非水溶剂中本来含有醇类或水分时,通过调节非水溶剂的纯化条件,不必特意加入醇类,可以得到同样的效果。一般来说,非水溶剂的纯化需要花费工夫,在工业上引起成本提高,而本发明中,可以无需多余的纯化来制备高性能的非水电解液,其价值非常大。In this embodiment, by adding alcohols to the purified non-aqueous solvent, the effect can be exerted, but when the non-aqueous solvent originally contains alcohols or water, by adjusting the purification conditions of the non-aqueous solvent, it is not necessary to add alcohols deliberately, which can to get the same effect. In general, the purification of non-aqueous solvents requires time and effort and increases industrial costs. However, in the present invention, a high-performance non-aqueous electrolytic solution can be produced without unnecessary purification, which is very valuable.
电解液[7]<电池的制作-1>Electrolyte [7] <Battery Production-1>
[正极的制造][production of positive electrode]
在N-甲基吡咯烷酮溶剂中混合90质量%作为正极活性物质的钴酸锂(LiCoO2)、5质量%的作为导电材料的乙炔黑和5质量%作为粘合剂的聚偏氟乙烯(PVdF),制成浆料。将得到的浆料涂布在厚度20μm的铝箔两面,并干燥,用压制机压延成厚度80μm,再将其切成宽52mm、长830mm大小,作为正极。其中,表面背面都在长度方向上设置50mm的未涂布部分,活性物质层的长度为780mm。Mix 90% by mass of lithium cobaltate (LiCoO 2 ) as a positive electrode active material, 5% by mass of acetylene black as a conductive material, and 5% by mass of polyvinylidene fluoride (PVdF ) to make slurry. The obtained slurry was coated on both sides of an aluminum foil with a thickness of 20 μm, dried, and rolled to a thickness of 80 μm by a press machine, and then cut into a size of 52 mm in width and 830 mm in length as a positive electrode. Among them, an uncoated portion of 50 mm was provided in the longitudinal direction on both the front and back sides, and the length of the active material layer was 780 mm.
[负极的制作][Production of Negative Electrode]
在98重量份人造石墨粉末KS-44(timcal公司制造,商品名)中添加100重量份作为增稠剂的羧甲基纤维素钠的水性分散液(羧甲基纤维素钠的浓度为1质量%)、2重量份作为粘合剂的苯乙烯-丁二烯橡胶的水性分散液(苯乙烯-丁二烯橡胶的浓度为50质量%),用分散器进行混合,制成浆料。将得到的浆料均匀涂布在作为负极集电体的厚度18μm的铜箔的两面,干燥后,用压制机压延成厚度为85μm,再将其切成宽56mm、长850mm的大小,作为负极。其中,表面背面都在长度方向上设置30mm的未涂布部分。Add 100 parts by weight of an aqueous dispersion of sodium carboxymethylcellulose as a thickener in 98 parts by weight of artificial graphite powder KS-44 (manufactured by timcal company, trade name) (the concentration of sodium carboxymethylcellulose is 1 mass %), 2 parts by weight of an aqueous dispersion of styrene-butadiene rubber as a binder (the concentration of styrene-butadiene rubber is 50% by mass), and mixed with a disperser to prepare a slurry. The obtained slurry was uniformly coated on both sides of a copper foil with a thickness of 18 μm as the negative electrode current collector, and after drying, it was rolled to a thickness of 85 μm with a press, and then cut into a size of 56 mm wide and 850 mm long as the negative electrode . Among them, an uncoated portion of 30 mm was provided in the longitudinal direction on both the front and back sides.
[电解液的制备][Preparation of Electrolyte Solution]
在干燥氩氛围下,在纯化的碳酸亚乙酯(EC)、碳酸二甲酯(DMC)和碳酸甲乙酯(EMC)的体积比3∶3∶4的混合溶剂中,以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6),制备非水电解液(1)。进一步使该非水电解液(1)中含有0.5质量%的根据Inorganic Nuclear Chemistry Letters(1969),5(7)的第581页~第582页中记载的方法制备的二氟磷酸锂,制备非水电解液(2)。Under a dry argon atmosphere, in a mixed solvent of purified ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) with a volume ratio of 3:3:4, 1 mol/L Concentration Dissolve fully dry lithium hexafluorophosphate (LiPF 6 ) to prepare non-aqueous electrolyte solution (1). Further, 0.5% by mass of lithium difluorophosphate prepared according to the method described on pages 581 to 582 of Inorganic Nuclear Chemistry Letters (1969), 5 (7) was contained in the non-aqueous electrolytic solution (1) to prepare a non-aqueous electrolytic solution (1). Water electrolyte (2).
[电池的组装][assembling the battery]
将正极和负极与聚乙烯制的隔板一起重叠卷绕并使得正极与负极不直接接触,制成电极体。使正极和负极的端子露出外部地收纳在电池罐中。然后,向其中注入5mL后述的非水电解液后,进行铆接成型,制造18650型圆筒电池。The positive electrode and the negative electrode were overlapped and wound together with a polyethylene separator so that the positive electrode and the negative electrode were not in direct contact to form an electrode body. The positive and negative terminals are housed in the battery can with the terminals exposed to the outside. Then, 5 mL of a non-aqueous electrolytic solution described later was injected thereinto, and then caulking was performed to manufacture a 18650-type cylindrical battery.
[电池的评价][evaluation of the battery]
(初期充放电)(initial charge and discharge)
对制造的圆筒状电池,在25℃下用0.2C的恒定电流恒定电压充电法充电至4.2V后,以0.2C的恒定电流放电至3.0V。将其进行5个循环使电池稳定。将此时的第5个循环的放电容量作为初期容量。另外,将1小时放电额定容量的电流值作为1C,所述额定容量取决于1小时率(one-hour-rate)的放电容量。The manufactured cylindrical battery was charged to 4.2V at 25° C. by a constant current constant voltage charging method of 0.2C, and then discharged to 3.0V by a constant current of 0.2C. This was performed for 5 cycles to stabilize the battery. The discharge capacity at the 5th cycle at this time was taken as the initial capacity. In addition, the current value of the 1-hour discharge rated capacity which depends on the 1-hour-rate discharge capacity is defined as 1C.
(循环试验)(cycle test)
对实施了初期充放电的电池,在60℃下以1C的恒定电流恒定电压充电至4.2V后,以1C的恒定电流放电至3.0V,将这样的充放电进行500个循环。将此时的第500个循环放电容量相对于第1个循环放电容量的比例作为循环保持率。The battery subjected to the initial charge and discharge was charged to 4.2V at a constant current and constant voltage of 1C at 60°C, and then discharged to 3.0V at a constant current of 1C, and such charge and discharge were performed for 500 cycles. The ratio of the discharge capacity at the 500th cycle to the discharge capacity at the first cycle at this time was defined as the cycle retention ratio.
(低温试验)(low temperature test)
对实施了初期充放电的电池,在25℃下用0.2C的恒定电流恒定电压充电法充电至4.2V后,在-30℃下实施0.2C的恒定电流放电。将此时的放电容量作为初期低温容量,将初期低温容量相对于初期容量的比例作为初期低温放电率。The battery subjected to the initial charge and discharge was charged to 4.2V by a 0.2C constant current constant voltage charging method at 25°C, and then a 0.2C constant current discharge was performed at -30°C. The discharge capacity at this time was taken as the initial low-temperature capacity, and the ratio of the initial low-temperature capacity to the initial capacity was taken as the initial low-temperature discharge rate.
此外,对循环试验后的电池在25℃下用0.2C的恒定电流恒定电压充电法充电至4.2V后,以0.2C的恒定电流放电至3.0V。对其进行3个循环,将其第3个循环的放电容量作为循环后容量。然后,对同一电池在25℃下以0.2C的恒定电流恒定电压充电法充电至4.2V后,在-30℃下实施0.2C的恒定电流放电。将此时的放电容量作为循环后低温容量,将循环后低温容量相对于循环后容量的比例作为循环后低温放电率。In addition, the battery after the cycle test was charged to 4.2V at 25°C by a constant current constant voltage charging method of 0.2C, and then discharged to 3.0V by a constant current of 0.2C. Three cycles were performed on it, and the discharge capacity at the third cycle was taken as the post-cycle capacity. Then, the same battery was charged to 4.2V by a 0.2C constant current constant voltage charging method at 25°C, and then a 0.2C constant current discharge was performed at -30°C. The discharge capacity at this time was taken as the post-cycle low-temperature capacity, and the ratio of the post-cycle low-temperature capacity to the post-cycle capacity was taken as the post-cycle low-temperature discharge rate.
电解液[7]实施例1Electrolyte [7] Embodiment 1
在非水电解液(2)中,以相对于全部电解液质量为1质量%的量混合碳酸亚乙烯酯(下面简称为“VC”),制备非水电解液(3)。使用该非水电解液(3),制作18650型电池,并测定循环保持率和低温放电率。结果如电解液[7]表1所示。另外,用离心分离机从初期充放电结束后的电池中回收电解液,测得的VC的量为0.40质量%。In the nonaqueous electrolytic solution (2), vinylene carbonate (hereinafter abbreviated as "VC") was mixed in an amount of 1% by mass relative to the mass of the entire electrolytic solution to prepare a nonaqueous electrolytic solution (3). Using this non-aqueous electrolytic solution (3), a 18650-type battery was produced, and the cycle retention rate and low-temperature discharge rate were measured. The results are shown in Table 1 of the electrolyte [7]. In addition, the electrolytic solution was recovered from the battery after the initial charge and discharge by a centrifuge, and the amount of VC measured was 0.40% by mass.
电解液[7]比较例1Electrolyte [7] Comparative Example 1
使用非水电解液(1),与电解液[7]实施例1同样地制作18650型电池,并测定循环保持率和低温放电率。结果如电解液[7]表1所示。Using the non-aqueous electrolytic solution (1), a 18650-type battery was produced in the same manner as in the electrolytic solution [7] Example 1, and the cycle retention rate and low-temperature discharge rate were measured. The results are shown in Table 1 of the electrolyte [7].
电解液[7]比较例2Electrolyte [7] Comparative Example 2
在非水电解液(1)中,以相对于总电解液质量为1质量%的量混合VC,制备非水电解液(4)。使用该非水电解液(4),与电解液[7]实施例1同样地制作18650型电池,并测定循环保持率和低温放电率。结果如电解液[7]表1所示。另外,用离心分离机从初期充放电结束后的电池中回收电解液,测得的VC的量为0.22质量%。In the nonaqueous electrolytic solution (1), VC was mixed in an amount of 1% by mass relative to the total electrolytic solution mass to prepare a nonaqueous electrolytic solution (4). Using this non-aqueous electrolytic solution (4), a 18650-type battery was produced in the same manner as in the electrolytic solution [7] Example 1, and the cycle retention rate and low-temperature discharge rate were measured. The results are shown in Table 1 of the electrolyte [7]. In addition, the electrolytic solution was recovered from the battery after the initial charge and discharge by a centrifuge, and the measured amount of VC was 0.22% by mass.
电解液[7]比较例3Electrolyte [7] Comparative Example 3
除了使用非水电解液(2),且不混合VC以外,与电解液[7]实施例1同样地制作18650型电池,并测定循环保持率和低温放电率。结果如电解液[7]表1所示。A 18650-type battery was produced in the same manner as in Example 1 of the electrolyte solution [7] except that the non-aqueous electrolyte solution (2) was used and VC was not mixed, and the cycle retention rate and low-temperature discharge rate were measured. The results are shown in Table 1 of the electrolyte [7].
电解液[7]比较例4Electrolyte [7] Comparative Example 4
在非水电解液(2)中,以相对于总电解液质量为5质量%的量混合VC,制备非水电解液(5)。在电解液[7]实施例1中,使用该非水电解液(5),制作18650型电池,并测定循环保持率和低温放电率。结果如电解液[7]表1所示。In the nonaqueous electrolytic solution (2), VC was mixed in an amount of 5% by mass relative to the total electrolytic solution mass to prepare a nonaqueous electrolytic solution (5). In the electrolyte solution [7] Example 1, the non-aqueous electrolyte solution (5) was used to manufacture a 18650-type battery, and the cycle retention rate and low-temperature discharge rate were measured. The results are shown in Table 1 of the electrolyte [7].
电解液[7]实施例2Electrolyte [7] Example 2
<电池的制作-2><Production of battery-2>
[正极的制作][making of positive electrode]
在N-甲基吡咯烷酮溶剂中混合90质量%作为正极活性物质的钴酸锂(LiCoO2)、5质量%的作为导电材料的乙炔黑和5质量%作为粘合剂的聚偏氟乙烯(PVdF),制成浆料。将得到的浆料涂布在厚度15μm的铝箔两面,并干燥,用压制机压延成厚度80μm,再将其切成具有宽100mm、长100mm大小的活性物质层和宽30mm的未涂布部分的形状,作为正极。Mix 90% by mass of lithium cobaltate (LiCoO 2 ) as a positive electrode active material, 5% by mass of acetylene black as a conductive material, and 5% by mass of polyvinylidene fluoride (PVdF ) to make slurry. The obtained slurry was coated on both sides of an aluminum foil with a thickness of 15 μm, dried, rolled to a thickness of 80 μm with a press, and cut into an active material layer having a width of 100 mm and a length of 100 mm and an uncoated portion of a width of 30 mm. shape, as the positive electrode.
[负极的制作][Production of Negative Electrode]
在98重量份人造石墨粉末KS-44(timcal公司制造,商品名)中添加100重量份作为增稠剂的羧甲基纤维素钠的水性分散液(羧甲基纤维素钠的浓度为1质量%)、2重量份作为粘合剂的苯乙烯-丁二烯橡胶的水性分散液(苯乙烯-丁二烯橡胶的浓度为50质量%),用分散器进行混合,制成浆料。将得到的浆料涂布在厚度10μm的铜箔两面,并干燥,用压制机压延成厚度为75μm,再将其切成具有宽104mm、长104mm大小的活性物质层和宽30mm的未涂布部分的形状,作为负极。In 98 parts by weight of artificial graphite powder KS-44 (manufactured by timcal company, trade name), add 100 parts by weight of an aqueous dispersion of sodium carboxymethyl cellulose as a thickener (the concentration of sodium carboxymethyl cellulose is 1 mass %), 2 parts by weight of an aqueous dispersion of styrene-butadiene rubber as a binder (the concentration of styrene-butadiene rubber is 50% by mass), and mixed with a disperser to prepare a slurry. The obtained slurry was coated on both sides of a copper foil with a thickness of 10 μm, dried, rolled to a thickness of 75 μm with a press, and then cut into an active material layer with a width of 104 mm and a length of 104 mm and an uncoated layer with a width of 30 mm. The shape of the part, as the negative pole.
[电池的组装][assembling the battery]
将32片正极和33片负极交替配置,各电极之间夹入多孔聚乙烯片隔板(厚度为25μm),进行叠层。此时,使正极活性物质面与负极活性物质面相对,并且不会露出负极活性物质面之外。将该正极和负极各自的未涂布部分焊接,制作集电片,将电极组密封在电池罐(外部尺寸:120×110×10mm)中。然后,在装有电极组的电池罐中注入20mL非水电解液(3),使电极充分浸透,密封而制作方形电池。32 positive electrodes and 33 negative electrodes were arranged alternately, and a porous polyethylene sheet separator (thickness: 25 μm) was sandwiched between the electrodes for lamination. At this time, the surface of the positive electrode active material faces the surface of the negative electrode active material without protruding from the surface of the negative electrode active material. The respective uncoated parts of the positive and negative electrodes were welded to produce current collector sheets, and the electrode group was sealed in a battery can (outer dimensions: 120×110×10 mm). Then, 20 mL of the non-aqueous electrolytic solution (3) was injected into the battery can containing the electrode group to fully soak the electrodes, and sealed to manufacture a square battery.
对这样制作的方形电池,采用与电解液[7]实施例1相同的方法实施循环试验、低温试验,并测定循环保持率和低温放电率。结果如电解液[7]表1所示。For the prismatic battery produced in this way, a cycle test and a low temperature test were carried out in the same manner as in Example 1 of the electrolyte solution [7], and the cycle retention rate and low temperature discharge rate were measured. The results are shown in Table 1 of the electrolyte [7].
电解液[7]比较例5Electrolyte [7] Comparative Example 5
电解液[7]实施例2中,使用非水电解液(1)来替代非水电解液(3),制作方形电池,并测定循环保持率和低温放电率。结果如电解液[7]表1所示。Electrolyte solution [7] In Example 2, the non-aqueous electrolyte solution (1) was used instead of the non-aqueous electrolyte solution (3) to make a square battery, and the cycle retention rate and low-temperature discharge rate were measured. The results are shown in Table 1 of the electrolyte [7].
电解液[7]比较例6Electrolyte [7] Comparative Example 6
电解液[7]实施例2中,使用非水电解液(4)来替代非水电解液(3),制作方形电池,并测定循环保持率和低温放电率。结果如电解液[7]表1所示。Electrolyte solution [7] In Example 2, the non-aqueous electrolyte solution (4) was used instead of the non-aqueous electrolyte solution (3) to make a square battery, and the cycle retention rate and low-temperature discharge rate were measured. The results are shown in Table 1 of the electrolyte [7].
电解液[7]比较例7Electrolyte [7] Comparative Example 7
电解液[7]实施例2中,使用非水电解液(2)来替代非水电解液(3),制作方形电池,并测定循环保持率和低温放电率。结果如电解液[7]表1所示。Electrolyte solution [7] In Example 2, the non-aqueous electrolyte solution (2) was used instead of the non-aqueous electrolyte solution (3) to make a square battery, and the cycle retention rate and low-temperature discharge rate were measured. The results are shown in Table 1 of the electrolyte [7].
电解液[7]表1Electrolyte [7] Table 1
[表60][Table 60]
由电解液[7]表1可知,非水电解液中含有碳酸亚乙烯酯和二氟磷酸盐的电解液[7]实施例1的锂二次电池与除了在非水电解液中不含有它们之外为相同结构的电解液[7]比较例1的锂二次电池相比,循环保持率、低温放电率都得到改善。From electrolytic solution [7] Table 1, it can be known that the lithium secondary battery of the electrolytic solution [7] embodiment 1 containing vinylene carbonate and difluorophosphate in the non-aqueous electrolytic solution is the same as the lithium secondary battery except that they are not contained in the non-aqueous electrolytic solution. In addition, compared with the lithium secondary battery of Comparative Example 1, the electrolyte solution [7] with the same structure has improved cycle retention rate and low-temperature discharge rate.
此外可知,即使与除了在非水电解液中仅含有VC之外为相同结构的电解液[7]比较例2的锂二次电池相比,循环保持率、低温放电率也都得到改善。循环保持率因VC的有无而明显变化,而通过向其中加入二氟磷酸盐,其效果增大。由初期充放电后的电池检测出的VC量存在约2倍的差异,认为其中残留的VC抑制了循环试验中的电池劣化。In addition, it can be seen that both the cycle retention rate and the low-temperature discharge rate are improved compared with the lithium secondary battery of Comparative Example 2 having the same structure as the electrolyte solution [7] except that only VC is contained in the non-aqueous electrolyte solution. The cycle retention varies significantly depending on the presence or absence of VC, but the effect is increased by adding difluorophosphate thereto. There is about a 2-fold difference in the amount of VC detected from the battery after the initial charge and discharge, and it is considered that the remaining VC suppresses battery deterioration in the cycle test.
由电解液[7]实施例1和电解液[7]比较例3可知,仅通过二氟磷酸盐不能充分提高循环保持率。此外,电解液[7]实施例1的锂二次电池与电解液[7]比较例3的锂二次电池相比,虽然初期低温放电率相同,但是不仅循环保持率有大的改善,而且循环后低温放电效率也得到提高。通过VC与二氟磷酸盐的协同效果,抑制了由于循环试验所导致的电池内部电阻的增大。From the example 1 of the electrolyte solution [7] and the comparative example 3 of the electrolyte solution [7], it can be seen that the cycle retention rate cannot be sufficiently improved only by difluorophosphate. In addition, although the initial low-temperature discharge rate of the lithium secondary battery of the electrolyte [7] Example 1 is the same as that of the lithium secondary battery of the electrolyte [7] Comparative Example 3, not only the cycle retention rate is greatly improved, but also The low-temperature discharge efficiency was also improved after cycling. By the synergistic effect of VC and difluorophosphate, the increase in the internal resistance of the battery due to the cycle test was suppressed.
以上在电解液[7]实施例2与电解液[7]比较例5~7之间也是同样的。The above is also the same between the electrolytic solution [7] Example 2 and the electrolytic solution [7] Comparative Examples 5 to 7.
若对电解液[7]实施例1的锂二次电池与含有较多VC的电解液[7]比较例4的锂二次电池进行比较,则可知电解液[7]比较例4中,虽然循环保持率相当优异,但是低温放电特性显著差,不能耐受在低温环境下的使用。If the lithium secondary battery of electrolyte [7] Example 1 is compared with the lithium secondary battery of electrolyte [7] Comparative Example 4 containing more VC, it can be seen that in electrolyte [7] Comparative Example 4, although The cycle retention rate is quite excellent, but the low-temperature discharge characteristics are remarkably poor, and it cannot withstand use in a low-temperature environment.
表中示出了采用10kHz交流法测定的直流电阻的值。本实验的方形电池与圆筒电池相比,电阻小、电容大。与电解液[7]实施例1相对于电解液[7]比较例1的低温特性提高程度相比,电解液[7]实施例2相对于电解液[7]比较例5的低温特性提高程度大,可知作为本发明的非水电解液的实施对象,电容高的二次电池或直流电阻小的二次电池是特别合适的。The table shows the values of DC resistance measured by the 10 kHz AC method. Compared with the cylindrical battery, the square battery in this experiment has smaller resistance and higher capacitance. Compared with the improvement degree of low-temperature characteristics of electrolyte solution [7] Example 1 relative to electrolyte solution [7] Comparative Example 1, the degree of improvement of low-temperature characteristics of electrolyte solution [7] Example 2 relative to electrolyte solution [7] Comparative Example 5 It can be seen that a secondary battery with high capacitance or a secondary battery with small DC resistance is particularly suitable as the object of implementation of the non-aqueous electrolyte solution of the present invention.
电解液[8]<二次电池的制作>Electrolyte [8] <Production of secondary battery>
[正极的制作][making of positive electrode]
在N-甲基吡咯烷酮溶剂中混合90质量%作为正极活性物质的钴酸锂(LiCoO2)、5质量%的作为导电材料的乙炔黑和5质量%作为粘合剂的聚偏氟乙烯(PVdF),制成浆料。将得到的浆料涂布在厚度15μm的铝箔两面,并干燥,用压制机压延成厚度80μm,再将其切成具有宽100mm、长100mm大小的活性物质层和宽30mm的未涂布部分的形状,作为正极。Mix 90% by mass of lithium cobaltate (LiCoO 2 ) as a positive electrode active material, 5% by mass of acetylene black as a conductive material, and 5% by mass of polyvinylidene fluoride (PVdF ) to make slurry. The obtained slurry was coated on both sides of an aluminum foil with a thickness of 15 μm, dried, rolled to a thickness of 80 μm with a press, and cut into an active material layer having a width of 100 mm and a length of 100 mm and an uncoated portion of a width of 30 mm. shape, as the positive electrode.
[负极的制作][Production of Negative Electrode]
在98重量份人造石墨粉末KS-44(timcal公司制造,商品名)中添加100重量份作为增稠剂的羧甲基纤维素钠的水性分散液(羧甲基纤维素钠的浓度为1质量%)、2重量份作为粘合剂的苯乙烯-丁二烯橡胶的水性分散液(苯乙烯-丁二烯橡胶的浓度为50质量%),用分散器进行混合,制成浆料。将得到的浆料涂布在厚度10μm的铜箔两面,并干燥,用压制机压延成厚度为75μm,再将其切成具有宽104mm、长104mm大小的活性物质层和宽30mm的未涂布部分的形状,作为负极。In 98 parts by weight of artificial graphite powder KS-44 (manufactured by timcal company, trade name), add 100 parts by weight of an aqueous dispersion of sodium carboxymethyl cellulose as a thickener (the concentration of sodium carboxymethyl cellulose is 1 mass %), 2 parts by weight of an aqueous dispersion of styrene-butadiene rubber as a binder (the concentration of styrene-butadiene rubber is 50% by mass), and mixed with a disperser to prepare a slurry. The obtained slurry was coated on both sides of a copper foil with a thickness of 10 μm, dried, rolled to a thickness of 75 μm with a press, and then cut into an active material layer with a width of 104 mm and a length of 104 mm and an uncoated layer with a width of 30 mm. The shape of the part, as the negative pole.
[电池的组装][assembling the battery]
将32片正极和33片负极交替配置,各电极之间夹入多孔聚乙烯片隔板(厚度为25μm),进行叠层。此时,使正极活性物质面与负极活性物质面相对,并且不会露出负极活性物质面之外。将该正极和负极各自的未涂布部分焊接,制作集电片,将电极组密封在带有排气阀的电池罐(外部尺寸:120×110×10mm)中。然后,在装有电极组的电池罐中注入20mL后述的非水电解液,使电极充分浸透,密封而制作方形电池。32 positive electrodes and 33 negative electrodes were arranged alternately, and a porous polyethylene sheet separator (thickness: 25 μm) was sandwiched between the electrodes for lamination. At this time, the surface of the positive electrode active material faces the surface of the negative electrode active material without protruding from the surface of the negative electrode active material. The respective uncoated parts of the positive and negative electrodes were welded to produce current collector sheets, and the electrode group was sealed in a battery can (outer dimensions: 120×110×10 mm) with a vent valve. Then, 20 mL of a non-aqueous electrolytic solution described later was poured into the battery can containing the electrode group, the electrodes were sufficiently permeated, and sealed to produce a square battery.
二次电池的1个电池外壳中收纳的电池要素所具有的电容,即二次电池的额定放电容量约为6安培小时(Ah),用10kHz交流法测定的直流电阻成分约为5毫欧(mΩ)。The capacitance of the battery element contained in one battery case of the secondary battery, that is, the rated discharge capacity of the secondary battery is about 6 ampere hours (Ah), and the DC resistance component measured by the 10 kHz alternating current method is about 5 milliohms ( mΩ).
电解液[8]<电池的评价>Electrolyte [8] <Battery Evaluation>
[循环保持率][Cycle Retention Rate]
[初期充放电][Initial charge and discharge]
在25℃下用0.2C的恒定电流恒定电压充电法充电至4.2V后,以0.2C的恒定电流放电至3.0V。将其进行5个循环使电池稳定。将此时的第5个循环的放电容量作为初期容量。而且,将1小时放电额定容量的电流值作为1C。After charging to 4.2V with a constant current and constant voltage charging method of 0.2C at 25°C, it was discharged to 3.0V with a constant current of 0.2C. This was performed for 5 cycles to stabilize the battery. The discharge capacity at the 5th cycle at this time was taken as the initial capacity. Furthermore, the current value of the rated discharge capacity for 1 hour was defined as 1C.
(循环试验)(cycle test)
对实施了初期充放电的电池,在60℃下以1C的恒定电流恒定电压法充电至4.2V,然后以1C的恒定电流放电至3.0V,将该充放电进行500个循环。将此时的第500个循环放电容量相对于第1个循环放电容量的比例作为循环保持率。The battery subjected to initial charge and discharge was charged to 4.2V at 60° C. with a constant current and constant voltage method of 1C, and then discharged to 3.0V with a constant current of 1C, and the charge and discharge were performed for 500 cycles. The ratio of the discharge capacity at the 500th cycle to the discharge capacity at the first cycle at this time was defined as the cycle retention ratio.
[初期低温放电率][Initial low temperature discharge rate]
(低温试验)(low temperature test)
对实施了初期充放电的电池,在25℃下用0.2C的恒定电流恒定电压充电法充电至4.2V后,在-30℃下实施0.2C的恒定电流放电。将此时的放电容量作为初期低温容量,将初期低温容量相对于初期容量的比例作为初期低温放电率。The battery subjected to the initial charge and discharge was charged to 4.2V by a 0.2C constant current constant voltage charging method at 25°C, and then a 0.2C constant current discharge was performed at -30°C. The discharge capacity at this time was taken as the initial low-temperature capacity, and the ratio of the initial low-temperature capacity to the initial capacity was taken as the initial low-temperature discharge rate.
[循环后低温放电率][Low temperature discharge rate after cycle]
此外,对循环试验后的电池在25℃下以0.2C的恒定电流恒定电压充电法充电至4.2V,然后以0.2C的恒定电流放电至3.0V。对其进行3个循环,将其第3个循环的放电容量作为循环后容量。然后,对同一电池在25℃下以0.2C的恒定电流恒定电压充电法充电至4.2V,然后在-30℃下实施0.2C的恒定电流放电。将此时的放电容量作为循环后低温容量,将循环后低温容量相对于循环后容量的比例作为循环后低温放电率。In addition, the battery after the cycle test was charged to 4.2V at 25°C by a constant current and constant voltage charging method of 0.2C, and then discharged to 3.0V by a constant current of 0.2C. Three cycles were performed on it, and the discharge capacity at the third cycle was taken as the capacity after cycle. Then, the same battery was charged to 4.2V by a 0.2C constant current constant voltage charging method at 25°C, and then a 0.2C constant current discharge was performed at -30°C. The discharge capacity at this time was taken as the post-cycle low-temperature capacity, and the ratio of the post-cycle low-temperature capacity to the post-cycle capacity was taken as the post-cycle low-temperature discharge rate.
电解液[8]实施例1Electrolyte [8] Embodiment 1
在干燥氩氛围下,向碳酸亚乙酯(EC)、碳酸甲乙酯(EMC)和碳酸二甲酯(DMC)的混合物(体积比2∶5∶3)中以1mol/L加入LiPF6,并进行溶解,使该混合溶液中含有0.3质量%六甲基环三硅氧烷、1质量%乙烯基碳酸亚乙酯,制备非水电解液。使用该非水电解液用上述方法制作电池,并测定循环保持率、初期低温放电率和循环后低温放电率。结果如电解液[8]表1所示。Under a dry argon atmosphere, LiPF 6 was added at 1 mol/L to a mixture of ethylene carbonate (EC), ethylmethyl carbonate (EMC) and dimethyl carbonate (DMC) (volume ratio 2:5:3), It was dissolved, and the mixed solution contained 0.3% by mass of hexamethylcyclotrisiloxane and 1% by mass of vinylethylene carbonate to prepare a non-aqueous electrolytic solution. Using this non-aqueous electrolytic solution, a battery was produced by the above-mentioned method, and the cycle retention rate, initial low-temperature discharge rate and low-temperature discharge rate after cycle were measured. The results are shown in Table 1 of the electrolyte [8].
电解液[8]实施例2Electrolyte [8] Embodiment 2
除了使用苯基二甲基氟硅烷来替代六甲基环三硅氧烷制成非水电解液以外,与电解液[8]实施例1同样地制作电池,并测定循环保持率、初期低温放电率和循环后低温放电率。结果如电解液[8]表1所示。Except using phenyldimethylfluorosilane instead of hexamethylcyclotrisiloxane to make a non-aqueous electrolyte, a battery was made in the same manner as in Example 1 of the electrolyte [8], and the cycle retention rate and initial low-temperature discharge were measured. rate and low temperature discharge rate after cycling. The results are shown in Table 1 of the electrolyte [8].
电解液[8]实施例3Electrolyte [8] Example 3
除了使用氟磺酸甲酯来替代六甲基环三硅氧烷制成非水电解液以外,与电解液[8]实施例1同样地制作电池,并测定循环保持率、初期低温放电率和循环后低温放电率。结果如电解液[8]表1所示。Except using methyl fluorosulfonate instead of hexamethylcyclotrisiloxane to make non-aqueous electrolyte, make battery in the same way as electrolyte [8] embodiment 1, and measure cycle retention rate, initial low temperature discharge rate and Low temperature discharge rate after cycle. The results are shown in Table 1 of the electrolyte [8].
电解液[8]实施例4Electrolyte [8] Embodiment 4
除了使用根据Inorganic Nuclear Chemistry Letters(1969),5(7)的第581页~第582页中记载的方法制备的二氟磷酸锂来替代六甲基环三硅氧烷制成非水电解液以外,与电解液[8]实施例1同样地制作电池,并测定循环保持率、初期低温放电率和循环后低温放电率。结果如电解液[8]表1所示。In addition to using lithium difluorophosphate prepared according to the method described on pages 581 to 582 of Inorganic Nuclear Chemistry Letters (1969), 5(7) to replace hexamethylcyclotrisiloxane to make a non-aqueous electrolyte , A battery was produced in the same manner as in Example 1 of the electrolyte solution [8], and the cycle retention rate, initial low-temperature discharge rate and low-temperature discharge rate after cycle were measured. The results are shown in Table 1 of the electrolyte [8].
电解液[8]实施例5Electrolyte [8] Embodiment 5
除了使用1,3-丙磺内酯替代乙烯基碳酸亚乙酯制成非水电解液以外,与电解液[8]实施例1同样地制作电池,并测定循环保持率、初期低温放电率和循环后低温放电率。结果如电解液[8]表1所示。Except using 1,3-propane sultone instead of vinyl ethylene carbonate to make non-aqueous electrolyte, make battery in the same way as electrolyte [8] embodiment 1, and measure cycle retention rate, initial low temperature discharge rate and Low temperature discharge rate after cycle. The results are shown in Table 1 of the electrolyte [8].
电解液[8]实施例6Electrolyte [8] Embodiment 6
除了使用γ-丁内酯替代乙烯基碳酸亚乙酯制成非水电解液以外,与电解液[8]实施例1同样地制作电池,并测定循环保持率、初期低温放电率和循环后低温放电率。结果如电解液[8]表1所示。Except that γ-butyrolactone was used instead of vinyl ethylene carbonate to make non-aqueous electrolyte, a battery was made in the same manner as in Example 1 of electrolyte [8], and the cycle retention rate, initial low temperature discharge rate and low temperature after cycle were measured. discharge rate. The results are shown in Table 1 of the electrolyte [8].
电解液[8]实施例7Electrolyte [8] Embodiment 7
除了使用氟碳酸亚乙酯替代乙烯基碳酸亚乙酯制成非水电解液以外,与电解液[8]实施例1同样地制作电池,并测定循环保持率、初期低温放电率和循环后低温放电率。结果如电解液[8]表1所示。In addition to using fluoroethylene carbonate instead of vinyl ethylene carbonate to make a non-aqueous electrolyte, a battery was made in the same manner as in Example 1 of the electrolyte [8], and the cycle retention rate, initial low-temperature discharge rate, and low-temperature after cycle were measured. discharge rate. The results are shown in Table 1 of the electrolyte [8].
电解液[8]实施例8Electrolyte [8] Embodiment 8
除了使用1,3-丙磺内酯替代乙烯基碳酸亚乙酯制成非水电解液以外,与电解液[8]实施例4同样地制作电池,并测定循环保持率、初期低温放电率和循环后低温放电率。结果如电解液[8]表1所示。Except using 1,3-propane sultone instead of vinyl ethylene carbonate to make non-aqueous electrolyte, make battery in the same way as electrolyte [8] embodiment 4, and measure cycle retention rate, initial low temperature discharge rate and Low temperature discharge rate after cycle. The results are shown in Table 1 of the electrolyte [8].
电解液[8]实施例9Electrolyte [8] Embodiment 9
除了使用γ-丁内酯替代乙烯基碳酸亚乙酯制成非水电解液以外,与电解液[8]实施例4同样地制作电池,并测定循环保持率、初期低温放电率和循环后低温放电率。结果如电解液[8]表1所示。Except using gamma-butyrolactone instead of vinyl ethylene carbonate to make a non-aqueous electrolyte, make a battery in the same way as the electrolyte [8] Example 4, and measure the cycle retention rate, initial low-temperature discharge rate and low-temperature after cycle discharge rate. The results are shown in Table 1 of the electrolyte [8].
电解液[8]实施例10Electrolyte [8] Embodiment 10
除了使用氟碳酸亚乙酯替代乙烯基碳酸亚乙酯制成非水电解液以外,与电解液[8]实施例4同样地制作电池,并测定循环保持率、初期低温放电率和循环后低温放电率。结果如电解液[8]表1所示。In addition to using fluoroethylene carbonate instead of vinyl ethylene carbonate to make a non-aqueous electrolyte, a battery was made in the same manner as in Example 4 of the electrolyte [8], and the cycle retention rate, initial low-temperature discharge rate, and low-temperature after cycle were measured. discharge rate. The results are shown in Table 1 of the electrolyte [8].
电解液[8]比较例1Electrolyte [8] Comparative Example 1
除了使非水电解液不含有六甲基环三硅氧烷以外,与电解液[8]实施例1同样地制作电池,并测定循环保持率、初期低温放电率和循环后低温放电率。结果如电解液[8]表1所示。Except that the non-aqueous electrolyte solution does not contain hexamethylcyclotrisiloxane, a battery was produced in the same manner as the electrolyte solution [8] Example 1, and the cycle retention rate, initial low-temperature discharge rate and low-temperature discharge rate after cycle were measured. The results are shown in Table 1 of the electrolyte [8].
电解液[8]比较例2Electrolyte [8] Comparative Example 2
除了使非水电解液不含有乙烯基碳酸亚乙酯以外,与电解液[8]实施例1同样地制作电池,并测定循环保持率、初期低温放电率和循环后低温放电率。结果如电解液[8]表1所示。Except that the non-aqueous electrolytic solution does not contain vinyl ethylene carbonate, a battery was produced in the same manner as the electrolytic solution [8] Example 1, and the cycle retention rate, initial low-temperature discharge rate and low-temperature discharge rate after cycle were measured. The results are shown in Table 1 of the electrolyte [8].
电解液[8]比较例3Electrolyte [8] Comparative Example 3
除了使非水电解液不含有六甲基环三硅氧烷和乙烯基碳酸亚乙酯以外,与电解液[8]实施例1同样地制作电池,并测定循环保持率、初期低温放电率和循环后低温放电率。结果如电解液[8]表1所示。Except making the non-aqueous electrolytic solution not contain hexamethylcyclotrisiloxane and vinyl ethylene carbonate, a battery was produced in the same manner as the electrolytic solution [8] Example 1, and the cycle retention rate, initial low-temperature discharge rate and Low temperature discharge rate after cycle. The results are shown in Table 1 of the electrolyte [8].
电解液[8]实施例11Electrolyte [8] Example 11
<二次电池的制作-2><Production of secondary battery-2>
[正极的制作][making of positive electrode]
在N-甲基吡咯烷酮溶剂中混合90质量%作为正极活性物质的钴酸锂(LiCoOx)、5质量%的作为导电材料的乙炔黑和5质量%作为粘合剂的聚偏氟乙烯(PVdF),制成浆料。将得到的浆料涂布在厚度20μm的铝箔两面,并干燥,用压制机压延成厚度80μm,再将其切成宽52mm、长830mm大小,作为正极。其中,表面背面都在长度方向上设置50mm的未涂布部分,活性物质层的长度为780mm。90% by mass of lithium cobaltate (LiCoOx) as a positive electrode active material, 5% by mass of acetylene black as a conductive material, and 5% by mass of polyvinylidene fluoride (PVdF) as a binder were mixed in N-methylpyrrolidone solvent , to make slurry. The obtained slurry was coated on both sides of an aluminum foil with a thickness of 20 μm, dried, and rolled to a thickness of 80 μm by a press machine, and then cut into a size of 52 mm in width and 830 mm in length as a positive electrode. Among them, an uncoated portion of 50 mm was provided in the longitudinal direction on both the front and back sides, and the length of the active material layer was 780 mm.
[负极的制作][Production of Negative Electrode]
在98重量份人造石墨粉末KS-44(timcal公司制造,商品名)中添加100重量份作为增稠剂的羧甲基纤维素钠的水性分散液(羧甲基纤维素钠的浓度为1质量%)、2重量份作为粘合剂的苯乙烯-丁二烯橡胶的水性分散液(苯乙烯-丁二烯橡胶的浓度为50质量%),用分散器进行混合,制成浆料。将得到的浆料均匀涂布在作为负极集电体的厚度18μm的铜箔的两面,干燥后,用压制机压延成厚度为85μm,再将其切成宽56mm、长850mm的大小,作为负极。其中,表面背面都在长度方向上设置30mm的未涂布部分。In 98 parts by weight of artificial graphite powder KS-44 (manufactured by timcal company, trade name), add 100 parts by weight of an aqueous dispersion of sodium carboxymethyl cellulose as a thickener (the concentration of sodium carboxymethyl cellulose is 1 mass %), 2 parts by weight of an aqueous dispersion of styrene-butadiene rubber as a binder (the concentration of styrene-butadiene rubber is 50% by mass), and mixed with a disperser to prepare a slurry. The obtained slurry was uniformly coated on both sides of a copper foil with a thickness of 18 μm as the negative electrode current collector, and after drying, it was rolled to a thickness of 85 μm by a press, and then cut into a size of 56 mm wide and 850 mm long, as the negative electrode . Among them, an uncoated portion of 30 mm was provided in the longitudinal direction on both the front and back sides.
[电池的组装][assembling the battery]
将正极和负极与聚乙烯制的隔板一起重叠卷绕并使得正极与负极不直接接触,制成电极体。使正极和负极的端子露出外部地收纳在电池罐中。然后,向其中注入5mL后述的非水电解液后,进行铆接成型,制造18650型圆筒电池。二次电池的1个电池外壳中收纳的电池要素所具有的电容,即该电池的额定放电容量约为0.7安培小时(Ah),用10kHz交流法测定的直流电阻成分约为35毫欧(mΩ)。The positive electrode and the negative electrode were overlapped and wound together with a polyethylene separator so that the positive electrode and the negative electrode were not in direct contact to form an electrode body. The positive and negative terminals are housed in the battery can with the terminals exposed to the outside. Then, 5 mL of a non-aqueous electrolytic solution described later was injected thereinto, and then caulking was performed to manufacture a 18650-type cylindrical battery. The capacitance of the battery element contained in one battery case of the secondary battery, that is, the rated discharge capacity of the battery is about 0.7 ampere hours (Ah), and the DC resistance component measured by the 10kHz alternating current method is about 35 milliohms (mΩ ).
使用电解液[8]实施例1中使用的非水电解液作为非水电解液,制作圆筒电池,并测定循环保持率、初期低温放电率和循环后低温放电率。结果如电解液[8]表1所示。Use the non-aqueous electrolyte used in Example 1 of the electrolyte [8] as the non-aqueous electrolyte to make a cylindrical battery, and measure the cycle retention rate, the initial low-temperature discharge rate and the low-temperature discharge rate after the cycle. The results are shown in Table 1 of the electrolyte [8].
电解液[8]实施例12Electrolyte [8] Example 12
除了使用电解液[8]实施例4中使用的非水电解液作为非水电解液以外,与电解液[8]实施例11同样地制作圆筒电池,并测定循环保持率、初期低温放电率和循环后低温放电率。结果如电解液[8]表1所示。Except using the non-aqueous electrolytic solution used in the electrolytic solution [8] Example 4 as the non-aqueous electrolytic solution, a cylindrical battery was produced in the same manner as the electrolytic solution [8] Example 11, and the cycle retention rate and the initial low-temperature discharge rate were measured. and low temperature discharge rate after cycling. The results are shown in Table 1 of the electrolyte [8].
电解液[8]比较例4Electrolyte [8] Comparative Example 4
除了使用电解液[8]比较例1中使用的非水电解液作为非水电解液以外,与电解液[8]实施例11同样地制作圆筒电池,并测定循环保持率、初期低温放电率和循环后低温放电率。结果如电解液[8]表1所示。Except using the non-aqueous electrolytic solution used in the electrolytic solution [8] Comparative Example 1 as the non-aqueous electrolytic solution, a cylindrical battery was produced in the same manner as the electrolytic solution [8] Example 11, and the cycle retention rate and initial low-temperature discharge rate were measured. and low temperature discharge rate after cycling. The results are shown in Table 1 of the electrolyte [8].
电解液[8]比较例5Electrolyte [8] Comparative Example 5
除了使用电解液[8]比较例2中使用的非水电解液作为非水电解液以外,与电解液[8]实施例11同样地制作圆筒电池,并测定循环保持率、初期低温放电率和循环后低温放电率。结果如电解液[8]表1所示。Except using the non-aqueous electrolytic solution used in the electrolytic solution [8] Comparative Example 2 as the non-aqueous electrolytic solution, a cylindrical battery was produced in the same manner as the electrolytic solution [8] Example 11, and the cycle retention rate and initial low-temperature discharge rate were measured. and low temperature discharge rate after cycling. The results are shown in Table 1 of the electrolyte [8].
电解液[8]比较例6Electrolyte [8] Comparative Example 6
除了使用电解液[8]比较例3中使用的非水电解液作为非水电解液以外,与电解液[8]实施例11同样地制作圆筒电池,并测定循环保持率、初期低温放电率和循环后低温放电率。结果如电解液[8]表1所示。Except using the non-aqueous electrolytic solution used in the electrolytic solution [8] Comparative Example 3 as the non-aqueous electrolytic solution, a cylindrical battery was produced in the same manner as the electrolytic solution [8] Example 11, and the cycle retention rate and initial low-temperature discharge rate were measured. and low temperature discharge rate after cycling. The results are shown in Table 1 of the electrolyte [8].
电解液[8]表1Electrolyte [8] Table 1
[表61][Table 61]
由电解液[8]表1可知,若对每个方形电池(电解液[8]实施例1~10,电解液[8]比较例1~3中使用)、圆筒电池(电解液[8]实施例11、12,电解液[8]比较例4~6中使用)进行比较,则非水电解液中同时含有至少一种特定化合物A和至少一种特定化合物B的电解液[8]实施例的锂二次电池,与不含有这些任意一种的电解液[8]比较例的锂二次电池相比,循环保持率、初期低温放电率和循环后低温放电率都得到改善。From the electrolyte [8] Table 1, it can be seen that if each square battery (electrolyte [8] embodiment 1-10, electrolyte [8] used in comparative examples 1-3), cylindrical battery (electrolyte [8] ] Embodiment 11,12, electrolyte solution [8] used in comparative examples 4~6) is compared, then the electrolyte solution [8] that contains at least one specific compound A and at least one specific compound B in the non-aqueous electrolyte solution simultaneously The lithium secondary battery of the example, compared with the lithium secondary battery of the comparative example that does not contain any of these electrolyte solutions [8], has improved cycle retention rate, initial low-temperature discharge rate, and low-temperature discharge rate after cycle.
如上所述,本实施例与本比较例的方形电池的额定放电容量为3安培小时(Ah)以上,直流电阻成分为10毫欧(mΩ)以下。另一方面,本实施例与本比较例的圆筒电池的额定放电容量小于3安培小时(Ah),直流电阻成分大于10毫欧(mΩ)。即,本实施例、本比较例的方形电池与圆筒电池相比,电阻小,电容大。而且,电解液[8]实施例1相对于电解液[8]比较例1的输出功率上升程度,比电解液[8]实施例11相对于电解液[8]比较例4的输出功率上升程度大,在电容大的二次电池或直流电阻小的二次电池中,本发明的效果更大。As described above, the rated discharge capacity of the prismatic batteries of this example and this comparative example is 3 ampere hours (Ah) or more, and the DC resistance component is 10 milliohms (mΩ) or less. On the other hand, the rated discharge capacity of the cylindrical batteries of this embodiment and this comparative example is less than 3 ampere hours (Ah), and the DC resistance component is greater than 10 milliohms (mΩ). That is, the prismatic batteries of this example and this comparative example have lower resistance and higher capacitance than cylindrical batteries. Moreover, the output power increase degree of the electrolyte solution [8] Example 1 relative to the electrolyte solution [8] Comparative Example 1 is higher than the output power increase degree of the electrolyte solution [8] Example 11 relative to the electrolyte solution [8] Comparative Example 4 Large, the effect of the present invention is greater in a secondary battery with a large capacitance or a secondary battery with a small DC resistance.
电解液[9]<二次电池的制作>Electrolyte [9] <Production of secondary battery>
[正极的制作][making of positive electrode]
在N-甲基吡咯烷酮溶剂中混合90质量%作为正极活性物质的钴酸锂(LiCoO2)、5质量%的作为导电材料的乙炔黑和5质量%作为粘合剂的聚偏氟乙烯(PVdF),制成浆料。将得到的浆料涂布在厚度15μm的铝箔两面,并干燥,用压制机压延成厚度80μm,再将其切成具有宽100mm、长100mm大小的活性物质层和宽30mm的未涂布部分的形状,作为正极。Mix 90% by mass of lithium cobaltate (LiCoO 2 ) as a positive electrode active material, 5% by mass of acetylene black as a conductive material, and 5% by mass of polyvinylidene fluoride (PVdF ) to make slurry. The obtained slurry was coated on both sides of an aluminum foil with a thickness of 15 μm, dried, rolled to a thickness of 80 μm with a press, and cut into an active material layer having a width of 100 mm and a length of 100 mm and an uncoated portion of a width of 30 mm. shape, as the positive electrode.
[负极的制作][Production of Negative Electrode]
在98重量份人造石墨粉末KS-44(timcal公司制造,商品名)中添加100重量份作为增稠剂的羧甲基纤维素钠的水性分散液(羧甲基纤维素钠的浓度为1质量%)、2重量份作为粘合剂的苯乙烯-丁二烯橡胶的水性分散液(苯乙烯-丁二烯橡胶的浓度为50质量%),用分散器进行混合,制成浆料。将得到的浆料涂布在厚度10μm的铜箔两面,并干燥,用压制机压延成厚度为75μm,再将其切成具有宽104mm、长104mm大小的活性物质层和宽30mm的未涂布部分的形状,作为负极。In 98 parts by weight of artificial graphite powder KS-44 (manufactured by timcal company, trade name), add 100 parts by weight of an aqueous dispersion of sodium carboxymethyl cellulose as a thickener (the concentration of sodium carboxymethyl cellulose is 1 mass %), 2 parts by weight of an aqueous dispersion of styrene-butadiene rubber as a binder (the concentration of styrene-butadiene rubber is 50% by mass), and mixed with a disperser to prepare a slurry. The obtained slurry was coated on both sides of a copper foil with a thickness of 10 μm, dried, rolled to a thickness of 75 μm with a press, and then cut into an active material layer with a width of 104 mm and a length of 104 mm and an uncoated layer with a width of 30 mm. The shape of the part, as the negative pole.
[电池的组装][assembling the battery]
将32片正极和33片负极交替配置,各电极之间夹入多孔聚乙烯片隔板(厚度为25μm),进行叠层。此时,使正极活性物质面与负极活性物质面相对,并且不会露出负极活性物质面之外。将该正极和负极各自的未涂布部分焊接,制作集电片,将电极组密封在带有排气阀的电池罐(外部尺寸:120×110×10mm)中。然后,在装有电极组的电池罐中注入20mL非水电解液,使电极充分浸透,密封而制作方形电池。32 positive electrodes and 33 negative electrodes were arranged alternately, and a porous polyethylene sheet separator (thickness: 25 μm) was sandwiched between the electrodes for lamination. At this time, the surface of the positive electrode active material faces the surface of the negative electrode active material without protruding from the surface of the negative electrode active material. The respective uncoated parts of the positive and negative electrodes were welded to produce current collector sheets, and the electrode group was sealed in a battery can (outer dimensions: 120×110×10 mm) with a vent valve. Then, 20 mL of non-aqueous electrolytic solution was poured into the battery can containing the electrode group, the electrodes were fully soaked, and sealed to produce a square battery.
[电容和直流电阻成分][Capacitance and DC resistance components]
该电池的额定放电容量(1个电池外壳中收纳的电池要素所具有的电容)为高容量、约为6Ah,用10kHz交流法测定的直流电阻成分约为5毫欧。The rated discharge capacity of this battery (capacitance of the battery element contained in one battery case) is a high capacity of about 6Ah, and the DC resistance component measured by the 10kHz AC method is about 5 milliohms.
[电池的评价][evaluation of the battery]
(容量的测定方法)(measurement method of capacity)
对未经充放电循环的新电池,在25℃、4.2V~3.0V的电压范围下,以0.2C的电流值(将1小时放电额定容量的电流值作为1C,所述额定容量取决于1小时率(one-hour-rate)的放电容量,下同)进行5个循环的初期充放电。将此时的第5个循环的0.2C放电容量作为容量。For a new battery that has not been charged and discharged, at 25°C and a voltage range of 4.2V to 3.0V, with a current value of 0.2C (the current value of the rated capacity of 1 hour discharge is taken as 1C, and the rated capacity depends on 1 One-hour-rate (one-hour-rate) discharge capacity, the same below) was performed for 5 cycles of initial charge and discharge. The 0.2C discharge capacity at the fifth cycle at this time was taken as the capacity.
(保存后5C放电容量)(5C discharge capacity after storage)
在60℃的高温环境下进行保存试验。将事先在25℃的环境下用恒定电流恒定电压法充电至充电上限电压4.2V的电池在60℃下保存1个月。对于保存后的电池,在25℃的环境下进行速度试验。即,将事先在25℃的环境下用恒定电流恒定电压法充电至充电上限电压4.2V的电池以相当于5C的恒定电流值进行放电,作为保存后5C放电容量。The storage test was carried out in a high-temperature environment of 60°C. The battery previously charged to a charging upper limit voltage of 4.2 V by a constant current constant voltage method in an environment of 25° C. was stored at 60° C. for one month. For the stored batteries, perform a speed test at 25°C. That is, a battery previously charged to a charging upper limit voltage of 4.2V by the constant current and constant voltage method in an environment of 25°C was discharged at a constant current value equivalent to 5C, which was used as the 5C discharge capacity after storage.
(过充电试验)(overcharge test)
在25℃环境下进行过充电试验。由放电状态(3V)以3C的恒定电流进行充电,观测其举动。其中,“阀工作”表示排气阀工作并释放电解液成分的现象,“破裂”表示电池容器以猛烈的势头破裂、内容物强制释放的现象。The overcharge test was carried out in an environment of 25°C. It was charged with a constant current of 3C from the discharged state (3V), and its behavior was observed. Among them, "valve operation" refers to the phenomenon in which the exhaust valve operates to release the electrolyte components, and "rupture" refers to the phenomenon in which the battery container ruptures violently and the contents are forcibly released.
电解液[9]实施例1Electrolyte [9] Embodiment 1
在干燥氩氛围下,向碳酸亚乙酯(EC)、碳酸二甲酯(DMC)、碳酸甲乙酯(EMC)的混合物(体积比3∶3∶4)中以1mol/L加入六氟磷酸锂(LiPF6),并进行溶解,在该混合溶液中混合1质量%的量的环己基苯(CHB)和0.3质量%的量的六甲基环三硅氧烷,制备非水电解液。使用该非水电解液用上述方法制作电池,并测定保存后5C放电容量。结果如电解液[9]表1所示。Under a dry argon atmosphere, add lithium hexafluorophosphate (LiPF 6 ), and dissolve, mix the cyclohexylbenzene (CHB) of the amount of 1 mass % and the hexamethylcyclotrisiloxane of the amount of 0.3 mass % in this mixed solution, prepare non-aqueous electrolytic solution. Using this non-aqueous electrolytic solution, a battery was produced by the above method, and the 5C discharge capacity after storage was measured. The results are shown in Table 1 of the electrolyte [9].
电解液[9]实施例2Electrolyte [9] Embodiment 2
使用混合了苯基二甲基氟硅烷来替代电解液[9]实施例1中的六甲基环三硅氧烷而制备的非水电解液,制作电池,并测定保存后5C放电容量。结果如电解液[9]表1所示。A non-aqueous electrolytic solution prepared by mixing phenyldimethylfluorosilane instead of hexamethylcyclotrisiloxane in Example 1 of the electrolytic solution [9] was used to fabricate a battery, and the 5C discharge capacity after storage was measured. The results are shown in Table 1 of the electrolyte [9].
电解液[9]实施例3Electrolyte [9] Embodiment 3
使用混合了甲磺酸三甲基甲硅烷基酯来替代电解液[9]实施例1中的六甲基环三硅氧烷而制备的非水电解液,制作电池,并测定保存后5C放电容量。结果如电解液[9]表1所示。Use the non-aqueous electrolyte prepared by mixing trimethylsilyl methanesulfonate to replace the hexamethylcyclotrisiloxane in the electrolyte [9] Example 1, make a battery, and measure the 5C discharge after storage capacity. The results are shown in Table 1 of the electrolyte [9].
电解液[9]实施例4Electrolyte [9] Embodiment 4
使用混合了根据Inorganic Nuclear Chemistry Letters(1969),5(7)的第581页~第582页中记载的方法制备的二氟磷酸锂来替代电解液[9]实施例1中的六甲基环三硅氧烷而制备的非水电解液,制作电池,并测定保存后5C放电容量。结果如电解液[9]表1所示。Use lithium difluorophosphate prepared according to the method described in Inorganic Nuclear Chemistry Letters (1969), 5 (7) on page 581 to page 582 to replace the hexamethyl ring in the electrolyte [9] Example 1 The non-aqueous electrolyte solution prepared from trisiloxane was used to make batteries, and the 5C discharge capacity after storage was measured. The results are shown in Table 1 of the electrolyte [9].
电解液[9]实施例5Electrolyte [9] Embodiment 5
使用混合了联苯来替代电解液[9]实施例1中的环己基苯而制备的非水电解液,制作电池,并测定保存后5C放电容量。结果如电解液[9]表1所示。A non-aqueous electrolytic solution prepared by mixing biphenyl instead of cyclohexylbenzene in Example 1 of the electrolytic solution [9] was used to fabricate a battery, and the 5C discharge capacity after storage was measured. The results are shown in Table 1 of the electrolyte [9].
电解液[9]实施例6Electrolyte [9] Embodiment 6
使用混合了苯基二甲基氟硅烷来替代电解液[9]实施例5中的六甲基环三硅氧烷而制备的非水电解液,制作电池,并测定保存后5C放电容量。结果如电解液[9]表1所示。A non-aqueous electrolytic solution prepared by mixing phenyldimethylfluorosilane instead of hexamethylcyclotrisiloxane in Example 5 of the electrolytic solution [9] was used to fabricate a battery, and the 5C discharge capacity after storage was measured. The results are shown in Table 1 of the electrolyte [9].
电解液[9]实施例7Electrolyte [9] Embodiment 7
使用混合了甲磺酸三甲基甲硅烷基酯来替代电解液[9]实施例5中的六甲基环三硅氧烷而制备的非水电解液,制作电池,并测定保存后5C放电容量。结果如电解液[9]表1所示。Use the non-aqueous electrolyte prepared by mixing trimethylsilyl methanesulfonate to replace the hexamethylcyclotrisiloxane in the electrolyte [9] embodiment 5, make the battery, and measure the 5C discharge after storage capacity. The results are shown in Table 1 of the electrolyte [9].
电解液[9]实施例8Electrolyte [9] Embodiment 8
使用混合了根据Inorganic Nuclear Chemistry Letters(1969),5(7)的第581页~第582页中记载的方法制备的二氟磷酸锂来替代电解液[9]实施例5中的六甲基环三硅氧烷而制备的非水电解液,制作电池,并测定保存后5C放电容量。结果如电解液[9]表1所示。Use lithium difluorophosphate prepared according to the method described in Inorganic Nuclear Chemistry Letters (1969), 5 (7) on page 581 to page 582 to replace the hexamethyl ring in the electrolyte [9] Example 5 The non-aqueous electrolyte solution prepared from trisiloxane was used to make batteries, and the 5C discharge capacity after storage was measured. The results are shown in Table 1 of the electrolyte [9].
电解液[9]实施例9Electrolyte [9] Embodiment 9
使用混合了叔戊基苯来替代电解液[9]实施例1中的环己基苯而制备的非水电解液,制作电池,并测定保存后5C放电容量。结果如电解液[9]表1所示。A non-aqueous electrolytic solution prepared by mixing tert-amylbenzene instead of cyclohexylbenzene in Example 1 of the electrolytic solution [9] was used to fabricate a battery, and the 5C discharge capacity after storage was measured. The results are shown in Table 1 of the electrolyte [9].
电解液[9]实施例10Electrolyte [9] Embodiment 10
使用混合了间三联苯的含量为3.7质量%、部分氢化率为42%的“间三联苯的部分氢化物”来替代电解液[9]实施例1中的环己基苯而制备的非水电解液,制作电池,并测定保存后5C放电容量。结果如电解液[9]表1所示。另外,电解液[9]实施例中使用的间三联苯的部分氢化物,使用以间三联苯为原料,在铂、钯或镍类催化剂的共存下,使其在高温加压条件下与氢气反应而得到的物质。此外,部分氢化率由通过气相色谱分析求得的间三联苯的部分氢化物的构成成分的组成比取平均值而确定。间三联苯的含量也由该气相色谱分析值求得。A non-aqueous electrolytic solution prepared by using a "partial hydrogenated product of m-terphenyl" mixed with a m-terphenyl content of 3.7% by mass and a partial hydrogenation rate of 42% instead of cyclohexylbenzene in the electrolyte [9] Example 1 solution, make a battery, and measure the 5C discharge capacity after storage. The results are shown in Table 1 of the electrolyte [9]. In addition, the partial hydrogenation of m-terphenyl used in the embodiment of the electrolyte [9] uses m-terphenyl as a raw material, and under the coexistence of platinum, palladium or nickel catalyst, it is mixed with hydrogen under high temperature and pressure. The substance obtained by the reaction. In addition, the partial hydrogenation rate was determined by taking the average of the composition ratios of the constituent components of the partially hydrogenated product of m-terphenyl obtained by gas chromatography analysis. The content of m-terphenyl was also obtained from the gas chromatographic analysis value.
电解液[9]比较例1Electrolyte [9] Comparative Example 1
使用电解液[9]实施例1中未混合六甲基环三硅氧烷而制备的非水电解液来制作电池,并测定5C放电容量。结果如电解液[9]表1所示。A battery was fabricated using the non-aqueous electrolyte solution prepared without mixing hexamethylcyclotrisiloxane in Example 1 of the electrolyte solution [9], and the 5C discharge capacity was measured. The results are shown in Table 1 of the electrolyte [9].
电解液[9]比较例2Electrolyte [9] Comparative Example 2
使用电解液[9]实施例5中未混合六甲基环三硅氧烷而制备的非水电解液来制作电池,并测定5C放电容量。结果如电解液[9]表1所示。A battery was fabricated using the non-aqueous electrolyte solution prepared without mixing hexamethylcyclotrisiloxane in Example 5 of the electrolyte solution [9], and the 5C discharge capacity was measured. The results are shown in Table 1 of the electrolyte [9].
电解液[9]比较例3Electrolyte [9] Comparative Example 3
使用电解液[9]实施例9中未混合六甲基环三硅氧烷而制备的非水电解液制造电池,对5C放电容量进行测定。结果如电解液[9]表1所示。A battery was manufactured using the non-aqueous electrolyte solution prepared without mixing hexamethylcyclotrisiloxane in Example 9 of the electrolyte solution [9], and the 5C discharge capacity was measured. The results are shown in Table 1 of the electrolyte [9].
电解液[9]比较例4Electrolyte [9] Comparative Example 4
使用电解液[9]实施例10中未混合六甲基环三硅氧烷而制备的非水电解液来制作电池,并测定5C放电容量。结果如电解液[9]表1所示。A battery was fabricated using the non-aqueous electrolyte solution prepared without mixing hexamethylcyclotrisiloxane in Example 10 of the electrolyte solution [9], and the 5C discharge capacity was measured. The results are shown in Table 1 of the electrolyte [9].
电解液[9]比较例5Electrolyte [9] Comparative Example 5
使用电解液[9]实施例1中未混合六甲基环三硅氧烷、CHB的任意一种而制备的非水电解液来制作电池,并测定5C放电容量。结果如电解液[9]表1所示。A battery was fabricated using the non-aqueous electrolytic solution prepared without mixing either hexamethylcyclotrisiloxane or CHB in Example 1 of the electrolytic solution [9], and the 5C discharge capacity was measured. The results are shown in Table 1 of the electrolyte [9].
电解液[9]表1Electrolyte [9] Table 1
[表62][Table 62]
由电解液[9]表1可知,在非水电解液中含有过充电防止剂和特定化合物的电解液[9]实施例1~10的锂二次电池,不言而喻地在过充电试验时可以避免电池破裂,在保存后的大电流(5C)放电试验中也表现出比电解液[9]比较例1~4的锂二次电池更优异的特性。该特性接近不含有过充电防止剂的电解液[9]比较例5的锂二次电池,可知在提高过充电安全性的同时,实用价值非常高。It can be seen from Table 1 of the electrolyte solution [9] that the lithium secondary batteries of the electrolyte solution [9] Examples 1 to 10 containing an overcharge preventing agent and a specific compound in the non-aqueous electrolyte solution are self-evident in the overcharge test. It can avoid battery rupture, and it also shows more excellent characteristics than the lithium secondary batteries of the electrolyte [9] Comparative Examples 1-4 in the high-current (5C) discharge test after storage. This characteristic is close to that of the lithium secondary battery of Comparative Example 5 of the electrolytic solution [9] that does not contain an overcharge preventing agent, and it can be seen that the practical value is very high while improving overcharge safety.
如上所述,通过使用具有下述特征的在非水溶剂中混合锂盐而形成的二次电池用非水电解液,可以同时满足高的大电流放电特性和过充电安全性,所述二次电池用非水电解液的特征是:含有过充电防止剂,还含有选自下述物质中的至少一种化合物,并且其在全部非水电解液中的含量为10ppm以上,所述物质包括:通式(1)表示的环状硅氧烷化合物、通式(2)表示的氟硅烷化合物、通式(3)表示的化合物、分子内具有S-F键的化合物、硝酸盐、亚硝酸盐、单氟磷酸盐、二氟磷酸盐、乙酸盐和丙酸盐。特别是对于1个电池外壳中收纳的电池要素所具有的电容为3安培小时(Ah)以上的以往难以同时满足这2个要素的电池,可以具有高实用性。As described above, high large-current discharge characteristics and overcharge safety can be simultaneously satisfied by using a non-aqueous electrolyte solution for a secondary battery formed by mixing a lithium salt in a non-aqueous solvent having the following characteristics. The non-aqueous electrolytic solution for batteries is characterized by: containing an overcharge preventing agent, and at least one compound selected from the following substances, and its content in all non-aqueous electrolytic solutions is more than 10ppm, and said substances include: Cyclic siloxane compound represented by general formula (1), fluorosilane compound represented by general formula (2), compound represented by general formula (3), compound having S-F bond in the molecule, nitrate, nitrite, mono Fluorophosphates, difluorophosphates, acetates and propionates. In particular, it is highly practical for a battery in which the capacity of the battery element housed in one battery case is 3 ampere-hours (Ah) or more, and it has been difficult to satisfy these two elements at the same time.
结构[1]~[5]实施例1Structure [1]~[5] Embodiment 1
《非水电解液的制作》"Preparation of Non-Aqueous Electrolyte"
在干燥氩氛围下,在纯化的碳酸亚乙酯(EC)、碳酸二甲酯(DMC)和碳酸甲乙酯(EMC)的体积比3∶3∶4的混合溶剂中以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。另外,使其含有0.3质量%的六甲基环三硅氧烷。Under a dry argon atmosphere, in a mixed solvent of purified ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) at a volume ratio of 3:3:4 at a concentration of 1 mol/L Dissolve well-dried lithium hexafluorophosphate (LiPF 6 ). In addition, 0.3% by mass of hexamethylcyclotrisiloxane was contained.
《正极的制作》"Positive Production"
正极活性物质为用下述方法合成的锂过渡金属复合氧化物,以组成式LiMn0.33Ni0.33Co0.33O2表示。以Mn∶Ni∶Co=1∶1∶1的摩尔比称量作为锰原料的Mn3O4、作为镍原料的NiO以及作为钴原料的Co(OH)2,向其中加入纯水,制成浆料,边搅拌边使用循环式介质搅拌型湿式珠磨机将浆料中的固体成分湿式粉碎至中值粒径0.2μm。The positive electrode active material is a lithium transition metal composite oxide synthesized by the following method, represented by the composition formula LiMn 0.33 Ni 0.33 Co 0.33 O 2 . Mn 3 O 4 as a manganese raw material, NiO as a nickel raw material, and Co(OH) 2 as a cobalt raw material were weighed at a molar ratio of Mn:Ni:Co=1:1:1, and pure water was added thereto to prepare For the slurry, wet pulverize the solid components in the slurry to a median particle size of 0.2 μm by using a circulating medium agitation type wet bead mill while stirring.
通过喷雾干燥器将浆料进行喷雾干燥,得到仅仅包含锰原料、镍原料、钴原料的粒径约5μm的大致球状的造粒粒子。在得到的造粒粒子中添加中值粒径3μm的LiOH粉末,并使Li的摩尔数与Mn、Ni和Co的总摩尔数之比为1.05,用高速混合机进行混合,得到镍原料、钴原料、锰原料的造粒粒子和锂原料的混合粉末。在空气流通下、950℃对该混合粉末烧制12小时(升降温速度为5℃/分钟)后,粉碎,通过网眼45μm的筛子,得到正极活性物质。该正极活性物质的BET比表面积为1.2m2/g,平均一次粒径为0.8μm,中值粒径d50为4.4μm,振实密度为1.6g/cm3。The slurry was spray-dried by a spray dryer to obtain roughly spherical granulated particles having a particle diameter of about 5 μm and containing only manganese raw materials, nickel raw materials, and cobalt raw materials. LiOH powder with a median diameter of 3 μm was added to the obtained granulated particles, and the ratio of the number of moles of Li to the total number of moles of Mn, Ni, and Co was 1.05, and mixed with a high-speed mixer to obtain nickel raw materials, cobalt Raw material, granulated particles of manganese raw material and mixed powder of lithium raw material. The mixed powder was calcined at 950° C. for 12 hours under air circulation (the heating and cooling rate was 5° C./min), then pulverized and passed through a sieve with a mesh size of 45 μm to obtain a positive electrode active material. The positive electrode active material had a BET specific surface area of 1.2 m 2 /g, an average primary particle size of 0.8 μm, a median particle size d 50 of 4.4 μm, and a tap density of 1.6 g/cm 3 .
在N-甲基吡咯烷酮溶剂中混合90质量%作为正极活性物质的钴酸锂(LiCoO2)、5质量%的作为导电材料的乙炔黑和5质量%作为粘合剂的聚偏氟乙烯(PVdF),制成浆料。将得到的浆料涂布在厚度15μm的铝箔两面,并干燥,用压制机压延成厚度81μm,再将其切成具有宽100mm、长100mm大小的活性物质层和宽30mm的未涂布部分的形状,作为正极。正极活性物质层的密度为2.35g/cm3,(一面的正极活性物质层的厚度)/(集电体的厚度)为2.2,L/(2×S2)为0.2。Mix 90% by mass of lithium cobaltate (LiCoO 2 ) as a positive electrode active material, 5% by mass of acetylene black as a conductive material, and 5% by mass of polyvinylidene fluoride (PVdF ) to make slurry. The obtained slurry was coated on both sides of an aluminum foil with a thickness of 15 μm, dried, rolled to a thickness of 81 μm with a press, and cut into an active material layer having a width of 100 mm and a length of 100 mm and an uncoated portion of a width of 30 mm. shape, as the positive electrode. The density of the positive electrode active material layer was 2.35 g/cm 3 , (thickness of positive electrode active material layer on one surface)/(thickness of current collector) was 2.2, and L/(2×S 2 ) was 0.2.
《负极的制作》"The Making of Negative Pole"
为了防止作为粒状碳质物质的市售的天然石墨粉末中混入粗大粒子,使用ASTM400目的筛子反复筛5次。将这样得到的负极材料作为碳质物质(A)。In order to prevent the commercially available natural graphite powder, which is a granular carbonaceous material, from being mixed with coarse particles, the sieve was repeatedly sieved 5 times using an ASTM400 mesh sieve. The negative electrode material thus obtained is referred to as a carbonaceous substance (A).
在碳质物质(A)中混合石脑油热解时得到的石油类重油,在非活性气体中实施1300℃的碳化处理,然后,通过对烧结物进行分级处理得到复合碳质物质,作为负极活性物质,所述复合碳质物质是在碳质物质(A)粒子表面上包覆了具有不同结晶性的碳质物质的复合碳质物质。分级处理时,为了防止混入粗大粒子,使用ASTM400目的筛子反复筛5次。从残炭率可以确定,得到的负极活性物质粉末被相对于95重量份石墨为5重量的低结晶性碳质物质包覆。负极活性物质的物性如结构[1]~[5]表1所示。Petroleum heavy oil obtained during the pyrolysis of naphtha is mixed with the carbonaceous substance (A), carbonized at 1300°C in an inert gas, and then the composite carbonaceous substance is obtained by classifying the sintered product as a negative electrode In the active material, the composite carbonaceous substance is a composite carbonaceous substance in which carbonaceous substances having different crystallinity are coated on the surface of the carbonaceous substance (A) particle. During the classification process, in order to prevent the mixing of coarse particles, use an ASTM400 mesh sieve to repeatedly sieve 5 times. It was confirmed from the carbon residue rate that the obtained negative electrode active material powder was coated with 5 parts by weight of low-crystalline carbonaceous material relative to 95 parts by weight of graphite. The physical properties of the negative electrode active material are shown in Table 1 with structures [1]-[5].
结构[1]~[5]表1Structure [1]~[5] Table 1
[表63][Table 63]
在98重量份上述负极活性物质中添加100重量份作为增稠剂的羧甲基纤维素钠的水性分散液(羧甲基纤维素钠的浓度为1质量%)、2重量份作为粘合剂的苯乙烯-丁二烯橡胶的水性分散液(苯乙烯-丁二烯橡胶的浓度为50质量%),用分散器进行混合,制成浆料。将得到的浆料涂布在厚度10μm的压延铜箔的两面,并干燥,用压制机压延成厚度为75μm,再将其切成具有宽104mm、长104mm大小的活性物质层和宽30mm的未涂布部分的形状,作为负极。此时的负极活性物质的密度为1.35g/cm3,L/(2×S2)为0.19。Add 100 parts by weight of an aqueous dispersion of sodium carboxymethyl cellulose as a thickener (the concentration of sodium carboxymethyl cellulose is 1% by mass) and 2 parts by weight of a binder in 98 parts by weight of the above-mentioned negative electrode active material. The aqueous dispersion of styrene-butadiene rubber (the concentration of styrene-butadiene rubber is 50% by mass) was mixed with a disperser to prepare a slurry. The obtained slurry was coated on both sides of a rolled copper foil with a thickness of 10 μm, dried, and rolled to a thickness of 75 μm with a press machine, and then cut into an active material layer with a width of 104 mm and a length of 104 mm and an uncoated copper foil with a width of 30 mm. The shape of the coated part, as the negative electrode. The density of the negative electrode active material at this time was 1.35 g/cm 3 , and L/(2×S 2 ) was 0.19.
《电池的制作》"Battery Making"
将32片正极和33片负极交替配置,各电极之间夹入多孔聚乙烯片隔板(厚度为25μm),进行叠层。此时,使正极活性物质面与负极活性物质面相对,并且不会露出负极活性物质面之外。将该正极和负极各自的未涂布部分之间捆束,进行点焊接,制作集电片,将电极组密封在铝制的电池罐(外部尺寸:120×110×10mm)中。作为电池罐,使用在盖部分具有正极和负极的集电端子、压力释放阀、非水电解液的注入口的电池罐。集电片和集电端子通过点熔接连接。然后,在装有电极组的电池罐中注入20mL非水电解液,使电极充分浸透,密封注入口,制作电池。正极的电极面积的总和相对于电池的外壳表面积总和之比为20.5,2×S1/T为264,电极组占有率为0.54。32 positive electrodes and 33 negative electrodes were arranged alternately, and a porous polyethylene sheet separator (thickness: 25 μm) was sandwiched between the electrodes for lamination. At this time, the surface of the positive electrode active material faces the surface of the negative electrode active material without protruding from the surface of the negative electrode active material. The uncoated parts of the positive and negative electrodes were bundled and spot-welded to form current collector sheets, and the electrode group was sealed in an aluminum battery can (outer dimensions: 120×110×10 mm). As the battery can, a battery can having positive and negative current collector terminals, a pressure release valve, and a non-aqueous electrolytic solution inlet in the lid portion was used. The collector tabs and collector terminals are connected by spot welding. Then, inject 20 mL of non-aqueous electrolytic solution into the battery can containing the electrode group to fully soak the electrodes, seal the injection port, and manufacture a battery. The ratio of the total electrode area of the positive electrode to the total surface area of the battery case was 20.5, 2×S 1 /T was 264, and the electrode group occupancy ratio was 0.54.
《电池的评价》"Battery Evaluation"
(电池容量的测定方法)(Measuring method of battery capacity)
对未经充放电的新电池,在25℃、4.1V~3.0V的电压范围下以电流值0.2C(将1小时放电额定容量的电流值作为1C,所述额定容量取决于1小时率(one-hour-rate)的放电容量,下同)进行5个循环的初期充放电。将此时的第5个循环的0.2C放电容量作为初期容量。电池评价的结果如结构[1]~[5]表2所示。For a new battery that has not been charged and discharged, at 25°C and a voltage range of 4.1V to 3.0V, use a current value of 0.2C (take the current value of the rated capacity of 1 hour discharge as 1C, and the rated capacity depends on the 1 hour rate ( One-hour-rate) discharge capacity, the same below) for 5 cycles of initial charge and discharge. The 0.2C discharge capacity at the fifth cycle at this time was taken as the initial capacity. The results of battery evaluation are shown in Table 2 for structures [1] to [5].
(初期输出功率的测定方法)(Measurement method of initial output power)
在25℃环境下通过0.2C的恒定电流进行150分钟充电,分别以0.1C、0.3C、1.0C、3.0C、10.0C放电10秒,测定第10秒的电压。将由电流-电压直线与下限电压(3V)围成的三角形的面积作为输出功率(W)。电池评价的结果如结构[1]~[5]表2所示。Charge at a constant current of 0.2C for 150 minutes at 25°C, discharge at 0.1C, 0.3C, 1.0C, 3.0C, and 10.0C for 10 seconds, and measure the voltage at 10 seconds. The area of the triangle surrounded by the current-voltage straight line and the lower limit voltage (3V) is taken as the output power (W). The results of battery evaluation are shown in Table 2 for structures [1] to [5].
(初期串联电阻的测定方法)(Measurement method of initial series resistance)
在25℃环境下通过0.2C的恒定电流进行150分钟充电,施加10kHz的交流,测定阻抗,作为直流电阻。电池评价的结果如结构[1]~[5]表2所示。Charge at a constant current of 0.2 C for 150 minutes in an environment of 25° C., apply an alternating current of 10 kHz, measure the impedance, and use it as a direct current resistance. The results of battery evaluation are shown in Table 2 for structures [1] to [5].
(循环试验(耐久后电池容量和耐久后输出功率的测定方法))(Cycle test (measuring method of battery capacity after endurance and output power after endurance))
在被视为锂二次电池的实际使用上限温度的60℃的高温环境下进行循环试验。用2C的恒定电流恒定电压法充电至充电上限电压4.1V后,以2C的恒定电流放电至放电终止电压3.0V,将此充放电循环作为1个循环,反复进行该循环直至500循环。对于循环试验结束后的电池,在25℃环境下以电流值0.2C进行3个循环充放电,将其第3个循环的0.2C放电容量作为耐久后电池容量。此外,对于循环试验结束后的电池,进行输出功率测定,作为耐久后输出功率,对串联电阻进行测定,作为耐久后串联电阻。电池评价的结果如结构[1]~[5]表2所示。The cycle test was performed in a high-temperature environment of 60° C., which is regarded as the practical upper limit temperature of the lithium secondary battery. After charging to the charging upper limit voltage of 4.1V by the constant current and constant voltage method of 2C, discharge to the end-of-discharge voltage of 3.0V by a constant current of 2C. This charge-discharge cycle is regarded as one cycle, and this cycle is repeated until 500 cycles. For the battery after the cycle test, it was charged and discharged for 3 cycles with a current value of 0.2C in an environment of 25°C, and the 0.2C discharge capacity of the third cycle was taken as the battery capacity after endurance. In addition, for the battery after the cycle test, the output power was measured as the output power after endurance, and the series resistance was measured as the series resistance after endurance. The results of battery evaluation are shown in Table 2 for structures [1] to [5].
(过充电试验)(overcharge test)
在25℃环境下进行过充电试验。由放电状态(3V)以3C的恒定电流进行充电,观测其举动。其中,“阀工作”表示排气阀工作并释放电解液成分的现象,“破裂”表示电池容器以猛烈的势头破裂、内容物强制释放的现象。电池评价的结果如结构[1]~[5]表2所示。The overcharge test was carried out in an environment of 25°C. It was charged with a constant current of 3C from the discharged state (3V), and its behavior was observed. Among them, "valve operation" refers to the phenomenon in which the exhaust valve operates to release the electrolyte components, and "rupture" refers to the phenomenon in which the battery container ruptures violently and the contents are forcibly released. The results of battery evaluation are shown in Table 2 for structures [1] to [5].
结构[1]~[5]实施例2Structure [1]~[5] Embodiment 2
使非水电解液中含有0.3质量%的甲磺酸三甲基甲硅烷基酯来替代结构[1]~[5]实施例1中的六甲基环三硅氧烷,除此之外,与结构[1]~[5]实施例1同样地制作电池,并同样地进行电池评价。结果如结构[1]~[5]表2所示。The trimethylsilyl methanesulfonate containing 0.3% by mass in the non-aqueous electrolytic solution replaces the hexamethylcyclotrisiloxane in the structure [1]~[5] embodiment 1, in addition, Batteries were produced in the same manner as in Example 1 of structures [1] to [5], and battery evaluations were performed in the same manner. The results are shown in Table 2 of structures [1]-[5].
结构[1]~[5]实施例3Structure [1]~[5] Embodiment 3
使非水电解液中含有0.3质量%的苯基二甲基氟硅烷来替代结构[1]~[5]实施例1中的六甲基环三硅氧烷,除此之外,与结构[1]~[5]实施例1同样地制作电池,并同样地进行电池评价。结果如结构[1]~[5]表2所示。The non-aqueous electrolyte contains 0.3% by mass of phenyldimethylfluorosilane to replace the hexamethylcyclotrisiloxane in the structure [1]~[5] embodiment 1, in addition, with the structure [ 1] to [5] A battery was produced in the same manner as in Example 1, and battery evaluation was performed in the same manner. The results are shown in Table 2 of structures [1]-[5].
结构[1]~[5]实施例4Structure [1]~[5] Embodiment 4
使非水电解液中含有0.3质量%的二氟磷酸锂来替代结构[1]~[5]实施例1中的六甲基环三硅氧烷,除此之外,与结构[1]~[5]实施例1同样地制作电池,并同样地进行电池评价。结果如结构[1]~[5]表2所示。Make the non-aqueous electrolytic solution contain 0.3 mass % lithium difluorophosphate to replace the hexamethylcyclotrisiloxane in the structure [1]~[5] embodiment 1, in addition, with the structure [1]~ [5] A battery was produced in the same manner as in Example 1, and battery evaluation was performed in the same manner. The results are shown in Table 2 of structures [1]-[5].
结构[1]~[5]比较例1Structure [1]~[5] Comparative Example 1
使结构[1]~[5]实施例1中的非水电解液中不含有六甲基环三硅氧烷,除此之外,与结构[1]~[5]实施例1同样地制作电池,并同样地进行电池评价。结果如结构[1]~[5]表2所示。Make structure [1]~[5] in the non-aqueous electrolytic solution in the embodiment 1 of [5] do not contain hexamethylcyclotrisiloxane, except that, make in the same way as structure [1]~[5] embodiment 1 battery, and perform battery evaluation in the same way. The results are shown in Table 2 of structures [1]-[5].
结构[1]~[5]表2Structure [1]~[5] Table 2
[表64][Table 64]
结构[1]~[5]实施例5Structure [1]~[5] Embodiment 5
《正极的制作》"Positive Production"
使用与结构[1]~[5]实施例1相同的正极活性物质,与结构[1]~[5]实施例1同样地制备浆料。将得到的浆料涂布在厚度15μm的铝箔的两面上,并进行干燥,用压制机压延成厚度81μm,再将其切成宽100mm、长3200mm大小的正极活性物质层,并具有在长度方向上每间隔20mm具有宽6mm的未涂布部分的形状,作为正极。正极活性物质层的密度为2.35g/cm3,(一面的正极活性物质层的厚度)/(集电体的厚度)为2.2。正极的宽度方向的长度相对于长度方向的长度的比率为32。Slurry was prepared in the same manner as in Example 1 of Structures [1] to [5] using the same positive electrode active material as in Example 1 of Structures [1] to [5]. The obtained slurry is coated on both sides of an aluminum foil with a thickness of 15 μm, dried, rolled into a thickness of 81 μm with a press, and then cut into a positive electrode active material layer with a width of 100 mm and a length of 3200 mm, and has The shape of an uncoated portion having a width of 6 mm at intervals of 20 mm was used as a positive electrode. The density of the positive electrode active material layer was 2.35 g/cm 3 , and (thickness of the positive electrode active material layer on one surface)/(thickness of the current collector) was 2.2. The ratio of the length in the width direction to the length in the longitudinal direction of the positive electrode was 32.
《负极的制作》"The Making of Negative Pole"
使用与结构[1]~[5]实施例1相同的负极活性物质,与结构[1]~[5]实施例1同样地制备浆料。将得到的浆料涂布在厚度10μm的压延铜箔的两面上,并进行干燥,用压制机压延成厚度75μm,再将其切成宽104mm、长3300mm大小的活性物质层,并具有在长度方向上每间隔20mm具有宽6mm的未涂布部分的形状,作为负极。此时的负极活性物质的密度为1.35g/cm3。Slurry was prepared in the same manner as in Example 1 of Structures [1] to [5] using the same negative electrode active material as in Example 1 of Structures [1] to [5]. The obtained slurry is coated on both sides of a rolled copper foil with a thickness of 10 μm, dried, rolled into a thickness of 75 μm by a press, and then cut into an active material layer with a width of 104 mm and a length of 3300 mm. The shape of an uncoated portion having a width of 6 mm at intervals of 20 mm in the direction was used as a negative electrode. The density of the negative electrode active material at this time was 1.35 g/cm 3 .
《电池的制作》"Battery Making"
在各电极之间夹着多孔聚乙烯片的微多孔膜隔板(厚度25μm)并使得正极与负极不直接接触,此外使未涂布部分为相反一侧地将正极和负极重叠并卷绕成圆形,制成电极体。在捆束卷绕的中心轴上将该正极和负极分别进行点焊接,制作集电片,制成电极组,将该电池组设置在铝制的电池罐(外部尺寸:36φ×120mm)中并使得负极集电片为底。负极集电片在罐底进行点焊接,形成电池罐为负极集电端子的结构。准备具有正极的集电端子、压力释放阀的电池罐盖,将正极集电片和正极集电端子通过点焊接而连接。然后,向装有电极组的电池罐中注入20mL非水电解液,使电极充分浸透,通过铆接成型密封电池罐盖,制作圆筒电池。正极的电极面积的总和相对于电池的外壳表面积总和之比为41.0,电极组占有率为0.58。A microporous membrane separator (thickness 25 μm) of a porous polyethylene sheet is sandwiched between each electrode so that the positive electrode and the negative electrode do not directly contact each other, and the positive electrode and the negative electrode are overlapped and wound into an uncoated part on the opposite side. Round shape, made of electrode body. The positive electrode and the negative electrode were respectively spot-welded on the central axis of the bundle winding to make a collector sheet to make an electrode group, and the battery group was set in an aluminum battery can (outer dimension: 36φ×120mm) and Make the negative electrode current collector sheet as the bottom. The negative electrode current collector is spot-welded at the bottom of the tank to form a structure in which the battery can is the negative electrode current collector terminal. A battery can lid having a positive electrode collector terminal and a pressure release valve was prepared, and the positive electrode collector tab and the positive electrode collector terminal were connected by spot welding. Then, inject 20 mL of non-aqueous electrolyte solution into the battery can with the electrode group to fully soak the electrodes, and seal the lid of the battery can by riveting to make a cylindrical battery. The ratio of the total electrode area of the positive electrode to the total surface area of the battery case was 41.0, and the electrode group occupancy ratio was 0.58.
《电池的评价》"Battery Evaluation"
除了使用上述圆筒电池之外,与结构[1]~[5]实施例1同样地进行电池评价。电池评价的结构如结构[1]~[5]表3所示。Battery evaluation was performed in the same manner as in Example 1 with structures [1] to [5] except that the above-mentioned cylindrical battery was used. The structure of the battery evaluation is shown in Table 3 with structures [1] to [5].
结构[1]~[5]实施例6Structure [1]~[5] Embodiment 6
除了在结构[1]~[5]实施例5中使非水电解液含有0.3质量%的甲磺酸三甲基甲硅烷基酯来替代六甲基环三硅氧烷以外,与结构[1]~[5]实施例5同样地制作电池,并同样地进行电池评价。结果如结构[1]~[5]表3所示。In addition to making the non-aqueous electrolyte solution contain 0.3% by mass of trimethylsilyl methanesulfonate to replace hexamethylcyclotrisiloxane in Example 5 of structures [1] to [5], the same structure as [1] ] to [5] A battery was produced in the same manner as in Example 5, and battery evaluation was performed in the same manner. The results are shown in Table 3 of structures [1]-[5].
结构[1]~[5]实施例7Structure [1]~[5] Embodiment 7
除了在结构[1]~[5]实施例5中使非水电解液含有0.3质量%的苯基二甲基氟硅烷来替代六甲基环三硅氧烷以外,与结构[1]~[5]实施例5同样地制作电池,并同样地进行电池评价。结果如结构[1]~[5]表3所示。In addition to making the nonaqueous electrolyte solution contain 0.3% by mass of phenyldimethylfluorosilane instead of hexamethylcyclotrisiloxane in Example 5 of structures [1] to [5], the structure [1] to [ 5] A battery was produced in the same manner as in Example 5, and battery evaluation was performed in the same manner. The results are shown in Table 3 of structures [1]-[5].
结构[1]~[5]实施例8Structure [1]~[5] Embodiment 8
除了在结构[1]~[5]实施例5中使非水电解液含有0.3质量%的二氟磷酸锂来替代六甲基环三硅氧烷以外,与结构[1]~[5]实施例5同样地制作电池,并同样地进行电池评价。结果如结构[1]~[5]表3所示。Except that in Example 5 of structures [1] to [5], the non-aqueous electrolyte solution contains 0.3% by mass of lithium difluorophosphate instead of hexamethylcyclotrisiloxane, and the structure [1] to [5] is implemented. A battery was produced in the same manner as in Example 5, and battery evaluation was performed in the same manner. The results are shown in Table 3 of structures [1]-[5].
结构[1]~[5]比较例2Structure [1]~[5] Comparative example 2
除了在结构[1]~[5]实施例5中使非水电解液不含有六甲基环三硅氧烷以外,与结构[1]~[5]实施例5同样地制作电池,并同样地进行电池评价。结果如结构[1]~[5]表3所示。Except that the non-aqueous electrolyte solution does not contain hexamethylcyclotrisiloxane in Example 5 of Structures [1] to [5], a battery was produced in the same manner as in Example 5 of Structures [1] to [5], and the same for battery evaluation. The results are shown in Table 3 of structures [1]-[5].
结构[1]~[5]表3Structure [1]~[5] Table 3
[表65][Table 65]
由结构[1]~[5]表2和结构[1]~[5]表3的结果可知,任意一个电池中,通过在非水电解液中含有特定化合物,输出功率、容量保持率、安全性都得到提高,即使在循环试验后,直流电阻成分的增加也小,电池容量和输出功率得到充分保持。From the results in Table 2 of structures [1]-[5] and Table 3 of structures [1]-[5], it can be seen that in any battery, by including a specific compound in the nonaqueous electrolyte, the output power, capacity retention rate, safety The performance is improved, and even after the cycle test, the increase of the DC resistance component is small, and the battery capacity and output power are fully maintained.
结构[6]实施例1Structure [6] Embodiment 1
《非水电解液的制作》"Preparation of Non-Aqueous Electrolyte"
在干燥氩氛围下,在纯化的碳酸亚乙酯(EC)、碳酸二甲酯(DMC)和碳酸甲乙酯(EMC)的体积比3∶3∶4的混合溶剂中以1mol/L的浓度溶解充分干燥的六氟磷酸锂(LiPF6)。并使其含有0.3质量%的六甲基环三硅氧烷。Under a dry argon atmosphere, in a mixed solvent of purified ethylene carbonate (EC), dimethyl carbonate (DMC) and ethyl methyl carbonate (EMC) at a volume ratio of 3:3:4 at a concentration of 1 mol/L Dissolve well-dried lithium hexafluorophosphate (LiPF 6 ). And it was made to contain 0.3 mass % of hexamethylcyclotrisiloxane.
《正极的制作》"Positive Production"
正极活性物质为采用下述方法合成的锂过渡金属复合氧化物,以组成式LiMn0.33Ni0.33Co0.33O2表示。以Mn∶Ni∶Co=1∶1∶1的摩尔比称量作为锰原料的Mn3O4、作为镍原料的NiO以及作为钴原料的Co(OH)2,向其中加入纯水制成浆料,一边搅拌一边使用循环式介质搅拌型湿式珠磨机将浆料中的固体成分湿式粉碎至中值粒径0.2μm。The positive electrode active material is a lithium transition metal composite oxide synthesized by the following method, represented by the composition formula LiMn 0.33 Ni 0.33 Co 0.33 O 2 . Weigh Mn 3 O 4 as a manganese raw material, NiO as a nickel raw material, and Co(OH) 2 as a cobalt raw material at a molar ratio of Mn:Ni:Co=1:1:1, and add pure water to make a slurry While stirring, the solid content in the slurry was wet pulverized to a median particle size of 0.2 μm using a circulating medium agitation type wet bead mill.
通过喷雾干燥器将浆料进行喷雾干燥,得到仅仅包含锰原料、镍原料、钴原料的粒径约5μm的大致球状的造粒粒子。在得到的造粒粒子中添加中值粒径3μm的LiOH粉末,并使Li的摩尔数与Mn、Ni和Co的总摩尔数之比为1.05,用高速混合机进行混合,得到镍原料、钴原料、锰原料的造粒粒子和锂原料的混合粉末。在空气流通下、950℃对该混合粉末烧制12小时(升降温速度为5℃/分钟)后,粉碎,通过网眼45μm的筛子,得到正极活性物质。该正极活性物质的BET比表面积为1.2m2/g,平均一次粒径为0.8μm,中值粒径d50为4.4μm,振实密度为1.6g/cm3。The slurry was spray-dried by a spray dryer to obtain roughly spherical granulated particles having a particle diameter of about 5 μm and containing only manganese raw materials, nickel raw materials, and cobalt raw materials. LiOH powder with a median diameter of 3 μm was added to the obtained granulated particles, and the ratio of the number of moles of Li to the total number of moles of Mn, Ni, and Co was 1.05, and mixed with a high-speed mixer to obtain nickel raw materials, cobalt Raw material, granulated particles of manganese raw material and mixed powder of lithium raw material. The mixed powder was calcined at 950° C. for 12 hours under air circulation (the heating and cooling rate was 5° C./min), then pulverized and passed through a sieve with a mesh size of 45 μm to obtain a positive electrode active material. The positive electrode active material had a BET specific surface area of 1.2 m 2 /g, an average primary particle size of 0.8 μm, a median particle size d 50 of 4.4 μm, and a tap density of 1.6 g/cm 3 .
在N-甲基吡咯烷酮溶剂中混合90质量%作为正极活性物质的钴酸锂(LiCoO2)、5质量%的作为导电材料的乙炔黑和5质量%作为粘合剂的聚偏氟乙烯(PVdF),制成浆料。将得到的浆料涂布在厚度15μm的铝箔两面,并干燥,用压制机压延成厚度81μm,再将其切成宽100mm、长100mm大小的正极活性物质层,并具有宽30mm的未涂布部分的形状,作为正极。正极活性物质层的密度为2.35g/cm3,(一面的正极活性物质层的厚度)/(集电体的厚度)为2.2,L/(2×S2)为0.2。Mix 90% by mass of lithium cobaltate (LiCoO 2 ) as a positive electrode active material, 5% by mass of acetylene black as a conductive material, and 5% by mass of polyvinylidene fluoride (PVdF ) to make slurry. The obtained slurry is coated on both sides of aluminum foil with a thickness of 15 μm, dried, and rolled to a thickness of 81 μm with a press, and then cut into a positive electrode active material layer with a width of 100 mm and a length of 100 mm, and has a width of 30 mm. The shape of the part, as the positive electrode. The density of the positive electrode active material layer was 2.35 g/cm 3 , (thickness of positive electrode active material layer on one surface)/(thickness of current collector) was 2.2, and L/(2×S 2 ) was 0.2.
《负极的制作》"The Making of Negative Pole"
为了防止作为粒状碳质物质的市售的天然石墨粉末中混入粗大粒子,使用ASTM400目的筛子反复筛5次。将这样得到的负极材料作为碳质物质(A)。In order to prevent the commercially available natural graphite powder, which is a granular carbonaceous material, from being mixed with coarse particles, the sieve was repeatedly sieved 5 times using an ASTM400 mesh sieve. The negative electrode material thus obtained is referred to as a carbonaceous substance (A).
在碳质物质(A)中混合石脑油热解时得到的石油类重油,在非活性气体中实施1300℃的碳化处理,然后,通过对烧结物进行分级处理得到复合碳质物质,作为负极活性物质,所述复合碳质物质是在碳质物质(A)粒子表面上包覆了具有不同结晶性的碳质物质的复合碳质物质。分级处理时,为了防止混入粗大粒子,使用ASTM400目的筛子反复筛5次。从残炭率可以确定,得到的负极活性物质粉末被相对于95重量份石墨为5重量的低结晶性碳质物质包覆。负极活性物质的物性如结构[6]表1所示。Petroleum heavy oil obtained during the pyrolysis of naphtha is mixed with the carbonaceous substance (A), carbonized at 1300°C in an inert gas, and then the composite carbonaceous substance is obtained by classifying the sintered product as a negative electrode In the active material, the composite carbonaceous substance is a composite carbonaceous substance in which carbonaceous substances having different crystallinity are coated on the surface of the carbonaceous substance (A) particle. During the classification process, in order to prevent the mixing of coarse particles, use an ASTM400 mesh sieve to repeatedly sieve 5 times. It was confirmed from the carbon residue rate that the obtained negative electrode active material powder was coated with 5 parts by weight of low-crystalline carbonaceous material relative to 95 parts by weight of graphite. The physical properties of the negative electrode active material are shown in Table 1 of the structure [6].
结构[6]表1Structure [6] Table 1
[表66][Table 66]
[3897]在98重量份上述负极活性物质中添加100重量份作为增稠剂的羧甲基纤维素钠的水性分散液(羧甲基纤维素钠的浓度为1质量%)、2重量份作为粘合剂的苯乙烯-丁二烯橡胶的水性分散液(苯乙烯-丁二烯橡胶的浓度为50质量%),用分散器进行混合,制成浆料。将得到的浆料涂布在厚度10μm的压延铜箔的两面,并干燥,用压制机压延成厚度为75μm,再将其切成宽104mm、长104mm大小的活性物质层,并具有宽30mm的未涂布部分的形状,作为负极。此时的负极活性物质的密度为1.35g/cm3,L/(2×S2)为0.19。In 98 parts by weight of the above-mentioned negative electrode active material, add 100 parts by weight of the aqueous dispersion of sodium carboxymethyl cellulose as a thickener (the concentration of sodium carboxymethyl cellulose is 1 mass % ) , 2 parts by weight as An aqueous dispersion of styrene-butadiene rubber as a binder (the concentration of styrene-butadiene rubber is 50% by mass) was mixed with a disperser to prepare a slurry. The obtained slurry is coated on both sides of a rolled copper foil with a thickness of 10 μm, dried, rolled into a thickness of 75 μm by a press, and then cut into an active material layer with a width of 104 mm and a length of 104 mm, and has a width of 30 mm. The shape of the uncoated part, as the negative electrode. The density of the negative electrode active material at this time was 1.35 g/cm 3 , and L/(2×S 2 ) was 0.19.
《电池的制作》"Battery Making"
将32片正极和33片负极交替配置,各电极之间夹入多孔聚乙烯片隔板(厚度为25μm),进行叠层。此时,使正极活性物质面与负极活性物质面相对,并且不会露出负极活性物质面之外。将该正极和负极各自的未涂布部彼此捆束,与作为集电端子的金属片一起通过点焊接连接,制作集电片,制成电极组。以上,多孔聚乙烯片在135℃以上堵塞孔。作为电池的外壳材料,使用依次叠层有聚丙烯膜、厚度0.04mm的铝箔和尼龙膜而得到的片(总计厚度0.1mm),使聚丙烯膜位于内面一侧来成型为矩形状杯,作为外壳。该聚丙烯膜的熔点为165℃。将上述电极组封入外壳中并使集电端子从杯上端的未密封部分向外露出,注入20mL非水电解液,使电极充分浸透。在减压下将杯上端热封而对其进行密封,制作电池。电池大致为方形,电池的正极的电极面积总和相对于外壳表面积(不含热封部分的外壳材料表面积)总和之比为22.6,(2×S1/T)为411。32 positive electrodes and 33 negative electrodes were arranged alternately, and a porous polyethylene sheet separator (thickness: 25 μm) was sandwiched between the electrodes for lamination. At this time, the surface of the positive electrode active material faces the surface of the negative electrode active material without protruding from the surface of the negative electrode active material. The uncoated parts of the positive and negative electrodes were bundled together, and connected by spot welding together with the metal sheet serving as a current collecting terminal to produce a current collecting sheet and an electrode group. Above, the porous polyethylene sheet clogged the pores above 135°C. As the battery case material, a sheet (total thickness 0.1 mm) obtained by laminating a polypropylene film, an aluminum foil with a thickness of 0.04 mm, and a nylon film in this order was used, and the polypropylene film was formed on the inner side to form a rectangular cup. shell. The polypropylene film had a melting point of 165°C. Enclose the above-mentioned electrode group in the casing and expose the collector terminal from the unsealed part of the upper end of the cup, and inject 20mL of non-aqueous electrolyte solution to fully soak the electrodes. The upper end of the cup was sealed by heat sealing under reduced pressure to produce a battery. The battery was roughly square, and the ratio of the total electrode area of the positive electrode to the total surface area of the case (the surface area of the case material excluding the heat-sealed portion) was 22.6, and (2×S 1 /T) was 411.
《电池的评价》"Battery Evaluation"
(电池容量的测定方法)(Measuring method of battery capacity)
对未经充放电循环的新电池,在25℃、4.1V~3.0V的电压范围下以电流值0.2C(将1小时放电额定容量的电流值作为1C,所述额定容量取决于1小时率(one-hour-rate)的放电容量,下同)进行5个循环初期充放电。将此时的第5个循环0.2C放电容量作为“电池容量”(Ah)。电池评价的结果如结构[6]表3所示。For a new battery that has not been charged and discharged cycled, at 25°C and a voltage range of 4.1V to 3.0V, use a current value of 0.2C (take the current value of the rated capacity of 1 hour discharge as 1C, and the rated capacity depends on the rate of 1 hour (one-hour-rate discharge capacity, the same below) for 5 cycles of initial charge and discharge. The 0.2C discharge capacity at the fifth cycle at this time was taken as "battery capacity" (Ah). The results of the battery evaluation are shown in Table 3 of the structure [6].
(直流电阻成分的测定方法)(Measurement method of DC resistance component)
在25℃环境下通过0.2C的恒定电流进行150分钟充电,施加10kHz的交流,测定阻抗,作为“直流电阻成分”(mΩ)。电池评价的结果如结构[6]表3所示。Charge for 150 minutes with a constant current of 0.2C in an environment of 25° C., apply an alternating current of 10 kHz, measure impedance, and use it as a “direct current resistance component” (mΩ). The results of the battery evaluation are shown in Table 3 of the structure [6].
(初期输出功率的测定方法)(Measurement method of initial output power)
在25℃环境下通过0.2C的恒定电流进行150分钟充电,分别以0.1C、0.3C、1.0C、3.0C、10.0C放电10秒,测定第10秒的电压。将由电流-电压直线与下限电压(3V)围成的三角形的面积作为“初期输出功率”(W)。电池评价的结果如结构[6]表3所示。Charge at a constant current of 0.2C for 150 minutes at 25°C, discharge at 0.1C, 0.3C, 1.0C, 3.0C, and 10.0C for 10 seconds, and measure the voltage at 10 seconds. The area of the triangle surrounded by the current-voltage line and the lower limit voltage (3V) is taken as the "initial output power" (W). The results of the battery evaluation are shown in Table 3 of the structure [6].
(循环试验)(cycle test)
(耐久后电池容量、耐久后直流电阻成分、耐久后输出功率的测定方法)(Measurement method of battery capacity after endurance, DC resistance component after endurance, output power after endurance)
在被视为锂二次电池的实际使用上限温度的60℃的高温环境下进行循环试验。用2C的恒定电流恒定电压法充电至充电上限电压4.1V后,以2C的恒定电流放电至放电终止电压3.0V,将该充放电循环作为1个循环,反复进行该循环直至500循环。对于循环试验结束后的电池,在25℃环境下以电流值0.2C进行3个循环充放电,将其第3个循环的0.2C放电容量作为“耐久后电池容量”。此外,对于循环试验结束后的电池,测定直流电阻成分,作为“耐久后直流电阻成分”,进行输出功率测定,作为“耐久后输出功率”。电池评价的结果如结构[6]表3所示。The cycle test was performed in a high-temperature environment of 60° C., which is regarded as the practical upper limit temperature of the lithium secondary battery. After charging to the charging upper limit voltage of 4.1V by the constant current and constant voltage method of 2C, it was discharged to the end-of-discharge voltage of 3.0V by the constant current of 2C. This charge-discharge cycle was regarded as one cycle, and this cycle was repeated until 500 cycles. For the battery after the cycle test, it is charged and discharged for 3 cycles with a current value of 0.2C in an environment of 25°C, and the 0.2C discharge capacity of the third cycle is taken as the "endurance battery capacity". In addition, the DC resistance component of the battery after the cycle test was measured as the "DC resistance component after endurance", and the output power was measured as "Output after endurance". The results of the battery evaluation are shown in Table 3 of the structure [6].
(过充电试验)(overcharge test)
在25℃环境下进行过充电试验。由放电状态(3V)以3C的恒定电流进行充电,观测其举动。其中,“阀工作”表示排气阀工作并释放非水电解液成分的现象,“破裂”表示电池容器以猛烈的势头破裂、内容物强制释放的现象。电池评价的结果如结构[6]表3所示。The overcharge test was carried out in an environment of 25°C. It was charged with a constant current of 3C from the discharged state (3V), and its behavior was observed. Among them, "valve operation" refers to the phenomenon in which the exhaust valve operates to release the non-aqueous electrolyte components, and "rupture" refers to the phenomenon in which the battery container ruptures violently and the contents are forcibly released. The results of the battery evaluation are shown in Table 3 of the structure [6].
(体积变化率的测定方法)(Measuring method of volume change rate)
在25℃环境下对放电状态(3V)的电池体积进行测定。体积通过向带有刻度容器中加入乙醇,将电池沉没在乙醇中来测定。将循环试验后的体积相对于循环试验前的体积的比例作为“体积变化率”。评价结果如结构[6]表3所示。The volume of the battery in the discharged state (3V) was measured in an environment of 25°C. Volume is determined by adding ethanol to a graduated container and submerging the cell in the ethanol. The ratio of the volume after the cycle test to the volume before the cycle test was defined as the "volume change rate". The evaluation results are shown in Table 3 of the structure [6].
结构[6]实施例2Structure [6] Embodiment 2
除了在结构[6]实施例1中使非水电解液含有0.3质量%的甲磺酸三甲基甲硅烷基酯来替代六甲基环三硅氧烷以外,与实施例1同样地制作电池,并同样地进行电池评价。结果如结构[6]表3所示。A battery was fabricated in the same manner as in Example 1, except that in Example 1 of structure [6], the nonaqueous electrolyte contained 0.3% by mass of trimethylsilyl methanesulfonate instead of hexamethylcyclotrisiloxane. , and perform battery evaluation in the same manner. The results are shown in Table 3 of the structure [6].
结构[6]实施例3Structure [6] Embodiment 3
除了在结构[6]实施例1中使非水电解液含有0.3质量%的苯基二甲基氟硅烷来替代六甲基环三硅氧烷以外,与结构[6]实施例1同样地制作电池,并同样地进行电池评价。结果如结构[6]表3所示。Produced in the same manner as structure [6] Example 1, except that the non-aqueous electrolytic solution contained 0.3% by mass of phenyldimethylfluorosilane instead of hexamethylcyclotrisiloxane in Structure [6] Example 1 battery, and perform battery evaluation in the same way. The results are shown in Table 3 of the structure [6].
结构[6]实施例4Structure [6] Embodiment 4
除了在结构[6]实施例1中使非水电解液含有0.3质量%的二氟磷酸锂来替代六甲基环三硅氧烷以外,与结构[6]实施例1同样地制作电池,并同样地进行电池评价。结果如结构[6]表3所示。Except that in structure [6] Example 1, the nonaqueous electrolyte solution contains 0.3% by mass of lithium difluorophosphate instead of hexamethylcyclotrisiloxane, a battery was produced in the same manner as in Structure [6] Example 1, and Battery evaluation was performed in the same manner. The results are shown in Table 3 of the structure [6].
结构[6]比较例1Structure [6] Comparative Example 1
除了在结构[6]实施例1中使非水电解液不含有六甲基环三硅氧烷以外,与结构[6]实施例1同样地制作电池,并同样地进行电池评价。结果如结构[6]表3所示。A battery was produced in the same manner as in Example 1 of Structure [6] except that the non-aqueous electrolyte solution did not contain hexamethylcyclotrisiloxane in Example 1 of Structure [6], and battery evaluation was performed in the same manner. The results are shown in Table 3 of the structure [6].
结构[6]表2Structure [6] Table 2
[表67][Table 67]
结构[6]表3Structure [6] Table 3
[表68][Table 68]
由结构[6]表3的结果可知,通过在非水电解液中含有特定的化合物,初期输出功率、容量保持率、安全性得到提高,即使在循环试验后也可充分保持电池容量和输出功率,电池体积的变化也小。From the results of structure [6] Table 3, it can be seen that by including specific compounds in the non-aqueous electrolyte, the initial output power, capacity retention rate, and safety are improved, and the battery capacity and output power can be fully maintained even after the cycle test , the change in battery volume is also small.
工业实用性Industrial Applicability
对本发明的锂二次电池的用途不特别限定,可以用于公知的各种用途中。作为具体的例子,可以举出笔记本个人电脑、笔输入式个人电脑、移动个人电脑、电子书播放器(Electronic book player)、便携式电话、便携式传真机、便携式复印机、便携式打印机、头戴受话器、摄录机、液晶电视、便利吸尘器、便携式CD、迷你式唱片播放器、无线电收发机、电子笔记本、计算器、存储卡、便携式磁带录音机、收音机、后备电源、电动机、汽车、摩托车、带电动机的自行车、自行车、照明器具、玩具、游戏机、钟表、电动工具、频闪仪、照相机等。本发明的锂二次电池,由于具有大容量、优异的寿命和高输出功率,气体产生少,即使是过充电时安全性也高,所以在特别是要求大容量的领域中,可以合适地广泛应用。The application of the lithium secondary battery of the present invention is not particularly limited, and it can be used in various known applications. Specific examples include notebook personal computers, pen input personal computers, mobile personal computers, electronic book players, cellular phones, portable facsimile machines, portable copiers, portable printers, headsets, VCRs, LCD TVs, convenient vacuum cleaners, portable CDs, mini record players, radio transceivers, electronic notebooks, calculators, memory cards, portable tape recorders, radios, backup power supplies, electric motors, automobiles, motorcycles, Bicycles, bicycles, lighting equipment, toys, game consoles, clocks, electric tools, stroboscopes, cameras, etc. The lithium secondary battery of the present invention has a large capacity, excellent life and high output power, produces less gas, and is highly safe even when overcharged, so it can be suitably widely used in fields requiring a large capacity. application.
虽然以上使用本发明和特定的方式进行了具体说明,但是对于本领域技术人员来说,不脱离本发明的意图和范围可以进行各种改变是显而易见的。而且,本申请基于以下的日本专利申请,通过引用援用其全部。Although the present invention and specific embodiments have been specifically described above, it is obvious for those skilled in the art that various changes can be made without departing from the intention and scope of the present invention. Also, this application is based on the following Japanese patent applications, all of which are incorporated by reference.
日本特愿2005-331255(申请日:2005年11月16日)Japanese Patent Application No. 2005-331255 (filing date: November 16, 2005)
日本特愿2005-331362(申请日:2005年11月16日)Japanese Patent Application No. 2005-331362 (filing date: November 16, 2005)
日本特愿2005-331391(申请日:2005年11月16日)Japanese Patent Application No. 2005-331391 (filing date: November 16, 2005)
日本特愿2005-331477(申请日:2005年11月16日)Japanese Patent Application No. 2005-331477 (filing date: November 16, 2005)
日本特愿2005-331513(申请日:2005年11月16日)Japanese Patent Application No. 2005-331513 (filing date: November 16, 2005)
日本特愿2005-339794(申请日:2005年11月25日)Japanese Patent Application No. 2005-339794 (filing date: November 25, 2005)
日本特愿2006-019863(申请日:2006年1月27日)Japanese Patent Application No. 2006-019863 (application date: January 27, 2006)
日本特愿2006-005622(申请日:2005年1月13日)Japanese Patent Application No. 2006-005622 (filing date: January 13, 2005)
日本特愿2005-367747(申请日:2005年12月21日)Japanese Patent Application No. 2005-367747 (filing date: December 21, 2005)
日本特愿2005-377366(申请日:2005年12月28日)Japanese Patent Application No. 2005-377366 (filing date: December 28, 2005)
日本特愿2005-349052(申请日:2005年12月2日)Japanese Patent Application No. 2005-349052 (filing date: December 2, 2005)
日本特愿2005-359061(申请日:2005年12月13日)Japanese Patent Application No. 2005-359061 (filing date: December 13, 2005)
日本特愿2006-019879(申请日:2006年1月27日)Japanese Patent Application No. 2006-019879 (application date: January 27, 2006)
日本特愿2006-013664(申请日:2005年1月23日)Japanese Patent Application No. 2006-013664 (filing date: January 23, 2005)
日本特愿2005-314043(申请日:2005年10月28日)Japanese Patent Application No. 2005-314043 (filing date: October 28, 2005)
日本特愿2005-331585(申请日:2005年11月16日)Japanese Patent Application No. 2005-331585 (filing date: November 16, 2005)
日本特愿2005-305368(申请日:2005年10月20日)Japanese Patent Application No. 2005-305368 (filing date: October 20, 2005)
日本特愿2005-344732(申请日:2005年11月29日)Japanese Patent Application No. 2005-344732 (filing date: November 29, 2005)
日本特愿2005-343629(申请日:2005年11月29日)Japanese Patent Application No. 2005-343629 (filing date: November 29, 2005)
日本特愿2005-332173(申请日:2005年11月16日)Japanese Patent Application No. 2005-332173 (filing date: November 16, 2005)
日本特愿2005-305300(申请日:2005年10月20日)Japanese Patent Application No. 2005-305300 (application date: October 20, 2005)
日本特愿2005-353005(申请日:2005年12月7日)Japanese Patent Application No. 2005-353005 (application date: December 7, 2005)
日本特愿2005-314260(申请日:2005年10月28日)Japanese Patent Application No. 2005-314260 (filing date: October 28, 2005)
日本特愿2005-369824(申请日:2005年12月22日)Japanese Patent Application No. 2005-369824 (filing date: December 22, 2005)
日本特愿2005-370024(申请日:2005年12月22日)Japanese Patent Application No. 2005-370024 (filing date: December 22, 2005)
附图说明Description of drawings
[图1](a)为负极[7]的实施例1~3和比较例1中使用的薄膜负极(1)的截面扫描型电子显微镜(SEM)照片。(b)是示出负极[7]的薄膜负极(1)的通过电子探针显微分析仪(EPMA)得到的元素Si在膜厚方向的质量浓度分布的照片。(c)是示出对负极[7]的薄膜负极(1)的通过电子探针显微分析仪(EPMA)得到的元素C在膜厚方向的质量浓度分布的照片。[ Fig. 1 ] (a) is a cross-sectional scanning electron microscope (SEM) photograph of a thin film negative electrode (1) used in Examples 1 to 3 and Comparative Example 1 of the negative electrode [7]. (b) is a photograph showing the mass concentration distribution of element Si in the film thickness direction of the thin-film negative electrode (1) of the negative electrode [7] obtained by an electron probe microanalyzer (EPMA). (c) is a photograph showing the mass concentration distribution of element C in the film thickness direction of the thin-film negative electrode (1) of the negative electrode [7] obtained by an electron probe microanalyzer (EPMA).
Claims (46)
Priority Applications (13)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202411245457.9A CN119133604A (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and non-aqueous electrolyte used therein |
| CN201510689192.6A CN105375064A (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and non-aqueous electrolyte used therein |
| CN202110646170.7A CN113571772A (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and nonaqueous electrolyte used therein |
| CN201910202173.4A CN110010850A (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and nonaqueous electrolyte used therein |
| CN201710119547.7A CN107069091A (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and non-aqueous electrolyte used therein |
| CN201210427884.XA CN102931434B (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and non-aqueous electrolyte used therein |
| CN201410514981.1A CN104659414B (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and non-aqueous electrolyte used therein |
| CN202110664313.7A CN113394458A (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and nonaqueous electrolyte used therein |
| CN202110664613.5A CN113594543A (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and nonaqueous electrolyte used therein |
| CN202510122565.5A CN119812477A (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and non-aqueous electrolyte used therein |
| CN201510151471.7A CN104752642B (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and non-aqueous electrolyte used therein |
| CN201910201995.0A CN110061283A (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and nonaqueous electrolyte used therein |
| CN201510504217.0A CN105186038B (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and non-aqueous electrolyte used therein |
Applications Claiming Priority (36)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP305368/2005 | 2005-10-20 | ||
| JP2005305368 | 2005-10-20 | ||
| JP305300/2005 | 2005-10-20 | ||
| JP2005305300 | 2005-10-20 | ||
| JP2005314043 | 2005-10-28 | ||
| JP314260/2005 | 2005-10-28 | ||
| JP2005314260 | 2005-10-28 | ||
| JP314043/2005 | 2005-10-28 | ||
| JP2005331585 | 2005-11-16 | ||
| JP331362/2005 | 2005-11-16 | ||
| JP2005331513 | 2005-11-16 | ||
| JP2005331477 | 2005-11-16 | ||
| JP2005331362 | 2005-11-16 | ||
| JP331391/2005 | 2005-11-16 | ||
| JP2005332173 | 2005-11-16 | ||
| JP331513/2005 | 2005-11-16 | ||
| JP331477/2005 | 2005-11-16 | ||
| JP2005331391 | 2005-11-16 | ||
| JP332173/2005 | 2005-11-16 | ||
| JP331585/2005 | 2005-11-16 | ||
| JP331255/2005 | 2005-11-16 | ||
| JP339794/2005 | 2005-11-25 | ||
| JP343629/2005 | 2005-11-29 | ||
| JP344732/2005 | 2005-11-29 | ||
| JP349052/2005 | 2005-12-02 | ||
| JP353005/2005 | 2005-12-07 | ||
| JP359061/2005 | 2005-12-13 | ||
| JP367747/2005 | 2005-12-21 | ||
| JP370024/2005 | 2005-12-22 | ||
| JP369824/2005 | 2005-12-22 | ||
| JP377366/2005 | 2005-12-28 | ||
| JP005622/2006 | 2006-01-13 | ||
| JP013664/2006 | 2006-01-23 | ||
| JP019879/2006 | 2006-01-27 | ||
| JP019863/2006 | 2006-01-27 | ||
| PCT/JP2006/320882 WO2007055087A1 (en) | 2005-10-20 | 2006-10-19 | Lithium secondary cell and nonaqueous electrolytic solution for use therein |
Related Child Applications (15)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201210427884.XA Division CN102931434B (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and non-aqueous electrolyte used therein |
| CN2010102426208A Division CN101894974A (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and nonaqueous electrolyte used therein |
| CN202411245457.9A Division CN119133604A (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and non-aqueous electrolyte used therein |
| CN202110664313.7A Division CN113394458A (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and nonaqueous electrolyte used therein |
| CN202110646170.7A Division CN113571772A (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and nonaqueous electrolyte used therein |
| CN201510151471.7A Division CN104752642B (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and non-aqueous electrolyte used therein |
| CN201910202173.4A Division CN110010850A (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and nonaqueous electrolyte used therein |
| CN201510504217.0A Division CN105186038B (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and non-aqueous electrolyte used therein |
| CN202510122565.5A Division CN119812477A (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and non-aqueous electrolyte used therein |
| CN201910201995.0A Division CN110061283A (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and nonaqueous electrolyte used therein |
| CN201710119547.7A Division CN107069091A (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and non-aqueous electrolyte used therein |
| CN201410514981.1A Division CN104659414B (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and non-aqueous electrolyte used therein |
| CN2011102544054A Division CN102324567A (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and non-aqueous electrolyte used therein |
| CN201510689192.6A Division CN105375064A (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and non-aqueous electrolyte used therein |
| CN202110664613.5A Division CN113594543A (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and nonaqueous electrolyte used therein |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN101292389A CN101292389A (en) | 2008-10-22 |
| CN101292389B true CN101292389B (en) | 2010-09-22 |
Family
ID=40035669
Family Applications (7)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN2006800390896A Ceased CN101292389B (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and nonaqueous electrolyte used therein |
| CN201710119547.7A Pending CN107069091A (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and non-aqueous electrolyte used therein |
| CN202110664613.5A Pending CN113594543A (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and nonaqueous electrolyte used therein |
| CN202110664313.7A Pending CN113394458A (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and nonaqueous electrolyte used therein |
| CN201910201995.0A Pending CN110061283A (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and nonaqueous electrolyte used therein |
| CN202110646170.7A Pending CN113571772A (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and nonaqueous electrolyte used therein |
| CN201910202173.4A Pending CN110010850A (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and nonaqueous electrolyte used therein |
Family Applications After (6)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201710119547.7A Pending CN107069091A (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and non-aqueous electrolyte used therein |
| CN202110664613.5A Pending CN113594543A (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and nonaqueous electrolyte used therein |
| CN202110664313.7A Pending CN113394458A (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and nonaqueous electrolyte used therein |
| CN201910201995.0A Pending CN110061283A (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and nonaqueous electrolyte used therein |
| CN202110646170.7A Pending CN113571772A (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and nonaqueous electrolyte used therein |
| CN201910202173.4A Pending CN110010850A (en) | 2005-10-20 | 2006-10-19 | Lithium secondary battery and nonaqueous electrolyte used therein |
Country Status (1)
| Country | Link |
|---|---|
| CN (7) | CN101292389B (en) |
Cited By (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102751532A (en) * | 2011-04-22 | 2012-10-24 | 株式会社日立制作所 | Lithium ion secondary battery and battery pack system |
| WO2014094221A1 (en) * | 2012-12-18 | 2014-06-26 | 湖州创亚动力电池材料有限公司 | Composite graphite cathode material for high-capacity lithium ion battery, and preparation method thereof |
| TWI508359B (en) * | 2012-07-23 | 2015-11-11 | Furukawa Electric Co Ltd | Surface-treated copper foil and method for manufacturing the same, lithium ion secondary battery electrode, and lithium ion secondary battery |
| CN105580184A (en) * | 2013-09-25 | 2016-05-11 | 国立大学法人东京大学 | Non-aqueous electrolyte secondary battery |
| US10686223B2 (en) | 2013-09-25 | 2020-06-16 | Kabushiki Kaisha Toyota Jidoshokki | Nonaqueous electrolyte secondary battery |
| US11335903B2 (en) | 2020-08-18 | 2022-05-17 | Group14 Technologies, Inc. | Highly efficient manufacturing of silicon-carbon composites materials comprising ultra low z |
| US11437621B2 (en) | 2015-08-28 | 2022-09-06 | Group14 Technologies, Inc. | Materials with extremely durable intercalation of lithium and manufacturing methods thereof |
| US11495793B2 (en) | 2013-03-14 | 2022-11-08 | Group14 Technologies, Inc. | Composite carbon materials comprising lithium alloying electrochemical modifiers |
| US11492262B2 (en) | 2020-08-18 | 2022-11-08 | Group14Technologies, Inc. | Silicon carbon composites comprising ultra low Z |
| US11611071B2 (en) | 2017-03-09 | 2023-03-21 | Group14 Technologies, Inc. | Decomposition of silicon-containing precursors on porous scaffold materials |
| US11611073B2 (en) | 2015-08-14 | 2023-03-21 | Group14 Technologies, Inc. | Composites of porous nano-featured silicon materials and carbon materials |
| US11639292B2 (en) | 2020-08-18 | 2023-05-02 | Group14 Technologies, Inc. | Particulate composite materials |
| US11661517B2 (en) | 2014-03-14 | 2023-05-30 | Group14 Technologies, Inc. | Methods for sol-gel polymerization in absence of solvent and creation of tunable carbon structure from same |
| US11707728B2 (en) | 2013-11-05 | 2023-07-25 | Group14 Technologies, Inc. | Carbon-based compositions with highly efficient volumetric gas sorption |
| US11718701B2 (en) | 2012-02-09 | 2023-08-08 | Group14 Technologies, Inc. | Preparation of polymeric resins and carbon materials |
| US12046744B2 (en) | 2020-09-30 | 2024-07-23 | Group14 Technologies, Inc. | Passivated silicon-carbon composite materials |
Families Citing this family (77)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2010176996A (en) * | 2009-01-28 | 2010-08-12 | Sanyo Electric Co Ltd | Nonaqueous electrolyte secondary battery |
| KR101096936B1 (en) * | 2009-07-23 | 2011-12-22 | 지에스칼텍스 주식회사 | Negative active material for rechargeable lithium battery, method of manufacturing the same and rechargeable lithium battery having the same |
| JP5463957B2 (en) | 2010-03-02 | 2014-04-09 | ソニー株式会社 | Non-aqueous electrolyte and battery |
| CN105870494B (en) * | 2010-06-25 | 2019-07-05 | Nec 能源元器件株式会社 | Lithium ion secondary battery |
| US9419271B2 (en) * | 2010-07-02 | 2016-08-16 | Semiconductor Energy Laboratory Co., Ltd. | Electrode material and method for forming electrode material |
| EP2600460B1 (en) * | 2010-07-28 | 2015-01-28 | LG Chem, Ltd. | Nonaqueous electrolyte solution for lithium secondary battery, and lithium secondary battery containing same |
| US9312559B2 (en) * | 2010-09-22 | 2016-04-12 | Toyota Jidosha Kabushiki Kaisha | Nonaqueous electrolyte secondary battery provided with a wound electrode body |
| JP5781294B2 (en) | 2010-11-16 | 2015-09-16 | 株式会社Adeka | Non-aqueous electrolyte secondary battery |
| CN102064344A (en) * | 2010-12-21 | 2011-05-18 | 东莞市杉杉电池材料有限公司 | Novel electrolyte for power battery |
| CN102544582A (en) * | 2010-12-29 | 2012-07-04 | 东莞市杉杉电池材料有限公司 | A kind of electrolyte solution of power lithium ion battery |
| CN102082290A (en) * | 2010-12-30 | 2011-06-01 | 奇瑞汽车股份有限公司 | High-voltage high-energy-density lithium ion battery and manufacturing method thereof |
| CN105576282B (en) | 2011-02-10 | 2018-11-06 | 三菱化学株式会社 | Nonaqueous electrolytic solution and nonaqueous electrolyte secondary battery using the nonaqueous electrolytic solution |
| US10186732B2 (en) * | 2011-03-04 | 2019-01-22 | Denso Corporation | Nonaqueous electrolyte solution for batteries, and nonaqueous electrolyte secondary battery using same |
| CN102332604A (en) * | 2011-03-09 | 2012-01-25 | 东莞新能源科技有限公司 | High-power lithium ion battery |
| CN104649301B (en) * | 2011-04-11 | 2017-07-14 | 三菱化学株式会社 | Method for producing lithium fluorosulfonate, lithium fluorosulfonate, nonaqueous electrolytic solution, and nonaqueous electrolyte secondary battery |
| KR101978726B1 (en) * | 2011-06-03 | 2019-05-15 | 가부시키가이샤 한도오따이 에네루기 켄큐쇼 | Power storage device and method of manufacturing the same |
| CN102263286A (en) * | 2011-06-10 | 2011-11-30 | 东莞新能源科技有限公司 | A high energy density lithium-ion battery |
| JP5561559B2 (en) * | 2011-10-06 | 2014-07-30 | トヨタ自動車株式会社 | Method for manufacturing lithium secondary battery |
| JP5915083B2 (en) * | 2011-10-31 | 2016-05-11 | トヨタ自動車株式会社 | Evaluation method of non-aqueous electrolyte secondary battery |
| JP5823261B2 (en) | 2011-11-10 | 2015-11-25 | 株式会社Adeka | Non-aqueous electrolyte and non-aqueous electrolyte secondary battery using the electrolyte |
| KR102087564B1 (en) * | 2011-11-18 | 2020-03-10 | 스미또모 가가꾸 가부시끼가이샤 | Laminated porous film, method for producing same, non-aqueous electrolyte secondary battery separator, laminated electrode sheet, and non-aqueous electrolyte secondary battery |
| KR101325555B1 (en) | 2011-12-09 | 2013-11-05 | 주식회사 엘지화학 | Lithium Secondary Battery Comprising Spherical Graphite as Anode Active Material |
| CN102522592A (en) * | 2011-12-20 | 2012-06-27 | 彩虹集团公司 | Non-aqueous electrolyte, preparation method of non-aqueous electrolyte and lithium ion secondary battery containing non-aqueous electrolyte |
| US20140322576A1 (en) * | 2012-02-29 | 2014-10-30 | Shin-Kobe Electric Machinery Co., Ltd. | Lithium Ion Battery |
| CN103311519B (en) * | 2012-03-07 | 2016-12-14 | 上海杉杉科技有限公司 | A kind of composite hard carbon cathode material and its production and use |
| WO2013180522A1 (en) * | 2012-05-31 | 2013-12-05 | 주식회사 엘지화학 | Lithium secondary battery |
| JP2014010977A (en) * | 2012-06-28 | 2014-01-20 | Sharp Corp | Electrode for nonaqueous electrolyte secondary battery and nonaqueous electrolyte secondary battery including the same |
| WO2014003165A1 (en) * | 2012-06-29 | 2014-01-03 | 三菱化学株式会社 | Nonaqueous electrolytic solution and nonaqueous electrolytic solution cell using same |
| CN104471778A (en) * | 2012-07-17 | 2015-03-25 | 丰田自动车株式会社 | Lithium secondary battery and manufacturing method thereof |
| JP6041343B2 (en) * | 2012-09-05 | 2016-12-07 | トヨタ自動車株式会社 | Lithium secondary battery and manufacturing method thereof |
| CN104662627B (en) * | 2012-09-28 | 2017-10-31 | 松下知识产权经营株式会社 | Electrode for capacitor and capacitor using same |
| JP6476116B2 (en) | 2013-07-29 | 2019-02-27 | 昭和電工株式会社 | Carbon material, battery electrode material, and battery |
| CN103560270B (en) * | 2013-10-30 | 2015-11-18 | 河南师范大学 | A kind of electrolyte for lithium ion battery |
| JP6365082B2 (en) * | 2014-08-01 | 2018-08-01 | セントラル硝子株式会社 | Non-aqueous electrolyte battery electrolyte and non-aqueous electrolyte battery using the same |
| JP6240586B2 (en) * | 2014-10-28 | 2017-11-29 | Jfeケミカル株式会社 | Graphite particles for negative electrode material of lithium ion secondary battery, negative electrode of lithium ion secondary battery and lithium ion secondary battery |
| CN107004895B (en) * | 2014-11-28 | 2019-10-11 | 三洋电机株式会社 | Non-aqueous electrolyte secondary battery |
| DE102016208250A1 (en) * | 2015-05-19 | 2016-11-24 | Semiconductor Energy Laboratory Co., Ltd. | Electrode, energy storage device and electronic device |
| WO2017154908A1 (en) * | 2016-03-07 | 2017-09-14 | 株式会社 東芝 | Non-aqueous electrolyte cell and cell pack |
| CN106785044B (en) * | 2017-02-13 | 2020-01-14 | 宁德新能源科技有限公司 | Electrolyte and secondary battery |
| CN108878975B (en) * | 2017-05-12 | 2020-02-21 | 宁德时代新能源科技股份有限公司 | Electrolyte and secondary battery including the same |
| JP7074132B2 (en) * | 2017-06-01 | 2022-05-24 | 昭和電工マテリアルズ株式会社 | Electrolytes and electrochemical devices |
| KR102337069B1 (en) * | 2017-06-30 | 2021-12-08 | 다이킨 고교 가부시키가이샤 | Electrolytes, electrochemical devices, secondary batteries and modules |
| KR102264636B1 (en) * | 2017-11-30 | 2021-06-15 | 주식회사 엘지에너지솔루션 | Non-aqueous electrolyte for secondary battery and secondary battery comprising the same |
| JP7225237B2 (en) * | 2017-12-13 | 2023-02-20 | ビーエーエスエフ ソシエタス・ヨーロピア | Electrolyte composition containing oligomeric phosphonate silyl ester |
| EP3748758B1 (en) * | 2018-01-31 | 2024-05-22 | Panasonic Intellectual Property Management Co., Ltd. | Non-aqueous electrolyte secondary battery, electrolyte solution, and method for manufacturing non-aqueous electrolyte secondary battery |
| CN108336291B (en) * | 2018-03-15 | 2024-01-12 | 朱玉荣 | Vehicle-mounted liquid-continuous battery |
| CN112997338B (en) * | 2018-09-12 | 2024-10-29 | 浦项控股股份有限公司 | Positive electrode active material, preparation method thereof and lithium secondary battery containing the same |
| KR20210104038A (en) * | 2018-12-19 | 2021-08-24 | 할도르 토프쉐 에이/에스 | Lithium cathode active material |
| CN109777460B (en) * | 2019-01-30 | 2021-07-16 | 虞定生 | Acicular petroleum coke and processing technology thereof |
| CN114175337A (en) * | 2019-08-06 | 2022-03-11 | Apb株式会社 | Manufacturing method of lithium ion battery |
| JP6705068B1 (en) * | 2020-01-17 | 2020-06-03 | 住友化学株式会社 | Positive electrode active material for all-solid-state lithium-ion battery, electrode and all-solid-state lithium-ion battery |
| CN111485173B (en) * | 2020-04-09 | 2020-12-08 | 广东德纳斯金属制品有限公司 | A new type of constant temperature material and its preparation method and application |
| KR102807564B1 (en) * | 2020-06-05 | 2025-05-13 | 닝더 엠프렉스 테크놀로지 리미티드 | Electrochemical devices and electronic devices |
| CN114730860B (en) * | 2020-06-23 | 2024-02-23 | 宁德时代新能源科技股份有限公司 | Secondary battery and device comprising same |
| CN113889595A (en) * | 2020-07-02 | 2022-01-04 | 洛阳月星新能源科技有限公司 | Preparation method of solid electrolyte coated graphite composite material |
| CN111769328B (en) * | 2020-07-10 | 2022-02-08 | 宁德新能源科技有限公司 | Electrolyte, electrochemical device and electronic device |
| EP4228037A4 (en) * | 2020-10-15 | 2024-10-30 | Ningde Amperex Technology Limited | Electrochemical device and electronic device |
| CN112151751B (en) * | 2020-10-15 | 2022-08-05 | 宁德新能源科技有限公司 | Electrochemical device and electronic device |
| CN112467214B (en) * | 2020-11-30 | 2022-03-01 | 远景动力技术(江苏)有限公司 | Electrolyte solution and lithium ion battery using same |
| KR102563523B1 (en) * | 2020-12-29 | 2023-08-10 | 한국과학기술연구원 | Method for manufacturing composite electrode for secondary battery, and composite electrode for secondary battery manufactured thereby |
| CN112928234B (en) * | 2021-02-01 | 2022-10-18 | 河南克能新能源科技有限公司 | Preparation method of positive electrode of lithium ion battery |
| CN112993381A (en) * | 2021-02-06 | 2021-06-18 | 苏州精诚智造智能科技有限公司 | Preparation method of high-rate lithium ion battery |
| CN112952198B (en) * | 2021-03-22 | 2023-03-10 | 华中科技大学 | Sulfur energy storage battery and method for improving rate capability and cycle stability of sulfur energy storage battery |
| CN113173108B (en) * | 2021-04-23 | 2022-12-13 | 中车青岛四方机车车辆股份有限公司 | Multi-stack fuel cell control method, control device, system and vehicle |
| CN113328143A (en) * | 2021-05-28 | 2021-08-31 | 远景动力技术(江苏)有限公司 | Non-aqueous electrolyte for lithium battery, preparation method of non-aqueous electrolyte and lithium ion battery |
| CN114628784B (en) * | 2021-10-28 | 2024-04-30 | 浙江超威创元实业有限公司 | High-low temperature general electrolyte and lithium ion battery containing same |
| CN114665064B (en) * | 2022-05-26 | 2022-08-30 | 宁德新能源科技有限公司 | Electrochemical device |
| CN116666729A (en) * | 2022-09-26 | 2023-08-29 | 欣旺达电动汽车电池有限公司 | Secondary battery and electricity utilization device |
| CN115458732B (en) * | 2022-10-31 | 2025-05-23 | 中南大学 | Sodium ion battery monocrystal positive electrode active material, and preparation method and application thereof |
| CN119330381B (en) * | 2022-12-05 | 2026-01-27 | 宁德时代新能源科技股份有限公司 | Alumina particles, method for producing the same, separator, battery, and electricity-using device |
| CN115954543A (en) * | 2022-12-28 | 2023-04-11 | 珠海冠宇电池股份有限公司 | a battery |
| WO2024229420A2 (en) * | 2023-05-03 | 2024-11-07 | The Board Of Trustees Of The Leland Stanford Junior University | Plasma-assisted rapid processing of high-throughput, solution deposited solid-state ionic conductors |
| CN117013066A (en) * | 2023-08-08 | 2023-11-07 | 常州千沐新能源有限公司 | Eutectic high-entropy electrolyte, preparation method and lithium ion battery |
| CN117154214B (en) * | 2023-10-30 | 2024-07-16 | 宁德时代新能源科技股份有限公司 | Electrolyte, secondary battery and electricity utilization device |
| CN118572192A (en) * | 2023-12-19 | 2024-08-30 | 宁德新能源科技有限公司 | Secondary battery and electronic device including the same |
| CN119764741B (en) * | 2024-08-22 | 2025-12-16 | 比亚迪股份有限公司 | A lithium-ion battery separator, its preparation method, and a lithium-ion battery using the same. |
| CN118825243B (en) * | 2024-09-10 | 2025-01-24 | 山东海化集团有限公司 | Preparation method and application of a carbon-supported FeCo diatomic porous lithium-sulfur battery composite material |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1567643A (en) * | 2003-01-09 | 2005-01-19 | 三星Sdi株式会社 | Electrolyte for rechargeable lithium battery and rechargeable lithium battery comprising same |
Family Cites Families (23)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE69605617T2 (en) * | 1995-07-24 | 2000-06-29 | Sumitomo Chemical Co., Ltd. | Lithium secondary battery with non-aqueous electrolyte |
| JP3975502B2 (en) * | 1997-03-17 | 2007-09-12 | ソニー株式会社 | Non-aqueous electrolyte secondary battery |
| US6150053A (en) * | 1997-06-06 | 2000-11-21 | Matsushita Electric Industrial Co., Ltd. | Non-aqueous electrolyte secondary battery |
| JP3439085B2 (en) * | 1997-08-21 | 2003-08-25 | 三洋電機株式会社 | Non-aqueous electrolyte secondary battery |
| JP3978881B2 (en) * | 1997-08-22 | 2007-09-19 | 宇部興産株式会社 | Non-aqueous electrolyte and lithium secondary battery using the same |
| US6060184A (en) * | 1998-07-09 | 2000-05-09 | Wilson Greatbatch Ltd. | Inorganic and organic nitrate additives for nonaqueous electrolyte in alkali metal electrochemical cells |
| JP2000243446A (en) * | 1998-12-25 | 2000-09-08 | Matsushita Electric Ind Co Ltd | Lithium secondary battery |
| NL1013484C2 (en) * | 1999-11-04 | 2001-05-09 | Tno | Method of manufacturing a rechargeable 3V Li-ion battery. |
| EP1357628A4 (en) * | 2001-01-04 | 2008-03-12 | Mitsubishi Chem Corp | WATER-FREE ELECTROLYTIC LIQUIDS AND LITHIUM SECONDARY BATTERIES THEREWITH |
| DE10209477A1 (en) * | 2002-03-05 | 2003-09-18 | Chemetall Gmbh | Electrochemical cell for a lithium ion battery with improved high temperature stability |
| AU2003221334A1 (en) * | 2002-03-08 | 2003-09-22 | Mitsubishi Chemical Corporation | Nonaqueous electrolyte and lithium-ion secondary battery containing the same |
| JP4233819B2 (en) * | 2002-06-25 | 2009-03-04 | 三菱化学株式会社 | Non-aqueous electrolyte secondary battery |
| WO2004030125A1 (en) * | 2002-09-26 | 2004-04-08 | Seimi Chemical Co., Ltd. | Positive electrode active substance for lithium secondary battery and process for producing the same |
| CN1294665C (en) * | 2003-08-15 | 2007-01-10 | 比亚迪股份有限公司 | Anode active material for non-aqueous secondary cell, and its preparing method and non-aqueous secondary cell using same |
| JP4450550B2 (en) * | 2002-11-21 | 2010-04-14 | 三井化学株式会社 | Non-aqueous electrolyte and secondary battery using the same |
| KR100515298B1 (en) * | 2003-03-24 | 2005-09-15 | 삼성에스디아이 주식회사 | A non-aqueous electrolyte and a lithium secondary battery comprising the same |
| US7374842B2 (en) * | 2003-04-30 | 2008-05-20 | Matsushita Battery Industrial Co., Ltd. | Non-aqueous electrolyte secondary battery |
| JP4283598B2 (en) * | 2003-05-29 | 2009-06-24 | Tdk株式会社 | Non-aqueous electrolyte solution and lithium ion secondary battery |
| KR100508923B1 (en) * | 2003-06-27 | 2005-08-17 | 삼성에스디아이 주식회사 | A non-aqueous electrolyte and a lithium secondary battery comprising the same |
| JP4407205B2 (en) * | 2003-08-22 | 2010-02-03 | 三菱化学株式会社 | Nonaqueous electrolyte for lithium secondary battery and lithium secondary battery using the same |
| JP2005123180A (en) * | 2003-09-26 | 2005-05-12 | Mitsubishi Chemicals Corp | Lithium composite oxide particles for lithium secondary battery positive electrode material, method for producing the same, and positive electrode for lithium secondary battery and lithium secondary battery using the same |
| JP2005251456A (en) * | 2004-03-02 | 2005-09-15 | Mitsubishi Chemicals Corp | Nonaqueous electrolytic solution for lithium secondary battery, and lithium secondary battery using the same |
| JP4794866B2 (en) * | 2004-04-08 | 2011-10-19 | パナソニック株式会社 | Cathode active material for non-aqueous electrolyte secondary battery, method for producing the same, and non-aqueous electrolyte secondary battery using the same |
-
2006
- 2006-10-19 CN CN2006800390896A patent/CN101292389B/en not_active Ceased
- 2006-10-19 CN CN201710119547.7A patent/CN107069091A/en active Pending
- 2006-10-19 CN CN202110664613.5A patent/CN113594543A/en active Pending
- 2006-10-19 CN CN202110664313.7A patent/CN113394458A/en active Pending
- 2006-10-19 CN CN201910201995.0A patent/CN110061283A/en active Pending
- 2006-10-19 CN CN202110646170.7A patent/CN113571772A/en active Pending
- 2006-10-19 CN CN201910202173.4A patent/CN110010850A/en active Pending
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1567643A (en) * | 2003-01-09 | 2005-01-19 | 三星Sdi株式会社 | Electrolyte for rechargeable lithium battery and rechargeable lithium battery comprising same |
Non-Patent Citations (3)
| Title |
|---|
| JP特开2004-171981A 2004.06.17 |
| JP特开2004-71458A 2004.03.04 |
| JP特开2005-123180A 2005.05.12 |
Cited By (35)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102751532A (en) * | 2011-04-22 | 2012-10-24 | 株式会社日立制作所 | Lithium ion secondary battery and battery pack system |
| US12084549B2 (en) | 2012-02-09 | 2024-09-10 | Group 14 Technologies, Inc. | Preparation of polymeric resins and carbon materials |
| US11725074B2 (en) | 2012-02-09 | 2023-08-15 | Group 14 Technologies, Inc. | Preparation of polymeric resins and carbon materials |
| US11718701B2 (en) | 2012-02-09 | 2023-08-08 | Group14 Technologies, Inc. | Preparation of polymeric resins and carbon materials |
| US11732079B2 (en) | 2012-02-09 | 2023-08-22 | Group14 Technologies, Inc. | Preparation of polymeric resins and carbon materials |
| US11999828B2 (en) | 2012-02-09 | 2024-06-04 | Group14 Technologies, Inc. | Preparation of polymeric resins and carbon materials |
| US12006400B2 (en) | 2012-02-09 | 2024-06-11 | Group14 Technologies, Inc. | Preparation of polymeric resins and carbon materials |
| US12497479B2 (en) | 2012-02-09 | 2025-12-16 | Group14 Technologies, Inc. | Preparation of polymeric resins and carbon materials |
| TWI508359B (en) * | 2012-07-23 | 2015-11-11 | Furukawa Electric Co Ltd | Surface-treated copper foil and method for manufacturing the same, lithium ion secondary battery electrode, and lithium ion secondary battery |
| WO2014094221A1 (en) * | 2012-12-18 | 2014-06-26 | 湖州创亚动力电池材料有限公司 | Composite graphite cathode material for high-capacity lithium ion battery, and preparation method thereof |
| US11495793B2 (en) | 2013-03-14 | 2022-11-08 | Group14 Technologies, Inc. | Composite carbon materials comprising lithium alloying electrochemical modifiers |
| US12418023B2 (en) | 2013-03-14 | 2025-09-16 | Group14 Technologies, Inc. | Composite carbon materials comprising lithium alloying electrochemical modifiers |
| US10686223B2 (en) | 2013-09-25 | 2020-06-16 | Kabushiki Kaisha Toyota Jidoshokki | Nonaqueous electrolyte secondary battery |
| US11011781B2 (en) | 2013-09-25 | 2021-05-18 | The University Of Tokyo | Nonaqueous electrolyte secondary battery |
| CN105580184B (en) * | 2013-09-25 | 2019-03-12 | 国立大学法人东京大学 | Non-aqueous electrolyte secondary battery |
| CN105580184A (en) * | 2013-09-25 | 2016-05-11 | 国立大学法人东京大学 | Non-aqueous electrolyte secondary battery |
| US12064747B2 (en) | 2013-11-05 | 2024-08-20 | Group14 Technologies, Inc. | Carbon-based compositions with highly efficient volumetric gas sorption |
| US11707728B2 (en) | 2013-11-05 | 2023-07-25 | Group14 Technologies, Inc. | Carbon-based compositions with highly efficient volumetric gas sorption |
| US12173165B2 (en) | 2014-03-14 | 2024-12-24 | Group14 Technologies, Inc. | Methods for sol-gel polymerization in absence of solvent and creation of tunable carbon structure from same |
| US11661517B2 (en) | 2014-03-14 | 2023-05-30 | Group14 Technologies, Inc. | Methods for sol-gel polymerization in absence of solvent and creation of tunable carbon structure from same |
| US11611073B2 (en) | 2015-08-14 | 2023-03-21 | Group14 Technologies, Inc. | Composites of porous nano-featured silicon materials and carbon materials |
| US12537192B2 (en) | 2015-08-14 | 2026-01-27 | Group14 Technologies, Inc. | Composites of porous nano-featured silicon materials and carbon materials |
| US11437621B2 (en) | 2015-08-28 | 2022-09-06 | Group14 Technologies, Inc. | Materials with extremely durable intercalation of lithium and manufacturing methods thereof |
| US11646419B2 (en) | 2015-08-28 | 2023-05-09 | Group 14 Technologies, Inc. | Materials with extremely durable intercalation of lithium and manufacturing methods thereof |
| US11495798B1 (en) | 2015-08-28 | 2022-11-08 | Group14 Technologies, Inc. | Materials with extremely durable intercalation of lithium and manufacturing methods thereof |
| US11611071B2 (en) | 2017-03-09 | 2023-03-21 | Group14 Technologies, Inc. | Decomposition of silicon-containing precursors on porous scaffold materials |
| US12155066B2 (en) | 2017-03-09 | 2024-11-26 | Group14 Technologies, Inc. | Decomposition of silicon-containing precursors on porous scaffold materials |
| US11804591B2 (en) | 2020-08-18 | 2023-10-31 | Group14 Technologies, Inc. | Highly efficient manufacturing of silicon-carbon composite materials comprising ultra low Z |
| US11639292B2 (en) | 2020-08-18 | 2023-05-02 | Group14 Technologies, Inc. | Particulate composite materials |
| US12057569B2 (en) | 2020-08-18 | 2024-08-06 | Group14 Technologies, Inc. | Highly efficient manufacturing of silicon-carbon composite materials comprising ultra low Z |
| US11611070B2 (en) | 2020-08-18 | 2023-03-21 | Group14 Technologies, Inc. | Highly efficient manufacturing of silicon-carbon composites materials comprising ultra low Z |
| US11498838B2 (en) | 2020-08-18 | 2022-11-15 | Group14 Technologies, Inc. | Silicon carbon composites comprising ultra low z |
| US11492262B2 (en) | 2020-08-18 | 2022-11-08 | Group14Technologies, Inc. | Silicon carbon composites comprising ultra low Z |
| US11335903B2 (en) | 2020-08-18 | 2022-05-17 | Group14 Technologies, Inc. | Highly efficient manufacturing of silicon-carbon composites materials comprising ultra low z |
| US12046744B2 (en) | 2020-09-30 | 2024-07-23 | Group14 Technologies, Inc. | Passivated silicon-carbon composite materials |
Also Published As
| Publication number | Publication date |
|---|---|
| CN107069091A (en) | 2017-08-18 |
| CN110010850A (en) | 2019-07-12 |
| CN113571772A (en) | 2021-10-29 |
| CN101292389A (en) | 2008-10-22 |
| CN113594543A (en) | 2021-11-02 |
| CN113394458A (en) | 2021-09-14 |
| CN110061283A (en) | 2019-07-26 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN101292389B (en) | Lithium secondary battery and nonaqueous electrolyte used therein | |
| CN102931434B (en) | Lithium secondary battery and non-aqueous electrolyte used therein | |
| US10720645B2 (en) | Carbon material for non-aqueous electrolyte secondary battery, negative electrode for non-aqueous electrolyte secondary battery, non-aqueous electrolyte secondary battery, and manufacturing method for carbon material for non-aqueous electrolyte secondary battery | |
| CN101894974A (en) | Lithium secondary battery and nonaqueous electrolyte used therein | |
| JP5671772B2 (en) | Lithium ion secondary battery | |
| JP2007200871A (en) | Lithium ion secondary battery |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| CP01 | Change in the name or title of a patent holder |
Address after: Tokyo, Japan Patentee after: MITSUBISHI CHEMICAL Corp. Address before: Tokyo, Japan Patentee before: MITSUBISHI RAYON Co.,Ltd. |
|
| CP01 | Change in the name or title of a patent holder | ||
| TR01 | Transfer of patent right |
Effective date of registration: 20170830 Address after: Tokyo, Japan Patentee after: MITSUBISHI RAYON Co.,Ltd. Address before: Tokyo, Japan Patentee before: MITSUBISHI CHEMICAL Corp. |
|
| TR01 | Transfer of patent right | ||
| TR01 | Transfer of patent right |
Effective date of registration: 20210118 Address after: Tokyo, Japan Patentee after: MITSUBISHI CHEMICAL Corp. Patentee after: Mu electrolyte Co.,Ltd. Address before: Tokyo, Japan Patentee before: MITSUBISHI CHEMICAL Corp. |
|
| TR01 | Transfer of patent right | ||
| IW01 | Full invalidation of patent right |
Decision date of declaring invalidation: 20220729 Decision number of declaring invalidation: 57429 Granted publication date: 20100922 |
|
| IW01 | Full invalidation of patent right |