CN101234105A - Pharmaceutical composition containing diabetosan and vildagliptin and preparation thereof - Google Patents
Pharmaceutical composition containing diabetosan and vildagliptin and preparation thereof Download PDFInfo
- Publication number
- CN101234105A CN101234105A CNA2008100558154A CN200810055815A CN101234105A CN 101234105 A CN101234105 A CN 101234105A CN A2008100558154 A CNA2008100558154 A CN A2008100558154A CN 200810055815 A CN200810055815 A CN 200810055815A CN 101234105 A CN101234105 A CN 101234105A
- Authority
- CN
- China
- Prior art keywords
- vildagliptin
- pharmaceutical composition
- metformin
- metformin hydrochloride
- tablet
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- SYOKIDBDQMKNDQ-XWTIBIIYSA-N vildagliptin Chemical compound C1C(O)(C2)CC(C3)CC1CC32NCC(=O)N1CCC[C@H]1C#N SYOKIDBDQMKNDQ-XWTIBIIYSA-N 0.000 title claims abstract description 34
- 229960001254 vildagliptin Drugs 0.000 title claims abstract description 34
- OETHQSJEHLVLGH-UHFFFAOYSA-N metformin hydrochloride Chemical compound Cl.CN(C)C(=N)N=C(N)N OETHQSJEHLVLGH-UHFFFAOYSA-N 0.000 title claims description 26
- 239000008194 pharmaceutical composition Substances 0.000 title claims description 17
- 238000002360 preparation method Methods 0.000 title abstract description 12
- XZWYZXLIPXDOLR-UHFFFAOYSA-N metformin Chemical compound CN(C)C(=N)NC(N)=N XZWYZXLIPXDOLR-UHFFFAOYSA-N 0.000 claims abstract description 43
- 239000000203 mixture Substances 0.000 claims abstract description 28
- 239000008187 granular material Substances 0.000 claims abstract description 13
- 239000002775 capsule Substances 0.000 claims abstract description 7
- 239000003826 tablet Substances 0.000 claims abstract description 7
- 239000007910 chewable tablet Substances 0.000 claims abstract description 4
- 239000007919 dispersible tablet Substances 0.000 claims abstract description 4
- 239000007938 effervescent tablet Substances 0.000 claims abstract description 4
- 239000006189 buccal tablet Substances 0.000 claims abstract description 3
- 238000002156 mixing Methods 0.000 claims abstract description 3
- 229960004329 metformin hydrochloride Drugs 0.000 claims description 26
- 150000001875 compounds Chemical class 0.000 claims description 14
- 208000001072 type 2 diabetes mellitus Diseases 0.000 claims description 9
- 238000009472 formulation Methods 0.000 claims description 8
- 229940068682 chewable tablet Drugs 0.000 claims description 3
- 235000005911 diet Nutrition 0.000 claims description 3
- 230000037213 diet Effects 0.000 claims description 3
- 230000015572 biosynthetic process Effects 0.000 claims description 2
- 229940046011 buccal tablet Drugs 0.000 claims description 2
- 239000007787 solid Substances 0.000 claims 1
- 206010012601 diabetes mellitus Diseases 0.000 abstract description 25
- 229960003105 metformin Drugs 0.000 abstract description 16
- 238000000034 method Methods 0.000 abstract description 6
- 239000000463 material Substances 0.000 abstract description 5
- 229940035752 metformin and vildagliptin Drugs 0.000 abstract description 3
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 abstract 3
- 241000700159 Rattus Species 0.000 description 17
- NOESYZHRGYRDHS-UHFFFAOYSA-N insulin Chemical compound N1C(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(NC(=O)CN)C(C)CC)CSSCC(C(NC(CO)C(=O)NC(CC(C)C)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CCC(N)=O)C(=O)NC(CC(C)C)C(=O)NC(CCC(O)=O)C(=O)NC(CC(N)=O)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CSSCC(NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2C=CC(O)=CC=2)NC(=O)C(CC(C)C)NC(=O)C(C)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2NC=NC=2)NC(=O)C(CO)NC(=O)CNC2=O)C(=O)NCC(=O)NC(CCC(O)=O)C(=O)NC(CCCNC(N)=N)C(=O)NCC(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC(O)=CC=3)C(=O)NC(C(C)O)C(=O)N3C(CCC3)C(=O)NC(CCCCN)C(=O)NC(C)C(O)=O)C(=O)NC(CC(N)=O)C(O)=O)=O)NC(=O)C(C(C)CC)NC(=O)C(CO)NC(=O)C(C(C)O)NC(=O)C1CSSCC2NC(=O)C(CC(C)C)NC(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CC(N)=O)NC(=O)C(NC(=O)C(N)CC=1C=CC=CC=1)C(C)C)CC1=CN=CN1 NOESYZHRGYRDHS-UHFFFAOYSA-N 0.000 description 16
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 11
- 239000008103 glucose Substances 0.000 description 11
- 210000004369 blood Anatomy 0.000 description 10
- 239000008280 blood Substances 0.000 description 10
- 239000000243 solution Substances 0.000 description 10
- 102000004877 Insulin Human genes 0.000 description 8
- 108090001061 Insulin Proteins 0.000 description 8
- 229940125396 insulin Drugs 0.000 description 8
- 230000037396 body weight Effects 0.000 description 7
- 102000007562 Serum Albumin Human genes 0.000 description 6
- 108010071390 Serum Albumin Proteins 0.000 description 6
- 230000001476 alcoholic effect Effects 0.000 description 5
- 238000001035 drying Methods 0.000 description 5
- 239000007779 soft material Substances 0.000 description 5
- 210000002784 stomach Anatomy 0.000 description 5
- ZSJLQEPLLKMAKR-GKHCUFPYSA-N streptozocin Chemical compound O=NN(C)C(=O)N[C@H]1[C@@H](O)O[C@H](CO)[C@@H](O)[C@@H]1O ZSJLQEPLLKMAKR-GKHCUFPYSA-N 0.000 description 5
- ZSJLQEPLLKMAKR-UHFFFAOYSA-N Streptozotocin Natural products O=NN(C)C(=O)NC1C(O)OC(CO)C(O)C1O ZSJLQEPLLKMAKR-UHFFFAOYSA-N 0.000 description 4
- 230000000694 effects Effects 0.000 description 4
- 229960001052 streptozocin Drugs 0.000 description 4
- 108010067722 Dipeptidyl Peptidase 4 Proteins 0.000 description 3
- 102100025012 Dipeptidyl peptidase 4 Human genes 0.000 description 3
- RWRDLPDLKQPQOW-UHFFFAOYSA-N Pyrrolidine Chemical compound C1CCNC1 RWRDLPDLKQPQOW-UHFFFAOYSA-N 0.000 description 3
- 239000003795 chemical substances by application Substances 0.000 description 3
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 3
- 201000010099 disease Diseases 0.000 description 3
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 3
- XMGQYMWWDOXHJM-UHFFFAOYSA-N limonene Chemical compound CC(=C)C1CCC(C)=CC1 XMGQYMWWDOXHJM-UHFFFAOYSA-N 0.000 description 3
- 239000011812 mixed powder Substances 0.000 description 3
- 239000007968 orange flavor Substances 0.000 description 3
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 3
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 2
- 229930195725 Mannitol Natural products 0.000 description 2
- 229920000168 Microcrystalline cellulose Polymers 0.000 description 2
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 2
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 2
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 2
- 235000010358 acesulfame potassium Nutrition 0.000 description 2
- 239000002671 adjuvant Substances 0.000 description 2
- 230000000295 complement effect Effects 0.000 description 2
- 229940079593 drug Drugs 0.000 description 2
- 239000003814 drug Substances 0.000 description 2
- 230000002708 enhancing effect Effects 0.000 description 2
- 210000001508 eye Anatomy 0.000 description 2
- 201000001421 hyperglycemia Diseases 0.000 description 2
- 230000001965 increasing effect Effects 0.000 description 2
- 230000002045 lasting effect Effects 0.000 description 2
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 2
- 239000000594 mannitol Substances 0.000 description 2
- 235000010355 mannitol Nutrition 0.000 description 2
- 229960001855 mannitol Drugs 0.000 description 2
- 208000030159 metabolic disease Diseases 0.000 description 2
- 235000019813 microcrystalline cellulose Nutrition 0.000 description 2
- 239000008108 microcrystalline cellulose Substances 0.000 description 2
- 229940016286 microcrystalline cellulose Drugs 0.000 description 2
- 238000012856 packing Methods 0.000 description 2
- 239000000741 silica gel Substances 0.000 description 2
- 229910002027 silica gel Inorganic materials 0.000 description 2
- 210000001519 tissue Anatomy 0.000 description 2
- KPYXMALABCDPGN-HYOZMBHHSA-N (4s)-5-[[(2s)-6-amino-1-[[(2s,3s)-1-[[(2s)-1-[[(2s)-1-[[(2s)-1-[[(2s)-1-[[(2r)-1-[[2-[[2-[[(1s)-3-amino-1-carboxy-3-oxopropyl]amino]-2-oxoethyl]amino]-2-oxoethyl]amino]-1-oxo-3-sulfanylpropan-2-yl]amino]-4-methyl-1-oxopentan-2-yl]amino]-1-oxopropan-2-yl]a Chemical compound NC(=O)C[C@@H](C(O)=O)NC(=O)CNC(=O)CNC(=O)[C@H](CS)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](C)NC(=O)[C@H](C(C)C)NC(=O)[C@H](CC(C)C)NC(=O)[C@H]([C@@H](C)CC)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@@H](NC(=O)[C@H](CC(O)=O)NC(=O)CNC(=O)[C@@H](N)CCCCN)CC1=CC=C(O)C=C1 KPYXMALABCDPGN-HYOZMBHHSA-N 0.000 description 1
- WBZFUFAFFUEMEI-UHFFFAOYSA-M Acesulfame k Chemical compound [K+].CC1=CC(=O)[N-]S(=O)(=O)O1 WBZFUFAFFUEMEI-UHFFFAOYSA-M 0.000 description 1
- 241000486679 Antitype Species 0.000 description 1
- 208000037157 Azotemia Diseases 0.000 description 1
- 206010008190 Cerebrovascular accident Diseases 0.000 description 1
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 1
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 1
- 101710198884 GATA-type zinc finger protein 1 Proteins 0.000 description 1
- 102000051325 Glucagon Human genes 0.000 description 1
- 108060003199 Glucagon Proteins 0.000 description 1
- DTHNMHAUYICORS-KTKZVXAJSA-N Glucagon-like peptide 1 Chemical compound C([C@@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](C)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCCCN)C(=O)NCC(=O)N[C@@H](CCCNC(N)=N)C(N)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](C)NC(=O)[C@H](C)NC(=O)[C@H](CCC(N)=O)NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](CC=1C=CC=CC=1)NC(=O)[C@@H](NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H](N)CC=1N=CNC=1)[C@@H](C)O)[C@@H](C)O)C(C)C)C1=CC=CC=C1 DTHNMHAUYICORS-KTKZVXAJSA-N 0.000 description 1
- 241001465754 Metazoa Species 0.000 description 1
- 241000699666 Mus <mouse, genus> Species 0.000 description 1
- 241000699670 Mus sp. Species 0.000 description 1
- 239000008118 PEG 6000 Substances 0.000 description 1
- 229920002584 Polyethylene Glycol 6000 Polymers 0.000 description 1
- 102100040918 Pro-glucagon Human genes 0.000 description 1
- 241000700157 Rattus norvegicus Species 0.000 description 1
- UIIMBOGNXHQVGW-DEQYMQKBSA-M Sodium bicarbonate-14C Chemical compound [Na+].O[14C]([O-])=O UIIMBOGNXHQVGW-DEQYMQKBSA-M 0.000 description 1
- UEDUENGHJMELGK-HYDKPPNVSA-N Stevioside Chemical compound O([C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@H]1O[C@]12C(=C)C[C@@]3(C1)CC[C@@H]1[C@@](C)(CCC[C@]1([C@@H]3CC2)C)C(=O)O[C@H]1[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O1)O)[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O UEDUENGHJMELGK-HYDKPPNVSA-N 0.000 description 1
- 208000006011 Stroke Diseases 0.000 description 1
- TVXBFESIOXBWNM-UHFFFAOYSA-N Xylitol Natural products OCCC(O)C(O)C(O)CCO TVXBFESIOXBWNM-UHFFFAOYSA-N 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- 229960004998 acesulfame potassium Drugs 0.000 description 1
- 239000000619 acesulfame-K Substances 0.000 description 1
- 125000002777 acetyl group Chemical group [H]C([H])([H])C(*)=O 0.000 description 1
- 210000000577 adipose tissue Anatomy 0.000 description 1
- 238000013459 approach Methods 0.000 description 1
- 210000000227 basophil cell of anterior lobe of hypophysis Anatomy 0.000 description 1
- 210000004204 blood vessel Anatomy 0.000 description 1
- 238000011095 buffer preparation Methods 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 230000019522 cellular metabolic process Effects 0.000 description 1
- 230000001684 chronic effect Effects 0.000 description 1
- 239000007979 citrate buffer Substances 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- -1 compound metformin hydrochloride Chemical class 0.000 description 1
- 239000007891 compressed tablet Substances 0.000 description 1
- 230000002950 deficient Effects 0.000 description 1
- 230000006735 deficit Effects 0.000 description 1
- 238000006731 degradation reaction Methods 0.000 description 1
- MTHSVFCYNBDYFN-UHFFFAOYSA-N diethylene glycol Chemical compound OCCOCCO MTHSVFCYNBDYFN-UHFFFAOYSA-N 0.000 description 1
- 230000004069 differentiation Effects 0.000 description 1
- 239000012153 distilled water Substances 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 239000007941 film coated tablet Substances 0.000 description 1
- 230000037406 food intake Effects 0.000 description 1
- 230000030136 gastric emptying Effects 0.000 description 1
- MASNOZXLGMXCHN-ZLPAWPGGSA-N glucagon Chemical compound C([C@@H](C(=O)N[C@H](C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(O)=O)C(C)C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](C)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CO)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CO)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](CC=1C=CC=CC=1)NC(=O)[C@@H](NC(=O)CNC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](N)CC=1NC=NC=1)[C@@H](C)O)[C@@H](C)O)C1=CC=CC=C1 MASNOZXLGMXCHN-ZLPAWPGGSA-N 0.000 description 1
- 229960004666 glucagon Drugs 0.000 description 1
- 230000034659 glycolysis Effects 0.000 description 1
- 238000006206 glycosylation reaction Methods 0.000 description 1
- 210000002216 heart Anatomy 0.000 description 1
- 230000002218 hypoglycaemic effect Effects 0.000 description 1
- MGXWVYUBJRZYPE-YUGYIWNOSA-N incretin Chemical class C([C@@H](C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](C)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CC=1NC=NC=1)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCCCN)C(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC=1NC=NC=1)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCC(N)=O)C(O)=O)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](CC=1C=CC=CC=1)NC(=O)[C@@H](NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H](N)CC=1C=CC(O)=CC=1)[C@@H](C)O)[C@@H](C)CC)C1=CC=C(O)C=C1 MGXWVYUBJRZYPE-YUGYIWNOSA-N 0.000 description 1
- 239000000859 incretin Substances 0.000 description 1
- 239000003112 inhibitor Substances 0.000 description 1
- 238000002347 injection Methods 0.000 description 1
- 239000007924 injection Substances 0.000 description 1
- 238000007689 inspection Methods 0.000 description 1
- 230000003914 insulin secretion Effects 0.000 description 1
- 210000003734 kidney Anatomy 0.000 description 1
- 150000002632 lipids Chemical class 0.000 description 1
- 210000003141 lower extremity Anatomy 0.000 description 1
- 235000019359 magnesium stearate Nutrition 0.000 description 1
- 206010025482 malaise Diseases 0.000 description 1
- HEBKCHPVOIAQTA-UHFFFAOYSA-N meso ribitol Natural products OCC(O)C(O)C(O)CO HEBKCHPVOIAQTA-UHFFFAOYSA-N 0.000 description 1
- 238000000465 moulding Methods 0.000 description 1
- 230000003387 muscular Effects 0.000 description 1
- 208000010125 myocardial infarction Diseases 0.000 description 1
- 210000005036 nerve Anatomy 0.000 description 1
- 210000000056 organ Anatomy 0.000 description 1
- 210000000496 pancreas Anatomy 0.000 description 1
- 230000000144 pharmacologic effect Effects 0.000 description 1
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 1
- 102000004169 proteins and genes Human genes 0.000 description 1
- 108090000623 proteins and genes Proteins 0.000 description 1
- 238000011552 rat model Methods 0.000 description 1
- 238000011084 recovery Methods 0.000 description 1
- 239000012744 reinforcing agent Substances 0.000 description 1
- 230000028327 secretion Effects 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 229910000029 sodium carbonate Inorganic materials 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 235000002639 sodium chloride Nutrition 0.000 description 1
- 239000007909 solid dosage form Substances 0.000 description 1
- 239000000600 sorbitol Substances 0.000 description 1
- 229960002920 sorbitol Drugs 0.000 description 1
- 229940013618 stevioside Drugs 0.000 description 1
- OHHNJQXIOPOJSC-UHFFFAOYSA-N stevioside Natural products CC1(CCCC2(C)C3(C)CCC4(CC3(CCC12C)CC4=C)OC5OC(CO)C(O)C(O)C5OC6OC(CO)C(O)C(O)C6O)C(=O)OC7OC(CO)C(O)C(O)C7O OHHNJQXIOPOJSC-UHFFFAOYSA-N 0.000 description 1
- 235000019202 steviosides Nutrition 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 230000001629 suppression Effects 0.000 description 1
- 208000011580 syndromic disease Diseases 0.000 description 1
- 208000009852 uremia Diseases 0.000 description 1
- 210000003462 vein Anatomy 0.000 description 1
- 239000000811 xylitol Substances 0.000 description 1
- 229960002675 xylitol Drugs 0.000 description 1
- 235000010447 xylitol Nutrition 0.000 description 1
- HEBKCHPVOIAQTA-SCDXWVJYSA-N xylitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)CO HEBKCHPVOIAQTA-SCDXWVJYSA-N 0.000 description 1
Landscapes
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
Abstract
The invention relates to medical composition containing metformin and Vildagliptin and a preparation method thereof. The medical composition is formed by taking metformin chloride and Vildagliptin as medical active components and blending with pharmaceutically acceptable auxiliary materials; the invention adopts the metformin chloride and Vildagliptin as materials and adds auxiliary materials with some special kinds and proportions to prepare and develop diversified oral preparations such as tablets, capsules, granules, dispersible tablets, chewable tablets, buccal tablets, effervescent tablets, effervescent granules according to the technical method of the invention. The medical composition of the invention can be used for treating diabetes type II that can not be properly cured by alimentary control and sports and also for diabetes type II that can not be controlled by simply using metformin chloride or Vildagliptin.
Description
Technical field
The present invention relates to a kind of is Pharmaceutical composition of active component and preparation method thereof, purposes with metformin hydrochloride and vildagliptin, belongs to medical technical field.
Background technology
Diabetes (Diabetes mellitus) be since a kind of syndrome of the caused glucose of cellular metabolism effect defective, protein and the lipid metabolic disorder of insufficient insulin or insulin along with the fall ill prolongation of time of diabetes, the intravital metabolism disorder of body is controlled well as can not get, the chronic complicating diseases that can cause tissues such as eye, kidney, nerve, blood vessel and heart, organ, so that final take place blind, lower limb are gangrenous, uremia, apoplexy or myocardial infarction, even threat to life.Diabetes are a kind of commonly encountered diseases, and along with growth in the living standard, the sickness rate of diabetes increases year by year.The prevalence of developed country's diabetes is up to 5%~10%, and the prevalence of China has reached 3%.
Type 2 diabetes mellitus also is adult's morbidity type diabetes, how to fall ill after 35~40 years old, accounts for diabetics more than 90%.The ability that produces insulin in the type 2 diabetes mellitus patient body is not to completely lose, insulin even generation are too much in the patient's body that has, but because the insulin resistant effect of body enhancing, and make the action effect of insulin not obvious, so the intravital insulin deficit of patient is a kind of relative shortage.
Compound recipe of the present invention is to be made up of two kinds of hypoglycemic medicines, and two kinds of drug mechanism differences can play complementary effect.Be used for the treatment of diet control and the type 2 diabetes mellitus that still can not suitably control of motion, also can be used for taking separately metformin hydrochloride or the out of contior type 2 diabetes mellitus of vildagliptin.
Metformin hydrochloride does not promote insulin secretion, and its blood sugar reducing function is mainly and promotes the fatty tissue ingestion of glucose, and muscular tissue anerobic glycolysis is increased, and increases the utilization of glucose, can also reduce glucose through gastral absorption.
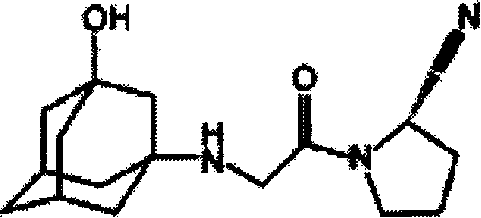
Vildagliptin (vildagliptin) is a kind of DPP IV (DPP-IV) inhibitor, also claim the incretin reinforcing agent, by suppressing the activity of DPP-IV, reduce the degradation speed of pancreas hyperglycemia sample peptide I GLP-1, and then under hyperglycemia concentration, stimulate secretion of insulin, and can discharge, promote beta Cell of islet propagation and approach such as differentiation and enhancing satietion to bring into play anti-type 2 diabetes mellitus function by delay gastric emptying, glucagon suppression.This product chemistry 1-[[(3-hydroxyl by name-1-adamantyl) amino] acetyl group]-2-cyano group-(S)-tetrahydro pyrrolidine, molecular formula is C
17H
25N
30
2, relative molecular mass is 303.4.Its chemical constitution is as follows:
By retrieval, do not see the pertinent literature and the patent report of relevant compound metformin hydrochloride and vildagliptin.
Summary of the invention
Having the purpose of this invention is to provide a kind of is the Pharmaceutical composition of active component with metformin hydrochloride and vildagliptin, and it is the active component that forms with metformin hydrochloride and vildagliptin, with the Pharmaceutical composition of mixing acceptable accessories formation.
This Pharmaceutical composition can be made into oral formulations.Its oral formulations includes but are not limited to any one solid dosage forms in tablet, capsule, granule, dispersible tablet, chewable tablet, buccal tablet, effervescent tablet, the effervescent granule.In described Pharmaceutical composition, the unit formulation content of its metformin hydrochloride is 125mg~2000mg.Be preferably 500mg~1000mg.The unit formulation content of vildagliptin is 25mg~200mg.Be preferably 50mg~100mg.Draw by analysis, the compound of described metformin hydrochloride and vildagliptin, special preferred compositions is 500mg+50mg in its per unit dosage, 1000mg+50mg, 500mg+100mg, 500mg+100mg.
Pharmaceutical composition of the present invention can be used for the treatment of diet control and the type 2 diabetes mellitus that still can not suitably control of motion, also can be used for taking separately metformin hydrochloride or the out of contior type 2 diabetes mellitus of vildagliptin.
The present invention also provides following pharmacological evaluation, further specifies the suitability of Pharmaceutical composition of the present invention.Experimental technique: the foundation of diabetes rat model: select adult Wistar rats for use, male, body weight 230~260g freely drinks water in the experimentation.Use pH4.5, (concentration is 10mgmL to 0.1mmolL citrate buffer preparation streptozotocin for Streptozotocin, STZ) solution
-1Behind the rat fasting 16h, streptozotocin solution 55mgkg
-1Inferior property lumbar injection.After 7 days, the socket of the eye vein is got blood and is surveyed blood glucose, with fasting glucose 〉=11.11mmolL
-1The person elects diabetic mice as.Selected diabetes rat is divided into 5 groups at random by body weight and blood glucose value.First group is diabetic model group, presses 10mLkg
-1Irritate stomach and give distilled water; Second group is compound metformin group (metformin and vildagliptin proportioning 500: 50), presses 200mgkg
-1Irritate stomach and give compound metformin solution; The 3rd group is the metformin group, presses 200mgkg
-1Irritate stomach and give metformin solution; The 4th group is the vildagliptin group, presses 200mgkg and irritates stomach to vildagliptin solution; And organize in contrast with the normal rat that body weight, Mus are complementary age, irritate stomach by 10mLkg and give ordinary water.Each is organized rat and weighed 1 time in per 3 days, and adjusts the administration volume according to the weight of animals.1 blood glucose of per 4 week mensuration, saccharifying serum albumin (GSP).
2. statistical disposition: statistical data all adopts the mean earthwork, and poor (x ± s) represent, each is organized between the group of data and relatively uses the t inspection statistics.
3 results:
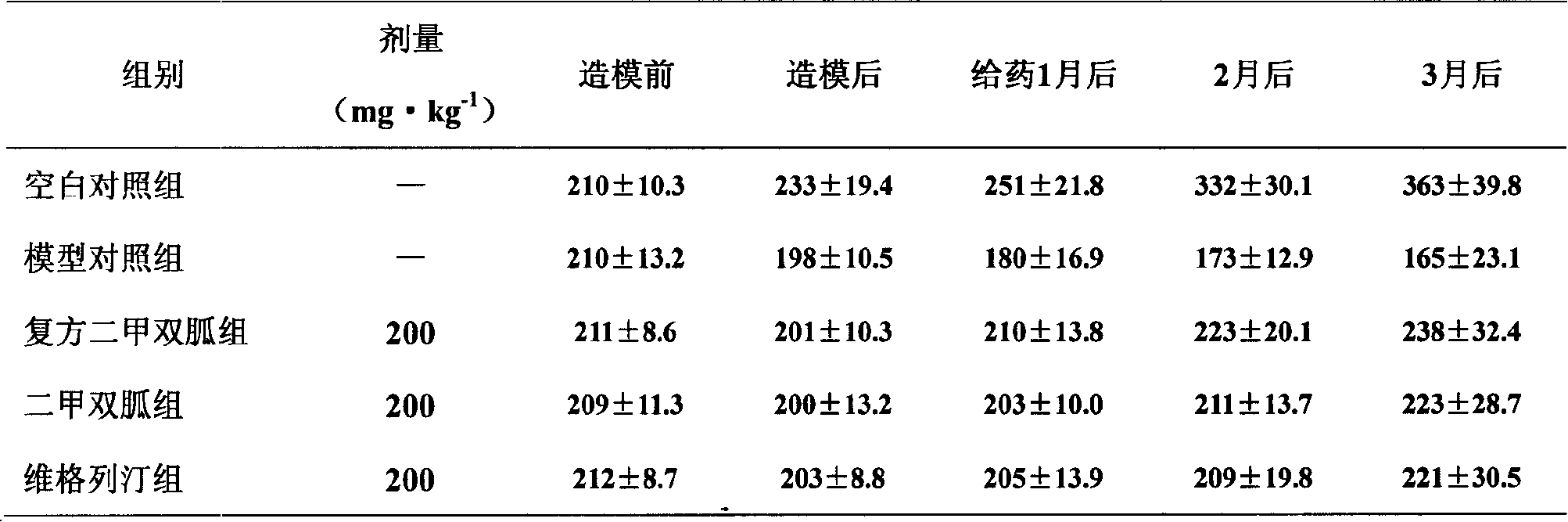
3.1 compound metformin is to the influence (seeing Table 1) of diabetes rat body weight.
Table 1 compound metformin to the influence of diabetes rat body weight (mg) (x ± s, n=15)
By table 1 as seen, after giving the streptozotocin moulding, the diabetes rat body weight significantly is lower than the normal control group, prolongation along with the course of disease, the model group rat body weight presents lasting downward trend (P<0.01), after giving the compound metformin treatment, the diabetes rat weight recovery is very fast, but still significantly is lower than normal control group (P<0.01).
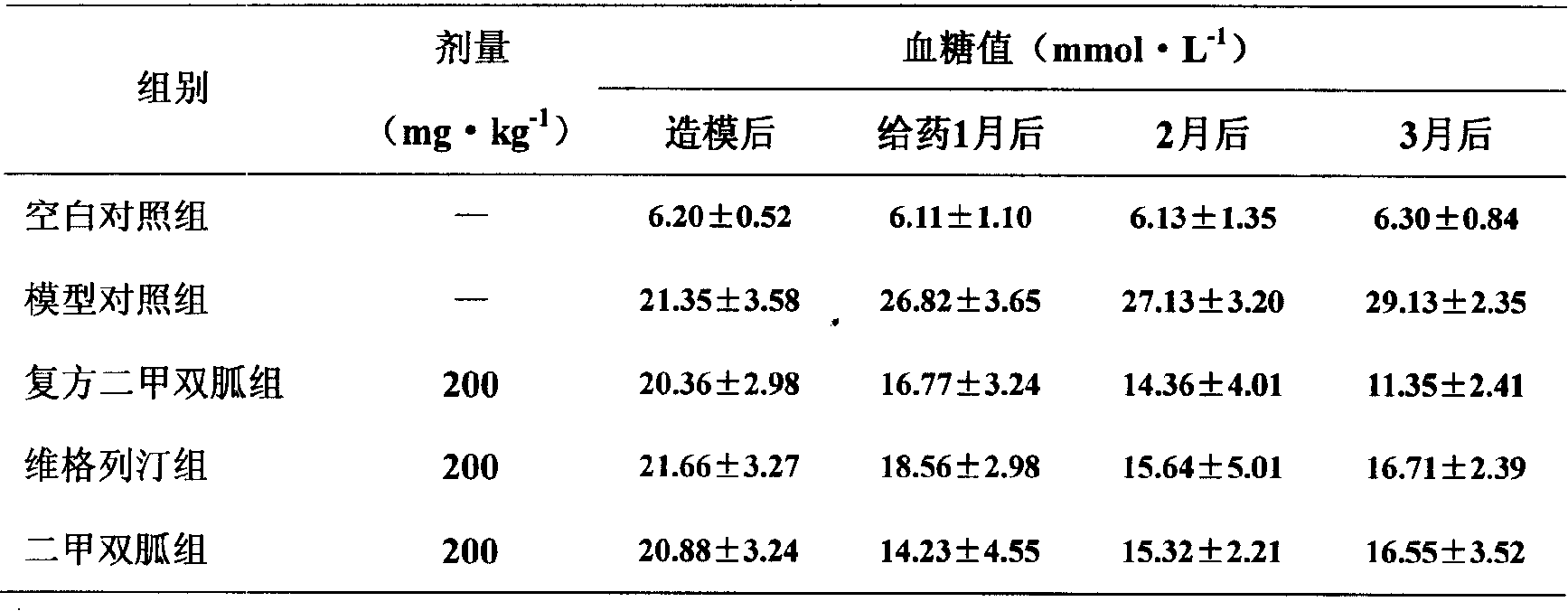
3.2 compound metformin is to the influence (seeing Table 2) of blood glucose in diabetic rats.
Table 2 compound metformin is to the influence of blood glucose in diabetic rats (x ± s.n=15)
By table 3 as seen, the normal matched group fasting blood sugar of diabetes rat significantly raises (P<0.01), and after the compound metformin treatment, the diabetes rat fasting glucose significantly reduces, and blood sugar reducing function is steady, and is lasting.
3.3 compound metformin is to the influence (seeing Table 3) of diabetes rat saccharifying serum albumin (GSP).
Table 3 compound metformin is to the influence of diabetes rat saccharifying serum albumin (GSP) (x ± s)
By table 3 as seen, the normal matched group saccharifying serum albumin of diabetes rat significantly raise (P<0.01), after the compound metformin treatment, diabetes rat saccharifying serum albumin content obviously reduces (P<0.01), the prompting compound metformin can be good at the fluctuation of blood sugar control, and can be good at suppressing the nonenzymatic glycosylation reaction of serum albumin.
The specific embodiment
Come metformin hydrochloride of the present invention and vildagliptin preparation done further specifying by following example, but be not limited in following example.
Embodiment 1 metformin hydrochloride and vildagliptin tablet
Prescription:
Preparation method:
Vildagliptin, microcrystalline Cellulose are crossed 80 mesh sieves respectively, and mix homogeneously is adopting the equivalent method of progressively increasing to mix with metformin hydrochloride, fully stirs to make evenly, and is standby; 50% alcoholic solution is joined in the mixed powder, the system soft material, 24 mesh sieves are granulated, drying, 20 mesh sieve granulate add micropowder silica gel, CMS-Na, adopt suitable punch die compressed tablets behind the mix homogeneously, promptly.
If carry out coating for above-mentioned tablet, then obtain coated tablet, can be Film coated tablets, enteric coatel tablets etc.
Embodiment 2: metformin hydrochloride and vildagliptin capsule
Prescription:
Preparation method:
Vildagliptin, metformin hydrochloride are all crossed 80 mesh sieves, progressively increase behind the method mix homogeneously, add magnesium stearate according to equivalent, mix homogeneously, packing, promptly.
Used capsule shells can be conventional capsule, also can be enteric coated capsule.
Embodiment 3: metformin hydrochloride and vildagliptin chewable tablet
Prescription:
Preparation method:
Supplementary materials such as vildagliptin, metformin, xylitol, mannitol are crossed 80 mesh sieves respectively, adopt equivalent incremental method mix homogeneously, add the 2%PVP-k30 alcoholic solution, the system soft material, 16 mesh sieves are granulated, drying, 20 mesh sieve granulate, the correctives, sweeting agent, the fluidizer mix homogeneously that add other, tabletting, promptly.
Embodiment 4: metformin hydrochloride and vildagliptin dispersible tablet
Prescription
Preparation method:
Supplementary materials such as vildagliptin, metformin hydrochloride, microcrystalline Cellulose are crossed 80 mesh sieves respectively, adopt equivalent incremental method mix homogeneously, standby; Add 75% alcoholic solution, the system soft material, 16 mesh sieves are granulated, drying, 20 mesh sieve granulate add other correctives orange flavor, sweeting agent stevioside, fluidizer micropowder silica gel, mix homogeneously such as disintegrating agent PPVP, L-HPC, tabletting, promptly.
Embodiment 5: metformin hydrochloride and vildagliptin effervescent tablet
Prescription
Preparation method:
Metformin hydrochloride and vildagliptin are crossed 80 mesh sieves, mix homogeneously; Get half mixed powder amount, with the citric acid mix homogeneously, with 50% alcoholic solution system soft material, 18 orders are granulated, drying, 16 order granulate; Get the surplus mixed powder again, mix with sodium carbonate, sodium bicarbonate, the same method is granulated; Two kinds of granules of gained mix, and add other adjuvant, and as mix homogeneously such as PEG6000, orange flavor, acesulfame potassium, sodium chloride, tabletting promptly.
Embodiment 5: metformin hydrochloride and vildagliptin granule
Prescription
Preparation method:
Metformin hydrochloride, vildagliptin, mannitol, sorbitol were waited 80 mesh sieves, mix homogeneously; With the 75% alcoholic solution system soft material of 5%PVPk-30,16 orders are granulated, drying, and 12 order granulate add other adjuvant, and as mix homogeneously such as orange flavor, acesulfame potassiums, packing is promptly.
Claims (7)
- The present invention for a kind of be the Pharmaceutical composition of active component with metformin hydrochloride and vildagliptin, it is characterized in that it is the active component that is formed by metformin hydrochloride and vildagliptin, with the Pharmaceutical composition of mixing acceptable accessories formation.
- 2. Pharmaceutical composition as claimed in claim 1 is characterized in that, can be made into oral formulations.
- 3. Pharmaceutical composition as claimed in claim 2 is characterized in that, its oral formulations is any one solid dosage formss such as tablet, capsule, granule, dispersible tablet, chewable tablet, buccal tablet, effervescent tablet, effervescent granule.
- 4. as the described Pharmaceutical composition of claim 1~3, it is characterized in that the unit formulation content of described metformin hydrochloride is 125mg~2000mg.Be preferably 500mg~1000mg.
- 5. as the described Pharmaceutical composition of claim 1~3, it is characterized in that the unit formulation content of described vildagliptin is 25mg~200mg.Be preferably 50mg~100mg.
- 6. as the described Pharmaceutical composition of claim 1~5, it is characterized in that the compound of described metformin hydrochloride and vildagliptin, special preferred compositions is 500mg+50mg in its unit dose, 1000mg+50mg, 500mg+100mg, 500mg+100mg.
- 7. the described Pharmaceutical composition of claim 1~6 can be used for the treatment of the type 2 diabetes mellitus that diet control and motion still can not suitably control, and also can be used for taking separately metformin hydrochloride or the out of contior type 2 diabetes mellitus of vildagliptin.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CNA2008100558154A CN101234105A (en) | 2008-01-09 | 2008-01-09 | Pharmaceutical composition containing diabetosan and vildagliptin and preparation thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CNA2008100558154A CN101234105A (en) | 2008-01-09 | 2008-01-09 | Pharmaceutical composition containing diabetosan and vildagliptin and preparation thereof |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN101234105A true CN101234105A (en) | 2008-08-06 |
Family
ID=39918172
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CNA2008100558154A Pending CN101234105A (en) | 2008-01-09 | 2008-01-09 | Pharmaceutical composition containing diabetosan and vildagliptin and preparation thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN101234105A (en) |
Cited By (39)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2010092163A3 (en) * | 2009-02-13 | 2010-10-21 | Boehringer Ingelheim International Gmbh | Antidiabetic medications comprising a dpp-4 inhibitor (linagliptin) optionally in combination with other antidiabetics |
| US7820815B2 (en) | 2004-11-05 | 2010-10-26 | Boehringer Ingelheim International Gmbh | Process for the preparation of chiral 8-(-3-aminopiperidin-1-yl) xanthines |
| US8106060B2 (en) | 2005-07-30 | 2012-01-31 | Boehringer Ingelheim International Gmbh | 8-(3-amino-piperidin-1-yl)-xanthines, their preparation, and their use as pharmaceuticals |
| US8119648B2 (en) | 2002-08-21 | 2012-02-21 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | 8-[3-amino-piperidin-1-yl]-xanthines, the preparation thereof and their use as pharmaceutical compositions |
| US8232281B2 (en) | 2006-05-04 | 2012-07-31 | Boehringer Ingelheim International Gmbh | Uses of DPP-IV inhibitors |
| EP2550957A1 (en) * | 2010-12-21 | 2013-01-30 | Sanovel Ilaç Sanayi Ve Ticaret Anonim Sirketi | Effervescent formulations of vildagliptin |
| US8513264B2 (en) | 2008-09-10 | 2013-08-20 | Boehringer Ingelheim International Gmbh | Combination therapy for the treatment of diabetes and related conditions |
| CN103285398A (en) * | 2013-06-28 | 2013-09-11 | 青岛黄海制药有限责任公司 | Compound preparation containing DPP-IV (dipeptidyl peptidase-IV) inhibitor and type-II diabetes medicine and preparation method thereof |
| US8551957B2 (en) | 2007-08-16 | 2013-10-08 | Boehringer Ingelheim International Gmbh | Pharmaceutical composition comprising a glucopyranosyl-substituted benzene derivate |
| CN103432089A (en) * | 2013-09-02 | 2013-12-11 | 天津市聚星康华医药科技有限公司 | Metformin hydrochloride chewable tablet and preparation method thereof |
| US8697868B2 (en) | 2004-02-18 | 2014-04-15 | Boehringer Ingelheim International Gmbh | 8-[3-amino-piperidin-1-yl]-xanthines, their preparation and their use as pharmaceutical compositions |
| CN103845326A (en) * | 2014-03-24 | 2014-06-11 | 江苏奥赛康药业股份有限公司 | Compound composition of vildagliptin and melbine and preparation method thereof |
| US8846695B2 (en) | 2009-01-07 | 2014-09-30 | Boehringer Ingelheim International Gmbh | Treatment for diabetes in patients with inadequate glycemic control despite metformin therapy comprising a DPP-IV inhibitor |
| US8853156B2 (en) | 2008-08-06 | 2014-10-07 | Boehringer Ingelheim International Gmbh | Treatment for diabetes in patients inappropriate for metformin therapy |
| US8865729B2 (en) | 2008-12-23 | 2014-10-21 | Boehringer Ingelheim International Gmbh | Salt forms of a xanthine compound |
| US8883800B2 (en) | 2011-07-15 | 2014-11-11 | Boehringer Ingelheim International Gmbh | Substituted quinazolines, the preparation thereof and the use thereof in pharmaceutical compositions |
| WO2015040460A1 (en) * | 2013-09-21 | 2015-03-26 | Effrx Pharmaceuticals Sa | Low-sodium effervescent pharmaceutical formulations |
| CN104557944A (en) * | 2014-12-31 | 2015-04-29 | 北京瑞都医药科技有限公司 | Hypoglycemic agent and preparation method thereof |
| US9034883B2 (en) | 2010-11-15 | 2015-05-19 | Boehringer Ingelheim International Gmbh | Vasoprotective and cardioprotective antidiabetic therapy |
| CN104825446A (en) * | 2015-06-04 | 2015-08-12 | 海口南陆医药科技有限公司 | Medicinal composition containing vildagliptin and metformin hydrochloride |
| EP2915527A1 (en) * | 2014-03-06 | 2015-09-09 | Sanovel Ilac Sanayi ve Ticaret A.S. | Pharmaceutical formulations of vildagliptin |
| US9149478B2 (en) | 2010-06-24 | 2015-10-06 | Boehringer Ingelheim International Gmbh | Diabetes therapy |
| US9155705B2 (en) | 2008-04-03 | 2015-10-13 | Boehringer Ingelheim International Gmbh | DPP-IV inhibitor combined with a further antidiabetic agent, tablets comprising such formulations, their use and process for their preparation |
| US9186392B2 (en) | 2010-05-05 | 2015-11-17 | Boehringer Ingelheim International Gmbh | Combination therapy |
| US9266888B2 (en) | 2006-05-04 | 2016-02-23 | Boehringer Ingelheim International Gmbh | Polymorphs |
| US9457029B2 (en) | 2009-11-27 | 2016-10-04 | Boehringer Ingelheim International Gmbh | Treatment of genotyped diabetic patients with DPP-IV inhibitors such as linagliptin |
| US9486526B2 (en) | 2008-08-06 | 2016-11-08 | Boehringer Ingelheim International Gmbh | Treatment for diabetes in patients inappropriate for metformin therapy |
| US9526728B2 (en) | 2014-02-28 | 2016-12-27 | Boehringer Ingelheim International Gmbh | Medical use of a DPP-4 inhibitor |
| US9526730B2 (en) | 2012-05-14 | 2016-12-27 | Boehringer Ingelheim International Gmbh | Use of a DPP-4 inhibitor in podocytes related disorders and/or nephrotic syndrome |
| US9555001B2 (en) | 2012-03-07 | 2017-01-31 | Boehringer Ingelheim International Gmbh | Pharmaceutical composition and uses thereof |
| US9713618B2 (en) | 2012-05-24 | 2017-07-25 | Boehringer Ingelheim International Gmbh | Method for modifying food intake and regulating food preference with a DPP-4 inhibitor |
| US20180185291A1 (en) | 2011-03-07 | 2018-07-05 | Boehringer Ingelheim International Gmbh | Pharmaceutical compositions |
| US10155000B2 (en) | 2016-06-10 | 2018-12-18 | Boehringer Ingelheim International Gmbh | Medical use of pharmaceutical combination or composition |
| US10406172B2 (en) | 2009-02-13 | 2019-09-10 | Boehringer Ingelheim International Gmbh | Pharmaceutical composition, methods for treating and uses thereof |
| CN112641776A (en) * | 2019-10-12 | 2021-04-13 | 江苏晶立信医药科技有限公司 | A pharmaceutical composition containing metformin or its pharmaceutically acceptable salt and Alogliptin or its pharmaceutically acceptable salt as active ingredients |
| US11033552B2 (en) | 2006-05-04 | 2021-06-15 | Boehringer Ingelheim International Gmbh | DPP IV inhibitor formulations |
| US11911388B2 (en) | 2008-10-16 | 2024-02-27 | Boehringer Ingelheim International Gmbh | Treatment for diabetes in patients with insufficient glycemic control despite therapy with an oral or non-oral antidiabetic drug |
| EP4171532A4 (en) * | 2020-06-25 | 2024-07-24 | Sanovel Ilac Sanayi Ve Ticaret Anonim Sirketi | STABLE COMBINATION OF VILDAGLIPTIN AND METFORMIN HCI |
| US12312352B2 (en) | 2012-05-14 | 2025-05-27 | Boehringer Ingelheim International Gmbh | Use of a DPP-4 inhibitor in SIRS and/or sepsis |
-
2008
- 2008-01-09 CN CNA2008100558154A patent/CN101234105A/en active Pending
Cited By (83)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8664232B2 (en) | 2002-08-21 | 2014-03-04 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | 8-[3-amino-piperidin-1-yl]-xanthines, the preparation thereof and their use as pharmaceutical compositions |
| US9321791B2 (en) | 2002-08-21 | 2016-04-26 | Boehringer Ingelheim International Gmbh | 8-[3-amino-piperidin-1-yl]-xanthines, the preparation thereof and their use as pharmaceutical compositions |
| US9108964B2 (en) | 2002-08-21 | 2015-08-18 | Boehringer Ingelheim International Gmbh | 8-[3-amino-piperidin-1-yl]-xanthines, the preparation thereof and their use as pharmaceutical compositions |
| US8119648B2 (en) | 2002-08-21 | 2012-02-21 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | 8-[3-amino-piperidin-1-yl]-xanthines, the preparation thereof and their use as pharmaceutical compositions |
| US8178541B2 (en) | 2002-08-21 | 2012-05-15 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | 8-[3-amino-piperidin-1-yl]-xanthines, the preparation thereof and their use as pharmaceutical compositions |
| US9556175B2 (en) | 2002-08-21 | 2017-01-31 | Boehringer Ingelheim International Gmbh | 8-[3-amino-piperidin-1-yl]-xanthines, the preparation thereof and thier use as pharmaceutical compositions |
| US10023574B2 (en) | 2002-08-21 | 2018-07-17 | Boehringer Ingelheim International Gmbh | 8-[3-amino-piperidin-1-yl]-xanthines, the preparation thereof and their use as pharmaceutical compositions |
| US10202383B2 (en) | 2002-08-21 | 2019-02-12 | Boehringer Ingelheim International Gmbh | 8-[3-amino-piperidin-1-yl]-xanthines, the preparation thereof and their use as pharmaceutical compositions |
| US8697868B2 (en) | 2004-02-18 | 2014-04-15 | Boehringer Ingelheim International Gmbh | 8-[3-amino-piperidin-1-yl]-xanthines, their preparation and their use as pharmaceutical compositions |
| US8883805B2 (en) | 2004-11-05 | 2014-11-11 | Boehringer Ingelheim International Gmbh | Process for the preparation of chiral 8-(3-aminopiperidin-1-yl)-xanthines |
| US8541450B2 (en) | 2004-11-05 | 2013-09-24 | Boehringer Ingelheim International Gmbh | Process for the preparation of chiral 8-(3-aminopiperidin-1yl)-xanthines |
| US7820815B2 (en) | 2004-11-05 | 2010-10-26 | Boehringer Ingelheim International Gmbh | Process for the preparation of chiral 8-(-3-aminopiperidin-1-yl) xanthines |
| US9499546B2 (en) | 2004-11-05 | 2016-11-22 | Boehringer Ingelheim International Gmbh | Process for the preparation of chiral 8-(3-aminopiperidin-1-yl)-xanthines |
| US9751855B2 (en) | 2004-11-05 | 2017-09-05 | Boehringer Ingelheim International Gmbh | Process for the preparation of chiral 8-(3-aminopiperidin-1-yl)-xanthines |
| US8106060B2 (en) | 2005-07-30 | 2012-01-31 | Boehringer Ingelheim International Gmbh | 8-(3-amino-piperidin-1-yl)-xanthines, their preparation, and their use as pharmaceuticals |
| US8637530B2 (en) | 2005-07-30 | 2014-01-28 | Boehringer Ingelheim International Gmbh | 8-(3-amino-piperidin-1-yl)-xanthines, their preparation, and their use as pharmaceuticals |
| US8673927B2 (en) | 2006-05-04 | 2014-03-18 | Boehringer Ingelheim International Gmbh | Uses of DPP-IV inhibitors |
| US9815837B2 (en) | 2006-05-04 | 2017-11-14 | Boehringer Ingelheim International Gmbh | Polymorphs |
| US9173859B2 (en) | 2006-05-04 | 2015-11-03 | Boehringer Ingelheim International Gmbh | Uses of DPP IV inhibitors |
| US10301313B2 (en) | 2006-05-04 | 2019-05-28 | Boehringer Ingelheim International Gmbh | Polymorphs |
| US12171767B2 (en) | 2006-05-04 | 2024-12-24 | Boehringer Ingelheim International Gmbh | Uses of DPP IV inhibitors |
| US9266888B2 (en) | 2006-05-04 | 2016-02-23 | Boehringer Ingelheim International Gmbh | Polymorphs |
| US10080754B2 (en) | 2006-05-04 | 2018-09-25 | Boehringer Ingelheim International Gmbh | Uses of DPP IV inhibitors |
| US9493462B2 (en) | 2006-05-04 | 2016-11-15 | Boehringer Ingelheim International Gmbh | Polymorphs |
| US12178819B2 (en) | 2006-05-04 | 2024-12-31 | Boehringer Ingelheim International Gmbh | DPP IV inhibitor formulations |
| US11033552B2 (en) | 2006-05-04 | 2021-06-15 | Boehringer Ingelheim International Gmbh | DPP IV inhibitor formulations |
| US11291668B2 (en) | 2006-05-04 | 2022-04-05 | Boehringer Ingelheim International Gmbh | Uses of DPP IV inhibitors |
| US8232281B2 (en) | 2006-05-04 | 2012-07-31 | Boehringer Ingelheim International Gmbh | Uses of DPP-IV inhibitors |
| US11919903B2 (en) | 2006-05-04 | 2024-03-05 | Boehringer Ingelheim International Gmbh | Polymorphs |
| US11084819B2 (en) | 2006-05-04 | 2021-08-10 | Boehringer Ingelheim International Gmbh | Polymorphs |
| US8551957B2 (en) | 2007-08-16 | 2013-10-08 | Boehringer Ingelheim International Gmbh | Pharmaceutical composition comprising a glucopyranosyl-substituted benzene derivate |
| US10022379B2 (en) | 2008-04-03 | 2018-07-17 | Boehringer Ingelheim International Gmbh | DPP-IV inhibitor combined with a further antidiabetic agent, tablets comprising such formulations, their use and process for their preparation |
| US9415016B2 (en) | 2008-04-03 | 2016-08-16 | Boehringer Ingelheim International Gmbh | DPP-IV inhibitor combined with a further antidiabetic agent, tablets comprising such formulations, their use and process for their preparation |
| US9155705B2 (en) | 2008-04-03 | 2015-10-13 | Boehringer Ingelheim International Gmbh | DPP-IV inhibitor combined with a further antidiabetic agent, tablets comprising such formulations, their use and process for their preparation |
| US10973827B2 (en) | 2008-04-03 | 2021-04-13 | Boehringer Ingelheim International Gmbh | DPP-IV inhibitor combined with a further antidiabetic agent, tablets comprising such formulations, their use and process for their preparation |
| US9486526B2 (en) | 2008-08-06 | 2016-11-08 | Boehringer Ingelheim International Gmbh | Treatment for diabetes in patients inappropriate for metformin therapy |
| US8853156B2 (en) | 2008-08-06 | 2014-10-07 | Boehringer Ingelheim International Gmbh | Treatment for diabetes in patients inappropriate for metformin therapy |
| US10034877B2 (en) | 2008-08-06 | 2018-07-31 | Boehringer Ingelheim International Gmbh | Treatment for diabetes in patients inappropriate for metformin therapy |
| US8513264B2 (en) | 2008-09-10 | 2013-08-20 | Boehringer Ingelheim International Gmbh | Combination therapy for the treatment of diabetes and related conditions |
| US11911388B2 (en) | 2008-10-16 | 2024-02-27 | Boehringer Ingelheim International Gmbh | Treatment for diabetes in patients with insufficient glycemic control despite therapy with an oral or non-oral antidiabetic drug |
| US8865729B2 (en) | 2008-12-23 | 2014-10-21 | Boehringer Ingelheim International Gmbh | Salt forms of a xanthine compound |
| US9212183B2 (en) | 2008-12-23 | 2015-12-15 | Boehringer Ingelheim International Gmbh | Salt forms of 1-[(4-methyl-quinazolin-2-yl)methyl]-3-methyl-7-(2-butyn-1-yl)-8-(3-(R)-amino-piperidin-1-yl)-xanthine |
| US8846695B2 (en) | 2009-01-07 | 2014-09-30 | Boehringer Ingelheim International Gmbh | Treatment for diabetes in patients with inadequate glycemic control despite metformin therapy comprising a DPP-IV inhibitor |
| EA029759B1 (en) * | 2009-02-13 | 2018-05-31 | Бёрингер Ингельхайм Интернациональ Гмбх | Antidiabetic medications comprising dpp-4 inhibitor (linagliptin) optionally in combination with other antidiabetic agents |
| JP2012517977A (en) * | 2009-02-13 | 2012-08-09 | ベーリンガー インゲルハイム インターナショナル ゲゼルシャフト ミット ベシュレンクテル ハフツング | An anti-diabetic drug comprising a DPP-4 inhibitor (linagliptin) optionally in combination with other anti-diabetic drugs |
| WO2010092163A3 (en) * | 2009-02-13 | 2010-10-21 | Boehringer Ingelheim International Gmbh | Antidiabetic medications comprising a dpp-4 inhibitor (linagliptin) optionally in combination with other antidiabetics |
| US10406172B2 (en) | 2009-02-13 | 2019-09-10 | Boehringer Ingelheim International Gmbh | Pharmaceutical composition, methods for treating and uses thereof |
| US12115179B2 (en) | 2009-02-13 | 2024-10-15 | Boehringer Ingelheim International Gmbh | Pharmaceutical composition, methods for treating and uses thereof |
| US10092571B2 (en) | 2009-11-27 | 2018-10-09 | Boehringer Ingelheim International Gmbh | Treatment of genotyped diabetic patients with DPP-IV inhibitors such as linagliptin |
| US9457029B2 (en) | 2009-11-27 | 2016-10-04 | Boehringer Ingelheim International Gmbh | Treatment of genotyped diabetic patients with DPP-IV inhibitors such as linagliptin |
| US9186392B2 (en) | 2010-05-05 | 2015-11-17 | Boehringer Ingelheim International Gmbh | Combination therapy |
| US9603851B2 (en) | 2010-05-05 | 2017-03-28 | Boehringer Ingelheim International Gmbh | Combination therapy |
| US10004747B2 (en) | 2010-05-05 | 2018-06-26 | Boehringer Ingelheim International Gmbh | Combination therapy |
| US9149478B2 (en) | 2010-06-24 | 2015-10-06 | Boehringer Ingelheim International Gmbh | Diabetes therapy |
| US9034883B2 (en) | 2010-11-15 | 2015-05-19 | Boehringer Ingelheim International Gmbh | Vasoprotective and cardioprotective antidiabetic therapy |
| US11911387B2 (en) | 2010-11-15 | 2024-02-27 | Boehringer Ingelheim International Gmbh | Vasoprotective and cardioprotective antidiabetic therapy |
| EP2550957A1 (en) * | 2010-12-21 | 2013-01-30 | Sanovel Ilaç Sanayi Ve Ticaret Anonim Sirketi | Effervescent formulations of vildagliptin |
| US10596120B2 (en) | 2011-03-07 | 2020-03-24 | Boehringer Ingelheim International Gmbh | Pharmaceutical compositions |
| US20180185291A1 (en) | 2011-03-07 | 2018-07-05 | Boehringer Ingelheim International Gmbh | Pharmaceutical compositions |
| US11564886B2 (en) | 2011-03-07 | 2023-01-31 | Boehringer Ingelheim International Gmbh | Pharmaceutical compositions |
| US8962636B2 (en) | 2011-07-15 | 2015-02-24 | Boehringer Ingelheim International Gmbh | Substituted quinazolines, the preparation thereof and the use thereof in pharmaceutical compositions |
| US8883800B2 (en) | 2011-07-15 | 2014-11-11 | Boehringer Ingelheim International Gmbh | Substituted quinazolines, the preparation thereof and the use thereof in pharmaceutical compositions |
| US9199998B2 (en) | 2011-07-15 | 2015-12-01 | Boehringer Ingelheim Internatioal Gmbh | Substituted quinazolines, the preparation thereof and the use thereof in pharmaceutical compositions |
| US9555001B2 (en) | 2012-03-07 | 2017-01-31 | Boehringer Ingelheim International Gmbh | Pharmaceutical composition and uses thereof |
| US10195203B2 (en) | 2012-05-14 | 2019-02-05 | Boehringr Ingelheim International GmbH | Use of a DPP-4 inhibitor in podocytes related disorders and/or nephrotic syndrome |
| US12312352B2 (en) | 2012-05-14 | 2025-05-27 | Boehringer Ingelheim International Gmbh | Use of a DPP-4 inhibitor in SIRS and/or sepsis |
| US9526730B2 (en) | 2012-05-14 | 2016-12-27 | Boehringer Ingelheim International Gmbh | Use of a DPP-4 inhibitor in podocytes related disorders and/or nephrotic syndrome |
| US9713618B2 (en) | 2012-05-24 | 2017-07-25 | Boehringer Ingelheim International Gmbh | Method for modifying food intake and regulating food preference with a DPP-4 inhibitor |
| CN103285398A (en) * | 2013-06-28 | 2013-09-11 | 青岛黄海制药有限责任公司 | Compound preparation containing DPP-IV (dipeptidyl peptidase-IV) inhibitor and type-II diabetes medicine and preparation method thereof |
| CN103285398B (en) * | 2013-06-28 | 2015-07-22 | 青岛黄海制药有限责任公司 | Compound preparation containing DPP-IV (dipeptidyl peptidase-IV) inhibitor and type-II diabetes medicine and preparation method thereof |
| CN103432089A (en) * | 2013-09-02 | 2013-12-11 | 天津市聚星康华医药科技有限公司 | Metformin hydrochloride chewable tablet and preparation method thereof |
| WO2015040460A1 (en) * | 2013-09-21 | 2015-03-26 | Effrx Pharmaceuticals Sa | Low-sodium effervescent pharmaceutical formulations |
| US9526728B2 (en) | 2014-02-28 | 2016-12-27 | Boehringer Ingelheim International Gmbh | Medical use of a DPP-4 inhibitor |
| EP2915527A1 (en) * | 2014-03-06 | 2015-09-09 | Sanovel Ilac Sanayi ve Ticaret A.S. | Pharmaceutical formulations of vildagliptin |
| CN103845326A (en) * | 2014-03-24 | 2014-06-11 | 江苏奥赛康药业股份有限公司 | Compound composition of vildagliptin and melbine and preparation method thereof |
| CN103845326B (en) * | 2014-03-24 | 2017-07-07 | 江苏奥赛康药业股份有限公司 | Compound of vildagliptin and melbine and preparation method thereof |
| CN104557944B (en) * | 2014-12-31 | 2017-05-31 | 北京瑞都医药科技有限公司 | A kind of hypoglycemic medicine and preparation method thereof |
| CN104557944A (en) * | 2014-12-31 | 2015-04-29 | 北京瑞都医药科技有限公司 | Hypoglycemic agent and preparation method thereof |
| CN104825446A (en) * | 2015-06-04 | 2015-08-12 | 海口南陆医药科技有限公司 | Medicinal composition containing vildagliptin and metformin hydrochloride |
| US10155000B2 (en) | 2016-06-10 | 2018-12-18 | Boehringer Ingelheim International Gmbh | Medical use of pharmaceutical combination or composition |
| US12364700B2 (en) | 2016-06-10 | 2025-07-22 | Boehringer Ingelheim International Gmbh | Medical use of pharmaceutical combination or composition |
| CN112641776A (en) * | 2019-10-12 | 2021-04-13 | 江苏晶立信医药科技有限公司 | A pharmaceutical composition containing metformin or its pharmaceutically acceptable salt and Alogliptin or its pharmaceutically acceptable salt as active ingredients |
| EP4171532A4 (en) * | 2020-06-25 | 2024-07-24 | Sanovel Ilac Sanayi Ve Ticaret Anonim Sirketi | STABLE COMBINATION OF VILDAGLIPTIN AND METFORMIN HCI |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN101234105A (en) | Pharmaceutical composition containing diabetosan and vildagliptin and preparation thereof | |
| CN101181264A (en) | Pharmaceutical composition taking metformin hydrochloride and vigelegting as active component as well as preparing method and uses thereof | |
| CN106924208A (en) | A kind of compound Dapagliflozin Metformin Extended-release Tablets and preparation method thereof | |
| CN101843617B (en) | Slow release preparation of compound Repaglinide-metformin hydrochloride | |
| CN1561980A (en) | Melbine hydrochloride enteric coatel slow-releasing preparation and its preparing method | |
| CN101647785B (en) | Gliclazide sustained-release tablet and preparation method thereof | |
| CN105233300B (en) | A kind of stable vildagliptin composition and preparation method thereof | |
| CN102727894B (en) | A kind of pharmaceutical composition and application thereof for the treatment of diabetes and complication thereof | |
| CN101496790A (en) | Acarbose chewable tablet | |
| CN105343056A (en) | Oral pharmaceutical composition for treating or preventing obesity-related hypertension and its application | |
| CN101524355A (en) | Compound preparation of antituberculosis medicaments, and preparation method thereof | |
| Kadhe et al. | Advances in drug delivery of oral hypoglycemic agents | |
| CN110384709A (en) | Composition and its application containing phloridzin and 1-DNJ | |
| CN101756993B (en) | Medical composition for losing weight or treating metabolic syndromes | |
| WO2005105109A1 (en) | Oral modified-release lozenges and their preparation method | |
| CN100496496C (en) | Compound metformin glipidide preparation for treating diabetes type II, and its preparation method | |
| CN112641776A (en) | A pharmaceutical composition containing metformin or its pharmaceutically acceptable salt and Alogliptin or its pharmaceutically acceptable salt as active ingredients | |
| CN100581549C (en) | Sustained-release preparation containing hydrochlorothiazide and preparation method thereof | |
| US11096890B2 (en) | Chewable dosage forms containing sitagliptin and metformin | |
| Chandramohan et al. | REVIEW ON DEVELOPMENT OF SUSTAINED RELEASE CAPSULES INCORPORATING MATRIX GRANULES OF GLIMEPIRIDE | |
| CN103405471A (en) | Compound preparation containing ilaprazole sodium | |
| CN101559052A (en) | Oral pharmaceutical composition containing mitiglinide and voglibose | |
| CN117731624A (en) | Solid pharmaceutical composition comprising GLP-1 receptor agonist and application | |
| JP2006519881A5 (en) | ||
| CN104434952B (en) | It is a kind of to treat pharmaceutical composition of diabetes and preparation method thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C02 | Deemed withdrawal of patent application after publication (patent law 2001) | ||
| WD01 | Invention patent application deemed withdrawn after publication |
Open date: 20080806 |