CN100594938C - Pulullan polysaccharide carrier material having the function of targeting therapy of cancer - Google Patents
Pulullan polysaccharide carrier material having the function of targeting therapy of cancer Download PDFInfo
- Publication number
- CN100594938C CN100594938C CN 200710057172 CN200710057172A CN100594938C CN 100594938 C CN100594938 C CN 100594938C CN 200710057172 CN200710057172 CN 200710057172 CN 200710057172 A CN200710057172 A CN 200710057172A CN 100594938 C CN100594938 C CN 100594938C
- Authority
- CN
- China
- Prior art keywords
- pulullan polysaccharide
- biotin
- carrier material
- polysaccharide
- pulullan
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
- 150000004676 glycans Chemical class 0.000 title claims abstract description 47
- 229920001282 polysaccharide Polymers 0.000 title claims abstract description 47
- 239000005017 polysaccharide Substances 0.000 title claims abstract description 47
- 239000012876 carrier material Substances 0.000 title claims abstract description 15
- 206010028980 Neoplasm Diseases 0.000 title claims abstract description 12
- 201000011510 cancer Diseases 0.000 title claims description 9
- 230000008685 targeting Effects 0.000 title abstract description 4
- 238000002560 therapeutic procedure Methods 0.000 title 1
- YBJHBAHKTGYVGT-ZKWXMUAHSA-N (+)-Biotin Chemical compound N1C(=O)N[C@@H]2[C@H](CCCCC(=O)O)SC[C@@H]21 YBJHBAHKTGYVGT-ZKWXMUAHSA-N 0.000 claims abstract description 54
- 239000011616 biotin Substances 0.000 claims abstract description 27
- 229960002685 biotin Drugs 0.000 claims abstract description 27
- 235000020958 biotin Nutrition 0.000 claims abstract description 27
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims abstract description 19
- VHYFNPMBLIVWCW-UHFFFAOYSA-N 4-Dimethylaminopyridine Chemical compound CN(C)C1=CC=NC=C1 VHYFNPMBLIVWCW-UHFFFAOYSA-N 0.000 claims abstract description 18
- 230000000694 effects Effects 0.000 claims abstract description 10
- 238000006467 substitution reaction Methods 0.000 claims abstract description 9
- QOSSAOTZNIDXMA-UHFFFAOYSA-N Dicylcohexylcarbodiimide Chemical compound C1CCCCC1N=C=NC1CCCCC1 QOSSAOTZNIDXMA-UHFFFAOYSA-N 0.000 claims abstract description 7
- 239000003960 organic solvent Substances 0.000 claims abstract description 7
- 238000002360 preparation method Methods 0.000 claims abstract description 7
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 claims description 12
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 claims description 10
- 239000000376 reactant Substances 0.000 claims description 10
- 238000006243 chemical reaction Methods 0.000 claims description 8
- 238000000502 dialysis Methods 0.000 claims description 8
- 238000001914 filtration Methods 0.000 claims description 6
- 239000007787 solid Substances 0.000 claims description 6
- 238000013019 agitation Methods 0.000 claims description 4
- 238000000151 deposition Methods 0.000 claims description 4
- 230000008021 deposition Effects 0.000 claims description 4
- 239000003937 drug carrier Substances 0.000 claims description 4
- 238000001035 drying Methods 0.000 claims description 4
- 239000000706 filtrate Substances 0.000 claims description 4
- 239000007788 liquid Substances 0.000 claims description 4
- 239000000385 dialysis solution Substances 0.000 claims description 3
- 239000013049 sediment Substances 0.000 claims description 3
- 239000000843 powder Substances 0.000 claims description 2
- 239000011859 microparticle Substances 0.000 abstract 1
- 230000009870 specific binding Effects 0.000 abstract 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 8
- 241001597008 Nomeidae Species 0.000 description 7
- 230000006287 biotinylation Effects 0.000 description 7
- 238000007413 biotinylation Methods 0.000 description 7
- 239000003814 drug Substances 0.000 description 7
- 210000004027 cell Anatomy 0.000 description 6
- 239000008367 deionised water Substances 0.000 description 5
- 229910021641 deionized water Inorganic materials 0.000 description 5
- 210000004881 tumor cell Anatomy 0.000 description 5
- 239000000463 material Substances 0.000 description 4
- IAZDPXIOMUYVGZ-WFGJKAKNSA-N Dimethyl sulfoxide Chemical group [2H]C([2H])([2H])S(=O)C([2H])([2H])[2H] IAZDPXIOMUYVGZ-WFGJKAKNSA-N 0.000 description 3
- 229920001218 Pullulan Polymers 0.000 description 3
- 239000004373 Pullulan Substances 0.000 description 3
- 239000004531 microgranule Substances 0.000 description 3
- 235000019423 pullulan Nutrition 0.000 description 3
- 239000000243 solution Substances 0.000 description 3
- DBTMGCOVALSLOR-UHFFFAOYSA-N 32-alpha-galactosyl-3-alpha-galactosyl-galactose Natural products OC1C(O)C(O)C(CO)OC1OC1C(O)C(OC2C(C(CO)OC(O)C2O)O)OC(CO)C1O DBTMGCOVALSLOR-UHFFFAOYSA-N 0.000 description 2
- RXVWSYJTUUKTEA-UHFFFAOYSA-N D-maltotriose Natural products OC1C(O)C(OC(C(O)CO)C(O)C(O)C=O)OC(CO)C1OC1C(O)C(O)C(O)C(CO)O1 RXVWSYJTUUKTEA-UHFFFAOYSA-N 0.000 description 2
- 241000219095 Vitis Species 0.000 description 2
- 235000009754 Vitis X bourquina Nutrition 0.000 description 2
- 235000012333 Vitis X labruscana Nutrition 0.000 description 2
- 235000014787 Vitis vinifera Nutrition 0.000 description 2
- 239000007864 aqueous solution Substances 0.000 description 2
- 108010053098 biotin receptor Proteins 0.000 description 2
- 150000001720 carbohydrates Chemical group 0.000 description 2
- 239000003153 chemical reaction reagent Substances 0.000 description 2
- 239000000470 constituent Substances 0.000 description 2
- 239000003431 cross linking reagent Substances 0.000 description 2
- 229960001760 dimethyl sulfoxide Drugs 0.000 description 2
- 238000009616 inductively coupled plasma Methods 0.000 description 2
- FYGDTMLNYKFZSV-UHFFFAOYSA-N mannotriose Natural products OC1C(O)C(O)C(CO)OC1OC1C(CO)OC(OC2C(OC(O)C(O)C2O)CO)C(O)C1O FYGDTMLNYKFZSV-UHFFFAOYSA-N 0.000 description 2
- 239000000047 product Substances 0.000 description 2
- 239000002904 solvent Substances 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- 238000005303 weighing Methods 0.000 description 2
- FYGDTMLNYKFZSV-BYLHFPJWSA-N β-1,4-galactotrioside Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@H](CO)O[C@@H](O[C@@H]2[C@@H](O[C@@H](O)[C@H](O)[C@H]2O)CO)[C@H](O)[C@H]1O FYGDTMLNYKFZSV-BYLHFPJWSA-N 0.000 description 2
- OWEGMIWEEQEYGQ-UHFFFAOYSA-N 100676-05-9 Natural products OC1C(O)C(O)C(CO)OC1OCC1C(O)C(O)C(O)C(OC2C(OC(O)C(O)C2O)CO)O1 OWEGMIWEEQEYGQ-UHFFFAOYSA-N 0.000 description 1
- LLIANSAISVOLHR-GBCQHVBFSA-N 5-[(3as,4s,6ar)-2-oxidanylidene-1,3,3a,4,6,6a-hexahydrothieno[3,4-d]imidazol-4-yl]pentanoic acid Chemical compound N1C(=O)N[C@@H]2[C@H](CCCCC(=O)O)SC[C@@H]21.N1C(=O)N[C@@H]2[C@H](CCCCC(=O)O)SC[C@@H]21 LLIANSAISVOLHR-GBCQHVBFSA-N 0.000 description 1
- 239000004382 Amylase Substances 0.000 description 1
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 1
- SXRSQZLOMIGNAQ-UHFFFAOYSA-N Glutaraldehyde Chemical compound O=CCCCC=O SXRSQZLOMIGNAQ-UHFFFAOYSA-N 0.000 description 1
- GUBGYTABKSRVRQ-PICCSMPSSA-N Maltose Natural products O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@@H](CO)OC(O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-PICCSMPSSA-N 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 238000003556 assay Methods 0.000 description 1
- 210000000170 cell membrane Anatomy 0.000 description 1
- 230000002939 deleterious effect Effects 0.000 description 1
- 229940079593 drug Drugs 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 230000002349 favourable effect Effects 0.000 description 1
- 239000012467 final product Substances 0.000 description 1
- 230000014509 gene expression Effects 0.000 description 1
- 239000008103 glucose Substances 0.000 description 1
- 210000003494 hepatocyte Anatomy 0.000 description 1
- 230000005847 immunogenicity Effects 0.000 description 1
- 210000004185 liver Anatomy 0.000 description 1
- 229920002521 macromolecule Polymers 0.000 description 1
- 238000000034 method Methods 0.000 description 1
- 230000003505 mutagenic effect Effects 0.000 description 1
- 231100000252 nontoxic Toxicity 0.000 description 1
- 230000003000 nontoxic effect Effects 0.000 description 1
- 230000009965 odorless effect Effects 0.000 description 1
- 230000002018 overexpression Effects 0.000 description 1
- 238000006116 polymerization reaction Methods 0.000 description 1
- 108090000623 proteins and genes Proteins 0.000 description 1
- 102000004169 proteins and genes Human genes 0.000 description 1
- 230000010837 receptor-mediated endocytosis Effects 0.000 description 1
- 108020003175 receptors Proteins 0.000 description 1
- 102000005962 receptors Human genes 0.000 description 1
- 231100000419 toxicity Toxicity 0.000 description 1
- 230000001988 toxicity Effects 0.000 description 1
- 230000001228 trophic effect Effects 0.000 description 1
- 229940088594 vitamin Drugs 0.000 description 1
- 229930003231 vitamin Natural products 0.000 description 1
- 235000013343 vitamin Nutrition 0.000 description 1
- 239000011782 vitamin Substances 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
Images
Landscapes
- Medicinal Preparation (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Polysaccharides And Polysaccharide Derivatives (AREA)
- Heterocyclic Carbon Compounds Containing A Hetero Ring Having Oxygen Or Sulfur (AREA)
Abstract
The invention relates to pulullan polysaccharide carrier material with targeting effect of tumour, which is pulullan polysaccharide derivative obtained by reacting biotin with pulullan polysaccharide,and the substitution degree of biotin is 20-85%. In organic solvent, the pulullan polysaccharide carrier material is prepared by reacting biotin, dicyclohexylcarbodiimide, 4-dimethylamino pyridine and pulullan polysaccharide and dialysing. The invention reduces the water solubility of pulullan polysaccharide by combining biotin to make it easy to form microparticles in water solution. And the ability of specific binding cell makes the said derivative be carrier material with targeting effect of tumour. The preparation method is simple, and the reproductivity is good.
Description
Technical field
The present invention relates to field of medicaments Chinese medicine carrier, particularly a kind of pulullan polysaccharide carrier material and preparation method thereof with cancer target effect.This carrier material is a biotinylation pulullan polysaccharide derivant.
Background technology
Pulullan polysaccharide (Pullulan) Chinese also is translated into pullulan, pullulan, general poly-polysaccharide or pulullan.It is the extracellular polysaccharide of the mould generation of falx of sprouting, and with α-1, it is main that the 6-glycosidic bond constitutes the homotype polysaccharide in conjunction with maltose, be glucose by α-1, the 4-glycosidic bond is combined into maltotriose, two ends are again with α-1, the 6-glycosidic bond is with other maltotriose combination, and macromolecule polysaccharide so repeatedly is formed by connecting.α-1,4-glycosidic bond same α-1, the ratio of 6-glycosidic bond is 2: 1, the degree of polymerization (D.P) is 100-5000.Commodity pulullan mean molecule quantity 2X10
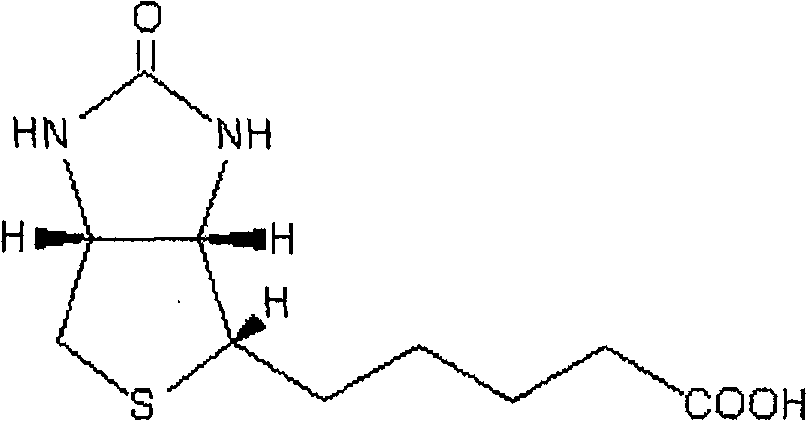
5, approximately form by 480 maltotrioses.This material is nontoxic, no mutagenic action, and colorless and odorless, edible, have excellent biological compatibility, can be degraded by the body endo-amylase, be a kind of excellent drug carrier material.Its aqueous solution viscosity is starkly lower than other polysaccharide solution, makes it more help practical application.Research shows that also pulullan polysaccharide can combine with the galactosylated acceptor of surface of hepatocytes, can be the medicine liver target new material is provided.But, because the water solublity of pulullan polysaccharide is extremely strong, so want in aqueous solution, to prepare can medicine carrying microgranule, must in preparation process, add cross-linking agent, as glutaraldehyde etc., these deleterious cross-linking agent are difficult to thoroughly remove at last handling process, will cause final product to have certain toxicity.In recent years, synthetic hydrophobically modified pulullan polysaccharide derivant has been widely used in the delivery system of number of chemical medicine, protein drug and genomic medicine as pharmaceutical carrier.
Biotin (biotin) claims biotin again, molecular formula C
10H
16N
2O
3S, chemical structural formula:
Biotin is a kind of water soluble vitamins of needed by human, but human body can not self be synthetic, must replenish by extraneous.Studies show that further the nearly all cell surface of human body all has biotin receptor expression, especially tumor cell.Tumor cell increases fast needs a large amount of trophic factors, and the biological example element is so cell surface meeting overexpression can form one like this and more normally organize the higher relatively zone in conjunction with biotin in conjunction with the receptor of biotin around tumor cell.Utilize these characteristics can be with the target spot of biotin receptor, by making material have the tumor cell targeting in conjunction with biological rope as a kind of specific recognition.
As micromolecular biotin, the advantage that more traditional tumor targeted molecular antibody has is its Stability Analysis of Structures, and non-immunogenicity has strong specific bond ability with cell surface, enters cell by receptor-mediated endocytosis by cell membrane.
Summary of the invention
The present invention aims to provide a kind of pulullan polysaccharide carrier material with cancer target effect and preparation method thereof, and this carrier material is a kind of biotinylation pulullan polysaccharide derivant that can the target tumor cell.The present invention is the characteristics in conjunction with pulullan polysaccharide and biotin, by in conjunction with biotin, reduced the water solublity of pulullan polysaccharide, make it be easy in water solublity, form microgranule, the ability of their specific bond cell makes this derivant become a kind of carrier material with cancer target effect simultaneously.
Pulullan polysaccharide carrier material with cancer target effect provided by the invention is the pulullan polysaccharide derivant that biotin and pulullan polysaccharide reaction obtain, the biotin substitution value is 20-85%, it is in organic solvent, biotin, dicyclohexylcarbodiimide and 4-dimethylamino pyridine and pulullan polysaccharide reaction prepare through dialysis.Concrete preparation method is as follows:
1) biotin under the room temperature, dicyclohexylcarbodiimide (DCC) and 4-dimethylamino pyridine (DMAP) are dissolved in the organic solvent, react 1-3 hour;
2) under agitation pulullan polysaccharide is added in the above-mentioned reactant liquor, reacted 3-10 days;
3) with reacting liquid filtering, remove DCU, filtrate is put into bag filter, and dialysis is 3-10 days in 1-10L water dialysis solution, changes liquid every half an hour;
4) white depositions after will dialysing is filtered and is collected, and washs 3-5 time respectively with organic solvent and water, and drying obtains the white solid powder.
Described organic solvent is dimethyl sulfoxide (DMSO).
Described biotin: DCC: DMAP: pulullan polysaccharide=1: 0.1~10: 0.01~10: 0.0001~0.001 (mol ratio).
Total reactant concentration is 0.1~25% in the described organic reagent.
The required organic reagent of described washing is: dichloromethane.
The molecular weight of described pulullan polysaccharide is (20,000-200,000) (1.2 * 10
-4-1.2 * 10
-3).Bag filter molecular cut off 2,000-40,000; Dialysis solution is a deionized water.
Pulullan polysaccharide carrier material with cancer target effect provided by the invention is the characteristics in conjunction with pulullan polysaccharide and biotin, by in conjunction with biotin, reduced the water solublity of pulullan polysaccharide, make it be easy in water solublity, form microgranule, the ability of their specific bond cell makes this derivant become a kind of carrier material with cancer target effect simultaneously.The synthetic pulullan polysaccharide carrier material of the present invention is a biotinylation pulullan polysaccharide derivant, and preparation method is simple, favorable reproducibility.
Description of drawings:
Fig. 1 is biotinylation Pulan polysaccharide derivates and pulullan polysaccharide IR spectrogram.
Fig. 2 is a biotinylation Pulan polysaccharide derivates
1The H-NRM spectrogram.(solvent: deuterated dimethyl sulfoxide: DMSO-d
6).
The specific embodiment
Embodiment 1:
Get biotin 2.44g, DCC2.06g and DMAP 611mg add dissolving fully in the 50ml anhydrous dimethyl sulphoxide (DMSO), and react 1 hour.Subsequently, the 244mg pulullan polysaccharide is joined in the above-mentioned reactant liquor reaction 6 days, be reflected under the magnetic agitation and carry out.After finishing, reaction, removes the sediment D CU in the reactant liquor with reactant liquor buchner funnel sucking filtration three times.The every 25ml of filtrate is put into a bag filter, again bag filter is put into 3L deionized water dialysis 6 days, changed water once in per 30 minutes.After dialysis finishes the white depositions in the bag filter is taken out, use the buchner funnel sucking filtration 3 times, the white solid on the funnel is collected, with 200ml washed with dichloromethane 3 times, reuse deionized water wash 3 times is after the drying, obtain white solid matter, the biotin substitution value is 81%.
Substitution value assay method: accurately take by weighing product 50mg, add dissolving with hydrochloric acid and be settled to 20ml.Hydrochloric acid solution is measured the S constituent content by inductively-coupled plasma spectrometer (IPC), again according to formula:
Substitution value %=S element (molal quantity)/anhydrous grape saccharide residue (molal quantity) * 100
Calculate the biotin substitution value.
Fig. 1 is biotinylation Pulan polysaccharide derivates and pulullan polysaccharide IR spectrogram.Fig. 2 is a biotinylation Pulan polysaccharide derivates
1The H-NRM spectrogram.(solvent: deuterated dimethyl sulfoxide: DMSO-d
6).
Embodiment 2:
Get biotin 1.22g, DCC1.06g and DMAP 300mg add dissolving fully among the anhydrous DMSO of 50ml, and react 1 hour.Subsequently, the 244mg pulullan polysaccharide is joined in the above-mentioned reactant liquor reaction 3 days, be reflected under the magnetic agitation and carry out.After finishing, reaction, removes the sediment D CU in the reactant liquor with reactant liquor buchner funnel sucking filtration three times.The every 25ml of filtrate is put into a bag filter, again bag filter is put into 3L deionized water dialysis 6 days, changed water once in per 30 minutes.After dialysis finishes the white depositions in the bag filter is taken out, use the buchner funnel sucking filtration 3 times, the white solid on the funnel is collected, with 200ml washed with dichloromethane 3 times, reuse deionized water wash 3 times obtains white solid matter after the drying.Accurately take by weighing product 50mg, add dissolving with hydrochloric acid and be settled to 20ml.Hydrochloric acid solution is measured the S constituent content by inductively-coupled plasma spectrometer (IPC), again according to formula:
Substitution value %=S element (molal quantity)/anhydrous grape saccharide residue (molal quantity) * 100
Calculating the biotin substitution value is 21%.
Claims (1)
1, a kind of pulullan polysaccharide carrier material with cancer target effect, by biotin and pharmaceutical carrier prepared in reaction, it is characterized in that: described pharmaceutical carrier is a pulullan polysaccharide, in organic solvent, biotin, dicyclohexylcarbodiimide and 4-dimethylamino pyridine and pulullan polysaccharide reaction, be prepared from through dialysis, the biotin substitution value is 81%, and concrete preparation method is as follows:
1) biotin under the room temperature, dicyclohexylcarbodiimide and 4-dimethylamino pyridine are dissolved in the organic solvent dimethyl sulfoxide, react 1-3 hour;
2) under agitation pulullan polysaccharide is added in the above-mentioned reactant liquor, reacted 3-10 days;
3) with reacting liquid filtering, remove the sediment D CU in the reactant liquor, filtrate is put into bag filter, and dialysis is 3-10 days in 1-10L water dialysis solution, changes liquid every half an hour;
4) white depositions after will dialysing is filtered and is collected, and washs 3-5 time respectively with dichloromethane and water, and drying obtains the white solid powder.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN 200710057172 CN100594938C (en) | 2007-04-20 | 2007-04-20 | Pulullan polysaccharide carrier material having the function of targeting therapy of cancer |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN 200710057172 CN100594938C (en) | 2007-04-20 | 2007-04-20 | Pulullan polysaccharide carrier material having the function of targeting therapy of cancer |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN101036788A CN101036788A (en) | 2007-09-19 |
| CN100594938C true CN100594938C (en) | 2010-03-24 |
Family
ID=38888097
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN 200710057172 Expired - Fee Related CN100594938C (en) | 2007-04-20 | 2007-04-20 | Pulullan polysaccharide carrier material having the function of targeting therapy of cancer |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN100594938C (en) |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101831000A (en) * | 2010-05-21 | 2010-09-15 | 中国医学科学院生物医学工程研究所 | Purification method of acetyl pullulan polysaccharide folate conjugate and preparation method of nanometer particles thereof |
| US10130587B2 (en) | 2011-01-11 | 2018-11-20 | Capsugel Belgium Nv | Hard capsules |
| US11576870B2 (en) | 2017-04-14 | 2023-02-14 | Capsugel Belgium Nv | Pullulan capsules |
| CN110678555B (en) | 2017-04-14 | 2023-10-13 | 比利时胶囊公司 | How to make pullulan |
| CN110051034B (en) * | 2019-05-13 | 2021-05-07 | 云南中烟工业有限责任公司 | Method for improving moisture retention and moisture resistance of cigarette cut tobacco |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1768860A (en) * | 2005-10-24 | 2006-05-10 | 天津大学 | Hydrazide-based assembly method of multiple biological functional factors on the surface of microparticles |
-
2007
- 2007-04-20 CN CN 200710057172 patent/CN100594938C/en not_active Expired - Fee Related
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1768860A (en) * | 2005-10-24 | 2006-05-10 | 天津大学 | Hydrazide-based assembly method of multiple biological functional factors on the surface of microparticles |
Non-Patent Citations (1)
| Title |
|---|
| Self-assembled nanoparticles of hydrophobically-modifiedpolysaccharide bearing vatiamin H as a targeted anti-cancerdrug delivery system. Kun Na,et al.European Journal of Pharmaceutical Science,No.18. 2003 * |
Also Published As
| Publication number | Publication date |
|---|---|
| CN101036788A (en) | 2007-09-19 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR102008768B1 (en) | Cyclodextrin-based polymers for delivering the therapeutic agents covalently bound thereto | |
| KR101347961B1 (en) | Cationized hyaluronic acid | |
| CN101805343A (en) | chemically modified small molecules | |
| CN100594938C (en) | Pulullan polysaccharide carrier material having the function of targeting therapy of cancer | |
| CA2913752A1 (en) | Cyclodextrin-based polymers for therapeutic delivery | |
| TWI482634B (en) | Biomedical composition | |
| CN100571781C (en) | Biotinylated pulullan polysaccharide nanometer grains and preparation method thereof | |
| CN102002117A (en) | Branched chitosan derivative and preparation method thereof | |
| CA2280949C (en) | Dextran derivatives, preparation method and applications as medicine with specific biological action | |
| Monaco et al. | Multi-arm star-shaped glycopolymers with precisely controlled core size and arm length | |
| CN103204998B (en) | Amphipathic polysaccharide/polypeptide block polymer containing azobenzene group and preparation method and application of block polymer | |
| Muhitdinov et al. | Homogenous synthesis of sodium cellulose sulfates with regulable low and high degree of substitutions with SO3/Py in N, N-dimethylacetamide/LiCl | |
| CN111333786B (en) | Preparation method of acid-sensitive adriamycin prodrug based on zwitterion and folic acid targeting | |
| Fan et al. | Synthesis and properties of functional glycomimetics through click grafting of fucose onto chondroitin sulfates | |
| CN108641018B (en) | A kind of sulfated heparin disaccharides grafting polymethyl acyl ethanol amine and preparation method thereof | |
| CN110358098A (en) | A kind of PEG bridging mannose-modified chitosan derivatives and preparation method thereof | |
| CN106749517B (en) | Polysaccharide derivative containing polypeptide targeting factor and preparation method thereof | |
| CN100562526C (en) | Glucose oligosulfate and preparation method thereof | |
| CN104744685A (en) | Three-arm polyethylene glycol derivative and preparation method thereof | |
| CN107375940A (en) | Nano medication with adhesion factor ICAM 1 for target spot prepares and its application | |
| Pedone et al. | Synthesis of two photolabile poly (ethylene glycol) derivatives for protein conjugation | |
| CN102977378A (en) | Amphipathic heparin-based block polymer and preparation method and application thereof | |
| JP5646164B2 (en) | Branched chitosan derivative | |
| TW200538457A (en) | Aqueous solutions comprising camptothecin | |
| CN103920163B (en) | A kind of paclitaxel complex, preparation method and applications |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| CF01 | Termination of patent right due to non-payment of annual fee | ||
| CF01 | Termination of patent right due to non-payment of annual fee |
Granted publication date: 20100324 Termination date: 20160420 |