CN100448846C - A kind of catalytic hydrogenation acylpyridine is the method for substituted piperidine compound hydrochloride - Google Patents
A kind of catalytic hydrogenation acylpyridine is the method for substituted piperidine compound hydrochloride Download PDFInfo
- Publication number
- CN100448846C CN100448846C CNB2006101443591A CN200610144359A CN100448846C CN 100448846 C CN100448846 C CN 100448846C CN B2006101443591 A CNB2006101443591 A CN B2006101443591A CN 200610144359 A CN200610144359 A CN 200610144359A CN 100448846 C CN100448846 C CN 100448846C
- Authority
- CN
- China
- Prior art keywords
- hydrochloride
- alkyl
- dichloroalkane
- reaction
- gem
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
Landscapes
- Hydrogenated Pyridines (AREA)
Abstract
本发明公开了一种催化氢化酰基吡啶为取代哌啶化合物盐酸盐的方法,本发明方法,是将式I或式II结构的酰基吡啶在Pd-C催化剂和偕二氯烷烃的存在下,进行催化氢化反应,得到取代哌啶化合物盐酸盐;其中,R为H、烷基或芳基;所述偕二氯烷烃通式为R1R2CCl2,其中,R1、R2为H、C1-C10的烷基或有1-5个氯原子和/或氟原子取代的C1-C10烷基。本发明通过在催化体系中加入偕二氯烷烃,在温和条件下顺利地将吡啶环还原为哌啶盐酸盐,而且,所生成的盐酸盐为产物的分离和纯化带来诸多方便。本发明方法产率高、选择性好、反应条件温和,并且分离过程简便。
The invention discloses a method for catalyzing the hydrogenation of acylpyridines into substituted piperidine compound hydrochlorides. The method of the invention is to use the acylpyridines with the structure of formula I or formula II in the presence of Pd-C catalyst and gem-dichloroalkane, Carry out catalytic hydrogenation reaction to obtain substituted piperidine compound hydrochloride; wherein, R is H, alkyl or aryl; the general formula of the geminal dichloroalkane is R 1 R 2 CCl 2 , wherein R 1 and R 2 are H, C1-C10 alkyl or C1-C10 alkyl substituted by 1-5 chlorine atoms and/or fluorine atoms. The invention successfully reduces the pyridine ring to piperidine hydrochloride under mild conditions by adding gem-dichloroalkane into the catalytic system, and the generated hydrochloride brings many conveniences to the separation and purification of products. The method of the invention has high yield, good selectivity, mild reaction conditions and simple and convenient separation process.
Description
技术领域 technical field
本发明涉及化合物的制备方法,特别是涉及一种催化氢化酰基吡啶为取代哌啶化合物盐酸盐的方法。The invention relates to a compound preparation method, in particular to a method for catalyzing the hydrogenation of acylpyridines to substituted piperidine compound hydrochlorides.
背景技术 Background technique
取代哌啶及其衍生物是药物研究中的重要药效团和合成中间体。如下所示:4-取代哌啶衍生物1不仅具有潜在的CCR5拮抗活性,也显示出很好的抗HIV-1活性(Shinichi,I.;Takashi,I.;Youichi,N.et al.J.Med.Chem.,2006,49,2784-2793.);Ifenprodil(2)具有抗高血压活性(Etsuo O.;Hitoshi T.;Hiroyuki H.et al.J.Med.Chem.,1993,36,417-420.);类似的二苯基酰胺衍生物3可作为钠离子通道的阻滞剂(IoannisR.;Sheryl H.and Roy D.S.J.Med.Chem.,1996,39,1514-1520.)。Substituted piperidines and their derivatives are important pharmacophores and synthetic intermediates in drug research. As shown below: 4-substituted piperidine derivative 1 not only has potential CCR5 antagonistic activity, but also shows good anti-HIV-1 activity (Shinichi, I.; Takashi, I.; Youichi, N.et al.J .Med.Chem., 2006,49,2784-2793.); Ifenprodil (2) has antihypertensive activity (Etsuo O.; Hitoshi T.; Hiroyuki H.et al.J.Med.Chem., 1993,36 , 417-420.); similar diphenylamide derivatives 3 can be used as blockers of sodium ion channels (IoannisR.; Sheryl H. and Roy D.S.J.Med.Chem., 1996, 39, 1514-1520.).
使用4-取代吡啶为原料在催化氢化条件下对吡啶环进行氢化还原是制备4-取代哌啶衍生物的重要方法之一。例如:专利文献报道,苯基吡啶基酮及其衍生物经水合肼还原得到相应的苄基吡啶化合物后,再用催化氢化的方法将吡啶环还原,得到相应的苄基哌啶化合物(Wick,A.;Frost,J.;Gaudilliere,B.;Bertin,J.;Dupont,R.;Rousseau,J.US Patent 4,690,931,1987)。又例如:α-苯基-4-吡啶甲醇在醋酸溶剂中使用相对较高的温度(60-80℃)和压力(8-10bar)进行Pd-C催化氢化,在氢解除去羟基的同时也将吡啶环还原成为哌啶环(Bela,A.;Agnes,P.;Gabor,T.;Laszlo,V.and Ferenc F.Eur.J.Org.Chem.,2004,3623-3632.)。Using 4-substituted pyridines as raw materials to carry out hydrogenation reduction of pyridine rings under catalytic hydrogenation conditions is one of the important methods for preparing 4-substituted piperidine derivatives. For example: patent literature reports, after phenylpyridyl ketone and derivative thereof obtain corresponding benzylpyridine compound through the reduction of hydrazine hydrate, then use the method for catalytic hydrogenation to reduce pyridine ring, obtain corresponding benzylpiperidine compound (Wick, A.; Frost, J.; Gaudilliere, B.; Bertin, J.; Dupont, R.; Rousseau, J. US Patent 4,690,931, 1987). Another example: α-phenyl-4-pyridinemethanol uses relatively high temperature (60-80°C) and pressure (8-10bar) to carry out catalytic hydrogenation of Pd-C in acetic acid solvent. Reduction of pyridine rings to piperidine rings (Bela, A.; Agnes, P.; Gabor, T.; Laszlo, V. and Ferenc F. Eur. J. Org. Chem., 2004, 3623-3632.).
但是,这两种最具代表性的方法均存在有一定的缺陷。例如:水合肼本身具有较大的毒性和操作的危险性;在醋酸溶剂中进行的氢化反应,基本上失去了环境友好的特性,不仅后处理较繁琐,而且只得到较低至中等的收率。However, both of these two most representative methods have certain defects. For example: hydrazine hydrate itself has great toxicity and danger of operation; the hydrogenation reaction carried out in acetic acid solvent basically loses the characteristics of environmental friendliness, not only the post-treatment is cumbersome, but also only obtains low to medium yield .
发明内容Contents of the invention
本发明的目的是提供一种催化氢化酰基吡啶为取代哌啶化合物盐酸盐的方法。The object of the present invention is to provide a method for catalytic hydrogenation of acylpyridines to be substituted piperidine compound hydrochlorides.
本发明所提供的催化氢化酰基吡啶为取代哌啶化合物盐酸盐的方法,是将式I或式II结构的酰基吡啶在Pd-C催化剂和偕二氯烷烃的存在下,进行催化氢化反应,得到取代哌啶化合物盐酸盐;The catalytic hydrogenation of acylpyridine provided by the present invention is a method for substituted piperidine compound hydrochloride, which is to carry out the catalytic hydrogenation reaction of the acylpyridine with the structure of formula I or formula II in the presence of Pd-C catalyst and gem-dichloroalkane, Obtain substituted piperidine compound hydrochloride;
其中,R为H、烷基或芳基;Wherein, R is H, alkyl or aryl;
所述偕二氯烷烃通式为R1R2CCl2,其中,R1、R2为H、C1-C10的烷基或有1-5个氯原子和/或氟原子取代的C1-C10烷基。The general formula of the gem-dichloroalkane is R 1 R 2 CCl 2 , wherein R 1 and R 2 are H, C1-C10 alkyl or C1-C10 substituted with 1-5 chlorine atoms and/or fluorine atoms alkyl.
在酰基吡啶中,R烷基时,优选的为C1-C20的直链烷基、支链烷基或环烷基;或者带有烷氧基或芳基取代基的C1-C20的直链烷基、支链烷基或环烷基。R为芳基时,优选的芳基为苯基或取代苯基,取代基为H、C1-C10的烷基、C1-C10的烷氧基。In acylpyridine, when R is an alkyl group, it is preferably a C1-C20 straight-chain alkyl, branched-chain alkyl or cycloalkyl; or a C1-C20 straight-chain alkane with an alkoxy or aryl substituent radical, branched alkyl or cycloalkyl. When R is an aryl group, the preferred aryl group is phenyl or substituted phenyl, and the substituents are H, C1-C10 alkyl, and C1-C10 alkoxy.
在本发明中,偕二氯烷烃可以是各种商品化试剂,常用的有二氯甲烷、1,1-二氯乙烷、1,1,2-三氯乙烷、1,1-二氯-2-氟乙烷、1,1-二氯-2,2,2-三氟乙烷、1,1-二氯丙烷、2,2-二氯丙烷、1,1-二氯-3,3-二甲基丁烷、1,1-二氯环戊烷和1,1-二氯环己烷等;优选为1,1-二氯乙烷或者1,1-二氯-3,3-二甲基丁烷。In the present invention, gem-dichloroalkane can be various commercial reagents, commonly used have dichloromethane, 1,1-dichloroethane, 1,1,2-trichloroethane, 1,1-dichloro -2-fluoroethane, 1,1-dichloro-2,2,2-trifluoroethane, 1,1-dichloropropane, 2,2-dichloropropane, 1,1-dichloro-3, 3-Dimethylbutane, 1,1-dichlorocyclopentane and 1,1-dichlorocyclohexane, etc.; preferably 1,1-dichloroethane or 1,1-dichloro-3,3 - Dimethylbutane.
本发明对于反应溶剂没有特殊要求,常用反应溶剂为MeOH、EtOH、i-PrOH、t-BuOH或EtOAc;优选为MeOH或EtOH。The present invention has no special requirements for the reaction solvent, and the commonly used reaction solvents are MeOH, EtOH, i-PrOH, t-BuOH or EtOAc; preferably MeOH or EtOH.
在本发明方法中,Pd-C催化剂和偕二氯烷烃的用量对于反应的进行影响不大,但可以影响反应的速度和反应时间,通常情况下,Pd-C催化剂的用量为所述酰基吡啶重量的1-100%;所述偕二氯烷烃与所述酰基吡啶的摩尔比为1∶1以上,甚至可以作为共溶剂,优选为1.1∶1。In the method of the present invention, the amount of Pd-C catalyst and gem-dichloroalkane has little effect on the reaction, but can affect the speed and reaction time of the reaction. Usually, the amount of Pd-C catalyst is about 100% of the acylpyridine 1-100% by weight; the molar ratio of the gem-dichloroalkane to the acylpyridine is more than 1:1, and can even be used as a co-solvent, preferably 1.1:1.
应用本发明方法,产物取代哌啶化合物盐酸盐有时会有两种:一种是哌啶醇盐酸盐,另一种是烷基哌啶盐酸盐。可以采用如下条件来提高反应的选择性:Applying the method of the present invention, the product substituted piperidine compound hydrochloride sometimes has two kinds: one is piperidinol hydrochloride, and the other is alkyl piperidine hydrochloride. The following conditions can be used to improve the selectivity of the reaction:
当R为芳基时,在常压和稍高于室温的温度(35-45℃)下,可以选择性地得到单一的4-(芳甲基)哌啶盐酸盐。When R is an aryl group, a single 4-(arylmethyl)piperidine hydrochloride can be selectively obtained under normal pressure and a temperature slightly higher than room temperature (35-45° C.).
当R为H或烷基时,在室温和40-80psi的氢压下,可以选择性地得到单一的(α-烷基)哌啶甲醇盐酸盐。When R is H or an alkyl group, a single (α-alkyl) piperidinemethanol hydrochloride can be selectively obtained at room temperature and a hydrogen pressure of 40-80 psi.
本发明通过在催化体系中加入偕二氯烷烃,在温和条件下顺利地将吡啶环还原为哌啶盐酸盐,而且,所生成的盐酸盐为产物的分离和纯化带来诸多方便。本发明方法产率高、选择性好、反应条件温和,并且分离过程简便。The invention successfully reduces the pyridine ring to piperidine hydrochloride under mild conditions by adding gem-dichloroalkane into the catalytic system, and the generated hydrochloride brings many conveniences to the separation and purification of products. The method of the invention has high yield, good selectivity, mild reaction conditions and simple and convenient separation process.
具体实施方式 Detailed ways
在通常的Pd-C催化氢化反应体系中加入偕二氯化物(R1R2CCl2),酰基吡啶[芳基吡啶基酮(4)或者烷基吡啶基酮(5)]几乎可以定量地、高度化学选择性地被转化为相应的4-(芳甲基)哌啶盐酸盐(6)或者(α-烷基)哌啶甲醇盐酸盐(9),反应方程式如下:Adding gem dichloride (R 1 R 2 CCl 2 ) to the usual Pd-C catalytic hydrogenation reaction system, the acylpyridine [aryl pyridyl ketone (4) or alkyl pyridyl ketone (5)] can be almost quantitatively , highly chemoselectively converted into corresponding 4-(arylmethyl)piperidine hydrochloride (6) or (α-alkyl)piperidinemethanol hydrochloride (9), the reaction equation is as follows:
根据对实验跟踪的结果,我们发现酰基吡啶[芳基(4-吡啶基)酮(4)和烷基吡啶基酮(5)]中的酮羰基在Pd-C催化氢化反应中首先被还原成为吡啶甲醇中间体(10和11)。然后,偕二氯化物在Pd-C催化氢化条件下,发生催化氢化脱氯反应。产生的氯化氢将吡啶甲醇中间体(10和11)原位转化成盐酸盐(12和13)。盐酸盐的生成不仅除去了吡啶氮原子对催化剂的毒化影响,而且将吡啶环进一步极化,使芳香稳定性进一步降低,反应历程如下。所以,不仅在温和条件下可以顺利地将吡啶环还原成为哌啶盐酸盐,而且生成的盐酸盐又给产物的分离和纯化带来诸多的方便,简单的过滤即可实现产物与原料的分离。而且,上述反应的反应条件非常温和,可以在低于35℃和80psi氢压下进行。According to the results of the experiment follow-up, we found that the ketone carbonyl in the acylpyridin [aryl(4-pyridyl) ketone (4) and alkylpyridyl ketone (5)] was first reduced to Pyridinemethanol intermediate (10 and 11). Then, the gem dichloride undergoes a catalytic hydrodechlorination reaction under the condition of Pd-C catalytic hydrogenation. The resulting hydrogen chloride converts the pyridinemethanol intermediates (10 and 11) to the hydrochloride salts (12 and 13) in situ. The generation of hydrochloride not only removes the poisoning effect of the pyridine nitrogen atom on the catalyst, but also further polarizes the pyridine ring, which further reduces the aromatic stability. The reaction process is as follows. Therefore, not only can the pyridine ring be successfully reduced to piperidine hydrochloride under mild conditions, but the generated hydrochloride brings a lot of convenience to the separation and purification of the product. Simple filtration can realize the separation of the product and the raw material. separate. Moreover, the reaction conditions of the above reaction are very mild, and can be carried out under 35° C. and 80 psi hydrogen pressure.
在本发明的实验条件下,当-R为芳基或烷基时,所得到的吡啶甲醇中间体(10和11)的反应活性表现出显著差异:当-R为芳基时,化合物10分子中的羟基由于受到相连芳基的活化,继续发生C-O键的氢解反应生成亚甲基,最后选择性地生成4-(芳甲基)哌啶盐酸盐(6)。而当-R为烷基时,化合物11分子中的羟基与烷烃相连,在本发明反应条件下可以控制完全不发生C-O键的氢解反应,最后选择性地生成(α-烷基)哌啶甲醇盐酸盐(9)。Under the experimental conditions of the present invention, when -R is an aryl group or an alkyl group, the reactivity of the obtained pyridinemethanol intermediates (10 and 11) shows a significant difference: when -R is an aryl group, the compound 10 molecule The hydroxyl group in is activated by the connected aryl group, and the hydrogenolysis reaction of the C-O bond continues to generate methylene, and finally selectively generates 4-(arylmethyl)piperidine hydrochloride (6). And when -R is an alkyl group, the hydroxyl group in the compound 11 molecule is connected with the alkane, and under the reaction conditions of the present invention, the hydrogenolysis reaction of the C-O bond can be controlled completely, and finally selectively generate (α-alkyl) piperidine Methanol hydrochloride (9).
在本发明中,偕二氯代烷烃(R2R1CCl2)是指在一个C原子上有两个取代氯原子的烷烃,分子中的R2和R1可以是氢原子或者C1-C10的烷基(直链烷基、支链烷基和环烷基),在这些烷烃链上可以有1-5个氯原子或者氟原子在偕二氯碳原子以外的任何位置上取代。最好选用6个碳原子以内的偕二氯代烷烃,这样在后处理时容易除去。实验证明,偕二氯代烷烃在本发明选择的Pd/C催化氢化实验条件下,所发生的还原去氯反应是一个受底物控制的过程。在没有含氮底物存在时,不会发生还原去氯反应。在含氮底物存在时,偕二氯代烷烃中的两个氯原子只有一个发生还原去氯反应生成氯化氢。所以,该反应任何时候均不会产生过量的氯化氢而使催化剂毒化。最常用的偕二氯代烷烃是那些商品化合物,例如:二氯甲烷、1,1-二氯乙烷、1,1,2-三氯乙烷、1,1-二氯-2-氟乙烷、1,1-二氯-2,2,2-三氟乙烷、1,1-二氯丙烷、2,2-二氯丙烷、1,1-二氯-3,3-二甲基丁烷、等等1,1-二氯环戊烷和1,1-二氯环己烷等等。最好使用1,1-二氯乙烷或者1,1-二氯-3,3-二甲基丁烷,因为它们的沸点相对较高,操作时容易定量进行。当它们释放出一个氯原子后生成的一氯化合物的沸点降低了许多,又容易从体系中除去。理论上,偕二氯代烷烃与底物的摩尔比是1∶1,但最好使用1.1∶1。过量的偕二氯代烷烃对反应的选择性和产率不产生负面影响。所以,在实际操作中偕二氯代烷烃的用量相对随意。In the present invention, gem-dichloroalkane (R 2 R 1 CCl 2 ) refers to an alkane with two substituted chlorine atoms on one C atom, and R 2 and R 1 in the molecule can be hydrogen atoms or C1-C10 Alkyl groups (straight-chain alkyl, branched-chain alkyl and cycloalkyl), in these alkane chains may have 1-5 chlorine atoms or fluorine atoms substituted at any position other than the geminal dichloro carbon atom. It is best to use gem-dichloroalkanes with less than 6 carbon atoms, which are easy to remove during post-processing. Experiments prove that under the selected Pd/C catalytic hydrogenation experimental conditions of the present invention, the reductive dechlorination reaction of gem-dichloroalkanes is a process controlled by the substrate. In the absence of nitrogenous substrates, reductive dechlorination does not occur. In the presence of nitrogen-containing substrates, only one of the two chlorine atoms in the gem-dichloroalkane undergoes a reductive dechlorination reaction to generate hydrogen chloride. Therefore, the reaction does not generate excess hydrogen chloride at any time to poison the catalyst. The most commonly used gem-dichloroalkanes are those commercial compounds such as: dichloromethane, 1,1-dichloroethane, 1,1,2-trichloroethane, 1,1-dichloro-2-fluoroethane alkanes, 1,1-dichloro-2,2,2-trifluoroethane, 1,1-dichloropropane, 2,2-dichloropropane, 1,1-dichloro-3,3-dimethyl Butane, etc. 1,1-dichlorocyclopentane and 1,1-dichlorocyclohexane, etc. It is best to use 1,1-dichloroethane or 1,1-dichloro-3,3-dimethylbutane because of their relatively high boiling point and easy quantification during operation. When they release a chlorine atom, the boiling point of the monochlorine compound generated is much lower, and it is easy to remove from the system. Theoretically, the molar ratio of gem-dichloroalkane to substrate is 1:1, but preferably 1.1:1 is used. An excess of gem-dichloroalkane does not negatively affect the selectivity and yield of the reaction. Therefore, in actual operation, the amount of gem-dichloroalkane is relatively arbitrary.
在发明中,催化氢化还原反应可以在MeOH、EtOH、i-PrOH、t-BuOH和EtOAc等溶剂中进行。实验证明在催化剂用量、反应温度、氢气压力等等相同的条件下,MeOH和EtOH为溶剂时产物的产率和选择性最好,更优选反应溶剂为MeOH。In the invention, the catalytic hydrogenation reduction reaction can be carried out in solvents such as MeOH, EtOH, i-PrOH, t-BuOH and EtOAc. Experiments have shown that under the same conditions of catalyst consumption, reaction temperature, hydrogen pressure, etc., the yield and selectivity of the product are the best when MeOH and EtOH are solvents, and the more preferred reaction solvent is MeOH.
本发明中,反应温度一般在反应溶剂的沸点之下;在高温条件下,可以加快反应速度,此时需要选择高沸点溶剂,以保证反应顺利进行。反应中,H2压力一般可控制在常压至80psi范围之内,提高H2压力,也有利于反应的进行,但对反应的设备有特殊的要求。In the present invention, the reaction temperature is generally below the boiling point of the reaction solvent; under high temperature conditions, the reaction speed can be accelerated. At this time, a high boiling point solvent needs to be selected to ensure the smooth progress of the reaction. During the reaction, the H 2 pressure can generally be controlled within the range of normal pressure to 80 psi, increasing the H 2 pressure is also beneficial to the reaction, but there are special requirements for the reaction equipment.
第一部分:芳基(4-吡啶基)酮(4)的氢化还原Part 1: Hydrogenation reduction of aryl(4-pyridyl)ketones (4)
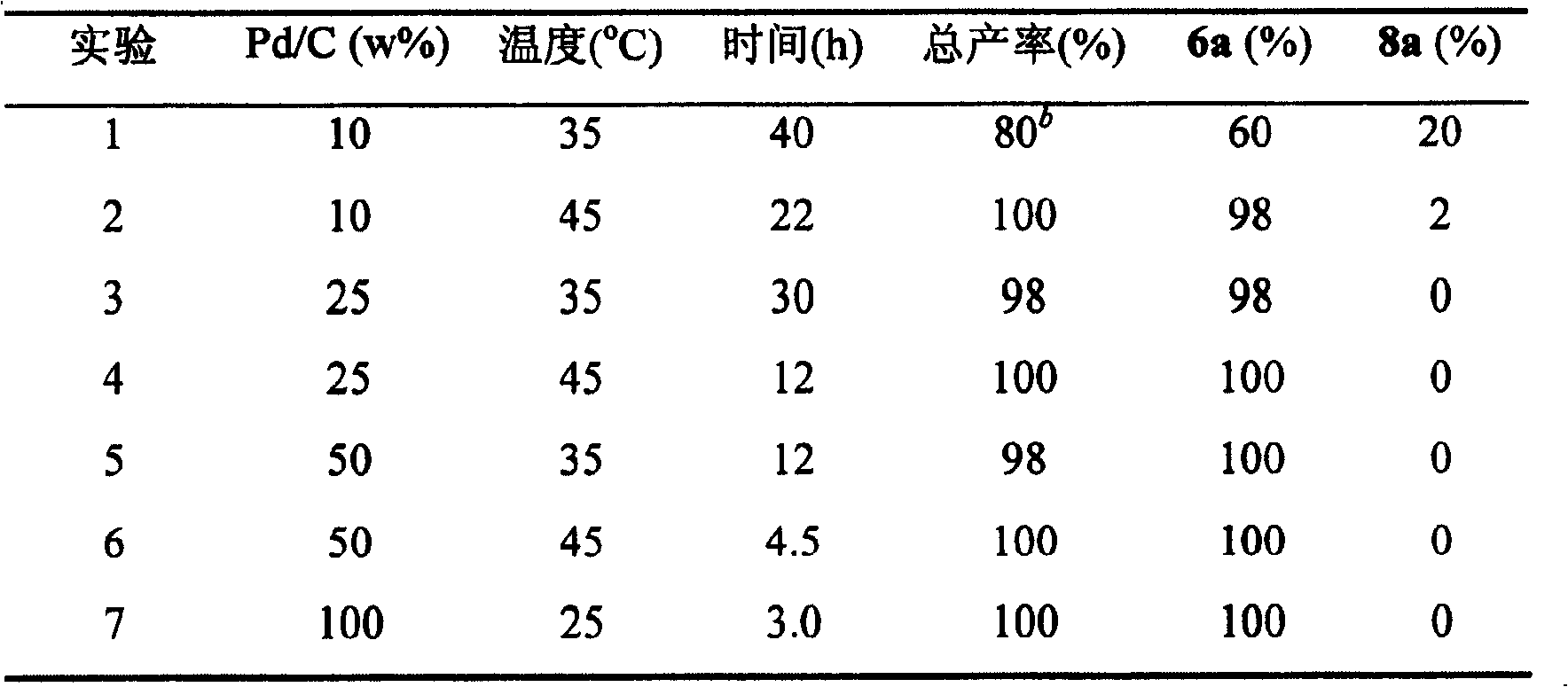
如下式所示:芳基(4-吡啶基)酮(4)的还原可用苯基(4-吡啶基)酮(4a)为例来说明。在本发明中,Pd-C催化剂中Pd的含量可以在1%-50%范围即可,实际情况主要受到反应压力、温度和时间的影响。一般可以方便地选用商品化的5%或者10%的Pd-C催化剂。从反应的综合条件考量,最好选用10%的Pd-C催化剂。10%Pd-C催化剂的用量一般为底物重量的1-100%即可,如表1所示。从经济的角度考量,最好选用25%;少于25%的用量仍然可以完成该反应,但是需要提高反应温度或者延长反应时间。从省时的角度考量,实验室操作最好选用50%的用量。使用超过100%的用量时则可以使反应在常温常压下进行。As shown in the following formula: the reduction of aryl (4-pyridyl) ketone (4) can be illustrated with phenyl (4-pyridyl) ketone (4a) as an example. In the present invention, the content of Pd in the Pd-C catalyst can be in the range of 1%-50%, and the actual situation is mainly affected by reaction pressure, temperature and time. Generally, a commercially available 5% or 10% Pd-C catalyst can be conveniently selected. Considering the comprehensive conditions of the reaction, it is best to use 10% Pd-C catalyst. The dosage of the 10% Pd-C catalyst is generally 1-100% of the weight of the substrate, as shown in Table 1. From an economic point of view, it is best to select 25%; the amount of less than 25% can still complete the reaction, but it needs to increase the reaction temperature or prolong the reaction time. From the perspective of saving time, it is best to use 50% of the dosage for laboratory operations. When the amount exceeds 100%, the reaction can be carried out at normal temperature and pressure.
表1.Pd/C催化剂用量对4a催化氢化反应的影响a Table 1. Effect of Pd/C catalyst dosage on 4a catalytic hydrogenation a
a.氢压=1大气压。b.母液中含有不成盐的混合物。a. Hydrogen pressure = 1 atmosphere. b. The mother liquor contains a non-salt-forming mixture.
在本发明中,芳基(4-吡啶基)酮中的芳基(-R)主要指苯基和取代苯基,取代基的范围包括氢原子(H);烷基(-R),其中包括1-10个碳原子的直链烷基、支链烷基和环烷基;烷氧基(-OR),其中R=1-10个碳原子的直链烷基、支链烷基和环烷基。取代基可以在苯环的邻位、间位、或对位取代,也可以同时在这些位置上进行取代。In the present invention, aryl (-R) in aryl (4-pyridyl) ketone mainly refers to phenyl and substituted phenyl, and the scope of substituent includes hydrogen atom (H); Alkyl (-R), wherein Including straight-chain alkyl, branched-chain alkyl and cycloalkyl of 1-10 carbon atoms; alkoxy (-OR), wherein R = straight-chain alkyl, branched-chain alkyl and Cycloalkyl. The substituents may be substituted at the ortho, meta, or para positions of the benzene ring, or may be substituted at these positions simultaneously.
如果苯环上有一个或者多个氟原子的话,反应受到明显的影响。例如:(4-氟苯基)(4-吡啶基)酮(4h)在同样的条件下生成4-(4-氟苯甲基)哌啶盐酸盐(6h)和(4-氟苯基)(4-哌啶基)甲醇盐酸盐(8h)的混合物。这主要是因为苯环对苄醇的活化明显降低,以至于反应中间体苄醇不能够完全发生羟基的氢解反应。但是,当使用CH2Cl2作为氯源时,该反应可以化学选择性地生成单一产物(4-氟苯基)(4-哌啶基)甲醇盐酸盐(8h)(表2)。这主要是因为CH2Cl2具有更高的氢化去氯活性,可以快速地生成吡啶盐酸盐。一旦吡啶盐酸盐被还原成为哌啶盐酸盐,原来的苄基醇就被转化成为哌啶基甲醇。因此,醇羟基的C-O氢解反应就完全被阻止。If there are one or more fluorine atoms on the benzene ring, the reaction is significantly affected. For example: (4-fluorophenyl) (4-pyridyl) ketone (4h) generates 4-(4-fluorobenzyl) piperidine hydrochloride (6h) and (4-fluorophenyl ) (4-piperidinyl)methanol hydrochloride (8h) mixture. This is mainly because the activation of benzyl alcohol by the benzene ring is significantly reduced, so that the reaction intermediate benzyl alcohol cannot completely undergo the hydrogenolysis reaction of the hydroxyl group. However, when CH2Cl2 was used as the chlorine source, the reaction could chemoselectively yield the single product (4-fluorophenyl)(4-piperidinyl)methanol hydrochloride (8h) (Table 2). This is mainly because CH 2 Cl 2 has a higher hydrodechlorination activity and can quickly generate pyridine hydrochloride. Once pyridine hydrochloride is reduced to piperidine hydrochloride, the original benzyl alcohol is converted to piperidinylmethanol. Therefore, the CO hydrogenolysis reaction of alcoholic hydroxyl groups is completely prevented.
表24-(4-氟苯甲酰基)吡啶(4h)的选择性催化氢化还原a Selective catalytic hydrogenation reduction of table 24-(4-fluorobenzoyl)pyridine (4h) a
a.Pd-C催化剂用量为50%w/w。b.实验1-4的母液中含有不成盐的混合物,但不影响分离。a. Pd-C catalyst dosage is 50% w/w. b. The mother liquor of experiments 1-4 contains a non-salt-forming mixture, but it does not affect the separation.
在本发明的实验条件下,芳基吡啶基酮反应的第一步是将羰基还原成为羟基,然后就产生二个竞争反应:a)吡啶环被还原成为哌啶环;b)羟基被氢解生成亚甲基。反应b的快慢对反应a不产生任何影响。但是,反应a首先完成的话,吡啶对羟基的活化能力就消失,因此反应b中羟基被氢解生成亚甲基的速度将大幅度地降低。所以,必须保持适当的反应温度,增加羟基的氢解能力,才能选择性地得到单一的4-(芳甲基)哌啶盐酸盐(6)。在常压下,35-55℃之间进行的反应给出最理想的反应速度、选择性、和产率,首选使用35℃。低于35℃时,有时产物中有少量的苄醇化合物。温度对反应速度的影响非常显著,例如:苯基(4-吡啶基)酮(4a)在35℃时需要12小时完成反应,在55℃时只需要4.5个小时。如果应用更高的温度则需要更换合适的溶剂,在实验室制备中没有必要。Under the experimental conditions of the present invention, the first step of the aryl pyridyl ketone reaction is to reduce the carbonyl to a hydroxyl group, and then two competing reactions are produced: a) the pyridine ring is reduced to a piperidine ring; b) the hydroxyl group is hydrogenolyzed Generate methylene. The speed of reaction b has no effect on reaction a. However, if reaction a is completed first, the activation ability of pyridine to hydroxyl will disappear, so the rate of hydrogenolysis of hydroxyl to form methylene in reaction b will be greatly reduced. Therefore, it is necessary to maintain an appropriate reaction temperature and increase the hydrogenolysis ability of the hydroxyl group to selectively obtain a single 4-(arylmethyl)piperidine hydrochloride (6). Under normal pressure, the reaction between 35-55°C gives the most ideal reaction speed, selectivity, and yield, and 35°C is preferred. When the temperature is lower than 35°C, sometimes there is a small amount of benzyl alcohol compound in the product. The influence of temperature on the reaction rate is very significant, for example: phenyl (4-pyridyl) ketone (4a) needs 12 hours to complete the reaction at 35°C, but only 4.5 hours at 55°C. If a higher temperature is applied, the appropriate solvent needs to be changed, which is not necessary in laboratory preparation.
反应中氢气压力的变化对反应速度和产物的选择性也有显著的的影响。如果保持反应温度在35-55℃之间,在15-80psi之间氢压下进行的反应均不对化学选择性产生明显的影响。但是在室温下,氢气压力的变化对反应选择性的影响相当明显。例如:苯基(4-吡啶基)酮(4a)在常压下生成1∶3的6a和8a的混合物;但是80psi氢气压力下定量地生成单一的产物4-(苯甲基)哌啶盐酸盐(6a)。The change of hydrogen pressure in the reaction also has a significant impact on the reaction rate and product selectivity. If the reaction temperature is kept between 35-55°C, the reactions carried out under the hydrogen pressure between 15-80psi have no obvious influence on the chemoselectivity. But at room temperature, the influence of the change of hydrogen pressure on the reaction selectivity is quite obvious. For example: phenyl (4-pyridyl) ketone (4a) generates a 1:3 mixture of 6a and 8a under normal pressure; however, a single product 4-(benzyl) piperidinium salt is quantitatively generated under 80 psi hydrogen pressure salt (6a).
按照如下实验条件:以ClCH2CHCl2或CH2Cl2作为偕二氯烷烃,MeOH为溶剂,在35℃、1atm氢压下,芳基(4-吡啶基)酮底物分子均可以顺利地被转化成为4-(芳甲基)哌啶盐酸盐(6)。如表3所示,所有的转变具有高度的化学选择性和几乎定量的产率。According to the following experimental conditions: using ClCH 2 CHCl 2 or CH 2 Cl 2 as gem-dichloroalkane, MeOH as solvent, at 35°C and 1 atm hydrogen pressure, aryl (4-pyridyl) ketone substrate molecules can be successfully It is converted into 4-(arylmethyl)piperidine hydrochloride (6). As shown in Table 3, all transformations were highly chemoselective and almost quantitative in yield.
表3.芳基(4-吡啶基)酮(4)的氢化还原举例Table 3. Examples of hydrogenation reduction of aryl (4-pyridyl) ketones (4)
a.Pd/C量为100%w/w;b.CH2Cl2被用做氯源。 a . Pd/C amount is 100% w/w; b. CH2Cl2 is used as chlorine source.
实施例1、4-苄基哌啶盐酸盐(6a)的制备The preparation of embodiment 1,4-benzylpiperidine hydrochloride (6a)
将4-苯甲酰基吡啶(4a,366mg,2.0mmol),10%Pd-C(183mg,50%w/w),和1,1,2-三氯乙烷(320mg,2.4mmol)在甲醇(30mL)中生成的悬浮液,在35℃和一个大气压的氢压下搅拌12h后,氢气吸收完全停止。过滤出Pd-C后,蒸去滤液中的甲醇,生成的白色固体用无水乙醚洗涤后,经CH3OH-Et2O重结晶得到4-苄基哌啶盐酸盐(6a)的白色晶体(42mg,98%)。4-Benzoylpyridine (4a, 366 mg, 2.0 mmol), 10% Pd-C (183 mg, 50% w/w), and 1,1,2-trichloroethane (320 mg, 2.4 mmol) were dissolved in methanol (30 mL), after stirring the resulting suspension at 35 °C under 1 atmosphere of hydrogen pressure for 12 h, hydrogen uptake completely ceased. After filtering out Pd-C, the methanol in the filtrate was distilled off, and the resulting white solid was washed with anhydrous ether, and then recrystallized from CH 3 OH-Et 2 O to obtain the white 4-benzylpiperidine hydrochloride (6a). Crystals (42 mg, 98%).
mp 180-181℃(MeOH-Et2O);mp 180-181°C (MeOH- Et2O );
IR:v3427,3371,3026,2733,1595,1497,1471,1452,1074cm-1;IR: v3427, 3371, 3026, 2733, 1595, 1497, 1471, 1452, 1074cm-1 ;
13C NMR:δ140.0,129.4,128.6,126.4,44.1,41.5,35.1,28.2; 13 C NMR: δ140.0, 129.4, 128.6, 126.4, 44.1, 41.5, 35.1, 28.2;
Calcd for C12H18ClN:C,68.07%;H,8.57%;N,6.62%;Found:C,67.94%;H,8.56%;N,6.51%.Calcd for C 12 H 18 ClN: C, 68.07%; H, 8.57%; N, 6.62%; Found: C, 67.94%; H, 8.56%; N, 6.51%.
实施例2、4-(4-甲基苄基)哌啶盐酸盐(6b)的制备The preparation of embodiment 2,4-(4-methylbenzyl)piperidine hydrochloride (6b)
按照实施例1相同的实验步骤,以(4-甲基苯基)-4-吡啶基酮(4b)为原料,得到4-(4-甲基苄基)哌啶盐酸盐(6b);According to the same experimental procedure as in Example 1, using (4-methylphenyl)-4-pyridyl ketone (4b) as a raw material, 4-(4-methylbenzyl)piperidine hydrochloride (6b) was obtained;
mp 218-220℃(MeOH-Et2O);mp 218-220°C (MeOH- Et2O );
IR:v3448,3382,3035,2505,1593,1515,1473,1451cm-1;IR: v3448, 3382, 3035, 2505, 1593, 1515, 1473, 1451cm -1 ;
13C NMR:δ136.8,135.5,129.2,129.0,43.9,41.2,35.2,28.2,20.4; 13 C NMR: δ136.8, 135.5, 129.2, 129.0, 43.9, 41.2, 35.2, 28.2, 20.4;
Calcd for C13H20ClN:C,69.16%;H,8.93%;N,6.20%;Found:C,69.18%;H,8.93%;N,6.14%.Calcd for C 13 H 20 ClN: C, 69.16%; H, 8.93%; N, 6.20%; Found: C, 69.18%; H, 8.93%; N, 6.14%.
实施例3、4-(2-甲基苄基)哌啶盐酸盐(6c)的制备The preparation of embodiment 3,4-(2-methylbenzyl)piperidine hydrochloride (6c)
按照实施例1相同的实验步骤,从(2-甲基苯基)-4-吡啶基酮(4c)得到4-(2-甲基苄基)哌啶盐酸盐(6c);According to the same experimental procedure as in Example 1, 4-(2-methylbenzyl)piperidine hydrochloride (6c) was obtained from (2-methylphenyl)-4-pyridylketone (4c);
mp 225-207℃(MeOH-Et2O);mp 225-207°C (MeOH- Et2O );
IR:v3447,3374,2726,1635,1592,1494,1456cm-1;IR: v3447, 3374, 2726, 1635, 1592, 1494, 1456cm-1 ;
13C NMR:δ138.2,136.9,130.4,130.3,126.6,125.9,44.1,38.7,34.0,28.3,18.6; 13 C NMR: δ138.2, 136.9, 130.4, 130.3, 126.6, 125.9, 44.1, 38.7, 34.0, 28.3, 18.6;
Calcd for C13H20ClN:C,69.16%;H,8.93%;N,6.20%;Found:C,69.11%;H,8.89%;N,6.26%.Calcd for C 13 H 20 ClN: C, 69.16%; H, 8.93%; N, 6.20%; Found: C, 69.11%; H, 8.89%; N, 6.26%.
实施例4、4-(4-甲氧基苄基)哌啶盐酸盐(6d)的制备The preparation of embodiment 4,4-(4-methoxybenzyl)piperidine hydrochloride (6d)
按照实施例1相同的实验步骤,从(4-甲氧基苯基)-4-吡啶基酮(4d)得到4-(4-甲氧基苄基)哌啶盐酸盐(6d);According to the same experimental procedure as in Example 1, 4-(4-methoxybenzyl)piperidine hydrochloride (6d) was obtained from (4-methoxyphenyl)-4-pyridylketone (4d);
mp 172-174℃(MeOH-Et2O);mp 172-174°C (MeOH- Et2O );
IR:v3448,2841,2723,1613,1593,1513,1455,1443,1246,1029cm-1;IR: v3448, 2841, 2723, 1613, 1593, 1513, 1455, 1443, 1246, 1029cm -1 ;
13C NMR:δ157.2,132.6,130.4,113.9,55.4,44.0,40.5,35.2,28.1; 13 C NMR: δ157.2, 132.6, 130.4, 113.9, 55.4, 44.0, 40.5, 35.2, 28.1;
Calcd for C13H20ClNO:C,64.59%;H,8.34%;N,5.79%;Found:C,64.48%;H,8.35%;N,5.66%.Calcd for C 13 H 20 ClNO: C, 64.59%; H, 8.34%; N, 5.79%; Found: C, 64.48%; H, 8.35%; N, 5.66%.
实施例5、4-(3-甲氧基苄基)哌啶盐酸盐(6e)的制备The preparation of embodiment 5,4-(3-methoxybenzyl)piperidine hydrochloride (6e)
按照实施例1相同的实验步骤,从(3-甲氧基苯基)-4-吡啶基酮(4e)得到4-(3-甲氧基苄基)哌啶盐酸盐(6e);According to the same experimental procedure as in Example 1, 4-(3-methoxybenzyl)piperidine hydrochloride (6e) was obtained from (3-methoxyphenyl)-4-pyridylketone (4e);
mp 150-152℃(MeOH-Et2O);mp 150-152°C (MeOH- Et2O );
IR:v3423,2840,2466,1611,1582,1488,1470,1454,1259,1046cm-1;IR: v3423, 2840, 2466, 1611, 1582, 1488, 1470, 1454, 1259, 1046cm -1 ;
13C NMR:δ158.9,141.8,129.7,122.2,114.8,111.7,55.3,44.0,41.5,35.0,28.2; 13 C NMR: δ158.9, 141.8, 129.7, 122.2, 114.8, 111.7, 55.3, 44.0, 41.5, 35.0, 28.2;
Calcd for C13H20ClNO:C,64.59%;H,8.34%;N,5.79%;Found:C,64.67%;H,8.28%;N,5.71%.Calcd for C 13 H 20 ClNO: C, 64.59%; H, 8.34%; N, 5.79%; Found: C, 64.67%; H, 8.28%; N, 5.71%.
实施例6、4-(2,4-二甲氧基苄基)哌啶盐酸盐(6f)的制备The preparation of embodiment 6,4-(2,4-dimethoxybenzyl)piperidine hydrochloride (6f)
按照实施例1相同的实验步骤,从(2,4-二甲氧基苯基)-4-吡啶基酮(4f)得到4-(2,4-二甲氧基苄基)哌啶盐酸盐(6f);According to the same experimental procedure as in Example 1, 4-(2,4-dimethoxybenzyl) piperidine hydrochloride was obtained from (2,4-dimethoxyphenyl)-4-pyridyl ketone (4f) salt (6f);
mp 178-180℃(MeOH-Et2O);mp 178-180°C (MeOH- Et2O );
IR:v3449,2932,1611,1506,1464,1146cm-1;IR: v3449, 2932, 1611, 1506, 1464, 1146cm -1 ;
13C NMR:δ158.8,158.2,131.4,120.4,104.8,98.6,55.44,55.36,44.0,35.2,33.9,28.3; 13 C NMR: δ158.8, 158.2, 131.4, 120.4, 104.8, 98.6, 55.44, 55.36, 44.0, 35.2, 33.9, 28.3;
Calcd for C14H22ClNO2:C,61.87%;H,8.16%;N,5.15%;Found:C,61.68%;H,8.18%;N,5.21%.Calcd for C 14 H 22 ClNO 2 : C, 61.87%; H, 8.16%; N, 5.15%; Found: C, 61.68%; H, 8.18%; N, 5.21%.
实施例7、4-(3,4-二甲基苄基)哌啶盐酸盐(6g)的制备The preparation of embodiment 7,4-(3,4-dimethylbenzyl)piperidine hydrochloride (6g)
按照实施例1相同的实验步骤,从(3,4-二甲基苯基)-4-吡啶基酮(4g)得到4-(3,4-二甲基苄基)哌啶盐酸盐(6g);According to the same experimental procedure as in Example 1, 4-(3,4-dimethylbenzyl) piperidine hydrochloride was obtained from (3,4-dimethylphenyl)-4-pyridyl ketone (4g) ( 6g);
mp 218-219℃(MeOH-Et2O);mp 218-219°C (MeOH- Et2O );
IR:v3420,2909,1586,12053420,2909,1586,1205cm-1;IR: v3420, 2909, 1586, 12053420, 2909, 1586, 1205cm-1 ;
13C NMR:δ137.3,136.2,133.7,130.4,129.5,126.7,43.9,41.3,35.3,28.3,19.1,18.7; 13 C NMR: δ137.3, 136.2, 133.7, 130.4, 129.5, 126.7, 43.9, 41.3, 35.3, 28.3, 19.1, 18.7;
Calcd for C14H22ClN:C,70.13%;H,9.25%;N,5.84%;Found:C,69.93%;H,9.18%;N,5.96%.Calcd for C 14 H 22 ClN: C, 70.13%; H, 9.25%; N, 5.84%; Found: C, 69.93%; H, 9.18%; N, 5.96%.
实施例8、4-(3,5-二甲基苄基)哌啶盐酸盐(6h)的制备Embodiment 8, the preparation of 4-(3,5-dimethylbenzyl)piperidine hydrochloride (6h)
按照实施例1相同的实验步骤,从(3,5-二甲基苯基)-4-吡啶基酮(4h)得到4-(3,5-二甲基苄基)哌啶盐酸盐(6h);According to the same experimental procedure as in Example 1, 4-(3,5-dimethylbenzyl) piperidine hydrochloride was obtained from (3,5-dimethylphenyl)-4-pyridyl ketone (4h) ( 6h);
mp 184-186℃(MeOH-Et2O);mp 184-186°C (MeOH- Et2O );
IR:v3442,2955,2768,1607,1588,1450cm-1;IR: v3442, 2955, 2768, 1607, 1588, 1450cm -1 ;
13C NMR:δ139.8,137.5,127.4,127.0,43.9,41.7,35.2,28.3,20.8; 13 C NMR: δ139.8, 137.5, 127.4, 127.0, 43.9, 41.7, 35.2, 28.3, 20.8;
Calcd for C14H22ClNO2:C,61.87%;H,8.16%;N,5.15%;Found:C,61.68%;H,8.18%;N,5.21%.Calcd for C 14 H 22 ClNO 2 : C, 61.87%; H, 8.16%; N, 5.15%; Found: C, 61.68%; H, 8.18%; N, 5.21%.
实施例9、α-(4-氟苯基)-4-哌啶甲醇盐酸盐(6i)的制备Example 9, Preparation of α-(4-fluorophenyl)-4-piperidinemethanol hydrochloride (6i)
按照实施例1相同的实验步骤,从(4-氟苯基)-4-吡啶基酮(4i)得到α-(4-氟苯基)-4-哌啶甲醇盐酸盐(6i);According to the same experimental procedure as in Example 1, α-(4-fluorophenyl)-4-piperidinemethanol hydrochloride (6i) was obtained from (4-fluorophenyl)-4-pyridylketone (4i);
mp 248-250℃(dec.),(MeOH-Et2O);mp 248-250°C (dec.), (MeOH-Et 2 O);
IR:v3339,2950,2813,2718,1509,1223cm-1;IR: v3339, 2950, 2813, 2718, 1509, 1223cm -1 ;
13C NMR:δ163.8,160.6,137.5,128.7,128.5,115.5,115.2,76.6,43.9,43.8,40.1,25.1,25.0; 13 C NMR: δ163.8, 160.6, 137.5, 128.7, 128.5, 115.5, 115.2, 76.6, 43.9, 43.8, 40.1, 25.1, 25.0;
Calcd for C12H17ClFNO:C,58.66%;H,6.97%;N,5.70%;Found:C,58.67%;H,7.12%;N,5.55%.Calcd for C 12 H 17 ClFNO: C, 58.66%; H, 6.97%; N, 5.70%; Found: C, 58.67%; H, 7.12%; N, 5.55%.
第二部分:烷基吡啶基酮(5)的还原Part II: Reduction of alkyl pyridyl ketones (5)
烷基吡啶基酮(5)的还原在催化剂的应用和偕二氯代烷烃两项上与芳基(4-吡啶基)酮(4)中的表述完全一致。The reduction of alkyl pyridyl ketones (5) is exactly the same as that of aryl (4-pyridyl) ketones (4) in terms of the use of catalysts and gem-dichloroalkanes.
在本发明中,底物烷基吡啶基酮可以是2-或者4-烷基吡啶基酮,其中的烷基(-R)可以包括氢原子(H);1-20个碳原子的直链烷基、支链烷基和环烷基,其中在距离酮羰基两个碳原子以外的任何碳原子上可以有烷氧基取代(-OR1,其中R1=1-10个碳原子的直链烷基、支链烷基和环烷基)和芳基取代(-Ar,其中Ar=苯基;在苯环任何位置上带有1-4个碳原子的直链烷烃、支链烷烃和环烷烃;或者苯环任何位置上带有1-4个碳原子的直链烷氧基、支链烷氧基和环烷氧基)。In the present invention, the substrate alkyl pyridyl ketone can be 2- or 4-alkyl pyridyl ketone, wherein the alkyl (-R) can include a hydrogen atom (H); a straight chain of 1-20 carbon atoms Alkyl, branched chain alkyl and cycloalkyl, wherein there may be alkoxy substitution on any carbon atom other than two carbon atoms from the ketone carbonyl group (-OR 1 , wherein R 1 = straight chain of 1-10 carbon atoms chain alkyl, branched chain alkyl and cycloalkyl) and aryl substitution (-Ar, where Ar = phenyl; straight-chain alkanes, branched-chain alkanes and cycloalkane; or straight-chain alkoxy, branched-chain alkoxy and cycloalkoxy with 1-4 carbon atoms at any position of the benzene ring).
如前所述:当烷基吡啶基酮(5)被氢化还原成为吡啶基苄醇(11)时,后续反应中存在有二个竞争反应:a)吡啶环被还原成为哌啶环;b)苄醇被氢解生成亚甲基。如果反应b先发生,则最后生成烷基哌啶盐酸盐(7)。如果反应a先发生,原来的吡啶基苄醇则成为哌啶基醇。因此,醇羟基完全失去被氢解的可能性,最后生成(α-烷基)哌啶甲醇盐酸盐(9)。As mentioned above: when the alkyl pyridyl ketone (5) is hydrogenated and reduced to pyridyl benzyl alcohol (11), there are two competing reactions in the subsequent reaction: a) the pyridine ring is reduced to a piperidine ring; b) Benzyl alcohol is hydrogenolyzed to form methylene. If reaction b occurs first, the alkylpiperidine hydrochloride (7) is finally formed. If reaction a occurs first, the original pyridyl benzyl alcohol becomes piperidyl alcohol. Therefore, the alcoholic hydroxyl group completely loses the possibility of being hydrogenolyzed, and finally generates (α-alkyl)piperidinemethanol hydrochloride (9).
按照前面所述的反应条件,虽然烷基吡啶基酮(5)的氢化还原总产率几乎总是定量完成,但生成的产物均为烷基哌啶盐酸盐(7)和(α-烷基)哌啶甲醇盐酸盐(9)的混合物。我们发现混合物中烷基哌啶盐酸盐(7)和(α-烷基)哌啶甲醇盐酸盐(9)的比例随着烷基链长的变化而变化。如表4所示:烷基的碳链越长,生成(α-烷基)哌啶甲醇盐酸盐(9)的比例越高。According to the reaction conditions described above, although the overall yield of the hydrogenation reduction of alkylpyridyl ketones (5) is almost always quantitative, the products generated are all alkylpiperidine hydrochlorides (7) and (α-alkane base) mixtures of piperidinemethanol hydrochloride (9). We found that the ratio of alkylpiperidine hydrochloride (7) and (α-alkyl)piperidinemethanol hydrochloride (9) in the mixture varied with the alkyl chain length. As shown in Table 4: the longer the carbon chain of the alkyl group, the higher the ratio of (α-alkyl) piperidinemethanol hydrochloride (9) is generated.
表4.烷基吡啶基酮(5)在氢化还原中竞争反应的变化规律Table 4. Variation of competition reaction of alkylpyridinyl ketone (5) in hydrogenation reduction
为了增加氢化还原产物的化学选择性,以4-吡啶基甲醛(5a)为例,对反应温度进行了测试。结果显示:升高反应温度可以明显加快反应速度。虽然有增加化合物7a比例的趋势,但在55℃时仍然有25%的产物9a。In order to increase the chemoselectivity of hydrogenated reduction products, the reaction temperature was tested by taking 4-pyridylcarbaldehyde (5a) as an example. The results show that increasing the reaction temperature can significantly speed up the reaction rate. Although there was a tendency to increase the proportion of compound 7a, there was still 25% product 9a at 55 °C.
接着对底物5a的反应压力进行了测试。结果显示:压力与温度对反应选择性的影响正好相反。增加反应压力不仅可以明显加快反应速度,而且具有显著增加化合物9a比例的趋势。在室温和60psi时的反应已经显现出高度的化学选择性,几乎定量地给出单一的产物9a。Then the reaction pressure of substrate 5a was tested. The results showed that the effects of pressure and temperature on the reaction selectivity were opposite. Increasing the reaction pressure can not only significantly speed up the reaction rate, but also has a tendency to significantly increase the proportion of compound 9a. The reaction at room temperature and 60 psi already showed a high degree of chemoselectivity, giving almost quantitatively the single product 9a.
对较短链长的烷基底物(5a-d,表4)来讲,较低的反应温度有利于反应的选择性,首选温度是室温。对较长链长的烷基底物来讲,实验室制备以室温最好。但是从节省时间的角度考量,可以在室温以上任何温度进行,当反应温度超过甲醇沸点时可以更换高沸点溶剂。为增加较短链长底物(5a-d,表4)的化学选择性,必须使用一定的氢压,80psi以内的氢压就可以达到非常满意的效果。较长链长的底物在常压下就具有非常满意的选择性,但是选择较高的压力可以节省时间。For alkyl substrates with shorter chain lengths (5a-d, Table 4), lower reaction temperatures favor the selectivity of the reaction, with room temperature being the preferred temperature. For alkyl substrates with longer chain lengths, room temperature is best for laboratory preparation. However, from the perspective of saving time, it can be carried out at any temperature above room temperature, and the high boiling point solvent can be replaced when the reaction temperature exceeds the boiling point of methanol. In order to increase the chemoselectivity of shorter chain length substrates (5a-d, Table 4), a certain hydrogen pressure must be used, and a hydrogen pressure within 80 psi can achieve very satisfactory results. Substrates with longer chain lengths have very satisfactory selectivity at normal pressure, but choosing a higher pressure can save time.
按照如下实验条件:以ClCH2CHCl2或CH2Cl2作为偕二氯烷烃,MeOH为溶剂,在15-80psi氢压下,烷基吡啶基酮底物分子(5)均可以顺利地被转化成为(α-烷基)哌啶基甲醇盐酸盐(9)。如表5所示,所有的氢化还原反应均以高度的化学选择性和几乎定量的产率生成相应的产物。According to the following experimental conditions: using ClCH 2 CHCl 2 or CH 2 Cl 2 as gem-dichloroalkane, MeOH as solvent, and under 15-80 psi hydrogen pressure, the alkyl pyridyl ketone substrate molecule (5) can be successfully converted It becomes (α-alkyl)piperidinylmethanol hydrochloride (9). As shown in Table 5, all hydrogenation reduction reactions lead to the corresponding products in highly chemoselective and almost quantitative yields.
表5.烷基吡啶基酮(5)的催化氢化还原Table 5. Catalytic hydrogenation reduction of alkylpyridyl ketones (5)
a.CH2Cl2被用做氯源,所有反应在常温下进行。a. CH 2 Cl 2 was used as chlorine source, and all reactions were carried out at room temperature.
实施例10、4-哌啶甲醇盐酸盐(9a)的制备The preparation of embodiment 10, 4-piperidinemethanol hydrochloride (9a)
将4-吡啶基甲醛(5a,214mg,2.0mmol),10%Pd-C(163mg,50%w/w),二氯甲烷(204mg,2.4mmol)的甲醇(30mL)混合物,在25℃和40psi的氢压下搅拌6.5h后,氢气吸收完全停止。过滤出Pd-C,蒸去滤液中的甲醇,生成的淡黄色固体用无水乙醚洗涤后,经CH3OH-Et2O重结晶得到4-哌啶甲醇盐酸盐(9a)的白色晶体(297mg,98%);4-pyridyl formaldehyde (5a, 214 mg, 2.0 mmol), 10% Pd-C (163 mg, 50% w/w), dichloromethane (204 mg, 2.4 mmol) in methanol (30 mL) mixture, at 25 ° C and After stirring for 6.5 h under a hydrogen pressure of 40 psi, hydrogen uptake completely ceased. Pd-C was filtered off, methanol in the filtrate was distilled off, and the resulting pale yellow solid was washed with anhydrous ether, then recrystallized from CH 3 OH-Et 2 O to obtain white crystals of 4-piperidinemethanol hydrochloride (9a) (297mg, 98%);
mp 128-130℃(MeOH-Et2O);mp 128-130°C (MeOH- Et2O );
v3261,2937,2888,2821,2731,2509,1617,1102,1016cm-1; v3261, 2937, 2888, 2821, 2731, 2509, 1617, 1102, 1016cm -1 ;
13C NMR:δ65.5,43.8,35.4,25.1; 13 C NMR: δ65.5, 43.8, 35.4, 25.1;
Calcd for C6H14ClNO:C,47.52%;H,9.31%;N,9.24%;Found:C,47.17%;H,9.41%;N,9.11%.Calcd for C 6 H 14 ClNO: C, 47.52%; H, 9.31%; N, 9.24%; Found: C, 47.17%; H, 9.41%; N, 9.11%.
实施例11、α-甲基-4-哌啶甲醇盐酸盐(9b)的制备Embodiment 11, the preparation of α-methyl-4-piperidinemethanol hydrochloride (9b)
按照实施例10相同的实验步骤,以4-乙酰基吡啶(5b)为原料,1,1,2-三氯乙烷为偕二氯烷烃,反应得到α-甲基-4-哌啶甲醇盐酸盐(9b);According to the same experimental procedure as in Example 10, using 4-acetylpyridine (5b) as a raw material, and 1,1,2-trichloroethane as a geminal dichloroalkane, the reaction gives α-methyl-4-piperidine methoxide acid salt (9b);
mp 108-110℃(MeOH-Et2O);mp 108-110°C (MeOH- Et2O );
IR:v3404,2968,2812,2722,2496,1602,1453,1057cm-1;IR: v3404, 2968, 2812, 2722, 2496, 1602, 1453, 1057cm -1 ;
13C NMR:δ70.4,44.1,44.0,40.2,28.2,24.6,24.3,19.1; 13 C NMR: δ70.4, 44.1, 44.0, 40.2, 28.2, 24.6, 24.3, 19.1;
Calcd for C7H16NOCl:C,50.75%;H,9.73%;N,8.46%;Found:C,50.66%;H,9.85%;N,8.44%.Calcd for C 7 H 16 NOCl: C, 50.75%; H, 9.73%; N, 8.46%; Found: C, 50.66%; H, 9.85%; N, 8.44%.
实施例12、α-乙基-4-哌啶甲醇盐酸盐(9c)的制备Example 12, the preparation of α-ethyl-4-piperidinemethanol hydrochloride (9c)
按照实施例10相同的实验步骤,以4-丙酰基吡啶(5c)为原料,1,1,2-三氯乙烷为偕二氯烷烃,反应得到α-乙基-4-哌啶甲醇盐酸盐(9c);According to the same experimental procedure as in Example 10, using 4-propionylpyridine (5c) as a raw material, and 1,1,2-trichloroethane as a geminal dichloroalkane, the reaction gives α-ethyl-4-piperidine methoxide acid salt (9c);
mp 106-108℃(MeOH-Et2O);mp 106-108°C (MeOH- Et2O );
v 3394,3068,2959,2936,2819,2715,1578,1378,994cm-1; v 3394, 3068, 2959, 2936, 2819, 2715, 1578, 1378, 994cm-1 ;
13Cδ75.6,44.2,44.1,38.3,25.8,25.1,23.8,9.4; 13C δ75.6, 44.2, 44.1, 38.3, 25.8, 25.1, 23.8, 9.4;
Calcd for C8H18ClNO:C,53.47%;H,10.10%;N,7.80%;Found:C,53.52%;H,10.14%;N,7.78%.Calcd for C 8 H 18 ClNO: C, 53.47%; H, 10.10%; N, 7.80%; Found: C, 53.52%; H, 10.14%; N, 7.78%.
实施例13、α-正丁基-4-哌啶甲醇盐酸盐(9d)的制备Example 13, Preparation of α-n-butyl-4-piperidinemethanol hydrochloride (9d)
将4-戊酰基吡啶(5d,326mg,2.0mmol),10%Pd-C(163mg,50%w/w),1,1,2-三氯乙烷(320mg,2.4mmol)的甲醇(30mL)混合物,在25℃和40psi的氢压下搅拌6.5h后,氢气吸收完全停止。过滤出Pd-C,蒸去滤液中的甲醇,生成的淡黄色固体用无水乙醚洗涤后,经CH3OH-Et2O重结晶得到α-正丁基-4-哌啶甲醇盐酸盐(9d)的白色晶体(415mg,100%);4-pentanoylpyridine (5d, 326 mg, 2.0 mmol), 10% Pd-C (163 mg, 50% w/w), 1,1,2-trichloroethane (320 mg, 2.4 mmol) in methanol (30 mL ) mixture, after stirring for 6.5 h at 25° C. under a hydrogen pressure of 40 psi, hydrogen uptake completely ceased. Filter out Pd-C, evaporate the methanol in the filtrate, wash the resulting light yellow solid with anhydrous ether, and recrystallize from CH 3 OH-Et 2 O to obtain α-n-butyl-4-piperidinemethanol hydrochloride White crystals of (9d) (415 mg, 100%);
mp 112-114℃(MeOH-Et2O);mp 112-114°C (MeOH- Et2O );
IR:v 3392,2954,2928,2860,2818,2716,1579,1449,1380,994cm-1;IR: v 3392, 2954, 2928, 2860, 2818, 2716, 1579, 1449, 1380, 994cm -1 ;
13C NMR:δ74.1,44.3,44.2,38.9,32.8,27.5,25.2,23.8,22.4,138; 13 C NMR: δ74.1, 44.3, 44.2, 38.9, 32.8, 27.5, 25.2, 23.8, 22.4, 138;
Calcd for C10H22ClNO:C,57.82%;H,10.67%;N,6.74%;Found:C,57.75%;H,10.79%;N,6.72%.Calcd for C 10 H 22 ClNO: C, 57.82%; H, 10.67%; N, 6.74%; Found: C, 57.75%; H, 10.79%; N, 6.72%.
实施例14、α-戊基-4-哌啶甲醇盐酸盐(9e)的制备Example 14, Preparation of α-pentyl-4-piperidinemethanol hydrochloride (9e)
按照实施例13相同的实验步骤,以4-己酰基吡啶(5e)为原料,反应得到α-戊基-4-哌啶甲醇盐酸盐(9e);According to the same experimental procedure as in Example 13, using 4-hexanoylpyridine (5e) as a raw material, the reaction gave α-pentyl-4-piperidinemethanol hydrochloride (9e);
mp 108-110℃(MeOH-Et2O);mp 108-110°C (MeOH- Et2O );
IR:v3393,3070,2951,2924,2859,2819,2716,1580,1466,1448,1379cm-1;IR: v3393, 3070, 2951, 2924, 2859, 2819, 2716, 1580, 1466, 1448, 1379cm -1 ;
13C NMR:δ74.0,44.2,44.1,38.9,33.1,31.4,25.2,24.9,23.7,22.3,13.7; 13 C NMR: δ74.0, 44.2, 44.1, 38.9, 33.1, 31.4, 25.2, 24.9, 23.7, 22.3, 13.7;
Calcd for C11H24ClNO:C,59.57%;H,10.91%;N,6.32%;Found:C,59.68%;H,10.79%;N,6.47%.Calcd for C 11 H 24 ClNO: C, 59.57%; H, 10.91%; N, 6.32%; Found: C, 59.68%; H, 10.79%; N, 6.47%.
实施例15、α-己基-4-哌啶甲醇盐酸盐(9f)的制备Example 15, Preparation of α-hexyl-4-piperidinemethanol hydrochloride (9f)
按照实施例13相同的实验步骤,以4-庚酰基吡啶(5f)为原料,反应得到α-己基-4-哌啶甲醇盐酸盐(9f);According to the same experimental procedure as in Example 13, 4-heptanoylpyridine (5f) was used as a raw material to react to obtain α-hexyl-4-piperidinemethanol hydrochloride (9f);
mp 98-100℃(MeOH-Et2O);mp 98-100°C (MeOH- Et2O );
IR:v3394,3370,3064,2955,2925,2715,1133cm-1;IR: v3394, 3370, 3064, 2955, 2925, 2715, 1133cm -1 ;
13C NMR:δ74.0,44.2,38.9,33.1,31.4,28.8,25.2,23.7,22.3,13.7; 13 C NMR: δ74.0, 44.2, 38.9, 33.1, 31.4, 28.8, 25.2, 23.7, 22.3, 13.7;
Calcd for C12H26ClNO:C,61.12;H,11.11;N,5.94;Found:C,61.28;H,11.07;N,5.87.Calcd for C 12 H 26 ClNO: C, 61.12; H, 11.11; N, 5.94; Found: C, 61.28; H, 11.07; N, 5.87.
实施例16、α-辛基-4-哌啶甲醇盐酸盐(9g)的制备Embodiment 16, the preparation of α-octyl-4-piperidinemethanol hydrochloride (9g)
按照实施例13相同的实验步骤,以4-壬酰基吡啶(5g)为原料,反应得到α-辛基-4-哌啶甲醇盐酸盐(9g);According to the same experimental procedure as in Example 13, 4-nonanoylpyridine (5 g) was used as a raw material to react to obtain α-octyl-4-piperidinemethanol hydrochloride (9 g);
mp 107-108℃(MeOH-Et2O);mp 107-108°C (MeOH- Et2O );
IR:v3392,3366,3067,2955,2923,2716,1108cm-1;IR: v3392, 3366, 3067, 2955, 2923, 2716, 1108cm-1 ;
13C NMR:δ73.9,44.1,44.0,33.6,31.9,29.7,29.4,25.8,25.4,23.5,22.6,13.9. 13 C NMR: δ73.9, 44.1, 44.0, 33.6, 31.9, 29.7, 29.4, 25.8, 25.4, 23.5, 22.6, 13.9.
Calcd for C14H30ClNO:C,63.73%;H,11.46%;N,5.31%;Found:C,63.47%;H,11.32%;N,5.29%.Calcd for C 14 H 30 ClNO: C, 63.73%; H, 11.46%; N, 5.31%; Found: C, 63.47%; H, 11.32%; N, 5.29%.
实施例17、α-(2-苯基乙基)-4-哌啶甲醇盐酸盐(9h)的制备Example 17, Preparation of α-(2-phenylethyl)-4-piperidinemethanol hydrochloride (9h)
按照实施例13相同的实验步骤,以4-(3-苯基丙酰基)-吡啶(5h)为原料,反应得到α-(2-苯基乙基)-4-哌啶甲醇盐酸盐(9h);According to the same experimental procedure as in Example 13, 4-(3-phenylpropionyl)-pyridine (5h) was used as raw material to react to obtain α-(2-phenylethyl)-4-piperidinemethanol hydrochloride ( 9h);
mp 170-171℃(MeOH-Et2O);mp 170-171°C (MeOH- Et2O );
IR:v3628,3382,2947,2825,2802,2725,1578,1495,1450cm-1;IR: v3628, 3382, 2947, 2825, 2802, 2725, 1578, 1495, 1450cm-1 ;
13C NMR:δ142.5,128.7,126.0,73.2,44.1,44.0,39.0,35.3,31.7,25.2,23.5; 13 C NMR: δ142.5, 128.7, 126.0, 73.2, 44.1, 44.0, 39.0, 35.3, 31.7, 25.2, 23.5;
Calcd for C14H22ClNO:C,65.74%;H,8.67%;N,5.48%;Found:C,65.68%;H,8.44%;N,5.50%.Calcd for C 14 H 22 ClNO: C, 65.74%; H, 8.67%; N, 5.48%; Found: C, 65.68%; H, 8.44%; N, 5.50%.
实施例18、α-丁基-2-哌啶甲醇盐酸盐(9i)的制备Example 18, Preparation of α-butyl-2-piperidinemethanol hydrochloride (9i)
按照实施例13相同的实验步骤,以2-(戊酰基)吡啶(5i)为原料,反应得到α-丁基-2-哌啶甲醇盐酸盐(9i);According to the same experimental procedure as in Example 13, using 2-(valeryl)pyridine (5i) as a raw material, the reaction gave α-butyl-2-piperidinemethanol hydrochloride (9i);
mp 132-134℃(MeOH-Et2O);mp 132-134°C (MeOH- Et2O );
IR:v3423,2956,2869,1486,1470,1447,1080cm-1;IR: v3423, 2956, 2869, 1486, 1470, 1447, 1080cm-1 ;
13C NMR:δ71.4,70.7,61.3,60.6,45.1,44.7,32.0,31.4,27.4,26.7,25.3,22.1,21.9,21.6,21.4,13.4; 13 C NMR: δ71.4, 70.7, 61.3, 60.6, 45.1, 44.7, 32.0, 31.4, 27.4, 26.7, 25.3, 22.1, 21.9, 21.6, 21.4, 13.4;
Calcd for C10H22ClNO:C,57.82%;H,10.67%;N,6.74%;Found:C,57.94%;H,10.48%;N,6.87%.Calcd for C 10 H 22 ClNO: C, 57.82%; H, 10.67%; N, 6.74%; Found: C, 57.94%; H, 10.48%; N, 6.87%.
Claims (11)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CNB2006101443591A CN100448846C (en) | 2006-12-04 | 2006-12-04 | A kind of catalytic hydrogenation acylpyridine is the method for substituted piperidine compound hydrochloride |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CNB2006101443591A CN100448846C (en) | 2006-12-04 | 2006-12-04 | A kind of catalytic hydrogenation acylpyridine is the method for substituted piperidine compound hydrochloride |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN1995017A CN1995017A (en) | 2007-07-11 |
| CN100448846C true CN100448846C (en) | 2009-01-07 |
Family
ID=38250237
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CNB2006101443591A Expired - Fee Related CN100448846C (en) | 2006-12-04 | 2006-12-04 | A kind of catalytic hydrogenation acylpyridine is the method for substituted piperidine compound hydrochloride |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN100448846C (en) |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4690931A (en) * | 1982-10-13 | 1987-09-01 | Synthelabo | Therapeutically useful 1-phenyl-2-piperidinoalkanol derivatives |
-
2006
- 2006-12-04 CN CNB2006101443591A patent/CN100448846C/en not_active Expired - Fee Related
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4690931A (en) * | 1982-10-13 | 1987-09-01 | Synthelabo | Therapeutically useful 1-phenyl-2-piperidinoalkanol derivatives |
Non-Patent Citations (2)
| Title |
|---|
| 哌啶催化合成工艺研究. 陈声宗.化学反应工程与工艺,第16卷第4期. 2002 |
| 哌啶催化合成工艺研究. 陈声宗.化学反应工程与工艺,第16卷第4期. 2002 * |
Also Published As
| Publication number | Publication date |
|---|---|
| CN1995017A (en) | 2007-07-11 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Liu et al. | N-Methylation of Amines with Methanol in the Presence of Carbonate Salt Catalyzed by a Metal–Ligand Bifunctional Ruthenium Catalyst [(p-cymene) Ru (2, 2′-bpyO)(H2O)] | |
| Lator et al. | Well-defined phosphine-free iron-catalyzed N-ethylation and N-methylation of amines with ethanol and methanol | |
| Brown et al. | Use of (Cyclopentadienone) iron tricarbonyl complexes for C–N bond formation reactions between amines and alcohols | |
| Liu et al. | Use of a cyclometalated iridium (III) complex containing a N∧ C∧ N-coordinating terdentate ligand as a catalyst for the α-alkylation of ketones and N-alkylation of amines with alcohols | |
| Malkov et al. | Asymmetric reduction of imines with trichlorosilane, catalyzed by sigamide, an amino acid-derived formamide: Scope and limitations | |
| Schley et al. | Oxidative synthesis of amides and pyrroles via dehydrogenative alcohol oxidation by ruthenium diphosphine diamine complexes | |
| Mo et al. | Rhodium (III)-Catalyzed C–H Activation/Alkyne Annulation by Weak Coordination of Peresters with O–O Bond as an Internal Oxidant | |
| JP5719115B2 (en) | Novel organometallic complex and method for producing amine compound | |
| Shen et al. | Merging visible-light photoredox and chiral phosphate catalysis for asymmetric Friedel–Crafts reaction with in situ generation of N-acyl imines | |
| Tan et al. | Catalytic conversion of N-heteroaromatics to functionalized arylamines by merging hydrogen transfer and selective coupling | |
| JP5216065B2 (en) | Pyridyltetrahydropyridines and pyridylpiperidines and methods for producing them | |
| Wei et al. | N-alkylation of amines with alcohols catalyzed by manganese (II) chloride or bromopentacarbonylmanganese (I) | |
| JP5847386B2 (en) | Method for producing amine compound | |
| Luo et al. | Autotandem catalyst: from acylhydrazones to N′, N′-methylaliphatic acylhydrazides via transfer hydrogenation/N-methylation with methanol catalyzed by a Cp* iridium complex bearing a functional ligand | |
| Luo et al. | pH-Mediated selective synthesis of N-allylic alkylation or N-alkylation amines with allylic alcohols via an Iridium catalyst in water | |
| Dethe et al. | Cp* Co (III)-Catalyzed Ketone-Directed ortho-C–H Activation for the Synthesis of Indene Derivatives | |
| Molnar et al. | Ruthenium-catalyzed C–H activation and coupling reactions in organic synthesis | |
| Sindhe et al. | Pyridine C (sp2)–H bond functionalization under transition-metal and rare earth metal catalysis | |
| Wu et al. | Copper-catalyzed sulfonylation/cyclization of pent-4-ynamides toward sulfonyl-functionalized pyrrol-2-ones | |
| CN110452270A (en) | Azepine Cabbeen palladium complex crystal and its synthetic method and preparing the application in alpha, beta-unsaturated ketone compound | |
| CN106458943A (en) | Synthesis of vortioxetine via (2,4-dimethylphenyl)(2-iodophenyl)sulfane intermediate | |
| CN100448846C (en) | A kind of catalytic hydrogenation acylpyridine is the method for substituted piperidine compound hydrochloride | |
| JPS5910337B2 (en) | Method for producing chlorinated phenylhydroxylamines | |
| NZ504338A (en) | Substituted pyridine or piperidine compounds and pharmaceutical compositions | |
| KR100707050B1 (en) | Method for preparing 10H-dibenzo [VII, VIII] [1,4] thiazepin-11-one |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| C17 | Cessation of patent right | ||
| CF01 | Termination of patent right due to non-payment of annual fee |
Granted publication date: 20090107 Termination date: 20101204 |