CN100435847C - Sustained release formulation containing poorly soluble principal drug - Google Patents
Sustained release formulation containing poorly soluble principal drug Download PDFInfo
- Publication number
- CN100435847C CN100435847C CNB200410043476XA CN200410043476A CN100435847C CN 100435847 C CN100435847 C CN 100435847C CN B200410043476X A CNB200410043476X A CN B200410043476XA CN 200410043476 A CN200410043476 A CN 200410043476A CN 100435847 C CN100435847 C CN 100435847C
- Authority
- CN
- China
- Prior art keywords
- acid
- alginate
- pharmaceutical compositions
- clarithromycin
- compositions according
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
- 239000000203 mixture Substances 0.000 title claims description 23
- 239000003814 drug Substances 0.000 title abstract description 25
- 229940079593 drug Drugs 0.000 title abstract description 22
- 238000013268 sustained release Methods 0.000 title abstract description 11
- 239000012730 sustained-release form Substances 0.000 title abstract description 11
- 238000009472 formulation Methods 0.000 title description 13
- 229960002626 clarithromycin Drugs 0.000 claims abstract description 36
- AGOYDEPGAOXOCK-KCBOHYOISA-N clarithromycin Chemical compound O([C@@H]1[C@@H](C)C(=O)O[C@@H]([C@@]([C@H](O)[C@@H](C)C(=O)[C@H](C)C[C@](C)([C@H](O[C@H]2[C@@H]([C@H](C[C@@H](C)O2)N(C)C)O)[C@H]1C)OC)(C)O)CC)[C@H]1C[C@@](C)(OC)[C@@H](O)[C@H](C)O1 AGOYDEPGAOXOCK-KCBOHYOISA-N 0.000 claims abstract description 36
- 235000010443 alginic acid Nutrition 0.000 claims abstract description 28
- 229920000615 alginic acid Polymers 0.000 claims abstract description 28
- FHVDTGUDJYJELY-UHFFFAOYSA-N 6-{[2-carboxy-4,5-dihydroxy-6-(phosphanyloxy)oxan-3-yl]oxy}-4,5-dihydroxy-3-phosphanyloxane-2-carboxylic acid Chemical compound O1C(C(O)=O)C(P)C(O)C(O)C1OC1C(C(O)=O)OC(OP)C(O)C1O FHVDTGUDJYJELY-UHFFFAOYSA-N 0.000 claims abstract description 20
- 229940072056 alginate Drugs 0.000 claims abstract description 19
- 239000008194 pharmaceutical composition Substances 0.000 claims abstract description 18
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 claims description 36
- IXPNQXFRVYWDDI-UHFFFAOYSA-N 1-methyl-2,4-dioxo-1,3-diazinane-5-carboximidamide Chemical compound CN1CC(C(N)=N)C(=O)NC1=O IXPNQXFRVYWDDI-UHFFFAOYSA-N 0.000 claims description 24
- 239000000661 sodium alginate Substances 0.000 claims description 24
- 235000010413 sodium alginate Nutrition 0.000 claims description 24
- 229940005550 sodium alginate Drugs 0.000 claims description 24
- 239000007937 lozenge Substances 0.000 claims description 19
- 239000002552 dosage form Substances 0.000 claims description 11
- 239000002253 acid Substances 0.000 claims description 9
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 9
- 239000007787 solid Substances 0.000 claims description 4
- JFCQEDHGNNZCLN-UHFFFAOYSA-N glutaric acid Chemical compound OC(=O)CCCC(O)=O JFCQEDHGNNZCLN-UHFFFAOYSA-N 0.000 claims description 3
- 239000003120 macrolide antibiotic agent Substances 0.000 claims description 3
- QBYIENPQHBMVBV-HFEGYEGKSA-N (2R)-2-hydroxy-2-phenylacetic acid Chemical compound O[C@@H](C(O)=O)c1ccccc1.O[C@@H](C(O)=O)c1ccccc1 QBYIENPQHBMVBV-HFEGYEGKSA-N 0.000 claims description 2
- BJEPYKJPYRNKOW-REOHCLBHSA-N (S)-malic acid Chemical compound OC(=O)[C@@H](O)CC(O)=O BJEPYKJPYRNKOW-REOHCLBHSA-N 0.000 claims description 2
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 claims description 2
- OFOBLEOULBTSOW-UHFFFAOYSA-N Propanedioic acid Natural products OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 claims description 2
- IWYDHOAUDWTVEP-UHFFFAOYSA-N R-2-phenyl-2-hydroxyacetic acid Natural products OC(=O)C(O)C1=CC=CC=C1 IWYDHOAUDWTVEP-UHFFFAOYSA-N 0.000 claims description 2
- KDYFGRWQOYBRFD-UHFFFAOYSA-N Succinic acid Natural products OC(=O)CCC(O)=O KDYFGRWQOYBRFD-UHFFFAOYSA-N 0.000 claims description 2
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric acid Natural products [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 claims description 2
- BJEPYKJPYRNKOW-UHFFFAOYSA-N alpha-hydroxysuccinic acid Natural products OC(=O)C(O)CC(O)=O BJEPYKJPYRNKOW-UHFFFAOYSA-N 0.000 claims description 2
- KDYFGRWQOYBRFD-NUQCWPJISA-N butanedioic acid Chemical compound O[14C](=O)CC[14C](O)=O KDYFGRWQOYBRFD-NUQCWPJISA-N 0.000 claims description 2
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 claims description 2
- 239000011976 maleic acid Substances 0.000 claims description 2
- 239000001630 malic acid Substances 0.000 claims description 2
- 235000011090 malic acid Nutrition 0.000 claims description 2
- 229960002510 mandelic acid Drugs 0.000 claims description 2
- 239000011975 tartaric acid Substances 0.000 claims description 2
- 235000002906 tartaric acid Nutrition 0.000 claims description 2
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 claims description 2
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 claims 5
- 239000003795 chemical substances by application Substances 0.000 claims 5
- ZDXPYRJPNDTMRX-VKHMYHEASA-N L-glutamine Chemical compound OC(=O)[C@@H](N)CCC(N)=O ZDXPYRJPNDTMRX-VKHMYHEASA-N 0.000 claims 1
- 230000003203 everyday effect Effects 0.000 claims 1
- ZDXPYRJPNDTMRX-UHFFFAOYSA-N glutamine Natural products OC(=O)C(N)CCC(N)=O ZDXPYRJPNDTMRX-UHFFFAOYSA-N 0.000 claims 1
- 230000002459 sustained effect Effects 0.000 claims 1
- ULGZDMOVFRHVEP-RWJQBGPGSA-N Erythromycin Chemical compound O([C@@H]1[C@@H](C)C(=O)O[C@@H]([C@@]([C@H](O)[C@@H](C)C(=O)[C@H](C)C[C@@](C)(O)[C@H](O[C@H]2[C@@H]([C@H](C[C@@H](C)O2)N(C)C)O)[C@H]1C)(C)O)CC)[C@H]1C[C@@](C)(OC)[C@@H](O)[C@H](C)O1 ULGZDMOVFRHVEP-RWJQBGPGSA-N 0.000 abstract description 20
- 238000013270 controlled release Methods 0.000 abstract description 18
- 229960003276 erythromycin Drugs 0.000 abstract description 7
- 150000001732 carboxylic acid derivatives Chemical class 0.000 abstract description 5
- 238000002560 therapeutic procedure Methods 0.000 abstract description 3
- 239000008203 oral pharmaceutical composition Substances 0.000 abstract description 2
- 238000004090 dissolution Methods 0.000 description 18
- 239000003826 tablet Substances 0.000 description 16
- 239000000243 solution Substances 0.000 description 11
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 8
- 239000011575 calcium Substances 0.000 description 8
- 229910052791 calcium Inorganic materials 0.000 description 8
- 239000011734 sodium Substances 0.000 description 8
- 229910052708 sodium Inorganic materials 0.000 description 8
- 238000012360 testing method Methods 0.000 description 8
- 239000007939 sustained release tablet Substances 0.000 description 7
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 6
- 230000000694 effects Effects 0.000 description 6
- 239000012086 standard solution Substances 0.000 description 6
- 238000004587 chromatography analysis Methods 0.000 description 5
- 230000002496 gastric effect Effects 0.000 description 5
- -1 sodium alginate Chemical compound 0.000 description 5
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 4
- VEUACKUBDLVUAC-UHFFFAOYSA-N [Na].[Ca] Chemical compound [Na].[Ca] VEUACKUBDLVUAC-UHFFFAOYSA-N 0.000 description 4
- 229960001126 alginic acid Drugs 0.000 description 4
- 239000000783 alginic acid Substances 0.000 description 4
- 150000004781 alginic acids Chemical class 0.000 description 4
- 239000000648 calcium alginate Substances 0.000 description 4
- 229960002681 calcium alginate Drugs 0.000 description 4
- 210000004051 gastric juice Anatomy 0.000 description 4
- 239000008187 granular material Substances 0.000 description 4
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 4
- 238000002360 preparation method Methods 0.000 description 4
- 239000012085 test solution Substances 0.000 description 4
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 3
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 3
- 150000001768 cations Chemical class 0.000 description 3
- AMTWCFIAVKBGOD-UHFFFAOYSA-N dioxosilane;methoxy-dimethyl-trimethylsilyloxysilane Chemical compound O=[Si]=O.CO[Si](C)(C)O[Si](C)(C)C AMTWCFIAVKBGOD-UHFFFAOYSA-N 0.000 description 3
- 210000001035 gastrointestinal tract Anatomy 0.000 description 3
- 239000000499 gel Substances 0.000 description 3
- 239000001866 hydroxypropyl methyl cellulose Substances 0.000 description 3
- 235000010979 hydroxypropyl methyl cellulose Nutrition 0.000 description 3
- 229920003088 hydroxypropyl methyl cellulose Polymers 0.000 description 3
- UFVKGYZPFZQRLF-UHFFFAOYSA-N hydroxypropyl methyl cellulose Chemical compound OC1C(O)C(OC)OC(CO)C1OC1C(O)C(O)C(OC2C(C(O)C(OC3C(C(O)C(O)C(CO)O3)O)C(CO)O2)O)C(CO)O1 UFVKGYZPFZQRLF-UHFFFAOYSA-N 0.000 description 3
- 238000004519 manufacturing process Methods 0.000 description 3
- 229940083037 simethicone Drugs 0.000 description 3
- 239000007974 sodium acetate buffer Substances 0.000 description 3
- 239000002904 solvent Substances 0.000 description 3
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 2
- 208000007882 Gastritis Diseases 0.000 description 2
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 2
- WHNWPMSKXPGLAX-UHFFFAOYSA-N N-Vinyl-2-pyrrolidone Chemical compound C=CN1CCCC1=O WHNWPMSKXPGLAX-UHFFFAOYSA-N 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 2
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 2
- 235000021355 Stearic acid Nutrition 0.000 description 2
- 206010000059 abdominal discomfort Diseases 0.000 description 2
- 239000004480 active ingredient Substances 0.000 description 2
- 230000000844 anti-bacterial effect Effects 0.000 description 2
- 238000009792 diffusion process Methods 0.000 description 2
- 238000010494 dissociation reaction Methods 0.000 description 2
- 230000005593 dissociations Effects 0.000 description 2
- 208000021302 gastroesophageal reflux disease Diseases 0.000 description 2
- 239000008101 lactose Substances 0.000 description 2
- 239000000314 lubricant Substances 0.000 description 2
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 2
- 239000002075 main ingredient Substances 0.000 description 2
- 239000012528 membrane Substances 0.000 description 2
- 238000000034 method Methods 0.000 description 2
- 229910000402 monopotassium phosphate Inorganic materials 0.000 description 2
- 235000019796 monopotassium phosphate Nutrition 0.000 description 2
- OQCDKBAXFALNLD-UHFFFAOYSA-N octadecanoic acid Natural products CCCCCCCC(C)CCCCCCCCC(O)=O OQCDKBAXFALNLD-UHFFFAOYSA-N 0.000 description 2
- 150000007524 organic acids Chemical class 0.000 description 2
- PJNZPQUBCPKICU-UHFFFAOYSA-N phosphoric acid;potassium Chemical compound [K].OP(O)(O)=O PJNZPQUBCPKICU-UHFFFAOYSA-N 0.000 description 2
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 2
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 2
- 239000011148 porous material Substances 0.000 description 2
- 229940069328 povidone Drugs 0.000 description 2
- 239000003755 preservative agent Substances 0.000 description 2
- 239000004172 quinoline yellow Substances 0.000 description 2
- 235000012752 quinoline yellow Nutrition 0.000 description 2
- 238000011160 research Methods 0.000 description 2
- 239000008117 stearic acid Substances 0.000 description 2
- 239000004408 titanium dioxide Substances 0.000 description 2
- METKIMKYRPQLGS-GFCCVEGCSA-N (R)-atenolol Chemical compound CC(C)NC[C@@H](O)COC1=CC=C(CC(N)=O)C=C1 METKIMKYRPQLGS-GFCCVEGCSA-N 0.000 description 1
- RTBFRGCFXZNCOE-UHFFFAOYSA-N 1-methylsulfonylpiperidin-4-one Chemical compound CS(=O)(=O)N1CCC(=O)CC1 RTBFRGCFXZNCOE-UHFFFAOYSA-N 0.000 description 1
- BDKLKNJTMLIAFE-UHFFFAOYSA-N 2-(3-fluorophenyl)-1,3-oxazole-4-carbaldehyde Chemical compound FC1=CC=CC(C=2OC=C(C=O)N=2)=C1 BDKLKNJTMLIAFE-UHFFFAOYSA-N 0.000 description 1
- SUBDBMMJDZJVOS-UHFFFAOYSA-N 5-methoxy-2-{[(4-methoxy-3,5-dimethylpyridin-2-yl)methyl]sulfinyl}-1H-benzimidazole Chemical compound N=1C2=CC(OC)=CC=C2NC=1S(=O)CC1=NC=C(C)C(OC)=C1C SUBDBMMJDZJVOS-UHFFFAOYSA-N 0.000 description 1
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 1
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 1
- DKMROQRQHGEIOW-UHFFFAOYSA-N Diethyl succinate Chemical compound CCOC(=O)CCC(=O)OCC DKMROQRQHGEIOW-UHFFFAOYSA-N 0.000 description 1
- 208000003870 Drug Overdose Diseases 0.000 description 1
- 206010013911 Dysgeusia Diseases 0.000 description 1
- 241000194033 Enterococcus Species 0.000 description 1
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 1
- WHUUTDBJXJRKMK-UHFFFAOYSA-N Glutamic acid Natural products OC(=O)C(N)CCC(O)=O WHUUTDBJXJRKMK-UHFFFAOYSA-N 0.000 description 1
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 description 1
- 206010028813 Nausea Diseases 0.000 description 1
- 206010033296 Overdoses Diseases 0.000 description 1
- 229930182555 Penicillin Natural products 0.000 description 1
- JGSARLDLIJGVTE-MBNYWOFBSA-N Penicillin G Chemical compound N([C@H]1[C@H]2SC([C@@H](N2C1=O)C(O)=O)(C)C)C(=O)CC1=CC=CC=C1 JGSARLDLIJGVTE-MBNYWOFBSA-N 0.000 description 1
- 239000002202 Polyethylene glycol Substances 0.000 description 1
- XBDQKXXYIPTUBI-UHFFFAOYSA-M Propionate Chemical compound CCC([O-])=O XBDQKXXYIPTUBI-UHFFFAOYSA-M 0.000 description 1
- 241000191940 Staphylococcus Species 0.000 description 1
- 241000194017 Streptococcus Species 0.000 description 1
- 208000025865 Ulcer Diseases 0.000 description 1
- 206010047700 Vomiting Diseases 0.000 description 1
- 239000003929 acidic solution Substances 0.000 description 1
- 125000001931 aliphatic group Chemical group 0.000 description 1
- WNROFYMDJYEPJX-UHFFFAOYSA-K aluminium hydroxide Chemical compound [OH-].[OH-].[OH-].[Al+3] WNROFYMDJYEPJX-UHFFFAOYSA-K 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- 229940069428 antacid Drugs 0.000 description 1
- 239000003159 antacid agent Substances 0.000 description 1
- 239000003242 anti bacterial agent Substances 0.000 description 1
- 230000000767 anti-ulcer Effects 0.000 description 1
- 239000007900 aqueous suspension Substances 0.000 description 1
- 229960002274 atenolol Drugs 0.000 description 1
- 229910052728 basic metal Inorganic materials 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 239000008280 blood Substances 0.000 description 1
- 210000004369 blood Anatomy 0.000 description 1
- 239000007853 buffer solution Substances 0.000 description 1
- FATUQANACHZLRT-KMRXSBRUSA-L calcium glucoheptonate Chemical compound [Ca+2].OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C(O)C([O-])=O.OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C(O)C([O-])=O FATUQANACHZLRT-KMRXSBRUSA-L 0.000 description 1
- 159000000007 calcium salts Chemical class 0.000 description 1
- 239000002775 capsule Substances 0.000 description 1
- 125000004432 carbon atom Chemical group C* 0.000 description 1
- 150000001735 carboxylic acids Chemical class 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 229960001380 cimetidine Drugs 0.000 description 1
- CCGSUNCLSOWKJO-UHFFFAOYSA-N cimetidine Chemical compound N#CNC(=N/C)\NCCSCC1=NC=N[C]1C CCGSUNCLSOWKJO-UHFFFAOYSA-N 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 238000004040 coloring Methods 0.000 description 1
- 150000001875 compounds Chemical class 0.000 description 1
- 239000003431 cross linking reagent Substances 0.000 description 1
- AAOVKJBEBIDNHE-UHFFFAOYSA-N diazepam Chemical compound N=1CC(=O)N(C)C2=CC=C(Cl)C=C2C=1C1=CC=CC=C1 AAOVKJBEBIDNHE-UHFFFAOYSA-N 0.000 description 1
- 229960003529 diazepam Drugs 0.000 description 1
- 238000007865 diluting Methods 0.000 description 1
- 239000003085 diluting agent Substances 0.000 description 1
- 238000010790 dilution Methods 0.000 description 1
- 239000012895 dilution Substances 0.000 description 1
- 125000002147 dimethylamino group Chemical group [H]C([H])([H])N(*)C([H])([H])[H] 0.000 description 1
- 238000007922 dissolution test Methods 0.000 description 1
- 231100000725 drug overdose Toxicity 0.000 description 1
- 235000019564 dysgeusia Nutrition 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- BEFDCLMNVWHSGT-UHFFFAOYSA-N ethenylcyclopentane Chemical compound C=CC1CCCC1 BEFDCLMNVWHSGT-UHFFFAOYSA-N 0.000 description 1
- 239000010408 film Substances 0.000 description 1
- 239000007888 film coating Substances 0.000 description 1
- 238000009501 film coating Methods 0.000 description 1
- 239000000706 filtrate Substances 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 238000007429 general method Methods 0.000 description 1
- 239000004220 glutamic acid Substances 0.000 description 1
- 235000013922 glutamic acid Nutrition 0.000 description 1
- 238000005469 granulation Methods 0.000 description 1
- 230000003179 granulation Effects 0.000 description 1
- 230000002949 hemolytic effect Effects 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 239000012729 immediate-release (IR) formulation Substances 0.000 description 1
- 238000001727 in vivo Methods 0.000 description 1
- 239000005414 inactive ingredient Substances 0.000 description 1
- NDDAHWYSQHTHNT-UHFFFAOYSA-N indapamide Chemical compound CC1CC2=CC=CC=C2N1NC(=O)C1=CC=C(Cl)C(S(N)(=O)=O)=C1 NDDAHWYSQHTHNT-UHFFFAOYSA-N 0.000 description 1
- 229960004569 indapamide Drugs 0.000 description 1
- 239000004615 ingredient Substances 0.000 description 1
- 239000007924 injection Substances 0.000 description 1
- 238000002347 injection Methods 0.000 description 1
- 229940099584 lactobionate Drugs 0.000 description 1
- JYTUSYBCFIZPBE-AMTLMPIISA-N lactobionic acid Chemical compound OC(=O)[C@H](O)[C@@H](O)[C@@H]([C@H](O)CO)O[C@@H]1O[C@H](CO)[C@H](O)[C@H](O)[C@H]1O JYTUSYBCFIZPBE-AMTLMPIISA-N 0.000 description 1
- 229960001375 lactose Drugs 0.000 description 1
- 238000004811 liquid chromatography Methods 0.000 description 1
- VTHJTEIRLNZDEV-UHFFFAOYSA-L magnesium dihydroxide Chemical compound [OH-].[OH-].[Mg+2] VTHJTEIRLNZDEV-UHFFFAOYSA-L 0.000 description 1
- 239000000347 magnesium hydroxide Substances 0.000 description 1
- 229910001862 magnesium hydroxide Inorganic materials 0.000 description 1
- 235000019359 magnesium stearate Nutrition 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 239000011159 matrix material Substances 0.000 description 1
- VAOCPAMSLUNLGC-UHFFFAOYSA-N metronidazole Chemical compound CC1=NC=C([N+]([O-])=O)N1CCO VAOCPAMSLUNLGC-UHFFFAOYSA-N 0.000 description 1
- 229960000282 metronidazole Drugs 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- CQDGTJPVBWZJAZ-UHFFFAOYSA-N monoethyl carbonate Chemical compound CCOC(O)=O CQDGTJPVBWZJAZ-UHFFFAOYSA-N 0.000 description 1
- 230000008693 nausea Effects 0.000 description 1
- 229960000381 omeprazole Drugs 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- 229940049954 penicillin Drugs 0.000 description 1
- 239000000546 pharmaceutical excipient Substances 0.000 description 1
- 239000000049 pigment Substances 0.000 description 1
- 229920001223 polyethylene glycol Polymers 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 229910001414 potassium ion Inorganic materials 0.000 description 1
- 239000000047 product Substances 0.000 description 1
- 235000013772 propylene glycol Nutrition 0.000 description 1
- 239000008213 purified water Substances 0.000 description 1
- 229940051201 quinoline yellow Drugs 0.000 description 1
- IZMJMCDDWKSTTK-UHFFFAOYSA-N quinoline yellow Chemical compound C1=CC=CC2=NC(C3C(C4=CC=CC=C4C3=O)=O)=CC=C21 IZMJMCDDWKSTTK-UHFFFAOYSA-N 0.000 description 1
- VMXUWOKSQNHOCA-LCYFTJDESA-N ranitidine Chemical compound [O-][N+](=O)/C=C(/NC)NCCSCC1=CC=C(CN(C)C)O1 VMXUWOKSQNHOCA-LCYFTJDESA-N 0.000 description 1
- 229960000620 ranitidine Drugs 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 238000005070 sampling Methods 0.000 description 1
- 230000028327 secretion Effects 0.000 description 1
- 239000012748 slip agent Substances 0.000 description 1
- 210000000813 small intestine Anatomy 0.000 description 1
- 235000017281 sodium acetate Nutrition 0.000 description 1
- 229940087562 sodium acetate trihydrate Drugs 0.000 description 1
- 229910000029 sodium carbonate Inorganic materials 0.000 description 1
- 229910001415 sodium ion Inorganic materials 0.000 description 1
- 239000004334 sorbic acid Substances 0.000 description 1
- 229940075582 sorbic acid Drugs 0.000 description 1
- 235000010199 sorbic acid Nutrition 0.000 description 1
- 239000000600 sorbitol Substances 0.000 description 1
- 229960004274 stearic acid Drugs 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 229960004291 sucralfate Drugs 0.000 description 1
- MNQYNQBOVCBZIQ-JQOFMKNESA-A sucralfate Chemical compound O[Al](O)OS(=O)(=O)O[C@@H]1[C@@H](OS(=O)(=O)O[Al](O)O)[C@H](OS(=O)(=O)O[Al](O)O)[C@@H](COS(=O)(=O)O[Al](O)O)O[C@H]1O[C@@]1(COS(=O)(=O)O[Al](O)O)[C@@H](OS(=O)(=O)O[Al](O)O)[C@H](OS(=O)(=O)O[Al](O)O)[C@@H](OS(=O)(=O)O[Al](O)O)O1 MNQYNQBOVCBZIQ-JQOFMKNESA-A 0.000 description 1
- 229960005404 sulfamethoxazole Drugs 0.000 description 1
- JLKIGFTWXXRPMT-UHFFFAOYSA-N sulphamethoxazole Chemical compound O1C(C)=CC(NS(=O)(=O)C=2C=CC(N)=CC=2)=N1 JLKIGFTWXXRPMT-UHFFFAOYSA-N 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 239000007916 tablet composition Substances 0.000 description 1
- 239000000454 talc Substances 0.000 description 1
- 229910052623 talc Inorganic materials 0.000 description 1
- 235000012222 talc Nutrition 0.000 description 1
- 239000010409 thin film Substances 0.000 description 1
- 235000010215 titanium dioxide Nutrition 0.000 description 1
- 231100000397 ulcer Toxicity 0.000 description 1
- 230000008673 vomiting Effects 0.000 description 1
Images
Landscapes
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicinal Preparation (AREA)
Abstract
本发明提供一种适用于每日投服型疗法中之持续释放型口服药学组合物,含有至少一种难溶解性主药(例如红霉素衍生物)。此药学组合物含有主药、一种水溶性藻酸盐及一种有机羧酸。本发明特别提供一种克拉霉素之控制释放型锭剂。The present invention provides a sustained-release oral pharmaceutical composition suitable for daily administration therapy, comprising at least one poorly soluble main drug (e.g., erythromycin derivative). The pharmaceutical composition comprises the main drug, a water-soluble alginate, and an organic carboxylic acid. The present invention particularly provides a controlled-release tablet of clarithromycin.
Description
技术领域 technical field
本发明系关于难溶解性主药,例如红霉素衍生物之口服持续释放配方,其可在胃肠液中被消化。本发明特别系关于可以每日一锭方式投服的克拉霉素(clarithromycin)药学组合物。The present invention relates to an oral sustained-release formulation of a poorly soluble main drug, such as an erythromycin derivative, which can be digested in gastrointestinal fluid. In particular, the present invention relates to a pharmaceutical composition of clarithromycin which can be administered in the form of one tablet per day.
背景技术 Background technique
红霉素和它的衍生物均属抗菌剂,彼等有广泛的抗菌力,和青霉素的抗菌活性有部份相同,特别是对于革兰氏阳性球菌,如肠球菌、溶血性链球菌A群、肺炎球菌及葡萄球菌,为有效的。通常服用的剂量是每天服用2-4次,连续服用10~14天。对于非住院患者而言,每天服用2~4次可能造成不方便,或会忘记服用;因此,最好是降低每日服用的次数。特别是对非住院患者,固态之持续释放配方可降低服用的次数。持续释放配方是为了使药物在血中浓度相对于时间的变化维持在稳定的状态,如此可使每天服用2~4次之患者避免在服用下一锭时有药物过量的情形产生。Erythromycin and its derivatives are antibacterial agents, they have a wide range of antibacterial activity, and some of the same antibacterial activity as penicillin, especially for Gram-positive cocci, such as enterococcus, hemolytic streptococcus group A , pneumococcus and staphylococcus are effective. The usual dosage is to take 2-4 times a day for 10-14 consecutive days. For ambulatory patients, taking 2 to 4 times a day may cause inconvenience, or forget to take; therefore, it is better to reduce the number of daily doses. Especially for non-hospitalized patients, the solid-state sustained-release formulation can reduce the number of doses. The sustained-release formula is to keep the blood concentration of the drug in a stable state relative to time, so that patients who take 2-4 times a day can avoid drug overdose when taking the next tablet.
使用藻酸盐胶来制备控制释放型药锭是已知的。一种水溶性的藻酸盐,例如藻酸钠,可与钙盐反应,如此可使藻酸盐被转变成不溶于水的藻酸钠-钙胶。可通过改变藻酸盐分子量、藻酸盐含量、多价阳离子及交联剂的类型、和(或)阳离子在藻酸盐中之含量度,来改变藻酸盐胶之控制释放性质。美国专利第4,842,866号中即有揭示利用藻酸钠和藻酸钠-钙来制备固态控制释放剂型。The use of alginate gels for the preparation of controlled release tablets is known. A water-soluble alginate, such as sodium alginate, can be reacted with a calcium salt so that the alginate is converted into a water-insoluble sodium alginate-calcium gel. The controlled-release properties of alginate gel can be changed by changing the molecular weight of alginate, the content of alginate, the type of multivalent cations and cross-linking agents, and (or) the content of cations in alginate. US Patent No. 4,842,866 discloses the use of sodium alginate and sodium-calcium alginate to prepare solid controlled release dosage forms.
上述所提及的专利技术并不能应用在含有水溶性很低之主药的配方中,例如,用克拉霉素和藻酸盐作成的配方,其溶离太慢且其在活体动物试验中之生体可利用性并无再现性。对于水溶性很低的药,可利用藻酸盐为基质并添加入有机酸,作成固态的控制释放剂型。美国专利第5,705,190号(其相当于中国台湾专利第429,154号)揭示一种控制释放剂型,含有水溶性藻酸盐类、藻酸盐复合物及有机羧酸(其可促进活性成分之溶离)。活性成分之一例子为克拉霉素,其很容易溶解于胃液中,以及于小肠的上端部位(pH 5.0)易溶解,在该部位克拉霉素最可能被吸收;但克拉霉素在小肠下端部位(pH 6到8)不易溶解,故为了增加在此部位之溶解度,在配方中使用柠檬酸。美国专利第5,705,190号所申请之口服药学组合物中,藻酸钠对藻酸钠-钙的重量比约为16∶1到1∶1,而有机酸(例如柠檬酸)对主药的摩尔比为1∶1。The patented technology mentioned above cannot be applied to the formulation containing the main drug with very low water solubility, for example, the formulation made of clarithromycin and alginate, its dissolution is too slow and its in vivo test results are limited. Bioavailability is not reproducible. For drugs with very low water solubility, alginate can be used as a matrix and organic acids can be added to make solid controlled release dosage forms. U.S. Patent No. 5,705,190 (which is equivalent to Taiwan Patent No. 429,154) discloses a controlled release dosage form containing water-soluble alginates, alginate complexes and organic carboxylic acids (which can promote the dissolution of active ingredients) . An example of an active ingredient is clarithromycin, which is readily soluble in gastric juices and in the upper part of the small intestine (pH 5.0), where clarithromycin is most likely to be absorbed; (
在美国专利第5,705,190号中,其固态控制释放之药学组合物包含藻酸钠、藻酸钠-钙和有机羧酸(例如柠檬酸)。然而,此配方无法解决或降低关于肠胃道不适方面之副作用,包括恶心、呕吐及味觉失常等症状。为了克服上述这些问题,因此发展出含有难溶性主药(例如红霉素)之改良控制释放剂型,如叙述在美国专利第6,010,718号者。这些配方包含了药学上可接受的高分子聚合物,如羟丙基甲基纤维素,来改善快速释放剂型之味觉,及降低肠胃道之副作用。美国专利第6,551,661B1号和美国专利第6,010,718号内容相似,但前者包含了较多对于病人之肠胃道之副作用研究结果,且证明服用克拉霉素之持续释放配方之病人显著较不会因为肠胃副作用而停药,且这些病人身上所发生之肠胃副作用显著较不严重。In US Patent No. 5,705,190, the solid controlled release pharmaceutical composition comprises sodium alginate, sodium alginate-calcium and organic carboxylic acid (such as citric acid). However, this formulation cannot resolve or reduce side effects related to gastrointestinal discomfort, including symptoms such as nausea, vomiting, and dysgeusia. In order to overcome the above-mentioned problems, an improved controlled-release dosage form containing a poorly soluble main drug (such as erythromycin) has been developed, as described in US Patent No. 6,010,718. These formulations contain pharmaceutically acceptable polymers, such as hydroxypropyl methylcellulose, to improve the taste of the immediate release dosage form and reduce gastrointestinal side effects. U.S. Patent No. 6,551,661B1 is similar to U.S. Patent No. 6,010,718, but the former includes more research results on the side effects of the gastrointestinal tract of patients, and it is proved that patients taking clarithromycin sustained-release formulations are significantly less likely to suffer from gastrointestinal side effects However, the drug was discontinued, and the gastrointestinal side effects occurred in these patients were significantly less serious.
如同美国专利第5,705,190号中所揭示的,XL锭(Abbott Laboratories Ltd.,Queenborough,Kent,ME11 5EL,英国,克拉霉素持续释放锭)含有500毫克的克拉霉素和其它非活性组份如:柠檬酸、藻酸钠、藻酸钠-钙、乳糖、聚乙烯吡咯酮(povidone)、滑石粉、硬脂酸、硬脂酸镁、羟丙基甲基纤维素、丙二醇、二氧化钛(E1711)、山梨酸、喹啉(quinoline)黄色色素(E104)等。而此配方所含之藻酸钠(水溶性的藻酸盐)、藻酸钠-钙(水不溶性的藻酸盐)、及柠檬酸(有机羧酸)系用来帮助克拉霉素溶离。藻酸钠与藻酸钠-钙的含量相差悬殊,所以要混合均匀是困难的。在美国专利第5,705,190号中虽提及藻酸钠对藻酸钠-钙的比是16∶1到1∶1,但其在XL锭中之实际重量比为8∶1。若将藻酸钠和藻酸钠-钙二者的含量调整至接近,例如其比例为2∶1或1∶1,则对生体相等性研究上之需求而言,克拉霉素之溶离速率仍然慢很多。As disclosed in U.S. Patent No. 5,705,190, XL tablets (Abbott Laboratories Ltd., Queenborough, Kent, ME11 5EL, UK, clarithromycin sustained release tablets) contain 500 mg of clarithromycin and other inactive ingredients such as: citric acid, sodium alginate, sodium alginate- Calcium, lactose, povidone, talc, stearic acid, magnesium stearate, hydroxypropyl methylcellulose, propylene glycol, titanium dioxide (E1711), sorbic acid, quinoline yellow coloring ( E104) etc. The sodium alginate (water-soluble alginate), sodium-calcium alginate (water-insoluble alginate), and citric acid (organic carboxylic acid) contained in this formula are used to help clarithromycin dissolve. The content of sodium alginate and sodium alginate-calcium is very different, so it is difficult to mix evenly. Although it is mentioned in U.S. Patent No. 5,705,190 that the ratio of sodium alginate to sodium alginate-calcium is 16:1 to 1:1, it is The actual weight ratio in XL ingots is 8:1. If the contents of sodium alginate and sodium alginate-calcium are adjusted to be close, for example, the ratio is 2:1 or 1:1, the dissolution rate of clarithromycin Still a lot slower.
发明内容 Contents of the invention
此发明的目的是提供一个供口服的含有难溶解性主药(例如红霉素衍生物)之控制释放型药学组合物,在此组合物中只需要用藻酸钠,便足以代替先前技艺中所使用之藻酸钠和藻酸钠-钙的组合物。藻酸钠可用以形成黏性的胶状溶液,且虽然可溶于水中,但不溶于pH小于3的酸性溶液中。所以藻酸钠可保护红霉素衍生物在胃液中不会快速溶解,因而改进红霉素衍生物之安定性。只用藻酸钠不仅可以控制红霉素衍生物之释放,也可避免在制造过程中各组份很难混合均匀的问题。The purpose of this invention is to provide a controlled-release pharmaceutical composition containing a poorly soluble main drug (such as erythromycin derivatives) for oral administration. In this composition, only sodium alginate is needed to replace the previous art. Compositions of sodium alginate and sodium-calcium alginate used. Sodium alginate can be used to form a viscous colloidal solution, and although soluble in water, it is insoluble in acidic solutions with a pH less than 3. So sodium alginate can protect erythromycin derivatives from rapidly dissolving in gastric juice, thus improving the stability of erythromycin derivatives. Only using sodium alginate can not only control the release of erythromycin derivatives, but also avoid the problem that the components are difficult to mix uniformly during the manufacturing process.
另一方面,本发明提供一种供口服的含有难溶解性主药(例如红霉素衍生物)之控制释放型药剂,且有膜衣包覆。本发明之药剂在与快速释放型药学组合物比较下,有较改良之口感,且肠胃道不适方面之副作用被降至最低。In another aspect, the present invention provides a controlled-release medicament containing a poorly soluble main drug (such as erythromycin derivatives) for oral administration and coated with a film coating. Compared with the rapid-release pharmaceutical composition, the medicament of the present invention has improved taste, and the side effects of gastrointestinal discomfort are minimized.
附图说明 Description of drawings
图1示出XL(以ORI代表)和本发明(以PBF代表)之克拉霉素持续释放型锭在pH5.0缓冲溶液中的溶离曲线图。Figure 1 shows Dissolution curves of clarithromycin sustained-release tablets of XL (represented by ORI) and the present invention (represented by PBF) in pH 5.0 buffer solution.
具体实施方式 Detailed ways
本发明提供一种每日服药一次的疗法,它是使需要此治疗之病人口服含有难溶解性主药(例如红霉素衍生物)之固态控制释放型药学组合物,其较好的剂型是锭剂。The present invention provides a therapy for taking medicine once a day, which is to make the patient in need of the therapy orally contain a solid-state controlled-release pharmaceutical composition containing a poorly soluble main drug (such as erythromycin derivatives), and its preferred dosage form is Lozenges.
红霉素是一种大环内酯抗生素,它溶解于酒精及一般的有机溶剂,但只微溶于水中;在低于pH 4之溶液中极端的不稳定。红霉素衍生物可包括以下诸类型:(1)在去氧糖胺之二甲基氨基上的酸加成盐,例如葡庚糖酸盐,乳糖醛酸盐和硬脂酸盐;(2)在去氧糖胺之OH基上之酯,例如乙基碳酸酯、乙基琥珀酸酯及丙酸酯;及(3)克拉霉素等。Erythromycin is a macrolide antibiotic, which is soluble in alcohol and common organic solvents, but only slightly soluble in water; it is extremely unstable in solutions below
本发明的药学组合物可包含已知之其它的药品以和红霉素衍生物合并使用,只要此种合并治疗为需要的或有利的;而这些与治疗胃炎、溃疡或胃食道逆流疾病(GERD)有关之其它药物,可为抗溃疡或抗胃炎的药物,例如选自:抑制胃分泌的化合物,如磺胺甲唑(sulfamethoxazole)、咪唑尼达(metronidazole)、西咪替丁(cimetidine)、吲达帕胺(indapamide)、阿替洛尔(atenolol)、地西泮(diazepam)、奥美拉唑(omeprazole)、雷尼替丁(ranitidine)、硫糖铝(sucralfate)等;或制酸剂如氢氧化镁、氢氧化铝、碳酸钠、迈利康(simethicone,亦称二甲基硅油或喜每赐康)等。The pharmaceutical composition of the present invention may contain other known drugs to be used in combination with the erythromycin derivatives, as long as such combined treatment is needed or beneficial; and these are related to the treatment of gastritis, ulcer or gastroesophageal reflux disease (GERD) The other related drugs can be anti-ulcer or anti-gastritis drugs, for example, selected from: compounds that inhibit gastric secretion, such as sulfamethoxazole, metronidazole, cimetidine, inda Indapamide, atenolol, diazepam, omeprazole, ranitidine, sucralfate, etc.; or antacids such as Magnesium hydroxide, aluminum hydroxide, sodium carbonate, simethicone (also known as simethicone or simethicone), etc.
上述这些药物在药学组合物中之含量范围可为整体组合物或整个药锭的20%到90%。对克拉霉素而言,此含量范围较佳可为整体组合物或整个药锭重量的40%到80%。The content range of the above-mentioned drugs in the pharmaceutical composition may be 20% to 90% of the whole composition or the whole tablet. For clarithromycin, the content range is preferably 40% to 80% by weight of the whole composition or the whole tablet.
本发明虽然较佳系使用藻酸钠,但其它阳离子,例如钾离子、铵离子及其它的碱性金属离子,亦可取代钠离子来形成水溶性的藻酸盐类。水溶性藻酸盐之含量范围可为整体组合物或整个药锭重量的10%到40%。Although sodium alginate is preferably used in the present invention, other cations, such as potassium ions, ammonium ions and other basic metal ions, can also replace sodium ions to form water-soluble alginates. The content of water-soluble alginate can range from 10% to 40% by weight of the whole composition or the whole tablet.
在本发明的控制释放配方中所需的有机羧酸为一有足够效力来创造低pH值微环境的酸,也就是于水解剂型之附近形成pH小于7的微环境。此主药对酸的重量比例范围可从1∶1到6∶1,而以4∶1的比例最佳。该酸以含有3到20个碳原子的脂肪族有机羧酸为佳,例如琥珀酸、酒石酸、苹果酸、戊二酸、顺丁烯二酸、麸胺酸、柠檬酸、杏仁酸等,而以柠檬酸最佳。The desired organic carboxylic acid in the controlled release formulations of the present invention is one that is sufficiently effective to create a low pH microenvironment, ie, a pH less than 7 microenvironment in the vicinity of the hydrolyzed dosage form. The weight ratio of the main ingredient to the acid can range from 1:1 to 6:1, and the ratio of 4:1 is the best. The acid is preferably an aliphatic organic carboxylic acid containing 3 to 20 carbon atoms, such as succinic acid, tartaric acid, malic acid, glutaric acid, maleic acid, glutamic acid, citric acid, mandelic acid, etc., and Citric acid is the best.
控制释放配方中的藻酸钠,在肠胃道的胃液中会反应成为藻酸,而藻酸可微溶于水中,且会在控制释放剂型的表层形成一层薄膜,用以控制药物的释放,此系因为该膜会控制药物的扩散。结果是,水不溶的藻酸盐,例如藻酸钠-钙,已不再需要被使用以控制克拉霉素的释放。The sodium alginate in the controlled-release formula will react into alginic acid in the gastric juice of the gastrointestinal tract, and the alginic acid is slightly soluble in water, and will form a film on the surface of the controlled-release dosage form to control the release of the drug. This is because the membrane controls the diffusion of the drug. As a result, water-insoluble alginates, such as sodium-calcium alginate, no longer need to be used to control the release of clarithromycin.
在本发明控制释放配方中作为非主成分的其它成分包括:药学上可接受之赋形剂、稀释剂、保存剂(防腐剂)、润滑剂、滑动剂、及经许可之色素。本发明之剂型也可在外层被涂覆与药物释放的控制或修正无关之物质。Other ingredients that are not main ingredients in the controlled-release formulation of the present invention include: pharmaceutically acceptable excipients, diluents, preservatives (preservatives), lubricants, slip agents, and approved pigments. The dosage forms of the present invention may also be coated on the outer layer with substances not related to the control or modification of drug release.
实施例1Example 1
1.控制释放型药锭的制粒1. Granulation of controlled release tablets
所有的锭剂配方系以如下述之一般制造方法而制备。克拉霉素、藻酸钠、乳糖和硬酯酸全部通过40号筛网,来移去任何大的结块,而这些已过筛的物质在混合机(Supermixer)中混合20分钟,再缓缓加入聚乙烯吡咯酮和柠檬酸溶液形成适合的颗粒。这些湿的颗粒通过16号筛网,且用60℃的热风干燥直到这些颗粒以卡氏(Karl Fisher)水份测定仪测定水分含量为5~7%。这些干燥的颗粒通过20号筛网后,和润滑剂在V型混合机上混合2分钟。All lozenge formulations were prepared by the general method of manufacture as described below. The clarithromycin, sodium alginate, lactose, and stearic acid were all passed through a No. 40 sieve to remove any large lumps, and the sieved material was mixed in a Supermixer for 20 minutes, then slowly A solution of povidone and citric acid was added to form suitable granules. These wet granules pass through a No. 16 sieve, and are dried with hot air at 60° C. until the granules have a moisture content of 5-7% as measured by a Karl Fisher moisture analyzer. After passing through a No. 20 screen, the dried granules were mixed with lubricant on a V-blender for 2 minutes.
2.打锭2. Ingot making
将旋转打锭机装设椭圆形的模具,分别将如以下表一所示的锭剂调配物I、II与III压成适合的厚度和脆度。锭剂调配物的组成如以下表I所示:The rotary tablet machine was equipped with an oval mold, and the tablet formulations I, II and III shown in Table 1 below were respectively pressed into suitable thickness and crispness. The composition of the lozenge formulation is shown in Table I below:
表ITable I
3.形成膜状涂层3. Form a film-like coating
使用一水性悬浮液来涂覆锭剂,该水性悬浮液包含羟丙基甲基纤维素、聚乙二醇、山梨醇、二氧化钛和纯水,结果约增加锭剂总重量之2.5%。The tablets were coated with an aqueous suspension comprising hydroxypropylmethylcellulose, polyethylene glycol, sorbitol, titanium dioxide and purified water, resulting in an increase of about 2.5% of the total weight of the tablets.
实施例2Example 2
溶离度研究Solubility studies
1.溶离度试验,依据美国药典第27版第463页(2004)1. Solubility test, according to page 463 of the 27th edition of the United States Pharmacopoeia (2004)
0.1M醋酸钠缓冲溶液:其制法为:取醋酸钠三水合物13.61g,置于1升容量瓶中,加水溶并稀释至容量,混合均匀,以0.1M醋酸调整其pH值为5.0。溶剂:900mL之上述0.1M醋酸钠缓冲溶液。0.1M sodium acetate buffer solution: the preparation method is as follows: take 13.61g of sodium acetate trihydrate, put it in a 1-liter volumetric flask, add water to dissolve it and dilute to volume, mix well, and adjust its pH value to 5.0 with 0.1M acetic acid. Solvent: 900 mL of the above 0.1 M sodium acetate buffer solution.
装置2:50rpmDevice 2: 50rpm
取样时间点:第1、2、4、6、8、10、12、18与24小时。Sampling time points: 1st, 2nd, 4th, 6th, 8th, 10th, 12th, 18th and 24th hours.
移动相:其制法为:取甲醇与0.067M磷酸二氢钾液之混合物(甲醇对0.067M磷酸二氢钾液之比例为650∶350),加磷酸调整其pH为4.0,以孔径0.5μm或更细之滤器过滤并脱气处理,必要时可再予以调整。Mobile phase: Its preparation method is: take a mixture of methanol and 0.067M potassium dihydrogen phosphate solution (the ratio of methanol to 0.067M potassium dihydrogen phosphate solution is 650:350), add phosphoric acid to adjust its pH to 4.0, and use a pore size of 0.5 μm or a finer filter and degassed, and adjusted if necessary.
标准品溶液:其制法为:取约27mg之经准确秤重的克拉霉素,在50mL定量瓶中以40mL之如上述溶剂溶解,若需要可进行摇动及超音波振荡,以确保溶解完全。以溶剂稀释至定量,混匀后以孔径0.5μm或更细之滤器过滤。取滤液作为标准品溶液。此溶液每mL含有约54μg之克拉霉素。Standard solution: The preparation method is as follows: take about 27mg of clarithromycin which has been accurately weighed, and dissolve it with 40mL of the above-mentioned solvent in a 50mL quantitative bottle. If necessary, shake and ultrasonically oscillate to ensure complete dissolution. Dilute to quantitative with a solvent, mix well and filter with a filter with a pore size of 0.5 μm or finer. Take the filtrate as the standard solution. This solution contained approximately 54 μg of clarithromycin per mL.
层析装置:液相层析装置,具波长210nm检测器,及4.6mm×15cm Inertsil ODS-2层析管:5μm,层析管温度保持在约50℃,移动相流速每分钟约1mL。将标准品溶液与受试品溶液进行层析,记录其反应:由克拉霉素波峰值所测得之管柱效率,其为不小于750理论板数,其波峰曳尾因子为不小于0.9亦不大于2;而重复注入之相对标准差为不大于2.0%。Chromatography device: liquid chromatography device, with a detector with a wavelength of 210nm, and a 4.6mm×15cm Inertsil ODS-2 chromatography tube: 5μm, the temperature of the chromatography tube is kept at about 50°C, and the flow rate of the mobile phase is about 1mL per minute. Perform chromatography on the standard solution and the test solution, and record the reaction: the column efficiency measured by the clarithromycin wave peak is not less than 750 theoretical plates, and the peak tailing factor is not less than 0.9. Not greater than 2; and the relative standard deviation of repeated injections is not greater than 2.0%.
测定法:取标准品溶液与受试品溶液(该受试品溶液为将取得之样品经定量之移动相稀释所形成之溶液,起始每mL含有约54μg之克拉霉素)等量(约20~50μL),分别注入层析装置层析之,记录其层析图,测计各主波峰。依照下式计算克拉霉素在溶离试验中所溶离的量(单位为μg):Determination method: take the standard solution and the test solution (the test solution is a solution formed by diluting the obtained sample with a quantitative mobile phase, and initially contains about 54 μg of clarithromycin per mL) (about 20-50 μL), respectively injected into the chromatographic device for chromatography, recorded the chromatogram, and measured the main peaks. Calculate the amount of clarithromycin dissolved in the dissolution test (in μg) according to the following formula:
900(CD)(ru/rs)900(CD)(r u /r s )
其中in
C:标准品溶液每mL含克拉霉素之μg数C: μg of clarithromycin per mL of the standard solution
D:配制受试品溶液时之适当稀释倍数D: Appropriate dilution factor when preparing the test product solution
ru和rs:分别为检品溶液及标准品溶液主成分之波峰值r u and r s : the peak values of the main components of the test solution and the standard solution, respectively
2.溶离度结果和讨论2. Dissolution Results and Discussion
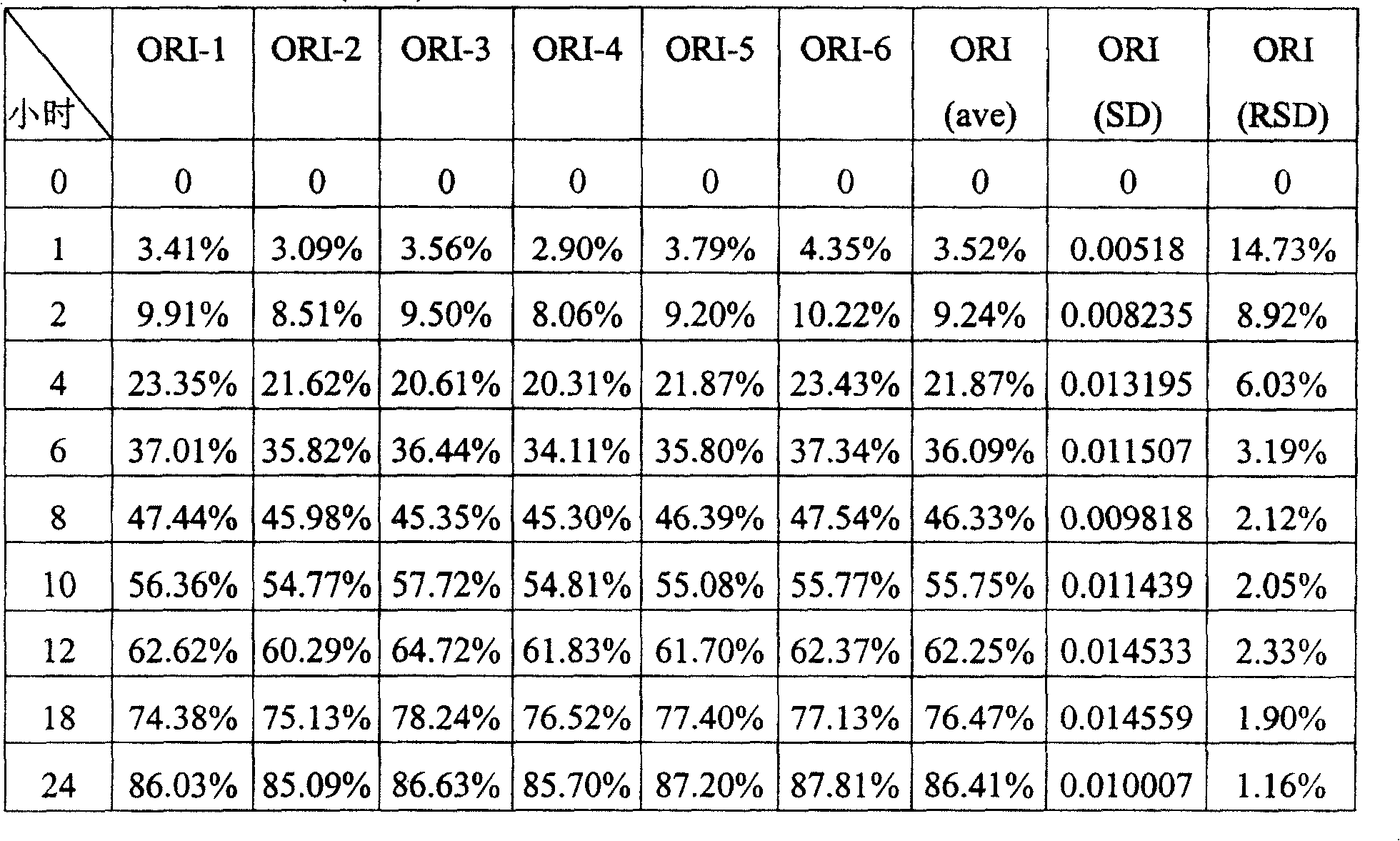
分别以得自Abbott Laboratories Ltd.(Queenborough,Kent,ME11 5EL,英国)的XL锭(克拉霉素,500mg)和本发明的持续释放型克拉霉素锭剂各6粒,在如上述之0.1M醋酸钠缓冲溶液中,进行如上述之溶离度研究,其溶离度试验的条件如上所述。而两者来自不同来源锭剂之溶离度结果如下表II示之。Respectively obtained from Abbott Laboratories Ltd. (Queenborough, Kent, ME11 5EL, UK) Each of XL tablets (clarithromycin, 500mg) and sustained-release clarithromycin lozenges of the present invention has 6 capsules. In the above-mentioned 0.1M sodium acetate buffer solution, the above-mentioned solubility research is carried out. The conditions of the test are as described above. The results of the solubility of the two tablets from different sources are shown in Table II below.
表II克拉霉素锭剂之溶离度研究Table II Dissolution studies of clarithromycin lozenges
本发明:本发明之持续释放型克拉霉素锭剂The present invention: sustained-release clarithromycin tablet of the present invention
二者的溶离曲线如图1所示,此图为XL(ORI)和本发明(PBF)之克拉霉素持续释放型锭的溶离曲线图。The dissociation curves of the two are shown in Figure 1, which is Dissolution profiles of clarithromycin sustained-release tablets of XL (ORI) and the present invention (PBF).
表III---在六次测试中,每一个XL锭剂(ORI)之溶离结果,平均值以(ave)表示,相对标准差以(RSD)表示Table III --- Among the six tests, each Dissolution results of XL lozenges (ORI), the average value is expressed as (ave), and the relative standard deviation is expressed as (RSD)
表IV---在六次测试中,每一个本发明之克拉霉素持续释放型锭剂(PBF)的溶离结果,平均值以(ave)表示,相对标准差以(RSD)表示Table IV --- in six tests, the dissolution results of each clarithromycin sustained-release lozenge (PBF) of the present invention, the average value is expressed in (ave), and the relative standard deviation is expressed in (RSD)
二者的溶离曲线如图1所示,此图为XL锭剂(ORI)和本发明(PBF)之克拉霉素持续释放型锭剂的溶离曲线图,由图1可见二者在各时间点的溶离度非常的接近,只除了在第十八小时之溶离度相差有5.46%(亦即,81.93%-76.47%=5.46%)外,其它各点均小于5%,因此二者在临床试验中,可能彼此为生体相等性的。The dissociation curves of the two are shown in Figure 1, which is The dissolution curves of the clarithromycin sustained-release tablet of the XL lozenge (ORI) and the present invention (PBF), as can be seen from Fig. 1, the dissolution rate of the two at each time point is very close, except at the tenth Except for the difference of 5.46% (that is, 81.93%-76.47%=5.46%) in the eight-hour solubility, the other points are all less than 5%. Therefore, the two may be biologically equivalent to each other in clinical trials.
此外,XL锭剂和本发明之克拉霉素持续型锭剂的每一锭的各个时间点溶离结果,如表III和表IV所示,在对于XL锭剂之六次测试中,在每个时间点的相对标准误差(RSD)在1.16%与14.37%之间,而对于本发明之克拉霉素持续释放型锭剂之六次测试中,在每个时间点的相对标准误差(RSD)在0.92%与10.49%之间。因此,XL锭剂和本发明之克拉霉素持续释放型锭剂的平均相对标准误差分别是4.71%和2.98%。此表示了本发明之多个克拉霉素持续释放型锭剂彼此间之溶离型态较相似,而相比之下显出,多个XL锭剂彼此间之溶离型态较不相似。此项优点系因为本发明中使用藻酸钠,反观XL锭剂中使用藻酸钠和藻酸钠-钙之组合物,而在XL锭剂之制造过程中该两种不同的藻酸盐会引起较难混合的问题。also, Dissolution results at each time point of XL lozenge and clarithromycin continuous lozenge of the present invention, as shown in table III and table IV, for In the six tests of the XL tablet, the relative standard deviation (RSD) at each time point was between 1.16% and 14.37%, while in the six tests of the clarithromycin sustained-release tablet of the present invention, in The relative standard deviation (RSD) for each time point was between 0.92% and 10.49%. therefore, The mean relative standard errors for the XL tablet and the clarithromycin sustained release tablet of the present invention were 4.71% and 2.98%, respectively. This shows that the dissolution patterns of multiple clarithromycin sustained-release tablets of the present invention are similar to each other, while in contrast, multiple The dissolution profiles of the XL lozenges were less similar to each other. This advantage is because sodium alginate is used in the present invention, on the other hand Sodium alginate and a combination of sodium alginate-calcium were used in XL lozenges, whereas in The two different alginates caused more difficult mixing problems during the manufacture of the XL lozenges.
本发明的优点Advantages of the invention
A.本发明在与使用两种不同藻酸盐来制备持续释放型锭剂之美国专利第5,705,190号比较下,显出为较易于操作且成本可较为降低。A. Compared with US Patent No. 5,705,190, which uses two different alginates to prepare sustained-release lozenges, the present invention is easier to operate and lower in cost.
B.在本发明之控制释放配方的藻酸钠,在肠胃道的胃液中会反应成为藻酸,而藻酸可微溶于水中,且会在控制释放剂型的表层形成一层薄膜,用以控制药物的释放,此系因为该膜会控制药物的扩散。B. The sodium alginate in the controlled-release formulation of the present invention will react into alginic acid in the gastric juice of the gastrointestinal tract, and alginic acid is slightly soluble in water, and will form a thin film on the surface of the controlled-release dosage form for use in The release of the drug is controlled because the membrane controls the diffusion of the drug.
C.由表II的数据可知,本发明之锭剂在和XL锭剂比较下,其溶离度并没有显著的差异;然而,由表III与IV可知,本发明者有明显较小的平均相对标准误差(RSD),因此可得知本发明之多个克拉霉素持续释放型锭剂彼此间之溶离型态较相似,而相比之下显出,多个XL锭剂彼此间之溶离型态较不相似。C. Known from the data of table II, lozenge of the present invention is in and Compared with XL lozenges, there is no significant difference in its dissolution rate; however, as can be seen from Tables III and IV, the inventors have significantly smaller average relative standard deviation (RSD), so it can be known that the present invention has multiple The dissolution profiles of clarithromycin sustained-release lozenges were relatively similar to each other, while in contrast multiple The dissolution profiles of the XL lozenges were less similar to each other.
Claims (9)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CNB200410043476XA CN100435847C (en) | 2004-05-13 | 2004-05-13 | Sustained release formulation containing poorly soluble principal drug |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CNB200410043476XA CN100435847C (en) | 2004-05-13 | 2004-05-13 | Sustained release formulation containing poorly soluble principal drug |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN1695739A CN1695739A (en) | 2005-11-16 |
| CN100435847C true CN100435847C (en) | 2008-11-26 |
Family
ID=35348728
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CNB200410043476XA Expired - Fee Related CN100435847C (en) | 2004-05-13 | 2004-05-13 | Sustained release formulation containing poorly soluble principal drug |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN100435847C (en) |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN115721626A (en) * | 2022-11-23 | 2023-03-03 | 浙江普利药业有限公司 | Clarithromycin sustained release tablet and preparation method thereof |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1205629A (en) * | 1995-12-19 | 1999-01-20 | 艾博特公司 | Controlled release formulation for poorly soluble basic drugs |
| CN1316899A (en) * | 1998-07-31 | 2001-10-10 | 大塚制药株式会社 | Pharmaceutical composition having improved taste |
| CN1415305A (en) * | 2002-11-25 | 2003-05-07 | 沈阳药科大学 | Sustained release preparation of roxithromycin |
-
2004
- 2004-05-13 CN CNB200410043476XA patent/CN100435847C/en not_active Expired - Fee Related
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1205629A (en) * | 1995-12-19 | 1999-01-20 | 艾博特公司 | Controlled release formulation for poorly soluble basic drugs |
| CN1316899A (en) * | 1998-07-31 | 2001-10-10 | 大塚制药株式会社 | Pharmaceutical composition having improved taste |
| CN1415305A (en) * | 2002-11-25 | 2003-05-07 | 沈阳药科大学 | Sustained release preparation of roxithromycin |
Also Published As
| Publication number | Publication date |
|---|---|
| CN1695739A (en) | 2005-11-16 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN1131027C (en) | Controlled release formulation for poorly soluble basic drugs | |
| US10441585B2 (en) | Formulations containing nalbuphine and uses thereof | |
| KR20130030306A (en) | Pharmaceutical compositions | |
| JP2020518611A (en) | Compositions with improved water solubility and bioavailability | |
| KR20210054137A (en) | Sustained-release pharmaceutical compositions for oral administration comprising rebamipide or pharmaceutically acceptable salt thereof | |
| WO1995015751A1 (en) | Combined antipyretic analgesic drug | |
| EP3305282A2 (en) | Composition of pranlukast-containing solid preparation with improved bioavailability and method for preparing same | |
| CN101747305A (en) | Five crystal forms of nicousamide compound, preparation method, pharmaceutical composition and application thereof | |
| MXPA01001120A (en) | Pharmaceutical compositions comprising ibuprofen and domperidone. | |
| CN100393302C (en) | Insoluble pharmaceutical composition osmotic pump controlled release preparation | |
| US20110060008A1 (en) | Pharmaceutical composition containing acetylcholine esterase inhibitor and method for the preparation thereof | |
| US5968906A (en) | Sucralfate preparations | |
| JP2010001242A (en) | Rebamipide solid preparation, and method for producing the same | |
| CN100435847C (en) | Sustained release formulation containing poorly soluble principal drug | |
| US20220288056A1 (en) | Pharmaceutical composition containing nitroxoline, nitroxoline oral solid tablet, preparation method therefor and use thereof | |
| CN120022270A (en) | Indobufen sustained-release preparation and preparation method and application thereof | |
| WO2021254409A1 (en) | Pharmaceutical composition of complex and preparation method therefor | |
| DK175526B1 (en) | Pharmaceutical composition and process for its preparation | |
| KR101561345B1 (en) | Controlled-release pharmaceutical composition of propionic acid derivatives | |
| WO2019230937A1 (en) | Solid oral dosage form having excellent dissolution properties | |
| KR101175816B1 (en) | Sustained release tablet for oral use | |
| US20050260263A1 (en) | Sustained release formulation for sparingly soluble main drugs | |
| TWI351290B (en) | ||
| CN112315924A (en) | Azithromycin composition and preparation method thereof | |
| CN107872973A (en) | Ranolazine multiple compressed tablets |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| CF01 | Termination of patent right due to non-payment of annual fee | ||
| CF01 | Termination of patent right due to non-payment of annual fee |
Granted publication date: 20081126 Termination date: 20180513 |