CN100434419C - Compound of monocyclic polysubstitution saturated cyclohexanones, prepartion method and usage - Google Patents
Compound of monocyclic polysubstitution saturated cyclohexanones, prepartion method and usage Download PDFInfo
- Publication number
- CN100434419C CN100434419C CNB2005100748051A CN200510074805A CN100434419C CN 100434419 C CN100434419 C CN 100434419C CN B2005100748051 A CNB2005100748051 A CN B2005100748051A CN 200510074805 A CN200510074805 A CN 200510074805A CN 100434419 C CN100434419 C CN 100434419C
- Authority
- CN
- China
- Prior art keywords
- compound
- formula
- extract
- cells
- saturated
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
- 150000001875 compounds Chemical class 0.000 title claims abstract description 88
- JHIVVAPYMSGYDF-UHFFFAOYSA-N cyclohexanone Chemical class O=C1CCCCC1 JHIVVAPYMSGYDF-UHFFFAOYSA-N 0.000 title claims abstract description 20
- 238000000034 method Methods 0.000 title claims description 22
- 125000002950 monocyclic group Chemical group 0.000 title abstract description 16
- 230000006907 apoptotic process Effects 0.000 claims abstract description 18
- 239000000284 extract Substances 0.000 claims abstract description 17
- 238000002360 preparation method Methods 0.000 claims abstract description 15
- 230000004663 cell proliferation Effects 0.000 claims abstract description 9
- 230000000259 anti-tumor effect Effects 0.000 claims abstract description 8
- 229940123587 Cell cycle inhibitor Drugs 0.000 claims abstract description 5
- 239000003112 inhibitor Substances 0.000 claims abstract description 4
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims description 21
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 18
- 239000001257 hydrogen Substances 0.000 claims description 17
- 229910052739 hydrogen Inorganic materials 0.000 claims description 17
- 241000871537 Choerospondias axillaris Species 0.000 claims description 14
- 235000014116 Spondias axillaris Nutrition 0.000 claims description 13
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 12
- 239000003814 drug Substances 0.000 claims description 9
- 239000000287 crude extract Substances 0.000 claims description 8
- 239000004480 active ingredient Substances 0.000 claims description 7
- 150000002431 hydrogen Chemical class 0.000 claims description 7
- 230000001939 inductive effect Effects 0.000 claims description 7
- 239000008194 pharmaceutical composition Substances 0.000 claims description 5
- 239000003139 biocide Substances 0.000 claims description 3
- 239000003937 drug carrier Substances 0.000 claims description 3
- 239000000546 pharmaceutical excipient Substances 0.000 claims description 3
- 230000008569 process Effects 0.000 claims description 3
- 210000004881 tumor cell Anatomy 0.000 claims description 3
- 230000022534 cell killing Effects 0.000 claims 1
- 239000003795 chemical substances by application Substances 0.000 claims 1
- 238000002386 leaching Methods 0.000 claims 1
- 125000001183 hydrocarbyl group Chemical group 0.000 abstract description 10
- 239000002246 antineoplastic agent Substances 0.000 abstract description 5
- 239000000411 inducer Substances 0.000 abstract description 4
- 238000002560 therapeutic procedure Methods 0.000 abstract 1
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 78
- 210000004027 cell Anatomy 0.000 description 58
- HTSGKJQDMSTCGS-UHFFFAOYSA-N 1,4-bis(4-chlorophenyl)-2-(4-methylphenyl)sulfonylbutane-1,4-dione Chemical compound C1=CC(C)=CC=C1S(=O)(=O)C(C(=O)C=1C=CC(Cl)=CC=1)CC(=O)C1=CC=C(Cl)C=C1 HTSGKJQDMSTCGS-UHFFFAOYSA-N 0.000 description 34
- 238000010828 elution Methods 0.000 description 28
- CSCPPACGZOOCGX-UHFFFAOYSA-N acetone Substances CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 26
- -1 saturated cyclohexanone compound Chemical class 0.000 description 20
- 230000002401 inhibitory effect Effects 0.000 description 14
- 206010028980 Neoplasm Diseases 0.000 description 12
- 230000022131 cell cycle Effects 0.000 description 12
- 238000001514 detection method Methods 0.000 description 12
- 238000012360 testing method Methods 0.000 description 12
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 11
- 230000000694 effects Effects 0.000 description 11
- 230000005764 inhibitory process Effects 0.000 description 11
- 125000001424 substituent group Chemical group 0.000 description 11
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 10
- 201000011510 cancer Diseases 0.000 description 10
- 239000000243 solution Substances 0.000 description 10
- 238000001228 spectrum Methods 0.000 description 9
- 206010006187 Breast cancer Diseases 0.000 description 8
- 208000026310 Breast neoplasm Diseases 0.000 description 8
- 229920006395 saturated elastomer Polymers 0.000 description 8
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 8
- 238000005481 NMR spectroscopy Methods 0.000 description 7
- 238000000684 flow cytometry Methods 0.000 description 7
- 230000001965 increasing effect Effects 0.000 description 7
- 150000002500 ions Chemical class 0.000 description 7
- 230000000877 morphologic effect Effects 0.000 description 7
- OKKJLVBELUTLKV-MZCSYVLQSA-N Deuterated methanol Chemical compound [2H]OC([2H])([2H])[2H] OKKJLVBELUTLKV-MZCSYVLQSA-N 0.000 description 6
- 238000004458 analytical method Methods 0.000 description 6
- 238000004128 high performance liquid chromatography Methods 0.000 description 6
- 238000000746 purification Methods 0.000 description 6
- 238000011160 research Methods 0.000 description 6
- 150000003839 salts Chemical class 0.000 description 6
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 5
- 206010009944 Colon cancer Diseases 0.000 description 5
- 208000001333 Colorectal Neoplasms Diseases 0.000 description 5
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 5
- 239000000969 carrier Substances 0.000 description 5
- 239000002026 chloroform extract Substances 0.000 description 5
- WORJEOGGNQDSOE-UHFFFAOYSA-N chloroform;methanol Chemical compound OC.ClC(Cl)Cl WORJEOGGNQDSOE-UHFFFAOYSA-N 0.000 description 5
- 230000001472 cytotoxic effect Effects 0.000 description 5
- 238000000655 nuclear magnetic resonance spectrum Methods 0.000 description 5
- 239000000523 sample Substances 0.000 description 5
- 239000000741 silica gel Substances 0.000 description 5
- 229910002027 silica gel Inorganic materials 0.000 description 5
- HIXDQWDOVZUNNA-UHFFFAOYSA-N 2-(3,4-dimethoxyphenyl)-5-hydroxy-7-methoxychromen-4-one Chemical compound C=1C(OC)=CC(O)=C(C(C=2)=O)C=1OC=2C1=CC=C(OC)C(OC)=C1 HIXDQWDOVZUNNA-UHFFFAOYSA-N 0.000 description 4
- 206010008342 Cervix carcinoma Diseases 0.000 description 4
- 241000196324 Embryophyta Species 0.000 description 4
- 206010033128 Ovarian cancer Diseases 0.000 description 4
- 206010061535 Ovarian neoplasm Diseases 0.000 description 4
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical compound [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 4
- 208000006105 Uterine Cervical Neoplasms Diseases 0.000 description 4
- 238000000862 absorption spectrum Methods 0.000 description 4
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 4
- 230000009702 cancer cell proliferation Effects 0.000 description 4
- 201000010881 cervical cancer Diseases 0.000 description 4
- 238000002983 circular dichroism Methods 0.000 description 4
- 230000008878 coupling Effects 0.000 description 4
- 238000010168 coupling process Methods 0.000 description 4
- 238000005859 coupling reaction Methods 0.000 description 4
- JHIVVAPYMSGYDF-PTQBSOBMSA-N cyclohexanone Chemical class O=[13C]1CCCCC1 JHIVVAPYMSGYDF-PTQBSOBMSA-N 0.000 description 4
- 229940079593 drug Drugs 0.000 description 4
- 239000012156 elution solvent Substances 0.000 description 4
- RTZKZFJDLAIYFH-UHFFFAOYSA-N ether Substances CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 4
- 238000002474 experimental method Methods 0.000 description 4
- 238000003919 heteronuclear multiple bond coherence Methods 0.000 description 4
- 230000002147 killing effect Effects 0.000 description 4
- 239000000463 material Substances 0.000 description 4
- 238000002844 melting Methods 0.000 description 4
- 230000008018 melting Effects 0.000 description 4
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 4
- 239000000203 mixture Substances 0.000 description 4
- 230000001338 necrotic effect Effects 0.000 description 4
- 230000003287 optical effect Effects 0.000 description 4
- 239000001301 oxygen Substances 0.000 description 4
- 229910052760 oxygen Inorganic materials 0.000 description 4
- 239000003208 petroleum Substances 0.000 description 4
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 4
- 238000000926 separation method Methods 0.000 description 4
- 239000002904 solvent Substances 0.000 description 4
- WVDDGKGOMKODPV-UHFFFAOYSA-N Benzyl alcohol Chemical compound OCC1=CC=CC=C1 WVDDGKGOMKODPV-UHFFFAOYSA-N 0.000 description 3
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 3
- 239000012980 RPMI-1640 medium Substances 0.000 description 3
- 230000001640 apoptogenic effect Effects 0.000 description 3
- 239000007864 aqueous solution Substances 0.000 description 3
- 230000005880 cancer cell killing Effects 0.000 description 3
- 238000004587 chromatography analysis Methods 0.000 description 3
- 238000005100 correlation spectroscopy Methods 0.000 description 3
- LOKCTEFSRHRXRJ-UHFFFAOYSA-I dipotassium trisodium dihydrogen phosphate hydrogen phosphate dichloride Chemical compound P(=O)(O)(O)[O-].[K+].P(=O)(O)([O-])[O-].[Na+].[Na+].[Cl-].[K+].[Cl-].[Na+] LOKCTEFSRHRXRJ-UHFFFAOYSA-I 0.000 description 3
- 238000009472 formulation Methods 0.000 description 3
- 230000006698 induction Effects 0.000 description 3
- 230000014759 maintenance of location Effects 0.000 description 3
- 239000002953 phosphate buffered saline Substances 0.000 description 3
- 230000035755 proliferation Effects 0.000 description 3
- 238000001953 recrystallisation Methods 0.000 description 3
- 231100000338 sulforhodamine B assay Toxicity 0.000 description 3
- 238000003210 sulforhodamine B staining Methods 0.000 description 3
- 238000004809 thin layer chromatography Methods 0.000 description 3
- XILIYVSXLSWUAI-UHFFFAOYSA-N 2-(diethylamino)ethyl n'-phenylcarbamimidothioate;dihydrobromide Chemical compound Br.Br.CCN(CC)CCSC(N)=NC1=CC=CC=C1 XILIYVSXLSWUAI-UHFFFAOYSA-N 0.000 description 2
- IOOMXAQUNPWDLL-UHFFFAOYSA-N 2-[6-(diethylamino)-3-(diethyliminiumyl)-3h-xanthen-9-yl]-5-sulfobenzene-1-sulfonate Chemical compound C=12C=CC(=[N+](CC)CC)C=C2OC2=CC(N(CC)CC)=CC=C2C=1C1=CC=C(S(O)(=O)=O)C=C1S([O-])(=O)=O IOOMXAQUNPWDLL-UHFFFAOYSA-N 0.000 description 2
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 2
- 229920002261 Corn starch Polymers 0.000 description 2
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 2
- BDAGIHXWWSANSR-UHFFFAOYSA-N Formic acid Chemical compound OC=O BDAGIHXWWSANSR-UHFFFAOYSA-N 0.000 description 2
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 2
- LRHPLDYGYMQRHN-UHFFFAOYSA-N N-Butanol Chemical compound CCCCO LRHPLDYGYMQRHN-UHFFFAOYSA-N 0.000 description 2
- 101100163901 Rattus norvegicus Asic2 gene Proteins 0.000 description 2
- GAMYVSCDDLXAQW-AOIWZFSPSA-N Thermopsosid Natural products O(C)c1c(O)ccc(C=2Oc3c(c(O)cc(O[C@H]4[C@H](O)[C@@H](O)[C@H](O)[C@H](CO)O4)c3)C(=O)C=2)c1 GAMYVSCDDLXAQW-AOIWZFSPSA-N 0.000 description 2
- 238000010521 absorption reaction Methods 0.000 description 2
- 230000009471 action Effects 0.000 description 2
- 239000007900 aqueous suspension Substances 0.000 description 2
- 238000003556 assay Methods 0.000 description 2
- 150000001721 carbon Chemical group 0.000 description 2
- 229910052799 carbon Inorganic materials 0.000 description 2
- 239000006285 cell suspension Substances 0.000 description 2
- 238000004440 column chromatography Methods 0.000 description 2
- 239000008120 corn starch Substances 0.000 description 2
- 239000006071 cream Substances 0.000 description 2
- 239000012043 crude product Substances 0.000 description 2
- 239000002552 dosage form Substances 0.000 description 2
- 238000001035 drying Methods 0.000 description 2
- 201000006549 dyspepsia Diseases 0.000 description 2
- 235000013399 edible fruits Nutrition 0.000 description 2
- 239000000469 ethanolic extract Substances 0.000 description 2
- 238000000605 extraction Methods 0.000 description 2
- 229930003944 flavone Natural products 0.000 description 2
- 150000002212 flavone derivatives Chemical class 0.000 description 2
- 235000011949 flavones Nutrition 0.000 description 2
- 235000019253 formic acid Nutrition 0.000 description 2
- 238000001052 heteronuclear multiple bond coherence spectrum Methods 0.000 description 2
- 238000003929 heteronuclear multiple quantum coherence Methods 0.000 description 2
- 238000011534 incubation Methods 0.000 description 2
- 238000007912 intraperitoneal administration Methods 0.000 description 2
- 238000001990 intravenous administration Methods 0.000 description 2
- 238000002955 isolation Methods 0.000 description 2
- 239000008101 lactose Substances 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- 239000006210 lotion Substances 0.000 description 2
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 2
- 239000002480 mineral oil Substances 0.000 description 2
- 235000010446 mineral oil Nutrition 0.000 description 2
- 238000002156 mixing Methods 0.000 description 2
- 230000004660 morphological change Effects 0.000 description 2
- 230000017074 necrotic cell death Effects 0.000 description 2
- 239000002674 ointment Substances 0.000 description 2
- 150000007524 organic acids Chemical class 0.000 description 2
- 235000019271 petrolatum Nutrition 0.000 description 2
- 239000000843 powder Substances 0.000 description 2
- 238000002953 preparative HPLC Methods 0.000 description 2
- 239000000047 product Substances 0.000 description 2
- XJMOSONTPMZWPB-UHFFFAOYSA-M propidium iodide Chemical compound [I-].[I-].C12=CC(N)=CC=C2C2=CC=C(N)C=C2[N+](CCC[N+](C)(CC)CC)=C1C1=CC=CC=C1 XJMOSONTPMZWPB-UHFFFAOYSA-M 0.000 description 2
- 230000002829 reductive effect Effects 0.000 description 2
- 239000012488 sample solution Substances 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- 238000006467 substitution reaction Methods 0.000 description 2
- 239000006228 supernatant Substances 0.000 description 2
- 239000003826 tablet Substances 0.000 description 2
- 238000010998 test method Methods 0.000 description 2
- 230000000699 topical effect Effects 0.000 description 2
- VHBFFQKBGNRLFZ-UHFFFAOYSA-N vitamin p Natural products O1C2=CC=CC=C2C(=O)C=C1C1=CC=CC=C1 VHBFFQKBGNRLFZ-UHFFFAOYSA-N 0.000 description 2
- LDVVTQMJQSCDMK-UHFFFAOYSA-N 1,3-dihydroxypropan-2-yl formate Chemical compound OCC(CO)OC=O LDVVTQMJQSCDMK-UHFFFAOYSA-N 0.000 description 1
- 238000001644 13C nuclear magnetic resonance spectroscopy Methods 0.000 description 1
- 238000005160 1H NMR spectroscopy Methods 0.000 description 1
- 238000001026 1H--1H correlation spectroscopy Methods 0.000 description 1
- LEACJMVNYZDSKR-UHFFFAOYSA-N 2-octyldodecan-1-ol Chemical compound CCCCCCCCCCC(CO)CCCCCCCC LEACJMVNYZDSKR-UHFFFAOYSA-N 0.000 description 1
- 208000004998 Abdominal Pain Diseases 0.000 description 1
- 241000208223 Anacardiaceae Species 0.000 description 1
- 241000240030 Campnosperma Species 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical group [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- 241000871539 Choerospondias Species 0.000 description 1
- OCUCCJIRFHNWBP-IYEMJOQQSA-L Copper gluconate Chemical class [Cu+2].OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C([O-])=O.OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C([O-])=O OCUCCJIRFHNWBP-IYEMJOQQSA-L 0.000 description 1
- 201000004624 Dermatitis Diseases 0.000 description 1
- 230000035519 G0 Phase Effects 0.000 description 1
- 230000010190 G1 phase Effects 0.000 description 1
- 230000004668 G2/M phase Effects 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 1
- CPELXLSAUQHCOX-UHFFFAOYSA-N Hydrogen bromide Chemical compound Br CPELXLSAUQHCOX-UHFFFAOYSA-N 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- 208000002260 Keloid Diseases 0.000 description 1
- 206010023330 Keloid scar Diseases 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 239000004264 Petrolatum Substances 0.000 description 1
- 206010050661 Platelet aggregation inhibition Diseases 0.000 description 1
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 1
- 229920001214 Polysorbate 60 Polymers 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- 230000018199 S phase Effects 0.000 description 1
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 1
- HVUMOYIDDBPOLL-XWVZOOPGSA-N Sorbitan monostearate Chemical compound CCCCCCCCCCCCCCCCCC(=O)OC[C@@H](O)[C@H]1OC[C@H](O)[C@H]1O HVUMOYIDDBPOLL-XWVZOOPGSA-N 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 description 1
- 239000007983 Tris buffer Substances 0.000 description 1
- 208000027418 Wounds and injury Diseases 0.000 description 1
- 206010000269 abscess Diseases 0.000 description 1
- 150000001242 acetic acid derivatives Chemical class 0.000 description 1
- PQLVXDKIJBQVDF-UHFFFAOYSA-N acetic acid;hydrate Chemical compound O.CC(O)=O PQLVXDKIJBQVDF-UHFFFAOYSA-N 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- 239000008186 active pharmaceutical agent Substances 0.000 description 1
- 239000000443 aerosol Substances 0.000 description 1
- 229910052783 alkali metal Inorganic materials 0.000 description 1
- 150000001340 alkali metals Chemical class 0.000 description 1
- 229910052784 alkaline earth metal Inorganic materials 0.000 description 1
- 150000001342 alkaline earth metals Chemical class 0.000 description 1
- 150000001413 amino acids Chemical class 0.000 description 1
- 230000003321 amplification Effects 0.000 description 1
- 230000003698 anagen phase Effects 0.000 description 1
- 230000001093 anti-cancer Effects 0.000 description 1
- 230000000118 anti-neoplastic effect Effects 0.000 description 1
- 229940041181 antineoplastic drug Drugs 0.000 description 1
- 125000003118 aryl group Chemical group 0.000 description 1
- 208000010668 atopic eczema Diseases 0.000 description 1
- 150000001558 benzoic acid derivatives Chemical class 0.000 description 1
- 235000019445 benzyl alcohol Nutrition 0.000 description 1
- 230000003115 biocidal effect Effects 0.000 description 1
- 230000004071 biological effect Effects 0.000 description 1
- 239000012490 blank solution Substances 0.000 description 1
- 230000037396 body weight Effects 0.000 description 1
- 239000002021 butanolic extract Substances 0.000 description 1
- 159000000007 calcium salts Chemical class 0.000 description 1
- 239000002775 capsule Substances 0.000 description 1
- 239000007963 capsule composition Substances 0.000 description 1
- 125000004432 carbon atom Chemical group C* 0.000 description 1
- 239000001569 carbon dioxide Substances 0.000 description 1
- 229910002092 carbon dioxide Inorganic materials 0.000 description 1
- 238000004113 cell culture Methods 0.000 description 1
- 229940081733 cetearyl alcohol Drugs 0.000 description 1
- HEDRZPFGACZZDS-OUBTZVSYSA-N chloroform-13c Chemical class Cl[13CH](Cl)Cl HEDRZPFGACZZDS-OUBTZVSYSA-N 0.000 description 1
- 238000001142 circular dichroism spectrum Methods 0.000 description 1
- 150000001860 citric acid derivatives Chemical class 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 201000010989 colorectal carcinoma Diseases 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 230000003013 cytotoxicity Effects 0.000 description 1
- 231100000135 cytotoxicity Toxicity 0.000 description 1
- 230000006378 damage Effects 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 230000018109 developmental process Effects 0.000 description 1
- 239000003085 diluting agent Substances 0.000 description 1
- 201000010099 disease Diseases 0.000 description 1
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 1
- 238000009826 distribution Methods 0.000 description 1
- 235000011869 dried fruits Nutrition 0.000 description 1
- 239000003995 emulsifying agent Substances 0.000 description 1
- 230000001804 emulsifying effect Effects 0.000 description 1
- 239000008387 emulsifying waxe Substances 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 239000002024 ethyl acetate extract Substances 0.000 description 1
- 235000003599 food sweetener Nutrition 0.000 description 1
- 230000012010 growth Effects 0.000 description 1
- 239000001963 growth medium Substances 0.000 description 1
- XLYOFNOQVPJJNP-ZSJDYOACSA-N heavy water Substances [2H]O[2H] XLYOFNOQVPJJNP-ZSJDYOACSA-N 0.000 description 1
- 238000002329 infrared spectrum Methods 0.000 description 1
- 238000001802 infusion Methods 0.000 description 1
- 238000002347 injection Methods 0.000 description 1
- 239000007924 injection Substances 0.000 description 1
- 208000014674 injury Diseases 0.000 description 1
- 238000007917 intracranial administration Methods 0.000 description 1
- 238000007918 intramuscular administration Methods 0.000 description 1
- 238000007913 intrathecal administration Methods 0.000 description 1
- 238000007914 intraventricular administration Methods 0.000 description 1
- 210000001117 keloid Anatomy 0.000 description 1
- 150000003893 lactate salts Chemical class 0.000 description 1
- 238000000622 liquid--liquid extraction Methods 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 235000019359 magnesium stearate Nutrition 0.000 description 1
- 150000002688 maleic acid derivatives Chemical class 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 239000002609 medium Substances 0.000 description 1
- 239000012528 membrane Substances 0.000 description 1
- 230000037323 metabolic rate Effects 0.000 description 1
- AFVFQIVMOAPDHO-UHFFFAOYSA-M methanesulfonate group Chemical class CS(=O)(=O)[O-] AFVFQIVMOAPDHO-UHFFFAOYSA-M 0.000 description 1
- 150000007522 mineralic acids Chemical class 0.000 description 1
- 239000003068 molecular probe Substances 0.000 description 1
- 239000000178 monomer Substances 0.000 description 1
- 229930014626 natural product Natural products 0.000 description 1
- 230000035407 negative regulation of cell proliferation Effects 0.000 description 1
- 239000000346 nonvolatile oil Substances 0.000 description 1
- 238000003199 nucleic acid amplification method Methods 0.000 description 1
- 235000003715 nutritional status Nutrition 0.000 description 1
- GLDOVTGHNKAZLK-UHFFFAOYSA-N octadecan-1-ol Chemical compound CCCCCCCCCCCCCCCCCCO GLDOVTGHNKAZLK-UHFFFAOYSA-N 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 150000003891 oxalate salts Chemical class 0.000 description 1
- 229940066842 petrolatum Drugs 0.000 description 1
- 150000007965 phenolic acids Chemical class 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- 229920001451 polypropylene glycol Polymers 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 235000013772 propylene glycol Nutrition 0.000 description 1
- 108090000623 proteins and genes Proteins 0.000 description 1
- 102000004169 proteins and genes Human genes 0.000 description 1
- 238000010298 pulverizing process Methods 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 238000007670 refining Methods 0.000 description 1
- 230000002441 reversible effect Effects 0.000 description 1
- 210000004706 scrotum Anatomy 0.000 description 1
- 238000002791 soaking Methods 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 239000001509 sodium citrate Substances 0.000 description 1
- NLJMYIDDQXHKNR-UHFFFAOYSA-K sodium citrate Chemical compound O.O.[Na+].[Na+].[Na+].[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O NLJMYIDDQXHKNR-UHFFFAOYSA-K 0.000 description 1
- 238000000638 solvent extraction Methods 0.000 description 1
- 239000001587 sorbitan monostearate Substances 0.000 description 1
- 235000011076 sorbitan monostearate Nutrition 0.000 description 1
- 229940035048 sorbitan monostearate Drugs 0.000 description 1
- 150000003431 steroids Chemical class 0.000 description 1
- 239000004575 stone Substances 0.000 description 1
- 238000007920 subcutaneous administration Methods 0.000 description 1
- 150000003890 succinate salts Chemical class 0.000 description 1
- 239000000375 suspending agent Substances 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 239000003765 sweetening agent Substances 0.000 description 1
- 150000003892 tartrate salts Chemical class 0.000 description 1
- MHXBHWLGRWOABW-UHFFFAOYSA-N tetradecyl octadecanoate Chemical compound CCCCCCCCCCCCCCCCCC(=O)OCCCCCCCCCCCCCC MHXBHWLGRWOABW-UHFFFAOYSA-N 0.000 description 1
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical class CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 1
- YNJBWRMUSHSURL-UHFFFAOYSA-N trichloroacetic acid Chemical compound OC(=O)C(Cl)(Cl)Cl YNJBWRMUSHSURL-UHFFFAOYSA-N 0.000 description 1
- LENZDBCJOHFCAS-UHFFFAOYSA-N tris Chemical compound OCC(N)(CO)CO LENZDBCJOHFCAS-UHFFFAOYSA-N 0.000 description 1
- 230000007306 turnover Effects 0.000 description 1
- 238000002211 ultraviolet spectrum Methods 0.000 description 1
- 230000000007 visual effect Effects 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
- 239000003871 white petrolatum Substances 0.000 description 1
Images
Landscapes
- Medicines Containing Plant Substances (AREA)
Abstract
The present invention relates to compounds of monocyclic polysubstitution saturated cyclohexanones having the formula I and containing a long chain hydrocarbon substituent, an extract containing the compounds, a preparation method and an application of the compounds and the extract. The compounds of monocyclic polysubstitution saturated cyclohexanones of the present invention can be used as cell cycle inhibitors, apoptosis inducers, cell proliferation inhibitors and anti-tumor agents for anti-tumor therapy.
Description
The technical field is as follows:
the invention relates to a monocyclic polysubstituted saturated cyclohexanone compound and a preparation method and application thereof. In particular to a monocyclic polysubstituted saturated cyclohexanone compound containing a long-chain hydrocarbon substituent, an extract containing the compound, a method for extracting, separating and purifying the compound and application of the extract and the compound in preparing a cell cycle inhibitor, an apoptosis inducer, a cell proliferation inhibitor, a cancer cell killing agent and an antitumor agent.
Background art:
monocyclic saturated cyclohexanone compounds refer to a series of compounds having different substituents attached to the carbon skeleton of cyclohexanone. The compound has a single skeleton structure, and different compounds are formed mainly due to different substituent groups and different substituent positions. Among them, some substituted monocyclic saturated cyclohexanones containing a long-chain hydrocarbon substituent have been reported in the literature. Such as the documents S.R. Johns, et al, Campnosperma extensions, the optically active long-chain 5-hydroxythex-2-enes and long-chain bicycles [3.3.1] none-3, 7-diodes: several substituted monocyclic saturated cyclohexanones containing one long-chain hydrocarbon group (seventeen or nineteen carbons) have been described by aust.j.chem., 40, 79-96, 1987. However, all the compounds described in the above documents are artificially transformed products. The compounds which are known so far are not only few in number and limited in substitution types, but also have antitumor activity and have the same substitution type as the compounds of the invention, and monocyclic polysubstituted saturated cyclohexanone compounds containing a long-chain hydrocarbon substituent are not reported so far.
Choerospondias axillaris (Roxb.) Burtt et Hill is a plant of Choerospondias genus of Anacardiaceae family, the bark and fruit of which are used as medicines mainly for treating pyocutaneous disease, injury from hot water, eczema of scrotum, indigestion, abdominal pain due to dyspepsia, sobering up and detoxication of fruit stone, and sore or abscess due to wind-toxicity keloid (edited by Jiangsu New institute of medicine, dictionary of Chinese medicine, Shanghai people's publishing house, 1977, page 397-. The dried fruit has been used as Chinese medicine "Guangzao" and has been imported into pharmacopoeia of 2000 th edition of China (national pharmacopoeia Committee, pharmacopoeia of people's republic of China, one division, Beijing, chemical industry Press, 2000 th year, page 32). Various chemical components such as flavone, phenolic acid, organic acid, amino acid, steroid and the like in choerospondias axillaris have been reported, and various activities such as antibiosis, platelet aggregation inhibition and the like of a choerospondias axillaris crude extract, total flavone or a monomer compound have also been reported (such as bear winterization and the like; the research overview and the application prospect of choerospondias axillaris plants in the aspect of medicines are shown in Guangdong pharmacy, 10 th period and 5 th page in 2000, and 8-10 th page). However, the antineoplastic activity and the active ingredients of the choerospondias axillaris, in particular to the monocyclic polysubstituted saturated cyclohexanone active ingredients related to the invention, are not reported in research so far.
The invention content is as follows:
the invention aims to provide a monocyclic polysubstituted saturated cyclohexanone compound containing a long-chain hydrocarbon substituent, which has the antitumor activities of cell cycle inhibition, cell apoptosis induction, direct killing of cancer cells and the like.
The inventor discovers for the first time that the crude extract of choerospondias axillaris has good antitumor activities such as killing cytotoxicity, apoptosis induction, cell cycle inhibition and inhibition on cancer cell proliferation on cancer cells, researches the active ingredients of the crude extract, and discovers that the monocyclic polysubstituted saturated cyclohexanone compound shown as the following formula I in the crude extract of choerospondias axillaris has the antitumor activity:
formula I
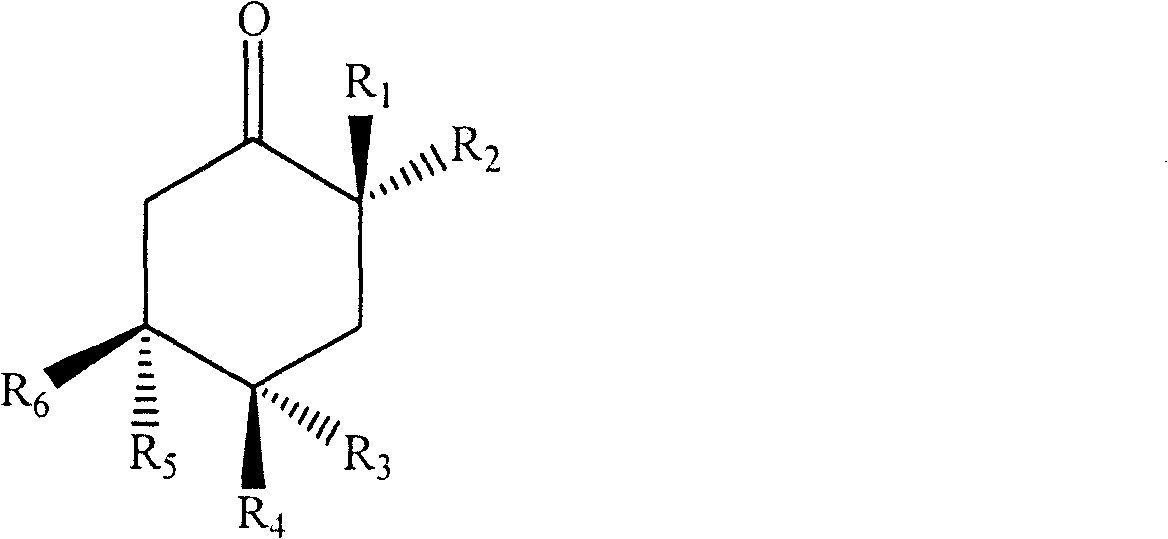
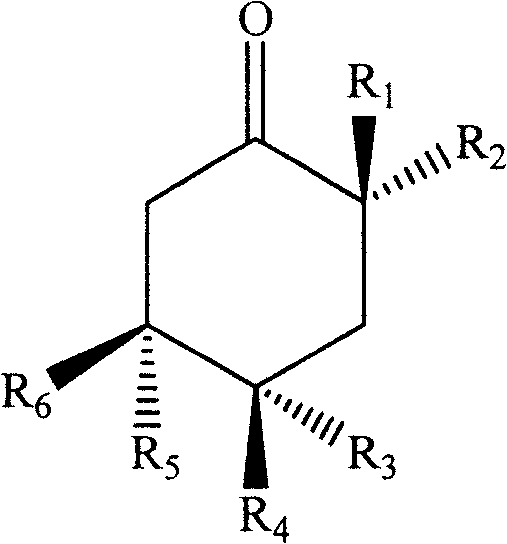
Wherein,
six-membered ring abstraction of Cyclohexanone4C1A chair configuration;
R1、R2、R3、R4、R5、R6is hydrogen, hydroxy, amino, methoxy or saturated or unsaturated straight chain or branched chain C optionally substituted by hydroxy, amino and the like15-20A hydrocarbon group, and, R1-R6At least one of them is said saturated or unsaturated straight chain or branched C optionally substituted with a substituent such as hydroxyl, amino or the like15-C20A hydrocarbyl group and the remaining at least three are oxygen-containing substituents selected from the group consisting of hydroxy or methoxy.
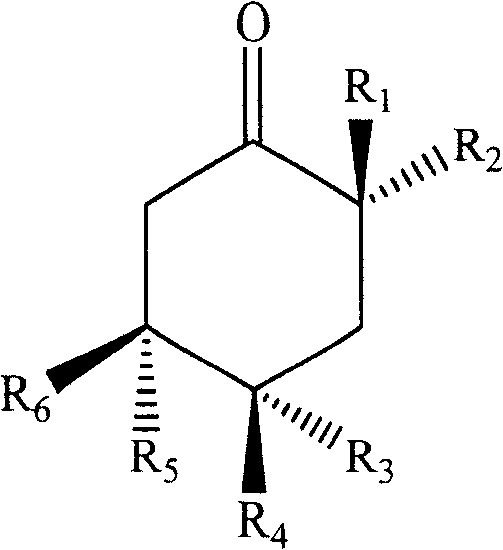
Accordingly, in a first aspect, the present invention relates to an extract characterized by comprising at least one monocyclic polysubstituted saturated cyclohexanone compound of formula I:
formula I
Wherein,
six-membered ring abstraction of Cyclohexanone4C1A chair configuration;
R1、R2、R3、R4、R5、R6is hydrogen, hydroxy, amino, methoxy or saturated or unsaturated straight chain or branched chain C optionally substituted by hydroxy, amino and the like15-20A hydrocarbon group, and, R1-R6At least one of them is said saturated or unsaturated straight chain or branched C optionally substituted with a substituent such as hydroxyl, amino or the like15-C20A hydrocarbyl group and the remaining at least three are oxygen-containing substituents selected from the group consisting of hydroxy or methoxy.
The second aspect of the invention relates to monocyclic polysubstituted saturated cyclohexanone compounds shown in formula I and pharmaceutically acceptable salts thereof:
formula I
Wherein,
six-membered ring abstraction of Cyclohexanone4C1A chair configuration;
R1、R2、R3、R4、R5、R6is hydrogen, hydroxy, amino, methoxy or saturated or unsaturated straight chain or branched chain C optionally substituted by hydroxy, amino and the like15-20A hydrocarbon group, and, R1-R6At least one of them is said saturated or unsaturated straight chain or branched C optionally substituted with a substituent such as hydroxyl, amino or the like15-C20A hydrocarbyl group and the remaining at least three are oxygen-containing substituents selected from the group consisting of hydroxy or methoxy.
A third aspect of the present invention relates to a process for preparing the above extract, which comprises extracting the relevant plant material, such as Choerospondias axillaris, with an alcohol or aqueous alcohol to obtain a crude extract, i.e., the extract.
In a fourth aspect, the invention relates to a process for the preparation of a compound of formula I as defined above, which comprises extracting the relevant plant material, such as choerospondias axillaris, with an alcohol or aqueous alcohol to obtain a crude extract, and then separating and purifying to obtain the compound of formula I.
A fifth aspect of the invention relates to a pharmaceutical composition comprising as active ingredient a monocyclic polysubstituted saturated cyclohexanone compound of formula I together with one or more pharmaceutically acceptable carriers or excipients.
The sixth aspect of the invention relates to the application of the monocyclic polysubstituted saturated cyclohexanone compound in the preparation of cell cycle inhibitors, apoptosis inducers, cell proliferation inhibitors, cancer cell killing agents and antitumor agents.
In one embodiment of the present invention, the present invention relates to monocyclic polysubstituted saturated cyclohexanones of the following formula I:
formula I
Wherein,
six-membered ring abstraction of Cyclohexanone4C1A chair configuration;
R1、R2、R3、R4、R5、R6is hydrogen, hydroxy, amino, methoxy or saturated or unsaturated straight chain or branched chain C optionally substituted by hydroxy, amino and the like15-20A hydrocarbon group, and, R1-R6At least one of them is said saturated or unsaturated straight chain or branched C optionally substituted with a substituent such as hydroxyl, amino or the like15-C20A hydrocarbyl group and the remaining at least three are oxygen-containing substituents selected from the group consisting of hydroxy or methoxy.
In a preferred embodiment of the present invention, the present invention relates to monocyclic polysubstituted saturated cyclohexanones of the following formula I:
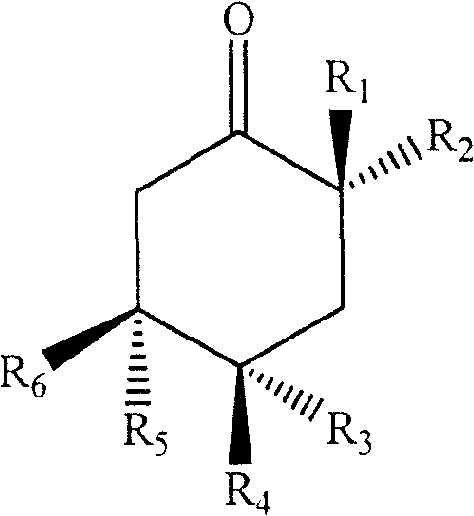
formula I
Wherein,
R1and R2Is hydroxy or (Z) -14-enenonadecyl, R3、R4、R5、R6Is hydroxyl or hydrogen.
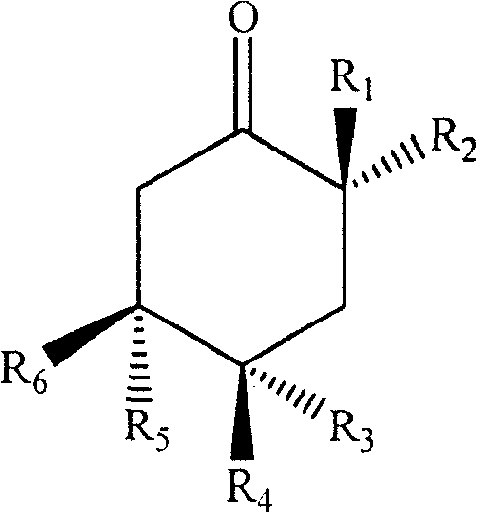
In a further preferred embodiment of the invention, R in the formula I1Is (Z) -14-enenonalkyl, R2、R4、R6Is hydroxy, R3、R5Is hydrogen.
In another further preferred embodiment of the invention, R in formula I2Is (Z) -14-enenonalkyl, R1、R3、R6Is hydroxy, R4、R5Is hydrogen.
Extracts and pure compounds of formula I containing the above compounds of formula I can be prepared by the following method: extracting plant material such as Choerospondias axillaris with alcohol or aqueous alcohol to obtain crude extract, and separating and purifying to obtain compound of formula I.
According to the invention, the alcohol employed in the above process is ethanol; the aqueous alcohol is 60-95% aqueous ethanol; the separation and purification includes conventional methods and means for separation and purification of natural products, such as liquid-liquid extraction, column chromatography, thin layer chromatography, high performance liquid chromatography, recrystallization and the like, which are well known to those skilled in the art. Wherein column chromatography, high performance liquid chromatography and recrystallization refining can be repeatedly carried out for many times.
The invention adopts a method for detecting cell morphological characteristics under a microscope by combining a Lissamine rhodamine B (SRB) method and a flow cytometry, and tests the effects of the compound shown in the formula I on cell proliferation inhibition, cell cycle inhibition, cell apoptosis induction, cancer cell direct killing and the like of mouse breast cancer tsFT210 cells, human colorectal cancer HCT-15 cells, human cervical cancer HeLa cells, human breast cancer MCF-7 cells and human ovarian cancer A2780 cells. Experiments prove that the compound shown in the formula I shows the biological activity of inhibiting the proliferation of tumor cells by inhibiting the cell cycle turnover, inducing the apoptosis of cancer cells or directly killing the cancer cells and the like, thereby having the anti-tumor effect.
The compound of the formula I can be prepared into antitumor drugs by being compatible with various pharmaceutically acceptable carriers, excipients or auxiliary materials, and is used for treating tumors.
The term "pharmaceutically acceptable salt" in the present invention may be a pharmaceutically acceptable inorganic or organic salt. The compounds having a basic group in formula I of the present invention may form pharmaceutically acceptable salts with inorganic acids, such as sulfate, hydrochloride, hydrobromide, phosphate; pharmaceutically acceptable salts can also be formed with organic acids such as acetates, oxalates, citrates, gluconates, succinates, tartrates, p-toluenesulfonates, methanesulfonates, benzoates, lactates, maleates, and the like. The compounds having an acidic group in formula I of the present invention may form pharmaceutically acceptable salts with alkali metals or alkaline earth metals, preferably but not limited to sodium, potassium, magnesium or calcium salts.
The compounds of the present invention may be administered alone or in the form of pharmaceutical compositions. The route of administration may be oral, parenteral or topical. The pharmaceutical composition can be formulated into various suitable dosage forms according to the administration route.
Pharmaceutical compositions of the compounds of the present invention may be administered in any of the following ways: oral, aerosol inhalation, rectal, nasal, buccal, topical, parenteral, e.g. subcutaneous, intravenous, intramuscular, intraperitoneal, intrathecal, intraventricular, intrasternal and intracranial injection or infusion, or via an external reservoir. Among them, oral, intraperitoneal or intravenous administration is preferable.
When administered orally, the compounds of the present invention may be formulated in any orally acceptable dosage form, including but not limited to tablets, capsules, aqueous solutions or suspensions. Among these, carriers for tablets generally include lactose and corn starch, and additionally, lubricating agents such as magnesium stearate may be added. Diluents used in capsule formulations generally include lactose and dried corn starch. Aqueous suspension formulations are generally prepared by mixing the active ingredient with suitable emulsifying and suspending agents. Optionally, some sweetener, aromatic or colorant may be added into the above oral preparation.
When applied topically to the skin, the compounds of the present invention may be formulated in a suitable ointment, lotion, or cream formulation wherein the active ingredient is suspended or dissolved in one or more carriers. Carriers that may be used in ointment formulations include, but are not limited to: mineral oil, liquid petrolatum, white petrolatum, propylene glycol, polyethylene oxide, polypropylene oxide, emulsifying wax and water; carriers that can be used in lotions or creams include, but are not limited to: mineral oil, sorbitan monostearate, tween 60, cetyl esters wax, cetearyl alcohol, 2-octyldodecanol, benzyl alcohol and water.
The compounds of the present invention may also be administered in the form of sterile injectable preparations, including sterile injectable aqueous or oleaginous suspensions or solutions. Among the carriers and solvents that may be employed are water, ringer's solution and isotonic sodium chloride solution. In addition, the sterilized fixed oil may also be employed as a solvent or suspending medium, such as a monoglyceride or diglyceride.
It is further noted that the dosage and method of administration of the compounds of the present invention will depend upon a variety of factors including the age, weight, sex, physical condition, nutritional status, the activity level of the compound, time of administration, metabolic rate, severity of the condition, and the subjective judgment of the treating physician. The preferred dosage is between 0.01-100mg/kg body weight/day.
The compound of formula I of the invention can also be used as a low molecular biological probe for inhibiting cell cycle or inducing apoptosis in life science research. When the compound of formula I is used as cell cycle inhibitor or apoptosis inducer in life science research, it can be dissolved in methanol or aqueous methanol, or dissolved in aqueous solution of dimethyl sulfoxide for application.
Description of the drawings:
FIG. 1 is a UV absorption spectrum of Compound I in methanol solution (40. mu.g/ml);
FIG. 2 is an infrared absorption spectrum (KBr) of Compound I;
FIG. 3 is of Compound I in deuterated chloroform1H nuclear magnetic resonance spectrum;
FIG. 4 is of Compound I in deuterated chloroform13C nuclear magnetic resonance spectrum;
FIG. 5 is a Circular Dichroism (CD) spectrum of Compound I in methanol solution (1.0 mg/ml);
FIG. 6 is a UV absorption spectrum of Compound II in methanol solution (40. mu.g/ml);
FIG. 7 is an infrared absorption spectrum (KBr) of Compound II;
FIG. 8 is of compound II in deuterated methanol1H nuclear magnetic resonance spectrum;
FIG. 9 is of compound II in deuterated methanol13C nuclear magnetic resonance spectrum;
FIG. 10 is a Circular Dichroism (CD) spectrum of Compound II in methanol solution (1.0 mg/ml);
FIG. 11 is a flow cytometric histogram of mouse breast cancer tsFT210 cells measured 17 hours after treatment with Compound I;
figure 12 is a flow cytometric histogram of mouse breast cancer tsFT210 cells measured 17 hours after treatment with compound II.
The specific implementation mode is as follows:
the following examples further illustrate the invention but are not intended to limit the invention thereto.
In the following examples, compounds of the present invention, hereinafter referred to as compound I, were identified by nuclear magnetic resonance or the like to have the following structures: a compound of formula I wherein the six-membered ring is taken4C1Configuration (absolute configuration 3S 4R 6R), R1Is (Z) -14-enenonalkyl, R2、R4、R6Is hydroxy, R3、R5Is hydrogen; the compound of the present invention, hereinafter referred to as compound II, has the following structure as identified by nuclear magnetic resonance or the like: a compound of formula I wherein the six-membered ring is taken4C1Configuration (absolute configuration 3S 4S 6S), wherein R2Is (Z) -14-enenonalkyl, R1、R3、R6Is hydroxy, R4、R5Is hydrogen:
formula I
The Arabic numbers on the carbocycle and the long chain indicate the index positions of the corresponding carbon atoms.
In the structural study of the compound, the melting point is measured by an X-4 type precision micro melting point tester of Beijing Tian Di Yu science and technology Limited liability company, and the temperature is not corrected. The specific rotation was measured by a JSASCO P-1020 polarimeter. Positive and negative ion TOF-MS and HR-TOF-MS were measured with an API 3000 liquid chromatography-mass spectrometer (ABI, USA) and an LCT mass spectrometer (Micromass, UK), respectively, an ultraviolet spectrum was measured with a Shimadzu UV2501PC ultraviolet spectrophotometer, and an infrared spectrum was measured with a Nicolet Magna-IR spectrometerTM550 type infrared spectrometer determination, nuclear magnetic resonanceSpectrum JEOLECllips-600 type superconducting nuclear magnetic resonance apparatus (600 MHz)1H-NMR,150MHz 13C-NMR). Circular Dichroism (CD) spectra were measured using JASCO J-810 Spectropolarimeter.
EXAMPLE 1 extraction and isolation of Compound I
The first step is as follows: preparation of extract containing Compound I
Pulverizing dried bark of Choerospondias axillaris (3.2kg), and soaking in ethanol at 25L room temperature for 7 days for 4 times. Mixing extractive solutions, concentrating, and drying to obtain 750g ethanol extract. 750g of the ethanol extract was suspended in 3L of water, and the mixture was extracted 4 times with chloroform (3L), ethyl acetate (3L) and n-butanol (3L) in this order to obtain 60g of the chloroform extract, 310g of the ethyl acetate extract, 300g of the n-butanol extract and 80g of the water layer residue, respectively. Wherein the chloroform extract is an extract containing compound I.
The second step is as follows: preparation of a chromatographic crude fraction containing Compound I
The chloroform extract (60g) was dried on a reduced pressure silica gel column (bed 7.5 cm. times.18.5 cm) and eluted with petroleum ether (P) -acetone (A) and methanol solvent system. The polarity of the elution solvent is increased by increasing the amount of acetone (A) in petroleum ether (P) to increase the polarity of the elution solvent in a gradient manner or by changing the amount of acetone (A) to methanol. Fractions were pooled according to TLC and Activity test to give 6 fractions Fr-1(3.2g, V)P∶VA100: 1 elution fraction), Fr-2(2.3g, V)P∶VA50: 1 elution fraction), Fr-3(2.0g, VP∶VA10: 1 elution fraction), Fr-4(10.8g, V)P∶VA5: 1 elution fraction), Fr-5(13.6g, V)P∶VA2: 1 elution fraction) and Fr-6(25g, methanol elution fraction). Subjecting Fr-5(13.6g) to dry vacuum silica gel column (5.0cm × 22cm), eluting with chloroform (C) -ethyl acetate (Ac), acetone, and methanol system, and combining the fractions according to thin layer detection and activity test to obtain 6 components Fr-5-1(4.1g, V)C∶VAc8: 1 elution fraction), Fr-5-2(980mg, V)C∶VAc4: 1 elution fraction), Fr-5-3(3.7g, VC∶VAc2: 1 elution fraction), Fr-5-4(1.1g, ethyl acetate elution fraction), Fr-5-5(1.8g, acetone elution fraction), Fr-5-6(0.8g, methanol elution fraction). Wherein Fr-5-3 is a chromatography crude component containing the compound I.
The third step: purification and purification of Compound I
Fr-5-3(3.7g) was applied to SephadexLH-20 column (bed 3.8 cm. times.36 cm) and eluted with chloroform-methanol (1: 1) to give a chromatography fraction mainly containing Compound I. This fraction was subjected to RP-18 reverse phase analytical high performance liquid chromatography (203nm detection) and when eluted with methanol gave an eluted fraction containing Compound I at a retention time of 8 minutes. Separating by RP-18 reversed-phase preparative high performance liquid chromatography (203nm detection) under the same conditions, eluting with methanol, and collecting corresponding eluate to obtain crude product of compound I. This crude compound I gave a single elution peak of compound I at retention time of 72 minutes in RP-18 reversed phase analytical HPLC (203nm detection) eluting with 80% methanol. Separating by RP-18 reversed phase preparative high performance liquid chromatography (203nm detection) under the same conditions, eluting with 80% methanol, collecting corresponding eluate, concentrating, and recrystallizing in chloroform-methanol to obtain pure compound I104 mg.
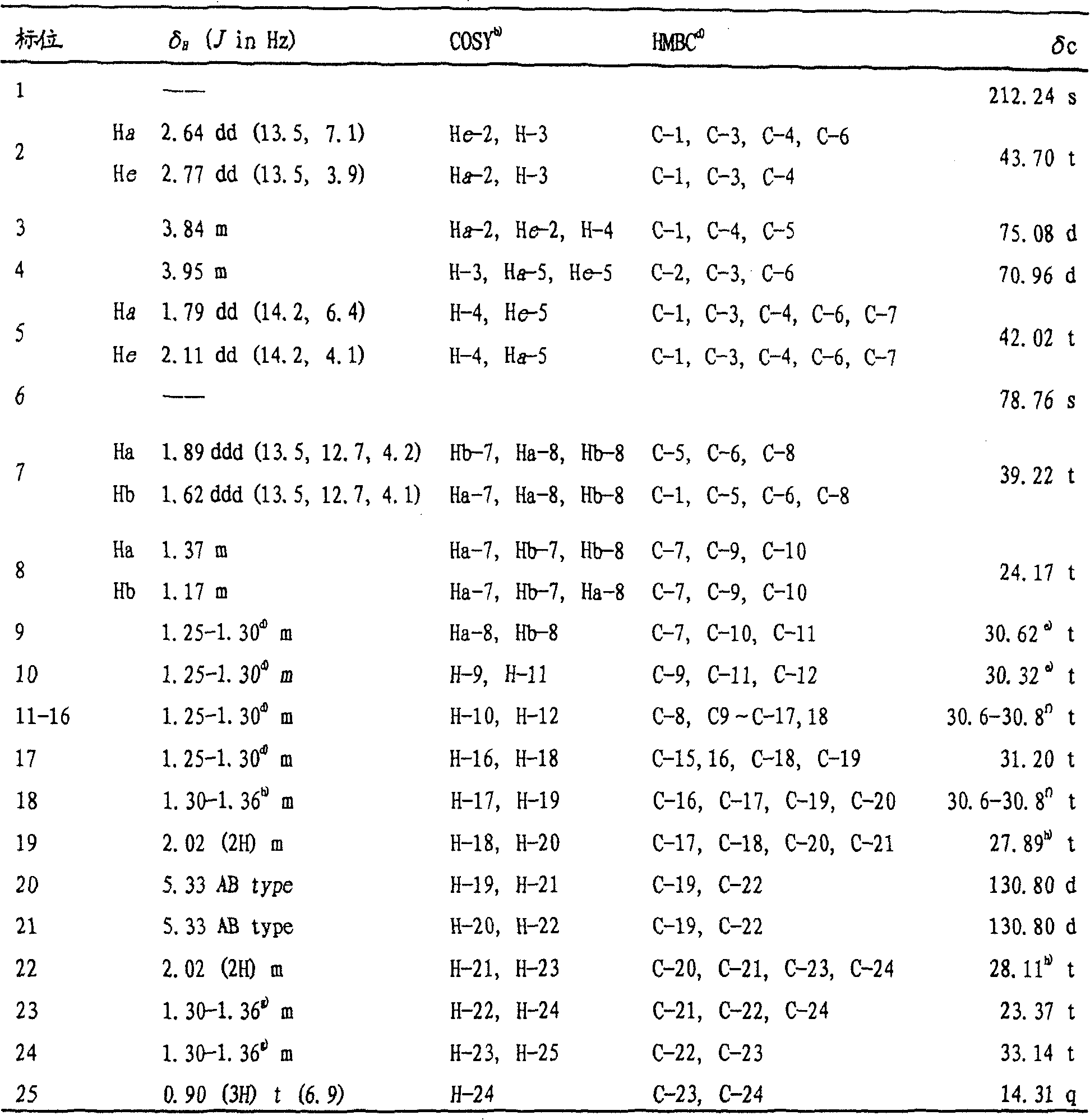
White crystalline powder of compound I with melting point of 75.1-76.2 deg.C, [ alpha ]]D 31+37.7°(c1.0,CHCl3) Molecular formula C25H46O4. Positive ion TOF-MSm/z: 428[ M + NH ]4]+,411[M+H]+,393[M+H-H2O]+,375[M+H-2H2O]+(ii) a Negative ion TOF-MS m/z: 455[ M-H + HCOOH]-,445[M+Cl]-,391[M-H-H2O]-(ii) a Positive ion HR-TOF-MSm/z: found 411.3475[ M + H]+Calculated value 411.3474([ M + H)]+)。UVλmaxnm (log ε) in MeOH: 201(3.64, end absorption). IR (KBr) vmax cm-1:3394,3282(OH),2920,2850(CH3&CH2),1718(CO),1465,1332,1166(C-O),1070,1004,651。CDλmax nm(mdeg)in MeOH at 1.0mg/ml:317(-21.3715),283.7(+58.3594),206.6(-30.5037),202.6(-1.16184)。1H and13the C NMR data are shown in Table 1.
TABLE 1 600MHz of Compound I in deuterated chloroform1H and 400MHz13C nuclear magnetic resonance dataa)
a) The method comprises the following steps Signals in the table are according to DEPT, PFG1H-1And attributing the analysis results of the difference spectra of H COSY, PFG HMQC, PFG HMBC and NOE. b) The method comprises the following steps The symbols in this column are represented in PFG1H-1The hydrogen nuclei in the H COSY spectrum, which are in the same line position with the hydrogen nuclei in the same line position, give coupling-related signals. c) The method comprises the following steps The symbols in this column are represented in the PFG HMBC spectra (A), (B), (C)1r JCHEither 8.3 or 4Hz) to the hydrogen nucleus in the same row index position gives the carbon atom of the HMBC coupling related signal. d) E), f): the signal cannot be attributed exactly because it overlaps with other signals. h) I): the signal assignments between two signals with the same superscript are interchangeable.
NOE difference spectrum test analysis of compound I: when Ha-2 was irradiated, NOE signals were observed on He-2, Ha-7, and Hb-7, respectively; when H-3 is irradiated, NOE signals are respectively observed on He-2, H-4, Ha-5, He-5 and 3-OH; when Ha-5 was irradiated, NOE signals were observed on H-3, H-4, He-5, respectively; when Ha-7 was irradiated, NOE signals were observed on Ha-2, He-5, Hb-7, Hb-8 and H-9, respectively; when He-7 was irradiated, a NOE signal was observed on Ha-7.
EXAMPLE 2 extraction and isolation of Compound II
The first step is as follows: preparation of extract containing Compound II
Referring to the first step of example 1, the same procedure was followed to obtain 110 g of chloroform extract, the main extract containing compound II, from 6 kg of choerospondias axillaris raw material.
The second step is as follows: preparation of a chromatographic crude fraction containing Compound II
60g of chloroform extract containing compound II was applied to a vacuum silica gel column (column bed 7.5 cm. times.18.5 cm) by dry method, and eluted with petroleum ether (P) -acetone (A) and methanol solvent system. The polarity of the elution solvent is increased by increasing the amount of acetone (A) in petroleum ether (P) to increase the polarity of the elution solvent in a gradient manner or by changing the amount of acetone (A) to methanol. Fractions were pooled according to thin layer assay and activity assay to give 6 fractions Fr-1(3.3g, V)P∶VA100: 1 elution fraction), Fr-2(2.1g, V)P∶VA50: 1 elution fraction), Fr-3(2.2g, VP∶VA10: 1 elution fraction), Fr-4(11.0g, VP∶VA5: 1 elution fraction), Fr-5(14.0g, VP∶VA2: 1 elution fraction) and Fr-6(24.5g, methanol elution fraction). Subjecting Fr-5(14.0g) to dry-process under reduced pressure on silica gel column (bed 5.0cm × 22cm), eluting with chloroform (C) -ethyl acetate (Ac), acetone, and methanol system, and combining the fractions according to thin layer detection and activity test to obtain 6 components Fr-5-1(4.3g, V)C∶VAc8: 1 elution fraction), Fr-5-2(1.0g, V)C∶VAc4: 1 elution fraction), Fr-5-3(3.8g, VC∶VAc2: 1 elution fraction), Fr-5-4(1.0g, ethyl acetate elution fraction), Fr-5-5(1.9g, acetone elution fraction), Fr-5-6(0.9g, methanol elution fraction). Wherein Fr-5-5 is a chromatography crude component containing a compound II.
The third step: purification and purification of Compound II
Fr-5-5(1.9g) was dissolved in an appropriate amount of chloroform-methanol (1: 1), and the resulting solution was applied to a SephadexLH-20 column (column bed 3.8 cm. times.36 cm), eluted with chloroform-methanol (1: 1) to give fractions Fr-5-5-1(0.8g), Fr-5-5-2(0.4g) and Fr-5-5-3(0.7g) in this order. Compound II is mainly concentrated in Fr-5-5-2, and thus Fr-5-5-2 was isolated by preparative silica gel thin layer chromatography (chloroform-methanol 7: 1 development) to give a crude product of Compound II (230 mg). In RP-18 reversed-phase analysis high performance liquid chromatography (203nm detection), when 83% methanol is used for elution, a single elution peak of the compound II is given at the position of 59 minutes of retention time, amplification experiment separation and refinement are carried out under the same conditions, RP-18 reversed-phase preparation high performance liquid chromatography (203nm detection) separation is carried out, corresponding elution components are collected, concentration is carried out, and the pure product of the compound II is obtained by recrystallization operation in methanol, namely 135 mg.
Compound II white crystal powder with melting point of 92-94 deg.C, [ alpha ]]D 31+31.4°(c1.0,CHCl3) Molecular formula C25H46O4. Positive ion TOF-MSm/z: 428[ M + NH ]4]+,411[M+H]+,393[M+H-H2O]+,375[M+H-H2O]+(ii) a Negative ion TOF-MSm/z: 469[ M-H + CH3COOH]-,455[M-H+HCOOH]-,445[M+Cl]-,391[M-H-H2O]-(ii) a Positive ion HR-TOF-MSm/z: found 411.3487[ M + H]+Calculated value 411.3474([ M + H)]+)。UVλmaxnm (log ε) in MeOH: 200(3.69, end absorption). IR (KBr) vmaxcm-1:3383(OH),2922,2850(CH3&CH2),1726(CO),1465,1457,1366,1080(C-O),699,644,621。CDλmaxnm(mdeg)in MeOHat 1.0mg/ml:319.5(-16.6417),284.4(+107.044),210.6(+9.10091),204.8(+30.7266)。1H and13the C NMR data are shown in Table 2.
TABLE 2 600MHz of Compound II in deuterated methanol1H and 400MHz13C nuclear magnetic resonance dataa)
a) The method comprises the following steps Signals in the table are according to DEPT, PFG1H-1And attributing the analysis results of the difference spectra of H COSY, PFG HMQC, PFG HMBC and NOE. b) The method comprises the following steps The symbols in this column are represented in PFG1H-1H COSY spectrum neutralizationThe hydrogen nuclei in the same row index position give the hydrogen nuclei for coupling-related signals. c) The method comprises the following steps The symbols in this column are represented in the PFG HMBC spectra (A), (B), (C)1r JCHEither 8.3 or 4Hz) to the hydrogen nucleus in the same row index position gives the carbon atom of the HMBC coupling related signal. d) F), g): the signal cannot be attributed exactly because it overlaps with other signals. e) I): the signal assignments between two signals with the same superscript are interchangeable.
NOE difference spectrum test analysis of compound II: when He-2 was irradiated, NOE signals were observed on Ha-2 and H-3, respectively; when H-3 was irradiated, NOE signals were observed on He-2 and Ha-5, respectively; NOE was observed on Ha-2 and He-5, respectively, when H-4 was irradiated; when Ha-5 was irradiated, NOE signals were observed on H-3, He-5, Ha-7 and Hb-7, respectively; when He-5 was irradiated, NOE signals were observed on H-4, Ha-5, Ha-7, respectively; when Ha-7 was irradiated, NOE signals were observed on He-5, Hb-7, Ha-8, and H-9, respectively; when He-7 was irradiated, NOE signals were observed on Ha-5, Ha-7, Ha-8, H-9.
Example 3 bioactivity test
Experimental sample and experimental method
Preparing a solution of a sample to be detected: the compound I isolated and purified in example 1 and the compound II isolated and purified in example 2 were each separately weighed out in an appropriate amount and prepared into a solution of a desired concentration with methanol for measurement of activity.
Cell lines and cell culture: the activity test adopts a mouse breast cancer tsFT210 cell line, a human colorectal cancer HCT-15 cell line, a human cervical cancer HeLa cell line, a human breast cancer MCF-7 cell line and a human ovarian cancer A2780 cell line.
The cells were subcultured in RPMI-1640 medium containing 10% FBS at 32 deg.C (tsFT210 cells) or 37 deg.C (HCT-15, HeLa, MCF-7 and A2780 cells) in an incubator with 5% carbon dioxide.
Cell proliferation inhibitory activity test method (lissamine rhodamine B method, SRB method): collecting human carcinoma of large intestine HCT-15 cells and human in logarithmic growth phasePreparing cervical cancer HeLa cells, human breast cancer MCF-7 cells and human ovarian cancer A2780 cells into 2 × 10 per ml of fresh RPMI-1640 culture medium5The cell suspension of each cell was inoculated into a 96-well plate at 200. mu.l per well, and 2. mu.l of each sample or blank solution at different concentrations was added thereto, followed by incubation at 37 ℃ for 24 hours. The cells cultured under the action of the medicine are taken, and the morphological change caused by the medicine treatment is observed under an optical microscope to judge whether the morphological characteristics of cell cycle inhibition, cell apoptosis or cell necrosis exist. The supernatant was then aspirated and 50. mu.l of 20% trichloroacetic acid was added to each well of cells, fixed at 4 ℃ for 1 hour, rinsed 5 times with water and air dried. 50 microliters of 0.4% SRB in acetic acid was added to each well and allowed to stand at room temperature for 30 minutes. Unbound free SRB dye was removed by washing 4 times with 1% acetic acid water. 150 μ L of Tris buffer (10mmol/L, pH 10.5) was added to each well to dissolve the protein-binding dye and the Optical Density (OD) at 520nm of each well was measured using a SPECTRA MAX Pus type microplate reader manufactured by MD. Three wells were placed for each concentration of sample in the same 96 well plate, three additional well blanks were placed, and the mean OD values in IR% (OD)Blank control-ODSample (A) Article (A))/ODBlank controlX 100% formula the inhibition of cell proliferation (IR%) was calculated at each concentration.
Flow cytometry test methods: taking tsFT210 cells in logarithmic growth phase, preparing the cells into a density of 2 x 10 per ml by using fresh RPMI-1640 medium5The cell suspension of each cell was inoculated into a 24-well plate at 0.5 ml per well, and 5. mu.l of each sample solution was added thereto, followed by incubation at 32 ℃ for 17 hours. The cells cultured under the action of the drug are taken, the morphological change caused by the drug treatment is observed under an optical microscope, the morphological characteristics of the cells such as cell cycle inhibition, cell apoptosis or cell necrosis are judged, and if necessary, the cells are photographed. The cells were then transferred from the 24-well plates to 1.5 ml Eppendorf centrifuge tubes, centrifuged at 3000 rpm at 4 ℃ for 3 minutes, the supernatant was aspirated, washed once with 0.5 ml Phosphate Buffered Saline (PBS) and centrifuged under the same conditions, and 150. mu.l of an aqueous solution of Propidium Iodide (PI) (5 mg in 100 ml water) was addedPI, 100mg sodium citrate and 200 mg NP-40), stained at 4 ℃ for 30 minutes, diluted with 150. mu.L PBS, and analyzed by flow cytometry to determine the DNA content distribution in the cells.
Results of the experiment
Inhibitory Activity of Compound I and Compound II on human cancer cell proliferation
In the SRB method test, the compound I and the compound II both show certain inhibitory activity to the cell proliferation of human colorectal cancer HCT-15 cells, and the test results of the inhibitory activity of different concentrations of the compound I and the compound II to the cell proliferation of human colorectal cancer HCT-15 cells are shown in Table 3.
TABLE 3 SRB assay results of compound I and compound II for inhibition of HCT-15 cell proliferation in human colorectal cancer
In the SRB method test, the compound I also shows inhibitory activity of different degrees on the proliferation of human cervical cancer HeLa cells, human ovarian cancer A2780 cells and human breast cancer MCF-7 cells, and the test results of the inhibitory activity of different concentrations of the compound I on the proliferation of the human cancer cells are shown in Table 4.
TABLE 4 SRB assay results for inhibition of human cancer cell proliferation by Compound I
The results in tables 3 and 4 show that both compound I and compound II show varying degrees of anticancer activity against the cancer cells tested in the SRB assay.
Flow cytometry results
Flow cytometry detection analysis results: tsFT210 cells were treated with different concentrations of compound I and compound II and assayed by flow cytometry. The results show that: compound I predominantly exhibits cell cycle inhibitory activity at concentrations ranging from 3.1 micrograms per ml to 12.5 micrograms per ml, inhibiting the cell cycle of tsFT210 cells at stage G0/G1; when the concentration exceeds 12.5 micrograms per milliliter, the apoptosis inducing activity is mainly presented, obvious apoptosis peaks are detected in a sub-G0/G1 region, and the apoptosis peaks are more obvious along with the increase of the concentration; when the concentration is above 50. mu.g per ml, some necrotic cytotoxic activity begins to be detected. Compound II inhibits the cell cycle of tsFT210 cells mainly in the G0/G1 phase in the concentration range of 6.2 micrograms per milliliter to 25 micrograms per milliliter, shows weak G2/M phase inhibition activity at the same time above 25 micrograms per milliliter, starts to show certain S phase inhibition activity at the concentration of above 50 micrograms per milliliter, and shows necrotic cytotoxic activity at the concentration of 100 micrograms per milliliter or above. The flow cytometry analysis and detection results are basically consistent with the morphological detection results observed under the microscope.
And (3) morphological detection results: observed under an optical inverted microscope, tsFT210 cells begin to show morphological features of apoptotic cells when treated with 12.5 micrograms of compound I and 25 micrograms of compound II per ml, and when treated with more than 25 micrograms of compound I per ml, the vast majority of the field shows the morphology typical of apoptotic cells, i.e. snowflake-shaped cells or membrane-bound cell debris; while the number of apoptotic cells in the visual field was increased by treatment with 50. mu.g or more of Compound II, the apoptosis-inducing activity of Compound II was significantly weaker than that of Compound I. When treated with more than 50 micrograms per milliliter of compound I and compound II, a portion of the cancer cells began to exhibit varying degrees of morphological characteristics typical of necrotic cells, and as the concentrations of compounds I and II increased, the necrotic cytotoxic activity also increased, indicating that compound I at high concentrations had some direct cytotoxic activity against mammalian cancer cells.
Conclusion
The experimental results show that the compound I and the compound II can play the anti-tumor role of inhibiting cancer cell proliferation by inhibiting cell cycle, inducing cancer cell apoptosis or directly killing cytotoxic activity to the cancer cells. Therefore, the compound of the present invention can be used as an antitumor agent for treating tumors, and can also be used as a low molecular probe for inducing apoptosis or inhibiting cell cycle for life science research for exploring the essence of life phenomenon.
Claims (8)
1. An extract characterized by containing at least one compound of the following formula I:
formula I
Wherein,
six-membered ring abstraction of Cyclohexanone4C1A chair configuration;
R1is (Z) -14-enenonalkyl, R2、R4、R6Is hydroxy, and R3、R5Is hydrogen, or
R2Is (Z) -14-enenonalkyl, R1、R3、R6Is hydroxy, and R4、R5Is hydrogen.
4. A pharmaceutical composition comprising as active ingredient a compound according to claim 2 or 3 together with one or more pharmaceutically acceptable carriers or excipients.
5. A method for preparing the extract according to claim 1, which comprises extracting Choerospondias axillaris with an alcohol or aqueous alcohol to obtain the extract.
6. A process for the preparation of a compound of formula I according to claim 2 or 3, which comprises leaching choerospondias axillaris with an alcohol or aqueous alcohol to obtain a crude extract, and separating and purifying to obtain the compound of formula I.
7. Use of an extract according to claim 1, or a compound of formula I according to claim 2 or 3, for the preparation of a cell cycle inhibitor, an apoptosis-inducing agent, an inhibitor of tumor cell proliferation or a tumor cell killing agent.
8. Use of an extract as claimed in claim 1, or a compound of formula I as claimed in claim 2 or 3, for the preparation of an antitumor medicament.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CNB2005100748051A CN100434419C (en) | 2005-06-03 | 2005-06-03 | Compound of monocyclic polysubstitution saturated cyclohexanones, prepartion method and usage |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CNB2005100748051A CN100434419C (en) | 2005-06-03 | 2005-06-03 | Compound of monocyclic polysubstitution saturated cyclohexanones, prepartion method and usage |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN1872838A CN1872838A (en) | 2006-12-06 |
| CN100434419C true CN100434419C (en) | 2008-11-19 |
Family
ID=37483460
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CNB2005100748051A Expired - Fee Related CN100434419C (en) | 2005-06-03 | 2005-06-03 | Compound of monocyclic polysubstitution saturated cyclohexanones, prepartion method and usage |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN100434419C (en) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10493072B2 (en) | 2014-10-24 | 2019-12-03 | Landos Biopharma, Inc. | Lanthionine synthetase C-like 2-based therapeutics |
| US11117881B2 (en) | 2019-12-20 | 2021-09-14 | Landos Biopharma, Inc. | Lanthionine c-like protein 2 ligands, cells prepared therewith, and therapies using same |
| US11197891B2 (en) | 2017-11-30 | 2021-12-14 | Landos Biopharma, Inc. | Therapies with lanthionine C-like protein 2 ligands and cells prepared therewith |
-
2005
- 2005-06-03 CN CNB2005100748051A patent/CN100434419C/en not_active Expired - Fee Related
Non-Patent Citations (4)
| Title |
|---|
| 南酸枣植物在药物方面的研究概况及其应用前景. 熊冬生,浦跃武,吴晓英.广东药学,第5期. 2000 |
| 南酸枣植物在药物方面的研究概况及其应用前景. 熊冬生,浦跃武,吴晓英.广东药学,第5期. 2000 * |
| 姜黄中所含香草环己酮提取物的抗变态反应活性(1). 史青.国外医学.中医中药分册,第6期. 2001 |
| 姜黄中所含香草环己酮提取物的抗变态反应活性(1). 史青.国外医学.中医中药分册,第6期. 2001 * |
Cited By (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10493072B2 (en) | 2014-10-24 | 2019-12-03 | Landos Biopharma, Inc. | Lanthionine synthetase C-like 2-based therapeutics |
| US10682349B2 (en) | 2014-10-24 | 2020-06-16 | Landos Biopharma, Inc. | Lanthionine synthetase C-like 2-based therapeutics |
| US10849895B2 (en) | 2014-10-24 | 2020-12-01 | Landos Biopharma, Inc. | Lanthionine synthetase C-like 2-based therapeutics |
| US11571419B2 (en) | 2014-10-24 | 2023-02-07 | Landos Biopharma, Inc. | Lanthionine synthetase C-like 2-based therapeutics |
| US12502388B2 (en) | 2014-10-24 | 2025-12-23 | Nimmune Biopharma, Inc. | Lanthionine synthetase C-like 2-based therapeutics |
| US11197891B2 (en) | 2017-11-30 | 2021-12-14 | Landos Biopharma, Inc. | Therapies with lanthionine C-like protein 2 ligands and cells prepared therewith |
| US11117881B2 (en) | 2019-12-20 | 2021-09-14 | Landos Biopharma, Inc. | Lanthionine c-like protein 2 ligands, cells prepared therewith, and therapies using same |
| US11377437B2 (en) | 2019-12-20 | 2022-07-05 | Landos Biopharma, Inc. | Lanthionine C-like protein 2 ligands, cells prepared therewith, and therapies using same |
| US12145920B2 (en) | 2019-12-20 | 2024-11-19 | Nimmune Biopharma, Inc. | Lanthionine c-like protein 2 ligands, cells prepared therewith, and therapies using same |
Also Published As
| Publication number | Publication date |
|---|---|
| CN1872838A (en) | 2006-12-06 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN107986951B (en) | Novel topoisomerase I inhibitor and pharmaceutical composition, preparation method and application thereof | |
| CN102516344A (en) | Compound with antitumor activity and preparation method and application thereof | |
| CN108689851B (en) | Tiglic alkane type diterpene compound and preparation method and application thereof | |
| CN115385924B (en) | A kind of cyclopentane benzofuran compound with anti-tumor activity and its application | |
| CN113101293B (en) | Application of ursolic acid derivative in preparing medicine for treating nervous system diseases | |
| He et al. | Highly oxygenated guaiane-type sesquiterpene lactones from Artemisia sacrorum and their antihepatoma activity | |
| CN100434419C (en) | Compound of monocyclic polysubstitution saturated cyclohexanones, prepartion method and usage | |
| CN114533719A (en) | Application of abietane diterpenoid compound in preparation of anti-inflammatory drugs | |
| CN102020649B (en) | Diketopiperazine compound as well as composition, preparation method and application thereof | |
| CN1872828B (en) | Monocyclic polysubstitution cyclohexenol, and compound of ketones, and preparation method, and usage | |
| CN107746421B (en) | Compound DICTYOPTERISIN F and its application in the preparation of antitumor drugs | |
| CN111548327A (en) | Norcarb-kaurine-type diterpenes and their preparation method and use in the preparation of antitumor drugs | |
| CN111808088B (en) | Compounds tersaphilone B and E, preparation method thereof and application thereof in preparing antitumor drugs | |
| CN107722096A (en) | A kind of steroid natural drug with antitumor action and its production and use | |
| CN117756621B (en) | New pentacyclic triterpenoid compounds and their extraction methods and uses | |
| CN100582110C (en) | Cyclohexanone saturated tricyclic (bridged) compound, preparation method and use thereof | |
| CN117586214B (en) | Linderane-type sesquiterpene dimer and preparation method and use thereof | |
| CN102618448B (en) | Drimane-type sesquialter terpene cyclohexenone derivative, preparation method thereof and application | |
| CN100347163C (en) | Cyclohexenone type bicyclo (condensed ring) compound and preparation method and application thereof | |
| CN111808155B (en) | Coumarin compounds of ginseng and their application in medicine | |
| CN119080724B (en) | Sesquiterpenoid compounds from Lycium bark and their preparation method and application | |
| JP4792596B2 (en) | Ishizu orchid extract and its preparation method and use | |
| CN116003371B (en) | Terpenoid, and extraction method and application thereof | |
| CN116789680B (en) | Daphnane-type macrocyclic diterpenoid compound with 4, 7-oxygen bridge structure, and preparation method and application thereof | |
| CN116283535B (en) | Sesquiterpenoids in daphne regale, and preparation method and application thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| CF01 | Termination of patent right due to non-payment of annual fee |
Granted publication date: 20081119 Termination date: 20150603 |
|
| EXPY | Termination of patent right or utility model |