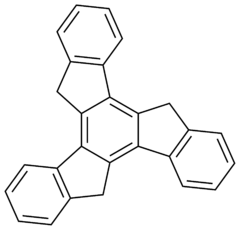
Truxene
| |
| Names | |
|---|---|
| Preferred IUPAC name
10,15-dihydro-5H-diindeno-[1,2-a:1',2'-c]fluorene | |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.008.132 |
| EC Number |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C27H18 | |
| Molar mass | 342.441 g·mol−1 |
| Density | 1.286 g/cm3 |
| Melting point | 378 °C (712 °F; 651 K) [1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Truxene is a polycyclic aromatic hydrocarbon. The molecule can be thought of as being made up of three fluorene units arranged symmetrically and sharing a common central benzene. Truxene is solid, and it is slightly soluble in water.
History
[edit]Truxene has been known since the end of the 19th century. J. Hausmann came across it in 1889 while investigating the reactions of 3-phenylpropionic acid with phosphorus pentoxide. He could not determine the exact structure but assumed it was a cyclic trimer of 1-indanone. According to him, it was formed by the condensation of 1-indanone resulting from intramolecular acylation of 3-phenylpropanoic acid.[2]
Frederic Stanley Kipping was able to confirm the structure of truxene in 1894 and obtained the compound by the trimerization of 1-indanone.[3]
Preparation
[edit]Truxene is prepared by the cyclotrimerization of 1-indanone in a mixture of acetic acid and concentrated hydrochloric acid. [4]
Uses
[edit]Truxene has a star shape, and it is therefore suitable as a starting point for the synthesis of dendrimers.[5]
Derivatives of truxene have also been used for the synthesis of liquid crystals[6] and fragments of fullerene.[7]
References
[edit]- ^ Harper, William L.; Smith, Wesley E. Process for synthesizing truxene; amorphous or graphitic carbon from indenes. 1970. US 3504044 A.
- ^ Hausmann, J. (July 1889). "Einwirkung von o -Cyanbenzylchlorid auf Natriummalonester. Untersuchung des α-Hydrindons". Berichte der Deutschen Chemischen Gesellschaft. 22 (2): 2019–2026. doi:10.1002/cber.18890220264.
- ^ Kipping, F. Stanley (1894). "XXIX. The formation of the hydrocarbon "truxene" from phenylpropionic acid, and from hydrindone". J. Chem. Soc., Trans. 65: 269–290. doi:10.1039/CT8946500269.
- ^ Amick, Aaron Warren (2008). Methodology Development for Use in Polycyclic Aromatic Hydrocarbon Synthesis. PhD thesis. p. 5. ISBN 978-0-549-75717-7.
- ^ Cao, Xiao-Yu; Zhang, Wen-Bin; Wang, Jin-Liang; Zhou, Xing-Hua; Lu, Hua; Pei, Jian (1 October 2003). "Extended π-Conjugated Dendrimers Based on Truxene". Journal of the American Chemical Society. 125 (41): 12430–12431. doi:10.1021/ja037723d. PMID 14531685.
- ^ Destrade, C.; Gasparoux, H.; Babeau, A.; Tinh, Nguyen Huu; Malthete, J. (May 1981). "Truxene Derivatives: A New Family of Disc-Like Liquid Crystals With an Inverted Nematic-Columnar Sequence". Molecular Crystals and Liquid Crystals. 67 (1): 37–47. doi:10.1080/00268948108070873.
- ^ Dehmlow, Eckehard V.; Kelle, Torsten (June 1997). "Synthesis of New Truxene Derivatives: Possible Precursors of Fullerene Partial Structures?". Synthetic Communications. 27 (11): 2021–2031. doi:10.1080/00397919708006804.