Advertisement
Research Article Free access | 10.1172/JCI76602
CD4+ and CD8+ T cell–dependent antiviral immunity requires STIM1 and STIM2
Patrick J. Shaw,1 Carl Weidinger,1 Martin Vaeth,1 Kevin Luethy,1 Susan M. Kaech,2 and Stefan Feske1
1Department of Pathology, New York University School of Medicine, New York, New York, USA and 2Department of Immunobiology, Yale University School of Medicine, New Haven, Connecticut, USA.
Address correspondence to: Stefan Feske, Department of Pathology, Experimental Pathology Program, New York University School of Medicine, 550 First Avenue, Smilow 316, New York, New York 10016, USA. Phone: 212.263.9066; E-mail: feskes01@nyumc.org.
Authorship note: Patrick J. Shaw and Carl Weidinger contributed equally to this work.
Find articles by Shaw, P. in: JCI | PubMed | Google Scholar
1Department of Pathology, New York University School of Medicine, New York, New York, USA and 2Department of Immunobiology, Yale University School of Medicine, New Haven, Connecticut, USA.
Address correspondence to: Stefan Feske, Department of Pathology, Experimental Pathology Program, New York University School of Medicine, 550 First Avenue, Smilow 316, New York, New York 10016, USA. Phone: 212.263.9066; E-mail: feskes01@nyumc.org.
Authorship note: Patrick J. Shaw and Carl Weidinger contributed equally to this work.
Find articles by Weidinger, C. in: JCI | PubMed | Google Scholar
1Department of Pathology, New York University School of Medicine, New York, New York, USA and 2Department of Immunobiology, Yale University School of Medicine, New Haven, Connecticut, USA.
Address correspondence to: Stefan Feske, Department of Pathology, Experimental Pathology Program, New York University School of Medicine, 550 First Avenue, Smilow 316, New York, New York 10016, USA. Phone: 212.263.9066; E-mail: feskes01@nyumc.org.
Authorship note: Patrick J. Shaw and Carl Weidinger contributed equally to this work.
Find articles by Vaeth, M. in: JCI | PubMed | Google Scholar
1Department of Pathology, New York University School of Medicine, New York, New York, USA and 2Department of Immunobiology, Yale University School of Medicine, New Haven, Connecticut, USA.
Address correspondence to: Stefan Feske, Department of Pathology, Experimental Pathology Program, New York University School of Medicine, 550 First Avenue, Smilow 316, New York, New York 10016, USA. Phone: 212.263.9066; E-mail: feskes01@nyumc.org.
Authorship note: Patrick J. Shaw and Carl Weidinger contributed equally to this work.
Find articles by Luethy, K. in: JCI | PubMed | Google Scholar
1Department of Pathology, New York University School of Medicine, New York, New York, USA and 2Department of Immunobiology, Yale University School of Medicine, New Haven, Connecticut, USA.
Address correspondence to: Stefan Feske, Department of Pathology, Experimental Pathology Program, New York University School of Medicine, 550 First Avenue, Smilow 316, New York, New York 10016, USA. Phone: 212.263.9066; E-mail: feskes01@nyumc.org.
Authorship note: Patrick J. Shaw and Carl Weidinger contributed equally to this work.
Find articles by Kaech, S. in: JCI | PubMed | Google Scholar
1Department of Pathology, New York University School of Medicine, New York, New York, USA and 2Department of Immunobiology, Yale University School of Medicine, New Haven, Connecticut, USA.
Address correspondence to: Stefan Feske, Department of Pathology, Experimental Pathology Program, New York University School of Medicine, 550 First Avenue, Smilow 316, New York, New York 10016, USA. Phone: 212.263.9066; E-mail: feskes01@nyumc.org.
Authorship note: Patrick J. Shaw and Carl Weidinger contributed equally to this work.
Find articles by Feske, S. in: JCI | PubMed | Google Scholar
Authorship note: Patrick J. Shaw and Carl Weidinger contributed equally to this work.
Published August 26, 2014 - More info
J Clin Invest. 2014;124(10):4549–4563. https://doi.org/10.1172/JCI76602.
Copyright © 2014, American Society for Clinical Investigation
Received: April 14, 2014; Accepted: July 10, 2014
Related article:
Abstract
Chronic infections induce a complex immune response that controls pathogen replication, but also causes pathology due to sustained inflammation. Ca2+ influx mediates T cell function and immunity to infection, and patients with inherited mutations in the gene encoding the Ca2+ channel ORAI1 or its activator stromal interaction molecule 1 (STIM1) are immunodeficient and prone to chronic infection by various pathogens, including
Authors
Ludovic Desvignes, Carl Weidinger, Patrick Shaw, Martin Vaeth, Theo Ribierre, Menghan Liu, Tawania Fergus, Lina Kozhaya, Lauren McVoy, Derya Unutmaz, Joel D. Ernst, Stefan Feske
-
Abstract
Calcium signaling is critical for lymphocyte function, and intracellular Ca2+ concentrations are regulated by store-operated Ca2+ entry (SOCE) through Ca2+ release–activated Ca2+ (CRAC) channels. In patients, loss-of-function mutations in CRAC channel components ORAI1 and STIM1 abolish SOCE and are associated with recurrent and chronic viral infections. Here, using mice with conditional deletion of Stim1 and its homolog Stim2 in T cells, we determined that both components are required for the maintenance of virus-specific memory CD8+ T cells and recall responses following secondary infection. In the absence of STIM1 and STIM2, acute viral infections became chronic. Early during infection, STIM1 and STIM2 were required for the differentiation of naive CD8+ T cells into fully functional cytolytic effector cells and mediated the production of cytokines and prevented cellular exhaustion in viral-specific CD8+ effector T cells. Importantly, memory and recall responses by CD8+ T cells required expression of STIM1 and STIM2 in CD4+ T cells. CD4+ T cells lacking STIM1 and STIM2 were unable to provide “help” to CD8+ T cells due to aberrant regulation of CD40L expression. Together, our data indicate that STIM1, STIM2, and CRAC channel function play distinct but synergistic roles in CD4+ and CD8+ T cells during antiviral immunity.
-
Introduction
Ca2+ signals play an important role in the function of CD4+ and CD8+ T cells (1, 2). Intracellular Ca2+ concentrations in T cells are predominantly regulated through Ca2+ release–activated Ca2+ (CRAC) channels in the plasma membrane (3, 4). CRAC channels are activated following T cell receptor (TCR) engagement, which leads to the activation of phospholipase Cγ, production of 1,4,5-inositol trisphosphate (IP3), and release of Ca2+ from ER Ca2+ stores via the opening of IP3 receptor channels. Ca2+ release, however, is not sufficient to sustain intracellular Ca2+ levels, cytokine production, and T cell activation (1, 5). Instead, Ca2+ release activates 2 proteins located in the ER membrane, stromal interaction molecule 1 (STIM1) and STIM2, which translocate to ER plasma membrane junctions (6, 7), where they bind and open ORAI1, the pore-forming subunit of the CRAC channel (8–10). Since this form of Ca2+ influx is dependent on the Ca2+ filling state of the ER, it is referred to as store-operated Ca2+ entry (SOCE) (2, 3, 11, 12).
The importance of CRAC channels for lymphocyte function is emphasized by the severe combined immunodeficiency–like (SCID-like) disease in patients with mutations in STIM1 and ORAI1 genes we characterized, whose T cells lack CRAC channel function and SOCE (8, 13–15). These patients are susceptible to recurrent and chronic viral infections, particularly those involving herpes viruses, including EBV, CMV, and human herpes virus 8 (HHV-8), which led to the development of virus-associated tumors in some patients (13, 14, 16, 17). These findings indicate an important role of CRAC channels in T cell–mediated antiviral and antitumor immunity. While T cells develop normally in ORAI1- and STIM1-deficient patients and mice, their function is severely impaired. CD4+ and CD8+ T cells show reduced antigen-specific proliferation in vitro and fail to produce IL-2, IFN-γ, TNF-α, and other cytokines (13, 18–22). We found that in cytotoxic CD8+ T cells, CRAC channels are required for controlling tumor growth in several mouse models of cancer and for tumor cell killing (23). Additionally, CRAC channels are required for the function of CD4+ T cells in vivo, as mice with T cell–specific deletion of Stim1 or Orai1 genes were protected from CD4+ T cell–mediated inflammation in animal models of multiple sclerosis and colitis (20, 24, 25).
How CRAC channels control antiviral immunity in vivo is poorly understood. CD8+ T cells are essential for antiviral immunity by killing virus-infected cells during the acute stages of infection and by providing long-term protection against viral infection through the generation and maintenance of memory CD8+ T cells. During an acute viral infection, naive virus–specific CD8+ T cells rapidly expand and differentiate into cytotoxic terminal effector (Teff) cells whose primary function is to kill virus-infected cells via the release of granzyme and perforin and the secretion of cytokines such as IFN-γ and TNF-α. Teff cells are characterized by high expression levels of the killer cell lectin-like receptor G1 (KLRG1) and the transcription factor T-bet, but low levels of IL-7 receptor α chain (IL-7Rα or CD127) (26). Following viral clearance, the Teff cell population contracts, whereas a smaller population of antigen-specific, long-lived memory CD8+ T cells persists that expresses high levels of CD127, but low levels of KLRG1 (26). The development, maintenance, and function of memory CD8+ T cells are controlled by a number of factors. These include the strength and frequency of TCR-antigen interactions (27, 28), costimulatory receptors and ligands on T cells and antigen-presenting cells (APCs), CD4+ T cell help (29, 30), cytokines (31), virus-neutralizing antibodies (32), and CD8+ T cell–intrinsic transcription factors like Eomesodermin (Eomes) (26, 33, 34). A defining trait of adaptive immunity is the rapid expansion of the long-lived memory CD8+ T cells upon secondary infection with virus (35). This recall response is controlled by a number of factors including IL-2 secretion by CD8+ or CD4+ T cells (36, 37), costimulatory signals such as CD40L (29, 38), and the exhaustion of CD8+ T cells (39). Recall responses to viral reinfection result in the proliferation of memory CD8+ T cells and their differentiation into effector cells that are able to kill virus-infected cells and provide strong protective immunity.
To better understand how CRAC channels control immunity to infection, we used mice with conditional deletion of Stim1 and Stim2 genes whose CD4+ and CD8+ T cells completely lack CRAC channel function and SOCE (21). We show that STIM1 and STIM2 contribute to the differentiation and function of effector CD8+ T cells during acute infection with lymphocytic choriomeningitis virus (LCMV). Importantly, SOCE regulated by STIM1 and STIM2 was essential for maintaining virus-specific memory CD8+ T cells and their ability to control viral infection. In addition, the function of memory CD8+ T cells and their ability to mediate recall responses and protection against secondary viral infection depended on STIM1 and STIM2. While some Teff cell functions were regulated in a CD8+ T cell–intrinsic manner, we observed that the maintenance of memory CD8+ T cells as well as their expansion and function upon reinfection were largely controlled by STIM1 and STIM2 in CD4+ T cells. Analyzing CD4+ T cells from STIM1/2-deficient mice and a patient with loss-of-function mutation in STIM1, we found that CRAC channels controlled the expression of CD40L in CD4+ T cells, which is required for the maintenance of memory CD8+ T cells and production of antiviral antibodies. Our findings identify a critical role of STIM1 and STIM2 in T cell–mediated antiviral immunity, protection from chronic viral infections, and recall responses to infectious pathogens, all of which have important implications for understanding T cell function during vaccination.
-
Results
STIM1 and STIM2 in T cells control immunity to acute viral infection and the maintenance of memory CD8+ T cells. To examine the role of STIM1 and STIM2, and thus CRAC channel–mediated SOCE, in T cells during antiviral immune responses, we used mice with conditional T cell–specific deletion of Stim1 and Stim2 genes. Stim1fl/fl Stim2fl/fl Cd4-Cre mice (referred to hereafter as DKO mice) lack SOCE completely in both CD4+ and CD8+ T cells (21), but not in CD4+ DCs (Supplemental Figure 1; supplemental material available online with this article; doi:10.1172/JCI76602DS1). Systemic exposure of mice to the Armstrong strain of LCMV (LCMVARM) causes an acute viral infection and a well-characterized CD8+ T cell response (40, 41). Despite the complete loss of SOCE in T cells from DKO mice, the mice were able to clear LCMVARM from the serum below detection levels by day 8 post infection (p.i.), similar to the levels detected in their WT littermates (Figure 1A). Viral titers were detectable in the livers of WT and DKO mice 8 days p.i. with only a moderate, nonsignificant elevation of titers in DKO mice compared with that detected in WT mice. At later time points (35–60 days p.i.), progressively increasing LCMV titers were observed in the serum and liver of DKO mice, whereas the littermate controls had cleared LCMV from the serum and liver (Figure 1A). Collectively, these findings show that STIM1 and STIM2 in T cells are critical to provide sterilizing immunity during the acute phase of infection and to prevent chronicity of a normally acute viral infection.
 Figure 1
Figure 1STIM1 and STIM2 in T cells control immunity to acute LCMV infection and the maintenance of virus-specific memory CD8+ T cells. Stim1fl/fl Stim2fl/fl Cd4-Cre (DKO) and WT mice were infected with LCMVARM (2 × 105 PFU; i.p.). (A) Viral titers in the sera and livers of mice. Each dot represents 1 mouse; horizontal lines show the means of the viral titers. ND, not detectable. (B) Representative flow cytometric plots of splenic CD8+ T cells from WT and DKO mice analyzed 8 days p.i. using DbNP396–404 and Db-GP33–41 tetramers. (C) Total numbers of LCMV-specific (DbNP396–404 tetramer+) CD8+ T cells in the spleens of WT (n = 5–9) and DKO (n = 7–9) mice at days 8, 35, and 60 p.i. (D–G) Expression of KLRG1 and CD127 (IL-7Rα) on DbNP396–404 tetramer+ splenic CD8+ T cells from WT (n = 5–9) and DKO (n = 7–9) mice (D). Total numbers of LCMV-specific terminal effector (KLRG1+CD127– in E) and memory CD8+ T cell populations (KLRG1+CD127+ in F, KLRG1–CD127+ in G). Numbers in E–G indicate fold change differences between WT and DKO mice. Numbers in dot plots in B and D represent the percentages of cells in gates. Statistical significance was calculated by Student’s t test (*P < 0.05; **P < 0.01; ***P < 0.001).
To investigate the CD8+ T cell response to LCMVARM infection, we analyzed the number of LCMV-specific CD8+ T cells using MHC class I tetramers against 2 distinct LCMV epitopes, DbNP396–404 and DbGP33–41. The total numbers of NP396–404- and GP33–41-specific CD8+ T cells were comparable in DKO and WT mice 8 and 35 days p.i., but were moderately reduced 60 days p.i. (Figure 1, B and C, and Supplemental Figure 2). To elucidate the role of STIM1 and STIM2 in the differentiation of CD8+ T cells into Teff and memory cells, we analyzed the expression of CD127 (IL-7Rα) and KLRG1 on WT and DKO LCMV-specific CD8+ T cells (26, 35, 42, 43). During the acute phase of LCMV infection (8 days p.i.), DKO mice showed reduced frequencies and absolute numbers of LCMV-specific KLRG1+CD127– Teff cells (Figure 1, D and E). By contrast, the frequencies and absolute numbers of KLRG1–CD127+ memory precursor CD8+ T cells were normal (Figure 1, D and G). During the memory phase of the CD8+ T cell response (35–60 days p.i.), the numbers of LCMV-specific KLRG1+CD127– Teff cells in DKO mice were equivalent to those in WT controls (Figure 1, D and E). Importantly, however, we observed a significant approximately 3-fold reduction in the number of LCMV-specific KLRG1–CD127+ and KLRG1+CD127+ memory CD8+ T cells in DKO mice (Figure 1, D, F, and G, and Supplemental Figure 2). Taken together, these findings suggest an early (8 days p.i.) defect in the differentiation of Teff cells and a later (35–60 days p.i.) defect in the maintenance of virus-specific memory CD8+ T cells in DKO mice. It is of note that reduced numbers of memory CD8+ T cells and viral recrudescence 35–60 days p.i. were only observed in LCMVARM-infected DKO mice but not in mice with T cell–specific deletion of Stim1 or Stim2 genes alone (Supplemental Figure 3), despite the fact that in T cells from Stim2fl/fl Cd4-Cre and Stim1fl/fl Cd4-Cre mice SOCE is moderately to severely impaired, respectively (20, 21). These findings indicate that STIM1 and STIM2 in T cells synergize to control antiviral immune responses to LCMV.
STIM1 and STIM2 regulate the function and differentiation of virus-specific effector CD8+ T cells. To understand whether recrudescence of LCMV was due to impaired viral clearance during acute infection, we analyzed the cytotoxic function of DKO CD8+ T cells. CD8+ T cells from WT and DKO P14 mice (which express a transgenic LMCV-specific TCR) were cocultured with LCMV GP33–41 peptide–pulsed target cells. We observed that DKO CD8+ T cells were significantly impaired in their ability to kill target cells in vitro (Figure 2A). Furthermore, when we isolated splenocytes from LCMVARM-infected WT and DKO mice and stimulated them with GP33–41 peptide or PMA and ionomycin in vitro, we found significantly reduced IFN-γ production in CD8+ T cells from DKO mice compared with that in WT controls (Figure 2B). Together, these data indicate that STIM1 and STIM2 are required for antiviral effector functions of CD8+ T cells, consistent with previous reports (23, 44).
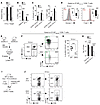 Figure 2
Figure 2STIM1 and STIM2 regulate the function and differentiation of effector CD8+ cells. (A) CD8+ cytotoxic CTLs from DKO P14 or WT P14 mice were cocultured with peptide-pulsed EL-4 cells; apoptosis was detected by annexin V staining. (B) Splenic CD8+ T cells from LCMVARM-infected WT (n = 6) and DKO (n = 5) mice were stimulated with PMA/ionomycin (iono) or GP33–41 peptide for 6 hours and IFN-γ production determined by flow cytometry. (C) Apoptosis and proliferation of LCMV-specific WT (n = 6) and DKO (n = 5) CD8+ T cells analyzed by annexin V and Ki67 staining. (D) T-bet and Eomes expression in CD8+ T cells from 3 mice per group. (E–H) Mixed BM chimeras were generated by reconstituting Rag2–/– mice with BM from WT (CD45.1) and DKO (CD45.2) mice and infected with LCMVARM (E). Frequency (F and G) and total number (H) of splenic effector CD8+ T cells of WT or DKO origin. Each dot in G represents 1 WT:DKO chimera; horizontal lines represent mean cell percentages. (I and J) Congenic CD45.1 WT mice were infected with LCMVARM and injected with 5 × 104 CD8+ T cells from DKO P14 and WT P14 mice. (J) Left panels show percentages of DbGP33–41 tetramer+ CD8+ T cells; right panels show percentages of transferred versus host cells among LCMV-specific cells. Plots are representative of 4 mice per group. Statistical significance was calculated using Student’s t test (*P < 0.05; **P < 0.01; ***P < 0.001). Bar graphs in A–D and H represent the means ± SEM. Numbers in F and J represent the percentage of cells.
In addition, the numbers of KLRG1+CD127– Teff cells were reduced by 2.6-fold in DKO mice compared with those in WT littermates 8 days p.i. (Figure 1, D and E), which likely contributes to the incomplete viral clearance. This reduction was not due to enhanced apoptosis or diminished proliferation of Teff cells (Figure 2C). Rather, we observed altered expression of the transcription factors T-bet and Eomes in virus-specific DKO CD8+ T cells (Figure 2D). T-bet plays an important role in Teff differentiation, and Eomes is required for memory CD8+ T cell differentiation and maintenance (45–47). On day 8 p.i., DKO CD8+ T cells showed decreased T-bet expression. Conversely, Eomes expression was elevated in DKO CD8+ T cells (Figure 2D). Thus, in the absence of STIM1 and STIM2, virus-specific CD8+ T cells are skewed toward a memory-like phenotype with reduced T-bet and increased Eomes levels, likely accounting for the reduction in Teff cells. This STIM1/2-dependent polarization occurs in response to LCMV infection, as T-bet and Eomes expression levels were comparable in naive CD8+ T cells from uninfected WT and DKO mice (Supplemental Figure 4, A and B).
To analyze whether STIM1 and STIM2 regulate the function and differentiation of Teff cells in a CD8+ T cell–intrinsic manner, we generated mixed BM chimeric mice to compare the response of WT and DKO Teff cells to LCMV infection in the same host (Figure 2E). In these WT:DKO chimeras, naive WT and DKO CD8+ T cells are initially present at a 1:1 ratio (data not shown). Eight days after infection of chimeras with LCMVARM, however, only approximately 11% of LCMV-specific CD8+ T cells were of DKO origin (CD45.1–; Figure 2F). Of all virus-specific WT CD8+ T cells, approximately half were Teff cells compared with only 17% DKO Teff cells (Figure 2, F and G). Accordingly, the absolute numbers of DKO Teff cells were strongly reduced (~12-fold) compared with those in WT mice (Figure 2H). To confirm these findings, we adoptively transferred CD8+ T cells from WT P14 or DKO P14 mice (which express a transgenic LCMV GP33–41-specific TCR) into LCMVARM-infected congenic CD45.1+ WT mice (Figure 2I). CD8+ T cells from WT P14, but not DKO P14, mice greatly expanded within 7 days after infection. WT P14 T cells accounted for approximately 90% of all virus-specific cells in the host mice compared with DKO P14 T cells, which accounted for only approximately 6% (Figure 2J). Since host mice have an intact immune system that can support Teff development of transferred DKO P14 T cells, these data show, together with those from WT:DKO chimeras, that STIM1 and STIM2 play a CD8+ T cell–intrinsic role in the differentiation of Teff cells during acute LCMV infection.
STIM1 and STIM2 regulate CD8 memory in a non-CD8+ T cell–intrinsic manner. We next investigated the mechanisms underlying impaired memory CD8+ T cell maintenance (Figure 1, D, F, and G) and whether this defect, like that of Teff cell differentiation, is also CD8+ T cell intrinsic. Expression levels of cytokine receptors that are critical for the development and homeostasis of memory CD8+ T cells such as IL-7R (CD127), IL-2Rα (CD25), IL-2Rβ, and IL-15R (48–50) were normal or elevated in virus-specific CD8+ T cells from DKO mice 8 and 35 days p.i. (Figure 1D and Supplemental Figure 4, C and D). Next, we asked whether the selective loss of memory CD8+ T cells in DKO mice is due to enhanced apoptosis. The number of virus-specific annexin V+ CD8+ T cells was significantly increased in DKO memory CD8+ T cells compared with that in WT littermates at days 35 and 60 p.i. (Figure 3A) (a similar increase was also observed in the Teff population; data not shown). While DKO memory CD8+ T cells proliferated more vigorously than did WT controls 35 days p.i., they stopped proliferating at day 60 p.i. (Figure 3B), suggesting that reduced numbers of memory CD8+ T cells in DKO mice at late time points after infection may be due to increased cell death in the absence of proliferation.
 Figure 3
Figure 3STIM1 and STIM2 regulate CD8 memory in a non-CD8+ T cell–intrinsic manner. (A–E) Increased expression of cell death, proliferation, exhaustion, and memory differentiation markers in DKO CD8+ T cells after LCMVARM infection. WT and DKO mice were analyzed for the frequencies of apoptotic (annexin V+ in A), proliferating (Ki67+ in B), and exhausted (Tim-3+, PD-1+ in C) splenic CD8+ T cells. Line graphs show the mean ± SEM of all DbNP396–404 tetramer+ CD8+ T cells over the course of LCMV infection in WT and DKO mice (3–6 per group). (D) Impaired IL-2 and IFN-γ production by splenic DKO CD8+ T cells after in vitro stimulation with GP33–41 peptide for 6 hours. Representative dot plots (left) and mean percentages (right) of IL-2+ cells among IFN-γ+ CD8+ T cells. (E) T-bet and Eomes expression (ratio of MFI values) in DbNP396–404 tetramer+ CD8+ T cells from 3 WT and 3 DKO mice analyzed by flow cytometry. (F–K) Frequencies (F) and total numbers (G) of DbNP396–404 tetramer+ CD8+ T cells in 5 WT:DKO chimeras. Green boxes highlight memory CD8+ T cells. (H–J) Frequencies of apoptotic (annexin V+ in H), proliferating (Ki67+ in I), and exhausted (Tim-3+, PD-1+ in J) splenic memory CD8+ T cells of WT and DKO origin. (K) T-bet/Eomes ratio in memory CD8+ T cells from 5 WT:DKO chimeras. Statistical significance was calculated using Student’s t test (*P < 0.05; **P < 0.01; ***P < 0.001). Bar graphs show the means ± SEM. Numbers in D and F represent the percentages of cells in gates.
During chronic infection, CD8+ T cells become exhausted, gradually losing their ability to proliferate as well as some effector functions such as IL-2 and IFN-γ secretion (51, 52). We investigated whether the loss of memory CD8+ T cells and viral recrudescence in DKO mice are due to CD8+ T cell exhaustion. We found that expression levels of molecules associated with T cell exhaustion such as T cell immunoglobulin mucin 3 (Tim-3) and programmed death 1 (PD-1) progressively increased with time p.i. in virus-specific CD8+ T cells from DKO, but not WT, mice (Figure 3C) and that DKO CD8+ T cells showed impaired IL-2 and IFN-γ production (Figure 3D). Viral titers at day 60 p.i. correlated negatively with IFN-γ production after in vitro restimulation with LCMV GP33–41 peptide and positively with the frequency of LCMV-specific CD8+ T cells expressing Tim-3 and PD-1 when both WT and DKO mice were included in a correlation analysis performed in analogy to that by Wherry et al. (ref. 53 and Supplemental Figure 5, A–E). However, viral titers did not correlate with Tim-3/PD-1 expression or with IFN-γ levels at day 60 p.i. when analyzed within the cohort of DKO mice alone, as would have been expected if the viral load determined the degree of CD8+ T cell exhaustion in DKO mice (Supplemental Figure 5, A–D, and F). Importantly, we observed that IFN-γ production by DKO CD8+ T cells was already severely impaired on day 8 p.i. when viral titers were low and comparable in WT and DKO mice (Supplemental Figure 5C). Furthermore, IFN-γ production was decreased and PD-1 levels were increased in Stim1fl/fl Cd4-Cre single-knockout mice in which LCMVARM infection did not become chronic and memory CD8+ T cell maintenance was normal (Supplemental Figure 3 and data not shown). Taken together, these data are consistent with the known role of Ca2+/NFAT in the transcriptional regulation of IFN-γ and argue against an important role for CD8+ T cell exhaustion in impaired antiviral immunity in DKO mice.
To test whether impaired memory CD8+ T cell maintenance could be due to altered expression of transcription factors that determine effector and memory fates of CD8+ T cells, we analyzed the levels of T-bet and Eomes in Teff and memory CD8+ T cell subsets of WT and DKO mice. Thirty-five and 60 days p.i., we observed that expression of T-bet was decreased and that of Eomes increased in DKO memory CD8+ T cells compared with expression levels in WT controls, resulting in a decreased T-bet/Eomes ratio (Figure 3E), which suggests that DKO CD8+ T cells are biased toward memory cells. These data indicate, together with normal numbers of KLRG1–CD127+ memory precursor cells 8 days p.i. (Figure 1G), that STIM1 and STIM2 do not control the initial development of memory CD8+ T cells but regulate their maintenance.
To understand whether the role of STIM1 and STIM2 in the maintenance of memory cells is intrinsic to CD8+ T cells, we analyzed LCMVARM-infected WT:DKO chimeric mice (Figure 3, F–K). The frequencies and absolute numbers of all LCMV-specific DKO CD8+ T cells were moderately reduced compared with those in WT controls 35 and 60 days p.i. (Figure 3, F and G). This was due to a selective approximately 33-fold decrease in the number of DKO Teff cells, consistent with the CD8+ T cell–intrinsic role of STIM1 and STIM2 in Teff cell differentiation (described in Figure 2). By contrast, the numbers of KLRG1+CD127+ and KLRG1–CD127+ memory CD8+ T cells as well as their apoptosis and proliferation rates were comparable between WT and DKO cells in the chimeric mice (Figure 3, G–I), indicating that STIM1 and STIM2 are not required for the maintenance of antiviral memory in a CD8+ T cell–intrinsic manner. DKO memory CD8+ T cells were maintained in the WT:DKO chimeras, although the expression level of Tim-3 remained high (Figure 3J), similar to that observed in DKO mice (Figure 3C). Likewise, T-bet/Eomes ratios were significantly decreased in DKO memory CD8+ T cell populations compared with those in WT cells (Figure 3K), similar to our observations in DKO mice (Figure 3E). Collectively, these data demonstrate that the maintenance of memory CD8+ T cells is restored in the presence of WT T cells (in WT:DKO chimeric mice) and is therefore not a CD8+ T cell–intrinsic function of STIM1 and STIM2.
STIM1 and STIM2 in CD4+ T cells are required for the maintenance of memory CD8+ T cells. Since the maintenance of DKO memory CD8+ T cells was restored in WT:DKO chimeras and CD4+ T cell help is known to be required to support memory CD8+ T cell responses (43, 54–56), we tested whether STIM1 and STIM2 in CD4+ T cells are critical for the maintenance of memory CD8+ T cells. We generated mixed BM chimeras with CD8a–/– mice in which greater than or equal to 90% of CD4+ T cells were of WT origin and CD8+ T cells of DKO or WT origin (Figure 4A and ref. 31). The resulting DKO:Cd8a–/– and WT:Cd8a–/– chimeras that have an essentially normal CD4+ T cell compartment were infected with LCMVARM. Sixty days p.i., the numbers of total virus–specific DKO CD8+ T cells and memory DKO CD8+ T cells in DKO:Cd8a–/– chimeras were comparable to those in WT:Cd8a–/– mice (Figure 4, B and C), indicating that the maintenance of DKO memory CD8+ T cells is restored in the presence of WT CD4+ T cells. Importantly, DKO:Cd8–/– chimeric mice were able to control viral infection completely, as LCMVARM titers were undetectable at days 8 and 60 p.i. (data not shown). Efficient control of LCMV infection in DKO:Cd8a–/– chimeras was achieved despite absent IL-2 and reduced IFN-γ production in DKO CD8+ T cells (Figure 4D).
 Figure 4
Figure 4STIM1 and STIM2 are required for CD4+ T cell help to maintain memory CD8+ T cells. (A–D) WT CD4+ T cells restore the maintenance of DKO memory CD8+ T cells. (A) Generation of WT:Cd8a–/– and DKO:Cd8a–/– chimeras. (B–D) Chimeras were infected with LCMVARM and analyzed 60 days p.i. for the frequencies (B) and absolute numbers (C) of LCMV-specific CD8+ T cells (mean ± SEM of cells from 6 WT and 6 DKO chimeras). (D) Intracellular cytokine staining for IL-2 and IFN-γ in splenic CD8+ T cells isolated from chimeras 60 days p.i. and restimulated with GP33–41 peptide for 5 hours in vitro. (E–L) WT CD8+ T cells require STIM1/2-dependent CD4+ T cell help for memory maintenance. (E) 5 × 104 WT P14 T cells (Thy1.1) were adoptively transferred into congenic WT or DKO mice and simultaneously infected with LCMVARM. (F–H) Frequencies of Thy1.1+KLRG1–CD127+ memory P14 T cells in the blood 8–60 days p.i. Bar graphs show the means ± SEM. (I and J) Frequencies (I) and total numbers (J) of splenic effector and memory subsets of Thy1.1+ WT P14 T cells 60 days p.i. (K) Serum LCMV titers. Each circle represents 1 mouse; horizontal lines represent the mean viral titers. (L) Tim-3 and PD-1 expression on splenic P14 T cells. Data in F–L are from 6 WT and 5 DKO mice. Statistical significance was calculated by Student’s t test (**P < 0.01; ***P < 0.001). Numbers in FACS plots represent percentages. Green boxes in B and F–H highlight memory CD8+ T cell populations.
To confirm the essential role of STIM1 and STIM2 in CD4+ T cells for memory CD8+ T cell maintenance, we transferred CD8+ T cells from WT P14 mice into congenic WT or DKO mice that were subsequently infected with LCMVARM (Figure 4, E–L). Similar frequencies of KLRG1–CD127+ P14 memory precursor cells were present 8 days p.i. in both WT and DKO host mice (Figure 4F). At later time points, memory P14 cells were almost completely absent in the blood (30 and 60 days p.i., Figure 4, G and H) and spleen (60 days p.i., Figure 4, I and J) of DKO mice compared with WT mice. By contrast, the numbers of effector P14 T cells in DKO mice 60 days p.i. were normal (Figure 4, I and J). The lack of memory P14 CD8+ T cells was associated with viral recrudescence in the serum of DKO, but not WT, mice (Figure 4K). Despite viral recrudescence in DKO mice, there was no increased expression of the exhaustion markers Tim-3 or PD-1 by transferred P14 cells in DKO mice compared with expression levels in WT mice (Figure 4L), and viral titers did not correlate with Tim-3 and PD-1 levels (Supplemental Figure 6, A and D). IFN-γ production by transferred WT P14 cells was moderately impaired in DKO mice compared with that in WT mice and correlated negatively with increased viral titers (Supplemental Figure 6, B and D). No such correlation was found between viral titers and IFN-γ production or PD-1/Tim-3 expression within the cohort of DKO recipient mice (Supplemental Figure 6, A–C, and E). Taken together, our data show that STIM1 and STIM2 regulate memory CD8+ T cell maintenance and antiviral immunity in a CD8+ T cell–extrinsic manner and do so largely independently of CD8+ T cell exhaustion.
STIM1 and STIM2 control CD40L expression and CD4+ T cell help to memory CD8+ T cells. The absence of CD4+ T cells during LCMV infection is known to result in a “helpless” CD8+ T cell response (29). We confirmed that while memory CD8+ T cells are initially present in Cd4–/– mice 8 days p.i. with LCMV, they are not maintained at later time points (days 80–100 p.i.; Supplemental Figure 7A), similar to our findings in DKO mice. To exclude the possibility that impaired memory CD8+ T cell maintenance in DKO mice is due to the absence of LCMV-specific CD4+ T cells, we analyzed the total numbers of virus-specific (I-Ab-GP66–77 tetramer+) CD4+ T cells in LCMVARM-infected WT and DKO mice and found that the numbers did not significantly differ between WT and DKO mice 8 and 35 days p.i. (Supplemental Figure 7, B and C). These findings indicated that STIM1 and STIM2 may be required for the function of CD4+ T cells and their ability to help memory CD8+ T cells. However, the Ca2+ dependent mechanisms regulating CD4+ T cell help are not well understood. One way in which CD4+ T cells provide help is the “licensing” of DCs through CD40L-CD40 interactions (29). We found that the total levels of CD40L protein in CD4+ T cells from the spleens of LCMV-infected DKO mice were significantly reduced 8 days p.i. compared with those in WT CD4+ T cells (Figure 5A). Since CD40L cycles extensively between the cell surface and intracellular compartments, we measured the expression of CD40L at the plasma membrane of CD4+ T cells following stimulation with LCMV GP61–80 peptide (57). CD4+ T cell activation resulted in a mobilization of CD40L to the plasma membrane of WT CD4+ T cells that was almost completely absent in DKO CD4+ T cells (Figure 5B). Consistent with the role of CD40L in DC licensing (29) and impaired CD40L surface expression on DKO CD4+ T cells, we found that the expression of MHC class II on CD11c+ DCs was significantly impaired in DKO mice 8 days p.i. (Figure 5C).
 Figure 5
Figure 5STIM1 and STIM2 control the maintenance of CD8 memory and generation of LCMV-specific antibodies by regulating CD40L expression on CD4+ T cells. (A and B) Impaired CD40L expression on DKO CD4+ T cells from LCMVARM-infected mice. (A) Total cellular CD40L in unstimulated splenic CD4+CD44+ T cells 8 days p.i. Bar graphs represent the mean MFI ± SEM of CD40L expression (5 mice per group). (B) Surface expression of CD40L on splenic CD4+ T cells 8 days p.i. and following stimulation with GP61–80 peptide in vitro. Bar graphs represent the means ± SEM of 3 repeat experiments. (C) MHC class II expression on splenic CD11c+ DCs 8 days p.i. Each dot represents 1 mouse; horizontal lines show the mean MFI. (D) Generation of WT:Cd40l–/– and DKO:Cd40l–/– chimeras used in E–H. (E and F) Frequencies (E) and absolute numbers (F) of LCMV-specific KLRG1–CD127+ memory CD8+ T cells 80 days p.i. with LCMVARM. Bar graphs in F show the mean ± SEM of cell numbers from 4 WT:Cd40l–/– and 4 DKO:Cd40l–/– chimeras. (G) Relative levels of LCMV-specific serum IgG (means ± SEM; 4–5 mice per group). (H) Impaired plasma membrane expression of CD40L on CD4+ T cells from a STIM1-deficient patient (PAT) (13) compared with that in a healthy donor (HD). CD4+ T cells were left unstimulated or stimulated with PMA/ionomycin for 5 hours. Representative histograms and mean MFI ± SEM from 3 experiments. Statistical significance was calculated by Student’s t test (*P < 0.05; **P < 0.01; ***P < 0.001). Numbers in FACS plots represent percentages.
We next investigated whether impaired CD40L expression on DKO CD4+ T cells is responsible for defective maintenance of memory CD8+ T cells. Stimulation of APCs with an agonistic anti-CD40 antibody was shown to enhance immune response against herpes virus (58), other infectious pathogens (59, 60), and tumors (61, 62) in the absence of CD4+ T cells. However, when we tested whether anti-CD40 injection of WT mice enhances the CD8+ T cell response to LCMVARM, we observed a significant loss, rather than an increase, of LCMV-specific memory CD8+ T cells (data not shown), consistent with a previous report (63). To be able to investigate the role of STIM1 and STIM2 in CD40L expression in the context of a memory CD8+ T cell response to LCMV infection in vivo, we generated DKO:Cd40l–/– and WT:Cd40l–/– chimeric mice (Figure 5, D–G). In these mice, half of the CD4+ T cells lack CD40L (Cd40l–/), and the other half is either DKO or WT. We hypothesized that if STIM1 and STIM2 are required for upregulation of CD40L on CD4+ T cells after LCMV infection in vivo, memory CD8+ T cell responses should be impaired in DKO:Cd40l–/– chimeras (all CD4+ T cells lack CD40L) but not in WT:Cd40l–/– chimeras (WT-derived CD4+ T cells [50%] express CD40L). Indeed, 80 days p.i. with LCMVARM, the frequencies (Figure 5E) and absolute numbers (Figure 5F) of LCMV-specific KLRG1–CD127+ memory CD8+ T cells were significantly reduced in DKO:Cd40l–/– chimeric mice compared with those in WT:Cd40l–/– chimeras. These data show that impaired CD40L expression on STIM1/2-deficient CD4+ T cells is responsible, at least in part, for the defective memory CD8+ T cell response in DKO mice. These findings are further supported by the impaired maintenance of CD127+ and CD62L+ memory CD8+ T cells we observed in Cd40l–/– mice after LCMV infection (Supplemental Figure 8).
CD40L is essential for the generation of virus-neutralizing antibodies, and LCMV-specific antibodies were shown to contribute to the maintenance of CD8+ T cell memory in LCMV-infected mice (32). Consistent with impaired CD40L expression on DKO CD4+ T cells, the levels of LCMV-specific IgG antibodies in DKO mice were significantly reduced 60 days p.i. compared with those in WT mice (Figure 5G). We next analyzed antibody titers in the WT:Cd40l–/– and DKO:Cd40l–/– chimeric mice. DKO:Cd40l–/– chimeras had significantly reduced LCMV-specific antibody titers compared with those in WT:Cd40l–/– chimeras (Figure 5G). As expected, we observed no reduction in LCMV-specific IgG antibody titers in DKO:Cd8a–/– chimeras (compared with WT:Cd8a–/– chimeras), as these mice have a WT CD4+ T cell compartment (Figure 5G). Collectively, these data demonstrate that impaired CD40L expression by DKO CD4+ T cells interferes with the production of LCMV-specific IgG antibodies and therefore very likely contributes to the impaired memory CD8+ T cell response in DKO mice. It is noteworthy that only partial defects in CD40L expression were observed on CD4+ T cells from Stim2fl/fl Cd4-Cre and Stim1fl/fl Cd4-Cre single-knockout mice (Supplemental Figure 9), consistent with their normal maintenance of memory CD8+ T cells after LCMVARM infection (Supplemental Figure 3).
To evaluate whether Ca2+ influx via STIM1 and STIM2 also regulates CD40L expression in human CD4+ T cells, we compared CD40L surface expression on primary human CD4+ T cells from a healthy donor and a patient with a loss-of-function mutation in STIM1 that abolishes Ca2+ influx (13). We found that CD40L expression on the surface of nonstimulated STIM1-deficient CD4+ T cells was already significantly lower than that in control T cells (Figure 5H). Stimulation with PMA-ionomycin led to robust CD40L mobilization in control cells, but not in STIM1-deficient CD4+ T cells. These results show that STIM1 is required for the expression of CD40L on human CD4+ T cells.
STIM1 and STIM2 are essential for memory CD8+ T cell function and recall responses to reinfection. Although residual memory CD8+ T cells are present in LCMV-infected DKO mice (Figure 1, D–G), we hypothesized that they are functionally impaired, given their exhausted phenotype, and abolished IL-2 production and the viral recrudescence in DKO mice. To investigate whether STIM1 and STIM2 are required for memory CD8+ T cell function and recall responses, we infected WT and DKO mice with LCMVARM and 60 days later rechallenged them with the clone 13 strain of LCMV (LCMVCL13). Seven days post reinfection (p.r.i.), we observed a severe defect in the expansion of LCMV-specific CD8+ T cells in DKO mice compared with that in WT mice (Figure 6, A and B). Accordingly, DKO mice failed to control reinfection with LCMVCL13, which was apparent in very high viral titers 7 days p.r.i. (Figure 6C), representing a 55-fold titer increase compared with the viral titers detected on day 60 after primary LCMVARM infection (Figure 1A). By contrast, WT mice completely cleared LCMVCL13. To determine whether the requirement for STIM1 and STIM2 signaling in recall responses by memory CD8+ T cells is intrinsic to CD8+ or CD4+ T cells, we used the WT and WT:Cd8a–/– and DKO:Cd8a–/– chimeric mice described above. Chimeric mice were infected with LCMVARM followed by reinfection with LCMVCL13 60 days later. Seven days after rechallenge, the numbers of LCMV-specific CD8+ T cells in DKO:Cd8a–/– chimeras increased by approximately 4-fold (compared with the numbers detected on day 60 p.i.), whereas those in WT:Cd8a–/– chimeras increased by approximately 10-fold (Figure 6D). Importantly, this partially restored recall response by DKO CD8+ T cells in the chimeric mice was sufficient to completely protect them against reinfection with LCMVCL13, as viral titers were undetectable in their sera 7 days p.r.i. (data not shown). This efficient recall response occurred despite the failure of DKO CD8+ T cells in DKO:Cd8a–/– chimeras to differentiate into KLRG1+CD127– effector cells (Figure 6E). The latter was associated with decreased T-bet and increased Eomes expression levels in DKO CD8+ T cells compared with those in WT cells (Figure 6F). Collectively, our data demonstrate that the expansion and function of memory CD8+ T cells during recall responses are critically dependent on STIM1 and STIM2 in CD4+ T cells.
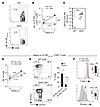 Figure 6
Figure 6STIM1 and STIM2 in CD4+ T cells are essential for recall responses to viral reinfection. WT and DKO mice (A–C) or Cd8a–/– chimeras (D–F) were infected with LCMVARM for 60 days, then reinfected with LCMVCL13, and analyzed 7 days p.r.i. (A–C) Impaired recall response in DKO mice. Representative contour plots (A) and total number (mean ± SEM in B) of LCMV-specific CD8+ T cells per spleen before and 7 days p.r.i. with LCMVCL13. Numbers in B indicate fold increase in LCMV-specific CD8+ T cells p.r.i. (C) Serum LCMV titers in WT (n = 7) and DKO (n = 6) mice 7 days p.r.i. Each dot represents 1 mouse; horizontal lines show the mean viral titers. (D–F) The presence of WT CD4+ T cells in chimeric mice restored recall responses by DKO CD8+ T cells. (D) Total number of LCMV-specific WT and DKO CD8+ T cells in the spleens of WT:Cd8a–/– (n = 4) and DKO:Cd8a–/– (n = 4) chimeras analyzed before and p.r.i. with LCMVCL13. Numbers indicate the fold increase of cell numbers 7 days p.r.i. (E and F) Expression of KLRG1 and CD127 (E) and T-bet and Eomes (F) on splenic LCMV-specific CD8+ T cells of WT or DKO origin in chimeras 7 days p.r.i. Line and bar graphs in D and E show the mean ± SEM of LCMV-specific CD8+ T cells from 4 mice per group. Statistical significance was calculated by Student’s t test (*P < 0.05; **P < 0.01; ***P < 0.001). Numbers in dot plots in A and E represent the percentages of cells in gates.
-
Discussion
We identified Ca2+ influx in T cells mediated by STIM1 and STIM2 as a critical signaling pathway required for CD8+ T cell immunity to viral infection. Together, STIM1 and STIM2 play distinct but synergistic roles in CD4+ and CD8+ T cells during antiviral immunity. CD8+ T cell–intrinsic functions of STIM1 and STIM2 include cytolytic effector functions and the differentiation of naive CD8+ T cells into virus-specific Teff cells. In addition, STIM1 and STIM2 in CD4+ T cells are essential to mediate antiviral CD8+ T cell responses by supporting (a) the maintenance of virus-specific memory CD8+ T cells, (b) memory CD8+ T cell function and effective recall responses to reinfection, and (c) the generation of virus-specific antibodies. We demonstrate an important role of STIM1 and STIM2 in the expression of CD40L on CD4+ T cells that was shown to be critical for the licensing of DCs (29) and the production of virus-specific antibodies, which in turn are required for the maintenance of memory CD8+ T cell responses. Accordingly, lack of STIM1 and STIM2 in T cells results in impaired CD4+ and CD8+ T cell immunity to primary and secondary viral challenge and chronification of a normally acute viral infection.
We were surprised to find that T cell–specific deletion of STIM1 and STIM2 had relatively little effect on the acute phase of the antiviral immune response, because the expansion of virus-specific CD4+ and CD8+ T cells and their ability to control viral replication in the first 8 days p.i. was largely normal. The numbers of LCMV-specific CD4+ and CD8+ T cells were similar and viral titers undetectable in the sera of WT and DKO mice, with only a moderate increase in LCMV titers in the livers of infected DKO mice. In vitro studies on the role of Ca2+ influx in cytotoxic lymphocytes had demonstrated that CRAC channels are important for the perforin-dependent cytolytic function of CD8+ T cells (44) and NK cells (64). This is consistent with our findings in this and a previous study that deletion of STIM1 and STIM2 impairs the ability of CD8+ Teff cells to release cytolytic granules and to kill target cells in vitro (23). Compromised cytotoxicity in the absence of STIM1 and STIM2 explains the lack of sterilizing immunity in DKO mice, resulting in viral recrudescence at days 35 and 60 p.i. The defect in cytotoxic effector function is compounded by reduced numbers of virus-specific KLRG1+CD127– Teff cells in DKO mice. This phenotype is substantially more pronounced in WT:DKO mixed BM chimeric mice and upon transfer of DKO P14 T cells into WT hosts, as DKO CD8+ Teff cells fail to expand and are strongly outcompeted by WT CD8+ Teff cells. A potential explanation for this differentiation defect is the significantly reduced level of T-bet in Teff cells, since T-bet regulates the differentiation of naive CD8+ T cells into Teff cells (46, 65). Collectively, our data demonstrate a CD8+ T cell–intrinsic role of STIM1 and STIM2 in the function, expansion, and differentiation of virus-specific Teff cells.
In contrast to moderate defects in acute antiviral immunity, we found that the memory T cell response to LCMV was severely impaired in DKO mice. The numbers of virus-specific KLRG1–CD127+ memory CD8+ T cells were markedly reduced in DKO mice 35 and 60 days p.i. However, the numbers of KLRG1–CD127+ memory CD8+ T cells were normal or increased in DKO mice 8 days p.i., suggesting that STIM1 and STIM2 are dispensable for their initial differentiation. Consistent with this conclusion, we found that protein levels of Eomes, a transcription factor essential for memory CD8+ T cell development, were increased and T-bet/Eomes ratios decreased in CD8+ T cells from DKO mice compared with levels in WT controls after LCMV infection. The apparent discrepancy between elevated Eomes expression and reduced numbers of memory CD8+ T cells at later stages of infection suggests that the Eomes-regulated developmental program from naive CD8+ T cells to memory CD8+ T cells is independent of STIM1 and STIM2, which is consistent with the normal numbers of KLRG1–CD127+ memory precursor CD8+ T cells we observed at day 8 p.i. Once developed, memory CD8+ T cells depend on STIM1 and STIM2 in CD4+ T cells for their maintenance, whereas Eomes expression in CD8+ T cells is not sufficient to sustain CD8 memory.
The maintenance of memory CD8+ T cells can be compromised by a number of factors, including CD8+ T cell exhaustion, a functional state characterized by impaired proliferation and production of IFN-γ, TNF-α, and IL-2 during chronic viral infection (52, 53). IL-2 and IFN-γ production was strongly impaired in DKO CD8+ T cells, which correlated with elevated viral titers in DKO mice. IFN-γ and IL-2 levels, however, are not suitable indicators of exhaustion in DKO mice, as their transcription is directly regulated by the Ca2+/calcineurin/NFAT pathway and is already impaired in DKO CD8+ T cells of noninfected mice (21). In fact, several lines of evidence indicate that CD8+ T cell exhaustion is not the primary cause of failed memory CD8+ T cell maintenance in the absence of STIM1 and STIM2: (a) the maintenance of LCMV-specific DKO memory CD8+ T cells was normal in WT:DKO chimeras despite their exhausted phenotype (high Tim-3 expression); (b) WT P14 T cells transferred into DKO mice failed to maintain their memory phenotype despite absent signs of exhaustion; (c) T cell–specific deletion of STIM1 alone does not impair the maintenance of memory CD8+ T cells or cause chronic LCMV infection despite increased expression levels of PD-1 and impaired IFN-γ production by CD8+ T cells.
CD4+ T cells are necessary to maintain virus-specific memory CD8+ T cells (66), and we show that this process is highly dependent on STIM1 and STIM2 in CD4+ T cells. This conclusion is based on normal numbers of LCMV-specific memory CD8+ T cells in WT:DKO and DKO:Cd8a–/– chimeric mice, which have a largely normal CD4+ T cell compartment in contrast to DKO mice. Conversely, we observed that adoptive transfer of LCMV-specific WT P14 T cells into DKO mice, whose CD4+ T cells lack STIM1 and STIM2, resulted in impaired maintenance of memory WT P14 T cells 35–60 days p.i. Although the numbers of LCMV-specific CD4+ T cells were comparable in WT and DKO littermate mice, STIM1- and STIM2-deficient CD4+ T cells were functionally impaired in their ability to provide T cell help. However, the molecular mechanisms by which STIM1 and STIM2 regulate CD4+ T cell function are unknown.
CD4+ T cells provide help to CD8+ T cells through a number of different mechanisms (29). Ligation of CD40 on DCs with CD40L expressed on CD4+ T cells has been shown to be critical for the maintenance of memory CD8+ T cells (32, 67–69), as CD40-CD40L interaction licenses DCs to prime CD8+ T cell responses (70, 71). We found that CD40L expression is tightly regulated by STIM1 and STIM2, since DKO CD4+ T cells have strongly reduced levels of CD40L at their cell surface before and after activation. Regulation of CD40L by STIM1 and STIM2 is consistent with the role of the Ca2+-dependent transcription factor NFAT in CD40L expression in murine and human T cells (72–74) and inhibition of de novo expression of CD40L by cyclosporine A, a calcineurin inhibitor (75). The important role of STIM1 and STIM2 in CD40L-dependent CD4+ T cell help for memory CD8+ T cell responses to LCMV is supported by the reduced numbers of memory CD8+ T cells in DKO:Cd40l–/– compared with WT:Cd40l–/– chimeras and the reduced MHC class II expression levels on DCs isolated from DKO mice, indicating that STIM1/2-dependent CD40L expression is required for DC licensing.
Furthermore, CD4+ T cell help for the production of LCMV-specific antibodies depends on CD40L (76). We found markedly reduced titers of LCMV-specific IgG antibodies in DKO mice and DKO:Cd40l–/– chimeras compared with those in WT mice and WT:Cd40l–/– chimeras, respectively. Since LCMV-specific antibodies support the maintenance of memory CD8+ T cells (32), the reduced production of antibodies against LCMV in DKO mice likely contributes to their memory CD8+ T cell defect. In addition, human CD4+ T cells from a STIM1-deficient patient had a profound defect in CD40L expression, likely explaining the chronic viral infections and impaired antibody responses to recall antigens observed in this and other CRAC channel–deficient patients (13, 77).
A defining hallmark of adaptive immunity is the ability to mount recall responses to reinfection. We show that recall responses by memory CD8+ T cells and their ability to control a secondary infection are critically dependent on STIM1 and STIM2 in CD4+ and, though to a lesser degree, CD8+, T cells. Not only were the numbers of virus-specific memory CD8+ T cells reduced in DKO mice, but the residual memory cells also failed to expand after reinfection with LCMV to become KLRG1+CD127– effector cells and to control viral replication. The expansion of memory CD8+ T cells and suppression of virus infection were largely restored in the presence of WT CD4+ T cells in DKO:Cd8a–/– chimeras, demonstrating that recall responses to virus infection are controlled predominantly by STIM1 and STIM2 in CD4+ T cells. It is noteworthy, however, that even in the presence of WT CD4+ T cells, DKO memory CD8+ T cells did not expand as efficiently as did WT CD8+ T cells and failed to become KLRG1+CD127– effector cells after reinfection. This may be due to the inability of STIM1/2-deficient memory CD8+ T cells in DKO:Cd8a–/– chimeras to produce IL-2. The transcription of IL-2 is regulated by the Ca2+-dependent transcription factor NFAT, and impaired SOCE diminishes NFAT activation and IL-2 production (21, 78, 79). While IL-2 does not modulate the number of virus-specific memory CD8+ T cells, it was shown to be essential for robust CD8+ T cell recall responses to reinfection with LCMV (37). A similar role of IL-2, albeit that produced by CD8+ T cells in an autocrine manner, in recall responses was reported by Feau et al. (36). A requirement for autocrine IL-2 produced by CD8+ T cells may explain the incomplete rescue of recall responses by LCMV-specific CD8+ T cells in DKO:Cd8a–/– chimeric mice. An additional explanation for an impaired, CD8+ T cell–intrinsic recall response may be the increased expression of the inhibitory receptors PD-1 and Tim-3 on DKO CD8+ T cells. Both proteins are associated with T cell exhaustion and impaired recall responses by CD8+ T cells (39, 51). While PD-1 expression was shown to be regulated by NFATc1 in vitro (80), in our studies, PD-1 levels were increased on LCMV-specific DKO CD8+ T cells. It is important to point out, however, that DKO memory CD8+ T cells in DKO:Cd8a–/– chimeras mounted a recall response that was sufficient to control reinfection with LCMVCL13, despite signs of exhaustion and failed differentiation into KLRG1+CD127– effector cells.
Our findings are clinically relevant because (a) they explain why STIM1- and ORAI1-deficient patients suffer from severe, chronic infections, and (b) they assess the benefits and risks of therapeutic CRAC channel inhibition, which is currently being explored for the treatment of autoimmune diseases. CRAC channelopathy in human patients due to mutations in STIM1 and ORAI1 genes is characterized by recurrent and chronic infections with CMV, EBV, HHV8, and other viruses, despite the presence of detectable EBV- and CMV-specific T cells (13–15, 77). In some of these patients, chronic infections caused the development of EBV-associated B cell lymphoma and HHV8- associated Kaposi sarcoma (13, 16, 17). The defective maintenance of memory CD8+ T cells, antibody production, and recall response to LCMV infection in DKO mice is reminiscent of the chronic viral infections in STIM1- and ORAI1-deficient patients and their lack of antigen-specific cellular and humoral immune responses (81). Surprisingly, and not predicted from in vitro experiments that showed defective function of cytotoxic effector CD8+ T cells in the absence of STIM1 and STIM2 (23), antiviral immunity is critically dependent on STIM1 and STIM2 function in CD4+ T cells. Both proteins synergize to enable CD4+ T cells to maintain memory CD8+ T cells through expression of CD40L and to facilitate recall responses to reinfection.
The dependence of memory CD8+ T cell responses on STIM1 and STIM2 has important implications for the development of CRAC channel inhibitors as therapeutics for the treatment of autoimmune diseases, inflammation, and cancer. Our findings caution that CRAC channel inhibition may result in chronification of (viral) infections and interfere with vaccination of infants and adults. In this context, it is important to note, however, that we observed impaired antiviral immunity against LCMV only in DKO mice, but not in mice lacking STIM1 or STIM2 individually. Since CD4+ T cells deficient for either STIM1 or STIM2 alone do not lack CRAC channel function completely, it is likely that their residual Ca2+ influx is sufficient to provide CD4+ T cell help for antiviral immunity. By contrast, partial reduction of Ca2+ influx in CD4+ T cells with individual deletion of STIM1, STIM2, or ORAI1 is sufficient to significantly attenuate proinflammatory CD4+ T cell function in murine models of autoimmune diseases (24). These different quantitative requirements for Ca2+ influx in antiviral immunity versus autoimmunity may provide a therapeutic window for CRAC channel inhibition in autoimmune and inflammatory disorders.
-
Methods
Mice. Stim1fl/fl Cd4-Cre, Stim2fl/flCd4-Cre, and Stim1fl/fl Stim2fl/flCd4-Cre (DKO) mice have been described previously (21). Unless otherwise stated, Cre-negative floxed littermates were used as WT controls. WT and DKO mice were further crossed with Tcra–/– and P14 TCR transgenic mice (Taconic) for more than 5 generations to generate WT P14 and DKO P14 mice. CD45.1, Cd8a–/–, Rag2–/–, and Cd40l–/– mice were purchased from The Jackson Laboratory. The WT P14 TCR transgenic mice used for adoptive transfer to congenic WT and DKO mice were a gift of R. Ahmed (Emory University, Atlanta, Georgia, USA).
Generation of mixed BM chimeras. To generate WT:DKO mixed BM chimeras, BM from DKO (CD45.2) and congenic WT (CD45.1) mice were mixed at a 1:1 ratio. To generate WT:Cd8a–/– and DKO:Cd8a–/– mixed BM chimeras, BM from Cd8a–/– mice was mixed with BM from either WT or DKO mice at a 9:1 ratio. To generate WT:Cd40l–/– and DKO:Cd40l–/– mixed BM chimeras, BM from Cd40l–/– mice was mixed with BM from either WT or DKO mice at a 1:1 ratio. In all cases, mixed BM was injected into congenic Rag2–/– mice lethally irradiated (11 Gy), and chimeras were used for experiments 6–8 weeks after reconstitution.
LCMV infection and analysis of viral titers. Stocks of the LCMVARM and LCMV clone 13 (LCMVCL13) strains were a gift of R. Ahmed. LCMVARM and LCMVCL13 were grown in BHK-21 cells and viral supernatant harvested as described (82). For primary infection, mice were injected i.p. with 2 × 105 PFU of LCMVARM. For secondary infections, mice previously infected with LCMVARM were injected i.v. with 2 × 106 PFU of LCMVCL13. Viral titers in serum or liver of infected mice as well as LCMV stocks were measured by viral plaque assays using Vero 76 cells as previously described (82). Briefly, confluent monolayers of Vero 76 cells were incubated with dilutions of samples for 1 hour. The sample solution was removed and replaced with a 1% agarose solution. Viral plaques were visualized 4 days later by neutral red solution.
Flow cytometry, antibodies, and tetramers. The following antibodies (clone number) for flow cytometry were purchased from eBioscience: CD4 (GK1.5), CD8 (53-6.7), CD44 (IM7), KLRG1 (2F1), CD127 (A7R34), IL-2Rα (PC61.5), IL-2Rβ (TM-b1), IL-15R (FAB551C), TRAIL (N2B2), CXCR4 (2B11), CXCR3 (CXCR3-173), T-bet (ebio4B10), Eomes (Dan11mag), Tim-3 (8B.2C12), PD-1 (J43), CD45.2 (104), CD45.1 (A20), IL-2 (JES6-5H4), IFN-γ (XMG1.2), CD40L (MR1), BrdU (Bu20a), human CD4 (OKT-4), human CD40L (24-31), CD11c (N418), and MHC class II (M5/114.15.2). Anti–IL-15R and annexin V were obtained from BD Biosciences. Class I MHC tetramers (H-2Db) complexed with peptides from LCMV glycoprotein (GP) 33-41 (KAVYNFATM) and LCMV nucleoprotein (NP) 396-404 (FQPQNGAFI), as well as class II tetramers (I-Ab) complexed with a peptide from LCMV GP 66–77 (DIYKGVYQFKSV) were generated by the NIH Tetramer Facility and conjugated with allophycocyanin (APC) and phycoerythrin (PE) fluorophores. Controls included CD8+ T cells from LCMV-infected mice left unstained, tetramer-stained CD8+ T cells from uninfected mice, or CD4+ T cells stained with APC-conjugated I-Ab tetramers loaded with CLIP peptide. Antibody and tetramer staining of CD4+ and CD8+ T cells was detected using an LSR II flow cytometer (BD Biosciences) and analyzed with FlowJo software (Tree Star Inc.).
In vitro killing assay. CD8+ T cells from WT P14 and DKO P14 mice expressing a transgenic LCMV GP33–41–specific TCR were isolated from the spleen by negative selection (STEMCELL Technologies), stimulated with plate-bound anti-CD3/anti-CD28 antibodies for 48 hours, and differentiated into cytotoxic lymphocytes (CTLs) for 6 to 7 days in the presence of 100 U/ml rhIL-2. CTLs were cocultured with EL-4 lymphoma cells that were labeled with 5 μM CFSE and coated with 10 μg/ml GP33–41 peptide at 1:1 and 5:1 ratios. After 90 minutes, CD8+ T cells and EL-4 cells were incubated with 4 μg/ml annexin V to measure apoptosis of EL-4 target cells.
LCMV-specific antibody measurements. Levels of LCMV-specific IgG antibodies in the serum of mice were measured by ELISA as described previously (83). Briefly, ELISA plates were coated overnight with lysates from LCMV-infected BHK-21 cells. LCMV-specific IgG was detected in serial dilutions of mouse serum using HRP goat anti-mouse IgG (Invitrogen), followed by the addition of 3,3′,5,5′-tetramethylbenzidine (TMB) substrate (BioLegend). Absorbance was measured at 450 nm using a SpectraMax M5 microplate reader (Molecular Devices).
CD40L measurements. Total CD40L expression was measured in murine CD4+ T cells permeabilized with 0.1% saponin. Plasma membrane expression of CD40L was measured as described previously (57). Briefly, splenocytes were isolated from LCMVARM-infected mice at day 8 p.i. and either left untreated or stimulated with LCMV GP61–80 peptide (30 μg/ml) or PMA (20 nM) plus ionomycin (1 μM) for 2 hours at 37°C in the presence of 1 μg/ml anti-CD40L antibody (clone MR1; eBioscience) to capture CD40L at the cell surface. Cells were washed, stained with additional antibodies against surface markers as indicated, and analyzed by flow cytometry. A similar protocol and anti-CD40L antibody (clone 24-31; eBioscience) were used for detection of CD40L at the surface of human CD4+ T cells.
Human T cell culture. T cells from healthy donors and a patient homozygous for a loss-of-function missense mutation (R429C) in STIM1 (13) were cultured as previously described (19). Briefly, 1 × 106 peripheral blood lymphocytes (PBLs) isolated by Ficoll gradient were stimulated with 1 μg/ml phytohemagglutinin (PHA-P) and allogenic feeder cells (1 × 106 irradiated PBLs and 1 × 105 irradiated B cells) and cultured in the presence of 20 U/ml rhIL-2 for 2 weeks. PBLs from the STIM1-deficient patient were provided by S. Ehl (Centre of Chronic Immunodeficiency, University Hospital Freiburg, Freiburg, Germany).
Statistics. P values were calculated using a 2-tailed, unpaired Student’s t test and Prism6 (GraphPad Software). Error bars show the mean SEM. P values less than 0.05 were considered statistically significant. For correlation analysis, SPSS software (IBM) was used, and statistics were calculated using Spearman’s Rho test.
Study approval. All animal experiments were conducted in accordance with protocols approved by the IACUC of New York University Langone Medical Center and Yale University. For experiments using primary human cells, informed consent for the studies was obtained from the patient’s family in accordance with the Declaration of Helsinki and IRB approval of the New York University School of Medicine.
-
Acknowledgments
We thank R. Ahmed for the gifts of LCMVARM and LCMVCL13 viral stocks and spleens of WT P14 mice. We thank M. Pipkin, H. Hu, and members of the Feske laboratory for helpful discussions. This work was funded by NIH grants R01AI097302 (to S. Feske), R37AI066232 and R01AI074699 (to S.M. Kaech), the Howard Hughes Medical Institute (to S.M. Kaech), postdoctoral fellowships by the National Multiple Sclerosis Society (to P.J. Shaw), and the Deutsche Forschungsgemeinschaft (DFG) (We 5303/1-1, to C. Weidinger and VA 882/1-1 to M. Vaeth).
Address correspondence to: Stefan Feske, Department of Pathology, Experimental Pathology Program, New York University School of Medicine, 550 First Avenue, Smilow 316, New York, New York 10016, USA. Phone: 212.263.9066; E-mail: feskes01@nyumc.org.
-
Footnotes
Conflict of interest: Stefan Feske is a cofounder of and scientific advisor for Calcimedica.
Reference information: J Clin Invest. 2014;124(10):4549–4563. doi:10.1172/JCI76602.
Kevin Luethy’s present address is: Laboratory of Neuronal Communication, Department of Human Genetics, KU Leuven, Leuven, Belgium.
See the related article at STIM1 controls T cell–mediated immune regulation and inflammation in chronic infection.
-
References
- Feske S. Calcium signalling in lymphocyte activation and disease. Nat Rev Immunol. 2007;7(9):690–702.View this article via: PubMed Google Scholar
- Hogan PG, Lewis RS, Rao A. Molecular basis of calcium signaling in lymphocytes: STIM and ORAI. Annu Rev Immunol. 2010;28:491–533.
- Feske S, Skolnik EY, Prakriya M. Ion channels and transporters in lymphocyte function and immunity. Nat Rev Immunol. 2012;12(7):532–547.View this article via: PubMed Google Scholar
- Lewis RS. Calcium signaling mechanisms in T lymphocytes. Annu Rev Immunol. 2001;19:497–521.
- Macian F. NFAT proteins: key regulators of T-cell development and function. Nat Rev Immunol. 2005;5(6):472–484.View this article via: PubMed Google Scholar
- Liou J, et al. STIM is a Ca2+ sensor essential for Ca2+-store-depletion-triggered Ca2+ influx. Curr Biol. 2005;15(13):1235–1241.
- Roos J, et al. STIM1, an essential and conserved component of store-operated Ca2+ channel function. J Cell Biol. 2005;169(3):435–445.
- Feske S, et al. A mutation in Orai1 causes immune deficiency by abrogating CRAC channel function. Nature. 2006;441(7090):179–185.
- Vig M, et al. CRACM1 is a plasma membrane protein essential for store-operated Ca2+ entry. Science. 2006;312(5777):1220–1223.
- Zhang SL, et al. Genome-wide RNAi screen of Ca(2+) influx identifies genes that regulate Ca(2+) release-activated Ca(2+) channel activity. Proc Natl Acad Sci U S A. 2006;103(24):9357–9362.
- Cahalan MD. STIMulating store-operated Ca(2+) entry. Nat Cell Biol. 2009;11(6):669–677.
- Shaw PJ, Feske S. Physiological and pathophysiological functions of SOCE in the immune system. Front Biosci (Elite Ed). 2012;4:2253–2268.View this article via: PubMed Google Scholar
- Fuchs S, et al. Antiviral and regulatory T cell immunity in a patient with stromal interaction molecule 1 deficiency. J Immunol. 2012;188(3):1523–1533.
- McCarl CA, et al. ORAI1 deficiency and lack of store-operated Ca2+ entry cause immunodeficiency, myopathy, and ectodermal dysplasia. J Allergy Clin Immunol. 2009;124(6):1311–1318.e7.View this article via: PubMed Google Scholar
- Picard C, et al. STIM1 mutation associated with a syndrome of immunodeficiency and autoimmunity. N Engl J Med. 2009;360(19):1971–1980.
- Byun M, et al. Whole-exome sequencing-based discovery of STIM1 deficiency in a child with fatal classic Kaposi sarcoma. J Exp Med. 2010;207(11):2307–2312.
- Sahin G, et al. Classic Kaposi sarcoma in 3 unrelated Turkish children born to consanguineous kindreds. Pediatrics. 2010;125(3):e704–e708.
- Feske S, Giltnane J, Dolmetsch R, Staudt LM, Rao A. Gene regulation mediated by calcium signals in T lymphocytes. Nat Immunol. 2001;2(4):316–324.
- Feske S, et al. Severe combined immunodeficiency due to defective binding of the nuclear factor of activated T cells in T lymphocytes of two male siblings. Eur J Immunol. 1996;26(9):2119–2126.
- Ma J, McCarl CA, Khalil S, Luthy K, Feske S. T-cell-specific deletion of STIM1 and STIM2 protects mice from EAE by impairing the effector functions of Th1 and Th17 cells. Eur J Immunol. 2010;40(11):3028–3042.
- Oh-Hora M, et al. Dual functions for the endoplasmic reticulum calcium sensors STIM1 and STIM2 in T cell activation and tolerance. Nat Immunol. 2008;9(4):432–443.
- Beyersdorf N, et al. STIM1-independent T cell development and effector function in vivo. J Immunol. 2009;182(6):3390–3397.
- Weidinger C, Shaw PJ, Feske S. STIM1 and STIM2-mediated Ca(2+) influx regulates antitumour immunity by CD8(+) T cells. EMBO Mol Med. 2013;5(9):1311–1321.
- Schuhmann MK, et al. Stromal interaction molecules 1 and 2 are key regulators of autoreactive T cell activation in murine autoimmune central nervous system inflammation. J Immunol. 2010;184(3):1536–1542.
- McCarl CA, et al. Store-operated Ca2+ entry through ORAI1 is critical for T cell-mediated autoimmunity and allograft rejection. J Immunol. 2010;185(10):5845–5858.
- Kaech SM, Cui W. Transcriptional control of effector and memory CD8(+) T cell differentiation. Nat Rev Immunol. 2012;12(11):749–761.View this article via: PubMed Google Scholar
- Zehn D, King C, Bevan MJ, Palmer E. TCR signaling requirements for activating T cells and for generating memory. Cell Mol Life Sci. 2012;69(10):1565–1575.
- Teixeiro E, et al. Different T cell receptor signals determine CD8+ memory versus effector development. Science. 2009;323(5913):502–505.
- Swain SL, McKinstry KK, Strutt TM. Expanding roles for CD4 T cells in immunity to viruses. Nat Rev Immunol. 2012;12(2):136–148.View this article via: PubMed Google Scholar
- Thomsen AR, Nansen A, Christensen JP, Andreasen SO, Marker O. CD40 ligand is pivotal to efficient control of virus replication in mice infected with lymphocytic choriomeningitis virus. J Immunol. 1998;161(9):4583–4590.View this article via: PubMed Google Scholar
- Cui W, Liu Y, Weinstein JS, Craft J, Kaech SM. An interleukin-21-interleukin-10-STAT3 pathway is critical for functional maturation of memory CD8+ T cells. Immunity. 2011;35(5):792–805.
- Bachmann MF, Hunziker L, Zinkernagel RM, Storni T, Kopf M. Maintenance of memory CTL responses by T helper cells and CD40-CD40 ligand: antibodies provide the key. Eur J Immunol. 2004;34(2):317–326.
- Lefrancois L, Obar JJ. Once a killer, always a killer: from cytotoxic T cell to memory cell. Immunol Rev. 2010;235(1):206–218.View this article via: PubMed Google Scholar
- Arens R, Schoenberger SP. Plasticity in programming of effector and memory CD8 T-cell formation. Immunol Rev. 2010;235(1):190–205.View this article via: PubMed Google Scholar
- Cui W, Kaech SM. Generation of effector CD8+ T cells and their conversion to memory T cells. Immunol Rev. 2010;236:151–166.
- Feau S, Arens R, Togher S, Schoenberger SP. Autocrine IL-2 is required for secondary population expansion of CD8(+) memory T cells. Nat Immunol. 2011;12(9):908–913.
- Williams MA, Tyznik AJ, Bevan MJ. Interleukin-2 signals during priming are required for secondary expansion of CD8+ memory T cells. Nature. 2006;441(7095):890–893.
- Eberlein J, Davenport B, Nguyen TT, Victorino F, Sparwasser T, Homann D. Multiple layers of CD80/86-dependent costimulatory activity regulate primary, memory, and secondary lymphocytic choriomeningitis virus-specific T cell immunity. J Virol. 2012;86(4):1955–1970.
- Fuse S, et al. Recall responses by helpless memory CD8+ T cells are restricted by the up-regulation of PD-1. J Immunol. 2009;182(7):4244–4254.
- Khanolkar A, Fuller MJ, Zajac AJ. T cell responses to viral infections: lessons from lymphocytic choriomeningitis virus. Immunol Res. 2002;26(1–3):309–321.View this article via: PubMed Google Scholar
- Butz E, Bevan MJ. Dynamics of the CD8+ T cell response during acute LCMV infection. Adv Exp Med Biol. 1998;452:111–122.
- Kaech SM, Tan JT, Wherry EJ, Konieczny BT, Surh CD, Ahmed R. Selective expression of the interleukin 7 receptor identifies effector CD8 T cells that give rise to long-lived memory cells. Nat Immunol. 2003;4(12):1191–1198.
- Huster KM, et al. Selective expression of IL-7 receptor on memory T cells identifies early CD40L-dependent generation of distinct CD8+ memory T cell subsets. Proc Natl Acad Sci U S A. 2004;101(15):5610–5615.
- Pores-Fernando AT, Zweifach A. Calcium influx and signaling in cytotoxic T-lymphocyte lytic granule exocytosis. Immunol Rev. 2009;231(1):160–173.
- Banerjee A, et al. Cutting edge: The transcription factor eomesodermin enables CD8+ T cells to compete for the memory cell niche. J Immunol. 2010;185(9):4988–4992.
- Joshi NS, et al. Inflammation directs memory precursor and short-lived effector CD8(+) T cell fates via the graded expression of T-bet transcription factor. Immunity. 2007;27(2):281–295.
- Intlekofer AM, et al. Effector and memory CD8+ T cell fate coupled by T-bet and eomesodermin. Nat Immunol. 2005;6(12):1236–1244.
- Schluns KS, Lefrancois L. Cytokine control of memory T-cell development and survival. Nat Rev Immunol. 2003;3(4):269–279.View this article via: PubMed Google Scholar
- Schluns KS, Williams K, Ma A, Zheng XX, Lefrancois L. Cutting edge: requirement for IL-15 in the generation of primary and memory antigen-specific CD8 T cells. J Immunol. 2002;168(10):4827–4831.
- Schluns KS, Kieper WC, Jameson SC, Lefrancois L. Interleukin-7 mediates the homeostasis of naive and memory CD8 T cells in vivo. Nat Immunol. 2000;1(5):426–432.
- Jin HT, et al. Cooperation of Tim-3 and PD-1 in CD8 T-cell exhaustion during chronic viral infection. Proc Natl Acad Sci U S A. 2010;107(33):14733–14738.View this article via: PubMed Google Scholar
- Wherry EJ. T cell exhaustion. Nat Immunol. 2011;12(6):492–499.View this article via: PubMed Google Scholar
- Wherry EJ, Blattman JN, Murali-Krishna K, van der Most R, Ahmed R. Viral persistence alters CD8 T-cell immunodominance and tissue distribution and results in distinct stages of functional impairment. J Virol. 2003;77(8):4911–4927.
- Shedlock DJ, Shen H. Requirement for CD4 T cell help in generating functional CD8 T cell memory. Science. 2003;300(5617):337–339.
- Sun JC, Bevan MJ. Defective CD8 T cell memory following acute infection without CD4 T cell help. Science. 2003;300(5617):339–342.
- Janssen EM, Lemmens EE, Wolfe T, Christen U, von Herrath MG, Schoenberger SP. CD4+ T cells are required for secondary expansion and memory in CD8+ T lymphocytes. Nature. 2003;421(6925):852–856.
- Koguchi Y, Thauland TJ, Slifka MK, Parker DC. Preformed CD40 ligand exists in secretory lysosomes in effector and memory CD4+ T cells and is quickly expressed on the cell surface in an antigen-specific manner. Blood. 2007;110(7):2520–2527.
- Sarawar SR, Lee BJ, Reiter SK, Schoenberger SP. Stimulation via CD40 can substitute for CD4 T cell function in preventing reactivation of a latent herpes virus. Proc Natl Acad Sci U S A. 2001;98(11):6325–6329.
- Demangel C, Palendira U, Feng CG, Heath AW, Bean AG, Britton WJ. Stimulation of dendritic cells via CD40 enhances immune responses to Mycobacterium tuberculosis infection. Infection and immunity. 2001;69(4):2456–2461.
- Rolph MS, Kaufmann SH. CD40 signaling converts a minimally immunogenic antigen into a potent vaccine against the intracellular pathogen Listeria monocytogenes. J Immunol. 2001;166(8):5115–5121.
- Murphy WJ, et al. Synergistic anti-tumor responses after administration of agonistic antibodies to CD40 and IL-2: coordination of dendritic and CD8+ cell responses. J Immunol. 2003;170(5):2727–2733.
- Hermans IF, Ritchie DS, Daish A, Yang J, Kehry MR, Ronchese F. Impaired ability of MHC class II–/– dendritic cells to provide tumor protection is rescued by CD40 ligation. J Immunol. 1999;163(1):77–81.View this article via: PubMed Google Scholar
- Bartholdy C, Kauffmann SO, Christensen JP, Thomsen AR. Agonistic anti-CD40 antibody profoundly suppresses the immune response to infection with lymphocytic choriomeningitis virus. J Immunol. 2007;178(3):1662–1670.
- Maul-Pavicic A, et al. ORAI1-mediated calcium influx is required for human cytotoxic lymphocyte degranulation and target cell lysis. Proc Natl Acad Sci U S A. 2011;108(8):3324–3329.
- Sullivan BM, Juedes A, Szabo SJ, von Herrath M, Glimcher LH. Antigen-driven effector CD8 T cell function regulated by T-bet. Proc Natl Acad Sci U S A. 2003;100(26):15818–15823.
- Sun JC, Williams MA, Bevan MJ. CD4+ T cells are required for the maintenance, not programming, of memory CD8+ T cells after acute infection. Nat Immunol. 2004;5(9):927–933.
- Andreasen SO, Christensen JE, Marker O, Thomsen AR. Role of CD40 ligand and CD28 in induction and maintenance of antiviral CD8+ effector T cell responses. J Immunol. 2000;164(7):3689–3697.
- Bachmann MF, et al. Cutting edge: distinct roles for T help and CD40/CD40 ligand in regulating differentiation of proliferation-competent memory CD8+ T cells. J Immunol. 2004;173(4):2217–2221.
- Sun JC, Bevan MJ. Cutting edge: long-lived CD8 memory and protective immunity in the absence of CD40 expression on CD8 T cells. J Immunol. 2004;172(6):3385–3389.
- Behrens G, et al. Helper T cells, dendritic cells and CTL Immunity. Immunol Cell Biol. 2004;82(1):84–90.
- Reis e Sousa C. Dendritic cells in a mature age. Nat Rev Immunol. 2006;6(6):476–483.View this article via: PubMed Google Scholar
- Lindgren H, Axcrona K, Leanderson T. Regulation of transcriptional activity of the murine CD40 ligand promoter in response to signals through TCR and the costimulatory molecules CD28 and CD2. J Immunol. 2001;166(7):4578–4585.
- Porter CM, Clipstone NA. Sustained NFAT signaling promotes a Th1-like pattern of gene expression in primary murine CD4+ T cells. J Immunol. 2002;168(10):4936–4945.
- Brunner M, Zhang M, Genin A, Ho IC, Cron RQ. A T-cell-specific CD154 transcriptional enhancer located just upstream of the promoter. Genes Immun. 2008;9(7):640–649.
- Koguchi Y, Gardell JL, Thauland TJ, Parker DC. Cyclosporine-resistant, Rab27a-independent mobilization of intracellular preformed CD40 ligand mediates antigen-specific T cell help in vitro. J Immunol. 2011;187(2):626–634.
- Borrow P, et al. CD40L-deficient mice show deficits in antiviral immunity and have an impaired memory CD8+ CTL response. J Exp Med. 1996;183(5):2129–2142.
- Feske S. Immunodeficiency due to defects in store-operated calcium entry. Ann N Y Acad Sci. 2011;1238:74–90.
- Feske S, Draeger R, Peter HH, Eichmann K, Rao A. The duration of nuclear residence of NFAT determines the pattern of cytokine expression in human SCID T cells. J Immunol. 2000;165(1):297–305.
- Luo C, et al. Recombinant NFAT1 (NFATp) is regulated by calcineurin in T cells and mediates transcription of several cytokine genes. Mol Cell Biol. 1996;16(7):3955–3966.View this article via: PubMed Google Scholar
- Oestreich KJ, Yoon H, Ahmed R, Boss JM. NFATc1 regulates PD-1 expression upon T cell activation. J Immunol. 2008;181(7):4832–4839.
- Feske S. ORAI1 and STIM1 deficiency in human and mice: roles of store-operated Ca2+ entry in the immune system and beyond. Immunol Rev. 2009;231(1):189–209.
- von Herrath M, Whitton JL. Animal models using lymphocytic choriomeningitis virus. Curr Protoc Immunol. 2001;Chapter 19:Unit 19.View this article via: PubMed Google Scholar
- Boettler T, et al. OX40 facilitates control of a persistent virus infection. PLoS Pathog. 2012;8(9):e1002913.
- Feske S. Calcium signalling in lymphocyte activation and disease. Nat Rev Immunol. 2007;7(9):690–702.
-
Version history
- Version 1 (August 26, 2014): No description
- Version 2 (October 1, 2014): No description



Copyright © 2024 American Society for Clinical Investigation
ISSN: 0021-9738 (print), 1558-8238 (online)
