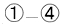|
|
1.IntroductionSaliva, which is produced by the salivary glands, has antibacterial properties that support oral health. Saliva also plays an important role in taste perception by dissolving the chemicals in food, enabling the flavor of the food to be detected. Because taste stimuli affect salivation,1, 2, 3, 4, 5 evaluation of saliva secretion (changes in stimulated saliva volume) could lead to the development of more objective gustatory and organoleptic tests. Salivary gland function is conventionally examined using imaging modalities, such as scintigraphy,6 magnetic resonance imaging (MRI),7 and ultrasonography.8 However, these techniques are somewhat invasive, high in cost, and impractical for daily use. The alternative methods are collecting saliva directly with a sampling device such as a Salivette® (cotton device)9 or using the passive drool method.10 Because these methods do not allow for the swallowing of saliva during measurement, they are not satisfactory for measuring saliva volume changes during organoleptic testing, which involves ingestion of drink or food. A method for estimating changes in saliva volume is thus needed that allows the participant to swallow, does not constrain the participant, and involves minimal cost. Our approach to meeting these conditions is to use near-infrared spectroscopy (NIRS), which is commonly used for measurements of biological metabolism, such as changes in hemoglobin (Hb) species concentration,11, 12, 13, 14 to evaluate changes in blood flow coupled with salivation. Because the NIRS technique can be used to noninvasively measure hemodynamic signals (relative changes in oxygenated and deoxygenated Hb concentration: oxy-Hb signal and deoxy-Hb signal) related to brain functional activity,15, 16 it should be able to detect vascular responses around the head and face accompanying saliva secretion.17 2.MethodTo evaluate the feasibility of this approach, we conducted three experiments. In the first experiment, we used NIRS measurement to simultaneously measure Hb signals around the temple areas and collected saliva volume to determine whether changes in the Hb signals in response to taste stimuli (Hb responses) were proportional to the saliva volume (experiment 1). We conducted the measurements around the temples because our pilot study indicated that conducting them at a more preferable position, anterior to the ears corresponding to the position of the parotid gland, is difficult due to the muscular motion accompanying stimulus intake. The second experiment was almost the same as the first except that the participants did not expel the mouth contents, but swallowed them, enabling us to investigate the effects of swallowing on Hb responses (experiment 2). Finally, we used functional magnetic resonance imaging (fMRI) to determine possible source regions of the Hb responses corresponding to changes in the stimulated saliva volume (experiment 3). 2.1.Experiment 12.1.1.ParticipantsFive healthy adults (two female and three male, aged 30–34) participated in experiment 1. The participants gave written informed consent before the experiment with approval from the Ethics Committee of Hitachi, Ltd. 2.1.2.Measurement sequenceA participant was seated facing a desk on which four plastic glasses were filled with 15 ml of either distilled water (water condition: W) or a sucrose solution (concentration: 96 g/l) (sweet condition: S). The tasting sequence consisted of three steps: (a) holding the glass with the right hand and raising the glass to the mouth, (b) 4 s later, taking the contents of the glass into the mouth, (c) another 4 s later, expelling the contents of the mouth back into the glass and returning the glass to the desk (Fig. 1). Each step was triggered by presenting a tone [(a) 1600 Hz, 200 ms; (b) 1000 Hz, 200 ms; and (c) 1600 Hz, 200 ms]. Two glasses of distilled water and two glasses of sucrose solution were used as taste stimuli in each measurement session. These stimuli were presented in the order of either W-S-W-S or S-W-S-W to counterbalance any order effects. To compare the Hb signals and volume of saliva secreted in response to the taste stimuli, the participants were instructed to empty the contents of their mouth into the same glass that had contained the water or sucrose solution. The glass was weighed before and after the measurement on a digital scale (ScoutTM Pro, Phaus Corporation, Parsippany, NJ, USA), and the change in the weight was defined as the saliva volume. Fig. 1Schematic diagrams of measurement sequence.
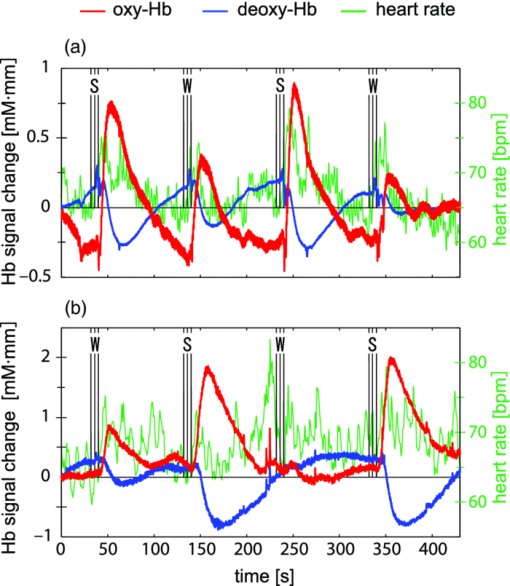
 2.1.3.Near-infrared spectroscopy systemWe used an optical topography system (ETG-7100, Hitachi Medical Corporation, Tokyo, Japan), which has eight irradiation positions and seven detection positions, making 22 source-detector pairs (channels: Chs) on each side of the head [Fig. 2a]. The midpoint between a source and a detector was regarded as a measurement (Ch) position. The system simultaneously irradiated light at 695±20 and 830±20 nm wavelengths through an optical fiber to an irradiation position. The reflected light was detected every 100 ms by using an avalanche photodiode through an optical fiber located 30 mm from the incident position. Optical fibers were used for both irradiating and detecting the lights. The average power of each light source was 1.5 mW, and each source was modulated at a distinctive frequency (1–10 kHz) to enable signal separation using a lock-in amplifier after detection. Fig. 2Response maps of Hb signals and representative time-courses of Hb responses in experiment 1: (a) Arrangement of incident, detection, and measurement positions (channels) and (b,c) maps of Oxy-Hb signals in response to (b) distilled water and (c) sucrose solution. Response value for each channel is indicated on color scale to right. Channels showing significant increases are marked with circles (p < 0.05). (d) Mean Hb signal changes (thick lines show oxy-Hb signals and thin lines show deoxy-Hb signals) in response to taste stimuli for channel 21 on the left side [(A–C) timing of taste stimuli (see Fig. 1)]. Error bars show standard error. 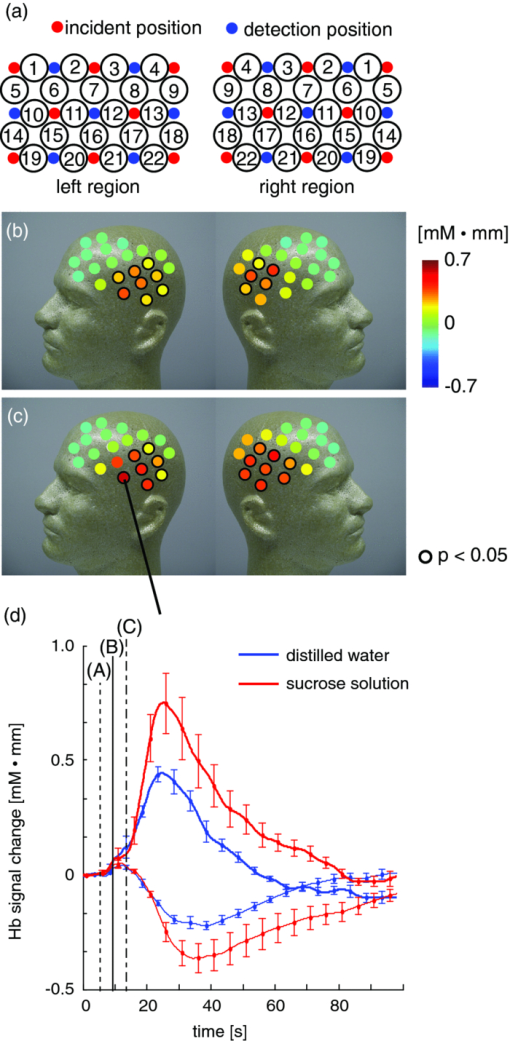 2.1.4.Data analysisThe NIRS data were processed using in-house software developed on MATLAB (Mathworks, Natick, MA, USA). The Hb signal, based on the obtained light signals at the two wavelengths, was calculated using the modified Lambert–Beer law. 17a The oxy- and deoxy-Hb signals (ΔC ′ oxy and ΔC ′ deoxy), which are the products of the concentration changes of the independent hemoglobin species (mM) and the effective optical path length (mm), were calculated using Eq. 1[TeX:] \documentclass[12pt]{minimal}\begin{document}\begin{equation} \Delta C'_{\rm oxy} = L\Delta C_{\rm oxy} = \frac{{ - \varepsilon _{{\rm deoxy} (\lambda 2)}\Delta A_{\rm (\lambda 1)}} + \varepsilon _{{\rm deoxy} {\rm (\lambda 1)}} \Delta A_{\rm (\lambda 2)}}{E}, \end{equation}\end{document}Eq. 2[TeX:] \documentclass[12pt]{minimal}\begin{document}\begin{equation} \Delta C'_{{\rm deoxy}} = L\Delta C_{{\rm deoxy}} = \frac{{\varepsilon _{{\rm oxy (}\lambda {\rm 2)}} \Delta A_{{\rm (}\lambda {\rm 1)}} - \varepsilon _{{\rm oxy (}\lambda {\rm 1)}} \Delta A_{{\rm (}\lambda {\rm 2)}} }}{E}, \end{equation}\end{document}Eq. 3[TeX:] \documentclass[12pt]{minimal}\begin{document}\begin{equation} E = \varepsilon _{{\rm deoxy (}\lambda {\rm 1)}} \varepsilon _{{\rm oxy (}\lambda {\rm 2)}} - \varepsilon _{{\rm deoxy }(\lambda {\rm 2)}} \varepsilon _{{\rm oxy (}\lambda {\rm 1)}}. \end{equation}\end{document}After the oxy- and deoxy-Hb signals were obtained, the oxy-Hb signals were processed as follows:
To evaluate the relationship between saliva volume and hemodynamic changes, we calculated Pearson's correlation coefficients between the weight of the collected saliva and the response values for each participant for each channel. 2.2.Experiment 22.2.1.ParticipantsThirteen healthy volunteers took part in experiment 2. As in experiment 1, they gave written informed consent before the experiment with approval from the Ethics Committee of Hitachi, Ltd. The data for two participants were excluded from the analysis because one had a fit of coughing during the experiment and the other took the solutions in the wrong order. The data used was thus from 11 participants (six male and five female, aged 24–40). 2.2.2.Measurement sequenceThe task sequence was almost the same as that in experiment 1 except that the participants did not expel but swallowed the mouth contents. That is, the participants swallowed the mouth contents 4 s after taking the glass contents into their mouth [Fig. 1c]. In addition, each participant had one more measurement trial in which the participant swallowed his or her own saliva without an intake of drink. Accordingly, we had three conditions: (i) a sweet condition (S) in which 15 ml of sucrose solution (96 g/l) was consumed, (ii) a water condition (W) in which 15 ml of distilled water was consumed, and (iii) a dummy condition (D) in which the participant swallowed his or her own saliva. Each participant had two measurement trials, one consisted of four times (D) [D-D-D-D] and another consisted of two alternations of (S) and (W) [S-W-S-W or W-S-W-S], with an interstimulation interval of 90 s. 2.2.3.Data analysisThe NIRS data were analyzed in the same manner as in experiment 1 until a response value was calculated (processes 1–5 above) for each condition. Across-participant paired t-tests (two samples) were then used to compare the response values between conditions—between (S) and (D), between (W) and (D), and between (S) and (W). 2.3.Experiment 32.3.1.ParticipantsSix healthy volunteers (two female and four male, aged 22–34) participated in the fMRI experiment (experiment 3) after giving written informed consent, which was approved by the Ethics Committee of the Ogawa Laboratories for Brain Function Research. 2.3.2.Measurement sequenceThe participants lay supine on a scanner bed, wore a headset for listening to the fMRI instructions, and held a mouthpiece between their lips. Two silicone tubes (LABORAN, 6 mm o.d., 4 mm i.d.; AS ONE Corporation, Osaka, Japan) were connected to the mouthpiece for delivering distilled water (water condition: W) or a sucrose solution (concentration: 96 g/l). The task sequence was the same as in experiment 2 except that it did not include the dummy condition (two alternations of (S) and (W) [S-W-S-W or W-S-W-S], with an interstimulation interval of 90 s). In addition, the amount of taste stimuli was reduced to 5 ml because swallowing 15 ml of liquid is difficult in a supine position. An experimenter standing by the scanner delivered the taste stimuli by using syringes. 2.3.3.MRI systemAll imaging was performed at the Ogawa Laboratories for Brain Function Research (Tokyo, Japan) using a 3-Tesla MRI system (Siemens Allegra, Erlangen, Germany). Functional images [blood-oxygenation-level-dependent (BOLD) signals] were collected in 30 horizontal slices covering the upper parts of the head with echo planar imaging software (voxel size: 3.6 × 3.6 × 5.0 mm; field of view (FOV): 280 × 280 mm; repetition time (TR): 1500 ms; echo time (TE): 20 ms; flip angle (FA): 90 deg). T1-weighted high-resolution anatomical images were also acquired in an additional run (voxel size: 0.9 × 0.9 × 5.0 mm; FOV: 280 × 280 mm; TR: 500 ms; TE: 9.1 ms; FA: 90°). 2.3.4.Data analysisWe analyzed the MRI data for each participant by using SPM2 software (Wellcome Department of Cognitive Neurology, University College London, United Kingom). First, the functional images were aligned with the first image and corrected for slice acquisition timing relative to the middle slice in each scan. The data, including the entire volume, were scaled to the grand mean of each slice and were smoothed with a Gaussian kernel (6 mm full width at half maximum). Next, to identify the source regions of the Hb responses shown in experiments 1 and 2, the oxy-Hb signal waveform obtained in experiment 2 was used as a regressor in a general linear model for statistical estimation. A statistical threshold was basically set to p < 0.05, using familywise error correction and spatial extent threshold (the areas of five or more contiguous significant voxels were defined as activation regions). 3.Results3.1.Experiment 1Both taste stimuli (distilled water and sucrose solution) induced a significant increase in the oxy-Hb signals (oxy-Hb response) around the temples [Figs. 2b, 2c]. The representative time course of the oxy-Hb response showed a peak with a latency of ∼20 s, and it took about 30–50 s to return to the baseline after the peak [Fig. 2d, thick lines]. The mean response value for the distilled water was ∼0.38 mM·mm, whereas that for the sucrose solution was ∼0.59 mM·mm. In contrast, the deoxy-Hb signals showed a significant decrease in response to the taste stimuli [Fig. 2d, thin lines] with the same tendency of larger amplitudes of response for the sucrose solution than for the distilled water. However, the deoxy-Hb responses were slower and smaller than the oxy-Hb responses; thus, we focused on the oxy-Hb signals in this study. The volumes of stimulated saliva corresponding to each taste stimuli are listed in Table 1. The paired t-test revealed that the volume corresponding to the sucrose solution was significantly higher than that corresponding to the distilled water [Fig. 3a; t = 2.82, p < 0.05]. This is consistent with the comparison of oxy-Hb responses around the temples [Fig. 2d]; thus, both the saliva volume and oxy-Hb response depended on the taste intensity (in this case, sweetness). Table 1Saliva volume (g) and representative channel by participant. Saliva volume was defined as the weight change (g) of a glass after the participant expelled his or her mouth contents into the glass (see 2. Method, 2.1 Experiment 1). The representative channel used for correlation analysis in experiment 1 is shown in the right column.
To confirm this finding, we performed correlation analysis between the saliva volume and response values for the oxy-Hb signals for each channel. The channel showing the highest correlation coefficient was located on the right temple (ch 21: r = 0.62, p < 0.005). Because the channel showing the highest correlation coefficient differed among participants, we selected a representative channel among the channels in the temple area (ch 18, 21, or 22) for each participant for further analysis (Table 1). The response values for the representative channels for the sucrose solution were larger than those for the distilled water [Fig. 3b; t = 3.28, p < 0.01], and the correlation coefficient between the saliva volume and response value was 0.81 (p < 0.0001) [Fig. 3c]. 3.2.Experiment 2We isolated the hemodynamic changes due to the sweet taste on the basis of the experimental design and analysis, in which the response values of the oxy-Hb signals were compared for the taste conditions. As shown in Fig. 4, significant oxy-Hb responses were found in the areas around the temples for both (S) and (W) relative to (D) (paired t-test, p < 0.01). Moreover, larger responses for (S) compared to (W) were found (p < 0.01) in a confined region [Fig. 4c]. The oxy-Hb responses showed a peak of ∼0.7 mM·mm with a latency of 19 s on average, and it took about 50–60 s to return to the baseline after the peak [Fig. 4d, thick lines]. Although motion artifacts were occasionally seen for the swallowing timing, the Hb signals in response to the taste stimuli were about 2–7 times larger and longer than the artifacts. In addition, the slower and smaller deoxy-Hb signals showed the same tendency in terms of the response amplitudes, depending on the stimuli conditions [Fig. 4d, thin lines]. Fig. 4Response maps of Hb signals and representative time-courses of Hb signals in experiment 2: (a) Comparison between water condition and dummy condition, (b) comparison between sweet condition and dummy condition, and (c) comparison between sweet condition and water condition. Channels showing significant increases are mar-ked with circles (p < 0.01). (d) Mean Hb signal changes (thick lines show oxy-Hb signals and thin lines show deoxy-Hb signals) in response to taste stimuli and dummy condition for channel 21 on the left side. Error bars show standard error.  We also calculated the heart rate changes from the Hb signals19 to compare the Hb responses with the patterns of heart rate change (Fig. 5). A moderate increase in the heart rate (HR) was seen after drinking either distilled water or sweetened water (5–7 bpm), but there was no correlation between the HR increase and oxy-Hb responses both for (S) (r = −0.01, p = 0.85) and (W) (r = 0.08, p = 0.11). 3.3.Experiment 3The fMRI analysis revealed activation in the extracranial regions [Fig. 6a] that might have been the source of the Hb responses observed in experiments 1 and 2. According to anatomical references,20, 21 the extracranial region likely expands into the temporal muscle, the temporal and sphenoid bone, the lateral and middle pterygoid muscle, and the pterygoid venous plexus close to the parotid gland. In particular, significant activation was commonly found around the frontal branch of the middle meningeal artery in the sphenoid bone20, 21 for all participants Fig. 6b]. Representative BOLD signals for a single trial (Fig. 7) indicate that there were larger responses to (S) than to (W), the same as indicated by the Hb signals. Fig. 6Activation regions corresponding to time course of NIRS signal revealed by fMRI analysis (p < 0.0001 with familywise error correction): (a) Activation regions for a representative participant. Global maximum is shown by cross point of blue lines in sagittal slice image, coronal slice image, and horizontal slice image. (b) Horizontal slice images of common activation regions in all six participants (A–F). Global maximums were found in these activation regions for four participants (A,B,C,E). 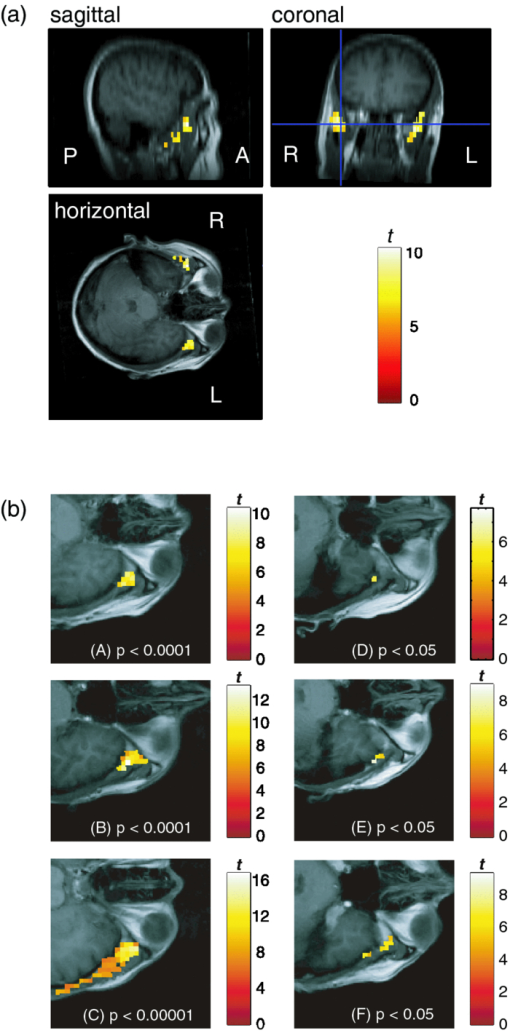 Fig. 7Representative time courses of BOLD signals in single trial. Examples of sequence (a) in W-S-W-S and (b) in S-W-S-W. Open and closed circles indicate presentation timing of distilled water and sucrose solution, respectively. Signal value in first scan was set to zero to correct baseline, and moving average of five scans (7.5 s) was used for smoothing. 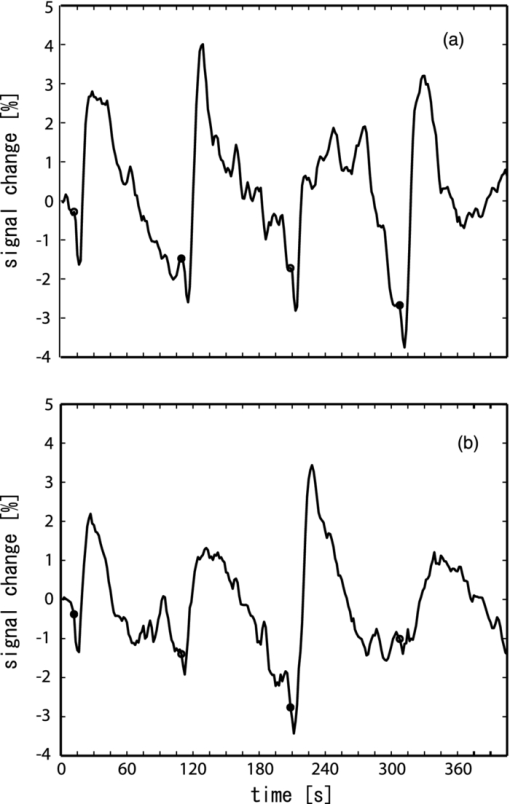 4.DiscussionTo our knowledge, this is the first report suggesting the use of NIRS to measure hemodynamic response accompanying saliva secretion under natural conditions in humans. Although NIRS cannot quantify the absolute value of the response due to indeterminable coefficient L [Eq. 1], the results for experiment 1 revealed a strong relationship between the oxy-Hb response and the saliva volume (Fig. 3). The correlation between the oxy-Hb response and the salivation volume when the volume was less than about 4.5 g was unclear. Nevertheless, our findings suggest that the oxy-Hb response reflects to a certain degree the change in the volume of stimulated saliva. Fig. 3Comparison of saliva volume and response value in Oxy-Hb signal: (a) Estimated saliva volume for two taste stimuli, (b) Oxy-Hb response value for two taste stimuli, and (c) correlation between estimated saliva volume and oxy-Hb response value. 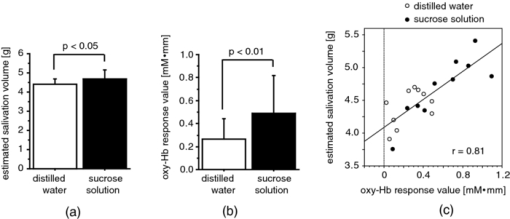 Similar Hb responses were reproduced in experiment 2 in which the participants did not expel but swallowed the mouth contents. As shown in Fig. 4d, there was a dip or a bump in the Hb time courses during the drinking motion, which is not evident in the time courses for experiment 1. We attribute it to the swallowing motion (i.e., it was an artifact of the experiment). It had a negligible effect, however, as additional analysis (Fig. 5) revealed that the effects of swallowing were small compared to the hemodynamic responses corresponding to the relative changes in saliva volume. The analysis also showed that the heart rate changes were not correlated to the responses. In addition, the fMRI experiment identified extracranial regions as a possible source for the Hb responses, although the exact source region could not be determined. Taken together, these findings suggest that NIRS can be applied to indirect measurement of relative volume changes in stimulated saliva during drinking. We speculate that blood supply to the parotid gland is the cause of the Hb responses corresponding to changes in the volume of stimulated saliva. It has been reported that stimulation of the preganglionic parasympathetic nerve increases salivary gland arterial flow, accompanied by copious salivary secretion.17 The Hb responses we observed were about 2–7 times larger than typical ones in the cerebral cortex.22 It is possible that these remarkably large and slow responses reflect the blood flow and oxygen consumption related to salivary secretion, which needs high-energy consumption. Many blood vessels are involved in the blood supply to the parotid gland, and the hemodynamic changes in these vessels have not yet been fully explained. There are arterial branches of the external carotid artery, the maxillary artery, the middle meningeal artery, and the superficial temporal artery around the parotid gland,20, 21 although there is a great individual difference in the allocation of these branches. Further studies are necessary to clarify the physiological mechanism of the significant change in the Hb signals in response to taste stimuli. In addition, it should be noted that there were some limitations in this study. First, we used only a sucrose solution, which is the least effective in stimulating salivation among the various gustatory stimuli, for comparison to distilled water. Although we had expected that these mild stimuli were sufficient to induce salivation, we would have been able to demonstrate a much greater difference from the results with distilled water if we had used an acid or salt solution as the comparative stimulus. Second, we used a primitive method to collect saliva, which gave us only a rough estimation of the mixed saliva volume consisting of parotid, submandibular, and sublingual saliva. Because the Hb responses in the NIRS measurements should be reflected by the blood supply to the parotid gland, a precise measurement of pure parotid saliva would have been better. However, we did not use a direct-measurement method, such as using a Lashley cup23 in order to avoid any effects of an instrument inside the mouth on salivation. Future work includes the use of a more precise method for saliva collection to help us obtain stronger evidence of the correspondence between Hb response and parotid function. AcknowledgmentsWe thank Dr. Seiji Ogawa and Dr. Yoshiaki Someya (Ogawa Laboratories for Brain Function Research), Dr. Takusige Katura and Noboru Moriya (Hitachi, Ltd., Advanced Research Laboratory), Dr. Hiroaki Kawamichi (Hitachi, Ltd., Systems Development Laboratory) for their technical assistance in the fMRI experiments; Dr. Masato Yasui and Dr. Yoshiro Soma (Keio University), Dr. Masanobu Kumada (Kumada Clinic), and Dr. Toshio Kaji, Dr. Joji Suzuki, and Dr. Takashi Shinagawa (Hitachi Yokohama Hospital) for their valuable contributions during our discussions, and Yuichi Mori and Kiyoshi Hasegawa (Business Incubation Division, Hitachi, Ltd.) for their helpful support. ReferencesC. Dawes,
“The composition of human saliva secreted in response to a gustatory stimulus and to pilocaprine,”
J. Physiol., 183
(2), 360
–368
(1966). Google Scholar
C. Dawes and
G. N. Jenkins,
“The effects of different stimuli on the composition of saliva in man,”
J. Physiol., 170 86
–100
(1964). Google Scholar
C. Dawes and
S. Watanabe,
“The effect of taste adaptation on salivary flow rate and salivary sugar clearance,”
J. Dent. Res., 66
(3), 740
–744
(1987). https://doi.org/10.1177/00220345870660030701 Google Scholar
S. Watanabe and
C. Dawes,
“A comparison of the effects of tasting and chewing foods on the flow rate of whole saliva in man,”
Arch. Oral Biol., 33
(10), 761
–764
(1988). https://doi.org/10.1016/0003-9969(88)90010-6 Google Scholar
S. Watanabe and
C. Dawes,
“The effects of different foods and concentrations of citric acid on the flow rate of whole saliva in man,”
Arch. Oral Biol., 33
(1), 1
–5
(1988). https://doi.org/10.1016/0003-9969(88)90089-1 Google Scholar
W. Aung, Y. Murata, R. Ishida, Y. Takahashi, N. Okada, and
H. Shibuya,
“Study of quantitative oral radioactivity in salivary gland scintigraphy and determination of the clinical stage of Sjogren's syndrome,”
J. Nucl. Med., 42
(1), 38
–43
(2001). Google Scholar
S. C. Simon-Zoula, C. Boesch, F. De Keyzer, and
H. C. Thoeny,
“Functional imaging of the parotid glands using blood oxygenation level dependent (BOLD)-MRI at 1.5T and 3T,”
J. Magn. Reson. Imaging, 27
(1), 43
–48
(2008). https://doi.org/10.1002/jmri.21071 Google Scholar
C. Martinoli, L. E. Derchi, L. Solbiati, G. Rizzatto, E. Silvestri, and
M. Giannoni,
“Color Doppler sonography of salivary glands,”
Am. J. Roentgenol., 163
(4), 933
–941
(1994). Google Scholar
L. Strazdins, S. Meyerkort, V. Brent, R. M. D’Souza, D. H. Broom, and
J. M. Kyd,
“Impact of saliva collection methods on sIgA and cortisol assays and acceptability to participants,”
J. Immunol. Methods, 307
(1-2), 167
–171
(2005). https://doi.org/10.1016/j.jim.2005.09.010 Google Scholar
D. A. Granger, K. T. Kivlighan, C. Fortunato, A. G. Harmon, L. C. Hibel, E. B. Schwartz, and
G. L. Whembolua,
“Integration of salivary biomarkers into developmental and behaviorally-oriented research: problems and solutions for collecting specimens,”
Physiol. Behav., 92
(4), 583
–590
(2007). https://doi.org/10.1016/j.physbeh.2007.05.004 Google Scholar
F. F. Jobsis,
“Noninvasive, infrared monitoring of cerebral and myocardial oxygen sufficiency and circulatory parameters,”
Science, 198
(4323), 1264
–1267
(1977). https://doi.org/10.1126/science.929199 Google Scholar
F. F. Jobsis,
“Non-invasive, infra-red monitoring of cerebral O2 sufficiency, bloodvolume, HbO2-Hb shifts and bloodflow,”
Acta Neurol. Scand. Suppl., 64 452
–453
(1977). Google Scholar
F. F. Jobsis-VanderVliet, C. A. Piantadosi, A. L. Sylvia, S. K. Lucas, and
H. H. Keizer,
“Near-infrared monitoring of cerebral oxygen sufficiency. I. spectra of cytochrome c oxidase,”
Neurol. Res., 10
(1), 7
–17
(1988). Google Scholar
S. Wray, M. Cope, D. T. Delpy, and
J. S. Wyatt,
“Characterization of the near infrared absorption spectra of cytochrome aa3 and haemoglobin for the non-invasive monitoring of cerebral oxygenation,”
Biochim. Biophys. Acta, 933
(1), 184
–192
(1988). https://doi.org/10.1016/0005-2728(88)90069-2 Google Scholar
B. Chance, Z. Zhuang, C. UnAh, C. Alter, and
L. Lipton,
“Cognition-activated low-frequency modulation of light absorption in human brain,”
Proc. Natl. Acad. Sci. USA, 90
(8), 3770
–3774
(1993). https://doi.org/10.1073/pnas.90.8.3770 Google Scholar
A. Villringer, J. Planck, C. Hock, L. Schleinkofer, and
U. Dirnagl,
“Near infrared spectroscopy (NIRS): a new tool to study hemodynamic changes during activation of brain function in human adults,”
Neurosci. Lett., 154
(1-2), 101
–104
(1993). https://doi.org/10.1016/0304-3940(93)90181-J Google Scholar
M. A. Lung,
“Variations in blood flow on mandibular glandular secretion to autonomic nervous stimulations in anaesthetized dogs,”
J. Physiol., 431 479
–493
(1990). Google Scholar
D. T. Delpy,M. Cope, P. van der Zee, S. Arridge, S. Wray and
J. Wyatt,
“Estimation of optical pathlength through tissue from direct time of flight measurement,”
Phys. Med. Biol., 33
(12), 1433
–1442
(1988). https://doi.org/10.1088/0031-9155/33/12/008 Google Scholar
H. Sato, M. Kiguchi, F. Kawaguchi, and
A. Maki,
“Practicality of wavelength selection to improve signal-to-noise ratio in near-infrared spectroscopy,”
Neuroimage, 21
(4), 1554
–1562
(2004). https://doi.org/10.1016/j.neuroimage.2003.12.017 Google Scholar
K. Yamazaki, M. Uchida, A. Obata, T. Katura, H. Sato, N. Tanaka, and
A. Maki,
“Comparison between spontaneous low-frequency oscillations in regional cerebral blood volume, and cerebral and plethysmographic pulsations,”
687
–690
(2007). Google Scholar
H.-J. Kretscmann and
W. Weinrich, Computertomographie and Magnetresonanztomographie, Igaku-Shoin Ltd., Tokyo
(1995). Google Scholar
L. Wilson-Pauwels, E. J. Akesson, P. A. Stewart, and
S. D. Spacey, Cranial Nerves in Health And Disease, Igaku-Shoin Ltd., Tokyo
(2004). Google Scholar
H. Sato, Y. Fuchino, M. Kiguchi, T. Katura, A. Maki, T. Yoro, and
H. Koizumi,
“Intersubject variability of near-infrared spectroscopy signals during sensorimotor cortex activation,”
J. Biomed. Opt., 10 044001
(2005). https://doi.org/10.1117/1.1960907 Google Scholar
K. S. Lashley,
“Reflex secretion of the human parotid gland,”
J. Exp. Psych., 1 461
–493
(1916). https://doi.org/10.1037/h0073282 Google Scholar
|