Volume 55 - Issue 4 - August 2007
Research Article
On the kinetics of ion exchange in phlogopite — An in situ AFM study
- Kirill Aldushin, Guntram Jordan, Elena Aldushina, Wolfgang W. Schmahl
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 339-347
-
- Article
- Export citation
-
The kinetics of cation exchange in phlogopite have been studied in situ by hydrothermal atomic force microscopy (HAFM). The exchange of interlayer K by octylammonium ions caused an increase in the interlayer distance and the formation of reaction fronts which can be locally resolved by AFM. The observed reaction fronts revealed substantial variations in their propagation rates — even within single interlayers. This observed variability in interlayer reactivity could mainly be attributed to chemical and structural inhomogeneities of the samples. A quantitative evaluation of the front propagation at representative sites yielded a diffusion coefficient of the K+ exchange by octylammonium of 1.2±0.6 × 10−11 cm2/s assuming negligible transport normal to the layers. The reverse reaction, i.e. the exchange of organic ions by K+, resulted in a retreat of the reaction fronts and a general restoration of the original morphological state. However, indications of structural alterations and areas with trapped octylammonium ions were found.
Adsorption of Eu3+ to smectites and fluoro-tetrasilicic mica
- Tomohiko Okada, Yusuke Ehara, Makoto Ogawa
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 348-353
-
- Article
- Export citation
-
The adsorption of Eu3+ from aqueous solution to natural Na+-montmorillonite (Kunipia F), synthetic saponite (Sumecton SA) and synthetic fluoro-tetrasilicic mica (Na+-TSM) clay samples was investigated. Adsorption capacities derived from the isotherms were 1.02, 0.71 and 1.00 meq/g of clay, respectively, for Kunipia F, Sumecton SA and Na+-TSM. The adsorption capacities were comparable to the cation exchange capacities of the clays, which were 1.19, 0.71 and 0.94 meq/g of clay, respectively. The greater slope of the TSM adsorption isotherm relative to the montmorillonite and saponite isotherms indicates a high affinity of Eu3+ for Na+-TSM. The high affinity of TSM for Eu3+ was thought to be related to the large electronegativity of the octahedral fluorine groups in TSM. Photoluminescence of adsorbed Eu3+ was observed for saponite and TSM, but not for montmorillonite. Quenching of Eu3+ luminescence by Fe in the montmorillonite structure is the probable reason for this phenomenon. The luminescence intensity varied with the amount of adsorbed Eu3+ for saponite and TSM as a result of self-quenching.
Eu incorporation behavior of a Mg-Al-Cl layered double hydroxide
- Hilde Curtius, Kristian Ufer
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 354-360
-
- Article
- Export citation
-
From leaching experiments with metallic uranium-aluminum research reactor fuel elements in repository-relevant MgCl2-rich salt brines, a Mg-Al layered double hydroxide (LDH) with chloride as the interlayer anion was identified as a crystalline secondary phase component. The incorporation behavior of europium into the structure of the Mg-Al-Cl LDH was investigated. Synthesis via co-precipitation was performed. The Mg-Al-Eu-Cl LDH obtained was treated with a concentrated ammonium carbonate solution. No release of Eu was detected; hence the molar stoichiometry of the LDH remained stable with respect to Mg, Al and Eu. This chemical behavior might be the first indication of the incorporation of Eu.
The material was further examined by powder X-ray diffraction. Structural parameters were obtained from comparisons of simulated and experimental diffraction patterns of a ${\rm{CO}}_3^{2 - }$
 -exchanged Mg-Al-Eu LDH and a Mg-Al LDH. The two materials showed different behaviors according to stacking order and lattice parameters. This is an indirect indication of the incorporation of Eu.
-exchanged Mg-Al-Eu LDH and a Mg-Al LDH. The two materials showed different behaviors according to stacking order and lattice parameters. This is an indirect indication of the incorporation of Eu.
The effect of amino acids on the dissolution rates of amorphous silica in near-neutral solution
- Motoharu Kawano, Sumine Obokata
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 361-368
-
- Article
- Export citation
-
Amino acids are ubiquitous in the Earth’s surface environments as reactive biological molecules produced by every living thing including bacteria. To evaluate the effects of amino acids on mineral dissolution and to reveal the mechanism by which they interact with the mineral surface, we performed dissolution experiments of X-ray amorphous silica in solution containing 0.1 mmol Na with 10.0 mmol amino acids such as cysteine, asparagine, serine, tryptophan, alanine, threonine, histidine, lysine and arginine in near-neutral solutions. Dissolution experiments in solutions of 0.1, 1.0 and 10.0 mmol NaCl without amino acids were also conducted as amino acid-free controls. The results of this study indicate that basic amino acids such as histidine, lysine and arginine can interact more strongly with the negatively charged surface of amorphous silica than other non-basic amino acids due to their greater dissociation, thus forming cationic species. This electrostatical interaction enhanced dissolution rates of amorphous silica by approximately one order of magnitude compared with amino acid-free controls. In contrast, no significant effect on the dissolution rates of amorphous silica was observed in solutions containing cysteine, asparagine, serine, tryptophan, alanine and threonine because of lesser interaction with the surface of amorphous silica.
Combined effects of tannate and ageing on structural and surface properties of aluminum precipitates
- G. Yu, U. K. Saha, L. M. Kozak, P. M. Huang
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 369-379
-
- Article
- Export citation
-
The influence of organics on the crystallization of Al precipitates has been well documented. However, the effects of organics and ageing on the transformation and structural configuration of Al precipitates in relation to their surface and charge properties are not fully understood. This study investigated the structural, microporous and surface properties of Al precipitates formed under the influence of tannate and ageing. The Al precipitates were synthesized at an initial Al concentration of 7 × 10−3 M, an OH/Al molar ratio (MR) of 3.0, and initial tannate/Al MRs of 0, 0.001, 0.01 and 0.1, and aged for 1, 10 and 40 days. As indicated by a decrease in gibbsite and bayerite and an increase in the oxalate-extractable Al contents, the non-crystalline precipitates increased with the increase of the initial tannate/Al MR. This observation is in accord with the X-ray diffraction and Fourier transform infrared (FTIR) data. The impact of tannic acid on the nature of the Al precipitates is also reflected in the increase of the contents of the pyrophosphate-extractable Al, which is indicative of organically bound Al. This observation is in agreement with the increase in the intensity of characteristic FTIR absorption bands of tannate and the organic C and adsorbed water contents. The decrease in the crystallinity of Al precipitates with increase in the tannate/Al MR resulted in the development of microporosity, increase in BET specific surface area and decrease of the average pore diameter and point of zero salt effect (PZSE). The FTIR absorption bands characteristic of tannate of the Al precipitates became weaker with ageing, in accord with the ageing-induced decrease in the contents of organic C and pyrophosphate-extractable Al. Ageing drastically decreased the BET specific surface area of the Al precipitates formed in the absence of tannate but this effect was less conspicuous for the products formed at the tannate/Al molar ratio of 0.1. The ageing-induced change in the PZSE of the Al precipitates formed both in the absence and presence of tannate was not significant. The results accomplished in this study are of fundamental significance to our understanding of the combined effects of organics and ageing on structural configuration of hydrolytic precipitates of Al in relation to their microporosity, surface and charge properties in the environment.
Soil Hydroxy-interlayered minerals: A re-interpretation of their crystallochemical properties
- Alain Meunier
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 380-388
-
- Article
- Export citation
-
Hydroxy-interlayered minerals (HIMs) are typical of moderately acidic soils. Barnhisel and Bertsch (1989) defined the hydroxy-interlayered clay minerals as a solid-solution series between smectite, vermiculite and pedogenic or aluminous chlorite end-members. Their experimental data for the relationship between the decrease in cation exchange capacity (CEC) and the amount of Al fixed in the interlayers of smectites and vermiculites is reinterpreted using calculated structural, chemical and X-ray diffraction (XRD) evidence. The adsorbed Al ions are in a 6-fold coordination state: [Al(OH)x(H2O)y](3−x)+ with x+y = 6. The polymerization process occurs before saturation of the exchange sites by Al ions. Some of the adsorbed Al ions form polynuclear cations keeping a constant positive charge.
X-ray diffraction patterns of oriented preparations in the ethylene glycol-solvated state suggest that HIMs consist of randomly interstratified expandable and chlorite-like layers (17 and 14.2 Å). Chlorite-like layers result from the selective adsorption of Al complex ions in specific interlayer zones that behave similarly to Al-chlorite (donbassite-like) with incomplete (60%) ‘gibbsite-like’ sheets (chlorite60). Using this framework, HIM XRD patterns can be interpreted by comparison with calculated chlorite60-dismectite mixed-layer mineral patternss using the NEWMOD software.
Weathering sequences of clay minerals in soils along a serpentinitic toposequence
- Z. Y. Hseu, H. Tsai, H. C. Hsi, Y. C. Chen
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 389-401
-
- Article
- Export citation
-
There has been limited research on clay mineral transformation in serpentinitic soils under humid tropical conditions. In this study, four soil pedons were selected along a toposequence from the summit (Entisol), shoulder (Vertisol), backslope (Alfisol) to footslope (Ultisol) positions to explore the contributions and the significance of landscape and weathering status of serpentinitic rock with regard to clay mineral transformations in eastern Taiwan. Experimental results indicated that the large amount of dithionite-citrate-bicarbonate-extractable Fe (Fed) and clay in the subsurface horizon were mainly caused by the strong leaching potential from intensive rainfall and weathering of the fine-grained parent rocks. The clay mineralogy reflected the clear weathering trend of the soils along the toposequence: (1) the soils on the summit and shoulder contained smectite and serpentine, which are predominant in the young soils derived from serpentinitic rocks; and (2) vermiculite gradually increased in the relatively old soils on backslope and footslope. The mineralogical transformations observed along the toposequence indicated that chlorite and serpentine, initially present in the Entisol on the summit, weather into smectite and interstratified chlorite-vermiculite in the intermediate soil on the shoulder under strong leaching and oxidizing conditions. Furthermore, vermiculite formed as the major weathering product of chlorite and smectite in the soil developed on the backslope. In addition to vermiculite, kaolinite and quartz formed in the soils on the footslope with the greatest concentration of Fed along the toposequence.
Mineralogy, geochemistry and genesis of smectite in pliocene volcaniclastic rocks of the doğanbey formation, beyşehir basin, Konya, Turkey
- Selahattin Kadir
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 402-422
-
- Article
- Export citation
-
Pliocene volcaniclastic lacustrine rocks of the Doğanbey formation in the Beyşehir region (central Anatolia) are composed of organic-matter-bearing claystone, clastic units and dolomite interbeds, suggesting an anaerobic, shallow swampy, lacustrine depositional environment. The depositional environment was subjected to periodic climatic change during which diagenesis occurred, and smectite, and locally palygorskite, were precipitated. Smectite flakes formed authigenically on feldspar and palygorskite fibers between dolomite rhombs and at the edges of smectite flakes. Increases in leaching of Na, K, Sr, Ba and Rb, increasing Al/Si ratios, and Fe with increasing degree of alteration reveal that hydration of volcaniclastic grains (feldspar, glass) by meteoric water — determined from O and H isotopic values — was the main cause of precipitation of beidellite and montmorillonite based on the tetrahedral charge/octahedral charge ratio, with average structural formulae of (Si7.72Al0.28)(Al3.20Fe0.53Mg0.25 Mn0.02Ti0.04) (Ca0.11Na0.09K0.11), and (Si7.88Al0.13)(Al3.18Fe0.53Mg0.18Mn0.02Ti0.05)(Ca0.11Na0.11K0.09), respectively. Therefore, the Doğanbey-area smectite is presumed to have formed by chemical weathering and dissolution-precipitation from feldspar and glass during diagenesis; palygorskite formed by direct precipitation from Mg-rich solutions during dolomitization, and by transformation from smectite in an alkaline lacustrine environment.
Metamorphic-hosted pyrophyllite and dickite occurrences from the hydrous Al-silicate deposits of the malatya-Pütürge region, central eastern Anatolia, Turkey
- Ömer Bozkaya, Hüseyin Yalçin, Zeynel Başibüyük, Gülcan Bozkaya
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 423-442
-
- Article
- Export citation
-
Hydrous Al-silicate deposits are found to the south of Pütürge in Malatya city, Turkey. The surrounding rocks consist of mylonitic granitic gneiss overlain by muscovite gneiss with kyanite-bearing metabasic schist lenses on top which are cut by silica veins containing prismatic tourmaline and speculante. Pyrophyllite is found within kyanite gneisses overlying the granitic gneisses. Fibrous, platy pyrophyllite is developed along the edges and cleavage planes of kyanite, whereas platy bunches of dickite occur as replacements of the relict kyanites as well as crack- and pore-fillings. Rocks forming the hydrous Al-silicate deposit contain 2M1 pyrophyllite, alunite, topaz, paragonite, dravite, dumortierite, chlorite and epidote as early hypogene minerals, and 2M1 dickite, diaspore, gibbsite, speculante, goethite and crandallite/goyazite as late hypogene minerals. On the basis of fluid inclusion and stable isotope data, it is estimated alterations to pyrophyllite and kaolinite occurred at temperatures are of 150 and 100°C, respectively, the minerals being formed by meteoric waters interacting with metamorphic rocks. Trace and REE variations are highly distinctive in terms of enrichment of most trace elements in pyrophyllite, whereas REEs are clearly abundant in dickite, indicating different conditions during formation such as early and late hypogene processes. The pyrophyllitic alteration took place in the late Cretaceous (69–71 Ma), whereas kaolinization occurred later.
Nanometer-scale chemical modification of nano-ball allophane
- Zaenal Abidin, Naoto Matsue, Teruo Henmi
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 443-449
-
- Article
- Export citation
-
Nano-ball allophane is a hydrous Al silicate with a hollow-sphere morphology that contains some defects or pores along the spherule walls. Enlargement of the pore openings by dilute alkali treatment was confirmed by cation exchange capacity determinations using various alkylammonium cations as replacement cations. An allophane sample with a low Si/Al ratio (0.67) was equilibrated with 10 mM CaCl2 (pH = 6.0) and the Ca2+ retained was extracted using aqueous 1 M NH4C1 or alkylammonium chloride salts. The Ca2+ extracted by
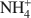 was 15.1 cmolc kg−1, but ${\rm{C}}{{\rm{H}}_3}{\rm{NH}}_3^ + $
was 15.1 cmolc kg−1, but ${\rm{C}}{{\rm{H}}_3}{\rm{NH}}_3^ + $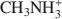 (mean diameter = 0.38 nm) only extracted 7.9 cmolc kg−1 of Ca2+. After 10 mM NaOH treatment (0.25 g:100 mL) of the allophane, the Ca2+ extracted by ${\rm{NH}}_4^ + $
(mean diameter = 0.38 nm) only extracted 7.9 cmolc kg−1 of Ca2+. After 10 mM NaOH treatment (0.25 g:100 mL) of the allophane, the Ca2+ extracted by ${\rm{NH}}_4^ + $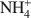 was 29.7 cmolc kg−1, 29.6 cmolc kg−1 by ${\rm{C}}{{\rm{H}}_3}{\rm{NH}}_3^ + $
was 29.7 cmolc kg−1, 29.6 cmolc kg−1 by ${\rm{C}}{{\rm{H}}_3}{\rm{NH}}_3^ + $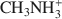 , and 29.4 cmolc kg−1 by ${{\rm{(C}}{{\rm{H}}_3}{\rm{)}}_2}{\rm{NH}}_2^ + $
, and 29.4 cmolc kg−1 by ${{\rm{(C}}{{\rm{H}}_3}{\rm{)}}_2}{\rm{NH}}_2^ + $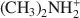 . The extraction of Ca2+ by the large ${{\rm{C}}_2}{{\rm{H}}_5}{\rm{NH}}_3^ + $
. The extraction of Ca2+ by the large ${{\rm{C}}_2}{{\rm{H}}_5}{\rm{NH}}_3^ + $ cation (mean diameter = 0.46 nm) only decreased to 26.1 cmolc kg−1, indicating that pore diameters were enlarged from ∼0.35 to 0.45 nm. The significant increase in Ca2+ retention after NaOH treatment was attributed to the dissociation of increased numbers of newly exposed silanol groups in the enlarged pores. The low Si/Al ratio of the NaOH-dissolved material (0.35) and the decreased intensity of the 348 cm−1 IR band also suggested selective dissolution of the pore region. For allophane with a high Si/Al ratio (0.99) and much accessory polymeric Si, dissolution of polymeric Si and of the pore region occurred simultaneously. Alkali treatment produced a smaller increase in pore size and Ca2+ retention for allophanes with large Si/Al ratios than for allophanes with small Si/Al ratios. It was concluded that by altering the dilute alkali treatment conditions and varying the Si/Al ratio of allophane, the extent of structural modification or pore enlargement of the hollow spheres might be controlled.
cation (mean diameter = 0.46 nm) only decreased to 26.1 cmolc kg−1, indicating that pore diameters were enlarged from ∼0.35 to 0.45 nm. The significant increase in Ca2+ retention after NaOH treatment was attributed to the dissociation of increased numbers of newly exposed silanol groups in the enlarged pores. The low Si/Al ratio of the NaOH-dissolved material (0.35) and the decreased intensity of the 348 cm−1 IR band also suggested selective dissolution of the pore region. For allophane with a high Si/Al ratio (0.99) and much accessory polymeric Si, dissolution of polymeric Si and of the pore region occurred simultaneously. Alkali treatment produced a smaller increase in pore size and Ca2+ retention for allophanes with large Si/Al ratios than for allophanes with small Si/Al ratios. It was concluded that by altering the dilute alkali treatment conditions and varying the Si/Al ratio of allophane, the extent of structural modification or pore enlargement of the hollow spheres might be controlled.
