WO2005075458A1 - Pyrimidine derivatives as orexin receptors antagonists - Google Patents
Pyrimidine derivatives as orexin receptors antagonists Download PDFInfo
- Publication number
- WO2005075458A1 WO2005075458A1 PCT/HU2005/000010 HU2005000010W WO2005075458A1 WO 2005075458 A1 WO2005075458 A1 WO 2005075458A1 HU 2005000010 W HU2005000010 W HU 2005000010W WO 2005075458 A1 WO2005075458 A1 WO 2005075458A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- alkyl
- hydroxy
- carboxylic acid
- phenyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 C*CC(C)N(C)CNC Chemical compound C*CC(C)N(C)CNC 0.000 description 5
- MCYGSOLWFMSXKQ-UHFFFAOYSA-N CC(C)(C)C(Oc1cc(C(CN(C)C(c(cnc(N(C)C)n2)c2[AlH2])=O)O)ccc1)=O Chemical compound CC(C)(C)C(Oc1cc(C(CN(C)C(c(cnc(N(C)C)n2)c2[AlH2])=O)O)ccc1)=O MCYGSOLWFMSXKQ-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/14—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing three or more hetero rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/20—Hypnotics; Sedatives
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/04—Anorexiants; Antiobesity agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D239/00—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings
- C07D239/02—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings
- C07D239/24—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings having three or more double bonds between ring members or between ring members and non-ring members
- C07D239/28—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings having three or more double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, directly attached to ring carbon atoms
- C07D239/32—One oxygen, sulfur or nitrogen atom
- C07D239/42—One nitrogen atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/04—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/06—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings linked by a carbon chain containing only aliphatic carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/12—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/02—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings
- C07D405/04—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/02—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings
- C07D405/12—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings
- C07D409/04—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings
- C07D409/12—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings
- C07D413/06—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings linked by a carbon chain containing only aliphatic carbon atoms
Definitions
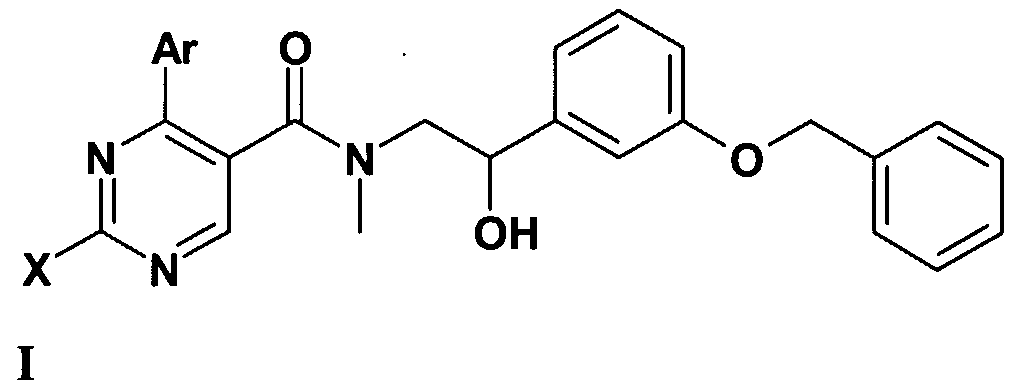
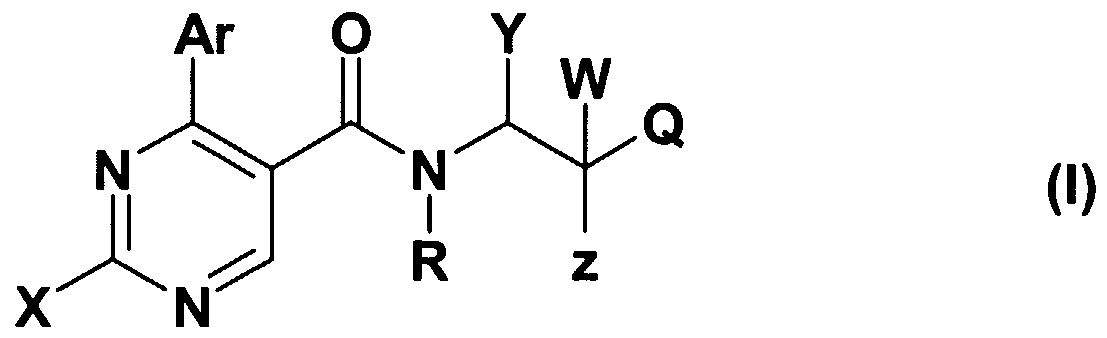
- the present invention relates to the orexin receptor antagonist compounds of the general formula (I), as well as their isomers, salts and solvates, to the pharmaceutical compositions containing them and to the therapeutic application thereof.
- hypocretin neuropeptides in other name hypocretin neuropeptides and their receptors were discovered in
- the orexinergic neuropeptides are formed in large amount in the neurons of the lateral hypothalamus, but via axonal transport processes, they also reach numerous remote areas of the nervous system. On the basis of experimental observations the orexinergic system seems to plays crutial role in the feeding, in the sleep-wake cycle and in the regulation of the autonomic nervous system processes.
- the orexin A and orexin B proteins are formed by the enzymatic cleavage of their only common precursor, the preproorexin protein molecule.
- Orexin A consists of 33 aminoacid residues with two intramolecular disulphide bridges.
- Orexin B is a linear protein consisting of 28 aminoacid residues.
- the aminoacid sequence of the orexin peptides has largely been conserved.
- the aminoacid sequences of the orexin A peptides are fully identical, whereas the aminoacid sequences of the orexin B proteins differ only in a few aminoacids.
- the orexin-producing neurons of the brain form a heterogenous cell population: one part of them exhibits leptin sensitivity, whereas the other part glucose sensitivity.
- orexinergic neurones are capable to express galanine, neuropeptide Y or dinorfme, in addition to the orexines.
- the orexin A and orexin B bind to specific receptors on the surface of the target cells: i.e. the orexin- 1 and orexin-2 receptors.
- the orexin- 1 receptors consist of 425, whereas the orexin-2 receptors of 444 aminoacid residues, and their aminoacid sequences are in 64% identical. Between the variants of the two orexin receptor types occuring in the different mammal species (man, pig, dog, mouse, rat) a considerable sequence-homology (of 91-98%) can be found.
- the aminoacid sequence of the human orexin- 1 receptor is in 94% identical with the aminoacid sequence of the rat, whereas the sequences of the human and rat orexin-2 receptors are in
- orexin A and B peptides bind with high affinity to both receptor types. Orexin affinities of the two receptor types were determined by intracellular Ca 2+ concentration measurements in recombinant systems (on CHO cells) and on hypothalamic neurons.
- the orexin A peptide was shown to be 10-50-fold more effective on the orexin- 1 receptors, demonstrating that this receptor type is selective towards orexin A.
- both neuropeptides exhibited similar, high activities, i.e. the orexin-2 receptors are not selective towards the orexin peptides.
- the orexin- 1 receptors -via the G q/ n sub-class G- proteins- may activate the phospholipase ⁇ (PLC ⁇ ) enzyme, whereas the orexin-2 receptors are supposed to bind also to the G q/ n and G; /0 or G s sub-classess of the G-proteins, thus beside the PLC ⁇ path, they may also influence the cAMP path.
- PLC ⁇ phospholipase ⁇
- the orexin- 1 and 2 receptor types are most frequent in the central nervous system (brain, spinal marrow), but they can also be found in numerous peripherical tissue types (as for instance in the hypophysis, in the adrenal glands, in the gastro-intestinal tract, in the pancreas and in the kidney).
- Orexines play important role in the regulation of the eating behizate, the sleeping-wake cycle, the neuroendocrinological processes and in the complex regulation of the energy consumption. Orexines in the central nervous system get in interaction with a number of specific neuron-nuclea, as for instance with the feeding centres of the hypothalamus, with the sleep-wake centres in the brain stam, with the symphatic and parasymphatic neuron nuclea and with the limbic system. After ventricular administration, orexines enhance in a dose-dependent manner the food-intake, the length of the time of wakefullness, the motoric activity, the speed of the metabolic processes, the heart rhythm and the blood pressure.
- Orexins and their receptors can also be found in the peripheric tissues. Orexins exert a direct effect on the hypophysis and on the hormon secretion of the adrenal glands and they influence considerably the digestion and absorbtion processes acting locally, along the gastro-intestinal tract.
- the orexin- A can effectively increase both in vitro and in vivo the insuline secretion of the pancrease and the leptin secretion of the lipides.
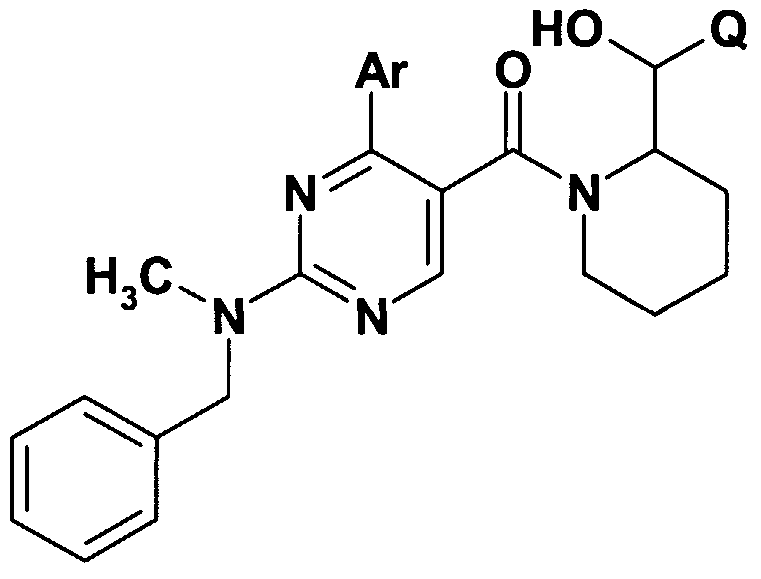
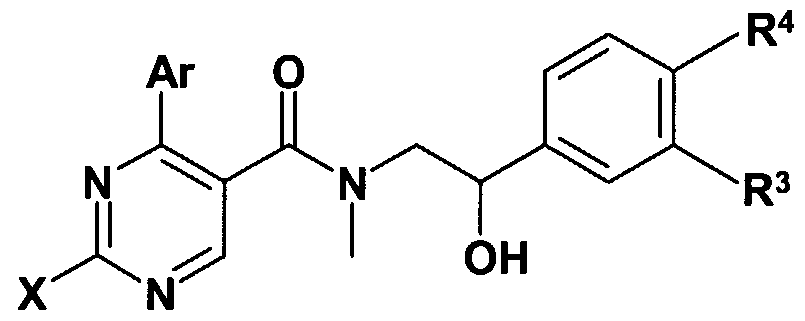
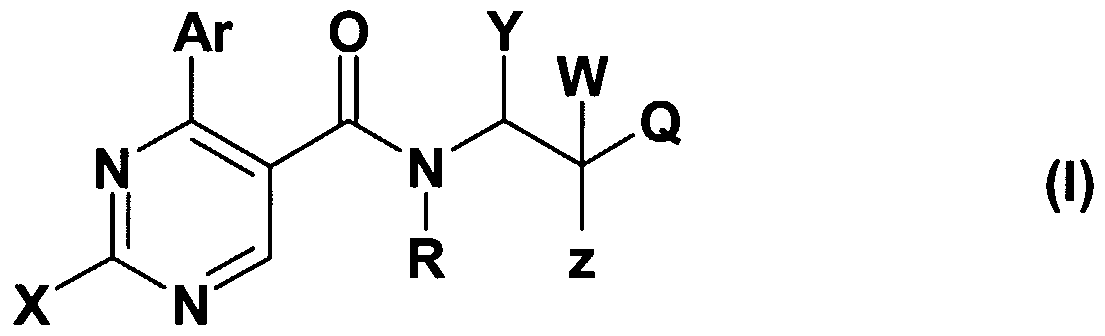
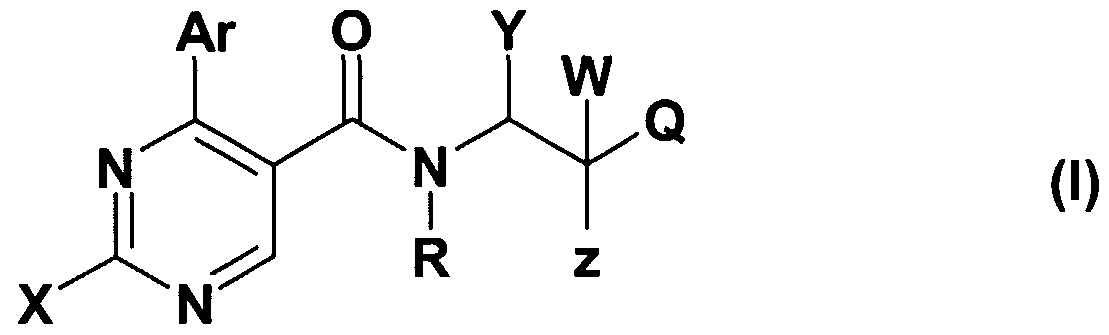
- X stands for CM-alkyl group; amino group -optionally substituted with one or two C M alkyl group, C ⁇ _ 4 -alkyl-S-group; saturated or partially saturated mono- or bicyclic moiety containing 1 or 2 or 3 heteroatoms (N, O or S) and connected to the pyrimidine ring through the nitrogen atom; or benzylamino-, phenylethylamino, N-C M alkylbenzylamino-, N-C M alkylphenylethylamino-, N-C M -hydroxyalkylbenzylamino-, N-C ⁇ .
- Z represents hydroxyl group, halogen atom, C ⁇ . 4 -alkoxy group, amino-group, C ⁇ . 4 -alkyl- amino-group, -NHCOC ⁇ . 4 -alkyl group;
- R + Y may mean -together with the included nitrogen and carbon atom- a
- Z + W together may mean an oxo group
- Q is a 5- or 6-membered heterocyclic ring containing 1-3 heteroatoms, optionally substituted with one or more identical or different C ⁇ . 4 -alkyl-, C ⁇ - -alkoxy- or hydroxyl group, or halogen atom, or a
- R is hydrogen atom, halogen atom, hydroxyl-, cyano-, C ⁇ . 4 - alkoxy- or C ⁇ . 4 -alkyl- group;
- R 3 stands for hydrogen atom, halogen atom, hydroxyl group, trihalogenomethyl group, amino-, cyano-, C ⁇ . 4 -alkylamino-, di(C ⁇ . 4 )alkylamino-, benzylamino-, benzyl-(Cj.
- R 7 is C M alkyl group, benzyl group or a phenyl group -optionally mono- or polysubstituted with halogen atom, C M alkoxy group, C M alkyl group or trihalogenomethyl group
- R 4 stands for hydrogen atom, halogen atom, hydroxyl group, cyano-group, C M alkoxy group or C alkyl group
- R 3 and R 4 together mean a -O-CH 2 -O-group; with the proviso that, if Q represents a 5- or 6-membered heterocyclic ring containing 1-3 heteroatom -optionally substituted with one or more identical or different C M alkyl-, C M alkoxy-, hydroxyl- group or halogen atom, W represents hydrogen atom, Z represents hydroxyl group, R + Y may form together with the included nitrogen and carbon atom a
- A represents NH- or NC ⁇ . 4 -alkyl or -CH 2 - group, and the meaning of Ar is as defined above, then the meaning X is different from C M alkyl- group or from an amino group -optionally substituted with one or two d. 4 alkyl group.
- W means hydrogen atom or C ⁇ . 4 -alkyl- group
- Z stands for hydroxyl group, halogen atom, C ⁇ . 4 -alkoxy group, amino group, Ci ⁇ -alkylamino group, -NHCOCM alkyl group
- Z + W form together an oxo-group
- R + Y form together with the included nitrogen and carbon atom a
- A stands for oxygen atom, -CH 2 -, -NH-, group and the value of n is 0, 1 or 2;
- Q represents a 5- or 6-membered heterocyclic ring containing 1-3 heteroatom -optionally substituted with one or more identical or different C M alkyl-, C M alkoxy-, hydroxyl- group or halogen atom, with the proviso that, if if W represents hydrogen atom, Z represents hydroxyl group, n is 1,
- A represents -NH-, or -NC M alkyl- or -CH 2 - group, then the meaning of X is different from C ⁇ . 4 alkyl group and different from an amino group -optionally substituted with one or more C M alkyl- group;
- a further group of the compounds of the general formula (I) are those where
- X and Ar have the meaning as defined in claim 1.
- W represents hydrogen atom or C ⁇ . 4 alkyl group
- Z stands for hydroxyl group, halogen atom, C M -alkoxy group, amino group, Ci. ⁇ alkylamino group, -NHCOC ⁇ . 4 -alkyl group; or Z + W form together an oxo-group; R + Y may form together with the included nitrogen and carbon atom a
- A stands for oxygen atom, -CH2-, -NH-, -NC M -alkyl- group and the value of n is 0, 1 or 2;
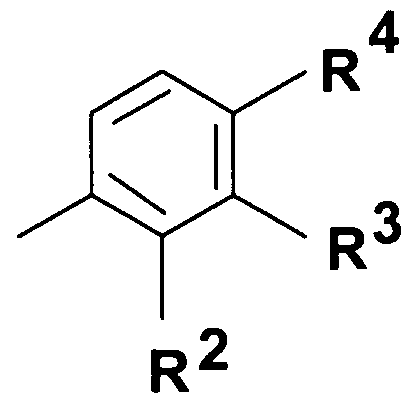
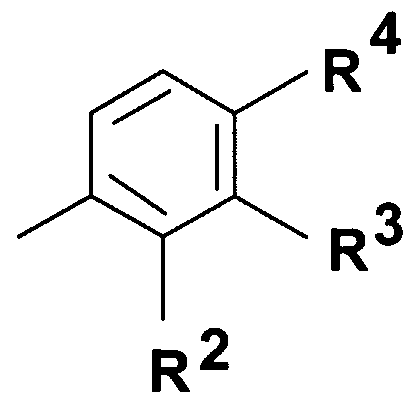
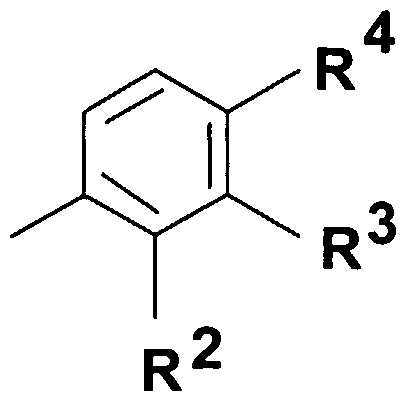
- Q represents - group where R 2 stands for hydrogen atom, halogen atom, hydroxyl group, Cj. 4 alkoxy group or C M alkyl group;
- R 3 stands for hydrogen atom, halogen atom, hydroxyl group, trihalogenomethyl group, amino-, C ⁇ . 4 -alkylamino-, di(C ⁇ )alkylamino-, benzylamino-, benzyl-(Cj. 4 )alkylamino- group, nitro group, benzyl group, phenylethyl group or C ⁇ .
- R 4 represents hydrogen atom, halogen atom, hydroxyl group, Ci ⁇ -alkoxy group or C M alkyl group; or
- Y represents hydrogen atom or C M alkyl group
- W represents hydrogen atom or C M alkyl group
- Z stands for hydroxyl group, halogen atom, C ⁇ . 4 -alkoxy group, amino group, Cj. ⁇ alkylamino group, -NHCOC ⁇ _ 4 -alkyl group; or
- R + Z form together a -(CH 2 ) m -G-group, where the value of m is 1, 2 or 3 and G represents oxygen atom, -CH 2 -, -NH- or -NC ⁇ _ 4 -alkyl- group;
- Q represents a 5- or 6-membered heterocyclic ring containing 1-3 heteroatom -optionally substituted with one or more identical or different C ⁇ . 4 alkyl-, C ⁇ . 4 alkoxy-, hydroxyl-group or halogen atom, or a group wherein R 2 represents hydrogen atom, halogen atom, hydroxyl group, Cj. 4 alkoxy group or
- R 3 stands for hydrogen atom, halogen atom, hydroxyl group, trihalogenomethyl group, amino-, C ⁇ . 4 -alkylamino-, di(C ⁇ . 4 )alkylamino-, benzylamino-, benzyl-(C ⁇ _ 4 )alkylamino- group, nitro group, benzyl group, phenylethyl group or C ⁇ . 4 -alkyl group,
- R 5 represents a Cj. 4 -alkyl group or benzyl group -optionally substituted with one or more halogen atom, trihalogenomethyl group, or
- R 4 stands for hydrogen atom, halogen atom, hydroxyl group, C alkoxy group or C M alkyl group;
- a further group of the compounds of the general formula (I) are those where
- R represents hydrogen atom or C M alkyl group
- Y represents hydrogen atom or CM alkyl group
- W represents hydrogen atom or C M alkyl group
- Z stands for hydroxyl group, halogen atom, CM-alkoxy group, amino group, CM-alkyl- amino group, -NHCOC ⁇ . 4 -alkyl group; or
- Q represents group wherein R 2 represents hydrogen atom, halogen atom, hydroxyl group, C alkoxy group or
- R 3 stands for hydrogen atom, halogen atom, hydroxyl group, trihalogenomethyl group, amino-, C ⁇ . 4 -alkylamino-, di(C ⁇ . 4 )alkylamino-, benzylamino-, benzyl-(C ⁇ - 4 )alkylamino- group, nitro group, benzyl group, phenylethyl group, d. 4 -alkyl group or
- R 5 represents a C ⁇ _ 4 -alkyl group or benzyl group -optionally substituted with one or more halogen atom, trihalogenomethyl group-;
- R 4 stands for hydrogen atom, halogen atom, hydroxyl group, C M alkoxy group or C M alkyl group;
- a further group of the compounds of the general formula (I) are those where
- R represents hydrogen atom or C ⁇ . 4 alkyl group
- Y represents hydrogen atom or C M alkyl group
- W represents hydrogen atom or C alkyl group
- Z stands for hydroxyl group, halogen atom, C ⁇ . 4 -alkoxy group, amino group, Cj. 4 - alkylamino group, -NHCOC ⁇ . 4 -alkyl group; or
- Q represents a 5- or 6-membered heterocyclic ring containing 1-3 heteroatom -optionally substituted with one or more identical or different C M alkyl-, alkoxy-, hydroxyl-group or halogen atom.
- a further group of the compounds of the general formula (I) are those where X represents benzylamino group, -N-C ⁇ . 4 -alkyl-benzyl-amino group - where the aromatic ring of the benzyl group may optionally be substituted with one or more identical or different C ⁇ . 4 -alkyl group, Cj ⁇ -alkoxy group, halogen atom or hydroxyl group;
- Ar means phenyl group -optionally substituted with one or more identical or different C M - alkyl group, Cj. 4 -alkoxy group, halogen atom trihalogenomethyl group or hydroxyl group;
- R+Y may form together with the included nitrogen atom and carbon atom a
- n 0, 1 or
- W represents hydrogen atom
- Z represents hydroxyl group
- Q represents a phenyl group or a 5- or 6-membered heterocyclic ring containing 1-3 identical or different heteroatom -optionally substituted with one or more identical or different halogen atom, Cj. 4 alkyl-, C M alkoxy-, hydroxyl-group.
- the denomination faced5- or 6-membered heterocyclic ring containing 1-3 identical or different heteroatoms may mean for example a 2-, 3- or 4-pyridyl or 2-thienyl or 3- thienyl group.
- the denomination arrivingsaturated or partially saturated mono- or bicycle containing 1-3 heteroatoms may mean for example pyrrolidinyl, morpholinyl, piperidinyl or tetrahydroisoquinolinyl moiety.
- the denomination spentHet means a heterocyclic moiety containing 1 or 2 identical or different heteroatoms (N, O or S), as for example a furanyl-, pyridyl-, thienyl- or morpholinyl group.
- the ,,4-7-membered cycloalkyl group may mean for example a cyclobutyl, cyclopentyl or cyclohexyl group.
- C M alkyl group we mean groups with straight or branched carbon chains, for instance a n-propyl, ethyl, n-butyl or tert.-butyl groups.
- C M alkoxy group we mean groups with straight or branched carbon chains, for instance a methoxy, ethoxy, isopropoxy or secondary butyloxy group.
- bicyclic ring optionally containing heteroatoms we mean for example a quinoline, isoquinoline, quinoxaline, benzotriazine or benzthiazole ring.
- halogen atom we mean fluoro, chloro, bromo or iodo atom.
- trihalogenomethyl group we mean for example trifluoromethyl-, trichloromethyl- or tribromomethyl- group.
- Orex-1 IC 50 22 nM
- Orex-2 IC 50 5800 nM
- Orex-1 IC 50 30 nM
- Orex-2 IC 50 3600 nM
- Orex-1 IC 50 12 nM
- Orex-2 IC 50 5400 nM
- Orex-1 IC 50 15 nM
- Orex-2 IC 50 320 nM
- Orex-1 IC 50 11 nM
- Orex-2 IC 50 6700 nM III
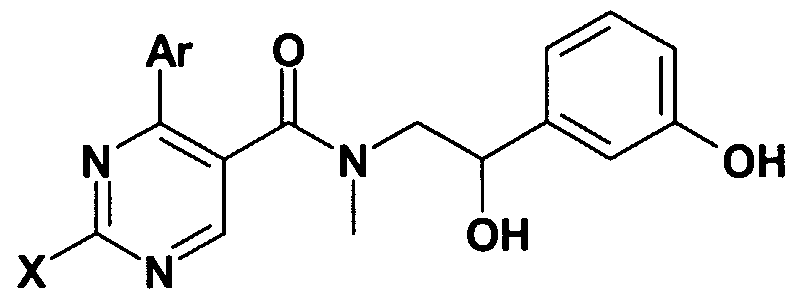
- Figure 1 shows the method of preparation of the compounds of the general formula (I).
- the acid of the general formula (II) wherein the meaning of Ar and X is as defined above- is transformed with an acid halogenide forming reagents, preferably with thionyl chloride, into the acid chloride, which is then reacted with the amine of the general formula (III) - wherein in the formula the meaning of R, Y, Z, W, Q is as defined above- in an inert solvent, (e.g. in dchloromethane or chloroform) in the presence of a base (e.g. triethylamine) or in pyridine, at room temperature or at reflux temperature.
- a base e.g. triethylamine
- the activating agent may be benzotriazol-1-yloxy- tris(pyrrolidino)phosphonium hexafluorophosphate (PyBOP) or benzotriazol-1-yloxy-tris- (dimethylamino)phosphonium hexafluorophosphate (BOP) or 4-(4,6-dimethoxy- 1,3,5- triazin-2-yl)-4-methylmo holinium chloride (DMTMM) or l-ethyl-3-(3'- dimethylaminopropyl)carbodiimide hydrochloride (EDC) with 1-hydroxybenzotriazole or 2,3,4,5,6-pentafluorophenol.
- PyBOP benzotriazol-1-yloxy- tris(pyrrolidino)phosphonium hexafluorophosphate
- BOP benzotriazol-1-yloxy-tris- (dimethylamino)phosphonium hexafluor
- the reaction is carried out in an inert sovent (e.g. in N,N- dimethylformamide, dichloromethane, tetrahydrofuran, dioxane, acetonitrile or in the mixture of thereof) at room temperature or under reflux conditions.
- an inert sovent e.g. in N,N- dimethylformamide, dichloromethane, tetrahydrofuran, dioxane, acetonitrile or in the mixture of thereof
- the compounds of the general formula (I) can also be prepared by using tetrafluorophenol bound to a solid phase polymeric support (for instance polystyrol-divinylbenzene-resin (J. M. Salvino et al. J. Comb. Chem. 2000, 2, 691) to activate the carboxylic acid.
- Functional groups of the compounds of the general formula (I) can be transformed into other functional groups (for example the substituents Q, R 2 , R 3 , R 4 , Z, W can be transformed into other Q, R 2 , R 3 , R 4 , Z, W substituents).
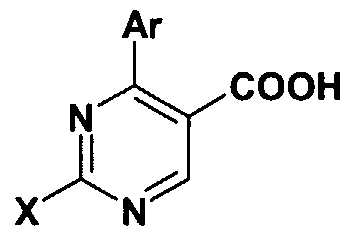
- the carboxylic acids of the general formula (II) -wherein the meaning of Ar and X is as defined above- are in part known from the literature or they can be prepared by methods known from the literature ( Figure 2.).
- the ⁇ -keto-esters of the general formula (IN) are available from the market or they can be synthesized by known methods (Gilman H. et al., J. Am. Chem. Soc. 1948, 70, 2755, Sicker D. et al., Coll. Czech. Chem. Commun. 1988, 53, 839-850, Wierenga W. et al., J. Org. Chem. 1979, 44, 310-311). Reaction of the compounds of the general formula (IN) with NN-dimethylformamide dimethyl acetale can be performed in the presence of solvent (for example in toluene at 60 °C; Herrero M.T.
- the enaminoesters of the general formula (N) with ⁇ -C- ⁇ dinucleofils for example with guanidine (VI), amidines (VI), S-methylisothiocarbamide (Nil), O-methylisocarbamide (NM) can be transformed into 2-substituted-pyrimidinecarboxylic acid esters (IX, X, XI) Breaux E. J. et al., J. Heterocyclic Chem. 1981, 18, 183, WO 00/73279).
- Ring closure can be effected in the presence of base (sodium ethylate or sodium hydrogen carbonate) in ethanol, NN-dimethylformamide or ⁇ -methylpyrrolidinone, at room temperature or at elevated temperature (80-100°C).
- base sodium ethylate or sodium hydrogen carbonate
- the 2-methylsulphanyl group of the compound of the general formula (X) can be oxidized with an oxidating agent (e.g. with 3-chloro-perbenzoic acid) into 2-methylsulphonyl group.
- the compounds of the general formula (XII) by reacting with a primary or secondary amine, in the presence of a solvent (e.g.
- esters of the general formula (IX) can be transformed into the esters of the general formula (IX) -where in the formula X represents a dialkylamino group, alkylamino group, arylamino group, arylalkylamino group, alkyl-arylalkylamino group, hetarylalkylamino group (for example hetarylmethylamino group), alkyl-hetarylalkylamino group (for example methyl- hetarylmethylamino group), amino group, 3,4-dihydro-lH-isoquinolin-2-yl group; the aryl group is a phenyl group optionally containing one or more substituents (e.g.
- halogen atom C ⁇ . 4 -alkyl group, C ⁇ . 4 -alkoxy group
- the hetaryl group is a 5- or 6-membered heteroaromatic ring containing 1-3 heteroatoms (nitrogen, oxygen or sulphur atom) (for example 2-pyridyl, 3-pyridyl, 4-pyridyl, 2-thienyl, 3-thienyl group).
- esters of the general formula (IX) where in the formula X represents a dialkylamino group, alkylamino group, arylamino group, arylalkylamino group, alkyl-arylalkylamino group, hetarylalkylamino group (for example hetarylmethylamino group), alkyl-hetarylalkylamino group (for example methyl-hetarylmethylamino group), amino group, 3,4-dihydro-lH- isoquinolin-2-yl group; the aryl group is a phenyl group optionally containing one or more substituents (e.g. halogen atom, C ⁇ .
- substituents e.g. halogen atom, C ⁇ .
- the hetaryl group is a 5- or 6-membered heteroaromatic ring containing 1-3 heteroatoms (nitrogen, oxygen or sulphur atom)
- 2-pyridyl, 3-pyridyl, 4-pyridyl, 2-thienyl, 3-thienyl group can also be prepared by reacting the ester of the formula (XI) with a primary or secondary amine at elevated temperature (up to 170 °C) in the presence of a solvent or without solvent.
- esters of the general formula (IX) -where in the formula the meanings of Ar and X are as defined above- can be hydrolyzed under acidic or basic conditions into the acids of the general formula (II) -where in the formula the meaning of Ar and X is as defined above.
- acid e.g. hydrochloric acid
- base e.g. sodium hydroxide, potassium hydroxide, sodium carbonate, potassium carbonate
- base e.g. sodium hydroxide, potassium hydroxide, sodium carbonate, potassium carbonate
- the acids of the general formula (II) can also be prepared by a novel method, as shown in Figure 3.
- the compounds of the general formula (XIII) can be synthesized by known methods, by the base-catalysed condensation of amidines with diethyl ethoxymethylenemalonate. The resulting compounds give with phosphoryl chloride the chloro-compounds of the general formula (XIV) (Dostert P. et al., Eur. J. Med. Chem. Chim. Ther. 1982, 17, 437-444).
- the chloro-compounds of the general formula (XIV) are transformed with aryl-boronic acids in dimethoxyethane, in the presence of tetrakis(triphenylphosphino)palladium catalyst into the compounds of the general formula (IX) -where in the formula Ar and X have the meanings as defined above.
- the esters of the general formula (IX) are hydrolyzed under acidic or basic conditions, preferably in the presence of base.
- the amines of the general formula (III) where in the formula the meanings of R, Y, Z, W and Q are defined above- can be purchased on the market or can be synthesized by methods known in the literature.
- the compounds of the general formula (XV), where in the formula A represents a CH, NH, N-Boc, N-(C ⁇ . 4 )-alkyl group, n 1, 2, 3 and PG means a protecting group (e.g.
- the compounds of the general formula (XVI) are those - wherein in the formula M represents a metal atom (e.g. lithium or magnesium atom), B represents a CH group, or a nitrogen atom, R 5 means hydrogen atom, hydroxyl group, halogen atom, C ⁇ . 4 -alkyl group, C ⁇ . 4 -alkoxy group.
- the substituent R 5 may take o-, m-, or p-position.
- the reaction of compounds (XV) with compounds (XVI) is performed in an inert solvent, for example in tetrahydrofuran or in ether, at low temperature (between 0 °C and -78 °C ).
- the protecting group (e.g. the t-butyloxycarbonyl-, trifluoroacetyl-, benzyloxycarbonyl group) of the compounds of the general formula (XVII) can be removed under acidic conditions (for example with trifluoroacetic acid in dichloromethane, with hydrochloric acid in dioxane or in dichloromethane), under basic conditions (for example with sodium hydroxide in aqueous methanol) or by catalytic hydrogenolysis (e.g. with palladium-on- charcoal catalyst in alcohol or in ethyl acetate).
- acidic conditions for example with trifluoroacetic acid in dichloromethane, with hydrochloric acid in dioxane or in dichloromethane
- basic conditions for example with sodium hydroxide in aqueous methanol
- catalytic hydrogenolysis e.g. with palladium-on- charcoal catalyst in alcohol or in ethyl acetate.
- the pharmaceutically acceptable salts of the compounds of the general formula (I) can be prepared by reacting the bases with the appropriate acids.
- solvates of the compounds of the general formula (I) we also mean the hydrates.
- the compounds of the general formula (I), as well as their pharmaceutically acceptable derivatives can be used for the treatment of diseases where human orexin receptors play a role, and for the treatment of which orexin receptor antagonists are needed.
- the orexin receptor antagonistic compounds of the general formula (I), and their pharmaceutically acceptable derivatives may be appropriate for the treatment of obesity and type-II diabetes (non-insuline dependent diabetes), furthermore for the treatment of sleeping disorders, narcolepsy, insomnia, jet-lag syndrome, for the treatment of sleeping disorders connected to neurological disorders, depression, anxiety, behavioral disorders, sexual disorders, neuropathic pain, pains connected to infections (like HIV), phantome pains, post-operative pains.
- the compounds of the general formula (I), and their pharmaceutically acceptable derivatives may be used for the treatment of stroke, heart- and lung diseases.
- the compounds of the general formula (I), as well as their pharmaceutically acceptable derivatives can be used for the treatment and prevention of diseases where human orexin receptor antagonists are needed for the treatment.
- the compounds according to the invention are used in the form of pharmaceutical composition.
- compositions contain the compounds of the general formula (I) or their derivatives together with pharmaceutically acceptable carriers and excipients.
- the compounds of the general formula (I), and their pharmaceutically acceptable derivatives can be administered by any of the traditional routes, e.g. by oral, parentheral, sublingual, nasal, rectal, or transdermal routes.
- the compounds of the general formula (I) and their pharmaceutically acceptable derivatives may be administered orally, in the forms of solid or liquide formulations, as for instance sirups, suspensions, emulsions, tablets or capsules.
- Liquid formulations contain the active component beside an appropriate liquid vehicle (e.g. water, ethanol, glycerine, polyethyleneglycole, oil) in the form of a solution or suspension.
- an appropriate liquid vehicle e.g. water, ethanol, glycerine, polyethyleneglycole, oil
- They may also contain colouring and odour agents.
- Tablets may contain the usual additives, e.g. magnesium stearate, starch, lactose, sucrose and cellulose.
- Hard and soft gelatine capsules can be prepared by the standard operations.
- Parenteral formulations contain the active ingredient in the form of a solution or suspension, prepared with a sterile aqueous carrier or with an appropriate oil, as for instance polyethylene glycol, polyvinylpyrrolidone, sesame oil or lecitine.
- an appropriate oil as for instance polyethylene glycol, polyvinylpyrrolidone, sesame oil or lecitine.
- Aerosols For nasal application aerosols, drops, gels or powders can be applied. Aerosols contain the active ingredient in the form of an aqueous or non-aqueous solution or suspension, in a closed container, in single or multiple doses.
- suppositories may be used which contain the usual excipients (e.g. cacao-butter or coconut-butter).
- excipients e.g. cacao-butter or coconut-butter.
- gels or dermal paches may be used.
- the doses of the compounds of the general formula (I) and their pharmaceutically acceptable derivatives used for the treatment or prevention of the above diseases depend on the nature of the disease and in general, a single dose is between 0.05 mg and 1000 mg and the daily dose is between 0.01 mg/kg and 100 mg/kg.
- 2-Dimethylamino-4-phenylpyrimidine-5-carboxylic acid (1.22 g, 5 mmol) is dissolved in tefrahydrofuran (60 ml). After 5 minutes of stirring to the solution are added: L- phenylephrine hydrochloride (1.22 g, 6 mmol) in tefrahydrofuran (50 ml), after another 5 minutes of stirring the N-methylmorpholine (1.21 g, 12 mmol) and after 2 minutes of stirring the 4-(4,6-dimethoxy-l,3,5-triazin-2-yl)-4-methylmorpholinium chloride (1.66 g, 6 mmol).
- the reaction mixture is stirred for 2 hours at room temperature, then heated at reflux temperature for 2 hours.
- the solvent is removed in vacuum.
- To the residue water (50 ml) is added, and the mixture is extracted with chloroform (75 ml), the exfract is washed with 10 % sodium carbonate solution (35 ml), with water (35 ml), dried over sodium sulphate and evaporated.
- the residue is chromatographed on silicagel using chloroform-methanol 100/1-100/5 mixture eluent. 0.36 g of the title compound is obtained, m.p.: 79-81 °C.
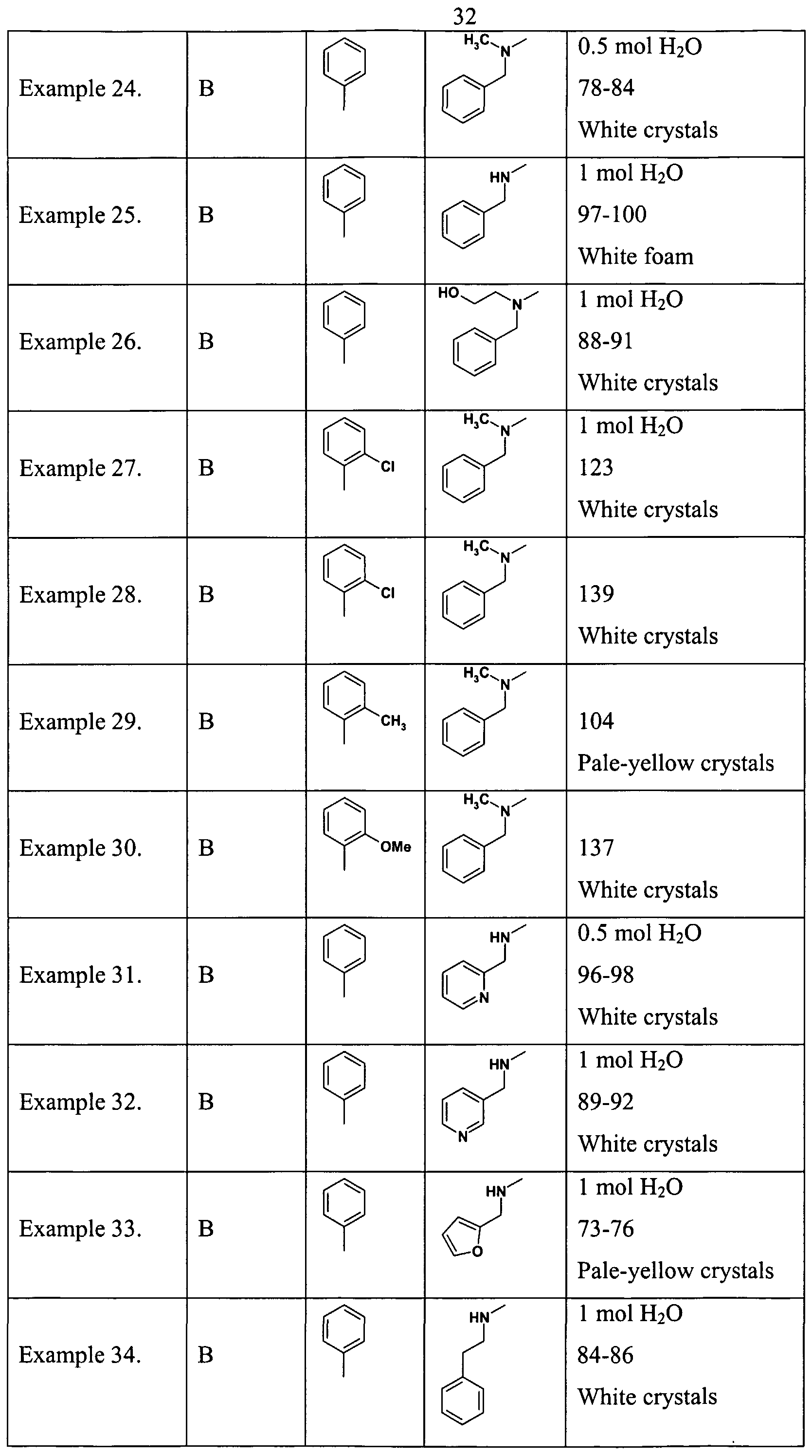
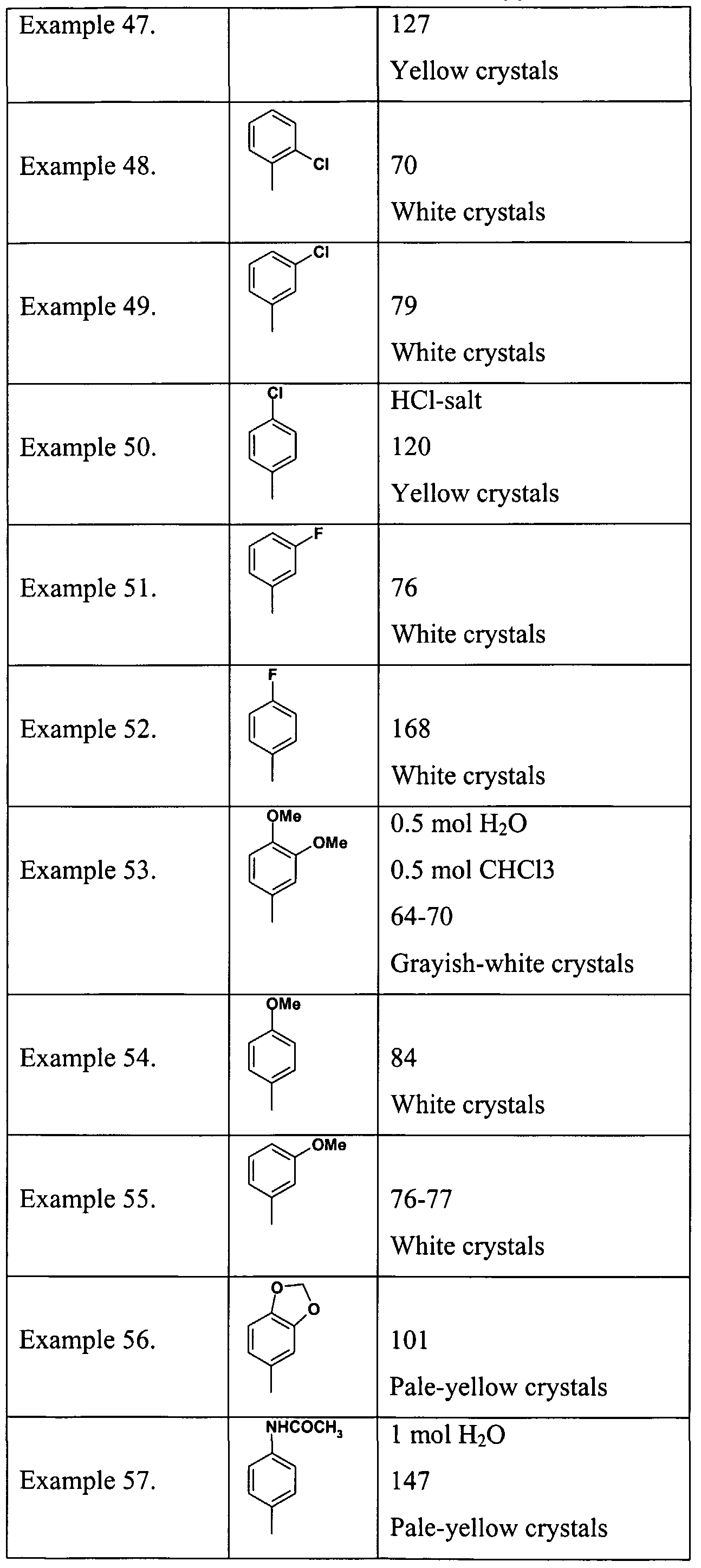
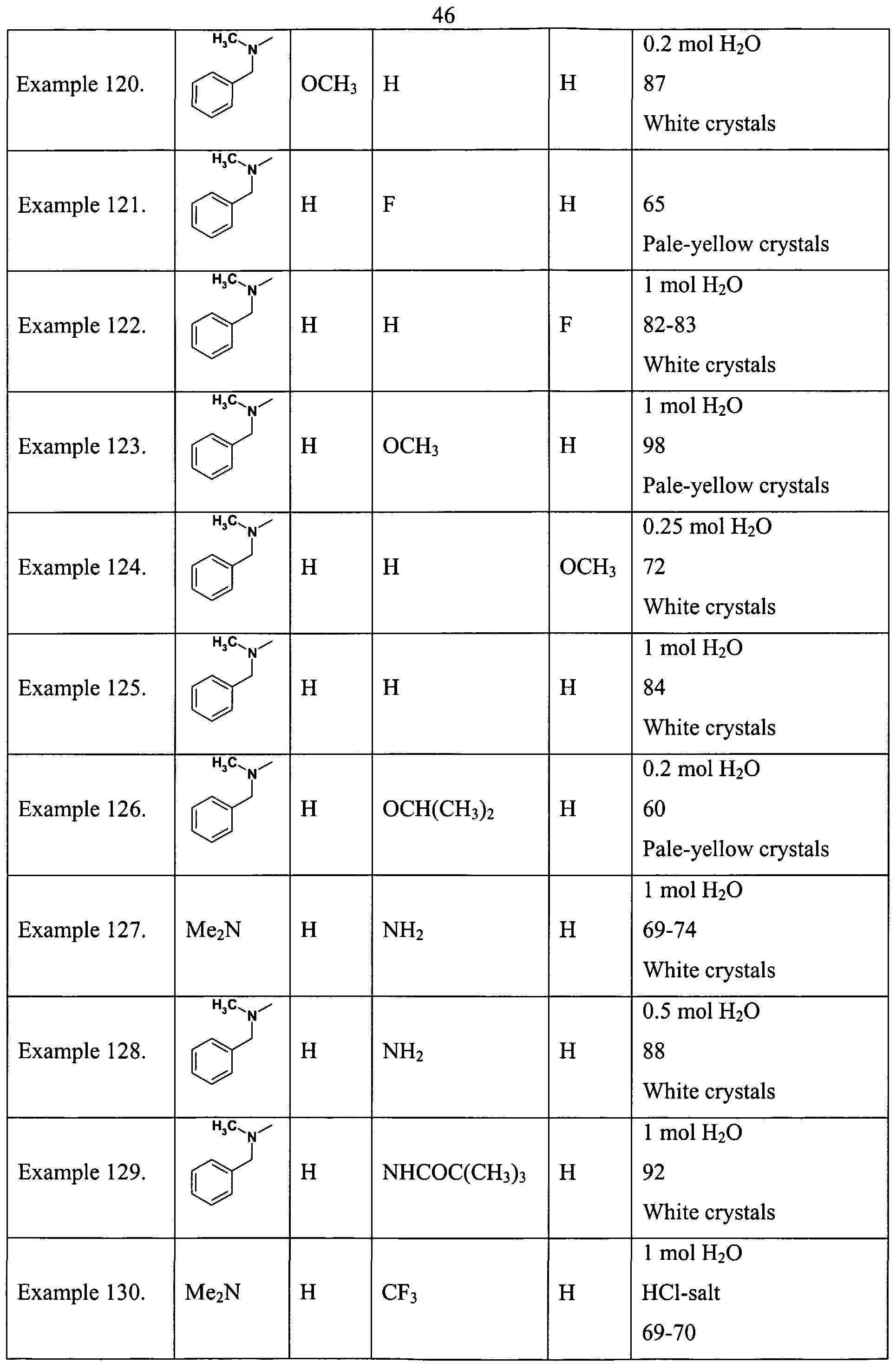
- Example 1 According to the process described in Example 1. are prepared the compounds of Tables 1. and 2.
- Example 17 4-Phenyl-2-[(thiophen-2-ylmethyl)amino]pyrimidine-5-carboxylic acid [2-hydroxy-2- (3-hydroxyphenyl)ethylJmethylamide (I) a) Ethyl 4-phenyl-2-methylsulphanylpyrimidine-5-carboxylate The mixture of ethyl benzoylacetate (9.61 g, 50 mmol) and NN-dimethylformamide dimethyl acetal (6.55 g, 55 mmol) was heated in microwave reactor for 10 minutes (30 W, — » 130 °C).
- Tetrakis(triphenylphosphino)palladium catalyst (0.25 g, 0.22 mmol) is dissolved under nitrogen atmosphere in dimethoxyethane (20 ml) and to this solution the solution of ethyl- 2-(benzylmethylamino)-4-chloropyrimidine-5-carboxylate (2.75 g, 9.0 mmol) in dimethoxyethane (20 ml) is added.
- 2-chlorophenylboronic acid (1.55 g, 9.9 mmol
- sodium carbonate (2.43 g, 23 mmol)
- dimethoxyethane (20 ml) and water 40 ml
- the material is prepared according to the method described in Example 17. Starting from 2-(Benzylmethylamino)-4-(2-chlorophenyl)pyrimidine-5-carboxylic acid and L- phenylephrine hydrochloride.M.p.:139 °C. Table 3.
- Example 41 White crystals 145-146
- Example 44 52-57 Yellow crystals 1.5 mol H 2 O
- Example 59 2-Dimethylamino-4-(3,4-dichIorophenyl)pyrimidine-5-carboxylic acid [2-(3- benzyloxyphenyl)-2-hydroxyethyl]methylamide
- Example 74 4-Phenyl-2-[(pyridin-2-ylmethyl)amino]pyrimidine-5-carboxylic acid (2-hydroxy-l- methyl-2-phenylethyl)methylamide
- Method B Starting from 4-phenyl-2-[(pyridin-2-ylmethyl)amino]pyrimidine-5-carboxylic acid (0.48 g, 1.5 mmol) and (lR,2S)-2-methylamino-l -phenyl- 1-propanol [(-)-ephedrine] (0.25 g, 1.5 mmol) according to the method described in Example 17. the title amide 0.42 g (58 %) is obtained as white crystalline matrerial; m.p.: 82 °C; [M+HJ + 468.
- the mixture is srirred at room temperature for 20 hours.
- the resin is filtered off, washed with NN-dimethylformamide, with tefrahydrofuran and finally with dichloromethane (3 x 10-10 ml).
- the reagent resin is then swelled in 2 ml of NN-dimethylformamide for 5 minutes under stirring, then 2-dimethylamino-4-phenylpyrimidine-5-carboxylic acid [2-(3-aminophenyl)- 2-hydroxyethyl]methylamide (39 mg, 0.10 mmol) is added to it.
- the mixture is srirred at room temperature for 20 hours.
- Ar phenyl
- X Me(C 6 H 5 CH 2 )N
- R 2 H
- R 3 OH
- hr- 125 I-orexin-A radioligand competition (displacement) experiments a fixed concentration of hr- 125 I-orexin-A is incubated with increasing concentrations of unlabeled test compound in the presence of highly purified plasmamembranes bearing either the human recombinant orexin- 1 (hr-OX-1) or the human recombinant orexin-2 (hr- OX-2) receptors.
- hr-OX-1 human recombinant orexin- 1
- hr- OX-2 human recombinant orexin-2
- Culturing the Chinese hamster ovarian cells expressing human recombinant orexin -1 or orexin-2 receptor proteins (CHO-hr-OX- 1 or CHO-hr-OX-2 cells) was carried out in cell culture medium (MEM medium, supplemented with 40 mg/1 prolin, 20 mg/1 gentamycin, 300 mg/1 geneticin, 10% dialysed fetal calf serum).
- Adherent cells were plated into Greiner flasks (175 cm 2 ). 4-6 days later culture medium was removed and cells were scarped in calcium- and magnesium-free phosphate buffered saline (PBS, 20 ml/flask). The cell suspension was cenfrifuged at 1,000 g for 5 minutes (4°C). The resulting pellet was resuspended and homogenized with a teflon pestle (4°C), then layered onto a discontinous sucrose gradient and cenfrifuged at 105,000 g. Plasmamembrane fraction accumulated in the interface between 14 and 34% sucrose layers was separated and pelleted by a further centrifugation step at 105,000 g for 60 min (4°C). The final pellet was resuspended in binding assay buffer and stored at -80°C up to the day of radioligand binding experiment. In vitro 125 I-orexin-A binding
- Test compounds were dissolved at a concentration of 1 mM in dimethylsulfoxide (DMSO).
- Serial dilution series were prepared from stock solutions (100% DMSO) with binding assay buffer in such a way that each samples contained a final concentration of 1% of DMSO in the receptor binding reaction mixture.
- samples were filtered through Whatman GF/C glass fibre filters using a SKATRON cell harvester, and the filters were washed with 5 ml of ice-cold buffer. The radioactivity remained on the filter was counted in a gamma counter (Wallac Automatic Gamma Counter 1470 Wizard).
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Engineering & Computer Science (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Diabetes (AREA)
- Hematology (AREA)
- Obesity (AREA)
- Biomedical Technology (AREA)
- Neurology (AREA)
- Neurosurgery (AREA)
- Emergency Medicine (AREA)
- Endocrinology (AREA)
- Child & Adolescent Psychology (AREA)
- Anesthesiology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Plural Heterocyclic Compounds (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
Abstract
Description
Claims
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP05708501A EP1718632B1 (en) | 2004-02-10 | 2005-02-08 | Pyrimidine derivatives as orexin receptor antagonists |
| JP2006552698A JP4825686B2 (en) | 2004-02-10 | 2005-02-08 | Pyrimidine derivatives as orexin receptor antagonists |
| DE602005014086T DE602005014086D1 (en) | 2004-02-10 | 2005-02-08 | PYRIMIDINE DERIVATIVES AS OREXINE RECEPTOR ANTAGONISTS |
| AT05708501T ATE429427T1 (en) | 2004-02-10 | 2005-02-08 | PYRIMIDINE DERIVATIVES AS OREXIN RECEPTOR ANTAGONISTS |
| US11/463,825 US7812031B2 (en) | 2004-02-10 | 2006-08-10 | Pyrimidine derivatives as orexin receptor antagonists |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| HU0400405A HUP0400405A3 (en) | 2004-02-10 | 2004-02-10 | Pyrimidine derivatives, process for producing them, their use, pharmaceutical compositions containing them and their intermediates |
| HUP0400405 | 2004-02-10 |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US11/463,825 Continuation US7812031B2 (en) | 2004-02-10 | 2006-08-10 | Pyrimidine derivatives as orexin receptor antagonists |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2005075458A1 true WO2005075458A1 (en) | 2005-08-18 |
| WO2005075458A8 WO2005075458A8 (en) | 2006-01-19 |
Family
ID=89981980
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/HU2005/000010 Ceased WO2005075458A1 (en) | 2004-02-10 | 2005-02-08 | Pyrimidine derivatives as orexin receptors antagonists |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US7812031B2 (en) |
| EP (1) | EP1718632B1 (en) |
| JP (1) | JP4825686B2 (en) |
| AT (1) | ATE429427T1 (en) |
| DE (1) | DE602005014086D1 (en) |
| HU (1) | HUP0400405A3 (en) |
| WO (1) | WO2005075458A1 (en) |
Cited By (37)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006050476A3 (en) * | 2004-11-03 | 2006-10-19 | Vertex Pharma | Pyrimidine derivatives as ion channel modulators and methods of use |
| WO2008008517A2 (en) | 2006-07-14 | 2008-01-17 | Merck & Co., Inc. | Bridged diazepan orexin receptor antagonists |
| WO2008017381A1 (en) | 2006-08-08 | 2008-02-14 | Sanofi-Aventis | Arylaminoaryl-alkyl-substituted imidazolidine-2,4-diones, processes for preparing them, medicaments comprising these compounds, and their use |
| WO2008009963A3 (en) * | 2006-07-21 | 2008-05-08 | Black James Foundation | Pyrimidine derivatives as modulators of parathyroid hormone receptors |
| WO2008069997A1 (en) | 2006-12-01 | 2008-06-12 | Merck & Co., Inc. | Substituted diazepan compounds as orexin receptor antagonists |
| WO2008147518A1 (en) | 2007-05-23 | 2008-12-04 | Merck & Co., Inc. | Pyridyl piperidine orexin receptor antagonists |
| WO2008150364A1 (en) | 2007-05-23 | 2008-12-11 | Merck & Co., Inc. | Cyclopropyl pyrrolidine orexin receptor antagonists |
| WO2009003997A1 (en) * | 2007-07-03 | 2009-01-08 | Glaxo Group Limited | Imidazo [1, 2-c] pyrimidin-2-ylmethylpiperidines as orexin receptor antagonists |
| WO2009021740A2 (en) | 2007-08-15 | 2009-02-19 | Sanofis-Aventis | Substituted tetrahydronaphthalenes, process for the preparation thereof and the use thereof as medicaments |
| WO2009043507A3 (en) * | 2007-09-11 | 2009-05-22 | Mondobiotech Lab Ag | Use of a peptide as a therapeutic agent |
| WO2009150614A1 (en) * | 2008-06-11 | 2009-12-17 | Actelion Pharmaceuticals Ltd | Tetrazole compounds as orexin receptor antagonists |
| WO2010003624A2 (en) | 2008-07-09 | 2010-01-14 | Sanofi-Aventis | Heterocyclic compounds, processes for their preparation, medicaments comprising these compounds, and the use thereof |
| WO2010044054A1 (en) * | 2008-10-14 | 2010-04-22 | Actelion Pharmaceuticals Ltd | Phenethylamide derivatives and their heterocyclic analogues |
| WO2010068601A1 (en) | 2008-12-08 | 2010-06-17 | Sanofi-Aventis | A crystalline heteroaromatic fluoroglycoside hydrate, processes for making, methods of use and pharmaceutical compositions thereof |
| WO2011023754A1 (en) | 2009-08-26 | 2011-03-03 | Sanofi-Aventis | Novel crystalline heteroaromatic fluoroglycoside hydrates, pharmaceuticals comprising these compounds and their use |
| EP2214676A4 (en) * | 2007-10-29 | 2011-08-03 | Merck Sharp & Dohme | OXININE RECEPTOR ANTAGONISTS SUBSTITUTED DIAZEPAN TYPE |
| WO2012120050A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Novel substituted phenyl-oxathiazine derivatives, method for producing them, drugs containing said compounds and the use thereof |
| WO2012120055A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Di- and tri-substituted oxathiazine derivates, method for the production thereof, use thereof as medicine and drug containing said derivatives and use thereof |
| WO2012120054A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Di- and tri-substituted oxathiazine derivates, method for the production thereof, use thereof as medicine and drug containing said derivatives and use thereof |
| WO2012120057A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Novel substituted phenyl-oxathiazine derivatives, method for producing them, drugs containing said compounds and the use thereof |
| WO2012120053A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Branched oxathiazine derivatives, method for the production thereof, use thereof as medicine and drug containing said derivatives and use thereof |
| WO2012120056A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Tetrasubstituted oxathiazine derivatives, method for producing them, their use as medicine and drug containing said derivatives and the use thereof |
| WO2012120058A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Oxathiazine derivatives which are substituted with benzyl or heteromethylene groups, method for producing them, their use as medicine and drug containing said derivatives and the use thereof |
| WO2012120051A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Benzyl-oxathiazine derivates substituted with adamantane or noradamantane, medicaments containing said compounds and use thereof |
| WO2012120052A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Oxathiazine derivatives substituted with carbocycles or heterocycles, method for producing same, drugs containing said compounds, and use thereof |
| WO2014047707A1 (en) | 2012-09-28 | 2014-04-03 | Cristália Produtos Químicos Farmacêuticos Ltda | Heteroaromatic compounds, method for preparing the compounds, pharmaceutical compositions, uses and method for treating acute and chronic pain |
| US9156819B2 (en) | 2011-10-19 | 2015-10-13 | Merck Sharp & Dohme Corp. | 2-pyridyloxy-4-nitrile orexin receptor antagonists |
| US9440982B2 (en) | 2012-02-07 | 2016-09-13 | Eolas Therapeutics, Inc. | Substituted prolines/piperidines as orexin receptor antagonists |
| US9499517B2 (en) | 2012-02-07 | 2016-11-22 | Eolas Therapeutics, Inc. | Substituted prolines / piperidines as orexin receptor antagonists |
| WO2017194548A1 (en) | 2016-05-10 | 2017-11-16 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and pharmaceutical compositions for the treatment of autoimmune inflammatory diseases |
| US10221170B2 (en) | 2014-08-13 | 2019-03-05 | Eolas Therapeutics, Inc. | Difluoropyrrolidines as orexin receptor modulators |
| WO2020188299A1 (en) * | 2019-03-20 | 2020-09-24 | Atrogi Ab | Heteroaryl(heterocyclyl)methanol compounds useful in the treatment of hyperglycaemia |
| US10894789B2 (en) | 2016-02-12 | 2021-01-19 | Astrazeneca Ab | Halo-substituted piperidines as orexin receptor modulators |
| US11357757B2 (en) | 2017-09-13 | 2022-06-14 | Atrogi Ab | Heteroaryl substituted beta-hydroxyethylamines for use in treating hyperglycaemia |
| US11427539B2 (en) | 2017-09-13 | 2022-08-30 | Atrogi Ab | Beta-hydroxy heterocyclic amines and their use in the treatment of hyperglycaemia |
| US11648216B2 (en) | 2017-09-13 | 2023-05-16 | Atrogi Ab | Fluorophenyl beta-hydroxyethylamines and their use in the treatment of hyperglycaemia |
| US11793774B2 (en) | 2017-09-13 | 2023-10-24 | Atrogi Ab | Chiral beta-hydroxyethylamines and their use in the treatment of hyperglycemia |
Families Citing this family (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2896799B1 (en) * | 2006-02-02 | 2008-03-28 | Sanofi Aventis Sa | SULFONAMIDE DERIVATIVES, THEIR PREPARATION AND THEIR THERAPEUTIC APPLICATION |
| US20080287452A1 (en) * | 2007-05-16 | 2008-11-20 | Wyeth | Heteroaryl/aryl pyrimidine analogs and their use as agonists of the wnt-beta-catenin cellular messaging system |
| US8263586B2 (en) | 2007-05-18 | 2012-09-11 | Merck Sharp & Dohme Corp. | OXO bridged diazepan orexin receptor antagonists |
| AR072899A1 (en) | 2008-08-07 | 2010-09-29 | Merck Sharp & Dohme | DERIVATIVES OF TERPIRIDINE-CARBOXAMIDE ANTAGONISTS OF OREXIN RECEPTORS, PHARMACEUTICAL COMPOSITIONS CONTAINING THEM AND USE OF THE SAME IN THE TREATMENT OF INSOMNIUM AND OBESITY. |
| US8466281B2 (en) * | 2008-10-21 | 2013-06-18 | Merck Sharp & Dohme Corp. | 2,5-disubstituted morpholine orexin receptor antagonists |
| AU2009308980B2 (en) * | 2008-10-30 | 2013-02-28 | Merck Sharp & Dohme Corp. | Isonicotinamide orexin receptor antagonists |
| AU2009308981A1 (en) * | 2008-10-30 | 2010-05-06 | Merck Sharp & Dohme Corp. | 2,5-disubstituted phenyl carboxamide orexin receptor antagonists |
| SI2491038T1 (en) | 2009-10-23 | 2016-08-31 | Janssen Pharmaceutica N.V. | Disubstituted octahy - dropyrrolo (3,4-c)pyrroles as orexin receptor modulators |
| CN101955463B (en) * | 2010-08-04 | 2012-01-04 | 重庆博腾制药科技股份有限公司 | Method for preparing rosuvastatin calcium intermediate |
| EP2708537A4 (en) * | 2011-05-10 | 2014-10-01 | Taisho Pharmaceutical Co Ltd | HETEROAROMATIC CYCLE DERIVATIVE |
| WO2015095111A1 (en) | 2013-12-18 | 2015-06-25 | Merck Sharp & Dohme Corp. | Diazepane orexin receptor antagonists |
| JP2017100950A (en) * | 2014-04-04 | 2017-06-08 | 大正製薬株式会社 | Oxo heterocyclic derivative |
| PH12018501903B1 (en) | 2016-03-10 | 2023-03-17 | Janssen Pharmaceutica Nv | Methods of treating depression using orexin-2 receptor antagonists |
| CN112920111B (en) * | 2019-12-06 | 2022-05-17 | 中国科学院大连化学物理研究所 | Polysubstituted pyridine derivative and synthetic method thereof |
Citations (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE244321C (en) * | ||||
| US2514380A (en) * | 1946-12-26 | 1950-07-11 | Hoffmann La Roche | Diamines and method of production |
| US2835669A (en) * | 1958-05-20 | Process for the production of substi- | ||
| GB1159263A (en) * | 1966-08-31 | 1969-07-23 | Smith Kline French Lab | Guanidine Derivatives and Process for Preparing the same |
| US3467662A (en) * | 1967-10-05 | 1969-09-16 | Sandoz Ag | 1-para-chlorophenyl-3-hydrazino - 1,5,6,7,8,8a-hexahydroimidazo(1,5-a)pyridine and process for its preparation |
| DE1955318A1 (en) * | 1968-11-08 | 1970-06-04 | Wander Ag Dr A | New pyrimidine derivatives and processes for their preparation |
| US3821244A (en) * | 1971-08-16 | 1974-06-28 | Mead Johnson & Co | 2-amino-4-phenyl-2-imidazolines |
| FR2241305A1 (en) * | 1973-08-20 | 1975-03-21 | Thomae Gmbh Dr K | |
| EP0638557A1 (en) * | 1993-08-13 | 1995-02-15 | Nihon Nohyaku Co., Ltd. | N-phenylpyri(mi)dine-N'-phenyl-urea derivatives, their preparation and their use as ACAT inhibitors |
| US6274588B1 (en) * | 1999-05-31 | 2001-08-14 | Hoffmann-La Roche Inc. | 4-phenyl-pyrimidine derivatives |
| WO2001096302A1 (en) * | 2000-06-16 | 2001-12-20 | Smithkline Beecham P.L.C. | Piperidines for use as orexin receptor antagonists |
| WO2002020495A2 (en) * | 2000-09-06 | 2002-03-14 | Chiron Corporation | Inhibitors of glycogen synthase kinase 3 |
| WO2003051872A1 (en) * | 2001-12-19 | 2003-06-26 | Smithkline Beecham P.L.C. | Ethylene diamine derivatives and their use as orexin-receptor antagonists |
| WO2004041791A1 (en) * | 2002-11-06 | 2004-05-21 | Glaxo Group Limited | N-aryl acetyl cyclic amine derivatives as orexin antagonists |
| WO2004041816A1 (en) * | 2002-11-06 | 2004-05-21 | Glaxo Group Limited | Azacyclic compounds as orexin receptor antagonist |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE2341925A1 (en) * | 1973-08-20 | 1975-03-06 | Thomae Gmbh Dr K | 2,4, (opt.5-), 6-substd. pyriminidines as antithrombic agents - e.g. 6-methyl-5-nitro-2-piperazino-4-thiomorpolino-pyrimidine |
| US4698340A (en) * | 1984-07-19 | 1987-10-06 | Fujisawa Pharmaceutical Co., Ltd. | Pyrimidine derivatives, processes for preparation thereof and composition containing the same |
| JPH07101940A (en) * | 1993-08-13 | 1995-04-18 | Nippon Nohyaku Co Ltd | Pyrimidine and pyridine derivatives, their production and uses |
| JP4623354B2 (en) * | 2000-09-14 | 2011-02-02 | 味の素株式会社 | Novel pyrimidine derivatives and novel pyridine derivatives |
| US7767680B2 (en) * | 2004-11-03 | 2010-08-03 | Vertex Pharmaceuticals Incorporated | Ion channel modulators and methods of use |
-
2004
- 2004-02-10 HU HU0400405A patent/HUP0400405A3/en unknown
-
2005
- 2005-02-08 WO PCT/HU2005/000010 patent/WO2005075458A1/en not_active Ceased
- 2005-02-08 AT AT05708501T patent/ATE429427T1/en not_active IP Right Cessation
- 2005-02-08 DE DE602005014086T patent/DE602005014086D1/en not_active Expired - Lifetime
- 2005-02-08 EP EP05708501A patent/EP1718632B1/en not_active Expired - Lifetime
- 2005-02-08 JP JP2006552698A patent/JP4825686B2/en not_active Expired - Fee Related
-
2006
- 2006-08-10 US US11/463,825 patent/US7812031B2/en not_active Expired - Fee Related
Patent Citations (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE244321C (en) * | ||||
| US2835669A (en) * | 1958-05-20 | Process for the production of substi- | ||
| US2514380A (en) * | 1946-12-26 | 1950-07-11 | Hoffmann La Roche | Diamines and method of production |
| GB1159263A (en) * | 1966-08-31 | 1969-07-23 | Smith Kline French Lab | Guanidine Derivatives and Process for Preparing the same |
| US3467662A (en) * | 1967-10-05 | 1969-09-16 | Sandoz Ag | 1-para-chlorophenyl-3-hydrazino - 1,5,6,7,8,8a-hexahydroimidazo(1,5-a)pyridine and process for its preparation |
| DE1955318A1 (en) * | 1968-11-08 | 1970-06-04 | Wander Ag Dr A | New pyrimidine derivatives and processes for their preparation |
| US3821244A (en) * | 1971-08-16 | 1974-06-28 | Mead Johnson & Co | 2-amino-4-phenyl-2-imidazolines |
| FR2241305A1 (en) * | 1973-08-20 | 1975-03-21 | Thomae Gmbh Dr K | |
| EP0638557A1 (en) * | 1993-08-13 | 1995-02-15 | Nihon Nohyaku Co., Ltd. | N-phenylpyri(mi)dine-N'-phenyl-urea derivatives, their preparation and their use as ACAT inhibitors |
| US6274588B1 (en) * | 1999-05-31 | 2001-08-14 | Hoffmann-La Roche Inc. | 4-phenyl-pyrimidine derivatives |
| WO2001096302A1 (en) * | 2000-06-16 | 2001-12-20 | Smithkline Beecham P.L.C. | Piperidines for use as orexin receptor antagonists |
| WO2002020495A2 (en) * | 2000-09-06 | 2002-03-14 | Chiron Corporation | Inhibitors of glycogen synthase kinase 3 |
| WO2003051872A1 (en) * | 2001-12-19 | 2003-06-26 | Smithkline Beecham P.L.C. | Ethylene diamine derivatives and their use as orexin-receptor antagonists |
| WO2004041791A1 (en) * | 2002-11-06 | 2004-05-21 | Glaxo Group Limited | N-aryl acetyl cyclic amine derivatives as orexin antagonists |
| WO2004041816A1 (en) * | 2002-11-06 | 2004-05-21 | Glaxo Group Limited | Azacyclic compounds as orexin receptor antagonist |
Non-Patent Citations (9)
| Title |
|---|
| BAKER B R, SHAPIRO H S: "Analogs of Tetrahydrofolic Acid. IX. Synthesis of N-[1-(2-Amino-4-hydroxy-6-phenyl-5-pyrimidyl)-3-propyl]p-aminobenzoyl-L-glutamic Acid, a "Nonclassical" Inhibitor of Some Folic Cofactor Area Enzymes", JOURNAL OF MEDICINAL CHEMISTRY, vol. 6, 1963, pages 664 - 669, XP002331361 * |
| BREAUX E J ET AL: "An improved general synthesis of 4-aryl-5-pyrimidinecarboxylates", JOURNAL OF HETEROCYCLIC CHEMISTRY, HETEROCORPORATION. PROVO, US, vol. 18, January 1981 (1981-01-01), pages 183 - 184, XP002117468, ISSN: 0022-152X * |
| DESHMUKH M B, SHELAR M A: "Efficient method for deamination of amino pyridines", NATIONAL ACADEMY SCIENCE LETTERS, vol. 21, no. 7, 1998, pages 247 - 249, XP009046773 * |
| DORIGO P, BENETOLLO F ET AL: "Synthesis and Cardiotonic Activity of Novel Pyrimidine Derivatives: Crystallographic and Quantum Chemical Studies", JOURNAL OF MEDICINAL CHEMISTRY, vol. 39, 1996, pages 3671 - 3683, XP002331359 * |
| MATOSIUK D ET AL: "Synthesis and CNS activity of 1-alkyl-5-arylimidazolidine -2-thiones", PHARMAZIE, vol. 52, no. 11, 1997, pages 71 - 72, XP001206542 * |
| PALANKI M S S ET AL: "Structure-Activity Relationship Studies of Ethyl 2-[(3-Methyl-2,5-dioxo(3-pyrrolinyl))amino]-4-(trifluoromethyl)pyrimidine-5-carboxylate: An Inhibitor of AP-1 and NF-kB Mediated Gene Expression", BIOORGANIC & MEDICINAL CHEMISTRY LETTERS, vol. 12, 2002, pages 2573 - 2577, XP002331360 * |
| REZENDE M C ET AL: "The preparation of potentially psychoactive beta-alkoxyphenethylamines", SYNTHETIC COMMUNICATIONS, vol. 25, no. 8, 1995, pages 1239 - 1247, XP009048646 * |
| SHIH N-Y ET AL: "Trans-4-Methyl-3-Imidazoyl Pyrrolidine as a potent, highly selective Histamine H3 Receptor Agonist in vivo", BIOORGANIC & MEDICINAL CHEMISTRY LETTERS, vol. 8, 1998, pages 243 - 248, XP002331363 * |
| WEINHARDT K ET AL: "Synthesis and Central Nervous System Properties of 2-[(Alkoxycarbonyl)amino]-4(5)-phenyl-2-imidazolines", JOURNAL OF MEDICINAL CHEMISTRY, vol. 27, 1984, pages 616 - 627, XP002331362 * |
Cited By (55)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8546414B2 (en) | 2004-11-03 | 2013-10-01 | Vertex Pharmaceuticals Incorporated | Ion channel modulators and methods of use |
| US7767680B2 (en) | 2004-11-03 | 2010-08-03 | Vertex Pharmaceuticals Incorporated | Ion channel modulators and methods of use |
| US8236815B2 (en) | 2004-11-03 | 2012-08-07 | Vertex Pharmaceuticals Incorporated | Ion channel modulators and methods of uses |
| WO2006050476A3 (en) * | 2004-11-03 | 2006-10-19 | Vertex Pharma | Pyrimidine derivatives as ion channel modulators and methods of use |
| WO2008008517A2 (en) | 2006-07-14 | 2008-01-17 | Merck & Co., Inc. | Bridged diazepan orexin receptor antagonists |
| WO2008009963A3 (en) * | 2006-07-21 | 2008-05-08 | Black James Foundation | Pyrimidine derivatives as modulators of parathyroid hormone receptors |
| WO2008017381A1 (en) | 2006-08-08 | 2008-02-14 | Sanofi-Aventis | Arylaminoaryl-alkyl-substituted imidazolidine-2,4-diones, processes for preparing them, medicaments comprising these compounds, and their use |
| WO2008069997A1 (en) | 2006-12-01 | 2008-06-12 | Merck & Co., Inc. | Substituted diazepan compounds as orexin receptor antagonists |
| US7951797B2 (en) | 2006-12-01 | 2011-05-31 | Merck Sharp & Dohme Corp. | Substituted diazepan orexin receptor antagonists |
| EP2392572A1 (en) | 2006-12-01 | 2011-12-07 | Merck Sharp & Dohme Corp. | Substituted diazepan compounds as orexin receptor antagonists |
| US8242121B2 (en) | 2007-05-23 | 2012-08-14 | Merck Sharp & Dohme Corp. | Pyridyl piperidine orexin receptor antagonists |
| US8569311B2 (en) | 2007-05-23 | 2013-10-29 | Merch Sharp & Dohme Corp. | Pyridyl piperidine orexin receptor antagonists |
| WO2008150364A1 (en) | 2007-05-23 | 2008-12-11 | Merck & Co., Inc. | Cyclopropyl pyrrolidine orexin receptor antagonists |
| WO2008147518A1 (en) | 2007-05-23 | 2008-12-04 | Merck & Co., Inc. | Pyridyl piperidine orexin receptor antagonists |
| WO2009003997A1 (en) * | 2007-07-03 | 2009-01-08 | Glaxo Group Limited | Imidazo [1, 2-c] pyrimidin-2-ylmethylpiperidines as orexin receptor antagonists |
| JP2010531849A (en) * | 2007-07-03 | 2010-09-30 | グラクソ グループ リミテッド | Imidazo [1,2-c] pyrimidin-2-ylmethylpiperidine derivatives as orexin receptor antagonists |
| WO2009021740A2 (en) | 2007-08-15 | 2009-02-19 | Sanofis-Aventis | Substituted tetrahydronaphthalenes, process for the preparation thereof and the use thereof as medicaments |
| WO2009043507A3 (en) * | 2007-09-11 | 2009-05-22 | Mondobiotech Lab Ag | Use of a peptide as a therapeutic agent |
| EP2214676A4 (en) * | 2007-10-29 | 2011-08-03 | Merck Sharp & Dohme | OXININE RECEPTOR ANTAGONISTS SUBSTITUTED DIAZEPAN TYPE |
| US8362009B2 (en) | 2007-10-29 | 2013-01-29 | Merck Sharp & Dohme Corp. | Substituted diazepan orexin receptor antagonists |
| WO2009150614A1 (en) * | 2008-06-11 | 2009-12-17 | Actelion Pharmaceuticals Ltd | Tetrazole compounds as orexin receptor antagonists |
| WO2010003624A2 (en) | 2008-07-09 | 2010-01-14 | Sanofi-Aventis | Heterocyclic compounds, processes for their preparation, medicaments comprising these compounds, and the use thereof |
| JP2012505263A (en) * | 2008-10-14 | 2012-03-01 | アクテリオン ファーマシューティカルズ リミテッド | Phenethylamide derivatives and their heterocyclyl analogs |
| WO2010044054A1 (en) * | 2008-10-14 | 2010-04-22 | Actelion Pharmaceuticals Ltd | Phenethylamide derivatives and their heterocyclic analogues |
| WO2010068601A1 (en) | 2008-12-08 | 2010-06-17 | Sanofi-Aventis | A crystalline heteroaromatic fluoroglycoside hydrate, processes for making, methods of use and pharmaceutical compositions thereof |
| WO2011023754A1 (en) | 2009-08-26 | 2011-03-03 | Sanofi-Aventis | Novel crystalline heteroaromatic fluoroglycoside hydrates, pharmaceuticals comprising these compounds and their use |
| WO2012120057A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Novel substituted phenyl-oxathiazine derivatives, method for producing them, drugs containing said compounds and the use thereof |
| WO2012120055A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Di- and tri-substituted oxathiazine derivates, method for the production thereof, use thereof as medicine and drug containing said derivatives and use thereof |
| WO2012120056A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Tetrasubstituted oxathiazine derivatives, method for producing them, their use as medicine and drug containing said derivatives and the use thereof |
| WO2012120058A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Oxathiazine derivatives which are substituted with benzyl or heteromethylene groups, method for producing them, their use as medicine and drug containing said derivatives and the use thereof |
| WO2012120051A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Benzyl-oxathiazine derivates substituted with adamantane or noradamantane, medicaments containing said compounds and use thereof |
| WO2012120052A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Oxathiazine derivatives substituted with carbocycles or heterocycles, method for producing same, drugs containing said compounds, and use thereof |
| WO2012120054A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Di- and tri-substituted oxathiazine derivates, method for the production thereof, use thereof as medicine and drug containing said derivatives and use thereof |
| WO2012120053A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Branched oxathiazine derivatives, method for the production thereof, use thereof as medicine and drug containing said derivatives and use thereof |
| WO2012120050A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Novel substituted phenyl-oxathiazine derivatives, method for producing them, drugs containing said compounds and the use thereof |
| US9156819B2 (en) | 2011-10-19 | 2015-10-13 | Merck Sharp & Dohme Corp. | 2-pyridyloxy-4-nitrile orexin receptor antagonists |
| US9440982B2 (en) | 2012-02-07 | 2016-09-13 | Eolas Therapeutics, Inc. | Substituted prolines/piperidines as orexin receptor antagonists |
| US9499517B2 (en) | 2012-02-07 | 2016-11-22 | Eolas Therapeutics, Inc. | Substituted prolines / piperidines as orexin receptor antagonists |
| US9896452B2 (en) | 2012-02-07 | 2018-02-20 | Eolas Therapeutics, Inc. | Substituted prolines/piperidines as orexin receptor antagonists |
| WO2014047707A1 (en) | 2012-09-28 | 2014-04-03 | Cristália Produtos Químicos Farmacêuticos Ltda | Heteroaromatic compounds, method for preparing the compounds, pharmaceutical compositions, uses and method for treating acute and chronic pain |
| US10221170B2 (en) | 2014-08-13 | 2019-03-05 | Eolas Therapeutics, Inc. | Difluoropyrrolidines as orexin receptor modulators |
| US11434236B2 (en) | 2016-02-12 | 2022-09-06 | Astrazeneca Ab | Halo-substituted piperidines as orexin receptor modulators |
| US12084437B2 (en) | 2016-02-12 | 2024-09-10 | Astrazeneca Ab | Halo-substituted piperidines as orexin receptor modulators |
| US10894789B2 (en) | 2016-02-12 | 2021-01-19 | Astrazeneca Ab | Halo-substituted piperidines as orexin receptor modulators |
| WO2017194548A1 (en) | 2016-05-10 | 2017-11-16 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods and pharmaceutical compositions for the treatment of autoimmune inflammatory diseases |
| US12036210B2 (en) | 2017-09-13 | 2024-07-16 | Atrogi Ab | Heteroaryl substituted beta-hydroxyethylamines for use in treating hyperglycaemia |
| US11427539B2 (en) | 2017-09-13 | 2022-08-30 | Atrogi Ab | Beta-hydroxy heterocyclic amines and their use in the treatment of hyperglycaemia |
| US11357757B2 (en) | 2017-09-13 | 2022-06-14 | Atrogi Ab | Heteroaryl substituted beta-hydroxyethylamines for use in treating hyperglycaemia |
| US11648216B2 (en) | 2017-09-13 | 2023-05-16 | Atrogi Ab | Fluorophenyl beta-hydroxyethylamines and their use in the treatment of hyperglycaemia |
| US11793774B2 (en) | 2017-09-13 | 2023-10-24 | Atrogi Ab | Chiral beta-hydroxyethylamines and their use in the treatment of hyperglycemia |
| US12280023B2 (en) | 2017-09-13 | 2025-04-22 | Atrogi Ab | Fluorophenyl beta-hydroxyethylamines and their use in the treatment of hyperglycaemia |
| CN113784961A (en) * | 2019-03-20 | 2021-12-10 | 阿托基公司 | Heteroaryl (heterocyclyl) carbinol compounds useful for treating hyperglycemia |
| TWI851687B (en) * | 2019-03-20 | 2024-08-11 | 瑞典商阿特羅吉有限公司 | Heteroaryl(heterocyclyl)methanol compounds useful in the treatment of hyperglycaemia |
| WO2020188299A1 (en) * | 2019-03-20 | 2020-09-24 | Atrogi Ab | Heteroaryl(heterocyclyl)methanol compounds useful in the treatment of hyperglycaemia |
| US12378216B2 (en) | 2019-03-20 | 2025-08-05 | Atrogi Ab | Heteroaryl(heterocyclyl)methanol compounds useful in the treatment of hyperglycaemia |
Also Published As
| Publication number | Publication date |
|---|---|
| EP1718632A1 (en) | 2006-11-08 |
| HUP0400405A3 (en) | 2009-03-30 |
| JP4825686B2 (en) | 2011-11-30 |
| JP2007522197A (en) | 2007-08-09 |
| HUP0400405A2 (en) | 2005-09-28 |
| WO2005075458A8 (en) | 2006-01-19 |
| US20070043037A1 (en) | 2007-02-22 |
| EP1718632B1 (en) | 2009-04-22 |
| US7812031B2 (en) | 2010-10-12 |
| DE602005014086D1 (en) | 2009-06-04 |
| HU0400405D0 (en) | 2004-04-28 |
| ATE429427T1 (en) | 2009-05-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2005075458A1 (en) | Pyrimidine derivatives as orexin receptors antagonists | |
| JP4980928B2 (en) | G protein-coupled receptor (GPR116) agonist and use thereof for the treatment of obesity and diabetes | |
| KR100985419B1 (en) | 2,3,6-trisubstituted-4-pyrimidone derivatives | |
| US20120157433A1 (en) | Heteroaryl Compounds as Kinase Inhibitors | |
| CN101466667A (en) | Azetidine derivatives as g-protein coupled receptor (GPR119) agonists | |
| CA2199621A1 (en) | Substituted aryl piperazines as neurokinin antagonists | |
| JP2008303217A (en) | Adenosine A2a receptor antagonist | |
| JP2012211204A (en) | Gpcr agonist | |
| MX2007008402A (en) | Substituted pyridinyl and pyrimidinyl derivatives as modulators of metabolism and the treatment of disorders related thereto. | |
| JP2011527331A (en) | Piperidinyl GPCR agonist | |
| NZ551673A (en) | 3-Aminocyclopentanecarboxamides as modulators of chemokine receptors | |
| JP3344997B2 (en) | Muscarinic antagonist | |
| CN114728170B (en) | Compounds active on nuclear receptors | |
| EA011487B1 (en) | 3-aminocyclopentanecarboxamides as modulators of chemokine receptors | |
| WO2001087849A2 (en) | Modulators of tnf- alpha signaling | |
| JP2013502448A (en) | Piperidine derivatives used as orexin antagonists | |
| AU2001259691A1 (en) | Modulators of TNF-alpha signaling | |
| CN101123964A (en) | G-protein coupled receptor (GPR116) agonists and their use for the treatment of obesity and diabetes | |
| KR20070007341A (en) | N-piperidine derivatives as Cr3 modulators | |
| US7919508B2 (en) | 3-piperidinylisochroman-5-ols as dopamine agonists | |
| US8003803B2 (en) | Fused ring thrombin receptor antagonists | |
| CA2527851A1 (en) | Hepatitis c virus inhibitors | |
| WO2009058919A1 (en) | Ccr5 antagonists as therapeutic agents | |
| WO2006013393A2 (en) | Quinoline derivatives as neurokinin receptor antagonists | |
| US20090143429A1 (en) | Quinoline Derivatives as Neurokinin Receptor Antagonists |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BW BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE EG ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NA NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SY TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): GM KE LS MW MZ NA SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LT LU MC NL PL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| CFP | Corrected version of a pamphlet front page | ||
| CR1 | Correction of entry in section i |
Free format text: IN PCT GAZETTE 33/2005 UNDER (81) ADD "SM" |
|
| DPEN | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed from 20040101) | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2005708501 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2006552698 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 11463825 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWW | Wipo information: withdrawn in national office |
Country of ref document: DE |
|
| WWP | Wipo information: published in national office |
Ref document number: 2005708501 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 11463825 Country of ref document: US |